Leveraging the ingenuity of carbon nanomaterials towards water quality amelioration
Malini S.
Bapat
 *a,
Hema
Singh
*a,
Hema
Singh
 b,
Shankar B.
Chaudhari
b,
Shankar B.
Chaudhari
 c,
Suresh
Shendage
c,
Raj
Kumar
c,
Suresh
Shendage
c,
Raj
Kumar
 d,
Arun
Karnwal
d,
Arun
Karnwal
 e and
Deepak
Kumar
e and
Deepak
Kumar
 *f
*f
aMKSSS's Cummins College of Engineering for Women, Karvenagar, Pune, Affiliated to Savitribai Phule Pune University, India. E-mail: malini.bapat@cumminscollege.in
bDepartment of Physics, Defence Institute of Advanced Technology, Girinagar, Pune-411025, India
cDepartment of Chemistry, KET'S V. G. Vaze College of Arts, Science and Commerce (Autonomous), Affiliated with University of Mumbai, Mulund, 400081, Maharashtra, India
dSchool of Health Sciences & Technology, UPES University, Dehradun, Uttarakhand, India
eDepartment of Microbiology, Graphic Era (Deemed to be University), Dehradun, Uttarakhand, India
fDepartment of Chemistry, Manipal University Jaipur, Jaipur, India. E-mail: deepak.sharma99967@gmail.com; deepak.kumar@jaipur.manipal.edu
First published on 1st October 2025
Abstract
In this review, carbon nanomaterials are evaluated for their application in the removal of major kinds of contaminants present in polluted waters. A comprehensive examination of their structure, properties, and synthesis pathways is done, and their action in pristine/composite form(s) for water remediation is explored. As water is the essence of life, the upkeep of its quality is imperative for the health and development of all living beings. With rapid urbanization, climate change, population growth and unsustainable practices, the availability and quality of water have been largely compromised. Carbonaceous nanomaterials are fascinating and possess exceptional properties with great potential for use in water quality amelioration. As per the UN Sustainable Development Goals (SDG 6 in particular), there is a need to design the right strategies for managing current water resources and making safe and affordable drinking water globally available. For recycling as well as treating wastewater for reuse, technological developments in material design are crucial as these form the basis of the water remediation process. This review examines the unique features of carbon nanomaterials and their engineering for water purification. Designing materials for filtration and water quality monitoring methods needs to be cohesive and must consider the environmental, social, financial and health aspects of the society. Therefore, this article details the structural features, preparation schemes, the properties of carbon materials and their mechanism of removal of impurities, such as heavy metals, VOCs, oils, dyes, alkyl halides, pharmaceuticals, PFAS and common organic contaminants present in wastewater. The detrimental effects of these contaminants on humans have been highlighted. The challenges and limitations of carbon materials, along with the technoeconomic analysis and future scope encompassing AI assistance, have been considered. This study presents an overview of the qualities of carbon nanomaterials and reiterates their dedicated action in water remediation.
1 Introduction
The advancements in science and technology have led to significant improvements in industrial production, resulting in higher living standards and sustainable economic growth in the entire world.1 This progress is often referred to as an “ever-flatter world”, indicating the increased interconnectedness of economies and societies around the globe. On the contrary, these positive outcomes have been accompanied by negative consequences for the environment. As industrial production and economic growth have expanded, so have the issues related to pollution. Communities worldwide are grappling with persistent pollution problems, which can have a far-reaching impact on the environment, public health, and quality of life. This dual nature underscores the need for sustainable practices that balance economic development with environmental conservation. A considerable portion of the Earth's crust is covered by water, which plays a crucial role in sustaining natural processes and maintaining the ecological balance. However, with the rapid growth of industries, freshwater resources are becoming increasingly scarce.2 In addition, water bodies such as seas, rivers, and lakes are facing rising pollution levels, primarily due to factors like urbanization and industrialization.3Fig. 1 depicts the availability of freshwater on the planet. As the availability of freshwater continues to decrease, the growing scarcity is raising serious concerns.4 Additionally, safe and clean drinking water is essential for the human health. Recycling, reusing and recovery of wastewater can be a boon, and water reclamation can thus make tons of litres of water available.Water contamination and pollution can have widespread and severe consequences on aquatic ecosystems, disrupt food chains, and degrade habitats. Moreover, polluted water poses serious risks to human health, leading to waterborne diseases, toxins entering the food chain, and long-term health problems.5 According to the WHO report, over one billion people lack access to safe drinking water. Mortality due to drinking water is a major concern and is one of the primary health goals in developing countries. The UNESCO 2021 World Water Development report has declared that about 9 million people die yearly due to poor drinking water.6 According to the SDGs adopted by world leaders in September 2015, a vision has been set for the world to be free of poverty, hunger, disease, and want. According to SDG 6.1, which focuses on water, it is recommended that by 2030, universal and equitable access to safe and affordable drinking water should be ensured for all. As per the WHO, 73% of the world's population were using safely managed drinking water services in 2022, and the rest of the people (more than 2 billion) had water without proper safety services, with most people being largely located in Africa and Asia. Diseases such as cholera, hepatitis A, polio, dysentery, typhoid, etc., result from contaminated water. The management of water in rural, urban, and agricultural areas is crucial; otherwise, hundreds of millions of people will be affected by contaminated and chemically polluted water.7 Thus, it is essential to implement comprehensive strategies for water management, pollution prevention, and sustainable water use. This involves a combination of technological, regulatory, and behavioural measures to ensure that water resources are conserved, protected, and made accessible to all, while also preserving the health and functioning of ecosystems.
Based on the water sources available, identification of contaminants and pollutants needs to be carried out. Physical removal of contaminants as well as disinfection are to be included. Fig. 2 depicts a general outline of the control measures, including pre-treatment, coagulation, flocculation, sedimentation, filtration, and disinfection carried out for wastewater treatment.
Water treatment involves a series of steps, and the water purification procedures involve filtration processes and disinfection prior to household distribution. The water from this process is passed through rapid gravity filters to remove any remaining suspended solids. After filtration, the water undergoes further treatment stages, which include additional oxidation and filtration (particularly for manganese removal), ozonation, and/or treatment with GAC. These steps are aimed at eliminating pesticides and certain trace organic compounds. Finally, the water is disinfected before it is released into the supply system.
There is, thus, an increasing demand for advanced water treatment, which has stimulated an intensive exploration of materials and technologies being developed by researchers. There are several techniques used presently for water purification, each with its own merits and weaknesses. The choice of technique often depends on the particular type of contaminant present in water from a source, the required level of filtration, and the available resources. Most of these techniques work on physical separation for the removal of solid contaminants based on size, charge, and adsorption capacity, as seen in Fig. 3.
The physical barrier used in many of these methods must be advanced materials that can efficiently remove the contaminants present in wastewater. The prevalent water cleansing techniques include mechanical filtration, activated carbon filtration, ion exchange, reverse osmosis, distillation, EDI, nanofiltration, membrane-based filtration, electrodialysis, UV disinfection, ozone treatment, biological filtration, and so on.8
1.1 Common methods adopted for water cleaning
1.2 Major types of water contaminants present in water and wastewater
To determine water quality, standard tests and procedures are advocated. The contaminants/impurity levels are expressed in ppm of mg L−1. The preliminary tests include checking alkalinity, hardness, acidity, BOD, COD, TDS, salinity, etc. Currently, ∼20% of the world's water is used by industries. Most industries, including chemicals, dyes, food manufacturing, beverages, textiles, pharmaceuticals, mining, oil refining, metal processing, and others like electroplating, tanning, printing, heating and cooling operations, and so on, release voluminous industrial effluents that contain heavy metals such as Pb, Cd, Cu, As, Ni, Cr, Zn and Hg, etc. Heavy metals are non-biodegradable, which means that they do not naturally break down over time, and they persist in the environment. These metals are known for their high levels of toxicity, and the discharge of these metals into water bodies is a threat to waterways on a global scale. Overall, there is an urgent need to address the issue of heavy metal contamination in waterways resulting from industrial activities.9Table 1 presents the heavy metal limits permissible in water and the associated health hazards.| Heavy metal with max. permissible limit | Health hazards for human beings |
|---|---|
| Arsenic (As)-0.01 ppm | - Gastrointestinal symptoms, cardiovascular and CNS disturbances, and even death in severe cases |
| - Bone marrow depression | |
| - Haemolysis (destruction of red blood cells) | |
| - Hepatomegaly (enlarged liver) | |
| - Melanosis (skin darkening) | |
| - Polyneuropathy (nerve damage) | |
| Zinc (Zn)-3.0 ppm | - Short-term illness and tolerance |
| Chromium (Cr)-0.05 ppm | - Nausea, vomiting, diarrhoea, and allergic dermatitis |
| Lead (Pb)-0.01 ppm | - In general, headache and fatigue |
| - Irritability, memory loss, difficulty concentrating, muscle weakness, seizures (in severe cases) | |
| - Abdominal pain, nausea, vomiting, constipation | |
| - Anaemia, pallor | |
| - Kidney dysfunction, increased urination | |
| - Learning & behavioral problems, developmental delays | |
| Mercury (Hg)-0.006 ppm | Acute exposure |
| -Lung damage, cough | |
| -Chest pain, shortness of breath | |
| -Nausea, vomiting, diarrhoea | |
| -Fever, weakness, metallic taste in the mouth | |
| Chronic exposure | |
| - Tremors, memory loss, difficulty concentrating | |
| - Irritability, mood swings, depression, anxiety | |
| - Excessive salivation, gum inflammation | |
| Copper (Cu)-2.0 ppm | - Liver damage, nausea, cramps, toxic to microorganisms if greater than 0.1 mg L−1 |
| Cadmium (Cd)-0.01 ppm | - Kidney damage |
| - Long-term exposure causes skeletal damage | |
| - Carcinogenic |
Some other inorganic pollutants can include minerals, salts, other metals, and compounds in the form of nitrates, sulphates, phosphates, ammonia, etc.11 Similarly, industries also discharge organic-based pollutants such as textile dyes, polyaromatic hydrocarbons, pharmaceuticals, active compounds, aliphatic and aromatic hydrocarbons, synthetic pesticides, detergents, food additives, insecticides, paints, synthetic fibres, plastics, solvents, and VOCs. The VOCs commonly detected in drinking water include formaldehyde (found in building and household products), benzene (a constituent of gasoline), trichloroethylene (a chemical used in septic system cleaners), and tetrachloroethylene (C2Cl4, used in the dry-cleaning industry). The major ECs are artificially prepared or naturally occurring chemicals, or they may include any microorganism that enters the water bodies and causes ecological or human health effects, as depicted in Fig. 4(a). ECs are categorized into groups such as pharmaceuticals and personal care products, surfactants, fire retardants, nanotechnology-related materials, plasticizers, and pesticides.12 Several ECs have been identified as endocrine-disrupting compounds because of their harmful effects on the endocrine system. EDCs are primarily derived from the sources as seen in Fig. 4(b). Used water from localities often contains EDCs such as oestrogen or certain phenolic compounds that interfere with the body's hormonal system. These chemicals, even in low concentrations (nanograms per liter), have been shown to exhibit noticeable hormonal effects. Natural steroid hormones like 17-estradiol and synthetic hormones such as EE2 are found in sewage. Additionally, chemicals like BPA and NP, which have weak estrogen-like properties, are commonly found in large quantities in water bodies.13 These contaminants are a growing concern for both human health and environmental safety, as they can disrupt the normal functioning of hormones in humans and wildlife.14
 | ||
| Fig. 4 (a) Effects of emerging contaminants on human beings. Reproduced from ref. 13 with permission from Elsevier, copyright [2022]. (b) Different classes and sources of endocrine disruptor compounds. (c) Permissible limits of VOCs by the WHO in ppm. (d) Microplastics detected in various industries and their surrounding areas. Reproduced from ref. 17 with permission from Elsevier Ltd, copyright [2023]. | ||
VOCs evaporate in the air and can be solubilized in water, thus affecting its quality. Fig. 4(c) depicts the permissible limits allowed by WHO. VOCs are present in paints, varnishes, cleaners, and various industrial products. They can change the smell and taste of water and also affect the health of children, the weak, and the elderly. Long-term exposure could lead to chronic conditions affecting the lungs, kidneys, and the CNS, and could also increase the risk of cancer. Anaemia, spleen damage, changes in the blood, changes in the adrenal glands, and nervous system damage are some other harmful effects.15 Thus, to mitigate the risks involved, water needs to be tested regularly for such contaminants. Filtration systems help mitigate the risks and ensure safer drinking water. Microplastics are the minuscule plastic fragments formed from decomposing plastic bottles and materials such as bags, which are present nearly everywhere, particularly in oceans, lakes, rivers, and aquatic animals. Global studies on tap water have detected that 83% of samples have microplastics in them; thus, it is a matter of serious concern. Given that more than 7 billion tons of plastic have been produced on earth up to the year 2015, and is further broken down into small-sized debris, it is quite likely that it ends up on the land and water around us. When plastic degrades to particles smaller than 5 mm in size, they are referred to as microplastics, and if they are smaller in diameter than 100 nm, they are called nanoplastics. Globally, 91% of the surface water contains microplastics having a size range that lies between 20–100 μm, 8% between 100–300 μm, and the rest of it being greater than 300 μm; composition-wise, these are made up of polyethylene roundings. Polyester, polyamide, polypropylene, polyacrylamide, polyurethane, polycarbonate, and polyethylene terephthalate are quite common.16 The microplastics can persist in the environment over long periods of time due to both environmental factors and their inherent characteristics, such as their hydrophobic nature, being low-density, fibrous, and recalcitrant. Fig. 4(d) depicts the considerable presence of microplastics in various water zones in our surroundings.17 In 2022, Cherniak et al.18 showed that microplastics can be removed at drinking water treatment plants by combining coagulation with aluminium hydroxide, flocculation, anthracite-sand filtration, and chlorination. The conventional water treatment showed removal efficiency between 40% and 55%, especially with respect to plastic fibres, while the granular activated carbon particles were efficient in the removal of 56–61% of microplastics. The unhealthy additives found in the microplastics have also been associated with cancer and EDCs. Researchers from Columbia University, in a recent study in January 2024, published their findings of microplastics, namely PVC, PS, PET, etc., present in three undisclosed popular drinking water brands sold in the US. It was disclosed by researchers that the plastic bits lie in the range of 11–40 lakhs per litre of bottled water. Nanoplastics are the most worrisome type of plastic pollution since they show heterogeneity in their size, morphology, and chemical composition.19 Further, the minuscule particles can cross the biological barriers and destroy individual cells and tissues in major organs, thereby disrupting cellular processes. They can deposit EDCs like perfluorinated and polyfluorinated substances such as PFAS, BPA, plasticizers like phthalates, heavy metals, flame retardant chemicals, etc. Also, 100 nm sized plastic materials are difficult to detect by microscopy. Among all those mentioned above, the poly- and per-fluoroalkyl substances are a group of persistent organic pollutants that exist indefinitely in the environment due to the strong C–F bond, which makes them resistant to attack by heat, alkali, and acid. The PFAS have a plethora of industrial applications, such as plastics, non-stick cookware, aqueous film-forming foams, and semiconductor manufacturing. In 2016, the US EPA issued a drinking water advisory level for PFOA and PFOS of 70 ng L−1. This rule enforces mandatory monitoring by 2027 and compliance by 2029.20a Nanofiltration, as well as reverse osmosis, are the leading technologies for PFAS removal owing to their high extraction efficiency. However, high costs of operation, energy requirement, sophistication, pre-treatment, etc., impose constraints. Landfills, a common waste disposal method, generate harmful leachates that pollute the groundwater and surface water. Thus, sustainable methods are required to address the PFAS contamination. The article by Tushar and Rowles et al. presents a comprehensive overview of sustainable PFAS treatment technologies.20b
To summarize, in the above sections, we have discussed the importance of clean water, major contaminants polluting water bodies, and the common methods adopted for improvement in water quality.
1.3 Water filtration methods
Wastewater filtration is essential for maintaining the ecological balance and preventing contamination of natural water sources. The process involves the removal of suspended particles, pollutants, and biological impurities from wastewater to render it safe for discharge into water bodies or for reuse. This process is indispensable for maintaining ecological balance and preventing the destruction of natural water sources. There are various types of wastewater filtration processes, such as physical filtration,21 biological filtration,22 chemical filtration,23 membrane filtration,24 and nanoparticle-based filtration.25 Physical filtration involves screening, which removes larger debris and solids from wastewater using physical barriers, while microfiltration uses porous membranes to separate suspended particles as small as one micron. Ultrafiltration uses fine membranes to eliminate particles as small as 0.01 microns, making it highly efficient in removing colloidal particles and macromolecules. Fig. 5 shows the various filtration levels for the removal of various sizes of impurities present in water. | ||
| Fig. 5 Filtration levels showing the removal of different-sized impurities from raw water in relation to applied pressure. | ||
Amongst the membrane-based technologies, RO is widely commercialized; however, it requires high energy and, thereby, incurs significant operational costs, which limit its broader applications.37 Polymeric materials have garnered significant research attention in the development of membrane technologies for water purification. These membranes offer several advantages: they are energy-efficient, easily scalable, and time-saving. They also demonstrate high water permeability and structural stability. Moreover, polymeric membranes exhibit excellent water selectivity, strong solute rejection even at low operational pressures, and high resistance to both oxidation and fouling. Polymeric membranes are often made from carbon-based polymers such as PVDF, PAN, PVA, PVC, PE/PP/polyamide, polysulfones, chitosan, etc. Several methods are employed in the fabrication of these membranes, including electrospinning,38 track-etching, vapor deposition, sol–gel processes, stretching, phase inversion, and interfacial polymerization.39 TFC membranes, which are critical for the commercialization of reverse osmosis and nanofiltration processes, are typically produced using IP technology. These membranes often feature a polyamide thin layer on top of a support material. The examples of monomers commonly used to create the polyamide thin layers are m-phenylenediamine and trimesoyl chloride. These membranes are effective in desalination applications. Fig. 6 depicts a general schematic, visualising the creation of a TFC membrane using DAP (2,6-diaminopyridine) and an amine on a PES support, followed by the addition trimesoyl chloride to undergo interfacial polymerization. In this image, an MPD (meta-phenylenediamine)-based membrane has been redesigned into a DAP-based membrane with structural superiority, having a robust structure and high chemical and photochemical stability with pH tolerance, which are useful for industrial applications.40
 | ||
| Fig. 6 Schematic representing a thin film composite membrane preparation by interfacial polymerization. Reproduced from ref. 40 under Creative Commons CC-BY, copyright [2025]. | ||
Membrane properties such as molecular structure, crystallinity, hydrophobicity/hydrophilicity, surface charge, and smoothness play a significant role in determining the permeate flux and overall performance in filtration processes.41,42 Polymer-based membranes, while commonly used, face challenges such as fouling, which results from the accumulation of surfactants, pore clogging, and structural deterioration after extended use.43 The fouling behaviour depends on the membrane's surface characteristics, including porosity, hydrophobicity, pore size, and morphology.44 Hydrophilic membranes tend to have better antifouling properties than hydrophobic ones. Membranes that recover higher permeate flux are typically more resistant to fouling. For hydrophobic membranes, surface roughness and a low surface energy are crucial factors that can be achieved through specific surface treatments. Incorporating nanomaterials into membranes can enhance their surface roughness, leading to the development of polymer nanocomposite membranes.45 When nanomaterials are mixed with polymers, they form nanocomposites with a multiphase structure, where at least one phase has nanoscale dimensions. It is well known that nanomaterials offer a range of beneficial properties, such as a large surface area for adsorption, unique surface chemistry, photocatalytic activity, antimicrobial effects, and various electrical and optical characteristics, all of which contribute to improved membrane performance. Various nanomaterials, such as organic and inorganic compounds (e.g., SiO2, TiO2, Ag, Fe, and zeolites), are used to modify polymer membranes.46 These materials impact the membrane's performance by influencing factors like solute selectivity and stability. Among these, carbon-based nanomaterials are particularly important for improving membrane properties. Several approaches can enhance membrane hydrophilicity and antifouling capabilities, such as coating,47 grafting,48 blending,49 or incorporating hydrophilic polymers and monomers into the membrane.50 In comparison to all these materials, it is important to note that carbon-based materials are environmentally friendly, exhibit low toxicity, and have simple preparation methods. They can exist in 0D, 1D, 2D, and 3D forms, are abundantly available, easy to functionalize, and possess excellent biocompatibility (Fig. 7(a)). Carbon-based materials contain carbon atoms bonded in special ways so that they exist in various morphologies such as spheres, sheets, ellipsoids, nanoribbons, and hollow tubes. Fullerenes, CNTs, carbon nanofibers, C black, graphene, nanodiamonds, C-dots, carbon onions, graphdiynes, etc., have different bonding, resulting in remarkable structures, and are well-known carbon forms. Based on a Scopus search, Fig. 7(b) depicts the research on wastewater treatment and purification, which was observed to be boosted from 2010 onwards (Trend 10 years). The growth from 2010 to 2020 is 10-fold and has doubled in the last five years.
 | ||
| Fig. 7 (a) Versatile nature of carbon materials that makes them valuable for water remediation. (b) Publication trend related to water treatment from 2010 (Science Direct database). (c) Trends in publications related to carbon-based technologies to treat pollutants and ECs in wastewater from the year 1950 to 2020. Reproduced from ref. 51 with permission from Elsevier, copyright [2023]. | ||
Fig. 7(c) shows that there has been an increase in publications utilizing carbon materials developed for wastewater treatment. The incorporation of carbon nanomaterials enables the optimization of pore size, increased surface area, and unique surface functionalities, thereby enhancing water permeability, thermomechanical stability, hydrophilicity, and antifouling properties in polymer-based nanocomposite membranes.52 Since all forms of carbon nanomaterials are biocompatible, they have numerous possibilities for both biomedical applications and water treatment.53 Detailed discussions on surface functionalization, interactions between carbon nanomaterials and polymers, as well as methods to enhance the carbon–polymer interface for bio-environmental applications, are needed, which are quite possible.54 The antibacterial and photocatalytic properties of carbon nanomaterials further improve membrane functionality. Thus, a major challenge in membrane research is improving the characteristics of carbon–polymer nanocomposite membranes, such as permeability, rejection, stability, and antifouling. This requires careful characterization of the carbon and carbon–polymer interfaces during loading, as well as an evaluation of load transfer efficiency in the nanocomposites. For water purification techniques, various C nanoforms such as 0D-fullerenes, quantum dots, 1-D such as CNTs, nanofibers, and rods, have been tested. The 2D carbon nanoforms, which include graphene, graphene oxide, etc., as well as mesoporous carbon forms, have been used over the period. Of all these, the 0D nanomaterials offer enhancement of the properties of polymer membranes due to their small size, morphology, and tunability, thus making the polymer smooth with better functionality and anti-fouling behaviour. The 0D carbon nanomaterials contribute to a smoother membrane surface, optimal pore size, enhanced permeability in thin-film nanocomposite membranes with increased surface hydrophilicity, resulting in higher water flux in composites. Biomass-derived carbon nanostructures in the form of sustainable porous spheres and high surface area fibers are also capable of adsorbing impurities present in water.55,56 The key advantage of the 0D carbon forms lies in their unique physical and surface properties, which facilitate the development of high-performance carbon-incorporated polymer membranes with a wide range of polymer types.
Hence, in this context, the review takes on the discussion of carbon nanomaterials at the forefront, which are uniquely suited for application in water purification systems for eliminating major pollutants and contaminants present in water. Significant efforts are being made by researchers to get together innovative, energy-efficient, and cost-effective wastewater treatment methods utilising novel materials and methods, which will be discussed further. Due to the remarkable properties of carbon nanomaterials, such as large surface area, unique morphologies, tunable porosity, biocompatibility, high strength, and remarkable adsorption properties, they are well-suited for application in water remediation either as individual materials or in a composite form. This review evaluates the strengths and limitations, and elaborates on the mechanism of carbon materials in their performance towards the removal of a variety of contaminants commonly present in water, such as heavy metals, pharmaceuticals, pesticides, endocrine-disrupting molecules, microplastics, PFAS, and others. The review aims to inspire the application of advanced materials, such as graphene, nanocellulose, CNTs, activated carbon, C quantum dots, graphynes, and C60, in energy-efficient, economical, and sustainable solutions to provide clean water. Having the desired properties and capabilities, the carbon nanomaterials truly stand out as significant materials in meeting this challenge. Thus, this article does not just comment on their success but also provides insight into the hurdles such as costs, scalability, potential toxicity, lack of regulatory framework, and limitations in real-world applications. It provides an overview of the use of carbon nanomaterials for water quality amelioration, as well as points out the sustainable and technological future scope.
2 Carbon-based nanomaterials for water treatment
Nanoparticle-based filtration is gaining attention in wastewater treatment for its potential to address water pollution challenges. Nanomaterials, like iron-based59 or carbon-based nanoparticles,60 offer unique properties and reactivity, enabling efficient pollutant removal. They are promising for applications like oil recovery and COD. Nanoparticles offer improved removal efficiencies, enhanced remediation techniques, and reduced environmental impacts compared to traditional methods. Carbon is among the top 5 most abundant elements in the universe. It forms bonds for ‘life’ with C, H, N, and other elements. Four of its electrons can participate in bond formation, which are strong covalent bonds, and double and triple bond formation is also possible. Thus, an unsurpassable number of varied stable structures are formed by carbon, such as the crystalline structures, the hardest material diamond, the layered structure of graphite, soft soot, bucky balls of fullerene, strong carbon nanotubes, the monoatomic honeycomb-structured layers of graphene, and highly activated carbon. Carbon nanomaterials are unique carbon forms with nanoscale dimensions, exhibiting extraordinary properties that are not found in bulk materials. These dimensions are in the range of one to a few hundred nanometers.61 These are frontier materials used in science and technology due to their unique properties and diverse applications. Carbon nanomaterials include various carbon allotropes, including amorphous carbon, graphite, diamonds, CNTs, and graphene oxide.62Fig. 8 shows a variety of carbon allotropes in 0D, 1D, 2D, and 3D morphologies. They attract the interest of researchers and companies all over the world due to their potential mechanical, electrical, and thermal properties, and have a major impact on industries like electronics, energy, medicine, environmental protection, and many more.63 To summarize, carbon-based nanomaterials with their unique structures and interactive properties with contaminants are well-suited for water purification.2.1 Carbon nanotubes (CNTs)
CNTs are a lightweight, robust 1-D carbon allotrope with exceptional mechanical, thermal, and electrical properties.64 They have a crystalline structure and consist of rolled-up graphene sheets, forming seamless cylinders with hexagonal lattices.65 The tubular structure with hexagonal lattice arrangements of CNT provides exceptional mechanical and electronic properties. CNTs are widely used across various fields, including biomedical, catalyst support, field emission, thermal conductivity, energy storage, molecular electronics, structural applications, and filtration.66 Single-walled carbon nanotubes (SWCNTs) and multi-walled carbon nanotubes (MWCNTs) are the primary types of carbon nanotubes, having a hexagonal honeycomb structure with varying properties. SWCNTs are single carbon atoms in cylindrical tubes, with diameters ranging from 0.7 to 2 nanometers. They have good mechanical, thermal, and electrical properties, making them strong, lightweight, and good conductors of heat and electricity.67 MWCNTs are made up of multiple layers of carbon atoms arranged in concentric cylindrical tubes, with diameters ranging from several nanometers to 100 nanometers.68 They exhibit remarkable mechanical and electrical properties, with a layered structure providing additional strength and thermal stability. MWCNTs are used in similar applications as SWCNTs, but their larger diameters make them suitable for certain mechanical and reinforcement applications. CNTs are corrosion-resistant and highly elastic, allowing them to bend and deform without breaking, making them suitable for applications requiring flexibility and resilience. Their large surface area makes them suitable for energy storage, drug delivery, and catalysis. The most utilized methods for CNT synthesis are (i) chemical vapor deposition,69 (ii) arc discharge, and (iii) laser ablation.70 The arc discharge method, first used in 1991 by Sumio Iijima, is a crucial technique for synthesizing carbon nanotubes.71 An electric arc was produced between the cathode and anode in an inert gas environment. The process generates high temperatures, causing carbon precursors to sublimate and form CNTs. The method can produce both multi-walled and single-walled CNTs, depending on the configuration of the electrodes. However, it has limitations, such as producing short tubes of random sizes and a higher defect rate. Low-temperature preparative methods like CVD are popular, but the arc discharge method remains a significant method in CNT synthesis research and continues to advance carbon nanotube technology. The CVD method is a common technique for the controlled growth of CNTs due to its simplicity, scalability, and potential for large-scale production.72 The process involves introducing carbon-containing precursor gases into a reactor with a catalyst material, which is heated to high temperatures. The catalyst's properties, such as size, distribution, and interaction with carbon species, influence the diameter, structure, and alignment of CNTs. CVD offers precise control over growth parameters, enabling the production of single-walled and multi-walled CNTs with varying properties. The method is being researched and developed to improve yield, purity, and scalability for widespread industrial use. Laser ablation is a widely employed method for the synthesis of CNTs, first used in 1993. It involves the high-intensity laser beam ablation of a carbon-rich feedstock, such as methane or carbon dioxide, in a quartz tube furnace. This process vaporizes carbon atoms, resulting in purer CNTs.73 Laser ablation is popular for SWCNTs due to its high purity. However, it is expensive and may require high-energy lasers. Researchers continue to optimize the laser ablation method, fine-tuning parameters and feedstock to produce CNTs with tailored characteristics for various applications, including electronics, nanosensors, and biomedical devices. Fig. 9(a–g) outlines the common methods for the synthesis of CNTs. | ||
| Fig. 9 (a) Structures of CNTs. Reproduced from ref. 74 with permission from Springer Nature, copyright [2020]. (b) Chemical vapour deposition. Reproduced from ref. 75 with permission from Taylor & Francis, LLC, copyright [2013]. (c) Arc-discharge process. Reproduced from ref. 76 with permission from RSC, copyright [2015]. (d) Laser furnace apparatus. Reproduced from ref. 77 with permission from Springer Nature, copyright [2016]. | ||
CNTs are considered a third-generation carbonaceous adsorbent material due to their unique structural and chemical properties.78Fig. 10 depicts the adsorption sites that CNTs have in their structure, with the potential to capture a variety of impurities as mentioned below.
 | ||
| Fig. 10 Adsorption sites of CNTs: (a) inner holes, (b) interstitial channels, (c) groove sites, and (d) exterior sites to capture the contaminants in wastewater. | ||
(a) Inner CNT holes: the hollow core of individual CNTs provides a significant site for adsorption. Contaminants can be adsorbed into these inner cavities, especially if their diameter is suitable enough to fit inside.
(b) Interstitial channels: these are the spaces between the CNTs when they are bundled together. These channels can trap small contaminants, particularly when the CNTs are organized in a form where these interstitial spaces are well-defined and accessible.
(c) Groove sites: the grooves refer to the indentations or crevices on the outer surface of the CNTs. These can also serve as adsorption sites, particularly for contaminants that are attracted to the specific surface features of the nanotubes.
(d) Exterior sites: the external surfaces of CNTs can adsorb contaminants through diverse interactions, which include the van der Waals forces, π–π interactions, or through functional groups attached to the CNT surface.
CNTs are effective adsorbents for various metal ions, including Cr2+, Cr4+, Ni2+, Sr2+, Pb2+, Zn2+, Cu2+, Cd2+, and Co2+. The factors that affect the sorption of metal ions on CNTs are as follows:79
(1) Ionic radius: larger ions are absorbed less efficiently. The order of various metal ions from largest to smallest is Pb2+ > Sr2+ > Ca2+ > Cd2+ > Mn2+ > Cu2+ > Zn2+ > Co2+ > Ni2+.
(2) Electronegativity: higher electronegativity improves adsorption on CNTs in the order of Pb2+ > Ni2+ > Cu2+ > Co2+ > Cd2+ > Mn2+ > Ca2+.
(3) Stability constant: metals with less soluble hydroxide complexes are better adsorbed.
Log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) K1 values: Pb(OH)+ (7.82) > Cu(OH)+ (7.00) > Zn(OH)+ (4.40) > Cd(OH)+ (4.17).
K1 values: Pb(OH)+ (7.82) > Cu(OH)+ (7.00) > Zn(OH)+ (4.40) > Cd(OH)+ (4.17).
(4) Reduction potential: lower reduction potential means stronger adsorption, in the following order: Pb2+/Pb (−0.1262 V) > Cu2+/Cu (−0.3419 V) > Cd2+/Cd (−0.4030 V) > Zn2+/Zn (−0.4618 V).
The SWCNTs and MWCNTs have both been used for metal removal from wastewater. SWCNTs exhibit a higher adsorption capacity for Cr(VI) ions (2.35 mg g−1) compared to MWCNTs (1.26 mg g−1) at pH 2.5. This indicates that SWCNTs are more effective in chromium adsorption as noted by Dehghani et al.80 K. Pillay et al.81 studied the adsorption efficiency of unfunctionalized and functionalized MWCNTs for chromium from aqueous solution. It was observed that unfunctionalized MWCNTs exhibit superior adsorption capability compared to functionalized MWCNTs, particularly for Cr(VI) ions. Functionalization of MWCNTs typically involves introducing oxygen-containing groups such as carboxyl (–COOH), hydroxyl (–OH), and amine (–NH2) groups and these groups introduce electron-rich sites. Cr(VI) in aqueous solution often exists in the form of negatively charged dichromate ions (Cr2O72− or HCrO4−). The functionalized MWCNTs, which are rich in electron-donating groups, result in the electrostatic repulsion of negatively charged dichromate ions and thus inhibit the adsorption process. An exception to this trend was observed by Atieh et al.,82 who attempted to modify MWCNTs by adding more carbonyl groups and studied Cr(III) removal from water using raw (R-) and COOH-modified (M-) carbon nanotubes (MCNTs). Their results showed that the maximum adsorption capacity increased as the pH was raised from 4 to 7. M-CNTs exhibited both faster and higher adsorption capabilities than R-CNTs, with R-CNTs achieving a maximum adsorption capacity of 0.3718 mg g−1 (7% removal), while M-CNTs achieved 0.5 mg g−1 (10% removal), which highlights the successful modification. However, carbon-based nanomaterials are so small that it is difficult to remove them from filtered water. In order to overcome this limitation, the CNTs are also impregnated with Fe3O4 for their easy and fast recovery from the filtered water [Fig. 11(a)].83 With the aid of a magnetic field, the water pollutants adsorbed on the nanoadsorbents can be successfully recovered. Fig. 11(b) shows the schematic for heavy metal adsorption on magnetic nanocomposites and their removal with a magnet.
 | ||
| Fig. 11 (a) Wastewater treatment using CNTs impregnated with magnetite to obtain purified water. Reproduced from ref. 83 with permission from Springer Nature, copyright [2020]. (b) Heavy metal adsorption on magnetic nanocomposites and their removal with a magnet. Reproduced from ref. 84 with permission from Elsevier copyright [2017]. | ||
In another report, CNTs impregnated with Fe3O4 were modified by adding MnO2 as metal-ion scavengers, which augmented the Cr removal efficacy of MWCNTs. At pH = 2, the nanocomposite (MnO2/Fe3O4/MWCNTs) displayed an adsorption capacity of 186 mg g−1 for Cr(VI). The removal was a consequence of the electrostatic interactions, cation exchange, and the formation of complexes on the surface.85 A chitosan-based gel was prepared by modifying MWCNTs with poly(acrylic acid) and poly(4-amino diphenylamine) by Kim et al. to enhance its chromium adsorption capacity from wastewater.86 Pyrrole polymer was used to wrap oxidized multi-walled carbon nanotubes, which proved to be an effective approach for Cr removal. The synthesized material exhibited a high affinity for chromium ions, with a distribution coefficient (Kd) calculated as 436.7 mg g−1 μM−1, indicating its strong adsorption capacity.87 Another polymer polyamine was used to modify carbon nanotubes to develop polyamine-carbon nanotubes (PA-CNTs) for Cr(VI) remediation with a capacity of 168.54 mg g−1. The carbon nanofibers (CNFs) were also reported to have an adsorption capacity of 227 mg g−1 at equilibrium toward Cr(VI), which is good, but the recyclability is a major issue that needs to be worked upon.88 Recently, Cr(VI) ions have been effectively removed from wastewater using magnetic multiwalled carbon nanotubes (MMWCNTs) in combination with deep eutectic solvents (DES). In this study, a deep eutectic solvent (DES) composed of a hydrogen bond donor (HBD) and a hydrogen bond acceptor (HBA) mixed at specific molar ratios was employed. By optimizing key parameters such as adsorbent dosage, contact time, and adsorption temperature, a Cr(VI) removal efficiency of over 95% was achieved. The adsorption process followed quasi-second-order kinetics and was well described by the Freundlich isotherm model, indicating multilayer adsorption on a heterogeneous surface. Modified multi-walled carbon nanotubes interacted with Cr(VI) through multiple mechanisms, including electrostatic attraction, reduction reactions, and complexation. The green solvent DES, with its extensive hydrogen bonding network, was effectively employed for the desorption of Cr(VI) from MMWCNTs, achieving a desorption efficiency of over 90%. Notably, the adsorbent retained high performance even after five consecutive adsorption–desorption cycles. These results suggest that the novel MMWCNTs, in combination with DES regeneration, offer significant potential for the efficient treatment of Cr(VI)-contaminated wastewater.89 In the literature, there are reports on the removal of As using CNTs. Imran Ali reported the removal of arsenite and arsenate, ∼91% and 92%, respectively, using MWCNTs at pH = 6.0 and 25 °C.90 In another work, Hassan Alijani and Shariatinia91 synthesised a zerovalent iron-doped MWCNT nanohybrid by using natural α-Fe2O3 as a precursor in situ via the CVD method. They reported 96% and 98.5% arsenite and arsenate adsorption onto the nanohybrid. Sankara Ramakrishnan et al. utilized ferrocene, which displayed dual functions: it acted as a floating catalyst in the formation of CNTs and also facilitated arsenic sorption.92 In another study, the carbon nanotubes were decorated with lignin, a biopolymer that was used to prepare a nanocomposite to capture Pb(II) ions from wastewater.93 It demonstrated an adsorption capacity of 235 mg L−1 for Pb(II) ions in wastewater. The enhanced removal efficiency of CNTs is attributed to the presence of several oxygen functionalizations on the surface, which would have enabled the removal of Pb(II) ions through electrostatic forces of attraction. The MWCNTs also exhibited an adsorption capacity of 88.62 mg g−1 and 99 mg g−1 for Cd(II) ions and Cu(II) ions, respectively, at a pH of ∼5 and having a dose of 0.1 g within an hour, with the initial concentration of metal ions being 10 mg L−1.94 Shao D., Hu J., and Wang X. used plasma-induced techniques for grafting chitosan onto MWCNTs for the expulsion of UO22+, Cu2+ (Fig. 12(a)), and Pb2+ from solution. Fig. 12(b) depicts the SEM of chitosan-grafted MWCNT. The resulting adsorption isotherms of UO22+, Cu2+and Pb2+ can be seen in Fig. 12(c) along with the other materials, which depict that MWCNTs grafted with chitosan are better candidates than MWCNTs.95
 | ||
| Fig. 12 (a) Representation of chitosan-grafted MWCNTs for copper adsorption from wastewater. (b) SEM images of chitosan-grafted MWCNTs. (c) Adsorption isotherms of UO22+, Cu2+and Pb2+ from solutions of chitosan-grafted MWCNTs and plasma-treated MWCNTs. Reproduced from ref. 95 with permission from WILEY-VCH, copyright [2010]. | ||
Apart from metal adsorption, SWCNTs and MWCNTs have been used for dye removal from industrial wastewater. Fugetsu et al. (2004) studied Ba2+-alginate-encapsulated MWCNTs for the removal of dyes such as acridine orange, orange-G, ethidium bromide, and eosin, reporting adsorption efficiency values of 0.31, 0.33, 0.43, and 0.44 mmol mg−1, respectively.96 In another work, Ghaedi et al.97 reported the adsorption efficiency of Eriochrome Cyanine R by comparing activated carbon (40.6 mg g−1) and MWCNTs (95.2 mg g−1) at varying parameters such as pH, temperature, and dye concentration, and found that the pH is an important factor for adsorption, and MWCNTs have better adsorption efficiency. Ma et al.98 prepared alkali-activated CNTs, which showed high adsorption efficiencies of 399 and 149 mg g−1 for methyl blue and methyl orange, respectively. Qu's group synthesized MWCNTs decorated with Fe2O3 nanoparticles for the extraction of neutral red and methylene blue from polluted water with the adsorption efficiency of 77.5 and 42.3 mg g−1, respectively.99 Similarly, Gong et al.100 developed magnetic MWCNTs for removing dyes such as neutral red, methylene blue, and brilliant cresyl blue from polluted water. In another study, Mallakpour and Rashidimoghadam developed glutaraldehyde cross-linked PVA/vitamin C–MWCNT (polyvinyl alcohol/vitamin C–multi-walled carbon nanotubes) composite membranes and investigated their efficiency in methylene blue adsorption from wastewater.101 Recently, Deepanraj et al. attached oxygen functional groups to the surface of MWCNTs and found an increase in the percentage of methylene blue adsorption. The membranes showed a high capacity for removing substantial amounts of methylene blue in a short time.102 Additionally, they maintained effectiveness over five cycles without any degradation in performance. Gao et al. prepared a magnetic polymer MWCNT nanocomposite for the elimination of azo dyes from wastewater.103 Zghal et al. studied graphite/CNT composites for the removal of cationic dyes, like Rh B, from dye-contaminated waters. Rhodamine B is a planar molecule that is readily adsorbed onto carbon graphite and carbon nanotube composites. The adsorption is primarily driven by hydrophobic interactions and other weak forces, notably π–π stacking between the dye's aromatic rings and the hexagonal carbon lattice of graphite and CNTs, thus making it a suitable candidate for large-scale environmental applications.104 Zhao and colleagues synthesized carboxyl-rich carbon nanotubes to enable the rapid removal of methyl red dye from aqueous solutions. These functionalized nanotubes demonstrated exceptional efficiency, removing 85% of the dye within just 5 minutes. The availability of abundant carboxyl (COOH) groups on the surface of the carbon nanotubes significantly increased the number of active sites accessible for dye adsorption, enhancing their performance. Moreover, the COOH-rich carbon nanotubes exhibited excellent reusability, maintaining their effectiveness over 10 consecutive cycles, highlighting their potential for sustainable water purification applications.105 In another study, researchers developed a magnetic alginate composite infused with MWCNTs and Fe3O4 nanoparticles (CNT–Alg–Fe3O4) to effectively remove dyes from industrial wastewater. Using response surface methodology with a central composite face-centered design, they optimized parameters such as pH, adsorbent dosage, and contact time. Under optimal conditions (pH = 3, 10 g L−1 adsorbent dose, 85.55 minutes contact time), the composite achieved a dye removal efficiency of 98.43% and an adsorption capacity of 138.97 dye per g. Notably, the adsorbent retained substantial efficacy over ten reuse cycles, with only an 11.94% reduction in performance, highlighting its potential for sustainable wastewater treatment applications.106 Reports indicate the use of CNTs for the removal of pharmaceutical waste. Specifically, MWCNTs have been employed for the removal of acetaminophen using modified –OH and –COO group MWCNTs. The MWCNTs followed the order of adsorption capacities as follows: ozone-treated MWCNTs (250 mg g−1) > chitosan-coated MWCNTs (205 mg g−1) > acid-treated MWCNTs (160 mg g−1) > NaOH-treated MWCNTs (130 mg g−1) > untreated MWCNTs (90 mg g−1).107 In a separate study, sodium hypochlorite-oxidized MWCNTs were assessed for their effectiveness in removing ciprofloxacin.108 A recent study introduced an innovative flow-through electro-Fenton system designed for the efficient degradation of tetracycline, an antibiotic contaminant in water. The system employed carbon nanotubes encapsulating α-Fe2O3 nanoparticles (Fe2O3-in-CNT) as a nanoconfined catalyst. This configuration enhanced the generation of hydrogen peroxide and reactive oxygen species (ROS), particularly singlet oxygen, leading to a 1.65-fold increase in degradation efficiency compared to traditional surface-coated catalysts. Additionally, the flow-through setup achieved degradation kinetics 5.1 times higher than conventional batch reactors. The process also resulted in degradation byproducts with reduced toxicity, offering a promising approach for environmental remediation.109 The CNTs exhibit good adsorptive capabilities for various reasons, such as (i) p–p electron–donor–acceptor interactions, (ii) electrostatic interactions, (iii) intermolecular hydrogen bonding, and (iv) comparable pore size. The first type of interaction mentioned is believed to be the critical driving force for the expulsion of organic pollutants or aromatic dyes. The adsorption of organic compounds with varying physicochemical properties, such as polarity, size, electron polarizability, and hydrophobic nature, onto SWCNTs and MWCNTs has been evaluated in the literature. For a given type of carbon nanotubes, the adsorption affinity showed a poor correlation with hydrophobicity but increased in the following order: nonpolar aliphatic < nonpolar aromatics < nitroaromatics. With respect to various nitroaromatics, the adsorption affinity was observed to increase with an increase in the number of –NO2 functional groups.
Chen and colleagues studied the adsorption behaviour of various aromatic compounds, including cyclohexane, benzene, and different chlorinated and nitro-substituted benzenes, on carbon nanotubes. They found that nitroaromatic compounds strongly adsorb onto the nanotubes, primarily due to π–π electron donor–acceptor interactions, where nitro groups act as electron acceptors and the nanotube's graphene surface functions as an electron donor. Moreover, nonpolar aromatic compounds showed higher adsorption compared to nonpolar aliphatic molecules, which was attributed to π–electron interactions between the planar aromatic rings and the nanotube surface. In the case of the bulky tetrachlorobenzene molecule, single-walled carbon nanotubes demonstrated a notably higher adsorption capacity per surface area than multi-walled nanotubes, suggesting the influence of molecular size and shape in adsorption.110 There have been many reports on the adsorption behaviour of DCB on CNTs; one of the groups investigated a broad pH range of 3–10 and achieved maximum sorption of 30.8 mg g−1 in 40 min.111 Chungsying Lu's group suggested that carbon nanotubes (CNTs) have significant potential for the elimination of trihalomethanes from water, due to their strong adsorption capacity and effective interactions with organic compounds.112,113 Gwendeline K. S. Wong and Richard D. Webster studied 47 trace atmospheric VOCs using GC-MS coupled with thermal desorption experiments. These experiments were performed under active/passive conditions, and comparisons with Carbopack X/Tenax were carried out. The results showed that 29 VOC analytes were adsorbed onto the SWCNT sample tube, and 37 VOC target analytes were identified in the Carbopack X/Tenax tubes. Methacrolein exhibited the highest normalised peak area ratio on the SWCNT sample tube, and the signal response was ∼3.2 times greater than that of the conventional multi-sorbent material used.114 Dioxin, a by-product in water treatment, is always of concern. Long and Yang115 found that carbon nanotubes adsorbed much more dioxin than activated carbon at low concentrations due to strong interactions between dioxin's benzene rings and the nanotube surface. Joseph et al. studied the adsorption of common EDCs, BPA (a commonly used plasticizer), and EE2 (synthetic estrogen) on SWCNTs from brackish waters. Their study revealed that the adsorptive capacity of SWCNTs was higher for EE2 than for BPA, likely due to its higher log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) KOW value, which may enhance its hydrophobic interactions with the SWCNTs.116 In another study, MWCNTs modified with iron oxide and manganese dioxide (MWCNT–Fe3O4–MnO2) were synthesized and subsequently used for the adsorption of BPA. A schematic diagram illustrating the BPA extraction process is presented in Fig. 13(a). Similarly, Fig. 13(b) shows the adsorption capacities of BPA on MWCNT–Fe3O4–MnO2 and other materials. The maximum adsorption capacities were 76.75 mg g−1 for MWCNTs, 44.52 mg g−1 for MWCNT–Fe3O4, and 132.89 mg g−1 for MWCNT–Fe3O4–MnO2. Fig. 13(c) shows the contact time for BPA adsorption onto MWCNT–Fe3O4 and MWCNT–Fe3O4–MnO2. MWCNT–Fe3O4–MnO2 exhibited the optimal contact time, reaching 150 minutes. Overall, MWCNT–Fe3O4–MnO2 proved to be the most efficient adsorbent, which could be due to its enhanced surface area and increased number of binding sites.117 The TEM images shown in Fig. 13(d–f) reveal that MnO2 is grown on the surface of Fe3O4, with MWCNTs serving as the structural backbone. VSM of MWCNT–Fe3O4 and MWCNT–Fe3O4–MnO2 are shown in Fig. 13(g).
KOW value, which may enhance its hydrophobic interactions with the SWCNTs.116 In another study, MWCNTs modified with iron oxide and manganese dioxide (MWCNT–Fe3O4–MnO2) were synthesized and subsequently used for the adsorption of BPA. A schematic diagram illustrating the BPA extraction process is presented in Fig. 13(a). Similarly, Fig. 13(b) shows the adsorption capacities of BPA on MWCNT–Fe3O4–MnO2 and other materials. The maximum adsorption capacities were 76.75 mg g−1 for MWCNTs, 44.52 mg g−1 for MWCNT–Fe3O4, and 132.89 mg g−1 for MWCNT–Fe3O4–MnO2. Fig. 13(c) shows the contact time for BPA adsorption onto MWCNT–Fe3O4 and MWCNT–Fe3O4–MnO2. MWCNT–Fe3O4–MnO2 exhibited the optimal contact time, reaching 150 minutes. Overall, MWCNT–Fe3O4–MnO2 proved to be the most efficient adsorbent, which could be due to its enhanced surface area and increased number of binding sites.117 The TEM images shown in Fig. 13(d–f) reveal that MnO2 is grown on the surface of Fe3O4, with MWCNTs serving as the structural backbone. VSM of MWCNT–Fe3O4 and MWCNT–Fe3O4–MnO2 are shown in Fig. 13(g).
 | ||
| Fig. 13 (a) Schematic illustrating the BPA extraction process. (b) Adsorption capacities of BPA on MWCNT–Fe3O4–MnO2 and other materials. (c) Contact time for BPA adsorption onto MWCNT–Fe3O4 and MWCNT–Fe3O4–MnO2. TEM images of (d) MWCNTs, (e) MWCNT–Fe3O4, and (f) MWCNT–Fe3O4–MnO2. (g) VSM of MWCNT–Fe3O4 and MWCNT–Fe3O4–MnO2. Reproduced from ref. 117 with permission from Elsevier, copyright [2020]. | ||
The nanomembranes used in filtration techniques such as RO, although excellent in ion rejection, the small pores and low permeate flux cannot meet the requirements of modern times. Thus, a highly efficient functional membrane for desalination is required, which exhibits stability and a good separation capability. As permeate flux increases, there is a concurrent loss in rejection efficiency. To bring about improvement in this matter, membranes containing CNT, MOFs, or GO have been incorporated into the polymers but these tend to agglomerate on the surface and create surface defects. Therefore, it has often been concluded that nanomaterials need to be functionalized. Researchers have functionalized CNTs with carboxyl groups or sulphonyl groups to prepare nanofiltration membranes. Zheng et al. prepared a polyethersulfone nanofiltration membrane having a polyamide layer with enhanced permeation flux and antifouling properties.118 Zhao et al.119 synthesized a novel sodium lignosulphate–CNT (SLS–CNT) interface polymeric membrane having a unique cobweb-like surface structure. The SLS–CNT series of interface polymeric membranes contained a large number of hydrophilic groups and water transport channels. In this configuration, the hydrophilically modified CNTs became untangled and extended through the polyamide (PA) layer under the influence of an electric field. This led to the creation of microporous water channels between the CNTs and the PA matrix. As a result, a distinctive membrane surface morphology formed, which exhibited a significantly enhanced permeability of 24.4 L m−2 h−1 bar−1. Fig. 14(a) shows the schematic for the formation of a PA–CNT on the surface of the PES substrate, resulting in a strong and unique membrane assembly formed by the interfacial polymerization reaction mechanism. Fig. 14(b) shows the CNT water channel mechanism in the absence of an electric field. Due to hydrophilic modifications of CNT and the applied electric field, a cobweb-like structure was obtained on the surface. Also, micropores between PA molecules and protruding CNT provide new water channels (Fig. 14(c)). The protruding SLS–CNT initially contacts the water layer, and numerous surface hydrophilic groups accelerate the adsorption of water molecules, allowing for high permeability via micropores. The nanofiltration membrane showed excellent long-term stability, while still maintaining a high permeability flux and Na2SO4 salt rejection rate (above 97%). A series of membranes with various SLS–CNT amounts, with/without passing DC, has been prepared. The permeation flux and rejection rates for MgCl2 and Na2SO4 salts were tested for the different membranes prepared. The SLS–CNT interfacial polymeric membrane showed satisfactory chlorine resistance and fouling resistance, as shown in Fig. 14(d).
 | ||
| Fig. 14 (a) Schematic of the formation of a PA–CNT on the PES substrate by an interfacial polymerization reaction. (b) CNT water channel mechanism in the absence of an electric field. (c) Due to hydrophilic modifications of CNT and the applied electric field, a cobweb-like structure is obtained on the surface. Micropores between PA molecules and protruding CNT provide new water channels. (d) Results of the permeation flux and rejection rates for various salts, which were tested for the different membranes prepared. Reproduced from ref. 119 with permission from Elsevier, copyright [2023]. | ||
To summarize, both SWNT and MWNTs are remarkable nanomaterials that are commonly prepared by CVD, laser ablation, arc discharge methods, etc. The structural features of CNTs are responsible for extracting contaminants, as well as the effects of functionalisation on their surface, making them effective in capturing heavy metals, dyes, pharmaceuticals, VOCs, etc. CNTs with polymeric membranes are capable of increasing the efficacy of salt rejection and fouling resistance during the desalination process.
2.2 Graphene
Graphene is a fascinating material with remarkable properties. It is a two-dimensional crystalline carbon, composed of a monolayer of carbon atoms arranged in a hexagonal lattice array.120 It was discovered by Andre Geim and Konstantin Novoselov in 2004. It is the thinnest material with a thickness of only 0.334 nm.121 Graphene's name comes from “graphite,” a three-dimensional carbon crystal with weakly coupled graphene layers, and the suffix “-ene”, which refers to hexagonal ring structures in polycyclic aromatic hydrocarbons.122 Graphene is a parent form for various graphitic structures, including graphite, carbon nanotubes, and buckyballs. Graphene's hexagonal lattice, with strong covalent bonds between carbon atoms, forms a perfect arrangement with short carbon–carbon bond lengths, resulting in exceptional strength and stability.123 Carbon in graphene's atomic structure forms strong covalent bonds with other carbon atoms, resulting in an exceptional solid hexagonal lattice framework. It shows exceptional strength and unique electronic properties due to sp2 hybridization, where three out of four valence electrons result in a flat, stable structure as seen in Fig. 15(a–e).124 | ||
| Fig. 15 (a) Carbon atom structure. (b) Electronic configuration of carbon atoms. (c) Atomic orbitals and their hybridization to form sp2-hybrids. (d) Graphene crystal lattice, with A and B representing carbon atoms from different sub-lattices, and a1 and a2 representing unit-cell vectors. (e) Double bond formation, i.e., sp2 hybridization. Reproduced from ref. 125 with permission from Taylor and Francis copyright [2018]. | ||
Graphene is synthesized through both top-down and bottom-up approaches, and it can also be fabricated on various substrates, as illustrated in Fig. 16(a and b).126,127 In the chemical synthesis of graphene oxide, graphite is oxidized using strong acids and oxidizing agents. Three primary methods have been developed over time: the Brodie method, the Staudenmaier method, and the Hummers' method128–130 (Fig. 16(c)). Fig. 16(d) demonstrates an efficient protocol in which graphene sheets are converted into graphene oxide in just five minutes using an oxidation technique.131
 | ||
| Fig. 16 Synthesis of graphene and graphene oxide. (a) Bottom-up and top-down routes to synthesize graphene. Reproduced from ref. 126, copyright [2016]. (b) Synthesis of graphene by different protocols. Reproduced from ref. 127 with permission from Springer Nature copyright [2020]. (c) Graphene oxide synthesis by chemical oxidation using Brodie's, Staudenmaier's and Hummers' methods. Reproduced from ref. 128, copyright [2019]. (d) Rapid synthesis of graphene oxide from graphene sheets in 5 minutes using oxidation. Reproduced from ref. 131, copyright [2021]. | ||
Graphene, being a strong, flexible material with robust carbon–carbon bonds, is a popular choice for room-temperature applications. Its delocalized π-electron system allows electrons to move rapidly, making it ideal for transparent electronic devices, gas separation, and filtration. Graphene's lightweight structure makes it ideal for various applications. It is biocompatible, non-toxic, and useful in energy storage devices and catalysis. Graphene's unique electronic properties, including massless Dirac fermions, make it a fascinating material for fundamental physics research.132 There are two derivatives of graphene, namely graphene oxide and reduced graphene oxide. GO, an oxidized form of graphene, was originally made in 1859 by oxidizing and exfoliating graphite. This approach introduces oxygen-containing functional groups (epoxy, carboxyl, or hydroxyl groups) and disturbs the sp2 structure of graphite layers.133 GO is water and solvent-dispersible and may be used in biomedical, sensor, solar cell, and energy storage applications. However, the sp2 bonding networks have been destroyed, so it can be regarded as an electrical insulator. Reduced graphene oxide is a type of graphene oxide in which the oxygen-containing functional groups have been removed.134 Thus, rGO is synthesized by eliminating oxygen-containing functional groups and reintroducing sp2 carbon. The reduction process influences the characteristics of rGO, specifically its conductivity. Highly reduced rGO has electrical conductivity qualities like pure graphene.135 GO is reduced using a variety of methods, including chemical reduction, thermal reduction, and electrochemical reduction. rGO is a more cost-effective material that is employed in energy storage, batteries, supercapacitors, and biomedical applications. GO and rGO differ in oxygen content, with GO being an electrical insulator and rGO being conductive.136 Various methods have been used to synthesize rGO, such as epitaxial growth, CVD, mechanical exfoliation, and chemical reduction.137 Mechanical exfoliation is the process of removing single layers of graphene from a graphite sample, whereas CVD is the process in which a carbon-containing gas at high temperatures in a reactor decomposes, resulting in a single carbon sheet.138 Chemical reduction eliminates oxygen-containing functional groups, while epitaxial growth develops graphene on a crystal surface. The Hummers' process is commonly used in the synthesis of graphene oxide, which includes reacting graphite with potassium permanganate in a strong sulfuric acid solution, as shown in Fig. 16(d). Ultrasonic treatment during oxidation aids in the separation of outer graphene layers.139 Graphene and its derivatives have made wonderful contributions in water desalination and water purification processes. The main benefits of graphene-based materials include high adsorption properties and easy availability of binding sites on modification.
Ruoff's group140 synthesized spongy graphene by reducing GO platelets. The spongy graphene showed a significant increment in the adsorption efficiency of oil and volatile organic solvent from 20 to 86 times its weight in comparison with polymers and expanded graphite, an alternative for oil sorbent. A polysulfone membrane used in the RO process for water shows fouling problems, thereby resulting in the reduction of the membrane's lifetime. Crock et al. prepared a polysulfone nanocomposite membrane using graphene nanoplatelets decorated with gold (Au) nanoparticles. In the RO membranes, osmotic pressure leads to a higher flux of water. It was observed by Crock et al. that Au-loading resulted in increased flux while graphene provides a controlled membrane structure.141 This group also worked on Pd and Pd–Au bimetallic nanocomposites.142 Khaliha et al.143 tailored the GO sheet and prepared a highly defective graphene oxide, named defective GO (dGO), which has an extremely high number of holes and vacancies due to the presence of an increased number of carboxylic groups. The material was tested for the selective adsorption of a mixture of eight EDCs, which includes OFLOX, fluoroquinolone, BPA, BP3, etc. The material was also compared with different forms of GO, such as Brodie-derived GO (bGO), prepared by the oxidation of graphite using fuming nitric acid with sodium chlorate, Hummers-derived GO (hGO), prepared by reacting potassium permanganate with NaNO3 in sulphuric acid for oxidation of graphite, and generating reduced GO (rGO) having an increasing number of ‘defects’ in the order of dGO > rGO > hGO > bGO as shown in Fig. 17(a–d). It was observed that the adsorption capacity of dGO for OFLOX and BP3 was the highest of all forms of GO, while a higher selectivity of rGO was found toward RhB dye (Fig. 17(k)). Molecular dynamics modelling was used by the authors to explain such selectivity [Fig. 17(e–j)]. The simulations were carried out using the AMBER force field to describe the interaction parameters of different forms of graphene oxide. The aqueous environment was represented using the water model [e.g., TIP3P or SPC/E], which is widely applied in carbon-based adsorption studies. Periodic boundary conditions were applied in all three directions (x, y, and z) to mimic bulk solution behavior and eliminate edge effects. The simulation box was sufficiently large to avoid spurious interactions between periodic images of the adsorbent sheets. Various functional groups, such as epoxy, hydroxyl, carbonyl, and carboxylic acid, were randomly distributed on the GO surfaces to reflect the experimental data, and different-sized vacancies were introduced into dGO to model real defects. The results showed that adsorption behavior was governed by both electrostatic interactions and structural features. OFLOX exhibited stronger interactions with hGO and bGO due to its localized positive charge, which favors electrostatic attraction with negatively charged GO groups. In contrast, RhB's charge is more delocalized, resulting in slightly weaker binding. On rGO, RhB showed significantly stronger interaction than OFLOX (−38.8 vs. −29.4 kcal mol−1), attributed to π–π stacking between RhB's aromatic rings and rGO's planar sp2 surface, as supported by a greater reduction in solvent-accessible surface area upon adsorption. For dGO, the simulations revealed a different mechanism: OFLOX adsorbed preferentially into the nanoscale cavities (∼331 Å3) formed by graphene defects, matching its molecular volume (∼284 Å3), while RhB, being bulkier (∼388 Å3), could not fit and instead adsorbed on the flat surface. This shape complementarity between OFLOX and dGO cavities was identified as the key factor behind dGO's selective adsorption behavior. Overall, the MD simulations not only confirmed experimental trends but also clarified how molecular size, charge distribution, and graphene surface features collectively influence adsorption efficiency and selectivity.
 | ||
| Fig. 17 Different forms of graphene oxide: (a) Brodie GO (bGO), (b) Hummers-derived GO (hGO), (c) reduced GO (rGO), (d) defected GO (dGO) with increasing number of defects. Adsorption of (e) OFLOX and (f) RhB on GO (g) OFLOX and (h) RhB on dGO (i) OFLOX and (j) RhB on rGO. (k) Removal % of the mixture of ECs for different forms of GO samples at contact times of 15 min, 1 h, 4 h, and 24 h. Reproduced from ref. 143 with permission from Elsevier copyright [2021]. | ||
Members of the US Army Corps of Engineers have, in the recent past, invented a water filtration membrane made using chitosan and graphene oxide. This technology is now available for integration into commercially available products and services through a patent.144 Another group has used GO and rGO 2D nanosheets for the removal of a mixture of fourteen selected PFASs present in tap water, followed by their removal from the treated water by combining their adsorption in batch and microfiltration on polyethersulfone hollow fibers. These fibers are water disinfecting but are unable to remove the EDCs from drinking water by adsorption or by physical filtration when passed through the membrane. It was observed that rGO was more effective in removing PFASs than graphene oxide from the tap water. Moreover, it was reported that both GO and rGO were able to remove more of the sulfonated compounds than the carboxylic analogues with an increase in the chain length. The authors compared their results (138 μg g−1) with granular activated carbon (43 μg g−1) results and found it to be 3 to 5 times more efficient for PFAS removal.145 Feldblyum et al.146 modified the surface of graphene oxide (GO) using the cationic surfactant cetyltrimethylammonium chloride to enhance its adsorption performance for the removal of PFAS mixtures from river water, including both short- and long-chain compounds. The surface functionalization was designed to increase electrostatic interactions and hydrophobic affinity toward PFAS molecules, resulting in improved adsorption kinetics and capacity. The modified GO achieved nearly 100% removal efficiency of PFAS in river water, highlighting the significant impact of surface chemistry optimization in the development of next-generation adsorbents for emerging contaminants. Recently, Trifoglio et al. developed alkyl amine-grafted graphene oxide nanosheets through a surface modification approach. The functionalization introduced hydrophobic and electrostatic interaction sites, effectively enhancing the affinity of GO toward PFAS molecules. The modified GO showed significantly higher adsorption capacities and faster removal rates compared to unmodified GO. The study assessed the role of the tertiary amine in PFAS adsorption by comparing GO-N,N-dimethylethylenediamine (a tertiary amine), GO-quaternized amine and GO-octylamine (no terminal amine). Results showed that the tertiary amine in N,N-dimethylethylenediamine significantly enhanced PFAS adsorption through electrostatic and hydrogen bonding interactions, thus highlighting the importance of functional group chemistry in adsorbent design.147 Mahofa et al. developed a β-cyclodextrin-modified graphene oxide membrane that effectively removes short-chain PFAS. The integration of β-CD creates specific binding sites within the GO nanochannels, effectively increasing intrapore energy barriers and hindering the transport of short-chain PFAS molecules. The membrane achieved over 90% retention of PFAS with high water permeability, significantly outperforming conventional membranes and offering a promising solution for advanced water purification.148 In order to remove egg albumin from wastewater, the authors initially dispersed silica uniformly on graphene oxide and then the SiO2–GO nanohybrid was doped onto a polysulfone membrane to prepare a nanohybrid membrane as SiO2–GO/PSU.149 The nanohybrid membrane was used in removing egg albumin from wastewater and it was found to exhibit better rejection properties in comparison with SiO2/PSU and GO/PSU hybrid membranes. This could be attributed to the high hydrophilicity and good dispersibility due to the synergistic effect of silica nanoparticles and graphene oxide. In yet another study, researchers incorporated GO into a cellulose triacetate (CA) membrane by dispersing graphene in different weight concentrations (0.25, 0.5, and 1% wt.) in the parent polymer. As a result, its thermal and mechanical properties were improved and the retention capacity for serum albumin was found to increase from 78% (CA membrane) to 99% in the composite membrane.150 Nipane et al.151 synthesized the rGO–ZnO nanorod nanocomposite as a photocatalyst for the degradation of MB and MO under UV irradiation. The zinc oxide nanorods absorb photons, generating electron–hole pairs, while the graphene sheets act as electron acceptors, quickly transferring electrons away from the zinc oxide. This synergistic effect prevents recombination and enhances the generation of reactive oxygen species, which are more effective at degrading the dye molecules. Methylene blue, a cationic dye, and methyl orange, an anionic dye, were observed to undergo degradation. The negative zeta potential of the composite indicates a greater absorption of the cationic dye MB than the anionic dye MO. Another group synthesised the same material, rGO–ZnO nanorod hydrogel, for methylene blue removal. The increase in efficiency was attributed to the laterally grown ZnO nanorods, which exhibit stronger electrostatic attractive forces and enhanced photocatalytic activity.152 Sun et al. incorporated magnetite into rGO and formed a nanocomposite that could efficiently remove rhodamine B and MG in water, with a removal rate of over 90%.153 In another work, metal ferrite (M = Cu, Co, or Ni)-enabled graphene oxide (MFe2O4@GO) nanocomposites were synthesized for methylene dye removal. The maximum adsorption capacities of MFe2O4@GO (where M = Cu, Co, or Ni) were 25.81, 50.15, and 76.34 mg g−1, respectively. The adsorption kinetics of methylene blue followed a pseudo-second-order model. Among the MFe2O4@GO nanocomposites studied, NiFe2O4@GO demonstrated the highest adsorption performance.154 Pan et al. developed a graphene oxide (GO) laminate membrane intercalated with Prussian blue nanocubes that acts as a photo-Fenton self-cleaning membrane for water purification. The nanocubes enhance water permeability and enable efficient dye degradation under visible light. The membrane achieved nearly 100% dye rejection, high water flux, and maintained performance over multiple cycles, showing strong potential for safe and effective water treatment.155 Graphene was also tested for separating oil from water. In this regard, graphene has also been found to be a promising candidate. A hydrophilic membrane can form a hydrogen bond with water, which helps in separating the oily particles, such as hydrocarbons, surfactants, grease, etc., from oil/water mixtures. Scientists have developed hydrophilic membranes by functionalizing hydrophobic graphite and graphene material. One of the research groups developed a method to increase the hydrophilic nature of the polyethersulfone membrane by using amine-functionalized graphene attached to polyacrylonitrile-co-maleimide. The water permeability was reported to have been increased by 43% with >99% selectivity. The modified graphene-based material exhibited the potential to be a new generation material for water filtration membranes.156 Schmidt et al.157 recently reported the fabrication of cellulose acetate membranes derived from biomass and subsequently modified with TiO2 and GO for the treatment of oil-in-water emulsions. The incorporation of TiO2 and GO significantly enhanced the membranes' hydrophilicity, mechanical strength, and antifouling properties, which are critical for maintaining flux and selectivity in complex wastewater streams. The study demonstrated improved oil rejection and permeability, indicating that the synergistic effects of TiO2 and GO play a pivotal role in enhancing separation performance. These results highlight the potential of bio-derived, nanocomposite membranes as promising candidates for sustainable oil–water separation technologies, particularly in industrial and environmental wastewater applications. Toxic metals in drinking water pose a significant problem, and graphene derivatives are also being investigated as a possible solution in this area, along with graphene composite aerogels, which are effective adsorbents suitable for use in water purification.158 In this regard, Rezaee et al.159 prepared a GO–PSU membrane and the presence of hydrophilic functional groups on GO enhanced the hydrophilicity and permeability of the synthesized membrane. The composite showed an increase in the removal of As in comparison to the pure polymer membrane. The increase in the removal of As was reported to be due to Donnan repulsion caused by negative charges induced by GO on the surface of the membrane. The heavy metals, such as Pb2+, Cu2+, and As3+, were successfully removed by using EDTA functionalized by magnetite and CS onto GO nanocomposites (EDTA-MCS/GO). Magnetic-functionalized graphene oxide nanocomposites exhibited the highest adsorption capacities of 206.52, 207.26, and 42.75 mg g−1 for Pb2+, Cu2+, and As3+, respectively. pH-related studies revealed that at higher pH, the nanocomposite acquires a negative charge, which attracts positively charged metal ions through electrostatic forces, leading to the adsorption of metals on the nanocomposite surface. Additionally, the authors reported that the rate of heavy metal ion adsorption is also dependent on the temperature of the solution, and with increasing temperature, the adsorption densities of Pb2+ and Cu2+ decreased significantly, while the adsorption density of As3+ increased. These results reveal that the adsorption of Pb2+ and Cu2+ on the EDTA-MCS/GO nanocomposite was an exothermic process, whereas the adsorption of As3+ was an endothermic reaction.160 Kumar and co-workers synthesized trioctylamine-modified GO that can adsorb 99.4% of the Cr(VI) present in water in less than an hour.161 Similarly, N. Pradeep Singh and co-workers synthesized a chitosan–graphene oxide composite material and studied the adsorption of hexavalent Cr(VI) using a batch mode process. The nanocomposite demonstrated an adsorption capacity of 104.16 mg g−1 at pH 2 within 420 minutes and was found to be recyclable for up to 10 cycles. The adsorption process was well described by the pseudo-second-order kinetic model and the Langmuir isotherm model.162
DFT studies reveal information such as adsorption geometry, bonding types, and electronic changes, which experiments alone cannot predict. The adsorption capacity, selectivity, and guidance in designing nanocomposites for enhanced adsorption can be laid out by DFT. Further, it can validate experiments, corroborate pH and temperature dependence, and offer guidelines for structural modifications. DFT analysis of Cr adsorption on functionalised GO is based on interactions between OH–, epoxy, and –C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O group on its surface. The adsorption energy is close to −150 kcal mol−1, and charge transfer occurs. These interactions between Cr(III) and GO are electrostatic, driven by van der Waals forces. Adsorption was observed to depend on pH and is optimal at 2–4 due to the protonation of surface functional groups and speciation of Cr(VI). The kinetics of this reaction follows a pseudo-second-order model, and chemisorption dominates. Langmuir isotherms fit better than Freundlich isotherms, indicating monolayer adsorption. The adsorption process is spontaneous, endothermic, and entropy-driven.163 In yet another study, a composite of ferrite nanoparticle with reduced graphene oxide was synthesized, and GO–Fe3O4 functionalized with 8-hydroxyquinoline proved to be highly efficient in the sorptive removal of Cr, achieving a capacity of 14.63 mg g−1 μM−1, higher than other compounds (e.g., GO, CS–GO, and RGO/NiO).164 Graphene can adsorb fluoride from water, but its adsorption capacity is low, since it has been observed that interactions between them are weak. There are reports in which basic aluminium sulfate has been deposited on porous graphene hydrogel. The primary advantage of using GHG is that its surface area is much larger than powdered graphene oxide. The adsorption capacity was measured to be 33.4 mg g−1 at an equilibrium fluoride concentration of 10.7 mg L−1.165 Chandra et al. removed arsenic ions from polluted water using magnetite-reduced GO and also found the positive effect of pH on the adsorption ability of the nanocomposite.166 Recently, Pryadko's group performed the in situ synthesis of a magnetic nanocomposite made from magnetite (Fe3O4) and rGO for As(III) removal from water. The nanocomposite demonstrated an impressive removal capacity of 14 mg g−1. Additionally, it achieved 81% adsorption within just one minute, surpassing the performance reported in literature.167 In another study, MnO2 was incorporated into a Fe3O4/graphene oxide composite for the removal of As(III) and As(V) from water. The MnO2 on the adsorbent surface facilitated the oxidation of As(III) to As(V) without the need for an external oxidant, while also contributing to the adsorption of arsenic in conjunction with Fe3O4.168 Hubdar Ali Maitlo's group169 compared various GO-based nanocomposites reported in the literature for chromium adsorption, including GO, chitosan-grafted graphene oxide,170 3-aminopropyltriethoxysilane-functionalized graphene oxide,171 reduced graphene oxide/NiO (RGO/NiO),172 and a composite of GO/chitosan/metal–organic framework [GO–CS@MOF (Zn(BDC)(DMF))],173 among others. The sorption capacities of these materials follow the order of GO–CS@MOF [Zn(BDC)(DMF)] (145 mg g−1) < GO–FH nanocomposite (212.8 mg g−1) < Ppy–GO–NC (625 mg g−1), corresponding to initial chromium concentrations of 962, 1923.5, and 3847 μM, respectively. Ibrahim and Mosa have doped graphite sheets with silver nanoparticles and evaluated their effectiveness as adsorbents for the removal of various heavy metal ions from water. The results demonstrated that the silver-doped graphite composite exhibited excellent adsorption performance, achieving removal efficiencies of 100% for Pb, 98% for Cu, 93% for Cr, 91% for Cd, 88% for Ni, 86% for Zn, 86% for Fe, and 33% for Hg.174 Soomro's group fabricated MoO2@GO and WO3@GO nanocomposite membranes using a simple method. These membranes exhibited strong stacking interactions and electrostatic attraction, leading to high separation performance. They achieved over 70% rejection of small ions like Na+ and Mg2+, and over 99% rejection of heavy metal ions such as Ni2+ and Pb2+, along with excellent water permeability (275 ± 10 L m−2 h−1 bar−1). These results highlight their strong potential for next-generation water purification and desalination technologies.175 Schmidt, Dou, and Sydlik introduced a sustainable, regenerable graphene-based water filter designed for household removal of heavy metals such as lead, cadmium, and mercury. This filter demonstrated removal efficiencies up to 65 times greater than those of conventional activated carbon filters. A notable feature of this technology is its reusability; the filter can be regenerated using simple treatments with hot water (80 °C) or vinegar, maintaining its performance over multiple cycles. This innovation offers a cost-effective and environmentally friendly solution for point-of-use water purification, addressing the critical need for accessible clean water in areas affected by heavy metal contamination.176 Additionally, there are reports investigating the adsorption behaviour of various organic pollutants on graphene in water. Wu et al. investigated the adsorption behaviour of various organic chemicals on graphene, including acrylonitrile, 1-naphthalenesulfonic acid, p-toluenesulfonic acid, and MB. The study explores how graphene interacts with different types of molecules—neutral, acidic, and dye compounds. It highlights organic molecules with larger sizes and suggests that more benzene rings result in faster adsorption rates and higher maximum adsorption capacities on graphene.177 Yu et al. investigated the adsorption capacities of benzene and toluene on rGO and GO surfaces. They found that the adsorption capacity for benzene was 276.4 mg g−1 on rGO and 216.2 mg g−1 on GO, while for toluene, it was 304.4 mg g−1 on rGO and 240.6 mg g−1 on GO. These results show that rGO has a higher adsorption capacity than GO. This could be attributed to the higher surface area of rGO (292.6 m2 g−1) compared to GO (236.4 m2 g−1). Additionally, the adsorption capacity for toluene was higher than for benzene, likely due to the presence of the methyl group in toluene, which can interact with the oxygen-containing groups on the surface of rGO through hydrogen bonding. This interaction enhances the adsorption of toluene on rGO.178 Another study explored the development of a composite material combining graphene quantum dots with zeolitic imidazolate framework-8 to effectively remove VOCs such as toluene and ethyl acetate from water. The composite demonstrated significantly improved adsorption capacities, achieving maximum values of 552.31 mg g−1 for toluene and 1408.59 mg g−1 for ethyl acetate; the corresponding removal efficiencies were 80.25% and 93.78%, respectively.179 In a recent study, graphene aerogels were explored as efficient adsorbents for water pollutants, with a comparison of two drying methods: freeze-drying and ambient pressure drying. The aerogel effectively adsorbed organic solvents such as acetone, ethanol, and methanol, as well as oils including pump oil, castor oil, and silicone oil. They also exhibited photocatalytic activity in degrading aqueous dye solutions like MB and Orange G.180
O group on its surface. The adsorption energy is close to −150 kcal mol−1, and charge transfer occurs. These interactions between Cr(III) and GO are electrostatic, driven by van der Waals forces. Adsorption was observed to depend on pH and is optimal at 2–4 due to the protonation of surface functional groups and speciation of Cr(VI). The kinetics of this reaction follows a pseudo-second-order model, and chemisorption dominates. Langmuir isotherms fit better than Freundlich isotherms, indicating monolayer adsorption. The adsorption process is spontaneous, endothermic, and entropy-driven.163 In yet another study, a composite of ferrite nanoparticle with reduced graphene oxide was synthesized, and GO–Fe3O4 functionalized with 8-hydroxyquinoline proved to be highly efficient in the sorptive removal of Cr, achieving a capacity of 14.63 mg g−1 μM−1, higher than other compounds (e.g., GO, CS–GO, and RGO/NiO).164 Graphene can adsorb fluoride from water, but its adsorption capacity is low, since it has been observed that interactions between them are weak. There are reports in which basic aluminium sulfate has been deposited on porous graphene hydrogel. The primary advantage of using GHG is that its surface area is much larger than powdered graphene oxide. The adsorption capacity was measured to be 33.4 mg g−1 at an equilibrium fluoride concentration of 10.7 mg L−1.165 Chandra et al. removed arsenic ions from polluted water using magnetite-reduced GO and also found the positive effect of pH on the adsorption ability of the nanocomposite.166 Recently, Pryadko's group performed the in situ synthesis of a magnetic nanocomposite made from magnetite (Fe3O4) and rGO for As(III) removal from water. The nanocomposite demonstrated an impressive removal capacity of 14 mg g−1. Additionally, it achieved 81% adsorption within just one minute, surpassing the performance reported in literature.167 In another study, MnO2 was incorporated into a Fe3O4/graphene oxide composite for the removal of As(III) and As(V) from water. The MnO2 on the adsorbent surface facilitated the oxidation of As(III) to As(V) without the need for an external oxidant, while also contributing to the adsorption of arsenic in conjunction with Fe3O4.168 Hubdar Ali Maitlo's group169 compared various GO-based nanocomposites reported in the literature for chromium adsorption, including GO, chitosan-grafted graphene oxide,170 3-aminopropyltriethoxysilane-functionalized graphene oxide,171 reduced graphene oxide/NiO (RGO/NiO),172 and a composite of GO/chitosan/metal–organic framework [GO–CS@MOF (Zn(BDC)(DMF))],173 among others. The sorption capacities of these materials follow the order of GO–CS@MOF [Zn(BDC)(DMF)] (145 mg g−1) < GO–FH nanocomposite (212.8 mg g−1) < Ppy–GO–NC (625 mg g−1), corresponding to initial chromium concentrations of 962, 1923.5, and 3847 μM, respectively. Ibrahim and Mosa have doped graphite sheets with silver nanoparticles and evaluated their effectiveness as adsorbents for the removal of various heavy metal ions from water. The results demonstrated that the silver-doped graphite composite exhibited excellent adsorption performance, achieving removal efficiencies of 100% for Pb, 98% for Cu, 93% for Cr, 91% for Cd, 88% for Ni, 86% for Zn, 86% for Fe, and 33% for Hg.174 Soomro's group fabricated MoO2@GO and WO3@GO nanocomposite membranes using a simple method. These membranes exhibited strong stacking interactions and electrostatic attraction, leading to high separation performance. They achieved over 70% rejection of small ions like Na+ and Mg2+, and over 99% rejection of heavy metal ions such as Ni2+ and Pb2+, along with excellent water permeability (275 ± 10 L m−2 h−1 bar−1). These results highlight their strong potential for next-generation water purification and desalination technologies.175 Schmidt, Dou, and Sydlik introduced a sustainable, regenerable graphene-based water filter designed for household removal of heavy metals such as lead, cadmium, and mercury. This filter demonstrated removal efficiencies up to 65 times greater than those of conventional activated carbon filters. A notable feature of this technology is its reusability; the filter can be regenerated using simple treatments with hot water (80 °C) or vinegar, maintaining its performance over multiple cycles. This innovation offers a cost-effective and environmentally friendly solution for point-of-use water purification, addressing the critical need for accessible clean water in areas affected by heavy metal contamination.176 Additionally, there are reports investigating the adsorption behaviour of various organic pollutants on graphene in water. Wu et al. investigated the adsorption behaviour of various organic chemicals on graphene, including acrylonitrile, 1-naphthalenesulfonic acid, p-toluenesulfonic acid, and MB. The study explores how graphene interacts with different types of molecules—neutral, acidic, and dye compounds. It highlights organic molecules with larger sizes and suggests that more benzene rings result in faster adsorption rates and higher maximum adsorption capacities on graphene.177 Yu et al. investigated the adsorption capacities of benzene and toluene on rGO and GO surfaces. They found that the adsorption capacity for benzene was 276.4 mg g−1 on rGO and 216.2 mg g−1 on GO, while for toluene, it was 304.4 mg g−1 on rGO and 240.6 mg g−1 on GO. These results show that rGO has a higher adsorption capacity than GO. This could be attributed to the higher surface area of rGO (292.6 m2 g−1) compared to GO (236.4 m2 g−1). Additionally, the adsorption capacity for toluene was higher than for benzene, likely due to the presence of the methyl group in toluene, which can interact with the oxygen-containing groups on the surface of rGO through hydrogen bonding. This interaction enhances the adsorption of toluene on rGO.178 Another study explored the development of a composite material combining graphene quantum dots with zeolitic imidazolate framework-8 to effectively remove VOCs such as toluene and ethyl acetate from water. The composite demonstrated significantly improved adsorption capacities, achieving maximum values of 552.31 mg g−1 for toluene and 1408.59 mg g−1 for ethyl acetate; the corresponding removal efficiencies were 80.25% and 93.78%, respectively.179 In a recent study, graphene aerogels were explored as efficient adsorbents for water pollutants, with a comparison of two drying methods: freeze-drying and ambient pressure drying. The aerogel effectively adsorbed organic solvents such as acetone, ethanol, and methanol, as well as oils including pump oil, castor oil, and silicone oil. They also exhibited photocatalytic activity in degrading aqueous dye solutions like MB and Orange G.180
To summarize, graphene, a fascinating 2D material, has been discussed in this sub-section with respect to its structure, properties exhibited, and its synthesis along with GO and rGO. The adsorption of oil, VOCs, antibiotics, and other organic pollutants has been discussed along with a comparison of bGO, hGO, rGO, and dGO for OFLOX, BP3, PFAS, and RhB removal. Functionalisation with amines and cyclodextrin, and composites with chitosan, MOFs, SiO2, ZnO, etc., to increase the efficiency level of purification of water have been explored. Further, improvement in membrane performance by intercalating various GOs with different materials capable of capturing heavy metals, oils and degradation of dyes has been discussed above.
2.3 Graphyne
Graphyne is a one-atom-thick, flat carbon network—similar to graphene. In 1987, Baughman et al.181 first predicted that graphynes (GYs) would form a series of stable crystalline carbon allotropes characterized by a high degree of sp hybridization. Composed of ethynyl units (sp-hybridized carbon) and aromatic rings (sp2-hybridized carbon), GYs were named for their structural resemblance to both graphite and ethyne.182 The coexistence of sp- and sp2-hybridized carbon atoms endows GYs with high π-conjugation, tunable electronic properties, a two-dimensional planar framework, and uniformly distributed pores (Fig. 18(a)).183a Over the past few decades, both theoretical predictions and experimental studies have explored the potential mechanical, optical, and electronic properties of GYs. Similar to graphene, GYs also possess a natural semiconductor band gap. They demonstrate exceptional electrical characteristics, including high carrier mobility and low carrier effective mass. The mechanical properties of GYs are strongly influenced by the number and arrangement of their acetylenic linkages. As seen in Fig. 18(b), graphynes can be classified as α-graphyne, β-graphyne, γ-graphyne, 6,6,12-graphyne, graphdiyne, and so on. In the graphyne family, to date, α-graphyne, β-graphyne, and 6,6,12-graphyne exist only as theoretical carbon allotropes; there are no experimental reports of their synthesis. γ-Graphyne possesses the highest stability in theory and experimentally, and there are also reports of its synthesis.183bFig. 18(c and d) depict the TEM images clearly indicating the hexagonal arrangement of C atoms and EDAX indicating a crystalline structure (Fig. 18(c)). Early studies proposed that γ-graphyne could be obtained through the controlled oligomerization of cyclo[12]carbon. Cui et al. reported a novel approach for GY synthesis using calcium carbide (CaC2) and perbromobenzene (C6Br6) by using ball milling for cross-coupling reactions (Fig. 18(e)). They reported that as-prepared γ-graphyne was a p-type semiconductor with a band gap of 2.58 eV.184 Yang and co-workers synthesized graphyne through ball milling using the same precursors, followed by calcination. They reported that the calcination step eliminated the defects introduced during ball milling.185 In yet another method, the Sonogashira cross-coupling procedure was used for the synthesis of γ-graphyne using hexabromobenzene and calcium carbide in the presence of copper acetylacetonate and palladium(II) acetate as catalyst.186 Graphdiyne (GDY), another member of the graphyne family, is typically synthesized using bottom-up coupling strategies. Graphdiyne substructures were achieved through the copper-mediated acetylenic coupling of terminal alkynes as well as palladium-catalyzed Sonogashira coupling using alkynyl halides.187,188 However, more stable precursors and effective catalytic systems make copper-mediated acetylenic coupling the most promising route for GDY synthesis. To date, four conventional copper-mediated coupling strategies—Glaser coupling,189 Glaser–Hay coupling,190 alkynylsilane coupling,191 and Eglinton coupling192 have been widely employed in fabricating GDY. Researchers have refined this approach to control the morphology (e.g., nanowalls and nanotubes) and scalability using controlled-release copper complexes or metal salt catalysts (Fig. 18(f)). Alternative methods include interface-assisted growth193 at liquid–liquid and gas–liquid interfaces to produce high-quality layered films, and low-temperature chemical vapor deposition (CVD) onto metal substrates like silver to form monolayer or few-layer films.194 Additional strategies, such as explosion-induced coupling195 or electron beam irradiation, offer ultrafast synthesis.196 | ||
| Fig. 18 (a) Molecular structures of graphene with the formation of graphyne and graphdiyne. Reproduced from ref. 183a with permission from the Royal Society of Chemistry, copyright [2009]. (b) Structural features of α, β and γ-graphdiynes. Reproduced from ref. 183b, Elsevier, copyright [2023]. (c) TEM images of graphyne at 100 nm, and HRTEM image (5 nm scale bar) of the aromatic ring network with the inset showing EDAX. Reproduced from ref. 183c, Springer Nature, copyright [2022]. (d) TEM depicting a hexagonal arrangement of carbon aromatic rings in a plane. Reproduced from ref. 183d, Wiley-VCH GmbH, copyright [2024]. (e) Different methods for the synthesis of graphyne. (f) Synthesis method of graphdiynes by a copper-mediated acetylenic coupling reaction. | ||
γ-Graphyne was synthesized using a modified mechanochemical method and employed as an adsorbent for the removal of organic dyes such as RhB, MB, and CR. The maximum adsorption capacities obtained from kinetic and isotherm analyses were 386.10, 357.14, and 254.45 mg g−1, respectively, which closely match the experimental values of 383.64, 359.72, and 242.44 mg g−1. The adsorption isotherm analysis by the authors shows that adsorption of organic dyes on γ-graphyne occurs via homogeneous and monolayer coverage and can be accurately modelled using the Langmuir isotherm. Density Functional Theory (DFT) calculations further demonstrated the primary adsorption sites for each dye: RhB preferentially binds to the trigonal rings, MB adsorbs mainly in the region between sp-hybridized carbon atoms and hexagonal rings, and CR interacts chiefly at the sp-bond regions. The negative electrostatic potentials at the specific atom on the dye—such as the oxygen atoms in RhB, nitrogen atoms in MB, and the sulfate (SO3) group in CR—facilitate strong interactions with the positive charge present on γ-graphyne. These interactions yield negative adsorption energies, which are favourable and lead to enhanced adsorption.197 Li et al. functionalized the alkyne linkages of γ-graphyne with sulfur moieties, conferring strong Lewis soft base characteristics that enhance the chemisorption of mercury ions (Hg(II)) from water. Equilibrium isotherm analysis fit the Langmuir model well, yielding an impressive maximum adsorption capacity of 675.7 mg g−1. The interactions involved are reversible Lewis soft acid–soft base bonds between Hg(II) ions and the sulfur-functionalized alkynyl groups, which underpin the adsorbent's exceptional reusability. After five adsorption–desorption cycles, the material retained over 91% of its capacity, although a slight performance drop occurred during the first cycle due to the irreversible adsorption of some Hg(II) on sulfur sites.198 Lin et al. synthesized the Ag3PO4@γ-graphyne composite as a photocatalyst for the degradation of norfloxacin, phenol, and 2-hydroxynaphthalene under visible light. The degradation efficiencies of phenol, 2-hydroxynaphthalene, and norfloxacin reached 100% in just 16, 5, and 8 minutes, respectively. LC-MS and 3D EEM analyses revealed that 2-hydroxynaphthalene and norfloxacin were primarily degraded into short-chain carboxylic acids. Free radical quenching experiments and ESR characterization indicated that various reactive species were involved in the degradation process, with photogenerated holes and superoxide radicals playing dominant roles.199 N-graphyne was in situ combined with BiOCl0.5Br0.5via a simple, one-step sonochemical method (Fig. 19(a)). The resulting N-graphyne/BiOCl0.5Br0.5 composite demonstrated significantly enhanced adsorption capacity and visible-light-driven photocatalytic degradation of levofloxacin (LEVO) compared to pristine BiOCl0.5Br0.5 (Fig. 19(b and c)).200 Another group decorated γ-graphyne on TiO2 nanotubes by a drop-coating method. The material showed 3.64-fold and 1.35-fold enhancements in the photoelectrocatalytic degradation of rhodamine B and levofloxacin, respectively, compared to pristine TiO2.201 Water desalination is essential for environmental protection and offers a sustainable solution to global water shortages. In this regard, different types of graphyne materials have been explored. Kou and co-workers202 investigated the performance of three graphyne-n nanosheets (graphyne-3, graphyne-4, and graphyne-5) as membranes for water desalination using molecular dynamics simulations. Their results showed that graphyne-3 achieved 100% salt rejection, indicating its strong potential for use in water desalination. Graphyne-4 nanosheets, on the other hand, exhibited superior salt rejection performance under an applied pressure of 100 MPa. It demonstrated more than 80% Na+ rejection and nearly 100% Cl− rejection, which is considered an acceptable rate for desalination. The lower passage of Cl− ions through graphyne-4 pores was attributed to their larger hydration shell compared to Na+ ions. Similar results were demonstrated by other workers; Zhu et al. studied the capability of the γ-graphyne-4 membrane for water desalination and concluded it to be a superior material to graphene. They also reported that the water flux increased linearly with the applied pressure.203 Wu's group204a also demonstrated that pristine γ-graphyne nanosheets exhibited higher salt rejection than conventional RO membranes. Furthermore, they reported that introducing negative charges onto the graphyne sheet could enhance both water permeability and salt rejection. To date, there have been no reports on γ-graphyne-1; therefore, Azamat et al. functionalized γ-graphyne-1 with hydroxyl, fluorine, and carboxylic acids (Fig. 19(d–f)) and found it to be equally effective for water desalination. Additionally, they reported that the salt rejection at <7.5 MPa applied pressure was 100% for all types of functionalized γ-graphyne-1 membranes but at high pressure, few ions could pass through the pores in some functionalized membranes (Fig. 19(g and h)).204b
 | ||
| Fig. 19 (a) A schematic showing the synthesis of the N-graphyne/BiOCl0.5Br0.5 composite, (b) photocatalytic degradation of LEVO, TC and RhB using the N-graphyne/BiOCl0.5Br0.5 composite and its comparison with pristine BiOCl0.5Br0.5. (c) Plausible mechanism for the degradation of LEVO, TC and RhB. Reproduced from ref. 200 with permission from RSC Pub copyright [2023]. Structures depicting γ-graphyne-1 functionalized with different functional groups: (d) hydroxyl, (e) fluorine and hydroxyl and (f) carboxylic acids. (g) Bar diagram showing the salt rejection of different functionalized pores at various applied pressures. (h) Graph showing the percentage salt rejection of γ-graphyne-1 functionalized with different groups at various applied pressures. Reproduced from ref. 204 with permission from [Elsevier B.V.] copyright [2020]. (i) Schematic depicting the adsorption of lead ions on graphdiyne. (j) SEM image of graphdiyne incorporated onto copper foam. (k) Pb2+ conc. in the filtrate and its removal efficiency by graphdiyne after a number of filter operations. (l) Removal efficiency of different concentrations of lead ions as a function of permeated volume in water. (m) Number of cycles for the removal efficiency of the graphdiyne-based foam filters. Reproduced from ref. 205, with permission from [WILEY-VCH] copyright [2017]. | ||
Graphdiyne (GDY), another member of the graphyne family, has emerged as a promising material for wastewater treatment. Graphdiyne has been reported in the literature for heavy metal removal, as its acetylenic linkages strongly interact with metal ions. In this context, Liu et al. demonstrated the removal of lead ions from contaminated water, achieving a maximum (Fig. 19(i)) adsorption capacity of 470.5 mg g−1 as determined by the Langmuir isotherm model. Furthermore, they incorporated graphdiyne onto copper foam and achieved a 99.6% removal efficiency for lead ions (Fig. 19(j–m)).205 In another study, heavy metals such as nickel, copper, and silver were investigated using Density Functional Theory. The results revealed that nickel atoms exhibited strong adsorption on the graphdiyne surface, while copper and silver underwent chemisorption.206 Graphdiyne has been reported in the literature as a material for removing organic pollutants from wastewater. Zhu et al. reported the removal of iodosulfuron-methyl sodium, a pesticide, from wastewater with a maximum adsorption capacity of up to 795.51 mg g−1, which is 130-fold higher than that of modified graphene oxide (6 mg g−1) and modified biochar (1.5 mg g−1) reported in previous studies.207 Zhu et al. developed a facile, safe, and scalable coupling reaction method by using six different transition-metal salts, including both Cu and Pd salts, to synthesize graphdiyne with diverse morphologies. Among these catalysts, CuSO4-catalyzed GDY stood out with over 90% yield, lower density, and fewer structural defects. This CuSO4GDY demonstrated broad applicability in water purification by effectively adsorbing a variety of organic pollutants—including dyes, tetracycline antibiotics, and neonicotinoid pesticides. Its dye adsorption performance was stronger than that of conventional adsorbents like graphene and activated carbon and comparable to MWCNTs.208 In another work, Thangavel and coworkers prepared a nanohybrid of graphdiyne–ZnO by the hydrothermal method and studied the degradation of methylene blue and rhodamine B. The degradation studies showed nearly twice the rate of degradation compared to bare ZnO nanoparticles. The authors proposed that the interfacial charge transfer from ZnO to GDY is a key mechanism. Graphdiyne, as a carbonaceous two-dimensional material, promotes the separation of photogenerated electron–hole pairs through efficient transfer across the interface, reducing recombination losses.209 Recently, copper-doped ZnO and graphdiyne (Cu/ZnO@GDY) were synthesized via a hydrothermal method. A photocatalytic membrane, Cu/ZnO@GDY/PES, was then prepared using polyethersulfone (PES) and applied in a photocatalytic membrane reactor for the degradation of tetracycline. The reactor demonstrated stable degradation efficiency during continuous flow operation.210 In many reports, non-metals are also doped on graphdiyne. Bai et al. reported boron-doped graphdiyne quantum dots loaded on hollow tubular carbon nitride for the degradation of 28 organic dyestuffs, BPA, antibiotics and bacterial destruction in a simulated wastewater system. The material exhibited enhanced degradability toward various pollutants, broad applicability in pollutant treatment, and promising potential for practical applications.211 Zhang's group synthesized sulfur-doped GDY nanosheets via a hydrothermal method. They demonstrated that sulfur-doped GDY effectively degrades dyes and pharmaceuticals, highlighting its potential as a promising candidate for wastewater treatment.212 Li et al. developed a graphdiyne-coated melamine sponge (GDYMS) using an in situ Glaser–Hay coupling reaction. This composite material exhibits superhydrophobic properties, enabling it to absorb oils and organic solvents up to 160 times its weight. GDYMS showed >97% oil–water separation efficiency. It remains stable in corrosive solvents, allowing for the extraction of various organic solvents such as chloroform, dichloromethane, ethyl acetate, petroleum ether, toluene, diesel, gasoline, and silicon oil. The GDYMS maintains its adsorption capacity over 100 reuse cycles, with a contact angle of 132°, indicating excellent recyclability and effectiveness.213 Gao et al. reported another study on oil–water separation, in which copper foam was used as a substrate to prepare nano-GDY coated with poly(dimethylsiloxane). The resulting material exhibited a contact angle of 171° (superhydrophobic foam) and achieved an oil–water separation efficiency >98%.214 Xue et al. demonstrated that graphdiyne is impermeable to water and ions, even at pressures as high as 250 MPa, suggesting that it is unsuitable for desalination applications.215 However, in 2020, Baghbani et al. conducted MD simulations of pristine graphdiyne membranes (without surface functionalization), using DFT-derived partial charges. Their work confirmed impermeability at pressures below 150 MPa but revealed that water is permeable at higher pressures, with flux increasing from 150 up to 650 MPa. Hence, salt rejection of 100% was observed at pressures below 400 MPa and 99.41% at 400 MPa. These results highlight the strong potential of pristine graphdiyne nanosheets for seawater desalination.216
To summarize, graphynes and graphdiynes are excellent 2D materials that could be very promising for water purification techniques for the removal of pesticides, heavy metals, dyes, and are especially useful in the case of desalination of brackish water. Despite great performances, the economic aspect related to their synthesis could act as a deterrent for commercial viability.
2.4 Fullerenes
Fullerenes are carbon nanomaterials that have distinct features, such as hollow, cage-like structures, which were discovered in 1985 by Kroto et al. when a graphite disc was vaporized on irradiation by a laser, and the carbon structures were studied.217 These nanomaterials have unique features, which include excellent thermal and electrical conductivity, electronic capabilities, and tensile strength.218 Fullerenes are unique carbon allotropes with an even number of C atoms, which are sp2 hybridized and range from C20 to C100, arranged in a hollow structural framework. C60 and C70 are more common than the other fullerenes, which are C20, C24, C180, etc., and they can be synthesized in varied dimensions and assemblies (Fig. 20). The carbon atoms have high symmetry and are quite stable, being a truncated icosahedron structure. According to Euler's isolated pentagon rule, a fullerene structure can contain multiple hexagonal faces, with a C2n molecule consisting of (n − 10) hexagons. The C60 fullerene, which is quite stable and obeys this principle, has n = 30, resulting in 20 hexagons and exactly 12 pentagons.219 | ||
| Fig. 20 Structure of a C60 fullerene molecule showing a highly symmetrical spherical geometry composed of 60 carbon atoms arranged in 20 hexagons and 12 pentagons, and its synthesis in varied dimensions. Reproduced from ref. 220, copyright [2020]. | ||
Fullerene molecules are durable and hard, with a high electron affinity, large molecular volume, a high surface-to-volume ratio, and strong resistance to degradation. These properties make them versatile nanomaterials with wide-ranging applications in nanotechnology, including drug delivery, photovoltaic cells, and catalysis.221 Fullerenes can act as electrophiles or electron-accepting groups, as there is poor electron delocalization due to the lack of double bonds in the pentagonal rings. Doping fullerenes with alkali or alkaline earth metals can induce superconductivity properties, and some fullerenes exhibit ferromagnetism, a permanent magnetic property.222 These properties have attracted significant research and offer exciting possibilities in technology. Fullerenes are synthesized by the arc discharge technique, which involves passing a high current through graphite electrodes in a chamber filled with inert gas, forming clusters of carbon atoms that condense into fullerenes; generally, 90% are C60 and 10% are C70 molecules, which can be separated by dissolving in aromatic solvents.223 Laser ablation involves the high-powered laser vaporization of graphite, cooling, and condensing carbon atoms. The field-induced high-intensity plasma method is considered an attractive option since it involves using an inexpensive powder and non-conductive hydrocarbon gas as the precursor materials.224 Chemical synthesis allows for the functionalization of fullerenes, with the attachment of various groups or molecules to the fullerene structure.225 The fullerenes behave like aromatic hydrocarbons as well as straight-chain alkenes. Since fullerenes have a high surface-to-volume ratio and can be functionalized, they are suitable adsorbate materials. The unique structure of fullerene, essentially C60 or the functionalized fullerene molecule, is capable of adsorbing a plethora of pollutants and impurities present in wastewater, including organic pollutants like benzene, toluene, xylene, ethylbenzene, etc.226 The fullerenes are also selective and sensitive for VOCs.227 An attractive feature of C60 is its ability to be derivatized into various forms, allowing the customization of its properties for specific applications. The spherical π-conjugated structure of C60, along with its significant pyramidalization angle, promotes chemical functionalization and enables its dissolution in a wide range of solvents. C60 can be functionalized with surface groups such as epoxy, carboxyl, and hydroxyl, which have the ability to interact with and attract water molecules. These functional groups provide strong adsorption capabilities, allowing interactions with organic molecules via covalent or non-covalent bonding. This makes functionalized fullerenes effective in capturing heavy metals, salts, oils, contaminants, and pathogens. The adsorption sites are limited to the fullerene surface, which contributes to rapid sorption kinetics. Moreover, fullerenes are composed of conjugated unsaturated carbon atoms arranged in a three-dimensional structure with surface functional groups. This structural uniformity results in a more consistent sorption energy distribution and further enhances the rate of sorption processes.228 The sorption of EDCs on the fullerene surface has been observed in a few studies.229,230 EDCs, such as BPA, having a butterfly-like structure, were reported to adsorb onto the curvature of fullerene as per molecular simulation studies. The π–π EDA system is the most probable critical mechanism for aromatic ring structures to adsorb on fullerene.231 Pan et al.232 showed that there was a relation between the sorption coefficient K and the surface area of the nano-adsorbent. The sorption of metal ions such as Cu, Pb, Ni, Zn, Ag, etc., has been tried on fullerenes; their sorption on the C60 surface is governed by different mechanisms. Silver ions are reduced on the surface, lead ions show a donor–acceptor relationship, while copper can be reduced and form complexes with the functional groups on the fullerene surface. Due to electron delocalization, fullerenes are strong electron acceptors and can absorb 6 electrons. The energy band gap (Eq) between the HOMO and LUMO for the different fullerenes lies between 1.5 and 1.98 eV. Thus, fullerenes can absorb UV light as well as visible light and act as an effective photocatalyst. Fullerene solutions can generate reactive oxygen species. The ROS can induce photocatalysis in chemical reactions. To prevent the quenching of excited states due to the aggregation of C60 molecules, the fullerenes need to be immobilized onto solid supports. When the fullerene was supported on semiconductor supports such as TiO2, MoS2, CdS, WO3, and ZnO, the photocatalytic activity was enhanced, and when supported on non-semiconductor supports, it can show self-photocatalytic activity; thus, fullerene-supported photocatalysts can be suitably applied in wastewater treatment. The fullerene/semiconductor photocatalysts can be synthesized by methods such as sol–gel, hydrothermal, simple adsorption, ball milling, ultrasonication, etc. When the TiO2/fullerene combination is used in visible light, not only is the adsorption of visible light enhanced but also the lifetime of the photo-induced carriers is enhanced for redox reactions; aggregation is also lowered. Thus, effective TiO2/C60-induced MB dye degradation was observed by Katsumata et al.233 Cho et al. observed that TiO2/C70 nanowires displayed better light harvesting properties than TiO2/C60 and, accordingly, greater MB degradation was observed for the former composites, such as TiO2/WO3/C60, TiO2/CdS/C60, and TiO2/CdSe/C60. The CdS-based composites showed maximum light absorption and also successfully degraded MO dye to the maximum extent.234 In a study by Vallant et al., C60 bound to silica was used for the solid-phase extraction of certain phosphopeptides and other biomolecules.235 Zhu et al. prepared a nanocomposite of C60 with ZnO to overcome the limitations of the photocorrosion of ZnO as well as to improve its photocatalytic performance. The resultant nanocomposite, having 1.5% C60, exhibited a stellar performance of 95% MB degradation, more than twice the efficiency of bare ZnO.236 Similarly, Behera et al. prepared a ZnFe2O4-fullerene photocatalyst that showed improved photocatalytic performance compared to ZnFe2O4 alone.237 In yet another study, a well-known semiconductor, CdS, which responds well in visible light and has a band gap of ∼2.2 eV, was coupled with C60. Cai, Hu et al. carried out a one-pot hydrothermal synthesis, and the resulting CdS/C60 composite exhibited higher photocurrent density and subsequently higher photocatalytic efficiency (97%) towards RhB degradation.238 A C60-modified Bi2WO6 photocatalyst exhibited 5 times higher photodegradation of MB.239 Along similar lines, the graphitic carbon nitride g-C3N4, a good organocatalyst with a band gap of 2.5–2.7 eV, shows good response in visible light. Its photocatalytic efficiency is restricted due to the recombination of charge carriers as well as inadequate solar absorption. Chai et al.240 introduced C60 into graphitic C3N4, which resulted in enhanced visible light absorption and decreased recombination of electron–hole pairs. Further, the dye RhB was effectively degraded by 97% in 60 min. The series of steps involved in photocatalysis has been explained in the following steps (i)–(iv):
| • Step 1: C60–C3N4 on interaction with light gives C60(e−) + g-C3N4 (h+) | (i) |
| • Step 2: C60(e−) + O2 → C60 + ˙O2− | (ii) |
| • Step 3: ˙O2− + 2e− + 2H+ → ˙OH + OH− | (iii) |
| • Step 4: RhB + h+ (˙O2−, ˙OH) → Products | (iv) |
By thermal treatment, Bai et al.241 prepared the C60–gC3N4 nanocomposite and tested it towards phenol and MB degradation. They observed a change in the band gap and reported thrice the efficiency of degradation of these compounds compared to pristine g-C3N4. It was also reported by them that the radical species generated in these reactions were potent enough to destroy E. coli bacteria present in a solution on irradiating with visible light. Thus, for treating wastewater, photocatalytic materials need to be cost-effective, demonstrate high photocatalytic efficiency, stability, and non-toxicity. Alheety et al. developed a fullerene-based nanocomposite and applied it in a magnetic solid-phase extraction method for the detection and removal of arsenic from drinking water samples collected from rivers, tap wells, and crude oil fields in Iraq.242 In yet another study by Baena et al., a C60–Na DDC derivative in toluene medium was prepared and assessed for lead detection at limits of 4–15 ng L−1. It showed stability, selectivity, and high adsorption efficiency as a sorbent material for metallics and organometallics at trace levels.243 Further, the photodegradation of organic contaminants and endocrine disruptors was checked using a tetrakis amino C60/silica photocatalyst, and it was observed to be effective for ranitidine, propranolol, trimethoprim, etc. Moreover, at pH = 10, the degradation of acetaminophen, BPA, and 4-chlorophenol was observed under fluorescent irradiation.244 The same photocatalyst was evaluated for oxidative degradation of some of the ECs, including pharmaceuticals such as acetaminophen, cimetidine, carbamazepine, ranitidine, propranolol, sulfisoxazole, etc. Kyriakopoulos et al. prepared a C60/MCM-41 photocatalyst using a dry impregnation method. The sample containing 3% by wt. of C60 showed 75% degradation of Orange-G. Fullerene-based photocatalysts could perform photocatalysis using O2, and such composites are promising materials for environmental remediation.245 In a study by Alekseeva et al., a fullerene-polystyrene composite was prepared by dissolving PS in a solution of C60 in o-xylene, with varying concentrations (0.01–0.1 wt%) of fullerene, making films, and evaporating the solvent. These composite films showed the effective extraction of heavy metal ions from aqueous electrolyte solutions, and the Langmuir model was used to show equilibrium adsorption of Cu ions by fullerene.246 Berezkin et al. found the adsorption of organic pollutants from pharmaceutical plant waste waters by C60 to be more effective as compared to soot or activated carbon.247 This was attributed to physical adsorption through dispersive interaction forces. The work of Cheng et al. proposed that the adsorption of two organic contaminants, namely, naphthalene and DCB, occurs not only on the fullerene surface but also an irreversible adsorption occurs within the aggregates of the C60 molecule.248 It was as early as 1994 that lead traces present in water were adsorbed on a C60-ammonium pyrrolidine dithiocarbamate complex, and Gallego et al. concluded that C60 had a much better adsorption capacity than AC or ODS.249 In the chapter written by Bottani et al., adsorption studies on fullerene were compiled, concluding that the efficiency and selectivity shown by C60 and C70 molecules resulted because they are good sorbents for the preconcentration of analytes and are especially suited for inorganic and organometallic compounds.250 Fullerenes have also been effectively used in polymeric membranes for the removal of contaminants from water. Industrial, ground, or domestic water has been treated with such nanocomposites for contaminant removal. The membrane characteristics, such as permeability, fouling, selectivity, pore size, stability, heat resistance, etc., have been studied. The sorption of species results from their penetration into the fullerene lattice or through the defects that act as micropores.251 A variety of nanocomposite filtration membrane technologies, such as microfiltration, ultrafiltration, and nanofiltration, RO, etc., are cutting-edge technologies, and fullerene covalently bonded to polymer matrices leads to an improvement in the properties of the nanocomposite characteristics. C60 as a nanofiller has been added to, say, polystyrene to make composite films for the removal of Cu(II), obeying the Langmuir model for the removal of Cu(II) with a sorption capacity of 14.6 mmol g−1.252 Being cost-intensive, fullerenes could be used in smaller amounts and decorated on AC, zeolites, etc. Cheng et al. studied the adsorption–desorption of naphthalene, a common organic pollutant, on C60 small aggregates, large aggregates, and C60 thin films. Distribution coefficients were found to be less than in the case of AC and in the following order: C60 large aggregate < C60 thin films < C60 small aggregates. The enhancement in dispersal was found to have a substantial effect on the adsorption properties.253 Toxic metals like Pb, Hg, As, etc., have been removed using fullerene-decorated polymeric membranes.254,255 Plisko et al. added 5% by wt. of hydroxy-functionalized C60 to polyamide membranes for water remediation, and the addition of the nanofillers was observed to give antifouling properties.256 Dmitrenko et al. prepared polyamide polyphenylene isophthalamide composite membranes having C nanofillers by solid-phase synthesis. The transport rate and permeation flux were enhanced by adding C60 particles to these membranes. Nafion with C60 molecules interspersed in the polymer was prepared, and the porosity and permeability were affected by the crystallinity and the dispersion of the particles in the Nafion matrix, which served as pores in the membrane.257 Tailoring the pore size, wettability, surface roughness, and other physical properties in polymer/nanomaterial composite needs to be standardized to achieve high-quality membranes with high selectivity. The polymer/fullerene nanocomposite membrane paves the way for improvement in the membrane surface properties, selectivity, and long-term stability.258 Further, porosity, selectivity, and permeability were dependent on the wt% of C60 and its bonding with the polymer membrane. Liu et al.259 made an epoxy-based nanocomposite membrane tailored with GO and grafted C60 molecules and used it for water desalination, as seen in Fig. 21(a–g). The membrane displayed a high water flux, up to 10.85 L h−1 m−2 bar−1. Ciotta et al. synthesized 2D carbon nanoparticles by the oxidative unfolding of the C60 fullerene. The optical properties, i.e., the UV absorption and PL, were studied for these particles in the absence and in the presence of Cu2+ and Pb2+ metal particles in solution. The increase in absorbance and the decrease in fluorescence intensity were related to the aggregation process, which finally resulted in the precipitation of the metal ions.260 Colloidal fullerenes have been reported to show inhibitory action against bacteria in aerobic and anaerobic conditions.261 Markovic et al.262 proposed that fullerene could generate ROS in the presence of oxygen and light, or could disrupt the electron transport or energy mechanism. Alvarez et al. proposed that aqueous fullerene suspensions induce oxidative stress in bacteria through a mechanism independent of ROS, supported by evidence of protein oxidation, alterations in cell membrane potential, and disruption of cellular respiration.263 In contrast to this, it was found recently in yet another study done by Zhang et al., the effects of a fullerene colloidal suspension on a variety of microorganisms. They found that the bacteria from the Acidovorax, Bacillus, and Cloacibacterium genera were enriched in the presence of C60 in a slightly acidic medium in aerobic conditions. They also observed that the C60 suspension caused an increase in denitrification in B. cereus S45.264 Therefore, fullerenes have been utilized in engineered systems to photocatalytically oxidize organic contaminants and to potentially inhibit or deactivate microbial activity. Thus, fullerene molecules contribute to enhancing the properties of the nanocomposites, affecting porosity, selectivity, permeability, transport rate, flux, and mechanical properties, anti-fouling, and improved surface properties.
 | ||
| Fig. 21 C60-grafted graphene oxide (GO) membranes for water desalination. (a) GO membrane without C60, (b) C60-grafted GO membrane, (c) optical micrograph of the cross-sectional area for GO laminates embedded in 81 μm thick epoxy, (d) GO–C60 membrane encapsulated with epoxy, (e) GO–C60 membrane inside the water desalination setup. (f) and (g) Schematic of the setup of a flat membrane made of GO and the C60 hybrid. Reproduced from ref. 259 with permission from the American Chemical Society copyright [2018]. | ||
To summarise this sub-section, fullerenes, the hollow cage-like carbon nanostructures, with their synthesis methods and their mode of operation to purify water, have been discussed. The functionalization of fullerene to result in suitable adsorbate materials capable of removing organic pollutants, solvents, EDCs, and heavy metals has been explained. In combination with semiconductors, self-photocatalytic activity examples have been cited to degrade dyes and other organics. Nanocomposites formed using C60 function well for the capture of metals and organometals; C60-decorated polymer membranes exhibit anti-fouling and serve as high-quality, stable membranes.
2.5 Carbon quantum dots
The exciting family of carbon-based nanoparticles known as carbon quantum dots has special characteristics and a wide range of applications. CQDs are a fascinating class of carbon nanoparticles with remarkable properties and versatile applications in various fields. Initially discovered in 2004, they have since been recognized as a novel class of fluorescent, small carbon nanomaterials with particle sizes less than 10 nm.265 Carbon quantum dots emit fluorescence when exposed to ultraviolet or visible light. This property is particularly attractive for bioimaging applications, where deep tissue bioimaging with deep red emission is particularly critical.266 Deep-red emissive carbonized polymer dots with exceptional FWHM have been successfully synthesized by researchers, showing a high quantum yield and outstanding biocompatibility.267 Carbon dots have found extensive biomedical applications due to their water solubility, less toxic nature, and biocompatibility. Their tunable fluorescence emission and excitation properties make them versatile probes for various sensing applications in fields like food analysis. The synthesis of carbon dots (Fig. 22(a and b)) has been explored through various methods, including hydrothermal, solvothermal, and microwave-assisted processes.267–269 Additionally, scientists have developed inventive techniques for creating carbon dots from natural resources like banana juice, watermelon peel, and pomelo peel, providing quick, affordable, and environmentally friendly alternatives. The properties may be affected by changes in the size of carbon dots and in the solvent or functionalization.270,271 As research in this field progresses, carbon dots are expected to revolutionize various industries, contributing to advancements in bioimaging, environmental protection, energy storage, sensing technologies, and more. CQDs have been synthesized using various synthetic methods, classified as top-down or bottom-up approaches. Top-down and bottom-up approaches used for synthesizing CQDs use small, scalable, and cost-effective materials. Fig. 22(a and b) reveals schemes related to the top-down and bottom-up strategies for CQD synthesis. The top-down method involves breaking down macromolecules into small CQDs through physical or chemical methods, while the bottom-up method involves polymerizing, building up, and carbonizing small molecules through chemical reactions. Microwave-assisted synthesis is a potential method for synthesis, reducing time and enhancing particle homogeneity.272 The arc discharge is another method used to synthesize CQDs decomposed from carbonaceous precursors in an anodic electrode driven by plasma gas generated in a sealed reactor.273 Laser ablation uses a high-energy laser pulse to irradiate the surface of the target to a thermodynamic state, rapidly heating it and evaporating it into a plasma state, and then crystallizing the same into nanoparticles. The as-obtained CQDs exhibit visible and tunable photoluminescence (PL), but the polydispersity may limit the specific surface area of CQDs during the electrocatalytic process. The process of acid oxidation is commonly employed for the transformation of bulk carbon into nanomaterials and introducing hydrophilic groups into the synthesized carbon dots. Microwave pyrolysis is a well-established bottom-up approach for the synthesis of CQDs, while hydrothermal/solvothermal synthesis involves dissolved organic molecules and polymers in water or solvent. Electrochemical methods easily tune particle size and PL performance. CQDs are thus carbon-based nanomaterials that possess remarkable optical properties, biocompatibility, cost-effectiveness, and easy synthesis, along with tuneable functionalizations and high quantum yields.274 Bawendi, Brus, and Yekimov received the Nobel Prize in 2023 for their work on CQDs.275 Owing to their large surface area, low toxicity, water solubility, and fluorescence properties, they can be very well used in water purification treatments. Table 2 presents various methods adopted by researchers for the synthesis as well as applications of CQDs. | ||
| Fig. 22 (a and b) Approaches for CQD synthesis. Reproduced from ref. 267 with permission from Elsevier Ltd copyright [2023]. (c) Photocatalytic behaviour of CQD with TiO2. Reproduced from ref. 268 with permission from RSC copyright [2018]. (d) Effects of solvents and increasing size of CQDs, resulting in an increasing degree of surface oxidation. Reproduced from ref. 269a and b with permission from ACS copyright [2016]. | ||
| CQD | Materials used for the preparation of CQDs | Method adopted & features | Application(s) | Ref. |
|---|---|---|---|---|
| 1 | Urea with sugarcane bagasse, carboxymethyl cellulose (CMC), or cellulose | One-step synthesis by a microwave heating method | Adsorption of Pb(II) from an aqueous solution containing CQDs. %Efficiency >96% | 276 |
| 2 | Action of ammonia from petroleum coke waste | Hydrothermal method | Monodisperse sub-5 nm CDs having 0.2% S and a 10% N-doped chitosan with CQD membrane and the capture of Cd2+ in alkaline medium, with the adsorption capacity of 112.4 mg g−1 at room temp | 277 |
| 3 | N-doped CQDs (N-CQD) produced from grass | Hydrothermal method | N-CQD that can adsorb 37% Cd2+ and 75% Pb2+ | 278 |
| The N-CQDs have high surface activity | The N-CQDs degrade six different types of dyes | |||
| 4 | Fluorescent CQDs made by using muskmelon peel as a starting material | Under ultrasonication | The CQD breakdown of Rh B was noted to be 99% under sunlight | 279 |
| 83.04% efficiency | ||||
| 5 | Waste rubber seed shells used for making CQDs | Numerous oxygen-functional groups (OH, COOH, and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O) on the CQD surface O) on the CQD surface |
The treatment of wastewater having both Congo Red and MB dyes, with clearance being 77% and 75%, respectively | 280 |
| 6 | N,S-co-doped CQDs produced using ammonium citrate/citric acid and L-cysteine as raw materials | Hydrothermal process. The change in fluorescence for the identification of phosphate in urine and surface water was observed, so quantitative analysis was also possible | An Fe3+ detection limit of 14.0 nM (∼0.8 μg L−1) was achieved using CQDs that required no excitation and gave 69% fluorescence quantum yields when a weak acid was used | 281 |
| 7 | N-doped CQD modified with Fe3O4 | Detection of Pb in various water samples | Removal of lead ions in vegetable and water samples | 282 |
| 8 | TiO2 and CQDS with a size of 5 nm were prepared | The hydrothermal technique was used | CQDs or CQD/TiO2 composites were used for the photocatalytic degradation of MO, which was reduced in concentration with time | 283 |
| Photocatalytic activity by CQDs/TiO2 under visible light (Fig. 22(c)) | ||||
| 9 | CQD/TiO2 containing a waterborne hyperbranched polyester nanocomposite | CQD semiconductor nanocomposites | Photocatalytic degradation of paraoxon ethyl pesticide under sunlight with an efficiency of 91% | 284 |
| 10 | Development of carbon dot-modified polyethersulfone membranes | Nanocomposite membranes | Introducing the carbon dots into PES membranes for wastewater treatment applications | 285 |
| 11 | CQDs on NH2-MIL-125 | High photocatalytic breakdown of organic contaminants under a broad spectrum | Photocatalytic action equated to NH2-MIL-125 for RhB breakdown in full-spectrum or even near-infrared light | 286 |
| 12 | Nitrogen-doped CQDs (N-CQDs) | High-quality N-doped adsorbent | Adsorption of Cd2+ and Pb2+ ions present in water | 287 |
| 13 | Nanocomposite (PECQDs/MnFe2O4) prepared by merging the polyethyleneimine CQDs with magnetic materials | The absence of CQD led to only 25% absorbance of U+ ions | Adsorption for the effective removal of uranium, ∼91%, by the nanocomposite | 288 |
| 14 | Fe3O4@NCQDs NCs were made via a co-precipitation reaction | Hydrothermal method to make NCQDs from lemon juice | Rapid removal of MB dye with over 90% efficiency | 289 |
| 15 | Iron oxide adsorbents: Fe3O4, C11–Fe3O4, and CQDs/C11–Fe3O4 have been synthesized | CQDs/C11–Fe3O4 adsorbs 93.9% of the benzopyrene | Eliminated benzopyrene from wastewater samples | 290 |
| 16 | Nitrogen-doped CQDs (NCQDs) have been prepared using dicyandiamide and citric acid | High-quality N-doped adsorbent | Adsorbent having NCQDs with iron oxide (Fe3O4) for the removal of Pb | 291 |
| 17 | CQDs having zinc–aluminum layered double hydroxide (CQDs/ZnAl-LDH) | N doped on the CQDs/ZnAl-LDH composite, adsorbed Cd2+ ions | After 20 minutes, the max. amount of Cd adsorbed on the CQDs/ZnAl-LDH composite was 12.60 mg g−1 | 292 |
Photocatalysis, being a versatile and efficacious green approach, is used to degrade organic pollutants present in the wastewater. To achieve the photocatalytic transformation of pollutants, a semiconductor such as CQD must absorb light of a certain wavelength range, causing an electron to be excited from the valence band to the conduction band. Eqn (v) shows the formation of positive holes (h+) in the VB of the photocatalyst:293
| Photocatalyst + hv → h+ VB + e−CB | (v) |
As seen in eqn (vi) and (vii), the electrons in the conduction band are captured by oxygen (O2) molecules to generate superoxide radicals (˙O2−), whereas the holes in the valence band split water molecules, producing hydroxyl free radicals.
| h+ VB + H2O → H+ + ˙OH | (vi) |
| e−CB + O2 → ˙O2− | (vii) |
According to eqn (viii)–(x), more species-to-species interactions may take place, resulting in the production of more ˙OH radicals.
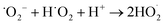 | (viii) |
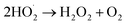 | (ix) |
| H2O2 → 2˙OH | (x) |
| ˙OH + EDC → CO2 + H2O | (xi) |
According to eqn (xi), pollutants that are adsorbed on the catalyst surface can be degraded by hydroxyl radicals to produce CO2 and H2O. Limitations related to the application of CQDs in water treatment involve expenses, scaling up, purity, etc., thus giving opportunities for scientists to overcome these limitations.294
In brief, CQDs have unique properties and are prepared by both top-down and bottom-up methods. They are capable of removing organic pollutants, heavy metals, dyes, etc., by processes such as adsorption, electrostatic forces, and coordination bonds. The action of CQDs as photocatalysts to produce ROS and break down pollutants makes them useful as biocompatible purifiers.
2.6 Nanodiamonds
Nanodiamonds (NDs) are tiny crystals with dimensions ranging from 1 to 100 nanometres in size. They are complex structures, as depicted in Fig. 23(A(a)), consisting of layered arrangements of carbon atoms. They have three sections: (1) the outer surface with functional groups, (2) the core, and (3) the shell's innermost face containing graphitic monolayer carbons and detached rings.295 | ||
| Fig. 23 A(a) Representation of the core shell structure of a nanodiamond. A(b) Functional groups attached to the surface of a ND. Reproduced from ref. 295 with permission from [Taylor & Francis] copyright [2015]. B(a) TEM along with the AFM images of aminated NDs and B(b) pristine NDs. B(c) FTIR spectra of NDs (red spectra: aminated nanodiamonds and blue spectra: pristine nanodiamonds). HR-XPS spectra of C, O and N for B(d) aminated NDs and B(e) pristine NDs. B(f) XRD spectra for aminated and pristine NDs. Reproduced from ref. 317 under CC BY-NC 4.0. (C) Illustration of nanodiamond adsorbents tailored with single- or double-arm amide-based thiourea chains for the separation of uranium metal based on spatial configuration and chelate ability. Reproduced from ref. 327 with permission from Elsevier copyright [2016]. | ||
Nanodiamond synthesis involves various methods to produce unique diamond nanoparticles with potential applications. The detonation method, discovered by Russian scientists through accidental explosions, involves detonating an explosive mixture, resulting in ultrafine nanodiamond nanoparticles at high temperatures.296 Other synthesis techniques include laser ablation, chemical vapor deposition, ultrasound cavitation, electron irradiation, high-pressure high-temperature synthesis, and autoclave synthesis. Laser ablation, HPHT, and detonation methods have been economically used for nanodiamond synthesis.297 In comparison with CNTs and other carbon nanoparticles, nanodiamonds stand out due to their unique functional groups, including carboxyl, hydroxyl, carbonyl, nitrogen, ketones, ethers, and sulfur, on their surface. Moreover, NDs exhibit several remarkable features, such as hydrophilicity, biocompatibility, chemical and mechanical stability, nontoxicity, low electron affinity, small primary particle size (averaging 4 to 5 nm), inertness, enormous surface area (300–500 m2 g−1), and exceptional adsorption capacity; these features make them a promising candidate for water purification & treatment.298 The ability of the ND surface to be functionalized by C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O, OH, COOH, amines, ether groups, etc., is shown in Fig. 23(A(b)). They can act as coupling agents and further attach to various functional groups, improving the adsorption capability of NDs for organic pollutants such as dyes and drugs by H bonding, electrostatic interactions, and π–π interactions between aromatic substances and surface π-conjugated structures of ND. NDs are nanosized versions of bulk diamond, having both sp3 and sp2-hybridized carbon atoms. Due to sp2 C shells, higher electrical conductivity is possible, and the greater the number of sp2 to sp3 hybridized C atoms, the more the band gap is reduced. An increased number of sp2 C atoms can be obtained by annealing the NDs in an inert atmosphere.299 Thiol-functionalized NDs coupled with Au nanoparticles have been used for sensing Cr(III) ions and β-adrenergic clenbuterol drugs in lake and tap water. A wine-red coloration changed to purple-blue due to coordinate bonds formed between the N–H and C
O, OH, COOH, amines, ether groups, etc., is shown in Fig. 23(A(b)). They can act as coupling agents and further attach to various functional groups, improving the adsorption capability of NDs for organic pollutants such as dyes and drugs by H bonding, electrostatic interactions, and π–π interactions between aromatic substances and surface π-conjugated structures of ND. NDs are nanosized versions of bulk diamond, having both sp3 and sp2-hybridized carbon atoms. Due to sp2 C shells, higher electrical conductivity is possible, and the greater the number of sp2 to sp3 hybridized C atoms, the more the band gap is reduced. An increased number of sp2 C atoms can be obtained by annealing the NDs in an inert atmosphere.299 Thiol-functionalized NDs coupled with Au nanoparticles have been used for sensing Cr(III) ions and β-adrenergic clenbuterol drugs in lake and tap water. A wine-red coloration changed to purple-blue due to coordinate bonds formed between the N–H and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O groups of ND with Cr(III) ions or clenbuterol molecules.300 Soylak et al. used magnetic, carboxylated NDs as solid-phase adsorbents to preconcentrate Zn dimethyldithiocarbamate and various common heavy metal ions such as Co(II), Cd(II), and Pb(II), and their extractive efficiency was investigated using real-life water samples. The extraction efficiency was further improved by producing Fe3O4@ND@GO hybrid adsorbents for the preconcentration of a drug called sildenafil citrate.301 Several researchers have reported that dyes and drugs adsorb on ND surfaces via electrostatic attraction, for example, acid orange 7 (azo compound), sulfapyridine, fluorescent dyes, sulfasalazine, and 5-aminosalicylic acid, in wastewater at pH 1–6.302,303 The present purification techniques are unable to purify hot wastewater produced by certain industries, such as oil recovery. The traditional RO method entails the use of cold water; thus, tremendous energy is wasted to cool it and then to reuse the water that needs to be heated for oil recovery. Behnam Khorshidi304 and his colleagues reported the synthesis of a thicker and thermally stable thin-film nanocomposite membrane by embedding nanodiamonds in polyamide, such that the hydrophilic surface of the nanodiamond augmented the surface wettability of the thin-film nanocomposite membrane, and the incorporation of nanodiamond into the polyamide reduced the contact angle from 88.4° for the pristine membrane to 58.3° in the nanocomposite membrane. The average roughness of the membranes increased from 108.4 nm (pristine membrane) to ∼178 nm nanocomposite membranes. The nanodiamond-modified membranes displayed a much higher pure water flux of about 76.5 LMH than the pristine membrane (17 LMH) at RT and 220 psi.
O groups of ND with Cr(III) ions or clenbuterol molecules.300 Soylak et al. used magnetic, carboxylated NDs as solid-phase adsorbents to preconcentrate Zn dimethyldithiocarbamate and various common heavy metal ions such as Co(II), Cd(II), and Pb(II), and their extractive efficiency was investigated using real-life water samples. The extraction efficiency was further improved by producing Fe3O4@ND@GO hybrid adsorbents for the preconcentration of a drug called sildenafil citrate.301 Several researchers have reported that dyes and drugs adsorb on ND surfaces via electrostatic attraction, for example, acid orange 7 (azo compound), sulfapyridine, fluorescent dyes, sulfasalazine, and 5-aminosalicylic acid, in wastewater at pH 1–6.302,303 The present purification techniques are unable to purify hot wastewater produced by certain industries, such as oil recovery. The traditional RO method entails the use of cold water; thus, tremendous energy is wasted to cool it and then to reuse the water that needs to be heated for oil recovery. Behnam Khorshidi304 and his colleagues reported the synthesis of a thicker and thermally stable thin-film nanocomposite membrane by embedding nanodiamonds in polyamide, such that the hydrophilic surface of the nanodiamond augmented the surface wettability of the thin-film nanocomposite membrane, and the incorporation of nanodiamond into the polyamide reduced the contact angle from 88.4° for the pristine membrane to 58.3° in the nanocomposite membrane. The average roughness of the membranes increased from 108.4 nm (pristine membrane) to ∼178 nm nanocomposite membranes. The nanodiamond-modified membranes displayed a much higher pure water flux of about 76.5 LMH than the pristine membrane (17 LMH) at RT and 220 psi.
Colon et al. synthesised and functionalized nanodiamonds, which were embedded into polyether sulfone and polyvinylidene membranes to be used for wastewater filtration. The incorporation of nanodiamonds into a polymer matrix was observed to show bactericidal reduction in comparison with the commercial membranes. The presence of nanodiamonds could influence water permeability due to their hydrophilic nature and their effect on membrane structure. The addition of nanodiamonds could potentially affect the pore size and distribution. As the loading content of NDs was increased from 0 to 1 wt%, the water content values of the nanocomposite membranes increased from 66.3% to 77.2%. These results suggest that the addition of nanodiamonds to polymer matrices can enhance their hydrophilicity and water flux without causing significant negative effects on their structural or separation properties.305 Molavi et al. reported that the thermal oxidation of NDs increases carboxylic acid content, improving their ability to adsorb anionic and cationic dyes. NDs enhance mechanical strength, thermal stability, and antifouling properties, making them valuable for improving membrane performance in filtration applications.306 In another study, detonation nanodiamonds307 containing multiple hydrophilic surface functional groups were utilized to prepare polyethersulfone (PES) nanofiltration membranes. To create the PES–DNDs nanocomposite membranes, the phase inversion method was employed. The incorporation of DNDs into nanocomposite membranes, even at low loading contents, has been shown to enhance their hydrophilicity. The addition of DNDs was shown to influence several structural characteristics of the membrane, including the porosity, mean pore size, and water content. As the DND loading content increased to 1 wt%, these properties were altered considerably. For example, the water content of the nanocomposite membranes increased from 66.3% to 77.2%. This increase suggests that the DNDs facilitate a greater retention of water within the membrane structure, likely due to the enhanced hydrophilic interactions provided by the DND surface functional groups. Furthermore, the membranes were tested for bovine serum albumin (BSA) filtration and dye separation in synthetic wastewater. A 0.5 wt% loading of DNDs in nanocomposite membranes achieved the highest dye removal efficiency for both C.I. Reactive Green 19 and C.I. Reactive Orange 29. A study by He and group explored the use of a B-doped nanodiamond film electrode in decolorizing an acid yellow azo dye through electrochemical polarization. Anodic polarization was found to be more effective than cathodic polarization as an electrode pre-treatment. The anodic polarization led to the transformation of the H-terminated surface of the B-doped nanodiamond into an O-terminated one, making it more hydrophilic. Moreover, the oxidation of surface functional groups and sp2 carbon occurred during anodic polarization, activating the electrode and significantly improving its electro-oxidation capability towards the acid yellow dye. Even after several cycles of reuse, the anodically polarized boron-doped nanodiamond maintained its ability to totally decolorize the dye.308 A study was conducted by Zhu et al. from Peking University using a boron-doped diamond electrode to explore the electrochemical oxidation of p-substituted phenols with various electron-withdrawing and -donating groups. The researchers performed both bulk electrolysis and voltammetry to investigate the degradation process. They found that the primary mechanism involved indirect electrochemical oxidation facilitated by OH radicals generated on the boron-doped diamond electrode. Furthermore, the study revealed that phenols with e−-withdrawing groups degenerated more efficiently than those with e−-donating groups, as the former were more readily released during the oxidation process.309 The TiO2@ND composite photocatalyst synthesis was carried out using a precipitation method. The study aimed to investigate in detail the impact of ND loading on the properties and action of TiO2. Characterization studies of the samples of TiO2@ND composites were also done. Further, the researchers examined various factors, such as the loading of TiO2@ND and the pH of the electrolyte, to understand their influence on the photocatalytic activity of the composites in degrading BPA, an endocrine-disrupting substance. It was observed that in neutral and acidic pH conditions, the degradation of BPA was enhanced. Notably, its complete degradation was accomplished using TiO2@ND, a photocatalytic composite with 8 mg of ND loading at pH = 5.1 under UV irradiation.310 Firstly, the presence of NDs enhanced light absorption, leading to more efficient utilization of UV irradiation. Secondly, the composites exhibited effective charge separation, preventing the recombination of photogenerated charge carriers. This resulted in a higher efficiency in generating reactive species responsible for BPA degradation, making the composites promising candidates for various environmental remediation applications and photocatalytic processes. The membranes of PANI/PVDF blended with ND showed better antifouling properties.311 Nanodiamonds were combined with molybdenum disulfide (MoS2) and subjected to a hydrothermal method to obtain the ND/MoS2 nanorod composite. This composite was used as an adsorbent in a vortex-assisted micro solid-phase extraction method to remove Cu(II) and Pb(II). The sorption capacities of the ND/MoS2 nanocomposites were measured as 49.33 mg g−1 for Cu(II) ions and 19.87 mg g−1 for Pb(II) ions.312 There are reports in which microbial moieties are used as substrates. Ozdemir et al. have immobilized Bacillus altitudinis bacteria onto ND and developed a thermophilic biosorbent. In this setup, the NDs act as a substrate for the bacteria, which serve to adsorb heavy metal ions. This biosorbent exhibited impressive adsorption capacities for Hg(II) (19.5 mg g−1), Co(II) (26.4 mg g−1), Cr(VI) (30.4 mg g−1), and Pb(II) (35.2 mg g−1). Further attempts were made on other microbial entities in which Armillaria mellea fungus was immobilized onto NDs and employed as a solid-phase extractor for Hg(II), Zn(II), and Cr(III) ions from diverse food samples.313,314 The unique features of ND as understood consist of high chemical stability, crystallinity, high surface area (300–500 m2 g−1), and high reactivity, making it suitable for water purification and treatment.315,316Fig. 23(C(a and b)) shows the TEM and AFM of (a) aminated (20 mV, positive zeta potential) and (b) pristine nanodiamonds (−8 mV zeta potential). Fig. 23(B(c)) shows the FTIR of aminated (red) and pristine nanodiamonds (blue). Fig. 23(B(d and e)) shows the high-resolution XPS for aminated and pristine nanodiamonds, while Fig. 23(B(f)) depicts the XRD for both functionalized and pristine nanodiamonds, respectively.317 It was also studied by some researchers how the degree of graphitization of NDs with annealing decreased surface charge but exhibited a high adsorption capacity for MB dye. The nanocarbon material, having core–shell sp3@sp2 carbon atoms composed of a diamond core and a graphitic shell, has garnered attention for its core–shell hybrid structure, surface chemistry, and catalytic activity.318
Regarding the removal of heavy metals from wastewater, the functional groups on the surface can play a significant role in adsorbing them on the ND surface. The presence of negatively charged species like hydroxyl or carboxyl groups causes the surface to have a negative charge.319 Thus, metal cations are readily adsorbed on the surface of ND due to the complex formation between opposite charges.320 Yudina et al. reported that complexation occurred between metal ions and the functionalized polar groups on the ND surface in the order of Fe(III) >Al(III) >Cu(II) > Mn(II) >Zn(II) >Cd(II)> Co(II)>Ni(II).321 Further, modified NDs could even adsorb actinide metals, which could be recovered along with heavy metals from water, as well as soil.322,323 Duffy et al. have shown that benzene sulfonic-functionalized NDs could remove MB with an adsorption capacity as much as 385 mg g−1.324 It was reported by Molavi et al. that carboxyl-functionalized NDs have thrice the efficiency of pristine ND to adsorb Cu(II), Pb(II) and NO3− ions. This happens via electrostatic interactions, ion exchange, complex formation of metal ions with the functionalized groups of ND, and physical adsorption of heavy metal ions to intercalated graphite layers.325 Matsumoto et al. developed an efficient DND support for a Cu-Prussian blue analogue that could adsorb hazardous, radioactive Cs+ in diluted artificial seawater and soil wastewater.326 Li and Zao et al. functionalized NDs with single-armed and double-armed ligands, as shown in Fig. 23(C), such that they could adsorb uranium ions from water. Both ND-DA and ND-SA showed good adsorption kinetics, a capacity of ∼200 mg g−1, and good selectivity towards uranium ions.327 Thus, adsorbents on the surface of ND can also selectively capture noble metals based on charge.288,328 ND was covalently functionalized with an ionic polymer on its surface to capture Cu(II) ions by He et al.329 and it was observed that adsorption increased with increasing temperature. A nanocomposite was prepared by surface functionalization of ND with ionic liquid for the adsorption of Congo red dye (CR). Compared to pristine ND, the functionalized ND exhibited around 93% removal efficiency of the dye, and this was possible even after 5 regeneration cycles.330 Hybridisation of 0D ND with 2D GO was carried out to make ND/GO microspheres used for the removal of Pb(II) ions from wastewater. A ratio of 85![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 15 for GO to ND exhibited the highest adsorption capacity for Pb(II) ions (303 mg g−1) as observed by Wu et al.331 The ND-MOF combination also showed improved physicochemical properties and was found to adsorb both cationic and anionic dyes. Such a hybrid structure of (0ND-UiO-X) showed good adsorption performance for MR, MO, MG, MB dyes in single and binary mixtures.332
15 for GO to ND exhibited the highest adsorption capacity for Pb(II) ions (303 mg g−1) as observed by Wu et al.331 The ND-MOF combination also showed improved physicochemical properties and was found to adsorb both cationic and anionic dyes. Such a hybrid structure of (0ND-UiO-X) showed good adsorption performance for MR, MO, MG, MB dyes in single and binary mixtures.332
NDs having a layer of carboxylic acid groups on their surface were used to prepare films with chitosan. These hybrids were tested for the removal of MO from wastewater, and the adsorption capacity was observed to be 454.5 mg g−1.333 Further, these modified NDs were cross-linked with a polymer and showed high adsorption behaviour towards Pb(II) ions.334 CR and MB dyes were removed from wastewater by using a thermally oxidised ND with UiO-66 MOFs.335 Another study by Wang et al. used ND coated with polydopamine and then functionalized with perfluorodecanethiol and attached to a PU sponge.336 It was observed to show high organic absorbance capacity and could separate oil/water mixtures. Further, Wang et al., by a low-cost and simple technique, fabricated ND-ODA@PDMS@MS for the selective removal of oily compounds from water. A method to improve the reactivity of ND towards persulfate ions was conducted via thermal annealing and dopant atom inclusion.337 As graphitisation increased with annealed temperature, the pore volume and surface area increased, and it exhibited a high capacity for persulfate activation. NDs can act as good photocatalysts in wastewater owing to the presence of several oxygen-rich functional groups, intense light scattering by sp3 to enhance the visible light adsorption range due to a narrow band gap.338,339 Khan et al. functionalized NDs with amino groups and used them as photocatalysts for RhB degradation340,341 and observed that annealing NDs improved their photocatalytic activity towards organic dyes. Further improvement in the photocatalytic activity of NDs in water was realised by combining them with photoactive semiconductors. Thus, a CQD/ND heterojunction was found to exhibit 98% photodegradation efficiency towards MB dye under sunlight due to superior charge carrier separation.342 ND-C60 heterostructures produced an aqueous colloidal suspension that produced ROS under visible light, which showed good dispersibility in water and good performance for the removal of furfural.343 Zhou made a series of g-C3N4/ND heterojunction materials, and their photocatalytic tendencies were investigated under UV-Vis irradiation; they showed high photocatalytic activity for MB degradation.344 Wang et al. in 2021 attached Ag nanoparticles to ND so that plasmon resonance energy transfer on the ND surface was observed to be the driving force for MB degradation under visible light.345 In another study, the Ag/ND/g-C3N4 photocatalyst was prepared to effectively degrade RhB dye from wastewater to produce H2 from water splitting. The silver nanoparticles were believed to be an electron reservoir, enhancing visible light absorbance capacity and promoting photogenerated charge separation efficiency.346 The combination of ZnSnO3/ND improved visible light photocatalytic activity such that in 2 h, 90% of TC was degraded. ND/ZnFe2O4 spherical particles also exhibited good photocatalytic activity for TC degradation as reported by Pan et al.347 This is beneficial for wastewater treatment. ND/TiO2 composites were tested for the photodegradation of amoxicillin and diphenhydramine drugs in UV-Vis light. The degradation of 10 ppm of BPA by ND/TiO2 was also reported by Huang et al.348 In another study, the ND/Fe2O3 composite was observed to degrade TC in water.349 Since membranes can be shaped in a variety of sizes and have high separation efficiency, good mass transport ability, reasonable flexibility, and can be scaled up as needed for installation, they are quite popular. Polymeric membranes are convenient, but they have a problem of fouling, and so surface modification by adding NDs is quite reasonable. Vatanpour and group reported incorporating DND particles into polyethersulfone membranes and they exhibited the highest water flux and rejection for dyes like Orange 29 and RG19.307 The same group used 1.5 wt% DND incorporated into a chitosan/PVA membrane, which enhanced the adsorption capacity of Pb ions to 121 mg g−1 from 29.5 mg g−1 for the pristine membrane.350 GO–ND nanocomposites were incorporated into PVC membranes to improve hydrophilicity. The addition of 0.1 wt% of GO + ND improved the antifouling performance.351 In a study by Kim et al., ND as co-catalysts were loaded onto WO3 as effective photocatalysts in visible light for the degradation of VOCs owing to charge separation, interfacial electron transfer, and the ND-induced lower overpotential of O2 reduction. The authors concluded that the graphitic layer over the sp3 diamond core plays an important role in the charge separation and interfacial electron transfer.352 Despite being an outstanding technology, membrane technology suffers from drawbacks such as membrane fouling as an outcome of its hydrophobic nature and/or interfacial structure that affect both performance and lifespan. The modification of the membrane is required to enhance the membrane permeability while maintaining a high solute rejection rate. PVDF is an excellent membrane material, although being highly hydrophobic makes it prone to fouling. Siddiqa et al. reported the fabrication of a PANI/PVDF composite membrane blended with NDs. A series of nanocomposite membranes with ND in the range 1–5% by wt% were blended by the solution casting method. It was observed that membranes with higher wt% of ND exhibited superior filtration efficiency owing to greater porosity, resistance to fouling, higher water flux, and salt rejection, and are suitable for application in wastewater treatment.353 In yet another interesting study carried out by Bland et al., quartz microfiber membranes were loaded with hydrogenated detonation NDs, which were highly electropositive. These composite membranes were used for the removal of viruses and negatively charged contaminant species present in drinking water. The loading of NDs on the membrane surface increased not only the surface area by four-fold but also enhanced the zeta potential of +45 mV at pH = 7. The membrane was effective in the retention of acid black 2 dye, and exhibited a LRV (membrane's log10 reduction value) of at least 6.2 for the MS2 bacteriophage at pH 5 to 9.354
To summarize, the unique structure of nanodiamonds and the synthesis methods such as CVD, ultrasound cavitation, HPHT, laser ablation, and detonation have been discussed. Functionalisation of NDs with –COOH, –OH, and –C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O groups for the removal of heavy metals, dyes, drugs, etc., and their composite formation with semiconductors for photocatalysis has been discussed here. The action of NDs and their adsorption capability for the removal of pollutants and microorganisms has also been summed up in this section.
O groups for the removal of heavy metals, dyes, drugs, etc., and their composite formation with semiconductors for photocatalysis has been discussed here. The action of NDs and their adsorption capability for the removal of pollutants and microorganisms has also been summed up in this section.
2.7 Activated carbon
Although not a nanoform of element carbon, activated carbon is a form of carbon that is prepared from carbonaceous materials. Organic waste materials can be pyrolysed and/or activated such that it results in the formation of a carbon-rich microporous material that is light and fluffy. Activated carbon, being highly porous with microfissures and fractions, has a microscopic sponge-like structure. Due to its highly porous nature and high surface area, it has an enhanced adsorption capacity and is an industrially important absorbent. Adsorption is a surface phenomenon where the contaminants present in the wastewater are adsorbed over the surface of the absorbents due to the force of attraction between them. This adsorption is due to internal pore diffusion, as well as adsorption and desorption phenomena. It is often activated with chemicals like KOH, H3PO4, etc., to enhance its adsorptive properties. AC obtained from a variety of carbon-rich precursors shows structural and morphological differences with divergent porous textures. The structure of AC is quite similar to graphite, with the hexagonal layers of carbon rings being close to each other at a distance of about 0.34 nm. AC is used for industrial wastewater treatment and is classified as powdered and granular. It is used for removing pesticides, organic pollutants, heavy metals, chlorinated compounds, pharmaceuticals, dyes, and is also effective for curbing microbial growth. Not only does it have good adsorption capacity but it can be regenerated chemically or thermally, although costly. The adsorption mechanism on AC is difficult to predict as it depends on various factors such as dispersive, electrostatic, and chemical interactions between the solute and adsorbent. The use of charcoal for water purification dates back to 1500 BC when the Hindus in India used it for water purification and the Egyptians used it as a medical adsorbent in metallurgical operations.The important properties that make AC so useful to remove contaminants and purify water are as follows:
(1) High surface area: surface area exceeding 1000 m2 g−1 on activation. It makes numerous porous sites available for the highly effective trapping of contaminants. The surface area is quite reactive due to the presence of abundant functional groups on the surface.
(2) Porous structure: the AC structure contains a micro- and macro-porous network throughout that makes the adsorption of a wide range of different-sized molecules effective. Microporosity in AC makes it effective for trapping small organic molecules and disinfection byproducts. Pore size distribution is an important criterion for effective adsorption.
(3) High adsorption capacity: AC adsorbs contaminants majorly through van der Waals forces and thus captures organic compounds, Cl2, pesticides, and other contaminants. There are numerous reactive sites available, which are conducive to heteroatom (N, P, S) doping.
(4) Chemical reactivity: the surface of activated carbon can be tailored to capture specific contaminants; thus, the targeted removal of pollutants is possible.
(5) Versatility: AC can be cost-effectively prepared from a variety of carbonaceous materials, such as coconut shells, olive pomace, fly ash, rice straw, wastepaper pulp, wood waste, etc. Each of these provides a different pore structure and possesses unique adsorption characteristics.
These properties make AC a useful material for adsorption applications, especially in water purification to improve the water quality by removing undesirable tastes, odors, and contaminants. Wastewater contains contaminants such as heavy metals,355 pharmaceuticals,356 fluorides,357 crude oil,358 dyes,359,360 venomous pollutants,361 antibiotics,362,363etc., which have been removed efficiently as per several reports. As an example, chromium(VI) found in wastewater is an acute contaminant resulting from the wide application of chromium metal in chemical industries, textile dyeing, and metallurgical operations. Cr(VI) is more toxic than Cr(III) and is commonly found in polluted water and soil owing to its stability, as well as mobility. Unfortunately, it is a carcinogen, and the WHO has set a permissible limit of 50 μg L−1 for Cr in potable water. The Cr(VI) adsorption on activated carbon was analysed by Ramirez et al. using FTIR, computational chemistry, and diffusion studies models.364 The results demonstrated that activated carbon had a high adsorption capacity for Cr(VI). DFT studies for Cr adsorption, as well as FTIR analysis, showed that the main mechanisms that exist in the Cr(VI) adsorption process on activated carbon are as follows: (i) complexation, (ii) electrostatic interactions, and (iii) the reduction of Cr(VI) to Cr(III); functional groups, such as esters, semiquinone, –OH, and carboxylic acids, are mainly responsible for these processes.
The activation of carbon raw material is carried out by the following: (i) physical activation, and (ii) chemical activation, as presented in Fig. 24. In the physical activation method, carbonaceous substances can undergo (a) carbonization, i.e., heating at 600–900 °C in the presence of an inert gas, (b) oxidation/activation in the presence of O2 or steam in the temperature range of 600–1200 °C. In chemical activation, the carbonaceous raw material in the absence of air is heated and treated with chemicals such as phosphoric acid, KOH, CaCl2, ZnCl2, etc., which infuse into the structure. These chemicals can degrade the wood fibres and cause a porous structure to develop.365
 | ||
| Fig. 24 Flowchart depicting activation methods for carbonaceous raw material by physical and chemical methods. | ||
The limitations of AC could include a short lifespan since it can become saturated with contaminants over time, thus reducing its effectiveness. It requires regular replacement/regeneration, and could be selective toward certain organic compounds and less effective for inorganic substances or heavy metals. Moreover, it is not so cost-effective for frequent replacement as it also needs regular maintenance. It could often be susceptible to bacterial breeding, affecting water quality. The disposal of loaded AC adsorbed with toxic substances is another cause of concern.
To sum up this section, organic waste materials, which are pyrolysed and activated to form carbonaceous and microporous materials, are called activated carbon. These are industrially useful and cost-effective carbon materials whose properties have been presented. They show very good adsorption behaviour towards contaminants present in wastewater, such as oils, fluorides, heavy metals, and pharmaceuticals, etc.
2.8 Carbonaceous biopolymer-nanocellulose
Carbon-based agro-waste materials like crop straw, rice husk, corn cob, sugarcane bagasse, groundnut shells, etc., are a rich source of cellulose, hemicellulose, lignin, etc., and the valorisation of these materials is much needed at present. These materials can be functionalized and made into suitable biocomposites, which can then be used for water purification processes. Resource utilization of agricultural waste products is important for sustainable development because they are energy-rich and contain a plethora of chemical substituents. Moreover, applying bio-waste materials in water purification provides a new opportunity for their valorisation. In 2019, crop straw was produced in quantities greater than a billion metric tonnes globally. A major part of the plant biomass on earth comprises lignocellulosic mass, which serves as a carbon-rich feedstock that is produced as an outcome of extensive agriculture and is rich in natural fiber that could be substituted for polymers as sustainable materials (Fig. 25).368 Plant cell walls are comprised of cellulose (35–50%), hemicellulose (20–35%), and lignin (10–25%); thus, a significant amount of cellulose is available naturally in all plant cell walls. It is a linear homopolysaccharide of β-1,4-linked anhydro-D-glucose units with repeating units of cellobiose. The anhydroglucose unit, the monomer of cellobiose, consists of three hydroxyl groups that can form intramolecular and intermolecular H bonds with adjacent glucose molecules, leading to a strong network of H-bonded glucose units in cellulose. Thus, there are four different types of cellulose, based on morphology, namely, Cellulose I, Cellulose II, Cellulose III, and Cellulose IV. Celluloses I and II are crystalline, but they differ in inter-sheet H bonding, which is present in the II polymorph. Moreover, II is obtained from I by alkali treatment and is thermodynamically more stable than I. Cellulose III is amorphous and obtained from polymorphs I and II on amination. Cellulose IV is obtained by treating Cellulose III with glycerol.366 On a chemical basis, cellulose can be classified into cellulose ester and cellulose ether based on treating native cellulose with organic acids, acid chlorides, etc. Cellulose thus has high mechanical strength but is still flexible owing to the presence of hydroxyl groups, as well as the ubiquitous H-bonding present throughout the chains. Cellulose fibrils aggregate into both crystalline and amorphous regions. Hence, the orderly arrangement at the crystalline zones renders strength and stiffness, while the amorphous regions contribute to a flexible nature. The cellulose fibrils can range from a few micrometers up to 100 nm in diameter, and several mm in length, thus having a good aspect ratio for the cellulosic biomass. The nanocellulose fibrils are derived from the cellulose fibers by a top-down approach, and these have a diameter smaller than 100 nm, and the length could be several micrometers. Thus, the nanocellulose fibre has outstanding strength with a light weight and low density. It has an elastic modulus greater than Kevlar and a high tensile strength up to 10 GPa. Moreover, the high surface area and presence of –OH functional groups make it possible to functionalize them by surface modification methods. Membrane filtration is widely used for water treatment, gas separation, biomedical applications, and as air filtration materials; for these purposes, polymers, biopolymers, and organic–inorganic hybrid materials are generally used.367 Also, membranes are used in water purification methods such as ultrafiltration, RO, electrodialysis, and nanofiltration. Thus, for membrane fabrication, nanocellulose obtained from renewable resources is an obvious choice based on its various physical properties, availability and biodegradability. Nanocellulose can be classified into two main types, namely cellulose nanocrystals and cellulose nanofibers, which are used in membrane production. These membranes could be used for filtration and separation processes. Nanocellulose membranes are effective at selectively filtering out particulates based on their structure, charge, size, weight, etc. (Fig. 25).368 | ||
| Fig. 25 Cellulose biopolymer is the basic unit of all plant biomass and largely available on the earth,368 and the nanocellulose obtained from plant biomass can be used in different ways to obtain pure water. Reproduced from ref. 406 with permission from ACS, copyright 2022. | ||
Consequently, the nanocellulose (NC) market is expected to grow to more than 950 million dollars in 2026 at a growth rate of 22.7%.369 Heavy metals and inorganic pollutants, along with certain organic pollutants released from industries, negatively impact human health, the environment, and aquatic life, and ultimately, bioaccumulate in the food chain. Table 3 shows that metal ions and other anions adversely affect the health of all living systems and, therefore, their removal from wastewater needs to be a priority. The removal methods include filtration, electrolysis, precipitation, adsorption, magnetic separation, crystallization, etc. Adsorption is one of the best methods since it allows the removal of ppm levels of metal impurities and dyes, etc., from wastewater. The lignocellulosic masses can serve as effective adsorbent materials. These materials can be functionalized such that specific impurities can be removed from the wastewater. The lignocellulosic biomass has a large number of –OH groups and can form –H bonds or complexes with metal ions.
| Pollutant in water | Carbon-based biomass used | Modification done | Conc. of pollutant adsorbed | References |
|---|---|---|---|---|
| As(V) | Coconut pith coir | As an anion exchanger | 1 mg L | 377 |
| As(V) | Acacia nilotica | Powdered stem | 50.8 mg g−1 | 378 |
| ClO4− | Cotton stalk | Amine cross-linked stalk | 83.8 mg g−1 | 385 |
| Cd | Orange peel | No modification | 128.23 mg g−1 | 386 |
| CN− | Rice husk | Increase in pH and temp. | 97% efficiency at optimum operating conditions | 387 |
| Cr | Coconut fiber | Composite with polyaniline | 0.25 mg mL−1 | 388 |
| Cu | Okra waste | Methyl methacrylate and acrylamide | 33.05 mg g−1 | 389 |
| F− | D. sissoo saw dust | Thermally activated | 0.7 mg g−1 | 390 |
| Hg | Walnut shell, hazel nut, pistachio shell | Mechanically ground to sizes 1–2 mm | >90% efficiency | 391 |
| Ni | Biomass gel | Carboxy methyl-doped boron nitride composites | 344 mg g−1 | 392 |
| NO3− | Coconut copra | Amine grafted | 49.9 mg g−1 | 393 |
| Pb | Cellulose nanofibers | Sodium alginate | 318.47 mg g−1 | 394 |
| PO43− | Sugarcane bagasse cellulose | Doping La into Mg–Al LDH and coating it on cellulose | 106.21 mg g−1 | 395 |
| SO42− | Pomelo peel biochar | ZrO2-modified | 35.21 mg g−1 | 396 |
| Zn | Hibiscus esculentus | Acrylamide + acrylic acid | 55.04 mg g−1 | 397 |
Attempts have been made to remove anionic pollutants present in wastewater, such as nitrates (NO3−), fluorides (F−), phosphates (PO43−), perchlorates (ClO4−), and cyanides (CN−), by functionalizing biomass with quaternary ammonium, amine groups, etc., as seen in Table 3. Impregnated bioadsorbents have been used for the adsorption of these anions; also, lignocellulosic masses can be grafted to improve the biosorption of these anions present in aqueous effluents. Acid-treated sunflower seed husks were used to adsorb nitrates from effluent water by Kristek Janković et al.398 The modified sunflower seeds showed an uptake of ∼13 mg g−1 for the NO3− ions. Quaternary amine functionalization was carried out on several cellulosic waste materials in a certain study that showed improved nitrate adsorption with a capacity of ∼240 mg g−1.399 Fluoride contamination of water is a serious problem, and Kong et al. have reviewed several publications related to carbon-based materials that suitably adsorb fluorides.400 Another review on the biosorption of fluorides from contaminated waters has been contributed by Halder et al.401
The presence of excess phosphates in water bodies can cause eutrophication and algal bloom and can adversely affect the ecology by depleting the oxygen levels. The removal of phosphates by biosorbent material like sugarcane bagasse was done by crosslinking chitosan with cellulose, such that a low-cost anion exchange adsorbent was obtained and a good level of phosphate was adsorbed on the surface.402 Using a quaternization reaction and ion-imprinting technology, a novel wheat straw anion-imprinted biosorbent was made for the adsorption of phosphates by modifying wheat straw. According to the report, the maximum phosphate adsorption capacity, calculated by the Langmuir model, was 23.5 mg g−1 at 45 °C.403 Shah et al.404 confirmed the uptake of phosphate and made a low-cost adsorbent by using magnetic Fe3O4 nanoparticle-impregnated tea waste that works well with an adsorption capacity of 227 mg g−1. Perchlorates are hazardous substances that are entering waterways due to explosives, propellants, or fireworks. They are difficult to remove, affect metabolic activities, and disrupt thyroid function and the CNS, especially in infants. Gao et al.385 functionalized a cotton stalk with epichlorohydrin, trimethylamine, and pyridine to capture perchlorate ions, which are rather difficult to remove from wastewater. The amine impregnated cotton stalk showed the adsorption capacity for perchlorate ions at 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) °C to be 83.3 mg g−1 which reduced to 80 mg g−1 at 40
°C to be 83.3 mg g−1 which reduced to 80 mg g−1 at 40![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) °C, the adsorption process being exothermic in nature. Suspended matter can be removed from water using nanocellulosic membranes effectively based on size exclusion principle.405 Filtration of microsized particles, colloids, and even nanosized matter can be carried out depending on the size of the nanopores in the membrane. The smaller the pore size, the greater the extent of removal of particles. Certain metal nanoparticles, such as Ag, Cu, and Au, and CNTs and CNFs can act as antibacterials because their small pore size, smooth surface, and thin films can deter bacteria, viruses and other microbes.406 The nanocellulosic membrane could be treated with reagents as shown in Fig. 26(a and b) to functionalize the surface and induce charge, thereby preventing microbial growth. Depending on the charge on the surface of the membrane, oppositely charged particles can be attracted and removed from the wastewater. By tailoring the pore size, surface charge, and properties of the membrane, it can be used for water purification407 as shown in Fig. 26(c). The image shows the deposition of a layer of nanocellulose on the surface of a filter paper by vacuum filtration. The concentration of impurities in water, the size of particles, pH of wastewater, flow rate, etc., affect the removal efficiency of suspended matter during purification. Since nanocellulose has good mechanical properties and can be functionalized, it can be a prime material for wastewater treatment.408 Improving the hydrophilicity and mechanical strength of nanocellulose membranes by controlling the surface charges and reactive sites can also reduce the cost of purification.
°C, the adsorption process being exothermic in nature. Suspended matter can be removed from water using nanocellulosic membranes effectively based on size exclusion principle.405 Filtration of microsized particles, colloids, and even nanosized matter can be carried out depending on the size of the nanopores in the membrane. The smaller the pore size, the greater the extent of removal of particles. Certain metal nanoparticles, such as Ag, Cu, and Au, and CNTs and CNFs can act as antibacterials because their small pore size, smooth surface, and thin films can deter bacteria, viruses and other microbes.406 The nanocellulosic membrane could be treated with reagents as shown in Fig. 26(a and b) to functionalize the surface and induce charge, thereby preventing microbial growth. Depending on the charge on the surface of the membrane, oppositely charged particles can be attracted and removed from the wastewater. By tailoring the pore size, surface charge, and properties of the membrane, it can be used for water purification407 as shown in Fig. 26(c). The image shows the deposition of a layer of nanocellulose on the surface of a filter paper by vacuum filtration. The concentration of impurities in water, the size of particles, pH of wastewater, flow rate, etc., affect the removal efficiency of suspended matter during purification. Since nanocellulose has good mechanical properties and can be functionalized, it can be a prime material for wastewater treatment.408 Improving the hydrophilicity and mechanical strength of nanocellulose membranes by controlling the surface charges and reactive sites can also reduce the cost of purification.
 | ||
| Fig. 26 (a) Functionalization on the cellulose surface and a variety of nanocellulosic forms. Reproduced from ref. 405 with permission from ACS copyright [2022]. (b) Agents used in the modification of cellulose and (c) nanocellulose membrane formation on a filter paper substrate by vacuum filtration. Reproduced from ref. 407 with permission from [Springer Nature] copyright [2018]. | ||
| Membrane | Dye adsorption capacity (mg g−1) | |||
|---|---|---|---|---|
| VBB | MV2 | RGG | Total | |
| a Conc. of dye: 2 mg L−1; vol: 30 mL; pH = 7.1; filtration pressure: 0.9 bar. | ||||
| NC fibers | 2.4 | 1.8 | 1.6 | 5.8 |
| GO/NC fibers | 3.0 (60) | 2.8 (100) | 2.3 (70) | 8.1 (230) |
The dye molecules were well separated irrespective of charge, and this was due to adsorption and size exclusion. It was further concluded that the GO/CNF membrane worked better than the nylon 66 membrane, having a similar pore structure. The high flux, stability, and good performance with easy fabrication make it a suitable material for the removal of organic contaminants, dyes, pesticides, heavy metal ions and microbes. Liu et al. prepared a double-layered GO/CNF composite membrane that exhibited extremely high-water permeation (18![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 123 ± 574 Lm−2 h−1 bar−1) and dye permeation 90 Lm−2 h−1 bar−1.433 Graphene nanomaterials are produced from aqueous suspensions and have the tendency to aggregate, which results in decreased surface area, which also affects the optical, mechanical, and electrical properties. On making composites with CNCs, better dispersive features were observed, along with higher flexibility, elongation, stability, conductivity, and enhanced adsorption capacity.434 For the adsorption and photocatalytic degradation of ciprofloxacin hydrochloride from aqueous solution, Anirudhan et al.435 prepared a novel photocatalyst by incorporating nano ZnO into GO/NC. This composite was used for the adsorption and further photodegradation of the antibiotic ciprofloxacin, which is used extensively for poultry. ZnO in GO enhanced the band gap to 2.8 eV. A composite was made with NC, and the degradation of ciprofloxacin was accomplished, using visible light, via electron–hole interactions. Water bodies are commonly used to get rid of antibiotics used in veterinary applications, and wastes from pharmaceutical plants, nursing homes, municipal water treatment plants, and poultry that contains antibiotics, eventually reaches waterbodies. Accumulations of antibiotics like ciprofloxacin could be present in water or soil surroundings, and could become sources for producing antibiotic-resistant bacteria that are harmful to human and animal health. A similar study was done for the removal of levofloxacin by Tao et al.436 The decoration of oxides like Cu2O/TiO2 on a cellulose/rG hydrogel framework for the degradation of MB for wastewater remediation was done by Zheng et al. Due to the oxides, higher catalytic activity was observed under UV light due to an efficient electron transfer process. Carboxymethyl cellulose (CMC) was added to GO flakes to make an aerogel composite by the hydrothermal method. The CMC added improved hydrophilicity and mechanical strength, and it could be used for wastewater treatment. The absorption efficiency for different organic liquids such as engine oil, DMSO, acetone, ethanol, etc., was studied for 5 regenerating cycles. The composite also showed good adsorption capacity for Rhodamine B with 186.33 mg g−1 as the adsorption activity.437 Yang et al. reported the preparation of a cellulose/GO composite that exhibited substantial adsorption of U(VI). The solution pH, adsorbent dosage, and contact time were varied, showing 97.5% U(VI) removal at an adsorbent dosage of 1.75g L−1 under slightly acidic conditions in about 1 hour.438 In yet another study, a NC/GO hybrid aerogel was ultrasonically prepared and tested for the removal of 21 types of antibiotics present in water. More than 69% of antibiotics were adsorbed with a max adsorption capacity of 418.7 mg g−1 for chloramphenicol and 454.6 for tetracyclines, as calculated by the Langmuir isotherm model, demonstrating the potential of NC/GO as an effective and economical adsorbent for antibiotic elimination from wastewater. The adsorption properties of GO were studied for heavy metal ions present in water. The graphene 2-D surface was functionalized by adding groups such as –COOH, –C
123 ± 574 Lm−2 h−1 bar−1) and dye permeation 90 Lm−2 h−1 bar−1.433 Graphene nanomaterials are produced from aqueous suspensions and have the tendency to aggregate, which results in decreased surface area, which also affects the optical, mechanical, and electrical properties. On making composites with CNCs, better dispersive features were observed, along with higher flexibility, elongation, stability, conductivity, and enhanced adsorption capacity.434 For the adsorption and photocatalytic degradation of ciprofloxacin hydrochloride from aqueous solution, Anirudhan et al.435 prepared a novel photocatalyst by incorporating nano ZnO into GO/NC. This composite was used for the adsorption and further photodegradation of the antibiotic ciprofloxacin, which is used extensively for poultry. ZnO in GO enhanced the band gap to 2.8 eV. A composite was made with NC, and the degradation of ciprofloxacin was accomplished, using visible light, via electron–hole interactions. Water bodies are commonly used to get rid of antibiotics used in veterinary applications, and wastes from pharmaceutical plants, nursing homes, municipal water treatment plants, and poultry that contains antibiotics, eventually reaches waterbodies. Accumulations of antibiotics like ciprofloxacin could be present in water or soil surroundings, and could become sources for producing antibiotic-resistant bacteria that are harmful to human and animal health. A similar study was done for the removal of levofloxacin by Tao et al.436 The decoration of oxides like Cu2O/TiO2 on a cellulose/rG hydrogel framework for the degradation of MB for wastewater remediation was done by Zheng et al. Due to the oxides, higher catalytic activity was observed under UV light due to an efficient electron transfer process. Carboxymethyl cellulose (CMC) was added to GO flakes to make an aerogel composite by the hydrothermal method. The CMC added improved hydrophilicity and mechanical strength, and it could be used for wastewater treatment. The absorption efficiency for different organic liquids such as engine oil, DMSO, acetone, ethanol, etc., was studied for 5 regenerating cycles. The composite also showed good adsorption capacity for Rhodamine B with 186.33 mg g−1 as the adsorption activity.437 Yang et al. reported the preparation of a cellulose/GO composite that exhibited substantial adsorption of U(VI). The solution pH, adsorbent dosage, and contact time were varied, showing 97.5% U(VI) removal at an adsorbent dosage of 1.75g L−1 under slightly acidic conditions in about 1 hour.438 In yet another study, a NC/GO hybrid aerogel was ultrasonically prepared and tested for the removal of 21 types of antibiotics present in water. More than 69% of antibiotics were adsorbed with a max adsorption capacity of 418.7 mg g−1 for chloramphenicol and 454.6 for tetracyclines, as calculated by the Langmuir isotherm model, demonstrating the potential of NC/GO as an effective and economical adsorbent for antibiotic elimination from wastewater. The adsorption properties of GO were studied for heavy metal ions present in water. The graphene 2-D surface was functionalized by adding groups such as –COOH, –C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O, –OH, which complex with metal ions. Due to the hydrophilic nature of GO, it was cross-linked on dialdehyde cellulose with triethylenetetramine by Wang et al.439 These composite adsorbents were studied for the removal of Cu(II) and Pb(II) ions. The adsorption capacity was approximately 65 mg g−1 and 80.9 mg g−1, respectively. The adsorbent materials were successfully tested over 4 cycles for the removal of ions. Several studies have been carried out for the removal of Cu2+, Ni2+, Hg2+, Cr6+, Pt2+, and As3+ by suitably modifying the cellulose surface.440 The adsorption of Ni2+ was greatly enhanced to 96% when NC was modified by GO. This is attributed to the involvement of the –COOH, –C
O, –OH, which complex with metal ions. Due to the hydrophilic nature of GO, it was cross-linked on dialdehyde cellulose with triethylenetetramine by Wang et al.439 These composite adsorbents were studied for the removal of Cu(II) and Pb(II) ions. The adsorption capacity was approximately 65 mg g−1 and 80.9 mg g−1, respectively. The adsorbent materials were successfully tested over 4 cycles for the removal of ions. Several studies have been carried out for the removal of Cu2+, Ni2+, Hg2+, Cr6+, Pt2+, and As3+ by suitably modifying the cellulose surface.440 The adsorption of Ni2+ was greatly enhanced to 96% when NC was modified by GO. This is attributed to the involvement of the –COOH, –C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O, and –OH groups present on the GO surface.441 In a study by Chattopadhyay et al., a facile one-pot synthesis of GO/NC fibrous composite was achieved. This composite was tested for the removal of methylene blue, an azo dye, from aqueous solution. The removal of 95% of MB in 2 h was observed, and the maximum absorption capacity of the nanocomposite was determined to be 751.88 mg g−1. It was predicted by the authors that the composite can be used for adsorption of toxic azo dyes.442 Six triazine pesticides were adsorbed by the CNF/GO composite. The strong electron-donor nature of the N, S and O atoms present in the different triazine pesticides have resulted in van der Waals interactions with the adsorbent. It was determined that 30 mL of the composite was required for adsorbing a 10 mL solution of the triazine pesticides. Adsorption was observed to be a spontaneous and endothermic process with an efficiency of 85% for 6 cycles of recycling the composite using organic solvent. Chantaso et al. derived cellulose from sugarcane leaves and fabricated filter membranes by adding varying quantities of GO using the vacuum filtration technique. As the concentration % of GO increased, the antimicrobial activity enhanced for E. coli bacteria and also towards the removal of particulate matter.443 Mathew et al. used TEMPO-oxidized cellulose fibers and nano GO sheets, which exhibited promising adsorption for Cu(II) removal and these experiments proved that GO/NC hybrid membranes have good robustness for water purification.444 Thus, carbon nanomaterials have been widely investigated by several researchers, and they have been proven to be very suitable for water treatment strategies. Yet, there are areas in which work is still required to be done in order to achieve cost effectiveness, selectivity and sustainability.
O, and –OH groups present on the GO surface.441 In a study by Chattopadhyay et al., a facile one-pot synthesis of GO/NC fibrous composite was achieved. This composite was tested for the removal of methylene blue, an azo dye, from aqueous solution. The removal of 95% of MB in 2 h was observed, and the maximum absorption capacity of the nanocomposite was determined to be 751.88 mg g−1. It was predicted by the authors that the composite can be used for adsorption of toxic azo dyes.442 Six triazine pesticides were adsorbed by the CNF/GO composite. The strong electron-donor nature of the N, S and O atoms present in the different triazine pesticides have resulted in van der Waals interactions with the adsorbent. It was determined that 30 mL of the composite was required for adsorbing a 10 mL solution of the triazine pesticides. Adsorption was observed to be a spontaneous and endothermic process with an efficiency of 85% for 6 cycles of recycling the composite using organic solvent. Chantaso et al. derived cellulose from sugarcane leaves and fabricated filter membranes by adding varying quantities of GO using the vacuum filtration technique. As the concentration % of GO increased, the antimicrobial activity enhanced for E. coli bacteria and also towards the removal of particulate matter.443 Mathew et al. used TEMPO-oxidized cellulose fibers and nano GO sheets, which exhibited promising adsorption for Cu(II) removal and these experiments proved that GO/NC hybrid membranes have good robustness for water purification.444 Thus, carbon nanomaterials have been widely investigated by several researchers, and they have been proven to be very suitable for water treatment strategies. Yet, there are areas in which work is still required to be done in order to achieve cost effectiveness, selectivity and sustainability.
Thus, to summarize, nanocellulose is a biopolymer extracted from agricultural waste and other cellulosic materials. Its structure, properties and modification by functionalizations have been explored in this sub-section. The efficacy of NC for the removal of all types of ions and heavy metals, and its capacity to remove organic pollutants, pharmaceuticals, dyes, and heavy metals as a composite material with graphene/GO have been discussed, with examples taken from literature.
An overview of the characteristics/strengths and challenges/weaknesses of the C-based nanomaterials discussed in this review for water purification is presented in Table 5.
| Form of carbon | Characteristics/supporting features | Challenges/limitations | References | |
|---|---|---|---|---|
| 1 | CNT | (1) 1D nanomaterial with high adsorption selectivity and capacity | (1) High cost of manufacturing and obtaining large quantities | 64–68 |
| (2) Methods of preparation are well established | (2) Difficult to synthesize SWNTS and scale up | |||
| (3) Easier regeneration, high conductivity | (3) Hard to disperse and functionalize CNTs | |||
| (4) Good adsorbent materials due to high surface area. Removes microbes and viruses, heavy metals effectively | (4) Exhibits a certain level of cytotoxicity. Difficult to develop biocompatible materials using CNTs | |||
| (5) Can be functionalized with hydroxyl, acid, and thiol groups on its surface | (5) No effective regulatory systems in place yet. | |||
| (6) Durable, high stability, short response time, especially in the form of nanocomposites | ||||
| (7) Ease of nanocomposite application | ||||
| 2 | Graphene | (1) A 2D nanomaterial made up of monoatomic graphite film with high strength | (1) Making graphene, GO, rGO on a large scale is a challenge | 120–124 |
| (2) High surface area with high absorbance capacity | (2) Not cost-effective | |||
| (3) And the ability to interact via π–π stacking, H bonding, and electrostatic interactions | (3) Toxic precursors | |||
| (4) Tunable surface chemistry | (4) Hydrophobic | |||
| (5) Can be made into GO and rGO forms, which are cost-effective | ||||
| (6) Selective for adsorbing organic dyes | ||||
| (7) Nanocomposites of GO with other metal nanoparticles can be used to adsorb heavy metals | ||||
| (8) Exhibits antibacterial activity in nanocomposite form | ||||
| 3 | Graphynes | (1) It is a 2D material | (1) Costly synthesis processes | 183–188 |
| (2) It can be synthesized in the lab | (2) The synthesis process is not easy | |||
| (3) It is a much more efficient material than most C nanoforms for wastewater treatment | ||||
| 4 | Fullerene | (1) 0D, ball-shaped nanomaterials | (1) Thermodynamically less stable than graphene or nanodiamond | 217–222 |
| (2) Hard materials that withstand high pressure and temperature | (2) Low thermal & electrical conductivity | |||
| (3) Soluble in common organic solvents | (3) Can also upregulate certain species of bacteria | |||
| (4) Can be antibacterial | (4) High cost | |||
| (5) Good adsorbents for heavy metals, dyes, and organic pollutants, especially for making composites with semiconductors/metal oxides | (5) Hydrophobic nature | |||
| (6) They have a lower density (1.65 g m−3) than diamonds | (6) Fullerenes may undergo degradation in the presence of light and oxygen | |||
| 5 | Carbon quantum dots | (1) 0D nanomaterial | (1) Lack of standard synthesis methods for scaling up | 265–271 |
| (2) Cost effective preparation methods | (2) Lack of long-term toxicity studies | |||
| (3) Biocompatibility | (3) Difficult to purify | |||
| (4) Versatile surface chemistry with possible functionalization | ||||
| (5) Water soluble and pollutant sensing capability | ||||
| (6) Unique optical properties | ||||
| 6 | Nanodiamonds | (1) 0D robust nanoform with both sp2 and sp3 C atoms | (1) Difficult to biodegrade in organisms | 295–300 |
| (2) High chemical stability | (2) Difficult to prepare monodisperse NDs | |||
| (3) Electrically conducting | (3) May have several defects in the structure | |||
| (4) Hard materials | (4) May persist in surroundings | |||
| (5) Large surface area with tunable characteristics | ||||
| (6) Is biocompatible and can undergo bioconjugation | ||||
| (7) Strong mechanical properties | ||||
| (8) Chemically inert | ||||
| 7 | Activated carbon | (1) Cost-effective preparation method | (1) Expensive chemical and thermal regeneration method | 445 and 446 |
| (2) High surface area and high degree of porosity | (2) Single-use ACs have a significant environmental impact | |||
| (3) High adsorption capacity | (3) Production from different carbon sources may have differing adsorption capacities and variable lifespans | |||
| (4) Effective for adsorbing organic pollutants, dyes, and heavy metals from industrial wastewater | (4) GAC production and reactivation consume substantial energy and have respiratory effects | |||
| (5) Reactivated GACs are environmentally sustainable and more cost-effective | (5) Disposal can lead to harmful byproducts due to incineration | |||
| (6) Removes undesirable odour and colour from water | ||||
| (7) Easier technology to use | ||||
| 8 | Cellulose | (1) Can be 2D, or 1D if fibres | (1) Production of nanocellulose on a large scale | 326, 329, 393 and 447 |
| (2) Most abundant natural biopolymer on earth | (2) Regeneration of cellulose-based materials requires harsh conditions | |||
| (3) Biocompatible & versatile | (3) Limitations in specificity | |||
| (4) Can be functionalized with a variety of functional groups | (4) Biofouling possible | |||
| (5) Can remove organic dyes, heavy metals, anions, pharmaceuticals and microbes | (5) Processing cost | |||
| (6) Can be made into membranes, sheets, aerogels, filters, and films | ||||
| (7) Possibilities of making NC hybrid membranes by 3D printing |
3 Technoeconomic analysis and challenges
The remarkable physicochemical properties of carbon-based nanomaterials facilitate their high visibility in applications related to water quality amelioration and include the following.(i) Adsorption-their high surface area, functionalized surfaces and ability to bond via complexation, H-bonding or electrostatic forces such as π–π EDA make them resourceful for the removal of various kinds of pollutants, such as heavy metals, ECs or PFAS.
(ii) Ability to form composites-carbon-based materials are very versatile when they are integrated with polymers to form composite materials with high-end capabilities. For example, membranes decorated with CNTs or activated carbon show better permeability, fouling resistance and longer life, in addition to polymer flexibility, tunability, hydrophobicity, and antimicrobial behaviour with stability for overall improvement in filtration.
(iii) Catalysis–materials such as CNTs, graphene, nanocellulose or their composites with TiO2, ZnO, etc., can bring about the catalytic or photocatalytic degradation of microbes and organic pollutants present in water.
As seen in the previous sections, carbon-based materials like CNT, nanocellulose or activated carbon produced from waste materials are capable of filtration, disinfection, pollutant removal, desalination, etc., in pristine or in composite form. These materials remain ubiquitous in water purification and are used in one or many ways. With rising pollution and the demand for pure water, effective water purification systems are required. With advancements in material synthesis, the cost of production of nanomaterials would decline with enhanced scalability. Moreover, the production of such materials from waste carbon sources or biomass could offer a sustainable solution. Thus, technological advancements in materials chemistry and engineering for the production of carbon-based nanomaterials using waste could be a technoeconomic advancement in the field of water amelioration. The fabrication of these materials is currently carried out via energy intensive processes such as CVD, laser ablation, arc discharge, acid hydrolysis, ball milling, hydrothermal, sol gel, or combustion/microwave/ultrasonication, which have potential scalability issues. Engineering water filtration systems using these materials would lead to higher costs that need to be offset by lower maintenance and higher efficiency and durability. Small-sized filters or portable ones are required because supplying lower-cost purification systems especially to remote places and villages is a potential challenge. The management of the spent carbon materials and disposal needs to be investigated since concerns of toxicity and the impact on the environment prevail. Regulations and frameworks/guidelines could be enforced for safe use and disposal. There is a significant gap that exists for carbon nanomaterial behaviour under varied environmental conditions, along with agglomeration issues, stability, transformations, and toxicity, and such studies need attention. Nanomaterial synthesis using biogenic processes or encapsulating/immobilizing nanomaterials could mitigate contamination issues. Further, life cycle assessments could be carried out to evaluate environmental trade-offs. Despite such challenges, carbon materials hold great promise for the improvement of water quality. Therefore, ongoing R&D for cost reduction, scalability, and improved efficiency with lower environmental impact could be the answer to this concern. Subsequently, smart water purification systems with optimized performance running on real-time data could be envisioned, as seen in Fig. 27.
4 Conclusion and future perspectives
Wastewater treatment, water quality remediation, and urban water management are primary sustainability development challenges, and marginalised groups face huge disparities in getting access to clean water and sanitation. Carbon-based nanomaterials such as graphene, GO, CNT, CDs, ACs, NDs, fullerenes, biopolymers, etc., offer promising wastewater treatment solutions with remarkable properties, potentially revolutionising treatment methods. These nanomaterials possess exceptional physicochemical properties, namely, a high surface area-to-volume ratio, tunable surface chemistry, handling flexibility, as well as unique electromechanical characteristics that enable interactions with diverse contaminants present in wastewater. Carbon-based nanomaterials are robust, have enhanced adsorption capacity, providing abundant active sites for capturing pollutants such as heavy metals, organic dyes, and pharmaceutical residues. This effective removal method offers a ground-breaking approach to contaminant removal from wastewater. Additionally, they are essential to sophisticated oxidation processes, which allow complicated contaminants to be broken down into byproducts that are safe for the environment. Incorporating carbon nanoparticles into nanocomposites enhances their wastewater treatment capabilities, enabling customised remedies to address specific contaminants. When carbon nanomaterials are combined with semiconductors/photocatalytic materials like titanium dioxide, a synergistic effect occurs, allowing pollutant destruction under UV and visible light. These substances have remarkable adsorption capacity, oxidative potential, selectivity, and synergy with other materials, making them suitable catalysts or substrates for transforming wastewater treatment methods. Challenges like nanoparticle aggregation, stability, cost, and environmental impacts require comprehensive research and mitigation strategies. Careful consideration of scalability and integration into the treatment infrastructure is essential. Carbon-based nanomaterials are promising for wastewater treatment, with research focusing on optimising properties, understanding interaction mechanisms, and developing efficient synthesis techniques. Common drawbacks that need to be overcome are cost-effectiveness and scaling up the production of the nanomaterials. Certain aspects of wastewater such as cytotoxicity studies, regulatory systems, standard scaling up procedures and environmental ramifications are still lacking. Collaborative efforts between researchers, engineers, and industries are essential to harness their full potential and transform them into sustainable wastewater treatment materials. Moreover, industrial effluent treatment needs to be strictly complied with under legal frameworks to maintain water quality standards.Future demands related to clean drinking water using sustainable technology entitles us to look at the use of AI in water purification. AI tools such as ML and neural network algorithms allow systems to learn from various types of datasets, adjust accordingly and make decisions automatically.
AI-driven algorithms can boost water efficiency, decrease fouling, manage pressure, temperature, and flow rate, and decrease operational time. Nanomembranes are advanced materials that are ultra-thin, with high surface area and good resilience and have the synergistic properties of polymers and carbon nanomaterials. AI can suggest composite nanomembrane compositions of polymers, CNTs, GO, graphynes, nanocellulose, etc., optimized for best purification with respect to ion selectivity, fouling mitigation, scaling up, energy usage and sustainability for area specific water samples. Thus, these are advanced materials that are apt at providing the necessary performance, versatility, and efficiency. With AI-driven algorithms, the optimization of membrane structure and design is possible.448 AI-designed models can suggest/identify suitable materials that display enhanced selectivity, permeability and fouling resistance. ML-driven computational models could simulate transport phenomenon, molecular interactions and water properties in order to identify other 3D materials capable of outperforming the existing technology. AI-based control systems can adjust the rate of water flow, pressure, cleaning cycle, energy use, and membrane fouling rate depending on the type and amounts of impurities present in the water sample. Thus, the integration of AI with the carbon-based nanomaterials is a game changing transformation in water quality amelioration. In conclusion, carbon materials have emerged as key materials in water and wastewater treatment. They are at the forefront of the future of wastewater treatment whether used in a pristine/composite manner. AI investment in water industries is expected to reach $6.3 billion by 2030. Also, AI is expected to save 20–30% of operational expenditure by optimizing the use of energy and chemical consumption in water treatment.449 AI tools can assess the performance of the adsorption process, which forms the basis for the removal of metal ions, dyes, organic pollutants, drugs, etc. However, more research work is needed to use AI techniques to upgrade the performance of water treatment. The selection of proper data, the application of AI tools and enhanced research can help overcome the challenges currently faced in water quality remediation and management. Fig. 28 shows a conceptual framework for attaining sustainable treatment towards water remediation.
The automation of water quality estimation through AI or XAI can contribute to potability and the evaluation of the impurities present in it.
(1) Through AI-enabled control for water purification systems using carbon-based nanomaterials such as graphene, nanocellulose, or CNTs, smarter filtration systems are practically possible. Decision-making based on real-time water quality data, adjustments in flow rates, pressure, membrane configurations, etc., could be optimized.
(2) Nanomaterials such as CQDs, CNTs, etc., paired with AI could improve the sensitivity and selectivity of the contaminants present in the water, and the purification system could be adjusted accordingly.
(3) The AI models with adaptive controls and sensors could make the purification of water efficient by optimizing the separation processes for specific pollutants. Further, the ML models could analyse the sensor data and identify contaminants present for an automated response to take action.450 They could also give warnings related to water safety and avert health problems related to water borne diseases.
(4) AI-based smart units could predict when maintenance is due, such as for exhausted filters or membranes, etc., and calculate the operational costs.
Fig. 29(a–d) shows the use of ML in mapping the quality of water using explainable AI, wherein the dataset shows that 70% is used for training and 30% is used for testing.
 | ||
| Fig. 29 Water quality prediction using AI-based models: (a) interfacing ML algorithms using XAI, (b) supervised learning models, (c) unsupervised learning models, and (d) hybrid models for water quality evaluation. Reproduced from ref. 450 with permission from Springer Nature copyright [2024]. | ||
Therefore, scalable and sustainable purification systems for both rural and urban areas are very much possible in the near future. Optimising characteristics, understanding interaction mechanisms, and developing efficient synthesis procedures, as well as reducing overall costs, should be the primary objectives of water quality amelioration. Sustainable water purification techniques and water management in rural and urban areas would require concerted efforts in materials and instrument development, innovative technologies, real-time monitoring, modelling aids, exploratory analysis using ML models, and planning strategies for the supply, treatment, reuse, and drainage. One also needs to determine whether air/water/soil quality and climate change are more pressing issues, considering environmental, social, or health issues, or ignorance in the name of progress. This review presents various water treatment methods using carbon nanomaterials and the trade-offs between them. Although carbon materials play a profound role in the treatment of contaminated water, stakeholders need to make informed decisions on the multifaceted issues involved while transitioning to advanced technologies. Clean water and water quality management, being a sustainable development goal, is a critical challenge and requires a concerted and integrative approach where AI adoption could offer innovative solutions for the future.
Author contributions
Malini S. Bapat and Hema Singh were involved in the idea creation, data curation, writing—original draft preparation. Shankar B. Chaudhari helped in methodology and data curation. Suresh Shendage contributed to methodology and data curation. Raj Kumar and Arun Karnwal were involved in writing, reviewing, and editing. Deepak Kumar helped in writing, reviewing, and editing.Conflicts of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.Abbreviations
| SDG | Sustainable Development Goals |
| PVA | Polyaniline |
| WHO | World Health Organization |
| DCB | 1,2-Dichlorobenzene |
| UNESCO | The United Nations Educational, Scientific and Cultural Organization |
| rGO | Reduced graphene oxide |
| GAC | Granular activated carbon |
| CVD | Chemical vapour deposition |
| PAC | Powdered activated carbon |
| dGO | Defective graphene oxide |
| EDI | Electrodeionization |
| OFLOX | Ofloxacin |
| BOD | Biochemical oxygen demand |
| BP3 | Benzophenone-3 |
| COD | Chemical oxygen |
| HGO | Hummers derived GO |
| TDS | Total dissolved solids |
| RhB | Rhodamine blue |
| VOC | Volatile organic compounds |
| GHG | Graphene hydrogel |
| EC | Emerging contaminants |
| PES | Polyethersulfone |
| PPCP | Pharmaceutical and personal care products |
| PSU | Polysulfone |
| EDC | Endocrine-disrupting compounds |
| CA | Cellulose acetate |
| DDT | Dichlorodiphenyltrichloroethane |
| G-PANCMI | Graphene-attached polyacrylonitrile-co-maleimide |
| E2 | Estradiol |
| EDTA | Ethylene diamine tetraacetic acid |
| EE2-17 | Ethinylestradiol |
| CS | Chitosan |
| BPA | Bisphenol A |
| HQ | Hydroquinone |
| NP | Nonylphenol |
| BAS | Basic aluminium sulfate |
| PPM | Parts per million |
| SSA | Specific surface area |
| PFAS | Perfluoroalkyl substances |
| AS-GO-3 | Aminopropyltriethoxysilane-functionalized graphene oxide |
| EPA | Environmental Protection Agency |
| RG | Reactive green |
| PFOA | Perfluoro octanoic acid |
| NM | Nanomaterial |
| PFOS | Perfluoro octane sulphonic acid |
| ODA | Octadecylamine |
| RBC | Rotating biological contactors |
| HOMO | Highest occupied molecular orbital |
| IP | Interfacial polymerisation |
| LUMO | Lowest occupied molecular orbital |
| TFC/TFN | Thin film composites/nanocomposites |
| ROS | Reactive oxygen species |
| CNT | Carbon nanotubes |
| MB | Methylene blue |
| DCC | N,N′-Dicyclohexylcarbodiimide |
| MCS | Magnetite & chitosan |
| AC | Activated carbon |
| Ppy | Polypyrrole |
| ODS | Ozone-depleting substance |
| MOF | Metal–organic frameworks |
| CQD | Carbon quantum dot |
| ND-DA | Nanodiamond with double arm ligands |
| CPD | Carbonized polymer dots |
| ND | Nanodiamond |
| FWHM | Full width at half maximum |
| HPHT | High-pressure high-temperature |
| PVDF | Polyvinylidene fluoride |
| FTIR | Fourier transform infrared |
| DND | Detonated nanodiamonds |
| CNS | Central nervous system |
| BSA | Bovine serum albumin |
| GN | Graphene |
| PANI | Polyaniline |
| CNC | Cellulose nanocrystals |
| MR | Methyl red |
| CNF | Cellulose nanofibers |
| MO | Methyl orange |
| OPP | Organophosphorus pesticides |
| MG | Malachite green |
| NC | Nanocellulose |
| TC | Tetracycline |
| CMC | Carboxymethylcellulose |
| TEMPO | 2,2,6,6-Tetramethylpiperidine-1-oxyl |
| CB | Conduction band |
| PL | Photoluminescence |
| XAI | Explainable AI |
| NCQD | Nitrogen-doped carbon quantum dots |
| ND-SA | Nanodiamond with single arm ligands |
| VB | Valence band |
| DDC | Dimethyldithiocarbamate |
Data availability
The data analysis for this work is included in the manuscript file. Furthermore, the software files can be requested from the corresponding author. No primary research results, software or code have been included and no new data were generated or analysed as part of this review.Acknowledgements
Author MB acknowledges the Research grant by MKSSS’s Cummins College of Engineering for Women, Pune. Author DK acknowledges the Manipal University, Jaipur, for providing the research environment and facilities.References
- K. Anderson, What Was the Industrial Revolution's Environmental Impact?, 2024.
- W. Musie and G. Gonfa, Fresh water resource, scarcity, water salinity challenges and possible remedies: A review, Heliyon, 2023, 9(8), e18685 CrossRef.
- J. Kastlunger, UNGA Resolution 76/300—the human right to a clean, healthy and sustainable environment, the possible impact on the establishment of a human right to a healthy environment, Doctoral dissertation, 2024 Search PubMed.
- Water UN, Water scarcity, United Nations, 2021.
- Y. L. Luo, Y. R. Pan, X. Wang, Z. Y. Wang, G. Daigger, J. X. Ma, L. H. Tang, J. Liu, N. Q. Ren and D. Butler, Leveraging the water-environment-health nexus to characterize sustainable water purification solutions, Nat. Commun., 2025, 16(1), 1269 CrossRef CAS PubMed.
- UN-Water, Summary progress update, SDG 6 – water and sanitation for all, 2021.
- L. Lin, H. Yang and X. Xu, Effects of water pollution on human health and disease heterogeneity: a review, Front. Environ. Sci., 2022, 10, 880246 CrossRef.
- D. Jaspal and A. Malviya, Composites for wastewater purification: A review, Chemosphere, 2020, 246, 125788 CrossRef CAS PubMed.
- J. Schullehner, D. Cserbik, P. Gago-Ferrero, J. Lundqvist and J. R. Nuckols, Integrating different tools and technologies to advance drinking water quality exposure assessments, J. Exposure Sci. Environ. Epidemiol., 2024, 34(1), 108–114 CrossRef CAS PubMed.
- M. S. Braga, R. F. Jaimes, W. Borysow, O. F. Gomes and W. J. Salcedo, Portable multispectral colorimeter for metallic ion detection and classification, Sensors, 2017, 17(8), 1730 CrossRef PubMed.
- P. Borah, M. Kumar and P. Devi, Types of inorganic pollutants: metals/metalloids, acids, and organic forms, Inorganic Pollutants in Water, Elsevier, 2020, pp. 17–31 Search PubMed.
- X. Li, X. Shen, W. Jiang, Y. Xi and S. Li, Comprehensive review of emerging contaminants: Detection technologies, environmental impact, and management strategies, Ecotoxicol. Environ. Saf., 2024, 278, 116420 CrossRef CAS PubMed.
- R. Kumar, M. Qureshi, D. K. Vishwakarma, N. Al-Ansari, A. Kuriqi, A. Elbeltagi and A. Saraswat, A review on emerging water contaminants and the application of sustainable removal technologies, Case Stud. Chem. Environ. Eng., 2022, 6, 100219 CrossRef CAS.
- S. Dutta, D. K. Haggerty, D. A. Rappolee and D. M. Ruden, Phthalate exposure and long-term epigenomic consequences: a review, Front. Genet., 2020, 11, 405 CrossRef CAS PubMed.
- G. U. Sikakwe, N. A. Nwachukwu and B. N. Igwe, Detection of human health risks resulting from contamination of borehole drinking water with harmful volatile organic chemicals and potentially toxic elements around petroleum products dispensing outlets, J. Hazard. Mater., 2025, 138508 CrossRef CAS PubMed.
- S. Chandra and K. B. Walsh, Microplastics in water: Occurrence, fate and removal, J. Contam. Hydrol., 2024, 104360 CrossRef CAS.
- H. Kye, J. Kim, S. Ju, J. Lee, C. Lim and Y. Yoon, Microplastics in water systems: A review of their impacts on the environment and their potential hazards, Heliyon, 2023, 9, 3 CrossRef.
- S. L. Cherniak, H. Almuhtaram, M. J. McKie, L. Hermabessiere, C. Yuan, C. M. Rochman and R. C. Andrews, Conventional and biological treatment for the removal of microplastics from drinking water, Chemosphere, 2022, 288, 132587 CrossRef CAS.
- N. Qian, X. Gao, X. Lang, H. Deng, T. M. Bratu, Q. Chen, P. Stapleton, B. Yan and W. Min, Rapid single-particle chemical imaging of nanoplastics by SRS microscopy, Proc. Natl. Acad. Sci. U. S. A., 2024, 121(3), e2300582121 CrossRef CAS.
- (a) J. C. DeWitt, J. Glüge, I. T. Cousins, G. Goldenman, D. Herzke, R. Lohmann, M. Miller, C. A. Ng, S. Patton, X. Trier and L. Vierke, Zürich II Statement on Per-and Polyfluoroalkyl Substances (PFASs): Scientific and Regulatory Needs, Environ. Sci. Technol. Lett., 2024, 11(8), 786–797 CrossRef CAS; (b) M. M. R. Tushar, Z. A. Pushan, N. Aich and L. S. Rowles, Balancing sustainability goals and treatment efficacy for PFAS removal from water, npj Clean Water, 2024, 7(1), 130 CrossRef CAS.
- S. Verma, A. Daverey and A. Sharma, Slow sand filtration for water and wastewater treatment–a review, Environ. Technol. Rev., 2017, 6(1), 47–58 CrossRef CAS.
- L. Jin, X. Sun, H. Ren and H. Huang, Biological filtration for wastewater treatment in the 21st century: A data-driven analysis of hotspots, challenges and prospects, Sci. Total Environ., 2023, 855, 158951 CrossRef CAS PubMed.
- M. Samer, Biological and Chemical Wastewater Treatment, Wastewater Treat. Eng., 2015, 1–50 CAS.
- E. Obotey Ezugbe and S. Rathilal, Membrane technologies in wastewater treatment: a review, Membranes, 2020, 10(5), 89 CrossRef PubMed.
- S. Singh, V. Kumar, R. Romero, K. Sharma and J. Singh, Applications of nanoparticles in wastewater treatment, Nanobiotechnology in Bioformulations, 2019, pp. 395–418 Search PubMed.
- R. W. Sneath, M. Shaw and A. G. Williams, Centrifugation for separating piggery slurry 1. The performance of a decanting centrifuge, J. Agric. Eng. Res., 1988, 39(3), 181–190 CrossRef.
- A. A. Kiss and O. M. Kattan Readi, An industrial perspective on membrane distillation processes, J. Chem. Technol. Biotechnol., 2018, 93(8), 2047–2055 CrossRef CAS.
- A. W. Patwardhan, Rotating biological contactors: a review, Ind. Eng. Chem. Res., 2003, 42(10), 2035–2051 CrossRef CAS.
- K. Windey, I. De Bo and W. Verstraete, Oxygen-limited autotrophic nitrification–denitrification (OLAND) in a rotating biological contactor treating high-salinity wastewater, Water Res., 2005, 39(18), 4512–4520 CrossRef CAS PubMed.
- S. Cortez, P. Teixeira, R. Oliveira and M. Mota, Rotating biological contactors: a review on main factors affecting performance, Rev. Environ. Sci. Bio/Technol., 2008, 7, 155–172 CrossRef CAS.
- H. Luo, C. Liu, D. He, J. Xu, J. Sun, J. Li and X. Pan, Environmental behaviours of microplastics in aquatic systems: A systematic review on degradation, adsorption, toxicity and biofilm under aging conditions, J. Hazard. Mater., 2022, 423, 126915 CrossRef CAS.
- N. A. Khan, S. Singh, E. A. López-Maldonado, P. F. Méndez-Herrera, J. R. López-López, U. Baig, P. C. Ramamurthy, N. M. Mubarak, R. R. Karri and I. H. Aljundi, Emerging membrane technology and hybrid treatment systems for the removal of micropollutants from wastewater, Desalination, 2023, 565, 16873 CrossRef.
- C. A. Quist-Jensen, F. Macedonio and E. Drioli, Membrane technology for water production in agriculture: Desalination and wastewater reuse, Desalination, 2015, 364, 17–32 CrossRef CAS.
- D. Namla, M. Oves, M. A. Alshaeri, S. M. Al-Maaqar, H. N. Y. Issa and G. Mangse, Nanofiltration as an advanced wastewater treatment technique: a comprehensive review, Discover Appl. Sci., 2025, 7(4), 1–22 Search PubMed.
- E. Obotey Ezugbe and S. Rathilal, Membrane technologies in wastewater treatment: a review, Membranes, 2020, 10(5), 89 CrossRef.
- R. W. Baker, Microfiltration, Membrane Technology and Applications, 2012, pp. 303–324 Search PubMed.
- S. F. Ahmed, F. Mehejabin, A. Momtahin, N. Tasannum, N. T. Faria, M. Mofijur, A. T. Hoang, D. V. N. Vo and T. M. I. Mahlia, Strategies to improve membrane performance in wastewater treatment, Chemosphere, 2022, 306, 135527 CrossRef CAS.
- A. H. Behroozi, M. Al-Shaeli and V. Vatanpour, Fabrication and modification of nanofiltration membranes by solution electrospinning technique: A review of influential factors and applications in water treatment, Desalination, 2023, 558, 116638 CrossRef CAS.
- B. S. Lalia, V. Kochkodan, R. Hashaikeh and N. Hilal, A review on membrane fabrication: Structure, properties and performance relationship, Desalination, 2013, 326, 77–95 CrossRef CAS.
- W. Suwaileh, S. Attar, F. Abu-Rub, A. El-Ghenym, K. ElSaid, A. Badreldin, M. Al-Hashimi, A. Abdel-Wahab and A. Abdala, Thin-film composite forward osmosis membrane with superior alkaline stability, J. Environ. Chem. Eng., 2025, 13(1), 114909 CrossRef CAS.
- J. Di, H. Wang, L. Zhang, Z. Chen, Y. Zhang, B. B. Mamba, M. Qiu, J. Guo and L. Shao, Recent progress in advanced polyamide nanofiltration membranes via interfacial polymerization for desalination and beyond, Desalination, 2024, 118167 CrossRef CAS.
- A. I. Osman, Z. Chen, A. M. Elgarahy, M. Farghali, I. M. Mohamed, A. K. Priya, H. B. Hawash and P. S. Yap, Membrane technology for energy saving: principles, techniques, applications, challenges, and prospects, Adv. Energy Sustainability Res., 2024, 5(5), 2400011 CrossRef.
- M. A. Ahmed, S. Amin and A. A. Mohamed, Fouling in reverse osmosis membranes: monitoring, characterization, mitigation strategies and future directions, Heliyon, 2023, 4, 9 Search PubMed.
- K. Xiao, Y. Xu, X. Cao, H. Xu and Y. Li, Advanced characterization of membrane surface fouling, in 60 Years of the Loeb-Sourirajan Membrane, Elsevier, 2022, pp. 499–532 Search PubMed.
- Q. Zhao, D. L. Zhao, F. Feng, T. S. Chung and S. B. Chen, Thin-film nanocomposite reverse osmosis membranes incorporated with citrate-modified layered double hydroxides (LDHs) for brackish water desalination and boron removal, Desalination, 2022, 527, 115583 CrossRef CAS.
- A. K. Priya, L. Gnanasekaran, P. S. Kumar, A. A. Jalil, T. K. Hoang, S. Rajendran, M. Soto-Moscoso and D. Balakrishnan, Recent trends and advancements in nanoporous membranes for water purification, Chemosphere, 2022, 303, 135205 CrossRef CAS.
- L. Liu, Y. Liu, J. Huang, X. Chen, S. Feng, Y. Wan and J. Luo, Strengthening nanofiltration membrane: Strategies for enhanced antifouling performance, Chem. Eng. J., 2025, 160964 CrossRef CAS.
- X. Zhang, J. Ma, J. Zheng, R. Dai, X. Wang and Z. Wang, Recent advances in nature-inspired antifouling membranes for water purification, Chem. Eng. J., 2022, 432, 134425 CrossRef CAS.
- M. H. Mruthunjayappa, N. S. Kotrappanavar and D. Mondal, Bioinspired engineering protein nanofibrils-based multilayered self-cleaning membranes for universal water purification, J. Hazard. Mater., 2022, 424, 127 CrossRef.
- X. Lu, Y. Peng, H. Qiu, X. Liu and L. Ge, Anti-fouling membranes by manipulating surface wettability and their anti-fouling mechanism, Desalination, 2017, 413, 127–135 CrossRef CAS.
- M. Coccia and E. Bontempi, New trajectories of technologies for the removal of pollutants and emerging contaminants in the environment, Environ. Res., 2023, 229, 115938 CrossRef CAS.
- K. A. Alrefaey, A. B. Abdel-Aziz, L. A. Said, I. S. Fahim and A. G. Radwan, Carbon Nanomaterials and Their Composites as Adsorbents, Springer International Publishing, Cham, 2024, pp. 39–62 Search PubMed.
- M. Nasrollahzadeh, M. Sajjadi, S. Iravani and R. S. Varma, Carbon-based sustainable nanomaterials for water treatment: state-of-art and future perspectives, Chemosphere, 2021, 263, 128005 CrossRef CAS.
- G. K. Prashanth, M. Gadewar, H. S. Lalithamba, S. Rao, K. V. Rashmi, K. V. Yatish, M. M. Swamy, N. P. Bhagya and M. K. Ghosh, Synthesis, and applications of carbon-integrated polymer composites and foams: A concise review, Inorg. Chem. Commun., 2024, 112614 CrossRef CAS.
- N. Roy, Y. A. Kumar, T. Ramachandran, A. M. Fouda, H. H. Hegazy, M. Moniruzzaman and S. W. Joo, Biomass-derived nanostructures and hydrothermal carbon spheres: A review of electrochemical applications in redox flow battery, J. Ind. Eng. Chem., 2025, 144, 228–254 CrossRef CAS.
- Y. Zhao, T. Ramachandran, A. Ghosh, A. G. Al-Sehemi, Y. A. Kumar, S. S. Rao, A. K. Yadav and D. Mani, Unlocking the potential of vanadium redox flow batteries: Recent advances in biomass lignin-based carbon fibers and future outlook, Biomass Bioenergy, 2025, 200, 108052 CrossRef CAS.
- M. B. Tahir Rasheed, F. Nabeel, M. Adeel and H. M. Iqbal, Environmentally related contaminants of high concern: Potential sources and analytical modalities for detection, quantification, and treatment, Environ. Int., 2019, 122, 52–66 CrossRef.
- M. Arbabi and N. Golshani, Removal of copper ions Cu (II) from industrial wastewater: A review of removal methods, Epidemiol. Health Syst. J., 2016, 3(3), 283–293 Search PubMed.
- S. Nizamuddin, M. T. H. Siddiqui, N. M. Mubarak, H. A. Baloch, E. C. Abdullah, S. A. Mazari, G. J. Griffin, M. P. Srinivasan and A. Tanksale, Iron oxide nanomaterials for the removal of heavy metals and dyes from wastewater, Nanoscale Mater. Water Purif., 2019, 447–472 CAS.
- S. Manimegalai, S. Vickram, S. R. Deena, K. Rohini, S. Thanigaivel, S. Manikandan, R. Subbaiya, N. Karmegam, W. Kim and M. Govarthanan, Carbon-based nanomaterial intervention and efficient removal of various contaminants from effluents–A review, Chemosphere, 2023, 312, 137319 CrossRef CAS.
- N. Jayaprakash, K. Elumalai, S. Manickam, G. Bakthavatchalam and P. Tamilselvan, Carbon nanomaterials: Revolutionizing biomedical applications with promising potential, Nano Mater. Sci., 2024 Search PubMed.
- L. L. Mokoloko, R. P. Forbes and N. J. Coville, The Transformation of 0-D Carbon Dots into 1-, 2-and 3-D Carbon Allotropes: A Minireview, Nanomaterials, 2022, 12, 2515 CrossRef CAS PubMed.
- D. Maiti, X. Tong, X. Mou and K. Yang, Carbon-based nanomaterials for biomedical applications: a recent study, Front. Pharmacol., 2019, 9, 1401 CrossRef.
- S. Zhang, Y. Ma, L. Suresh, A. Hao, M. Bick, S. C. Tan and J. Chen, Carbon nanotube reinforced strong carbon matrix composites, ACS Nano, 2020, 14(8), 9282–9319 CrossRef CAS.
- R. Ghasempour and H. Narei, CNT basics and characteristics, in Carbon Nanotube-Reinforced Polymers, Elsevier, 2018, pp. 1–24 Search PubMed.
- J. L. Blackburn, A. J. Ferguson, C. Cho and J. C. Grunlan, Thermoelectric materials: carbon-nanotube-based thermoelectric materials and devices, Adv. Mater., 2018, 30(11), 1870072 CrossRef.
- S. Shalu, K. Dasgupta, A. Kumari and B. D. Ghosh, Carbon Nanotubes: A Concise Review of The Synthesis Techniques, Properties, and Applications, Carbon Nanotubes and Nanoparticles, 2019, pp. 81–106 Search PubMed.
- Á. Kukovecz, G. Kozma and Z. Kónya, Multi-walled Carbon Nanotubes, Springer handbook of nanomaterials, 2013, pp. 147–188 Search PubMed.
- C. H. See and A. T. Harris, A review of carbon nanotube synthesis via fluidized-bed chemical vapor deposition, Ind. Eng. Chem. Res., 2007, 46(4), 997–1012 CrossRef CAS.
- A. Aqel, K. M. Abou El-Nour, R. A. Ammar and A. Al-Warthan, Carbon nanotubes, science and technology part (I) structure, synthesis and characterisation, Arabian J. Chem., 2012, 5(1), 1–23 CrossRef CAS.
- Y. Ando and S. Iijima, Preparation of carbon nanotubes by arc-discharge evaporation, Jpn. J. Appl. Phys., Part 2, 1993, 32, L107 CrossRef CAS.
- A. K. Madikere Raghunatha Reddy, A. Darwiche, M. V. Reddy and K. Zaghib, Review on Advancements in Carbon Nanotubes: Synthesis, Purification, and Multifaceted Applications, Batteries, 2025, 11(2), 71 CrossRef CAS.
- A. E. Agboola, Development and Model Formulation of Scalable Carbon Nanotube Processes: HiPCO and CoMoCAT Process Models, Louisiana State University and Agricultural & Mechanical College, 2005 Search PubMed.
- V. Negri, J. Pacheco-Torres, D. Calle and P. López-Larrubia, Carbon Nanotubes in Biomedicine. Surface-Modified Nanobiomaterials for Electrochemical and Biomedicine Applications, Springer, 2020, pp. 177–217 Search PubMed.
- R. Siddheswaran, D. Manikandan, R. E. Avila, C. E. Jeyanthi and R. V. Mangalaraja, Formation of carbon nanotube forest over spin-coated Fe2O3 reduced thin-film by chemical vapor deposition, Fullerenes, Nanotubes Carbon Nanostruct., 2015, 23(5), 392–398 CrossRef CAS.
- B. P. Singh, D. K. Saket, A. P. Singh, S. Pati, T. K. Gupta, V. N. Singh, S. R. Dhakate, S. K. Dhawan, R. K. Kotnala and R. B. Mathur, Microwave shielding properties of Co/Ni attached to single walled carbon nanotubes, J. Mater. Chem. A, 2015, 3(25), 13203–13209 RSC.
- R. Das, Z. Shahnavaz, M. E. Ali, M. M. Islam and S. B. Abd Hamid, Can we optimize arc discharge and laser ablation for well-controlled carbon nanotube synthesis?, Nanoscale Res. Lett., 2016,(1), 510 CrossRef.
- X. Liu, M. Wang, S. Zhang and B. Pan, Application potential of carbon nanotubes in water treatment: a review, J. Environ. Sci., 2013, 25(7), 1263–1280 CrossRef CAS.
- B. Arora and P. Attri, Carbon nanotubes (CNTs): a potential nanomaterial for water purification, J. Compos. Sci., 2020, 4(3), 135 CrossRef CAS.
- M. H. Dehghani, M. M. Taher, A. K. Bajpai, B. Heibati, I. Tyagi, M. Asif, S. Agarwal and V. K. Gupta, Removal of noxious Cr (VI) ions using single-walled carbon nanotubes and multi-walled carbon nanotubes, Chem. Eng. J., 2015, 279, 344–352 CrossRef CAS.
- K. Pillay, E. M. Cukrowska and N. J. Coville, Multi-walled carbon nanotubes as adsorbents for the removal of parts per billion levels of hexavalent chromium from aqueous solution, J. Hazard. Mater., 2009, 166(2–3), 1067–1075 CrossRef CAS.
- M. A. Atieh, O. Y. Bakather, B. S. Tawabini, A. A. Bukhari, M. Khaled, M. Alharthi, M. Fettouhi and F. A. Abuilaiw, Removal of chromium (III) from water by using modified and nonmodified carbon nanotubes, J. Nanomater., 2010, 2010(1), 232378 CrossRef.
- A. Mudhoo and M. Sillanpää, Magnetic nanoadsorbents for micropollutant removal in real water treatment: a review, Environ. Chem. Lett., 2021, 19(6), 4393–4413 CrossRef CAS.
- J. Xu, Z. Cao, Y. Zhang, Z. Yuan, Z. Lou, X. Xu and X. Wang, A review of functionalized carbon nanotubes and graphene for heavy metal adsorption from water: Preparation, application, and mechanism, Chemosphere, 2018, 195, 351–364 CrossRef CAS PubMed.
- C. Luo, Z. Tian, B. Yang, L. Zhang and S. Yan, Manganese dioxide/iron oxide/acid oxidized multi-walled carbon nanotube magnetic nanocomposite for enhanced hexavalent chromium removal, Chem. Eng. J., 2013, 234, 256–265 CrossRef CAS.
- M. K. Kim, K. S. Sundaram, G. A. Iyengar and K. P. Lee, A novel chitosan functional gel included with multiwall carbon nanotube and substituted polyaniline as adsorbent for efficient removal of chromium ion, Chem. Eng. J., 2015, 267, 51–64 CrossRef CAS.
- M. Bhaumik, S. Agarwal, V. K. Gupta and A. Maity, Enhanced removal of Cr (VI) from aqueous solutions using polypyrrole wrapped oxidized MWCNTs nanocomposites adsorbent, J. Colloid Interface Sci., 2016, 470, 257–267 CrossRef CAS.
- D. Jia, Z. Jing, Y. Duan and J. Li, Ultrafast removal of Cr (VI) ions using polyamine modified carbon nanotubes, J. Taiwan Inst. Chem. Eng., 2022, 133, 104265 CrossRef CAS.
- L. Wang, Y. Zhu, L. Ma, X. Hai, X. Li, Z. Yang, Y. Gao, M. Yuan, H. Xiong, M. Chen and X. Ma, Efficient removal of Chromium (VI) from wastewater based on magnetic multiwalled carbon nanotubes coupled with deep eutectic solvents, Chemosphere, 2024, 362, 142732 CrossRef CAS PubMed.
- I. Ali, Microwave assisted economic synthesis of multi walled carbon nanotubes for arsenic species removal in water: batch and column operations, J. Mol. Liq., 2018, 271, 677–685 CrossRef CAS.
- H. Alijani and Z. Shariatinia, Effective aqueous arsenic removal using zero valent iron doped MWCNT synthesized by in situ CVD method using natural α-Fe2O3 as a precursor, Chemosphere, 2017, 171, 502–511 CrossRef CAS.
- N. Sankararamakrishnan, D. Chauhan and J. Dwivedi, Synthesis of functionalized carbon nanotubes by floating catalytic chemical vapor deposition method and their sorption behavior toward arsenic, Chem. Eng. J., 2016, 284, 599–608 CrossRef CAS.
- Z. Li, J. Chen and Y. Ge, Removal of lead ion and oil droplet from aqueous solution by lignin-grafted carbon nanotubes, Chem. Eng. J., 2017, 308, 809–817 CrossRef CAS.
- N. M. Mubarak, J. N. Sahu, E. C. Abdullah, N. S. Jayakumar and P. Ganesan, Microwave assisted multiwall carbon nanotubes enhancing Cd (II) adsorption capacity in aqueous media, J. Ind. Eng. Chem., 2015, 24, 24–33 CrossRef CAS.
- D. Shao, J. Hu and X. Wang, Plasma induced grafting multiwalled carbon nanotube with chitosan and its application for removal of UO, Cu2+, and Pb2+ from aqueous solutions, Plasma Processes Polym., 2010, 7(12), 977–985 CrossRef CAS.
- B. Fugetsu, S. Satoh, T. Shiba, T. Mizutani, Y. B. Lin, N. Terui, Y. Nodasaka, K. Sasa, K. Shimizu, T. Akasaka and M. Shindoh, Caged multiwalled carbon nanotubes as the adsorbents for affinity-based elimination of ionic dyes, Environ. Sci. Technol., 2004, 38(24), 6890–6896 CrossRef CAS PubMed.
- M. Ghaedi, A. Shokrollahi, H. Hossainian and S. N. Kokhdan, Comparison of activated carbon and multiwalled carbon nanotubes for efficient removal of Eriochrome cyanine R (ECR): kinetic, isotherm, and thermodynamic study of the removal process, J. Chem. Eng. Data, 2011, 56(7), 3227–3235 CrossRef CAS.
- J. Ma, F. Yu, L. Zhou, L. Jin, M. Yang, J. Luan, Y. Tang, H. Fan, Z. Yuan and J. Chen, Enhanced adsorptive removal of methyl orange and methylene blue from aqueous solution by alkali-activated multiwalled carbon nanotubes, ACS Appl. Mater. Interfaces, 2012, 4(11), 5749–5760 CrossRef CAS PubMed.
- S. Qu, F. Huang, S. Yu, G. Chen and J. Kong, Magnetic removal of dyes from aqueous solution using multi-walled carbon nanotubes filled with Fe2O3 particles, J. Hazard. Mater., 2008, 160(2–3), 643–647 CrossRef CAS PubMed.
- J. L. Gong, B. Wang, G. M. Zeng, C. P. Yang, C. G. Niu, Q. Y. Niu, W. J. Zhou and Y. Liang, Removal of cationic dyes from aqueous solution using magnetic multi-wall carbon nanotube nanocomposite as adsorbent, J. Hazard. Mater., 2009, 164(2–3), 1517–1522 CrossRef CAS PubMed.
- S. Mallakpour and S. Rashidimoghadam, Poly (vinyl alcohol)/Vitamin C-multi walled carbon nanotubes composites and their applications for removal of methylene blue: Advanced comparison between linear and nonlinear forms of adsorption isotherms and kinetics models, Polymer, 2019, 160, 115–125 CrossRef CAS.
- B. Deepanraj, V. Tirth, A. Algahtani and E. M. Elsehly, The performance of multi-walled carbon nanotubes-based filters with acid functionalization for enhanced methylene blue removal from water resources, Alexandria Eng. J., 2024, 88, 310–316 CrossRef.
- H. Gao, S. Zhao, X. Cheng, X. Wang and L. Zheng, Removal of anionic azo dyes from aqueous solution using magnetic polymer multi-wall carbon nanotube nanocomposite as adsorbent, Chem. Eng. J., 2013, 223, 84–90 CrossRef CAS.
- S. Zghal, I. Jedidi, M. Cretin, S. Cerneaux and M. Abdelmouleh, Adsorptive removal of Rhodamine B dye using carbon graphite/cnt composites as adsorbents: Kinetics, isotherms and thermodynamic study, Materials, 2023, 16(3), 1015 CrossRef CAS PubMed.
- B. Zhao, Y. Zhao, P. Liu, Y. L. Men and Y. X. Pan, Boosting the adsorption and removal of dye from water by COOH-functionalized carbon nanotubes, Green Chem. Eng., 2023, 4(1), 88–98 CrossRef CAS.
- A. Azari, R. Nabizadeh, A. H. Mahvi and S. Nasseri, Magnetic multi walled carbon nanotubes-loaded alginate for treatment of industrial dye manufacturing effluent: adsorption modelling and process optimisation by central composite face-central design, Int. J. Environ. Anal. Chem., 2021, 103(7), 1509–1529 CrossRef.
- L. Yanyan, T. A. Kurniawan, A. B. Albadarin and G. Walker, Enhanced removal of acetaminophen from synthetic wastewater using multi-walled carbon nanotubes (MWCNTs) chemically modified with NaOH, HNO3/H2SO4, ozone, and/or chitosan, J. Mol. Liq., 2018, 251, 369–377 CrossRef.
- F. Yu, S. Sun, S. Han, J. Zheng and J. Ma, Adsorption removal of ciprofloxacin by multi-walled carbon nanotubes with different oxygen contents from aqueous solutions, Chem. Eng. J., 2016, 285, 588–595 CrossRef CAS.
- W. Chen, F. Wang, X. Hou, M. Li, L. Jia, X. Li, S. Li, X. Zhang, H. Wang and Z. Bian, Efficient degradation of tetracycline contaminants by a flow-through Electro-Fenton system:Carbon nanotube-encapsulated α-Fe2O3 under nanoconfinement, Chem. Eng. J., 2025, 507, 160449 CrossRef CAS.
- W. Chen, L. Duan and D. Zhu, Adsorption of polar and nonpolar organic chemicals to carbon nanotubes, Environ. Sci. Technol., 2007, 41(24), 8295–8300 CrossRef CAS.
- X. Peng, Y. Li, Z. Luan, Z. Di, H. Wang, B. Tian and Z. Jia, Adsorption of 1,2- dichlorobenzene from water to carbon nanotubes, Chem. Phys. Lett., 2003, 376(1–2), 154–158 CrossRef CAS.
- C. Lu, Y. L. Chung and K. F. Chang, Adsorption of trihalomethanes from water with carbon nanotubes, Water Res., 2005, 39(6), 1183–1189 CrossRef CAS.
- C. Lu, Y. L. Chung and K. F. Chang, Adsorption thermodynamic and kinetic studies of trihalomethanes on multiwalled carbon nanotubes, J. Hazard. Mater., 2006, 138(2), 304–310 CrossRef CAS.
- G. K. Wong and R. D. Webster, Active and passive adsorption of 47 trace atmospheric volatile organic compounds onto carbon nanotubes, RSC Adv., 2021, 11(48), 29913–29919 RSC.
- R. Q. Long and R. T. Yang, Carbon nanotubes as superior sorbent for dioxin removal, J. Am. Chem. Soc., 2001, 123(9), 2058–2059 CrossRef CAS PubMed.
- L. Joseph, J. Heo, Y. G. Park, J. R. Flora and Y. Yoon, Adsorption of bisphenol A and 17α-ethinyl estradiol on single walled carbon nanotubes from seawater and brackish water, Desalination, 2011, 281, 68–74 CrossRef CAS.
- X. Guo, Y. Huang, W. Yu, X. Yu, X. Han and H. Zhai, Multi-walled carbon nanotubes modified with iron oxide and manganese dioxide (MWCNTs-Fe3O4− MnO2) as a novel adsorbent for the determination of BPA, Microchem. J., 2020, 157, 104867 CrossRef CAS.
- J. Zheng, M. Li, K. Yu, J. Hu, X. Zhang and L. Wang, Sulfonated multiwall carbon nanotubes assisted thin-film nanocomposite membrane with enhanced water flux and anti-fouling property, J. Membr. Sci., 2017, 524, 344–353 CrossRef CAS.
- C. Zhao, C. Liu, W. Wang, Z. Zhang, X. Yang, J. Tian, X. Zhang and B. Zhao, Interfacial polymerization layer with CNT providing fast water channels under electric field for efficient desalination of nanofiltration membranes, Desalination, 2023, 565, 116825 CrossRef CAS.
- P. Ares and K. S. Novoselov, Recent advances in graphene and other 2D materials, Nano Mater. Sci., 2022, 4(1), 3–9 CrossRef CAS.
- E. Gerstner, Nobel prize 2010: Andre geim & Konstantin Novoselov, Nat. Phys., 2010, 6(11), 836 Search PubMed.
- S. Ren, P. Rong and Q. Yu, Preparations, properties and applications of graphene in functional devices: A concise review, Ceram. Int., 2018, 44(11), 11940–11955 CrossRef CAS.
- S. K. Tiwari, S. Sahoo, N. Wang and A. Huczko, Graphene research and their outputs: Status and prospect, J. Sci.: Adv. Mater. Devices, 2020, 5(1), 10–29 Search PubMed.
- N. Slepičková Kasálková, P. Slepička and V. Švorčík, Carbon nanostructures, nanolayers, and their composites, Nanomaterials, 2021, 11(9), 2368 CrossRef.
- G. Yang, L. Li, W. B. Lee and M. C. Ng, Structure of graphene and its disorders: a review, Sci. Technol. Adv. Mater., 2018, 19(1), 613–648 CrossRef CAS PubMed.
- J. Luis Valverde Palomino, A. Romero Izquierdo, M. Sánchez Silva and M. del Prado Lavín López, Chapter: Optimization of the Synthesis Procedures of Graphene and Graphite Oxide, 2016 Search PubMed.
- Y. Wu, S. Wang and K. Komvopoulos, A review of graphene synthesis by indirect and direct deposition methods, J. Mater. Res., 2020, 35(1), 76–89 CrossRef CAS.
- A. Adetayo and D. Runsewe, Synthesis and fabrication of graphene and graphene oxide: a review, Open J. Compos. Mater., 2019, 9(02), 207 CAS.
- L. Staudenmaier, Verfahren zur darstellung der graphitsäure, Ber. Dtsch. Chem. Ges., 1898, 31(2), 1481–1487 CrossRef CAS.
- W. S. Hummers Jr and R. E. Offeman, Preparation of graphitic oxide, J. Am. Chem. Soc., 1958, 80(6), 1339 CrossRef.
- M. C. Costa, V. S. Marangoni, P. R. Ng, H. T. Nguyen, A. Carvalho and A. H. Castro Neto, Accelerated synthesis of graphene oxide from graphene, Nanomaterials, 2021, 11(2), 551 CrossRef CAS PubMed.
- D. Chen, L. Tang and J. Li, Graphene-based materials in electrochemistry, Chem. Soc. Rev., 2010, 39(8), 3157–3180 RSC.
- O. C. Compton and S. T. Nguyen, Graphene oxide, highly reduced graphene oxide, and graphene: versatile building blocks for carbon-based materials, Small, 2010, 6(6), 711–723 CrossRef CAS PubMed.
- N. M. S. Hidayah, W. W. Liu, C. W. Lai, N. Z. Noriman, C. S. Khe, U. Hashim and H. C. Lee, Comparison on graphite, graphene oxide and reduced graphene oxide: Synthesis and characterization, in AIP Conference Proceedings, AIP Publishing, 2017, vol. 1892, no. 1 Search PubMed.
- N. Morimoto, T. Kubo and Y. Nishina, Tailoring the oxygen content of graphite and reduced graphene oxide for specific applications, Sci. Rep., 2016, 6(1), 21715 CrossRef CAS.
- T. Van Khai, H. G. Na, D. S. Kwak, Y. J. Kwon, H. Ham, K. B. Shim and H. W. Kim, Significant enhancement of blue emission and electrical conductivity of N-doped graphene, J. Mater. Chem., 2012, 22(34), 17992–18003 RSC.
- B. W. Pratama and W. S. B. Dwandaru, Synthesis of reduced graphene oxide based on thermally modified liquid-phase exfoliation, Nano Express, 2020, 1(1), 010023 CrossRef CAS.
- Z. Y. Xia, S. Pezzini, E. Treossi, G. Giambastiani, F. Corticelli, V. Morandi, A. Zanelli, V. Bellani and V. Palermo, The exfoliation of graphene in liquids by electrochemical, chemical, and sonication-assisted techniques: a nanoscale study, Adv. Funct. Mater., 2013, 23(37), 4684–4693 CrossRef CAS.
- M. C. Costa, V. S. Marangoni, P. R. Ng, H. T. Nguyen, A. Carvalho and A. H. Castro Neto, Accelerated synthesis of graphene oxide from graphene, Nanomaterials, 2021, 11(2), 551 CrossRef CAS.
- H. Bi, X. Xie, K. Yin, Y. Zhou, S. Wan, L. He, F. Xu, F. Banhart, L. Sun and R. S. Ruoff, Spongy graphene as a highly efficient and recyclable sorbent for oils and organic solvents, Adv. Funct. Mater., 2012, 22(21), 4421–4425 CrossRef CAS.
- C. A. Crock, A. R. Rogensues, W. Shan and V. V. Tarabara, Polymer nanocomposites with graphene-based hierarchical fillers as materials for multifunctional water treatment membranes, Water Res., 2013, 47(12), 3984–3996 CrossRef CAS.
- C. A. Crock, R. Şengür-Taşdemir, İ. Koyuncu and V. V. Tarabara, High throughput catalytic dechlorination of TCE by hollow fiber nanocomposite membranes with embedded Pd and Pd-Au catalysts, Sep. Purif. Technol., 2017, 179, 265–273 CrossRef CAS.
- S. Khaliha, T. D. Marforio, A. Kovtun, S. Mantovani, A. Bianchi, M. L. Navacchia, M. Zambianchi, L. Bocchi, N. Boulanger, A. Iakunkov and M. Calvaresi, Defective graphene nanosheets for drinking water purification: Adsorption mechanism, performance, and recovery, FlatChem, 2021, 29, 100283 CrossRef CAS.
- V. F. Medina, J. Mattei-Sosa and C. S. Griggs, Method of Recycling Chitosan and Graphene Oxide Compound, US Pat., 10,414,659, 2019 Search PubMed.
- S. Khaliha, A. Bianchi, A. Kovtun, F. Tunioli, A. Boschi, M. Zambianchi, D. Paci, L. Bocchi, S. Valsecchi, S. Polesello and A. Liscio, Graphene oxide nanosheets for drinking water purification by tandem adsorption and microfiltration, Sep. Purif. Technol., 2022, 300, 121826 CrossRef CAS.
- M. N. Pervez, T. Jiang, J. K. Mahato, A. K. Ilango, Y. Kumaran, Y. Zuo, W. Zhang, H. Efstathiadis, J. I. Feldblyum, M. V. Yigit and Y. Liang, Surface modification of graphene oxide for fast removal of per-and polyfluoroalkyl substances (PFAS) mixtures from river water, ACS ES&T Water, 2024, 4(7), 2968–2980 Search PubMed.
- A. Trifoglio, S. Mantovani, S. Khaliha, A. Kovtun, T. D. Marforio, M. Calvaresi and M. Melucci, Tailoring graphene oxide nanosheets by alkyl amine grafting for enhanced adsorption of PFASs in drinking water: a combined theoretical and experimental study, Nanoscale, 2025, 17, 12124–12133 RSC.
- E. Mahofa, S. El Meragawi, M. A. Vilayatteri, S. Dwivedi, M. R. Panda, P. Jovanović, A. C. van Duin, B. Freeman, A. Tanksale and M. Majumder, Manipulating Intrapore Energy Barriers in Graphene Oxide Nanochannels for Targeted Removal of Short-Chain PFAS, ACS Nano, 2025, 19, 14742–14755 CrossRef CAS.
- H. Wu, B. Tang and P. Wu, Development of novel SiO2–GO nanohybrid/polysulfone membrane with enhanced performance, J. Membr. Sci., 2014, 451, 94–102 CrossRef CAS.
- M. Ionita, L. E. Crica, S. I. Voicu, A. M. Pandele and H. Iovu, Fabrication of cellulose triacetate/graphene oxide porous membrane, Polym. Adv. Technol., 2016, 27(3), 350–357 CrossRef CAS.
- S. V. Nipane, P. V. Korake and G. S. Gokavi, Graphene-zinc oxide nanorod nanocomposite as photocatalyst for enhanced degradation of dyes under UV light irradiation, Ceram. Int., 2015, 41(3), 4549–4557 CrossRef CAS.
- H. N. Tien and S. H. Hur, Fabrication of 3D structured ZnO nanorod/reduced graphene oxide hydrogels and their use for photo-enhanced organic dye removal, J. Colloid Interface Sci., 2015, 437, 181–186 CrossRef.
- H. Sun, L. Cao and L. Lu, Magnetite/reduced graphene oxide nanocomposites: one step solvothermal synthesis and use as a novel platform for removal of dye pollutants, Nano Res., 2011, 4, 550–562 CrossRef CAS.
- A. R. B. Bayantong, Y. J. Shih, D. C. Ong, R. R. M. Abarca, C. D. Dong and M. D. G. de Luna, Adsorptive removal of dye in wastewater by metal ferrite-enabled graphene oxide nanocomposites, Chemosphere, 2021, 274, 129518 CrossRef CAS PubMed.
- Y. Pan, Z. Shi, J. Li, Z. Zhang, X. Li, Z. Zhuang, Y. Mo, J. Liang, Z. Wang, M. An and Q. Luo, Graphene oxide laminates intercalated with Prussian blue nanocube as a photo-Fenton self-cleaning membrane for enhanced water purification, J. Membr. Sci., 2023, 672, 121465 CrossRef CAS.
- J. A. Prince, S. Bhuvana, V. Anbharasi, N. Ayyanar, K. V. K. Boodhoo and G. Singh, Ultra-wetting graphene-based PES ultrafiltration membrane–a novel approach for successful oil-water separation, Water Res., 2016, 103, 311–318 CrossRef CAS.
- D. V. Schmidt, T. L. Costa, D. F. Cipriano, C. S. Meireles, C. J. Dalmaschio and J. C. Freitas, Biomass-Derived Cellulose Acetate Membranes Modified with TiO2/Graphene Oxide for Oil-In-Water Emulsion Treatment, ACS Omega, 2024, 9(39), 40882–40896 CrossRef CAS.
- T. Ramachandran, N. Roy, H. H. Hegazy, I. S. Yahia, Y. A. Kumar, M. Moniruzzaman and S. W. Joo, From graphene aerogels to efficient energy storage: current developments and future prospects, J. Alloys Compd., 2025, 1010, 177248 CrossRef CAS.
- R. Rezaee, S. Nasseri, A. H. Mahvi, R. Nabizadeh, S. A. Mousavi, A. Rashidi, A. Jafari and S. Nazmara, Fabrication and characterization of a polysulfone-graphene oxide nanocomposite membrane for arsenate rejection from water, J. Environ. Health Sci. Eng., 2015, 13, 1–11 CrossRef CAS PubMed.
- A. Shahzad, W. Miran, K. Rasool, M. Nawaz, J. Jang, S. R. Lim and D. S. Lee, Heavy metals removal by EDTA-functionalized chitosan graphene oxide nanocomposites, RSC Adv., 2017, 7, 9764–9771 RSC.
- A. S. K. Kumar, S. S. Kakan and N. Rajesh, A novel amine impregnated graphene oxide adsorbent for the removal of hexavalent chromium, Chem. Eng. J., 2013, 230, 328–337 CrossRef CAS.
- M. S. Samuel, J. Bhattacharya, S. Raj, N. Santhanam, H. Singh and N. P. Singh, Efficient removal of Chromium (VI) from aqueous solution using chitosan grafted graphene oxide (CS-GO) nanocomposite, Int. J. Biol. Macromol., 2019, 121, 285–292 CrossRef CAS PubMed.
- S. Singh, A. G. Anil and B. Uppara, Adsorption and DFT investigations of Cr (VI) removal using nanocrystals decorated with graphene oxide, npj Clean Water, 2024, 7, 17 CrossRef CAS.
- A. Sheikhmohammadi, S. M. Mohseni, M. Sardar, M. Abtahi, S. Mahdavi, H. Keramati, Z. Dahaghin, S. Rezaei, M. Almasian, M. Sarkhosh and M. Faraji, Application of graphene oxide modified with 8-hydroxyquinoline for the adsorption of Cr (VI) from wastewater: optimization, kinetic, thermodynamic and equilibrium studies, J. Mol. Liq., 2017, 233, 75–88 CrossRef CAS.
- Y. Chen, Q. Zhang, L. Chen, H. Bai and L. Li, Basic aluminum sulfate@ graphene hydrogel composites: preparation and application for removal of fluoride, J. Mater. Chem. A, 2013, 1(42), 13101–13110 RSC.
- V. Chandra, J. Park, Y. Chun, J. W. Lee, I. C. Hwang and K. S. Kim, Water-dispersible magnetite-reduced graphene oxide composites for arsenic removal, ACS Nano, 2010, 4(7), 3979–3986 CrossRef CAS.
- A. Pryadko, Y. R. Mukhortova, V. V. Botvin, I. Y. Grubova, M. R. Galstenkova, D. V. Wagner, E. Y. Gerasimov, E. V. Sukhinina, A. G. Pershina, A. L. Kholkin and M. A. Surmeneva, A comprehensive study on in situ synthesis of a magnetic nanocomposite of magnetite and reduced graphene oxide and its effectiveness at removing arsenic from water, Nano-Struct. Nano-Objects, 2023, 36, 101028 CrossRef CAS.
- X. Luo, C. Wang, S. Luo, R. Dong, X. Tu and G. Zeng, Adsorption of As (III) and As (V) from water using magnetite Fe3O4-reduced graphite oxide–MnO2 nanocomposites, Chem. Eng. J., 2012, 187, 45–52 CrossRef CAS.
- H. A. Maitlo, K. H. Kim, V. Kumar, S. Kim and J. W. Park, Nanomaterials-based treatment options for chromium in aqueous environments, Environ. Int., 2019, 130, 104748 CrossRef CAS PubMed.
- H. V. Tran, T. L. Tran, T. D. Le, T. D. Le, H. M. Nguyen and L. T. Dang, Graphene oxide enhanced adsorption capacity of chitosan/magnetite nanocomposite for Cr (VI) removal from aqueous solution, Mater. Res. Express, 2018, 6(2), 025018 CrossRef.
- C. He, Z. Yang, J. Ding, Y. Chen, X. Tong and Y. Li, Effective removal of Cr (VI) from aqueous solution by 3-aminopropyltriethoxysilane-functionalized graphene oxide, Colloids Surf., A, 2017, 520, 448–458 CrossRef CAS.
- K. Zhang, H. Li, X. Xu and H. Yu, Synthesis of reduced graphene oxide/NiO nanocomposites for the removal of Cr (VI) from aqueous water by adsorption, Microporous Mesoporous Mater., 2018, 255, 7–14 CrossRef CAS.
- M. S. Samuel, V. Subramaniyan, J. Bhattacharya, C. Parthiban, S. Chand and N. P. Singh, A GO-CS@ MOF [Zn (BDC)(DMF)] material for the adsorption of chromium (VI) ions from aqueous solution, Composites, Part B, 2018, 152, 116–125 CrossRef CAS.
- F. A. Ibrahim and S. M. A. E. H. Mosa, Graphite recycling by doping nano materials for water treatment, J. Inorg. Organomet. Polym. Mater., 2024, 34(4), 1564–1571 CrossRef CAS.
- F. Soomro, F. H. Memon, M. Khan, M. Iqbal, A. Ibrar, A. A. Memon, J. H. Lim, K. H. Choi and K. H. Thebo, Ultrathin graphene oxide-based nanocomposite membranes for water purification, Membranes, 2023, 13(1), 64 CrossRef CAS PubMed.
- S. J. Schmidt, W. Dou and S. A. Sydlik, Regeneratable graphene-based water filters for heavy metal removal at home, ACS ES&T Water, 2023, 3(8), 2179–2185 Search PubMed.
- T. Wu, X. Cai, S. Tan, H. Li, J. Liu and W. Yang, Adsorption characteristics of acrylonitrile, p-toluenesulfonic acid, 1-naphthalenesulfonic acid and methyl blue on graphene in aqueous solutions, Chem. Eng. J., 2011, 173(1), 144–149 CrossRef CAS.
- L. Yu, L. Wang, W. Xu, L. Chen, M. Fu, J. Wu and D. Ye, Adsorption of VOCs on reduced graphene oxide, J. Environ. Sci., 2018, 67, 171–178 CrossRef CAS.
- H. Li, Y. Cheng, J. Li, T. Li, J. Zhu, W. Deng, J. Zhu and D. He, Preparation and adsorption performance study of graphene quantum dots@ ZIF-8 composites for highly efficient removal of volatile organic compounds, Nanomaterials, 2022, 12(22), 4008 CrossRef CAS PubMed.
- G. Gorgolis, M. Kotsidi, G. Paterakis, N. Koutroumanis, C. Tsakonas and C. Galiotis, Graphene aerogels as efficient adsorbers of water pollutants and their effect on drying methods, Sci. Rep., 2024, 14(1), 8029 CrossRef CAS PubMed.
- R. H. Baughman, H. Eckhardt and M. Kertesz, Structure-property predictions for new planar forms of carbon: Layered phases containing sp2 and sp atoms, J. Chem. Phys., 1987, 87(11), 6687–6699 CrossRef CAS.
- A. Hayat, M. Sohail, S. B. Moussa, M. K. Al-Muhanna, W. Iqbal, Z. Ajmal, S. Raza, Y. Al-Hadeethi and Y. Orooji, State, synthesis, perspective applications, and challenges of Graphdiyne and its analogues: A review of recent research, Adv. Colloid Interface Sci., 2023, 319, 102969 CrossRef CAS.
- (a) S. W. Cranford, D. B. Brommer and M. J. Buehler, Extended graphynes: simple scaling laws for stiffness, strength and fracture, Nanoscale, 2012, 4(24), 7797–7809 RSC; (b) J. Li and Y. Han, Artificial carbon allotrope γ-graphyne: Synthesis, properties, and applications, Giant, 2023, 13, 100140 CrossRef CAS; (c) Z. Zhou, Y. Tan, Q. Yang, A. Bera, Z. Xiong, M. Yagmurcukardes, M. Kim, Y. Zou, G. Wang, A. Mishchenko and I. Timokhin, Gas permeation through graphdiyne-based nanoporous membranes, Nat. Commun., 2022, 13(1), 4031 CrossRef CAS PubMed; (d) T. Song, H. Liu, H. Zou, C. Wang, S. Shu, H. Dai and L. Duan, Metal-Free Wet Chemistry for the Fast Gram-Scale Synthesis of γ-Graphyne and its Derivatives, Angew. Chem., Int. Ed., 2024, 63(50), e202411228 CrossRef CAS.
- Q. Li, Y. Li, Y. Chen, L. Wu, C. Yang and X. Cui, Synthesis of γ-graphyne by mechanochemistry and its electronic structure, Carbon, 2018, 136, 248–254 CrossRef CAS.
- C. Yang, Y. Li, Y. Chen, Q. Li, L. Wu and X. Cui, Mechanochemical Synthesis of γ-Graphyne with Enhanced Lithium Storage Performance, Small, 2019, 15(8), 1804710 CrossRef.
- M. Barua, A. Saraswat and C. N. Rao, A novel method for synthesis of γ-graphyne and their charge transfer properties, Carbon, 2022, 200, 247–252 CrossRef CAS.
- J. A. Marsden and M. M. Haley, Carbon networks based on dehydrobenzoannulenes. 5. Extension of two-dimensional conjugation in graphdiyne nanoarchitectures, J. Org. Chem., 2005, 70(25), 10213–10226 CrossRef CAS.
- K. Sonogashira, Y. Tohda and N. Hagihara, A convenient synthesis of acetylenes: catalytic substitutions of acetylenic hydrogen with bromoalkenes, iodoarenes and bromopyridines, Tetrahedron Lett., 1975, 16(50), 4467–4470 CrossRef.
- J. Zhou, J. Li, Z. Liu and J. Zhang, Exploring approaches for the synthesis of few-layered graphdiyne, Adv. Mater., 2019, 31(42), 1803758 CrossRef CAS PubMed.
- R. Sakamoto, N. Fukui, H. Maeda, R. Matsuoka, R. Toyoda and H. Nishihara, The accelerating world of graphdiynes, Adv. Mater., 2019, 31(42), 1804211 CrossRef CAS PubMed.
- G. Li, Y. Li, X. Qian, H. Liu, H. Lin, N. Chen and Y. Li, Construction of tubular molecule aggregations of graphdiyne for highly efficient field emission, J. Phys. Chem. C, 2011, 115(6), 2611–2615 CrossRef CAS.
- F. Diederich and Y. Rubin, Synthetic approaches toward molecular and polymeric carbon allotropes, Angew Chem. Int. Ed. Engl., 1992, 31(9), 1101–1123 CrossRef.
- R. Matsuoka, R. Sakamoto, K. Hoshiko, S. Sasaki, H. Masunaga, K. Nagashio and H. Nishihara, Crystalline graphdiyne nanosheets produced at a gas/liquid or liquid/liquid interface, J. Am. Chem. Soc., 2017, 139(8), 3145–3152 CrossRef CAS.
- R. Liu, X. Gao, J. Zhou, H. Xu, Z. Li, S. Zhang, Z. Xie, J. Zhang and Z. Liu, Chemical vapor deposition growth of linked carbon monolayers with acetylenic scaffoldings on silver foil, Adv. Mater., 2017, 29(18), 1604665 CrossRef.
- Z. Zuo, H. Shang, Y. Chen, J. Li, H. Liu, Y. Li and Y. Li, A facile approach for graphdiyne preparation under atmosphere for an advanced battery anode, Chem. Commun., 2017, 53(57), 8074–8077 RSC.
- J. Kuang, J. Yu, Q. Zhang, S. Zhu, R. Wang, J. Ma, J. Wan, H. Han, Z. He, N. Ma and Y. Zhang, Graphdiyne Production in a Flash: High-Yield Direct Synthesis by Electron Beam Irradiation, Adv. Mater., 2025, 2506979 CrossRef CAS PubMed.
- J. Yang, Z. Bi, S. Zhang, L. Zeng, Y. Zhang, Y. Wang, J. Yan, W. Zhao, Y. Dai and J. Yun, Synthesis of γ-graphyne by modified mechanochemistry with enhanced adsorption of organic dyes, Diamond Relat. Mater., 2022, 129, 109336 CrossRef CAS.
- Y. Li, X. Xu, B. Qiang, Y. Li, Y. Lu and C. Li, Turn hazardous endosulfan into S-doped alkynyl carbon material for energy storage and Hg (II) adsorption via a green mechanochemical process, ACS Sustain. Chem. Eng., 2022, 10(28), 9216–9224 CrossRef CAS.
- Y. Lin, H. Liu, C. Yang, X. Wu, C. Du, L. Jiang and Y. Zhong, Gama-graphyne as a photogenerated electrons transfer layer enhances photocatalytic performance of silver phosphate, Appl. Catal., B, 2020, 264, 118479 CrossRef.
- S. Li, M. Sun, X. Huang, H. Chen, J. Zhao and Z. Li, In situ sonochemical synthesis of flower-like N-graphyne/BiOCl0.5 Br 0.5 microspheres for efficient removal of levofloxacin, Dalton Trans., 2024, 53(3), 917–931 RSC.
- B. Gao, M. Sun, W. Ding, Z. Ding and W. Liu, Decoration of γ-graphyne on TiO2 nanotube arrays: Improved photoelectrochemical and photoelectrocatalytic properties, Appl. Catal., B, 2021, 281, 119492 CrossRef CAS.
- J. Kou, X. Zhou, H. Lu, F. Wu and J. Fan, Graphyne as the membrane for water desalination, Nanoscale, 2014, 6(3), 1865–1870 RSC.
- C. Zhu, H. Li, X. C. Zeng, E. G. Wang and S. Meng, Quantized water transport: ideal desalination through graphyne-4 membrane, Sci. Rep., 2013, 3(1), 3163 CrossRef PubMed.
- (a) B. Wu, H. Jin, J. Yin, W. Zhang, X. Tang, P. Zhang and Y. Ding, Water and salt permeability of monolayer graph-n-yne: Molecular dynamics simulations, Carbon, 2017, 123, 688–694 CrossRef CAS; (b) J. Azamat, N. B. Baghbani and H. Erfan-Niya, Atomistic understanding of functionalized γ-graphyne-1 Nanosheet membranes for water desalination, J. Membr. Sci., 2020, 604, 118079 CrossRef CAS.
- R. Liu, J. Zhou, X. Gao, J. Li, Z. Xie, Z. Li, S. Zhang, L. Tong, J. Zhang and Z. Liu, Graphdiyne filter for decontaminating lead-ion-polluted water, Adv. Electron. Mater., 2017, 3(11), 1700122 CrossRef.
- A. H. Mashhadzadeh, A. M. Vahedi, M. Ardjmand and M. G. Ahangari, Investigation of heavy metal atoms adsorption onto graphene and graphdiyne surface: A density functional theory study, Superlattices Microstruct., 2016, 100, 1094–1102 CrossRef CAS.
- J. Zhu, S. Xiang, B. Zhang, J. Wang, C. Li, C. Pan, Y. Xu and Y. Ma, Oxygen-defective graphdiyne for ultra-efficient removal of sulfonylurea herbicides from aqueous solution, J. Environ. Chem. Eng., 2022, 10(3), 107724 CrossRef CAS.
- J. Zhu, D. Liu, C. Li, B. Zhang, J. Wang, W. Wu, J. Ji and Y. Ma, Facile and large-scale synthesis of polymorphic graphdiyne catalyzed by transition metal salts for organic pollutant removal, RSC Adv., 2021, 11(56), 35408–35414 RSC.
- S. Thangavel, K. Krishnamoorthy, V. Krishnaswamy, N. Raju, S. J. Kim and G. Venugopal, Graphdiyne–ZnO nanohybrids as an advanced photocatalytic material, J. Phys. Chem. C, 2015, 119(38), 22057–22065 CrossRef CAS.
- X. Sun, H. Yang, J. Xie, G. Teng, J. He, Z. Zhao and C. Zhang, A photocatalyst combined of copper doped ZnO and graphdiyne (Cu/ZnO@ GDY) for photocatalytic degradation of tetracycline: Mechanism and application, Water Res., 2025, 278, 123345 CrossRef CAS.
- Q. Bai, C. Zhang, Q. Zhu, L. Wang, M. Liu, W. W. Yu, F. Du and N. Sui, Metal-Free Boron Doped Graphdiyne Quantum Dots Loaded Hollow Tubular Carbon Nitride as Novel Photocatalysts for Peroxymonosulphate Mediated Efficient Water Pollutant Degradation, DOI: DOI:10.2139/ssrn.4184112.
- J. Zhang, Q. Bai, X. Bi, C. Zhang, M. Shi, W. W. Yu, F. Du, L. Wang, Z. Wang, Z. Zhu and N. Sui, Piezoelectric enhanced peroxidase-like activity of metal-free sulfur doped graphdiyne nanosheets for efficient water pollutant degradation and bacterial disinfection, Nano Today, 2022, 43, 101429 CrossRef CAS.
- J. Li, Y. Chen, J. Gao, Z. Zuo, Y. Li, H. Liu and Y. Li, Graphdiyne sponge for direct collection of oils from water, ACS Appl. Mater. Interfaces, 2018, 11(3), 2591–2598 CrossRef PubMed.
- X. Gao, J. Zhou, R. Du, Z. Xie, S. Deng, R. Liu, Z. Liu and J. Zhang, Robust superhydrophobic foam: a graphdiyne-based hierarchical architecture for oil/water separation, Adv. Mater., 2016, 28(1), 168–173 CrossRef CAS.
- M. Xue, H. Qiu and W. Guo, Exceptionally fast water desalination at complete salt rejection by pristine graphyne monolayers, Nanotechnology, 2013, 24(50), 505720 CrossRef.
- N. B. Baghbani, J. Azamat, H. Erfan-Niya, S. Majidi and L. Khazini, Molecular insights into water desalination performance of pristine graphdiyne nanosheet membrane, J. Mol. Graphics Modell., 2020, 101, 107729 CrossRef PubMed.
- H. W. Kroto, J. R. Heath, S. C. O'Brien, R. F. Curl and R. E. Smalley, C60: Buckminsterfullerene, Nature, 1985, 318(6042), 162–163 CrossRef CAS.
- M. Paradise and T. Goswami, Carbon nanotubes–production and industrial applications, Mater. Des., 2007, 28(5), 1477–1489 CrossRef CAS.
- W. Kratschmer, L. D. Lamb and D. R. Hoffman, Nature, 1990, 347–354 Search PubMed.
- K. Ariga and L. K. Shrestha, Zero-to-one (or more) nanoarchitectonics: How to produce functional materials from zero-dimensional single-element unit, fullerene, Mater. Adv., 2021, 2(2), 582–597 RSC.
- S. Kulkarni, S. B. Chaudhari, S. S. Chikkamath, R. S. Kurale, T. S. Thopate, S. Praveenkumar, S. Ghotekar, P. Patil and D. Kumar, Potential applications of fullerenes in drug delivery and medical advances, Inorg. Chem. Commun., 2024, 113829 Search PubMed.
- S. Margadonna and K. Prassides, Recent advances in fullerene superconductivity, J. Solid State Chem., 2002, 168(2), 639–652 CrossRef CAS.
- P. I. Kyesmen, A. Onoja and A. N. Amah, Fullerenes synthesis by combined resistive heating and arc discharge techniques, SpringerPlus, 2016, 5, 1–7 CrossRef CAS.
- H. Itagaki, Y. Fujiwara, Y. Minowa, Y. Ikehara, T. Kaneko, T. Okazaki, Y. Iizumi, J. Kim and H. Sakakita, Synthesis of endohedral-fullerenes using laser ablation plasma from solid material and vaporized fullerenes, AIP Adv., 2019, 9(7), 075324 CrossRef.
- G. B. Daniel, K. Nachimuthu and J. L. Nallasivam, Functionalization of [C60] -Fullerene: A Recent Update, Chem.–Asian J., 2024, 202401800 Search PubMed.
- A. Serrano and M. Gallego, Fullerenes as sorbent materials for benzene, toluene, ethylbenzene, and xylene isomers preconcentration, J. Sep. Sci., 2006, 29(1), 33–40 CrossRef CAS PubMed.
- Y. Li and M. Seymour, Fullerenes and carbon nano-onions for environmental application, Nanotechnology for Water Treatment and Purification, Springer International Publishing, 2014, pp. 145–158 Search PubMed.
- K. Guo, N. Li, L. Bao and X. Lu, Fullerenes and derivatives as electrocatalysts: Promises and challenges, Green Energy Environ., 2024, 9(1), 7–27 CrossRef CAS.
- V. K. Gupta and T. A. Saleh, Sorption of pollutants by porous carbon, carbon nanotubes and fullerene-an overview, Environ. Sci. Pollut. Res., 2013, 20, 2828–2843 CrossRef CAS PubMed.
- I. Ion, R. M. Senin, B. Vasile and A. C. Ion, Influence of the matrix of aqueous solutions on the adsorption of endocrine disruptors by fullerene C60, J. Environ. Eng. Landscape Manage., 2019, 27(1), 1–11 CrossRef.
- I. Ion, G. R. Ivan, R. M. Senin, S. M. Doncea, M. C. Capra, O. Oprea, G. Stinga, O. Orbulet and A. C. Ion, Adsorption of triclocarban (TCC) onto fullerene C60 in simulated environmental aqueous conditions, Sep. Sci. Technol., 2019, 54(17), 2759–2772 CrossRef CAS.
- B. Pan, D. Lin, H. Mashayekhi and B. Xing, Adsorption and hysteresis of bisphenol A and 17α-ethinyl estradiol on carbon nanomaterials, Environ. Sci. Technol., 2008, 42(15), 5480–5485 CrossRef CAS PubMed.
- K. I. Katsumata, N. Matsushita and K. Okada, Preparation of TiO2-Fullerene Composites and Their Photocatalytic Activity under Visible Light, Int. J. Photoenergy, 2012, 1, 256096 Search PubMed.
- E. C. Cho, J. H. Ciou, J. H. Zheng, J. Pan, Y. S. Hsiao, K. C. Lee and J. H. Huang, Fullerene C70 Decorated TiO2 nanowires for visible-light-responsive photocatalysts, Appl. Surf. Sci., 2015, 355, 536–546 CrossRef CAS.
- R. M. Vallant, Z. Szabo, S. Bachmann, R. Bakry, M. Najam-ul-Haq, M. Rainer, N. Heigl, C. Petter, C. W. Huck and G. K. Bonn, Development and application ofC60-fullerene bound silica for solid-phase extraction of biomolecules, Anal. Chem., 2007, 79(21), 8144–8153 CrossRef CAS PubMed.
- H. Fu, T. Xu, S. Zhu and Y. Zhu, Photocorrosion inhibition and enhancement of photocatalytic activity for ZnO via hybridization with C60, Environ. Sci. Technol., 2008, 42(21), 8064–8069 CrossRef CAS.
- A. Behera, S. Mansingh, K. Das and K. Parida, Synergistic ZnFe2O4-carbon allotropes nanocomposite photocatalyst for norfloxacin degradation and Cr (VI) reduction, J. Colloid Interface Sci., 2019, 544, 96–111 CrossRef CAS PubMed.
- Q. Cai, Z. Hu, Q. Zhang, B. Li and Z. Shen, Fullerene (C60)/CdS nanocomposite with enhanced photocatalytic activity and stability, Appl. Surf. Sci., 2017, 403, 151–158 CrossRef CAS.
- S. Zhu, T. Xu, H. Fu, J. Zhao and Y. Zhu, Synergetic effect of Bi2WO6 photocatalyst with C60 And enhanced photoactivity under visible irradiation, Environ. Sci. Technol., 2007, 41(17), 6234–6239 CrossRef CAS.
- B. Chai, X. Liao, F. Song and H. Zhou, Fullerene modified C3N4 composites with enhanced photocatalytic activity under visible light irradiation, Dalton Trans., 2014, 43(3), 982–989 RSC.
- X. Bai, L. Wang, Y. Wang, W. Yao and Y. Zhu, Enhanced oxidation ability of g-C3N4 photocatalyst via C60 modification, Appl. Catal., B, 2014, 152, 262–270 CrossRef.
- M. A. Alheety, A. Raoof, S. A. Al-Jibori, A. Karadağ, A. I. Khaleel, H. Akbaş and O. Uzun, Eco-friendly C60-SESMP-Fe3O4 inorganic magnetizable nanocomposite as high-performance adsorbent for magnetic removal of arsenic from crude oil and water samples, Mater. Chem. Phys., 2019, 231, 292–300 CrossRef CAS.
- J. R. Baena, M. Gallego and M. Valcárcel, Speciation of lead in environmental waters by preconcentration on a new fullerene derivative, Anal. Chem., 2002, 74(7), 1519–1524 CrossRef CAS PubMed.
- J. Lee, S. Hong, Y. Mackeyev, C. Lee, E. Chung, L. J. Wilson, J. H. Kim and P. J. Alvarez, Photosensitized oxidation of emerging organic pollutants by tetrakis C60 amino Fullerene-derivatized silica under visible light irradiation, Environ. Sci. Technol., 2011, 45(24), 10598–10604 CrossRef CAS PubMed.
- J. Kyriakopoulos, A. T. Papastavrou, G. D. Panagiotou, M. D. Tzirakis, K. S. Triantafyllidis, M. N. Alberti, K. Bourikas, C. Kordalis, M. Orfanopoulos and A. Lycourghiotis, Deposition of fullerene C60 on the surface of MCM-41 via the one-step wet impregnation method: Active catalysts for the singlet oxygen mediated photooxidation of alkenes, J. Mol. Catal. A: Chem., 2014, 381, 9–15 CrossRef CAS.
- O. V. Alekseeva, N. A. Bagrovskaya and A. V. Noskov, Study of Hybrid Polystyrene Materials with Incorporated Fullerene Nanoparticles, Proceedings of the International Conference Nanomaterials Applications and Properties, 2014, vol. 3 (2), p. 02NNSA04(5pp) Search PubMed.
- V. I. Berezkin, I. V. Viktorovskiı, L. V. Golubev, V. N. Petrova and L. O. A. Khoroshko, Comparative Study of the Sorption Capacity of Activated Charcoal, Soot, and Fullerenes for Organochlorine compounds, Tech. Phys. Lett., 2002, 28(11), 885–888 CrossRef CAS.
- X. Cheng, A. T. Kan and M. B. Tomson, Uptake and sequestration of naphthalene and 1, 2-dichlorobenzene by C60, J. Nanopart. Res., 2005, 7, 555–567 CrossRef CAS.
- M. Gallego, Y. Petit de Pena and M. Valcarcel, Fullerenes as sorbent materials for metal preconcentration. Analytical Chemistry 66(22):4074-078. B) Bui TX and Choi H (2009) Nanoscale carbon materials for contaminant separation, in Nanotechnologies for Water Environment Applications, 1994, pp. 269–311 Search PubMed.
- Adsorption by Carbons, ed. Bottani E. J. and Tascón J. M., Elsevier, 2011 Search PubMed.
- K. Kaneko, C. Ishii, T. Arai and H. Suematsu, Defect-associated microporous nature of C 60 crystals, J. Phys. Chem., 1993, 97(26), 6764–6766 CrossRef CAS.
- O. V. Alekseeva, N. A. Bagrovskaya and A. V. Noskov, Sorption of heavy metal ions by fullerene and polystyrene/fullerene film compositions, Prot. Met. Phys. Chem., 2016, 52, 443–447 CAS.
- X. Cheng, A. T. Kan and M. B. Tomson, Naphthalene adsorption and desorption from aqueous C60 fullerene, J. Chem. Eng. Data, 2004, 49(3), 675–683 CrossRef CAS.
- A. E. Burakov, E. V. Galunin, I. V. Burakova, A. E. Kucherova, S. Agarwal, A. G. Tkachev and V. K. Gupta, Adsorption of heavy metals on conventional and nanostructured materials for wastewater treatment purposes: A review, Ecotoxicol. Environ. Saf., 2018, 148, 702–712 CrossRef CAS PubMed.
- S. R. Yashas, B. Shahmoradi, K. Wantala and H. P. Shivaraju, Potentiality of polymer nanocomposites for sustainable environmental applications: A review of recent advances, Polymer, 2021, 233, 124184 CrossRef CAS.
- T. V. Plisko, A. S. Liubimova, A. V. Bildyukevich, A. V. Penkova, M. E. Dmitrenko, V. Y. Mikhailovskii, G. B. Melnikova, K. N. Semenov, N. V. Doroshkevich and A. I. Kuzminova, Fabrication and characterization of polyamide-fullerenol thin film nanocomposite hollow fiber membranes with enhanced antifouling performance, J. Membr. Sci., 2018, 551, 20–36 CrossRef CAS.
- M. E. Dmitrenko, A. V. Penkova, A. I. Kuzminova, R. R. Atta, A. A. Zolotarev, A. S. Mazur, O. S. Vezo, E. Lahdenranta, D. A. Markelov and S. S. Ermakov, Development and investigation of novel polyphenylene isophthalamide pervaporation membranes modified with various fullerene derivatives, Sep. Purif. Technol., 2019, 226, 241–251 CrossRef CAS.
- A. Kausar, I. Ahmad, M. Maaza and M. H. Eisa, State-of-the-art of polymer/fullerene C60 nanocomposite membranes for water treatment: conceptions, structural diversity and topographies, Membranes, 2022, 13(1), 27 CrossRef PubMed.
- Y. Liu, B. Phillips, W. Li, Z. Zhang, L. Fang, J. Qiu and S. Wang, Fullerene-tailored graphene oxide interlayer spacing for energy-efficient water desalination, ACS Appl. Nano Mater., 2018,(11), 6168–6175 CrossRef CAS.
- E. Ciotta, P. Prosposito, D. Moscone, N. Colozza and R. Pizzoferrato, Detection and removal of heavy-metal ions in water by unfolded-fullerene nanoparticles, in AIP Conference Proceedings, AIP Publishing, 2019, vol. 2145, p. 1 Search PubMed.
- R. E. Letts, T. C. Pereira, M. R. Bogo and J. M. Montserrat, Biologic responses of bacteria communities living at the mucus secretion of common carp (Cyprinus carpio) after exposure to the carbon nanomaterial fullerene (C60), Arch. Environ. Contam. Toxicol., 2011, 61, 311–317 CrossRef CAS.
- Z. Markovic, B. Todorovic-Markovic, D. Kleut, N. Nikolic, S. Vranjes-Djuric, M. Misirkic, L. Vucicevic, K. Janjetovic, A. Isakovic, L. Harhaji and B. Babic-Stojic, The mechanism of cell-damaging reactive oxygen generation by colloidal fullerenes, Biomaterials, 2007, 28(36), 5437–5448 CrossRef CAS PubMed.
- D. Y. Lyon and P. J. Alvarez, Fullerene water suspension (nC60) exerts antibacterial effects via ROS-independent protein oxidation, Environ. Sci. Technol., 2008, 42(21), 8127–8132 CrossRef CAS.
- F. Huang, L. Ge, B. Zhang, Y. Wang, H. Tian, L. Zhao, Y. He and X. Zhang, A fullerene colloidal suspension stimulates the growth and denitrification ability of wastewater treatment sludge-derived bacteria, Chemosphere, 2014, 108, 411–417 CrossRef CAS PubMed.
- Y. Song, S. Zhu and B. Yang, Bioimaging based on fluorescent carbon dots, RSC Adv., 2014, 4(52), 27184–27200 RSC.
- F. Yuan, S. Li, Z. Fan, X. Meng, L. Fan and S. Yang, Shining carbon dots: Synthesis and Biomedical and optoelectronic applications, Nano Today, 2016, 11(5), 565–586 CrossRef CAS.
- (a) H. L. Yang, L. F. Bai, Z. R. Geng, H. Chen, L. T. Xu, Y. C. Xie, D. J. Wang, H. W. Gu and X. M. Wang, Carbon quantum dots: Preparation, optical properties, and biomedical applications, Mater. Today Adv., 2023, 18, 100376 CrossRef CAS; (b) C. Xia, S. Zhu, T. Feng, M. Yang and B. Yang, Evolution and synthesis of carbon dots: from carbon dots to carbonized polymer dots, Adv. Sci., 2019, 6(23), 1901316 CrossRef CAS.
- S. Sharma, A. Umar, S. K. Mehta, A. O. Ibhadon and S. K. Kansal, Solar light driven photocatalytic degradation of levofloxacin using TiO 2/carbon-dot nanocomposites, New J. Chem., 2018, 42(9), 7445–7456 RSC.
- (a) H. Ding, S. B. Yu, J. S. Wei and H. M. Xiong, Full-color light-emitting carbon dots with a surface-state-controlled luminescence mechanism, ACS Nano, 2016, 10(1), 484–491 CrossRef CAS PubMed; (b) D. Pan, J. Zhang, Z. Li and M. Wu, Hydrothermal route for cutting graphene sheets into blue-luminescent graphene quantum dots, Adv. Mater., 2010, 22(6), 734–738 CrossRef CAS.
- J. Shen, Y. Zhu, C. Chen, X. Yang and C. Li, Facile preparation and upconversion luminescence of graphene quantum dots, Chem. Commun., 2011, 47(9), 2580–2582 RSC.
- P. Wang, J. H. Liu, H. Gao, Y. Hu, X. Hou, G. E. LeCroy, C. E. Bunker, Y. Liu and Y. P. Sun, Host–guest Carbon dots as high-performance fluorescence probes, J. Mater. Chem. C, 2017,(25), 6328–6335 RSC.
- T. V. de Medeiros, J. Manioudakis, F. Noun, J. R. Macairan, F. Victoria and R. Naccache, Microwave-assisted synthesis of carbon dots and their applications, J. Mater. Chem. C, 2019, 7(24), 7175–7195 RSC.
- A. S. Rasal, S. Yadav, A. Yadav, A. A. Kashale, S. T. Manjunatha, A. Altaee and J. Y. Chang, Carbon quantum dots for energy applications: a review, ACS Appl. Nano Mater., 2021, 4(7), 6515–6541 CrossRef CAS.
- S. Sen and A. Bose, Carbon dots: A review of innovations, applications, challenges, and future prospects, Inorg. Chem. Commun., 2025, 113852 CrossRef CAS.
- https://www.nobelprize.org/prizes/chemistry/2023/bawendi/facts/ .
- H. A. S. Tohamy, M. El-Sakhawy and S. Kamel, Eco-friendly synthesis of carbon quantum dots as An effective adsorbent, J. Fluoresc., 2023, 33(2), 423–435 CrossRef CAS.
- K. Jlassi, K. Eid, M. H. Saliem, A. M. Abdullah, M. M. Chehimi and I. Krupa, Rational synthesis, characterization, and application of environmentally friendly (polymer–carbon dot) hybrid composite film for fast and efficient UV-assisted Cd 2+ removal from water, Environ. Sci. Eur., 2020, 32, 1–3 CrossRef.
- M. Sabet and K. Mahdavi, Green synthesis of high photoluminescence nitrogen-doped carbon quantum dots from grass via a simple hydrothermal method for removing organic and inorganic water pollution, Appl. Surf. Sci., 2019, 463, 283–291 CrossRef CAS.
- M. Preethi, C. Viswanathan and N. Ponpandian, A metal-free, dual catalyst for the removal of Rhodamine B using novel carbon quantum dots from muskmelon peel under sunlight and ultrasonication:A green way to clean the environment, J. Photochem. Photobiol., A, 2022, 426, 113765 CrossRef CAS.
- N. U. Nizam, M. M. Hanafiah, E. Mahmoudi and A. W. Mohammad, Synthesis of highly fluorescent carbon quantum dots from rubber seed shells for the adsorption and photocatalytic degradation of dyes, Sci. Rep., 2023, 13(1), 12777 CrossRef CAS.
- S. Hijazi, A. S. Ganie, M. M. Rahman and W. A. Shah, Facile synthesis of surface-functionalized fluorescent carbon quantum dots for the selective detection of ferric ions, Environ. Sci.: Nano, 2023, 10(12), 3281–3294 RSC.
- M. Mashkani, A. Mehdinia, A. Jabbari, Y. Bide and M. R. Nabid, Preconcentration and extraction of lead ions in vegetable and water samples by N-doped carbon quantum dots conjugated with Fe3O4 as a green and facial adsorbent, Food Chem., 2018, 239, 1019–1026 CrossRef CAS PubMed.
- F. F. Hasan, N. A. W. Mohd Zahidi, V. S. Bahar, U. R. R. P. Remli and A. Abd Aziz, Carbon quantum dots (CQDs) based composites photocatalyst for wastewater purification, in Materials Science Forum Trans, Tech Publications Ltd, 2022, vol. 1056, pp. 111–117 Search PubMed.
- D. Hazarika, D. Saikia, K. Gupta, M. Mandal and N. Karak, Photoluminescence, self-cleaning and photocatalytic behavior of waterborne hyperbranched polyester/carbon dot@ TiO2 nanocomposite, ChemistrySelect, 2018, 3(22), 6126–6135 CrossRef CAS.
- H. Koulivand, A. Shahbazi, V. Vatanpour and M. Rahmandoost, Development of carbon dot-modified polyethersulfone membranes for enhancement of nanofiltration, permeation and antifouling performance, Sep. Purif. Technol., 2020, 230, 115895 CrossRef.
- Y. Wang and A. Hu, Carbon quantum dots: synthesis, properties and applications, J. Mater. Chem. C, 2014, 2(34), 6921–6939 RSC.
- M. Sabet and K. Mahdavi, Green synthesis of high photoluminescence nitrogen-doped carbon quantum dots from grass via a simple hydrothermal method for removing organic and inorganic water pollutants, Appl. Surf. Sci., 2019, 463, 283–291 CrossRef CAS.
- S. Huang, S. Jiang, H. Pang, T. Wen, A. M. Asiri, K. A. Alamry, A. Alsaedi, X. Wang and S. Wang, Dual functional nanocomposites of magnetic MnFe2O4 and fluorescent carbon dots for efficient U (VI) removal, Chem. Eng. J., 2019, 368, 941–950 CrossRef CAS.
- A. Tadesse, D. RamaDevi, M. Hagos, G. Battu and K. Basavaiah, Synthesis of nitrogen doped Carbon quantum dots/magnetite nanocomposites for efficient removal of methyl blue dye pollutants from contaminated water, RSC Adv., 2018, 8(16), 8528–8536 RSC.
- D. Yang, S. K. Tammina, X. Li and Y. Yang, Enhanced removal and detection of benzo[α]pyrene in environmental water samples using carbon dots-modified magnetic nanocomposites, Ecotoxicol. Environ. Saf., 2019, 170, 383–390 CrossRef CAS.
- Z. Mou, H. Zhang, Z. Liu, J. Sun and M. Zhu, Ultrathin BiOCl/nitrogen-doped graphene quantum dots composites with strong adsorption and effective photocatalytic activity for the degradation of antibiotic ciprofloxacin, Appl. Surf. Sci., 2019, 496, 143655 CrossRef CAS.
- O. Rahmanian, M. Dinari and M. K. Abdolmaleki, Carbon quantum dots/layered double hydroxide hybrid for fast and efficient decontamination of Cd (II): The adsorption kinetics and isotherms, Appl. Surf. Sci., 2018, 428, 272–279 CrossRef CAS.
- Y. Yao, H. Zhang, K. Hu, G. Nie, Y. Yang, Y. Wang, X. Duan and S. Wang, Carbon dots based photocatalysis for environmental applications, J. Environ. Chem. Eng., 2022, 10(2), 107336 CrossRef CAS.
- U. V. Agbogo, B. S. Rifore, C. T. Arum, P. D. Lorver, J. Mathew and S. A. Tanko, Carbon quantum dots for wastewater treatment: present progress and future prospects, FUDMA J. Sci., 2024, 8(1), 93–102 CrossRef.
- S. Jabeen, A. Kausar, B. Muhammad, S. Gul and M. Farooq, A review on polymeric nanocomposites of nanodiamond, carbon nanotube, and nanobifiller: structure, preparation and properties, Polym.-Plast. Technol. Eng., 2015, 54(13), 1379–1409 CrossRef CAS.
- N. R. Greiner, D. S. Phillips, J. D. Johnson and F. Volk, Diamonds in detonation soot, Nature, 1988, 333(6172), 440–442 CrossRef CAS.
- V. V. Danilenko, On the history of the discovery of nanodiamond synthesis, Phys. Solid State, 2004, 46, 595–599 CrossRef CAS.
- P. Aprà, L. Mino, A. Battiato, P. Olivero, S. Sturari, M. C. Valsania, V. Varzi and F. Picollo, Interaction of nanodiamonds with water: impact of surface chemistry on hydrophilicity, aggregation and electrical properties, Nanomaterials, 2021, 11(10), 2740 CrossRef PubMed.
- S. E. Elugoke, O. E. Fayemi, A. S. Adekunle, T. T. Nkambule, B. B. Mamba and E. E. Ebenso, Conductive nanodiamond-based detection of neurotransmitters: one decade, few sensors, ACS Omega, 2021, 6(29), 18548–18558 CrossRef CAS PubMed.
- M. Shellaiah and K. W. Sun, Conjugation of cysteamine functionalized nanodiamond to gold nanoparticles for pH enhanced colorimetric detection of Cr3+ ions demonstrated by real water sample analysis, Spectrochim. Acta, Part A, 2023, 286, 121962 CrossRef CAS PubMed.
- E. Yilmaz, H. Ulusoy, Ö. Demir and M. Soylak, A new magnetic nanodiamond/graphene oxide Hybrid (Fe3O4@ ND@ GO) material for pre-concentration and sensitive determination of sildenafil in alleged herbal aphrodisiacs by HPLC-DAD system, J. Chromatogr., 2018, B1084, 113–121 Search PubMed.
- H. Molavi, K. Mirzaei, E. Jafarpour, A. Mohammadi, M. S. Salimi, M. Rezakazemi, M. M. Nadagouda and T. M. Aminabhavi, Wastewater treatment using nanodiamond and related materials, J. Environ. Manage., 2024, 349, 119349 CrossRef CAS PubMed.
- V. Mochalin, O. Shenderova, D. Ho and Y. Gogotsi, The properties and applications of nanodiamonds, Nano-Enabled Med. Appl., 2020, 313–350 Search PubMed.
- P. Karami, B. Khorshidi, L. Shamaei, E. Beaulieu, J. B. Soares and M. Sadrzadeh, Nanodiamond-enabled thin-film nanocomposite polyamide membranes for high-temperature water treatment, ACS Appl. Mater. Interfaces, 2020, 12(47), 53274–53285 CrossRef CAS.
- A. Colon, J. Avalos, B. R. Weiner, G. Morell and R. Ríos, Water treatment membranes embedded with a stable and bactericidal nanodiamond material, J. Water Health, 2023, 21(5), 601–614 CrossRef PubMed.
- M. Zamani, M. Aghajanzadeh, H. Molavi, H. Danafar and A. Shojaei, Thermally oxidized nanodiamond: An effective sorbent for separation of methotrexate from aqueous media: synthesis, characterization, In vivo and in vitro biocompatibility study, J. Inorg. Organomet. Polym. Mater., 2019, 29, 701–709 CrossRef CAS.
- V. Vatanpour, R. S. E. Naeeni, A. Ghadimi, A. Karami and B. Sadatnia, Effect of detonation nanodiamond on the properties and performance of polyethersulfone nanocomposite membrane, Diamond Relat. Mater., 2018, 90, 44–255 CrossRef.
- Y. He, H. Lin, Z. Guo, W. Zhang, H. Li and W. Huang, Recent developments and advances in boron-doped diamond electrodes for electrochemical oxidation of organic pollutants, Sep. Purif. Technol., 2019, 212, 802–821 CrossRef CAS.
- X. Zhu, S. Shi, J. Wei, F. Lv, H. Zhao, J. Kong, Q. He and J. Ni, Electrochemical oxidation characteristics of p-substituted phenols using a boron-doped diamond electrode, Environ. Sci. Technol., 2007, 41(18), 6541–6546 CrossRef CAS.
- Y. M. Hunge, A. A. Yadav, S. Khan, K. Takagi, N. Suzuki, K. Teshima, C. Terashima and A. Fujishima, Photocatalytic degradation of bisphenol A using titanium dioxide@nanodiamond composites under UV light illumination, J. Colloid Interface Sci., 2021, 582, 1058–1066 CrossRef CAS.
- A. Siddiqa, A. Majid, F. Saira, S. Farooq, R. Qureshi and S. Qaisar, Nanodiamond embedded polyaniline/polyvinylidene fluoride nanocomposites as microfiltration membranes for removal of industrial pollution, RSC Adv., 2023, 13(42), 29206–29214 RSC.
- H. Chen, J. Shen and Y. Zhang, Molybdenum disulfide/nanodiamonds hybrid for high electromagnetic absorption, Diamond Relat. Mater., 2021, 118, 108535 CrossRef CAS.
- S. Ozdemir, E. Kilinc, K. S. Celik, V. Okumus and M. Soylak, Simultaneous pre concentrations of Co2+, Cr6+, Hg2+ and Pb2+ ions by Bacillus altitudinis immobilized nanodiamond prior to their determinations in food samples by ICP-OES, Food Chem., 2017, 215, 447–453 CrossRef CAS PubMed.
- S. Ozdemir, E. Kılınç, F. Şen and M. Soylak, Development of Armillae mellea immobilized nanodiamond the pre concentrations of Cr (III), Hg (II) and Zn (II), Anal. Biochem., 2021, 17, 114122 CrossRef.
- E. V. Perevedentseva, A. Karmenyan and C. L. Cheng, Spectroscopy of Nanodiamond Surface: Investigation and Applications. Novel Aspects of Diamond: from Growth to Applications, 2019, pp. 363–413 Search PubMed.
- R. Bogdanowicz, Functionalized nanodiamonds as a perspective green carbo-catalyst for removal of emerging organic pollutants, Curr. Opin. Solid State Mater. Sci., 2022, 26(3), 100991 CrossRef CAS.
- D. Khanal, Q. Lei, G. Pinget, D. A. Cheong, A. Gautam, R. Yusoff, B. Su, S. Yamaguchi, A. Kondyurin, J. C. Knowles and G. Georgiou, The protein corona determines the cytotoxicity of nanodiamonds: implications of corona formation and its remodelling on nanodiamond applications in biomedical imaging and drug delivery, Nanoscale Adv., 2020, 2(10), 4798–4812 RSC.
- Y. Lin, Z. Feng, L. Yu, Q. Gu, S. Wu and D. S. Su, Insights into the surface chemistry and electronic properties of sp2 and sp3-hybridized nanocarbon materials for catalysis, Chem. Commun., 2017, 53(35), 4834–4837 RSC.
- H. Molavi, A. Shojaei and A. Pourghaderi, Rapid and tunable selective adsorption of dyes using thermally oxidized nanodiamond, J. Colloid Interface Sci., 2018, 524, 52–64 CrossRef CAS.
- F. Saberi-Movahed and D. W. Brenner, What drives adsorption of ions on the surface of nanodiamonds in aqueous solutions?, arXiv, 2021, preprint, arXiv:2102.09187, DOI:10.48550/arXiv.2102.09187.
- E. B. Yudina, A. E. Aleksenskii, I. G. Fomina, A. V. Shvidchenko, D. P. Danilovich, I. L. Eremenko and A. Y. Vul, Interaction of carboxyl groups with rare metal ions on the surface of detonation nanodiamonds, Eur. J. Inorg. Chem., 2019, 39–40, 4345–4349 CrossRef.
- Y. Buchatskaya, A. Romanchuk, R. Yakovlev, A. Shiryaev, I. Kulakova and S. Kalmykov, Sorption of actinides onto nanodiamonds, Radiochim. Acta, 2015, 103(3), 205–211 CrossRef CAS.
- G. Speranza, Carbon nanomaterials: Synthesis, functionalization and sensing applications, Nanomaterials, 2021, 11(4), 967 CrossRef CAS PubMed.
- E. Duffy, X. He, P. N. Nesterenko and B. Paull, Hierarchical porous graphitic carbon monoliths with detonation nanodiamonds: synthesis, characterisation and adsorptive properties, J. Mater. Sci., 2015, 50, 6245–6259 CrossRef CAS.
- D. S. Volkov, P. K. Krivoshein, I. V. Mikheev and M. A. Proskurnin, Pristine detonation nanodiamonds as regenerable adsorbents for metal cations, Diamond Relat. Mater., 2020, 108121 CrossRef CAS.
- K. Matsumoto, H. Yamato, S. Kakimoto, T. Yamashita, R. Wada, Y. Tanaka, M. Akita and T. Fujimura, A highly efficient adsorbent Cu-Prussian Blue@ nanodiamond for cesium in diluted artificial Seawater and soil-treated wastewater, Sci. Rep., 2018, 8(1), 5807 CrossRef PubMed.
- X. Zhao, S. Zhang, C. Bai, B. Li, Y. Li, L. Wang, R. Wen, M. Zhang, L. Ma and S. Li, Nano-diamond particles functionalized with single/double-arm amide–thiourea ligands for adsorption of metal ions, J. Colloid Interface Sci., 2016, 469, 109–119 CrossRef CAS.
- M. S. Gutiérrez, K. A. López, J. Morey and M. N. Piña, Introducing Selectivity on Carbonaceous Material: Removing Noble Salts, Au3+, and Ag+ from Aqueous Media by Nanodiamonds Functionalized with Squaramides, Materials, 2020, 13(5), 1086 CrossRef.
- Z. He, Q. Huang, L. Mao, H. Huang, M. Liu, J. Chen, F. Deng, N. Zhou, X. Zhang and Y. Wei, Direct surface modification of nanodiamonds with ionic copolymers for fast adsorptive removal of copper ions with high efficiency, Colloid Interface Sci. Commun., 2020, 37, 100278 CrossRef CAS.
- G. Yang, H. Huang, J. Chen, D. Gan, F. Deng, Q. Huang, Y. Wen, M. Liu, X. Zhang and Y. Wei, Preparation of ionic liquids functionalized nanodiamonds-based composites through the Michael addition reaction for efficient removal of environmental pollutants, J. Mol. Liq., 2019, 296, 111874 CrossRef CAS.
- W. Wu, F. Gao, J. Qu, Y. Zang, M. Fan and K. Hua, Self-Assembly of Graphene Oxide/Nanodiamond Microspheres with High Adsorption for Pb (II) Ions, ChemistrySelect, 2022, 7(10), 202104436 CrossRef.
- M. Neshastehgar, P. Rahmani, A. Shojaei and H. Molavi, Enhanced adsorption removal performance of UiO-66 by rational hybridization with nanodiamond, Microporous Mesoporous Mater., 2020, 296, 110008 CrossRef CAS.
- M. Raeiszadeh, A. Hakimian, A. Shojaei and H. Molavi, Nanodiamond-filled chitosan as an efficient adsorbent for anionic dye removal from aqueous solutions, J. Environ. Chem. Eng., 2018, 6(2), 3283–3294 CrossRef CAS.
- A. B. Çiğil, O. A. Urucu and M. V. Kahraman, Nanodiamond-containing polyethyleneimine hybrid materials for lead adsorption from aqueous media, J. Appl. Polym. Sci., 2019, 36(46), 48241 CrossRef.
- F. Ahmadijokani, H. Molavi, A. Bahi, S. Wuttke, M. Kamkar, O. J. Rojas, F. Ko and M. Arjmand, Electrospun nanofibers of chitosan/polyvinyl alcohol/UiO-66/nanodiamond: Versatile adsorbents for wastewater remediation and organic dye removal, Chem. Eng. J., 2023, 457, 141176 CrossRef CAS.
- H. Wang, Q. Zhao, K. Zhang, F. Wang, J. Zhi and C. X. Shan, Superhydrophobic nanodiamond-functionalized melamine sponge for oil/water separation, Langmuir, 2022, 38(37), 11304–11313 CrossRef CAS PubMed.
- P. Shao, Y. Jing, X. Duan, H. Lin, L. Yang, W. Ren, F. Deng, B. Li, X. Luo and S. Wang, Revisiting the graphitized nanodiamond-mediated activation of peroxymonosulfate: singlet oxygenation versus electron transfer, Environ. Sci. Technol., 2021, 55(23), 16078–16087 CrossRef CAS PubMed.
- F. Bernat-Quesada, C. Vallés-García, E. Montero-Lanzuela, A. López-Francés, B. Ferrer, H. G. Baldoví and S. Navalón, Hybrid sp2/sp3 nanodiamonds as heterogeneous metal-free ozonation catalysts in water, Appl. Catal., B, 2021, 299, 120673 CrossRef CAS.
- L. X. Su, Y. Cao, H. S. Hao, Q. Zhao and J. Zhi, Emerging applications of nanodiamonds in photocatalysis, Funct. Diamond, 2022,(1), 93–109 Search PubMed.
- M. Khan, A. Hayat, S. K. Mane, T. Li, N. Shaishta, D. Alei, T. K. Zhao, A. Ullah, A. Zada, A. Rehman and W. U. Khan, Functionalized nano diamond composites for photocatalytic hydrogen evolution and effective pollutant degradation, Int. J. Hydrogen Energy, 2020, 45(53), 29070–29081 CrossRef CAS.
- B. Liang, L. Zhang, W. Wang, G. Xu, W. Zhang, Y. Zhang, R. Zhang, L. Yang and L. Zhang, Nanodiamond core/onion-like carbon shell materials with excellent visible light photocatalytic activity, Mater. Res. Express, 2019, 6(4), 045609 CrossRef.
- X. Sun, C. Wang, L. Liu, Q. Wang, S. Cheng, N. Gao, J. Liu and H. Li, Enhanced solar-light-driven photocatalysis by carbon-nanodots/nanodiamonds heterojunction, Desalin. Water Treat., 2020, 206, 439–446 CrossRef CAS.
- H. Lee, J. S. Lee, K. J. Moor, H. I. Kim, S. R. Kim, G. Gim, J. Lee, H. H. Kim, T. M. Fahmy, J. H. Kim and C. Lee, Hand-ground fullerene-nanodiamond composite for photosensitized water treatment and photodynamic cancer therapy, J. Colloid Interface Sci., 2021, 587, 101–109 CrossRef CAS PubMed.
- L. Zhou, H. Zhang, X. Guo, H. Sun, S. Liu, M. O. Tade and S. Wang, Metal-free hybrids of graphitic carbon nitride and nanodiamonds for photoelectrochemical and photocatalytic applications, J. Colloid Interface Sci., 2017, 493, 275–280 CrossRef CAS PubMed.
- Z. Wang, F. Zhang, A. Ning, D. Lv, G. Jiang and A. Song, Nanosilver supported on inert nanodiamond as a direct plasmonic photocatalyst for degradation of methyl blue, J. Environ. Chem. Eng., 2021, 9(1), 104912 CrossRef CAS.
- L. X. Su, Q. Lou, C. X. Shan, D. L. Chen, J. H. Zang and L. J. Liu, Ag/Nanodiamond/g-C3N4 heterostructures with enhanced visible-light photocatalytic performance, Appl. Surf. Sci., 2020, 525, 146576 CrossRef CAS.
- J. Pan, H. Sun, K. Chen, Y. Zhang, P. Shan, W. Shi and F. Guo, Nanodiamonds decorated yolk-shell ZnFe2O4 sphere as magnetically separable and recyclable composite for boosting antibiotic degradation performance, Chin. J. Chem. Eng., 2023, 54, 162–172 CrossRef CAS.
- D. Huang, G. Zhang, J. Yi, M. Cheng, C. Lai, P. Xu, C. Zhang, Y. Liu, C. Zhou, W. Xue and R. Wang, Progress and challenges of metal-organic frameworks-based materials for SR-AOPs applications in water treatment, Chemosphere, 2021, 263, 127672 CrossRef CAS PubMed.
- J. Pan, F. Guo, H. Sun, M. Li, X. Zhu, L. Gao and W. Shi, Nanodiamond decorated 2D hexagonal Fe2O3 nanosheets with a Z-scheme photogenerated electron transfer path for enhanced photocatalytic activity, J. Mater. Sci., 2021, 56, 6663–6675 CrossRef CAS.
- V. Vatanpour, E. Salehi, N. Sahebjamee and M. Ashrafi, Novel chitosan/polyvinyl alcohol thin membrane adsorbents modified with detonation nanodiamonds: preparation, characterization, and adsorption performance, Arabian J. Chem., 2020, 13(1), 1731–1740 CrossRef CAS.
- S. Khakpour, Y. Jafarzadeh and R. Yegani, Incorporation of graphene oxide/nanodiamond nanocomposite into PVC ultrafiltration membranes, Chem. Eng. Res. Des., 2019, 152, 60–70 CrossRef CAS.
- H. I. Kim, H. N. Kim, S. Weon, G. H. Moon, J. H. Kim and W. Choi, Robust co-catalytic performance of nanodiamonds loaded on WO3 for the decomposition of volatile organic compounds under visible light, ACS Catal., 2016, 6(12), 8350–8360 CrossRef CAS.
- A. Siddiqa, A. Majid, F. Saira, S. Farooq, R. Qureshi and S. Qaisar, Nanodiamond embedded polyaniline/polyvinylidene fluoride nanocomposites as microfiltration membranes for removal of industrial pollution, RSC Adv., 2023, 13(42), 29206–29214 RSC.
- H. A. Bland, I. A. Centeleghe, S. Mandal, E. L. Thomas, J. Y. Maillard and O. A. Williams, Electropositive nanodiamond-coated quartz microfiber membranes for virus and dye filtration, ACS Appl. Nano Mater., 2021, 4(3), 3252–3261 CrossRef CAS.
- I. Pet, M. N. Sanad, M. Farouz, M. M. ElFaham, A. El-Hussein, M. A. El-Sadek, R. A. Althobiti and A. Ioanid, Recent Developments in the Implementation of Activated Carbon as Heavy Metal Removal Management, Water Conserv. Sci. Eng., 2024, 9(2), 62 CrossRef.
- L. Nielsen, M. J. Biggs, W. Skinner and T. J. Bandosz, The effects of activated carbon surface features on the reactive adsorption of carbamazepine and sulfamethoxazole, Carbon, 2014, 80, 419–432 CrossRef CAS.
- D. S. Senewirathna, S. Thuraisingam, S. Prabagar and J. Prabagar, Fluoride removal in drinking water using activated carbon prepared from palmyrah (Borassus flabellifer) nut shells, Curr. Res. Green Sustainable Chem., 2022, 5, 100304 CrossRef CAS.
- N. O. Ogbodo, C. O. Asadu, C. A. Ezema, M. I. Onoh, O. C. Elijah, I. S. Ike and O. E. Onoghwarite, Preparation and Characterization of activated carbon from agricultural waste (Musa-paradisiaca peels) for the remediation of crude oil contaminated water, J. Hazard. Mater. Adv., 2021, 2, 100010 CAS.
- S. Husien, R. M. El-taweel, A. I. Salim, I. S. Fahim, L. A. Said and A. G. Radwan, Review of activated carbon adsorbent material for textile dyes removal: Preparation, and modelling, Curr. Res. Green Sustainable Chem., 2022, 5, 100325 CrossRef CAS.
- P. K. Malik, Dye removal from wastewater using activated carbon developed from sawdust: adsorption equilibrium and kinetics, J. Hazard. Mater., 2004, 113(1–3), 81–88 CrossRef CAS PubMed.
- M. Lewoyehu, Comprehensive review on synthesis and application of activated carbon from agricultural residues for the remediation of venomous pollutants in wastewater, J. Anal. Appl. Pyrolysis, 2021, 159, 105279 CrossRef CAS.
- X. Zhang, W. Guo, H. H. Ngo, H. Wen, N. Li and W. Wu, Performance evaluation of powdered activated carbon for removing 28 types of antibiotics from water, J. Environ. Manage., 2016, 172, 193–200 CrossRef CAS.
- A. Cruz-Cruz, A. Rivas-Sanchez, G. Gallareta-Olivares, R. B. González-González, M. F. Cárdenas-Alcaide, H. M. Iqbal and R. Parra-Saldívar, Carbon-based materials: adsorptive removal of antibiotics from water, Water Emerging Contam. Nanoplast., 2023, 2(1), N–A Search PubMed.
- A. Ramirez, R. Ocampo, S. Giraldo, E. Padilla, E. Flórez and N. Acelas, Removal of Cr (VI) from an aqueous solution using an activated carbon obtained from teakwood sawdust: Kinetics, equilibrium, and density functional theory calculations, J. Environ. Chem. Eng., 2020, 8(2), 103702 CrossRef CAS.
- R. Ganjoo, S. Sharma, A. Kumar and M. M. A. Daouda, in Activated Carbon Progress and Applications, ed. C. Verma and M. A. Quraishi, The Royal Society of Chemistry, 2023, ch. 1, pp. 1–22 Search PubMed.
- S. P. Kimothi, S. Panwar and A. Khulbe, Creating wealth from agricultural waste, Indian council of agricultural research, New Delhi 0-172, 2020.
- T. Heinze, Cellulose: Structure and Properties, Advances in Polymer Science, Springer, International Publishing, 2015, pp. 1–52 Search PubMed.
- B. Joseph, V. K. Sagarika, C. Sabu, N. Kalarikkal and S. Thomas, Cellulose nanocomposites: Fabrication and biomedical applications, J. Bioresour. Bioprod., 2020, 5(4), 223–237 CAS.
- P. R. Sharma, S. K. Sharma, T. Lindström and B. Hsiao, Nanocellulose-enabled membranes for water purification: Perspectives, Adv. Sustainable Syst., 2020, 4(5), 1900114 CrossRef CAS.
- A. Bhardwaj, M. Bansal, K. Wilson, S. Gupta and M. Dhanawat, Lignocellulose biosorbents: Unlocking the potential for sustainable environmental cleanup, Int. J. Biol. Macromol., 2025, 294, 139497 CrossRef CAS.
- A. Hamissa, A. Lodi, M. Seffen, E. Finocchio, R. Botter and A. Converti, Sorption of Cd (II) and Pb (II) from aq sol. onto Agave americana fibers, Chem. Eng. J., 2010, 159(1–3), 67–74 CrossRef.
- A. P. Marín, J. F. Ortuno, M. I. Aguilar, V. F. Meseguer, J. Sáez and M. Lloréns, Use of chemical modification to determine the binding of Cd (II), Zn (II) and Cr (III) ions by orange waste, Biochem. Eng. J., 2010, 53(1), 2–6 CrossRef.
- G. Sun and W. Shi, Sunflower stalks as adsorbents for the removal of metal ions from wastewater, Ind. Eng. Chem. Res., 1998, 37(4), 1324–1328 CrossRef CAS.
- M. Chiban, A. Soudani, F. Sinan and M. Persin, Wastewater treatment by batch adsorption method onto micro-particles of dried Withania frutescens plant as a new adsorbent, J. Environ. Manage., 2012, 95, S61–S65 CrossRef CAS PubMed.
- T. Altun and E. Pehlivan, Removal of Cr (VI) from aqueous solutions by modified walnut shells, Food Chem., 2012, 132(2), 693–700 CrossRef CAS.
- Y. Kim, J. Park, J. Bang, J. Kim, H. J. Jin and H. W. Kwak, Highly efficient Cr (VI) remediation by cationic functionalized nanocellulose beads, J. Hazard. Mater., 2022, 426, 128078 CrossRef CAS.
- T. S. Anirudhan and M. R. Unnithan, Arsenic (V) removal from aqueous solutions using an anion exchanger derived from coconut coir pith and its recovery, Chemosphere, 2007, 66(1), 60–66 CrossRef CAS.
- J. A. Baig, T. G. Kazi, A. Q. Shah, G. A. Kandhro, H. I. Afridi, S. Khan and N. F. Kolachi, Biosorption studies on the powder stem of Acacia nilotica: removal of arsenic from surface water, J. Hazard. Mater., 2010, 178(1–3), 941–948 CrossRef CAS.
- W. Cao, Z. Wang, H. Ao and B. Yuan, Removal of Cr (VI) by corn stalk-based anion exchanger: the extent and rate of Cr (VI) reduction as side reaction, Colloids Surf., A, 2018, 539, 424–432 CrossRef CAS.
- P. Kaur, K. Raghuvanshi, S. Kumar and A. Kumar, NaOH-treated rice husk, a lignocellulosic biomaterial for removal of Cr (VI) from polluted water, Polym. Bull., 2024, 81(18), 17265–17298 CrossRef CAS.
- S. Zhang, J. Cao, P. Yang, Y. Xie, H. Wang, Y. Mao, K. Ning and Q. Zhang, Adsorption and aggregation of Cu2+ on carboxymethylated sugarcane bagasse: Adsorption behavior and mechanism, J. Hazard. Mater., 2024, 465, 133297 CrossRef CAS PubMed.
- H. S. Jahin, A. I. Khedr and H. E. Ghannam, Banana peels as a green bioadsorbent for removing metals ions from wastewater, Discover Water, 2024, 4(1), 36 CrossRef.
- Y. Zheng, K. Sun, N. Wen, H. Rao, H. Ye and B. Yang, An all-biomass adsorbent: competitive removal and correlative mechanism of Cu2+, Pb2+, Cd2+ from multi-element aqueous solutions, Polym. Bull., 2023, 80(12), 12619–12640 CrossRef CAS.
- L. S. Mustapha, S. O. Jacob-Oricha, M. D. Yahya, S. Y. Lau, A. S. Yusuff and K. S. Obayomi, Effective removal of Cr (VI) and Pb (II) ions from mining wastewater using eco-friendly synthesized magnesium oxide nanoparticles incorporated rice husk ash, Environ. Adv., 2024, 15, 100507 CrossRef CAS.
- X. Xu, B. Gao, X. Huang, J. Ling, W. Song and Q. Yue, Physicochemical characteristics of epichlorohydrin, pyridine and trimethylamine functionalized cotton stalk and its adsorption/desorption properties for perchlorate, J. Colloid Interface Sci., 2015, 440, 219–228 CrossRef CAS PubMed.
- T. F. Akinhanmi, E. A. Ofudje, A. I. Adeogun, P. Aina and I. M. Joseph, Orange peel as low-cost adsorbent in the elimination of Cd (II) ion: kinetics, isotherm, thermodynamic and optimization evaluations, Bioresour. Bioprocess., 2020, 7, 1–6 CrossRef.
- M. I. Al-Ubaidy, Evaluation of parameters affecting the rice husks performance on cyanide ion removal from wastewater, Wasit J. Eng. Sci., 2018, 6(2), 66–74 CrossRef.
- S. Jha, R. Gaur, S. Shahabuddin, I. Ahmad and N. Sridewi, Kinetic and isothermal investigations on the use of low-cost coconut fiber-polyaniline composites for the removal of chromium from wastewater, Polymers, 2022, 14(20), 4264 CrossRef CAS.
- A. S. Singha and A. Guleria, Synthesis and applications of functional polymers from natural okra fibers for removal of Cu (II) ions from aqueous solution, J. Nat. Fibers, 2015, 12(6), 587–603 CrossRef CAS.
- S. Joshi, M. Garg and S. Jana, Thermal activated adsorbent from D. sissoo sawdust for fluoride removal: Batch study, J. Inst. Eng. India Ser. E, 2022, 103(2), 323–337 CrossRef CAS.
- M. Dias, J. Pinto, B. Henriques, P. Figueira, E. Fabre, D. Tavares, C. Vale and E. Pereira, Nutshells as efficient biosorbents to remove cadmium, lead, and mercury from contaminated solutions, Int. J. Environ. Res. Public Health, 2021, 18(4), 1580 CrossRef CAS.
- X. Li, B. Wang, W. Zhang, X. Zhang and X. Wang, Adsorption of Aqueous Nickel Ion by Biomass Carboxymethyl Cellulose-Doped Boron Nitride Composites and Its Subsequent Energy Storage, Polymers, 2025, 17(5), 567 CrossRef CAS PubMed.
- M. Kalaruban, P. Loganathan, W. G. Shim, J. Kandasamy, H. H. Ngo and S. Vigneswaran, Enhanced removal of nitrate from water using amine-grafted agricultural wastes, Sci. Total Environ., 2016, 565, 503–510 CrossRef CAS PubMed.
- H. Zhao, X. K. Ouyang and L. Y. Yang, Adsorption of lead ions from aqueous solutions by porous cellulose nanofiber–sodium alginate hydrogel beads, J. Mol. Liq., 2021, 324, 115122 CrossRef CAS.
- Y. Wang, J. Li, Y. Yuan, Y. Si, J. Xu, M. Li and X. Peng, La (OH)3 loaded magnetic nanocomposites derived from sugarcane bagasse cellulose for phosphate adsorption: characterization, performance and mechanism, Colloids Surf., A, 2021, 626, 127060 CrossRef CAS.
- H. Ao, W. Cao, Y. Hong, J. Wu and L. Wei, Adsorption of sulfate ion from water by zirconium oxide-modified biochar derived from pomelo peel, Sci. Total Environ., 2020, 708, 135092 CrossRef CAS PubMed.
- A. Guleria, G. Kumari and E. C. Lima, Cellulose-g-poly-(acrylamide-co-acrylic acid) polymeric bioadsorbent for the removal of toxic inorganic pollutants from wastewaters, Carbohydr. Polym., 2020, 228, 115396 CrossRef CAS.
- A. Kristek Janković, M. Habuda-Stanić, H. Dong, A. Tutić, Ž. Romić, M. Ergović Ravančić, T. Landeka Dragičević and M. Šiljeg, Utilization of Modified Sunflower Seed as Novel Adsorbent for Nitrates Removal from Wastewater, Water, 2024, 16(1), 73 CrossRef.
- J. Maculewicz, A. D. O'Sullivan, D. Barker, K. T. Wai, S. Basharat and R. Bello-Mendoza, Novel quaternary ammonium functionalized cellulosic materials for nitrate adsorption from polluted waters, Water, Air, Soil Pollut., 2025, 236(1), 1–5 CrossRef.
- J. He, Y. Yang, Z. Wu, C. Xie, K. Zhang, L. Kong and J. Liu, Review of fluoride removal from water environment by adsorption, J. Environ. Chem. Eng., 2020, 8(6), 104516 CrossRef CAS.
- S. Mukherjee and G. Halder, A review on the sorptive elimination of fluoride from contaminated wastewater, J. Environ. Chem. Eng., 2018, 6(1), 1257–1270 CrossRef CAS.
- A. Manyatshe, M. O. Balogun, T. T. Nkambule, Z. E. Cele and T. A. Msagati, Chemical modification of sugarcane bagasse with chitosan for the removal of phosphates in aqueous solution, in AIP Conference Proceedings, AIP Publishing, 2020, Nov 26, vol. 2289, no. 1 Search PubMed.
- L. Zhang, F. Meng, W. Song, X. Ren, J. Wang, X. Cai, X. Li, Y. Li and L. Yan, Enhanced selective removal of phosphate in the presence of high-strength nitrate using straw-based anion imprinted biosorbent, J. Environ. Chem. Eng., 2022, 10(3), 108060 CrossRef CAS.
- K. H. Shah, M. Fareed, M. Waseem, S. Shahida, M. R. Hatshan, S. Sarfraz, A. Batool, M. Fahad, T. Ahmad, N. S. Shah and K. Ha, Tea-Waste-Mediated Magnetic Oxide Nanoparticles as a Potential Low-Cost Adsorbent for Phosphate (PO43−) Anion Remediation, Water, 2023, 15(20), 3541 CrossRef CAS.
- J. Kaur, P. Sengupta and S. Mukhopadhyay, Critical review of bio adsorption on modified cellulose and removal of divalent heavy metals (Cd, Pb, and Cu), Ind. Eng. Chem. Res., 2022, 61(5), 1921–1954 CrossRef CAS.
- R. Das, T. Lindstrom, P. R. Sharma, K. Chi and B. S. Hsiao, Nanocellulose for sustainable water purification, Chem. Rev., 2022, 122(9), 8936–9031 CrossRef CAS PubMed.
- Z. Wang, W. Zhang, J. Yu, L. Zhang, L. Liu, X. Zhou, C. Huang and Y. Fan, Preparation of nanocellulose/filter paper (NC/FP) composite membranes for high-performance filtration, Cellulose, 2019, 26, 1183–1194 CrossRef CAS.
- R. Goswami, S. Singh, P. Narasimhappa, P. C. Ramamurthy, A. Mishra, P. K. Mishra, H. C. Joshi, G. Pant, J. Singh, G. Kumar and N. A. Khan, Nanocellulose: A comprehensive review investigating its potential as an innovative material for water remediation, Int. J. Biol. Macromol., 2024, 254, 127465 CrossRef CAS PubMed.
- N. F. Japri, Z. A. Majid, S. K. Ghoshal, W. H. Danial, H. H. See and M. Z. Othman, On the versatility of graphene-cellulose composites: An overview and bibliometric assessment, Carbohydr. Polym., 2024, 20, 121969 CrossRef.
- A. S. Norfarhana, R. A. Ilyas and N. J. Ngadi, A review of nanocellulose adsorptive membrane as multifunctional wastewater treatment, Carbohydr. Polym., 2022, 291, 119563 CrossRef CAS.
- J. T. Orasugh and S. S. Ray, Nanocellulose-graphene Oxide-Based Nanocomposite for Adsorptive Water Treatment in Functional Polymer Nanocomposites for Wastewater Treatment, Springer International Publishing, Cham, 2022, pp. 1–53 Search PubMed.
- D. Trache, A. F. Tarchoun, M. Derradji, T. S. Hamidon, N. Masruchin, N. Brosse and M. H. Hussin, Nanocellulose: from fundamentals to advanced applications, Front. Chem., 2020, 8, 392 CrossRef CAS PubMed.
- Z. Wang, L. Song, Y. Wang, X. F. Zhang and J. Yao, Construction of a hybrid graphene oxide/nanofibrillated cellulose aerogel used for the efficient removal of methylene blue and tetracycline, J. Phys. Chem. Solids, 2021, 150, 109839 CrossRef CAS.
- P. Joshi, O. P. Sharma, S. K. Ganguly, M. Srivastava and O. P. Khatri, Fruit waste-derived cellulose and graphene-based aerogels: Plausible adsorption pathways for fast and efficient removal of organic dyes, J. Colloid Interface Sci., 2022, 608, 2870–2883 CrossRef CAS PubMed.
- T. C. Mokhena, M. J. Mochane, A. Mtibe, S. Sigonya, B. Ntsendwana, E. G. Masibi, L. Sikhwivhilu and T. S. Motsoeneng, Recent advances on nanocellulose-graphene oxide composites: a review, Cellulose, 2024,(12), 7207–7249 CrossRef CAS.
- J. Gao, J. Wang, M. Cai, Q. Xu, J. Zhang, X. Cao, J. Zhang and Y. Chen, Advanced superhydrophobic and multifunctional nanocellulose aerogels for oil/water separation: A review, Carbohydr. Polym., 2023, 300, 120242 CrossRef CAS PubMed.
- K. Sharma, P. Choudhary, A. Majeed, S. Guleria, M. Kumar, A. K. Rana and G. Rajauria, Cellulose based membranes, hydrogels and aerogels for water treatment application, Ind. Crops Prod., 2025, 225, 120474 CrossRef CAS.
- F. Suo, G. Xie, J. Zhang, J. Li, C. Li, X. Liu, Y. Zhang, Y. Ma and M. Ji, A carbonised sieve-like corn straw cellulose–graphene oxide composite for organophosphorus pesticide removal, RSC Adv., 2018, 8(14), 7735–7743 RSC.
- L. Maiuolo, V. Algieri, F. Olivito, M. A. Tallarida, P. Costanzo, A. Jiritano and A. De Nino, Chronicle of nanocelluloses (NCs) for catalytic applications: Key advances, Catalysts, 2021, 11(1), 96 CrossRef CAS.
- V. Calvo, C. Martínez-Barón, L. Fuentes, W. K. Maser, A. M. Benito and J. M. González-Domínguez, Nanocellulose: The Ultimate Green Aqueous Dispersant for Nanomaterials, Polymers, 2024, 16(12), 1664 CrossRef CAS PubMed.
- J. Hui, H. You, A. Van Beek, J. Zhang, A. Elahi, J. R. Downing, L. E. Chaney, D. Lee, E. A. Ainsworth, S. Chaudhuri and J. B. Dunn, Biorenewable Exfoliation of Electronic-Grade Printable Graphene Using Carboxylated Cellulose Nanocrystals, ACS Appl. Mater. Interfaces, 2024, 16(42), 5753443 CrossRef.
- V. T. Nguyen, L. Q. Ha, T. D. Nguyen, P. H. Ly, D. M. Nguyen and D. Hoang, Nanocellulose and graphene oxide aerogels for adsorption and removal of methylene blue from an aqueous environment, ACS Omega, 2021, 7(1), 1003–1013 CrossRef.
- V. T. Nguyen, L. Q. Ha, L. C. Van, P. T. Huynh, D. M. Nguyen, V. P. Nguyen, T. H. Tran and D. Hoang, Antibiotics tetracycline adsorption and flame-retardant capacity of eco-friendly aerogel-based nanocellulose, graphene oxide, polyvinyl alcohol, and sodium bicarbonate, J. Environ. Chem. Eng., 2023, 11(2), 109523 CrossRef CAS.
- J. Wei, S. H. Gui, J. H. Wu, D. D. Xu, Y. Sun, X. Y. Dong, Y. Y. Dai and Y. F. Li, Nanocellulose-graphene oxide hybrid aerogel to water purification, Appl. Environ. Biotechnol., 2019, 4(1), 11–17 CrossRef.
- L. Chen, Y. Li, S. Hu, J. Sun, Q. Du, X. Yang, Q. Ji, Z. Wang, D. Wang and Y. Xia, Removal of methylene blue from water by cellulose/graphene oxide fibres, J. Exp. Nanosci., 2016, 11(14), 1156–1170 CrossRef CAS.
- H. Hosseini, A. Zirakjou, D. J. McClements, V. Goodarzi and W. H. Chen, Removal of methylene blue from wastewater using ternary nanocomposite aerogel systems: Carboxymethyl cellulose grafted by polyacrylic acid and decorated with graphene oxide, J. Hazard. Mater., 2022, 421, 126752 CrossRef CAS.
- Y. Ma, Y. Hu, X. Yang, Q. Shang, Q. Huang, L. Hu, P. Jia and Y. Zhou, Fabrication, functionalization and applications of cellulose-based aerogels: A review, Int. J. Biol. Macromol., 2024, 26, 138114 Search PubMed.
- N. I. Aris, N. A. Rahman, M. H. Wahid, N. Yahaya, A. S. Abdul Keyon and S. Kamaruzaman, Superhydrophilic graphene oxide/electrospun cellulose nanofibre for efficient adsorption of organophosphorus pesticides from environmental samples, R. Soc. Open Sci., 2020, 7(3), 192050 CrossRef CAS.
- E. A. Elkhatib and H. M. Hamadeen, Nanomaterials for Removal of Organophosphorus Pesticides from Wastewater in Agricultural and Environmental Nanotechnology: Novel Technologies and Their Ecological Impact, Springer Nature Singapore. Singapore, 2023, pp. 583–617 Search PubMed.
- L. Valencia, S. Monti, S. Kumar, C. Zhu, P. Liu, S. Yu and A. P. Mathew, Nanocellulose/graphene oxide layered membranes: elucidating their behaviour during filtration of water and metal ions in real time, Nanoscale, 2019, 11(46), 22413–22422 RSC.
- L. Chen, Y. Li, S. Hu, J. Sun, Q. Du, X. Yang, Q. Ji, Z. Wang, D. Wang and Y. Xia, Removal of methylene blue from water by cellulose/graphene oxide fibres, J. Exp. Nanosci., 2016, 11(14), 1156–1170 CrossRef CAS.
- P. M. da Silva, N. G. Camparotto, T. F. Neves, V. R. Mastelaro, B. Nunes, C. S. Picone and P. Prediger, Instantaneous adsorption and synergic effect in simultaneous removal of complex dyes through nanocellulose/graphene oxide nanocomposites: Batch, fixed-bed experiments and mechanism, Environ. Nanotechnol. Monit. Manage., 2021, 16, 100584 CAS.
- P. Liu, C. Zhu and A. P. Mathew, Mechanically robust high flux graphene oxide-nanocellulose membranes for dye removal from water, J. Hazard. Mater., 2019, 371, 484–493 CrossRef CAS.
- D. Trache, V. K. Thakur and R. Boukherroub, Cellulose nanocrystals/graphene hybrids—a promising new class of materials for advanced applications, Nanomaterials, 2020, 10(8), 1523 CrossRef CAS PubMed.
- T. S. Anirudhan and J. R. Deepa, Nano-zinc oxide incorporated graphene oxide/nanocellulose composite for the adsorption and photo catalytic degradation of ciprofloxacin hydrochloride from aqueous solutions, J. Colloid Interface Sci., 2017, 490, 343–356 CrossRef CAS.
- J. Tao, J. Yang, C. Ma, J. Li, K. Du, Z. Wei, C. Chen, Z. Wang, C. Zhao and X. Deng, Cellulose nanocrystals/graphene oxide composite for the adsorption and removal of levofloxacin hydrochloride antibiotic from aqueous solution, R. Soc. Open Sci., 2020, 7(10), 200857 CrossRef CAS PubMed.
- A. L. Zheng, S. Sabidi, T. Ohno, T. Maeda and Y. Andou, Cu2O/TiO2 decorated on cellulose nanofiber/reduced graphene hydrogel for enhanced photocatalytic activity and its antibacterial applications, Chemosphere, 2022, 286, 131731 CrossRef CAS PubMed.
- M. C. Yang, Y. Q. Tseng, K. H. Liu, Y. W. Cheng, W. T. Chen, W. T. Chen, C. W. Hsiao, M. C. Yung, C. C. Hsu and T. Y. Liu, Preparation of amphiphilic chitosan–graphene oxide–cellulose nanocrystalline composite hydrogels and their biocompatibility and antibacterial properties, Appl. Sci., 2019, 9(15), 3051 CrossRef CAS.
- M. Yao, Z. Wang, Y. Liu, G. Yang and J. Chen, Preparation of dialdehyde cellulose grafted graphene oxide composite and its adsorption behavior for heavy metals from aqueous solution, Carbohydr. Polym., 2019, 212, 345–351 CrossRef CAS PubMed.
- L. Wang, K. Li, K. Copenhaver, S. Mackay, M. E. Lamm, X. Zhao, B. Dixon, J. Wang, Y. Han, D. Neivandt and D. A. Johnson, Review on nonconventional fibrillation methods of producing cellulose nanofibrils and their applications, Biomacromolecules, 2021, 22(10), 4037–4059 CrossRef CAS PubMed.
- J. Zhao, Z. Wei, L. Sun, Y. Wang, X. Wu, T. Wang, Z. Wang and Y. Fu, A novel cellulose-based composite hydrogel microsphere material: for efficient adsorption of Co (II) and Ni (II) ions in water, J. Inorg. Organomet. Polym. Mater., 2025, 35(2), 898–918 CrossRef CAS.
- A. Zaman, J. Orasugh, P. Banerjee, S. Dutta, M. S. Ali, D. Das, A. Bhattacharya and D. Chattopadhyay, Facile one-pot in-situ synthesis of novel graphene oxide-cellulose nanocomposite for enhanced azo dye adsorption at optimized conditions, Carbohydr. Polym., 2020, 246, 116661 CrossRef CAS PubMed.
- M. Chantaso, K. Chaiyong, R. Meesupthong, N. Yingkamhaeng, L. N. Diem, S. Torgbo, P. Sukyai, S. Khantayanuwong, B. Puangsin and P. Srichola, Sugarcane leave-derived cellulose nanocrystal/graphene oxide filter membrane for efficient removal of particulate matter, Int. J. Biol. Macromol., 2023, 234, 123676 CrossRef CAS.
- Z. Karim, M. Hakalahti, T. Tammelin and A. P. Mathew, In situ TEMPO surface functionalization of nanocellulose membranes for enhanced adsorption of metal ions from aqueous medium, RSC Adv., 2017, 7(9), 5232–5241 RSC.
- I. Kozyatnyk and I. Yakupova, Impact of chemical and physical treatments on the structural and surface properties of activated carbon and hydrochar, ACS Sustain. Chem. Eng., 2025, 7(6), 2500–2507 CrossRef.
- K. K. Kishibayev, J. Serafin, R. R. Tokpayev, T. N. Khavaza, A. A. Atchabarova, D. A. Abduakhytova, Z. T. Ibraimov and J. Sreńscek-Nazzal, Physical and chemical properties of activated carbon synthesized from plant wastes and shungite for CO2 capture, J. Environ. Chem. Eng., 2021, 9(6), 106798 CrossRef CAS.
- A. Randhawa, S. D. Dutta, K. Ganguly, T. V. Patil, D. K. Patel and K. T. Lim, A review of properties of nanocellulose, its synthesis, and potential in biomedical applications, Appl. Sci., 2022, 12(14), 7090 CrossRef CAS.
- A. Krishnan, T. Sundaram, B. Nagappan and Y. Devarajan, Integrating artificial intelligence in nanomembrane systems for advanced water desalination, Results Eng., 2024, 24, 103321 CrossRef CAS.
- G. Alam, I. Ihsanullah, M. Naushad and M. Sillanpää, Applications of artificial intelligence in water treatment for optimization and automation of adsorption processes: Recent advances and prospects, Chem. Eng. J., 2022, 427, 130011 CrossRef CAS.
- M. K. Nallakaruppan, E. Gangadevi, M. L. Shri, B. Balusamy, S. Bhattacharya and S. Selvarajan, Reliable water quality prediction and parametric analysis using explainable AI models, Sci. Rep., 2024, 14(1), 7520 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2025 |






