Unlocking sunlight driven photocatalysis: synthesis, diversity, and applications of COF-based S-scheme heterojunctions
Irshad
Ahamd
 *abd,
Zaheer Ud Din
Babar
*abd,
Zaheer Ud Din
Babar
 e,
Yifei
Zhang
*a,
Ayman
Al-Qattan
f,
Samia Ben
Ahmed
g and
Gao
Li
e,
Yifei
Zhang
*a,
Ayman
Al-Qattan
f,
Samia Ben
Ahmed
g and
Gao
Li
 *abc
*abc
aInstitute of Catalysis for Energy and Environment, College of Chemistry and Chemical Engineering Shenyang Normal University, Shenyang 110034, China. E-mail: sszyf2017@126.com
bSchool of Chemistry and Chemical Engineering, Inner Mongolia Normal University, Hohhot 010018, China. E-mail: ligao_2019@qq.com
cDalian Institute of Chemical Physics, CAS, Dalian 116023, China
dDepartment of Physics, University of Agriculture Faisalabad, Pakistan. E-mail: irshadmahar55@yahoo.com
eDepartment of Physics “Ettore Pancini”, University of Naples Federico II, Piazzale Tecchio, 80, 80125 Naples, Italy
fEnergy and Building Research Center, Kuwait Institute for Scientific Research, Safat, 13109, Kuwait
gDepartment of Chemistry College of Science, King Khalid University, P. O. Box 9004, Abha, Saudi Arabia
First published on 8th April 2025
Abstract
Covalent organic frameworks (COFs) are highly porous and crystalline organic polymers with remarkable thermal and chemical stability. Their tunable structures and properties have enabled their applications in diverse fields. However, COFs suffer from significant drawbacks, including poor processability, strong self-stacking tendencies, limited electrical conductivity, restricted ion transport owing to pore blockage, and quick recombination of photogenerated electron–hole pairs. To address these limitations, construction of heterojunctions between COFs and other semiconductors has emerged as an effective approach. In particular, the S-scheme heterojunction design has recently attracted increasing interest owing to its ability to suppress charge carrier recombination while preserving strong redox capability, thereby enhancing photocatalytic efficiency. Despite these advantages, there is a scarcity of comprehensive reviews focusing on COF-based S-scheme heterojunctions. This review provides a detailed overview of the structural and functional properties of these materials in photocatalysis. It further discusses various synthesis strategies and charge-transfer mechanisms involved in constructing S-scheme heterojunctions by integrating COFs with different semiconductor materials. Additionally, recent advancements in COF-based S-scheme heterojunction photocatalysts are summarized, highlighting their various applications. Finally, the persistent challenges and potential future research directions in this domain are critically examined.
Introduction
Photocatalysis is a vital strategy for harnessing solar energy and converting it into chemical energy for various applications.1 However, conventional inorganic semiconductor photocatalysts face several challenges, including wide band gaps, limited structural tunability, and a lack of abundant active sites.2 These drawbacks contribute to the low efficiency of photocatalytic processes, thereby restricting their scalability for industrial applications.3 As a result, there is a growing need for advanced materials and innovative structural designs to improve photocatalytic performance. Recent years have witnessed significant progress in the development of porous materials with tailored structures for photocatalysis.3 Among them, metal–organic frameworks (MOFs), covalent organic frameworks (COFs), hydrogen-bonded organic frameworks (HOFs), and porous organic polymers (POPs) have attracted considerable interest.4,5 MOFs, composed of metal clusters and organic ligands connected by coordination bonds, exhibit well-ordered structures, high porosity, and tunable properties.6 However, their practical application in photocatalysis is hindered by limited chemical stability.7 Similarly, HOFs, which rely on hydrogen bonding interactions to form defined structures with low densities, also suffer from poor chemical stability, limiting their functional versatility.8 POPs, in contrast, are highly stable due to their covalent bonding networks, but their irregular pores and undefined structures make it challenging to establish structure–property relationships.9Among these porous materials, COFs stand out as a distinct class of crystalline, covalently linked polymers with well-defined structures.10 Their ability to integrate the advantageous properties of MOFs, HOFs, and POPs while addressing their limitations makes them attractive candidates for photocatalytic applications.11 COFs exhibit semiconducting behavior and a tailorable electronic structure, which permits for specific control over light absorption and charge transfer processes.12 The high surface area and porous architecture of COFs provide numerous active sites for catalysis while minimizing particle aggregation, thereby improving reaction efficiency.13 Furthermore, their chemical versatility enables fine-tuning of their properties through rational design and modification, allowing them to accomplish the specific prerequisites of different photocatalytic reactions.14 The structural flexibility of COFs offers additional advantages, as their framework composition can be engineered to optimize physical and electronic properties.15 By adjusting the building blocks, functional groups, and connectivity, COFs can be tailored for enhanced light absorption, charge separation, and catalytic activity.16 These properties make COFs promising platforms for a wide range of applications, such as photocatalysis, sensors, gas separation, and drug delivery.17,18 In photocatalysis, their ability to facilitate electron transfer and provide efficient reaction sites plays a crucial role in improving reaction rates and selectivity.19 In spite of these benefits, the photocatalytic efficiency of COFs remains hindered by their relatively large band gaps and slow charge transfer kinetics.20 These factors reduce their ability to utilize a broad range of solar radiation and hinder the overall reaction efficiency.21 Additionally, recombination of photogenerated electron–hole pairs further limits their large-scale application.22 To overcome these limitations, researchers have developed various strategies, including the construction of heterojunction composites, metal doping, and the incorporation of co-catalysts.23–25 Among these, the design of heterojunction systems has proven to be particularly impressive in promoting charge separation and increasing photocatalytic efficiency.
Heterojunction photocatalysts function by forming an interface between different semiconductor materials to facilitate charge transfer and reduce recombination.26 Traditional heterojunction designs, such as type-II and Z-scheme configurations, have demonstrated success in improving photocatalytic activity.27 However, these systems have inherent drawbacks, including insufficient redox potential and inefficient charge separation.28 Recently, an innovative approach, S-scheme heterojunctions, has emerged as a potential substitute for overcoming these limitations.29 The S-scheme heterojunction is formed by coupling an oxidation photocatalyst (OP) with a reduction photocatalyst (RP). Upon integration, electrons from the RP migrate to the OP, leading to band bending at the interface. This charge migration results in the generation of an internal electric field directed from the RP to OP. Owing to coulombic interactions and the inherent electric field, non-contributory charge carriers recombine, while high-energy electrons and holes remain effectively separated. This unique charge transfer mechanism enhances photocatalytic performance by maintaining a strong redox potential while simultaneously reducing charge carrier recombination,30–32 resulting in S-scheme heterojunctions exhibiting superior photocatalytic activity compared to traditional heterojunction designs.
Integrating COFs with S-scheme heterojunctions presents an outstanding approach for further increasing photocatalytic performance.33 The combination of the COF's high surface area, tunable band structure, and stability with the efficient charge separation process of S-scheme heterojunctions leads to improved light absorption and enhanced photocatalytic efficiency.34 By leveraging the synergistic effects of COFs and S-scheme heterojunctions, researchers have explored applications in diverse photocatalytic fields, including hydrogen production, CO2 reduction, and H2O2 synthesis.35,36 COF-based S-scheme heterojunctions capitalize on the advantages of both COFs and S-scheme charge transfer pathways, leading to increased reaction rates and improved photocatalytic efficiency.37 The porous structure of COFs facilitates a beneficial environment for reactant adsorption and mass transport, while the heterojunction design ensures optimal charge carrier dynamics. This integration enables COFs to serve as efficient photocatalytic platforms for renewable energy conversion and environmental remediation.38 Despite these advancements, research on COF-based S-scheme heterojunctions is still in its infancy, and plentiful barriers remain. For instance, the synthesis of COF-based S-scheme heterojunctions is limited by factors such as poor interfacial compatibility, difficulty in achieving precise structural alignment, and challenges in controlling crystallinity and porosity.33–37 These issues can hinder effective charge transfer and overall material stability.
The rational design and precise synthesis of COFs with tailored electronic structures for optimal charge transfer require further exploration. In addition, the recyclability and scalability of these candidates require further investigation to enable practical applications. Addressing these challenges through advanced material design and improved synthetic methodologies is crucial for unlocking the full potential of COF-based S-scheme heterojunction photocatalysts. Currently, the existing literature lacks a comprehensive review that systematically examines recent progress in COF-based S-scheme heterojunction photocatalysts. Considering the accelerating interest in this domain, a detailed analysis of their synthesis strategies, charge transfer mechanisms, and photocatalytic applications is essential for guiding future research. This review provides a detailed overview of the latest status of research, addresses existing challenges, and proposes future directions for advancing this promising field. By consolidating recent findings and offering insights into their potential applications, this review seeks to contribute to the continued development of high-performance COF-based S-scheme heterojunction photocatalysts. This review provides an in-depth review of COF-based S-scheme heterojunction photocatalysts, covering their fundamental properties, fabrication methods, various types of COF-based S-scheme heterojunctions, and photocatalytic mechanisms. Additionally, this review highlights recent advancements in their applications in hydrogen production, CO2 reduction, and H2O2 production. Finally, existing obstacles and future viewpoints are highlighted to provide further insights to promote this emerging research field.
Photocatalytic properties of COFs
COFs are largely structured organic polymers with remarkable porosities, offering significant potential for photocatalytic applications. The attractive properties of the COFs are discussed below.Morphology
COFs can be synthesized in 2D and 3D forms, with each structure providing distinct advantages for different applications such as separation, sensing, and catalysis.39,40 The most promising and emerging applications of COFs are photocatalytic solar fuel generation, where they effectively convert CO2 into useful products, including CH4, CO, CH3OH, and HCOOH, reduce N2 to NH3, and split water into H2 using solar energy.41–43 COFs are prepared using techniques such as imine condensation, boronate ester formation, and Schiff base condensation, which links organic monomers to create extended networks with periodic pores. These monomers contain active centers, such as boronic acids, amines, and aldehydes, facilitating the fabrication of intricate 2D or 3D structures.32 In 2D COFs, the organic molecules are arranged in layers connected by π–π stacking interactions.44 These interactions stabilize the structure and create channels for photocatalysis. The large surface area provided by these layers is crucial for photocatalytic applications, while π–π stacking also aids in charge transport, making 2D COFs suitable for photocatalysis.45,46 Additionally, 2D COFs are adaptable, allowing for modifications in surface area, pore size, and electrical properties by choosing specific building blocks and adjusting the synthesis process.47 On the other hand, 3D COFs have a more robust structure because of their extensive network of covalent bonds in three dimensions, offering high mechanical and thermal stability.48 This makes them ideal for applications that require durability and long-term performance under harsh conditions. The interconnected pores of 3D COFs provide a large surface area for reactant adsorption, making them valuable in processes like carbon capture and water splitting.29 While 2D COFs are more common, creating 3D COFs presents challenges due to the limited availability of stereoscopic building blocks and crystallization issues.49 Nevertheless, top-down or bottom-up methods have been used to produce COF nanosheets, providing opportunities to study the relationship between structure and performance for catalytic improvements.50Charge separation and migration
Among the main photocatalytic parameters, COFs have the ability to produce photoinduced charge carriers upon light exposure, which is enhanced by the careful choice of building materials that promote excellent charge migration, enabling the transformation of sunlight into chemical energy.51 By optimizing the electronic properties of COFs, such as through the design of donor–acceptor pairs or improving π-conjugation, their photocatalytic efficiency can be further increased.52 The combination of efficient electron transfer and structured electron channels makes COFs promising candidates for CO2 reduction and other photocatalytic applications.53 By incorporating photosensitizers or designing COFs with alternating donor–acceptor units, researchers can optimize charge transfer properties and enhance photocatalytic performance, while adjusting the bandgap and electronic properties further improves their efficiency.54 COFs' ability to form thin films is also advantageous for applications requiring fast mass and charge transport, though synthesizing highly crystalline and stable COF thin films can be challenging; however, techniques like layer-by-layer assembly, interfacial assembly, and support-assisted growth have made significant progress, demonstrating promise in heterogeneous photocatalysis, particularly for long-term stability and efficiency.55 In recent years, strategies such as designing donor–acceptor COFs, incorporating photosensitizers, and creating COF heterojunctions have been explored to improve photocatalytic activity.56 COFs can form heterojunctions with materials like inorganic semiconductors, MOFs, and carbon-based materials, which enhances their photocatalytic performance by providing the migration of photogenerated charge carriers, improving charge separation, and increasing efficiency for energy production and pollutant removal applications.57 The efficiency of COFs in photocatalysis is largely influenced by their capability to enable charge separation and transport, as the strength of exciton binding energy dictates how readily excitons dissociate into individual charge carriers.58 For optimal performance, excitons must migrate effectively through the COF network to reach active sites before recombining, making minimizing recombination and ensuring effective electron–hole separation key to enhancing the availability of free carriers for catalytic activity.59 Charge transport mechanisms enable these carriers to reach reaction sites where they contribute to photocatalytic transformations, and various strategies can improve this process, including the incorporation of heterojunctions, protonation, or the induction of an internal electric field.60 Heterojunctions establish an interface between materials with distinct band alignments, which aids in electron–hole separation, while protonation alters the electronic properties of COFs, leading to increased charge carrier mobility.61 Additionally, an internal electric field helps steer electrons and holes in opposite directions, thereby reducing their recombination rate and enhancing overall catalytic efficiency.62Surface area and porosity
Larger surface areas and well-defined pores provide optimal environments for substrates to interact with catalytic sites, facilitating efficient reactions.63 The highly porous nature of COFs, with customizable surface areas and pore sizes, enhances their ability to adsorb reactants and convert them into valuable products.64 Their stability, both chemically and physically, ensures durability and recyclability as photocatalysts, making them an ideal choice for photocatalytic applications.65 Additionally, COFs are crystalline materials formed through covalent bonding between multifunctional monomers, resulting in well-defined 2D and 3D structures.66 The geometry, pore distribution, and surface area of COFs might be carefully controlled through the picking of appropriate organic building blocks, enabling fine-tuning for specific applications.67 For photocatalytic applications, the surface area and pore size of COFs are critical factors. Adjustments to the stacking arrangement of COF nanosheets can influence the pore sizes and catalytic performance, while incorporating metal nanoparticles further enhances their photocatalytic properties. The combination of precise pore control, large surface areas, and the ability to host catalytic sites makes COFs highly effective for photocatalytic applications.68 COFs possess a modular design, allowing for precise control of their structural attributes, including topology, symmetry, pore size, and connectivity. This structural versatility is achieved by selecting organic precursors with distinct molecular lengths and symmetries.69 Due to their highly crystalline nature, COFs exhibit a substantial surface area and a uniform distribution of pores, which play a crucial role in offering abundant photocatalytic sites, facilitating substrate accommodation, and ensuring efficient mass transport during catalytic reactions.70 Additionally, their well-defined framework serves as an excellent matrix for anchoring molecular catalysts or photosensitizers, enhancing their applicability in heterogeneous photocatalysis.71 By carefully designing the pore architecture through the selection of specific monomeric units, it is possible to tailor both pore dimensions and morphology. Moreover, post-synthetic modifications offer additional avenues for fine-tuning the porous framework, as pore organization and surface area are closely tied to the crystallinity and packing arrangement of COFs. For example, highly crystalline COF-1 exhibits a notably greater specific surface area (2143 m2 g−1) in contrast to its amorphous counterpart, COP-1 (13.8 m2 g−1).72 The extensive surface area of COFs ensures accessible catalytic sites, while their porous network supports rapid charge migration toward the surface, optimizing their photocatalytic efficacy.Versatility
The versatility of COF structures offers considerable potential for tailoring materials for specific photocatalytic applications.51 This can be achieved through two primary approaches: the bottom-up method, where monomers are designed to form the desired COF directly, and post-synthetic modifications, where functional groups are added to existing COF structures.52 These modifications include metalation, where metal ions are introduced into the COFs, and other techniques like click reactions that introduce new functional groups to enhance properties.53 Chemical conversions, such as oxidation–reduction or cyclization, can also modulate the structure, providing additional means to fine-tune COF properties.54 COFs' high surface area, lightweight elements, and ability to adjust pore size (ranging from micropores to mesopores) render them perfect for a variety of applications.60 The pore size and shape can be customized through monomer design, although predicting these characteristics in 3D COFs is more challenging due to potential interpenetration of structures.61 Researchers control pore size using three strategies: adjusting the length of building blocks, modifying substituent groups, and altering the stacking arrangement of COF nanosheets.62 The pore size of COFs can also be customized. For example, halogens can be used to modify the pore environment of 2D COFs, enhancing their photocatalytic performance, while functionalized nanosheets can improve photocatalytic activity for fuel generation and pollutant removal.63 COFs' low density and high surface area, along with their ability to host functional groups, make them attractive for a variety of applications.64Stability
Stability is crucial for the practical use of COFs, especially for post-synthetic modifications.73 COFs generally exhibit effective thermal durability due to their strong covalent bonds, which enable them to maintain structural integrity at temperatures beyond 500 °C under inert conditions.74 While early boron-derived COFs showed inadequate chemical durability in water or acidic/basic environments, advancements have significantly improved their stability.54 Stronger linkages, such as hydrazine, ether, azine, and imine bonds, have enhanced the chemical stability of COFs, even under harsh conditions.55 Post-synthetic strategies, such as converting a 2D imine-linked COF into a more stable thiazole-linked version, further improve their stability and suitability for practical applications.75 COFs' thermal and chemical stability, due to the robust covalent bonds in their structure, allow them to maintain their properties in challenging environments.43 COFs' stability plays a key role in their practical use for photocatalytic applications. Advances in linkage chemistry have greatly improved the stability of COFs, making them more robust in various environments. Some COFs are stabilized through processes like aza-Diels–Alder cycloaddition or sulfurization, increasing their resilience under harsh conditions.76 A crucial aspect in the rational design of photocatalysts is ensuring both photostability and chemical durability. From the standpoint of reusability, it is vital that photocatalysts maintain long-term stability while withstanding different environmental factors such as acidic or basic conditions, oxidative species, co-catalysts, and sacrificial reagents.77 COFs can achieve remarkable stability by carefully selecting molecular components and robust linkages, thereby sustaining their photocatalytic efficiency in diverse reactions.78 Furthermore, COFs inherently resist photodegradation and photobleaching, enhancing their longevity under light exposure.49 In certain cases, their photochemical robustness can be further improved through protonation or post-synthetic modifications.65Band position
The performance of photoreactions depends on the alignment of the CB and VB of the material. By designing COFs with suitable energy levels, researchers can optimize their ability to absorb visible light (400–800 nm), enhance charge separation, and improve photocatalytic performance.79 Modifications, such as halogenation or the addition of functional groups like sulfone or porphyrin, can further tune COFs' energy levels and boost photocatalytic activity.80 Improved electron transfer and charge carrier mobility contribute to more efficient photocatalytic processes.81 Additionally, the inhibition of electron–hole recombination through structural modifications, such as chlorination or fluorination, enhances the overall photocatalytic performance.82 For a photochemical reaction to occur, the absorbed photon must have energy ≥ the bandgap of the catalyst. COFs exhibit photocatalytic activity that is highly dependent on their optical band structure, which ideally should be around 1.80 eV to maximize solar energy utilization.83 Typically, COFs possess optical band gaps in the range 1.50–3.0 eV, with a predominant value near 2.20 eV due to their extensive π-conjugation.33 The energy levels of COFs can be tailored by incorporating extended aromatic moieties, donor–acceptor pairs, or heteroatoms into their molecular structure, as well as by modifying their linkages or applying post-synthetic alterations.43 Beyond the band gap, the absolute energy levels of COFs are critical for photocatalytic processes. Functionalizing COFs or altering their linkage structures can significantly influence the band gap and band alignment, improving charge carrier dynamics and light absorption.70 Upon irradiation, electrons migrate from the VB to the CB, producing electron–hole pairs that transfer to the surface and drive redox processes.62 The presence of π-conjugation in COFs plays a key role in reducing the band gap, thereby enhancing their ability to capture light.51 Additionally, incorporating specific structural components or heteroatoms can further optimize electronic properties. The introduction of functional groups such as imidazole, porphyrin, or sulfone into the COF backbone can enhance light absorption.75 The inherent bandgap of COFs generally falls between 1.30 eV and 2.90 eV, allowing them to absorb light up to 800 nm.34 As a result, COFs with well-matched energy levels and strong optical absorption can serve as efficient photocatalysts.Crystallinity
COFs stand out from amorphous porous organic polymers (POPs) because of their ordered crystalline structures.84 This well-defined arrangement ensures that π-conjugated units are periodically positioned, allowing precise control over both topology and stacking through molecular engineering.85 The optical and electronic behavior of COFs is significantly influenced by the stacking arrangement and interlayer distances, as these factors regulate orbital interactions in both the ground and excited states.61 Unlike conventional organic polymers, COFs are synthesized using a bottom-up strategy that strategically organizes electron donors (D) and acceptors (A) in a repeating pattern.28 This structural design enhances charge separation upon photoexcitation while mitigating charge recombination. Additionally, the extended periodicity in layered COFs facilitates the efficient transport of charge carriers or excitons across the material, reducing recombination losses.39 Moreover, the crystalline structure of COFs contributes to increased charge carrier lifetime and kinetic energy, both of which are essential for efficient photocatalysis. Achieving high crystallinity in COFs requires careful selection of molecular precursors along with optimized reaction conditions, including solvent choice, catalyst type, reaction temperature, and synthesis duration.25 The long-range order in COFs reflects the structured positioning of building units at both local and extended scales. Several factors play a critical role in the formation of highly crystalline COFs. Due to variations in reaction dynamics and thermodynamic stability, the selection of linkage chemistry and molecular components greatly impacts the final crystallinity.86 Furthermore, reaction parameters such as temperature, pressure, monomer concentration, and synthesis duration are crucial in controlling nucleation and ensuring the formation of large, well-ordered COF crystals,69 among which optimization of the monomer concentration and reaction temperature is particularly vital for promoting crystallization. Typically, solvothermal synthesis of highly crystalline COFs requires a reaction period of 3 to 7 days.87 For instance, PI-COF-1, a polyimide-linked COF, was successfully synthesized over 5 to 7 days under solvothermal conditions.38 In general, well-ordered COFs exhibit better photocatalytic efficiency in contrast to their disordered counterparts, as their high crystallinity reduces defect sites, suppresses charge carrier recombination, and enhances charge transport efficiency.85 Despite their promising properties for photocatalysis, challenges remain in enhancing the conductivity of COFs. Solutions include developing hybrids with mobile semiconductors or increasing the crystallinity and conjugation of COFs.68 The well-defined pore structures, high surface area, and wide light harvesting capabilities of COFs make them excellent candidates for heterogeneous photocatalysis, particularly in solar fuel generation under harsh conditions.69 Their high crystallinity and periodic arrangements promote charge separation and migration, which is pivotal for photocatalytic efficiency.35Heterojunction formation of COFs
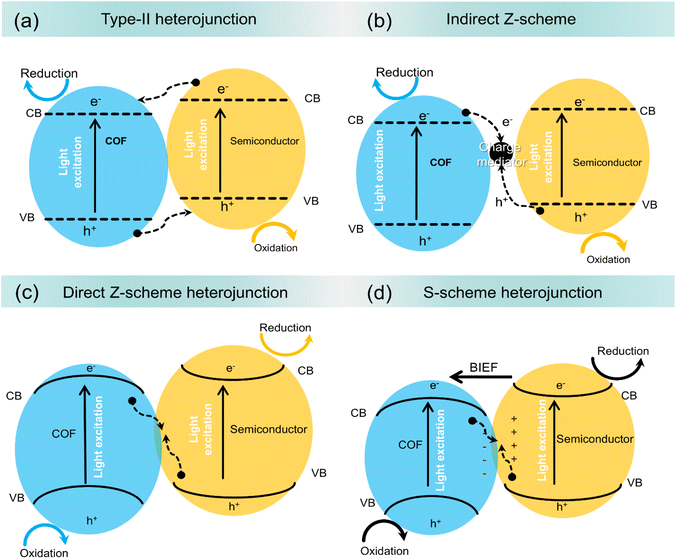
Most COFs possess a relatively large bandgap, which restricts their ability to efficiently absorb and exploit solar radiation, leading to low photocatalytic efficiency. This is partly due to the slow electron transfer rate and reduced quantum efficiency.25,32 One potential solution is to combine COFs with semiconductors, as the high porosity and large surface area of COFs can enhance the dispersion of coupled materials. This improved dispersion support hinders cluster formation and ensures a more regular supply of reactive sites, thereby enhancing the photocatalytic process.26,31 The formation of heterojunctions between COFs and semiconductor materials further improves the overall performance by enhancing light absorption, particularly in the visible range.30 These heterojunctions suppress the recombination of charge carriers, and increasing the transfer efficiency of photogenerated electron–hole pairs. As a result, the separation and transfer of charges are enhanced, creating supportive conditions for photochemical reactions.27 There are three main types of heterojunctions commonly employed in COF-semiconductor systems: type-II, Z-scheme, and S-scheme heterojunctions. The configuration of each heterojunction dictates the behavior of the photoinduced charge pairs at the interface between the two materials. In these configurations, the bandgaps of the semiconductors differ, and their CB and VB positions align to facilitate charge transfer between the materials. Herein, we discuss three different types of COF-based heterojunction photocatalysts, Fig. 1.COF-based type II heterojunction
In a type-II heterojunction, when the semiconductor has a higher CB energy level and a lower VB energy level than the integrated COF, as shown in Fig. 1a, the photoinduced electrons from the CB of the semiconductor move to the CB of the COF. Meanwhile, photogenerated holes from the VB of the COF migrate to the VB of the integrated semiconductor.28,44 This transfer results in the spatial separation of photoinduced charge carriers, diminishing the likelihood of recombination and improving the efficiency of the photocatalytic reaction.33 However, type-II heterojunction systems have some limitations. A major issue is the repulsion between photoinduced electrons in the CBs of the COF and integrated semiconductor.27 This repulsion can slow down or prevent the efficient migration of photoinduced electrons from the CB of the semiconductor to that of the COF.85 Additionally, this repulsion hinders the migration of photoinduced holes from the VB of the COF to the VB of the integrated semiconductor.69 From a thermodynamic standpoint, this reduces the overall redox capacity of the system, limiting its ability to drive photocatalytic reactions. Consequently, although type-II heterojunctions can be effective in some cases, this mechanistic limitation hinders their potential in traditional photocatalytic applications. For example, Yu et al. employed an in situ approach to couple TiO2 with three different COFs (TTh-COF, Th-COF, and Ph-COF) to form type-II heterojunctions to study the photocatalytic activity for uranium reduction.88Compared to the pure COF and TiO2, the XPS spectra of the TTh-COF-TiO2 heterojunction showed that the binding energies of the COF orbitals shifted positively, while those of TiO2 orbitals shifted negatively, as shown in Fig. 2a and b. This revealed not only the transport of electrons from TTh-COF to TiO2 due to the generation of N–Ti covalent bonds but also confirmed the development of the heterojunction between TiO2 and TTh-COF. Furthermore, the extended light absorption ability of TTh-COF-TiO2 compared with that of pure TiO2 (Fig. 2c) verified that heterojunction formation effectively promoted light absorption. Similarly, the smallest diameter in the EIS Nyquist plots and the highest photocurrent density of TTh-COF-TiO2 validated the successful formation of the heterojunction. The optimized TTh-COF-TiO2 photocatalyst displayed a maximum uranium reduction efficiency of 98% within 40 min of visible-light illumination. Additionally, six recycling experiments revealed only a small performance loss, from 98% to 95.3%, indicating a strong stability. Upon light excitation, the photogenerated electrons in the CB of TTh-COF shifted to the CB of TiO2 through the N–Ti covalent bond, whereas holes migrated from the VB of TiO2 to that of TTh-COF. This resulted in the enhanced separation of photogenerated charge pairs. Ultimately, the accumulated electrons in the CB of TiO2 removed uranium, whereas holes in the VB of TTh-COF were depleted by methanol. Similarly, Wang et al. reported that the development of a type-II heterojunction between CdS and TpPa-1 COF provided enhanced separation and transfer of photogenerated charge pairs, leading to an NH3 generation rate of 241 μmol g−1 h−1 from N2 fixation, which was 3- and 17-fold superior to that of CdS and TpPa-1, respectively.90
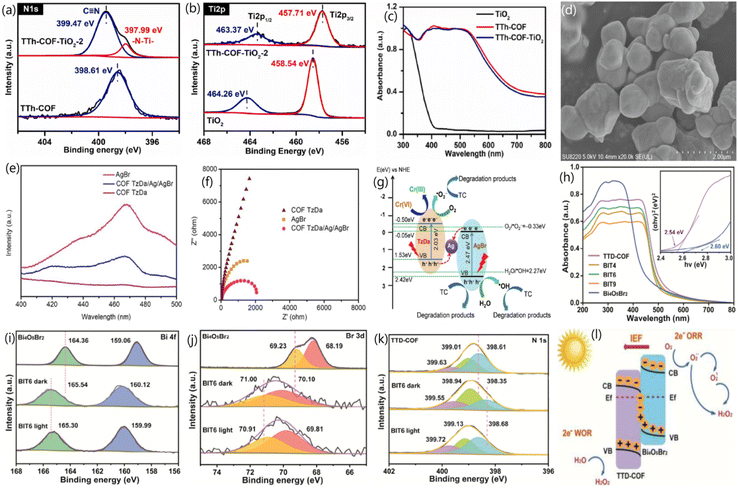 | ||
| Fig. 2 High resolution XPS spectra of pure materials and TTh-COF-TiO2 heterojunction (a) N 1s, (b) Ti 2p, and (c) absorption spectra of fabricated materials,88 copyright 2024, Elsevier (d) SEM image, (e) PL spectra, (f) EIS Nyquist plots, and (g) ASS Z-scheme electron–hole transfer process of TzDa/Ag/AgBr,71 copyright 2022, Elsevier. (h) Absorption spectra and high-resolution XPS spectra of different materials: (i) Bi 4f, (j) Br 3d, (k) N 1s, and (l) S-scheme charge-migration mechanism of Bi4O5Br2/COF,89 copyright 2024, American Chemical Society. | ||
COF-based Z-scheme heterojunction
In type-II heterojunctions, two semiconductors with different band structures are coupled, which helps suppress the recombination of photoinduced charge carriers by spatially separating them. However, this separation can reduce the photocatalyst's redox capability because electrons and holes are not available at the same site for oxidation and reduction reactions, resulting in a lower overall performance.81 Z-scheme heterojunctions were proposed to address the above limitation, mimicking the catalytic mechanism of photosynthesis. This structure retains a high redox potential while separating the electrons and holes, enhancing photocatalytic performance.82 Z-scheme heterojunctions can be categorized as indirect Z-scheme heterojunctions and direct Z-scheme heterojunctions depending on the existence of charge migration mediators, as shown in Fig. 1b and c.83 Indirect Z-scheme systems use mediators, which can be liquid-phase electron donor/acceptor pairs or solid-state mediators like noble metals.57 In conventional Z-scheme heterojunctions, two semiconductors are paired with electron donor/acceptor species in the liquid phase.23 The component with a more negative reducibility preserves the photoinduced electrons, while the component with a superior oxidizability preserves the photoinduced holes, preventing recombination, and retaining a strong redox potential. All-solid-state (ASS) Z-scheme heterojunctions use precious metals as charge mediators, preventing reverse reactions typical in liquid-phase systems.24 The solid-state mediator provides better stability and suppresses unwanted reactions that could degrade the photocatalyst.88Direct Z-scheme heterojunctions combine the advantages of both the indirect types. They preserve both excellent redox properties and efficient electron–hole separation, maintaining strong oxidative and reductive capabilities without relying on mediators. This results in higher photocatalytic efficiency.90 For instance, Shi et al. utilized Ag as a charge mediator between AgBr and COF TzDa to construct the ASS Z-scheme heterojunction using the in situ technique for photocatalytic pollutant removal under simulated sunlight irradiation.90 The microstructure analysis (Fig. 2d) revealed the simultaneous dispersion of AgBr and Ag nanoparticles onto COF TzDa nanosheets, forming strong interfacial contacts that facilitated rapid mass transport and extended light absorption, thereby enhancing the photoreaction. Additionally, the porous structure of COF TzDa/Ag/AgBr increases the surface area, promoting catalyst–light interactions, charge migration, and reactant transport through the pores. This was confirmed by the lower PL intensity and smaller EIS Nyquist arc radius for TzDa/Ag/AgBr compared to the pure materials, as shown in Fig. 2e and f. The optimized TzDa/Ag/AgBr photocatalyst demonstrated a maximum TC removal rate of 80% within 30 min, which was ascribed to the widened optical absorption, promoted electron–hole separation, and large surface area. Furthermore, TzDa/Ag/AgBr exhibited negligible performance loss after four consecutive recycling experiments, with no changes in the XRD results, indicating promising efficiency and structural stability of the photocatalyst. Under light irradiation, the photoinduced electrons in the CB of AgBr recombined with the photoinduced holes via the Ag mediator, leading to the increased separation of photoinduced electrons in the CB of COF TzDa and holes in the VB of AgBr, as illustrated in Fig. 2g. These preserved electrons reduce O2 to ˙O2− radicals, where holes react with H2O to produce ˙OH radicals, which subsequently degrade pollutants.
Similarly, Yang et al. formed a direct Z-scheme WO3/Tp-TAPB heterojunction for photocatalytic H2O2 production under visible light excitation.66 The microstructure examination confirmed the effective integration of WO3 nanocubes with COF nanosheets, resulting in extended light absorption and improved transfer and separation of photogenerated charge pairs. Additionally, compared to pure catalysts, the improved surface area of the heterojunction not only provided numerous reactive sites for redox reactions but also promoted light interaction and reactant diffusion, enhancing the photocatalytic performance. Consequently, the optimized WO3/Tp-TAPB exhibited the highest H2O2 production rate of 1.49 mmol g−1 h−1, which was approximately 72 and 3 fold higher than those of WO3 and Tp-TAPB, respectively. Furthermore, the optimized WO3/Tp-TAPB catalyst retained both its performance and structural stability after four recycling experiments, confirming the robust stability of the photocatalyst. Upon light excitation, the photogenerated electrons in the CB of WO3 directly perished with holes in the VB of Tp-TAPB, thus maintaining the photoinduced electrons and holes in the CB of Tp-TAPB and VB of WO3, respectively. These retained electrons and holes then produce ˙O2− and ˙OH radicals, leading to the generation of H2O2. COFs have attracted significant attention for their use in Z-scheme heterojunctions for photocatalysis, especially in liquid-phase systems. However, these systems face challenges such as unwanted charge migration from the CB of the coupled component to the CB of the COF, which leads to charge recombination and reverse reactions that reduce efficiency.91 Additionally, the liquid-phase nature of these systems limits their applications. COF-based ASS Z-scheme heterosystems rely on expensive precious metals as charge mediators to facilitate electron transfer.47 While these metals can improve charge separation, they also increase the overall cost and interfere with the semiconductor's ability to absorb light, diminishing the system's overall efficiency.27 Furthermore, the precise mechanisms of charge transfer in these systems are still not fully understood, leading to ongoing debates. A more recent development, the direct Z-scheme heterojunction, addresses these issues by eliminating the need for electron mediators. This system involves two semiconductors in direct contact, which lowers costs and retains strong redox capabilities.53 However, despite its advantages, it shares similarities with older Z-scheme systems, and some still mistakenly associate it with the drawbacks of reverse electron transfer seen in those previous models.92 To overcome these limitations, Yu et al. documented the idea of the S-scheme heterojunction.93
COF-based S-scheme heterojunction
The S-scheme heterojunction is designed to optimize charge separation and minimize reverse reactions by combining two semiconductors with different energy levels: the more closely integrated, the difference in their Ef levels is eliminated, resulting in the migration of free electrons from the reduced photocatalyst (RP) to the oxidized photocatalyst (OP).28 This electron transfer facilitates a positively charged layer near the RP, bending its band structure upwards, while a negatively charged layer develops near the side of OP, bending its band structure downward, as shown in Fig. 1d.68 The resulting IEF, which points from the RP to the OP, plays a crucial role in separating charge carriers and preventing recombination. It also aids in the recombination and perishing of photogenerated holes in the VB of the RP with electrons in the CB of the OP, consuming unnecessary charge carriers and improving the system's efficiency.56 Additionally, this electric field and the Coulomb repulsion between electrons prevent the unwanted transport of photoinduced electrons from the CB of the RP to the CB of the OP, effectively inhibiting reverse reactions and maintaining a strong redox capacity in the system.94 As a result, the S-scheme heterojunction is perceived as a more excellent and cost-effective alternative to conventional Z-scheme and type-II heterojunctions, addressing the limitations of older systems and enhancing photocatalytic performance.For example, Yue et al. integrated TTD-COF strips with Bi4O5Br2 spheres through a solvothermal process to create an S-scheme heterojunction, with the aim of enhancing the photocatalytic performance for H2O2 production.89 Microstructure analysis demonstrated that TTD-COF was effectively wrapped around Bi4O5Br2via strong interfacial interactions, resulting in increased surface area and enhanced light absorption (Fig. 2h). Additionally, the higher current density of the heterojunction, in contrast to bare semiconductors, further confirmed the enhanced separation and transport of photoinduced charge pairs, attributed to the generation of the S-scheme heterojunction between Bi4O5Br2 and TTD-COF.
Consequently, the Bi4O5Br2/COF photocatalyst exhibited the highest H2O2 production rate of 5221 μmol g−1 h−1, nearly double that of TTD-COF alone. XPS spectra showed that the binding energies of Bi4O5Br2 orbitals exhibited a red shift, while those of TTD-COF displayed a blue shift, indicating the migration of free electrons from Bi4O5Br2 to TTD-COF due to the formation of the heterojunction. Under light irradiation, however, the binding energies of the Bi4O5Br2 and TTD-COF orbitals in the heterojunction exhibited a reverse shift compared to the dark state, as shown in Fig. 2i–k, confirming that photoinduced electrons migrated from TTD-COF to Bi4O5Br2 in accordance with the S-scheme charge migration mechanism. The preserved electrons in the CB of Bi4O5Br2 and holes in the VB of TTD-COF generated ˙O2−and ˙OH radicals, leading to H2O2 production. Similarly, An et al. employed a self-assembly technique to integrate TiO2 with Tp-Tta COF via an S-scheme heterojunction and investigated the photocatalytic performance for CO2 reduction.49 Microstructure analysis confirmed a strong interface interaction between Tp-Tta COF and TiO2 nanosheets, which resulted in an increased surface area that promoted mass transport and charge migration. Additionally, the XPS study confirmed the successful fabrication of the TiO2/COF heterojunction, which extended the light absorption, providing additional photoinduced charge carriers to support the photoreaction. The optimized TiO2/COF heterojunction exhibited the highest CO generation efficiency of 11.6 μmol h−1, 14.5 fold greater compared to that of pure TiO2. EPR spectra confirmed the development of the S-scheme charge transfer between TiO2 and COF. Upon light excitation, the photogenerated electrons in the CB of TiO2 perished with holes in the VB of COF, leading to the separation of electrons in the CB of COF and holes in the VB of TiO2, thus facilitating CO2 reduction. Although the formation of COF-based S-scheme heterojunctions has immense potential for photocatalytic applications, research in this field is still in its early stages. Consequently, significant research is expected in the near future to explore this emerging area.
Synthesis methods
The formation of COF-based S-scheme heterojunctions is a potential approach for developing highly efficient photocatalysts. Owing to their unique properties, integrating COFs with other semiconductors presents a challenging synthetic approach. Additionally, precise control over the crystallinity and porosity of COFs during heterojunction formation requires careful attention to factors such as the pressure, processing time, reaction temperature, and solvent combinations. These parameters play a critical role in maintaining the balance between the synthesis and crystallization of COF-based S-scheme heterojunctions. The common preparation methods for these photocatalysts are summarized in Fig. 3.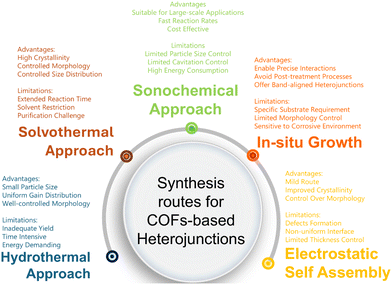 | ||
| Fig. 3 Summary of various synthesis methods discussed to synthesize COF-based S-scheme heterojunctions with corresponding advantages and limitations. | ||
Hydrothermal synthesis
The hydrothermal synthesis route is extensively adopted, especially in the preparation of COF-based photocatalysts because it is economical, as it relies primarily on water as the solvent, which is abundant and inexpensive.95 Hydrothermal synthesis offers several advantages, including the ability to produce photocatalysts with good grain development, small particle sizes, uniform distribution, and well-controlled morphology.96 These features are essential for photocatalytic reactions, as they improve the COF-based material's efficiency in applications, including hydrogen production, pollution degradation, and other energy-related reactions. One of the pivotal characteristics of the hydrothermal method is its ability to produce high-crystallinity COF-based materials with precise control over their morphology.97 Notably, the precursors of the COF and selected semiconductor are mixed in a solvent, typically water, under stirring or ultrasonic treatment, and then placed in an autoclave. The process typically involves dissolving the sample in water and heating it to a specific temperature (80–200 °C), where it undergoes re-crystallization.98 During this period, surfactants or templates may be added to guide the formation of the desired structure. These surfactants facilitate the self-assembly of particles, and as the crystals grow, they form the desired shape, whether they are fine particles, thin films, or more complex structures, such as hollow particles.99 Overall, the hydrothermal method offers precise control over the crystallization process, making it ideal for synthesizing a wide range of photocatalytic materials with specific properties.100 These materials are typically used for applications that require a high surface area, efficient electron transport, and excellent photocatalytic performance. The capability to regulate the size, shape, and distribution of the nanoparticles allows for the design of advanced materials with tailored properties, which is a significant advantage in fields like environmental remediation and renewable energy.101For example, Zhang et al. employed a hydrothermal method to integrate BiOBr and TzDa COF to evaluate its photocatalytic performance in Cr(VI) reduction.102 The precursors of both materials were dissolved in ethylene glycol (EG) under stirring, and the resulting uniform solution was transferred to an autoclave for hydrothermal treatment at 120 °C for 6 h. The final product was collected, washed, and dried at 60 °C for 6 h. The synthetic process is illustrated in Fig. 4a. As shown in Fig. 4b, microstructure analysis revealed that the BiOBr nanoplates (∼100 nm) were effectively anchored with the porous TzDa COF, enhancing the surface area, light absorption, and charge separation, thereby improving the photocatalytic performance. Consequently, the optimized BiOBr/TzDa COF photocatalyst achieved a Cr(VI) reduction efficiency of 97% within 40 min under visible-light irradiation. The microwave-assisted hydrothermal synthesis, which uses microwaves to generate heat, has also been effectively employed to prepare COF-based S-scheme heterojunctions.103 Replacing conventional heating with microwave heating accelerates the reaction rate, provides better control over the process, yields higher product purity, and enhances efficiency.87 However, the use of polar solvents is essential for effective heating during the reaction.28 For example, Bao et al. employed a microwave-assisted hydrothermal process to fabricate a TpBD COF/ZnIn2S4 S-scheme heterojunction to investigate its photocatalytic H2 generation performance.48 The precursors, in the required molar ratio, were mixed in water under ultrasonication before initiating the microwave-assisted hydrothermal reaction, as depicted in Fig. 4c. SEM analysis (Fig. 4d) revealed that the TpBD COF particles (2–7 μm) were effectively integrated with the flower-like ZnIn2S4 structures, providing abundant active sites and enhanced light absorption. Furthermore, the strong interfacial contact between TpBD COF and ZnIn2S4 promoted the separation and migration of photogenerated carriers, resulting in an effective H2 generation efficiency of 2304 μmol g−1 h−1 under visible light. In addition, in situ hydrothermal synthesis was employed to fabricate COF-based S-scheme heterojunction photocatalysts. This approach ensures strong interfacial interactions, improves the separation and transport of photoinduced charge pairs, and allows precise control over the heterojunction's morphology.33 As an example, Wang et al. utilized an in situ hydrothermal process to fabricate defective ZnIn2S4/TpPa-1 S-scheme photocatalysts for H2 generation.104 Microstructure analysis revealed that flower-like ZnIn2S4 nanosheets were intimately connected with block-like TpPa-1, resulting in an improved surface area, widened optical absorption, and enhanced charge dynamics. The optimized ZnIn2S4/TpPa-1 photocatalyst achieved an outstanding H2 production efficiency of 2745 μmol g−1 h−1 under visible light. Despite the promising potential of in situ hydrothermal synthesis, its application in the fabrication of COF-based S-scheme heterojunction photocatalysts remains limited. However, it is anticipated that this method will attract increasing attention in the development of S-scheme photocatalysts with yields suitable for industrial applications.
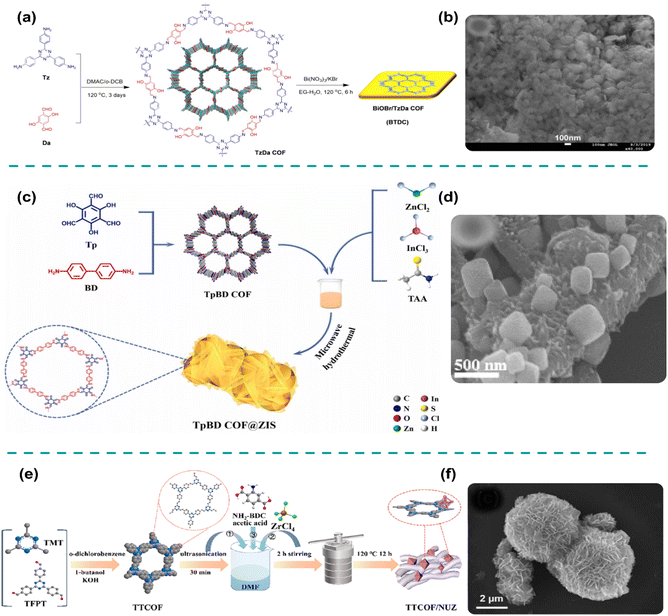 | ||
| Fig. 4 (a) Synthetic process and (b) SEM image of BiOBr/TzDa COF. Reproduced with permission from ref. 102, copyright 2021, Elsevier. (c) Synthetic scheme, and (d) SEM image of TpBD COF/ZnIn2S4. Reproduced with permission from ref. 48, copyright 2023, Elsevier. (e) Synthetic scheme, and (f) SEM image of COF/MOF. Reproduced with permission from ref. 81, copyright 2022, American Chemical Society. | ||
The hydrothermal technique is effective in generating highly crystalline COFs with well-defined particle sizes and minimal aggregation.103 However, its confined reaction conditions result in relatively low yields.33 This method, though suitable for large-scale synthesis, is time-intensive, energy-demanding, and often requires specialized equipment.48 It enables the formation of COFs under controlled conditions, yielding materials with minimal structural defects and uniform morphology.102 However, prolonged exposure to high temperatures and pressures poses challenges for industrial applications. Moreover, COFs with borate linkages struggle with stability in humid environments, limiting their usability in specific applications.104
Solvothermal synthesis
The solvothermal preparation technique is performed within a sealed container under high pressure and temperature above the boiling point of the chosen solvent. This technique begins by dissolving precursors in a solvent (such as tetrahydrofuran, dimethyl sulfoxide, N,N-dimethylformamide or 1,4-dioxane).105,106 Once dissolved, the mixture is kept in a stainless-steel Teflon-lined autoclave to withstand the high temperatures and pressures involved.107 The autoclave is then heated in an oven, typically within a temperature range of 100 °C to 200 °C, to facilitate the reaction. The duration of the heating process generally ranges from 48 to 96 h, but can be extended by relying on the features of the precursors and the complexity of the desired product.108,109 During this reaction, the process occurs heterogeneously, meaning that the different components in the mixture interact in various phases, which is key for the formation of COF-based heterojunctions. As the autoclave heats up, the pressure inside the container increases significantly. This elevated pressure accelerates the reaction rate, which in turn promotes the nucleation of particles and the gradual growth of COF crystals.110 After the reaction is complete, the final product is isolated using a centrifuge, a process that can be repeated in different stages to ensure complete separation of the crystalline products.85 This is known as the solvothermal reaction and is highly effective in synthesizing COF-based heterojunctions, as it allows for a controlled environment that promotes crystal formation. For example, Niu et al. used N,N-dimethylformamide (DMF) to develop a COF/MOF S-scheme heterojunction via solvothermal synthesis, investigating its photocatalytic performance for CO2 reduction.81 In this process (Fig. 4e), the required amounts of COF and MOF precursors were mixed in DMF under stirring, followed by a solvothermal treatment at 120 °C for approximately 12 hours. TEM analysis (Fig. 4f) revealed a robust interfacial contact between the COF and MOF, which facilitated the migration and separation of photoinduced charges. This was confirmed by the smallest EIS Nyquist arc diameter of the composite, in contrast to that of the pure materials. Consequently, the COF/MOF photocatalyst exhibited excellent CO generation performance, which was 5 and 4.4 fold superior to that of MOF and COF, respectively. Similarly, the precursors of g-C3N4 and COF were mixed in a DMF solution under stirring, followed by heat treatment at 120 °C for one day under vacuum.85 The g-C3N4/COF S-scheme heterojunction was successfully developed. The COF films grown on g-C3N4 enhanced the surface area, widened the light-harvesting range, and promoted the separation and transfer of photogenerated electron–hole pairs, leading to a sevenfold increase in hydrogen generation performance in contrast to bare g-C3N4. The selectivity of solvents plays a crucial role during the solvothermal synthesis of COF-based S-scheme heterojunction photocatalysts.30 In some cases, a combination of solvents has been shown to improve crystallinity and porosity, which in turn promotes surface area, light absorption, and charge separation.47 However, despite its effectiveness, the solvothermal synthesis route has some drawbacks, particularly its long reaction times, significant energy consumption required to maintain high temperatures for extended periods, and the need for specialized, bulky equipment that can handle high-pressure conditions.To overcome these challenges, microwave-assisted solvothermal synthesis has been introduced as a faster and more energy-efficient alternative. Microwaves, first discovered in 1946 by P. L. Spencers, are a form of electromagnetic radiation with wavelengths ranging from 1 m to 1 mm and frequencies ranging from 300 MHz to 300 GHz.111 Local microwave ovens usually operate at a frequency of 2.45 GHz.48 Microwave-assisted solvothermal synthesis takes advantage of these microwaves to accelerate chemical reactions, significantly reducing the time required for synthesis.55 This method can carry out reactions within just 4 minutes to 4 hours, depending on the nature of the reaction and the materials involved. The process works using an oscillating electric field generated by microwaves, which causes the dipole molecules present in the reaction mixture to move rapidly. This motion generates heat, which further accelerates the chemical reactions.112,113 The heat generated by this process improves the nucleation rate of COF-based materials, leading to faster crystal formation.114 One of the major advantages of microwave-assisted solvothermal synthesis is that it offers precise control over both temperature and pressure, which is vital for achieving high-quality products. This method is particularly beneficial for synthesizing materials with specific morphological characteristics, as it permits a better command over the shape and dimension of the final product.38 Additionally, the microwave-assisted solvothermal method produces high crystallization rates and improved purity, making it ideal for applications that require materials with well-defined structures and properties. The shorter synthesis times and the ability to tailor product morphology make microwave-assisted synthesis a potential substitute to conventional methods such as solvothermal fabrication, which typically require much longer reaction times and more cumbersome equipment.58 For instance, microwave-assisted solvothermal processes have demonstrated greater efficacy than the conventional solvothermal method in increasing the surface area and light absorption of the TpPa-1/BiVO4 S-scheme heterojunction photocatalyst, leading to nearly 72-fold higher H2O2 production compared to pure BiVO4 under visible light.55 Notably, the solvothermal method is a simple and efficient technique for synthesizing COF-based S-scheme heterojunctions, enabling faster reaction rates and larger yields, thereby facilitating the development of high-performance photocatalysts. The in situ solvothermal growth of COF onto a semiconductor forms a heterojunction characterized by robust interfacial interactions between the constituent semiconductors, enhanced porosity and surface area, and strong chemical stability.114 Furthermore, this direct integration of materials optimizes band alignment, reduces interfacial charge migration resistance, and consequently improves electron–hole separation and photocatalytic efficiency.28 As an example, Sun et al. utilized an in situ solvothermal growth process to fabricate a WO3/TpPa-1 S-scheme heterojunction with improved surface area, distributed light harvesting, and enhanced charge separation, achieving nearly fivefold higher H2 production compared to pure TpPa-1 under visible light.86 The solvothermal approach is widely utilized for synthesizing COF-based materials due to its straightforward process and ability to produce high-quality crystals.85 Despite these advantages, it demands extended reaction times, organic solvents, and strict reaction conditions, making it challenging to control.108 This method typically employs high-dielectric, proton-donating solvents, while pH adjustments through acids or bases help in achieving desired material properties.106 The reaction occurs at temperatures exceeding the solvent's boiling point, ensuring higher yields with controlled morphology, size distribution, and crystallinity.81 However, solvent removal from the COF pores is a complex process, often requiring vacuum drying or solvent-based washing with ethanol or methanol.85
Electrostatic self-assembly synthesis
Self-assembly is a process in which small components spontaneously organize themselves into larger ordered structures. This phenomenon is seen in nature, such as when molecules form cell membranes, viruses, or biopolymer fibers.41 Recently, scientists and engineers have adapted this process to create nanoscale materials in the laboratory using a bottom-up approach. In this approach, the individual components arrange themselves into specific, pre-designed structures to form larger systems, minimizing the overall free energy of the system.46One common method of self-assembly involves electrostatic interactions, which are especially effective in producing heterojunctions.49 Electrostatic self-assembly relies on electrostatic interactions between nanoparticles of opposite charges, causing them to naturally align and organize in a medium. This method is gentle and allows for specific regulation of the shape and size of the assembled particles.58 For example, when using electrostatic interactions, COFs and semiconductors with opposite charges can attract each other, resulting in their assembly into the desired structure.65 Since the self-assembly process occurs under mild conditions, it preserves the original properties of COFs and semiconductors, leading to a heterojunction that retains the characteristics of both materials.92 Additionally, the self-assembly process prevents these materials from aggregating.96 As an example, Wang et al. employed a self-assembly method to couple Tp-Tta COF with g-C3N4, forming an S-scheme g-C3N4/Tp-Tta COF heterojunction for photocatalytic CO2 reduction under visible light.46 The precursors of both semiconductors were mixed in water, ultrasonicated, and heated at 80 °C until the solvent evaporated to obtain the heterojunction. The synthesis scheme is shown in Fig. 5a. XRD analysis confirmed that the original phases of both materials were preserved in the heterojunction, with no impurities or transformation phases observed, demonstrating the effectiveness of the self-assembly synthesis. The TEM image (Fig. 5b) revealed that the self-assembly process effectively distributed Tp-Tta COF nanosheets onto g-C3N4, creating an interfacial contact that facilitated the separation and migration of charge carriers while extending light absorption up to 700 nm, as validated by DRS and PL analyses. Consequently, the optimized g-C3N4/Tp-Tta COF system exhibited the maximized CO generation rate of 11.25 μmol h−1, which was 45 fold greater compared to that of pure g-C3N4.
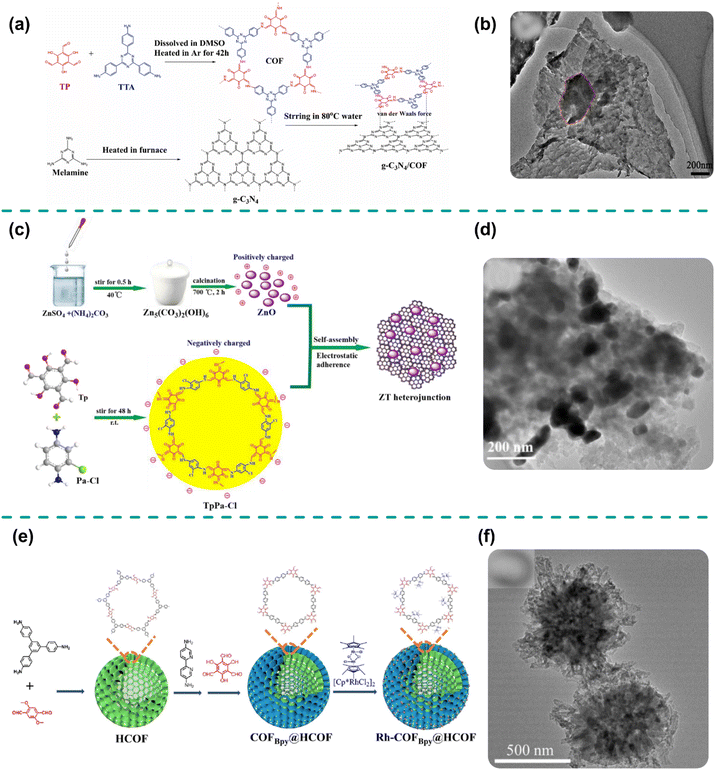 | ||
| Fig. 5 (a) Synthetic process and (b) TEM image of g-C3N4/Tp-Tta COF. Reproduced with permission from ref. 46, copyright 2022, Elsevier. (c) Synthetic process, and (d) TEM image of ZnO/TpPa-Cl. Reproduced with permission from ref. 50, copyright 2022, Elsevier. (e) Synthesis process and (f) TEM image of Rh-COF@COF. Reproduced with permission from ref. 105, copyright 2023, Elsevier. | ||
Similarly, Zhang et al. utilized a self-assembly process to integrate positively charged ZnO nanoparticles with negatively charged TpPa-Cl, creating an S-scheme heterojunction for photocatalytic H2O2 production.50 Initially, the authors employed Schiff-base condensation to synthesize TpPa-Cl, followed by calcination of ZnO precursors at 700 °C to obtain ZnO, as depicted in Fig. 5c. Subsequently, ZnO nanoparticles were anchored onto TpPa-Cl. XRD patterns confirmed that no phase transformation occurred in either ZnO or TpPa-Cl. Moreover, TEM analysis (Fig. 5d) demonstrated that the ZnO nanoparticles were intimately anchored onto TpPa-Cl, enhancing the porosity and surface area, thereby facilitating reactant adsorption and increasing the density of active sites. Additionally, the robust interfacial interaction between ZnO and TpPa-Cl significantly improved the separation and transport of photoinduced charge carriers, resulting in a maximum H2O2 generation efficiency of 2443 μmol g−1 h−1 under simulated sunlight, which was nearly nine times higher than that of pure TpPa-Cl. However, the limitations of electrostatic forces often constrain the complexity and diversity of the structures that can be developed, thereby hindering further advancements in these COF-based heterojunctions. This method ensures precise control over the interaction between COFs and semiconductors, leading to improved crystallinity and tunable functional properties.41 It enables synthesis under mild conditions, preserving structural integrity and enhancing material stability.50 However, achieving a uniform interface remains difficult, and controlling COF thickness is a challenge.46 Additionally, defects and interface irregularities formed during the assembly process may hinder the photocatalytic efficiency of the resulting material.96
In situ growth
The in situ growth synthesis of COF-based S-scheme heterojunctions is a versatile method that hinges on the intrinsic characteristics of the materials involved. This approach takes advantage of the functional groups and surface properties of both the COFs and semiconductors, enabling precise interactions essential for seamless integration.115 Without the need for additional post-treatment processes, this strategy simplifies the fabrication while ensuring robust interfacial contact. Such a contact is pivotal in creating band-aligned heterojunctions that enhance charge carrier dynamics, leading to faster separation and migration of photogenerated charge carriers.116 Moreover, this synthesis method ensures the uniform deposition of COFs onto the semiconductor surface, resulting in a well-ordered, porous structure. This uniformity not only enhances the material's adsorption ability for reactants but also enhances their diffusion efficiency within the photocatalyst framework.117 The maximized porosity further contributes to a higher density of active sites, thereby supporting prolonged carrier lifetimes and improving the overall photocatalytic efficiency.117 Additionally, the covalent interactions that develop during this process impart remarkable thermal stability to the material. This stability is critical for maintaining the integrity and performance of the photocatalyst under extreme operating conditions, making it highly durable for long-term applications.118 The synergy between the structural, interfacial, and electronic properties enabled by this method paves the way for the development of advanced photocatalytic systems with superior sunlight conversion capabilities.11 For instance, Zhao et al. utilized an in situ growth approach to prepare a hierarchical core–shell S-scheme heterojunction by integrating hierarchical Rh-COF with a hollow COF, where Rh-COF functioned as the shell.105Fig. 5e illustrates the schematic preparation of the hierarchical Rh-COF@COF core–shell structure. TEM analysis (Fig. 5f) revealed that the hollow COF core was encapsulated with a hierarchical Rh-COF shell, resulting in an increased surface area that enhanced the catalyst–light interaction and facilitated mass transport. This structural optimization significantly improved the separation and transport of the photoinduced charge carriers, thereby supporting the photocatalytic reaction. The photocatalytic performance of the S-scheme Rh-COF@COF photocatalyst was 3.7 times higher than that of pure Rh-COF under visible-light irradiation.Similarly, Qiu et al. employed an in situ growth process to grow In2S3 nanosheets onto COF substrates, aiming to evaluate their photocatalytic performance for H2O2 generation.69 The authors initially synthesized COF through Schiff base condensation of TAPB and DMTP, which was subsequently dissolved with InCl3 in water. Indium ions coordinated with the active sites on the COF, while TAA decomposed to release sulfur ions, leading to the formation of In2S3 nanosheets robustly anchored to the COF. Microstructural analysis revealed that the core–shell nanostructure of COF/In2S3 developed through the network growth of In2S3 nanosheets on the COF. This integration increased the surface area from 87 to 267 m2 g−1, extended the light absorption edge to 700 nm, and enhanced the separation and transfer of photogenerated charge pairs, as confirmed by various characterization techniques. The optimized COF/In2S3 photocatalyst achieved a remarkable H2O2 generation efficiency of 5713.2 μmol g−1 h−1 in the absence of a sacrificial agent. Xia et al. adopted a similar strategy, initially preparing COF via Schiff base condensation between TG and DHPA.106 The synthesized COF was then mixed with bismuth nitrate, allowing Bi ions to adsorb onto the COF. These ions were subsequently reduced to Bi2O3 nanoparticles, which were strongly anchored to the COF via robust interfacial interactions. The resulting heterojunction displayed increased porosity and surface area, which facilitated mass transport and reactant diffusion. This design yielded a 5.6-fold enhancement in H2O2 production compared that with of pure Bi2O3, which was attributed to the improved separation and transfer of photogenerated charge pairs. Hence, the in situ growth of COF-based S-scheme heterojunctions offers effective control over morphology, promotes band alignment between coupled semiconductors, and enhances the separation and transport of electron–hole pairs, collectively boosting photocatalytic performance. This technique enhances charge separation, minimizes electron–hole recombination, and improves overall material stability.11 The strong interfacial bonding promotes effective reactant diffusion and increased catalytic performance.115 Furthermore, the uniform porosity contributes to better adsorption capacity and selectivity for organic pollutants.118 Enhanced conductivity and reduced interfacial resistance further improve long-term stability and ion transport efficiency.116 Additionally, the robust bonding between COFs and 2D materials offers excellent resistance to corrosion. However, controlling the morphology during synthesis can be difficult, sometimes leading to structural imperfections.105
Other synthesis methods
The sonochemical synthesis process has gained attention in photocatalyst fabrication owing to its advantages such as low power consumption, rapid reaction rates, and cost-effectiveness. The generation and collapse of acoustic cavitation bubbles create localized hot spots, leading to extreme temperatures (>5000 K) and pressures (>1000 atm) within short durations.119 These dynamic conditions enable rapid heating and cooling cycles, promoting COF-based S-scheme photocatalyst crystallization.120 This process enhances crystallinity while optimizing properties such as size, morphology, and defect structure, which are vital for photocatalytic efficiency. Sonochemically prepared S-scheme photocatalysts feature large surface areas, which increase the number of active sites for photocatalytic reactions.121 This also improves mass transport and reactant diffusion, ensuring effective interactions with active sites. This method achieves high reaction rates, as seen in its short synthesis duration (∼60 min), making it efficient and scalable.28 Moreover, it enables the formation of heterostructures and S-scheme interfaces with improved light absorption and charge separation, enhancing sunlight-driven photocatalysis.42 Tailoring parameters such as ultrasonic power, frequency, solvent, and precursors offers precise control over structural and functional properties, making the sonochemical method versatile and valuable for synthesis of advanced materials.60For example, Zhang et al. utilized a sonochemical method to integrate BiOBr nanosheets onto TpPaCl2, forming an S-scheme TpPaCl2/BiOBr system for the photocatalytic generation of H2O2.94 The authors initially synthesized TpPaCl2via Schiff-base polymerization of Tp and PaCl2. Subsequently, TpPaCl2 was mixed with KBr and bismuth nitrate, which served as BiOBr precursors, in a water–glycerol mixture under sonication for 30 min to fabricate the TpPaCl2/BiOBr heterojunction. Microstructural analysis revealed that the TpPaCl2 microspheres were partially covered with BiOBr nanoparticles, resulting in an increased surface area and porosity. These structural improvements facilitated the formation of additional active sites, enhanced mass transport, and reduced charge recombination rate. The band position alignment between the two materials established an S-scheme heterojunction, achieving a remarkable H2O2 generation rate of 3749 μmol g−1 h−1 under simulated sunlight, which is 27 times higher than that of bare BiOBr. Thus, the sonochemical process effectively enhances the surface area and porosity, highlighting its potential for the industrial-scale synthesis of COF-based S-scheme heterojunctions. Thermal annealing involves heating the constituent precursors to high temperatures, which enables the formation of specific microstructures with enhanced crystallinity, composition, and interface properties. This process increases surface area and improves charge separation, thereby supporting photocatalytic activity.68 For example, Niu et al. employed thermal annealing to develop an S-scheme CuWO4/COF heterojunction with improved photocatalytic performance for CO2 reduction under visible light.34 The COF was fabricated by dispersing specific amounts of TMT and TFPT in a mixture of 1-butanol and o-dichlorobenzene, followed by a solvothermal reaction at 120 °C for 72 hours. CuWO4 was prepared by dissolving copper nitrate and sodium tungstate dihydrate in water, followed by co-precipitation and calcination. To construct the heterojunction, the required amount of COF dissolved in methanol was ultrasonically mixed with CuWO4 and heated at 80 °C, followed by calcination at 250 °C for 1 h. Microstructural analysis showed that CuWO4 nanoparticles were distributed along the COF nanofibers, resulting in an improved surface area, extended light harvesting, and promoted electron–hole separation. This led to a CO generation level of 7.17 μmol g−1 h−1, highly surpassing that of pure COF (1.6 μmol g−1 h−1). Despite its potential, thermal annealing remains underexplored for the synthesis of COF-based S-scheme heterojunctions.
Wet chemical synthesis is a highly versatile method that provides precise control over the crystallinity, particle size, and morphology of COF-based S-scheme heterojunctions by carefully selecting precursors, shape-controlling agents, and reducing agents.122 Surfactants play a crucial role by modulating the surface energy, driving crystallization, and promoting stable crystal structures, which enhance charge separation and migration.123
This approach also offers scalability and cost-effectiveness, operating under mild conditions while enabling uniform COF deposition on substrates for improved photocatalytic performance.124 However, challenges include maintaining stoichiometry and homogeneity and the potential for impurities from additives while ensuring consistency.125 Despite these limitations, wet chemical synthesis remains a promising strategy, with future efforts needed to optimize reaction conditions, develop sustainable surfactants, and improve scalability for industrial applications. For instance, Zhang et al. employed a wet chemistry technique to anchor MgIn2S4 nanosheets onto a hollow COF, forming an S-scheme heterojunction for visible-light-activated photocatalytic H2O2 generation.64 The authors initially synthesized COF via Schiff-based polymerization and crystallization between TPT-CHO and TAPB, followed by the application of wet chemistry to grow MgIn2S4 nanosheets on the COF surface. Microstructure analysis revealed robust interfacial interactions between the MgIn2S4 nanosheets and COF, leading to an increased surface area, widened light harvesting, and enhanced mass transport to favor the photocatalytic reaction. As a result, the optimized MgIn2S4/COF heterojunction achieved a remarkable H2O2 generation level of 4.52 mmol g−1 h−1, which was 9.4 fold superior to that of bare COF, highlighting the effectiveness of wet chemistry synthesis in developing heterojunctions with superior photocatalytic properties. However, material aggregation during synthesis reduces the surface area and limits the carrier transfer rates, partially compromising the photocatalytic performance.39 However, the method remains cost-effective and suitable for industrial applications.
Various kinds of COF-based S-scheme heterojunctions
The porosity and high surface area of COFs play a pivotal role in the prevention of semiconductor aggregation when coupled with suitable semiconductors. This attribute ensures the accessibility of a larger density of photocatalytic reactive sites.28 Moreover, the framework and porous structure of COFs are instrumental in reducing carrier transport distances when combined with other semiconductors, thereby accelerating the transfer of photoinduced carriers.30 Importantly, compared to traditional heterojunctions like type II or Z-scheme configurations, the S-scheme heterojunctions exhibit a notable extension in the photoinduced carrier lifetime while significantly decreasing the resistance of the composite material. This results in an enhanced redox capability, underlining the superior performance of these composites.40 In the context of classification, COF-based S-scheme heterojunctions can be categorized by their heterojunction structure or COF modification strategies. From a structural perspective, these heterojunctions are divided into single-and dual S-scheme heterojunctions. However, from the standpoint of modification strategies, the focus shifts to COF–COF heterojunctions, MOF–COF heterojunctions, and S-scheme heterojunctions involving co-catalysts coupled with Schottky junctions. Although these methodologies have gained recognition, they remain at various stages of development, with ongoing research aimed at addressing challenges and fostering innovation.Structural level
Single S-scheme heterojunctions
At the structural level, the integration of semiconductor materials with COFs to construct single S-scheme heterojunctions has gained significant attention. The selection of materials in this context is influenced by differences in the Ef levels, CB, and VB of the involved components. Materials with higher Ef levels and more negative CB values are classified as RP, whereas those with lower Ef levels and more positive VB values are categorized as OP.45,58 While earlier studies primarily emphasized on n-type semiconductors for this construction, recent advancements have expanded the scope to include p-type semiconductors, broadening the versatility of the S-scheme heterojunction framework, as shown in Fig. 6.Zhao et al. employed an in situ encapsulation method to construct an S-scheme W18O49@COF heterojunction, achieving enhanced photocatalytic performance for aerobic oxidation under visible light.35 SEM analysis (Fig. 7a) confirmed the effective wrapping of W18O49 nanobundles with TpPa-H COF through robust interfacial interactions, resulting in a high surface area and abundant pore channels that facilitated mass transport and suppressed recombination of photoinduced electron–hole pairs. This was further validated by photoluminescence (PL) and electrochemical impedance spectroscopy (EIS) Nyquist studies (Fig. 7b and c), which demonstrated the lowest PL intensity and smallest EIS Nyquist arc radius for the W18O49/TpPa-H heterojunction compared to its individual components. Additionally, the absorption spectra corroborated the effectiveness of the S-scheme heterojunction by exhibiting enhanced light absorption, which provided abundant charges for supporting photocatalytic reactions. Consequently, the W18O49/TpPa-H photocatalyst achieved 100% benzylamine conversion within 4 h, which is approximately three times higher than that of pure TpPa-H. This improvement was ascribed to the smaller work function of TpPa-H (5.62 eV) than that of W18O49 (8.05 eV), which induced spontaneous electron migration from W18O49 to TpPa-H upon integration. This migration results in band bending and the generation of interfacial BIEF, facilitating charge separation and migration. Under light irradiation, the photoinduced electrons in the CB of W18O49 recombined with holes in the VB of TpPa-H, effectively separating the high-potential electrons in the CB of TpPa-H and the holes in the VB of W18O49 to drive the aerobic oxidation reaction (Fig. 7d).
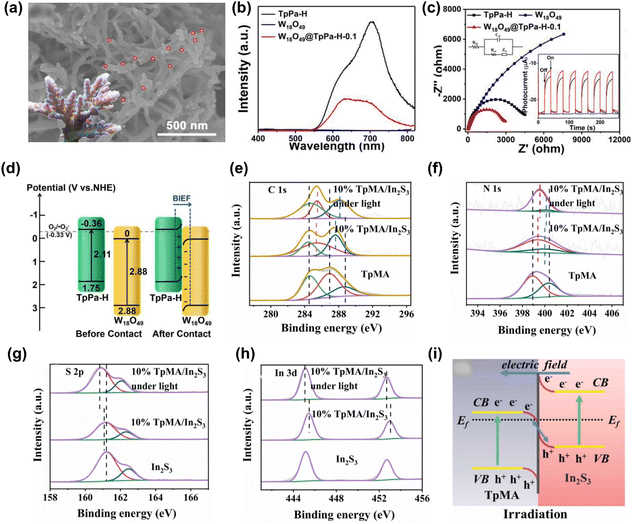 | ||
| Fig. 7 (a) SEM image, (b) PL spectra, (c) EIS Nyquist plots, and (d) charge transfer mechanism of W18O49@COF. Reproduced with permission from ref. 35, copyright 2024, Elsevier. XPS analysis (e) C 1s, (f) N 1s, (g) S 2p, (h) In 3d, and (i) charge-transfer mechanism of TpMa-H/In2S3. Reproduced with permission from ref. 84, copyright 2024, Elsevier. | ||
Similarly, Chen et al. developed a single S-scheme heterojunction by integrating In2S3 nanosheets with TpMa nanotubes to enhance photocatalytic performance for H2O2 production.84 TEM analysis confirmed the successful integration of In2S3 nanosheets with TpMa nanotubes via strong interfacial interactions. The absorption spectra revealed that the generation of the TpMa-H/In2S3 system significantly broadened light harvesting, increasing the excitation and availability of photoinduced charge carriers. The improved separation and migration of these charges were validated by the lowest EIS Nyquist arc radius observed for the optimized TpMa-H/In2S3 heterojunction compared with its counterparts. Consequently, the TpMa-H/In2S3 heterojunction exhibited a maximum H2O2 generation level of 311.07 μmol L−1. The development of an S-scheme system between TpMa-H and In2S3 was further confirmed by in situ XPS analysis. As shown in Fig. 7e–h, the binding energies of the TpMa-H orbitals exhibited a negative shift, whereas those of In2S3 showed a positive shift after heterojunction formation, indicating electron migration from In2S3 to TpMa-H. Under light irradiation, the binding energies of the component orbitals reversed their shifts in contrast to the dark state. This indicates that the photogenerated electrons and holes in the CB of TpMa-H and holes in the VB of In2S3 recombine, enhancing the separation of electrons in the CB of TpMa-H and holes in the VB of In2S3. These separated charge carriers facilitate the redox reactions necessary for H2O2 production (Fig. 7i).
Qi et al. reported improved photocatalytic performance for SMT degradation by forming an S-scheme heterosystem between COF and g-C3N4 using an in situ polymerization process.87 Microstructural analysis demonstrated that the COF nanosheets were intimately coated onto the g-C3N4 substrate via strong π–π interactions, resulting in an enhanced surface area with abundant charge transport channels and numerous catalytic sites to support photoreactions. The formation of the heterojunction also promoted charge carrier separation and migration, as verified by the diminished PL intensity and smallest EIS Nyquist arc diameter. Consequently, the optimized COF/g-C3N4 photocatalyst exhibited a 6.6-fold improvement in SMT degradation performance compared to pure COF. Upon light irradiation, the photogenerated electrons in the CB of g-C3N4 recombined and perished with holes in the VB of COF, enhancing the separation of electrons in the CB of COF and holes in the VB of g-C3N4. This process generates superoxide and hydroxyl radicals, which actively degrade SMT. Although these studies achieved significant performance improvements, further analysis is required to establish the recyclability and scalability of these materials in various operational environments, such as real wastewater matrices or continuous flow reactors. In addition, the development of COF-based S-scheme heterojunctions introduces a new dimension of π–π interactions, which warrants a deeper investigation into how these interactions influence the electronic band structures and active site accessibility. Future research should also address the eco-friendliness and cost-effectiveness of synthetic processes to align these advancements with sustainable development goals and ensure practical viability beyond laboratory conditions. Integrating computational studies, such as DFT simulations, can further direct the formation of S-scheme heterojunctions by predicting the optimal material combinations and their electronic properties for specific applications.
Dual S-scheme heterojunctions
The formation of single S-scheme heterojunctions involving COFs as one of the components has demonstrated a significant improvement in photocatalytic efficiency by enhancing the separation of high-potential photoinduced charge carriers.48 However, their photocatalytic efficiency remains insufficient to meet the demands of large-scale applications. Recently, the concept of dual S-scheme heterojunctions has garnered considerable attention owing to their superior ability to facilitate charge-carrier separation on a larger scale. This enhancement is attributed to the increased driving force provided by the synergistic effects of two BIEFs formed at the interfaces of the two junctions.36 The formation of dual S-scheme heterojunctions requires specific prerequisites, including proper energy band alignment, appropriate band edge positions, and a sufficient dissimilarity in Ef levels between the component materials.29 These factors collectively ensure the effective formation of dual S-scheme heterojunctions, resulting in optimal charge transfer and separation. For researchers interested in the principles governing charge transfer and separation, the conditions necessary for heterojunction formation, and the precise roles of band edge positions and Ef level differences, we recommend our recently published review paper on dual S-scheme heterojunction photocatalysts. This paper provides an in-depth exploration of all aspects related to these dual S-scheme heterojunctions.94 In this section, we specifically emphasize on the enhanced performance exhibited by COF-based dual S-scheme heterojunctions, emphasizing their potential for advancing photocatalytic applications. For instance, Fan et al. transitioned the S-scheme CdS@COF core–shell heterojunction, which exhibited poor photocatalytic performance, into a high-performance dual S-scheme CdS/BiVO4@COF heterojunction by introducing BiVO4 at the interface of CdS@COF.109 Notably, the role of the COF shell shifted from an OP to an RP in the dual S-scheme heterojunction because of the significant matching of the band structures and the Ef level differences. The authors first anchored BiVO4 nanoparticles onto the exterior of CdS, which was subsequently enveloped by a COF shell. The resulting heterostructure exhibited an enhanced surface area due to the combined effect of the three components, thereby extending light absorption, improving mass transport, and promoting electron–hole separation. The CdS/BiVO4@COF photocatalyst achieved the maximum syngas and O2 yields of 453.6 and 230.5 μmol g−1 h−1, respectively. These improvements were ascribed to the increased separation of high-potential charges driven by the generation of two BIEFs at the interfaces. The enhanced charge separation was validated by the highest current density and lowest PL intensity of CdS/BiVO4@COF compared to those of CdS@COF and pure materials. KPFM analysis was employed to authenticate the S-scheme charge-migration mechanism, and the results are presented in Fig. 8. For the CdS@COF heterojunction, a high surface potential difference of 60.6 mV between CdS and COF indicated the formation of an interfacial BIEF owing to the charge redistribution process in the dark. Under light irradiation, the surface potential of CdS decreased, whereas that of COF increased, lowering the potential difference between CdS and COF to 49.42 mV, indicating the transfer of photogenerated electrons from the COF to CdS. In contrast, the CdS/BiVO4@COF heterojunction demonstrated a higher potential in the outermost region and a reduced potential in the middle region, suggesting the flow of free carriers from CdS and COF to BiVO4. Additionally, in contrast with CdS@COF, the potential difference of CdS/BiVO4@COF in the dark increased to 80.15 mV, which then decreased to 36.9 mV upon light exposure. This significant potential difference under dark and light conditions confirmed that the photogenerated electrons in the CB of CdS and COF perished with photoinduced holes in the VB of BiVO4, thereby confirming the dual S-scheme charge migration mechanism in CdS/BiVO4@COF.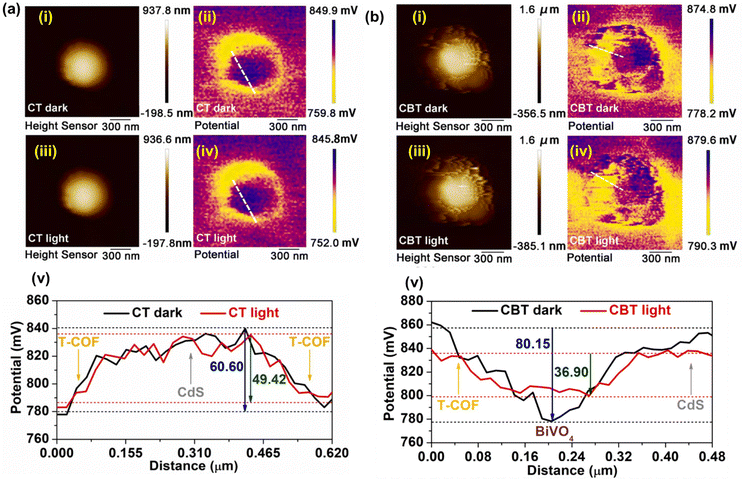 | ||
| Fig. 8 KPFM analyses of (a) CdS/COF and (b) CdS/BiVO4@COF: AFM studies (left) and KPFM analyses (right) under dark and light irradiation; (bottom) line-scanning surface potential curves under dark and light irradiation. Reproduced with permission from ref. 109, copyright 2024, Elsevier. | ||
Similarly, Lu et al. demonstrated that the formation of a dual S-scheme α-Fe2O3/g-C3N4/COF heterojunction effectively enhanced the visible-light-driven photoactivity for TC degradation.115 Microstructural analysis revealed that the net structure of the COF, featuring wide pore routes, was effectively integrated with lamellar-shaped g-C3N4 and hexagonal α-Fe2O3. This integration facilitates a large surface area and high porosity, promoting the dispersion of active sites, offering additional carrier transport channels, and reducing the charge diffusion distance, thereby impeding the recombination of photogenerated charge pairs. Furthermore, the unique heterostructure broadens light harvesting, generating abundant charges for the redox processes. Consequently, the TC degradation rate reached 0.00561 min−1, which was 2.5 and 2 fold superior to those of the COF and α-Fe2O3/g-C3N4, respectively. Additionally, the optimal candidate exhibited robust stability over five continuous cycles without structural degradation, highlighting its potential for large-scale applications. Upon light excitation, the photogenerated electrons in the CB of α-Fe2O3 recombine with holes in the VB of g-C3N4 and COF, enhancing the separation of electrons in the CBs of g-C3N4 and COF and holes in the VB of α-Fe2O3. This process generates superoxide and hydroxyl radicals, which effectively degrade the TC. Despite the remarkable ability of COF-based dual S-scheme heterojunctions to enhance the photocatalytic performance, few studies have explored this strategy. However, given the exceptional properties of COFs and the charge separation potential of dual S-scheme heterojunctions, this approach is anticipated to witness a significant surge in research interest in the near future.
COF level
COF–MOF-based S-scheme heterojunctions
The effective preparation of porous MOFs enhances the surface area, offering abundant active sites and providing shortened charge transport channels, which collectively increase the adsorption of reactants, elevate reaction activity, and accelerate the separation and transfer of photogenerated electron–hole pairs.45 These features significantly improve the photocatalytic efficiency by addressing key restrictions like recombination of electron–hole pairs and limited interaction between the catalyst and reactants.60 Furthermore, the nanostructure and composition of MOFs can be modulated to achieve extended light absorption, a critical factor in exciting and generating sufficient charge carriers to facilitate redox reactions under various light conditions.11 This structural tunability, combined with their versatile properties, has made MOFs a prominent subject of attention in the domain of photocatalysis. In addition to these advantages, the development of a COF-MOF S-scheme system synergistically integrates the strengths of both materials. This unique heterojunction not only enhances the surface area and extends light harvesting capacity but also significantly improves charge separation efficiency, which is vital for sustaining high photocatalytic activity.75 The spatially directed charge transfer within the S-scheme heterojunction ensures effective exploitation of the generated charges, thereby boosting photocatalytic reactions.39 As an example, the MOF-808-NH2 was coupled with TpTt-COF through a hydrothermal method to form an S-scheme heterojunction for photocatalytic AP removal under visible light.83 Microstructural analysis revealed a core–shell structure, where rod-like TpTt-COF grew onto the surface of rope-like MOF-808-NH2, increasing the surface area to 54.95 m2 g−1 and creating wide pore channels to enhance mass transport and light absorption. The formation of C–N bonds facilitates charge transport, improving the separation and transport of photoinduced charge pairs. Consequently, the optimal MOF-808-NH2/TpTt-COF heterojunction achieved an AP removal rate of 98% within 2 h with negligible performance loss and structural degradation after four recycling experiments, confirming the potential of S-scheme heterojunctions. Niu et al. employed a solvothermal process to develop S-scheme heterojunctions by integrating COF nanofibers with MOF octahedra in varying molar ratios for photocatalytic CO2 reduction under visible light.81 SEM analysis (Fig. 9a) demonstrated that MOF was effectively embedded onto COF with strong interfacial interactions, enhancing carrier separation and transport. The higher surface area of the COF/MOF heterojunction (900 m2 g−1) compared to that of the individual components facilitated CO2 adsorption and activation, provided abundant active sites, and shortened the charge transport distance. This was validated by the diminished PL intensity and the smallest EIS Nyquist arc diameter of the optimized heterojunction, as shown in Fig. 9b and c. TAS analysis (Fig. 9d and e) further confirmed prolonged charge carrier lifetimes, with the average relaxation lifespan of the heterojunction (242.1 ps) significantly exceeding that of the MOF (119 ps) within the 550–800 nm wavelength range. Consequently, the optimized COF/MOF heterojunction achieved a CO generation rate of 6.56 μmol g−1 h−1, 5- and 4.4-fold superior to those of the MOF and COF, respectively. XPS spectra provided evidence of the S-scheme charge transport route. As Fig. 9f–i show, the binding energies of N 1s and C 1s in COF exhibited a red shift within the heterojunction under dark conditions, whereas Zr 3d and O 1s in MOF showed a blue shift, indicating electron migration from COF to MOF. Under light irradiation, the binding energies of the constituent orbitals were reversed, verifying photoinduced electron migration from the MOF to COF. This separation of electrons in the CB of the COF and holes in the VB of the MOF enabled CO2 and H2O to react, generating CO, H+, and O2. Similarly, Hu et al. utilized ultrasonic treatment to integrate H2N-Cu-MOF nanosheets with flower-like TpPa-1-COF, developing an S-scheme heterosystem to enhance H2 generation performance.67 The resulting nanostructure improved the surface area, widened light harvesting, and reduced charge diffusion distances, promoting efficient charge separation and migration. The optimized H2N-Cu-MOF/TpPa-1-COF photocatalyst exhibited a hydrogen generation efficiency of 4.19 mmol g−1 h−1, surpassing TpPa-1-COF and H2N-Cu-MOF by factors of 22.1 and 46.5, respectively. Stability analysis over four cycles revealed a negligible performance loss, confirming the robustness of the photocatalyst. ISIXPS analysis verified the S-scheme charge migration route, where electrons in the CB of TpPa-1-COF reduced H+ to H2 and holes in the VB of H2N-Cu-MOF were scavenged by ascorbic acid. While these studies show that the S-scheme heterojunction effectively facilitates spatial charge separation, there is scope to explore the impact of external factors, such as solution pH, temperature, and competing ions, on the stability and robustness of these heterojunctions. Moreover, leveraging in situ techniques, including XPS, fs-TAS, and KPFM, could offer additional insights into the transient behavior of charge carriers and their interaction with reactants during the photocatalytic process. Overall, while these studies exemplify the potential of MOF/COF S-scheme heterojunctions, future research should prioritize scalable synthesis methods, enhanced mechanistic understanding, and long-term stability in complex environments to transition these materials to industrial-scale photocatalytic applications.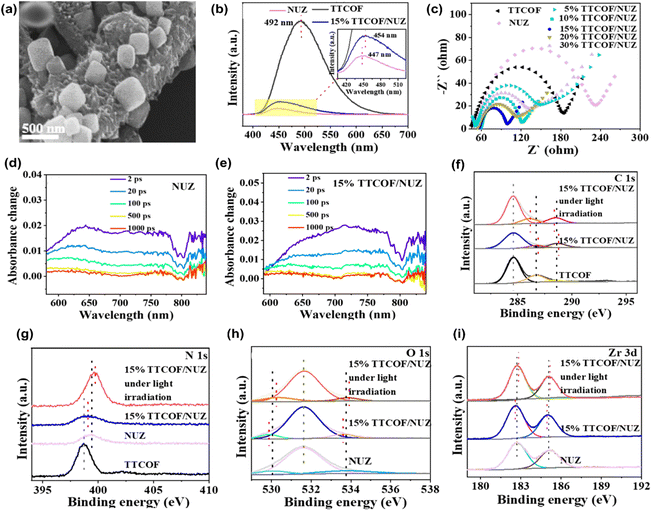 | ||
| Fig. 9 (a) SEM image, (b) PL spectra, (c) EIS Nyquist plots, (d and e) TAS analysis, ex situ/in situ XPS analysis, (f) C 1s, (g) N 1s, (h) O 1s, and (i) Zr 3d of COF/MOF. Reproduced with permission from ref. 81, copyright 2022, American Chemical Society. | ||
COF–COF-based S-scheme heterojunctions
This novel approach leverages the distinctive features of individual COFs, including their tunable porosity, high crystallinity, and large surface areas, while overcoming their intrinsic limitations, such as inefficient charge separation and low light absorption.37 By grafting one COF onto another, the resulting heterojunctions not only optimize charge carrier dynamics but also enhance light harvesting capabilities through synergistic interactions.27 Furthermore, the integration ensures more efficient utilization of the visible light and improves the redox potential of the photocatalyst.42 These atomic-level charge transfer pathways, facilitated by the strong interfacial coupling between COF units, reduce the recombination rates of photogenerated carriers, and prolong their lifetimes.61 This, in turn, significantly boosts the efficiency of photocatalytic reactions, particularly for applications such as hydrogen production, CO2 reduction, organic transformation, and pollutant removal.For example, Luan et al. employed an in situ process to decorate Pt nanoparticles and COF-316 nanosheets into TbBpy-COF nanosheets, thereby fabricating an S-scheme COF-316/Pt@TbBpy-COF heterojunction for evaluating the efficiency of overall water splitting under visible light.68 TEM analysis (Fig. 10a) confirmed the intimate integration of COF-316 nanosheets with nanoribbons of TbBpy-COF, along with the effective decoration of Pt particles within the pores of COF. The formation of the COF-316/Pt@TbBpy-COF heterostructure via π–π interactions effectively enhanced the surface area, promoting the availability of catalytic centers and facilitating catalyst–light interactions to generate abundant charge carriers. Moreover, COF-316/Pt@TbBpy-COF exhibited the highest current density and the lowest PL intensity compared to its counterparts, verifying that the heterojunction significantly increased the separation and migration of photoinduced electron–hole pairs. Consequently, this heterostructure acquired a maximum H2 generation of 220.4 μmol g−1 h−1 with no performance loss over five consecutive recycling experiments, and an AQY of 5.75% at 500 nm. EPR analysis further verified the S-scheme charge-separation route. As shown in Fig. 10b, the DMPO-˙OH signal for COF-316/TbBpy-COF was considerably larger than those for pure COF-316 and TbBpy-COF, confirming the accumulation of photogenerated charge pairs with a strong redox capacity, a hallmark of the S-scheme heterojunction. Under light irradiation, the photogenerated electrons and holes in the CB of COF-316 and the VB of TbBpy-COF recombined, enhancing the separation of electrons and holes in the CB of TbBpy-COF and the VB of COF-316, respectively. These separated charge carriers subsequently drive water splitting, producing H2 and O2.
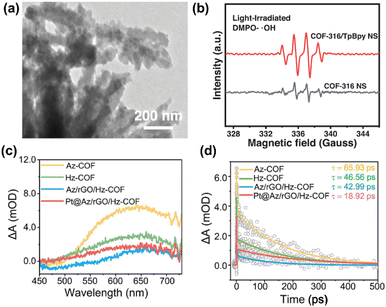 | ||
| Fig. 10 (a) TEM image and (b) DMPO-˙OH spectra of COF-316/Pt@TbBpy-COF. Reproduced with permission from ref. 68, copyright 2024, Wiley. (c) TAS analysis, and (d) TRPL analysis of Az-COF/rGO/Hz-COF. Reproduced with permission from ref. 82, copyright 2024, American Chemical Society. | ||
Niu et al. synthesized an S-scheme heterostructured system by integrating Az-COF and Hz-COF using a solvothermal process, with rGO serving as a mediator for overall water splitting.82 UPS analysis revealed that the work function of Az-COF (3.72 eV) was inferior to that of Hz-COF (4.39 eV), representing charge migration from Az-COF to Hz-COF upon integration. This migration created an interfacial IEF, which served as a dynamic force for the improved separation of photogenerated electrons in Az-COF and holes in Hz-COF under light irradiation. TAS analysis (Fig. 10c) verified that the intensity of the excited absorption peaks in the Az-COF/rGO/Hz-COF heterojunction was higher than that in the pure COFs, signifying the enhanced separation of photoinduced electron–hole pairs due to the S-scheme heterojunction. TRPL analysis (Fig. 10d) further supported this observation, showing the shortest charge recombination lifetime for Az-COF/rGO/Hz-COF, indicating accelerated charge separation and migration across the interface. The Az-COF/rGO/Hz-COF photocatalyst achieved the highest H2 and O2 levels of 120 and 58 μmol g−1 h−1, respectively. Hence, the modularity and structural diversity of COFs provide further opportunities to fine-tune the electronic properties of these heterojunctions, enabling precise control of their photocatalytic behavior. These studies revealed that controlling the interfacial coupling between COFs is essential for optimizing charge–transfer pathways, as challenges such as mismatched energy levels and insufficient contact can hinder the formation of efficient heterojunctions. Advanced synthesis techniques, including template growth and molecular pre-design, can overcome these limitations and enhance photocatalytic performance. Notably, these photocatalysts highlight their potential; however, scalability remains a concern because of the challenges of maintaining crystallinity and structural integrity in large-scale synthesis methods, such as in situ decoration and solvothermal processes. Furthermore, integrating computational studies such as DFT calculations with experimental analyses such as EPR, UPS, TAS, and TRPL could provide deeper insights into interfacial charge dynamics and band alignment, offering a direction for the rational formation of next-generation COF-based heterojunctions.
COF-QD-based S-scheme heterojunctions
Quantum dots (QDs) exhibit several matchless characteristics that render them highly appropriate for enhancing the photocatalytic performance, especially when integrated into COF-based S-scheme heterojunction photocatalysts. Their ability to absorb light in the visible region allows for efficient utilization of solar energy, which is a crucial factor in promoting photocatalytic reactions.126 Furthermore, the quantum confinement effect in QDs facilitates multiple exciton generation, enabling the emission of up to three electrons per absorbed photon, which is a significant improvement compared to the single-electron emission typical of bulk semiconductors.127 This characteristic is particularly advantageous for boosting the charge carrier density in photocatalytic systems. Additionally, QDs possess excellent charge separation and transport capabilities because of their nanoscale dimensions and large intrinsic dipole moments, which align well with the charge transport pathways in S-scheme heterojunctions.128 The high exciton coefficient of QDs in the visible region further enhances their light absorption efficiency, ensuring effective solar spectrum utilization.129 Moreover, their luminescence properties contribute to reducing charge carrier recombination, thereby improving photocatalytic efficiency.33 When integrated with COF semiconductors, QDs extend the visible-light harvesting extent of the resulting heterojunction, ensuring complementary light harvesting.130 These synergistic properties make QDs an exceptional component in COF-based S-scheme heterojunction photocatalysts, where efficient charge separation, enhanced light absorption, and optimized charge transport are vital for driving advanced photocatalytic applications.For instance, Zhao et al. used a self-assembly process to integrate a flower-like COF with ZnSe QDs, thereby developing an S-scheme heterojunction for photocatalytic H2O2 production.58 Microstructure analysis confirmed the uniform decoration of ZnSe QDs onto the flower-like COF, with a robust interfacial interaction. The absorption spectra revealed that the absorption shoulder of the COF extended from 690 nm to 810 nm upon integration with ZnSe QDs (Fig. 11a), resulting in increased excitation and the creation of more photoinduced electron–hole pairs. Furthermore, TPRL analysis indicated that the average lifetime of ZnSe/COF was shorter than those of bare COF and ZnSe, verifying the reduced recombination of high-potential photoinduced charge carriers. This was also supported by the smallest EIS Nyquist arc radius of ZnSe/COF compared with its competitors. The optimized ZnSe/COF exhibited the highest H2O2 generation level (1895 μmol g−1 h−1) under light irradiation. XPS analysis not only confirmed the formation of the heterojunction but also authenticated the S-scheme charge transfer pathway between the COF and ZnSe. As Fig. 11b–e show, without light irradiation, the binding energies of COF orbitals in ZnSe/COF exhibited a negative shift compared to those of pure COF orbitals, while the binding energies of ZnSe in ZnSe/COF were higher than those of pure ZnSe orbitals. This variation in the binding energy confirmed the transfer of free carriers from ZnSe to COF upon the creation of the heterojunction. Conversely, upon light exposure, the binding energies of the constituent orbitals of both materials within the heterojunction exhibited a reverse shift, validating the performance of photogenerated electrons in the LUMO of COF with holes in the VB of ZnSe, which is consistent with the S-scheme mechanism (Fig. 11f). The separated electrons in the CB of ZnSe reduced O2 and H+ to generate H2O2, whereas holes in the HOMO of COF were oxidized through ethanol.
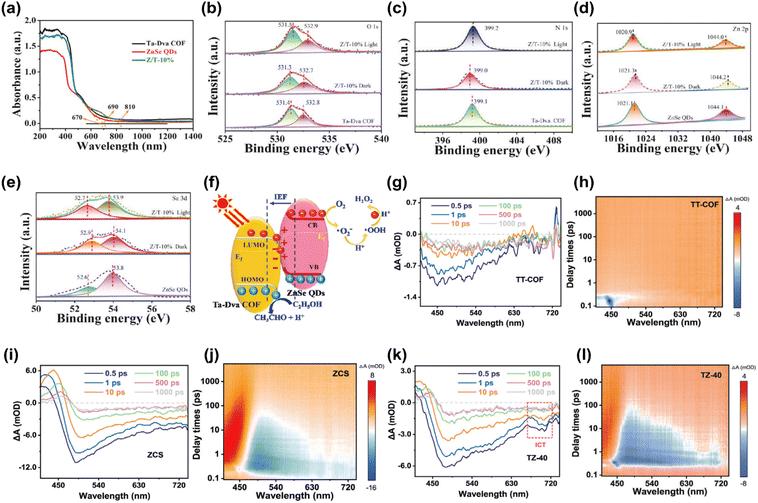 | ||
| Fig. 11 (a) Light absorption spectra, ex situ/in situ XPS analysis (b) O 1s, (c) N 1s, (d) Zn 2p, (e) Se 3d, and (f) charge transfer mechanism of ZnSe/COF. Reproduced with permission from ref. 58. Copyright 2024, Elsevier. TAS analysis of (g and h) COF, (i and j) ZnCdS, and (k and l) COF/ZnCdS. Reproduced with permission from ref. 97, copyright 2024, Wiley. | ||
The presence of high-energy surface atoms in QDs can lead to particle agglomeration during photocatalytic reactions, reducing the surface area and charge transport channels. However, the high surface area and plentiful active sites of COF can effectively reduce QD agglomeration. For example, Ma et al. reported that the creation of an S-scheme heterojunction between ZnCdS QDs and COF effectively mitigated agglomeration and promoted the uniform dispersion of ZnCdS QDs.97 This resulted in a substantial enhancement in the surface area of COF/ZnCdS, providing abundant active sites and charge transport channels, which reduced recombination and increased the generation of charge carriers, as evidenced by the maximized current density and extended light absorption. Consequently, the optimized COF/ZnCdS exhibited the highest H2O2 generation efficiency of 5171 μmol g−1 h−1 under visible light, which is double that of the pure COF. The enhanced performance was ascribed to the retention of high-potential photoinduced charge pairs without perishing owing to the formation of an S-scheme heterojunction between ZnCdS and COF. The S-scheme charge-transfer pathway was verified using fs-TAS analysis (Fig. 11g–l). Under light irradiation, pure COF showed maximum absorption at 480 nm, whereas pure ZnCdS exhibited absorption peaks at 430 and 485 nm. Compared with a single exciton, the second exciton showed a red shift for ZnCdS, representing a decrease in the energy required to introduce the second exciton. Similarly, the COF/ZnCdS composite exhibited two absorption peaks at 420 and 495 nm, showing a red shift in the second exciton aligned with ZnCdS. However, TAS analysis of COF/ZnCdS showed an additional negative absorption at 700 nm, attributed to the migration of electrons from ZnCdS to COF, aligned with the S-scheme carrier transport route. Similarly, He et al. anchored CsPbBr3 QDs onto TPA-COF to construct an S-scheme heterojunction, leading to enhanced photocatalytic efficiency for CO2 reduction.116 Microstructure analysis confirmed the uniform dispersion of CsPbBr3 QDs onto the TPA-COF surface, leading to extended light harvesting and improved electron–hole separation, thus supporting the photocatalytic reaction. The optimized CsPbBr3/TPA-COF exhibited maximum CH4 and CO generation rates of 13.7 and 41.2 μmol g−1 h−1, respectively. fs-TAS analysis confirmed the S-scheme charge migration pathway. Compared with pure materials, the formation of an additional signal and the shortening of lifetimes (τ1 and τ2) in TPA-COF/CsPbBr3 relative to pure TPA-COF and CsPbBr3 verified the formation of new charge transport channels and the migration of charge from TPA-COF to CsPbBr3, aligned with the S-scheme route. These results emphasize the potential of COF-based materials for overcoming the intrinsic limitations of QD photocatalysts. However, to fully exploit the capabilities of S-scheme heterojunctions, further research should focus on elucidating the interplay between the structural stability, electronic interactions, and charge dynamics under realistic conditions. Such studies will pave the way for the rational design of next-generation photocatalysts with enhanced performance and durability.
Applications
COF-based S-scheme heterojunction photocatalysts have demonstrated significant potential for various applications. Table 1 lists several applications of these advanced materials. Although their current use primarily focuses on photocatalysis, their unique properties also suggest their potential in areas such as gas storage, chemical sensing, biomedicine, and beyond. COF-based S-scheme heterojunctions are particularly notable for photocatalytic applications, such as hydrogen generation, CO2 conversion, degradation of environmental pollutants, and organic transformation, as shown in Fig. 12.| Catalyst | Synthesis method | Light source/λ (nm) | Application | Performance | Ref. |
|---|---|---|---|---|---|
| COF/CdS | Self-assembly | 300 W Xe/>420 | H2 generation | 8.67 mmol h−1 g−1 | 41 |
| COF/ZnIn2S4 | Condensation reaction | 300 W Xe/>400 | 0.70 mmol h−1 g−1 | 42 | |
| COFs-Ph@CdS | Hydrothermal | 300 W Xe | 0.73 mmol h−1 g−1 | 44 | |
| TpBD COF/ZnIn2S4 | Hydrothermal | 300 W Xe/>420 | 2.30 mmol h−1 g−1 | 48 | |
| ZnIn2S4/TpPa-1COF | Hydrothermal | 300 W Xe/>420 | 2.75 mmol h−1 g−1 | 104 | |
| WO3/TpPa-1COF | Hydrothermal | 300 W Xe/>420 | 19.89 mmol h−1 g−1 | 86 | |
| BiOBr/COF | Solvothermal | 300 W Xe/>420 | 7.79 mmol h−1 g−1 | 85 | |
| TiO2/COF | — | 300 W Xe/>420 | 15.33 mmol h−1 g−1 | 51 | |
| CdS/COF | Self-assembly | 300 W Xe/>420 | 15.1 mmol h−1 g−1 | 52 | |
| COF/g-C3N4 | Chemical approach | 300 W Xe/>420 | 1.15 mmol h−1 g−1 | 56 | |
| COF/PUP | In situ growth | 350 W Xe/>420 | 94.5 mmol h−1 g−1 | 57 | |
| BiOBr/TpBD COF | In situ transformation | 300 W Xe/>420 | 16.17 mmol h−1 g−1 | 62 | |
| COF/ZnxCd1−xS | Self-assembly | 300 W Xe/>400 | 8.41 mmol h−1 g−1 | 65 | |
| MOF/TpPa-1 COF | Ultrasonic treatment | 300 W Xe/320–780 | 4.19 mmol h−1 g−1 | 67 | |
| COF/CeO2 | In situ growth | 300 W Xe/>420 | CO2 reduction | CO: 66.2 μmol g−1 | 40 |
| g-C3N4/COF | Self-assembly | 300 W Xe/>400 | CO: 11.25 μmol h−1 | 46 | |
| TiO2/COF | In situ growth | 300 W Xe | CO: 7.51 μmol h−1 | 47 | |
| TiO2/COF | Self-assembly | 300 W Xe | CO: 11.6 μmol h−1 | 49 | |
| TiO2/COF | Co-precipitation | 300 W Xe | CO: 50.5 μmol h−1 g−1 | 59 | |
| COF/ZnIn2S4 | In situ growth | 300 W Xe/>420 | CO: 1117 μmol h−1 g−1 | 66 | |
| Zr-MOF/Pro-COF | In situ growth | 300 W Xe | CO: 106.35 μmol g−1 | 11 | |
| COF/MOF | Solvothermal | 300 W Xe/>420 | CO: 6.56 μmol g−1 h−1 | 81 | |
| ZnO/COF | Interfacial schiff-base reaction | 300 W Xe | CO: 11.07 μmol g−1; CH4: 4.86 μmol g−1 | 91 | |
| TiO2/BTTA COF | In situ growth | 300 W Xe/350–780 | H2O2 production | 0.74 mmol L−1 h−1 | 37 |
| COF/In2S3 | Hydrothermal | 300 W Xe/>420 | 0.31 mmol L−1 h−1 | 84 | |
| BiOBr/COF | In situ growth | 300 W Xe | 3.75 mmol g−1 h−1 | 94 | |
| ZnO/COF | Self-assembly | 300 W Xe | 2.44 mmol g−1 h−1 | 50 | |
| TpPa-1COF/BIVO4 | Solvothermal | LED/420 | 0.72 mmol g−1 h−1 | 55 | |
| ZnSe/COF | Self-assembly | 300 W Xe | 1.90 mmol g−1 h−1 | 58 | |
| MgIn2S4/COF | Wet chemistry | 300 W Xe/>420 | 4.52 mmol g−1h−1 | 64 | |
| COF/In2S3 | In situ growth | 360 W Xe | 5.71 mmol g−1 h−1 | 69 | |
| CdS/COF | Hydrothermal | 300 W Xe/>420 | 0.61 mmol L−1 h−1 | 80 | |
| ZnCdS/COF | — | 300 W Xe/>420 | 5.17.1 mmol g−1 h−1 | 97 | |
| Bi4O5Br2/COF | Hydrothermal | White LED/400–700 | 5.22 mmol g−1 h−1 | 89 | |
| SnS2/COF | Hydrothermal | 300 W Xe/420–800 | 1.47 mmol g−1 h−1 | 117 |
Hydrogen production
The environmental challenges posed by fossil fuels necessitate a shift toward renewable and eco-friendly energy sources. Among these, hydrogen is emerging as a promising energy carrier owing to its large energy density and clean-burning properties.41 Hydrogen, with an energy content of 120–142 MJ kg−1, is expected to dominate global energy generation by 2080, and current annual production exceeds 44.5 million tons.44 However, the majority of hydrogen is still derived from steam methane reforming, which accounts for approximately 95% of global production.42 To make hydrogen production greener and more sustainable, photocatalytic water splitting offers an efficient method for converting solar energy into hydrogen fuel.84 Since Fujishima and Honda's breakthrough in 1972, significant advancements have been made in photocatalytic hydrogen production.94 However, many semiconductor photocatalysts capable of water splitting exhibit wide bandgaps and are only activated under UV light, which represents a small fraction of the solar spectrum. This limitation emphasizes the need for visible-light-responsive photocatalysts that are efficient, stable, environmentally safe, and cost-effective for large-scale hydrogen production.50 To achieve efficient water splitting, the photocatalyst must satisfy several criteria. It must absorb light effectively to generate electron–hole pairs, minimize charge recombination, and prevent photocorrosion or toxic byproduct formation during the reaction. Additionally, the bandgap energy should ideally range between 1.23 eV and 3.0 eV to balance thermodynamic feasibility and visible-light activation.51 For overall water splitting, the CB must be more negative than the H+/H2 potential (0 V), while the VB must be more positive than the O2/H2O potential (1.23 V). These requirements ensure that the photocatalyst can drive the redox reactions necessary for splitting water into hydrogen and oxygen.56 COFs are particularly advantageous for this application because of their high surface area and porous frameworks that provide abundant active sites and minimize particle aggregation. The integration of COFs with S-scheme heterojunctions enhances charge carrier separation while maintaining high redox capability, resulting in synergistic performance improvements.131For example, an in situ growth process was employed to decorate positively charged ZnIn2S4 onto negatively charged COF, forming an S-scheme COF/ZnIn2S4 core–shell heterojunction for photocatalytic H2 generation.42 Microstructural analysis (Fig. 13a) revealed that flower-like ZnIn2S4, serving as the shell, enveloped the spherical COF nanoparticles, creating a robust interface between the core and shell. This design enhanced the surface area and porosity, promoted efficient mass transport, and reduced the charge-diffusion distance. These improvements were corroborated by the EIS Nyquist spectra (Fig. 13b), which demonstrated that the heterojunction significantly reduced the interfacial charge transfer resistance and accelerated charge migration and separation. Additionally, the substantial changes in the binding energies of the C 1s, N 1s, Zn 2p, In 3d, and S 2p orbitals in the COF/ZnIn2S4 system compared to those of pure ZnIn2S4 and COF confirmed the successful formation of the heterojunction. The COF/ZnIn2S4 heterojunction attained an outstanding H2 production rate of 695 μmol g−1 h−1, which was further enhanced to 2711 μmol g−1 h−1 after the introduction of Pt as a co-catalyst. Moreover, this heterojunction displayed an AQY of 2.45% at 400 nm and a 98.9% increase in lifetime, in contrast to the bare COF. Upon light irradiation, the photogenerated electrons in the LUMO of the COF perished with holes in the VB of ZnIn2S4, effectively retaining high-potential electrons in the CB of ZnIn2S4 and holes in the HOMO of the COF, as illustrated in Fig. 13c. The preserved electrons react with H+ to generate H2, whereas the holes are utilized by Na2S/Na2SO3. The S-scheme charge-migration mechanism was further validated by EPR analysis (Fig. 13d). Pure COF did not exhibit any DMPO-˙O2 signals owing to its insufficient reduction potential (0.12 V vs. NHE) compared to the standard ˙O2− potential (−0.33 V vs. NHE). Conversely, ZnIn2S4 displayed a strong DMPO-˙O2 signal due to its adequate CB potential (−1.14 V vs. NHE). Notably, the COF/ZnIn2S4 system showed a significantly enhanced DMPO-˙O2 signal, indicating the pile-up of high-potential electrons in the CB of ZnIn2S4 rather than their migration to the low-potential COF, which aligned well with the S-scheme charge transport route. This study underscores that in situ anchoring of the ZnIn2S4 shell around the COF core not only enhances charge separation but also facilitates numerous reactive sites, facilitating efficient photocatalytic H2 generation.
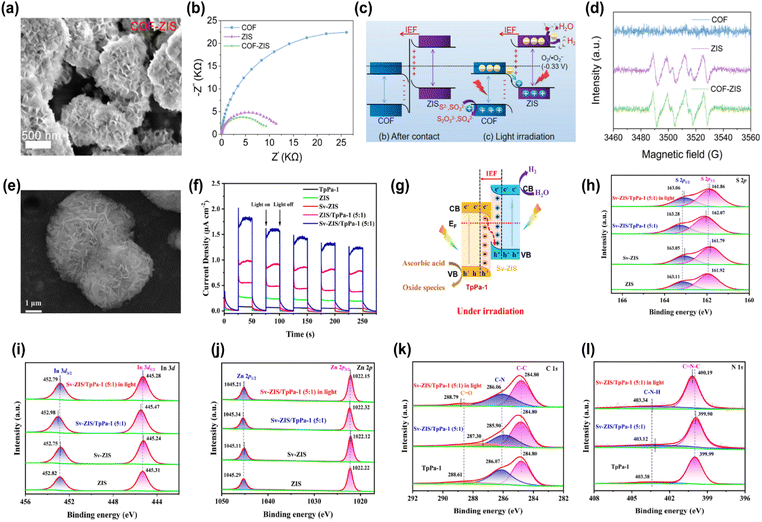 | ||
| Fig. 13 (a) SEM image, (b) EIS Nyquist plots, (c) charge transfer mechanism, and (d) DMPO analysis of COF/ZnIn2S4. Reproduced with permission from ref. 42. Copyright 2024, Elsevier. (e) SEM image, (f) photocurrent response, (g) charge transfer mechanism, ex situ/in situ XPS analysis, (h) S 2p, (i) In 3d, (j) Zn 2p, (k) C 1s, and (l) N 1s of SVs-ZnIn2S4/TpPa-1. Ref. 104, copyright 2024, Elsevier. | ||
Defect formation is critical for optimizing the photocatalytic properties of COF-based materials, particularly S-scheme heterojunction systems. Defects introduce active sites, enhance reactant–catalyst interactions, and create mid-gap states that expand visible-light absorption. These structural irregularities act as charge trapping centers, reducing electron–hole recombination and complementing the spatial charge separation of S-scheme heterojunctions.132 Additionally, defects improve interfacial bonding within the heterojunction, boosting charge transfer efficiency.133 As a self-modification strategy, defect engineering maintains the chemical stability and durability of COF-based materials, making it an effective approach to enhance photocatalytic performance without compromising stability.
For example, Wang et al. used a hydrothermal method to introduce sulfur vacancies (SVs) in ZnIn2S4, subsequently integrating SVs-ZnIn2S4 onto TpPa-1 at varying mass ratios to construct S-scheme heterojunctions for photocatalytic H2 generation.104 TEM analysis (Fig. 13e) confirmed that flower-like spheres of SVs-ZnIn2S4, composed of nanosheets, were uniformly distributed on block-like TpPa-1, establishing a robust interfacial interaction. This configuration enhances the surface area and increases the density of the catalytic centers. The successful creation of SVs in ZnIn2S4 was validated through EPR analysis, which demonstrated their role as charge-trapping sites, facilitating the separation of photoinduced electron–hole pairs. This was also supported by the highest photocurrent density recorded for the SVs-ZnIn2S4/TpPa-1 heterojunction compared with its pure counterparts, as shown in Fig. 13f. Moreover, the formation of SVs introduced impurity levels below the CB of ZnIn2S4, effectively reducing its bandgap and extending its light harvesting range. This extension facilitated the generation of additional charges for the redox processes. Consequently, the optimal SVs-ZnIn2S4/TpPa-1 achieved an impressive hydrogen production level of 2745 μmol g−1 h−1, with an AQY of 4.67% at 420 nm. Furthermore, the photocatalyst demonstrated exceptional stability and maintained its performance and structural integrity after multiple consecutive cycles. Upon light irradiation, the photogenerated electrons in the CB of TpPa-1 perished with holes in the VB of SVs-ZnIn2S4 (Fig. 13g), enhancing the separation of the high-potential electrons in the CB of SVs-ZnIn2S4, which subsequently reduced H+ to H2. The S-scheme charge migration route was verified using ISIXPS analysis (Fig. 13h–l). The binding energies of the Zn 2p, In 3d, and S 2p orbitals of SVs-ZnIn2S4, as well as C 1s and N 1s in TpPa-1, exhibited red and blue shifts in the dark state within the heterojunction compared to those of the pure materials, demonstrating the migration of free electrons from SVs-ZnIn2S4 to TpPa-1. Upon light irradiation, the binding energies shifted in the reverse direction, confirming that photoinduced electrons from TpPa-1 migrated to SVs-ZnIn2S4, which matched well with the S-scheme charge transfer route. Thus, the formation of defects has a profound effect on the photocatalytic efficiency of S-scheme heterojunctions. In COF-based S-scheme heterojunctions, defects not only modulate the electronic properties and energy levels of the integrated component but also enhance reaction kinetics by introducing additional active sites and charge trap locations.33
Furthermore, defect engineering extends light absorption and manipulates carrier density, supporting the separation and migration of photoinduced charges.47 It is thus established that defect formation induces a red shift in light harvesting due to the production of intermediate levels. However, it is important to note that this extension in light absorption occurs at the cost of the redox potential of the photoinduced electron–hole pairs. A redox potential that is too low cannot overcome the potential barrier for hydrogen generation, adversely impacting the photocatalytic performances.44 Additionally, defects can act as charge recombination centers, which increase the recombination level of photogenerated electron–hole pairs, further reducing the photocatalytic H2 generation efficiency.58 Therefore, future studies should focus on optimizing the defect density and distribution within the heterojunction by selecting advanced synthetic methods to mitigate the negative effects on light absorption and charge redox potential, ultimately enhancing H2 generation performance.
Microstructure optimization is another crucial approach for improving the H2 generation performance of COF-based S-scheme photocatalysts.132 Various specific COF nanostructures, such as 0D (e.g., nanoparticles and quantum dots), 1D (e.g., nanorods, nanotubes, and nanowires), 2D (e.g., nanosheets and nanoplates), and 3D (e.g., nanonetwork structures and nanoflowers), each with different physiochemical properties, have been used to construct S-scheme heterojunctions. These nanostructures enable the controllable formation and utilization of photogenerated charge carriers.134,135 Among these, 2D COF-based S-scheme heterojunctions stand out because of their ability to increase the surface area, provide abundant active sites, shorten charge diffusion distances, and offer multiple charge transport channels. These advantages help decrease the recombination rate of photogenerated charge carriers and extend the light absorption. For example, Sun et al. adopted an in situ process to decorate WO3 nanoplates onto the surface of flower-like TpPa-1-COF to develop an S-scheme WO3/TpPa-1-COF heterojunction for photocatalytic H2 production.86 TEM analysis confirmed a strong interfacial interaction between TpPa-1-COF and WO3, facilitating an increased surface area that improved mass transportation and provided frequent active sites to favor the photoreaction. The maximized current density and lowest EIS Nyquist arc diameter of the optimized WO3/TpPa-1-COF heterojunction, compared to its counterparts, demonstrated that the creation of the S-scheme heterojunction effectively improved the separation and transfer of photoinduced charge pairs, leading to a higher density of charges available for redox processes. Consequently, the optimized WO3/TpPa-1-COF heterojunction exhibited a maximum hydrogen generation efficiency of 19.89 mmol g−1 h−1, which was five times higher than that of the pure TpPa-1-COF. Moreover, the photocatalyst showed an AQE of 12.4% at 420 nm with negligible performance and structural loss, even after four recycling experiments, demonstrating its strong stability. Under light irradiation, the photogenerated electrons in the CB of WO3 perished with holes in the VB of TpPa-1-COF, enhancing the separation of the high-potential electrons in the CB of TpPa-1-COF and holes in the VB of WO3. The retained electrons in the CB of TpPa-1-COF migrated to Pt, where they reacted with H+ to produce hydrogen, whereas the holes in the VB of WO3 were captured by ascorbic acid. This work revealed that controlling the microstructure can enhance the surface area and the separation and transfer of photogenerated charge carriers, thus promoting photocatalytic H2 generation. Other examples of COF-based S-scheme heterojunctions that are considered excellent photocatalysts for H2 generation are listed in Table 1. In comparison to binary COF-based heterojunctions, dual S-scheme heterojunctions offer enhanced charge transport and separation capabilities. However, to the best of our knowledge, no COF-based dual S-scheme heterojunctions for photocatalytic H2 generation have been reported to date. Future studies should explore this approach, as the combined effects of the three semiconductors could significantly extend light absorption and promote charge separation, minimize electron–hole recombination, and ultimately improve photocatalytic performance.
CO2 reduction
Photocatalytic CO2 reduction, alongside hydrogen evolution, holds significant promise for addressing environmental and energy challenges.34 Converting CO2 into fuels such as methane and methanol not only mitigates atmospheric CO2 levels but also offers alternative energy sources.40 However, CO2 reduction poses considerable difficulties due to its chemical inertness and the need for high over-potentials to drive multi-electron transfer processes.46 This complexity makes the reaction less straightforward compared to hydrogen generation and often results in poor product selectivity. Despite these hurdles, photocatalytic CO2 reduction under ambient conditions has emerged as a potential approach capable of lowering greenhouse gas emissions while generating beneficial compound feedstocks.47 Since the groundbreaking research by Inoue and colleagues, numerous studies have highlighted the potential of this method for generating products such as carbon monoxide, methanol, and methane.49 A key area of recent research has focused on the application of COF-based S-scheme heterojunctions for photocatalytic CO2 capture and conversion. These materials, composed of porous networks formed from organic linkers, boast high surface areas and tailorable pore structures, rendering them efficient outlets for CO2 adsorption.59 Advancements in COF-based S-scheme heterojunction design have enhanced both the adsorption capacity and selectivity for CO2. Such modifications are particularly important for separating CO2 from mixed gas streams, which is a common requirement for industrial processes and power plant emissions. The ability of COFs to selectively target CO2, even in the presence of other gases, is a critical advantage in practical applications. Additionally, the reusability of these materials further enhances their appeal as sustainable tools for carbon capture and conversion technologies.32 The implications of the above extend beyond theoretical interest, offer tangible solutions for lowering greenhouse gas emissions, and promote sustainable energy applications.For instance, Wang et al. reported an enhanced photocatalytic CO2 conversion efficiency utilizing an S-scheme g-C3N4 (NH)/Tp-Tta COF heterojunction synthesized through a self-assembly process.46 TEM analysis revealed that Tp-Tta COF nanosheets were uniformly distributed over the surface of g-C3N4 (NH) nanosheets through strong interfacial interactions. This homogeneous scattering increases the surface area, providing abundant active sites, reducing charge diffusion distances, and creating multiple charge transport channels. These factors collectively facilitated mass transport and broadened light absorption, both of which are crucial for supporting the photocatalytic reaction. The optical properties were validated using absorption spectra and PL analysis (Fig. 14a and b). The heterojunction exhibited the most pronounced extension in light harvesting and the lowest PL intensity compared with its individual components. These results indicate that the strong interfacial bonding and π–π interactions between g-C3N4 (NH) and Tp-Tta COF effectively enhanced the light absorption and charge separation. Consequently, the optimized photocatalyst achieved a CO generation level of 11.25 μmol h−1, which is 3.2 fold superior to that of pure g-C3N4 (NH). Moreover, the photocatalyst maintained stable CO generation over a continuous 7-hours period. The S-scheme charge-transfer route within the g-C3N4 (NH)/Tp-Tta COF heterojunction was confirmed by EPR analysis (Fig. 14c). Pure Tp-Tta COF exhibited no DMPO-˙O2− signal due to its CB potential being less negative than the O2/˙O2− redox potential (−0.33 V vs. NHE). Conversely, g-C3N4 (NH) showed a distinct DMPO-˙O2 signal, confirming that its photogenerated electrons had sufficient reduction potential. The g-C3N4 (NH)/Tp-Tta COF heterojunction displayed a significantly stronger DMPO-˙O2− signal, indicating the accumulation of high-energy photoinduced electrons in the CB of g-C3N4 (NH) owing to the S-scheme charge-migration mechanism. In the absence of this mechanism, electrons would migrate from the high-potential g-C3N4 (NH) to the lower-potential Tp-Tta COF, resulting in a diminished DMPO-˙O2− signal. Upon light excitation, the preserved electrons in the CB of g-C3N4 (NH) converted CO2 to CO, whereas the holes in the VB of Tp-Tta COF oxidized TEOA. Thus, the intimate interaction and creation of the S-scheme heterojunction between g-C3N4 (NH) and Tp-Tta COF are key factors in the enhanced CO2 reduction performance.
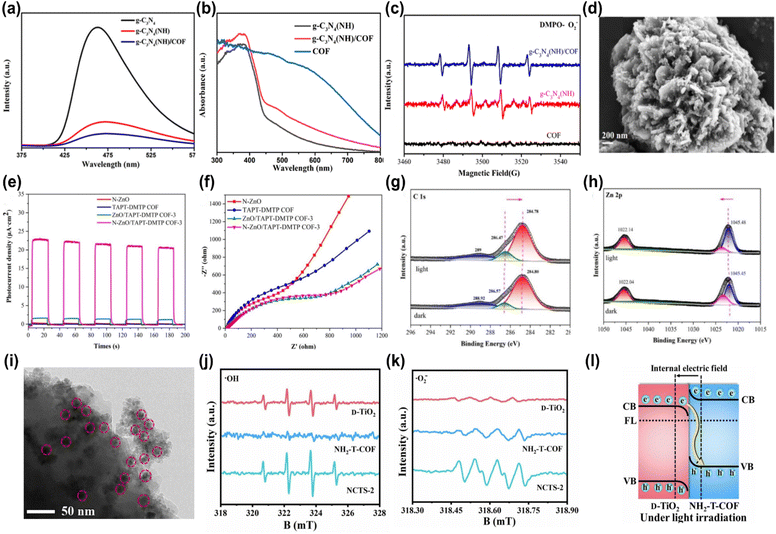 | ||
| Fig. 14 (a) PL analysis, (b) absorption spectra, and (c) EPR analysis of g-C3N4 (NH)/Tp-Tta COF. Reproduced with permission from ref. 46, copyright 2022, Elsevier. (d) SEM image, (e) photocurrent response, (f) EIS Nyquist plots, in situ XPS spectra (g) C 1s, and (h) Zn 2p of N–ZnO/TAPT-DMTP COF. Reproduced with permission from ref. 91, copyright 2025, Elsevier. (i) TEM image, (j) DMPO-˙O2− signal, (k) DMPO-˙OH signal, and (l) charge-transfer mechanism of N-COF/TiO2. Reproduced with permission from ref. 47, copyright 2022, Elsevier. | ||
The energy band structure design plays a critical role in controlling the charge transfer behavior and the redox capabilities of COF-based S-scheme heterojunctions.136 Carefully tuning the band positions not only governs the directionality and efficiency of photogenerated electron–hole pair separation but also aligns the thermodynamics to favor CO2 reduction processes.137 Therefore, selecting semiconductors with appropriate band structures and rationally adjusting their electronic properties are essential strategies to optimize charge transfer and separation in COF-based S-scheme heterojunctions. As an example, Mu et al. employed an imine-bridged strategy to couple a D–A COF, specifically TAPT-DMTP COF, onto imine-modified ZnO (N–ZnO) with matching energy bands, forming an S-scheme N-ZnO/TAPT-DMTP COF heterojunction for CO2 reduction.91 SEM analysis (Fig. 14d) confirmed the successful integration of flower-like N–ZnO onto the coral-like surface of TAPT-DMTP COF, resulting in a porous morphology with an increased surface area. This structure provides abundant active sites and shortens the charge diffusion distance, facilitating the separation and transfer of photogenerated electron–hole pairs. The electrochemical measurements further supported these results. The N–ZnO/TAPT-DMTP COF heterojunction displayed the highest photocurrent density and lowest EIS Nyquist arc diameter among its counterparts (Fig. 14e and f). This behavior suggests that the amine groups in N–ZnO interact strongly with DMTP, creating robust interfacial coupling and enhancing charge-transport channels for rapid charge migration. XPS analysis confirmed the formation of the N–ZnO/TAPT-DMTP COF heterojunction. A significant negative shift in the C 1s binding energy and a positive shift in the Zn 2p binding energy, compared to the pure materials, indicated electron transfer from N–ZnO to TAPT-DMTP COF. Under light irradiation, these shifts reversed (Fig. 14g and h), indicating that the migration of photoinduced electrons from the TAPT-DMTP COF to N–ZnO—aligned well with the S-scheme charge transfer route. The optimized N–ZnO/TAPT-DMTP COF photocatalyst demonstrated superior CO and CH4 generation levels of 11.07 and 4.86 μmol g−1, respectively, significantly outperforming the individual components. Additionally, the optimized N–ZnO/TAPT-DMTP COF photocatalyst exhibited insignificant efficiency and structural loss after five consecutive runs, indicating its high stability. Under illumination, photogenerated electrons in the CB of N–ZnO perished with holes in the VB of the TAPT-DMTP COF owing to the synergistic effects of BIEF, band bending, and electrostatic attraction. This process enhances the separation of high-energy electrons in the CB of the TAPT-DMTP COF and holes in the VB of N–ZnO. The retained electrons in the CB of the TAPT-DMTP COF facilitated the reduction of CO2 into CO and CH4, whereas the holes in the VB of N–ZnO oxidized water to produce ˙OH radicals. More research efforts are expected to deepen the understanding of the matching energy band structure to realize charge transfer dynamics through advanced techniques, quantifying the role of BIEF using DFT to guide more effective interface engineering while optimizing the morphology (e.g., nanosheets or hollow structures) to improve the surface area and active site exposure. Expanding light harvesting through plasmonic enhancements or co-catalyst incorporation could further improve performance, while exploring alternative reaction conditions and scaling synthesis methods for real-world applications would advance the practical potential of photocatalysts.
The interface quality of COF-based S-scheme heterojunctions plays a pivotal role in governing the migration and separation efficiency of photogenerated charge carriers.29 Enhancing the photocatalytic performance hinges on improving the interfacial contact between integrated materials, as the interface is central to facilitating efficient charge carrier migration and separation.44 Strengthening the interface bonding is crucial for reducing interfacial charge transfer resistance. One of the most efficient processes to improve the interface bonding strength is in situ growth for constructing S-scheme heterojunctions. For example, Wang et al. employed an in situ anchoring approach to integrate defective TiO2 onto the surface of T-COF, forming an S-scheme heterojunction with improved interfacial bonding strength for photocatalytic CO2 reduction.47 TEM analysis (Fig. 14i) showed that TiO2 nanoparticles were uniformly distributed across the surface of T-COF spheres, indicating successful integration with robust interfacial contact. The resulting porous morphology contributed to an increased surface area and larger pore size, facilitating CO2 migration to the exposed active sites for redox reactions. EXAFS analysis confirmed that TiO2 strongly riveted onto the T-COF network via coordination with N atoms, validating the formation of a stable heterojunction with robust interfacial interactions. The w.f. of TiO2 (5.10 eV) exceeded that of T-COF (4.85 eV), indicating the transport of free electrons from T-COF to TiO2 and the generation of an interfacial BIEF directed from T-COF to TiO2. EPR study verified the S-scheme carrier transfer between T-COF and TiO2, as shown in Fig. 14j and k. Defective TiO2 exhibited no DMPO-˙O2− signal but showed a significant DMPO-˙OH signal, suggesting that its electrons lacked sufficient reduction potential, whereas its holes possessed a strong oxidation potential. Conversely, the lack of a DMPO-˙OH signal and the presence of a pronounced DMPO-˙O2− signal in the EPR spectra of T-COF highlight the effectiveness of the photoinduced electrons. In contrast, the EPR spectra of N-COF/TiO2 exhibited strong intensities for both the DMPO-˙O2− and DMPO-˙OH signals, indicating the accumulation of high-potential electrons in the conduction band of T-COF and holes in the valence band of TiO2, which matched well with the S-scheme transfer route (Fig. 14l). The optimized N-COF/TiO2 photocatalyst demonstrated excellent CO2 reduction performance, achieving an AQY of 6.81% at 365 nm, which was 8- and 9-fold superior to that of T-COF and TiO2, respectively. Furthermore, this photocatalyst exhibited robust stability over five successive rounds, with an insignificant efficiency reduction. These results indicate the significance of precise interface engineering in COF-based heterojunctions. In situ growth methods have demonstrated potential for enhancing interfacial bonding and photocatalytic efficiency, although the underlying atomic-level mechanisms require further investigation. Comparative studies with other semiconductor materials can also provide a deeper understanding of the advantages and limitations of COF-based S-scheme heterojunctions.
H2O2 production
Hydrogen peroxide (H2O2), a green reactive oxygen species (ROS) with strong oxidative stability, is ideal for antibacterial applications.1 It is widely used in sanitation and environmental remediation. Semiconductor photocatalysis offers a sustainable, renewable, and cost-effective method for H2O2 synthesis, eliminating the need for expensive and hazardous precious-metal catalysts. This solar-driven process enables on-site H2O2 production, thereby reducing storage and transportation risks.138 Recent advances in semiconductor photocatalysis have focused on enhancing H2O2 production efficiency and selectivity.139 S-scheme heterojunctions have proven effective in addressing conventional photocatalytic limitations by combining semiconductors with different bandgaps and electronic properties. This structure promotes superior charge separation and transfer: electrons in the CB of one semiconductor migrate to another with a more negative potential, while holes move to the VB of the other semiconductor with a more positive potential.140,141 This reduces electron–hole recombination and enhances photocatalytic performance. For H2O2 synthesis, this mechanism allows electrons to reduce oxygen on the photocatalyst surface, forming H2O2, while holes oxidize various substrates.142 Acidic conditions enhance proton availability, aiding hydroxyl radical generation, which further reacts to form H2O2, completing the cycle.143 H2O2 photocatalytic production involves the oxygen reduction reaction (ORR) and water oxidation reaction (WOR). The ORR follows a two-electron pathway to produce superoxide anions (O2−), which convert to H2O2, or a four-electron pathway reducing oxygen directly to water.144 WOR oxidizes water to generate hydroxyl radicals, contributing to H2O2 formation.145 The photocatalyst's selectivity for the two-electron ORR, requiring a CB potential more negative than 0.68 V, is crucial for high H2O2 yields.146 COFs offer tunable porosity, large surface area, and structural flexibility, optimizing light absorption and charge dynamics essential for redox reactions.147 COFs' chemical stability and functionalizable surfaces make them suitable for diverse environmental conditions.For example, Xu et al. employed an in situ growth approach to decorate SnS2 nanoplates onto TaTp COF via a hydrothermal process, forming an S-scheme SnS2/TaTp COF heterojunction to enhance photocatalytic H2O2 production.117 Microstructural analysis (Fig. 15a) revealed that the nanoplates, composed of agglomerated nanoparticles, were effectively grown on the TaTp COF surface, ensuring intimate interfacial contact. This structure resulted in an increased surface area, providing abundant active sites and efficient charge transfer routes to facilitate the photoreaction. The formation of the SnS2/TaTp COF heterojunction not only extended the light absorption, generating additional charge carriers, but also improved the charge separation and transport owing to the excellent conductivity of SnS2. This effectively suppressed electron–hole recombination, as evidenced by the smallest arc diameter in the EIS Nyquist plot and the highest photocurrent density observed for SnS2/TaTp COF compared to its counterparts. The optimized heterojunction exhibited H2O2 production efficiencies 4.6- and 2.1-fold superior to that of bare SnS2 and TaTp COF, respectively. Moreover, stability tests over four consecutive cycles demonstrated no loss in performance or structural integrity, confirming the high stability of the SnS2/TaTp COF photocatalyst. To verify the S-scheme charge transfer route, in situ XPS analysis was conducted (Fig. 15b–f). Under dark conditions, the binding energies of the C 1s, N 1s, and O 1s orbitals in the SnS2/TaTp COF heterojunction exhibited a blue shift, whereas the Sn 3d and S 2p orbitals indicated a positive shift, indicating the migration of free electrons from SnS2 to TaTp COF. Upon light irradiation, the binding energies of C 1s, O 1s, Sn 3d, and S 2p reversed, confirming the transfer of photogenerated electrons from TaTp COF to SnS2, in alignment with the S-scheme charge transfer route. During photocatalysis, the retained electrons in the CB of SnS2 reduce O2 to ˙O2−, which consequently reduces H+ to produce H2O2. Simultaneously, the photoinduced holes in the VB of TaTp COF oxidize H2O, producing O2 and H+. These findings emphasize the potential of engineered heterojunctions in solar-driven H2O2 production, while underscoring the need for further optimization in interface engineering, electronic structure modulation, and long-term stability to advance scalable photocatalytic applications.
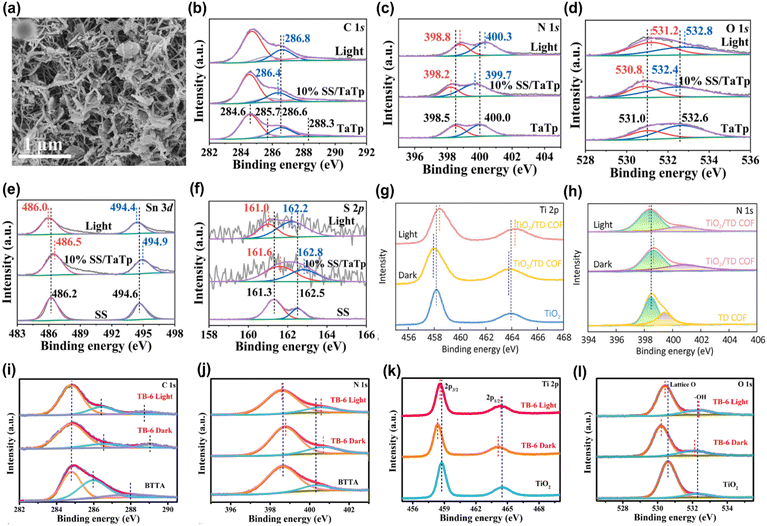 | ||
| Fig. 15 (a) SEM image and XPS analysis (b) C 1s, (c) N 1s, (d) O 1s, (e) Sn 3d, and (f) S 2p of SnS2/TaTp COF. Reproduced with permission from ref. 117, copyright 2025, Elsevier. XPS analysis of (g) Ti 2p and (h) N 1s of TiO2/COF. Reproduced with permission from ref. 118. Copyright 2025, Elsevier. XPS analysis (i) C 1s, (j) N 1s, (k) Ti 2p, and (l) O 1s of TiO2/BTTA (reproduced with permission from ref. 37 copyright 2023, Elsevier). | ||
For heterojunction photocatalysts, the semiconductor surface serves as a pivotal site for redox reactions, whereas the interface plays a critical role in controlling the migration and separation of photogenerated charge pairs. Consequently, the microstructure of the heterojunction and its interface significantly influences charge migration and the extent of redox processes, thus affecting the overall photocatalytic efficiency. To optimize the structure of S-scheme heterojunctions, controlling the morphology is essential to enhance light transmission depth and expose additional active sites, fostering better interaction between active sites and reactants, thus boosting photocatalytic efficiency. For instance, Zhao et al. assembled TiO2 nanoparticles onto a flower-like COF to construct an S-scheme TiO2/COF heterojunction with robust interfacial contact, resulting in enhanced photocatalytic performance for H2O2 production.118 TEM examination indicated that 5 nm TiO2 nanoparticles were uniformly distributed across the surface of the flower-like COF, indicating successful integration. This integration leads to extended light absorption, promoting increased charge generation. Furthermore, the fs-TA decay kinetics demonstrated a reduced average carrier lifetime for TiO2/COF compared to pure TiO2, while exhibiting a longer lifetime than pure COF. This indicates enhanced separation and transport of photoinduced charge pairs due to the generation of an S-scheme heterojunction between TiO2 and COF. The optimized TiO2/COF photocatalyst achieved a maximum H2O2 generation level of 1326 μmol g−1 h−1, maintaining excellent structural integrity and performance stability over five successive recycling experiments. The superior w.f. of TiO2 (6.73 eV) in contrast to that of COF (5.04 eV) facilitated the transfer of free electrons from COF to TiO2, leading to the formation of an interfacial IEF. This phenomenon was corroborated by the XPS analysis. As depicted in Fig. 15g and h, the binding energies of the Ti 2p and N 1s orbitals exhibited negative and positive shifts within the TiO2/COF heterojunction under dark conditions compared to pure TiO2 and COF, respectively, indicating electron migration from COF to TiO2 upon integration. Upon light excitation, the binding energies of the Ti 2p and N 1s orbitals moved in the opposite direction, signifying the transport of photoinduced electrons from the CB of TiO2 to the HOMO of COF, which matched well with the S-scheme charge transfer route. The retained electrons in the LUMO of the COF reacted with O2 to produce ˙O2− radicals, which subsequently reacted with H+ to yield H2O2. Simultaneously, the retained holes in the VB of TiO2 oxidize water to generate ˙OH radicals.
Optimized interfacial contact areas facilitate abundant charge transport channels, ensuring swift transfer and effective separation of photogenerated electron–hole pairs.29 Heterojunctions formed through mere physical mixing of semiconductors provide limited interfacial contact, resulting in insufficient charge carrier separation and migration, thereby diminishing photocatalytic performance.48 Conversely, the formation of core–shell heterojunctions offers extensive active surfaces, enabling effective charge carrier migration and utilization.22 The robust interfacial interaction between the core and shell not only provides ample charge diffusion and transport pathways but also enhances light absorption and utilization due to multiple light reflections within the cavity of the core–shell heterojunction. For instance, Yang et al. reported enhanced photocatalytic performance for H2O2 production using an S-scheme TiO2/BTTA core–shell heterojunction.37 Microstructural analysis revealed a rough surface on the TiO2/BTTA composite; attributed to the in situ anchoring of BTTA nanoparticles onto TiO2 nanofibers which produce a porous shell, promoting the contact of reactants to the reactive centers on the TiO2 core. This structural modification led to an increased surface area and abundant active sites, thereby promoting mass transport and enhancing redox reaction capacity. The creation of the heterojunction was verified by Raman spectroscopy, which displayed characteristic peaks corresponding to both TiO2 and BTTA. XPS analysis (Fig. 15i–l) demonstrated shifts in the binding energies, providing deeper insight into the charge-transfer behavior. The N 1s and C 1s orbitals in the TiO2/BTTA heterojunction exhibited a positive shift compared to pure BTTA, whereas the Ti 2p and O 1s orbitals showed a negative shift relative to pure TiO2 under dark conditions. These observations indicate electron migration from BTTA to TiO2.
Under light irradiation, the binding energies shifted in the opposite direction, suggesting photoinduced electron transfer from TiO2 to BTTA, which matched well with the S-scheme charge transfer route. The optimized TiO2/BTTA photocatalyst achieved a maximum H2O2 production rate of 740 μmol L−1 h−1 and a furoic acid transformation efficiency of 96%. Moreover, the photocatalyst exhibited negligible performance degradation and no structural changes after four consecutive cycles, confirming its high stability. Upon light exposure, the photoexcited electrons in the CB of TiO2 perish with holes in the HOMO of BTTA. This route enhances the separation of high-energy electrons in the LUMO of BTTA and holes in the VB of TiO2. The retained electrons reduce O2 to superoxide radicals, leading to H2O2 formation, while the holes oxidize ethanol and water to produce ˙C2H5OH and ˙OH radicals, which subsequently undergo dehydrogenation to form furoic acid. However, despite these promising results, future studies should explore the scalability of such heterojunctions, recyclability under real-world conditions, and the potential impact of different reaction environments on charge separation ability. In addition, a deeper insight into the interplay between the structural properties and charge dynamics could pave the way for further optimization, potentially extending the application of such heterojunctions to broader photocatalytic processes, including water splitting and CO2 reduction.
Organic transformation
Photocatalytic organic synthesis provides a green and sustainable strategy for producing valuable chemicals by utilizing solar energy for power reactions while reducing harmful byproducts.148 Traditional photocatalysts, such as metal complexes and organic dyes, often suffer from drawbacks such as instability, photodegradation, and high costs.149 To overcome these issues, researchers are actively exploring novel photocatalysts that offer enhanced efficiency, long-term stability, and cost-effectiveness, particularly those free from precious metals.150 COFs have attracted attention as potential candidates owing to their high surface area, robust stability, and customizable structures. Consequently, the application of COFs in photocatalytic organic synthesis has gained significant traction, leading to notable discoveries and improvements. However, a major challenge in COF-based photocatalysis is the rapid recombination of photoinduced charges, which restricts their performance.151 One effective strategy to address this concern is the development of S-scheme heterojunctions, which facilitate charge separation by selectively directing electrons and holes, thereby improving the photocatalytic activity. For example, Zhao et al. employed an in situ encapsulation strategy to synthesize a series of S-scheme heterojunctions incorporating W18O49 nanobundles and TpPa-H COF, aiming to explore their potential in the photocatalytic transformation of benzylamine into imine.152 SEM analysis (Fig. 16a) confirmed that the W18O49 nanobundles were effectively encapsulated within TpPa-H, forming a robust interfacial contact that enhanced mass transport and light absorption. This strong interface facilitates the availability of numerous active sites and a high density of charge carriers, thereby promoting the photocatalytic reaction. Furthermore, XPS analysis demonstrated that the binding energies of the W 4f orbital in W18O49 (Fig. 16c) exhibited a blue shift in the heterojunction compared with pristine W18O49. Conversely, the C 1s orbital in TpPa-H (Fig. 16b) displays a red shift relative to that of pure TpPa-H. These shifts provide compelling evidence of electron migration from TpPa-H to W18O49, driven by the formation of a well-defined heterojunction with strong interfacial interactions. Additionally, the optimized W18O49/TpPa-H heterojunction exhibited the smallest EIS Nyquist arc diameter and lowest PL intensity, indicating that heterojunction formation significantly reduced the interfacial charge migration resistance. This enhancement facilitated efficient charge separation, thereby promoting redox reactions.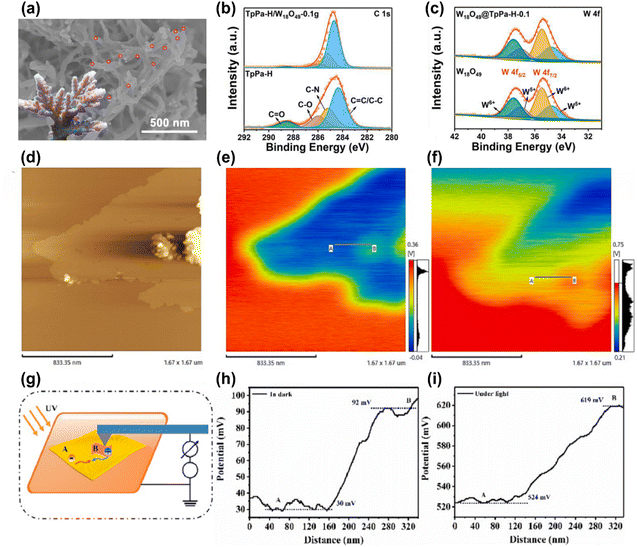 | ||
| Fig. 16 (a) SEM image, XPS spectra (b) C 1s, and (c) W 4f of pure materials and W18O49/TpPa-H heterojunction. Ref. 152, copyright 2024, Elsevier, (d) AFM image, surface potential distribution of the W18O49/TpPa-H heterojunction; (e) dark state; (f) light irradiation; (g) photoexcitation of KPFM; line scanning potential difference (h) dark state; and (i) light irradiation. Ref. 153, copyright 2023, Elsevier. | ||
As a result, the optimized heterojunction achieved an outstanding benzylamine conversion efficiency of 99% within 240 min, which was approximately three and ten times higher than those of TpPa-H and W18O49, respectively. Furthermore, recycling experiments confirmed the exceptional stability of the developed heterojunction, as no noticeable variation in the benzylamine conversion rate or changes in the XRD patterns were observed, indicating the preservation of both performance and structural integrity. The w.f. of W18O49 (8.05 eV) was considerably larger than that of TpPa-H (5.62 eV), leading to the generation of an interfacial BIEF from TpPa-H to W18O49. This results in band bending owing to electron migration upon integration. Within this system, the photogenerated electrons in the CB of W18O49 perished with holes in TpPa-H, thereby enhancing the charge separation by accumulating electrons in TpPa-H and holes in W18O49. These electrons subsequently react with O2 to generate superoxide radicals, whereas the holes react with benzylamine to form intermediate ions, which subsequently react with superoxide radicals to yield the final imine product. Future research should focus on tailoring the heterojunction interface through defect engineering, doping, or ligand functionalization to further modulate charge transfer dynamics and enhance selectivity.
The morphology of the component materials in an S-scheme heterojunction plays a crucial role in determining the interface quality and interfacial charge transport pathways.154 Compared to nanoparticles or larger blocks, the fabrication of nanosheets and tiny blocks not only strengthens the interfacial interaction between the constituent materials but also provides abundant active sites and short charge transport channels. This, in turn, enhanced the charge separation efficiency and accelerated redox reactions, ultimately leading to superior photocatalytic performance.155 For example, Sun et al. documented the development of an S-scheme CdS/COF heterojunction to evaluate its efficiency in hydrogen production alongside an organic transformation process.153 TEM imaging confirmed the successful integration of TpBD onto the CdS nanosheets, forming a robust interface that extended light absorption (500–600 nm), thereby generating a large number of charges for redox processes. Furthermore, the CdS/TpBD heterojunction exhibited a higher current density and a lower EIS Nyquist arc diameter than its pure components, validating the improved separation and migration of the photogenerated charge pairs. This improvement was attributed to the presence of abundant charge-transport pathways and reduced charge-transport impedance. Consequently, the S-scheme photocatalyst achieved an excellent hydrogen production rate of 15.1 mmol g −1 h −1 while simultaneously converting ascorbic acid to 2,3-diketo-L-gulonic acid with high efficiency. To further substantiate the S-scheme charge transport route between CdS and TpBD, a KPFM analysis was conducted. AFM imaging (Fig. 16d) confirmed the anchoring of TpBD onto CdS nanosheets. As illustrated in Fig. 16e, the surface potential of point A (CdS) was superior to that of point B (TpBD) in the absence of light, generating a potential difference of 62 mV and establishing a BIEF from TpBD to CdS. Upon light irradiation, the surface potential of both materials increased because of their n-type conductivity, leading to a relatively higher surface potential difference of 95 mV between points A and B (Fig. 16f–i). This shift indicated the transport of photoinduced electrons from CdS to TpBD under the influence of BIEF (Fig. 16g). Consequently, the photoinduced holes in the VB of TpBD recombine with electrons in the CB of CdS, following the S-scheme charge migration mechanism. The remaining electrons in the CB of TpBD participated in the reduction of H+ to generate hydrogen, whereas the holes in the VB of CdS facilitated the oxidation of ascorbic acid into C6H8O7. However, the scalability and real-world applicability of CdS/COF S-scheme heterojunctions remain unclear. Investigating its performance under natural sunlight conditions and in flow-reactor systems could bridge the gap between laboratory-scale research and industrial applications. Addressing these gaps is essential for advancing S-scheme heterojunctions beyond fundamental research for practical and sustainable photocatalytic applications.
The construction of an S-scheme heterojunction by combining organic COFs with an appropriate inorganic material featuring a suitable band structure effectively merges the inherent advantages of porous COFs with the charge-separation capabilities of S-scheme systems. This synergy enhances the charge-carrier diffusion and transport, thereby facilitating photocatalytic reactions. For example, an in situ growth technique was applied to prepare ZnIn2S4 nanosheets on Tp-Tta nanoplates, forming an S-scheme heterojunction to investigate their cooperative photocatalytic performance for H2 production and organic transformation.156 Microstructural analysis confirmed that ZnIn2S4 nanosheets were vertically aligned on Tp-Tta nanoplates with strong interfacial contact, leading to enhanced light scattering, improved absorption, and more effective transfer and separation of photoinduced charges to drive the reaction. The XPS analysis provided compelling evidence for heterojunction formation. The constituent orbitals of ZnIn2S4 exhibited a positive binding energy shift within the ZnIn2S4/Tp-Tta system compared to that of pure ZnIn2S4, whereas Tp-Tta showed a negative shift relative to its pristine form. This binding energy shift not only confirmed the charge transfer between the two materials but also highlighted the presence of BIEF and band bending. Under light irradiation, the reversal of these orbital shifts further authenticated the S-scheme charge-transport route. As a result, the ZnIn2S4/Tp-Tta photocatalyst demonstrated outstanding performance, achieving maximum H2 production and furfural generation levels of 9.73 and 12.1 mmol g−1 h−1, respectively, while maintaining excellent structural stability. Mechanistically, the photogenerated electrons in the CB of ZnIn2S4 actively reacted with H+ to generate hydrogen, whereas the holes in the VB of Tp-Tta oxidized furfuryl alcohol to generate furfuryl. Future research should focus on defect engineering and interface modifications to enhance charge separation efficiency and product selectivity for broader photocatalytic applications.
Summary and future perspectives
In summary, COF-based S-scheme heterojunctions have emerged as a potential strategy to address the inherent limitations of COFs, such as insufficient light absorption, rapid charge recombination, and restricted redox potential. By integrating S-scheme heterojunctions, these materials exhibit enhanced photocatalytic properties, benefiting from improved charge-separation efficiency, extended light absorption, and superior redox capabilities. Despite rapid advancements in this field, a comprehensive review of COF-based S-scheme heterojunction photocatalysts is still lacking. This work, therefore, provides an in-depth discussion of synthesis approaches, structural configurations, and charge transfer mechanisms, particularly distinguishing S-scheme heterojunctions from other heterojunction categories, such as type-II and Z-schemes. Furthermore, various preparation strategies and recent developments in COF-based S-scheme heterojunctions are systematically summarized, followed by an exploration of their application in diverse photocatalytic processes. Although these photocatalysts have demonstrated significant potential, the field remains in its early phases, facing various constraints that need to be addressed for broader practical applications. Key aspects, such as structural stability, charge transport efficiency, and large-scale synthesis, require further research and optimization. This review focuses on facilitating a valuable understanding of the current advancements and inspires future advancements, ultimately promoting the growth of COF-based S-scheme heterojunction photocatalysts for a wider range of applications. Although noteworthy progress has been made in this promising domain, there are still a number of challenges for further exploration.(I) To fully harness the advantages of COFs in S-scheme heterojunctions, a deeper insight into the electronic connections between different materials is essential. This includes clarifying how these interactions influence charge carrier behavior, particularly in terms of separation efficiency and transfer dynamics. Moreover, the impact of the surrounding reaction conditions on the photocatalytic efficiency, such as variations in temperature, humidity, and chemical composition, requires further exploration to optimize operational efficiency.
(II) Despite their promising properties, the synthesis of COF-based S-scheme heterojunction photocatalysts typically relies on energy-intensive methods that involve high temperature and pressure. These approaches not only increase production costs, but also pose environmental concerns.157 The development of scalable and sustainable fabrication techniques that minimize energy consumption and eliminate hazardous reagents is a crucial step toward broader adoption in industrial applications. To address these challenges, researchers are actively exploring alternative synthetic pathways, including low-temperature in situ growth, green solvent approaches, and self-assembly methods.
(III) Accurate characterization is fundamental to understanding the structural and functional properties of these photocatalysts. However, techniques such as scanning electron microscopy (SEM), transmission electron microscopy (TEM), X-ray diffraction (XRD), X-ray photoelectron spectroscopy (XPS), and Raman spectroscopy have been used. Advancements in in situ and operando characterization techniques, such as real-time X-ray diffraction (XRD) and transmission electron microscopy (TEM), will enable the direct observation of structural and electronic changes during photocatalytic reactions. Additionally, transient absorption spectroscopy (TAS) offers insights into charge carrier dynamics, whereas in situ X-ray absorption spectroscopy (XAS) can provide information on electronic state evolution, enhancing the mechanistic understanding of these materials.
(IV) From a morphological perspective, most COF-based photocatalysts feature predominantly microporous architecture, which can limit the mass transport and active site accessibility. Engineering hierarchical porosity by integrating mesopores and macropores or designing more complex architectures, such as hollow or open-framework structures, could enhance diffusion properties and catalytic performance. Techniques such as template-assisted synthesis, defect engineering, and selective etching are viable routes for achieving these structural modifications.
(V) Another constraint is the limited range of semiconducting materials currently integrated into COF-based S-scheme heterojunctions. Most studies have focused on various oxides, sulfides, and carbonaceous materials. The inclusion of other potential photocatalysts, including metal nitrides, bismuth oxyhalides, and metal selenides, can significantly enhance the efficiency and versatility of these heterojunctions. Moreover, although COF/non-COF heterojunctions have been extensively studied, the development of COF-on-COF heterojunctions remains relatively unexplored. Such all-COF structures offer unique advantages in terms of tunability, electronic coupling, and interfacial charge transport.
(VI) A deeper exploration of the correlation between the material structure and performance is necessary to fully realize the potential of COF-based S-scheme heterojunctions in photocatalysis. In situ spectroscopic techniques, including infrared and Raman spectroscopy, can provide real-time monitoring of the structural and compositional changes during photocatalytic reactions.158 Additionally, maximizing the concurrent consumption of photoinduced electrons and holes in redox reactions could enable high-value chemical transformation. Because the porous nature of COFs influences reactant diffusion, investigating the interfacial pore connectivity is critical for optimizing the mass transfer properties.159 Finally, translating these materials from laboratory-scale powders to macroscale configurations such as gels, films, or membranes would greatly enhance their practical applicability in various photocatalytic processes.
(VII) The formation of heterojunctions inherently involves lattice mismatch owing to differences in crystal structures, leading to strain-induced defects, increased charge recombination, and reduced photocatalytic performance.160 This issue is particularly significant in COF-based S-scheme heterojunctions, where interfacial defects hinder the charge transport. To overcome this challenge, strategies such as epitaxial growth, strain modulation, and interfacial passivation can minimize the mismatch-induced defects and enhance charge separation. van der Waals epitaxy offers a promising route by reducing the strain at the heterointerface, whereas advanced buffer layers with tailored mechanical and electronic properties can alleviate structural disparities without impeding charge dynamics. Additionally, linker engineering, molecular-level design, and π–π stacking optimization can improve interfacial compatibility and charge transport. Future research should focus on rationally designing charge-transport materials with tunable lattice parameters, integrating computational approaches to optimize heterojunction architecture, and developing scalable fabrication methods to enhance the stability and efficiency of COF-based S-scheme photocatalysts for practical solar energy conversion applications.
By advancing these key research directions, the development and application of COF-based S-scheme heterojunction photocatalysts can be accelerated significantly, opening new possibilities for their integration into sustainable energy and environmental technologies.
Data availability
Data availability is not applicable to this article as no new data were created or analyzed in this study.Author contributions
I. A. and G. L. designed the concept of this work. I. A., Z. U. D. B., A. A.-O. and S. B. A. wrote the first manuscript. Y. Z., G. L. and S. B. A. got the fund and wrote the final manuscript. All the authors discussed the results and commented on the manuscript.Conflicts of interest
There are no conflicts to declare.Acknowledgements
Gao Li acknowledge the financial support by the National Natural Science Foundation of China (22172167) and Open fund of State Key Laboratory of Catalysis (DICP 2024SKL-A-006). The authors extend their appreciation to the Deanship of Research and Graduate Studies at King Khalid University for funding this work through the Large Research Project under grant number RGP2/376/46.References
- J. Z. Hassan, A. Raza, U. Qumar and G. Li, Sustainable Mater. Technol., 2022, 33, e00478 CrossRef CAS.
- Q. Shi, Z. Qin, C. Yu, A. Waheed, H. Xu, Y. Gao, H. Abroshan and G. Li, Nano Res., 2020, 13, 939–946 CrossRef CAS.
- F. Xu and B. Weng, J. Mater. Chem. A, 2023, 11, 4473–4486 RSC.
- Q. Li, Z.-Q. Wu, D. Li, T.-H. Liu, H.-y. Yin, X.-B. Cai, W. Zhu, Z.-L. Fan and R.-Z. Li, J. Mater. Chem. A, 2023, 11, 2957–2968 RSC.
- M. S. Khalid, Z. Li, Y. Miao, X. Gu, Y. Zhang and G. Li, ACS Appl. Nano Mater., 2025, 8, 6201–6209 CrossRef CAS.
- G. Li, C. Zhu, X. Xi and Y. Cui, Chem. Commun., 2009, 2118–2120 Search PubMed.
- S. Ghosh, H. Küçükkeçeci, R. P. Paitandi, V. Weigelt, V. Dippold, S. Seki and A. Thomas, J. Mater. Chem. A, 2024, 12, 247–255 Search PubMed.
- Z. Zhao, W. Chen, G. Zhang and Y. Chen, J. Mater. Chem. A, 2023, 11, 26052–26062 Search PubMed.
- Y. Liu, X. Jiang, L. Chen, Y. Cui, Q.-Y. Li, X. Zhao, X. Han, Y.-C. Zheng and X.-J. Wang, J. Mater. Chem. A, 2023, 11, 1208–1215 Search PubMed.
- F. Huang, Y. Wang, X. Dong and X. Lang, J. Mater. Chem. A, 2024, 12, 7036–7046 RSC.
- J. Wang, Z. Dai, L. Wang, D. Zhang, Y. Wang, J. Li, F. Zhou and J. Huang, J. Mater. Chem. A, 2023, 11, 2023–2030 RSC.
- C. Yang, H. Hu, C. Qian and Y. Liao, J. Mater. Chem. A, 2023, 11, 25899–25909 RSC.
- I. M. A. Mekhemer, M. M. Elsenety, A. M. Elewa, K. D. G. Huynh, M. M. Samy, M. G. Mohamed, D. M. Dorrah, D. C. K. Hoang, A. F. Musa, S.-W. Kuo and H.-H. Chou, J. Mater. Chem. A, 2024, 12, 10790–10798 Search PubMed.
- Z. Li, T. Deng, S. Ma, Z. Zhang, G. Wu, J. Wang, Q. Li, H. Xia, S.-W. Yang and X. Liu, J. Am. Chem. Soc., 2023, 145, 8364–8374 CrossRef CAS PubMed.
- H. He, R. Shen, P. Zhang, G. Liang and X. Li, J. Mater. Chem. A, 2024, 12, 227–232 Search PubMed.
- J. Sun, J. Chakraborty, M. Deng, A. Laemont, X. Feng, Y.-Y. Liu and P. Van Der Voort, J. Mater. Chem. A, 2023, 11, 21516–21540 Search PubMed.
- X. Yu, K. Gong, S. Tian, G. Gao, J. Xie and X.-H. Jin, J. Mater. Chem. A, 2023, 11, 5627–5635 RSC.
- C. Liu, D.-L. Ma, P.-J. Tian, C. Jia, Q.-Y. Qi, G.-F. Jiang and X. Zhao, J. Mater. Chem. A, 2024, 12, 16063–16069 RSC.
- T. Wang, R. Zhang, P. Zhai, M. Li, X. Liu and C. Li, J. Mater. Chem. A, 2024, 12, 1292–1299 RSC.
- W. Dong, Y. Xiao, Z. Qin, B. Qiao and L. Li, J. Mater. Chem. A, 2023, 11, 14760–14767 RSC.
- I. Ahmad, Y. Zhang, S. A. AlFaify, G. Li and I. M. Ashraf, J. Alloys Compd., 2024, 178309 Search PubMed.
- Z. Li, L. Xu, Z. Babar, A. Raza, Y. Zhang, X. Gu, Y. Miao, Z. Zhao and G. Li, Nano Res., 2024, 17, 4729–4736 Search PubMed.
- Q. Li, Y. Zhang, L. Xu, H. Chen, L. Zhang, M. S. Khalid, Z. Li, Z. Li and G. Li, Appl. Catal., B, 2025, 361, 124706 CrossRef CAS.
- Y. Sun, Q. Shi, X. Gu, B. Wang, B. Lumbers and G. Li, J. Colloid Interface Sci., 2024, 662, 76–86 CrossRef CAS PubMed.
- Q. Shi, X. Zhang, Z. Li, A. Raza and G. Li, ACS Appl. Mater. Interfaces, 2023, 15, 30161–30169 CrossRef CAS PubMed.
- H. Shi, Q. Shi, S. Piracha and G. Li, J. Mater. Chem. A, 2024, 12, 20270–20277 RSC.
- Q. Shi, G. Ping, X. Wang, H. Xu, J. Li, J. Cui, H. Abroshan, H. Ding and G. Li, J. Mater. Chem. A, 2019, 7, 2253–2260 Search PubMed.
- H. Wang, J. Fan, J. Zou, Y. Zheng, D. Wang and J. Jiang, J. Mater. Chem. A, 2024, 12, 30429–30441 RSC.
- A. Raza, Z. Qin, S. Ahmad, M. Ikram and G. Li, J. Environ. Chem. Eng., 2021, 9, 106569 CrossRef CAS.
- R. Xiong, X. Ke, W. Jia, Y. Xiao, B. Cheng and S. Lei, J. Mater. Chem. A, 2023, 11, 2178–2190 RSC.
- S. Zhang, X. Gong, Q. Shi, G. Ping, H. Xu, A. Waleed and G. Li, ACS Omega, 2020, 5, 15942–15948 CrossRef CAS PubMed.
- P. Sanwal, X. Gu, Y. Zhang and G. Li, Precis. Chem., 2025 DOI:10.1021/prechem.4c00087.
- Y. Pan, Z. Huang, D. Zheng and C. Yang, J. Colloid Interface Sci., 2023, 644, 19–28 CrossRef CAS PubMed.
- Q. Niu, Q. Chen, G. Huang, L. Li, Y. He and J. Bi, J. Colloid Interface Sci., 2023, 643, 102–114 Search PubMed.
- X. Zhao, A. Li, D. Yang, T.-Y. Qiu, Z. Zhao, S.-L. Wang, X. Mu and H.-Q. Tan, J. Colloid Interface Sci., 2024, 653, 67–76 Search PubMed.
- J. Bi, Z. Zhang, J. Tian and G. Huang, J. Colloid Interface Sci., 2024, 661, 761–771 CrossRef CAS PubMed.
- Y. Yang, J. Liu, M. Gu, B. Cheng, L. Wang and J. Yu, Appl. Catal., B, 2023, 333, 122780 CrossRef CAS.
- Y. Wen, Z. Huang, M. Zhao and L. Zhao, Appl. Surf. Sci., 2022, 596, 153485 CrossRef CAS.
- M.-L. Xu, J.-R. Li, X.-M. Wu, T. Yu, G.-Y. Qin, F.-J. Wang, L.-N. Zhang, K. Li and X. Cheng, Appl. Surf. Sci., 2022, 602, 154371 CrossRef CAS.
- S. Li, H. Yu, Y. Wang, S. Wang, L. Zhang, P. Zhu, C. Gao and J. Yu, Appl. Surf. Sci., 2023, 615, 156335 CrossRef CAS.
- F. Wang, B. Da, Y. Jin, P. Sanwal, L. Cui, S. Chen, J. Xu, B. Xue and G. Li, J. Environ. Sci., 2025, 155, 37–47 Search PubMed.
- Y. Chen, Y. Li, W. Chen, W. Xu, Z. Han, A. Waheed, Z. Ye, G. Li and A. Baiker, Nano Res., 2022, 15, 1366–1374 CrossRef CAS.
- A. Raza, M. Ikram, S. Guo, A. Baiker and G. Li, Adv. Sustainable Syst., 2022, 6, 2200087 Search PubMed.
- D. You, Z. Pan and Q. Cheng, J. Alloys Compd., 2023, 930, 167069 Search PubMed.
- S. He, Q. Rong, H. Niu and Y. Cai, Appl. Catal., B, 2019, 247, 49–56 CrossRef CAS.
- J. Wang, Y. Yu, J. Cui, X. Li, Y. Zhang, C. Wang, X. Yu and J. Ye, Appl. Catal., B, 2022, 301, 120814 CrossRef CAS.
- Y. Wang, Z. Hu, W. Wang, Y. Li, H. He, L. Deng, Y. Zhang, J. Huang, N. Zhao, G. Yu and Y.-N. Liu, Appl. Catal., B, 2023, 327, 122419 Search PubMed.
- S. Bao, Q. Tan, S. Wang, J. Guo, K. Lv, S. A. C. Carabineiro and L. Wen, Appl. Catal., B, 2023, 330, 122624 CrossRef CAS.
- X. An, J. Bian, K. Zhu, R. Liu, H. Liu and J. Qu, Chem. Eng. J., 2022, 442, 135279 CrossRef CAS.
- Y. Zhang, J. Qiu, B. Zhu, M. V. Fedin, B. Cheng, J. Yu and L. Zhang, Chem. Eng. J., 2022, 444, 136584 CrossRef CAS.
- Y.-P. Zhang, W. Han, Y. Yang, H.-Y. Zhang, Y. Wang, L. Wang, X.-J. Sun and F.-M. Zhang, Chem. Eng. J., 2022, 446, 137213 Search PubMed.
- G. Sun, J. Zhang, B. Cheng, H. Yu, J. Yu and J. Xu, Chem. Eng. J., 2023, 476, 146818 CrossRef CAS.
- Z. Hu, Y. Wang, L. Wang, Q. Wang, Q. Zhang, F. Cui and G. Jiang, Chem. Eng. J., 2024, 479, 147534 CrossRef CAS.
- M. Zhang, H. Cao, M. Wu, Y. Zhang, J. Zhu, J. Xu, J. Deng and T. Zhou, Chem. Eng. J., 2024, 487, 150684 Search PubMed.
- Z. Tong, D.-K. Ma, J. Wu, X. Hu, N. Jiang, X. Yang and T. Peng, Chem. Eng. J., 2024, 496, 154262 Search PubMed.
- P. Dong, A. Zhang, T. Cheng, J. Pan, J. Song, L. Zhang, R. Guan, X. Xi and J. Zhang, Chin. J. Catal., 2022, 43, 2592–2605 Search PubMed.
- Z. Liang, R. Shen, P. Zhang, Y. Li, N. Li and X. Li, Chin. J. Catal., 2022, 43, 2581–2591 CrossRef CAS.
- Y. Zhao, C. Yang, S. Zhang, G. Sun, B. Zhu, L. Wang and J. Zhang, Chin. J. Catal., 2024, 63, 258–269 CrossRef CAS.
- L. Wang, G. Huang, L. Zhang, R. Lian, J. Huang, H. She, C. Liu and Q. Wang, J. Energy Chem., 2022, 64, 85–92 Search PubMed.
- H.-X. Zhang, S.-H. Ma, H.-X. Wang, S.-C. Li, H.-Y. Shen, D.-M. Kong, F.-Y. Wang and L.-N. Zhu, J. Environ. Chem. Eng., 2024, 12, 111899 CrossRef CAS.
- Z. Huang, C. Zhang, W. Zhang and H. Liu, J. Environ. Chem. Eng., 2024, 12, 114501 CrossRef CAS.
- H. Ran, X. Liu, J. Fan, Y. Yang, L. Zhang, Q. Guo, B. Zhu and Q. Xu, J. Materiomics, 2025, 11, 100918 CrossRef.
- J. Sun, H. Liu, S. Wang, Y. Zhang, C. Bie and L. Zhang, J. Materiomics, 2025, 11, 100975 CrossRef.
- Y. Zhang, Y. Wang, Y. Liu, S. Zhang, Y. Zhao and J. Zhang, J. Materiomics, 2025, 11, 100985 CrossRef.
- Z. Fan, X. Guo, F. Liu, Y. Li, L. Zhang and Z. Jin, Appl. Mater. Today, 2022, 29, 101637 CrossRef.
- S. Yuan, Y. Feng, S. Liang, Y. Zhang, J. Zhao, Y. Wang, P. Yang and Q. Gu, Mater. Today Energy, 2025, 48, 101804 CrossRef CAS.
- H. Hu, X. Zhang, K. Zhang, Y. Ma, H. Wang, H. Li, H. Huang, X. Sun and T. Ma, Adv. Energy Mater., 2024, 14, 2303638 CrossRef CAS.
- B.-B. Luan, X. Chu, Y. Wang, X. Qiao, Y. Jiang and F.-M. Zhang, Adv. Mater., 2024, 36, 2412653 CrossRef CAS PubMed.
- J. Qiu, K. Meng, Y. Zhang, B. Cheng, J. Zhang, L. Wang and J. Yu, Adv. Mater., 2024, 36, 2400288 CrossRef CAS PubMed.
- R. Gao, R. Shen, C. Huang, K. Huang, G. Liang, P. Zhang and X. Li, Angew. Chem., Int. Ed., 2025, 64, e202414229 CrossRef CAS PubMed.
- Z. Shi, Z. Chen, Y. Zhang, X. Wang, T. Lu, Q. Wang, Z. Zhan and P. Zhang, Sep. Purif. Technol., 2022, 288, 120717 CrossRef CAS.
- Y.-P. Zhang, H.-L. Tang, H. Dong, M.-Y. Gao, C.-C. Li, X.-J. Sun, J.-Z. Wei, Y. Qu, Z.-J. Li and F.-M. Zhang, J. Mater. Chem. A, 2020, 8, 4334–4340 RSC.
- B. Dong, Y. Wan, Q. Cheng, H. Zhou and Z. Pan, Environ. Sci.: Nano, 2022, 9, 2799–2814 RSC.
- L. Sun, W. Wang, T. Kong, H. Jiang, H. Tang and Q. Liu, J. Mater. Chem. A, 2022, 10, 22531–22539 RSC.
- J. Pang, W. Chen, J. Hu, J. Cheng, M. Tang, Z. Liu and R. Tan, Catal. Sci. Technol., 2023, 13, 6198–6210 RSC.
- H. Shi, Q. Shi, X. Gu, B. Wang, B. Lumbers and G. Li, J. Colloid Interface Sci., 2024, 673, 527–536 CrossRef CAS PubMed.
- Y. Tang, Y. Wang, X. Cheng and H. Zhang, J. Mater. Chem. A, 2025, 13, 3350–3358 RSC.
- Q. Xiong, Q. Shi, X. Gu, X. Sheng, Y. Sun, H. Shi, L. Xu and G. Li, J. Colloid Interface Sci., 2024, 654, 1365–1377 CrossRef CAS PubMed.
- P. Dong, T. Cheng, J.-l. Zhang, J. Jiang, L. Zhang, X. Xi and J. Zhang, ACS Appl. Energy Mater., 2023, 6, 1103–1115 CrossRef CAS.
- J. Z. Hassan, A. Zaheer, A. Raza and G. Li, Sustainable Mater. Technol., 2023, 36, e00609 CrossRef CAS.
- Q. Niu, S. Dong, J. Tian, G. Huang, J. Bi and L. Wu, ACS Appl. Mater. Interfaces, 2022, 14, 24299–24308 Search PubMed.
- Q. Niu, W. Chen, G. Pan, L. Li, Y. Yu, J. Bi, L. Wu and J. C. Yu, ACS Mater. Lett., 2024, 6, 1411–1417 CrossRef CAS.
- P. Chen, M. Yan, J. Shi, M. Feng, Q. Li, H. Chen, Y. Xie, J. Lei, M. Wakeel, B. Hu and X. Wang, Chem.–Eur. J., 2025, e202500305 CrossRef PubMed.
- H. Chen, S. Gao, G. Huang, Q. Chen, Y. Gao and J. Bi, Appl. Catal., B, 2024, 343, 123545 CrossRef CAS.
- J. Zhang, F. B. Busari, Y. Zhang, S. Guo, Y. Zhao, B. Wang, Q. Zeng, Z. Zhao and G. Li, Nano Res. Energy, 2025, 4, e9120140 CrossRef.
- L. Sun, L. Li, J. Fan, Q. Xu and D. Ma, J. Mater. Sci. Technol., 2022, 123, 41–48 CrossRef CAS.
- L. Qi, C. Xiao, W. Lu, H. Zhang, Y. Zhou, J. Qi, Y. Yang, Z. Zhu and J. Li, Sep. Purif. Technol., 2025, 354, 128758 CrossRef CAS.
- S. Yu, C. Li, Y. Lin, J. Zhang, Y. Liu and F. Yu, Sep. Purif. Technol., 2024, 341, 126888 CrossRef CAS.
- J.-Y. Yue, Z.-X. Pan, P. Yang and B. Tang, ACS Mater. Lett., 2024, 6, 3932–3940 CrossRef CAS.
- C. Wang, S. Shi, F. Duan, S. Lu, H. Zhu, M. Du, X. Chen and M. Chen, J. Mater. Chem. A, 2022, 10, 16524–16532 RSC.
- M. Mu, L. Meng, S. Ma, W. Chen, X. Yin and G. Bai, J. Colloid Interface Sci., 2025, 686, 63–74 CrossRef CAS PubMed.
- R. Gao, R. Shen, C. Huang, K. Huang, G. Liang, P. Zhang and X. Li, Angew. Chem., Int. Ed., 2025, 64, e202414229 CrossRef CAS PubMed.
- L. Zhang, J. Zhang, H. Yu and J. Yu, Adv. Mater., 2022, 34, 2107668 CrossRef CAS PubMed.
- H. Zhang, J. Liu, Y. Zhang, B. Cheng, B. Zhu and L. Wang, J. Mater. Sci. Technol., 2023, 166, 241–249 CrossRef CAS.
- L. Wang, J. Mao, G. Huang, Y. Zhang, J. Huang, H. She, C. Liu, H. Liu and Q. Wang, Chem. Eng. J., 2022, 446, 137011 CrossRef CAS.
- Y. Wang, H. Li, Q. Lin, J. Zhao, X. Fang, N. Wen, Z. Zhang, Z. Ding, R. Yuan, X.-H. Huang and J. Long, ACS Catal., 2023, 13, 15493–15504 CrossRef CAS.
- X. Ma, S. Li, Y. Gao, N. Li, Y. Han, H. Pan, Y. Bian and J. Jiang, Adv. Funct. Mater., 2024, 34, 2409913 CrossRef CAS.
- Y. Xu, Z. Sun, S. Fan, X. Han, L. Li, Z. Gao and C. Wang, J. Mater. Chem. A, 2024, 12, 27180–27205 RSC.
- G. Zhao, C. Yang, W. Meng and X. Huang, J. Mater. Chem. A, 2024, 12, 3209–3229 RSC.
- T. Mazuelo, T. Naranjo, M. Gomez-Mendoza, A. Herrero Pizarro, L. Collado, M. Barawi, F. Gándara, M. Liras and V. A. de la Peña O'Shea, J. Mater. Chem. A, 2024, 12, 1476–1487 RSC.
- C. Huang, T. Huang, X. L. Li, W. Zhou and M. Ding, J. Mater. Chem. A, 2024, 12, 8734–8746 RSC.
- Y. Zhang, Z. Chen, Z. Shi, T.-t. Lu, D. Chen, Q. Wang and Z. Zhan, Sep. Purif. Technol., 2021, 275, 119216 CrossRef CAS.
- N. Arif, M. N. Zafar, M. Batool, M. Humayun, M. A. Iqbal, M. Younis, L. Li, K. Li and Y.-J. Zeng, J. Mater. Chem. C, 2024, 12, 12653–12691 RSC.
- S.-D. Wang, L.-Y. Huang, L.-J. Xue, Q. Kang, L.-L. Wen and K.-L. Lv, Appl. Catal., B, 2024, 358, 124366 CrossRef CAS.
- H. Zhao, L. Wang, G. Liu, Y. Liu, S. Zhang, L. Wang, X. Zheng, L. Zhou, J. Gao, J. Shi and Y. Jiang, ACS Catal., 2023, 13, 6619–6629 CrossRef CAS.
- Y. Xia, K. Zhang, H. Yang, L. Shi and Q. Yi, Acta Phys.-Chim. Sin., 2024, 40, 2407012 CrossRef.
- H. Hu, X. Sun, H. Li, H. Pan, Y. Ma, H. Huang and T. Ma, Small, 2025, 21, 2407117 CrossRef CAS PubMed.
- Y.-N. Gong, X. Guan and H.-L. Jiang, Coord. Chem. Rev., 2023, 475, 214889 CrossRef CAS.
- Z. Fan, Y. Cai, Z. Yang, X. Zhang, R. Shao, S. Zhong, L. Zhao, D. Liu and S. Bai, Appl. Catal., B, 2024, 351, 123979 CrossRef CAS.
- H. He, S. Chen, W. Bi, X. Wen, S. Sun, P. Zhang, R. Shen and X. Li, Sol. RRL, 2023, 7, 2300532 CrossRef CAS.
- Q. Shi, Z. Qin, S. Sharma and G. Li, Chem. Rec., 2021, 21, 879–892 CrossRef CAS PubMed.
- P. Mou, G. Wan, L. Wu, D. Liu and G. Wang, J. Mater. Chem. A, 2023, 11, 4345–4354 RSC.
- J. Xu, W. Ma, P. He, Y. Zhou, X. Liu, Y. Chen, P. Zuo and Q. Zhuang, J. Mater. Chem. A, 2023, 11, 330–340 RSC.
- Q. Xia, Z. Han, Z. Zhang, Z. Huang, X. Wang, J. Chang, Q. Chen and M. Chen, J. Mater. Chem. C, 2023, 11, 4552–4569 RSC.
- W. Lu, T. Qin, B. Wang, J. Li, H. Zhu, G. Bai, Y. Li, L. Chen and X. Yan, ChemPhotoChem, 2023, 7, e202200230 CrossRef CAS.
- Y. He, P. Hu, J. Zhang, G. Liang, J. Yu and F. Xu, ACS Catal., 2024, 14, 1951–1961 CrossRef CAS.
- D. X. Xu, S. Dong, J. Lv, G. Huang, Q. Chen and J. Bi, Appl. Surf. Sci., 2025, 689, 162489 CrossRef.
- Y. Zhao, Y. Zhang, H. Tan, C. Ai and J. Zhang, J. Materiomics, 2025, 11, 100970 CrossRef.
- J. Zhang, H. Wang, Y. Zhang, Z. Li, D. Yang, D. Zhang, T. Tsukuda and G. Li, J. Phys. Chem. Lett., 2023, 14, 4179–4184 CrossRef CAS PubMed.
- G. Sahoo, T. G. Senthamaraikannan, H. S. Jeong, P. Bandyopadhyay, D.-H. Lim, S. K. Nayak and S. M. Jeong, J. Mater. Chem. A, 2024, 12, 15019–15035 RSC.
- Z. Qin, D. Zhao, L. Zhao, Q. Xiao, T. Wu, J. Zhang, C. Wan and G. Li, Nanoscale Adv., 2019, 1, 2529–2536 RSC.
- F. K. Busari, Z. Babar, A. Raza and G. Li, Sustainable Mater. Technol., 2024, 40, e00958 CrossRef CAS.
- J. Ding, Z. Peng, Z. Wang, C. Zeng, Y. Feng, M. Yang, G. Hu, J. Luo and X. Liu, J. Mater. Chem. A, 2024, 12, 28023–28031 RSC.
- X. Li, L. Gao, M. Wang, D. Lv, P. He, Y. Xie, X. Zhan, J. Li and Z. Lin, J. Mater. Chem. A, 2023, 11, 6772–6795 RSC.
- A. Raza, A. A. Rafi, J. Z. Hassan, A. Rafiq and G. Li, Appl. Surf. Sci. Adv., 2023, 15, 100402 CrossRef.
- H.-L. Wu, M.-Y. Qi, Z.-R. Tang and Y.-J. Xu, J. Mater. Chem. A, 2023, 11, 3262–3280 RSC.
- W. Song, G. Qi and B. Liu, J. Mater. Chem. A, 2023, 11, 12482–12498 RSC.
- Z. Yang, H. Li, T. Xu, M. She, J. Chen, X. Jia, P. Liu, X. Liu and J. Li, J. Mater. Chem. A, 2023, 11, 2679–2689 RSC.
- M. Yu, P. Li, R. Huang, C. Xu, S. Zhang, Y. Wang, X. Gong and X. Xing, J. Mater. Chem. B, 2023, 11, 734–754 RSC.
- A. Tiwari, S. Walia, S. Sharma, S. Chauhan, M. Kumar, T. Gadly and J. K. Randhawa, J. Mater. Chem. B, 2023, 11, 1029–1043 RSC.
- J. Zhang, W. Zhou, J. Zhao, L. Xu, X. Jiang, Z. Li, Y. Peng and G. Li, Small, 2025, 21, 2408324 CrossRef CAS PubMed.
- X. Ren, M. Li, L. Qiu, X. Guo, F. Tian, G. Han, W. Yang and Y. Yu, J. Mater. Chem. A, 2023, 11, 5754–5765 RSC.
- L. Chen, G. Chen, C. Gong, Y. Zhang, Z. Xing, J. Li, G. Xu, G. Li and Y. Peng, Nat. Commun., 2024, 15, 10501 CrossRef CAS PubMed.
- X. Liu, Y. Peng, Z. Liang, L. Wang, S. Han, Z. Dou, X. Lu, J. Tong, J. Liu and J. Li, J. Mater. Chem. C, 2023, 11, 2871–2879 RSC.
- J. Prakash, P. Kumar, N. Saxena, Z. Pu, Z. Chen, A. Tyagi, G. Zhang and S. Sun, J. Mater. Chem. A, 2023, 11, 10015–10064 RSC.
- Z. Ali, J. Ma, M. Hong and R. Sun, J. Mater. Chem. A, 2023, 11, 3297–3314 RSC.
- K. Zheng, J. Zhang, D. Zhao, Y. Yang, Z. Li and G. Li, Nano Res., 2019, 12, 501–507 CrossRef CAS.
- S. Guo, S. Zhang, Q. Fang, H. Abroshan, H. J. Kim, M. Haruta and G. Li, ACS Appl. Mater. Interfaces, 2018, 10, 40599–40607 Search PubMed.
- Z. Li, Y. Xie, J. Gao, X. Zhang, J. Zhang, Y. Liu and G. Li, J. Mater. Chem. A, 2023, 11, 26573–26579 RSC.
- J. Shen, X. Qiu and Y. Zhu, J. Mater. Chem. A, 2023, 11, 11704–11711 RSC.
- Q. Shi, A. Raza, L. Xu and G. Li, J. Colloid Interface Sci., 2022, 625, 750–760 CrossRef CAS PubMed.
- N. Wang, Y.-S. Zhang, D.-D. Wei, H.-M. Duan, L.-M. Mo and H.-Y. Wang, J. Mater. Chem. A, 2023, 11, 4162–4169 RSC.
- M. Tamtaji, S. Cai, W. Wu, T. Liu, Z. Li, H.-Y. Chang, P. R. Galligan, S.-i. Iida, X. Li, F. Rehman, K. Amine, W. A. Goddard and Z. Luo, J. Mater. Chem. A, 2023, 11, 7513–7525 RSC.
- J. Zhang, Y. Xie, Q. Jiang, S. Guo, J. Huang, L. Xu, Y. Wang and G. Li, J. Mater. Chem. A, 2022, 10, 16920–16927 Search PubMed.
- Z. Li, H. Wang, Y. Xie, Y. Zhang, J. Gao, Y. Xu, L. Chen, L. Xu, E. Wang and G. Li, J. Colloid Interface Sci., 2025, 681, 16–24 Search PubMed.
- Y. Yao, H. Wang, K. Dong, H. Li, J. Liang, R. Li, S. Sun, Z. Cai, X. He, D. Zheng, Y. Luo, S. Alfaifi, D. Ma, W. Hu and X. Sun, J. Mater. Chem. A, 2023, 11, 22154–22160 RSC.
- W. Yu, Z. Zhu, C. Hu, S. Lin, Y. Wang, C. Wang, N. Tian, Y. Zhang and H. Huang, J. Mater. Chem. A, 2023, 11, 6384–6393 RSC.
- H. Chen, H. S. Jena, X. Feng, K. Leus and P. Van Der Voort, Angew. Chem., Int. Ed., 2022, 61, e202204938 CrossRef CAS PubMed.
- Z. Wang, Z. Qi, S. Wang, J. Du, W. Dai, F. Lu and Q. Deng, J. Colloid Interface Sci., 2025, 683, 736–745 CrossRef CAS PubMed.
- M. Deng, J. Chakraborty, G. Wang, K. S. Rawat, L. Bourda, J. Sun, I. Nath, Y. Ji, P. Geiregat, V. Van Speybroeck and X. Feng, J. Am. Chem. Soc., 2025, 147, 10219–10230 CrossRef CAS PubMed.
- M. Lei, Y. Xue, L. Gan, B. Li, Y. Lu, Q. Yang, W. Wang, S. Wang, Q. Su and Q. Wu, J. Mol. Catal., 2025, 577, 114976 CrossRef CAS.
- X. Zhao, A. Li, D. Yang, T. Y. Qiu, Z. Zhao, S. L. Wang, X. Mu and H. Q. Tan, J. Colloid Interface Sci., 2024, 653, 67–76 CrossRef CAS PubMed.
- G. Sun, J. Zhang, B. Cheng, H. Yu, J. Yu and J. Xu, Chem. Eng. J., 2023, 476, 146818 CrossRef CAS.
- S. Ghasemi, A. Parastesh, M. Padervand, H. Ren, X. Li, A. Labidi, M. Signoretto, E. A. Dawi, T. Hamzehlouyan, E. Lichtfouse and C. Wang, Curr. Opin. Chem. Eng., 2025, 47, 101059 CrossRef.
- M. Bilal, M. Q. Alfaifi, S. B. Ahmed, M. M. Abduljawad, Y. I. Alrashed, E. S. Aldurahim and Y. A. Alassmy, Mater. Sci. Semicond. Process., 2025, 186, 109051 CrossRef CAS.
- Y. Wang, H. Li, Q. Lin, J. Zhao, X. Fang, N. Wen, Z. Zhang, Z. Ding, R. Yuan, X. H. Huang and J. Long, ACS Catal., 2023, 13(23), 15493–15504 CrossRef CAS.
- D. Golberg, Y. Bando, C. C. Tang and C. Y. Zhi, Adv. Mater., 2007, 19, 2413–2432 CrossRef CAS.
- J. Yang, Z. Chen, L. Zhang and Q. Zhang, ACS Nano, 2024, 18, 21804–21835 CrossRef CAS PubMed.
- L. Yao, A. M. Pütz, H. Vignolo-González and B. V. Lotsch, J. Am. Chem. Soc., 2024, 146, 9479–9492 CrossRef CAS PubMed.
- S.-P. Qi, R.-T. Guo, Z.-X. Bi, Z.-R. Zhang, C.-F. Li and W.-G. Pan, Small, 2023, 19, 2303632 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2025 |