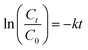Synthesis of carbon quantum dots decorated titanium disilicide: a novel hybrid solar-driven photocatalyst for sustainable wastewater treatment
Sarfaraz
Mahmood
a,
Arisha
Bi
a,
Sneha
Shukla
a,
Samina
Husain
 b,
Jai
Prakash
b,
Jai
Prakash
 *c and
Saif Ali
Chaudhry
*a
*c and
Saif Ali
Chaudhry
*a
aDepartment of Chemistry, Jamia Millia Islamia, New Delhi 110025, India. E-mail: saifchaudhry09@gmail.com
bCentre for Nanoscience and Nanotechnology, Jamia Millia Islamia, New Delhi 110025, India
cDepartment of Chemistry, National Institute of Technology Hamirpur, Himachal Pradesh-177005, India. E-mail: jaip@nith.ac.in
First published on 11th August 2025
Abstract
Producing an efficient solar driven semiconductor photocatalyst is required for harnessing the maximum amount of solar light for sustainable application in solving real-world problems. In the present work, a novel nanohybrid titanium disilicide/carbon quantum dot (TiSi2/CQD)-based composite photocatalyst was prepared via sonication followed by one-pot hydrothermal treatment. Several spectroscopic and microscopic techniques were used to investigate the successful composite formation with the detection of new electrostatic interactions and an overall increase in crystallinity and optoelectronic as well as photocatalytic properties. The TiSi2/CQD nanohybrid photocatalyst was tested for the visible light (400 W LED lamp) driven photocatalytic degradation of eosin yellowish (EY) and Nile blue (NB), an anionic and a cationic dye, respectively. The TiSi2/CQD photocatalyst showed excellent photodegradation efficiency of 83.7% (NB) and 95.1% (EY) as compared to TiSi2. To mimic the real wastewater conditions, the TiSi2/CQD photocatalyst was also tested with a mixture of two dyes (NB/EY) under direct natural sunlight conditions, with emphasis on the role of various parameters and their optimization (i.e., catalytic dosage, initial dye concentration and pH) in achieving the maximum degradation efficiency of 99%. The optoelectronic properties of TiSi2 were enhanced by decorating its surface with CQDs acting as an electron reservoir and significantly improved the visible light absorption, photocurrent response and charge carrier-transfer dynamics while decreasing the electron–hole recombination rate. As a result, the TiSi2/CQD composite showed excellent photodegradation with excellent recyclability up to three consecutive cycles. The kinetic studies showed that the photodegradation reactions followed the first order kinetics. This article introduces an economic, thermally stable and relatively non-toxic substitute for metal oxide-based semiconductor photocatalysts for excellent visible light activity.
1. Introduction
In this era of great industrial advancement, a large volume of untreated and harmful chemical waste is being discharged into waterbodies. Insecticides, pesticides, textile dyes, heavy metals and pharmaceuticals, etc. are some of the most common water contaminants, among which textile dyes own the largest share of about 20 percent.1 Commonly used textile dyes such as Nile blue (NB) and eosin yellowish (EY) are harmful chemicals with high toxicity, posing the risk of carcinogenic, teratogenic, and mutagenic health hazards.2,3 In addition, these highly stable dye molecules decreases light penetration in the water bodies due to their intense colors, thus reducing the rate of photosynthesis for the marine autotrophs.2,4 As a result, the marine organisms are being greatly harmed due to the formation of a toxic environment owing to the decrease in the dissolved oxygen.5 The untreated disposal and extensive usage of textile dyes had therefore become one of the biggest wastewater issues. Many techniques such as adsorption, photocatalysis, membrane filtration, ion exchange, coagulation and flocculation are employed to remove these harmful wastewater contaminants.Photocatalysis is considered as one of the prominent methods as it permanently breaks down the target pollutant into smaller (non-harmful) fragments. It is a process which involves the absorption of incident light on the catalyst surface resulting in the emission of photoinduced electrons, generating an electron–hole pair which in turn participates in the redox processes to initiate and speed up chemical reactions.6 In general, a photocatalyst is a material that transforms incident light energy into chemical energy. These materials can be employed to decompose unwanted chemical residues and pollutants under sunlight.7 Pioneers like Boddy (https://www.doi.org/10.1021/ar2003013%5d), Honda, and Fujishima (https://www.sciencedirect.com/science/article/pii/S2949839224000981 – ref. 17) demonstrated the immense potential of photocatalysis in the late 1960s, where the increased concern about energy sustainability had been very beneficial for the research progress in the domain of photocatalysis.8 Photocatalysts have applications in environmental remediation, hydrogen and hydrocarbon production, water splitting, self-cleaning coatings, drug delivery, etc. Metal oxide based semiconducting materials such as TiO2, ZnO, SnO2, WO3, similar doped materials, such as BiVO4, Fe2O3, co-doped metal oxides, and their composites had been extensively explored for their application in the domain of photocatalytic degradation of micropollutants.9 The major advantages of using metal oxide semiconductors are their biocompatibility, exceptional stability, and a favorable combination of their electronic structure, generation and transport of charge carriers and excited lifetimes of electrons, which had made them promising materials for photocatalytic applications.9,10 However, there are some disadvantages which limit their catalytic potential, making the photocatalytic process inefficient. Primarily, a large bandgap and a larger interfacial distance lead to a relatively slower charge carrier movement, causing a weaker visible light response.11,12 TiO2 and ZnO are two such extensively explored photocatalysts, both having bandgaps between 3.2 and 3.37 eV, limiting their activation to the ultraviolet (UV) region.13 They can only absorb UV light, which makes up only a small fraction (4–5%) of the solar radiation and is transparent across 95–96% of the solar spectrum that reaches the Earth's surface.14 The reliance of almost all the semiconductors on the UV light radiation (wavelength <390 nm) limits their range of applications to direct sunlight and conversely increases the energy consumption for artificial UV light driven photocatalysis.15 Another major drawback of TiO2, ZnO or similar photocatalysts arises from the rapid recombination rate of electron–hole pairs, thereby decreasing the quantum efficiency.16 Also, adsorption of target species on the catalyst surface plays an important role in the photocatalytic mechanism. The previously discussed catalysts have low adsorption capacity for hygroscopic contaminants owing to the weak charge separations and lower functionalities on their surface.13 Non-uniform distribution, agglomeration, difficult recovery of catalyst post-water treatment and photo-corrosion in alkaline solutions under UV irradiation are some of the secondary drawbacks of metal oxide based photocatalysts.17 Regarding the scale of toxicity, metal oxide semiconductors, such as TiO2 and ZnO, are considered highly toxic at the nanoscale where light irradiation can increase the toxicity to a great extent.18 TiO2 nanoparticles have been reported to be 347 times more toxic to E. coli under light conditions than under dark conditions.19 The IARC had classified TiO2 as a Group 2B carcinogen and genotoxic to humans.20
To tackle the above discussed drawbacks and harness the fullest potential of solar radiation by keeping the environmental hazards in mind, a new class of photocatalysts must be introduced with a narrow bandgap, relatively faster photoinduced charge separation and transfer tendency with a relatively lower electron–hole recombination rate. One such noble photocatalyst with the above desired properties had been introduced in the present research, which can emerge as an efficient material for solar energy harvesting. TiSi2 is an inorganic nanomaterial with a distinct two-dimensional (2D) morphology, high thermal and chemical stability and unique optoelectronic properties.21,22 TiSi2 is relatively unexplored as a photocatalyst in spite of having great optoelectronic properties such as high electrical conductivity, narrow bandgap, high thermal stability and non-toxicity. It had a bandgap ranging from 1.5 eV to 3.4 eV (corresponding to a wavelength from 800 nm to 360 nm) with prominent light absorption ability from the near-UV to the visible range.23 The material also possesses an outstanding thermal stability and excellent electrical conductivity.24,25 Additionally, TiSi2 does not exert toxicity on the health of living organisms and is considered relatively stable and environment friendly.26 Such characteristics make TiSi2 a promising candidate to be a sustainable photocatalyst and hence needs to be investigated further.
In the present work, the photocatalytic activity of TiSi2 has been explored, which is a noble material in the domain of photocatalytic degradation reactions. The aim is to improve the photocatalytic properties of TiSi2 for promising applications in the removal of dyes from the wastewater under visible light irradiation as well as in direct sunlight, showing its real practical application. It is noteworthy that in spite of having such exceptional optoelectronic properties TiSi2 struggled to achieve a very fast electron–hole recombination rate owing to its high conductivity.21,27 In order to increase the charge separation, an electron reservoir must be introduced which can delocalize the photoinduced electron and suppress the charge recombination rate. Carbon quantum dots (CQDs) are well known for their excellent electron reservoir and transport capabilities, which increase the electron–hole separation time by successfully trapping and transferring the photo-excited electrons, thereby improving the overall photocatalytic performance.28,29 Other benefits of combining CQDs with TiSi2 are that (i) CQDs can improve the light absorption capacity of TiSi2 and (ii) help in better adsorption of the target species on the composite surface due to the presence of abundant polar functionalities on CQDs.30 Many similar studies have been conducted, where CQDs are combined with other photocatalyst semiconductors like metal oxide semiconductors to get the desired photocatalytic properties such as TiO2/CQDs,31 L-CQDs/ZnO, etc.32 There are many methods of synthesis of TiSi2, namely self-propagating high-temperature synthesis (SHS), blast furnace slag, mechanical alloying, rapid thermal process (RTP) and chemical vapor deposition (CVD). The most feasible method for the bulk preparation of TiSi2 nanoparticles is mechanical alloying with subsequent annealing. However, this synthesis method had a drawback of having a small amount of unreacted precursors due to the collision inaccuracy but can be separated out later from the bulk.33 The CQDs were synthesized by the hydrothermal method for homogeneous particle size distribution, high density and extra purity.34
Lastly, the novel TiSi2/CQD composite was prepared by the one-pot hydrothermal method where the required ratio of TiSi2 nanoparticles and CQDs were mixed for high pressure hydrothermal treatment to yield a uniform and highly crystalline nanomaterial with improved optoelectronic properties and hence improved photocatalytic properties.35 Moreover, TiSi2 or the TiSi2/CQD composite has not been explored for photocatalytic reactions to date. The current research introduces a novel, non-toxic, highly efficient and visible light active composite nano-catalyst with a detailed analysis of its morphological and optoelectronic properties. The photocatalytic test involves a comparison of degradation efficiencies of the catalyst under white LED (visible light radiation) and open sunlight for an anionic as well as a cationic dye and a comparison with other prominent photocatalysts reported in the recent literature of similar compositions such as CQDs/ZnO36 and CQDs/TiO2.37
2. Experimental
2.1 Materials
Ti and Si powders of 3N grade were supplied by Sigma-Aldrich for the synthesis of TiSi2. Citric acid and ethylene glycol used to synthesize CQDs were obtained from Loba Chemie Pvt Ltd, New Delhi. L-Histidine was supplied by Garg Chemicals, New Delhi, which was used to increase the N and O containing functionalities in the CQDs. Ethylenediaminetetraacetic acid (EDTA), p-benzoquinone (BQ) and isopropyl alcohol (IPA) were supplied by SRL Pvt. Ltd, New Delhi. The other reagents including NB and EY were used as received. Deionized (DI) water, having resistivity >18.7 MΩ, was employed as the solvent. Analytical grade reagents were used throughout the experiment without any prior treatment.2.2 Preparation of TiSi2
Fig. 1(a) depicts the stepwise synthesis of TiSi2; in brief, the elemental Ti and Si powders were mixed in a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 molar ratio and subjected to mechanical alloying using a planetary ball mill (QSHO4L, Torrey Hills Tech., LLC) at 260 rpm, maintaining the inert atmosphere using Ar gas. Stainless steel balls (9.5 mm diameter) were used to maintain the ball to sample mass ratio as 18
2 molar ratio and subjected to mechanical alloying using a planetary ball mill (QSHO4L, Torrey Hills Tech., LLC) at 260 rpm, maintaining the inert atmosphere using Ar gas. Stainless steel balls (9.5 mm diameter) were used to maintain the ball to sample mass ratio as 18![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1. After 10 h of milling time, an intermediary U-phase was obtained, which was then converted to pure TiSi2 after 3 h of vacuum annealing at 800 °C.38
1. After 10 h of milling time, an intermediary U-phase was obtained, which was then converted to pure TiSi2 after 3 h of vacuum annealing at 800 °C.38
2.3 Preparation of CQDs
CQDs were synthesized by one-step hydrothermal treatment (Fig. 1(b)) where citric acid was used as a carbon source and L-histidine as a carbonization agent with ethylene glycol as a passivating agent. Briefly, 1.6 g of citric acid was dissolved in a 30 mL mixture containing equal volumes of ethylene glycol and DI water. A separate mixture of 1.0 g L-histidine (C6H9N3O2) dissolved in 10 mL of DI water was prepared. The two obtained mixtures were stirred separately for 10 min and then mixed and stirred for 10 min again. The resulting solution was then transferred to a Teflon autoclave (50 mL) and was subjected to hydrothermal treatment at 130 °C for 24 h and then cooled at room temperature (25 °C). The undispersed solids and impurities were removed by centrifugation at 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 rpm for 15 min. The supernatant was further dialyzed using a 3.5 kDa dialysis membrane to remove any residual impurity in the CQD sample.
000 rpm for 15 min. The supernatant was further dialyzed using a 3.5 kDa dialysis membrane to remove any residual impurity in the CQD sample.
2.4 Preparation of the TiSi2/CQD composite
The composite was prepared by ultrasonication followed by hydrothermal treatment, as described in Fig. 1(c). To prepare 20% wt/vol composite, 10 mL of CQDs and 0.2 mg TiSi2 in 20 mL DI were separately sonicated to obtain a well-dispersed set of solutions. These two solutions were then mixed and subjected to ultrasonic treatment for 1 h for the formation of necessary heterojunctions.39 The sample was then transferred to the Teflon autoclave (50 mL) for hydrothermal treatment at 130 °C for 12 h. The resulting composite was left to cool slowly for homogeneous nucleation, centrifuged twice at 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 rpm for 15 min and dried at 60 °C for 6 h.
000 rpm for 15 min and dried at 60 °C for 6 h.
2.5 Characterization of the TiSi2/CQD composite
The X-ray diffraction (XRD) patterns of the powder samples were analyzed using a Bruker-D8 Discover diffractometer (CIF, JMI) with Cu kα radiation (300 W) in the 10 to 80° (2θ) scanning range. The Fourier transform infrared (FT-IR) spectroscopy was done on a PerkinElmer Spectrum Two N spectrometer (MNIT, Jaipur, India) after formation of KBr pellets with transmittance ranging from 4000 to 400 cm−1. A high-resolution ZEISS EVO 18 microscope (SAIF, AIIMS, New Delhi) equipped with an energy dispersive X-ray spectroscopy (AMETEK EDAX) detector was used to perform field emission scanning electron microscopy (FESEM) and energy dispersive X-ray (EDX) analysis to obtain the surface images, elemental composition and elemental mapping images. The lattice characteristics, size and morphology of CQDs and TiSi2/CQDs composite were analyzed by high-resolution transmission electron microscopy (HR-TEM) using a Krios Cryo-TEM Talos instrument (SAIF, AIIMS, New Delhi) with an acceleration voltage of 220 kV. A Thermo Scientific NEXA surface analyzer (CIF, IIT-Jammu) with a background vacuum pressure of 10−7 mbar and a band-pass energy of 200 eV was used to conduct XPS measurements to determine the oxidation and bonding states of the present elements. UV-vis diffuse reflectance (UV-DRS) spectroscopy of the samples was performed on an Agilent Cary 5000 instrument (USIC, DU, New Delhi) having an integrating sphere with a sampling interval of 0.50 nm, and the bandgap was further calculated using the Kubelka–Munk equation. A WITec alpha300 laser photoluminescence spectrometer (USIC, DU, New Delhi) was employed to obtain the photoluminescence (PL) spectra with an excitation wavelength of 325 nm. Photocurrent response tests were performed on a CORRTEST CS250 electrochemical workstation. The photocurrent response was calculated in 0.5 M Na2SO4 aqueous solution with a three-electrode system. A glassy carbon electrode (GCE, 0.3 cm diameter) was employed as the working electrode, a platinum wire as the counter electrode and a saturated calomel electrode as the reference. The TiSi2 or TiSi2/CQD sample was dispersed in water to prepare a suspension, and a microliter (μL) amount of sample was dropped on the glassy GCE. After drying the GCE, 10 μL Nafion solution (5 wt%) was dropped on it. Keeping a stable open circuit voltage, the light was turned on and off every 20 seconds (s). Cyclic voltammetry (CV) was carried out in 0.1 M tetrabutylammonium perchlorate acetonitrile solution, where Ag+/AgCl (0.01 M AgNO3 in acetonitrile solution) was taken as the reference electrode in the dark and under visible light irradiation on a PalmSens potentiostat (CNN, JMI). Electrochemical impedance spectroscopy (EIS) measurements were performed on a BioLogic SP-150 workstation with 50 mL of 0.5 M Na2SO4 solution as the electrolyte and Ag+/AgCl as the reference electrode. Electron spin resonance (ESR) measurements were conducted on a JEOL JES-X320 instrument with 1 mg per mL photocatalyst and 50 mM DMPO in methanol and aqueous medium in the dark and under 10 min of visible light irradiation. A PerkinElmer UV-2100 UV-vis spectrometer was used to determine the absorbance for photodegradation studies.2.6 Photoreaction parameter optimizations and photocatalytic activity tests
To test the photocatalytic activity of the novel TiSi2/CQD composite, different anionic as well as cationic dyes were subjected to the photodegradation experiment under a visible lamp (white LED-400 W). NB and EY, a cationic and an anionic dye, respectively, were selected for the degradation experiment and optimization. For the purpose of parameter optimizations and photodegradation reactions throughout this research, 50 mL dye solutions after catalyst loading were kept in dark for 30 min to attain adsorption desorption equilibrium and then under visible light irradiation for 90 min at a distance of 25 cm with mild stirring. 5 mL aliquots were taken out every 10 min for adsorption and every 15 min for the photocatalytic studies. The photocatalyst was separated from the solution via centrifugation at 8000 rpm for 5 min, and the filtered aliquots were then analyzed using a UV-vis spectrophotometer. The detailed procedure of parameter optimization and explanation is presented in the SI. The optimum catalyst dosage for NB and EY was found to be 0.4 g L−1 (20 mg) and 0.5 g L−1 (25 mg), respectively (Fig. S1(a and c)). The highest dye degradation efficiency was shown for 20 ppm (∼56.5 μM) NB and 10 ppm (∼14.5 μM) EY (Fig. S1(b and d)). The influence of pH was also tested for both the dyes, which helped in a better understanding of the photodegradation mechanism. After the complete optimization of reaction parameters, a final reaction set was employed for 3 h where the photocatalytic efficiency of the TiSi2/CQD composite was compared with TiSi2 and self-degradation tendency (photolysis) of the dyes. At the completion of the photocatalytic experiment, the catalyst was recovered by physical separation and centrifugation and washed several times with DI water and ethanol to desorb the dye molecules from the catalytic surface.2.7 Scavenger tests
To determine the role and influence of different reactive oxygen species (ROS) in a photocatalytic reaction, different scavenging experiments were carried out. EDTA (10 mM), BQ (1 mM) and IPA![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O (2
H2O (2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 18) were employed to investigate the influence of hole (h+), superoxide radical (˙O2−) and hydroxyl radical (˙OH) scavengers, respectively. The scavenger test was carried out with the exact same procedure mentioned in the previous sections.
18) were employed to investigate the influence of hole (h+), superoxide radical (˙O2−) and hydroxyl radical (˙OH) scavengers, respectively. The scavenger test was carried out with the exact same procedure mentioned in the previous sections.
3. Results and discussion
3.1 Characterization of CQD, TiSi2 and CQD/TiSi2 composite
The X-ray Diffraction (XRD) pattern of TiSi2 (Fig. 2(a)) displays distinct diffraction peaks at 32.3°, 39.2°, 40.1°, 43.4°, 46.6°, and 47.3°, corresponding to the (111), (311), (040), (022), and (331) planes, respectively, of a face-centered orthorhombic structure. The sharp peaks at 29.1°, 46.9°, and 69.5° indicate the presence of small amounts of Si, and the peaks at 35.5°, 52.4°, and 64.4° can be attributed to the presence of a trace amount of Ti which was unreacted and is present in the final sample, which is one of the drawbacks of the mechanical alloying method.21,33 A similar result was also reported by Liu et al.25 Conversely, the XRD pattern of the TiSi2/CQD composite showed similar peaks at 32.3°, 39.2°, 40.2°, 43.4°, 46.5°, and 47.3°, corresponding to the (111), (311), (040), (022), and (331) planes of the composite material, respectively, where the increase in the peak intensity indicates the increase in the crystallinity as a whole, compared to that of the pure TiSi2 phase. A very few low intensity peaks are detected (29.8°, 41.0°, 54.4° and 66.5°) for the carbon species in the TiSi2/CQD spectrum because of a relatively lower concentration and poor crystallinity of CQDs. These results strongly suggest that there is no change in the crystal structure of TiSi2 after the incorporation of CQDs.40,41 The interplanar distance (d-spacing) using Bragg's law was also calculated and was found to be 0.236 nm and 0.220 nm corresponding to (311) and (040) planes, respectively.42The Fourier transform infrared (FTIR) spectra of TiSi2, CQDs and the TiSi2/CQD composite suggest the presence of different functional groups and verify the successful loading of CQDs on the surface of TiSi2, as displayed in Fig. 2(b). CQDs exhibit a broad absorption band from 3534 to 3442 cm−1 due to the N–H/O–H stretching vibration.37 The absorption band at 2962 cm−1 corresponds to C–H stretching vibrations. The peak at 1715 cm−1 indicates the presence of heteroaromatic imidazole skeletons in the CQDs. The peaks present at 1654 cm−1 and 1568 cm−1 correspond to the stretching vibration of –C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O of amide43 and C
O of amide43 and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C stretching vibrations, respectively. This suggests the formation of C
C stretching vibrations, respectively. This suggests the formation of C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C unsaturated bonds in the CQDs, which agrees with the lattice spacing measurements from HR-TEM.44 The absorption band at 1385 cm−1 represents the characteristic flexural vibrations of C–NH, where L-histidine was the source of N content.45,46 The stretching vibrations of C–O and C–N are displayed by the small peaks at 1127 cm−1 and 1025 cm−1, respectively.47 The C–N stretching band is observed at 1025 cm−1, and C–H and N–H bending vibrations are located at 875 cm−1 and 607 cm−1, respectively.43
C unsaturated bonds in the CQDs, which agrees with the lattice spacing measurements from HR-TEM.44 The absorption band at 1385 cm−1 represents the characteristic flexural vibrations of C–NH, where L-histidine was the source of N content.45,46 The stretching vibrations of C–O and C–N are displayed by the small peaks at 1127 cm−1 and 1025 cm−1, respectively.47 The C–N stretching band is observed at 1025 cm−1, and C–H and N–H bending vibrations are located at 875 cm−1 and 607 cm−1, respectively.43
The broad band situated at 3425 cm−1 for TiSi2 could be ascribed to O–H stretching, arising due to the absorbed moisture, which is also supported by the O–H bending vibration band at 1632 cm−1.48 A medium broad peak at around 1100 cm−1 can be assigned to Si–O–Si stretching, indicating that a trace amount of SiO2 is formed as impurity, which is also confirmed by XRD.49 Finally, the peaks in the range of 850 cm−1 to 550 cm−1 in both the TiSi2 and TiSi2/CQD samples correspond to the characteristic Si–Ti–Si stretching, which is slightly shifted towards lower wavenumbers in the composite. A great weakening of almost all the peaks is observed in the spectrum of composite material, indicating a good chemical connection between the two substances and can be attributed to the formation of Ti–O–C bonds between TiSi2 and the CQDs.50 The peaks present at 3750 cm−1, 2325 cm−1, 1100 cm−1 and 1025 cm−1 in the CQD spectrum were also detected in the spectrum of the TiSi2/CQD composite at a lower intensity due to less abundance of carbon content. A decrease in the intensity of the FTIR spectra is observed for the TiSi2/CQD composite, which can be attributed to the formation of a heterostructure, providing evidence for the successful loading of CQDs on the TiSi2 surface. FESEM was performed to investigate the surface morphologies of both TiSi2 and the TiSi2/CQD composite at different magnifications. An overall similar morphology was observed because the CQD loadings were in relatively lower concentration. It is evident from Fig. 2(c) that the surface of TiSi2 nanoparticles had an uneven particle size distribution and flat surfaces. On the other hand, the surface of the TiSi2/CQD composite was found to have agglomerated, spherical shaped, and evenly distributed particles, as clearly depicted in Fig. 2(d). A cauliflower-like morphology can be observed in the FESEM image of the composite. The overall increase in the homogeneity (which can also be an effect of hydrothermal treatment) perfectly aligns with the increase of crystallinity detected in the composite material by XRD.
Determination of the composition, elemental analysis and elemental mapping of pure TiSi2 and TiSi2/CQD nanocomposite were carried out using EDX. Fig. 2(e) presents the EDX spectrum of TiSi2, showing that it has Ti, Si and O (trapped moisture), whereas the EDX spectrum in Fig. 2(f) shows the presence of Ti, Si, C, O, and N in the TiSi2/CQD composite, which confirms the successful loading of CQDs on the TiSi2 surface. Fig. S2 and S3 depict the FESEM images of TiSi2 and TiSi2/CQD composite, respectively, along with elemental mapping of all the elements. The distribution of Ti and Si on the TiSi2 surface (Fig. S2) and Ti, Si, C, O, and N on the TiSi2/CQD surface (Fig. S3) was analyzed. A uniform distribution of the C, N, and O elements (Fig. S3(e–g)) revealed that the CQDs were dispersed homogeneously throughout the TiSi2 matrix.
The investigation of the average size, shape and physical appearance of the CQDs was carried out using HRTEM, as shown in Fig. 3(a). The average diameter of CQDs was determined after taking measurements of 20 random CQDs and was found to be 2.3–3.8 nm with a mean diameter of about 3.0 nm for which the size distribution chart is displayed in Fig. 3(b). Pure TiSi2 (Fig. 3(c)) appears as a clear (sheet-like) material with a characteristic fringe size of 0.220 nm in the HRTEM image, as shown in (Fig. 3(c), inset). It is clearly depicted in the TEM image (Fig. 3(d)) of the TiSi2/CQD composite that the CQDs (dark circular dots) are homogeneously distributed on the (grey sheet-like) surface of TiSi2, which were evidently absent in pure TiSi2. The (040) crystalline facet of anatase TiSi2 (Fig. 3(e)) and the (311) crystalline facet of graphitic carbon from CQDs (Fig. 3(e and f)) correspond to the lattice fringe distances of 0.220 nm and 0.236 nm, respectively.42,51 The interplanar (d) spacing calculated through HRTEM scaling was found to be in agreement with the value calculated from XRD data. Hence, TEM analyses of pure TiSi2 and TiSi2/CQDs confirm the proper decoration of CQDs on the TiSi2 surface.
XPS measurements were conducted to get a better understanding of the elemental states and chemical composition in TiSi2 and the TiSi2/CQD composite. The XPS survey spectrum (Fig. 4(a)) detects the presence of five main elements Ti (17.2%), Si (41.4%), O (13.5%), C (24.8%) and N (3.1%) in the composite, whereas only Ti (31.4%) and Si (63.1%) were majorly present in pure TiSi2 sample with trace amounts of O (4.9%) and C (0.6%) from the absorbed atmospheric moisture or CO2. The detection of C, O and N elements indicates the successful decoration of the TiSi2 surface by the CQDs. Three binding energy peaks are detected in the Ti 2p XPS spectrum of TiSi2 (Fig. 4(b)), where peaks at 454.1, 459.0 and 464.65 eV refer to Ti (2p3/2), Ti (2p3/2) and Ti (2p1/2), respectively.27 The differences in splitting energies between the Ti peaks are 4.9 and 5.65 eV, suggesting that Ti is present in Ti0 and Ti4+ oxidation states.
Comparing the peak positions of pure TiSi2 with those present in the TiSi2/CQDs composite, a downward shift was observed in the binding energies of Ti0 (454.1 eV to 453.46 eV) and Ti4+ (459.0 eV to 458.5 eV), respectively, as clearly depicted in Fig. 4(b and c). In the Ti 2p spectrum of the TiSi2/CQDs composite (Fig. 4(c)), the peak intensity of Ti0 is increased, whereas the peak intensity of Ti4+ shows a subsequent decrease, compared to the Ti 2p spectrum of pure TiSi2 (Fig. 4(b)). This change in the peak intensities indicates the reduction of Ti4+ to Ti0, which could be attributed to the electron donor capabilities of CQDs, facilitating the reduction process.27,52 The Si 2p spectrum (Fig. 4(d)) displays two peaks at 99.17 and 104.1 eV corresponding to the Si (2p 3/2) for Si0 and Si (2p 3/2) for Si4+, respectively. A similar trend of downshift in binding energies was observed for Si0 (99.17 eV to 98.6 eV) and Si4+ (104.1 eV to 103.5 eV) while comparing the Si 2p spectrum of pure TiSi2 with TiSi2/CQD, as displayed in Fig. 4(d and e), respectively. The shift of the relative peak position is attributed to the interaction of TiSi2 with CQDs in the TiSi2/CQD composite.53 This downshift in the binding energies of the composite spectra could be due to new bond formations or other electrostatic interactions between the TiSi2 and loaded CQDs causing the weakening of Si–Ti–Si bonds.54 The existence of electrostatic bonding environments between the TiSi2 and CQDs can also be explained by the Si 2p spectra having deconvoluted peaks at 102.1 eV and 104.0 eV, which can be attributed to –Si–C– and –Si–O– linkages (Fig. 4(e)).55,56 Despite the fact that –Si–C– was detected in the composite, its strength was weak compared to the significantly intense –Si–O– peak, indicating that oxygen functionalities of the CQD surface play an important role in the formation of the TiSi2/CQD composite. In the narrow scan of C 1s (Fig. 4(f)), the deconvoluted peaks of the C 1s spectra at 284.50, 285.14, 286.29, and 288.42 eV were assigned to the C–C, C–O, C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O and COOH, respectively, indicating the abundance of functionalities in the CQDs. Fig. 4(g) depicts O 1s spectra deconvoluted into three different peaks with binding energies at 529.5, 530.5, and 531.4 eV attributed to Ti–O, C–O and Ti–N–O, respectively. The peaks at 529.5 and 531.4 eV could be ascribed to the Ti–O and Ti–N–O interaction between Ti of TiSi2 and oxygen/nitrogen containing functionalities present in CQDs.57 The N 1s spectrum was also evaluated and is presented in Fig. S4. Therefore, it can be concluded that the composite was primarily formed by the electrostatic interactions between the hydroxyl or oxygen functionalities of CQDs and TiSi2.58
O and COOH, respectively, indicating the abundance of functionalities in the CQDs. Fig. 4(g) depicts O 1s spectra deconvoluted into three different peaks with binding energies at 529.5, 530.5, and 531.4 eV attributed to Ti–O, C–O and Ti–N–O, respectively. The peaks at 529.5 and 531.4 eV could be ascribed to the Ti–O and Ti–N–O interaction between Ti of TiSi2 and oxygen/nitrogen containing functionalities present in CQDs.57 The N 1s spectrum was also evaluated and is presented in Fig. S4. Therefore, it can be concluded that the composite was primarily formed by the electrostatic interactions between the hydroxyl or oxygen functionalities of CQDs and TiSi2.58
The valence band (VB) maximum (EVBM) of TiSi2 was also calculated from the XPS survey scan, as presented in Fig. 5(a).59 The UV-vis diffuse reflectance spectra of the samples are illustrated in Fig. 5(b). The pure TiSi2 and TiSi2/CQD composite exhibit a strong absorbance in the UV-vis range from 300 to 800 nm. The two main peaks present at 335 nm and 408 nm correspond to the π–π* and n–π* transitions, respectively. The composite showed enhanced absorption capability as compared to the pure TiSi2 attributed to the formation of chemical linkages between TiSi2 and CQDs, as discussed above. A similar phenomenon was observed for the RuO2-1/TiSi2 composite, as reported by Mou et al., 2013.21 Functionalization of the TiSi2 surface with CQDs had further enhanced the absorption ability of TiSi2 more towards the visible range, as also reported by Zhang et al. and others for TiO2/CQDs, where the bandgap was substantially decreased from 3.07 to 2.68 eV after CQD incorporation, justifying the role of CQDs in enhancing the light absorption of the composite.60–62
 | ||
| Fig. 5 (a) VB maximum (EVBM) of TiSi2. Optical evaluations of TiSi2 and TiSi2/CQDs: (b) UV-vis diffuse reflectance spectra and (c) Tauc plot for bandgap calculations. | ||
Furthermore, the bandgap (Eg) of both TiSi2 and TiSi2/CQD composite was calculated by the modified Kubelka–Munk equation,63 as illustrated in Fig. 5(c). The bandgap of pure TiSi2 was calculated to be 1.9 eV, as also reported by Mou et al., 2013 (ref. 21) and Zhang et al., 2019.27 On the other hand, the bandgap of the TiSi2/CQD composite was found to be slightly decreased to 1.8 eV (optimum for visible light harvesting),64 attributed to functionalization of TiSi2 with CQDs, which enhanced its visible light absorption by narrowing the bandgap. The reduction in the band gap of the TiSi2/CQDs composite is due to the introduction of a mid-gap state arising from the interaction between the electronic states of the CQDs and the TiSi2 matrix. These mid-gap states generate new energy levels, resulting from the interaction of the conduction band (CB) of TiSi2 and the quantum confinement effects of CQDs, effectively lowering the overall bandgap. This reduction in bandgap allows the TiSi2/CQD composite to absorb more visible light, extending the absorption range and enhancing the photocatalytic efficiency under visible light.65,66 Similarly, Khosroshahi et al.67 achieved the bandgap of 1.8 eV of the high-pressure rock salt polymorph of ZnO which showed enhanced photocatalytic activity compared to ZnO with a wurtzite crystal structure (3.1–3.4 eV). Similarly, Elsayed et al.68 reported that the bandgap for practical and efficient usage of a photocatalyst must be from 1.0 to 2.0 eV. Hence, it can be ascertained that the TiSi2/CQD composite can utilize the visible spectrum of light and can be an efficient visible light photocatalyst.
3.2 Photocatalytic activity of TiSi2 and TiSi2/CQD composite
The TiSi2/CQD composite was tested for its adsorption capacity for both the NB and EY dyes separately, in a dark environment before light irradiation. The adsorption–desorption equilibrium was established within 30 min for both the dyes and the catalyst under mild stirring in the dark. The photocatalytic degradation activity of the composite and pure TiSi2 for both the dyes and their self-degradation ability are presented in Fig. 6(a and b). An abrupt decrease in the C/Co for both the dyes can be seen in the first 15 min of visible light irradiation, indicating the initiation of the photodegradation reaction.The percentage degradation efficiency of the dyes for the composite as well as pure TiSi2 is displayed through a histogram in Fig. 6(c and d). Within 90 min of light irradiation, the self-degradation ability of NB and EY was found to be 22.2% and 35.2%, respectively. The degradation efficiency with pure TiSi2 within the same reaction time was found to be 67.6% for NB and 72.5% for EY. On the other hand, the composite showed a remarkable degradation efficiency of about 83.7% and 95.1% for NB and EY, respectively, in just 90 min visible light irritation. UV absorbance spectra of photocatalytic degradation with TiSi2/CQDs as the photocatalyst are displayed in Fig. 6(e) for NB and Fig. 6(f) for EY. The composite was also tested for a mixture containing equal volumes of the two dyes having same concentrations (20 ppm each) under a white LED lamp as well as in direct sunlight (Fig. 7) with a catalyst dosage of 0.4 g L−1 (20 mg). The composite showed astonishing degradation efficiencies of 99.5% (NB) and 61.3% (EY) for the dye mixture under direct sunlight compared to 76.0% (NB) and 71.9% (EY) under a white LED in just 90 min (Fig. 7(c and d)). Rapid and efficient degradation was achieved for NB under sunlight (Fig. 7(b)), whereas EY showed better degradation results under a white LED source (Fig. 7(a)). These results illustrate that the TiSi2/CQD composite can efficiently harness the energy of sunlight for photocatalytic reactions and can be further tuned by the CQD loading.
Fig. S5 compares the zero(th), pseudo-first and second order kinetics of the photocatalytic degradation reactions under optimized conditions for both NB and EY. The plot of −ln(Ct/C0) versus irradiation time (t) is linearly fitted with a convincing correlation factor of 0.93 (R2 ≥ 0.927) for NB and 0.98 (R2 ≥ 0.976) for EY, which suggests that both the reactions follow pseudo-first order kinetics (Fig. 8(a)). The kinetic studies discovered that the photodegradation of NB as well as EY under optimized conditions were in accordance with the pseudo-first order rate formula (eqn (1)):
The effect of pH change was also studied at optimized values of catalytic dosage and the initial dye concentration. A series of photodegradation experiments were carried out at pH ranging from 4 to 10 for both the dyes, where NB showed the best degradation of 81.1% at pH 8 (Fig. 8(c)). At pH 4, NB showed 62% degradation, which was substantially decreased to only 54% at pH 10. The TiSi2 surface is positive or negatively charged at a pH value lesser or greater than PZC (pHPZC = 5.1). The photocatalyst showed the best efficiency when the solution had a greater pH (pH = 8) than the PZC of TiSi2. In the alkaline environment, NB is in a cationic form (NB+), but the catalyst surface becomes negatively charged due to the mixing of the OH− radical with the holes at the catalyst surface causing the cationic dye to get absorbed more, hence increasing the degradation efficiency. When the pH value of the solution is decreased from 8 to 4, i.e., at a pH lower than pHPZC, the NB is cationic and the catalytic surface is also positively charged; therefore, the degradation efficiency decreases drastically due to the electrostatic repulsion between the NB and the catalyst surface.70 The degradation efficiency of the dye is the least at pH 10, and this may be due to the zwitterion formation in the dye which in turn increases the dimerization of NB dye due to interaction of the positive and negative ends.71 Therefore, NB and the catalyst surface had negligible interaction due to the decrease in charge density. Similarly, the degradation efficiency with pH change can be explained for EY (anionic dye), which showed a degradation of 94.3% at a lower pH of 4, whereas the degradation efficiency gradually decreased to 72.8% while increasing the pH to 10 (Fig. 8(d)). The effect of pH variations on the degradation efficiency of the NB and EY dyes is also consistent with the results in other literature, such as those reported by Shafaee et al., 2018.71
To check the stability and reusability of the TiSi2/CQD composite, the recyclability test was conducted for the photodegradation of EY, while all the reaction parameters were maintained as optimized earlier. After the photocatalytic degradation of EY, the catalyst was recollected and made ready to be used after removing any adsorbed dyes by washing with ethanol and drying. The composite showed a remarkable degradation efficiency of approximately 82.1% for the EY dye (Fig. 8(e and f)) even after three consecutive cycles of active photocatalysis, indicating high stability and multi-cyclic activity. The cyclic activity test results showed that the TiSi2/CQD composite is structurally stable and had good reusability, which is advantageous for practical applications.
3.3 Mechanism of enhanced photocatalytic activity of the TiSi2/CQD composite
TiSi2 showed excellent photodegradation for NB and EY, which was significantly enhanced by the incorporation of CQDs in the TiSi2 matrix. CQDs played a vital role in improving the optical properties of TiSi2, which further led to improved photocatalytic activity.It is well known that the charge separation and transfer efficiency are closely related to the photocatalytic performance. CQDs are well known for their electron trapping and storage capacity. Hence, the photogenerated electrons from TiSi2 can hover in the conducting and well conjugated network of CQDs,72 which in turn inhibits the electron–hole recombination rate. The availability of excited electrons increases the rate of free radical generation and initiation of the photodegradation reaction. This can be further explained by the PL measurements. Fig. 9(a) depicts the PL spectra of pure TiSi2 and TiSi2/CQD composite excited at 325 nm. The recombination rate of the photoinduced charge carriers is directly proportional to the PL intensity.73 It is evident from the PL spectra that the composite displays significant attenuation in the PL intensity compared to pure TiSi2. This illustrates that the CQDs predominantly play the role of electron reservoirs, and their incorporation in the TiSi2 matrix can greatly hinder the photogenerated electron–hole recombination rate.
Transient photocurrent response (PCR) as a function of time for the pure as well as composite materials was recorded. PCR deals with the charge separation and transfer efficiencies of the photo-generated electron–hole pair. Both the pure TiSi2 and TiSi2/CQD composite exhibit fast and reproducible PCR under several on–off cycles (20 s) of visible light irradiation (Fig. 9(b)). It is to be noted that the TiSi2/CQD composite exhibited a stronger PCR which was about twice as large compared to pure TiSi2. These results clearly indicate that the TiSi2/CQD composite possesses a higher charge separation and transfer capacity due to the synergetic effect of CQDs. In order to validate the PCR results and investigate the charge transfer dynamics of the photo-induced charge carriers, CV measurements were conducted. For electrochemical analysis, a slurry of the photocatalyst was coated on the 1 cm × 1 cm carbon/graphite electrode (CE), which was then dried and made ready for the CV tests. Fig. 9(c and d) present the CV charts of pure TiSi2 and TiSi2/CQD composite, with very smooth CV curves in the absence of any light source. On the other hand, when the same samples were subjected to light irradiation, the pure as well as composite samples showed rather rough CV curves. The peaks or flickering on the CV curve can be related to the process of charge transfer or movement of electrons and ions between the electrode material via an electrolyte.74 It can be argued that the generation of unexpected oxidation peaks corresponding to their applied potentials arises due to the gain of extra energy by the electrons from the incident light radiation, giving rise to an electron transfer process. The CV curves clearly depict that the oxidation peaks are much larger and more concentrated in the CV curve of the TiSi2/CQD composite compared to pure TiSi2, indicating that the CQDs have successfully amplified the charge carrier and transfer dynamics of TiSi2. The CV data perfectly align with the PL and PCR results and illustrate that the CQDs played an important role in reducing the electron–hole recombination rate of TiSi2 and enhancing the charge separation and transfer efficiencies of the photo-induced charge carriers, in turn improving the photocatalytic activity of TiSi2.
The EIS technique is employed to compare the charge transfer processes of the materials. Fig. 9(e) shows the EIS results of TiSi2 and TiSi2/CQD electrodes presented as Nyquist plots. The radius of the semi-circular plot of TiSi2/CQDs is much smaller than that of TiSi2. The trend demonstrates a significant reduction in the charge transfer resistance within the TiSi2/CQD interface compared to pure TiSi2, which is in agreement with the results of PL and PCR measurements. The CB position of the TiSi2 was measured by the Mott–Schottky (MS) plots.75Fig. 9(f) presents the MS plot of pure TiSi2 with a positive slope of the plot, confirming the n-type semiconductor behavior.76 The position of the CB of the n-type semiconductor is considered very close to the flat bandgap of the semiconductor. The CB potential (ECB) of pure TiSi2 was estimated by MS plots and was found to be −0.40 V vs. Ag/AgCl (−0.18 V vs. NHE). By using the relationship of ECB and bandgap (Eg), the valence band potential (EVB) was calculated for pure TiSi2, which appeared at 1.5 V vs. Ag/AgCl (1.72 V vs. NHE). Additionally, the valence band maximum (EVBM) was also evaluated by measuring the valence band region of pure TiSi2 using XPS, as shown in Fig. 5(a).
In order to understand the photodegradation reaction mechanism, the contributions of reaction initiators were studied through the degradation reaction itself. It is universally acknowledged that in any photo-activated or photodegradation reaction, the three major reaction initiators are photogenerated holes (h+), hydroxyl radicals (˙OH) and superoxide radicals (˙O2−). Ethylenediaminetetraacetic acid (EDTA), isopropyl alcohol (IPA) and p-benzoquinone (BQ) were employed as hole (h+), hydroxyl radical (˙OH) and superoxide radical (˙O2−) scavengers, respectively. In contrast, in the presence of different free radical scavengers, some of the reactive holes or radical species may be scavenged, which in turn inhibits the activity of the photocatalyst. The test results are displayed in Fig. 10(a), where the degradation efficiency for NB dye was almost the same as that of IPA but was slightly reduced for EDTA. A significant reduction in the degradation efficiency to 59.3% is observed when BQ was employed as a radical scavenger. This means that superoxide radicals (˙O2−) play a major role in the photodegradation mechanism of NB. Degradation of NB was favored by the ˙O2− (anionic) radical species, which can show better interaction with the NB (cationic) dye due to electrostatic interactions.77–79 On the other hand, IPA as a radical scavenger reduced the degradation efficiency down to 45.3% in the case of EY dye (Fig. 10(b)), indicating the major role of hydroxyl radicals (˙OH) in the degradation mechanism of EY dye.80 Hence, the contributions of the reactive species for the degradation of NB dye follows the order: ˙O2− > h+ > ˙OH. Whereas the contributions of reactive species for EY dye degradation are in the order: ˙OH > h+ > ˙O2−.81 To identify and confirm the radical species involved in the reduction of NB and EY dyes by TiSi2/CQDs, ESR experiments using DMPO as the spin-trapping agent were carried out. Fig. 10(c) shows the ESR spectra of DMPO-trapped ˙OH in aqueous medium and DMPO-trapped ˙O2− in methanol medium, which have been investigated in the active radical species for TiSi2/CQDs. Four characteristic peaks (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2
2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2
2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) of DMPO–˙OH adducts and four characteristic distorted peaks of the DMPO–˙O2− adducts were observed in TiSi2/CQD nanocomposite systems under visible light irradiation, but no signal was detected in the dark. The detected ˙OH characteristic signal peaks are mainly because the water molecule is oxidized by h+ to generate ˙OH.82 Similarly, the ˙O2− radicals are generated when photogenerated electrons from the catalyst's CB interact with the adsorbed molecular oxygen (O2) in the vicinity of the catalyst surface.83 It is clear from the ESR plots that the ˙OH had relatively more intense peaks compared to that of ˙O2− indicating the slightly higher concentration of ˙OH, thereby explaining the higher degradation percentage of EY. The ESR experiment provides evidence for the generation of ˙OH and ˙O2− radical species formed in TiSi2/CQDs under visible light irradiation. Hence, it can be ascertained that the TiSi2/CQD composite would generate relatively more active radical species due to the lower electron–hole recombination compared to pure TiSi2 having a higher electron–hole recombination, as already explained by PL and Nyquist plots.
1) of DMPO–˙OH adducts and four characteristic distorted peaks of the DMPO–˙O2− adducts were observed in TiSi2/CQD nanocomposite systems under visible light irradiation, but no signal was detected in the dark. The detected ˙OH characteristic signal peaks are mainly because the water molecule is oxidized by h+ to generate ˙OH.82 Similarly, the ˙O2− radicals are generated when photogenerated electrons from the catalyst's CB interact with the adsorbed molecular oxygen (O2) in the vicinity of the catalyst surface.83 It is clear from the ESR plots that the ˙OH had relatively more intense peaks compared to that of ˙O2− indicating the slightly higher concentration of ˙OH, thereby explaining the higher degradation percentage of EY. The ESR experiment provides evidence for the generation of ˙OH and ˙O2− radical species formed in TiSi2/CQDs under visible light irradiation. Hence, it can be ascertained that the TiSi2/CQD composite would generate relatively more active radical species due to the lower electron–hole recombination compared to pure TiSi2 having a higher electron–hole recombination, as already explained by PL and Nyquist plots.
Based on the results obtained, a plausible mechanism for the activity of TiSi2/CQD composite for the photocatalytic dye degradation is depicted in Fig. 11. Evidently, CQDs acted as electron reservoirs and played a key role in suppressing the electron–hole recombination rate of TiSi2 by accelerating the separation and transfer of charge carriers from the CB of TiSi2 to the well delocalized electrophilic network of CQDs.84,85 An increase in the light absorption capacity and improved visible light utilization was achieved by the coupling of CQDs with pure TiSi2. Upon irradiation of visible light on the TiSi2 surface, an electron jumps out from the VB (1.5 V vs. Ag/AgCl) to the CB (−0.4 V vs. Ag/AgCl) leaving a positively charged vacancy behind (holes), overcoming the bandgap (1.9 eV), as a consequence of the photoelectric effect. The electron after losing the energy tends to recombine readily with the holes at the VB of TiSi2. CQDs (being electrophilic) act as electron reservoirs, and their presence in the vicinity of the CB of TiSi2 in the TiSi2/CQD composite lowers the electron–hole recombination rate to a significant degree. The increase in the charge separation time and higher charge densities of both the extreme ends acts as the ROS generation centers. The e− separated and delocalized in the CQDs acts as the reduction center of molecular O2 and generates ˙O2− species. The holes generated in the valence band region of TiSi2 interacts with H2O and generates ˙OH radical species.86 The adsorbed NB and EY molecules on the surface of TiSi2/CQD composite get readily oxidized by ˙O2− and ˙OH radical species, respectively, and further decompose to give CO2 and H2O as the major degradation products. Similar results can be found in multiple literature reports such as those from Saleem et al.77 and Sarwan et al.,87 who found ˙O2− and ˙OH are ROS responsible for the photodegradation and mineralization of NB. Saleem et al.77 proposed a degradation mechanism where the ROS attacks at a double bond (unsaturated sites) present in the aromatic ring of the NB dye. The breakdown of conjugation leads to the decolourization of dye with the formation of smaller intermediate fragments, such as 1,2-dihydroxy naphthalene, benzene-1,4-diol, 1,4-benzoquinone and dicarboxylic acid, which further mineralized to from CO2 and H2O within 100 min. Whereas Vignesh et al.88 and Debnath et al.80 found ˙OH as the ROS responsible for the photocatalytic mineralization of the EY dye, which was supported by studying the degradation products through mass spectrometry.80,89 Vignesh et al. studied the degradation intermediates of EY by GC, where propanedioic acid, 3,5-dibromocyclohex-5-ene-1,2,4-trione and benzoic acid were found as intermediates, which further degraded into CO2 and H2O in 180 min. It must be noted that the degradation efficiencies in our study are comparable to those found in the study conducted by Debnath et al.80 with a marginally less reaction time. Hence, it can be ascertained that both the dyes must be completely mineralized by the TiSi2/CQD composite during the course of the photodegradation reaction. The plausible mechanism of the photodegradation reaction of NB and EY dyes by the composite can be illustrated as follows:
| TiSi2/CQD + hν → e− + h+ | (1) |
| H2O + h+ → H+ + ˙OH | (2) |
| O2 + e− → ˙O2− | (3) |
| O2− + 2H+ → H2O2 | (4) |
| H2O2 → 2˙OH | (5) |
| ˙O2− + NB → CO2 + H2O | (6) |
| ˙OH + EY → CO2 + H2O | (7) |
 | ||
| Fig. 11 Plausible mechanism of the photocatalytic degradation reaction by the TiSi2/CQD composite under sunlight. | ||
Table 1 comprises the experimental results of some similar well-explored photocatalysts, which shall be compared with the TiSi2/CQD composite. The results clearly distinguish the novelty of the TiSi2/CQD composite which showed remarkable degradation efficiency with a relatively lower catalytic dosage and higher dye concentrations. The simple synthesis method with lower toxicity, better cyclic reusability and the remarkable ability to harness the visible light energy make TiSi2 a photocatalyst worthy of exploration.
| Material | Catalyst dosage | Dye conc. (mg L−1) | Target pollutant | Light source | Time (min) | Degradation efficiency (%) |
|---|---|---|---|---|---|---|
| TiO2-CQDs90 | Photo-catalytic film | 20 | RhB | 500 W Xe lamp with an UV filter | 150 | 77 |
| TiO2/CQDs91 | 2 g L−1 | 10 | RhB | 500 W Xe lamp with a 420 nm wavelength | 90 | ∼57 |
| CTP/TiO2 (ref. 92) | 1 g L−1 | 10 | RhB | 300 W Xe lamp with a cut-off filter | 360 | 91.1 |
| C-dots/TiO2 (ref. 93) | 0.2 g L−1 | 50 | RhB | 300 W Xe arc lamp cut-off filter (λ ≥ 400 nm) | 90 | 44 |
| P25/NCQDs94 | 6 wt% | 10 | MB | Simulated sunlight | 60 | 91 |
| CQDs/TiO2 (ref. 95) | 0.25 g L−1 | 10 | MB | 500 W Xe | 120 | 86 |
| CQDs/TiO2 nanohybrid96 | 1 g L−1 | 10 | MB | 80 W LED | 60 | 41 |
| CQDs/TiO2 nanotubes97 | — | 5 | MB | 500 W Xe | 100 | ∼35 |
| CQDs/TiO2/Fe2O3 (ref. 98) | 0.5 g L−1 | 20 | MB | 500 W Xe | 180 | 86.5 |
| NCQDs/TiO2 (ref. 99) | 1 g L−1 | 10 | MB | 42 W LED | 420 | 86 |
| CQD/MH100 | 50 mg L−1 | 20 | MB | 400 W halogen spotlight | 90 | 87.7 |
| TiO2/CQDs/BiOX101 (X = Cl, Br, I) | 0.28 g L−1 | 5 | MO | Simulated sunlight | 180 | 80 |
| TiSi2/CQDs (this work) | 0.4 g L−1 | 20 | NB | 400 W LED lamp | 120 | 83.7 |
| TiSi2/CQDs (this work) | 0.5 g L−1 | 10 | EY | 400 W LED lamp | 120 | 95.1 |
4. Conclusion
Novel photocatalysts, TiSi2 and TiSi2/CQD composite, were synthesized by annealing following mechanical alloying and hydrothermal methods, respectively. These photocatalysts were successfully characterized using various spectroscopic and microscopic techniques such as XRD, XPS, FTIR, UV-visible, FESEM, EDX, HRTEM, etc. for investigating optoelectronic, morphological and photocatalytic properties, etc. The composite was systematically optimized for parameters such as catalytic dosage, initial dye concentrations and pH. The photocatalytic activity of TiSi2 and TiSi2/CQD composite was investigated for NB and EY dyes as target pollutants. The TiSi2/CQD composite showed enhanced degradation efficiencies of 83.7% (NB) and 95.1% (EY) compared to 67.6% (NB) and 72.5% (EY) for the pure TiSi2. The composite also showed promising results for the degradation of the dye mixture, which mimics the real wastewater conditions, and the efficiency of about 99.5% (NB) and 61.3% (EY) was achieved in just 90 min of direct sunlight irradiation. The TiSi2/CQD composite photocatalyst showed the first order kinetics with degradation rate constants of 0.014 m−1 (NB) and 0.028 m−1 (EY), which are significantly higher compared to values obtained for pure TiSi2. Multiple electrochemical and optical studies such as PL, CV and EIS were conducted to analyze the role of CQDs in the capture and transfer of photoinduced electrons. The participation of different reactive radical species was examined in detail by scavenger studies and supported by the ESR measurements, where ˙O2− and ˙OH radicals were predominantly active in the photodegradation of NB and EY dyes, respectively. This research shows the important role of CQDs in promoting the photodegradation efficacy of TiSi2 by their remarkable capability to act as an e− reservoir, in turn suppressing the electron–hole recombination rate by accelerating the separation and transfer of photoinduced charge carriers from the CB of TiSi2. Hence, the TiSi2/CQD composite has proved its remarkable capability as an efficient visible light active, multi-cyclic photocatalyst. More series of TiSi2/CQD composites with different mass ratios can be optimized to get an even better reaction efficiency. TiSi2 in itself has magnificent optoelectronic properties and can be coupled with different materials to achieve excellence in the domain of visible light driven photocatalysis for multiple real-world applications.Conflicts of interest
There are no conflicts to declare.Data availability
The data that support the findings of this study are available from the authors.Photoreaction parameter optimizations and photocatalytic activity test, FESEM image and respective elemental mapping of TiSi2 and TiSi2/CQD composite, high-resolution N 1s spectra of TiSi2/CQD, zero-order, pseudo-first order and pseudo-second order photodegradation kinetics. See DOI: https://doi.org/10.1039/d5ta02427g.
Acknowledgements
Sarfaraz Mahmood thanks all the co-authors and acknowledges the support from the Environmental Chemistry Lab (JMI), Central Instrumental Facility (CIF, JMI) and Center for Nanoscience and Nanotechnology (CNN, JMI). Special thanks to Mr Sahil Thakur (NIT-Hamirpur), Rupam Sharma (NIT-Hamirpur) and Dr Gyan Singh (CNN, JMI) for their support and mentorship.References
- S. Dutta, S. Adhikary, S. Bhattacharya, D. Roy, S. Chatterjee, A. Chakraborty, D. Banerjee, A. Ganguly, S. Nanda and P. Rajak, Contamination of Textile Dyes in Aquatic Environment: Adverse Impacts on Aquatic Ecosystem and Human Health, and Its Management Using Bioremediation, J. Environ. Manage., 2024, 353, 120103, DOI:10.1016/j.jenvman.2024.120103.
- Q. Wang, C. Cai, M. Wang, Q. Guo, B. Wang, W. Luo, Y. Wang, C. Zhang, L. Zhou, D. Zhang, Z. Tong, Y. Liu and J. Chen, Efficient Photocatalytic Degradation of Malachite Green in Seawater by the Hybrid of Zinc-Oxide Nanorods Grown on Three-Dimensional (3D) Reduced Graphene Oxide(RGO)/Ni Foam, Materials, 2018, 11(6), 1004, DOI:10.3390/ma11061004.
- F. M. Drumond Chequer, G. A. R. de Oliveira, E. R. Anastacio Ferraz, J. Carvalho, M. V. Boldrin Zanoni, and D. P. de Oliveir, Textile Dyes: Dyeing Process and Environmental Impact, in Eco-Friendly Textile Dyeing and Finishing, InTech, 2013, DOI:10.5772/53659.
- F. Barancheshme and M. Munir, Strategies to Combat Antibiotic Resistance in the Wastewater Treatment Plants, Front. Microbiol., 2018, 8, 2603, DOI:10.3389/fmicb.2017.02603.
- C. Hayward, K. E. Ross, M. H. Brown and H. Whiley, Water as a Source of Antimicrobial Resistance and Healthcare-Associated Infections, Pathogens, 2020, 9(8), 667, DOI:10.3390/pathogens9080667.
- S. Khan, T. Noor, N. Iqbal and L. Yaqoob, Photocatalytic Dye Degradation from Textile Wastewater: A Review, ACS Omega, 2024, 9(20), 21751–21767, DOI:10.1021/acsomega.4c00887.
- S. Thakur, A. Bi, S. Mahmood, Samriti, O. Ruzimuradov, R. Gupta, J. Cho and J. Prakash, Graphene Oxide as an Emerging Sole Adsorbent and Photocatalyst: Chemistry of Synthesis and Tailoring Properties for Removal of Emerging Contaminants, Chemosphere, 2024, 352, 141483, DOI:10.1016/j.chemosphere.2024.141483.
- A. Fujishima and K. Honda, Electrochemical Photolysis of Water at a Semiconductor Electrode, Nature, 1972, 238(5358), 37–38, DOI:10.1038/238037a0.
- C. Karthikeyan, P. Arunachalam, K. Ramachandran, A. M. Al-Mayouf and S. Karuppuchamy, Recent Advances in Semiconductor Metal Oxides with Enhanced Methods for Solar Photocatalytic Applications, J. Alloys Compd., 2020, 828, 154281, DOI:10.1016/j.jallcom.2020.154281.
- F. T. Geldasa, M. A. Kebede, M. W. Shura and F. G. Hone, Experimental and Computational Study of Metal Oxide Nanoparticles for the Photocatalytic Degradation of Organic Pollutants: A Review, RSC Adv., 2023, 13(27), 18404–18442, 10.1039/D3RA01505J.
- R. Li, T. Gao, Y. Wang, Y. Chen, W. Luo, Y. Wu, Y. Xie, Y. Wang and Y. Zhang, Engineering of Bimetallic Au–Pd Alloyed Particles on Nitrogen Defects Riched g-C3N4 for Efficient Photocatalytic Hydrogen Production, Int. J. Hydrogen Energy, 2024, 63, 1116–1127, DOI:10.1016/j.ijhydene.2024.03.113.
- F. Zhou, J. Zhang, Y. Zhang, Y. Wu, Y. Wang and W. Luo, Palladium-Copper Bimetallic Catalysts for Electroreduction of CO2 and Nitrogenous Species, Coord. Chem. Rev., 2024, 509, 215802, DOI:10.1016/j.ccr.2024.215802.
- H. Dong, G. Zeng, L. Tang, C. Fan, C. Zhang, X. He and Y. He, An Overview on Limitations of TiO2-Based Particles for Photocatalytic Degradation of Organic Pollutants and the Corresponding Countermeasures, Water Res., 2015, 79, 128–146, DOI:10.1016/j.watres.2015.04.038.
- M. Karmaoui, A. B. Jorge, P. F. McMillan, A. E. Aliev, R. C. Pullar, J. A. Labrincha and D. M. Tobaldi, One-Step Synthesis, Structure, and Band Gap Properties of SnO 2 Nanoparticles Made by a Low Temperature Nonaqueous Sol–Gel Technique, ACS Omega, 2018, 3(10), 13227–13238, DOI:10.1021/acsomega.8b02122.
- K. Davis, R. Yarbrough, M. Froeschle, J. White and H. Rathnayake, Band Gap Engineered Zinc Oxide Nanostructures via a Sol–Gel Synthesis of Solvent Driven Shape-Controlled Crystal Growth, RSC Adv., 2019, 9(26), 14638–14648, 10.1039/C9RA02091H.
- S. Banerjee, S. C. Pillai, P. Falaras, K. E. O'Shea, J. A. Byrne and D. D. Dionysiou, New Insights into the Mechanism of Visible Light Photocatalysis, J. Phys. Chem. Lett., 2014, 5(15), 2543–2554, DOI:10.1021/jz501030x.
- B. Abebe, H. C. A. Murthy and E. Amare, Enhancing the Photocatalytic Efficiency of ZnO: Defects, Heterojunction, and Optimization, Environ. Nanotechnol., Monit. Manage., 2020, 14, 100336, DOI:10.1016/j.enmm.2020.100336.
- S. Thakur, A. Ojha, S. K. Kansal, N. K. Gupta, H. C. Swart, J. Cho, A. Kuznetsov, S. Sun and J. Prakash, Advances in Powder Nano-Photocatalysts as Pollutant Removal and as Emerging Contaminants in Water: Analysis of Pros and Cons on Health and Environment, Adv. Powder Mater., 2024, 3(6), 100233, DOI:10.1016/j.apmate.2024.100233.
- J. Hou, L. Wang, C. Wang, S. Zhang, H. Liu, S. Li and X. Wang, Toxicity and Mechanisms of Action of Titanium Dioxide Nanoparticles in Living Organisms, J. Environ. Sci., 2019, 75, 40–53, DOI:10.1016/j.jes.2018.06.010.
- M. Skocaj, M. Filipic, J. Petkovic and S. Novak, Titanium Dioxide in Our Everyday Life; Is It Safe?, Radiol. Oncol., 2011, 45(4), 227–247, DOI:10.2478/v10019-011-0037-0.
- Z. Mou, S. Yin, M. Zhu, Y. Du, X. Wang, P. Yang, J. Zheng and C. Lu, RuO2/TiSi2/Graphene Composite for Enhanced Photocatalytic Hydrogen Generation under Visible Light Irradiation, Phys. Chem. Chem. Phys., 2013, 15(8), 2793, 10.1039/c2cp44270a.
- G. Medeiros-Ribeiro, D. A. A. Ohlberg, D. R. Bowler, R. E. Tanner, G. A. D. Briggs and R. Stanley Williams, Titanium Disilicide Nanostructures: Two Phases and Their Surfaces, Surf. Sci., 1999, 431(1–3), 116–127, DOI:10.1016/S0039-6028(99)00434-3.
- S. Banerjee, S. K. Mohapatra and M. Misra, Water Photooxidation by TiSi 2 –TiO 2 Nanotubes, J. Phys. Chem. C, 2011, 115(25), 12643–12649, DOI:10.1021/jp106879p.
- J. Pelleg and G. Sade, TiB2/TiSi2 Bilayer Fabrication by Various Techniques: Structure and Contact Properties, Phys. B, 2006, 371(1), 20–34, DOI:10.1016/j.physb.2005.10.001.
- J. Liu, Y. Bai, P. Chen, N. Cui and H. Yin, Reaction Synthesis of TiSi2 and Ti5Si3 by Ball-Milling and Shock Loading and Their Photocatalytic Activities, J. Alloys Compd., 2013, 555, 375–380, DOI:10.1016/j.jallcom.2012.12.100.
- Titanium silicide (TiSi2), National Library of Medicine, pubchem.ncbi, https://pubchem.ncbi.nlm.nih.gov/compound/6336889.
- C. Zhang, A. Liu, K. Li, Y. Du and P. Yang, One-Step Synthesis of MoS2/TiSi2 via an In Situ Photo-Assisted Reduction Method for Enhanced Photocatalytic H2 Evolution under Simulated Sunlight Illumination, Catalysts, 2019, 9(3), 299, DOI:10.3390/catal9030299.
- W. Wu, L. Zhan, K. Ohkubo, Y. Yamada, M. Wu and S. Fukuzumi, Photocatalytic H2 Evolution from NADH with Carbon Quantum Dots/Pt and 2-Phenyl-4-(1-Naphthyl)Quinolinium Ion, J. Photochem. Photobiol., B, 2015, 152, 63–70, DOI:10.1016/j.jphotobiol.2014.10.018.
- A. Aghamali, M. Khosravi, H. Hamishehkar, N. Modirshahla and M. A. Behnajady, Synthesis and Characterization of High Efficient Photoluminescent Sunlight Driven Photocatalyst of N-Carbon Quantum Dots, J. Lumin., 2018, 201, 265–274, DOI:10.1016/j.jlumin.2018.04.061.
- H.-L. Yang, L.-F. Bai, Z.-R. Geng, H. Chen, L.-T. Xu, Y.-C. Xie, D.-J. Wang, H.-W. Gu and X.-M. Wang, Carbon Quantum Dots: Preparation, Optical Properties, and Biomedical Applications, Mater. Today Adv., 2023, 18, 100376, DOI:10.1016/j.mtadv.2023.100376.
- A. Mozdbar, A. Nouralishahi, S. Fatemi and F. S. Talatori, The Impact of Carbon Quantum Dots (CQDs) on the Photocatalytic Activity of TiO2 under UV and Visible Light, J. Water Proc. Eng., 2023, 51, 103465, DOI:10.1016/j.jwpe.2022.103465.
- H. Liang, X. Tai, Z. Du and Y. Yin, Enhanced Photocatalytic Activity of ZnO Sensitized by Carbon Quantum Dots and Application in Phenol Wastewater, Opt. Mater., 2020, 100, 109674, DOI:10.1016/j.optmat.2020.109674.
- J. Xu, M. Jin, X. Shi, Q. Li, C. Gan and W. Yao, Preparation of TiSi2 Powders with Enhanced Lithium-Ion Storage via Chemical Oven Self-Propagating High-Temperature Synthesis, Nanomaterials, 2021, 11(9), 2279, DOI:10.3390/nano11092279.
- L. Zhang, X. Yang, Z. Yin and L. Sun, A Review on Carbon Quantum Dots: Synthesis, Photoluminescence Mechanisms and Applications, Luminescence, 2022, 37(10), 1612–1638, DOI:10.1002/bio.4351.
- S. Azam, Mn/Cu/O/Chitosan Nanocomposites: Hydrothermal Synthesis, Characterization, and Its Application for Adsorptive Removal of Methylene Blue from Wastewater, J. Nanostruct., 2022, 12(3), 746–753, DOI:10.22052/JNS.2022.03.027.
- J.-J. Xu, Y.-N. Lu, F.-F. Tao, P.-F. Liang and P.-A. Zhang, ZnO Nanoparticles Modified by Carbon Quantum Dots for the Photocatalytic Removal of Synthetic Pigment Pollutants, ACS Omega, 2023, 8(8), 7845–7857, DOI:10.1021/acsomega.2c07591.
- J. Zhang, Q. Liu, J. Wang, H. He, F. Shi, B. Xing, J. Jia, G. Huang and C. Zhang, Facile Preparation of Carbon Quantum Dots/TiO2 Composites at Room Temperature with Improved Visible-Light Photocatalytic Activity, J. Alloys Compd., 2021, 869, 159389, DOI:10.1016/j.jallcom.2021.159389.
- Y. Kaneko and M. Nagumo, Intermediate Phase in Synthesis of TiSi2 by Mechanical Alloying and Annealing, Mater. Sci. Eng., A, 2004, 375–377, 825–828, DOI:10.1016/j.msea.2003.10.089.
- Y. Xie, G. Ali, S. H. Yoo and S. O. Cho, Sonication-Assisted Synthesis of CdS Quantum-Dot-Sensitized TiO 2 Nanotube Arrays with Enhanced Photoelectrochemical and Photocatalytic Activity, ACS Appl. Mater. Interfaces, 2010, 2(10), 2910–2914, DOI:10.1021/am100605a.
- M. Li, M. Wang, L. Zhu, Y. Li, Z. Yan, Z. Shen and X. Cao, Facile Microwave Assisted Synthesis of N-Rich Carbon Quantum Dots/Dual-Phase TiO2 Heterostructured Nanocomposites with High Activity in CO2 Photoreduction, Appl. Catal., B, 2018, 231, 269–276, DOI:10.1016/j.apcatb.2018.03.027.
- T. Shen, Q. Wang, Z. Guo, J. Kuang and W. Cao, Hydrothermal Synthesis of Carbon Quantum Dots Using Different Precursors and Their Combination with TiO2 for Enhanced Photocatalytic Activity, Ceram. Int., 2018, 44(10), 11828–11834, DOI:10.1016/j.ceramint.2018.03.271.
- S. Y. Chen, Z. X. Shen, K. Li, A. K. See and L. H. Chan, Synthesis and Characterization of Pure C40 TiSi2, Appl. Phys. Lett., 2000, 77(26), 4395–4397, DOI:10.1063/1.1329864.
- D. K. Dang, C. Sundaram, Y.-L. T. Ngo, W. M. Choi, J. S. Chung, E. J. Kim and S. H. Hur, Pyromellitic Acid-Derived Highly Fluorescent N-Doped Carbon Dots for the Sensitive and Selective Determination of 4-Nitrophenol, Dyes Pigm., 2019, 165, 327–334, DOI:10.1016/j.dyepig.2019.02.029.
- Y. Liu, W. Li, P. Wu, C. Ma, X. Wu, M. Xu, S. Luo, Z. Xu and S. Liu, Hydrothermal Synthesis of Nitrogen and Boron Co-Doped Carbon Quantum Dots for Application in Acetone and Dopamine Sensors and Multicolor Cellular Imaging, Sens. Actuators, B, 2019, 281, 34–43, DOI:10.1016/j.snb.2018.10.075.
- Y. Wang, W. Weng, H. Xu, Y. Luo, D. Guo, D. Li and D. Li, Negatively Charged Molybdate Mediated Nitrogen-Doped Graphene Quantum Dots as a Fluorescence Turn on Probe for Phosphate Ion in Aqueous Media and Living Cells, Anal. Chim. Acta, 2019, 1080, 196–205, DOI:10.1016/j.aca.2019.07.023.
- H. Yang, L. He, S. Pan, H. Liu and X. Hu, Nitrogen-Doped Fluorescent Carbon Dots for Highly Sensitive and Selective Detection of Tannic Acid, Spectrochim. Acta, Part A, 2019, 210, 111–119, DOI:10.1016/j.saa.2018.11.029.
- X. Jiang, D. Qin, G. Mo, J. Feng, C. Yu, W. Mo and B. Deng, Ginkgo Leaf-Based Synthesis of Nitrogen-Doped Carbon Quantum Dots for Highly Sensitive Detection of Salazosulfapyridine in Mouse Plasma, J. Pharm. Biomed. Anal., 2019, 164, 514–519, DOI:10.1016/j.jpba.2018.11.025.
- B. Huang, X. Jia, Y. Li, A. Zheng and Y. Sun, Chiral Mesoporous Silicates Immobilizing Titanium Dioxide for Catalytic Asymmetric Epoxidation of Alkenes, Catal. Surv. Asia, 2017, 21(1), 13–27, DOI:10.1007/s10563-016-9222-x.
- W. S. Ahn, K. K. Kang and K. Y. Kim, Synthesis of TS-1 by Microwave Heating of Template-Impregnated SiO2–TiO2 Xerogels, Catal. Lett., 2001, 72(3/4), 229–232, DOI:10.1023/A:1009084011436.
- X. Li, J. Niu, J. Zhang, H. Li and Z. Liu, Labeling the Defects of Single-Walled Carbon Nanotubes Using Titanium Dioxide Nanoparticles, J. Phys. Chem. B, 2003, 107(11), 2453–2458, DOI:10.1021/jp026887y.
- Y. Deng, M. Chen, G. Chen, W. Zou, Y. Zhao, H. Zhang and Q. Zhao, Visible–Ultraviolet Upconversion Carbon Quantum Dots for Enhancement of the Photocatalytic Activity of Titanium Dioxide, ACS Omega, 2021, 6(6), 4247–4254, DOI:10.1021/acsomega.0c05182.
- D. Chu, C. Zhang, P. Yang, Y. Du and C. Lu, WS2 as an Effective Noble-Metal Free Cocatalyst Modified TiSi2 for Enhanced Photocatalytic Hydrogen Evolution under Visible Light Irradiation, Catalysts, 2016, 6(9), 136, DOI:10.3390/catal6090136.
- Y. Liu, S. Roy, S. Sarkar, J. Xu, Y. Zhao and J. Zhang, A Review of Carbon Dots and Their Composite Materials for Electrochemical Energy Technologies, Carbon Energy, 2021, 3(5), 795–826, DOI:10.1002/cey2.134.
- R. K. Upadhyay, N. Soin and S. S. Roy, Role of Graphene/Metal Oxide Composites as Photocatalysts, Adsorbents and Disinfectants in Water Treatment: A Review, RSC Adv., 2014, 4(8), 3823–3851, 10.1039/C3RA45013A.
- A. Kaur, P. Chahal and T. Hogan, Selective Fabrication of SiC/Si Diodes by Excimer Laser Under Ambient Conditions, IEEE Electron Device Lett., 2016, 37(2), 142–145, DOI:10.1109/LED.2015.2508479.
- J. Guo, K. Song, B. Wu, X. Zhu, B. Zhang and Y. Shi, Atomically Thin SiC Nanoparticles Obtained via Ultrasonic Treatment to Realize Enhanced Catalytic Activity for the Oxygen Reduction Reaction in Both Alkaline and Acidic Media, RSC Adv., 2017, 7(37), 22875–22881, 10.1039/C7RA01701D.
- J. Zhang, Y. Cao, P. Zhao, T. Xie, Y. Lin and Z. Mu, Visible-Light-Driven Pollutants Degradation with Carbon Quantum Dots/N-TiO2 under Mild Condition: Facile Preparation, Dramatic Performance and Deep Mechanism Insight, Colloids Surf., A, 2020, 601, 125019, DOI:10.1016/j.colsurfa.2020.125019.
- H. Ouyang, H. Huang, H. Wang and X. Zheng, The Morphology Evolution of Nitrogen-Doped Carbon Quantum Dots/Hollow TiO2 Composites and Their Applications in Photocatalysis, J. Mater. Sci., 2020, 55(3), 976–989, DOI:10.1007/s10853-019-03991-x.
- M. Zubair, I.-H. Svenum, M. Rønning and J. Yang, Core-Shell Nanostructures of Graphene-Wrapped CdS Nanoparticles and TiO2 (CdS@G@TiO2): The Role of Graphene in Enhanced Photocatalytic H2 Generation, Catalysts, 2020, 10(4), 358, DOI:10.3390/catal10040358.
- J. Zhang, Q. Liu, J. Wang, H. He, F. Shi, B. Xing, J. Jia, G. Huang and C. Zhang, Facile Preparation of Carbon Quantum Dots/TiO2 Composites at Room Temperature with Improved Visible-Light Photocatalytic Activity, J. Alloys Compd., 2021, 869, 159389, DOI:10.1016/j.jallcom.2021.159389.
- A. Mozdbar, A. Nouralishahi, S. Fatemi and F. S. Talatori, The Impact of Carbon Quantum Dots (CQDs) on the Photocatalytic Activity of TiO2 under UV and Visible Light, J. Water Proc. Eng., 2023, 51, 103465, DOI:10.1016/j.jwpe.2022.103465.
- Y. Zhang, Z.-R. Tang, X. Fu and Y.-J. Xu, TiO2− Graphene Nanocomposites for Gas-Phase Photocatalytic Degradation of Volatile Aromatic Pollutant: Is TiO2− Graphene Truly Different from Other TiO2− Carbon Composite Materials?, ACS Nano, 2010, 4(12), 7303–7314, DOI:10.1021/nn1024219.
- E. Jimenez-Relinque, I. Llorente and M. Castellote, TiO2 Cement-Based Materials: Understanding Optical Properties and Electronic Band Structure of Complex Matrices, Catal. Today, 2017, 287, 203–209, DOI:10.1016/j.cattod.2016.11.015.
- C.-H. Ri, Y.-S. Kim, U.-G. Jong, Y.-H. Kye, S.-H. Ryang and C.-J. Yu, First-Principles Study on Structural, Electronic and Optical Properties of Perovskite Solid Solutions KB 1−x Mg x I 3 (B = Ge, Sn) toward Water Splitting Photocatalysis, RSC Adv., 2021, 11(42), 26432–26443, 10.1039/D1RA04534B.
- P. K. Prabhakaran, S. Balu, G. Sridharan, D. Ganapathy and A. K. Sundramoorthy, Development of Eco-Friendly CQDs/TiO 2 Nanocomposite for Enhanced Photocatalytic Degradation of Methyl Orange Dye, Eng. Res. Express, 2025, 7(1), 015002, DOI:10.1088/2631-8695/ada553.
- S. Tong, J. Zhou, L. Ding, C. Zhou, Y. Liu, S. Li, J. Meng, S. Zhu, S. Chatterjee and F. Liang, Preparation of Carbon Quantum Dots/TiO2 Composite and Application for Enhanced Photodegradation of Rhodamine B, Colloids Surf., A, 2022, 648, 129342, DOI:10.1016/j.colsurfa.2022.129342.
- H. Razavi-Khosroshahi, K. Edalati, J. Wu, Y. Nakashima, M. Arita, Y. Ikoma, M. Sadakiyo, Y. Inagaki, A. Staykov, M. Yamauchi, Z. Horita and M. Fuji, High-Pressure Zinc Oxide Phase as Visible-Light-Active Photocatalyst with Narrow Band Gap, J. Mater. Chem. A, 2017, 5(38), 20298–20303, 10.1039/C7TA05262F.
- M. H. Elsayed, M. Abdellah, A. Z. Alhakemy, I. M. A. Mekhemer, A. E. A. Aboubakr, B.-H. Chen, A. Sabbah, K.-H. Lin, W.-S. Chiu, S.-J. Lin, C.-Y. Chu, C.-H. Lu, S.-D. Yang, M. G. Mohamed, S.-W. Kuo, C.-H. Hung, L.-C. Chen, K.-H. Chen and H.-H. Chou, Overcoming Small-Bandgap Charge Recombination in Visible and NIR-Light-Driven Hydrogen Evolution by Engineering the Polymer Photocatalyst Structure, Nat. Commun., 2024, 15(1), 707, DOI:10.1038/s41467-024-45085-6.
- K. V. Kumar, K. Porkodi and F. Rocha, Langmuir–Hinshelwood Kinetics – A Theoretical Study, Catal. Commun., 2008, 9(1), 82–84, DOI:10.1016/j.catcom.2007.05.019.
- T. Nguyen Thi Thu, N. Nguyen Thi, V. Tran Quang, K. Nguyen Hong, T. Nguyen Minh and H. Le Thi, N. Synthesis, Characterisation, and Effect of PH on Degradation of Dyes of Copper-Doped TiO 2, J. Exp. Nanosci., 2016, 11(3), 226–238, DOI:10.1080/17458080.2015.1053541.
- M. Shafaee, E. K. Goharshadi, M. Mashreghi and M. Sadeghinia, TiO 2 Nanoparticles and TiO 2 @graphene Quantum Dots Nanocomposites as Effective Visible/Solar Light Photocatalysts, J. Photochem. Photobiol., A, 2018, 357, 90–102, DOI:10.1016/j.jphotochem.2018.02.019.
- R. Wang, K.-Q. Lu, Z.-R. Tang and Y.-J. Xu, Recent Progress in Carbon Quantum Dots: Synthesis, Properties and Applications in Photocatalysis, J. Mater. Chem. A, 2017, 5(8), 3717–3734, 10.1039/C6TA08660H.
- Y. J. Liu, H. L. Wang, S. J. Li, S. G. Wang, W. J. Wang, W. T. Hou, Y. L. Hao, R. Yang and L. C. Zhang, Compressive and Fatigue Behavior of Beta-Type Titanium Porous Structures Fabricated by Electron Beam Melting, Acta Mater., 2017, 126, 58–66, DOI:10.1016/j.actamat.2016.12.052.
- X. Hu, W. Han, M. Zhang, D. Li and H. Sun, Enhanced Adsorption and Visible-Light Photocatalysis on TiO2 with in Situ Formed Carbon Quantum Dots, Environ. Sci. Pollut. Res., 2022, 29(37), 56379–56392, DOI:10.1007/s11356-022-19810-x.
- S. Ashok, N. Kumaresan, H. C. D. Souza, T. F. de Oliveira and V. Ganesh, An Investigation on G-C3N4/ZnS/SnO2 Ternary Nanocomposites for Electrochemical Alkaline Water Splitting and Photocatalytic Methylene Blue Decomposition Reactions, J. Solid State Electrochem., 2025, 29(2), 389–400, DOI:10.1007/s10008-024-06099-8.
- T. V. H. Luu, V. C. Nguyen, T. D. Thuy Tran, V. D. Doan, T. L. Nguyen, N. X. Dung and H. P. Dang, A Novel 3D Fe 2 O 3 @ZnBi 2 O 4 n–p Heterojunction with High Photocatalytic Activity under Visible Light, Nanoscale Adv., 2025, 7(10), 2942–2954, 10.1039/D4NA01039F.
- M. N. Saleem, A. Shah, N. Ullah, J. Nisar and F. J. Iftikhar, Detection and Degradation Studies of Nile Blue Sulphate Using Electrochemical and UV-Vis Spectroscopic Techniques, Catalysts, 2023, 13(1), 141, DOI:10.3390/catal13010141.
- B. Sarwan, B. Pare and A. D. Acharya, Heterogeneous Photocatalytic Degradation of Nile Blue Dye in Aqueous BiOCl Suspensions, Appl. Surf. Sci., 2014, 301, 99–106, DOI:10.1016/j.apsusc.2014.01.136.
- W. Yang, K. Ding, G. Chen, H. Wang and X. Deng, Synergistic Multisystem Photocatalytic Degradation of Anionic and Cationic Dyes Using Graphitic Phase Carbon Nitride, Molecules, 2023, 28(6), 2796, DOI:10.3390/molecules28062796.
- S. Debnath, N. Ballav, H. Nyoni, A. Maity and K. Pillay, Optimization and Mechanism Elucidation of the Catalytic Photo-Degradation of the Dyes Eosin Yellow (EY) and Naphthol Blue Black (NBB) by a Polyaniline-Coated Titanium Dioxide Nanocomposite, Appl. Catal., B, 2015, 163, 330–342, DOI:10.1016/j.apcatb.2014.08.011.
- W. Albuquerque, P. Trigueiro, B. V. Silva, L. Neves, L. C. Almeida and R. R. Peña-Garcia, A Novel RuO2@ZnO-Alginate-Halloysite Composite for the Effective Degradation of Eosin Yellow Dye and Ciprofloxacin Drug, Mater. Res. Bull., 2025, 182, 113178, DOI:10.1016/j.materresbull.2024.113178.
- Y. Shao, D. You, Y. Wan, Q. Cheng and Z. Pan, An Investigation on a WO3/MoO3−x Heterojunction Photocatalyst for Excellent Photocatalytic Performance and Enhanced Molecular Oxygen Activation Ability, React. Chem. Eng., 2024, 9(1), 70–81, 10.1039/D3RE00396E.
- F. Zhang, Y. Peng, X. Yang, Z. Li and Y. Zhang, Enhanced Photo-Assisted Fenton Degradation of Antibiotics over Iron-Doped Bi-Rich Bismuth Oxybromide Photocatalyst, Nanomaterials, 2022, 13(1), 188, DOI:10.3390/nano13010188.
- H. Yu, Y. Zhao, C. Zhou, L. Shang, Y. Peng, Y. Cao, L.-Z. Wu, C.-H. Tung and T. Zhang, Carbon Quantum Dots/TiO2 Composites for Efficient Photocatalytic Hydrogen Evolution, J. Mater. Chem. A, 2014, 2(10), 3344, 10.1039/c3ta14108j.
- X. Zeng, Z. Wang, N. Meng, D. T. McCarthy, A. Deletic, J. Pan and X. Zhang, Highly Dispersed TiO2 Nanocrystals and Carbon Dots on Reduced Graphene Oxide: Ternary Nanocomposites for Accelerated Photocatalytic Water Disinfection, Appl. Catal., B, 2017, 202, 33–41, DOI:10.1016/j.apcatb.2016.09.014.
- G. Rajender, J. Kumar and P. K. Giri, Interfacial Charge Transfer in Oxygen Deficient TiO2-Graphene Quantum Dot Hybrid and Its Influence on the Enhanced Visible Light Photocatalysis, Appl. Catal., B, 2018, 224, 960–972, DOI:10.1016/j.apcatb.2017.11.042.
- B. Sarwan, B. Pare and A. D. Acharya, Synthesis of Mn/NiO and Mn/BiOCl Nanoparticles for Degradation of Nile Blue Dye Contaminated Water under Visible Light Illumination, Part. Sci. Technol., 2020, 38(6), 659–666, DOI:10.1080/02726351.2019.1570991.
- K. Vignesh, A. Suganthi, M. Rajarajan and R. Sakthivadivel, Visible Light Assisted Photodecolorization of Eosin-Y in Aqueous Solution Using Hesperidin Modified TiO2 Nanoparticles, Appl. Surf. Sci., 2012, 258(10), 4592–4600, DOI:10.1016/j.apsusc.2012.01.035.
- M. U. Sadiq, A. Shah, A. Zahid, A. Javaid and F. J. Iftikhar, Nanosensor for the Detection of Eosin Yellow and Its Photocatalytic Degradation Using Phytosynthesized Cerium Oxide Nanoparticles, RSC Adv., 2025, 15(6), 4095–4110, 10.1039/D4RA08231A.
- W. Wang, Y. Ni and Z. Xu, One-Step Uniformly Hybrid Carbon Quantum Dots with High-Reactive TiO2 for Photocatalytic Application, J. Alloys Compd., 2015, 622, 303–308, DOI:10.1016/j.jallcom.2014.10.076.
- R. Liu, H. Li, L. Duan, H. Shen, Y. Zhang and X. Zhao, In Situ Synthesis and Enhanced Visible Light Photocatalytic Activity of C-TiO 2 Microspheres/Carbon Quantum Dots, Ceram. Int., 2017, 43(12), 8648–8654, DOI:10.1016/j.ceramint.2017.03.184.
- J. Zhang, Q. Liu, H. He, F. Shi, G. Huang, B. Xing, J. Jia and C. Zhang, Coal Tar Pitch as Natural Carbon Quantum Dots Decorated on TiO2 for Visible Light Photodegradation of Rhodamine B, Carbon, 2019, 152, 284–294, DOI:10.1016/j.carbon.2019.06.034.
- W. Liu, Y. Li, F. Liu, W. Jiang, D. Zhang and J. Liang, Visible-Light-Driven Photocatalytic Degradation of Diclofenac by Carbon Quantum Dots Modified Porous g-C3N4: Mechanisms, Degradation Pathway and DFT Calculation, Water Res., 2019, 151, 8–19, DOI:10.1016/j.watres.2018.11.084.
- Y. Zeng, D. Chen, T. Chen, M. Cai, Q. Zhang, Z. Xie, R. Li, Z. Xiao, G. Liu and W. Lv, Study on Heterogeneous Photocatalytic Ozonation Degradation of Ciprofloxacin by TiO2/Carbon Dots: Kinetic, Mechanism and Pathway Investigation, Chemosphere, 2019, 227, 198–206, DOI:10.1016/j.chemosphere.2019.04.039.
- J. Ke, X. Li, Q. Zhao, B. Liu, S. Liu and S. Wang, Upconversion Carbon Quantum Dots as Visible Light Responsive Component for Efficient Enhancement of Photocatalytic Performance, J. Colloid Interface Sci., 2017, 496, 425–433, DOI:10.1016/j.jcis.2017.01.121.
- Z. W. Heng, W. C. Chong, Y. L. Pang and L. C. Sim, Photocatalytic Degradation of Methylene Blue under Visible Light Using Carbon Dot/Titanium Dioxide Nanohybrid, IOP Conf. Ser.:Mater. Sci. Eng., 2020, 991(1), 012092, DOI:10.1088/1757-899X/991/1/012092.
- M. Sun, X. Ma, X. Chen, Y. Sun, X. Cui and Y. Lin, A Nanocomposite of Carbon Quantum Dots and TiO 2 Nanotube Arrays: Enhancing Photoelectrochemical and Photocatalytic Properties, RSC Adv., 2014, 4(3), 1120–1127, 10.1039/C3RA45474F.
- J. Zhang, M. Kuang, J. Wang, R. Liu, S. Xie and Z. Ji, Fabrication of Carbon Quantum Dots/TiO2/Fe2O3 Composites and Enhancement of Photocatalytic Activity under Visible Light, Chem. Phys. Lett., 2019, 730, 391–398, DOI:10.1016/j.cplett.2019.06.011.
- J. Zhang, X. Zhang, S. Dong, X. Zhou and S. Dong, N.-Doped Carbon Quantum Dots/TiO2 Hybrid Composites with Enhanced Visible Light Driven Photocatalytic Activity toward Dye Wastewater Degradation and Mechanism Insight, J. Photochem. Photobiol., A, 2016, 325, 104–110, DOI:10.1016/j.jphotochem.2016.04.012.
- B. Y. Yu and S.-Y. Kwak, Carbon Quantum Dots Embedded with Mesoporous Hematite Nanospheres as Efficient Visible Light-Active Photocatalysts, J. Mater. Chem., 2012, 22(17), 8345, 10.1039/c2jm16931b.
- X. Qu, M. Liu, J. Yang, C. Wang, H. Meng, S. Li, L. Shi and F. Du, A Novel Ternary TiO 2/CQDs/BiOX (X = Cl, Br, I) Heterostructure as Photocatalyst for Water Purification under Solar Irradiation, J. Solid State Chem., 2018, 264, 77–85, DOI:10.1016/j.jssc.2018.05.002.
| This journal is © The Royal Society of Chemistry 2025 |