Ultra-broadband microwave absorption performance of multi-scale designed Fe3O4@NiCo2O4/PANI/NRGO aerogels
Yousef
Mehdizadeh
a,
Jamileh
Seyed-Yazdi
 *a,
Farnaz
Jamadi
*a,
Farnaz
Jamadi
 ab,
Fatemeh
Ebrahimi-Tazangi
a,
Seyyed Jalal
Seyyedyazdi
ab,
Fatemeh
Ebrahimi-Tazangi
a,
Seyyed Jalal
Seyyedyazdi
 c,
Seyed Mohammad
Hosseiny
c,
Seyed Mohammad
Hosseiny
 a and
José
Teixeira
a and
José
Teixeira
 d
d
aDepartment of Physics, Faculty of Science, Vali-e-Asr University of Rafsanjan, Rafsanjan, Iran. E-mail: j.seyedyazdi@gmail.com
bDepartment of Physics, Sirjan University of Technology, Sirjan, Iran
cDepartment of Computer Science, Vali-e-Asr University of Rafsanjan, Rafsanjan, Iran
dLaboratoire Leon Brillouin (CEA/CNRS) CEA Saclay, France
First published on 18th June 2025
Abstract
In this study, we developed a microstructure design and an appropriate multicomponent strategy to optimize a microwave absorber: Fe3O4@NiCo2O4/PANI/NRGO aerogels. We synthesized Fe3O4 hollow spheres, Fe3O4@NiCo2O4, Fe3O4@NiCo2O4/PANI, and finally, Fe3O4@NiCo2O4/PANI/NRGO aerogels using hydrothermal and in situ polymerization methods. The maximum reflection loss of the aerogels can reach −41.8 dB at 16.7 GHz with a thickness of only 2.0 mm; the bandwidth, with absorption above 90%, is 13.3 GHz (4.7–18 GHz for matching thicknesses of 1.6–5.0 mm). The maximum reflection loss of the Fe3O4@NiCo2O4 sample reaches −50.9 dB at 5.7 GHz with a matching thickness of 3.0 mm; the bandwidth with a reflection loss less than −10 dB is 15.0 GHz (3–18 GHz for 1.0–5.0 mm matching thicknesses). The enhanced electromagnetic absorption performance is due to synergetic magnetic loss, dielectric loss, and conduction loss in the porous multi-scaled structure with a hollow interior, including various rich interfaces provided by the distribution of different components in the heterostructures. Our results provide a new approach for designing and constructing excellent microwave-absorbing materials for practical applications.
1 Introduction
As electronic devices and advanced communication technologies permeate our lives, the threat of electromagnetic pollution has intensified, threatening both human health and the functionality of essential electronic systems. In response, microwave absorbing materials (MAMs) are a promising solution, attracting considerable attention due to their potential to mitigate electromagnetic interference.1–3 Emerging from post-World War II radar advances, MAMs are now integral to applications ranging from wireless communication to stealth military technologies. The escalating proliferation of electronic devices amplifies the urgency for efficient MAMs. Recent innovations in materials science, particularly with carbon-based composites, led to the development of lightweight, durable absorbers with enhanced absorption capabilities and broadened operational bandwidths.4–6 This quest for high-performance MAMs is crucial not only to protect public health, but also to ensure the reliability of our interconnected systems.7Spinel nickel cobaltite (NiCo2O4) is a ternary metal oxide that exhibits remarkable properties, including excellent oxidation resistance, strong magnetism, and significantly higher electrical conductivity, at least two orders of magnitude larger than that of analogous nickel and cobalt oxides.8 With its simple preparation process and abundant availability, NiCo2O4 is increasingly recognized as an exceptional candidate for applications in microwave absorption and other advanced technological fields.9–12
The two main strategies to improve the absorption performance are based on magnetic loss and dielectric loss (including conductive loss). Iron oxides such as Fe3O4 are among the best options to improve the magnetic loss of the absorbers.13–17 Graphene and graphene-based materials are among the best choices to enhance the conductive loss and dielectric loss of microwave-absorbing materials.18–21 Synergistically regulating the polarization loss and conductivity loss of carbon-based materials is a crucial scientific challenge for optimizing their microwave absorption performance. The nanocomposites integrating carbon-based materials, magnetic materials, and conductive polymers demonstrate a profound synergistic effect, optimizing impedance matching and thereby significantly enhancing microwave attenuation efficiency through a harmonious interplay of magnetic and dielectric loss mechanisms. Many researchers have developed and examined different composite materials to explore and advance promising microwave absorbers. Yan et al.22 synthesized lilac flower-like CoNiZnO@nitrogen-doped carbon composites through self-assembly and pyrolysis. The RLmin reaches −70.70 dB at 11.04 GHz with 2.17 mm thickness, and the effective absorption bandwidth (EAB) reaches 4.48 GHz (13.52–18.00 GHz) at 1.55 mm. Hu et al.23 using a simple immersion-annealing strategy, developed a dual-functional NiCo2O4/NiCoO2 co-embedded porous bio-carbon composite, presenting numerous heterogeneous interfaces, which exhibits excellent bacteriostatic properties and microwave absorption. The RLmin reaches −38.2 dB at 2 mm thickness, and the EAB reaches 7.01 GHz at 2.31 mm. Li et al.24 developed FeCo/FeCoNi@N-doped porous carbon composites through a thermal decomposition method followed by pyrolysis in an inert atmosphere, resulting in significantly enhanced microwave absorption performance due to their multi-component system and porous architecture. The RLmin reaches −67 dB at 14.7 GHz with a thickness of 1.91 mm, and the corresponding EAB reaches 6.24 GHz, covering the whole Ku band.
Previous research indicates that heterogeneous, multilayer nanostructured composites can enhance absorption performance. In this study, various multi-component MAMs composed of Fe3O4, NiCo2O4, PANI, and NRGO were prepared using different methods. Among them, the multiscale-designed Fe3O4@NiCo2O4/PANI/NRGO aerogels demonstrate exceptional microwave absorption performance, covering the bandwidth from the C band to the Ku band, and exhibiting strong absorption capabilities despite their thin profile. This performance may stem from the strong synergistic effects that improve impedance matching and overall attenuation. Furthermore, CST simulations were performed to model electromagnetic wave absorbing performance of our materials. The structural, morphological, magnetic, and microwave absorption properties of these nanocomposites were studied in depth.
2 Experimental
2.1 Materials
FeCl3·6H2O (Purity: 98%), GO, Fe(NO3)2·9H2O, Na3C6H5O7·2H2O, CH4·N2O, deionized water (DIW), C3H3NaO2, Ni(NO3)2·6H2O, Co(NO3)2·6H2O, C6H12O6, C2H4(NH2)2 (EDA), HCl, aniline (C6H5NH2), and (NH4)2S2O8 were obtained from Merck or Sigma-Aldrich and used without further purification.2.2 Synthesis of hollow Fe3O4 (S1)
We synthesized Fe3O4 hollow spheres using a hydrothermal method. First, 4.2 g FeCl3·6H2O was dispersed in 120 mL DIW for 20 minutes. Then, we added 3 g of urea and 8 g of sodium citrate dihydrate, stirring the mixture for an additional 25 minutes. In a separate container, 1.8 g of sodium polyacrylate was stirred in 120 mL of DIW for 5 minutes, resulting in a solution resembling white jelly. We then added the iron chloride solution dropwise to the jelly, stirring for 1 hour to ensure thorough mixing. The mixture was transferred to an autoclave and heated to 200 °C for 24 hours to facilitate the hydrothermal reaction. Afterward, the final product was washed with ethanol and DIW until a neutral pH of 7 was achieved and dried at 60 °C for 24 hours.2.3 Synthesis of Fe3O4@NiCo2O4 (S2)
Hollow Fe3O4 was used for synthesizing the nanocomposites and aerogels (S2, S3, and S4). First, 0.4 g Ni(NO3)2·6H2O and 0.8 g Co(NO3)2·6H2O were stirred in 100 mL DIW for 10 minutes, resulting in a purple solution. We then added 150 μL of EDA and 0.4 g of urea to the mixture. Next, 0.27 g C6H12O6 and hollow Fe3O4 were introduced while stirring for 30 minutes. The mixture was then transferred to an oven set at 180 °C for 20 hours to undergo a hydrothermal process. Afterward, the resulting product was washed with ethanol and DIW and subsequently dried. Finally, the material was annealed at 400 °C for 2 hours to complete the calcination.2.4 Synthesis of Fe3O4@NiCo2O4/PANI (S3)
The Fe3O4@NiCo2O4/PANI composite was synthesized through in situ polymerization. First, 33 mL of HCl was mixed with 150 mL of DIW. Next, 1.4 g of the S2 sample was dispersed in 50 mL of DIW and added to the acidic solution. Then, 0.7 g of aniline was incorporated while dispersing the mixture using an ultrasonic cleaner. The resulting mixture was stirred in an ice bath at 0–5 °C for 60 minutes. Following this, 0.7 g (NH4)2S2O8 was dissolved in 60 mL of DIW and stirred for 10 minutes. Then, (NH4)2S2O8 solution was added dropwise to the previous mixture, which was stirred for an additional 7 hours. At the end of the polymerization process, the product (S3) was separated using a magnet, washed with ethanol and DIW, and then dried at room temperature.2.5 Synthesis of Fe3O4@NiCo2O4/PANI/NRGO aerogels (S4)
To prepare Fe3O4@NiCo2O4/PANI/NRGO, 0.9 g of graphene oxide (GO)25 was dispersed in 200 mL of DIW for 1 hour. Next, 0.4 g of the S3 sample was dissolved in 100 mL of DIW. The two solutions were then combined and mixed thoroughly. Subsequently, 1.8 mL of EDA was added dropwise while the mixture was stirred for 90 minutes. The solution was transferred to an autoclave and then heated in an oven at 120 °C for 20 hours. A black solid cake hydrogel was formed, which was thoroughly washed with ethanol and DIW to remove impurities and residual reactants. Subsequently, the product was transferred to a freeze-dryer set at −50 °C for 48 hours, resulting in sublimation by drying under vacuum, which results in the formation of the porous aerogel structure.The prepared samples—Fe3O4, Fe3O4@NiCo2O4, Fe3O4@NiCo2O4/PANI, and Fe3O4@NiCo2O4/PANI/NRGO—are denoted as S1, S2, S3, and S4, respectively. A vector network analyzer (VNA) was employed to measure the electromagnetic parameters. The samples were uniformly mixed with paraffin at specific ratios: 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50 for S1, 15
50 for S1, 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 85 for S2, 30
85 for S2, 30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 70 for S3, and 30
70 for S3, and 30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 70 for S4. The steps involved in preparing these nanocomposite samples are illustrated in Fig. 1.
70 for S4. The steps involved in preparing these nanocomposite samples are illustrated in Fig. 1.
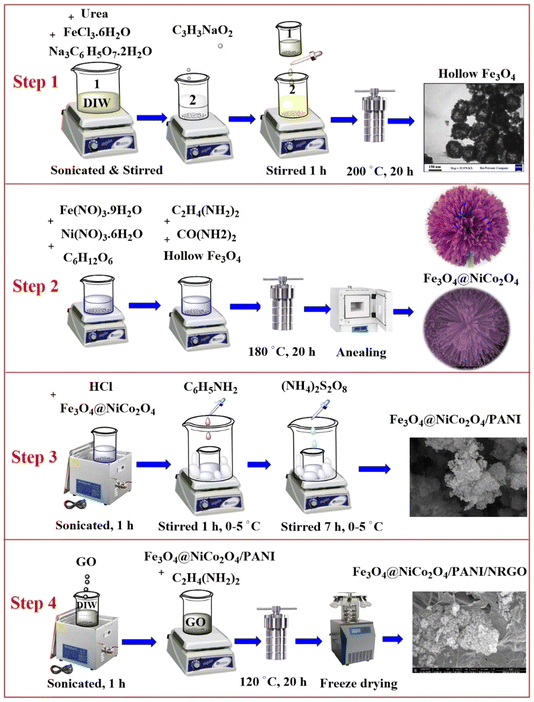 | ||
| Fig. 1 Schematic representation of the synthesis process for (S1) Fe3O4 hollow spheres, (S2) Fe3O4@NiCo2O4, (S3) Fe3O4@NiCo2O4/PANI nanocomposites, and (S4) Fe3O4@NiCo2O4/PANI/NRGO aerogels. | ||
3 Results and discussion
XRD (X-ray diffraction) analysis was conducted to confirm the composition and crystal structure of the nanocomposites and aerogels, as shown in Fig. 2, where distinct diffraction peaks for NiCo2O4, Fe3O4, PANI, and RGO are represented by different symbols. The peaks at 18.9°, 31.2°, 36.7°, 44.6°, 55.4°, 59.1°, and 65° correspond to the (111), (220), (311), (400), (422), (511), and (440) crystal planes of NiCo2O4, respectively. Bragg peaks at 2θ = 18.4°, 30.2°, 35.5°, 43.1°, 53.7°, 57.1°, 62.8°, and 74.2° correspond to the (111), (220), (311), (400), (422), (333), (440), and (533) crystal planes of Fe3O4.26 The low-intensity diffraction peaks at around 2θ = 11.3°, 12.7°, and 15.3° correspond to the (011), (020), and (200) crystal planes of the PANI polymer chain.27,28 Additionally, a low-intensity peak at 2θ = 16.19° is attributed to the (011) plane of the PANI polymer chain. The crystallite sizes of the nanoparticles, estimated using Scherrer's equation, are 56.7, 44.8, 56.3, and 30.1 nm for S1, S2, S3, and S4, respectively.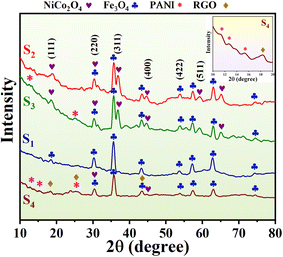 | ||
| Fig. 2 XRD spectra of the synthesized nanocomposites and aerogels: (S1) Fe3O4 hollow spheres, (S2) Fe3O4@NiCo2O4, (S3) Fe3O4@NiCo2O4/PANI nanocomposites, and (S4) Fe3O4@NiCo2O4/PANI/NRGO aerogels. | ||
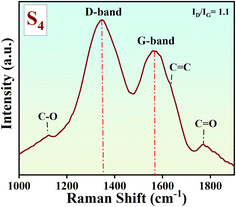
Raman spectroscopy was performed using a 532 nm excitation laser. The Raman spectrum of the S4 aerogel (Fig. 3) displays D- and G-bands at 1353 cm−1 and 1578 cm−1, respectively. This shows a blue shift in the D-band and a red shift in the G-band compared to those of free-standing RGO, which has D-band and G-band peaks at 1335 cm−1 and 1590 cm−1, respectively. The D-band is associated with the disordered or defect-induced A1g breathing mode of carbon atoms, indicating structural imperfections in sp2 domains. While disorder can coexist with sp3 hybridization in certain carbon materials (e.g., amorphous carbons), the D-band primarily signals defects rather than a direct sp2 to sp3 transition. The G-band arises from the E2g vibration mode of ordered sp2 hybridized carbon atoms in graphitic structures. The intensity ratio of the D band to the G band (ID/IG) reflects the extent of graphitization, where a lower ratio indicates a higher degree of graphitization.29–31
FTIR (Fourier transform infrared) spectroscopy was employed to confirm the presence of various functional groups and identify the types of bonds in the nanocomposites and aerogels. Fig. 4 shows the FTIR spectra for samples S1, S2, S3, and S4. The peak observed at 3450 cm−1 is attributed to the stretching vibration of hydroxyl groups, indicating water adsorption on the sample surfaces. For NiCo2O4, a peak related to the Ni–O bond is present at 558 cm−1, while Fe3O4 shows a peak at 562 cm−1 corresponding to the Fe–O bond; these peaks overlap in sample S2. Additionally, NiCo2O4 has a weak Co–O peak at 648 cm−1 in the core@shell sample (S2). Samples S3 and S4, which contain PANI, show related bonds as depicted in Fig. 4. The N–H stretching peak (approximately 3450 cm−1) of PANI may broaden due to hydrogen bonding with oxygen-containing groups in NRGO. The C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C stretching peaks (around 1600 cm−1 and ∼1500 cm−1) of PANI may slightly shift due to π–π interactions with the graphene sheets in NRGO. The metal–oxygen peaks (approximately 550–650 cm−1) appear weaker when the metal oxides are well-coated with PANI and NRGO.32,33
C stretching peaks (around 1600 cm−1 and ∼1500 cm−1) of PANI may slightly shift due to π–π interactions with the graphene sheets in NRGO. The metal–oxygen peaks (approximately 550–650 cm−1) appear weaker when the metal oxides are well-coated with PANI and NRGO.32,33
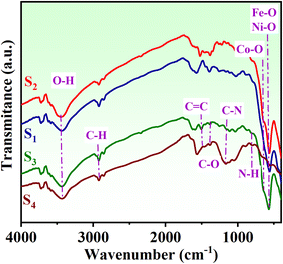 | ||
| Fig. 4 FTIR analysis of (S1) Fe3O4 hollow spheres, (S2) Fe3O4@NiCo2O4, (S3) Fe3O4@NiCo2O4/PANI nanocomposites, and (S4) Fe3O4@NiCo2O4/PANI/NRGO aerogels. | ||
The morphology, particle size, composition, elemental distribution, and characterization of the samples were analyzed using SEM, EDS, TEM, and elemental mapping, as illustrated in Fig. 5.
A vibration sample magnetometer (VSM) measures the magnetic properties of the nanocomposites. As shown in Fig. 6, the hysteresis loop of the samples varies with the applied magnetic field, ranging from −14![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 to 14
000 to 14![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 Oe. The inset of Fig. 6 highlights low hysteresis, indicative of superparamagnetic behavior in sample S1. In contrast, the hysteresis loops for S2, S3, and S4 demonstrate ferromagnetic characteristics at room temperature. The magnetic saturation values for S1, S2, S3, and S4 are 78.0, 39.1, 38.7, and 21.7 (emu g−1), respectively, measured at a maximum magnetic field of 1.4 T. As expected, the Fe3O4 hollow spheres exhibit the highest magnetic saturation, resulting in greater magnetic responsiveness. The introduction of a NiCo2O4 shell on the Fe3O4 core reduces magnetic saturation. Importantly, in this experiment, PANI adopts a configuration within the structure that does not alter magnetic properties, which is crucial for metacomposites,34 as nano/micro scale arrangements can significantly impact overall performance. Typically, adding RGO (in this case, NRGO) results in decreased magnetic saturation of the aerogels.
000 Oe. The inset of Fig. 6 highlights low hysteresis, indicative of superparamagnetic behavior in sample S1. In contrast, the hysteresis loops for S2, S3, and S4 demonstrate ferromagnetic characteristics at room temperature. The magnetic saturation values for S1, S2, S3, and S4 are 78.0, 39.1, 38.7, and 21.7 (emu g−1), respectively, measured at a maximum magnetic field of 1.4 T. As expected, the Fe3O4 hollow spheres exhibit the highest magnetic saturation, resulting in greater magnetic responsiveness. The introduction of a NiCo2O4 shell on the Fe3O4 core reduces magnetic saturation. Importantly, in this experiment, PANI adopts a configuration within the structure that does not alter magnetic properties, which is crucial for metacomposites,34 as nano/micro scale arrangements can significantly impact overall performance. Typically, adding RGO (in this case, NRGO) results in decreased magnetic saturation of the aerogels.
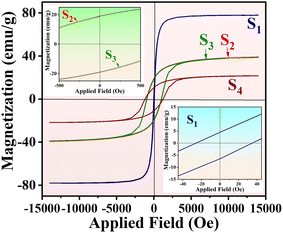 | ||
| Fig. 6 VSM analysis of synthesized nanocomposites: (S1) Fe3O4 hollow spheres, (S2) Fe3O4@NiCo2O4, (S3) Fe3O4@NiCo2O4/PANI nanocomposites, and (S4) Fe3O4@NiCo2O4/PANI/NRGO aerogels. | ||
To investigate the porosity and surface area characteristics of the aerogels, N2 adsorption–desorption isotherms35,36 were obtained, as shown in Fig. 7. The isotherm exhibits a Type II profile, typical of nanoporous materials, which indicates a gradual nitrogen uptake at low relative pressures followed by a more significant increase at higher pressures, with minimal hysteresis. This behavior suggests well-defined mesoporosity and effective pore connectivity within the aerogel structure, facilitated by defect-rich carbon (NRGO) and polyaniline (PANI) layers that create interconnected conductive networks. The surface area of sample S4 is 23.19 m2 g−1, with a mean pore diameter of approximately 3.99 nm.
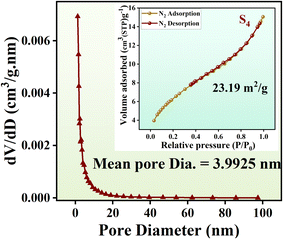 | ||
| Fig. 7 N2 adsorption–desorption isotherm, surface area, and estimated pore size distribution curves for Fe3O4@NiCo2O4/PANI/NRGO aerogels (S4). | ||
4 Microwave absorption analysis
To investigate the microwave absorption mechanism, we measured the electromagnetic parameters–relative complex permittivity (εr = ε′ − jε′′) and relative complex permeability (μr = μ′ − jμ′′)—of nanocomposites and aerogels using a vector network analyzer over the frequency range of 1–18 GHz. The real parts of the complex permittivity and permeability (ε′ and μ′) are related to the storage of electric and magnetic energy, while the imaginary parts (ε′′ and μ′′) correspond to energy dissipation. The dielectric tangent loss (tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δε = ε′′/ε′) and magnetic tangent loss (tan
δε = ε′′/ε′) and magnetic tangent loss (tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δμ = μ′′/μ′) are used to demonstrate overall wave attenuation performance.
δμ = μ′′/μ′) are used to demonstrate overall wave attenuation performance.
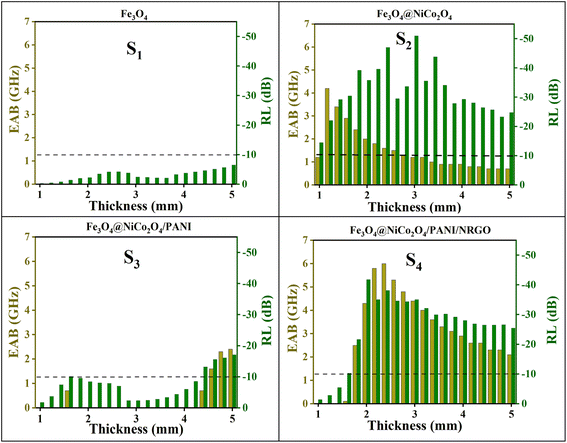
The microwave absorption performance of the nanocomposites can be represented visually by using the strength of the reflection loss (RL). Based on transmission line theory, RL values associated with the electromagnetic parameters can be calculated using the following formulae:37
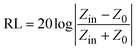 | (1) |
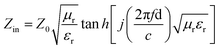 | (2) |
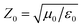 is the impedance of free space. In this equation, f represents the microwave frequency, d is the layer thickness of the absorber, and c is the velocity of light in free space.
is the impedance of free space. In this equation, f represents the microwave frequency, d is the layer thickness of the absorber, and c is the velocity of light in free space.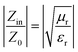 | (3) |
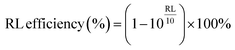 | (4) |
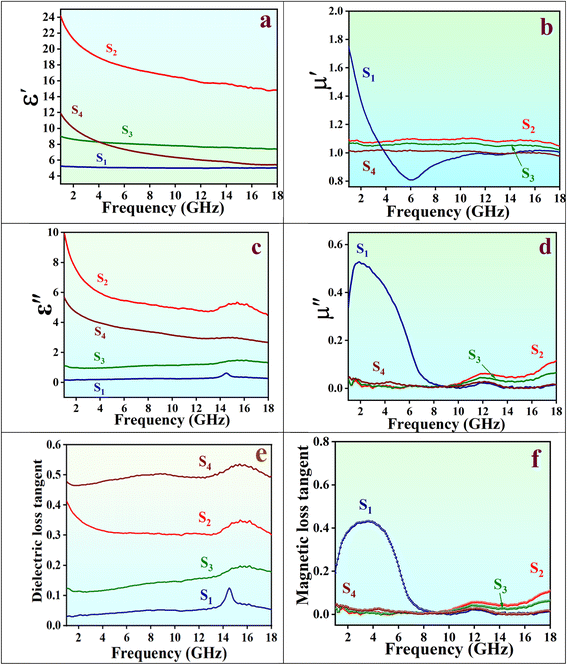
The variations of electromagnetic parameters with frequency are illustrated in Fig. 8. Fig. 8a displays the frequency dispersion effect for ε′, which shows a decreasing trend across the entire frequency range. This decrease occurs because dipole orientations align with low-frequency electric fields but cannot keep up with rapid fluctuations in high-frequency fields.38 The S1 sample (Fe3O4 hollow spheres) exhibits the lowest and primarily frequency-independent ε′ and ε′′ values due to its limited polarization loss capabilities. In contrast, these parameters are enhanced in other samples, reaching their highest values in the case of the S2 sample (NiFe2O4/Fe3O4 core@shells), closely followed by S4 aerogels, as shown in Fig. (8a, c and e), due to enhanced interfacial polarization.39 The identical resonance peaks observed in the tan δε curves across all samples suggest similar polarization loss mechanisms. Regarding μ′ and μ′′ performance, the S1 sample shows the highest magnetic energy storage capability below 4 GHz and the greatest magnetic energy dissipation below 9 GHz. Above these frequencies, the S2 sample demonstrates superior performance in terms of μ′ and μ′′ (Fig. 8b and d). Dielectric tangent loss (tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δε = ε′′/ε′) and magnetic tangent loss (tan
δε = ε′′/ε′) and magnetic tangent loss (tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δμ = μ′′/μ′) are presented in Fig. 8e and f. For all composites except S1, dielectric polarization processes primarily govern electromagnetic energy attenuation. Effective microstructure engineering that enhances interfacial polarization, and composition control to manipulate dielectric loss, are key strategies for developing superior wave absorbers, particularly in the case of S4 aerogels. The high dielectric tangent loss values of S4 aerogels indicate that the quality of carbonization, enhanced by the introduction of RGO, significantly influences the electromagnetic parameters, emphasizing its crucial role in increasing dielectric loss through enhanced polarization capabilities.
δμ = μ′′/μ′) are presented in Fig. 8e and f. For all composites except S1, dielectric polarization processes primarily govern electromagnetic energy attenuation. Effective microstructure engineering that enhances interfacial polarization, and composition control to manipulate dielectric loss, are key strategies for developing superior wave absorbers, particularly in the case of S4 aerogels. The high dielectric tangent loss values of S4 aerogels indicate that the quality of carbonization, enhanced by the introduction of RGO, significantly influences the electromagnetic parameters, emphasizing its crucial role in increasing dielectric loss through enhanced polarization capabilities.
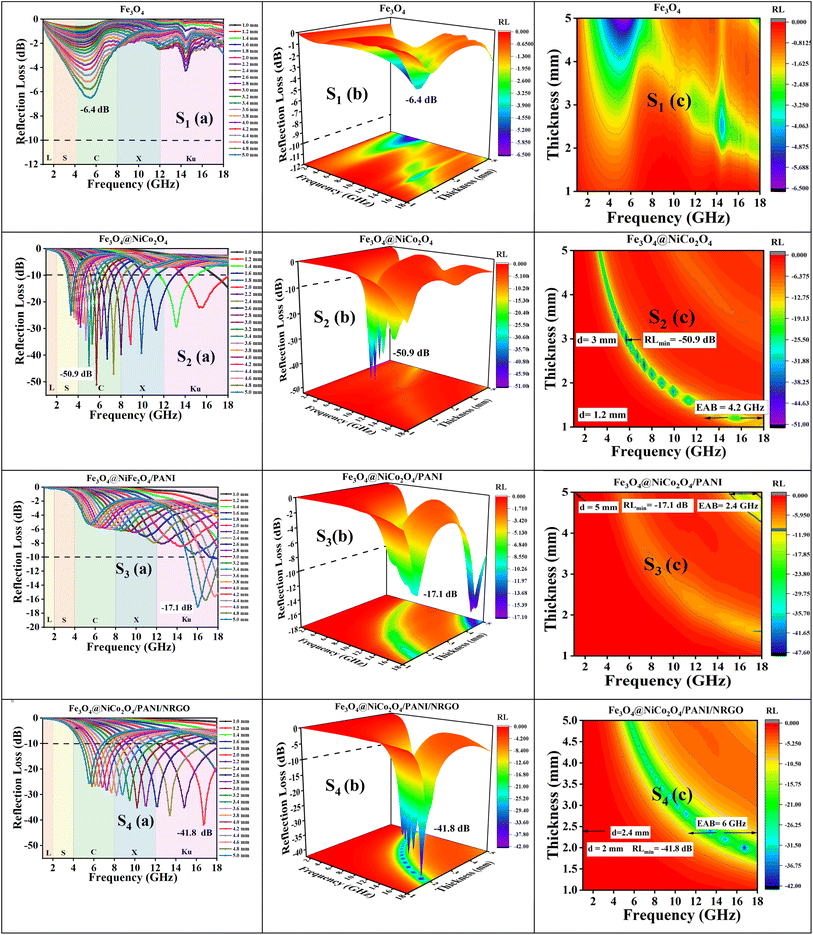
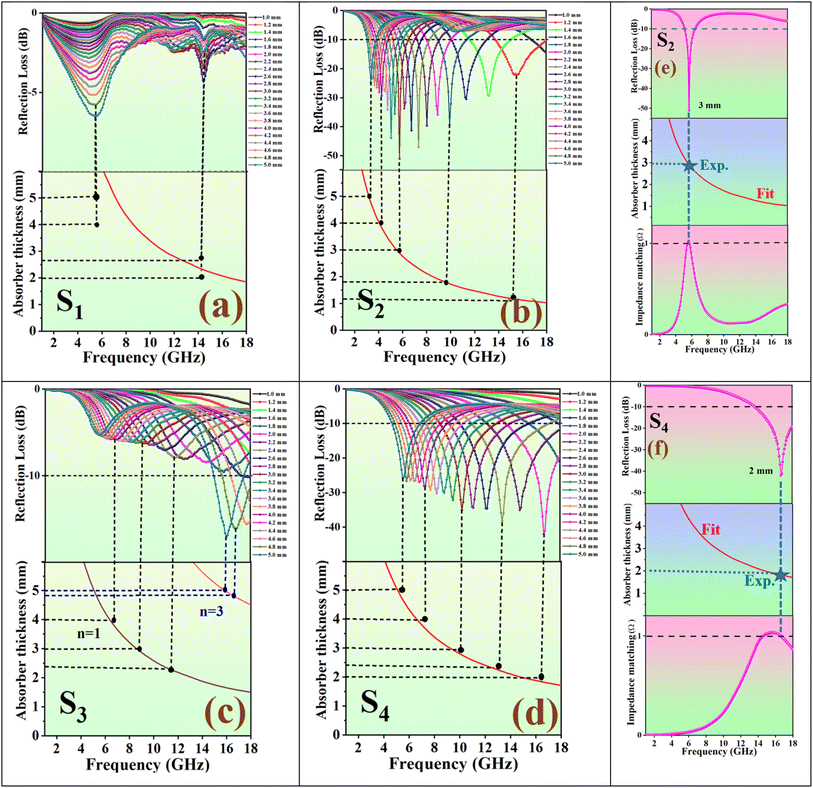
Fig. 9 presents 2D reflection loss data and 3D reflection loss maps for absorbers with varying layer thicknesses, accompanied by a 2D contour diagram that illustrates the relationship between thickness and frequency for the nanocomposites and aerogels. Panels S2(a) and S4(a) in Fig. 9 showcase exceptional electromagnetic wave absorption performance across the entire frequency range, covering from S to Ku bands for S2 and the C to Ku bands for S4. Table 1 summarizes the RLmin, absorber thickness, EAB, coercive field (Hc), remanence magnetization (Mr), and magnetic saturation (Ms) of all prepared samples.
| Sample | RLmin (dB) | f (GHz) | t m (mm) | EAB (GHz) | |Hc|(Oe) | M r (emu g−1) | M S (emu g−1) |
|---|---|---|---|---|---|---|---|
| Fe3O4 (S1) | −6.4 | 5.4 | 5 | 0 | 34.5 | 6.9 | 78 |
| Fe3O4@NiCo2O4 (S2) | −50.9 | 5.7 | 3 | 4.2 | 1100 | 19.2 | 39.1 |
| Fe3O4@NiCo2O4/PANI (S3) | −17.1 | 16 | 5 | 2.4 | 1100 | 18.8 | 38.7 |
| Fe3O4@NiCo2O4/PANI/NRGO (S4) | −41.8 | 16.7 | 2 | 6 | 1250 | 10.6 | 21.7 |
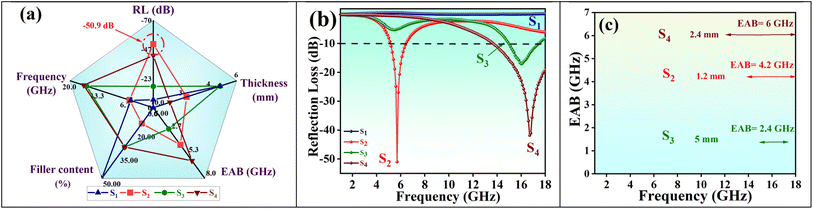
The radar chart in Fig. 10(a) compares and summarizes the electromagnetic wave absorption parameters for the prepared nanocomposites and aerogels. Fig. 10(b) displays the optimal RLmin values, while Fig. 10(c) presents the optimal EAB achieved using the optimized thickness across all samples at varying frequencies.
Fig. 11 offers additional information on RL and EAB at various thicknesses for comparative analysis. A higher RL indicates better microwave absorption; however, an ideal absorber should exhibit high RL, a wide EAB, and minimal thickness simultaneously. According to eqn (4), both S2 and S4 achieve over 99% electromagnetic wave absorption. Thus, considering EAB values and thickness, we conclude that S2 and S4 demonstrate exceptional performance.
 | ||
| Fig. 11 RL and EAB versus thickness for (S1) Fe3O4 hollow spheres, (S2) Fe3O4@NiCo2O4, (S3) Fe3O4@NiCo2O4/PANI nanocomposites, and (S4) Fe3O4@NiCo2O4/PANI/NRGO aerogels. | ||
In transmission line theory for electronic circuits, the matching thickness can be determined under the quarter-wavelength matching condition (eqn (5)). Fig. 12 shows the calculated matching thicknesses of the prepared samples as a function of frequency.40 By analyzing the peaks associated with RLmin, we find that an ideal absorber's matching thicknesses correspond to the λ/4 and 3λ/4 curves, closely aligning with thicknesses that meet the nλ/4 condition. At these matched thicknesses, microwaves reflect off the surface, achieving a 180° phase difference between the incident and reflected waves. This phase cancellation results in the dissipation of electromagnetic waves at the interface.41,42
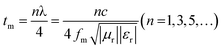 | (5) |
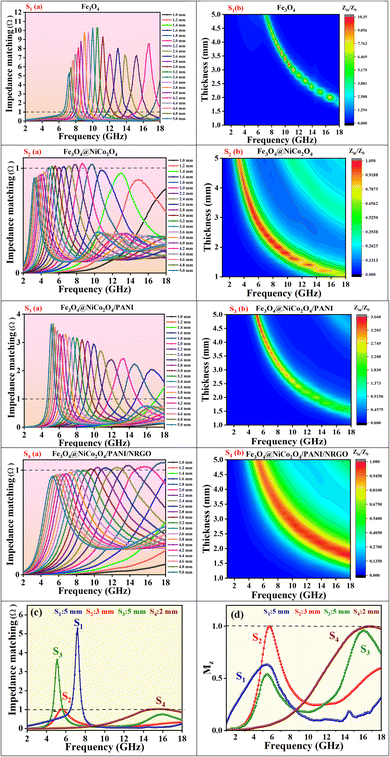
4.1 Impedance matching
When designing microwave absorbers and their composite materials, a key factor to optimize absorption performance is the simultaneous improvement of impedance matching and attenuation capability. Effective impedance matching is crucial for electromagnetic absorption, as it maximizes the efficient transmission of incident microwaves into the absorber. Impedance matching is defined in eqn (6):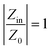 | (6) |
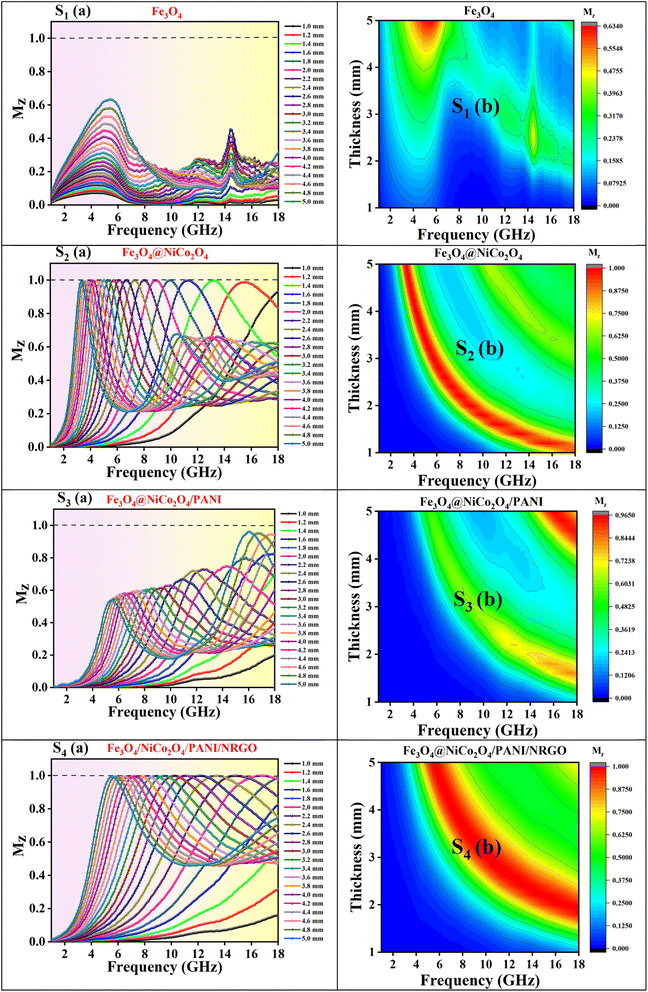
This means that when the characteristic impedance of a material is very close to that of free space, electromagnetic waves can effectively penetrate the material, leading to minimal reflection at the surface of the absorber layer. To assess impedance matching, consider the parameter Z = |Zin/Z0|, which represents the ratio of the input impedance to the impedance of free space,43 and the input impedance matching coefficient (Mz), as defined in eqn (7):44
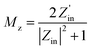 | (7) |
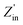 is the real part of the normalized input impedance.45Fig. 13 and 14 illustrate the trend of impedance matching using 2D contour maps for various samples as a function of frequency, allowing for easy comparison with RLmin values.
is the real part of the normalized input impedance.45Fig. 13 and 14 illustrate the trend of impedance matching using 2D contour maps for various samples as a function of frequency, allowing for easy comparison with RLmin values.
 | ||
| Fig. 14 2D and contour diagrams of the input impedance matching coefficient for S1 (a and b), S2 (a and b), S3 (a and b), and S4 (a and b). | ||
Fig. 13d presents the optimal input impedance matching coefficient for the samples, while Fig. 14 provides a detailed overview of the impedance matching coefficient across all samples. As the matching coefficient MZ approaches 1, impedance matching improves, allowing a greater portion of the incident electromagnetic waves to enter the absorber. Additionally, when |Zin/Z0| = 1, EM waves can fully penetrate the material, resulting in zero reflection. The closer |Zin/Z0| is to 1, the more effectively incident microwaves can penetrate the material. Better impedance matching occurs for higher tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δε samples (e.g. S4).46 High impedance matching is crucial for effective absorption, as it indicates that most incident microwaves pass through the absorbing materials rather than being reflected at their surface. Notably, a higher absorption performance correlates with improved impedance matching. In the frequency range of 1–18 GHz, the |Zin/Z0| values for samples S2 and S4 samples are consistently close to 1, demonstrating suitable impedance matching that contributes to their outstanding microwave absorption performance.
δε samples (e.g. S4).46 High impedance matching is crucial for effective absorption, as it indicates that most incident microwaves pass through the absorbing materials rather than being reflected at their surface. Notably, a higher absorption performance correlates with improved impedance matching. In the frequency range of 1–18 GHz, the |Zin/Z0| values for samples S2 and S4 samples are consistently close to 1, demonstrating suitable impedance matching that contributes to their outstanding microwave absorption performance.
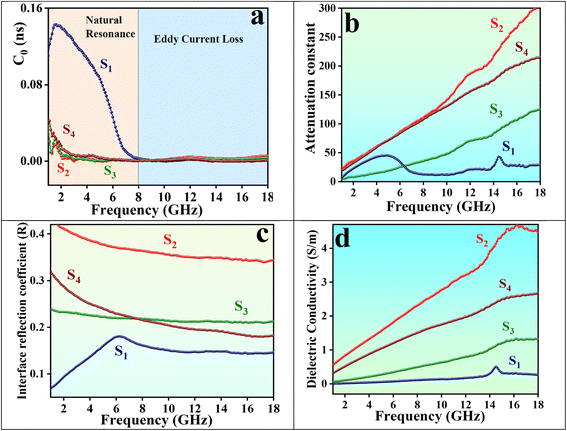
In many cases, magnetic loss is jointly dominated by natural and exchange resonance.47 However, eddy current loss can significantly impact magnetic loss in the GHz frequency range. The rapid fluctuations in the magnetic field at these high frequencies induce substantial circulating currents (eddy currents) in conductive materials, leading to considerable energy dissipation as heat. The contribution of the eddy current effect can be analyzed using the coefficient C0, as defined in eqn (9):48,49
| μ′′ = 2/3πμ0(μ′)2σd2f | (8) |
| C0 = μ′′(μ′)−2f−1 = 2/3πμ0σd2 = constant | (9) |
If C0 remains constant within the measured frequency range, magnetic loss is attributed to eddy currents. As shown in Fig. 15a, eddy current loss is negligible at frequencies below 8 GHz. However, at higher frequencies, with constant C0 values, eddy current loss becomes the main contributor to magnetic loss. This phenomenon is related to the skin effect, whereby the penetration depth of microwaves decreases at high frequencies, resulting in localized current dissipation at the surface that causes microwave attenuation and improves absorption performance at these frequencies.50
When microwaves can readily penetrate the absorber (i.e., with impedance matching close to 1) and the absorber demonstrates strong attenuation capability (high α values), the incident microwaves are effectively dissipated. The attenuation constant α, which characterizes the absorber's attenuation performance and is illustrated in Fig. 15b, is a crucial parameter for evaluating the absorption capabilities of materials. It is determined by the values of permittivity and permeability, as defined in eqn (10):51
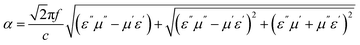 | (10) |
When the characteristic impedance of the absorbers matches or approaches that of free space, microwaves can easily enter the absorbers without reflection. The interface reflection coefficient R is defined as the ratio of the reflected wave to the incident wave, as derived from eqn (11),52 and illustrated in Fig. 15c:
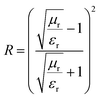 | (11) |
An optimal low R-value indicates effective impedance matching, allowing more electromagnetic waves to penetrate the material instead of being reflected. Reducing R through impedance-matching techniques is essential for enhancing electromagnetic wave penetration and maximizing absorption efficiency.
Conduction loss occurs when charge carriers move through a material, resulting in energy dissipation as heat. Generally, materials with higher conductivity experience greater conduction loss. Several factors influence dielectric conductive loss, including material conductivity, morphology, defects, and heterogeneous interfaces. Hollow structures can enhance conductivity and conduction loss by increasing the volume fraction of the absorbent material, creating a more extensive conductive network, and facilitating electron transport. This leads to improved electromagnetic wave attenuation. Additionally, hollow structures optimize impedance matching, allowing a greater number of electromagnetic waves to penetrate the material while promoting multiple reflections and scattering, which further enhances energy dissipation.53 Dielectric conductivity is defined by using eqn (12) and illustrated in Fig. 15d.
| σ = 2πfε0ε′′ | (12) |
Fig. 15d shows that S2, which has core@shell structures with hollow Fe3O4 cores, and S4, composed of aerogels with multi-dimensional structures, offer conductive pathways for efficient electron transport, resulting in higher dielectric conductivity among the samples.
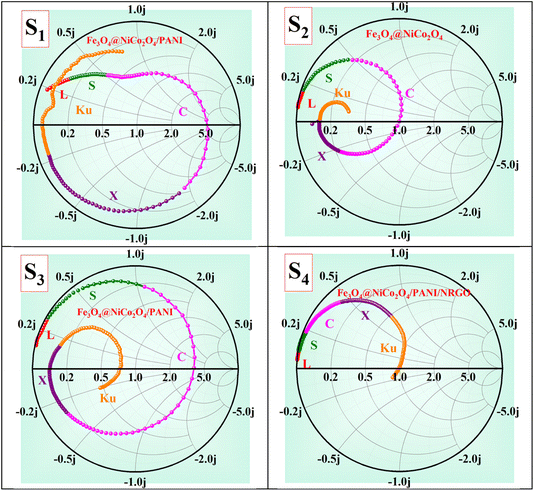
Fig. 16 presents the Smith chart characteristics for all the prepared samples. The center point of the Smith chart represents the matching point; the closer the curve is to the center, the better the impedance-matching conditions.1,54,55 According to Fig. 16, the S2 and S4 samples show the best impedance matching—S2 in the C band, and S4 in the Ku band—consistent with the findings in Fig. 13 and 14.
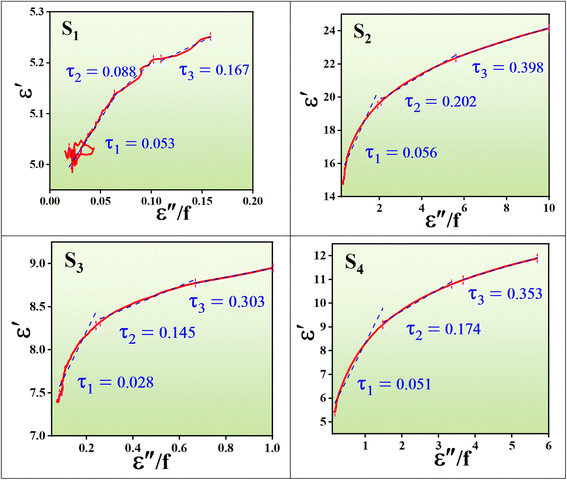
Fig. 17 displays both 3D and 2D Cole–Cole curves for the prepared samples. Each Debye relaxation process occurring at the interfaces, or due to dipole polarization, corresponds to a semicircle on the ε" versus ε′ plot (referred to as Cole–Cole plots), which can be mathematically expressed by using eqn (13):
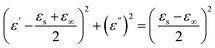 | (13) |
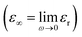 and εs
and εs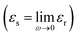 represent the optical (high-frequency limit) and static dielectric constants, respectively.56 The semicircle curves in Fig. 17 depict the dielectric relaxation process, while the slopes of the long straight tails on the Cole–Cole curves provide an estimate of conductive loss capacity (Fig. 18). Generally, a larger semicircle radius suggests a stronger dielectric relaxation.41 The presence of multiple semicircles, indicative of significant polarization effects, implies that several relaxation processes occur, leading to a reduction in the associated conduction loss over the frequency range.57,58
represent the optical (high-frequency limit) and static dielectric constants, respectively.56 The semicircle curves in Fig. 17 depict the dielectric relaxation process, while the slopes of the long straight tails on the Cole–Cole curves provide an estimate of conductive loss capacity (Fig. 18). Generally, a larger semicircle radius suggests a stronger dielectric relaxation.41 The presence of multiple semicircles, indicative of significant polarization effects, implies that several relaxation processes occur, leading to a reduction in the associated conduction loss over the frequency range.57,58
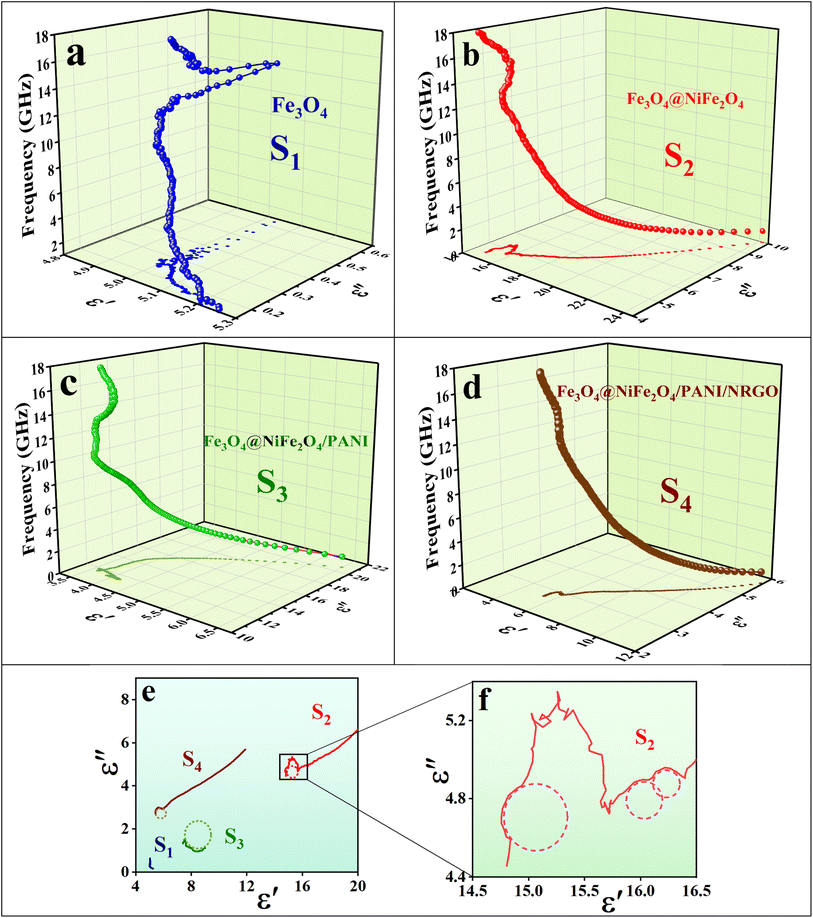 | ||
| Fig. 17 3D Cole–Cole curves (a–d), 2D Cole–Cole curves of all samples (e), and the 2D Cole–Cole curve for S2 (f). | ||
Fig. 19 compares the absorption parameters from our study (S2 and S4) with those from related research, including RLmin, EAB, and absorption thickness.
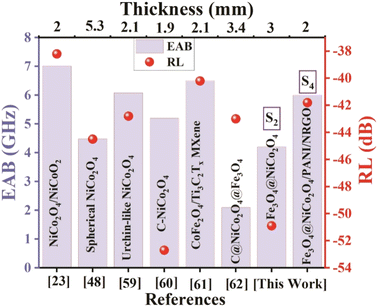 | ||
| Fig. 19 Benchmarking of microwave absorption performance in this work (S2 and S4) against reported studies.23,48,59–62 | ||
4.2 Radar cross-section (RCS)
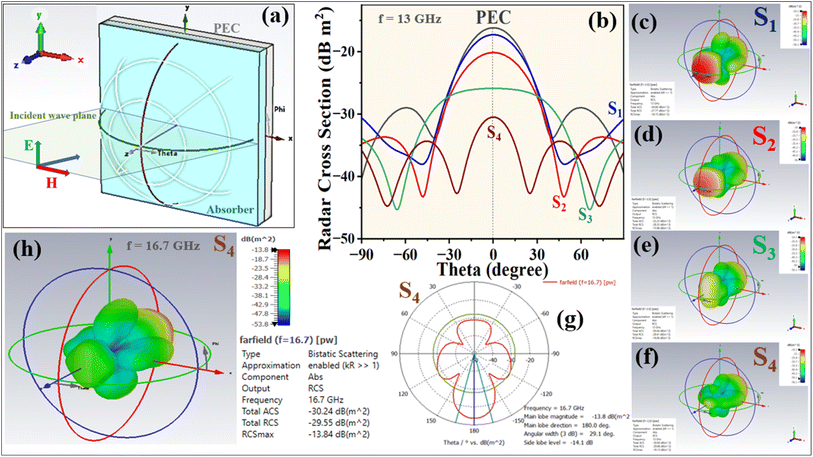
In radar technology applications, the microwave attenuation capabilities of the absorbers were evaluated by assessing the RCS values of a metal back sheet model—a perfect electric conductor (PEC) coated with the prepared samples—using CST simulation software. The RCS value (σ) is given by the following equation (Eq. 14):2,7,63,64σ(dBm2) = 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) log(4πS|Es/Ei|2/λ2) log(4πS|Es/Ei|2/λ2) | (14) |
The PEC board was defined and positioned within a 30 × 30 × 2.4 mm3 environment. Following the simulation, 3D color maps and 1D RCS values ranging from −90° to 90° are obtained and are presented in Fig. 20. When electromagnetic waves are directed at the PEC coated with the samples, the resulting RCS values are lower than those of the pristine PEC. The scattering of the incident wave is significantly reduced for some samples after applying the absorbers. Fig. 20(a) illustrates the schematic of the RCS simulated model. Fig. 20(b–f) display the RSC results at 13 GHz for all samples, while Fig. 20(g and h) shows the corresponding results for the aerogels at 16.7 GHz, all at ϕ = 90°. This sequence closely aligns with the RL values at 13 GHz across all four nanocomposites and underscores the exceptional performance of the (S4) aerogels at 16.7 GHz with a thickness of 2.4 mm (see Fig. 9). The simulation results indicate that the S4 aerogels have an improved ability to dissipate electromagnetic waves, effectively reducing microwave scattering. Consequently, our prepared aerogels demonstrate strong potential for practical applications involving electromagnetic wave management.
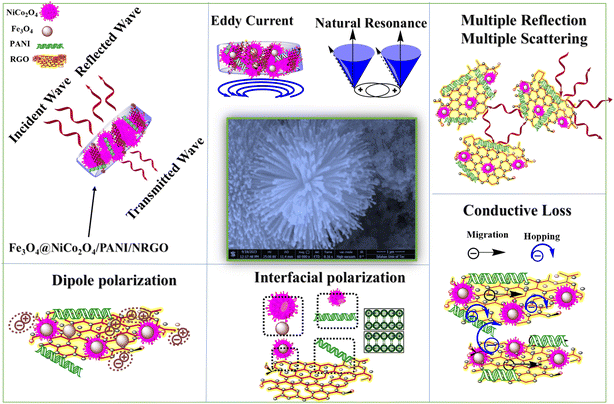
Our nanocomposites and aerogels consist of components with varying conductivity, permittivity, and permeability. The accumulation of charges at the heterojunction interfaces between these different components, combined with the numerous defects and functional groups in NRGO sheets, leads to dipole polarization. The integration of PANI and NRGO forms a conductive network that enhances the propagation of electromagnetic waves. Conductive loss mainly results from electron migration within these networks and electron hopping between different NRGO sheets. Additionally, magnetic components generate eddy currents when exposed to alternating electromagnetic waves and exhibit ferromagnetic resonance in the GHz frequency range. Fig. 21 illustrates a schematic diagram highlighting various loss mechanisms, including dipole polarization due to defect-induced dipole moments, interfacial polarization at heterogeneous boundaries, conduction loss, multiple reflections, and magnetic loss in Fe3O4@NiCo2O4/PANI/RGO (S4) aerogels.
5 Conclusion
In summary, four materials were synthesized using hydrothermal and in situ polymerization methods: (S1) Fe3O4, (S2) Fe3O4@NiCo2O4, (S3) Fe3O4@NiCo2O4/PANI nanocomposites, and (S4) Fe3O4@NiCo2O4/PANI/NRGO aerogels. The S2 nanocomposite exhibits exceptional microwave absorption performance, achieving a minimum reflection loss (RLmin) of −50.9 dB at 5.7 GHz and an effective absorption bandwidth (EAB) of 4.2 GHz at a matching thickness of 3 mm. Notably, S2 maintains an RLmin below −10 dB up to 15.0 GHz across various thicknesses, demonstrating strong electromagnetic wave absorption capabilities throughout the S, C, X, and Ku bands. The S4 aerogels feature a unique structural design characterized by a hollow interior and porous architecture, achieving an RLmin = −41.8 dB at 16.7 GHz and the widest EAB of 6 GHz at a matching thickness of 2 mm. They maintain over 90% absorption across a broad bandwidth of 13.3 GHz, effectively covering the C, X, and Ku frequency bands. The synergistic effects of NRGO and PANI contribute to the formation of a robust conductive network within the nanocomposites and aerogels, facilitating microwave dissipation through combined conductive, dielectric, and magnetic loss mechanisms, thereby functioning as efficient broadband absorbers. The multi-scale design of the selected components results in a multi-layered heterogeneous porous structure with a hollow interior, enabling fine-tuning of effective permittivity, permeability, and conductivity. This enhances impedance matching and attenuation properties via synergistic loss mechanisms. Furthermore, CST simulation results corroborate the excellent microwave energy dissipation performance of the prepared nanocomposites and aerogels, underscoring their promising potential for practical applications.List of abbreviations
| DIW: | Deionized water |
| EAB: | Effective absorption bandwidth |
| EDS: | Energy-dispersive X-ray spectroscopy |
| EM: | Electromagnetic |
| FESEM: | Field emission scanning electron microscopy |
| FTIR: | Fourier transform infrared |
| GO: | Graphene oxide |
| MA: | Microwave absorption |
| MAMs: | Microwave-absorbing materials |
| MWCNT: | Multi-walled carbon nanotube |
| PANI: | Polyaniline |
| PEC: | Perfect electric conductor |
| RGO: | Reduced graphene oxide |
| RL: | Reflection loss |
| RT: | Room temperature |
| TEM: | Transmission electron microscopy |
| VSM: | Vibrating-sample magnetometry |
| XRD: | X-ray diffraction |
List of symbols
| c | Speed of the electromagnetic wave |
| f | Frequency of the microwave |
| λ | Wavelength of the microwave |
tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δε δε | Dielectric loss tangent |
tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δμ δμ | magnetic loss tangent |
| t m | Matching thickness |
| Z 0 | Impedance of free space |
| Z in | Input impedance |
| α | Attenuation coefficient |
| ε 0 | Permittivity of free space |
| ε r | Relative permittivity |
| ε′ | Real part of relative permittivity |
| ε′′ | Imaginary part of relative permittivity |
| μ 0 | Permeability of free space |
| μ r | Relative permeability |
| μ′ | Real part of relative permeability |
| μ′′ | Imaginary part of relative permeability |
| σ | Dielectric conductivity |
Data availability
All data generated or analyzed during this study are included in this article.Conflicts of interest
The authors declare no competing interests.References
- F. Jamadi, J. Seyed-Yazdi, F. Ebrahimi-Tazangi and S. M. Hosseiny, The impact of RGO and MWCNT/RGO on the microwave absorption of NiFe2O4@Fe3O4 in the presence or absence of PANI, J. Mater. Chem. A, 2024, 12, 32981–33002, 10.1039/D4TA04128C.
- J. Qiu, X. Liu, C. Peng, S. Wang, R. Wang and W. Wang, Porous metal microsphere M@C-rGO (metal = Mn, Fe, Co, Ni, Cu) aerogels with high low-frequency microwave absorption, strong thermal insulation and superior anticorrosion performance, J. Mater. Chem. A, 2024, 12, 21997, 10.1039/d4ta04051a.
- W. Yan, J. Luo, Y. Li, M. Liu, Y. Wu, Z. Dai and X. Li, Construction of CoO/Co9S8/NC composites with low-frequency and broadband electromagnetic wave absorption, Carbon, 2024, 228, 119338, DOI:10.1016/j.carbon.2024.119338.
- R. Islam, Y. Sood, H. Mudila, A. Ohlan and A. Kumar, Microwave absorbing properties of polypyrrole-based 2D nanocomposites, J. Mater. Chem. A, 2024, 12, 31004–31027, 10.1039/D4TA05676K.
- Y. Lin, X. Zhou, Y. Wang, J. Hou, D. Wang, G. Tong and Yu Cao, Progress of MOFs composites in the field of microwave absorption, Carbon, 2025, 238, 120241, DOI:10.1016/j.carbon.2025.120241.
- J. Wu, T. Wang and J. Kong, Microwave absorption properties of Ni/porous carbon fiber composites derived from flax fiber templates, Carbon, 2025, 238, 120305, DOI:10.1016/j.carbon.2025.120305.
- S. Wang, X. Zhang, S. Hao, J. Qiao, Z. Wang, L. Wu, J. Liu and F. Wang, Nitrogen-Doped Magnetic-Dielectric-Carbon Aerogel for High-Efficiency Electromagnetic Wave Absorption, Nano-Micro Lett., 2024, 16, 16, DOI:10.1007/s40820-023-01244-w.
- X. Wang, X. Han, J. Liu, G. Sun, Y. Feng, Y. Zhang, M. Ma, Z. Guo and Y. Li, Research progress of NiCo2O4-based composites in microwave absorption: a review, J. Mater. Sci., 2025, 60, 3–31, DOI:10.1007/s10853-024-10482-1.
- X. Zeng, C. Zhao, Y. Yin, T. Nie, N. Xie, R. Yu and G. D. Stucky, Construction of NiCo2O4 nanosheets-covered Ti3C2Tx MXene heterostructure for remarkable electromagnetic microwave absorption, Carbon, 2022, 193, 26e34, DOI:10.1016/j.carbon.2022.03.029.
- Y. Fu, W. Wang, H. Miao, Y. Cui, Y. Chen, Z. Wang, G. Xiao, B. Xue and W. Jiang, Exploration of microwave absorptivity and catalysis of CMF@Co-MoS2 for microwave-initiated depolymerization of lignin, Chem. Eng. J., 2024, 501, 157591, DOI:10.1016/j.cej.2024.157591.
- C. Lai, X. Qu, H. Zhao, S. W. Hong and L. Kwan, Improved performance in asymmetric supercapacitors utilized by dual ion-buffering reservoirs based on honeycomb-structured NiCo2O4 and 3D rGO-PPy aerogels, Appl. Surf. Sci., 2022, 586, 152847, DOI:10.1016/j.apsusc.2022.152847.
- X. Li, J. Feng, Y. Du, J. Bai, H. Fan, H. Zhang, Y. Peng and F. Li, One-pot synthesis of CoFe2O4/graphene oxide hybrids and their conversion into FeCo/graphene hybrids for lightweight and highly efficient microwave absorber, J. Mater. Chem. A, 2015, 3, 5535, 10.1039/c4ta05718j.
- Z. Su, W. Zhang, J. Lu, L. Tian, S. Yi, Y. Zhang, S. Zhou, Bo Niu and D. Long, Oxygen-vacancy-rich Fe3O4/carbon nanosheets enabling high-attenuation and broadband microwave absorption through the integration of interfacial polarization and charge-separation polarization, J. Mater. Chem. A, 2022, 10, 8479, 10.1039/d2ta00080f.
- M. Ma, Y. Bi, Z. Jiao, J. Yue, Z. Liao, Y. Wang, Y. Ma and W. Huang, Facile fabrication of metal–organic framework derived Fe/Fe3O4/FeN/N-doped carbon composites coated with PPy for superior microwave absorption, J. Colloid Interface Sci., 2022, 608, 525–535, DOI:10.1016/j.jcis.2021.09.169.
- X. Shi, Z. Wu, Z. Liu, J. Lv, Z. Zi and R. Che, Interface engineering in the hierarchical assembly of carbon-confined Fe3O4 nanospheres for enhanced microwave absorption, J. Mater. Chem. A, 2022, 10, 8807, 10.1039/d1ta11005e.
- T. Yan, XiC. Ye, E. He, G. Qi, Y. Wang, Y. Ye and H. Wu, GR-Fe3O4/PLA 3D printing composite materials with excellent microwave absorption properties, J. Alloys Compd., 2024, 972, 172799, DOI:10.1016/j.jallcom.2023.172799.
- Na Chen, X.-F. Pan, Z.-J. Guan, Ya-J. Zhang, K.-J. Wang and J.-T. Jiang, Flower-like hierarchical Fe3O4-based heterostructured microspheres enabling superior electromagnetic wave absorption, Appl. Surf. Sci., 2024, 642, 158633, DOI:10.1016/j.apsusc.2023.158633.
- X. Huang, J. Wei, Y. Zhang, B. Qian, Q. Jia, J. Liu, X. Zhao and G. Shao, Ultralight magnetic and dielectric aerogels achieved by Metal–Organic Framework initiated gelation of graphene oxide for enhanced microwave absorption, Nano-Micro Lett., 2022, 14(1), 107, DOI:10.1007/s40820-022-00851-3.
- S. Li, T. Ma, Z. Chai, Z. Zhang, M. Zhu, X. Tang, X. Zhao, Y. Lu, Q. Lan, Z. Wang, F. He, Z. Wang and T. Liu, Graphene-based magnetic composite foam with hierarchically porous structure for efficient microwave absorption, Carbon, 2023, 207, 105–115, DOI:10.1016/j.carbon.2023.02.066.
- X. Xu, F. Ran, Z. Fan, Z. Cheng, T. Lv, L. Shao and Y. Liu, Bimetallic Metal–Organic Framework-derived pomegranate-like nanoclusters coupled with CoNi-doped graphene for strong wideband microwave absorption, ACS Appl. Mater. Interfaces, 2020, 12(15), 17870–17880, DOI:10.1021/acsami.0c01572.
- F. Zhang, N. Li, J.-F. Shi, L. Xu, Li-C. Jia, Y.-Yi Wang and D.-X. Yan, Recent progress on carbon-based microwave absorption materials for multifunctional applications: A review, Composites, Part B, 2024, 283, 111646, DOI:10.1016/j.compositesb.2024.111646.
- J. Yan, W. Bai, Ce Cui, S. Jiang, W. Wang, H. Tang and R. Guo, Metal–organic framework-derived lilac flower-like CoNiZnO@nitrogen-doped carbon composites via a trapping microwave strategy for efficient absorption, J. Mater. Chem. A, 2024, 12, 28794, 10.1039/d4ta05222f.
- T. Hu, Di Lan, J. Wang, X. Zhong, G. Bu and P. Yin, Construction of NiCo2O4/NiCoO2 co-embedded porous bio-carbon with rich heterogeneous interfaces for excellent bacteriostatic microwave radiation protection, Carbon, 2025, 232, 119798, DOI:10.1016/j.carbon.2024.119798.
- P. B. Li, Y. Z. Zhao, Y. Zhao, J. F. Yan, H. T. Zhao, W. Zhao, J. N. Yun, C. Chen, Z. H. Deng and Z. Y. Zhang, Trimetallic prussian blue analogue derived FeCo/FeCoNi@NPC composites for highly efficient microwave absorption, Composites, Part B, 2022, 246, 110268, DOI:10.1016/j.compositesb.2022.110268.
- M. Darvishi, M. Jamali-Paghaleh, F. Jamali-Paghaleh and J. Seyed-Yazdi, Two methods for microwave irradiation synthesis of TiO2–ZnO–graphene ternary hybrids with enhanced photocatalytic activity, Mater. Res. Express, 2017, 4, 016501, DOI:10.1088/2053-1591/aa52cf.
- Z. Çıplak and N. Yıldız, Ag@Fe3O4 nanoparticles decorated NrGO nanocomposite for supercapacitor application, J. Alloys Compd., 2023, 941, 169024, DOI:10.1016/j.jallcom.2023.169024.
- A. Henaish, B. I. Salem, T. M. Meaz, Y. A. Alibwaini, A. W. Ajlouni and O. M. Hemeda, et al., Synthesize, characterization, dielectric, linear and nonlinear optical properties of Ni-Al Ferrite/PANI nanocomposite film, Opt. Mater., 2021, 119, 111397, DOI:10.1016/j.optmat.2021.111397.
- Y. Zhang, J. Liu, Y. Zhang, J. Liu and Y. Duan, Facile synthesis of hierarchical nanocomposites of aligned polyaniline nanorods on reduced graphene oxide nanosheets for microwave absorbing materials, RSC Adv., 2017, 7(85), 54031–54038, 10.1039/c7ra08794b.
- P. Zhou, S. Zhang, W. Zhou, C. Sun, C. Hu, S. Yuan, J. Li, C. Liu, Y. Zhao, Y. Kuang, S. Wang, Q. Zhang, Y. Liu and L. Wang, Hollow engineering of Co/N-doped C@carbon aerogel with hierarchical structure boosting interfacial polarization for ultra-thin microwave absorption and thermal insulation, Chem. Eng. J., 2024, 495, 153561, DOI:10.1016/j.cej.2024.153561.
- M. Huang, L. Wang, Ke Pei, W. You, X. Yu, Z. Wu and R. Che, Multidimension-Controllable Synthesis of MOF-Derived Co@N-Doped Carbon Composite with Magnetic-Dielectric Synergy toward Strong Microwave Absorption, Small, 2020, 2000158, DOI:10.1002/smll.202000158.
- Z. Yao, F. Liu, S. Xu, X. Zhang, C. Rong, Z. Xiong, J. Yuan, Yu Yi, X. Zhu and Y. Huajun, Facile synthesis of La2O3/Co@N-doped carbon nanotubes via Prussian blue analogues toward strong microwave absorption, Carbon, 2022, 196, 763–773, DOI:10.1016/j.carbon.2022.05.041.
- J. Shen, Q. Wang, K. Zhang, S. Wang, Li Lu, S. Dong, S. Zhao, J. Chen, R. Sun, Y. Wang and Z. Jian, Wenzhi ZhangFlexible carbon cloth based solid-state supercapacitor from hierarchical holothurian-morphological NiCo2O4@NiMoO4/PANI, Electrochim. Acta, 2019, 320, 134578, DOI:10.1016/j.electacta.2019.134578.
- B. Dai, Y. Ma, S. Feng, H. Wang, M. Ma, J. Ding and X. Yin, et al., Fabrication of one-dimensional M (Co, Ni)@polyaniline nanochains with adjustable thickness for excellent microwave absorption properties, J. Colloid Interface Sci., 2022, 627, 113–125, DOI:10.1016/j.jcis.2022.06.137.
- H. Gu, J. Guo, M. A. Khan, D. P. Young, T. D. Shen, S. Wei and Z. Guo, Magnetoresistive polyaniline–silicon carbide metacomposites: plasma frequency determination and high magnetic field sensitivity, Phys. Chem. Chem. Phys., 2016, 18, 19536–19543, 10.1039/C6CP02694J.
- X. Liang, X. Xu, Z. Man, B. Quan, B. Sun, J. Chen, W. Gu and G. Ji, Zinc oxide/nanoporous carbon hybrid materials derived from metal–organic frameworks with different dielectric and absorption performances, Inorg. Chem. Front., 2019, 6, 2521, 10.1039/c9qi00648f.
- W. Qi, P. Zhang, X. Guo, W. Jiang, X. Tao, P. K. Shen and Z. Q. Tian, Atomic spin engineering of Fe–N–C by axial chlorine-ligand modulation for lightweight and efficient electromagnetic wave absorption, J. Colloid Interface Sci., 2025, 692, 137464, DOI:10.1016/j.jcis.2025.137464.
- K. Luo, S. Zhang, Y. Liu, H. Wu, X. Fan, Y. Cao and J. Huang, Hierarchical architecture bioinspired CNTs/CNF electromagnetic wave absorbing materials, Carbon, 2023, 207, 198–206, DOI:10.1016/j.carbon.2023.03.024.
- Li Xiao, H. Zhou, J. Zhang, X. Zhang, M. Li, J. Zhang, M. A. Darwish, T. Zhou, S.-K. Sun, e L. Xie and Di Zhou, Construction of shell-like carbon superstructures through anisotropically oriented self-assembly for distinct electromagnetic wave absorption, J. Mater. Chem. A, 2024, 12, 4057, 10.1039/d3ta06822f.
- Z. Xiang, C. Huang, Y. Song, B. Deng, X. Zhang, X. Zhu, D. Batalu, O. Tutunaru and W. Lu, Rational construction of hierarchical accordion-like Ni@porous carbon nanocomposites derived from metal-organic frameworks with enhanced microwave absorption, Carbon, 2020, 167, 364e377, DOI:10.1016/j.carbon.2020.06.015.
- W. Yan, J. Luo, Y. Li, M. Liu, Y. Wu, Z. Dai and X. Li, Construction of CoO/Co9S8/NC composites with low-frequency and broadband electromagnetic wave absorption, Carbon, 2024, 228, 119338, DOI:10.1016/j.carbon.2024.119338.
- Y. Cai, R. Zhou, S. Zhang, M. Qiao, Y. Cheng, Y. Zhu, H. Luo, P. Chen, X. Li and Fu Chen, Constructing conductively hierarchical Co3Fe7/NC@MWCNTs composites by binary MOF-on-MOF hybrids: a promising approach for boosting microwave absorption, Chem. Eng. J., 2025, 507, 160896, DOI:10.1016/j.cej.2025.160896.
- Z. Yao, S. Xu, X. Zhang, J. Yuan, C. Rong, Z. Xiong, X. Zhu, Yi Yu, H. Yu, S. Kang and F. Kuang, CuCo Nanocube/N-Doped Carbon Nanotube Composites for Microwave Absorption, ACS Appl. Nano Mater., 2023, 6, 1325–1338, DOI:10.1021/acsanm.2c04972.
- F. Ebrahimi-Tazangi and J. Seyed-Yazdi, Seyedeh Hoda Hekmatara, α-Fe2O3@CoFe2O4/GO nanocomposites for broadband microwave absorption by surface/interface effects, J. Alloys Compd., 2022, 900, 163340, DOI:10.1016/j.jallcom.2021.163340.
- J. Wang, Y. Wang, J. Cheng, Y. Fu, Li Yao, W. Nie, J. Wang, B. Liu, D. Zhang, G. Zheng and M. Cao, Abundant vacancies induced high polarization-attenuation effects in flower-like WS2 microwave absorbers, J. Mater. Sci. Technol., 2024, 194, 193–202, DOI:10.1016/j.jmst.2024.01.085.
- Z. Huang, J. Cheng, H. Zhang, Y. Xiong, Z. Zhou, Q. Zheng, G. Zheng, D. Zhang and M. Cao, High-performance microwave absorption enabled by Co3O4 modified VB-group laminated VS2 with frequency modulation from S-band to Ku-band, J. Mater. Sci. Technol., 2022, 107, 155–164, DOI:10.1016/j.jmst.2021.08.005.
- D. Wu, Y. Wang, S. Deng, Di Lan, Z. Xiang and Q. He, Heterostructured CoFe@N-doped carbon porous polyhedron for efficient microwave absorption, Nano Res., 2023, 16, 1859–1868, DOI:10.1007/s12274-022-5341-z.
- J. Zheng, J. Zheng, X. Zhang, P. Yue, W. Cai, H. Chen, L. Qiao, Y. Ying, J. Yu, J. Li, W. Li and S. Che, Microwave Absorption Properties and Characterization of Porous Permanent/Soft Magnetic Composite Iron Nitride with Heterogeneous Interfaces, Adv. Funct. Mater., 2025, 2424988, DOI:10.1002/adfm.202424988.
- X. Zhou, Z. Jia, X. Zhang, B. Wang, X. Liu, B. Xu, L. Bi and G. Wu, Electromagnetic wave absorption performance of NiCo2X4 (X = O, S, Se, Te) spinel structures, Chem. Eng. J., 2021, 420, 129907, DOI:10.1016/j.cej.2021.129907.
- F. Ebrahimi-Tazangi, S. Hoda Hekmatara and J. Seyed-Yazdi, Remarkable microwave absorption of GO-SiO2/Fe3O4via an effective design and optimized composition, J. Alloys Compd., 2021, 854, 157213, DOI:10.1016/j.jallcom.2020.157213.
- Z. Jiao, J. Hu, M. Ma, Y. Liu, J. Zhao, X. Wang, S. Luan and L. Zhang, One-dimensional core–shell CoC@CoFe/C@PPy composites for high-efficiency microwave absorption, J. Colloid Interface Sci., 2023, 650, 2014–2023, DOI:10.1016/j.jcis.2023.07.072.
- J. Li, Di Lan, Y. Cheng, Z. Jia, P. Liu, X. Shi, H. Guo, A. Feng, X. Feng, G. Wu and P. Yin, Constructing mixed-dimensional lightweight magnetic cobalt-based composites heterostructures: An effective strategy to achieve boosted microwave absorption and self-anticorrosion, J. Mater. Sci. Technol., 2024, 196, 60–70, DOI:10.1016/j.jmst.2024.02.016.
- Y. Bai, L. He, P. Lv, C. Ge, H. Xu and X. Zhang, Impedance matched (hydroxylated nano-BN/reduced graphene oxide)@Fe3O4/polyaniline composite for efficient microwave absorption and thermal management, Mater. Chem. Phys., 2023, 295, 127193, DOI:10.1016/j.matchemphys.2022.127193.
- S. Wang, X. Zhang, S. Hao, J. Qiao, Z. Wang, L. Wu, J. Liu and F. Wang, Nitrogen-doped magnetic-dielectric-carbon aerogel for high-efficiency electromagnetic wave absorption, Nano-Micro Lett., 2024, 16, 16, DOI:10.1007/s40820-023-01244-w.
- F. Ebrahimi-Tazangi, A. Maazallahi, P. Livreri and J. Seyed-Yazdi, MnFe2O4/Fe3O4/PANI/rGO heterogeneous nanocomposites: outstanding dual-broadband nano-coating microwave absorbers, J. Alloys Compd., 2024, 1002, 175149, DOI:10.1016/j.jallcom.2024.175149.
- J. Lu, X. Di, M. Yuan, B. Sun, C. Zhou, Y. Luo, Y. Chen and H. Zou, In-situ construction of phase-separated interconnected PVA@RGO frameworks using dual-crosslinking/reduction steps for broadband microwave absorption and enhanced structural performance of aramid honeycombs, Carbon, 2024, 228, 119360, DOI:10.1016/j.carbon.2024.119360.
- Bo Dai, F. Dong, H. Wang, Y. Qu, J. Ding, Y. Ma, M. Ma and T. Li, Fabrication of CuS/Fe3O4@polypyrrole flower-like composites for excellent electromagnetic wave absorption, J. Colloid Interface Sci., 2023, 634, 481–494, DOI:10.1016/j.jcis.2022.12.029.
- Z. Liao, M. Ma, Z. Tong, R. Wang, Y. Bi, Y. Chen, K. L. Chung and Y. Ma, Fabrication of ZnFe2O4/C@PPy composites with efficient electromagnetic wave absorption properties, J. Colloid Interface Sci., 2021, 602, 602–611, DOI:10.1016/j.jcis.2021.06.042.
- S. Cheng, D. Sheng, S. Mukherjee, W. Dong, Y. Huang, R. Cao, A. Xie, R. A. Fischer and W. Li, Carbon nanolayer-mounted single metal sites enable dipole polarization loss under electromagnetic field, Nat. Commun., 2024, 15, 9077, DOI:10.1038/s41467-024-53465-1.
- H. Wu, M. Qin and L. Zhang, NiCo2O4 constructed by different dimensions of building blocks with superior electromagnetic wave absorption performance, Composites, Part B, 2020, 182, 107620, DOI:10.1016/j.compositesb.2019.107620.
- C. Han, M. Zhang, W.-Q. Cao and M.-S. Cao, Electrospinning and in-situ hierarchical thermal treatment to tailor C-NiCo2O4 nanofibers for tunable microwave absorption, Carbon, 2021, 171, 953–962, DOI:10.1016/j.carbon.2020.09.067.
- Na Tian, C. Wang and C. You, Synthesis of nanospherical CoFe2O4/Ti3C2Tx MXene composites with enhanced microwave absorbing performance, J. Alloys Compd., 2023, 967, 171796, DOI:10.1016/j.jallcom.2023.171796.
- S. Wei, X. Wang, B. Zhang, M. Yu, Y. Zheng, Y. Wang and J. Liu, Preparation of hierarchical core-shell C@NiCo2O4@Fe3O4 composites for enhanced microwave absorption performance, Chem. Eng. J., 2017, 314, 477–487, DOI:10.1016/j.cej.2016.12.005.
- Z. Lin, H. Xiao, Y. Chen and H. Cai, Petaloid magnetic bimetallic CoNi/CNTs carbides self-assembled with MXene for exceptional electromagnetic wave absorption performance, J. Alloys Compd., 2025, 1020, 179368, DOI:10.1016/j.jallcom.2025.179368.
- H. Dong, S. Gao, C. Yu, Z. Wang, H. Zhang, Y. Huang, T. Zhao and Y. Li, Design and performance of 3D-Printed ABS@rGO/CF/CeO2 composites for microwave absorption and mechanical strength, Chem. Eng. J., 2024, 499, 156696, DOI:10.1016/j.cej.2024.156696.
| This journal is © The Royal Society of Chemistry 2025 |