Scalable, thermally stabilized MOF-graphene fibers with hierarchical porosity for high-performance energy storage devices
Narges
Nobakht
*ab,
Mohammad
Hosseini
 a,
Seyyed Ahmad
Etghani
ac,
Mohamad Ali
Sanjari Shahrezaei
a,
Ali
Sajedi-Moghaddam
a,
Seyyed Ahmad
Etghani
ac,
Mohamad Ali
Sanjari Shahrezaei
a,
Ali
Sajedi-Moghaddam
 *a and
Seyed Hamed
Aboutalebi
*a and
Seyed Hamed
Aboutalebi
 *ad
*ad
aCondensed Matter National Laboratory, Institute for Research in Fundamental Sciences, Tehran 19395-5531, Iran. E-mail: hamedaboutalebi@ipm.ir; nargesnobakht@ipm.ir; a.sajedi@ipm.ir
bBattScout Company, London Road, Bishopdown, Salisbury, England SP1 3, UK
cSchool of Biological Sciences, Institute for Research in Fundamental Sciences, Tehran, 19395-5746, Iran
dSchool of Quantum Physics & Matter, Institute for Research in Fundamental Sciences, Tehran, 19395-5531, Iran
First published on 29th July 2025
Abstract
Next-generation energy storage devices require electrodes that combine high charge storage capacity, mechanical robustness, and scalable fabrication. Here, we present a new class of thermally stabilized, hierarchically porous hybrid fibers integrating copper benzene tricarboxylate (Cu-BTC) metal–organic frameworks (MOFs) with liquid crystalline graphene oxide, addressing long-standing challenges in MOF processing for fiber-based architectures. Incorporating Keggin-type phosphotungstic acid polyoxometalate enhances the thermal stability of the Cu-BTC framework, enabling wet spinning and thermal reduction to produce conductive MOF-reduced graphene oxide hybrid fibers. In addition to providing high surface area and hierarchical porosity, the MOF structure introduces redox-active sites that contribute to pseudocapacitance, further improving charge storage. The resulting fibers exhibit outstanding electrochemical performance in a symmetric two-electrode configuration, delivering a gravimetric capacitance of 476.9 F g−1 at 0.77 A g−1 and maintaining 307.7 F g−1 even at a high current density of 7.69 A g−1, demonstrating remarkable rate capability. The fibers also show excellent cycling stability, with 96.7% capacitance retention over 4000 cycles. Moreover, the hybrid device achieves an areal energy density of 29.9 μWh cm−2 at a power density of 0.35 mW cm−2, and retains 19.29 μWh cm−2 even at a higher power density of 3.47 mW cm−2, significantly outperforming many previously reported fiber-based supercapacitors. Beyond electrochemical function, the fibers demonstrate exceptional mechanical strength (Young's modulus >42 GPa), offering a rare combination of durability and performance. This work establishes a versatile platform for integrating MOFs into flexible, high-performance, and scalable fiber-based energy storage devices.
Introduction
Supercapacitors (SCs) are critical for sustainable and efficient energy storage, with applications ranging from load leveling in electric vehicles to powering next-generation wearable devices.1–4 Among them, electric double-layer capacitors (EDLCs) have attracted significant attention due to their high power density, fast charge–discharge rates, and excellent cycling stability. These characteristics arise from their non-faradaic energy storage mechanism.5,6 However, the limited energy density of EDLCs, attributed to their purely electrostatic charge storage, restricts their widespread application in energy-demanding devices. Traditional efforts to improve EDLCs have focused on optimizing carbon-based materials, such as reduced graphene oxide (rGO) and carbon nanotubes, through advanced processing and synthetic techniques.7–11 However, carbon-based materials often lack well-defined hierarchical pore structures that integrate micropores for charge storage and mesopores for ion transport, limiting their performance.12–14 This limitation is particularly evident in high-performance two-dimensional (2D) materials like graphene, which often suffer from slow charging dynamics and scalability issues due to their confined geometries.8,15–18Metal–organic frameworks (MOFs) have recently gained attention as a compelling alternative to carbon-based materials due to their tunable porosity, exceptionally high surface areas (2000–8000 m2 g−1),19 and the ability to integrate micropores and mesopores within a single structure.20–23 Combining tunable organic ligands and functional metal ions enables precise control over MOF synthesis, allowing for tailored pore sizes and structures optimized for specific applications.22,24–27 Unlike carbon-based materials, MOFs can maintain highly ordered porosity and ultrathin walls, resembling those of 2D materials, while also facilitating the simultaneous diffusion and trapping of ions of different sizes. This dual-pore structure is advantageous for designing multifunctional SCs capable of operating with various electrolytes. Micropores enhance capacitance by providing a large surface area for charge storage, while mesopores improve charge transfer kinetics.
Despite their advantages, MOFs suffer from poor electrical conductivity, chemical instability in common electrolytes, and thermal sensitivity during processing, limiting their practical application in SCs.20,28–32 As a result, the theoretical promise of MOFs—offering capacitance values exceeding those of 2D materials—has yet to be realized in practical devices. Although MOF-based composites, such as MOF-graphene,33 and MOF-CNTs,34 have demonstrated high performance in three-electrode configurations, their performance in practical two-electrode configurations remains subpar. For instance, the best-reported performance for 2D conductive MOFs in a two-electrode configuration is 110 F g−1 at a low current density of 0.05 A g−1, which drops to below 40 F g−1 at a more practical rate of 1 A g−1—a value comparable to commercial carbon-based materials.14 Bridging this performance gap requires innovative strategies to improve ion and electron transport in MOF-based electrodes.
Transforming MOF-based materials into fiber-shaped architectures represents a promising strategy to address these challenges. Fiber-shaped electrode materials offer advantages such as enhanced ion diffusion pathways, mechanical flexibility, and compatibility with flexible and wearable devices. However, despite their potential, no studies have reported the fabrication of fiber-shaped MOF-graphene hybrid structures. Such hybrid architectures are anticipated to synergistically combine the high conductivity of rGO with the large surface area and tunable porosity of MOFs, resulting in improved charge storage capacity, faster ion transport, and enhanced mechanical robustness.
Here, we report the first thermally stabilized, hierarchically porous MOF-graphene hybrid fibers for high-performance SC applications. By incorporating Keggin-type phosphotungstic acid (HPW) into a copper benzene tricarboxylate (Cu-BTC, HKUST-1) framework, we enhance the thermal stability of the MOF, enabling its integration with liquid crystalline graphene oxide (LCGO) dispersions. The stabilized MOF-GO mixture is processed via wet spinning to form continuous, unidirectional fibers that retain hierarchical porosity. Subsequent thermal reduction transforms GO into rGO, imparting electrical conductivity without compromising the MOF structure. The resulting hybrid fibers exhibit high capacitance, long-term cycling stability, and superior mechanical properties, positioning them as scalable and versatile electrode materials for next-generation fiber-based energy storage devices.
Results and discussion
Thermal stabilization of Cu-BTC MOF via polyoxometalate (POM) incorporation
MOFs are widely recognized for their tunable porosity and high surface areas.21,22 Cu-BTC, in particular, uniquely combines high surface area, redox-active copper sites and benign, water-based synthesis—all critical for scalable fiber SCs. Its redox-active copper centers contribute pseudocapacitance through reversible Cu2+/Cu+ transitions, directly enhancing energy density. In addition to these functional advantages, Cu-BTC offers practical benefits, including (i) being a cost-effective precursor, (ii) straightforward aqueous synthesis under ambient conditions, and (iii) established scalability for fiber processing. In addition, Cu-BTC frameworks offers a high surface area exceeding 1500 m2 g−1 and a hierarchical porous structure, making them suitable for encapsulating POMs.35 The Cu-BTC framework comprises three distinct pores: the smallest, an octahedral cavity (∼6 Å in diameter), is located at the center of [Cu12(C9H3O6)8(H2O)12] units, while the other two pores, formed by the periodic arrangement of these units, measure approximately 11 Å and 15 Å. The smallest pore is hydrophobic due to its benzyl-rich core and the saturation of all metal coordination sites, whereas the larger pores are hydrophilic due to the presence of coordinated water molecules interacting with accessible Cu sites. The largest of these pores remains unoccupied in pristine Cu-BTC, providing a potential site for incorporating HPW.36 However, the poor thermal stability of Cu-BTC, like other MOFs, often limits its use in fiber formation and coagulation-based processing.37,38To enhance the thermal stability of Cu-BTC, we introduced HPW into the framework, leading to the spontaneous formation of an HPW-integrated Cu-BTC MOF, hereafter referred to as PM, under ambient conditions (Fig. 1a and S1). The presence of HPW immobilizes benzene-1,3,5-tricarboxylate linkers, restricting their lateral movement and preventing structural reorientation.39 This integration significantly improves the thermal resilience of the MOF, effectively stabilizing its architecture even after surfactant removal.
Thermogravimetric analysis (TGA) shows that pristine Cu-BTC exhibits significant weight loss beginning at 200 °C, with almost complete degradation by 350 °C (Fig. 1b). In contrast, PM retains over 80% of its initial weight at 300 °C, demonstrating substantial thermal stability. This stability exceeds the temperature typically required for the thermal reduction of GO (200–220 °C),8 making PM suitable for composite fiber fabrication. The improved thermal stability of PM is attributed to synergistic interactions between HPW and the Cu-BTC framework. HPW forms electrostatic interactions with Cu2+ sites, reducing linker mobility and enhancing framework rigidity.40 Additionally, HPW acts as a molecular stabilizer, preventing pore collapse and healing structural defects.41 Its high intrinsic thermal stability further delays decomposition, contributing to the overall robustness of the composite.42
Furthermore, the surface chemistry of PM was analyzed using Fourier transform infrared (FTIR) spectroscopy, as shown in Fig. 1c. The spectrum exhibits characteristic peak at 727 cm−1, which are attributed to Cu–O stretching vibration.43 Additional bands appear at 796 and 898 cm−1, corresponding to W–O–W vibrations, while a distinct peak at 982 cm−1 is assigned to W![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretching. A strong band at 1080 cm−1 is ascribed to the PO4 group of the HPW.44 The spectral region between 1300 and 1700 cm−1 is associated with the vibrations of carboxylate ligands, confirming the successful coordination of BTC to the copper centers.45 These observations collectively validate the presence of both the Cu-BTC and the HPW indicating the structural integrity of PM.
O stretching. A strong band at 1080 cm−1 is ascribed to the PO4 group of the HPW.44 The spectral region between 1300 and 1700 cm−1 is associated with the vibrations of carboxylate ligands, confirming the successful coordination of BTC to the copper centers.45 These observations collectively validate the presence of both the Cu-BTC and the HPW indicating the structural integrity of PM.
Nitrogen adsorption–desorption measurements confirm that the PM superstructure maintains high porosity. The Brunauer–Emmett–Teller (BET) surface area of PM reaches 1684.7 m2 g−1 (Fig. 1d). Barrett–Joyner–Halenda (BJH) analysis reveals a broad pore size distribution, with dominant peaks at 1.7 nm and 3.9 nm, corresponding to micropores and mesopores, respectively (Fig. 1e). The hierarchical pore structure, combining micro- and mesoporosity, promotes electrolyte accessibility and ion diffusion, critical for enhancing charge storage in SC electrodes.
Fabrication of hybrid fibers and printed structures
Building on the demonstrated thermal stabilization of the MOF structure through POM (HPW) incorporation, the resulting PM superstructure was integrated with LCGO dispersions to enhance processability (Fig. S2 and S3). While the PM structure alone exhibits limited colloidal stability in water,46 the incorporation of HPW—a highly polar POM—significantly increases the hydrophilicity of the Cu-BTC framework. This enhanced hydrophilicity facilitates homogeneous dispersion and effective decoration of large GO monolayer sheets in LCGO dispersions (Fig. S4) through simple mixing. The strong interfacial compatibility between PM and GO arises from matching chemistries and π–π interactions, leading to the formation of a hybrid PMGO structure.Zeta potential measurements confirm the improved colloidal stability of the hybrid PMGO dispersion (Fig. S5). Pristine PM exhibits a relatively low zeta potential of −13.8 mV, indicating poor stability in aqueous media. In contrast, GO sheets display a highly negative zeta potential of −36.6 mV, characteristic of stable dispersions due to strong electrostatic repulsion. The PMGO hybrid achieves an intermediate zeta potential of −29.2 mV, signifying enhanced dispersion stability relative to PM alone. These results confirm that incorporating GO improves the colloidal behavior of the PM structure, facilitating stable hybrid ink formation. The synergy between PM and LCGO not only stabilizes the hybrid dispersion but also enables direction-dependent processability. This hybrid ink benefits from the physical and chemical cohesion of its components, preventing PM aggregation and maintaining mesogen alignment without inducing particle flocculation. Consequently, the system supports advanced processing techniques, including direct ink printing and both wet and dry spinning.
Subsequent thermal treatment facilitates the reduction of GO to rGO, converting PMGO into PMrGO. This reduction step ensures the required electrical conductivity and mechanical integrity for electrode fabrication. The intrinsic direction-dependent properties of printed continuous fiber architectures, stemming from weak out-of-plane transport characteristics, can be exploited to enhance the capacitive performance of MOFs.47–49 Since the thickness of individual fibers, yarns, or printed layers remains constrained, this approach mitigates issues related to slow charge dynamics.8 Electrode mass can be easily increased by printing additional layers or integrating multiple fibers and yarns onto the electrode substrate. However, standalone 3D MOF architectures remain challenging to fabricate, as they typically require coating onto other fibers or direct structural integration.50–52
A promising strategy involves utilizing LCGO dispersions as a matrix to guide the self-assembly of MOFs and GO (Fig. 2). Techniques such as wet spinning or printing enable the fabrication of unidirectional architectures; here, we used a continuous direct printing method into a coagulation bath to form the final structures, as demonstrated in previous studies.8,48,49,52 Photographs of the wet spinning process and mechanical flexibility of the resulting PMGO fibers are shown in Fig. S6. This method eliminates the need for additional cross-linking, enabling 3D self-assembled and aligned microstructures without binders.49 Maintaining a slightly acidic dispersion (pH 3–4) of large GO was critical for preserving pendant multifunctional oxide and sulfate groups on the GO surface. Interactions between the secondary building units of the highly polar PM MOF and these functional groups play a crucial role in minimizing MOF crystal aggregation and improving dispersion. Compared to conventional casting, this approach yields hybrid composites that exhibit enhanced molecular transport and improved accessibility to MOF and rGO active sites, optimizing performance.
Characterization of as-prepared hybrid architectures
The morphology and microstructure of the fabricated products were analyzed using field emission scanning electron microscopy (FESEM), as shown in Fig. 3 and S7. The morphology of the as-prepared PM is presented in Fig. 3a-I, while Fig. 3a-II to IV illustrate the morphology of pristine GO fibers printed using isopropanol as the coagulation bath.The cross-sectional FESEM image (Fig. 3a-IV) reveals a highly porous yet ordered architecture in the GO fibers, a feature attributed to the rapid extraction of water during coagulation. This process promotes the structured alignment of GO sheets.
To enhance the architecture, a PM-first approach was employed, in which PM was integrated with LCGO prior to fiber formation, resulting in PM-decorated GO fibers (Fig. 3b-I to IV). The as-prepared PMGO demonstrates an exfoliated and ordered architecture across its diameter (Fig. S7). The cross-sectional analysis (Fig. 3b-IV) of the printed fibers highlights their porous morphology and a directionally constrained exfoliated arrangement. Notably, lateral cohesion between adjacent GO sheets, facilitated by van der Waals interactions and hydrogen bonding, enhances the structural integrity of both pristine and hybrid fibers. Additionally, energy-dispersive X-ray spectroscopy (EDS) (Fig. S8) confirms the elemental composition of PMGO, detecting elements such as copper (Cu), tungsten (W), phosphorus (P), oxygen (O), and carbon (C). Cu originates from the MOF, while P and W are contributed by HPW, and C is attributed to GO and MOF.
TGA of PMGO was conducted prior to the thermal reduction of GO to rGO (Fig. S9). The integration of GO within the as-prepared architecture reinforced structural integrity up to 300 °C, with a minor weight loss of approximately 5% below 100 °C, likely due to moisture desorption. A standard weight loss of approximately 20% was observed, which is characteristic of the GO reduction process. The final PMGO structure exhibited remarkable thermal stability, with no significant degradation up to 300 °C, surpassing the required temperature range (200–220 °C) for the typical reduction process. To mitigate the re-agglomeration and restacking of graphene sheets while preserving the structural integrity of the architecture, the thermal treatment was carefully controlled within this temperature range. This reduction process endowed the final architecture with electrical conductivity, crucial for energy storage applications, without compromising fiber integrity (Fig. S10).
Post-reduction micrographs (Fig. 3c-I to IV) confirm the structural stability of the reduced fibers, with no observable structural collapse or swelling, thereby demonstrating the effectiveness of the employed reduction method. The presence of protruding graphene sheets in Fig. 3c-IV further emphasizes the uniformity of the reduction process and the robustness of the final architecture.
Building on the structural advantages of the PMrGO architecture, we observed a remarkable Young's modulus of approximately 42.64 GPa—nearly twice that of native rGO fibers. This value significantly exceeds those of cross-linked GO and rGO-based fibers, such as GO fibers coagulated in chitosan (around 22.6 GPa) and calcium chloride (around 20.1 GPa),49 as well as all previously reported GO and rGO fibers (Table S1).8 This enhancement in mechanical strength is attributed to a higher degree of hydrogen bonding, promoted by the acidic conditions used during hybrid fiber formation, leading to stronger material interactions. Additionally, strong coordination bonding between copper centers in the MOF and oxygen-containing functional groups on the GO sheets facilitates more efficient stress transfer across the hybrid interface. The MOF particles also act as rigid spacers that suppress layer restacking and restrict slippage between GO sheets, thereby increasing resistance to deformation.53,54
Raman spectroscopy was employed to investigate the structural evolution of the PMGO hybrid upon thermal reduction to PMrGO. As shown in Fig. 4a, both samples exhibit two prominent Raman bands near 1350 cm−1 and 1592 cm−1, corresponding to the D band (disorder-induced) and G band (graphitic E2g mode) of graphene-based materials, respectively. Notably, the intensity ratio (ID/IG) increases from 0.885 in PMGO to 1.059 in PMrGO after thermal treatment, confirming the successful reduction of GO to rGO. This increase indicates the formation of new graphitic domains with smaller sp2 clusters, which is a typical feature of partially restored graphitic lattices during the reduction of GO.
Besides the D and G bands, additional Raman features were observed below 1000 cm−1, which can be attributed to the vibrational modes of the PM component of the hybrid. Peaks detected in the low-frequency region (<500 cm−1) are associated primarily with lattice vibrations and metal–oxygen stretching modes originating from copper-containing units within the Cu-BTC framework. Meanwhile, peaks appearing in the intermediate-frequency region (500–900 cm−1) arise mainly from vibrational modes of the organic linkers in the benzene-1,3,5-tricarboxylate (BTC) moieties.43 In particular, the observed band near 801 and 899 cm−1 can be assigned to edge-sharing W–Oc–W and W–Oe–W vibrations of the HPW, confirming its presence and structural integrity within the composite.55
Fig. 4b and c present the nitrogen adsorption–desorption isotherms and corresponding BJH pore-size distribution curves for the PMrGO hybrid architecture. The BET analysis reveals a high specific surface area of 2240.2 m2 g−1 for PMrGO, substantially exceeding the surface area of the pristine PM component (1684.7 m2 g−1, see Fig. 2d), reflecting the additional contribution from the rGO network and the synergistic porosity arising from hybridization. The BJH pore-size distribution shown in Fig. 4b indicates dominant peaks at approximately 1.5 nm and 3.4 nm, similar to those observed for the PM component (Fig. 2e), although minor variations in peak positions hint at the subtle effects of incorporating rGO into the hybrid structure.
Furthermore, X-ray diffraction (XRD) analysis of PMrGO after heat treatment at 200 °C reveals that the modified PM framework remains structurally intact (Fig. 4d). The d-spacing distribution includes three distinct regions: 0.3–0.6 nm, 0.8 nm, and 1.5–1.7 nm, characteristic of self-stacked, solvated graphene films known for their high electrochemical capacitance. Notably, this broad range of pore sizes aligns with nitrogen adsorption–desorption isotherm results, where the 1.5 nm peak confirms microporous structures facilitating ion diffusion. It should be noted that gas adsorption techniques cannot resolve pore sizes in the 0.3–0.6 nm range, which are critical for charge storage.8 These limitations underscore the need for complementary characterization methods to evaluate the material's pore architecture fully.
The hierarchical porosity of PMrGO plays a crucial role in electrochemical performance. Larger pores enable rapid electrolyte diffusion, supporting high charge/discharge rates, while the smaller pores enhance charge retention, improving capacitance (Fig. 4e and f). Together, this combination of structural integrity and tunable porosity not only can support superior SC performance but also suggests broader applicability in energy storage systems, such as lithium-ion batteries, where minimal volume expansion and structural durability are critical for long-term cycling stability.
Electrochemical performance of PMrGO hybrid architectures
To systematically enhance the SC performance of the developed materials, we investigated the influence of architecture, composition, and electrolyte environment on the electrochemical behavior of the PMrGO system through cyclic voltammetry (CV) measurements. All electrodes were fabricated with practical thicknesses and tested using commercial-grade current collectors, separators, and electrolytes to ensure relevance to real-world devices.Fig. 5a compares the CV profiles of hybrid fiber architectures with and without MOF, revealing a striking contrast in their electrochemical behavior. The PrGO electrode exhibits a predominantly quasi-rectangular CV profile across various scan rates, indicative of EDLC behavior, with minor deviations suggesting limited pseudocapacitive contributions from redox-active HPW species (Fig. S11). In contrast, the PMrGO electrode shows a significantly distorted CV shape with a higher current response, highlighting enhanced pseudocapacitive contributions resulting from incorporating MOFs. The enhanced charge storage behavior is attributed to the redox-active Cu2+ centers in the Cu-BTC MOF, which undergo reversible faradaic reactions.56,57 The Cu-BTC nodes undergo a fully reversible Cu2+ ⇌ Cu+ redox transition. This rapid, surface-accessible faradaic process supplements the electric-double-layer charge stored on the rGO scaffold, boosting capacitance. Because the rigid MOF lattice is in intimate electrical contact with the conductive rGO network, the Cu centers remain stable during cycling. The synergy of these two charge-storage modes—Cu redox pseudocapacitance plus rGO EDLC—accounts for the high capacitance observed.
The effect of structural format on energy storage performance is shown in Fig. 5b, where fiber-based PMrGO electrodes outperform their non-fiber counterparts at a scan rate of 50 mV s−1. The fiber architecture displays a larger CV area and more pronounced pseudocapacitive behavior due to its aligned and continuous structure. It promotes efficient ion diffusion and electron transport, maximizing the synergistic effect between PM and the rGO matrix. In contrast, the non-fiber composite exhibits a more rectangular CV shape with reduced current response at different scan rates (Fig. S12).
To optimize composition, PMrGO fiber electrodes were fabricated at different PM-to-rGO weight ratios. The 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 weight ratio exhibited the best capacitive behavior, with the largest CV curve area and highest current response, indicating optimal charge storage performance (Fig. 5c). While increasing the PM content to 2
1 weight ratio exhibited the best capacitive behavior, with the largest CV curve area and highest current response, indicating optimal charge storage performance (Fig. 5c). While increasing the PM content to 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 still maintained reasonable performance, further increases beyond this ratio reduced adhesion between the active material and the electrode substrate, causing peeling and electrode failure. This reduction in adhesion is attributed to the decreased relative content of GO, which in our binder-free design also serves as a structural binder, leading to weaker cohesion and reduced mechanical integrity. It should be noted that variation of the PM content in the hybrid structure results in deviation of the CV profiles of the hybrid structure, highlighting the role of the PM structure in inserting the pseudocapacitance in the PMrGO hybrid architecture (Fig. S13).
1 still maintained reasonable performance, further increases beyond this ratio reduced adhesion between the active material and the electrode substrate, causing peeling and electrode failure. This reduction in adhesion is attributed to the decreased relative content of GO, which in our binder-free design also serves as a structural binder, leading to weaker cohesion and reduced mechanical integrity. It should be noted that variation of the PM content in the hybrid structure results in deviation of the CV profiles of the hybrid structure, highlighting the role of the PM structure in inserting the pseudocapacitance in the PMrGO hybrid architecture (Fig. S13).
Fig. 5d shows the CV curves of PMrGO in sodium chloride (NaCl), sulfuric acid (H2SO4), and potassium hydroxide (KOH) at 50 mV s−1, where NaCl exhibits the largest enclosed area, followed by H2SO4 and KOH, reflecting its highest capacitance. This trend is supported by rate capability tests (Fig. 5e), with NaCl maintaining superior capacitance retention at increasing scan rates. This enhanced electrochemical performance can be attributed to preserving redox integrity within the polyoxometalate-modified Cu-BTC framework in NaCl electrolyte. The non-reactive nature of Na+ ions helps maintain the coordination environment of redox-active Cu2+ centers, enabling consistent pseudocapacitive behavior—consistent with previous findings that neutral electrolytes better preserve MOF redox activity by minimizing structural degradation and ionic interference.58 In contrast, protons (H+) and hydroxide ions (OH−) may disrupt metal–ligand interactions or induce framework reconfiguration, reducing faradaic contributions. This observation aligns with reports on Cu-BTC, where aqueous acidic and basic conditions lead to structural degradation due to competitive binding of protons or nucleophilic attack by hydroxide ions at the Cu–O coordination sites. Such interactions destabilize the paddlewheel clusters and collapse the framework, thereby diminishing electrochemical performance.59 Consequently, neutral electrolytes offer a more stable environment for maintaining the structural integrity and redox activity of copper-based MOFs during electrochemical processes.
In light of these findings, PMrGO with a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 PM-to-rGO mass ratio, fabricated in fiber form, and tested in NaCl electrolyte, was selected for further investigation (Fig. 5f). This optimized configuration facilitates the full utilization of EDLC and pseudocapacitance, supported by the high inherent conductivity of the thermally reduced PMrGO architecture.
1 PM-to-rGO mass ratio, fabricated in fiber form, and tested in NaCl electrolyte, was selected for further investigation (Fig. 5f). This optimized configuration facilitates the full utilization of EDLC and pseudocapacitance, supported by the high inherent conductivity of the thermally reduced PMrGO architecture.
Following optimization of the PM-rGO hybrid-based electrochemical system, the energy storage properties of the PMrGO hybrid fibers were systematically investigated in a 1 M NaCl electrolyte. The CV curves (Fig. 6a) recorded at various scan rates exhibit minimal distortion, maintaining a similar shape even at a high scan rate of 200 mV s−1, indicating efficient charge transport and excellent rate capability. Galvanostatic charge–discharge (GCD) profiles at varying current densities are shown in Fig. 6b. The non-ideal GCD curves, deviating from the triangular shape typical of EDLC electrodes, indicate the dominance of reversible surface redox reactions in the PMrGO hybrid architecture. Remarkably, even at high current densities, the as-printed PMrGO hybrid architecture exhibits no observable ohmic drop (Fig. S15). This confirms the excellent electrical conductivity and high-rate capability of the PMrGO electrode. The PMrGO hybrid electrode exhibits a remarkable gravimetric capacitance of 476.9 F g−1 at a current density of 0.77 A g−1, corresponding to an areal capacitance of 430.56 mF cm−2 (Fig. 6c and Table S2). Even at a higher current density of 7.69 A g−1, the electrode maintains a gravimetric capacitance of 307.7 F g−1 and an areal capacitance of 277.78 mF cm−2, retaining approximately 65% of its initial capacitance. These results highlight the excellent rate capability and stable charge storage performance of the PMrGO device, even under high-rate operating conditions.
 | ||
| Fig. 6 Electrochemical performance of PMrGO hybrid fiber SCs in a two-electrode configuration using a 1 M NaCl electrolyte. (a) CV profiles at scan rates ranging from 5 to 200 mV s−1. (b) GCD profiles at various current densities. (c) Gravimetric capacitance and corresponding rate capability at different current densities. (d) Comparison of the areal and gravimetric capacitance of PMrGO fiber electrode with other reported fiber electrodes, including hollow rGO fiber,60 coaxial rGO fiber,61 EM-rGO/MXene fiber,64 NS-GF@RuO2 fiber,65 MXene/rGO fiber,63 and rGO/MnO2 fiber.62 (e) Nyquist plot; the inset shows a magnified view of the high-frequency region. (f) Bode plot in the frequency range from 0.1 Hz to 0.5 MHz. (g) Power-law dependence of current on scan rate. (h) b-Values at different potentials. (i) Areal Ragone plot of the PMrGO device, showing its energy and power densities in comparison with state-of-the-art hybrid fiber-based SCs, including PANI/GF,67 CuS|P-CuGFs//AC-GFs,68 rGO/PEDOT:PSS,60 N-doped SiNs/GF,69 NS-GF@RuO2,65 CNT/MXene/GF,70 rGO/MXene,64 and MoS2/GF.71 (j) Photograph demonstrating the practical application of two PMrGO fiber-based SC cells connected in series to power a commercial red LED. (k) Long-term cycling stability of PMrGO and PrGO devices at a scan rate of 20 mV s−1 over 4000 cycles. | ||
The superior performance of the PMrGO hybrid electrode is further underscored by its position in the areal versus gravimetric capacitance plot (Fig. 6d), where it outperforms a range of previously reported graphene-based fiber electrodes. These include hollow structured rGO fibers (304.5 mF cm−2 and 63.1 F g−1 at 0.08 mA cm−2),60 coaxial rGO fibers (205 mF cm−2 and 182 F g−1 at 10 mV s−1),61 rGO/MnO2 fiber (16.7 mF cm−2 and 65 F g−1 at 0.05 mA cm−2),62 MXene/rGO (372.2 mF cm−2 and 327.5 F g−1 at 10 mV s−1),63 EM-rGO/MXene (550.96 mF cm−2 and 110.89 F g−1 at 20 mV s−1),64 and NS-GF@RuO2 (20 mF cm−2 and 68.88 F g−1 at 0.2 A g−1).65 In comparison, the PMrGO system delivers a unique combination of high areal and gravimetric capacitance, reflecting the synergistic integration of redox-active MOFs with conductive and aligned graphene networks in the hybrid fiber architecture. It is also important to note that, for a comprehensive evaluation of device performance, a relatively high mass loading of 1.3 mg per electrode was deliberately used in the symmetric device. The as-printed architecture offers a distinct advantage in this regard, as increasing the mass does not adversely affect the ion diffusion path, which remains confined to the radius of the printed structure.
To examine the kinetics of ion transport in the PMrGO SC, electrochemical impedance spectroscopy (EIS) was performed at open-circuit voltage. As shown in Fig. 6e, the Nyquist plot exhibits a minimal equivalent series resistance (ESR) of 1 Ω, derived from the high-frequency intercept on the real axis. This low ESR reflects the combined contributions of the electrolyte's ionic conductivity and low interfacial contact resistances within the device. The absence of a semicircle in the high-frequency region further confirms negligible charge transfer resistance (Rct), consistent with rapid ion adsorption–desorption kinetics facilitated by the open-porous architecture of the PMrGO electrode. In the low-frequency region, the impedance spectrum displays a relatively steep slope, indicative of predominantly capacitive behavior characteristic of double-layer charge storage. The Bode plot for the PMrGO electrode is shown in Fig. 6f, providing further insight into its EIS behavior. At a low frequency of 0.1 Hz, the phase angle registers −52.6°, indicating the pseudocapacitive nature of the device. Upon closer examination, the characteristic frequency (f0) at a phase angle of −45° is approximately 9.99 Hz. Based on this value, the relaxation time constant (τ0 = 1/f0) is calculated to be about 100 ms. This short time constant signifies the excellent power capability of the PMrGO device and its rapid charge–discharge response. Additionally, the impedance modulus is lower than 545 Ω in the low-frequency region and approaches 1.1 Ω in the high-frequency region, providing clear evidence of the electrode's good electrochemical conductivity.
To further understand the charge storage kinetics mechanism of PMrGO SCs, the capacitive properties were examined through CV curves at various scanning rates, utilizing the equation66
| I = aνb | (1) |
A b-value of 0.5 indicates a diffusion-controlled process associated with faradaic intercalation, while a b-value of 1.0 corresponds to a purely capacitive behavior. As shown in Fig. 6g, the PMrGO device exhibits a strong linear relationship between current and scan rate. The b-values determined at different potentials (Fig. 6h) range from 0.52 to 0.73, indicating a mixed charge storage mechanism predominantly governed by diffusion-controlled pseudocapacitive processes. The Ragone plot (Fig. 6i) demonstrates the potential of the PMrGO device for high-energy- and high-power-density SCs. The PMrGO device achieves an impressive areal energy density (EA) of 29.89 μWh cm−2 at an areal power density (PA) of 0.35 mW cm−2 and retains 19.29 μWh cm−2 even at a higher power density of 3.47 mW cm−2, within a modest 1 V potential window (Table S2). Notably, the PMrGO architecture demonstrates some of the highest energy density values among reported prepared hybrid fiber-based architectures, including PANI/GF,67 CuS|P-CuGFs//AC-GFs,68 rGO/PEDOT:PSS,60 N-doped SiNs/GF,69 NS-GF@RuO2,65 CNT/MXene/GF,70 rGO/MXene,64 and MoS2/GF71 (Table S3).
Additionally, the PMrGO fiber-based SCs exhibit a gravimetric energy density (EG) of 16.56 Wh kg−1 and a gravimetric power density (PG) of 0.19 kW kg−1 at a current density of 0.77 A g−1. When the current density is increased tenfold to 7.69 A g−1, approximately 65% of the initial energy density is retained (10.68 Wh kg−1), while the power density increases to 1.92 kW kg−1 (Table S2). Notably, these values exceed those of commercial activated carbon-based SCs, which typically exhibit energy densities below 10 Wh kg−1.72 These results provide compelling evidence that the combination of porosity, conductivity, and pseudocapacitive contributions leads to exceptional energy storage performance. Furthermore, the practical application of the fabricated PMrGO device is demonstrated by powering a red LED using two SC cells connected in series (Fig. 6j).
Moreover, the PMrGO device exhibited excellent cycling stability, retaining 96.7% of its initial capacitance after 4000 charge–discharge cycles during CV testing at 20 mV s−1 (Fig. 6k). In contrast, the capacitance retention of the PrGO device decreased to 58.8% under the same conditions. This highlights the critical role of the MOF in enhancing the cycle life of the hybrid PMrGO fiber SC. POMs often suffer from poor cycling stability due to structural degradation and performance loss during repeated charge–discharge cycles.73 However, immobilizing POMs within the MOF effectively preserves their structural integrity and stability in the electrolyte, mitigating degradation and significantly improving their long-term cycling performance.
Fig. 7 offers a comprehensive overview of how the PMrGO composite's structure and porosity evolve before and after the 4000-cycle stability test. Complementary characterizations including SEM, Gas sorption along with BJH analysis, XRD, FTIR and Raman spectroscopy were performed to reinforce the validity of the electrochemical observations to align with best practices for accurate and reproducible materials analysis.74 SEM image (Fig. 7a) reveal that, following cycling, the originally well-defined cubic morphology of the Cu-BTC crystals decorating the PMrGO surface becomes less distinct, exhibiting a partial loss of faceted edges and a transition toward a distorted cubic morphology. This morphological alteration correlates with the observed reduction in crystallinity in the XRD patterns (Fig. 7d), where the intensity of the Cu-BTC (111) reflection75 at 5.8° diminishes after cycling, while the positions of all peaks remain unchanged, confirming that the framework structure persists without major phase transformations. Such selective attenuation of the (111) reflection suggests a loss of long-range order along this crystallographic direction, consistent with the observed rounding of the cubic crystal morphology.
Textural properties derived from nitrogen adsorption–desorption isotherms (Fig. 7b) show a moderate decrease in specific surface area, from 2240.2 m2 g−1 to 2044.5 m2 g−1 after cycling, indicating partial pore collapse or blockage. BJH pore size distribution analyses (Fig. 7c) further reveal shifts in pore size peaks, with the appearance of slightly larger mesopores after cycling, which may reflect partial structural rearrangement or defect formation within the MOF framework. Despite these changes, both FTIR (Fig. 7e) and Raman spectra (Fig. 7f) remain nearly unchanged after cycling, demonstrating that the chemical functionalities of the framework and the composite remain preserved, with no significant alterations in bonding environments.
In addition to structural and spectroscopic characterization, EIS was performed after 4000 charge–discharge cycles to further evaluate the interfacial and conductive stability of the PMrGO electrode (Fig. S16). The EIS spectra revealed minimal changes in the high-frequency region, where the overlapping intercepts on the real axis indicate stable series resistance, reflecting retained electrolyte conductivity and preserved interfacial contact resistances. However, the low-frequency region exhibited a reduced phase angle, signifying a deviation from ideal capacitive behavior and increased resistive contributions within the electrode–electrolyte system. This shift is attributed to structural or interfacial degradation, such as pore clogging or a reduction in ion-accessible surface area during cycling, as evidenced by BET and BJH analyses (Fig. 7b and c), which introduce resistive losses and disrupt capacitive double-layer formation at lower frequencies. Overall, these results indicate that while the PMrGO composite retains its chemical integrity and general framework structure after stability cycling, there is a partial loss of crystallinity and a mild decrease in porosity, likely associated with morphological rounding of the cubic MOF crystals and defect formation. Such structural evolution is not uncommon in MOF-based materials under cycling conditions and highlights the balance between preserving structural features and achieving long-term stability in composite materials.
Conclusions
In conclusion, we present a significant advancement in fiber-based SCs through thermally stabilized MOF-graphene hybrid fibers fabricated via wet-spinning polyoxometalate-enhanced Cu-BTC MOFs and LCGO. The optimized architecture achieves a remarkable gravimetric capacitance of 476.9 F g−1 at 0.77 A g−1 with good rate capability (307.7 F g−1 at 7.7 A g−1) and excellent cycling stability (96.7% retention over 4000 cycles) in practical two-electrode configurations. The hybrid fibers simultaneously exhibit robust mechanical properties (Young's modulus >42 GPa) and hierarchical porosity, addressing the common challenges of balancing electrochemical performance with mechanical integrity in fiber electrodes. The successful integration of MOF-derived pseudocapacitance with graphene's conductivity through the scalable wet-spinning process used here offers a promising approach for fabricating structurally integrated, binder-free fiber-based energy storage devices. This work provides valuable insights into designing multifunctional electrode materials by combining the complementary advantages of MOFs and graphene in fiber architectures.Experimental section
Synthesis of GO
GO was synthesized via a modified Hummers' method, following our previous reports.10,49 At the start, 0.5 grams of graphite flakes were added to a 250 mL glass vessel, which was then set in an ice bath. Then, 50 mL of 98% sulfuric acid (H2SO4) was added to the vessel. Under vigorous stirring, 3.5 g of potassium permanganate (KMnO4) was slowly added and the resulting mixture was left overnight. For neutralization, 50 mL of deionized (DI) water was added dropwise during high shear mixing. Then, 7 mL of hydrogen peroxide (diluted in 13 mL DI water) was added to neutralize the KMnO4. After an additional 30 minutes of stirring, the GO particles were washed and centrifuged using a hydrochloric acid (HCl) solution (9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 water to HCl by volume). They were then centrifuged again and rinsed with deionized water until the solution reached a pH of approximately 5 to 6. Finally, the GO particles were diluted with DI water and exfoliated by gentle shaking.
1 water to HCl by volume). They were then centrifuged again and rinsed with deionized water until the solution reached a pH of approximately 5 to 6. Finally, the GO particles were diluted with DI water and exfoliated by gentle shaking.
Synthesis of PM
To synthesize PM, 0.21 g of BTC (1 mmol) and 0.10 g of cetyltrimethylammonium bromide (CTAB) were dissolved in 14 mL of absolute ethanol. Subsequently, 0.18 g of HPW and 1.45 g of copper(II) nitrate trihydrate (Cu(NO3)2·3H2O) were dissolved in 12 mL of DI water and added dropwise to the BTC-CTAB solution under stirring for approximately 30 minutes. The mixture was left undisturbed at room temperature for 2 days, leading to the formation of a blue precipitate. The precipitate was collected, washed three times with DI water, and dried at 60 °C for 24 hours. CTAB was removed using Soxhlet extraction with ethanol for 24 hours. The final product was air-dried at 60 °C, achieving a yield of 93%.Printing PMrGO hybrid architectures
PMrGO hybrid fibers were fabricated through a three-step process. First, PM and GO suspensions were mixed at varying weight ratios and sonicated in an ultrasonic bath for 30 minutes to achieve a homogeneous dispersion. The resulting mixture was then loaded into plastic syringes and extruded into a rotating coagulation bath containing isopropanol. During extrusion, shear forces induced alignment of the nematic domains in the hybrid liquid crystal dispersion, enabling the formation of continuous, unidirectional fibers. Isopropanol was selected as the coagulation medium instead of conventional acetone due to its lower residue content and milder effect on the fiber structure, which helped preserve morphology after solvent evaporation.After fiber formation, the as-spun PMGO fibers were thermally annealed at 200 °C to reduce GO to rGO. This step created a conductive network while maintaining the structural integrity of the hybrid architecture, resulting in the final PMrGO fibers. For comparison, PMGO fibers were also obtained by air-drying under tension at room temperature without thermal reduction. Pure GO fibers were prepared following the same procedure, omitting the addition of PM.
Fabrication of PMrGO hybrid electrodes
The working electrodes were fabricated by printing PMGO architectures into an isopropanol coagulation bath, then deposition onto pre-cleaned titanium sheets (1.2 cm × 1.2 cm) for device assembly in a symmetric two-electrode configuration. The PMGO-coated titanium sheets were subsequently annealed at 200 °C for 2 hours to reduce GO to rGO, resulting in the final PMrGO hybrid architectures.Materials characterizations
Atomic force microscopy (AFM) and scanning electron microscopy (SEM) analyses were performed on GO sheets deposited onto pre-cleaned, silanized silicon wafers with a 300 nm SiO2 layer, following our previously reported method. The silane solution was prepared by mixing 3-aminopropyltriethoxysilane and water (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9 v/v), with one drop of HCl added to catalyze hydrolysis. Pre-cut silicon substrates were immersed in this solution for 30 minutes and rinsed thoroughly with Millipore water. GO sheets were deposited onto the silanized substrates by immersing them in a GO dispersion (50 mg mL−1) for 5 seconds, then rinsing in clean Millipore water for 30 seconds and air-drying. The uniformity of GO sheet deposition was confirmed via optical microscopy (OM) before AFM and SEM imaging. FESEM (JEOL JSM-7500FA and Zeiss EVO MA 15), both equipped with EDS detectors, was used to analyze morphology and elemental composition. The lateral size distribution of over 100 isolated GO sheets was measured using ImageJ software. The diameter of an equal-area circle was used to define the lateral size of ultra-large GO sheets. AFM characterization was conducted in tapping mode under ambient conditions using an MFP-3D system (Asylum Research).
9 v/v), with one drop of HCl added to catalyze hydrolysis. Pre-cut silicon substrates were immersed in this solution for 30 minutes and rinsed thoroughly with Millipore water. GO sheets were deposited onto the silanized substrates by immersing them in a GO dispersion (50 mg mL−1) for 5 seconds, then rinsing in clean Millipore water for 30 seconds and air-drying. The uniformity of GO sheet deposition was confirmed via optical microscopy (OM) before AFM and SEM imaging. FESEM (JEOL JSM-7500FA and Zeiss EVO MA 15), both equipped with EDS detectors, was used to analyze morphology and elemental composition. The lateral size distribution of over 100 isolated GO sheets was measured using ImageJ software. The diameter of an equal-area circle was used to define the lateral size of ultra-large GO sheets. AFM characterization was conducted in tapping mode under ambient conditions using an MFP-3D system (Asylum Research).
TGA was performed on all samples using a Q600 TGA/DSC2 (TA Instruments) at a heating rate of 10 °C min−1 under ambient air. Nitrogen sorption experiments were conducted using a BELSORP Mini adsorption system (BEL Japan Inc.) to determine the specific surface area and pore size distribution. The birefringence of the liquid crystal dispersions and the printed architectures was investigated using a custom-built polarized OM setup in transmission mode.
XRD patterns were obtained using a Philips X'Pert Pro powder diffractometer with Cu Kα radiation (λ = 0.154 nm). Zeta potential measurements were carried out using the Brookhaven 90 Plus instrument. The morphology of the printed fiber architectures was further examined by FESEM (Zeiss Sigma 300). The Young's modulus of the samples was measured using an NHT3 nanoindentation tester (Anton Paar) equipped with a Berkovich diamond indenter. The data were analyzed using the Oliver–Pharr method. Raman spectra were collected using an XploRA™ PLUS confocal micro-Raman spectrometer (Horiba France SAS) equipped with a CCD detector and a 532 nm laser. FTIR spectra were recorded using a Spectrum 100 spectrometer (PerkinElmer, USA) operating in the wavenumber range of 2000–450 cm−1, with a wavelength accuracy of 1 cm−1.
Electrochemical characterizations
The SC performance was evaluated through CV (Metrohm DropSens, SPELEC) and GCD (NEWARE battery tester, BTS 4000) experiments conducted in a two-electrode configuration. The mass of the active material on each electrode was measured to be 1.3 mg. All CV and GCD data exhibited consistent repeatability, and the reported values represent averaged results. The EIS measurements were performed at open circuit voltage by applying a sinusoidal potential over a frequency range of 0.1 Hz to 0.5 MHz.Electrochemical calculations
The gravimetric capacitance (CG, F g−1) and areal capacitance (CA, F cm−2) of a single electrode in the symmetric device were calculated from the GCD discharge curves according to the following equations:76,77 | (2) |
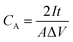 | (3) |
The gravimetric energy density (EG, Wh kg−1) and power density (PG, W kg−1) of the device were calculated using the following equations:76,77
 | (4) |
 | (5) |
The areal energy density (EA, μWh cm−2) and power density (PA, mW cm−2) of the device were calculated according to the following equations:76,77
 | (6) |
 | (7) |
Conflicts of interest
The authors declare no competing financial interest.Data availability
The data supporting this article have been included as part of the SI.Detailed experimental procedures, characterization and energy performance metrics are included in SI. See DOI: https://doi.org/10.1039/d5ta02969d.
Acknowledgements
This research was supported by Iran National Science Foundation (INSF) under project No. 4022382. Instrumental characterization and materials preparation facilities were provided by the Condensed Matter National Laboratory at the Institute for Research in Fundamental Sciences (IPM) in Tehran, Iran. Additionally, financial support for equipment purchase was granted by the INSF under project number 4022382.References
- E. Pomerantseva, F. Bonaccorso, X. Feng, Y. Cui and Y. Gogotsi, Science, 2019, 366, eaan8285 CrossRef PubMed.
- J. Huang, Y. Xie, Y. You, J. Yuan, Q. Xu, H. Xie and Y. Chen, Adv. Funct. Mater., 2023, 33, 2213095 CrossRef.
- R. Sharma, H. Kumar, G. Kumar, S. Sharma, R. Aneja, A. K. Sharma, R. Kumar and P. Kumar, Chem. Eng. J., 2023, 468, 143706 CrossRef.
- F. Wang, X. Wu, X. Yuan, Z. Liu, Y. Zhang, L. Fu, Y. Zhu, Q. Zhou, Y. Wu and W. Huang, Chem. Soc. Rev., 2017, 46, 6816–6854 RSC.
- P. Simon and Y. Gogotsi, Nat. Mater., 2020, 19, 1151–1163 CrossRef CAS PubMed.
- H. Shao, Y.-C. Wu, Z. Lin, P.-L. Taberna and P. Simon, Chem. Soc. Rev., 2020, 49, 3005–3039 RSC.
- S. H. Aboutalebi, A. T. Chidembo, M. Salari, K. Konstantinov, D. Wexler, H. K. Liu and S. X. Dou, Energy Environ. Sci., 2011, 4, 1855–1865 RSC.
- S. H. Aboutalebi, R. Jalili, D. Esrafilzadeh, M. Salari, Z. Gholamvand, S. Aminorroaya Yamini, K. Konstantinov, R. L. Shepherd, J. Chen, S. E. Moulton, P. C. Innis, A. I. Minett, J. M. Razal and G. G. Wallace, ACS Nano, 2014, 8, 2456–2466 CrossRef CAS PubMed.
- A. Chidembo, S. H. Aboutalebi, K. Konstantinov, M. Salari, B. Winton, S. A. Yamini, I. P. Nevirkovets and H. K. Liu, Energy Environ. Sci., 2012, 5, 5236–5240 RSC.
- M. M. Islam, S. H. Aboutalebi, D. Cardillo, H. K. Liu, K. Konstantinov and S. X. Dou, ACS Cent. Sci., 2015, 1, 206–216 CrossRef CAS PubMed.
- B. Liu, Q. Zhang, L. Zhang, C. Xu, Z. Pan, Q. Zhou, W. Zhou, J. Wang, L. Gu and H. Liu, Adv. Mater., 2022, 34, 2106309 CrossRef CAS.
- D. K. Singha, R. I. Mohanty, P. Bhanja and B. K. Jena, Mater. Adv., 2023, 4, 4679–4706 RSC.
- J. W. Gittins, K. Ge, C. J. Balhatchet, P.-L. Taberna, P. Simon and A. C. Forse, J. Am. Chem. Soc., 2024, 146, 12473–12484 CrossRef CAS PubMed.
- D. Sheberla, J. C. Bachman, J. S. Elias, C.-J. Sun, Y. Shao-Horn and M. Dincă, Nat. Mater., 2017, 16, 220–224 CrossRef CAS.
- S. Zhai and Y. Chen, Acc. Mater. Res., 2022, 3, 922–934 CrossRef CAS.
- M. F. El-Kady, V. Strong, S. Dubin and R. B. Kaner, Science, 2012, 335, 1326–1330 CrossRef CAS.
- M. Ghidiu, M. R. Lukatskaya, M.-Q. Zhao, Y. Gogotsi and M. W. Barsoum, Nature, 2014, 516, 78–81 CrossRef CAS PubMed.
- W. Chen, J. Gu, Q. Liu, M. Yang, C. Zhan, X. Zang, T. A. Pham, G. Liu, W. Zhang, D. Zhang, B. Dunn and Y. Morris Wang, Nat. Nanotechnol., 2022, 17, 153–158 CrossRef CAS PubMed.
- E. Rafiee and N. Nobakht, J. Mol. Catal. A: Chem., 2015, 398, 17–25 CrossRef CAS.
- N. Nobakht, S. A. Etghani, M. Hosseini and S. H. Aboutalebi, J. Energy Chem., 2024, 97, 388–418 CrossRef CAS.
- Z. A. Sandhu, M. A. Raza, N. S. Awwad, H. A. Ibrahium, U. Farwa, S. Ashraf, A. Dildar, E. Fatima, S. Ashraf and F. Ali, Mater. Adv., 2024, 5, 30–50 RSC.
- S.-J. Shin, J. W. Gittins, C. J. Balhatchet, A. Walsh and A. C. Forse, Adv. Funct. Mater., 2024, 34, 2308497 CrossRef CAS.
- N. Nobakht, M. A. Faramarzi, A. Shafiee, M. Khoobi and E. Rafiee, Int. J. Biol. Macromol., 2018, 113, 8–19 CrossRef CAS.
- S. Bi, H. Banda, M. Chen, L. Niu, M. Chen, T. Wu, J. Wang, R. Wang, J. Feng, T. Chen, M. Dincă, A. A. Kornyshev and G. Feng, Nat. Mater., 2020, 19, 552–558 CrossRef CAS PubMed.
- D. Feng, T. Lei, M. R. Lukatskaya, J. Park, Z. Huang, M. Lee, L. Shaw, S. Chen, A. A. Yakovenko, A. Kulkarni, J. Xiao, K. Fredrickson, J. B. Tok, X. Zou, Y. Cui and Z. Bao, Nat. Energy, 2018, 3, 30–36 CrossRef CAS.
- L. Niu, T. Wu, M. Chen, L. Yang, J. Yang, Z. Wang, A. A. Kornyshev, H. Jiang, S. Bi and G. Feng, Adv. Mater., 2022, 34, 2200999 CrossRef CAS PubMed.
- E. Rafiee and N. Nobakht, Acta Chim. Slov., 2016, 63, 309–316 CrossRef CAS PubMed.
- M. Zhao and S. Tong, Energy Fuels, 2024, 38, 13796–13818 CrossRef CAS.
- B. Yang, B. Li and Z. Xiang, Nano Res., 2023, 16, 1338–1361 CrossRef CAS.
- W. Zhao, Y. Zeng, Y. Zhao and X. Wu, J. Energy Storage, 2023, 62, 106934 CrossRef.
- G. Song, Y. Shi, S. Jiang and H. Pang, Adv. Funct. Mater., 2023, 33, 2303121 CrossRef CAS.
- B. He, Q. Zhang, Z. Pan, L. Li, C. Li, Y. Ling, Z. Wang, M. Chen, Z. Wang, Y. Yao, Q. Li, L. Sun, J. Wang and L. Wei, Chem. Rev., 2022, 122, 10087–10125 CrossRef CAS PubMed.
- K. Jayaramulu, M. Horn, A. Schneemann, H. Saini, A. Bakandritsos, V. Ranc, M. Petr, V. Stavila, C. Narayana, B. Scheibe, Š. Kment, M. Otyepka, N. Motta, D. Dubal, R. Zbořil and R. A. Fischer, Adv. Mater., 2021, 33, 2004560 CrossRef CAS PubMed.
- M. Lu, G. Wang, X. Yang and B. Hou, Nano Res., 2022, 15, 6112–6120 CrossRef CAS.
- C.-Y. Sun, S.-X. Liu, D.-D. Liang, K.-Z. Shao, Y.-H. Ren and Z.-M. Su, J. Am. Chem. Soc., 2009, 131, 1883–1888 CrossRef CAS PubMed.
- M. Bagheri, A. Melillo, B. Ferrer, M. Y. Masoomi and H. Garcia, ACS Appl. Mater. Interfaces, 2022, 14, 978–989 CrossRef CAS PubMed.
- D. T. Lee, Z. Dai, G. W. Peterson, M. G. Hall, N. L. Pomerantz, N. Hoffman and G. N. Parsons, Adv. Funct. Mater., 2022, 32, 2108004 CrossRef CAS.
- H. K. Kim, W. S. Yun, M.-B. Kim, J. Y. Kim, Y.-S. Bae, J. Lee and N. C. Jeong, J. Am. Chem. Soc., 2015, 137, 10009–10015 CrossRef CAS PubMed.
- L. H. Wee, C. Wiktor, S. Turner, W. Vanderlinden, N. Janssens, S. R. Bajpe, K. Houthoofd, G. Van Tendeloo, S. De Feyter, C. E. A. Kirschhock and J. A. Martens, J. Am. Chem. Soc., 2012, 134, 10911–10919 CrossRef CAS PubMed.
- S.-M. Liu, Z. Zhang, X. Li, H. Jia, M. Ren and S. Liu, Adv. Mater. Interfaces, 2018, 5, 1801062 CrossRef.
- D. Mustafa, E. Breynaert, S. R. Bajpe, J. A. Martens and C. E. A. Kirschhock, Chem. Commun., 2011, 47, 8037–8039 RSC.
- Y.-J. Wang, Y.-Y. Zhou, H.-G. Hao, M. Song, N. Zhang, S. Yao, J.-H. Yan, Z.-M. Zhang and T.-B. Lu, Inorg. Chem., 2018, 57, 1342–1349 CrossRef CAS PubMed.
- D. Mohanadas, M. A. A. Mohd Abdah, N. H. N. Azman, J. Abdullah and Y. Sulaiman, Int. J. Hydrogen Energy, 2021, 46, 35385–35396 CrossRef.
- E. Rafiee, N. Nobakht and L. Behbood, Res. Chem. Intermed., 2017, 43, 951–969 CrossRef.
- C. Petit, J. Burress and T. J. Bandosz, Carbon, 2011, 49, 563–572 CrossRef.
- I. Spanopoulos, C. Tsangarakis, E. Klontzas, E. Tylianakis, G. Froudakis, K. Adil, Y. Belmabkhout, M. Eddaoudi and P. N. Trikalitis, J. Am. Chem. Soc., 2016, 138, 1568–1574 CrossRef.
- D. Esrafilzadeh, R. Jalili, E. M. Stewart, S. H. Aboutalebi, J. M. Razal, S. E. Moulton and G. G. Wallace, Adv. Funct. Mater., 2016, 26, 3105–3117 CrossRef.
- S. Naficy, R. Jalili, S. H. Aboutalebi, R. A. Gorkin III, K. Konstantinov, P. C. Innis, G. M. Spinks, P. Poulin and G. G. Wallace, Mater. Horiz., 2014, 1, 326–331 RSC.
- R. Jalili, S. H. Aboutalebi, D. Esrafilzadeh, R. L. Shepherd, J. Chen, S. Aminorroaya-Yamini, K. Konstantinov, A. I. Minett, J. M. Razal and G. G. Wallace, Adv. Funct. Mater., 2013, 23, 5345–5354 CrossRef.
- A. M. Eagleton, E. K. Ambrogi, S. A. Miller, N. Vereshchuk and K. A. Mirica, Angew. Chem., Int. Ed., 2023, 62, e202309078 CrossRef.
- G. W. Peterson, D. T. Lee, H. F. Barton, T. H. Epps and G. N. Parsons, Nat. Rev. Mater., 2021, 6, 605–621 CrossRef.
- Y. Zhang, S. Yuan, X. Feng, H. Li, J. Zhou and B. Wang, J. Am. Chem. Soc., 2016, 138, 5785–5788 CrossRef.
- R. Kumar, D. Raut, U. Ramamurty and C. N. R. Rao, Angew. Chem., Int. Ed., 2016, 55, 7857–7861 CrossRef PubMed.
- K. Jayaramulu, S. Mukherjee, D. M. Morales, D. P. Dubal, A. K. Nanjundan, A. Schneemann, J. Masa, S. Kment, W. Schuhmann, M. Otyepka, R. Zbořil and R. A. Fischer, Chem. Rev., 2022, 122, 17241–17338 CrossRef PubMed.
- A. E. R. S. Khder, H. M. A. Hassan and M. S. El-Shall, Appl. Catal., A, 2014, 487, 110–118 CrossRef CAS.
- D. Mohanadas, M. A. A. Mohd Abdah, N. H. N. Azman, T. B. S. A. Ravoof and Y. Sulaiman, Sci. Rep., 2021, 11, 11747 CrossRef CAS PubMed.
- M. Saraf, R. Rajak and S. M. Mobin, J. Mater. Chem. A, 2016, 4, 16432–16445 RSC.
- G. Lee, G. Park and S. S. Park, J. Am. Chem. Soc., 2024, 146, 29767–29772 CrossRef CAS PubMed.
- Y. Duan, H. Li, X. Shi, C. Ji, J. Imbrogno and D. Zhao, Ind. Eng. Chem. Res., 2025, 64, 5372–5382 CrossRef CAS.
- G. Qu, J. Cheng, X. Li, D. Yuan, P. Chen, X. Chen, B. Wang and H. Peng, Adv. Mater., 2016, 28, 3646–3652 CrossRef CAS PubMed.
- X. Zhao, B. Zheng, T. Huang and C. Gao, Nanoscale, 2015, 7, 9399–9404 RSC.
- J. Zhang, X. Yang, Y. He, Y. Bai, L. Kang, H. Xu, F. Shi, Z. Lei and Z.-H. Liu, J. Mater. Chem. A, 2016, 4, 9088–9096 RSC.
- Q. Yang, Z. Xu, B. Fang, T. Huang, S. Cai, H. Chen, Y. Liu, K. Gopalsamy, W. Gao and C. Gao, J. Mater. Chem. A, 2017, 5, 22113–22119 RSC.
- N. He, S. Patil, J. Qu, J. Liao, F. Zhao and W. Gao, ACS Appl. Energy Mater., 2020, 3, 2949–2958 CrossRef CAS.
- S. H. Noh, H. B. Lee, K. S. Lee, H. Lee and T. H. Han, ACS Appl. Mater. Interfaces, 2022, 14, 29867–29877 CrossRef PubMed.
- J. Wang, J. Polleux, J. Lim and B. Dunn, J. Phys. Chem. C, 2007, 111, 14925–14931 CrossRef CAS.
- X. Zheng, L. Yao, Y. Qiu, S. Wang and K. Zhang, ACS Appl. Energy Mater., 2019, 2, 4335–4344 CrossRef CAS.
- B. Bai, J. Shui, Y. Wang, Z. Su, L. Qiu and P. Du, ACS Appl. Nano Mater., 2024, 7, 12387–12398 CrossRef CAS.
- B. Bai, L. Qiu, Y. Yuan, L. Song, J. Xiong and P. Du, J. Energy Storage, 2023, 63, 106984 CrossRef.
- R. Xiang, J. Zhang, X. Yang, Y. Liu, C. Lu, X. Wang and K. Zhang, ACS Appl. Energy Mater., 2022, 5, 13212–13221 CrossRef CAS.
- M. Tang, Y. Wu, J. Yang and Y. Xue, J. Alloys Compd., 2020, 828, 153622 CrossRef CAS.
- L. Liu, P.-L. Taberna, B. Dunn and P. Simon, ACS Energy Lett., 2021, 6, 4311–4316 CrossRef CAS.
- Y. Zhang, Y. Li, H. Guo, Y. Guo and R. Song, Mater. Chem. Front., 2024, 8, 732–768 RSC.
- S. H. Aboutalebi, Nanoscale Adv. Mater., 2025, 2, 10–15 Search PubMed.
- S. Iwamoto, R. Nakayama, S. Chon, R. Shimizu and T. Hitosugi, J. Mater. Chem. A, 2024, 12, 17492–17500 RSC.
- Y. Shao, M. F. El-Kady, C.-W. Lin, G. Zhu, K. L. Marsh, J. Y. Hwang, Q. Zhang, Y. Li, H. Wang and R. B. Kaner, Adv. Mater., 2016, 28, 6719–6726 CrossRef CAS PubMed.
- B. Yao, S. Chandrasekaran, H. Zhang, A. Ma, J. Kang, L. Zhang, X. Lu, F. Qian, C. Zhu, E. B. Duoss, C. M. Spadaccini, M. A. Worsley and Y. Li, Adv. Mater., 2020, 32, 1906652 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2025 |






