Sterically directed, site-preferential selenization of discrete copper sulfide nanoclusters
Michael J.
Trenerry
 ,
Eryck
García L.
,
Eryck
García L.
 ,
Alex
Lovstedt
,
Alex
Lovstedt
 ,
Lauren E.
Way
,
Lauren E.
Way
 ,
Jan-Niklas
Boyn
,
Jan-Niklas
Boyn
 and
Gwendolyn A.
Bailey
and
Gwendolyn A.
Bailey
 *
*
University of Minnesota – Twin Cities, Minneapolis, MN 55455, USA. E-mail: gbailey@umn.edu
First published on 15th September 2025
Abstract
The controlled incorporation of selenide ions into metal sulfide phases is of great interest to chemists and engineers because of the numerous potential uses of sulfoselenide materials for energy storage and catalysis. However, the need for high-performing devices places stringent demand on compositional and site control in these materials. Herein, we study selenide incorporation into the soluble phosphine-supported copper sulfide nanoclusters [Cu12S6(dppo)4] (1-S: dppo = 1,8-bis(diphenylphopshino)octane) using Se(SiMe3)2 as a selenide source. Selenization occurs preferentially at the [Cu4S4] “equator” sites of the prolate core, with favored retention of sulfides in the apical [Cu4S] positions as judged by X-ray crystallography studies. DFT studies indicate that the site preference arises from the greater steric accessibility of the equatorial sites located under the flexible octane bridges relative to the apical sites covered by four P–Ph moieties, which gives rise to thermodynamic energy differences of ∼5 kcal mol−1. Further solution studies of [Cu12E6(dppo)4] 1-E (E = S, Se) elucidate key differences between the core structures and ligand flexibility in solution. While 77Se NMR studies indicate that 1-Se maintains its prolate core solution, variable-temperature NMR analysis suggest that the dppo ligand is significantly more flexible in 1-Se than 1-S as a result of the larger size of Se, which results in inward puckering of the Se–Cu–Se linkages in the [Cu4Se4] equator. The Se–Cu–Se puckering provides additional room for flexion and rotation of the octane arm and likely contributes to the site preference of selenization. These findings suggest powerful means of tuning metal chalcogenide structure and function via fine tailoring of selenide positioning.
Introduction
Metal chalcogenides have emerged as promising alternatives to precious metals for a range of applications including energy storage, sensing, and catalysis.1–5 Global demand for these materials is anticipated to increase as the world's population grows and precious metal supplies become depleted. However, simple binary chalcogenides MEx (E = S, Se) have limited scope.6 This limitation renders the development of ternary and quaternary materials, including mono- and bimetallic “sulfoselenides,” critical for the achievement of superior performance.6–8 Recently, selenide doping has become popular in the field of photo- and electrochemical catalysis. For example, Mo(SxSe(1−x))2 alloys were found to have significantly lower onset potentials for the hydrogen evolution reaction (HER) compared to either pure MoS2 or MoSe2, with performances in certain cases approaching that of Pt.9,10 Similar effects have been noted in Cu2Mo(SxSe(1−x))4 and Cu2W(SxSe(1−x))4 nanostructures11,12 as well as Co(SxSe(1−x))2 nanowires13 and Cu2(SxSe(1−x)) layered materials on Cu foam (Fig. 1, top).14 In particular, the Cu2(SxSe(1−x)) layers achieved a current density of 100 mA cm−2 at a low overpotential of 315 mV, in contrast to the Cu2S catalyst which required 453 mV to achieve the same activity.14 Varying the sulfide to selenide ratio in CdSxSe(1−x) nanorods has also been found to tune product selectivity in electrochemical synthesis gas production, with CO![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2 ratios varying between 4
H2 ratios varying between 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 to 1
1 to 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4 according to the value of x (0 ≤ x ≤ 1).15
4 according to the value of x (0 ≤ x ≤ 1).15
However, metal sulfoselenide catalysts still underperform relative to Pt group metals, likely in part due to difficulties controlling the crystallinity, surface structure, and active site homogeneity.10,12,14 To this end, researchers have turned to the investigation of atomically precise nanoclusters (APNCs).16–20 As discrete, well-defined analogues of the bulk materials, APNCs are defined as 1–3 nm nanoparticles consisting of a few to hundreds of metal atoms stabilized by organic ligands.16 Significant interest in APNCs, including those incorporating core chalcogenide atoms, arises from their size-dependent properties, which bridge the gap between discrete metal complexes and plasmonic materials and as such exhibit distinct electronic, optical, and magnetic attributes.16,21 For catalysis, the well-defined structures of APNCs enables tailoring and stabilization of specific active site configurations through controlled cluster synthesis and ligand stabilization strategies.19,22 Owing to the increased density of active sites, their catalytic activity can even exceed that of the corresponding bulk materials.22–24 In addition, methods for their incorporation into well-defined superatomic cluster arrays have given rise to the development of atomically well-defined extended solids with tunable dimensionality and physicochemical properties.25–27
By extension from the bulk materials, the controlled substitution of chalcogenides to yield sulfoselenide clusters and arrays with fixed compositions and selenide placements could provide one further lever for enhancing catalyst performance. At the atomically precise level, however, chalcogenide exchange reactivity of metal sulfide/selenide clusters has been scarcely explored. In an early example, Holm and Reynolds studied mixtures of [Fe4S4(SR)4]2− and [Fe4Se4(SR)4]2− clusters, which spontaneously exchanged in acetonitrile solutions leading to the formation of mixed [Fe4S(4−x)Sex(SR)4]2− species in near-equilibrium proportions.28 Similar results were reported for [Fe4E4(SR)4]3− and [Fe2E2(SR)4]0/1−/2− clusters (E = S, Se). Fedin and coworkers later reported the selective replacement of equatorial persulfide sites in [Mo3(μ3-S)(μ2-S)2X6]2− (X = Cl, Br, NCS) with selenides upon treatment with KSeCN or Ph3PSe.29 Rees and coworkers then extended these principles to Fe- and Mo-containing metalloenzymes of bacterial nitrogenase, finding that exogenous selenide from KSeCN replaces all sulfide sites of the [Fe4S4] cluster found in the iron protein,30 while it selectively replaces the sulfide bridge sites of the FeMo-cofactor responsible for nitrogen fixation.31 In an unrelated example, Dehnen and coworkers synthesized the multinary cluster [(CuPPh3)6(E)6(SnFc)2] (Fc = ferrocenyl) by reaction of [(SnFc)4Se6] with Na2S in the presence of a Cu source.32 While these examples demonstrate feasibility, generalizable strategies for controlled and site-selective selenization of metal chalcogenide APNCs have not been established.
Recently, our research team confirmed the unique solution stability of the [Cu12S6] nanocluster core when stabilized by chelating 1,8-bis(diphenylphosphino)octane (dppo) ligands.33–35 The cluster [Cu12S6(dppo)4] (1-S) exhibited a hydrodynamic radius (rH) of 9.4 Å in solutions of THF-d8 or C6D6 as determined by 1H DOSY NMR, consistent with an intact cluster core based on reported crystallographic data.33 We also showed that 1-S is redox-active, exhibiting a chemically quasireversible redox couple at −0.50 V vs. Fc0/+ in THF/electrolyte solutions. These properties, together with the known photoluminescent activity of copper chalcogenide clusters, make them promising candidates for application in photo/electrochemical catalysis and light-emitting devices.34,36–38 As such, we became interested in the development of sulfoselenide analogues of 1-S (Fig. 1, bottom). The dppo ligand creates two distinct environments for the sulfide sites in 1-S, where the octane linker surrounding the equatorial sites is more flexible and less sterically demanding than the PPh groups surrounding the apical site (Fig. 1, space-filling diagrams in bottom right). In addition, short o-CH–S interactions to the PPh groups further rigidify the steric environment surrounding the apical sites. We reasoned that this ligand-differentiated environment could result in a site preference for placement of Se in the less sterically constrained equatorial sites due to its slightly larger size (ionic radius: 1.98 Å, vs. 1.84 Å for S2−). At the same time, a kinetic preference could arise from the strong o-CH–S interactions to PPh, which would disfavour Cu–S bond cleavage and replacement with Se at the apical sites. If successful, this strategy would provide a blueprint for the development of well-defined sulfoselenide APNCs and extended crystalline materials with controlled colocalization of sulfide and selenide.
Herein, we describe a strategy for site-preferential chalcogenide exchange of [Cu12S6(dppo)4] (1-S) at the equatorial sites. Thus, XRD analysis of crystals grown from sulfide exchange reactions with Se(SiMe3)2 unequivocally demonstrate a ∼35% enhancement in selenide substitution at the equatorial positions (1.22![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 Se
1 Se![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) S, vs. 0.69
S, vs. 0.69![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 Se
1 Se![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) S in apical sites). Interestingly, crystals grown from the de novo synthesis of [Cu12SxSe(6−x)(dppo)4] from 2
S in apical sites). Interestingly, crystals grown from the de novo synthesis of [Cu12SxSe(6−x)(dppo)4] from 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 Se(SiMe3)2
1 Se(SiMe3)2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) S(SiMe3)2 reveal a similar site preference, suggesting that the positional isomerism of sulfoselenide clusters may be controlled through dynamic self-sorting. DFT calculations confirm the key role of the dppo ligands in dictating a thermodynamic site preference, and provide insight into subtle electronic changes imparted by selenization. Together, these results indicate a powerful, potentially generalizable strategy for enriching selenium substitution in specific nanocluster sites, which could be important in enabling the next level of atomistic control over structure, properties, and reactivity of mixed sulfoselenide materials.
S(SiMe3)2 reveal a similar site preference, suggesting that the positional isomerism of sulfoselenide clusters may be controlled through dynamic self-sorting. DFT calculations confirm the key role of the dppo ligands in dictating a thermodynamic site preference, and provide insight into subtle electronic changes imparted by selenization. Together, these results indicate a powerful, potentially generalizable strategy for enriching selenium substitution in specific nanocluster sites, which could be important in enabling the next level of atomistic control over structure, properties, and reactivity of mixed sulfoselenide materials.
Results and discussion
The selenide nanocluster [Cu12Se6(dppo)4] (1-Se) has been previously reported,36 but prior to this work, its solution characterization was unknown. To confirm the stability of 1-Se in solution, we first subjected it to an in-depth 1D/2D NMR analysis. The 1H and 31P{1H} NMR spectra collected in THF-d8 revealed a spectral signature distinct from that of the free dppo ligand and 1-S. Like for 1-S,33 the 1H NMR spectral signature suggested that the [Cu12Se6] nanocluster core was preserved in solution via a symmetrical, locked ligand configuration. In particular, the two Ph rings within each –PPh2 unit are rendered inequivalent, with one of the o-CH signals shifted substantially downfield relative to the free dppo ligand (δH 8.37, vs. 8.43 ppm in 1-S; Fig. 2a). The downfield shift arises from CH–S/Se interactions between the cisoid phenyl o-CH and apical S/Se in 1-S/Se, which remain short in 1-Se (avg. 2.829 Å from XRD; Fig. 2b, left).33,39 This distance is only 0.05 Å longer than the corresponding CH–S distances in 1-S, suggesting considerable steric constraint with the larger chalcogenide. The aliphatic protons of the octane linker are also distinguished as pairs of diastereotopic multiplets except the geminal delta signals, which are coalesced at room temperature. The reported crystallographic α-CH–Se distances for equatorial Se in 1-Se are much longer than for the apical Se (avg. 3.261 Å), suggesting less steric constraint. A 1H DOSY NMR analysis confirmed the intactness of 1-Se in solution, with the proton signals correlating to a uniformly small diffusion coefficient consistent with the expected hydrodynamic radius of the intact nanocluster (rH = 9.4 Å, vs. rXRD = 9.4 Å from crystallography; Fig. S3). The measured hydrodynamic radius of 1-Se is essentially the same as that of 1-S,33 despite small differences in the crystallographic Cu–S/Se bond distances reported for each species.36 | ||
| Fig. 2 (a) 1H NMR spectra (THF-d8, 400 MHz) of 1-S and 1-Se showing that the conformationally restricted ligand environment in 1-Se is similar to that in 1-S. Ph o/m/p-CH signals belonging to cisoid and transoid Ph rings are denoted with c and t subscripts, respectively. (b) Truncated crystallographic structures (CCDC: 1044373)36 showing representative CH–Se distances in the apical and equatorial view. Colour scheme: dark grey (cisoid Ph–C); light grey (transoid Ph–C); orange (P); blue (Cu); red (Se); white (H). H atoms are omitted except cisoid Ph o-CH and octane α-CHa, which are shown interacting with S. | ||
Direct interrogation of the core structure in 1-Se was facilitated by 77Se{1H} NMR spectroscopy. This marks an important development from previous work on Cu12S6(dppo)4, which was limited to the 1H, 13C, and 31P nuclei of the supporting ligands.33 Thus, a triplet and broad singlet observed at −646.8 ppm and −173.2 ppm were assigned to the four equivalent equatorial Se atoms and two apical Se atoms of the [Cu12Se6] core, respectively (Fig. 3). These assignments are corroborated by DFT predictions of 77Se chemical shifts (Section S6 of the SI) and are consistent with the expected relative intensities of the signals. Additionally, the triplet splitting pattern for the signal at −646.8 ppm is consistent with two-bond 77Se–31P coupling between equatorial Se sites and phosphine ligands bound at opposite ends of the cluster. Furthermore, the J-coupling constant of 2J(77Se, 31P) = 99.5 Hz closely matches a literature report of cis-disposed M(PPh3)2(SeNR)2 complexes (M = Pd, Pt) in which 2J(77Se, 31P) = 90 and 137 Hz, respectively.40 The nearly 500 ppm difference in chemical shifts between the apical and equatorial sites of 1-Se is attributed to differences in paramagnetic shielding effects, as confirmed by DFT calculations of the NMR shielding tensors (Table S7). Together, these results demonstrate that the elongated cuboctahedral core of 1-Se is maintained in the solution phase, resulting in symmetry-distinct apical and equatorial selenide sites possessing substantially different 77Se chemical shifts. They also add to a limited body of literature disclosing 77Se NMR shifts of discrete metal chalcogenide clusters, for which site-differentiation detected by 77Se NMR spectroscopy is particularly rare.41,42 Of relevance to this work, previous reports by Corrigan and coworkers identified only one 77Se signal for each metal selenide cluster 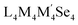 (L = a cyclic(alkyl)(amino)carbene or N-heterocyclic carbene ligand; M and M′ = Cu, Ag, or Au: δSe = −655 to −447 ppm), indicating that all selenide sites are symmetry equivalent.41,42
(L = a cyclic(alkyl)(amino)carbene or N-heterocyclic carbene ligand; M and M′ = Cu, Ag, or Au: δSe = −655 to −447 ppm), indicating that all selenide sites are symmetry equivalent.41,42
Variable-temperature NMR spectroscopy
The differences in the 1H NMR spectra of 1-S and 1-Se suggest that Se substitution may lead to differences in ligand dynamics, which in turn may impact reactivity at S/Se. The room-temperature NMR spectra differ most notably in the broadness of the signals for geminal δ-proton resonances, which present as a broad singlet in 1-Se but are split into two distinct peaks for the diastereotopic pair in 1-S.To further understand how Se substitution impacts solution structure and dynamics, we undertook a comprehensive variable-temperature 1H NMR study of 1-S and 1-Se (Fig. 4a). By cooling the solutions in ca. 20 °C increments, we determined the coalescence temperature of the geminal δ-protons to be −26 °C in 1-Sevs. 60 °C in 1-S. The drastically different coalescence temperatures in these two species indicates that the torsion of alkyl C–C bonds is significantly more restricted for dppo ligands bound to the [Cu12S6] core than to the analogous [Cu12Se6] core. This trend may be rationalized by differences in the core structures of 1-S and 1-Se as seen in the reported crystallographic data for each compound (Fig. 4b).34,36 According to these data, the three-bond S–Cu–S linkages comprising the Cu4S4 “equator” substructure of 1-S are roughly linear (avg. 175.2°), whereas the analogous Se–Cu–Se linkages in 1-Se are puckered inwards (avg. 167.6°) such that there is greater separation between the central Cu atoms at each face of the nanocluster core and the alkyl chains of dppo ligands. This gives the dppo ligand more freedom to rotate, contributing presumably to the dramatically lower coalescence temperature of geminal δ-CH in 1-Se. At the same time, inward puckering of Cu4Se4 substructure leads to increased strain at both Cu and Se, which deviate from their ideal bond angles. These experiments thus confirm the retention of chemical strain in the solid and solution state induced by selenization in 1-Se. In metal sulfoselenide materials, chemical strain arising from the relatively long M–Se linkages has been proposed to play a key role in enhancing catalytic activity. For example, it has been associated with a decrease in the free energy of hydrogen absorption (ΔGH*) in Cu2W(SxSe(1−x))4 leading to improved HER activity.12
 | ||
| Fig. 4 (a) Partial variable-temperature 1H NMR spectra (400 MHz, THF-d8) of 1-S (left) and 1-Se (right) showing coalescence of key δ geminal methylene signals at 60 °C and −26 °C, respectively. The full 1H and 31P{1H} NMR spectra are shown in Fig. S17–S22. (b) Partial structures of 1-S and 1-Se showing differences in the bond metrics for the equatorial Cu4S4 and Cu4Se4 moieties (adapted from the reported crystallographic data).36 Color scheme: blue (Cu), yellow (S), red (Se). | ||
Chalcogenide exchange
The above analysis suggests that 1-Se retains its core structure in solution and that while the apical Se sites are constricted by the PPh groups, the equatorial sites are significantly less encumbered due to puckering of the Cu4Se4 equator, which leads to much greater inherent flexibility of the octane linker. We therefore hypothesized that the dramatic steric differences between the apical and equatorial Se sites would lead to a measurable thermodynamic preference for substitution at the equatorial positions, while a kinetic preference could arise from steric blocking of apical sites in 1-S by the PPh groups.To test this hypothesis, we developed synthetic methodology to effect partial selenization of [Cu12S6(dppo)4]. Thus, 13 equiv. Se(SiMe3)2 were added to a benzene solution of 1-S and stirred at RT over the course of several days (Fig. 5a). During this time, an unidentified gray precipitate formed and the colour of the filtered supernatant changed from red to orange, matching the appearance of independently synthesized samples of 1-Se. MALDI-MS analysis of samples at three-day intervals revealed clusters of peaks with characteristic m/z and isotope patterns matching those for the expected 1-S, 1-Se, and intermediate sulfoselenide clusters [Cu12SxSe(6−x)(dppo)4] (0 ≤ x ≤ 6; Fig. 5b, S8 and S9). The extent of selenization increased with later time points and ultimately reached complete conversion to 1-Se after 9 days. Simultaneously, the 1H and 31P{1H} NMR spectra revealed a gradual decrease in the intensity of signals for 1-S and the simultaneous formation of selenium-substituted congeners, with complete conversion to 1-Se over 196 hours (Fig. S10 and S11). The 31P{1H} NMR spectra also indicated the formation of free dppo and phosphine selenides, indicating partial cluster degradation. While the mechanism of selenization is currently unclear, the MALDI-MS spectra suggest an associative mechanism in which Se(SiMe3)2 binds to the [Cu12S6] cluster core prior to sulfide release. In particular, minor peaks corresponding to clusters with expanded cores bearing seven chalcogenides (e.g. [Cu12S6Se], [Cu12Se7], etc.) were observed to rise and decay over the course of selenization (Fig. 5, S8 and S9). Interestingly, no new silicon-containing peaks were detected in the 29Si NMR spectra, suggesting that the silane groups may be contained in the gray precipitate.
Of note, chalcogenide exchange proceeded in only one direction for this system, as the analogous reaction of 1-Se and S(SiMe3)2 did not lead to the formation of 1-S or any sulfoselenide intermediates (Fig. S12 and S13). While thermodynamics favour the formation of copper(I) sulfide phases over selenide,37 the absence of reactivity with S(SiMe3)2 is consistent with the higher bond dissociation energy for the S–Si bond (619(13) kJ mol−1) relative to Se–Si (531(25) kJ mol−1).
The degree of site preference for substitution at the equatorial vs. apical sites was assessed by single crystal XRD studies. A filtered aliquot of the reaction solution after three days was crystallized by slow evaporation (Fig. 6a). Single crystal XRD followed by structure determination and refinement yielded a structure solution in which sulfoselenide species [Cu12SxSe(6−x)(dppo)4] were cocrystallized with substitutional disorder (R1 = 0.0686, wR2 = 0.1109 on all data; Fig. 6b). The best model yielded 41% Se at the apical sites vs. 55% at the equatorial sites, consistent with a significant preference for Se substitution at the equatorial sites.
Similar results were obtained from the de novo synthesis of sulfoselenide clusters (Fig. 6b, bottom). Thus, reaction of 12 CuOAc and dppo with a 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 mixture of S(SiMe3)2 and Se(SiMe3)2, followed by filtration and slow evaporation of the solvent, led to the growth of single crystals of [Cu12SxSe(6−x)(dppo)4] (Fig. S27). Data refinement likewise yielded a substitutionally disordered structure showing 65% Se substitution on the apical position, vs. 80% substitution at the equatorial position (R1 = 0.0758, wR2 = 0.1122 on all data; Fig. 6b, right). These results indicate a similar preference for Se substitution at the equatorial sites. This preference is notable given that Cu–S/Se bond formation occurs in the de novo synthesis from monometallic inputs via smaller cluster aggregates,37,43 and hence, kinetic site preferences arising from the local steric environments of a fully assembled cluster are not expected to play a major role. The similar site preference observed for the two methods suggests that the enhanced Se occupancy at the equatorial sites may be thermodynamically controlled.
1 mixture of S(SiMe3)2 and Se(SiMe3)2, followed by filtration and slow evaporation of the solvent, led to the growth of single crystals of [Cu12SxSe(6−x)(dppo)4] (Fig. S27). Data refinement likewise yielded a substitutionally disordered structure showing 65% Se substitution on the apical position, vs. 80% substitution at the equatorial position (R1 = 0.0758, wR2 = 0.1122 on all data; Fig. 6b, right). These results indicate a similar preference for Se substitution at the equatorial sites. This preference is notable given that Cu–S/Se bond formation occurs in the de novo synthesis from monometallic inputs via smaller cluster aggregates,37,43 and hence, kinetic site preferences arising from the local steric environments of a fully assembled cluster are not expected to play a major role. The similar site preference observed for the two methods suggests that the enhanced Se occupancy at the equatorial sites may be thermodynamically controlled.
DFT analysis of chalcogen substitution at 1-S
To help elucidate the origins of site preference for selenide substitution at 1-S, we next conducted combined geometry optimization and thermochemical calculations via DFT. These calculations focused on 1-S′, 1-Se′, where the dppo ligand was truncated to PPh2Et, and 16 sulfoselenide constitutional isomers [Cu12SxSe(6−x)(PPh2Et)8] (0 < x < 6). Geometry optimizations and thermochemical calculations were performed using unrestricted Kohn–Sham DFT using the BP86 functional44,45 in combination with the def2-SVP basis set.46 Similar methods were found previously to give rise to good agreement between predicted electronic transitions and UV-vis electronic absorption spectra for 1-S.36 In addition, the character of the frontier orbitals was found to be similar between dppo-supported 1-S and truncated 1-S′, supporting the validity of using truncated ligands for electronic structure analysis (Fig. S35). In general, we find reasonable agreement between the DFT-predicted bond metrics for the truncated structures and crystallographic bond metrics for 1-S and 1-Se, although the Cu–Cu bond lengths in the computed structures are consistently underestimated (Table S8).33Consistent with chalcogenide substitution patterns observed by XRD, the ground-state thermochemical calculations consistently predicted selenide placement at the equatorial sites to be more favourable, with a ca. 5 kcal mol−1 penalty each time an apical sulfide and equatorial selenide were exchanged (Fig. 7 and S29). To confirm that selenization is sterically directed, thermochemical calculations were carried out on the geometry-optimized clusters [Cu12SxSe(6−x)(PMe3)8] stabilized by sterically undemanding PMe3 ligands. In this case, chalcogenide exchange between an apical sulfide and equatorial selenide incurred only a ca. 0.5 kcal mol−1 penalty (Fig. 7) and both the apical and equatorial sites were relatively sterically uncongested compared to the structures of 1-S′ and 1-Se′ (Fig. S29 and S33). These calculations confirm that the sterically-differentiated ligand environments in 1-S/Se are likely responsible for the equatorial site preference of selenization in 1-S.
To understand the impact of selenization on electronic structure, we also undertook a molecular orbital analysis of 1-S′, 1-Se′, and four constitutional isomers of [Cu12S6Se3(PPh2Et)8]. Interestingly, analysis of the calculated HOMOs and LUMOs revealed minimal impact of selenization on the electronic structure. The calculated HOMO–LUMO gaps for all six isomers were within 1.5 kcal mol−1, while the shape of the HOMOs and LUMOs were similar for all compounds investigated (Fig. 8). For all structures, the HOMOs displayed localized electron density on the metal and chalcogenide atoms, with the electron density on S/Se and Cu predominantly displaying p- and d-orbital character, respectively. A natural charge analysis carried out using the NBO7 (ref. 47) software further confirmed that the charge distribution is similar regardless of selenization, with average natural charges on Cu of 0.508 and 0.497 on Cu for 1-S′ and 1-Se′, respectively (Table S7). Additionally, the average charge was consistently higher on apical Cu atoms (∼0.55) and lower on equatorial Cu atoms (∼0.40) for all structures calculated.
Energies of the singly oxidized states at the neutral geometry were also calculated to obtain the vertical ionization energies (IEs; Table S10). Although 1-S′ and the partially selenized clusters have similar IEs, the IE of 1-Se′ was seen to decrease from ca. 108.5 to 107.1 kcal mol−1 consistent with electrochemical characterization of independently prepared 1-Se showing that complete selenization renders the cluster core only slightly more reducing than 1-S (Fig. S25).48 Likewise, the calculated SOMOs and spin density plots of oxidized clusters [1-S′]+ and [1-Se′]+ show that the effect of selenization on electron and spin delocalization is minimal. As the Se content increases, the extent of electron delocalization from the Cu atoms onto the chalcogen atoms slightly increases, with a total spin of 0.505 on Cu and 0.126 on Se atoms in [1-Se′]+ compared to 0.554 on Cu and 0.105 on S in [1-S′]+ (Table S11). Meanwhile, electron delocalization in the mixed sulfoselenide clusters is intermediate compared to either [1-S′]+ or [1-Se′]+ and largely invariant to the specific positionality of Se. The increased delocalization in 1-Se′ likely contributes to the slightly lower IE by stabilizing the oxidized state.
This computational analysis offers an atomically precise perspective on how subtle differences in electronic structure can engender dramatic reactivity differences in metal sulfides, selenides, and mixed sulfoselenides. For example, nanocubes of β-Cu2Se exhibited remarkably high faradaic efficiencies for C2+ products during electrocatalytic CO2 reduction, with 84% ethanol and 15% acetate being formed at relatively modest applied potentials (−0.6 V vs. RHE),49 while Cu2S nanocubes produced primarily H2 and formic acid at similar applied potentials.50 The exceptional C2+ selectivity of β-Cu2Se was attributed to the Cuδ+ character of the Cu atoms (0 < δ < 1), which are more electron-rich in Cu2Se compared to Cu2S resulting in higher CO dwell times.49 The DFT calculations presented herein indicate that relatively minor differences in electron delocalization shift the IE and redox potentials to lower values, making it effectively easier to remove an electron. Meanwhile, partial selenization leads to similar IE but increases chemical strain.12 This apparently has a synergistic impact on HER performance, as demonstrated in the mixed sulfoselenide materials Cu2SxSe(1−x) and Cu2M(SxSe(1−x))4 (M = Mo, W).11,12,14
Conclusions
Herein, we report a method for the sterically directed, site-preferential selenization of discrete copper sulfide nanoclusters. Thus, selenization of [Cu12S6(dppo)4] (1-S) is dictated by the differentiated ligand bulk in apical [Cu4E] pyramids and central [Cu4E4] equator sites, which results in steric constraints being imposed on Se due to its slightly larger size. Additionally, puckering of the [Cu4Se4] equator in 1-Se leads to decreased steric encumbrance between the equatorial Se and dppo octane linkages. These differences result in thermodynamic energy differences of ca. 5 kcal mol−1 between sulfoselenide constitutional isomers in which Se is placed in the equatorial vs. apical sites. Importantly, measurable site preferences arise from even relatively subtle differences in local steric environments, visualized in Fig. 1b. These findings suggest that even larger differences could be obtained if the ligand environment were deliberately manipulated to maximize steric differences as demonstrated previously in other cluster systems.51–53 Additionally, the use of organic ligands to sterically impede certain surface sites of bulk materials (explored in other contexts54,55) could lead to preferential selenization of unencumbered sites. Greater control over selenization in these systems could lead to better fine-tuning of reactivity and physicochemical properties towards the next generation of multinary sulfoselenide materials.Author contributions
This research was conceptualized and the manuscript written with joint contributions from MJT and GAB. MJT performed all non-crystallographic experiments and analysis, and performed geometry optimizations and thermochemical calculations via DFT. EGL and AL collected the crystallographic data and refined the structure solutions. LEW and JNB performed the molecular orbital analysis, IE calculations, and spin density plots of 1-S′, 1-Se′, constitutional isomers of [Cu12S6Se3(PPh2Et)8], and their oxidized analogs.Conflicts of interest
There are no conflicts to declare.Data availability
CCDC 2466271 and 2466272 contain the supplementary crystallographic data for this paper.56a,bSupplementary information: NMR spectra, MALDI mass spectra, electrochemical data, X-ray diffraction data, computational details, and Cartesian coordinates of DFT-optimized structures. See DOI: https://doi.org/10.1039/d5ta05259a.
Acknowledgements
This research was supported by startup funds through the University of Minnesota – Twin Cities and an ACS PRF grant, under award number 67165-DNI3. NMR, MALDI-MS, and single crystal XRD experiments were carried out in the LeClaire-Dow Instrumentation Facility at UMN. NMR characterization was supported by the National Institutes of Health Office of the Director (award #S10OD011952). MALDI-MS characterization was supported by a University of Minnesota Research Infrastructure Investment grant. Single crystal XRD experiments were supported by a National Science Foundation Major Research Instrumentation grant (award #1229400). LEW acknowledges support from the 3M Science and Technology Graduate Fellowship.References
- S. Palchoudhury, K. Ramasamy, J. Han, P. Chen and A. Gupta, Transition metal chalcogenides for next-generation energy storage, Nanoscale Adv., 2023, 5, 2724–2742 RSC.
- M. A. Reynolds, Minireview on the Evolution of Tetrathiometallate Salts as Protean Building Blocks of Catalysts and Materials for the Energy Transition: Recent Advances and Future Perspectives, Energy Fuels, 2021, 35, 15285–15300 CrossRef CAS.
- R.-W. Huang, Y.-S. Wei, X.-Y. Dong, X.-H. Wu, C.-X. Du, S.-Q. Zang and T. C. W. Mak, Hypersensitive dual-function luminescence switching of a silver-chalcogenolate cluster-based metal–organic framework, Nat. Chem., 2017, 9, 689–697 CrossRef CAS PubMed.
- K. Nakamae, T. Nakajima, Y. Ura, Y. Kitagawa and T. Tanase, Facially Dispersed Polyhydride Cu9 and Cu16 Clusters Comprising Apex-Truncated Supertetrahedral and Square-Face-Capped Cuboctahedral Copper Frameworks, Angew. Chem., Int. Ed., 2020, 59, 2262–2267 CrossRef CAS PubMed.
- D. M. Khaidar, W. N. R. W. Isahak, Z. A. C. Ramli and K. N. Ahmad, Transition metal dichalcogenides-based catalysts for CO2 conversion: An updated review, Int. J. Hydrogen Energy, 2024, 68, 35–50 CrossRef CAS.
- J. T. Perryman and J. M. Velázquez, Design Principles for Multinary Metal Chalcogenides: Toward Programmable Reactivity in Energy Conversion, Chem. Mater., 2021, 33, 7133–7147 CrossRef CAS.
- J. C. Ortiz-Rodríguez and J. M. Velázquez, Structure–reactivity relationships in Chevrel phase electrocatalysts for small-molecule reduction reactions, Curr. Opin. Electrochem., 2022, 34, 101002 CrossRef.
- Y. Hu, J. Zhu, X. Wang, X. Zheng, X. Zhang, C. Wu, J. Zhang, C. Fu, T. Sheng and Z. Wu, Mo4+-Doped CuS Nanosheet-Assembled Hollow Spheres for CO2 Electroreduction to Ethanol in a Flow Cell, Inorg. Chem., 2024, 63, 9983–9991 CrossRef CAS PubMed.
- H. Zhou, F. Yu, Y. Huang, J. Sun, Z. Zhu, R. J. Nielsen, R. He, J. Bao, W. A. Goddard III, S. Chen and Z. Ren, Efficient hydrogen evolution by ternary molybdenum sulfoselenide particles on self-standing porous nickel diselenide foam, Nat. Commun., 2016, 7, 12765 CrossRef CAS PubMed.
- R. Bar-Ziv, O. E. Meiron and M. Bar-Sadan, Enhancing the catalytic activity of the alkaline hydrogen evolution reaction by tuning the S/Se ratio in the Mo(SxSe1−x)2 catalyst, Nanoscale, 2018, 10, 16211–16216 RSC.
- A. P. Tiwari, D. Kim, Y. Kim, O. Prakash and H. Lee, Highly active and stable layered ternary transition metal chalcogenide for hydrogen evolution reaction, Nano Energy, 2016, 28, 366–372 CrossRef CAS.
- A. P. Tiwari, A. Azam, T. G. Novak, O. Prakash and S. Jeon, Chemical strain formation through anion substitution in Cu2WS4 for efficient electrocatalysis of water dissociation, J. Mater. Chem. A, 2018, 6, 7786–7793 RSC.
- K. Wang, C. Zhou, D. Xi, Z. Shi, C. He, H. Xia, G. Liu and G. Qiao, Component-controllable synthesis of Co(SxSe1−x)2 nanowires supported by carbon fiber paper as high-performance electrode for hydrogen evolution reaction, Nano Energy, 2015, 18, 1–11 CrossRef CAS.
- S. Kumaravel, D. Kumar, S. Sam Sankar, A. Karmakar, R. Madhu, K. Bera, H. N. Dhandapani, S. Nagappan, S. Chakraborty and S. Kundu, Vacancy-fused multiple layers of copper sulfoselenide superstructures: a propitious HER electrocatalyst in acidic medium, Catal. Sci. Technol., 2023, 13, 694–704 RSC.
- R. He, A. Zhang, Y. Ding, T. Kong, Q. Xiao, H. Li, Y. Liu and J. Zeng, Achieving the Widest Range of Syngas Proportions at High Current Density over Cadmium Sulfoselenide Nanorods in CO2 Electroreduction, Adv. Mater., 2018, 30, 1705872 CrossRef PubMed.
- R. Jin, C. Zeng, M. Zhou and Y. Chen, Atomically Precise Colloidal Metal Nanoclusters and Nanoparticles: Fundamentals and Opportunities, Chem. Rev., 2016, 116, 10346–10413 CrossRef CAS PubMed.
- I. Chakraborty and T. Pradeep, Atomically Precise Clusters of Noble Metals: Emerging Link between Atoms and Nanoparticles, Chem. Rev., 2017, 117, 8208–8271 CrossRef CAS PubMed.
- Y. Du, H. Sheng, D. Astruc and M. Zhu, Atomically Precise Noble Metal Nanoclusters as Efficient Catalysts: A Bridge between Structure and Properties, Chem. Rev., 2020, 120, 526–622 CrossRef CAS PubMed.
- R. Jin, G. Li, S. Sharma, Y. Li and X. Du, Toward Active-Site Tailoring in Heterogeneous Catalysis by Atomically Precise Metal Nanoclusters with Crystallographic Structures, Chem. Rev., 2021, 121, 567–648 CrossRef CAS PubMed.
- Atomically Precise Nanochemistry, ed. R. Jin and D.-e. Jiang, John Wiley & Sons Ltd, Hoboken, NJ, 2023 Search PubMed.
- Y. Niihori, S. Miyajima, A. Ikeda, T. Kosaka and Y. Negishi, Vertex-Shared Linear Superatomic Molecules: Stepping Stones to Novel Materials Composed of Noble Metal Clusters, Small Sci., 2023, 3, 2300024 CrossRef CAS PubMed.
- Y. Liu, J. Yu, Y. Lun, Y. Wang, Y. Wang and S. Song, Ligand Design in Atomically Precise Copper Nanoclusters and Their Application in Electrocatalytic Reactions, Adv. Funct. Mater., 2023, 33, 2304184 CrossRef CAS.
- J. Kibsgaard, T. F. Jaramillo and F. Besenbacher, Building an appropriate active-site motif into a hydrogen-evolution catalyst with thiomolybdate [Mo3S13]2− clusters, Nat. Chem., 2014, 6, 248–253 CrossRef CAS PubMed.
- T. Kawawaki, T. Okada, D. Hirayama and Y. Negishi, Atomically precise metal nanoclusters as catalysts for electrocatalytic CO2 reduction, Green Chem., 2024, 26, 122–163 RSC.
- A. Pinkard, A. M. Champsaur and X. Roy, Molecular Clusters: Nanoscale Building Blocks for Solid-State Materials, Acc. Chem. Res., 2018, 51, 919–929 CrossRef CAS PubMed.
- E. A. Doud, A. Voevodin, T. J. Hochuli, A. M. Champsaur, C. Nuckolls and X. Roy, Superatoms in materials science, Nat. Rev. Mater., 2020, 5, 371–387 CrossRef.
- J.-M. Li, T.-Y. Xu, Y.-L. Zhao, X.-L. Hu and K.-H. He, Two 6/10-connected Cu12S6 cluster-based organic frameworks: crystal structure and proton conduction, Dalton Trans., 2021, 50, 7484–7495 RSC.
- J. G. Reynolds and R. H. Holm, Core chalcogenide atom substitution reactions of [Fe2X2(SR)4]2− and [Fe4X4(SR)4]2−,3− clusters (X = sulfur, selenium), Inorg. Chem., 1981, 20, 1873–1878 CrossRef CAS.
- V. P. Fedin, M. N. Sokolov, V. Ye. Fedorov, D. S. Yufit and Yu. T. Struchkov, Reactions of triangular Mo3S7X62− (X = Cl, Br, NCS) complexes with KSCN and KSeCN, resulting in stereoselective substitution of sulfur atom in asymmetrically coordinated μ2S2 ligand. X-ray structure of (PPN)2Mo3(μ3-S)(μ2-SSe)3Cl6, Inorg. Chim. Acta, 1991, 179, 35–40 CrossRef CAS.
- T. M. Buscagan, J. T. Kaiser and D. C. Rees, Selenocyanate derived Se-incorporation into the nitrogenase Fe protein cluster, eLife, 2022, 11, e79311 CrossRef CAS.
- T. Spatzal, K. A. Perez, J. B. Howard and D. C. Rees, Catalysis-dependent selenium incorporation and migration in the nitrogenase active site iron-molybdenum cofactor, eLife, 2015, 4, e11620 CrossRef PubMed.
- Z. You, J. Bergunde, B. Gerke, R. Pöttgen and S. Dehnen, Ferrocenyl-Functionalized Sn/Se and Sn/Te Complexes: Synthesis, Reactivity, Optical, and Electronic Properties, Inorg. Chem., 2014, 53, 12512–12518 CrossRef CAS PubMed.
- M. J. Trenerry and G. A. Bailey, Ditopic Ligand Effects on Solution Structure and Redox Chemistry in Discrete [Cu12S6] Clusters with Labile Cu–S bonds, Nanoscale, 2024, 16, 16048–16057 RSC.
- X.-X. Yang, I. Issac, S. Lebedkin, M. Kühn, F. Weigend, D. Fenske, O. Fuhr and A. Eichhöfer, Red-luminescent biphosphine stabilized ‘Cu12S6’ cluster molecules, Chem. Commun., 2014, 50, 11043–11045 RSC.
- S. Dehnen, A. Schäfer, D. Fenske and R. Ahlrichs, New Sulfur- and Selenium-Bridged Copper Clusters; Ab Initio Calculations on [Cu2nSen(PH3)m] Clusters, Angew. Chem., Int. Ed. Engl., 1994, 33, 746–749 CrossRef.
- A. Eichhöfer, G. Buth, S. Lebedkin, M. Kühn and F. Weigend, Luminescence in Phosphine-Stabilized Copper Chalcogenide Cluster Molecules—A Comparative Study, Inorg. Chem., 2015, 54, 9413–9422 CrossRef.
- O. Fuhr, S. Dehnen and D. Fenske, Chalcogenide clusters of copper and silver from silylated chalcogenide sources, Chem. Soc. Rev., 2013, 42, 1871–1906 RSC.
- M. J. Trenerry, M. Kallumkal, E. Garcia L. and G. A. Bailey, CO2 Reduction Using Chalcogen-Modified Copper Materials and Nanoclusters: Progress and Challenges, ChemRxiv, 2025, preprint, DOI:10.26434/chemrxiv-2025-3mgg3.
- H. A. Fargher, T. J. Sherbow, M. M. Haley, D. W. Johnson and M. D. Pluth, C–H⋯S hydrogen bonding interactions, Chem. Soc. Rev., 2022, 51, 1454–1469 RSC.
- T. Chivers, D. D. Doxsee and R. W. Hilts, Preparation and phosphorus-31 and selenium-77 NMR spectra of platinum and palladium complexes of a P2N4Se2 ring, Inorg. Chem., 1993, 32, 3244–3249 CrossRef CAS.
- A. M. Polgar, F. Weigend, A. Zhang, M. J. Stillman and J. F. Corrigan, A N-Heterocyclic Carbene-Stabilized Coinage Metal-Chalcogenide Framework with Tunable Optical Properties, J. Am. Chem. Soc., 2017, 139, 14045–14048 CrossRef CAS PubMed.
- A. M. Polgar, A. Zhang, F. Mack, F. Weigend, S. Lebedkin, M. J. Stillman and J. F. Corrigan, Tuning the Metal/Chalcogen Composition in Copper(I)–Chalcogenide Clusters with Cyclic (Alkyl)(amino)carbene Ligands, Inorg. Chem., 2019, 58, 3338–3348 CrossRef CAS PubMed.
- S. Dehnen, A. Eichhöfer and D. Fenske, Chalcogen-Bridged Copper Clusters, Eur. J. Inorg. Chem., 2002, 279–317 CrossRef.
- A. D. Becke, Density-functional exchange-energy approximation with correct asymptotic behavior, Phys. Rev. A: At., Mol., Opt. Phys., 1988, 38, 3098–3100 CrossRef CAS PubMed.
- J. P. Perdew, Density-functional approximation for the correlation energy of the inhomogeneous electron gas, Phys. Rev. B: Condens. Matter Mater. Phys., 1986, 33, 8822–8824 CrossRef PubMed.
- F. Weigend and R. Ahlrichs, Balanced basis sets of split valence, triple zeta valence and quadruple zeta valence quality for H to Rn: Design and assessment of accuracy, Phys. Chem. Chem. Phys., 2005, 7, 3297–3305 RSC.
- E. D. Glendening, J. K. Badenhoop, A. E. Reed, J. E. Carpenter, J. A. Bohmann, C. M. Morales, P. Karafiloglou, C. R. Landis and F. Weinhold, NBO 7.0, Theoretical Chemistry Institute, University of Wisconsin, Madison, 2018. Search PubMed.
- B. Zheng, X.-D. Chen, S.-L. Zheng and R. H. Holm, Selenium as a Structural Surrogate of Sulfur: Template-Assisted Assembly of Five Types of Tungsten–Iron–Sulfur/Selenium Clusters and the Structural Fate of Chalcogenide Reactants, J. Am. Chem. Soc., 2012, 134, 6479–6490 CrossRef CAS PubMed.
- A. Saxena, W. Liyanage, J. Masud, S. Kapila and M. Nath, Selective electroreduction of CO2 to carbon-rich products with a simple binary copper selenide electrocatalyst, J. Mater. Chem. A, 2021, 9, 7150–7161 RSC.
- C. Guo, Y. Guo, Y. Shi, X. Lan, Y. Wang, Y. Yu and B. Zhang, Electrocatalytic Reduction of CO2 to Ethanol at Close to Theoretical Potential via Engineering Abundant Electron-Donating Cuδ+ Species, Angew. Chem., Int. Ed., 2022, 61, e202205909 CrossRef CAS PubMed.
- J. A. Kephart, B. S. Mitchell, A. Chirila, K. J. Anderton, D. Rogers, W. Kaminsky and A. Velian, Atomically Defined Nanopropeller Fe3Co6Se8(Ph2PNTol)6: Functional Model for the Electronic Metal–Support Interaction Effect and High Catalytic Activity for Carbodiimide Formation, J. Am. Chem. Soc., 2019, 141, 19605–19610 CAS.
- D. A. Reed, T. J. Hochuli, N. A. Gadjieva, S. He, R. A. Wiscons, A. K. Bartholomew, A. M. Champsaur, M. L. Steigerwald, X. Roy and C. Nuckolls, Controlling Ligand Coordination Spheres and Cluster Fusion in Superatoms, J. Am. Chem. Soc., 2022, 144, 306–313 CrossRef CAS PubMed.
- T. M. Bostelaar, A. C. Brown, A. Sridharan and D. L. M. Suess, A general method for metallocluster site-differentiation, Nat. Synth., 2023, 2, 740–748 CrossRef CAS.
- C. M. Crudden, J. H. Horton, I. I. Ebralidze, O. V. Zenkina, A. B. McLean, B. Drevniok, Z. She, H.-B. Kraatz, N. J. Mosey, T. Seki, E. C. Keske, J. D. Leake, A. Rousina-Webb and G. Wu, Ultra stable self-assembled monolayers of N-heterocyclic carbenes on gold, Nat. Chem., 2014, 6, 409–414 CrossRef CAS PubMed.
- D.-H. Nam, P. De Luna, A. Rosas-Hernández, A. Thevenon, F. Li, T. Agapie, J. C. Peters, O. Shekhah, M. Eddaoudi and E. H. Sargent, Molecular enhancement of heterogeneous CO2 reduction, Nat. Mater., 2020, 19, 266–276 CrossRef CAS PubMed.
- (a) M. J. Trenerry, E. Garcia L., A. Lovstedt, L. T. Way, J.-N. Boyn and G. A. Bailey, CCDC 2466271: Experimental Crystal Structure Determination, 2025, DOI:10.5517/ccdc.csd.cc2nsc4z; (b) M. J. Trenerry, E. Garcia L., A. Lovstedt, L. T. Way, J.-N. Boyn and G. A. Bailey, CCDC 2466272: Experimental Crystal Structure Determination, 2025, DOI:10.5517/ccdc.csd.cc2nsc50.
| This journal is © The Royal Society of Chemistry 2025 |






