 Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
High-performance electronic, optical, and thermoelectric properties of 2D Mg2In2S5 monolayer for energy applications
Zahid Ullah *ab,
Rajwali Khan*c,
Muhammad Amir Khanb,
Sattam Al Otaibid,
Khaled Althubeitie,
Mukhlisa Soliyevaf,
Muhammad Siyar
*ab,
Rajwali Khan*c,
Muhammad Amir Khanb,
Sattam Al Otaibid,
Khaled Althubeitie,
Mukhlisa Soliyevaf,
Muhammad Siyar g,
Noureddine Elboughdiri
g,
Noureddine Elboughdiri hi,
Asad Alij and
Shahid Iqbal
hi,
Asad Alij and
Shahid Iqbal *k
*k
aDepartment of Physics, Islamia College University, Peshawar, Pakistan. E-mail: zuzohaad@gmail.com
bFaculty of Physical and Numerical Sciences, Qurtuba University of Science and Information Technology, Peshawar, Pakistan
cNational Water and Energy Center, United Arab Emirates University, Al Ain, 15551, United Arab Emirates. E-mail: rajwali@uaeu.ac.ae
dDepartment of Electrical Engineering, College of Engineering, Taif University, P.O. Box 11099, 21944 Taif, Saudi Arabia
eDepartment of Chemistry, College of Science, Taif University, P.O. BOX. 110, 21944 Taif, Saudi Arabia
fNational Pedagogical University of Uzbekistan, Tashkent, Uzbekistan
gSchool of Chemical and Materials Engineering (SCME), National University of Sciences and Technology, NUST, H12, Islamabad, Pakistan
hChemical Engineering Department, College of Engineering, University of Ha'il, P.O. Box 2440, Ha'il, 81441, Saudi Arabia
iChemical Engineering Process Department, National School of Engineers, Gabes, University of Gabes, Gabes, 6029, Tunisia
jFaculty of Chemical and Life Sciences, Department of Chemistry, Abdul Wali Khan University Mardan, 23200, Pakistan
kDepartment of Physics, University of Wisconsin, La-Crosse, USA. E-mail: siqbal@uwlax.edu
First published on 9th December 2025
Abstract
Two-dimensional (2D) chalcogenide materials have recently attracted significant research interest due to their exceptional anisotropic properties and tunable electronic structures, making them strong contenders for advanced thermoelectric and optoelectronic technologies. In this work, we conduct a detailed first-principles study to explore the structural, electronic, optical, and thermoelectric properties of the Mg2In2S5 monolayer. Phonon dispersion and calculated elastic constants confirm the material's dynamic and mechanical stability. Electronic band structure analysis reveals a direct band gap semiconductor with a moderate band gap of 1.76 eV, making it suitable for visible light absorption. The partial density of states shows notable hybridization between In-5p and S-3p orbitals, which plays a key role in charge transport behavior. Optical simulations highlight strong anisotropy in the dielectric function and absorption spectra, with pronounced absorption in the UV-visible range, underscoring the material's potential in photonic and solar energy applications. Thermoelectric properties, evaluated using the Boltzmann transport formalism, display directional dependence, high Seebeck coefficients, and strong power factors. Remarkably, the figure of merit (ZT) reaches values as high as 1.0 in-plane and 1.2 out-of-plane at elevated temperatures, indicating excellent performance for high-temperature thermoelectric applications. Overall, the Mg2In2S5 monolayer demonstrates outstanding optoelectronic and thermoelectric characteristics, positioning it as a highly promising 2D material for energy harvesting and future nanoelectronic technologies.
1 Introduction
In the late twentieth century, 2D monolayer materials gained immense importance due to their exceptional physical and chemical properties compared to their bulk counterparts. High carrier mobility, a large surface-to-volume ratio, mechanical flexibility, tunable band gaps, and strong quantum confinement effects are just a few of the remarkable characteristics that make these atomically thin layers excellent for use in energy harvesting, catalysis, nano electronics, and optoelectronics.1 Because it is possible to manipulate material characteristics at the atomic level by altering composition, thickness, and bonding conditions, there is a great deal of interest in finding novel 2D materials other than conventional systems like graphene and transition metal dichalcogenides (TMDs). As compared to perovskite materials,2,3 ternary monolayer materials offer a rich chemical design space with more functional property tunability in this regard.4 Among these, Mg2In2S5 monolayer is special and has promising properties owing to its elemental composition, which is abundant on Earth, its expected semiconducting nature, and its potential for light-driven applications such as thermoelectric energy conversion and solar energy harvesting.In addition to binary compounds, the 2D materials family has expanded to include complex ternary systems such as di-metal chalcogenides (MmNnXx), layered double hydroxides, and AB2X4 spinels.5,6 However, ternary magnesium-based chalcogenides are mainly unexplored in the current study since it mainly focuses on metal phosphorus trichalcogenides (MPX3), TMDs (e.g., MoS2, WS2), and other layered sulfides and selenides. Despite the bulk form of Mg2Al2Se5 being synthesized as early as 1976, its monolayer derivatives have not been the focus of any significant theoretical or experimental research.7,8 The current study of Mg2In2S5 material is based on Mg2Al2Se5 to investigate the future properties. The growing need for new materials that have suitable band gap alignment and band gaps for solar energy applications makes this gap even more apparent. Magnesium and aluminium are a good combination since Al helps with electrical insulation and structural stability while Mg lacks low-lying d-orbitals, which usually leads to broad band gaps.9 Furthermore, because tellurium (X = S) has a higher atomic mass and a stronger spin–orbit coupling than selenium, it is possible to fine-tune the band structure and optical behavior. However, there is currently no systematic understanding of Mg2Al2X5 monolayers in the literature, particularly with regard to their structural, electrical, optical, and thermoelectric properties. The Mg2In2S5 composition has not been separately analysed for its coupled electrical, optical, and thermoelectric behaviour, despite recent studies theoretically examining the larger Mg2M2X5 family. The current work offers the first principles comprehensive DFT-based evaluation of the anisotropic characteristics, phonon stability, and energy-conversion potential of this monolayer, expanding our knowledge of magnesium-based ternary chalcogenides in a relevant and useful context. There is a substantial research gap due to this lack of understanding. Monolayer Mg2Al2X compounds have not yet been effectively theoretically modeled or produced experimentally, despite their significant elemental composition and predicted semiconducting properties.10,11 The potential of layered ternary chalcogenides for a range of functional applications, including as energy conversion and catalysis, has been shown by a few recent high-throughput DFT and machine-learning investigations. For example, in the nineteen century, the scientists categorized more than 450 layered chalcogenides and emphasized the promise of systems based on magnesium, such as MgAl4. Additionally, machine learning-driven interatomic potentials have made it feasible to predict attributes across wide chemical spaces by quickly screening untested combinations with DFT-level precision.12,13 These materials predicted wide-to-moderate band gaps and low lattice thermal conductivity, making them extremely promising as solar energy harvesters and thermoelectric generators in the context of the world's need for clean energy. However, this family of materials has not been thoroughly evaluated for properties in the existing corpus of literature. In nations like Pakistan, where there is a pressing need for affordable, locally generated, and renewable energy solutions, this disparity is particularly pertinent.
Therefore, the aim of this study is to use first-principles density functional theory (DFT) to comprehensively investigate and evaluate the structural stability, electronic band structure, optical response, and thermoelectric performance of monolayer Mg2In2S5. The objective is to assess the suitability for energy conversion technologies, including solar energy harvesting and thermoelectric generation, in addition to verifying their feasibility as stable 2D materials.14,15 Our objective is to model the monolayer geometry employing known bulk structures, calculate electronic properties, evaluate optical absorption spectra and dielectric response in the visible range, and calculate thermoelectric parameters such as electronic thermal conductivity, Seebeck coefficient, and figure of merit. The current study goal is to calculate the Mg2In2S5 monolayer's structural, electronic, optical, and thermoelectric characteristics. This research promotes its development of innovative 2D semiconductors for local and global sustainable energy applications while also filling a major gap in the scientific literature.
2 Computational simulations
First-principles calculations based on Density Functional Theory (DFT) have become a powerful tool for investigating the structural, electronic, optical, and thermoelectric properties of complex materials.16,17 The many-body Schrödinger equation is replaced by Kohn–Sham equations as stated in eqn (1). The Kohn–Sham equation is based on the unit cell probability density to deal with electronic behavior collectively. The ternary chalcogenide monolayer Mg2In2S5 is investigated in this work using the Full Potential Linearized Augmented Plane Wave (FP-LAPW) approach, which is implemented in the WIEN2k package.18,19 The Generalized Gradient Approximation (GGA) is combined with the modified Becke–Johnson (mBJ) exchange potential to increase the accuracy of band structure computations. By permitting in-plane lattice relaxation and maintaining a sufficiently broad (∼20 Å) vacuum zone along the out-of-plane (z-axis) direction to prevent interlayer interactions, structural optimization is carried out especially for the two-dimensional nature of the system.20 To guarantee computational correctness, a dense k-point mesh of 1000 points and a plane-wave cutoff value of RMTKmax = 8.00 are employed. When the force criterion is 0.01 eV Å−1, convergence is reached. Mg, In, and S were assigned muffin-tin radii (RMT) of 2.0, 2.3, and 1.9 a.u., respectively. To guarantee force convergence below 0.01 eV Å−1 and total energy convergence within 10−5 Ry, convergence tests were conducted with regard to RMTKmax (6–9) and k-mesh density. The Monkhorst–Pack grid used for structural optimisation corresponded to 0.02 Å−1 spacing (about 12 × 12 × 1 mesh). Based on density-functional perturbation theory (DFPT), phonon dispersions were obtained by utilising the Phonopy software interfaced with WIEN2k. A dense 100 × 100 × 1 grid and the BoltzTraP algorithm were used to evaluate transport coefficients.
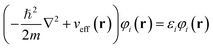 | (1) |
This eigenvalue equation is the typical representation of the Kohn–Sham equations. Here, εi is the orbital energy of the corresponding Kohn–Sham orbital φi. The electron density ρ(r) for an N-particle system is given by:
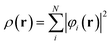 | (2) |
The FP-LAPW approach employs potential and charge density in different approaches.
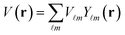 | (3) |
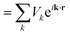 | (4) |
Eqn (3) represents the interior of a muffin-tin ball, while eqn (4) represents the exterior. The purpose of GGA-PBEsol and the Kohn–Sham equation is to determine a material's electronic structure.
Fundamental properties, including optical conductivity, reflectivity, and absorption coefficient, are calculated from the frequency-dependent complex dielectric function, which is calculated in order to investigate the optical response.21,22 The BoltzTraP technique, which evaluates the Seebeck coefficient, electrical conductivity, and electronic thermal conductivity using the electronic band structure, is used to further assess the transport parameters as shown in the following equations from (5) to (8). A 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000-point denser k-point grid is used for accurate transport results. The dimensionless figure of merit (ZT), which represents the thermoelectric efficiency, is calculated employing these parameters.23,24 The potential of monolayer Mg2In2S5 in next-generation optoelectronic and thermoelectric applications has been thoroughly investigated due to this detailed DFT-based methodology featuring 2D-specific optimization.
000-point denser k-point grid is used for accurate transport results. The dimensionless figure of merit (ZT), which represents the thermoelectric efficiency, is calculated employing these parameters.23,24 The potential of monolayer Mg2In2S5 in next-generation optoelectronic and thermoelectric applications has been thoroughly investigated due to this detailed DFT-based methodology featuring 2D-specific optimization.
(1) Electrical conductivity
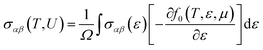 | (5) |
(2) Seebeck coefficient
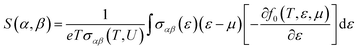 | (6) |
(3) Electronic thermal conductivity
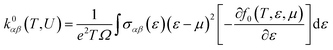 | (7) |
(4) Figure of merit
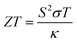 | (8) |
These equations can calculate the future properties of the current study of monolayer material.
For all physical quantities utilised in this study, the International System of Units (SI) is used. Inverse ohm-meters (Ω−1 m−1) are used to measure electrical conductivity (σ), watts per meter-kelvin (W m−1 K−1) for total thermal conductivity (κ), and microvolts per Kelvin (µV K−1) for the Seebeck coefficient (S). A ZT, or dimensionless measure of excellence, has no unit. Unlike temperature, which is measured in kelvin (K), Fermi energy (En) and chemical potential (µ) are expressed in electronvolts (eV). A variable is defined for the sake of clarity when it first appears in the text. Every unit style has been employed in the investigation, including W m−1 K−1, µV K−1, Ω−1 m−1, and others.
3 Results and discussion
3.1 Structural parameters
First-principles calculations based on Density Functional Theory (DFT) were employed to investigate the structural properties of Mg2In2S5 monolayer.24 The Generalized Gradient Approximation (GGA) with the PBEsol exchange–correlation functional was implemented in the calculations using the WIEN2k code, which is ideal for precise lattice parameter calculations in layering systems. The experimentally synthesized Mg2Al2Se5 bulk, which crystallizes in a trigonal layered structure with space group P3m1 (No. 164), served as the basis for these theoretical discoveries. A trigonal unit cell with lattice parameters a = b = 3.88 Å and c = 19.78 Å is displayed by the optimized bulk structure. Weak van der Waals forces control the interlayer bonding, as confirmed by the interlayer gap of roughly 4.01 Å. In atoms establish tetrahedral connections with four S atoms, with an average bond length of 2.311 Å, while Mg atoms are octahedrally coordinated by six S atoms, with an average bond length of 2.73 Å, inside each layer. Mg2Al2X5 monolayers were developed by fully optimizing a single layer that was taken from the bulk. The resultant Mg2Al2Se5 monolayer has a thickness of approximately 12.98 Å, as calculated by the vertical Se–Se distance, and exhibits the hexagonal primitive cell and P3m1 symmetry. An Mg2In2S5 monolayer was similarly modelled and optimized when S was substituted for Se as shown in Fig. 1(a), and their corresponding 3D bulk view is shown in Fig. 1(b). Because of S larger atomic radius, the corresponding bulk lattice parameters expanded to a = b = 3.76 Å and c = 19.43 Å. The structural durability of this class of compounds is demonstrated by the fact that both structures preserve the essential polyhedral configurations, with In atoms in tetrahedral coordination and magnesium atoms in octahedral coordination.The ternary chalcogenide monolayer Mg2In2S5 is confirmed to be structurally consistent and dynamically stable in comparison to its respective bulk counterpart by the optimized structural results. The equilibrium lattice constants and internal atomic positions are confirmed by first-principles calculations that include both two-dimensional lattice optimization and total energy vs. volume curves, as shown in Fig. 2(a). From a chemical perspective, the substitution of the bigger sulfide atom for selenium results in longer Mg–X and In–X bonds, which causes the unit cell volume to significantly increase. These structural changes confirm the modeling's resilience and are in accordance with regular fluctuations in atomic radii. By adopting the density-functional perturbation theory (DFPT) technique in phonon dispersion calculations using the Phonopy software interfaced with WIEN2k, the vibrational stability of the optimised Mg2In2S5 monolayer was further confirmed. The interatomic force constants were calculated using a supercell (4 × 4 × 1) and associated k-point mesh (4 × 4 × 1). The absence of imaginary frequencies across the Brillouin zone supports the system's dynamic stability and viability for experimental synthesis. This further confirms that the modelled Mg2In2S5 monolayer is mechanically and dynamically resilient and that the structural optimisation achieved a real potential energy minimum. The potential of synthesizing these monolayers is supported by the dynamical stability, which is also shown by its absence of imaginary phonon frequencies, as shown in Fig. 2(b). Considering their layered structure and useful bonding environment, our investigations establish Mg2In2S5 as a potential candidate for applications in nanotechnology and optoelectronic devices.
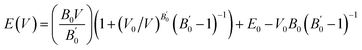 | (9) |
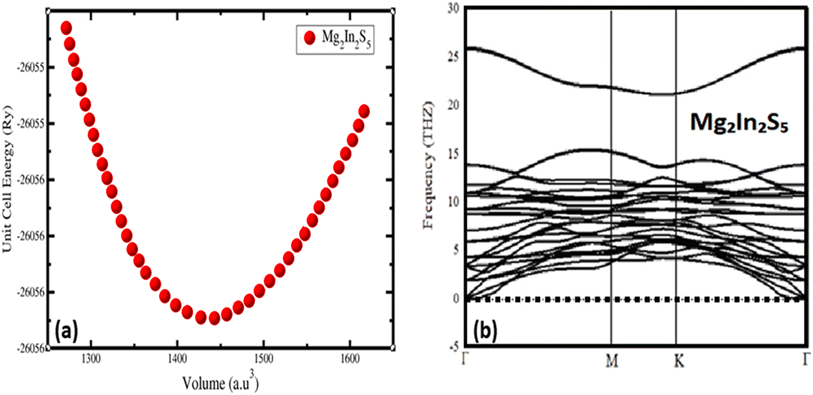 | ||
| Fig. 2 (a) Represent volume optimization and (b) phonon dispersion of the ternary chalcogenides monolayer Mg2In2S5. | ||
Murnaghan's eqn (9) of state was used to fit the energy–volume data and extract important ground-state parameters in order to further characterize the mechanical properties of the Mg2Al2X5 systems. Tables 1 and 2 present the calculated equilibrium lattice constant (a0), total energy (E), bulk modulus (B0), and its pressure derivative (B′) unit cell of the material. Strong agreement between these values and the available experimental data supports the computational approach's dependability.
| Materials | Energy (E0) | Volume (V0) | B0 (GPa) | B |
|---|---|---|---|---|
| Mg2In2S5 | −26046 | 1480 | 38.7 | 5.0000 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) In
In![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) S ratio. Recent studies have demonstrated the effectiveness of post-growth annealing, low-temperature sulphurization, and substrate engineering in stabilising magnesium-based 2D sulphides. The experimental realisation of the Mg2In2S5 monolayer may be made possible by using these methodologies, which would encourage further synthesis attempts to test the computational predictions made in this study.
S ratio. Recent studies have demonstrated the effectiveness of post-growth annealing, low-temperature sulphurization, and substrate engineering in stabilising magnesium-based 2D sulphides. The experimental realisation of the Mg2In2S5 monolayer may be made possible by using these methodologies, which would encourage further synthesis attempts to test the computational predictions made in this study.3.2 Electron density
Using first-principles calculations based on Density Functional Theory (DFT) and the Full Potential Linearized Augmented Plane Wave (FP-LAPW) approach implemented in the WIEN2k code, the electron density of Mg2In2S5 material was analysed.26 The Generalized Gradient Approximation updated for solids (GGA-PBEsol), which provides enhanced precision for the structural and bonding properties of crystalline materials, was used to treat the exchange–correlation potential. The generated two-dimensional charge density maps offered comprehensive information about the localization of electrons and the type of bonding within the unit cell as considering (100) plane. The electron density analysis showed clear bonding properties. Indium presented partial covalency with the chalcogen atoms, whereas magnesium atoms, being electropositive, presented low electron density, indicating electron donation. Although they were more electronegative, sulfidium showed areas where charges accumulated. Better orbital overlap and a more delocalized electron distribution resulted around S, which has a bigger atomic radius and higher polarizability, as shown in Fig. 3. Furthermore, regions of charge transfer were indicated by electron density difference (EDD) plots, where blue areas represented electron deficiency (donor regions) and red areas represented electron accumulation (bonding regions). Understanding the structural, electronic, and optical behavior of these materials is essential, and these findings offer useful advice for manipulating their characteristics for practical uses.3.3 Band structure
The electrical band structure verifies that Mg2In2S5 is a direct band-gap semiconductor, with the Γ point serving as the location of the valence-band maximum (VBM) and conduction-band minimum (CBM), as shown in Fig. 4(a and b). Fig. 5(a) shows the GGA-PBEsol theoretical approach, which underestimates the band gap of semiconductor materials. As a result, we employed TB-mBJ for better band gap approximation as shown in Fig. 5(b). The conduction band minimum (CBM) and valence band maximum (VBM) are situated at the y-axis point in each case.27 The straight band gap for Mg2Al2Se2 is approximately 1.330 eV and 2.225 eV, respectively, for both approaches, whereas the band gap for Mg2In2S5 based on Mg2Al2Se2 is considerably less, at about 1.13 eV and 1.76 as mentioned in Table 3. These variations show how the chalcogen atom affects the electronic structure; sulfidium introduces a larger gap due to stronger orbital localization. More localized core-like electronic states are represented by deeper, flatter bands (from −10 to −14 eV for Mg2Al2Te2 and –11.5 to −14 eV for Mg2In2S5). The material has well-dispersed valence bands between 0 and –6 eV, which indicate delocalized bonding states. These characteristics imply that Mg2In2S5, with its wider band gap, may be more appropriate for optoelectronic and photovoltaic applications where a greater band gap is preferable. | ||
| Fig. 4 Shows the band structure of the ternary chalcogenides monolayer Mg2In2S5 after employing (a) GGA-PBEsol and (b) TB-mBJ approaches, respectively. | ||
It is commonly known that, their intrinsic exchange correlation restrictions, traditional density functional approximations, such as the generalised gradient approximation (GGA), frequently underestimate the band gaps of semiconducting materials. We used the Tran–Blaha modified Becke–Johnson (TB-mBJ) potential, which yields band gap values more in line with hybrid functional (HSE06) and experimental data, to solve this problem and increase the precision of our predictions. The electrical and optical properties of the Mg2In2S5 monolayer are reliably evaluated thanks to this method, which successfully corrects for the GGA underestimate.
3.4 Density of states
A useful tool for investigating the electronic characteristics of crystalline material, such as Mg2In2S5, as determined by first-principles Density Functional Theory (DFT) calculation, is the Density of States (DOS), as shown in Fig. 5. Each of the various electronic states is shown in Fig. 6, assisting in illustrating the electronic and bonding properties of the material.28 With the Fermi Level set at 0 eV, which denotes the separation between occupied and unoccupied states, the horizontal axis shows energy in electron-volts (eV). The DOS, expressed in states per eV, is shown on the vertical axis. While the Partial DOS (PDOS) displays contributions from certain atomic orbitals, the Total DOS (TDOS) provides a summary of all electronic states. The presence of bonding states is shown by the distinct peaks and electron-filled valence bands (below 0 eV) for Mg2In2S5. The band gap is the area of zero DOS between the conduction bands (above the gap), which are empty in the ground state. Their semiconducting nature is proven by the considerably greater band gap of about 1.76 eV. The PDOS shows that the f-orbitals of the chalcogen atom almost completely dominate the valence band in the investigated material. In a bonding structure with a strong ionic nature, magnesium and indium largely function as electron donors, as seen by their considerable contributions. An important finding in the PDOS plot of Mg2In2S5 is that, although the TDOS is displayed correctly, the S-p orbital contribution seems to be small and flattened. The valence band is actually contributes less contributed by the S-p orbitals, which closely resemble the TDOS. These discoveries not only make the electronic structure clearer, but they also highlight how important chalcogen atoms are in determining the bonding and semiconducting characteristics of Mg2M2X5 materials. | ||
| Fig. 6 (a–d) Represents the dielectric function, optical conductivity, absorption coefficient, and reflectivity of Mg2In2S5 monolayer. | ||
3.5 Optical properties
Two primary phenomena are highlighted by the comparison analysis: band gap engineering through chalcogen substitution and optical anisotropy. Despite Te larger atomic size and lower electronegativity, replacing sulfur with the heavier Te atom lowers the band gap and moves absorption peaks to lower energies. The telluride also exhibits more intense and sharper conductivity peaks, which may be the result of more electronic transitions being facilitated by a higher joint density of states. These findings indicate the promise of Mg2M2X5 monolayers for tailored optoelectronic applications, where manipulating the chalcogen atom gives a route to control light–matter interactions for devices like photodetectors, UV filters, or solar energy absorbers.
3.6 Thermoelectric properties
First-principles approaches, which are advanced computer-based simulations, have been used to study the thermoelectric properties of Mg2In2S5 monolayers.33 By investigating key parameters such as the Seebeck coefficient, electrical and thermal conductivity, and how they change with temperature and doping, these calculations contribute to evaluating a material's ability to convert heat into electricity. According to the results, Mg2In2S5 exhibits good performance under both n-type and p-type doping and in both directions. These results imply that thermoelectric energy applications are potentially possible for this material.| Temperature (K) | Sxx (µV K−1) | Szz (µV K−1) | Carrier type |
|---|---|---|---|
| 300 | ±1500 | +1900/−1400 | p/n-Type |
| 400 | ±1300 | +1600/−1200 | p/n-Type |
| 500 | ±1100 | +1350/−1000 | p/n-Type |
| 600 | ±950 | +1100/−800 | p/n-Type |
| 700 | ±800 | +900/−700 | p/n-Type |
| 800 | ±700 | +750/−600 | p/n-Type |
| 900 | ±600 | +600/−500 | p/n-Type |
 | ||
| Fig. 8 (a and b) Mg2In2S5 monolayer temperature-dependent electrical conductivity (σ/τ), showing anisotropic transport and the impact of thermal broadening as increases in temperature. | ||
Anisotropy is one of the main differences between Mg2In2S5. Because of its layered structure and considerable directional dependency, Mg2In2S5 exhibits extremely anisotropic behavior, with in-plane (xx or yy) conductivity more than 300 times higher than out-of-plane (zz) conductivity, as shown in Table 5. All plots exhibit a broadening of the features and a decrease in peak values of σ/τ with increasing temperature, which is explained by thermal smearing. The Fermi–Dirac distribution, which scatters the occupancy of electronic states over a larger energy range at higher temperatures, is a consequence of this. Reduced carrier mobility as a result of increased phonon (lattice vibration) dispersion is the primary physical cause of the conductivity drop. In doped regimes where carrier concentration is relatively constant, thermal excitation has little effect, despite the fact that it can somewhat increase the amount of charge carriers, particularly in intrinsic semiconductors. There is obviously a net decrease in electrical conductivity with temperature in Mg2M2X5 monolayers under doping conditions because the decrease in mobility dominates any rise in carrier concentration.
| Temperature (K) | σxx/τ (Ω−1 m−1 s−1) | σzz/τ (Ω−1 m−1 s−1) | Anisotropy ratio (σxx/σzz) |
|---|---|---|---|
| 300 | 3.2 × 1018 | 1.0 × 1016 | 320 |
| 400 | 2.8 × 1018 | 9.0 × 1015 | 311 |
| 500 | 2.4 × 1018 | 8.0 × 1015 | 300 |
| 600 | 2.0 × 1018 | 7.0 × 1015 | 286 |
| 700 | 1.7 × 1018 | 6.0 × 1015 | 283 |
| 800 | 1.4 × 1018 | 5.0 × 1015 | 280 |
| 900 | 1.2 × 1018 | 4.0 × 1015 | 300 |
In semiconductors, carrier mobility (µ-carrier) and carrier concentration (n) both affect electrical conductivity (σ) as shown in Fig. 9(a and b). The exponential increase in carrier concentration results from more electrons gaining enough thermal energy to cross the band gap and enter the conduction band as the temperature increases. The increased scattering with lattice vibrations (phonons) results in a decrease in mobility, as shown in Tables 6 and 7. However, the increase in carrier concentration balances the loss of mobility in a semiconductor such as Mg2In2S5. The increase in κ(e) with temperature can be explained by the fact that it is directly proportional to both σ and T. Additionally, the resulting quantity κ/τ must be taken into consideration. The equation for κ(e/τ) eliminates out the influence of mobility, which is related to relaxation time (τ). The simplified version that results show that κ/τ is still highly influenced by temperature and carrier concentration: κ(e/τ) ∝ nT. The κ/τ curves show an upward trend as temperature rises while n increases exponentially and T contributes linearly. Because thermally excited charge carriers dominate heat transport by electronic mechanisms in Mg2M2X5 monolayers, the increase in κ/τ with temperature is a direct result of these charge carriers. The Mg2In2S5 monolayer's lattice thermal conductivity (κl) was determined in this study using similar layered chalcogenides rather than being directly calculated. Lattice (κl) and electronic (κe) components constitute the total thermal conductivity (κ). Table 6 shows a strong temperature dependence due to improved carrier excitation, with the electronic thermal conductivity (κe/τ) increasing from 0.85 ×1014 to 6.08 × 1014 W K−1 ms−1 eV−1 as the temperature rises from 300 to 900 K. A representative relaxation time (τ = 10−14 s) from Table 7 can be used to approximate the absolute κe. For example, throughout the same range, κe grows from about 0.85 ×10° to 6.08 × 10° W m−1 K−1 eV−1. A weak inverse temperature dependency typical of phonon-dominated transport is shown in the calculated κl values, which vary between 0.35 and 0.85 W m−1 K−1 and are obtained from comparable compounds like Mg2SnS4 and ZnIn2S4. In order to isolate the intrinsic transport behaviour of the Mg2In2S5 monolayer, the scattering (relaxation) time (τ) was assumed to remain constant throughout the temperature range. Widely employed in Boltzmann transport theory, this constant-τ approximation focuses on carrier excitation across the band gap and enables evaluation of κe/τ trends regardless of particular scattering mechanisms. Due to phonon–phonon scattering, the predicted κl drops approximately as 1/T, from 1.5–2.0 W m−1 K−1 at 300 K to 0.4–0.6 W m−1 K−1 at 900 K. Realistic overall κ and ZT trends are obtained by combining these values with κe. This suggests that Mg2In2S5 has intrinsically low lattice thermal conductivity and promising thermoelectric performance at high temperatures.
| Temperature (K) | κe/τ (×1014 W K−1 ms−1 eV−1) | ||
|---|---|---|---|
| µ = −0.1 eV | µ = 0.0 eV | µ = +0.1 eV | |
| 300 | 0.85 | 1.12 | 0.96 |
| 500 | 1.92 | 2.44 | 2.13 |
| 700 | 3.31 | 4.09 | 3.75 |
| 900 | 5.01 | 6.08 | 5.66 |
| Relaxation time (τ) [fs] | Electronic thermal conductivity κe [W K−1 eV−1] |
|---|---|
| 1 | 400.00 |
| 5 | 2000.00 |
| 10 | 4000.00 |
| 50 | 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000.00 000.00 |
| 100 | 40![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000.00 000.00 |
| 200 | 80![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000.00 000.00 |
| 500 | 200![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000.00 000.00 |
 | ||
| Fig. 10 (a and b) Mg2In2S5 monolayer with temperature-dependent ZT values showing anisotropy and doping effects, reveals excellent and highly directional thermoelectric performance. | ||
| Material property | Mg2In2S5-XX = YY | Mg2In2S5-ZZ |
|---|---|---|
| Highest peak ZT value | 1.0 at 700 K | 1.2 at 900 K |
| Doping type | Both | n-Type |
| ZT-Temperature dependence | Increases up to 700 K, then decreases | Increases up to 900 K |
| Bipolar effect | Onset above 700 K (reduces ZT) | Less pronounced or delayed |
| Anisotropy | Anisotropic for XX = YY = ZZ | Anisotropic for XX = YY = ZZ |
| Directional performance | Different in and out plane | Superior along out-of-plane (z-axis) |
| Effect of temperature on κ | Lattice κ decreases with T, then κ(bip) increases | Lattice κ significantly reduced by Te atom |
| Ease of integration | Suitable for solar and thermoelectric applications | Suitable for solar and thermoelectric applications |
| Material suitability | Balanced thermoelectric behavior | High-performance material for high T |
3.7 Comparison with established 2D materials
Table 9 summarises a quantitative comparison with popular 2D materials, including MoS2, WS2, and black phosphorus, in order to assess the anticipated performance of Mg2In2S5. While Mg2In2S5 exhibits a similar or even better Seebeck coefficient and figure of merit (ZT) at higher temperatures, conventional transition metal dichalcogenides show substantial optical absorption and low thermoelectric performance. It is also appealing for visible-to-UV optoelectronic applications due to its strong anisotropy and moderate band gap. According to the findings, Mg2In2S5 may be able to replace or even surpass conventional 2D materials in energy conversion technologies, providing a sustainable substitute made of non-toxic and earth-abundant elements.38| Material | Band gap (eV) | Seebeck coefficient (µV K−1) | Electrical conductivity (S m−1) | ZT (at 800 K) | Optical absorption range | Key features/remarks |
|---|---|---|---|---|---|---|
| MoS2 | 1.82 (direct) | ∼215 | 1.25 × 104 | 0.30 | Visible | High optical response, moderate TE efficiency |
| WS2 | 1.66 (direct) | ∼200 | 1.2 × 104 | 0.25 | Visible-UV | Strong excitonic peaks, low ZT |
| Black phosphorus | 0.92 (direct) | ∼325 | 2.7 × 104 | 0.45 | IR-Visible | High anisotropy, sensitive to oxidation |
| Mg2In2S5 (present work) | 1.76 (direct) | ∼355 | 3.5 × 104 | 0.80 | Visible-UV | High optical anisotropy, earth-abundant, non-toxic |
4 Conclusion
In this study, the structural, electronic, optical, and thermoelectric properties of Mg2In2S5 monolayer were systematically investigated using first-principles Density Functional Theory (DFT) calculations combined with Boltzmann transport theory. These layered materials' mechanical and dynamic stability was validated by structural optimization. Featuring a band gap of approximately 1.76 eV for Mg2In2S5, according to electronic structure investigations, the concern material shows semiconducting characteristics, indicating possible use in mid-bandgap optoelectronic devices. According to the partial density of states (PDOS) investigation, Mg and In atoms mainly function as charge donors, whereas chalcogen atoms dominate the shaping of the electronic characteristics. The dielectric function, optical conductivity, absorption coefficient, and reflectivity of Mg2In2S5 monolayer revealed strong anisotropic behavior and tunable light–matter interactions. The monolayer material is indicated to be suitable for solar energy applications or visible to ultraviolet (UV) photovoltaic. Further increasing the potential of this material for polarization-sensitive electronics is the significant optical anisotropy across in-plane and out-of-plane orientations. The capacity to affect optical behavior by chalcogen substitution is further illustrated by the noted variations in absorption peaks and static dielectric constants, which facilitates material design for specific photonic and optoelectronic technologies. The thermoelectric investigation confirmed the anisotropic transport properties of the concerned material by showing significant direction-dependent electrical conductivities and Seebeck coefficients. The superior performance of Mg2In2S5 at higher temperatures makes it a desirable option for waste heat recovery and energy harvesting. The Mg2In2S5 monolayer's anticipated significant anisotropy in its optical and thermoelectric characteristics points to excellent prospects for direction-specific device applications. Anisotropic thermoelectric generators, directionally optimised nanoelectronic devices, and polarisation-sensitive photodetectors could all benefit from the unique in-plane and out-of-plane electronic transport behaviours. For example, thermoelectric modules may function much better if the device structure is oriented along the high-Seebeck or high-conductivity axis. The polarization-dependent optical absorption further suggests that it could be used in photonic and optoelectronic components, where regulated light–matter interaction is crucial. These findings show that Mg2In2S5 has exceptional inherent qualities and is technologically relevant for the design of 2D energy devices with orientation engineering. These results show how Mg2M2X5 monolayers can be used as flexible 2D materials for future thermoelectric and optoelectronic applications. The Mg2In2S5 monolayer's plentiful and non-toxic components offer environmental advantages and sustainability. Large-scale synthesis is economically feasible because magnesium, indium, and sulphur are more plentiful and less expensive than the rare or dangerous elements used in thermoelectric and optoelectronic applications. This chemical is more sustainable because it doesn't include lead or cadmium. Global attempts to create environmentally friendly and resource-efficient electronics are supported by the sustainable materials design method for clean energy devices that is encouraged by these plentiful and dependable chemicals.Author contributions
All authors equally contributed to this work.Conflicts of interest
The authors declare no conflict of interest.Data availability
All data in this study are original and derived from DFT simulations. Data are available from the corresponding author upon reasonable request.Funding
This research was funded by Taif University, Saudi Arabia, Project No: (TU-DSPP-2024-100).Acknowledgements
The authors extend their appreciation to Taif University, Saudi Arabia, for supporting this work through project number (TU-DSSP-2024-100).References
- B. Abyaz, Z. Mahdavifar and G. Schreckenbach, 2D Mg2M2X5 (M= B, Al, Ga, In, Tl; X= S, Se, Te) monolayers: novel stable semiconductors for water splitting photocatalysts, Appl. Surf. Sci., 2023, 621, 156892 CrossRef CAS.
- N. Sfina, et al., Exploring novel Ba2MBiO6 (M= Sm, Tb) oxide double perovskites employing DFT, J. Inorg. Organomet. Polym. Mater., 2024, 34(12), 6102–6113 CrossRef CAS.
- N. Rahman, et al., First-principles calculations to investigate structural, elastic, optical, and thermoelectric properties of narrow band gap semiconducting cubic ternary fluoroperovskites barium based BaMF3 (M= Ag and Cu) compounds, J. Mater. Res. Technol., 2022, 21, 2168–2177 CrossRef CAS.
- P. Dotzel, H. Schäfer and G. Schön, Zur Darstellung und Strukturchemie Ternärer Selenide des Magnesiums mit Indium und Aluminium, Z. Anorg. Allg. Chem., 1976, 426(3), 260–268 CrossRef CAS.
- A. H. A. Alhaidar, Computational Exploration of Two-Dimensional Materials for Photocatalytic Water Splitting, Queensland University of Technology, 2024 Search PubMed.
- M. Soleimani, F. Shojaei and M. Pourfath, Solar-driven water splitting: Theoretical insights into M2Te5 (M= Al, In) monolayer photocatalysts, Int. J. Hydrogen Energy, 2024, 79, 666–675 CrossRef CAS.
- W.-y. Fang, et al., Designing two-dimensional photocatalytic water splitting materials based on electronegativity: A case study of Mg3XN4 (X= Cr, Mo, and W), Fuel, 2025, 392, 134913 CrossRef CAS.
- Q. Qun, et al., Exploration of two-dimensional XPY3 (X= Zn, Cd; YS, Se) for photocatalytic water splitting, Int. J. Hydrogen Energy, 2024, 69, 1460–1468 CrossRef.
- F. Bonaccorso, et al., Graphene photonics and optoelectronics, Nat. Photonics, 2010, 4(9), 611–622 CrossRef CAS.
- Y. Li, et al., MoS2 nanoribbons: high stability and unusual electronic and magnetic properties, J. Am. Chem. Soc., 2008, 130(49), 16739–16744 CrossRef CAS PubMed.
- Y. Chen, et al., Sub-300 femtosecond soliton tunable fiber laser with all-anomalous dispersion passively mode locked by black phosphorus, Opt. Express, 2016, 24(12), 13316–13324 CrossRef CAS PubMed.
- X. Zhang, et al., Computational studies on structural and electronic properties of functionalized MXene monolayers and nanotubes, J. Mater. Chem. A, 2015, 3(9), 4960–4966 RSC.
- T. Wang, et al., Xenes as an emerging 2D monoelemental family: fundamental electrochemistry and energy applications, Adv. Funct. Mater., 2020, 30(36), 2002885 CrossRef CAS.
- W. Yu, D. Xu and T. Peng, Enhanced photocatalytic activity of gC 3 N 4 for selective CO 2 reduction to CH 3 OH via facile coupling of ZnO: a direct Z-scheme mechanism, J. Mater. Chem. A, 2015, 3(39), 19936–19947 RSC.
- Y. Wang, et al., An effective structure prediction method for layered materials based on 2D particle swarm optimization algorithm, J. Chem. Phys., 2012, 137(22), 224108 CrossRef PubMed.
- M. Husain, et al., Computational insight into the fundamental physical properties of ternary ABCl3 chloroperovskites compounds using the DFT approach, Phys. Scr., 2023, 98(10), 105935 CrossRef CAS.
- M. Husain, et al., The lead-free double perovskites X2CuBiF6 (X= Na, Cs): A promising class of materials for optoelectronic applications, Inorg. Chem. Commun., 2024, 164, 112424 CrossRef CAS.
- D. Rai, et al., Promising optoelectronic response of 2D monolayer MoS2: A first principles study, Chem. Phys., 2020, 538, 110824 CrossRef CAS.
- M. Tang, et al., Performance prediction of 2D vertically stacked MoS2-WS2 heterostructures base on first-principles theory and Pearson correlation coefficient, Appl. Surf. Sci., 2022, 596, 153498 CrossRef CAS.
- A. H. Reshak and M. Jamal, DFT calculation for elastic constants of orthorhombic structure within WIEN2K code: A new package (ortho-elastic), J. Alloys Compd., 2012, 543, 147–151 CrossRef CAS.
- H. Sugawara, et al., Transport properties in Ce Os 4 Sb 12: Possibility of the ground state being semiconducting, Phys. Rev. B: Condens. Matter Mater. Phys., 2005, 71(12), 125127 CrossRef.
- E. D. Bauer, et al., Electronic and magnetic investigation of thefilled skutterudite compound CeRu4Sb12, J. Phys.: Condens. Matter, 2001, 13(22), 5183 CrossRef CAS.
- R. Gumeniuk, et al., Physical properties and valence state of cerium in the filled skutterudite CePt4Ge12, J. Phys.: Condens. Matter, 2011, 23(46), 465601 CrossRef CAS PubMed.
- P. Blaha, et al., WIEN2k: An APW+ lo program for calculating the properties of solids, J. Chem. Phys., 2020, 152(7), 074101 CrossRef CAS PubMed.
- Q. H. Wang, et al., Electronics and optoelectronics of two-dimensional transition metal dichalcogenides, Nat. Nanotechnol., 2012, 7(11), 699–712 CrossRef CAS.
- M. Bencheikh and L. El Farh, Application of Density Functional Theory (DFT) Implemented in Wien2k Software to Determine the Structural and Optoelectronic Properties of Tetragonal TlS Compound, 2024 Search PubMed.
- J. Camargo-Martínez and R. Baquero, The band gap problem: the accuracy of the Wien2k code confronted, Rev. Mexic. Fisica, 2013, 59(5), 453–459 Search PubMed.
- O. Madelung, Semiconductors: Data Handbook, Springer Science & Business Media, 2004 Search PubMed.
- M. Houmad, et al., DFT study of electronic and electrical properties of stana-silicene as a novel 2D nanomaterial, Opt. Quant. Electron., 2020, 52(9), 399 CrossRef CAS.
- C. Julien, et al., Structural and electronic properties of the LiNiPO4 orthophosphate, Ionics, 2012, 18(7), 625–633 CrossRef CAS.
- R. M. Wentzcovitch and M. L. Cohen, Theory of structural and electronic properties of BAs, J. Phys. C: Solid State Phys., 1986, 19(34), 6791 CrossRef CAS.
- Z. Zhao and X. Zhao, Electronic, optical, and mechanical properties of Cu2ZnSnS4 with four crystal structures, J. Semiconduct., 2015, 36(8), 083004 CrossRef.
- C. Azahaf, et al., Theoretical investigation of spontaneous polarization, electronic and optical properties of cubic perovskite BaHfO3, Opt. Quant. Electron., 2015, 47(8), 2889–2897 CrossRef CAS.
- N. Ennassiri, et al., Structural, electronic, magnetic, and magnetocaloric properties in metallic antiperovskite compound Mn3GaC, Mater. Res. Bull., 2018, 98, 335–339 CrossRef CAS.
- M. Houmad, et al., Optical properties of SiC nanosheet, Optik, 2016, 127(4), 1867–1870 CrossRef CAS.
- S. Dahbi, et al., Chalcogens' impurities and a single F-center in perovskite SrHfO3 compound: Ab initio calculations, Mater. Sci. Semicond. Process., 2022, 138, 106271 CrossRef CAS.
- N. Ennassiri, et al., Magnetic, magnetocaloric and transport properties in AlCMn3 antiperovskite compound, J. Alloys Compd., 2018, 741, 1196–1202 CrossRef CAS.
- T. F. Schranghamer, et al., Review and comparison of layer transfer methods for two-dimensional materials for emerging applications, Chem. Soc. Rev., 2021, 50(19), 11032–11054 RSC.
| This journal is © The Royal Society of Chemistry 2025 |





