Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Application of thiopyrylium trifluoromethanesulfonates as Lewis acids in organocatalytic reactions
Noriyoshi
Nagahora
 *,
Chikako
Yoshimura
,
Mana
Hotta
and
Yuzuki
Watanabe
*,
Chikako
Yoshimura
,
Mana
Hotta
and
Yuzuki
Watanabe
Department of Chemistry, Faculty of Science, Fukuoka University, Fukuoka 814-0180, Japan. E-mail: nagahora@fukuoka-u.ac.jp
First published on 30th September 2025
Abstract
The successful application of thiopyrylium trifluoromethanesulfonate (triflate) as organic Lewis-acidic catalysts in electro-philic aromatic bromination, acetalization, cascade, and three-component reactions is disclosed. All these transformations provided the desired products in good yield. In particular, a catalytic amount of thiopyrylium triflate was used to activate N-bromosuccinimide for the bromination of various aromatic compounds under ambient and dark conditions.
Catalyst-mediated organic synthesis is a widely used and versatile tool in the field of organic chemistry. In particular, the use of transition metals as Lewis-acidic catalysts has attracted considerable attention.1 By definition, a Lewis acid has a low-lying lowest unoccupied molecular orbital (LUMO) that can accept lone-pair electrons.2 Compared with transition-metal catalysis, synthetic chemistry using metal-free Lewis-acidic organocatalysts is less explored, despite the inherent interest of synthesizing high purity molecules without metals in the fields of materials science and pharmaceutical research.
Recently, cationic organic salts, i.e., cationic organic molecules that bear counter anions, have been sporadically used for promoting specific target transformations such as electrophilic aromatic substitutions and aza-Diels–Alder reactions,3 paving the way for the design of cationic organic Lewis-acidic catalysts. For instance, Mukaiyama and coworkers have used carbocations as Lewis acids for the first time and employed triphenylcarbenium salts as catalysts for aldol, allylations, and Michael reactions.4 Cyclopropenium,5 tropylium,6 pyridinium,7 bromonium,8 and iodonium salts9 have also been applied as catalysts in several organic reactions as electron-pair acceptors. In addition, organocatalysis using chalcogen(IV) species such as sulfonium,10 selenonium,11 and telluronium salts12 has recently received much attention; however, reports on catalytic reactions using tertiary chalcogenium compounds remain limited.
Among the aforementioned compounds, thiopyrylium salts are of interest owing to their unique electronic structure and as new building blocks for sulfur-containing aromatic molecules.13 In 2020, we reported the synthesis of a new series of thiopyrylium salts via the Lewis- or Brønsted-acid-promoted intramolecular cyclization of diarylthioethers.14 The resulting thiopyrylium compounds bear a cationic organic framework, as revealed by their spectroscopic characterization, and can be handled under atmospheric conditions. Owing to the presence of a cationic sulfur atom whose charge is delocalized over a conjugated π-electron system, we envisioned that the thiopyrylium salt could serve as a Lewis acid in solution. Here, we demonstrate for the first time that thiopyrylium trifluoromethanesulfonates (triflates) can be used as a Lewis acid in bromination, acetalization, cascade, and multi-component reactions. In particular, we describe the application of thiopyrylium triflates as a Lewis acid to activate N-bromosuccinimide (NBS) for bromination reactions. This reaction is especially important because traditional electrophilic brominations using molecular bromine to obtain brominated aromatic hydrocarbons, which are relevant compounds in metal-catalyzed cross-coupling reactions, pharmaceuticals, and naturally occurring products,1 often suffer from complications associated with toxicity and operational complexity. Therefore, intense efforts have been devoted to enhancing the inherently low reactivity of NBS as an alternative bromination reagent using catalytic activators.15
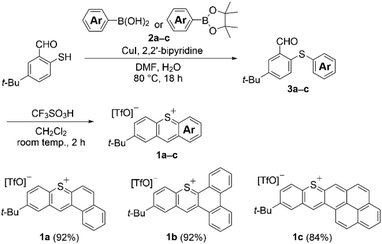
We initially synthesized new thiopyrylium triflates 1a–c using our previously developed intramolecular cyclization (Scheme 1).14 Coupling reactions of 5-(tert-butyl)-2-sunfanylbenzaldehyde with naphthalen-2-ylboronic acid (2a), phenanthren-9-ylboronic acid (2b), and pyren-2-ylboronic acid pinacol ester (2c) afforded diarylthioethers 3a–c, respectively, in moderate yields (see SI for details). Then, treatment of thioethers 3a–c with trifluoromethanesulfonic acid (TfOH) at room temperature afforded thiopyrylium triflates 1a–c in good yields via intramolecular cyclization. Thiopyrylium triflates 1a–c are thermally stable under atmospheric conditions and do not exhibit any signs of decomposition or hygroscopicity.
We first investigated the Lewis acidity of thiopyrylium triflates 1a–c using the Gutmann–Beckett method,16 which is based on the use of trioctylphosphine oxide (n-Oc3P![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O) as a probe to detect the chemical-shift changes (ΔδP) in the 31P NMR spectra of the evaluated systems upon addition of n-Oc3P
O) as a probe to detect the chemical-shift changes (ΔδP) in the 31P NMR spectra of the evaluated systems upon addition of n-Oc3P![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O. Adding one equivalent of n-Oc3P
O. Adding one equivalent of n-Oc3P![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O to a solution of 1a–c in CD3CN resulted in downfield shifts of ΔδP = 12.0, 16.3, and 19.5 ppm, respectively, compared to the chemical shift of free n-Oc3P
O to a solution of 1a–c in CD3CN resulted in downfield shifts of ΔδP = 12.0, 16.3, and 19.5 ppm, respectively, compared to the chemical shift of free n-Oc3P![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O.17 These results suggest that thiopyrylium triflates 1a–c inherently behave as Lewis acids in solution, whereby 1b and 1c exhibiting higher Lewis acidity than 1a. Thus, intermolecular interaction between these salts and trioctylphosphine oxide would be affected by their molecular structures of 1a–c.
O.17 These results suggest that thiopyrylium triflates 1a–c inherently behave as Lewis acids in solution, whereby 1b and 1c exhibiting higher Lewis acidity than 1a. Thus, intermolecular interaction between these salts and trioctylphosphine oxide would be affected by their molecular structures of 1a–c.
Then, the intermolecular interaction of thiopyrylium triflate 1b with NBS was investigated via NMR spectroscopy using a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 stoichiometric mixture of thiopyrylium salt 1b and NBS. The 1H and 13C NMR spectra of the mixture in CDCl3 showed slightly shifted signals compared with those of free 1b and NBS, indicating that no covalent bond was formed between 1b and NBS. Therefore, thiopyrylium triflate 1b behaves as a soft Lewis acid toward NBS in solution. Moreover, theoretical investigations on the interactions between thiopyrylium cations 1a′–c′ and NBS were conducted using the Gaussian 16 programs suite. In all cases, the interactions were exothermic. In particular, thiopyrylium cation 1b′ and NBS showed the most effective interaction with an exothermic energy of 1.98 kcal mol−1, which is consistent with the experimentally observed affinity of 1b for NBS. Regarding the optimized structure, interaction between HOMO of 1b′ and LUMO of NBS might be caused by a planar stacked conformation of the 1b′/NBS complex.
1 stoichiometric mixture of thiopyrylium salt 1b and NBS. The 1H and 13C NMR spectra of the mixture in CDCl3 showed slightly shifted signals compared with those of free 1b and NBS, indicating that no covalent bond was formed between 1b and NBS. Therefore, thiopyrylium triflate 1b behaves as a soft Lewis acid toward NBS in solution. Moreover, theoretical investigations on the interactions between thiopyrylium cations 1a′–c′ and NBS were conducted using the Gaussian 16 programs suite. In all cases, the interactions were exothermic. In particular, thiopyrylium cation 1b′ and NBS showed the most effective interaction with an exothermic energy of 1.98 kcal mol−1, which is consistent with the experimentally observed affinity of 1b for NBS. Regarding the optimized structure, interaction between HOMO of 1b′ and LUMO of NBS might be caused by a planar stacked conformation of the 1b′/NBS complex.
Having examined the Lewis acidity of 1a–c in solution, we tested their catalytic activity in the electrophilic aromatic bromination of anisole by reacting anisole with NBS in the presence of 5.0 mol% of 1a–c under dark conditions. The results are summarized in Table 1.
First, we confirmed that the bromination did not proceed without thiopyrylium triflate or any additive (entry 1). Using TfOH as a Brønsted acid enhanced the bromination, resulting in the formation of 4-bromoanisole in 63% yield (entry 2); however, the addition of both TfOH and 2,4,6-trimethylpyridine did not afford the desired product (entry 3). The bromination proceeded smoothly in the presence of 5.0 mol% of thiopyrylium triflate 1b under dark conditions to furnish 4-bromoaniole in 58% yield (entry 5), whereas thiopyrylium triflates 1a and 1c afforded 4-bromoaniole in lower yield, i.e., 48% and 33%, respectively (entries 4 and 6). The observed lower activity for 1c might be caused by its low solubility toward CH2Cl2. No significant change in the yield was observed when using 1b in the presence of 2,4,6-trimethylpyridine (entry 7). These results confirmed that thiopyrylium triflate 1b effectively catalyzes this electrophilic aromatic bromination as a Lewis acid and that the 1b-catalyzed bromination was not affected by the presence of a Lewis base.
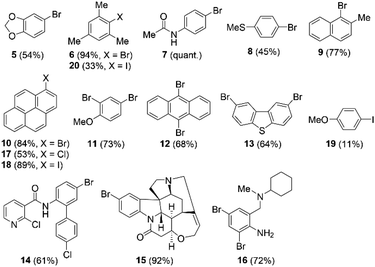
With the optimized reaction conditions in hand, we next explored the substrate scope of the electrophilic bromination by NBS (Chart 1). In the presence of 5.0 mol% of 1b, 1,3-benzodioxole, mesitylene, acetanilide and thioanisole reacted with NBS to afford monobrominated compounds 5, 6, 7, and 8 in 54%, 94%, quantitative, and 45% yield, respectively. Although naphthalene did not undergo the desired bromination, 2-methylnaphthalene readily furnished 1-bromo-2-methylnaphthalene (9) in 77% yield. When using pyrene, the reaction proceeded regioselectively to produce 1-bromopyrene (10) in 84% yield. In the cases of anisole, anthracene, and dibenzo[b,d]thiophene, two-fold bromination occurred preferentially to afford 2,4-dibromoanisole (11), 9,10-dibromoanthracene (12), and 2,8-dibromodibenzo[b,d]thiophene (13) in 73%, 68%, and 64% yield, respectively. We confirmed that the thiopyrylium-catalyzed bromination can also be applied to agricultural chemicals and alkaloid molecules. Specifically, boscalid and strychnine reacted under the applied bromination conditions to furnish brominated derivatives 14 and 15 in 61% and 92% yield, respectively. Dibromoaniline derivative 16, an important compound in the field of pharmaceutical chemistry, was also obtained via two-fold bromination. Conversely, 2-methylquinoline, 9H-thioxanthen-9-one, 2,5-dimethylthiophene, phenanthrene, neopentylbenzene, and 1-bromo-3-chlorobenzene were not converted to the corresponding brominated products under the applied reaction conditions. The monobromination of anisole was also performed using N-bromophthalimide instead of NBS under the otherwise optimized conditions, to afford 4 in 74% isolated yield. Moreover, chlorination and iodination reactions using N-chloro- and N-iodosuccinimide, respectively, were successfully conducted, which furnished 1-chloropyrene (17), 1-iodopyrene (18), 4-iodoanisole (19), and iodomesitylene (20) in 53%, 89%, 11%, and 33% isolated yield, respectively (Chart 1). Thus, the thiopyrylium-triflate-promoted bromination, chlorination, and iodination is suitable for a variety of aromatic hydrocarbons, sulfur-containing heterocycles, agricultural chemicals, and alkaloids.
The robustness of the developed organic catalytic system was tested by recovering the catalyst after the reaction and using the reused catalyst for new reaction cycles. Upon completion of the bromination of pyrene in the presence of 1b, the reaction mixture was separated by centrifugation at 0 °C, affording an orange solid. The obtained solid was then treated repeatedly with another portion of pyrene and NBS in CH2Cl2. The catalytic system could be reused at least five times with complete conversion and virtually no activity loss. Specifically, the product yield from the first to the fifth experiments was 91%, 100%, 86%, 86%, and 82%. These results demonstrate the robustness of catalyst 1b in these electrophilic bromination reactions.
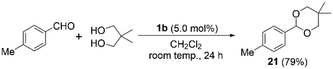
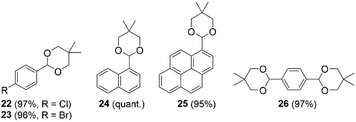
Having confirmed the activity and stability of thiopyrylium triflate 1b in electrophilic bromination reactions, we examined its ability to catalyze other organic reactions. Thiopyrylium triflate 1b was thus applied in the acetalization reaction of benzaldehyde derivatives using a diol. First, the acetalization reaction of p-tolualdehyde and 2,2-dimethylpropane-1,3-diol proceeded using 5.0 mol% of 1b at room temperature (Scheme 2). The 1H NMR spectrum of the reaction mixture revealed the complete conversion of the substrate within 24 h. After chromatographic purification, acetal 21 was isolated in 79% yield. Then, we evaluated the effect of different substituents on the benzene ring of the benzaldehyde precursors. Installing an electron-withdrawing group on the aromatic ring did not significantly alter the cyclization outcome (cf. compounds 22 and 23 in Chart 2). When using 1-naphthaldehyde, the acetalization smoothly proceeded to provide acetal 24 in quantitative isolated yield. π-Expanded 1-pyrenecarboxaldehyde successfully reacted with the diol in the presence of 1b (5.0 mol%) to afford the corresponding acetal (25) in 95% isolated yield. Terephthalaldehyde also proved to be a suitable substrate, which provided bisacetal derivative 26 in good yield. These results clearly demonstrate the versatility of 1b, whose sufficient Lewis acidity renders it suitable for a variety of catalytic reactions.
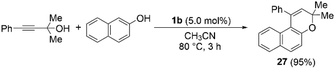
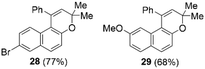
Next, we turned our attention to expand the reaction of propargylic alcohols with naphthalen-2-ol in the presence of thiopyrylium triflate 1b as a Lewis acid (Scheme 3). First, we confirmed the complete recovery of the substrates in the absence of 1b. When using 5.0 mol% of 1b, this cascade reaction smoothly proceeded at 80 °C within 3 h according to 1H NMR spectroscopy monitoring. 3H-Benzochromene derivative 27 was obtained in 95% isolated yield after chromatographic separation. In addition, this direct construction of the 3H-benzochromene framework provided 28 and 29 in excellent yield under the applied conditions (Chart 3). Thus, propargylic alcohols could be activated by thiopyrylium triflate 1b to undergo the Friedel–Crafts reaction with naphthalen-2-ol to give 3H-benzochromene derivatives.
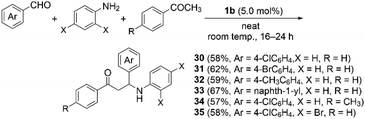
Finally, we examined Mannich reactions catalyzed by thiopyrylium triflate 1b as a model three-component reaction (Scheme 4). In the absence of 1b at room temperature, the control reaction did not lead to the desired three-component product. Meanwhile, in the presence of 5.0 mol% of 1b, the reaction of 4-chlorobenzaldehyde, aniline, and acetophenone furnished Mannich product 30 in 58% isolated yield. 4-Bromobenzaldehyde, p-tolualdehyde, and 1-naphthaldehyde were also tolerated as substrates instead of 4-chlorobenzaldehyde, providing three-component products 31, 32, and 33 in 62%, 59%, and 67% yield, respectively. In the case of substituted acetophenone and aniline derivatives, the reactions smoothly produced 34 and 35 in 57% and 58% yield, respectively. These results confirm that thiopyrylium triflate 1b also works as a Lewis-acidic organocatalyst in Mannich-type condensations to afford the three-component products in moderate yield.
In summary, we have for the first time, successfully used thiopyrylium triflate 1b as a Lewis-acidic organocatalyst in electrophilic bromination reactions of aromatic compounds, acetalization reactions of benzaldehydes, cascade reactions, and Mannich reactions. The corresponding products were obtained in good yield. DFT calculations suggested the formation of noncovalent interactions between the thiopyrylium cation fragment and the imide/carbonyl group of NBS or the benzaldehydes. Further studies on the application of thiopyrylium salts to catalyze other organic reactions are currently in progress in our laboratory.
Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: experimental details, characterization data, and supporting figures and tables. See DOI: https://doi.org/10.1039/d5ob01353d.References
- P. Ruiz-Castillo and S. L. Buchwald, Chem. Rev., 2016, 116, 12564–12649 CrossRef CAS PubMed
.
- A. Corma and H. García, Chem. Rev., 2003, 103, 4307–4365 CrossRef CAS PubMed
.
-
(a) J. An, R. M. Denton, T. H. Lambert and E. D. Nacsa, Org. Biomol. Chem., 2014, 12, 2993–3003 RSC
; (b) V. R. Naidu, S. Ni and J. Franzén, ChemCatChem, 2015, 7, 1896–1905 CrossRef CAS
; (c) D. J. M. Lyons, R. D. Crocker, M. Blümel and T. V. Nguyen, Angew. Chem., Int. Ed., 2017, 56, 1466–1484 CrossRef CAS PubMed
.
-
(a) T. Mukaiyama, S. Kobayashi and S.-i. Shoda, Chem. Lett., 1984, 13, 907–910 CrossRef
; (b) T. Mukaiyama, S. Kobayashi and S.-i. Shoda, Chem. Lett., 1984, 13, 1529–1530 CrossRef
; (c) M. Ohshima, M. Murakami and T. Mukaiyama, Chem. Lett., 1985, 14, 1871–1874 CrossRef
; (d) M. Yanagisawa and T. Mukaiyama, Chem. Lett., 2001, 30, 224–225 CrossRef
.
- R. Chen, T. Zheng, X. Jiang and Y.-Y. Yeung, ACS Catal., 2024, 14, 9198–9206 CrossRef CAS
.
-
(a) D. J. M. Lyons, R. D. Crocker, D. Enders and T. V. Nguyen, Green Chem., 2017, 19, 3993–3996 RSC
; (b) U. P. N. Tran, G. Oss, D. P. Pace, J. Ho and T. V. Nguyen, Chem. Sci., 2018, 9, 5145–5151 RSC
; (c) G. Oss, J. Ho and T. V. Nguyen, Eur. J. Org. Chem., 2018, 3974–3981 CrossRef CAS
; (d) M. A. Hussein, V. T. Huynh, R. Hommelsheim, R. M. Koenigs and T. V. Nguyen, Chem. Commun., 2018, 54, 12970–12973 RSC
; (e) C. Empel, T. V. Nguyen and R. M. Koenigs, Org. Lett., 2021, 23, 548–553 CrossRef CAS PubMed
; (f) N. N. H. Ton, B. K. Mai and T. V. Nguyen, J. Org. Chem., 2021, 86, 9117–9133 CrossRef CAS PubMed
; (g) Rekha, S. Sharma, G. Singh and R. V. Anand, ACS Org. Inorg. Au, 2022, 2, 186–196 CrossRef CAS PubMed
.
-
(a) S. Das, D. Pekel, J.-M. Neudörfl and A. Berkessel, Angew. Chem., Int. Ed., 2015, 54, 12479–12483 CrossRef CAS PubMed
; (b) M. Shaw, Y. Kumar, R. Thakur and A. Kumar, Beilstein J. Org. Chem., 2017, 13, 2385–2395 CrossRef CAS PubMed
; (c) T. Ghosh, A. Mukherji and P. K. Kancharla, Org. Lett., 2019, 21, 3490–3495 CrossRef CAS PubMed
.
-
(a) Y. Yoshida, S. Ishikawa, T. Mino and M. Sakamoto, Chem. Commun., 2021, 57, 2519–2522 RSC
; (b) Y. Yoshida, T. Mino and M. Sakamoto, ACS Catal., 2021, 11, 13028–13033 CrossRef CAS
.
-
(a) Y. Zhang, J. Han and Z.-J. Liu, RSC Adv., 2015, 5, 25485–25488 RSC
; (b) F. Heinen, E. Engelage, A. Dreger, R. Weiss and S. M. Huber, Angew. Chem., Int. Ed., 2018, 57, 3830–3833 CrossRef CAS PubMed
; (c) B. Ye, J. Zhao, K. Zhao, J. M. McKenna and F. D. Toste, J. Am. Chem. Soc., 2018, 140, 8350–8356 CrossRef CAS PubMed
; (d) F. Heinen, D. L. Reinhard, E. Engelage and S. M. Huber, Angew. Chem., Int. Ed., 2021, 60, 5069–5073 CrossRef CAS PubMed
.
-
(a) S. Kaneko, Y. Kumatabara, S. Shimizu, K. Maruoka and S. Shirakawa, Chem. Commun., 2017, 53, 119–122 RSC
; (b) S. Liu, K. Maruoka and S. Shirakawa, Angew. Chem., Int. Ed., 2017, 56, 4819–4823 CrossRef CAS PubMed
; (c) C. Q. He, C. C. Lam, P. Yu, Z. Song, M. Chen, Y.-h. Lam, S. Chen and K. N. Houk, J. Org. Chem., 2020, 85, 2618–2625 CrossRef CAS PubMed
; (d) A. S. Novikov and D. S. Bolotin, Org. Biomol. Chem., 2022, 20, 7632–7639 RSC
; (e) M. V. Il'in, A. S. Novikov and D. S. Bolotin, J. Org. Chem., 2022, 87, 10199–10207 CrossRef PubMed
; (f) Y. Lu, Q. Liu, Z.-X. Wang and X.-Y. Chen, Angew. Chem., Int. Ed., 2022, 61, e202116071 CrossRef CAS PubMed
.
-
(a) E. J. Lenardão, S. R. Mendes, P. C. Ferreira, G. Perin, C. C. Silveira and R. G. Jacob, Tetrahedron Lett., 2006, 47, 7439–7442 CrossRef
; (b) E. J. Lenardão, E. L. Borges, S. R. Mendes, G. Perin and R. G. Jacob, Tetrahedron Lett., 2009, 49, 1919–1921 CrossRef
; (c) E. J. Lenardão, J. d. O. Feijó, S. Thurow, G. Perin, R. G. Jacob and C. C. Silveira, Tetrahedron Lett., 2009, 50, 5215–5217 CrossRef
; (d) X. He, X. Wang, Y.-L. S. Tse, Z. Ke and Y.-Y. Yeung, Angew. Chem., Int. Ed., 2018, 57, 12869–12873 CrossRef CAS PubMed
; (e) X. He, X. Wang, Y.-L. S. Tse, Z. Ke and Y.-Y. Yeung, ACS Catal., 2021, 11, 12632–12642 CrossRef CAS
; (f) K. Okuno, T. Nakamura and S. Shirakawa, Asian J. Org. Chem., 2021, 10, 655–659 CrossRef CAS
; (g) A. Y. B. Ángel, P. R. O. Campos and E. E. Alberto, Molecules, 2023, 28, 3056 CrossRef PubMed
.
-
(a) B. Zhou and F. P. Gabbaï, Chem. Sci., 2020, 11, 7495–7500 RSC
; (b) B. Zhou and F. P. Gabbaï, Organometallics, 2021, 40, 2371–2374 CrossRef CAS
; (c) R. Weiss, E. Aubert, P. Pale and V. Mamane, Angew. Chem., Int. Ed., 2021, 60, 19281–19286 CrossRef CAS PubMed
.
-
(a) N. Nagahora, T. Kushida, K. Shioji and K. Okuma, Organometallics, 2019, 38, 1800–1808 CrossRef CAS
; (b) N. Nagahora, K. Kitahara, Y. Mizuhata, N. Tokitoh, K. Shioji and K. Okuma, J. Org. Chem., 2020, 85, 7748–7756 CrossRef CAS PubMed
.
-
(a) N. Nagahora, R. Tanaka, T. Tada, A. Yasuda, Y. Yamada, K. Shioji and K. Okuma, Org. Lett., 2020, 22, 6192–9196 CrossRef CAS PubMed
; (b) K. Hirano, A. Yasuda, T. Sasamori, K. Shioji, K. Okuma and N. Nagahora, Heterocycles, 2021, 102, 451–464 CrossRef CAS
.
-
(a) F. Mo, J. M. Yan, D. Qiu, F. Li, Y. Zhang and J. Wang, Angew. Chem., Int. Ed., 2010, 49, 2028–2032 CrossRef CAS PubMed
; (b) K. Tanemura, T. Suzuki, Y. Nishida, K. Satsumabayashi and T. Horaguchi, Chem. Lett., 2003, 32, 932–933 CrossRef CAS
; (c) Y. Nishii, M. Ikeda, Y. Hayashi, S. Kawauchi and M. Miura, J. Am. Chem. Soc., 2019, 142, 1621–1629 CrossRef PubMed
; (d) C. N. Kona, R. Oku, S. Nakamura, M. Miura, K. Hirano and Y. Nishii, Chem, 2024, 10, 402–413 CrossRef CAS
.
-
(a) M. A. Beckett, G. C. Strickland, J. R. Holland and K. S. Varma, Polymer, 1996, 37, 4629–4631 CrossRef CAS
; (b) R. R. Golwankar, T. D. Curry II, C. J. Paranjothi and J. D. Blakemore, Inorg. Chem., 2023, 62, 9765–9780 CrossRef CAS PubMed
.
- We also observed that adding one equivalent of n-Oc3P
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O to a solution of tropylium tetrafluoroborate/triphenylcarbenium tetrafluoroborates in CD3CN resulted in downfield shifts of ΔδP = 5.01/24.4 ppm, respectively, compared to the chemical shift of free n-Oc3P
O to a solution of tropylium tetrafluoroborate/triphenylcarbenium tetrafluoroborates in CD3CN resulted in downfield shifts of ΔδP = 5.01/24.4 ppm, respectively, compared to the chemical shift of free n-Oc3P![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O.
O.
| This journal is © The Royal Society of Chemistry 2025 |