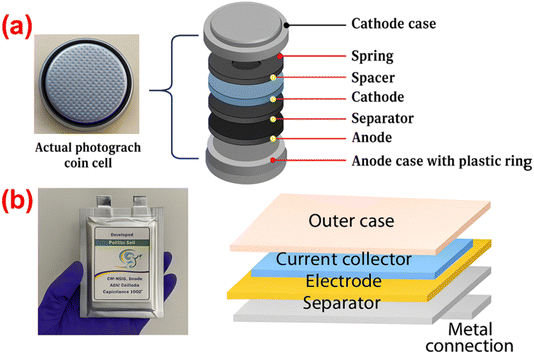
Recent advances in hybrid supercapacitors: a review of high performance materials and scalable fabrication techniques
Nargish
Parvin†
a,
Dhananjaya
Merum†
ab,
Misook
Kang
 b,
Sang Woo
Joo
b,
Sang Woo
Joo
 a,
Jae Hak
Jung
*c and
Tapas Kumar
Mandal
a,
Jae Hak
Jung
*c and
Tapas Kumar
Mandal
 *a
*a
aSchool of Mechanical Engineering, Yeungnam University, Gyeongsan 38541, Republic of Korea. E-mail: nargish.parvin@gmail.com; msdhananjaya51@gmail.com; swjoo@yu.ac.kr; tps.mndl@gmail.com
bCollege of Natural Sciences, Department of Chemistry, Yeungnam University, Gyeongsan 38541, Republic of Korea. E-mail: mskang@ynu.ac.kr
cSchool of Chemical Engineering, Yeungnam University, Gyeongsan, 38541, Republic of Korea. E-mail: jhjung@ynu.ac.kr
First published on 30th May 2025
Abstract
Hybrid supercapacitors (HSCs) have emerged as a transformative energy storage technology, bridging the gap between traditional capacitors and batteries by combining high power density with significant energy storage capacity. This review comprehensively examines the recent advancements in materials and fabrication techniques for HSCs. We discuss the fundamental principles and mechanisms that distinguish HSCs, highlighting the roles of various electrode materials, including carbon-based materials, metal oxides, conducting polymers, and emerging composite materials such as metal–organic frameworks (MOFs) and covalent organic frameworks (COFs). The review also delves into the diversity of electrolyte materials, encompassing aqueous, organic, and solid-state electrolytes, and their impact on the performance of HSCs. Key fabrication methods, including chemical vapor deposition (CVD) and hydrothermal synthesis, are outlined, emphasizing their contributions to enhancing material properties and device performance. Performance metrics such as capacitance, energy density, and cycling stability are critically evaluated. Applications of HSCs in consumer electronics, electric vehicles, and grid storage are explored, demonstrating their versatility and potential. Finally, the review addresses current challenges and future directions in the field, focusing on material scalability, environmental impact, and developing next-generation HSCs. This synthesis aims to provide insights and stimulate further research in the rapidly evolving landscape of hybrid supercapacitors.
1. Introduction
1.1. Overview of energy storage technologies
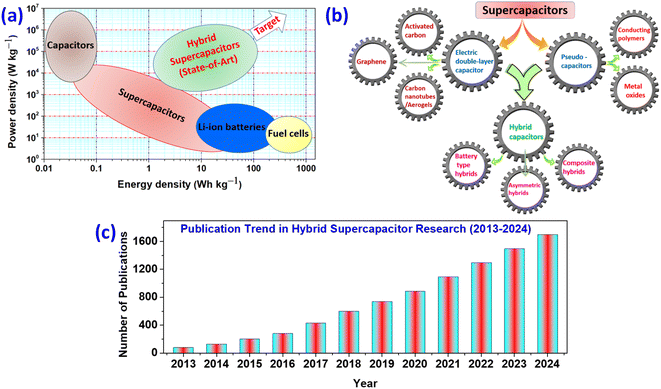
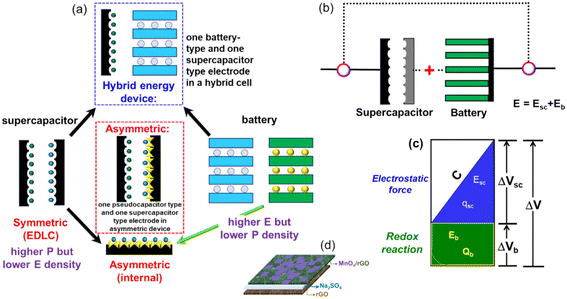
In contemporary society, the energy demand is continuously increasing, and modern organizations have historically met this need by utilizing fossil fuels. Excess use of conventional fossil fuels such as coal, oil, and natural gas contributes to global warming, negatively impacting societal development and human health. To address these issues, the world has been promoting the development of clean and sustainable energy alternatives to replace fossil fuels. Through this approach, we can generate substantial amounts of clean and sustainable energy by harnessing natural sources such as wind, tidal, hydro, and solar energy.1–4 However, environmental conditions can affect the durability and stability of devices used to generate electrical power. Consequently, energy storage devices must be developed to store acquired energy for various applications. Batteries, supercapacitors, and fuel cells are examples of electrochemical energy storage (EES) technologies that convert chemical energy into electrical energy and offer viable alternatives due to their unique properties. EES devices are widely used in consumer electronics such as mobile phones, iPods, laptops, digital cameras, emergency doors, military and aerospace equipment, and hybrid vehicles.5–10 Batteries and supercapacitors have garnered significant interest in academia and industry over the last several decades due to their high energy density, extended shelf life, high power density, and cycle stability. Li-ion batteries have a high specific energy density (100–280 W h kg−1) but limited power density (0.2–1.8 kW kg−1) and shorter lifecycles due to sluggish charge–discharge kinetics and degradation over time.11–13 Supercapacitors, on the other hand, have a high power density and good capacity retention but a lower energy density of 5–30 W h kg−1.14,15Fig. 1a depicts the Ragone plot, which compares the energy density (W h kg−1) and power density (W kg−1) of various electrochemical energy storage devices. The illustration shows that while traditional capacitors have a higher power density than batteries or fuel cells, their energy density is lower. A conventional capacitor can be charged or discharged very quickly and generate high power, but it cannot store much energy per unit mass or volume. Batteries and fuel cells have higher energy storage capacity, but their dynamic performance is low.16,17 Supercapacitors are considered the intermediate between conventional capacitors and batteries as they store more energy than capacitors and more power than batteries. Supercapacitors can charge and discharge quickly while maintaining a respectable energy storage capacity per unit mass.Supercapacitors (SCs) are generally categorized into EDLCs and pseudocapacitors. EDLCs store energy through the electrostatic separation of charges at the electrode–electrolyte interface, while pseudocapacitors involve faradaic reactions for charge storage.18 Despite their advantages, traditional supercapacitors face limitations in energy density, which restricts their application in high-energy-demand scenarios.19 In general, the electrochemical efficiency of the active materials, electrolyte selection, and the system's stable voltage window may all influence supercapacitor performance. As a consequence, much research has been conducted to produce innovative electrode materials with novel structures and device designs to enable successful ion and electron transport over a short diffusion length. Furthermore, the commercial usage of many other types of SCs, such as EDLC, pseudo, and hybrid, is categorized based on their charging methodologies or the materials utilized in the electrodes, as illustrated in Fig. 1b.
1.2. Importance of hybrid supercapacitors
Hybrid supercapacitors (HSCs) have emerged as a promising solution to bridge the performance gap between batteries and supercapacitors. By integrating battery-like and capacitor-like materials, HSCs aim to achieve both high energy density and high power density.20 This hybrid approach leverages the fast charge–discharge characteristics of capacitive materials and the substantial energy storage capabilities of battery-type materials.21 The development of HSCs addresses the critical need for advanced energy storage systems in various sectors, including electric vehicles (EVs), portable electronics, and renewable energy integration.22,23 For instance, HSCs can significantly enhance the efficiency and performance of EVs by providing rapid acceleration and energy recuperation during braking.24 In portable electronics, the compact size and high performance of HSCs can lead to longer battery life and faster charging times.251.3. Scope and objectives of the review
This review aims to provide a comprehensive analysis of the latest advancements in the materials and fabrication techniques for hybrid supercapacitors. We will explore the fundamental principles underlying HSCs, focusing on the synergistic effects of combining different types of electrode materials. Specifically, we will examine the roles of carbon-based materials, metal oxides, conducting polymers, and emerging composites such as metal–organic frameworks (MOFs) and covalent organic frameworks (COFs). Furthermore, the review will delve into the various types of electrolytes used in HSCs, including aqueous, organic, and solid-state electrolytes, and their impact on the overall performance of the devices. We will discuss key fabrication methods, such as CVD and hydrothermal synthesis that are instrumental in enhancing the material properties and device performance of HSCs. In addition to material and fabrication insights, this review will evaluate HSCs' performance metrics, such as capacitance, energy density, and cycling stability. We will highlight their practical applications in consumer electronics, electric vehicles, and grid storage, showcasing their versatility and potential.Despite the availability of several recent reviews on supercapacitors, this work uniquely distinguishes itself through a comprehensive, materials-to-device-level exploration of hybrid supercapacitors. In particular, it integrates a comparative evaluation of diverse electrode materials, including emerging MOFs and COFs-with a detailed analysis of advanced fabrication techniques, such as hydrothermal synthesis and chemical vapor deposition. Unlike prior reviews, this article also emphasizes the critical influence of various electrolytes and the structure–property–performance relationships governing HSC efficiency. Furthermore, it maps technological developments to practical applications in electric vehicles, wearable devices, and smart grids, while addressing key challenges related to device integration, environmental sustainability, and industrial scalability. This multidimensional approach provides new insights and bridges important knowledge gaps, making the review a timely and valuable resource for researchers and engineers working on next-generation energy storage technologies. Finally, the review will address the current challenges and future directions in the field, emphasizing the importance of material scalability, environmental impact, and the development of next-generation HSCs. By providing a thorough understanding of the advancements and ongoing research in hybrid supercapacitors, this review aims to stimulate further innovation and application in this rapidly evolving field.
1.4. Methodology of the review
This review was conducted using a structured and systematic approach to identify, analyze, and synthesize current knowledge on hybrid supercapacitors, focusing on high-performance materials and scalable fabrication techniques. The methodology comprises literature collection, selection criteria, data extraction, thematic categorization, and analysis of emerging trends.Scopus, Web of Science, Google Scholar, ScienceDirect, IEEE Xplore, SpringerLink, MDPI, ACS Publications, RSC Publications, etc.
• The search covered peer-reviewed articles published between 2013 and 2025 to capture a decade of advancements. The following keywords and Boolean operators were used:
• “Hybrid supercapacitors”
• “Electrode materials for supercapacitors”
• “High-performance supercapacitor materials”
• “Metal oxides and conducting polymers in energy storage”
• “Carbon nanomaterials and supercapacitor electrodes”
• “Scalable fabrication techniques for supercapacitors”
• “Sol–gel synthesis supercapacitors”
• “CVD supercapacitor electrodes”
• “Ionic liquids supercapacitor electrolytes”
• “Flexible energy storage devices”
• “Recent trends in energy storage”
1.4.1.1. Inclusion and exclusion criteria. Inclusion criteria:
• Articles published in English between 2013 and 2024.
• Peer-reviewed journals, review articles, and high-impact conference proceedings.
• Studies focusing on hybrid supercapacitors, electrode/electrolyte materials, and fabrication strategies.
• Articles reporting experimental, computational, or theoretical advancements in performance metrics (e.g., capacitance, energy density, power density).
Exclusion criteria:
• Articles not directly related to hybrid supercapacitors.
• Duplicate records or preliminary notes without peer review.
• Patents, non-scientific literature, and preprints without adequate data validation.
1.4.1.2. Data extraction and analysis. From the selected articles, the following information was extracted:
• Material type (e.g., carbon-based, metal oxides, polymers, composites)
• Synthesis methods (e.g., sol–gel, CVD, hydrothermal, electrospinning)
• Electrochemical performance metrics (e.g., specific capacitance, energy/power density, cycle life)
• Electrolyte systems (e.g., ionic liquids, solid-state, aqueous)
• Device configurations (e.g., flexible/wearable, symmetric/asymmetric, hybrid systems)
Quantitative and qualitative data were then systematically categorized into thematic sections to evaluate key trends, performance improvements, scalability, and real-world applicability.
• Advanced electrode and electrolyte materials
• Synthesis and fabrication methods (sol–gel, CVD, hydrothermal, etc.)
• Role of ionic liquids and emerging electrolyte systems
• Device configurations and applications
• Future perspectives and commercialization potential
This structure ensures a holistic view of the progress in hybrid supercapacitor technologies, aiding both new researchers and experienced scientists in identifying knowledge gaps and opportunities.
2. Fundamentals of hybrid supercapacitors
2.1. Basic principles and mechanisms
Hybrid supercapacitors are advanced energy storage devices that aim to bridge the gap between conventional capacitors and batteries by combining the merits of both. The fundamental principle of HSCs lies in their ability to store and deliver energy through a combination of electric double-layer capacitance and faradaic (redox) reactions. This synergy enables HSCs to achieve higher energy densities than traditional supercapacitors while maintaining superior power densities compared to batteries.26 The basic structure of an HSC typically consists of two different types of electrodes: one with battery-like properties (faradaic) and the other with capacitor-like properties (non-faradaic). Fig. 2 graphically illustrates the charge storage mechanism in energy storage systems such as EDLCs, pseudocapacitors, batteries, and hybrid supercapacitors. EDLCs (Fig. 2a), which commonly include carbon-based electrodes such as activated carbon, CNTs, or graphene, store energy via non-faradaic processes. Specifically, energy is stored by accumulating electrostatic charges at the electrode–electrolyte interface, resulting in an electrical double layer.27 This highly reversible method enables quick charge–discharge cycles, which contributes to high power density. According to the Helmholtz model, the specific capacitance of EDLCs is mostly determined by the accessible surface area of the electrode and the effective separation distance between charged species28,29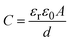 | (1) |
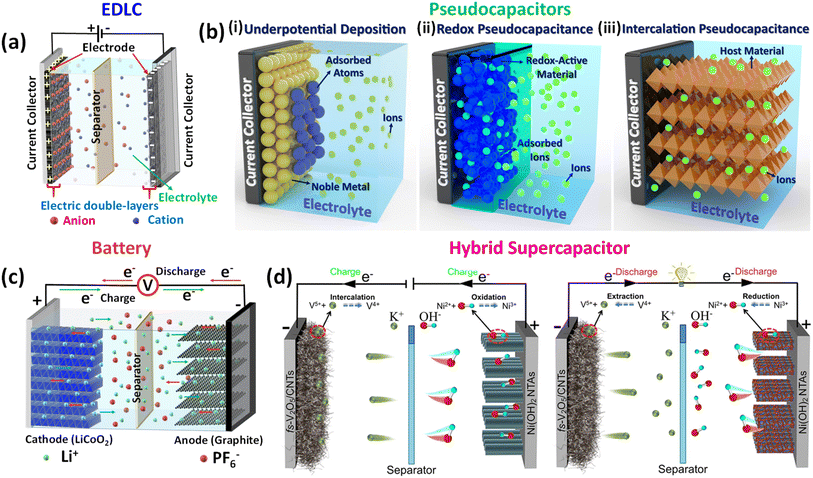 | ||
| Fig. 2 Schematic illustration of (a) electrochemical double-layer capacitor (EDLC); (b(i)) underpotential deposition, (b(ii)) redox pseudocapacitance, and (b(iii)) intercalation pseudocapacitance.45 Reproduced from ref. 45 with permission from Elsevier, copyright 2022, (c) battery, and (d) hybrid supercapacitor.46 Reproduced from ref. 46 with permission from Elsevier, copyright 2022. | ||
Pseudocapacitive behavior can be classified into three types:
(i) Underpotential deposition (Fig. 2b(i)) is the adsorption of atoms or ions on electrode surfaces at potentials higher than their equilibrium redox potential. This mechanism is common in noble metals (e.g., Pt, Au) and results in high capacitance owing to strong surface bonding.28 The general response is as follows:
| M + xCz+ + xZe− ↔ C.M | (2) |
(ii) Redox pseudocapacitance (Fig. 2b(ii)) is defined as reversible surface or near-surface redox interactions between oxidized (Oxd) and reduced (Red) species that are accompanied by cation adsorption. Typical compounds are RuO2, MnO2, and conductive polymers.28,30 This reaction is generally described as
| Oxd. + zC+ + ze− ↔ RedCz | (3) |
(iii) Intercalation pseudocapacitance (Fig. 2b(iii)) involves the insertion of cations into the structure of redox-active materials (e.g., Nb2O5, MnO2, NiO) without inducing a crystallographic phase change. This mechanism combines features of both batteries and capacitors, providing fast ion transport and good rate capability.28,31 The typical reaction is:
| MAy + xLi+ + xe− ↔ LixMAy | (4) |
Moreover, A lithium-ion battery (LIB) typically consists of a cathode, an anode, and a separator soaked with an electrolyte (Fig. 2c). Graphite is widely employed as the anode material, while lithium-rich transition metal oxides serve as cathodes. The charge–discharge process is governed by electrochemical redox reactions at the electrodes, accompanied by the reversible intercalation and de-intercalation of lithium ions into and out of the electrode host structures. This reversibility is critical to ensuring the recharge-ability and long-term cycling performance of battery. The electrolyte promotes ionic conductivity while maintaining electronic insulation, which drives electrons to move through an external circuit, allowing for the transformation of chemical energy into electrical energy. This fundamental operational principle remains consistent across various next-generation battery chemistries beyond conventional LIBs.32,33
In a standard configuration of a LIB utilizing graphite for the anode and lithium cobalt oxide (LiCoO2) for the cathode, the electrochemical reactions can be represented as follows:
| Anode: C6 + xLi+ + xe− ⇌ LixC6 | (5) |
| Cathode: LiCoO2 ⇌ Li1−xCoO2 + xLi+ + xe− | (6) |
Among state-of-the-art cathode materials in commercial LIBs are lithium nickel manganese cobalt oxides (LiNixMnyCozO2, where x + y + z = 1), LiCoO2, LiMn2O4, and LiFePO4, with graphite continuing to be the most widely used anode material.34 Modern LIBs typically demonstrate cycle lifespans that can surpass several thousand charge–discharge cycles, influenced by the particular chemistry and operational parameters.
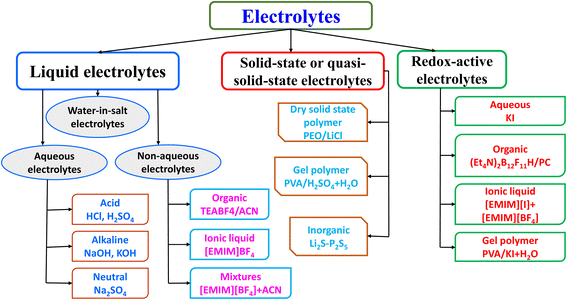
In recent years, significant advancements have been made in the design and fabrication of hybrid supercapacitors. Researchers have explored various nanostructured materials to improve the performance of HSCs. For example, the use of nanostructured metal oxides, such as MnO2 nanowires and NiO nanosheets, has been shown to enhance the electrochemical properties of battery-type electrodes by providing a larger surface area and shorter ion diffusion paths.35 Similarly, carbon-based materials like graphene and carbon nanotubes have been widely studied for their high surface area and excellent conductivity, which improve the performance of EDLC electrodes.36,37 A novel dual-ion hybrid supercapacitor (Fig. 2d) was designed by Shi et al. using Ni(OH)2 nanotube arrays as a positive electrode and V2O5 directly grown on freestanding carbon nanotubes (CNTs) as a negative electrode. Moreover, the development of advanced hybrid materials, such as MOFs and COFs, has opened new avenues for enhancing the performance of HSCs.38 These materials offer tunable porosity and high surface area, facilitating efficient ion transport and increasing the overall capacitance of the device.23,39 Electrolytes also play a crucial role in the performance of HSCs.40 Aqueous electrolytes, such as sulfuric acid and alkaline solutions (potassium hydroxide, sodium hydroxide, and lithium hydroxide), are commonly used due to their high ionic conductivity and low cost. However, the voltage window of aqueous electrolytes is limited, which restricts the energy density of HSCs.41,42 To overcome this limitation, organic electrolytes and ionic liquids, which offer a wider voltage window and higher energy density, are being increasingly explored.43,44 The basic principles and mechanisms of hybrid supercapacitors revolve around the integration of capacitor-like and battery-like materials to achieve a balanced performance in terms of energy and power density. Continued research in material science and electrochemistry is essential to further improve the efficiency and application potential of HSCs, making them a viable solution for the next generation of energy storage technologies.
2.2. Types of hybrid supercapacitors
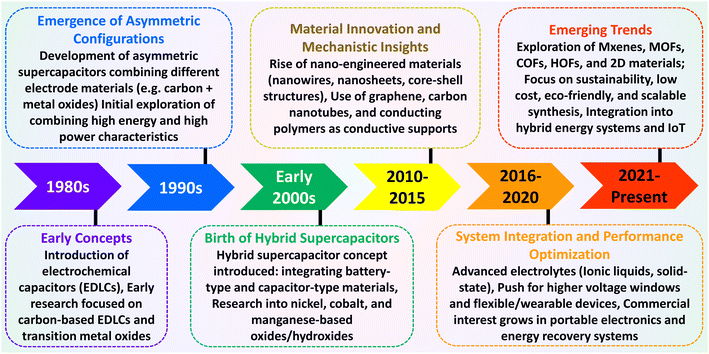
The evolution of hybrid supercapacitors (Fig. 3) has progressed significantly in recent decades, propelled by the necessity to connect high-energy batteries with high-power supercapacitors. The 1980s marked the establishment of foundational principles for electrochemical capacitors, with initial devices predominantly utilizing electric-double-layer-capacitors (EDLCs) that incorporated carbon-based materials.47 In the 1990s, the concept of asymmetric supercapacitors emerged, presenting configurations that integrated materials with varying charge storage mechanisms to improve energy density.48 The early 2000s noticed the introduction of hybrid supercapacitors, which combined faradaic (battery-type) and non-faradaic (capacitor-type) electrodes to achieve high energy and power densities concurrently.49 Over the next decade, the focus of investigation transitioned to the nanoscale engineering of electrode materials, such as metal oxides, sulfides, and carbon composites, to enhance specific capacitance, rate capability, and cycling stability.50 From 2016 to 2020, developments in electrolyte systems, device design, and flexible architectures played a significant role in enhancing performance and broadening application possibilities.51–53 In recent years, attention has shifted to sustainable synthesis methods, the investigation of innovative 2D materials like MXenes, MOFs, COFs, and HOFs materials.39,54–56 Integration into hybrid energy systems and Internet of Things (IoT), the use of machine learning for material discovery and device optimization57,58 establishing hybrid supercapacitors as promising candidates for next-generation energy storage technologies.Hybrid capacitors are classified into three categories according to their charge storage mechanism in electrodes: asymmetric, battery-type, and composite.59,60Fig. 4 illustrates the diverse structural configurations and electrochemical mechanisms underpinning hybrid supercapacitor (HSC) systems. Panel (a) presents fundamental electrode design strategies, showcasing how individual capacitive or battery-type electrodes can be combined in asymmetric configurations to exploit both high power and high energy densities. In (b), a symmetrical configuration known as Cap-Bat is depicted, where two battery-type electrodes exhibit capacitive behavior within an aqueous electrolyte, facilitating stable cycling and increased voltage windows. Panel (c) compares the charge-potential characteristics of supercapacitive and battery-type electrodes, highlighting the linear, triangular profiles typical of capacitive systems versus the plateaued profiles associated with faradaic reactions. Finally, panel (d) exemplifies an asymmetric HSC device that couples a double-layer electrode with a redox-active electrode, enabling enhanced energy storage through synergistic charge accumulation mechanisms. These design variations demonstrate how strategic electrode pairing and device architecture can significantly influence electrochemical performance, offering tailored solutions for applications ranging from portable electronics to grid-scale energy storage.61Table 1 provides an overview of the electrochemical processes happing at the anode and cathode in different types of supercapacitors, including symmetric, asymmetric, battery-type hybrid, and composite hybrid systems. To enhance the understanding of hybrid supercapacitors, we focus on the strategic selection of positive and negative electrode materials, their distinct electrochemical properties, and the synergistic effects that contribute to total device performance. The following sections thoroughly describe these issues, providing supporting analyses from CV and GCD measurements.
 | ||
| Fig. 4 Schematic representation of hybrid supercapacitor configurations and charge storage behavior. (a) Overview of various electrode arrangements, including single-mode capacitive or battery-type electrodes and their integration in asymmetric and hybrid energy devices. (b) Symmetrical hybrid configuration (Cap-Bat-type), combining two battery-type electrodes with capacitive behavior in an aqueous electrolyte environment. (c) Comparative charge-potential profiles demonstrating the distinct electrochemical characteristics of supercapacitive and battery-type electrodes. (d) Example architecture of an asymmetric hybrid supercapacitor integrating a capacitive electrode and a faradaic electrode for enhanced energy and power density. Reproduced from ref. 61 with permission from nature portfolio, copyright 2024. | ||
| S. No. | Type of supercapacitor | Anode mechanism | Cathode mechanism |
|---|---|---|---|
| 1 | Symmetric | EDLC or pseudocapacitance | EDLC or pseudocapacitance |
| 2 | Asymmetric | EDLC | Pseudocapacitance |
| 3 | Battery-type hybrid | EDLC or pseudocapacitance | Battery-type faradaic |
| 4 | Composite hybrid | EDLC or pseudocapacitance | Binary or ternary composites |
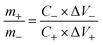 | (7) |
 | ||
| Fig. 5 Schematic MgCo2O4@MnO2//AC ASC and electrochemical characteristic CV and GCD plots.71 Reproduced from ref. 71 with permission from Springer Nature, copyright 2019. | ||
For instance, composites of graphene with metal oxides or conducting polymers have demonstrated synergistic effects, resulting in higher specific capacitance and better cycling stability.74–76 Electrolytes also play a crucial role in the performance of AHSCs. Aqueous electrolytes, such as potassium hydroxide (KOH) and sulfuric acid (H2SO4), are commonly used due to their high ionic conductivity and low cost. However, they are limited by a narrow voltage window. To overcome this limitation, organic electrolytes and ionic liquids, which offer a wider voltage window and higher energy density, are increasingly being explored.77,78 The asymmetric hybrid supercapacitors represent a versatile and efficient energy storage solution by integrating capacitive and faradaic electrodes. The continued research and development in this field, focusing on advanced materials and optimized electrode designs, holds promise for further improving the performance and application potential of AHSCs.
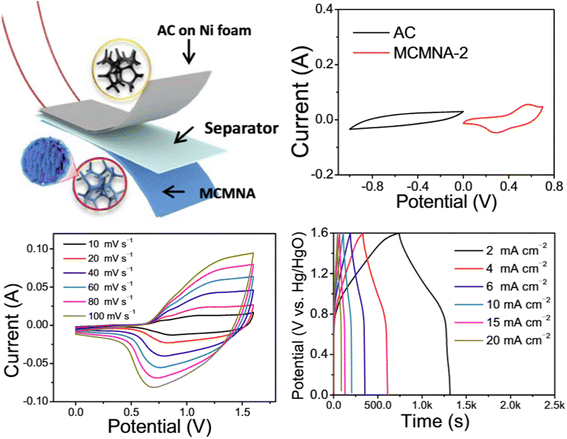
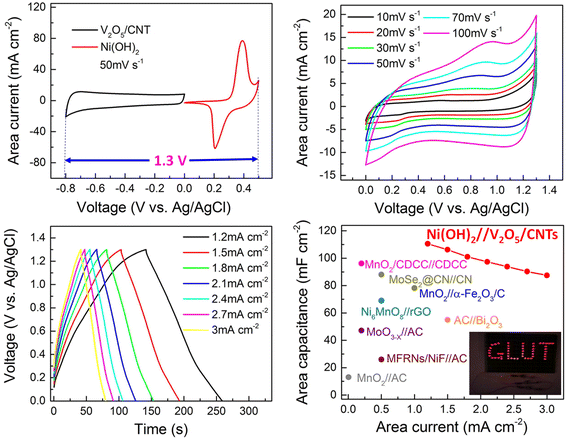
The electrolyte selection is another critical to the optimization of BHS systems. An ideal electrolyte must be electrochemically stable and compatible with both electrode types in order to maintain balanced charge storage kinetics. Electrolyte incompatibility may cause polarization and asymmetric charge transfer, resulting in decreased performance and device deterioration. Recent research has emphasized the benefits of utilizing aqueous electrolytes in BHS, especially in systems with metal oxide or hydroxide electrodes. These electrolytes provide strong ionic conductivity, environmental friendliness, and cheaper cost than organic or ionic liquid-based systems, while still giving suitable voltage windows when combined with appropriate electrode materials. For example, Shi et al. designed a potassium-based dual-ion hybrid supercapacitor (DHSC), which gives a striking illustration of how combining battery-type and pseudocapacitor-type electrodes may considerably improve energy storage performance. In this device, Ni(OH)2 nanotube arrays operate as the cathode, leveraging OH− ions for battery-type faradaic processes, while V2O5-anchored carbon nanotubes serve as the anode, exploiting K+ ions via pseudocapacitive mechanisms. This beneficial interaction between dual-ion species enables efficient charge storage and transportation, leading in high energy density and operating voltage. The combination of a 2 M KOH aqueous electrolyte with a quasi-solid-state structure enhances the safety, flexibility, and mechanical integrity of the device. The electrochemical performance and practical application of the DHSC device is shown in Fig. 6. The charge storage mechanism in the device involves the reversible insertion/extraction of K+ and OH− ions at the electrodes. This strategic design not only improves electrochemical performance, but also demonstrates the practicality of adaptable, high-performance energy storage systems. The DHSC operates at 1.3 V, has a maximum energy density of 25.4 μW h cm−2, and retains 81.4% capacity after 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles. Furthermore, the gadget performs reliably under bending stress and can power realistic loads such as 50 LEDs when connected in series.46
000 cycles. Furthermore, the gadget performs reliably under bending stress and can power realistic loads such as 50 LEDs when connected in series.46
 | ||
| Fig. 6 Electrochemical performance of the battery-type hybrid supercapacitor based on Ni(OH)2 and free-standing V2O5 on CNTs with 1.3 V voltage in alkaline (KOH) electrolyte.46 Reproduced from ref. 46 with permission from Elsevier, copyright 2022. | ||
Furthermore, the adaptability of BHS design allows for many classifications depending on the active ions and electrolytes utilized. These include lithium-ion and sodium-ion hybrid supercapacitors, acidic and alkaline BHS systems, BHS with redox-active electrolytes, and those that use pseudocapacitive electrodes.80 Each design has distinct benefits in terms of energy storage capacity, operating voltage, and cycle stability, allowing for customized solutions for a wide range of applications, from portable electronics to grid-level energy management. Even though its promising characteristics, the commercialization of BHS faces various hurdles. These include increasing the cycle endurance of faradaic electrodes, optimizing the interface between the two electrode types, and scaling up synthesis procedures to produce consistent and cost-effective materials. Continued research into enhanced material design, electrode/electrolyte interface engineering, and system integration will be essential for fully realizing the promise of battery-type hybrid supercapacitors in next-generation energy storage systems.
Li et al. report a significant progression in the improvement of composite hybrid supercapacitors by designing a novel positive electrode composed of polyaniline integrated with a three-dimensional porous Ti3C2Tx MXene framework. This innovative composite structure effectively tackles the prevalent issue of MXene oxidation at positive potentials by increasing the work function through the integration of PANI, as supported by first-principle calculations. The resulting PANI@Ti3C2Tx MXene electrode exhibits an impressive volumetric capacitance of 1632 F cm−3 at a scan rate of 10 mV s−1, and retains a notable 827 F cm−3 even at a high scan rate of 5000 mV s−1. Fig. 7a illustrates the CV curves of both electrodes, along with the GCD plots of the assembled Ti3C2Tx MXene//PANI@Ti3C2Tx MXene hybrid supercapacitor. The device delivers a remarkable energy density of 50.6 W h L−1 and an exceptional power density of 127 kW L−1. The results demonstrate the capability of polymer@MXene composites to improve the performance of hybrid supercapacitors, presenting a promising avenue for advancing high-energy and high-power energy storage systems.82
 | ||
| Fig. 7 (a) Electrochemical performance (CV and GCD curves) of M-Ti3C2Tx//PANI@M-Ti3C2Tx hybrid supercapacitor,82 Reproduced from ref. 82 with permission from Wiley, copyright 2019. (b) The schematic representation and electrochemical profiles (CV and GCD curves) of the Ni-NMO-rGO-CoO//AC all-solid-state hybrid supercapacitor.83 Reproduced from ref. 83 with permission from John Wiley and Sons Ltd., copyright 2022. | ||
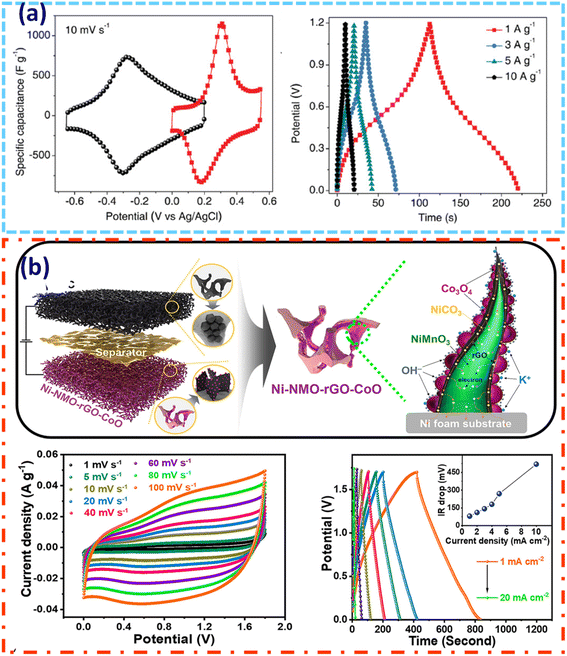
However, Ranjith et al. successfully fabricated a high-performance ternary composite electrode composed of NiCO3/NiMnO3-rGO-Co3O4 (Ni-NMO-rGO-CoO) nanoflake arrays (NFAs) on a conductive 3D nickel foam substrate via a two-step hydrothermal process. As shown in Fig. 7b, the schematic design and electrochemical profiles clearly indicate the efficient charge storage behavior and outstanding performance of this ternary composite hybrid supercapacitor. This hierarchical, layer-by-layer structure intelligently blends three functional components to provide synergistic advantages in composite hybrid supercapacitor applications. Reduced graphene oxide acts as a flexible and conductive interlayer, allowing volume variations during cycling and improving structural stability. Co3O4 nanograins are evenly dispersed across the rGO-wrapped NiMnO3 matrix, promoting fast ion transport and increasing electrochemical activity. The designed Ni-NMO-rGO-CoO ternary composite has a high specific capacity of 188.8 mA h g−1 at 1 mA g−1. The Ni-NMO-rGO-CoO//AC hybrid supercapacitor has an exceptional energy density of 57.2 W h kg−1 and a power density of 472.2 W kg−1. It also has outstanding cycling stability, keeping 97.7% of its capacitance after 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles, demonstrating its potential for next-generation portable energy storage technologies.83
000 cycles, demonstrating its potential for next-generation portable energy storage technologies.83
Moreover, the fibrous, woven structure of CNTs acts as an excellent conductive framework for the consistent development of PPy, promoting effective transport of electrons and ions. Furthermore, this architecture facilitates a three-dimensional charge distribution, optimizing the use of active materials while reducing internal resistance. The mechanical strength of the CNT backbone effectively reduces the volumetric changes typically observed in pseudocapacitive polymers during charge/discharge cycles. The integration of these structures results in improved cycle life and mechanical stability, effectively connecting the high energy density characteristic of batteries with the rapid response capabilities of supercapacitors. For example, investigations have demonstrated that CNT-PPy composites can maintain more than 90% of their capacitance after 5000 cycles, exhibiting stability comparable to that of conventional EDLC systems.79 Similarly, hybrid electrodes based on reduced graphene oxide and metal oxides like MnO2 have demonstrated specific capacitances that exceed 300 F g−1, significantly outperforming the individual components due to the synergistic interaction between EDLC and pseudocapacitance.84 Composite designs facilitate the adjustment of energy and power densities while providing the opportunity to engineer electrode architecture tailored to specific applications, including wearable electronics and high-voltage asymmetric supercapacitors. Ongoing progress in material integration, nanoscale engineering, and interfacial optimization is anticipated to significantly improve the performance and scalability of these hybrid supercapacitor systems.
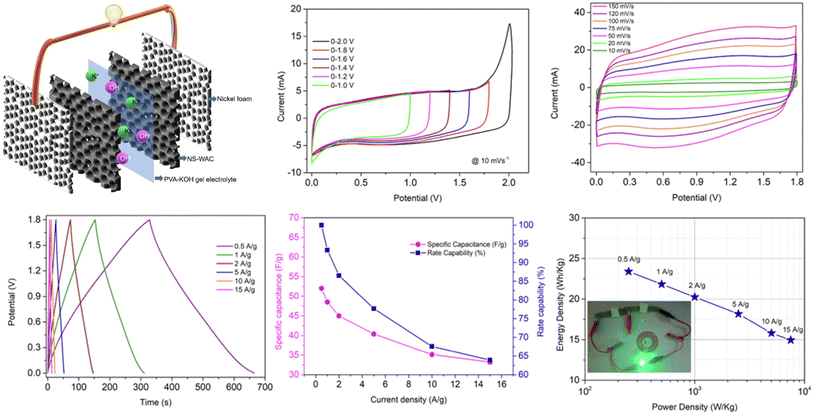
Anand et al. recently constructed a symmetric solid-state supercapacitor utilizing nitrogen and sulfur co-doped activation carbon (NS-WAC), sourced from economical walnut shell biomass, which demonstrates promising electrochemical performance. As shown in Fig. 8, the device architecture consists of NS-WAC electrodes coated on nickel foam with a PVA-KOH gel electrolyte. The CV and GCD profiles reveal a combination of electric double-layer capacitance and pseudocapacitive behavior, with high stability across varying scan rates and current densities. The SSC device achieves a specific capacitance of 52 F g−1 at 0.5 A g−1, with 1.8 V voltage, and delivers a maximum energy density of 23.4 W h kg−1 at a power density of 250 W kg−1, along with excellent cycling durability (91.8% retention after 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles). Furthermore, a volumetric capacitance of 74.36 F cm−3 reflects its high packing density, supporting its suitability for compact and portable energy storage applications.86
000 cycles). Furthermore, a volumetric capacitance of 74.36 F cm−3 reflects its high packing density, supporting its suitability for compact and portable energy storage applications.86
 | ||
| Fig. 8 Schematic representation of NS-WAC//NS-WAC device fabrication and their electrochemical performance. Reproduced from ref. 86 with permission from Elsevier, copyright 2024. | ||
For instance, a high-voltage (2.7 V) symmetric supercapacitors utilizing nitrogen-doped graphene materials have shown remarkable performance improvements due to enhanced electrical conductivity and increased active sites for charge storage.87 Recent advancements have focused on developing nanocomposites that synergize the properties of different materials. For example, composites of graphene and transition metal oxides, such as MnO2, RuO2, SnO2, and Co3O4, exhibit both high capacitance and excellent cycling stability, making them ideal candidates for SHSC electrodes.88–90 Another promising direction in SHSC research is the incorporation of MOFs and COFs. These materials provide high surface areas, tunable porosity, and customizable functionality, which are beneficial for enhancing the capacitive and faradaic performance of the supercapacitor. Studies have shown that MOF-derived carbon materials can achieve high energy densities while maintaining superior rate capabilities.91–93 In addition, the electrolyte also plays a crucial role in the performance of SHSCs. Aqueous electrolytes, while providing high ionic conductivity, are often limited by their narrow electrochemical window. To address this, organic electrolytes and ionic liquids, which offer wider voltage windows and better stability, are increasingly being utilized in SHSCs to achieve higher energy densities.87,94 In terms of practical applications, SHSCs have been demonstrated to be effective in various energy storage systems, including portable electronics and electric vehicles. Their ability to deliver high power outputs with moderate energy densities makes them suitable for applications requiring quick charge and discharge cycles.95 So, symmetric hybrid supercapacitors represent a promising class of energy storage devices that leverage the combined advantages of EDLC and pseudocapacitance. Continued research focusing on advanced materials, innovative composites, and optimized electrolytes has the potential to further enhance the performance and application range of SHSCs, making them a viable solution for future energy storage challenges.
2.3. Comparison with conventional capacitors and batteries
Hybrid supercapacitors combine elements from both supercapacitors and batteries to bridge the performance gap between these two traditional energy storage technologies. Understanding their distinctions and advantages requires a comprehensive comparison with conventional supercapacitors and batteries regarding energy density, power density, cycle life, and operational characteristics.Conventional capacitors, often called electrochemical capacitors, are characterized by their high power and low energy densities. They store energy through electrostatic charge separation at the electrode–electrolyte interface, which provides rapid charge and discharge capabilities but limits the total energy stored per unit volume.96 In contrast, batteries store energy through chemical reactions, leading to higher energy densities but at the cost of lower power density and slower charge–discharge rates.97,98 Hybrid supercapacitors aim to combine the best of both worlds by integrating materials and mechanisms that enhance both energy and power densities.
For instance, asymmetric hybrid supercapacitors use a combination of capacitive and pseudocapacitive materials, resulting in improved energy storage capabilities while maintaining high power delivery.99 These configurations allow HSCs to achieve intermediate energy densities compared to conventional supercapacitors and batteries. Capacitors generally exhibit superior cycle life compared to batteries, thanks to their electrostatic charge storage mechanism, which involves less degradation of materials over numerous charge–discharge cycles.100 Batteries, particularly lithium-ion types, tend to degrade more quickly due to the chemical changes occurring during each cycle, leading to reduced cycle life and performance.101 Hybrid supercapacitors, leveraging the advantages of both systems, tend to offer a balance in cycle life. While they do not match the cycle life of traditional supercapacitors, they generally outperform batteries in this regard. Advanced materials and electrode designs in HSCs are continuously being developed to further enhance their cycle stability.102,103 The operational voltage range of conventional capacitors is often limited to a lower voltage window due to electrolyte constraints. This limitation affects their energy density and overall performance. Batteries, however, can operate over a broader voltage range but face issues with energy efficiency and safety.104 Hybrid supercapacitors address these challenges by employing advanced electrolytes and electrode materials that support wider voltage windows and improved safety profiles. For example, HSCs using high-voltage electrolytes or hybrid materials can extend their operational voltage range while maintaining efficient energy storage and delivery.105
➢ In contrast to batteries, hybrid supercapacitors have a significantly wider working temperature range. Batteries can operate at +50 °C; however, hybrid supercapacitors can work safely at +80 °C, making them more appropriate for usage in very hot conditions.106,107
➢ Hybrid supercapacitors perform better not just at high temperatures but also at low temperatures. They may help systems or devices operate with steady performance even at −30 °C, whereas batteries have difficulty working at −5 °C.107
➢ Hybrid supercapacitors charge much more quickly than batteries. Typically, this occurs within seconds. Battery specialists use the C-rate to measure how quickly a battery charges or discharges. Experts generally assess the C-rate of hybrid supercapacitors at 20 °C, which is around 7 times quicker than the speed of batteries.108
➢ Hybrid supercapacitors provide substantially better power density than batteries. In this regard, batteries are at a disadvantage because the chemical processes that store and release energy take time.95
➢ Hybrid supercapacitors are more environmentally friendly than batteries in terms of the materials used, production process, and disposal. They also have fewer hazardous compounds or heavy metals than batteries.109
➢ The chemical potential has a far higher energy storage capacity than static electricity. Applications requiring more storage capacity often use batteries.65
➢ Hybrid supercapacitors have notably longer life cycles than batteries. A conventional battery is capable of sustaining 3500 charge/discharge cycles; however, a hybrid supercapacitor can often withstand over 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles. Due to a broad operating temperature range and eco-friendly components, this may result in significant material and cost savings.110,111
000 cycles. Due to a broad operating temperature range and eco-friendly components, this may result in significant material and cost savings.110,111
➢ When compared to batteries, hybrid supercapacitors are far safer and less prone to burst.79
The choice between supercapacitors, batteries, and hybrid systems often depends on the specific application requirements. Conventional supercapacitors are ideal for applications requiring rapid energy discharge and high power output, such as regenerative braking systems in vehicles. Batteries are suited for applications needing high energy storage, like portable electronics and electric vehicles. Hybrid supercapacitors, with their balanced performance characteristics, find applications in scenarios requiring both high power and moderate energy density, such as in grid energy storage systems and load leveling. So, hybrid supercapacitors offer a compelling alternative to conventional supercapacitors and batteries by integrating strengths from both technologies. They provide improved energy density while maintaining high power density and stability, making them suitable for diverse and demanding applications. Continued research into advanced materials and design strategies will further enhance the performance and applicability of hybrid supercapacitors.
In conclusion, hybrid supercapacitors (HSCs) have emerged as a promising class of energy storage devices that effectively bridge the performance gap between traditional supercapacitors and batteries. By synergistically combining electric double-layer capacitance with faradaic redox reactions, HSCs offer an optimal balance of high power density, long cycle life, and enhanced energy density. Their versatility in design ranging from asymmetric and symmetric configurations to advanced composite systems along with the integration of nanostructured materials such as MOFs, COFs, and heteroatom-doped carbons, has significantly boosted their electrochemical performance. Furthermore, the strategic selection of electrolytes plays a pivotal role in maximizing energy output and operational stability. Collectively, these advancements position HSCs as highly efficient and scalable solutions for future applications in portable electronics, electric mobility, and grid-level energy storage.
3. Electrode materials for hybrid supercapacitors
3.1. Carbon-based materials
The incorporation of carbon-based materials in electrochemical energy storage, especially in supercapacitors, has undergone incredible innovations from the early 19th century to the current era. Initial investigations concentrated on the conductive characteristics of graphite in voltaic cells, establishing a foundation for subsequent advancements. During the 1970s and 1980s, a significant development occurred with the introduction of supercapacitors that utilized activated carbon, recognized for its substantial surface area and outstanding electrical characteristics.112,113 Improvements continued throughout the 1990s, which included an increasing interest in innovative carbon structures like carbon nanotubes (CNTs) and graphene. The unique structural and electrical features of these materials expand the potential applications of carbon in energy storage. Carbon exhibits outstanding chemical versatility and is capable of forming a diverse array of allotropes such as graphite, diamond, fullerenes, nanotubes, and graphene.114–118 The structures display unique dimensionalities and morphologies, which have been the subject of thorough scientific exploration.Carbon, being a prevalent and crucial component of the Earth's crust, is notable for its capacity to create materials that exhibit high electrical conductivity, considerable surface-to-volume ratios, and versatile structural characteristics. The characteristics of carbon-based materials render them particularly advantageous for supercapacitor electrodes, focusing on the attainment of high energy density and swift charge–discharge performance.119,120 Furthermore, carbon materials are appreciated for their lightweight characteristics, thermal and chemical stability, cost-effectiveness, and compatibility with the environment. The performance of carbon-based electrodes can be optimized by adjusting factors like porosity, surface area, graphitization degree, the presence of functional groups, and doping with heteroatoms. The modifications improve electrochemical properties and enable customized functionality for particular storage applications.121–123 However, despite the many benefits, a key challenge persists: enhancing the interaction between carbon materials and electrolytes to boost energy density, power density, and long-term stability in cutting-edge supercapacitor systems. To provide a comprehensive understanding of the electrochemical performance and structural advantages of carbon-based materials in hybrid supercapacitors, the subsequent sections critically examine three representative classes: activated carbon, graphene and graphene oxide, and carbon nanotubes.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4 ratio of sugarcane bagasse and KOH sources, achieving 3554 m2 g−1 of surface area at 800 °C, and significantly increasing charge storage capacity.124 Li et al. demonstrated that AC with a well-developed microporous structure achieved a specific surface area exceeding 3000 m2 g−1, leading to a notable enhancement in charge storage capacity.125 In recent decades, the techniques of synthesizing AC have enabled the production of materials with specific surface areas of up to 3000–4000 m2 g−1, which have shown considerable promise in energy storage applications because of their unique physical and chemical characteristics.126–128
4 ratio of sugarcane bagasse and KOH sources, achieving 3554 m2 g−1 of surface area at 800 °C, and significantly increasing charge storage capacity.124 Li et al. demonstrated that AC with a well-developed microporous structure achieved a specific surface area exceeding 3000 m2 g−1, leading to a notable enhancement in charge storage capacity.125 In recent decades, the techniques of synthesizing AC have enabled the production of materials with specific surface areas of up to 3000–4000 m2 g−1, which have shown considerable promise in energy storage applications because of their unique physical and chemical characteristics.126–128
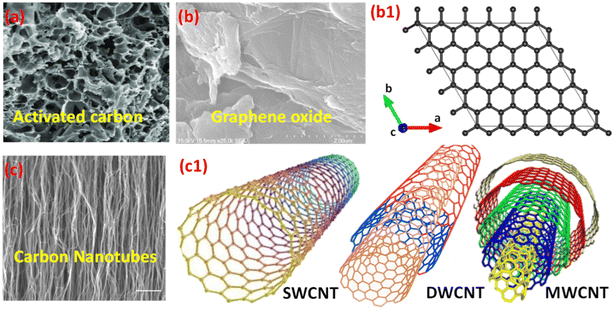 | ||
| Fig. 9 (a) SEM image of activated carbon,115 Reproduced from ref. 115 with permission from Elsevier, copyright 2023; (b & b1) SEM image of graphene oxide and its schematic structure using vesta software; (c) SEM image of carbon nanotubes,116 Reproduced from ref. 116 with permission from Springer Nature, copyright 2021, and (c1) schematic structures of SWCNT,118 DWCNT, and MWCNT,118 Reproduced from ref. 118 with permission from Springer Nature, copyright 2021. | ||
The electrochemical performance of activated carbon in hybrid supercapacitors is largely governed by its electrical conductivity and the effective utilization of its surface area. Recent advancements have focused on improving AC's conductivity by incorporating conductive additives or modifying the carbon structure. Chen et al. reported that doping AC with nitrogen and sulfur elements enhanced its electrical conductivity and capacitance performance, achieving a specific capacitance of 275 F g−1 in a hybrid supercapacitor configuration. This modification not only improves the charge storage capability but also contributes to better overall device performance.129 Hilal Pecenek et al. (2023) worked on biomass-derived activated carbon as a negative electrode for hybrid supercapacitors. It is synthesized from orange peels via a sequential carbonization and activation process. The device delivered a high specific capacitance of 330 F g−1, excellent energy density of 244.8 W h kg−1 and power density of 1.15 kW kg−1 at a current density of 1 A g−1.130 Yanhua Li et al., used a two-step KOH activation approach to create three-dimensionally O-doped, interconnected hierarchical porous carbons with an ultrahigh surface area of up to 4048.2 m2 g−1. It can achieve 419 F g−1 at 0.5 A g−1 and has high cycling stability, with 90.1% retention even after 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles at 10 A g−1 in a three-electrode configuration. In contrast, a two-electrode system exhibits 30.3 W h kg−1 of energy density at 250 W kg−1 and 16.0 W h kg−1 of energy density at higher power density, i.e., 5000 W kg−1.131 Soheil et al. found that sulfur-doping activated carbon increases electrochemical performance by 440 F g−1 at 1 A g−1 and 90% of capacitance retention at 20 A g−1 after 5000 cycles.132 These modifications not only improves the charge storage capability but also contributes to better overall device performance. Using a hydrothermal technique, Susmitha et al. synthesized nitrogen and sulfur-co-doped activated carbon nanosheets (NSAC) for electrochemical supercapacitors. At 0.7 A g−1, the symmetric supercapacitor, produced 417 F g−1 of specific capacitance and demonstrated outstanding cycling stability at 5 A g−1, retaining 76% of its original capacitance after 60
000 cycles at 10 A g−1 in a three-electrode configuration. In contrast, a two-electrode system exhibits 30.3 W h kg−1 of energy density at 250 W kg−1 and 16.0 W h kg−1 of energy density at higher power density, i.e., 5000 W kg−1.131 Soheil et al. found that sulfur-doping activated carbon increases electrochemical performance by 440 F g−1 at 1 A g−1 and 90% of capacitance retention at 20 A g−1 after 5000 cycles.132 These modifications not only improves the charge storage capability but also contributes to better overall device performance. Using a hydrothermal technique, Susmitha et al. synthesized nitrogen and sulfur-co-doped activated carbon nanosheets (NSAC) for electrochemical supercapacitors. At 0.7 A g−1, the symmetric supercapacitor, produced 417 F g−1 of specific capacitance and demonstrated outstanding cycling stability at 5 A g−1, retaining 76% of its original capacitance after 60![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles.133 Subir et al. produced porous activated carbon (PAC) from petroleum coke and activated it with KOH, yielding 2105.6 m2 g−1 of specific surface area. The stacked layers of carbon atoms in a two-dimensional hexagonal structure provide 470 F g−1 of specific capacitance at 0.5 A g−1 and ultra-stable performance at 10 A g−1, retaining 98% capacitance even after 15
000 cycles.133 Subir et al. produced porous activated carbon (PAC) from petroleum coke and activated it with KOH, yielding 2105.6 m2 g−1 of specific surface area. The stacked layers of carbon atoms in a two-dimensional hexagonal structure provide 470 F g−1 of specific capacitance at 0.5 A g−1 and ultra-stable performance at 10 A g−1, retaining 98% capacitance even after 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles.134 Anant et al. investigated the biomass of Phyllanthus emblica leaves to produce activated carbon as an electrode material for supercapacitors. The developed material has a large surface area of 1244 m2 g−1, which results in 336 F g−1 of specific capacitance at 1 A g−1 and strong cycle stability. Furthermore, a symmetric supercapacitor provides 29 W h kg−1 of maximum energy density and 6750 W kg−1 of maximum power density, as well as exceptional cycling stability, i.e., 95% retention at 16 A g−1 for up to 25
000 cycles.134 Anant et al. investigated the biomass of Phyllanthus emblica leaves to produce activated carbon as an electrode material for supercapacitors. The developed material has a large surface area of 1244 m2 g−1, which results in 336 F g−1 of specific capacitance at 1 A g−1 and strong cycle stability. Furthermore, a symmetric supercapacitor provides 29 W h kg−1 of maximum energy density and 6750 W kg−1 of maximum power density, as well as exceptional cycling stability, i.e., 95% retention at 16 A g−1 for up to 25![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles135 Activated carbon exhibits remarkable chemical stability and durability, making it a suitable choice for long-term applications in hybrid supercapacitors. The resistance to degradation and capacity loss over numerous charge–discharge cycles is a key factor in the selection of AC as an electrode material. The aforementioned investigations have shown that AC-based electrodes maintain high capacitance and stable performance even after extensive cycling.
000 cycles135 Activated carbon exhibits remarkable chemical stability and durability, making it a suitable choice for long-term applications in hybrid supercapacitors. The resistance to degradation and capacity loss over numerous charge–discharge cycles is a key factor in the selection of AC as an electrode material. The aforementioned investigations have shown that AC-based electrodes maintain high capacitance and stable performance even after extensive cycling.
In hybrid supercapacitors, activated carbon is often paired with pseudocapacitive materials to achieve a balance between high energy and power densities. This combination leverages the high surface area of AC and the faradaic charge storage of materials such as transition metal oxides or conductive polymers. Recent studies have explored various hybrid configurations, such as AC combined with manganese dioxide or polyaniline, to optimize performance metrics. Wang et al. reported that combining AC with MnO2 led to a hybrid supercapacitor with an energy density of 30 W h kg−1 and a power density of 10 kW kg−1, showcasing the effectiveness of such combinations.136 Young-Geun Lee et al. prepared phosphorus and boron codoped activated carbon to improve the wettability and electrical properties to enhance the hybrid supercapacitor's performance. The arranged zinc-ion hybrid supercapacitors exhibited a superior electrochemical performance with an excellent specific capacity of 169.4 mA h g−1 at 0.5 A g−1 and outstanding ultrafast longevity indicated by 88% capacity retention for up to 30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles at 10 A g−1.137 Imen et al. created reduced graphene oxide nanosheets and activated carbon from olive stones with polyaniline surface decoration to build supercapacitors. The PAni@OS-rGO (1
000 cycles at 10 A g−1.137 Imen et al. created reduced graphene oxide nanosheets and activated carbon from olive stones with polyaniline surface decoration to build supercapacitors. The PAni@OS-rGO (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) has a specific capacitance of 582.6 F g−1 at 0.1 A g−1. At 7.0 A g−1, the particular capacitance remained at 312.5 F g−1, indicating a high retention rate of 53.64%. The material's excellent cycling stability, with a capacity retention of 90.07% after 3000 cycles, demonstrates its potential as a supercapacitor electrode.138 Salama et al. used a simple hydrothermal process to produce magnesium ferrites supported by activated carbon from orange peels. The MF45-AC electrode material exhibited remarkable electrochemical performance, with a specific capacitance of 870 F g−1 at a current density of 1 A g−1. The constructed electrodes were fairly stable throughout cycling. Even after 5000 cycles, AC, MF, and MF45-AC retained 89.6%, 94.2%, and 95.1% of their original specific capacitance, respectively.139 Witecka et al. effectively fabricated activated carbon/iron oxide (90 wt%:10 wt%; ACF) composite electrodes with chitosan as a binder for supercapacitor applications using electrophoretic deposition. ACF electrodes were employed in a symmetric two-electrode cell configuration utilizing a gel polymer electrolyte, PVdF(HFP)-PC-Mg(ClO4)2, demonstrating a specific capacitance of 54.4 F g−1, alongside commendable energy and power densities of 4.7 W h kg−1 and 1.2 kW kg−1, respectively. In addition, it exhibits remarkable electrochemical stability (91.5%) for approximately 10
1) has a specific capacitance of 582.6 F g−1 at 0.1 A g−1. At 7.0 A g−1, the particular capacitance remained at 312.5 F g−1, indicating a high retention rate of 53.64%. The material's excellent cycling stability, with a capacity retention of 90.07% after 3000 cycles, demonstrates its potential as a supercapacitor electrode.138 Salama et al. used a simple hydrothermal process to produce magnesium ferrites supported by activated carbon from orange peels. The MF45-AC electrode material exhibited remarkable electrochemical performance, with a specific capacitance of 870 F g−1 at a current density of 1 A g−1. The constructed electrodes were fairly stable throughout cycling. Even after 5000 cycles, AC, MF, and MF45-AC retained 89.6%, 94.2%, and 95.1% of their original specific capacitance, respectively.139 Witecka et al. effectively fabricated activated carbon/iron oxide (90 wt%:10 wt%; ACF) composite electrodes with chitosan as a binder for supercapacitor applications using electrophoretic deposition. ACF electrodes were employed in a symmetric two-electrode cell configuration utilizing a gel polymer electrolyte, PVdF(HFP)-PC-Mg(ClO4)2, demonstrating a specific capacitance of 54.4 F g−1, alongside commendable energy and power densities of 4.7 W h kg−1 and 1.2 kW kg−1, respectively. In addition, it exhibits remarkable electrochemical stability (91.5%) for approximately 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles. These findings validated the stability of the employed technology and its prospective use in energy storage and conversion.140 Simran et al. used a hydrothermal technique to synthesize MnCo2O4/AC/PPy ternary composites. The prepared ternary composite (MAP-20) outperformed virgin MnCo2O4 with a capacitance of 838.8 F g−1 at 4 A g−1. The asymmetric device with MAP-20 and MnO2/AC electrodes achieved a remarkable energy density of 88.12 W h kg−1 at 1.6 kW kg−1. The asymmetric device also achieved an impressive cyclic performance of 89.68% over 10
000 cycles. These findings validated the stability of the employed technology and its prospective use in energy storage and conversion.140 Simran et al. used a hydrothermal technique to synthesize MnCo2O4/AC/PPy ternary composites. The prepared ternary composite (MAP-20) outperformed virgin MnCo2O4 with a capacitance of 838.8 F g−1 at 4 A g−1. The asymmetric device with MAP-20 and MnO2/AC electrodes achieved a remarkable energy density of 88.12 W h kg−1 at 1.6 kW kg−1. The asymmetric device also achieved an impressive cyclic performance of 89.68% over 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles. The real-time applicability of the device was also tested by turning ON a 39-red LED panel. For around 45 minutes, three asymmetric cells linked in series lighted the display. All of these results point to the fact that mixing several effective electrode materials in a manner that works well together increases supercapacitor electrochemical performance.141 Shah et al. provide a novel two-step pyrolysis technique for producing activated carbon nanosheets from jute sticks (JAC). The optimized JAC-2 material has a high yield (24%) and specific surface area (2601 m2 g−1). In addition, they adopt a novel in situ synthesis approach to create NiCoLDH-1@JAC-2 hybrid nanocomposites by merging JAC nanosheets with nickel–cobalt-layered double hydroxide nanoflowers. These nanocomposites act as positive electrode material in all-solid-state asymmetric hybrid supercapacitors, whereas JAC-2 acts as a negative electrode material, resulting in impressive performance metrics. Using a PVA/KOH solid electrolyte, the HSCs attain a specific capacitance of 750 F g−1 at 0.5 A g−1, and an energy density of 100 W h kg−1 at 250 W kg−1 of power density. They also exhibit excellent cycle stability (95% even after undergoing 10
000 cycles. The real-time applicability of the device was also tested by turning ON a 39-red LED panel. For around 45 minutes, three asymmetric cells linked in series lighted the display. All of these results point to the fact that mixing several effective electrode materials in a manner that works well together increases supercapacitor electrochemical performance.141 Shah et al. provide a novel two-step pyrolysis technique for producing activated carbon nanosheets from jute sticks (JAC). The optimized JAC-2 material has a high yield (24%) and specific surface area (2601 m2 g−1). In addition, they adopt a novel in situ synthesis approach to create NiCoLDH-1@JAC-2 hybrid nanocomposites by merging JAC nanosheets with nickel–cobalt-layered double hydroxide nanoflowers. These nanocomposites act as positive electrode material in all-solid-state asymmetric hybrid supercapacitors, whereas JAC-2 acts as a negative electrode material, resulting in impressive performance metrics. Using a PVA/KOH solid electrolyte, the HSCs attain a specific capacitance of 750 F g−1 at 0.5 A g−1, and an energy density of 100 W h kg−1 at 250 W kg−1 of power density. They also exhibit excellent cycle stability (95% even after undergoing 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 GCD cycles).142
000 GCD cycles).142
Recent innovations in the field have focused on enhancing the performance of AC-based electrodes through advanced processing techniques and functionalization. For example, novel methods like microwave-assisted activation and plasma treatment have been employed to create high-performance AC electrodes with tailored properties. These advancements enable better control over pore size distribution and surface chemistry, further improving the electrochemical characteristics of AC-based electrodes.143,144 AC plays a crucial role in the development of hybrid supercapacitors due to its exceptional properties, particularly its high surface area, good electrical conductivity, and chemical stability. The porous structure of activated carbon provides an extensive surface area, facilitating efficient charge storage through electrostatic interactions. This characteristic is particularly beneficial in supercapacitors, where rapid charge–discharge cycles are essential for high power density. The ability of activated carbon to store a large amount of charge in its pores without undergoing significant structural changes makes it a durable material with excellent cyclic stability, which is critical for long-term energy storage applications. In addition to its surface area, the conductivity of activated carbon enables the efficient transport of ions and electrons during the charge–discharge process. This conductivity, while not as high as in metals or certain advanced nanomaterials like graphene, is sufficient for most energy storage applications when combined with its large surface area. The stability of activated carbon, particularly its resistance to chemical degradation in various electrolytes, further enhances its suitability for supercapacitors, making it a reliable and sustainable choice in both aqueous and organic electrolyte systems. Ongoing research efforts are focused on improving the performance of activated carbon in hybrid supercapacitors by exploring novel synthesis methods and surface modifications. For instance, researchers are investigating the incorporation of heteroatoms such as nitrogen, oxygen, or sulfur into the carbon matrix to enhance the electrochemical activity of the material. These dopants can introduce additional active sites for charge storage, increase conductivity, and improve the overall energy density of the supercapacitor. In addition, advances in pore structure engineering, such as the development of hierarchical porosity (micro-, meso-, and macro-pores), aim to optimize ion transport and storage, allowing for faster ion diffusion and enhanced charge storage capabilities.
Another area of innovation involves the integration of activated carbon with other materials, such as transition metal oxides, conducting polymers, and graphene, to create hybrid electrodes. These composite materials can synergistically combine the advantages of different components, such as the high capacitance of metal oxides and the fast charge–discharge properties of activated carbon. The resulting hybrid materials offer enhanced performance, including higher energy and power densities, improved cycle life, and better overall stability, making them attractive for next-generation energy storage devices. Looking forward, future research is expected to focus on developing cost-effective and environmentally friendly methods for producing activated carbon with tailored properties. Biomass-derived activated carbon, produced from sustainable sources like agricultural waste, is gaining attention as a green alternative to traditional carbon precursors. Additionally, new processing techniques, such as template-assisted synthesis and chemical activation, are being explored to further control the porosity, surface chemistry, and morphology of activated carbon materials. So the activated carbon continues to be a key material in the field of hybrid supercapacitors due to its unique combination of high surface area, conductivity, and stability. As research progresses, the development of advanced processing techniques and hybrid material strategies will likely lead to even greater improvements in the energy storage capabilities of activated carbon-based supercapacitors, positioning them as essential components in the future of energy storage technologies.
Graphene, a single layer of carbon atoms arranged in a hexagonal lattice (Fig. 9b1), is renowned for its remarkable electrical conductivity, mechanical strength, and high surface area. As an electrode material in hybrid supercapacitors, graphene contributes to both high capacitance and power density. Recent research has focused on improving graphene's performance through various methods, including chemical doping and nano-structuring.147
Graphene's excellent electrical conductivity is due to its delocalized π-electron system, allowing for efficient charge transfer. This feature is critical for increasing the rate capability and power density of hybrid supercapacitors.148 Graphene oxide, reduced graphene oxide, N-doped graphene, and halogenated graphene are examples of chemically functionalized graphene materials. RGO has a high electrical conductivity (6300 S cm−1), while GO is an insulator or semiconductor. The RGO structure almost exactly mimics pure graphene's extraordinary surface area (2600 m2 g−1).149,150 Functionalizing graphene oxide with additional materials can enhance its performance in hybrid supercapacitors. For instance, Hou et al. developed amine-functionalized graphene (F-rGO)/polyaniline composites for supercapacitor applications. F-rGO/PANI has a much higher specific capacitance value (597 F g−1 at 0.5 A g−1) compared to F-rGO (210 F g−1) and pure PANI (400 F g−1). That's not the cycling stability of porous F-rGO/PANI (89%) after 2000 cycles at 1 A g−1 was much higher than that of F-rGO/PANI (75%) and pure PANI (65%). The energy density of PF-rGO/PANI is 34.9 W h kg−1 at a power density of 1.27 kW kg−1. The results show that regulating the morphology allows for a large amount of energy storage, and the mesoporous structure helps to stabilize the composite during electrochemical cycling. As a result, the combination of electrode materials with adjustable morphologies and mesoporous structures might provide an alternate approach to energy storage applications.151 Samira et al. produced a Ni(II)@amine-functionalized graphene oxide composite for a high-performance supercapacitor. The experimental study demonstrated a high capacitance of 1136 F g−1 at a current density of 2 A g−1, with notable capacitance retention of 84% at 5 A g−1 and commendable stability over 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles in a three-electrode configuration. Furthermore, an asymmetric two-electrode device (Ni@A-GO//AC) exhibited an impressive specific capacitance of 478 F g−1 at 2 A g−1, alongside commendable stability (87%) over 5000 cycles, indicating its potential for practical application as a high-performance supercapacitor.152 Haghshenas et al. established a straightforward and environmentally benign technique for synthesizing Arg-functionalized graphene sheets (Arg/GO), which can serve as electrodes in supercapacitors. The presence of amine and carboxylic acid groups in the Arg structure alters the surface chemistry of graphene. This enhances the wettability of the electrode, facilitating the diffusion of electrolyte ions into the electrode surface and inducing pseudo-faradaic reactions. The Arg/GO symmetric supercapacitor device has a specific capacitance of 295 F g−1 at a current density of 1 A g−1 and demonstrates significant cycling stability, retaining 94% capacitance after 4000 cycles. Taking into account environmental friendliness with the exceptional power and energy density of 2204 W kg−1 and 50 W h kg−1, respectively.153 Aliakbari et al. investigated a 5-amino-substituted 1,10-phenanthroline-functionalized graphene oxide (N-FGO) complexed with cobalt(II) and manufactured a nanocomposite (N-FGO-Co nanocomposite) used in a practical symmetrical supercapacitor. The alteration and synthesis of this complex markedly enhanced the morphological characteristics and electrochemical performance of the active material, which functioned as the electrodes for the supercapacitor. The composite electrode exhibited a specific capacitance of 512 F g−1 at a current density of 1 A g−1 and maintained 95.2% of its charge retention after 5000 charge–discharge cycles. The N-FGO-Co//N-FGO-Co system exhibits a specific capacity of 268 F g−1 at 0.5 A g−1, with an energy density of 9.2 W h kg−1 and a power density of 8000 W kg−1. After 5000 cycles at a current density of 16 A g−1, the symmetrical supercapacitor maintains 93.9% of its original capacity.154 Recent advancements in the preparation and modification of graphene and graphene oxide have led to significant improvements in their electrochemical performance. For example, Yolanda et al. employed a hydrothermal method to synthesize reduced graphene oxide (rGO) decorated with iron oxide nanoparticles, which enhanced the capacitance and stability of the supercapacitor. This composite material exhibited a specific capacitance of 345 F g−1 and maintained over 90% of its capacitance after 10
000 cycles in a three-electrode configuration. Furthermore, an asymmetric two-electrode device (Ni@A-GO//AC) exhibited an impressive specific capacitance of 478 F g−1 at 2 A g−1, alongside commendable stability (87%) over 5000 cycles, indicating its potential for practical application as a high-performance supercapacitor.152 Haghshenas et al. established a straightforward and environmentally benign technique for synthesizing Arg-functionalized graphene sheets (Arg/GO), which can serve as electrodes in supercapacitors. The presence of amine and carboxylic acid groups in the Arg structure alters the surface chemistry of graphene. This enhances the wettability of the electrode, facilitating the diffusion of electrolyte ions into the electrode surface and inducing pseudo-faradaic reactions. The Arg/GO symmetric supercapacitor device has a specific capacitance of 295 F g−1 at a current density of 1 A g−1 and demonstrates significant cycling stability, retaining 94% capacitance after 4000 cycles. Taking into account environmental friendliness with the exceptional power and energy density of 2204 W kg−1 and 50 W h kg−1, respectively.153 Aliakbari et al. investigated a 5-amino-substituted 1,10-phenanthroline-functionalized graphene oxide (N-FGO) complexed with cobalt(II) and manufactured a nanocomposite (N-FGO-Co nanocomposite) used in a practical symmetrical supercapacitor. The alteration and synthesis of this complex markedly enhanced the morphological characteristics and electrochemical performance of the active material, which functioned as the electrodes for the supercapacitor. The composite electrode exhibited a specific capacitance of 512 F g−1 at a current density of 1 A g−1 and maintained 95.2% of its charge retention after 5000 charge–discharge cycles. The N-FGO-Co//N-FGO-Co system exhibits a specific capacity of 268 F g−1 at 0.5 A g−1, with an energy density of 9.2 W h kg−1 and a power density of 8000 W kg−1. After 5000 cycles at a current density of 16 A g−1, the symmetrical supercapacitor maintains 93.9% of its original capacity.154 Recent advancements in the preparation and modification of graphene and graphene oxide have led to significant improvements in their electrochemical performance. For example, Yolanda et al. employed a hydrothermal method to synthesize reduced graphene oxide (rGO) decorated with iron oxide nanoparticles, which enhanced the capacitance and stability of the supercapacitor. This composite material exhibited a specific capacitance of 345 F g−1 and maintained over 90% of its capacitance after 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles.155 The stability and durability of graphene and graphene oxide in hybrid supercapacitors are crucial for their practical application. Graphene-based electrodes generally exhibit high chemical and mechanical stability, which is essential for long-term performance. Zhao et al. highlighted that rGO electrodes maintained over 85% of their initial capacitance after 20
000 cycles.155 The stability and durability of graphene and graphene oxide in hybrid supercapacitors are crucial for their practical application. Graphene-based electrodes generally exhibit high chemical and mechanical stability, which is essential for long-term performance. Zhao et al. highlighted that rGO electrodes maintained over 85% of their initial capacitance after 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles, demonstrating their robustness.156
000 cycles, demonstrating their robustness.156
Recent innovations in the field have focused on enhancing the performance of graphene and GO through advanced processing techniques. For example, Kumar et al. used a straightforward hydrothermal technique to nucleate the hexagonal NiCO2S4 hierarchical nanostructure on reduced graphene oxide (rGO) sheets for utilization as an electrode material in supercapacitors. The composite Rgo-NiCo2S4 materials exhibited several advantages, such as enhanced electrolyte diffusion channels, accelerated electron transport, and abundant active sites. This was achievable because to the synergistic interplay between the hierarchical structure of NiCo2S4 and the high conductivity of rGO. At a current density of 1 A g−1, the hybrid composites exhibited a superior specific capacitance of 972 F g−1. The rGO-NiCo2S4//AC ASC device exhibited a specific capacitance of 208 F g−1 at 1 A g−1, and maintained 94.1% of its capacitance after 2000 cycles. The rGO-NiCo2S4//AC ASC device exhibited a superior energy density value of 56.62 W h kg−1, which is double that of the NiCo2S4//AC ASC device at 23.26 W h kg−1. Furthermore, the magnitude of power density remained almost constant at 701.37 W kg−1 for both devices.157 Nirosha et al. synthesized a rGO/MWCNT/Co3O4 composite by a simple hydrolysis technique followed by heating. Due to their excellent electrical conductivity and exceptional carrier mobility, rGO and MWCNTs serve as optimal matrices for hybrid supercapacitors, while their composites with metal oxides demonstrate improved electrochemical properties owing to the synergistic effects and integration of various dimensionalities. The rGO/MWCNT/Co3O4 composite exhibited an exceptional specific capacitance of 922 F g−1 at a current density of 1 A g−1, with an insignificant capacitance decline after 2000 cycles. The rGO/MWCNT/Co3O4 composite exhibited a power density of 2000 W kg−1 and an energy density of 32.2 W h kg−1 when used in a symmetric supercapacitor. The supercapacitor constructed with the composite exhibited 83% capacitance retention over 2000 cycles at 3 A g−1.158 Wu and others discovered that physically compressing Fe2O3NDs/rGO aerogel allowed them to be employed as flexible, stand-alone electrodes. These electrodes demonstrated excellent specific capacitance (347.4 F g−1 at 1 A g−1), high rate capability (184 F g−1 at 10 A g−1), and good cycle stability. The Fe2O3 NDs/rGO//Co3O4 NDs/rGO asymmetric supercapacitor has a high specific capacitance of 69.1 F g−1 at 1 A g−1 and an exceptional energy density of 21.6 W h kg−1 at 750 W kg−1, as well as good cycling stability with a capacity retention of 94.3% after 5000 cycles. This study offers a potential approach to designing high-performance graphene-based composite electrodes and significant inspiration for the development of innovative flexible energy-storage devices.159 However, Ramachandran et al. reported that graphene aerogels and their composites have attracted considerable attention due to their large surface area, tunable porosity, and excellent electrical conductivity, making them promising candidates for multifunctional applications. They also noted that advanced synthesis techniques such as self-assembly, additive manufacturing, and heteroatom doping have significantly expanded the applicability of these materials in areas including energy storage and environmental remediation. Despite these developments, challenges related to large-scale synthesis, structural uniformity, and process optimization must be addressed to facilitate their transition from laboratory research to industrial implementation.160 Manibalan et al. developed a new heterostructure nanocomposite, C/Ru@NiO-CuO (CRNC), appropriate for energy storage applications. The CRNC nanocomposite exhibits a nanospherical morphology and a highly crystalline heterostructure. It is a dependable electrode material that offers several advantages for supercapacitor applications, including excellent conductivity, substantial surface area, and multiple oxidation states. In a three-electrode cell design, the synthesized CRNC demonstrates superior electrochemical performance at 0.5 A g−1, achieving a specific capacitance of 318 F g−1. Following 5000 GCD cycles, the electrode exhibits a commendable capacity retention rate of 86.7%. An asymmetric solid-state supercapacitor was fabricated by positioning the separator between the CRNC and rGO/α-Co3O4, achieving a specific capacitance of 152.5 F g−1 at 0.5 A g−1, and maintaining 87% capacitance retention after 3500 GCD cycles. Additinally, the ASC provides energy and power density metrics of 21.18 W h kg−1 and 1800 W kg−1, respectively. The CRNC//rGO/α-Co3O4 configuration demonstrates excellent structural stability, prolonged cycle stability, superior rate capacity, and advantageous compatibility.161 Liu et al. (2021) developed a scalable method for producing graphene oxide with tunable oxygen content, which allowed for better control over the material's electrochemical properties.162 Moreover, the integration of graphene with conductive polymers or other nanomaterials has been explored to further enhance the performance of hybrid supercapacitors.163 Graphene and graphene oxide are critical materials in the development of high-performance hybrid supercapacitors. Their exceptional electrical conductivity, high surface area, and ability to form composites make them valuable components in achieving superior energy and power densities. Ongoing research and technological advancements continue to optimize their properties and expand their applications in advanced energy storage systems.
Carbon nanotubes exhibit sp2 bonding, characterized by a honeycomb lattice structure, each atom is bonded to three adjacent atoms, similar to graphite. Carbon nanotubes are categorized into single-walled carbon nanotubes (SWCNTs), double-walled carbon nanotubes (DWCNTs), and multi-walled carbon nanotubes (MWCNTs), shown in Fig. 9c1. SWCNTs consist of a solitary graphene cylinder, whereas MWCNTs include numerous concentric graphene cylinders. DWCNTs are a kind of multi-walled carbon nanotubes composed of two tubes. DWCNTs integrate the characteristics of SWCNTs and MWCNTs, with a diameter that lies between the two primary types of CNTs. As a result, they integrate the performance of both varieties of carbon nanotubes.166,167 The electrical conductivity of CNTs, stemming from their unique π-electron delocalization, significantly enhances charge storage and transfer processes in supercapacitors.168 The specific surface area of the material ranges from a few tens (single CNT) up to 500–1700 m2 g−1, allowing substantial charge buildup.169–171 CNTs exhibit superior electrochemical performance compared to traditional carbon materials. Their high electrical conductivity ensures efficient charge transport, which is crucial for high power density in supercapacitors. Song et al. developed an innovative and sustainable carbon-based material termed hollow porous carbon particles, which encapsulate multi-wall carbon nanotubes (CNTs@HPC) for use in supercapacitors. The composite has macro pores, meso pores, and micro pores, resulting in a distinctive hierarchical porous structure with a total specific surface area of 913 m2 g−1. The presence of residual silicon–aluminum elements in CNT@HPC, a direct result of the unique synthesis approach involving nickel phyllosilicate in CDM, accounts for the heightened hydrophilicity. This manufacturing method results in excellent conductivity, facilitating rapid movement of electrolyte ions and enhancing capacitive performance. The optimal CNT@HPC has a specific capacitance of 306 F g−1 at 1 A g−1.172 Mandal et al. synthesized multi-walled carbon nanotubes by a straightforward and economical pyrolysis technique for supercapacitor applications. The synthesized MWCNTs electrode exhibits a peak specific capacitance of 278 F g−1 at 1 A g−1 and 257 F g−1 at 5 mV s−1. The engineered MWCNTs electrode has shown remarkable retention, preserving 97% capacitance after 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles.173 These results may lead to the creation of energy storage systems that are more efficient and resilient, appropriate for diverse applications. The high conductivity of CNTs results in low internal resistance and improved charge/discharge rates.
000 cycles.173 These results may lead to the creation of energy storage systems that are more efficient and resilient, appropriate for diverse applications. The high conductivity of CNTs results in low internal resistance and improved charge/discharge rates.
To further enhance the performance of CNTs, they are often combined with other materials, such as metal oxides or conductive polymers. For example, blending CNTs with transition metal oxides like MnO2, Co3O4 or Fe2O3 can significantly boost the pseudocapacitive behavior and overall capacitance of hybrid supercapacitors. Sun et al. documented a flexible asymmetric supercapacitor exhibiting elevated energy and power densities, constructed from nanostructured MnO2 nanospheres and Bi2O3 nanoshells affixed to a three-dimensional interconnected carbon nanotube sponge in a neutral hydrogel electrolyte. An ASC device exhibiting flexibility, with negative and positive electrodes composed of CNTs@Bi2O3 and CNTs@MnO2, operates at 1.8 V, with a substantial capacitance of 2.4 F cm−3 (82 F g−1 at 1 A g−1) and an impressive energy density of 75.4 μW h cm−2 (37 W h kg−1 at a power density of 1554 W kg−1). Capacitor retention is maintained at around 98% after 3000 fast GCD cycles. Furthermore, the adaptable ASC device in this study employs a nontoxic hydrogel electrolyte, making it environmentally sustainable and possibly advantageous for wearable electronic applications.174 Mandal et al. have developed a novel hybrid composite of activated carbon/MWCNTs-ZnFe2O4 using a straightforward and eco-friendly approach. At a current density of 1 A g−1, the composite exhibits the highest specific capacitance of 609 F g−1. The long-term stability test indicated that 91% capacitance was sustained after 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles at 1 A g−1, confirming the exceptional cycling performance of the produced composite. The incorporation of MWCNTs and ZFO into the host AC offers superior storage performance and cycle stability.175 According to Luo et al., they created high-performance flexible supercapacitors out of fabric covered with polypyrrole (PPy) and composed of carbon nanotubes and cotton. Electrodes are made using a simple and scalable knitting and chemical polymerization method. This fabric contains 13.2 mg of PPy and has a high areal capacitance (4192 mF cm−2), strong rate capability (50% capacitance retention at 100 mA cm−2), and excellent cycling stability (95% capacitance retention after 5000 cycles). The fabric electrodes from a quasi-solid-state symmetric supercapacitor with a capacitance of 1947 mF cm−2 at 5 mA cm−2, maximum energy density of 0.17 mW h cm−2, and maximum power density of 25.6 mW cm−2. These findings open up possibilities for producing high-performance, flexible supercapacitors in a scalable manner.176 Kshetri et al. constructed a three-part carbon aerogel structure by integrating graphene (Gr), carbon nanofibers (CNFs), and carbon nanotubes. The electrode material composed of CNTs@Gr-CNF exhibits superior electrochemical performance. The material has a specific capacitance of 521.5 F g−1 at 0.25 A g−1 and retains 98% of its capacitance after 10
000 cycles at 1 A g−1, confirming the exceptional cycling performance of the produced composite. The incorporation of MWCNTs and ZFO into the host AC offers superior storage performance and cycle stability.175 According to Luo et al., they created high-performance flexible supercapacitors out of fabric covered with polypyrrole (PPy) and composed of carbon nanotubes and cotton. Electrodes are made using a simple and scalable knitting and chemical polymerization method. This fabric contains 13.2 mg of PPy and has a high areal capacitance (4192 mF cm−2), strong rate capability (50% capacitance retention at 100 mA cm−2), and excellent cycling stability (95% capacitance retention after 5000 cycles). The fabric electrodes from a quasi-solid-state symmetric supercapacitor with a capacitance of 1947 mF cm−2 at 5 mA cm−2, maximum energy density of 0.17 mW h cm−2, and maximum power density of 25.6 mW cm−2. These findings open up possibilities for producing high-performance, flexible supercapacitors in a scalable manner.176 Kshetri et al. constructed a three-part carbon aerogel structure by integrating graphene (Gr), carbon nanofibers (CNFs), and carbon nanotubes. The electrode material composed of CNTs@Gr-CNF exhibits superior electrochemical performance. The material has a specific capacitance of 521.5 F g−1 at 0.25 A g−1 and retains 98% of its capacitance after 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 GCD cycles at 5 A g−1 in 6 M KOH. The CNTs@Gr-CNF//NiCo2S4 hybrid supercapacitor has a specific capacitance of 218 F g−1 at 1 A g−1 and an energy density of 62.13 W h kg−1 at a power density of 789.66 W kg−1. Furthermore, the device has exceptional cyclic stability, retaining 91.7% of its specific capacitance after 10
000 GCD cycles at 5 A g−1 in 6 M KOH. The CNTs@Gr-CNF//NiCo2S4 hybrid supercapacitor has a specific capacitance of 218 F g−1 at 1 A g−1 and an energy density of 62.13 W h kg−1 at a power density of 789.66 W kg−1. Furthermore, the device has exceptional cyclic stability, retaining 91.7% of its specific capacitance after 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles. The findings indicate that the CNTs@Gr-CNF material has exceptional properties, making it suitable for use as a negative electrode in advanced hybrid supercapacitors.177 Sham lal et al. constructed a symmetric hybrid supercapacitor using SWCNTs/TiO2 nanocomposite electrodes and PVA/H2SO4 gel electrolyte. Symmetric HSCs have a high capacitance of 144 F g−1, a specific energy of 20 W h kg−1, and an impressive cycling life of 95% up to 50
000 cycles. The findings indicate that the CNTs@Gr-CNF material has exceptional properties, making it suitable for use as a negative electrode in advanced hybrid supercapacitors.177 Sham lal et al. constructed a symmetric hybrid supercapacitor using SWCNTs/TiO2 nanocomposite electrodes and PVA/H2SO4 gel electrolyte. Symmetric HSCs have a high capacitance of 144 F g−1, a specific energy of 20 W h kg−1, and an impressive cycling life of 95% up to 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles.178 Dang et al. use one-step phosphorization of nickel cobalt hydroxide to create homologous 3D NiCoP/CoP heteronanosheet networks supported on N-doped carbon nanotubes. At a Ni/Co ratio of 1
000 cycles.178 Dang et al. use one-step phosphorization of nickel cobalt hydroxide to create homologous 3D NiCoP/CoP heteronanosheet networks supported on N-doped carbon nanotubes. At a Ni/Co ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2, N-CNTs@NiCoP/CoP exhibit a high specific capacity of 152 mA h g−1 at 1 A g−1, as well as superior rate capability with a capacity retention of 61% even at 30 A g−1. The hybrid supercapacitor, composed of an N-CNTs@NiCoP/CoP cathode and a porous carbon anode generated from ZIF-67, has a high energy density of 45.5 W h kg−1 at a power density of 784 W kg−1 and is exceptionally stable, retaining 87% of its energy after 10
2, N-CNTs@NiCoP/CoP exhibit a high specific capacity of 152 mA h g−1 at 1 A g−1, as well as superior rate capability with a capacity retention of 61% even at 30 A g−1. The hybrid supercapacitor, composed of an N-CNTs@NiCoP/CoP cathode and a porous carbon anode generated from ZIF-67, has a high energy density of 45.5 W h kg−1 at a power density of 784 W kg−1 and is exceptionally stable, retaining 87% of its energy after 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles at 12 A g−1. It has excellent electrochemical performance because to the significant synergistic impact of NiCoP and CoP in their heterojunctions, greater surface area, and active sites created by the 3D nanosheet network and conductive N-CNT support.179 Ahmad et al. reported that the hybrid electrode is composed of hexyl-aminated carbon nanotubes (HA-CNTs) with high electrical conductivity and surface functionalities, as well as a porous Mn-Ni(3-hydroxypyridine-2-carboxylic acid) metal–organic framework (Mn-Ni(3-HPCA) MOF) for fast ion diffusion. The Mn-Ni(3-HPCA)MOF/HA-CNT hybrid had an impressive specific capacitance of 673.3 F g−1 at 0.25 A g−1 and maintained 72.3% of its capacitance at 10 A g−1. Hybrid Mn-Ni(3-HPCA) MOF/HA-CNT//XGnP supercapacitor that works with a KOH-loaded ploy(acrylonitrile-co-1-vinylimidazole) gel is strong and long-lasting. It demonstrated an impressive energy density of 67.9 W h kg−1 at a power density of 300 W kg−1 and possessed high cycling stability (87.1% after 10
000 cycles at 12 A g−1. It has excellent electrochemical performance because to the significant synergistic impact of NiCoP and CoP in their heterojunctions, greater surface area, and active sites created by the 3D nanosheet network and conductive N-CNT support.179 Ahmad et al. reported that the hybrid electrode is composed of hexyl-aminated carbon nanotubes (HA-CNTs) with high electrical conductivity and surface functionalities, as well as a porous Mn-Ni(3-hydroxypyridine-2-carboxylic acid) metal–organic framework (Mn-Ni(3-HPCA) MOF) for fast ion diffusion. The Mn-Ni(3-HPCA)MOF/HA-CNT hybrid had an impressive specific capacitance of 673.3 F g−1 at 0.25 A g−1 and maintained 72.3% of its capacitance at 10 A g−1. Hybrid Mn-Ni(3-HPCA) MOF/HA-CNT//XGnP supercapacitor that works with a KOH-loaded ploy(acrylonitrile-co-1-vinylimidazole) gel is strong and long-lasting. It demonstrated an impressive energy density of 67.9 W h kg−1 at a power density of 300 W kg−1 and possessed high cycling stability (87.1% after 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles).180 Ma et al. created a three-dimensional (3D) linked network structure by merging it with conductive carbon nanotubes that penetrate MOF-derived Co-Ni-S composite spheres. 3D linked design considerably improves electronic structure, charge-transfer capacity, and pseudocapacitive properties. The hybrid electrode composed of Co-Ni-S and CNTs exhibits a high specific capacitance of 540.6 C g−1 at 1 A g−1 and a rate capability of 69.9% at 10 A g−1. The Co-Ni-S/CNTs//AC hybrid supercapacitor achieves a high energy density of 63.5 W h kg−1 at 800 W kg−1 and retains 83.0% of its original capacitance after 10
000 cycles).180 Ma et al. created a three-dimensional (3D) linked network structure by merging it with conductive carbon nanotubes that penetrate MOF-derived Co-Ni-S composite spheres. 3D linked design considerably improves electronic structure, charge-transfer capacity, and pseudocapacitive properties. The hybrid electrode composed of Co-Ni-S and CNTs exhibits a high specific capacitance of 540.6 C g−1 at 1 A g−1 and a rate capability of 69.9% at 10 A g−1. The Co-Ni-S/CNTs//AC hybrid supercapacitor achieves a high energy density of 63.5 W h kg−1 at 800 W kg−1 and retains 83.0% of its original capacitance after 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles. The good performance underlines the need of the 3D integrated design for future energy storage.181
000 cycles. The good performance underlines the need of the 3D integrated design for future energy storage.181
Functionalizing CNTs with various chemical groups or incorporating them into hybrid structures can improve their compatibility with electrolytes and enhance their electrochemical performance. Research by Faisal et al. explored the functionalization of CNTs with nitrogen-doped graphene, which improved the material's electrochemical stability and specific capacitance by facilitating better interaction with electrolytes.182 Additionally, encapsulating CNTs in carbon-based matrices or polymer matrices can enhance their mechanical stability and electrochemical performance. The long-term stability and mechanical strength of CNTs are critical for the practical application of supercapacitors. CNTs exhibit excellent cycling stability, with minimal capacity loss over extended charge/discharge cycles. According to the work by Razzaq et al. CNT-based electrodes maintained over 90% of their initial capacitance after 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles, indicating their high durability.183 Despite their advantages, there are challenges associated with the use of CNTs in supercapacitors, such as difficulties in large-scale synthesis and dispersion issues. Future research is focused on addressing these challenges through improved synthesis techniques, better dispersion methods, and exploring new applications for CNT-based materials.184 Continued development in this area will likely lead to even greater enhancements in the performance of hybrid supercapacitors. Carbon nanotubes represent a powerful class of materials for the advancement of hybrid supercapacitors. Their exceptional electrical conductivity, high surface area, and stability make them highly effective as electrode materials. Ongoing research into functionalization, composite formation, and large-scale production will further enhance the capabilities and practical applications of CNTs in energy storage technologies.
000 cycles, indicating their high durability.183 Despite their advantages, there are challenges associated with the use of CNTs in supercapacitors, such as difficulties in large-scale synthesis and dispersion issues. Future research is focused on addressing these challenges through improved synthesis techniques, better dispersion methods, and exploring new applications for CNT-based materials.184 Continued development in this area will likely lead to even greater enhancements in the performance of hybrid supercapacitors. Carbon nanotubes represent a powerful class of materials for the advancement of hybrid supercapacitors. Their exceptional electrical conductivity, high surface area, and stability make them highly effective as electrode materials. Ongoing research into functionalization, composite formation, and large-scale production will further enhance the capabilities and practical applications of CNTs in energy storage technologies.
3.2. Metal oxides and hydroxides
MnO2 (Fig. 10a) is a widely studied transition metal oxide known for its high theoretical specific capacitance (1370 F g−1). It is favored for its abundant availability, low cost, and environmental friendliness.194 Recent advances have focused on improving MnO2's conductivity and stability using various synthesis methods for nanostructured forms such as nanowires, nanorods, nanosheets, nanotubes, nanospheres, nanoflowers, and nanoparticles.195 According to Gaini Zhang et al., manganese oxide nanosheets with many in-plane nanopores are created by an in situ redox reaction and an etching procedure. This new method offers a wide ion-accessible surface area for effective charge storage, yielding porous MnO2 nanosheets with a higher specific capacitance (274 F g−1 at 0.25 A g−1) and desired rate capability (98.4% of initial capacitance after 5000 cycles at 5 A g−1).196 Xiaoli Dai et al. produced MnO2 electrodes using ultrasonic-assisted electrodeposition. The electrodes manufactured at a current density of 7 mA cm−2 exhibit a high specific capacitance of 415.4 F g−1 at 1 A g−1 in the supercapacitor and 818.1 mA h g−1 at 1 A g−1 after 200 cycles in lithium-ion batteries.197 For instance, a study by Su et al. demonstrated that MnO2 nanowires achieved a specific capacitance of 472 F g−1 with excellent cycle stability.198
 | ||
| Fig. 10 A schematic structure of (a) MnO2, (b)Co3O4, (c) NiO (using Vesta software) (d) layered double hydroxides (LDH), Reproduced from ref. 185 with permission from MDPI, copyright 2019, (e) polyaniline (PANI), and (f) polypyrrole (PPy). | ||
Co3O4 (Fig. 10b) is another significant TMO known for its high specific capacitance (3560 F g−1) and electrical conductivity. It provides multiple oxidation states, facilitating efficient charge storage and transfer.199 Chrisma Rose Babu et al., synthesized Co3O4 nanoparticles using the in situ hydrothermal method with varying pH levels in the precursor solution. At a pH of 10, the prepared cobalt nanoparticles exhibit 1195.05 F g−1 of specific capacitance at a current density of 1.5 A g−1 in a three-electrode system. While the symmetric supercapacitor device delivered 870.6 F g−1 of specific capacitance at 5 A g−1 with an energy density of 77.3 W h kg−1 and power density of 1997.7 W kg−1, along with 88% of cycling stability after 970 GCD cycles.200 Recent research has shown that Co3O4-based supercapacitors can deliver specific capacitances up to 800 F g−1 4. Incorporating of Co3O4 into hybrid systems with carbon-based materials or conductive polymers further enhances performance by improving conductivity and charge distribution. NiO (Fig. 10c) has great attention due to its extra-high specific capacitance (3750 F g−1), excellent thermal and electrochemical stability. The pseudocapacitive behavior of NiO is attributed to its ability to participate in redox reactions involving Ni2+/Ni3+ ions.201 Mamta et al., derived NiO nanoparticles using aloe vera as a reducing agent through a simple green synthesis process and found that the synthesized nanoparticles display a specific capacitance of 336 F g−1 at a current density of 0.76 A g−1. However, NiO//NiO symmetric device delivered a specific capacitance of 239 F g−1, a specific energy density of 47.8 W h kg−1, and a power density of 545 W kg within a 1.2 V voltage window (−0.6 to 0.6 V).202 Research by Li et al. (2021) highlighted that NiO nanostructures exhibited a specific capacitance of 435.25 F g−1 when combined with reduced graphene oxide (rGO).203 NiO's integration with other materials often leads to synergistic effects, enhancing overall supercapacitor performance.
Unfortunately, the poor electrical conductivity of MnO2, Co3O4, and NiO limits the full utilization of their high specific capacitance. Hence, researchers employ different strategies (dimension reduction, composite formation, doping, morphology control, coating, electrolyte modification, etc.) to enhance the electrical conductivity and improve the specific surface area of the electrode.204 The combination of transition metal oxides with carbon-based materials or conductive polymers forms composites that leverage the advantages of both materials. Such hybrid systems can enhance conductivity, improve mechanical stability, and provide higher capacitance. For example, combining MnO2 with CNTs or graphene has significantly improved supercapacitor performance. A study by Singu et al. reported that MnO2/graphene composites achieved a specific capacitance of 674 F g−1 with excellent rate capability and cycling stability.205 Despite their advantages, transition metal oxides face challenges like limited electronic conductivity and potential material dissolution in electrolytes. Ongoing research aims to address these issues through the development of novel synthesis methods, optimization of nanostructures, and exploration of hybrid materials to enhance performance and stability.206 Advances in these areas will contribute to the practical application of TMOs in hybrid supercapacitors. Transition metal oxides are vital for advancing hybrid supercapacitors due to their high pseudocapacitance, rich electrochemical activity, and versatility. Continued research into optimizing these materials and developing effective composite systems will further enhance the performance and applicability of hybrid supercapacitors in various energy storage applications.
LDHs (Fig. 10d) consist of positively charged metal hydroxide layers intercalated with exchangeable anions, which provide a high degree of flexibility in tailoring the material's properties. Typically, LDHs are synthesized through co-precipitation, hydrothermal, or sol–gel methods. The general formula for LDHs is [M1−X2+MX3+(OH)2AX/nn−·mH2O], where M2+ and M3+ are divalent (e.g. Ni, Co, Cu, Mg, etc.) and trivalent (e.g. Al, Fe, Mn, Cr, etc.) metal cations, respectively, and An− represents the interlayer anions (e.g., Cl−, OH−, NO3−, SO42−, and organic anions), x can be any value between 0.2 and 0.33, and ‘m’ is the number of crystal water molecules.208,209 Recent advances have focused on optimizing LDH synthesis to enhance their electrochemical performance. For instance, Long et al. effectively synthesized a cobalt aluminum layered double hydroxide and its derivatives by hydrothermal, alkali-etching, and phosphorization processes. The CoAl-LDHs electrode had a high capacitance of 983.1 F g−1 at 1 A g−1, a better rate capability of 88.64% at 10 A g−1, and outstanding cycling stability (92.68% retention after 5000 cycles at 10 A g−1). Furthermore, the CoAl LDH//graphene asymmetric capacitor has an energy density of 16.29 W h kg−1 at a power density of 700 W kg−1, outstanding coulombic efficiency (90.85% at 5 A g−1), and impressive capacitance retention (82.84% after 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles).210 Rajendran et al. used in situ etching/ion exchange to effectively manufacture NiCo-LDHs from a self-templated Ni-MOF. The NiCo-LDH/10 sample has superior electrochemical properties, with a maximum specific capacity of 1272 C g−1 at 2 A g−1. Furthermore, the NiCo-LDH/10//CNT asymmetric supercapacitor delivered a maximum energy density of 36.1 W h kg−1, while retaining remarkable stability (103.9%) and coulombic efficiency (100.4%) even after 5000 cycles.211
000 cycles).210 Rajendran et al. used in situ etching/ion exchange to effectively manufacture NiCo-LDHs from a self-templated Ni-MOF. The NiCo-LDH/10 sample has superior electrochemical properties, with a maximum specific capacity of 1272 C g−1 at 2 A g−1. Furthermore, the NiCo-LDH/10//CNT asymmetric supercapacitor delivered a maximum energy density of 36.1 W h kg−1, while retaining remarkable stability (103.9%) and coulombic efficiency (100.4%) even after 5000 cycles.211
LDHs exhibit significant pseudocapacitive behavior due to the faradaic redox reactions occurring within their layered structures. This characteristic is crucial for hybrid supercapacitors, where LDHs contribute to both the capacitive and energy storage mechanisms. However, interlayer hydrogen bonding makes LDH aggregate more readily. This significantly limits electrochemical sites and decreases specific surface area. To solve this problem, conductive supports such as nickel foam, carbon nanotubes, carbon spheres, and graphene have been assembled. For example, Zhang et al. presented reduced graphene–oxide supported nickel–cobalt LDH nanosheets as a supercapacitor electrode material. The addition of graphene could significantly increase conductivity, while the supported NiCo-LDH could successfully prevent graphene from self-aggregation. This electrode achieved a capacitance of 1675 F g−1 at 1 A g−1 and 83.8% capacitance retention at 10 A g−1. Furthermore, the NiCo-LDH/rGO//AC asymmetric supercapacitor demonstrated a high energy density of 49.9 W h kg−1 at a power density of 375.2 W kg−1 and 62.2% of capacitance retention after 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles at 5 A g−1.212 Similarly, Luo et al. created nickel–cobalt–oxyselenide as a nanowire and nickel–cobalt–LDH nanosheets as shells on carbon fiber (CF@NiCoO4Se3/NixCoy-LDH) for supercapacitors. This novel nanocluster has a multitude of active sites and high electrical conductivity. The core–shell structure can exhibit an outstanding capacitance of 3270 F g−1 at 1 A g−1. In addition, CF@NiCoO4Se3/Ni2Co1-LDH//AC demonstrated a high energy density of 89.7 W h kg−1 at a power density of 800 W kg−1, along with 95.6% capacitance maintained after 10
000 cycles at 5 A g−1.212 Similarly, Luo et al. created nickel–cobalt–oxyselenide as a nanowire and nickel–cobalt–LDH nanosheets as shells on carbon fiber (CF@NiCoO4Se3/NixCoy-LDH) for supercapacitors. This novel nanocluster has a multitude of active sites and high electrical conductivity. The core–shell structure can exhibit an outstanding capacitance of 3270 F g−1 at 1 A g−1. In addition, CF@NiCoO4Se3/Ni2Co1-LDH//AC demonstrated a high energy density of 89.7 W h kg−1 at a power density of 800 W kg−1, along with 95.6% capacitance maintained after 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 GCD cycles.213 Thirukumaran et al. synthesized a composite material i.e., NiCo-LDH grown on carbon nanoballs. The composite electrode with 50% carbon nanoballs can deliver 1220 F g−1 of specific capacitance and retain 86% of its initial capacitance after 5000 cycles.214 These hybrid systems demonstrate the potential for designing high-performance supercapacitors with tailored properties for specific applications. Despite their advantages, the application of LDHs in supercapacitors faces challenges such as limited electrical conductivity and scalability of synthesis processes. Ongoing research is focused on addressing these issues by exploring novel synthesis techniques, optimizing material compositions, and developing scalable production methods. The integration of LDHs with other functional materials and advanced fabrication techniques is expected to enhance their performance and broaden their applicability in energy storage devices. Future studies should focus on improving the electrochemical performance of LDHs, developing cost-effective synthesis methods, and exploring new hybrid configurations to meet the growing demand for high-performance supercapacitors. Layered double hydroxides represent a promising class of materials for hybrid supercapacitors due to their high specific capacitance, structural stability, and flexibility in anion exchange. Continued research and development in optimizing LDH-based materials and their composites will contribute to advancements in energy storage technologies, offering improved performance and practical applications in various fields.
000 GCD cycles.213 Thirukumaran et al. synthesized a composite material i.e., NiCo-LDH grown on carbon nanoballs. The composite electrode with 50% carbon nanoballs can deliver 1220 F g−1 of specific capacitance and retain 86% of its initial capacitance after 5000 cycles.214 These hybrid systems demonstrate the potential for designing high-performance supercapacitors with tailored properties for specific applications. Despite their advantages, the application of LDHs in supercapacitors faces challenges such as limited electrical conductivity and scalability of synthesis processes. Ongoing research is focused on addressing these issues by exploring novel synthesis techniques, optimizing material compositions, and developing scalable production methods. The integration of LDHs with other functional materials and advanced fabrication techniques is expected to enhance their performance and broaden their applicability in energy storage devices. Future studies should focus on improving the electrochemical performance of LDHs, developing cost-effective synthesis methods, and exploring new hybrid configurations to meet the growing demand for high-performance supercapacitors. Layered double hydroxides represent a promising class of materials for hybrid supercapacitors due to their high specific capacitance, structural stability, and flexibility in anion exchange. Continued research and development in optimizing LDH-based materials and their composites will contribute to advancements in energy storage technologies, offering improved performance and practical applications in various fields.
3.3. Conducting polymers
PANI can be synthesized through various methods, including oxidative polymerization of aniline in the presence of an oxidizing agent. This process yields different forms of PANI, such as emeraldine salt, emeraldine base, and pernigraniline, each with distinct electrical and electrochemical properties.218,219 Emeraldine salt, in particular, exhibits high electrical conductivity and is widely used in energy storage applications. Recent advances have focused on optimizing the synthesis of PANI to enhance its performance in supercapacitors. For instance, Amarnath et al. created porous PANI by polymerization of aniline in a non-toxic reagent such as brine solution. The prepared porous PANI electrodes exhibited a high specific capacitance of 662 F g−1, which is 2.5 times greater than that of conventional PANI.220 Chu et al. synthesized polyaniline nanowires at different temperatures through a simple hydrothermal method. The prepared PANI nanowires at 80 °C delivered the highest specific capacitance of 540 F g−1 at 0.5 A g−1 and 82% of capacitance maintained after 1000 cycles at 5 A g−1 in 0.5 M H2SO4 electrolyte solution.221 Zhao et al. prepared PANI nanobelts by low-temperature-induced polymerization. Temperature, p-phenylenediamine and mixing degree of raw materials highly affect nanobelts' uniformity. The uniform-shaped PANI nanobelts obtained from the rotating packed bed have a specific capacitance of 304.4 F g−1 at 0.5 A g−1 and 40.65% capacitance retention after 1000 cycles.222 The electrochemical performance of PANI in hybrid supercapacitors is largely attributed to its redox-active nature. During the charging and discharging processes, PANI undergoes reversible redox reactions due to its different oxidation states, contributing to its high capacitance.223 This characteristic enables PANI to deliver high energy and power densities in supercapacitor devices.
Recent studies have demonstrated that in situ oxidative polymerization successfully coated nanostructured porous PANI on a flexible carbon cloth substrate. The uniform-coated PANI on carbon cloth electrodes reached a maximum capacitance of 691 F g−1 with 94% capacitance retention after 2000 charge–discharge cycles.224 To further enhance the performance of PANI in supercapacitors, researchers have explored the use of PANI composites with other materials such as carbon nanotubes, graphene, and metal oxides. These composites leverage the high surface area and conductivity of carbon materials while maintaining the high pseudocapacitance of PANI. For instance, Amirmohammad et al. fabricated PANI/GO/CuFe2O4 nanocomposite using in situ polymerization. The ternary composite electrode had the highest specific capacitance of 614.76 F g−1 at 1 A g−1 and 88% of cycle stability even after 3500 cycles. In addition, PANI/GO/CuFe2O4//GO asymmetric supercapacitor demonstrated energy density of 49.72 W h kg−1 and power density of 923 W kg−1.225 Zhao et al. prepared graphene oxide/polyaniline composite, where trifluoroacetic acid serves as the doping protonic acid, followed by filtration and the final reduction steps. The composite electrode delivered a specific capacitance of 810 F g−1 and good rate capability. The rGO/PANI based symmetric all-solid-state supercapacitor exhibited a highest energy density 62.2 W h kg−1 at a power density of 800 W kg−1, along with capacitance retention of 91.3% after 2000 cycles.226 Abeer et al. prepared CuO@Cu4O3/rGO/PANI nanocomposite by using in situ strategy based on co-precipitation and incorporation process of rGO, followed by in situ oxidative polymerization of aniline monomer. The composite electrode exhibited a high specific capacitance of 508 F g−1 at 1 A g−1.226 Hou et al., studied amine-functionalized graphene and mesoporous graphene were composited with PANI. F-rGO/PANI composite electrode delivered high specific capacitance of 597 F g−1 than PF-rGO/PANI (489 F g−1) at 1 A g−1. However, F-rGO/PANI and PF-rGO/PANI electrodes maintained 75% and 89% of initial capacitance after 2000 cycles at 1 A g−1.151 Despite its advantages, PANI-based materials face challenges such as limited mechanical stability and poor conductivity in certain oxidation states. Recent research is focused on addressing these issues by developing new doping strategies and incorporating PANI into more stable composite matrices. Future research aims to further optimize the synthesis and processing conditions of PANI to enhance its performance in hybrid supercapacitors. Additionally, exploring new doping methods and composite structures will be crucial for overcoming the existing limitations and achieving even higher performance levels. Polyaniline remains a key material in the development of hybrid supercapacitors due to its high specific capacitance, favorable electrochemical properties, and versatility. The ongoing advancements in PANI synthesis and composite technologies are expected to further enhance its performance and broaden its applications in energy storage devices.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.4, efficient electrolyte channels and quicker electron routes were formed. This resulted in a linked ultralong and uniform PPy-GS nanowire with the greatest surface area, smallest average pore size, lowest contact angle, and highest conductivity. The conductivity of PPy-GS-40% reached 13.54 S cm−1, compared to 1.17 S cm−1 for PPy particles and 3.84 S cm−1 for the PPy-CTAB nanowire. Furthermore, the maximum specific capacitance of PPy-GS reached 556 F g−1 at 1 A g−1, compared to 233 F g−1 for PPy particles and 397 F g−1 for the PPy-CTAB nanowire. Even after 2000 cycles at 1.0 A g−1, the original capacitance remained at 85.4%. The PPy-GS-40% symmetrical supercapacitor has an energy density of 49.4 W h kg−1 and a powder density of 400 W kg−1.229
0.4, efficient electrolyte channels and quicker electron routes were formed. This resulted in a linked ultralong and uniform PPy-GS nanowire with the greatest surface area, smallest average pore size, lowest contact angle, and highest conductivity. The conductivity of PPy-GS-40% reached 13.54 S cm−1, compared to 1.17 S cm−1 for PPy particles and 3.84 S cm−1 for the PPy-CTAB nanowire. Furthermore, the maximum specific capacitance of PPy-GS reached 556 F g−1 at 1 A g−1, compared to 233 F g−1 for PPy particles and 397 F g−1 for the PPy-CTAB nanowire. Even after 2000 cycles at 1.0 A g−1, the original capacitance remained at 85.4%. The PPy-GS-40% symmetrical supercapacitor has an energy density of 49.4 W h kg−1 and a powder density of 400 W kg−1.229
Bhoyar et al. investigated PPy synthesis using laccase isolated from Lentinus tigrinus SSB_W2 in the presence of several mediators, including ABTS, HBT, SA, p-CA, and VA. The PPy-ABTS has higher decomposition temperatures, showing greater thermal stability up to 400 °C. PPy-ABTS has a higher specific capacitance (242 F g−1 at 10 mV s−1 from CV and 153 F g−1 at 1 A g−1 from GCD curves) and a capacitance retention of over 90% after 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles.230 Iurchenkova et al. investigated how the synthetic media used to create polypyrroles altered their atomic, electrical, and morphological structures. They also investigated the electrochemical behavior of polypyrroles in water-based electrolytes with varying ions and pH levels in a supercapacitor. The research shows that the synthetic medium affects the size of polymer particles rather than their form. It is vital to note that the kind of electrolyte, the nitrogen species and oxygen-containing functional groups in polypyrrole's structure, and the particle size all have an impact on its electrochemical capabilities. The ideal electrolyte for employing polypyrrole in supercapacitor is neutral. Polypyrrole produced in acetonitrile has a specific capacitance of around 385 F g−1 and remains 60% stable after over 10
000 cycles.230 Iurchenkova et al. investigated how the synthetic media used to create polypyrroles altered their atomic, electrical, and morphological structures. They also investigated the electrochemical behavior of polypyrroles in water-based electrolytes with varying ions and pH levels in a supercapacitor. The research shows that the synthetic medium affects the size of polymer particles rather than their form. It is vital to note that the kind of electrolyte, the nitrogen species and oxygen-containing functional groups in polypyrrole's structure, and the particle size all have an impact on its electrochemical capabilities. The ideal electrolyte for employing polypyrrole in supercapacitor is neutral. Polypyrrole produced in acetonitrile has a specific capacitance of around 385 F g−1 and remains 60% stable after over 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles in a 1 M NaCl solution.231
000 cycles in a 1 M NaCl solution.231
To further enhance the performance of PPy in hybrid supercapacitors, researchers have developed various composites by combining PPy with other materials, such as carbon-based materials, metal oxides, and conductive polymers. These composites leverage the high surface area and electrical conductivity of the additives while maintaining the high pseudocapacitance of PPy. For instance, Jyothibasu et al. prepared polypyrrole nanoparticles via a simple in situ chemical polymerization using the plant-derived material curcumin as a bioavailable template. A PPyNP//f-CNT freestanding composite electrode of high mass loading was prepared after blending the mixtures of the prepared PPyNP and functionalized CNTs. The prepared free-standing composite electrode displayed a high areal capacitance of 4585 mF cm−2 at a current density of 2 mA cm−2. A symmetric PPyNP/f-CNT//PPyNP/f-CNT all-solid-state supercapacitor delivered a maximum areal energy and power density of 129.24 μW h cm−2 and 12.5 mW cm−2, respectively. In addition, the device exhibited 79.03% of capacitance retention after 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles.232 Nady et al. fabricated PPy/NiO nanocomposite electrode using electrochemical deposition technique at 4 A cm−2 for supercapacitors. The synergistic effect of NiO and PPy along with uniform porous network structure improves the accessibility of electrolyte ions for faradaic reactions. Hence, the composite electrode exhibited a high specific capacitance of 679 F g−1 at 1 A g−1, and capacitance retention of 83.9% of its initial capacitance after 1000 cycles at 1 A g−1.233 Ding et al. prepared PPy-modified Ni3S2 nanosheet electrodes through a hydrothermal method and a subsequent electrodeposition approach. The novel electrode can shorten the ion diffusion path and enhance its conductivity, resulting the electrode exhibited a capacity of 1484 C g−1 at 1 A g−1 and high cycling stability. Moreover, Ni3S2/PPy//AC hybrid supercapacitor demonstrated a maximal energy density of 74.9 W h kg−1 at power density of 1356.4 W kg−1 along with 91.7% of capacity retention even after 10
000 cycles.232 Nady et al. fabricated PPy/NiO nanocomposite electrode using electrochemical deposition technique at 4 A cm−2 for supercapacitors. The synergistic effect of NiO and PPy along with uniform porous network structure improves the accessibility of electrolyte ions for faradaic reactions. Hence, the composite electrode exhibited a high specific capacitance of 679 F g−1 at 1 A g−1, and capacitance retention of 83.9% of its initial capacitance after 1000 cycles at 1 A g−1.233 Ding et al. prepared PPy-modified Ni3S2 nanosheet electrodes through a hydrothermal method and a subsequent electrodeposition approach. The novel electrode can shorten the ion diffusion path and enhance its conductivity, resulting the electrode exhibited a capacity of 1484 C g−1 at 1 A g−1 and high cycling stability. Moreover, Ni3S2/PPy//AC hybrid supercapacitor demonstrated a maximal energy density of 74.9 W h kg−1 at power density of 1356.4 W kg−1 along with 91.7% of capacity retention even after 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles at 4 A g−1.234 Porous structured rGO/PPy electrodes were prepared by Dang et al. using electrochemical co-deposition and reduction. The porous structure not only improves the electroactive sites of the electrodes but also actively promotes the diffusion efficiency of the electrolyte ions. As a result, the electrode delivered a maximum capacitance of 1091.2 mF cm−2 at 1 mA cm−2 and maintained 72.8% capacitance retention after 5000 cycles at 10 mA cm−2.235 Zhang et al. reported that a thin layer of graphene oxide and PPy might undergo a redox reaction via a moderate hydrothermal procedure. This produces a reduced graphene oxide/PPy hydrogel layer that adheres to the carbon cloth. The as-prepared electrode has a high capacitance of 1221 mF cm−2 at 1 mA cm−2 and good cycle stability. A symmetric supercapacitor with two ideal electrodes may reach an energy density of 70.4 μW h cm−2 with a power density of 0.7 mW cm−2 and a superior capacitance retention of 81.7% after 5000 cycles at 20 mA cm−2.236 Despite its advantages, PPy-based materials face several challenges, including issues related to mechanical stability, conductivity variations, and processing difficulties. To address these challenges, ongoing research is focused on optimizing the synthesis conditions, improving the stability of PPy, and developing new composite materials. Future research aims to explore novel doping strategies, improve the mechanical properties of PPy-based materials, and develop advanced composites to further enhance the performance of PPy in hybrid supercapacitors. Additionally, research is needed to explore scalable and cost-effective synthesis methods for practical applications. Polypyrrole remains a key material in the field of hybrid supercapacitors due to its high specific capacitance, favorable electrochemical properties, and versatility. The continued advancements in PPy synthesis and composite technologies are expected to further enhance its performance and expand its applications in energy storage devices.
000 cycles at 4 A g−1.234 Porous structured rGO/PPy electrodes were prepared by Dang et al. using electrochemical co-deposition and reduction. The porous structure not only improves the electroactive sites of the electrodes but also actively promotes the diffusion efficiency of the electrolyte ions. As a result, the electrode delivered a maximum capacitance of 1091.2 mF cm−2 at 1 mA cm−2 and maintained 72.8% capacitance retention after 5000 cycles at 10 mA cm−2.235 Zhang et al. reported that a thin layer of graphene oxide and PPy might undergo a redox reaction via a moderate hydrothermal procedure. This produces a reduced graphene oxide/PPy hydrogel layer that adheres to the carbon cloth. The as-prepared electrode has a high capacitance of 1221 mF cm−2 at 1 mA cm−2 and good cycle stability. A symmetric supercapacitor with two ideal electrodes may reach an energy density of 70.4 μW h cm−2 with a power density of 0.7 mW cm−2 and a superior capacitance retention of 81.7% after 5000 cycles at 20 mA cm−2.236 Despite its advantages, PPy-based materials face several challenges, including issues related to mechanical stability, conductivity variations, and processing difficulties. To address these challenges, ongoing research is focused on optimizing the synthesis conditions, improving the stability of PPy, and developing new composite materials. Future research aims to explore novel doping strategies, improve the mechanical properties of PPy-based materials, and develop advanced composites to further enhance the performance of PPy in hybrid supercapacitors. Additionally, research is needed to explore scalable and cost-effective synthesis methods for practical applications. Polypyrrole remains a key material in the field of hybrid supercapacitors due to its high specific capacitance, favorable electrochemical properties, and versatility. The continued advancements in PPy synthesis and composite technologies are expected to further enhance its performance and expand its applications in energy storage devices.
3.4. Emerging materials
MOFs exhibit high surface areas and tunable porosity, which are advantageous for supercapacitor electrodes. The large surface area facilitates increased charge storage, while the porosity allows for effective ion transport.238 Because of their rich skeleton types, large specific surface area, adjustable pore structure, and many functional groups, MOF materials have broad application prospects in energy storage, gas adsorption and separation, catalysis, chemical sensing, biomedicine, and other fields. Recent studies have shown that MOFs can provide high specific capacitance and excellent rate performance when integrated into supercapacitor electrodes. For instance, Wang et al. (2024) reported that a MOF-based electrode achieved a specific capacitance of 896.1 C g−1, demonstrating its high potential for energy storage applications.239
MOFs are a relatively new but rapidly developing family of porous materials that have attracted significant interest in chemistry and materials research due to their distinct features. MOFs are classified into various types based on their configuration: MIL series MOFs, PCN series MOFs, CPL series MOFs, UIO series MOFs, ZIF series MOFs, and IRMOF series MOFs.240 MIL (Materials Institute Lavoisier) series MOFs are composed of carboxylate ligands (e.g., 1,3,5-benzene tricarboxylate, terephthalate, and isophthalate) and trivalent metal ions (V, Cr, Fe, Al, Ga, ln, lanthanides), as well as metal ions in various oxidation states (e.g., Ti(IV)).241 PCN (porous coordination network) series MOFs feature cubic octahedral nanopore cages that create a pore-cage-pore channel-like architecture in space, with enormous potential for gas storage.242,243 CPL (coordination pillared layer) series MOFs are built by scaffolding 2D Cu(II) and pyrazine-2,3-dicarboxylate layers with dipyridyl ligands. Modifying the pillar ligands allows for systematic control of channel diameters and surface functionalities in CPL series MOFs, which can be produced easily at room temperature. The series of MOFs exhibit exceptional flexibility and a distinct guest-responsive character.244,245 UIO (University of Oslo) series MOFs are three-dimensional porous materials of Zr4+ and dicarboxylic acid ligands. The UIO series has greater chemical and thermal stability than other MOF series. Although the ligands of UIO-MOFs vary in length, UiO-64, UiO-66, UiO-67, UiO-68, UiO-69, and numerous derivatives have the same reticular structure, and altering the ligand does not affect the thermal stability of UIO-MOFs.244,246 ZIF series MOFs are based on zeolitic imidazolate frameworks formed by copolymerizing zinc and cobalt ions with imidazole-type linkers. Zn or Co replace the tetrahedral Si/Al clusters in ZIF structures, whereas bridging imidazolates take the place of the bridging oxygen (O).242,247 IRMOF (isoreticular) series MOFs have the same net architecture. In other words, they have the same topology since they are made of the same organic linkers. For example, IRMOF-1 is made up of tetrahedral Zn4O clusters joined together in a cubic unit by terephthalate links. The terephthalate linker was replaced with additional bidentate linear groups, resulting in a family of MOFs with comparable shape and cubic arrangement known as IRMOF-2, IRMOF-3, etc., or IRMOF-n.248,249 MOFs, especially their composites and derived materials, have been the subject of great attention in recent years. Raissa et al. studied how cobalt content and ligand exchange affect the performance of bimetallic NiCo-MOF, aiming to optimize it. The study found that incorporating cobalt changes the abundance of Ni3+ and Co2+, both electrochemically active species. The results show that a Ni/Co ratio of 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 in NiCo-MOF outperforms (437 C g−1 at 0.5 A g−1) than other ratios. Ligand exchange with a phosphate group increases capacity to 522 C g−1 at 0.5 A g−1. KNiCoPO4//AC has an energy density of 22 W h kg−1 at 363 W kg−1. After 5000 cycles at 5 A g−1, the device retains 99% of initial specific capacitance.250 Meng et al. employed a one-pot solvothermal approach to load Ni/Co-MOFs onto graphite nanosheets and create Ni/Co-MOFs/GNS nanohybrids. GNS, like carbon materials, have high electronic conductivity and stability. The development of Ni/Co-MOFs on the GNS may prevent them from adhering together, improve their spread, increase specific surface area, disclose more redox active sites, and, ultimately, improve electrochemical performance. The Ni/Co-MOFs/GNS nanohybrid had a higher specific capacity of 1080 C g−1 at 1 A g−1. In addition, Ni/Co-MOFs/GNS//AC asymmetric supercapacitor delivered a energy density of 28.7 W h kg−1 with a power density of 849.9 W kg−1.251 Composites made of metal–organic frameworks have considerable potential as electrode materials for supercapacitors due to their ability to combine the benefits of MOFs with other functional materials, such as high conductivity and distinctive electrochemical characteristics.
1 in NiCo-MOF outperforms (437 C g−1 at 0.5 A g−1) than other ratios. Ligand exchange with a phosphate group increases capacity to 522 C g−1 at 0.5 A g−1. KNiCoPO4//AC has an energy density of 22 W h kg−1 at 363 W kg−1. After 5000 cycles at 5 A g−1, the device retains 99% of initial specific capacitance.250 Meng et al. employed a one-pot solvothermal approach to load Ni/Co-MOFs onto graphite nanosheets and create Ni/Co-MOFs/GNS nanohybrids. GNS, like carbon materials, have high electronic conductivity and stability. The development of Ni/Co-MOFs on the GNS may prevent them from adhering together, improve their spread, increase specific surface area, disclose more redox active sites, and, ultimately, improve electrochemical performance. The Ni/Co-MOFs/GNS nanohybrid had a higher specific capacity of 1080 C g−1 at 1 A g−1. In addition, Ni/Co-MOFs/GNS//AC asymmetric supercapacitor delivered a energy density of 28.7 W h kg−1 with a power density of 849.9 W kg−1.251 Composites made of metal–organic frameworks have considerable potential as electrode materials for supercapacitors due to their ability to combine the benefits of MOFs with other functional materials, such as high conductivity and distinctive electrochemical characteristics.
Strategic use of inorganic metal centers and organic ligands may generate MOF materials with a wide range of structures and functional groups. Various techniques, including “standard one-pot synthesis” and “innovative manufacturing” approaches, may be used to produce MOF materials. Various conventional preparation methods, including hydrothermal,252 microwave-assisted,253 electrochemical deposition,254 mechanistic,255 and ultrasonic chemicals,256 are economically viable and environmentally friendly methods for directly extracting complex compounds from readily available materials without the requirement to isolate intermediates. Innovative manufacturing techniques such as spray-drying approach,257 kinetic modulation,258 seed induction,259 and template synthesis260 are being developed to systematically manage the reaction process and achieve a precise synthesis of MOF materials. Nevertheless, all approaches possess limitations in accurately predicting the composition and attributes of the intended result. The synthesis of MOFs involves the coordination of metal ions with organic ligands, and various methods such as solvothermal, hydrothermal, and microwave-assisted synthesis have been employed to produce MOFs with desired properties. Functionalization of MOFs with conductive materials, such as graphene or carbon nanotubes, further enhances their electrochemical performance. For example, MOF-5/graphene composites have demonstrated superior capacitance and conductivity compared to pure MOF-5.261,262
MOFs-based hybrid supercapacitors often exhibit impressive performance metrics, including high energy and power densities. Integrating MOFs with carbon-based materials or conducting polymers can synergistically improve the overall performance of hybrid supercapacitors. A novel bimetallic and dual-linker metal–organic framework (Co-V-MOF) with a microflower form was synthesized by Anwer et al. using the Solvo-hydrothermal technique. This Co-V-MOF exhibits outstanding electrochemical characteristics as a result of its large porosity and large surface area. The boosted Co-V-MOF electrode exhibited a remarkable specific capacitance of 1711.1 F g−1. The distinctive Co-V-MOF//AC electrode arrangement improves electrochemical performance by enhancing electrical conductivity and minimizing ion diffusion routes. The specific capacitance of this hybrid device is measured to be 163.97 F g−1 when using a scan rate of 10 mV s−1. It exhibits astounding energy and power densities of 70.65 W h kg−1 and 835.1 W kg−1, respectively. Consistently and exceptionally, it achieves a performance of 92.21% stability for 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles.263 Hussain and colleagues created a novel freestanding/flexible electrode by coupling porous reduced graphene oxide carbonized nanofiber with conductive bimetallic MOFs (c-MOF@GNF) using electrospinning and in situ c-MOF production in a sequential method. At 1 A g−1, the specific capacitance of the c-MOF@GNF material was 1820 F g−1, with 92.2% of its capacitance remaining after 14
000 cycles.263 Hussain and colleagues created a novel freestanding/flexible electrode by coupling porous reduced graphene oxide carbonized nanofiber with conductive bimetallic MOFs (c-MOF@GNF) using electrospinning and in situ c-MOF production in a sequential method. At 1 A g−1, the specific capacitance of the c-MOF@GNF material was 1820 F g−1, with 92.2% of its capacitance remaining after 14![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles. These qualities are due to its high specific surface area, wettability, conductivity, and availability of active sites. The flexible c-MOF@GNF//GNF asymmetric devices have a high energy density of 52.2 W h kg−1 with a power density of 799.6 W kg−1. The device also demonstrates excellent rate capability and long-term cycle stability, as seen by a 90.6% capacitance retention after 12
000 cycles. These qualities are due to its high specific surface area, wettability, conductivity, and availability of active sites. The flexible c-MOF@GNF//GNF asymmetric devices have a high energy density of 52.2 W h kg−1 with a power density of 799.6 W kg−1. The device also demonstrates excellent rate capability and long-term cycle stability, as seen by a 90.6% capacitance retention after 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles, indicating their practical application.264 One challenge with MOFs is their stability under operational conditions. Recent advancements have focused on enhancing the stability of MOFs in electrolyte solutions and during cycling. For example, Liu et al. reported that by coating MOFs with conductive polymers, the stability and electrochemical performance of MOF-based electrodes were significantly improved. Debendra et al. used a controlled temperature calcination technique to produce Fe3C-integrated electrospun porous carbon nanofiber mats obtained from MIL-88A. These mats are capable of supporting the adhesion of Fe-MOFs on their surface. The methodology used facilitated the transformation of Fe-MOFs into Fe2O3/NPC while preserving the shape of the nanorods. Integrating MIL-88A-derived Fe3C into the negative electrode reduces its inherent resistance and enhances its overall performance. The Fe2O3/NPC-350@Fe3C/EPCNFs electrode, shows a noteworthy specific capacitance of 531 F g−1 at 1 A g−1 and maintained 90% of initial capacitance after 20
000 cycles, indicating their practical application.264 One challenge with MOFs is their stability under operational conditions. Recent advancements have focused on enhancing the stability of MOFs in electrolyte solutions and during cycling. For example, Liu et al. reported that by coating MOFs with conductive polymers, the stability and electrochemical performance of MOF-based electrodes were significantly improved. Debendra et al. used a controlled temperature calcination technique to produce Fe3C-integrated electrospun porous carbon nanofiber mats obtained from MIL-88A. These mats are capable of supporting the adhesion of Fe-MOFs on their surface. The methodology used facilitated the transformation of Fe-MOFs into Fe2O3/NPC while preserving the shape of the nanorods. Integrating MIL-88A-derived Fe3C into the negative electrode reduces its inherent resistance and enhances its overall performance. The Fe2O3/NPC-350@Fe3C/EPCNFs electrode, shows a noteworthy specific capacitance of 531 F g−1 at 1 A g−1 and maintained 90% of initial capacitance after 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles. In addition, a symmetric supercapacitor with an energy density of 21.6 W h kg−1 at a power density of 499.05 W kg−1 and 93% of cycling stability (20
000 cycles. In addition, a symmetric supercapacitor with an energy density of 21.6 W h kg−1 at a power density of 499.05 W kg−1 and 93% of cycling stability (20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles at 20 A g−1) suggest that the electrode produced for this supercapacitor is suitable for practical use in energy storage systems.265 Ongoing research is aimed at optimizing the synthesis and functionalization of MOFs for supercapacitor applications. Novel MOF structures, improved synthesis methods, and better integration strategies with other materials are areas of active investigation. Additionally, scaling up the production of MOF-based electrodes while maintaining their performance remains a crucial aspect for practical applications.266,267
000 cycles at 20 A g−1) suggest that the electrode produced for this supercapacitor is suitable for practical use in energy storage systems.265 Ongoing research is aimed at optimizing the synthesis and functionalization of MOFs for supercapacitor applications. Novel MOF structures, improved synthesis methods, and better integration strategies with other materials are areas of active investigation. Additionally, scaling up the production of MOF-based electrodes while maintaining their performance remains a crucial aspect for practical applications.266,267
COFs are notable for their structural stability and porosity. Their high surface area and well-defined pore structures facilitate efficient ion transport and storage. Recent studies have demonstrated that COFs can offer high specific capacitance and excellent rate performance in supercapacitor applications.270 For instance, constructing conductive COFs is viable for developing high-performance COF-based supercapacitors. Li et al. developed a Ni-COF using Ni(II)-salphen units. It can conduct electricity up to 1.3 × 10−2 S cm−1. Ni-COF has a high specific capacitance of 1257 F g−1 at 1 A g−1 and 94% cycling stability after 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles. The AC//Ni-COF asymmetric supercapacitor produced a high energy density of 130 W h kg−1 and power density of 839 W kg−1. The COF without Ni2+ had an electrical conductivity of 8.4 × 10−6 S cm−1 and a substantially lower capacitance of 184 F g−1 at 1 A g−1. Strong π-conjugation and square-planar Ni(II) coordination may have contributed to the considerable rise in electrical conductivity, further improving electrochemical performance.271 Several COFs have been explored for supercapacitor applications, including TADF-COFs, TpPa-COFs, and COF-5. These frameworks offer various properties such as high electrical conductivity and stability. TADF-COFs, for example, have shown superior charge storage capabilities due to their large surface area and high porosity.272,273 Similarly, TpPa-COFs have demonstrated excellent capacitance retention and cycling stability due to their strong covalent linkages and stable framework structure.274,275
000 cycles. The AC//Ni-COF asymmetric supercapacitor produced a high energy density of 130 W h kg−1 and power density of 839 W kg−1. The COF without Ni2+ had an electrical conductivity of 8.4 × 10−6 S cm−1 and a substantially lower capacitance of 184 F g−1 at 1 A g−1. Strong π-conjugation and square-planar Ni(II) coordination may have contributed to the considerable rise in electrical conductivity, further improving electrochemical performance.271 Several COFs have been explored for supercapacitor applications, including TADF-COFs, TpPa-COFs, and COF-5. These frameworks offer various properties such as high electrical conductivity and stability. TADF-COFs, for example, have shown superior charge storage capabilities due to their large surface area and high porosity.272,273 Similarly, TpPa-COFs have demonstrated excellent capacitance retention and cycling stability due to their strong covalent linkages and stable framework structure.274,275
TADF-COFs are a class of covalent organic frameworks designed to exhibit thermally activated delayed fluorescence, a property that significantly enhances their performance in optoelectronic and energy storage applications. For supercapacitor applications, TADF-COFs are highly attractive due to their large surface area and high porosity, which are crucial for maximizing ion storage and transport. The interconnected porous structure allows for rapid ion diffusion, improving both the charge–discharge rates and the overall energy density. Furthermore, the molecular arrangement of TADF-COFs often facilitates efficient charge separation and transfer due to their conjugated structures, which contribute to high electrical conductivity.272,273 The combination of these properties allows for superior charge storage capabilities compared to other types of COFs. Additionally, their tunable electronic properties, owing to the specific donor–acceptor interactions in TADF-COFs, make them highly adaptable for energy storage devices requiring both high power and energy densities.
TpPa-COFs, constructed from terephthalaldehyde (Tp) and phenylenediamine (Pa), are another class of COFs that have gained attention in supercapacitor applications. These COFs are known for their strong covalent linkages, typically involving Schiff-base chemistry, which results in a highly stable framework. This stability is crucial for maintaining structural integrity during long-term charge–discharge cycling, leading to excellent capacitance retention over thousands of cycles. TpPa-COFs also feature a highly ordered crystalline structure, enhancing their mechanical robustness and ensuring that the ion diffusion pathways remain open and accessible throughout cycling.274,275 Furthermore, the chemical stability of the TpPa framework, even in harsh electrolytic environments, makes them ideal for use in aqueous and organic electrolytes. The strong covalent bonds in the structure minimize the risk of framework collapse or degradation, allowing TpPa-COFs to exhibit outstanding cycling stability compared to other porous materials.
COF-5 is one of the first reported COFs constructed from boronic acid building blocks, forming a two-dimensional, highly porous, and crystalline framework.276,277 COF-5 has been extensively studied for various applications, including supercapacitors, due to its high surface area and well-defined pore structure. The key advantage of COF-5 in supercapacitor applications lies in its ability to achieve high charge storage capacity due to its large surface area, which facilitates the adsorption of ions from the electrolyte. The boronic ester linkages in COF-5 contribute to a rigid framework that is relatively stable under operating conditions. However, boron-based linkages are often sensitive to moisture, limiting the practical applications of COF-5 in humid or aqueous environments without modification. Despite this, COF-5's intrinsic high porosity and electrical properties make it a promising candidate for supercapacitors, especially when paired with protective coatings or encapsulated in hybrid structures to improve stability. Fig. 12 showcases the chemical structures of distinct building blocks, each designed with unique geometrical features that directly influence their assembly and functional properties within complex materials. The diversity in geometry—from linear, planar, to more intricate 3D structures—enables precise control over the spatial arrangement and interactions of these molecules in a final composite material. Geometrical variation among these building blocks is not merely structural; it also dictates the types of bonding interactions (such as π–π stacking, hydrogen bonding, and covalent bonding) that can occur during material formation. Linear geometries, for example, often facilitate straightforward polymerization or chain-like assemblies, allowing for high flexibility and adaptability in dynamic environments. Planar structures, on the other hand, promote efficient stacking interactions which are favorable for the creation of layered, high-density materials, often resulting in enhanced electronic and optical properties due to close molecular packing. In more complex, branched, or 3D geometries, the building blocks can introduce steric bulk and functional diversity that prevent aggregation, which is beneficial in applications requiring solubility and dispersion, such as drug delivery or catalysis. Additionally, 3D geometries can create voids or channels within the material matrix, leading to porous structures ideal for applications in gas storage, separation, or catalysis. The strategic design of these building blocks with specific geometrical characteristics enables tailoring of the final material's mechanical stability, electronic properties, and reactivity. Thus, the varied geometries of these chemical structures are not just fundamental to molecular design but are also essential in enabling the versatile, tunable properties required for next-generation materials with applications across fields such as biomedicine, electronics, and energy storage. Fig. 12 thus underscores the importance of geometry in chemical design, providing a molecular blueprint for achieving targeted functionality in advanced materials.
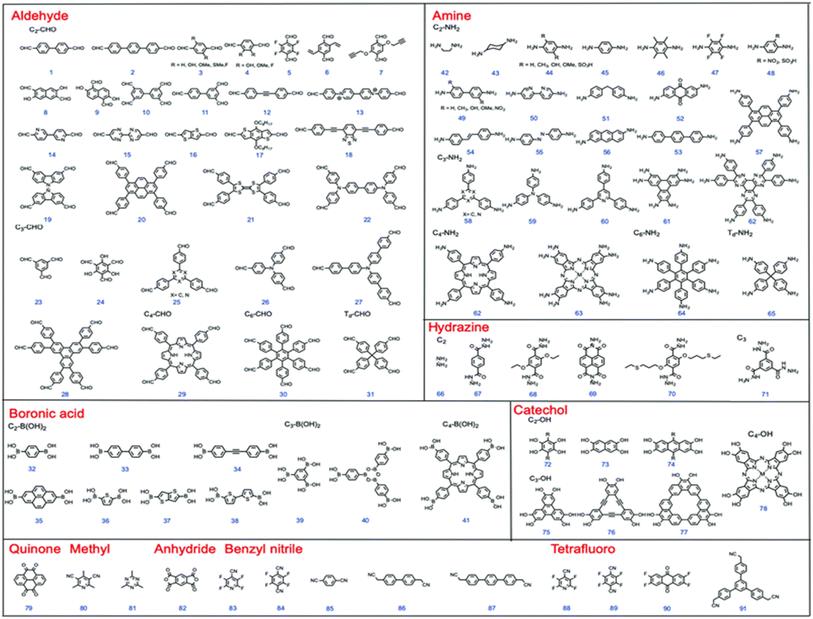 | ||
| Fig. 12 Chemical structures of foundational building blocks exhibiting various geometries,278 Reproduced from ref. 278 with permission from the Royal Society of Chemistry, copyright 2020. | ||
The synthesis of COFs is a crucial process that involves precise control over reaction conditions to yield highly ordered, porous crystalline structures with tailored properties. COFs are generally synthesized through the condensation of organic monomers that form stable covalent bonds, such as boronate esters, imines, or triazines, which are linked under controlled conditions to create frameworks with predictable pore sizes, shapes, and functionalities. Various synthesis methods have been developed to optimize the structural and functional properties of COFs, each offering distinct advantages in terms of crystallinity, pore accessibility, and scalability. Solvothermal and hydrothermal synthesis: solvothermal and hydrothermal methods are among the most commonly used techniques in COF synthesis.279 These approaches involve heating monomers in a solvent (organic or water-based) at elevated temperatures within sealed vessels, often for extended periods. The choice of solvent, temperature, and reaction time plays a critical role in determining the morphology and crystallinity of the resulting COF. Solvothermal synthesis, in particular, allows for high degrees of customization in the framework, as solvents and additives can influence monomer orientation and pore structure. However, these methods can be time-intensive, making scalability a challenge for industrial applications. Microwave-assisted synthesis: microwave-assisted synthesis has emerged as a promising technique to enhance the efficiency of COF production. By providing rapid, uniform heating, microwave synthesis can significantly reduce reaction times (often from days to hours or even minutes) while improving crystallinity.
Additionally, this method facilitates better control over particle size and morphology, as the rapid heating minimizes the likelihood of aggregation. Microwave-assisted synthesis is particularly advantageous for producing COFs with precise, reproducible structures, making it a preferred choice for applications requiring batch consistency and scalability. Functionalization with conductive materials: to improve the electrochemical properties of COFs, functionalization with conductive materials such as graphene or carbon nanotubes is a common strategy. COFs alone may lack the necessary electrical conductivity for applications in energy storage; however, functionalizing COFs with conductive additives can bridge this gap by providing a pathway for electron transport. Graphene, with its high surface area and conductivity, has proven especially effective. Liu et al. demonstrated that COF-5 composites with reduced graphene oxide (rGO) exhibited a marked increase in electrical conductivity and capacitance, enhancing performance in supercapacitor applications. This composite approach not only retains the porosity and structural stability of the COF but also benefits from the synergistic effects of rGO's high conductivity, which facilitates rapid charge–discharge cycles.279–281
Post-synthetic modification and functional group incorporation: beyond conductive material integration, post-synthetic modification techniques enable further functionalization of COFs by adding desired chemical groups, such as sulfonic acid groups or amines, directly onto the COF structure. These modifications can improve the affinity for specific ions or enhance stability under operational conditions, making COFs more versatile in various applications. Functional group incorporation can also increase the COF's affinity for conductive additives, thereby improving the uniformity and effectiveness of material integration. Overall, advancements in COF synthesis and functionalization have expanded their potential applications, especially in fields requiring high electrochemical performance, such as supercapacitors, batteries, and catalysis. By refining synthesis techniques and exploring novel functionalization methods, researchers can optimize COFs for specific applications, improving properties such as conductivity, stability, and ion transport. The combination of these strategies is paving the way for COFs to play a significant role in next-generation energy storage and conversion technologies.
The synthesis method depicted in Fig. 13 demonstrates an innovative approach for constructing 2D polymers with high structural precision, achieved at the air–water interface using surfactant monolayers. This interfacial synthesis leverages the unique environment created by surfactants, which not only stabilizes monomers but also restricts their movement to two dimensions, thus facilitating planar polymerization. The reaction between 2DPI and 2DPA, as shown in panel (b), indicates a carefully controlled process where monomer alignment at the interface is critical for achieving ordered polymer networks. TEM and AC-HRTEM images in panels (c) and (d) provide direct evidence of the polymer's crystallinity and structural regularity, with the atomic-level resolution of AC-HRTEM revealing periodic lattice structures that confirm the success of the 2D polymerization technique. This crystallinity is essential for potential electronic and optical applications, where uniformity and periodicity at the molecular level translate into predictable electronic properties. The AFM image in panel (e) further elucidates the film's smooth topology, indicating a thin, continuous structure that could be advantageous in applications requiring large surface-area-to-volume ratios, such as catalysis or sensing. Collectively, these images validate the method's efficacy in producing high-quality 2D polymers, highlighting the potential of this synthesis strategy to enable scalable production of structurally precise, functionally tunable 2D materials for diverse scientific and industrial applications.
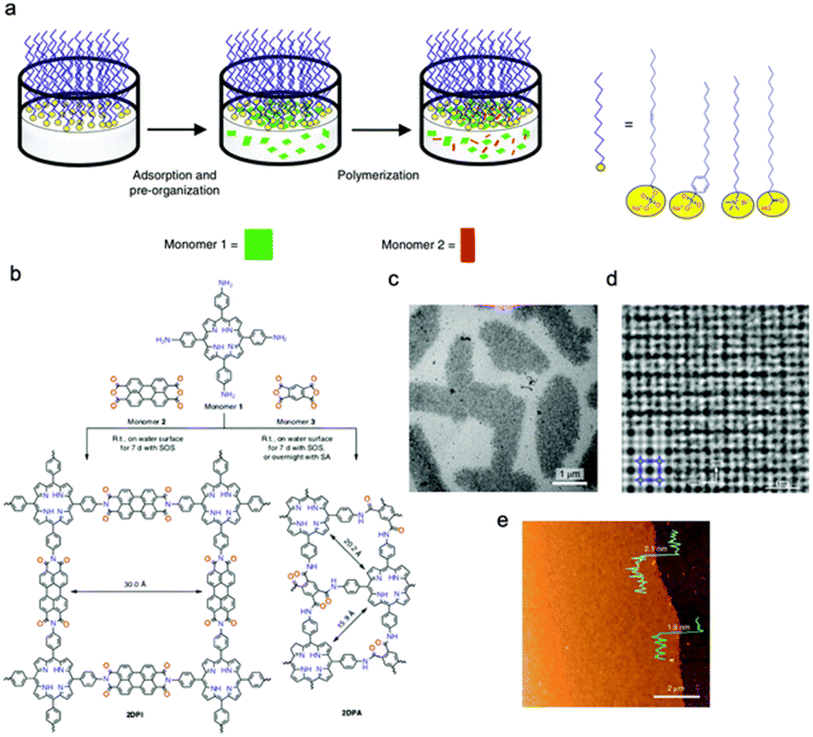 | ||
| Fig. 13 (a) Diagram illustrating the synthesis process for 2D polymers formed at the water–air interface, facilitated by surfactant monolayers. (b) Diagram showing the reaction between 2DPI and 2DPA. (c) TEM image depicting the 2DPI film. (d) AC-HRTEM image showcasing 2DPI. (e) AFM image showing the 2DPI film structure. Reproduced from ref. 282 with permission from Nature Publishing Group, copyright 2019. | ||
COFs (Covalent Organic Frameworks) have gained significant attention for their potential in hybrid supercapacitors, thanks to their high surface area, tunable porosity, and customizable chemical functionalities. These properties make COFs highly efficient for charge storage and ion transport, contributing to their notable energy and power densities. However, COFs alone can suffer from limitations in electrical conductivity and cycling stability. To address these challenges, researchers have explored the integration of COFs with other high-performance materials, such as carbon-based materials and conductive polymers, creating hybrid structures that harness the unique advantages of each component. One successful approach is the combination of COFs with carbon-based materials, like graphene, to enhance conductivity and overall structural integrity. For example, COF/graphene composites leverage the conductive network of graphene, which supports rapid electron transfer and provides mechanical stability, while the COF component contributes to high energy storage capacity through ion adsorption and desorption within its porous structure. Ibrahim et al. demonstrated that COF/graphene composites could achieve an impressive energy density of 0.8 A g−1 and a power density of 234.0 F g−1. This synergy between COFs and graphene enhances the composite's capacitive performance, achieving a balance between high energy density, characteristic of batteries, and high power density, which is typical for supercapacitors.283 Furthermore, integrating COFs with conductive polymers, such as polyaniline or polypyrrole, has shown potential in improving electrochemical performance. Conductive polymers not only improve the electrical conductivity of the hybrid supercapacitor but also offer pseudocapacitive behavior, which boosts energy storage through faradaic reactions. This approach effectively combines the double-layer capacitance of COFs with the pseudocapacitance from conducting polymers, resulting in a hybrid with superior energy density and cycling stability. In these systems, the COF provides a structured matrix with substantial ion-accessible surface area, while the polymer fills voids and interstitial spaces, facilitating efficient ion transport. The incorporation of COFs into hybrid supercapacitors thus opens pathways for achieving high-performance energy storage devices. By combining COFs with materials that provide complementary electrical and mechanical properties, hybrid supercapacitors can deliver substantial energy and power densities, making them competitive with both batteries and traditional supercapacitors. Continued research on optimizing these hybrids, including the exploration of different COF structures and functionalization strategies, could unlock even greater performance, with promising applications in portable electronics and electric vehicles where high energy density and rapid power delivery are essential.
Despite their high tunability and unique porosity, COFs face substantial challenges regarding stability in electrolyte solutions, particularly during prolonged electrochemical cycling. COFs often exhibit susceptibility to degradation under operational conditions, primarily due to interactions with electrolytes and mechanical stresses that weaken their covalent bonds. Stability concerns in COFs arise from both chemical and structural vulnerabilities, limiting their practical applications in energy storage devices. To address these limitations, various stabilization strategies have emerged. Encapsulation within protective layers, for example, helps isolate COFs from harsh electrolyte interactions, preserving their structure over extended cycles. Conductive polymer coatings have been particularly promising, as they provide a dual benefit: physical protection of the COF and enhanced electrical conductivity. Recently, researchers demonstrated a significant improvement in stability and electrochemical performance by coating COF electrodes with conductive polymers. This approach mitigates the dissolution and structural collapse often seen in COF materials, extending their lifespan under cyclic stress.284,285 Additionally, covalent bonding reinforcement strategies have been developed to stabilize the linkages within COFs. For instance, modifying the framework with stronger covalent bonds, such as boron–nitrogen linkages or incorporating metal–organic cross-links, can reduce bond cleavage rates. These modifications can enhance framework rigidity, thus reducing the likelihood of degradation during cycling. Thermal and chemical post-treatments have also shown promise in improving COF robustness by reinforcing interlayer interactions, further enhancing stability. While these approaches mark significant progress, the long-term stability of COFs in high-energy applications remains a challenge. Future research is likely to focus on hybrid materials and advanced coatings that offer multifunctional protection, aiming to make COFs a more reliable and practical choice for energy storage and conversion devices.
Ongoing research aims to optimize the synthesis and functionalization of COFs for supercapacitor applications. Innovations in COF design, including the development of new building blocks and improved synthesis methods, are expected to enhance the performance and practicality of COF-based supercapacitors. Additionally, efforts to scale up production while maintaining performance are critical for practical applications.286,287
Although significant progress and attention have been given to supercapacitors, selecting the appropriate and effective electrode material remains challenging. To choose a material suitable for achieving high energy density in supercapacitors, the following criteria must be met:
➢ High specific surface area to enable optimal specific capacitance
➢ Accurate rate capability and stable capacitance characteristics
➢ Excellent ionic conductivity
➢ A sufficient number of electro-active sites
➢ Adequate thermal, physical, and chemical stability to ensure cyclic stability and support sustainable manufacturing
A supercapacitor would not be complete without the electrolyte, which is responsible for balancing and transporting charges between the two electrodes. Electrolytes may influence the essential factors that affect supercapacitor devices, including power density, cyclic stability, capacity, and energy density. The physical and chemical parameters of the electrolyte are critical to how a supercapacitor works. The interior structure of active materials may significantly influence the interaction between electrode and electrolyte.288 A suitable electrolyte is essential for supercapacitors to be safe and work well. Fig. 14 illustrates the classification of electrolytes into liquid, solid, or quasi-solid-state. The liquid electrolytes are further classified as aqueous and non-aqueous. Researchers focus on the aqueous system because of its strong ionic conductivity and inexpensive cost. Non-aqueous electrolytes, namely organic-based electrolytes, are widely employed in commercial supercapacitors. A supercapacitor with a non-aqueous electrolyte costs about $2400 per kW per h, which is much more than regular batteries.289 The solid-state electrolyte acts as both a mild ionic conductor and a separator, significantly reducing packing size. However, it has certain drawbacks, including poor electrolyte–electrode interaction and a low capacitance value. So, the optimal electrolyte that fits all of the criteria of a supercapacitor device has yet to be developed.
3.5. Aqueous electrolytes
Aqueous electrolytes are critical components in hybrid supercapacitors, influencing their performance, safety, and environmental impact. These electrolytes, based on water as the solvent, are widely used due to their low cost, high ionic conductivity, and environmental friendliness. The selection and optimization of aqueous electrolytes involve various factors such as electrolyte concentration, pH, and ionic species, which collectively impact the electrochemical performance of hybrid supercapacitors.➢ Types of aqueous electrolytes: aqueous electrolytes generally include solutions of inorganic salts, organic acids, or a combination of both. The most common inorganic salts used are KOH, H2SO4, and sodium sulfate (Na2SO4). These electrolytes are favored for their high ionic conductivity and broad electrochemical stability windows.290,291 For example, KOH is widely used in supercapacitors due to its ability to provide high specific capacitance and power density.292
➢ Performance characteristics: the performance of aqueous electrolytes in hybrid supercapacitors is closely related to their ionic conductivity and electrochemical stability. High ionic conductivity facilitates efficient charge transfer and faster charge/discharge rates. The electrochemical stability window defines the maximum voltage that can be applied without electrolyte decomposition.10,293 For instance, Zhang et al. prepared Co3O4/ACC@RGO//ACC@RGO hybrid supercapacitor device with neutral low-concentration aqueous electrolytes and achieved excellent cycling stability and a wide potential window from 0 to 2.2 V.294
➢ Advantages and limitations: aqueous electrolytes offer several advantages, including high ionic conductivity, low cost, and safety compared to organic electrolytes. However, their practical application is limited by their narrower electrochemical stability window and lower energy density compared to non-aqueous electrolytes. This limitation restricts their use in high-voltage applications.295 Recent research has focused on improving the performance of aqueous electrolytes by exploring novel ionic liquids and electrolyte additives. For example, the addition of ionic liquids to aqueous electrolytes has been shown to enhance their electrochemical stability and extend the operating voltage range.296,297 Additionally, the use of advanced electrolyte formulations, such as mixed aqueous-organic systems, has demonstrated improved energy density and cycle stability.298
➢ Novel electrolyte combinations: combining different salts or integrating organic acids with inorganic salts can tailor the properties of the electrolyte. For instance, solutions of potassium sulfate (K2SO4) and citric acid have been reported to offer a balance between high ionic conductivity and stability, leading to enhanced performance in supercapacitors.299 Another example includes the development of high-concentration electrolyte systems that provide increased capacitance and power density.300
➢ Applications and future directions: aqueous electrolytes are particularly suitable for applications requiring high safety and environmental friendliness. Future research is expected to focus on optimizing the composition of aqueous electrolytes to improve their performance metrics, such as enhancing their electrochemical stability and broadening their operational voltage range. Exploring innovative electrolyte formulations and additives will likely lead to advancements in hybrid supercapacitor technology.295,301,302
3.6. Organic electrolytes
Organic electrolytes are gaining prominence in hybrid supercapacitors due to their advantageous properties, such as broader electrochemical stability windows, higher energy density, and enhanced safety compared to traditional aqueous electrolytes. This section explores the types, characteristics, and recent advancements in organic electrolytes for hybrid supercapacitors.➢ Types of organic electrolytes: organic electrolytes are typically classified into two main categories: organic solvents with dissolved salts and ionic liquids. Organic solvents include common solvents such as acetonitrile, propylene carbonate, and ethylene carbonate, often paired with salts like tetraethylammonium tetrafluoroborate (TEABF4) or lithium hexafluorophosphate (LiPF6);302,303 ionic liquids, such as 1-ethyl-3-methylimidazolium tetrafluoroborate (EMIM-BF4), are salts in the liquid state at room temperature and provide high stability and low volatility.304,305
➢ Electrochemical performance: organic electrolytes generally offer a higher electrochemical stability window compared to aqueous systems, often exceeding 2.5 V, which enables hybrid supercapacitors to achieve greater energy densities.306 For instance, acetonitrile-based electrolytes combined with TEABF4 can operate effectively up to 3.5 V, which is beneficial for increasing the voltage and energy density of supercapacitors.307,308 Ionic liquids provide even broader stability windows, up to 4 V or higher, which can significantly enhance the energy storage capabilities of hybrid supercapacitors.309
➢ Advantages and limitations: the primary advantages of organic electrolytes include their wide electrochemical stability windows, high ionic conductivity, and safety features. However, they also present certain limitations such as higher cost, potential environmental impact, and limited ionic conductivity compared to aqueous electrolytes.310,311 For instance, while ionic liquids offer exceptional stability, their lower viscosity can reduce the ionic conductivity, impacting the charge/discharge rates.312,313 Recent research has focused on developing new organic electrolyte formulations and optimizing their performance. Innovations such as gel-type electrolytes, which combine organic solvents with gelling agents, have been shown to improve the mechanical stability and ionic conductivity of the electrolyte while maintaining its electrochemical performance.314,315 Furthermore, the development of new ionic liquids with lower viscosities and enhanced conductivity has also been explored.316
➢ Emerging trends: researchers are investigating hybrid organic-aqueous electrolyte systems to combine the best features of both types. For example, organic–inorganic hybrid electrolytes have demonstrated improved electrochemical stability and higher energy density compared to purely organic or aqueous systems.317 Additionally, the use of eco-friendly and biodegradable organic solvents is being explored to address environmental concerns associated with organic electrolytes.318,319 Organic electrolytes are particularly suited for high-energy applications in hybrid supercapacitors. Future research is expected to focus on enhancing the ionic conductivity of organic electrolytes, developing new ionic liquids with better performance characteristics, and addressing environmental impacts. Innovations in electrolyte formulations and the development of hybrid systems will likely drive advancements in supercapacitor technology.10,320,321
3.7. Ionic liquids
Ionic liquids (ILs) are a class of organic salts that remain in the liquid state over a wide temperature range, making them highly suitable for various energy storage applications, including hybrid supercapacitors. Their unique properties, such as high ionic conductivity, wide electrochemical stability window, and low volatility, make them an attractive choice for enhancing the performance of supercapacitors.➢ Characteristics of ionic liquids: ionic liquids are characterized by their ability to remain liquid at room temperature or slightly above due to their low melting points. They combine organic cations and inorganic anions, contributing to their diverse physicochemical properties.322,323 Commonly used ionic liquids include 1-ethyl-3-methylimidazolium tetrafluoroborate (EMIM-BF4) and 1-butyl-3-methylimidazolium hexafluorophosphate (BMIM-PF6).324,325 These ILs offer high ionic conductivity, excellent thermal stability, and a broad electrochemical window, which can exceed 4 V in some cases.326,327
➢ Electrochemical performance: using ionic liquids in hybrid supercapacitors enhances their electrochemical performance by providing high voltage stability and improving energy density. For instance, EMIM-BF4 has been shown to provide an electrochemical stability window from 2.35 to 4.5 V, which allows for greater energy storage than traditional aqueous electrolytes.328 The high ionic conductivity of ionic liquids also facilitates efficient charge and discharge processes, resulting in improved power density and cycle life.329,330
➢ Advantages and challenges: ionic liquids offer several advantages, including non-flammability, low vapor pressure, and chemical stability, which make them safer and more environmentally friendly compared to conventional organic solvents.331,332 However, challenges remain, such as their relatively high cost and viscosity, which can affect the ionic conductivity and overall performance of supercapacitors.333,334 Additionally, the compatibility of ionic liquids with various electrode materials and the potential for environmental impact need further investigation.335,336 Recent research has focused on improving the performance and cost-effectiveness of ionic liquids for supercapacitor applications. Innovations include the development of novel ionic liquid formulations with enhanced ionic conductivity and broader electrochemical stability windows.41,330 For example, new classes of ionic liquids with tailored anions and cations have been synthesized to optimize their performance in supercapacitors.337,338 Additionally, research is exploring the use of ionic liquid-based gel electrolytes, which combine the advantages of ionic liquids with the mechanical stability of gels.339,340
➢ Emerging trends: the integration of ionic liquids with other electrolyte systems, such as hybrid ionic liquid-aqueous electrolytes, is an emerging trend that aims to combine the benefits of both types.341 Moreover, there is ongoing research into ionic liquids' environmental impact and recyclability to address sustainability concerns.310,342 Innovations in this field will lead to more efficient, cost-effective, and environmentally friendly energy storage solutions.302,343,344 Ionic liquids hold significant potential for enhancing the performance of hybrid supercapacitors in applications requiring high energy density and stability. Future research will likely focus on optimizing ionic liquid formulations, improving their cost and environmental impact, and exploring new applications in energy storage and beyond.
So the ionic liquids (ILs) offer promising advantages for hybrid supercapacitors due to their wide electrochemical stability window, high ionic conductivity, and thermal stability. They enable higher energy and power densities compared to traditional electrolytes, enhancing overall device performance. Despite challenges such as high viscosity and cost, ongoing research into novel IL formulations and gel-based systems aims to overcome these limitations. Future developments are expected to focus on improving sustainability, compatibility with various electrodes, and expanding their applicability in advanced energy storage technologies.
3.8. Solid-state electrolytes
Advantages:
➢ Enhanced safety: gel electrolytes reduce leakage risks associated with liquid electrolytes, thus improving the overall safety of supercapacitors. They also exhibit reduced flammability compared to organic solvents used in traditional electrolytes.348,349
➢ Improved mechanical properties: the gel matrix provides flexibility and mechanical robustness, which is advantageous for wearable and flexible electronics. This flexibility also facilitates better integration into various device architectures.350,351
➢ Wide electrochemical stability window: gel electrolytes can operate across a broad voltage range, enhancing the performance of hybrid supercapacitors. This stability contributes to higher energy and power densities.339,352
Recent studies have focused on developing new gel electrolyte formulations to further enhance their performance. For example, researchers have investigated the use of hybrid polymers such as poly(vinylidene fluoride) (PVDF) combined with ionic liquids to achieve higher ionic conductivity and better thermal stability.353,354 Additionally, advanced synthesis techniques such as electrospinning have been employed to create nanostructured gel electrolytes with improved performance metrics.355,356 Despite their advantages, gel electrolytes face challenges such as limited ionic conductivity at low temperatures and degradation over extended use. Ongoing research aims to address these issues by exploring novel polymer matrices and ionic salts.315,357,358 Furthermore, there is a growing interest in developing eco-friendly and cost-effective gel electrolytes to meet the increasing demand for sustainable energy storage solutions.
Advantages:
➢ Enhanced safety and stability: polymer electrolytes eliminate the risk of leakage and reduce flammability compared to liquid electrolytes, enhancing the safety of supercapacitors. Their solid-state nature also contributes to better thermal stability and longer lifespan.362
➢ Flexibility and processability: the flexibility of polymer electrolytes makes them suitable for use in flexible and wearable electronic devices. Additionally, their processability allows for easy integration into various device architectures.351
➢ Wide electrochemical window: polymer electrolytes can operate over a broad voltage range, contributing to the high energy density of hybrid supercapacitors. This electrochemical stability is crucial for achieving high-performance energy storage.363
Recent research has focused on enhancing the performance of polymer electrolytes through various strategies. For example, the incorporation of inorganic fillers or nanomaterials into polymer matrices has been shown to improve ionic conductivity and mechanical properties.361 Researchers have also explored the use of new polymer chemistries and combinations to develop electrolytes with superior performance characteristics.364 Advanced techniques such as electrospinning have been employed to create nanofiber-based polymer electrolytes with enhanced properties.365 Despite their advantages, polymer electrolytes face challenges such as limited ionic conductivity at low temperatures and potential degradation over long-term use. Addressing these challenges involves exploring new polymer materials, optimizing the polymer–electrolyte interface, and developing strategies to enhance the ionic conductivity at various operating temperatures.366,367 Furthermore, there is a growing interest in developing eco-friendly and cost-effective polymer electrolytes to meet the increasing demand for sustainable energy storage solutions.318 Finally, a detailed comparison of the electrochemical performances of the electrode materials, evaluated under both three-electrode and two-electrode configurations, is summarized and presented in Table 2. This comparison includes parameters such as the potential window, specific capacitance at corresponding current densities, cyclic stability, power density, energy density, and the respective electrolytes used in each configuration, providing a comprehensive overview of their electrochemical behavior.
| S. No. | Three-electrode configuration | Stability | Two-electrode configuration | Ref. | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Electrode | Electrolyte | Potential window | Specific capacitance at a current density | Device and electrolyte | Potential window | Specific capacitance @ current density | Power density and energy density | Stability | |||
| 1 | F-rGO/PANI | 1 M H2SO4 | 0.0 to 0.8 V | 597 F g−1 @ 0.5 A g−1 | 75% after 2000 cycles @ 1 A g−1 | — | 151 | ||||
| 2 | Carbon aerogel | 1 M H2SO4 | 0.0 to 0.8 V | 138 F g−1 @ 0.5 A g−1 | 102% after 5000 cycles @ 1 A g−1 | — | — | — | 10.06 W h kg−1 @ 181.06 W kg−1 | — | 368 |
| 3 | NSAC | 1 M H2SO4 | 0.0 to 1.0 V | 417 F g−1 @ 0.7 A g−1 | 76% after 60![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles at 5 A g−1 000 cycles at 5 A g−1 |
— | 133 | ||||
| 4 | PAC | 6 M KOH | −1.0 to 0.0 V | 470 F g−1 @ 0.5 A g−1 | 98% after 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles at 10 A g−1 000 cycles at 10 A g−1 |
— | 134 | ||||
| 5 | AC/MWCNTs/ZnFe2O4 | 3 M KOH | −0.2 to 0.6 V | 609 F g−1 @ 1 A g−1 | 91% after 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles @ 1 A g−1 000 cycles @ 1 A g−1 |
— | 175 | ||||
| 6 | α-MnO2 nanowires | 1 M Na2SO4 | 0.0 to 0.8 V | 43.8 F g−1 @ 0.5 A g−1 | 99% after 1000 cycles @ 0.5 A g−1 | — | 369 | ||||
| 7 | NiCo-LDH/50 CNB | 1 M KOH | 0.0 to 0.5 V | 1220 F g−1 @ 1 A g−1 | 91% after 5000 cycles @ 1 A g−1 | — | 214 | ||||
| 8 | PANI nanowires | 0.5 M H2SO4 | −0.2 to 0.8 V | 540 F g−1 @ 0.5 A g−1 | 82% after 1000 cycles @ 5 A g−1 | — | 370 | ||||
| 9 | Camellia seed shell-derived activated carbon | 3 M KOH | −1.0 to 0.0 V | 305 F g−1 at 0.5 A g−1 | — | CSSC-KOH//CSSC-KOH; KOH | 0–1.3 V | 65.5 F g−1 @ 0.3 A g−1 | 15.37 W h kg−1 at 195 W kg−1 | 96% after 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles @ 2 A g−1 000 cycles @ 2 A g−1 |
371 |
| 10 | BW-AC | 6 M KOH | 0.0 to 1 V | 149 F g−1 @ 0.5 A g−1 | — | — | 3.3 W h kg−1 @ 3930 W kg−1 | 85.6% after 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles @ 5 A g−1 000 cycles @ 5 A g−1 |
372 | ||
| BW-AC | 1 M TEA-BF4 organic electrolyte | 0.0 to 2.7 V | 106 F g−1 @ 0.5 A g−1 | — | 12.7 W h kg−1 @ 9164 W kg−1 | 79.6% after 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles @ 5 A g−1 000 cycles @ 5 A g−1 |
|||||
| 11 | V-5 | 3 M KOH/water/DMF | 0.0 to 0.68 V | 306 F g−1 @ 1.2 A g−1 | 98% after 5000 cycles @ 5 A g−1 | Graphite//V-5 (AC); KOH/water/DMF/PVA | 0–2.2 V | 110.7 F g−1 @ 3.5 A g−1 | 61.54 W h kg−1 @ 3500 W kg−1 | 92% after 5000 cycles | 373 |
| 12 | Lotus seedpods derived carbon | 6 M KOH | −1.0 to 0.0 V | 419 F g−1 @ 0.5 A g−1 | 90.1% after 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles at 10 A g−1 000 cycles at 10 A g−1 |
LSAC-Tpre 400-K3//LSAC-Tpre 400-K3 | −1.0 to 0.0 V | — | 30.3 W h kg−1 @ 250 W kg−1 | — | 131 |
| 13 | MnCo2O4/AC/PPy | 6 M KOH | −1.0 to 1.0 V | 834.84 F g−1 @ 4 A g−1 | 94.14% after 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles @ 4 A g−1 000 cycles @ 4 A g−1 |
MAP-20//MnO2/AC ASC | 0.0 to 1.6 V | 247.85 F g−1 @ 2 A g−1 | 88.12 W h kg−1 @ 1.6 kW kg−1 | 89.68% after 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles @ 2 A g−1 000 cycles @ 2 A g−1 |
141 |
| 14 | PF-rGO/PANI | 1 M H2SO4 6 M KOH | 0.0 to 0.8 V | 489 F g−1 @ 0.5 A g−1 | 89% after 2000 cycles @ 1 A g−1 | 151 | |||||
| rGO-NiCo2S4 | 0.0 to 0.45 V | 972 F g−1 @ 1 A g−1 | — | rGO-NiCo2S4//AC ASC | 0.0 to 1.4 V | 208 F g−1 @ 1 A g−1 | 56.62 W h kg−1 @ 701.37 W kg−1 | 94.1% after 2000 cycles @ 3 A g−1 | |||
| 15 | C/Ru@NiO-CuO | 2 M KOH | 0.0 to 0.6 V | 318 F g−1 @ 0.5 A g−1 | 86.7% after 5000 cycles @ 10 A g−1 | CRNC//rGO-Co3O4 | 0.0 to 1.0 V | 152.5 F g−1 @ 0.5 A g−1 | 21.18 W h kg−1 @ 250 W kg−1 | 87% after 3500 cycles @ 10 A g−1 | 161 |
| 16 | Fe2O3 NDs/RGO | 6 M KOH | 0.0 to 0.6 V | 347.4 F g−1 @ 1 A g−1 | — | a-Fe2O3 NDs/rGO//Co3O4 NDs/rGO | 0.0 to 1.5 V | 69.1 F g−1 @ 1 A g−1 | 21.6 W h kg−1 @ 750 W kg−1 | 94.3% after 5000 cycles @ 5 A g−1 | 159 |
| 17 | CNTs@Gr-CNF | 6 M KOH | −1.0 to 0.0 V | 521.5 F g−1 @ 0.25 A g−1 | 98% after 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles @ 5 A g−1 000 cycles @ 5 A g−1 |
CNTs@Gr-CNF//NiCo2S4/Ni-foam | 0.0 to 1.5 V | 218 F g−1 @ 1 A g−1 | 62.13 W h kg−1 @ 789.66 W kg−1 | 91.7% after 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles at 5 A g−1 000 cycles at 5 A g−1 |
177 |
| 18 | Mn-Ni(3-HPCA)MOF/HA-CNT | 3 M KOH | −0.6 to 0.0 V | 6733 F g−1 @ 0.25 A g−1 | — | Mn-Ni(3-HPCA)MOF/HA-CNT//XGnP ASC | 0 to 1.7 V | 287.6 F g−1 @ 0.3 A g−1 | 67.9 W h kg−1 @300 W kg−1 | 87.1% after 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles @ 10 A g−1 000 cycles @ 10 A g−1 |
180 |
| 19 | Co-Ni-S/CNTs | 6 M KOH | 0.0 to 0.5 V | 540.6 C g−1 @ 1 A g−1 | — | Co-Ni-S/CNTs//AC | 0.0 to 1.6 V | 178.6 F g−1 @ 1 A g−1 | 63.5 W h kg−1 @ 800 W kg−1 | 83% after 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles @ 10 A g−1 000 cycles @ 10 A g−1 |
181 |
| 20 | Co3O4@MnO2@PPy | 2 M KOH | 0.0 to 1.1 V | 977 F g−1 @ 1 A g−1 | 105% after 400 cycles @ 1 A g−1 | Co3O4@MnO2@PPy//AC ASC | 0.0 to 1.8 V | 102.78 F g−1 @ 1 A g−1 | 166 W h kg−1 @ 3.3 kW kg−1 | 120% after 5000 cycles @ 15 mA cm−2 | 374 |
| 21 | MnO2/Ni-Mn-S | 2 M KOH | 0.0 to 0.6 V | 1575 F g−1 @ 1 A g−1 | — | MnO2/Ni-Mn-S//N-Rgo HSC; PVA-KOH gel | 0.0 to 1.8 V | 67 F g−1 @ 3 mA cm−2 | 31.5 W h kg−1 @ 937.5 W kg−1 | 97.6% after 11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles @ 10 mA cm−2 000 cycles @ 10 mA cm−2 |
375 |
| 22 | NPMCF | 1 M Na2SO4 | −0.2 to 1.2 V | 687 F g−1 | — | CPCF//NPMCF ASC; PVA/Na2SO4 | 0.0 to 2.2 V | 143 F g−1 @ 1 A g−1 | 71.6 W h kg−1 @ 492.7 W kg−1 | — | 376 |
| 23 | CPCF | 6 M KOH | −1.0 to 0.0 V | 227 F g−1 @ 1 A g−1 | 92.68% after 5000 cycles @ 10 A g−1 | CoAl LDH//Gr ASC; 6 M KOH | 0.0 to 1.4 V | 59.86 F g−1 @ 1 A g−1 | 16.29 W h kg−1 @ 700 W kg−1 | 82.84% after 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles @ 4 A g−1 000 cycles @ 4 A g−1 |
210 |
| CoAl LDH | 0.0 to 0.4 V | 983.1 F g−1 @ 1 A g−1 | |||||||||
| 24 | NiCo-LDHs | 3 M KOH | 0.0 to 0.38 V | 1272 C g−1 @ 2 A g−1 | — | NiCo-LDHs//CNTs ASC; 3 M KOH | 0.0 to 1.3 V | 153.8 F g−1 @ 1 A g−1 | 36.1 W h kg−1 @ 649 W kg−1 | 103.9% after 5000 cycles @ 4 A g−1 | 377 |
| 25 | NiCo-LDH/rGO | 2 M KOH | 0.0 to 0.45 V | 1675 F g−1 @ 1 A g−1 | 81.2% after 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles @ 10 A g−1 000 cycles @ 10 A g−1 |
NiCo-LDH/rGO//AC ASC; 2 M KOH | 0.0 to 1.5 V | 159.6 F g−1 @ 0.5 A g−1 | 49.9 W h kg−1 @ 375.2 W kg−1 | 62.2% after 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles @ 5 A g−1 000 cycles @ 5 A g−1 |
212 |
| 26 | CF@NCOSe/Ni2Co1-LDH | 6 M KOH | 0.0 to 0.5 V | 454.17 mA h g−1 @ 1 A g−1 | — | CF@NCOSe/Ni2Co1-LDH//AC HSC; 6 M KOH | 0.0 to 1.6 V | 252.4 F g−1 @ 1 A g−1 | 89.7 W h kg−1 @ 800 W kg−1 | 95.6% after 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles @ 10 A g−1 000 cycles @ 10 A g−1 |
213 |
| 27 | MXene/PANI | 6 M KOH | 0.0 to 1.0 V | 563 F g−1 @ 0.5 A g−1 | 95.15% after 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles @ 5 A g−1 000 cycles @ 5 A g−1 |
MXene/PANI//AC ASC; 7 M KOH | 0.0 to 1.2 V | 262 F g−1 @ 0.5 A g−1 | 22.67 W h kg−1 @ 0.217 kW kg−1 | 90.82% after 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles @ 5 A g−1 000 cycles @ 5 A g−1 |
378 |
| 28 | Ni3S2/PPy | 3 M KOH | 0.0 to 0.5 V | 1484 C g−1 @ 1 A g−1 | 116.7% after 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles @ 10 A g−1 000 cycles @ 10 A g−1 |
Ni3S2/PPy//AC | 0.0 to 1.6 V | 93.6 C g−1 @ 0.5 A g−1 | 74.9 W h kg−1 @ 1356.4 W kg−1 | 91.7% after 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles @ 4 A g−1 000 cycles @ 4 A g−1 |
234 |
| 29 | rGO/PPy | 0.3 M NaClO4 | −0.2 to 0.8 V | 531 F g−1 @ 1 mA cm−2 | 71.7% after 8000 cycles 20 mA cm−2 | rGO/PPy//rGO/PPy | 0.0 to 1.0 V | 110 F g−1 | 15.3 W h kg−1 @1.44 kW kg−1 | 81.7% after 5000 cycles @ 20 mA cm−2 | 236 |
| 30 | — | P&B-AC//Zn HSC | 0.3 to 1.9 V | 169.4 mA h g−1 @ 0.5 A g−1 | 169.4 W h kg−1 @ 500 W kg; 66.7 W h kg−1 @ 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 W kg−1 000 W kg−1 |
95% up to 2000 cycles @ 0.5 A g−1 and 88% up to 30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 @ 10 A g−1 000 @ 10 A g−1 |
137 | ||||
| 31 | — | Zn-aMEGO HSC; 3 M Zn(CF3SO3)2 | 0.0 to 1.9 V | 210 F g−1 @ 0.1 A g−1 | 106.3 W h kg−1 & 31.4 kW kg−1 | 93% after 80![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cyles @ 8 A g−1 000 cyles @ 8 A g−1 |
379 | ||||
| 32 | — | BiFeO3/g-C3N4//AC (synthesized from orange peels) | 0–2.4 V | 330 F g−1 @ 1 A g−1 | 244.8 W h kg−1 @ 1.155 kW kg−1 | 80% after 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles 000 cycles |
130 | ||||
| 33 | — | ACF/GPE/ACF | 0.0 to 1.0 V | 54.4 F g−1 @ 0.1 A g−1 | 4.7 W h kg−1 @ 1.2 kW kg−1 | 91.5% after 5000 cycles @ 0.1 A g−1 | 140 | ||||
| 34 | NiCo-MOF | 2 M NaOH | 0.0 to 0.45 V | 522 C g−1 @ 0.5 A g−1 | 99% after 5000 cycles @ 5 A g−1 | KNiCoPO4//AC; 2 M NaOH | 0.0 to 1.45 V | 76 F g−1 @ 0.5 A g−1 | 22 W h kg−1 @ 363 W kg−1 | 99% after 5000 @ 5 A g−1 | 250 |
| 35 | Ni/Co-MOFs/GNS nanohybrid | 6 M KOH | 0.0 to 0.5 V | 1080 C g−1 @ 1 A g−1 | 92% after 1000 cycles @ 5 A g−1 | NiCo/GNS//ASC; 6 M KOH | 0.0 to 1.7 V | 71.5 F g−1 @ 1 A g−1 | 28.7 W h kg−1 @ 849.9 W kg−1 | — | 251 |
| 36 | Co-V-MOF | 1 M KOH | −0.2 to 0.85 V | 1711.1 F g−1 @ 1 A g−1 | 92.1% after 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles @ 5 A g−1 000 cycles @ 5 A g−1 |
Co-V-MOF//AC; PVA-KOH | 0.0 to 1.2 V | 187.5 F g−1 @ 1 A g−1 | 0.8 kW kg−1 @ 70.65 W h kg−1 | 82.6% after 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles @ 10 A g−1 000 cycles @ 10 A g−1 |
263 |
| 37 | c-MOF@GNF | 2 M KOH | 0.0 to 0.42 V | 1820 F g−1 @ 1 A g−1 | 92.2% after 14![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles @ 20 A g−1 000 cycles @ 20 A g−1 |
c-MOF@GNF//GNF; PVA-KOH | 0.0 to 1.6 V | 146.8 F g−1 @ 1 A g−1 | 52.2 W h kg−1 @ 799.6 W h kg−1 | 90.6% after 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles @ 10 A g−1 000 cycles @ 10 A g−1 |
264 |
| 38 | TFPDQ-COF/GO | 1 M NaCl | −1.2 V to 0.0 V | 429 F g−1 @ 2 A g−1 | — | TFPDQGO//AC; 1 M NaCl | 0.0 to 1.6 V | 118.5 F g−1 @ 1 A g−1 | 59.4 W h kg−1 @ 950 W kg−1 | 80.6% after 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles @ 10 A g−1 000 cycles @ 10 A g−1 |
380 |
| 39 | DAAQ-COFs/Ti3C2Tx | 1 M H2SO4 | −0.5 to 0.2 V | 361 F g−1 @ 1 A g−1 | 89.1% after 30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles @ 5 A g−1 000 cycles @ 5 A g−1 |
CMFs/CCMP; PVA/H2SO4 gel | 0.0 to 1.4 V | 100 F g−1 @ 0.5 A g−1 | 27.5 W h kg−1 @ 350 W kg−1 | 88.9% after 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles @ 5 A g−1 000 cycles @ 5 A g−1 |
381 |
| 40 | VA-Ti3C2Tx@COF-LZU 1 | 1 M H2SO4 | −0.6 to 0.2 V | 787 F g−1 @ 1 A g−1 | — | VA-Ti3C2Tx@COF-LZU 1//RGO FASCs; 1 M H2SO4 | 0.0 to 1.4 V | 398 F g−1 @ 1 A g−1 | 27 W h kg−1 @ 573 W kg−1 | 89% after 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles @ 5 A g−1 000 cycles @ 5 A g−1 |
382 |
4. Design and fabrication techniques
4.1. Electrode preparation methods
 | ||
| Fig. 15 (a) Schematic illustration of CVD method, Reproduced from ref. 399; (b) sol–gel method, Reproduced from ref. 400 and (c) hydrothermal method to synthesize NiCo2O4 nanostructures, Reproduced from ref. 401 with permission from nature portfolio, copyright 2022. | ||
Applications in electrode fabrication:
➢ Graphene and graphene oxide: CVD is extensively used for the synthesis of high-quality graphene and graphene oxide films, which are integral to advanced supercapacitor electrodes due to their high electrical conductivity and large surface area.385–387 Recent advancements in CVD have enabled the production of graphene films with exceptional uniformity and scalability, enhancing their performance in hybrid supercapacitors.388
➢ Metal oxides and nitrides: CVD is also employed to deposit metal oxides (such as MnO2) and nitrides (such as TiN), which are used in supercapacitor electrodes due to their high capacitance and electrochemical stability. The ability to control the stoichiometry and morphology of these materials via CVD has led to significant improvements in the performance and longevity of supercapacitors.389–391
➢ Carbon nanotubes: the deposition of CNTs using CVD has become a standard method due to their high mechanical strength, electrical conductivity, and large surface area. CVD allows for the growth of vertically aligned CNT arrays and carbon nanotubes/graphene hybrids, which can be directly integrated into supercapacitor electrodes to enhance their electrochemical performance392–395
Recent developments in CVD techniques have focused on improving the quality and functionality of deposited films. Innovations such as low-temperature CVD and plasma-enhanced CVD have expanded the range of materials that can be effectively deposited and integrated into supercapacitor electrodes.396,397 Moreover, the use of novel precursor materials and the optimization of deposition parameters have led to significant improvements in the performance characteristics of electrodes fabricated using CVD.398 So the Chemical Vapor Deposition (CVD) has emerged as a critical technique for fabricating high-performance electrode materials in hybrid supercapacitors, owing to its precision, scalability, and ability to produce uniform, high-purity films. Its application in synthesizing graphene, metal oxides, nitrides, and carbon nanotubes has significantly enhanced the electrochemical performance and stability of supercapacitor electrodes. Recent advancements, including low-temperature and plasma-enhanced CVD, continue to broaden its applicability and improve material functionality for next-generation energy storage systems.
➢ Start with a solution including pure chemical precursors;
➢ The process of transforming the solution into a sol, which is then followed by a gel through hydrolysis and polycondensation;
➢ The sol forms into desired shapes such as bulk, film, and fiber during the gelation process;
➢ The gel undergoes thermal treatment to transform into a solid material at low temperatures.
However, it is crucial to recognize the presence of many variances. The “sol–gel method” is the procedure of solidifying a substance by treating a formed compact of gel particles made from a solution or sol with heat.404,405
Principles of the sol–gel method:
➢ Preparation of sol: the sol–gel process begins with the preparation of a sol, which is a stable dispersion of colloidal particles in a solvent. Typically, metal alkoxides or metal chlorides are used as precursors, which undergo hydrolysis and polycondensation reactions to form a network of interconnected particles in the sol.406 The choice of precursors and solvents significantly influences the properties of the final material.
➢ Gelation: as the sol evolves, it forms a gel network through polymerization reactions. This gel network provides a three-dimensional structure that can be further processed to enhance its electrochemical properties.407,408 The gel is typically allowed to age, during which it strengthens and becomes more interconnected.
➢ Drying and heat treatment: the gel is then dried to remove solvents, which may lead to the formation of a xerogel or an aerogel depending on the drying conditions. Subsequent heat treatment (calcination) is used to remove organic residues and achieve the desired crystalline phase and porosity.409,410
The sol–gel method is extensively employed for synthesizing metal oxides, such as MnO2, Co3O4, and Fe2O3, which are used as active materials in supercapacitor electrodes due to their high capacitance and stability.411–413 The sol–gel process allows for precise control over the stoichiometry and morphology of these metal oxides, enhancing their performance. Sol–gel-derived carbon materials, including carbon xerogels and aerogels, are also used as electrode materials. These materials offer high surface area and excellent conductivity, making them suitable for supercapacitors.414,415 The sol–gel method enables the creation of porous carbon structures with tunable properties. The sol–gel technique is useful for fabricating composite electrodes by incorporating active metal oxides or conducting polymers into a carbon matrix. This approach enhances the overall performance of hybrid supercapacitors by combining the advantages of different materials.416,417 Recent advancements in sol–gel processing have focused on optimizing the synthesis parameters and developing new precursor materials to enhance the properties of supercapacitor electrodes. Innovations such as one-pot sol–gel processes and the use of functionalized precursors have led to improved performance and broader applicability.418,419 Additionally, the integration of nanostructured materials into sol–gel matrices has been shown to significantly enhance the electrochemical performance of supercapacitor electrodes.420 So the sol–gel method offers a versatile and precise approach for synthesizing advanced electrode materials for supercapacitors, enabling control over composition, porosity, and nanostructure. Its ability to produce metal oxides and porous carbon materials with high surface area and conductivity enhances capacitance and stability. Recent innovations, including one-pot synthesis and nanostructure integration, further improve performance, making the sol–gel method a promising route for developing next-generation energy storage devices.
Principles of hydrothermal synthesis:
➢ Reaction conditions: hydrothermal synthesis is performed in a sealed vessel, often referred to as an autoclave, where the precursor materials are dissolved in a solvent (typically water) and subjected to high temperatures and pressures. The high-pressure environment facilitates the dissolution of precursors and promotes the nucleation and growth of the desired material.422,423 The temperature and pressure conditions can be adjusted to control the size, shape, and crystallinity of the resulting product.
➢ Formation of nanostructures: during the hydrothermal process, nanoparticles or nanostructures are formed through nucleation and growth mechanisms. The solvent acts as both a medium and a reactant, which allows for the uniform distribution of precursor materials and promotes the formation of well-defined nanostructures.424,425 The controlled environment of the autoclave helps in achieving the desired phase and morphology.
➢ Post-synthesis processing: after synthesis, the hydrothermal products often require washing, filtering, and drying to remove any residual reagents or by-products.426,427 Further treatments, such as annealing, can be employed to enhance the material properties and achieve the desired crystalline phase.
Hydrothermal synthesis is extensively used to produce metal oxides such as NiO, MnO2, and titanium dioxide (TiO2) for supercapacitor electrodes. These metal oxides are valued for their high specific capacitance and stability. The method allows for precise control over the particle size and morphology, which influences the electrochemical performance of the electrodes.428–430 This technique can also be used to synthesize carbon-based materials, including carbon spheres and reduced-graphene oxide (rGO), which are incorporated into supercapacitor electrodes to enhance conductivity and surface area.431,432 Hydrothermal synthesis of carbon materials often involves the use of organic precursors or templates to guide the formation of nanostructures. Hydrothermal synthesis enables the creation of composite materials by combining metal oxides or carbon materials with other active components. These composites often exhibit synergistic effects, leading to improved electrochemical performance.433,434 For example, composites of metal oxides with carbon nanotubes or graphene have been shown to enhance charge storage and conductivity. Recent research in hydrothermal synthesis has focused on optimizing reaction conditions to produce materials with enhanced properties. Innovations include the development of hybrid hydrothermal processes that integrate other synthesis methods, such as sol–gel, to create advanced electrode materials.435–437 Additionally, the exploration of new precursors and solvents has led to the discovery of novel materials with superior performance.
4.2. Cell assembly techniques
Principles of coin cell assembly:
➢ Cell design and components: a typical coin cell consists of several key components (Fig. 16a): two electrodes (positive and negative), a separator, an electrolyte, and a casing. The electrodes are usually composed of active materials coated onto current collectors, often made from metals like aluminum or copper. The separator, typically a porous membrane, prevents short-circuiting between electrodes while allowing ionic conductivity. The electrolyte, which can be aqueous, organic, or ionic liquid-based, facilitates charge transport.440,441
➢ Assembly process: the assembly of a coin cell involves placing the electrode materials onto current collectors, followed by stacking them with the separator and electrolyte in a coin cell casing. The cell components are compressed and sealed within the casing to ensure mechanical stability and to prevent leakage of the electrolyte.442,443 Proper alignment and contact between the electrodes and current collectors are crucial for optimal performance.
➢ Performance testing: once assembled, coin cells are subjected to electrochemical testing to evaluate their performance characteristics, including capacitance, energy density, power density, and cycle stability. Techniques such as cyclic voltammetry (CV), galvanostatic charge–discharge (GCD), and electrochemical impedance spectroscopy (EIS) are commonly used.444–446
Recent developments and advances of coin cell:
➢ Recent advancements have focused on improving the miniaturization and standardization of coin cells to facilitate more accurate and consistent testing of supercapacitor materials. This includes the development of standardized test protocols and cell designs to ensure reproducibility across different research laboratories.447,448 Coin cells are increasingly used for testing advanced materials, such as nanomaterials, composites, and novel electrolytes. The compact size of coin cells allows for high-throughput screening of new materials, helping to accelerate the development of high-performance supercapacitors.23,449 There has been significant research into optimizing electrolytes for use in coin cells. This includes the development of new electrolyte formulations that enhance the performance of supercapacitors, such as ionic liquids and gel electrolytes. These advancements aim to improve the stability, conductivity, and overall efficiency of the devices.41,450–453
Automation and robotic systems are being integrated into the coin cell assembly process to increase precision and efficiency. Automated systems can handle repetitive tasks, reduce human error, and improve the consistency of assembled cells.454,455 Despite its advantages, coin cell assembly also faces challenges, such as the need for precise control of cell assembly conditions and the potential for inconsistencies in the results due to manual assembly processes. Future research is likely to focus on addressing these challenges by developing more advanced assembly techniques and improving the reproducibility and reliability of coin cells.15
Principles of pouch cell assembly:
➢ Cell structure and components: pouch cells (Fig. 16b) consist of several layers: two electrodes (positive and negative), a separator, and an electrolyte. These components are assembled within a flexible pouch-like casing made from polymer films, such as aluminum or polypropylene. The pouch design allows for the encapsulation of the cell components, providing protection and flexibility.456,457 The electrodes, typically coated on metal foils, are stacked with a separator in between and infused with the electrolyte.
➢ Assembly process: the assembly of pouch cells involves the following steps:
○ Electrode preparation: electrodes are prepared by coating active materials onto current collectors and drying them.
○ Stacking and encapsulation: the electrodes and separator are stacked in the desired configuration and then placed into the pouch. The pouch is sealed, often through heat sealing or adhesive bonding, to ensure the cell's integrity and prevent leakage.458,459
○ Electrolyte filling: the electrolyte is introduced into the pouch either before or after sealing, depending on the specific assembly process.460 This step is crucial for achieving optimal ionic conductivity and electrochemical performance.
➢ Performance testing: pouch cells are tested for various electrochemical properties, including capacitance, energy density, power density, and cycle stability.461,462 Techniques such as cyclic voltammetry, galvanostatic charge–discharge, and electrochemical impedance spectroscopy are used to evaluate their performance under realistic conditions.
Recent developments and advances of pouch cell:
Flexible and lightweight designs: recent advancements in pouch cell technology focus on developing flexible and lightweight designs, which are particularly useful for wearable electronics and portable energy storage applications. Innovations in pouch materials and sealing techniques have enhanced the flexibility and mechanical properties of these cells.463,464High energy density: efforts are being made to increase the energy density of pouch cells by optimizing electrode materials and electrolytes. This includes the development of high-capacity electrodes and advanced electrolytes that improve the overall energy storage capability of the cells.465Enhanced safety and durability: improvements in pouch cell safety and durability are critical for practical applications. Research has focused on enhancing the mechanical strength of the pouch materials and improving the stability of the electrolyte to prevent issues such as leakage and degradation.461,466Scalability and production techniques: advances in production techniques aim to enhance the scalability of pouch cell manufacturing. Automation and high-throughput methods are being integrated to increase the efficiency and consistency of pouch cell production.453,467 Pouch cell technology faces challenges such as ensuring uniformity in large-scale production and addressing potential issues related to mechanical stability and electrolyte leakage. Future research is likely to focus on overcoming these challenges by developing more robust materials, improving manufacturing processes, and enhancing the overall performance and reliability of pouch cells.468,469 Ultimate the recent advancements in pouch cell technology have significantly improved their flexibility, energy density, safety, and scalability, making them ideal for modern portable and wearable applications. However, challenges such as mechanical stability and large-scale production uniformity persist. Future research will likely emphasize robust material development and optimized manufacturing techniques to further enhance pouch cell performance and reliability.
4.3. Integration with other devices
The integration of supercapacitors with other devices is a critical aspect of advancing their practical applications and enhancing their performance in real-world scenarios. This section explores the various approaches and technologies used to integrate supercapacitors with other electronic and energy systems, focusing on the challenges, methodologies, and recent advancements.➢ Hybrid energy storage systems: supercapacitors are often combined with batteries to create hybrid energy storage systems that leverage the strengths of both technologies. Batteries provide high energy density, while supercapacitors offer high power density and fast charge/discharge rates. This hybrid approach is used in applications like electric vehicles and renewable energy systems, where it helps balance the energy supply and demand effectively.7,472
➢ Power management systems: in power management systems, supercapacitors are integrated to enhance the stability and efficiency of energy distribution. They act as buffer storage to smooth out fluctuations in power supply and demand. For instance, in grid energy storage applications, supercapacitors can support the grid during peak loads and improve the reliability of renewable energy sources.473,474
➢ Flexible and wearable electronics: integration with flexible and wearable electronics is an emerging trend where supercapacitors are incorporated into flexible substrates or textiles. This integration allows for the development of flexible energy storage solutions that can be seamlessly integrated into clothing or other wearable items, providing power for sensors and other electronic components.475–477
➢ Thermal management: supercapacitors and integrated devices must manage heat dissipation effectively to prevent overheating and ensure long-term reliability. Thermal management solutions, such as heat sinks and thermal interface materials, are employed to manage the temperature and maintain optimal operating conditions.479,480
➢ Design considerations: the design of integrated systems must account for factors such as space constraints, mechanical stress, and environmental conditions. Advanced design techniques, including computer-aided design (CAD) and simulation, are used to optimize the integration process and ensure the system's performance and durability.481
➢ Experimental complexity and data interpretation: while in situ and operando approaches are critical for real-time monitoring of electrochemical phenomena, their application is often hampered by complicated experimental setups and difficulties in understanding dynamic, multidimensional data.482
➢ Material deterioration and analytical limitations: material deterioration during operation complicates long-term stability studies, as does the low resolution and sensitivity of present analytical methods.483
➢ Standardization and accessibility: the absence of defined methods, as well as the high cost of modern characterization techniques, limit their widespread use in research and practical applications.482
To address these issues, enhanced experimental settings, the incorporation of multi-modal analytical methodologies, and breakthroughs in high-resolution, low-damage instruments are required. Furthermore, the creation of cost-effective and standardized procedures is projected to broaden access to these instruments, improving performance assessment and accelerating the development of integrated, high-efficiency supercapacitor systems.
➢ Miniaturization and flexibility: advances in miniaturization and flexible electronics have enabled the development of compact and adaptable supercapacitors that can be easily integrated into various devices. This includes the use of nano-engineered materials and flexible substrates to create high-performance supercapacitors for wearable and portable applications.25,485
➢ Smart integration technologies: the integration of smart technologies, such as wireless communication and sensing, with supercapacitors is an area of active research. These technologies enable the development of intelligent energy storage systems that can monitor and adapt to changing conditions in real-time.455,478,486
5. Performance metrics and testing
5.1. Capacitance and energy density
Understanding the performance metrics of hybrid supercapacitors is crucial for evaluating their suitability for various applications. Among these metrics, capacitance and energy density are key parameters that provide insights into the storage capacity and energy efficiency of the devices.Capacitance:
Capacitance (C) is a fundamental property of supercapacitors, representing their ability to store electric charge.487 It is defined by the equation:
| C = Q/V | (8) |
The specific capacitance can be calculated from the integral area of the CV curve using the following equation
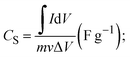 | (9) |
From, GCD curves with linear charge–discharge curves (EDLCs)
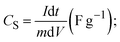 | (10) |
From, GCD curves with non-linear charge–discharge curves (pseudocapacitor/battery type supercapacitor electrodes)
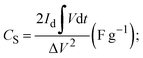 | (11) |
 is the integral area under the discharge curve, ΔV is the potential window, respectively.489
is the integral area under the discharge curve, ΔV is the potential window, respectively.489
Recent advancements in electrode materials have significantly enhanced the capacitance of hybrid supercapacitors. For instance, incorporating graphene and carbon nanotubes has increased the surface area and improved ion accessibility, leading to higher capacitance values.490 Additionally, the development of composite materials, such as carbon–metal oxide hybrids, has further boosted the capacitance by combining the high surface area of carbon with the pseudocapacitive properties of metal oxides.491
Energy Density:
Energy density (E) is another critical performance metric, indicating the energy stored per unit volume or mass. It is given by:
| E = 1/2CS(ΔV)2 (W h kg−1) | (12) |
Advancements in materials science have led to significant improvements in the energy density of hybrid supercapacitors. For example, the use of MOFs and COFs as electrode materials has been reported to enhance energy density due to their high surface area and tunable pore structures.493,494 These materials facilitate efficient ion transport and increase the overall storage capacity. Moreover, the optimization of electrolytes has also contributed to higher energy densities. Ionic liquids and solid-state electrolytes offer wider electrochemical windows, enabling higher operating voltages and thus increasing the energy density.495 Hybrid supercapacitors utilizing these advanced electrolytes can achieve energy densities comparable to those of traditional batteries while maintaining superior power density and cycle life.
Testing and measurement:
Accurate measurement of capacitance and energy density is essential for characterizing the performance of hybrid supercapacitors. In general, the electrochemical capacitor and battery performance of active electrodes are evaluated using electrochemical techniques such as cyclic voltammetry, galvanostatic charge–discharge tests, and electrochemical impedance spectroscopy. Fig. 17 displays typical CV and GCD profiles for EDLC, pseudocapacitor, and battery materials. These three methodologies aid in deciphering kinetic responses and determining the charge-storage process in active electrode materials.
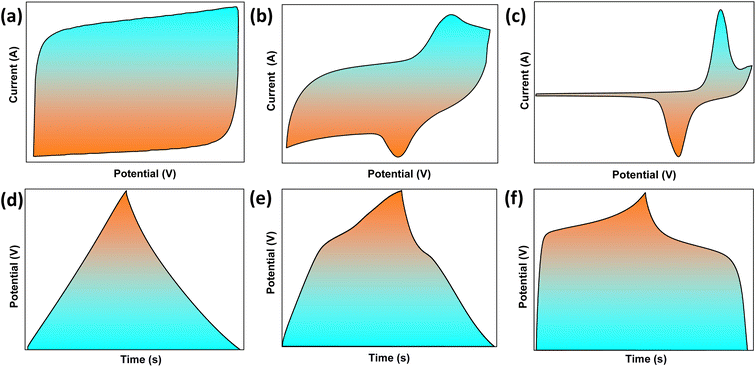 | ||
| Fig. 17 Characteristic metrics of CV and GCD of different electrode types (a, and d) EDLC, (b, and e) pseudocapacitive, and (c, and f) battery type. | ||
The following formula called as power law, which provides the power law dependency of the current response (I) on the scan rate (v), make estimating electrode kinetics easy to understand: I = avb; here, “a” is constant, parameter “b” with a value of 0.5 indicates a diffusion controlled process in which the current response is proportional to the square root of the scan rate. When “b” = 1, it suggests a surface-controlled process with a linear response to the scan rate (Iαv). A “b” value of 0.5 to 1 implies a hybrid energy storage system that switches from battery-like to capacitive behavior.496,497
➢ Cyclic voltammetry: CV measures the current response of the supercapacitor to a linearly varying voltage. The shape of the CV curves provides information about the capacitive behavior and the presence of redox reactions.498
➢ Galvanostatic charge–discharge: GCD tests involve charging and discharging the supercapacitor at a constant current. The resulting voltage profiles are used to calculate capacitance, energy density, rate capability and cycling stability. GCD is particularly useful for evaluating the rate capability and cycle stability of the supercapacitor.496
➢ Electrochemical impedance spectroscopy: EIS analyzes the frequency response of the supercapacitor to a small alternating current signal. It provides insights into the resistance and capacitance of the device, helping to identify factors limiting performance.499
Recent studies have highlighted the use of graphene and its derivatives to enhance capacitance and energy density. For instance, a study demonstrated that graphene-MnO2 composites exhibit high specific capacitance and energy density due to synergistic effects between the components.500 The development of nanostructured materials, such as nanowires and nanotubes, has been shown to improve ion transport and increase active surface area, resulting in higher capacitance and energy density.501 Research on advanced electrolytes, including ionic liquids and gel polymers, has led to significant enhancements in energy density. These electrolytes provide wider electrochemical windows and better thermal stability, making them ideal for high-performance supercapacitors.502
So the capacitance and energy density are key indicators of the energy storage capabilities of electroactive polymers (EAPs), directly influencing their suitability for applications such as flexible supercapacitors and wearable energy devices. Enhancing these parameters requires careful material engineering, including the integration of high surface area nanomaterials, improved polymer conductivity, and the development of optimized composite architectures. Recent advancements have demonstrated that synergistic combinations of conductive fillers with EAP matrices can significantly increase charge storage capacity while maintaining flexibility and mechanical stability. These improvements in capacitance and energy density pave the way for high-performance, lightweight, and adaptable energy storage systems tailored for next-generation electronic applications.
5.2. Power density
Power density is a crucial performance metric for hybrid supercapacitors, reflecting their ability to deliver energy quickly. It is defined as the amount of power per unit volume (or mass) and is given by the equation:| P = E/Δt (W kg−1) | (13) |
Mechanisms influencing power density:
➢ Electrode materials: the choice of electrode materials significantly impacts the power density of hybrid supercapacitors. Carbon-based materials, such as activated carbon, graphene, and carbon nanotubes, offer high surface area and excellent electrical conductivity, enhancing power density.36 Metal oxides and hydroxides, such as MnO2, NiO, and Co(OH)2, provide high pseudocapacitance, which contributes to higher power output.504
➢ Composite materials: the use of composite materials, such as carbon–metal oxide and carbon–polymer composites, can further enhance power density. These materials combine the high surface area and conductivity of carbon with the pseudocapacitive properties of metal oxides or the flexibility and processability of polymers.505 For example, graphene–polyaniline composites have demonstrated high power densities due to their synergistic effects.506
➢ Electrolytes: the type of electrolyte used also plays a significant role in determining power density. Aqueous electrolytes, such as sulfuric acid and potassium hydroxide, offer high ionic conductivity, resulting in low internal resistance and high power density.507,508 However, their voltage windows are limited, which can restrict energy density. On the other hand, organic electrolytes and ionic liquids provide wider voltage windows, leading to higher energy and power densities.509
➢ Cell configuration: the design and configuration of the cell also affect power density. For instance, the use of thin electrodes can reduce ion diffusion paths, thereby enhancing power density.510 Additionally, advanced fabrication techniques, such as 3D printing and laser scribing, can create well-organized electrode structures that improve ion transport and power output.511
Recent studies have focused on enhancing power density through the use of graphene and its derivatives. A study demonstrated that graphene-MnO2 hybrids exhibit high power densities due to their excellent conductivity and high surface area.500 Similarly, graphene aerogels have been used to develop supercapacitors with high power density and mechanical flexibility.512 The use of nanostructured electrodes, such as nanowires and nanotubes, has been shown to improve power density by providing efficient pathways for ion transport. For example, CNTs decorated with metal oxides have demonstrated enhanced power densities due to their high surface area and fast electron transport capabilities.513 Hybrid electrolytes, combining the benefits of aqueous and organic electrolytes, have been developed to achieve high power density. These electrolytes offer high ionic conductivity and wide voltage windows, enabling high power and energy densities.514
Performance testing:
Accurate measurement of power density involves testing techniques such as cyclic voltammetry, galvanostatic charge–discharge, and electrochemical impedance spectroscopy.
➢ Cyclic voltammetry: CV measures the current response to a linearly varying voltage, providing information on the capacitive behavior and reaction kinetics. High scan rates in CV tests are used to evaluate power density, as they simulate high discharge rates.515
➢ Galvanostatic charge–discharge: GCD tests involve charging and discharging the supercapacitor at a constant current. The resulting voltage profiles are used to calculate power density. Short discharge times in GCD tests indicate high power density.516
➢ Electrochemical impedance spectroscopy: EIS analyzes the frequency response of the supercapacitor, providing insights into resistance and capacitance. Low equivalent series resistance (ESR) values obtained from EIS indicate high power density.517,518
So the power density is a vital parameter that determines the ability of electroactive polymers (EAPs) to deliver energy rapidly, which is especially important for applications requiring quick response times such as sensors, actuators, and energy harvesting devices. Recent research efforts have focused on enhancing the intrinsic conductivity and optimizing the microstructure of EAPs to facilitate faster ion and electron transport. Incorporating conductive fillers, engineering nano-architectures, and improving interfacial charge transfer have proven effective in boosting power density. These advancements position EAP-based systems as promising candidates for next-generation devices where high power output and rapid energy delivery are essential for performance and efficiency.
5.3. Cycling stability
Cycling stability is a critical performance metric for hybrid supercapacitors, reflecting their ability to maintain capacity and efficiency over numerous charge–discharge cycles. It is a vital factor for practical applications, as it determines the longevity and reliability of the device. High cycling stability ensures that the supercapacitor can be used repeatedly without significant degradation in performance.519Mechanisms influencing cycling stability:
➢ Electrode material degradation: the stability of electrode materials under prolonged cycling influences the overall cycling stability of hybrid supercapacitors. Carbon-based materials, such as graphene and carbon nanotubes, exhibit excellent cycling stability due to their robust structure and high conductivity.520 While providing high capacitance, metal oxides and conducting polymers often suffer from structural degradation and dissolution during cycling, which limits their stability.36
➢ Electrolyte stability: the type and stability of the electrolyte also significantly impact cycling stability. Aqueous electrolytes, though highly conductive, can lead to corrosion and oxidation of the electrodes. On the other hand, organic electrolytes and ionic liquids, with their wide electrochemical windows and low volatility, offer better stability, though they may have issues with ionic conductivity and cost.521 Recent advances in hybrid electrolytes aim to combine the benefits of both to enhance cycling stability.522
➢ Structural integrity: maintaining the structural integrity of electrodes during repeated charge–discharge cycles is crucial. The use of nanostructured materials and composites can help improve mechanical stability. For instance, incorporating flexible polymers into carbon-based materials or using 3D architectures can mitigate the stress and strain during cycling, thus enhancing stability.523,524
A study demonstrated that graphene aerogels, due to their 3D interconnected network, show minimal degradation over 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles, maintaining over 95% of their initial capacitance.525 Similarly, CNT-based electrodes have been shown to retain high stability due to their excellent mechanical properties and conductivity.526 Composite materials, such as MnO2/graphene and NiO/CNT, have exhibited improved cycling stability compared to pure metal oxides. The conductive carbon matrix supports the metal oxide particles, preventing agglomeration and dissolution during cycling.527 Conducting polymers like polyaniline (PANI) and polypyrrole have been integrated with carbon materials to enhance cycling stability. These composites leverage the high capacitance of conducting polymers while the carbon matrix provides structural support and conductivity.528 The development of hybrid electrolytes that combine the high ionic conductivity of aqueous solutions with the wide voltage window of organic solvents has shown promise in enhancing cycling stability. These electrolytes reduce electrode degradation and improve overall device longevity.529
000 cycles, maintaining over 95% of their initial capacitance.525 Similarly, CNT-based electrodes have been shown to retain high stability due to their excellent mechanical properties and conductivity.526 Composite materials, such as MnO2/graphene and NiO/CNT, have exhibited improved cycling stability compared to pure metal oxides. The conductive carbon matrix supports the metal oxide particles, preventing agglomeration and dissolution during cycling.527 Conducting polymers like polyaniline (PANI) and polypyrrole have been integrated with carbon materials to enhance cycling stability. These composites leverage the high capacitance of conducting polymers while the carbon matrix provides structural support and conductivity.528 The development of hybrid electrolytes that combine the high ionic conductivity of aqueous solutions with the wide voltage window of organic solvents has shown promise in enhancing cycling stability. These electrolytes reduce electrode degradation and improve overall device longevity.529
Performance testing:
➢ Galvanostatic charge–discharge: cycling stability is often evaluated using GCD tests, where the supercapacitor is subjected to continuous charge and discharge cycles at a constant current. The retention of capacitance and efficiency over thousands of cycles is recorded to assess stability.530
➢ Electrochemical impedance spectroscopy: EIS is used to monitor changes in internal resistance and capacitance during cycling. An increase in resistance or a decrease in capacitance over cycles indicates degradation of the electrode materials or the electrolyte.531
➢ Cyclic voltammetry: CV measurements at different stages of cycling can reveal changes in electrochemical behavior. A consistent CV profile over multiple cycles indicates good cycling stability.532
Ultimate the cycling stability is a critical performance metric for electroactive polymers (EAPs) used in energy storage and sensing applications, as it reflects the material's ability to maintain consistent functionality over prolonged operational cycles. High cycling stability ensures reliable and durable operation, which is essential for practical deployment in flexible electronics, biomedical devices, and self-powered systems. Recent advancements in polymer design, including the incorporation of robust backbones, crosslinking strategies, and hybridization with nanomaterials, have significantly improved the structural integrity and electrochemical resilience of EAPs under repeated use. These innovations contribute to reduced degradation and better retention of electrochemical performance, making EAPs more viable for long-term, real-world applications.
5.4. Rate capability
Rate capability is a crucial performance metric for hybrid supercapacitors, determining their ability to deliver energy at high power rates. It reflects the supercapacitor's efficiency in maintaining capacitance and energy storage when subjected to varying charge–discharge rates. High rate capability is essential for applications requiring rapid energy delivery, such as in electric vehicles and power grids.Factors influencing rate capability:
➢ Electrode material properties: the intrinsic properties of electrode materials significantly affect rate capability. Materials with high electrical conductivity and large surface area, such as graphene, carbon nanotubes, and certain metal oxides, are preferred due to their efficient charge transport and storage capabilities.112 For example, a study highlighted that graphene-based supercapacitors maintained high capacitance even at high current densities due to their excellent conductivity and surface area.533
➢ Electrode architecture: the architecture of the electrodes, including their porosity and structural design, also influences rate capability. Hierarchical and porous structures facilitate rapid ion transport and reduce diffusion distances, enhancing rate performance. For instance, electrodes with a mesoporous structure showed improved rate capability due to better electrolyte penetration and ion accessibility.534
➢ Electrolyte properties: the choice of electrolyte impacts the rate capability by influencing ion mobility and conductivity. Aqueous electrolytes provide high ionic conductivity, supporting high rate performance, but may have limitations in voltage range. On the other hand, organic electrolytes and ionic liquids, with their wider electrochemical windows, can offer better rate capability at higher voltages despite lower ionic conductivity.297 Hybrid electrolytes combining these advantages are being developed to optimize rate capability.22
Recent studies have shown that hybrid supercapacitors using graphene and CNTs exhibit superior rate capability due to their high electrical conductivity and large surface area. For example, a graphene/CNT composite electrode maintained over 90% of its capacitance at high current densities, demonstrating excellent rate performance.535 Metal oxide-based electrodes, such as MnO2 and RuO2, have been extensively studied for their high capacitance and good rate capability. The integration of metal oxides with conductive materials like graphene enhances the rate capability by improving electron transport pathways. A MnO2/graphene composite showed significant improvement in rate performance compared to pure MnO2.437 Conducting polymers such as polyaniline (PANI) and polypyrrole have been used in hybrid supercapacitors for their high pseudocapacitance. When combined with carbon materials, these polymers exhibit enhanced rate capability due to improved conductivity and structural stability. A study demonstrated that a PANI/graphene composite maintained high capacitance retention at various current densities.536 The development of hybrid electrolytes, which combine the high ionic conductivity of aqueous electrolytes with the wide voltage range of organic electrolytes, has shown promise in improving rate capability. These electrolytes enable rapid ion transport while supporting high voltage operation, enhancing the overall rate performance of hybrid supercapacitors.537
Performance testing:
1. Galvanostatic charge–discharge: rate capability is often evaluated using GCD tests at different current densities. The ability of the supercapacitor to retain capacitance at higher current densities indicates good rate performance. For instance, a hybrid supercapacitor tested at varying current densities retained over 80% of its initial capacitance, showcasing its excellent rate capability.538
2. Cyclic voltammetry: CV measurements at different scan rates are used to assess rate capability. A consistent CV profile at higher scan rates indicates efficient charge transfer and high rate performance. A study on a hybrid electrode showed minimal distortion in the CV curves at higher scan rates, reflecting good rate capability.539
3. Electrochemical impedance spectroscopy: EIS is employed to analyze the charge transfer resistance and ion diffusion behavior. Lower charge transfer resistance and faster ion diffusion correlate with better rate capability. An EIS study on a hybrid supercapacitor demonstrated low resistance and rapid ion transport, supporting its high rate performance.540
So the rate capability of electroactive polymers (EAPs) plays a pivotal role in determining their effectiveness in high-performance energy storage and conversion devices. Superior rate capability ensures that these materials can sustain rapid charge and discharge cycles without significant loss in capacity, making them highly suitable for applications requiring fast energy delivery, such as wearable electronics, biomedical devices, and flexible sensors. Advances in material design, including molecular engineering, nanostructuring, and composite formation, have significantly improved ion and electron transport pathways within EAPs, thereby enhancing their rate performance. Continued research focusing on optimizing structural features and electrode–electrolyte interfaces will be essential to further boost the rate capabilities of EAPs for real-world applications.
6. Applications of hybrid supercapacitors
Hybrid supercapacitors have evolved as useful energy storage device owing to their high power density, quick charge–discharge cycles, and extended lifetime. Fig. 18 depicts its applicability in crucial sectors such as renewable energy integration, electric mobility, industrial robotics, and smart grid systems. Supercapacitors are opening the way for advances in energy management by bridging the gap between conventional capacitors and batteries, therefore facilitating the shift to sustainable and energy-efficient technology. | ||
| Fig. 18 Applications of supercapacitors in various sectors, including renewable energy systems, electric vehicles, industrial automation, and portable electronics. | ||
6.1. Consumer electronics
Hybrid supercapacitors have emerged as pivotal energy storage devices in consumer electronics. They are driven by their superior electrochemical performance, including high power density, rapid charge–discharge capability, and long cycle life. These attributes address the increasing demand for efficient and reliable power sources in various consumer electronics, such as smartphones, laptops, wearable devices, and portable multimedia players.Advantages in consumer electronics:
➢ High power density: HSCs provide high power density, making them ideal for devices requiring quick energy bursts. This characteristic is particularly beneficial for applications such as camera flashes, LED flashes in smartphones, and other high-current demanding functions in portable electronics.23 For instance, a hybrid supercapacitor integrated with lithium-ion batteries (LIBs) can significantly enhance the performance of devices by providing the necessary power surges without compromising the overall energy capacity.541
➢ Rapid charge–discharge capability: the fast charge–discharge capability of HSCs is crucial for consumer electronics that require frequent and quick charging cycles. This feature enhances user convenience by reducing downtime and ensuring that devices are ready for use in a shorter time. Recent studies have demonstrated that HSCs with graphene-based electrodes exhibit excellent rate performance, achieving near-full charge in minutes, which is significantly faster than conventional batteries.25
➢ Long cycle life: HSCs exhibit long cycle life, which is essential for consumer electronics subjected to repeated charging and discharging cycles. This durability translates to extended device lifetimes and reduced maintenance costs. For example, Sun et al. designed Cr-ion HSCs with porous graphitized carbon fabric, Cr metal and LiCl–CrCl3 as cathode, anode and electrolyte. CHSC delivered 1.47 mW h cm−2 energy density with 9.95 mW cm−2 and 95.4% capacity retention after 30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles.542
000 cycles.542
Notable applications:
Smartphones and tablets: the integration of HSCs in smartphones and tablets can address the increasing power demands of advanced features such as high-resolution displays, multi-core processors, and 5G connectivity. HSCs can provide the necessary power boosts for these features without significantly increasing device size or weight. A study on hybrid Li-ion capacitors (LICs) showed enhanced performance in powering smartphones, providing both high energy and power densities.48
Wearables: wearable electronics, including smartwatches, fitness trackers, and smart glasses, benefit greatly from the compact size and high power density of HSCs. These devices require energy storage solutions that are lightweight and capable of delivering power efficiently. Research on flexible hybrid supercapacitors, utilizing materials such as graphene and MXenes, has demonstrated their suitability for wearable applications, offering both flexibility and high electrochemical performance.543–545
Portable media devices and laptops: devices like portable media players and digital cameras can leverage the fast charging capabilities of HSCs to enhance user experience. For example, a graphene-based supercapacitor was used to power a portable media player, achieving rapid charging and extended playback times compared to traditional batteries.546 The use of HSCs in laptops and ultrabooks can lead to significant improvements in battery life and performance, particularly in terms of fast charging and efficient energy management. HSCs can supplement traditional LIBs, providing quick power bursts for intensive tasks and reducing overall charging time. A hybrid capacitor-battery system in laptops has been shown to deliver enhanced power management, supporting both high performance and long battery life.547
The integration of HSCs with existing battery technologies and electronic components requires careful consideration of compatibility and interface issues. Ensuring seamless operation and optimal performance in diverse consumer electronics is a key area of ongoing research.548,549 Continuous advancements in electrode and electrolyte materials are crucial for further enhancing the performance of HSCs in consumer electronics. Innovations such as 2D materials, metal–organic frameworks, and advanced polymer electrolytes hold promise for achieving higher energy and power densities, along with improved mechanical properties.550,551 The cost-effectiveness and scalability of HSCs are vital for their widespread adoption in consumer electronics. Research efforts are focused on developing low-cost, scalable manufacturing processes and materials to make HSCs commercially viable for large-scale applications.552 Electroactive polymers (EAPs) present transformative opportunities for next-generation consumer electronics by enabling the development of flexible, lightweight, and self-powered devices. Their unique ability to transduce mechanical stimuli into electrical signals makes them ideal for applications such as wearable electronics, touch-sensitive interfaces, and portable energy harvesters. These materials support the miniaturization and multifunctionality demanded by modern gadgets while enhancing user interaction through responsive and adaptive features. To fully leverage their potential, ongoing efforts are needed to optimize their electrical performance, mechanical stability, and compatibility with large-scale fabrication processes tailored to consumer markets.
6.2. Electric vehicles
Hybrid supercapacitors are rapidly gaining traction in the electric vehicle (EV) sector due to their exceptional energy and power density, complementing modern EVs' energy storage and delivery requirements. Their unique characteristics make them highly suitable for applications that demand high power delivery and extended cycle life, addressing key performance metrics critical to EV operations.553Advantages of HSCs in electric vehicles:
➢ Enhanced power delivery: HSCs offer high power density, which is crucial for EVs requiring rapid acceleration and high performance. Delivering quick bursts of power complements traditional lithium-ion batteries (LIBs), which are generally optimized for energy storage rather than high power output. Recent studies have demonstrated that integrating HSCs with LIBs can significantly improve acceleration performance and regenerative braking efficiency.554 For example, hybrid capacitor-battery systems have shown a 15% improvement in acceleration performance compared to standard battery systems.555
➢ Improved regenerative braking: regenerative braking systems benefit from the fast charge–discharge capabilities of HSCs. During braking, HSCs can rapidly absorb and store the energy recovered from braking processes, which can then be swiftly released during acceleration. This process enhances overall vehicle efficiency and reduces energy losses.554 A recent study on hybrid supercapacitors used in regenerative braking systems highlighted their ability to capture up to 30% more energy compared to conventional systems.556
➢ Extended battery life: the integration of HSCs helps extend the life of LIBs by taking over high power duties and reducing the stress on the battery. This results in improved battery longevity and reduces the frequency of replacements, leading to lower long-term costs.557 Research has shown that using HSCs in conjunction with LIBs can reduce battery wear and tear, resulting in a lifespan extension.558
Recent developments and implementations:
Hybrid energy storage systems: the combination of HSCs with LIBs in hybrid energy storage systems (HESS) is becoming increasingly popular in EVs. These systems leverage the strengths of both technologies to optimize performance, providing a balance between high energy density and high power density. For instance, a study on HESS using graphene-based supercapacitors and lithium iron phosphate batteries demonstrated significant improvements in both acceleration and efficiency.559,560
Fast-charging infrastructure: the use of HSCs in charging infrastructure, such as fast chargers and charging stations, enhances the efficiency of power delivery to EVs. HSCs can provide high power density during rapid charging processes, reducing the overall charging time. Recent advancements in fast-charging technologies have integrated HSCs to achieve charging times of under 10 minutes for a significant charge.554
Advanced battery management systems (BMS): smart BMS incorporating HSCs can improve the overall performance and reliability of EVs. These systems ensure optimal utilization of both batteries and supercapacitors, balancing their load and managing power delivery efficiently. Recent research on smart BMS has demonstrated improved energy management and increased system reliability in EVs using HSCs.549
One of the primary challenges is the cost and scalability of HSCs for large-scale EV applications. Although HSCs offer significant performance benefits, their high production costs can hinder widespread adoption. Research is ongoing to develop cost-effective manufacturing techniques and scalable production processes to make HSCs more accessible to the EV market.561,562 Continued advancements in electrode and electrolyte materials are essential for enhancing the performance of HSCs in EVs. Innovations such as new carbon-based materials, advanced metal oxides, and novel polymer composites hold promise for achieving higher energy densities and improved thermal stability.561,563 Ensuring seamless integration of HSCs with existing EV systems and components is critical. Researchers are focusing on developing compatible interfaces and integration strategies to optimize the performance of hybrid energy storage systems in various vehicle configurations.564,565 Electroactive polymers (EAPs) offer considerable potential for enhancing energy harvesting and sensing systems in electric vehicles (EVs), contributing to improved energy efficiency and vehicle intelligence. Their flexibility, lightweight nature, and ability to convert mechanical energy from motion or vibrations into electrical signals make them suitable for integration into vehicle components such as tires, suspension systems, and seats. These capabilities can support auxiliary power generation, structural health monitoring, and smart interface functions, promoting more sustainable and intelligent vehicle designs. However, further advancements in material durability, power output, and integration techniques are necessary to fully realize their practical application in the EV sector.
6.3. Grid storage
Hybrid supercapacitors are gaining increasing attention for their application in grid energy storage systems due to their ability to deliver high power densities and rapid charge/discharge capabilities. This section provides a comprehensive review of the role of HSCs in grid storage, focusing on their benefits, recent advancements, and ongoing challenges.102Advantages in grid storage:
Peak load management and frequency and voltage stabilization: HSCs in grid storage systems often require rapid response capabilities to manage peak load demands and stabilize the grid. HSCs are particularly suited for these applications due to their high power density, which allows them to discharge energy quickly during peak periods.566 Recent studies have shown that HSCs can effectively reduce peak power demands by up to 15–25% in grid systems, thereby enhancing the overall efficiency of power distribution. This capability is critical for integrating intermittent renewable energy sources and maintaining grid stability.
Long-term durability: the ability of HSCs to undergo rapid charge and discharge cycles makes them ideal for balancing short-term fluctuations in energy supply and demand. Their rapid response time helps mitigate issues related to supply and demand imbalances, such as frequency regulation and voltage stabilization.567,568 Recent research highlights that HSCs can handle up to 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 charge–discharge cycles without significant degradation, making them suitable for long-term grid applications.569
000 charge–discharge cycles without significant degradation, making them suitable for long-term grid applications.569
High energy efficiency: hybrid supercapacitors offer improved energy efficiency compared to traditional battery systems due to their lower internal resistance and higher charge efficiency. This efficiency translates into reduced energy losses during storage and retrieval, which is essential for optimizing grid storage systems. Studies have demonstrated that integrating HSCs into grid storage systems can enhance overall energy efficiency by approximately 15–20%.570,571
Recent developments and innovations:
Renewable energy integration: HSCs are increasingly being integrated with renewable energy sources, such as wind and solar, to address the intermittency issues associated with these technologies. Combining HSCs with renewable energy sources can provide a stable and reliable power supply by storing excess energy during periods of high production and discharging it during low production.572
Advanced materials: recent advancements in this area have shown that HSCs can improve the reliability of renewable energy integration by up to 20%. The development of advanced materials for HSCs is a key area of research aimed at enhancing their performance in grid storage applications. Innovations in electrode materials, such as graphene-based composites and transition metal oxides, have led to significant improvements in energy and power densities. For example, the use of graphene composites has been shown to increase the energy density of HSCs by up to 30%.573,574
Cost-effective production: these advancements are crucial for improving the performance and economic viability of grid storage systems. Cost is a significant factor affecting the widespread adoption of HSCs in grid storage applications. Recent research has focused on developing cost-effective manufacturing techniques, such as scalable synthesis methods and low-cost materials, to reduce the overall cost of HSCs.575 Innovations in manufacturing processes have demonstrated the potential to lower the cost of HSCs, making them more competitive with traditional storage technologies.576
One of the primary challenges in deploying HSCs for grid storage is scalability. The integration of HSCs into large-scale grid storage systems requires the development of scalable infrastructure and efficient system integration strategies. Ongoing research is focused on addressing these challenges by developing scalable technologies and optimizing system design.577 Ensuring the long-term stability and reliability of HSCs in grid storage applications is essential for their successful deployment. Research is being conducted to assess the long-term performance and degradation mechanisms of HSCs, with the goal of improving their durability and operational lifespan.578 The adoption of HSCs in grid storage applications also involves navigating regulatory and standardization issues. The development of industry standards and regulatory frameworks is necessary to ensure the safe and effective use of HSCs in grid systems. Collaboration between researchers, industry stakeholders, and regulatory agencies is crucial for addressing these issues and promoting the widespread adoption of HSC technologies.579
The integration of electroactive polymer (EAP)-based energy harvesting systems into grid storage applications presents a promising avenue for enhancing the flexibility and resilience of future power systems. Their inherent lightweight nature, mechanical compliance, and adaptability to varying environmental stimuli position EAPs as potential components for decentralized energy capture and storage, especially in portable and wearable electronics connected to smart grids. While their energy density and long-term durability currently limit large-scale deployment, advances in material engineering such as hybrid composites and multilayer architectures are expected to bridge the performance gap. Continued research into cost-effective fabrication and energy conversion efficiency could enable EAP-based technologies to complement existing grid storage solutions, fostering a more sustainable and distributed energy infrastructure.
6.4. Industrial applications
Hybrid supercapacitors are emerging as a transformative technology for various industrial applications due to their high power density, rapid charge/discharge capabilities, and long cycle life. This section reviews the current state of HSCs in industrial applications, highlighting their benefits, recent advancements, and areas of ongoing research.HSCs are increasingly being utilized in energy backup systems for industrial applications. Their ability to provide instantaneous power makes them ideal for supporting critical infrastructure during power outages or fluctuations. Recent studies have demonstrated that HSCs can enhance the reliability of backup power systems by providing rapid response and high power output. Integrating HSCs into industrial backup systems improved the energy efficiency and reliability of power delivery.568,580 The integration of HSCs in power management systems for industrial machinery has shown promising results. Their high power density and rapid charge/discharge capabilities allow them to smooth out power fluctuations and stabilize the energy supply to machinery. This application is crucial for preventing machinery malfunctions and ensuring smooth operation. Recent studies demonstrated that HSCs could reduce power fluctuations in industrial machinery, leading to improved operational stability.581–584 Hybrid supercapacitors are also playing a significant role in integrating renewable energy sources into industrial processes. Their ability to store excess energy generated from renewable sources and provide it on demand helps in maintaining a stable energy supply. Recent advancements have shown that HSCs can effectively store and dispatch energy from sources such as wind and solar power. According to Kerim et al. incorporating supercapacitors into photovoltaic energy systems might solve problems like low energy density and high costs while simultaneously improving efficiency, reliability, and energy storage capacity.585 In high-power applications, such as industrial pulse power systems and electric drives, HSCs offer significant advantages. Their high power density allows them to deliver large amounts of power in short bursts, which is essential for applications that require rapid energy delivery. Duan et al. highlighted that HSCs can be used effectively in industrial pulse power systems, providing a substantial increase in power output and operational efficiency.586 HSCs are being explored for their potential in electromagnetic interference (EMI) shielding applications in industrial environments. Their conductive properties make them suitable for mitigating EMI, which can interfere with sensitive electronic equipment. Recent research demonstrated that HSCs can effectively reduce EMI in industrial settings, enhancing the performance and reliability of electronic devices.587 Thermal management in industrial systems is another area where HSCs are proving valuable. Their thermal conductivity properties can be leveraged to manage heat dissipation in electronic and power systems. Scientists reported that HSCs can help improve thermal management in industrial applications by efficiently dissipating heat, thereby enhancing the longevity and performance of electronic components.588
Despite their advantages, the widespread adoption of HSCs in industrial applications faces several challenges. These include high manufacturing costs, scalability issues, and the need for specialized infrastructure. Future research is focused on addressing these challenges by developing cost-effective manufacturing methods, improving material performance, and optimizing system integration.
6.5. Techno-economic outlook and application prospects
The translation of electroactive polymer (EAP)-based self-powered actuators and biosensors from laboratory-scale innovations to real-world biomedical applications hinges not only on scientific breakthroughs but also on techno-economic feasibility. While the unique properties of EAPs—such as high flexibility, low density, and responsiveness to external stimuli—make them attractive for next-generation wearable and implantable systems, several economic and technical considerations must be addressed to facilitate commercial deployment.Material costs and scalability: EAPs, including ionic polymer-metal composites (IPMCs), dielectric elastomers (DEs), and piezoelectric polymers like PVDF and its copolymers, vary significantly in material cost and processability. Although the raw materials for most EAPs are relatively inexpensive, the cost escalates when considering functionalization steps, electrode deposition, and device packaging. For instance, noble metals like platinum and gold, often used as electrodes, increase the overall cost. Therefore, ongoing research into alternative low-cost materials—such as conductive carbon-based fillers (e.g., CNTs, graphene), printable conductive inks, and biodegradable polymers is essential for ensuring scalability without compromising performance.
Manufacturing and integration challenges: one of the major techno-economic bottlenecks is the fabrication of EAP-based devices with consistent performance at scale. Techniques like spin-coating, inkjet printing, or 3D printing must be optimized for high-throughput, reproducibility, and compatibility with biomedical-grade materials. Furthermore, integration with existing electronics, flexible substrates, and encapsulation layers adds complexity and cost. To mitigate these challenges, the development of modular manufacturing strategies and roll-to-roll processing for continuous production is being actively explored.
Device longevity and reliability: from a commercial perspective, the long-term stability and reliability of EAP-based actuators and sensors are critical. These devices must withstand cyclic deformations, physiological conditions (humidity, temperature, pH), and biofouling without degradation. Ensuring device robustness over time reduces replacement frequency and maintenance costs, thereby improving their cost-effectiveness for clinical and consumer applications. Investment in durability testing, encapsulation technologies, and self-healing materials can substantially boost the commercial appeal.
Regulatory and market considerations: commercializing EAP-based biomedical devices also entails navigating regulatory pathways such as FDA and CE approvals. This involves rigorous safety and efficacy testing, biocompatibility assessment, and quality control, which contribute to development costs and time-to-market. Nevertheless, the growing demand for wearable biosensors, soft robotics in surgery, and personalized healthcare tools provides a strong market pull. Strategic partnerships between academia, industry, and healthcare providers are essential to bridge the gap between research and market-ready products.
Cost-to-performance trade-offs: from a techno-economic standpoint, a favorable cost-to-performance ratio is key for EAP-based devices to compete with traditional electronic sensors or actuators. Although EAPs may initially incur higher development costs, their multifunctionality, lightweight design, and energy autonomy (via self-powered mechanisms) could lead to lower operational and maintenance costs over the product lifecycle. For example, self-powered biosensors reduce the need for frequent battery replacements or external power sources, which is especially beneficial in remote or resource-limited healthcare settings.
Commercial prospects and emerging markets: the application prospects for EAP-based self-powered devices span several sectors, including personalized medicine, prosthetics, soft robotics, environmental monitoring, and human–machine interfaces. The growing emphasis on telemedicine, decentralized diagnostics, and real-time health monitoring presents lucrative opportunities. In particular, markets in aging populations, military-grade wearable systems, and sports performance tracking are expected to see significant adoption of such technologies in the near future.
So, while EAP-based self-powered actuators and biosensors offer transformative potential, their widespread adoption will depend on overcoming manufacturing, integration, regulatory, and economic hurdles. Advancements in cost-effective materials, scalable fabrication techniques, and collaborative commercialization models will be pivotal in realizing their full impact across biomedical and industrial domains.
7. Challenges and future directions
7.1. Material challenges
The advancement of hybrid supercapacitors is closely linked to the development and optimization of electrode and electrolyte materials. Despite the progress made, several material-related challenges persist that impact the performance, cost, and scalability of HSCs. This section reviews the key material challenges and recent research efforts aimed at addressing these issues. One of the fundamental challenges in HSCs is balancing energy density and power density. While high energy density is critical for applications requiring extended energy storage, high power density is essential for applications needing rapid discharge and charge cycles. Recent research has focused on developing materials that can simultaneously enhance both properties. For instance, Zhang et al. investigated advanced hybrid electrode materials that offer improved energy and power densities by optimizing the nanostructure of the active materials.589 However, achieving this balance remains a significant hurdle in material science. Another challenge is the long-term stability of materials used in HSCs. Electrode materials must endure repeated charge and discharge cycles without significant degradation, which can lead to decreased performance and shorter lifespans. Recent work by Liu et al. has explored novel coating techniques and composite structures to enhance the durability of electrode materials, such as carbon-based and metal–oxide electrodes.475 Despite these advancements, achieving long-term stability across diverse operational conditions remains a challenge. The cost of materials and the scalability of production processes are critical factors influencing the commercialization of HSCs. Many high-performance materials, such as graphene and certain metal oxides, are expensive and difficult to scale up. Recent studies have addressed these issues by exploring more cost-effective materials and scalable synthesis methods. For example, Mathan et al. discussed the development of low-cost, scalable production methods for graphene-based supercapacitor electrodes.590 However, the challenge of reducing costs while maintaining performance continues to be significant. The environmental impact and safety of materials used in HSCs are gaining increasing attention. Materials must be both environmentally benign and safe for use in various applications. Recent research has focused on developing eco-friendly and non-toxic materials. For instance, Wang et al. reviewed the use of bio-based and biodegradable materials in supercapacitors, aiming to reduce environmental impact. Addressing these concerns is crucial for the widespread adoption of HSC technologies.591 The interface between electrode materials and electrolytes can significantly affect the performance of HSCs. Poor compatibility can lead to increased resistance, reduced efficiency, and shorter lifespan. Recent studies, such as those by Zhao et al. have reviewed various electrode–electrolyte combinations to optimize interface properties.592 Ensuring compatibility and optimizing the interface remain key challenges in material science for HSCs. Accurate characterization of materials at various scales is essential for understanding their performance in HSCs. Advanced characterization techniques can provide insights into materials' structural, chemical, and electrochemical properties. Research by Su et al. and Sun et al. highlighted using advanced microscopy and spectroscopy techniques to characterize new electrode materials and improve their design.593,594 Continued development of characterization methods is vital for advancing material science in HSC technology.7.2. Cost and scalability
The cost and scalability of hybrid supercapacitors remain pivotal challenges impacting their widespread adoption and commercialization. Addressing these issues requires a comprehensive understanding of the factors influencing material costs, production processes, and overall economic viability.The cost of raw materials significantly affects the overall expense of HSCs. Many high-performance materials, such as advanced carbon allotropes (e.g., graphene) and transition metal oxides, are expensive due to their complex synthesis and limited availability. For instance, recent research by Horn et al. and Tiwari et al. highlights that the high cost of graphene is a major barrier to its widespread use in supercapacitors, despite its superior performance characteristics.595,596 Similarly, Liang et al. discuss the cost challenges associated with rare and expensive transition metals used in supercapacitor electrodes.597 Scalable and cost-effective production methods are critical for the commercialization of HSCs. Traditional synthesis methods, such as CVD and sol–gel processing, often involve expensive equipment and complex procedures. Recent advancements have aimed at developing more economical and scalable techniques. For example, Zhang et al. introduced a low-cost, scalable method for synthesizing high-quality graphene oxide, which could potentially reduce the cost of graphene-based supercapacitors.598 Similarly, Delbari et al. reviewed on novel sol–gel method that reduce the production costs of metal oxide-based electrodes for flexible supercapacitors.599 Balancing cost and performance is a significant challenge. Lower-cost materials often compromise performance, while high-performance materials may be prohibitively expensive. Recent studies have focused on finding optimal trade-offs. For instance, Javed et al. reviewed on hybrid materials for hybrid supercapacitors that combine cost-effective components with high-performance additives to balance cost and efficiency.600 This approach aims to make high-performance HSCs more affordable without sacrificing their capabilities. Scaling up production from laboratory to industrial scale presents challenges. Processes that are feasible at a small scale may become costly and inefficient when scaled up. Research by Gulam smdani et al. has examined the economic implications of scaling up synthesis methods and proposed strategies to improve scalability and cost-efficiency.601 Addressing these issues is crucial for the commercial viability of HSC technologies. The recyclability and recovery of materials used in HSCs also impact their cost and environmental footprint. Efficient recycling processes can help reduce material costs and mitigate environmental concerns. Ali et al. (2023) reviewed various recycling methods for supercapacitor materials, highlighting the potential for cost reduction through effective recovery techniques.602 Developing efficient recycling processes is essential for making HSCs more sustainable and economically viable. Market adoption and production costs are interconnected. High production costs can inhibit market growth, while increased market adoption can drive economies of scale and reduce costs. Recent studies have explored how market dynamics influence production costs and vice versa. For instance, Ravindranath et al. analyzed the impact of market trends on the cost of supercapacitor materials and production methods, emphasizing the need for strategies to enhance market adoption.563 Government policies and regulations can also affect the cost and scalability of HSC technologies. Supportive policies and incentives for research and development can help lower costs and accelerate commercialization. Research by Wang et al. (2023) discussed the role of government policies in supporting the development and scaling of advanced energy storage technologies, including supercapacitors.603 Aligning policy frameworks with technological advancements is crucial for reducing costs and fostering growth in the HSC industry.
7.3. Environmental impact and sustainability
The environmental impact and sustainability of hybrid supercapacitors are increasingly recognized as critical factors influencing their development and commercialization. As the demand for advanced energy storage solutions grows, understanding and mitigating the environmental footprint of these technologies becomes paramount.The sourcing of raw materials for HSCs, particularly those involving rare or toxic elements, poses environmental and ethical concerns. Materials such as graphene, transition metal oxides, and conducting polymers often involve complex extraction processes that can be environmentally damaging. For instance, the extraction of rare earth metals used in some supercapacitor electrodes can lead to significant ecological disruption and pollution.604 Recent studies have explored more sustainable sources or alternative materials with lower environmental impacts.605,606 The environmental impact of manufacturing processes for HSCs includes energy consumption, waste generation, and chemical emissions. Advanced fabrication techniques like CVD and sol–gel processes often require significant energy and produce chemical byproducts.607 Researchers are focusing on developing cleaner, less energy-intensive methods that can reduce the environmental footprint of these processes.608 End-of-life management of HSCs is a critical area for environmental sustainability. The disposal and recycling of spent supercapacitors can be challenging, particularly for those containing hazardous materials. Effective recycling processes are essential to recover valuable materials and reduce waste. For instance, Leonard et al. reviewed various recycling strategies for supercapacitor materials and proposed methods to enhance material recovery and minimize environmental impact.609 Efficient recycling technologies are needed to address the challenges associated with the disposal of supercapacitors. The development of sustainable materials for HSCs is a growing area of research. Researchers are investigating alternative materials that offer similar or superior performance to traditional ones but with lower environmental impact. For example, Liu et al. have explored the use of bio-derived materials and green synthesis methods to reduce the environmental impact of supercapacitor electrodes.610 Such approaches aim to minimize the ecological footprint of HSCs while maintaining high performance. Life cycle assessment (LCA) is a comprehensive approach to evaluating the environmental impact of HSCs throughout their lifecycle. LCA considers factors such as raw material extraction, manufacturing, usage, and end-of-life disposal. Recent studies have applied LCA to HSCs to identify key areas for improvement and to guide the development of more sustainable technologies.611,612 This holistic view helps in understanding the overall environmental impact and guides decision-making towards more sustainable practices. Government policies and regulations play a crucial role in promoting environmental sustainability in the development and deployment of HSCs. Policies that encourage the use of environmentally friendly materials and processes, and that support recycling and waste management, can significantly impact the sustainability of HSC technologies.479 Recent research by Benjamin et al. highlights the need for stronger regulatory frameworks and incentives to support sustainable practices in the energy storage sector.613 Market adoption of HSCs is influenced by their environmental impact. Technologies with lower environmental footprints are more likely to gain acceptance in the market. As consumer and regulatory pressures increase, there is a growing emphasis on developing HSCs that meet sustainability criteria. Studies by Chen et al. have analyzed the relationship between market adoption and environmental impact, providing insights into how sustainability considerations affect the commercialization of HSCs.614 Green chemistry approaches are increasingly applied to the development of HSCs to minimize environmental impact. These approaches focus on designing materials and processes that reduce the use of hazardous substances and enhance sustainability. Recent advancements include the use of non-toxic solvents, recyclable materials, and energy-efficient synthesis methods.615,616 Such innovations are critical for advancing the sustainability of HSC technologies.
7.4. Future research directions
The field of hybrid supercapacitors is rapidly evolving, and future research will play a crucial role in addressing current limitations and advancing technology. Several key areas of focus are emerging, which hold promise for enhancing the performance, sustainability, and applicability of HSCs. Future research should focus on the development of novel electrode materials that offer improved performance characteristics, such as higher capacitance, energy density, and stability. Advanced 2D materials like MXenes, Prussian blue frameworks, MOFs, COFs, and HOFs are gaining attention for their high surface area and tunable properties.55,56,617,618Fig. 19 illustrated a schematic outlines the key research directions and innovations driving the evolution of hybrid supercapacitors, including advancements in electrode materials, electrolytes, fabrication methods, and system integration for next-generation energy storage applications.Recent studies, such as those by Cheng et al. have demonstrated the potential of MOFs in enhancing the performance of supercapacitors.619 Similarly, COFs are being explored for their exceptional porosity and stability, as highlighted by Umer et al.279 Exploring new electrolyte systems is crucial for improving the overall performance of HSCs. Researchers are investigating solid-state electrolytes and ionic liquids to overcome the limitations of traditional aqueous and organic electrolytes. For instance, recent work by Wu et al. has focused on developing gel electrolytes with enhanced ionic conductivity and stability.620 Similarly, Meisner et al. have proposed advanced polymer electrolytes that offer improved performance in various operating conditions.621 The integration of HSCs with emerging technologies such as flexible electronics, wearable devices, and energy harvesting systems presents a promising avenue for future research. The development of flexible and stretchable HSCs could lead to innovative applications in wearable electronics. Studies by Liu et al. highlight the integration of HSCs into flexible substrates, which could significantly enhance the versatility of energy storage solutions.22 Future research must also address the environmental impact of HSCs throughout their lifecycle. Efforts are needed to develop sustainable materials and processes, as well as effective recycling methods. Researchers like Meskhar et al. have explored eco-friendly synthesis routes and recyclable materials to reduce the environmental footprint of HSCs.622 Additionally, Yin et al. have investigated the use of green chemistry approaches in the development of supercapacitors.623 The optimization of fabrication techniques is crucial for scaling up the production of HSCs and improving their performance. Research into more efficient and cost-effective manufacturing processes, such as the sol–gel method and hydrothermal synthesis, is essential. Recent studies demonstrated that insights into optimizing these methods for better material properties and performance.384,624–626 Innovative device architectures and configurations can significantly impact the performance of HSCs. Research into novel hybrid configurations and electrode designs can lead to improvements in energy and power density. For example, Chen et al. have investigated new hybrid supercapacitor designs that integrate multiple electrode materials to enhance overall performance.627 Improving testing methodologies and performance metrics is essential for better evaluating HSCs. Future research should focus on developing more accurate and comprehensive testing protocols to assess parameters such as rate capability, cycling stability, and energy density. Studies by Yu Yin et al. have proposed new performance metrics and testing techniques to better understand the behavior of HSCs under different conditions.628 Interdisciplinary collaboration among material scientists, engineers, and chemists is vital for advancing HSC technology. Collaborative efforts can lead to breakthroughs in material development, device design, and application integration. Recent research underscores the importance of such collaborations in driving innovation and overcoming existing challenges.479,629,630
8. Conclusion
8.1. Summary of key findings
The review of hybrid supercapacitors underscores their pivotal role in advancing energy storage technologies, showcasing significant progress in materials, design, and applications. This section summarizes the key findings from the reviewed literature and highlights the advancements and future directions in the field. Recent developments in electrode materials for HSCs have led to notable improvements in performance metrics. Carbon-based materials, including activated carbon, graphene, and carbon nanotubes, continue to be at the forefront due to their high surface area and conductivity. For example, graphene and its derivatives have been shown to significantly enhance the energy density and power density of HSCs. Metal oxides, particularly transition metal oxides and layered double hydroxides, have also demonstrated substantial improvements in capacitance and stability. Additionally, conducting polymers such as PANI and PPy have contributed to enhancing the charge storage capability of HSCs. The development of advanced electrolytes has been crucial for optimizing HSC performance. Aqueous, organic, and ionic liquid electrolytes have been extensively studied. Recent advancements in solid-state electrolytes, including gel and polymer electrolytes, have provided solutions for enhancing safety and stability while maintaining high ionic conductivity. The evolution of design and fabrication techniques has significantly impacted the performance and scalability of HSCs. Techniques such as CVD, sol–gel methods, and hydrothermal synthesis have been refined to produce high-quality electrodes. Cell assembly methods, including coin and pouch cell assemblies, have been optimized to improve energy and power density. The assessment of HSC performance through metrics such as capacitance, power density, cycling stability, and rate capability has highlighted their competitive advantages over conventional energy storage systems. Recent studies emphasize the importance of accurate testing protocols to evaluate the long-term stability and efficiency of HSCs under various operational conditions. Hybrid supercapacitors are being increasingly integrated into diverse applications, from consumer electronics and electric vehicles to grid storage and industrial systems. Their high power density and long cycle life make them suitable for applications requiring rapid charge–discharge cycles and reliable performance. Future research directions include exploring new materials, enhancing scalability, and addressing environmental sustainability to further advance the field.8.2. Potential impact on energy storage technologies
Hybrid supercapacitors represent a transformative advancement in energy storage technologies, merging the high power density characteristics of supercapacitors with the enhanced energy density typical of batteries. This synergy positions HSCs as pivotal components in the next generation of energy storage systems, with significant implications across various sectors. The integration of diverse electrode materials, including carbon-based materials (e.g., graphene and carbon nanotubes), metal oxides, and conducting polymers, has markedly improved the performance of HSCs. These advancements have led to substantial increases in both energy and power densities, enabling HSCs to deliver rapid bursts of energy while maintaining a high energy storage capacity. This enhanced performance makes HSCs highly suitable for applications requiring both high power and high energy, such as in electric vehicles and grid storage. HSCs exhibit versatility across various applications, from consumer electronics to large-scale grid storage systems. Their ability to deliver high power and maintain stability over numerous charge–discharge cycles makes them ideal for use in electric vehicles, where both quick acceleration and efficient energy recovery are crucial. Additionally, their role in grid storage solutions addresses the challenge of balancing supply and demand by providing rapid energy discharge capabilities. The development of advanced electrolytes, including organic electrolytes, ionic liquids, and solid-state electrolytes, has further enhanced the performance and safety of HSCs. Solid-state electrolytes, in particular, offer improved safety profiles by reducing the risk of leakage and flammability, thus making HSCs more viable for a broader range of applications. The potential environmental impact and sustainability of HSCs are crucial considerations. Advances in material science and fabrication techniques are addressing these concerns by developing more sustainable and recyclable materials. However, ongoing research is needed to minimize the environmental footprint associated with the production and disposal of HSCs. Future research is poised to focus on several key areas, including the development of novel materials with enhanced properties, optimization of fabrication processes, and reduction of costs. Innovations in material science, such as the exploration of MOFs and COFs, promise to further elevate the performance of HSCs. Additionally, integrating HSCs with emerging technologies, such as advanced energy management systems, could significantly broaden their application scope. The hybrid supercapacitors hold substantial promise for advancing energy storage technologies, offering a unique blend of high power and energy density. Their continued development and integration into various applications are expected to drive significant improvements in energy storage efficiency and performance, paving the way for a more sustainable and technologically advanced future.8.3. Final remarks
In conclusion, hybrid supercapacitors represent a significant leap forward in the realm of energy storage technologies. By synergistically combining the rapid charge–discharge capabilities of supercapacitors with the high energy density of batteries, they offer a versatile and efficient solution to the limitations of conventional energy storage systems. Advances in materials science, especially in electrode development using carbon-based substances, metal oxides, conducting polymers, and their composites, have substantially enhanced both energy and power densities, as well as cyclic stability.Furthermore, innovations in electrolyte technologies, including aqueous, organic, ionic liquid, and solid-state systems, have expanded the operational voltage windows, safety profiles, and environmental adaptability of hybrid supercapacitors. Simultaneously, progress in design and fabrication techniques such as scalable nanostructuring, layer-by-layer assembly, and additive manufacturing has enabled high-performance architectures and miniaturized systems suitable for emerging applications.
Looking ahead, future research in hybrid supercapacitors should focus on the following key directions:
• Development of multifunctional and sustainable materials: designing eco-friendly, low-cost, and abundant electrode and electrolyte materials with high redox activity, mechanical flexibility, and long-term stability will be crucial.
• Structural optimization and nanostructuring strategies: engineering hierarchical and porous architectures at the micro/nanoscale will improve ion/electron transport, energy density, and durability.
• Exploration of novel hybrid systems: integrating metal–organic frameworks (MOFs), MXenes, 2D materials, and bio-derived nanomaterials could unlock new charge storage mechanisms and improve performance metrics.
• Smart integration with energy systems: embedding hybrid supercapacitors into wearable electronics, IoT devices, and hybrid energy systems (e.g., solar + supercapacitor) will broaden their applicability.
• AI-driven design and modeling: employing artificial intelligence and machine learning for predictive modeling, materials screening, and system optimization will accelerate innovation cycles.
• Green and scalable manufacturing techniques: emphasizing roll-to-roll processing, 3D printing, and solvent-free fabrication will enhance commercial scalability and environmental sustainability.
• Lifecycle and recyclability considerations: addressing end-of-life recycling, reusability of components, and environmental impact will be essential for sustainable deployment.
As hybrid supercapacitors continue to evolve, they are self-contained to play a transformative role across sectors ranging from consumer electronics and electric vehicles to grid storage and biomedical systems. Their ability to deliver rapid bursts of energy while maintaining long operational lifetimes positions them as indispensable components in the future of energy storage and management. The ongoing convergence of materials innovation, smart fabrication, and system-level integration will be instrumental in shaping the next generation of high-performance, cost-effective, and sustainable hybrid supercapacitors.
Data availability
No new data were generated or analyzed in this study. The information presented in this review is based on previously published literature, which has been appropriately cited. Therefore, data sharing is not applicable to this article.Author contributions
N. P., D. M., and T. K. M. contributed to conceptualization, original draft preparation, and writing review and editing, as well as software development. M. K. validation, visualization, and S. W. J. responsible for validation, visualization, and editing. J. H. J. and T. K. M. contributed to validation, visualization, and writing review and editing. All authors have read and agreed to the published version of the manuscript. N. P. and D. M. contributed equally to this work.Conflicts of interest
The authors declare no conflicts of interest.Acknowledgements
This work was supported by the National Research Foundation of Korea (NRF) grant funded by the Korean government NRF-2019R1A5A8080290.References
- J. Wang and W. Azam, Geosci. Front., 2024, 15, 101757 CrossRef CAS.
- D. Merum, S. K. Arla, D. Radhalayam, A. M. Tighezza, S. P. Mooni and S. W. Joo, J. Energy Storage, 2024, 99, 113189 CrossRef.
- L. Mu, S. Qiu, G. Zhao, B. Zhang, W. Liao, N. Zhao and X. Xu, J. Mater. Chem. A, 2024, 12, 1714–1724 RSC.
- A. Raihan, Energy Nexus, 2023, 9, 100180 CrossRef CAS.
- Reenu, Sonia, L. Phor, A. Kumar and S. Chahal, J. Energy Storage, 2024, 84, 110698 CrossRef.
- K. C. S. Lakshmi and B. Vedhanarayanan, Batteries, 2023, 9, 202 CrossRef CAS.
- O. S. Adedoja, E. R. Sadiku and Y. Hamam, Polymers, 2023, 15, 2272 CrossRef CAS PubMed.
- L. Xu, J. Li, H. Shuai, Z. Luo, B. Wang, S. Fang, G. Zou, H. Hou, H. Peng and X. Ji, J. Energy Chem., 2022, 67, 524–548 Search PubMed.
- K. O. Oyedotun, J. O. Ighalo, J. F. Amaku, C. Olisah, A. O. Adeola, K. O. Iwuozor, K. G. Akpomie, J. Conradie and K. A. Adegoke, J. Electron. Mater., 2023, 52, 96–129 CrossRef CAS.
- X. Gao, J. Yang, Z. Xu, Y. Nuli and J. Wang, Energy Storage Materials, 2023, 54, 382–402 Search PubMed.
- F. M. N. U. Khan, M. G. Rasul, A. S. M. Sayem and N. Mandal, Energy Reports, 2023, 9, 11–21 CrossRef.
- D. P. Dubal and P. Gomez-Romero, Materials Today Energy, 2018, 8, 109–117 CrossRef.
- M. Duduta, S. de Rivaz, D. R. Clarke and R. J. Wood, Batteries Supercaps, 2018, 1, 131–134 CrossRef.
- H. A. Khan, M. Tawalbeh, B. Aljawrneh, W. Abuwatfa, A. Al-Othman, H. Sadeghifar and A. G. Olabi, Energy, 2024, 295, 131043 CrossRef CAS.
- Y. M. Volfkovich, J. Electroanal. Chem., 2024, 963, 118290 CrossRef CAS.
- M. Girirajan, A. K. Bojarajan, I. N. Pulidindi, K. N. Hui and S. Sangaraju, Coord. Chem. Rev., 2024, 518, 216080 CrossRef CAS.
- C. N. Mohammad, B. N. Murthy, H. M. Shareef and S. Ghosh, in 2024 7th International Conference on Devices, Circuits and Systems (ICDCS), IEEE, 2024, pp. 216–220 Search PubMed.
- K. K. Garlapati, S. Naskar, S. K. Martha and B. B. Panigrahi, ACS Appl. Energy Mater., 2024, 6950, DOI:10.1021/acsaem.4c00962.
- I. Offen-Polak, M. Auinat, N. Sezin, Y. Ein-Eli and M. Balaish, J. Power Sources, 2021, 498, 229902 CrossRef CAS.
- K. T. VanSant, A. C. Tamboli and E. L. Warren, Joule, 2021, 5, 514–518 CrossRef.
- S. S. Shah, Batteries, 2024, 10, 168 CrossRef CAS.
- S. Liu, J. Yang, P. Chen, M. Wang, S. He, L. Wang and J. Qiu, Electrochem. Energy Rev., 2024, 7, 25 CrossRef CAS.
- R. Mohanty, U. A. Mohanty and K. Parida, Energy Fuels, 2024, 38, 13585–13611 CrossRef CAS.
- N. Loura, S. Mor, D. Nandal, V. Nandal and V. Dhull, Energy Storage, 2024, e559, DOI:10.1002/est2.559.
- Q. Muhammad Saqib, A. Mannan, M. Noman, M. Y. Chougale, C. S. Patil, Y. Ko, J. Kim, S. R. Patil, M. Yousuf, R. Ali Shaukat, Y. Pyo Jeon, D. Dubal and J. Bae, Chem. Eng. J., 2024, 490, 151857 CrossRef CAS.
- P. Saha, N. C. D. Nath, M. M. Islam, M. A. Aziz and A. J. S. Ahammad, Electrochim. Acta, 2024, 144892 CrossRef CAS.
- S. B. Aziz, A. R. Murad, R. T. Abdulwahid, D. M. Aziz, A. A. Abdalrahman, R. M. Abdullah, M. F. Z. Kadir, O. G. Abdullah, N. A. Halim and J. Hassan, Int. J. Biol. Macromol., 2024, 273, 133203 CrossRef CAS PubMed.
- Y. Shao, M. F. El-Kady, J. Sun, Y. Li, Q. Zhang, M. Zhu, H. Wang, B. Dunn and R. B. Kaner, Chem. Rev., 2018, 118, 9233–9280 CrossRef CAS PubMed.
- H. Huang and M. Niederberger, Nanoscale, 2019, 11, 19225–19240 RSC.
- S. Fleischmann, J. B. Mitchell, R. Wang, C. Zhan, D. Jiang, V. Presser and V. Augustyn, Chem. Rev., 2020, 120, 6738–6782 CrossRef CAS PubMed.
- S. J. Panchu, K. Raju and H. C. Swart, ChemElectroChem, 2024, e202300810, DOI:10.1002/celc.202300810.
- N. Nitta, F. Wu, J. T. Lee and G. Yushin, Mater. Today, 2015, 18, 252–264 CrossRef CAS.
- M. Li, J. Lu, Z. Chen and K. Amine, Adv. Mater., 2018, 30, e1800561 CrossRef PubMed.
- A. Manthiram, Nat. Commun., 2020, 11, 1550 CrossRef CAS PubMed.
- M. U. Khalid, S. Zulfiqar, M. F. Warsi, I. Shakir, M. S. Al-Buriahi and E. W. Cochran, Energy Fuels, 2024, 38, 5506–5521 CrossRef CAS.
- A. Tundwal, H. Kumar, B. J. Binoj, R. Sharma, G. Kumar, R. Kumari, A. Dhayal, A. Yadav, D. Singh and P. Kumar, RSC Adv., 2024, 14, 9406–9439 RSC.
- S. Alosious, S. R. Tee and D. J. Searles, J. Phys. Chem. C, 2024, 128, 2190–2204 CrossRef CAS.
- T. Ramachandran, R. K. Raji and M. Rezeq, J. Mater. Chem. A, 2025, 13, 12855–12890 RSC.
- Y. Chen, Y. Shi, G. Song, B. Yang and H. Pang, Next Mat., 2025, 6, 100323 Search PubMed.
- Y. Anil Kumar, S. Vignesh, T. Ramachandran, A. M. Fouda, H. H. Hegazy, M. Moniruzzaman and T. Hwan Oh, J. Ind. Eng. Chem., 2025, 145, 191–215 CrossRef CAS.
- M. Saha, A. Kumar, R. Kanaoujiya, K. Behera and S. Trivedi, Energy Fuels, 2024, 38, 8528–8552 CrossRef CAS.
- S. Lan, C. Yu, J. Yu, X. Zhang, Y. Liu, Y. Xie, J. Wang and J. Qiu, Small, 2024, e2309286, DOI:10.1002/smll.202309286.
- I. G. da Silva, M. Anouti, M. F. Montemor and I. M. Marrucho, ACS Sustain. Chem. Eng., 2024, 12, 1365–1377 CrossRef CAS.
- S. S. Shah and M. A. Aziz, J. Chem. Environ., 2024, 1–45 Search PubMed.
- M. Heydari Gharahcheshmeh and K. Chowdhury, Energy Adv., 2024, 3, 2668–2703 RSC.
- C. Shi, J. Sun, Y. Pang, Y. Liu, B. Huang and B.-T. Liu, J. Colloid Interface Sci., 2022, 607, 462–469 Search PubMed.
- Supercapacitorsed, ed. F. Béguin and E. Frąckowiak, Wiley, 2013 Search PubMed.
- S. V. Sadavar, S. Lee and S. Park, Adv. Sci., 2024, 2403172, DOI:10.1002/advs.202403172.
- S. Subhadarshini, N. K. Peyada, D. K. Goswami and N. C. Das, Energy Fuels, 2024, 38, 11455–11493 CrossRef CAS.
- M. Cui and X. Meng, Nanoscale Adv., 2020, 2, 5516–5528 RSC.
- X. Zhang, C. Jiang, J. Liang and W. Wu, J. Mater. Chem. A, 2021, 9, 8099–8128 RSC.
- J. Pu, Y. Wang, X. Chen, C. Guan and C. Cheng, Nano Futures, 2020, 4, 032002 CrossRef CAS.
- C. Y. Chan, Z. Wang, H. Jia, P. F. Ng, L. Chow and B. Fei, J. Mater. Chem. A, 2021, 9, 2043–2069 RSC.
- C. Valentini, V. Montes-García, D. Pakulski, P. Samorì and A. Ciesielski, Small, 2024, 20(52), 202410544 Search PubMed.
- K. Patra, S. Dey, C. Solanki, A. Sengupta and V. K. Mittal, ACS Appl. Eng. Mater., 2025, 3, 1130–1165 CrossRef CAS.
- Z. Xu, Y. Liu, Z. Wu, R. Wang, Q. Wang, T. Li, J. Zhang, J. Cheng, Z. Yang, S. Chen, M. Miao and D. Zhang, Chem. Eng. J., 2020, 387, 124071 CrossRef CAS.
- K. Sayed, M. Aref, A. G. Abokhalil and M. A. Mossa, in Energy Storage Devices – A Comprehensive Overview, IntechOpen, 2025 Search PubMed.
- E. G. Shankar, M. V. Paranjape and J. S. Yu, Adv. Compos. Hybrid Mater., 2025, 8, 149 CrossRef CAS.
- J. Mangaiyarkkarasi and J. Shanthalakshmi Revathy, in Innovations in Next-Generation Energy Storage Solutions, IGI Global, 2025, pp. 599–616 Search PubMed.
- A. A. Alguail, A. H. Al-Eggiely, M. M. Gvozdenović, B. Z. Jugović and B. N. Grgur, J. Power Sources, 2016, 313, 240–246 CrossRef CAS.
- S. Gupta, S. B. Carrizosa and B. Aberg, Sci. Rep., 2024, 14, 29277 CrossRef CAS PubMed.
- V. Molahalli, G. Soman, V. S. Bhat, A. Shetty, A. Alodhayb and G. Hegde, RSC Adv., 2024, 14, 32314–32326 RSC.
- D. Merum, R. R. Nallapureddy, M. R. Pallavolu, T. K. Mandal, R. R. Gutturu, N. Parvin, A. N. Banerjee and S. W. Joo, ACS Appl. Energy Mater., 2022, 5, 5561–5578 CrossRef CAS.
- D. Gao, Z. Luo, C. Liu and S. Fan, Green Energy Environ., 2023, 8, 972–988 Search PubMed.
- N. I. Jalal, R. I. Ibrahim and M. K. Oudah, J. Phys.: Conf. Ser., 2021, 1973(1), 17 CrossRef.
- H. V. T. Nguyen, A. Bin Faheem, J. Kim, J. Lee, Q. Jin, B. Koo, W. Kim and K. K. Lee, ACS Appl. Energy Mater., 2023, 6, 3155–3166 Search PubMed.
- X. Pan, Q. Li, T. Wang, T. Shu and Y. Tao, Nanoscale, 2024, 16, 3701–3713 RSC.
- M. I. Ul Hoque and R. Holze, Polymers, 2023, 15, 1–27 CrossRef PubMed.
- N. Wu, X. Bai, D. Pan, B. Dong, R. Wei, N. Naik, R. R. Patil and Z. Guo, Adv. Mater. Interfaces, 2021, 8, 1–17 Search PubMed.
- J. Huang, K. Yuan and Y. Chen, Adv. Funct. Mater., 2022, 32, 1–68 Search PubMed.
- J. Xu and L. Wang, Sci. Rep., 2019, 9, 12557 CrossRef PubMed.
- S. Panigrahy, P. Panda, A. Shekhawat and S. Barman, ACS Appl. Nano Mater., 2023, 6, 3825–3834 CrossRef CAS.
- S. Abbasi, F. Hekmat and S. Shahrokhian, J. Alloys Compd., 2023, 957, 170452 Search PubMed.
- W. Ye, J. Cai, F. Yu, X. Li and X. Wang, Biomass Bioenergy, 2021, 145, 105949 CrossRef CAS.
- H. Zong, A. Zhang, J. Dong, Y. He, H. Fu, H. Guo, F. Liu, J. Xu and J. Liu, Chem. Eng. J., 2023, 475, 146088 CrossRef CAS.
- M. Ashraf, S. S. Shah, I. Khan, M. A. Aziz, N. Ullah, M. Khan, S. F. Adil, Z. Liaqat, M. Usman, W. Tremel and M. N. Tahir, Chem.–Eur. J., 2021, 27, 6973–6984 Search PubMed.
- P. Sinha, K. K. Kar and A. K. Naskar, Adv. Mater. Technol., 2022, 7, 1–14 Search PubMed.
- S. A. Mane, A. A. Kashale, G. P. Kamble, S. S. Kolekar, S. D. Dhas, M. D. Patil, A. V. Moholkar, B. R. Sathe and A. V. Ghule, J. Alloys Compd., 2022, 926, 166722 CrossRef CAS.
- A. Dutta, S. Mitra, M. Basak and T. Banerjee, Energy Storage, 2022, e339, DOI:10.1002/est2.339.
- W. Zuo, R. Li, C. Zhou, Y. Li, J. Xia and J. Liu, Adv. Sci., 2017, 1600539, DOI:10.1002/advs.201600539.
- J. Cherusseri, D. Pandey and J. Thomas, Batteries Supercaps, 2020, 3, 860–875 CrossRef CAS.
- K. Li, X. Wang, S. Li, P. Urbankowski, J. Li, Y. Xu and Y. Gogotsi, Small, 2020, 1906851, DOI:10.1002/smll.201906851.
- K. S. Ranjith, S. M. Ghoreishian, N. R. Chodankar, G. S. R. Raju, S. J. Patil, Y. S. Huh and Y. Han, Int. J. Energy Res., 2022, 46, 15066–15080 CrossRef CAS.
- P. R. Bueno, J. Power Sources, 2019, 414, 420–434 Search PubMed.
- A. Muzaffar, M. B. Ahamed, K. Deshmukh and J. Thirumalai, Renewable Sustainable Energy Rev., 2019, 101, 123–145 Search PubMed.
- S. Anand, M. Wasi Ahmad, A. Syed, A. H. Bahkali, M. Verma, B. Hye Kim and A. Choudhury, J. Ind. Eng. Chem., 2024, 129, 309–320 CrossRef CAS.
- S. Thareja and A. Kumar, Energy Technol., 2020, 8, 1–11 Search PubMed.
- R. B. Rakhi and H. N. Alshareef, J. Power Sources, 2011, 196, 8858–8865 Search PubMed.
- K. P. Annamalai, X. Zheng, J. Gao, T. Chen and Y. Tao, Carbon, 2019, 144, 185–192 CrossRef CAS.
- R. B. Rakhi, W. Chen, D. Cha and H. N. Alshareef, J. Mater. Chem., 2011, 21, 16197–16204 RSC.
- M. R. Biradar, C. R. K. Rao, S. V. Bhosale and S. V. Bhosale, Energy Fuels, 2023, 37, 4671–4681 CrossRef CAS.
- R. Bhosale, S. Bhosale, D. Narale, C. Jambhale and S. Kolekar, Langmuir, 2023, 39, 12075–12089 CrossRef CAS PubMed.
- D. Acharya, A. Muthurasu, T. H. Ko, R. M. Bhattarai, T. Kim, S. H. Chae, S. Saidin, K. Chhetri and H. Y. Kim, ACS Appl. Energy Mater., 2023, 6, 9196–9206 CrossRef CAS.
- B. Karamanova, E. Mladenova, M. Thomas, N. Rey-Raap, A. Arenillas, F. Lufrano and A. Stoyanova, Gels, 2023, 983, DOI:10.3390/gels9120983.
- J. Cherusseri, D. Pandey and J. Thomas, Batteries Supercaps, 2020, 3, 860–875 CrossRef CAS.
- J. Sun, B. Luo and H. Li, Adv. Energy Sustainability Res., 2022, 2100191, DOI:10.1002/aesr.202100191.
- W. He, W. Guo, H. Wu, L. Lin, Q. Liu, X. Han, Q. Xie, P. Liu, H. Zheng, L. Wang, X. Yu and D. Peng, Adv. Mater., 2021, 2005937, DOI:10.1002/adma.202005937.
- T. Ramachandran, H. Butt, L. Zheng and M. Rezeq, J. Energy Storage, 2024, 99, 113425 CrossRef.
- R. Thota, H. Lim and J. H. Bang, Chem. Eng. J., 2022, 431, 133467 Search PubMed.
- E. Pameté, L. Köps, F. A. Kreth, S. Pohlmann, A. Varzi, T. Brousse, A. Balducci and V. Presser, Adv. Energy Mater., 2023, 2301008, DOI:10.1002/aenm.202301008.
- F. Wu, J. Maier and Y. Yu, Chem. Soc. Rev., 2020, 49, 1569–1614 RSC.
- D. P. Chatterjee and A. K. Nandi, J. Mater. Chem. A, 2021, 9, 15880–15918 RSC.
- F. Xing, Z. Bi, F. Su, F. Liu and Z. Wu, Adv. Energy Mater., 2022, 2200594, DOI:10.1002/aenm.202200594.
- F. C. Krause, J.-P. Jones, M. C. Smart, K. B. Chin and E. J. Brandon, Electrochim. Acta, 2021, 374, 137898 CrossRef CAS.
- M. Pathak, D. Bhatt, R. C. Bhatt, B. S. Bohra, G. Tatrari, S. Rana, M. C. Arya and N. G. Sahoo, Chem. Rec., 2023, e202300236, DOI:10.1002/tcr.202300236.
- G. Yang, J. Huang, X. Wan, Y. Zhu, B. Liu, J. Wang, P. Hiralal, O. Fontaine, Y. Guo and H. Zhou, Nano Energy, 2021, 90, 106500 CrossRef CAS.
- Y. Jiang, K. Ma, M. Sun, Y. Li and J. Liu, Energy Environ. Mater., 2023, 6, 1–8 Search PubMed.
- A. Lahyani, A. Sari, I. Lahbib and P. Venet, J. Energy Storage, 2016, 6, 222–231 CrossRef.
- D. Gao, Z. Luo, C. Liu and S. Fan, Green Energy Environ., 2023, 8, 972–988 CrossRef CAS.
- Y. E. Durmus, H. Zhang, F. Baakes, G. Desmaizieres, H. Hayun, L. Yang, M. Kolek, V. Küpers, J. Janek, D. Mandler, S. Passerini and Y. Ein-Eli, Adv. Energy Mater., 2020, 2000089, DOI:10.1002/aenm.202000089.
- H. He, J. Lian, C. Chen, Q. Xiong and M. Zhang, Chem. Eng. J., 2021, 421, 129786 CrossRef CAS.
- S. Islam, H. Weerasinghe, D. M. Prado, A. N. Gonzaga and C. Burda, Energy Fuels, 2025, 39, 8369–8390 CrossRef CAS.
- S. Iqbal, H. Khatoon, A. Hussain Pandit and S. Ahmad, Mater. Sci. Energy Technol., 2019, 2, 417–428 Search PubMed.
- G. Kothandam, G. Singh, X. Guan, J. M. Lee, K. Ramadass, S. Joseph, M. Benzigar, A. Karakoti, J. Yi, P. Kumar and A. Vinu, Adv. Sci., 2023, 2301045, DOI:10.1002/advs.202301045.
- R. Ahmad Aftab, S. Zaidi, A. Aslam Parwaz Khan, M. Arish Usman, A. Y. Khan, M. Tariq Saeed Chani and A. M. Asiri, Alexandria Eng. J., 2023, 71, 355–369 CrossRef.
- T. Hajilounezhad, R. Bao, K. Palaniappan, F. Bunyak, P. Calyam and M. R. Maschmann, npj Comput. Mater., 2021, 7, 134 CrossRef CAS.
- I. Rafique, A. Kausar, Z. Anwar and B. Muhammad, Polym.-Plast. Technol. Eng., 2016, 55, 312–333 CrossRef CAS.
- R. Devi and S. S. Gill, Microsyst. Technol., 2021, 27, 3225–3233 CrossRef CAS.
- N. Yang, S. Yu, J. V. Macpherson, Y. Einaga, H. Zhao, G. Zhao, G. M. Swain and X. Jiang, Chem. Soc. Rev., 2019, 48, 157–204 RSC.
- S. Jha, B. Akula, P. Boddu, M. Novak, H. Enyioma, R. Cherradi and H. Liang, Mater. Today Sustain., 2023, 22, 100375 Search PubMed.
- Y. Anil Kumar, G. Koyyada, T. Ramachandran, J. H. Kim, S. Sajid, M. Moniruzzaman, S. Alzahmi and I. M. Obaidat, Nanomaterials, 2023, 13, 1049 CrossRef CAS PubMed.
- M. S. Waris, A. H. Chaudhary, S. Anwer, P. P. I. Sultana, S. A. A. Nami and M. Z. Khan, Energy Fuels, 2023, 37, 19433–19460 CrossRef CAS.
- D. R. Lobato-Peralta, P. U. Okoye and C. Alegre, J. Power Sources, 2024, 617, 235140 CrossRef CAS.
- M. Dwiyaniti, A. Elang Barruna, R. Muhamad Naufal, I. Subiyanto, R. Setiabudy and C. Hudaya, IOP Conf. Ser.:Mater. Sci. Eng., 2020, 909, 012018 CAS.
- Y. Li, D. Zhang, Y. Zhang, J. He, Y. Wang, K. Wang, Y. Xu, H. Li and Y. Wang, J. Power Sources, 2020, 448, 227396 CrossRef CAS.
- A. T. S. C. Brandão, S. State, R. Costa, P. Potorac, J. A. Vázquez, J. Valcarcel, A. F. Silva, L. Anicai, M. Enachescu and C. M. Pereira, ACS Omega, 2023, 8, 18782–18798 CrossRef PubMed.
- W. Yang, C. Zhu, L. Li, W. Wang and Z. Yang, Funct. Mater. Lett., 2021, 14, 2150022 CrossRef CAS.
- V. Skrypnychuk, N. Boulanger, A. Nordenström and A. Talyzin, J. Phys. Chem. Lett., 2020, 11, 3032–3038 CrossRef CAS PubMed.
- H. Chen, F. Yu, G. Wang, L. Chen, B. Dai and S. Peng, ACS Omega, 2018, 3, 4724–4732 CrossRef CAS PubMed.
- H. Pecenek, F. K. Dokan, M. S. Onses, E. Yılmaz and E. Sahmetlioglu, J. Energy Storage, 2023, 64, 107075 CrossRef.
- Y. Li, J. Mei, L. Wu, Q. Xu and Z. Li, Int. J. Hydrogen Energy, 2024, 49, 67–80 CAS.
- S. Hajibaba, S. Gholipour, M. Pourjafarabadi, A. Bakhshayesh, M. M. Byranvand, M. Saliba and Y. Abdi, J. Energy Storage, 2024, 79, 110044 CrossRef.
- S. Uppugalla, R. Pothu, R. Boddula, M. A. Desai and N. Al-Qahtani, Emergent Mater., 2023, 6, 1167–1176 CrossRef CAS.
- S. K. Pati, Y. Hwang, H.-M. Lee, B.-J. Kim and S. Park, Carbon Lett., 2024, 34, 153–162 CrossRef CAS.
- A. Agrawal, A. Gaur and A. Kumar, J. Energy Storage, 2023, 66, 107395 CrossRef.
- J.-G. Wang, Y. Yang, Z.-H. Huang and F. Kang, Carbon, 2013, 61, 190–199 CrossRef CAS.
- Y.-G. Lee and G.-H. An, ACS Appl. Mater. Interfaces, 2020, 12, 41342–41349 CrossRef CAS PubMed.
- I. Benchikh, A. O. Ezzat, L. Sabantina, Y. Benmimoun and A. Benyoucef, Polymers, 2024, 16, 421 CrossRef CAS PubMed.
- R. S. Salama, M. S. Gouda, M. F. A. Aboud, F. T. Alshorifi, A. A. El-Hallag and A. K. Badawi, Sci. Rep., 2024, 14, 8223 CrossRef CAS PubMed.
- A. Witecka, P. Pietrzyk-Thel, M. Krajewski, K. Sobczak, A. Wolska and A. Jain, J. Alloys Compd., 2024, 985, 174040 CrossRef CAS.
- S. Kour, P. Kour and A. L. Sharma, Nanoscale, 2024, 16, 13627–13641 RSC.
- S. S. Shah, M. A. Aziz, M. Ali, A. S. Hakeem and Z. H. Yamani, Small, 2023, 2306665, DOI:10.1002/smll.202306665.
- J. Wan, G. Zhang, H. Jin, J. Wu, N. Zhang, B. Yao, K. Liu, M. Liu, T. Liu and L. Huang, Carbon, 2022, 191, 340–349 CrossRef CAS.
- R. Kumar, S. Sahoo, E. Joanni, R. K. Singh and K. K. Kar, ACS Appl. Mater. Interfaces, 2022, 14, 20306–20325 CrossRef CAS PubMed.
- N. Li, S. Tang, Y. Dai and X. Meng, J. Mater. Sci., 2014, 49, 2802–2809 CrossRef CAS.
- H. Sabeeh, S. Zulfiqar, M. Aadil, M. Shahid, I. Shakir, M. A. Khan and M. F. Warsi, Ceram. Int., 2020, 46, 21064–21072 CrossRef CAS.
- Y. Du, P. Xiao, J. Yuan and J. Chen, Coatings, 2020, 10, 892 CrossRef CAS.
- M. Yu, R. Dong and X. Feng, J. Am. Chem. Soc., 2020, 142, 12903–12915 CrossRef CAS PubMed.
- P. Ramesh, A. A. Kalicharan, J. Pitchaimani and R. Koutavarapu, New J. Chem., 2023, 47, 4609–4613 RSC.
- Y. Wang, Y. Chen, S. D. Lacey, L. Xu, H. Xie, T. Li, V. A. Danner and L. Hu, Mater. Today, 2018, 21, 186–192 CrossRef CAS.
- Z. Hou, S. Zou and J. Li, J. Alloys Compd., 2020, 827, 154390 CrossRef CAS.
- S. Mohammadi, A. M. Homayounfard and S. M. Mousavi-Khoshdel, ACS Appl. Energy Mater., 2024, 7, 6142–6154 CrossRef CAS.
- M. Haghshenas, M. Mazloum-Ardakani, L. Amiri-Zirtol and F. Sabaghian, Int. J. Hydrogen Energy, 2021, 46, 30219–30229 CrossRef CAS.
- R. Aliakbari, E. Kowsari, H. R. Naderi, S. Ramakrishna, A. Chinnappan and M. D. Najafi, J. Alloys Compd., 2022, 914, 165371 CrossRef CAS.
- Y. Belaustegui, I. Rincón, F. Fernández-Carretero, P. Azpiroz, A. García-Luís and D. A. P. Tanaka, Chem. Eng. J. Adv., 2021, 6, 100094 CrossRef CAS.
- B. Zhao, D. Chen, X. Xiong, B. Song, R. Hu, Q. Zhang, B. H. Rainwater, G. H. Waller, D. Zhen, Y. Ding, Y. Chen, C. Qu, D. Dang, C.-P. Wong and M. Liu, Energy Storage Materials, 2017, 7, 32–39 CrossRef.
- S. Kumar, S. Sekar, A. K. Kaliamurthy and S. Lee, J. Mater. Res. Technol., 2021, 12, 2489–2501 CrossRef CAS.
- N. James, S. Simon, S. Rajeevan, S. C. George and S. P. Balakrishnan, J. Macromol. Sci., Part B: Phys., 2024, 63, 750–771 CrossRef CAS.
- C. Wu, Z. Zhang, Z. Chen, Z. Jiang, H. Li, H. Cao, Y. Liu, Y. Zhu, Z. Fang and X. Yu, Nano Res., 2021, 14, 953–960 CrossRef CAS.
- M. Z. Abadin, A. Abdul Wahid and M. Usman, J. Microsc., 2025 DOI:10.1111/jmi.13427.
- G. Manibalan, Y. Govindaraj, R. Rajaram, R. Mallappa Dugani, G. Ramalingam, G. Murugadoss, L. Neelakantan and R. Murugavel, Energy Fuels, 2024, 38, 15744–15761 CrossRef CAS.
- W. Liu and G. Speranza, ACS Omega, 2021, 6, 6195–6205 CrossRef CAS PubMed.
- M. Sadiq, M. U. Islam, M. M. Moharam, E. A. M. Saleh and S. U. Asif, J. Mater. Sci.: Mater. Electron., 2024, 35, 1011 CrossRef CAS.
- Y. Pathaare, A. M. Reddy, P. Sangrulkar, B. Kandasubramanian and A. Satapathy, Hybrid Adv., 2023, 3, 100041 CrossRef.
- H. T. Das, S. Dutta, T. E. Balaji, N. Das, P. Das, N. Dheer, R. Kanojia, P. Ahuja and S. K. Ujjain, Chemosensors, 2022, 10, 223 CrossRef CAS.
- T. Arunkumar, R. Karthikeyan, R. Ram Subramani, K. Viswanathan and M. Anish, Int. J. Ambient Energy, 2020, 41, 452–456 CrossRef CAS.
- A. H. Hamad, M. N. Rashko, S. M. Hamad and A. A. Barzinjy, Eurasian J. Sci. Eng., 2022, 54–68, DOI:10.23918/eajse.v8i2p54.
- M. G. Tadesse, A. S. Ahmmed and J. F. Lübben, J. Compos. Sci., 2024, 8, 53 CrossRef CAS.
- V. D. Silva, Y. J. Dias, D. A. Macedo, E. S. Medeiros and A. L. Yarin, Appl. Mater. Today, 2023, 31, 101776 CrossRef.
- A. Khan and K. A. Alamry, Surface Modified Carbon Nanotubes: An Introduction, ACS Symposium Series, 2022, ch. 1, vol. 1424, pp. 1–25, DOI:10.1021/bk-2022-1424.ch001.
- B. Dziejarski, J. Serafin, K. Andersson and R. Krzyżyńska, 2023, 24, 100483.
- G. Song, C. Li, T. Wang, K. H. Lim, F. Hu, S. Cheng, E. Hondo, S. Liu and S. Kawi, Small, 2024, 2305517, DOI:10.1002/smll.202305517.
- M. Mandal, I. Alam, M. R. Sahoo, S. Patra, A. Mahapatra, A. K. Nayak, S. Subudhi and P. Mahanandia, Chem. Phys. Lett., 2024, 853, 141536 CrossRef CAS.
- J. Sun, S. Li, Y. Li, X. Zhang and Z. Yang, ACS Appl. Energy Mater., 2024, 7, 7450–7458 CrossRef CAS.
- M. Mandal, S. Subudhi, I. Alam, B. Subramanyam, S. Patra, J. Raiguru, S. Das and P. Mahanandia, Inorg. Chem. Commun., 2021, 123, 108332 CrossRef CAS.
- X. Luo, Y. Liang, W. Weng, Z. Hu, Y. Zhang, J. Yang, L. Yang and M. Zhu, Energy Storage Materials, 2020, 33, 11–17 CrossRef.
- T. Kshetri, D. T. Tran, D. C. Nguyen, N. H. Kim, K. Lau and J. H. Lee, Chem. Eng. J., 2020, 380, 122543 CrossRef CAS.
- M. S. Lal, R. Badam, N. Matsumi and S. Ramaprabhu, J. Energy Storage, 2021, 40, 102794 CrossRef.
- T. Dang, D. Wei, G. Zhang, L. Wang, Q. Li, H. Liu, Z. Cao, G. Zhang and H. Duan, Electrochim. Acta, 2020, 341, 135988 CrossRef CAS.
- M. W. Ahmad, J. Singh, A. Syed, A. H. Bahkali, M. Subramaniam, D.-J. Yang and A. Choudhury, Energy Fuels, 2024, 38, 14672–14684 CrossRef CAS.
- Z. Ma, R. Zheng, Y. Liu, Y. Ying and W. Shi, J. Colloid Interface Sci., 2021, 602, 627–635 CrossRef CAS PubMed.
- S. N. Faisal, C. M. Subramaniyam, E. Haque, M. M. Islam, N. Noorbehesht, A. K. Roy, M. S. Islam, H. K. Liu, S. X. Dou, A. T. Harris and A. I. Minett, ACS Appl. Energy Mater., 2018, 8b00845 CrossRef.
- A. Abdul Razzaq, Y. Yao, R. Shah, P. Qi, L. Miao, M. Chen, X. Zhao, Y. Peng and Z. Deng, Energy Storage Materials, 2019, 16, 194–202 CrossRef.
- K. J. Hughes, K. A. Iyer, R. E. Bird, J. Ivanov, S. Banerjee, G. Georges and Q. A. Zhou, ACS Appl. Nano Mater., 2024, 7, 18695–18713 CrossRef CAS.
- R. P. Wijitwongwan, S. G. Intasa-ard and M. Ogawa, ChemEngineering, 2019, 3, 68 CrossRef CAS.
- M. Manikandan, T. Prasankumar, D. Murugesan, M. Dinesh, V. Vignesh, R. Govindaraj and P. Ramasamy, in Nanostructured Materials for Energy Storage, Wiley, 2024, pp. 1143–1187 Search PubMed.
- X. Wu, in Flexible Supercapacitors, Wiley, 2022, pp. 157–178 Search PubMed.
- Y.-S. Lee, J. K. Alagarasan, K. Dasha Kumar, T. Ramachandran, H.-J. Kim, M. S. Khan, Y. Anil Kumar, M. Lee and S. Kim, J. Phys. Chem. Solids, 2024, 192, 112114 CrossRef CAS.
- N. Parveen, J. Energy Storage, 2023, 68, 107762 CrossRef.
- S. Chondath, L. Bansal, D. K. Rath, N. Ahlawat, B. Sahu, S. Tiwari, A. Chaudhary and R. Kumar, Sustainable Energy Fuels, 2025, 9, 3304–3314 RSC.
- J. J. William, L. Chitra, B. Saravanakumar, K. Vignesh, P. Rathineshwaran and T. Praveen Kumar, ACS Sustainable Resour. Manage., 2025, 2, 402–412 CrossRef CAS.
- S. Jayakumar, P. C. Santhosh, M. M. Mohideen and A. V. Radhamani, J. Alloys Compd., 2024, 976, 173170 CrossRef CAS.
- Q. Qin, T. Wang, Z. Li, G. Zhang, H. Jang, L. Hou, Y. Wang, M. Gyu Kim, S. Liu and X. Liu, J. Energy Chem., 2024, 88, 94–102 CrossRef CAS.
- M. Zhang, H. Zheng, H. Zhu, Z. Xu, R. Liu, J. Chen, Q. Song, X. Song, J. Wu, C. Zhang and H. Cui, Vacuum, 2020, 176, 109315 CrossRef CAS.
- S. Kour, S. Tanwar and A. L. Sharma, J. Alloys Compd., 2022, 910, 164834 CrossRef CAS.
- G. Zhang, J. Zhang, H. Yang and X. Li, Mater. Lett., 2022, 306, 130858 CrossRef CAS.
- X. Dai, M. Zhang, T. Li, X. Cui, Y. Shi, X. Zhu, P. Wangyang, D. Yang and J. Li, Vacuum, 2022, 195, 110692 CrossRef CAS.
- X. Su, Z. Liang, Q. He, Y. Guo, G. Luo, S. Han and L. Yu, Nanotechnology, 2024, 35, 265402 CrossRef CAS PubMed.
- H. Zhang, S. Geng, M. Ouyang, M. Mao, F. Xie and D. J. Riley, Small, 2022, 18(9), 2106391 CrossRef CAS PubMed.
- C. R. Babu, A. V. Avani, T. S. Xavier, M. Tomy, S. Shaji and E. I. Anila, J. Energy Storage, 2024, 80, 110382 CrossRef.
- N. Joseph, P. M. Shafi, J. S. Sethulakshmi, R. Karthik, A. C. Bose and J.-J. Shim, Electrochim. Acta, 2021, 399, 139392 CrossRef CAS.
- M. Bulla, V. Kumar, R. Devi, S. Kumar, A. K. Sisodiya, R. Dahiya and A. K. Mishra, Sci. Rep., 2024, 14, 7389 CrossRef CAS PubMed.
- X. Li, J. Li, Y. Zhang and P. Zhao, Colloids Surf., A, 2021, 622, 126653 CrossRef CAS.
- M. Zhi, C. Xiang, J. Li, M. Li and N. Wu, Nanoscale, 2013, 5, 72–88 RSC.
- B. S. Singu, E. S. Goda and K. R. Yoon, J. Ind. Eng. Chem., 2021, 97, 239–249 CrossRef CAS.
- T. Ramachandran, A. I. Mourad, R. K. Raji, R. Krishnapriya, N. Cherupurakal, A. Subhan and Y. Al-Douri, Int. J. Energy Res., 2022, 46, 16823–16838 CrossRef CAS.
- Z. Lin, X. Li, H. Zhang, B. Bin Xu, P. Wasnik, H. Li, M. V. Singh, Y. Ma, T. Li and Z. Guo, Inorg. Chem. Front., 2023, 10, 4358–4392 RSC.
- X. Gao, P. Wang, Z. Pan, J. P. Claverie and J. Wang, ChemSusChem, 2020, 13, 1226–1254 CrossRef CAS PubMed.
- C. Jing, B. Dong and Y. Zhang, Energy Environ. Mater., 2020, 3, 346–379 CrossRef CAS.
- K. Li, H. Teng, X. Dai, Y. Wang, D. Wang, X. Zhang, Y. Yao, X. Liu, L. Feng, J. Rao and Y. Zhang, CrystEngComm, 2022, 24, 2081–2088 RSC.
- R. Ramachandran, Y. Lan, Z. X. Xu and F. Wang, ACS Appl. Energy Mater., 2020, 3, 6633–6643 CrossRef CAS.
- L. Zhang, P. Cai, Z. Wei, T. Liu, J. Yu, A. A. Al-Ghamdi and S. Wageh, J. Colloid Interface Sci., 2021, 588, 637–645 CrossRef CAS PubMed.
- R. Luo, Q. Yang, Y. Liu, L. Sun, C. Wang, M. Chen and W. Shi, Inorg. Chem. Front., 2023, 10, 192–200 RSC.
- T. Periyasamy, S. P. Asrafali, S.-C. Kim and J. Lee, Polymers, 2024, 16, 2005 CrossRef CAS PubMed.
- S. Goswami, S. Nandy, E. Fortunato and R. Martins, J. Solid State Chem., 2023, 317, 123679 CrossRef CAS.
- N. K and C. S. Rout, RSC Adv., 2021, 11, 5659–5697 RSC.
- Q. Wang, J. Li, D. Wang, J. Niu, P. Du, J. Liu and P. Liu, Electrochim. Acta, 2020, 349, 136348 CrossRef CAS.
- M. Beygisangchin, S. Abdul Rashid, S. Shafie, A. R. Sadrolhosseini and H. N. Lim, Polymers, 2021, 13, 2003 CrossRef CAS PubMed.
- M. L. Mota, A. Carrillo, A. J. Verdugo, A. Olivas, J. M. Guerrero, E. C. De la Cruz and N. Noriega Ramírez, Molecules, 2019, 24, 1621 CrossRef CAS PubMed.
- A. Chellachamy Anbalagan and S. N. Sawant, Polymer, 2016, 87, 129–137 CrossRef CAS.
- J. Chu, X. Li, Q. Li, J. Ma, B. Wu, X. Wang, R. Zhang, M. Gong and S. Xiong, High Perform. Polym., 2020, 32, 258–267 CrossRef CAS.
- Y. Zhao, S. Xu, L. Chang, W. He, W. Wu, K. Wang, M. Wang and D. Li, Mater. Lett., 2021, 286, 129207 CrossRef CAS.
- T. P. Anandhu, R. R. Mohan, J. Cherusseri, R. R and S. J. Varma, Electrochim. Acta, 2022, 425, 140740 CrossRef CAS.
- D. J. Ahirrao, A. K. Pal, V. Singh and N. Jha, J. Mater. Sci. Technol., 2021, 88, 168–182 CrossRef CAS.
- A. K. Ghasemi, M. Ghorbani, M. S. Lashkenari and N. Nasiri, Electrochim. Acta, 2023, 439, 141685 CrossRef CAS.
- Z. Zhao, Z. Liu, Q. Zhong, Y. Qin, A. Xu, W. Li and J. Shi, ACS Appl. Energy Mater., 2020, 3, 8774–8785 CrossRef CAS.
- M. S. Zoromba, M. H. Abdel-Aziz, M. Bassyouni, A. M. Abusorrah, A. Attar, N. Baghdadi and N. Salah, Phys. E, 2021, 134, 114889 CrossRef CAS.
- T. Samwang, N. M. Watanabe, Y. Okamoto, S. Srinives and H. Umakoshi, ACS Omega, 2023, 8, 48946–48957 CrossRef CAS PubMed.
- Y. Wang, H. Wang, W. Zhang, G. Fei, K. Shu, L. Sun, S. Tian, H. Niu, M. Wang, G. Hu and Y. Duan, ACS Appl. Polym. Mater., 2023, 5, 1254–1263 CrossRef CAS.
- S. S. Bhoyar, V. B. Sangale, M. A. Desai, P. A. Khot, S. K. Pardeshi, S. D. Sartale and K. M. Kodam, J. Power Sources, 2024, 607, 234599 CrossRef CAS.
- A. A. Iurchenkova, T. Kallio and E. O. Fedorovskaya, Electrochim. Acta, 2021, 391, 138892 CrossRef CAS.
- J. Parayangattil Jyothibasu, M.-Z. Chen and R.-H. Lee, ACS Omega, 2020, 5, 6441–6451 CrossRef CAS PubMed.
- J. El Nady, A. Shokry, M. Khalil, S. Ebrahim, A. M. Elshaer and M. Anas, Sci. Rep., 2022, 12, 3611 CrossRef CAS PubMed.
- J. Ding, T. Xia, Q. Xia, G. Li and Y. Qu, ACS Appl. Nano Mater., 2022, 5, 654–661 CrossRef CAS.
- F. Dang, Y. Cai and P. Yang, ACS Appl. Energy Mater., 2024, 7, 5832–5838 CrossRef CAS.
- Y. Zhang, A. Xu, Y. Yu, S. Ye, Z. Zhao, W. Cao, S. Zhang and Y. Qin, Langmuir, 2024, 40, 1399–1407 CrossRef CAS PubMed.
- D. Li, A. Yadav, H. Zhou, K. Roy, P. Thanasekaran and C. Lee, Glob. Chall., 2023, 2300244, DOI:10.1002/gch2.202300244.
- R. Zhang, Z. Zhang, J. Jiang and H. Pang, J. Energy Storage, 2023, 65, 107217 CrossRef.
- L. Wang, Q. He, F. Xiao, L. Yang, Y. Jiang, R. Su, P. He, H. Lei, B. Jia and B. Tang, Electrochim. Acta, 2024, 484, 144082 CrossRef CAS.
- D. Wang, H. Yao, J. Ye, Y. Gao, H. Cong and B. Yu, Small, 2024, 20(47), 2404350 CrossRef CAS PubMed.
- V. Shrivastav, S. Sundriyal, K. Kim, R. K. Sinha, U. K. Tiwari and A. Deep, Int. J. Energy Res., 2020, 44, 6269–6284 CrossRef CAS.
- K.-B. Wang, Q. Xun and Q. Zhang, EnergyChem, 2020, 2, 100025 CrossRef.
- K. Wang, R. Bi, M. Huang, B. Lv, H. Wang, C. Li, H. Wu and Q. Zhang, Inorg. Chem., 2020, 59, 6808–6814 CrossRef CAS PubMed.
- Z. Cao, R. Momen, S. Tao, D. Xiong, Z. Song, X. Xiao, W. Deng, H. Hou, S. Yasar, S. Altin, F. Bulut, G. Zou and X. Ji, Nano-Micro Lett., 2022, 14, 181 CrossRef CAS PubMed.
- W. He, X.-B. Mu, J. Lei, W. Yuan, P. Zhang and Q.-G. Zhai, Cryst. Growth Des., 2023, 23, 4346–4356 CrossRef CAS.
- K. Jayaramulu, M. Horn, A. Schneemann, H. Saini, A. Bakandritsos, V. Ranc, M. Petr, V. Stavila, C. Narayana, B. Scheibe, Š. Kment, M. Otyepka, N. Motta, D. Dubal, R. Zbořil and R. A. Fischer, Adv. Mater., 2021, 33(4), 2004560 CrossRef CAS PubMed.
- K. Wang, Y. Guo and Q. Zhang, Small Struct., 2021, 2100115, DOI:10.1002/sstr.202100115.
- N. Muzaffar, A. M. Afzal, H. H. Hegazy and M. W. Iqbal, J. Energy Storage, 2023, 64, 107142 CrossRef.
- P. G. Raje, S. R. Gurav, M. R. Waikar, A. S. Rasal, J.-Y. Chang and R. G. Sonkawade, J. Energy Storage, 2022, 56, 105700 CrossRef.
- Raissa, N. L. W. Septiani, S. Wustoni, F. Failamani, N. Wehbe, M. Eguchi, H. Nara, S. Inal, V. Suendo and B. Yuliarto, J. Power Sources, 2024, 603, 234423 CrossRef CAS.
- T.-L. Meng, Y.-X. Ma, J.-W. Wang, X.-D. Zhang, L. Lei, J. Li and F. Ran, J. Mater. Sci.: Mater. Electron., 2024, 35, 1592 CrossRef CAS.
- H. Guo, H. Zhang, N. Wu, Z. Pan, C. Li, Y. Chen, Y. Cao and W. Yang, J. Alloys Compd., 2023, 934, 167779 CrossRef CAS.
- H. Lavillunière, T.-N. Pham-Truong, T.-K.-L. Nguyen, C. Vancaeyzeele and P.-H. Aubert, ACS Appl. Energy Mater., 2024, 7, 1723–1734 CrossRef.
- S. Salehi, M. H. Ehsani and M. Aghazadeh, J. Alloys Compd., 2023, 940, 168885 CrossRef CAS.
- B. Nath, in Synthesis of Metal-Organic Frameworks via Water-Based Routes, Elsevier, 2024, pp. 93–120 Search PubMed.
- J. Liu, Q. Xia, L. Wang, Q. Hu, N. M. Shinde and A. Zhou, ACS Appl. Mater. Interfaces, 2024, 49380–49391, DOI:10.1021/acsami.4c09616.
- N. Wang, Y. Wei, M. Chang, J. Liu and J.-X. Wang, ACS Appl. Mater. Interfaces, 2022, 14, 10712–10720 CrossRef CAS PubMed.
- R. Batool, S. D. Dhas, A. C. Mendhe, A. Ghazal, I. Kim and D. Kim, J. Mater. Chem. A, 2024, 12, 21230–21242 RSC.
- S. Saha, M. Kandasamy, D. Potphode, C. S. Sharma, B. Chakraborty, N. Hameed and N. Salim, J. Energy Storage, 2024, 99, 113294 CrossRef.
- C. Huang, W. Sun, Y. Jin, Q. Guo, D. Mücke, X. Chu, Z. Liao, N. Chandrasekhar, X. Huang, Y. Lu, G. Chen, M. Wang, J. Liu, G. Zhang, M. Yu, H. Qi, U. Kaiser, G. Xu, X. Feng and R. Dong, Angew. Chem., Int. Ed., 2024, e202313591, DOI:10.1002/anie.202313591.
- D. K. Singha, R. I. Mohanty, P. Bhanja and B. K. Jena, Mater. Adv., 2023, 4, 4679–4706 RSC.
- M. Zhang, Y. Shan, Q. Kong and H. Pang, FlatChem, 2022, 32, 100332 CrossRef CAS.
- A. H. Anwer, M. M. Zubair, F. Mashkoor, A. Benamor, I. Hasan, M. Shoeb and C. Jeong, J. Alloys Compd., 2024, 970, 172512 CrossRef CAS.
- N. Hussain, Z. Abbas, K. Nabeela and S. M. Mobin, J. Mater. Chem. A, 2024, 12, 17642–17650 RSC.
- D. Acharya, A. Muthurasu, T. H. Ko, R. M. Bhattarai, T. Kim, S.-H. Chae, S. Saidin, K. Chhetri and H. Y. Kim, ACS Appl. Energy Mater., 2023, 6, 9196–9206 CrossRef CAS.
- D. Liu, D. Zou, H. Zhu and J. Zhang, Small, 2018, 14(37), 1801454 CrossRef PubMed.
- A. Hayat, S. Rauf, B. Al Alwan, A. El Jery, N. Almuqati, S. Melhi, M. A. Amin, Y. Al-Hadeethi, M. Sohail, Y. Orooji and W. Lv, Materials Today Energy, 2024, 41, 101542 CrossRef CAS.
- C. Pei, M. S. Choi, X. Yu, H. Xue, B. Y. Xia and H. S. Park, J. Mater. Chem. A, 2021, 9, 8832–8869 RSC.
- H. Vardhan, A. Nafady, A. M. Al-Enizi and S. Ma, Nanoscale, 2019, 11, 21679–21708 RSC.
- W. Lee, H. Li, Z. Du and D. Feng, Chem. Soc. Rev., 2024, 53, 8182–8201 RSC.
- T. Li, W.-D. Zhang, Y. Liu, Y. Li, C. Cheng, H. Zhu, X. Yan, Z. Li and Z.-G. Gu, J. Mater. Chem. A, 2019, 7, 19676–19681 RSC.
- Z. Wu, Y. Liu, C. Zou and W. Guo, Next Energy, 2023, 1, 100041 CrossRef.
- P. Sudhakar, A. K. Gupta, D. B. Cordes and E. Zysman-Colman, Chem. Sci., 2024, 15, 545–554 RSC.
- C. G. Gruber, L. Frey, R. Guntermann, D. D. Medina and E. Cortés, Nature, 2024, 630, 872–877 CrossRef CAS PubMed.
- S. Liu, L. Yao, Y. Lu, X. Hua, J. Liu, Z. Yang, H. Wei and Y. Mai, Mater. Lett., 2019, 236, 354–357 CrossRef CAS.
- L. K. Ritchie, A. Trewin, A. Reguera-Galan, T. Hasell and A. I. Cooper, Microporous Mesoporous Mater., 2010, 132, 132–136 CrossRef CAS.
- P. J. Waller, F. Gándara and O. M. Yaghi, Acc. Chem. Res., 2015, 48, 3053–3063 CrossRef CAS PubMed.
- J. Li, X. Jing, Q. Li, S. Li, X. Gao, X. Feng and B. Wang, Chem. Soc. Rev., 2020, 49, 3565–3604 RSC.
- U. Shahzad, H. M. Marwani, M. Saeed, A. M. Asiri, M. R. Repon, R. H. Althomali and M. M. Rahman, Chem. Rec., 2023, e202300285, DOI:10.1002/tcr.202300285.
- T. K. Mandal, N. Parvin, K. Mishra, S. Mohandoss and Y. R. Lee, Microchim. Acta, 2019, 186, 833 CrossRef CAS PubMed.
- N. Parvin, T. K. Mandal and S. W. Joo, J. Mater. Chem. C, 2023, 11, 16398–16410 RSC.
- K. Liu, H. Qi, R. Dong, R. Shivhare, M. Addicoat, T. Zhang, H. Sahabudeen, T. Heine, S. Mannsfeld, U. Kaiser, Z. Zheng and X. Feng, Nat. Chem., 2019, 11, 994–1000 CrossRef CAS PubMed.
- M. Ibrahim, H. N. Abdelhamid, A. M. Abuelftooh, S. G. Mohamed, Z. Wen and X. Sun, J. Energy Storage, 2022, 55, 105375 CrossRef.
- M. Ghaderi, H. Bi and K. Dam-Johansen, Adv. Colloid Interface Sci., 2024, 323, 103055 CrossRef CAS PubMed.
- C. Wang, Z. Zhang, Y. Zhu, C. Yang, J. Wu and W. Hu, Adv. Mater., 2022, 2102290, DOI:10.1002/adma.202102290.
- H. Ben, W. Du, J. Zhao, Y. Wang, Y. Wu, F. Lin, Y. Lei, S. Zhou, J. Pu, M. Ye, J. Du, X. Zhang, Y. Fan and L. Lv, Coord. Chem. Rev., 2024, 517, 216003 CrossRef CAS.
- K. Li, X. Quan and B. Yan, Sens. Actuators, B, 2023, 392, 134078 CrossRef CAS.
- B. Pal, S. Yang, S. Ramesh, V. Thangadurai and R. Jose, Nanoscale Adv., 2019, 1, 3807–3835 RSC.
- K. Nasrin, S. Gokulnath, M. Karnan, K. Subramani and M. Sathish, Energy Fuels, 2021, 35, 6465–6482 CrossRef CAS.
- G. Wang, L. Zhang and J. Zhang, Chem. Soc. Rev., 2012, 41, 797–828 RSC.
- D. Majumdar, in Flexible Supercapacitor Nanoarchitectonics, Wiley, 2021, pp. 349–412 Search PubMed.
- P. Krishnan and V. Biju, Bull. Mater. Sci., 2021, 44, 288 CrossRef CAS.
- X. Wang, Z. You and D. Ruan, Chin. J. Chem., 2006, 24, 1126–1132 CrossRef CAS.
- M. Zhang, H. Fan, Y. Gao, N. Zhao, C. Wang, J. Ma, L. Ma, A. K. Yadav, W. Wang, W. S. Vincent Lee, T. Xiong, J. Xue and Z. Xia, ACS Appl. Mater. Interfaces, 2020, 12, 17659–17668 CrossRef CAS PubMed.
- H. Zhang, X. Liu, H. Li, I. Hasa and S. Passerini, Angew. Chem., Int. Ed., 2021, 60, 598–616 CrossRef CAS PubMed.
- K. L. Van Aken, M. Beidaghi and Y. Gogotsi, Angew. Chem., 2015, 127, 4888–4891 CrossRef.
- Y. Wang, K. Xue, C. Yan, Y. Li, X. Zhang, K. Su, P. Ma, S. Wan and J. Lang, Batteries, 2024, 10, 54 CrossRef CAS.
- P. Wang, Y. Zhong, J. Wang, H. Zhou, G. Sun, X. Sui and Z. Wang, Adv. Funct. Mater., 2024, 2414563, DOI:10.1002/adfm.202414563.
- C. Agarwal and A. K. Pandey, Environ. Sci.:Adv., 2023, 2, 1306–1339 CAS.
- M. Zhang, W. Wang, X. Liang, C. Li, W. Deng, H. Chen and R. Li, Chin. Chem. Lett., 2021, 32, 2217–2221 CrossRef CAS.
- S. Chen, M. Zhang, P. Zou, B. Sun and S. Tao, Energy Environ. Sci., 2022, 15, 1805–1839 RSC.
- H. Rashid Khan and A. Latif Ahmad, J. Ind. Eng. Chem., 2025, 46–66, DOI:10.1016/j.jiec.2024.07.014.
- R. Zhang, X. Shi, O. C. Esan and L. An, Glob. Chall., 2022, 6(12), 2200050 CrossRef PubMed.
- I. F. Martínez-Galmiche, N. Ramírez-Corona, C. Conde-Mejía, K. B. Sánchez-Sánchez, R. Gani and A. Jiménez-Gutiérrez, Chem. Eng. Res. Des., 2022, 188, 238–248 CrossRef.
- A. Rao, T. Bhakyapaibul, J. Rovey, D. A. Levin and H. Chew, in AIAA Scitech 2023 Forum, American Institute of Aeronautics and Astronautics, Reston, Virginia, 2023 Search PubMed.
- J.-C. Liu, Z.-H. Huang and T.-Y. Ma, Small Struct., 2020, 2000020, DOI:10.1002/sstr.202000020.
- R. R. Galimzyanov, S. V. Stakhanova, I. S. Krechetov, A. T. Kalashnik, M. V. Astakhov, A. V. Lisitsin, A. Y. Rychagov, T. R. Galimzyanov and F. S. Tabarov, J. Power Sources, 2021, 495, 229442 CrossRef CAS.
- P. W. Ruch, D. Cericola, A. Foelske-Schmitz, R. Kötz and A. Wokaun, Electrochim. Acta, 2010, 55, 4412–4420 CrossRef CAS.
- C. Lian, H. Liu, C. Li and J. Wu, AIChE J., 2019, 65, 804–810 CrossRef CAS.
- S. S. de Jesus and R. Maciel Filho, Renewable Sustainable Energy Rev., 2022, 157, 112039 CrossRef CAS.
- S. Shahzad, A. Shah, E. Kowsari, F. J. Iftikhar, A. Nawab, B. Piro, M. S. Akhter, U. A. Rana and Y. Zou, Glob. Chall., 2018, 1800023, DOI:10.1002/gch2.201800023.
- H. Ohno, Bull. Chem. Soc. Jpn., 2006, 79, 1665–1680 CrossRef CAS.
- W.-L. Yuan, X. Yang, L. He, Y. Xue, S. Qin and G.-H. Tao, Front. Chem., 2018, 6, 00059 CrossRef PubMed.
- P. C. Marr and A. C. Marr, Green Chem., 2016, 18, 105–128 RSC.
- J. Castillo, A. Santiago, X. Judez, I. Garbayo, J. A. Coca Clemente, M. C. Morant-Miñana, A. Villaverde, J. A. González-Marcos, H. Zhang, M. Armand and C. Li, Chem. Mater., 2021, 33, 8812–8821 CrossRef CAS.
- J. Pereira, R. Souza, A. Moreira and A. Moita, Ionics, 2024, 30, 4343–4385 CrossRef CAS.
- M. J. Lee, D. O. Shin, J. Y. Kim, J. Oh, S. H. Kang, J. Kim, K. M. Kim, Y. M. Lee, S. O. Kim and Y.-G. Lee, Energy Storage Materials, 2021, 37, 306–314 CrossRef.
- T. Bertaglia, C. M. Costa, S. Lanceros-Méndez and F. N. Crespilho, Mater. Adv., 2024, 5, 7534–7547 RSC.
- A. Kityk, V. Pavlik and M. Hnatko, Adv. Colloid Interface Sci., 2024, 334, 103310 CrossRef CAS PubMed.
- X. Qiu, N. Wang, Z. Wang, F. Wang and Y. Wang, Angew. Chem., Int. Ed., 2021, 60, 9610–9617 CrossRef CAS PubMed.
- H. Zhou, C. Liu, J.-C. Wu, M. Liu, D. Zhang, H. Song, X. Zhang, H. Gao, J. Yang and D. Chen, J. Mater. Chem. A, 2019, 7, 9708–9715 RSC.
- F. Philippi and T. Welton, Phys. Chem. Chem. Phys., 2021, 23, 6993–7021 RSC.
- Y.-Q. Zhang, G.-C. Xu and M. Li, Dalton Trans., 2021, 50, 12287–12291 RSC.
- M. Shamsipur, A. A. M. Beigi, M. Teymouri, S. M. Pourmortazavi and M. Irandoust, J. Mol. Liq., 2010, 157, 43–50 CrossRef CAS.
- R. P. Swatloski, J. D. Holbrey and R. D. Rogers, Green Chem., 2003, 5, 361 RSC.
- S. Pan, M. Yao, J. Zhang, B. Li, C. Xing, X. Song, P. Su and H. Zhang, Front. Chem., 2020, 261, DOI:10.3389/fchem.2020.00261.
- H. Niu, L. Wang, P. Guan, N. Zhang, C. Yan, M. Ding, X. Guo, T. Huang and X. Hu, J. Energy Storage, 2021, 40, 102659 CrossRef.
- A. Ray and B. Saruhan, Materials, 2021, 14, 2942 CrossRef CAS PubMed.
- H. Liu and H. Yu, J. Mater. Sci. Technol., 2019, 35, 674–686 CrossRef CAS.
- S. Xue, Y. Liu, K. Hou, M. He and X. Liu, J. Power Sources, 2024, 615, 235097 CrossRef CAS.
- I. Jhanji, Int. J. Eng. Sci., 2023, 11, 57–67 Search PubMed.
- S. M. S. Hussain, A. A. Adewunmi, O. S. Alade, M. Murtaza, A. Mahboob, H. J. Khan, M. Mahmoud and M. S. Kamal, J. Taiwan Inst. Chem. Eng., 2023, 153, 105195 CrossRef CAS.
- M. Mirzaei-Saatlo, E. Asghari, H. Shekaari, B. Pollet and R. Vinodh, Electrochim. Acta, 2024, 474, 143499 CrossRef CAS.
- C. Bodin, B. Gelinas, J. Deng, K. Pithaksinsakul, Y. Zhu, D. Rochefort and O. Fontaine, Curr. Opin. Colloid Interface Sci., 2023, 64, 101677 CrossRef CAS.
- G. Yang, Y. Song, Q. Wang, L. Zhang and L. Deng, Mater. Des., 2020, 190, 108563 CrossRef CAS.
- K. Matuszek, S. L. Piper, A. Brzęczek-Szafran, B. Roy, S. Saher, J. M. Pringle and D. R. MacFarlane, Adv. Mater., 2024, 2313023, DOI:10.1002/adma.202313023.
- A. Dutta, W. Millar, D. S. Silvester and T. Banerjee, New J. Chem., 2024, 48, 17787–17801 RSC.
- K. Sheoran, H. Kaur, S. S. Siwal and V. K. Thakur, Adv. Energy Sustainability Res., 2023, 2300021, DOI:10.1002/aesr.202300021.
- P. E. Lokhande, V. Kadam, C. Jagtap, D. D. Mohite, R. Udayabhaskar, P. V. Thangavelu, S. M. H. Qaid and A. Kumar, Energy Technol., 2024, 2400995, DOI:10.1002/ente.202400995.
- X. Chen, L. Yi, J. Liu, Z. Luo, Y. Shen and X. Wang, J. Power Sources, 2024, 603, 234433 CrossRef CAS.
- D. Kasprzak and J. Liu, Sustainable Mater. Technol., 2023, 38, e00726 CrossRef CAS.
- S. Beil, M. Markiewicz, C. S. Pereira, P. Stepnowski, J. Thöming and S. Stolte, Chem. Rev., 2021, 121, 13132–13173 CrossRef CAS PubMed.
- S. I. Wong, H. Lin, T. Ma, J. Sunarso, B. T. Wong and B. Jia, Mater. Rep.: Energy, 2022, 2, 100093 CAS.
- S. Dalvand, A. Foroozandeh, A. Heydarian, F. S. Nasab, M. Omidvar, N. Yazdanfar and A. Asghari, Ionics, 2024, 30, 1857–1870 CrossRef CAS.
- S. M. A. Nayem, S. Islam, S. S. Shah, N. Sultana, M. N. Shaikh, A. Aziz and A. J. S. Ahammad, in Biomass-Based Supercapacitors, Wiley, 2023, pp. 383–401 Search PubMed.
- H. G. Redda, Y. Nikodimos, W.-N. Su, R.-S. Chen, S.-K. Jiang, L. H. Abrha, T. M. Hagos, H. K. Bezabh, H. H. Weldeyohannes and B. J. Hwang, Mater. Today Commun., 2021, 26, 102102 CrossRef CAS.
- M. F. Aziz, M. H. Buraidah, M. A. Careem and A. K. Arof, Electrochim. Acta, 2015, 182, 217–223 CrossRef CAS.
- M. M. Amaral, R. Venâncio, A. C. Peterlevitz and H. Zanin, J. Energy Chem., 2022, 67, 697–717 CrossRef CAS.
- Y.-H. Lin, W.-T. Huang, Y.-T. Huang, Y.-N. Jhang, T.-T. Shih, M. Yılmaz and M.-J. Deng, Polymers, 2023, 15, 3438 CrossRef CAS PubMed.
- S. Kuang, Z. Huang, Y. Huang, D. Shao, X. Jia, B. Feng, Y. Yan, H. Hu and X. Yu, J. Alloys Compd., 2022, 918, 165688 CrossRef CAS.
- T. K. L. Nguyen and T.-N. Pham-Truong, Polymers, 2024, 16, 2506 CrossRef CAS PubMed.
- K. Gong, H. Lee, Y. Choi, G. Jung, K. Keum, J. W. Kim and J. S. Ha, Electrochim. Acta, 2024, 475, 143585 CrossRef CAS.
- E. C. Fluker, S. Pathreeker and I. D. Hosein, J. Phys. Chem. C, 2023, 127, 16579–16587 CrossRef CAS PubMed.
- J. P. Serra, A. Fidalgo-Marijuan, J. C. Barbosa, D. M. Correia, R. Gonçalves, J. M. Porro, S. Lanceros-Mendez and C. M. Costa, ACS Appl. Polym. Mater., 2022, 4, 5909–5919 CrossRef CAS PubMed.
- A. La Monaca, A. Paolella, A. Guerfi, F. Rosei and K. Zaghib, Electrochem. Commun., 2019, 104, 106483 CrossRef CAS.
- G. Zheng, Z. Zeng, Y. Chen, X. Wang, D. Sun and C. Cui, J. Energy Storage, 2024, 86, 111285 CrossRef.
- M. S. Ahmed, M. Islam, B. Raut, S. Yun, H. Y. Kim and K.-W. Nam, Gels, 2024, 10, 563 CrossRef CAS PubMed.
- Z. Hong, H. Tian, Z. Fang, Y. Luo, H. Wu, F. Zhao, Q. Li, S. Fan and J. Wang, ChemElectroChem, 2024, e202300759, DOI:10.1002/celc.202300759.
- C. Yonchai, P. Kidkhunthod, S. Siriroj, J. Padchasri, S. Sonsupap, S. Nijpanich, P. Pakawanit, N. Chanlek, S. Maensiri and S. M. Smith, Materialia, 2024, 35, 102114 CrossRef CAS.
- R. Whba, M. S. Su'ait, F. Whba and A. Ahmad, J. Power Sources, 2024, 606, 234539 CrossRef CAS.
- K. Daems, P. Yadav, K. B. Dermenci, J. Van Mierlo and M. Berecibar, Renewable Sustainable Energy Rev., 2024, 191, 114136 CrossRef CAS.
- W. Liu, Z. Li, F. Pan, Q. He and Q. Zhang, RSC Adv., 2023, 13, 34652–34659 RSC.
- A. D. Shuaibu, S. S. Shah, A. S. Alzahrani and M. A. Aziz, J. Energy Storage, 2025, 107, 114851 CrossRef.
- Z. Yang, W. Ye, X. Lei, D. Schweigert, H.-K. Kwon and A. Khajeh, npj Comput. Mater., 2024, 10, 296 CrossRef CAS.
- S. Borah, A. K. Guha, L. Saikia and M. Deka, J. Alloys Compd., 2021, 886, 161173 CrossRef CAS.
- S. Huo, L. Sheng, W. Xue, L. Wang, H. Xu, H. Zhang and X. He, InfoMat, 2023, e12394, DOI:10.1002/inf2.12394.
- Z. Li, J. Fu, X. Zhou, S. Gui, L. Wei, H. Yang, H. Li and X. Guo, Adv. Sci., 2023, 2201718, DOI:10.1002/advs.202201718.
- K. Yang, Q. Fan, Y. Zhang, G. Ren, X. Huang and P. Fu, RSC Adv., 2024, 14, 1123–1133 RSC.
- X. Tian, X. Cheng, S. Liao, J. Chen, P. Lv and Q. Wei, ACS Appl. Mater. Interfaces, 2023, 52415–52426, DOI:10.1021/acsami.3c10671.
- J. Chu, X. Li, Q. Li, J. Ma, B. Wu, X. Wang, R. Zhang, M. Gong and S. Xiong, High Perform. Polym., 2020, 32, 258–267 CrossRef CAS.
- J. Yang, J. Peng, Y. Lei, Y. Tang, P. Liu, J. Zeng, C. Yi, Y. Shen, L. Zheng and X. Wang, ACS Appl. Energy Mater., 2024, 7, 469–478 CrossRef.
- N. Boulanger, A. V. Talyzin, S. Xiong, M. Hultberg and A. Grimm, Colloids Surf., A, 2024, 680, 132684 CrossRef CAS.
- B. H. Poornima, M. Marimuthu and T. Vijayakumar, Surf. Interfaces, 2024, 44, 103665 CrossRef CAS.
- Z. Wang, S. Pan, B. Wang, J. Qi, L. Tang and L. Liu, J. Electrochem. Sci. Technol., 2021, 12, 346–357 CrossRef CAS.
- Y. K. Sonia and S. K. Meher, Energy Fuels, 2023, 37, 4010–4025 CrossRef CAS.
- Z. Karami, F. Hekmat, M. Chougale, S. Shahrokhian and D. P. Dubal, Small, 2023, 2404506, DOI:10.1002/smll.202404506.
- R. Ramachandran, Y. Lan, Z.-X. Xu and F. Wang, ACS Appl. Energy Mater., 2020, 3, 6633–6643 CrossRef CAS.
- Y. Li, P. Kamdem and X.-J. Jin, J. Alloys Compd., 2021, 850, 156608 CrossRef CAS.
- S. Wu, Y. Chen, T. Jiao, J. Zhou, J. Cheng, B. Liu, S. Yang, K. Zhang and W. Zhang, Adv. Energy Mater., 2019, 1902915, DOI:10.1002/aenm.201902915.
- L. Xu, Y. Liu, Z. Ding, X. Xu, X. Liu, Z. Gong, J. Li, T. Lu and L. Pan, Small, 2023, 2307843, DOI:10.1002/smll.202307843.
- N. An, Z. Guo, C. Guo, M. Wei, D. Sun, Y. He, W. Li, L. Zhou, Z. Hu and X. Dong, Chem. Eng. J., 2023, 458, 141434 CrossRef CAS.
- X. Zhu, Y. Zhang, Z. Man, W. Lu, W. Chen, J. Xu, N. Bao, W. Chen and G. Wu, Adv. Mater., 2023, 2307186, DOI:10.1002/adma.202307186.
- B. Qin, H. Ma, M. Hossain, M. Zhong, Q. Xia, B. Li and X. Duan, Chem. Mater., 2020, 32, 10321–10347 CrossRef CAS.
- P. Forouzandeh, P. Ganguly, R. Dahiya and S. C. Pillai, J. Power Sources, 2022, 519, 230744 CrossRef CAS.
- Z. Shi, H. Ci, X. Yang, Z. Liu and J. Sun, ACS Nano, 2022, 16, 11646–11675 CrossRef CAS PubMed.
- S. Sahoo, G. Sahoo, S. M. Jeong and C. S. Rout, J. Energy Storage, 2022, 53, 105212 CrossRef.
- M. Bahri, S. H. Gebre, M. A. Elaguech, F. T. Dajan, M. G. Sendeku, C. Tlili and D. Wang, Coord. Chem. Rev., 2023, 475, 214910 CrossRef CAS.
- M. J. Madito, K. S. Matshoba, F. U. Ochai-Ejeh, N. Mongwaketsi, C. B. Mtshali, M. Fabiane and N. Manyala, Surf. Coat. Technol., 2020, 383, 125230 CrossRef CAS.
- P. Mouchani, R. Sarraf-Mamoory and H. Aghajani, J. Alloys Compd., 2022, 904, 163895 CrossRef CAS.
- Q. Zhang, F. Yang, C. Zhang, H. Dong, J. Sui, L. Yu, Y. Chen, J. Yu and L. Dong, Appl. Surf. Sci., 2021, 551, 149417 CrossRef CAS.
- T. K. Nguyen, S. H. Yu, J. Yan and D. H. C. Chua, J. Mater. Sci., 2020, 55, 15588–15601 CrossRef CAS.
- X. Zang, Y. Jiang, M. Sanghadasa and L. Lin, Sens. Actuators, A, 2020, 304, 111886 CrossRef CAS.
- Z. Niu, Y. Zhang, Y. Zhang, X. Lu and J. Liu, J. Alloys Compd., 2020, 820, 153114 CrossRef CAS.
- H.-L. Hsu, M. Miah, S. K. Saha, J.-H. Chen, L.-C. Chen and S.-Y. Hsu, Mater. Today Chem., 2021, 22, 100569 CrossRef CAS.
- N. Bardi, T. Giannakopoulou, A. Vavouliotis and C. Trapalis, ACS Appl. Nano Mater., 2020, 3, 10003–10013 CrossRef CAS.
- E. Bertran-Serra, S. Rodriguez-Miguel, Z. Li, Y. Ma, G. Farid, S. Chaitoglou, R. Amade, R. Ospina and J.-L. Andújar, Nanomaterials, 2023, 13, 2533 CrossRef CAS PubMed.
- Z. Wang, J. Chen, S. Sun, Z. Huang, X. Zhang, X. Li and H. Dong, Energy Storage Materials, 2022, 50, 161–185 CrossRef.
- R. Konar and G. D. Nessim, Mater. Adv., 2022, 3, 4471–4488 RSC.
- R. Bhoria, in Graphene and its derivatives – synthesis and applications, IntechOpen, 2019 Search PubMed.
- S. Sopan Mahato, D. Mahata, S. Panda and S. Mahata, in Sol-Gel Method – Recent Advances, IntechOpen, 2023 Search PubMed.
- K. Jang, K. R. Park, C. Bin Mo, S. Kim, J. Jeon, S. Lim, C. Ahn, H. Han, D. Kim, S. H. Lee, K. M. Kim and S. Mhin, Sci. Rep., 2022, 12, 19546 CrossRef CAS PubMed.
- M. Dai, D. Zhao and X. Wu, Chin. Chem. Lett., 2020, 31, 2177–2188 CrossRef CAS.
- D. Navas, S. Fuentes, A. Castro-Alvarez and E. Chavez-Angel, Gels, 2021, 7, 275 CrossRef CAS PubMed.
- D. Bokov, A. Turki Jalil, S. Chupradit, W. Suksatan, M. Javed Ansari, I. H. Shewael, G. H. Valiev and E. Kianfar, Adv. Mater. Sci. Eng., 2021, 21, 5102014 CrossRef.
- S. Sakka, J. Sol-Gel Sci. Technol., 2022, 102, 478–481 CrossRef CAS.
- F. Liu, D. Feng, H. Yang and X. Guo, Sci. Rep., 2020, 10, 4331 CrossRef CAS PubMed.
- G. J. Owens, R. K. Singh, F. Foroutan, M. Alqaysi, C.-M. Han, C. Mahapatra, H.-W. Kim and J. C. Knowles, Prog. Mater. Sci., 2016, 77, 1–79 CrossRef CAS.
- A. De, M. S. Kim, A. Adhikari, R. Patel and S. Kundu, J. Mater. Chem. A, 2024, 12, 19720–19756 RSC.
- F. Putz, A. Waag, C. Balzer, S. Braxmeier, M. S. Elsaesser, L. Ludescher, O. Paris, W. J. Malfait, G. Reichenauer and N. Hüsing, Microporous Mesoporous Mater., 2019, 288, 109578 CrossRef CAS.
- G. Veselov and A. Vedyagin, Materials, 2023, 16, 6566 CrossRef CAS PubMed.
- I. Shaheen, K. S. Ahmad, C. Zequine, R. K. Gupta, A. G. Thomas and M. A. Malik, Energy, 2021, 218, 119502 CrossRef CAS.
- A. Pal, T. Das, S. Ghosh and M. Nandi, Dalton Trans., 2020, 49, 12716–12730 RSC.
- B. Thi Hang, VNU J. Sci.: Math. – Phys., 2023, 39(3) DOI:10.25073/2588-1124/vnumap.4848.
- X. Wu, X. Zhang, X. Wang, C. Zhang, Q. Zhu, A. Du, Z. Zhang and J. Shen, J. Porous Mater., 2022, 29, 87–95 CrossRef CAS.
- K. I. Nargatti, A. R. Subhedar, S. S. Ahankari, A. N. Grace and A. Dufresne, Carbohydr. Polym., 2022, 297, 120039 CrossRef CAS PubMed.
- M. A. Raza, Z. U. Rehman, M. G. Tanvir and M. F. Maqsood, in Renewable Polymers and Polymer-Metal Oxide Composites, Elsevier, 2022, pp. 195–251 Search PubMed.
- B. De, et al., Transition Metal Oxide-/Carbon-/Electronically Conducting Polymer-Based Ternary Composites as Electrode Materials for Supercapacitors, in Handbook of Nanocomposite Supercapacitor Materials II, ed. K. Kar, Springer Series in Materials Science, Springer, Cham, 2020, vol. 302, DOI:10.1007/978-3-030-52359-6_15.
- S. Soltani, N. Khanian, U. Rashid and T. S. Yaw Choong, RSC Adv., 2020, 10, 16431–16456 RSC.
- V. M. Adimule, S. S. Nandi, S. S. Kerur, S. A. Khadapure and S. Chinnam, Top. Catal., 2022 DOI:10.1007/s11244-022-01571-z.
- S. Verma, S. Arya, V. Gupta, S. Mahajan, H. Furukawa and A. Khosla, J. Mater. Res. Technol., 2021, 11, 564–599 CrossRef CAS.
- N. T. Shelke, M. A. Yewale, R. A. Kadam, U. T. Nakate, A. A. Jadhavar and D. K. Shin, Surf. Interfaces, 2023, 43, 103555 CrossRef CAS.
- Y.-Q. Niu, J.-H. Liu, C. Aymonier, S. Fermani, D. Kralj, G. Falini and C.-H. Zhou, Chem. Soc. Rev., 2022, 51, 7883–7943 RSC.
- Z. Wang, L. Yang, C. Xu, J. Cheng, J. Zhao, Q. Huang and C. Yang, Green Carbon, 2023, 1, 193–209 CrossRef.
- M. Ahmed, in Polymer Science and Nanotechnology, Elsevier, 2020, pp. 361–399 Search PubMed.
- V. Harish, M. M. Ansari, D. Tewari, A. B. Yadav, N. Sharma, S. Bawarig, M.-L. García-Betancourt, A. Karatutlu, M. Bechelany and A. Barhoum, J. Taiwan Inst. Chem. Eng., 2023, 149, 105010 CrossRef CAS.
- A. Morales, J. Labidi and P. Gullón, Sci. Total Environ., 2021, 764, 142800 CrossRef CAS PubMed.
- M. H. Marzbali, S. Kundu, P. Halder, S. Patel, I. G. Hakeem, J. Paz-Ferreiro, S. Madapusi, A. Surapaneni and K. Shah, Chemosphere, 2021, 279, 130557 CrossRef CAS PubMed.
- S. D. Dhas, P. S. Maldar, M. D. Patil, A. B. Nagare, M. R. Waikar, R. G. Sonkawade and A. V. Moholkar, Vacuum, 2020, 181, 109646 CrossRef CAS.
- A. Marin-Flores, E. M. Arce-Estrada, A. Romero-Serrano, A. Rivera-Benitez, J. López-Rodríguez and A. Hernández-Ramírez, Int. J. Electrochem. Sci., 2022, 17, 221053 CrossRef CAS.
- R. Lakra, R. Kumar, S. Kumar, D. Thatoi and A. Soam, Mater. Today: Proc., 2023, 74, 863–866 CAS.
- E. Tovar-Martinez, C. E. Sanchez-Rodriguez, J. D. Sanchez-Vasquez, M. Reyes-Reyes and R. López-Sandoval, Diamond Relat. Mater., 2023, 136, 110010 CrossRef CAS.
- D. Torres, S. Pérez-Rodríguez, D. Sebastián, J. L. Pinilla, M. J. Lázaro and I. Suelves, Nanomaterials, 2020, 10, 1056 CrossRef CAS PubMed.
- R. Wang, X. Li, Z. Nie, Y. Zhao and H. Wang, J. Energy Storage, 2021, 38, 102479 CrossRef.
- M. Zhong, M. Zhang and X. Li, Carbon Energy, 2022, 4, 950–985 CrossRef CAS.
- M. R. Pallavolu, K. G. Krishna, G. Nagaraju, P. S. S. Babu, S. Sambasivam and A. Sreedhar, J. Ind. Eng. Chem., 2022, 116, 428–437 CrossRef CAS.
- K. Batool, M. Rani, S. M. Osman, M. Sillanpää, R. Shafique, S. Khan and M. Akram, Diamond Relat. Mater., 2024, 143, 110904 CrossRef CAS.
- Y. Zhao, H. Hao, T. Song, X. Wang, C. Li and W. Li, J. Alloys Compd., 2022, 914, 165343 CrossRef CAS.
- R. Dugas, J. D. Forero-Saboya and A. Ponrouch, Chem. Mater., 2019, 31, 8613–8628 CrossRef CAS PubMed.
- G. Wu, Y. Yang, R. Zhu, W. Yang, H. Yang and H. Zhou, Energy Environ. Sci., 2023, 16, 4320–4325 RSC.
- K. P. Shwetha, M. K. Sudha Kamath, Y. N. Athreya, C. K. Rastogi, K. Nagaraju, A. Khosla and C. Manjunatha, J. Energy Storage, 2024, 75, 109404 CrossRef.
- R. I. M. Vitto, M. T. Natividad and S. T. Palisoc, J. Power Sources, 2023, 582, 233547 CrossRef CAS.
- M. B. Arvas, N. Karatepe, M. Gencten and Y. Sahin, New J. Chem., 2021, 45, 6928–6939 RSC.
- L. Vivas and D. P. Singh, Front. Energy Res., 2022, 9, 794604 CrossRef.
- J. Miao, C. Zhou, X. Yan, H. Jiang, M. You, Y. Zhu, Y. Li, W. Zhou and X. Cheng, Energy Fuels, 2021, 35, 2766–2774 CrossRef CAS.
- S. D. Sutar, I. Patil, H. Parse, P. Mukherjee and A. Swami, Small, 2024, 2403552, DOI:10.1002/smll.202403552.
- S. C. Sekhar, B. Ramulu, S. J. Arbaz, S. K. Hussain and J. S. Yu, Small Methods, 2021, 2100335, DOI:10.1002/smtd.202100335.
- N. Van Toan, H. Sui, J. Li, T. T. K. Tuoi and T. Ono, J. Energy Storage, 2024, 81, 110446 CrossRef.
- G. Saeed, T. Kang, J. S. Byun, D. Min, J. S. Kim, S. V. Sadavar and H. S. Park, Energy Mater., 2024, 400023, DOI:10.20517/energymater.2023.81.
- S. Radhakrishnan, A. Patra, G. Manasa, M. A. Belgami, S. Mun Jeong and C. S. Rout, Adv. Sci., 2024, 2305325, DOI:10.1002/advs.202305325.
- C. Peng, X. Huang, M. Zhao, S. Liao, Q. Yang, N. Yang and S. Yu, J. Energy Chem., 2025, 426–457, DOI:10.1016/j.jechem.2024.08.051.
- M. Diantoro, H. Rahmadani, N. I. M. Atturoifah, Nasikhudin, I. C. Zawaidah and Aripriharta, J. Phys.: Conf. Ser., 2024, 2734, 012021 CrossRef CAS.
- P. E. Lokhande, U. S. Chavan, S. Bhosale, A. Kalam and S. Deokar, in Flexible Supercapacitor Nanoarchitectonics, Wiley, 2021, pp. 277–313 Search PubMed.
- B. K. Saikia, S. M. Benoy, M. Bora, D. Neog, D. Bhattacharjya, A. Rajbongshi and P. Saikia, J. Energy Storage, 2024, 78, 110312 CrossRef.
- G. Mora-Aquino, Á. L. Rodríguez-Morales, F. López-Huerta, E. Delgado-Alvarado, E. A. Elvira-Hernández and A. L. Herrera-May, Sens. Actuators, A, 2024, 372, 115321 CrossRef CAS.
- A. He, J. He, L. Cao, J. Chen, B. Cheng, R. Ma, Y. Ding, G. Yang and F. Yi, Adv. Mater. Technol., 2024, 2301931, DOI:10.1002/admt.202301931.
- M. D. L. Garayt, M. B. Johnson, L. Laidlaw, M. A. McArthur, S. Trussler, J. E. Harlow, J. R. Dahn and C. Yang, J. Electrochem. Soc., 2023, 170, 080516 CrossRef CAS.
- A. Schmid, C. Ellersdorfer, M. Raffler, N. Karajan and F. Feist, J. Power Sources, 2023, 581, 233510 CrossRef CAS.
- M. Merabtene, P. Tanninen, J. Wolf, F. Kayatz, M. Hauptmann, E. Saukkonen, A. Pesonen, T. Laukala, J. Varis and V. Leminen, Packag. Technol. Sci., 2023, 36, 667–679 CrossRef CAS.
- M. E. McGovern, D. D. Bruder, E. D. Huemiller, T. J. Rinker, J. T. Bracey, R. C. Sekol and J. A. Abell, J. Power Sources, 2023, 561, 232742 CrossRef CAS.
- M. Grandjean, M. Perrey, X. Randrema, J. Laurier, P. Chenevier, C. Haon and S. Liatard, J. Power Sources, 2023, 585, 233646 CrossRef CAS.
- S. M. Benoy, A. Hazarika, M. Bora, A. Rajbongshi, D. Sarmah, M. K. Phukan and B. K. Saikia, ACS Appl. Energy Mater., 2024, 7, 6045–6061 CrossRef CAS.
- T. Kedara Shivasharma, A. C. Mendhe, R. Sahu and B. R. Sankapal, J. Colloid Interface Sci., 2024, 676, 739–754 CrossRef CAS PubMed.
- Y. Xiong, X. Yan, T. Li, H. Jin, Z. Chen, X. Xu, X. Ji and X. Ge, Chem. Eng. J., 2023, 451, 138913 CrossRef CAS.
- J. K. Yadav, B. Rani, A. Tiwari and A. Dixit, J. Renewable Sustainable Energy, 2024, 014101, DOI:10.1063/5.0184499.
- J. Wu, X. Zhang, Z. Ju, L. Wang, Z. Hui, K. Mayilvahanan, K. J. Takeuchi, A. C. Marschilok, A. C. West, E. S. Takeuchi and G. Yu, Adv. Mater., 2021, 2101275, DOI:10.1002/adma.202101275.
- W. Fu, K. Turcheniuk, O. Naumov, R. Mysyk, F. Wang, M. Liu, D. Kim, X. Ren, A. Magasinski, M. Yu, X. Feng, Z. L. Wang and G. Yushin, Mater. Today, 2021, 48, 176–197 CrossRef CAS.
- G. F. Hawes, S. Rehman, Y. Rangom and M. A. Pope, Int. Mater. Rev., 2023, 68, 323–364 CrossRef CAS.
- S. Kim, G. Park, S. J. Lee, S. Seo, K. Ryu, C. H. Kim and J. W. Choi, Adv. Mater., 2023, 2206625, DOI:10.1002/adma.202206625.
- J. Li, J. Fleetwood, W. B. Hawley and W. Kays, Chem. Rev., 2022, 122, 903–956 CrossRef CAS PubMed.
- Y. Mi, Y. Lu, X. Wang, Z. Zhao, X. Cao and N. Wang, Batteries, 2022, 8, 215 CrossRef CAS.
- J.-H. Lee, G. Yang, C.-H. Kim, R. L. Mahajan, S.-Y. Lee and S.-J. Park, Energy Environ. Sci., 2022, 15, 2233–2258 RSC.
- A. G. Abo-Khalil, A. Sobhy, M. A. Abdelkareem and A. G. Olabi, Int. J. Thermofluids, 2023, 20, 100477 CrossRef.
- H. Guentri, T. Allaoui, M. Mekki and M. Denai, J. Energy Storage, 2021, 39, 102578 CrossRef.
- C. R. Arunkumar, U. B. Manthati and S. Punna, J. Energy Storage, 2022, 50, 104232 CrossRef.
- K. Liu, T. Duan, F. Zhang, X. Tian, H. Li, M. Feng, R. Wang, B. Jiang and K. Zhang, J. Mater. Chem. A, 2024, 12, 20606–20637 RSC.
- A. H. Khadem, Adv. Mater. Interfaces, 2023, 2300724, DOI:10.1002/admi.202300724.
- R. Speranza, P. Zaccagnini, A. Scalia, E. Tresso and A. Lamberti, J. Power Sources, 2023, 583, 233581 CrossRef CAS.
- H. Sheng, Y. Ma, H. Zhang, J. Yuan, F. Li, W. Li, E. Xie and W. Lan, Adv. Mater. Technol., 2024, 2301796, DOI:10.1002/admt.202301796.
- N. R. Chodankar, S. V. Karekar, M. Safarkhani, A. M. Patil, P. A. Shinde, R. B. Ambade, J. Kim, Y. Han, Y. Huh, A. al Ghaferi and E. Alhajri, Adv. Funct. Mater., 2024, 2406819, DOI:10.1002/adfm.202406819.
- S. A. Beknalkar, A. M. Teli, V. V. Satale, R. U. Amate, P. J. Morankar, M. A. Yewale and J. C. Shin, Energy Storage Materials, 2024, 66, 103217 CrossRef.
- J. N. Chukwunweike, C. C. Eze, I. Abubakar, L. O. Izekor and A. A. Adeniran, World J. Adv. Res. Rev., 2024, 23, 2538–3557 CrossRef.
- T. Ramachandran, R. K. Raji, S. Palanisamy, N. Renuka and K. Karuppasamy, J. Ind. Eng. Chem., 2025, 145, 144–168 CrossRef CAS.
- R. Malik, V. K. Tomer and M. Sain, EES Batteries, 2025, 1, 119–152 RSC.
- X. Wu, A. Chen, X. Yu, Z. Tian, H. Li, Y. Jiang and J. Xu, ACS Nano, 2024, 18, 20957–20979 CrossRef CAS PubMed.
- X.-L. Shi, L. Wang, W. Lyu, T. Cao, W. Chen, B. Hu and Z.-G. Chen, Chem. Soc. Rev., 2024, 9254–9305, 10.1039/D4CS00361F.
- S. Vanmathi, H. Awasthi, A. Pal and S. Goel, Sci. Rep., 2024, 14, 16953 CrossRef CAS PubMed.
- S. Kumar, E. Majhi, A. S. Deshpande and M. Khandelwal, Carbon Trends, 2024, 16, 100385 CrossRef CAS.
- R. Zhang, Y. Guo, J. Ning, J. Sun, T. Tao and H. He, J. Alloys Compd., 2024, 1006, 176218 CrossRef CAS.
- M. Sethi, U. S. Shenoy and D. K. Bhat, Nanoscale Adv., 2020, 2, 4229–4241 RSC.
- B. Freitas, W. G. Nunes, D. M. Soares, F. C. Rufino, C. M. Moreira, L. M. Da Silva and H. Zanin, Mater. Adv., 2021, 2, 4264–4276 RSC.
- M. Bilal, Z. U. Rehman, J. Hou, S. Ali, S. Ullah and J. Ahmad, in Metal Oxide-Carbon Hybrid Materials, Elsevier, 2022, pp. 25–60 Search PubMed.
- L. Jing, K. Zhuo, L. Sun, N. Zhang, X. Su, Y. Chen, X. Hu, R. Feng and J. Wang, J. Am. Chem. Soc., 2024, 146, 14369–14385 CrossRef CAS PubMed.
- J. Xue, Z. Sun, B. Sun, C. Zhao, Y. Yang, F. Huo, A. Cabot, H. K. Liu and S. Dou, ACS Nano, 2024, 18, 17439–17468 CrossRef CAS PubMed.
- D. N. Ampong, E. Effah, E. A. Tsiwah, A. Kumar, E. Agyekum, E. N. A. Doku, O. Issaka, F. O. Agyemang, K. Mensah-Darkwa and R. K. Gupta, Coord. Chem. Rev., 2024, 519, 216121 CrossRef CAS.
- H. Kwon, S. Kim, H. Park, J. S. Park, J. Park, Y. Lee, S. J. Yang and U. H. Choi, Adv. Funct. Mater., 2024, 2406727, DOI:10.1002/adfm.202406727.
- D. Merum, L. N. Ambadi, H. O. Mahammad, M. R. Pallavolu, M. Goddati, J. Lee, B. A. Al-Asbahi, R. Pitcheri, A. N. Banerjee and S. W. Joo, J. Alloys Compd., 2023, 950, 169771 CrossRef CAS.
- R. S. Desai, V. S. Jadhav, P. S. Patil and D. S. Dalavi, Mater. Adv., 2024, 5, 920–960 RSC.
- M. Dhananjaya, N. Guru Prakash, A. Lakshmi Narayana and O. M. Hussain, J. Electron. Mater., 2020, 49, 1922–1934 CrossRef CAS.
- A. Lakshmi-Narayana, M. Dhananjaya, C. M. Julien, S. W. Joo and C. V. Ramana, ACS Appl. Mater. Interfaces, 2023, 15, 20925–20945 CrossRef CAS PubMed.
- A. Rao, S. Bhat and S. De, Electrochim. Acta, 2024, 487, 144152 CrossRef CAS.
- H. Yu, J. Zhang, I. Shaheen, M. Ahmad, X. Chen, B. Akkinepally and I. Hussain, J. Ind. Eng. Chem., 2024, 129, 53–68 CrossRef CAS.
- D. Deb, in Ionic Liquids - Recent Advances, IntechOpen, 2024 Search PubMed.
- A. Aghmadi and O. A. Mohammed, Batteries, 2024, 10, 141 CrossRef CAS.
- D. Malavekar, S. Pujari, S. Jang, S. Bachankar and J. H. Kim, Small, 2024, 2312179, DOI:10.1002/smll.202312179.
- S. Alam, S. Jadoon, M. Z. Iqbal, H. H. Hegazy, Z. Ahmad and I. S. Yahia, J. Energy Storage, 2024, 85, 110955 CrossRef.
- A. Saeed, S. Khalid and M. Naeem Anjum, ChemistrySelect, 2024, e202303527, DOI:10.1002/slct.202303527.
- J. Zhang, K. Wang, P. Lu, J. Gao, Z. Cao, F. Mo, D. Ho, B. Li and H. Hu, Adv. Funct. Mater., 2023, 2310775, DOI:10.1002/adfm.202310775.
- Y.-Y. Hsieh and H.-Y. Tuan, Energy Storage Materials, 2024, 68, 103361 CrossRef.
- S. Arora and N. Verma, RSC Appl. Polym., 2024, 2, 317–355 RSC.
- W. Xie, Z. Zhang and X. Gao, Electrochim. Acta, 2024, 493, 144396 CrossRef CAS.
- J. Ma, S. Zheng, Y. Fu, X. Wang, J. Qin and Z.-S. Wu, Chem. Sci., 2024, 15, 5451–5481 RSC.
- K. M. A. Raihan, S. Sahoo, T. Nagaraja, S. Sigdel, B. LaCroix, C. M. Sorensen and S. R. Das, APL Energy, 2024, 016104, DOI:10.1063/5.0186302.
- R. K. Nare, S. Ramesh, V. Kakani, Y. Haldorai, C. Karthikeyan, B. P. Kumar, N. Siva Kumar, M. Asif, S. N. Kumar, D. P. Babu, K. R. Reddy and V. R. Pasupuleti, Diamond Relat. Mater., 2023, 141, 110660 CrossRef.
- J. Xie, D. Lin, H. Lei, S. Wu, J. Li, W. Mai, P. Wang, G. Hong and W. Zhang, Adv. Mater., 2023, 2306508, DOI:10.1002/adma.202306508.
- A. Rützler, J. Büttner, J. Oechsler, S. E. Balaghi, S. Küspert, N. Ortlieb and A. Fischer, Adv. Funct. Mater., 2024, 2401188, DOI:10.1002/adfm.202401188.
- B. Liu, Y. Ye, M. Yang, Y. Liu, H. Chen, H. Li, W. Fang and J. Qiu, Adv. Funct. Mater., 2023, 2310534, DOI:10.1002/adfm.202310534.
- A. Kachura, M. S. Zaman and O. Trescases, in 2024 IEEE Transportation Electrification Conference and Expo (ITEC), IEEE, 2024, pp. 1–6 Search PubMed.
- A. C. Lazanas and M. I. Prodromidis, ACS Meas. Sci. Au, 2023, 3, 162–193 CrossRef CAS PubMed.
- J. Zhang, M. Gu and X. Chen, Micro Nano Eng., 2023, 21, 100229 CrossRef.
- Z. He, C. Zhang, Z. Zhu, Y. Yu, C. Zheng and F. Wei, Adv. Funct. Mater., 2024, 2408285, DOI:10.1002/adfm.202408285.
- A. Sharma, R. Sharma, R. C. Thakur and L. Singh, J. Energy Chem., 2023, 82, 592–626 CrossRef CAS.
- C. Shen, W. Feng, Y. Yu, H. Wang, Y. Cheng, C. Dong, J. Gu, A. Zheng, X. Liao, X. Xu and L. Mai, Adv. Funct. Mater., 2024, 2304511, DOI:10.1002/aenm.202304511.
- G. Liu, Z. Zhang, Z. Li, L. Guo and L. Ning, 2D Materials, 2023, 10, 022002 CrossRef CAS.
- N. Parvin, V. Kumar, S. Park, T. K. Mandal and S. W. Joo, Small, 2024, 2407386, DOI:10.1002/smll.202407386.
- K. Abdou Ahmed Abdou Elsehsah, Z. Ahmad Noorden and N. Mat Saman, Heliyon, 2024, 10, e37071 CrossRef CAS PubMed.
- X.-M. Liu, Z. Dong Huang, S. Woon Oh, B. Zhang, P.-C. Ma, M. M. F. Yuen and J.-K. Kim, Compos. Sci. Technol., 2012, 72, 121–144 CrossRef CAS.
- Y. Dessie, S. Tadesse, R. Eswaramoorthy and Y. Adimasu, All Life, 2021, 14, 541–568 CrossRef CAS.
- B. Maria Mahimai, E. Li, J. Pang, J. Zhang and J. Zhang, J. Energy Storage, 2024, 96, 112598 CrossRef.
- J. P. Pender, G. Jha, D. H. Youn, J. M. Ziegler, I. Andoni, E. J. Choi, A. Heller, B. S. Dunn, P. S. Weiss, R. M. Penner and C. B. Mullins, ACS Nano, 2020, 14, 1243–1295 CrossRef CAS PubMed.
- Q. Wu, T. He, Y. Zhang, J. Zhang, Z. Wang, Y. Liu, L. Zhao, Y. Wu and F. Ran, J. Mater. Chem. A, 2021, 9, 24094–24147 RSC.
- X. Shu, W. Yang, B. Yang, K. Wei, K. Punyawudho and C. Liu, Eng. Failure Anal., 2024, 155, 107764 CrossRef CAS.
- X. Duan, C. Wang, F. Lv, T. Liu, X. Liu, Q. Wang and S. Yuan, Electrochim. Acta, 2025, 512, 145510 CrossRef CAS.
- F. Mashkoor, M. Shoeb, J. A. Khan, M. A. Gondal and C. Jeong, J. Energy Storage, 2024, 104, 114578 CrossRef.
- F. Yang, K. Lv, X. Zhao, D. Kong, N. Kong, Z. Luo, J. Tao, J. Zhou, J. M. Razal and J. Zhang, Chem. Eng. J., 2024, 494, 153246 CrossRef CAS.
- E. S. Sowbakkiyavathi, S. P. Arunachala Kumar, D. K. Maurya, B. Balakrishnan, J. Z. Guo and A. Subramania, Adv. Compos. Hybrid Mater., 2024, 7, 130 CrossRef CAS.
- A. Umar, F. Ahmed, N. Ullah, S. A. Ansari, S. Hussain, A. A. Ibrahim, H. Qasem, S. A. Kumar, M. A. Alhamami, N. Almehbad, H. Algadi, T. Almas, A. F. Selim and S. Baskoutas, Electrochim. Acta, 2024, 479, 143743 CrossRef CAS.
- N. M. Ghazali, N. F. Mazuki, M. H. Sulaiman, K. Aoki, Y. Nagao and A. S. Samsudin, Solid State Ionics, 2024, 414, 116650 CrossRef CAS.
- F. Zhang, T. Zhang, X. Yang, L. Zhang, K. Leng, Y. Huang and Y. Chen, Energy Environ. Sci., 2013, 6, 1623 RSC.
- Y. Wang, Y. Jin, W. Tian, B. Fan, D. Ding, Z. Zhao, Z. Chen, Z. Guo and P. Ren, Ind. Crops Prod., 2024, 222, 120035 CrossRef CAS.
- B. Ramulu, J. A. Shaik, A. R. Mule and J. S. Yu, Mater. Sci. Eng., R, 2024, 160, 100820 CrossRef.
- K. Raut, A. Shendge, J. Chaudhari, R. Lamba and N. F. Alshammari, J. Energy Storage, 2024, 82, 110324 CrossRef.
- B. Sun, N. Wang, X. Xie, L. Zhong, L. He, M. Xiang, K. Liang and W. Hu, Angew. Chem., Int. Ed., 2024, e202408569, DOI:10.1002/anie.202408569.
- L. Vaught, R. Sellers, B. Shirani Bidabadi, A. A. Polycarpou and A. Amiri, Chem. Eng. J., 2024, 492, 152234 CrossRef CAS.
- L. Pradhan, B. Mohanty, G. Padhy, R. Kumar Trivedi, D. Prasad Das, B. Chakraborty and B. Kumar Jena, Chem. Eng. J., 2024, 497, 154587 CrossRef CAS.
- Z. He, L. Yao, W. Guo, N. Sun, F. Wang, Y. Wang, R. Wang and F. Wang, Adv. Funct. Mater., 2023, 2305251, DOI:10.1002/adfm.202305251.
- M. Vandana, K. Bijapur, G. Soman and G. Hegde, Crit. Rev. Solid State Mater. Sci., 2024, 49, 335–370 CrossRef CAS.
- Y. Izadi and R. Beiranvand, IEEE Trans. Power Electron., 2023, 38, 15671–15692 Search PubMed.
- S. S. Shah, M. A. Aziz and Z. H. Yamani, Chem. Rec., 2022, e202200018, DOI:10.1002/tcr.202200018.
- S. M. Abu, M. A. Hannan, M. S. Hossain Lipu, M. Mannan, P. J. Ker, M. J. Hossain and T. M. I. Mahlia, J. Cleaner Prod., 2023, 394, 136246 CrossRef CAS.
- J. S. Teixeira, R. S. Costa, A. L. Pires, A. M. Pereira and C. Pereira, Dalton Trans., 2021, 50, 9983–10013 RSC.
- X. Wu, H. Yang, M. Yu, J. Liu and S. Li, Materials Today Energy, 2021, 21, 100739 CrossRef CAS.
- M. Pantrangi, E. Ashalley, M. K. Hadi, H. Xiao, Y. Zhang, W. Ahmed, N. Singh, A. Alam, U. Younis, F. Ran, P. Liang and Z. Wang, Energy Storage Materials, 2024, 73, 103791 CrossRef.
- A. Urooj and A. Nasir, World Electr. Veh. J., 2024, 15, 342 CrossRef.
- G. G. Njema, R. B. O. Ouma and J. K. Kibet, J. Renewable Energy, 2024, 2024, 1–35 CrossRef.
- G. Ren, H. Wang, C. Chen and J. Wang, Sustain. Energy Technol. Assess., 2021, 44, 100998 Search PubMed.
- J. Partridge and D. I. Abouelamaimen, Energies, 2019, 12, 2683 CrossRef CAS.
- D. Karimi, H. Behi, J. Van Mierlo and M. Berecibar, Designs, 2022, 6, 53 CrossRef.
- J. Zhang, Q. Zhang, G. Zhang, Y. Chen, S. Liu and L. Jiang, J. Energy Storage, 2023, 73, 109225 CrossRef.
- M. Yaseen, M. A. K. Khattak, M. Humayun, M. Usman, S. S. Shah, S. Bibi, B. S. U. Hasnain, S. M. Ahmad, A. Khan, N. Shah, A. A. Tahir and H. Ullah, Energies, 2021, 14, 7779 CrossRef CAS.
- S. Liu, L. Wei and H. Wang, Appl. Energy, 2020, 278, 115436 CrossRef CAS.
- S. Rajagopal, R. Pulapparambil Vallikkattil, M. Mohamed Ibrahim and D. G. Velev, Condens. Matter, 2022, 7, 6 CrossRef CAS.
- M. B. F. Ahsan, S. Mekhilef, T. K. Soon, M. B. Mubin, P. Shrivastava and M. Seyedmahmoudian, Int. J. Energy Res., 2022, 46, 19826–19854 CrossRef.
- R. T. Yadlapalli, R. R. Alla, R. Kandipati and A. Kotapati, J. Energy Storage, 2022, 49, 104194 CrossRef.
- L. Zhang, X. Hu, Z. Wang, J. Ruan, C. Ma, Z. Song, D. G. Dorrell and M. G. Pecht, Renewable Sustainable Energy Rev., 2021, 139, 110581 CrossRef.
- R. S. Sankarkumar and R. Natarajan, Int. Trans. Electr. Energy Syst., 2021, 12819 Search PubMed.
- H. Tang, S. Wang and H. Li, Energy, 2021, 219, 119598 CrossRef.
- R. T. Yadlapalli, A. Kotapati, R. Kandipati and C. S. Koritala, J. Energy Storage, 2022, 50, 104212 CrossRef.
- M. Khalid, Energies, 2019, 12, 4559 CrossRef CAS.
- K. Khan, A. K. Tareen, M. Aslam, A. Mahmood, Q. Khan, Y. Zhang, Z. Ouyang, Z. Guo and H. Zhang, Prog. Solid State Chem., 2020, 58, 100254 CrossRef CAS.
- Q. Zhang, L. Wang, G. Li and Y. Liu, J. Energy Storage, 2020, 31, 101721 CrossRef.
- A. Al-Qahtani, Z. Farooq and S. Almutairi, in Alternative Energies and Efficiency Evaluation, IntechOpen, 2022 Search PubMed.
- B. R. Ravada, N. R. Tummuru and B. N. L. Ande, IEEE J. Emerg. Sel. Top. Power Electron., 2021, 2, 334–342 Search PubMed.
- Z. Li, P. Yu, W. Zhong, M. Zhang, Z. Li, A. Cheng, Y. Liang, L. Miao, X. Yang and H. Zhang, Carbon, 2021, 176, 1–10 CrossRef CAS.
- Z. Qiu, Z. Liu, G. Wang, C. Huangfu, Z. Li, Y. Yan, C. Chi, P. Gao, X. Lu, S. Zhang, T. Wei and Z. Fan, Energy Storage Materials, 2023, 60, 102824 CrossRef.
- Wangmo, A. Helwig and J. Bell, J. Energy Storage, 2024, 95, 112701 CrossRef.
- J. Jiang, L. Yao, H. Peng, G. Wei, Y. Tian, L. Sun, P. Dai, P. Cai, Y. Zou, H. Zhang, F. Xu and B. Zhang, ACS Appl. Mater. Interfaces, 2024, 16, 22102–22112 CrossRef CAS PubMed.
- F. Parolin, P. Colbertaldo and S. Campanari, Energy Convers. Manage., 2022, 266, 115650 CrossRef CAS.
- H. Soleimani, D. Habibi, M. Ghahramani and A. Aziz, Energies, 2024, 17, 3291 CrossRef.
- A. A. Nkembi, M. Simonazzi, D. Santoro, P. Cova and N. Delmonte, Batteries, 2024, 10, 88 CrossRef CAS.
- A. M. Adeyinka, O. C. Esan, A. O. Ijaola and P. K. Farayibi, Sustainable Energy Research, 2024, 11, 26 CrossRef.
- A. Fatih Guven, A. Y. Abdelaziz, M. Mahmoud Samy and S. Barakat, Energy Convers. Manage., 2024, 312, 118560 CrossRef.
- R. Punyavathi, A. Pandian, A. R. Singh, M. Bajaj, M. B. Tuka and V. Blazek, Sci. Rep., 2024, 14, 5661 CrossRef CAS PubMed.
- X. Zhang, H. Yi, Y. Wen, Q. Li, Z. Wang, Y. Luo and F. Zhuo, IEEE Trans. Ind. Appl., 2024, 60, 7225–7239 Search PubMed.
- L. Wang, Q.-S. Vo and A. V. Prokhorov, IEEE Trans. Ind. Appl., 2018, 54, 50–60 Search PubMed.
- K. Karabacak, Energy Storage and Conversion, 2024, 2, 436 CrossRef.
- J. Duan, J. Liu, Q. Xiao, S. Fan, L. Sun and G. Wang, Energy, 2019, 169, 1242–1258 CrossRef.
- M. Antunes, Polymers, 2024, 16, 195 CrossRef CAS PubMed.
- H. Liu, K. Moon, J. Li, Y. Xie, J. Liu, Z. Sun, L. Lu, Y. Tang and C.-P. Wong, Nano Energy, 2020, 77, 105058 CrossRef CAS.
- Y. Zhang, Y. Zhang, C. Li, X. Yan, S. Hu, R. Yin, Y. Wei, K. Gao and H. Gao, Coord. Chem. Rev., 2024, 519, 216103 CrossRef CAS.
- S. Mathan, M. Selvaraj, M. A. Assiri, K. Kandiah and R. Rajendran, Surf. Interfaces, 2024, 51, 104707 CrossRef CAS.
- R. Wang, Y. Feng, D. Li, K. Li and Y. Yan, Green Chem., 2024, 26, 9075–9103 RSC.
- L. Zhao, Y. Peng, P. Dou, Y. Li, T. He and F. Ran, InfoMat, 2024, e12597, DOI:10.1002/inf2.12597.
- X. Su, J. Ye and Y. Zhu, J. Energy Chem., 2021, 54, 242–253 CrossRef CAS.
- Z. Sun, J. Pan, W. Chen, H. Chen, S. Zhou, X. Wu, Y. Wang, K. Kim, J. Li, H. Liu, Y. Yuan, J. Wang, D. Su, D. Peng and Q. Zhang, Adv. Energy Mater., 2023, 2303165, DOI:10.1002/aenm.202303165.
- M. Horn, B. Gupta, J. MacLeod, J. Liu and N. Motta, Curr. Opin. Green Sustainable Chem., 2019, 17, 42–48 CrossRef.
- S. K. Tiwari, A. K. Thakur, A. De Adhikari, Y. Zhu and N. Wang, Nanomaterials, 2020, 10, 2046 CrossRef CAS PubMed.
- R. Liang, Y. Du, P. Xiao, J. Cheng, S. Yuan, Y. Chen, J. Yuan and J. Chen, Nanomaterials, 2021, 11, 1248 CrossRef CAS PubMed.
- Y. Zhang, Y. Xu, R. Liu and Y. Niu, Chem. Eng. J., 2023, 461, 141985 CrossRef CAS.
- S. A. Delbari, L. S. Ghadimi, R. Hadi, S. Farhoudian, M. Nedaei, A. Babapoor, A. Sabahi Namini, Q. Van Le, M. Shokouhimehr, M. Shahedi Asl and M. Mohammadi, J. Alloys Compd., 2021, 857, 158281 CrossRef CAS.
- M. S. Javed, S. Asim, T. Najam, M. Khalid, I. Hussain, A. Ahmad, M. A. Assiri and W. Han, Carbon Energy, 2022, e271, DOI:10.1002/cey2.271.
- G. Smdani, M. R. Islam, A. N. Ahmad Yahaya and S. I. Bin Safie, Energy Environ., 2023, 34, 1094–1141 CrossRef.
- G. A. M. Ali, Z. H. Bakr, V. Safarifard and K. F. Chong, Recycled Nanomaterials for Energy Storage (Supercapacitor) Applications, in Waste Recycling Technologies for Nanomaterials Manufacturing, ed. A. S. H. Makhlouf and G. A. M. Ali, Topics in Mining, Metallurgy and Materials Engineering, Springer, Cham, 2021, DOI:10.1007/978-3-030-68031-2_7.
- Q. Wang, Z. Tan, G. De, L. Pu and J. Wu, Energy Sci. Eng., 2019, 7, 3147–3159 CrossRef CAS.
- S.-S. Chai, W.-B. Zhang, J.-L. Yang, L. Zhang, M. M. Theint, X.-L. Zhang, S.-B. Guo, X. Zhou and X.-J. Ma, RSC Sustainability, 2023, 1, 38–71 RSC.
- M. Mao, Y. Qi, K. Lu, Q. Chen, X. Xie, X. Li, Z. Lin, L. Chai and W. Liu, Environ. Sci. Technol., 2024, 58, 14013–14021 CrossRef CAS PubMed.
- A. S. Patil, A. V. Patil, C. G. Dighavkar, V. A. Adole and U. J. Tupe, Chem. Phys. Lett., 2022, 796, 139555 CrossRef CAS.
- A. U. Khan, A. Waris, D. Zamel, V. B. Mbayachi, A. Muhaymin, A. Safdar, Z. Shahid, M. Al Dalawi, U. y Umna and M. Khalil, in Chemical Physics of Polymer Nanocomposites, Wiley, 2024, pp. 15–44 Search PubMed.
- A. I. Osman, Z. Chen, A. M. Elgarahy, M. Farghali, I. M. A. Mohamed, A. K. Priya, H. B. Hawash and P. Yap, Adv. Energy Sustainability Res., 2024, 2400011, DOI:10.1002/aesr.202400011.
- L. U. Okonye and J. Ren, Heliyon, 2024, 10, e35285 CrossRef CAS PubMed.
- T. Liu and H. Yabu, Adv. Energy Sustainability Res., 2023, 2300168, DOI:10.1002/aesr.202300168.
- K. Dissanayake and D. Kularatna-Abeywardana, J. Energy Storage, 2024, 96, 112563 CrossRef.
- M. Reveles-Miranda, V. Ramirez-Rivera and D. Pacheco-Catalán, Renewable Sustainable Energy Rev., 2024, 192, 114196 CrossRef.
- B. Achzet, D. Ott, R. Fleiner, M. Gornik, A. Thorenz and C. Helbig, Cleaner Chem. Eng., 2024, 100122 CrossRef.
- S. Chen, D. Chen and J.-H. Lin, Renewable Sustainable Energy Rev., 2024, 204, 114817 CrossRef.
- A. Muzaffar, M. B. Ahamed and C. M. Hussain, Renewable Sustainable Energy Rev., 2024, 195, 114324 CrossRef CAS.
- A. M. Amani, L. Tayebi, E. Vafa, M. Abbasi, A. Vaez, H. Kamyab, S. Chelliapan, M. J. Azizli and R. Bazargan-Lari, J. Cleaner Prod., 2024, 436, 140606 CrossRef.
- S. Giri, M. Roy, S. Bandyopadhyay, S. Das, S. Mangal and S. Samanta, J. Mater. Chem. A, 2025 10.1039/D5TA01469G.
- Y. Anil Kumar, S. S. Sana, T. Ramachandran, M. A. Assiri, S. Srinivasa Rao and S. C. Kim, Dalton Trans., 2024, 53, 10770–10804 RSC.
- H. Cheng, J. Li, T. Meng and D. Shu, Small, 2024, 2308804, DOI:10.1002/smll.202308804.
- W. Yang, W. Yang, J. Zeng, Y. Chen, Y. Huang, J. Liu, J. Gan, T. Li, H. Zhang, L. Zhong and X. Peng, Prog. Mater. Sci., 2024, 144, 101264 CrossRef CAS.
- Q. J. Meisner, S. Jiang, P. Cao, T. Glossmann, A. Hintennach and Z. Zhang, J. Mater. Chem. A, 2021, 9, 25927–25933 RSC.
- H. Meskher, D. Ghernaout, A. K. Thakur, F. S. Jazi, Q. F. Alsalhy, S. S. Christopher, R. Sathyamurhty and R. Saidur, Mater. Today Commun., 2024, 38, 108517 CrossRef CAS.
- Y. Yin, Q. Liu, Y. Zhao, T. Chen, J. Wang, L. Gui and C. Lu, Energy Fuels, 2023, 37, 3523–3554 CrossRef CAS.
- S. D. Dhas, P. N. Thonge, A. C. Mendhe, J. Yun and D. Kim, Renewable Sustainable Energy Rev., 2024, 204, 114819 CrossRef CAS.
- K. A. Elsehsah, Z. A. Noorden and N. M. Saman, J. Energy Storage, 2024, 97, 112788 CrossRef.
- Q. Yang, Z. Lu, J. Liu, X. Lei, Z. Chang, L. Luo and X. Sun, Prog. Nat. Sci.:Mater. Int., 2013, 23, 351–366 CrossRef.
- D. Chen, K. Jiang, T. Huang and G. Shen, Adv. Mater., 2019, 1901806, DOI:10.1002/adma.201901806.
- Y. Yin, S. Zhang, Y. Liu, Z. Huang, W. Sun, M. Zhang, E. Zhou, H. Wu, L. Yang, X. Guan and P. Yin, ACS Omega, 2024, 9, 11883–11894 CrossRef CAS PubMed.
- L. Gaba, P. Siwach, K. Aggarwal, S. Dahiya, R. Punia, A. S. Maan, K. Singh and A. Ohlan, Adv. Colloid Interface Sci., 2024, 332, 103268 CrossRef CAS PubMed.
- S. Ponnada, M. S. Kiai, O. Eroglu, C.-O. Kim, R. K. Sharma and G. S. Martynkova, Energy Fuels, 2024, 18242–18264, DOI:10.1021/acs.energyfuels.4c02987.
Footnote |
| † These authors equally contributed to this work. |
| This journal is © The Royal Society of Chemistry 2025 |