A transparent, tough, highly stretchable and self-adhesive zwitterionic dual-network eutectogel for wearable flexible sensors†
Zhuangzhuang
Ma
a,
Jiale
Zhang
a,
Zelin
Ma
b,
Minghao
Lou
a,
Peijin
Zou
a,
Hongqiang
Wang
 *b and
Lichao
Jia
*a
*b and
Lichao
Jia
*a
aKey Laboratory of Applied Surface and Colloid Chemistry, National Ministry of Education, Shaanxi Key Laboratory for Advanced Energy Devices, Shaanxi Engineering Lab for Advanced Energy Technology, School of Materials Science and Engineering, Shaanxi Normal University, 620 West Chang'an Street, Xi'an, Shaanxi 710119, China. E-mail: lichaojia@snnu.edu.cn
bState Key Laboratory of Solidification Processing, Center for Nano Energy Materials, School of Materials Science and Engineering, Northwestern Polytechnical University, Shaanxi Joint Labortary of Graphene, Xi'an, 710072, P. R. China. E-mail: hongqiang.wang@nwpu.edu.cn
First published on 27th February 2025
Abstract
Eutectogels possess significant potential for use in wearable flexible sensors due to their low volatility, chemical stability, and high ionic conductivity. However, most eutectogels face challenges such as intransparency, non-adhesion, and limited mechanical strength, which hinder their practical applications. In this study, we designed and successfully constructed a novel zwitterionic dual-network (DN) eutectogel to address these limitations and analysed the interactions within the polymer by molecular dynamics. This eutectogel comprises a pre-polymerized network of [2-(methacryloyloxy)ethyl]dimethyl-(3-sulfopropyl) (DMAPS) and physically crosslinked acrylic acid-N-(2-hydroxyethyl)acrylamide copolymers (p(AA-co-HEMAA)). The DN eutectogels exhibit exceptional properties, including high strength (0.569 MPa), elongation at break (637%), toughness (2.529 MJ m−3), and high transparency (∼89%). Additionally, they possess good adhesion and stability. Flexible sensors with high sensitivity, a wide detection range, and excellent stability were fabricated from these eutectogels and tested with various applications, including limb motion detection, handwriting recognition, and Morse code translation. This study not only showcases a high-performance eutectogel-based sensing material but also paves the way for the development of multifunctional wearable devices in the future.
1. Introduction
Recently, flexible sensors have garnered increasing research attention and hold significant promise across various domains, including wearable sensing,1 smart medicine,2 soft robotics,3 and human–computer interaction.4 Typically, hydrogel-based sensors are hindered by inherent issues such as freezing at low temperatures, water evaporation at high temperatures, and limited long-term durability.5 Nevertheless, ionogel-based strain sensors have attracted significant interest due to their distinctive three-dimensional structure, high permeability, ionic conductivity, and compatible mechanical properties with various biological tissues.6 However, the typically high cost and toxicity of ionic liquids greatly limit their widespread use.7,8 Therefore, developing a novel ionic conductive gel material that combines affordability, high stretchability, non-toxicity, and environmental resilience remains a critical challenge.Deep eutectic solvents (DESs) are typically formed when hydrogen bond acceptors (HBAs, e.g., choline chloride) and hydrogen bond donors (HBDs, e.g., ethylene glycol) are mixed at specific molar ratios through extensive hydrogen bonding interactions, yielding eutectic mixtures with depressed melting points.9 These eutectogels are not only safe, low cost, and environmentally friendly, but also exhibit low volatility and excellent ionic conductivity. Consequently, they are an ideal alternative to expensive ionogels and temperature-resistant hydrogels, making them a hot research topic.10 By adjusting the chemical structures and proportion of DES and gelling agents, the key properties of eutectogels, such as ionic conductivity, mechanical properties, shape memory and self-healing properties can be easily modulated.11 However, the single network structures with poor mechanical properties significantly hinder their application.12
Since the pioneering work on polymeric DN gel structures reported by Gong et al., considerable attention has been paid to optimizing DN gels to enhance their mechanical strength and toughness.13 For example, Ko et al. discovered that a DN hydrogel based on polyacrylamide (AAm) and sodium alginate exhibited fast rebound and no hysteresis.14 Liu et al. formed a DN structure by combining carboxymethyl chitosan (CMC) with acrylic acid (AA), which demonstrated excellent self-healing (74.3% recovery of tensile strength after 1 hour) and tunable mechanical properties.15 Luo et al. physically crosslinked polyvinyl alcohol (PVA) with p(AM-co-AA) copolymers, resulting in excellent mechanical properties (2.1 MPa tensile stress, 6.5 MJ m−3 toughness, 0.4 MPa elastic modulus) and strong adhesion to various material surfaces.16 These findings indicate the feasibility of constructing a dual network structure to enhance the mechanical properties of gels. However, studies on eutectogels based on DN structures are rarely reported.
[2-(Methacryloyloxy)ethyl]dimethyl-(3-sulfopropyl) (DMAPS) as a typical zwitterionic compounds have received much attentions for their application in gel materials due to their unique molecular structure and multifunctional properties. Usually, zwitterionic compounds contain both positively charged quaternary ammonium groups and negatively charged sulfonic acid groups, the balanced distribution of charges within the molecule endows them excellent anti-freezing, anti-drying, and low bioadhesion properties. At the same time, these ionic groups can form dynamic cross-links with the functional groups in the polymer network through electrostatic interaction and hydrogen bonding, potentially enhancing the mechanical strength and tensile properties of the gel. In addition, the hydrophilicity and ionic conductivity of the zwitterionic compounds could help to improve the environmental adaptability and electrical properties of the gels, making them an ideal candidate for flexible electronics.17–19 At present, zwitterionic compounds, especially DMAPS, perform well in hydrogels. However, only a few studies have been applied to DN eutectogels, and the corresponding principle in enhancing eutectogel performance have not yet been clarified.
In this study, a zwitterionic DN eutectogel based on choline chloride–ethylene glycol ([ChCl][EG]) deep eutectic solvent was successfully synthesized, achieving transparency, strong adhesion, and mechanical strength. And the intra-polymer interactions were simulated with molecular dynamics. The DN eutectogel exhibited high strength (0.569 MPa) and elongation at break (637%), strong toughness (2.529 MJ m−3), high transparency (∼89%), good adhesion, and long-term stability. Furthermore, the DN eutectogel was utilized as a flexible sensor for applications such as limb motion detection, handwriting recognition, and Morse code translation. This work demonstrates a high-performance eutectogel-based sensing material and introduces a novel approach for the modification of eutectogels.
2. Experimental section
2.1 Chemicals
The reagents used in the experiments were analytically pure and used without further purification. Choline chloride (ChCl, C5H14ClNO, 98%), ethylene glycol (EG, C2H6O2, 98%), acrylic acid (AA, C3H4O2, 99%), N-(2-hydroxyethyl)acrylamide (HEMAA, C5H9NO2, 98%) and [2-(methacryloyloxy)ethyl]dimethyl-(3-sulfopropyl) (DMAPS, C11H21NO5S, 97%) were provided by Aladdin Biochemical Co. Ltd, (Shanghai, China). N,N-Methylenebis(acrylamide) (MBAA, C7H10N2O2, 99%) and 2-hydroxy-4′-(2-hydroxyethoxy)-2-methylpropiophenone (I2959, C12H16O4, 98%) were purchased from Macklin Biochemical Co. Ltd, (Shanghai, China).2.2 Synthesis of deep eutectic solvents
The deep eutectic solvents were prepared similarly to those previously reported in the literature.20 The required amount of ChCl (13.96 g, 0.1 mol) was dried under vacuum for 2 hours to remove the water (the remaining ChCl had to be placed back in the drying box to prevent ChCl from getting wet), then quickly mixed with EG (12.41 g, 0.1 mol) in a nitrogen purge atmosphere and stirred for 1 hour at 80 °C to form a homogeneous, colorless liquid, which is the deep eutectic solvent (during this time the whole system was sealed to ensure that it would not get wet). Finally, the resulting deep eutectic solvent was placed in a vacuum oven (vacuum: −0.1 MPa, ambient temperature: 25 °C, ambient humidity: 30% RH). It is important to note that after each use of the deep eutectic solvent, the solution needs to be purged with nitrogen and sealed again before placing in the vacuum oven to ensure the stability of the deep eutectic solvent.2.3 Synthesis of pDMAPS/p(AA-co-HEMAA) DN eutectogels
The different mass fractions (1 wt%, 2 wt%, 5 wt% and 10 wt%) of DMAPS monomers were dissolved in 2.56 mL [ChCl][EG], respectively. Then 0.0045 g I2959 was added to each of the above solutions and stirred for 1 h until fully dissolved. Subsequently, they were placed under UV light (365 nm, 40 W) for 1 h to polymerize the solutions into dispersions with different pDMAPS contents. Then 2.56 mL AA monomer, 1.58 mL HEMAA monomer, 0.0087 g MBAA and 0.0875 g I2959 were added one by one in the dispersions with different pDMAPS contents and stirred overnight to fully dissolve them. Next, the above solutions were transferred into customized glass molds and placed under 365 nm UV light (365 nm, 40 W) for 30 min to construct pDMAPS/p(AA-co-HEMAA) DN eutectogels (AHD eutectogels). The eutectogels synthesized with different mass fractions (1 wt%, 2 wt%, 5 wt%, and 10 wt%) of DMAPS monomers were named as A6H4D1, A6H4D2, A6H4D5, and A6H4D10, respectively. The pAA eutectogels (AA eutectogels) and p(AA-co-HEMAA) eutectogels (AH eutectogels) were prepared with the similar method as pDMAPS/p(AA-co-HEMAA) eutectogels (see the detail in ESI†). The pAA eutectogels with different AA contents (30 wt%, 40 wt%, 50 wt%, and 60 wt%) were named as AA3, AA4, AA5, and AA6, respectively. The p(AA-co-HEMAA) eutectogels with different AA to HEMAA mass ratios (10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0, 8
0, 8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2, 6
2, 6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4, 4
4, 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 6, 2
6, 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 8, and 0
8, and 0![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10) were named as AA, A8H2, A6H4, A4H6, A2H8 and HEMAA.
10) were named as AA, A8H2, A6H4, A4H6, A2H8 and HEMAA.
2.4 Characterization
The transmittance of the eutectogels was measured using a UV-Vis spectrophotometer (Lambda 950, PerkinElmer, USA) in the range of 400–800 nm. The in situ morphology of the eutectogels were examined by a tungsten filament scanning electron microscope (SU3050, Hitachi, Japan) and a field emission scanning electron microscope (SU8020, Hitachi, Japan). The structure and composition of the eutectogels were operated using a high-resolution X-ray diffractometer (Smartlab9, Japan, Cu Kα1 radiation, λ = 0.15406 nm) and Fourier-transform infrared spectrometer (Tensor II, Bruker, Germany) with attenuated total reflectance (ATR) mode. Chemical state of the eutectogels were examined with X-ray photoelectron spectra (XPS, Kratos Analytical, AXIS ULTRA, Japan). The small-angle X-ray scattering (SAXS) and wide-angle X-ray scattering (WAXS) data of the eutectogels were collected on the SAXS beamline (Xeuss 3.0, France). Dynamic mechanical analysis was performed on a TA Instrument (DMA Q850, Artemis). The frequency sweep ranged from 0 to 20 Hz with a strain of 1%. The AA, HEMAA and DMAPS was constructed with Materials Studio, ver. 2019. The calculation of surface electrostatic potential (ESP) was performed in the DMol3 module.2.5 Adhesion testing of eutectogels
A piece of eutectogel (1.0 cm × 1.0 cm × 0.1 cm) was sandwiched between two pieces of aluminum, copper, glass, wood, plastic and ceramic, respectively, and pressed with a 100 g weight for 10 min to make full contact. Then, the adhesion strength of the gel was measured by a universal testing machine (990B, Dongguan Zhiqu Precision Instruments Co., Ltd) with a tensile speed of 2 mm min−1. Finally, the adhesion strength was obtained by calculating the maximum force divided by the overlapping contact area.2.6 Mechanical testing of eutectogels
A universal testing machine (990B, Dongguan Zhiqu Precision Instrument Co., Ltd) with a 500 N transducer was used to evaluate the mechanical properties of the eutectogel. The size of the samples was 3.0 cm × 1.0 cm × 0.1 cm, and each sample was tested at least three times with the tensile rate of 100 mm min−1. The tensile stress (σn) in the test was calculated by the following equation: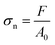 | (1) |
The fracture elongation (ε) of the experimental co-polymerized gel was calculated by the following equation:
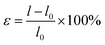 | (2) |
2.7 Ionic conductivity testing of eutectogels
The ionic conductivity of the eutectogel was tested using an electrochemical workstation (CHI660E, Shanghai CH Instruments Co., Ltd). The size of the sample was 1.0 cm × 1.0 cm × 0.1 cm. Stainless steel sheets were adhered to the upper and lower ends of the sample to reduce the contact resistance. The electrochemical impedance spectra (EIS) was measured by holding the upper and lower sides of the eutectogel in the frequency range of 0.1 Hz to 100 kHz. The ionic conductivity (σe) of the sample was calculated by the following equation: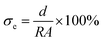 | (3) |
2.8 Sensing performance of eutectogels
The response of the eutectogel-based sensors in tension/compression was evaluated by connecting copper wires at both ends of the eutectogel using a universal testing machine (990B, Dongguan Zhiqu Precision Instruments Co., Ltd) in combination with a source measurement unit (B2912A, Keysight, USA). The response value (ΔR/R0) of the eutectogel-based sensor was calculated by the following equation: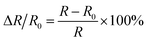 | (4) |
The gauge factor (GF) of the eutectogel-based sensor was calculated by the following equation:
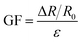 | (5) |
The pressure sensitivity (S) was defined as:
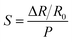 | (6) |
The human motion detection experiments were conducted in compliance with The Code of Ethics of the World Medical Association (Declaration of Helsinki). All experiments involving human participants were reviewed and approved by the Human Research Ethics Committee of Shaanxi Normal University. Informed consent was obtained from each participant prior to the study.
2.9 Biosafety assessment
An in vitro cytotoxicity assessment was conducted using mouse fibroblasts (L929). Cells were cultured in DMEM medium supplemented with 10% fetal bovine serum (FBS) at 37 °C under a 5% CO2 atmosphere. A6H4D5 eutectogel samples, sterilized by UV irradiation for 30 minutes, were co-cultured with L929 cells at varying concentrations (0%, 25%, 50%, 75%, and 100%) for 24 hours. For MTT assay, logarithmic-phase L929 cells were adjusted to a density of 5 × 103 cells per well in 96-well plates. After 24 hours of incubation with the A6H4D5 eutectogel extracts, the culture medium was replaced with 100 μL of fresh medium containing 10% MTT reagent, followed by 4 hours of incubation under the same conditions. Additionally, after co-culturing the extracts with L929 cells (8 × 104 cells per mL) for 24 h, live/dead staining was performed using the BB-4126 kit (Bestbio Biotech, China). Cells were imaged directly via laser scanning confocal microscopy (Leica TCS SP8), where viable cells fluoresced green (calcein AM) and dead cells fluoresced red (propidium iodide). Experimental parameters, including cell density, incubation time, and MTT protocol, were strictly aligned with standardized cytotoxicity testing guidelines to ensure reproducibility.3. Results and discussion
3.1 Material characterization
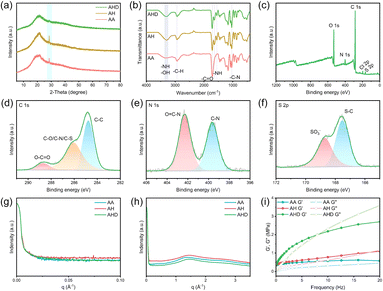
Fig. 1(a) depicts the preparation procedure of AHD eutectogel. Specifically, the DMAPS monomer was first dissolved in [ChCl][EG] and polymerized under 365 nm UV light to form pDMAPS polymer chains. Due to the interaction between the reactive groups in the pDMAPS molecule and [ChCl][EG], the pDMAPS polymer chains stabilized in the solvent, forming a homogeneous dispersion. Subsequently, AA and HEMAA monomers were added to the solution and polymerized to form p(AA-co-HEMAA) copolymer chains.21–23 During polymerization, the acryloyl and sulfonylpropyl side chains of pDMAPS cross-link with the copolymer chains via MBAA, forming a three-dimensional network structure.24,25 In this system, the AA block in p(AA-co-HEMAA) increases the rigidity and stability of the eutectogel due to its higher polarity, while the HEMAA block enhances flexibility and elasticity owing to the presence of the hydroxyethyl side chain.26 Additionally, the pDMAPS polymer chains ensure the flexibility of the eutectogel.27 The electrostatic potential distribution (ESP) analysis of AA, HEMAA, and DMAPS monomers was conducted using the DMol3 module in MS software as shown in Fig. 1(b). Evidently, DMAPS strongly interacts with AA and HEMAA due to its unique zwitterionic character. Moreover, molecular dynamics simulations of the gels using the Forcite module in MS software revealed that a dense hydrogen bonding network was formed between DES and DMAPS/AA-HEMAA (Fig. S1†). Specifically, the quaternary ammonium group in ChCl (a hydrogen bond acceptor) and the hydroxyl group of EG (a hydrogen bond donor) interacted via hydrogen bonds with the sulfonic acid group of DMAPS as well as the carboxyl, hydroxyl, and amide groups of AA-HEMAA. These multidirectional hydrogen bonds not only enhance the interfacial bonding between the polymer chains and DES, but also build a dense three-dimensional cross-linked network. Under external forces, the hydrogen bonds reversibly break to dissipate energy while dynamically reconfiguring to maintain the integrity of the network, thus significantly enhancing the toughness of the eutectogels.28,29 The high miscibility of p(AA-co-HEMAA) and pDMAPS in [ChCl][EG] ensures excellent homogeneity, resulting in outstanding optical transparency of the eutectogel (Fig. S2†). As shown in Fig. 1(c), the AHD eutectogel exhibits high transparency in the visible region (400–800 nm) with an average transmittance of 89%. When the eutectogel is placed over white paper printed with “SNNU”, the text paper remains clearly visible, demonstrating its exceptional permeability. Furthermore, the prepared eutectogel exhibits excellent tensile and mechanical properties during twisting and stretching experiments (Fig. 1(d)).The structures of AA eutectogel, AH eutectogel, and AHD eutectogel were characterized by X-ray diffraction (XRD). As shown in Fig. 2(a), all three eutectogels exhibit broad diffraction peaks near the 2 theta value of 21.34°, indicating their natural amorphous structures.30 Additionally, the AA eutectogel and AH eutectogel show a sharp diffraction peak at 28.58°, likely due to the partial crystallization of acrylic acid during the polymerization process.31 This diffraction peak is significantly weakened in the AHD eutectogel, which can be attributed to the strong interaction between pDMAPS and p(AA-co-HEMAA). These data suggest the high transparency and good mechanical properties of the AHD eutectogels.32
The chemical structures of AA eutectogel, AH eutectogel, and AHD eutectogel were characterized by FT-IR spectroscopy. As shown in Fig. 2(b), the broad peak at 3303 cm−1 in the AA eutectogel corresponds to the stretching vibration of –OH in carboxyl group and [ChCl][EG]. The peak at 2931 cm−1 and the double peaks at 1000–1100 cm−1 are related to the stretching and bending vibrations of –C–H. The peak at 1712 cm−1 can be attributed to the stretching vibration of –C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O in the carboxyl group.33–35 The enhanced peak intensity at 3303 cm−1 in the AH eutectogel compared to the AA eutectogel is likely due to the telescopic vibrational of –NH in HEMAA. Two new peaks at 1629 cm−1 and 1555 cm−1 can be assigned to the bending vibrational of –NH in HEMAA.36,37 Meanwhile, the increased intensity of the peak at 1034 cm−1 is attributed to the –C–N stretching vibration in HEMAA.38 These results indicate the successful copolymerization of AA with HEMAA. Additionally, no new peaks were observed in the AHD eutectogel, which may be due to the low content of pDMAPS.
O in the carboxyl group.33–35 The enhanced peak intensity at 3303 cm−1 in the AH eutectogel compared to the AA eutectogel is likely due to the telescopic vibrational of –NH in HEMAA. Two new peaks at 1629 cm−1 and 1555 cm−1 can be assigned to the bending vibrational of –NH in HEMAA.36,37 Meanwhile, the increased intensity of the peak at 1034 cm−1 is attributed to the –C–N stretching vibration in HEMAA.38 These results indicate the successful copolymerization of AA with HEMAA. Additionally, no new peaks were observed in the AHD eutectogel, which may be due to the low content of pDMAPS.
The composition of the gel was characterized using X-ray photoelectron spectroscopy (XPS). In Fig. 2(c), the AHD eutectogel shows distinct peaks corresponding to C, N, O, S and Cl. Notably, three characteristic peaks of C 1s at 284.8, 286.1, and 288.7 eV correspond to carbon–carbon single bonds (–C–C–), carbon atoms bonded to oxygen (–C–O–), carbon–sulfur (–C–S–), carbon–nitrogen (–C–N–), and carbon atoms in the carbonyl group, respectively (Fig. 2(d)). The N 1s spectra show peaks at 399.65 and 402.3 eV, which can be attributed to carbon–nitrogen (–C–N–) and ammonium nitrogen (–N+(CH3)2CH2) (Fig. 2(e)). The peaks in the S 2p spectra at 167.55 and 168.7 eV are assigned to carbon–sulfur (–C–S–) and sulfonate anions (–SO3−) (Fig. 2(f)).39 These results confirm the successful preparation of the AHD eutectogel. The structural homogeneity of the three eutectogels was assessed using small-angle X-ray scattering (SAXS) and wide-angle X-ray scattering spectroscopy (WAXS). As shown in Fig. 2(g, h) and S3,† the absence of characteristic patterns implies that there is no phase separation, suggesting a homogeneous distribution of the polymer network, which ensures high mechanical properties of the materials. Additionally, crystallization phenomena observed in XRD were not detected in this analysis, likely due to the presence of only a few sparsely dispersed crystalline domains within the gel system. Dynamic thermomechanical analysis (DMA) of the three eutectogels revealed that the storage modulus and loss modulus of the AHD eutectogel were significantly enhanced compared to those of both the AA eutectogel and the AH eutectogel. This finding strongly supports the effectiveness of the DN structure in improving the mechanical properties of the eutectogels (Fig. 2(i)).
To investigate the morphological changes of the eutectogels, all samples containing [ChCl][EG] were characterized using scanning electron microscope (SEM). As shown in Fig. S4(a),† the AA eutectogel exhibits a loose microstructure, which may be related to the partial crystallization of acrylic acid, resulting in poor mechanical properties. Compared with AA eutectogel, AH eutectogel (Fig. S4(b)†) demonstrates a more uniform surface distribution and a more homogeneous microstructure. With the construction of the dual-network, the uniformity and density of AHD eutectogel is improved further, manifesting the advantage of the dual-network system in enhancing the structural strength and stability of the gels (Fig. S4(c)†). After soaking the eutectogels in ultrapure water for 24 hours to remove [ChCl][EG] and freeze-drying to retain the intact polymer network structure, SEM tests were performed to investigate the network morphology of the three different eutectogel systems. As shown in Fig. S4(d),† the AA eutectogel exhibited a macroporous structure with disorganized pore diameters, leading to poor mechanical properties.40 The copolymerization of HEMAA with AA (Fig. S4(e)†), resulted in a polymer network with interconnected pores that are more densely arranged, although the pore sizes remain inhomogeneous. In contrast, the AHD eutectogel demonstrated a spongy structure (Fig. S4(f)†) with dense, uniform pores of about 10 μm in diameter. This structure facilitates energy transfer during the strain process of the co-polymerized gel, resulting in a better mechanical flexibility.41
3.2 Mechanical properties
To evaluate the effect of different monomer content, the mechanical properties of AA eutectogel, AH eutectogel, and AHD eutectogel were characterized. First, the effect of varying AA content with 30 wt%, 40 wt%, 50 wt%, and 60 wt% in AA eutectogels was explored, and the samples were named AA3, AA4, AA5, and AA6, respectively (when the content of AA was as high as 70 wt%, the eutectogel could not be formed). As shown in Fig. S5(a),† the tensile strength and elongation at break of the eutectogels substantially increased with the increase in the mass fraction of AA. At a mass fraction of 60% AA, the tensile strength reached 0.402 MPa, and the elongation at break was 427%. The corresponding calculated toughness was 1.227 MJ m−3 and the elastic modulus was 0.26 MPa (Fig. S5(a′)†). Consequently, the monomer content in the following system was fixed at 60 wt%.Further optimization of the mechanical properties of eutectogels was achieved by varying the proportion of AA and HEMAA monomers. The mass ratios of AA to HEMAA monomers were set at 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0, 8
0, 8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2, 6
2, 6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4, 4
4, 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 6, 2
6, 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 8 and 0
8 and 0![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10, respectively, and the samples were named AA, A8H2, A6H4, A4H6, A2H8, and HEMAA. The A6H4 eutectogel exhibited the strongest tensile strength of 0.573 MPa with elongation at break of 515% (Fig. S5(b)†). The toughness was approximately 1.737 MJ m−3, and the elastic modulus was about 0.171 MPa (Fig. S5(b′)†). It is well known that the lower the elastic modulus, the easier the material deforms. Based on these results, the A6H4 eutectogel was selected for subsequent studies.
10, respectively, and the samples were named AA, A8H2, A6H4, A4H6, A2H8, and HEMAA. The A6H4 eutectogel exhibited the strongest tensile strength of 0.573 MPa with elongation at break of 515% (Fig. S5(b)†). The toughness was approximately 1.737 MJ m−3, and the elastic modulus was about 0.171 MPa (Fig. S5(b′)†). It is well known that the lower the elastic modulus, the easier the material deforms. Based on these results, the A6H4 eutectogel was selected for subsequent studies.
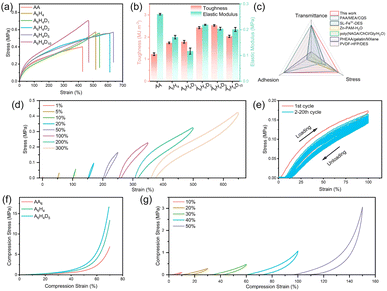
To further improve the toughness, a second polymer network of pDMAPS was added to the A6H4 eutectogel to form a dual network structure. The mechanical properties of the eutectogels were investigated by adjusting the pDMAPS content. The pDMAPS content was varied from 1 wt% to 10 wt%, and the samples were named A6H4D1, A6H4D2, A6H4D5, and A6H4D10. As demonstrated in Fig. 3(a), the elongation at break initially increased and then decreased with the increase in pDMAPS content, while the tensile strength continued to increase. When the pDMAPS content was 5 wt%, the elongation at break was the highest (637%), and the tensile strength reached 0.569 MPa. As depicted in Fig. 3(b), the A6H4D5 eutectogel exhibited the highest toughness (2.529 MJ m−3) and an elastic modulus of (0.204 MPa). These results clearly indicate that pDMAPS significantly enhances the toughness and mechanical properties of the eutectogels. The prepared A6H4D5 eutectogel outperforms most previously reported eutectogels, offering good tensile strength, high transparency, high toughness, low elastic modulus, and excellent adhesion properties (Fig. 3(c) and Table S1†).42–47
To estimate the energy dissipation ability of A6H4D5 eutectogel more intuitively, continuous loading–unloading cycles were performed under different strains of 1%, 5%, 10%, 20%, 50%, 100%, 200% and 300% (Fig. 3(d)). A typical hysteresis phenomenon was observed with increasing strain, suggesting significant energy consuming during cyclic tests.48 Additionally, continuous loading–unloading cyclic tests at a fixed strain were conducted to investigate the fatigue resistance of the A6H4D5 eutectogel (Fig. 3(e), S6(a and b)†). The A6H4D5 eutectogel exhibited a pronounced hysteresis phenomenon during the first tensile cycle, likely due to the reversible interactions of hydrogen bonds. As the number of cycles increased, the area of the hysteresis loop and the maximum stress gradually decreased, corresponding to the destruction of the chemical bonds in the double network structure of the eutectogel.49 Moreover, more than 85% of the maximum stress was maintained after 20 cycles, indicating good fatigue resistance. And the eutectogel was also tested for 1000 cycles of rapid cycling (tensile speed: 500 mm min−1), and the maximum stress could still be maintained at 65%. These results demonstrate that the A6H4D5 eutectogel possesses excellent resilience and mechanical reversibility.
The compressive properties of AA eutectogel, AH eutectogel, and AHD eutectogel were evaluated through compression tests. As shown in Fig. 3(f), all materials demonstrated exceptional compressive abilities, withstanding up to 70% deformation. Notably, the AHD eutectogel exhibited the highest compressive strength at 16.66 MPa. Continuous cyclic compression tests were conducted on the A6H4D5 eutectogel under varying compressive strains. As depicted in Fig. 3(g), the A6H4D5 eutectogel efficiently deformed and quickly returned to its original state under different compressive strains, indicating excellent fatigue resistance during compression. These results highlight the superior mechanical properties of the A6H4D5 eutectogel, making it suitable for use in flexible wearable electronic devices.
3.3 Adhesion studies
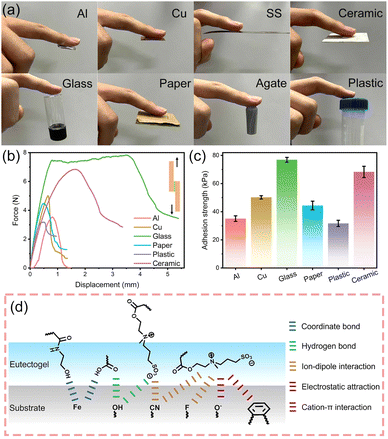
Eutectogels, due to their excellent adhesion properties, can adhere closely to substrates without additional adhesives, allowing accurate monitoring of signal changes.50 The adhesion of A6H4D5 eutectogel was explored, as shown in Fig. 4(a). The A6H4D5 eutectogel can firmly adhere to aluminium, copper, stainless steel (SS), ceramic sheets, glass, paper, agate, and plastic surfaces, confirming its excellent adhesion properties. Furthermore, the eutectogels were sandwiched between substrates, and the bond strength was quantitatively measured by a lap-shear test (Fig. 4(b)). The eutectogel showed the best adhesion to glass, with an adhesion force of 7.77 N. The adhesion strengths were calculated by dividing the maximum adhesion force by the overlapping contact area. As shown in Fig. 4(c), the adhesion strengths of the aluminium sheets, copper sheets, glass sheets, paper, plastic, and ceramic were 35.1 kPa, 50.2 kPa, 76.9 kPa, 44.4 kPa, 31.7 kPa, and 68.3 kPa, respectively. This excellent adhesion of the eutectogel to polar substrates is attributed to a combination of coordinate bond, hydrogen bond, electrostatic attraction, ion–dipole, and cation–π interactions (Fig. 4(d)), which is expected to be applied to human skin, robotic arms, and smart robots in the future.513.4 Strain sensing performance studies
To evaluate the ionic conductivity and strain sensitivity, the eutectogels were connected to an LED in a closed circuit, as illustrated in Fig. S7(a and b)† and demonstrated in Movie S1.† In its original state, the A6H4D5 eutectogel exhibits excellent conductivity. When the eutectogel is stretched, the LED dims slightly, which can be attributed to the ionic mobility within [ChCl][EG]. The rapid change in resistance of the eutectogel under tensile strain indicates its potential for real-time sensing applications. Additionally, electrochemical impedance spectroscopy (EIS) tests were conducted on AA eutectogel, AH eutectogel, and AHD eutectogel with varying pDMAPS content, as shown in Fig. S7(c) and (d).† All samples exhibit low resistance values. The ionic conductivity of the A6H4D5 eutectogel is approximately 2.731 × 10−2 mS cm−1.Benefiting from its excellent electrical conductivity, high tensile strength, and resilience, the sensing performance of the A6H4D5 eutectogel-based strain sensor was evaluated. Fig. 5(a) shows the relationship between tensile strain and relative resistance change, which was used to determine the gauge factor (GF). The sensor exhibits a GF of 0.883 for small strains (0–150%) and a GF of 1.541 for larger strains (150–600%). This sensitivity is comparable to that of current gel-based strain sensors, making it suitable for everyday applications.
To assess the stability of the sensor's response, loading–unloading cycle tests were conducted at various strains. As shown in Fig. 5(b and c), the sensor's electrical signals accurately track strain changes in real time, with response values increasing progressively with strain. The electrical signal changes are symmetrical during loading and unloading. Furthermore, the A6H4D5 eutectogel-based sensor was subjected to multiple loading–unloading cycles at a fixed strain. As shown in Fig. 5(d), at small strains, the response values remain stable without baseline drift or numerical attenuation as the number of cycles increased, indicating the sensor's reliability for small strains. At strains of 100% and 200%, the response values slightly decrease with increasing cycles. For instance, at 200% strain, the response value of the 5th cycle decreased by approximately 7.7% compared to the first cycle. This reduction is primarily due to the disruption of the dual network structures under successive stretching.
To further assess the stability of the sensor response, the A6H4D5 eutectogel-based sensor was subjected to 20 cycles at 50% strain, with tensile rates ranging from 10 mm min−1 to 500 mm min−1. As shown in Fig. 5(e), the sensor demonstrates good stability throughout the cycling tests, suggesting its potential for detecting human movement and biomedical signals. Fig. 5(f) illustrates that the response values of the A6H4D5 eutectogel-based sensors remain consistent after strain holding, indicating excellent repeatability. The response and recovery times of the sensor at 20% strain are 564 ms and 520 ms respectively (Fig. 5(g)), demonstrating the sensor's quick reaction to strain. Additionally, the sensor was subjected to 1600 load–unload cycles at 50% strain. A slight drop in the electrical signal occurs at the beginning of the cycle due to stress relaxation and dynamic cross-link reorganization of the dual-network. However, as the material gradually stabilizes, the sensor maintains a relatively stable electrical signal, which demonstrates its excellent mechanical durability and impressive repeatability (Fig. 5(h)).
The A6H4D5 eutectogel-based sensor also exhibits excellent pressure-sensing properties. As shown in Fig. S8(a),† the pressure sensitivity of the sensor can be categorized into three regions: 0.684 kPa−1 (0–14 kPa), 0.125 kPa−1 (14–90 kPa), and 0.097 kPa−1 (90–200 kPa). The sensor's response decreases with increasing pressure, which is attributed to the shortening of the conductive path and a reduction in relative resistance. Additionally, the GF value of the sensor gradually decreases with increasing compressive strain (Fig. S8(b)†). The sensor's electrical signals can accurately track changes in strain in real time (Fig. S8(c)†). At a fixed strain, response values remain stable without baseline drift or numerical decay as the number of loading–unloading cycles increases (Fig. S8(d)†), indicating that the sensor is suitable for applications involving small strains. Moreover, the sensor's response remains nearly constant across different velocities (10–500 mm min−1), demonstrating stable and reproducible feedback (Fig. S8(e)†).
The A6H4D5 eutectogel-based sensor is also capable of detecting various human activities and monitoring health in practical scenarios. As shown in Fig. 6(a) and Movie S2,† the sensor was installed at finger joints to monitor finger movements. When the finger is bent sequentially to angles of 30°, 60°, and 90°, the resistance changes distinctly, and the electrical signal remains highly repeatable and stable. The sensor accurately reflects the degree of finger bending in real time, even when the finger transitions rapidly between straightening and bending at 90° (Fig. 6(b)). Similarly, when the sensor is attached to the wrist and elbow, it effectively tracks resistance changes during bending and relaxing processes, showing quick recovery and responsiveness (Fig. 6(c) and (d)). Moreover, the electrical signal from the A6H4D5 eutectogel-based sensor remained stable during multiple instances of elbow bending to 90° (Fig. 6(e)). When attached to the back of the neck, the sensor effectively monitored head nodding motions (Fig. 6(f)). The sensor also successfully detected various leg movements when attached to the knee, such as deep squatting, knee bending, walking, fast walking, and running, by analyzing signal strength, frequency, and peak shape (Fig. 6(g–i)). Furthermore, it could recognize facial expressions and complex muscle movements, such as smiling (Fig. 6(j)). The in vitro cytotoxicity of the A6H4D5 eutectogel-based strain sensor was evaluated by culturing L929 cells with extracts of the A6H4D5 eutectogel. Cells incubated with the A6H4D5 eutectogel extracts exhibited over 85% viability compared to the control group even at 100% cell concentration, in a clean medium (Fig. 6(h)), confirming no significant release of cytotoxic components. After 24 hours of exposure to the A6H4D5 eutectogel extract, L929 cells retained normal morphology with no observable signs of apoptosis or necrosis (Fig. 6(i)). These results indicate that the A6H4D5 eutectogel possesses outstanding biosafety and biocompatibility, supporting its potential application in wearable health monitoring systems for human motion and physiological signal detection.5,52
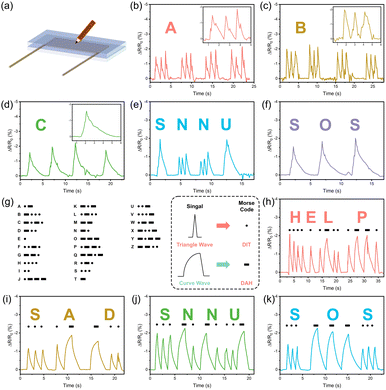
Additionally, the conversion of writing signals from handwriting recording devices into text has gained significant attention in human–computer interaction systems. Gel sensors have shown promise as handwriting carriers, capable of recognizing various human writing content due to the different stresses applied along the sliding path.53 To explore this capability, handwriting experiments were conducted by wrapping the hydrogel with VHB tape for stable operation and connecting it to the experimental apparatus with wires as electrodes on both sides (Fig. 7(a)). Each letter (A, B, and C) produces a unique output signal (Fig. 7(b–d)). When writing the words “SNNU” (Fig. 7(e)) and “SOS” (Fig. 7(f)), the sensor provided clear and distinct electrical signals, demonstrating its potential for monitoring human writing behavior effectively.
Moreover, the sensor was tested for Morse code recognition and translation. Tapped Morse code was successfully recognized and translated by distinguishing between dots and horizontal lines (tapping and long-pressing) (Fig. 7(g)). Electrical signal representations for words such as “HELP”, “SAD”, “SNNU”, and “SOS” were accurately displayed (Fig. 7(h–k)). This capability indicates that the sensor could facilitate encrypted information transmission, further expanding its application range in flexible wearable technology.
4. Conclusion
In summary, we successfully synthesized the pDMAPS chains by dissolving DMAPS monomers in [ChCl][EG], and then added AA and HEMAA monomers to polymerize p(AA-co-HEMAA) chains. This process resulted in the cross-linking of pDMAPS and p(AA-co-HEMAA) chains, forming a robust three-dimensional network. The pDMAPS component provided enhanced viscosity and high elasticity, while the p(AA-co-HEMAA) component improved mechanical strength. Mechanical property evaluations demonstrated that the copolymerization of HEMAA and the cross-linking of pDMAPS significantly enhanced the mechanical properties of the eutectogel. Among the various formulations, the A6H4D5 eutectogel exhibited exceptional performance, with high tensile strength (0.569 MPa), elongation at break (637%), high toughness (2.529 MJ m−3), and high optical transparency (∼89%). Additionally, it showed excellent viscosity and stability. Finally, the eutectogel was assembled into a flexible sensor and successfully applied to detect various limb motions, recognize handwriting, and translate Morse code. This study highlights the exceptional performance of the eutectogel-based strain sensor and presents a novel approach for enhancing eutectogel materials.Data availability
The authors confirm that the data supporting the findings of this study are available within the article and its ESI.†Author contributions
Zhuangzhuang Ma: conceptualization, validation, visualization, methodology, writing – review &editing, and writing – original draft. Jiale Zhang: validation, formal analysis, and visualization. Zelin Ma: validation and software. Minghao Lou and Peijin Zou: visualization. Hongqiang Wang: resources, supervision, and project administration. Lichao Jia: funding acquisition, supervision, project administration, and conceptualization.Conflicts of interest
There are no conflicts to declare.Acknowledgements
This work was financially supported by the fund of the State Key Laboratory of Solidification Processing in NWPU (Grant No. SKLSP202407), and the Young Scientist Initiative Project of School of Materials Science and Engineering at Shaanxi Normal University (2023YSIP-MSE-SNNU001).Notes and references
- X. Xiong, Y. Chen, Z. Wang, H. Liu, M. Le, C. Lin, G. Wu, L. Wang, X. Shi, Y.-G. Jia and Y. Zhao, Nat. Commun., 2023, 14, 1331 CrossRef CAS PubMed.
- J. Yi and Y. Xianyu, Adv. Funct. Mater., 2022, 32, 2113012 CrossRef CAS.
- S. Song, F. Fallegger, A. Trouillet, K. Kim and S. P. Lacour, Sci. Robot., 2023, 8, eadd1002 CrossRef PubMed.
- K. Zhai, H. Wang, Q. Ding, Z. Wu, M. Ding, K. Tao, B.-R. Yang, X. Xie, C. Li and J. Wu, Adv. Sci., 2023, 10, 2205632 CrossRef CAS PubMed.
- J. Zhang, Z. Ma, M. Li, M. Lou, H. Wang and L. Jia, Adv. Funct. Mater., 2024, 2415694 Search PubMed.
- Z. Zhang, L. Qian, B. Zhang, C. Ma and G. Zhang, Angew. Chem., Int. Ed., 2024, e202410335 CAS.
- L. Li, W. Li, X. Wang, X. Zou, S. Zheng, Z. Liu, Q. Li, Q. Xia and F. Yan, Angew. Chem., Int. Ed., 2022, 61, e202212512 CrossRef CAS PubMed.
- K. Q. Fan, W. C. Wei, Z. Q. Zhang, B. Liu, W. B. Feng, Y. P. Ma and X. J. Zhang, Chem. Eng. J., 2022, 449, 137878 CrossRef CAS.
- B. B. Hansen, S. Spittle, B. Chen, D. Poe, Y. Zhang, J. M. Klein, A. Horton, L. Adhikari, T. Zelovich, B. W. Doherty, B. Gurkan, E. J. Maginn, A. Ragauskas, M. Dadmun, T. A. Zawodzinski, G. A. Baker, M. E. Tuckerman, R. F. Savinell and J. R. Sangoro, Chem. Rev., 2021, 121, 1232–1285 CrossRef CAS PubMed.
- H. Zhang, N. Tang, X. Yu, M.-H. Li and J. Hu, Adv. Funct. Mater., 2022, 32, 2206305 CrossRef CAS.
- S. J. He, H. L. Ren, Y. Y. Chen, D. Rong and Q. F. Rong, J. Energy Storage, 2023, 72, 108619 CrossRef.
- J. Liu, L. Zhou, P. Zhang, Y. Zhao, H. Wei and Y. Yu, CCS Chem., 2023, 1–13 CrossRef.
- J. P. Gong, Y. Katsuyama, T. Kurokawa and Y. Osada, Adv. Mater., 2003, 15, 1155–1158 Search PubMed.
- S. Ko, A. Chhetry, D. Kim, H. Yoon and J. Y. Park, ACS Appl. Mater. Interfaces, 2022, 14, 31363–31372 CrossRef CAS PubMed.
- C. L. Liu, Z. Y. Xu, S. Chandrasekaran, Y. P. Liu and M. Y. Wu, Carbohydr. Polym., 2023, 303, 120468 CrossRef CAS PubMed.
- Y. C. Luo, M. L. Yu, Y. T. Zhang, Y. Y. Wang, L. Long, H. H. Tan, N. Li, L. J. Xu and J. X. Xu, Nano Energy, 2022, 104, 107955 CrossRef CAS.
- J. Hu, J. Guo, J. Zhao, Z. Chen, M. Kalulu, G. Chen and G. Fu, ACS Appl. Mater. Interfaces, 2024, 16, 10671–10681 CrossRef CAS PubMed.
- M. Ajmal, S. Demirci, M. Siddiq, N. Aktas and N. Sahiner, Colloids Surf., A, 2015, 486, 29–37 CrossRef CAS.
- Y. Wu, Y. Deng, K. Zhang, Y. Wang, L. Wang and L. Yan, J. Mater. Chem. A, 2022, 10, 17721–17729 RSC.
- Y. Zhang, Y. Wang, Y. Guan and Y. Zhang, Nat. Commun., 2022, 13, 6671 CrossRef CAS PubMed.
- Y. C. Song, H. Li, T. H. Shan, P. P. Yang, S. W. Li, Z. Liu, C. T. Liu and C. Y. Shen, Carbohydr. Polym., 2023, 302, 120377 CrossRef CAS PubMed.
- B. C. Zhao, H. B. Jiang, Z. K. Lin, S. F. Xu, J. Xie and A. P. Zhang, Carbohydr. Polym., 2019, 224, 115022 CrossRef PubMed.
- A. Elizalde-Cárdenas, M. González-Torres, G. Leyva-Gómez, H. Cortés, O. González-Mendoza, M. A. Pérez-Díaz, C. Pineda and R. M. Ribas-Aparicio, Mater. Lett., 2022, 324, 132783 CrossRef.
- C. X. Lei, Y. H. Guo, W. X. Guan, H. Y. Lu, W. Shi and G. H. Yu, Angew. Chem., Int. Ed., 2022, 61, e202200271 CrossRef CAS PubMed.
- Y. Zhao, N. Yang, X. Chu, F. Sun, M. U. Ali, Y. Zhang, B. Yang, Y. Cai, M. Liu, N. Gasparini, J. Zheng, C. Zhang, C. Guo and H. Meng, Adv. Mater., 2023, 35, 2211617 CrossRef CAS PubMed.
- J. Zhu, M. Cho, Y. Li, T. He, J. Ahn, J. Park, T.-L. Ren, C. Lee and I. Park, Nano Energy, 2021, 86, 106035 CrossRef CAS.
- R. Eelkema and A. Pich, Adv. Mater., 2020, 32, 1906012 CrossRef CAS PubMed.
- Y. Zheng, H. Liu, J. Wang, T. Cui, J. Zhu and Z. Gui, Adv. Fiber Mater., 2024, 6, 195–213 CrossRef CAS.
- G. Ge, Y. Zhang, X. Xiao, Y. Gong, C. Liu, C. Lyu, W. L. Ong, G. W. Ho, Z. Yang and W. Huang, Adv. Funct. Mater., 2024, 34, 2310963 CrossRef CAS.
- H. Yamaguchi, S. Nakanishi, H. Iba and T. Itoh, J. Power Sources, 2015, 275, 1–5 CrossRef CAS.
- S.-A. Chen and H.-T. Lee, Macromolecules, 1995, 28, 2858–2866 CrossRef CAS.
- J. Chen, Y. Wang, L. Li, Y.-E. Miao, X. Zhao, X.-P. Yan, C. Zhang, W. Feng and T. Liu, ACS Appl. Mater. Interfaces, 2023, 15, 16109–16117 CrossRef CAS PubMed.
- Y. L. Zhao, H. L. Cheng, Y. X. Li, J. C. Rao, S. Z. Yue, Q. J. Le, Q. Qian, Z. Liu and J. Y. Ouyang, J. Mater. Chem. A, 2022, 10, 4222–4229 RSC.
- V. Nesslinger, A. G. Orive, D. Meinderink and G. Grundmeier, J. Colloid Interface Sci., 2022, 615, 563–576 CrossRef CAS PubMed.
- K. Kumar, M. Bisht, H. Arora, S. M. Ghoreishian, R. Umapathi, Y. S. Huh and P. Venkatesu, ACS Sustain. Chem. Eng., 2022, 10, 9991–10002 CrossRef CAS.
- G. Ge, K. Mandal, R. Haghniaz, M. Li, X. Xiao, L. Carlson, V. Jucaud, M. R. Dokmeci, G. W. Ho and A. Khademhosseini, Adv. Funct. Mater., 2023, 33, 2207388 CrossRef CAS PubMed.
- Y. Go, H.-Y. Park, Y. Zhu, K. Yoo, J. Kwak, S.-H. Jin and J. Yoon, Adv. Funct. Mater., 2023, 33, 2215193 CrossRef CAS.
- H. Zhi, X. B. Zhang, F. Y. Wang and L. Feng, ACS Appl. Mater. Interfaces, 2022, 14, 52422–52429 CrossRef CAS PubMed.
- J. Yang, L. Chang, H. Deng and Z. Cao, ACS Nano, 2024, 18, 18980–18991 CrossRef CAS PubMed.
- S. Q. Liu, Y. H. Zhong, X. L. Zhang, M. H. Pi, X. Y. Wang, R. J. Zhu, W. Cui and R. Ran, ACS Appl. Mater. Interfaces, 2022, 14, 15641–15652 CrossRef CAS PubMed.
- Z. Sun, Y. D. Kuang, M. Ahmad, Y. Huang, S. Yin, F. Seidi and S. Wang, Carbohydr. Polym., 2023, 305, 120556 CrossRef CAS PubMed.
- Y. Feng, J. Yu, D. Sun, C. Dang, W. Ren, C. Shao and R. Sun, Nano Energy, 2022, 98, 107284 CrossRef CAS.
- B. Guo, M. Yao, S. Chen, Q. Yu, L. Liang, C. Yu, M. Liu, H. Hao, H. Zhang, F. Yao and J. Li, Adv. Funct. Mater., 2024, 34, 2315656 CrossRef CAS.
- Z. Li, S. Zhang, Z. Jiang, D. Cai, C. Gu and J. Tu, Mater. Chem. Phys., 2021, 267, 124701 CrossRef CAS.
- Y. Liu, X. Zhang, B. Li, H. Chen, H. Li, J. Chen and H. Dong, Chem. Eng. J., 2023, 461, 141965 CrossRef CAS.
- Y. Wu, Y. Deng, K. Zhang, J. Qiu, J. Wu and L. Yan, ACS Appl. Energy Mater., 2022, 5, 3013–3021 CrossRef CAS.
- Y. Wu, L. Yang, J. Wang, S. Li, X. Zhang, D. Chen, Y. Ma and W. Yang, ACS Appl. Mater. Interfaces, 2023, 15, 36759–36770 CrossRef CAS PubMed.
- K. Xue, C. Shao, J. Yu, H. Zhang, B. Wang, W. Ren, Y. Cheng, Z. Jin, F. Zhang, Z. Wang and R. Sun, Adv. Funct. Mater., 2023, 33, 2305879 CrossRef CAS.
- J. S. Yang, Y. W. Feng, B. Z. Wang, J. S. Miao, S. H. Wei, H. Li, L. T. Mo and Z. Y. Qin, Chem. Eng. J., 2023, 474, 145544 CrossRef CAS.
- X. Wang, W. Han, J. Yang, P. Cheng, Y. Wang, C. Feng, C. Wang, H. Zhang, Y. Sun and G. Lu, Sens. Actuators, B, 2022, 361, 131707 CrossRef CAS.
- H. Ren, Z. Zhang, X. Cheng, Z. Zou, X. Chen and C. He, Sci. Adv., 2023, 9, eadh4327 CrossRef CAS PubMed.
- M. Li, J. Zhang, G. Chen, Y. Liu, D. Xie, M. Li, Y. Zhang, J. Song and Z. Luo, Int. J. Biol. Macromol., 2025, 303, 140657 CrossRef CAS PubMed.
- Y. Liu, F. Zhuo, J. Zhou, L. Kuang, K. Tan, H. Lu, J. Cai, Y. Guo, R. Cao, Y. Fu and H. Duan, ACS Appl. Mater. Interfaces, 2022, 14, 54276–54286 CrossRef CAS PubMed.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: https://doi.org/10.1039/d5ta00422e |
| This journal is © The Royal Society of Chemistry 2025 |