Mixed matrix membranes of PIM-1 incorporating MOF-808 functionalized with amino acids for enhanced CO2/CH4 separation
Dalia
Refaat
 ab,
Mohamed
Yahia
ab,
Mohamed
Yahia
 *abcd,
Harol David
Martínez-Hernández
ef,
Monica
Jimenez-Ruiz
e,
Vanessa
Galván
*abcd,
Harol David
Martínez-Hernández
ef,
Monica
Jimenez-Ruiz
e,
Vanessa
Galván
 fg,
Viktor
Petrenko
fg,
Viktor
Petrenko
 gh,
Roberto
Fernández de Luis
gh,
Roberto
Fernández de Luis
 *g and
Joaquín
Coronas
*g and
Joaquín
Coronas
 ab
ab
aInstituto de Nanociencia y Materiales de Aragón (INMA), CSIC-Universidad de Zaragoza, Zaragoza 50018, Spain
bChemical and Environmental Engineering Department, Universidad de Zaragoza, Zaragoza 50018, Spain
cChemistry Department, Faculty of Science, Helwan University, Cairo, 11795, Egypt. E-mail: mohamed.yahia@science.helwan.edu.eg
dCenter for Cooperative Research on Alternative Energies (CIC energiGUNE), Basque Research and Technology Alliance (BRTA), Alava Technology Park, Albert Einstein 48, Vitoria-Gasteiz 01510, Spain. E-mail: myahia@cicenergigune.com
eInstitut Laue Langevin, 71 Avenue des Martyrs, CS 20156, 38042 Grenoble, France
fDepartment of Organic and Inorganic Chemistry, Faculty of Science and Technology, University of the Basque Country UPV/EHU, Sarriena auzoa z/g, 48940 Leioa, Spain
gBCMaterials, Basque Center for Materials, Applications and Nanostructures, UPV/EHU Science Park, 48940 Leioa, Spain. E-mail: roberto.fernandez@bcmaterials.net
hIKERBASQUE, Basque Foundation for Science, 48009 Bilbao, Spain
First published on 10th October 2025
Abstract
This study investigates the enhancement of CO2/CH4 separation performance in mixed matrix membranes (MMMs) by incorporating amino acid-functionalized MOF-808 (MOF-808@AA; AA = Lys, Arg and Cys) into a polymer of intrinsic microporosity (PIM-1) matrix. MMMs containing 10 wt% MOF-808@AA were fabricated via solution casting. The structural and physicochemical properties of PIM-1, MOF-808@AA, and the resulting MMMs were characterized using 1H-NMR, FTIR, TGA, SEM, XRD, SAXS, N2/77 K and CO2/15 °C isotherms, and high-pressure CO2 adsorption. Collectively, the characterization results confirm the successful incorporation of MOF-808@AA into the PIM-1 membranes, with no detectable alteration of the nanostructure of the PIM-1 matrix. Gas separation measurements revealed a notable increase in both CO2 permeability and CO2/CH4 selectivity upon incorporation of MOF-808@AA fillers. Among the tested membranes, the PIM-1/MOF-808@Lys MMM functionalized with lysine exhibited the best performance, achieving a CO2 permeability of 14![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 354 ± 201 Barrer and a CO2/CH4 selectivity of 26.4 ± 1.8. Aging effects were also studied, showing a reduction in CO2 permeability accompanied by a slight increase in selectivity over time. In addition, the single and mixed gas experiments at elevated pressures (up to 6 bar) showed that CO2 permeability under mixed gas conditions was slightly higher than in single gas experiments, supporting the potential of these MMMs for application in realistic CO2/CH4 separation processes under practical operating conditions. Inelastic neutron scattering (INS) and density functional theory (DFT) calculations confirmed that CO2 molecules preferentially interact with the amino groups introduced by AA functionalization. DFT simulations further revealed that while CO2 and CH4 access similar adsorption sites, the interaction energy of CO2 with amino groups is approximately three times higher than that of CH4. These findings demonstrate that MOF-808@AA fillers can significantly enhance the CO2-philicity of PIM-1-based MMMs, offering a promising approach for efficient CO2/CH4 separation compared to pristine PIM-1 or unmodified PIM-1/MOF-808 composites.
354 ± 201 Barrer and a CO2/CH4 selectivity of 26.4 ± 1.8. Aging effects were also studied, showing a reduction in CO2 permeability accompanied by a slight increase in selectivity over time. In addition, the single and mixed gas experiments at elevated pressures (up to 6 bar) showed that CO2 permeability under mixed gas conditions was slightly higher than in single gas experiments, supporting the potential of these MMMs for application in realistic CO2/CH4 separation processes under practical operating conditions. Inelastic neutron scattering (INS) and density functional theory (DFT) calculations confirmed that CO2 molecules preferentially interact with the amino groups introduced by AA functionalization. DFT simulations further revealed that while CO2 and CH4 access similar adsorption sites, the interaction energy of CO2 with amino groups is approximately three times higher than that of CH4. These findings demonstrate that MOF-808@AA fillers can significantly enhance the CO2-philicity of PIM-1-based MMMs, offering a promising approach for efficient CO2/CH4 separation compared to pristine PIM-1 or unmodified PIM-1/MOF-808 composites.
1. Introduction
Natural gas derived from fossil fuels represents one of the most important energy vectors for our society today. As a sustainable alternative, biogas is a non-fossil fuel that can be obtained from the valorization of different waste deposits. The composition of natural gas or biogas depends on their origin, but they typically show a significant percentage of carbon dioxide (CO2) as a secondary component.1 Indeed, the removal of CO2 from methane-rich (CH4) gas streams is key to enhance their quality and improve their overall energy density. This is particularly important in the context of biogas production and conditioning prior to its injection into natural gas distribution pipeline systems.1,2 Overall, the CO2 separation from methane-rich streams is key to prevent reductions in their energy density and, in parallel, mitigate potential adverse effects on their compression and transportation.Consequently, there is a pressing need to develop efficient, selective and cost-effective technologies for CO2 separation from CH4.1,2 Today, a variety of industrial techniques, including cryogenic separation, absorption and membrane separation, are applied to this end.3–6 Of these methods, recent advances in mixed matrix membrane (MMM)-based separation have steadily approached practical application in comparison to traditional technologies.3–6
On the one hand, polymeric membrane production is easy to scale up, with a reduced footprint and cost, and thus, making it an appealing option for CO2 separation.1,2 On the other hand, it is challenging to modify the internal structure of the polymeric selective layer in order to improve both permeability and selectivity simultaneously. Consequently, the goal is to develop membrane materials that provide an exceptional selectivity to minimize the methane losses while maintaining a high permeability to decrease the production costs.7–9 In this sense, MMMs effectively combine the favorable processing capabilities of polymeric materials with the outstanding transport characteristics of specific fillers, making them highly promising for gas separation.10,11 The main aim is to develop membranes that achieve superior permeability and selectivity values, with the goal of surpassing the so-called Robeson upper bound.4,5,12 By incorporating inorganic nano-fillers (i.e. porous silicas, carbons, zeolites, and MOFs) into the polymer framework, it is possible to enhance the selective permeation through mechanisms such as molecular sieving, solubility modulation, or barrier effect and disorder within the polymer chains.13,14 However, a significant challenge in fabricating MMMs is ensuring a uniform distribution of the inorganic nano-fillers within the thin selective layer of the polymer matrix, in addition to obtaining a proper filler–polymer interaction. Excessive filler concentrations can adversely affect both the performance and the mechanical integrity of the membranes.15,16 Additionally, the incompatibility between the polymer and the nano-filler decreases the selectivity by creating low-selectivity interfaces.17,18
Polymers of intrinsic microporosity (PIMs) have partially overcame the limitations exhibited by the selective thin layers of classic MMM technologies. These polymeric porous membranes combine high permeability with satisfactory selectivity arising from their interconnected micro-cavities and the presence of spiro-bisindane moieties in their structure (Scheme 1a). Together, these features introduce considerable steric hindrance that enhances the fractional free volume (FFV) and rigidity of the polymer chains.19–21 Among the PIM family, PIM-1 is the most thoroughly investigated polymer, recognized for its excellent solubility, robust physicochemical and thermal stability. For instance, PIM-1 exhibits superior attributes in CO2/CH4 separation, achieving selectivity values ranging from about 7.5 to 33.5.22 Building on this foundation, the incorporation of inorganic or organic fillers into PIM-based matrices has been demonstrated to enhance the key performance metrics of these porous polymer membranes for gas separation applications.11,15 Nevertheless, the integration of porous fillers that actively contribute to gas separation, particularly by enhancing selectivity, remains relatively underexplored. Preliminary studies, however, suggest strong potential for synergistic effects between the fillers and the PIM matrix.15
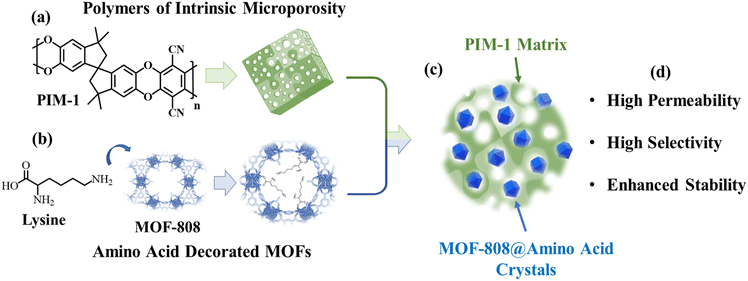 | ||
| Scheme 1 General strategy to combine (a) polymers of intrinsic microporosity and (b) amino acid functionalized MOF-808 to (c) develop mixed matrix membranes with (d) enhanced properties. | ||
This is precisely where metal organic frameworks (MOFs) offer significant advantages over other conventional porous materials (Scheme 1a). MOFs are constructed via the coordination of metal clusters and organic linkers, whose geometry, connectivity, and chemical functionality can be systematically tailored.23,24 This tunability allows precise modulation of the pore size, aperture and, more importantly, the selective interactions and/or diffusivity of target gas molecules such as CO2 and CH4. Furthermore, the incorporation of MOF fillers into MMMs for gas separation, including those based on PIMs, offers additional advantages, such as mitigating physical ageing, enhancing resistance to plasticization, and improving interfacial compatibility with the surrounding polymer matrix.25–30 In detail, it has been demonstrated that CO2/CH4 separation in PIM-1/MOF MMMs is influenced by the MOF-filler features, such as crystal structure, particle size,31 shape32 and surface chemistry,33,34 as well as the MOF loading and membrane processing conditions.35,36
Yet, further opportunities remain to be explored in terms of the post-synthetic functionalization versatility of MOF nanofillers to enhance their selectivity in gas separation via membrane applications, particularly for the purification of CO2/CH4 mixtures.37–41 In contrast to other Zr–MOFs, six of the twelve coordination positions of the Zr6(μ3-O)4(μ3-OH)4 clusters are “linker-defective” in MOF-808. This structural feature enables the straightforward post-synthetic functionalization of these sites with amino acids (Scheme 1b), thereby imparting the parent MOF with exceptional CO2 adsorption capacity at very low relative pressures.42 An important question arising from these adsorption studies is whether the exceptional CO2 affinity observed in amino acid-functionalized MOF-808 can be effectively translated into PIM-1/MOF MMMs, where gas separation is governed by diffusion across the composite matrix (Scheme 1c).
Although some studies have reported the incorporation of MOF-808@AA (AA = amino acid) into different polymer matrices as MMMs for various applications,43,44 this is the first time such MOF-808@AA materials have been integrated into a PIM-1 matrix applied for gas separation purposes. Thus, in this work, we have explored the performance for CO2/CH4 separation of PIM-1/MOF-808@AA MMMs with a 10 wt% loading of MOF-808@AA. Cysteine, arginine and lysine have been selected as the AAs to be integrated into MOF-808 due to the varied accessibility and chemical reactivity of their amino groups.38,45 The resulting PIM-1/MOF-808@AA MMMs were tested for their CO2/CH4 separation efficiency at 35 °C, comparing their performance with the adsorption capacity and selectivity values obtained for the bare MOF-808@AA materials. The CO2 and CH4 interaction with MOF-808@AA was studied by means of inelastic neutron spectroscopy (INS) and density functional theory (DFT) calculations, confirming the preference of MOF-808@AA to interact with CO2 in comparison to CH4, even though they share similar adsorption sites. All in all, we have demonstrated that the selectivity shown by the amino acid decorated MOF fillers is transferred to the PIM-1/MOF-808@AA membranes in terms of a significant CO2 selectivity gain without permeability loss (Scheme 1d).
2. Experimental
2.1. PIM-1 polymer and MOF crystals synthesis and assembly
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, v
1, v![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) v) solutions. The trimesic acid does not dissolve completely at this stage. The ZrCl4 solution was added dropwise to the acid one in a 1 L screw-capped glass jar reactor under stirring until a clear solution was obtained. The reactor was immersed in an oil bath for refluxing. After 1 h of reaction, the solution started to become cloudy, and once completed after 24 h, a white suspension of MOF-808 nanoparticles was obtained. The material was collected by centrifugation and washed twice with water (12 h each) and twice with MeOH (12 h each). The sample was finally activated at 80 °C for 24 h.
v) solutions. The trimesic acid does not dissolve completely at this stage. The ZrCl4 solution was added dropwise to the acid one in a 1 L screw-capped glass jar reactor under stirring until a clear solution was obtained. The reactor was immersed in an oil bath for refluxing. After 1 h of reaction, the solution started to become cloudy, and once completed after 24 h, a white suspension of MOF-808 nanoparticles was obtained. The material was collected by centrifugation and washed twice with water (12 h each) and twice with MeOH (12 h each). The sample was finally activated at 80 °C for 24 h.
2.2. Fabrication of PIM-1/MOF-808@AA MMMs
A 10 wt% loading of the MOF in the PIM-1-based MMMs was selected as the ideal value based on previous investigations.47,48 Following this approach, PIM-1@MOF-808 and PIM-1/MOF-808@AA MMMs were fabricated as follows. First, PIM-1 was dissolved in CHCl3. Meanwhile, dispersions of MOF-808 and MOF-808@AA were prepared by sonicating them in CHCl3 for 1 h. Once the PIM-1 solution was ready, it was combined with the MOF dispersions and stirred continuously for another 3 h to ensure thorough homogeneity of the mixture. In the following, it was cast into glass Petri dishes and left at room temperature for 3 days to allow the solvent to fully evaporate. Prior to membrane testing, both the pure PIM-1 membranes and the MMMs were soaked in methanol for 3 h, followed by drying under vacuum at 60 °C overnight. Finally, the samples were stored in a dark place under ambient conditions.2.3. Characterization of PIM-1, MOFs and MMMs
The 1H NMR analysis, as shown in Fig. S1–S4, was conducted using a Bruker Avance III 400 spectrometer (Bruker, Billerica, MA, USA), operating at 400 MHz for proton frequency at 25 °C, with CDCl3 serving as the internal reference. PIM-1 molecular weight was determined through gel permeation chromatography (GPC) with multi-angle light scattering (MALS) detection. For 1H-NMR analysis, 20 mg of MOF-808 and MOF-808@AA samples were digested in a NaOH deuterated water solution (1 M, 0.7 mL). The solution was recovered carefully with a syringe, preventing the uptake of the powdered material settled at the bottom of the centrifuge tube. The experiments were performed on a Bruker AV500 equipped with a BBI probe and Z-axis gradients, operating at a frequency of 500 MHz for proton and 125.77 MHz for carbon. Once the 1H-NMR spectra were acquired, the AA content per formula unit was calculated by integrating the signals associated with the corresponding AA, acetic acid and trimesic acid molecules. A Bruker Vertex 70 FTIR spectrometer, equipped with a DTGS detector and Golden Gate diamond ATR accessory, was used for Fourier transform infrared spectroscopy (FTIR) measurements. Thermogravimetric analysis (TGA) was carried out on a Mettler Toledo TGA 851e instrument.To assess the crystallinity of the membranes and MOFs. Powder X-ray diffraction (XRD) data were acquired using a Panalytical Empyrean diffractometer, with Cu Kα1 radiation (λ = 0.154 nm). The patterns were collected between 5° and 40° at a rate of 0.03° per second. N2 adsorption isotherms at −196 °C were recorded with a Micromeritics Tristar 3000 analyzer, and the BET specific surface area (SSA) was calculated using the Brunauer–Emmett–Teller method. Scanning electron microscopy (SEM) images were captured using an Inspect F50 scanning electron microscope (FEI), operated at 10 kV. Membrane thickness was measured at 5–6 different points using a micrometer, with thickness ranging between 100 and 200 μm. High-pressure adsorption measurements in MOF-808 and MOF-808@Lys were performed using a high-pressure volumetric gas sorption analyzer iSorb. Prior to the measurements, the samples were degassed under vacuum at 120 °C for 4 h. Thermodiffractometry was performed on a Bruker D8 Advance Vantec diffractometer (Cu Kα radiation) equipped with a variable-temperature stage HTK2000 for the measurements performed in the 30 to 500 °C temperature range. Patterns were registered each 10 °C (2θ range = 5–40°, step size = 0.01°, and exposure time = 0.5 s per step). The position and intensity of the (311) plane were analyzed and are illustrated in Fig. S5–S8. For additional details about characterization techniques refer to the SI. Small-Angle X-ray Scattering (SAXS) measurements were carried out using a Xenocs system on membrane sections with an area of 1 cm2, employing a Cu Kα radiation source (λ ≈ 1.542 Å) operated at 40 kV and 40 mA. The sample-to-detector distance was set to 108 cm, providing access to a momentum transfer range of 0.01 < q < 0.2 Å−1.
2.4. Gas mixture permeation
A circular membrane (with an area of 2.12 cm2) was cut and positioned within a custom-designed module composed of two stainless steel sections and a 316LSS macroporous disc support with a pore size of 20 μm, secured using Viton O-rings, as shown in Fig. S9. The temperature was maintained at 35 °C by placing the permeation module in a Memmert oven. For the separation process, a 50/50 cm3(STP) min−1 CO2/CH4 gas mixture was fed at 3 bar, while the permeate side of the membrane was swept at atmospheric pressure using 2 cm3(STP) min−1 of He. The gas streams were controlled via mass-flow regulators (Alicat). CO2 and CH4 concentrations in the permeate were continuously analyzed using an Agilent 990 micro-gas chromatograph. After a steady state was reached (approximately 3 h), the permeability values for CO2 and CH4 were calculated in Barrer units (10−10 cm3(STP) cm cm−2 s−1 cmHg−1) by dividing the gas flux by the membrane area and the partial pressure, then multiplying by the membrane thickness. The CO2/CH4 selectivity was obtained as the ratio of their permeabilities. The formulas used for calculating permeability (eqn (1)) and selectivity (eqn (2)) are provided below.49,50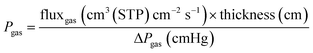 | (1) |
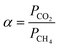 | (2) |
To assess the impact of physical aging on membrane performance, the membranes were kept in a dry cabinet for 60 days to avoid exposure to moisture. Afterward, gas permeation tests were performed on the aged membranes without any methanol treatment. For accuracy and consistency, each membrane was tested five times before and after the aging period.
Furthermore, time-lag experiments were performed with repeated measurements (at least three times) to ensure accuracy and reproducibility. The downstream pressure was kept significantly lower than the upstream pressure (not exceeding 0.1% of the upstream pressure) to meet the boundary conditions of the time-lag method and maintain a steady-state flux. Further details on the time-lag method, including measurements and calculations, can be found in our prior work.49,51 The experimental setup (Fig. S10) and the equations used to determine diffusion coefficient (D), permeability (P), and solubility (S) are provided in the SI.
2.5. Inelastic neutron scattering in situ characterization
Among the spectroscopic techniques, inelastic neutron scattering (INS) appears as an ideal experimental technique to study such systems for two reasons: (i) the incoherent scattering length of H is almost 100 times higher than that of other elements and, therefore, with INS, those vibrational modes where the H atoms are involved are mainly visible; and (ii) the absence of selection rules, which make INS particularly sensitive toward all the vibrational modes involving H atoms. The INS spectra were measured at 10 K in the range of energy transfers from 160 to 2000 cm−1 (that corresponds to 20–250 meV) with an energy resolution of ΔE/ΔE ∼2% using the IN1-Lagrange neutron spectrometer installed at the hot source of the high-flux reactor at the Institut Laue-Langevin in Grenoble. The energy transfer was calculated by subtracting 4.5 meV, the constant final energy of the Pyrolytic Graphite (PG) analyzers, from the energy of the incoming neutrons selected with a focusing Cu (220) single crystal. To the extent that the final wave vector (kf) is much smaller than the initial wave vector (ki) (and therefore high Q), the observed intensity is directly proportional to the generalized density of states (GDOS), that is, the hydrogen partial density of states in the case of hydrogenated materials. Data sets were then normalized to monitor counts and corrected for empty cell measurements.The INS experiments were conducted after degassing the MOF-808 (1.438 g), MOF-808@Lys (1.564 g) and MOF-808@Arg (1.512 g) samples overnight at 120 °C under vacuum. Following the acquisition of preliminary data, one bar of CO2 was introduced into the measurement cell, and spectra for all three materials were recorded again. The absorption bands were assigned based on the INS spectral simulations obtained from the optimized structures of the simplified model obtained by DFT calculations. The energy corresponding to the interactions of CO2 and CH4 molecules with the AAs was also calculated from simplified models. A detailed description of the data acquisition, minimization of structural models, the identification of the INS absorption maxima and, last but not least, the calculations of the absorption energy for CO2 and CH4 molecules can be found in the SI.
3. Results and discussion
3.1. PIM-1 and MOF-808@AA synthesis and functionalization
The PIM-1 structure after its synthesis was confirmed by analyzing the 1H-NMR and GPC spectra, as illustrated in our previous work.47 In detail, Fig. S1a shows the synthesis reaction between TTSBI and TFTPN monomers conducting PIM-1. The PIM-1 structure was confirmed by analyzing its 1H-NMR spectrum, which is presented in Fig. S1b. The spectrum displays the characteristic signals for the H atoms of the aromatic rings, which appear between 6.50 and 6.85 ppm, as well as for the H atoms of the five-membered rings, which are observed between 2.20 and 2.35 ppm. The signals for the H atoms of the –CH3 groups are visible between 1.30 and 1.39 ppm. The obtained spectrum serves as evidence of the correct PIM-1 polymeric structure. Additionally, the molecular weight of PIM-1 was assessed through GPC, revealing a monomodal peak in the molecular weight distribution curve, as depicted in Fig. S1c. The apparent molecular weight of PIM-1 was determined to be 75 kDa, accompanied by a PDI of 1.85. The 1H NMR and GPC analyses confirmed the successful synthesis of the PIM-1 polymer, consistent with the previous studies.11,14,46All the solvent-assisted ligand incorporation (SALI) conditions employed to decorate MOF-808 with amino acid molecules, the crystallinity of the MOF-808@AA samples after the SALI process, and the degree of functionalization of each of the samples have been detailed in Table S1 (see the SI). After preliminary characterization, the samples that maintain their crystallinity and, in parallel, exhibit the highest Zr6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) AA molar ratio were selected for their complete characterization and integration into PIM membranes (see the SI for further details). Furthermore, 1H-NMR analyses were performed for the parent MOF-808 and the amino acid (AA) functionalized MOF-808@Lys, MOF-808@Arg and MOF-808@Cys (Fig. S2–S4) after the digestion of the samples under basic conditions. The integration of the 1H-NMR signals associated with the AA, trimesate and acetate species has allowed establishment of their molar relation per MOF-808 formula unit. To this end, the 8.35 ppm signal of the trimesic acid was taken as a reference and compared to those obtained for Arg, Cys and Lys molecules. A nominal formula of [Zr6O4(OH)4(BTC)2(OH)6−x(H2O)6−x(AA+OH−)x] is proposed, where x denotes the molar equivalence of amino acids per Zr6O4(OH)4 cluster. Water and hydroxyl pairs were employed to balance the linker defective positions at the zirconium clusters non-capped by acetate groups. For instance, the 1H-NMR signal associated with acetate groups was negligible. The water solutions of the amino acids employed for the MOF-808 functionalization showed a pH close to 5. Given the pKa values of the amino and carboxyl groups, amino acids are expected to exist as zwitterions in solution. Upon integration into MOF-808, the negative charge of the carboxylate group is neutralized through coordination to the zirconium hexameric clusters. Consequently, the amino acid is stabilized within the pore space of MOF-808 as a heterogenized protonated amine. This protonated –NH3+ group originates from proton transfer from a water molecule, resulting in the formation of an –NH3+/OH− pair that is stabilized within the MOF-808 structure. By considering all these points, from the 1H-NMR analysis it can be concluded that we were able to integrate near three amino acid molecules per formula unit in the MOF-808 structure, as detailed in Table 1.
AA molar ratio were selected for their complete characterization and integration into PIM membranes (see the SI for further details). Furthermore, 1H-NMR analyses were performed for the parent MOF-808 and the amino acid (AA) functionalized MOF-808@Lys, MOF-808@Arg and MOF-808@Cys (Fig. S2–S4) after the digestion of the samples under basic conditions. The integration of the 1H-NMR signals associated with the AA, trimesate and acetate species has allowed establishment of their molar relation per MOF-808 formula unit. To this end, the 8.35 ppm signal of the trimesic acid was taken as a reference and compared to those obtained for Arg, Cys and Lys molecules. A nominal formula of [Zr6O4(OH)4(BTC)2(OH)6−x(H2O)6−x(AA+OH−)x] is proposed, where x denotes the molar equivalence of amino acids per Zr6O4(OH)4 cluster. Water and hydroxyl pairs were employed to balance the linker defective positions at the zirconium clusters non-capped by acetate groups. For instance, the 1H-NMR signal associated with acetate groups was negligible. The water solutions of the amino acids employed for the MOF-808 functionalization showed a pH close to 5. Given the pKa values of the amino and carboxyl groups, amino acids are expected to exist as zwitterions in solution. Upon integration into MOF-808, the negative charge of the carboxylate group is neutralized through coordination to the zirconium hexameric clusters. Consequently, the amino acid is stabilized within the pore space of MOF-808 as a heterogenized protonated amine. This protonated –NH3+ group originates from proton transfer from a water molecule, resulting in the formation of an –NH3+/OH− pair that is stabilized within the MOF-808 structure. By considering all these points, from the 1H-NMR analysis it can be concluded that we were able to integrate near three amino acid molecules per formula unit in the MOF-808 structure, as detailed in Table 1.
| Sample | Formula |
|---|---|
| MOF-808@Arg | Zr6O4(OH)7.2(H2O)3.2(C9O6H3)2((C6H15N4O2)(OH)2)2.8 |
| MOF-808@Cys | Zr6O4(OH)7.2(H2O)3.2(C9O6H3)2((C3H7NO2S)(OH))2.8 |
| MOF-808@Lys | Zr6O4(OH)6.8(H2O)2.8(C9O6H3)2((C6H15N2O2)(OH)2)3.2 |
3.2. PIM-1/MOF-808@AA membrane characterization
First, powder XRD was employed to corroborate that the MOF-808 and MOF-808@AA materials remain stable after their incorporation into PIM-1 membranes. The XRD patterns of the parent MOF-808 and of AA functionalized MOF-808@AA are presented in Fig. 1a. The diffraction patterns of MOF-808 and MOF-808@AA display a very similar signature in terms of the position of the main intensities (i.e. 8.4° (311), 8.7° (222), 10.1° (400) and 11.0° (311)). Consistent with the previously published literature, the integration of amino acid molecules does not alter the long-range order of the initial material.11,52–54 In addition, the crystallinity of the AA decorated materials is similar to that shown by the parent MOF-808. The XRD data were further analyzed by pattern matching analyses in order to discard the presence of secondary phases after the synthesis. As derived from the final fittings (Fig. S5–S8), the position of the main diffraction intensities, and hence the cell parameters (see Table S1), do not vary after the solvent-assisted ligand installation (SALI) of AAs. Although slight differences in the relative intensity of the diffraction intensities have been observed, the variations are not enough to determine the crystallographic position of the AA molecules via Rietveld refinement. | ||
| Fig. 1 XRD patterns of (a) MOF-808 and MOF-808@AA and (b) PIM-1/MOF-808@AA MMMs. FTIR spectra of (c) MOF-808 and MOF-808@AA and (d) PIM-1 and PIM-1/MOF-808@AA MMMs. | ||
In a second step, the PIM-1, PIM-1/MOF-808 and PIM-1/MOF-808@AA MMMs were studied by XRD analysis as well (Fig. 1b) to corroborate the integrity of the MOF structure after its integration into the polymeric membrane. As expected, the XRD pattern of PIM-1 highlights its amorphous nature (Fig. 1b). The two broad diffraction peaks, located at 13.4° and 18.0°, reflect chain-to-chain spacings of 0.49 nm and 0.65 nm, respectively. These spacings are linked to the polymer chain packing efficiency within the PIM-1 ladder-like, microporous structure.14,46,55 Additionally, the XRD data of PIM-1/MOF-808 and PIM-1/MOF-808@AA MMMs reveal both the long-range ordering of the PIM-1 matrix, as well as the stability of the crystal structure of the MOF after its integration into the polymeric scaffold. Both PIM-1 and MOF-808 retain their structures, indicating that there are no significant alterations in their structures.
Furthermore, MOF- and PIM-based MMMs were studied by IR spectroscopy. The FTIR spectra of MOF-808 and MOF-808@AA (Fig. 1c) are dominated by the characteristic vibrational bands of MOF-808. The main bands correspond to the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O symmetric and antisymmetric vibrations located between 1600 cm−1 and 1400 cm−1, associated with the carboxylate groups of the organic linkers. Upon functionalization with amino acids, MOF-808@AA exhibits new signals, particularly in the 3200–3400 cm−1 range, which are indicative of N–H stretching vibrations from the amino groups of the amino acid molecules. These changes confirm the successful amino acid functionalization of MOF-808, as previously corroborated by 1H-NMR. In addition, PIM-1 and PIM-1/MMMs were subjected to FTIR spectroscopy (Fig. 1d) to investigate the interactions between MOFs and PIM-1. For PIM-1, the absorption bands located at 2239 cm−1 and 1265 cm−1 are linked to the vibrational modes of the nitrile groups, specifically the C
O symmetric and antisymmetric vibrations located between 1600 cm−1 and 1400 cm−1, associated with the carboxylate groups of the organic linkers. Upon functionalization with amino acids, MOF-808@AA exhibits new signals, particularly in the 3200–3400 cm−1 range, which are indicative of N–H stretching vibrations from the amino groups of the amino acid molecules. These changes confirm the successful amino acid functionalization of MOF-808, as previously corroborated by 1H-NMR. In addition, PIM-1 and PIM-1/MMMs were subjected to FTIR spectroscopy (Fig. 1d) to investigate the interactions between MOFs and PIM-1. For PIM-1, the absorption bands located at 2239 cm−1 and 1265 cm−1 are linked to the vibrational modes of the nitrile groups, specifically the C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N stretching. Moreover, the absorption bands in the range of 2800–2900 cm−1 and at 1446 cm−1 are associated with methylene (CH2) stretching and bending vibration modes, respectively.14,46 The IR signal associated with the C–O stretching mode appears within the 1350–1250 cm−1 region. The band identified at 1009 cm−1 is a result of the characteristic C–N stretching, whereas that at 874 cm−1 is linked to the aromatic sp2 C–H bending. After incorporating MOF-808@AA into the PIM-1 matrix, the IR spectra of the resulting PIM-1/MOF-808@AA MMMs show additional bands associated with the C
N stretching. Moreover, the absorption bands in the range of 2800–2900 cm−1 and at 1446 cm−1 are associated with methylene (CH2) stretching and bending vibration modes, respectively.14,46 The IR signal associated with the C–O stretching mode appears within the 1350–1250 cm−1 region. The band identified at 1009 cm−1 is a result of the characteristic C–N stretching, whereas that at 874 cm−1 is linked to the aromatic sp2 C–H bending. After incorporating MOF-808@AA into the PIM-1 matrix, the IR spectra of the resulting PIM-1/MOF-808@AA MMMs show additional bands associated with the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O symmetric and antisymmetric vibrations arising from the trimesate linkers, but more importantly, additional signals located in the 3200–3400 cm−1 region, attributed to the N–H stretching from the amino acid functionalization of MOF-808. This observation confirms the effective embedding of the MOF-808@AA particles into the PIM-1 polymer matrix. The lack of shifts in the IR signals arising from PIM and MOF-808 components indicates that they are stabilized by weak interactions that do not affect their molecular components at the interphase.
O symmetric and antisymmetric vibrations arising from the trimesate linkers, but more importantly, additional signals located in the 3200–3400 cm−1 region, attributed to the N–H stretching from the amino acid functionalization of MOF-808. This observation confirms the effective embedding of the MOF-808@AA particles into the PIM-1 polymer matrix. The lack of shifts in the IR signals arising from PIM and MOF-808 components indicates that they are stabilized by weak interactions that do not affect their molecular components at the interphase.
N2 sorption isotherms at 77 K were employed to evaluate the microporosity of MOF-808, its amino acid-functionalized derivatives (MOF-808@AA) and PIM-1. Fig. S11 and Table 2 confirm the microporous nature of MOF-808 and MOF-808@AA materials.30,56–60 PIM-1 exhibits a high BET SSA of 751 m2 g−1, which aligns with its well-known combination of type I and IV isotherms.61–63 This indicates a dual porosity system, consisting of both micro and mesopores, where the mesopores are accessible through the micropores, as indicated by the prominent hysteresis. Such structural properties make PIM-1 highly favorable for gas adsorption, enhancing its performance in gas separation applications.14,64 Similarly, MOF-808 demonstrates a significant BET SSA of 792 m2 g−1. This value falls significantly within the range of the experimental values previously reported for MOF-808 synthesized under comparable conditions, confirming consistency with existing data.47,59 The functionalization of MOF-808 with amino acids (lysine, arginine and cystine) results in a reduction in the BET surface area and pore size due to partial pore occupation of the amino acid molecules.38,45 Specifically, MOF-808@Cys exhibits the highest BET surface area (518 m2 g−1), followed by MOF-808@Lys (362 m2 g−1) and MOF-808@Arg (235 m2 g−1). Still, the significant reduction of the surface area after the functionalization of MOF-808 suggests that some degree of structural collapse could be induced during the amino acid integration into the material, since the degree of AA functionalization and the size of the amino acids cannot account alone for this surface area decay.65,66 Despite this reduction, the pore space of MOF-808 remains accessible, suggesting that the AA-functionalized MOFs retain a significant gas adsorption capacity. These AA modifications could tolerate the gas transport properties of MOF-808 in MMMs when combined with PIM-1, underscoring the importance of AA selection in optimizing these materials for advanced gas separation applications.
| Sample | N2 adsorption analysis | |||
|---|---|---|---|---|
| Langmuir SSA (m2 g−1) | BET SSA (m2 g−1) | Pore volume (cm3 g−1) | Micropore volume (cm3 g−1) | |
| PIM-1 powder | 945 ± 10 | 751 ± 3 | 0.50 | 0.20 |
| MOF-808 | 976 ± 5 | 792 ± 2 | 0.38 | 0.24 |
| MOF-808@Arg | 354 ± 6 | 235 ± 5 | 0.15 | 0.07 |
| MOF-808@Cys | 645 ± 4 | 518 ± 2 | 0.31 | 0.18 |
| MOF-808@Lys | 465 ± 3 | 362 ± 6 | 0.23 | 0.12 |
| PIM-1 membrane | 485 ± 7 | 350 ± 4 | 0.28 | 0.7 |
| PIM-1/MOF-808 (10 wt%) | 741 ± 3 | 560 ± 2 | 0.35 | 1.1 |
| PIM-1/MOF-808@Lys (10 wt%) | 685 ± 4 | 460 ± 1 | 0.30 | 0.9 |
Furthermore, to investigate the pore structure and its impact on gas transport behavior in the fabricated membranes, N2 adsorption BET analysis was conducted on PIM-1 powder, PIM-1 membrane, PIM-1/MOF-808 (10 wt%), and PIM-1/MOF-808@Lys (10 wt%) MMMs, as shown in Table 2 and Fig. S11. The pristine PIM-1 powder exhibited a high specific surface area (751 m2 g−1) and pore volume (0.50 cm3 g−1), consistent with its intrinsic microporosity. Upon membrane casting, these values decreased to 350 m2 g−1 and 0.28 cm3 g−1, respectively, due to partial pore collapse and chain rearrangement during film formation. Incorporation of 10 wt% MOF-808 and MOF-808@Lys into the PIM-1 matrix resulted in an increased surface area (560 and 460 m2 g−1) and pore volume (0.35 and 0.30 cm3 g−1), respectively, reflecting the contribution of the porous MOF-808 and MOF-808@Lys particles and improved free volume within the membrane structure.
In addition, the CO2 sorption isotherms at 15 °C were measured for MOF-808 and MOF-808@AA to further evaluate the enhanced affinity of amino acid-functionalized MOF-808 towards CO2 (Fig. S11c). As anticipated, amino acid functionalities markedly enhance the CO2 affinity of the parent MOF-808. The CO2 uptake at 1 bar increases progressively from 1 mmol g−1 for pristine MOF-808 to 2.5, 3.0, and 4.0 mmol g−1 for MOF-808@Arg, MOF-808@Cys, and MOF-808@Lys, respectively. It is important to highlight the notable differences in performance between the MOF-808@AA materials developed in this study and those previously reported by Chen and co-workers.66 However, under conditions approaching 1 bar, MOF-808@Lys exhibits a significantly enhanced performance, achieving an uptake approximately 4.0 mmol g−1 higher than the values reported by Chen's team. It is true that the degree of amino acid functionalization is slightly higher in our case, as well as differences in the functionalization protocols, which may significantly influence the performance of these materials.
The thermogravimetric analysis (TGA) (Fig. 2a), differential thermal analysis (DTA) (Fig. S12) and thermo-diffraction data (TDX) (Fig. S13) provide complementary insights into the thermal stability of MOF-808 and its AA-functionalized derivatives. The TGA curve for the parent MOF-808 reveals three main weight losses. The first weight loss, below 150 °C, corresponds to the desorption of physically adsorbed water (or solvent loss, as observed between 30 °C and 200 °C). The second weight loss, occurring between 200 °C and 450 °C, is linked to the release of acetate modulators and dehydration of zirconium clusters, which correlates with a loss of crystallinity in the diffraction patterns, signaling a progressive collapse of the framework. Lastly, the final weight loss at around 500 °C is attributed to the decomposition of the organic linker. Upon functionalization with AAs, the thermal degradation profile varies significantly. The MOF-808@AA samples, particularly MOF-808@Arg and MOF-808@Lys, exhibit earlier weight losses in the 200–400 °C range. Given the value of the weight loss observed between 200 and 300 °C, it is likely that a significant percentage of the amino acids installed into the framework are released within this temperature range. The degradation of the amino acid molecules incorporated within the crystal structure of MOF-808 is likely to proceed via a multistep process, in which pendant functional groups (e.g., –NH2 or –SH) are likely to be thermally released at distinct temperatures from the decomposition of the molecular backbone and the subsequent calcination of the trimesate linkers. This results in multistep degradation characterized by the occurrence of distinct intermediate decomposition events. Furthermore, the weight loss resulting from the degradation of amino acid molecules is substantially greater than that of the acetate modulators. Consequently, the contribution of amino acid decomposition to the overall mass loss observed in the TGA profile is significantly higher than that attributed to acetate modulators in the parent material.
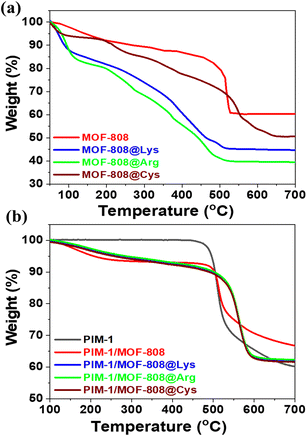 | ||
| Fig. 2 TGA curves of (a) MOF-808 and MOF-808@AA and (b) PIM-1, PIM-1/MOF808 (10 wt%) and PIM-1/MOF-808@AA (10 wt%) MMMs. | ||
The thermal stability of the MOF-808 and MOF-808@AA materials was studied in parallel by thermo-diffraction. In detail, the position and the intensity of the first two diffraction maxima were monitored during the heating process (Fig. S13). Thus, the starting point of the thermal degradation can be defined as the temperature where the diffraction peaks start to (i) displace towards higher 2θ values due to the structural collapse or (ii) lose intensity. The comparison of the relative position of the (311) plane shown in Fig. S13 indicates that there is a continuous but significant contraction of the structures above 220 °C, regardless of the functionalization installed in the framework. In parallel, the variation in the relative intensity reveals a decay for MOF-808 and Cys and Arg variants above 250 °C. In comparison, MOF-808@Lys shows a significant degradation at 210 °C.
Fig. 2b depicts the TGA curves of PIM-1 and PIM-1/MOF-808@AA MMMs, revealing the thermal characteristics of the pure polymer and the composite membranes. PIM-1 exhibits excellent thermal stability, with minimal weight loss until around 500 °C, where significant degradation of the polymer backbone occurs. The incorporation of MOF-808 or MOF-808@AA into polymer PIM-1 results in only a minor variation of the thermal profile, indicating that the MOF fillers did not adversely affect the thermal stability of PIM-1. It is interesting to note here that the relatively moderate loading of the MOF (10 wt%) is not expected to induce a drastic variation in the thermal degradation of the polymer unless the polymer itself is affected by the integration of the MOF-808 nanoparticles. It is also interesting to note that the DTA curves (Fig. S12) show a sharpening of the degradation step of PIM occurring between 500 and 600 °C when MOF-808 fillers are incorporated into the polymeric matrix. This thermal consistency and the stability enhancement observed upon adding MOF-808@AA into the polymer suggest strong interfacial compatibility between the PIM-1 polymer and the MOF-808@AA fillers.
SEM images in Fig. 3a–d and S14 provide insight into the morphology and the particle size of MOF-808 and MOF-808@AA, highlighting the absence of significant aggregation of the octahedral-like crystals. The SEM analysis shows that the functionalization of MOF-808 with amino acids (AAs) via the SALI method did not significantly affect the overall crystal morphology. However, a slight increase in the average particle size was observed for the AA-functionalized samples compared to pristine MOF-808. Specifically, the average crystal sizes were as follows: MOF-808 ≈ (450 ± 50 nm), MOF-808@Lys ≈ (750 ± 30 nm), MOF-808@Arg ≈ (800 ± 55 nm) and MOF-808@Cys ≈ (650 ± 25 nm), respectively. These findings indicate that the incorporation of amino acids leads to a noticeable increase in the crystal size while retaining the original morphology. Furthermore, Fig. 3a, b and S15 display the SEM micrographs for PIM-1 and the PIM-1/MOF-808@AA MMMs. The images provide insight into the surface morphology (the microporous character evidenced from the N2 isotherm) of the PIM-1 matrix, similar to that reported in previous studies.14,55 For MMMs, the SEM images reveal the homogeneous incorporation of MOF-808 and MOF-808@AA particles into the PIM-1 matrix. It is important to highlight the good dispersion of the MOF particles within the PIM polymer with minimal aggregation. The SEM cross-section reveals a porous structure with uniformly dispersed MOF-808 particles embedded within the PIM matrix, while the individual MOF crystals appear well integrated, contributing to a continuous and interconnected porous network throughout the MMMs. The absence of large voids or defects at the MOF–polymer interface indicates strong interfacial adhesion, which is crucial for maintaining the mechanical stability and gas separation performance of the membranes.
 | ||
| Fig. 3 (a) and (b) SEM images of (a.1) MOF-808, (a.2) PIM-1/MOF-808, (b.1) MOF-808@Lys and (b.2) PIM-1/MOF-808@Lys. | ||
In addition to SEM analysis, small-angle X-ray scattering (SAXS) was employed to further investigate the nanoscale structural modifications of the PIM-1 membrane matrix upon incorporation of MOF-808 and MOF-808@AA fillers (Fig. S16). It is important to note that SAXS provides structural information on heterogeneities within the PIM-1 matrix over a length scale ranging from approximately 200 nm down to 2 nm.
As shown in the Fig. S16, the scattering profile of the pristine PIM-1 membrane exhibits a smooth curve with a characteristic power-law decay, indicative of large, polydisperse domains exhibiting fractal-like organisation. This scattering behaviour is attributed to the presence of micron-sized air-filled voids within the membrane. Upon inclusion of MOF-808 and its amino acid derivatives, a distinct diffraction peak emerges in the high-q region, corresponding to the first Bragg reflection of MOF-808 (see Fig. S16). Notably, the SAXS profiles of all MOF-containing membranes display similar features, suggesting that the inclusion of the fillers does not disrupt the overall hierarchical organisation of the polymer matrix across the studied length scales. The primary difference between these membranes and the pristine PIM-1 lies in the scattering intensity, which is attributed to the variation in X-ray scattering length density between the MOF particles and the polymer (i.e. the MOF content of the membranes). In fact, all samples maintain the same power-law trend, indicating comparable fractal-like structural organisation.67–70
3.3. Separation performance of PIM-1/MOF-808@AA MMMs
The CO2 permeability and CO2/CH4 selectivity of pristine PIM-1, PIM-1/MOF-808 and PIM-1/MOF-808@AA MMMs are provided in Table S2 and Fig. 4a, b. The incorporation of 10 wt% MOF-808 and its amino acid-functionalized variants (MOF-808@AA) into PIM-1 resulted in marked improvements in both permeability and CO2/CH4 selectivity (Fig. 4a). The PIM-1 membrane exhibits a CO2 permeability of 9564 ± 278 Barrer and a CO2/CH4 selectivity of 17.9 ± 2.0. The incorporation of MOF-808 into the PIM-1 matrix led to a significant increase in both values, yielding a CO2 permeability of 11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 370 ± 235 Barrer and a selectivity of 18.6 ± 2.0. Notably, the PIM-1/MOF-808@Lys membrane exhibited the highest CO2 permeability of 14
370 ± 235 Barrer and a selectivity of 18.6 ± 2.0. Notably, the PIM-1/MOF-808@Lys membrane exhibited the highest CO2 permeability of 14![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 354 ± 201 Barrer and a CO2/CH4 selectivity of 26.4 ± 1.8 before aging. Similarly, PIM-1/MOF-808@Arg achieved a permeability of 14
354 ± 201 Barrer and a CO2/CH4 selectivity of 26.4 ± 1.8 before aging. Similarly, PIM-1/MOF-808@Arg achieved a permeability of 14![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 056 ± 193 Barrer and a selectivity of 18.5 ± 1.8, while PIM-1/MOF-808@Cys shows a permeability of 13
056 ± 193 Barrer and a selectivity of 18.5 ± 1.8, while PIM-1/MOF-808@Cys shows a permeability of 13![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 877 ± 244 Barrer and a selectivity of 18.5 ± 1.6.
877 ± 244 Barrer and a selectivity of 18.5 ± 1.6.
 | ||
| Fig. 4 CO2 permeability and mixed CO2/CH4 selectivity for the bare PIM-1 membrane and PIM-1/MOF-808 and PIM-1/MOF-808@AA (10 wt%) MMMs: (a) before aging and (b) after aging (60 days). | ||
The physical aging characteristics of the membranes were also assessed in the second step of this study. After 60 days of aging under dark conditions and room-temperature (out of the permeation setup), a decrease in CO2 permeability and a slight increase in CO2/CH4 selectivity were observed across all MMMs, as shown in Table S2 and Fig. 4b. Previous studies have demonstrated that PIM-1, known for its high free fractional volume (FFV), undergoes a physical aging. This aging phenomenon is typical in PIM-type polymers,71 where the polymer structure gradually reorients towards a pseudo-equilibrium state. This local rearrangement reduces the FFV, causing a drop in permeability while slightly improving selectivity.22,63 However, contrary to the bare polymer membrane, PIM-1/MOF-808@Lys continued to outperform the other MMMs, retaining a CO2 permeability of 13![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 241 ± 244 Barrer and a CO2/CH4 selectivity of 27.5 ± 1.5. This represents a 10% decrease in permeability but a 5% enhancement in selectivity in comparison to the bare PIM-1. Similarly, PIM-1/MOF-808@Arg showed a permeability of 13
241 ± 244 Barrer and a CO2/CH4 selectivity of 27.5 ± 1.5. This represents a 10% decrease in permeability but a 5% enhancement in selectivity in comparison to the bare PIM-1. Similarly, PIM-1/MOF-808@Arg showed a permeability of 13![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 015 ± 199 Barrer with a selectivity of 20.9 ± 1.3, while PIM-1/MOF-808@Cys achieved a permeability of 12
015 ± 199 Barrer with a selectivity of 20.9 ± 1.3, while PIM-1/MOF-808@Cys achieved a permeability of 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 135 ± 185 Barrer and a selectivity of 20.5 ± 1.4. The increase in selectivity after aging can be attributed to the reduced FFV within the polymer, which limits CH4 diffusion more than that of CO2, thereby enhancing the CO2/CH4 selectivity. These findings underscore that AA modifications, particularly with Lys, not only improve the initial separation performance but also enhance the long-term stability of the MMMs, highlighting the potential of these materials for efficient and durable CO2/CH4 separation.
135 ± 185 Barrer and a selectivity of 20.5 ± 1.4. The increase in selectivity after aging can be attributed to the reduced FFV within the polymer, which limits CH4 diffusion more than that of CO2, thereby enhancing the CO2/CH4 selectivity. These findings underscore that AA modifications, particularly with Lys, not only improve the initial separation performance but also enhance the long-term stability of the MMMs, highlighting the potential of these materials for efficient and durable CO2/CH4 separation.
Last but not least, the permeability and selectivity data obtained for all the membranes were included within the Robeson plot shown in Fig. 5a. The slope of the upper bound (UB) lines for selectivity-permeability is dependent on the kinetic diameter of the permeating gas pairs, and any variations along the slope are directly linked with their diffusivities.3,4 The position of our data in Fig. 5a clearly indicates that the addition of unmodified MOF-808 to the PIM-1 matrix improves its performance enough to surpass the 2018 UB line, but still does not exceed the 2019 UB limit for the CO2/CH4 separation. In contrast, the performances of MMMs PIM-1/MOF-808@Arg and PIM-1/MOF-808@Cys approach the 2019 Robeson UB line.
 | ||
| Fig. 5 (a) Mixed (CO2/CH4) selectivity as a function of CO2 permeability linked with the 1991,4 2008 (ref. 12) and 2019 (ref. 22) upper-bounds before and after aging for pristine PIM-1, PIM-1/MOF-808 (10 wt%), and PIM-1/MOF-808@AA (10 wt%) MMMs. (b) High-pressure CO2 adsorption isotherms for MOF-808 and MOF-808@Lys materials. | ||
All in all, taking into consideration all the results obtained, it is clear that the integration of MOF-808 nanoparticles into polymer PIM-1 induces an alteration of the pore structure of the MMM, improving its CO2 permeability, but at the same time, maintaining its selectivity. The uniformly dispersed and interconnected porous structure observed in PIM-1/MOF-808 MMMs, which is significantly different from that observed in the parent PIM-1, could tentatively explain this improvement. Small variations in the structure of PIM-1/MOF-808@AA MMMs are also observed when compared to PIM-1/MOF-808, providing experimental evidence that can further explain the additional permeability gain exhibited by the membranes when MOF-808 is functionalized with AAs. In comparison, the selectivity gain observed for MOF-808 and MOF-808@AA decorated PIM-1 is not so straightforward to explain. If the DFT-based calculations of the adsorption energies for CO2 by the amino groups and side chains of amino acids are considered, Arg-based MMMs should show the best performance in comparison to Lys and Cys ones, since arginine shows the largest interaction energies with CO2, both when adsorbed in the guanidinium side chain or at the α-NH2 group. Nevertheless, the experimental results point out that only PIM-1/MOF-808@Lys increases its selectivity in comparison to the other studied mixed matrix membranes. When comparing the CO2 adsorption capacity and affinity of AAs in solution, lysine outperforms arginine.72 The study reported recently by Lyu et al. demonstrated that the CO2 uptake of MOF-808@Lys at low relative pressures also improves the adsorption capacity values of other AA functionalized MOF-808 variants.73 Nevertheless, the adsorption capacity values for arginine and cysteine variants were not reported in this last investigation.
To complement previous investigations, high-pressure CO2 adsorption isotherms were studied for MOF-808 and MOF-808@Lys materials. Before discussing the results, it is important to emphasize that adsorption and diffusion are distinct processes that cannot be directly correlated. However, CO2 selectivity values obtained from adsorption can provide qualitative insights into the variations observed in the selectivity of PIM-1/MOF-808@AA membranes.
Notably, the incorporation of lysine into the MOF-808 structure significantly alters the isotherm, as shown in Fig. 5b. AA functionalization enhances adsorption in the low CO2 pressure range (0–0.25 bar). Beyond this pressure, CO2 uptake in MOF-808@Lys is lower than in the parent MOF-808. This is expected, as lysine molecules partially occupy the pore space, facilitating CO2 interaction at lower pressures but hindering further molecular access at higher pressures.
The evaluation of CO2 permeability (the single and mixed gases) as a function of pressure up to 6 bar (the highest pressure that our set-up can reach) for PIM-1, PIM/MOF-808, and PIM/MOF-808@AA (10 wt%) MMMs is presented in Table S3 and Fig. S17. The results show that the CO2 permeability (PCO2) increases progressively with pressure up to 6 bar for all tested membranes. Among the studied membranes, the PIM-1/MOF-808@Lys (10 wt%) membranes consistently exhibited the highest PCO2 values across the pressure range, followed by PIM-1/MOF-808@AA (10 wt%), PIM-1/MOF-808 (10 wt%) and pristine PIM-1. A moderate increase in PCO2 was also observed with increasing pressure, particularly in the membranes containing functionalized MOFs. This behavior is attributed to the dual-sorption nature of glassy polymers such as PIM-1, where elevated pressures activate additional free volume, thereby enhancing CO2 uptake. The incorporation of lysine-functionalized MOFs introduces CO2-philic groups (e.g., –NH2 and –COOH), which strengthen gas–MOF interactions and increase CO2 solubility within the matrix. Interestingly, CO2 permeability values obtained from mixed-gas measurements were slightly higher than those from single-gas experiments, a trend that is often observed in microporous systems such as PIM-1-based MMMs.47,49,74–76 This enhancement is primarily due to the synergistic effects between gas species in mixed environments. In particular, CO2, being more condensable than CH4, preferentially interacts with the available sorption sites and microporous domains, displacing CH4 and facilitating a more efficient transport through the membrane. This competitive sorption mechanism, combined with the tailored interfacial compatibility introduced by the functionalized MOF, can lead to higher overall permeability and selectivity under mixed-gas conditions. Such behavior aligns well with previous findings reported for PIM-1-based MMM systems.47,49,74–76
The single CO2 permeability for PIM-1, PIM-1/MOF-808 (10 wt%), and PIM-1/MOF-808@Lys (10 wt%) was conducted as a function of different temperatures (35, 50, 75 and 100 °C), as shown in Table S4 and Fig. S18. The results showed that the single CO2 permeability decreases with increasing temperature for all membranes, with the highest permeability values recorded for PIM-1/MOF-808@Lys (10 wt%). This behavior can be explained by the exothermic nature of gas sorption in glassy polymers, where sorption capacity and consequently gas solubility decline as temperature increases.77 While higher temperatures can enhance gas diffusivity by increasing polymer chain mobility, in our case the reduction in solubility is the dominant factor, resulting in an overall drop in permeability, with negative values of activation energy for CO2.77 From a plasticization perspective, this observation is highly relevant, as plasticization in glassy polymers is typically initiated by excessive gas sorption, which leads to polymer swelling and loss of selectivity. The reduced solubility of CO2 at elevated temperatures limits polymer chain swelling, thereby minimizing the risk of plasticization. Consequently, the observed trend of decreasing CO2 permeability with temperature not only reflects the thermodynamic sorption behavior of the membranes but also provides evidence of their stability under conditions where plasticization effects could otherwise compromise long-term performance. Therefore, the combined pressure and temperature dependent permeability studies suggest that the incorporation of MOF-808, particularly functionalized with lysine, should improve the plasticization resistance of PIM-1 based membranes while maintaining high gas separation performance. This observed behavior further underscores the promising potential of PIM-1/MOF composite membranes in realistic gas separation processes.
Furthermore, the activation energy (Ea) for CO2 permeability (PCO2) was calculated for the tested MMMs at different temperatures (35–100 °C), as represented by Arrhenius plots of the results obtained (ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) PCO2vs. 1/T),78 as shown in Table S4 and Fig. S18b. The calculated values revealed a distinct trend, decreasing in magnitude from −12.70 kJ mol−1 for pure PIM-1 to −11.72 kJ mol−1 for the PIM-1/MOF-808 (10 wt%) MMM, and further to −9.77 kJ mol−1 for the PIM-1/MOF-808@Lys (10 wt%) MMM. This progressive reduction in Ea signifies a fundamental change in the CO2 transport mechanism. The high negative Ea of pristine PIM-1 is characteristic of a solution-diffusion process in a rigid, glassy polymer matrix, where permeability is strongly hindered by decreasing temperature.77 The incorporation of MOF-808 slightly mitigates this temperature sensitivity, likely by introducing additional diffusion pathways. Most notably, the significantly lower Ea for the lysine-functionalized composite strongly suggests the emergence of a facilitated transport mechanism. The amine groups on the lysine moieties provide reactive sites for reversible reaction with CO2, effectively reducing the energy barrier for permeation and making the membrane's performance more resilient to operational temperature variations.
PCO2vs. 1/T),78 as shown in Table S4 and Fig. S18b. The calculated values revealed a distinct trend, decreasing in magnitude from −12.70 kJ mol−1 for pure PIM-1 to −11.72 kJ mol−1 for the PIM-1/MOF-808 (10 wt%) MMM, and further to −9.77 kJ mol−1 for the PIM-1/MOF-808@Lys (10 wt%) MMM. This progressive reduction in Ea signifies a fundamental change in the CO2 transport mechanism. The high negative Ea of pristine PIM-1 is characteristic of a solution-diffusion process in a rigid, glassy polymer matrix, where permeability is strongly hindered by decreasing temperature.77 The incorporation of MOF-808 slightly mitigates this temperature sensitivity, likely by introducing additional diffusion pathways. Most notably, the significantly lower Ea for the lysine-functionalized composite strongly suggests the emergence of a facilitated transport mechanism. The amine groups on the lysine moieties provide reactive sites for reversible reaction with CO2, effectively reducing the energy barrier for permeation and making the membrane's performance more resilient to operational temperature variations.
The solubility and diffusion coefficients obtained from time-lag experiments for PIM-1, PIM/MOF-808, and PIM/MOF-808@AA are presented in Table S5 and Fig. S19. The measurements were specifically performed at 10 wt% MOF loading, as supported by our previous studies,47–49 which demonstrated that this concentration provides an optimal balance between performance and material dispersion. Higher loadings, such as 15 wt%, have been reported to cause MOF particle agglomeration, which can disrupt the membrane microstructure and reduce gas separation efficiency. In comparison to pristine PIM-1, the incorporation of MOF-808 led to notable improvements in both solubility and diffusion coefficients, attributed to the porous nature of MOF-808 and its affinity for CO2. The PIM-1/MOF-808@Lys (10 wt%) membrane exhibited the highest CO2/CH4 selectivity among all tested samples. This enhancement is attributed to a balanced contribution from both increased solubility and moderately improved diffusion selectivity. The functionalization of MOF-808 with lysine introduces polar amino groups that interact favorably with CO2, leading to enhanced gas sorption and a higher solubility coefficient. This effect is especially pronounced in the solubility selectivity, which reflects the stronger affinity of the functionalized filler toward CO2 over CH4. While it is generally understood that strong sorption interactions may slow down gas diffusion, our time-lag measurements confirm that the diffusion coefficient of CO2 in the PIM/MOF-808@Lys membrane remains sufficiently high. This indicates that lysine functionalization does not significantly hinder gas mobility. Instead, it appears to promote better filler dispersion and improved interfacial compatibility with the PIM-1 matrix, thus facilitating selective and efficient gas transport. Overall, the combination of enhanced CO2-philic interactions and preserved diffusivity explains the superior gas separation performance of the PIM-1/MOF-808@Lys MMM.
Finally, to evaluate the influence of MOF loading on gas separation performance, a systematic study was conducted using PIM-1-based MMMs incorporating MOF-808 and its amino acid-functionalized derivatives (MOF-808@Lys and MOF-808@Arg) at different loadings (5, 10, and 15 wt%). As shown in Fig. S20 and Table S6, the CO2 permeability and CO2/CH4 selectivity progressively increased with MOF loading from 5 to 10 wt%, reaching the best values at 10 wt%. This enhancement is attributed to the presence of well-dispersed MOF particles that introduce additional free volume and diffusion pathways through their porous structures and their interface with the PIM-1 matrix. The incorporation of Zr-based MOFs also contributes to selective gas sorption due to their intrinsic CO2-philic character and the presence of functional groups in the MOF linkers, which improve compatibility with the polymer matrix. However, at 15 wt% loading, a slight decline in both CO2 permeability and solubility was observed. This decrease is likely due to MOF particle agglomeration, which can lead to non-uniform filler dispersion, partial pore blockage, and the formation of non-selective voids, ultimately hindering efficient gas transport. Among the functionalized MOFs, PIM-1/MOF-808@Lys (10 wt%) delivered the best performance, achieving a CO2 permeability of 14![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 354 ± 201 Barrer and a CO2/CH4 selectivity of 26.4 ± 1.8. Interestingly, although MOF-808@Arg contains more amino groups than lysine, the PIM-1/MOF-808@Lys membranes outperformed the arginine-based ones. This could be due to several factors: (i) lysine offers a better balance between sorption and diffusion, likely due to its simpler structure and less steric hindrance, which facilitates gas transport; (ii) the stronger interaction between lysine-functionalized MOFs and the PIM-1 matrix may promote a more uniform filler distribution and improved interface adhesion; and (iii) the larger side groups in arginine may impose stricter diffusion constraints within the membrane, reducing overall gas mobility despite the higher density of amino groups. These results highlight the importance of optimizing both MOF loading and surface functionalization. While the general trend of improved gas separation with increasing MOF content was observed, the optimum performance was consistently achieved at 10 wt% loading for both MOF-808@Lys and MOF-808@Arg systems, with lysine yielding the best combination of permeability and selectivity due to favorable solubility–diffusion trade-offs and better interfacial compatibility mentioned previously in Table S5 and Fig. S19. In line with these findings, the next section demonstrates that the adsorption sites of the Lys-modified MOF are much more energetic for CO2 than for CH4, in agreement with the MMM performance shown above.
354 ± 201 Barrer and a CO2/CH4 selectivity of 26.4 ± 1.8. Interestingly, although MOF-808@Arg contains more amino groups than lysine, the PIM-1/MOF-808@Lys membranes outperformed the arginine-based ones. This could be due to several factors: (i) lysine offers a better balance between sorption and diffusion, likely due to its simpler structure and less steric hindrance, which facilitates gas transport; (ii) the stronger interaction between lysine-functionalized MOFs and the PIM-1 matrix may promote a more uniform filler distribution and improved interface adhesion; and (iii) the larger side groups in arginine may impose stricter diffusion constraints within the membrane, reducing overall gas mobility despite the higher density of amino groups. These results highlight the importance of optimizing both MOF loading and surface functionalization. While the general trend of improved gas separation with increasing MOF content was observed, the optimum performance was consistently achieved at 10 wt% loading for both MOF-808@Lys and MOF-808@Arg systems, with lysine yielding the best combination of permeability and selectivity due to favorable solubility–diffusion trade-offs and better interfacial compatibility mentioned previously in Table S5 and Fig. S19. In line with these findings, the next section demonstrates that the adsorption sites of the Lys-modified MOF are much more energetic for CO2 than for CH4, in agreement with the MMM performance shown above.
3.4. Dynamics of MOF-808@Lys1+ studied by INS
Inelastic neutron scattering (INS) spectroscopy is a technique that extracts precise information about the molecular vibrations – especially those associated with hydrogen atoms. Indeed, the analysis of the INS data opens the room to understand the dynamics of specific atoms or molecules and of the surrounding chemical environment disturbing them. In this study, INS was employed for two aims: first, to elucidate the binding modes of the zwitterion form of lysine amino acid to MOF-808, and second, to elucidate the adsorption sites and their energy of interaction with CO2 and CH4 molecules upon adsorption. This study focused on lysine decorated MOF-808, since it is the one showing the best performance in terms of selectivity of CO2 and CH4 separation when integrated in PIM membranes.Before presenting the INS data and the DFT calculations used for interpretation, it is essential to define the protonation state of the amino acid once stabilized within MOF-808. As detailed in the experimental section, the slightly acidic pH of the lysine solution used for functionalization suggests that lysine is stabilized in its zwitterionic form (hereafter referred to as Lys+). Notably, this zwitterionic form carries an overall positive charge, due to the presence of two ammonium groups and one carboxylate group. Upon incorporation into MOF-808, the carboxylate group coordinates to the zirconium clusters, anchoring the molecule to the framework, while the amine groups remain exposed within the pore space. We hypothesize that, similar to amines in aqueous solution at intermediate pH, the –NH2 groups of lysine interact with surrounding water molecules, accept a proton, and become stabilized as –NH3+/OH− ion pairs. Therefore, the models used for DFT minimization and subsequent INS data calculations are based on the protonated form of the lysine molecule, hereafter referred to as Lys+.
Fig. 6a shows the experimental INS spectra for Lys1+, MOF-808 (red line) and MOF-808@Lys1+, containing the GDOS as a function of wavenumber or energy. Preliminary band identification enabled correlating the specific vibrational bands M1 (939 cm−1), M2 (1093 cm−1) and M3 (1420 cm−1) of MOF-808 with the bending of structural hydroxyls of the cluster (M1–M3 being bands marked in Fig. 4a),79 bending of C–H bonds in the BTC3− linkers,79 and the symmetrical stretching of the O–C–O groups belonging to the trimesate linkers or acetate modulators, respectively.80,81
For MOF-808@Lys1+, the M1 and M2 bands remained unaltered after functionalization of MOF-808, while the M3 signal disappeared. The last variation suggests that Lys1+ is likely displacing the acetate modulators bonded to the linker defective positions of the Zr6 clusters. On the other hand, the bands associated with Lys1+ have been identified in the INS spectra in Fig. 6a and labelled with the letters A–I. The vibrational bands of Lys1+ obtained from the DFT calculation have been assigned using the same codes in Fig. S16 and Table S3. Most of the band associated with the vibrational modes of Lys1+ remain unaltered when the amino acid was integrated into MOF-808. These signals are marked with blue strips in Fig. 6a and b. In contrast, these bands associated with Lys1+ that show intensity or position variations in the INS spectra of free and Lys1+ immobilized into MOF-808 are marked with violet stripes in the same plot. It is clear from the comparison between the spectra that the bands labeled as B and C disappeared when lysine is installed into the structure of MOF-808, whilst the bands labeled as A and D to I (marked as such in Fig. 4a) are still present after the functionalization. In order to assist the interpretation of the INS data when Lys1+ is included in the MOF-808 structure, the vibrational density of states (VDOS) of the amino acid in its zwitterion form (Fig. 6b and S21) was calculated. A simplified description of the assignment of vibrational modes is provided below, whilst a detailed description of each of the individual signals has been supplied in the SI (Table S7 and Fig. S22). The same letter and color codes shown in Fig. 6a have been employed to identify the calculated vibrational modes (Table S7 and Fig. S22). Upon qualitative visualization of the vibrational normal modes associated with Lys1+ shown in Fig. S22, it was identified that its skeleton loses some degree of vibrational freedom, inducing the disappearance of the B (770 cm−1) and C (884 cm−1) bands. The Lys1+ model obtained by the DFT calculation allowed identification of the band B as rocking (ρ) of H–C–H bonds and twisting (τ) of H–C–H and H–N–H groups. Similarly, the C band was linked to the τ and ρ vibrational modes of different H–C–H groups, and to the scissoring (δ) of H–C–N and H–N–H groups. As suggested by the calculated INS spectrum (Fig. 6b and S23), the two bands disappearing during the Lys1+ anchoring to the MOF have contribution from carbon atoms, gC, confirming that the main chain and the lateral amino group of the amino acid have lost vibrational freedom when the amino acid is anchored to the clusters (Fig. 4b). In addition, the F and I bands (see orange stripes in Fig. 6b) are associated mainly with the vibrations of the H atoms, and the A, D, E, G and H bands (see blue stripes in Fig. 6b) are linked to both the contribution of H and C vibrations. All in all, the variations in the INS spectra indicate that Lys1+ bonded to the clusters acquired rigidity mainly in the main carbon skeleton, but still does not lose all its vibrational freedom associated with the hydrogen bonds of CH2 and NH3 groups.
Moreover, INS experiments were conducted on MOF-808@Lys1+ loaded with 1 bar of CO2. Fig. 6c presents the experimental GDOS for the parent MOF-808@Lys1+ under vacuum (blue line) and after exposure to 1 bar of CO2 (magenta line). The most notable changes observed upon CO2 adsorption are (I) the blueshift of the A band from 726 to 733 cm−1, attributed to slight variations in the rocking vibration of the C–H bonds within the Lys skeleton, and (II) the emergence of the N band at 1180 cm−1 (see Table S7). The other signals remain unchanged, with only minor variations in intensity. When calculating the theoretical VDOS for Lys1+, Lys1+ interacting with a CO2 molecule or Lys1+ in its carbamate form, the third spectrum is the one that fits better with the experimental data (Fig. S21–S23). In detail, the simulation achieved to reproduce the blueshift of the A band from 718 to 735 cm−1,82 in parallel, the generation of the N band N 1188 cm−1, which is ascribed to the bending vibration of the lateral O–H bond in the carbamic acid form of Lys (see Fig. 6d). This is supported by electrostatic potential maps, which show that the lateral regions of Lys exhibit the highest potential distribution, making them prone to interacting with the negative poles of CO2 molecules seeking charge balance. This initial interaction between CO2 and the lateral Lys chain is likely the first step in the subsequent chemisorption reaction, leading to the formation of carbamic acid species under dry conditions.
At this stage, taking into consideration the conclusion of INS analysis performed under static absorption conditions, while we do not rule out the formation of carbamate species during the CO2/CH4 gas separation process in PIM/MOF-808@AA membranes, we employed DFT calculations to identify the preferential adsorption sites of CO2 and CH4 molecules in MOF-808@Lys instead of the MOF-808@Lys-carbamate form. Additionally, we analyzed their interaction energies with Lys1+ as gas loading increases, simulating the effect of pressure in the separation process. Regardless of the gas type, DFT identifies five distinct interaction sites: two associated with the terminal ammonium group, two linked to the lateral ammonium site and one specific to CO2, related to the CH2 group of Lys. For the latter, it remains to be confirmed whether the adsorption is primarily stabilized by surrounding molecules rather than the CH2 groups themselves. Overall, the interaction energies at these sites are three to six times higher for CO2 compared to CH4, a trend that aligns with the selectivity enhancement observed in MMM PIM/MOF-808@Lys compared to the bare PIM-1 membrane.
4. Conclusion
This study has demonstrated that incorporating amino acid-functionalized MOF-808 into PIM-1 MMMs significantly enhances CO2/CH4 separation performance, improving both permeability and selectivity. Functionalization of MOF-808 with amino acids possessing different lateral chains and amino groups (e.g., lysine, arginine, and cysteine) modified the textural properties and BET surface area of the MOF, resulting in enhanced CO2 adsorption capacity and affinity. Consequently, PIM-1/MOF-808@AA MMMs exhibited superior gas transport performance, selectivity, and long-term stability compared to the pristine PIM-1 membranes.Among the tested membranes, PIM-1/MOF-808@Lys achieved the highest CO2 permeability and CO2/CH4 selectivity, approaching the 2019 Robeson upper bound. Optimized MMMs with 10 wt% MOF loading maintained excellent performance even after 60 days of aging, highlighting their suitability for practical CO2/CH4 separation applications such as natural gas purification and biogas upgrading.
Complementary INS and DFT analyses provided molecular-level insight into the enhanced performance. Zwitterionic lysine (Lys1+) anchors to the defect sites on MOF-808, displacing acetate groups and restricting vibrational freedom, particularly in the CH2 and NH3+ moieties. This creates multiple CO2 interaction sites, facilitating carbamic acid formation and selective binding over CH4. The strong correlation between molecular interactions and macroscopic gas transport highlights lysine-functionalized MOF-808 as an effective strategy to tune selectivity and permeability in PIM-based MMMs.
Overall, this work provides a clear structure–property relationship, demonstrating that amino acid functionalization of MOF-808 provides a versatile and efficient approach to develop high-performance, stable MMMs for advanced CO2/CH4 separation applications.
Author contributions
D. Refaat: investigation, methodology, validation, formal analysis, writing-original draft, writing-review & editing. H. D. Martínez-Hernández, M. Jimenez-Ruiz, V. Galván, and V. Petrenko: investigation, validation, formal analysis, writing-original draft. M. Yahia, R. Fernández de Luis, and J. Coronas: conceptualization, methodology, visualization, supervision, funding acquisition, writing-original draft, writing-review & editing.Conflicts of interest
The authors declare that they have no financial or commercial conflicts of interest.Data availability
The data supporting the findings of this study are available within the SI. Additional data can be made available by the authors upon reasonable request. Supplementary information: the SI provides detailed experimental and computational data, including 1H-NMR and GPC analysis, X-ray diffraction, N2 and CO2 adsorption isotherms, thermodiffraction data, SEM and SAXS characterization, gas separation measurements under various conditions, solubility and diffusion studies, as well as computational analyses of Vibrational Density of States (VDOS) and DFT calculations. See DOI: https://doi.org/10.1039/d5ta04211a.Acknowledgements
This investigation received financial support through multiple funding sources: MCIN/AEI/10.13039/501100011033 and ERDF (“A way of making Europe”) provided support via grants PID2022-138582OB-I00 and PID2021-122940OB-C31; the European Union's NextGenerationEU program and MCIN/AEI/10.13039/501100011033 jointly funded TED2021-130621B-C4; and MICIU/AEI/10.13039/501100011033 supported CEX2023-001286-S. Researcher M. Yahia was supported through the María Zambrano fellowship program, financed by both the European Union's NextGenerationEU initiative and Spain's Ministerio de Universidades. Additional financial backing came from the Government of Aragón (T68_23R). The research team expresses sincere appreciation for the technical resources and instrumentation made available by Universidad de Zaragoza, particularly through the ELECMI ICTS Laboratorio de Microscopías Avanzadas national research facility and the Servicio General de Apoyo a la Investigación-SAI. H. D. Martinez-Hernandez acknowledges the Institute Laue-Langevin and the Spanish MCIN for his PhD/CFR contract (Reference: ESP-2-2023). Also, H. D. Martinez-Hernandez, M. Jiménez Ruiz and R. Fernández de Luis thank J. M. Porro for his contribution to the discussion on the experimental setup and data treatment of neutron scattering experiments. The authors gratefully acknowledge Dr Oleksandr Tomchuk (STFC RAL, UK) for the help with SAXS measurements.References
- Y. Zhang, J. Sunarso, S. Liu and R. Wang, Int. J. Greenhouse Gas Control, 2013, 12, 84–107 CrossRef CAS.
- H. Yang, Z. Xu, M. Fan, R. Gupta, R. B. Slimane, A. E. Bland and I. Wright, J. Environ. Sci., 2008, 20, 14–27 CrossRef CAS PubMed.
- B. D. Freeman, Macromolecules, 1999, 32, 375–380 CrossRef CAS.
- L. M. Robeson, J. Membr. Sci., 1991, 62, 165–185 CrossRef CAS.
- L. M. Robeson, B. D. Freeman, D. R. Paul and B. W. Rowe, J. Membr. Sci., 2009, 341, 178–185 CrossRef CAS.
- S. Basu, A. L. Khan, A. Cano-Odena, C. Liu and I. F. J. Vankelecom, Chem. Soc. Rev., 2010, 39, 750–768 RSC.
- X. Zou and G. Zhu, Adv. Mater., 2018, 30, 1700750 CrossRef PubMed.
- R. W. Baker and B. T. Low, Macromolecules, 2014, 47, 6999–7013 CrossRef CAS.
- S. Wang, X. Li, H. Wu, Z. Tian, Q. Xin, G. He, D. Peng, S. Chen, Y. Yin and Z. Jiang, Energy Environ. Sci., 2016, 9, 1863–1890 RSC.
- G. Dong, H. Li and V. Chen, J. Mater. Chem. A, 2013, 1, 4610–4630 RSC.
- M. R. Khdhayyer, E. Esposito, A. Fuoco, M. Monteleone, L. Giorno, J. C. Jansen, M. P. Attfield and P. M. Budd, Sep. Purif. Technol., 2017, 173, 304–313 CrossRef CAS.
- L. M. Robeson, J. Membr. Sci., 2008, 320, 390–400 CrossRef CAS.
- C. E. Powell and G. G. Qiao, J. Membr. Sci., 2006, 279, 1–49 CrossRef CAS.
- M. Yahia, Q. N. P. Le, N. Ismail, M. Essalhi, O. Sundman, A. Rahimpour, M. M. Dal-Cin and N. Tavajohi, Microporous Mesoporous Mater., 2021, 312, 110761 CrossRef CAS.
- T.-S. Chung, L. Y. Jiang, Y. Li and S. Kulprathipanja, Prog. Polym. Sci., 2007, 32, 483–507 CrossRef CAS.
- N. A. H. M. Nordin, A. F. Ismail, A. Mustafa, R. S. Murali and T. Matsuura, RSC Adv., 2015, 5, 30206–30215 RSC.
- Y. Li, T.-S. Chung, C. Cao and S. Kulprathipanja, J. Membr. Sci., 2005, 260, 45–55 CrossRef CAS.
- D.-Y. Kang and J. S. Lee, Langmuir, 2023, 39, 2871–2880 CrossRef CAS PubMed.
- C. A. Scholes and S. Kanehashi, Membranes, 2019, 9, 41 CrossRef PubMed.
- N. B. McKeown and P. M. Budd, Chem. Soc. Rev., 2006, 35, 675–683 RSC.
- N. B. McKeown, Curr. Opin. Chem. Eng., 2022, 36, 100785 CrossRef.
- B. Comesaña-Gándara, J. Chen, C. G. Bezzu, M. Carta, I. Rose, M.-C. Ferrari, E. Esposito, A. Fuoco, J. C. Jansen and N. B. McKeown, Energy Environ. Sci., 2019, 12, 2733–2740 RSC.
- P. Llewellyn, G. Maurin and J. Rouquerol, in Adsorption by Powders and Porous Solids, ed. F. Rouquerol, J. Rouquerol, K. S. W. Sing, P. Llewellyn and G. Maurin, Academic Press, Oxford, 2nd edn, 2014, pp. 565–610, DOI:10.1016/B978-0-08-097035-6.00014-0.
- A. Karmakar, A. V. Desai and S. K. Ghosh, Coord. Chem. Rev., 2016, 307, 313–341 CrossRef CAS.
- E. Adatoz, A. K. Avci and S. Keskin, Sep. Purif. Technol., 2015, 152, 207–237 CrossRef CAS.
- B. Zornoza, C. Tellez, J. Coronas, J. Gascon and F. Kapteijn, Microporous Mesoporous Mater., 2013, 166, 67–78 CrossRef CAS.
- J. M. Luque-Alled, C. Moreno and P. Gorgojo, Curr. Opin. Chem. Eng., 2023, 39, 100901 CrossRef.
- J. M. Luque-Alled, A. W. Ameen, M. Alberto, M. Tamaddondar, A. B. Foster, P. M. Budd, A. Vijayaraghavan and P. Gorgojo, J. Membr. Sci., 2021, 623, 118902 CrossRef CAS.
- Y. Feng, W. Yan, Z. Kang, X. Zou, W. Fan, Y. Jiang, L. Fan, R. Wang and D. Sun, Chem. Eng. J., 2023, 142873 CrossRef CAS.
- M. Rafiul Hasan, A. Moriones, M. Malankowska and J. Coronas, Sep. Purif. Technol., 2023, 304, 122355 CrossRef CAS.
- N. A. H. M. Nordin, A. F. Ismail, A. Mustafa, R. S. Murali and T. Matsuura, RSC Adv., 2014, 4, 52530–52541 RSC.
- A. Sabetghadam, B. Seoane, D. Keskin, N. Duim, T. Rodenas, S. Shahid, S. Sorribas, C. L. Guillouzer, G. Clet, C. Tellez, M. Daturi, J. Coronas, F. Kapteijn and J. Gascon, Adv. Funct. Mater., 2016, 26, 3154–3163 CrossRef CAS PubMed.
- S. Hwang, W. S. Chi, S. J. Lee, S. H. Im, J. H. Kim and J. Kim, J. Membr. Sci., 2015, 480, 11–19 CrossRef CAS.
- S. Sorribas, B. Zornoza, C. Téllez and J. Coronas, J. Membr. Sci., 2014, 452, 184–192 CrossRef CAS.
- D. Liu, Y. Wu, Q. Xia, Z. Li and H. Xi, Adsorption, 2013, 19, 25–37 CrossRef CAS.
- P. Tanvidkar, S. Appari and B. V. R. Kuncharam, Rev. Environ. Sci. Bio/Technol., 2022, 21, 539–569 CrossRef CAS.
- L. Sze Lai, Y. Fong Yeong, K. Keong Lau and M. Shariff Az, J. Appl. Sci., 2014, 14, 1161–1167 CrossRef.
- A. Valverde, G. I. Tovar, N. A. Rio-López, D. Torres, M. Rosales, S. Wuttke, A. Fidalgo-Marijuan, J. M. Porro, M. Jiménez-Ruiz, V. García Sakai, A. García, J. M. Laza, J. L. Vilas-Vilela, L. Lezama, M. I. Arriortua, G. J. Copello and R. Fernández de Luis, Chem. Mater., 2022, 34, 9666–9684 CrossRef CAS.
- R. Newar, N. Akhtar, N. Antil, A. Kumar, S. Shukla, W. Begum and K. Manna, Angew. Chem., Int. Ed., 2021, 60, 10964–10970 CrossRef CAS PubMed.
- J. Canivet, S. Aguado, G. Bergeret and D. Farrusseng, Chem. Commun., 2011, 47, 11650–11652 RSC.
- Y. Zhao, Z. Zhang, Q. Gao and W. Wei, ACS Appl. Nano Mater., 2025, 8, 7190–7199 CrossRef CAS.
- N. Tien-Binh, D. Rodrigue and S. Kaliaguine, J. Membr. Sci., 2018, 548, 429–438 CrossRef CAS.
- Z. Gu, S. Yu, J. Zhu, P. Li, X. Gao and R. Zhang, Desalination, 2020, 493, 114661 CrossRef CAS.
- A. Katare and B. Mandal, ACS Appl. Nano Mater., 2023, 6, 4821–4833 CrossRef CAS.
- A. Valverde, E. Alkain, N. A. Rio-López, L. Lezama, A. Fidalgo-Marijuan, J. M. Laza, S. Wuttke, J. M. Porro, I. Oyarzabal, M. Jiménez-Ruiz, V. García Sakai, P. L. Arias, I. Agirrezabal-Telleria and R. Fernández de Luis, J. Mater. Chem. A, 2024, 12, 4555–4571 RSC.
- M. D. Guiver, M. Yahia, M. M. Dal-Cin, G. P. Robertson, S. Saeedi Garakani, N. Du and N. Tavajohi, Macromolecules, 2020, 53, 8951–8959 CrossRef CAS PubMed.
- M. Yahia, L. A. Lozano, J. M. Zamaro, C. Téllez and J. Coronas, Sep. Purif. Technol., 2024, 330, 125558 CrossRef CAS.
- M. Pérez-Miana, J. M. Luque-Alled, M. Yahia, Á. Mayoral and J. Coronas, J. Mater. Chem. A, 2024, 12, 10316–10328 RSC.
- M. Yahia, D. Refaat, J. Coronas and C. Tellez, Sep. Purif. Technol., 2025, 356, 129825 CrossRef CAS.
- D. Refaat, M. Yahia and J. Coronas, Process Saf. Environ. Prot., 2024, 192, 401–411 CrossRef CAS.
- L. Martínez-Izquierdo, A. Perea-Cachero, M. Malankowska, C. Téllez and J. Coronas, J. Environ. Chem. Eng., 2022, 10, 108324 CrossRef.
- Z. Li, W. Zhang, M. Tao, L. Shen, R. Li, M. Zhang, Y. Jiao, H. Hong, Y. Xu and H. Lin, Chem. Eng. J., 2022, 435, 134804 CrossRef CAS.
- L. A. Lozano, L. A. S. Hoyos, B. M. C. Faroldi and J. M. Zamaro, Mater. Today Commun., 2022, 31, 103540 CrossRef CAS.
- G. Morales, M. Paniagua, D. de la Flor, M. Sanz, P. Leo, C. López-Aguado, H. Hernando, S. A. Orr, K. Wilson, A. F. Lee and J. A. Melero, Fuel, 2023, 339, 127465 CrossRef CAS.
- L. Hao, K.-S. Liao and T.-S. Chung, J. Mater. Chem. A, 2015, 3, 17273–17281 RSC.
- C. Ardila-Suárez, A. M. Díaz-Lasprilla, L. A. Díaz-Vaca, P. B. Balbuena, V. G. Baldovino-Medrano and G. E. Ramírez-Caballero, CrystEngComm, 2019, 21, 3014–3030 RSC.
- H. Reinsch, S. Waitschat, S. M. Chavan, K. P. Lillerud and N. Stock, Eur. J. Inorg. Chem., 2016, 2016, 4490–4498 CrossRef CAS.
- L. Valenzano, B. Civalleri, S. Chavan, S. Bordiga, M. H. Nilsen, S. Jakobsen, K. P. Lillerud and C. Lamberti, Chem. Mater., 2011, 23, 1700–1718 CrossRef CAS.
- M. Lammert, C. Glißmann and N. Stock, Dalton Trans., 2017, 46, 2425–2429 RSC.
- L. A. Lozano, C. M. Iglesias, B. M. C. Faroldi, M. A. Ulla and J. M. Zamaro, J. Mater. Sci., 2018, 53, 1862–1873 CrossRef CAS.
- P. M. Budd, B. S. Ghanem, S. Makhseed, N. B. McKeown, K. J. Msayib and C. E. Tattershall, Chem. Commun., 2004, 230–231, 10.1039/B311764B.
- N. B. McKeown, P. M. Budd, K. J. Msayib, B. S. Ghanem, H. J. Kingston, C. E. Tattershall, S. Makhseed, K. J. Reynolds and D. Fritsch, Chem.–Eur. J., 2005, 11, 2610–2620 CrossRef CAS PubMed.
- N. B. McKeown, Polymer, 2020, 202, 122736 CrossRef CAS.
- M. Tian, S. Rochat, H. Fawcett, A. D. Burrows, C. R. Bowen and T. J. Mays, Adsorption, 2020, 26, 1083–1091 CrossRef CAS.
- A. Loosen, F. de Azambuja and T. N. Parac-Vogt, Mater. Adv., 2022, 3, 2475–2487 RSC.
- O. I.-F. Chen, C.-H. Liu, K. Wang, E. Borrego-Marin, H. Li, A. H. Alawadhi, J. A. R. Navarro and O. M. Yaghi, J. Am. Chem. Soc., 2024, 146, 2835–2844 CrossRef CAS PubMed.
- M. Benzaqui, R. Semino, N. Menguy, F. Carn, T. Kundu, J.-M. Guigner, N. B. McKeown, K. J. Msayib, M. Carta, R. Malpass-Evans, C. Le Guillouzer, G. Clet, N. A. Ramsahye, C. Serre, G. Maurin and N. Steunou, ACS Appl. Mater. Interfaces, 2016, 8, 27311–27321 CrossRef CAS PubMed.
- D. Lombardo, P. Calandra and M. A. Kiselev, Molecules, 2020, 25, 5624 CrossRef CAS PubMed.
- C.-S. Tsao, M.-S. Yu, T.-Y. Chung, H.-C. Wu, C.-Y. Wang, K.-S. Chang and H.-L. Chen, J. Am. Chem. Soc., 2007, 129, 15997–16004 CrossRef CAS PubMed.
- T. Alebrahim, L. Huang, H. K. Welgama, N. Esmaeili, E. Deng, S. Cheng, D. Acharya, C. M. Doherty, A. J. Hill, C. Rumsey, M. Trebbin, T. R. Cook and H. Lin, ACS Appl. Mater. Interfaces, 2024, 16, 11116–11124 CrossRef CAS PubMed.
- R. Swaidan, B. Ghanem, M. Al-Saeedi, E. Litwiller and I. Pinnau, Macromolecules, 2014, 47, 7453–7462 CrossRef CAS.
- R. Ramezani, S. Mazinani and R. Di, Felice, 2022, 38, 273–299 CAS.
- H. Lyu, O. I.-F. Chen, N. Hanikel, M. I. Hossain, R. W. Flaig, X. Pei, A. Amin, M. D. Doherty, R. K. Impastato, T. G. Glover, D. R. Moore and O. M. Yaghi, J. Am. Chem. Soc., 2022, 144, 2387–2396 CrossRef CAS PubMed.
- S. He, B. Zhu, S. Li, Y. Zhang, X. Jiang, C. Hon Lau and L. Shao, Sep. Purif. Technol., 2022, 284, 120277 CrossRef CAS.
- A. E. Gemeda, M. G. De Angelis, N. Du, N. Li, M. D. Guiver and G. C. Sarti, J. Membr. Sci., 2017, 524, 746–757 CrossRef CAS.
- S. Thomas, I. Pinnau, N. Du and M. D. Guiver, J. Membr. Sci., 2009, 333, 125–131 CrossRef CAS.
- M. Yamato, M. Akatsuka and H. Kawakami, Polymer, 2024, 308, 127413 CrossRef CAS.
- M. Y. M. Abdelrahim, C. F. Martins, L. A. Neves, C. Capasso, C. T. Supuran, I. M. Coelhoso, J. G. Crespo and M. Barboiu, J. Membr. Sci., 2017, 528, 225–230 CrossRef CAS.
- K. I. Hadjiivanov, D. A. Panayotov, M. Y. Mihaylov, E. Z. Ivanova, K. K. Chakarova, S. M. Andonova and N. L. Drenchev, Chem. Rev., 2021, 121, 1286–1424 CrossRef CAS PubMed.
- I. Romero-Muñiz, C. Romero-Muñiz, I. del Castillo-Velilla, C. Marini, S. Calero, F. Zamora and A. E. Platero-Prats, ACS Appl. Mater. Interfaces, 2022, 14, 27040–27047 CrossRef PubMed.
- J. Xu, J. Liu, Z. Li, X. Wang, Y. Xu, S. Chen and Z. Wang, New J. Chem., 2019, 43, 4092–4099 RSC.
- Y. Ma, W. Lu, X. Han, Y. Chen, I. da Silva, D. Lee, A. M. Sheveleva, Z. Wang, J. Li, W. Li, M. Fan, S. Xu, F. Tuna, E. J. L. McInnes, Y. Cheng, S. Rudić, P. Manuel, M. D. Frogley, A. J. Ramirez-Cuesta, M. Schröder and S. Yang, J. Am. Chem. Soc., 2022, 144, 8624–8632 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2025 |

