Open Access Article
Open Access ArticleReactivity of an aza-dipyrrin and an aza-BODIPY motif bearing two 2-pyridyl units: an emissive NNN binding pocket
Rosinah Liandrah
Gapare
 a,
Roberto M.
Diaz-Rodriguez
a,
Roberto M.
Diaz-Rodriguez
 ab,
Victoria A.
Williams
ab,
Victoria A.
Williams
 a,
Mark
Atwood
a,
Michael J.
Cotnam
a,
James W.
Hilborn
a,
Erin R.
Johnson
a,
Mark
Atwood
a,
Michael J.
Cotnam
a,
James W.
Hilborn
a,
Erin R.
Johnson
 a,
Katherine N.
Robertson
a,
Katherine N.
Robertson
 c and
Alison
Thompson
c and
Alison
Thompson
 *a
*a
aDepartment of Chemistry, Dalhousie University, Halifax, Nova Scotia B3H 4J3, Canada. E-mail: Alison.Thompson@dal.ca
bDepartment of Chemistry and Biomolecular Sciences, University of Ottawa, Ottawa, Ontario K1N 6N5, Canada
cDepartment of Chemistry, Saint Mary's University, Halifax, Nova Scotia B3H 3C3, Canada
First published on 3rd November 2025
Abstract
The synthesis and reactivity of an aza-dipyrrin and an aza-BODIPY motif each featuring two 2-pyridyl rings appended to the pyrrolic units are reported. The tricoordinate NNN binding domain was complexed to various metals, with Zn(II) analogues isolated and crystallographically characterised. Furthermore, the addition of alcohols across the aza-fulvenium unit provided an amino-decorated framework that served as a convenient pro-ligand for turn-on emissive metal sensing.
Introduction
Boron-containing ring systems have garnered significant attention due to their applications across diverse fields including materials science, optoelectronics, catalysis, sensors and medicinal agents.1–3 Among these, boron complexes of dipyrrins, called BODIPYs (4,4-disubstituted-4-bora-3a,4a-diaza-s-indacenes, Fig. 1, left) shine on account of their exceptional photophysical properties. These include high fluorescence quantum yields (Φf), tunable absorption/emission wavelengths, and remarkable (photo)stability. Such attributes make BODIPYs indispensable in applications such as bioimaging and light-harvesting, and as sensors for environmental and biological analytes.4Aza-BODIPYs are distinguished from BODIPYs by featuring a nitrogen atom at the central (i.e., meso) position of the dipyrrinato skeleton (Fig. 1, middle). This variation influences the photochemical properties; for example, aza-BODIPYs typically exhibit absorption and emission in the red-NIR region of the spectrum, which is a highly advantageous characteristic for biological applications. In addition, the aza-BODIPY framework (and its resultant properties) can be tuned through structural modification, such as introduction of electron-withdrawing or electron-donating aryl substituents.5–8 The incorporation of functional aryl groups, as substituents on the dipyrrolic core, is thus an attractive approach to developing new application-focused materials.
To this end, a one-pot synthesis of 2-phenyl-4-pyridylpyrroles used 4-acetylpyridineoxime tosylate and 4′-methoxyacetophenones as starting materials, courtesy of an in situ Neber rearrangement that liberates a highly electrophilic 2H-azirine.9 This protocol was a key component in a multi-step pathway to generate aza-dipyrrins. However, applicability was limited to aza-dipyrrinato systems bearing pyridyl units on just one of the two pyrrolic moieties, due to the approach involving reaction of an α-free pyrrole with an α-nitrosated pyrrole. As the synthesis and isolation of α-nitroso pyridyl-substituted pyrroles remains elusive,10 this step-wise approach is unsuited to the preparation of aza-dipyrrins bearing a pyridyl unit on each of the pyrroles in the framework. Nevertheless, the tunability of photophysical and photochemical properties of aza-BODIPYs bearing the electron-withdrawing pyridyl motif on the pyrrolic framework offers significant allure. Furthermore, a 3,7-dipyridyl aza-dipyrrin presents a potential secondary coordinating pocket defined by the two pyridyl motifs and the meso-nitrogen atom, extending opportunities for tridentate NNN chelation9,11 alongside the primary pyrrole-based site through which –BR2 can coordinate.12
In this vein, a one-pot synthesis of the bis(2-pyridyl) aza-BODIPY 1 (Fig. 1, right) from 2-phenyl-4-(2-pyridyl)pyrrole enabled the absorption and emission maxima to be determined.13 Furthermore, binding with Hg(II) resulted in a bathochromic shift in the absorption characteristics, and identified the potential of 1 as a highly selective chemosensor. Intrigued by the potential applications14–19 for the two complementary coordinating pockets, we targeted an improved synthesis of 1. The potential of the neutral, tridentate NNN pocket consisting of the meso-nitrogen atom plus the two pyridyl nitrogen atoms, as well as the bidentate NN monoanionic pocket featuring the two pyrrolic nitrogen atoms, was also explored.
Results and discussion
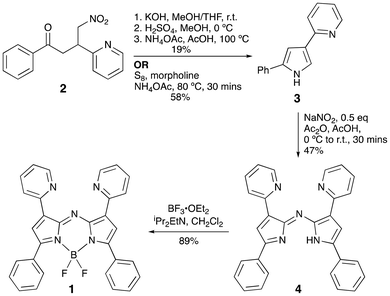
Access to 1,3,7,9-tetraaryl aza-dipyrrins typically stems from the Michael addition of nitromethane to the corresponding chalcone precursors, themselves produced by aldol condensation of the corresponding aromatic aldehyde with acetophenone.8 Accordingly, reaction of 2-pyridinecarboxaldehyde and acetophenone in the presence of LiOH·H2O and Et2O/EtOH, rather than via the reported approach involving KOH/MeOH,20 provided 1-phenyl-3-(2-pyridyl)-2-propen-1-one. Subsequent reaction with nitromethane in the presence of Et2NH/MeOH at reflux temperature provided the nitrobutanone 2 (Scheme 1).A reported procedure13 for converting nitrobutanone 2 into the key pyridyl-appended pyrrole 3 leveraged a Nef reaction, involving various sequential basic and acidic steps performed over two days to ensure condensation, deprotection and oxidation. We found this route to 3 to be procedurally heavy and inefficient, ultimately providing capricious yields upon scale-up. To address these limitations, pyrrole formation utilising sulfur and morpholine alongside an ammonia source was investigated, as this approach was reported to afford diaryl-substituted pyrroles in high yields after short reaction times.21 Nitrobutanone 2, morpholine, ammonium acetate, and S8, along with ethyl acetate to aid stirring, were thus reacted for 30 minutes, which led to a dark orange-colored mixture (Scheme 1). Further addition of ethyl acetate led to precipitation of the excess sulfur; then, filtration, aqueous extraction of the mixture, and purification via column chromatography provided pyrrole 3 in an efficient and scalable fashion.
Attention then turned to a one-pot synthesis of aza-dipyrrin 4 (Scheme 1), i.e., the formal condensation product of the reaction of 2 eq. of 3 with an NO+ source. Following the published procedure,13 a solution of 3 in Ac2O/AcOH was reacted with NaNO2 whilst stirring first at 0 °C and then at 80 °C. Brief examination10 revealed that, under the reaction conditions, the nitrosopyrrole is formed immediately upon addition of NaNO2 yet rapid decomposition occurs on heating. The significant degradation of the nitrosopyrrole formed in situ therefore impedes the formation of the desired aza-dipyrrin 4. The reaction mixture was thus maintained at 0 °C, whereby the characteristically vivid green 2-nitroso-3,5-diarylpyrrole formed efficiently. Gradual warming to room temperature subsequently enabled the reaction of the remaining pyrrole 3 with the nitrosated species, resulting in production of 4. Isolation involved extraction using NaHCO3/CH2Cl2, followed by purification via column chromatography on neutral alumina to furnish 4 as a dark blue solid in 47% yield. A crystal structure of 4 (Fig. 2, left) confirms the two pyridyl moieties of the aza-dipyrrin flanking the nitrogen atom in the meso position. The aza-dipyrrinato core of 4 lies essentially coplanar with the two phenyl rings (angle between planes <6°); the two pyridyl rings, while freely rotatable, are slightly tilted cf. the core in the solid-state structure (up to 20°).
Although previously isolated from a one-pot reaction starting from pyrrole 3,13 stirring a solution of 4 in CH2Cl2 with BF3·Et2O and NEt3 for 24 hours yielded only trace amounts of the desired aza-BODIPY 1. However, replacing NEt3 with iPr2EtN and reducing the reaction time to just 30 minutes led to a significantly improved outcome. After purification via column chromatography on silica, the desired aza-BODIPY 1 was isolated as a green solid in 89% yield, a success that was maintained on scales of up to 2 g. A crystal structure of 1 (Fig. 2, right) confirmed the presence of the –BF2 unit. Unlike the corresponding ligand, the aryl substituents around the aza-dipyrrinato core of 1 are notably more deviated from coplanarity (by up to 50° for the phenyl substituent at C1, and 17–25° for the others).
With reliable syntheses in hand, attention turned to investigating the reactivity of the aza-BODIPY 1 and the dipyrrin 4. The presence of pyridyl groups on the framework offer the opportunity to quench emission via photoinduced electron transfer, leading us to explore the effects of methylation. Indeed, the redox properties of N,N′-4,4-dialkyl bipyridinium systems (viologens) have been extensively studied for use in diverse fields like electrocatalysis, electro- and photochromism, herbicides, and energy storage systems.22–25 Treatment of the aza-BODIPY 1 with CH3I resulted in a yellowing of the dark green solution alongside formation of an orange precipitate. The precipitate, collected by filtration, was identified as the dimethylated analogue 5 (Fig. 3, left). The filtrate was concentrated to provide a green oil, which was not further purified; analysis of the corresponding NMR data confirmed methylation at just one of the pyridyl nitrogen atoms. The NMR spectra of 5 are representative of an unsymmetrical structure, presumably as a result of the two alkylated pyridyl groups exhibiting different conformational preferences. Protonation of 1, to provide the triflate salt 6a (Fig. 3, right) and the perchlorate salt 6b, also demonstrated reactivity at the pyridyl nitrogen atoms as confirmed by X-ray crystallography (Fig. 4 and SI).
Efforts to determine the pKa of 1 using absorption spectroscopy were complicated by evident solvatochromic behaviour. The dimethylated analogue 5 also exhibited solvatochromicity: a solution of 5 in MeCN was green in colour; solutions in MeOH, DMSO and DMF were orange; and those in THF and acetone were yellow (Fig. S10). Solvent-dependent behaviour was further evident in NMR spectra, as illustrated in Fig. 5.
The photophysical properties of the dipyridyl-containing aza-dipyrrinato constructs are presented in Table 1. Complexation of 4 with –BF2 to form the aza-BODIPY 1 results in an anticipated red shift in absorbance maximum, and a corresponding shift in emission along with a significant increase in quantum yield. The methylated analogue 5 retains the absorption characteristics of the BODIPY 1 and, unsurprisingly, methylation results in quenched emission.
| Compound | Solvent | λ max,abs (nm) | ε (M−1 cm−1) | λ max,em (nm) | Stokes shift (nm) | Φ f |
|---|---|---|---|---|---|---|
| a Wavelength of maximum absorbance. b Wavelength of maximum fluorescence emission. | ||||||
| 1 | CH2Cl2 | 664 | 82![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000 |
703 | 39 | 0.27 |
| 4 | CH2Cl2 | 604 | 29![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000 |
664 | 60 | 0.004 |
| 5 | MeCN | 664 | 44![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000 |
706 | 42 | 0.04 |
| 6 | MeCN | 634 | 11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000 |
731 | 97 | 0.18 |
| 7 | MeCN | 438 | 9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000 |
676 | 238 | 0.03 |
| 8 | MeCN | 438 | 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000 |
704 | 266 | 0.02 |
| 1-Zn(ClO4)2 | MeCN | 694 | 73![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000 |
745 | 51 | 0.13 |
| 4-Zn(ClO4)2 | MeCN | 648 | 34![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000 |
712 | 64 | 0.006 |
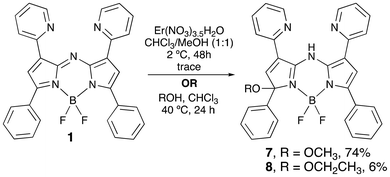
Encouraged by the propensity of the tridentate NNN pocket towards methylation and protonation, the coordination reactivity of 1 was next explored. With a view to evaluating the potential complexation of lanthanides within the tridentate pocket, a solution of 1 in CHCl3/MeOH was reacted with 1 eq. Er(NO3)3·5H2O, as shown in Scheme 2. Over the course of four days the colour gradually changed from deep green, through yellowish-green, and eventually to a deep blue solution from which some solid material precipitated. After this time, a small number of orange crystals were obtained, isolated via filtration and washed with pentane.
The 1H NMR spectrum (Fig. 6, top) of a solution of this material in CD3CN displayed a methyl signal at 3.5 ppm, suggestive of the presence of a methoxy group. Furthermore, two singlets each integrating to 1H, and with chemical shifts corresponding to a H-atom on the carbon backbone of a pyrrolic ring and a vinylic hydrogen atom, provided evidence suggestive of desymmetrisation cf. 1. The peaks corresponding to the aryl protons attributable to the pyridyl rings further supported desymmetrisation. The 1H NMR spectrum featured a signal at 14.7 ppm, likely indicative of a highly deshielded NH unit.
Given these data, and the sharpness of the NMR signals, the isolated material was evidently not the expected BODIPY-based lanthanide complex. Given that the –BF2 motif remained coordinated to the pyrrolic nitrogen atoms, as confirmed by 11B and 19F NMR spectroscopy, it seemed reasonable to conclude that the observed NH signal originated with the meso-nitrogen atom: as such, addition of methanol across the azafulvenium moiety of 1, to form 7, was suspected (Scheme 2), and was confirmed by an X-ray crystal structure (Fig. 7) and ESI+ mass spectrometry.
 | ||
| Fig. 7 Structure of compound 7 with the solvent omitted for clarity. Thermal ellipsoids have been drawn at the 50% probability level. Hydrogen atoms are included (not labelled). | ||
Presumably, the Lewis acidic nature of Er(NO3)3·5H2O, and/or the Brønsted acidity of the methanolic solution, catalysed the addition of alcohol to form 7. Pleasingly, simply stirring a methanolic solution of 1 at 40 °C for 24 h (Scheme 2) resulted in formation of 7 in a much-improved yield. The ethanol adduct 8 was obtained via similar means (Scheme 2) and the structure confirmed via X-ray crystallographic analysis (see SI), although only a trace amount of the corresponding adduct was isolated when 1 was stirred with isopropanol. Addition of alcohol across the azafulvene moiety of the aza-BODIPY 1 results in a colour change from deep green to orange, with the absorption maximum at 438 nm (Table 1).26 Furthermore, as a consequence of the demise of the fully conjugated dipyrrinato framework (Fig. S21), the fluorescence emission of 1 is, unsurprisingly, lost upon formation of 7 and 8. Anticipating that emission might be restored upon complexation, the optical behaviour of 7 was studied in the presence of Mn2+, Fe3+, NH4+, Ni2+, Cu2+, Zn2+, Na+, K+, Mg2+, Fe2+, Ca2+, Al3+ and Sb3+. Upon the addition of Zn2+, Sb3+ or Al3+ to a solution of 7 in MeCN, significantly red-shifted absorption properties were observed (Fig. S28); the addition of the other metal ions resulted in less dramatic changes. Intriguingly, addition of solutions of Zn2+ or Sb3+ to a solution of 7 restored emission (Fig. 8) and resulted in maxima at 745 nm (excitation at 600 nm), i.e. bathochromically shifted cf. the aza-BODIPY 1. In contrast, the addition of Al3+ to a solution of 7 resulted in an emission maximum at 660 nm. Addition of the other metal ions effected only insignificant changes in emission or spectral position. Thus, the addition of select metal ions clearly results in complexation within the NNN tridentate pocket, and the –BF2 unit originating with 7 is maintained upon the addition of the metal salts. The solutions containing Zn2+ and Sb3+ both absorb in the near-IR region (λmax,abs = 694 nm for the solution containing 7 and Zn2+; λmax,abs = 684 nm for the solution containing 7 and Sb3+), presumably as a result of enhanced coplanarisation of the aza-dipyrrinato core and the pyridyl units. Impressively, the effect of this planarisation on the absorption maxima is greater than that of the red-shifting observed upon formation of the aza-BODIPY 1 from the aza-dipyrrin 4.
 | ||
| Fig. 8 Emission spectra of solutions of 7 in MeCN (0.13 mM) upon the addition of 20 eq. of metal ions (λex = 600 nm). | ||
Given the NNN-tridentate binding opportunities offered by the two pyridyl units and the meso-nitrogen of the aza-dipyrrinato system, the potential of 7 for metal ion sensing was explored.13,26–31 The addition of increasing concentrations of Zn(ClO4)2 in MeCN to a solution of 7 in MeCN was monitored using absorbance (Fig. 9) and emission (Fig. S30) spectroscopies. As the concentration of Zn2+ increased, emission at 745 nm increased concurrently. The addition of Zn2+ to a solution of 7 in MeCN results in a dramatic enhancement in absorption in the 600–750 nm range (Fig. 9); similar behaviour is observed in the emission characteristics (Fig. S30). Importantly, 7 has negligible absorption and emission in the 600–750 nm range, and so detection of metal ions using 7 constitutes a turn-on sensor. This characteristic also renders 7 a superior metal ion sensor than the fluorescent aza-BODIPY 1.13
 | ||
| Fig. 9 Changes in absorption spectra of a solution of 7 in MeCN (5 μM) upon the addition of Zn(ClO4)2 (0.5–14 eq.) in MeCN. Inset: plot of absorbance at 664 nm as a function of [Zn2+]. | ||
To provide a rationale for the complexation chemistry observed for this system, the geometry of the putative BODIPY complex ion with Zn2+ (i.e.1-Zn2) was optimised using B86bPBE0-XDM/lightdense32 and the ZORA scalar relativistic correction using the FHI-aims program.33 The optimised geometry demonstrated a planar binding arrangement about Zn, with a Zn–Npyridyl bond length of 1.926 Å and Zn–Nmeso bond lengths of 1.921 Å (Fig. 10).
While previous studies have explored the metal-sensing properties of the aza-BODIPY 1,13 there are no reports to date regarding the isolation and characterisation of its complexes. X-ray diffraction analysis of a single crystal grown from the mixture resulting from the reaction of 7 with Zn(ClO4)2 in MeCN revealed the complex 1-Zn(ClO4)2 (Fig. 11). Evidently, the methoxy adduct 7 acts as a proligand, complexing the metal with loss of methanol upon the addition of Zn(ClO4)2. In agreement with the computational predictions, the metal ion is complexed by the neutral tridentate NNN binding pocket of 1. However, the Zn2+ centre was found to be further coordinated by acetonitrile and one perchlorate ion (with the second perchlorate ion remaining uncoordinated), deviating the metal ion from coplanarity with the donor atoms in the NNN pocket. Given that 7 is non-emissive at >600 nm, the sensing ability of the proligand is realised by turn-on emission through in situ generation of the emissive aza-BODIPY motif. Quenching of the reaction mixture containing 1-Zn(ClO4)2 with water and extraction into CH2Cl2 revealed a mild instability to aqueous conditions manifesting as a (partial) loss of the –BF2 unit, which was confirmed crystallographically (see SI).
The aza-dipyrrin 4 also complexes Zn2+ upon reaction with Zn(ClO4)2, again positioning the metal in the neutral tridentate NNN pocket (see SI). Intriguingly, complexation of metals with 4via the NNN pocket leaves the bidentate aza-dipyrrinato binding motif available for further complexation, and enables future exploration of the sensing properties of this versatile pyridyl-appended dipyrrinato framework.
Conclusions
A refined synthetic route to the 3,7-(2-pyridyl)-appended aza-BODIPY 1 is presented herein, accompanied by isolation and characterisation of the parent aza-dipyrrin 4. The chemical reactivity of the aza-dipyrrin 4 and the aza-BODIPY 1 was evaluated, including methylation and protonation of the pyridyl nitrogen atoms. Most importantly, it is demonstrated that 1 can add unhindered alcohols across the azafulvene unit, and that the resulting non-emissive adducts sense metal complexation in a turn-on-emissive manner. Consequently, we report herein the first isolation, characterisation and solid-state structure of a complex featuring a metal ion in the neutral tridentate NNN pocket, which consists of the two pyridyl nitrogen atoms and the meso-nitrogen atom of the aza-dipyrrinato framework. Of further note, complexation of 4via the neutral tridententate NNN binding pocket leaves the bidentate aza-dipyrrinato binding motif available for further complexation, and a variety of potential applications can therefore be envisioned.Author contributions
Conceptualisation: RLG, RD-R and AT. Funding acquisition: AT. Investigation: RLG, RD-R, VAW, MA, MJC, JWH, ERJ and KNR. Project administration: AT. Supervision: AT. Writing – original drafts: RLG and AT. Writing – review & editing: RLG, RD-R, VAW, MA, JWH, ERJ, KNR and AT.Conflicts of interest
There are no conflicts to declare.Data availability
Data supporting this article have been included as part of the supplementary information (SI). Supplementary information: synthesis details and characterisation data. See DOI: https://doi.org/10.1039/d5ob01628b.CCDC 2494132, 2494133 and 2486550–2486557 contain the supplementary crystallographic data for this paper.34a–j
Acknowledgements
We thank Dr Michael Lumsden and Mr Xiao Feng (both at Dalhousie University) for sharing their expertise in NMR spectroscopy and mass spectrometry, respectively. This work was supported, in part, by NSERC of Canada via Discovery Grants and the CREATE Training Program in BioActives (510963). This work was enabled, in part, thanks to funding from the Canada Research Chairs Program (funding reference number 950-232829). This research was enabled, in part, by funding from the Canada Foundation for Innovation (JELF 39824) and Research Nova Scotia (Research Opportunities Fund 2020-1208).References
- R. J. Grams, W. L. Santos, I. R. Scorei, A. Abad-García, C. A. Rosenblum, A. Bita, H. Cerecetto, C. Viñas and M. A. Soriano-Ursúa, Chem. Rev., 2024, 124, 2441–2511 CrossRef CAS PubMed.
- Y. Su and R. Kinjo, Chem. Soc. Rev., 2019, 48, 3613–3659 RSC.
- Z. Huang, S. Wang, R. D. Dewhurst, N. V. Ignat'ev, M. Finze and H. Braunschweig, Angew. Chem., Int. Ed., 2020, 59, 8800–8816 CrossRef CAS PubMed.
- R. L. Gapare and A. Thompson, Chem. Commun., 2022, 58, 7351–7359 RSC.
- C. Schäfer, J. Mony, T. Olsson and K. Börjesson, J. Org. Chem., 2022, 87, 2569–2579 CrossRef.
- A. Bessette and G. S. Hanan, Chem. Soc. Rev., 2014, 43, 3342–3405 RSC.
- J. K. G. Karlsson and A. Harriman, J. Phys. Chem. A, 2016, 120, 2537–2546 CrossRef CAS PubMed.
- Y. Ge and D. F. O'Shea, Chem. Soc. Rev., 2016, 45, 3846–3864 RSC.
- C. Liu, Y. Jin, X. Ji, W. Zhao and X. Dong, Chem. – Eur. J., 2022, 28, e202201503 CrossRef CAS PubMed.
- E. B. Brown, R. L. Gapare, J. W. Campbell, A. Alkaş, S. Sequeira, J. W. Hilborn, S. M. Greening, K. N. Robertson and A. Thompson, Org. Biomol. Chem., 2024, 22, 6122–6128 RSC.
- C. Liu, C. Liu, X. Ji, W. Zhao and X. Dong, J. Med. Chem., 2024, 67, 15908–15924 CrossRef CAS PubMed.
- E. J. González Lopez, A. M. Sarotti and D. A. Heredia, Org. Lett., 2025, 27, 8602–8606 CrossRef.
- A. Coskun, M. D. Yilmaz and E. U. Akkaya, Org. Lett., 2007, 9, 607–609 CrossRef CAS PubMed.
- W. Wu, H. Guo, W. Wu, S. Ji and J. Zhao, J. Org. Chem., 2011, 76, 7056–7064 CrossRef CAS PubMed.
- Y. Chen, J. Zhao, L. Xie, H. Guo and Q. Li, RSC Adv., 2012, 2, 3942–3953 RSC.
- M. D. Ward, Coord. Chem. Rev., 2010, 254, 2634–2642 CrossRef CAS.
- A. D'Aléo, F. Pointillart, L. Ouahab, C. Andraud and O. Maury, Coord. Chem. Rev., 2012, 256, 1604–1620 CrossRef.
- W. Thor, H.-Y. Kai, Y.-H. Yeung, Y. Wu, T.-L. Cheung, L. K. B. Tam, Y. Zhang, L. J. Charbonnière, P. A. Tanner and K.-L. Wong, JACS Au, 2024, 4, 3813–3822 CrossRef CAS PubMed.
- E. Hemmer, N. Venkatachalam, H. Hyodo, A. Hattori, Y. Ebina, H. Kishimoto and K. Soga, Nanoscale, 2013, 5, 11339–11361 RSC.
- A. Rosas-Sánchez, R. A. Toscano, J. G. López-Cortés and M. C. Ortega-Alfaro, Dalton Trans., 2014, 44, 578–590 RSC.
- M. Adib, N. Ayashi, F. Heidari and P. Mirzaei, Synlett, 2016, 1738–1742 CrossRef CAS.
- C. L. Bird and A. T. Kuhn, Chem. Soc. Rev., 1981, 10, 49–82 RSC.
- M. Kathiresan, B. Ambrose, N. Angulakshmi, D. E. Mathew, D. Sujatha and A. M. Stephan, J. Mater. Chem. A, 2021, 9, 27215–27233 RSC.
- B. Ambrose, R. Naresh, S. Deshmukh, M. Kathiresan and P. Ragupathy, Energy Fuels, 2023, 37, 18226–18242 CrossRef CAS.
- A. Iriel, J. M. Novo, G. B. Cordon and M. G. Lagorio, Photochem. Photobiol., 2014, 90, 107–112 CrossRef CAS PubMed.
- F.-Z. Li, Z. Wu, C. Lin, Q. Wang and G.-C. Kuang, Results Chem., 2022, 4, 100384 CrossRef CAS.
- J. Du, J. Fan, X. Peng, H. Li, J. Wang and S. Sun, J. Fluoresc., 2008, 18, 919–924 CrossRef CAS PubMed.
- X. Peng, J. Du, J. Fan, J. Wang, Y. Wu, J. Zhao, S. Sun and T. Xu, J. Am. Chem. Soc., 2007, 129, 1500–1501 CrossRef CAS PubMed.
- L. Zeng, E. W. Miller, A. Pralle, E. Y. Isacoff and C. J. Chang, J. Am. Chem. Soc., 2006, 128, 10–11 CrossRef CAS PubMed.
- B. Wang, P. Li, F. Yu, P. Song, X. Sun, S. Yang, Z. Lou and K. Han, Chem. Commun., 2013, 49, 1014–1016 RSC.
- Y. Wu, X. Peng, B. Guo, J. Fan, Z. Zhang, J. Wang, A. Cui and Y. Gao, Org. Biomol. Chem., 2005, 3, 1387–1392 RSC.
- A. J. A. Price, A. Otero-de-la-Roza and E. R. Johnson, Chem. Sci., 2023, 14, 1252–1262 RSC.
- V. Blum, R. Gehrke, F. Hanke, P. Havu, V. Havu, X. Ren, K. Reuter and M. Scheffler, Comput. Phys. Commun., 2009, 180, 2175–2196 CrossRef CAS.
- (a) CCDC 2494132: Experimental Crystal Structure Determination, 2025, DOI:10.5517/ccdc.csd.cc2pqbwn; (b) CCDC 2494133: Experimental Crystal Structure Determination, 2025, DOI:10.5517/ccdc.csd.cc2pqbxp; (c) CCDC 2486550: Experimental Crystal Structure Determination, 2025, DOI:10.5517/ccdc.csd.cc2pgg9y; (d) CCDC 2486551: Experimental Crystal Structure Determination, 2025, DOI:10.5517/ccdc.csd.cc2pggbz; (e) CCDC 2486552: Experimental Crystal Structure Determination, 2025, DOI:10.5517/ccdc.csd.cc2pggc0; (f) CCDC 2486553: Experimental Crystal Structure Determination, 2025, DOI:10.5517/ccdc.csd.cc2pggd1; (g) CCDC 2486554: Experimental Crystal Structure Determination, 2025, DOI:10.5517/ccdc.csd.cc2pggf2; (h) CCDC 2486555: Experimental Crystal Structure Determination, 2025, DOI:10.5517/ccdc.csd.cc2pggg3; (i) CCDC 2486556: Experimental Crystal Structure Determination, 2025, DOI:10.5517/ccdc.csd.cc2pggh4; (j) CCDC 2486557: Experimental Crystal Structure Determination, 2025, DOI:10.5517/ccdc.csd.cc2pggj5.
| This journal is © The Royal Society of Chemistry 2025 |