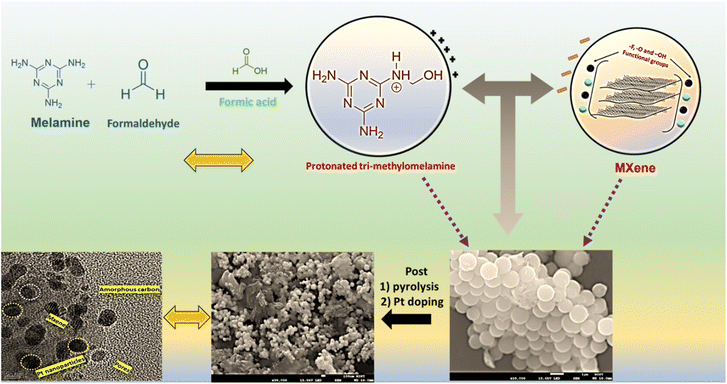
Template-based synthesis of porous and wrinkled Pt-supported MXenes for enhanced bifunctional electrocatalysis of the ORR and HER
Rupali S.
Mane
 a,
Higgins M.
Wilson
b,
Bhagyashree N.
Patil
a,
Sang Joon
Lee
a,
Higgins M.
Wilson
b,
Bhagyashree N.
Patil
a,
Sang Joon
Lee
 b and
Neetu
Jha
b and
Neetu
Jha
 *a
*a
aDepartment of Physics, Institute of Chemical Technology Mumbai, Nathalal Parekh Marg, Mumbai-400019, India. E-mail: nr.jha@ictmumbai.edu.in
bDepartment of Mechanical Engineering, Pohang University of Science and Technology, 77 Cheongam-Ro, Nam-Gu, Pohang-37673, Gyeongbuk, Republic of Korea
First published on 28th August 2025
Abstract
This study proposes a novel strategy to develop a platinum-supported, highly porous MXene (Pt/PMx) catalyst via a “template-based transformation” approach and evaluates its bifunctional electrocatalytic properties for the oxygen reduction reaction (ORR) and hydrogen evolution reaction (HER). The strategy introduces defects into nitrogen MXene sheets, enhancing their surface area and porosity. The synthesized PMx effectively mitigates the restacking of the MXene sheets and facilitates the uniform dispersion of the ultrasmall platinum nanoparticles across their surface. The Pt/PMx catalyst exhibits excellent ORR activity, achieving an onset potential (Eon) of 0.93 V (Eon for Pt/C = 0.94 V) in an acidic medium and 0.94 V (Eon for Pt/C = 0.92 V) in an alkaline medium, along with superior chronoamperometric stability for 40![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 s. Comprehensive evaluation of intrinsic parameters, including active surface area, mass activity, CO poisoning tolerance, and durability, via accelerated degradation tests confirms the robustness of the electrocatalyst support. The significant interaction between the Pt nanoparticles and the support enhances H+ adsorption and charge transport and, thereafter, HER activity, which is attributed to the improved porosity and conductivity of the support. The strong interaction between Pt and PMx was found to be the key for excellent stability towards the ORR (20
000 s. Comprehensive evaluation of intrinsic parameters, including active surface area, mass activity, CO poisoning tolerance, and durability, via accelerated degradation tests confirms the robustness of the electrocatalyst support. The significant interaction between the Pt nanoparticles and the support enhances H+ adsorption and charge transport and, thereafter, HER activity, which is attributed to the improved porosity and conductivity of the support. The strong interaction between Pt and PMx was found to be the key for excellent stability towards the ORR (20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 potential cycles) and HER (∼40 h). This synthesis strategy provides a unique and novel pathway for designing highly porous, conductive, and hydrophilic Pt-based catalyst supports for improved ORR and HER performance.
000 potential cycles) and HER (∼40 h). This synthesis strategy provides a unique and novel pathway for designing highly porous, conductive, and hydrophilic Pt-based catalyst supports for improved ORR and HER performance.
Introduction
The dwindling of energy resources such as fossil fuels has led to an insalubrious eco-balance. Thus, other sustainable technologies, such as fuel cells (FCs), water electrolyzers, and metal–air batteries, are receiving considerable attention.1,2 Accordingly, global efforts are being made to commercialise these technologies. However, these electrochemical systems face some undeniable challenges even when employing commercial electrocatalytically active materials such as Pt/C, which encounters sluggish kinetics in vital electrochemical reactions such as the oxygen reduction reaction (ORR) and hydrogen evolution reaction (HER). Additionally, Pt/C catalysts encounter issues such as low stability, CO poisoning, and dissolution and agglomeration of Pt when subjected to practical use in long-run processes.3Vulcan XC72R carbon, a support used in commercial catalysts,4 has been reported to undergo degradation and Ostwald ripening, which are known for causing the aforementioned issues.5,6 Considering these hurdles, advancements are being made in replacing carbon as the support in Pt/C with an alternative conducting, non-degradable, and enduring support. In pursuit of this direction, several types of carbon materials, including carbon nanotubes, nitrogen-doped carbon supports, and graphene, have been employed to investigate the durability and anti-poisoning ability of catalysts.7–9 Despite their excellent properties, such as conductivity, conducive structure, and surface area, these materials fail when they are subjected to practical operating conditions such as high acidity, extreme temperatures, and high humidity. Hence, highly conducting, corrosion-resistant robust materials such as titanium carbides, i.e. MXenes, can be considered a potential candidate as a support for Pt. MXenes, a 2D carbide family material, can be synthesized by adopting the etching process of Mn+1AXn (n = 1, 2 or 3; M = transition metal; A = 13 or 14 group elements, e.g. aluminum and carbon; and X = carbon or/and nitrogen). MXenes have been widely utilized due to their significant properties such as metal end structure, high stability and conductivity.10 Owing to their exceptional properties, several studies have been reported on MXenes as supports for ORR and other applications.11–13 However, despite these properties, MXenes demonstrate low catalytic activity as electrocatalysts due to their scarcity of active sites and tendency of their sheets to agglomerate or restack. These factors can be effectively addressed by tailoring their electronic structure, tuning morphology, doping transition metal atoms, etc.
Guo et al. designed MXene nanofibers using a hydrolyzation process, followed by etching. These nanofibers showed a low overpotential towards HER application due to their high surface area and exposed active sites due to their tuned morphology.14 Singh et al. synthesized an ultrathin single-layer MXene sheet by intercalating tetrapropylammonium hydroxide (TPAOH) into the pristine MXene and studied its ORR kinetics. The enhanced catalytic activity was attributed to its improved electronic conductivity.15 It was observed that doping a transition metal into MXene alters the Gibbs free energy of the overall catalyst, subsequently leading to an improved catalytic performance. Fang et al. grew MoS2 on MXene via the hydrothermal technique and formed a nanoflower-type morphology. Benefiting from this 3D architecture, the catalyst showed enhanced HER activity.16 Similarly, Shahid et al. reported the synthesis of a 3D MXene-reduced graphene oxide composite with NiSe2 for water splitting application. MXene, when optimized structurally or doped with other entities, exhibits an excellent electrocatalytic performance owing to its improved electrical conductivity and electrolyte ion transfer rate.17 However, despite the numerous studies on non-noble metal-doped MXenes as electrocatalysts by tailoring various factors such as controlled surface area, structure, and morphology for energy conversion applications, their catalytic activity has been found to be nominal compared to Pt. In this direction, Wei et al. first studied the synergy between Pt NPs and MXene via first-principle calculations. The results displayed that MXene not only acts as a conducting support but also provides high structural stability and modulates the overall electronic structure, favouring the electrocatalytic activity.18 Later, Chen et al. reported the synthesis of TiC-supported Pt NPs (Pt/TiC) by thermal treatment. Here, Pt/TiC was synthesized by calcining a mixture of Pt precursor and TiC in an air and H2 atmosphere, respectively. The results demonstrated superior ORR activity when the catalyst was calcined in an H2 atmosphere. This was attributed to the well-dispersed, ultrasmall size (∼2.2 nm) NPs of Pt on TiC. However, when the mixture was calcined in air, the size of the Pt NPs was found to be increased to 6.7 nm, given that TiC tends to oxidize under the latter condition, leading to the formation of PtOx species and preventing the uniform dispersion of Pt NPs. This further explains the reduced ORR activity obtained under the latter condition.19 Thus, to tackle these issues, Wei et al. synthesized Pt NPs supported on Ti3C2X2 using the reflux method. Here, the addition of the Pt precursor and MXene took place under an inert atmosphere (N2), leading to the formation of ultrasmall Pt NPs with a size of 3–4 nm on Ti3C2X2. The results demonstrated the enhanced durability and catalytic activity of the as-synthesized catalyst. Here, a minimal increase in the size of the Pt NPs was observed in the post-durability study.8 Enhancing the interaction between the metal support to prevent the degradation of platinum and corrosion of the support has emerged as another crucial strategy to augment the durability of catalysts. In this regard, Hrnjić et al. reported a synergistic relationship between the conductive support, i.e. MXene, and Pt NPs. It was observed that the strong interaction between the metal and Pt induces strain in the structure and controls the size of the NPs, thereby enhancing the ORR activity.20 Similarly, Jackson et al. developed boron carbide-supported Pt for enhanced ORR activity. They found that electronic modulation between the support and Pt NPs controls the ORR activity and stability. This is related to the charge transfer/redistribution between the support and metal, which modulates the Fermi energy level, and hence the activity of the catalyst.21 Xiu et al. reported a multilayered hollow structure supporting ultrafine Pt NPs as an effective catalyst for seawater splitting. Herein, the synergy between the catalyst interface and ultrafine Pt improves the charge transport, descriptor ion adsorption/activation and mass exchange, which effectively enhance the catalyst activity.22
The common problem in these approaches is the agglomeration/restacking and oxidation of the MXene sheets during the synthesis and/or post-electrochemical study. This leads to an increase in the size of the Pt NPs, hampering the durability and activity of the catalyst. Thus, to address this issue effectively, we explore the utilisation of a highly porous MXene sheet as a catalyst support. This type of structure is known to achieve the uniform distribution of Pt NPs, which also helps with efficient mass transport owing to its hierarchical pore structure. Bridging the above-mentioned two significant strategies, i.e., controlled morphology and tuned porosity with simultaneous doping of MXene with noble/non-noble metals, is challenging. Also, 3D MXenes show low porosity and minimal adhesion with the doped species.23 Cui et al. demonstrated the synthesis of crumpled nitrogen-doped MXene wrapped with nitrogen-doped amorphous carbon (denoted as NMXC) through a melamine-based template-guided microsphere route for modulating the surface chemistry and expanding the inter-lamellar spacing of the MXene. This strategy enlarges the work function, reducing the electrochemical oxidation, and hence improving the stability of the material. This group studied the performance of zinc-based hybrid supercapacitors using this electrode material.
Herein, we report the (2–3–2)D transformative strategy that first transforms two-dimensional sheet-like MXene into a 3-dimensional spherical morphology, followed by bursting of this MXene sphere to again form 2-D highly porous and crumpled MXene nanosheets. This strategy not only creates defects and increases the porosity of the catalyst but also addresses the major restacking and oxidation issues of MXene sheets. Post-pyrolysis, a hydrothermal route was employed to disperse Pt NPs on the highly poriferous Mx sheet (PMx). The Pt-supported highly porous MXene in the catalyst gives rise to excellent ORR and HER catalytic activity, along with excellent durability. Furthermore, a comprehensive examination of the intrinsic parameters, such as active surface area and mass activities, was conducted through CO poisoning and stripping studies, in conjunction with an accelerated durability test (ADT). It was observed that this strategy paves the way for the design and optimization of Pt NP-decorated highly conducting supports for energy conversion applications.
Result and discussion
The synthesis process involves the polymerisation of the melamine-formaldehyde (MF) adduct in formic acid.24 The acidic condition was found to be susceptible to the protonation of MF, making it more electrophilic towards the reaction25 and forming the polymer chain of tri-methylomelamine (M–NHCH2OH, where M represents the core ring part of melamine).26 In the succeeding phase, the MXene sheets containing negatively charged functional groups (–F, –O, and –OH) are electrostatically attracted to the protonated MF complex, leading to colloidal coagulation. The resulting pure white supernatant was washed multiple times with deionized water, and subsequently dried, yielding perfect spherical 3D MXene spheres (MxSP).27–30 This spherical structure transforms into highly porous and wrinkled MXene (PMx) post-pyrolysis at 550 °C, as shown in Scheme 1. The direct mixing of melamine with MXene does not form the required porous structure but limits the mass transport diffusivity. Alternatively, the protonated structure of the MF template acts as a spacer between the MXene sheets. After confirming the transformation from a 3D spherical structure to 2D porous nature in the Mx sheet (PMx), Pt NPs were deposited hydrothermally to obtain the hybrid active electrocatalyst i.e. Pt/PMx. This approach of 3D to 2D transformation not only addresses the aggregation/restacking issue of the MXene layers but also avoids the oxidation of the MXene sheets and provides highly porous networks with complementary N as active sites for the ORR kinetics (detailed ORR mechanism is shown in Fig. S1). This porous network with a wrinkled morphology was found to be conducive for dispersing Pt nanoparticles uniformly on the MXene layers, further improving the kinetics of the hybrid catalyst.Fig. 1 illustrates a schematic representation of the catalyst and corresponding FE-SEM analysis. As depicted in Fig. 1a, pristine MXene shows a well-exfoliated bundle of thin lamellar sheet-like structures due to the delamination of the Al layer post-HF etching. Fig. 1b corresponds to the FE-SEM image of 3D MXene. Here, following the condensation process, the protonated MF spheres, which are electrostatically associated with the 2D pristine MXene sheets, exhibit a spherical morphology (∼700 nm to 800 nm), with remnants of MXene sheets present on the their surface. Briefly, the MXene lamellar layers get separated and adsorb on MF, resulting in a 3D spherical architecture of MXene with a size of a few nm. Elemental mapping further confirms the presence of Ti, N, C, and O (Fig. S2a and b). The absence of –F species further confirms the electrostatic interaction between the protonated MF and negatively charged MXene. In the subsequent pyrolysis step, melamine in the 3D structure bursts at 550 °C and forms a 2D-N doped highly porous structure (PMx), which effectively protects the MXene layers from restacking (Fig. 1c). To confirm the wrinkled nature and purity of the material, elemental mapping was performed (Fig. S2c and d). The wrinkled structure of the obtained PMx was found to prevent the restacking of the MXene layers and improve the surface area of the material. Additionally, the presence of N species was found to boost the catalytic activity towards ORR and HER by providing active sites during the electrochemical reaction. The successful deposition of Pt nanoparticles (NPs) on the highly porous PMx sheets (Pt/PMx) was confirmed by the FE-SEM image shown in Fig. 1d, along with the corresponding elemental mapping (Fig. 1e–g), which clearly illustrates the uniform distribution of Pt, Ti, C, N, and O. To quantitatively evaluate the elemental composition, energy-dispersive X-ray spectroscopy (EDAX) analysis was conducted (Fig. S2e).
It was found that the N species present in PMx greatly enhance the surface properties of MXene, including its electron distribution, conductivity, and surface hydrophilicity. This was further confirmed by performing contact angle measurement on Pt/PMx, as shown in Fig. S3a. The obtained contact angle for the hybrid material in aqueous medium and the electrolytes used for electrochemical testing illustrates values of less than 90°, affirming the hydrophilicity of the composite material. Also, the conductivity of the electrocatalyst can be confirmed by electrochemical impedance spectroscopy, as depicted by electrochemical characterisation.
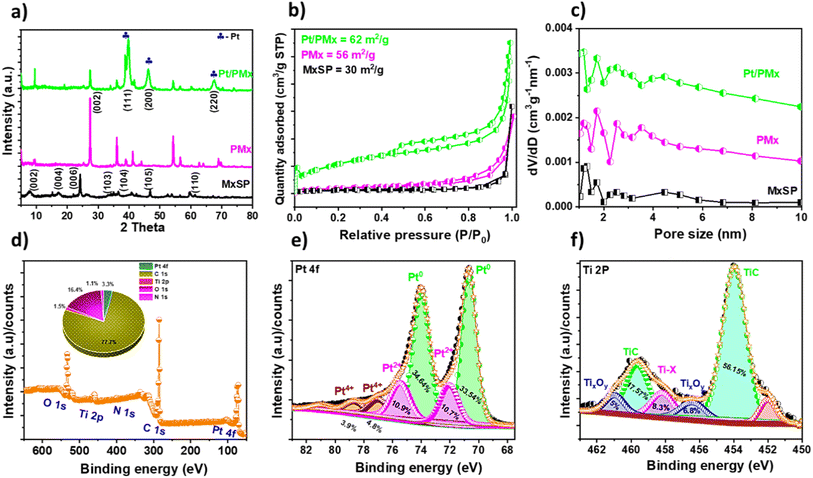
The XRD diffractograms of MxSP, PMx, and Pt/PMx are shown in Fig. 2a. The diffraction pattern of MXene (Fig. S3b) depicts the presence of characteristic peaks in comparison with the pristine MAX phase, i.e. Ti3AlC2 (JCPDS 52-0875). This is due to the HF treatment employed to obtain MXene from MAX. The slight shift in the peak at 9.8° towards a lower angle of 9.3° was observed predominantly for the (002)31 peaks of MXene owing to the increased d-spacing (dMAX – 9.1 Å to dMXene – 9.45 Å) as a result of the exfoliation of the sheets post-delamination. An analogous XRD pattern was obtained for MxSP except for the peak at 9.4°, which corresponds to the (002) plane and shows a conspicuous shift towards a lower angle (at 7.9°). This is mainly attributed to the sheet bending and random orientation of post-doping MXene onto MF.23 As shown in Fig. 2 and S3c, the XRD pattern of the PMx sample exhibits several low-intensity peaks at approximately 9.5°, 17.2°, and 25.3°, which correspond to the (002), (004), and (006) planes of MXene, respectively, indicating that the 3D MXene spheres retain the partial structural characteristics of MXene even after pyrolysis.32–34 The slight shift in these peaks toward higher 2θ values suggests a decrease in d-spacing, which is attributed to the 3D structural disintegration during thermal treatment. In addition, the appearance of high-intensity peaks at higher angles such as at 25.9°, 36°, and 55° indicates the partial formation of rutile TiO2, likely due to the oxidation of the surface functional groups (e.g., OH and –O) present on MXene, even under an inert atmosphere.33 Specifically, the peak at 27.3° is assigned to the (002) plane of carbon, which corresponds to the d-spacing of approximately 3.4 Å. This shift in d-spacing is primarily attributed to pyrolysis-induced structural rearrangements, which lead to the development of a porous carbonaceous matrix surrounding the MXene layers, thereby forming a highly porous 2D partially amorphous architecture.35,36 Furthermore, the XRD pattern of Pt/PMx validates the presence of sharp peaks at 40.2°, 46.4°, and 67.9° (JCPDS no. 01-1194).23,37,38 These peaks are ascribed to the (111), (200), and (220) planes, further confirming the successful doping of FCC-structured Pt NPs onto PMx.39
The surface area and pore size measurement of the prepared catalysts were analysed by using nitrogen adsorption–desorption BET isotherms and Barrett–Joyner–Halenda (BJH) measurements. As illustrated in Fig. 2b, the highest value of the surface area was obtained for Pt/PMx (62 m2 g−1) with a type IV isotherm. This could be attributed to the poriferous nature of the PMx sheets and Pt NPs during the hydrothermal process, which exposes more pores and leads to an increase in the high surface area. MxSP and PMx show mixed isotherms phases of type II and IV isotherms with BET values of 30 and 56 m2 g−1, respectively. This corroborates the presence of a multi-porous network, confirming the presence of both macroporosity and mesopores, with the presence of a small amount of micropores.36,40 This was further confirmed by the BJH study, as shown in Fig. 2c. The pore size of MxSP is in the range of 1 nm to 3.5 nm, implying the presence of both micro and mesopores in the material. Alternatively, PMx exhibits the presence of different pores with diameters of 1 nm, 2 nm, 2.5 nm, and 3.7 nm, ranging from micropores to various-size mesopores. This is mainly attributed to the pyrolysis treatment, which led to the conversion of the 3D MXene spheres to 2D highly porous sheets. This confirms that converting 3D to wrinkled MXene sheets reduces the face-to-face restacking, enhancing its porosity and surface area (Fig. S4a). The results were found to be consistent with the composite (Pt/PMx) material, demonstrating a negligible change in the porosity of the hybrid material.
The chemical compositions and bonding configuration were further elucidated by X-ray photoelectron spectroscopy (XPS). A statistical comparison of the survey spectra (Fig. S4b) for MXene and Pt/PMx reveals a higher proportion of C 1s moieties in Pt/PMx than in MXene, which can be attributed to the pyrolysis of MxSP, during which the melamine template spheres burst into sheets, resulting in a highly porous, hydrophilic active PMx framework. The Ti 2p spectra of MXene exhibits a significantly intense peak, while the survey spectrum of the composite reveals a minimal peak, indicating that a substantial portion of the MXene surface has been transformed and utilized during the composite formation process. Additionally, the absence of F 1s spectra in the composite affirms the reaction mechanism, where the negatively charged functional groups (–F, –O, and –OH) are electrostatically attracted to the protonated MF complex, thereafter forming PMx.41 Furthermore, the noticeable shift in Ti 2p towards a higher BE indicates electron transfer between MXene and Pt, confirming a strong metal support interaction (Fig. S4b, inset).42Fig. 2d depicts the survey spectrum of the hybrid catalyst, i.e. Pt/PMx. The obtained spectrum illustrates the presence of C, N, O, Pt, and Ti elements, without any impurities. It also confirms the doping of N in the MXene sheets with the most intense peak for Npyro, as explained by the highly resolved N band shown in Fig. S4c. The deconvoluted N 1s spectra observed for Pt/PMx confirm that N moieties (originated from MF template) successfully diffused in the composite material, indicating the metal support interaction and found to be conducive for ORR and HER. As depicted, the spectra exhibit five major N-species, namely pyridinic N (Npyri), pyrrolic N (Npyro), metal–N (M–N), graphitic N (Ngra), and N oxide (Noxide), with the corresponding binding energies of ∼397.9 eV (15.5 at%), 398.8 eV (33.1 at%), 399.8 eV (33.86 at%), 400.63 (13.02 at%) eV, and 401.5 eV (4.48 at%), respectively.2,43 The presence of an intense peak with high area under the curve obtained for pyridinic and pyrrolic N-moieties is found to be conductive for ORR and HER, as reported in most previous reports.2,43 The other high percentage peak obtained for M–N indicates chemical adsorption between Pt and PMx. The deconvoluted Pt 4f spectra (Fig. 2e) confirm the existence of Pt0 at 70.68 eV (34.64 at%) and 74 eV (33.56 at%), Pt2+ at 72.04 eV (10.92 at%) and 75.44 eV (10.79 at%), and Pt4+ at 76.97 eV (4.88 at%) and 78.67 eV (3.58 at%), respectively.10,44 The highly intense peak with the maximum atomic percentage corresponds to metallic Pt, i.e. Pt0, which implies less coverage of poisonous species (oxides). The high percentage of Pt2+ followed by Pt4+ is mainly attributed to Pt(OH)2, where the M–OH bond confirms the successful deposition of Pt NPs onto the porous MXene network via chemisorption, and subsequently strong metal–support interaction. Furthermore, the Pt 4f spectra, particularly for Pt0 and Pt2+, exhibit a shift towards a lower binding energy relative to the conventional Pt species.45 This is mainly attributed to the electron sharing between PMx and Pt, which modulates the d-band of Pt/PMx. The altered d-band level was determined to be beneficial for electrocatalytic activity. This confirms the significant synergistic effect between the support (PMx) and Pt nanoparticles.46,47 To further substantiate this, the high-resolution valence band X-ray photoelectron spectroscopy (VB-XPS) spectrum is correlated with the adsorption strength of the intermediate, as shown in Fig. S4d. This result illustrates a downshift in the VB, i.e., shifting in d-band, not only due to Pt-doping but also due to the reaction process, which subsequently adjusts the Fermi energy and the d-band in the composite.46,48–50 The deconvoluted Ti 2p spectra (as shown in Fig. 2f) correspond to the intense TiC peaks located at 453.9 eV and 459.7 eV with the atomic percentages of 56.15 at% and 17.57 at%, respectively.51 The peak located at 458.16 eV is attributed to the Ti–X species, while the peaks located at 460.9 and 456.4 eV show the presence of TiXOY species. This could be attributed to the surface functional groups, mainly (–OH) present in MXene. Therefore, it can be affirmed that MXene plays a vital role in enhancing the physical properties of the catalyst such as surface area and pore size and modulating its electronic structure by optimizing the d-band center of Pt with the help of another transition metal, i.e. Ti.
The morphology, porosity, and size of the MxSP, PMx, and Pt/PMx particles were examined by HR-TEM analysis. Fig. 3a and b show a 3D MxSP sphere (inset) with the MXene layer acquiring the shape of MF. The obtained SAED (Fig. 3c) pattern illustrates the distribution of nanoparticles corresponding to metallic Ti in MXene, which is different from the hexagonal pattern observed for the bare MXene (Fig. S5a–c).52 The porous network obtained post-pyrolysis of MxSP, i.e. PMx, can evidently be observed in Fig. 3d and e. The respective SAED pattern (Fig. 3f) confirms the equally distributed Ti NPs. Fig. 3g–i confirm the uniform dispersion of Pt NPs onto PMx. It was found that the wrinkled sheets help in enhancing the adsorption of Pt on the surface of PMx, further improving the descriptor adhesion and smooth mass transfer during catalytic reactions.41 The inset of Fig. 3g and S5d further confirms the uniform distribution of Pt NPs on the wrinkled and highly porous PMx. The corresponding TEM images also indicate the formation of an amorphous structure around Pt NPs with a d spacing of 0.19 nm and 0.23 nm for the (200) and (111) planes, respectively. Post-deposition, the Pt NPs were found to be embedded in PMx uniformly with an average particle size of ∼4 nm. The corresponding SAED pattern represents a polycrystalline nature, confirming the same (Fig. 3i).
 | ||
| Fig. 3 HR-TEM analysis alongside the respective SAED patterns. (a–c) MxSP, (d–f) PMx and (g–i) Pt/PMx. | ||
The electrochemical activity of the catalyst for ORR
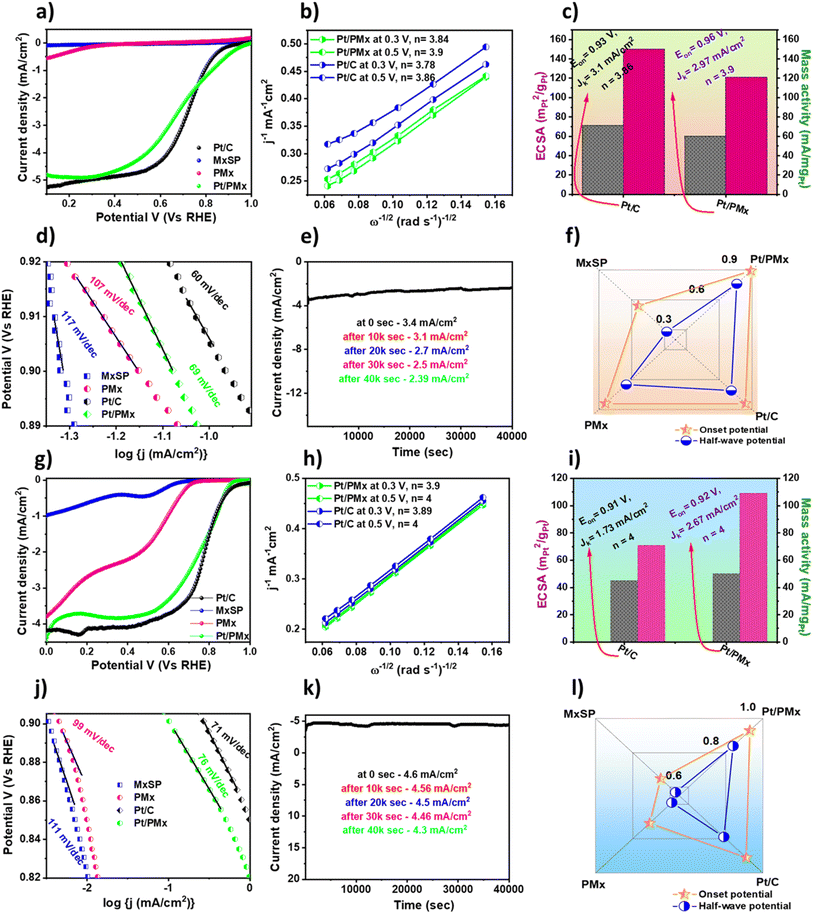
The electrochemical evaluation of the synthesized catalysts for ORR activity was performed using a three-electrode rotating disk electrode set-up in both acidic and alkaline media and the data is compared with commercial Pt/C (20 wt% Pt loading). Prior to the experiment, the electrolyte was purged with N2 for about 30 min to remove all the dissolved oxygen intermediates. Fig. 4 illustrates the electrochemical properties of the synthesized catalysts in O2-saturated 0.5 M H2SO4. A characteristic trend was observed when the catalyst was subjected to the hydrodynamic polarisation study. Fig. 4a depicts the typical LSV curve obtained for the as-synthesized catalysts. The LSV curve depicts a high onset potential of 0.96 V vs. RHE for Pt/PMx compared to MxSP (0.46 V) < PMx (0.82 V) < Pt/C (0.93 V). This is mainly attributed to the modified surface chemistry and strong interaction between the doped Pt NPs on PMx compared to Pt/C, where the available multi-porous nature of the catalyst and hydrophilicity lead to a favourable ORR pathway under acidic conditions. The porous matrix serves as a mass transport channel that allows enough exposure for oxygen as a descriptor in the catalytic reaction. To confirm this, the ORR performance of Pt-doped MxSP (Pt/MxSP) was compared with that of Pt/PMx (Fig. S6). Although LSV illustrates the conducive behaviour of Pt/MxSP towards ORR activity, the obtained performance compared with Pt/PMx illustrates a more positive onset alongside excellent current density. This reveals the role of PMx in the strong interaction of the metal-doped carbon support with the active centers. The diffusion-controlled nature of the prepared catalysts was studied by recording polarisation curves at different rotation speeds; this reveals the kinetics of the catalyst. As illustrated in Fig. S6, the obtained current density was found to increase with an increase in rotation speed owing to the hydrodynamic nature of the electrolyte solution, which increases with speed. The ORR kinetics were further analyzed by calculating the electron transfer number (n) at two different potentials in the limiting region. The results illustrate the absolute 4e− pathways for both Pt/PMx and Pt/C with n values of 3.9 and 3.86 respectively (Fig. 4b). Additionally, other parameters such as half-wave potential, kinetic current density (Jk), Tafel slope, ECSA, and mass activity were extrapolated from Fig. 4c and summarized in Table 1. The Tafel slope obtained for the as-synthesized catalyst is illustrated in Fig. 4d, where first-order kinetics was observed with Tafel slopes of 60 mV dec−1 (Pt/PMx) and 69 mV dec−1 (Pt/C), which indicates a smooth mass transport rate with boosted rate-determining step. In FCs, the stability and durability of the catalyst have been found to play a vital role in the commercialisation of electrocatalysts. Therefore, to emphasize the stability of the prepared Pt/PMx, the catalyst was subjected to a 40![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 s chronoamperometric stability test and the changes in current density were measured, as shown in Fig. 4e. The results depict a small decrease in the current density from 3.4 mA cm−2 (initial) to ∼2.4 mA cm−2, demonstrating the good electrochemical stability of the catalyst. This is attributed to its highly conducting and porous support originating from PMx, preventing degradation and poisoning of the catalyst, which confirms the activity of PMx towards ORR. The active nature of the support was further observed when the half-wave and onset potential of the pristine catalyst were compared with that of the hybrid catalyst (Fig. 4f). Regardless of the fact that the onset of Pt/PMx was found to be significantly higher than that of Pt/C, the MA and ECSA were found to be higher for Pt/C. Another objective of this work was to analyse the durability and poisoning resistance of Pt/PMx in comparison with Pt/C. This was tested by subjecting the catalyst to a long-term durability study (20
000 s chronoamperometric stability test and the changes in current density were measured, as shown in Fig. 4e. The results depict a small decrease in the current density from 3.4 mA cm−2 (initial) to ∼2.4 mA cm−2, demonstrating the good electrochemical stability of the catalyst. This is attributed to its highly conducting and porous support originating from PMx, preventing degradation and poisoning of the catalyst, which confirms the activity of PMx towards ORR. The active nature of the support was further observed when the half-wave and onset potential of the pristine catalyst were compared with that of the hybrid catalyst (Fig. 4f). Regardless of the fact that the onset of Pt/PMx was found to be significantly higher than that of Pt/C, the MA and ECSA were found to be higher for Pt/C. Another objective of this work was to analyse the durability and poisoning resistance of Pt/PMx in comparison with Pt/C. This was tested by subjecting the catalyst to a long-term durability study (20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 CV cycles) and anti-poisoning study against CO, as explained in a later section. Additionally, the comprehensive intrinsic findings post-poisoning study further affirm the significant role of PMx as a support.
000 CV cycles) and anti-poisoning study against CO, as explained in a later section. Additionally, the comprehensive intrinsic findings post-poisoning study further affirm the significant role of PMx as a support.
| Before | Post-ADT (20k cycles) | Post-stripping | |||||||
|---|---|---|---|---|---|---|---|---|---|
| E on (V) | ECSA (mPt2 gPt−1) | MA (mA μgPt−1) | E on (V) | ECSA (mPt2 gPt−1) | MA (mA μgPt−1) | E on (V) | ECSA (mPt2 gPt−1) | MA (mA μgPt−1) | |
| Acidic medium (0.5 M H 2 SO 4 ) | |||||||||
| Pt/C | 0.93 | 63 | 150 | 0.91 | 40 | 96 | 0.89 | 48 | 114 |
| Pt/PMx | 0.96 | 60 | 121 | 0.93 | 47 | 84 | 0.94 | 53 | 97 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
|||||||||
| Basic medium (0.1 M KOH) | |||||||||
| Pt/C | 0.91 | 45 | 71 | 0.89 | 39 | 59 | 0.89 | 31 | 50 |
| Pt/PMx | 0.92 | 50 | 109 | 0.90 | 43 | 84 | 0.89 | 43 | 84 |
Based on the excellent electrochemical activity of the catalyst in acidic medium, the catalyst was investigated for its ORR activity in an alkaline medium (0.1 M KOH). Similar protocols were followed for the measurements under alkaline conditions. As shown in Fig. 4g, Pt/PMx follows the same trend with a sharp cathodic peak at the most positive potential. As depicted in Fig. 4i, Pt/PMx exhibits the onset potential of 0.92 V (Pt/C – 0.92 V) with the current density (j) of 4.35 mA cm−2 and half-wave potential (E1/2) of 0.825 V (for Pt/C, E1/2 – 0.80 V and j – 4.2 mA cm−2). As elucidated, the improved activity of Pt/PMx can be attributed to the poriferous nature of PMx, along with its high conductivity and hydrophilicity. To further elucidate the role of PMx compared to MxSP, the ORR activity of Pt/MxSP was evaluated alongside Pt/PMx. Although Pt/MxSP exhibited a substantial electrocatalytic performance, Pt/PMx demonstrated a more favourable onset potential and higher limiting current density, highlighting its superior ORR activity (Fig. S8). This is mainly attributed to the porosity and high surface area of PMx compared to MxSP. Following this, the ECSA obtained for Pt/PMx was found to be higher at 50 m2 gPt−1 than for Pt/C (45 m2 gPt−1). For further perception, the LSV curve of the prepared catalyst was obtained at different rotation speeds (400–1600 rpm). As expected, the current density was enhanced with an increase in the rotation speed, as shown in Fig. S8. Correspondingly, the K–L plot of the catalyst (Fig. 4h) was obtained, which exhibits good linearity, revealing its first-order kinetics. The n value for Pt/PMx is 3.9 and 4 at potentials of 0.3 V and 0.5 V, respectively. However, the n value for Pt/C at the same potentials was found to be 3.89 and 4, respectively. The Tafel slope (Fig. 4j) for Pt/PMx (71 mV dec−1) was found to be in the same range as that of Pt/C (76 mV dec−1), illustrating the faster kinetics of the catalyst in comparison to Pt/C.53 Further, the chronoamperometric stability was tested to assess the stability of the catalyst in an alkaline environment. Fig. 4k exhibits a negligible loss in current density from 4.6 mA cm−2 to 4.3 mA cm−2, even after 40![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 s in the stability tests. This confirms that PMx is a concrete support and conducive to ORR activity. This was further observed in Fig. 4l, where the half-wave and onset potential of the pristine materials are compared with the hybrid catalyst.
000 s in the stability tests. This confirms that PMx is a concrete support and conducive to ORR activity. This was further observed in Fig. 4l, where the half-wave and onset potential of the pristine materials are compared with the hybrid catalyst.
Intrinsic behaviour of the catalyst towards CO poisoning and stability
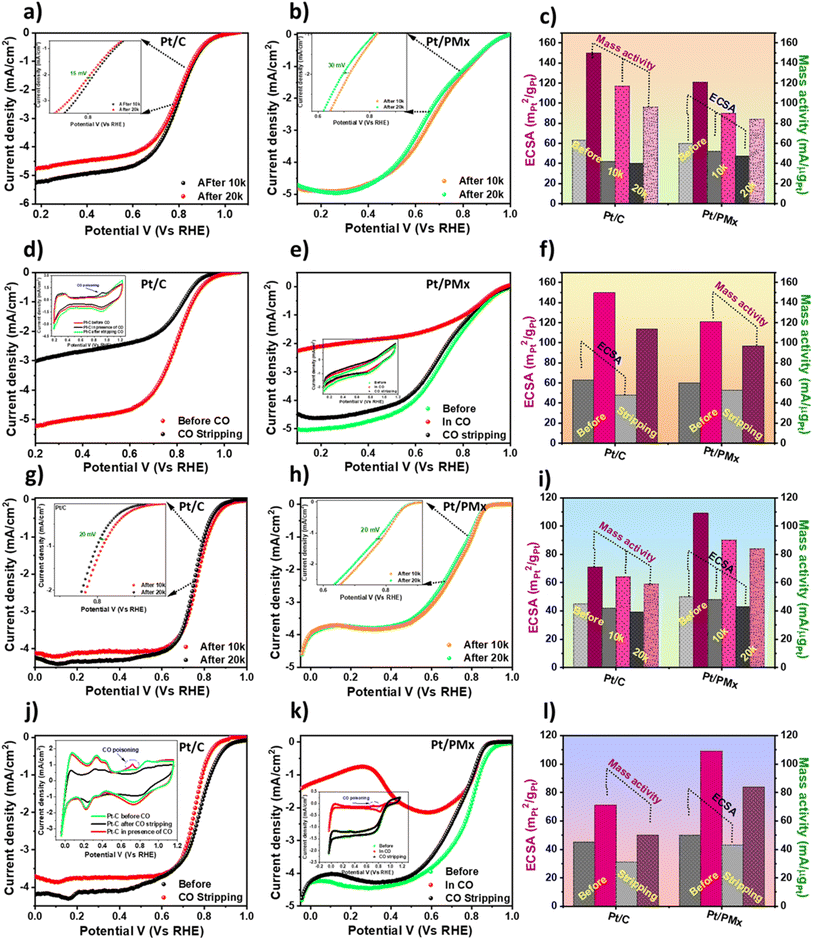
In addition to the electrochemical study, the catalyst was further subjected to a thorough durability and CO poisoning/stripping study. As is well known in the literature, Pt/C depicts enhanced activity given its stronger rate-determining step, and hence the strong adsorption of the descriptors.54 However, with the prolonged test, it has also been found that Pt/C tends to leach out/aggregate or alter the activity. Thus, the main objective of this study is to boost the durability and poisoning resistance of Pt-based materials. Therefore, despite knowing the facts and observed results, although Pt/C exhibits excellent MA and ECSA, it is a major concern to observe if it remains intact post-durability test. Hence, we performed a durability study and compared the intrinsic properties of the as-synthesized catalyst and Pt/C (Fig. 5a and b). An accelerated durability test (ADT) was performed by recording 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 CV cycles in an active region, i.e. with a potential window of 0.6 V to 1 V (vs. RHE). Fig. 5c represents the data post-durability study, where it can be observed that LSV scarcely shows any detrimental effect on Eon, current density, MA, and ECSA, demonstrating the excellent stability of the catalyst. The findings suggest that even though the Pt/C catalyst possess a higher MA and ECSA, the activity of the commercial catalyst decreased by about two-times more than that of Pt/PMx after ADT. This could be attributed to the poriferous nature and higher stability of PMx, which inhibits leaching, aggregation, and loss of active centers despite the number of cycles.
000 CV cycles in an active region, i.e. with a potential window of 0.6 V to 1 V (vs. RHE). Fig. 5c represents the data post-durability study, where it can be observed that LSV scarcely shows any detrimental effect on Eon, current density, MA, and ECSA, demonstrating the excellent stability of the catalyst. The findings suggest that even though the Pt/C catalyst possess a higher MA and ECSA, the activity of the commercial catalyst decreased by about two-times more than that of Pt/PMx after ADT. This could be attributed to the poriferous nature and higher stability of PMx, which inhibits leaching, aggregation, and loss of active centers despite the number of cycles.
As reported in various reports in the literature, the ORR mechanism of Pt-based and even Pt-free electrocatalysts tends to change in basic medium,44,54,55 depending on several factors such as the interaction of hydroxyl ions and contribution of active centers such as C–N, metal–N, and Pt centers. This study reflects the activity obtained for Pt/PMx compared to Pt/C.56 As shown in Fig. 5g and h, Pt/PMx illustrates a higher MA and ECSA than Pt/C. This could be attributed to the conductivity and hydrophilicity of the substrate. Additionally, the in situ N-doping originating from MF pyrolysis could also contribute to the enhanced activity of the catalyst. Then, the impact of ADT on the study in basic medium was determined, as represented in Fig. 5i. As expected, the results demonstrate barely any shift in the onset potential, current density, and MA. This further confirms the excellent contribution of the support towards the electrocatalytic activity of the catalyst.
Following the enhanced durability of the catalyst, the poisoning resistivity of the catalyst was studied by CO stripping. The CV and LSV curves were recorded in acidic electrolyte before purging (Fig. 5d and e and inset), post CO saturation in the electrolyte, and after stripping (by purging N2 and O2 simultaneously), respectively. As observed, a CV obtained in the presence of CO + O2-saturated electrolyte exhibits a small peak corresponding to the oxidation on the surface of the catalyst. However, it was observed that the CV nearly remained intact after the CO stripping treatment (N2, and then O2 purging for 30 min). For a clearer illustration of the obtained results, LSV was recorded before and after CO stripping, which illustrates a negligible change in onset potential, Eon (0.92 V), and current density j (∼5 mA cm−2) value for Pt/PMx. However, Pt/C exhibited a greatly limited current density and onset potential. This is because CO is lethal to the Pt centers and blocks the active adsorption of oxygen species, reducing the activity of the material. Our study observed that replacing the Vulcan C substrate with the conducting and porous MXene greatly protects the active sites during poisoning. Then, the effect of poisoning on the catalyst was determined in terms of MA and ECSA. A similar protocol was employed to obtain the activity of the catalyst in basic medium (Fig. 5j and k). As studied,56 the C–N sites remain intact in a CO environment. This could be the main reason why Pt/PMx tended to remain intact post-poisoning due to the presence of N-doped active sites in PMx, protecting the Pt centers from harsh CO exposure. To confirm the above-mentioned results, HR-TEM was performed post-ADT, as shown in Fig. S8. As observed, it was found that although the wrinkles in PMx were found to be altered, the MXene layers protect the Pt NPs from the harsh acid environment. Table 1 summarises the durability and poisoning study performed in acidic and basic media.
The electrochemical activity of the catalyst towards HER
After ascertaining the excellent performance in ORR, the catalyst was subjected to test its HER activity in both acidic and alkaline media. The HER activity of the as-synthesized materials was examined in N2-saturated 0.5 M H2SO4 using a three-electrode setup. To avoid Pt dissociation, a graphite rod was used as the counter electrode in this case. The polarization curve, as shown in Fig. 6a, displays the onset potential of 298 mV at the current density of 50 mA cm−2 (Pt/C – 260 mV). The adoption of porous crumbled and nitrogen doped MXene as a support not only enhances the porosity but also increases the conductivity and hydrophilicity of the catalyst. As shown in Fig. 6b, the obtained EIS depicts a small semi-circle for Pt/PMx. This might be attributed to the high conductivity of MXene as a support and its interaction with Pt, which provides strong catalytic active sites. To evaluate the rate of the reaction, the Tafel slope was obtained using the potential vs. log(current density) plot, as shown in Fig. 6c. The reaction rate of 35.1 mV dec−1 was obtained for Pt/PMx, which is found to be comparable with the standard Pt/C (31 mV dec−1).In general, HER activity exhibits a three-step reaction mechanism in acidic media,57 as follows: (a) primary adsorption of H+ ions, i.e. Volmer step (H3O+ + e− + M ⇌ M–H + H2O), (b) electrochemical desorption, Heyrovsky step (M–H + H3O+ + e− ⇌ H2 + H2O + M), and (c) chemical recombination, Tafel mechanism (2M–H ⇌ H2 + 2M), with theoretical Tafel slopes of 120 mV dec−1, 40 mV dec−1, and 30 mV dec−1, respectively.6,43,58 A lower Tafel value indicates the Volmer–Heyrovsky mechanism, with H+ adsorption as the rate-determining step. This implies fast H2 generation efficiency, as further confirmed by ECSA and TOF calculations. The ECSA was analyzed using the electrochemical double-layer capacitance. CV was recorded at various scan rates in the non-faradic region, as shown in Fig. S9a–d for the as-synthesized catalysts. These CV curves were utilized to calculate Cdl (Fig. S9a′–d′). The value obtained for Pt/PMx is 537 cm2 in comparison with that of Pt/C of 1020 cm2. Furthermore, the intrinsic behavior was examined by calculating the TOF of the catalyst. The TOF of Pt/PMx was found to be 5.03 s−1, which is greater than that of Pt/C (4.78 s−1). Here, TOF refers to the amount of H2 evolved per Pt at the active site per second (reference is S1). Alongside intrinsic activity, the obtained overpotentials of the catalyst at different current densities (10, 50, 100, and 150 mA cm−2) were plotted against that of Pt/C (inset of Fig. 6d), demonstrating that its response is comparable to the commercial material. The chronoamperometry stability (∼35 h) obtained for Pt/PMx (Fig. 6d) exhibits a transient performance. The current rapidly increased until the catalyst reached its stable state, and thereafter a constant response at ∼51 mV (10 mA cm−2) was maintained, demonstrating excellent stability even in an acidic medium. To evaluate the stability of the as-synthesized catalyst, XRD analysis was conducted after the stability test, as shown in Fig. S10. The results depicts no change in the peak positions; however, a noticeable decrease in peak intensity was observed. These changes suggest possible alterations in the structural integrity and phase stability of the catalyst. In addition to their excellent HER activity in acidic medium, the as-synthesized materials were further tested in the alkaline electrolyte of 0.1 M KOH. Fig. 6e depicts the polarization curve obtained in N2-saturated 0.1 M KOH. Pt/PMx exhibits enhanced HER activity compared to the other electrode materials, and the onset potential was found to be close to that of Pt/C. The hybrid catalyst shows an onset potential of 92 mV, which is close to that of the benchmark Pt/C (89 mV). However, it is much lower than that of MxSP and PMx, indicating its superior electrocatalytic activity. Further, the EIS obtained at this potential (Fig. 6f) depicts a smaller semi-circle, i.e. lower Rct, compared to Pt/C and other synthesized catalysts. These results confirm the hydrophilic, porous and conducting nature of MXene, which boosts the mass transport rate of the descriptors in Pt/PMx. To comprehend the reaction mechanism, the Tafel slope was obtained by linear fitting of the overpotential (η) vs. log(j) plot in the low η region (Fig. 6g). The Tafel slopes obtained for the synthesized catalysts are 140 mV dec−1, 133 mV dec−1, 67.9 mV dec−1 and 64.8 mV dec−1 for MxSP, PMx, Pt/PMx and Pt/C, respectively. This represents the Volmer–Heyrovsky mechanism of hydrogen evolution (H* + H+ + e−n → H2). In contrast, the pristine catalyst shows a sluggish Volmer mechanism (H+ + e−n → H*). Apparently, the hydrophilic nature of MXene gives rise to the Volmer–Heyrovsky mechanism. It also improves the overall hydrophilicity of Pt/PMx, which aids in the smooth adoption and dissociation process of the adsorbate, i.e. hydronium ions. Further, to evaluate the intrinsic activity, the ECSA (Fig. S11a–d), Cdl (Fig. S11a′–d′) and TOF were evaluated with respect to the number of active Pt sites. The results illustrate that the ECSA values follow the order of MxSP (42.5 cm2) < PMx (47 cm2) < Pt/PMx (300 cm2) < Pt/C (475 cm2). Pt/PMx shows a TOF of 2.8 s−1 in alkaline medium, which is greater than Pt/C (2.7 s−1). Further, the chronoamperometric response recorded in 1 M KOH medium shows a transient performance with no decay in current density even after 35 h, demonstrating excellent stability in alkaline medium. This constant response is mainly attributed to the strong support, i.e. PMx, employed for Pt NPs, confirming the absence of degradation, poisoning, or agglomeration when subjected to long-term stability.
Conclusion
This work emphasizes the development highly poriferous, hydrophilic and conducting MXene-supported Pt NPs derived from the “template-based transformation” strategy. Herein, we report the (2–3–2)D transformative strategy, which first transforms two-dimensional sheet-like MXene to a 3-dimensional spherical morphology, followed by the formation of 2-D highly porous and wrinkled nanosheet of MXene post-pyrolysis treatment. This reduces the large dead volume, aggregation, and restacking and enhances the conductivity and hydrophilicity of pristine MXene. The FE-SEM and HR-TEM images confirm the wrinkled structure of PMx, where Pt NPs were found to be uniformly distributed with an average size of ∼4 nm. The wrinkled nature of PMx was found to beneficial to prevent the restacking of MXene and expose a greater number of active sites. Additionally, this porous and wrinkled sheet provides in situ active N sites. The XPS analysis affirms the strong Pt and support interaction, which tunes the electronic structure of Pt/PMx, further improving the electrocatalytic performance for ORR and HER. The obtained performance was found to be comparable with commercial Pt/C. Additionally, when employed against long-term ORR stability, i.e. for 40![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 s chronoamperometric stability and ADT of 20
000 s chronoamperometric stability and ADT of 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 potential CV cycles in ORR, the as-designed catalyst exhibited better stability than the commercial Pt/C. The wrinkled and porous nature of the catalyst was found to protect against CO poisoning. The comprehensive electrochemical and intrinsic analyses post-stability and poisoning study reveal the excellent stability and anti-poisoning ability of the catalyst. Similarly, the HER study reveals improved stability for ∼40 h in both acidic and alkaline media. Additionally, the obtained HER performance for the hybrid catalyst illustrates a lower overpotential and better TOF for Pt supported on porous MXene compared to carbon. The (2–3–2)D transformation strategy paves the way for the design of conducive strong supports for Pt-based materials, leading to a catalyst performance that is comparable with Pt/C.
000 potential CV cycles in ORR, the as-designed catalyst exhibited better stability than the commercial Pt/C. The wrinkled and porous nature of the catalyst was found to protect against CO poisoning. The comprehensive electrochemical and intrinsic analyses post-stability and poisoning study reveal the excellent stability and anti-poisoning ability of the catalyst. Similarly, the HER study reveals improved stability for ∼40 h in both acidic and alkaline media. Additionally, the obtained HER performance for the hybrid catalyst illustrates a lower overpotential and better TOF for Pt supported on porous MXene compared to carbon. The (2–3–2)D transformation strategy paves the way for the design of conducive strong supports for Pt-based materials, leading to a catalyst performance that is comparable with Pt/C.
Author contributions
Rupali S. Mane: conceptualization, methodology, investigation, formal analysis, visualization, validation, writing – original draft. Higgins Wilson: conceptualization, data curation, writing – reviewing and editing original draft. Bhagyashree N. Patil: data analysis, investigation. Sang Joon Lee: resources, writing – reviewing and editing original draft, funding acquisition, project administration. Neetu Jha: conceptualization, funding acquisition, writing – reviewing and editing original draft, project administration.Conflicts of interest
The author declares that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.Data availability
The data will be made available on request.Supplementary information is available. See DOI: https://doi.org/10.1039/d5ta04239a.
Acknowledgements
This research work was sponsored by the Department of Science and Technology (DST/TMD/HCF/2K18/130), University Grant Commission-FRP (F.4-5(84-FRP) & (21-FRP)/2014(BSR)) and the Mahatma Jyotiba Phule Research Programme. Authors H. M. Wilson and S. J. Lee thank the National Research Foundation of Korea (NRF) for the financial support through Korea Research Fellowship (Brain pool program/2021H1D3A2A01099482). We would like to thank IIT Bombay and IIT Mandi SAIF Department for carrying out sample analysis.References
- Z. Manappadan and K. Selvaraj, Energy Adv., 2022, 1, 886–899 RSC.
- R. S. Mane, G. Periyasamy and N. Jha, Electrochim. Acta, 2024, 481, 143916 CrossRef CAS.
- S. Sui, X. Wang, X. Zhou, Y. Su, S. Riffat and C. Liu, J. Mater. Chem. A, 2017, 5, 1808–1825 RSC.
- Y. Sun, B. Huang, Y. Li, Y. Xing, M. Luo, N. Li, Z. Xia, Y. Qin, D. Su, L. Wang and S. Guo, Chem. Mater., 2019, 31, 8136–8144 CrossRef CAS.
- P. Q. Phan, R. Naraprawatphong, P. Pornaroontham, J. Park, C. Chokradjaroen and N. Saito, Mater. Adv., 2021, 2, 322–335 RSC.
- S. B. Alex, L. Vazhayal, P. P. Waghmaitar, R. R. Urkude, B. B. Chandashive, D. Khushalani and S. K. Haram, ACS Appl. Energy Mater., 2024, 7, 1890–1901 CrossRef CAS.
- J. Lee, N. Son, B. H. Park, S. Kim, D. Bae, M. Kim, S. W. Joo and M. Kang, Appl. Surf. Sci., 2022, 600, 154048 CrossRef CAS.
- X. Xie, S. Chen, W. Ding, Y. Nie and Z. Wei, Chem. Commun., 2013, 49, 10112–10114 RSC.
- J. Xu, D. Aili, Q. Li, C. Pan, E. Christensen, J. O. Jensen, W. Zhang, G. Liu, X. Wang and N. J. Bjerrum, J. Mater. Chem. A, 2013, 1, 9737–9745 RSC.
- C. Zhang, B. Ma, Y. Zhou and C. Wang, J. Electroanal. Chem., 2020, 865, 114142 CrossRef CAS.
- Z. Zhang, C. Liu, Y. Dai, B. Liu, P. Guo, F. Tu, M. Ma, L. Shen, Z. Zhao, Y. Liu, Y. Zhang, L. Zhao and Z. Wang, ACS Appl. Energy Mater., 2022, 5, 14957–14965 CrossRef CAS.
- N. Ma, N. Li, T. Wang, X. Ma and J. Fan, J. Mater. Chem. A, 2022, 10, 1390–1401 RSC.
- Q. Zhao, Y. Zhang, C. Ke, X. Yang and W. Xiao, Nanoscale, 2023, 15, 17516–17524 RSC.
- W. Yuan, L. Cheng, Y. An, H. Wu, N. Yao, X. Fan and X. Guo, ACS Sustainable Chem. Eng., 2018, 6, 8976–8982 CrossRef CAS.
- S. G. Peera, C. Liu, A. K. Sahu, M. Selvaraj, M. C. Rao, T. G. Lee, R. Koutavarapu, J. Shim and L. Singh, Adv. Mater. Interfaces, 2021, 8, 2100975 CrossRef CAS.
- J.-J. Huang, X.-Q. Liu, F.-F. Meng, L.-Q. He, J.-X. Wang, J.-C. Wu, X.-H. Lu, Y.-X. Tong and P.-P. Fang, J. Electroanal. Chem., 2020, 856, 113727 CrossRef CAS.
- K. Chaudhary, B. Basha, S. Zulfiqar, S. Yousaf, E. W. Cochran, M. S. Al-Buriahi, M. Farooq Warsi and M. Shahid, Fuel, 2023, 351, 128856 CrossRef CAS.
- D. Kan, R. Lian, D. Wang, X. Zhang, J. Xu, X. Gao, Y. Yu, G. Chen and Y. Wei, J. Mater. Chem. A, 2020, 8, 17065–17077 RSC.
- L. Yang, Y. C. Kimmel, Q. Lu and J. G. Chen, J. Power Sources, 2015, 287, 196–202 CrossRef CAS.
- A. Hrnjić, A. R. Kamšek, L. Bijelić, A. Logar, N. Maselj, M. Smiljanić, J. Trputec, N. Vovk, L. Pavko, F. Ruiz-Zepeda, M. Bele, P. Jovanovič and N. Hodnik, ACS Catal., 2024, 14, 2473–2486 CrossRef PubMed.
- C. Jackson, G. T. Smith, D. W. Inwood, A. S. Leach, P. S. Whalley, M. Callisti, T. Polcar, A. E. Russell, P. Levecque and D. Kramer, Nat. Commun., 2017, 8, 15802 CrossRef CAS PubMed.
- L. Xiu, W. Pei, S. Zhou, Z. Wang, P. Yang, J. Zhao and J. Qiu, Adv. Funct. Mater., 2020, 30, 1910028 CrossRef CAS.
- L. Xiu, Z. Wang, M. Yu, X. Wu and J. Qiu, ACS Nano, 2018, 12, 8017–8028 CrossRef CAS PubMed.
- W. Si, Q. Xie, Y. Shen and Z. Wang, Catal. Lett., 2024, 154, 1819–1827 CrossRef CAS.
- M. T. Benson, Ind. Eng. Chem. Res., 2003, 42, 4147–4155 CrossRef CAS.
- L. Liu, Z.-H. Xie, Q.-F. Deng, X.-X. Hou and Z.-Y. Yuan, J. Mater. Chem. A, 2017, 5, 418–425 RSC.
- B. Alič, U. Šebenik and M. Krajnc, Polymers, 2024, 16, 2463 CrossRef PubMed.
- W. Bao, L. Liu, C. Wang, S. Choi, D. Wang and G. Wang, Adv. Energy Mater., 2018, 8, 1702485 CrossRef.
- G. León, N. Paret, P. Fankhauser, D. Grenno, P. Erni, L. Ouali and D. L. Berthier, RSC Adv., 2017, 7, 18962–18975 RSC.
- J. Wen, S. Zhang, Y. Liu and Y. Zhai, J. Colloid Interface Sci., 2022, 624, 338–347 CrossRef CAS PubMed.
- R. Mane, S. Mane, V. Somkuwar, N. Thombre, A. Patwardhan and N. Jha, Battery Energy, 2023, 2, 20230019 CrossRef CAS.
- K. Allen-Perry, W. Straka, D. Keith, S. Han, L. Reynolds, B. Gautam and D. E. Autrey, Materials, 2021, 14, 694 CrossRef CAS PubMed.
- A. E. Allah, RSC Adv., 2023, 13, 9983–9997 RSC.
- A. Chen, H. Wei, Z. Peng, Y. Wang, S. Akinlabi, Z. Guo, F. Gao, S. Duan, X. He, C. Jia and B. B. Xu, Small, 2024, 20, 2404011 CrossRef CAS PubMed.
- L. Yu, Z. Fan, Y. Shao, Z. Tian, J. Sun and Z. Liu, Adv. Energy Mater., 2019, 9, 1901839 CrossRef.
- X. Tao, L. Zhang, X. He, L. Fang, H. Wang, L. Zhang, L. Yu and G. Zhu, Molecules, 2022, 27, 4890 CrossRef CAS PubMed.
- Y. Liu, X. Zeng, Y. Wu, H. U. Khan, F. Xiao, B. Y. Xia and Y. Sun, Adv. Sens. Energy Mater., 2024, 100091 CrossRef.
- M. N. Kumar, B. Govindh and N. Annapurna, Asian J. Chem., 2017, 29, 2541–2544 CrossRef CAS.
- O. Martinez-Mora, L. F. Leon-Fernandez, M. Velimirovic, F. Vanhaecke, K. Tirez, J. Fransaer and X. Dominguez-Benetton, Mater. Adv., 2023, 4, 6183–6191 RSC.
- X. Duan, C. Wang, F. Lv, T. Liu, X. Liu, Q. Wang and S. Yuan, Electrochim. Acta, 2025, 512, 145510 CrossRef CAS.
- Z. Kang, J. Cai, D. Ye, H. Zhao, J. Luo and J. Zhang, Chem. Eng. J., 2022, 446, 137443 CrossRef CAS.
- K. M. Naik, K. Hashisake, T. Hamada, E. Higuchi and H. Inoue, J. Mater. Chem. A, 2022, 10, 13987–13997 RSC.
- R. S. Mane, A. R. Shakeelur Rahema, T. Kothawade, H. Chakraborty and N. Jha, Fuel, 2022, 323, 124420 CrossRef CAS.
- X. Ye, Y. Xue, K. Li, W. Tang, X. Han, X. Zhang, Z. Song, Z. Zheng and Q. Kuang, Mater. Adv., 2021, 2, 5479–5486 RSC.
- T. M. Pham, K. Im and J. Kim, Appl. Surf. Sci., 2023, 611, 155740 CrossRef CAS.
- K. M. Naik, E. Higuchi and H. Inoue, Nanoscale, 2020, 12, 11055–11062 RSC.
- F. T. Angerasa, C.-Y. Chang, E. A. Moges, W.-H. Huang, K. Lakshmanan, Y. Nikodimos, J.-F. Lee, N. G. Habtu, M.-C. Tsai, W.-N. Su and B. J. Hwang, Mater. Today Energy, 2023, 34, 101312 CrossRef CAS.
- G. Zhang, C. Ma, Q. He, H. Dong, L. Cui, L. Li, L. Li, Y. Wang and X. Wang, iScience, 2023, 26, 106622 CrossRef CAS PubMed.
- X. Wang, L. Bai, J. Lu, X. Zhang, D. Liu, H. Yang, J. Wang, P. K. Chu, S. Ramakrishna and X.-F. Yu, Angew. Chem., Int. Ed., 2019, 58, 19060–19066 CrossRef CAS PubMed.
- K. M. Naik, E. Higuchi and H. Inoue, J. Power Sources, 2020, 455, 227972 CrossRef CAS.
- H. Cui, H. Mi, C. Ji, F. Guo, Y. Chen, D. Wu, J. Qiu and H. Xie, J. Mater. Chem. A, 2021, 9, 23941–23954 RSC.
- R. S. Mane, D. Zaroliwalla, G. Periyasamy and N. Jha, Small, 2025, 2503228 CrossRef CAS PubMed.
- P. Ganesan, A. Staykov, A. Mufundirwa, T. Sugiyama, H. Shu, M. Uejima and N. Nakashima, Mater. Adv., 2022, 3, 6539–6548 RSC.
- V. C. A. Ficca, C. Santoro, E. Placidi, F. Arciprete, A. Serov, P. Atanassov and B. Mecheri, ACS Catal., 2023, 13, 2162–2175 CrossRef CAS.
- H. Meng, S. Pei, H. Li and Y. Zhang, ACS Omega, 2021, 6(16), 11059–11067 CrossRef CAS PubMed.
- R. S. Mane, A. S. Nair, R. I. Jafri and N. Jha, ACS Appl. Nano Mater., 2024, 7, 23454–23465 CrossRef CAS.
- K. M. Naik, E. Higuchi and H. Inoue, Int. J. Hydrogen Energy, 2023, 48, 30741–30750 CrossRef CAS.
- J. N. Tiwari, S. Sultan, C. W. Myung, T. Yoon, N. Li, M. Ha, A. M. Harzandi, H. J. Park, D. Y. Kim, S. S. Chandrasekaran, W. G. Lee, V. Vij, H. Kang, T. J. Shin, H. S. Shin, G. Lee, Z. Lee and K. S. Kim, Nat. Energy, 2018, 3, 773–782 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2025 |