A dual-functional S-scheme Ti3C2/MoS2–ZnIn2S4 heterojunction for accelerated photocatalytic H2 evolution and efficient solar evaporators†
Xin
Xiong‡
a,
Longji
Gui‡
a,
Li
Yang
b,
Jintao
Li
a,
Songsong
Xu
a,
Fang
Yu
*a,
Jingwen
Qian
 *a,
Tao
Mei
*a,
Tao
Mei
 a and
Xianbao
Wang
a and
Xianbao
Wang
 *a
*a
aHubei Collaborative Innovation Center for Advanced Organic Chemical Materials, Ministry-of-Education Key Laboratory for the Green Preparation and Application of Functional Materials, Hubei Key Laboratory of Polymer Materials (Hubei University), School of New Energy and Electrical Engineering, Hubei University, Wuhan, 430062, China. E-mail: ichqjw123@163.com
bSchool of Materials Science and Engineering, Yancheng Institute of Technology, Yancheng, 224051, China
First published on 23rd April 2025
Abstract
The present utilization of photocatalysts in energy conversion fails to meet the increasing demands for enhanced H2 evolution efficiency and optimal solar energy utilization, primarily due to the rapid recombination of photogenerated carriers at the interface and suboptimal solar utilization rate. This study employs interface engineering of S-heterojunctions to integrate MoS2–ZnIn2S4 onto the surface of the charge transport layer MXene. The high conductivity and layered structure of MXene effectively accelerate charge transfer, reduce electron–hole recombination efficiency, and enhance hydrogen production performance. When this composite material is loaded onto a three-dimensional porous natural silk aerogel (NSA) substrate, it utilizes the photothermal effect of infrared light to simultaneously enhance the generation of hydrogen and water vapor. The photothermal effect of this system exhibits a conversion efficiency of 94.2%, significantly elevating the system temperature and increasing the hydrogen production rate. Under these synergistic effects, the composite material achieves a maximum hydrogen production efficiency of 33.0 mmol g−1 h−1 at room temperature, representing a sixfold improvement compared to MoS2–ZnIn2S4, while the water evaporation rate of Ti3C2/MoS2–ZnIn2S4 NSAs reaches 2.6 kg m−2 h−1. This bifunctional system demonstrates exceptional adaptability and hydrogen production potential, offering a novel solution for solar-driven seawater desalination and hydrogen evolution.
1. Introduction
The rapid population growth and industrial expansion have led to a significant increase in the demand for water and clean energy, which are crucial for fostering a sustainable green society.1–5 Solar-powered water evaporation is emerging as a viable technique for maximizing the use of solar energy for seawater desalination.6–8 Meanwhile, solar-driven photocatalytic water splitting into hydrogen presents a promising pathway toward achieving a sustainable and uncontaminated energy landscape.9 Despite notable advancements in this research area, the majority of existing semiconductor photocatalysts face several limitations, including low solar energy utilization rates, rapid recombination of electron–hole pairs, and challenges in recovering photocatalyst powders. These factors collectively impede their efficacy in photocatalytic H2 production and practical applications. Various strategies, such as elemental doping, heterojunction construction, and structural and defect engineering, have been developed to enhance the H2 evolution efficiency of photocatalysts.10–12 Notably, almost 50% of solar radiation consists of infrared light,13 which poses challenges for utilization in photocatalytic water splitting due to its low energy. Recently, photothermal-assisted photocatalysis has given new ideas to enhance the efficiency of photocatalytic hydrogen production.14 The photothermal effect has the potential to elevate the local temperature of the photocatalyst, thereby facilitating the chemical reaction and increasing the production of hydrogen.14,15 However, in the practical application of powder photothermal photocatalytic materials, most of the thermal energy generated by the photothermal effect dissipates into the surrounding water, resulting in an ineffective enhancement of solar utilization.The integration of interfacial photo-vapor conversion technology with photocatalytic hydrogen evolution technology presents a novel approach to enhance the solar energy utilization efficiency of powder photocatalysts.16 Interfacial photo-vapor technology, initially introduced in 2014 (ref. 17), can significantly improve the efficiency of solar energy conversion by localizing solar-thermal energy conversion at the air/liquid interface. By designing a system that combines photothermal photocatalysts on interfacial substrates, simultaneous generation of hydrogen and pure water is realized, providing an effective way to deal with the global resource crisis, addressing both clean water and energy crises.16,18–20 To realize this dual-function system, it is crucial to identify materials that exhibit both efficient photocatalytic and photothermal properties.
Most photothermal semiconductors have a narrow bandgap structure, which enables better light absorption compared to wide band gap materials.21,22 Molybdenum disulfide, a typical metal sulfide semiconductor material with a narrow direct band gap, has been widely applied as either a photo-vapor conversion material or a cocatalyst for photocatalytic water splitting, owing to its suitable band structure, wide absorption range of ultraviolet to near-infrared light and adjustable two-dimensional structure.23–25 However, in the process of photocatalytic hydrogen production, the semiconductor's narrow bandgap leads to the quick recombination of photogenerated electrons and holes. The traditional type II heterojunction has the advantage of charge separation, and there is no doubt that the disadvantage of this heterojunction is that it sacrifices redox ability, and the electrostatic repulsion also affects the transfer efficiency of the carriers.26–28 Additionally, the disadvantages of traditional Z-type heterojunctions are more obvious, such as dependence on the solution environment and side reaction competition.28,29 In order to improve carrier transport and separation, Yu and his team proposed a novel S-type heterojunction,28,30,31 which opens up a new path for subsequent research. The photocatalytic system consists of a reductive photocatalyst (RP) and an oxidative photocatalyst (OP). When RP and OP come into contact, electrons spontaneously diffuse from RP to OP, resulting in the formation of positively and negatively charged electron layers at their interface. This electron transfer establishes an internal electric field (IEF) directed from RP to OP.32–35 Concurrently, the accumulation of electrons and holes induces upward and downward band bending in RP and OP, respectively.36 Furthermore, photogenerated electrons in the conduction band of OP recombine with holes in the valence band of RP due to Coulombic attraction. The synergistic effects of the internal electric field, band bending, and Coulombic interaction collectively enhance the redox capabilities of the system.37,38 Notably, the photogenerated electrons in the conduction band of RP and holes in the valence band of OP are preserved and subsequently participate in photocatalytic reactions.39
In recent years, the S-type heterojunction and various interfacial control technologies proposed by researchers have opened a new path for the design of photocatalysts. For examples, Kong proposed a specific S–Zn charge transfer pathway at the OP/RP interface.40 A heterogeneous interface formed between the two phases to optimize the electron arrangement on the catalyst surface.41 In addition, the efficient photocatalytic oxidation of benzyl alcohol was achieved by modifying ZnIn2S4 with dual cocatalysts,42 along with the significant differences in Fermi energy levels and band structures. The internal electric field (IEF) and band bending at the heterojunction interface promoted photocatalytic carrier separation and transport.43 Furthermore, it is proposed that a dual effect, encompassing both the internal electric field (IEF) and the photothermal effect within the S-scheme heterojunction, effectively enhances photocatalytic performance.44
Herein, a Ti3C2/MoS2–ZnIn2S4 S-scheme heterojunction was skillfully prepared by a hydrothermal method and oil bath deposition method. Ti3C2 (MXene), a novel two-dimensional layered material, exhibits exceptional electrical conductivity and a 2D/2D van der Waals structure with MoS2, thereby facilitating robust interaction forces and substantial contact areas. The structural design for the introduction of Ti3C2 into MoS2–ZnIn2S4 composites helps promote the formation of S-scheme heterojunctions.45,46 Natural silk nanofibril aerogels (NSAs) with controllable structures and great mechanical elasticity were treated as photothermal substrate materials to load Ti3C2/MoS2–ZnIn2S4 composites. The three-dimensional macroporous channels in the floating NSAs ensure that liquid water continuously reaches the surface, where it is then converted into water vapor and H2 by Ti3C2/MoS2–ZnIn2S4 composites. Benefiting from the photothermal effect and the S-scheme heterojunction, the maximum hydrogen production efficiency of the composite was observed to be 33.0 mmol g−1 h−1 at room temperature. The water evaporation rate based on the Ti3C2/MoS2–ZnIn2S4 NSAs could reach 2.6 kg m−2 h−1 under standard sunlight, and the photothermal conversion efficiency could reach 93.4%. This work maximizes the exploitation of solar energy resources and provides a novel solution for the realization of efficient vapor and hydrogen production simultaneously.
2. Results and discussion
2.1 Composition and structure analysis
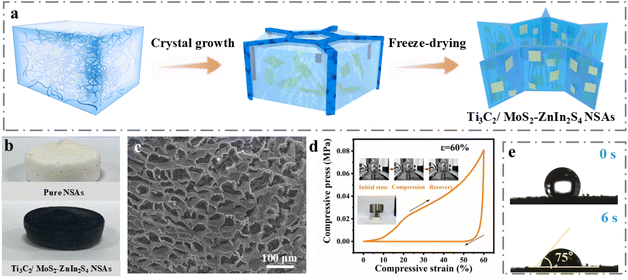
The preparation of materials and methods is shown in the ESI† based on previously published work.24,47,48 The morphology and structural features of the photothermal–photocatalytic composite play an important role in the final performance of the solar interfacial water evaporation and hydrogen production dual-function system. Fig. 1a shows the schematic diagram of the synthesis of the three-dimensional Ti3C2/MoS2–ZnIn2S4 heterojunction photothermal photocatalyst. First, accordion-shaped Ti3C2 MXene was synthesized by etching Ti3AlC2 powder with HF solution. Fig. S1a† shows the morphology of Ti3AlC2 and Fig. 1b shows Ti3C2 MXene after the etching of Ti3AlC2 powder. It can be seen that the tightly connected laminate structure of Ti3AlC2 becomes loose after etching, and the Ti3C2 MXene shows an accordion-like structure. After loading MoS2, the accordion-like Ti3C2's two-dimensional slits and regular lamellar surface are full of petal-like MoS2 (Fig. 1c), and the organ-like grain morphology of Ti3C2 powder provides a good substrate for the growth of MoS2 nanosheets. Finally, the photocatalyst ZnIn2S4 was deposited on MoS2/Ti3C2 by using a simple oil bath method. The sheets of ZnIn2S4 were intimately filled in the interstices of Ti3C2 and wrapped around MoS2 to establish a heterojunction, as shown in Fig. 1d. In addition, as shown in Fig. S2,† the elements S, Ti, Mo, C, Zn and In were uniformly distributed on the surface of the ternary composite Ti3C2/MoS2–ZnIn2S4. For comparison, the samples of pure ZnIn2S4, MoS2, Ti3C2/ZnIn2S4, and MoS2/ZnIn2S4 were prepared. From the SEM images in Fig. S1c–f,† it could be seen that both ZnIn2S4 and MoS2 exhibited relatively comparable nanosheet-like structures, which form nano-spheres when agglomerated with each other. The XRD patterns in Fig. S3a† demonstrated a change in the material phase from Ti3AlC2 to Ti3C2. The diffraction peaks of samples after HF etching located at the 2θ of 8.6°, 18.54°, 27.82°, and 60.71° could be observed, which were indexed to the (002), (004), (006), and (100) peaks of Ti3C2, demonstrating the successful preparation of Ti3C2 MXene. The Raman spectrum in Fig. S3b† further proved that the 2D Ti3C2 exhibited a good degree of graphitization. The XRD pattern of Ti3C2/MoS2–ZnIn2S4 in Fig. 1e demonstrated characteristic peaks of ZnIn2S4, Ti3C2, and MoS2, indicating the successful preparation of Ti3C2/MoS2–ZnIn2S4 composites. X-ray electron spectroscopic (XPS) characterization was performed to further verify the elemental composition of the as-synthesized materials. In Fig. 1f, the doublet present at approximately 229.6 eV and 232.8 eV in the Mo 3d spectrum was assigned to Mo4+ 3d5/2 and 3d3/2. Two characteristic peaks corresponding to Mo6+ near 234.7 and 236.3 eV might come from the slight oxidation of surface Mo. The In 3d spectrum in Fig. 1g showed two peaks near 452.3 and 444.7 eV corresponding to In 3d5/2 and In 3d3/2. In Fig. S4† of Ti 2p spectra, the peaks at 453.1 and 459.8 eV corresponded to the Ti3+ peaks of Ti3C2. The Zn 2p spectrum showed two peaks near 1044.8 and 1021.9 eV corresponding to Zn 2p3/2 and 2p1/2. These results all proved the successful preparation of Ti3C2/MoS2–ZnIn2S4.The efficiency of solar water evaporation systems depends not only on the loaded photothermal conversion material but also on the substrate material of the evaporation device. NSAs with high flexibility and hydrophilic properties were prepared from natural silk cocoons, as shown in Fig. 2a, in which PVA solution was inserted to enhance the mechanical properties and water transport of the polymer network. The NSAs loaded with different materials were obtained by the vertical freeze-drying method. The black Ti3C2/MoS2–ZnIn2S4 NSAs and white control groups (pure NSAs) are shown in Fig. 2b. It could be seen that the surface of NSAs loaded with Ti3C2/MoS2–ZnIn2S4 was wrinkled, compared to the surface of pure NSAs, which was porous and smooth. Moreover, Fig. 2c and S5† show the organ-like flakes of Ti3C2/MoS2–ZnIn2S4 successfully embedded on the surface of NSAs, which verified the feasibility of the synthesis experiment. Meanwhile, it could be seen in the SEM images that the NSAs maintained excellent porous morphology. Therefore, a stress–strain test was performed, as shown in Fig. 2d. When the NSAs were compressed to 60% of their starting height, the surface was able to withstand a pressure of 0.08 MPa, which showed that the NSAs had favorable mechanical properties and strength. As shown in Fig. 2e, dynamic contact angle tests were performed between water droplets and the surface of Ti3C2/MoS2–ZnIn2S4 NSAs. It was observed that Ti3C2/MoS2–ZnIn2S4 NSAs could be moistened within 6 s, revealing good hydrophilicity for excellent water transport. The above characterization studies have been conducted to ensure that the Ti3C2/MoS2–ZnIn2S4 NSAs can be applied properly in various environments.
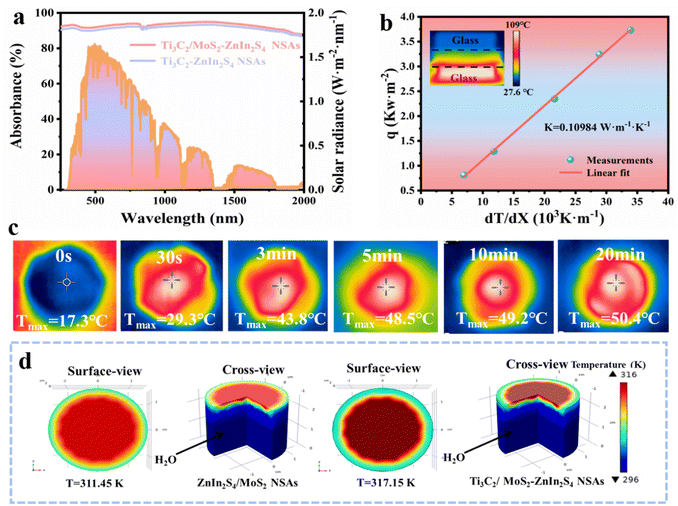
The photothermal effect was one of the factors for improving the activity of photocatalysts. In Fig. 3a, the light absorption abilities of the samples were investigated by ultraviolet-visible-near infrared (UV-vis-NIR) absorption spectroscopy. Ti3C2/MoS2–ZnIn2S4 NSAs exhibited intensive absorption throughout the entire spectrum in the region of 250–2000 nm, which exceeded 90%. The thermal dissipation of Ti3C2/MoS2–ZnIn2S4 NSAs is also depicted in Fig. 3b. Ti3C2/MoS2–ZnIn2S4 NSAs with a low thermal conductivity of 0.11 W m−1 K−1 can efficiently convert sunlight into thermal energy. The surface temperature variations of the samples under one sun illumination were conducted for exploration of the photothermal conversion ability (Fig. 3c, S5 and S6†). Ti3C2/MoS2–ZnIn2S4 NSAs could respond rapidly to temperature, rising from 17.3 to 50.4 °C in 20 minutes. The equilibrium temperature of Ti3C2/MoS2–ZnIn2S4 NSAs was much higher than those of ZnIn2S4/MoS2 NSAs and ZnIn2S4 NSAs. Simultaneously, COMSOL multiphysics simulations by building two heat transfer models for the surface and cross-section of Ti3C2/MoS2–ZnIn2S4 NSAs and ZnIn2S4/MoS2 NSAs were used to further verify the thermal management, as shown in Fig. 3d. Ti3C2/MoS2–ZnIn2S4 NSAs with excellent thermal localization performance undoubtedly favored the enhancement of vapor production.
2.2 Photocatalytic H2 generation performance
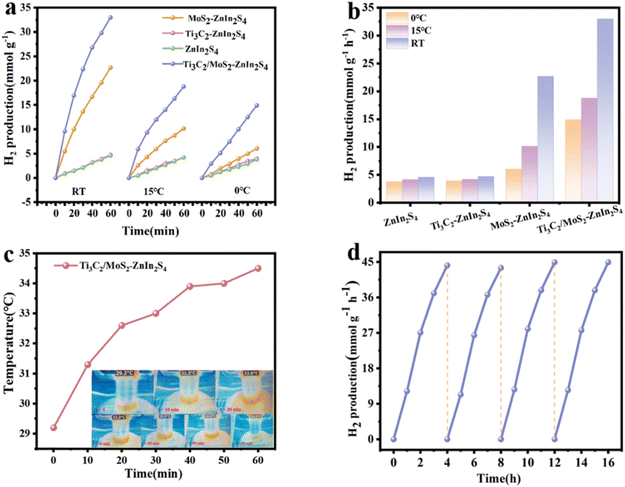
The synthesized photocatalysts were irradiated with AM 1.5 light to evaluate the H2 production performance in an experimental system with cooling water to control the temperature. As shown in Fig. 4a and b, when the temperature of the system was controlled at 0 °C, pristine ZnIn2S4 showed poor H2 evolution activity. It might be caused by the weak charge separation efficiency and low light absorption efficiency. After combining ZnIn2S4 with the co-catalyst MoS2 to form a heterogeneous structure, the photocatalytic hydrogen production activity was improved to 6.1 mmol g−1 h−1. Moreover, the Ti3C2/MoS2–ZnIn2S4 composite showed the highest H2 production of 14.9 mmol g−1 h−1, which was 3.9, 3.8 and 2.5 times higher than that of pure ZnIn2S4, Ti3C2/ZnIn2S4 and MoS2/ZnIn2S4. When the system temperature was higher than 0 °C, the H2 production rates of all four samples exhibited a large increase. Especially for the Ti3C2/MoS2–ZnIn2S4 composite, the average H2 production increased from 14.9 mmol g−1 h−1 at 0 °C to 33.0 mmol g−1 h−1 at an actual operating temperature of nearly 34.6 °C due to the photothermal effect, as shown in Fig. 4b (the RT in Fig. 4a and b represents the condition without temperature control). The apparent quantum efficiencies (AQEs) at 420 nm and 375 nm were 0.029% and 2.042%, respectively (Fig. S7†). AQE demonstrates a correlation with the characteristic absorption of ZnIn2S4, indicating that the photoelectrons essential for photocatalytic hydrogen production primarily originate from ZnIn2S4, while Ti3C2/MoS2 functions as a co-catalyst. The temperature change of the system under light exposure for 60 min was recorded by infrared thermography (Fig. 4c). It was shown that the photothermal effect led to an increase in the temperature of the system from 29.1 to a staggering 34.6 °C.Under light exposure, the hydrogen production of efficiency of the photocatalyst would be greatly promoted. In addition, the stability of Ti3C2/MoS2–ZnIn2S4 was evaluated by cycling experiments. After 4 cycles, the amount of photocatalytic H2 yield decreased slightly (Fig. 4d). The slight increase in hydrogen production efficiency from the third cycle test may be due to the photothermal evaporation effect, and the high temperature promotes the reaction. With the continuous evaporation of water in the reactor, the concentration of the reaction solution increases, so that the reaction rate is accelerated. In addition, we have carried out a long-term stability test of the sample, as shown in Fig. S8;† the hydrogen production rate of the sample left for 4 months and left for 2 months can reach 6.6687 mmol g−1 h−1 and 7.5082 mmol g−1 h−1 (temperature controlled at 0 °C). As the sulfide nature of our sample renders it susceptible to oxidation, which will inevitably compromise its efficiency over time. As shown in Fig. S9,† the hydrogen production rates of Ti3C2/MoS2–ZnIn2S4 powder (at 0 °C) and NSAs compared to other reported materials were demonstrated. The hydrogen production of this material is better than that of most of the materials reported in the literature. The hydrogen production rate of Ti3C2/MoS2–ZnIn2S4 NSAs under one sunlight was also detected by gas chromatography. Three-dimensional macroporous aerogels of Ti3C2/MoS2–ZnIn2S4 can produce hydrogen at a rate of 4.7 mmol g−1 h−1, and the hydrogen production efficiency on the aerogel surface with a radius of 1.8 cm could reach 374.2 μmol g−1 h−1 cm−2. The XRD and Raman patterns of the cycled Ti3C2/MoS2–ZnIn2S4 sample are shown in Fig S10.† The maintained original structure and composition reveal good structural robustness, and no signs of conversion of Ti3C2 to TiO2 were observed.
The photocatalytic mechanism can be verified by optical and photoelectrochemical tests. Based on this, we studied the separation and transport efficiency of photogenerated carriers of photocatalysts by photoluminescence (PL) spectroscopy and time-resolved photoluminescence attenuation spectroscopy (TRPL). As shown in Fig. 5a, all samples showed emission peaks around 560 nm. The pure ZnIn2S4 showed a rather high intensity of PL emission, due to the high recombination rate of photo-generated electron–hole pairs. When Ti3C2 and MoS2 were combined with ZnIn2S4, the PL intensity of the ternary composite photothermal-photocatalytic material decreased, following the order ZnIn2S4 > MoS2/ZnIn2S4 > Ti3C2/ZnIn2S4 > Ti3C2/MoS2–ZnIn2S4, which obviously indicates that the recombination of photogenerated charge carriers is significantly suppressed. This proved that the construction of ternary S-type heterojunctions significantly improved the separation and migration rate to suppress the generation of photogenerated charge carriers.49 The photogenerated carrier lifetimes were fitted by TRPL to analyze the kinetic behavior of photogenerated carriers in these composite photocatalysts. As shown in Fig. 5b, the average carrier lifetime of Ti3C2/MoS2–ZnIn2S4 was reduced to 1.6428 ns compared to pure ZnIn2S4, Ti3C2/ZnIn2S4, and MoS2/ZnIn2S4 (5.2284, 1.9400, and 2.0577 ns) by a biexponential kinetic function, and the specific fitting parameters are included in Table S1.† The significant reduction in the lifetime of photogenerated carriers may be due to the excellent conductivity of Ti3C2, which generates an interfacial electric field from MoS2 to MXene and then to the surface of ZnIn2S4, which effectively accelerates the transport and separation of carriers.50,51 To further investigate the charge transport and separation properties of the samples, transient photocurrent response (I−t) and electrochemical impedance spectroscopy (EIS) measurements were performed on ZnIn2S4, MoS2/ZnIn2S4, Ti3C2/ZnIn2S4, and Ti3C2/MoS2–ZnIn2S4 composites. In the EIS Nyquist results (Fig. 5c), the smaller the arc radius, the smaller the transport resistance of the carriers.52 It is obvious that Ti3C2/MoS2–ZnIn2S4 has a circle with the smallest radius, which indicates that due to the excellent conductivity of Ti3C2, the electron transport resistance of the composite is significantly reduced, and the transfer of photogenerated carriers is promoted.53 In Fig. 5d, all samples showed fast photocurrent responses. The current density of ZnIn2S4 is the lowest, which was due to severe photogenerated carrier recombination and poor visible light absorption. The highest photocurrent density belonged to Ti3C2/MoS2–ZnIn2S4, which was consistent with the PL results, implying a significant inhibition of charge recombination in the composite heterostructure.
Fig. 6a and b show high-resolution XPS spectra of Zn 2p, In 3d and Mo 3d of ZnIn2S4, MoS2–ZnIn2S4, and Ti3C2/MoS2–ZnIn2S4. Comparing MoS2–ZnIn2S4 composites with pure ZnIn2S4 and MoS2, there is no shift in the Zn 2p, In 3d and Mo 3d peak positions. The peaks of the MoS2–ZnIn2S4 composites in the XPS spectrum are largely unshifted, possibly due to a surface defect at the interface of the two phases that severely hinders the transfer of electrons as electron trapping centers.54,55 For Ti3C2/MoS2–ZnIn2S4 photocatalysts, the binding energies of Zn 2p and ln 3d elements shifted by −0.4 eV and −0.1 eV, respectively, compared to ZnIn2S4. Conversely, Fig. 6c shows that the binding energies of Mo 3d3/2 and 3d5/2 shifted from 232.3 eV and 229.1 eV to 232.8 eV and 229.6 eV, respectively. The observed shifts were attributed to the creation of Ti3C2 bridges and electron exchange from MoS2 to ZnIn2S4 at the heterointerfaces of Ti3C2/MoS2–ZnIn2S4 materials. Ti3C2 assisted the formation of an S-type heterojunction and accelerated the charge transfer between MoS2 and ZnIn2S4. More importantly, the close contact between Ti2C3/MoS2 and ZnIn2S4 effectively reduces the recombination of photoexcited carriers, resulting in more electrons participating in the reaction56 so that the recombination of photogenerated carriers at the interface was inhibited and the photogenerated electron transfer was promoted.57 As shown in Fig. 6d, the absorption edge of ZnIn2S4 alone was approximately 506 nm, which is located in the visible region. The absorption spectrum of Ti3C2/MoS2–ZnIn2S4 exhibited stronger absorption in the ultraviolet and visible regions, which confirms a significant increase in the light absorption capacity of the sample, resulting in a higher solar energy utilization rate.58 Moreover, the Tauc diagram obtained by the conversion of UV-vis diffuse absorption spectra using the Kubelka–Munk formula was used to determine the band gap (Eg) of photocatalysts.59 As shown in Fig. S11,† the band gap energies of Ti3C2/MoS2–ZnIn2S4 and Ti3C2/ZnIn2S4 were calculated to be 1.95 eV and 2.15 eV. Such a narrower band gap can provide a wider wavelength range and higher solar conversion efficiency for light absorption.60
The Mott–Schottky results are shown in Fig. S12.† Combined with the bandgap energy, we drew a band structure schematic illustration of MoS2–ZnIn2S4 and Ti3C2/MoS2–ZnIn2S4 in Fig. 6e and f, respectively. Based on these experimental data and theoretical analysis, we propose the photocatalytic mechanism and possible charge transfer mechanism at the interface of the Ti3C2/MoS2–ZnIn2S4 S-type heterojunction. Prior to the addition of Ti3C2, the charge transfer between MoS2–ZnIn2S4 was severely hampered. Moreover, the influence of weak redox ability and electrostatic repulsion also makes the recombination of photogenerated electrons and holes more serious. Ti3C2 MXene forms a 2D/2D van der Waals structure with MoS2, enabling robust interaction forces and substantial contact areas. Meanwhile, Ti3C2 MXene with good conductivity acts as an intermediate electron acceptor, facilitating the rapid transfer of interfacial charge from MoS2 to MXene and then to the ZnIn2S4 surface.56,61 Additionally, Ti3C2 MXene can form Schottky junctions between surfaces and other photocatalysts, generating a built-in electric field that prevents the recombination of photogenerated electrons and holes,62 and Ti3C2 can also be used as an ohmic hydrogen evolution cocatalyst to provide electron transfer pathways and active sites for photocatalytic hydrogen evolution.63 However, in this material system, ZnIn2S4 is the main light-absorbing material, and its photogenerated electrons react with water to produce hydrogen. Combined with the XPS and hydrogen production results, Ti3C2/ZnIn2S4 samples shows little improvement in hydrogen evolution performance, compared with pure ZnIn2S4. This indicates that Ti3C2 mainly aids in connecting two heterojunction components, thus creating better electron transport pathways. Furthermore, it enhances interfacial contact, ensuring close interaction among the three phases and reducing the recombination of photogenerated charge carriers. Increase the number of electrons involved in the reaction. Moreover, the compact structure formed by Ti3C2/MoS2 and ZnIn2S4 has been shown to be effective in reducing the recombination of photoexcited carriers, thereby increasing the number of electrons participating in the reaction. In addition, ZnIn2S4 with many active sites provides the necessary channels for electron transport in two-dimensional lamellae,64 allowing effective charge migration from the bulk phase to the surface.57 Upon the fabrication of the S-type heterostructure, the superior charge transfer properties and intimate interfacial contact of Ti3C2 facilitate electron transfer. Driven by the disparity in Fermi energy levels between ZnIn2S4 (RP) and MoS2 (OP), electrons migrate from ZnIn2S4 to MoS2, culminating in equilibrium. This electron flux engenders an electron-rich layer at the MoS2 interface and a hole-rich layer at the ZnIn2S4 interface,65,66 driving the generation of an intrinsic electric field from ZIS to MS. The accumulation of electrons and holes leads to the upward and downward bending of the energy bands of ZIS and MS, respectively. Under illumination, due to the action of the intrinsic electric field, band bending and Coulombic forces, the photogenerated holes with weak oxidizing ability in ZIS and the photogenerated electrons with weak reducing ability in MS recombine at the Ti3C2 interface. The photogenerated electrons and holes with strong redox ability are retained to participate in the reaction,67 so the photogenerated electrons in the ZnIn2S4 conduction band react with H2O in water to produce H2.
2.3 Solar evaporation
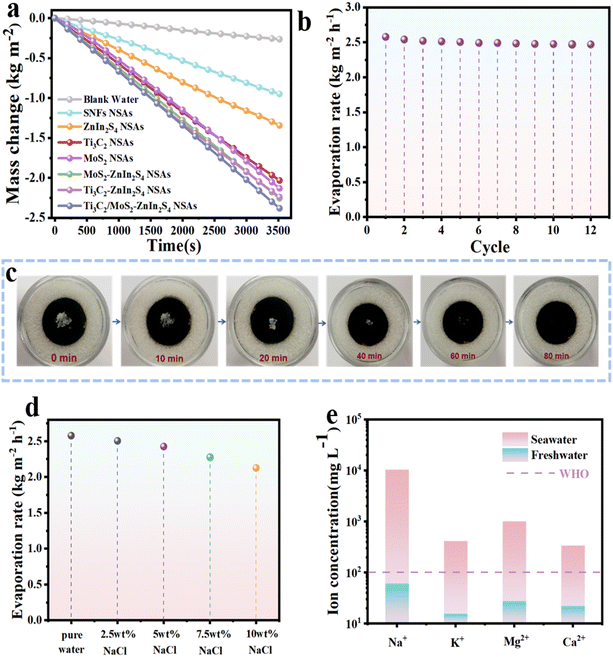
It is noteworthy that the clever combination of hydrophilic and photothermal properties in the as-prepared samples resulted in unique advantages for solar desalination. As shown in Fig. 7a, Ti3C2/MoS2–ZnIn2S4 NSAs delivered an evaporation rate of 2.6 kg m−2 h−1, which outperformed other samples. The corresponding photothermal conversion efficiency of Ti3C2/MoS2–ZnIn2S4 NSAs was calculated to be 94.2% (Note S1, ESI†). The evaporation rate of Ti3C2/MoS2–ZnIn2S4 NSAs remained around 2.5 kg m−2 h−1 under one sun illumination for 12 cycles, confirming the cycling stability of solar evaporation performance of Ti3C2/MoS2–ZnIn2S4 NSAs (Fig. 7b). Besides, 0.2 g of NaCl was placed on the surface of Ti3C2/MoS2–ZnIn2S4 NSAs and could be redissolved in solution after 60 minutes of light exposure, which was attributed to mighty water absorption and fast transport ability (Fig. 7c). For a more persuasive evaluation of the practical solar desalination capacity of Ti3C2/MoS2–ZnIn2S4 NSAs, the evaporation rates in low-salinity and high-salinity brine were tested as shown in Fig. 7d. Owing to the decline in vapor pressure under high salinity, the evaporation rates of Ti3C2/MoS2–ZnIn2S4 NSAs gradually decreased with the increase of the salinity, demonstrating the significant impact of salt concentration on the water evaporation.68 However, even in 10 wt% NaCl solution, the evaporation rate still reached 2.1 kg m−2 h−1. Meanwhile, the condensed water generated by Ti3C2/MoS2–ZnIn2S4 NSAs was collected, and the concentrations of the Na+, Mg2+, K+, and Ca2+ ions were detected with inductively coupled plasma atomic emission spectrometry (ICP-OES). The concentrations of the four cations decreased significantly, which complied with the World Health Organization (WHO) drinking water standards.To further investigate the freshwater and H2 generation capacity of Ti3C2/MoS2–ZnIn2S4 NSAs under a practical environment, outdoor experiments through a home-made device were performed on a sunny day, as shown in Fig. 8a. The surface temperature of Ti3C2/MoS2–ZnIn2S4 NSAs, solar radiation intensity, ambient temperature, and mass change were tracked every 30 min. The outdoor ambient temperature and solar intensity varied frequently. The maximum surface temperature was up to 54 °C at 2:30 PM, and the evaporation rate was 2.8 kg m−2 h−1. Under this environment, vapor was continuously produced from Ti3C2/MoS2–ZnIn2S4 NSAs and condensed in the top cover (Fig. S13a and b, ESI†). In addition, some H2 bubbles were adsorbed onto the top cover, as shown in Fig. S13c.† Thus, the developed Ti3C2/MoS2–ZnIn2S4 NSAs exhibited significant advantages and tremendous potential for efficient freshwater and H2 generation.
3. Conclusion
In summary, our investigation focuses on the organic and tight integration of photothermal conversion and photocatalytic hydrogen production using solar interfacial water evaporation technology to maximize the utilization and conversion of renewable energy through solar evaporation and water-splitting energy production strategies. In this work, we have developed Ti3C2/MoS2–ZnIn2S4 NSAs as solar evaporators to enhance vapor generation. The NSAs have favorable mechanical properties and low thermal conductivity (0.11 W m−1 K−1 in the dry state). A high evaporation rate of up to 2.6 kg m−2 h−1 of Ti3C2/MoS2–ZnIn2S4 NSAs under one sun was achieved, yielding a corresponding photothermal conversion efficiency of 93.4%. In addition, Ti3C2 not only promotes the construction of MoS2–ZnIn2S4 S-scheme heterojunctions, which accelerate the separation of photogenerated electron–hole pairs, but also significantly enhances the transference of photogenerated electrons due to its excellent conductivity and thus significantly improves the efficiency of hydrogen precipitation evolution. The H2 yield of Ti3C2/MoS2–ZnIn2S4 significantly increases to 33.0 mmol g−1 h−1 at RT. The Ti3C2/MoS2–ZnIn2S4 NSA dual-function system designed in this study has good adaptability and hydrogen production potential and offers great potential for designing efficient water cracking photocatalysts for solar desalination and hydrogen precipitation. The findings of the present study indicate that, to realize an efficient solar hydrogen and water production bifunctional device, it is necessary to design the catalyst material with a suitable bandgap photocatalyst, as well as a narrow bandgap co-catalyst with photothermal properties. Furthermore, it is desirable to design the interface of this heterojunction to enhance the carrier separation at the interface at high temperatures.Data availability
The data supporting this article have been included as part of the ESI.†Conflicts of interest
There are no conflicts to declare.Acknowledgements
This work was financially supported by the National Natural Science Foundation of China (Grant No. 52406237), the Wuhan Science and Technology Bureau of China (Grant No. 2018010401011280), and the Department of Science and Technology of Hubei Province, China (No. 2024BAB096).References
- S. Sun, X. Zhou, H. Liu, Y. Jiang, H. Zhou, C. Zhang and G. Fu, Water Res., 2021, 194, 116931 CrossRef CAS
.
- M. T. H. van Vliet, E. R. Jones, M. Flörke, W. H. P. Franssen, N. Hanasaki, Y. Wada and J. R. Yearsley, Environ. Res. Lett., 2021, 16, 024020 CrossRef CAS
.
- C. J. Vörösmarty, P. B. McIntyre, M. O. Gessner, D. Dudgeon, A. Prusevich, P. Green, S. Glidden, S. E. Bunn, C. A. Sullivan, C. R. Liermann and P. M. Davies, Nature, 2010, 467, 555–561 CrossRef
.
- G. Ni, H. Zandavi, M. Javid, S. Boriskina, T. Cooper and G. Chen, Energy Environ. Sci., 2018, 11, 1510–1519 RSC
.
- X. Wu, G. Y. Chen, G. Owens, D. Chu and H. Xu, Mater. Today Energy, 2019, 12, 277–296 CrossRef
.
- J. Huang, H. Zheng and H. Kong, Energy Convers. Manage., 2024, 299, 117806 CrossRef
.
- J. Liu, S. Zhang, J. Wang and Q. Lan, Desalination, 2025, 597, 118356 CrossRef CAS
.
- X. Wu, Y. Lu, X. Ren, P. Wu, D. Chu, X. Yang and H. Xu, Adv. Mater., 2024, 36, 2313090 CrossRef
.
- V.-H. Dang, T.-A. Nguyen, M.-V. Le, D. Q. Nguyen, Y. H. Wang and J. C. S. Wu, Chem. Eng. J., 2024, 484, 149213 CrossRef CAS
.
- L. Guo, Z. Yang, K. Marcus, Z. li, B. Luo, L. Zhou, X. Wang, Y. Du and Y. Yang, Energy Environ. Sci., 2018, 11, 106–114 RSC
.
- M. Liu, Y. Chen, J. Su, J. Shi, X. Wang and L. Guo, Nat. Energy, 2016, 1, 16151 CrossRef CAS
.
- Q. Wang, T. Hisatomi, Q. Jia, H. Tokudome, M. Zhong, C. Wang, Z. Pan, T. Takata, M. Nakabayashi, N. Shibata, Y. Li, I. D. Sharp, A. Kudo, T. Yamada and K. Domen, Nat. Mater., 2016, 15, 611–615 CrossRef CAS
.
- G. H. Timmermans, S. Hemming, E. Baeza, E. A. J. van Thoor, A. P. H. J. Schenning and M. G. Debije, Adv. Opt. Mater., 2020, 8, 2000738 CrossRef CAS
.
- J. Lu, Y. Shi, Z. Chen, X. Sun, H. Yuan, F. Guo and W. Shi, Chem. Eng. J., 2023, 453, 139834 CrossRef CAS
.
- D. Aguilar-Ferrer, T. Vasileiadis, I. Iatsunskyi, M. Ziółek, K. Żebrowska, O. Ivashchenko, P. Błaszkiewicz, B. Grześkowiak, R. Pazos, S. Moya, M. Bechelany and E. Coy, Adv. Funct. Mater., 2023, 33, 2304208 CrossRef CAS
.
- H. Han, K. Huang, Y. Yao, Z. Li and X. Meng, Chem. Eng. J., 2022, 450, 138419 CrossRef CAS
.
- W. Xiong, Y. Chen, M. Hao, L. Zhang, T. Mei, J. Wang, J. Li and X. Wang, Appl. Therm. Eng., 2015, 91, 630–637 CrossRef CAS
.
- J. Bai, W. Chen, L. Hao, R. Shen, P. Zhang, N. Li and X. Li, Chem. Eng. J., 2022, 447, 137488 CrossRef CAS
.
- S. Guo, X. Li, J. Li and B. Wei, Nat. Commun., 2021, 12, 1343 CrossRef CAS
.
- P. Liu, Y.-b. Hu, X.-Y. Li, L. Xu, C. Chen, B. Yuan and M.-L. Fu, Angew. Chem., Int. Ed., 2022, 61, e202208587 CrossRef CAS
.
- D. Ghim, Q. Jiang, S. Cao, S. Singamaneni and Y.-S. Jun, Nano Energy, 2018, 53, 949–957 CrossRef CAS
.
- Z. Wang, W. Xu, K. Yu, S. Gong, H. Mao, R. Huang and Z. Zhu, Sol. RRL, 2021, 5, 2100183 CrossRef CAS
.
- J. Xiao, Y. Guo, W. Luo, D. Wang, S. Zhong, Y. Yue, C. Han, R. Lv, J. Feng, J. Wang, W. Huang, X. Tian, W. Xiao and Y. Shen, Nano Energy, 2021, 87, 106213 CrossRef CAS
.
- Z. Guo, Z. Chen, Z. Shi, J. Qian, J. Li, T. Mei, J. Wang, X. Wang and P. Shen, Sol. Energy Mater. Sol. Cells, 2020, 204, 110227 CrossRef CAS
.
- X. Yang, Y. Yang, L. Fu, M. Zou, Z. Li, A. Cao and Q. Yuan, Adv. Funct. Mater., 2018, 28, 1704505 CrossRef
.
- J. Low, J. Yu, M. Jaroniec, S. Wageh and A. A. Al-Ghamdi, Adv. Mater., 2017, 29, 1601694 CrossRef
.
- J. Low, C. Jiang, B. Cheng, S. Wageh, A. A. Al-Ghamdi and J. Yu, Small Methods, 2017, 1, 1700080 CrossRef
.
- Q. Xu, L. Zhang, B. Cheng, J. Fan and J. Yu, Chem, 2020, 6, 1543–1559 CAS
.
- K. Maeda, ACS Catal., 2013, 3, 1486–1503 CrossRef CAS
.
- R. Yang, L. Mei, Y. Fan, Q. Zhang, R. Zhu, R. Amal, Z. Yin and Z. Zeng, Small Methods, 2021, 5, 2100887 CrossRef CAS PubMed
.
- L. Wang, B. Cheng, L. Zhang and J. Yu, Small, 2021, 17, 2103447 CrossRef CAS PubMed
.
- L. Zhang, J. Zhang, H. Yu and J. Yu, Adv. Mater., 2022, 34, 2107668 CrossRef CAS
.
- Y.-P. Zhang, W. Han, Y. Yang, H.-Y. Zhang, Y. Wang, L. Wang, X.-J. Sun and F.-M. Zhang, Chem. Eng. J., 2022, 446, 137213 CrossRef CAS
.
- B. Zhu, J. Liu, J. Sun, F. Xie, H. Tan, B. Cheng and J. Zhang, J. Mater. Sci. Technol., 2023, 162, 90–98 CrossRef CAS
.
- B. Zhu, J. Sun, Y. Zhao, L. Zhang and J. Yu, Adv. Mater., 2024, 36, 2310600 CrossRef CAS
.
- Z. Jin and Y. Wu, Appl. Catal., B, 2023, 327, 122461 CrossRef CAS
.
- B. He, P. Xiao, S. Wan, J. Zhang, T. Chen, L. Zhang and J. Yu, Angew. Chem., Int. Ed., 2023, 62, e202313172 CrossRef CAS
.
- M. Gu, Y. Yang, B. Cheng, L. Zhang, P. Xiao and T. Chen, Chin. J. Catal., 2024, 59, 185–194 CrossRef CAS
.
- Z. Jin, H. Li and J. Li, Chin. J. Catal., 2022, 43, 303–315 CrossRef CAS
.
- Z. Kong, D. Zhang, J. Liu, X.-Y. Ji, P. Cai, X. Pu and H. Zhang, Nano Energy, 2025, 138, 110890 CrossRef CAS
.
- M.-Y. Ma, H.-Z. Yu, L.-M. Deng, L.-Q. Wang, S.-Y. Liu, H. Pan, J.-W. Ren, M. Y. Maximov, F. Hu and S.-J. Peng, Tungsten, 2023, 5, 589–597 CrossRef
.
- Y. Wei, Y. Wu, J. Wang, Y.-H. Wu, Z. Weng, W.-Y. Huang, K. Yang, J.-L. Zhang, Q. Li, K.-Q. Lu and B. Han, J. Mater. Chem. A, 2024, 12, 18986–18992 RSC
.
- J. Yu, P. Su, D. Zhang, H. Zhao, N. Yang, T. Liang, D. Zhang and X. Pu, Sep. Purif. Technol., 2025, 354, 128694 CrossRef CAS
.
- R. Zhang, R. Liu, Z. Ding, J. Ma, T. Wang, D. Zhang, J. Liu, P. Cai and X. Pu, J. Colloid Interface Sci., 2025, 682, 568–577 CrossRef CAS
.
- J. Pang, R. G. Mendes, A. Bachmatiuk, L. Zhao, H. Ta, T. Gemming, H. Liu, Z. Liu and M. Rümmeli, Chem. Soc. Rev., 2019, 48, 72–133 RSC
.
- Z. Guo, W. Zhou, N. Arshad, Z. Zhang, D. Yan, M. S. Irshad, L. Yu and X. Wang, Carbon, 2022, 186, 19–27 CrossRef CAS
.
- Q. Wang, S. Yan, G. Han, X. Li, R. You, Q. Zhang, M. Li and D. L. Kaplan, Compos. Sci. Technol., 2020, 187, 107950 CrossRef CAS
.
- A. Feng, Y. Yu, Y. Wang, F. Jiang, Y. Yu, L. Mi and L. Song, Mater. Des., 2017, 114, 161–166 CrossRef CAS
.
- S. Wan, W. Wang, B. Cheng, G. Luo, Q. Shen, J. Yu, J. Zhang, S. Cao and L. Zhang, Nat. Commun., 2024, 15, 9612 CrossRef CAS PubMed
.
- L. Bi, R. Zhang, K. Zhang, Y. Lin, D. Wang, X. Zou and T. Xie, ACS Sustainable Chem. Eng., 2019, 7, 15137–15145 CrossRef CAS
.
- T. Jiang, T. Xie, L. Chen, Z. Fu and D. Wang, Nanoscale, 2013, 5, 2938–2944 RSC
.
- P. Wang, Z. Shen, Y. Xia, H. Wang, L. Zheng, W. Xi and S. Zhan, Adv. Funct. Mater., 2019, 29, 1807013 CrossRef
.
- K. Huang, C. Li and X. Meng, J. Colloid Interface Sci., 2020, 580, 669–680 CrossRef CAS PubMed
.
- J. Zhang, J. Cheng, M. Song, H. Song, Y. Wang, S.-F. Yin and P. Chen, Chem. Eng. J., 2024, 496, 153713 CrossRef CAS
.
- N. Zhang, Z. Zhai, Y. Yan, X. Feng, S. Chen, Y. Zhao and B. Zhang, Sep. Purif. Technol., 2025, 362, 131694 CrossRef CAS
.
- J. Ran, G. Gao, F.-T. Li, T.-Y. Ma, A. Du and S.-Z. Qiao, Nat. Commun., 2017, 8, 13907 CrossRef CAS
.
- T. Su, C. Men, L. Chen, B. Chu, X. Luo, H. Ji, J. Chen and Z. Qin, Adv. Sci., 2022, 9, 2103715 Search PubMed
.
- Y. Li, J. Ma, L. Xu, T. Liu, T. Xiao, D. Chen, Z. Song, J. Qiu and Y. Zhang, Adv. Sci., 2023, 10, 2207514 CrossRef CAS
.
- L. Zeng, F. Zhe, Y. Wang, Q. Zhang, X. Zhao, X. Hu, Y. Wu and Y. He, J. Colloid Interface Sci., 2019, 539, 563–574 CrossRef CAS
.
- J. Xu, J. Shen, H. Jiang, X. Yu, W. Ahmad Qureshi, C. Maouche, J. Gao, J. Yang and Q. Liu, J. Ind. Eng. Chem., 2023, 119, 112–129 CrossRef CAS
.
- H. Zeng, Z. Li, G. Li, X. Cui, M. Jin, T. Xie, L. Liu, M. Jiang, X. Zhong, Y. Zhang, H. Zhang, K. Ba, Z. Yan, Y. Wang, S. Song, K. Huang and S. Feng, Adv. Energy Mater., 2022, 12, 2102765 CrossRef CAS
.
- Y. Yang, Z. Zeng, G. Zeng, D. Huang, R. Xiao, C. Zhang, C. Zhou, W. Xiong, W. Wang, M. Cheng, W. Xue, H. Guo, X. Tang and D. He, Appl. Catal., B, 2019, 258, 117956 CrossRef CAS
.
- X. Qin, L. Ji and A. Zhu, Colloids Surf., A, 2023, 676, 132198 CrossRef CAS
.
- Y. Xu, A. Yan, L. Jiang, F. Huang, D. Hu, G. Duan and F. Zheng, J. Alloys Compd., 2022, 895, 162504 CrossRef CAS
.
- J. Li, J. Yao, Q. Yu, X. Zhang, S. A. C. Carabineiro, X. Xiong, C. Wu and K. Lv, Appl. Catal., B, 2024, 351, 123950 CrossRef CAS
.
- J. Li, X. Zhang, X. Xiong, C. Wu, Y. Jin and K. Lv, Sep. Purif. Technol., 2025, 354, 129444 CrossRef CAS
.
- Y. Xu, Q. Liu, J. Zhu, H. Zhang, J. Liu, R. Chen, J. Yu, Y. Li and J. Wang, Chem. Eng. J., 2022, 435, 134958 CrossRef CAS
.
- Y. Tang, N. Wang, Y. He, Y. Wang, Y. Shan, H. Zhang and Y. Sun, J. Hydrol., 2023, 617, 129033 CrossRef
.
Footnotes |
| † Electronic supplementary information (ESI) available. See DOI: https://doi.org/10.1039/d5ta00933b |
| ‡ Authors contributed equally to this work. |
| This journal is © The Royal Society of Chemistry 2025 |