Open Access Article
Open Access ArticleDigold photosensitizer for visible light [2 + 2] cycloaddition and E/Z isomerization reactions
Tommaso
Ruggiero
a,
James
Fortwengler
bc,
Kristof
Van Hecke
 a,
Mark E.
Thompson
a,
Mark E.
Thompson
 bc,
Thomas S. A.
Heugebaert
bc,
Thomas S. A.
Heugebaert
 d and
Steven P.
Nolan
d and
Steven P.
Nolan
 *a
*a
aDepartment of Chemistry and Center for Sustainable Chemistry, Ghent University, Krijgslaan 289, building S-3, 9000 Ghent, Belgium. E-mail: steven.nolan@ugent.be
bMork Family Department of Chemical Engineering and Materials Science, University of Southern California, Los Angeles, California 90089, USA
cDepartment of Chemistry, University of Southern California, Los Angeles, California 90089, USA. E-mail: met@usc.edu
dDepartment of Green Chemistry and Technology, Ghent University, Coupure Links 653, 9000 Ghent, Belgium. E-mail: thomas.heugebaert@ugent.be
First published on 17th October 2025
Abstract
Energy transfer photocatalysis has emerged as a powerful platform for a wide variety of transformations, yet advances in this area are often limited by the visible-light absorption of catalysts and the very high cost of state-of-the-art iridium-based sensitizers. We report the synthesis, characterization, and photocatalytic use of a novel digold(I) complex, {[Au(IPr)]2(DHIC)} (PhotAu3), designed to overcome these limitations by absorbing visible light in the blue region and proving catalytically effective under 450 nm LED irradiation. The complex was successfully applied to a range of EnT-mediated photocatalytic reactions, including intramolecular [2 + 2] cycloadditions and E/Z isomerizations, achieving high conversions at low catalyst loadings in short reaction times.
Introduction
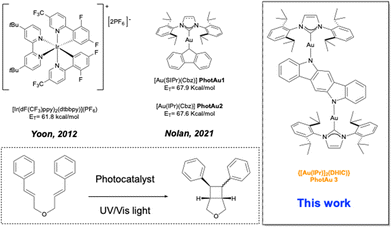
Over the past fifteen years, the area of photocatalysis has experienced remarkable growth, becoming a powerful synthetic tool enabling efficient late-stage functionalization and the construction of very diverse molecular frameworks.1–4 While photoredox catalysis via electron transfer (ET) has largely dominated the field,5–7 energy transfer (EnT) catalysis, particularly through a triplet–triplet energy transfer (TTEnT) mechanism,8 has recently gained attention as a promising strategy for molecular assembly.9–12 Although a wide variety of organosensitizers,13–17 including ruthenium,18–20 and other transition metal-based photocatalysts21–24 have been explored in energy transfer (EnT) photocatalysis, iridium complexes remain the state-of-the-art9,25 due to their exceptionally long excited triplet-state lifetimes, high quantum yields, tunable ligand frameworks, and, most notably, their high triplet energy (Et) values that permit excitation of a broad range of organic substrates.26 Despite recent advances where Et values of up to 73 kcal mol−1 have been achieved,27,28 the majority of Ir-based photosensitizers still fall below the 64 kcal mol−1 threshold, thereby oftentimes limiting substrate scope. Furthermore, their complex multi-step syntheses,29 high cost,30 limited compatibility with green solvents due to their ionic nature, and poor recyclability31,32 collectively highlight the pressing need for more sustainable, cost-effective, and efficient alternatives. As a more cost-effective and synthetically accessible alternative to iridium-based photosensitizers, Nolan and co-workers have developed and made commercially available33 the gold carbazolyl complexes [Au(SIPr)(Cbz)] (PhotAu1) (SIPr = [N,N-bis(2,6-diisopropylphenyl)imidazolin-2-ylidene]; Cbz = carbazolyl) and [Au(IPr)(Cbz)] (PhotAu2) (IPr = [N,N-bis(2,6-diisopropylphenyl)imidazol-2-ylidene]) which exhibit high triplet energies (67.9 and 67.6 kcal mol−1, respectively), remarkably long lifetimes in solution (266 and 335 μs respectively), and intense energy emission, enabling previously inaccessible energy transfer (EnT) transformations such as the intramolecular [2 + 2] cycloaddition of unsubstituted indoles in notably short reaction times.34,35 The successful use of these organogold complexes in additional EnT photocatalytic processes further demonstrates their potential and versatility. These have shown effective in the intermolecular [2 + 2] cycloaddition of coumarins,36 isooxazolines37 and benzothiophenes38 with various alkenes, the cyclization of acrylamides39 and in the synthesis of cycloheptaindoles.40Dinuclear gold(I) complexes have only recently been studied in catalysis,41,42 materials science43–45 and as promising anticancer agents.46,47 Dinuclear amido-bearing NHC complexes for photocatalysis have few precedents, and this synthetic challenge represents an opportunity to explore the fundamental question of synergistic bimetallic interactions and their impact on photocatalytic behavior and reactivity. In this regard, novel dinuclear [(bisNHC){Au(Cbz)}2] complexes have been described.48 Investigation of their photocatalytic activity and photophysical properties revealed that, while their properties resemble those of [Au(SIPr)(Cbz)], the NHC-Au-Cbz fragments in the binuclear complexes appear to operate independently from one another in solution without the apparent existence of cooperative interaction. Additionally, binuclear Janus-type and tandem-NHC gold complexes reported by Thompson and coworkers exhibit thermally activated delayed fluorescence (TADF), although they have not been employed as sensitizers, and their synthesis remains challenging.49,50 A major limitation of the new gold complexes PhotAu1 and PhotAu2 is the position of their absorbance in the UV region. Irradiation in this section of the spectrum can lead to photochemical initiation of reactions without the need for a sensitizer.34 Thus far, only light sources emitting at 365 nm have been successfully used in experiments conducted by the Nolan group with gold sensitizers. In contrast, iridium-based photocatalysts can operate with 450 nm irradiation, utilizing visible blue light, which is generally considered a milder and more selective condition than UV light irradiation. The use of visible light is preferred due to its lower energy consumption, safer handling, and reduced potential for degradation of both the starting material and products, as well as a lower likelihood of side reactions and byproduct formation.51–53 To overcome these issues, we sought to design a gold photocatalyst that could operate (like iridium complexes) in this region of the visible light spectrum. Here, we report the facile synthesis of a novel gold sensitizer [Au(IPr)]2(DHIC) (PhotAu3) and its catalytic use in [2 + 2] cycloaddition and E/Z isomerization reactions (Scheme 1).
Results and discussion
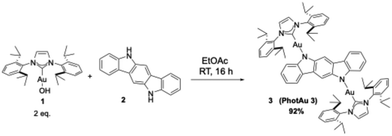
Inspired by the catalytic behavior of amido-complexes and the efficiency of carbazole in these key photocatalytically active architectures, 5,11-dihydroindolo[3,2-b]carbazole (2) was employed as a new nitrogen donor. The initial synthetic approach involved the weak base route with [Au(IPr)(Cl)], K2CO3 and 2 (see SI). Unfortunately, this first attempt proved unsuccessful. We reasoned that an alternative gold nucleophilic synthon could be deployed to form the amido–gold bond. To this end, [Au(IPr)(OH)] (1)54 was employed and cleanly led to the formation of PhotAu3 (3) in ethyl acetate at room temperature, according to Scheme 2. The excellent solubility of 3 in THF permitted full spectroscopic characterization as well as the growth of single crystals, suitable for X-ray diffraction study. Photoluminescent spectroscopy was also possible in this solvent. The crystal structure of PhotAu3 exhibits an asymmetric unit, consisting of two halves of symmetry-independent molecules, with different spatial disposition of the NHC and amido fragments (Fig. 1). One molecule (Fig. 1, left) presents the NHC moiety and the donor ligand on the same plane, while the other shows the two fragments lying perpendicular to one another. The second molecular conformation is similar to the one exhibited by PhotAu1.35 Interestingly, other similar molecular structures bearing IPr show conformations with the two fragments set on the same plane.55–58 Absorption spectra of the free ligand 2 with that of PhotAu3, measured in THF solution, show that the last absorption band of 5,11-dihydroindolo[3,2-b]carbazole ends at around 420 nm, with the last peak at 405 nm (see SI). By adding the IPr-Au moiety to the donor ligand, the number of π-conjugated orbitals increases, thus red-shifting the absorption band of the complex: for PhotAu3 two new bands are present with maximum absorption at 400, 440, and 470 nm, still absorbing up to 480 nm, indicating the ability to absorb from a standard blue LED at 450 nm.The PL emission spectrum shows a peak of λmax = 562 nm, which indicates that PhotAu3 has an ET value of ca. 50.9 kcal mol−1. This value is in line with reactivity found using the catalyst, which proved unreactive with substrates possessing higher values than 60 kcal mol−1. The PLQY reported for the catalyst is 3% and the lifetime of the photoluminescence is 39 μs in THF (Table 1).
| λ max | Φ PLQY | τ (μs) | k r (103 s−1) | k nr (105 s−1) |
|---|---|---|---|---|
| 562 | 3% | 39 | 7.7 | 2.5 |
Both of those values are inferior compared to those reported for PhotAu1 and PhotAu2. The low kr is most likely a result of emission from a ligand locally excited triplet state (3LE). The vibronic fine structure seen in the emission band is characteristic of LC-based emission, whereas CT-based emission is typically broad and featureless. To further support the 3LC assignment, the phosphorescence spectrum of PhotAu3 in 2Me-THF was collected at 77 K. The 77 K spectrum and the room temperature spectra are the same, consistent with the emission at both temperatures coming from the 3LE state. We have found in previous (carbene)M(amido) complexes that cooling the sample in a polar solvent destabilizes the CT and often gives rise to 3LC emission.57,59–62 Here the 3LC emission is observed at both RT and 77 K. The gold is facilitating rapid intersystem crossing from the 1ICT to the 3ICT, which internally converts to the 3LE state. The involvement of the 3LE state leads to the long-measured lifetime, which is beneficial for photocatalysis requiring diffusion of the substrate to the excited catalyst (Fig. 2).
 | ||
| Fig. 2 Molar absorptivity of the DHIC ligand and PhotAu3 in THF, as well as photoluminescence spectra at room temperature of PhotAu3 in THF and at 77 K in 2-MeTHF are shown. | ||
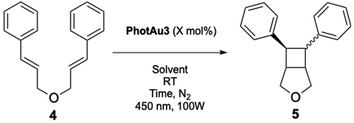
Our new photosensitizer was initially tested in the intramolecular [2 + 2] cycloaddition of substrates that have previously been investigated using iridium catalysts and our gold photosensitizers. Cinnamyl ether 4 was initially selected as the model substrate.34,35,48 Compound 4 has previously been tested by Yoon and coworkers as a benchmark for EnT catalysis.63 In their seminal work, 1 mol% of the [Ir(dF(CF3)ppy)2(dtbbpy)][PF6] sensitizer was used and the reaction proceeded in very dilute DMSO solution (0.01 M). Ruthenium sensitizers were unable to mediate this reaction as triplet energies of styrenes are estimated to be ca. 60 kcal mol−1.64
Optimization focused mainly on decreasing the catalyst loading and reaction time while confirming that visible light could facilitate the reaction. We employed 450 nm irradiation wavelength at 100 W. Surprisingly, full conversion and a yield of 96% were obtained after 30 minutes with 0.5 mol% of the catalyst in both THF and 2Me-THF (entries 2 and 3, Table 2). By decreasing the catalyst loading to 0.25 mol%, a conversion of 72% was reached after 1 h and 83% after 2 h (entries 4 and 5, Table 2). Control experiments confirmed both the need for light and for photosensitizer (entries 6 and 7, Table 2). To confirm the need for gold, compound 2 (the carbazole ligand alone) was used as photocatalyst in a 10 mol% amount (entry 8, Table 2). At the 450 nm irradiation for 6 hours, a poor 23% conversion was obtained. These catalytic data agree with expected reactivity based on the absorption spectra of both catalyst and ligand 5,11-dihydroindolo[3,2-b]carbazole.
| Entry | Mol% | Solvent | Time (h) | Conv. (%) | Yield (%) |
|---|---|---|---|---|---|
| a General reaction conditions: 4 (0.1 mmol, 1 eq.), PhotAu3 (0.5–0.25 mol%), THF or 2Me-THF, room temperature, 0.5–6 h, under nitrogen, under 450 nm light irradiation at 100 W. NMR conversion and yield are calculated with 1,3,5-trimethoxybenzene as internal standard as average of two runs. b Isolated yield. c Reaction performed in the dark. d 5,11-Dihydroindolo[3,2-b]carbazole used as catalyst. e Benzophenone used as catalyst. f Thioxantone used as catalyst. | |||||
| 1 | 0.5 | THF | 1 | 100 | 98 |
| 2 | 0.5 | THF | 0.5 | 100 | 96 |
| 3 | 0.5 | 2Me-THF | 0.5 | 100 | 96 (90) |
| 4 | 0.25 | THF | 1 | 72 | 68 |
| 5 | 0.25 | THF | 2 | 83 | 73 |
| 6 | 0 | THF | 6 | 0 | 0 |
| 7 | 0.5c | THF | 6 | 0 | 0 |
| 8 | 10d | THF | 6 | 23 | 17 |
| 9 | 4e | THF | 1 | 0 | 0 |
| 10 | 4f | THF | 1 | 15 | 9 |
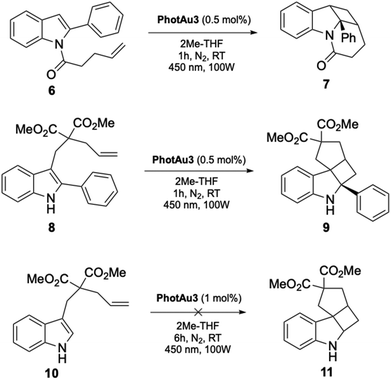
An additional family of spiroindoline precursor substrates was examined. This series has previously been examined by You,65–69 Fu,70 Koenig16 and Oderinde.71 Polycyclic spiroindolines are privileged scaffolds found in numerous naturally occurring alkaloids.72 In order to generate the products by photocatalysis, it is often necessary to pre-functionalize the precursors, as branched indoles, with the introduction of an electron-withdrawing group on the C2 of the indole, since this functionalization has been shown to significantly lower the excitation threshold of the precursor molecule, thereby facilitating the photochemical transformation.65 Keeping in mind the exciting results brought by compound 4, the optimization of intramolecular [2 + 2] cycloaddition of tethered indoles at low catalyst loading and short reaction times was investigated (Table 2).
Both substrates 6 and 8 reached full conversion after 1 hour with 0.5 mol% of catalyst (1 mol% gold). Once again THF and 2Me-THF were selected as solvents to fully solubilize the sensitizer. Interestingly but not surprisingly, compound 10, without the phenyl group at C2, did not react under these conditions as it possesses an ET value of 65 kcal mol−1 (ref. 61) which is well above the value at which the photocatalyst can operate (Scheme 3, see SI for reaction conditions).
The E/Z isomerization of double bonds via EnT photocatalysis was next explored. The synthesis of Z-cinnamyl ethers, their corresponding alcohols and, more generally, Z-alkenes present a significant challenge. Various conceptually distinct methodologies have been developed to tackle this challenge including Wittig reactions,73 cross-coupling of alkenyl halides,74 stereoselective semi-hydrogenation of alkynes,75Z-selective olefin metathesis,76 and Peterson olefination reactions.77 However, these reported transformations generally suffer from limited selectivity for the Z-configuration and often depend on energy-intensive reagents or costly transition metal-based catalysts.
Therefore, an alternative strategy that enables the synthesis of the Z-isomer from the more accessible E-isomer, while allowing diversification from a common and easily available intermediate, is highly desirable. Various types of photosensitizers have been used recently for this purpose, obtaining the Z isomer from its E counterpart through E/Z isomerization. Those photocatalysts include iridium-based triplet photosensitizers,78–80 organic dyes such as riboflavin,81 aromatic ketones like 2-iodo-9-fluorenone,82 and covalent organic frameworks (COFs).83,84 Our first substrate candidate was cinnamyl alcohol (12) (Scheme 4), which has been reported by Wang and coworkers to yield the corresponding Z isomer with a ratio of Z/E = 81/19 in a system with state-of-the-art fac-Ir(ppy)3 iridium sensitizer at 1 mol% in THF with 24 h of blue LED irradiation.85 The experiments, reported in Table 3, show that reaction times longer than 2 hours were unnecessary to reach high Z/E ratio (entries 1, 2 and 3). A 0.1 M solution showed the same result as that of 0.2 M (entry 4). A catalyst loading of 0.5 mol% allowed Z/E ratios of 74/26 (entry 7). These findings suggest an equilibrium-based reaction. Similar results were observed for the stilbene substrate (14) (Scheme 4), which reached a Z/E ratio of 79/21 with 0.5 mol% catalyst in just 1 hour. Reactions performed in THF proved better than those conducted in Me-THF in both cases.
| Entry | Mol% | Solvent | Time (h) | Conc. (M) | Z/E |
|---|---|---|---|---|---|
| a General reaction conditions: 15 (0.2 mmol, 1 eq.), PhotAu3 (0.25–2.0 mol%), THF or 2Me-THF, room temperature, 1–16 h, under nitrogen, under 450 nm light irradiation at 100 W. Z/E ratios were confirmed by NMR spectroscopy, average of two runs. b Reaction performed in the dark. c Benzophenone used as catalyst. d Thioxanthone used as catalyst. | |||||
| 1 | 1 | THF | 16 | 0.2 | 74/26 |
| 2 | 1 | THF | 6 | 0.2 | 74/26 |
| 3 | 1 | THF | 2 | 0.2 | 74/26 |
| 4 | 1 | THF | 2 | 0.1 | 74/26 |
| 5 | 1 | THF | 1 | 0.2 | 70/30 |
| 6 | 2 | THF | 1 | 0.2 | 70/30 |
| 7 | 0.5 | THF | 2 | 0.2 | 74/26 |
| 8 | 0.5 | 2Me-THF | 2 | 0.2 | 56/44 |
| 9 | 0.25 | THF | 2 | 0.2 | 27/73 |
| 10 | 0 | THF | 2 | 0.2 | 0/100 |
| 11 | 0.5b | THF | 2 | 0.2 | 0/100 |
| 12 | 4c | THF | 2 | 0.2 | 0/100 |
| 13 | 4d | THF | 2 | 0.2 | 23/77 |
A recent report disclosed the application of a new organic sensitizer that is able to yield Z-stilbene with Z/E = 74/26,86 while with state-of-the-art iridium catalysts it is possible to reach Z/E ratios of 99/1.87 Control experiments confirmed that for the two substrates examined with PhotAu3, both 450 nm irradiation and the sensitizer are necessary for the reaction to occur (entries 10 and 11, Table 3). To complete this section of the study, the isomerization reactions were carried out with common and commercially available photosensitizers such as benzophenone88 and thioxanthone,89 which have been used extensively in a variety of photochemical transformations.90
These two photocatalysts proved completely ineffective in the isomerization reactions. This can be easily understood as the absorbance band of benzophenone does not go above ca. 380 nm while thioxanthone absorbs around 410 nm.91 The results shown in Tables 2 and 3 confirm the inefficacy of benzophenone in reactions involving 4 and 12. Since thioxanthone can absorb a portion of visible light emitted, we were able to reach 15% conversion with 4 as substrate and 23/77 Z/E ratio for cinnamyl alcohol with 4 mol% of catalyst loading. PhotAu3 proves significantly more effective.
Conclusions
A simple synthetic protocol to access a new digold photosensitizer, which has been fully characterized by spectroscopic and X-ray diffraction studies on a single crystal, has been established. This new photocatalyst is the first gold complex to be employed successfully for energy transfer photocatalysis under visible light irradiation and proves a viable alternative to blue light absorbing state-of-the-art iridium sensitizers. PhotAu3 has been deployed as catalyst in three different intramolecular [2 + 2] cycloadditions, and two E/Z isomerizations at low catalyst loading and proved effective in short reaction times. Further studies involving extensive reaction scope in various transformations using PhotAu3 are ongoing. The evolution of the synthetic design of gold complexes for photocatalysis is being further explored in our laboratories.Author contributions
T. R.: conceptualization, investigation and synthesis and catalysis experiments; characterization NMR and/or SC-XRD; writing and editing of the manuscript. J. F.: photophysical analysis and characterization of the catalyst and writing part of the manuscript on the topic. K. V. H.: review and editing of manuscript; secured funding. M. E. T.: supervision of the work; writing and editing of the manuscript. T. S. A. H.: supervision of the work; writing and editing of the manuscript; secured funding. S. P. N.: conceptualization, supervision of the project and work; writing and editing of the manuscript; secured funding.Conflicts of interest
There are no conflicts to declare.Data availability
Additional data supporting this article have been included as part of the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d5ob01503k.CCDC 2465569 contains the supplementary crystallographic data for this paper.92
Acknowledgements
We gratefully acknowledge support from the Special Research Fund (BOF/24J/2023/084 to K. V. H.) of Ghent University, the Special Research Fund (BOF/STA/202102/003 to T. S. A. H.) of Ghent University and The Research Foundation – Flanders (FWO) (G0A6823N to S. P. N). Umicore AG is thanked for generous gifts of materials.References
- L. Marzo, S. K. Pagire, O. Reiser and B. König, Angew. Chem., Int. Ed., 2018, 57, 10034–10072 CrossRef CAS PubMed.
- B. König, Eur. J. Org. Chem., 2017, 1979–1981 CrossRef.
- N. Corrigan, S. Shanmugam, J. Xu and C. Boyer, Chem. Soc. Rev., 2016, 45, 6165–6212 RSC.
- T. P. Yoon, M. A. Ischay and J. Du, Nat. Chem., 2010, 2, 527–532 CrossRef CAS PubMed.
- N. A. Romero and D. A. Nicewicz, Chem. Rev., 2016, 116, 10075–10166 CrossRef CAS PubMed.
- S. P. Pitre and L. E. Overman, Chem. Rev., 2022, 122, 1717–1751 CrossRef CAS PubMed.
- M. Reckenthäler and A. G. Griesbeck, Adv. Synth. Catal., 2013, 355, 2727–2744 CrossRef.
- F. Strieth-Kalthoff and F. Glorius, Chem, 2020, 6, 1888–1903 CAS.
- Q.-Q. Zhou, Y.-Q. Zou, L.-Q. Lu and W.-J. Xiao, Angew. Chem., Int. Ed., 2019, 58, 1586–1604 CrossRef CAS PubMed.
- J. Großkopf, T. Kratz, T. Rigotti and T. Bach, Chem. Rev., 2022, 122, 1626–1653 CrossRef PubMed.
- F. Strieth-Kalthoff, M. J. James, M. Teders, L. Pitzer and F. Glorius, Chem. Soc. Rev., 2018, 47, 7190–7202 RSC.
- S. Dutta, J. E. Erchinger, F. Strieth-Kalthoff, R. Kleinmans and F. Glorius, Chem. Soc. Rev., 2024, 53, 1068–1089 RSC.
- W. C. de Souza, B. T. Matsuo, P. M. Matos, J. T. M. Correia, M. S. Santos, B. König and M. W. Paixão, Chem. – Eur. J., 2021, 27, 3722–3728 CrossRef CAS PubMed.
- X. Li, R. J. Kutta, C. Jandl, A. Bauer, P. Nuernberger and T. Bach, Angew. Chem., Int. Ed., 2020, 59, 21640–21647 CrossRef CAS PubMed.
- A. Hölzl-Hobmeier, A. Bauer, A. V. Silva, S. M. Huber, C. Bannwarth and T. Bach, Nature, 2018, 564, 240–243 CrossRef PubMed.
- A. B. Rolka and B. Koenig, Org. Lett., 2020, 22, 5035–5040 CrossRef CAS PubMed.
- E. Pylova, B. Lasorne, N. D. McClenaghan, G. Jonusauskas, M. Taillefer, S. N. Konchenko, A. Prieto and F. Jaroschik, Chem. – Eur. J., 2024, 30, e202401851 CrossRef CAS PubMed.
- T. R. Blum, Z. D. Miller, D. M. Bates, I. A. Guzei and T. P. Yoon, Science, 2016, 354, 1391–1395 CrossRef CAS PubMed.
- F. M. Hörmann, C. Kerzig, T. S. Chung, A. Bauer, O. S. Wenger and T. Bach, Angew. Chem., 2020, 132, 9746–9755 CrossRef.
- Y.-Q. Zou, S.-W. Duan, X.-G. Meng, X.-Q. Hu, S. Gao, J.-R. Chen and W.-J. Xiao, Tetrahedron, 2012, 68, 6914–6919 CrossRef CAS.
- P.-K. Chow, G. Cheng, G. S. M. Tong, C. Ma, W.-M. Kwok, W.-H. Ang, C. Y.-S. Chung, C. Yang, F. Wang and C.-M. Che, Chem. Sci., 2016, 7, 6083–6098 RSC.
- N. Hu, H. Jung, Y. Zheng, J. Lee, L. Zhang, Z. Ullah, X. Xie, K. Harms, M.-H. Baik and E. Meggers, Angew. Chem., Int. Ed., 2018, 57, 6242–6246 CrossRef CAS PubMed.
- M. Beliaeva, A. Belyaev, E. V. Grachova, A. Steffen and I. O. Koshevoy, J. Am. Chem. Soc., 2021, 143, 15045–15055 CrossRef CAS PubMed.
- C. Cruché, W. Neiderer and S. K. Collins, ACS Catal., 2021, 11, 8829–8836 CrossRef.
- M. Bera, H. S. Hwang, T.-W. Um, S. M. Oh, S. Shin and E. J. Cho, Org. Lett., 2022, 24, 1774–1779 CrossRef CAS PubMed.
- D. Jespersen, B. Keen, J. I. Day, A. Singh, J. Briles, D. Mullins and J. D. I. Weaver, Org. Process Res. Dev., 2019, 23, 1087–1095 CrossRef CAS PubMed.
- L. Schmid, F. Glaser, R. Schaer and O. S. Wenger, J. Am. Chem. Soc., 2022, 144, 963–976 CrossRef CAS PubMed.
- T. O. Paulisch, L. A. Mai, F. Strieth-Kalthoff, M. J. James, C. Henkel, D. M. Guldi and F. Glorius, Angew. Chem., Int. Ed., 2022, 61, e202112695 CrossRef CAS PubMed.
- M. S. Lowry, J. I. Goldsmith, J. D. Slinker, R. Rohl, R. A. Pascal, G. G. Malliaras and S. Bernhard, Chem. Mater., 2005, 17, 5712–5719 CrossRef CAS.
- In February 2023, Umicore reports an average price of 29.6 € per mmol for Ir (https://pmm.umicore.com/en/prices/iridium/), and 12.1 € per mmol for gold (https://pmm.umicore.com/en/prices/gold/).
- Z.-Y. Xu, Y. Luo, D.-W. Zhang, H. Wang, X.-W. Sun and Z.-T. Li, Green Chem., 2020, 22, 136–143 RSC.
- J. Ma, F. Strieth-Kalthoff, T. Dalton, M. Freitag, J. L. Schwarz, K. Bergander, C. Daniliuc and F. Glorius, Chem, 2019, 5, 2854–2864 CAS.
- Strem catalogue numbers: 79-0000 [Au(IPr)(Cbz)]; 79-0002 [Au(SIPr)(Cbz)].
- E. A. Martynova, V. A. Voloshkin, S. G. Guillet, F. Bru, M. Beliš, K. V. Hecke, C. S. J. Cazin and S. P. Nolan, Chem. Sci., 2022, 13, 6852–6857 RSC.
- N. V. Tzouras, E. A. Martynova, X. Ma, T. Scattolin, B. Hupp, H. Busen, M. Saab, Z. Zhang, L. Falivene, G. Pisanò, K. Van Hecke, L. Cavallo, C. S. J. Cazin, A. Steffen and S. P. Nolan, Chem. – Eur. J., 2021, 27, 11904–11911 CrossRef CAS PubMed.
- V. A. Voloshkin, M. Villa, E. A. Martynova, M. Beliš, K. V. Hecke, P. Ceroni and S. P. Nolan, Chem. Sci., 2024, 15, 4571–4580 RSC.
- S. G. Guillet, A. A. Logvinov, V. A. Voloshkin, E. A. Martynova and S. P. Nolan, Org. Lett., 2023, 25, 1403–1408 CrossRef CAS PubMed.
- E. A. Martynova, V. V. Voloshkin, M. Villa, A. Fiorentino, M. Beliš, K. V. Hecke, P. Ceroni and S. P. Nolan, J. Catal., 2025, 442, 115850 CrossRef CAS.
- Y. Zhao, S. G. Guillet, V. A. Voloshkin and S. P. Nolan, Org. Lett., 2024, 26, 10322–10327 CrossRef CAS PubMed.
- Y. Zhao, V. A. Voloshkin, E. A. Martynova, B. Maity, L. Cavallo and S. P. Nolan, Chem. Commun., 2024, 60, 3174–3177 RSC.
- T. A. C. A. Bayrakdar, F. Nahra, D. Ormerod and S. P. Nolan, J. Chem. Technol. Biotechnol., 2021, 96, 3371–3377 CrossRef.
- T. A. C. A. Bayrakdar, F. Nahra, J. V. Davis, M. M. Gamage, B. Captain, M. Temprado, M. Marazzi, M. Saab, K. V. Hecke, D. Ormerod, C. D. Hoff and S. P. Nolan, Organometallics, 2020, 39, 2907–2916 CrossRef.
- R. W. Y. Man, C.-H. Li, M. W. A. MacLean, O. V. Zenkina, M. T. Zamora, L. N. Saunders, A. Rousina-Webb, M. Nambo and C. M. Crudden, J. Am. Chem. Soc., 2018, 140, 1576–1579 CrossRef CAS PubMed.
- H. Shen, S. Xiang, Z. Xu, C. Liu, X. Li, C. Sun, S. Lin, B. K. Teo and N. Zheng, Nano Res., 2020, 13, 1908–1911 CrossRef CAS.
- M. Jin, R. Ando, M. J. Jellen, M. A. Garcia-Garibay and H. Ito, J. Am. Chem. Soc., 2021, 143, 1144–1153 CrossRef CAS PubMed.
- N. Mirzadeh, M. A. Bennett and S. K. Bhargava, Coord. Chem. Rev., 2013, 257, 2250–2273 CrossRef CAS.
- N. Mirzadeh, T. S. Reddy and S. K. Bhargava, Coord. Chem. Rev., 2019, 388, 343–359 CrossRef CAS.
- X. Ma, V. A. Voloshkin, E. A. Martynova, M. Beliš, M. Peng, M. Villa, N. V. Tzouras, W. Janssens, K. V. Hecke, P. Ceroni and S. P. Nolan, Catal. Sci. Technol., 2023, 13, 4168–4175 RSC.
- J. Ma, J. Schaab, S. Paul, S. R. Forrest, P. I. Djurovich and M. E. Thompson, J. Am. Chem. Soc., 2023, 145, 20097–20108 CrossRef CAS PubMed.
- T. Li, D. G. Shlian, P. I. Djurovich and M. E. Thompson, Chem. – Eur. J., 2021, 27, 6191–6197 CrossRef CAS PubMed.
- S. Poplata, A. Tröster, Y.-Q. Zou and T. Bach, Chem. Rev., 2016, 116, 9748–9815 CrossRef CAS PubMed.
- N. Hoffmann, Synthesis, 2004, 481–495 CrossRef CAS.
- M. T. Crimmins and T. L. Reinhold, in Organic Reactions, John Wiley & Sons, Ltd, 2004, pp. 297–588 Search PubMed.
- S. Gaillard, A. M. Z. Slawin and S. P. Nolan, Chem. Commun., 2010, 46, 2742–2744 RSC.
- S. Heo, B. H. Jhun, S. Woo, H. S. Kim, I. Kim, J. Kim, W. Son, Y. Jung, K. Kim, J. Cho and Y. You, Adv. Opt. Mater., 2024, 12, 2302148 CrossRef CAS.
- I. Ibni Hashim, N. V. Tzouras, W. Janssens, T. Scattolin, L. Bourda, S. Bhandary, K. Van Hecke, S. P. Nolan and C. S. J. Cazin, Chem. – Eur. J., 2022, 28, e202201224 CrossRef CAS PubMed.
- R. Hamze, M. Idris, D. S. Muthiah Ravinson, M. C. Jung, R. Haiges, P. I. Djurovich and M. E. Thompson, Front. Chem., 2020, 8, 401 CrossRef CAS PubMed.
- J. Ma, J. Schaab, P. Djurovich and M. E. Thompson, chemrxiv, 2023, preprint, chemrxiv:2023-03j7h, DOI:10.26434/chemrxiv-2023-03j7h.
- R. Hamze, J. L. Peltier, D. Sylvinson, M. Jung, J. Cardenas, R. Haiges, M. Soleilhavoup, R. Jazzar, P. I. Djurovich, G. Bertrand and M. E. Thompson, Science, 2019, 363, 601–606 CrossRef CAS PubMed.
- R. Hamze, S. Shi, S. C. Kapper, D. S. Muthiah Ravinson, L. Estergreen, M.-C. Jung, A. C. Tadle, R. Haiges, P. I. Djurovich, J. L. Peltier, R. Jazzar, G. Bertrand, S. E. Bradforth and M. E. Thompson, J. Am. Chem. Soc., 2019, 141, 8616–8626 CrossRef CAS PubMed.
- S. Shi, M. C. Jung, C. Coburn, A. Tadle, M. R. Daniel Sylvinson, P. I. Djurovich, S. R. Forrest and M. E. Thompson, J. Am. Chem. Soc., 2019, 141, 3576–3588 CrossRef CAS PubMed.
- T.-Y. Li, S.-J. Zheng, P. I. Djurovich and M. E. Thompson, Chem. Rev., 2024, 124, 4332–4392 CrossRef CAS PubMed.
- Z. Lu and T. P. Yoon, Angew. Chem., Int. Ed., 2012, 51, 10329–10332 CrossRef CAS PubMed.
- T. Ni, R. A. Caldwell and L. A. Melton, J. Am. Chem. Soc., 1989, 111, 457–464 CrossRef CAS.
- M. Zhu, C. Zheng, X. Zhang and S.-L. You, J. Am. Chem. Soc., 2019, 141, 2636–2644 CrossRef CAS PubMed.
- M. Zhu, X. Zhang, C. Zheng and S.-L. You, ACS Catal., 2020, 10, 12618–12626 CrossRef CAS.
- M. Zhu, H. Xu, X. Zhang, C. Zheng and S.-L. You, Angew. Chem., Int. Ed., 2021, 60, 7036–7040 CrossRef CAS PubMed.
- M. Zhu, X.-L. Huang, S. Sun, C. Zheng and S.-L. You, J. Am. Chem. Soc., 2021, 143, 13441–13449 CrossRef CAS PubMed.
- M. Zhu, X.-L. Huang, H. Xu, X. Zhang, C. Zheng and S.-L. You, CCS Chem., 2020, 3, 652–664 CrossRef.
- Z. Zhang, D. Yi, M. Zhang, J. Wei, J. Lu, L. Yang, J. Wang, N. Hao, X. Pan, S. Zhang, S. Wei and Q. Fu, ACS Catal., 2020, 10, 10149–10156 CrossRef CAS.
- M. S. Oderinde, E. Mao, A. Ramirez, J. Pawluczyk, C. Jorge, L. A. M. Cornelius, J. Kempson, M. Vetrichelvan, M. Pitchai, A. Gupta, A. K. Gupta, N. A. Meanwell, A. Mathur and T. G. M. Dhar, J. Am. Chem. Soc., 2020, 142, 3094–3103 CrossRef CAS PubMed.
- M. Ishikura, K. Yamada and T. Abe, Nat. Prod. Rep., 2010, 27, 1630 RSC.
- A. Pelter, E. Buss and E. Colclough, J. Chem. Soc., Chem. Commun., 1987, 297–299 RSC.
- E. Negishi, Z. Huang, G. Wang, S. Mohan, C. Wang and H. Hattori, Acc. Chem. Res., 2008, 41, 1474–1485 CrossRef CAS PubMed.
- H. Lindlar, Helv. Chim. Acta, 1952, 35, 446–450 CrossRef CAS.
- M. J. Koh, R. K. M. Khan, S. Torker and A. H. Hoveyda, Angew. Chem., Int. Ed., 2014, 53, 1968–1972 CrossRef CAS PubMed.
- L. F. van Staden, D. Gravestock and D. J. Ager, Chem. Soc. Rev., 2002, 31, 195–200 RSC.
- K. Nakajima, X. Guo and Y. Nishibayashi, Chem. – Asian J., 2018, 13, 3653–3657 CrossRef CAS PubMed.
- J. J. Molloy, J. B. Metternich, C. G. Daniliuc, A. J. B. Watson and R. Gilmour, Angew. Chem., Int. Ed., 2018, 57, 3168–3172 CrossRef CAS PubMed.
- H. Zhang, Q. Xu, L. Yu and S. Yu, Eur. J. Org. Chem., 2020, 1472–1477 CrossRef CAS.
- K. Livingstone, M. Tenberge, F. Pape, C. G. Daniliuc, C. Jamieson and R. Gilmour, Org. Lett., 2019, 21, 9677–9680 CrossRef CAS PubMed.
- W. Cai, H. Fan, D. Ding, Y. Zhang and W. Wang, Chem. Commun., 2017, 53, 12918–12921 RSC.
- M. Bhadra, S. Kandambeth, M. K. Sahoo, M. Addicoat, E. Balaraman and R. Banerjee, J. Am. Chem. Soc., 2019, 141, 6152–6156 CrossRef CAS PubMed.
- A. López-Magano, X. Solans-Monfort, N. Salaverri, L. Marzo, R. Mas-Ballesté and J. Alemán, Sol. RRL, 2024, 8, 2300768 CrossRef.
- H. Li, H. Chen, Y. Zhou, J. Huang, J. Yi, H. Zhao, W. Wang and L. Jing, Chem. – Asian J., 2020, 15, 555–559 CrossRef CAS PubMed.
- C.-X. Liu, Y. Yao, Z.-W. Zhou, S. Qin, Z.-P. Yu, F.-Y. Tao, W.-D. Li, X.-Q. Yu and N. Wang, ChemSusChem, 2025, 18, e202401387 CrossRef CAS PubMed.
- D. C. Fabry, M. A. Ronge and M. Rueping, Chem. – Eur. J., 2015, 21, 5350–5354 CrossRef CAS PubMed.
- S. I. Faßbender, J. J. Molloy, C. Mück-Lichtenfeld and R. Gilmour, Angew. Chem., Int. Ed., 2019, 58, 18619–18626 CrossRef PubMed.
- N. F. Nikitas, P. L. Gkizis and C. G. Kokotos, Org. Biomol. Chem., 2021, 19, 5237–5253 RSC.
- I. K. Sideri, E. Voutyritsa and C. G. Kokotos, Org. Biomol. Chem., 2018, 16, 4596–4614 RSC.
- J. Corpas, P. Mauleón, R. Gómez Arrayás and J. C. Carretero, Adv. Synth. Catal., 2022, 364, 1348–1370 CrossRef CAS.
- CCDC 2465569: Experimental Crystal Structure Determination, 2025, DOI:10.5517/ccdc.csd.cc2nrmhk.
| This journal is © The Royal Society of Chemistry 2025 |