Open Access Article
Open Access ArticleMicrowave assisted cobalt(III)-catalysed C–H aminocarbonylation reactions with isocyanates for the synthesis of thiophenecarboxamides†
Borja
Taboada-Seras
 ,
Carlos
Santiago
,
Carlos
Santiago
 ,
Eider
Sustatxa
,
Asier
Carral-Menoyo
*,
Nuria
Sotomayor
,
Eider
Sustatxa
,
Asier
Carral-Menoyo
*,
Nuria
Sotomayor
 * and
Esther
Lete
* and
Esther
Lete
 *
*
Departamento de Química Orgánica e Inorgánica, Facultad de Ciencia y Tecnología, Universidad del País, Vasco/Euskal Herriko Unibertsitatea UPV/EHU, Apdo. 644., 48080 Bilbao, Spain. E-mail: nuria.sotomayor@ehu.eus
First published on 25th June 2025
Abstract
A mild and efficient cobalt(III)-catalysed C–H aminocarbonylation procedure for thiophenes and benzo[b]thiophenes under MW assisted conditions has been developed. Site selectivity is controlled by the use of pyridine and pyrimidine directing groups. The reaction is effective with aromatic isocyanates with wide substitution patterns, although aliphatic isocyanates showed less reactivity. The introduced amide group can act as directing group in further iterative C–H functionalization reactions, allowing the diversification of the heterocyclic structures. DFT calculations have shed light on the mechanistic course and reactivity patterns.
Introduction
Amides are ubiquitous structural motifs in natural occurring molecules, pharmaceuticals, agrochemicals1 and polymers or materials.2 In addition, carboxamides play a crucial role in organic synthesis as versatile building blocks3 or directing groups for aromatic metalation4 or transition-metal catalysed C–H functionalization reactions.5 Although many efficient methodologies have been developed for amide synthesis,6 one of their main drawbacks is their low atom-economy. In fact, catalytic amide bond formation7 has been identified by the ACS Green Chemistry Institute Pharmaceutical Roundtable (ACS GCIPR) as a key initiative for green chemistry.8 In this context, transition metal-catalysed C–H activation9 has become an essential synthetic tool to install amide motifs in arenes and heteroarenes, reducing the number of steps and waste generation, as there is no need to use prefunctionalized substrates. The regioselectivity can be controlled by using directing groups and/or ligands or even the substrate and the method presents a high degree of functional group compatibility.10 Thus, efficient protocols for ruthenium-,11 rhodium- or rhenium- catalysed12 C(sp2)–H aminocarbonylation reactions with isocyanates have been reported (Scheme 1A), using different directing groups to control the regioselectivity. In the case of palladium catalysis, two different approaches have been developed. On one hand, the decarboxylative ortho-aminocarbonylation of heteroaromatic carboxylic acids with isothiocyanates has been described, where the carboxyl acts as a traceless directing group. On the other hand, the aminocarbonylation of aryl and heteroaryl C(sp2)–H bonds has been achieved using CO and nitroarenes as the nitrogen source.13Over the past years, cobalt has emerged as one of the most promising earth-abundant first-row metals to replace these classical transition-metals.14 A significant breakthrough in this chemistry has been the development of air-stable high-valent Cp*Co(III) catalysts15 for catalytic generation of nucleophilic Co(III) organometallic species in directed C–H bond functionalization reactions, mainly through electrophilic base-assisted mechanisms, such as CMD.16 Nevertheless, only a few reports have appeared to date describing the Co(III)-catalysed C(sp2)–H aminocarbonylation for the synthesis of aromatic carboxamides and their application to heteroarenes (e.g. thiophene and benzothiophene) has been far less investigated. In 2015 Ellman17 and Ackermann18 independently reported the first Cp*Co(III)-catalysed C–H aminocarbonylation reactions with isocyanates as electrophiles and pyrazole as the directing group, though mainly for the synthesis of benzamides (Scheme 1B). Although Ackermann demonstrated that the isocyanate electrophiles can be generated in situ from the corresponding N-acylazides,18 the use of N-acylazides can also lead to amidation reactions under similar reaction conditions, as reported by Punniyamurthy and col.19 for the Cp*Co(III)-catalysed C–H activation of indoles with a 2-pyrimidine directing group. Related amidation reactions have been reported under Co(III) catalysis using other electrophiles (N-chloroacetamides,20 dioxazolones19,21 or α-benzoylketene dithioacetals22) avoiding Curtius rearrangement. Recently, Ellman23 extended the procedure to cascade reactions between 1,3-dienes and isocyanates for synthesis of α-quaternary amides. Regarding the introduction of amides into heteroarenes, Maji24 developed a thiocarbamate-directed Cp*Co(III)-catalysed aminocarbonylation /cascade annulation of pyrroles and indoles with isocyanates that provide access diverse pyrrolecarboxamides (Scheme 1C). On the other hand, the aminocarbonylation of indoles can also be achieved with amines and CO using low-valent cobalt catalysis.25
Considering the potential and the limitations of amide functional group insertion into (hetero)arenes, we became interested in the Co(III)-catalysed C–H aminocarbonylation reaction for the synthesis of thiophene- and benzo[b]thiophenecarboxamides. The synthesis of N-arylthiophen-3-carboxamides has attracted the interest of synthetic chemists as they have been identified as human dihydroorotate dehydrogenase (DHODH) inhibitors involved in the treatment of autoimmune diseases (rheumatoid arthritis and multiple sclerosis) and cancer therapy.26 The thiophene- and benzo[b]thiophene-2-carboxamide motifs are also essential to overcome cancer chemoresistance.27 Additionally, these derivatives can be used as agrochemical fungicides.28 Structural variations may cause important differences in the mechanism of action of this type of drugs. For example, the introduction of heteroaryl motifs on the thiophene would give heterobiaryl scaffolds as pyridyl- or pyrimidinyl-thiophenecarboxamides with applications both in medicinal chemistry29 and material science.30 This type of biheteroarylcarboxamides have been previously synthetized via palladium-catalysed C-3 arylation reactions of thiophene-2-carboxamides, though a limited number of aromatic substituents (phenyl, 2-methylthiophenyl and quinol-8-yl) have been tested at the amide nitrogen atom.31 The palladium-catalysed C-2 arylation reaction of thiophene-3-carboxamides32 has also allowed the introduction of a pyridyl group, though the procedure required the use of a perfluorotoluimide as directing group. In the context of our interest in Cp*Co(III) catalysed C–H functionalization reactions,33 herein we report a microwave-assisted cobalt(III)-catalysed C–H aminocarbonylation of thiophenes and benzo[b]thiophenes with isocyanates using pyridine or pyrimidine as directing groups to control site selectivity (Scheme 1D). The use of MW irradiation significantly shortens the reaction times compared to standard thermal conditions. The introduced N-arylamide group can act as directing group in further iterative C–H functionalization reactions, allowing the diversification of the heterocyclic structures. Besides, DFT studies have shed light on the reaction mechanism.
Results and discussion
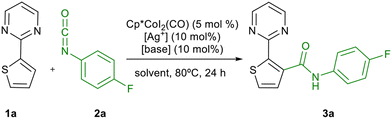
We started studying the reaction of thiophene 1a and 4-fluorophenyl isocyanate 2a as a model, using Cp*CoI2(CO) as catalyst in DCE at 70 °C, based on the reaction conditions described by Ackerman as starting point.18 Under these reaction conditions, the amide 3a was obtained in low yield, recovering unreacted starting material (Table 1, entry 1.). When the temperature was increased to 80 °C and the reaction time was extended to 24 h, the yield increased to 78% (Table 1, entry 2). Next, different bases were tested. When CsOAc and RbOAc were used, amide 3a was obtained in 84% yield (Table 1, entries 3 and 4). The yield dropped when AgTFA or CsOPiv where used, recovering unreacted 1a (Table 1, entries 5 and 6). Full conversion was observed with sodium pivalate, obtaining 3a in a 91% yield (Table 1, entry 7). The use of other silver salts, such as AgNTf2 or AgPF6 did not improve the yield (Table 1, entries 8 and 9), and the reaction did not proceed in presence of silver tetrafluoroborate (Table 1, entry 10). Regarding solvents, the use toluene and trifluorotoluene gave lower yields (Table 1, entries 11 and 12 vs. entry 7).| Entry | Solventa | [Ag+] | [Base] |
3a![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) b (%) b (%) |
|---|---|---|---|---|
| a The reactions were carried out with 1a (0.5 mmol), Cp*CoI2(CO)2 (0.025 mmol), [Ag+] (0.05 mmol), [base] (0.05 mmol) and 2a (1 mmol) in 20 mL sealed reaction tubes inserted in a heating block. The temperature refers to the external temperature of the heating block. b Yield (%) of isolated pure compound. c Unreacted 1a was recovered. d 70 °C, 16 h. e No Cp*Co(CO)I2 was added. | ||||
| 1 | DCE | AgSbF6 | AgOAc | 27c,d |
| 2 | DCE | AgSbF6 | AgOAc | 78c |
| 3 | DCE | AgSbF6 | CsOAc | 84c |
| 4 | DCE | AgSbF6 | RbOAc | 84c |
| 5 | DCE | AgSbF6 | AgTFA | 55c |
| 6 | DCE | AgSbF6 | CsOPiv | 57c |
| 7 | DCE | AgSbF6 | NaOPiv·H2O | 91 |
| 8 | DCE | AgNTf2 | NaOPiv·H2O | 85c |
| 9 | DCE | AgPF6 | NaOPiv·H2O | 65c |
| 10 | DCE | AgBF4 | NaOPiv·H2O | —c |
| 11 | Toluene | AgSbF6 | NaOPiv·H2O | 82c |
| 12 | PhCF3 | AgSbF6 | NaOPiv·H2O | 81c |
| 13 | DCE | AgSbF6 | — | 40 |
| 14 | DCE | — | NaOPiv·H2O | nr |
| 15e | DCE | AgSbF6 | NaOPiv·H2O | nr |
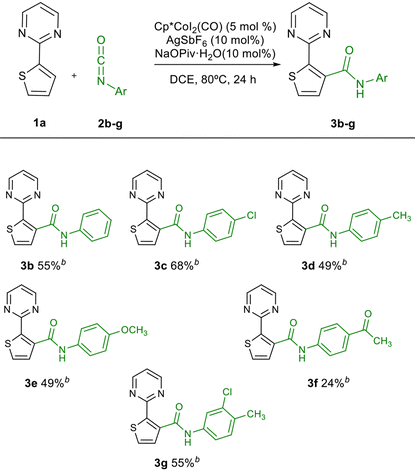
With the optimized reaction conditions in hand, we checked that the carboxylate base has a positive effect on reactivity (Table 1, entry 13), and that the cobalt complex and the silver salt are essential for the C–H activation process (Table 1, entries 14 and 15). Once the optimal reaction conditions were selected, the reaction was extended to aromatic isocyanates 2b–g (Table 2). However, in all cases, lower reactivity was observed for these isocyanates, regardless of the electronic nature of the substituents. The amides 3b–g were obtained in modest yields, recovering unreacted 1a in all cases. At this point, we thought that this would be a suitable scenario to study the effect of MW irradiation on these reactions. The acceleration effect of MW on transition metal-catalysed cross-coupling reactions is well known,34 although the application of MW in C–H activation reactions is still less extended.35 In this context, our group reported recently the microwave-assisted palladium(II)-catalysed C-3 acylation of thiophenes with aldehydes via C(sp2)–H activation.36 Recent examples with Rh(III) catalysts have also been described,37 but this technique has been scarcely applied in Co(III) catalysis.38
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) a
a
After a brief optimisation of the catalyst loading and time, (see Table S1 in ESI†), amide 3a could be obtained in a 90% yield at 100° C in a sealed reaction tube under MW irradiation with a maximum power of 200 W after only 1 hour (Table 3).
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) a
a
| a Yield (%) of pure isolated product. Reactions were done in a 0.5 mmol scale using 10 mL sealed reaction tubes. The indicated temperature, obtained using a maximum power of 200 W, refers to the internal reaction temperature measured by an infrared sensor. b 5 mol% of catalyst was used. c Reaction done in 1 mmol scale. d Reaction done in 2 mmol scale. |
|---|

|
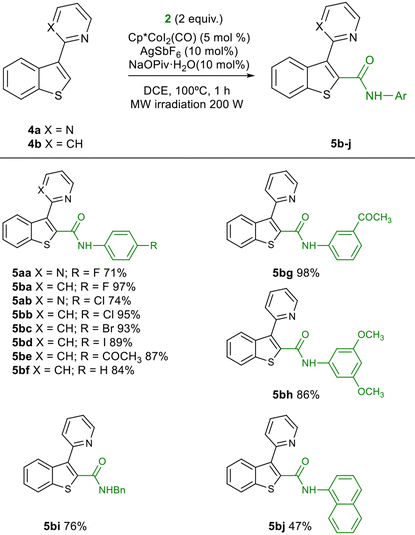
The catalyst loading could be reduced from 5 mol% to 2.5 mol% without a big erosion of the yield (82%). At this point we also checked the use of pyridine as directing group, instead of pyrimidine. 2-(Thiophen-2-yl)pyridine was used as substrate under these reaction conditions, obtaining a significantly lower yield of the corresponding amide (50%, see Scheme S1 in ESI†). The presence of a directing group is also fundamental to obtain the observed reactivity and regioselectivity. In fact, when 2-methyl- and 2-phenylthiophene were tested as substrates, no reaction was observed with 2a under the optimized conditions (see Scheme S2 in ESI†). Considering these results, 1a was used as substrate, and the reaction was extended to a variety of aromatic isocyanates 2b–2u. The reaction works with both electron deficient and electron rich isocyanates, although electron deficient isocyanates were more reactive (for example, 3avs. 3d or 3e), as has been reported previously,18,24 although both 3e and 3f were obtained in low yield using 5 mol% of the catalyst. The meta-substituted isocyanates gave in general good results (3j–3q). The reaction is also compatible with ortho-substituted (3r, 3s) and disubstituted isocyanates (3g, 3s, 3t). Alkyl isocyanates were also reactive, leading to the corresponding amides 3v–3x in moderate yield. In some cases, the yields could be improved using 5 mol% of the catalyst. The reaction could also be scaled up to 1 and 2 mmol (Table 3, 3a) with consistent yields.
The structure of the amides was unambiguously confirmed by X-ray analysis of one of the derivatives (3i, Fig. 1).39
 | ||
| Fig. 1 ORTEP plot of compound 3i with thermal ellipsoids at the 50% probability level (CCDC 2463953†). | ||
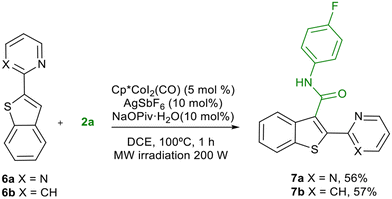
We next extended the reaction for the C-2 aminocarbonylation of benzothiophenes (Table 4). Pyrimidine and pyridine were incorporated as directing groups at C-3 of benzothiophene (4a,b). Pyrimidine resulted an efficient directing group in the reaction of 4a with isocyanates 2a,b, obtaining 5aa and 5ab in good yields under the optimised conditions. However, in this case pyridine was a more efficient directing group, obtaining 5ba and 5bb in almost quantitative yields (Table 4). The reaction was then extended to a variety of isocyanates obtaining the corresponding benzo[b]thiophene-2-carboxamides 5bc to 5bj in generally high yields. The aminocarbonylation in C-3 of the benzothiophene ring was also feasible, introducing the directing group in C-2 (Scheme 2). Under the optimised reaction conditions, both pyrimidine and pyridine behaved as efficient directing groups, leading to moderate yields of amides 7a and 7b.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) a
a
| a Yield (%) of pure isolated product. Reactions were done in a 0.5 mmol scale using 10 mL sealed reaction tubes. The indicated temperature, obtained using a maximum power of 200 W, refers to the internal reaction temperature measured by an infrared sensor. |
|---|
|
With the experimental results in hand, we decided to carry out DTF studies to elucidate the mechanism operating in the reaction. All structures were optimized using density functional theory (DFT) as implemented in Gaussian 16,40 with B3LYP41 as functional, 6-31G(d,p) as basis set for non-metallic atoms, and LANL2DZ42 as basis set for cobalt. Final energies were obtained performing single-point calculations on the previously optimized structures at M0643/6-311++G(d,p) level of theory for non-metallic atoms and SDD basis set for cobalt,44 introducing solvation factors with the IEF-PCM45 method, and 1,2-dichloroethane as solvent. The stationary points were characterized by frequency calculations in order to verify that they have the right number of imaginary frequencies.
For the mechanistic calculations (Fig. 2), we used the cationic complex [Cp*Co(OAc)]+ as starting point, which has been previously proposed as the catalytic active species.46,33b Coordination of this species with a molecule of substrate 1a comes with a stabilizing effect of −14.1 kcal mol−1 to give intermediate A, which can undergo a non-rate-determining and reversible C–H activation step (ΔG‡ = 17.4 kcal mol−1), rendering B. To confirm the reversible character of the C–H activation step, deuteration experiments were carried out, observing a 70% deuterium incorporation when the substrate was subjected to the reaction conditions in the presence of 4-acetylphenyl isocyanate 2f and 10 equivalents of AcOD (Scheme 3). These results are in agreement with related examples.18,24 After this C–H activation process, ligand exchange occurs, releasing a molecule of acetic acid and incorporating phenyl isocyanate 2b coupling partner to generate complex C, which undergoes a rate-determining migratory insertion to the N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bond with an activation energy of 13.4 kcal mol−1, forming intermediate D. After coordination of acetic acid to give E, a fast and irreversible proto-demetalation process (ΔG‡ = 1.2 kcal mol−1) takes place giving complex F. At this point, the desired product 3b is released through a ligand exchange that also regenerates initial complex A.
C bond with an activation energy of 13.4 kcal mol−1, forming intermediate D. After coordination of acetic acid to give E, a fast and irreversible proto-demetalation process (ΔG‡ = 1.2 kcal mol−1) takes place giving complex F. At this point, the desired product 3b is released through a ligand exchange that also regenerates initial complex A.
 | ||
| Fig. 2 Free energy profile of [Cp*CoOAc]+ catalysed C–H aminocarbonylation of 1a with 2b characterised at M06/6-311++G(d,p) level of theory (energy values expressed in kcal mol−1). | ||
With these computational results in hand, the catalytic cycle depicted in Scheme 4 can be proposed for the aminocarbonylation of thiophenes. First of all, [Cp*Co(CO)I2] would undergo ligand exchange with NaOPiv and AgSbF6, present in the reaction media, to form active catalytic species [Cp*Co(OPiv)]+. This species would then coordinate a molecule of substrate 1a. Generated complex I is then proposed to undergo C–H activation, rendering II, which forms III after release of pivalic acid and coordination of the corresponding isocyanate coupling partner. Afterwards, migratory insertion to the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N double bond would occur, leading to intermediate IV. This complex can coordinate a molecule of pivalic acid to form V, which undergoes proto-demetalation. The corresponding amidated product 3 would be released from species VI after coordination of another molecule of 1a, regenerating initial complex I.
N double bond would occur, leading to intermediate IV. This complex can coordinate a molecule of pivalic acid to form V, which undergoes proto-demetalation. The corresponding amidated product 3 would be released from species VI after coordination of another molecule of 1a, regenerating initial complex I.
Finally, we decided to study the use of the introduced N-arylamide group as directing group for further iterative Cp*Co(III) catalysed C–H functionalization reactions, allowing the diversification of the heterocyclic structures. To showcase this possibility, we selected reported allylation, alkylation and alkenylation reactions, using amides 3a,b as substrates (Scheme 5). The N-arylamide group behaved indeed as an efficient directing group for allylation reactions. Using an allyl benzoate as allylating agent under the reaction conditions described by our group,33b or ally carbonate under the conditions described by Glorious,47 the 4-allylated thiophenes were obtained in good yields, significantly higher than the yields obtained for the allylation of thiophenes using N-methyl33b or N,N-dimethyl amides47 as directing groups. C-4 alkylation with N-benzylmaleimide was also possible in high yield, using the conditions previously described.48 Finally, alkenylation with ethyl acrylate was also possible under Matsunaga conditions,49 although in lower yield.
Conclusions
In conclusion, the cobalt(III)-catalysed C–H aminocarbonylation of thiophenes and benzo[b]thiophenes takes place efficiently using pyridine or pyrimidine as directing groups to control site selectivity. The use of MW irradiation significantly shortens the reaction time to 1 h, obtaining generally high yields of the amides. The reaction is effective with aromatic isocyanates with a wide substitution pattern, although aliphatic isocyanates showed less reactivity. DFT mechanistic studies have shown that the reaction proceeds via a non-rate-determining and reversible C–H activation step, followed by coordination with the isocyanate, rate-determining migratory insertion to the N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bond and fast protodemetalation. The introduced N-arylamide group can act as directing group in further iterative C–H functionalization reactions, allowing the diversification of the heterocyclic structures.
C bond and fast protodemetalation. The introduced N-arylamide group can act as directing group in further iterative C–H functionalization reactions, allowing the diversification of the heterocyclic structures.
Author contributions
A. Carral-Menoyo, N. Sotomayor and E. Lete conceptualized and supervised the project. N. Sotomayor and E. Lete wrote the original draft, reviewed, and edited the manuscript. B. Taboada-Seras and C. Santiago did the major chemical experimental part and wrote the first experimental draft of ESI.† E. Sustatxa participated in the chemical experimental work. A. Carral-Menoyo was responsible for the computational study. N. Sotomayor and E. Lete were responsible for funding acquisition. All authors have read and agreed to the published version of the manuscript.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the ESI.†Acknowledgements
Ministerio de Ciencia e Innovación (PID2022-137365NB-100 funded by MCIN/AEI/10.13039/501100011033) and Gobierno Vasco (IT1558-22) are gratefully acknowledged for their financial support. Technical and human support provided by Servicios Generales de Investigación SGIker (UPV/EHU, MINECO, GV/EJ, ERDF, EU and ESF) is also acknowledged.References
- C. Lamberth and J. Dinges, Different Roles of Carboxylic Functions in Pharmaceuticals and Agrochemicals, in Bioactive Carboxylic Compound Classes: Pharmaceuticals and Agrochemicals, ed. C. Lamberth and J. Dinges, Wiley, Weinheim, 2016, ch. 1, pp. 1–11 Search PubMed.
- For some representative reviews, see: (a) X. Guo, A. Facchetti and T. J. Marks, Imide- and Amide-Functionalized Polymer Semiconductors, Chem. Rev., 2014, 114, 8943–9021 CrossRef CAS PubMed; (b) K. Marchildon, Polyamides - still strong after seventy years, Macromol. React. Eng., 2011, 5, 22–54 CrossRef CAS.
- See for example: (a) X. B. Pang, P. F. Su and X. Z. Shu, Reductive Cross-Coupling of Unreactive Electrophiles, Acc. Chem. Res., 2022, 55, 2491–2509 CrossRef CAS PubMed; (b) C. W. Liu and M. Szostak, Decarbonylative cross-coupling of amides, Org. Biomol. Chem., 2018, 16, 7998–8010 RSC; (c) S. L. MacNeil, M. Gray, D. G. Gusev, L. E. Briggs and V. Snieckus, Carbanionic Friedel-Crafts Equivalents. Regioselective Directed Ortho, and Remote Metalation-C-N Cross Coupling Routes to Acridones and Dibenzo[b,f]azepinones, J. Org. Chem., 2008, 73, 9710–9719 CrossRef CAS PubMed; (d) N. Sotomayor and E. Lete, Aryl and heteroaryllithium compounds by metal-halogen exchange. Synthesis of carbocyclic and heterocyclic systems, Curr. Org. Chem., 2003, 7, 275–300 CrossRef CAS.
- For some representative reviews, see: (a) R. D. Jansen-van Vuuren, S. S. A. Liu, M. A. J. Miah, J. Cerkovnik, J. Kosmrlj and V. Snieckus, The Versatile and Strategic O-Carbamate Directed Metalation Group in the Synthesis of Aromatic Molecules: An Update, Chem. Rev., 2024, 124, 7731–7828 CrossRef CAS PubMed; (b) D. Tilly, J. Magolan and J. Mortier, Directed Remote Aromatic Metalations: Mechanisms and Driving Forces, Chem. – Eur. J., 2012, 18, 3804–3820 CrossRef CAS PubMed; (c) V. Snieckus, Directed Ortho Metalation - Tertiary Amide and O-Carbamate Directors in Synthetic Strategies For Polysubstituted Aromatics, Chem. Rev., 1990, 90, 879–933 CrossRef CAS.
- (a) R. Thakur, Y. Jaiswal and A. Kumar, Primary amides: Sustainable weakly coordinating groups in transition metal-catalyzed C–H bond functionalization reactions, Tetrahedron, 2021, 93, 132313 CrossRef CAS; (b) Q. Zheng, C.-F. Liu, J. Chen and G.-W. Rao, C–H Functionalization of Aromatic Amides, Adv. Synth. Catal., 2020, 362, 1406–1446 CrossRef CAS; (c) D. D. Subhedar, A. A. Mishra and B. M. Bhanage, N-Methoxybenzamide: A Versatile Directing Group for Palladium-, Rhodium- and Ruthenium-Catalyzed C−H Bond Activations, Adv. Synth. Catal., 2019, 361, 4149–4195 CrossRef CAS; (d) R. Das, G. S. Kumar and M. Kapur, Amides as Weak Coordinating Groups in Proximal C–H Bond Activation, Eur. J. Org. Chem., 2017, 5439–5459 CrossRef CAS; (e) R.-Y. Zhu, M. E. Farmer, Y.-Q. Chen and J.-Q. Yu, A Simple and Versatile Amide Directing Group for C-H Functionalizations, Angew. Chem., Int. Ed., 2016, 55, 10578–10599 CrossRef CAS PubMed; G. Rouquet and N. Chatani, Catalytic Functionalization of C(sp2)-H and C(sp3)-H Bonds by Using Bidentate Directing Groups, Angew. Chem., Int. Ed., 2013, 52, 11726–11743 Search PubMed; (f) M. Engle, T.-S. Mei, M. Wasa and J.-Q. Yu, Weak Coordination as a Powerful Means for Developing Broadly Useful C–H Functionalization Reactions, Acc. Chem. Res., 2012, 45, 788–802 CrossRef PubMed.
- (a) R. M. de Figueiredo, J.-S. Suppo and J.-M. Campagne, Nonclassical Routes to Amide Bond Formation, Chem. Rev., 2016, 116, 12029–12122 CrossRef CAS PubMed; (b) R. M. Lanigan and T. D. Sheppard, Recent Developments in Amide Synthesis: Direct Amidation of Carboxylic Acids and Transamidation Reactions, Eur. J. Org. Chem., 2013, 7453–7465 CrossRef CAS; (c) C. A. G. N. Montalbetti and V. Falque, Amide Bond Formation and Peptide Coupling, Tetrahedron, 2005, 61, 10827–10852 CrossRef CAS.
- (a) E. Massolo, M. Pirola and M. Benaglia, Amide Bond Formation Strategies: Latest Advances on a Dateless Transformation, Eur. J. Org. Chem., 2020, 4641–4651 CrossRef CAS; (b) H. Lundberg, F. Tinnis, N. Selander and H. Adolfsson, H. Catalytic amide formation from non-activated carboxylic acids and amines, Chem. Soc. Rev., 2014, 43, 2714–2742 RSC; (c) V. R. Pattabiraman and J. Bode, Rethinking amide bond synthesis, Nature, 2011, 480, 471–479 CrossRef CAS PubMed; (d) S. D. Roughley and A. M. Jordan, The Medicinal Chemist's Toolbox: An Analysis of Reactions Used in the Pursuit of Drug Candidates, J. Med. Chem., 2011, 54, 3451–3479 CrossRef CAS PubMed; (e) E. Valeur and M. Bradley, Amide bond formation: beyond the myth of coupling reagents, Chem. Soc. Rev., 2009, 38, 606–631 RSC.
- D. J. C. Constable, P. J. Dunn, J. D. Hayler, G. R. Humphrey, J. L. Leazer Jr, R. J. Linderman, K. Lorenz, J. Manley, B. A. Pearlman, A. Wells, A. Zaks and T. Y. Zhang, Key green chemistry research areas-a perspective from pharmaceutical manufacturers, Green Chem., 2007, 9, 411–420 RSC.
- (a) Handbook of C-H Functionalization, ed. D. Maiti, Wiley-VCH, Weinheim, 2023 Search PubMed; (b) F. Dalton, T. Faber and F. Glorius, C−H Activation: Toward Sustainability and Applications, ACS Cent. Sci., 2021, 7, 245–261 CrossRef PubMed; (c) A. Lei, W. Shi, W. Liu, H. Zhang and C. He, Oxidative Cross-Coupling Reactions, Wiley-VCH, Weinheim, 2017 Search PubMed; (d) Science of Synthesis. Cross-Coupling and Heck-type Reactions 3. Metal-Catalyzed Heck-Type Reactions and C−H Couplings via C−H Activation, ed. M. Larhed, Georg Thieme Verlag, Stuttgart, Workbench edn, 2013 Search PubMed; (e) J. Wencel-Delord and F. Glorius, C–H bond activation enables the rapid construction and late-stage diversification of functional molecules, Nat. Chem., 2013, 5, 369–375 CrossRef CAS PubMed; (f) C. Jia, T. Kitamura and Y. Fujiwara, Catalytic functionalization of arenes and alkanes via C−H bond activation, Acc. Chem. Res., 2001, 34, 633–639 CrossRef CAS PubMed.
- (a) C. Sambiagio, D. Schönbauer, R. Blieck, G. Dao-Huy, T. Pototschnig, P. Schaaf, T. Wiesinger, M. F. Zia, J. Wencel-Delord, T. Besset, B. U. W. Maes and M. Schnürch, A comprehensive overview of directing groups applied in metal-catalysed C–H functionalisation chemistry, Chem. Soc. Rev., 2018, 47, 6603–6743 RSC; (b) M. Beller and X. F. Wu, Transition metal catalyzed carbonylation reactions, Springer, Heidelberg, 2013 CrossRef.
- (a) E. Y. Lai, B. Yuan, L. Ackermann and M. J. Johansson, Ruthenium(II)-Catalyzed aminocarbonylation with Isocyanates through weak coordinating groups, Chem. – Eur. J., 2023, 29, e202302023 CrossRef CAS PubMed; (b) L. Lin, X.-J. Zhang, X. Xu, Y. Zhao and Z. Shi, Ru3(CO)12-Catalyzed Modular Assembly of Hemilabile Ligands by C-H Activation of Phosphines with Isocyanates, Angew. Chem., Int. Ed., 2023, 62, e202214584 CrossRef CAS PubMed; (c) P. Karishma, A. Gogia, S. K. Mandal and R. Sakhuja, Ruthenium Catalyzed C-H amidation and Carbocyclization using Isocyanates: An Access to Amidated 2-phenylphthalazine-1,4-diones and Indalolo[1,2-b]phthalazine-triones, Adv. Synth. Catal., 2021, 363, 762–775 CrossRef CAS; (d) S. De Sakar and L. Ackermann, Ruthenium(II)-Catalyzed C-H Activation with Isocyanates: A Versatile Route to Phthalimides, Chem. – Eur. J., 2014, 20, 13932–13936 CrossRef PubMed; (e) For an example of substrate control, see: S. M. A. Shakoor, S. Kumari, S. Khullar, S. K. Mandal, A. Kumar and R. Sakhuja, Ruthenium(II)-Catalyzed Regioselective Ortho Amidation of Imidazo Heterocycles with Isocyanates, J. Org. Chem., 2016, 81, 12340–12349 CrossRef CAS PubMed.
- (a) Y. Chen, Z. Li, Y. Xu, T. Huang, L. Hai, R. Nie and Y. Wu, Access to Amides and Lactams via Pyridotriazole as a Transformable Directing Group, J. Org. Chem., 2023, 88, 15597–15607 CrossRef CAS PubMed; (b) T. Jeong, S. H. Lee, N. K. Mishra, U. De, J. Park, P. Dey, J.-H. Kwak, Y. H. Jung, H. S. Kim and I. S. Kim, Synthesis and Cytotoxic Evaluation of N-Aroylureas through Rhodium(III)-Catalyzed C−H Functionalization of Indolines with Isocyanates, Adv. Synth. Catal., 2017, 359, 2329–2336 CrossRef CAS; (c) J. Xu, N. Sharma, U. K. Sharma, Z. Li, G. Song and E. V. Van der Eycken, Cationic Rhodium(III)-Catalyzed Direct C-2 Carboxamidation of Indoles with Isocyanates via C-H Bond Functionalization, Adv. Synth. Catal., 2015, 357, 2615–2621 CrossRef CAS; (d) Y. Kuninobu, Y. Tokunaga and K. Takai, Rhenium-catalyzed Amidation of Heteroaromatic Compounds via C–H Bond Activation, Chem. Lett., 2007, 36, 872–873 CrossRef CAS; (e) Y. Kuninobu, K. Kikuchi, Y. Tokunaga, Y. Nishina and K. Takai, Hydroarylation of acetylenes, acrylates, and isocyanates with heteroaromatic compounds under rhenium catalysis, Tetrahedron, 2008, 64, 5974–5981 CrossRef CAS.
- (a) R. N. P. Tulichala, M. Shankar and K. C. K. Swamy, Palladium-Catalyzed Decarboxylative ortho-Amidation of Indole-3-carboxylic Acids with Isothiocyanates Using Carboxyl as a Deciduous Directing Group, J. Org. Chem., 2018, 83, 4375–4383 CrossRef CAS PubMed; (b) F. Zhou, D.-S. Wang, X. Guan and T. G. Driver, Nitroarenes as the Nitrogen Source in Intermolecular Palladium-Catalyzed Aryl C–H Bond Aminocarbonylation Reactions, Angew. Chem., Int. Ed., 2017, 56, 4530–4534 CrossRef CAS PubMed.
- For selected reviews, see: (a) R. Mandal, B. Garai and B. Sundararaju, Weak-Coordination in C-H Bond Functionalizations Catalyzed by 3d Metals, ACS Catal., 2022, 12, 3452–3506 CrossRef CAS; (b) L. Lukasevics, A. Cizikovs and L. Grigorjeva, C-H bond functionalization by high-valent cobalt catalysis: current progress, challenges and future perspectives, Chem. Commun., 2021, 57, 10827–10841 RSC; (c) P. Gandeepan, T. Müller, D. Zell, G. Cera, S. Warratz and L. Ackermann, 3d Transition Metals for C-H Activation, Chem. Rev., 2019, 119, 2192–2452 CrossRef CAS PubMed; (d) O. Planas, C. J. Whiteoak and X. Ribas, Recent Advances in Cobalt-Catalyzed Cross-Coupling Reactions, in Non-Noble Metal Catalysis: Molecular Approaches and Reactions, ed. R. J. M. K. Gebbink and M.-E. Moret, Wiley-VCH, Weinheim, 2019, pp. 297–328 Search PubMed; (e) A. Baccalini, S. Vergura, P. Dolui, G. Zanoni and D. Maiti, Recent advances in cobalt-catalysed C-H functionalizations, Org. Biomol. Chem., 2019, 17, 10119–10141 RSC; (f) O. Planas, P. G. Chirila, C. J. Whiteoak and X. Ribas, Current Mechanistic Understanding of Cobalt-Catalyzed C-H Functionalization, in Advances in Organometallic Chemistry, ed. P. Pérez, Academic Press, San Diego, 2018, vol. 69, pp. 209–282 Search PubMed; (g) M. Moselage, J. Li and L. Ackermann, Cobalt-catalyzed C-H activation, ACS Catal., 2016, 6, 498–525 CrossRef CAS.
- T. Yoshino, H. Ikemoto, S. Matsunaga and M. Kanai, A Cationic High-Valent Cp*CoIII Complex for the Catalytic Generation of Nucleophilic Organometallic Species: Directed C-H Bond Activation, Angew. Chem., Int. Ed., 2013, 52, 2207–2211 CrossRef CAS PubMed.
- (a) A. Carral-Menoyo, N. Sotomayor and E. Lete, Cp*Co(III)-Catalyzed C-H Hydroarylation of Alkynes and Alkenes and Beyond: A Versatile Synthetic Tool, ACS Omega, 2020, 5, 24974–24993 CrossRef CAS PubMed; (b) J. Ghorai and P. Anbarasan, Developments in Cp*CoIII-Catalyzed C-H Bond Functionalizations, Asian J. Org. Chem., 2019, 8, 430–455 CrossRef CAS; (c) T. Yoshino and S. Matsunaga, Cp*CoIII-Catalyzed C–H Functionalization and Asymmetric Reactions Using External Chiral Sources, Synlett, 2019, 1384–1400 CAS; (d) T. Yoshino and S. Matsunaga, (Pentamethylcyclopentadienyl)-cobalt(III)-Catalyzed C−H Bond Functionalization: From Discovery to Unique Reactivity and Selectivity, Adv. Synth. Catal., 2017, 359, 1245–1262 CrossRef CAS; (e) T. Yoshino and S. Matsunaga, High-valent cobalt-catalyzed CH bond functionalization, Adv. Organomet. Chem., 2017, 68, 197–247 CrossRef.
- J. R. Hummel and J. A. Ellman, Cobalt(III)-Catalyzed C–H Bond Amidation with Isocyanates, Org. Lett., 2015, 17, 2400–2403 CrossRef CAS PubMed.
- J. Li and L. Ackermann, Cobalt(III)-Catalyzed Aryl and Alkenyl C-H Aminocarbonylation with Isocyanates and Acyl Azides, Angew. Chem., Int. Ed., 2015, 54, 8551–8554 CrossRef CAS PubMed.
- T. A. Shah, P. B. De, S. Pradhan, S. Banerjee and T. Punniyamurthy, Cp*Co(III)-Catalyzed Regioselective C2 Amidation of Indoles Using Acyl Azides, J. Org. Chem., 2019, 84, 16278–16285 CrossRef CAS PubMed.
- V. V. Nagesh and A. B. Pawar, Harnessing Dual Reactivity of N-Chloroamides for Cascade C–H Amidation/Chlorination of Indoles under Cobalt-Catalysis: Overriding Hofmann Rearrangement Pathway Leading to Aminocarbonylation, Org. Lett., 2024, 26(49), 10523–10528 CrossRef CAS PubMed.
- B. Khan, V. Dwivedi and B. Sundararaju, Cp*Co(III)-Catalyzed o-Amidation of Benzaldehydes with Dioxazolones Using Transient Directing Group Strategy, Adv. Synth. Catal., 2020, 362, 1195–1200 CrossRef CAS.
- Q. Jia, L. Kong and X. Li, Cobalt(III)-catalyzed C–H amidation of weakly coordinating sulfoxonium ylides and α-benzoylketene dithioacetal, Org. Chem. Front., 2019, 6, 741–745 RSC.
- J. Yeo, J. P. Tassone and J. A. Ellman, Synthesis of α-Quaternary Amides via Cp*Co(III)-Catalyzed Sequential C-H Bond Addition to 1,3-Dienes and Isocyanates, Org. Lett., 2024, 26, 9769–9774 CrossRef CAS PubMed.
- S. Saha and M. S. Maji, Cp*Co(III)-catalyzed thiocarbamate-directed C–H aminocarbonylation, amination, and cascade annulation of pyrroles, Chem. Commun., 2022, 58, 10865–10868 RSC.
- (a) P. Wei, J. Ying and X.-F. Wu, Cobalt(II)-Catalyzed Intermolecular Aminocarbonylation of Indoles with Amines, Org. Lett., 2023, 25, 7700–7704 CrossRef CAS PubMed; (b) Q. Gao, J.-M. Lu, L. Yao, S. Wang, J. Ying and X.-F. Wu, Cobalt-Catalyzed Direct C–H Carbonylative Synthesis of Free (NH)-Indolo[1,2-a]quinoxalin-6(5H)-ones, Org. Lett., 2021, 23, 178–182 CrossRef CAS PubMed.
- (a) J. Leban, M. Kralik, J. Mies, R. Baumgartner, M. Gassen and S. Tasler, Biphenyl-4-ylcarbamoyl thiophene carboxylic acids as potent DHODH inhibitors, Bioorg. Med. Chem. Lett., 2006, 16, 267–270 CrossRef CAS PubMed; (b) Y. Zhou, L. Tao, X. Zhou, Z. Zuo, J. Gong, X. Liu, Y. Zhou, C. Liu, N. Sang, H. Liu, J. Zou, K. Gou, X. Yang and Y. Zhao, DHODH and cancer: promising prospects to be explored, Cancer Metab., 2021, 9, 22 CrossRef PubMed.
- (a) Archna, S. Pathania and P. A. Chawla, Thiophene-based derivatives as anticancer agents: An overview on decade's work, Bioorg. Chem., 2020, 101, 104026 CrossRef CAS PubMed; (b) R. Mudududdla, S. K. Guru, A. Wani, S. Sharma, P. Joshi, R. A. Vishwakarma, A. Kumar, S. Bhushan and S. B. Bharate, 3-(Benzo[d,][1,3]dioxol-5-ylamino)-N-(4-fluorophenyl)thiophene-2-carboxamide overcomes cancer chemoresistance via inhibition of angiogenesis and P-glycoprotein efflux pump activity, Org. Biomol. Chem., 2015, 13, 4296–4309 RSC.
- (a) G. A. White and S. G. Georgopoulos, Target Sites of Carboxamides, in Target Sites of Fungicide Action, ed. W. Koeller, CRC Press, Boca Raton, 2018, ch. 1, pp. 1–29 Search PubMed; (b) G. A. White and S. G. Georgopoulos, Thiophene carboxamide fungicides: Structure-activity relationships with the succinate dehydrogenase complex from wild-type and carboxin- resistant mutant strains of Aspergillus nidulans, Pestic. Biochem. Physiol., 1986, 25, 188–204 CrossRef CAS.
- (a) S. Pathania, R. K. Narang and R. K. Rawal, Role of sulphur-heterocycles in medicinal chemistry: An update, Eur. J. Med. Chem., 2019, 180, 486–508 CrossRef CAS PubMed; (b) R. Shah and P. K. Verma, Therapeutic importance of synthetic thiophene, Chem. Cent. J., 2018, 12, 137 CrossRef CAS PubMed; (c) C. M. Adams, D. Belanger, D. Bevan, T. Ehara, L. Ferrara, N. Ji, M. Kato, D. Liu, E. Meredith, M. Mogi, J. J. Powers and G. Prasanna, Thiophen-2-yl-pyridin-2-yl-1H-pyrazole-4-carboxylic acid derivatives and the use thereof as soluble guanylate cyclase activators, WO2016001876A1, 2016, Novartis AG . Search PubMed; (d) H. K. Patel, M. I. Siklos, H. Abdelkarim, E. L. Mendonca, A. Vaidya, P. A. Petukhov and G. R. J. Thatcher, A Chimeric SERM–Histone Deacetylase Inhibitor Approach to Breast Cancer Therapy, ChemMedChem, 2014, 9, 602–613 CrossRef CAS PubMed; (e) K. Ester, M. Hranjec, I. Piantanida, I. Caleta, I. Jarak, K. Pavelic, M. Kralj and G. Karminski-Zamola, Novel derivatives of pyridylbenzo[b]thiophene-2-carboxamides and benzo[b]thieno[2,3-c]naphthyridin-2-ones: minor structural variations provoke major differences of antitumor action mechanisms, J. Med. Chem., 2009, 52, 2482–2492 CrossRef CAS PubMed; (f) J. F. dit Chabert, B. Marquez, L. Neville, L. Joucla, S. Broussous, P. Bouhours, E. David, S. Pellet-Rostaing, B. Marquet, N. Moreau and M. Lemaire, Synthesis and evaluation of new arylbenzo[b]thiophene and diarylthiophene derivatives as inhibitors of the NorA multidrug transporter of Staphylococcus aureus, Bioorg. Med. Chem., 2007, 15, 4482–4497 CrossRef PubMed; (g) P. D. Dominic, P. J. John and I. K. Kenneth, Preparation of heterocyclylcarbonylanthranilic acid derivatives as agrochemical fungicides, WO9616954A1, 1996, Agrevo UK Limited Search PubMed.
- (a) Y. Gui, Y. Zhao, X. Li, T. Liang, S. Zhao and Z. Zhang, Catalyst-Controlled Regiodivergent C−H Alkynylation of 2-Pyridylthiophenes, Adv. Synth. Catal., 2025, 367, e20240085 CrossRef, and references therein. (b) L. Ding, Z.-D. Yu, X.-Y. Wang, Z.-F. Yao, Y. Lu, C.-Y. Yang, J.-Y. Wang and J. Pei, Polymer Semiconductors: Synthesis, Processing, and Applications, Chem. Rev., 2023, 123, 7421–7497 CrossRef CAS PubMed; (c) W. Gao, Y. Wan, Z. Zhang, H. Wu, T. Liu and G. Zhang, The Hofmann reaction involving annulation of o-(pyridin-2-yl)aryl amides selectively and rapidly leads to potential photocatalytically active 6H-pyrido[1,2-c]quinazolin-6-one derivatives, Green Chem., 2020, 22, 7955–7961 RSC; (d) M.-H. Chen, F.-X. Wang, J.-J. Cao, C.-P. Tan, L.-N. Ji and Z.-W. Mao, Light-Up Mitophagy in Live Cells with Dual-Functional Theranostic Phosphorescent Iridium(III) Complexes, ACS Appl. Mater. Interfaces, 2017, 9, 13304–13314 CrossRef CAS PubMed; (e) R. Kroon, A. Diaz de Zerio-Mendaza, S. Himmelberger, J. Bergqvist, O. Backe, G. C. Faria, F. Gao, A. Obaid, W. Zhuang, D. Gedefaw, E. Olsson, O. Inganas, A. Salleo, C. Muller and M. R. Andersson, A New Tetracyclic Lactam Building Block for Thick, Broad-Bandgap Photovoltaics, J. Am. Chem. Soc., 2014, 136, 11578–11581 CrossRef CAS PubMed; (f) L. Flamigni, A. Barbieri, C. Sabatini, B. Ventura and F. Barigelletti, Photochemistry and Photophysics of Coordination Compounds: Iridium, Top. Curr. Chem., 2007, 281, 143–203 CrossRef CAS; (g) A. Tsuboyama, H. Iwawaki, M. Furugori, T. Mukaide, J. Kamatani, S. Igawa, T. Moriyama, S. Miura, T. Takiguchi, S. Okada, M. Hoshino and K. Ueno, Homoleptic Cyclometalated Iridium Complexes with Highly Efficient Red Phosphorescence and Application to Organic Light-Emitting Diode, J. Am. Chem. Soc., 2003, 125, 12971–12979 CrossRef CAS PubMed.
- (a) K. S. Larbi, H. Y. Fu, N. Laidaoui, K. Beydoun, A. Miloudi, D. E. Abed, S. Djabbar and H. Doucet, Palladium-Based Catalytic System for the Direct C3-Arylation of Furan-2-carboxamides and Thiophene-2-carboxamides, ChemCatChem, 2012, 4, 815–823 CrossRef; (b) R. Padmavathi, R. Sankar, B. Gopalakrishnan, R. Parella and S. A. Babu, Pd(OAc)2/A gOAc Catalytic System Based Bidentate Ligand Directed Regiocontrolled C–H Arylation and Alkylation of the C-3 Position of Thiophene- and Furan-2-carboxamides, Eur. J. Org. Chem., 2015, 3727–3742 CrossRef CAS.
- M. H. Daniels, J. R. Armand and K. L. Tan, Sequential Regioselective C−H Functionalization of Thiophenes, Org. Lett., 2016, 18, 3310–3313 CrossRef CAS PubMed.
- (a) A. Carral-Menoyo, N. Sotomayor and E. Lete, Amide-Directed Intramolecular Co(III)-Catalyzed C–H Hydroarylation of Alkenes for the Synthesis of Dihydrobenzofurans with a Quaternary Center, J. Org. Chem., 2020, 85, 10261–10270 CrossRef CAS PubMed; (b) A. Carral-Menoyo, I. Barbolla, C. Santiago, M. Espinel, N. Sotomayor, E. Gómez-Bengoa and E. Lete, Directed C−H Allylation of Aromatic Carboxamides with Allyl Aryl Ethers under Cp*Co(III)-Catalysis, Eur. J. Org. Chem., 2024, e202301090 CrossRef CAS.
- For some reviews, see, for example; (a) P. V. Mehta and E. V. Van der Eycken, Microwave-assisted C–C bond forming cross-coupling reactions: an overview, Chem. Soc. Rev., 2011, 40, 4925–4936 RSC; (b) J. P. Soni, K. S. Chemitikanti, S. V. Joshi and N. Shankaraia, The microwave-assisted syntheses and applications of non-fused single-nitrogen-containing heterocycles, Org. Biomol. Chem., 2020, 18, 9737–9761 RSC; (c) M. Rahman, S. Ghosh, D. Bhattacherjee, G. V. Zyryanov, A. K. Bagdi and A. Hajra, Recent Advances in Microwave-assisted Cross-Coupling Reactions, Asian J. Org. Chem., 2022, 11, e202200179 CrossRef CAS.
- P. C. Dhanush, P. V. Saranya and G. Anilkumar, Microwave assisted C-H activation reaction: An overview, Tetrahedron, 2022, 105, 132614 CrossRef CAS.
- C. Santiago, X. Jiménez-Aberasturi, E. Leicea, M. G. Lete, N. Sotomayor and E. Lete, Microwave-assisted palladium catalysed C–H acylation with aldehydes: synthesis and diversification of 3-acylthiophenes, Org. Biomol. Chem., 2022, 20, 852–861 RSC.
- See, for instance: (a) N. Sharma, V. Bahadur, U. K. Sharma, D. Saha, Z. Li, Y. Kumar, J. Colaers, B. K. Singh and E. V. Van der Eycken, Diversification of Peptidomimetics and Oligopeptides through Microwave-Assisted Rhodium(III)-Catalyzed Intramolecular Annulation, Adv. Synth. Catal., 2019, 361, 4442 CrossRef; (b) H.-R. Chen, I. J. Barve, H.-S. Hsieh, K.-M. Liu, G. P. Pawar and C.-M. Suna, Microwave-Assisted Rhodium(III)-Catalyzed [3+3] Annulation of 2-Benzyl-2H-Indazole-6-carboxylic Acids with Iodonium Ylides: A Regioselective Synthesis of Indazole-Fused Chromenes, Adv. Synth. Catal., 2025, 367, e202400756 CrossRef CAS.
- For an example of a related amidation reaction, see: J. Huang, Y. Huang, T. Wang, Q. Huang, Z. Wang and Z. Chen, Microwave-Assisted Cp*CoIII-Catalyzed C−H Activation/Double C−N Bond Formation Reactions to Thiadiazine 1-Oxides, Org. Lett., 2017, 19, 1128–1131 CrossRef CAS PubMed.
- CCDC 2463953†contains the supplementary crystallographic data for 3i.
- M. J. Frisch, G. W. Trucks, H. B. Schlegel, G. E. Scuseria, M. A. Robb, J. R. Cheeseman, G. Scalmani, V. Barone, G. A. Petersson, H. Nakatsuji, X. Li, M. Caricato, A. V. Marenich, J. Bloino, B. G. Janesko, R. Gomperts, B. Mennucci, H. P. Hratchian, J. V. Ortiz, A. F. Izmaylov, J. L. Sonnenberg, D. Williams-Young, F. Ding, F. Lipparini, F. Egidi, J. Goings, B. Peng, A. Petrone, T. Henderson, D. Ranasinghe, V. G. Zakrzewski, J. Gao, N. Rega, G. Zheng, W. Liang, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, T. Vreven, K. Throssell, J. A. Jr Montgomery, J. E. Peralta, F. Ogliaro, M. J. Bearpark, J. J. Heyd, E. N. Brothers, K. N. Kudin, V. N. Staroverov, T. A. Keith, R. Kobayashi, J. Normand, K. Raghavachari, A. P. Rendell, J. C. Burant, S. S. Iyengar, J. Tomasi, M. Cossi, J. M. Millam, M. Klene, C. Adamo, R. Cammi, J. W. Ochterski, R. L. Martin, K. Morokuma, O. Farkas, J. B. Foresman and D. J. Fox, Gaussian 16, Revision C.01, Gaussian, Inc., Wallingford CT, 2016 Search PubMed.
- (a) C. Lee, W. Yang and R. G. Parr, Development of the Colle-Salvetti correlation-energy formula into a functional of the electron density, Phys. Rev. B:Condens. Matter Mater. Phys., 1988, 37, 785–789 CrossRef CAS PubMed; (b) A. D. Becke, Density–functional thermochemistry. III. The role of exact exchange, J. Chem. Phys., 1993, 98, 5648–5652 CrossRef CAS; (c) W. Kohn, A. D. Becke and R. G. Parr, Density Functional Theory of Electronic Structure, J. Phys. Chem., 1996, 100, 12974–12980 CrossRef CAS.
- T. H. Dunning Jr and P. J. Hay, in Modern Theoretical Chemistry, ed. H. F. Schaefer III, Plenum, New York, 1977, vol. 3, pp. 1–28 Search PubMed.
- Y. Zhao and D. G. Truhlar, The M06 suite of density functionals for main group thermochemistry, thermochemical kinetics, noncovalent interactions, excited states, and transition elements: two new functionals and systematic testing of four M06-class functionals and 12 other functionals, Theor. Chem. Acc., 2008, 120, 215–241 Search PubMed.
- (a) M. Dolg, U. Wedig, H. Stoll and H. Preuss, Energy-adjusted ab initio pseudopotentials for the first-row transition elements, J. Chem. Phys., 1987, 86, 866–872 CrossRef CAS; (b) D. Andrae, U. Häußermann, M. Dolg, H. Stoll and H. Preuß, Energy-adjusted ab initio pseudopotentials for the second and third row transition elements, Theor. Chim. Acta, 1990, 77, 123–141 CrossRef CAS.
- (a) E. Cancés, B. Mennucci and J. Tomasi, A new integral equation formalism for the polarizable continuum model: Theoretical background and applications to isotropic and anisotropic dielectrics, J. Chem. Phys., 1997, 107, 3032–3041 CrossRef; (b) M. Cossi, V. Barone, B. Mennucci and J. Tomasi, Ab initio study of ionic solutions by a polarizable continuum dielectric model, Chem. Phys. Lett., 1998, 286, 253–260 CrossRef CAS; (c) J. Tomasi, B. Mennucci and E. Cancès, The IEF version of the PCM solvation method: an overview of a new method addressed to study molecular solutes at the QM ab initio level, J. Mol. Struct.:THEOCHEM, 1999, 464, 211–226 CrossRef CAS.
- For selected examples, see: (a) P. G. Chirila, J. Adams, A. Dirjal, A. Hamilton and J. C. Whiteoak, Cp*Co(III)-Catalyzed Coupling of Benzamides with α,β-Unsaturated Carbonyl Compounds: Preparation of Aliphatic Ketones and Azepinones, Chem. – Eur. J., 2018, 24, 3584–3589 CrossRef CAS PubMed; (b) A. Kenny, A. Pisarello, A. Bird, P. G. Chirila, A. Hamilton and C. J. Whiteoak, A challenging redox neutral Cp*Co(III)-catalysed alkylation of acetanilides with 3-buten-2-one: synthesis and key insights into the mechanism through DFT calculations, Beilstein J. Org. Chem., 2018, 14, 2366–2374 CrossRef CAS PubMed.
- T. Gensch, S. Vásquez-Céspedes, D.-G. Yu and F. Glorius, Cobalt(III)-Catalyzed Directed C−H Allylation, Org. Lett., 2015, 17, 3714–3717 CrossRef CAS PubMed.
- (a) Z. Zhang, S. Han, M. Tang, L. Ackermann and J. Li, C−H Alkylations of (Hetero)Arenes by Maleimides and Maleate Esters through Cobalt(III) Catalysis, Org. Lett., 2017, 19, 3315–3318 CrossRef CAS PubMed; (b) R. Mandal, B. Emayavaramban and B. Sundararaju, Cp*Co(III)-Catalyzed C−H Alkylation with Maleimides Using WeaklyCoordinating Carbonyl Directing Groups, Org. Lett., 2018, 20, 2835–2838 CrossRef CAS PubMed.
- Y. Suzuki, B. Sun, T. Yoshino, M. Kanai and S. Matsunaga, Cp*Co(III)-catalyzed oxidative C-H alkenylation of benzamides with ethyl acrylate, Tetrahedron, 2015, 71, 4552–4556 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available: Synthesis and characterization data for compounds 1–10. Deuterium incorporation experiment on 1a. Computational data. Copies of 1H and 13C NMR (and 19F NMR) spectra of amides 3, 5, 7–10. CCDC 2463953. For ESI and crystallographic data in CIF or other electronic format see DOI: https://doi.org/10.1039/d5ob00781j |
| This journal is © The Royal Society of Chemistry 2025 |