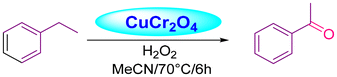Recent progress in the synthesis of CuCr2O4 nanomaterials and their composites for catalytic, energy, and biomedical applications: a state-of-the-art-review
Kajalben
Patel
a,
Mamta
Patil
a,
Yogita
Abhale
*a,
Kun-Yi Andrew
Lin
b,
Kar Ban
Tan
 c,
Ankush
Chauhan
d,
Abhinav
Kumar
c,
Ankush
Chauhan
d,
Abhinav
Kumar
 e,
Vishnu
Adole
e,
Vishnu
Adole
 f,
Majid S.
Jabir
f,
Majid S.
Jabir
 g and
Suresh
Ghotekar
g and
Suresh
Ghotekar
 *d
*d
aDepartment of Chemistry, Government College Daman (Veer Narmad South Gujarat University, Surat), Daman 396210, UT of DNH & DD, India. E-mail: mahayogi.85@gmail.com
bDepartment of Environmental Engineering & Innovation and Development Center of Sustainable Agriculture, National Chung Hsing University, Taichung, 40227, Taiwan
cDepartment of Chemistry, Faculty of Science, University Putra Malaysia, 43400 UPM, Serdang, Selangor, Malaysia
dCentre for Herbal Pharmacology and Environmental Sustainability, Chettinad Academy of Research and Education, Kelambakkam, Tamil Nadu 603103, India. E-mail: ghotekarsuresh7@gmail.com
eCentre for Research Impact & Outcome, Chitkara University Institute of Engineering and Technology, Chitkara University, Rajpura 140401, Punjab, India
fDepartment Chemistry, Mahatma Gandhi Vidyamandir's Loknete Vyankatrao Hiray Arts, Science and Commerce College (Savitribai Phule Pune University), Panchavati, Nashik, Maharashtra 422003, India
gCollege of Applied Sciences, University of Technology, Baghdad, Iraq
First published on 2nd October 2025
Abstract
Copper chromite nanoparticles (CuCr2O4 NPs) are in the spotlight of modern nanoscience due to their versatile applications across various scientific and industrial domains. This review addresses the synthesis of CuCr2O4 NPs, metal-doped CuCr2O4 NPs, and nanocomposites (NCs), emphasizing the key parameters, viz., reaction time, pH, temperature, precursor concentration, and the choice of fuel or surfactant, influencing their morphology, crystallinity, and functional properties. CuCr2O4 NPs are fabricated using several chemical methods, namely, sol–gel, co-precipitation, hydrothermal, solution combustion, and ball-milling, with different types of fuels or surfactants serving as complexing agents. These materials exhibit remarkable thermal, electrical, and catalytic characteristics. The discussion extends to their broad spectrum of applications, including biological activities such as antimicrobial, anticancer, and bone regeneration as well as their genotoxic impact. We also explore their role in photocatalysis, heterogeneous catalysis, and electrochemical applications, including supercapacitors and lithium-ion batteries. The emerging utility of CuCr2O4 NPs in sensor development is also highlighted. This review covers the recent advancements in the synthesis of CuCr2O4 NPs, highlighting their key challenges and future directions. Analyzing the structure–property relationships and the influence of synthesis parameters on their performance provides valuable insights into optimizing the design and functionalization of CuCr2O4 NPs for specific applications.
1. Introduction
In recent years, nanotechnology has emerged in the limelight through its diverse applicability in various fields, ushering in what can be called a “nano-era”. Richard Feynman's (1959) talk “There is plenty of room at the bottom” and the invention of the scanning tunneling microscope (1981) paved the way for the development of nanoscience and nanotechnology. Japanese scientist Norio Taniguchi first used the term “nanotechnology” to describe semiconductor processes at the nanoscale about fifteen years after Feynman's discussion. They promoted the idea that materials may be processed, separated, consolidated, and deformed by a single atom or molecule as part of nanotechnology.1In the present scenario, there has been considerable interest in advancing spinel oxide nanoparticles (NPs) across various disciplines of study. Their beneficial traits, including affordability, enhanced efficiency, effortless recuperation, and superior recyclability, suggest that they are potent catalysts.2–5 The remarkable catalytic activity of spinel oxides makes them noteworthy. Spinel oxides are inexpensive, harmless, and extremely stable materials with good acid and alkali resistance and high melting temperatures, and their NPs have a comparatively large surface area. Due to these qualities, they can be utilized as solid heterogeneous catalysts in organic chemical reactions.6–8 The most common type of spinel chromite is binary transition metal oxides, M2+B23+O42−, where M = Mg2+, Mn2+, Co2+, Ni2+, Cu2+, or Zn2+. Spinel nanostructures have garnered attention due to their diverse applications in various fields, including nanomedicine, photocatalysis, catalysis, energy storage, lithium-ion batteries, and sensors.2,9–11 CuCr2O4 is a fascinating chromate spinel due to its cost-effectiveness and non-toxicity. As a tetragonally deformed normal spinel, CuCr2O4 has an axial ratio (c/a) of less than 1, suggesting that its c-axis is compressed relative to its a-axis.7 Cr3+ ions occupy the octahedral (B) sites, while Cu2+ ions are positioned at the tetrahedral (M) sites, and the perfect crystal is anticipated to be an insulator. At normal and high temperatures, spinel CuCr2O4 crystallizes into a cubic phase and tetragonal phase, respectively. Its first-order phase transition occurs at approximately 854 K (upon heating).12,13 According to the Jahn-Teller phenomenon, spinel oxides containing copper have excellent thermal stability.14
Numerous approaches have been documented in the literature for preparing CuCr2O4 NPs, including hydrothermal,15 co-precipitation,16 sol–gel,17 solution combustion,8 microwave-assisted,18 and mechanical ball milling.19 Various fuels and surfactants, such as ethylene glycol, CTAB, tartaric acid, citric acid, glycine, and glucose, have been utilized as capping agents during the synthesis process. These agents are crucial in the synthesis of NPs, given that they enhance the monodispersity, reduce the particle size, and minimize the aggregation of nanoparticles. Surfactants reduce the surface tension of the solution, lowering the energy required to create a new phase from the precursor ions.20
CuCr2O4 has been identified as a p-type semiconductor (Eg = 1.40 eV) for a range of photocatalytic applications.15,21–27 Materials known as catalysts accelerate chemical reactions without being consumed by the reaction; however, they are frequently difficult to separate and recover.28–30 A green and cost-effective catalyst should have minimal preparation costs, high activity, selectivity, stability, ease of separation, and recyclability.31–33 Surprisingly, CuCr2O4 NPs function as a heterogeneous catalyst in several reactions, such as the thermal disintegration of ammonium perchlorate (AP),34 oxidation of aniline to azoxybenzene,35 preparation of pyrazine,36 synthesis of triarylamine,16 oxidation of benzene to phenol,15,24 hydrogenation of 2-furfuraldehyde,37 and synthesis of biscoumarin and pyrano[c]chromene,38 and this catalyst effectively catalyzes toluene oxidation by interfering with C–H bond activation39 and synthesis of [1]benzopyran azo dyes, etc.40
Intriguingly, CuCr2O4 nanomaterials/composites show a broad spectrum of biological activities, including anticancer,41 antibacterial,42,43 bone regeneration,44 hemolysis,41 and genotoxicity tests,45 with significant potential. Furthermore, CuCr2O4 nanomaterials and composites have been studied for energy storage applications. The development of high-energy and high-power batteries,46 as well as hybrid supercapacitor electrodes due to their capacitive characteristics, cyclic stability, and high power density,47 is crucial for meeting energy storage needs. Rechargeable metal–air batteries based on oxygen electrocatalysis are a viable alternative, particularly when the energy source is intermittent, such as solar or wind. These batteries use bifunctional oxygen electrocatalysis, which includes both oxygen reduction (ORR) and oxygen evolution processes (OER).48 Also, CuCr2O4 has potential as an electrocatalyst in electrochemical hydrogen storage systems, providing a sustainable solution to renewable energy technology.49
This review thoroughly examined CuCr2O4 NPs. The current literature was gathered from Google Scholar, ResearchGate, PubMed, Scopus, and books using the search terms “synthesis + CuCr2O4 NPs/doped metal/nanocomposites + energy storage + biological + catalytic/photocatalytic applications”. Fig. 1 shows a pie chart of the data received from these keywords. A significant amount of studies has been done on the synthesis and characterization of CuCr2O4 NPs that can be applied in a variety of scientific and engineering domains. The current summary will help researchers better understand the opportunities and challenges associated with the production of CuCr2O4 NPs.
 | ||
| Fig. 1 Data of the number of papers published on “synthesis of CuCr2O4 nanomaterials and their applications” from Google Scholar, ResearchGate, PubMed, Scopus, and Book database. | ||
2. Synthesis of CuCr2O4 NPs
The growth and production processes of CuCr2O4 NPs determine their structure and shape. To date, CuCr2O4 NPs have been synthesized using several processes, such as sol–gel, co-precipitation, solution combustion, precipitation, hydrothermal, microwave-assisted procedures, and mechanical ball milling, which are summarized in Table 1.| Synthesis method | Morphology | Particle size (nm) | Characterization techniques | Applications | Ref. |
|---|---|---|---|---|---|
| Sol–gel combustion | Laminate, octahedral, and spherical | — | XRD, UV-Vis, FTIR, DSC, TEM, SEM-EDX, ATR | Selective absorber coatings | 17 |
| Sol–gel | Spherical | 30–55 | XRD, FESEM, TEM, TGA-DSC | Catalytic activity | 50 |
| Semi spherical | — | XRD, UV-vis-DRS, FTIR, SEM | Photocatalytic activity | 51 | |
| Sol–gel | Spherical | 37 | FT-IR, XRD, BET, FESEM-EDS | Electrochemical hydrogen storage | 49 |
| Green synthesis | Spherical | 54 | |||
| Sol–gel | Quasi-spherical | 20–60 | XRD, UV–Vis, FTIR, TEM, SEM, RSR, MEM | Photocatalytic activity | 52 |
| Uniform | 850 °C < 100 | XRD, FTIR. SEM, VSM | Microwave absorber | 53 | |
| Poly tetragonal | — | XRD, FTIR, SEM | Antibacterial activity | 42 | |
| Spherical | 33 | UV-Vis-DRS, FESEM, XRD, FTIR | — | 55 | |
| Cubic | 20–60 | XRD, FTIR, SEM. TGA/DTA | Catalytic activity | 56 | |
| — | 34 | XRD, XPS, SEM, HRTEM-SAED | Sensor | 60 | |
| — | 20–60 | XRD, TEM, TG-DTA | — | 61 | |
| Octahedral | 20–30 | XRD, FESEM, EDAX, TG–DTG | — | 62 | |
| Pechini | — | 500 °C-30 | XRD, TG-DTA, TEM-SAED | Catalytic combustion | 57 |
| 700 °C-100 | |||||
| Pechini & modified Pechini | — | 650 °C-200 | XRD, SEM | — | 58 |
| Sol–gel self-combustion | Tetragonal | 82.56 | XRD, FESEM, EDS, HRTEM-SAED, XPS, CIS | Electrical conductivity | 59 |
| Solution combustion self-combustion | Spherical | 200 and 225 | XRD, TGA-DTA, SEM, BET, XPS | Catalytic activity | 8 |
| Combustion | Irregular | 25.16 | UV-VIS-NIR, XRD, BET, SEM | — | 63 |
| Spherical | CuCr2O4-35–40 | XRD, FTIR, BET, HRSEM, HRTEM, GC-MS | Polypropylene pyrolysis oil into diesel | 66 | |
| NiCuCr2O4-38–46 | |||||
| Spherical | 135.2 | XRD, FTIR, TEM, SEM, EDAX, TGA-DTA | Catalytic activity | 65 | |
| Self-combustion | Spherical | — | XRD, UV-vis-DRS, FTIR, and SEM-EDX | Photocatalytic activity | 64 |
| Flow microreactor | — | 192–300 | XRD, FTIR, FESEM, 1H, 13C NMR | — | 87 |
| Co-precipitation | Spherical | 20–30 | XRD, IR, SEM | Catalytic activity | 16 |
| Spherical | 13.1 | SEM, TEM, DLLS | Catalytic activity | 69 | |
| Spherical | Uniform | XRD, FTIR, SEM, 1H, 13C NMR | Catalytic activity | 67 | |
| Octahedral | — | XRD, SEM, TGA-DTA | Photocatalytic activity | 71 | |
| Spherical | 22–28 | XRD, FTIR, SEM, 1H, 13C NMR | Catalytic activity | 68 | |
| Spherical and cubic | 26–30 | XRD, TEM, Raman spectroscopy, X-ray microanalyses | Catalytic activity | 92 | |
| Irregular | 20–30 | XRD, SEM, 1H and 13C NMR | Catalytic activity | 38 | |
| Quasi-spherical | 600 °C-15.1 | XRD, TEM, BET | Photocatalytic activity | 23 | |
| 700 °C-21.7 | |||||
| Spherical | 48 | XRD, SEM, UV-vis, TG–DSC | Dye sensitized solar cells | 72 | |
| Inverse-coprecipitation | Spherical | 520 °C-72 | XRD, FTIR, FESEM, TG–DSC | — | 70 |
| Hydrothermal | Almost uniform | 20–45 | XRD, XPS, ICP-AES, SEM, TEM, TGA | Catalytic activity | 15 |
| Almost uniform (25–50 nm) | CuCr2O4 fresh-35, CuCr2O4 spent after five reuses-39 | XRD, ICP-AES, XPS, EXAFS, SEM, TEM, TGA | Catalytic activity | 74 | |
| Uniform | 20–60 | XRD, ICP-AES, XPS, FTIR, SEM, TEM, TGA | Catalytic activity | 75 | |
| Almost uniform (25–50 nm) | CuCr2O4 fresh-35, CuCr2O4 spent after five reuses-36 | XRD, XPS, SEM, TEM, TGA, ICP-AES | Catalytic activity | 35 | |
| Spherical | 20–40 | XRD, XPS, ICP-AES, SEM, TEM, TGA | Catalytic activity | 15 | |
| Irregular-500 °C | — | XRD, UV–vis-DRS, TEM, TG–DSC, XPS, zeta potential | Photocatalytic activity | 22 | |
| Cubic-600–800 °C | |||||
| Uniform | 20 | XRD, FTIR, SEM, TEM | Supercapacitor | 47 | |
| Spherical | 12 | XRD, SEM, TEM | Catalytic activity | 79 | |
| Almost uniform | 30–60 | XRD, FTIR, SEM-EDAX, TEM, XPS | Catalytic activity | 76 | |
| Sphere | 23 | XRD, FTIR, SEM, EDS, 1H, 13C NMR | Catalytic activity | 40 | |
| — | 7.9–23.9 | XRD, FTIR, SEM, TEM, TPR, TGA, BET | Catalytic activity | 77 | |
| Uniform | 20–40 | XRD, ICP-AES, XPS, TPR, BET, SEM, TEM, EXAFS | Catalytic activity | 39 | |
| Uniform | CuCr2O4-35 | XRD, FTIR, UV-Vis, FESEM, TGA | Photocatalytic activity | 80 | |
| Mn0.5Cu0.5Cr2O4-90 | |||||
| Uniform | 20–50 | XRD, ICP-AES, FTIR, SEM, TEM, TGA, XPS | Catalytic activity | 78 | |
| — | 45 | XRD, TEM, SEM, EDAX | Catalytic activity | 93 | |
| 800 °C-spherical | 100–300 | XRD, SEM-EDX | Solar absorber coatings | 14 | |
| Irregular and spherical | CuCr2O4-30.17 | XRD, FT-IR, UV–Vis, FE-SEM, HR-TEM, BET | Photocatalytic activity | 81 | |
| CuCr2O4–citric acid-26.38, CuCr2O4–tartaric acid-13.13 nm | |||||
| Mechanical ball milling | — | 30 | XRD, FTIR, SEM, TEM, BET, TGA/DSC | Catalytic activity | 84 |
| Ultrasonic dispersion method and a mechanical grinding | Spherical | — | XRD, FTIR, SEM, EDS, DSC–TG–DTG | Catalytic activity | 85 |
| Microwave assisted co-precipitation | Flower | 21–46 | XRD, UV-Vis, IR, FESEM, HRTEM | — | 18 |
| Microwave | Spherical | Citric acid-23, oleic acid-8.43 | XRD, FTIR, SEM-EDS | — | 82 |
| Coconut oil-9.62 | |||||
| Urea-15.48 | |||||
| Hexagonal | 41–25 | UV-vis, XRD, FTIE, SEM, TEM | Dye-sensitized solar cell | 83 | |
| Microwave combustion | Irregular plate | 37–26 | XRD, SEM | Antibacterial and electrical conductivity | 43 |
| Sputtering | — | 500 °C-30–40 | XRD, SEM, AFM, TEM | — | 89 |
| 800 °C-100 | |||||
| Wet chemical grinding | Semi-spherical | 40 | XRD, SEM, TEM, DSC/TG | Catalytic activity | 90 |
| Co-precipitation, ultrasound, and green chemistry | — | 41.51 | XRD, FT-IR, EDX, SEM, zeta potential | Anti-breast cancer activity | 41 |
2.1. Pristine CuCr2O4 NPs
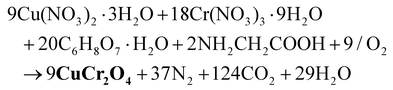 | (1) |
Hosseini et al. produced CuCr2O4 NPs using a temperature-programmed procedure after a sol–gel method in the presence of urea, and the resulting particles were homogeneous, well-separated, and had a sphere-like morphology.50 In another study, the researchers used sucrose as a template in the synthesis process and compared the template-assisted and template-free approaches.51 Lachini et al. manufactured CuCr2O4 NPs using two different methods, sol–gel synthesis with stearic acid and green synthesis using Aloe vera. Researchers successfully investigated the performance of these materials as electrocatalysts in electrochemical hydrogen storage.49 Benrighi et al. found that CuCr2O4 NPs have a spinel structure with an orthorhombic phase, specifically belonging to the Fddd space group.52
Peymanfar et al. investigated the microwave absorption properties of NPs produced using silicone rubber as a polymeric matrix. In the first study, they investigated a CuCr2O4/silicone rubber nanocomposite, which has a microwave absorption of 48.56 decibels (dB) at 10.9 GHz, a thickness of 2.6 mm, and more than 92.99% microwave absorption along the X-band frequency.53 In the second investigation, they created CuCr2O4/poly(vinylidene fluoride) (PVDF) nanocomposites. They achieved a maximal reflection loss of −65.57 dB at 14 GHz with a thickness of 2 mm, and absorbed a bandwidth of 7.73 GHz with more than 10 dB absorption. CuCr2O4 NPs play a promising function in improving microwave absorption properties in polymeric matrices.54 As discussed in a previous report, Akbar Mirzai and team synthesized CuCr2O4 NPs using glucose and created antibacterial films using PVDF and silicone rubber.42
Habibi et al. employed the citrate sol–gel synthesis method to synthesize CuCr2O4 NPs at room temperature using a fuel. The XRD data from the samples indicated the development of the CuCr2O4 nanocomposite at a calcination temperature of 700 °C.55 Wang et al. synthesized nanochromates MCr2O4 (M = Co, Ni, Cu, and Zn) using the sol–gel technique. The structural and morphological traits were disclosed employing XRD and SEM.56 The Pechini method involves dissolving Cu and Cr salts, and then adding citric acid to produce a homogeneous solution. This mixture was heated at 95 °C for many hours (h) to evaporate the water, yielding dark brown, clear, viscous gels. After that, the gel was dried at 160 °C for 2 h, resulting in frothy dark powders known as the precursor to CuCr2O4/CuO nanocomposites.57 Additionally, Billman et al. manufactured CuCr2O4 NPs utilizing two nitrate–citrate combustion synthesis sol–gel methods, as indicated in Fig. 2A, i.e., one with ethylene glycol and neutralized with ammonium hydroxide (Pechini technique (Pechini, 1967)) and one without (modified Pechini method). The Pechini technique yields a broader particle size distribution, characterized by generally smaller and more scattered agglomerates, as shown in Fig. 2B, which exhibits a more branching structure with larger cluster sizes.58
 | ||
| Fig. 2 (A) Diagram of Pechini and modified Pechini method and (B) graphic showing the differences between the two methods for producing gel structures from chelated complexes.58 | ||
Javed et al. conducted a detailed analysis of the physical characteristics of CuCr2O4 NPs. For the study of their structural attributes, a schematic representation was generated using the VESTA program (Fig. 3). CuCr2O4 NPs have a single-phase, tetragonally distorted normal spinel structure with an I41/amd space group in the crystal lattice. In the CuO4 tetrahedral geometry, Cu(II) bonds have shorter lengths and angles compared to Cr(III) bonds in the CrO6 octahedron. The TEM images revealed d-spacing values of 0.27310 nm. XPS analysis revealed the presence of several electrical routes in the CuCr2O4 spinel system, including Cu2+–O2−–Cr2+, Cr3+–O2−–Cr3+, Cr6+–O2−–Cr6+, and Cr3+–O2−–Cr6+.59
 | ||
| Fig. 3 Schematic of the tetragonally distorted CuCr2O4: (a) unit cell, (b) extended crystal structure, and (c) 2D and (d) 3D electron density maps.59 | ||
Kong et al. reported that the CuCr2O4 material has a pore structure that allows rapid analyte transport and generates well-dispersed, small-sized NPs with a minimal polyhedral content.60 Cui et al. created various types of spinel NPs, including CoAl2O4 (blue), CoCr2O4 (bluish green), ZnFe2O4 (brown), and CuCr2O4 (black), employing propylene oxide as a gelation agent via the sol–gel technique, which reduces the required calcination temperature and minimizes particle agglomeration.61 Habibi et al. investigated the decomposition mechanism of Cu(NO3)2·3H2O and Cr(NO3)3·9H2O precursors, as well as the copper chromite xerogel precursor, using thermal analysis. The results revealed that mass loss for copper(II) nitrate and chromium(III) nitrate occurred at 258 °C and 140 °C, respectively. In comparison, the major mass loss for the dried copper chromite gel occurred at 310 °C, confirming the decomposition of nitrates and the phase transformation leading to the complete crystallization of CuCr2O4.62
Pan et al. described the synthesis process of CuCr2O4 NPs. They used the appropriate mixed amount of Cu(NO3)2·3H2O and CrCl3·6H2O, along with (CH2)6N4 and NH4HCO3, and then agitated for 1 h. After aging the mixture for 12 h, the samples were dried in a vacuum at 105 °C for 6 h, and the dry precursor was obtained. After that, the precursor was calcined at 400 °C, 450 °C, 500 °C, and 600 °C for 3 h each.71 A mono-dispersed CuCr2O4 nanopowder was effectively generated using an alcohol–aqueous mixture and applied as photoanodes in dye-sensitized solar cells.72
Paul et al. manufactured CuCr2O4 NPs utilizing a co-precipitation method with a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 molar ratio of Cu and Cr hydroxide precursors. Initially, urea decomposed to NH3 and CO2 (eqn (2)). NH3 reacts with water molecules to form NH4+ and OH− (eqn (3)). The free OH− ions subsequently mix with Cu2+ and Cr3+ ions to form Cu(OH)2 and Cr(OH)3 (eqn (4) and (5)), respectively. The sample was calcined to produce CuCr2O4 NPs (eqn (6)).23
2 molar ratio of Cu and Cr hydroxide precursors. Initially, urea decomposed to NH3 and CO2 (eqn (2)). NH3 reacts with water molecules to form NH4+ and OH− (eqn (3)). The free OH− ions subsequently mix with Cu2+ and Cr3+ ions to form Cu(OH)2 and Cr(OH)3 (eqn (4) and (5)), respectively. The sample was calcined to produce CuCr2O4 NPs (eqn (6)).23
| (NH2)2CO + H2O → 2NH3 + CO2 | (2) |
| NH3 + H2O → NH4+ + OH− | (3) |
| Cu2+ + 2OH− → Cu(OH)2 | (4) |
| Cr3+ + 3OH− → Cr(OH)3 | (5) |
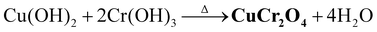 | (6) |
Ismael et al. produced CuCr2O4 NPs via a hydrothermal method. Fig. 4 shows how colloidal CuCr2O4 was re-dispersed in acetone after being recovered from its production medium using an ultrasonic probe. Ammonium perchlorate (AP) particles, followed by dissolution in acetone and colloidal CuCr2O4 NPs, were integrated into the AP matrix using the antisolvent method.79
 | ||
| Fig. 4 Integration of CuCr2O4 NPs into AP.79 | ||
Soleimani et al. prepared CuCr2O4 and Mn0.5Cu0.5Cr2O4 and compared their effectiveness in degrading malachite green with CuCr2O4 NPs.80 Pleasingly, Acharyya et al. reported the 3D raspberry-like morphology of CuCr2O4 in one of their studies. Crystal-face attraction, electrostatic fields, van der Waals forces, hydrophobic interactions, hydrogen bonds, and the autogenous pressure in the autoclave influence the creation of the raspberry morphology. The “soft template” CTAB and Ostwald ripening also have a substantial contribution. Cr6+ precursors, such as K2Cr2O7, play a significant role in forming the raspberry-like structure, but Cr3+ precursors, including CrCl3 and Cr(NO3)3·9H2O, do not. Synthesizing chromium(III) oxide with CTAB, (NH4)2Cr2O7, and hydrazine resulted in a characteristic 3D strawberry-like texture, underlining the significance of Cr6+ in self-assembly (Fig. 5).78
 | ||
| Fig. 5 Schematic of the production of CuCr2O4 spinel catalyst with a 3D raspberry-like structure.78 | ||
Rubin et al. hydrothermally synthesized CuFeMnO4 and CuCr2O4 NPs, whereas large-batch sintering produced Cu0.5Cr1.1Mn1.4O4. These samples were annealed at 800 °C for various durations, including 100, 300, 1000, and 2000 h. After 2000 h, XRD data verified that the relevant peaks are indeed that of CuCr2O4, and there is no reason to assume a phase transition from CuCr2O4 to CuCrO2 (delafossite).14 Al-Wasidi et al. demonstrated the facile synthesis of CuCr2O4 NPs using citric acid and tartaric acid templates, as well as in their absence.81
Appalakutti et al. fabricated Cu–Cr–O nanocomposites using a continuous flow microreactor. As depicted in Fig. 6, Cu, Cr nitrate, and surfactant (PVP and CTAB) mixtures were filled in a syringe at varying molar ratios. Alternatively, another syringe was filled with a citric acid solution in a 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
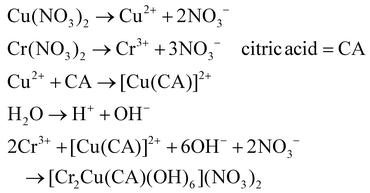
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 molar ratio relative to the total metal ion concentration. The microreactor was placed in a water bath at 80 °C, with a flow rate ranging from 25 to 150 mL h−1. After that, the nanopowder was generated by calcining at 200 °C for a specific period of 2 h. The hydrolysis and polycondensation reaction (eqn (7)–(10)) mechanisms are provided below.87
1 molar ratio relative to the total metal ion concentration. The microreactor was placed in a water bath at 80 °C, with a flow rate ranging from 25 to 150 mL h−1. After that, the nanopowder was generated by calcining at 200 °C for a specific period of 2 h. The hydrolysis and polycondensation reaction (eqn (7)–(10)) mechanisms are provided below.87
 | ||
| Fig. 6 Illustration of the fabrication of CuCr2O4 NPs in a continuous flow microchannel reactor.87 | ||
Hydrolysis reaction
| (7) |
(Precursor A)
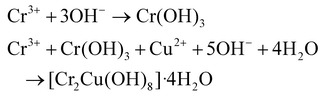 | (8) |
(Precursor B)
| 14H+ + 14NO3− → 14HNO3 |
Polycondensation by dehydration
(Precursor A)
 | (9) |
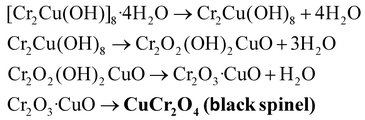 | (10) |
Interestingly, Chiu and research team used an electrospinning approach to produce 1-D CuCr2O4 fibers.88 Tsai and colleagues successfully examined the production of transparent conducting Cu–Cr–O films on fused silica substrates utilizing DC reactive magnetron sputtering with a single equimolar Cu–Cr alloy target and a post-annealing method in a controlled Ar atmosphere at 500–800 °C for 2 h.89
The wet mechanical grinding method and the vacuum freeze-drying process present a promising technique for easily producing Cu-based NPs, which have potential applications in the mass preparation of other nanosized catalysts. The Cu-based NPs, including CuO, CuCr2O4, and copper β-resorcylate, are illustrated in Fig. 7a and b, which detail the synthesis and drying processes, respectively.90
 | ||
| Fig. 7 Diagram of the production of Cu-based NPs: (a) vacuum freeze-drying and (b) wet mechanical grinding.90 | ||
Ahmed et al. produced CuCr2O4 using rosemary (Rosmarinus officinalis) leaf extract, employing three techniques including co-precipitation, ultrasonication, and green chemistry. Researchers evaluated its anti-breast cancer efficacy using the MCF-7 cell line, which revealed encouraging results.41 The CuCrO2 phase develops at a temperature of 750–800 °C for all five mixes. CuO, Cr2O3, and CuCr2O4 phases exist in this temperature range; hence, the CuCrO2 phase is produced by reacting the spinel phase with CuO.91
In conclusion, various physical, chemical, and green synthesis methods have been employed to fabricate CuCr2O4 nanomaterials. Among them, the sol–gel method is the most widely used due to its low calcination temperature, excellent chemical homogeneity, and ability to produce monodispersed NPs with a controlled size and morphology. The citric acid-assisted sol–gel technique is particularly effective, given that citric acid functions as a chelating agent, forming homogeneous metal–citrate gels that promote the uniform mixing of metal ions and result in smaller crystallite sizes. According to the literature, this method typically yields nanoparticles with small sizes and spherical morphology, which enhance their surface area and improves their catalytic performance. The improved homogeneity of CuCr2O4 synthesized via this approach significantly boosts its photocatalytic activity, especially in the degradation of antibiotics, making it more advantageous than conventional synthesis techniques. Additionally, the sol–gel approach facilitates the incorporation of capping agents, such as amino acids, and green alternatives, such as sucrose. These stabilizing agents effectively minimize the agglomeration, enhance the crystallinity, and refine the morphology of the particles. Therefore, the sol–gel method, when combined with suitable chelating and capping agents, provides a versatile, efficient, and environmentally friendly approach for synthesizing high-performance CuCr2O4 nanostructures for various applications.
2.2. Synthesis of metal-doped CuCr2O4 NPs
Batool et al. manufactured Cd- and Nd-doped CuCr2O4 NPs (MxCu1−xCr2O4) with x values of 0.2, 0.4, 0.6, and 0.8. The band gap of the parent compound was ascertained employing photoluminescence (PL) spectra and found to be 4.96 eV. When elements were doped in the parent compound at varying concentrations, the band gap (Eg) decreased for Nd (2.43 eV) and Cd (2.42 eV). Additionally, microscopic images revealed that the texture of pure CuCr2O4 NPs was tetragonal and that doping with other compounds alters the structure according to the characteristics of the NPs.94 Anusree et al. produced Ba and Co-doped CuCr2O4 NPs using a hydrothermal technique and evaluated the catalytic efficacy of the created NPs for the heat breakdown of AP.95 These studies, conducted by Acharyya et al., reported the formation of nanoclusters of Cu(II) supported by CuCr2O4 NPs. They investigated their catalytic activity in the selective hydroxylation of benzene to phenol,24 the oxyamination of benzene to aniline,96 and the selective oxidation of ethanol.97Bai et al. reported the synthesis of Fe and Mn-doped CuCr2O4 NPs with the formula CuCr2−x−yFexMnyO4. The doped pigments exhibited a higher UV-visible absorbance than CuCr2O4, attributed to electron transfer between O2− → Fe3+, O2− → Mn3+, and d–d intra-atomic transitions of Fe3+ and Mn3+. The CuCr1.93Fe0.07O4 black pigment showed a higher NIR solar reflectance (R* = 27.7%) than the CuCr2O4 black pigment (R* = 19.0%), which was ascribed to the decreased concentration of free carriers.98
Yadagiri et al. employed the sol–gel method for generating CuCr2O4 and Fe-substituted CuCr2O4. XRD analysis of CuCr2−xFexO4 revealed that for x = 0.0, the phase is tetragonal, whereas the other three compounds (x = 0.5, 1.0, and 1.5) have a cubic phase. It was discovered that the average crystalline size increased with Fe substitution.99 Ramezanalizadeh et al. created La-doped CuCr2O4 NPs. The photocatalytic study of doped NPs revealed the outstanding degradation of contaminated dyes.100 Jagadeesan et al. synthesized pure CuCr2O4 and doped CuCr2−xMgxO4 (x = 0.00–0.1%, 0.2%, and 0.3%) nanocomposites. In short, the Mg-doped sample has a higher specific surface area than the pure CuCr2O4 sample.101 Interestingly, Zn-doped CuCr2O4 was synthesized at different concentrations and calcined at various temperatures.102 Interestingly, scientists utilized CuCr2O4 black oxide pigment doped with various metals, including Mn, Ni, Co, Al, Zn, and Sn, to create solar-absorbing spinel structures. Among them, the Mn–CuCr2O4 pigment exhibited much higher levels of solar absorption than the other metal dopants.103
Using a microscopic model and Green's function theory, Apostolov et al. theoretically investigated the magnetic, electric, and optical characteristics of both pure and ion-doped CuCr2O4 in bulk and nanoparticle forms. Their results showed that the magnetization and band gap energy (Eg) decrease with an increase in the nanoparticle size. Magnetization and polarization (P) peaked when doped with Co ions, while both qualities decreased as the Pr ion concentration increased. Therefore, compared to that substituted with rare-earth elements, Co-doped CuCr2O4 NPs are more appropriate for spintronic applications. Alternatively, Pr ion doping raises the Eg, which makes it more suitable for solar cell technology.104
Table 2 summarizes the diverse synthetic approaches, characterization techniques, and applications of metal-doped CuCr2O4 NPs.
| Doped nanoparticles | Synthesis method | Morphology | Particle size (nm) | Characterization techniques | Applications | Ref. |
|---|---|---|---|---|---|---|
| CuCr2O4 | Sol–gel | Spherical | >100 | XRD, EDS, SEM, PL, Raman spectroscopy | — | 94 |
| Cu1−xCdxCr2O4 and Cu1−xNdxCr2O4 | — | 25–55 | ||||
| Ba and Co doped CuCr2O4 | Hydrothermal | Irregular | CuCr2O4-42 | XRD, SEM, TEM, BET | Catalytic activity | 95 |
| CoCuCr2O4-52 | ||||||
| BaCuCr2O4-32 | ||||||
| Cu(II) oxide | Hydrothermal | — | 2–8 | XRD, SEM, TEM, XPS, FTIR, TGA, TPR, BET, XANES, ICP-AES | Catalytic activity | 24 |
| CuCr2O4 NPs | ∼55 | |||||
| Cu(II) supported CuCr2O4 | — | — | ∼10 | XRD, XPS, SEM, TEM, TPR, TGA | Catalytic activity | 96 |
| 30–60 | ||||||
| Cu(II)-supported CuCr2O4 | Hydrothermal | — | 30–60 | XRD, BET, ICP-AES, SEM, TEM, TGA | Catalytic activity | 97 |
| Fe/Mn co-doped CuCr2O4 | Co-precipitation | Octahedron | — | XRD, UV-vis-NIR, XPS, SEM, Raman spectroscopy | — | 98 |
| CuCr2O4 and Fe-substituted CuCr2O4 | Sol–gel | Rectangular | — | XRD, UV-vis, Mossbauer, Raman spectroscopy, FESEM | — | 99 |
| La–CuCr2O4 | Sol–gel Pechini | Spherical | — | XRD, SEM, FTIR, DRS, VSM | Photocatalytic activity | 100 |
| CuCr2O4 | Combustion | Irregular | 49–53 | XRD, FTIR, FESEM, TEM, BET | Catalytic activity | 101 |
| Mg (0.1%) CuCr2O4 | 43–39 | |||||
| Mg (0.2%) CuCr2O4 | 40–31 | |||||
| Mg (0.3%) CuCr2O4 | 39–29 | |||||
| CuCr2O4 | Hydrothermal and co-precipitation | Octahedral | 71.84 | XRD, UV–vis–NIR, SEM | Solar power | 103 |
| CuCrMnO4 | Octahedral | 38.07 | ||||
| CuCrNiO4 | Flake | 51.88 | ||||
| CuCrCoO4 | Flake | 33.71 | ||||
| CuCrAlO4 | Irregular rods and polyhedral | 31.64 | ||||
| CuCrZnO4 | Flake | 38.41 | ||||
| CuCrSnO4 | Octahedral | 86.36 | ||||
| ZnCuCr2O4 | Sol–gel | — | — | XRD, SEM-EDS UV, PL, Raman spectroscopy | — | 102 |
| Cu1−xMgxCr2O4 | Sol–gel | Quasi spherical | — | XRD, UV, SEM | — | 105 |
2.3. Synthesis of CuCr2O4 nanocomposites
The interaction of NPs with the matrix and particle–particle interactions are important variables in determining the properties of nanocomposites. Strong interactions between particles and epoxy can enhance the mechanical properties of nanocomposites.106–108 Lao et al. created CuCr2O4 NPs using the sol–gel technique. Then, a suspension was created by diluting the CuCr2O4 sample with glucose. Ag(NH3)2OH was created by titrating a 0.01 mol L−1 AgNO3 solution from a diluted NH3·H2O solution. They combined the suspension with the Ag solution. Then, the mixture was centrifuged and dried, yielding a final weight ratio of CuCr2O4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Ag of 9
Ag of 9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1. The SEM investigation revealed that the surfaces of the NPs were shiny and smooth. Following electroless plating with Ag, nanosized particles were found to be equally dispersed on the surface of CuCr2O4, indicating that Ag was efficiently coated onto the CuCr2O4 surface.46 Kien and colleagues used the sol–gel and impregnation methods to produce CuCr2O4·Pd/γ-Al2O3.109 As previously described in their studies, they first synthesised Bi2O3.110 Subsequently, the complexing-coprecipitation method was employed to synthesise CuCr2O4–CuO. As depicted in Fig. 8, electrostatic interaction between NH4+ and Bi2O3 NPs resulted in the coating of NH4+ on Bi2O3 surfaces because of their higher specific surface area and plentiful OH−. Cu2+ and Cr3+ salts were then deposited on the Bi2O3 surfaces. NH4+ on Bi2O3 particles exhibits a strong complexing interaction with Cu2+, and it may be absorbed on the surface of Bi2O3, forming [Cu(NH3)4]2+. Similarly, Cr3+ can be coated on the surface of Bi2O3 particles through electrostatic interaction between [Cu(NH3)4]2+ and [Cr(OH)4]−. After calcination, monodisperse spherical Bi2O3/CuCr2O4–CuO core/shell nanocomposites were produced.111
1. The SEM investigation revealed that the surfaces of the NPs were shiny and smooth. Following electroless plating with Ag, nanosized particles were found to be equally dispersed on the surface of CuCr2O4, indicating that Ag was efficiently coated onto the CuCr2O4 surface.46 Kien and colleagues used the sol–gel and impregnation methods to produce CuCr2O4·Pd/γ-Al2O3.109 As previously described in their studies, they first synthesised Bi2O3.110 Subsequently, the complexing-coprecipitation method was employed to synthesise CuCr2O4–CuO. As depicted in Fig. 8, electrostatic interaction between NH4+ and Bi2O3 NPs resulted in the coating of NH4+ on Bi2O3 surfaces because of their higher specific surface area and plentiful OH−. Cu2+ and Cr3+ salts were then deposited on the Bi2O3 surfaces. NH4+ on Bi2O3 particles exhibits a strong complexing interaction with Cu2+, and it may be absorbed on the surface of Bi2O3, forming [Cu(NH3)4]2+. Similarly, Cr3+ can be coated on the surface of Bi2O3 particles through electrostatic interaction between [Cu(NH3)4]2+ and [Cr(OH)4]−. After calcination, monodisperse spherical Bi2O3/CuCr2O4–CuO core/shell nanocomposites were produced.111
 | ||
| Fig. 8 Schematic of the spherical Bi2O3/CuCr2O4–CuO core/shell NCs.111 | ||
Balasurya et al. synthesized NCs using the co-precipitation method to synthesize CuCr2O4 NPs. Using these NPs and Bi salt, they synthesized CuCr2O4–BiI3O9 nanocomposites via a sonochemical method.45 Ghorai and co-workers described CuCr2O4/BiOBr NC. Sol–gel combustion produces spherical CuCr2O4 NPs, which are then decorated on BiOBr plates using precipitation. CTAB was used as the Br source and template (Fig. 9).112
 | ||
| Fig. 9 Fabrication procedure of CuCr2O4/BiOBr nanocomposite.112 | ||
A CuCr2O4@CaFe–LDO hybrid was synthesized by Oladipo and group. They investigated its photocatalytic potential for treating olive mill effluent (OMW). These nanocomposites demonstrated outstanding absorption and photocatalytic activity.113,114 The researchers synthesized various nanocomposites, including CuCr2O4–CeO2, using the sol–gel and co-precipitation methods, as well as a combination of these techniques.115 Additionally, they employed the impregnation method,116 reflux condensation,26 and ultrasonication-assisted methods117 to synthesize Cu/CuCr2O4, CuO–CuCr2O4, and mesoporous CuCr2O4–g-C3N4 heterojunctions, respectively.
Hummers’ approach was adapted to produce graphene oxide.118 Graphene nanocomposites with electron-donating inclusions increase the electronic significance of the molecules they support.119–121 g-C3N4–NS/CuCr2O4 using refluxing,119 CuCr2O4/GO using co-precipitation,120 MXene (Ti3C2Tx)/CuCr2O4 used wet-chemical sonication-assisted co-precipitation approach,122 nanospherical–CuCr2O4 nanocomposite decorated with polyaniline (PANI),123 heterogeneous p-CuCr2O4/n-SnO2,124 and MCr2O4/TiO2 (M–Cu, Ni) using the co-precipitation method.125
Gao et al. employed a high-temperature phase separation technique to synthesize CuCr2O4/Cr2O3 nanogranular from Cu-doped Cr2O3 NPs, as confirmed by XRD tests. Its magnetic susceptibility has two transitions, i.e., CuCr2O4 becomes ferrimagnetic at 130 K, and Cr2O3 becomes antiferromagnetic at 310 K.126
Mechanical milling is a viable solid-state method for creating new CuCrO2/CuCr2O4 nanocomposites. This approach affects both the outcome of the reaction product and the phase transitions that occur during the process. This research compares the heat treatment and characteristics of the nanocomposite to that of single-phase delafossite.127
As depicted in the reaction pathway in Fig. 10, firstly CuCr2O4 NPs were fabricated via a sol–gel auto-combustion approach. Then, calcined NPs were dispersed in epoxy resin and polyurethane (PU) using ultrasonication to synthesize EP/PU/CuCr2O4 nanocomposites. By adding CuCr2O4 NPs to the EP/PU matrix, the tensile test showed that the mechanical properties were greatly improved. The ideal composition was 0.76 wt% CuCr2O4 NPs and 2.6 wt% polyurethane in the epoxy resin. Additionally, molecular dynamics simulations showed how the NPs affected the mechanical properties and interaction energy of the nanocomposite.128
 | ||
| Fig. 10 Schematic layout of the synthesis of EP/PU/CuCr2O4 nanocomposites.128 | ||
The synthesis of CuCr2O4 NPs via the co-precipitation technique, using 6 M KOH as the precipitating agent, is illustrated in Fig. 11a. The TGA examination of the as-dried sample, as shown in Fig. 11b, indicates a weight loss of 25% between 28 °C and 260 °C because of the removal of C2H5OH and H2O and a slight weight loss of 5.5% between 400 °C and 460 °C because of the elimination of HCl. According to the study, annealing should occur at 460 °C, while slower heating rates could lower this temperature. Using XRD analysis, Fig. 11c verifies the generation of CuCr2O4 NPs, and Fig. 11d displays the optical absorbance band. The band gap of CuCr2O4 NPs is 3.35 eV, according to Fig. 11e.129
 | ||
| Fig. 11 (a) Diagram of the synthesis of CuCr2O4 (with an image of the final product), (b) TGA, (c) XRD spectra of the NPs, with an inset showing thw crystal structure, (d) UV-visible spectra, and (e) optical band gap energy of CuCr2O4 NPs.129 | ||
Susanti et al. produced N-doped reduced GO via hydrothermal temperature changes. XRD investigation revealed that the d-spacing value increased with temperature, indicating the presence of more substituted N atoms. The EDX examination revealed that the sample treated at a hydrothermal temperature of 160 °C had the highest N concentration (14.88%).130 Interestingly, CuCr2O4 particles were supported on macroscopic polystyrene beads using a low-temperature ultrasonic technique.8 According to the SEM pictures, the CuCr2O4 particles completely coated the PS surface.131 TEM examination revealed that CuCr2O4 was uniformly distributed on rGO.132
Intriguingly, Shi et al. synthesized CuCr2O4/SiO2 composite membranes by impregnating a quartz glass fibrous (QGF) membrane with Cu and Cr precursors, followed by drying at 110 °C and calcining at 700 °C. Flat and frisbee-shaped membranes with varying CuCr2O4 loadings were produced with the frisbee-shaped samples assembled with round EPS disks to enable floating and provide heat barrier functionality for water distillation performance testing (Fig. 12).133
 | ||
| Fig. 12 Frisbee-shaped CuCr2O4/SiO2 composite membrane disk synthesis and EPS assembly to generate a composite CuCr2O4/SiO2-EPS tandem-structured disk.133 | ||
Holec et al. used the sol–gel method to produce ZnCr2O4/SiO2, CuCr2O4/SiO2, and CdCr2O4/SiO2, with HNO3 as an acid catalyst, formamide (modifier), methanol (solvent), and TEOS (precursor). Furthermore, they studied the magnetic behavior of the synthesized NPs; among them, the magnetic behavior of CuCr2O4 exhibits a ZFC-FC bifurcation at 150 K, hysteresis necking at 2 K, indicating two magnetic phases, and a wide anomaly at 30 K attributed to a surface spin superparamagnetic-like phenomenon.134 Ghorai et al. and their research team constructed CuCr2O4 and TiO2 by chemically depositing CuCr2O4 on a Ti precursor via sonochemical treatment, followed by thermal treatment.48 Sivaranjani et al. observed that adding CuCr2O4 increased the particle size and decreased the full-width at half-maximum of the TiO2 patterns, indicating improved crystallinity. According to the XRD analysis, CuCr2O4 improves the crystal structure of TiO2 NPs.118
Khan et al. found that coupling the V2O5/CuCr2O4–MCM-41 nanocomposite interface considerably improves the photocatalytic decomposition of alizarine red (AR) and bromothymol blue (BTB), as well as photoinhibition of Staphylococcus aureus and Escherichia coli bacteria.135 Hosseini et al. employed CTAB and the sol–gel method to conduct a qualitative and quantitative study of CuCr2O4 NCs with different Cu and Cr molar ratios. Titration procedures were employed to determine the mass percentages of Cr2O3 and CuO. For each sample, the total percentage of Cr2O3 and CuO was approximately 100%, indicating the absence of impurities and confirming that the samples contained only Cr2O3 and CuO.136 Vinothkumar et al. reported the synthesis of CuCr2O4/Cr2O3 NCs involving dissolving Cu(II) and Cr(III) precursors in dist. H2O with stirring to obtain a homogeneous solution. Liquid ammonia is then added as a precipitating agent. The resulting precipitate is dried and calcined to form CuCr2O4/Cr2O3 NCs. The researchers successfully studied the supercapacitor performance of CuCr2O4/Cr2O3 composites on nickel foam, as demonstrated in Fig. 13.137
 | ||
| Fig. 13 Schematic layout of the synthesis of CuCr2O4/Cr2O3 composite on an Ni foam for supercapacitors.137 | ||
Barman et al. created nano-copper chromite-loaded H-mordenite by combining equal quantities of copper and chromium salt precursors, followed by the addition of triethanolamine, while continuously stirring. To avoid precipitation, the pH was adjusted using HNO3. The solution was heated, and the resulting fluffy mass was calcined at 923 K. The particle size was found to be 31 nm by TEM examination. The nano-copper chromite was then blended with H-mordenite in different ratios, mixed with 20% bentonite as a binder, extruded, and dried overnight at 373 K.138 By thermally breaking down Cu(II) and Cr(III) carboxylate-type precursors that were generated in situ inside a silica matrix by a redox reaction with 1,3-propanediol, Ştefănescu et al. created CuCr2O4/SiO2 and Cu2Cr2O4/SiO2 NCs that included 50 wt% of the corresponding oxides. Upon heating, the precursors decomposed at around 300 °C into amorphous CuO and Cr2O3−x. Cr2O3−x crystallized to α-Cr2O3 at 400 °C, which reacted with CuO to form Cu2Cr2O4. Further heating to 800 °C and 1000 °C resulted in the formation of residual CuO and well-crystallized Cu2Cr2O4, respectively. In both composites, the oxides were uniformly distributed in the amorphous silica matrix. The samples produced at various annealing temperatures were analyzed using thermal techniques.139
Abbasi and colleagues used the sol–gel process to create spherical CuCr2O4 NPs with various amino acids as capping agents. They successfully examined how the molar ratio of the stabilizing agent affected the purity, particle size, and shape of the copper chromite. Moreover, a photodeposition method was used to create CuCr2O4/Ag nanostructures. They reported that the surface roughness and particle size increase in the CuCr2O4/Ag nanostructures compared to pristine CuCr2O4, indicating effective surface modification through Ag coating and potential impact on photocatalytic capabilities.140
Table 3 summarizes the diverse fabrication strategies, characterization techniques, and applications of CuCr2O4 nanocomposites.
| Nanocomposites | Synthesis method | Morphology | Particle size (nm) | Characterization techniques | Applications | Ref. |
|---|---|---|---|---|---|---|
| Ag-coated CuCr2O4 | Sol–gel | — | — | XRD, SEM-EDS, TEM, TG-DSC | Li-ion batteries | 46 |
| CuCr2O4·Pd/γ-Al2O3 | Sol–gel and impregnation | Rod | — | XRD, SEM-EDS, BET | Catalytic activity | 109 |
| Bi2O3/CuCr2O4–CuO core/shell | Co-precipitation | Spherical | 78 | XRD, TEM, EDS, FTIR | Catalytic activity | 111 |
| CuCr2O4 | Co-precipitation | Flake | — | XRD, UV-vis-DRS XPS, SEM-EDAX, SAED, BET, EIS, PL | Photocatalytic activity and genotoxic effect | 45 |
| CuCr2O4–BiI3O9 | Sono chemical | CuCr2O4 | ||||
| Cube | ||||||
| BiI3O9 | ||||||
| CuCr2O4 | Sol–gel combustion | Spherical | 30–80 | XRD, UV-Vis, DRS, FESEM, TEM, HRTEM, EDX, XPSPL, BET, Raman spectroscopy | Photocatalytic and electrochemical activity | 112 |
| CuCr2O4/BiOBr | Co-precipitation | |||||
| CuCr2O4 | Sol–gel | Spherical | 65 | XRD, UV-vis, FTIR, PL | Photocatalytic activity | 141 |
| BiVO4 | Chemical route | |||||
| CuCr2O4@CaFe-LDO | — | — | — | XRD, UV–Vis, DRS XPS, SEM-EDX, TEM, TG–DTA | Photocatalytic activity | 114 |
| CuCr2O4@CaFe-LDO | — | — | 12.51 | — | Photocatalytic activity | 113 |
| CuCr2O4–CeO2 | Sol–gel combustion | Semi-spherical | 15 | XRD, UV-Vis DRS, TEM, EDS, XPS, Raman spectroscopy, PL, BET | Photocatalytic activity | 115 |
| Cu/CuCr2O4 | Impregnation | — | 50–100 | XRD, FTIR, HRTEM, FESEM, XPS, XANES/EXAFS | Catalytic activity | 116 |
| CuCrO2/CuCr2O4 | Ball milling | — | 174 ± 5 | XRD, UV-vis, FESEM, XPS, DTA, FESEM, EDS, TEM, SAED | Photocatalytic activity | 127 |
| CuCr2O4/CuO | Co-precipitation | — | — | SEM/EDAX, ICP/AES, XRD, and XPS | Catalytic activity | 142 |
| CuO–CuCr2O4 | Reflux condensation | Flower | — | XRD, UV-vis-DRS, TEM, PL, FESEM | Photocatalytic activity | 26 |
| EP/PU/CuCr2O4 | Sol–gel auto combustion | Octahedral | — | XRD, UV-vis, FTIR, SEM, XPS, TG–DSC | — | 128 |
| CuCr2O4–g-C3N4 | Ultrasonication-assisted | Irregular | 12–16 | XRD, FTIR, UV–vis DRS, TEM, PL, XPS, BET, EDX | Photocatalytic activity | 117 |
| g-C3N4–NS/CuCr2O4 | Refluxing | — | 12.6 | XRD, UV-Vis DRS, FTIR, SEM, EDX, TEM, XPS, PL, BET, TGA | Photocatalytic activity | 119 |
| CuCr2O4/GO | Co-precipitation | Quasi-spherical | GO-1.39 | XRD, SEM, TEM, EDS, PL, Raman spectroscopy | Energy storage and electrochemical activity | 143 |
| CuCr2O4/GO-3.54 | ||||||
| CuCr2O4 and MWCNT/CuCr2O4 | Co-precipitation | Quasi-spherical | — | XRD, UV–Vis, FTIR, XPS, SEM-EDS, BET, HRTEM-SAED | Supercapacitor | 129 |
| MXene/CuCr2O4 | Co-precipitation | — | CuCr2O4-21.2, Ti3C2Tx-25.5 | UV-vis, SEM-EDX, zeta potential, Raman spectroscopy, PL, FTIR | Energy storage | 122 |
| Ti3C2Tx/CuCr2O4-8.7 | ||||||
| N-doped graphene CuCr2O4 | Sonication-assisted mechanical mixing | — | — | XRD, FTIR, XPS, SEM-EDS, STEM | Supercapacitor | 121 |
| N-doped rGO CuCr2O4 | Sol–gel | — | — | XRD, FTIR, SEM-EDX | Supercapacitor | 130 |
| PANI: CuCr2O4 | — | Nano spherical | — | XRD, UV–Vis, FTIR, XPS, BET, TEM, and PL | Photocatalytic activity | 123 |
| CuCr2O4–PS | Low-temperature ultrasonic | — | — | XRD, FTIR, UV–Vis, SEM-EDS, BET | Photocatalytic activity | 131 |
| CuCr2O4/rGO | Hydrothermal | — | 50 | XRD, FTIR, TEM, TGA, XPS, zeta potential, Raman spectroscopy | Catalytic activity | 132 |
| CuCr2O4/SiO2 | — | Frisbee | — | XRD, UV-vis-NIR, SEM, IR | Solar driven water evaporation | 133 |
| CuCr2O4/SiO2 | Sol–gel | — | — | XRD, TEM | — | 134 |
| CuCr2O4/SnO2 | Sol–gel | — | — | XRD, XPS, FTIR | Photocatalytic activity | 124 |
| CuCr2O4 | Sol–gel | Spherical | 25–30 | XRD, TEM-SAED, SEM-EDX, BET | Photocatalytic and photoeletrocatalytic activity | 48 |
| TiO2 | Sonication | Semi sphere | 6–10 | |||
| CuCr2O4/TiO2 | Co-precipitation | — | 54 | XRD, UV-vis | Dye sensitized solar cell | 125 |
| CuCr2O4/TiO2 | Sol–gel | — | 50 | XRD, UV-Vis-DRS, TEM | Photocatalytic activity | 144 |
| CuCr2O4·0.7TiO2–rGO | Sol–gel assisted Hummers’ method | Spherical | 100 | XRD, FTIR, SEM-EDX, XPS, TGA-DTA, Raman spectroscopy | Catalytic activity | 145 |
| CuCr2O4/TiO2 | Sol–gel | Spherical | 25–40 | XRD, SEM, TEM | Catalytic activity | 146 |
| CuCr2O4/TiO2 | Sol–gel | — | 66 | XRD, DRS-UV, BET, FT-IR | Photocatalytic activity | 118 |
| V2O5/CuCr2O4/MCM-41 | Hydrothermal | CuCr2O4/MCM-41-hexagonal | 32 | XRD, UV–Vis, FTIR, BET, HRTEM, SEM-EDX, TGA | Photocatalytic and antibacterial activity | 135 |
| V2O5-spherical | 6 | |||||
| CuCr2O4 nanocomposites | Sol–gel | Pseudospherical | 50–500 | XRD, SEM, EDAX, FTIR | Photocatalytic activity | 136 |
| CuCr2O4–Cr2O3 | Co-precipitation | Spherical | ±25 | XRD, FTIR, UV-Vis, SEM, EDX TEM, SAED, XPS, EIS, BET | Supercapacitors | 137 |
| 50 mass% CuCr2O4/SiO2 | Modified sol–gel | — | 43 | XRD, FTIR | — | 139 |
| 50 mass%Cu2Cr2O4/SiO2 | 50.5 | |||||
| CuCr2O4/Ag-leucine | Sol–gel | Quasi-spherical | 30–60 | ICP-OES, DRS, XRD, SEM, EDS, TEM | Photocatalytic activity | 140 |
3. The impact of experimental parameters on CuCr2O4 NPs physico-chemical properties
3.1. pH
According to Pakzad et al., particle size is significantly affected by pH and the amount of fuel. Doubling the amount of fuel greatly increases the particle size. They observed that low citric acid (4 mmol)![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) glycine (10 mmol) and high citric acid (8 mmol)
glycine (10 mmol) and high citric acid (8 mmol)![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) glycine (20 mmol) ratios resulted in average particle sizes of 0.74 and 5.18 μm, respectively. It was also discovered that as the fuel/oxidizer ratio increased, the particle shape shifted toward octahedral structures, with larger particles observed at pH 9. They concluded that particle size increases with an increase in the fuel/nitrate ratio or solution pH.17
glycine (20 mmol) ratios resulted in average particle sizes of 0.74 and 5.18 μm, respectively. It was also discovered that as the fuel/oxidizer ratio increased, the particle shape shifted toward octahedral structures, with larger particles observed at pH 9. They concluded that particle size increases with an increase in the fuel/nitrate ratio or solution pH.17
3.2. Time
Mobini et al. investigated the effect of aging time on material structure and found that prolonged aging significantly influenced crystallinity. They reported that the surface area increased from 20.4 to 42.8 m2 g−1 as the aging period progressed. At the same time, the overall crystallinity decreased, accompanied by an increase in amorphous content and the development of different crystalline phases.77 Chiu et al. delved into the impact of time on nanoparticle morphology. The topology of the fibers was altered significantly as the sintering time increased. All the fibers had a coarse morphology, and this roughness increased with sintering time.88 Surprisingly, researchers aimed to study the development of 3D raspberry-like structures. They conducted time-dependent experiments at 180 °C utilizing a hydrothermal approach. As seen in Fig. 14, after 18 h of crystallization, the particles formed a raspberry-like shape with homogeneous diameters. However, prolonged crystallization (>18 h) led to the development of polydispersed particles.78 Similarly, Yuan et al. used a Teflon-lined stainless-steel autoclave heated to 180 °C and maintained for 11 h to synthesise cubic-like CuCr2O4 particles.22 | ||
| Fig. 14 SEM images of CuCr2O4 NPs synthesized using hydrothermal techniques at 180 °C for (a) 6 h, (b) 18 h, (c) 72 h, (d) 7 days, (e) after 7 days of constant stirring, and (f) after 7 days of aging.78 | ||
3.3. Concentration
Sankudevan et al. found that increasing the Mn doping concentration in CuCr2O4 NPs reduced the particle size from 37 to 26 nm, as confirmed by XRD analysis.43 Pure CuO and CuO–CuCr2O4 NCs were made with various Cr3+ concentrations. The monoclinic structure of pure CuO is reflected in its XRD. CuO–CuCr2O4 with a 10% Cr3+ concentration exhibits a weak peak at 18.12°, indicating the presence of CuCr2O4, which has a tetragonal structure. The CuCr2O4 peak intensifies when the Cr3+ concentration increases (CuO–CuCr2O4: 20% Cr3+ concentration and CuO–CuCr2O4: 30% Cr3+ concentration), whereas the CuO peaks diminish. The crystallite size decreases with an increase in Cr3+ content, indicating that CuCr2O4 inhibits CuO growth and may diminish the lattice characteristics of CuO.26 Batool et al. reported that the grain size reduced when the Cd and Nd doping concentrations increased, which was further confirmed by SEM analysis.102 Ramezanalizadeh et al. investigated the effect of varying the La loading on the morphology, size, and crystallinity of CuCr2O4 NPs using FESEM analysis. According to the experimental findings, the shape of the nanostructure changed dramatically with an increase in La concentration, going from plate-like at 0.35 La loading to spherical-like at 0.7, as shown in Fig. 15. Additionally, a marked change in particle size was observed. These structural and morphological changes, as supported by XRD data, indicate that the addition of La to the CuCr2O4 lattice has a significant impact on the crystalline phases and physical properties of the material.100 | ||
| Fig. 15 FESEM images of CuCr1.65La0.35O4 (A–C), CuCr1.3La0.7O4 (D–F), and CuCrLaO4 (G–I).100 | ||
3.4. Temperature
Hosseini et al. investigated several factors, including the Cu/Cr ratio and calcination temperature in the range of 500–750 °C, to examine the formation of diverse phases, including CuCr2O4, CuO, and CuCrO2. It was established that the phase composition of the resulting CuCr2O4 NPs is determined by the molar ratio of Cu–Cr in the initial reactants. When the reaction temperature is set to 600 °C and the Cu/Cr molar ratio is 0.5, pure spinel CuCr2O4 NPs are obtained.50Tavakoli et al. demonstrated that the phase structure of the CuCr2O4 compound depends on the initial reactant temperature and the molar ratio of Cu![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Cr.70 Pal et al. observed that CuCr2O4 samples synthesized at 400–500 °C formed NPs with slight agglomeration, while calcination at 550 °C led to significant particle growth and the formation of predominantly octahedral-shaped structures. The particle size increased in tandem with the calcination temperature, primarily due to the aggregation of smaller particles at higher temperatures.71 Chiu et al. tested a sample sintered at temperatures in the range of 500–800 °C for varying periods of time. The fiber sintered at 500 °C included a combination of Cr2O3 and CuCr2O4, but sintering at 600–800 °C produced single-phase CuCr2O4. The mean fiber diameter reached 500 nm. Secondary-phase Cr2O3 was discovered when the fiber was sintered at 700 °C for 5 and 10 min, while single-phase CuCr2O4 occurred when the sintering time was raised to 15–30 min.88 Mobinia's study suggested that the thermal sintering of particles at high temperatures causes the BET surface area to decrease, given that sintering reduces the porosity and crystal size.77
Cr.70 Pal et al. observed that CuCr2O4 samples synthesized at 400–500 °C formed NPs with slight agglomeration, while calcination at 550 °C led to significant particle growth and the formation of predominantly octahedral-shaped structures. The particle size increased in tandem with the calcination temperature, primarily due to the aggregation of smaller particles at higher temperatures.71 Chiu et al. tested a sample sintered at temperatures in the range of 500–800 °C for varying periods of time. The fiber sintered at 500 °C included a combination of Cr2O3 and CuCr2O4, but sintering at 600–800 °C produced single-phase CuCr2O4. The mean fiber diameter reached 500 nm. Secondary-phase Cr2O3 was discovered when the fiber was sintered at 700 °C for 5 and 10 min, while single-phase CuCr2O4 occurred when the sintering time was raised to 15–30 min.88 Mobinia's study suggested that the thermal sintering of particles at high temperatures causes the BET surface area to decrease, given that sintering reduces the porosity and crystal size.77
Billman et al. suggested that the modified Pechini method produces smaller particle sizes and increased agglomeration. The measured particle growth kinetics are consistent with a diffusion-limited, inhibited grain growth model.58 Similarly, Tsai et al. revealed that higher temperatures increase the particle size by boosting atomic diffusion, and also enlarge the voids by inducing atoms to cluster around the void margins due to capillary forces. They further reported that annealing the nanostructure at 500 °C transformed the film structure from amorphous to a mixture of monoclinic CuO and spinel CuCr2O4. Delafossite CuCrO2 was formed by further annealing at 600 °C, which depleted the CuCr2O4 phase. Increasing the annealing temperature also altered the microstructure, evolving from compact nanograins to larger agglomerated grains with voids, resulting in a rougher surface.89 Similarly, Batool et al. also observed the effect of temperature on the morphology of CuCr2O4 NPs doped with Mg. The researchers observed that at lower temperatures, the particles exhibited quasi-spherical shapes with low homogeneity. As the temperature increased, the particle size increased, and the shapes became less uniform. The particles showed higher aggregation, less spherical appearance, and more disorder at 750 °C.105
3.5. Fuel
Capping agents play a crucial role in the synthesis of nanoparticles by increasing the monodispersity, reducing the particle size, and preventing the aggregation of NPs. Sathiskumar and colleagues studied Cu–Cr–O NC oxide catalysts manufactured using various Cu–Cr mole ratios to better understand the impact of the mole ratio on the final product, using fuels such as glycine and citric acid.8 Bisht et al. utilized capping agents, namely, urea, coconut oil, oleic acid, and citric acid, to generate CuCr2O4 NPs. It was observed that improved surface capping occurred in the presence of oleic acid.82 Paymenfar et al. studied the influence of a capping agent on CuCr2O4 NPs using FESEM analysis. According to the analysis, they concluded that the uncapped particles exhibited a semi-spherical morphology with noticeable aggregation and low monodispersity. In contrast, the use of sucrose as a green capping agent reduced the particle size, minimized the agglomeration, and greatly enhanced the monodispersity. The findings demonstrate the importance of the capping agent in regulating the shape and crystallinity of nanoparticles during sol–gel production.51 To adjust morphology and particle size, Kumar et al. employed a variety of surfactants, including PEG, CTAB, and PVP. Among them, the CuCr2O4 material made with CTAB surfactant exhibits the greatest specific capacitance due to its larger surface area, rougher surface, and enhanced ion adsorption active sites resulting from its spherical shape.73 Al-Wasidi et al. created CuCr2O4 NPs with and without citric acid and tartaric acid as templates. The presence of organic acids caused lower crystallite sizes. Organic templates had a considerable influence on crystallite size and morphology; the nanoparticles produced without templates had irregular forms, but those with templates were spherical.81 The impact of glycine, alanine, valine, and leucine as capping agents in the synthesis of CuCr2O4 NPs was examined by Ali Abbasi et al. Glycine, the smallest amino acid, has a weak steric barrier due to its tiny molecular size, resulting in the development of agglomerated particles with an unknown shape. In contrast, the NPs synthesised with leucine as both a capping and reducing agent have a well-defined, sphere-like shape. Among the amino acids investigated, leucine exhibited the most pronounced steric hindrance effect, resulting in more uniform and smaller particles. Based on the structural properties of the amino acids, it can be concluded that larger amino acids provide more steric hindrance, which is advantageous for the controlled synthesis of subminiature NPs with enhanced shape and reduced agglomeration.140Table 4 describes the diverse parameter-dependent fabrication of CuCr2O4 nanomaterials.
| Synthesis method | Parameters for the synthesis of CuCr2O4 NPs | Ref. | ||||||
|---|---|---|---|---|---|---|---|---|
| Concentration of Cu salt or weight | Concentration of Cr salt or weight | Molar ratio Cr3+![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Cu2+ Cu2+ |
pH | Temperature | Time (h) | |||
| Reaction (°C) | Calcination (°C) | |||||||
| Sol–gel | 0.01 mol | 0.02 mol | — | — | 95 | 600 | 3 | 50 |
| — | — | — | — | 50 | 700 | 6 | 52 | |
| 0.01 | 0.02 | — | — | 90 | 750 and 850 | 4 | 53 | |
| — | — | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
8.5 | 90 | 300 and 850 | 6 and 4 | 42 | |
| 2 mmol | 4 mmol | — | — | 70 | — | — | 55 | |
| — | — | — | 10–11 | 80 | 820 | 4 | 56 | |
| 0.799 g | 1.833 g | — | — | 180 | 600 | 4 | 60 | |
| — | — | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
7 | 120 | 900 | 24 | 46 | |
| — | — | 0.5 | 3 | — | 550 | 5 | 109 | |
| 0.005 mol | 0.01 mol | — | — | 80 | 800 | 1.5 | 141 | |
| — | — | — | — | 95 | 750 | 4 | 143 | |
| 0.1 mol | 0.2 mol | — | — | 50–65 | 500 | 2 | 121 | |
| — | — | — | — | 40 | 800, 900, 1000, 1100 | 2 | 134 | |
| 0.005 mol | 0.01 mol | — | — | 80 | 300, 500, 700 | 1.5 | 144 | |
| 5 mmol | 10 mmol | — | — | 80 | 600 | 3 | 146 | |
| 5 mM | 10 mM | — | — | 95 | 600 | 3 | 118 | |
| 0.01 | 0.02 | — | ∼7 | 90 | 750 | 4 | 102 | |
| 0.01 mol | 0.02 mol | — | — | 95 | 600 | 3 | 99 | |
| 2.0 g | 10 g | — | 8 | 180 | 750 | 6 | 27 | |
| 0.01296 | 0.02592 | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
— | 90 | 800 | 3 | 139 | |
| 0.5 g (2 mmol) | 1.658 g (4 mmol) | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
7 | 70 | 1000 | 2 | 62 | |
| 1 mmol | 2 mmol | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
— | 70 | 650 | 3 | 140 | |
| Sol–gel auto combustion | 7.5 mmol | 15 mmol | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
8–10 | 90 | 950 | 1 | 17 |
| — | — | — | — | 120 | 900 | 10 | 59 | |
| 0.9053 g | 3 g | — | — | 80 | 700 | 4 | 112 | |
| 2.152 mmol | 4.298 mmol | — | 7 | 70 | 700 | 3 | 128 | |
| 0.9057 g | 3 g | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
— | 80 | 700 | 4 | 48 | |
| Sol–gel | 1 mmol | 2 mmol | — | — | 80 | 650 | 3 | 49 |
| Green synthesis | 1 mmol | 2 mmol | — | — | 100 | 650 | 3 | |
| Pechini | 0.07 mol | 0.1 mol | — | — | 95 | 600 | 3 | 57 |
| Pechini-and modified Pechini | — | — | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
7 | 80 | 650, 750, and 850 | 1 and 24 h | 58 |
| Combustion | 36.23 g | 60 g | — | — | — | 750–800 | 2 | 65 |
| 0.5 M | 1 M | — | — | 400 | 5 | 101 | ||
| Self-combustion | 10 mmol | 20 mmol | — | — | 90 | 600 | 3 | 64 |
| Co-precipitation | 1.4 g | 4.0 g | — | 10 | 80 | 600 | 5 | 16, 38 and 67–69 |
| 0.020 mol | 0.040 mol | — | — | — | 400, 450, 500 and 600 | 3 | 71 | |
| 0.35 M | 0.35 M | — | — | — | 300 | 6 | 92 | |
| Reduced with H2 300 | 8 | |||||||
| 1.208 g | 4.0015 g | — | — | 110 | 600 and 700 | 4 | 23 | |
| 0.5 M | 0.5 M | — | 8 | 90 | 800 | 5 | 111 | |
| 0.1 M | 0.2 M | — | 11 | 80 | 700 | 4 | 45 | |
| 0.005 mol | 0.01 mol | — | 8–9 | — | 1000 | 2 | 72 | |
| 1.70 g | 5.32 g | — | — | 85 | 400 | 4 | 129 | |
| — | — | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
— | — | 700 | 6 | 131 | |
| 0.05 mol L−1 | — | 8–9 | — | — | 900 | 1 | 98 | |
| Inverse-coprecipitation | 0.005 mol | 0.01 mol | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
9 | — | 400, 520, and 800 | 2 | 70 |
| Hydrothermal | 1.5 g | 5.0 g | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
8 | 180 | 750 | 6 | 74 and 75 |
| 3.5 g | 11 g | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
8 | 200 | 700 | - | 35 | |
| 5 mmol | 10 mmol | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
10–10.3 | 70 | 500–800 | 4 | 22 | |
| 2.25 g | 7.5 g | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
8 | 180 | 750 | 6 | 24 | |
| 5.54 g | 15.4 g | — | 7 | — | 500 | 5 | 47 | |
| 2.3 g | 7.5 g | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
8 | 180 | 750 | 6 | 76 | |
| 3.8 g | 12.3 g | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
9 | 180 | 650 | 6 | 39 | |
| 0.1 g | 0.27 g | — | — | 120 | 400 | 3 | 40 | |
| — | — | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
9, 10, 11, 12 | 160, 180 and 200 (11 h) | 400, 500, 600 and 700 | 4 | 77 | |
| 1 mmol | 2 mmol | — | 14 | 180 | 700 | 5 | 80 | |
| 2.6 g | 2.7 | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
— | 180 | 800 | 6 | 78 | |
| 1 mmol | 2 mmol | — | — | — | — | — | 93 | |
| 1 M | 2 M | — | 10–11.5 | 200 | 550 | 5 | 14 | |
| Equal | Equal | — | 7 | 200 | 750 | 5 | 95 and 103 | |
| 5.5 g | 16 g | — | 8 | 200 | 750 | — | 97 | |
| 2.30 g | 7.50 g | — | 10 | 180 | 700 | 6 | 81 | |
| Microwave | 1 mmol | 2 mmol | — | — | — | 500–800 | 3 | 82 |
| Ultrasonication assisted | 5.5 g | 15.4 g | — | — | 40 | 550 | 3 | 117 |
| Refluxing | 0.03 g | 0.19 g | — | 10 | 96 | 520 | 4 | 119 |
| — | 5.54 g | 15.4 g | — | 7 | 60 | 500 | 5 | 123 |
| — | 0.03 g | 0.07 g | — | 10 | 60 | 500 | 4 | 135 |
| Co-precipitation, ultrasound, and green chemistry | 1.7 g | 0.5 M | — | 7 | — | 650 | 4 | 41 |
4. Proposed mechanism for plant extract-mediated green synthesis of CuCr2O4 NPs
This eco-friendly process produces nanoparticles that are more stable, less prone to deformation and aggregation, and promote the adsorption of phytochemicals on their surface, thereby accelerating the reaction times.147 Lachini et al. employed an aqueous extract of Aloe vera leaves as a reducing agent for the synthesis of CuCr2O4 NPs.49Fig. 16 presents the probable mechanistic process for the green synthesis of CuCr2O4 NPs. The proposed reaction mechanism demonstrates the stabilising and reducing effects of active phytochemicals. Numerous active phytochemicals, including anthraquinones, polysaccharides, flavonoids, vitamins, enzymes, saponins, tannins, and sterols, are found in Aloe vera leaves.148–150 To suggest the possible reaction mechanism, a flavonoid has been chosen as a model biomolecule.151 The electrostatic interaction between the hydroxyl groups of flavonoids and the cations of metal precursors causes aromatic hydroxyl groups to bind to copper and chromium ions, and stable complexes are formed between the metal ions and flavonoids. After calcination, the complex breaks down, yielding CuCr2O4 NPs.Furthermore, the CuCr2O4 NPs were created by reducing Cu2+ and Cr3+ ions using bioactive phytochemicals, such as polyphenols, flavonoids, and terpenoids, which are present in rosemary leaf extract and serve as natural stabilizing and reducing agents. The reduction was then finished by adding NaBH4, and the crystalline spinel structure was achieved by calcining the mixture at 650 °C.41
5. Applications of CuCr2O4 nanomaterials
5.1. CuCr2O4 nanomaterials as catalysts
In an aqueous medium, ethyl acetoacetate, hydrazine hydrate, and aldehydes underwent a pseudo-five-component reaction to produce 4,4′-(arylmethanediyl)bis(3-methyl-1H-pyrazol-5-ols), according to Safaei-Ghomi et al. (Scheme 1).67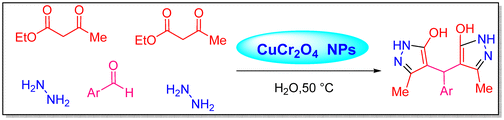 | ||
| Scheme 1 Production of 4,4′-(arylmethanediyl)bis(3-methyl-1H-pyrazol-5-ol) analogues using catalytic amounts of CuCr2O4 NPs.67 | ||
Safaei-Ghomi et al. utilized CuCr2O4 NPs as a catalyst in the Ullman reaction. C–N cross-coupling reactions between anilines and iodobenzenes were performed in toluene under reflux conditions with a nitrogen atmosphere, using KOH as the base and a CuCr2O4 nanocatalyst under ligand-free conditions. This nanocatalyst enhanced the pace and efficiency of triarylamine synthesis (Scheme 2).16
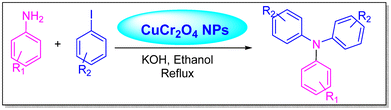 | ||
| Scheme 2 Triarylamine synthesis using CuCr2O4 NPs.16 | ||
Shahbazi-Alavi et al. described a one-pot pseudo-five-component condensation reaction utilizing a nano-CuCr2O4 catalyst to produce bis-thiazolidinones (Scheme 3). The creation of primary imine intermediates, followed by the attack of the sulphur atoms of thioglycolic acid on the activated imine groups, intramolecular cyclization, and the removal of water, results in the cyclized product bis-thiazolidines, according to the suggested mechanism. By activating the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O, C
O, C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N, and S–H groups, the nano-CuCr2O4 surface atoms improve the interaction with nucleophiles. These surface atoms are hubs for catalytically stimulating chemical processes.68
N, and S–H groups, the nano-CuCr2O4 surface atoms improve the interaction with nucleophiles. These surface atoms are hubs for catalytically stimulating chemical processes.68
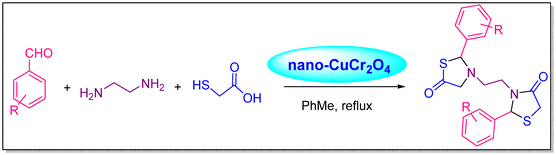 | ||
| Scheme 3 Bis-thiazolidine synthesis using nano-CuCr2O4 catalyst.68 | ||
The study by Shahbazi-Alavi et al. employed a similar process, but they incorporated ammonium acetate as a key component to alter the synthesis of arylated dipyrazolodihydropyridine derivatives. When ammonium acetate, EAA, 4-nitrobenzaldehyde, and hydrazine hydrate were reacted in ethanol at room temperature, using nano-CuCr2O4 as a catalyst, significant yields of tetrahydrodipyrazolopyridines were produced (Scheme 4). Furthermore, the recyclability of the catalyst was demonstrated up to five cycles in the model reaction.152
 | ||
| Scheme 4 Preparation of tetrahydrodipyrazolopyridines using nano-CuCr2O4.152 | ||
Wołosiak-Hnat et al. produced a CuCr2O4 catalyst with a more crystalline structure and a higher copper concentration than the commercial catalyst, enabling greater selectivity for 1,2-propanediol. At 210 °C, 80 wt% aqueous glycerol and 8 wt% catalyst resulted in the maximum glycerol conversion of 76.9% and 90.7% selectivity to 1,2-propanediol compared to the inserted glycerol, respectively. The studies were conducted under an initial hydrogen pressure of 3.0 MPa, stirring speed of 100 rpm, reaction time of 24 h, and pressure of 5.6 MPa throughout the process. Under the same conditions, the maximum selectivity for 1,3-propanediol was just 2.5% at a glycerol conversion rate of 30.9%. This study suggested that the catalyst must be present for the C–O bond in alcohols to be activated for hydrogenolysis. The hydrogenolysis of glycerol to 1,2- and 1,3-propanediol is generally considered a two-step procedure. Glycerol is dehydrated to produce acetol or 3-hydroxypropanal in the first stage. Hydrogenating these intermediates to 1,2- and 1,3-propanediol, respectively, is the second step (Scheme 5).92
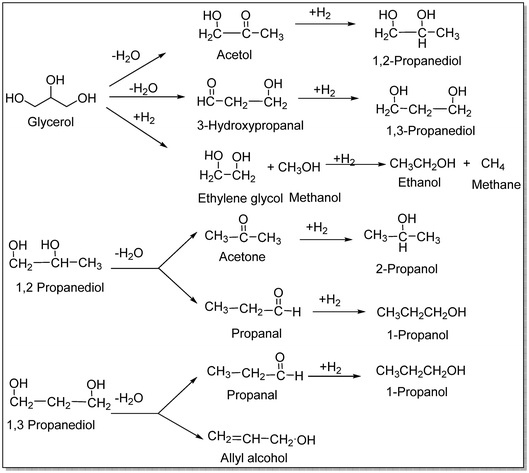 | ||
| Scheme 5 Possible pathways for the catalytic hydrogenolysis of glycerol.92 | ||
Karimi-Jaberi et al. described the formation of benzylidene bis(4-hydroxycoumarin) and dihydropyrano[c]chromene analogs in H2O at RT with nano-CuCr2O4 as a catalyst. This process is eco-friendly, consumes less time, produces high yields, and the catalyst can be reused, making it simple, practical, and sustainable (Scheme 6a and b).38
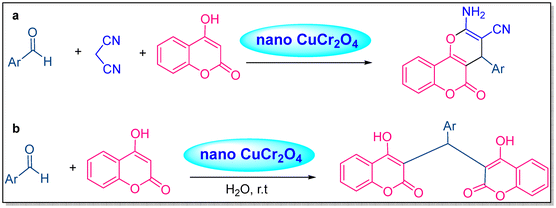 | ||
| Scheme 6 (a) Synthesis of dihydropyrano[c]chromene and (b) benzylidene bis(4-hydroxycoumarin).38 | ||
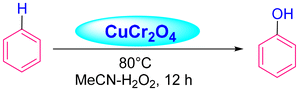
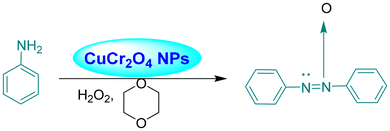
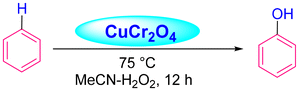
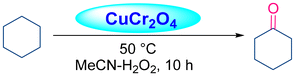
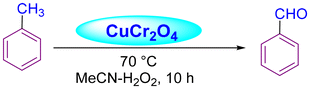
Acharyya and research team have published multiple reports on the synthesis of CuCr2O4 NPs and their catalytic activity. In this report, a nano-catalyst with improved thermal stability and enhanced catalytic performance in the hydroxylation of benzene to phenol, oxidation of ethylbenzene to acetophenone, and oxidation of aniline to azoxybenzene was generated, utilizing H2O2 as the oxidizing agent. The study found that at 80 °C, the benzene conversion was 72.5% with 94% phenol selectivity (entry 1, Table 5).74 In comparison, at 70 °C, the ethylbenzene conversion was 68.5%, yielding 78% acetophenone (entry 2, Table 5),75 aniline conversion was 78%, with 92% selectivity to azoxybenzene (entry 3, Table 5),35 at 75 °C the benzene conversion of 67% with 94% phenol selectivity (entry 4, Table 5),15 and at 50 °C, 70% cyclohexane conversion and 85% selectivity towards cyclohexanone (entry 5, Table 5),76 and 57.5% toluene conversion with 84.4% selectivity of benzaldehyde. CuCr2O4 NPs can be reused several times without losing effectiveness (entry 6, Table 5).39 Similarly, the effect of Cu(II) nanoclusters supported by CuCrO4 spinel NPs on the selective oxidation of ethanol was investigated. Without a solvent, this process yielded 65% conversion of ethanol to acetic acid with 97% selectivity at 75 °C, utilizing H2O2 as the liquid-phase oxidant.97 However, Table 5 describes the diverse organic transformations catalyzed by CuCr2O4 NPs.
In another study, Cu(II) nanoclusters supported on CuCr2O4 NPs were used to explore their catalytic activity in the sustained oxyamination conversion of benzene to aniline via a direct procedure employing NH3 and H2O2. The technique was efficient in terms of activity, selectivity, mild reaction conditions, and catalyst. The Cu(II) component was found to significantly affect both the conversion rate and product selectivity.96
Scheme 7 illustrates that ultra-small Cu(II) oxide supported on CuCr2O4 spinel NPs participates in converting benzene to phenol when air is used as an oxidant. The textural promoter Cr(III) prevents the sintering of Cu(II) oxide, thereby preserving the surface area of the catalyst during the reaction. In this process, the supported Cu(II) species is reduced by benzene to Cu(I) species A, which subsequently transitions into its canonical form B. Simultaneously, benzene generates a peroxide radical (HOO˙) and a benzene radical cation (C6H6˙+). The benzene radical cation then reacts with species B to form complex C, which subsequently converts into its canonical form D. After that, a proton is removed by complex D, restoring aromaticity and creating intermediate E. Phenol and Cu(II) oxide species F are produced when intermediate E dissociates. The interaction between species F and the concurrently generated hydroxyl radical (˙OH) results in the production of intermediate G. The catalytic cycle is finished when a benzene molecule reduces G back to Cu(I) species, creating a benzene radical cation that rejoins the cycle.24
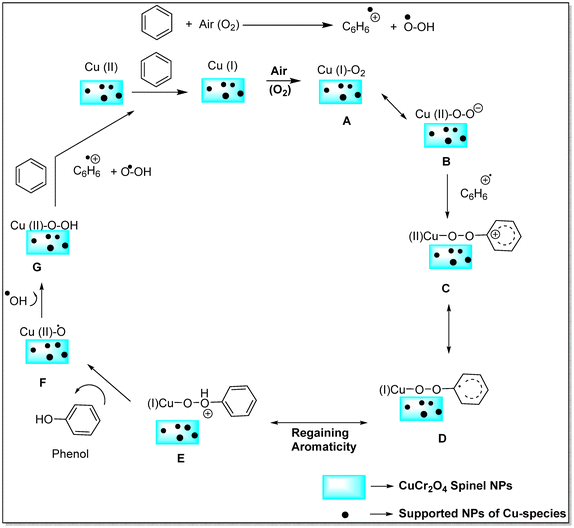 | ||
| Scheme 7 Possible process for phenol production over ultra-small Cu(II) based on CuCr2O4 NPs.24 | ||
Chen et al. found that CuCr2O4 was the most effective catalyst, achieving a cyclohexane conversion rate of up to 95.8% and a cyclohexanone selectivity of 78.1%. The catalyst also showed good cycle stability after five cycles.132
Abdolmohammadi's research shows that CuCr2O4 NPs have a larger surface area due to their acidic coordinated sites. CuCr2O4 NPs interact with the carbonyl oxygen group in (E)-1,2-diphenyl-1-diazene to generate an intermediate. This intermediate then undergoes a Knoevenagel reaction with 4-hydroxycoumarin, resulting in the formation of an alkene. Following that, a Michael-type addition occurs, in which another mole of 4-hydroxycoumarin interacts with the alkene. After cyclization and dehydration procedures, 7-(4-hydroxy-2-oxo-2H-[1]benzopyran-3-yl)-9-[(E)-2-(phenyl)-1-diazenyl]-6H,7H-[1]benzopyrano[4,3-b][1]benzopyran-6-one is produced (Scheme 8).40
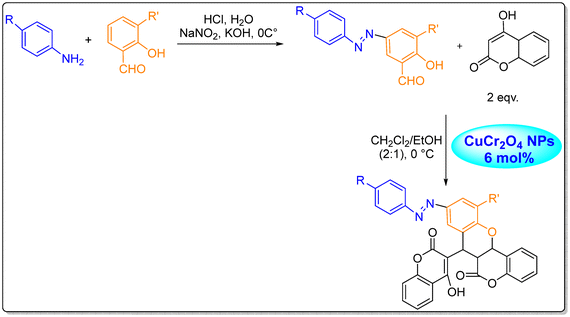 | ||
| Scheme 8 Formation of [1]benzopyran azo dyes catalyzed by CuCr2O4 NPs.40 | ||
The long-term stability test results show that the catalyst exhibited excellent stability, even at 400 °C in the presence of CO2 and water vapor. The calcination temperature is a key factor in improving the catalytic performance in the oxidation of CO.77
Interestingly, a zinc oxide-doped, strongly acidic zeolite catalyst increased the formation of a toluene intermediate during the catalytic pyrolysis of cellulose. As shown in Fig. 17, CuCr2O4@MCM-41 exhibited strong catalytic activity. H2O2, as an oxidizing agent, under optimal conditions of 60 °C and 4 h, resulted in 87.5% cresol selectivity and 83.0% conversion.153
 | ||
| Fig. 17 CuCr2O4@MCM-41 catalyst used for the efficient catalytic pyrolysis of cellulose and catalytic oxidation.153 | ||
Scheme 9 depicts a multicomponent reaction involving 3,4-methylenedioxyphenol, aromatic aldehydes, and C–H-activated acidic compounds, such as dimedone. This method confirmed the universality of the route by producing high yields of xanthenones and chromenopyrimidinediones. Water was used as a green, non-volatile solvent, and ultrasonic irradiation was employed as a safe energy source, enabling the quick synthesis of products.154
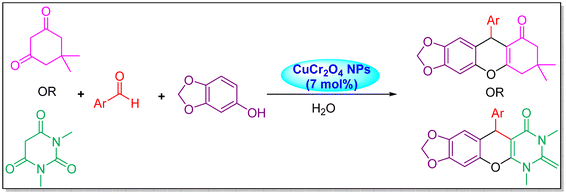 | ||
| Scheme 9 Ultrasound-based, green, and sustainable method for xanthenone and chromenopyrimidinedione derivatives.154 | ||
Kien and colleagues examined the fact that Pd may enhance the catalytic efficacy of spinel CuCr2O4 and Pd catalytic processes for the oxidation of carbon monoxide.109 Using 0.100 g of catalyst, a temperature range of 30–450 °C, and a gas flow of 15 L h−1, the study employed the temperature programmed surface reaction (TPSR) method to investigate a mixture of 1% CO, 20% O2, and 79% N2. The overall catalytic activity was considerably enhanced by distributing Pd NPs on CuCr2O4 NPs.109 The core/shell NPs demonstrated promising catalytic activity for CO oxidation.111 According to the researchers, HCOOH is more difficult to create than CH3OH due to its more complex chemical structure. The conversion of CO2 into CH3OH is most likely the predominant reaction. Furthermore, the selectivity of HCOOH and CH3OH at 110–180 °C with calcined Cu/CuCr2O4 catalysts demonstrates alterations in the CO2 conversion pathway.116
Shojaei et al. investigated the catalytic activity of CuCr2O4 and CuCr2O4/TiO2 with an optimal chromite-to-TiO2 molar ratio, catalyst amount, oxidant type and amount, and solvents. CuCr2O4–TiO2 heterojunctions significantly enhance the catalytic activity in converting benzylic alcohols to aldehydes, particularly under microwave irradiation with t-BHP and H2O2 as oxidants.146
As seen in Scheme 10, homolytic cleavage of the H2O2 molecules gives two ˙OH radicals. The resulting radicals combine with Cr3+ and Cu2+ ions on the surface of CuCr2O4, producing Cu2+ and Cu+ ions, respectively, as well as hydroperoxyl. The hydroperoxy radicals also further react with hydroxyl radicals on the surface, producing water and oxygen. Veratryl alcohol (VA) molecules are absorbed into the surface of CuCr2O4, generating metal–alkoxide intermediates and adsorbing hydrogen through O–H bond breakage. The surface O–H group is generated when the adsorbed hydrogen bonds with the surface lattice oxygen and coordinates with the carbon atom of the methyl group in the VA molecule. After activating the C–H bond in the β site, veratraldehyde and water are formed.101
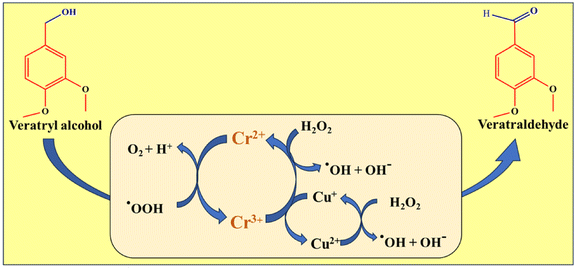 | ||
| Scheme 10 Oxidation of VA to veratraldehyde.101 | ||
Ghasemi et al. utilized CuCr2O4 NPs, along with a basic ionic liquid, to fabricate cyclopropanecarboxylic acids. They fabricated (±)-trans-2-phenylcyclopropanecarboxylic acid using trans-cinnamic acid as the starting material, along with the NPs. They utilized various solvent forms, but THF gave the optimal results (Scheme 11).155
 | ||
| Scheme 11 Reaction of functionalized trans-cinnamic acids via cyclopropanation.155 | ||
Barman et al. presented a method for hydrogenating acetone using a catalyst. Firstly, chromite reversibly reacts with hydrogen, resulting in the formation of a decreased spinel and the release of copper as surface-bound particles. Acetone adsorbs onto Cu0 atoms as π-complexes, getting electrons from the metal and a proton from chromite, resulting in negative charges. The oxidised copper returns to the spinel. A second proton is injected, and a proton from the acidic medium combines with the OH groups to produce a carbonium ion and water. This ion conducts electrophilic substitution with benzene to form cumene (Scheme 12).138 As depicted in Fig. 18, the catalytic performance of CuCr2O4 NPs for various organic transformations was demonstrated.
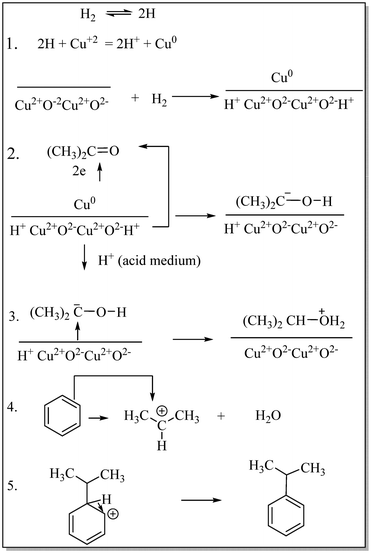 | ||
| Scheme 12 Mechanism of reductive isopropylation of benzene with acetone.138 | ||
Patil and colleagues studied the thermal decomposition of p-type nano-CuO and CuCr2O4 using AP. The DSC analysis concluded that the electrochemical technique produced NPs with better catalytic effects on HTD and AP. Nano-CuCr2O4 outperformed nano-CuO in reducing high-temperature decomposition by 118 °C at a 2 wt% concentration. Nano-CuO and CuCr2O4 produced significant heat emissions of 5.430 and 3.921 kJ g−1, respectively.156 A similar investigation was conducted by Shukri Ismael et al. They proposed that the transition metal Cr3+ in CuCr2O4 exhibits hole conductivity due to its partially filled outermost d-orbitals. It rapidly takes electrons released by ClO4−, generating Cr2+, which then migrates to the surface of the catalyst and reacts with NH4+ to generate ammonia and hydrogen atoms. As a result, the AP ion and its intermediate products can absorb electrons, accelerating the thermal deterioration of AP (Fig. 19).79
 | ||
| Fig. 19 Schematic of the CuCr2O4 catalyst for AP decomposition.79 | ||
Ashraf et al. reported that copper chromite was efficiently incorporated into an ammonium nitrate (AN) matrix. The possible effect of copper chromite on the thermal behavior and decomposition enthalpy of AN was examined using TGA and DSC. Copper chromite had a considerable impact on the primary breakdown process of AN. AN decomposition was entirely transformed from a strong endothermic reaction (+1554 J g−1) to a strong exothermic reaction (−1625 J g−1).93 Nano-CuCr2O4 noticeably enhances the degradation temperature of AP/HTPB (ammonium perchlorate/hydroxy-terminated polybutadiene propellant) and AP/CMDB (ammonium perchlorate/composite modified double-base propellant), hence increasing the apparent heat of degradation. Furthermore, the burning rates of nano-CuCr2O4–AP/HTPB and nano-CuCr2O4–AP/CMDB propellants increased to 1.60 mm s−1 and 3.23 mm s−1, representing an increase of 28.3% and 26.3%, respectively.19
The TGA/DSC study verified that nano CuCr2O4 had a higher catalytic effect on ammonium dinitramide (AND) than nano CuO. Fuyao Chen and team believed that this could be because the catalytic activity of the two NPs differed for the preliminary decomposition product (AN) of ADN. They also conducted an ignition test on raw ADN and several copper-based catalyst/ADN composites. Raw ADN produced only a small amount of smoke at 200 ms, with the most intense decomposition time at 514 ms. The entire decomposition process of four distinct catalysts/ADN composites took less than 500 ms, indicating that copper-based catalysts can greatly facilitate ADN decomposition, and nano CuCr2O4 had a stronger catalytic effect on ADN decomposition than nano CuO. Moreover, researchers explored the adsorption capacity for NH3 on the selected typical crystal face of nano CuCr2O4 (211) and the typical crystal faces of nano CuO (002) and (200). The (211) crystal face of nano CuCr2O4 showed the greatest negative value of Eads among the three models, implying that nano CuCr2O4 has stronger adsorption ability to the NH3 molecule than that of nano CuO, which may be the primary explanation for the excellent catalytic performance of nano CuCr2O4 for AN (Fig. 20).84
 | ||
| Fig. 20 Schematic of the calculation and modeling process of adsorption heat between nano CuCr2O4 and NH3.84 | ||
Li et al. proposed that the efficient phase interaction between spinel CuCr2O4 and delafossite CuCrO2 is present in Cu–Cr–O NCs. The catalyst with Cu/Cr of 0.7 shows excellent catalytic performance, a low-pressure exponent (0.43), and a high burning rate at all pressures.57 Hosseini et al. found that adding CuCr2O4 NPs to pure AP decreases its decomposition temperature by 56 °C to 84 °C and increases the apparent heat released by 516 to 608 J g−1.50 Several studies have shown that AP decomposes at high temperatures.85,90,142 CuCr2O4 NPs demonstrated remarkable catalytic activity on ultrafine AP, as revealed by the lowest high-temperature decomposition temperature and Gibbs free energy of the nano-CuCr2O4/ultrafine AP composite generated via ball milling for 6–12 h.85
Wang et al. investigated the catalytic effects of chromate NPs on the thermal decomposition of FAP and CL-20. The inclusion of chromate nanoparticles significantly decreased the decomposition temperature of FAP. Specifically, the produced CuCr2O4 NPs reduced the decomposition temperature of FAP by 57.7 °C, demonstrating that CuCr2O4 has significantly greater catalytic effectiveness than other chromates.56 Viswanath et al. studied nano CuCr2O4 as a ballistic modifier in a typical solid rocket propellant (SRP) composition. According to the report, 17.5% CuO was required to achieve the desired phase for its application as a potential ballistic modifier. This excess CuO in CuCr2O4 enhances the burn rate of the typical SRP.65 The addition of 5 wt% Cu–Cr–O–0.7TiO2–rGO catalyst to AP considerably improved the thermal decomposition. Compared to the industrial catalyst (activated CuCr2O4), it showed a 133.61% greater solid composite propellant (SCP) burn rate. With 0.6% catalyst in AP, the burn rate increased from 4.866 mm s−1 (pure AP) to 8.531 mm s−1, exceeding that of the industrial catalyst of 6.385 mm s−1 at 33 bar and displaying good ballistic performance.145 The superior catalytic performance of metal-doped CuCr2O4 can be attributed to the presence of mixed active sites, including Co2+ and Ba2+ sites, as well as Cu2+ and Cr2+ sites. The mechanism indicates that the interaction between the catalyst and AP, which benefits from more active metal sites in the doped catalyst, demonstrates significant potential for AP decomposition. Furthermore, factors such as pore volume and the presence of active sites, as presented by doping, are also critical in influencing the catalytic performance.95
5.2. CuCr2O4 nanomaterials as photocatalysts
Environmental issues, notably water pollution, have received much attention in the last few years.157 One of the main types of contaminants in wastewater is dyes.158,159 Wastewater containing dyes has a significant impact on humans, animals, and the ecosystem because many dyes are not readily biodegradable and are highly toxic to the human body and aquatic life.160,161 Photocatalytic activity has garnered significant attention recently due to its potential applications in environmental remediation, energy conversion, and sustainable chemical processes. This study investigated the effects of using a catalyst, without a catalyst, and with hydrogen peroxide (H2O2). Recent studies have illustrated that H2O2 is essential for photocatalytic activity and significantly accelerates the degradation rate.162 Paul et al. proposed that CuCr2O4 NPs act as photocatalysts by absorbing solar energy, exciting electrons from the valence band (VB) to the conduction band (CB) and leaving holes in the VB. Dye is oxidized into innocuous chemicals by these holes, and hydroxyl radicals (OH˙) are produced through the reduction of H2O2. The high bandgap energy and vast surface area of CuCr2O4 NPs promote the formation of electron–hole pairs, while inhibiting recombination, significantly increasing the photocatalytic efficiency.23Pan et al. suggested that a pH of 9 plays a crucial role in the catalytic process of the samples. At greater concentrations, p-nitrophenol was successfully destroyed by 0.3 g L−1 of CuCr2O4 calcined at 400 °C, with excellent reproducibility.71 Yuan et al. observed photocatalytic activity using Rhodamine B (RhB), methyl orange (MO), and methylene blue. The absorption peaks of RhB (554 nm) and MB (664 nm) decreased significantly with an increase in the irradiation time and almost disappeared by 60 min. In contrast, the characteristic absorption peak of MO at 464 nm only slightly decreases with an increase in exposure time.22
Soleimani and team investigated the degradation of malachite green (MG) using CuCr2O4 and Mn0.5Cu0.5Cr2O4. The results show that Mn0.5Cu0.5Cr2O4 is more effective than CuCr2O4. In the photodegradation process, morphology and surface area significantly influence the efficiency. CuCr2O4 exhibits an agglomerated shape and a lower surface area, which results in reduced photodegradation capability. The Mn0.5Cu0.5Cr2O4 sample exhibits lower overall electronegativity than CuCr2O4, which affects its band gap and facilitates easier electron transitions between the conduction and valence bands.80
Fig. 21 illustrates the predicted photocatalytic mechanism for the decomposition of tartrazine azo dye. Under visible light (≥1.25 eV), CuCr2O4 generates electron–hole pairs (eqn (11)). Electrons react with oxygen to form ˙O2− (eqn (12)). At the same time, holes in the valence band interact with H2O to produce ˙OH radicals (eqn (13)). These reactive species, especially ˙OH and h+, break down the azo dye into CO2, H2O, phenols, aromatic amines, and acids such as oxalic, tartaric, and malic (eqn (14)).64
| CuCr2O4 + visible light → e−CB + h+VB | (11) |
| e−CB + O2 → ˙O2− | (12) |
| h+VB + H2O → ˙OH | (13) |
 | (14) |
 | ||
| Fig. 21 Proposed mechanism for photocatalytic degradation of a dye.64 | ||
Peymanfar et al. proposed that photocatalytic degradation under both visible and dark light conditions performed better than expected, suggesting that the capped CuCr2O4 structures and the produced CuCr2O4 function as photocatalytic materials rather than adsorbents.51
The impacts of pH, catalyst dosage, and initial concentration on photocatalytic decomposition were examined by Benrighi et al. They found that dye degradation was significantly influenced by the pH of the solution, with the best degradation taking place at pH values close to 4.0. Dye degradation decreases significantly as the pH increases from 4.0 to 10. This reduction can be related to the negative charge that anionic dyes acquire at higher pH values. It might impede degradation by repelling them from the surface of the photocatalyst due to charge repulsion.52
Han et al. examined the photocatalytic degradation of Congo red dye and antibiotics, including TCH, metformin hydrochloride (MFH), oxycline hydrochloride, and naproxen sodium (NPS). CuAl2O4 oxides reveal high photocatalytic performance in TCH and OCH, while MFH and NPS exhibit poor photocatalytic activity.86 The electronic band structure (Fig. 22a and c) and density of states (DOS) of nanomaterials were computed using the DFT function. The Fermi values of CuCr2O4 and BiI3O9 were 4.32 and 1.41 eV, according to Fig. 22b and d, respectively. The DOS analysis of pure CuCr2O4 and BiI3O9 revealed electron excitation at different orbitals. CuCr2O4–BiI3O9-10% NCs outperformed other concentrations and pure CuCr2O4 and BiI3O9 NPs in terms of photocatalytic activity. The efficient photodegradation is attributed to the effective generation of heterojunctions, improved charge carrier separation and migration, and a wide surface area. OH˙ significantly contributed to cefixime (CEF) deterioration. The proposed Z-scheme is effective in photodegrading CEF.45
 | ||
| Fig. 22 Electronic band structure, work function, and atomic arrangement of CuCr2O4 (a and b) and BiI3O9 NPs (c and d).45 | ||
Ghorai et al. studied the photocatalytic performance of a CuCr2O4/BiOBr nanocomposite for degrading RhB and tetracycline hydrochloride (TC-HCl) under household LED light, as shown in Fig. 23.112
 | ||
| Fig. 23 Layout of the dye degradation setup.112 | ||
The BiVO4–CuCr2O4 (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.25) nanocomposite exhibits ∼90% photocatalytic efficiency for MB dye degradation under visible light, which increases with PANI addition. This enhancement is attributed to the improved charge separation of electron–hole pairs, further strengthened by the incorporation of PANI.141
0.25) nanocomposite exhibits ∼90% photocatalytic efficiency for MB dye degradation under visible light, which increases with PANI addition. This enhancement is attributed to the improved charge separation of electron–hole pairs, further strengthened by the incorporation of PANI.141
According to Oladipo et al., 1.0 g CuCr2O4@CaFe-LDO removed 66% COD without an oxidant after 300 min in the dark. However, after 180 min of reaction, the CuCr2O4@CaFe-LDO/K2S2O8/sunlight system removed ∼99% and 98.3% COD and colour, respectively. The crude OMW contained seven phenolic compounds, the most abundant of which were hydroxy tyrosol (76.84%) and tyrosol (15.14%) [Fig. 24].113 Without an oxidant, CuCr2O4@CaFe-LDO degraded phenol by 54.8% after 4 h in the dark. Within 40 min, the deterioration reached 90% when exposed to sunshine and 0.05 M K2S2O8. After 120 min, the CuCr2O4@CaFe-LDO/K2S2O8 system, with UV assistance, achieved 99% phenolic degradation, 95.1% COD removal, and 84.6% BOD removal.114
 | ||
| Fig. 24 Probable reaction pathway of the CuCr2O4@CaFe-LDO/K2S2O8/sunlight system. (b) Illustration of the catalyst performance under both dark and light conditions.113 | ||
It was discovered that the 10% CuCr2O4–CeO2 nanocomposite sample exhibited a remarkable photo-Fenton performance. Tests using coumarin fluorescence probes and radical trapping verified that ˙OH is the main factor causing RhB degradation. Visible LED light promotes the production of ˙OH during the Fenton-like reaction more efficiently than under dark conditions. The favorable redox reaction between Ce4+/Ce3+ and Cu2+/Cu+ couples triggers a dark Fenton-like reaction. However, the low-molecular-weight compounds produced during degradation accumulate on the active sites of the catalyst, reducing its reusability. Fig. 25 illustrates the photo-Fenton reaction mechanisms.115
 | ||
| Fig. 25 Photo Fenton-like reaction process of CuCr2O4–CeO2 NCs.115 | ||
The degradation of MO and MB dyes followed a pseudo-first-order kinetic model.127 Mageshwari et al. analyzed the time-dependent UV-vis absorption spectra of MO and MB in the presence of CuO and CuO–CuCr2O4 (20% Cr3+). After 270 min with CuO and 145 min with CuO–CuCr2O4, the MO absorption peak at 464 nm, which was associated with the azo (–N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N–) bond, disappeared. Likewise, after 360 min with CuO and 180 min with CuO–CuCr2O4, the MB peak at 665 nm disappeared. The time-dependent UV–vis absorption spectra in Fig. 26 demonstrate the effective degradation of MO and MB dyes by the CuO and CuO–CuCr2O4 samples. The dyes gradually lost color, indicating their complete degradation. In the case of MO, the peak at 464 nm disappeared after 270 min with CuO and 145 min with CuO–CuCr2O4. In the case of MB, the 665 nm peak vanished after 360 min with CuO and 180 min with CuO–CuCr2O4, indicating that the CuO–CuCr2O4 sample exhibited superior photocatalytic activity compared to pure CuO.26 The photocatalytic efficacy of g-C3N4-NS/CuCr2O4 NCs was evaluated by degrading MB and RhB dyes and phenol under visible light. The nanocomposite performed best with a 10 wt% CuCr2O4 loading.119
N–) bond, disappeared. Likewise, after 360 min with CuO and 180 min with CuO–CuCr2O4, the MB peak at 665 nm disappeared. The time-dependent UV–vis absorption spectra in Fig. 26 demonstrate the effective degradation of MO and MB dyes by the CuO and CuO–CuCr2O4 samples. The dyes gradually lost color, indicating their complete degradation. In the case of MO, the peak at 464 nm disappeared after 270 min with CuO and 145 min with CuO–CuCr2O4. In the case of MB, the 665 nm peak vanished after 360 min with CuO and 180 min with CuO–CuCr2O4, indicating that the CuO–CuCr2O4 sample exhibited superior photocatalytic activity compared to pure CuO.26 The photocatalytic efficacy of g-C3N4-NS/CuCr2O4 NCs was evaluated by degrading MB and RhB dyes and phenol under visible light. The nanocomposite performed best with a 10 wt% CuCr2O4 loading.119
 | ||
| Fig. 26 Time -dependent UV–vis absorption spectra of (a and b) MO and (c and d) MB dye solutions in the presence of CuO and CuO–CuCr2O4 (20% Cr3+ concentration) samples under UV irradiation.26 | ||
Mohamed et al. investigated CuCr2O4–g-C3N4 photocatalysts for photocatalytic hydrogen generation utilizing glycerol under visible light illumination (Fig. 27). Their findings revealed that the CuCr2O4 content in the heterojunction had a substantial impact on photocatalytic performance, with a 3 wt% composition being the optimal. At this ideal level, the 3 wt% CuCr2O4–g-C3N4 heterojunctions produced thirty-times more hydrogen from glycerol than pure g-C3N4.117
 | ||
| Fig. 27 Proposed mechanism for H2 gas generation using the CuCr2O4–g-C3N4 heterojunction.117 | ||
According to the results, when exposed to simulated sunlight, pure CuCr2O4 or TiO2 showed a weak photocatalytic performance for generating hydrogen from an aqueous oxalic acid solution. However, CuCr2O4/TiO2 heterojunction coupling showed noticeably higher photocatalytic activity. A mass concentration of 0.8 g L−1 exhibited promising hydrogen evolution capability when calcined at 500 °C.144 Baoum et al. investigated the bandgap energy of CuCr2O4 and PANI@CuCr2O4 composites using UV-vis spectroscopy. The bandgap values for pure CuCr2O4 and 0.02%, 0.04%, 0.06%, and 0.08% PANI@CuCr2O4 were 2.12, 2.00, 1.92, 1.73, and 1.72 eV, respectively. The outcomes indicate that the incorporation of PANI reduces the bandgap, thereby enhancing the photocatalytic activity of the nanocomposites.123 Daimalah et al. demonstrated the significance of pH and dose in photocatalytic activity. An increase in catalyst dosage resulted in a higher abatement rate. Furthermore, as the pH decreased from 9 to 3.5, the cefixime (CFX) removal efficiency improved from 30% to 94%.131
Ghorai et al. investigated the photocatalytic activity of dyes and antibiotics, yielding promising results. As demonstrated in Fig. 28, radical trapping experiments revealed that in the PFL reaction, dye abatement decreased significantly with IPA and AgNO3, confirming that ˙OH radical and excited electrons are the main reactive species, respectively. Na2EDTA and BQ had minimal effects, indicating the limited roles of holes and superoxide species, respectively. In the DFL reaction, the degradation activity decreased significantly in the presence of IPA.48
 | ||
| Fig. 28 Impact of various radical quenchers on the PFL and DFL activity of the 100% CuCr2O4/TiO2 nanocomposite for MB, RhB, and MO degradation.48 | ||
Sivaranjani et al. stated that increasing the number of photons per unit area with an increase in light intensity enhances the deterioration. Furthermore, the photocatalytic process exhibits a positive relationship with pH, with the highest degradation occurring at a pH of 7.0. Degradation is reported to diminish above pH 7.0 because of the reaction between the ˙OH and h+ generated by the semiconductor. Compared to other anions such as Cl−, HCO3−, CH3−, and NO3−, the presence of SO42− anions was considered to cause the most deterioration. The bonding behavior of the complexes and the contribution of molecular orbitals (HOMO and LUMO) were predicted by DOS plots.118
As shown in Fig. 29, the alizarine red dye molecule absorbs light energy, exciting electrons in V2O5/CuCr2O4–MCM-41, which generates ROS, such as ˙OH. These ˙OH groups interact with alizarine red molecules, breaking the dye into smaller, non-toxic components such as CO2, SO42−, and H2O.135
 | ||
| Fig. 29 Plausible mechanism for the decomposition of Alizarin Red dye.135 | ||
Ramezanalizadeha et al. highlighted the synergistic involvement of La in improving the photocatalytic activity. Notably, CuCrLaO4 demonstrated remarkable degradation of both RhB and MO dye.100 Hosseini et al. investigated the photocatalytic activity of CuCr2O4 against p-nitrophenol contamination. The CuO content of the samples considerably influenced their photocatalytic performance. The findings demonstrate that the Cu/Cr molar ratio has a significant impact on the photocatalytic efficiency of the samples.136
Al-Wasidi et al. studied the degradation of Acid Orange 7 dye using a CuCr2O4 photocatalyst (Fig. 30). Under visible light, the copper chromite photocatalyst absorbs energy equivalent to or greater than its band gap, resulting in electron–hole (e−/h+) pairs on its surface. The photogenerated electrons combine with adsorbed oxygen molecules to form superoxide radicals (O2˙−), and holes react with water molecules to produce hydroxyl radicals (˙OH). These reactive species, particularly hydroxyl radicals and holes, effectively breakdown Acid Orange 7 dye into CO2, H2O, or other byproducts.81
 | ||
| Fig. 30 Suggested approach, utilizing CuCr2O4 as a photocatalyst for the degradation of Acid Orange 7 dye.81 | ||
Abbasi et al. found that CuCr2O4 and CuCr2O4/Ag nanostructures effectively degraded MO, MB, and RhB dyes. The researchers found that using leucine-capped CuCr2O4/Ag (3%) nanostructures instead of CuCr2O4 NPs improved the RhB dye degradation efficiency from 76% to 91%. Their study also highlighted the role of pH in the breakdown process. As shown in Fig. 31, the surface of the particles under acidic conditions exhibits a significant positive charge, which inhibits cationic dye adsorption due to electrostatic repulsion. The best degrading efficiency for RhB was reached under neutral circumstances. The catalytic efficiency of leucine-capped CuCr2O4/Ag (3%) for RhB degradation was 91% at pH 7 compared to 73% at pH 5.140
 | ||
| Fig. 31 Effect of pH on the RhB adsorption and desorption on the CuCr2O4/Ag nanostructure surfaces.140 | ||
Furthermore, Fig. 32 and Table 6 describe the decomposition of diverse dyes and antibiotics in detail using CuCr2O4 NPs.
 | ||
| Fig. 32 Illustration of the photocatalytic decomposition of various dyes and antibiotics employing pristine CuCr2O4, metal-doped CuCr2O4, and their nanocomposites. | ||
| Dye/antibiotic | Dye conc. | CuCr2O4 NPs loaded | Band gap | Light source | Degradation (%) | Degradation time (h) | Cycles | Rate constant for degradation (min−1) | Ref. |
|---|---|---|---|---|---|---|---|---|---|
| p-Nitrophenol | 20 mg l−1 | 100 mg | — | Visible light | 400 °C-98.4 | 180 | 10 | — | 71 |
| 450 °C-95.7 | |||||||||
| 500 °C-86.8 | |||||||||
| 550 °C-8512 | |||||||||
| 600 °C-77.2 | |||||||||
| MB | 10 mg L−1 | 10 mg | 1.4 | Solar light | 80.6 | 18 | 5 | 0.132 | 23 |
| MO | 92.3 | 18 | 0.147 | ||||||
| RhB | 93.6 | 12 | 0.2095 | ||||||
| RhB | — | 20 mg (600 °C) | — | Visible light | 96.8 | 60 | 5 | — | 22 |
| MB | 99.5 | ||||||||
| MO | 14 | ||||||||
| MG (Mn0.5Cu0.5Cr2O4) | 10 ppm | 40 mg | 1.37 | Solar light | 100 | 40 | 3 | — | 80 |
| Tartrazine azo | — | 0.5 g | 1.25 | Visible light | 99.6 | 120 | 4 | 0.04578 | 64 |
| MB CuCr2O4 | 10 mg L−1 | 0.03 g | 2.4 | Visible light | 98 | 90 | 5 | — | 51 |
| MO capped-CuCr2O4 | 87 capped-CuCr2O4 | 100 | 5 | ||||||
| Sunset yellow | 0.8 g L−1 | 10 ppm | 0.9 | Visible light | 97 | 4 h | 4 | 0.01685 | 52 |
| CEF | — | — | 1.83 CuCr2O4 | Visible light | 97.2 | 200 | 6 | 0.0128 | 45 |
| 2.31 BiI3O9 | |||||||||
| RhB | 10 mg L−1 | 25 mg | 1.48 CuCr2O4 | Led light | 95.97 | 15 | 4 | 0.22427 | 112 |
| TC-HCl | 10 ppm | 25 mg | 2.62 BiOBr | 96.7 | 300 | ||||
| RhB | 10 ppm | 50 mg | 2.81 CuCr2O4 | Visible LED light | 99 | 15 | — | — | 115 |
| MB | 1.40 CeO2 | 85 | |||||||
| MO | 60 | ||||||||
| MB | 2 mg L−1 | 0.2 g | 2.64 | Visible light | 84 | 180 | 3 | 0.0052 | 127 |
| MO | 57 | 0.0025 | |||||||
| MB | 15 mg L−1 | 50 mg | 1.40 CuCr2O4 | UV light | 97.16 | 145 | 0.01803 | 26 | |
| MO | 15 mg L−1 | 1.63 CuO | 98.16 | 180 | 0.02211 | ||||
| RhB | 2.50 × 10−5 M | — | 1.4 CuCr2O4 | Visible light | 98.8 | 210 | 5 | — | 119 |
| 2.7 g-C3N4 | |||||||||
| Acridine orange dye | — | 2.0 g L−1 | 1.73 | Visible light | 100 | 40 | 5 | — | 123 |
| CFX | 10 ppm | 1 g L−1 | 0.93 | Visible light | CuCr2O4-PS-94 | 180 | 4 | — | 131 |
| CuCr2O4-60 (pH-3.5) | |||||||||
| Crystal violet | 15 mg L−1 | 1 mg mL−1 | 1.39-CuCr2O4 | Sun light | — | 90 | 0.012 | — | 124 |
| 3.07-SnO2 | |||||||||
| MB | 10 ppm | 50 mg | 1.4-CuCr2O4 | LED light | 99.2 | 15 | — | 0.30794 | 48 |
| RhB | 3.1-TiO2 | 97.2 | 0.24358 | ||||||
| MO | 89.6 | 0.14943 | |||||||
| TC-HCl | 99 (PFL) | ||||||||
| 58 (DFL) | 15 | ||||||||
| Norfloxacin (NORF) | 97 (PFL) | ||||||||
| 74 (DFL) | 75 | ||||||||
| MG | — | 30 mg | 6.48 | Visible light | 86 | — | — | 0.9855 | 118 |
| AR | — | 40 mg | 1.3 | Visible light | 90 | 60 | — | — | 135 |
| BTB | 90 | 60 | |||||||
| RhB | 10 mg l−1 (pH = 7) | 20 mg | 2.5, 2.6 and 2.65 | LED visible light | 98 | 90 | 6 | — | 100 |
| MO | 99 | 60 | |||||||
| 4-NP | 20 ppm | 25 mg | 1.4 | UV light | 97.2 | 100 | — | 0.0330 | 136 |
| Acid orange 7 | 100 mg L−1 | 0.10 g | CuCr2O4-1.25 | Visible light | 87.38 | 40 min | — | 0.053 | 81 |
| 25 mg L−1 | CuCr2O4–citric acid-1.88 | 96.52 | 0.083 | ||||||
| 25 mg L−1 | CuCr2O4–tartaric acid-1.92 | 98.81 | 0.115 | ||||||
| RhB | 10 ppm | 0.04 g | — | UV light | Leucine–CuCr2O4/Ag (1%)-81 | 60 min | — | — | 140 |
| 3.05 | Leucine–CuCr2O4/Ag (3%)-87 | ||||||||
| — | Leucine–CuCr2O4/Ag (5%)-91% |
5.3. Energy storage application of CuCr2O4 nanomaterials
 | ||
| Fig. 33 Ppy/CuCr2O4 nanocomposite as a pseudocapacitor.47 | ||
Rajeswari et al. examined the photovoltaic efficiency of different materials. Among them, CuCr2O4 had the lowest photovoltaic efficiency, lowest fill factor, and highest short-circuit photocurrent density. In contrast, La-doped oxides had the highest short-circuit photocurrent density and fill factor. The photoelectric efficiency of the La-doped CuCr2O4 electrode was mainly owing to its shape and the synergistic effect of a dispersive active center.83 Bhagwan et al. demonstrated the application of CuCr2O4 NPs in supercapacitors, achieving a specific capacity of 72.5 mAh g−1 at 1 A g−1. Incorporating MWCNTs significantly enhanced the performance, yielding a higher capacity of 108.5 mAh g−1. The MWCNT/CuCr2O4//AC HSC device produced a noteworthy energy density of 32.46 Wh kg−1 and a power density of 775 W kg−1. The tremendous potential of this material for energy storage applications was demonstrated through hands-on demonstrations, which powered gadgets, including LEDs, a kitchen timer, and a toy fan.129 The maximum capacitance of 370.5 F g−1 of the CuCr2O4/GO nanocomposite was attained with a 0.1 M H2SO4 aqueous solution, indicating its potential for capacitive energy storage.120,143
Sarkar et al. found that the 5 wt% CuCr2O4 NG nanocomposite exhibited a maximum specific capacitance of 530.6 F g−1 at a current density of 0.5 A g−1 and demonstrated excellent cycling stability, retaining 98.3% of its capacity after 5000 GCD cycles. CuCr2O4 enhances the supercapacitive performance by serving as a redox-active material and spacer between NG layers, thereby maintaining structural integrity, preventing agglomeration, and minimizing the capacitance loss.121 According to Susanti et al., CV and EIS measurements showed that the hybrid supercapacitor electrode (N–rGO/CuCr2O4) successfully developed a hybrid supercapacitor by combining the characteristics of EDLC and pseudocapacitors.130 Kumar et al. suggested that the NPs exhibited potential as an asymmetric supercapacitor.73
Vinothkumar et al. explored the potential of CuCr2O4/Cr2O3@NF as an efficient material for energy storage. Fig. 34(a) depicts the galvanostatic charge–discharge (GCD) profiles recorded at different current densities (from 1 A g−1 to 15 A g−1), which display noticeable potential plateaus with non-linear features, indicating the pseudocapacitive behavior of the material. As shown in Fig. 34(b), it is evident that the specific capacitance decreases as the current density increases. Despite this trend, the material exhibits a high specific capacitance of approximately 1027.5 F g−1 at 1 A g−1 and maintains a good performance even at higher rates. The comparison chart in Fig. 34(c) highlights the superior electrochemical performance of CuCr2O4/Cr2O3 NCs over other spinel chromite-based materials. Finally, Fig. 34(d) presents the effective redox pseudocapacitance mechanism involving Cu and Cr ion pairs, enhanced by the well-dispersed nanoparticles and high surface area, which facilitates rapid ion diffusion and redox activity. Furthermore, the asymmetric device (CuCr2O4/Cr2O3@NF//AC@NF) delivers an energy density of 38.89 Wh kg−1 at 800.02 W kg−1 and retains 88.67% of its capacitance after 5000 charge–discharge cycles.137
 | ||
| Fig. 34 (a) GCD curves of CuCr2O4/Cr2O3@NF from 1 to 15 A g−1, (b) Cspvs. current density of CuCr2O4/Cr2O3@NF, (c) Csp comparison with earlier Cr-related spinel electrodes, and (d) redox reactions of CuCr2O4/Cr2O3@NF.137 | ||
 | ||
| Fig. 35 Highlighted area in the CV of (a) BiOBr, (b) CuCr2O4, and (c) 20% CuCr2O4/BiOBr in 0.1 M KCl/CH3OH/ferrocene shows the change in potential in each case, with GC serving as the working electrode, Pt wire serving as the counter electrode, and Ag/AgCl serving as the reference electrode.112 | ||
Shafique et al. described the electrochemical stability of a nanoelectrode material, which was tested for commercial uses by scanning modified electrodes for 500 cycles in 1 M KOH and 0.1 M H2SO4 electrolytes at a scan rate of 100 mV s−1. EIS spectra show a quicker electron transfer rate in 0.1 M H2SO4, with a maximum specific capacitance of 445.5 F g−1 determined using CV.122 Furthermore, a three-electrode setup using CuCr2O4–CTAB as the electrode material produced capacitance values of up to 965 F g−1 at a scan rate of 1 A g−1. Additionally, it provides excellent cyclic stability in a 1 M Na2SO4 electrolyte, retaining 91% of its initial capacitance after 5000 cycles.73
Using a simple drop-coating method, Kong et al. successfully fabricated a non-enzymatic electrochemical H2O2 sensor. The broad apparent active area of the CuCr2O4/GCE probe, low charge transfer resistance, and the synergistic redox coupling of CuCr2O4 and GCE were responsible for its high sensitivity (886 μA mM−1 cm−2), low detection limit (160 nM), and wide linear detection range (0.3 μM to 8.1 mM) in 0.1 M NaOH.60 Lao et al. reported the electrochemical performance of a CuCr2O4Ag composite. Compared to the uncoated CuCr2O4, the Ag coating on CuCr2O4 improves its coulombic efficiency, cycling stability, and initial discharge capacity, making it a more efficient anode material for lithium-ion batteries.46
5.4. Biological activities of CuCr2O4 nanomaterials/composite
In the study by Ahmed et al., they examined the toxicity of CuCr2O4 NPs in MCF-7 cells using the MTT test. The rosemary essential nanoemulsion helps prevent cancer. This study found that rosemary-mediated CuCr2O4 NPs had a stronger cell-killing effect than Nolvadex-D. The IC50 value of the synthesized NPs was 247.7 ppm. Furthermore, a hemolysis assay was performed on red blood cells (RBCs) using different concentrations of the compound. The results revealed no hemolysis at any tested concentration, suggesting that the NPs are non-toxic. This confirms the absence of cytotoxic effects on RBC in the toxicity screening test.41 Mirzaei et al. tested the antibacterial activities of bare or modified NPs against Gram-positive (S. aureus) and Gram-negative (E. coli) bacteria in silicone rubber and PVDF matrices. They discovered that the size of the NPs and the matrix medium are critical for inhibiting bacterial growth.42 Khan et al. studied antibacterial activities achieved through the photoinhibition of both E. coli and S. aureus bacteria. V2O5/CuCr2O4/MCM-41 inhibited E. coli and S. aureus in a zone of 18 ± 0.3 mm and 22 ± 0.2 mm, respectively, when exposed to visible light. The nanostructures efficiently inhibited bacterial growth and survival, demonstrating their suitability for applications in water disinfection and V2O5/CuCr2O4/MCM-41 antibacterial coatings.135 Heydari et al. evaluated the bone-forming ability of human adipose-derived stem cells (hADSCs) using a nanocomposite scaffold synthesized from glycerol and azelaic acid (Gl–Az) with CuCr2O4 NPs. The findings demonstrated that the Gl–Az–1%CuCr2O4 scaffold considerably raised bone differentiation markers in hADSCs after 14 days and exhibited good mechanical strength, biocompatibility, and degradability.44 CuCr2O4–BiI3O9 NCs were tested for their potential genotoxicity on Allium cepa root tips. The mitotic cycle and nuclei index impact the genotoxic nature of NCs and phase disruption. Modifying the stages of the cycle impairs protein synthesis and DNA replication, thereby enhancing the cytotoxicity. Furthermore, the NCs showed no chromosomal aberrations or micronucleus production in Allium cepa, indicating their non-genotoxic nature.45 Moreover, Tables 7 and 8 describe the biological and toxicological activities of CuCr2O4 nanomaterials/composites.| Nanomaterials | Applications | Cell line/bacteria | Results | Mechanism of action | Observations | Ref. |
|---|---|---|---|---|---|---|
| Rosemary mediated-CuCr2O4 NPs | Anti-breast cancer (MTT assay) | MCF-7 | IC50-247.7 ppm | Generation of reactive oxygen species (ROS), induced oxidative stress, mitochondrial damage | Dose-dependent cell death: up to 64% reduction at 320 ppm after 24 h; apoptosis observed | 41 |
| Modified CuCr2O4/silicone rubber (soaked in Tween80) | Antibacterial activity (Agar diffusion method) | E. coli | ZOI | Damage cell membrane | Size and medium are crucial factors in preventing bacterial development because a higher surface area-to-volume ratio promotes better interactions | 42 |
| S. aureus | 11 mm | |||||
| 12 mm | ||||||
| Bare CuCr2O4/silicone rubber (soaked in Tween80) | E. coli | 12 mm | ||||
| S. aureus | 14 mm | |||||
| V2O5/CuCr2O4/MCM-41 (light) | Antibacterial activity (Agar well diffusion method) | E. coli | ZOI | Disruption of the cell membrane, and generation of ROS | Blocked bacterial growth and viability | 135 |
| 18 ± 0.3 mm | ||||||
| MIC | ||||||
| 50 μg mL−1 | ||||||
| S. aureus | ZOI | |||||
| 22 ± 0.2 mm | ||||||
| MIC | ||||||
| 40 μg mL−1 |
| Applications | Effective dose | Mechanism of action | Observations | Ref. |
|---|---|---|---|---|
| Homolysis (blood cells) | 50, 250, and 500 μg mL−1 | Disruption of cell membranes | Non-toxic nature for all concentration (no homolysis) | 41 |
| Genotoxicity (Allium cepa root tips) | — | DNA damage, chromosomal aberrations | Dependent on the NCs concentration | 45 |
5.5. Miscellaneous applications of CuCr2O4 nanomaterials/composite
Pakzad et al. investigated spectrally selective absorbers in thermoset acrylate-based coatings on aluminium substrates. The larger particle sizes of the produced CuCr2O4 NPs exhibit superior solar selectivity. This results in lower thermal emittance (εT) in the infrared region (2.5–20 μm) and higher solar absorption (αs) across the solar spectrum (0.3–2.5 μm).17 The η of DSSC was 44% higher than that of the pure TiO2 film electrode. CuCr2O4/TiO2 composite electrodes can reduce charge recombination by slowing down reverse electron transfer due to their insulating properties. Furthermore, spinel CuCr2O4 is a narrow-bandgap semiconductor with a gap energy of 1.14 eV, which facilitates the absorption of solar energy in the visible light range, thereby improving the performance of DSSCs.72 Hu et al. described the doping of MCr2O4 into TiO2 film electrodes, which boosted their absorbance. CuCr2O4/TiO2 showed superior light responsiveness than NiCr2O4/TiO2. 2% CuCr2O4 increased the photoelectric conversion efficiency by 54%, whereas 1% NiCr2O4 improved it by 30%.125 Rubin and colleagues investigated solar absorber coatings. The maximum solar absorptance was observed in the porous Cu0.5Cr1.1Mn1.4O4 (Fig. 36), which was 97.1% prior to thermal annealing and remained unchanged during thermal testing, reaching 97.2% after 2000 h. Alternatively, the solar absorptance of Pyromark 2500 significantly decreased, increased from 96.4% to 94.6%.14 | ||
| Fig. 36 Dotted line displaying the anticipated trend for solar absorptance value at 800 °C after 100, 300, 1000, and 2000 h.14 | ||
Javed et al. explored the temperature and frequency effect of the electrical conduction and dielectric relaxation mechanisms in CuCr2O4, tetragonally distorted spinel chromite.59 The exchange bias behavior was discussed about the exchange coupling between the ferrimagnetic CuCr2O4 (130 K) and the antiferromagnetic Cr2O3 (310 K). The exchange bias in CuCr2O4–Cr2O3 was attributed to the exchange coupling between ferrimagnetic CuCr2O4 (130 K) and antiferromagnetic Cr2O3 (310 K). Strong exchange bias was noticed in the field-cooled hysteresis loop at low temperatures, with both the exchange bias field and coercive field decreasing as the temperature increased, vanishing at around 200 K.126
6. Challenges and opportunities
Despite significant advancements, several challenges remain in the synthesis and application of CuCr2O4 NPs. Achieving precise control over particle size, shape, and crystallinity is challenging, given that the synthesis parameters such as temperature, pH, reaction duration, and surfactants significantly influence the final properties. Scalability and cost-effectiveness also pose hurdles, given that many synthesis methods require high energy inputs or expensive precursors, limiting their commercial viability. Furthermore, to ensure a consistent performance, additional research is also required to determine the stability and reproducibility of CuCr2O4 NPs under various environmental conditions. Concerns regarding potential cytotoxicity and environmental impact must be addressed before the widespread use of biomedical and industrial applications. According to the literature, CuCr2O4 nanocomposites and metal-doped CuCr2O4 NPs have demonstrated a remarkable capacity for energy storage and photocatalytic activity. Additional research is required, especially in the area of green synthesis. Only two studies have reported the green production of CuCr2O4 NPs from natural sources to date. This opens up a new avenue for research on sustainability. Additionally, creative approaches are necessary to enhance the compatibility and efficiency when integrating these NPs into practical devices, such as sensors, batteries, and catalysts.However, numerous opportunities exist to overcome these challenges and expand the potential of CuCr2O4-based materials. Advanced functionalization through metal doping and nanocomposite formation can significantly enhance their catalytic, electrochemical, and biomedical properties. The development of sustainable and green synthesis techniques that use less energy and environmentally benign precursors can improve the scalability, while lessening their negative effects on the environment. CuCr2O4 NPs have enormous potential for anticancer treatments, antimicrobial coatings, and targeted drug delivery in biomedical applications. Their excellent electrochemical properties also make them ideal candidates for next-generation energy storage devices, including supercapacitors and Li-ion batteries. Furthermore, their integration into smart sensor technologies might result in extremely sensitive and specific detection systems for industrial applications, healthcare diagnostics, and environmental monitoring. By addressing existing challenges and leveraging these opportunities, CuCr2O4 NPs can be further optimized for cutting-edge applications, driving innovation across multiple scientific and technological domains.
7. Conclusion
In this review paper, we provide an overview of breakthroughs in CuCr2O4 nanostructures, including pristine NPs, metal-doped variants, and nanocomposites, with diverse applications spanning biological, catalytic, electrochemical, and sensing fields. Several procedures exist for the synthesis of CuCr2O4 nanomaterials, including sol–gel, hydrothermal, solution combustion, co-precipitation, and electrospinning approaches. The fabrication of these NPs is significantly influenced by variables such as temperature, pH, reaction time, precursor concentration, and the selection fuel or surfactant, which directly impact their structural and functional properties. In this review, we demonstrated that CuCr2O4 NPs demonstrate significant catalytic activity in organic transformations. They also serve as an excellent enhancer of the burning rate for AP-based solid propellant systems. Furthermore, they have demonstrated outstanding potential in energy storage devices such as Li-ion batteries and supercapacitors, as well as electrocatalysis for the OER and HER. CuCr2O4 NPs show promise in nanomedicine, including anticancer, antibacterial, and genotoxic properties. Their photocatalytic activity has also been widely investigated for the decomposition of dyes and antibiotics. CuCr2O4 NPs have great promise in these fields due to their unique physical and magnetic features. Future studies should focus on developing sustainable and scalable synthesis techniques, while exploring novel applications of CuCr2O4-based nanomaterials. By bridging the gap between synthesis and application, this review aims to contribute to the advancement of copper chromite nanotechnology for next-generation scientific and industrial innovations.Conflicts of interest
The authors declare no conflict of interest.Data availability
No primary research results, software or code have been included and no new data were generated or analysed as part of this review.References
- J. Hulla, S. C. Sahu and A. W. Hayes, Nanotechnology: History and future, Hum. Exp. Toxicol., 2015, 34(12), 1318–1321 CrossRef CAS PubMed.
- S. Ghotekar, et al., Spinel ZnCr2O4 nanorods synthesized by facile sol-gel auto combustion method with biomedical properties, J. Sol-Gel Sci. Technol., 2023, 105(1), 176–185 CrossRef CAS PubMed.
- K. T. Thendral, M. Amutha and R. Ragunathan, Design and development of copper cobaltite (CuCo2O4) nanoparticle for antibacterial anticancer and photocatalytic activity, Mater. Lett., 2023, 349, 134720 CrossRef.
- R. Kalia, et al., Photocatalytic degradation properties of Li–Cr ions substituted CoFe2O4 nanoparticles for wastewater treatment application, Phys. Status solidi A, 2022, 219(8), 2100539 CrossRef CAS.
- Y. Gao, et al., Spinel CoMn2O4 hollow nanospheres for very wide linear and sensitive detection of hydrogen peroxide, J. Alloys Compd., 2022, 897, 163158 CrossRef CAS.
- H. Adkins and R. Connor, The catalytic hydrogenation of organic compounds over copper chromite, J. Am. Chem. Soc., 1931, 53(3), 1091–1095 CrossRef CAS.
- A. M. Kawamoto, L. C. Pardini and L. C. Rezende, Synthesis of copper chromite catalyst, Aerosp. Sci. Technol., 2004, 8(7), 591–598 CrossRef CAS.
- P. Sathiskumar, C. Thomas and G. Madras, Solution combustion synthesis of nanosized copper chromite and its use as a burn rate modifier in solid propellants, Ind. Eng. Chem. Res., 2012, 51(30), 10108–10116 CrossRef CAS.
- S. Ghotekar, et al., A novel green synthesis of honey-mediated CoCr2O4 nanoparticles and their expeditious heterogeneous catalytic role for the synthesis of 5-aryl-[1, 2, 4] triazolidine-3-thiones, Res. Chem. Intermed., 2024, 50(1), 49–68 CrossRef CAS.
- A. S. Namini, et al., Synthesis and characterization of novel ZnO/NiCr2O4 nanocomposite for water purification by degradation of tetracycline and phenol under visible light irradiation, Mater. Res. Bull., 2021, 139, 111247 CrossRef CAS.
- B. Sriram, et al., Deep eutectic solvent-mediated synthesis of spinel zinc chromite nanoparticles: a simple label-free electrochemical sensor for dopamine and ascorbic acid, ACS Appl. Nano Mater., 2023, 6(19), 17593–17602 CrossRef CAS.
- O. Crottaz, F. Kubel and H. Schmid, Jumping crystals of the spinels NiCr2O4 andCuCr2O4, J. Mater. Chem., 1997, 7(1), 143–146 RSC.
- L. Patron, et al., New synthetic route in obtaining copper chromite I. Hydrolysis of some soluble salts, Mater. Res. Bull., 2001, 36(7–8), 1269–1276 CrossRef CAS.
- E. B. Rubin, Y. Chen and R. Chen, Optical properties and thermal stability of Cu spinel oxide nanoparticle solar absorber coatings, Sol. Energy Mater. Sol. Cells, 2019, 195, 81–88 CrossRef CAS.
- S. S. Acharyya, et al., Cetyl alcohol mediated synthesis of CuCr2O4 spinel nanoparticles: a green catalyst for selective oxidation of aromatic C–H bonds with hydrogen peroxide, RSC Adv., 2015, 5(7), 4838–4843 RSC.
- J. Safaei-Ghomi, Z. Akbarzadeh and B. Khojastehbakht-Koopaei, C–N cross-coupling reaction catalysed by reusable CuCr2O4 nanoparticles under ligand-free conditions: a highly efficient synthesis of triarylamines, RSC Adv., 2015, 5(37), 28879–28884 RSC.
- E. Pakzad, Z. Ranjbar and M. Ghahari, Synthesized of octahedral cupper chromite spinel for spectrally selective absorber (SSA) coatings, Prog. Org. Coat., 2019, 132, 21–28 CrossRef CAS.
- Monika, R. Sharma and A. Singh, Structural and optical properties of chromium oxide in CuCr2O4−δ nanostructured materials, Mater. Today: Proc., 2023, 1–5 CAS.
- B. Gou, et al., Effect of Nano–Copper Chromite on the Thermal Decomposition and Combustion of AP–Based Solid Propellants, Propellants, Explos., Pyrotech., 2022, 47(10), e202200087 CrossRef CAS.
- X. Sun, et al., A CTAB-assisted hydrothermal orientation growth of ZnO nanorods, Mater. Chem. Phys., 2003, 78(1), 99–104 CrossRef.
- R. Mohamed, Synthesis and characterization of AgCl@ graphitic carbon nitride hybrid materials for the photocatalytic degradation of atrazine, Ceram. Int., 2015, 41(1), 1197–1204 CrossRef CAS.
- W. Yuan, X. Liu and L. Li, Synthesis, characterization and photocatalytic activity of cubic-like CuCr2O4 for dye degradation under visible light irradiation, Appl. Surf. Sci., 2014, 319, 350–357 CrossRef CAS.
- B. Paul, et al., Facile synthesis of spinel CuCr2O4 nanoparticles and studies of their photocatalytic activity in degradation of some selected organic dyes, J. Alloys Compd., 2015, 648, 629–635 CrossRef CAS.
- S. S. Acharyya, et al., Synergistic effect between ultrasmall Cu(II) oxide and CuCr2O4 spinel nanoparticles in selective hydroxylation of benzene to phenol with air as oxidant, ACS Catal., 2015, 5(5), 2850–2858 CrossRef CAS.
- A. Sanoop, R. Rajeev and B. K. George, Synthesis and characterization of a novel copper chromite catalyst for the thermal decomposition of ammonium perchlorate, Thermochim. Acta, 2015, 606, 34–40 CrossRef CAS.
- K. Mageshwari, et al., Novel CuCr2O4 embedded CuO nanocomposites for efficient photodegradation of organic dyes, Appl. Surf. Sci., 2015, 353, 95–102 CrossRef CAS.
- M. H. Ghasemi and E. Kowsari, The synergistic effect of copper chromite spinel nanoparticles (CuCr 2 O 4) and basic ionic liquid on the synthesis of cyclopropanecarboxylic acids, Res. Chem. Intermed., 2016, 42, 7963–7975 CrossRef CAS.
- W. Zhang, et al., Progress of ethylene action mechanism and its application on plant type formation in crops, Saudi J. Biol. Sci., 2020, 27(6), 1667–1673 CrossRef CAS PubMed.
- X. Zenggang, et al., Research on image retrieval algorithm based on combination of color and shape features, J. Signal Process. Syst., 2021, 93, 139–146 CrossRef.
- C. Zuo, et al., Transport of intensity phase retrieval and computational imaging for partially coherent fields: The phase space perspective, Opt. Laser Eng., 2015, 71, 20–32 CrossRef.
- C. Zuo, et al., High-speed three-dimensional shape measurement for dynamic scenes using bi-frequency tripolar pulse-width-modulation fringe projection, Opt. Laser Eng., 2013, 51(8), 953–960 CrossRef.
- X. Li, et al., Silver–catalyzed decarboxylative allylation of difluoroarylacetic acids with allyl sulfones in water, Chem. – Asian J., 2020, 15(7), 1175–1179 CrossRef CAS PubMed.
- Y. Liu, et al., Hierarchical nanocomposite electrocatalyst of bimetallic zeolitic imidazolate framework and MoS2 sheets for non-Pt methanol oxidation and water splitting, Appl. Catal., B, 2019, 258, 117970 CrossRef CAS.
- P. Li, et al., A novel hydrolysis method to synthesize chromium hydroxide nanoparticles and its catalytic effect in the thermal decomposition of ammonium perchlorate, Thermochim. Acta, 2012, 544, 71–76 CrossRef CAS.
- S. S. Acharyya, S. Ghosh and R. Bal, Catalytic oxidation of aniline to azoxybenzene over CuCr2O4 spinel nanoparticle catalyst, ACS Sustainable Chem. Eng., 2014, 2(4), 584–589 CrossRef CAS.
- B. M. Latha, V. Sadasivam and B. Sivasankar, A highly selective synthesis of pyrazine from ethylenediamine on copper oxide/copper chromite catalysts, Catal. Commun., 2007, 8(7), 1070–1073 CrossRef CAS.
- D. Liu, et al., Deactivation mechanistic studies of copper chromite catalyst for selective hydrogenation of 2-furfuraldehyde, J. Catal., 2013, 299, 336–345 CrossRef CAS.
- Z. Karimi-Jaberi, et al., Nano-copper chromite (nano-CuCr2O4): a novel and efficient catalyst for the synthesis of biscoumarin and pyrano [c] chromene derivatives in water at room temperature, Res. Chem. Intermed., 2016, 42, 4641–4650 CrossRef CAS.
- S. S. Acharyya, et al., Preparation of the CuCr2O4 spinel nanoparticles catalyst for selective oxidation of toluene to benzaldehyde, Green Chem., 2014, 16(5), 2500–2508 RSC.
- S. Abdolmohammadi and S. Dahi, Azar, Sustainable synthesis of [1] benzopyran azo dyes using CuCr2O4 NPs., J. Heterocycl. Chem., 2021, 58(11), 2181–2188 CrossRef CAS.
- H. S. Ahmed, M. Hammadi and W. Majeed, Green Synthesis of CuCr2O4 Nano Composite from Rosemary Extract and Evaluation of its Anti-Breast Cancer Properties, J. Nanostruct., 2024, 14(4), 1183–1190 CAS.
- A. Mirzaei, R. Peymanfar and N. Khodamoradipoor, Investigation of size and medium effects on antimicrobial properties by CuCr2O4 nanoparticles and silicone rubber or PVDF, Mater. Res. Express, 2019, 6(8), 085412 CrossRef CAS.
- P. Sankudevan, et al., Enhancement of luminescence mechanisms in structural, morphological, and catalytic properties of undoped CuCr2O4 and Mn-doped CuCr2O4, J. Cluster Sci., 2023, 34(3), 1527–1534 CrossRef CAS.
- M. Heydari, et al., The role of copper chromite nanoparticles on physical and bio properties of scaffolds based on poly (glycerol-azelaic acid) for application in tissue engineering fields, Cell Tissue Res., 2023, 391(2), 357–373 CrossRef CAS PubMed.
- S. Balasurya, et al., Novel self-propelled CuCr2O4-BiI3O9 nanojets for the efficient photodegradation of organics in wastewater: A non-genotoxic nanomaterial for water treatment, J. Water Process Eng., 2023, 53, 103657 CrossRef.
- M. Lao, et al., Enhanced electrochemical performance of Ag-coated CuCr2O4 as anode material for lithium-ion batteries, Ceram. Int., 2014, 40(8), 11899–11904 CrossRef CAS.
- J. S. Shayeh, et al., A novel route for electrosynthesis of CuCr2O4 nanocomposite with p-type conductive polymer as a high performance material for electrochemical supercapacitors, J. Colloid Interface Sci., 2017, 496, 401–406 CrossRef CAS PubMed.
- K. Ghorai, et al., Anatase TiO2 decorated CuCr2O4 nanocomposite: A versatile photocatalyst under domestic LED light irradiation, Appl. Surf. Sci., 2021, 568, 150838 CrossRef CAS.
- S. A. Lachini, A. Eslami and M. Enhessari, A comparative study of sol-gel and green synthesized CuCr2O4 nanoparticles as an electrode material for enhanced electrochemical hydrogen storage, Int. J. Hydrogen Energy, 2024, 88, 841–849 CrossRef CAS.
- S. G. Hosseini, R. Abazari and A. Gavi, Pure CuCr2O4 nanoparticles: synthesis, characterization and their morphological and size effects on the catalytic thermal decomposition of ammonium perchlorate, Solid State Sci., 2014, 37, 72–79 CrossRef CAS.
- R. Peymanfar and H. Ramezanalizadeh, Sol-gel assisted synthesis of CuCr2O4 nanoparticles: an efficient visible-light driven photocatalyst for the degradation of water pollutions, Optik, 2018, 169, 424–431 CrossRef CAS.
- Y. Benrighi, et al., Characterization and application of the spinel CuCr2O4 synthesized by sol–gel method for sunset yellow photodegradation, J. Sol-Gel Sci. Technol., 2022, 101(2), 390–400 CrossRef CAS.
- R. Peymanfar and N. Khodamoradipoor, Preparation and Identification of CuCr2O4 Nanoparticles and Investigation of Its Microwave Absorption Characteristics at X-Band Frequency Using Silicone Rubber Polymeric Matrix, Proceedings, 2018, 1–6 Search PubMed.
- R. Peymanfar and N. Khodamoradipoor, Preparation and characterization of copper chromium oxide nanoparticles using modified sol–gel route and evaluation of their microwave absorption properties, Phys. Status solidi A, 2019, 216(11), 1900057 CrossRef.
- M. H. Habibi and F. Fakhri, Fabrication and Characterization of CuCr2O4 Nanocomposite by XRD, FESEM, FTIR, and DRS, Synth. React. Inorg., Met.-Org., Nano-Met. Chem., 2016, 46(6), 847–851 CrossRef CAS.
- Y. Wang, et al., Nanochromates MCr2O4 (M = Co, Ni, Cu, Zn): preparation, characterization, and catalytic activity on the thermal decomposition of fine AP and CL-20, ACS Omega, 2019, 5(1), 327–333 CrossRef PubMed.
- W. Li and H. Cheng, Synthesis and characterization of Cu-Cr-O nanocomposites, J. Cent. South Univ. Technol., 2007, 14(3), 291–295 CrossRef CAS.
- J. Billman, et al., CuCr2O4 particle growth and evolution across sol–gel routes and calcination profiles, Adv. Appl. Ceram., 2023, 122(3–4), 197–214 CrossRef CAS.
- M. Javed, et al., Temperature and frequency dependent response of electrical conduction and dielectric relaxation mechanism in CuCr2O4 tetragonally distorted spinel chromite, Ceram. Int., 2024, 50(3), 4767–4781 CrossRef CAS.
- D.-R. Kong, et al., Non-enzymatic CuCr2O4/GCE amperometric sensor for high sensing and rapid detection of nM level H2O2, Microchem. J., 2023, 194, 109343 CrossRef CAS.
- H. Cui, M. Zayat and D. Levy, Sol-gel synthesis of nanoscaled spinels using propylene oxide as a gelation agent, J. Sol-Gel Sci. Technol., 2005, 35, 175–181 CrossRef CAS.
- M. H. Habibi and F. Fakhri, Sol–gel combustion synthesis and characterization of nanostructure copper chromite spinel, J. Therm. Anal. Calorim., 2014, 115, 1329–1333 CrossRef CAS.
- B. S. Barros, et al., Preparation and characterization of spinel MCr2O4 (M = Zn, Co, Cu and Ni) by combustion reaction, J. Metastable Nanocryst. Mater., 2004, 20, 325–332 Search PubMed.
- P. de Jesus Cubas, et al., Synthesis of CuCr2O4 by self-combustion method and photocatalytic activity in the degradation of Azo Dye with visible light, J. Photochem. Photobiol., A, 2020, 401, 112797 CrossRef.
- J. V. Viswanath, et al., Copper chromite as ballistic modifier in a typical solid rocket propellant composition: A novel synthetic route involved, J. Energ. Mater., 2018, 36(1), 69–81 CrossRef.
- P. Tamizhdurai, et al., Renewable Energy from Waste Plastic: Hydroprocessing of Polypropylene Pyrolysis Oil to Diesel Fuel Using Novel Catalyst. SSRN, 2024, 1–29 Search PubMed.
- J. Safaei-Ghomi, B. Khojastehbakht-Koopaei and S. Zahedi, Copper chromite nanoparticles as an efficient and recyclable catalyst for facile synthesis of 4, 4′-(arylmethanediyl) bis (3-methyl-1 H-pyrazol-5-ol) derivatives, Chem. Heterocycl. Compd., 2015, 51, 34–38 CrossRef CAS.
- H. Shahbazi-Alavi, J. Safaei-Ghomi and S. H. Nazemzadeh, CuCr2O4 nanoparticles: An efficient heterogeneous catalyst for the synthesis of bis-thiazolidinones, Org. Chem. Res., 2018, 4(1), 116–123 Search PubMed.
- M. Edrissi, S. Hosseini and M. Soleymani, Synthesis and characterisation of copper chromite nanoparticles using coprecipitation method, Micro Nano Lett., 2011, 6(10), 836–839 CrossRef CAS.
- H. Tavakoli, M. R. Sarraf and A. Zarei, Inverse Co-precipitation Synthesis of Copper Chromite Nanoparticles, 2016 Search PubMed.
- L. Pan, et al., Highly photocatalytic activity for p-nitrophenol degradation with spinel-structured CuCr2O4, Micro Nano Lett., 2012, 7(5), 415–418 CrossRef.
- Z. Q. Hu, et al., Preparation and photoelectric properties of CuCr2O4 nanopowders, Adv. Mater. Res., 2011, 284, 974–979 Search PubMed.
- S. A. Kumar, T. Shahanas and G. Harichandran, Synthesis and fabrication of tetragonal copper chromite nanospheres for usage as the cathode material in asymmetric supercapacitors, J. Energy Storage, 2025, 106, 114839 CrossRef.
- S. S. Acharyya, et al., Facile synthesis of CuCr2O4 spinel nanoparticles: a recyclable heterogeneous catalyst for the one pot hydroxylation of benzene, Catal. Sci. Technol., 2014, 4(12), 4232–4241 RSC.
- S. S. Acharyya, S. Ghosh and R. Bal, Surfactant promoted synthesis of CuCr2O4 spinel nanoparticles: a recyclable catalyst for one-pot synthesis of acetophenone from ethylbenzene, Ind. Eng. Chem. Res., 2014, 53(51), 20056–20063 CrossRef CAS.
- S. S. Acharyya, et al., Fabrication of CuCr2O4 spinel nanoparticles: A potential catalyst for the selective oxidation of cycloalkanes via activation of Csp3–H bond, Catal. Commun., 2015, 59, 145–150 CrossRef CAS.
- S. Mobini, F. Meshkani and M. Rezaei, Surfactant-assisted hydrothermal synthesis of CuCr2O4 spinel catalyst and its application in CO oxidation process, J. Environ. Chem. Eng., 2017, 5(5), 4906–4916 CrossRef CAS.
- S. S. Acharyya, S. Ghosh and R. Bal, Fabrication of three-dimensional (3D) raspberry-like copper chromite spinel catalyst in a facile hydrothermal route and its activity in selective hydroxylation of benzene to phenol, ACS Appl. Mater. Interfaces, 2014, 6(16), 14451–14459 CrossRef CAS PubMed.
- S. Ismael, et al., Facile Synthesis and Catalytic Activity Assessment of Copper Chromite Nanoparticles for Ammonium Perchlorate Decomposition, J. Phys.: Conf. Ser., 2022, 1–11 Search PubMed.
- F. Soleimani, M. Salehi and A. Gholizadeh, Synthesis and characterization of new spinel Mn0.5Cu0.5Cr2O4 and degradation of Malachite Green from wastewater in comparison with CuCr2O4, Int. J. Nano Dimens., 2019, 10(3), 260–271 CAS.
- A. S. Al-Wasidi, et al., Facile hydrothermal synthesis of copper chromite nanoparticles for efficient photocatalytic degradation of acid orange 7 dye, J. Inorg. Organomet. Polym. Mater., 2022, 1–12 Search PubMed.
- N. Bisht, et al., Novel microwave synthesis of copper chromite nanoparticles, Inorg. Chem. Commun., 2021, 134, 109072 CrossRef CAS.
- G. Rajeswari, et al., Enhancement of the structure, solar cells and vibrational studies of undoped CuCr2O4 and La-doped CuCr2O4 semiconductor compounds, Heliyon, 2022, 8(4), 1–6 CrossRef PubMed.
- F. Chen, et al., Comparative Study of Typical Copper-Based Catalysts for Catalyzing Thermal Decomposition of High Energy Oxidant Ammonium Dinitramide. Available at SSRN 4903523.
- D. Zhang, et al., Significantly enhanced thermal decomposition of mechanically activated ammonium perchlorate coupling with nano copper chromite, ACS Omega, 2021, 6(24), 16110–16118 CrossRef CAS PubMed.
- M. Han, et al., Spinel CuB2O4 (B = Fe, Cr, and Al) oxides for selective adsorption of Congo red and photocatalytic removal of antibiotics, ACS Appl. Nano Mater., 2022, 5(8), 11194–11207 CrossRef CAS.
- S. Appalakutti, et al., Process intensification of copper chromite (CuCr2O4) nanoparticle production using continuous flow microreactor, Chem. Eng. Process., 2015, 89, 28–34 CrossRef CAS.
- T.-W. Chiu, C.-H. Tu and Y.-T. Chen, Fabrication of electrospun CuCr2O4 fibers, Ceram. Int., 2015, 41, S399–S406 CrossRef CAS.
- D.-C. Tsai, et al., Annealing effect on the structural and optoelectronic properties of Cu-Cr-O thin films deposited by reactive magnetron sputtering using a single CuCr target, Mater. Sci.-Pol., 2023, 41(1), 191–201 CrossRef.
- G.-z. Hao, et al., Preparation and characterization of copper-based nanoparticles as catalyst for ammonium perchlorate (AP), in 2015 IEEE 15th International Conference on Nanotechnology (IEEE-NANO), IEEE, 2015.
- J. Schorne-Pinto, et al., Thermodynamic and structural properties of CuCrO2 and CuCr2O4: experimental investigation and phase equilibria modeling of the Cu–Cr–O system, J. Phys. Chem. C, 2021, 125(27), 15069–15084 CrossRef CAS.
- A. Wołosiak-Hnat, E. Milchert and G. Lewandowski, The influence of technological parameters on hydrogenolysis of glycerol in the presence of CuCr2O4 catalyst, J. Adv. Oxid. Technol., 2012, 15(2), 405–417 Search PubMed.
- A. Ashraf, et al., The potentials of copper chromite nanoparticles on ammonium nitrate decomposition: Towards eco-friendly oxidizers for green solid propellants, J. Phys.: Conf. Ser., 2024, 1–9 Search PubMed.
- K. Batool, et al., Facile chemical synthesis of cadmium and neodymium doped copper chromite by sol gel method, J. Nanoscope, 2021, 2(2), 163–184 CrossRef.
- T. Anusree and A. A. Vargeese, Enhanced performance of barium and cobalt doped spinel CuCr2O4 as decomposition catalyst for ammonium perchlorate, J. Solid State Chem., 2022, 315, 123481 CrossRef CAS.
- S. S. Acharyya, S. Ghosh and R. Bal, Direct catalytic oxyamination of benzene to aniline over Cu(II) nanoclusters supported on CuCr2O4 spinel nanoparticles via simultaneous activation of C–H and N–H bonds, Chem. Commun., 2014, 50(87), 13311–13314 RSC.
- S. S. Acharyyaa, S. Ghosh, S. Adak, R. Singh, V. Kumar and R. Bal, Highly efficient Cu2+/CuCr2O4 nanoparticles catalyst for selective oxidation of ethanol, Sci. Lett., 2015, 4, 153 Search PubMed.
- M. Bai, et al., Synthesis and characterization of Fe/Mn co-doped CuCr2O4 black pigment with high near-infrared reflectance, Sol. Energy, 2022, 234, 240–250 CrossRef CAS.
- K. Yadagiri, et al., Structural transformation and magnetic properties of Fe-substituted nano CuCr2O4 spinel structure, Ceram. Int., 2024, 50(3), 4987–4993 CrossRef.
- H. Ramezanalizadeh, R. Peymanfar and N. Khodamoradipoor, Design and development of a novel lanthanum inserted CuCr2O4 nanoparticles photocatalyst for the efficient removal of water pollutions, Optik, 2019, 180, 113–124 CrossRef CAS.
- S. Jagadeesan, et al., Liquid phase selective oxidation of veratryl alcohol to veratraldehyde using pure and Mg-doped copper chromite catalysts, RSC Adv., 2024, 14(25), 18093–18102 RSC.
- K. Batool, et al., Synthesis and characterization of Zinc-Doped Copper Chromites by sol gel method, J. Nanoscope, 2021, 2(1), 15–28 CrossRef.
- Y. Youn, et al., Effects of metal dopings on CuCr2O4 pigment for use in concentrated solar power solar selective coatings, ACS Appl. Energy Mater., 2019, 2(1), 882–888 CrossRef CAS.
- A. T. Apostolov, I. N. Apostolova and J. M. Wesselinova, Magnetic, electric and optical properties of ion doped CuCr2O4 nanoparticles, Magnetochemistry, 2022, 8(10), 122 CrossRef CAS.
- K. Batool, et al., Nanosized magnesium doped copper chromites spinel particles synthesis and characterization, ECS J. Solid State Sci. Technol., 2020, 9(12), 126005 CrossRef CAS.
- S. Khostavan, et al., The effect of interaction between nanofillers and epoxy on mechanical and thermal properties of nanocomposites: theoretical prediction and experimental analysis, Adv. Polym. Technol., 2019, 2019(1), 8156718 Search PubMed.
- K. T. Marla and J. C. Meredith, Simulation of interaction forces between nanoparticles: End-grafted polymer modifiers, J. Chem. Theory Comput., 2006, 2(6), 1624–1631 CrossRef CAS PubMed.
- H. Eslami, M. Rahimi and F. Müller-Plathe, Molecular dynamics simulation of a silica nanoparticle in oligomeric poly (methyl methacrylate): a model system for studying the interphase thickness in a polymer–nanocomposite via different properties, Macromolecules, 2013, 46(21), 8680–8692 CrossRef CAS.
- P. T. Kien, N. Q. Thiet and T. Van Khai, Promoting aspect of palladium nanoparticles on spinel Pd@ CuCr2O4/γ–Al2O3 catalytic system in carbon monoxide oxidation, Vietnam J. Sci. Technol. B, 2017, 55(1), 70–76 Search PubMed.
- W. Li, Facile synthesis of monodisperse Bi2O3 nanoparticles, Mater. Chem. Phys., 2006, 99(1), 174–180 CrossRef CAS.
- W. Li and H. Cheng, Bi2O3/CuCr2O4–CuO core/shell structured nanocomposites: Facile synthesis and catalysis characterization, J. Alloys Compd., 2008, 448(1–2), 287–292 CrossRef CAS.
- K. Ghorai, et al., Facile synthesis of CuCr2O4/BiOBr nanocomposite and its photocatalytic activity towards RhB and tetracycline hydrochloride degradation under household visible LED light irradiation, J. Alloys Compd., 2021, 867, 157947 CrossRef CAS.
- A. A. Oladipo, CuCr2O4@ CaFe–LDO photocatalyst for remarkable removal of COD from high-strength olive mill wastewater, J. Colloid Interface Sci., 2021, 591, 193–202 CrossRef CAS PubMed.
- A. A. Oladipo, Rapid photocatalytic treatment of high-strength olive mill wastewater by sunlight and UV-induced CuCr2O4@CaFe–LDO, J. Water Process Eng., 2021, 40, 101932 CrossRef.
- K. Ghorai, et al., Facile synthesis of CuCr2O4/CeO2 nanocomposite: A new Fenton like catalyst with domestic LED light assisted improved photocatalytic activity for the degradation of RhB, MB and MO dyes, Appl. Surf. Sci., 2021, 536, 147604 CrossRef CAS.
- C.-L. Chiang, et al., Conversion of hydrogen/carbon dioxide into formic acid and methanol over Cu/CuCr2O4 catalyst, Int. J. Hydrogen Energy, 2017, 42(37), 23647–23663 CrossRef CAS.
- R. M. Mohamed and M. W. Kadi, Generation of hydrogen gas using CuCr2O4-g-C3N4 nanocomposites under illumination by visible light, ACS Omega, 2021, 6(6), 4485–4494 CrossRef CAS PubMed.
- S. K. Sivaranjani, et al., Efficiency of CuCr2O4/Titanium dioxide nanoparticles composite for organic dye removal in aqueous solutions, Environ. Res., 2023, 236, 116692 CrossRef CAS PubMed.
- A. Akhundi and A. Habibi-Yangjeh, Graphitic carbon nitride nanosheets decorated with CuCr2O4 nanoparticles: novel photocatalysts with high performances in visible light degradation of water pollutants, J. Colloid Interface Sci., 2017, 504, 697–710 CrossRef CAS PubMed.
- S. K. Korde, et al., Experimental investigations on copper-based nanoparticles for energy storage applications, 2023 Search PubMed.
- S. Sarkar, R. Akshaya and S. Ghosh, Nitrogen doped graphene/CuCr2O4 nanocomposites for supercapacitors application: effect of nitrogen doping on coulombic efficiency, Electrochim. Acta, 2020, 332, 135368 CrossRef CAS.
- R. Shafique, et al., Fabrication and characterization of MXene/CuCr2O4 nanocomposite for diverse energy applications, J. Mater. Res. Technol., 2023, 24, 2668–2677 CrossRef CAS.
- A. Baoum, M. Amin and R. Mohamed, Development of CuCr2O4 nanocomposite adopting decoration with polyaniline for acridine orange dye degradation, Appl. Nanosci., 2020, 10, 1501–1510 CrossRef CAS.
- H. Lahmar, et al., Photocatalytic degradation of crystal violet dye on the novel CuCr2O4/SnO2 hetero-system under sunlight, Optik, 2020, 219, 165042 CrossRef CAS.
- Z. Q. Hu, et al., Preparation and photoelectric properties of spinel MCr2O4 (M = Cu, Ni) nanopowders, Adv. Mater. Res., 2012, 415, 1594–1598 Search PubMed.
- Y. Gao, et al., Exchange bias effect in CuCr2O4/Cr2O3 nanogranular systems, J. Alloys Compd., 2016, 673, 126–130 CrossRef CAS.
- A. Mohammadi, V. Mahdikhah and S. Sheibani, Mechano-thermally synthesized CuCrO2/CuCr2O4 nanocomposite and improvement of photocatalytic activity by structure modification, Opt. Mater., 2022, 131, 112716 CrossRef CAS.
- M. Sarafrazi, A. R. Ghasemi and M. Hamadanian, Synergistic effect between CuCr2O4 nanoparticles and plasticizer on mechanical properties of EP/PU/CuCr2O4 nanocomposites: Experimental approach and molecular dynamics simulation, J. Appl. Polym. Sci., 2020, 137(46), 49425 CrossRef CAS.
- J. Bhagwan and J. I. Han, Facile synthesis of MWCNT/CuCr2O4 nanocomposite for aqueous hybrid supercapacitor, Inorg. Chem. Commun., 2024, 170, 113325 CrossRef CAS.
- D. Susanti, A. N. R. Pratama and H. Nurdiansah, The Effect of Nitrogen on the Capacitive Properties of N-Doped rGO/CuCr2O4 Composites as Materials for Supercapacitors, Eng. Chem., 2024, 5, 49–56 CrossRef.
- M. Daimalah, N. A. Laoufi and A. Mouheb, CuCr2O4-polystyrene immobilized photocatalyst: Facile fabrication, characterization and enhanced photodegradation of cefixime under visible light, Mater. Sci. Semicond. Process., 2022, 146, 106675 CrossRef CAS.
- Z. Chen, et al., Synthesis of CuCr2O4/Reduced Graphene Oxide Composite: A Green Catalyst for Selective Oxidation of Cyclohexane to Cyclohexanone with Hydrogen Peroxide, ChemistrySelect, 2017, 2(33), 10941–10945 CrossRef CAS.
- Y. Shi, et al., A robust CuCr2O4/SiO2 composite photothermal material with underwater black property and extremely high thermal stability for solar–driven water evaporation, Adv. Sustainable Syst., 2018, 2(3), 1700145 CrossRef.
- P. Holec, et al., ACr2O4/SiO2 (A = Zn, Cu, Cd) nanocomposites, their preparation and physical properties, IOP Conf. Ser.: Mater. Sci. Eng., 2011, 1–4 Search PubMed.
- I. A. Khan, et al., Dual-functional nanostructures: Photocatalytic and biomedical applications of vanadium oxide activated CuCr2O4/MCM-41, Chem. Eng. Sci., 2024, 287, 119714 CrossRef CAS.
- S. G. Hosseini, H. Sharifnezhad and M. Fathollahi, Quantitative and qualitative characterization of pure copper chromite nanocomposites for photodegradation of p-nitrophenol in aqueous medium, J. Electron. Mater., 2019, 48(7), 4207–4216 CrossRef CAS.
- V. Vinothkumar, et al., One-step designing of spinel CuCr2O4/Cr2O3 nanostructures as efficient positive electrode for a high-performance supercapacitor, Surf. Interfaces, 2024, 54, 105290 CrossRef CAS.
- S. Barman, et al., Kinetics of reductive isopropylation of benzene with acetone over nano-copper chromite-loaded H-mordenite, Ind. Eng. Chem. Res., 2006, 45(10), 3481–3487 CrossRef CAS.
- M. Ştefănescu, et al., Preparation and characterization of CuCr2O4/SiO2 and Cu2Cr2O4/SiO2 nanocomposites obtained from carboxylate complex combinations, J. Therm. Anal. Calorim., 2019, 138, 1887–1894 CrossRef.
- A. Abbasi, et al., Investigation of the synergic effect of silver on the photodegradation behavior of copper chromite nanostructures, J. Mater. Sci.: Mater. Electron., 2019, 30, 13994–14006 CrossRef CAS.
- R. Bajaj, M. Sharma and D. Bahadur, Visible light-driven novel nanocomposite (BiVO4/CuCr2O4) for efficient degradation of organic dye, Dalton Trans., 2013, 42(19), 6736–6744 RSC.
- S. G. Hosseini, et al., Orthogonal array design method for optimization experiments of composite oxides (CuCr2O4/CuO) synthesis and investigation of their catalytic activity on thermal decomposition of ammonium perchlorate particles, J. Energ. Mater., 2024, 42(2), 187–205 CrossRef CAS.
- R. Shafique, et al., Copper chromite/graphene oxide nanocomposite for capacitive energy storage and electrochemical applications, Int. J. Environ. Sci. Technol., 2022, 19(8), 7517–7526 CrossRef CAS.
- J. Yan, et al., CuCr2O4/TiO2 heterojunction for photocatalytic H2 evolution under simulated sunlight irradiation, Sol. Energy, 2009, 83(9), 1534–1539 CrossRef CAS.
- H. Kumar, et al., The effect of reduced graphene oxide on the catalytic activity of Cu–Cr–O–TiO2 to enhance the thermal decomposition rate of ammonium perchlorate: an efficient fuel oxidizer for solid rocket motors and missiles, RSC Adv., 2017, 7(58), 36594–36604 RSC.
- A. F. Shojaei, F. Shirini and E. Hedayati, Comparison of selective oxidation of aromatic alcohols using copper(II) chromite-titanium dioxide nanocomposite at reflux, light irradiation, and microwave conditions, Inorg. Nano-Met. Chem., 2017, 47(9), 1312–1317 CrossRef CAS.
- M. Nasrollahzadeh, et al., Green synthesis of CuO nanoparticles using aqueous extract of Thymus vulgaris L. leaves and their catalytic performance for N-arylation of indoles and amines, J. Colloid Interface Sci., 2016, 466, 113–119 CrossRef CAS PubMed.
- F. Nalimu, et al., Review on the phytochemistry and toxicological profiles of Aloe vera and Aloe ferox, Future J. Pharm. Sci., 2021, 7(1), 145 CrossRef PubMed.
- M. Sánchez, et al., Pharmacological update properties of Aloe vera and its major active constituents, Molecules, 2020, 25(6), 1324 CrossRef PubMed.
- S. Arunkumar and M. Muthuselvam, Analysis of phytochemical constituents and antimicrobial activities of Aloe vera L. against clinical pathogens, World J. Agric. Sci., 2009, 5(5), 572–576 CAS.
- K. Patel, et al., Insights into the solar-light-driven photocatalysis of greenly synthesized CoCr2O4 nanoparticles using Azadirachta indica, J. Sol-Gel Sci. Technol., 2025, 1–18 Search PubMed.
- H. Shahbazi-Alavi, et al., Nano-CuCr2O4: an efficient catalyst for a one-pot synthesis of tetrahydrodipyrazolopyridine, J. Chem. Res., 2016, 40(6), 361–363 CrossRef CAS.
- Y. Luo, et al., Selective catalytic transformation of cellulose into bio-based cresol with CuCr2O4@ MCM-41 catalyst, Cellulose, 2022, 29(1), 303–319 CrossRef CAS.
- S. Abdolmohammadi, H. Shahrokhi and S. Dahi-Azar, An Efficacious Synthesis of Xanthenones and Chromenopyrimidinediones Catalyzed by Copper Chromite Nanoparticles and Preliminary Assessment of Their Bioactivity, Polycyclic Aromat. Compd., 2024, 44(1), 418–430 CrossRef CAS.
- M. H. Ghasemi and E. Kowsari, The synergistic effect of copper chromite spinel nanoparticles (CuCr2O4) and basic ionic liquid on the synthesis of cyclopropanecarboxylic acids, Res. Chem. Intermed., 2016, 42, 7963–7975 CrossRef CAS.
- P. R. Patil, V. e. N. Krishnamurthy and S. S. Joshi, Effect of nano–copper oxide and copper chromite on the thermal decomposition of ammonium perchlorate, Propellants, Explos., Pyrotech., 2008, 33(4), 266–270 CrossRef CAS.
- H. Lahmar, et al., Photocatalytic reduction of Cr(VI) on the new hetero-system CuCr2O4/ZnO, J. Mol. Catal. A: Chem., 2012, 353, 74–79 CrossRef.
- R. Vinu, S. U. Akki and G. Madras, Investigation of dye functional group on the photocatalytic degradation of dyes by nano-TiO2, J. Hazard. Mater., 2010, 176(1–3), 765–773 CrossRef CAS PubMed.
- X. Liu, et al., Light down-converting characteristics of ZnO–Y2O2S: Eu3+ for visible light photocatalysis, J. Colloid Interface Sci., 2013, 404, 150–154 CrossRef CAS PubMed.
- P. Amornpitoksuk, et al., Effect of phosphate salts (Na3PO4, Na2HPO4, and NaH2PO4) on Ag3PO4 morphology for photocatalytic dye degradation under visible light and toxicity of the degraded dye products, Ind. Eng. Chem. Res., 2013, 52(49), 17369–17375 CrossRef CAS.
- X. Cai, et al., Photocatalytic degradation properties of Ni(OH)2 nanosheets/ZnO nanorods composites for azo dyes under visible-light irradiation, Ceram. Int., 2014, 40(1), 57–65 CrossRef CAS.
- K. Pagar, et al., Bio-inspired Sustainable Fabrication of CdO Nanoparticles Using Citrus sinensis Peel Extract for Photocatalytic Degradation of Rhodamine B Dye, Top. Catal., 2024, 67(17), 1169–1182 CrossRef CAS.
- H. Lund, Renewable energy strategies for sustainable development, Energy, 2007, 32(6), 912–919 CrossRef.
| This journal is © The Royal Society of Chemistry 2025 |