Recent progress in electrochemical rearrangement reactions
Zhaojiang
Shi†
 *,
Chao-Yan
Wen†
,
Li-Xue
Yang
,
Jianan
Li
* and
Xiaoxia
Sun
*,
Chao-Yan
Wen†
,
Li-Xue
Yang
,
Jianan
Li
* and
Xiaoxia
Sun
 *
*
Jiangxi Provincial Key Laboratory of Organic Functional Molecules; Institute of Organic Chemistry, Jiangxi Science and Technology Normal University, Nanchang 330013, P. R. China. E-mail: zj1084552735@163.com; lijianan77@yeah.net; xxsun@jxstnu.edu.cn
First published on 28th January 2025
Abstract
Rearrangement reactions are among the most critical transformations in synthetic chemistry, enabling the construction of complex molecules from simple starting materials through the cleavage and reformation of chemical bonds. Synthetic electrochemistry, as a sustainable synthesis method, eliminates the need for stoichiometric redox reagents, significantly advancing green chemistry. Over the past decade, numerous electrochemically promoted rearrangement reactions have been developed, demonstrating the broad applicability of electrochemistry in facilitating rearrangement processes. This review highlights the application of electrochemistry in rearrangements, focusing on functional group migrations, ring expansion reactions, and selective migratory cyclization reactions.
1. Introduction
A reaction in which the molecular skeleton undergoes cleavage and reorganization through chemical bonds to generate structural isomers is typically known as a rearrangement. This process normally involves the migration of functional groups within the molecule to form isomers of the original compound or the release of simple molecules (such as H2O or SO2) to produce other compounds. Rearrangement reactions enable the synthesis of complex target compounds from simple starting materials by adjusting the position of atoms or groups within or between molecules.1 Therefore, these reactions can effectively build carbon skeletons that are difficult to synthesize by traditional methods, greatly aiding the synthesis of natural products and drugs. Traditional rearrangement reactions are typically categorized as ionic nucleophilic rearrangements and electrophilic rearrangements.2 These reactions have been extensively developed, and many named reactions fall into this category,3 such as Hofmann,4 Smiles–Truce,5 and Beckmann.6 Although this type of rearrangement has been well developed, the process involves the use of stoichiometric oxidants and toxic, harmful reagents, which limits practical application development.In recent years, due to the promotion of the green chemistry concept, the electrochemical synthesis technology has experienced a revival.7 Electrochemical organic synthesis is considered a green and efficient method for carrying out oxidative or reductive reactions through electron transfer, eliminating the need for stoichiometric oxidants and reductants.8 Compared to traditional organic synthesis, electrochemical organic synthesis offers advantages of mild conditions, shorter reaction times, and simpler operation. Furthermore, most reactions do not require special conditions such as high temperature or pressure, making them more beneficial for industrial applications. Recently, there has been significant progress in electrochemical-mediated cyclizations,9 C–H functionalization reactions,10 and difunctionalization of alkenes and alkynes,11 among others.12 With the rapid growth of electrochemical chemistry, electrochemical-mediated rearrangement reactions have emerged as an important research area in organic chemistry, garnering widespread attention from the chemical community.13 Thus, in this review, we summarize the latest progress in the field of electrochemical rearrangements over the past decade, focusing on functional group migrations, ring expansions and migratory cyclization reactions, to provide new perspectives and references for researchers in this field (Scheme 1).
2. Electrochemical functional group migrations
Functional group migration is one of the most effective synthetic strategies for rapidly constructing high-value-added compounds from simple molecules. Among these reactions, the selective migration of groups remains one of the most challenging issues. Traditionally, such transformations often rely on stoichiometric oxidants or expensive photocatalysts, which has limited their further development. With the rapid advancement of electrochemical technologies, several sustainable electrochemically driven functional group migration reactions have been developed.2.1 1,2-Functional group migrations
The traditional Hofmann rearrangement requires stoichiometric halogen and sodium hydroxide, and the use of these toxic and harmful reagents limits its practical application. To address this, Zhang et al. reported an electrochemical Hofmann rearrangement using NaBr as a mediator in an undivided cell under constant current conditions (Scheme 2A).14 The synthetic strategy is well compatible with various commercially available medicine derivatives (2a and 2c) and amantadine derivatives (2b) demonstrating its practicality in synthesis. The proposed mechanism begins with the cathodic reduction of MeOH, which generates H2 and the base MeO−. Meanwhile, the bromine produced at the anode reacts with amide (1a) to form an intermediate (A). Intermediate (A) then undergoes Hofmann rearrangement, followed by nucleophilic attack by the methoxy group to form the target product (2d, Scheme 2B). In 2023, Cantillo and co-workers realized the electrochemical Hofmann rearrangement of primary amines to synthesize methyl carbamates using a spinning cylinder electrode cell.15Recently, the bifunctionalization of alkenes mediated by free radicals has emerged as a powerful tool for the synthesis of various bioactive molecules.16 In 2019, the Lei group reported the electrochemical oxidation of allyl alcohols (3) to synthesize β-trifluoromethyl ketones (5) via a 1,2-migration process (Scheme 3A).17 This reaction features mild conditions and simple operation, and eliminates the need for metal catalysis or chemical oxidants. Notably, this protocol enables not only the migration of various aryl groups (5a–c) but also the formation of alkyl-migration product (5d). Subsequently, Ackermann et al. extended this approach, achieving electrochemical 1,2- and 1,4-aryl migration of allyl alcohols (3, Scheme 3B).18
In 2023, the Xiong group developed an efficient electrochemical 1,2-migration reaction of allyl alcohol derivatives (7) to synthesize γ-keto sulfones (10) containing a β-quaternary carbon center (Scheme 4A).19 A possible mechanism was proposed (Scheme 4B). Firstly, p-toluenesulfohydrazide (9) loses electrons and deprotonates at the anode forming an unstable intermediate (A), which then releases N2 and transforms into the sulfonyl radical intermediate (B). Next, intermediate (B) reacts with substrate (8a) via radical addition to generate intermediate (C). Subsequently, intermediate (C) undergoes a 1,2-phenyl migration producing intermediate (D), which is further oxidized at the anode to form the cation (E). Finally, intermediate (E) undergoes deprotonation to yield the target product (10a).
Although the electrochemical 1,2-migration of allyl alcohols has been well developed, there are few reports on the corresponding flow chemical syntheses. Recently, Fang and co-workers revealed a continuous-flow electrochemical synthesis of α-alkynyl ketones (13 and 15) via radical 1,2-alkynyl migration (Scheme 5).20 This strategy eliminates the need for metal catalysts or chemical oxidants, operates under mild conditions, exhibits high reaction efficiency, and accommodates a broad substrate range. As a result, this continuous-flow electrochemical synthesis strategy has broad potential applications.
1,2-Diaryl compounds are widely found in various pharmaceutical molecules.21 These compounds can be efficiently synthesized via the 1,2-aryl migration strategy, in which the 1,1-diaryl ethyl radical is the key intermediate to realize 1,2-aryl migration.22 Traditional synthesis strategies often require the additional introduction of free radicals or chemical oxidants, which limits the substrate scope.23 Recently, electrochemical decarboxylation has emerged as a promising alternative without transition-metal catalysts, photocatalysts, external oxidants, or high temperatures.24 Therefore, electrochemical decarboxylation is an ideal method to obtain radical species from carboxylic acids. In 2020, the Lei group reported an electrochemical oxidative decarboxylation and 1,2-aryl migration strategy for the synthesis of 1,2-diaryl ethers (18, Scheme 6A).25 The synthetic strategy exhibits good substrate compatibility, and methanol (18a) as well as cyclohexanol (18b) can be effectively applied in the reaction. It is worth noting that the reaction can still be realized when one of the two aryl groups is replaced by methoxy (18c) or benzyl (18d).
Through cyclic voltammetry studies, it is determined that the oxidation peak appears at 1.68 V (vs. Ag/AgCl in MeOH/DCE) when 16a and 1.0 equivalent of nBu4NOAc are mixed. Mechanistic investigations suggested that 3,3-diphenylpropionic acid (16a) undergoes deprotonation in the presence of acetate to produce carboxylate (A), which is oxidized at the anode to generate a carboxyl radical (B). The next decarboxylation reaction can obtain the primary carbon radical (C). A subsequent 1,2-aryl migration and anodic oxidation step generate a benzyl carbocation (E). Finally, the reaction between the benzyl carbocation (E) and methanol can yield the desired product (18a) with the help of acetate (Scheme 6B).
The Beckmann rearrangement has been widely applied in the synthesis of pharmaceuticals, pesticides, and natural products.26 It remains one of the most important methods for synthesizing amide compounds. Recently, several improved Beckmann rearrangement reactions have been reported, including metal-complex-catalyzed,27 boronic acid-mediated,28 and photocatalytic29 rearrangement reactions. Although these methods have shown good results, the development of more efficient and environmentally friendly synthesis strategies remains essential. The Guan group developed an electrochemical Beckmann rearrangement (Scheme 7A).30 This strategy efficiently synthesizes amide derivatives (20) by direct electrolysis of ketoximes at room temperature under constant current conditions. The reaction exhibits a broad substrate scope and functional group compatibility, accommodating substrates such as aniline (20a), indole (20b), styrene (20c), and methyl (20d).
Mechanistic studies proposed the following pathway that ketoxime (20e, Eox = 1.42 V vs. Ag/AgCl in DCE/HFIP) undergoes a single-electron transfer to form a radical cation (A). Then intermediate (A) is attacked by nucleophilic water in the system to form intermediate (B), which undergoes a 1,2-rearrangement to generate a radical intermediate (C). Subsequently, the intermediate (C) is reduced at the cathode or reacts with another ketoxime (19) via chain propagation, closing the catalytic cycle to obtain the intermediate (D), which is protonated to form the intermediate (E). Finally, intermediate (E) loses one molecule of water to produce (F), which tautomerizes to form the product amide (20e, Scheme 7B).
Alkynes are one of the most significant organic functional groups, commonly found in many natural products as well as bioactive compounds.31 Meanwhile, alkynes are often used as key intermediates in synthesis. Therefore, it is crucial to develop efficient and convenient synthetic methods for creating unsymmetrical alkynes. In 2021, Didier and co-workers developed an electrochemical intramolecular rearrangement of trialkynylorganoborates to synthesize unsymmetrical alkyne compounds (17, Scheme 8A).32 All kinds of alkynes (17a and 17b), heterocycles (17c), and natural products (17d) can be successfully produced using this simple and mild reaction.
The authors proposed a potential mechanism (Scheme 8B). First, the aryl group of the trialkynylarylborate (16a, Eox = 1.14 V vs. SCE) undergoes oxidation at the anode, resulting in the formation of intermediate (A). Subsequently, a C–C bond can be created through either π bond addition or σ bond cleavage, leading to intermediates (B) and (C), respectively. Finally, intermediate (C) undergoes further oxidation to eliminate boron, facilitating the re-aromatization of the desired product (17).
2.2 1,3-Functional group migrations
In early investigations, imine derivatives were synthesized by the 1,3 (O → N) acyl migration strategy (Mumm rearrangement) of O-acyl isoamides. However, this approach requires the preparation of unstable precursor imidoyl chlorides,33 which restricts its applicability. The synthesis of imides using O-acyl isoamides generated in situ from readily available substrates under mild conditions is environmentally desirable. To address this, the Sun group developed an electrochemical functionalization of alkenes by a four-component cascade reaction cascade to synthesize imides (20, Scheme 9A).34 This reaction uses simple and readily accessible starting materials, the conditions are mild, and the substrate range is wide. Different types of benzoic acids (20a–c) and alcohol (20d) can be effectively utilized. Additionally, the electrochemical four-component reaction can be successfully scaled up to gram quantities, highlighting the practical utility of this synthesis method.Based on mechanistic studies (Scheme 9B), it is proposed that styrene (18a, Eox = 1.6 V vs. SCE in MeCN) is oxidized at the anode to form a free radical cation intermediate (A). This intermediate is then reacted with the nucleophilic reagent MeO− to create radical intermediate (B), which is subsequently anodized to produce cation (C). The intermolecular trapping of cation (C) with acetonitrile leads to the formation of carbocation (D), which can combine with PhCOO− to generate intermediate (E). Finally, due to its structural instability, intermediate (E) quickly undergoes a Mumm rearrangement to furnish the desired product (20a).
Recently, the Sun group reported an electrochemical three-component reaction cascade Mumm rearrangement which was developed for the synthesis of imides (22, Scheme 10A).35 This method utilizes commercially available aryl acids (19), nitriles, and alkylbenzenes (21) as substrates, allowing for the creation of various imine derivatives through electrolysis at a constant current (7 mA) without using metal catalysts or chemical oxidants. Following this, the Huang group introduced an electrochemically promoted decarboxylation of carboxylic acid (23) followed by a Mumm rearrangement to produce imide derivatives (24, Scheme 10B).36 The reaction conditions are mild, exhibit good tolerance for different functional groups, and yield a range of imides in moderate to high amounts.
Incorporating –CF3 and –CN functional groups into carbonyl structural units can alter the inherent properties of drug molecules.37 Among these, trimethylsilyl nucleophilic reagents, such as Me3SiCF3 and Me3SiCN, are easily available,38 and they are non-toxic trifluoromethyl and cyanide sources that react with carbonyl electrophilic reagents, in which the slow release of CF3− or CN− anions in aprotic solvents requires necessary initiators.39 To address this, Yuan et al. developed an initiator-free electrochemical trifluoromethylsilylation and cyanosilylation of aldehydes (Scheme 11A).40 The reaction has a wide range of substrates, and accommodates aromatic aldehydes (27a–d), heterocyclic aldehydes (27e and 27f), and alkyl aldehydes (27g and 27h). Mechanistic studies indicated that Me3SiCF3 (26a) is initially reduced at the cathode to form an anionic intermediate (A), which then reacts with benzaldehyde (25a) to form another intermediate (B). This intermediate (B) subsequently undergoes intramolecular CF3 migration and loses electrons at the anode, resulting in the desired product (27a, Scheme 11B).
The selective functionalization of the allylic C (sp3)–H bond enables the construction of a diversified molecular skeleton which can be further transformed into high-value-added products.41 Currently, allylic C–H bond functionalization is primarily restricted to terminal alkenes or substrates with a single allylic site,42 which limits its broader application. Additionally, isothiocyanates are commonly found in natural products and functional materials.43 Conventional methods for synthesizing isothiocyanates are not only harsh but also involve toxic thio-reagents.44 Therefore, it is crucial to develop greener and more efficient direct C(sp3)–H isothiocyanation techniques. Recently, the Guo group reported an electrochemically promoted allylic C(sp3)–H isothiocyanation of internal alkenes (Scheme 12A).45 The method not only has high chemical selectivity and position selectivity but also has wide functional group tolerance and excellent selectivity (30a and 30b). Additionally, it can be applied for the late-stage isothiocyanation of bioactive molecules (30c).
The control experiments and DFT calculations showed that TMSNCS plays a dual role of HAT reagent and coupling partner (Scheme 12B). Initially, the SCN anion is oxidized at the anode to produce thiocyanogen, which exists in equilibrium with the SCN radical. Subsequently, SCN radicals can selectively capture the hydrogen atom of the substrate (28a) and generate allylic radical species (A). Then, intermediate (A) is captured by (SCN)2 to produce intermediate (B). Finally, the thermodynamically favored isothiocyanate (30a) is obtained via a [3,3]-sigmatropic rearrangement.
2.3 1,4-Functional group migrations
The remote functional group transfer strategy is a special type of organic transformation in the field of organic synthesis because it can synthesize some high-value-added compounds that are difficult to obtain by traditional methods.46 Among them, remote functional group migration by forming intramolecular cyclic intermediates or transition states has been developed.47 These reactions mainly focus on photocatalysis and metal catalysis.1,48 In 2018, the Pan group reported the electrochemical enhancement of 1,4-alkynyl migration in tertiary alcohol derivatives, achieving 1,2-sulfonylation/alkynylation of alkenes (Scheme 13A).49 The reaction affords various products of alkynyl migration (33a and 33b) or alkenyl migration (33a and 33b) with moderate to excellent yields under constant current electrolysis.Mechanistic investigations suggested that sodium sulfinate (32) is oxidized at the anode to produce oxygen-centered radical (A). This radical intermediate (A) can resonate to form a more stable sulfonyl radical (B). Subsequently, the radical (B) and substrate (31) form an intermediate (C) through radical addition. Following this, intermediate (C) undergoes intramolecular radical cyclization to produce a vinyl radical (D), which then experiences selective C–C bond cleavage to yield a free radical (E). Finally, the intermediate (E) is dehydrogenated and oxidized at the anode to generate the target product (33, Scheme 13B).
Meanwhile, the Guo group reported a method for the direct electrooxidation sulfonylation/heteroarylation of alkenes using sulfinic acid, which operates under mild conditions and is effective for various heteroaromatic hydrocarbons (Scheme 14A).50 Shortly after, the Wang group developed the electrochemical bifunctionalization of olefins through the remote migration of radical groups (Scheme 14B).51 In 2022, Morrill et al. developed an electrochemical azidocyanation reaction of olefins through a 1,4-nitrile strategy. This method has high substrate tolerance and is suitable for various alkene-containing cyanohydrins, providing a good way to synthesize 1,2-azidonitriles (Scheme 14C).52 In the same year, Ye et al. developed the electrochemical oxidation of inorganic sulfites with alcohols to generate alkoxysulfonyl radicals, which are used in the subsequent difunctionalization of allylic alcohols to provide various sulfonate esters (Scheme 14D).53
In 2020, the Guo group reported that the electrochemically triggered process by the N radical promotes remote heteroaryl migration (Scheme 15A).54 This approach is environmentally friendly and efficient, exhibiting strong compatibility with various functional groups and high atomic efficiency. Mechanistic studies revealed that intermediate (A) is formed from the substrate (37a) through TFE anion exchange. Concurrently, the organic catalyst (Cat. 1, Eox = 0.84 V and 1.65 V vs. SCE in MeCN) is oxidized at the anode to form a stable radical cation intermediate (Cat. 1*), and then the intermediate (A) is oxidized to form a radical intermediate (B). Then, the intermediate (B) undergoes intramolecular radical cyclization to obtain spiro radical intermediate (C). Finally, the intermediate (D) is produced through C–C bond cleavage, which was oxidized by the (cat. 1*) to form the desired product (38a, Scheme 15B).
Meanwhile, the Mo group developed an electrochemical reaction to regulate the migration of 1,2-and 1,4-functional groups in β-hydroxycarboxylic acid (Scheme 16A).55 The synthetic strategy controls the anodic oxidation of carboxylic acid by electrochemistry through a one-electron or two-electron pathway, resulting in 1,4-aryl transfer or semipinacol-type 1,2-group transfer products with excellent chemical selectivity. At the same time, this synthetic strategy can synthesize a series of β-keto acid esters (40a–d) and ketones (41a–d) with excellent yields. A potential mechanism is proposed for 1,4-migration (Scheme 16B-1). The deprotonated substrate (i.e., the carboxylate, Eox = 0.77 V vs. Fc+/0 in MeCN/H2O) undergoes single-electron oxidation to form O-center radical species (A). This is followed by intramolecular radical cyclization, resulting in radical species (B). At the same time, there is an alternative pathway (path b) where an electron-rich arene is first oxidized, and then an intramolecular nucleophilic carboxylate attacks it to create an intermediate (B). Finally, the intermediate (B) is further oxidized and deprotonated to obtain the target product (40a). The study of the 1,2-migration mechanism shows that the substrate (39a) is oxidized at the anode to remove a molecule of CO2 to obtain the intermediate (E). Subsequently, the intermediate (E) was further oxidized and loses hydrogen ions to produce the target product (41a, Scheme 16B-2).
 | ||
| Scheme 16 Electrochemically promoted migration of 1,2- and 1,4-functional groups of β-hydroxycarboxylic acids. | ||
Phosphorothioates containing C–S–P (O) bonds are widely found in agrochemicals and active biomolecules.56 In recent years, reagents containing phosphorus sulfide have been developed and effectively utilized in phosphorothiolation reactions.57 Additionally, elemental sulfur (S8) is a cost-effective, readily available, and abundant resource, making it an appealing option for creating C–S–P (O) bonds.58 In 2023, the Cao group realized the electrochemical regioselective C–H phosphorothioation reaction of indolizines by using elemental sulfur (S8) as the sulfur source, leading to the synthesis of various mercapto-phosphono-substituted indolizine derivatives through intramolecular S- to C-[1,4] phosphoryl migration (Scheme 17A).59 The migration products (45) can be synthesized efficiently and conveniently via electrolysis at a constant current of 10 mA, accommodating a wide range of substrates, with moderate to good yields for various substituted indolizines (45a–45c) and H-phosphonates (45d).
A potential mechanism was proposed for the formation of phosphorothiolation products (44). Initially, Br2 is produced by anodic oxidation. Then, Br2 reacts with O,O-diethyl S-hydrogen phosphorothioate (A) to form intermediate (B). Next, indolizine (42a) reacts with intermediate (B) to obtain intermediate (C) which then deprotonates to form the phosphorothioate product (44a). For the formation of the rearrangement product (45a), the compound (44a) undergoes single-electron oxidation to provide a radical cation (D), which breaks the S–P bond to produce (EtO)2P(O)+ and a sulfur-centered radical (E). Subsequently, the free radical (E) is isomerized to form a free radical intermediate (F). Following this, the intermediate (F) captures (EtO)2P(O)+ to generate free radical cation (G). Finally, the radical cation (G) is spontaneously reduced by another molecule (44a), or the charge may be passed from the cation radical back to the electrode (backward electron transfer). Finally, the rearrangement product (45a) is formed (Scheme 17B).
The β-arylethylamine structure is widely found in some endogenous neurotransmitters and several drugs used for treating central nervous system diseases.60 Among them, the structure can be prepared by radical desulfonylative rearrangement of arylsulfonamides, with the generation of a β-amino radical being crucial for these reactions.61 In recent years, numerous methods for producing β-amino radicals under mild conditions have been developed.62 Although these synthetic strategies have made great achievements, there is still a great need for simpler, milder, atomic-economical, and wider methods. Recently, Claraz et al. reported an electrochemically promoted radical fluoromethylation of N-allylbenzamides (46), leading to the synthesis of various functionalized β-arylethylamine derivatives (48) through 1,4-aryl migration (Scheme 18A).63 This reaction can access valuable tri- and di-fluorocontaining arylethylamine derivatives (48a and 48b) and realize 1,4-migration for substrates containing strong electron-withdrawing groups (48c) or electron-rich groups (48d).
A potential reaction mechanism was proposed by density functional theory (DFT) calculations (Scheme 18B). Initially, the substrate (47a, Eox = 0.73 V vs. Fc+/0 in DCE/MeOH) is oxidized at the anode and SO2 is removed to obtain CF3˙ radical (A). Then, the substrate (46a) and the radical (A) undergo radical addition to obtain an intermediate (B) which undergoes intramolecular radical cyclization to produce a spiro radical intermediate (C). Next, selective C–C bond cleavage occurs to obtain carbamoyl radical (D). Subsequently, the intermediate (D) is oxidized at the anode to produce the corresponding carbamoyl cation (E), which undergoes nucleophilic addition of methanol to produce carbamoyl cation (F). Finally, the target product (48a) is obtained by deprotonation.
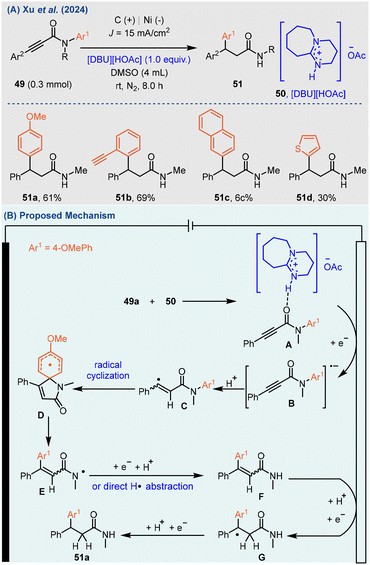
Very recently, the Xu group used [DBU][HOAc] as a hydrogen bonding donor to achieve 1,4-aryl migration of N-arylpropiolamides by an electrochemical reduction strategy, resulting in the synthesis of various diarylpropanamide compounds (Scheme 19A).64 The reductive activation of chemical bonds at lower negative potentials allows for high tolerance and selectivity towards different functional groups. Various substituted aromatic hydrocarbons (51a–c) and thiophene (51d) can also realize 1,4-migration. It is worth noting that [DBU][HOAc] is used as both a hydrogen bond donor and a supporting electrolyte for the first time.
The mechanism investigations revealed that the substrate (49a) interacts with [DBU][HOAc] (50) to form a complex (A, Ered = −2.3 V vs. Ag/AgNO3 in MeCN) via hydrogen bonding. This complex is then reduced at the cathode to yield an intermediate (B). Following this, intermediate (B) undergoes protonation to generate vinyl radical (C), which subsequently undergoes intramolecular radical cyclization to form a spiro intermediate (D). Then, the (D) is selectively cleaved by the C–N bond to produce amide radical intermediate (E). The (E) is abstracted by a hydrogen atom or reduced by SET and then protonated to produce intermediate (F). Next, intermediate (F) is reduced at the cathode and deprotonated to obtain (G). Finally, (G) obtained the target product (51a) through a radical-polar crossover (RPC) (Scheme 19B).
2.3 1,5-Functional group migrations
In 2019, the Guo group discovered that amidyl radicals could be produced through the electrochemical reduction of N–O bonds. Following this, they synthesized a variety of amide derivatives (53) via intramolecular 1,5-aryl migration (Scheme 20A).65 This synthetic strategy is suitable for the post-modification of hydroxylamine derivatives (53a and 53b) with different substituents and bioactive molecules (53c). A potential mechanism was proposed (Scheme 20B). Initially, the substrate (52a, Ered = −1.52 V vs. SCE in DMSO) is reduced at the cathode to produce an intermediate (A). Then, the N–O bond is cleaved to generate amidyl radical (B). Next, intramolecular free radical Smiles rearrangement occurs to form radical intermediate (C). Finally, the cathodic reduction and protonation of (C) generates the target product (53a).Aminophenol exists widely in the molecular structures of many compounds.66 Nowadays, the synthetic methods for polysubstituted aminophenol are mainly realized by metal catalysis and ligands.67 Thus, it is crucial to develop more environmentally friendly and cost-effective synthesis methods. In 2023, the He group developed an electrochemical organoselenium-catalyzed 1,5-amide migration of N-aryloxyamides to synthesize a series of aminophenol derivatives (56a and 56b) (Scheme 21A).68 Meanwhile, when the para-substituent on the benzene ring of the substrate (54) is not hydrogen, the obtained product is an aminoketone derivative (57a and 57b).
The mechanism studies showed that the catalyst (55, Eox = 1.54 V and 1.91 V vs. Ag/AgCl in MeCN) is oxidized and activated at the anode to form selenium cation (A) and phenyl selenium radical (A). Meanwhile, radical (B) can also produce selenium cation (A) through single-electron transfer at the anode. Following this, intermediate (A) combines with the bromine anion to form PhSeBr and reacts with the substrate (54) to form Se–N intermediate (C). Intermediate (C) undergoes two [2,3] sigmatropic rearrangements to obtain para-aminated intermediate (F, R2 = H) or product (57) (R2 ≠ H). Finally, the intermediate (F) removes cation (A) under the action of HBr to complete the cycle and afford the final target product (56a, Scheme 21B).
3. Electrochemical ring expansion reactions
The synthesis of medium-sized rings is difficult due to the unfavorable enthalpy and entropy barriers present in their transition states.69 The common strategy to achieve this synthetic goal is ring expansion from more easily available five-membered or six-membered ring compounds.70 In 2014, Peters and colleagues demonstrated an electrochemical direct reduction ring extension reaction using a silver cathode, successfully synthesizing six- to seven-membered ring compounds (59, Scheme 22A).71 First, the substrate (58a, Ered = −0.6 V and −1.51 V vs. SCE in DMF) is reduced at the cathode to generate a radical intermediate (A), which then quickly generates a cyclopropyl alkoxy radical (B). This radical (B) subsequently undergoes a ring-expansion reaction to form an intermediate (C), which ultimately abstracts a hydrogen atom from the solvent DMF to yield the desired product (59a, Scheme 22B).Later, Christoffers et al. reported the electrochemical reduction and ring extension reaction of cyclic α-(ortho-iodophenyl)-β-oxoesters to synthesize a series of benzannulated cycloalkanone carboxylic esters (61, Scheme 23A).72 The reaction exhibits good substrate tolerance, allowing for the synthesis of benzocycloheptanone (61a and 61b), nonanone (61c), and decanone (61d) in moderate to good yields. The mechanism studies showed that the substrate (60a) is reduced at the cathode to obtain phenyl anion derivative (A). Subsequently, the anion center attacks the carbonyl group (activated by TMSCl) to produce a tricyclic intermediate (B) with a cis-configuration. Finally, the target product (61a) is obtained by hydrolysis (Scheme 23B).
A structure containing an all-carbon quaternary stereocenter is widely found in many bioactive natural products and pharmaceuticals.73 Among these, the semipinacol rearrangement of allylic alcohols has emerged as a popular method for creating carbonyl compounds with α-quaternary carbon centers.74 Traditional synthesis methods often rely on chemical oxidants or metal reagents, which significantly restrict their broader application.75 Therefore, developing a more greener and efficient synthesis strategy is necessary. In 2019, the Zhang group used cheap and stable RSO2Na (R = CF3, Ph) as a free radical precursor to realize electrochemical phenol rearrangement of allyl alcohol (Scheme 24A).76 This strategy does not need oxidants and metal reagents, and the conditions are mild. It can synthesize various β-trifluoromethyl (64a and 64b) and sulfonated ketones (64c and 64d) with moderate to excellent yields.
A potential mechanism was proposed (Scheme 24B). Initially, CF3SO2Na undergoes oxidation via SET at the anode, resulting in the formation of a sulfonyl radical. This is followed by a rapid release of SO2, leading to the creation of a CF3 radical (B). Next, radical addition occurs with the substrate (62a) to obtain radical (C), which is further oxidized to cation (D). Finally, the target product (64a) is obtained through the deprotonation process of ring expansion.
Around the same period, Kim et al. also reported the electrochemical trifluoromethylation/ring extension reaction of alkenyl alcohols to synthesize a series of β-CF3 substituted ketones (Scheme 25A).77 Then, Kim and co-workers continued to report the electrochemical oxidation of alkenylcyclobutanols, radical selenylation/ring expansion reactions (Scheme 25B),78 along with radical arylsulfonylation and ring expansion reactions (Scheme 25C).79
In 2019, the Zhang group used inorganic halide salts as sources of halogen to synthesize a series of β-halocarbonyl compounds (70) with an all-carbon α-quaternary center through the electrochemical halogenation/ring expansion reaction of allyl alcohol (Scheme 26A).80 This reaction features mild conditions and a wide range of substrates, which can not only synthesize five-membered (70a and 70d) and six-membered (70b) β-halocarbonyl compounds but also realize the construction of spirocyclic skeletons (70c and 70e). The mechanism study shows that halide anions are first oxidized to halogen molecules at the anode. Subsequently, halogen molecules react with the double bond of the substrate (68a) to generate an intermediate (A), which then undergoes a semipinacol rearrangement to yield the desired product (70a, Scheme 26B).
 | ||
| Scheme 26 Approaches for synthesising β-halocarbonyls via electrochemical rearrangement of allylic alcohols. | ||
Medium-sized nitrogen-containing heterocycles, specifically those with 8 to 11-membered rings, are commonly found in bioactive compounds and natural products.81 However, this kind of molecular skeleton is mainly synthesized by intramolecular carbonylation,82 closed-ring metathesis (RCM)83, Claisen rearrangement,84etc.85 Therefore, these synthetic strategies require the use of metal catalysts or stoichiometric oxidants, which greatly limits their application scope. In 2020, the Ruan group designed an electrochemical strategy to synthesize annulated medium-sized lactams (72) by promoting the cleavage of C–C bonds through N radicals (Scheme 27A).86 The synthetic strategy features mild conditions, and 8–11 annulated lactams (70a–d) can be synthesized with moderate to good yields without metal reagents and chemical oxidants.
A potential mechanism was proposed by DFT calculations (Scheme 27B). Initially, the N–H bond in the substrate (71a, Eox = 2.2 V vs. Ag/AgCl in MeCN) is oxidized at the anode to produce an amido radical intermediate (A), which undergoes intramolecular cyclization to form a radical intermediate (B). Next, the selective C–C bond of intermediate (B) is cleaved to generate a neutral radical (C). Finally, (C) undergoes single electron oxidation and deprotonation to produce the target product (72b). Additionally, the Lei group realized the electrochemical ring-expanding reaction between molecules by using benzocyclic ketone and aniline.87
The benzoxazinone skeleton exists widely in some drug molecules and bioactive compounds.88 Currently, most of these compounds’ synthesis methods require the use of strong alkali or stoichiometric organometallic reagents, and the steps are cumbersome and the atom economy is poor.89 Therefore, developing more greener, efficient, and sustainable synthetic methods under mild conditions is particularly important. In 2022, Maulide et al. reported the electrochemical rearrangement of 3-hydroxyoxindoles to synthesize benzoxazinone derivatives (Scheme 28A).90 This reaction demonstrates broad tolerance for different functional groups, allowing various nucleophiles (74a and 74b) and alkyl (74c and 74d) substitutions to participate.
The mechanism studies showed that the substrate (73a) is oxidized at the anode to form the peroxide intermediate (A), which can be rearranged to form the intermediate (B) (Scheme 28C) in two possible ways. The ring-expanding reaction is carried out by a Baeyer–Villiger type rearrangement or an intermediate (B) which can be produced by an oxa-Dowd–Beckwith-type rearrangement formed in the molecule on the epoxide. Subsequently, the intermediate (B) was further oxidized to obtain a cationic intermediate (C), which was further captured by methanol to obtain the target product (74a, Scheme 28B).
4. Electrochemical migratory cyclization reactions
Electrochemically promoted radical cyclization has been developed rapidly in recent decades and provided a powerful tool for the synthesis of nitrogen-containing heterocyclic compounds.9c,10a,91 Among them, the migratory cyclization reaction can quickly realize the recombination of the molecular skeleton to obtain a brand-new cyclic compound. However, there are few studies on electrochemically promoted migratory cyclization reactions, because the occurrence of ortho-cyclization reactions often accompanies the occurrence of migratory cyclization reactions, so it is difficult to control its selectivity.In 2019, the Ye group reported for the first time that the [4 + 2] annulation-rearrangement-aromatization of styrene was electrochemically promoted to synthesize functional naphthalene derivatives (Scheme 29A).92 This reaction is notable for not requiring a metal catalyst or oxidant, demonstrating excellent atom economy. Additionally, it shows strong substrate tolerance, allowing various substituted styrenes (76a–d) to afford the desired product with moderate to good yields and chemical selectivity.
The proposed mechanism is that TBPA (Eox = 1.15 V vs. SCE in MeCN) is first oxidized at the anode to form the radical cation TBPA (TBPAC˙+). Following this, the electron-rich styrene (75a) is oxidized by SET to obtain free radical cation (A) and TBPA. Substrate (75a) attacks cation (A) to form intermediate (B), which undergoes intramolecular radical cyclization and deprotonation to obtain (D). Subsequently, (D) loses electrons and protons to obtain intermediate (E), which is further oxidized and protonated to obtain carbon radical (F). Intermediate (F) is oxidized to (G). Finally, the secondary carbon cation is rearranged into a more stable tertiary carbon cation (H) and deprotonated to obtain the migratory cyclization product (76a, Scheme 29B).
Triazolopyridinone derivatives are widely used in medicine, materials, and agricultural chemicals.93 However, there are no reports on sterically hindered substituted triazolopyridine derivatives. In 2020, the Zhang group reported an electrochemical rearrangement for effective synthesis of triazolopyridine from various alkyl carboxylic acids, which are difficult to obtain by traditional methods (Scheme 30A).94 This synthetic strategy features simple and mild conditions and a wide range of substrates. It can not only synthesize various conventional functionalized triazolopyridines (78a and 78b) but also proves the potential application value of this synthetic strategy for the late-stage modification of bioactive molecules (78c) and amino acid derivatives (78d).
The proposed mechanism is that triarylamine (NAr3) is first oxidized at the anode to form the radical cation NAr3 (NAr3˙+). Next, the substrate (77) undergoes SET oxidation to generate the radical cation (A) and NAr3. Following this, the intermediate (A) is deprotonated to produce a trans-diazo compound, which can be easily converted into a cis-diazo compound (C). Finally, an intramolecular nucleophilic attack of the carbonyl group by the pyridine nitrogen followed by a concerted 1,2-alkyl migration from carbon to nitrogen affords the rearranged product (78) via intermediate (D) (Scheme 30B).
In 2021, the Guo group developed the electrochemical synthesis of cinnoline derivatives (81) by using ortho-alkynyl acetophenones and sulfonyl hydrazides as starting materials through organic catalytic radical cyclization and migration (Scheme 31A).95 The method features mild conditions, excellent regioselectivity, and wide functional group tolerance, and can obtain various substituted cinnoline derivatives (81a–d) with moderate to excellent yields.
The mechanism studies showed that (Cat 1Eox = 2.32 V vs. Ag/AgCl in MeCN/TFE/H2O) is first oxidized to free radical cation intermediate (Cat 1˙+) at the anode. Meanwhile, the intermediate (A) generates anion (B) under the action of a base, which is converted into radical (C) by the intermediate (Cat 1˙+). This is followed by (C) intramolecular radical cyclization and Smiles rearrangement to obtain intermediate (E). The alkyl radical intermediates (F) and (E) are formed by resonance. Subsequently, the radical coupling of (F) and O2 generates the corresponding tetraoxide intermediate (G), and the continuous O–O and C–N bond homolysis furnishes diazo radical (H). Then (H) generates aryl radical (I) by intramolecular cyclization, which is oxidized by (Cat 1˙+) to form cationic intermediate (J). Finally, the target product (81a) is obtained by deprotonation (Scheme 31B).
The core skeleton of diazoxide compounds is nitrogen-containing six-membered heterocyclic compounds containing sulfanilamide groups. These compounds are widely found in nature and used in clinical research because of their good biological activities.96 As a bioisostere, benzoxathiazine dioxide has potential hypoglycemic and fungicidal effects.97 However, currently, only a few synthetic methods generally have some disadvantages, such as the need for the preparation of starting materials in advance, complex synthetic routes, limited substrate range, low yield, and many by-products, which do not conform to the concept of green chemistry.98 Therefore, developing an efficient, novel, green synthesis method to construct these compounds conveniently is crucial.
In 2022, Ye and co-workers used simple and easily available N-acylsulfonamides to efficiently synthesize various benzoxathiazine dioxides (83) through an electrochemical oxidation migratory cyclization strategy (Scheme 32A).99 The reaction avoids SO2 removal under mild electrochemical conditions, reduces environmental pollution, and improves atom economy. This method has a wide range of substrates, good functional group tolerance, and good compatibility with heterocyclic quinoline (83b). It is worth noting that this strategy allows for the late modification of pharmaceutically active molecules (83c and 83d). The preliminary activity test results indicate that benzoxathiazine dioxide shows potential anti-tumor activity.
The possible mechanism was proposed based on experimental results and DFT calculations (Scheme 32B). The N–H bond in the substrate (82a, Eox = 2.40 V vs. Fc+/0 in MeCN) first forms intramolecular hydrogen bonds with acetate. Then, the radical (B) is generated at the anode through proton-coupled electron transfer (PCET), which can be rapidly isomerized to form O radical species (C). Subsequently, spirocyclic cyclohexadienyl radical intermediate (D) was generated by intramolecular radical cyclization. The reaction then proceeds through two possible reaction pathways. Path a involves radical migration (D to E) and subsequent removal of a proton to form (F), which is further oxidized to obtain the target product (83a). Radical (D) may generate the corresponding carbocation (G) through single electron oxidation in path b. After cationic migration (G to H) and deprotonation, the migratory cyclization product (83a) is obtained.
In the same year, the Zhang group reported the synthesis of benzothiophene derivatives (86) via a migratory cyclization reaction between sulfonyl hydrazides and alkynes under electrochemical conditions (Scheme 33A).100 The experimental results indicate that the reaction proceeds through an intermediate quaternary spirocyclisation, ultimately leading to the migratory cyclization process. Computational research has revealed the selectivity and compatibility of drug molecules and demonstrates the potential applications of the protocols. The reaction has good substrate tolerance, and the target product can be obtained in moderate yields for various sulfonyl hydrazides (86a and 86c) and alkynes (86b and 86d).
The potential mechanism was proposed (Scheme 33B). Initially, sulfonyl hydrazides (84a, Eox = 1.80 V and 2.48 V vs. Ag/AgCl in MeCN) are deprotonated, oxidized at the anode, and released with nitrogen to generate sulfonyl radical (B). Then (B) reacts with (85a, Eox = 2.67 V vs. Ag/AgCl in MeCN) to generate alkenyl radical (C), which is further oxidized by the anode to generate alkenyl cation intermediate (D). (D) undergoes intramolecular cyclization to form a quaternary spirocyclization species (E). Then, ring extension generation involving 1,2-S-migration occurs rapidly to form (F). Finally, further oxidation and deprotonation lead to the formation of the product (86a).
The free radical is a typical highly active species, but it can still form different products through selective transformation.101 The key to achieving high selectivity in free radical reactions lies in effectively controlling the competitive reaction. Recently, Ye and co-workers reported the synthesis of a series of sultam-fused pyridinone derivatives (88 and 89) via electrochemical selective migratory cyclization and ortho-cascade cyclization of 2-alkynylbenzenesulfonamides (Scheme 34A).102 It is found that the incorporation of an extra 2-methyl substituent leads to the selective migration of the acyl group of the key spirocyclic cation intermediate, thus promoting a cascade migration cyclization process.
The potential mechanism was proposed based on DFT calculations (Scheme 34B). Firstly, the N–H bond in the substrate (87c) preferentially combines with acetate to form intramolecular hydrogen bonds, and then the nitrogen radical (B) is generated at the anode by PCET. Subsequently, intramolecular cyclization occurs rapidly to obtain carbon radical (C). Then, there are two possible reaction pathways. In the first reaction pathway (path a), the intermediate (C) attacks the ortho-position of the benzene ring to undergo radical cyclization, which is further oxidized at the anode and then dehydrogenates to obtain the cyclization product (88c). Another reaction pathway (path b) is that the intermediate (C) attacks the carbon atom (C1) to obtain the spiro radical intermediate (E), which is further oxidized into a cationic species (E′). Subsequently, the cationic species (E′) undergoes C1–C2 bond cleavage and recombination to obtain the intermediate (F) which then dehydrogenates to obtain the product of acyl migration (89c). If the C1–C3 bond of the cationic species (E′) cleaves, the intermediate (G) is obtained. Finally, the cyclization product is obtained by a deprotonation process (88c).
5. Miscellaneous rearrangements
The rearrangement of O-aryl thiocarbamate to S-aryl thiocarbamate at high temperatures is commonly called the Newman–Kwart rearrangement (NKR).103 The conventional approach necessitates the utilization of elevated temperatures (>200 °C). In 2018, Francke et al. reported that electrolytic O-aryl thiocarbamate can be rearranged to obtain S-aryl thiocarbamate under constant current conditions (Scheme 35A).104 A variety of S-aryl thiocarbamate derivatives (91a–c) can be obtained at room temperature with moderate to excellent yields. Meanwhile, continuous-flow electrochemical synthesis can achieve almost quantitative yields without using electrolytes, which proves the potential application value of this synthesis strategy. The mechanism study shows that the substrate (90a) is oxidized to radical cation (A) at the anode. Then (A) undergoes intramolecular cyclization and selective cleavage of the C–O bond occurs to form intermediate (C). Ultimately, the desired product (91a) is achieved through a reduction process, which may occur via reverse electron transfer (BET) or a radical chain mechanism (91a, Scheme 35B).The formation of oxidized C–N bonds by C–H/C–C bond cleavage has aroused great interest from scientists, and this synthetic strategy has been widely used in the synthesis of pharmaceutical intermediates and materials.105 In 2019, Jiao and co-workers reported the synthesis of aniline derivatives (94a–d) by electrochemical oxidative cleavage of C–C bonds of alkylarenes (Scheme 36A).106 This strategy uses a cheap and durable graphite plate as the electrode and can be carried out without using any external catalyst or oxidant. The protocol is green, sustainable, efficient, and easy to operate.
The mechanism studies showed that the substrate (92a, Eox = 2.05 V and 2.39 V vs. Ag/AgNO3 in MeCN) is oxidized at the anode, thus generating free radical cation (A). Subsequently, benzyl radical (B) is generated by deprotonation, which is further oxidized to form intermediate cation (C). Then, nucleophilic organic azides (93) attack cation (C) to produce (D). Next, intermediate (E) is formed by a Schmidt-type rearrangement of intermediate (D), and dinitrogen gas is released as the driving force. Finally, isomerization and hydrolysis produce alkylanilines (94a) and benzaldehydes (Scheme 36B).
Heteroarylethylamine compounds exhibit good biological activity and have been widely used in medicine and agricultural chemicals.107 In 2022, Zhu and co-workers reported the synthesis of various β-heteroaryl-γ-trifluoromethylamine derivatives by electrochemically promoting the heteroaryltrifluoromethylation reaction of allylamine (97, Scheme 37A).108 The synthetic strategy demonstrates good substrate tolerance and enables the migration of various heteroaryl groups (97a–c) as well as the late-stage modification of bioactive compounds (97d).
The mechanism studies showed that CF3SO2Na (Eox = 1.37 V vs. Ag/AgCl in MeCN) is initially oxidized at the anode, releasing SO2 to obtain CF3 radical (A). Subsequently, the radical (A) is added to the double bond of the substrate (95a) to form the radical (B). Through intramolecular radical cyclization, the spirocyclic intermediate (C) is obtained. Next, the intermediate (C) releases SO2 through a Smiles rearrangement to generate nitrogen-free radical (D). Finally, the radical intermediate (D) is reduced by SO2 in the aqueous solution to obtain the product (97a, Scheme 36C). Recently, the Pan group reported the electrochemical synthesis of β-difluoromethylamide compounds (100) from N-benzenesulfonylacrylamide and difluorine reagents (Scheme 37B).109
The spirooxindole skeleton exists widely in many natural products and biologically active molecules.110 Most of the traditional syntheses of this molecular skeleton require the oxidative rearrangement of indole using chemical oxidants.111 However, these chemical oxidants are dangerous and toxic and will produce stoichiometric harmful chemical waste. In 2022, Tong et al. reported an electrochemical oxidation rearrangement of tetrahydro-β-carbolines in a zero-gap flow cell to synthesize spirooxindole derivatives (Scheme 38A).112 Almost simultaneously, the Xu group also reported the synthesis of spirooxindoles by electrochemical rearrangement in an undivided flow cell (Scheme 38B).113 These synthetic strategies feature a good substrate scope, and various spirooxindole derivatives (902a–d) were obtained with moderate to excellent yields. The mechanistic investigations revealed that LiBr is oxidized to HOBr at the anode. Then, HOBr oxidizes the substrate (101a) to bromo-indoline intermediate (A). After the addition of water, spirobenzindole (102a) is produced through a semi-pinacol rearrangement (Scheme 38C).
The Ferrier rearrangement (FR) is a nucleophilic substitution with an allylic rearrangement, resulting in the conversion of glycal into a 2,3-unsaturated glycosyl derivative.114 This strategy has been widely used in the synthesis of many natural products.115 Recently, Mazzarella et al. reported an electrochemically promoted Ferrier rearrangement to synthesize a variety of unsaturated glycosyl derivatives (Scheme 39A).116 This synthetic strategy exhibits wide substrate compatibility, and various nucleophiles can provide 2,3-unsaturated glycosyl derivatives (105a–c) with high yields and excellent diastereoselectivities. This sustainable method is expected to expand the electrochemical application in sugar chemistry. The mechanism study shows that glucose (103a) generates radical cation (A) through single electron oxidation at the anode. Subsequently, the acetoxy group is lost, and it evolves into an oxonium species (B). The substrate (104a) reacts with the intermediate (B) by deprotonation to form the product (105a, Scheme 39B).
Small cycloalkanes have moderate ring strain and usually require dense functionalization to induce the bias or distal activation of (hetero) aromatic rings via single-electron oxidation for relieving the tension.117 Very recently, Wu and co-workers reported that a variety of oxazoline and oxazine derivatives (107a–108a) were synthesized by electrochemical oxidation directly activating alkyl cyclopropanes/butanes (Scheme 40A).118 This strategy applies to a wide range of substrates. Notably, products derived from cyclobutanes undergo formal ring contraction to cyclopropanes (109a and 109b). This electrochemical synthesis strategy marks significant progress in the skeleton rearrangement reactions driven by the strain release of moderately strained rings and provides a sustainable and efficient synthetic route for the construction of complex heterocyclic compounds.
The mechanism studies showed that cyclopropane and cyclobutane in substrates (106a, Eox = 2.16 V and 2.30 V vs. Ag/AgCl in MeCN) and (106c) are oxidized at the anode to produce radical cation (A). Subsequently, the radicals (B) and (D) are formed by an intramolecular cyclization reaction. The radical (B) is further oxidized at the anode to generate cation (C) and then deprotonated to generate oxazoline (107a). On the other hand, the radical (D) captures oxygen to form peroxy radical (E), forming carbon radical (F) through 1,5-hydrogen atom transfer (HAT). Subsequently, free radical (F) participates in the substitution to form a cyclopropyl ring (109a), and at the same time releases hydrogen peroxide free radical (Scheme 40B).
6. Conclusions
As a green synthesis method, electrochemical synthesis provides powerful support for exploring new reactions. In the past decade, there has been a growing number of reports on electrochemical rearrangements. This review mainly introduces electrochemically promoted functional group migrations, ring expansions, and migratory cyclization reactions. It is worth noting that electrochemical rearrangements can synthesize numerous molecular structures that are challenging to create using traditional methods. In particular, electrochemically promoted migratory cyclization reactions can often get unexpected results.Although this field is still in the stage of vigorous development after a decade of progress, many reactions remain undeveloped, such as the discovery of new functional group migrations, enantioselective electrochemical rearrangements, and rearrangement reactions involving the retention of small molecules. However, we believe that with the rapid development of electrochemistry, this field will be gradually explored. More importantly, it highlights the unique charm of electrochemistry in exploring new reactions and will stimulate the further application of electrochemical synthesis strategies in the pharmaceutical and chemical fields.
Data availability
This is a review article with no new data. All the compounds have been reported before.Conflicts of interest
There are no conflicts to declare.Acknowledgements
This work was supported by the Jiangxi Provincial Key Laboratory of Organic Functional Molecules (No: 2024SSY05141).References
- X. Wu, Z. Ma, T. Feng and C. Zhu, Radical-mediated rearrangements: past, present, and future, Chem. Soc. Rev., 2021, 50, 11577–11613 RSC.
- H. Wu, Q. Wang and J. Zhu, Recent Advances in Catalytic Enantioselective Rearrangement, Eur. J. Org. Chem., 2019, 1964–1980 CrossRef CAS.
- C. M. Rojas, in Molecular Rearrangements in Organic Synthesis, Wiley-VCH, New York, 2015, DOI:10.1002/9781118939901.
- X. Huang, M. Seid and J. W. Keillor, A Mild and Efficient Modified Hofmann Rearrangement, J. Org. Chem., 1997, 62, 7495–7496 CrossRef CAS PubMed.
- (a) W. E. Truce, W. J. Ray, O. L. Norman and D. B. Eickemeyer, Rearrangements of Aryl Sulfones. I. The Metalation and Rearrangement of Mesityl Phenyl Sulfone, J. Am. Chem. Soc., 1958, 80, 3625–3629 CrossRef CAS; (b) L. A. Warren and S. Smiles, CXVII.—iso-β-Naphthol sulphide, J. Chem. Soc., 1930, 956–963 RSC.
- A. H. Blatt, The Beckmann Rearrangement, Chem. Rev., 1933, 12, 215–260 CrossRef CAS.
- (a) B. A. Frontana-Uribe, R. D. Little, J. G. Ibanez, A. Palma and R. Vasquez-Medrano, Organic electrosynthesis: a promising green methodology in organic chemistry, Green Chem., 2010, 12, 2099–2119 RSC; (b) M. Yan, Y. Kawamata and P. S. Baran, Synthetic Organic Electrochemical Methods Since 2000: On the Verge of a Renaissance, Chem. Rev., 2017, 117, 13230–13319 CrossRef CAS PubMed; (c) A. Bhadani and M. Kathiresan, Electrochemical switching in mechanically interlocked molecules (MIMs), Org. Chem. Front., 2024, 11, 2954–2980 RSC; (d) K. Grudzień, A. Zlobin, J. Zadworny, K. Rybicka-Jasińska and B. Sadowski, Modern photo- and electrochemical approaches to aryl radical generation, Org. Chem. Front., 2024, 11, 5232–5277 RSC.
- (a) C. Kingston, M. D. Palkowitz, Y. Takahira, J. C. Vantourout, B. K. Peters, Y. Kawamata and P. S. Baran, A Survival Guide for the “Electro-curious”, Acc. Chem. Res., 2020, 53, 72–83 CrossRef CAS; (b) X.-Q. Xie, W. Zhou, R. Yang, X.-R. Song, M.-J. Luo and Q. Xiao, Electroreduction strategy: a sustainable tool for the generation of aryl radicals, Org. Chem. Front., 2024, 11, 4318–4342 RSC.
- (a) P. R. D. Murray, J. H. Cox, N. D. Chiappini, C. B. Roos, E. A. McLoughlin, B. G. Hejna, S. T. Nguyen, H. H. Ripberger, J. M. Ganley, E. Tsui, N. Y. Shin, B. Koronkiewicz, G. Qiu and R. R. Knowles, Photochemical and Electrochemical Applications of Proton-Coupled Electron Transfer in Organic Synthesis, Chem. Rev., 2022, 122, 2017–2291 CrossRef CAS PubMed; (b) C. Pratley, S. Fenner and J. A. Murphy, Nitrogen-Centered Radicals in Functionalization of sp2 Systems: Generation, Reactivity, and Applications in Synthesis, Chem. Rev., 2022, 122, 8181–8260 CrossRef CAS PubMed; (c) P. Xiong and H.-C. Xu, Chemistry with Electrochemically Generated N-Centered Radicals, Acc. Chem. Res., 2019, 52, 3339–3350 CrossRef CAS.
- (a) M. D. Kärkäs, Electrochemical strategies for C–H functionalization and C–N bond formation, Chem. Soc. Rev., 2018, 47, 5786–5865 RSC; (b) R. Shaw, N. Sihag, H. Bhartiya and M. R. Yadav, Harnessing photocatalytic and electrochemical approaches for C–H bond trifluoromethylation and fluoroalkylation, Org. Chem. Front., 2024, 11, 954–1014 RSC.
- (a) J. Liu, J.-P. Wan and Y. Liu, Electrochemical difunctionalization of alkenes and alkynes for the synthesis of organochalcogens involving C–S/Se bond formation, Org. Chem. Front., 2024, 11, 597–630 RSC; (b) Y. Zheng, W. Lu, C. Chen, Y. Lu and S. Huang, Recent advances in electrochemical difunctionalization of alkenes and alkynes for the synthesis of organohalides, Org. Chem. Front., 2024, 11, 5306–5324 RSC.
- C. Yin, S. Tang, J. Mei, X. Hu and H. Zhang, Electrochemical synthesis and transformation of organoboron compounds, Org. Chem. Front., 2023, 10, 3361–3377 RSC.
- D. Saha, I. M. Taily, R. Kumar and P. Banerjee, Electrochemical rearrangement protocols towards the construction of diverse molecular frameworks, Chem. Commun., 2021, 57, 2464–2478 RSC.
- L. Li, M. Xue, X. Yan, W. Liu, K. Xu and S. Zhang, Electrochemical Hofmann rearrangement mediated by NaBr: practical access to bioactive carbamates, Org. Biomol. Chem., 2018, 16, 4615–4618 RSC.
- B. K. Malviya, C. Bottecchia, K. Stone, D. Lehnherr, F. Lévesque, C. O. Kappe and D. Cantillo, Multigram Electrochemical Hofmann Rearrangement Using a Spinning Three-Dimensional Anode, Org. Process Res. Dev., 2023, 27, 2183–2191 CrossRef CAS.
- (a) N. Fu, G. S. Sauer and S. Lin, A general, electrocatalytic approach to the synthesis of vicinal diamines, Nat. Protoc., 2018, 13, 1725–1743 CrossRef CAS; (b) Z.-L. Li, G.-C. Fang, Q.-S. Gu and X.-Y. Liu, Recent advances in copper-catalysed radical-involved asymmetric 1,2-difunctionalization of alkenes, Chem. Soc. Rev., 2020, 49, 32–48 RSC; (c) X. Wu and C. Zhu, Radical-Mediated Remote Functional Group Migration, Acc. Chem. Res., 2020, 53, 1620–1636 CrossRef CAS.
- Z. Guan, H. Wang, Y. Huang, Y. Wang, S. Wang and A. Lei, Electrochemical Oxidative Aryl(alkyl)trifluoromethylation of Allyl Alcohols via 1,2-Migration, Org. Lett., 2019, 21, 4619–4622 CrossRef CAS PubMed.
- D. Wang, B. Yuan, J. Xu and L. Ackermann, Electrochemical Rearrangement for Remote Functionalizations of Unactivated Alkenes, Chem. – Eur. J., 2023, 29, e202300600 CrossRef CAS PubMed.
- W. Xia, Y. Yang, X. Zhang, L. Hu and Y. Xiong, Electrochemical synthesis of γ-keto sulfones containing a β-quaternary carbon center via 1,2-migration, Green Chem., 2023, 25, 8273–8279 RSC.
- Z. Mao, Y. Zhou, J. Zhang, C. Liu, C.-S. Wang, X. Yang, H. Qin, Z. Fang and K. Guo, Difunctionalization of alkenes proceeding with radical 1,2-alkynyl migration in batch and continuous-flow modes, New J. Chem., 2024, 48, 1735–1740 RSC.
- C. Elger, P. Halász, J. Maia, L. Almeida and P. Soares-da-Silva, Efficacy and safety of eslicarbazepine acetate as adjunctive treatment in adults with refractory partial-onset seizures: A randomized, double-blind, placebo-controlled, parallel-group phase III study, Epilepsia, 2009, 50, 454–463 CrossRef CAS.
- Z.-M. Chen, X.-M. Zhang and Y.-Q. Tu, Radical aryl migration reactions and synthetic applications, Chem. Soc. Rev., 2015, 44, 5220–5245 RSC.
- (a) W. R. Bowman and J. M. D. Storey, Synthesis using aromatic homolytic substitution—recent advances, Chem. Soc. Rev., 2007, 36, 1803–1822 RSC; (b) Z. Cong, T. Miki, O. Urakawa and H. Nishino, Synthesis of Dibenz[b,f]oxepins via Manganese(III)-Based Oxidative 1,2-Radical Rearrangement, J. Org. Chem., 2009, 74, 3978–3981 CrossRef CAS PubMed; (c) W. Kong, M. Casimiro, E. B. Merino and C. Nevado, Copper-Catalyzed One-Pot Trifluoromethylation/Aryl Migration/Desulfonylation and C(sp2)–N Bond Formation of Conjugated Tosyl Amides, J. Am. Chem. Soc., 2013, 135, 14480–14483 CrossRef CAS PubMed; (d) A. Bunescu, Q. Wang and J. Zhu, Copper-Catalyzed Cyanomethylation of Allylic Alcohols with Concomitant 1,2-Aryl Migration: Efficient Synthesis of Functionalized Ketones Containing an α-Quaternary Center, Angew. Chem., Int. Ed., 2015, 54, 3132–3135 CrossRef CAS.
- (a) H. Kurihara, T. Fuchigami and T. Tajima, Kolbe Carbon−Carbon Coupling Electrosynthesis Using Solid-Supported Bases, J. Org. Chem., 2008, 73, 6888–6890 CrossRef CAS; (b) A. Wiebe, T. Gieshoff, S. Möhle, E. Rodrigo, M. Zirbes and S. R. Waldvogel, Electrifying Organic Synthesis, Angew. Chem., Int. Ed., 2018, 57, 5594–5619 CrossRef CAS PubMed.
- F. Bu, L. Lu, X. Hu, S. Wang, H. Zhang and A. Lei, Electrochemical oxidative decarboxylation and 1,2-aryl migration towards the synthesis of 1,2-diaryl ethers, Chem. Sci., 2020, 11, 10000–10004 RSC.
- R. M. de Figueiredo, J.-S. Suppo and J.-M. Campagne, Nonclassical Routes for Amide Bond Formation, Chem. Rev., 2016, 116, 12029–12122 CrossRef CAS PubMed.
- (a) H. J. Kiely-Collins, I. Sechi, P. E. Brennan and M. G. McLaughlin, Mild, calcium catalysed Beckmann rearrangements, Chem. Commun., 2018, 54, 654–657 RSC; (b) M. Arisawa and M. Yamaguchi, Rhodium-Catalyzed Beckmann Rearrangement, Org. Lett., 2001, 3, 311–312 CrossRef CAS.
- X. Mo, T. D. R. Morgan, H. T. Ang and D. G. Hall, Scope and Mechanism of a True Organocatalytic Beckmann Rearrangement with a Boronic Acid/Perfluoropinacol System under Ambient Conditions, J. Am. Chem. Soc., 2018, 140, 5264–5271 CrossRef CAS PubMed.
- V. P. Srivastava, A. K. Yadav and L. D. S. Yadav, The Beckmann Rearrangement Executed by Visible-Light-Driven Generation of Vilsmeier-Haack Reagent, Synlett, 2014, 665–670 CAS.
- L. Tang, Z.-L. Wang, Y.-H. He and Z. Guan, An Electrochemical Beckmann Rearrangement: Traditional Reaction via Modern Radical Mechanism, ChemSusChem, 2020, 13, 4929–4936 CrossRef CAS PubMed.
- (a) D. Wang and S. Gao, Sonogashira coupling in natural product synthesis, Org. Chem. Front., 2014, 1, 556–566 RSC; (b) C. Lamberth, Alkyne chemistry in crop protection, Bioorg. Med. Chem., 2009, 17, 4047–4063 CrossRef CAS PubMed.
- A. Music, C. M. Nuber, Y. Lemke, P. Spieß and D. Didier, Electro-alkynylation: Intramolecular Rearrangement of Trialkynylorganoborates for Chemoselective C(sp2)–C(sp) Bond Formation, Org. Lett., 2021, 23, 4179–4184 CrossRef CAS PubMed.
- (a) D. Y. Curtin and L. L. Miller, The isolation and rearrangement of simple isoimides (iminoanhydrides), Tetrahedron Lett., 1965, 6, 1869–1876 CrossRef; (b) J. S. P. Schwarz, Preparation of acyclic isoimides and their rearrangement rates to imides, J. Org. Chem., 1972, 37, 2906–2908 CrossRef CAS.
- X. Zhang, T. Cui, X. Zhao, P. Liu and P. Sun, Electrochemical Difunctionalization of Alkenes by a Four-Component Reaction Cascade Mumm Rearrangement: Rapid Access to Functionalized Imides, Angew. Chem., Int. Ed., 2020, 59, 3465–3469 CrossRef CAS.
- Q. Chu, Z. Feng, S. Zhang, P. Liu and P. Sun, Three-component reaction for the synthesis of imides enabled by electrochemical C(sp3)–H functionalization, Green Chem., 2023, 25, 6728–6732 RSC.
- P. Jiang, C. Liang, T. He, R. Liu, X. Meng, Y. Zheng and S. Huang, Electrochemical Decarboxylation/Mumm Rearrangement towards Imides, Eur. J. Org. Chem., 2024, e202400469 CrossRef CAS.
- H.-J. Böhm, D. Banner, S. Bendels, M. Kansy, B. Kuhn, K. Müller, U. Obst-Sander and M. Stahl, Fluorine in Medicinal Chemistry, ChemBioChem, 2004, 5, 637–643 CrossRef.
- (a) D. A. Evans, G. L. Carroll and L. K. Truesdale, Synthetic applications of trimethylsilyl cyanide. Efficient synthesis of .beta.-aminomethyl alcohols, J. Org. Chem., 1974, 39, 914–917 CrossRef CAS; (b) G. K. S. Prakash and A. K. Yudin, Perfluoroalkylation with Organosilicon Reagents, Chem. Rev., 1997, 97, 757–786 CrossRef CAS PubMed; (c) G. K. S. Prakash, P. V. Jog, P. T. D. Batamack and G. A. Olah, Taming of Fluoroform: Direct Nucleophilic Trifluoromethylation of Si, B, S, and C Centers, Science, 2012, 338, 1324–1327 CrossRef CAS PubMed.
- (a) X. Liu, C. Xu, M. Wang and Q. Liu, Trifluoromethyltrimethylsilane: Nucleophilic Trifluoromethylation and Beyond, Chem. Rev., 2015, 115, 683–730 CrossRef CAS; (b) C. P. Johnston, T. H. West, R. E. Dooley, M. Reid, A. B. Jones, E. J. King, A. G. Leach and G. C. Lloyd-Jones, Anion-Initiated Trifluoromethylation by TMSCF3: Deconvolution of the Siliconate–Carbanion Dichotomy by Stopped-Flow NMR/IR, J. Am. Chem. Soc., 2018, 140, 11112–11124 CrossRef CAS PubMed.
- H. Yang, Y. Shen, Z. Xiao, C. Liu, K. Yuan and Y. Ding, The direct trifluoromethylsilylation and cyanosilylation of aldehydes via an electrochemically induced intramolecular pathway, Chem. Commun., 2020, 56, 2435–2438 RSC.
- (a) R. R. Karimov and J. F. Hartwig, Transition-Metal-Catalyzed Selective Functionalization of C(sp3)−H Bonds in Natural Products, Angew. Chem., Int. Ed., 2018, 57, 4234–4241 CrossRef CAS; (b) R. Wang, Y. Luan and M. Ye, Transition Metal–Catalyzed Allylic C(sp3)–H Functionalization via η3-Allylmetal Intermediate, Chin. J. Chem., 2019, 37, 720–743 CrossRef CAS; (c) R. Manoharan and M. Jeganmohan, Recent Advancements in Allylic C(sp3)–H Functionalization of Olefins Catalyzed by Rh(III) or Ir(III) Complexes, Eur. J. Org. Chem., 2020, 7304–7319 CrossRef CAS; (d) D. L. Golden, S.-E. Suh and S. S. Stahl, Radical C(sp3)–H functionalization and cross-coupling reactions, Nat. Rev. Chem., 2022, 6, 405–427 CrossRef CAS PubMed.
- (a) T. A. F. Nelson and S. B. Blakey, Intermolecular Allylic C−H Etherification of Internal Olefins, Angew. Chem., Int. Ed., 2018, 57, 14911–14915 CrossRef CAS; (b) C. Huang, R.-N. Ci, J. Qiao, X.-Z. Wang, K. Feng, B. Chen, C.-H. Tung and L.-Z. Wu, Direct Allylic C(sp3)−H and Vinylic C(sp2)−H Thiolation with Hydrogen Evolution by Quantum Dots and Visible Light, Angew. Chem., Int. Ed., 2021, 60, 11779–11783 CrossRef CAS PubMed.
- (a) Y. Terada, H. Masuda and T. Watanabe, Structure–Activity Relationship Study on Isothiocyanates: Comparison of TRPA1-Activating Ability between Allyl Isothiocyanate and Specific Flavor Components of Wasabi, Horseradish, and White Mustard, J. Nat. Prod., 2015, 78, 1937–1941 CrossRef CAS PubMed; (b) L. Romeo, R. Iori, P. Rollin, P. Bramanti and E. Mazzon, Isothiocyanates: An Overview of Their Antimicrobial Activity against Human Infections, Molecules, 2018, 23, 624 CrossRef.
- (a) Z. Fu, W. Yuan, N. Chen, Z. Yang and J. Xu, Na2S2O8-mediated efficient synthesis of isothiocyanates from primary amines in water, Green Chem., 2018, 20, 4484–4491 RSC; (b) C. Kiaku, J. M. Walsh, M. C. Leech, D. L. Poole, J. Mason, I. C. A. Goodall, P. Devo and K. Lam, Electrochemical Isothiocyanation of Primary Amines, Org. Lett., 2023, 25, 1147–1150 CrossRef CAS.
- X. Gao, H. He, K. Miao, L. Zhang, S.-F. Ni, M. Li and W. Guo, Electrochemical Allylic C(sp3)–H Isothiocyanation via [3,3]-Sigmatropic Rearrangement, Org. Lett., 2024, 26, 4554–4559 CrossRef CAS PubMed.
- (a) W. Li, W. Xu, J. Xie, S. Yu and C. Zhu, Distal radical migration strategy: an emerging synthetic means, Chem. Soc. Rev., 2018, 47, 654–667 RSC; (b) X. Wu, S. Wu and C. Zhu, Radical-mediated difunctionalization of unactivated alkenes through distal migration of functional groups, Tetrahedron Lett., 2018, 59, 1328–1336 CrossRef CAS.
- (a) W. Thaharn, D. Soorukram, C. Kuhakarn, P. Tuchinda, V. Reutrakul and M. Pohmakotr, Radical Cyclization/ipso-1,4-Aryl Migration Cascade: Asymmetric Synthesis of 3,3-Difluoro-2-propanoylbicyclo[3.3.0]octanes, Angew. Chem., Int. Ed., 2014, 53, 2212–2215 CrossRef CAS; (b) Y. Li, B. Liu, H.-B. Li, Q. Wang and J.-H. Li, Oxidative radical 1,2-alkylarylation of alkenes with α-C(sp3)–H bonds of acetonitriles involving 1,2-aryl migration, Chem. Commun., 2015, 51, 1024–1026 RSC; (c) Y. Zeng, C. Ni and J. Hu, Recent Advances in the One-Step Synthesis of Distally Fluorinated Ketones, Chem. – Eur. J., 2016, 22, 3210–3223 CrossRef CAS PubMed.
- (a) Y. Xu, Z. Wu, J. Jiang, Z. Ke and C. Zhu, Merging Distal Alkynyl Migration and Photoredox Catalysis for Radical Trifluoromethylative Alkynylation of Unactivated Olefins, Angew. Chem., Int. Ed., 2017, 56, 4545–4548 CrossRef CAS; (b) X. Tang and A. Studer, Alkene 1,2-Difunctionalization by Radical Alkenyl Migration, Angew. Chem., Int. Ed., 2018, 57, 814–817 CrossRef CAS.
- Y. Gao, H. Mei, J. Han and Y. Pan, Electrochemical Alkynyl/Alkenyl Migration for the Radical Difunctionalization of Alkenes, Chem. – Eur. J., 2018, 24, 17205–17209 CrossRef CAS.
- M.-W. Zheng, X. Yuan, Y.-S. Cui, J.-K. Qiu, G. Li and K. Guo, Electrochemical Sulfonylation/Heteroarylation of Alkenes via Distal Heteroaryl ipso-Migration, Org. Lett., 2018, 20, 7784–7789 CrossRef CAS.
- Z. Zou, W. Zhang, Y. Wang, L. Kong, G. Karotsis, Y. Wang and Y. Pan, Electrochemically Promoted Fluoroalkylation–Distal Functionalization of Unactivated Alkenes, Org. Lett., 2019, 21, 1857–1862 CrossRef CAS.
- A. C. Seastram, M. D. Hareram, T. M. B. Knight and L. C. Morrill, Electrochemical alkene azidocyanation via 1,4-nitrile migration, Chem. Commun., 2022, 58, 8658–8661 RSC.
- C. Zhang, M. Yang, Y. Qiu, M. Song, H. Wang, M. Yang, W. Xie, J. Wu and S. Ye, Alkoxysulfonyl radical species: acquisition and transformation towards sulfonate esters through electrochemistry, Chem. Sci., 2022, 13, 11785–11791 RSC.
- C. Liu, Q. Jiang, Y. Lin, Z. Fang and K. Guo, C- to N-Center Remote Heteroaryl Migration via Electrochemical Initiation of N Radical by Organic Catalyst, Org. Lett., 2020, 22, 795–799 CrossRef CAS PubMed.
- Z. Zhang, L. Zhang, X. Zhang, J. Yang, Y. Yin, Y. Jiang, C. Zeng, G. Lu, Y. Yang and F. Mo, Anodic oxidation triggered divergent 1,2- and 1,4-group transfer reactions of β-hydroxycarboxylic acids enabled by electrochemical regulation, Chem. Sci., 2020, 11, 12021–12028 RSC.
- (a) S. Cogoi, V. Rapozzi, F. Quadrifoglio and L. Xodo, Anti-gene Effect in Live Cells of AG Motif Triplex-Forming Oligonucleotides Containing an Increasing Number of Phosphorothioate Linkages, Biochemistry, 2001, 40, 1135–1143 CrossRef CAS PubMed; (b) T. Ozturk, E. Ertas and O. Mert, A Berzelius Reagent, Phosphorus Decasulfide (P4S10), in Organic Syntheses, Chem. Rev., 2010, 110, 3419–3478 CrossRef CAS PubMed; (c) N.-S. Li, J. K. Frederiksen and J. A. Piccirilli, Synthesis, Properties, and Applications of Oligonucleotides Containing an RNA Dinucleotide Phosphorothiolate Linkage, Acc. Chem. Res., 2011, 44, 1257–1269 CrossRef CAS PubMed; (d) T. S. Kumar, T. Yang, S. Mishra, C. Cronin, S. Chakraborty, J.-B. Shen, B. T. Liang and K. A. Jacobson, 5′-Phosphate and 5′-Phosphonate Ester Derivatives of (N)-Methanocarba Adenosine with in Vivo Cardioprotective Activity, J. Med. Chem., 2013, 56, 902–914 CrossRef CAS PubMed.
- (a) X.-Y. Chen, M. Pu, H.-G. Cheng, T. Sperger and F. Schoenebeck, Arylation of Axially Chiral Phosphorothioate Salts by Dinuclear PdI Catalysis, Angew. Chem., Int. Ed., 2019, 58, 11395–11399 CrossRef CAS; (b) Y. Guo, Y. Luo, S. Mu, J. Xu and Q. Song, Photoinduced Decarboxylative Phosphorothiolation of N-Hydroxyphthalimide Esters, Org. Lett., 2021, 23, 6729–6734 CrossRef CAS PubMed; (c) Z. Zheng, S. Shi, Q. Ma, Y. Yang, Y. Liu, G. Tang and Y. Zhao, Synthesis of δ-phosphorothiolated alcohols by photoredox/copper catalyzed remote C(sp3)–H phosphorothiolation of N-alkoxypyridinium salts, Org. Chem. Front., 2021, 8, 6845–6850 RSC.
- (a) J. Xu, X. Yu and Q. Song, Silver-Catalyzed Radical-Involved Cascade Cyclization of Diphenylphosphine with Cinnamamides: Access to 2-Phosphinoyl-3H-pyrrolo[1,2-a]indoles, Org. Lett., 2017, 19, 980–983 CrossRef CAS PubMed; (b) P. Zhang, G. Yu, W. Li, Z. Shu, L. Wang, Z. Li and X. Gao, Copper-Catalyzed Multicomponent Trifluoromethylphosphorothiolation of Alkenes: Access to CF3-Containing Alkyl Phosphorothioates, Org. Lett., 2021, 23, 5848–5852 CrossRef CAS PubMed; (c) P. Zhang, W. Li, X. Zhu, Y. Li, X. Zhao, S. Shi, F. Zhu, J. Lin and X. Gao, Photoredox and Copper-Catalyzed Sulfonylphosphorothiolation of Alkenes toward β-Sulfonyl Phosphorothioates, Adv. Synth. Catal., 2022, 364, 3316–3320 CrossRef CAS.
- X. Liu, W. Jiang, C. Huang, S. Ma, Q. Wang and H. Cao, Electrochemical phosphorothiolation and 1,4-S→C phospho-Fries rearrangement: controlled access to phosphorothiolated and mercapto-phosphono substituted indolizines, Org. Chem. Front., 2023, 10, 5198–5204 RSC.
- (a) S. Freeman and J. F. Alder, Arylethylamine psychotropic recreational drugs: a chemical perspective, Eur. J. Med. Chem., 2002, 37, 527–539 CrossRef CAS; (b) A. Gallardo-Godoy, A. Fierro, T. H. McLean, M. Castillo, B. K. Cassels, M. Reyes-Parada and D. E. Nichols, Sulfur-Substituted α-Alkyl Phenethylamines as Selective and Reversible MAO-A Inhibitors: Biological Activities, CoMFA Analysis, and Active Site Modeling, J. Med. Chem., 2005, 48, 2407–2419 CrossRef CAS; (c) A. H. Lewin, H. A. Navarro and S. W. Mascarella, Structure–activity correlations for β-phenethylamines at human trace amine receptor 1, Bioorg. Med. Chem., 2008, 16, 7415–7423 CrossRef CAS PubMed.
- (a) Z. Liu, Y. Wang, Z. Wang, T. Zeng, P. Liu and K. M. Engle, Catalytic Intermolecular Carboamination of Unactivated Alkenes via Directed Aminopalladation, J. Am. Chem. Soc., 2017, 139, 11261–11270 CrossRef CAS; (b) Y.-D. Du, B.-H. Chen and W. Shu, Direct Access to Primary Amines from Alkenes by Selective Metal-Free Hydroamination, Angew. Chem., Int. Ed., 2021, 60, 9875–9880 CrossRef CAS; (c) Y. Cai, S. Chatterjee and T. Ritter, Photoinduced Copper-Catalyzed Late-Stage Azidoarylation of Alkenes via Arylthianthrenium Salts, J. Am. Chem. Soc., 2023, 145, 13542–13548 CrossRef CAS; (d) V. Pozhydaiev, C. Muller, J. Moran and D. Lebœuf, Catalytic Synthesis of β-(Hetero)arylethylamines: Modern Strategies and Advances, Angew. Chem., Int. Ed., 2023, 62, e202309289 CrossRef CAS PubMed.
- (a) R. Loven and W. N. Speckamp, A novel 1,4 arylradical rearrangement, Tetrahedron Lett., 1972, 13, 1567–1570 CrossRef; (b) H. J. Köhler and W. N. Speckamp, Intramolecular diastereospecific aryl radical substitution, J. Chem. Soc., Chem. Commun., 1980, 142–143, 10.1039/C39800000142; (c) M. Tada, H. Shijima and M. Nakamura, Smiles-type free radical rearrangement of aromatic sulfonates and sulfonamides: syntheses of arylethanols and arylethylamines, Org. Biomol. Chem., 2003, 1, 2499–2505 RSC; (d) D. M. Whalley, H. A. Duong and M. F. Greaney, A visible light-mediated, decarboxylative, desulfonylative Smiles rearrangement for general arylethylamine syntheses, Chem. Commun., 2020, 56, 11493–11496 RSC; (e) A. R. Allen, J.-F. Poon, R. C. McAtee, N. B. Watson, D. A. Pratt and C. R. J. Stephenson, Mechanism of Visible Light-Mediated Alkene Aminoarylation with Arylsulfonylacetamides, ACS Catal., 2022, 12, 8511–8526 CrossRef CAS.
- E. Derat, G. Masson and A. Claraz, Electrochemically–Driven 1,4−Aryl Migration via Radical Fluoromethylation of N–Allylbenzamides: a Straightforward Access to Functionalized β–Arylethylamines, Angew. Chem., Int. Ed., 2024, 63, e202406017 CrossRef CAS PubMed.
- L. Lan, K. Xu and C. Zeng, The merger of electro-reduction and hydrogen bonding activation for a radical Smiles rearrangement, Chem. Sci., 2024, 15, 13459–13465 RSC.
- X. Chang, Q. Zhang and C. Guo, Electrochemical Reductive Smiles Rearrangement for C–N Bond Formation, Org. Lett., 2019, 21, 10–13 CrossRef CAS.
- (a) T. Ohba, T. Yamauch, K. Higashiyama and N. Takahashi, Potent anticancer activities of novel aminophenol analogues against various cancer cell lines, Bioorg. Med. Chem., 2007, 15, 847–853 CrossRef CAS PubMed; (b) M. Chinnapattu, K. I. Sathiyanarayanan and P. S. Iyer, Synthesis and biological evaluation of adamantane-based aminophenols as a novel class of antiplasmodial agents, Bioorg. Med. Chem. Lett., 2015, 25, 952–955 CrossRef CAS; (c) L. Wang, L. Zhao, X. Jia, L. Jiang, Y. Song, Q. Ye and Z. Lyu, Aminophenols increase proliferation of thyroid tumor cells by inducing the transcription factor activity of estrogen receptor α, Biomed. Pharmacother., 2019, 109, 621–628 CrossRef CAS.
- (a) T. Xu and H. Alper, Pd-Catalyzed Chemoselective Carbonylation of Aminophenols with Iodoarenes: Alkoxycarbonylation vs Aminocarbonylation, J. Am. Chem. Soc., 2014, 136, 16970–16973 CrossRef CAS PubMed; (b) T. Xu, F. Sha and H. Alper, Highly Ligand-Controlled Regioselective Pd-Catalyzed Aminocarbonylation of Styrenes with Aminophenols, J. Am. Chem. Soc., 2016, 138, 6629–6635 CrossRef CAS PubMed; (c) F. Sha and H. Alper, Ligand- and Additive-Controlled Pd-Catalyzed Aminocarbonylation of Alkynes with Aminophenols: Highly Chemo- and Regioselective Synthesis of α,β-Unsaturated Amides, ACS Catal., 2017, 7, 2220–2229 CrossRef CAS.
- L. Gao, Z.-F. Wang, L.-W. Wang, H.-T. Tang, Z.-Y. Mo and M.-X. He, Electrochemical selenium-catalyzed para-amination of N-aryloxyamides: access to polysubstituted aminophenols, Org. Biomol. Chem., 2023, 21, 7895–7899 RSC.
- (a) J. Fastrez, Macrocyclization versus polymerization in polycondensation reactions under high-dilution conditions: a theoretical study, J. Phys. Chem., 1989, 93, 2635–2642 CrossRef CAS; (b) G. A. Molander, Diverse Methods for Medium Ring Synthesis, Acc. Chem. Res., 1998, 31, 603–609 CrossRef CAS.
- (a) P. Dowd and W. Zhang, Free radical-mediated ring expansion and related annulations, Chem. Rev., 1993, 93, 2091–2115 CrossRef CAS; (b) J. R. Donald and W. P. Unsworth, Ring-Expansion Reactions in the Synthesis of Macrocycles and Medium-Sized Rings, Chem. – Eur. J., 2017, 23, 8780–8799 CrossRef CAS.
- E. A. Wappes, M. S. Mubarak and D. G. Peters, Electrochemical Reduction of 1-Bromomethyl-2-oxocycloalkane-1-carboxylates at Silver Cathodes in Dimethylformamide: One-Carbon Ring-Expansion Reactions, J. Electrochem. Soc., 2014, 161, G122 CrossRef CAS.
- J. Strehl, C. Kahrs, T. Müller, G. Hilt and J. Christoffers, Electrochemical-Induced Ring Transformation of Cyclic α-(ortho-Iodophenyl)-β-oxoesters, Chem. – Eur. J., 2020, 26, 3222–3225 CrossRef CAS PubMed.
- (a) Y. Schun and G. A. Cordell, Studies on the NMR Spectroscopic Properties of Gelsemine–Revisions and Refinements, J. Nat. Prod., 1985, 48, 969–971 CrossRef CAS; (b) H. Ishikawa, D. A. Colby, S. Seto, P. Va, A. Tam, H. Kakei, T. J. Rayl, I. Hwang and D. L. Boger, Total Synthesis of Vinblastine, Vincristine, Related Natural Products, and Key Structural Analogues, J. Am. Chem. Soc., 2009, 131, 4904–4916 CrossRef CAS PubMed; (c) X. Zhou, T. Xiao, Y. Iwama and Y. Qin, Biomimetic Total Synthesis of (+)-Gelsemine, Angew. Chem., Int. Ed., 2012, 51, 4909–4912 CrossRef CAS.
- (a) T. J. Snape, Recent advances in the semi-pinacol rearrangement of α-hydroxy epoxides and related compounds, Chem. Soc. Rev., 2007, 36, 1823–1842 RSC; (b) B. Wang and Y. Q. Tu, Stereoselective Construction of Quaternary Carbon Stereocenters via a Semipinacol Rearrangement Strategy, Acc. Chem. Res., 2011, 44, 1207–1222 CrossRef CAS; (c) X.-M. Zhang, Y.-Q. Tu, F.-M. Zhang, Z.-H. Chen and S.-H. Wang, Recent applications of the 1,2-carbon atom migration strategy in complex natural product total synthesis, Chem. Soc. Rev., 2017, 46, 2272–2305 RSC.
- (a) Z.-M. Chen, W. Bai, S.-H. Wang, B.-M. Yang, Y.-Q. Tu and F.-M. Zhang, Copper-Catalyzed Tandem Trifluoromethylation/Semipinacol Rearrangement of Allylic Alcohols, Angew. Chem., Int. Ed., 2013, 52, 9781–9785 CrossRef CAS PubMed; (b) H. Egami, R. Shimizu, Y. Usui and M. Sodeoka, Iron-catalyzed trifluoromethylation with concomitant C–C bond formation via 1,2-migration of an aryl group, Chem. Commun., 2013, 49, 7346–7348 RSC; (c) X. Liu, F. Xiong, X. Huang, L. Xu, P. Li and X. Wu, Copper-Catalyzed Trifluoromethylation-Initiated Radical 1,2-Aryl Migration in α,α-Diaryl Allylic Alcohols, Angew. Chem., Int. Ed., 2013, 52, 6962–6966 CrossRef CAS PubMed; (d) Z.-M. Chen, Z. Zhang, Y.-Q. Tu, M.-H. Xu, F.-M. Zhang, C.-C. Li and S.-H. Wang, A Mn(iii)/TEMPO-co-mediated tandem azidation–1,2-carbon migration reaction of allylic silyl ethers, Chem. Commun., 2014, 50, 10805–10808 RSC; (e) X.-Z. Shu, M. Zhang, Y. He, H. Frei and F. D. Toste, Dual Visible Light Photoredox and Gold-Catalyzed Arylative Ring Expansion, J. Am. Chem. Soc., 2014, 136, 5844–5847 CrossRef CAS PubMed.
- J.-C. Kang, Y.-Q. Tu, J.-W. Dong, C. Chen, J. Zhou, T.-M. Ding, J.-T. Zai, Z.-M. Chen and S.-Y. Zhang, Electrochemical Semipinacol Rearrangements of Allylic Alcohols: Construction of All-Carbon Quaternary Stereocenters, Org. Lett., 2019, 21, 2536–2540 CrossRef CAS PubMed.
- H. I. Jung, Y. Kim and D. Y. Kim, Electrochemical trifluoromethylation/semipinacol rearrangement sequences of alkenyl alcohols: synthesis of β-CF3-substituted ketones, Org. Biomol. Chem., 2019, 17, 3319–3323 RSC.
- Y. J. Kim and D. Y. Kim, Electrochemical Radical Selenylation/1,2-Carbon Migration and Dowd–Beckwith-Type Ring-Expansion Sequences of Alkenylcyclobutanols, Org. Lett., 2019, 21, 1021–1025 CrossRef CAS PubMed.
- Y. J. Kim and D. Y. Kim, Electrochemical radical arylsulfonylation/semipinacol rearrangement sequences of alkenylcyclobutanols: Synthesis of β-sulfonated cyclic ketones, Tetrahedron Lett., 2019, 60, 1287–1290 CrossRef CAS.
- C. Chen, J.-C. Kang, C. Mao, J.-W. Dong, Y.-Y. Xie, T.-M. Ding, Y.-Q. Tu, Z.-M. Chen and S.-Y. Zhang, Electrochemical halogenation/semi-pinacol rearrangement of allylic alcohols using inorganic halide salt: an eco-friendly route to the synthesis of β-halocarbonyls, Green Chem., 2019, 21, 4014–4019 RSC.
- (a) H. Zhang, S. Qiu, P. Tamez, G. T. Tan, Z. Aydogmus, N. V. Hung, N. M. Cuong, C. Angerhofer, D. D. Soejarto, J. M. Pezzuto and H. H. S. Fong, Antimalarial Agents from Plants II. Decursivine, A New Antimalarial Indole Alkaloid from Rhaphidophora decursiva, Pharm. Biol., 2002, 40, 221–224 CrossRef CAS; (b) N. Brown, B. Xie, N. Markina, D. VanderVelde, J.-P. H. Perchellet, E. M. Perchellet, K. R. Crow and K. R. Buszek, Synthesis of a natural product-inspired eight-membered ring lactam library via ring-closing metathesis, Bioorg. Med. Chem. Lett., 2008, 18, 4876–4879 CrossRef CAS PubMed; (c) J. Li, J. Li, Y. Xu, Y. Wang, L. Zhang, L. Ding, Y. Xuan, T. Pang and H. Lin, Asymmetric synthesis and biological activities of natural product (+)-balasubramide and its derivatives, Nat. Prod. Res., 2016, 30, 800–805 CrossRef CAS PubMed.
- (a) S.-M. Lu and H. Alper, Sequence of Intramolecular Carbonylation and Asymmetric Hydrogenation Reactions: Highly Regio- and Enantioselective Synthesis of Medium Ring Tricyclic Lactams, J. Am. Chem. Soc., 2008, 130, 6451–6455 CrossRef CAS PubMed; (b) R. Mancuso, D. S. Raut, N. Marino, G. De Luca, C. Giordano, S. Catalano, I. Barone, S. Andò and B. Gabriele, A Palladium-Catalyzed Carbonylation Approach to Eight-Membered Lactam Derivatives with Antitumor Activity, Chem. – Eur. J., 2016, 22, 3053–3064 CrossRef CAS PubMed.
- (a) H. M. A. Hassan, Recent applications of ring-closing metathesis in the synthesis of lactams and macrolactams, Chem. Commun., 2010, 46, 9100–9106 RSC; (b) N. Hegmann, L. Prusko, N. Diesendorf and M. R. Heinrich, In Situ Conformational Fixation of the Amide Bond Enables General Access to Medium-Sized Lactams via Ring-Closing Metathesis, Org. Lett., 2018, 20, 7825–7829 CrossRef CAS PubMed.
- (a) B. Zhou, L. Li, X.-Q. Zhu, J.-Z. Yan, Y.-L. Guo and L.-W. Ye, Yttrium-Catalyzed Intramolecular Hydroalkoxylation/Claisen Rearrangement Sequence: Efficient Synthesis of Medium-Sized Lactams, Angew. Chem., Int. Ed., 2017, 56, 4015–4019 CrossRef CAS PubMed; (b) M. Kunitski, N. Eicke, P. Huber, J. Köhler, S. Zeller, J. Voigtsberger, N. Schlott, K. Henrichs, H. Sann, F. Trinter, L. P. H. Schmidt, A. Kalinin, M. S. Schöffler, T. Jahnke, M. Lein and R. Dörner, Double-slit photoelectron interference in strong-field ionization of the neon dimer, Nat. Commun., 2019, 10, 1 CrossRef CAS PubMed.
- T. C. Stephens and W. P. Unsworth, Consecutive Ring-Expansion Reactions for the Iterative Assembly of Medium-Sized Rings and Macrocycles, Synlett, 2020, 133–146 CAS.
- Z. Xu, Z. Huang, Y. Li, R. Kuniyil, C. Zhang, L. Ackermann and Z. Ruan, Catalyst-free, direct electrochemical synthesis of annulated medium-sized lactams through C–C bond cleavage, Green Chem., 2020, 22, 1099–1104 RSC.
- K. Liu, C. Song, X. Jiang, X. Dong, Y. Deng, W. Song, Y. Yang and A. Lei, Electrochemical Ring Expansion to Synthesize Medium-Sized Lactams Through C–C Bond Cleavage, CCS Chem., 2020, 3, 2233–2244 CrossRef.
- (a) R. C. Rizzo, M. Udier-Blagović, D.-P. Wang, E. K. Watkins, M. B. K. Smith, R. H. Smith, J. Tirado-Rives and W. L. Jorgensen, Prediction of Activity for Nonnucleoside Inhibitors with HIV-1 Reverse Transcriptase Based on Monte Carlo Simulations, J. Med. Chem., 2002, 45, 2970–2987 CrossRef CAS PubMed; (b) P. Zhang, E. A. Terefenko, A. Fensome, J. Wrobel, R. Winneker, S. Lundeen, K. B. Marschke and Z. Zhang, 6-Aryl-1,4-dihydro-benzo[d][1,3]oxazin- 2-ones: A Novel Class of Potent, Selective, and Orally Active Nonsteroidal Progesterone Receptor Antagonists, J. Med. Chem., 2002, 45, 4379–4382 CrossRef CAS PubMed.
- (a) S. S. Nikam, P.-W. Yuen, B. E. Kornberg, B. Tobias and M. F. Rafferty, Novel Use of Substituted 1,4-Dihydrobenz[d][1,3]oxazin-2-ones in the Synthesis of Important Aminomethyl o-Nitroanilines, J. Org. Chem., 1997, 62, 9331–9334 CrossRef CAS; (b) C. E. Houlden, G. C. Lloyd-Jones and K. I. Booker-Milburn, Facile Double-Lithiation of a Transient Urea: Vicarious ortho-Metalation of Aniline Derivatives, Org. Lett., 2010, 12, 3090–3092 CrossRef CAS PubMed; (c) Y. Zhao, B. Huang, C. Yang, Q. Chen and W. Xia, Sunlight-Driven Forging of Amide/Ester Bonds from Three Independent Components: An Approach to Carbamates, Org. Lett., 2016, 18, 5572–5575 CrossRef CAS PubMed; (d) E. M. Larin, A. Torelli, J. Loup and M. Lautens, One-Pot, Three-Step Synthesis of Benzoxazinones via Use of the Bpin Group as a Masked Nucleophile, Org. Lett., 2021, 23, 2720–2725 CrossRef CAS PubMed.
- M. Vayer, M. Pastor, C. Kofink and N. Maulide, Electrochemical Rearrangement of 3-Hydroxyoxindoles into Benzoxazinones, Org. Lett., 2022, 24, 27–32 CrossRef CAS.
- N. Chen and H.-C. Xu, Electrochemical generation of nitrogen-centered radicals for organic synthesis, Green Synth. Catal., 2021, 2, 165–178 CrossRef.
- Y. Ma, J. Lv, C. Liu, X. Yao, G. Yan, W. Yu and J. Ye, Electrochemical [4 + 2] Annulation-Rearrangement-Aromatization of Styrenes: Synthesis of Naphthalene Derivatives, Angew. Chem., Int. Ed., 2019, 58, 6756–6760 CrossRef CAS PubMed.
- (a) A. A. Alhaider, M. A. Abdelkader and E. J. Lien, Design, synthesis and pharmacological activities of 2-substituted 4-phenylquinolines as potential antidepressant drugs, J. Med. Chem., 1985, 28, 1394–1398 CrossRef CAS PubMed; (b) M. Giannangeli, N. Cazzolla, M. R. Luparini, M. Magnani, M. Mabilia, G. Picconi, M. Tomaselli and L. Baiocchi, Effect of Modifications of the Alkylpiperazine Moiety of Trazodone on 5HT2A and α1 Receptor Binding Affinity, J. Med. Chem., 1999, 42, 336–345 CrossRef CAS PubMed; (c) C.-L. Lee, W.-H. Lee and C.-H. Yang, The effects of co-sensitization in dye-sensitized solar cells, J. Mater. Sci., 2013, 48, 3448–3453 CrossRef CAS.
- Z. Ye, Y. Wu, N. Chen, H. Zhang, K. Zhu, M. Ding, M. Liu, Y. Li and F. Zhang, Enantiospecific electrochemical rearrangement for the synthesis of hindered triazolopyridinone derivatives, Nat. Commun., 2020, 11, 3628 CrossRef CAS PubMed.
- C. Cai, Y. Lu, C. Yuan, Z. Fang, X. Yang, C. Liu and K. Guo, Organocatalytic Electrosynthesis of Cinnolines through Cascade Radical Cyclization and Migration, ACS Sustainable Chem. Eng., 2021, 9, 16989–16996 CrossRef CAS.
- (a) B. Pirotte, P. de Tullio, P. Lebrun, M. H. Antoine, J. Fontaine, B. Masereel, M. Schynts, L. Dupont, A. Herchuelz and J. Delarge, 3-(Alkylamino)-4H-pyrido[4,3-e]1,2,4-thiadiazine 1,1-dioxides as powerful inhibitors of insulin release from rat pancreatic B-cells: a new class of potassium channel openers?, J. Med. Chem., 1993, 36, 3211–3213 CrossRef CAS PubMed; (b) C. Nicolas, M. Verny, I. Giraud, M. Ollier, M. Rapp, J.-C. Maurizis and J.-C. Madelmont, New Quaternary Ammonium Oxicam Derivatives Targeted toward Cartilage: Synthesis, Pharmacokinetic Studies, and Antiinflammatory Potency, J. Med. Chem., 1999, 42, 5235–5240 CrossRef CAS; (c) W. A. Coetzee, Multiplicity of effectors of the cardioprotective agent, diazoxide, Pharmacol. Ther., 2013, 140, 167–175 CrossRef CAS.
- A. F. Kornahrens, A. B. Cognetta III, D. M. Brody, M. L. Matthews, B. F. Cravatt and D. L. Boger, Design of Benzoxathiazin-3-one 1,1-Dioxides as a New Class of Irreversible Serine Hydrolase Inhibitors: Discovery of a Uniquely Selective PNPLA4 Inhibitor, J. Am. Chem. Soc., 2017, 139, 7052–7061 CrossRef CAS PubMed.
- (a) E. Wertheim, Preparation of N-Benzoyl-o-aminobenzenesulfonamide. Condensation to Heterocyclic Compounds1, J. Am. Chem. Soc., 1934, 56, 971–973 CrossRef CAS; (b) S. Suzue and T. Irikura, Studies on Hypoglycemic Agents. IV. Synthesis of 1, 4, 3-Benzoxathiazine-4, 4-dioxides, Chem. Pharm. Bull., 1968, 16, 806–813 CrossRef CAS; (c) T. Iwakawa, H. Tamura, A. Murabayashi and Y. Hayase, Cycloaddition in Synthesis of Sulfonamide Derivatives. IV. One-Pot Synthesis of 3-Dimethylamino-4, 1, 2-benzoxathiazine 1, 1-Dioxides, 3-Methoxy-4-methyl-1, 2, 4-benzothiadiazine 1, 1-Dioxide and 3-Dimethylamino-1, 4, 2-benzodithiazine 1, 1-Dioxides, Chem. Pharm. Bull., 1991, 39, 1939–1943 CrossRef CAS.
- Z. Shi, Y. Li, N. Li, W.-Z. Wang, H.-K. Lu, H. Yan, Y. Yuan, J. Zhu and K.-Y. Ye, Electrochemical Migratory Cyclization of N-Acylsulfonamides, Angew. Chem., Int. Ed., 2022, 61, e202206058 CrossRef CAS PubMed.
- R. Li, D. Yuan, M. Ping, Y. Zhu, S. Ni, M. Li, L. Wen and L.-B. Zhang, Electrochemically-promoted synthesis of benzo[b]thiophene-1,1-dioxides via strained quaternary spirocyclization, Chem. Sci., 2022, 13, 9940–9946 RSC.
- (a) M. P. Sibi and T. R. Rheault, in Radicals in Organic Synthesis, 2001, pp. 461–478, DOI:10.1002/9783527618293.ch23; (b) J. C. Walton, in Encyclopedia of Radicals in Chemistry, Biology and Materials, 2012, DOI:10.1002/9781119953678.rad001.
- Z. Shi, S. Dong, T. Liu, W.-Z. Wang, N. Li, Y. Yuan, J. Zhu and K.-Y. Ye, Electrochemical cascade migratory versus ortho-cyclization of 2-alkynylbenzenesulfonamides, Chem. Sci., 2024, 15, 2827–2832 RSC.
- G. C. Lloyd-Jones, J. D. Moseley and J. S. Renny, Mechanism and Application of the Newman-Kwart O→S Rearrangement of O-Aryl Thiocarbamates, Synthesis, 2008, 661–689 CAS.
- T. Broese, A. F. Roesel, A. Prudlik and R. Francke, An Electrocatalytic Newman–Kwart-type Rearrangement, Org. Lett., 2018, 20, 7483–7487 CrossRef CAS.
- (a) H. M. L. Davies and M. S. Long, Recent Advances in Catalytic Intramolecular C–H Aminations, Angew. Chem., Int. Ed., 2005, 44, 3518–3520 CrossRef CAS PubMed; (b) D.-S. Kim, W.-J. Park and C.-H. Jun, Metal–Organic Cooperative Catalysis in C–H and C–C Bond Activation, Chem. Rev., 2017, 117, 8977–9015 CrossRef CAS PubMed.
- Y. Adeli, K. Huang, Y. Liang, Y. Jiang, J. Liu, S. Song, C.-C. Zeng and N. Jiao, Electrochemically Oxidative C–C Bond Cleavage of Alkylarenes for Anilines Synthesis, ACS Catal., 2019, 9, 2063–2067 CrossRef CAS.
- (a) D. E. Nichols and C. D. Nichols, Serotonin Receptors, Chem. Rev., 2008, 108, 1614–1641 CrossRef CAS PubMed; (b) K. Karrouchi, S. Radi, Y. Ramli, J. Taoufik, Y. N. Mabkhot, F. A. Al-aizari and M. H. Ansar, Synthesis and Pharmacological Activities of Pyrazole Derivatives: A Review, Molecules, 2018, 23, 134 CrossRef PubMed.
- J. Lan, K. Lin, X. Zhang and T. Zhu, Stereoselective migratory heteroaryltrifluoromethylation of allylic amines via electrosynthesis, Green Chem., 2022, 24, 6138–6144 RSC.
- Z.-L. Lei, Z.-C. Ding, S.-H. Li, F.-H. Cui, H.-T. Tang and Y.-M. Pan, Electrochemical synthesis of β-difluoromethylamide compounds by N-benzenesulfonylacrylamide with difluorine reagents, Chem. Commun., 2024, 60, 7614–7617 RSC.
- (a) Y. Zhao, S. Yu, W. Sun, L. Liu, J. Lu, D. McEachern, S. Shargary, D. Bernard, X. Li, T. Zhao, P. Zou, D. Sun and S. Wang, A Potent Small-Molecule Inhibitor of the MDM2−p53 Interaction (MI-888) Achieved Complete and Durable Tumor Regression in Mice, J. Med. Chem., 2013, 56, 5553–5561 CrossRef CAS PubMed; (b) N. Ye, H. Chen, E. A. Wold, P.-Y. Shi and J. Zhou, Therapeutic Potential of Spirooxindoles as Antiviral Agents, ACS Infect. Dis., 2016, 2, 382–392 CrossRef CAS PubMed; (c) L.-M. Zhou, R.-Y. Qu and G.-F. Yang, An overview of spirooxindole as a promising scaffold for novel drug discovery, Expert Opin. Drug Discovery, 2020, 15, 603–625 CrossRef CAS PubMed.
- (a) N. Finch and W. I. Taylor, The Conversion of Tetrahydro-β-Carboline Alkaloids into Oxindoles. the Structures and Partial Syntheses of Mitraphylline and Rhyncophylline, J. Am. Chem. Soc., 1962, 84, 1318–1320 CrossRef CAS; (b) J. Shavel and H. Zinnes, Oxindole Alkaloids. I. Oxidative-Rearrangement of Indole Alkaloids to their Oxindole Analogs, J. Am. Chem. Soc., 1962, 84, 1320–1321 CrossRef CAS; (c) P. S. Baran and J. M. Richter, Enantioselective Total Syntheses of Welwitindolinone A and Fischerindoles I and G, J. Am. Chem. Soc., 2005, 127, 15394–15396 CrossRef CAS PubMed.
- Y. Zheng, Y. T. Cheung, L. Liang, H. Qiu, L. Zhang, A. Tsang, Q. Chen and R. Tong, Electrochemical oxidative rearrangement of tetrahydro-β-carbolines in a zero-gap flow cell, Chem. Sci., 2022, 13, 10479–10485 RSC.
- D. Liu and H.-C. Xu, Electrochemical Rearrangement of Indoles to Spirooxindoles in Continuous Flow, Eur. J. Org. Chem., 2023, e202200987 CrossRef CAS.
- V. Jose, E. J. Diana, U. S. Kanchana and T. V. Mathew, Current trends and advancements in Ferrier and Petasis-Ferrier rearrangement, J. Organomet. Chem., 2023, 991, 122691 CrossRef CAS.
- (a) K. Fukaya, K. Kodama, Y. Tanaka, H. Yamazaki, T. Sugai, Y. Yamaguchi, A. Watanabe, T. Oishi, T. Sato and N. Chida, Synthesis of Paclitaxel. 2. Construction of the ABCD Ring and Formal Synthesis, Org. Lett., 2015, 17, 2574–2577 CrossRef CAS PubMed; (b) A. K. Ghosh, G. C. Reddy, A. J. MacRae and M. S. Jurica, Enantioselective Synthesis of Spliceostatin G and Evaluation of Bioactivity of Spliceostatin G and Its Methyl Ester, Org. Lett., 2018, 20, 96–99 CrossRef CAS PubMed.
- C. Qi, G. Goti, A. Sartorel, L. Dell'Amico and D. Mazzarella, Electrochemical Ferrier Rearrangement of Glycals, Org. Lett., 2024, 26, 9328–9333 CrossRef CAS PubMed.
- (a) R. Gianatassio, J. M. Lopchuk, J. Wang, C.-M. Pan, L. R. Malins, L. Prieto, T. A. Brandt, M. R. Collins, G. M. Gallego, N. W. Sach, J. E. Spangler, H. Zhu, J. Zhu and P. S. Baran, Strain-release amination, Science, 2016, 351, 241–246 CrossRef CAS; (b) R. Gianatassio and D. Kadish, Direct Alkylation of 1-Azabicyclo[1.1.0]butanes, Org. Lett., 2019, 21, 2060–2063 CrossRef CAS PubMed; (c) M. Ociepa, A. J. Wierzba, J. Turkowska and D. Gryko, Polarity-Reversal Strategy for the Functionalization of Electrophilic Strained Molecules via Light-Driven Cobalt Catalysis, J. Am. Chem. Soc., 2020, 142, 5355–5361 CrossRef CAS PubMed; (d) N. Jha, P. Mishra and M. Kapur, Strained cycloalkanols in C–C bond formation reactions: a boon in disguise!, Org. Chem. Front., 2023, 10, 4941–4971 RSC.
- J. Qi, C. Wang, G. Wang, P. O’Neill, S. R. Dubbaka, H. T. Ang, X. Chen and J. Wu, Strain-Release-Driven Electrochemical Skeletal Rearrangement of Non-Biased Alkyl Cyclopropanes/Butanes, Angew. Chem., 2025, 64, e202413723 CrossRef CAS PubMed.
Footnote |
| † These authors contributed equally to this work. |
| This journal is © the Partner Organisations 2025 |