Open Access Article
Open Access ArticleA hollow mesoporous magnetic silica-based nanocomposite supported on copper ferrite functionalized with phosphomolybdic acid as a nanocatalyst for the synthesis of 1,4-dihydropyridines
Mahla Dadaei and
Hossein Naeimi *
*
Department of Organic Chemistry, Faculty of Chemistry, University of Kashan, Kashan, 87317, Islamic Republic of Iran. E-mail: naeimi@kashanu.ac.ir; Tel: +98 3155912397 Tel: +98 3155912388
First published on 9th December 2025
Abstract
Porous nanomaterials are substances characterized by structures containing holes. Due to their extensive porous surface areas, these materials exhibit high reactivity and are of use as catalysts. In this study, a hollow nanocatalyst of magnetic mesoporous copper ferrite functionalized with phosphomolybdic acid was prepared and characterized by XRD, SEM, EDX, FT-IR, and VSM techniques that are each separately discussed in the text. This catalyst was used in a reaction to synthesize 1,4-dihydropyridine derivatives through a three-component reaction of barbituric acid, ammonium acetate and various benzaldehyde derivatives. After purification of the products with ethanol, the products were identified using physical and spectroscopic data, such as melting point, FT-IR, and 1H NMR. Also, this catalyst can be recovered and reused five times without losing its catalytic activity.
Introduction
Recently, magnetic nanoparticles have attracted significant attention from researchers due to their desirable features that enable their application in various scientific fields such as nanocatalysts, biopharmaceuticals,1 magnetic resonance imaging (MRI),2 magnetic particle imaging,3 and data storage.4 To date, various methods have been proposed for the synthesis of magnetic nanoparticles.5When synthesizing magnetic nanoparticles, several important factors must be considered, such as the intrinsic magnetic properties, size and structure of the nanoparticles, surface coating and surface charge, stability in aqueous environments, as well as their non-toxicity. Using an ideal synthetic method, it is possible to appropriately control the size, shape, surface coating, and colloidal stability of magnetic nanoparticles. Among nanoscale magnetic materials, in addition to iron oxide, other iron-containing compounds also exhibit the magnetic properties required for biomedical applications. Ferrites are particularly valuable due to their ability to have their physicochemical properties controlled by an external magnetic field, making them highly suitable for other applications such as.6,7 Nanoparticles designed for such purposes must exhibit low toxicity, high structural stability, and significant superparamagnetic behaviour at room temperature (RT). Among magnetic nanoparticles, spinel ferrites (SFs) are represented by the chemical formula MFe2O4, where M refers to a divalent cation such as Co, Cu, Fe, Mn, or Ni. Among SFs, particularly cobalt ferrite (CoFe2O4) and copper ferrite (CuFe2O4) have attracted significant attention due to their high coercivity, notable mechanical and chemical stability, and pronounced magnetocrystalline anisotropy, making them important from physical, chemical, and biomedical perspectives.8–10
Depending on the synthesis technique and calcination temperature, CuFe2O4 can crystallize in two crystal structures, namely cubic spinel and tetragonal. Common nanoparticle synthesis methods include hydrothermal,11 sonochemical,12 sol–gel,13 citrate–nitrate,14,15 co-precipitation16,17 and solid-state18 techniques. Recently, CuFe2O4 materials with various morphologies have been studied and reported. To date, CuFe2O4 spinel has been extensively utilized in diverse fields, including heterogeneous catalysis,6,7 photocatalysis,6 and energy storage11 and as an anode material for batteries.13
Porous materials consist of a network of regular and irregular pores or cavities, which possess a large surface area and controllable pore size.19,20 Mesoporous materials are widely employed as drug carriers due to their exceptional properties, including a high surface area-to-volume ratio, tunable particle size,21 and excellent biocompatibility and biodegradability.22
In recent years, research on the synthesis of mesoporous silica nanoparticles for biomedical applications has dramatically increased, leading to significant advancements in this field. In 2001, MCM-41 was first introduced as a drug delivery system; subsequently, other mesoporous silica frameworks such as SBA-15 and MCM-48 have been identified and extensively studied as carriers for drug delivery and controlled release systems.23,24 Mesoporous silica nanoparticles are now commonly utilized in medicine for the targeted delivery of therapeutic agents and bioactive compounds within the body.
Solid catalysts based on ordered mesoporous materials have garnered considerable attention due to their uniform and well-defined distribution of active catalytic sites on the substrate surface.25 These materials exhibit a relatively homogeneous and controllable density of catalytically active centres, which contributes to the consistent reproducibility of synthesized catalysts.26,27 Moreover, the presence of ordered mesopores significantly alleviates diffusion limitations encountered in microporous catalysts such as zeolites, thereby enhancing mass transport properties.28 Compared to catalysts supported on conventional nanoparticles, mesoporous solid-supported catalysts demonstrate superior catalytic activity, stability, and ease of diffusion, making them highly advantageous for various catalytic applications.
Pyridopyrimidine is an aromatic compound with the formula C7H5N3. It is obtained from the coupling of two compounds, pyridine and pyrimidine. The coupling of the pyridopyrimidine ring can be carried out from different parts, each of which exhibits different properties and characteristics. The pyrido[3,2-d]pyrimidine compound is one of these heterocycles.29 Pyridopyrimidine derivatives have a wide range of biological properties such as anti-hypertensive, anti-cancer, anti-viral, and anti-bacterial.30
In recent years, the synthesis of pyridopyrimidine derivatives has been reported in the presence of different catalysts such as SBA-15-SO3H,31 DIPEAc,32 MWCNTs@L-His/Cu(II),33 MNPs-NPBG-SA,34 and ionic liquids.35 Although those works report convenient methods, the present method compared with previously reported catalysts has some advantages such as thermal and chemical stability, highly activity, simplicity of procedure, easy reaction work-up, recyclability, clean method and resulting products having high purity.
Today, magnetic nanoparticles are used as catalysts in multicomponent reactions,36–40 including the Hantzsch reaction, under various conditions.41–43
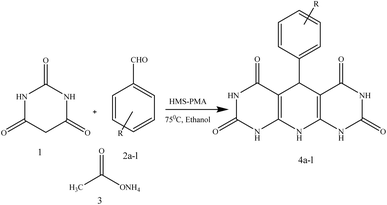
In continuation of our research interest in catalytic reactions,44–47 in this research, the synthesis of 1,4-dihydropyridine derivatives was explored using barbituric acid, ammonium acetate, and various benzaldehyde derivatives. The reactions were carried out by using a hollow mesoporous magnetic copper ferrite silica functionalized with phosphomolybdic acid as a nanocatalyst in ethanol at 75 °C.
Experimental
General information
The chemicals used in this study were purchased from Fluka and Merck chemical companies and used without further purification. The nanostructures were characterized using a Holland Philips X'Pert powder X-ray diffractometer (XRD) with Cu Kα radiation (λ = 0.154056 nm), operated at a scanning rate of 2° min−1 over a 2θ range of 10° to 100°. Fourier-transform infrared (FT-IR) spectra were recorded using KBr pellets with a PerkinElmer 781 spectrophotometer and a Nicolet Impact 400 FT-IR spectrometer. 1H NMR spectra were recorded in DMSO-d6 with a Bruker DRX-400 spectrometer using tetramethylsilane as an internal standard. The morphology of the nanocomposites was examined using scanning electron microscopy (SEM; SIGMA VP). Elemental analysis of the catalyst was carried out by energy-dispersive X-ray spectroscopy (EDX) using an Oxford Instruments system. Magnetic properties of the nanoparticles were measured using vibrating sample magnetometry (VSM; PPMS-9T) at 300 K at Kashan University, Iran. Thermogravimetric analysis (TGA) was performed using a thermal analyser (TGA 1100, Sanaf Electronic Industries). Brunauer–Emmett–Teller (BET) analysis was performed using a Belsorp Mini X. Melting points were determined using a Yanagimoto micro melting point apparatus and are reported uncorrected. The purity of the substrates and the progress of the reactions were monitored by thin-layer chromatography (TLC) on silica gel plates (Polygram SILG/UV 254, Merck).Catalyst preparation
5-Phenyl-5,10-dihydropyrido[2,3-d:6,5-d′]dipyrimidine-2,4,6,8-(1H,3H,7H,9H)-tetraone (4a). White solid, m.p. (°C): 303–305, m.p. (°C) rep.: 301–303;48 IR (KBr) ν (cm−1): 3400 (NH), 2971 (C–H, sp2 stretch), 1698, 1615 (C
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), 1402, 1513 (C
O), 1402, 1513 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C, Ar); 1H NMR (DMSO-d6, 400 MHz) δ (ppm): 10.14 (s, 4H, NH), 7.22 (s, 1H, NH), 7.12 (t, 3H, Ar, J = 10.1 Hz), 7.02 (d, 2H, Ar, J = 7.7 Hz), 5.92 (s, 1H, CH).
C, Ar); 1H NMR (DMSO-d6, 400 MHz) δ (ppm): 10.14 (s, 4H, NH), 7.22 (s, 1H, NH), 7.12 (t, 3H, Ar, J = 10.1 Hz), 7.02 (d, 2H, Ar, J = 7.7 Hz), 5.92 (s, 1H, CH).
5-(4-Chlorophenyl)-5,10-dihydropyrido[2,3-d:6,5-d′]dipyrimidine-2,4,6,8-(1H,3H,7H,9H)-tetraone (4b). White solid, m.p. (°C): 295–297, m.p. (°C) rep.: 296–298;48 IR (KBr) ν (cm−1): 3210 (NH), 2924 (C–H, sp2 stretch), 1694 (C
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), 1478, 1609 (C
O), 1478, 1609 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C, Ar); 1H NMR (DMSO-d6, 400 MHz) δ (ppm): 10.10 (s, 4H, NH), 7.20 (d, 2H, Ar, J = 8.2 Hz), 7.10 (s, 1H, NH), 7.02 (d, 2H, Ar, J = 8.1).
C, Ar); 1H NMR (DMSO-d6, 400 MHz) δ (ppm): 10.10 (s, 4H, NH), 7.20 (d, 2H, Ar, J = 8.2 Hz), 7.10 (s, 1H, NH), 7.02 (d, 2H, Ar, J = 8.1).
5-(4-Nitrophenyl)-5,10-dihydropyrido[2,3-d:6,5-d′]dipyrimidine-2,4,6,8-(1H,3H,7H,9H)-tetraone (4c). White solid, m.p. (°C): 302–304, m.p. (°C) rep.: 298–300;48 IR (KBr) ν (cm−1): 3398 (NH), 2971 (C–H, sp2 stretch), 1616 (C
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), 1401 (C
O), 1401 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C, Ar); 1H NMR (DMSO-d6, 400 MHz) δ (ppm): 10.18 (s, 4H, NH), 8.05 (d, 2H, Ar, J = 8.5 Hz), 7.22 (d, 2H, Ar, J = 8.4 Hz), 7.08 (s, 1H, NH), 6.04 (s, 1H, CH).
C, Ar); 1H NMR (DMSO-d6, 400 MHz) δ (ppm): 10.18 (s, 4H, NH), 8.05 (d, 2H, Ar, J = 8.5 Hz), 7.22 (d, 2H, Ar, J = 8.4 Hz), 7.08 (s, 1H, NH), 6.04 (s, 1H, CH).
5-(4-Isopropylphenyl)-5,10-dihydropyrido[2,3-d:6,5-d′]dipyrimidine-2,4,6,8-(1H,3H,7H,9H)-tetraone (4d). Orange solid, m.p. (°C): 297–298, m.p. (°C) rep.: 302–304;48 IR (KBr) ν (cm−1): 3178 (NH), 3033 (C–H, sp2 stretch), 1653 (C
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), 1495, 1444 (C
O), 1495, 1444 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C, Ar); 1H NMR (DMSO-d6, 400 MHz) δ (ppm): 9.98 (s, 4H, NH), 7.25 (s, 1H, NH), 6.68 (d, 2H, Ar, J = 11.9 Hz), 6.65 (d, 2H, Ar, J = 7.9 Hz), 5.83 (s, 1H, CH), 2.78 (s, 6H).
C, Ar); 1H NMR (DMSO-d6, 400 MHz) δ (ppm): 9.98 (s, 4H, NH), 7.25 (s, 1H, NH), 6.68 (d, 2H, Ar, J = 11.9 Hz), 6.65 (d, 2H, Ar, J = 7.9 Hz), 5.83 (s, 1H, CH), 2.78 (s, 6H).
5-(3-Nitrophenyl)-5,10-dihydropyrido[2,3-d:6,5-d′]dipyrimidine-2,4,6,8-(1H,3H,7H,9H)-tetraone (4e). White solid, m.p. (°C): 300–302, m.p. (°C) rep.: 297–299;48 IR (KBr) ν (cm−1): 3224 (NH), 2923 (C–H, sp2 stretch), 1694, 1622 (C
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), 1515, 1406 (C
O), 1515, 1406 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C, Ar); 1H NMR (DMSO-d6, 400 MHz) δ (ppm): 10.18 (s, 4H, NH), 7.93 (d, 1H, Ar, J = 5.2 Hz), 7.81 (s, 1H, Ar), 7.47 (d, 2H, CH, J = 4.6 Hz), 7.07 (s, 1H, NH), 6.05 (s, 1H, CH).
C, Ar); 1H NMR (DMSO-d6, 400 MHz) δ (ppm): 10.18 (s, 4H, NH), 7.93 (d, 1H, Ar, J = 5.2 Hz), 7.81 (s, 1H, Ar), 7.47 (d, 2H, CH, J = 4.6 Hz), 7.07 (s, 1H, NH), 6.05 (s, 1H, CH).
5-(4-Bromophenyl)-5,10-dihydropyrido[2,3-d:6,5-d′]dipyrimidine-2,4,6,8-(1H,3H,7H,9H)-tetraone (4f). White solid, m.p. (°C): 304–306, m.p. (°C) rep.: 301–303;48 IR (KBr) ν (cm−1): 3239 (NH), 2923 (C–H, sp2 stretch), 1692 (C
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), 1602, 1473 (C
O), 1602, 1473 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C, Ar); 1H NMR (DMSO-d6, 400 MHz) δ (ppm): 10.04 (s, 4H, NH), 7.31 (d, 2H, Ar, J = 8.0 Hz), 7.02 (s, 1H, NH), 6.95 (d, 2H, Ar, J = 8.0 Hz), 5.89 (s, 1H, CH).
C, Ar); 1H NMR (DMSO-d6, 400 MHz) δ (ppm): 10.04 (s, 4H, NH), 7.31 (d, 2H, Ar, J = 8.0 Hz), 7.02 (s, 1H, NH), 6.95 (d, 2H, Ar, J = 8.0 Hz), 5.89 (s, 1H, CH).
5-(4-Fluorophenyl)-5,10-dihydropyrido[2,3-d:6,5-d′]dipyrimidine-2,4,6,8-(1H,3H,7H,9H)-tetraone (4g). White solid, m.p. (°C): 297–299, m.p. (°C) rep.: 295–297;48 IR (KBr) ν (cm−1): 3209 (NH), 2932 (C–H, sp2 stretch), 1688 (C
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), 1602 (C
O), 1602 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C, Ar); 1H NMR (DMSO-d6, 400 MHz) δ (ppm): 10.08 (s, 4H, NH), 7.09 (s, 1H, NH), 6.99 (d, 2H, Ar, J = 23.1 Hz), 6.95 (d, 2H, Ar, J = 8.5 Hz), 5.89 (s, 1H, CH).
C, Ar); 1H NMR (DMSO-d6, 400 MHz) δ (ppm): 10.08 (s, 4H, NH), 7.09 (s, 1H, NH), 6.99 (d, 2H, Ar, J = 23.1 Hz), 6.95 (d, 2H, Ar, J = 8.5 Hz), 5.89 (s, 1H, CH).
5-(2,4-Dichlorophenyl)-5,10-dihydropyrido[2,3-d:6,5-d′]dipyrimidine-2,4,6,8-(1H,3H,7H,9H)-tetraone (4h). White solid, m.p. (°C): 285–287, m.p. (°C) rep.: 290–292;48 IR (KBr) ν (cm−1): 3361 (NH), 3052 (C–H, sp2 stretch), 1694, 1622 (C
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), 1525, 1401 (C
O), 1525, 1401 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C, Ar); 1H NMR (DMSO-d6, 400 MHz) δ (ppm): 10.08 (s, 4H, NH), 7.32 (s, 1H, Ar), 7.23 (d, 2H, Ar, J = 7.1 Hz), 7.08 (s, 1H, NH), 5.77 (s, 1H, CH).
C, Ar); 1H NMR (DMSO-d6, 400 MHz) δ (ppm): 10.08 (s, 4H, NH), 7.32 (s, 1H, Ar), 7.23 (d, 2H, Ar, J = 7.1 Hz), 7.08 (s, 1H, NH), 5.77 (s, 1H, CH).
5-(p-Tolyl)-5,10-dihydropyrido[2,3-d:6,5-d′]dipyrimidine-2,4,6,8-(1H,3H,7H,9H)-tetraone (4i). White solid, m.p. (°C): 294–296, m.p. (°C) rep.: 298–300;48 IR (KBr) ν (cm−1): 3195 (NH), 3001 (C–H, sp2 stretch), 1612 (C
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), 1402 (C
O), 1402 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C, Ar); 1H NMR (DMSO-d6, 400 MHz) δ (ppm): 10.03 (s, 4H, NH), 7.32 (s, 1H, NH), 6.93 (d, 2H, Ar, J = 7.9 Hz), 6.88 (d, 2H, Ar, J = 7.2 Hz), 5.87 (s, 1H, CH).
C, Ar); 1H NMR (DMSO-d6, 400 MHz) δ (ppm): 10.03 (s, 4H, NH), 7.32 (s, 1H, NH), 6.93 (d, 2H, Ar, J = 7.9 Hz), 6.88 (d, 2H, Ar, J = 7.2 Hz), 5.87 (s, 1H, CH).
5-(4-Hydroxyphenyl)-5,10-dihydropyrido[2,3-d:6,5-d′]dipyrimidine-2,4,6,8-(1H,3H,7H,9H)-tetraone (4j). Yellow solid, m.p. (°C): 255–257, m.p. (°C) rep.: 260–262;48 IR (KBr) ν (cm−1): 3195 (NH), 3001 (C–H, sp2 stretch), 1612 (C
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), 1402 (C
O), 1402 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C, Ar); 1H NMR (DMSO-d6, 400 MHz) δ (ppm): 10.01 (s, 4H, NH), 8.88 (s, 1H, OH), 7.11 (s, 1H, NH), 6.78 (d, 2H, Ar, J = 8.1 Hz), 6.52 (d, 2H, Ar, J = 8.0 Hz), 5.80 (s, 1H, CH).
C, Ar); 1H NMR (DMSO-d6, 400 MHz) δ (ppm): 10.01 (s, 4H, NH), 8.88 (s, 1H, OH), 7.11 (s, 1H, NH), 6.78 (d, 2H, Ar, J = 8.1 Hz), 6.52 (d, 2H, Ar, J = 8.0 Hz), 5.80 (s, 1H, CH).
5-(3-Hydroxyphenyl)-5,10-dihydropyrido[2,3-d:6,5-d′]dipyrimidine-2,4,6,8-(1H,3H,7H,9H)-tetraone (4k). Yellow solid, m.p. (°C): 288–290, m.p. (°C) rep.: 283–285;48 IR (KBr) ν (cm−1): 3195 (NH), 3001 (C–H, sp2 stretch), 1612 (C
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), 1402 (C
O), 1402 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C, Ar); 1H NMR (DMSO-d6, 400 MHz) δ (ppm): 10.04 (s, 4H, NH), 8.91 (s, 1H, OH), 7.27 (s, 1H, Ar), 7.09 (s, 1H, NH), 6.97 (d, 1H, Ar, J = 8.0 Hz), 6.90 (t, 1H, Ar, J = 7.4 Hz), 6.41 (d, 1H, Ar, J = 8.2 Hz), 5.84 (s, 1H, CH).
C, Ar); 1H NMR (DMSO-d6, 400 MHz) δ (ppm): 10.04 (s, 4H, NH), 8.91 (s, 1H, OH), 7.27 (s, 1H, Ar), 7.09 (s, 1H, NH), 6.97 (d, 1H, Ar, J = 8.0 Hz), 6.90 (t, 1H, Ar, J = 7.4 Hz), 6.41 (d, 1H, Ar, J = 8.2 Hz), 5.84 (s, 1H, CH).
5-(4-Methoxyphenyl)-5,10-dihydropyrido[2,3-d:6,5-d′]dipyrimidine-2,4,6,8-(1H,3H,7H,9H)-tetraone (4l). Yellow solid, m.p. (°C): 304–306, m.p. (°C) rep.: 300–302;48 IR (KBr) ν (cm−1): 3195 (NH), 3001 (C–H, sp2 stretch), 1612 (C
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), 1402 (C
O), 1402 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C, Ar); 1H NMR (DMSO-d6, 400 MHz) δ (ppm): 10.02 (s, 4H, NH), 7.67 (s, 1H, NH), 6.90 (d, 2H, Ar, J = 8.4 Hz), 6.70 (d, 2H, Ar, J = 8.4 Hz), 5.85 (s, 1H, CH), 3.66 (s, 3H, CH).
C, Ar); 1H NMR (DMSO-d6, 400 MHz) δ (ppm): 10.02 (s, 4H, NH), 7.67 (s, 1H, NH), 6.90 (d, 2H, Ar, J = 8.4 Hz), 6.70 (d, 2H, Ar, J = 8.4 Hz), 5.85 (s, 1H, CH), 3.66 (s, 3H, CH).
Results and discussion
Preparation and characterization of the catalyst
To prepare the catalyst, we have chosen a mixture of Cu(NO3)2, FeCl3 and NaOH dissolved in deionized water by heating. In the next step, the nano magnetic material in the presence of CTAB was coated with silica. The resulting CuFe2O4 nanoparticles were then allowed to react under vigorous stirring with an appropriate concentration of tetraethyl orthosilicate. Then, the reaction of CuFe2O4@SiO2 with phosphomolybdic acid under room temperature condition for 18 h was carried out. The synthetic path for the preparation of the catalyst is shown in Scheme 1. In order to characterize the catalyst structure, the prepared nanocatalyst was analyzed using FT-IR, XRD, SEM, EDX, TGA, BET and VSM techniques.Fig. 1a–c show the FT-IR spectra of CuFe2O4 MNPs, CuFe2O4@SiO2 and CuFe2O4@SiO2@PMA nanocomposite, respectively. The FT-IR spectrum of CuFe2O4 MNPs shows peaks characteristic of Cu–O at 431 cm−1 and Fe–O 524 cm−1 (Fig. 1a). Fig. 1b shows the FT-IR spectrum of CuFe2O4@SiO2 MNPs. The bands at 1074 cm−1 and 806 cm−1 are attributed to the vibration of Si–O–Si bonds (Fig. 1b). Fig. 1c shows the FT-IR spectrum of CuFe2O4@SiO2@PMA. The broad band at 3342 cm−1 is due to the O–H acidic group. The P–O stretching frequency at 950 cm−1 and the Mo–O stretching frequency at 795 cm−1 are observed (Fig. 1c).
The structures of the CuFe2O4 NPs, CuFe2O4@SiO2, and magnetic nanocatalyst were characterized using XRD as shown in Fig. 2. At 35° were observed the peaks of copper, iron, and molybdenum metals, and the region between 20° and 30° was amorphous, showing a silicate peak. As can be seen in this figure, in the XRD pattern of CuFe2O4, the crystal peaks for (220), (311), (222), (400), (422), (511) and (440) appeared, indicative of the presence of cubic CuFe2O4 with Fd![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m space group (JCPDS card no. 01-077-0010).
m space group (JCPDS card no. 01-077-0010).
In order to prove the synthesis of the nanocatalyst, the SEM image of the hollow mesoporous copper ferrite nanocatalyst functionalized with phosphomolybdic acid is shown in Fig. 3 and it shows the changes in the catalyst surface before and after the reaction with phosphomolybdic acid.
Furthermore, the EDX results are shown in Fig. 4, confirming the signals for iron, copper, molybdenum, oxygen, phosphorus, and silicon elements and so indicating the successful synthesis of the nanocatalyst. The results are shown in Table 1, with the presence of Mo: 2.71, Cu: 3.36, Fe: 10.37, P: 2.06, Si: 23.83 and O: 58.41 in the nanocomposite. Also, a uniform distribution of the elements among the samples, as well as their relative concentration can be seen in the results obtained from the elemental mapping of the samples. Based on the elements present in the CuFe2O4@SiO2@PMA nanocatalyst, the mapping images are shown in Fig. 5.
| Entry | Element | Atomic% |
|---|---|---|
| 1 | O | 58.41 |
| 2 | Si | 23.83 |
| 3 | P | 2.06 |
| 4 | Mo | 2.71 |
| 5 | Fe | 10.37 |
| 6 | Cu | 3.36 |
The magnetic properties of CuFe2O4 nanoparticles and CuFe2O4@SiO2@PMA were investigated by the VSM technique at room temperature (Fig. 6). As can be seen, the prepared nanocomposite exhibits paramagnetic behaviour with saturation magnetization of 2.1 emu g−1 and for CuFe2O4 nanoparticles the value is 7.9 emu g−1. This result shows that the saturation magnetization of the sample decreases with functionalization and coating. The reason for the easy separation of CuFe2O4@SiO2@PMA as a magnetic nanocatalyst is its high saturation magnetization.
TGA provides valuable information on the thermal stability and the amount of organic and inorganic components of the catalyst. The weight loss stage below 200 °C usually corresponds to the loss of physically adsorbed water and volatile organic solvents. The weight loss between 300 and 800 °C related to the decomposition of organic functional groups confirms the successful functionalization of the nanoparticles with phosphomolybdic acid (Fig. 7).
The N2 adsorption–desorption isotherms of CuFe2O4@SiO2@PMA are shown in Fig. 8. The surface area of CuFe2O4@SiO2@PMA is determined as 95.241 m2 g−1 and the average pore diameter of the catalyst is 5.1765 nm. These results successfully demonstrated the porosity and nanostructure of the catalyst.
 | ||
| Fig. 8 N2 adsorption–desorption: (a) BET results for CuFe2O4@SiO2@PMA; (b) the isotherm of CuFe2O4@SiO2@PMA; and (c) the BJH results for CuFe2O4@SiO2@PMA. | ||
Evaluating the catalytic activity of the nanocomposite
In this protocol, the catalytic activity of CuFe2O4@SiO2@PMA in the synthesis of chromene compounds was investigated. In order to determine the most appropriate amount of magnetic nanocatalyst, solvent, and temperature for the reaction to obtain the highest efficiency, the reaction of barbituric acid, ammonium acetate and benzaldehyde was treated as a model (Scheme 2). At first, to determine the most suitable catalyst amount in the reaction, different amounts of prepared nanocomposite were used as heterogeneous catalyst in the model reaction under thermal conditions and EtOH solvent. The results are indicated in Table 2.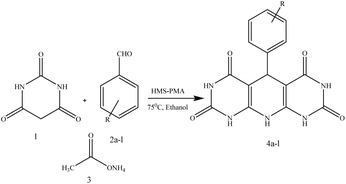 | ||
| Scheme 2 The synthesis of 5-phenyl-5,10-dihydropyrido[2,3-d:6,5-d′]dipyrimidine-2,4,6,8-(1H,3H,7H,9H)-tetraone using the heterogeneous nanocatalyst. | ||
As can be seen from Table 2, the reaction was efficiently performed using 0.03 g of the catalyst at 75 °C and ethanol as a solvent and the desired product was obtained (Table 2, entry 3).
Then, to optimize the reaction temperature, the reaction was carried out in ethanol at various temperatures (Table 3). The best results were obtained at 75 °C (Table 3, entry 4).
In continuation, the reaction solvent was optimized. The reaction of benzaldehyde (0.5 mmol), barbituric acid (1 mmol), ammonium acetate (0.6 mmol) and CuFe2O4@SiO2@PMA (0.03 g) as catalyst under thermal conditions (75 °C) was carried out in various solvents (Table 4). The best results were obtained using ethanol as a solvent (Table 4, entry 4).
Finally after optimization of the reaction conditions, syntheses of 1,4-dihydropyridine derivatives were carried out with the reaction of different aromatic aldehydes including those with electron-withdrawing and electron-donating groups. The corresponding results are depicted in Table 5.
| Entry | R | Product | Time (min) | Yieldb (%) |
|---|---|---|---|---|
| a Reaction conditions: benzaldehyde (0.5 mmol), barbituric acid (1 mmol), ammonium acetate (0.6 mmol), catalyst (0.03 g) under thermal conditions (75 °C) and ethanol as a solvent.b Isolated yields. | ||||
| 1 | H | 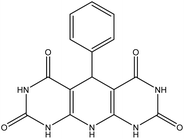 |
10 | 90 |
| 2 | 4-Chloro | 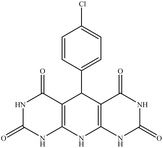 |
7 | 95 |
| 3 | 4-Nitro | 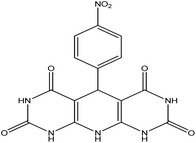 |
9 | 92 |
| 4 | 4-Dimethyl | 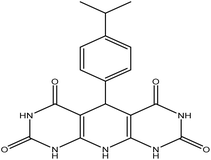 |
14 | 85 |
| 5 | 3-Nitro | 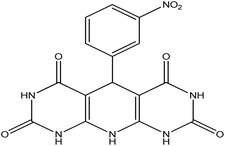 |
9 | 90 |
| 6 | 4-Bromo | 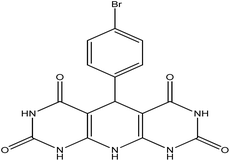 |
10 | 94 |
| 7 | 4-Fluoro |  |
9 | 90 |
| 8 | 2,4-Dichloro |  |
8 | 93 |
| 9 | 4-Methyl |  |
12 | 80 |
| 10 | 4-Hydroxy |  |
25 | 80 |
| 11 | 3-Hydroxy | 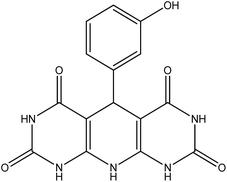 |
15 | 85 |
| 12 | 4-Methoxy | 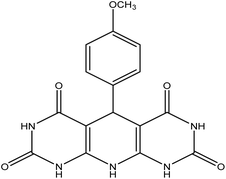 |
18 | 75 |
Furthermore, the present work was compared with other previously reported works concerning the synthesis of 1,4-dihydropyridines. The corresponding results are indicated in Table 6. The present method compared with the previously reported ones has some advantages such as high yields, short reaction times, simplicity of procedure, easy reaction work-up, recyclability and resulting products having high purity.
| Entry | Product | Time (min) | Yield (%) | Conditions | Ref. |
|---|---|---|---|---|---|
| 1 | 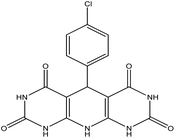 |
45 | 92 | DIPEAc, RT, solvent free | 32 |
| 7 | 95 | CuFe2O4@SiO2@PMA, 75 °C, ethanol | This work | ||
| 2 | 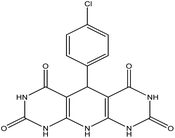 |
60 | 95 | MWCNTs@L-His/Cu(II), reflux, ethanol | 33 |
| 7 | 95 | CuFe2O4@SiO2@PMA, 75 °C, ethanol | This work | ||
| 3 | 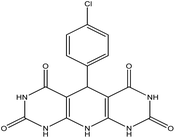 |
60 | 88 | US, 60 °C, H2O | 49 |
| 7 | 95 | CuFe2O4@SiO2@PMA, 75 °C, ethanol | This work | ||
| 4 | 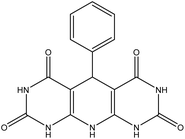 |
60 | 87 | US, 60 °C, H2O | 49 |
| 10 | 90 | CuFe2O4@SiO2@PMA, 75 °C, ethanol | This work |
Proposed reaction mechanism for the synthesis of 1,4-dihydropyridine derivatives
The proposed reaction mechanism for the synthesis of 1,4-dihydropyridines involves the reaction of barbituric acid, aromatic aldehydes, and ammonium acetate in the presence of the CuFe2O4@SiO2@PMA catalyst as illustrated in Scheme 3. Initially, the carbonyl group of the benzaldehyde is activated by the catalyst resulting in a reduction of the electron density on the carbonyl carbon, making it more susceptible to nucleophilic attack. Then, the Knoevenagel condensation reaction between the activated aldehyde and barbituric acid gives compound I.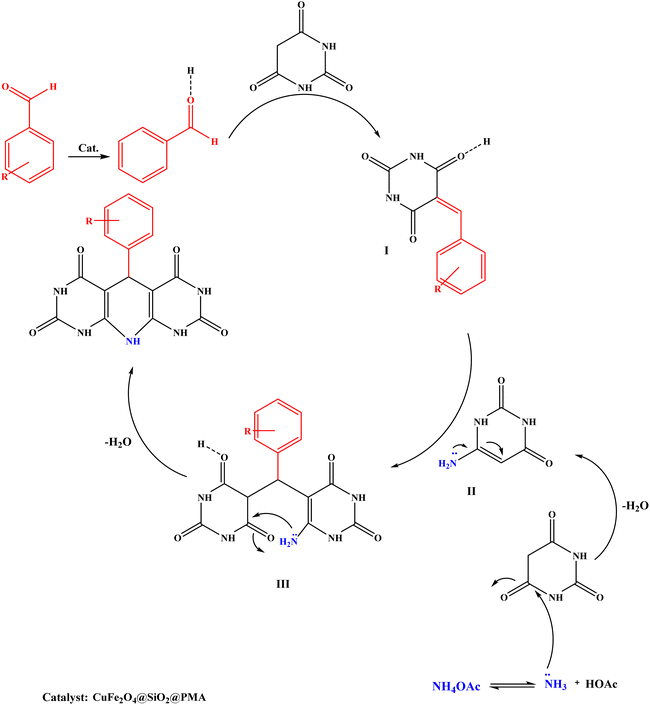 | ||
| Scheme 3 The proposed reaction mechanism for the synthesis of 1,4-dihydropyridine derivatives with CuFe2O4@SiO2@PMA as catalyst. | ||
Simultaneously with this reaction, ammonia reacts with another barbituric acid and creates enamine compound II. In the next step, a Michael-type addition of compound II to compound I and tautomerization lead to intermediate III. The product is formed by intramolecular attack of the amino group onto the activated carbonyl group in intermediate III. This process is followed by cyclization and dehydration to produce 1,4-dihydropyridine compounds.50
Catalyst reusability
After the completion of the reaction, the heterogeneous catalyst was separated from the reaction mixture using an external magnet. Following its separation, it was washed with ethyl acetate to remove any remaining organic compounds. Then, the recovered catalyst was dried in an oven at 50 °C and reused in the model reaction. Fig. 9 shows its catalytic activity for five cycles. Furthermore, to prove the stability and performance of the recovered nanocatalyst, the recovered catalyst was analyzed using FT-IR, XRD and FE-SEM methods. As seen, there is no significant difference between the FT-IR, XRD and FE-SEM analyses of the initial and recovered nanocomposite (Fig. 10–12). To test the heterogeneous nature of the catalyst, a hot filtration test was carried out for the synthesis of 4j product under optimum reaction conditions. After 15 min of the reaction, the catalyst was separated from the reaction mixture using an external magnet and then the filtrate was allowed to react further for the other half of the reaction time. The results showed no further product after the separation of the catalyst, and thus the catalyst acts heterogeneously in the reaction media without any leaching of its active sites.Conclusions
In this research, different compounds of dihydropyrido derivatives were synthesized. The synthesis of these derivatives was carried out under mild conditions and in the presence of a hollow mesoporous nanocomposite. This nanocomposite was prepared and identified as an efficient catalyst for the one-pot synthesis of 1,4-dihydropyridine derivatives under clean and mild reaction conditions. High yields, short reaction times, environmental friendliness, reusability and the easy recovery of the catalyst using an external magnet are the important features of this methodology. The products were identified based on physical and spectroscopic data, such as melting point, FT-IR and 1H NMR analyses. Also, the catalyst was characterized using FT-IR, XRD, EDX, SEM, TGA, BET and VSM techniques.Conflicts of interest
There are no conflicts to declare.Data availability
The datasets generated and analyzed during the current study are available from the Hossein Naeimi repository (E-mail: naeimi@kashanu.ac.ir).Supplementary information (SI): FT-IR and 1H NMR spectra. See DOI: https://doi.org/10.1039/d5ra06191a.
Acknowledgements
The authors are grateful to University of Kashan for supporting this work through grant no. 159148/86.References
- M. Fan, J. Yan, H. Tan, Y. Miao and X. Hu, J. Mater. Chem. B, 2014, 2, 8399–8405 RSC.
- B. Gleich, C. Rümenapp and A. Haase, Pharm. Res., 2012, 29, 1165–1179 CrossRef.
- Z. Zhou, L. Yang, J. Gao and X. Chen, Adv. Mater., 2019, 31, 1804567 CrossRef.
- N. A. Frey, S. Peng, K. Cheng and S. Sun, Chem. Soc. Rev., 2009, 38, 2532–2542 RSC.
- L. Gloag, M. Mehdipour, D. Chen, R. D. Tilley and J. J. Gooding, Adv. Mater., 2019, 31, 1904385 CrossRef CAS.
- S. Nikazar, M. Barani, A. Rahdar, M. Zoghi and G. Z. Kyzas, ChemistrySelect, 2020, 5, 12590 CrossRef CAS.
- L. Arda, N. Dogan and C. Boyraz, J. Supercond. Novel Magn., 2018, 31, 365 CrossRef CAS.
- M. K. Satheeshkumar, E. R. Kumar, P. Indhumathi, C. Srinivas, M. Deepty, S. Sathiyaraj, N. Suriyanarayanan and D. L. Sastry, Inorg. Chem. Commun., 2020, 111, 107578 CrossRef CAS.
- S. M. Rathod, A. R. Chavan, S. S. Jadhav, K. M. Batoo, M. Hadi and E. H. Raslan, Chem. Phys. Lett., 2021, 765, 138308 CrossRef CAS.
- F. Sharifianjazi, M. Moradi, N. Parvin, A. Nemati, A. J. Rad, N. Sheysi, A. Abouchenari, A. Mohammadi, S. Karbasi, Z. Ahmadi, A. Esmaeilkhanian, M. Irani, A. Pakseresht, S. Sahmani and M. S. Asl, Ceram. Int., 2020, 46, 18391 CrossRef CAS.
- Y. J. Yang, C. Jiang, S. Chen, N. Wang, P. Yang, M. Liu and Y. Cheng, J. Electroanal. Chem., 2022, 918, 116385 CrossRef.
- K. Shahzad, S. Mushtaq, M. Rizwan, W. Khalid, M. Atif, F. Din Ud, N. Ahmad, R. Abbasi and Z. Ali, Mater. Sci. Eng., C, 2021, 119, 111444 CrossRef CAS PubMed.
- R. A. Raimundo, V. D. Silva, L. S. Ferreira, F. J. A. Loureiro, D. P. Fagg, D. A. Macedo, U. U. Gomes, R. M. Gomes, M. M. Soares and M. A. Morales, J. Alloys Compd., 2023, 940, 168783 CrossRef CAS.
- S. Kumar, A. Ohlan, P. Kumar and V. Verma, J. Supercond. Novel Magn., 2020, 33, 1187 CrossRef CAS.
- A. A. Al-Juaid, Mater. Res. Express, 2023, 10, 026101 CrossRef.
- B. Yalcin, J. Nanopart. Res., 2022, 24, 271 CrossRef CAS.
- M. Chithra, C. N. Anumol, V. Argish, B. N. Sahu and S. C. Sahoo, Mater. Sci., 2023, 34, 806 CAS.
- G. A. Lone and M. Ikram, J. Alloys Compd., 2023, 934, 167891 CrossRef CAS.
- D. H. Everett, Pure Appl. Chem., 1972, 31, 577–638 CrossRef.
- H. Zheng, F. Gao and V. Valtchev, J. Mater. Chem. A, 2016, 4, 16756–16770 RSC.
- X. Du and J. He, Langmuir, 2010, 26, 10057–10062 CrossRef CAS PubMed.
- A. B. D. Nandiyanto, A. Suhendi, T. Ogi, T. Iwaki and K. Okuyama, Colloids Surf., A, 2012, 396, 96–105 CrossRef CAS.
- C. Yu, B. Tian, J. Fan, G. D. Stucky and D. Zhao, Chem. Commun., 2001, 2726–2727 RSC.
- T. Asefa and Z. Tao, Can. J. Chem., 2012, 90, 1015–1031 CrossRef CAS.
- M. A. Balestriere, K. Schuhladen, K. H. Seitz, A. R. Boccaccini, S. M. Cere and J. Ballarre, J. Electroanal. Chem., 2020, 876, 114735 CrossRef CAS.
- W. Zhang, H. He, Y. Tian, K. Lan, Q. Liu, C. Wang, Y. Liu, A. Elzatahry, R. Che, W. Li and D. Zhao, Chem. Sci., 2019, 10, 1664–1670 RSC.
- X. Lu, G. Hasegawa, K. Kanamori and K. Nakanishi, J. Sol-Gel Sci. Technol., 2020, 1–21 Search PubMed.
- Y. Wu, W. Zhao, Y. Qiang, Z. Chen, L. Wang, X. Gao and Z. Fang, Carbon, 2020, 159, 292–302 CrossRef CAS.
- M. Jang, Synthesis and Biological Evaluation of Bicyclic Heterocycles, 2009, pp. 1–150 Search PubMed.
- T. Mohamed, M. K. Mann and P. P. N. Rao, RSC Adv., 2017, 736, 22360–22368 RSC.
- S. Rostamizadeh, L. Tahershamsi and N. Zekri, J. Iran. Chem. Soc., 2015, 12, 1381–1389 CrossRef CAS.
- M. R. Bhosle, L. D. Khillare, J. R. Mali, A. P. Sarkate, D. K. Lokwani and S. V. Tiwari, New J. Chem., 2018, 42, 18621–18632 RSC.
- H. Saeidiroshan and L. Moradi, J. Organomet. Chem., 2019, 893, 1–10 CrossRef CAS.
- D. M. Gholami, N. Foroughifar, A. Khajeh-Amiri and H. N. Pasdar, J. Chin. Chem. Soc., 2018, 65, 1356–1369 CrossRef.
- H. Naeimi, A. Didar, Z. Rashid and Z. Zahraie, J. Antibiot., 2017, 70, 845–852 CrossRef CAS.
- H. Singh, A. Kachore, V. Aggarwal, E. Bala, Saima, R. Kumar, P. Kumar and P. K. Verma, ChemistrySelect, 2025, 10, e202501004 CrossRef CAS.
- H. Singh, A. Kachore, V. Aggarwal, E. Bala, Saima, M. Selvaraj and P. K. Verma, Chem. Rec., 2025, 25, e202500069 CrossRef CAS PubMed.
- E. Bala, A. Kachore, V. Aggarwal, H. Singh, Saima, M. Selvaraj, R. Kumar and P. K. Verma, JPhys Mater., 2025, 708, 135961 CAS.
- A. Kachore, E. Bala, V. Aggarwal, H. Singh, Saima, M. H. Adam Suleiman, M. Selvaraj and P. K. Verma, J. Ind. Eng. Chem., 2025, 146, 494–505 CrossRef CAS.
- V. Aggarwal, A. Kachore, E. Bala, H. Singh, M. Selvaraj, M. A. Assiri, Saima, R. Kumar and P. K. Verma, J. Phys. Chem. Solids, 2026, 208, 113060 CrossRef CAS.
- V. Aggarwal, A. Kachore, E. Bala, H. Singh, M. Selvaraj, M. A. Assiri, Saima, R. Kumar, R. Sharma and P. K. Verma, J. Taiwan Inst. Chem. Eng., 2025, 175, 106249 CrossRef CAS.
- H. Singh, Himanshu, A. Kachore, V. Aggarwal, E. Bala, Saima, R. Kumar and P. K. Verma, Langmuir, 2025, 41, 23163–23181 CrossRef CAS.
- G. Lavanya, S. A. Khadar, I. Jariya, M. Sasikala, V. Kalpana, A. Padmanaban, J. Wang and B. M. Ali, ChemistrySelect, 2024, 9, e202403664 CrossRef CAS.
- M. Dadaei and H. Naeimi, RSC Adv., 2021, 11, 15360 RSC.
- H. Naeimi and M. Dadaei, RSC Adv., 2015, 5, 76221 RSC.
- M. Ansari, A. H. Ghasemi and H. Naeimi, RSC Adv., 2025, 15, 18535–18547 RSC.
- H. Hassanpour and H. Naeimi, RSC Adv., 2024, 14, 17296–17305 RSC.
- D. M. Gholami, N. Foroughifar, A. Khajeh-Amiri and H. N. Pasdar, J. Chin. Chem. Soc., 2018, 65, 1356–1369 CrossRef.
- M. H. Mosslemin and M. R. Nateghi, Ultrason. Sonochem., 2010, 17, 162–167 CrossRef CAS PubMed.
- H. Naeimi and A. Didar, Ultrason. Sonochem., 2017, 34, 889–895 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2025 |