 Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
High-temperature composite phase change material with “concrete-like” strength even beyond the eutectic temperature
Yuto
Shimizu
 a,
Takahiro
Kawaguchi
b,
Joshua Chidiebere
Mba
a,
Takahiro
Kawaguchi
b,
Joshua Chidiebere
Mba
 b,
Tomokazu
Nakamura
b,
Keita
Tanahashi
b,
Melbert
Jeem
b,
Tomokazu
Nakamura
b,
Keita
Tanahashi
b,
Melbert
Jeem
 b and
Takahiro
Nomura
b and
Takahiro
Nomura
 *b
*b
aGraduate School of Engineering, Hokkaido University, Kita 13 Nishi 8, Kita-ku, Sapporo, 060-8628, Japan
bFaculty of Engineering, Hokkaido University, Kita 13 Nishi 8, Kita-ku, Sapporo, 060-8628, Japan. E-mail: nms-tropy@eng.hokudai.ac.jp; Tel: +81 11 706 6849; Fax: +81 11 706 6842
First published on 14th August 2025
Abstract
The need for medium-to-long-term thermal energy storage has increased in tandem with the widespread adoption of variable renewable energy sources. Latent heat storage using metallic phase change materials (PCMs) presents a promising solution by combining high thermal conductivity with high heat storage density. Recent advances in microencapsulated PCMs (MEPCMs) have addressed corrosion and leakage issues commonly associated with metallic PCMs. This study develops an MEPCM composite using an Al–Cu–Si-based MEPCM as the primary component. This system exhibits a suitable structure, performance, and strength for achieving GW h-scale thermal energy storage systems. The MEPCM composite is fabricated by microencapsulating Al–Cu–Si eutectic alloy powder, which has a eutectic temperature of 520 °C, with an Al oxide shell formed via chemical conversion and heat-oxidation treatments, followed by mixing with an alumina sintering aid, forming, and sintering. The composite exhibits a heat storage density of approximately 1.0 GJ m−3 under a temperature difference (ΔT) of 200 °C, which is 2–3 times higher than that of conventional sensible heat storage materials. It retains both its heat storage performance and structural integrity after 1000 thermal cycles. The thermal conductivity ranges from 4.1 to 6.6 W m−1 K−1 between 300 and 600 °C. This study is the first to evaluate the compressive strength of composite PCMs in the liquid state, where the composite retains a compressive strength of 32 MPa at 600 °C, which is comparable to that of ordinary concrete. In the solid state, the composite exhibits 83 MPa at room temperature and 49 MPa at 500 °C. Therefore, MEPCM composites are expected to possess substantial compressive strength for applications in medium-to-high-temperature heat storage and thermal management systems, even beyond the eutectic temperature of the PCM.
1. Introduction
With the large-scale deployment of variable renewable energy aiming to achieve a decarbonized society, advancements in thermal energy storage (TES) and thermal management technologies have become increasingly crucial. The variability of renewable energy has been extensively explored, and strategies using power-to-X systems to convert and store renewable energy, including chemical or thermal energy, are essential.1 Recently, power-to-heat-to-power energy storage systems, also known as Carnot batteries, have been proposed. Carnot batteries excel over other energy storage technologies in terms of economic viability and geographical constraints.2–4 In addition, power-to-X-related processes, including the Sabatier reaction,5–11 Fischer–Tropsch synthesis,12–14 solid oxide fuel cells, and reversible solid oxide cells,15–17 have demonstrated that thermal management technologies can enhance device and plant performances, extend their service life, and reduce waste heat. Accordingly, the advancement of heat storage and management technologies is indispensable not only for strengthening sector coupling among the electricity, industry, and transportation sectors,18–20 but also for improving the efficiency of each process.In TES systems, three main heat storage technologies are widely recognized: sensible heat storage (SHS), latent heat storage (LHS), and thermochemical heat storage (THS).21–23 SHS is based on the principle of specific heat capacity, storing heat by raising the temperature of solid or liquid materials. Owing to its ease of handling, SHS is the most widely adopted technology; however, its heat storage density is lower than those of LHS and THS. By contrast, LHS uses the latent heat of solid–liquid phase transitions in phase change materials (PCMs) and offers several advantages, including a high heat-storage density, the ability to supply heat at a constant phase change temperature, operation using heat alone, and excellent reusability.21,24–26 THS systems utilize heat released by reversible thermochemical reactions. Although THS demonstrates excellent heat storage performance, it faces more challenges than SHS and LHS, including a shorter material lifetime, lower heat transfer efficiency, and more complex system design due to the involvement of chemical reactions.21,23,27 Among these technologies, LHS may be used to achieve a simple TES system due to its inherent characteristics and is also suitable for thermal regulation in processes with temperature fluctuations, such as industrial waste heat and heat from chemical reactions.28,29 Thermal management using PCMs has been studied and commercialized for low-temperature applications, such as electronic devices30–33 and Li-ion batteries.34–40 In the high-temperature range, its application to the thermal regulation of chemical reactions has been demonstrated at the laboratory scale.9–11,41,42
PCMs face several practical challenges including leakage during melting, chemical reactivity, and phase separation.43 The microencapsulation of PCMs is an effective solution to these issues.28,44–46 The development of microencapsulated PCMs (MEPCMs) has been explored for both organic systems,44,47–50 which operate at low temperatures, and inorganic salt51–55 and metal alloy systems,28,45,46 which operate at medium-to-high temperatures. While organic MEPCMs have attained practical applications,48–50 the technology for microencapsulated molten salt and alloy-based PCMs remains in its early stages. However, in alloy-based MEPCMs, the development of an Al–Si alloy core with an Al2O3 shell56 has initiated a phase of exponential growth in recent years, resulting in various metallic PCMs with different melting points46 (such as Ga,57–59 Sn,60–65 Sn-alloy,66,67 Zn–Al,68–72 Al–Cu–Si,73 Al–Cu,74 Al–Si,56,75–82 Al–Si–Fe,83 Al–Ni,84 Al,85–88 and Cu–Si–Al89 systems) being encapsulated in ceramic shells. The development of these alloy-based MEPCMs provides a foundation for fabricating LHS materials and thermal management systems applicable in medium-to-high-temperature ranges.
Metal and alloy-based MEPCMs, which function both as LHS materials and ceramic particles, exhibit excellent handling and formability, making them highly promising for use in heat storage and thermal management devices. Composite PCMs have been developed using Al–Si-based MEPCMs,90–93 which are among the most advanced alloy-based MEPCMs. Furthermore, heat storage and thermal regulation devices have been explored at the laboratory scale,9–11,41,94,95 with the development of kW h-scale packed-bed LHS systems.96 Recent reports on the development of Al–Si-based MEPCM composites have demonstrated thermal cycling durability of up to 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles.90 In addition, progress has been made with composites utilizing MEPCMs based on Zn–Al,70,71 Sn,97,98 and Sn alloys,99–101 which continue to evolve as part of broader MEPCM development. Research on thermal management technologies using MEPCMs in temperature-varying processes is advancing. Koide et al. proposed using MEPCMs as catalyst supports in CO2 methanation (the Sabatier reaction), a hydrogen utilization technology, to mitigate catalytic thermal runaway caused by exothermic reactions.9 Similarly, Cholila et al. and Takizawa et al. reported the effectiveness of MEPCMs in regulating reaction heat in ammonia decomposition42 and in chemical-loop reverse water–gas shift reaction processes,41 respectively. Similar thermal management systems using MEPCMs could be employed in the Sabatier reaction, ammonia thermal decomposition, and reverse water–gas shift reaction, alongside use in various catalytic chemical reactions and temperature-varying processes (such as chemical batteries,34–40,102 dry reforming,103,104 and solid oxide fuel cells15–17).
000 cycles.90 In addition, progress has been made with composites utilizing MEPCMs based on Zn–Al,70,71 Sn,97,98 and Sn alloys,99–101 which continue to evolve as part of broader MEPCM development. Research on thermal management technologies using MEPCMs in temperature-varying processes is advancing. Koide et al. proposed using MEPCMs as catalyst supports in CO2 methanation (the Sabatier reaction), a hydrogen utilization technology, to mitigate catalytic thermal runaway caused by exothermic reactions.9 Similarly, Cholila et al. and Takizawa et al. reported the effectiveness of MEPCMs in regulating reaction heat in ammonia decomposition42 and in chemical-loop reverse water–gas shift reaction processes,41 respectively. Similar thermal management systems using MEPCMs could be employed in the Sabatier reaction, ammonia thermal decomposition, and reverse water–gas shift reaction, alongside use in various catalytic chemical reactions and temperature-varying processes (such as chemical batteries,34–40,102 dry reforming,103,104 and solid oxide fuel cells15–17).
In LHS, selecting or developing PCMs with appropriate operating temperatures for specific applications is essential. Furthermore, beyond Al–Si-based and other established systems, developing composite materials comprising alloy-based MEPCMs with various melting points is anticipated to advance future TES and thermal management technologies. Accordingly, this study focuses on eutectic Al–Cu–Si alloy-based MEPCMs with a melting point of 520 °C,73,105 which could be employed in medium-to-high-temperature thermal utilization technologies. Heat storage technologies operating in this temperature range have been practically implemented in concentrating solar power (CSP) systems using steam Rankine cycles.106–111 Moreover, both the CSP and TES components of Carnot batteries, which are expected to be developed in the future, could benefit from this technology.112,113
To the best of our knowledge, this study is the first to evaluate the mechanical strength of composite PCMs at high temperatures exceeding the melting point of PCMs. While several studies have assessed the compressive strength of composite PCMs based on molten salts,114–120 Sn-based alloys,100 and Al121 systems, all these evaluations have been conducted at room temperature, and none have been performed under high-temperature conditions where the PCM exists in a liquid state. However, in packed-bed TES, thermal stresses induced by the thermal expansion of storage materials pose a risk of structural failure in either the storage medium itself or its container.122–124 Such cyclic thermal stress resulting from repeated heating and cooling is a well-known concern referred to as thermal ratcheting. Concerning this phenomenon, numerical analyses of thermo-mechanical effects in high-temperature heat storage systems have been conducted based on experimental assessments of material mechanical properties.125,126 Therefore, to realize the large-scale implementation of TES and thermal management systems using MEPCM composites, experimental evaluation of mechanical strength at high temperatures is essential.
This study evaluates the compressive strength of composite PCMs when the PCM component is in the molten state and discusses the corresponding failure mechanisms. This investigation provides insight into the compressive fracture properties of alloy-based MEPCM composites with the MEPCM as the primary component. In particular, this study aims to develop composite materials with the Al–Cu–Si-based MEPCM as the main component and to evaluate their thermophysical properties, thermal cycling durability, heat storage performance, and thermomechanical properties, along with their compressive strength and compressive fracture mechanisms under high-temperature conditions.
2. Materials and methods
2.1 MEPCM composite preparation
Fig. 1 illustrates the fabrication procedure of the MEPCM composite, which can be broadly categorized into two main steps: (1) MEPCM preparation using Al–Cu–Si alloy particles and (2) MEPCM composite preparation. In step 1, Al–26.5% Cu–5.4% Si (mass%) powder (particle size: d < 45 μm) (Hikari Material Industry Co., Ltd., Japan) was used as the raw material. The Al–Cu–Si particles were encapsulated via a two-step process comprising a chemical conversion treatment followed by a heat-oxidation treatment (steps 1-1 and 1-2 in Fig. 1). First, 20 g of Al–Cu–Si powder was treated in boiling distilled water containing 16.7 g L−1 of Al(OH)3 for 3 h, resulting in the formation of alumina precursors (AlOOH and Al(OH)3) on the particle surface. The treated powder was filtered and dried overnight at 100 °C. In step 1-2, the chemically treated powder underwent heat-oxidation by being heated in ambient air from room temperature to 1100 °C at a rate of 10 °C min−1, followed by isothermal holding for 3 h in a muffle furnace. Fig. 2 presents photographs, scanning electron microscopy (SEM) images, energy dispersive X-ray spectroscopy (EDS) elemental mapping images, and the particle size distribution of the prepared MEPCM samples. The particle size distribution was evaluated using the Segment Anything Model for nanoparticles, as reported by Larsen et al.127 Consistent with a previous study,73 the MEPCMs were prepared as powders, each comprising an Al alloy core encapsulated by an Al oxide shell (Fig. 2b). In addition, the average particle size of the MEPCM powder was measured as 25.2 μm. In step 2, the MEPCM prepared in step 1 was mixed with a sintering aid powder, shaped into green bodies, and subsequently sintered to form the MEPCM composite (steps 2-1 and 2-2 in Fig. 1). Furthermore, α-Al2O3 (average particle size: 0.15 μm; purity: 99.99%; Taimei Chemicals Co., Ltd., Japan) was used as a sintering aid for the MEPCM composite. The Al–Cu–Si alloy-based MEPCM and the α-Al2O3 powder were wet-mixed at a volume ratio of 8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2. The mixed powder was then die-pressed into green bodies with a diameter of 10 mm at 20 MPa for 1 min. The green bodies were sintered by heating in ambient air from room temperature to 1100 °C at a rate of 5 °C min−1, followed by isothermal holding for 1 h in a muffle furnace. After isothermal holding at 1100 °C, the muffle furnace was programmed to cool at a rate of −5 °C min−1. The composite samples were fabricated with weight adjustments to ensure that the height of the green bodies did not exceed approximately 10 mm.
2. The mixed powder was then die-pressed into green bodies with a diameter of 10 mm at 20 MPa for 1 min. The green bodies were sintered by heating in ambient air from room temperature to 1100 °C at a rate of 5 °C min−1, followed by isothermal holding for 1 h in a muffle furnace. After isothermal holding at 1100 °C, the muffle furnace was programmed to cool at a rate of −5 °C min−1. The composite samples were fabricated with weight adjustments to ensure that the height of the green bodies did not exceed approximately 10 mm.
2.2 Analyses
The cross-sectional structure of the MEPCM composite samples was observed and analyzed using SEM and EDS (SEM-EDS; JSM-7001FA, JEOL Ltd., Japan). Fig. S1 provides a schematic of the observed composite region. The sample cross section was prepared by cutting along the central axis of the cylindrical composite, followed by polishing with emery paper. The phase structure of the composite samples was analyzed using powder X-ray diffraction (XRD; MiniFlex600, Rigaku, Japan) with a Cu Kα radiation source. The powder samples for XRD measurements were obtained by grinding the prepared composite samples with a mortar and pestle. The apparent density (ρ0) of the composite samples at 25 °C was measured using a gas pycnometer (Ultrapycnometer 1000, Quantachrome Instruments, USA). The prepared composite samples were used as-is during the gas pycnometer measurements. The coefficient of linear thermal expansion (αCLTE) of the samples was measured using a thermomechanical analyzer (TMA; TMA7300, Hitachi High-Tech Science Co., Japan). Three composite samples with diameters of 10 mm and heights of approximately 10 mm were prepared and measured using the TMA. The melting and solidification characteristics of the samples were evaluated using differential scanning calorimetry (DSC; DSC3+, Mettler Toledo, Switzerland). For the DSC measurements, 15 mg of the composite sample, pulverized using a mortar and pestle, was placed in a 30 μL alumina sample pan. The samples were heated to 650 °C under an Ar gas flow of 50 mL min−1 and then cooled, with a heating/cooling rate of ±5 °C min−1. The thermal diffusivity (αTD) and specific heat (Cp) of the samples were measured using the laser flash analyzer (TC-7000, ULVAC, Japan) over the temperature range of 300–600 °C. For laser flash analysis, samples of the fabricated MEPCM composite were used after thinning to a 1 mm thickness by polishing. The thermal conductivity (k) was calculated using the density (ρ) at a given temperature—determined from the measured ρ0 and αCLTE—together with αTD and Cp. The values of ρ and k were calculated as| ρ = ρ0/(1 + 3αCLTE(T − T0)), | (1) |
| k = αTD × Cp × ρ. | (2) |
2.3 Thermal cyclic test
To evaluate the cyclic durability of heat storage and release, the samples were subjected to 500 and 1000 cycles of heating and cooling within the temperature range corresponding to the melting and solidification of the Al–Cu–Si alloy. For the thermal cycling test, MEPCM composite samples with a diameter and height of 10 and ∼4 mm were used. Fig. 3a shows a photograph of the apparatus used for the thermal cyclic test, while Fig. 3b presents the gas temperature near the sample as measured by thermocouples. The samples were placed in a quartz tube with flowing air and were heated and cooled using two mobile electric furnaces. The temperatures of the two furnaces were set at 400 and 650 °C, respectively. First, the sample was placed at the center of the 400 °C furnace and held until the air temperature in the vicinity of the sample reached a steady state. Then, the 650 °C furnace was moved to the sample position and held there for 6 min, after which the 400 °C furnace was returned to the sample position. Thermal cycling was performed for 500 and 1000 cycles, with heating at 650 °C for 6 min and cooling at 400 °C for 9 min. Based on the gas temperature near the sample during thermal cycling (Fig. 3b), it was confirmed that the sample was heated and cooled within a temperature range of approximately 420–620 °C. The cross-sectional structure and the melting and solidification behaviors of the samples after thermal cycling were analyzed using SEM-EDS and DSC, along with those of the as-prepared MEPCM composite samples. | ||
| Fig. 3 (a) Photograph of the apparatus used for the thermal cyclic test, and (b) changes in the gas temperature near the sample. | ||
2.4 Compression test
The composite samples were subjected to compression tests at constant temperatures of 24 (i.e., room temperature (RT)), 500, 550, and 600 °C using a universal testing machine (SHIMADZU Co. Ltd., AUTOGRAPH AG-IS 100 kN AG-X/R). The samples attached to thermocouples for temperature control were placed in an electric furnace and compression tested at a compression rate of 0.5 mm min−1 in ambient air. Compression tests were performed until the samples were fractured. The setup for the compression tests at an elevated temperature is shown in Fig. 4. Composite samples of 10 mm in diameter and height were used as the compression test specimens. Details of diameter, height, and parallelism for all specimens used in the compression test are listed in Table S1. The cross-sectional sample structure after the compression test was analyzed using SEM-EDS. The as-prepared MEPCM composite samples were also tested.3. Results
3.1 Structure
Fig. 5 presents multiple characterizations of the Al–Cu–Si-based MEPCM composite: a photograph of the sample (Fig. 5a), SEM and EDS mapping of the cross-sectional structure (Fig. 5b), the XRD pattern (Fig. 5c), and the calculated theoretical density along with the measured apparent and bulk densities (Fig. 5d). As observed in Fig. 5a, no alloy leakage or damage due to cracking occurred even after sintering at 1100 °C. In the sample cross section, microcapsules of Al–Cu–Si alloy covered with Al oxide were sintered to each other while maintaining their respective capsule structures. From the XRD pattern, alloy components Al, Al2Cu, Si, and Al oxides α-Al2O3 and CuAl2O4 were detected in the composite. Referring to the cross-sectional observation in Fig. 5b, the alloy component comprised the MEPCM core, α-Al2O3 comprised the MEPCM shell and sintering aid, and CuAl2O4 was formed as part of the MEPCM shell. As shown in Fig. 5d, the theoretical density, apparent density, and bulk density of the composite were 3.52, 3.45, and 2.48 g cm−3, respectively. The theoretical density was calculated from the densities of the MEPCM (3.41 g cm−3) and the sintering aid, α-Al2O3 (3.96 g cm−3). The mixer volume ratio was 8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2. The apparent density was measured using the gas pycnometer, while the bulk density was calculated using the dimensions and weights of the composite samples. These density measurements revealed that the composite sample contained 28.1% and 2.0% open and closed porosities, respectively.
2. The apparent density was measured using the gas pycnometer, while the bulk density was calculated using the dimensions and weights of the composite samples. These density measurements revealed that the composite sample contained 28.1% and 2.0% open and closed porosities, respectively.
3.2 Phase change properties and thermal cyclic durability
Fig. 6a and b show the DSC curves of the composite during heating and cooling, respectively. An expanded view of the curves in the temperature range of 530–590 °C for the first cycle and after 500 and 1000 thermal cycles is shown in Fig. 6c. The melting onset temperature (Tm,onset) of the as-prepared composite (first cycle) was observed at 519 °C, followed by the main melting peak (Pm1) at 521 °C. Most of the latent heat of melting was attributed to Pm1, although a minor peak (Pm2) was observed at 573 °C. The total latent heat capacity of melting (ΔHm) was 190 J g−1. After 500 and 1000 thermal cycles, the values of Tm,onset, Pm1, and Pm2 remained unchanged compared to the first cycle. In addition, ΔHm showed negligible variation before and after the thermal cyclic tests. According to the DSC cooling curves in Fig. 6b and c, the solidification onset temperature (Ts,onset) of the composite was determined to be 553 °C. However, Ts,onset did not correspond to the main solidification stage, as the primary solidification peaks, Ps1 and Ps2, were observed at 496 and 489 °C, respectively, at lower temperatures. The total latent heat capacity of solidification (ΔHs) was 189 J g−1, which was similar to the latent heat capacity of melting. Similar to the melting behavior, there was almost no change in the Ts,onset, Ps1, Ps2, and ΔHs values before and after the thermal cyclic tests. A discussion of the heat storage properties and thermal cyclic durability of this system based on the results of this cycle test is provided in Section 4.1. | ||
| Fig. 6 DSC curves of the MEPCM composite during (a) heating and (b) cooling. (c) Expanded view in the temperature range of 530–590 °C for the first cycle and after 500 and 1000 thermal cycles. | ||
Fig. 7a-1–a-3 present SEM images and Fig. 7b-1–b-3 present EDS elemental mapping of the sample cross sections after being subjected to 1000 thermal cycles. Similar to the MEPCM composite sample before the thermal cyclic tests (Fig. 5b), the sample cross section exhibited a sintered structure comprising MEPCM with an Al–Cu–Si alloy core and an Al oxide shell. Thus, the composite sample retained its original microcapsule structure even after 1000 thermal cycles. Notably, Cu was not observed in EDS mapping (Fig. 7b-1–b-3) due to limitations in the number of overlapping colors. However, individual elemental mappings including Cu were observed, as shown in Fig. S2. Based on the analytical results obtained using the DSC and SEM-EDS, the developed composite, using Al–Cu–Si-based MEPCM as the base material, possessed excellent thermal cycling durability.
 | ||
| Fig. 7 (a-1–a-3) SEM images and (b-1–b-3) EDS elemental mapping of the sample cross section after 1000 thermal cycles; magnifications: ×200, ×500, and ×2000. | ||
3.3 Thermophysical properties
Fig. 8 presents the TMA curves of the Al–Cu–Si MEPCM composite samples. The samples exhibited monotonic thermal expansion with increasing temperature. Table 1 presents the average αCLTE values for the MEPCM composite sample at 50–500 °C in intervals of 50 °C. αCLTE remained almost constant at 10.6–12.3 × 10−6 °C−1 in the temperature range of 50–350 °C. Conversely, in the temperature range of 350–500 °C, αCLTE decreased from 11.5 to 6.1 × 10−6 °C−1. Table S2 presents the values of αCLTE in the temperature range of 50–500 °C for each of the three TMA measurements shown in Fig. 8. A discussion of the thermal expansion characteristics of this system is provided in Section 4.2.| Temperature [°C] | Liner thermal expansion coefficient, αCLTE [10−6 °C−1] |
|---|---|
| 50–100 | 10.6 |
| 100–150 | 11.6 |
| 150–200 | 12.1 |
| 200–250 | 12.3 |
| 250–300 | 12.2 |
| 300–350 | 11.5 |
| 350–400 | 9.4 |
| 400–450 | 7.1 |
| 450–500 | 6.1 |
Fig. 9a and b present the thermal diffusivity and specific heat and thermal conductivity of the Al–Cu–Si MEPCM composite sample, respectively. In addition, Table S3 presents the values of the thermophysical components plotted in Fig. 9. The thermal diffusivity of the composite samples gradually decreased from 0.0277 to 0.0140 cm2 s−1 between 300 and 500 °C, where the MEPCM core alloy remained in its solid phase. Conversely, the specific heat increased from 0.96 to 1.18 J g−1 K−1 over this temperature range. Upon exceeding the melting point of the MEPCM core alloy at 520 °C, the thermal diffusivity, which decreased up to 500 °C, improved and reached 0.178 at both 550 and 600 °C. The increase in specific heat observed below the melting point stabilized or slightly decreased when the temperature exceeded the melting point, reaching 1.22 and 1.11 at 550 and 600 °C, respectively. Similarly, the experimental thermal conductivity (kex; Fig. 9b) exhibited a change in temperature dependence beyond the melting point. The value of kex gradually decreased from 6.6 to 4.1 W m−1 K−1 between 300 and 500 °C. However, when heated from 500 °C to above the melting point of the MEPCM core alloy at 550 °C, the value of kex increased from 4.1 to 5.3 W m−1 K−1. The experimental results regarding the thermal conductivity are discussed in Section 4.3 in comparison with the estimated effective thermal conductivity.
3.4 Compression fracture behavior
Fig. 10a and b present the stress-testing machine displacement curves for the composite samples during compression testing at RT and 500 and 600 °C, respectively. Due to the high-temperature conditions of these tests, displacement measurements on the samples included minor errors originating from the testing machine. As precise correction of these errors is extremely challenging, the horizontal axis (Fig. 10) represented the measured testing machine displacement. The RT compression test results (Fig. 10a) indicated that the three tested samples underwent elastic deformation up to a displacement of approximately 0.14 mm. Subsequently, the samples demonstrated compressive strengths of 86, 81, and 77 MPa, with an average value of 81.3 MPa. After attaining these compressive strengths, the stress rapidly decreased to approximately 33 MPa. Following this drop in stress, one of the samples (sample 3) fractured due to a significant crack at a testing machine displacement of 0.22 mm. The other two samples (samples 1 and 2) exhibited gradual decrease in stress within a testing machine displacement range of 0.22–0.48 mm and fractured when the displacement reached 0.48 mm. However, even in samples 1 and 2 tested at RT, stress decreased sharply immediately after reaching the compressive stress value. Cracking was assumed to have occurred at that point. In the stress–displacement curves obtained at 500 and 600 °C (Fig. 10b), three primary stages of compressive failure behavior were observed. In stage 1, the initial phase of compression deformation, the elastic deformation behavior was evident. The compressive strength of 49 MPa was attained after passing stage 1 at 500 °C, where the MEPCM core was in its solid state. The compressive strength of 31 MPa was attained at 600 °C, where the MEPCM core was in its liquid state. Thus, the compressive strength of the MEPCM composite samples decreased due to the melting of the MEPCM core alloy. In stage 2, after attaining the compressive strength, differing behaviors were observed at 500 and 600 °C. At 500 °C, stress gradually decreased with increasing displacement. At 600 °C, stress fluctuated with increasing displacement, demonstrating a gradual decrease overall. Subsequently, in stage 3, both samples at 500 and 600 °C exhibited a reduced rate of stress decrease relative to the increase in displacement, thereby eventually stabilizing. The final stress at the fracture and the maximum displacement were 28.8 MPa and 1.01 mm at 500 °C, and 21.1 MPa and 1.51 mm at 600 °C, respectively. The stress–displacement curves for the compression tests of three specimens at 500, 550, and 600 °C are shown in Fig. S3. At 550 °C, the alloy core of MEPCM was mostly in its liquid phase, resulting in almost identical test results as those at 600 °C. Table S4 presents the maximum load and compressive strength values for all compression tests that were performed. | ||
| Fig. 10 Stress-testing machine displacement curves for the composite samples during compression tests at (a) room temperature and (b) 500 and 600 °C. | ||
Fig. 11 presents the photographs of MEPCM composite samples compressed and fractured at 500 and 600 °C. The sample that fractured at 500 °C exhibited only cracks, whereas that fractured at 600 °C showed both cracks and alloy leakage.
 | ||
| Fig. 11 Photographs of the MEPCM composite samples compressed and fractured at (a) 500 and (b) 600 °C. | ||
Fig. 12 presents the SEM images and EDS elemental mapping of the cross sections of the composite samples after compression testing at RT, 500 °C, and 600 °C. The SEM images in Fig. 12a-1–c-1 show that cracks formed due to compression in all samples, propagating approximately 45° from the bottom edge of the composite samples. The crack orientation was consistent with the general phenomenon in compression testing, where maximum shear stress occurs at 45°. As shown in Fig. 12a-2–a-5, the cross section of the sample compressed at RT exhibited two structural features: (1) cracks and (2) Al alloy core/Al oxide shell MEPCM. Notably, MEPCM (the main component of the composite) retained its microcapsule structure in nearly all particles even after the compressive fracture. For the sample compressed at 500 °C, three distinct microstructural regions were observed: (1) cracks, (2) regions where MEPCM retained its structure, and (3) regions where MEPCM was crushed, resulting in a mottled mixture of Al alloy and Al oxide (Fig. 12b-2–b-5). The regions marked as (2) and (3) were separated by the crack region (1). In the sample compressed at 600 °C, four microstructural features were observed: (1) cracks, (2) regions where MEPCM retained its structure, (3) regions surrounding the cracks where mostly Al oxide remained, and (4) regions where MEPCM was crushed, forming a mixture of Al alloy and Al oxide (Fig. 12c-2–c-5). Hence, the cracks in region (1) formed within region (3) after compression at 600 °C.
 | ||
| Fig. 12 SEM images and EDS elemental mapping of the cross sections of composite samples after compression testing at (a) RT, (b) 500 °C, and (c) 600 °C. | ||
4. Discussion
4.1 Heat storage properties and thermal cyclic durability
The MEPCM composite fabricated in this study comprised an Al–26.5% Cu–5.4% Si alloy with a near-eutectic composition as PCMs.73,105 DSC measurements (Fig. 6) revealed a primary melting peak at the eutectic temperature of approximately 520 °C. In addition, a minor endothermic peak observed at 573 °C during melting was attributed to Al oxidation during the composite fabrication process, which altered the alloy composition from the eutectic composition. Fig. S4 presents the phase diagram of Al–Cu0.68403Si0.31597, illustrating that a decrease in Al concentration from the eutectic composition increased the liquidus temperature. The shift in alloy composition due to oxidation during Al-alloy-based MEPCM fabrication has been reported in previous studies.73,84As shown in Fig. 6 and 7, the MEPCM composite samples exhibited no significant changes in LHS capacity or structural integrity even after 1000 thermal cycles. When the MEPCM composite samples lacked sufficient durability, structural failure of the microcapsules or alloy oxidation resulted in the deterioration of heat storage performance or leakage of the alloy.90,93,97 Therefore, the Al–Cu–Si MEPCM composite fabricated in this study exhibited excellent thermal cycling durability.
4.2 Thermal expansion characteristics
The values of αCLTE for α-Al2O3 were 4.6 and 7.1 × 10−6 °C−1 at 20 and 500 °C,128 respectively, while those for pure Al were 21.8 and 29.5 × 10−6 °C−1 at 50 and 500 °C, respectively.129 The value for ADC12 (Al–Si–Cu alloy) was 20.1 × 10−6 °C−1 from 50 to 200 °C,130 while that for A6061 (Al–Mg–Si alloy) was 24.3 × 10−6 °C−1 from 50 to 500 °C.131 Therefore, the value of αCLTE for the Al–Cu–Si MEPCM composite was lower than that of conventional Al alloys and approximately equal to that of α-Al2O3. Al alloys were dispersed as particles in the internal structure of MEPCM composites, whereas alumina formed a continuous structure. Notably, the thermal expansion coefficient of Al alloys was reduced by incorporating particles with low thermal expansion coefficients, such as alumina and SiC.129,131,132 In the MEPCM composite, αCLTE decreased as the temperature increased above 350 °C, attributable to the increased solubility of Si in the Al solid solution, which caused the Si crystals to dissolve into the Al solid solution. Si dissolved in the Al solid solution exhibited a smaller volume than Si crystals.133 Therefore, as the temperature increased, Si dissolved in Al, leading to a decrease in αCLTE for the MEPCM composite. Similar phenomena were reported by Huber et al. using A359 (AlSi10Mg) and A356 (AlSi7Mg).1324.3 Effective thermal conductivity
As presented in Section 3.3, the thermal conductivity of the MEPCM composite samples ranged from 4.1 to 6.6 W m−1 K−1 within the temperature range of 300–600 °C. The average value of 4.8 W m−1 K−1 was recorded (Fig. 7b). However, the thermal conductivities of the individual components, including the Al–Cu–Si alloy and alumina, were approximately 55–130 W m−1 K−1 and 8–12 W m−1 K−1,105,128 respectively, within the same temperature range. Thus, the presence of significant thermal resistance within the composite samples was observed.In this section, the effective thermal conductivity of the Al–Cu–Si MEPCM composite is estimated using theoretical models for porous media. This analysis provides insights into the achievable thermal conductivity of the MEPCM composite for future research and development.
The estimated effective thermal conductivity of the MEPCM composite (keff-comp) can be calculated using the Russell model, as shown in eqn (3):134,135
 | (3) |
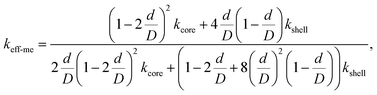 | (4) |
 | (5) |
 represents the effective thermal conductivity in a vacuum, ks is the thermal conductivity of a solid, ε is the porosity ratio, ε0 is the maximum porosity ratio, Dp is the particle diameter, and hp is the heat transfer coefficient between coarse particle contact surfaces. The value of ks corresponds to the effective thermal conductivity simulating the core–shell structure of the Al–Cu–Si alloy and alumina (Fig. S6). The porosity ε was set at 0.301, while the maximum porosity ε0 was taken as 0.476, based on the most open packing powder filling fraction.139 In addition, the term (hpDp/ks) represents a dimensionless number indicating the degree of consolidation, referred to as the consolidation parameter. Fig. 13b shows the temperature-dependent curves of
represents the effective thermal conductivity in a vacuum, ks is the thermal conductivity of a solid, ε is the porosity ratio, ε0 is the maximum porosity ratio, Dp is the particle diameter, and hp is the heat transfer coefficient between coarse particle contact surfaces. The value of ks corresponds to the effective thermal conductivity simulating the core–shell structure of the Al–Cu–Si alloy and alumina (Fig. S6). The porosity ε was set at 0.301, while the maximum porosity ε0 was taken as 0.476, based on the most open packing powder filling fraction.139 In addition, the term (hpDp/ks) represents a dimensionless number indicating the degree of consolidation, referred to as the consolidation parameter. Fig. 13b shows the temperature-dependent curves of  for cases where the ratio of kex (Fig. 9b) to ks (Fig. S6) is 0.282 and 0.351. When the average kex in the 300–500 °C range was used as kex, the ratio kex,ave. 300–500/ks was 0.282, while for the 550–600 °C range, it was 0.351 (=kex,ave. 550–600/ks). From this comparison, it is inferred that the thermal resistance within the composite is lower when the MEPCM core is in the liquid phase compared to the solid phase. Furthermore, the consolidation parameters below and above the melting point of the MEPCM core alloy were found to be 0.65 and 0.82, respectively. This result also suggests that interparticle heat transfer is enhanced when the MEPCM core is in the liquid phase; this improvement could be due to the melting expansion of the MEPCM core alloy, which eliminates the voids within the MEPCM shell and thereby promotes interparticle heat transfer. Consequently, although a decrease in thermal conductivity would typically be expected after melting (according to Fig. 13a), an increase in the experimental value was observed from just below the melting point to above it.
for cases where the ratio of kex (Fig. 9b) to ks (Fig. S6) is 0.282 and 0.351. When the average kex in the 300–500 °C range was used as kex, the ratio kex,ave. 300–500/ks was 0.282, while for the 550–600 °C range, it was 0.351 (=kex,ave. 550–600/ks). From this comparison, it is inferred that the thermal resistance within the composite is lower when the MEPCM core is in the liquid phase compared to the solid phase. Furthermore, the consolidation parameters below and above the melting point of the MEPCM core alloy were found to be 0.65 and 0.82, respectively. This result also suggests that interparticle heat transfer is enhanced when the MEPCM core is in the liquid phase; this improvement could be due to the melting expansion of the MEPCM core alloy, which eliminates the voids within the MEPCM shell and thereby promotes interparticle heat transfer. Consequently, although a decrease in thermal conductivity would typically be expected after melting (according to Fig. 13a), an increase in the experimental value was observed from just below the melting point to above it.
The thermal conductivity of the MEPCM composite sample fabricated in this study (with a porosity of 30.1%) was found to be significantly lower than the estimated keff-comp value shown in Fig. 13a. This suggests that the thermal resistance between solid particles is relatively high. To improve the thermal conductivity of the composite, fabrication of a denser material with enhanced sinterability is essential. This may be achieved by adjusting the particle size distribution of the mixed powder containing MEPCM and sintering aids140,141 or using sintering aids with high sinterability such as TiO2, La2O3, and ZrO2.142,143 Even though high-temperature sintering could be employed in MEPCM composites, the possible decrease in latent heat capacity due to the oxidation of the alloy should be carefully avoided. In addition, the Russell model used for the estimation occasionally overestimates the effective thermal conductivity.144,145 Therefore, this estimation represents one of the possible scenarios that can be achieved using MEPCM composites. To improve the usefulness of this estimation method, further research is required to develop MEPCM composites with different porosities using this method and to fit the estimated values with experimental thermal conductivities.
4.4 Compressive fracture: mechanism and key characteristics
As described in Section 3.4, the cross-sectional structures of the composite samples after compression failure at RT, 500 °C, and 600 °C were distinct. Fig. 14 illustrates the expected failure mechanisms at 500 °C and 600 °C, where stages 1, 2, and 3 correspond to those in Fig. 8. Stage 1 represents the elastic deformation region at both 500 °C and 600 °C, where no irreversible changes (such as MEPCM damage or crack formation) occur. In stage 2 at 500 °C, MEPCM degradation and the formation of microcracks between MEPCM particles occurs, gradually reducing the internal voids. By contrast, in stage 2 at 600 °C, the MEPCM shell is more easily damaged than at 500 °C because the core alloy is in the liquid phase, resulting in the leakage of the alloy from the MEPCM. The stress fluctuations observed in Fig. 8b could be due to the sequential rupture-like breakage of individual MEPCM particles. The difference in the stage 2 mechanism had a significant impact on compressive strength as the compressive strength of the sample at 600 °C was significantly lower than that at 500 °C. Stage 3 at 500 °C results from the breakdown and densification of the microcapsule structure in stage 2, leading to an almost complete elimination of voids. The elimination of voids increases resistance to compressive deformation, resulting in a smaller rate of decrease in stress with increasing displacement (Fig. 10b). The densified area eventually fractures when there is no more allowance for deformation. The crushed, dense MEPCM layer and the final crack formed in this process correspond to the crushed MEPCM (3) and crack areas (1) shown in Fig. 12b-2. Stage 3 at 600 °C forms a region where only Al oxide remains in a significant quantity. This is because the deformation that occurs during stage 2 caused the elimination of voids within the composite and the ejection of the PCM liquid, leaving only the Al oxide composed of the MEPCM shell and the sintering aids. The formation of a dense layer of oxide increases resistance to deformation and reduces the rate of decrease in stress with respect to an increase in displacement. Eventually, the tolerance limit of the dense Al oxide layer is exceeded, leading to fracture. The structures resulting from Al oxide densification and crack initiation correspond to the cracked (1) and crushed (3) Al oxide regions shown in Fig. 12c-2. The region of the crushed MEPCM (4) shown in Fig. 12c-2 is considered to be the microstructure corresponding to the stress fluctuations in Fig. 10b at stage 3. Stage 3 at 600 °C in Fig. 10b shows that the stress fluctuations remain, although they are less prominent than those in stage 2. This suggests that the MEPCM continues to rupture outside of the region of maximum shear stress. In addition, the maximum displacement is greater at 600 °C than at 500 °C because of the ejection of the internal PCM as a liquid at 600 °C (Fig. 11). Finally, the stress–displacement curve at RT (Fig. 10a) shows a sudden drop in stress after reaching the compressive strength, and it is inferred that the specimen cracks at that point. | ||
| Fig. 14 Schematic of the compressive fracture mechanism of the core alloy in the MEPCM composite during the solid and liquid phases (stages 1, 2, and 3 correspond to the notations in Fig. 10b). | ||
The compressive fracture mechanism supports the assertion that even when the PCM in the MEPCM composite was in its liquid phase, the liquid was rarely exposed to the surface until the compressive strength was reached. This property serves as a significant advantage because each PCM particle is covered by a ceramic shell. Although molten salt PCM composites have been considered medium-to-high-temperature LHS materials, these composite PCMs are porous materials impregnated with PCM.114–119 PCM was assumed to seep out onto the surface when compressed while the PCM inside was in a molten state. Such PCM seepage may be reversible to some extent as a microscopic phenomenon occurring at the scale of a single grain. However, in a packed-bed heat storage system, PCM is assumed to spread irreversibly between composite PCMs and onto the surface of the heat storage container. This phenomenon can lead to the agglomeration of composite PCM particles and degradation of the material due to the reaction between the PCM and the container. Therefore, even the composite PCM has limited applications due to concerns regarding its low resistance to thermal ratcheting. Conversely, composites with MEPCM as the main component do not experience PCM wetting and spreading even when in contact with other particles or containers because the PCM is not exposed to the surface if the applied load remains below the compressive strength. Moreover, in our previous studies on Al–Si MEPCM composites in lab-scale to kW h-class bench-scale packed-bed LHS devices, no agglomeration or breakage of heat storage particles was observed even after heat storage and release tests.94–96
Table 2 presents the compressive strengths of the MEPCM composite samples, conventional refractory materials, and concrete. The compressive strength values of refractory bricks were obtained from typical values for silica brick, alumina brick, high-alumina brick, and chamotte brick, which are used in practical applications.146 For concrete, the compressive strength values of ordinary and high-strength concrete were taken from the Japanese Industrial Standards for ready-mixed concrete (JIS 5308: 2024).147 The compressive strengths of the composite sample at RT (81 MPa) and 500 °C (49 MPa) were equal to or higher than those of the silica brick (44 MPa), alumina brick (65 MPa), high-alumina brick (83 MPa), chamotte brick (49 MPa), and high-strength concrete (45–60 MPa). Furthermore, even at 600 °C, when the core alloy of the composite sample was in its liquid state, its compressive strength (32 MPa) remained comparable to that of ordinary concrete (18–45 MPa). These findings indicated that although the compressive strength of the Al–Cu–Si MEPCM composite decreased at higher temperatures, it retained a strength that was comparable to that of ordinary concrete even when its internal structure was in a liquid state. Therefore, the composite sample was considered to exhibit sufficiently high compressive strength for use as a heat storage material. This discussion is significant for the future scaling-up of packed-bed LHS and thermal management systems, which have previously been explored MEPCM composites.41,42,94–96
| Material [ref. no.] | Compressive strength [MPa] |
|---|---|
| a Compressive strength of MEPCM composites is the average of three compression tests performed. b 95.0% SiO2–1.0% Al2O3–1.0% Fe2O3 (bricks for hot stove).146 c 41.6% SiO2–53.6% Al2O3–1.5% Fe2O3 (for heating furnace).146 d 20% SiO2–77% Al2O3–1% Fe2O3 (bricks for hot stove).146 e 58% SiO2–38% Al2O3–2% Fe2O3 (bricks for hot stove).146 | |
| Al–Cu–Si MEPCM composite (at RT)a | 81 |
| Al–Cu–Si MEPCM composite (at 500 °C)a | 49 |
| Al–Cu–Si MEPCM composite (at 600 °C)a | 32 |
| Silica brickb | 44 |
| Alumina brickc | 65 |
| High alumina brickd | 83 |
| Chamotte bricke | 49 |
| Ordinary concrete147 | 18–45 |
| High strength concrete147 | 45–60 |
4.5 Comparison with conventional sensible heat storage materials
Fig. 15 presents the volume-based heat storage densities at ΔT = 100 and 200 °C for conventional SHS materials such as reinforced concrete, silica brick, rock, high-alumina brick, molten nitrate, and the Al–Cu–Si MEPCM composite fabricated in this study. Table 3 presents the density, specific heat, and thermal conductivity of each of the heat storage materials shown in Fig. 15.136,148–150 When the latent heat component of the MEPCM composite was combined with a sensible heat component at ΔT = 100 °C or 200 °C, the resulting heat storage density reached 0.74 and 1.0 GJ m−3, respectively. Assuming ΔT = 100 or 200 °C, the MEPCM composite exhibited a heat storage density that was 3–4 or 2–3 times higher than that of other SHS materials, respectively. Furthermore, as shown in Table 3, the thermal conductivity of the MEPCM composite was higher than that of conventional SHS materials. Therefore, the MEPCM composite can facilitate the design of heat storage systems with a higher heat storage density, compactness, and more rapid heat exchange compared to conventional SHS materials. | ||
| Fig. 15 Heat storage density per unit volume at ΔT = 100 and 200 °C for conventional solid and liquid sensible heat storage materials and Al–Cu–Si MEPCM composite. | ||
| Material (at temperature) [ref. no.] | Density [kg m−3] | Specific heat [kJ kg−1 K−1] | Thermal conductivity [W m−1 K−1] |
|---|---|---|---|
| Reinforced concrete (ave. at 200–400 °C)148 | 2200 | 0.85 | 1.5 |
| Silica brick (ave. at 200–700 °C)148 | 1820 | 1.0 | 1.5 |
| Rock149 | 2240 | 1.0 | — |
| High alumina brick (1027 °C)136 | 2200 | 1.07 | 1.7 |
| NaNO3–40% KNO3 (molten salt) (300 °C)150 | 1730 | 1.54 | 0.6 |
| Al–Cu–Si MEPCM composite (ave. 300–600 °C) | 2480 | 1.07 | 5.5 |
It should be noted that the Al–Cu–Si MEPCM composite was compared with conventional SHS materials because these materials are generally used in similar temperature ranges for heat storage applications. Previously developed alloy-based LHS composites include the SnBi58 alloy and Al–Si alloy-based phase change composites, which exhibit phase change temperatures of 150 and 577 °C, respectively.90,100 Because the operating temperatures of these materials differ from that of the material investigated in this study, this comparison is provided only for reference. Fig. S7 shows the volume-based heat storage densities of these materials at ΔT = 100 °C. The SnBi58 alloy and Al–Si alloy-based MEPCM composites exhibited heat storage densities of 0.40 and 0.56 GJ m−3, respectively. As the Al–Cu–Si MEPCM composite developed in this study has a heat storage density of 0.74 GJ m−3 at ΔT = 100 °C, this verifies that this material exhibits a high heat storage density compared to alloy-based LHS materials.
5. Conclusion
In this study, Al–Cu–Si MEPCM composites were developed as medium-to-high-temperature LHS materials, and their structures, thermophysical properties, heat storage performance, thermal cycling durability, and compressive fracture characteristics were evaluated. This study is the first to investigate the compressive strength and fracture mechanism of LHS composites under high-temperature conditions where the PCM exists in a liquid state. The key findings of this study are summarized as follows:• The Al–Cu–Si MEPCM composite was fabricated as a sintered structure in which the MEPCM, comprising an Al–Cu–Si alloy core and an Al oxide shell, was co-sintered with alumina powder serving as a sintering aid. The resulting composite exhibited a bulk density of 2.48 g cm−3 and a porosity of 30.1%.
• The composite exhibited a specific heat of 0.96–1.22 J g−1 K−1 and a thermal conductivity of 4.1–6.6 W m−1 K−1 over a temperature range of 300–600 °C. Under heat storage conditions with a temperature difference (ΔT) of 200 °C, the composite achieved a heat storage density of approximately 1.0 GJ m−3, which was two-to-three times that of conventional SHS materials.
• The MEPCM composite retained its structural integrity after undergoing 1000 thermal cycles, with no observed changes in its phase change temperature or latent heat capacity.
• Even at high temperatures where the PCM existed in a liquid state, the MEPCM composite prevented the PCM from being exposed at the surface until its compressive strength limit was reached. This behavior represents a distinctive advantage over conventional composite PCMs, which typically comprise porous media impregnated with PCM and are more prone to leakage under similar conditions.
• At RT, the MEPCM composite exhibited a compressive strength of 81 MPa, which was comparable to or greater than that of silica bricks, alumina bricks, and high-strength concrete. Remarkably, even at 600 °C—when the internal Al alloy was in a molten state—the composite retained a compressive strength of 32 MPa, which was comparable to that of ordinary concrete.
Future research will focus on further densifying the MEPCM composite to enhance its thermal conductivity, heat storage density, and mechanical strength. Moreover, repeated thermal deformation tests within the elastic deformation range of MEPCM composites, which were not assessed in the present compression tests, are required to further investigate the durability of MEPCM composites against thermal ratcheting. Considering the applications of these materials, packed-bed heat storage and heat exchanger systems incorporating Al–Cu–Si MEPCM composites should be investigated, and thermal management technologies that use latent heat to maintain temperature-fluctuating processes at a constant phase change temperature should be developed.
Author contributions
Yuto Shimizu: investigation, data curation, formal analysis, visualization, funding acquisition, writing – original draft, writing – review & editing. Takahiro Kawaguchi: writing – review & editing, data curation. Joshua Chidiebere Mba: writing – review & editing, data curation. Tomokazu Nakamura: writing – review & editing, data curation. Keita Tanahashi: writing – review & editing, data curation. Melbert Jeem: writing – review & editing, data curation. Takahiro Nomura: conceptualization, funding acquisition, methodology, project administration, supervision, writing – review & editing.Conflicts of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.Data availability
The authors confirm that the data supporting the findings of this study are available within the article or its SI.Supplementary information, which includes supplementary figures and tables, is available. See DOI: https://doi.org/10.1039/d5ta03994k.
Acknowledgements
This paper was partially supported by the JSPS KAKENHI (grant number JP22K18993 and JP23KJ0050) and the Light Metal Educational Foundation of Japan. A part of this work was conducted at Hokkaido University, supported by the “Nanotechnology Platform” Program of the Ministry of Education, Culture, Sports, Science and Technology (MEXT), Japan.References
- E. T. Sayed, A. G. Olabi, A. H. Alami, A. Radwan, A. Mdallal, A. Rezk and M. A. Abdelkareem, Energies, 2023, 16, 1415 Search PubMed.
- M. C. Argyrou, P. Christodoulides and S. A. Kalogirou, Renewable Sustainable Energy Rev., 2018, 94, 804–821 Search PubMed.
- W.-D. Steinmann, Renewable Sustainable Energy Rev., 2017, 75, 205–219 Search PubMed.
- Q. Yong, Y. Tian, X. Qian and X. Li, Appl. Therm. Eng., 2022, 215, 119048 Search PubMed.
- F. Kosaka, T. Yamaguchi, Y. Ando, T. Mochizuki, H. Takagi, K. Matsuoka and K. Kuramoto, Int. J. Hydrogen Energy, 2021, 46, 4116–4125 Search PubMed.
- Y. Li, J. An, S. Wang, Z. Geng and H. Dong, Adv. Powder Technol., 2024, 35, 104671 Search PubMed.
- S. Sakamoto, T. Matsuoka and Y. Nakamura, Catal. Lett., 2020, 150, 2928–2936 Search PubMed.
- D. Sun and D. S. Simakov, J. CO2 Util., 2017, 21, 368–382 Search PubMed.
- H. Koide, A. Gunji, M. Sugimasa, T. Kawaguchi, C. Tamzysi and T. Nomura, Appl. Energy, 2025, 380, 124942 Search PubMed.
- T. Takahashi, H. Koide, H. Sakai, D. Ajito, A. Kurniawan, Y. Kunisada and T. Nomura, Sci. Rep., 2021, 11, 7539 Search PubMed.
- C. Tamzysi, T. Takahashi, Y. Kunisada, A. Kurniawan and T. Nomura, Chem. Eng. J., 2025, 506, 159753 Search PubMed.
- V. Dieterich, A. Buttler, A. Hanel, H. Spliethoff and S. Fendt, Energy Environ. Sci., 2020, 13, 3207–3252 Search PubMed.
- Y. Liu, O. Ersen, C. Meny, F. Luck and C. Pham-Huu, ChemSusChem, 2014, 7, 1218–1239 Search PubMed.
- A. O. Odunsi, T. S. O'Donovan and D. A. Reay, Appl. Therm. Eng., 2016, 93, 1377–1393 Search PubMed.
- M. Promsen, K. Selvam, Y. Komatsu, A. Sciazko, S. Kaneko and N. Shikazono, Energy Convers. Manage., 2022, 255, 115309 Search PubMed.
- S. Santhanam, M. P. Heddrich, M. Riedel and K. A. Friedrich, Energy, 2017, 141, 202–214 Search PubMed.
- G. Xiao, A. Sun, H. Liu, M. Ni and H. Xu, Appl. Energy, 2023, 331, 120383 Search PubMed.
- N. Javanshir, S. Syri, S. Tervo and A. Rosin, Energy, 2023, 266, 126423 Search PubMed.
- J. Ramsebner, R. Haas, A. Ajanovic and M. Wietschel, Wiley Interdiscip. Rev.: Energy Environ., 2021, 10, e396 Search PubMed.
- E. Zeyen, M. Victoria and T. Brown, Nat. Commun., 2023, 14, 3743 Search PubMed.
- T. Liang, A. Vecchi, K. Knobloch, A. Sciacovelli, K. Engelbrecht, Y. Li and Y. Ding, Renewable Sustainable Energy Rev., 2022, 163, 112478 Search PubMed.
- L. Seyitini, B. Belgasim and C. C. Enweremadu, J. Energy Storage, 2023, 62, 106919 Search PubMed.
- A. J. Carrillo, J. González-Aguilar, M. Romero and J. M. Coronado, Chem. Rev., 2019, 119, 4777–4816 Search PubMed.
- H. M. Ali, T.-u. Rehman, M. Arıcı, Z. Said, B. Duraković, H. I. Mohammed, R. Kumar, M. K. Rathod, O. Buyukdagli and M. Teggar, Prog. Energy Combust. Sci., 2024, 100, 101109 Search PubMed.
- T. Nomura, N. Okinaka and T. Akiyama, ISIJ Int., 2010, 50, 1229–1239 Search PubMed.
- B. Zalba, J. M. Marı́n, L. F. Cabeza and H. Mehling, Appl. Therm. Eng., 2003, 23, 251–283 Search PubMed.
- Y. Zhao, C. Y. Zhao, C. N. Markides, H. Wang and W. Li, Appl. Energy, 2020, 280, 115950 Search PubMed.
- S. Wang, K. Lei, Z. Wang, H. Wang and D. Zou, Chem. Eng. J., 2022, 438, 135559 Search PubMed.
- Y. Ouyang, Y. Luo, X. Gao, H. Gao, G. Wang and X. Shu, Chin. J. Catal., 2024, 60, 128–157 Search PubMed.
- X. Hu, X. Gong, F. Zhu, X. Xing, Z. Li and X. Zhang, Renewable Energy, 2023, 212, 227–237 Search PubMed.
- W. Hua, L. Zhang and X. Zhang, J. Mol. Liq., 2021, 340, 117183 Search PubMed.
- Z.-w. Li, L.-c. Lv and J. Li, Int. J. Heat Mass Transfer, 2016, 98, 550–557 Search PubMed.
- G. Liu, K. Shang, S. Chen and J. Shen, Compos. Sci. Technol., 2025, 111120 Search PubMed , in press..
- R. Fan, N. Zheng and Z. Sun, Int. J. Heat Mass Transfer, 2021, 166, 120753 Search PubMed.
- R. Kizilel, R. Sabbah, J. R. Selman and S. Al-Hallaj, J. Power Sources, 2009, 194, 1105–1112 Search PubMed.
- Z. Ling, F. Wang, X. Fang, X. Gao and Z. Zhang, Appl. Energy, 2015, 148, 403–409 Search PubMed.
- Z. Ling, Z. Zhang, G. Shi, X. Fang, L. Wang, X. Gao, Y. Fang, T. Xu, S. Wang and X. Liu, Renewable Sustainable Energy Rev., 2014, 31, 427–438 Search PubMed.
- S. Mousavi, A. Zadehkabir, M. Siavashi and X. Yang, Appl. Energy, 2023, 334, 120643 Search PubMed.
- M. Safdari, R. Ahmadi and S. Sadeghzadeh, Energy, 2020, 193, 116840 Search PubMed.
- E. Shengxin, Y. Cui, Y. Liu and H. Yin, Energy, 2023, 282, 128805 Search PubMed.
- K. Takizawa, D. A. R. Sanjeew, N. Yagihashi, K. Mimura, Y. Shimizu, M. Jeem and T. Nomura, Chem. Eng. J., 2025, 504, 158558 Search PubMed.
- C. Tamzysi, Y. Shimizu, T. Nakamura, M. Jeem, K. Tanahashi, T. Kawaguchi, K. Mimura, A. Kurniawan and T. Nomura, React. Chem. Eng., 2025, 10, 593–604 Search PubMed.
- H. Mehling, Appl. Sci., 2023, 13, 8682 Search PubMed.
- Q. Zhang, Y. Li, C. Liu, X. Wu, X. Zhang, J. Song, Y. Mao and K. Yuan, J. Mater. Chem. A, 2024, 12, 32526–32547 Search PubMed.
- S. Zhu, M. T. Nguyen and T. Yonezawa, Nanoscale Adv., 2021, 3, 4626–4645 Search PubMed.
- C. Shi, M. Xu, X. Guo, S. Zhu and D. Zou, Adv. Funct. Mater., 2025, 35, 2412914 Search PubMed.
- X. Lu, R. Qian, X. Xu, M. Liu, Y. Liu and D. Zou, Nano Energy, 2024, 124, 109520 Search PubMed.
- W. Su, J. Darkwa and G. Kokogiannakis, Renewable Sustainable Energy Rev., 2015, 48, 373–391 Search PubMed.
- N. P. Pasarkar, M. Yadav and P. A. Mahanwar, J. Polym. Res., 2023, 30, 13 Search PubMed.
- Y. Sheikh, M. O. Hamdan and S. Sakhi, J. Energy Storage, 2023, 72, 108472 Search PubMed.
- J. Lee and B. Jo, Sol. Energy Mater. Sol. Cells, 2021, 222, 110954 Search PubMed.
- J. Li, L. Li, H. Wang, X. Zhu, Y. Chen and L. Jia, Energy Technol., 2020, 8, 2000550 Search PubMed.
- S. Mo, B. Xiao, B. Mo, J. Chen, L. Jia, Z. Wang and Y. Chen, Energy Fuels, 2023, 37, 7490–7500 Search PubMed.
- S. Park and B. Jo, Sol. Energy Mater. Sol. Cells, 2024, 272, 112905 Search PubMed.
- H. Zhang, D. Shin and S. Santhanagopalan, Renewable Energy, 2019, 134, 1156–1162 Search PubMed.
- T. Nomura, C. Zhu, N. Sheng, G. Saito and T. Akiyama, Sci. Rep., 2015, 5, 9117 Search PubMed.
- K. Kashiyama, T. Kawaguchi, K. Dong, H. Sakai, N. Sheng, A. Kurniawan and T. Nomura, Energy Storage, 2020, 2, e177 Search PubMed.
- K. Dong, T. Kawaguchi, Y. Shimizu, H. Sakai and T. Nomura, ACS Omega, 2022, 7, 28313–28323 Search PubMed.
- K. Ishii, K. Kawayama and K. Fumoto, J. Energy Storage, 2023, 59, 106426 Search PubMed.
- M. Jeem, R. Ishida, M. Kondo, Y. Shimizu, T. Kawaguchi, K. Dong, A. Kurniawan, Y. Kunisada, N. Sakaguchi and T. Nomura, ACS Appl. Mater. Interfaces, 2024, 16, 3509–3519 Search PubMed.
- J. Bao, D. Zou, S. Zhu, Q. Ma, Y. Wang and Y. Hu, Chem. Eng. J., 2021, 415, 128965 Search PubMed.
- K. Lei, J. Bao, X. Zhao, H. Wang and D. Zou, Chem. Eng. J., 2022, 446, 137020 Search PubMed.
- J. Lu, N. Sheng and C. Zhu, Sol. Energy Mater. Sol. Cells, 2022, 239, 111652 Search PubMed.
- N. Sheng, H. Guo, C. Zhu and Z. Rao, J. Energy Storage, 2022, 56, 106069 Search PubMed.
- N. Sheng, J. Lu, J. Hu, R. Zhu, L. Fadillah, C. Wang, C. Zhu, Z. Rao and H. Habazaki, Chem. Eng. J., 2021, 420, 129906 Search PubMed.
- C.-C. Lai, S.-M. Lin, Y.-D. Chu, C.-C. Chang, Y.-L. Chueh and M.-C. Lu, Nano Energy, 2016, 25, 218–224 Search PubMed.
- S. Wang, X. Zhao, Z. Wang, Y. Zhang, H. Wang and D. Zou, J. Clean. Prod., 2023, 417, 138058 Search PubMed.
- T.-H. Hsu, C.-H. Chung, F.-J. Chung, C.-C. Chang, M.-C. Lu and Y.-L. Chueh, Nano Energy, 2018, 51, 563–570 Search PubMed.
- T. Kawaguchi, T. Nakamura, M. Kondo, J. C. Mba, K. Dong, Y. Shimizu, M. Jeem and T. Nomura, Chem. Eng. J., 2024, 498, 154782 Search PubMed.
- T. Kawaguchi, H. Sakai, R. Ishida, Y. Shimizu, A. Kurniawan and T. Nomura, J. Energy Storage, 2022, 55, 105577 Search PubMed.
- T. Kawaguchi, H. Sakai, N. Sheng, A. Kurniawan and T. Nomura, Appl. Energy, 2020, 276, 115487 Search PubMed.
- Q. Li, X. Ma, X. Zhang, J. Ma, X. Hu and Y. Lan, Mater. Lett., 2022, 308, 131208 Search PubMed.
- Y. Shimizu, S. Cho, T. Kawaguchi, K. Tanahashi, K. Dong, T. Nakamura, A. Kurniawan, M. Jeem and T. Nomura, J. Energy Storage, 2024, 75, 109535 Search PubMed.
- C. Zhou, L. Jiang, Z. Gu, C. Wang, L. He, L. Huang, Z. Li and K. Li, Chem. Eng. J., 2023, 471, 144610 Search PubMed.
- N. Sheng, C. Zhu, G. Saito, T. Hiraki, M. Haka, Y. Hasegawa, H. Sakai, T. Akiyama and T. Nomura, J. Mater. Chem. A, 2018, 6, 18143–18153 Search PubMed.
- J. C. Mba, H. Sakai, K. Dong, Y. Shimizu, M. Kondo, T. Nakamura, M. Jeem and T. Nomura, J. Energy Storage, 2024, 94, 112066 Search PubMed.
- T. Nomura, J. Yoolerd, N. Sheng, H. Sakai, Y. Hasegawa, M. Haga, G. Saito and T. Akiyama, Sol. Energy Mater. Sol. Cells, 2018, 187, 255–262 Search PubMed.
- R. Qian, K. Weng, M. Liu, L. Huang and D. Zou, Chem. Eng. J., 2024, 496, 153929 Search PubMed.
- N. Sheng, C. Zhu, H. Sakai, Y. Hasegawa, T. Akiyama and T. Nomura, Sol. Energy Mater. Sol. Cells, 2019, 200, 109925 Search PubMed.
- K. Wang, T. Zhang, T. Wang, C. Xu, F. Ye and Z. Liao, Chem. Eng. J., 2022, 436, 135054 Search PubMed.
- H. Wei, C. Wang, S. Yang, J. Han, M. Yang, J. Zhang, Y. Lu and X. Liu, Chem. Eng. J., 2020, 391, 123539 Search PubMed.
- J. Zhang, M. Zhang, H. Li, H. Gu, D. Chen, C. Zhang, Y. Tian, E. Wang and Q. Mu, J. Mater. Sci. Technol., 2024, 176, 48–56 Search PubMed.
- J. Zhang, M. Zhang, H. Gu, H. Li, A. Huang, S. Yang, T. Long and X. Zhang, Chem. Eng. J., 2024, 485, 150119 Search PubMed.
- Y. Shimizu, T. Kawaguchi, H. Sakai, K. Dong, A. Kurniawan and T. Nomura, Sol. Energy Mater. Sol. Cells, 2022, 246, 111874 Search PubMed.
- T. Kawaguchi, J. Yoolerd, H. Sakai, Y. Shimizu, A. Kurniawan and T. Nomura, Sol. Energy Mater. Sol. Cells, 2022, 237, 111540 Search PubMed.
- Q. Li, X. Ma, X. Zhang, J. Ma, J. Liu, X. Hu and Y. Lan, J. Alloys Compd., 2021, 888, 161606 Search PubMed.
- T. Nomura, J. Yoolerd, N. Sheng, H. Sakai, Y. Hasegawa, M. Haga and T. Akiyama, Sol. Energy Mater. Sol. Cells, 2019, 193, 281–286 Search PubMed.
- K. Li, X. Cheng, N. Li, X. Zhu, Y. Wei, K. Zhai and H. Wang, J. Mater. Chem. A, 2017, 5, 24232–24246 Search PubMed.
- M. Aoki, M. Jeem, Y. Shimizu, T. Kawaguchi, M. Kondo, T. Nakamura, C. Fushimi and T. Nomura, Mater. Adv., 2024, 5, 675–684 Search PubMed.
- T. Kawaguchi, M. Jeem, A. Kurniawan, K. Dong, M. Kondo, Y. Sato, T. Nakamura and T. Nomura, J. Energy Storage, 2024, 75, 109635 Search PubMed.
- T. Kawaguchi, H. Sakai, Y. Shimizu, K. Dong, A. Kurniawan and T. Nomura, ISIJ Int., 2022, 62, 2567–2572 Search PubMed.
- T. Kawaguchi, Y. Shimizu, K. Dong, A. Kurniawan and T. Nomura, Energy Storage, 2024, 6, e493 Search PubMed.
- H. Sakai, N. Sheng, A. Kurniawan, T. Akiyama and T. Nomura, Appl. Energy, 2020, 265, 114673 Search PubMed.
- H. Koide, A. Kurniawan, T. Takahashi, T. Kawaguchi, H. Sakai, Y. Sato, J. N. Chiu and T. Nomura, Energy, 2022, 238, 121746 Search PubMed.
- A. Kurniawan, R. Tsukiashi, Y. Shimizu, Y. Sato, T. Nakamura, J. N. Chiu and T. Nomura, Appl. Therm. Eng., 2025, 259, 124696 Search PubMed.
- T. Nakamura, Y. Sato, L. Shan, J. N. Chiu, S. Fujii and T. Nomura, Experimental Study of a Bench Scale Packed-bed Latent Heat Storage Unit with Al-Si based PCM pellets, Proceedings of the 16th IEA ES TCP International Conference on Energy Storage, Lyon, France, 2024 Search PubMed.
- Q. Ma, D. Zou, Y. Wang and K. Lei, Ceram. Int., 2021, 47, 24240–24251 Search PubMed.
- X. Yao, Y. Chang, H. Gu, J. Guo and D. Zou, Chem. Eng. J., 2023, 477, 146967 Search PubMed.
- Z. Wang, K. Lei, S. Wang, F. Wang, Y. Zhang and D. Zou, Ceram. Int., 2023, 49, 19136–19148 Search PubMed.
- Z. Wang, M. Liu, Y. Chang, J. Guo and D. Zou, Ceram. Int., 2023, 49, 36560–36571 Search PubMed.
- Z. Wang, S. Zhu, X. Zhao, L. Huang and D. Zou, Int. J. Therm. Sci., 2022, 176, 107478 Search PubMed.
- S. P. Vudata and D. Bhattacharyya, Int. J. Heat Mass Transfer, 2021, 181, 122025 Search PubMed.
- Z. Alipour, V. B. Borugadda, H. Wang and A. K. Dalai, Chem. Eng. J., 2023, 452, 139416 Search PubMed.
- M. Li, Z. Sun and Y. H. Hu, J. Mater. Chem. A, 2021, 9, 12495–12520 Search PubMed.
- A. Rawson, C. Villada, M. Kolbe, V. Stahl and F. Kargl, Energy Storage, 2022, 4, e299 Search PubMed.
- A. H. Alami, A. G. Olabi, A. Mdallal, A. Rezk, A. Radwan, S. M. A. Rahman, S. K. Shah and M. A. Abdelkareem, Int. J. Thermofluids, 2023, 18, 100340 Search PubMed.
- E. González-Roubaud, D. Pérez-Osorio and C. Prieto, Renewable Sustainable Energy Rev., 2017, 80, 133–148 Search PubMed.
- Y. Tian and C.-Y. Zhao, Appl. Energy, 2013, 104, 538–553 Search PubMed.
- D. L. Barth, J. E. Pacheco, W. J. Kolb and E. E. Rush, J. Sol. Energy Eng., 2002, 124, 170–175 Search PubMed.
- U. Herrmann, B. Kelly and H. Price, Energy, 2004, 29, 883–893 Search PubMed.
- P. Speidel, B. Kelly, M. Prairie, J. Pacheco, R. Gilbert and H. Reilly, J. Phys. IV, 1999, 9, Pr3-181–Pr3-187 Search PubMed.
- V. Novotny, V. Basta, P. Smola and J. Spale, Energies, 2022, 15, 647 Search PubMed.
- A. Vecchi, K. Knobloch, T. Liang, H. Kildahl, A. Sciacovelli, K. Engelbrecht, Y. Li and Y. Ding, J. Energy Storage, 2022, 55, 105782 Search PubMed.
- J. Liu, J. Xu, Z. Su, Y. Zhang and T. Jiang, Sol. Energy Mater. Sol. Cells, 2023, 251, 112114 Search PubMed.
- H. Yang, W. Zhang, Y. Zhu, Y. Shao, Y. Shao and X. Zhang, J. Energy Storage, 2022, 56, 106047 Search PubMed.
- F. Jiang, Z. Ge, X. Ling, D. Cang, L. Zhang and Y. Ding, Renewable Energy, 2021, 179, 327–338 Search PubMed.
- F. Jiang, L. Zhang, D. Cang, X. Ling and Y. Ding, Ceram. Int., 2021, 47, 26301–26309 Search PubMed.
- L. Sang and Y. Xu, J. Energy Storage, 2020, 31, 101611 Search PubMed.
- G. Xu, G. Leng, C. Yang, Y. Qin, Y. Wu, H. Chen, L. Cong and Y. Ding, Sol. Energy, 2017, 146, 494–502 Search PubMed.
- G. Leng, G. Qiao, Z. Jiang, G. Xu, Y. Qin, C. Chang and Y. Ding, Appl. Energy, 2018, 217, 212–220 Search PubMed.
- Q. Li, L. Cong, X. Zhang, B. Dong, B. Zou, Z. Du, Y. Xiong and C. Li, Sol. Energy Mater. Sol. Cells, 2020, 211, 110511 Search PubMed.
- J. R. Eggers, M. von der Heyde, S. H. Thaele, H. Niemeyer and T. Borowitz, J. Energy Storage, 2022, 55, 105780 Search PubMed.
- E. Garitaonandia, P. Arribalzaga, I. Miguel and D. Bielsa, Energies, 2024, 17, 1515 Search PubMed.
- G. Zanganeh, A. Pedretti, S. Zavattoni, M. Barbato and A. Steinfeld, Sol. Energy, 2012, 86, 3084–3098 Search PubMed.
- V. Dreißigacker, H. Müller-Steinhagen and S. Zunft, Heat Mass Tran., 2010, 46, 1199–1207 Search PubMed.
- T. Mitterlehner, G. Kartnig and M. Haider, FME Trans., 2020, 48, 427–431 Search PubMed.
- R. Larsen, T. L. Villadsen, J. K. Mathiesen, K. M. Jensen and E. D. Boejesen, ChemRxiv, 2023, preprint, DOI:10.26434/chemrxiv-2023-k73qz-v2.
- R. G. Munro, J. Am. Ceram. Soc., 1997, 80, 1919–1928 Search PubMed.
- S. Elomari, M. D. Skibo, A. Sundarrajan and H. Richards, Compos. Sci. Technol., 1998, 58, 369–376 Search PubMed.
- Y. Shimizu and T. Nomura, J. Alloys Compd., 2025, 1017, 179006 Search PubMed.
- S. Elomari, R. Boukhili and D. J. Lloyd, Acta Mater., 1996, 44, 1873–1882 Search PubMed.
- T. Huber, H. P. Degischer, G. Lefranc and T. Schmitt, Compos. Sci. Technol., 2006, 66, 2206–2217 Search PubMed.
- E. J. Mittemeijer, P. V. Mourik and T. H. De Keijser, Philos. Mag. A, 1981, 43, 1157–1164 Search PubMed.
- H. W. Russell, J. Am. Ceram. Soc., 1935, 18, 1–5 Search PubMed.
- M. Matsushita, M. Monde and Y. Mitsutake, Int. J. Hydrogen Energy, 2014, 39, 9718–9725 Search PubMed.
- J. S. o. T. Properties, Thermophysical Properties Handbook, Yokendo Co. Ltd., 1st edn, 2008 Search PubMed.
- J. Czyzewski, A. Rybak, K. Gaska, R. Sekula and C. Kapusta, Materials, 2020, 13, 5480 Search PubMed.
- D. S. Smith, S. Fayette, S. Grandjean, C. Martin, R. Telle and T. Tonnessen, J. Am. Ceram. Soc., 2003, 86, 105–111 Search PubMed.
- D. Kunii and J. M. Smith, AIChE J., 1960, 6, 71–78 Search PubMed.
- R. Bjørk, V. Tikare, H. L. Frandsen and N. Pryds, J. Am. Ceram. Soc., 2013, 96, 103–110 Search PubMed.
- T.-S. Yeh and M. D. Sacks, J. Am. Ceram. Soc., 1988, 71, C484–C487 Search PubMed.
- C. Qian, K. Hu, Z. Shen, Q. Wang, P. Li and Z. Lu, Ceram. Int., 2023, 49, 17506–17523 Search PubMed.
- Z. Yang, Z. Yin, D. Wang, H. Wang, H. Song, Z. Zhao, G. Zhang, G. Qing, H. Wu and H. Jin, Mater. Chem. Phys., 2020, 241, 122453 Search PubMed.
- N. Kucukdogan, L. Aydin and M. Sutcu, Thermochim. Acta, 2018, 665, 76–84 Search PubMed.
- I. H. Tavman, Int. Commun. Heat Mass Transfer, 1998, 25, 723–732 Search PubMed.
- The Ceramic Society of Japan, Handbook of Ceramics, Gihodo Shuppan Co., Ltd., 2nd edn, 2002 Search PubMed.
- JIS A5308: 2024 Ready-Mixed Concrete (In Japanese) Search PubMed.
- A. Gil, M. Medrano, I. Martorell, A. Lázaro, P. Dolado, B. Zalba and L. F. Cabeza, Renewable Sustainable Energy Rev., 2010, 14, 31–55 Search PubMed.
- S. Hasnain, Energy Convers. Manage., 1998, 39, 1127–1138 Search PubMed.
- T. Bauer, N. Pfleger, N. Breidenbach, M. Eck, D. Laing and S. Kaesche, Appl. Energy, 2013, 111, 1114–1119 Search PubMed.
| This journal is © The Royal Society of Chemistry 2025 |







