Exploring spinel oxides from bimetallic to high-entropy with a focus on the structure and performance in the oxygen evolution reaction†
Mahalakshmi
Vedanarayanan
a,
Chandrasekaran
Pitchai
 a and
Chih-Ming
Chen
a and
Chih-Ming
Chen
 *ab
*ab
aDepartment of Chemical Engineering, National Chung Hsing University, 145 Xingda Rd., South District, Taichung 402202, Taiwan. E-mail: chencm@nchu.edu.tw
bInnovation and Development Center of Sustainable Agriculture, National Chung Hsing University, 145 Xingda Rd., South District, Taichung 402202, Taiwan
First published on 20th May 2025
Abstract
Spinel oxides have emerged as highly promising electrocatalysts for the oxygen evolution reaction (OER) due to their unique structural characteristics, exceptional stability, and remarkable electrochemical performance. Their well-ordered crystal structures promote efficient catalytic activity, while their durability ensures stability under demanding electrochemical conditions. These attributes make them ideal candidates for sustainable energy applications, such as water splitting and clean energy production. Additionally, the ability to modify their electronic and surface properties enables optimization, further enhancing their performance and offering a competitive alternative to noble metal-based electrocatalysts. This review begins with an overview of the basic mechanisms of the OER, such as the lattice oxygen mechanism (LOM) and the traditional adsorbate evolution mechanism (AEM). It then outlines the OER pathways in spinel electrocatalysts, focusing on key performance descriptors that determine their catalytic effectiveness. Thus, additional efforts are necessary to develop spinel materials that offer multiple advantages to fulfill these requirements. With continued research and innovation, the commercialization of OER catalysts shows significant potential for advancing energy conversion technologies.
1. Introduction
The drive for sustainable energy solutions has become a pivotal necessity in responding to the escalating global energy demands and the urgent environmental crises that threaten our future. With a high gravimetric energy density and no carbon footprint, hydrogen stands out among the other options as a clean and effective energy transporter.1–3 Leveraging these advantages, electrochemical water splitting has drawn a lot of interest as a feasible hydrogen production technique, providing a workable way to reduce reliance on fossil fuels and address the environmental and energy challenges.4–6Eqn (1)–(5) describe the two basic processes that produce hydrogen and oxygen in the potent process of electrochemical water splitting. At the cathode, hydrogen is produced via the hydrogen evolution reaction (HER), where protons are reduced to form hydrogen gas. Simultaneously, at the anode, water undergoes oxidation in the oxygen evolution reaction (OER), releasing oxygen gas. These two reactions work in tandem, unlocking the potential of water as a renewable source for clean energy, offering an efficient pathway to sustainable hydrogen production.7–10In acidic solutions:
| HER: 4H+(aq) + 4e− → 2H2(g) | (1) |
| OER: 2H2O(l) → O2(g) + 4H+(aq) + 4e− | (2) |
In neutral and alkaline solutions:
| HER: 4H2O(l) + 4e− → 2H2(g) + 4OH−(aq) | (3) |
| OER: 4OH−(aq) → O2(g) + 2H2O(l) + 4e− | (4) |
Overall water splitting reaction:
| 2H2O(l) → O2(g) + 2H2(g) | (5) |
According to theory, the water-splitting reaction requires a voltage of 1.23 V between the cathode and anode. To overcome the kinetic challenges, however, an additional potential referred to as the overpotential is required. Among the two reactions, the OER is the key limiting factor, as it involves a four-electron transfer and the creation of an O–O bond, whereas the HER only requires the transfer of two electrons.11,12 This highlights the importance of developing OER electrocatalysts capable of lowering the overpotential and improving the rate of reaction. Iridium (Ir), ruthenium (Ru), and their oxides are examples of noble-metal-based catalysts that have been extensively researched due to their exceptional OER performance and stability in both alkaline and acidic environments.13–15 However, their high cost and limited availability present significant challenges to large-scale commercialization.
The search for efficient, noble-metal-free electrocatalysts for the OER has intensified in recent years, with a focus on materials like carbon-based compounds, carbon nitride, and a wide range of transition-metal derivatives, including phosphides, nitrides, selenides, oxides, carbides, hydroxides/oxyhydroxides, sulfides, and phosphates.16–20 Among these, spinel transition-metal oxides have attracted significant attention owing to their cost-effectiveness, ease of synthesis, and structural and compositional versatility. These materials are particularly valued for their stability in alkaline environments, earth-abundant nature, and eco-friendly characteristics.21–23 Additionally, spinels exhibit fascinating properties such as excellent optical, electrical, magnetic, and catalytic behavior, attributed to their diverse compositions, specific electronic structures, and variable valence states.24
Spinel transition metal oxides possess a unique and highly ordered crystal structure, typically represented by AB2O4. In this structure, “A” and “B” are metal cations, often transition metals, that occupy specific sites within the crystal lattice. The distribution of metal cations among two types of interstitial sites—octahedral (B-sites) and tetrahedral (A-sites)—and the cubic close-packed arrangement of oxygen ions define the spinel structure's face-centered cubic (FCC) layout. In the ideal spinel structure, the “A” cations occupy the tetrahedral sites, while the “B” cations occupy the octahedral sites.25–28 This arrangement leads to a stable, highly symmetric crystal lattice with a distinct three-dimensional network that contributes to the material's stability, especially in various chemical environments.
The ability to substitute different metal ions at both A and B sites allows for a wide range of compositions, enabling the tailoring of the material's electronic, magnetic, and catalytic properties.29,30 The structural flexibility of spinels is one of the reasons they are so widely studied for different applications, including energy conversion and storage. The spinel structure's tunable nature, in combination with the diverse oxidation states of the metal cations, further enhances the material's catalytic activity, particularly in reactions like oxygen evolution and hydrogen evolution, making them ideal candidates for sustainable energy technologies as shown in Fig. 1.31
2. Mechanism of the oxygen evolution reaction
The oxygen evolution reaction has traditionally been described within the framework of metal-cation redox processes. However, recent studies have increasingly challenged this conventional understanding, revealing limitations in the adsorption mechanism that relies on metal-cation redox processes and prompting a re-evaluation of the underlying reaction pathways. For instance, pH-dependent OER activity observed on the RHE scale suggests the involvement of non-concerted proton–electron transfer steps, deviating from the typical concerted pathway. Additionally, growing evidence indicates the participation of lattice oxygen in the reaction mechanism, highlighting the limitations of surface-bound intermediate models. Moreover, enhancements in OER activity have been correlated with changes in the bulk electronic structure, such as elevated oxygen 2p-band centers and stronger metal–oxygen hybridization. These insights collectively point to a more complex reaction landscape, necessitating a broader mechanistic understanding beyond traditional adsorption-based frameworks.32Therefore, the OER in electrocatalysis progresses through two primary mechanisms, determined by the catalyst material and its interaction with adsorbed species. These mechanisms are the Adsorbate Evolution Mechanism (AEM) and the Lattice Oxygen Mediated Mechanism (LOM), as shown in Fig. 2. The AEM, considered the traditional OER pathway, relies on the adsorption and sequential oxidation of intermediates on the catalyst surface. The catalytic activity in this mechanism is strongly influenced by the binding strength of intermediates to active sites, which must be carefully balanced to achieve efficient reaction kinetics.33,34 Conversely, the LOM incorporates the lattice oxygen of the catalyst into the reaction via lattice oxygen redox chemistry, enabling direct oxygen release under OER potentials. This mechanism not only enhances the catalytic process by overcoming the limitations of the AEM but also similarly provides a broader understanding of the superior activity observed in certain solid-phase catalysts. By delving into these mechanisms and their key parameters, researchers can develop next-generation OER electrocatalysts with optimized performance and efficiency.35–37
2.1 Adsorbate evolution mechanism (AEM)
The AEM is one of the primary pathways for the OER, particularly on catalysts that strongly bind to oxygen-containing intermediates. In the AEM, the reaction proceeds through a series of intermediate species adsorbed on the surface of the catalyst, as shown in Fig. 2a. The hydroxyl intermediate (HO*) is created when OH− ions adsorb onto the catalyst's active sites (*), frequently at oxygen vacancy sites. This adsorbed hydroxyl group undergoes deprotonation, releasing H+ and e−, thereby forming an O* intermediate. The next step involves the reaction of this oxygen intermediate (O*) with another OH− ion, leading to the formation of a peroxo intermediate (HOO*). In the final step, the peroxo intermediate undergoes deprotonation to release molecular oxygen (O2), while regenerating the active site for further catalytic cycles.38–40 This process is governed by concerted proton-electron transfer (CPET) at each stage. The complete set of reactions is as follows:| OH− + * → HO* + e− | (6) |
| HO* → O* + e− + H+ | (7) |
| O* + OH− → HOO* + e− | (8) |
| HOO* → * + O2(g) + e− + H+ | (9) |
2.2 Lattice oxygen-mediated mechanism (LOM)
The LOM operates differently from the conventional AEM by involving two neighboring metal sites as shown in Fig. 2b, in contrast to the single metal site in the AEM. In the LOM, the reaction begins with the deprotonation of two hydroxyl intermediates (HO*) on the metal sites, resulting in the formation of two metal-oxo species. Instead of forming the peroxo intermediate (HOO*) that is observed in the AEM, these nearby metal-oxo species then directly couple to create an O–O bond. Ultimately, molecular oxygen (O2) is released, and the two-metal valency is reserved by hydroxide ions, regenerating active sites. A major advantage of the LOM is that, since HOO* is not an intermediate, it eliminates the scaling relationship between HO* and HOO*, which is typically a limiting factor in the AEM. As a result, the LOM enables water oxidation without the limitation of a minimal overpotential of about 0.37 V.41 Although the LOM was historically considered unlikely for heterogeneous water oxidation catalysts due to the assumption that the O–O coupling step requires a large activation barrier, recent research has shown that this mechanism can be effectively utilized, particularly in molecular catalysts involving transition metals. This progress has led to significant advancements in the design of catalysts that harness the LOM for an efficient OER.42–44For a better understanding, Fig. 3 offers a detailed overview of the proposed LOM and AEM pathways for NiFe2O4 and CoFe2O4 during the OER. It highlights the distinct processes involved in O–O bond formation in spinel oxides, which are known for their unique catalytic properties in the OER. In the LOM, O–O bond formation occurs through direct coupling between an oxygen atom adsorbed on a metal site and a lattice oxygen atom. Conversely, the AEM pathway involves O–O bond formation solely on oxygen atoms adsorbed on metal sites. These mechanisms are crucial for understanding the efficiency of spinel oxides, as the ability to transition between these pathways significantly affects the catalytic activity and stability of the materials during the OER.45
 | ||
| Fig. 3 Proposed LOM and AEM pathways for NiFe2O4 and CoFe2O4 during the OER. Reproduced with permission from ref. 45. Copyright 2022, American Chemical Society. | ||
2.3 Key parameters for assessing OER performance
The OER performance of electrocatalysts is highly dependent on experimental conditions. However, the lack of standardization in measurement protocols, apparatus setups, and operating parameters often leads to significant variation. This inconsistency makes it difficult to reliably compare the performance of different materials. In the following section, we discuss key factors that influence OER measurements, including experimental parameters, electrode configurations, and other essential components of the testing setup as shown in Fig. 4.46 | ||
| Fig. 4 Summary of important key parameters to evaluate OER activity, including experimental conditions and evaluation methods. Reproduced with permission from ref. 46. Copyright 2022, Wiley. | ||
2.3.1.1 Overpotential (η). Overpotential, represented by the symbol η, is the extra potential needed to overcome the inherent kinetic constraints in electrochemical water splitting for the OER. In comparison to the reversible hydrogen electrode (RHE), the OER's reversible thermodynamic potential is 1.23 V. The η at the desired current density, without iR compensation (eqn (10)), is given by
| ηOER = ERHE − 1.23 V | (10) |
In an ideal scenario, the applied potential would exactly match the equilibrium potential of a reaction. However, in reality, a higher potential is necessitated to overcome the energy barriers associated with the electron transfer processes. This additional potential can be described by the Nernst equation:
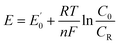 | (11) |
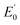 is the standard potential, and C0 and CR are the concentrations of the oxidized and reduced species, respectively. The discrepancy between the applied potential and the equilibrium potential is known as the overpotential, η. In practice, η is the extra potential that must be applied to reach a desired current.47,48
is the standard potential, and C0 and CR are the concentrations of the oxidized and reduced species, respectively. The discrepancy between the applied potential and the equilibrium potential is known as the overpotential, η. In practice, η is the extra potential that must be applied to reach a desired current.47,48
2.3.1.2 Tafel slope (b). An essential metric for understanding the reaction's kinetics and mechanism is the Tafel slope, which is especially useful for identifying the rate-limiting step. It is measured in mV dec−1 and can be obtained from the Tafel plot, which is generated by plotting log(j) against η from the polarization curve. The Tafel slope can be expressed using the following equation:
η = a + b![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) log(j) log(j) | (12) |
2.3.1.3 Exchange current density (j0). The rate of electron transfer between the electrode and the electrolyte in the absence of net electrolysis, usually at an overpotential (η) of 0 V, is represented by the exchange current density (j0), a crucial electrochemical parameter. It indicates the current density at which the rates of oxidation and reduction reactions at the electrodes are balanced, meaning that the forward and reverse reactions occur at the same rate. An inherent characteristic of the electrode surface, j0, is affected by the electrolyte, temperature, and electrocatalyst material. A high exchange current density typically suggests a high rate of electron transfer, which is often associated with a higher performing electrocatalyst for reactions like the OER. In practical terms, j0 can be determined by extrapolating the linear portion of the Tafel plot (log(j) vs. overpotential (η)) to the x-axis. A higher j0 generally indicates faster reaction kinetics and better overall catalyst performance.51
2.3.1.4 Turnover frequency (TOF). An essential activity indicator that offers a more thorough comprehension of an electrocatalyst's intrinsic OER performance is turnover frequency (TOF). It quantifies the rate at which oxygen is produced or reactants are consumed per unit time, relative to the amount of electrocatalyst present, at a given overpotential. The TOF is calculated using the equation:
| TOF = (j × NA)/(nF × Γ) | (13) |
2.3.1.5 Faradaic efficiency (FE). Faradaic efficiency (FE), which essentially reflects the conversion efficiency from electrons to oxygen, is a metric used to evaluate how effectively electrons are transferred during the OER. It is calculated by dividing the charge required to create oxygen (O2) by the total charge that has flowed through the circuit. The FE is expressed by the following equation:
| FE = αnF/Q | (14) |
2.3.1.6 Mass and specific activities. Under the presumption of smooth, planar surfaces, current density which is commonly represented in mA cm−2 traditionally only takes the electrodes' geometrical area into account. This presumption, however, is not true for the majority of electrocatalysts, particularly those with rough surfaces that are characteristic of nanostructured materials, which frequently have lower masses. Two more activity measures, bulk activity and specific activity, have been added by researchers to guarantee equitable comparisons between various electrocatalysts.
Mass activity, which is the current normalized by the catalyst loading and represented in amperes per gram (A g−1), can be used to evaluate the OER performance of electrocatalysts with various topographies and morphologies. However, specific activity normalizes the current using either the Brunauer–Emmett–Teller (BET) surface area or the electrochemical surface area (ECSA). When comparing electrocatalysts with different surface areas but comparable structures, specific activity is very useful. In contrast to geometrical area-normalized current density, ECSA-normalized current density is more responsive to changes in catalyst loading and more accurately represents the inherent catalytic characteristics of the electrocatalyst. It is crucial to remember that certain activity and mass are usually reported at a predetermined overpotential.55,56
2.3.1.7 Stability. Achieving exceptional stability is critical for catalysts to meet the demands of real-world applications. While transition metal compounds typically exhibit strong stability in alkaline electrolytes, their performance often deteriorates in acidic solutions due to degradation and dissolution, which limits their practical use. In response, considerable research has focused on improving the stability of these catalysts, with the structural modification of the working electrode playing a key role. Self-supporting electrodes, where catalysts are directly grown on the electrode surface, generally offer greater stability than those applied via drop-casting.
To evaluate the stability of OER catalysts, various techniques are employed. A widely used method is the accelerated degradation test (ADT), which involves subjecting the catalyst to several hundred or thousand cycles of cyclic voltammetry (CV) at high scan rates. Afterward, a linear sweep voltammetry (LSV) curve is recorded to determine the change in overpotential at a given current density. A smaller increase in overpotential is indicative of better stability. Furthermore, chronopotentiometry (U–t curves at a constant current density) or chronoamperometry (I–t curves at a constant potential) can be used to evaluate stability over extended time periods. A catalyst is considered stable for the OER if its current density remains consistent in chronoamperometry or if the overpotential increases only slightly in chronopotentiometry.57,58
To enhance catalyst performance, it is often beneficial to deposit the active material on a conductive and compatible substrate. A standard three-electrode setup typically involves a platinum (Pt) counter electrode, a reference electrode (such as Ag/AgCl, Hg/Hg2Cl2, or Hg/HgO), and the working electrode. The reference electrode is crucial for maintaining a stable potential and must be selected according to the test environment. For example, Ag/AgCl is not ideal under high-pH conditions because hydroxide ions can react to form Ag2O, which may clog the junction. In highly alkaline systems, more suitable reference electrodes include the reversible hydrogen electrode (RHE) or Hg/HgO in KOH solution. It is also important to avoid using metal counter electrodes in strong acids, as they are prone to corrosion and dissolution.61,62
Binders are essential for maintaining adhesion and facilitating charge and mass transport, but they must not interfere with the catalyst's activity. Nafion 117 is widely used due to its good conductivity and adhesion properties. In larger-scale applications, electrodes are commonly prepared using nickel mesh or plates. Techniques such as vacuum plasma spraying of Ni–Al alloys or electroplating Ni–Zn, followed by alkaline leaching, are used to generate porous, high-surface-area Raney nickel electrodes. Electrodeposition of Ni on a stainless steel mesh is another practical method. Catalyst loading must be optimized: low loading may cause incomplete coverage, while high loading can lead to aggregation, rough surfaces, and restricted mass transport, ultimately reducing the effective activity of the catalyst.63,64
3. Designing spinel-type electrocatalysts for the OER
Spinels are a diverse group of compounds characterized by the formula AB2X4, where A represents metals like Li, Mn, Zn, or Co, B includes elements such as Al, Cr, Fe, or Ni, and X refers to anions like O, S, or Se. The structure consists of metal A in smaller tetrahedral sites, metal B in larger octahedral sites, and the anion X at the corners of polyhedra. The oxidation states of A and B adjust to ensure charge neutrality, commonly forming configurations like A2+B3+2X2−4 or A4+B2+2X2−4. Due to the size difference in interstitial spaces, smaller cations tend to occupy the tetrahedral positions, while larger ones prefer the octahedral sites.65,66According to the work of Zhao et al., spinels exhibit various cation distributions, where A and B cations are distributed in different proportions within tetrahedral and octahedral sites, respectively. Based on these cation arrangements, spinels can be categorized into three types: normal, inverse, and complex spinels. To clearly represent these configurations, the formula A1−λBλ(AλB2−λ)X4 is introduced. In this notation, the cations before the parentheses occupy the tetrahedral positions, while those inside the parentheses reside in the octahedral sites. When λ = 0, the structure corresponds to a normal spinel (Fig. 5a), such as MgAl2O4 where Mg2+ occupies the tetrahedral sites and Al3+ resides in the octahedral ones. When λ = 1, it is an inverse spinel (Fig. 5b), such as NiFe2O4, where half of the Fe3+ ions are in tetrahedral sites, and the other half, along with Ni2+, are in octahedral sites represented as Fe(NiFe)O4. For values of 0 < λ < 1, the structure is considered a complex spinel (Fig. 5c). An example is CuAl2O4, where both Cu2+ and Al3+ ions partially occupy both tetrahedral and octahedral sites, denoted as Cu1−λAlλ(CuλAl2−λ)O4.67
 | ||
| Fig. 5 Representative structures of (a) a normal spinel (MgAl2O4), (b) an inverse spinel (NiFe2O4), and (c) a complex spinel (CuAl2O4) in different styles and views. The green and purple polyhedra correspond to octahedral and tetrahedral metal occupation sites, respectively. Reproduced with permission from ref. 67. Copyright 2017, American Chemical Society. | ||
Spinel oxides are ideal for the OER due to their high stability, excellent conductivity, and tunable properties. Their robust structure ensures durability in harsh electrochemical conditions, while the flexibility in metal incorporation allows optimization for enhanced catalytic performance. Additionally, spinel oxides provide multiple active sites for the OER and are made from cost-effective, earth-abundant materials, making them both efficient and sustainable for energy conversion applications.
3.1 Structural design
Although spinel-type materials have attracted attention, their OER performance is still below par. Increasing the number of exposed active sites is a well-established technique for increasing electrocatalytic activity. Tailoring the material's morphology to provide more accessible sites could improve catalytic efficiency. Hollow multimetallic spinel oxides are considered promising for this purpose. Research shows that octahedral sites, with their redox-active properties, are crucial for oxygen electrocatalysis, and the transition metals in these sites play a critical role in the process.68 Kim et al. have established a technique for the electrochemical preparation of uniform and well-adhering ZnCo2O4, a spinel material where Zn2+ replaces Co2+ exclusively in the Td sites of Co3O4, while Co3+ content in the octahedral (Oh) sites remains unchanged. In contrast, in NiCo2O4 and MnCo2O4, Ni and Mn predominantly occupy the octahedral sites. Additionally, since Ni and Mn can exist in mixed oxidation states (Ni2+/Ni3+ for Ni and Mn3+/Mn4+ for Mn), incorporating them into Co3O4 results in more complex modifications of the Co2+/Co3+ ratio. The stable oxidation state and fixed coordination of Zn2+ in the ZnCo2O4 spinel structure provide a unique opportunity to examine the catalytic role of Co ions in the tetrahedral sites during the OER. By comparing the OER performances of Co3O4 and ZnCo2O4, it may be possible to determine whether ZnCo2O4 could offer similar catalytic activity. If it does, ZnCo2O4 could present a more cost-effective and environmentally friendly alternative to Co3O4 for OER catalysis.693.2 Adjustment of valence states
The electrical characteristics of the metal active sites can be considerably altered by adding high-valence metal atoms to 3d transition metal-based materials. This enhances the interaction between the sites and intermediates associated with oxygen. Through air annealing, Luo et al. have synthesized W6+ doped spinel W–NiCo2O4-2 with a hierarchical sheet-like shape by introducing a high-valence oxyanion exchange method. The increased electrolyte contact, larger specific surface area, and more edge active sites of the hierarchical porous nanosheets enable efficient mass transport and charge transfer. By improving the eg orbital electron filling (eg = 1) at W sites and facilitating electron transfer from Co to W, W6+ doping increases the valence state of Co. This modification improves intrinsic activity by promoting the production of metal oxyhydroxide (M–OOH) active species during the OER process. Additionally, W6+ lowers the oxygen intermediates' free energy barrier and adsorption energy, greatly improving the reaction kinetics.703.3 Defect modification
By optimizing the electronic configuration, defect engineering is a potent technique to increase the number of active sites and improve catalyst conductivity. In spinel-type metal oxides, surface defects are primarily classified as oxygen (anion) defects and metal (cation) defects. Oxygen defects, the most extensively studied due to their low formation energy, can significantly alter the spinel surface's electronic structure and energy gap, thereby improving OER performance. However, maintaining stable oxygen defects in oxygen-rich environments poses a challenge. Metal defects, although less common due to their higher formation energy, offer distinct advantages. These include inducing semimetallic behavior and promoting significant charge delocalization, which can greatly enhance conductivity and provide unique catalytic properties.71 Liu et al. have synthesized Zn- and Al-substituted Co3O4, denoted as ZnxCo1−xAl2O4 (x = 0, 0.2, 0.4, 0.6, 0.8, and 1), and evaluated their OER activity. Zn2+ and Al3+ were chosen as substitution ions due to their non-variable valence states and preference for occupying tetrahedral and octahedral sites, respectively, over Co ions. While CoAl2O4 was anticipated to show the highest activity due to the catalytic efficiency of Co2+, the Zn0.6Co0.4Al2O4 composition exhibited the best OER performance among all samples. Additionally, Al ions stabilized Co2+ in the spinel structure, which remained intact even after 1200 OER cycles, highlighting its durability and stability.723.4 Engineering of composition and interfaces
Building on advancements in composition and interface engineering, significant efforts have focused on developing spinel-based heterostructures to enhance OER electrocatalysis. Jin et al. have achieved remarkable improvement in the intrinsic activity of Co3O4 spinel oxide by introducing CeO2 to form a Co3O4/CeO2 nanocomposite. The inclusion of CeO2 modulated the local bonding environment of Co2O4, facilitating the oxidation of Co3+ to catalytically active Co4+ species, which substantially boosted OER performance. As a result, the Co2O4/CeO2 composite delivered an impressive current density of 10 mA cm−2 at 423 mV in a 0.5 M H2SO4 electrolyte, as shown in Fig. 6. Beyond exceptional OER activity, this heterojunction demonstrated outstanding electrochemical stability under harsh acidic conditions.73,74 | ||
| Fig. 6 Electrochemical characterization of Co3O4 and Co3O4/CeO2 in 0.5 M H2SO4 solution. (a) iR-corrected CV curves of both catalysts; the inset shows the overpotential (with the error bar) required for each catalyst to reach a geometric catalytic current density of 10 mA cm−2 based on the averages of three individual electrodes. (b) Magnified CV curve of the Co3O4 catalyst that highlights the three pre-OER redox features and the corresponding C1, C2, and C3 cathodic peaks. Reproduced with permission from ref. 73. Copyright 2021, Nature Communications. | ||
3.5 Surface remodeling
The OER process often induces changes in surface composition or structure, particularly in spinel-type materials, leading to variations in electrocatalytic performance. Studies have shown that electrocatalysts undergo surface reconstruction during electrochemical reactions, forming in situ active species with higher oxidation states that significantly enhance OER performance. Xu et al. have developed a straightforward method to promote the self-reconstruction of CoAl2O4, thereby improving its OER activity. Their investigation revealed that Fe substitution facilitates surface reconstruction in CoAl2O4 and introduces a unique self-terminating reconstruction behavior in CoFe0.25Al1.74O4, ensuring stable surface chemistry. Electrochemical measurements confirmed that the Fe-substituted CoAl2O4 catalyst surpasses several perovskites and IrO2, showcasing its superior catalytic capabilities.75,764. Noble and non-noble metal-based spinel oxide frameworks
4.1 Ru and Ir-based spinel oxides
To improve the charge-transfer process, Fan et al. created RuO2/Co3O4–RuCo@NC composites with a lot of metal–semiconductor contacts. By partially oxidizing RuCo alloys embedded within a nitrogen-doped carbon (NC) framework, RuO2/Co3O4–RuCo@NC heterostructures were formed, featuring unique metal–semiconductor interfaces and favorable charge redistribution. Transmission electron microscopy (TEM) images show uniformly dispersed nanoparticles within the porous matrix. High-resolution TEM (HRTEM) further reveals distinct lattice fringes, indicating the high crystallinity of the nanoparticles. High-angle annular dark-field scanning TEM (HAADF-STEM) and elemental mapping (Fig. 7) confirm the uniform distribution of Co, Ru, O, C, and N elements throughout the structure at the nanoscale. | ||
| Fig. 7 Schematic illustration of the synthesis process of RuO2/Co3O4–RuCo@NC (a). TEM (b and c) and HRTEM (d) images of RuO2/Co3O4–RuCo@NC. Enlarged HRTEM images and FFT patterns in section I (e and f) and section II (g and h). HAADF-STEM elemental mappings (i) of RuO2/Co3O4–RuCo@NC. Reproduced with permission from ref. 77. Copyright 2019, American Chemical Society. | ||
As a result, the composite demonstrated remarkable electrocatalytic capabilities for the evolution of oxygen under acidic conditions. Remarkably, it achieved a low overpotential of 247 mV for the OER and a cell voltage of 1.66 V for overall water splitting at a current density of 10 mA cm−2. RuO2/Co3O4–RuCo@NC exhibits the lowest Tafel slope of 89 mV dec−1, outperforming RuO2 (179 mV dec−1), indicating enhanced OER kinetics in acidic media. Its long-term stability was confirmed via chronopotentiometry, maintaining a stable OER potential over 8 hours at a constant current density of 10 mA cm−2, as shown in Fig. 8.77
 | ||
| Fig. 8 (a) Polarization curves, (b) η10, and (c) Tafel plots of Co3O4–Co@NC, RuO2/Co3O4–RuCo@NC, and RuO2 for the OER in 0.5 M H2SO4 solution. (d) Comparison of η10 on various acidic OER electrocatalysts. (e) Chronopotentiometric curves of Co3O4–Co@NC, RuO2/Co3O4–RuCo@NC, and RuO2 at a constant current density of ∼10 mA cm−2. Reproduced with permission from ref. 77. Copyright 2019, American Chemical Society. | ||
Fonseca et al. have reported that Fe3O4/RuO2@NEU7 demonstrated outstanding OER performance. The catalyst reached a current density of 10 mA cm−2 in 1 M KOH with an ultralow overpotential of only 250 mV. According to the exchange current density data, this remarkable activity was ascribed to quick electron transfer and an optimum catalytic surface area. The synergistic interaction between RuO2 and Fe2O4, potentially driven by electrostatic or magnetic interactions, contributed to the enhanced performance. This synergy involved tuning charge density (oxidation states) and facilitating exchange interactions between similar spins in RuO2 and Fe3O4 with OER intermediates.78 In comparison to IrO2, which had a Tafel slope of 68.2 mV dec−1 and an onset potential of 1.48 V, Zaman et al.'s study of IrO2-decorated Co3O4 nanorods revealed a considerably steeper Tafel slope of 51.2 mV dec−1 and a 30 mV improvement in onset potential. This outstanding performance was attributed to the one-dimensional nanorod-shaped assembly of substrate particles and the synergistic impact of metal cations, which are driven by their electronic modulations.79 By incorporating atomically distributed Ir atoms into a spinel Co3O4 lattice, Liu et al. have created an acidic OER catalyst. This catalyst exhibited exceptional performance, achieving an overpotential as low as 226 mV at 10 mA cm−2 and an impressive TOF of 3.15 s−1 at η = 300 mV, outperforming commercial IrO2 by three orders of magnitude. Furthermore, it demonstrated outstanding durability, with a lifespan of up to 500 h at 10 mA cm−2 and exceptional corrosion resistance in acidic OER.80 According to the work of Wang et al., a Ru/RuO2–Co3O4 catalyst was synthesized by thermally treating a Ru/ZIF-67 precursor at 350 °C in air. SEM images reveal a porous structure, enhancing the catalyst's surface area, while TEM images show hollow, irregular particles around 200 nm in diameter. The 0.216 nm fringe spacing corresponds to the (002) facet of Ru, and the 0.255 nm spacing matches the (101) plane of RuO2. The FFT pattern shows diffraction spots from RuO2 (110) and Co3O4 (222) planes, as shown in Fig. 9.
 | ||
| Fig. 9 Phase and structural characterization: (a) XRD patterns of Co3O4 and Ru/RuO2–Co3O4, (b) TEM image, (c and g) HRTEM images, (d, e, h and i) magnified HRTEM images, (f) FFT pattern corresponding to the region in (c), (j) FFT pattern corresponding to the region in (g), and (k–n) elemental mapping of Ru/RuO2–Co3O4. Reproduced with permission from ref. 81. Copyright 2023, American Chemical Society. | ||
The catalyst demonstrated excellent durability, operating for over 19 hours with an overpotential of 226 mV at 10 mA cm−2, as shown in Fig. 10. It outperformed many recent catalysts and commercial RuO2 in acidic media. Key structural features, such as the hollow structure, oxygen vacancies, carbon coating, and the interface between Co3O4 and RuO2, contributed to its outstanding catalytic activity.81
 | ||
| Fig. 10 Electrochemical OER performance of Ru/RuO2–Co3O4 in 0.1 M HClO4. (a) Polarization curves, (b) Tafel plots, (c) Cdl values, and (d) Nyquist plots for Ru/RuO2–Co3O4 and references. (e) OER polarization curves of Ru/RuO2–Co3O4 and commercial RuO2 before and after 1000 cycles. (f) Electrochemical stability evaluation by chronopotentiometry at 10 mA cm−2. Reproduced with permission from ref. 81. Copyright 2023, American Chemical Society. | ||
A RuO2/(Co, Mn)3O4 nanocomposite with a low Ru concentration of 2.51 wt% has been created by Niu et al. as an effective OER catalyst for acidic media (0.5 M H2SO4). Despite the low Ru loading, the catalyst showed remarkable acidic OER activity, achieving an overpotential of 270 mV at 10 mA cm−2, and improved long-term stability when compared to the benchmark RuO2 catalyst.82 Shan et al. have investigated the electrocatalytic OER performance of Ir0.06Co2.94O4 in O2-saturated 0.1 M HClO4 electrolytes. According to rotating ring-disk electrode (RRDE) studies, the catalyst showed an onset potential of roughly 1.45 V vs. the reversible hydrogen electrode (RHE). It performed exceptionally well at an overpotential of 300 mV (1.53 V vs. RHE), with a noteworthy OER current density of 5.99 mA cm−2.83 Yin et al. have presented a new method for co-electrodepositing iridium single atoms (Ir–SAs) onto ultrathin NiCo2O4 porous nanosheets (Ir–NiCo2O4 NSs). The catalyst's OER activity and stability in acidic environments were greatly increased by the coupling of surface-exposed Ir–SAs with oxygen vacancies (Vo). With an ultralow overpotential of 240 mV at 10 mA cm−2 and sustained stability for 70 hours, the Ir–NiCo2O4 NSs showed exceptional performance. Their exceptional catalytic effectiveness was further demonstrated by the TOF of 1.13 and 6.70 s−1 at overpotentials of 300 and 370 mV, respectively.84
Using a glassy carbon electrode (GCE), Zhu et al. investigated the OER performance of commercial IrO2 and Co3O4 in addition to Ir–Co3O4 and as-synthesised Co3O4 in 0.5 M H2SO4. Ir–Co3O4 showed the best OER performance out of all the samples that were evaluated. Ir–Co3O4 needed an η of 236 mV to reach a current density of 10 mA cm−2, which was significantly less than that of Co3O4 (412 mV) and commercial Co3O4 (511 mV).85
4.2 (Ni, Zn, Cu, Fe, and Mn) incorporated Co3O4-based bimetallic spinel oxides
In recent years, bimetallic spinel oxides have emerged as highly promising candidates for the OER, often demonstrating performance that rivals or even surpasses conventional state-of-the-art (SoA) catalysts such as IrO2 and RuO2. Their superior activity is largely attributed to the synergistic interactions between dual metal centers, which effectively tune the electronic structure, enhance charge transfer capabilities, and optimize the binding energies of key reaction intermediates. The inherent mixed-valence nature of spinel structures ((A2+)(B3+)2O4) facilitates efficient electron hopping between divalent and trivalent cations, resulting in excellent electrical conductivity, a crucial factor in electrocatalytic processes. Compared to more complex multiatomic systems, bimetallic spinels offer a favorable balance of high catalytic efficiency, structural stability, and material simplicity. These attributes make them attractive, cost-effective alternatives to noble metal-based catalysts for practical and scalable water-splitting applications.86The incorporation of a second metal modulates the electronic band structure, optimizing the adsorption energies of key OER intermediates (*OH, *O, and *OOH) and lowering overpotentials. It also alters cation distribution between tetrahedral and octahedral sites, promoting oxygen vacancy formation and efficient electron transport. Additionally, redox synergy from mixed-valence states (e.g., Co3+/Co2+ and Fe3+/Fe2+) facilitates multi-electron transfer, while the second metal enhances structural integrity, minimizing degradation during prolonged electrolysis. Most spinels reported for the OER are Fe-based or Co-based compounds, typically classified according to the B-site cation as Fe-based (AFe2O4), Co-based (ACo2O4), and Mn-based (AMn2O4) spinels. By varying the A-site with different non-precious metals, diverse compositions with enhanced catalytic properties can be achieved. Fe-based spinels, known for their high conductivity, rich redox chemistry, and low toxicity, generally adopt an inverse spinel structure, where A2+ cations (e.g., Ni2+, Co2+, Mn2+, and Zn2+) occupy octahedral sites, while Fe3+ ions are distributed across both tetrahedral and octahedral sites. Their Fe3+/Fe2+ and A3+/A2+ redox couples contribute significantly to OER activity. Similarly, Co-based spinels (ACo2O4) have been widely studied, where Co3+ resides in octahedral sites and A2+ cations (e.g., Ni2+, Cu2+, Zn2+, and Mn2+) occupy tetrahedral sites. The addition of a second transition metal enhances electrical conductivity and introduces more redox-active sites, thereby boosting OER performance.87
 | ||
| Fig. 11 (A and C) SEM and TEM images of NiCo2O4 NNs and (B and D) NiCo2O4 NSs. Reproduced with permission from ref. 88. Copyright 2014, American Chemical Society. | ||
The overpotentials required to achieve 5 mA cm−2 and 10 mA cm−2 catalytic currents for NiCo2O4 NNs were about 165 mV and 323 mV lower than those for NiCo2O4 NSs and CoPi, respectively. The enhanced kinetics of NiCo2O4 NNs were evident from a lower Tafel slope of 292 mV dec−1, compared to 393 mV dec−1 for NiCo2O4 NSs. This suggests that the NiCo2O4 nanoneedles, with a more hydroxylated surface and greater amounts of physisorbed and chemisorbed water, contributed to superior water oxidation performance, as shown in Fig. 12.88
 | ||
| Fig. 12 (A) LSV, (B) controlled potential electrolysis, and (C and D) Tafel plots of NiCo2O4 NNs and NiCo2O4 NSs recorded in 1 M KOH solution at a scan rate of 5 mV s−1 with a three-electrode system applying SCE as the reference electrode. Reproduced with permission from ref. 88. Copyright 2014, American Chemical Society. | ||
Cui et al. have found that core–ring NiCo2O4 nanoplatelets exhibit excellent OER performance in alkaline electrolysis, with an overpotential of 0.315 V at 100 mA cm−2. The core–ring structure increases the active surface area and exposes more active Co atoms, enhancing OER efficiency.89 A NiCo2O4 spinel nanowire array catalyst was coated onto a rotating-disk electrode by Jin et al. and evaluated at 400 and 2500 rpm with a scan rate of 10 mV s−1 in an O2-saturated 0.1 M KOH solution. With a notable rise in anodic current at the same applied potential, the OER's onset potential was found to be approximately 0.60 V, 0.12 V lower than that of a pure carbon electrode. According to these findings, NiCo2O4 spinel nanowire arrays show promise as OER catalysts.90 Su et al. have attributed the enhanced OER electrocatalytic activity and stability of one-dimensional NiCo2O4 nanowire arrays (NWAs) to several factors. The NiCo2O4 NWAs' 1D ordered morphology facilitates electron transport, while the open spaces between the nanowires improve electrolyte diffusion, and their mesoporous structure increases the surface area for reactions. In the spinel lattice, Ni substitution increases conductivity and produces oxygen-deficient structures by replacing Co³⁺ with lower-valence Ni at octahedral positions. Furthermore, Ni-doped nanowire arrays improve ion intercalation and electrode material use by offering more active sites and effective diffusion paths for electrolyte ions. Cyclic voltammetry measurements showed that the ECSA of NiCo2O4 increased to 140 cm2, compared to 112.5 cm2 for Co3O4 and 63.75 cm2 for NiO. Finally, oxygen vacancies and structural distortions promote OH− adsorption, accelerating OER kinetics.91 Chen et al. have reported that the overpotential of NiCo2O4 nanoframes at a current density of 10 mA cm−2 is 265 mV, which surpasses that of Co3O4 nanoframes (319 mV) and NiO nanoframes (442 mV). This suggests that NiCo2O4 nanoframes exhibit superior catalytic activity due to the presence of nanosheets having a mesoporous structure.92
As seen in Fig. 13, Liu and colleagues created hierarchical ZnxCo3−xO4 nanostructures with secondary nanoneedles on primary rhombus-shaped pillars that provide excellent mechanical adhesion, high porosity, and active-site density. The structures achieved a low η of 0.32 V with a Tafel slope of 51 mV dec−1, as shown in Fig. 14. Their exceptional performance and stability were attributed to the optimized Co ion environment and hierarchical design, making them efficient electrocatalysts for water oxidation.93
 | ||
Fig. 13 SEM images of ZnxCo3−xO4 arrays: (A) pure Co3O4, (B) Zn/Co = 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4, (C) Zn/Co = 1 4, (C) Zn/Co = 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3, and (D) Zn/Co = 1 3, and (D) Zn/Co = 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2. The inset shows high-magnification SEM images taken from the as-obtained ZnxCo3−xO4 samples. Reproduced with permission from ref. 93. Copyright 2014, American Chemical Society. 2. The inset shows high-magnification SEM images taken from the as-obtained ZnxCo3−xO4 samples. Reproduced with permission from ref. 93. Copyright 2014, American Chemical Society. | ||
 | ||
Fig. 14 (A) Cyclic voltammograms of ZnxCo3−xO4 electrodes recorded at a potential sweep rate of 5 mV s−1 (B and C) State polarization curves recorded at a scan rate of 0.5 mV s−1 and their corresponding Tafel plots (a, Ir/C; b, Co3O4; c, ZnxCo3−xO4-1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4; d, ZnxCo3−xO4-1 4; d, ZnxCo3−xO4-1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3; and e, ZnxCo3−xO4-1 3; and e, ZnxCo3−xO4-1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2). (D) Chronoamperometric responses (percentage of retained current vs. operation time) of ZnxCo3−xO4-1 2). (D) Chronoamperometric responses (percentage of retained current vs. operation time) of ZnxCo3−xO4-1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 and Ir/C electrodes kept at 1.63 V vs. RHE in 1 M KOH solution. Reproduced with permission from ref. 93. Copyright 2014, American Chemical Society. 3 and Ir/C electrodes kept at 1.63 V vs. RHE in 1 M KOH solution. Reproduced with permission from ref. 93. Copyright 2014, American Chemical Society. | ||
Ramsundar et al. have demonstrated that nanostructured ZnxCo3−xO4 is an effective catalyst for the OER. The substitution of Co2+ with Zn2+ in Co3O4 increases the high-spin Co3+ content, which enhances OER performance. ZnxCo3−xO4 showed a low η of 250 mV and a turnover frequency of around 3 × 103 s−1.94 Menezes et al. have found that ZnCo2O4 outperforms traditional Co3O4 as an OER catalyst. It shows a reduced electrode potential of 1.66 V, lower than the 1.70 V of Co3O4. Furthermore, the Tafel slope for ZnCo2O4 is significantly smaller at 38.5 mV dec−1, highlighting its superior reaction kinetics for oxygen evolution.95 Han et al. have reported that the ZnxCo3−xO4 film demonstrates excellent stability and a low η of about 0.33 V (and 0.35 V at 20 mA cm−2) for 10 h, with a Tafel slope of 39 mV dec−1 for the OER. When compared to Co3O4, ZnxCo3−xO4 exhibits improved OER performance due to surface structural alterations and a greater density of accessible active Co3+ sites, which are made possible by the addition of Zn2+.96 Zhang et al. have shown that a ZnCo2O4 micro-spindle catalyst exhibits an η of 0.389 V, with a Tafel slope of 59.54 mV dec−1. The catalyst maintains stable OER performance over 7200 s without a significant increase in overpotential. Its impressive activity, low Tafel slope, and long-term stability highlight its potential for real-world applications.97
 | ||
| Fig. 15 (a and b) SEM and (c and d) TEM images of Vo-MnCo2O4 with different magnifications. (e and f) HRTEM images of Vo-MnCo2O4 (inset: FFT image). (g) HADDF image and (h–j) STEM-EDS mapping images of Mn, Co, and O of Vo-MnCo2O4. Reproduced with permission from ref. 106. Copyright 2021, Elsevier Ltd. | ||
 | ||
| Fig. 16 (a) The OER polarization curves in 0.1 M KOH, (b) corresponding overpotentials at 10 mA cm−2 and current densities at 1.68 V vs. RHE, and (c) Tafel plots. Reproduced with permission from ref. 106. Copyright 2021, Elsevier Ltd. | ||
4.3 (Ni, Co, Zn, Cu, and Mn) incorporated Fe3O4-based bimetallic spinel oxides
 | ||
| Fig. 17 (a–c) SEM images (different magnification) and (d) XRD pattern of CoFe2O4 NPs on CFP. (e–i) Elemental mapping analysis of CoFe2O4 NPs on CFP; (e) SEM image of the spot used for the elemental mapping: (f) Co map, (g) Fe map, (h) O map, and (i) C map. The scale bar for (f–i) is the same as that in (e). Reproduced with permission from ref. 112. Copyright 2015, American Chemical Society. | ||
 | ||
| Fig. 18 (a) Cyclic voltammetry (CV) measurement of CoFe2O4 NPs on CFP and bare CFP recorded at a scan rate of 20 mV s−1 in 1 M NaOH. Inset shows the zoomed-in current density versus RHE plot. (b) Tafel plot of the corresponding samples in (a). (c) Current density versus time of CoFe2O4 NPs on CFP measured at a fixed potential of 0.675 V versus Hg/HgORE in 1 M NaOH. The plots are shown without any iR correction. Reproduced with permission from ref. 112. Copyright 2015, American Chemical Society. | ||
Yan et al. have used NaBH4 in a straightforward reduction process to create CoFe2O4 nanosheets (NSs). The resulting reduced CoFe2O4 NSs are rich in oxygen-deficient sites, providing more active sites and enhanced conductivity compared to the pristine CoFe2O4 hollow nanospheres. These reduced nanosheets achieved an η of 320 mV for the OER.113
4.4 Ternary spinel oxides
Ramakrishnan et al. have synthesized a ternary metal-based inverse spinel oxide (ISO) catalyst, NiCrFeO4, supported on carbon, to enhance OER efficiency. This catalyst exhibits remarkable OER activity, achieving a low η of 264 mV and a Tafel slope of 45 mV dec−1. Additionally, it shows exceptional stability, with sustained performance over 40 h at a constant current density of 10 mA cm−2.124 Yan et al. have used nickel foam as the substrate and a straightforward hydrothermal process, followed by calcination, to create FeNiCo2O4 ternary mixed metal spinel oxides. OER tests reveal that FeNiCo2O4 exhibits exceptional electrocatalytic activity, with a low η of 350 mV. Furthermore, it demonstrates a smaller Tafel slope of 27 mV dec−1, outperforming other samples with varying Fe/Ni ratios, indicating its superior catalytic performance.125 Chen et al. have prepared cobalt-nickel-iron oxide catalysts electrochemically activated on 3D carbon fiber electrodes. These catalysts showed remarkable OER performance, with a low η of 232 mV, a Tafel slope of 37.6 mV dec−1, and prolonged duration stability, sustaining electrolysis for over 100 h.126 Karkera et al. have synthesized CoMnWO4via a hydrothermal method, which outperformed the state-of-the-art OER catalyst IrO2. It achieved a current density of 10 mA cm−2 with a lower η of 0.4 V, compared to IrO2 (0.46 V). Additionally, the catalyst exhibited an enhanced mass activity of 50 A g−1 and a TOF of 0.3526 s−1 at 0.4 V.127 Gupta et al. have highlighted the exceptional performance of LiNiAlO2, achieving activity levels at 1.56 V comparable to the benchmark IrO2 catalyst. With a Tafel slope of 44 mV dec−1, the catalyst demonstrated remarkable durability during linear sweep voltammetry and chronoamperometry tests, efficiently managing oxygen bubble accumulation.128 Zhu et al. have fabricated a layered LiCoFeO2 (LCFO2) catalyst through a straightforward sol–gel process, showcasing outstanding OER activity and stability under alkaline conditions. The catalyst delivered an overpotential of ≈0.34 V at 10 mA cm−2 and a Tafel slope of 50 mV dec−1. Its superior performance arises from the synergistic effects of Co and Fe, which optimize the electronic structure, generate abundant oxidative oxygen species (O22−/O−), enhance charge transfer, and improve O2 desorption. This study establishes LCFO2 as a highly efficient, durable, and affordable OER electrocatalyst.129 Lu et al. have proposed an electrochemical lithium tuning strategy in organic electrolytes to enhance the catalytic activity of materials for the OER in aqueous solutions. The optimized catalyst, LiCoNiFeO2, demonstrated exceptional performance with an onset potential of approximately 1.47 V vs. RHE and a low Tafel slope of ∼35 mV dec−1. This electrochemically delithiated material outperformed the benchmark iridium/carbon catalyst, showcasing its potential as an efficient OER electrocatalyst.130 Roy et al. have reported that the NiCoFeO4 catalyst, benefiting from the synergistic effect of optimally balanced Fe and Co, achieved a reduced η of 310 mV and maintained a constant current density for 22 h during chronoamperometric testing. The superior OER performance of NiCoFeO4 was attributed to its higher covalency, the electronic configuration of Co3+, and a high concentration of oxygen vacancies, which collectively enhanced its catalytic efficiency.131 To activate porous spinel NiFe2O4 nanowires with up to four different metal cation substitutions, Zhang et al. introduced a eutectic dealloying strategy. The selected area electron diffraction (SAED) pattern reveals concentric rings, confirming the polycrystalline nature of the NiFeXO4 nanowires. High-resolution TEM images display distinct lattice fringes with interplanar spacings of 2.50 Å and 2.07 Å, corresponding to the (311) and (400) planes of spinel oxides, respectively. Moreover, EDS elemental mapping confirms the uniform distribution of the substituted cations throughout the nanowire structure, as shown in Fig. 19. | ||
| Fig. 19 Microstructures of porous spinel NiFeXO4 nanowires. (a) TEM image of NiFeXO4 (X = Fe, Ni, Al, Mo, Co, Cr) porous nanowires at low magnification. (b and c) High-resolution HAADF-STEM image and SAED pattern of the NiFeXO4 porous nanowires. (e and f) Atomic resolution images show d spacings of two random areas labeled in (d). (h–n) EDS elemental mapping images of O, Fe, Ni, Al, Co, Cr, and Mo of the NiFeXO4 nanowire are shown in (g). Reproduced with permission from ref. 132. Copyright 2023, American Chemical Society. | ||
The overpotentials required to achieve a high current density of 300 mA cm−2 comparable to that used in commercial electrolyzers highlight the superior activity of NiFeXO4. It requires only 288 mV, which is approximately 150–205 mV lower than that of NiFeYO4 (438 mV) and NiFe2O4 (493 mV), as shown in Fig. 20. To further evaluate the catalytic kinetics, Tafel slopes were analyzed. NiFeXO4 exhibits the lowest Tafel slope at 53.3 mV dec−1, outperforming NiFeYO4 (58.4 mV dec−1), NiFe2O4 (65.5 mV dec−1), and NiFe-LDH (75.0 mV dec−1), indicating more favorable reaction kinetics, as shown in Fig. 20.132
 | ||
| Fig. 20 Electrochemical characterization of porous spinel NiFeXO4 nanowires. (a) OER polarization curves. (b) Overpotentials at typical current densities of 10 and 300 mA cm−2. (c) Tafel slope values. Reproduced with permission from ref. 132. Copyright 2023, American Chemical Society. | ||
4.5 Quaternary spinel oxides
Piñeiro-García et al. have investigated the catalytic performance of quaternary mixed oxides (X–Sn–Mo–Sb, where X = Mn, Fe, Co, or Ni) for the oxygen evolution reaction (OER) under both acidic and alkaline conditions. These oxides were fabricated on FTO glass substrates via atmospheric plasma spraying. While all X–MO coatings exhibited similar structural characteristics, subtle variations in surface morphology were noted among the different compositions, as shown in Fig. 21. | ||
| Fig. 21 (a) Coatings produced via SPPS using a solution precursor containing the desired cation composition. The mixed oxide coatings are produced by applying several layers followed by thermal annealing in air. (b) SEM images of the quaternary mixed oxides (X–Sn–Mo–Sb) deposited on FTO, from left to right: Mn-MO, Fe-MO, Co-MO, and Ni-MO. Reproduced with permission from ref. 133. Copyright 2024, American Chemical Society. | ||
The research focused on assessing the efficiency and stability of the X-MO (mixed oxides) coatings under diverse conditions to determine their suitability as OER catalysts. In acidic media, Co-MO showed the best performance, with overpotentials of 816 mV and 870 mV at current densities of 5 mA cm−2 (η5) and 10 mA cm−2 (η10), respectively, followed by Mn-MO at 940 mV for η5. Co-MO (80 mV dec−1) fared better than Ni-MO (121 mV dec−1), Mn-MO (133 mV dec−1), and Fe-MO (151 mV dec−1), according to the Tafel slope study. In alkaline media, Co-MO demonstrated excellent electrocatalytic performance with overpotentials of 440 mV and 520 mV at η10 and η50, respectively, while Mn-MO exhibited a higher overpotential of 750 mV at η10.
The Tafel slopes for the catalysts were 41 mV dec−1 for Co-MO, 57 mV dec−1 for Ni-MO, 77 mV dec−1 for Mn-MO, and 89 mV dec−1 for Fe-MO, establishing Co-MO as a reliable candidate for the oxygen evolution reaction across various pH conditions, as shown in Fig. 22.133
 | ||
Fig. 22 (a) X-MO coatings produced on FTO in 1 M KOH using a 1 mV s−1 scan rate, with iR-corrected polarization curves (dashed lines indicate the activity after the stability test). (b) Tafel slopes evaluated at a 1 mV s−1 scan rate in KOH = 1 M. (c) Nyquist plot analysis by electrochemical impedance spectroscopy of X-MOs in KOH = 1 M at 1.7 V vs. RHE; the corresponding circuit model is displayed in the inset. (d) SEM pictures. (e) Normalized metal content in relation to Sb. The atomic ratios are 1.14![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.08 0.08![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.68 0.68![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2, 1.22 2, 1.22![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.17 0.17![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.77 0.77![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2, 1.12 2, 1.12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.31 0.31![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.73 0.73![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2, and 1.12 2, and 1.12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.32 0.32![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.67 0.67![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 for X 2 for X![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Sn Sn![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Mo Mo![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Sb, where X = Mn, Fe, Co, or Ni. Reproduced with permission from ref. 133. Copyright 2024, American Chemical Society. Reproduced with permission from ref. 133. Copyright 2024, American Chemical Society. Sb, where X = Mn, Fe, Co, or Ni. Reproduced with permission from ref. 133. Copyright 2024, American Chemical Society. Reproduced with permission from ref. 133. Copyright 2024, American Chemical Society. | ||
4.6 High entropy spinel oxides
High-entropy oxides (HEOs) have emerged as a promising new class of electrocatalysts. Comprising five or more equimolar cations in a single-phase structure, these materials known as high-entropy materials (HEMs) have garnered significant attention in fields such as energy storage, fuel cells, and catalysis. Their appeal lies in the remarkable tunability achieved by varying the constituent elements and their concentrations, along with enhanced physical and chemical stability arising from the high configurational entropy of mixing. In complex metal oxides, constructing a fully periodic bulk model for every possible elemental configuration is often computationally impractical. To address this, a more efficient strategy involves examining local mixing effects near the active site. As shown in Fig. 23, this method selects a central metal atom and allows for the substitution of its neighbouring atoms—either metal cations or ligands—to simulate various local environments. This localized approach can be extended to include further coordination shells while remaining computationally manageable and site-specific. It enables the development of both bulk and surface impurity models, with surface models particularly useful for investigating mixing enthalpy and catalytic properties such as the oxygen evolution reaction.134 | ||
| Fig. 23 HEO impurity model for M3O4 spinel. (a) OER active site with adsorbed O* (red) surrounded by five metal neighbours (Cr, Co, Mn, Ni, and Fe) within 3.5 Å. Inset: top view showing active sites and nearby atoms. (b) Relative mixing enthalpy per metal for various surface active sites, referenced to bulk values. Reproduced with permission from ref. 134. Copyright 2023, Nature Communications. | ||
From a thermodynamic perspective, configurational entropy (Sconfig) reaches its maximum when all components are present in equal proportions, as in equimolar mixtures. High entropy plays a stabilizing role only when the enthalpy of mixing is significantly positive (endothermic), which is the case in entropy-stabilized materials. However, in many high-entropy materials where the mixing enthalpy is negative (exothermic), entropy does not dominate the Gibbs free energy landscape. As a result, these materials are not truly stabilized by entropy alone but rather by a combination of enthalpic and entropic effects.135
In heterogeneous catalysis, the efficiency of a catalyst largely depends on the nature and availability of its active sites, which govern key processes such as adsorption of reactants, bond transformation, charge transfer, stabilization of intermediates, and eventual product release. High-entropy spinels (HESs), composed of multiple metal cations with varying oxidation states, present a unique advantage by offering a richer and more diverse array of active sites than traditional spinel catalysts, as shown in Fig. 24. This enhanced activity is not merely the result of individual metal contributions, but rather stems from the collective and synergistic behavior of the elements, enabled by their uniform distribution and strong inter-element interactions. This collaborative effect allows HESs to effectively accommodate a wide range of reaction intermediates and pathways, thereby significantly improving catalytic performance. Interestingly, research has shown that catalytic efficiency in these systems cannot be directly predicted by the proportion of each element, highlighting the complex and cooperative nature of the active sites in HESs.136
 | ||
| Fig. 24 Relationships between the structure, properties and catalytic performance of HESs. Reproduced with permission from ref. 136. Copyright 2024, Elsevier Ltd. | ||
Wang et al. have studied entropy changes in the high entropy spinel oxide Zn(CrMnFeCoNi)2O4 during OER, revealing that the electrochemical cycling cause Zn leaching and surface reconstruction forming high entropy spinel oxide which increase surface entropy.137 Katzbaer et al. have explored the use of a high-entropy aluminate spinel oxide, (Fe0.2Co0.2Ni0.2Cu0.2Zn0.2)Al2O4 (A5Al2O4), as a catalyst for the OER in alkaline solutions. The A5Al2O4 catalyst exhibited superior performance, achieving a low η of 400 mV, surpassing the performance of single-metal catalysts. Its enhanced catalytic activity is attributed to the unique composition of the high-entropy spinel structure, which results in a reduced band gap, boosting its efficiency in facilitating the OER.138
He et al. synthesized high-entropy spinel oxides, specifically (Fe0.2Co0.2Ni0.2Cr0.2Mn0.2)3O4 (L-HEOs), using a simple room-temperature combustion method, with metal nitrates as oxidants and citric acid as a complexing agent. The SEM image of the L-HEOs shows a porous surface, a result of gas released during the synthesis process. TEM analysis reveals that large nanoparticles form through the aggregation of smaller particles, approximately 10 nm in size. HRTEM images further show numerous randomly distributed lattice stripes, indicating a high number of grain boundaries within the L-HEOs. The observed lattice fringes in the HRTEM images correspond to interplanar distances of 4.87 Å, 2.94 Å, 2.56 Å, and 2.43 Å, corresponding to the (111), (220), (311), and (222) planes of the spinel structure, respectively. Additionally, energy dispersive X-ray (EDX) spectroscopy confirms that the molar ratio of Fe, Co, Ni, Cr, and Mn is approximately 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, verifying the composition of the entropy-stabilized oxides, as shown in Fig. 25. With a Tafel slope of 50.27 mV dec−1 and an overpotential of 275 mV at 10 mA cm−2, the catalyst demonstrated increased OER activity. Furthermore, it demonstrated exceptional long-term stability, maintaining its performance for 60 h at 10 mA cm−2 and 40 h at 250 mA cm−2, attributed to its high entropy and strong structural stability, as shown in Fig. 26.139
1, verifying the composition of the entropy-stabilized oxides, as shown in Fig. 25. With a Tafel slope of 50.27 mV dec−1 and an overpotential of 275 mV at 10 mA cm−2, the catalyst demonstrated increased OER activity. Furthermore, it demonstrated exceptional long-term stability, maintaining its performance for 60 h at 10 mA cm−2 and 40 h at 250 mA cm−2, attributed to its high entropy and strong structural stability, as shown in Fig. 26.139
 | ||
| Fig. 25 (a) Synthesis diagram of L-HEOs, (b) SEM image of L-HEOs, (c) TEM image of L-HEOs, (d) HRTEM image of L-HEOs, (e) HRTEM image of amplification of the blue box in (d) and (f) EDX mapping of L-HEOs. Reproduced with permission from ref. 139. Copyright 2023, Elsevier Ltd. | ||
 | ||
| Fig. 26 (a) Representative LSV curves of L-HEOs, H-HEOs and commercial RuO2 in 1 M KOH. Insert in (a) shows the corresponding overpotentials at 10 and 50 mA cm−2; (b) Tafel slope derived from (a); (c) Tafel slope and overpotential at 10 mA cm−2 of L-HEOs with previously reported OER catalysts in 1 M KOH. Reproduced with permission from ref. 139. Copyright 2023, Elsevier Ltd. | ||
Talluri et al. have synthesized a high-entropy spinel oxide electrocatalyst, (CoCrFeMnNi)3O4, through a straightforward soft chemical method, resulting in a spherical morphology. With a low η of 220 mV, a Tafel slope of 100 mV dec−1, and an onset potential of 1.45 V vs. RHE, the catalyst showed outstanding OER activity.140 Zhang et al. have fabricated self-supported high-entropy spinel oxides on carbon cloth (CC), creating the composite (Fe0.2Co0.2Ni0.2Mn0.2Cr0.2)3O4@CC (referred to as FeCoNiMnCr@CC). This catalyst exhibited a notable η of 287 mV for the OER, coupled with exceptional long-term stability. The enhanced durability of FeCoNiMnCr@CC is attributed to the stable crystal structure of the high-entropy material and its strong adhesion to the carbon cloth support.141 For the first time, Amarnath et al. have created high-entropy metal oxide nanoparticles (Ni, Fe, Mn, Cu, Zn)3O4 using a thermal plasma technique. With a Tafel slope of 54 mV dec−1 and an η of 308 mV at a current density of 50 mA cm−2, the OER performance demonstrated that HEO@350 °C exhibited exceptional electrochemical activity. Additionally, it demonstrated good catalytic stability during a 30 hour chronoamperometry analysis.142 Triolo et al. have prepared high-entropy spinel oxide nanofibers, such as (Cr,Mn,Fe,Co,Ni), (Cr,Mn,Fe,Co,Zn), and (Cr,Mn,Fe,Ni,Zn), featuring a granular structure and oxygen-deficient surface through electrospinning and calcination at a temperature of 500 or 600 °C. These materials were tested as electrocatalysts for the OER in alkaline solutions. Among them, the (CrMnFeCoNi)3O4 nanofibers calcined at 500 °C showed the best performance, with the lowest η of 360 mV and a Tafel slope of 41 mV dec−1.143 Iwase et al. have developed high-entropy spinel oxides (HE-SOs) through a supercritical hydrothermal method, utilizing an acetate solution of the first five transition metals (Mn, Fe, Co, Ni, and Zn). These HE-SOs achieved an η of 330 mV in 1 M KOH, demonstrating exceptional catalytic activity for the OER. Their effectiveness as OER catalysts was demonstrated by the Tafel slope, which was measured to be 36.7 mV dec−1.144 Table S1† and Fig. 27 provide a comparison of the overpotentials of noble and non-noble metal spinel oxides in the oxygen evolution reaction, showcasing the varying electrocatalytic performances between the two categories.
 | ||
| Fig. 27 Graphical representation of overpotentials of reported spinel oxides with their respective reference number listed in Table S1.† | ||
5. Conclusions, challenges and future perspectives
Spinel oxides have gained great interest as OER catalysts due to their unusual crystal structure, extensive redox chemistry, and high stability. However, despite their promising qualities, various hurdles prevent their practical deployment in water electrolysis and renewable energy technologies. The large overpotential, which causes significant energy losses during the OER, is one of the main problems. The total energy efficiency of water splitting is decreased by the high voltages needed to drive the reaction effectively in many spinel oxides. Another important drawback is low intrinsic electrical conductivity, which slows down the catalytic process and prevents quick charge transfer due to inadequate electron transport. This problem is especially noticeable in metal-based oxides, where electron transport is constrained by the insulating properties of certain metal–oxygen links.Phase change and structural deterioration with extended electrochemical operation present another significant obstacle. Certain spinel oxides undergo structural rebuilding under extremely oxidative circumstances, which can result in the dissolution of important metal species into the electrolyte or the loss of active sites. As a result, catalytic performance gradually deteriorates, decreasing their long-term durability. Another process that happens in spinel oxides during the OER is surface reconstruction, in which the surface structure dynamically alters as a result of the reaction. This transition is a double-edged sword because, although it can occasionally reveal novel active sites and increase catalytic activity, it is frequently challenging to manage. In addition, scalability and cost-effectiveness continue to be major challenges because many high-performance spinel oxides contain rare or costly transition metals like ruthenium, nickel, or cobalt. Utilizing such components raises production costs and restricts widespread commercial use. Furthermore, the catalyst–substrate interaction is a critical factor in stability because poor adhesion between the catalyst and the conducting substrate can cause material detachment, which further impairs lifetime and performance.
A number of prospective tactics have been put forth to solve these issues. Doping and elemental substitution are two efficient methods that alter the electronic structure and improve conductivity of spinel lattices by introducing heteroatoms like iron, manganese, or non-metal elements. Additionally, by optimizing the active metal centers' oxidation states, this tactic can increase their catalytic efficiency. The creation of porous or nanostructured spinel oxides boosts the electrochemically active surface area and speeds up mass transfer, making nanostructuring and morphology engineering another exciting avenue. For example, mesoporous or hollow shapes can increase electrolyte accessibility, which can improve reaction kinetics.
Furthermore, hybridization with conductive materials like graphene, MXenes, or carbon nanotubes can greatly improve the quality of charge transfer. Improved electrochemical performance can result from combining spinel oxides with highly conductive materials to increase the catalyst's overall electrical conductivity. Optimizing the interface between spinel oxides and the supporting electrode is another important way to improve mechanical stability and stop catalyst detachment.
To ensure that spinel oxides can be produced affordably without compromising their functioning, scalable and eco-friendly synthesis methods must also be developed. Hydrothermal, solvothermal, and electrodeposition processes are examples of environmentally benign methods for producing highly effective catalysts. Bifunctional spinel oxides can significantly boost energy efficiency and make them more attractive for large-scale hydrogen production. Industrial applications on a large scale remain scarce. To overcome these challenges, the following aspects must be taken into account: the controllable and practical synthesis of affordable spinel-type composite materials for scalable production. Spinel oxides are among the most efficient and multifunctional catalysts for various energy-related reactions. To date, synthesis methods such as wet chemistry, sol–gel, laser pulsed, plasma deposition, and pyrolysis have been employed to create spinel-type composites.145,146 The introduction of additional external field energy is anticipated to allow for non-contact control during synthesis, opening up new possibilities for the future of spinel-type composites and solid-state chemistry. This external energy could take the form of a magnetic field, electric field, light energy, or the ultrasonic-assisted reduction method that we have proposed. Utilizing methods such as X-ray photoelectron spectroscopy (XPS) and in situ X-ray absorption spectroscopy (XAS), advanced operando studies offer important real-time insights into the structural evolution of spinel oxides during the OER. Designing more resilient catalysts and comprehending surface changes are made easier through these findings. When paired with advanced characterization techniques and precise synthesis methods, the internal mechanisms of composite materials require further investigation. Current research suggests that the properties of composites may influence the overall electronic state. Additionally, there is growing interest in dual-functional catalysts capable of efficiently driving both the hydrogen evolution reaction in complete water splitting and the oxygen evolution reaction.146
Spinel oxides have diverse applications in CO2, oxygen, and nitrogen reduction processes. In CO2 reduction, they facilitate multi-electron transfer, enhancing CO2 conversion to valuable products like carbon monoxide and hydrocarbons. Their high surface area and tunable electronic properties improve selectivity, efficiency, and stability, making them suitable for long-term applications. In the oxygen reduction reaction (ORR), spinel oxides exhibit excellent catalytic performance, aiding the efficient reduction of oxygen to water, which is crucial for energy conversion systems like fuel cells and metal–air batteries. For nitrogen reduction, spinel oxides activate nitrogen molecules, enabling ammonia production under mild conditions. Their high conductivity and efficient charge transfer make them a sustainable alternative to traditional ammonia synthesis, offering a more energy-efficient approach. Thus, spinel oxides play a critical role in multiple electrochemical processes, contributing to advancements in energy storage, environmental sustainability, and chemical production.147
Computational modeling and machine learning (ML) play a pivotal role in predicting and optimizing the properties of spinel oxides, especially in the development of next-generation catalysts for a wide range of applications, such as electrocatalysis and energy storage. As the demand for more efficient, stable, and sustainable materials grows, the role of computational tools and machine learning becomes even more critical in identifying and designing materials with superior performance. Computational methods like Density Functional Theory (DFT) provide detailed insights into the atomic and electronic structure of spinel oxides, enabling the prediction of their behavior in various catalytic processes and energy storage systems. These simulations help understand how structural modifications, such as doping or changes in lattice arrangement, influence catalytic activity, stability, and overall performance. On the other hand, ML algorithms analyze large datasets from both experimental and simulation studies, rapidly screening vast material spaces to predict the properties of new spinel oxide compositions. By uncovering correlations between composition, structure, and properties, ML accelerates the design of novel materials. In combination, computational modeling and machine learning allow researchers to optimize spinel oxides for applications in energy storage, catalysis, and other fields, paving the way for the development of next-generation catalysts that are more efficient, cost-effective, and scalable.148
In conclusion, despite the enormous promise of spinel oxides as OER catalysts, a number of obstacles must be overcome, including high overpotential, poor conductivity, structural instability, and scaling problems. To improve their stability and efficiency, future research should concentrate on operando investigations, conductive hybrid materials, morphological engineering, and doping techniques. Spinel oxides can become next-generation water electrolysis catalysts and aid in the development of clean and renewable energy technologies by overcoming these constraints through creative material design and sustainable synthesis techniques.
Data availability
The data supporting this article have already been included in the main text and/or as part of the ESI.†Conflicts of interest
The authors declare no conflict of interest.Acknowledgements
We acknowledge support from the National Science and Technology Council of Taiwan through Grant No. 113-2221-E-005-007-MY3. We also acknowledge the financial support from the “Innovation and Development Center of Sustainable Agriculture” from The Featured Areas Research Center Program within the framework of the Higher Education Sprout Project by the Ministry of Education (MOE) in Taiwan.References
- H. Ishaq, I. Dincer and C. Crawford, Int. J. Hydrogen Energy, 2022, 47(62), 26238–26264 CrossRef CAS.
- S. E. Hosseini and M. A. Wahid, Renewable Sustainable Energy Rev., 2016, 57, 850–866 CrossRef CAS.
- F. Dawood, M. Anda and G. M. Shafiullah, Int. J. Hydrogen Energy, 2020, 45, 3847–3869 CrossRef CAS.
- X. Shang, J. H. Tang, B. Dong and Y. Sun, Sustainable Energy Fuels, 2020, 4(7), 3211–3228 RSC.
- K. B. M. Ismail, M. Arun Kumar, S. Mahalingam, J. Kim and R. Atchudan, Materials, 2023, 16, 4471 CrossRef CAS PubMed.
- N. N. Loganathan, V. Perumal, B. R. Pandian, R. Atchudan, T. N. J. I. Edison and M. Ovinis, J. Energy Storage, 2022, 50, 104149 CrossRef.
- C. Pitchai, S. M. Gopalakrishnan and C. M. Chen, Energy Fuels, 2024, 38(3), 2235–2247 CrossRef CAS.
- J. Yu, T. A. Le, N. Q. Tran and H. Lee, Chem.–Eur. J., 2020, 26, 6423–6436 CrossRef CAS.
- Z. Li, X. Zhang, C. Ou, Y. Zhang, W. Wang, S. Dong and X. Dong, Coord. Chem. Rev., 2023, 491, 215381 CrossRef.
- J. Wang, X. Yue, Y. Yang, S. Sirisomboonchai, P. Wang, X. Ma, A. Abudula and G. Guan, J. Alloys Compd., 2020, 819, 153346 CrossRef CAS.
- J. Mohammed-Ibrahim and H. Moussab, J. Energy Chem., 2021, 57, 299–342 Search PubMed.
- K. Zhang and R. Zou, Small, 2021, 17(37), 2100129 CrossRef CAS.
- L. Tian, Z. Li, X. Xu and C. Zhang, J. Mater. Chem. A, 2021, 9(23), 13459–13470 RSC.
- G. Gao, Z. Sun, X. Chen, G. Zhu, B. Sun, Y. Yamauchi and S. Liu, Appl. Catal., B, 2024, 339, 123584 CrossRef.
- Y. Zi, C. Zhang, J. Zhao, Y. Cheng, J. Yuan and J. Hu, Small, 2024, 20(50), 2406657 CrossRef CAS PubMed.
- Y. Xue, S. Sun, Q. Wang, Z. Dong and Z. Liu, J. Mater. Chem. A, 2018, 6(22), 10596–10626 Search PubMed.
- M. Vedanarayanan, C. M. Chen and M. G. Sethuraman, ACS Appl. Energy Mater., 2024, 7(17), 7260–7271 CrossRef CAS.
- Z. G. Yang, H. M. Xu, T. Y. Shuai, Q. N. Zhan, Z. J. Zhang, K. Huang, C. Dai and G. R. Li, Nanoscale, 2023, 15(22), 11777–11800 RSC.
- M. Vedanarayanan, C. Pitchai and S. M. Gopalakrishnan, New J. Chem., 2024, 48(14), 6420–6429 RSC.
- H. Wang, S. Zhu, J. Deng, W. Zhang, Y. Feng and J. Ma, Chin. Chem. Lett., 2021, 32(1), 291–298 CrossRef CAS.
- A. Lebechi, A. K. Ipadeola, K. Eid, A. M. Abdullah and K. I. Ozoemena, Nanoscale, 2022, 14, 10717–10737 RSC.
- H. Xu, J. Yuan, G. He and H. Chen, Coord. Chem. Rev., 2023, 478, 214869 CrossRef.
- X. Yue, X. Qin, Y. Chen, Y. Peng, C. Liang, M. Feng, X. Qiu, M. Shao and S. Huang, Adv. Sci., 2021, 8(17), 2101653 CrossRef CAS.
- S. Deka, Dalton Trans., 2022, 52, 839–856 RSC.
- F. Bosi, C. Biagioni and M. Pasero, Eur. J. Mineral., 2019, 31, 183–192 CrossRef CAS.
- J. X. Flores-Lasluisa, F. Huerta, D. Cazorla-Amorós and E. Morallón, Environ. Res., 2022, 214, 113731 CrossRef CAS.
- S. Chandrasekaran, C. Bowen, P. Zhang, Z. Li, Q. Yuan, X. Ren and L. Deng, J. Mater. Chem. A, 2018, 6, 11078–11104 RSC.
- R. Wei, X. Bu, W. Gao, R. A. B. Villaos, G. MacAm, Z. Q. Huang, C. Lan, F. C. Chuang, Y. Qu and J. C. Ho, ACS Appl. Mater. Interfaces, 2019, 11(36), 33012–33021 CrossRef CAS.
- H. Xu, J. Yuan, G. He and H. Chen, Coord. Chem. Rev., 2023, 475, 214869 CrossRef CAS.
- X. M. Liu, X. Cui, K. Dastafkan, H. F. Wang, C. Tang, C. Zhao, A. Chen, C. He, M. Han and Q. Zhang, J. Energy Chem., 2020, 46, 290–302 Search PubMed.
- Y. Zhou, S. Sun, C. Wei, Y. Sun, P. Xi, Z. Feng and Z. J. Xu, Adv. Mater., 2019, 31(41), 1902509 CrossRef CAS PubMed.
- A. Grimaud, O. Diaz-Morales, B. Han, W. T. Hong, Y. L. Lee, L. Giordano and Y. Shao-Horn, Nat. Chem., 2017, 9(5), 457–465 CrossRef CAS.
- M. I. Jamesh and X. Sun, J. Power Sources, 2018, 400, 31–68 CrossRef CAS.
- C. Pitchai, M. Vedanarayanan and S. Mathur Gopalakrishnan, New J. Chem., 2023, 47(30), 14355–14363 RSC.
- Q. Liang, G. Brocks and A. Bieberle-Hütter, J. Phys Energy, 2021, 3(2), 026001 CrossRef CAS.
- P. Chandrasekaran, T. N. Jebakumar Immanuel Edison and M. G. Sethuraman, Int. J. Hydrogen Energy, 2020, 45(53), 28800–28811 CrossRef CAS.
- X. Liu, Z. He, M. Ajmal, C. Shi, R. Gao, L. Pan, Z. F. Huang, X. Zhang and J. J. Zou, Trans. Tianjin Univ., 2023, 29(4), 247–253 CrossRef CAS.
- A. Zagalskaya and V. Alexandrov, ACS Catal., 2020, 10(6), 3650–3657 CrossRef CAS.
- J. Song, C. Wei, Z. F. Huang, C. Liu, L. Zeng, X. Wang and Z. J. Xu, Chem. Soc. Rev., 2020, 49, 2196–2214 RSC.
- M. Vedanarayanan and S. Mathur Gopalakrishnan, Energy Fuels, 2023, 37(16), 12204–12214 CrossRef CAS.
- J. Song, C. Wei, Z. F. Huang, C. Liu, L. Zeng, X. Wang and Z. J. Xu, Chem. Soc. Rev., 2020, 49(7), 2196–2214 RSC.
- N. Zhang and Y. Xiong, J. Phys. Chem. C, 2023, 127, 2147–2159 CrossRef CAS.
- Y. Pan, X. Xu, Y. Zhong, L. Ge, Y. Chen, J. P. M. Veder, D. Guan, R. O'Hayre, M. Li, G. Wang, H. Wang, W. Zhou and Z. Shao, Nat. Commun., 2020, 11(1), 2002 CrossRef CAS.
- X. Xu, Y. Pan, Y. Zhong, C. Shi, D. Guan, L. Ge, Z. Hu, Y. Y. Chin, H. J. Lin, C. Te Chen, H. Wang, S. P. Jiang and Z. Shao, Adv. Sci., 2022, 9(14), 2200530 CrossRef CAS.
- O. N. Avcı, L. Sementa and A. Fortunelli, ACS Catal., 2022, 12(15), 9058–9073 CrossRef.
- M. Yu, E. Budiyanto and H. Tüysüz, Angew. Chem., Int. Ed., 2022, 61(1), e202103824 CrossRef CAS.
- S. Niu, S. Li, Y. Du, X. Han and P. Xu, ACS Energy Lett., 2020, 5(4), 1083–1087 CrossRef CAS.
- P. Chandrasekaran, T. N. J. I. Edison and M. G. Sethuraman, Fuel, 2022, 320, 123947 CrossRef CAS.
- C. Pitchai, M. Vedanarayanan, C. M. Chen and S. M. Gopalakrishnan, Int. J. Hydrogen Energy, 2024, 72, 755–763 CrossRef CAS.
- P. Chandrasekaran, T. N. J. I. Edison and M. G. Sethuraman, Fuel, 2022, 320, 123947 CrossRef CAS.
- X. Lu and C. Zhao, Nat. Commun., 2015, 6(1), 1–7 Search PubMed.
- S. Anantharaj, P. E. Karthik and S. Noda, Angew. Chem., Int. Ed., 2021, 60(41), 23051–23067 CrossRef CAS.
- M. Vedanarayanan, C. Pitchai, C. M. Chen and S. M. Gopalakrishnan, Electrochim. Acta, 2024, 497, 144529 CrossRef CAS.
- M. Görlin, P. Chernev, J. F. De Araújo, T. Reier, S. Dresp, B. Paul, R. Krähnert, H. Dau and P. Strasser, J. Am. Chem. Soc., 2016, 138(17), 5603–5614 CrossRef.
- L. Yu, S. Sun, H. Li and Z. J. Xu, Fundam. Res., 2021, 1(4), 448–452 CrossRef CAS.
- C. Spöri, P. Briois, H. N. Nong, T. Reier, A. Billard, S. Kühl, D. Teschner and P. Strasser, ACS Catal., 2019, 9(8), 6653–6663 CrossRef.
- S. Chandrasekaran, C. Zhang, Y. Shu, H. Wang, S. Chen, T. N. J. I. Edison and Z. Han, Coord. Chem. Rev., 2021, 449, 214209 CrossRef CAS.
- I. A. Khan, P. Morgen, R. Sharma and S. M. Andersen, J. Phys. Chem. C, 2024, 128(7), 2828–2833 CrossRef CAS.
- M. Yu, G. Moon, E. Bill and H. Tüysüz, ACS Appl. Energy Mater., 2019, 2(2), 1199–1209 CrossRef CAS.
- M. David, C. Ocampo-Martínez and R. Sμnchez-PeḈa, J. Energy Storage, 2019, 23, 392–403 CrossRef.
- Y. Wei, C.-H. Shin, E. B. Tetteh, B.-J. Lee and J.-S. Yu, ACS Appl. Energy Mater., 2020, 3, 822–830 CrossRef CAS.
- L. J. Enman, A. E. Vise, M. Burke Stevens and S. W. Boettcher, ChemPhysChem, 2019, 20, 3089–3095 CrossRef CAS PubMed.
- M. Schalenbach, O. Kasian and K. J. J. Mayrhofer, Int. J. Hydrogen Energy, 2018, 43, 11932–11938 CrossRef CAS.
- A. N. Colli, H. H. Girault and A. Battistel, Materials, 2019, 12, 1336 CrossRef CAS.
- E. J. W. Verwey and E. L. Heilmann, J. Chem. Phys., 1947, 15(4), 174–180 CrossRef CAS.
- K. E. Sickafus, J. M. Wills and N. W. Grimes, J. Am. Ceram. Soc., 1999, 82(12), 3279–3292 CrossRef CAS.
- Q. Zhao, Z. Yan, C. Chen and J. Chen, Chem. Rev., 2017, 117(15), 10121–10211 CrossRef CAS PubMed.
- S. V. Devaguptapu, S. Hwang, S. Karakalos, S. Zhao, S. Gupta, D. Su, H. Xu and G. Wu, ACS Appl. Mater. Interfaces, 2017, 9(51), 44567–44578 CrossRef CAS PubMed.
- T. W. Kim, M. A. Woo, M. Regis and K. S. Choi, J. Phys. Chem. Lett., 2014, 5(13), 2370–2374 CrossRef CAS.
- J. Luo, X. Wang, Y. Gu, S. Wang, Y. Li, T. Wang, Y. Liu, Y. Zhou and J. Zhang, Chem. Eng. J., 2023, 472, 144839 CrossRef CAS.
- J. Zheng, X. Peng, Z. Xu, J. Gong and Z. Wang, ACS Catal., 2022, 12(16), 10245–10254 CrossRef CAS.
- W. Liu, M. Kamiko, I. Yamada and S. Yagi, Mater. Adv., 2022, 3(20), 7513–7519 RSC.
- J. Huang, H. Sheng, R. D. Ross, J. Han, X. Wang, B. Song and S. Jin, Nat. Commun., 2021, 12(1), 3036 CrossRef CAS.
- G. Zhang, M. Chen, C. Li, B. Wu, J. Chen, W. Xiang, X. Wen, D. Zhang, G. Cao and W. Li, Chem. Eng. J., 2022, 443, 136434 CrossRef CAS.
- T. Wu, S. Sun, J. Song, S. Xi, Y. Du, C. Bo, W. Aji Sasangka, H. Liao, C. Lip Gan and G. G. Scherer, Nat. Catal., 2019, 2(9), 763–772 CrossRef CAS.
- J. Zhang, Y. Ye, B. Wei, F. Hu, L. Sui, H. Xiao, L. Gui, J. Sun, B. He and L. Zhao, Appl. Catal., B, 2023, 330, 122661 CrossRef CAS.
- Z. Fan, J. Jiang, L. Ai, Z. Shao and S. Liu, ACS Appl. Mater. Interfaces, 2019, 11(51), 47894–47903 CrossRef CAS.
- J. Fonseca and S. Choi, Catal. Sci. Technol., 2020, 10(24), 8265–8282 RSC.
- W. Q. Zaman, W. Sun, Z. H. Zhou, Y. Wu, L. Cao and J. Yang, ACS Appl. Energy Mater., 2018, 1(11), 6374–6380 CrossRef CAS.
- Y. Liu, Y. Chen, X. Mu, Z. Wu, X. Jin, J. Li, Y. Xu, L. Yang, X. Xi, H. Jang, Z. Lei, Q. Liu, S. Jiao, P. Yan, X. Li and R. Cao, ACS Catal., 2023, 13(6), 3757–3767 CrossRef CAS.
- T. Wang, Z. Li, H. Jang, M. G. Kim, Q. Qin and X. Liu, ACS Sustain. Chem. Eng., 2023, 11(13), 5155–5163 CrossRef CAS.
- S. Niu, X.-P. Kong, S. Li, Y. Zhang, J. Wu, W. Zhao and P. Xu, Appl. Catal., B, 2021, 297, 120442 CrossRef CAS.
- J. Shan, C. Ye, S. Chen, T. Sun, Y. Jiao, L. Liu, C. Zhu, L. Song, Y. Han, M. Jaroniec, Y. Zhu, Y. Zheng and S. Z. Qiao, J. Am. Chem. Soc., 2021, 143(13), 5201–5211 CrossRef CAS PubMed.
- J. Yin, J. Jin, M. Lu, H. Zhang, Y. Peng and C. H. Yan, J. Am. Chem. Soc., 2020, 142(43), 18378–18386 CrossRef CAS PubMed.
- Y. Zhu, J. Wang, T. Koketsu, M. Kroschel, J. M. Chen, S. Y. Hsu, G. Henkelman, Z. Hu, P. Strasser and J. Ma, Nat. Commun., 2022, 13(1), 7754 CrossRef CAS PubMed.
- G. B. Wang, C. S. Hsu and H. M. Chen, Dalton Trans., 2020, 49(48), 17505–17510 RSC.
- J. O. Olowoyo and R. J. Kriek, Small, 2022, 18(41), 2203125 CrossRef CAS.
- H. Shi and G. Zhao, J. Phys. Chem. C, 2014, 118(45), 25939–25946 CrossRef CAS.
- B. Cui, H. Lin, J. B. Li, X. Li, J. Yang and J. Tao, Adv. Funct. Mater., 2008, 18(9), 1440–1447 CrossRef CAS.
- C. Jin, F. Lu, X. Cao, Z. Yang and R. Yang, J. Mater. Chem. A, 2013, 1(39), 12170–12177 RSC.
- Y. Z. Su, Q. Z. Xu, G. F. Chen, H. Cheng, N. Li and Z. Q. Liu, Electrochim. Acta, 2015, 174, 1216–1224 CrossRef CAS.
- Z. Chen, B. Zhao, Y. C. He, H. R. Wen, X. Z. Fu, R. Sun and C. P. Wong, Mater. Chem. Front., 2018, 2(6), 1155–1164 RSC.
- X. Liu, Z. Chang, L. Luo, T. Xu, X. Lei, J. Liu and X. Sun, Chem. Mater., 2014, 26(5), 1889–1895 CrossRef CAS.
- R. M. Ramsundar, V. K. Pillai and P. A. Joy, Phys. Chem. Chem. Phys., 2018, 20(46), 29452–29461 RSC.
- P. W. Menezes, A. Indra, A. Bergmann, P. Chernev, C. Walter, H. Dau, P. Strasser and M. Driess, J. Mater. Chem. A, 2016, 4(25), 10014–10022 RSC.
- S. Han, S. Liu, R. Wang, X. Liu, L. Bai and Z. He, ACS Appl. Mater. Interfaces, 2017, 9(20), 17186–17194 CrossRef CAS.
- J. Zhang, D. Zhang, Y. Yang, J. Ma, S. Cui, Y. Li and B. Yuan, RSC Adv., 2016, 6(95), 92699–92704 RSC.
- X. Du, X. Zhang, Z. Xu, Z. Yang and Y. Gong, Int. J. Hydrogen Energy, 2018, 43(10), 5012–5018 CrossRef CAS.
- A. T. Aqueel Ahmed, B. Hou, H. S. Chavan, Y. Jo, S. Cho, J. Kim, S. M. Pawar, S. N. Cha, A. I. Inamdar, H. Kim and H. Im, Small, 2018, 14(28), 1800742 CrossRef.
- A. T. Aqueel Ahmed, S. M. Pawar, A. I. Inamdar, H. Kim and H. Im, Adv. Mater. Interfaces, 2020, 7(2), 1901515 CrossRef CAS.
- S. K. Bikkarolla and P. Papakonstantinou, J. Power Sources, 2015, 281, 243–251 CrossRef CAS.
- C. Alegre, C. Busacca, A. Di Blasi, O. Di Blasi, A. S. Aricò, V. Antonucci and V. Baglio, Mater. Today Energy, 2020, 18, 100508 CrossRef CAS.
- W. Yan, Z. Yang, W. Bian and R. Yang, Carbon, 2015, 92, 74–83 CrossRef CAS.
- W. Wang, L. Kuai, W. Cao, M. Huttula, S. Ollikkala, T. Ahopelto, A. P. Honkanen, S. Huotari, M. Yu and B. Geng, Angew. Chem. Int. Ed., 2017, 56(47), 14977–14981 CrossRef CAS.
- X. Cao, C. Jin, F. Lu, Z. Yang, M. Shen and R. Yang, J. Electrochem. Soc., 2014, 161(5), H296–H300 CrossRef CAS.
- K. Zeng, W. Li, Y. Zhou, Z. Sun, C. Lu, J. Yan, J. H. Choi and R. Yang, Chem. Eng. J., 2021, 421, 127831 CrossRef CAS.
- M. S. Al-Hoshan, J. P. Singh, A. M. Al-Mayouf, A. A. Al-Suhybani and M. N. Shaddad, Int. J. Electrochem. Sci., 2012, 7(6), 4959–4973 CrossRef CAS.
- Z. Liu, B. Tang, X. Gu, H. Liu and L. Feng, Chem. Eng. J., 2020, 395, 125170 CrossRef CAS.
- P. Balasubramanian, S. Bin He, A. Jansirani, H. H. Deng, H. P. Peng, X. H. Xia and W. Chen, Chem. Eng. J., 2021, 405, 126732 CrossRef CAS.
- L. S. Ferreira, T. R. Silva, J. R. D. Santos, V. D. Silva, R. A. Raimundo, M. A. Morales and D. A. Macedo, Mater. Chem. Phys., 2019, 237, 121847 CrossRef CAS.
- C. Mahala, M. D. Sharma and M. Basu, Electrochim. Acta, 2018, 273, 462–473 CrossRef CAS.
- A. Kargar, S. Yavuz, T. K. Kim, C. H. Liu, C. Kuru, C. S. Rustomji, S. Jin and P. R. Bandaru, ACS Appl. Mater. Interfaces, 2015, 7(32), 17851–17856 CrossRef CAS.
- K. L. Yan, X. Shang, Z. Z. Liu, B. Dong, S. S. Lu, J. Q. Chi, W. K. Gao, Y. M. Chai and C. G. Liu, Int. J. Hydrogen Energy, 2017, 42(38), 24150–24158 CrossRef CAS.
- S. D. Jituri, R. P. Nikam, V. J. Mane, S. B. Shaikh, C. D. Lokhande and S. H. Mujawar, J. Mater. Sci.: Mater. Electron., 2023, 34(27), 1842 CrossRef CAS.
- Y. Hu, X. Zhao, F. Li, Q. Dong, B. Wen, D. Sun, W. Liang and X. Lyu, ACS Appl. Energy Mater., 2023, 6(19), 9985–9993 CrossRef CAS.
- D. E. Lee, M. Danish, A. Husain and W. K. Jo, J. Alloys Compd., 2024, 1000, 175048 CrossRef CAS.
- M. Li, M. Lu, J. Yang, J. Xiao, L. Han, Y. Zhang and X. Bo, J. Alloys Compd., 2019, 809, 151766 CrossRef CAS.
- K. Baruah, A. Kant, P. Gaijon, S. Ghosh and M. R. Singh, Appl. Surf. Sci. Adv., 2021, 6, 100130 CrossRef.
- Y. Liu, Z. Niu, Y. Lu, L. Zhang and K. Yan, J. Alloys Compd., 2018, 735, 654–659 CrossRef CAS.
- J. Kim, J. Lee, C. Liu, S. Pandey, S. W. Joo, N. Son and M. Kang, Appl. Surf. Sci., 2021, 546, 149124 CrossRef.
- X. Liu, Y. Q. Huo, L. K. Yan, N. Fan, K. Z. Cai and Z. M. Su, Chem.–Eur. J., 2020, 26(63), 14397–14404 Search PubMed.
- X. Wu, Y. Niu, B. Feng, Y. Yu, X. Huang, C. Zhong, W. Hu and C. M. Li, ACS Appl. Mater. Interfaces, 2018, 10(24), 20440–20447 CrossRef CAS PubMed.
- Y. Chen, Z. Shi, S. Li, J. Feng, B. Pang, L. Yu, W. Zhang and L. Dong, J. Electroanal. Chem., 2020, 860, 113930 CrossRef CAS.
- P. Ramakrishnan, K. B. Lee and J. I. Sohn, Appl. Surf. Sci., 2021, 566, 150653 Search PubMed.
- K. L. Yan, X. Shang, Z. Li, B. Dong, X. Li, W. K. Gao, J. Q. Chi, Y. M. Chai and C. G. Liu, Appl. Surf. Sci., 2017, 416, 371–378 CrossRef CAS.
- W. Chen, H. Wang, Y. Li, Y. Liu, J. Sun, S. Lee, J. S. Lee and Y. Cui, ACS Cent. Sci., 2015, 1(5), 244–251 CrossRef CAS PubMed.
- G. Karkera, T. Sarkar, M. D. Bharadwaj and A. S. Prakash, ChemCatChem, 2017, 9(19), 3681–3690 CrossRef CAS.
- A. Gupta, W. D. Chemelewski, C. B. Mullins and J. B. Goodenough, Adv. Mater., 2015, 27(39), 6063–6067 CrossRef CAS.
- Y. Zhu, W. Zhou, Y. Chen, J. Yu, M. Liu and Z. Shao, Adv. Mater., 2015, 27(44), 7150–7155 CrossRef CAS PubMed.
- Z. Lu, H. Wang, D. Kong, K. Yan, P. C. Hsu, G. Zheng, H. Yao, Z. Liang, X. Sun and Y. Cui, Nat. Commun., 2014, 5(1), 4345 CrossRef CAS PubMed.
- S. Roy and S. Roy, Sustainable Energy Fuels, 2024, 8(17), 3854–3864 RSC.
- Q. Zhang, Y. Hu, H. Wu, X. Zhao, M. Wang, S. Wang, R. Feng, Q. Chen, F. Song, M. Chen and P. Liu, ACS Nano, 2023, 17(2), 1485–1494 CrossRef CAS.
- A. Pineiro-Garcia, X. Wu, E. J. Canto-Aguilar, A. Kuzhikandathil, M. Rafei and E. Gracia-Espino, ACS Appl. Mater. Interfaces, 2024, 16(51), 70429–70441 CrossRef CAS PubMed.
- J. Baek, M. D. Hossain, P. Mukherjee, J. Lee, K. T. Winther, J. Leem and X. Zheng, Nat. Commun., 2023, 14(1), 5936 CrossRef CAS PubMed.
- M. Fracchia, M. Coduri, P. Ghigna and U. Anselmi-Tamburini, J. Eur. Ceram. Soc., 2024, 44(2), 585–594 CrossRef CAS.
- Y. Mo, X. Guan, S. Wang and X. Duan, Chem. Sci., 2025, 16, 1652–1676 RSC.
- J. Wang, S. Sun, S. Xi, Y. Sun, S. J. H. Ong, Z. W. Seh and Z. J. Xu, J. Phys. Chem. C, 2024, 128, 4978–4987 CrossRef CAS.
- R. R. Katzbaer, F. M. dos Santos Vieira, I. Dabo, Z. Mao and R. E. Schaak, J. Am. Chem. Soc., 2023, 145(12), 6753–6761 CrossRef CAS.
- L. He, H. Kang, G. Hou, X. Qiao, X. Jia, W. Qin and X. Wu, Chem. Eng. J., 2023, 460, 141675 CrossRef CAS.
- B. Talluri, K. Yoo and J. Kim, J. Environ. Chem. Eng., 2022, 10(1), 106932 CrossRef CAS.
- R. Zhang, Z. Xu, Z. Du, Y. Wan, S. Yuan, F. Zeng, J. Xu, Z. Meng, X. Hu and H. Tian, Inorg. Chem., 2023, 62(46), 19052–19059 CrossRef CAS PubMed.
- P. Amarnath, R. Madhu, K. Praveen, S. Govindarajan, S. Kundu and Y. Subramaniam, ACS Appl. Energy Mater., 2023, 6(11), 5899–5911 CrossRef CAS.
- C. Triolo, K. Moulaee, A. Ponti, G. Pagot, V. Di Noto, N. Pinna, G. Neri and S. Santangelo, Adv. Funct. Mater., 2024, 34(6), 2306375 CrossRef CAS.
- K. Iwase and I. Honma, ACS Appl. Energy Mater., 2022, 5(8), 9292–9296 CrossRef CAS.
- E. L. Ayuk, M. O. Ugwu and S. B. Aronimo, Chem. Res. J., 2017, 2(5), 97–123 CAS.
- Z. Shao, X. Wu, X. Wu, S. Feng and K. Huang, Mater. Chem. Front., 2023, 7(21), 5288–5308 RSC.
- J. X. Flores-Lasluisa, F. Huerta, D. Cazorla-Amorós and E. Morallón, Environ. Res., 2022, 214, 113731 CrossRef CAS.
- S. Fujii, J. Hyodo, K. Shitara, A. Kuwabara, S. Kasamatsu and Y. Yamazaki, Sci. Technol. Adv. Mater., 2024, 25(1), 2416383 CrossRef PubMed.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: https://doi.org/10.1039/d5ta01363a |
| This journal is © The Royal Society of Chemistry 2025 |





