Modulating charge transfer pathway via halide tuning of layered Bi-oxyhalides on an MOF-derived CuO nanorod photocathode†
Sneha
Joseph
 a,
Mohit
Kumar
a,
Mohit
Kumar
 ab,
Suryansh
Chalamalasetty
a,
Bhagatram
Meena
ab,
Suryansh
Chalamalasetty
a,
Bhagatram
Meena
 a and
Subrahmanyam
Challapalli
a and
Subrahmanyam
Challapalli
 *a
*a
aDepartment of Chemistry, Indian Institute of Technology Hyderabad, Sangareddy, Kandi, Telangana 502285, India. E-mail: csubbu@iith.ac.in
bDepartment of Chemistry and Biochemistry, School of Science Computing and Engineering, Swinburne University of Technology, Hawthorn, VIC 3122, Australia
First published on 21st January 2025
Abstract
Charge transfer pathways in heterojunctions are crucial for determining photoexcited charge generation, separation, and kinetics of surface reactions. Nature-inspired Z-scheme and type-I heterojunctions are the most sought mechanisms in photo-assisted processes aimed at charge separation and achieving higher photovoltages, thereby improving photoconversion efficiencies. Herein, we present a novel strategy to control the charge transfer pathway in p–n heterojunctions by modulating the halide in a metal oxyhalide combined with metal–organic framework (MOF)-templated CuO nanorods (M-CuO NRs). Charge transfer mechanisms and kinetics across p–n heterojunctions were systematically investigated using M-CuO NRs/BiOX (X = Cl, Br, and I) photocathodes for photoelectrochemical (PEC) hydrogen evolution reactions. Varying halide species led to transition in the charge transfer mechanism, resulting in distinct PEC conversion characteristics. Furthermore, J = −5.6 mA cm−2 at 0 VRHE and an HC-STH (%) of 0.95% at 0.34 VRHE were achieved for M-CuO NRs/BiOI, while J = −5.1 mA cm−2 at 0 VRHE and an HC-STH (%) of 0.85% at 0.40 VRHE were obtained for M-CuO NRs/BiOBr. Our findings demonstrate that modulating the halide in BiOX significantly impacts charge transfer pathways, enhancing PEC conversion kinetics and hydrogen evolution under solar irradiation. Comprehensive characterizations of M-CuO NRs/BiOX photoelectrodes were conducted, including crystalline structure, morphology, photoelectrochemical performance, and photophysical properties.
Introduction
Continuous and thorough research into renewable energy technology is necessary to address the energy generation gap between conventional and non-conventional sources and ensure a sustainable future.1–3 Photoelectrochemical (PEC) water splitting is a highly prospective technique for sustainable generation of hydrogen, leveraging solar energy to drive the redox reactions involved in water dissociation, thereby providing a renewable and eco-friendly alternative to conventional fossil fuel-based processes. PEC-generated hydrogen can be directly used as green hydrogen or further processed into hydrocarbons such as ethane and methane, as well as aid in carbon sequestration through the Fischer–Tropsch process.4,5 The commercial viability of hydrogen production using PEC technology is severely hampered by the employment of thin film photoelectrodes in PEC cells, which pose significant hurdles such as reduced photoconversion efficiency and photodegradation susceptibility. Most PEC studies predominantly concentrate on n-type materials such as TiO2 (ref. 6–8) and BiVO4,9,10 whereas investigations into photocathodes are comparatively limited. Currently, assessing the limitations in material bandgap tunability and modulation of electronic bands presents a significant challenge, restricting researchers to a limited range of materials available for photoelectrode fabrication. This limitation impedes the investigation and advancement of more effective photoelectrochemical systems since the capacity to precisely adjust these characteristics is essential to maximize photoelectrode performance.Precise control of the conduction and valence bands in semiconductors is a potent and promising way to customize the material's electrical properties, thus maximizing its performance for specific applications. To satisfy a variety of technological requirements, these band topologies can be carefully adjusted to optimize light absorption, increase charge carrier mobility, and fine-tune electronic band gaps. This strategy holds significant promise for advancing semiconductor-based devices in areas such as photovoltaics, photoelectrocatalysis, photocatalysis, and electronic sensors, where precise control of material properties is essential for achieving high efficiency and performance.11 The choice of halides and cation size in perovskites plays a key role in tuning the band gap by altering the electronic structure and crystal geometry by changes in orbital overlap and antibonding interactions. For instance, increasing the proportion of halides such as bromine compared to iodine can widen the band gap. Moreover, geometrical features such as octahedral tilting and bond angles also influence the band gap, highlighting the interplay between composition and crystal structure.12,13
Layered bismuth-based oxyhalides (BiOX, X: Cl, Br, and I) is a V–VI–VII ternary compound semiconductor class that has garnered considerable attention due to their unique layered structure owing to van der Waals interaction and ability to modulate halides within the crystal lattice, allowing for precise control over band positions and bandgaps. Although Bi-oxyhalides have been successfully employed in photocatalysis for the degradation of organic pollutants and pharmaceutical compounds, their potential for application in PEC water splitting remains underexplored and warrants further investigation.14–16 The 30% CuO/BiOI composite demonstrated improved photodegradation of methyl orange, primarily driven by ˙O2− radicals, with additional contributions from holes (h+) and ˙OH radicals. This enhancement is due to efficient charge separation and reactive species generation under visible light.17 In a PEC bioanalytical system, using a hierarchical nanostructured BiOI nanosheet/nickel oxide (NiO) film as the photocathode and glucose oxidases (GOx) as the oxidase, a glucose-controlled increase in cathodic photocurrent was noted.18 Also, bismuth oxyiodides have the potential of photocurrent switching between n-type and p-type.19 Reports have documented the calcination of the precursor molecule Bi7O9I3, leading to the formation of a sequence of bismuth oxyiodides. The enhanced photocatalytic activity observed was attributed to iodine vacancy formation at high temperatures, occurring before the material experiences a phase transition. These vacancies were associated with an increase in band gap, downward shifts in band potentials, and a transition in the semiconductor behaviour from p-type to n-type, which collectively improved the material's photocatalytic efficiency.20 Poznyak et al. were the first to report photocurrent switching behaviour of bismuth oxyiodide (BiOI) photoelectrodes.21 BiOI microplatelets exhibited the photoelectrochemical photocurrent switching (PEPS) effect, where a shift from anodic to cathodic photocurrent was observed at approximately 0.5 VNHE.22
Another category of materials known as metal organic frameworks (MOFs) have sparked widespread interest owing to control over diverse crystal facets, morphology, high porosity, and active sites. They have gained attention as precursors,23–25 photosensitizers,26–28 and co-catalysts29–32 owing to their tunable structures and abundant active sites, making them valuable for photoelectrochemical (PEC) water splitting.33 MOFs demonstrate significant potential as sacrificial templates in the synthesis of metal oxides, providing ample surface area and numerous active sites for enhanced functionality.34,35 Similar kind of works are presented by Cardenas-Morcoso et al., wherein porous cobalt-oxide co-catalysts, based on a cobalt–imidazolium MOF (ZIF-67), have been used as cocatalysts for enhancing the performance of BiVO4-based photoanodes.36 The MOF-derived cocatalyst accelerated the photoanodic OER and thus greatly improved the overall PEC performance. Incorporation of cocatalysts sourced from MOFs boosts the productivity of the photoanodic oxygen evolution reaction (OER), thereby leading to a notable improvement in the overall photoelectrochemical (PEC) systems' performance. In a similar fashion, a template of MOF-on-MOF (metal–organic framework) was utilized as the material to create a composite of In2O3/CuO p–n heterojunction. More specifically, the process involved the growth of Cu-BDC on the exterior of a hexagonal rod-shaped MIL-68(In) to establish a heterostructure known as MIL-68(In)/Cu-BDC. Subsequent annealing yielded the hybrid to be developed into an In2O3/CuO p–n heterojunction composite, while preserving the primary hollow hexagonal rod-shaped structure of the MOF.37
In this report, we propose a strategy to precisely control the charge transfer mechanism and modulate the kinetics of charge transfer in a heterojunction photocathode by tuning halides in layered BiOX (X = Cl, Br, I), decorated over MOF-derived CuO nanorods (M-CuO NRs). Using Cu-BTC MOF as a precursor for M-CuO NRs synthesis through a modified chemical treatment, the resultant M-CuO NRs/BiOX heterojunction photocathodes were thoroughly investigated. Study on M-CuO NRs/BiOI and M-CuO NRs/BiOBr revealed ameliorated PEC activity with J = −5.6 mA cm−2 at 0 VRHE, HC-STH of 0.95% at 0.34 VRHE and J = −5.1 mA cm−2 at 0 VRHE, HC-STH of 0.85% at 0.40 VRHE, respectively, compared to M-CuO NRs (J = −4.1 mA cm−2 at 0 VRHE, HC-STH of 0.40% at 0.40 VRHE). The study reveals that modulating the halide in BiOX significantly alters the charge transfer mechanisms, thus enhancing photoelectrochemical conversion kinetics and hydrogen evolution under solar illumination. Charge transfer pathways affect the photostability in such a way that M-CuO NRs/BiOI shows higher photostability than M-CuO NRs/BiOBr and M-CuO NRs. Comprehensive characterization of M-CuO NRs/BiOX photoelectrodes was performed to examine their crystalline structure, morphology, photoelectrochemical behaviour, and photophysical properties.
Experimental section
Synthesis of M-CuO NRs and fabrication of photocathode
M-CuO NRs were synthesized through an energy-efficient method using Cu-BTC MOF as a precursor. The synthesis procedure and detailed characterization of Cu-BTC can be found in the ESI.† The as-synthesized Cu-BTC (0.6 g) was added to an NaOH (0.2 M, 30 mL) solution under continuous stirring (600 rpm) at room temperature. The initial blue color of the solution gradually turned dark, becoming fully brownish black after few minutes. The residue was collected at 5-, 10-, and 15 min intervals, washed three times with deionized water and finally with ethanol, and the obtained Cu(OH)2 was then dried overnight at 60 °C. M-CuO NRs photocathodes were prepared via doctor blading. A mixture of deionized water and acetylacetone (4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) was mixed with 10 mg of Cu (OH)2 and 1–2 drops of Triton X-100 to form a paste. This paste was applied onto FTO substrates and calcined at 400 °C for 2 h to obtain M-CuO NRs photocathode.
1) was mixed with 10 mg of Cu (OH)2 and 1–2 drops of Triton X-100 to form a paste. This paste was applied onto FTO substrates and calcined at 400 °C for 2 h to obtain M-CuO NRs photocathode.
Synthesis of BiOX (X = Cl, Br, I)
BiOX was synthesised by a simple co-precipitation reaction, which is well known. Bi(NO3)3·5H2O (0.364 g) and KI (0.125 g) were separately dissolved in 20 mL of ethanol (solution A) and 40 mL of distilled water (solution B) respectively with continuous stirring and ultrasonication. Solution B was then added gradually to solution A under constant stirring. The pH was controlled at 7 using NH3·H2O, followed by stirring at 70 °C for 3 hours. After cooling to room temperature, the dark brick red precipitate was collected, washed, and dried overnight at 80 °C. The synthesis procedure described above was also employed to prepare BiOCl and BiOBr by substituting KI with KCl and KBr, respectively, while maintaining the stoichiometric ratio for the reactants. Trivalent bismuth cations remain stable in strongly acidic aqueous solutions, but as the pH increases, they hydrolyse to form bismuthyl ions (BiO+). In the presence of X− ions, this can cause the spontaneous precipitation of BiOX as a fine powder (Fig. S7, ESI†). Therefore, meticulous pH control is essential for the controlled growth of bismuth oxyhalide nanostructures.38Fabrication of M-CuO NRs/BiOX photoelectrodes
Pre-synthesized M-CuO NRs photocathodes were drop-cast with BiOX solutions to prepare M-CuO NRs/BiOX photoelectrodes. BiOX solutions were made in isopropyl alcohol (IPA) with varying concentrations (0.03, 0.06, and 0.09 mg mL−1) and then drop-cast onto the M-CuO NRs photocathodes. The IPA solvent was allowed to evaporate at a regulated rate since the deposition was carried out on FTO substrates that were set on a hot plate maintained at 50 °C. The electrodes underwent annealing at 90 °C after deposition.Results and discussion
Synthesis and characterization of M-CuO NRs and BiOX (X: Cl, Br, and I)
The synthetic protocol for the sequential fabrication of M-CuO NRs and BiOI microflowers arranged in M-CuO NRs/BiOI photoelectrode is shown in a pictorial representation (Fig. 1a). Moreover, CuO photocathodes were fabricated using several processes, including hydrothermal synthesis, pyrolysis, and calcination for comparison. Detailed synthetic methodologies are included in the ESI (Fig. S1).† Scanning electron micrographs revealed the morphological characteristics of Cu-BTC octahedrons transitioning (inset Fig. 1b) to 1D nanorods (Fig. 1b and c) after chemical treatment, while H-CuO exhibited a flake-like structure (Fig. S3b, ESI†). M-CuO (O2) presented a truncated octahedron (Fig. S3c, ESI†). The primary cause of the shrinkage and truncation of M-CuO (O2) might be the loss of both C and H from organic ligands upon the thermal treatment of parent Cu-BTC.23 BiOI presented very peculiar microflowers (Fig. 1d and e) and BiOBr showed aggregated flakes-like structure (Fig. 1f and g). On further analysis of M-CuO NRs/BiOI, microflowers having an average diameter ∼ 3.5 μm were found to be arranged on top of M-CuO NRs in a layer-by-layer assembly pattern (Fig. 1h, k–m). M-CuO NRs were also found to be distributed on the petals of BiOI microflowers through the higher magnification of M-CuO NRs/BiOI (Fig. 1i and j).The X-ray diffraction (XRD) patterns of Cu-BTC show distinct and sharp peaks at 2θ = 9.52°, 11.68°, 13.3°, 14.6°, 16.5°, 17.46°, and 19.06°, which can be correlated with the crystal planes of the face-centered-cubic (lattice parameter a = 26.34300 Å) (220), (222), (400), (331), (422), (511), and (440), respectively, belonging to the Fm![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m space group. The XRD pattern indicates that the CuO coated over FTO has 2θ peaks at 32°, 35°, 39°, 49°, 53°, and 58° that are indexed to monoclinic CuO (JCPDS Card No. 45-937) crystal planes of (110), (
m space group. The XRD pattern indicates that the CuO coated over FTO has 2θ peaks at 32°, 35°, 39°, 49°, 53°, and 58° that are indexed to monoclinic CuO (JCPDS Card No. 45-937) crystal planes of (110), (![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 11), (202), (020), and (
11), (202), (020), and (![[2 with combining macron]](https://www.rsc.org/images/entities/char_0032_0304.gif) 02), respectively (Fig. 2a). Furthermore, as MOF-derived samples such as M-CuO(N2), M-CuO(O2), and M-CuO NRs lack peaks at about 0–30°, it can be inferred that the Cu-BTC was completely converted into CuO.39 The XRD patterns of the CuO nanostructures coated on FTO and prepared using various synthetic processes are shown in Fig. S4, ESI.† XRD investigation showed a distinct collection of diffraction peaks at 9.6°, 29.65°, 31.7°, 37.49°, 39.25°, 45.5°, 51.4°, 55.2°, 60.2°, 61.8°, 66.38°, and 74.1° to confirm the phase purity in BiOI. A strong structural proof of the material's crystallinity and phase integrity was provided by the precise correspondence of each of these peaks to a specific crystallographic plane, including (001), (002), (101), (102), (112), (200), (113), (202), (211), (204), (220), and (214). Its lattice constants are a = b = 4 Å and c = 9.1 Å, and the crystal structure is tetragonal.40 In Fig. S5, ESI,† the XRD patterns of BiOCl, BiOBr, and their composite with M-CuO NRs are presented. The detection of characteristic peaks corresponding to both M-CuO NRs and the respective BiOX phases within the XRD pattern of the M-CuO NRs/BiOX composite serve as compelling authentication for the successful fabrication of a biphasic composite structure, signifying the coexistence and integration of both constituent materials in a singular, well-defined system.
02), respectively (Fig. 2a). Furthermore, as MOF-derived samples such as M-CuO(N2), M-CuO(O2), and M-CuO NRs lack peaks at about 0–30°, it can be inferred that the Cu-BTC was completely converted into CuO.39 The XRD patterns of the CuO nanostructures coated on FTO and prepared using various synthetic processes are shown in Fig. S4, ESI.† XRD investigation showed a distinct collection of diffraction peaks at 9.6°, 29.65°, 31.7°, 37.49°, 39.25°, 45.5°, 51.4°, 55.2°, 60.2°, 61.8°, 66.38°, and 74.1° to confirm the phase purity in BiOI. A strong structural proof of the material's crystallinity and phase integrity was provided by the precise correspondence of each of these peaks to a specific crystallographic plane, including (001), (002), (101), (102), (112), (200), (113), (202), (211), (204), (220), and (214). Its lattice constants are a = b = 4 Å and c = 9.1 Å, and the crystal structure is tetragonal.40 In Fig. S5, ESI,† the XRD patterns of BiOCl, BiOBr, and their composite with M-CuO NRs are presented. The detection of characteristic peaks corresponding to both M-CuO NRs and the respective BiOX phases within the XRD pattern of the M-CuO NRs/BiOX composite serve as compelling authentication for the successful fabrication of a biphasic composite structure, signifying the coexistence and integration of both constituent materials in a singular, well-defined system.
The Raman spectra of M-CuO NRs are shown in Fig. 2b. Three Raman peaks are visible at 286, 333, and 615 cm−1, as shown in Fig. 2b.41 M-CuO NRs have two molecules in each primitive cell, placing them in the group of mathematical equation space. With the symmetry of 4Au + 5Bu + Ag + 2Bg, there are nine zone-center optical phonon modes; only three of the Ag + 2Bg modes are Raman-active. By comparing the vibrational spectra of M-CuO NRs, we were able to attribute the Ag mode's peak at 286 cm−1 and the Bg mode's peaks at 333 and 615 cm−1.42–44 In the Raman spectrum of BiOBr, there are two A1g, B1g, and Eg Raman-active modes. The excellent crystallinity is confirmed by the pointed peaks. The bands at 56.2, 94, and 111 cm−1 for BiOBr are attributed to the A1g internal Bi–Br stretching mode. The Eg Bi–Br stretching mode is assigned to the band at 158 cm−1.45 The B1g mode may be responsible for the band at 390 cm−1 produced by the motion of oxygen atoms.46 Three unique vibration peaks were seen in the Raman analysis of pure BiOI, and they were found at 55, 94 and 145 cm−1. These peaks matched the A1g and Eg phonon stretching modes of the Bi–I moieties, respectively.47,48
To learn more about the stability of each bare material, we performed thermogravimetric analysis (TGA) studies under O2 atmosphere. All samples experienced an initial weight loss due to the loss of physically adsorbed water molecules. Cu-BTC began to collapse when the temperature was raised to approximately 360 °C because of the polymer's backbone cleavage (carbonation), losing roughly 44% of its weight in the process (Fig. S2b, ESI†). M-CuO NRs experienced a phase change from CuO to Cu2O during heating to about 720 °C due to thermal breakdown. Cu(II) was reduced to Cu(I) in this process, and oxygen was released from the crystal structure in the process (Fig. S2b, ESI†).49 For BiOI, BiOBr, and BiOCl, the catalysts remain extremely stable up to temperatures of 368 °C, 536 °C, and 607 °C, respectively. With the additional increase in temperature for BiOI, BiOBr, and BiOCl, respectively, the impulsive breakdown of Bi2O3 and the halogen gases explained the noticeable weight loss of 36%, 16%, and 14%. For the intended experimental temperature range, the catalysts were found to be extremely stable overall (Fig. S8, ESI†).50
HR-XPS spectra of Cu 2p shows peaks at 934.3 and 954.2 eV corresponding Cu 2p3/2 and Cu 2p1/2, respectively, for M-CuO NRs. On the other hand, Cu 2p peaks in M-CuO NRs/BiOI shifted 0.7 eV to a lower binding energy, i.e., 933.6 eV and 953.5 eV for Cu 2p3/2 and Cu 2p1/2, respectively. Minor shift towards lower binding energy in XPS was observed in Cu 2p owing to the change/movement of electron density around M-CuO NRs in M-CuO NRs/BiOI compared to CuO (Fig. 2c). On the other hand, the decreased electron density over BiOI in M-CuO NRs/BiOI resulted in tightly bound electrons in Bi 4f, which led to increased binding energy of Bi 4f electrons compared to pristine BiOI. Along with Cu 2p peaks, satellite peaks were observed at 942.4 and 962.4 eV, which correspond to 3d–4s shakeup transitions, which were not observable in the filled 3d shells and metallic states. The broad O 1s core-level spectrum can be fitted to three distinct peaks denoted as O1, O2, and O3. O2 binding with Cu is represented by the O1 component at a lower energy of 529.9 eV. The presence of Cu and O was confirmed by the O3 component at 534.6 eV, which can be attributable to adsorbed molecular water. The O2 component at 531.7 eV is related to the chemisorbed oxygen on the surface of M-CuO NRs (Fig. 2d).51 Bi 4f peaks with binding energy of 158.4 eV and 163.7 eV are attributed to Bi 4f7/2 and Bi 4f5/2, respectively. However, on the fabrication of M-CuO NRs/BiOI, Bi 4f peaks shifted by 0.4 eV to a higher binding energy owing to a decrease in electron density, insinuating a shift in the electron cloud from BiOI to M-CuO NRs (Fig. 2e). Satellite peaks are at 1.0 eV away from the Bi 4f7/2 and Bi 4f5/2 main peaks, corresponding to the reported values.52 HR-XPS of I 3d peaks was observed with satellite peaks and a doublet centred at 618.6 eV and 630.1 eV for 3d5/2 and 3d3/2, respectively, in BiOI (Fig. 2f). A minor shift to a lower binding energy was observed in M-CuO NRs/BiOI with the doublet centered at 618.4 eV and 629.9 eV for 3d5/2 and 3d3/2, respectively. Change in the surrounding charge environment led to electron movement in the fabricated heterojunction from BiOI to M-CuO NRs, resulting in minor shifts in I 3d high-resolution XPS.53,54
Transmission electron microscopy analysis of M-CuO NRs (Fig. 3a) and HR-TEM showed a fringe width of 0.23 nm corresponding to (111) plane observed in the experimental XRD pattern (Fig. 3b). TEM micrographs of M-CuO NRs/BiOBr were observed with two different fringes corresponding to M-CuO NRs and BiOBr (0.28 nm, (011) plane) (Fig. 3d–f) along with the EDS pattern for the presence of corresponding elements Cu, O, Bi, and Br (Fig. 3g). Similarly, two different patterns with different fringe widths for M-CuO NRs and BiOI micro flowers were observed in M-CuO NRs/BiOI (Fig. 3h–j). EDS analysis of M-CuO NRs/BiOI showed the presence of elements, i.e., Cu, O, Bi, and I, and the SAED pattern showed a polycrystalline nature (Fig. 3k).
Photoelectrochemical studies
To conduct photoelectrochemical measurements, a PGSTAT302 N workstation (Metrohm instruments) was utilized to provide a three-electrode arrangement. The counter electrode (CE) was a Pt rod, while the reference electrode (RE) was a saturated Ag/AgCl electrode. AM 1.5G simulated solar light produced from a 150 W LED source and measured to 100 mW cm−2 was used to illuminate the front surface of the photoelectrodes in an area of 0.25 cm2. Measurements using electrochemical impedance spectroscopy (EIS) in a 0.1 M Na2SO4 solution were made at 0 V vs. RHE, with an AC amplitude of 20 mV and a frequency range from 0.1 Hz to 1 MHz.CuO photocathodes synthesised through various methods, i.e., hydrothermal, pyrolysis, calcination, and chemical treatment, were subjected to PEC characterizations using J–V polarization curves (Fig. 4a). CuO synthesized through Cu-BTC precursor's chemical treatment using NaOH followed by annealing (M-CuO NRs) resulted in the highest photocurrent density among other synthetic methods. We investigated the effect of time and annealing temperature for Cu-BTC chemical treatment in NaOH solution. Among different times, 15 min dipping of Cu-BTC in NaOH showed the highest photocurrent response (−4.1 mA cm−2 at 0 VRHE) (Fig. 4a and S9, ESI†). The pre-oxidation of Cu-BTC prior to annealing in the presence of air and morphological transformation to 1D nanorods led to significant photo-response compared to other counterparts as 1D nanostructures are known to have long diffusional length and time, showing decreased rate of recombination and faster charge transfer.51,55 The photocurrent density on the M-CuO NRs did not exhibit any appreciable degradation after a 3 h study. But the photocurrent density was stabilized at a much lower photocurrent than that obtained in LSV measurements. This leads to poor faradaic efficiency of the M-CuO NRs photoelectrodes.
The deposition of BiOX X: Cl, Br, and I on M-CuO NRs was tested and studied by modulating different parameters, i.e., amount of BiOX deposited and annealing temperature. In terms of thermal stability, BiOX were found to be air stable and their photoactivity degrade rapidly at about 300 °C. Three different concentrations of BiOX were drop-casted (BiOX_x representing the concentration) and optimal concentrated M-CuO NRs/BiOX_x photocathodes were subjected to different annealing temperatures of 90 °C, 150 °C, and 300 °C and then tested. The J–V polarization curve under continuous illumination of bare BiOX (X: Cl, Br, and I) was supplemented for comparison (Fig. S11, ESI†). M-CuO NRs/BiOCl_x (x: 3, 6, and 9) were tested having different concentrations of BiOCl. A decrease in the photocurrent density was observed in case of M-CuO NRs/BiOCl_3 (−3.4 mA cm−2), M-CuO NRs/BiOCl_6 (−2.2 mA cm−2) and M-CuO NRs/BiOCl_9 (−2.8 mA cm−2) at 0 VRHE compared to M-CuO NRs (−4.1 mA cm−2) (Fig. 4b). On annealing M-CuO NRs/BiOCl_3 at different temperatures, degradation in photoactivity was found owing to the dissociation of BiOCl. M-CuO NRs/BiOCl_150 and M-CuO NRs/BiOCl_300 showed identical photoactivity in J–V curves while J for M-CuO NRs/BiOCl_90, it remained −3.4 mA cm−2 at 0 VRHE (Fig. 4c). In previous reports, Zhang et al. fabricated an NiS/CuInS2/BiOCl photocathode with stable photoresponse for over 5.5 hours, achieving a hydrogen production rate of 0.048 μmol h−1 and 89.5% faradaic efficiency. BiOCl was employed as the hole transport layer in the device.56 (001)-oriented p-type BiOCl thin films, synthesized via mist CVD, exhibited enhanced near-UV absorption from oxygen vacancies. Films showed a higher residual carrier density of approximately −2.3 mA cm−2 compared to others.57
The deposition of BiOBr for fabricating M-CuO NRs/BiOBr_x (where x: 3, 6, and 9) led to an increment in photo-response only for M-CuO NRs/BiOBr_3 with J = −5.1 mA cm−2 while M-CuO NRs/BiOBr_6 (−3.6 mA cm−2) and M-CuO NRs/BiOBr_9 (−2 mA cm−2) at 0 VRHE showed a decrease in photoactivity (Fig. 4d). With increasing annealing temperature, the photoactivity further decreased, particularly at 150 °C (−1.8 mA cm−2) and 300 °C (−1.4 mA cm−2) at 0 VRHE (Fig. 4e). In the literature, BiOBr, when integrated with MoS2, formed a van der Waals heterojunction, stabilizing its lattice during the hydrogen evolution reaction (HER). 50%-MoS2/BiOBr yielded −16.5 mA cm−2, but the performance drops significantly after the second cycle due to BiOBr reduction, indicated by the irreversible redox peak of Bi3+ to Bi0.58 In another study by Shang et al., at an external bias of 2 V, the photocurrent density of the C/BiOBr composite rose to 7.74 mA cm−2, compared to 4.51 mA cm−2 for pristine BiOBr. This increase demonstrated effectiveness of C doping in enhancing the photocatalytic performance of BiOBr.46
BiOI microflowers' deposition over M-CuO NRs resulted in the enhancement of photocurrent density with M-CuO NRs/BiOI_6, showing highest J = −5.6 mA cm−2 and M-CuO NRs/BiOI_3, M-CuO NRs/BiOI_9 delivering J = −5.1 mA cm−2 and −4.3 mA cm−2 at 0 VRHE, respectively (Fig. 4f). Significant degradation in photocurrent density was observed at 150 °C (−2.2 mA cm−2) and 300 °C (−1.8 mA cm−2) at 0 VRHE, which is attributed to BiOI degradation/transformation to Bi2O3 (Fig. 4g). Enhancing BiOI-based photocathodes through the combination of hydrogenation, nanostructuring, and plasmonic effects (Au NPs) has been shown to be a successful tactic. Au@H-BiOI photocathodes attained a photocurrent density of 3.6 ± 0.1 mA cm−2 at 0 VRHE under AM 1.5G irradiation, with over 80% faradaic efficiency. This is due to their high surface area of nanostructured films, increased electrical conductivity, and plasmonic effects.59 Optimized n-type BiOI nanoplatelet thin films exhibit photocurrents of ∼0.9 mA cm−2 at 0.4 VAg/AgCl. This performance results from a balanced combination of favourable morphology, high crystallinity, controlled impurity levels, and effective optical absorption.60 Kinetics of charge generation and transfer mechanism influences the photostability of the electrodes. The highest half-cell solar-to-hydrogen conversion efficiency was recorded for M-CuO NRs/BiOI (0.95% @ 0.34 VRHE) and M-CuO NRs/BiOBr (0.85% @ 0.4 VRHE) (Fig. 4i). Chronoamperometric analysis of the fabricated photoelectrodes revealed higher stability of M-CuO NRs/BiOI compared to M-CuO NRs/BiOBr and M-CuO NRs (Fig. 4j). Deposition of BiOI microflowers on M-CuO NRs was able to surpass the inherent photocorrosion property of CuO to some extent. The XRD patterns of the M-CuO NRs and M-CuO NRs/BiOI photoelectrodes before and after photostability studies are provided (Fig. S12 and S13, ESI†). A notable reduction in peak intensity is likely attributed to the diminished crystallinity of the material following photostability studies. Furthermore, the photoreduction of CuO to the Cu2O phase is lesser in the M-CuO NRs/BiOI photoelectrode when compared to the bare M-CuO NRs photoelectrode. The fewer Cu2O phases after stability studies clearly indicate this observation and thereby strengthen the influence of BiOI on the stability of M-CuO NRs.
Through statistical analysis, 5 photocathodes of each type were tested under continuous illumination of AM 1.5G. Box plots of PEC parameters such as photocurrent density and Eonset are depicted in Fig. 4k and l, respectively. The reference for Eonset is considered as the potential at which the photocurrent density reaches 0.1 mA cm−2vs. RHE. The photocurrent density of M-CuO NRs/BiOI showed higher J among others (Fig. 4k); meanwhile, the onset potential for M-CuO NRs/BiOBr was found to be higher than that of M-CuO NRs/BiOI (Fig. 4l). The fabricated M-CuO NRs/BiOI and M-CuO NRs/BiOBr have shown promising performance in comparison with the other reported photoelectrode materials (Table S5, ESI†).
Optical studies
To understand the optical properties, UV-DRS was performed over various samples including M-CuO NRs synthesized through different synthetic protocols, BiOX, and M-CuO NRs/BiOX (X: Cl, Br, and I) (Fig. 5a–c). M-CuO NRs shows a broad absorption spectrum in the wavelength range between 300 and 800 nm, and the estimated direct band gap of 1.8 eV calculated through Kubelka–Munk (K–M) function. The band gap of H-CuO and M-CuO (O2) was evaluated to be 1.52 and 1.56 eV, respectively (Fig. S10, ESI†). A smoother surface and more specular reflection as opposed to diffuse reflection were suggested by the more oscillations in the H-CuO nanoflake reflectance spectra.51 M-CuO NRs films, on the other hand, have rougher surfaces compared to other nanostructures, which result in longer optical pathways for light within the films and more diffuse reflectance. Nanorods efficiently capture solar light and increase the carrier diffusion length due to the random distribution of particles. The suitable nanostructure of M-CuO NRs allows for enhanced light harvesting because of a strong light scattering effect and less reflection. BiOI microflowers showed an intense absorption with an absorption edge starting from 625 nm, BiOBr showed an absorption starting at 400 nm, and BiOCl absorbed at 350 nm (Fig. 5b). The bandgap calculated using the K–M plot for BiOI, BiOBr, and BiOCl was 1.8, 2.8, and 3.3 eV, respectively. After the deposition of BiOX (X: Cl, Br, and I) on M-CuO NRs, the absorption characteristics of the fabricated heterojunction of M-CuO NRs/BiOX (X: Br and I) were enhanced except for M-CuO NRs/BiOCl compared to M-CuO NRs (Fig. 5c). Following absorption analysis, the emission characteristics of M-CuO NRs and M-CuO NRs/BiOX (X: Cl, Br, and I) revealed the rate of recombination through steady state photoluminescence spectroscopy (PL) (Fig. 5d). PL spectrum of the synthesized nanocomposite was obtained at an excitation wavelength (λex) of 370 nm. A sluggish rate of exciton recombination is typically associated with weak, intense peaks, indicating a prolonged lifetime for excitons.61 M-CuO NRs/BiOBr and M-CuO NRs/BiOI revealed lower rate of recombination as compared to M-CuO NRs, while M-CuO NRs/BiOCl revealed a higher rate of recombination attributed to the decreased intensity of emission for M-CuO NRs/BiOX (X: Br and I) (Fig. 5d). Time-resolved photoluminescence spectroscopy (TRPL) revealed an elongated lifetime of photogenerated charge carrier in M-CuO NRs/BiOX (X: Cl, Br, and I). M-CuO NRs/BiOI (1.75 ns) lifetime is 4-fold higher than that of M-CuO NRs (0.48 ns) while that of M-CuO NRs/BiOBr and M-CuO NRs/BiOCl was 0.63 ns and 0.45 ns, respectively (Fig. 5e and Table S3, ESI†).Incident photon-to-current conversion efficiency (IPCE%) of M-CuO NRs, M-CuO NRs/BiOX_x (x: Cl, Br, and I) was measured to study the wavelength-dependent photo-response along with integrated photocurrent density (Jsc) under AM 1.5G illumination (Fig. 6a). M-CuO NRs/BiOI was observed with the highest IPCE (%) of ∼28% at 675 nm with Jsc = 6.3 mA cm−2, M-CuO NRs/BiOBr showed 15% at 675 nm with Jsc = 4.3 mA cm−2, M-CuO NRs showed 12% at 675 nm with Jsc = 3.5 mA cm−2 and M-CuO NRs/BiOCl showed lowest 7% at 675 nm with Jsc = 1.7 mA cm−2. Below 900 nm, M-CuO NRs/BiOI showed higher IPCE than M-CuO NRs/BiOBr and M-CuO NRs. Evaluation of charge transfer phenomenon and kinetics of M-CuO NRs/BiOX (X: Br and I) through electrochemical impedance spectroscopy (EIS) analysis revealed a lower charge transfer resistance (Rct) for M-CuO NRs/BiOI (2.74 kΩ) compared to that of M-CuO NRs/BiOBr (3.15 kΩ) and M-CuO NRs (3.96 kΩ) owing to a swift charge transfer through bulk and faster surficial reaction (Fig. 6b) (Table S1, ESI†).62 Correlating EIS with the Bode phase plot, M-CuO NRs/BiOI with lower phase difference and elongated electron lifetime indicates longer diffusion length of a photogenerated electron. However, a large phase difference in M-CuO NRs/BiOBr indicates slow kinetics of charge transfer compared to M-CuO NRs/BiOI but a longer electron lifetime than M-CuO NRs means better charge separation at the heterojunction (Fig. 6c and Table S2, ESI†). The impact of BiOX (X: Cl, Br, and I) on the bulk charge separation efficiency and surface charge transfer efficiency of M-CuO NRs was analysed (Fig. S14, ESI†). The J–V profile of the photocathodes was examined in a 0.1 M Na2SO4 (pH 6.18) electrolyte, incorporating H2O2 as an electron scavenger to guarantee the thorough capture of interfacial photogenerated electrons. M-CuO NRs/BiOI demonstrated a notably enhanced bulk charge separation efficiency and surface charge transfer efficiency (ηsep = 43%, ηct = 40%) at 0 V_RHE when compared to the bare M-CuO NRs (ηsep = 38%, ηct = 28%). In the meantime, M-CuO NRs/BiOBr exhibited a bulk charge separation efficiency of 41% and a surface charge transfer efficiency of 36%. Conversely, M-CuO NRs/BiOCl demonstrated the lowest bulk charge separation efficiency at 35% and a surface charge transfer efficiency of 28%.
Mechanistic and charge transfer studies
Mott–Schottky plots (M–S) of M-CuO NRs, BiOX (X: Cl, Br, and I) recorded at 1 kHz reveal the p-type nature of M-CuO NRs and n-type nature of BiOX (X: Cl, Br, and I) with different flat band potentials (EFB). EFB for p-type M-CuO NRs represents the valence band aligned at 0.98 V while for n-type BiOX, EFB represents a conduction band at −0.83 V, −0.51, and 0.53 V for BiOCl, BiOBr and BiOI, respectively (Fig. 6d–g and Table S4, ESI†). Bandgap, conduction and valence band positions were calculated using electrochemical and absorption studies of thin film semiconductor photocathodes (Scheme 1).63,64 Charge transfer pathways inferred from the PEC, absorption, emission characterizations and mechanistic studies revealed a Z-scheme of charge transfer between M-CuO NRs and BiOI in M-CuO NRs/BiOI, while M-CuO NRs/BiOBr revealed a type-II heterojunction and type-I scheme of charge transfer in the M-CuO NRs/BiOCl photocathode. To manifest Z scheme mechanism in M-CuO NRs/BiOI, O2˙− trapping experiments were conducted using nitro blue tetrazolium chloride (NBT) as a molecular probe showed that NBT reacts with O2˙− radicals in a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4 ratio to form a formazan derivative,65 with decreased UV absorption at 260 nm indicating O2˙− formation (Fig. 6h). The absorbance vs. irradiation time plots of NBT aqueous solution for different time intervals are presented in Fig. 6h. Based on the ability of negative CuO conduction band potential to reduce dioxygen to O2˙− under visible light, the migration of charge carriers in a Z-scheme mechanism was confirmed in this system.66 The reduction potential of O2 to O2˙− is roughly −0.33 eV compared to NHE. Given that the conduction band of BiOI is positioned at +0.53 eV, the formation of superoxide radicals is not feasible. In the case of M-CuO NRs, with the conduction band positioned at −0.82 eV, the generation of O2˙− is noted. The NBT that reacted with the M-CuO NRs/BiOI binary composite shows decreased absorbance at 260 nm in NBT spectral analysis, which confirms the formation of superoxide on the M-CuO NRs unit and supports a Z-scheme charge transfer mechanism. The significant decrease in NBT absorption over time for M-CuO NRs/BiOI relative to pristine M-CuO NRs suggests suppressed charge recombination and improved electron accumulation on M-CuO NRs, which promotes effective O2 reduction to O2˙− (Fig. 6i). The experimental details for O2˙− trapping and the absorbance vs. irradiation time plots of NBT aqueous solution after reaction with M-CuO NRs (Fig. S15, ESI†) are given in the ESI.†67,68
4 ratio to form a formazan derivative,65 with decreased UV absorption at 260 nm indicating O2˙− formation (Fig. 6h). The absorbance vs. irradiation time plots of NBT aqueous solution for different time intervals are presented in Fig. 6h. Based on the ability of negative CuO conduction band potential to reduce dioxygen to O2˙− under visible light, the migration of charge carriers in a Z-scheme mechanism was confirmed in this system.66 The reduction potential of O2 to O2˙− is roughly −0.33 eV compared to NHE. Given that the conduction band of BiOI is positioned at +0.53 eV, the formation of superoxide radicals is not feasible. In the case of M-CuO NRs, with the conduction band positioned at −0.82 eV, the generation of O2˙− is noted. The NBT that reacted with the M-CuO NRs/BiOI binary composite shows decreased absorbance at 260 nm in NBT spectral analysis, which confirms the formation of superoxide on the M-CuO NRs unit and supports a Z-scheme charge transfer mechanism. The significant decrease in NBT absorption over time for M-CuO NRs/BiOI relative to pristine M-CuO NRs suggests suppressed charge recombination and improved electron accumulation on M-CuO NRs, which promotes effective O2 reduction to O2˙− (Fig. 6i). The experimental details for O2˙− trapping and the absorbance vs. irradiation time plots of NBT aqueous solution after reaction with M-CuO NRs (Fig. S15, ESI†) are given in the ESI.†67,68
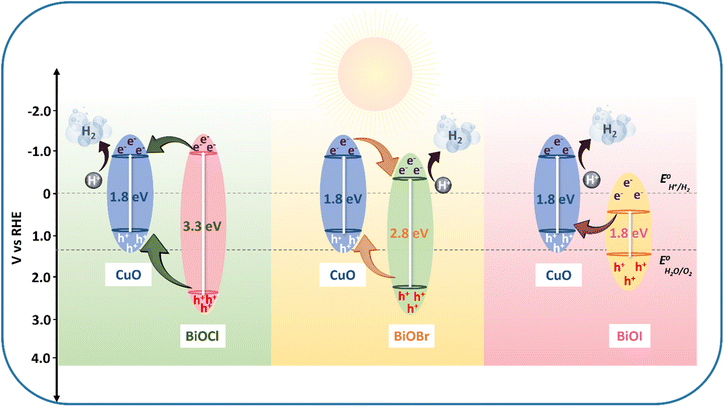 | ||
| Scheme 1 Proposed charge transfer mechanism in the p–n heterojunction of M-CuO NRs/BiOCl, M-CuO NRs/BiOBr, and M-CuO NRs/BiOI. | ||
Conclusions
In this work, we outlined an inventive strategy to modulate the charge transfer mechanisms and PEC performances of heterojunction photocathodes by tuning halides in BiOX (X = Cl, Br, I) deposited on M-CuO NRs. Diverging from the conventional, energy-intensive pyrolysis route for metal oxide synthesis from 3D-MOFs, a more efficient and eco-friendlier alternative was explored. The deposition of BiOI microflowers onto M-CuO NRs significantly enhanced the photocurrent density, with M-CuO NRs/BiOI_6 achieving the highest performance, exhibiting a photocurrent density of J = −5.6 mA cm−2 and HC-STH of 0.95%. Meanwhile, BiOBr deposition resulted in J = −5.1 mA cm−2 at 0 VRHE and HC-STH of 0.85% at 0.40 VRHE compared to M-CuO NRs (J = −4.1 mA cm−2 at 0 VRHE, HC-STH of 0.40% at 0.40 VRHE). M-CuO NRs/BiOI exhibited the highest IPCE (∼28%) at 675 nm, corresponding to an integrated current density (Jsc) of 6.3 mA cm−2. In comparison, M-CuO NRs/BiOBr achieved an IPCE of 15% at 675 nm with Jsc = 4.3 mA cm−2, while M-CuO NRs showed the lowest IPCE of 12% at 675 nm with Jsc = 3.5 mA cm−2. The inherent photocorrosion tendency of CuO was mitigated through the deposition of BiOX, enhancing its stability under photoelectrochemical conditions. Time-resolved photoluminescence spectroscopy (TRPL) revealed an extended lifetime of photogenerated charge carriers in M-CuO NRs/BiOX (X = Cl, Br, I). Among these, M-CuO NRs/BiOI exhibited the longest lifetime at 1.75 ns, which is approximately four times higher than that of bare M-CuO NRs (0.48 ns). In addition, the lifetimes for M-CuO NRs/BiOBr and M-CuO NRs/BiOCl were 0.63 ns and 0.45 ns, respectively.As the halide in BiOX transitioned from Cl to I, the charge transfer mechanism evolved from a Type I alignment to a Type II band alignment and ultimately to a Z-scheme mechanism. This progression was attributed to the systematic modulation of the electronic structure and band energetics, driven by the increasing electronegativity and orbital contributions of the halides.
Data availability
The data supporting this article have been included as part of the ESI,† including Chemicals, Characterization techniques, XRD, TGA, SEM, Kubelka–Munk's plot, PEC measurements, LSV polarization curves, Tables for EIS equivalent circuit parameters, Band positions, TRPL lifetime parameters and performance comparison with previous reports.Conflicts of interest
Authors declare no competing conflict of interest.Acknowledgements
S. J. sincerely acknowledges the Department of Science and Technology (India) for the INSPIRE initiative, which provided fellowship to pursue PhD. M. K. gratefully thanks CSIR-India for the Senior Research Fellowship and SUPRA fellowship from Swinburne University of Technology. C. S. acknowledges JICA Friendship 2.0 programme & Greenko School of sustainability for the funding. Authors would also like to extend their special thanks to Dr Poondla Vijayakumar for the XPS and TGA characterizations, Mrs Melody P. R. C. for XRD characterizations and Ms. N. J. L. Sivani for SEM characterizations.References
- J. Su and L. Vayssieres, ACS Energy Lett., 2016, 1, 121–135 CrossRef CAS.
- A. Hussain, S. M. Arif and M. Aslam, Renewable Sustainable Energy Rev., 2017, 71, 12–28 CrossRef.
- J. K. Yadav, B. Rani, P. Saini and A. Dixit, Energy Adv., 2024, 3, 927–944 RSC.
- M. Joseph, B. Meena, R. Joy, S. Joseph, R. K. Sethi, S. N. Remello, S. Haridas and C. Subrahmanyam, Sustainable Energy Fuels, 2024, 8, 3412–3418 RSC.
- E. Kalamaras, M. M. Maroto-Valer, M. Shao, J. Xuan and H. Wang, Catal. Today, 2018, 317, 56–75 CrossRef CAS.
- T. Zhou, J. Wang, S. Chen, J. Bai, J. Li, Y. Zhang, L. Li, L. Xia, M. Rahim, Q. Xu and B. Zhou, Appl. Catal., B, 2020, 267, 118599 CrossRef CAS.
- T. Zhou, S. Chen, L. Li, J. Wang, Y. Zhang, J. Li, J. Bai, L. Xia, Q. Xu, M. Rahim and B. Zhou, Appl. Catal., B, 2020, 269, 118776 CrossRef CAS.
- T. Zhou, L. Li, J. Li, J. Wang, J. Bai, L. Xia, Q. Xu and B. Zhou, Chem. Eng. J., 2021, 425, 131770 CrossRef CAS.
- T. Zhou, S. Chen, J. Wang, Y. Zhang, J. Li, J. Bai and B. Zhou, Chem. Eng. J., 2021, 403, 126350 CrossRef CAS.
- T. Zhou, J. Wang, Y. Zhang, C. Zhou, J. Bai, J. Li and B. Zhou, Chem. Eng. J., 2022, 431, 133414 CrossRef CAS.
- M. H. Miah, M. U. Khandaker, M. B. Rahman, M. Nur-E-Alam and M. A. Islam, RSC Adv., 2024, 14, 15876–15906 RSC.
- S. Meloni, G. Palermo, N. Ashari-Astani, M. Grätzel and U. Rothlisberger, J. Mater. Chem. A, 2016, 4, 15997–16002 RSC.
- D. P. McMeekin, G. Sadoughi, W. Rehman, G. E. Eperon, M. Saliba, M. T. Hörantner, A. Haghighirad, N. Sakai, L. Korte, B. Rech, M. B. Johnston, L. M. Herz and H. J. Snaith, Science, 2016, 351, 151–155 CrossRef CAS PubMed.
- Z.-Q. Wang, H. Wang, X.-F. Wu and T.-L. Chang, J. Alloys Compd., 2020, 834, 155025 CrossRef CAS.
- K.-H. Ye, Z. Chai, J. Gu, X. Yu, C. Zhao, Y. Zhang and W. Mai, Nano Energy, 2015, 18, 222–231 CrossRef CAS.
- C. Liu, J. Zhou, J. Su and L. Guo, Appl. Catal., B, 2019, 241, 506–513 CrossRef CAS.
- A. Sudharani, K. Sunil Kumar, R. Mangiri, Y. C. Ratnakaram and R. P. Vijayalakshmi, Mater. Chem. Phys., 2021, 258, 123891 CrossRef CAS.
- L. Zhang, Y. F. Ruan, Y. Y. Liang, W. W. Zhao, X. D. Yu, J. J. Xu and H. Y. Chen, ACS Appl. Mater. Interfaces, 2018, 10, 3372–3379 CrossRef CAS PubMed.
- M. E. Kazyrevich, M. V. Malashchonak, A. V. Mazanik, E. A. Streltsov, A. I. Kulak and C. Bhattacharya, Electrochim. Acta, 2016, 190, 612–619 CrossRef CAS.
- M. Long, P. Hu, H. Wu, Y. Chen, B. Tan and W. Cai, J. Mater. Chem. A, 2015, 3, 5592–5598 RSC.
- S. K. Poznyak and A. I. Kulak, Electrochim. Acta, 1990, 35, 1941–1947 CrossRef CAS.
- P. Kwolek and K. Szaciłowski, Electrochim. Acta, 2013, 104, 448–453 CrossRef CAS.
- L. Shi, D. Benetti, F. Li, Q. Wei and F. Rosei, Appl. Catal., B, 2020, 263, 118317 CrossRef CAS.
- T. Jiao, C. Lu, D. Zhang, K. Feng, S. Wang, Z. Kang and J. Zhong, Appl. Catal., B, 2020, 269, 118768 CrossRef CAS.
- C. Hou, T. Li, Y. Chen and W. Fu, Chempluschem, 2015, 80, 1465–1471 CrossRef CAS PubMed.
- L. Zhang, P. Cui, H. Yang, J. Chen, F. Xiao, Y. Guo, Y. Liu, W. Zhang, F. Huo and B. Liu, Adv. Sci., 2015, 3, 1–6 Search PubMed.
- L. Zhang, P. Cui, H. Yang, J. Chen, F. Xiao, Y. Guo, Y. Liu, W. Zhang, F. Huo and B. Liu, Adv. Sci., 2019, 6, 1802373 CrossRef PubMed.
- S. R. Vaddipalli, S. R. Sanivarapu, S. Vengatesan, J. B. Lawrence, M. Eashwar and G. Sreedhar, ACS Appl. Mater. Interfaces, 2016, 8, 23049–23059 CrossRef CAS PubMed.
- S. Zhou, K. Chen, J. Huang, L. Wang, M. Zhang, B. Bai, H. Liu and Q. Wang, Appl. Catal., B, 2020, 266, 118513 CrossRef CAS.
- W. Zhan, Q. Kuang, J. Zhou, X. Kong, Z. Xie and L. Zheng, J. Am. Chem. Soc., 2013, 135, 1926–1933 CrossRef CAS PubMed.
- W. Yang, R. R. Prabhakar, J. Tan, S. D. Tilley and J. Moon, Chem. Soc. Rev., 2019, 48, 4979–5015 RSC.
- G. Yilmaz, C. F. Tan, Y. Lim and G. W. Ho, Adv. Energy Mater., 2019, 9, 1802983 CrossRef.
- R. Dutta, R. Shrivastav, M. Srivastava, A. Verma, S. Saxena, N. K. Biswas, V. R. Satsangi and S. Dass, Int. J. Hydrogen Energy, 2022, 47, 5192–5210 CrossRef CAS.
- M. Huang, K. Mi, J. Zhang, H. Liu, T. Yu, A. Yuan, Q. Kong and S. Xiong, J. Mater. Chem. A, 2017, 5, 266–274 RSC.
- J.-S. Li, S.-L. Li, Y.-J. Tang, K. Li, L. Zhou, N. Kong, Y.-Q. Lan, J.-C. Bao and Z.-H. Dai, Sci. Rep., 2014, 4, 5130 CrossRef CAS PubMed.
- D. Cardenas-Morcoso, R. Ifraemov, M. García-Tecedor, I. Liberman, S. Gimenez and I. Hod, J. Mater. Chem. A, 2019, 7, 11143–11149 RSC.
- L. Shi, D. Benetti, Q. Wei and F. Rosei, Small, 2019, 19, 2300606 CrossRef PubMed.
- L. Armelao, G. Bottaro, C. Maccato and E. Tondello, Dalton Trans., 2012, 41, 5480 RSC.
- Y. Song, X. Li, C. Wei, J. Fu, F. Xu, H. Tan, J. Tang and L. Wang, Sci. Rep., 2015, 5, 8401 CrossRef PubMed.
- N. J. Calos, J. S. Forrester and G. B. Schaffer, J. Solid State Chem., 1996, 122, 273–280 CrossRef CAS.
- M. Rashad, M. Rüsing, G. Berth, K. Lischka and A. Pawlis, J. Nanomater., 2013, 2013, 1 Search PubMed.
- W. Reichardt, F. Gompf, M. Aïn and B. M. Wanklyn, Z. Phys. B: Condens. Matter., 1990, 81, 19–24 CrossRef CAS.
- J. Chrzanowski and J. C. Irwin, Solid State Commun., 1989, 70, 11–14 CrossRef CAS.
- J. F. Xu, W. Ji, Z. X. Shen, W. S. Li, S. H. Tang, X. R. Ye, D. Z. Jia and X. Q. Xin, J. Raman Spectrosc., 1999, 30, 413–415 CrossRef CAS.
- J. Wang, Y. Zhang, L. Tian, F. Liu and Q. Xia, J. Nanopart. Res., 2014, 16, 2691 CrossRef.
- J. Shang, Y. Gao, B. Zhao, S. Shen and X. Wang, J. Mater. Sci.:Mater. Electron., 2020, 31, 20858–20867 CrossRef CAS.
- P. Wu, L. Feng, Y. Liang, X. Zhang, X. Li, S. Tian, H. Hu, G. Yin and S. Khan, J. Mater. Sci.:Mater. Electron., 2020, 31, 5385–5401 CrossRef CAS.
- J. E. D. Davies, J. Inorg. Nucl. Chem., 1973, 35, 1531–1534 CrossRef CAS.
- S. R. Son, K. S. Go and S. D. Kim, Ind. Eng. Chem. Res., 2009, 48, 380–387 CrossRef CAS.
- B. T. Shiferraw, A. F. Baye and H. Kim, Int. J. Hydrogen Energy, 2024, 51, 435–450 CrossRef CAS.
- S. Cheng, X. Gao, S. DelaCruz, C. Chen, Z. Tang, T. Shi, C. Carraro and R. Maboudian, J. Mater. Chem. B, 2019, 7, 4990–4996 RSC.
- H. Liu, W. Cao, Y. Su, Y. Wang and X. Wang, Appl. Catal., B, 2012, 111–112, 271–279 CrossRef CAS.
- Q. Geng, H. Xie, W. Ji, Y. Li, S. Chen, B. Deng, R. Jin and F. Dong, J. Phys. Chem. C, 2024, 128, 8331–8340 CrossRef CAS.
- X. Zhang, W. Yang, M. Gao, H. Liu, K. Li and Y. Yu, Green Energy Environ., 2022, 7, 66–74 CrossRef CAS.
- T. R. Harris-Lee, F. Marken, C. L. Bentley, J. Zhang and A. L. Johnson, EES Catal., 2023, 1, 832–873 RSC.
- Y. Zhang, Y. Dong, G. Wang, P. Jiang, S. Zhao, Y. Li, X. Wu, H. Miao, J. Li, J. Lyu, Y. Wang and Y. Zhu, Catal. Lett., 2020, 150, 1337–1345 CrossRef CAS.
- Z. Sun, S. Qin, D. Oka, H. Zhang, T. Fukumura, Y. Matsumoto and B. Mei, Inorg. Chem., 2023, 62, 8914–8922 CrossRef CAS PubMed.
- M. Wang, S. Osella, B. Torre, M. Crisci, F. Schmitz, R. Altieri, E. Di Fabrizio, H. Amenitsch, B. Sartori, Z. Liu, T. Gatti and F. Lamberti, ChemCatChem, 2024, 16, e202400282 CrossRef CAS.
- J. Gan, B. B. Rajeeva, Z. Wu, D. Penley and Y. Zheng, Electrochim. Acta, 2016, 219, 20–27 CrossRef CAS.
- N. T. Hahn, S. Hoang, J. L. Self and C. B. Mullins, ACS Nano, 2012, 6, 7712–7722 CrossRef CAS PubMed.
- S. Kumar, Bhawna, A. Gupta, R. Kumar, A. Bharti, A. Kumar and V. Kumar, J. Phys. Chem. C, 2023, 127, 7095–7106 CrossRef CAS.
- J. K. Yadav, B. Rani, P. Saini, A. P. Pandey and A. Dixit, Sustainable Energy Fuels, 2024, 8, 4617–4627 RSC.
- W. Guo, W. D. Chemelewski, O. Mabayoje, P. Xiao, Y. Zhang and C. B. Mullins, J. Phys. Chem. C, 2015, 119, 27220–27227 CrossRef CAS.
- R. Joy, B. Meena, M. Kumar, M. Jospeh, S. Joseph, C. Subrahmanyam and S. Haridas, Catal. Today, 2024, 433, 114669 CrossRef CAS.
- H. Huang, S. Tu, C. Zeng, T. Zhang, A. H. Reshak and Y. Zhang, Angew. Chem., Int. Ed., 2017, 56, 11860–11864 CrossRef CAS PubMed.
- Y. Su, X. Yu, X. Fu, Q. Zhu, L. Liu, Y. Zhu and Y. Zhang, Electrochim. Acta, 2020, 344, 136144 CrossRef CAS.
- D. Prusty, S. Mansingh, L. Acharya, L. Paramanik and K. M. Parida, RSC Adv., 2022, 12, 1265–1277 RSC.
- D. Kandi, D. P. Sahoo, S. Martha and K. Parida, Adv. Mater. Interfaces, 2019, 6, 1900370 CrossRef.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: https://doi.org/10.1039/d4ta08319a |
| This journal is © The Royal Society of Chemistry 2025 |






