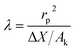A review of the design, applications, and mechanisms of electrically assisted reverse osmosis and nanofiltration processes
Haya
Nassrullah
ab,
Jamaliah
Aburabie
a,
Shabin
Mohammed
c,
Nidal
Hilal
a and
Raed
Hashaikeh
*a
aNYUAD Water Research Center, New York University Abu Dhabi, P. O. Box 129188, Abu Dhabi, United Arab Emirates. E-mail: raed.hashaikeh@nyu.edu
bChemical and Biomolecular Engineering Department, New York University, Tandon School of Engineering, NY 11201, USA
cDepartment of Chemical Engineering, Higher Colleges of Technology, Abu Dhabi, United Arab Emirates
First published on 14th August 2025
Abstract
Reverse osmosis (RO) and nanofiltration (NF) are the main membrane technologies used for water treatment applications. Despite their widespread use, these technologies face several limitations, including membrane fouling, limited selectivity towards certain ions or contaminants, and a lack of tunability in separation performance. The use of electricity, either by applying an external electric field or by integrating electrically conductive membranes, has shown potential to enhance membrane performance and expand process capabilities. Electrically assisted RO and NF processes have been investigated for nearly two decades, yet their development remains at an early stage due to several knowledge gaps. This paper provides a comprehensive review of studies on electrically assisted RO and NF systems, with a focus on membrane fabrication methods, system design considerations, mechanisms of action, and application areas. The aim is to highlight key limitations and research gaps to help guide future improvements of these processes. The future of electrically assisted NF and RO processes lies in the development of scalable fabrication strategies, evaluation of membrane stability, benchmarking against conventional systems, improved module design, and consideration of both energy and economic requirements.
1. Introduction
Water scarcity has become a persistent challenge affecting individuals and communities in many parts of the world. Between 1996 and 2005, it was estimated that approximately four billion people experienced severe water scarcity for at least one month each year.1 With the compounding effects of climate change, population growth, and economic development,2–4 this number is expected to rise. In fact, by 2050, water demand is projected to increase by 20–30%,5,6 indicating the critical need for potential solutions to water scarcity. Desalination and wastewater reclamation offer effective solutions to reduce water scarcity and achieve water security across the globe.7,8 Among the available technologies, Reverse Osmosis (RO) and Nanofiltration (NF) membrane processes are pivotal advanced filtration solutions for water treatment.9 RO membranes are capable of rejecting the smallest contaminants in water, such as monovalent ions.10 NF membranes have a larger pore size than RO, with a surface charge allowing them to exhibit high rejection for polyvalent ions and low molecular weight organic solutes.10,11 Despite the promising perspectives for RO and NF in desalination and wastewater treatment, these technologies still suffer from several limitations including membrane fouling, insufficient rejection of certain contaminants, limited selectivity, and lack of tunability.The use of electricity with membranes, referred to as electrically assisted membrane processes, is an effective technique introduced to overcome limitations and improve the performance of membranes. Electrically assisted membrane processes are categorized into two main approaches, as illustrated in Fig. 1a: electro-filtration and electrically conductive membranes (ECMs). Electro-filtration involves applying an external electric field across a conventional membrane module. When applied to NF and RO membranes, electro-filtration is termed electro-nanofiltration (ENF) and electro-reverse osmosis (ERO), respectively. ECMs are membranes fabricated from electrically conductive materials that function simultaneously as filters and electrodes during operation. It is important to note that the schematics in Fig. 1a illustrate the use of a flat-sheet ECM; however, hollow-fiber and tubular ECMs are also used as discussed elsewhere.12
The use of electricity in water treatment dates back to 1959, when Bier,13 applied an electric field to dewater colloidal suspensions. Several studies followed Bier's work, offering improvements on the proposed process and modifications to the theoretical equations explaining the process for better understanding.14–16 Around the same time, Giddings17 and Grushka et al.18 introduced a process named field-flow fractionation, wherein an electric field applied perpendicular to the flow was used to separate macromolecules or particles. Building on these studies, Henry Jr et al.19 developed electro-filtration to enhance filtration performance. Following Henry's work, several studies were published in the literature on electro-microfiltration (EMF)20–22 and electro-ultrafiltration (EUF).23–27 It was not until 1997 that Pupunat et al.28 introduced ENF, which was developed for anion fractionation (SO42− and Cl−), and since then, the use of ENF has expanded to various applications. More recently, in 2014, ERO was introduced.29 A timeline of the progression of ERO and ENF applications is presented in Fig. 1b.
Besides the development of electro-filtration processes, in the late 20th century, researchers began working on fabricating ECMs. With considerable advancements in conductive polymer research, the first microporous ECM was fabricated in 1985 using polyacetylene.30 Since then, various ECMs have been developed for microfiltration (MF) and ultrafiltration (UF).12 The development of NF and RO ECMs came much later in time relative to MF and UF membrane, mainly due to the fabrication challenges.31 The first tight NF ECM was developed in 2013 by Lannoy et al.,32 and was later patented in 2017.33 In the past five years, several research groups have introduced innovative fabrication methods for RO and NF ECMs, resulting in an increase in publications in this area. Fig. 1b shows the timeline of the applications of RO and NF ECMs up to this year.
With the surge in studies on RO and NF ECMs, and the increasing interest in electrically assisted RO and NF processes over the past two decades (as shown in Fig. 1c), a comprehensive review on this topic has become necessary.
Most previously published review papers on electrically assisted membrane technologies have focused on one aspect of the field which is ECMs and did not cover electro-filtration processes. These reviews are outdated, with the latest publication in 2021.12,34,35 Only a few reviews covered electro-filtration processes including EUF and EMF, but they too are outdated.36,37 Other reviews have focused on ECMs prepared from specific materials, such as 1D and 2D nanomaterials,38 carbon-based,39–41 inorganic and ceramic,42–44 and polymer-based45,46 or for particular applications, including toxic pollutant removal,47 fouling mitigation and control,48,49 advanced oxidation processes,50 electrooxidation of organic pollutants,51 organic wastewater treatment,52 and drinking water purification.53
A major limitation of all these previous review papers, whether they focus on ECMs, discuss electro-filtration systems briefly, or concentrate on specific materials or applications of ECMs, is that the majority of them have focused on UF and MF processes, as the use of electricity was predominantly applied in these technologies in the past.31 None has provided a systematic review of electrically assisted RO and NF processes.
Therefore, this paper aims to provide a comprehensive review on electrically assisted RO and NF processes, including ERO and ENF systems as well as NF and RO ECMs. It covers the design of ENF and ERO systems, and the fabrication methods for RO and NF ECMs. It also explores the diverse applications of electricity in each technology and elucidates the underlying mechanisms driving the performance enhancements. Additionally, this review discusses the limitations of current approaches and offers future perspectives to guide ongoing research. By addressing these gaps, this review aims to provide researchers with valuable insights to further advance the field of electrically assisted RO and NF membrane processes and optimize their performance.
2. Design of electrically assisted NF and RO systems
As discussed earlier, to enhance the performance of RO and NF systems using electricity, researchers have explored the application of external electric fields in both non-conductive and ECM systems (Fig. 1a). The former technique involves applying an electric field externally to a conventional membrane system to influence the separation process. The electric field affects the movement of charged particles and ions within the feed solution and across the membrane. On the other hand, ECMs can interact with the feed solution differently compared to conventional non-conductive membranes. This conductivity can be intrinsic, due to the material properties of the membrane, or achieved through coating or doping with conductive materials. Subsequent sections will discuss both cases with special attention to the design and fabrication aspects of each approach.2.1. Non-conductive membrane systems (ERO and ENF)
In ERO and ENF, a conventional membrane is placed between two electrodes in a custom-designed filtration cell. This configuration provides several advantages over using ECMs.54,55 For instance, producing ECMs with precise molecular-level sieving capabilities is both costly and challenging, complicating their scalability and widespread use. They can also be prone to electrochemical degradation, which may compromise membrane integrity and lead to inconsistent separation performance over time. The geometry of the electrode is determined by the design of the membrane module. For example, tubular membrane systems are often paired with rod-shaped electrodes due to their compatibility with the cylindrical geometry, while flat sheet membrane modules utilize rectangular electrodes to match their planar structure. In sandwiched membrane configurations, electrode size and spacing must be carefully managed. Based on previous studies, the spacing between electrodes significantly affects the electric field strength which changes both the flux and separation performance, and therefore must be optimized.56,57 Ideally, electrode spacing should be minimized to maximize the electric field effect but not so much as to cause electrical short circuits. Additionally, the electrode positioned on the permeate side of the module must possess sufficient porosity to allow unobstructed flow of the filtered permeate. In terms of electrode materials, previous research has often implemented commercially available options such as stainless steel, copper, and titanium sheets. However, some studies in the past have used lab-made electrodes with improved efficiency and durability for ERO and ENF systems. For example, a custom-designed Ti/TiO2–RuO2 porous electrode has been developed, either used alone or in combination with stainless steel mesh in studies led by Benqiao He and his research team.57–59 The fabrication of these electrodes adopts a patented technology, which involves a conducting base made from a combination of metal oxides, film-forming metals, and non-film-forming conductors.60 This method enhances both the efficiency and durability of the electrodes. Their unique fabrication process aims to address common issues associated with titanium-based electrodes, offering improvements in performance and longevity. David M. Warsinger and colleagues recently investigated the use of porous SnO2–Sb electrodes, fabricated via a modified sol–gel method, as anodes for ENF aimed at removing Perfluorooctanoic Acid (PFOA) through electrochemical reactions.61 The electrode preparation method was selected to ensure stability under the applied voltages required for electrochemical reactions, reducing the risk of degradation or the formation of undesired byproducts during treatment.The studies discussed so far have used a sandwich configuration, with electrodes placed on opposite sides of the membrane, but alternative designs have also been proposed. A recent investigation introduced an innovative design where both electrodes were positioned within the feed chamber to assess the performance of ERO.62 The study used carbon cloth and a platinum plate as the cathode and anode, respectively, with a separation gap of 2.5 mm. The authors suggest that this design eliminated the need for a conductive membrane or modifications to the membrane surface with a conductive layer. Accordingly, the use of external electrodes preserves the original membrane surface properties. However, the electrode spacing requires further optimization to reduce energy consumption. Alternatively, a recent study examined the potential dual functionality of the electrode.63 It evaluated two different anode configurations while keeping the cathode fixed as a porous stainless-steel mesh in the permeate section. The first configuration involved a mixed titanium oxide plate coated with ruthenium and iridium, positioned 2 mm above the membrane. The second configuration utilized a similar mixed metal oxide mesh with diamond-shaped openings placed directly on the membrane surface, simultaneously serving as a spacer. Positioning the electrode on the membrane surface effectively reduced the distance between the electrodes to the thickness of the membrane. This setup allowed the electrode to function both as a spacer and as a means to enhance biofouling and scaling control through the production of chlorine and protons. This design integrates the conventional RO configuration with an electrochemical process, offering a promising approach for scalable industrial applications.
In general, recent advancements in electrode design and fabrication are enhancing the effectiveness and durability of ERO and ENF systems. The integration of innovative materials and design strategies, such as porous electrodes and dual-function components, provides promising solutions to address the limitations of traditional ECMs. A key design challenge for ERO and ENF processes is the increased external voltage requirement due to the resistance posed by the membranes. Therefore, it is crucial to focus on optimizing overall energy consumption by exploring novel configurations. As these technologies continue to evolve, they have the potential to improve their scalability and operational efficiency, paving the way for more effective and sustainable membrane filtration processes.
2.2. ECM systems
In RO and NF systems incorporating ECMs, the membrane itself serves as an anode or cathode alongside a counter electrode. The design of these systems focuses primarily on developing membranes that combine high separation performance with excellent electrical conductivity and stability. Consequently, a key challenge in this area is achieving precise molecular sieving capabilities that meet the selectivity requirements of RO and NF processes. The fabrication of ECMs has been effectively achieved using a range of materials, including both organic and inorganic options such as conducting polymers, carbon-based materials, and metals.35 The literature reveals two primary approaches to fabricating ECMs: (1) developing membranes with intrinsic electrical conductivity and (2) applying surface modifications to commercial or lab-made membranes.Historically, polyaniline (PANI) has been recognized as the most widely used polymeric material for developing ECMs, owing to its stable chemical properties and low production costs. PANI is often enhanced with suitable dopants, such as HCl or organic acids, to improve its electrical conductivity.64 However, a common challenge is HCl leaching over time, which can lead to a gradual decrease in membrane conductivity. Furthermore, the doped casting solution is typically used in membrane fabrication via non-solvent-induced phase inversion, resulting in significant organic waste. To address these issues, a recent study proposed a novel approach in which PANI membranes produced through phase inversion are immersed in solutions of long-chain inorganic salts, such as poly(sodium 4-styrene sulfonate), sodium dodecyl sulfate (SDS), and sodium dodecylbenzene sulfonate (SDBS).65 This method minimizes waste generation and reduces fabrication costs, with the potential for reuse. However, due to the limited electrical conductivity of conducting polymers, the use of nanomaterials, whether as building blocks or as fillers in a polymer matrix has emerged as the most effective approach for creating membranes with high electrical conductivity. Materials such as MXenes, reduced graphene oxide (rGO, the conductive form of typically insulating GO), carbon nanotubes (CNTs), and carbon nanostructures (CNS) have shown significant effectiveness in this context where membrane fabrication is accomplished through techniques such as interfacial polymerization, casting, or vacuum filtration.
Our team has previously developed membranes with enhanced conductivity using a polymer/CNS composite. The fabrication process involved casting, followed by a combination of chemical cross-linking and evaporative casting. In the initial study, we proposed casting a homogeneous solution of alginate and CNS, followed by chemical cross-linking and the removal of residual solvents.66 Pore formation was achieved through the elution of pore-forming agents of varying molecular sizes. In a subsequent study, a homogeneous dispersion of networked cellulose (NC) was combined with CNS and underwent chemical cross-linking. This was followed by evaporative casting, resulting in membranes that demonstrated impressive conductivity of up to 6044 ± 6.404 S m−1.67 The fibrous morphology of NC, along with controlled chemical cross-linking, facilitated membranes with varying molecular weight cut-offs without the aid of pore-forming agents. However, the dense nature of the membranes produced through casting and evaporation often results in low permeability, despite exhibiting high electrical conductivity.
To overcome the low permeability and potential defects associated with directly mixing conductive components into the polymer matrix, a two-step fabrication approach involving distinct layers has been proposed by many researchers. This method consists of two phases: first, constructing a layer of conductive material, followed by polymerization and cross-linking. An example of this technique is the combination of vacuum filtration and subsequent IP, which produces thin film composite (TFC) ECMs. The unique architecture of TFC membranes allows for the optimization of support, intermediate, and selective layers to achieve the desired membrane properties. Leveraging the electrical conductivity of CNTs, numerous studies have reported the development of nanocomposite membranes via IP on CNT-deposited supports.68–71 This approach has yielded nanocomposite RO and NF membranes with electrical conductivity values reaching up to 3707 ± 284 S m−1.71 However, despite this high electrical conductivity, several challenges persist in their fabrication. First, the use of vacuum filtration for incorporating conducting materials complicates the scalability of the production process. Additionally, the compatibility and stability of the functional interlayer during prolonged operation require further investigation. As an alternative, modifications to IP using functional molecules have been suggested; however, the resulting electrical conductivity levels fall short of those achieved with nanocomposite membranes.72–75 For example, a study by Wang et al. demonstrated the incorporation of electroactive molecules, specifically Cucurbit[n]uril molecular containers, into the organic phase prior to IP, resulting in the formation of voltage-gated membranes.75 Similarly, a recent investigation reported the development of electrically responsive membranes by utilizing conductive monomers, such as oligoaniline, as a replacement for the traditional piperazine (PIP) monomer in the amine phase.72 These innovative approaches address the challenges associated with incorporating conductive fillers within the polymer matrix and illustrate the potential for scalable production of electrically responsive membranes via straightforward IP techniques. However, the costs related to these novel molecules, along with their comparatively lower electrical conductivity, necessitate further investigation and evaluation.
Alternatively, Jiansuxuan Chen et al.76 proposed the fabrication of TFC ECM on hydrogel-coated stainless-steel mesh. The use of stainless steel, rather than conventional non-woven fabric, enhanced the membrane electrical conductivity. However, the authors did not provide the overall electrical conductivity exhibited by the TFC membranes, as the hydrogel and polyamide layers likely influenced the conductivity offered by the stainless-steel mesh. Moreover, the use of stainless steel as a substrate limits the mechanical flexibility of the membrane. In addition to traditional IP, researchers have investigated the fabrication of NF ECMs by polymerizing PANI on supports decorated with CNTs.77,78 However, the scalability might be questionable, as vacuum filtration is used to deposit the CNT layer, and the maximum conductivity achieved with this method remains lower than that of membranes produced through IP.
Mere filtration has been identified as a straightforward technique for creating ECMs, especially when utilizing 2D materials as building blocks. This fabrication process involves either vacuum or pressure to filter nanomaterials at a desired concentration and volume homogenously dispersed in an appropriate solvent, onto a suitable membrane support. This method is facile and allows precise control over the membrane thickness.79 As mere assembly of nanomaterials through filtration could compromise the stability and robustness of the membrane, researchers have also demonstrated chemical cross-linking or cation intercalation to stabilize the membrane.80–82 For instance, molten salt intercalation was introduced as an alternative to the conventional cation intercalation involving aqueous solution that can potentially enter the interlayers and induce swelling.82 This method facilitated interlayer spacing modulation in addition to enhancing the structural integrity of the laminated layers.
As mentioned before, filtration-assisted membrane fabrication is used for 2D materials, making the optimization of membrane thickness critical. This is due to the highly tortuous pathways created by the stacking of nanosheets within the selective layer. As a result, it is essential to either fabricate an extremely thin selective layer or intercalate nanomaterials to increase the interlayer spacing. The intercalated material functions as a spacer and may also enhance the overall electrical properties of the membrane. For instance, when the 2D material exhibits limited electrical conductivity, it is often combined with CNTs, either as a single layer or through a two-layer approach using sequential deposition. The two-layer approach through sequential deposition, enables the formation of a thin selective layer by first filtering a CNT network onto the support, followed by lamination of the active material at the desired thickness.80,83 In contrast, depositing a mixed solution as a single layer can minimize restacking and preserve interlayer spacing, maintaining open channels that facilitate water permeation.84,85 The major advantage of filtration-assisted membrane fabrication is that it allows the membrane formation merely by the assembly of nanoparticles without involving polymers other than the membrane support. This enables fabrication of membranes with outstanding conducting properties even up to 1.1 × 105 S m−1 as reported in.83 However, membranes made from 2D materials using vacuum filtration are known to have poor reproducibility and lack mechanical robustness, making them unsuitable for the high-pressure conditions required in RO and NF systems. These membranes are often developed for fundamental studies, and further research is needed to improve their durability and scalability.
Surface modification using conductive components is also reported as a facile strategy to develop ECMs. The key task is to identify a suitable approach that can impart electrical conductivity without disturbing the membrane performance, particularly the permeance. Sputter coating of titanium on ceramic membranes was reported to maintain the inherent surface properties including surface wettability and structure, while still achieving a remarkable electrical conductivity of 5.1 × 102 S m−1.86 However, the applicability of e-beam deposition is limited to ceramic membranes, as the high-energy process can damage polymeric substrates. In a different study, Bongyeon Jung et al.87 proposed surface coating with percolating functionalized CNTs using an airbrush spray on commercial RO membranes, however, further modification with polyvinyl alcohol (PVA) solution was necessary to ensure the stability of the coated layer and improve the surface wettability of the membrane. Recently, a more versatile facile and inexpensive approach was demonstrated that can be extended to a variety of conducting materials on non-conductive membranes. This approach consists of two steps: (1) the deposition of a glutaraldehyde-conducting material composite onto the membrane via vacuum or pressure-assisted assembly, and (2) cross-linking using a poly(ethyleneimine) (PEI) solution. This technique is applicable to a wide range of conducting materials, with the final conductivity influenced by the specific material used for modification. However, a significant challenge remains in scaling up the pressure/vacuum filtration process for the deposition of conducting elements on the membrane surface.88 The fabrication methods of NF and RO ECMs are illustrated in Fig. 2 and summarized in Table 1, along with key remarks on each approach. Based on this comparison, interfacial polymerization emerges as the most suitable method for fabricating RO and NF ECMs, as it builds on a well-established membrane fabrication technique and yields membranes with good selectivity and permeability, comparable to commercial membranes. Although further work is needed to evaluate feasibility and scalability, ECMs fabricated via interfacial polymerization currently demonstrate the best performance and durability among all ECMs reported in literature. Overall, scalability remains the key limitation across most ECM fabrication methods mainly due to the high cost of conductive materials, such as CNTs and advanced 2D materials and the lack of established industrial-scale production processes. Moreover, there is limited evaluation of the economic feasibility of ECMs, and as a result, we lack a comprehensive understanding of their practical viability and long-term stability.
| Method | Description | Remarks | References |
|---|---|---|---|
| Casting/phase inversion | Casting the conductive polymeric solution followed by non-solvent induced phase inversion | High scalability, very low electrical conductivity | 64 and 65 |
| Casting/evaporation | Casting the membrane solution and cross-linking reaction followed by evaporation of residual solvents | Low scalability, possibilities of defects, low permeability, high electrical conductivity | 66 and 67 |
| Filtration | Vacuum or pressure assisted assembly on suitable membrane supports | Low scalability, very high electrical conductivity and is suitable only for fabrication with 2D materials alone or in combination | 79–85, 89 and 90 |
| Vacuum filtration or electrically conductive ultrafiltration membrane + interfacial polymerization | Vacuum filtration to deposit the conducting layer or use of an electrically conductive ultrafiltration support, followed by interfacial polymerization to form a selective layer | Low scalability, high electrical conductivity, high permselectivity | 68–71, 91 and 92 |
| Vacuum filtration + polymerization | Vacuum filtration to deposit the conducting layer followed by polymerization forming a selective layer | Low scalability, low permselectivity, high electrical conductivity | 77, 78 and 93 |
| Interfacial polymerization | Interfacial polymerization using conducting monomers or conducting fillers | High scalability, very low electrical conductivity, high permselectivity | 72–75 |
| Coating | Surface pre-treatment to induce conductivity through sputter/spray coating | Low scalability, high conductivity, low stability of the coated layer | 86 and 87 |
3. Applications of electricity and governing mechanisms
3.1. Salt rejection
RO is currently the leading desalination technology worldwide. The global desalination capacity by RO continues to expand as the process undergoes improvements to reduce costs. Commercial RO membranes demonstrate high rejection of salts, achieving up to 99.8% rejection of 32 g per L NaCl feed.94 In comparison, NF exhibits higher permeate flux and requires lower feed pressure, making it an attractive candidate for desalination.93 However, NF shows high rejection for multivalent salt ions (e.g. SO42−, Mg2+, Ca2+) but relatively low rejection for monovalent salt ions (e.g. Na+, Cl−).10,11 For instance, commercial NF200 membranes exhibits only 60% rejection of 35 g per L NaCl at 30 bars.95 This limitation is due to insufficient electrostatic interaction between the charged membrane and monovalent ions, coupled with the fact that the pore size of the membrane is larger than the hydrated ion. Consequently, despite its advantages in terms of permeate production and energy requirements, NF is inadequate as a standalone process for desalination to meet the required permeate quality.10Over the past two decades, several studies have explored the potential of ENF and NF ECMs to improve or regulate monovalent salt rejection in NF. Noël et al.96 were the first to investigate the effect of an electric field on membrane permeability and NaCl rejection. Their results indicated that under varying magnitudes and polarities of applied potential, there was no clear correlation between NaCl rejection and its concentration in the feed. However, in the absence of an electric potential, NaCl rejection decreased as its concentration in the feed increased. Based on these findings, the authors concluded that the electric field did not directly influence ion transport but rather affected the membrane itself. Without electricity, the observed rejection performance was attributed to conformational changes in the SO3− groups in the membrane polymer (sulfonated poly (2,6-dimethyl-1,4-phenylene oxide) (SPPO)) leading to larger pore sizes at higher NaCl concentrations.97 In contrast, under the electric field, the functional group adopted a conformation that remained unaffected by the salt concentration in the feed, justifying the minimal changes in NaCl rejection under different magnitudes and polarities of the electric field. These observations were specific to this membrane type and did not indicate a promising application of ENF for improving monovalent salt rejection. Subsequently, Han et al.,98,99 demonstrated that the rejection of 1 g per L Na2SO4, and 1 g per L MgCl2 was improved from 68% to 93%, and from 54.1% to 81.2% by using a PA NF membrane as a cathode, and a chitosan membrane as an anode, respectively. The authors attributed this enhanced rejection to the increased surface charge density of the membrane, which strengthens Donnan exclusion. This mechanism arises from electrostatic interactions between the membrane surface charge and ions in the feed.10,100 However, these studies appear to contain a significant misinterpretation of results. Since PA and chitosan are electrically insulating polymers, they cannot function as electrodes, as they would not close the electric circuit. The analysis of these studies was shallow, and the conclusions do not accurately reflect the actual mechanisms driving the enhanced rejection. The effect of the electric field on salt rejection remained unclear until the study by Sun et al.,54 which demonstrated that applying an electric field across the membrane, with the anode placed below the membrane, enhanced salt rejection. They found that salt rejection was related to the membrane's specific capacitance, such that membranes with higher specific capacitance showed higher responsiveness to the electric field. Greater responsiveness implies more significant enhancement in rejection, as an increase in specific capacitance results in more negative charges on the membrane under an electric field. Following the same configuration proposed by Sun et al.,54 a more recent study on the effect of electric field on salt rejection was conducted by Li et al.57 They observed that increasing the distance between electrodes reduced the rejection enhancement, suggesting that the enhancement is dependent on the electric field intensity. This is because the enhancement was partly due to the improved ion diffusion by the dragging force of the electric field. Another important finding was that the effect of the electric field was more pronounced in membranes with a higher structural coefficient (λ):
Salt rejection can also be enhanced by applying electricity to NF ECMs.67,77,78,85,93,101 The optimal results of several studies are presented in Table 2 and the main underlying mechanisms are illustrated in Fig. 3a. Hu et al.85 introduced ECMs fabricated from CNTs and rGO. When used as a cathode, these membranes exhibited significantly enhanced rejection of Na2SO4, NaCl, and CaCl2. The rejection of salt without electricity was attributed to the membrane's intrinsic specific capacitance, due to the in-plane conjugation of rGO and CNTs. The enhanced rejection observed when electricity was applied was attributed to additional sources of capacitance. These include pseudo-capacitance, resulting from the coordination between positive ions in the feed and the oxygen-containing functional groups in the membrane, and electrostatic double layer (EDL) capacitance, which forms due to the accumulation of counter-ions from the feed solution onto the membrane surface. A higher total capacitance led to stronger Donnan interactions such that ions in the bulk solution had to overcome strong electrostatic repulsion forces to pass through the membrane. It is important to note that since the rejection was based on capacitance, the membrane's performance as an anode was only slightly different from its performance as a cathode. However, in all filtration experiments where electricity was applied, the improvement in ion rejection was associated with a reduction in permeate flux. Conversely, Zhang et al.93 and Xing et al.101 demonstrated that salt rejection was enhanced without compromising permeate flux when a negative bias was applied to the membrane. They attributed this improvement to the increased surface charge density of the membranes. Essentially, when an external potential is applied, additional charges are generated on the membrane. To further investigate the mechanism behind electrical enhancement, Donnan steric pore model (DSPM) analysis was conducted. The results revealed that the ion concentration difference across the membrane-feed and membrane–permeate interfaces increased in magnitude with the application of electricity. This occurred because more counterions (cations) were adsorbed on the membrane surface when a negative bias was applied, causing a greater difference in ion concentration between the bulk solution and the area near the membrane surface. As a result, the Donnan potential difference increased, estimated from modelled ion concentrations at the membrane interface, which hindered ion transfer through the membrane. In other studies, changes in salt rejection were also attributed to membrane surface charge, but the mechanism was verified using different methods.67,77,78 Xu et al.77,78 applied the Derjaguin–Landau–Verwey–Overbeek (DLVO) theory to describe the interaction energy between the charged membrane surface and salt ions, both with and without a negative bias applied to the membrane. The interaction energy consists of two components: van der Waals interaction energy and electrostatic EDL interaction energy. The results showed that the membrane exhibited positive interaction energy, indicating repulsive forces. Under an applied external potential, the magnitude of the interaction energy increased. This increase was attributed to the enhanced EDL interaction energy, which is a function of membrane potential, as the van der Waals energy remained unchanged. The authors found a positive correlation between interaction energy and membrane surface charge density, suggesting that the increased interaction between the membrane and ions was due to enhanced membrane surface charge density. Nassrullah et al.67 showed that the percentage of change in rejection when the membrane was used as a cathode differed from when it was used as an anode. This discrepancy, indicated that applying electricity affected the EDL structure within the membrane nanochannels, as EDL formation depends on the magnitude and sign of membrane charge. Consequently, this resulted in changes in the Donnan potential at the membrane/bulk interface. The charges induced on the membrane by the applied external potential led to a depletion of co-ions in the EDL structure within the membrane nanochannels. This depletion caused the accumulation of counter-ions at the membrane/electrolyte interface, leading to changes in the Donnan potential difference between the membrane and the bulk solution. Since positive and negative ions exhibit different valence and diffusion coefficients, they formed different EDL arrangements inside the membrane pores.102,103 Thus, the change in the percentage of rejection when electricity was applied varied accordingly.
| Membrane | Effective filtration area (cm2) | Distance between electrodes (mm) | Feed flowrate (L h−1) | Counter electrode (anode) | Applied pressure (bar) | Salt rejection performance without electricity | Applied DC voltage (V) | Salt rejection performance with electricity | Ref. |
|---|---|---|---|---|---|---|---|---|---|
| rGO-CNT on PVDF | 9.08 | 10 | — | Titanium | 1 | 50 mM Na2SO4: ∼30% | 1 | 50 mM Na2SO4: ∼70% | 85 |
| 50 mM NaCl: ∼25% | 50 mM NaCl: 65% | ||||||||
| 50 mM CaCl2: ∼55% | 50 mM CaCl2: ∼78% | ||||||||
| PANI-PSS/CNT on PVDF | 8.04 | 1 | 2.4 | Titanium mesh | 2 | 5 mM Na2SO4: 81.6% | 2.5 | 5 mM Na2SO4: 93% | 93 |
| 5 mM NaCl: 53.9% | 5 mM NaCl: 82.4% | ||||||||
| GO and EDA-PSS/CNT on PVDF | 6.4 | 0.8 | 2.4 | Titanium mesh | 1 | 2 mM NaCl: 52.4% to 78.3% | 3 | 2 mM NaCl: 78.3% | 56 |
| PANI-PSSA/CNT on PVDF | 7.07 | 20 | — | Titanium ruthenium mesh | 5 | 2 mM Na2SO4: 87.73% | 2.5 | 2 mM Na2SO4: 94.6% | 78 |
| 2 mM NaCl: 61.25% | 2 mM NaCl: 70.42% | ||||||||
| NC-CNS-NCC | 14 | 2 | 50 | Titanium mesh | 5 | 7 mM Na2SO4: 75.6% | 1 | 7 mM Na2SO4: 81.7% | 67 |
| 7 mM NaCl: 43.8% | 7 mM NaCl: 54.6% | ||||||||
| rGO-MoS2 on PVDF | 7.2 | — | 40 | Titanium mesh | — | 5 mM Na2SO4: ∼85% | 2.5 | 5 mM Na2SO4: 93.6% | 101 |
| 5 mM NaCl: ∼65% | 5 mM NaCl: 81.4% | ||||||||
| VARGO on PVDF | 0.012 | 2 | 2.4 | Titanium mesh | 0.8 | 5 mM NaCl: 53.8% | 2.5 | 5 mM NaCl: 88.7% | 105 |
| MXene and cCNF on PVDF | 8.5 | 2 | 15 | Titanium mesh | — | 5 mM Na2SO4: 80.4% | 2 | 5 mM Na2SO4: 88.7% | 89 |
| 5 mM NaCl: 46.2% | 5 mM NaCl: 60.6% |
All the studies discussed so far on NF ECMs have shown that the enhanced salt rejection is often accompanied by a decrease in permeate flux, whether slight or significant. This trade-off between permeability and selectivity is a common challenge in membrane technologies.104 However, achieving simultaneous improvements in salt rejection and water flux is possible.56,105–107 The mechanisms behind enhanced water flux using electricity is summarized in Fig. 3b. Zhang et al.56 introduced a membrane composed of an ethylenediamine–polystyrene sulfonate intercalated graphene oxide selective layer deposited on electrically conductive CNTs (GO&EDA-PSS/CNT). This membrane exhibited both higher water flux and increased salt rejection when an electric voltage was applied across the GO layer. The authors used COMSOL multiphysics numerical simulation to investigate the mechanism behind this enhancement. The results indicated that applying an external electric field (with the membrane serving as a cathode) generates electroosmotic flow, which aligns with the direction of the pressure-driven flow, leading to increased water transport across the membrane. Similarly, Zhang et al.105 developed a vertically aligned reduced GO (VARGO) membrane featuring nanochannels that are parallel to the direction of water transport. When a negative bias was applied to the membrane, the water contact angle (WCA) decreased as the voltage increased. The accelerated entry of water molecules into the membrane nanochannels upon the application of electricity was attributed to the enhanced charge density of the membrane, which led to the accumulation of more counterions on the walls of the nanochannels. The attractive forces between the counterions and the nanochannel walls, combined with the repulsive forces between the nanochannel walls, contribute to expanding the interface, resulting in lower water-wall interfacial tension.
3.2. Salt ion selectivity
As mentioned earlier, NF exhibits higher rejection of multivalent ions compared to monovalent ions. Due to this property, NF has been used to selectively separate SO42− over Cl− in various applications, such as reducing sulfate content in seawater for oil recovery and meeting purity requirements for the chloralkali process.108,109 Previous studies have investigated the effect of an electric field on SO42−/Cl− selectivity.28,107,110 Pupunat et al.28 were the first to introduce the ENF process. They examined several conditions, including different ratios of salt concentrations, overall concentration, voltage, and transmembrane pressure, to understand the effect of the electric field on ion rejection. They found that at a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio of Na2SO4
1 ratio of Na2SO4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) NaCl and applied voltage of 7.5 V with the cathode placed on the permeate side, the rejection of SO42− was only slightly improved, while the rejection of Cl− significantly increased from −17% to 63% and the rejection of Na+ decreased from ∼50% to ∼−200%. Moreover, higher voltage led to a more pronounced effect on ion rejection, whereas higher salt concentrations and increased pressure reduced the impact of the electric field. These results were supported by changes in pH and current density, showing that greater changes in co-ion/counter-ion selectivity were associated with more significant shifts in pH and current density. Based on these findings, the authors concluded that the electric field could be used to manipulate the kinetics of ion transport, though they did not provide an explanation for the underlying mechanism. Similarly, a recent study by Guo et al.107 on ENF, with a solution containing 15 g per L Na2SO4 and 127 g per L NaCl, showed that NaCl rejection significantly increased from 50.9% to 77.6% and Na2SO4 rejection improved from 77.6% to 99.7%. From these studies, it can be concluded that while the electric field enhances Cl− rejection more than SO42−, it may be a useful tool for improving overall salt rejection, but not necessarily for enhancing SO42−/Cl− selectivity. However, the mechanism behind this behavior has not been discussed in these studies and remains unclear.
NaCl and applied voltage of 7.5 V with the cathode placed on the permeate side, the rejection of SO42− was only slightly improved, while the rejection of Cl− significantly increased from −17% to 63% and the rejection of Na+ decreased from ∼50% to ∼−200%. Moreover, higher voltage led to a more pronounced effect on ion rejection, whereas higher salt concentrations and increased pressure reduced the impact of the electric field. These results were supported by changes in pH and current density, showing that greater changes in co-ion/counter-ion selectivity were associated with more significant shifts in pH and current density. Based on these findings, the authors concluded that the electric field could be used to manipulate the kinetics of ion transport, though they did not provide an explanation for the underlying mechanism. Similarly, a recent study by Guo et al.107 on ENF, with a solution containing 15 g per L Na2SO4 and 127 g per L NaCl, showed that NaCl rejection significantly increased from 50.9% to 77.6% and Na2SO4 rejection improved from 77.6% to 99.7%. From these studies, it can be concluded that while the electric field enhances Cl− rejection more than SO42−, it may be a useful tool for improving overall salt rejection, but not necessarily for enhancing SO42−/Cl− selectivity. However, the mechanism behind this behavior has not been discussed in these studies and remains unclear.
NF, with its unique separation properties compared to other membrane-based processes, shows great potential for achieving precise selectivity on the sub-nanometer scale.75 The separation of Mg2+ from Li+, particularly from Salt Lake brine, which is a primary resource for Li+ has recently gained significant attention. However, Mg2+ is also present in these brines and shares similar properties with Li+ in terms of reactivity and hydration radius (Mg2+: 4.28 Å, Li+: 3.82 Å) making their separation challenging. Excellent separation of Mg2+ from Li+ has been achieved using positively charged NF membranes,111,112 however this is often associated with the permeation of small amount of Mg2+, which impacts the purity of Li+ in the permeate. Recently, Li et al.59 demonstrated that the separation between Mg2+ and Li+ increased in an ENF process as the current density increased until complete separation was achieved. At current density of 2.83 mA cm−2 (corresponding to 3.7 V), the rejection of Mg2+ exceeded 99.97%, while the rejection of Li+ was −104.75%, resulting in a separation factor (SMg2+/Li+) of 6825. This behavior was attributed to two factors: (1) Li+ was more easily dehydrated under the electric field compared to Mg2+ ions (as demonstrated by molecular dynamic simulations), and (2) the membrane was in situ reconstructed to become positively charged (as confirmed by zeta potential measurements and XPS analysis of the membranes). Further understanding of the effect of the electric field was obtained in a subsequent study,58 where the sensitivity of selectivity to process parameters and the structural coefficient of the membrane was examined. The study found that to achieve high Mg2+/Li+ separation, it is critical to optimize the feed concentration, pH value, Mg2+/Li+ mass ratio, and pressure, as all these parameters play a role in affecting ion rejection. ENF was also found to be effective in improving the separation of cations with the same charge, such as Mg2+/Sr2+.113 The enhanced separation of Mg2+ and Sr2+ with electricity is due to the promoted dehydration of Sr2+ under an electric field, leading to its permeation, while Mg2+ is rejected by the NF membrane and those which permeate through the membrane precipitate on the permeate side with the generated OH− where the cathode is placed.
Although ENF has proven effective in achieving ion selectivity, a key limitation across these studies is the high voltage required to overcome the resistance of the conventional membrane, resulting in increased energy consumption. To address this, one promising strategy is to fabricate the selective layer directly on an electrically conductive support, which minimizes the distance between the electrodes and reduces the overall resistance. Recently, Lv et al.114 implemented this approach by fabricating a PA membrane on a conductive support incorporating MXene and CNTs. They reported comparable Mg2+/Li+ separation performance to previous ENF studies,58,59 while requiring a lower applied voltage. However, in our view, it remains difficult to conclude whether this design is superior to conventional ENF systems, as the spacing between the electrodes differed across the studies which is a factor that significantly influences separation performance.
3.3. Fouling mitigation
Fouling is one of the major challenges affecting all pressure-driven membrane technologies, including RO and NF.115,116 The main indications of fouling are reduced membrane flux, compromised permeate quality, and a loss of efficiency over time. Fouling also leads to economic losses due to increased energy consumption as pressure requirements rise, the necessity for additional pretreatments, increased need for membrane cleaning, and a decrease in the lifespan of the membrane.117,118 For instance, Jafari et al.119 estimate the cost of fouling in RO plants, fed with surface water or municipal wastewater effluent, to be around 24% of operational expenses (OPEX), while in NF plants, which are fed with anoxic groundwater, it is approximately 11%. Fouling is classified into reversible and irreversible based on the possibility of cleaning and restoring membrane performance.120 Reversible fouling results from cake layer formation or concentration polarization of the rejected materials at the membrane surface, and can be cleaned physically by backwashing or surface washing or chemically with cleaning agents. Irreversible fouling, due to chemisorption of materials to the membrane surface and pore clogging, is more challenging to clean, often requiring harsh extensive chemical cleaning or membrane replacement.Developing unique and effective strategies to mitigate fouling in RO and NF membranes is an active and ongoing area of research. Conventionally, fouling control methods include pretreatment of the feed to NF and RO systems using chemicals or pretreatment technologies, which can render the membrane process less sustainable.118,121 Alternatively, fouling can be reduced by using ECMs or applying an electric field. Fouling is classified to three types: organic, inorganic (scaling), and biofouling, depending on the type of contaminants causing it, with organic fouling being the most common.
NF and RO ECMs have proven effective in reducing fouling. Ahmed et al.122 developed an NF membrane with a high electrical conductivity of 2200 S m−1. They suggested that the membrane's electrical conductivity could be utilized for membrane cleaning via electrolysis, as their demonstration showed that hydrogen bubbles formed on the membrane during electrolysis. Other studies have shown images of fouled NF and RO ECMs before and after electrolysis-based cleaning, as shown Fig. 4a.66,67,123 However, none of these studies have demonstrated membrane performance in the presence of foulants during filtration, nor have they explored the effect of electricity on flux recovery. Recent studies have reported filtration tests of NF ECMs to reduce organic fouling, along with proposed mechanisms.65,74,76,80,81,86,107 For instance, Chen et al.76 fabricated a PA layer on a gel-coated stainless steel mesh. The antifouling performance evaluation revealed that the decline in permeate flux over time was reduced when the membrane was used as a cathode (Fig. 4b). They attributed the reduced fouling to two factors: (1) microbubbles formed on the membrane surface due to water electrolysis, and (2) enhanced electrostatic repulsive forces between the membrane and the foulant (bovine serum albumin (BSA)). The microbubbles formed at the membrane surface are hydrogen bubbles that generate localized turbulence and shear forces, physically disrupting foulant deposition and enhancing surface cleaning through in situ aeration.124–127 While the antifouling effect might be due to hydrogen evolution by electrolysis, as discussed and demonstrated in other studies,66,67,86,122,123 the role of electrostatic repulsion is uncertain in this case. The authors did not report the electrical conductivity of the PA layer facing the feed, and if it was non-conductive, applying voltage would not induce surface charge effects. Conversely, Liu et al.65 suggested that reduced fouling was due to electrostatic repulsive forces, as the active membrane layer facing the feed was electrically conductive (made from PANI, a conductive polymer). They verified changes in membrane charge with electricity by measuring the zeta potential of the membrane after applying voltage. These measurements were conducted across a range of pH values, and at each pH, the zeta potential consistently became more negative as the voltage increased (Fig. 4c).
 | ||
| Fig. 4 (a) Images of the original membrane, with foulants and after electrolysis reprinted from ref. 66. Copyright 2023, with permission from Elsevier; effect of applied voltage on (b) normalized flux over time used with permission of Royal Society of Chemistry, from ref. 75. Copyright 2022; permission conveyed through copyright Clearance Center, Inc. and (c) zeta potential measurements reprinted from ref. 64. Copyright 2024, with permission from Elsevier; (d) comparison of flux decline rate with different membrane cleaning methods and electrically conductive and conventional membranes reprinted from ref. 73. Copyright 2024, with permission from Elsevier. | ||
In addition to enhanced repulsive forces and bubble generation on the membrane, the electrophoresis phenomenon also contributes to reducing organic fouling. In this phenomenon, charged particles move away from the membrane surface and toward the oppositely charged electrode due to the effect of the electric field.80 This phenomenon is dependent on electric field strength and does not require an ECM. Noël et al.128 showed that using ENF, permeability was maintained at a higher value over time compared to when no electric field was applied, suggesting that the electric field may have delayed fouling layer formation. The potential of ENF to reduce organic fouling raises questions about the necessity and effectiveness of ECMs. Moreover, it is crucial to compare electrically-assisted cleaning with other cleaning methods. Jia et al.74 conducted experiments using a commercial NF270 membrane, a non-ECM (M-0), and ECMs based on PEDOT (M-2) for 120 minutes of filtration of sodium alginate (an organic foulant) and NaCl. They then subjected the membranes to different cleaning methods for 2 minutes and repeated the fouling-cleaning process for three cycles. The flux decline rate (FDR%) results showed that electro-assisted cleaning was the most effective method (Fig. 4d). Among the different membranes tested with electro-assisted cleaning, the ECM (M-2) exhibited the lowest FDR%, demonstrating that it combines the effects of electrostatic repulsion and electrophoresis. This suggests that using ECMs for organic fouling mitigation or cleaning is more effective than electrofiltration with conventional membranes. Most studies have applied an electric field continuously, which is an energy-intensive approach.12 Therefore, for practical applications, it is more sustainable to use intermittent voltage application. Anis et al.86 showed that the intermittent use of electricity with a titanium-coated ceramic NF membrane was effective in reducing the decline in flux over time.
RO and NF membranes are prone to inorganic fouling (scaling) when used in water desalination applications. Scaling occurs when insoluble precipitates form from ions such as Ca2+, SO42−, and other metal ions, then they exceed their solubility threshold at the membrane/solution interface due to concentration polarization.12,42,129 The accumulation of these precipitates or salt crystals on the membrane surface leads to a decline in permeate flux and can cause physical damage to the membrane, ultimately degrading its salt rejection performance. The most common scalants are CaSO4 and CaCO3. CaCO3 dissolves readily in acidic environments and can be controlled by adjusting the pH of the solution. However, CaSO4 is pH-insensitive, and therefore requires the use of antiscalants to control scaling.130 Antiscalants, however, have several limitations, including high costs, the risk of water eutrophication in discharged water, and the potential to increase membrane biofouling.71,131,132 Duan et al.68 demonstrated that CaCO3 scaling on an RO ECM (used as an anode) could be effectively removed. As shown in Fig. 5a, the permeate flux declined over the first 460 minutes due to CaCO3 formation. When an electric potential of 2.5 V was applied for 30 minutes with the pressure shut off, the flux was recovered by 98%, then it began to decline again once the process resumed without electricity. The removal of CaCO3 was attributed to water oxidation on the membrane surface, which generated H+ ions, lowering the pH of the solution, thereby facilitating the dissolution of the CaCO3 deposits. The study also showed that using the membrane continuously as an anode could significantly slow down CaSO4 scaling. When a positive potential is applied to the membrane, it becomes charged, and counter-ions form multiple layers to balance this charge, to maintain electroneutrality. Precipitation occurs when counter-ions and co-ions are present in equal ratios. The rate of salt crystal growth depends on the local ratio of cations to anions near the membrane surface.133 Therefore, rapid precipitation or nucleation does not occur on the membrane but rather away from it, as the co-ions and counter-ions are present in nearly equal concentrations, and the resulting precipitates are carried away with the flow. This study demonstrated that continuous DC voltage is effective in reducing scaling. AC voltage has also been shown to contribute to scaling reduction.55,71 Jung et al.71,134 showed that applying an AC voltage of 4 V and 1 Hz to an NF ECM significantly reduced scaling by both CaSO4 and CaCO3. They monitored the membrane flux with and without electricity using a CaSO4 solution. At 55% water recovery, the flux of the electrified membrane reached 17.16 ± 1.77 LMH, compared to 15.08 ± 1.97 LMH for the non-electrified membrane. This reduction in scaling was attributed to electrophoretic mixing caused by polarity switching. As shown in Fig. 5b, anions and cations move in opposite directions based on the membrane's charge, switching direction according to the frequency of the applied AC potential. As a result, the ions do not have enough residence time to co-locate and form nucleations. Besides using ECMs, Shang et al.55 demonstrated that antiscaling could be achieved with ENF using both DC and AC voltages (Fig. 5c). When a negative DC potential was applied, with the electrode below the membrane acting as the cathode, a greater reduction in flux was observed as scaling increased. This occurs due to the migration of Ca2+ ions toward the membrane, where carboxylic acid groups on the membrane surface provide interaction sites for Ca2+, facilitating pre-nucleation cluster formation. Conversely, applying a positive DC voltage moves Ca2+ away from the membrane, thereby reducing scaling. AC voltage also reduced scaling; however, beyond a certain frequency, it becomes ineffective and results in a greater flux reduction than the control. This is likely because the time available for membrane charging and cation/anion movement is insufficient, causing ions to remain in close proximity.
 | ||
| Fig. 5 (a) Effect of CaCO3 scaling on flux before and after applying electricity used with permission of Royal Society of Chemistry, from ref. 67. Copyright 2014; permission conveyed through Copyright Clearance Center, Inc. ; (b) schematic illustration of ion movement due to polarity reversal reprinted from ref. 70. Copyright 2023, with permission from Elsevier; (c) average flux decline rates due to CaSO4 scaling at different conditions reprinted from ref. 55. Copyright 2023, with permission from Elsevier; (d) effect of biofouling on flux under different conditions (no flushing, flushing, and flushing with electricity) (adapted) with permission from ref. 32. Copyright 2013 American Chemical Society. | ||
ECMs have proven highly effective in mitigating biofouling. Lannoy et al.,32 developed the first RO ECM for biofouling mitigation. The study showed that applying an alternating potential of 1.5 V to the membrane reduced flux decline (as shown in Fig. 5d). For instance, after 80 hours, the flux decline in the ECM with applied voltage was 45%, whereas the same decline occurred after just 22 hours in the ECM without electricity and after 30 hours in the non-ECM with electricity. The circles in Fig. 5d indicate instances of back-flushing, clearly showing that flushing the ECM helped recover flux, which was not the case in the other scenarios. The mechanism behind this performance was not fully explained, but the study suggested that alternating potentials could cause instabilities in the system, affecting pH and EDL, creating a less favorable environment for bacteria and disrupting biofouling formation. Additionally, a negative potential repels bacteria, while a positive potential creates oxidizing conditions, leading to bacterial inactivation. This membrane was a significant milestone in the field of high-pressure ECMs, as it was the first RO ECM reported in the literature and was later patented.33 Subsequently, the first startup, “Active Membrane,” was launched in 2021, as shown in Fig. 1c.
Feed spacers are a key component in spiral wound membrane modules, designed to enhance fluid mixing and turbulence within the feed channel.135,136 During biofouling, biofilm formation occurs not only on the membrane surface but also on the feed spacer, contributing to performance decline. Recent studies have demonstrated that incorporating electrically conductive materials into feed spacers can effectively reduce biofouling on both surfaces.137,138 For instance, Pisharody et al.137 used a laser-induced graphene-based spacer operated in resistor mode and observed significantly lower biofilm accumulation when an electric current was applied. The associated temperature increase was minimal, only 0.4–1.8 °C at 10–12 V, suggesting that the antifouling effect was not due to bacterial inactivation by ohmic heating. The authors hypothesized that the reduced biofouling was due to direct oxidation or other electric effects which were not explained or investigated. While these findings are promising, further research is needed on module design that accommodates electrically conductive spacers. As with conventional spacers, several parameters need to be investigated to optimize system performance.
3.4. Rejection/degradation of water contaminants
The treatment of contaminated saline water presents a significant global challenge due to the coexistence of salts and hazardous pollutants in these systems.139 Brine, seawater, and industrial effluents often contain dyes, phenolic compounds, heavy metals, nitrite, PFOA, and boron, which are toxic and resistant to conventional treatment methods. Traditional approaches such as adsorption, biological treatment, and chemical precipitation often fail to address the complexity of these systems due to the high ionic strength and interference from salts. RO and NF ECMs, ERO, and ENF have emerged as advanced systems offering enhanced contaminant rejection and/or degradation, as discussed in this section and summarized in Table 3.12 Two main mechanisms explain contaminant removal in these systems. Electrostatic interaction between the membrane and contaminants occurs in the non-faradaic voltage region, where no electrochemical reactions take place and charged contaminants are repelled or attracted without chemical change. Electrochemical degradation happens at higher voltages in the faradaic region, where electrochemical redox reactions on the membrane or electrode surfaces break down contaminants through oxidation or reduction.| Ref. | Pollutant | Electricity configuration | Electrode | Electrical properties of the electrode | Effective filtration area (cm2) | Counter electrode | Applied pressure (bar) | Performance without electricity | Performance with electricity | Dominant mechanism |
|---|---|---|---|---|---|---|---|---|---|---|
| a OG: orange G, MLB: methylene blue, MO: methyl orange, CR: congo red, MV: methyl violet, MG: methyl green, AF: acid fuchsin, RB: rose bengal, IC: indigo carmine, MYB: methylene blue. b Approximate values were extracted from figures. | ||||||||||
| 140 | Dye | NF ECM | rGO-MXene | Electrical conductivity: 1100 S m−1 | 8.04 | Titanium mesh | 1 | 20 mg per L OG: 56% and ∼21 LMH per bar | At −2 V: 20 mg per L OG: 91.4% and ∼23 LMH per bar | Enhanced electrostatic repulsion except for MLB electrochemical reduction |
| 20 mg per L MLB: 35.5% | 20 mg per L MLB: 97.8% | |||||||||
| 20 mg per L MO: 44.1% | 20 mg per L MO: 68.7% | |||||||||
| 20 mg per L CR: ∼57% | 20 mg per L CR: 92.5% | |||||||||
| 84 | MXene/CNT | Electrical conductivity: 236 S m−1 | 7 | Titanium mesh | 1 | 20 mg per L OG: 64.8% and ∼23.5 LMH per bar | At −3 V: 20 mg per L OG: 96.3% and ∼28 LMH per bar | Enhanced electrostatic repulsion improves rejection while permeation is improved through relieved concentration polarization and membrane fouling | ||
| 20 mg per L MO: 27.2% and ∼25 LMH per bar | 20 mg per L MO: 87.8% and ∼28 LMH per bar | |||||||||
| 79 | MXene | — | — | — | Vacuum | 20 mg per L MV: 28% and 68 LMH | At −1 V: 20 mg per L MV: 61% and 36 LMH | Enhanced electrostatic adsorption of dyes with opposite charge to the membrane accumulating in the pores causes improved rejection and reduced flux while repulsion of dyes with similar charge to the membrane facilitates dye transport through the membrane leading to degraded rejection and enhanced flux | ||
| 100 mg per L CR: 26% and 69 LMH | ||||||||||
| 100 mg per L CR: 86% and 66 LMH | At 1 V: 20 mg per L MV: 1% and 151 LMH | |||||||||
| 100 mg per L CR: 99% and 22 LMH | ||||||||||
| 75 | PA incorporating BABD-CB | Electrical conductivity: ∼0.135 S m−1 | — | — | 6 | 50 mg per L MG: ∼20% | At −20 V: 50 mg per L MG: ∼100% and ∼22 LMH per bar | Electrostatic interactions between the dye and the membrane. As an anode, negatively charged dye was connected with the holes on the membrane through van der Waals forces, as a cathode, positively charged dye combined with the electrons on the membrane surface | ||
| 50 mg per L AF: ∼15% and ∼26 LMH per bar | ||||||||||
| At 20 V: 50 mg per L MG: ∼15% and ∼26 LMH per bar | ||||||||||
| 50 mg per L AF: ∼100% and ∼22 LMH per bar | ||||||||||
| 80 | CNT/MXene-PEI | Area-specific resistance: 0.85 Ω cm−2 | 7.065 | — | 1 | 10 ppm MLB: 48% and 66 LMH per bar | At −2 V: 10 ppm MLB: 98% and ∼92 LMH per bar | Enhanced electrostatic repulsion | ||
| 10 ppm RB: ∼62% | 10 ppm RB: ∼96% | |||||||||
| 72 | ATPA | Electrical conductivity: ∼0.018 S m−1 | 12.56 | — | 8 | 50 ppm AF: 36.37% and ∼90 LMH per bar | At −50 V: 50 ppm AF: ∼3% and ∼66 LMH per bar | Structural transformation leading to changes in the surface charge | ||
| 50 ppm MG: ∼75% and ∼70 LMH per bar | ||||||||||
| 50 ppm MG: 24.67% and ∼90 LMH per bar | At 50 V: 50 ppm AF: ∼100% and 85.8 LMH per bar | |||||||||
| 50 ppm MG: ∼20% and ∼70 LMH per bar | ||||||||||
| 82 | Al3+ intercalated MXene | Electrical conductivity: ∼240 S m−1 | — | Titanium mesh | 1 | 100 ppm MO: 46.2% | At −2 V: 100 ppm MO: 90.3% | Enhanced electrostatic repulsion | ||
| 100 ppm OG: 73.2% | 100 ppm OG: 94.2% | |||||||||
| 90 | MXene-CP6 | — | — | — | 2 | 50 ppm MG: ∼55% | At −10 V: 50 ppm MG: 98% | Voltage-dependent modulation of interlayer spacing (enabled by CP6 intercalation) | ||
| 50 ppm IC: ∼75% | ||||||||||
| 50 ppm IC: ∼30% | At 10 V: 50 ppm MG: 0% | |||||||||
| 50 ppm IC: ∼0% | ||||||||||
| 73 | PA-POMs on PANI/PVDF | — | 9.62 | Titanium mesh | 4 | 1000 ppm nitrite (NaNO2) | 1000 ppm nitrite (NaNO2) | Enhanced electrostatic repulsion/electrocatalytic degradation | ||
| At −20 V: 50 ppm MO: <95% | ||||||||||
| 50 ppm MO: 0% | 50 ppm MYB: >5% | |||||||||
| At 20 V: the effect was reversed | ||||||||||
| 50 ppm MYB: 0% | Effectively degrade 82% of MO after 15 h | |||||||||
| 141 | Phenolic compounds | ECM | CPANI/CNT | Electrical conductivity: 680 S m−1 | 8.04 | Titanium mesh | 2 | 5 ppm bisphenol A: 31.9% and 6.8 LMH per bar | At 2 V: 5 ppm BPA: 98.1% and 6.8–6.2 LMH per bar | Electrochemical oxidation of BPA |
| 142 | Tannic acid and Ca(OH)2/Ti3C2Tx on PA | — | 16 | — | 6 | 1000 ppm phenol: 85.1% and ∼28 LMH per bar | At −3 V: 1000 ppm phenol: 90% and ∼37 LMH per bar | Electrostatic repulsion | ||
| 1000 ppm 4-nitrophenol: 91.1% and ∼27 LMH per bar | 1000 ppm 4-nitrophenol: ∼94% and ∼33 LMH per bar | |||||||||
| 1000 ppm BPA: 94.6% and ∼23 LMH per bar | 1000 ppm BPA: ∼97.5% and ∼30 LMH per bar | |||||||||
| 143 | Heavy metals | NF ECM | SrF2/Ti3C2Tx nanoparticles on PA/PVDF | — | 16 | Copper mesh | 6 | Mn2+: 93.6% | At 2 V: Mn2+: 98.8% | Photo electrocatalysis (PEC). The generation of electrons, holes, superoxide radicals, and hydroxyl radicals during PEC enhances the rejection of heavy metal ions |
| Ni2+: 90.5% | Ni2+: 95.3% | |||||||||
| Cu2+: 90.7% | Cu2+: 95.7% | |||||||||
| Zn2+: 92.2% | Zn2+: 97.3% | |||||||||
| Permeance of Mn2+ = 21.35–22.01 LMH per bar | Permeance of Mn2+ = 28.32–28.78 LMH per bar | |||||||||
| 144 | ENF | Titanium sheet | NA | 3.14 | Titanium sheet | 6 | 1000 ppm Na2WO4: ∼95% | 1000 ppm Na2WO4: ∼97% | Electro-deionization regenerating the ion exchange layer by water electrolysis and enhances the removal of both heavy metal cations and oxyanions | |
| 1000 ppm Na2CrO4: ∼83% | 1000 ppm Na2CrO4: ∼90% | |||||||||
| 1000 ppm NiCl2: ∼84% | 1000 ppm NiCl2: ∼96% | |||||||||
| 1000 ppm CdCl2: ∼78% | 1000 ppm CdCl2: ∼93% | |||||||||
| 145 | Nitrate | NF ECM | MXene-Cu on nylon membrane | — | 7.065 (calculated from the reported membrane diameter of ∼3 cm) | Platinum sheet | NA | — | At −1.3 V: 30 ppm nitrate in 100 ml of 1 mM Na2SO4: 85% nitrate conversion to NH3 in 1 h at 300 LMH using closed-cycle flow-through (CFT) | Electrocatalytic reduction of nitrate to ammonia via Cu catalyst |
| 73 | Nitrite | NF ECM | PA-POMs on PANI/PVDF | — | 9.62 | Titanium mesh | 4 | 1000 ppm nitrite (NaNO2): nitrite removal is 23.1% | At −20 V: 1000 ppm nitrite (NaNO2): nitrite removal is 81% | Enhanced Donnan effect and electrocatalytic reduction at the cathode (nitrite to nitrogen) and oxidation at the anode (nitrite to nitrate) |
| 61 | PFOA | ENF | Porous titanium disc | NA | — | Porous titanium disc coated with SnO2–Sb | 4.14 | 0.5 ppm PFOA and non-saline: 45% | At −30 V: 0.5 ppm PFOA and non-saline: 97% and 90% fluoride recovery after 48 h | Electro-oxidation of PFOA into fluoride and PFCAs |
| 87 | Boron | RO ECM | CNT and PVA coating on commercial RO membrane | Sheet resistance: 61.63 ± 7.49 Ω/□ | 40 | Titanium wire coated with platinum | 34.47 | 35![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 ppm NaCl with 5 ppm boron (pH 6.55): 63.99% 000 ppm NaCl with 5 ppm boron (pH 6.55): 63.99% |
At −5 V (pH = 10.83): 35![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 ppm NaCl with 5 ppm boron 90.05% 000 ppm NaCl with 5 ppm boron 90.05% |
Elevated pH at the membrane surface through electrochemical reactions transforms boric acid to borate |
| 62 | ERO | Carbon cloth placed on the surface of a commercial RO membrane | NA | 33.34 | Platinum plate | 41.4 | 35![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 ppm NaCl with 5 ppm boron: ∼75% 000 ppm NaCl with 5 ppm boron: ∼75% |
At −4 V (pH ∼8): 35![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 ppm NaCl with 5 ppm boron: ∼93.8% 000 ppm NaCl with 5 ppm boron: ∼93.8% |
Elevated pH near the membrane surface through electrochemical reactions transforms boric acid to borate | |
| 69 | Chlorine | RO ECM | PA/CNT on polysulfone | Electrical conductivity: 2839 ± 129.9 S m−1 | 40 | Platinum coated plate | 10 | Exposure to 1000 ppm NaOCl: 3.5% chlorine uptake (at 1 h exposure) and 9.3% chlorine uptake (at 24 h exposure) | Exposure to 1000 ppm NaOCl: at −2 V: not detected chlorine uptake (at 1 h exposure) and 3.8% chlorine uptake (at 24 h exposure) | Electrochemical reduction of chlorine to chloride on membrane surface |
| 2000 ppm NaCl: NaCl rejection 74% after 24 h chlorination | 2000 ppm NaCl: NaCl rejection 88% after 24 h chlorination | |||||||||
| 63 | ERO | Woven stainless-steel mesh | NA | 33.15 | Titanium mixed metal oxide plate coated with ruthenium and iridium | 50 | — | Chlorine content in the brine: 0.05 ppm at −1.5 V | Electrochemical oxidation of chloride to free chlorine at anode for biofouling control | |
| 0.125 ppm at −2.1 V | ||||||||||
Beyond enhanced rejection, applying electricity can also support in situ degradation of dyes, yielding a retentate stream with reduced concentration of pollutant. For example, Wang et al.140 reported that Methylene Blue (MLB) exhibited improved rejection from 35.5% to 97.8% as the voltage increased from 0 to −2.0 V which was attributed to the electrochemical reduction of MLB. The enhanced removal of dye could be associated with an increase in water flux thus breaking the selectivity permeability trade-off. In a recent study on MXene-based membranes, rejection of MLB under −2 V increased from 48.63% to 97.92%, while water flux improved from 66 to 92 LMH per bar.80 The flux enhancement with electricity was linked to electrophoretic forces, which minimized dye accumulation on the membrane surface and kept the transport channels open. While most studies emphasize electrostatic repulsion as the mechanism behind the improved dye removal, Li et al.79 reported the opposite trend. When the membrane and dye had opposite charges, electrostatic attraction led to dye intercalation within the lamellar channels, forming a barrier that increased dye rejection but reduced water flux. Conversely, when the membrane and dye shared the same charge, electrostatic repulsion prevented dye accumulation, leading to lower rejection and higher water flux. In this section, we have discussed several mechanisms that contribute to improved dye rejection using NF ECMs. A key takeaway from these studies is that the governing mechanism is not universal; rather, it is determined by the membrane (materials and properties) and its interaction with the contaminant when electricity is applied.
3.5. Pore tuning and structural changes
Controlling membrane microstructure, including pore tuning and surface topography, is a key goal in membrane manufacturing. Once conventional membranes are formed, their pore size and surface properties are usually fixed. However, intense electric fields can be used as a tool to modify the properties and performance of pre-formed membranes. Darestani et al.29,159 demonstrated that an intense electric field can be implemented to alter the microstructure and separation performance of pre-formed NF and RO membranes. The membranes were sandwiched between two electrodes and subjected to intense DC voltage while being heated to a temperature between the glass transition and melting points of the polymer, as polymer chain movement is only possible above the glass transition temperature. In this approach, the electric field was applied before membrane testing, not during filtration experiments. Under the right treatment time and voltage, NF and RO membranes showed improved rejection of polyethylene glycol (PEG) with a molecular weight of 10 kDa, and salts (100 mM NaCl and 50 mM MgCl2), respectively, while the flux decline was minimal. The authors attributed these changes to a smoother membrane surface and a tightened microstructure after electric field application. Although they presented this method as a potential way to modify membrane microstructure, the reasons behind these changes remain unclear, and using an intense electric field is energy-intensive and impractical on a large scale. As an alternative, electrically responsive membranes, whose filtration properties can be tuned during operation, offer a more practical solution. These membranes help overcome some of the limitations associated with fixed membrane properties, such as fouling and restricted applications.160 A prominent example is PANI, the most extensively studied intrinsically conducting polymer. Several studies have shown that applying voltage to PANI membranes results in reduced permeate flux and enhanced rejection of various molecular weights of PEG.64,161,162 These changes in filtration performance were attributed to the narrowed pore size due to polymer chain swelling, which occurs as acid dopants rearrange in the polymer structure under applied voltage (illustrated in Fig. 6a).64 Further studies confirmed that the type of acid dopant affects membrane tunability, as different dopants led to varied changes in filtration performance under electricity.161 Following a similar mechanism, Zhu et al.70 revealed that the pore size of an PA NF ECM can be tuned using electricity. This study marked a significant milestone in pore size regulation, achieving angstrom-level control, which is essential for precise solute separation. When placed in an electrolyte solution, weakly charged PA membranes deswell and contract to reduce effective pore size under the pore flow model. In contrast, when the membrane is strongly charged, the counterion concentration within the polymer network exceeds that in the bulk solution, creating an osmotic pressure difference and causing membrane swelling163 (Fig. 6b, left). Based on this theory, applying external voltage induces swelling, leading to a larger pore size (Fig. 6b, right). Changes in membrane volume leading to modulated pore size can also be induced by redox-driven ion insertion and expulsion, as demonstrated in a recent study using polypyrrole (PPy).164 Under a reducing potential, cations were inserted into the polymer network, causing it to swell and contract the pores; under an oxidizing potential, the cations were expelled, leading to polymer shrinkage and pore expansion. To further enhance tunability, hydroxyl functional groups were introduced to strengthen electrostatic and van der Waals interactions within the nanochannels. This design enabled reversible and dynamic tuning of membrane selectivity and water flux during operation. | ||
| Fig. 6 Schematic illustration of (a) asymmetric PANI membrane and effect of voltage on membrane swelling degree reprinted from ref. 63. Copyright 2018, with permission from Elsevier, (b) swelling behavior of PA based on the charge adapted with permission from ref. 69. Copyright 2023 Springer Nature, and (c) structural expansion in the channel size of rGO membrane due to electricity (adapted) with permission from ref. 82. Copyright 2023 American Chemical Society; (d) reversible changes in performance of rGO intercalated PPy membrane when applied voltage switches between 0.5 V and −2.5 V (the feed solution was 2 g per L Na2SO4 solution) reprinted from ref. 162. Copyright 2023, with permission from Elsevier. | ||
In addition to polymeric membranes, 2D lamellar membranes, such as GO, have emerged for smart gating applications. For instance, Wei et al.83 developed an rGO membrane in which the channel size could be electrochemically tuned by applying a positive potential (oxidation) to the membrane. Freshwater flux was found to double after electrochemical treatment, indicating that the structural expansion resulted from water molecule intercalation into the restacked regions, driven by hydrogen bond interactions with hydroxyl groups electrochemically produced at the edges of rGO nanosheets (as shown in Fig. 6c). While this study showed promising results for electrochemically expanding rGO channels, the change was applied outside of the filtration setup, and no discussion of reversibility was provided. It is possible that the changes in the membrane structure were irreversible, which would limit the application of such membranes. Similarly, in a recent study on rGO membranes intercalated with AT, the smallest repeating unit of PANI, it was found that applying a cathodic potential resulted in an increase in interlayer spacing, due to the electrochemical adsorption of counterions by AT.165 However, the authors did not evaluate whether the expanded interlayer spacing returned to its original state after testing or during cycles of voltage on/off, leaving the reversibility of the structural change unconfirmed. In contrast, Wu et al.166 demonstrated that the pore size of an rGO membrane intercalated with PPy can be reversibly regulated under applied voltage during the filtration process (Fig. 6d). In this case, the change in pore size was due to volume changes in PPy caused by the insertion and removal of cations (in this case, Na+) when oxidation and reduction potentials were applied, respectively. While electrically modulating membrane pore size is a powerful tool for controlling separation performance, evaluating the reversibility of the membrane structural changes is critical for practical applicability.
4. Conclusions and recommendations
Electrically assisted NF and RO systems have attracted growing attention as a means to enhance membrane performance, and expand the functionality of conventional membrane processes. This review examined the current state of knowledge on fabrication strategies, performance outcomes, application areas, and mechanisms of these systems. The points below summarize the key conclusions, identify research gaps, and provide recommendations to guide future research to support the advancements of these systems:• ERO and ENF systems offer promising alternatives to ECMs through recent advances in electrode design, including dual-function components that act as both spacers and electrodes. However, current studies are limited to lab-scale experiments using simple flat-sheet designs. Therefore, further research is needed on membrane module design, specifically how to accommodate electrodes, manage their connections, and ensure durability during operation.
• Several fabrication methods for NF and RO ECMs have been reported in the literature, but all have limitations. Interfacial polymerization appears to be the most promising approach, as it is commercially established and has been shown to produce ECMs with performance comparable to commercial membranes. Therefore, future research should focus on improving the fabrication of RO and NF ECMs via interfacial polymerization, with an emphasis on enhancing electrical conductivity, membrane performance, and scalability.
• Many studies on NF and RO ECMs refer to their membranes as electrically conductive, even when they either do not measure electrical conductivity or sheet resistance, or report values that are low compared to other studies in the literature. As a result, the mechanisms described in these studies, if based on the assumption of electrical conductivity, may not be accurate. It is therefore essential to establish a minimum value that defines what qualifies as an ECM. Moreover, the electrical and electrochemical properties of membranes should be thoroughly characterized and clearly reported.
• The specific ECM properties that should be optimized to enhance responsiveness to electrical assistance remain unclear. For instance, how does electrical conductivity affect the membrane's response to electricity? Further research is needed to understand the fundamental effects of membrane characteristics, such as electrical conductivity, specific capacitance, and pore structure on performance with electricity. In addition, studies should explore how these properties interact and collectively impact membrane performance.
• ECMs have shown promising results for enhanced salt rejection, particularly in cases where increased rejection is accompanied by improved water permeability. However, despite these advancements, most of these membranes still fall short of achieving salt rejection levels comparable to commercial NF membranes when electricity is not applied. For NF ECMs to demonstrate a comparative advantage, their performance without electricity should be at least comparable to that of commercial NF membranes, allowing for further enhancement when electricity is applied.
• There are only a few studies exploring the use of electric fields for ion separation. One notable example is the use of membranes to separate Mg2+ from Li+, presenting a potential method for Li+ recovery. However, it remains unclear how this approach compares with currently proposed methods for Li+ recovery in terms of feasibility or effectiveness.
• In applications where voltage is used to induce changes in membrane pore size, one critical aspect often overlooked is the reversibility of this effect. Although the ability to alter membrane structure and pore size using electricity is a powerful tool, it is not useful if the changes are irreversible. Therefore, testing for reversibility is a critical aspect that must be included in such studies.
• In the context of fouling mitigation, many studies lack critical analysis or supporting data to evaluate the effectiveness of ERO and ENF systems. A key issue is that membrane performance is usually compared between conditions with and without electricity, without considering comparisons against conventional membrane cleaning methods. While it is evident that membranes without electricity will suffer from fouling, the real question is whether electrically-assisted fouling mitigation is superior to traditional methods. Furthermore, most tests using electrically-assisted systems are conducted with synthetic solutions rather than real water samples. While synthetic solutions are useful for understanding mechanisms, they are insufficient for evaluating the true effectiveness of electricity in fouling reduction or membrane cleaning. Additionally, the use of various foulants in antifouling studies creates difficulties in comparing membranes developed by different research groups or assessing different methods of applying electricity. To address this, consistent use of the same model foulants, as suggested in a previous review,31 is recommended.
• Regarding contaminant removal, electrically assisted systems that only extract contaminants generate a stream concentrated with those contaminants, creating an additional challenge. Therefore, it is more effective to focus on degrading the contaminants. In electrically assisted systems for contaminant degradation, it is crucial to minimize the formation of toxic and undesirable byproducts while achieving high faradaic efficiency by preventing undesirable faradaic reactions at the counter electrode. However, the practicality of employing such systems, particularly those requiring relatively high pressure, remains debatable. For instance, UF membranes have demonstrated the capability to achieve complete removal and degradation of dyes without generating toxic byproducts, offering a fast and reliable solution for water treatment which could be employed in a portable system due to their low-pressure requirements. In comparison, the viability of NF systems for similar applications is questionable as it requires high pressure for operating. These systems could prove their viability if they were used in alternative applications, such as the recovery of valuable ions, the conversion or transport of nitrate into ammonia, or the transformation of pollutants in waste streams into value-added organic products.
• A significant concern with ECMs is their stability under applied electricity, which is often not adequately examined. Most studies lack sufficient data on long-term stability testing and the electrochemical properties of these membranes. To better understand membrane durability, we recommend conducting long-term tests under applied electricity for at least 24 hours at the lab scale to assess structural and chemical stability over time. In addition, cyclic voltage testing (cycling between 0 V and the optimized operating voltage) should be performed to confirm that applying electricity does not cause permanent changes to the membrane's chemical or physical structure and that any observed changes are reversible. Electrochemical characterization is also essential, particularly for membranes operated at voltages where electrochemical reactions may occur such as those used for contaminant degradation.
• The economic viability of electrically assisted systems compared to conventional methods is still overlooked. Techno-economic evaluations, including feasibility studies, are necessary to determine acceptable membrane material costs, electrode costs, and additional energy consumption. Large-scale or pilot-scale testing is crucial to assess the practicality of these systems. Key considerations include whether these technologies can extend membrane lifespan and justify the additional costs. Testing with real water samples is also vital for a more accurate assessment of their potential. Moreover, the design of suitable configurations and modules for electrically assisted systems has been repeatedly discussed and highlighted by researchers,34,54 yet no concrete actions have been taken to address this issue.
Author contributions
Haya Nassrullah: writing – original draft, writing – review & editing, conceptualization, visualization. Jamaliah Aburabie: writing – original draft, visualization. Shabin Mohammed: writing – original draft. Nidal Hilal: supervision. Raed Hashaikeh: supervision, writing – review & editing, funding acquisition, conceptualization.Conflicts of interest
There are no conflicts to declare.Data availability
No primary research results, software or code have been included and no new data were generated or analysed as part of this review.Acknowledgements
The authors would like to thank Tamkeen for its financial support (project CG007) under the NYUAD Research Institute Award.References
- M. M. Mekonnen and A. Y. Hoekstra, Sci. Adv., 2016, 2, e1500323 CrossRef PubMed.
- M. Kummu, J. H. A. Guillaume, H. de Moel, S. Eisner, M. Flörke, M. Porkka, S. Siebert, T. I. E. Veldkamp and P. J. Ward, Sci. Rep., 2016, 6, 38495 CrossRef CAS PubMed.
- B. D. Richter, D. Abell, E. Bacha, K. Brauman, S. Calos, A. Cohn, C. Disla, S. F. O'Brien, D. Hodges, S. Kaiser, M. Loughran, C. Mestre, M. Reardon and E. Siegfried, Water Policy, 2013, 15, 335–363 CrossRef.
- C. J. Vörösmarty, P. Green, J. Salisbury and R. B. Lammers, Science, 2000, 289, 284–288 CrossRef PubMed.
- P. Greve, T. Kahil, J. Mochizuki, T. Schinko, Y. Satoh, P. Burek, G. Fischer, S. Tramberend, R. Burtscher, S. Langan and Y. Wada, Nat. Sustain., 2018, 1, 486–494 CrossRef.
- United Arab Emirates, Ministry of Foreign Affairs, in Ripple Effect: Water Scarcity - The Hidden Threat to Global Security and Prosperity, 2023 Search PubMed.
- H. Nassrullah, S. F. Anis, R. Hashaikeh and N. Hilal, Desalination, 2020, 491, 114569 CrossRef CAS.
- K. K. Kesari, R. Soni, Q. M. S. Jamal, P. Tripathi, J. A. Lal, N. K. Jha, M. H. Siddiqui, P. Kumar, V. Tripathi and J. Ruokolainen, Water, Air, Soil Pollut., 2021, 232, 208 CrossRef CAS.
- Reverse Osmosis and Nanofiltration Membranes Market Size, Growth Opportunities, https://www.linkedin.com/pulse/reverse-osmosis-nanofiltration-membranes-market-cpzqc, accessed July 29, 2024.
- J. Aburabie, H. Nassrullah and R. Hashaikeh, in Nanofiltration for Sustainability, CRC Press, 2023 Search PubMed.
- A. Iris Schäefer, in Nanofiltration: Principles, Applications, and New Materials, John Wiley & Sons, Ltd, 2021, pp. xxiii–xxx Search PubMed.
- M. Sun, X. Wang, L. R. Winter, Y. Zhao, W. Ma, T. Hedtke, J.-H. Kim and M. Elimelech, ACS EST Eng., 2021, 1, 725–752 CrossRef CAS.
- B. M, in Electrophoresis, Theory, Methods and Applicationss, Academic Press, New York, 1959, p. 263 Search PubMed.
- F. C. Cooper, Q. M. Mees and M. Bier, J. Sanit. Eng. Div., 1965, 91, 13–26 CrossRef CAS.
- S. P. Moulik, F. C. Cooper and M. Bier, J. Colloid Interface Sci., 1967, 24, 427–432 CrossRef CAS.
- M. Bier, in Membrane Processes in Industry and Biomedicine: Proceedings of a Symposium Held at the 160th National Meeting of the American Chemical Society, under the Sponsorship of the Division of Industrial and Engineering Chemistry, Chicago, Illinois, September 16 and 17, 1970, ed. M. Bier, Springer US, Boston, MA, 1971, pp. 233–266 Search PubMed.
- J. C. Giddings, Sep. Sci., 1966, 1, 123–125 Search PubMed.
- E. Grushka, K. D. Caldwell, M. N. Myers and J. C. Giddings, Sep. Purif. Methods, 1973, 2, 127–151 CrossRef CAS.
- J. D. Henry Jr, L. F. Lawler and C. H. A. Kuo, AIChE J., 1977, 23, 851–859 CrossRef.
- G. Akay and R. J. Wakeman, J. Membr. Sci., 1997, 131, 229–236 CrossRef CAS.
- R. J. Wakeman, Food Bioprod. Process., 1998, 76, 53–59 CrossRef CAS.
- C. Visvanathan and R. B. Aim, Sep. Sci. Technol., 1989, 24, 383–398 CrossRef CAS.
- N. Mameri, S. Oussedik, R. Yeddou, D. L. Piron, D. Belhocine, H. Lounici and H. Grib, Sep. Purif. Technol., 1999, 17, 203–211 CrossRef CAS.
- W. R. Bowen and H. A. M. Sabuni, Ind. Eng. Chem. Res., 1991, 30, 1573–1579 CrossRef CAS.
- H. Yukawa, K. Shimura, A. Suda and A. Maniwa, J. Chem. Eng. Jpn., 1983, 16, 305–311 CrossRef CAS.
- G. M. Rios, H. Rakotoarisoa and B. Tarodo de la Fuente, J. Membr. Sci., 1988, 38, 147–159 CrossRef CAS.
- C. Mullon, J. M. Radovich and B. Behnam, Sep. Sci. Technol., 1985, 20, 63–72 CrossRef CAS.
- L. Pupunat, G. M. Rios, R. Joulié and M. Persin, Sep. Sci. Technol., 1998, 33, 67–81 CrossRef CAS.
- M. T. Darestani, T. C. Chilcott and H. G. L. Coster, J. Membr. Sci., 2014, 449, 158–168 CrossRef CAS.
- K. B. WAGENER, in Materials Science of Synthetic Membranes, American Chemical Society, 1985, vol. 269, pp. 429–445 Search PubMed.
- M. J. Larocque, A. Gelb, D. R. Latulippe and C.-F. de Lannoy, Sep. Purif. Technol., 2022, 287, 120482 CrossRef CAS.
- C.-F. de Lannoy, D. Jassby, K. Gloe, A. D. Gordon and M. R. Wiesner, Environ. Sci. Technol., 2013, 47, 2760–2768 CrossRef CAS PubMed.
- University of California and Duke University, US Pat., US9802163B2, 2017 Search PubMed.
- X. Zhu and D. Jassby, Acc. Chem. Res., 2019, 52, 1177–1186 CrossRef CAS PubMed.
- N. H. Barbhuiya, U. Misra and S. P. Singh, Environ. Sci.:Water Res. Technol., 2021, 7, 671–705 RSC.
- C. Li, X. Guo, X. Wang, S. Fan, Q. Zhou, H. Shao, W. Hu, C. Li, L. Tong, R. R. Kumar and J. Huang, Electrochim. Acta, 2018, 287, 124–134 CrossRef CAS.
- Y. Shen and A. R. Badireddy, Membranes, 2021, 11, 820 CrossRef CAS PubMed.
- A. B. Alayande, K. Goh, M. Son, C.-M. Kim, K.-J. Chae, Y. Kang, J. Jang, I. S. Kim and E. Yang, Membranes, 2021, 11, 5 CrossRef CAS PubMed.
- X. Fan, G. Wei and X. Quan, Environ. Sci.: Nano, 2023, 10, 11–40 RSC.
- Y. Liu, F. Liu, N. Ding, X. Hu, C. Shen, F. Li, M. Huang, Z. Wang, W. Sand and C.-C. Wang, Chin. Chem. Lett., 2020, 31, 2539–2548 CrossRef CAS.
- J. J. Patil, A. Jana, B. A. Getachew, D. S. Bergsman, Z. Gariepy, B. D. Smith, Z. Lu and J. C. Grossman, J. Mater. Chem. A, 2021, 9, 3270–3289 RSC.
- Z. Lin, L. Liu, C. Zhang, P. Su, X. Zhang, X. Li and Y. Jiao, Chem. Eng. J., 2024, 493, 152474 CrossRef CAS.
- D. Kong, Y. Zhao, R. Wang, J. Li, J. Li and J. Ma, ACS EST Eng., 2023, 3, 2123–2146 CrossRef CAS.
- Z. Zhang, G. Huang, Y. Li, X. Chen, Y. Yao, S. Ren, M. Li, Y. Wu and C. An, Chem. Eng. J., 2022, 427, 131987 CrossRef CAS.
- J. Hao, L. Wang, X. Wang, J. Wang, M. He, X. Zhang, J. Wang, L. Nie and J. Li, Environ. Sci.:Water Res. Technol., 2023, 10, 105–127 RSC.
- F. Ahmed, B. S. Lalia, V. Kochkodan, N. Hilal and R. Hashaikeh, Desalination, 2016, 391, 1–15 CrossRef CAS.
- Y. Mo, L. Zhang, X. Zhao, J. Li and L. Wang, J. Hazard. Mater., 2022, 436, 129162 CrossRef CAS PubMed.
- J. Kim, J. Lee, S. Lee, L. Tijing, H. K. Shon and S. Hong, Desalination, 2024, 578, 117445 CrossRef CAS.
- P. Formoso, E. Pantuso, G. De Filpo and F. P. Nicoletta, Membranes, 2017, 7, 39 CrossRef PubMed.
- G. Ren, R. Li, M. Zhao, Q. Hou, T. Rao, M. Zhou and X. Ma, Chem. Eng. J., 2023, 451, 138907 CrossRef CAS.
- C. Trellu, B. P. Chaplin, C. Coetsier, R. Esmilaire, S. Cerneaux, C. Causserand and M. Cretin, Chemosphere, 2018, 208, 159–175 CrossRef CAS PubMed.
- Z. Pan, C. Song, L. Li, H. Wang, Y. Pan, C. Wang, J. Li, T. Wang and X. Feng, Chem. Eng. J., 2019, 376, 120909 CrossRef CAS.
- Y. Qi, D. Li, S. Zhang, F. Li and T. Hua, J. Environ. Sci., 2024, 141, 102–128 CrossRef CAS PubMed.
- J. Sun, C. Hu, B. Wu, H. Liu and J. Qu, J. Membr. Sci., 2020, 604, 118077 CrossRef CAS.
- C. Shang, T. Zhang, J. Y. Lee and S. Zhang, J. Membr. Sci., 2023, 675, 121549 CrossRef CAS.
- H. Zhang, X. Quan, S. Chen, H. Yu and J. Niu, Environ. Sci. Technol., 2020, 54, 15433–15441 CrossRef CAS PubMed.
- Q. Li, H. Liu, Y. Ji, Z. Cui, F. Yan, M. Younas, J. Li and B. He, Desalination, 2022, 535, 115825 CrossRef CAS.
- Q. Li, Y. Liu, Y. Liu, Y. Ji, Z. Cui, F. Yan, J. Li, M. Younas and B. He, J. Membr. Sci., 2022, 662, 120982 CrossRef CAS.
- Q. Li, H. Liu, B. He, W. Shi, Y. Ji, Z. Cui, F. Yan, Y. Mohammad and J. Li, J. Membr. Sci., 2022, 641, 119880 CrossRef CAS.
- Chemnor Ag, US Pat., US3632498A, 1972 Search PubMed.
- Y. Ji, Y. J. Choi, Y. Fang, H. S. Pham, A. T. Nou, L. S. Lee, J. Niu and D. M. Warsinger, Environ. Sci. Technol., 2023, 57, 18519–18528 CrossRef CAS PubMed.
- X. Bao, W. Long, H. Liu and Q. She, J. Membr. Sci., 2021, 637, 119639 CrossRef CAS.
- W. Long, J. W. Koo, Z. Yuan and Q. She, Water Res., 2024, 249, 120982 CrossRef CAS PubMed.
- L. Xu, S. Shahid, A. K. Holda, E. A. C. Emanuelsson and D. A. Patterson, J. Membr. Sci., 2018, 552, 153–166 CrossRef CAS.
- Y. Liu, Q. Li, Y. Ji, M. Younas and B. He, Sep. Purif. Technol., 2024, 348, 127531 CrossRef CAS.
- J. Aburabie, H. Nassrullah and R. Hashaikeh, Chemosphere, 2023, 310, 136907 CrossRef CAS PubMed.
- H. Nassrullah, J. Aburabie, N. Hilal and R. Hashaikeh, J. Water Proc. Eng., 2023, 56, 104405 CrossRef.
- W. Duan, A. Dudchenko, E. Mende, C. Flyer, X. Zhu and D. Jassby, Environ. Sci.: Processes Impacts, 2014, 16, 1300–1308 RSC.
- N. K. Khanzada, D. Jassby and A. K. An, Chem. Eng. J., 2022, 435, 134858 CrossRef CAS.
- Y. Zhu, L. Gui, R. Wang, Y. Wang, W. Fang, M. Elimelech, S. Lin and J. Jin, Nat. Commun., 2023, 14, 2373 CrossRef CAS PubMed.
- B. Jung, S. Ma, C. Miang Khor, N. Khalid Khanzada, A. Anvari, X. Wang, S. Im, J. Wu, U. Rao, A. Kyoungjin An, E. M. V. Hoek and D. Jassby, Chem. Eng. J., 2023, 452, 139216 CrossRef CAS.
- L. Xiao, J. Bai, X. Chen, W. Lai, L. Gong, H. Liu, L. Shan and S. Luo, Ind. Eng. Chem. Res., 2024, 63, 9151–9161 CrossRef CAS.
- M.-M. Zhang, A.-K. Li, M.-J. Tang, Q.-Y. He, Y.-H. Peng, R.-J. Fan, S.-P. Sun and X.-L. Cao, J. Membr. Sci., 2024, 699, 122668 CrossRef CAS.
- T.-Z. Jia, R. Feng, C. Cui, Q. Chen, L. Cseri, R.-F. Zhou, G. Szekely, X.-L. Cao and S.-P. Sun, Water Res., 2024, 252, 121251 CrossRef CAS PubMed.
- Y. Wang, R.-Z. Liang, T.-Z. Jia, X.-L. Cao, Q. Wang, J.-R. Cao, S. Li, Q. Shi, L. Isaacs and S.-P. Sun, J. Am. Chem. Soc., 2022, 144, 6483–6492 CrossRef CAS PubMed.
- J. Chen, X. Wang, Y. Li, R. Dai and Z. Wang, Environ. Sci.:Water Res. Technol., 2022, 8, 2652–2662 RSC.
- L. Xu, S. Liu, Z. Zheng, L. Yu, K. Li, J. Wang and J. Wang, Sep. Purif. Technol., 2023, 308, 122910 CrossRef CAS.
- L. Xu, S. Liu, L. Yu, K. Li, Y. Zhang, J. Wang and J. Wang, J. Membr. Sci., 2022, 663, 121046 CrossRef CAS.
- J. Li, C. Xu, J. Long, Z. Ding, R. Yuan and Z. Li, ACS Appl. Nano Mater., 2022, 5, 7373–7381 CrossRef CAS.
- X. Fang, M. Shi, Z. Zhang, X. Zhang, X. Yan, F. Li, S. Jiang and Y. Liu, Sep. Purif. Technol., 2024, 339, 126753 CrossRef CAS.
- Z. Zandi, M. Rastgar, M. Mohseni, M. D. Firouzjaei, W. Dilokekunakul, B. Anasori, C. D. Vecitis, R. Keller, M. Wessling, M. Elliott, A. Rahimpour and M. Sadrzadeh, Adv. Funct. Mater., 2024, 34, 2401970 CrossRef CAS.
- C. Yuan, Y. Li, L. Qian, J. Xing, X. Wang, H. Zhang, L. Du, G. Wei, S. Chen and X. Quan, ACS EST Eng., 2023, 3, 2233–2242 CrossRef CAS.
- G. Wei, L. Du, H. Zhang, J. Xing, S. Chen and X. Quan, Environ. Sci. Technol., 2023, 57, 3843–3852 CrossRef CAS PubMed.
- G. Yi, L. Du, G. Wei, H. Zhang, H. Yu, X. Quan and S. Chen, J. Membr. Sci., 2022, 658, 120719 CrossRef CAS.
- C. Hu, Z. Liu, X. Lu, J. Sun, H. Liu and J. Qu, J. Mater. Chem. A, 2018, 6, 4737–4745 RSC.
- S. F. Anis, B. S. Lalia, R. Hashaikeh and N. Hilal, Sep. Purif. Technol., 2022, 303, 122228 CrossRef CAS.
- B. Jung, C. Y. Kim, S. Jiao, U. Rao, A. V. Dudchenko, J. Tester and D. Jassby, Desalination, 2020, 476, 114212 CrossRef CAS.
- W. Zhang, N. G. P. Chew and O. Coronell, Environ. Sci. Technol. Lett., 2023, 10, 1135–1141 CrossRef CAS PubMed.
- Z. Ma, P. Liu, W. Chen, C. Wang, Y. Qiu, L.-F. Ren and J. Shao, Sep. Purif. Technol., 2025, 360, 131101 CrossRef CAS.
- X. Ding, M. Nie, S. Cheng, R. Li, Y. Wang, A. S. Ali, X. Han and Y. Sun, Sep. Purif. Technol., 2025, 365, 132667 CrossRef CAS.
- Z. Zhang, Z. Zhou, S. Feng, L. Wu and S. Gao, Desalination, 2025, 601, 118582 CrossRef CAS.
- H. Nassrullah, S. Aristizabal, J. Aburabie, N. Hilal and R. Hashaikeh, npj Clean Water, 2025, 8, 44 CrossRef CAS.
- H. Zhang, X. Quan, X. Fan, G. Yi, S. Chen, H. Yu and Y. Chen, Environ. Sci. Technol., 2019, 53, 868–877 CrossRef CAS PubMed.
- K. P. Lee, T. C. Arnot and D. Mattia, J. Membr. Sci., 2011, 370, 1–22 CrossRef CAS.
- M. Pontié, J. S. Derauw, S. Plantier, L. Edouard and L. Bailly, Desalin. Water Treat., 2013, 51, 485–494 CrossRef.
- I. M. Noël, R. E. Lebrun and C. R. Bouchard, Desalination, 2003, 155, 243–254 CrossRef.
- I. M. Noël, R. E. Lebrun and C. R. Bouchard, Desalination, 2003, 155, 229–242 CrossRef.
- R. Han, X. Ma and Y. Dai, Desalin. Water Treat., 2017, 87, 76–81 CrossRef CAS.
- R. Han, S. Zhang and X. Jian, Desalin. Water Treat., 2016, 57, 28531–28536 CrossRef CAS.
- A. E. Yaroshchuk, Membr. Technol., 1998, 1998, 9–12 Search PubMed.
- J. Xing, H. Zhang, G. Wei, L. Du, S. Chen, H. Yu and X. Quan, Environ. Sci. Technol., 2023, 57, 615–625 CrossRef CAS PubMed.
- C. Cheng, G. Jiang, G. P. Simon, J. Z. Liu and D. Li, Nat. Nanotechnol., 2018, 13, 685–690 CrossRef CAS PubMed.
- G. Jiang, C. Cheng, D. Li and J. Z. Liu, Nano Res., 2016, 9, 174–186 CrossRef CAS.
- H. B. Park, J. Kamcev, L. M. Robeson, M. Elimelech and B. D. Freeman, Science, 2017, 356, eaab0530 CrossRef PubMed.
- H.-G. Zhang, X. Quan, L. Du, G.-L. Wei, S. Chen, H.-T. Yu and Y.-C. Dong, Proc. Natl. Acad. Sci. U. S. A., 2023, 120, e2219098120 CrossRef CAS.
- H. Zheng, X. Meng, Y. Yang, J. Chen and S. Huo, Chem. Eng. Res. Des., 2023, 193, 738–750 CrossRef CAS.
- F. Guo, J. Miao, L. Xu, Q. Zhou and T. Deng, Desalination, 2023, 555, 116526 CrossRef CAS.
- A. Loreti Hupsel, C. P. Borges, F. V. da Fonseca and G. M. Barbosa, Water Sci. Technol., 2024, 89, 454–469 CrossRef PubMed.
- Q. Wang, Y. Wang, B.-Z. Chen, T.-D. Lu, H.-L. Wu, Y.-Q. Fan, W. Xing and S.-P. Sun, Ind. Eng. Chem. Res., 2019, 58, 12280–12290 CrossRef CAS.
- L. Pupunat, G. M. Rios and R. Joulié, Sep. Sci. Technol., 1999, 34, 1947–1962 CrossRef CAS.
- H.-Z. Zhang, Z.-L. Xu, H. Ding and Y.-J. Tang, Desalination, 2017, 420, 158–166 CrossRef CAS.
- X. Li, C. Zhang, S. Zhang, J. Li, B. He and Z. Cui, Desalination, 2015, 369, 26–36 CrossRef CAS.
- H. Liu, Q. Li, B. He, Z. Sun, F. Yan, Z. Cui and J. Li, Membranes, 2022, 12, 57 CrossRef CAS.
- J. Lv, Y. Wang, B. Hui, Y. Ji, H. Wang, M. Younas and B. He, J. Membr. Sci., 2025, 714, 123413 CrossRef CAS.
- A. Matin, T. Laoui, W. Falath and M. Farooque, Sci. Total Environ., 2021, 765, 142721 CrossRef CAS PubMed.
- W. Zheng, Y. Chen, X. Xu, X. Peng, Y. Niu, P. Xu and T. Li, J. Water Proc. Eng., 2024, 59, 104876 CrossRef.
- L. N. Nthunya, M. F. Bopape, O. T. Mahlangu, B. B. Mamba, B. Van der Bruggen, C. A. Quist-Jensen and H. Richards, J. Environ. Manage., 2022, 301, 113922 CrossRef CAS PubMed.
- A. I. Schäfer, N. Andritsos, A. J. Karabelas, E. M. V. Hoek, R. Schneider and M. Nyström, Nanofiltration: Principles, Applications, and New Materials, 2021 Search PubMed.
- M. Jafari, M. Vanoppen, J. M. C. van Agtmaal, E. R. Cornelissen, J. S. Vrouwenvelder, A. Verliefde, M. C. M. van Loosdrecht and C. Picioreanu, Desalination, 2021, 500, 114865 CrossRef CAS.
- W. Guo, H.-H. Ngo and J. Li, Bioresour. Technol., 2012, 122, 27–34 CrossRef CAS PubMed.
- S. Jiang, Y. Li and B. P. Ladewig, Sci. Total Environ., 2017, 595, 567–583 CrossRef CAS PubMed.
- F. E. Ahmed, B. S. Lalia, N. Hilal and R. Hashaikeh, Desalination, 2017, 406, 60–66 CrossRef CAS.
- F. E. Ahmed, R. Hashaikeh and N. Hilal, Desalination, 2019, 470, 114118 CrossRef CAS.
- L. Shen, Q. Wu, Q. Ye, H. Lin, J. Zhang, C. Chen, R. Yue, J. Teng, H. Hong and B.-Q. Liao, Water Res., 2023, 243, 120353 CrossRef CAS PubMed.
- X. Lu, L. Shen, Q. Zeng, J. Du, C. Chen, J. Teng, W. Yu, Y. Xu and H. Lin, J. Membr. Sci., 2024, 692, 122310 CrossRef CAS.
- W. Yu, Y. Liu, Y. Xu, R. Li, J. Chen, B.-Q. Liao, L. Shen and H. Lin, J. Membr. Sci., 2019, 581, 401–412 CrossRef CAS.
- Y. Liu, L. Shen, Z. Huang, J. Liu, Y. Xu, R. Li, M. Zhang, H. Hong and H. Lin, J. Membr. Sci., 2022, 641, 119925 CrossRef CAS.
- I. M. Noël, R. Lebrun and C. R. Bouchard, Desalination, 2000, 129, 125–136 CrossRef.
- X. Liao, Y. J. Lim, M. Khayet, Y. Liao, L. Yao, Y. Zhao and A. G. Razaqpur, Water Res., 2023, 244, 120511 CrossRef CAS PubMed.
- A. Antony, J. H. Low, S. Gray, A. E. Childress, P. Le-Clech and G. Leslie, J. Membr. Sci., 2011, 383, 1–16 CrossRef CAS.
- A. Sweity, Y. Oren, Z. Ronen and M. Herzberg, Water Res., 2013, 47, 3389–3398 CrossRef CAS PubMed.
- J. S. Vrouwenvelder, S. A. Manolarakis, H. R. Veenendaal and D. van der Kooij, Desalination, 2000, 132, 1–10 CrossRef CAS.
- F. Ahmi and A. Gadri, Desalination, 2004, 166, 427–434 CrossRef.
- University of California, US Pat., US20240109039A1, 2024 Search PubMed.
- Y. Ibrahim and N. Hilal, ACS Appl. Eng. Mater., 2024, 2, 1094–1109 CrossRef CAS.
- X. Qian, A. Anvari, E. M. V. Hoek and J. R. McCutcheon, Desalination, 2023, 556, 116518 CrossRef CAS.
- L. Pisharody, C. Thamaraiselvan, E. Manderfeld, S. P. Singh, A. Rosenhahn and C. J. Arnusch, Adv. Mater. Interfaces, 2022, 9, 2201443 CrossRef CAS.
- Y. Baek, H. Yoon, S. Shim, J. Choi and J. Yoon, Environ. Sci. Technol. Lett., 2014, 1, 179–184 CrossRef CAS.
- R. Alam, S. U. Khan, M. Usman, M. Asif and I. H. Farooqi, Process Saf. Environ. Prot., 2022, 158, 625–643 CrossRef CAS.
- X. Wang, H. Zhang, X. Wang, S. Chen, H. Yu and X. Quan, Front. Environ. Sci. Eng., 2022, 17, 1 Search PubMed.
- H. Zhang, L. Du, J. Xing, G. Wei and X. Quan, Front. Environ. Sci. Eng., 2022, 17, 59 CrossRef.
- H. Zheng, X. Meng, D. Liu, J. Wu and S. Huo, J. Water Proc. Eng., 2023, 55, 104084 CrossRef.
- H. Zheng, X. Meng, J. Wu, D. Liu and S. Huo, Chemosphere, 2023, 342, 140152 CrossRef CAS PubMed.
- Z.-J. Fu, S.-K. Jiang, X.-Y. Chao, C.-X. Zhang, Q. Shi, Z.-Y. Wang, M.-L. Liu and S.-P. Sun, Water Res., 2022, 222, 118888 CrossRef CAS PubMed.
- X. Yang, G. Wei, J. Cao, Z. Ding, R. Yuan, J. Long and C. Xu, ACS Sustainable Chem. Eng., 2024, 12, 3378–3389 CrossRef CAS.
- R. Al-Tohamy, S. S. Ali, F. Li, K. M. Okasha, Y. A.-G. Mahmoud, T. Elsamahy, H. Jiao, Y. Fu and J. Sun, Ecotoxicol. Environ. Saf., 2022, 231, 113160 CrossRef CAS PubMed.
- P. Moradihamedani, Polym. Bull., 2022, 79, 2603–2631 CrossRef CAS.
- N. Panigrahy, A. Priyadarshini, M. M. Sahoo, A. K. Verma, A. Daverey and N. K. Sahoo, Environ. Technol. Innovation, 2022, 27, 102423 CrossRef CAS.
- R. Picetti, M. Deeney, S. Pastorino, M. R. Miller, A. Shah, D. A. Leon, A. D. Dangour and R. Green, Environ. Res., 2022, 210, 112988 CrossRef CAS PubMed.
- A. Popova, R. Rattanakom, Z.-Q. Yu, Z. Li, K. Nakagawa and T. Fujioka, Water Res., 2023, 244, 120484 CrossRef CAS PubMed.
- L. Dirani, G. M. Ayoub, L. Malaeb and R. M. Zayyat, J. Environ. Chem. Eng., 2024, 12, 113325 CrossRef CAS.
- D. M. Wanninayake, J. Environ. Manage., 2021, 283, 111977 CrossRef CAS PubMed.
- Md. N. Pervez, T. Jiang and Y. Liang, J. Water Proc. Eng., 2024, 63, 105471 CrossRef.
- S. Brendel, É. Fetter, C. Staude, L. Vierke and A. Biegel-Engler, Environ. Sci. Eur., 2018, 30, 9 CrossRef PubMed.
- N. Hilal, G. J. Kim and C. Somerfield, Desalination, 2011, 273, 23–35 CrossRef CAS.
- G. M. Geise, H.-S. Lee, D. J. Miller, B. D. Freeman, J. E. McGrath and D. R. Paul, J. Polym. Sci., Part B:Polym. Phys., 2010, 48, 1685–1718 CrossRef CAS.
- J. Wang, S. Hao, P. Qi, W. Wang and Y. Hu, J. Membr. Sci., 2024, 709, 123092 CrossRef CAS.
- C. Liu, W. Wang, B. Yang, K. Xiao and H. Zhao, Water Res., 2021, 195, 116976 CrossRef CAS PubMed.
- M. T. Darestani, T. C. Chilcott and H. G. L. Coster, J. Membr. Sci., 2014, 452, 367–378 CrossRef CAS.
- L. Fei, L. Lu, C. Chen, L. Shen, B. Li, M. Zhou, J. Xu, B. Wang and H. Lin, ACS EST Eng., 2023, 3, 862–873 CrossRef CAS.
- R. Rohani and I. I. Yusoff, J. Polym. Res., 2019, 26, 125 CrossRef.
- R. Rohani and I. I. Yusoff, Iran. Polym. J., 2019, 28, 789–800 CrossRef CAS.
- V. H. Hegde, M. F. Doherty and T. M. Squires, Science, 2022, 377, 186–191 CrossRef CAS PubMed.
- Y. Wu, Z. Chen, C. Lu, C. Hu and J. Qu, ACS Nano, 2025, 19, 4993–5004 CrossRef CAS PubMed.
- J. Hao, J. Li, L. Wang, M. He, J. Wang, X. Wang, X. Zhang and J. Xin, J. Membr. Sci., 2025, 724, 123993 CrossRef CAS.
- Y. Wu, Z. Gu, C. Lu, C. Hu and J. Qu, Water Res., 2023, 244, 120478 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2025 |