Open Access Article
Open Access ArticleEco-efficient C–H alkynylation of indoles via mechanochemical ruthenium catalysis
Rajaram
Maayuri
 ,
Vikash
Kumar
,
Sourav
Maiti
and
Parthasarathy
Gandeepan
,
Vikash
Kumar
,
Sourav
Maiti
and
Parthasarathy
Gandeepan
 *
*
Department of Chemistry, Indian Institute of Technology Tirupati, Yerpedu – Venkatagiri Road, Yerpedu Post, Tirupati District, Andhra Pradesh 517619, India. E-mail: pgandeepan@iittp.ac.in
First published on 10th September 2025
Abstract
A regioselective C2-alkynylation of indoles via ruthenium(II)-catalyzed C–H activation using bromoalkynes is demonstrated under both solution-phase and mechanochemical conditions. The solvent-minimized mechanochemical method delivers comparable yields with reduced reaction time and improved green metrics. Broad substrate scope, gram-scale applicability, and post-functionalization showcase the synthetic utility of this approach. This work underscores the potential of mechanochemistry as a sustainable and operationally simple alternative for direct C–H functionalization of heterocycles.
Indole derivatives are ubiquitous scaffolds found in a wide array of natural products, pharmaceuticals, agrochemicals, and advanced materials, making their selective functionalization a topic of enduring interest.1,2 Among various synthetic approaches, direct C–H activation3 has emerged as a powerful and sustainable strategy for the late-stage modification of heterocycles, including indoles. Over the past two decades, substantial progress has been made in the regioselective functionalization of indoles through C–H functionalization.4 In particular, alkynylated indoles serve as valuable building blocks for the construction of complex organic molecules with potential applications in medicinal chemistry and materials science.5 However, direct C2-alkynylation of indoles has been relatively underexplored compared to C2-arylation, alkylation, and alkenylation. In this context, work by Waser,6 Li,7 Shi,8 and others9 has demonstrated the regioselective incorporation of alkynyl groups at the C2-position of indoles via directed transition metal-catalyzed C–H activation using hypervalent iodine–alkyne reagents. Building on these advances, Ackermann10 and others9c,11 developed methods employing bromoalkynes as alkynylating agents for C2-selective functionalization of indoles. Despite their efficiency, these methodologies often require volatile and toxic organic solvents, elevated temperatures, and prolonged reaction times—factors that compromise the overall greenness and sustainability of the C–H activation protocols.
In recent years, solvent-free or solvent-less mechanochemical C–H activation reactions using ball-milling techniques have emerged as an attractive alternative to conventional solution-phase methods.12 These approaches offer several advantages, including reduced or no solvent use and significantly shorter reaction times. In this context, Bolm reported the mechanochemical C2-alkynylation of indoles using the sensitive hypervalent iodine-based reagent ethynylbenziodoxolone (EBX) as the alkyne source, catalyzed by the expensive [Cp*RhCl2]2 in the presence of AgNTf2 as an additive.13 While efficient, the reliance on costly rhodium catalysts, silver additives, and sensitive iodine(III) reagents limits the broader applicability of this method.
To address these limitations, we sought to develop a cost-effective and sustainable mechanochemical approach for the C2-selective alkynylation of indoles (Scheme 1). Our strategy employs the inexpensive ruthenium-based catalyst14 [Ru(p-cymene)Cl2]2 and readily accessible bromoalkynes under ball-milling conditions. Furthermore, we aimed to compare the efficiency, practicality, and green chemistry metrics of this mechanochemical protocol with its solution-phase counterpart, thereby evaluating the potential of mechanochemistry as a viable and greener alternative for C–H alkynylation.
 | ||
| Scheme 1 Ruthenium(II)-catalyzed C2-alkynylation of indoles through solution-phase and mechanochemical strategy. | ||
We began our study by optimizing the reaction conditions for the synthesis of 2-alkynylated indole carboxamide 3a from N-methoxy-1H-indole-1-carboxamide (1a) and (bromoethynyl)triisopropylsilane (2a) (Table S1). Following detailed optimization, we identified that treatment of 1a (0.21 mmol, 1.0 equiv.) with 2a (0.42 mmol, 2.0 equiv.) in the presence of 2.5 mol% of [Ru(p-cymene)Cl2]2 and 1.0 equiv. of Na2CO3 in 2.0 mL of 2,2,2-trifluoroethanol (TFE) at room temperature for 24 h afforded 3a in 84% isolated yield (Scheme 2). This reaction condition was designated as the standard solution-phase condition (Condition A). In parallel, we performed optimization studies under mechanochemical conditions to identify an optimal protocol (Table S2). Milling a mixture of 1a (0.21 mmol, 1.0 equiv.), 2a (0.42 mmol, 2.0 equiv.), [Ru(p-cymene)Cl2]2 (2.5 mol%), Na2CO3 (1.0 equiv.), and TFE (100 μL, used as a liquid-assisted grinding (LAG) additive) in a 5 mL stainless-steel (SS) jar with three SS balls (ϕ = 5 mm) at 30 Hz for 3 h furnished 3a in 80% isolated yield (Scheme 2). This optimized condition was designated as the standard mechanochemical condition (Condition B).
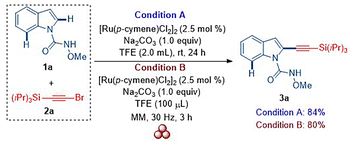 | ||
| Scheme 2 Ruthenium(II)-catalyzed solution-phase or mechanochemical synthesis of C2-alkynylated indoles. | ||
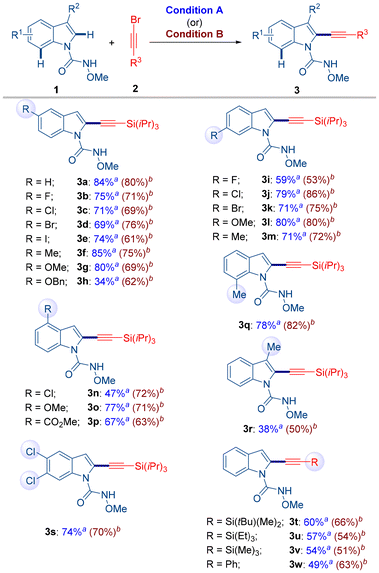
With the optimized reaction conditions A and B established, a series of indole substrates (1a–1s) were subjected to alkynylation with 2a (Scheme 3). Various indoles bearing substituents at the 4-, 5-, 6-, and 7-positions underwent smooth C2-alkynylation to afford the corresponding products (3a–3s) in satisfactory yields under both conditions. Notably, halogen substituents on the indole ring were well tolerated, offering potential for further diversification via cross-coupling strategies. The sterically hindered 3-methylindole substrate (1r) also participated in the reaction, albeit with reduced efficiency due to steric hindrance at the C3 position. In a similar fashion, alkynylation reactions employing bromoalkynes bearing alternative silyl groups in place of Si(iPr)3 were conducted with 1a, delivering the corresponding products (3t–3v) in good yields. Furthermore, (bromoethynyl)benzene (2e) also underwent reaction with 1a to afford the desired product 3w in good yield under conditions A and B. These results highlight the broad applicability of both protocols across diverse indole substrates and bromoalkyne partners. Overall, the reaction scope illustrates that both solution-phase and mechanochemical conditions deliver comparable efficiencies in most cases. However, considering the significantly shorter reaction times and reduced solvent usage, the mechanochemical approach offers distinct advantages in terms of operational simplicity and environmental sustainability.
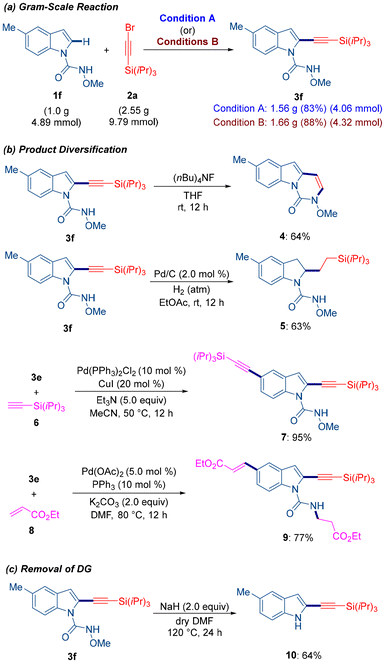
Next, we evaluated the scalability of the reaction. As shown in Scheme 4a, a gram-scale reaction between 1f and 2a afforded the desired product 3f in 83% yield under solution-phase conditions and 88% yield under mechanochemical conditions, demonstrating the practicality of both approaches. To showcase the synthetic utility of the alkynylated product, 3f was treated with tetrabutylammonium fluoride (TBAF) to furnish the corresponding pyrimidoindolone derivative 4 (Scheme 4b). Furthermore, hydrogenation of 3f using Pd/C under a hydrogen atmosphere provided the indoline derivative 5 in 63% yield. We also demonstrated the use of cross-coupling strategies for the functionalization of halogen-substituted products. For example, compound 3e underwent a Sonogashira coupling reaction with 6 and a Heck coupling with 8 to afford products 7 and 9, respectively. Finally, treatment of 3f with NaH successfully removed the N-methoxy carboxamide directing group, affording product 10 in good yield (Scheme 4c).
Based on the experimental results and literature precedents,14b,c we propose a plausible catalytic cycle involving a cyclometalated ruthenium complex 11 (Fig. 1). Coordination of alkynyl bromide 2a to complex 11, followed by migratory insertion, generates a seven-membered ruthenacycle intermediate 13. This intermediate subsequently undergoes β-bromo elimination to afford species 14. The resulting complex then reacts with another molecule of 1a in the presence of Na2CO3 to release the product 3a and regenerate the ruthenacycle intermediate 11, completing the catalytic cycle.
After achieving a broad substrate scope and demonstrating scalability, our focus turned to evaluating the robustness of both sets of reaction conditions. Accordingly, we assessed the reaction efficiency for forming product 3a under various sensitivity assessment parameters.15 For Condition A, we evaluated factors such as catalyst loading, the amounts of base and alkynylating reagents, reaction time, solvent volume, and reaction scale. As shown in the radar diagram (Fig. 2a), variations in these parameters resulted in only minor deviations from the standard yield. Similarly, Condition B was also subjected to sensitivity analysis. Gratifyingly, the corresponding radar diagram (Fig. 2b) revealed minimal deviation from the standard yield, highlighting the robustness of the optimized conditions.
 | ||
| Fig. 2 Sensitivity assessment of the reaction Conditions A and B. (a) Radar diagram for Condition A. (b) Radar diagram for Condition B. c = concentration. T = temperature. t = time. cat. = catalyst. | ||
To evaluate the eco-friendliness of the two developed strategies, we analyzed the green chemistry metrics (GCM) for the gram-scale synthesis of compound 3f from 1f (1.0 g, 4.89 mmol) and 2a (2.55 g, 9.79 mmol).16 As shown in Table 1, both strategies exhibit satisfactory green chemistry metrics. Notably, the mechanochemical approach demonstrates superior green metrics compared to the solution-phase method. It delivers a significantly lower E-factor (2.33 vs. 19.47) and improved process mass intensity (PMI = 3.33 vs. 20.48), indicating reduced waste generation and material input. Moreover, enhanced mass productivity and EcoScale values further underscore its operational and environmental efficiency.
| Entry | Green chemistry metrics | Ideal value | Obtained value for product 3f | |
|---|---|---|---|---|
| Solution phase approach | Mechanochemical approach | |||
| 1 | Chemical yield (CY) | 100% | 82.95 | 88.33 |
| 2 | E-factor | 0 | 19.47 | 2.33 |
| 3 | Process mass Intensity (PMI) | 1 | 20.48 | 3.33 |
| 4 | Atom economy (AE) | 100% | 82.61 | 82.61 |
| 5 | Carbon efficiency (CE) | 100% | 66.66 | 66.66 |
| 6 | Reaction mass efficiency (RME) | 100% | 43.94 | 46.76 |
| 7 | Optimum efficiency (OE) | 100% | 53.18 | 56.60 |
| 8 | Mass productivity (MP) | 100% | 4.88 | 30.56 |
| 9 | Waste intensity (WI) | 0 | 0.95 | 0.70 |
| 10 | Percentage conversion (PC) | 100% | 100 | 100 |
| 11 | Percentage selectivity (PS) | 100% | 82.95 | 88.33 |
| 12 | EcoScale | 100% | 68.47 | 73.17 |
In conclusion, we have developed a regioselective C2-alkynylation of indoles employing a ruthenium(II)-catalyzed C–H activation strategy under both solution-phase and mechanochemical conditions. Using readily available bromoalkynes and a cost-effective [Ru(p-cymene)Cl2]2 catalyst enabled efficient functionalization across a broad range of indole substrates, with both protocols delivering comparable yields. The mechanochemical approach, in particular, demonstrated operational simplicity, shorter reaction times, and reduced solvent consumption. Scalability was confirmed through gram-scale synthesis, and the synthetic utility of the alkynylated products was exemplified through downstream transformations. Sensitivity assessments highlighted the robustness of both protocols, while green chemistry metric analysis revealed the superior environmental profile of the mechanochemical method. These findings underscore the potential of mechanochemistry as a practical and sustainable alternative for C–H alkynylation in complex molecule synthesis.
Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the SI. Supplementary information: experimental details, characterization data of the isolated products, and copies of the NMR spectra of new products. See DOI: https://doi.org/10.1039/d5ob01198a.Acknowledgements
The authors are grateful to the Anusandhan National Research Foundation (ANRF) India (Grant No. CRG/2023/008708), and the Ministry of Education (MoE), India (Grant No. MoE-STARS/STARS-2/2023-0685) for financial support. The authors also thank the Indian Institute of Technology Tirupati for providing infrastructure, facilities, and fellowship to V. K. The Council of Scientific and Industrial Research (CSIR), India, is sincerely acknowledged for awarding a Senior Research Fellowship (SRF) to R. M.References
- (a) L. Du, L. Dian, S. A. Newmister, Y. Xia, G. Luo, D. H. Sherman and S. Li, Chem. Rev., 2025, 125, 1718–1804 CrossRef CAS PubMed; (b) J. Dhuguru and R. Skouta, Molecules, 2020, 25, 1615 CrossRef CAS; (c) E. El-Sawy, A. Abdelwahab and G. Kirsch, Synthesis, 2018, 4525–4538 CAS.
- (a) S. Das, New J. Chem., 2023, 47, 13729–13775 RSC; (b) M. Inman and C. J. Moody, Chem. Sci., 2013, 4, 29–41 RSC; (c) S. Cacchi and G. Fabrizi, Chem. Rev., 2011, 111, PR215–PR283 CrossRef PubMed; (d) K. Krüger, A. Tillack and M. Beller, Adv. Synth. Catal., 2008, 350, 2153–2167 CrossRef; (e) G. R. Humphrey and J. T. Kuethe, Chem. Rev., 2006, 106, 2875–2911 CrossRef CAS.
- (a) S. K. Sinha, S. Guin, S. Maiti, J. P. Biswas, S. Porey and D. Maiti, Chem. Rev., 2021, 122, 5682–5841 CrossRef PubMed; (b) T. Rogge, N. Kaplaneris, N. Chatani, J. Kim, S. Chang, B. Punji, L. L. Schafer, D. G. Musaev, J. Wencel-Delord, C. A. Roberts, R. Sarpong, Z. E. Wilson, M. A. Brimble, M. J. Johansson and L. Ackermann, Nat. Rev. Methods Primers, 2021, 1, 43 CrossRef CAS; (c) T. Dalton, T. Faber and F. Glorius, ACS Cent. Sci., 2021, 7, 245–261 CrossRef CAS; (d) P. Gandeepan, T. Müller, D. Zell, G. Cera, S. Warratz and L. Ackermann, Chem. Rev., 2019, 119, 2192–2452 CrossRef CAS; (e) C. G. Newton, S. G. Wang, C. C. Oliveira and N. Cramer, Chem. Rev., 2017, 117, 8908–8976 CrossRef CAS; (f) K. Murakami, S. Yamada, T. Kaneda and K. Itami, Chem. Rev., 2017, 117, 9302–9332 CrossRef CAS; (g) C. Liu, J. Yuan, M. Gao, S. Tang, W. Li, R. Shi and A. Lei, Chem. Rev., 2015, 115, 12138–12204 CrossRef CAS PubMed; (h) G. Song, F. Wang and X. Li, Chem. Soc. Rev., 2012, 41, 3651–3678 RSC; (i) T. Satoh and M. Miura, Chem. – Eur. J., 2010, 16, 11212–11222 CrossRef CAS.
- (a) J. Wen and Z. Shi, Acc. Chem. Res., 2021, 54, 1723–1736 CrossRef CAS PubMed; (b) K. Urbina, D. Tresp, K. Sipps and M. Szostak, Adv. Synth. Catal., 2021, 363, 2723–2739 CrossRef CAS; (c) P. Kumar, P. J. Nagtilak and M. Kapur, New J. Chem., 2021, 45, 13692–13746 RSC; (d) S. Pradhan, P. B. De, T. A. Shah and T. Punniyamurthy, Chem. – Asian J., 2020, 15, 4184–4198 CrossRef CAS; (e) T. A. Shah, P. B. De, S. Pradhan and T. Punniyamurthy, Chem. Commun., 2019, 55, 572–587 RSC; (f) J. Kalepu, P. Gandeepan, L. Ackermann and L. T. Pilarski, Chem. Sci., 2018, 9, 4203–4216 RSC; (g) J. A. Leitch, Y. Bhonoah and C. G. Frost, ACS Catal., 2017, 7, 5618–5627 CrossRef CAS.
- F. Diederich, P. J. Stang and R. R. Tykwinski, Acetylene chemistry: chemistry, biology and material science, Wiley-VCH, Weinheim, 2005 Search PubMed.
- G. L. Tolnai, S. Ganss, J. P. Brand and J. Waser, Org. Lett., 2012, 15, 112–115 CrossRef.
- F. Xie, Z. Qi, S. Yu and X. Li, J. Am. Chem. Soc., 2014, 136, 4780–4787 CrossRef CAS.
- Z.-Z. Zhang, B. Liu, C.-Y. Wang and B.-F. Shi, Org. Lett., 2015, 17, 4094–4097 CrossRef CAS.
- (a) J. Zhang, M. Wang, H. Wang, H. Xu, J. Chen, Z. Guo, B. Ma, S.-R. Ban and H.-X. Dai, Chem. Commun., 2021, 57, 8656–8659 RSC; (b) Y. Liu, F. Chang, Q. Jiang, Z. Ma and C. Liu, Synlett, 2017, 658–662 Search PubMed; (c) T. Li, Z. Wang, W. B. Qin and T. B. Wen, ChemCatChem, 2016, 8, 2146–2154 CrossRef CAS.
- (a) Z. Ruan, N. Sauermann, E. Manoni and L. Ackermann, Angew. Chem., Int. Ed., 2017, 56, 3172–3176 CrossRef CAS PubMed; (b) N. Sauermann, M. J. González and L. Ackermann, Org. Lett., 2015, 17, 5316–5319 CrossRef CAS PubMed; (c) N. Kaplaneris, J. Son, L. Mendive-Tapia, A. Kopp, N. D. Barth, I. Maksso, M. Vendrell and L. Ackermann, Nat. Commun., 2021, 12, 3389 CrossRef CAS PubMed.
- (a) Y. Zhao, X. Li, P. Zhou, X. Han, C. Zhang, T. Liang, S. Zhao and Z. Zhang, Org. Lett., 2024, 26, 7285–7290 CrossRef CAS; (b) S. M. Khake, V. Soni, R. G. Gonnade and B. Punji, Chem. – Eur. J., 2017, 23, 2907–2914 CrossRef CAS.
- (a) J. F. Reynes, F. Leon and F. García, ACS Org. Inorg. Au, 2024, 4, 432–470 CrossRef CAS PubMed; (b) N. Fantozzi, J.-N. Volle, A. Porcheddu, D. Virieux, F. García and E. Colacino, Chem. Soc. Rev., 2023, 52, 6680–6714 RSC; (c) X. Yang, C. Wu, W. Su and J. Yu, Eur. J. Org. Chem., 2022, 2022, e202101440 CrossRef CAS; (d) K. J. Ardila-Fierro and J. G. Hernández, ChemSusChem, 2021, 14, 2145–2162 CrossRef CAS PubMed; (e) A. Porcheddu, E. Colacino, L. De Luca and F. Delogu, ACS Catal., 2020, 10, 8344–8394 CrossRef CAS; (f) J. L. Howard, Q. Cao and D. L. Browne, Chem. Sci., 2018, 9, 3080–3094 RSC; (g) J. G. Hernández and C. Bolm, J. Org. Chem., 2017, 82, 4007–4019 CrossRef; (h) S. L. James, C. J. Adams, C. Bolm, D. Braga, P. Collier, T. Friščić, F. Grepioni, K. D. M. Harris, G. Hyett, W. Jones, A. Krebs, J. Mack, L. Maini, A. G. Orpen, I. P. Parkin, W. C. Shearouse, J. W. Steed and D. C. Waddell, Chem. Soc. Rev., 2012, 41, 413–447 RSC; (i) N. K. Narayanan and M. Schnürch, ChemCatChem, 2025, 17, e00457 CrossRef CAS.
- G. N. Hermann, M. T. Unruh, S. H. Jung, M. Krings and C. Bolm, Angew. Chem., Int. Ed., 2018, 57, 10723–10727 CrossRef CAS PubMed.
- (a) P. Zhou, C. Wang, G. Wan, W. Zheng, Z. Wei, T. Liang, J. Jiang and Z. Zhang, J. Org. Chem., 2024, 89, 10953–10964 CrossRef CAS; (b) P. Zhou, X. Liang, Z. Xu, H. Chen, Z. Wei, T. Liang, J. Jiang and Z. Zhang, Chem. Commun., 2024, 60, 6679–6682 RSC; (c) R. Boobalan, P. Gandeepan and C.-H. Cheng, Org. Lett., 2016, 18, 3314–3317 CrossRef CAS PubMed.
- (a) M. L. Schrader, F. R. Schäfer, F. Schäfers and F. Glorius, Nat. Chem., 2024, 16, 491–498 CrossRef CAS; (b) L. Pitzer, F. Schäfers and F. Glorius, Angew. Chem., Int. Ed., 2019, 58, 8572–8576 CrossRef CAS.
- (a) J. Martínez, J. F. Cortés and R. Miranda, Processes, 2022, 10, 1274 CrossRef; (b) M. Tobiszewski, M. Marć, A. Gałuszka and J. Namieśnik, Molecules, 2015, 20, 10928–10946 CrossRef CAS; (c) F. Roschangar, R. A. Sheldon and C. H. Senanayake, Green Chem., 2015, 17, 752–768 RSC; (d) C. R. McElroy, A. Constantinou, L. C. Jones, L. Summerton and J. H. Clark, Green Chem., 2015, 17, 3111–3121 RSC; (e) K. Van Aken, L. Strekowski and L. Patiny, Beilstein J. Org. Chem., 2006, 2, 3 Search PubMed; (f) A. D. Curzons, D. N. Mortimer, D. J. C. Constable and V. L. Cunningham, Green Chem., 2001, 3, 1–6 RSC.
| This journal is © The Royal Society of Chemistry 2025 |

![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Condition A: 1 (0.21 mmol, 1.0 equiv.), 2 (0.42 mmol, 2.0 equiv.), [Ru(
Condition A: 1 (0.21 mmol, 1.0 equiv.), 2 (0.42 mmol, 2.0 equiv.), [Ru(