 Open Access Article
Open Access ArticleThe role of composition and diameter in the crystal purity of InAsxP1−x nanowires
Giada
Bucci
 a,
Vladimir G.
Dubrovskii
a,
Vladimir G.
Dubrovskii
 b,
Valentina
Zannier
b,
Valentina
Zannier
 *a,
Fabio
Beltram
*a,
Fabio
Beltram
 a and
Lucia
Sorba
a and
Lucia
Sorba
 a
a
aNEST, Scuola Normale Superiore of Pisa and Istituto Nanoscienze - Consiglio Nazionale delle Ricerche (CNR), 56127, Pisa, Italy. E-mail: valentina.zannier@nano.cnr.it
bFaculty of Physics, St. Petersburg State University, Universitetskaya Embankment 7-9, 199034 St. Petersburg, Russia
First published on 31st July 2025
Abstract
Ternary InAsxP1−x nanowires are widely considered promising building blocks for fundamental studies and applications in nano- and opto-electronics. However, it is admittedly challenging to maintain the necessary control over the crystal purity of nanowires. Furthermore, the crystal phase trends in ternary III–V nanowires remain generally unknown. In this work, we present wurtzite InAsxP1−x nanowires with different compositions x, ranging from 0 to 0.54, grown via chemical beam epitaxy on InP(111)B substrates using Au colloidal catalysts of different diameters. Transmission electron microscopy studies reveal that a pure wurtzite phase of the nanowires requires larger InAs fractions x for larger diameters of Au colloids (in particular, x = 0.36 for 20 nm diameter and x = 0.54 for 30 nm diameter Au colloids). We developed a model for the critical composition of thin vapor–liquid–solid III–V ternary nanowires xc corresponding to the transition from a polytypic structure at x < xc to a pure wurtzite structure at x > xc. For the InAsxP1−x material, the critical composition increases with the nanowire diameter, which explains our experimental findings. These results shed more light on the composition- and diameter-dependent polytypism, offering valuable insights for the design and crystal phase control of InAsxP1−x and other III–V ternary nanowires.
1. Introduction
III–V semiconductor nanowires (NWs) have been extensively studied due to their unique structural, electronic, and optical properties. Thanks to their high aspect ratio, NWs can relax strain radially, enabling the integration of highly lattice-mismatched materials without introducing dislocations. This feature enables greater flexibility in realizing NW heterostructures composed of dissimilar materials and bandgap engineering in such heterostructures compared to epi-layers.1 NWs are often synthesized via a metal-assisted vapor–liquid–solid (VLS) growth method. By tuning the growth conditions, substrate orientation, catalyst material and NW diameter, both hexagonal wurtzite (WZ) and cubic zincblende (ZB) crystal phases can be obtained in III–V NWs.2 A pure WZ phase is never observed in bulk III–V materials, but it is easily achieved in binary III–V NWs. This crystal phase is of great interest due to several intriguing properties, like the possibility of realizing the vanishing fine-structure splitting in WZ quantum dots (QDs)3 or obtaining a direct band gap structure of GaP, AlGaAs and some other III–V materials.4,5 Recently, significant efforts have been devoted to the synthesis and investigation of III–V ternary NWs, which offer promising applications due to their bandgap tunability and the emergence of novel properties. Among ternary alloys, InAsxP1−x has attracted particular interest due to its potential in a wide range of technologies. This material exhibits excellent piezoelectric properties6,7 and a tunable direct band gap ranging from 0.351 eV to 1.351 eV,8 making it highly suitable for applications in optoelectronics, quantum computing, and quantum information technologies. Additionally, the low effective mass and high electron mobility in InAsxP1−x are attractive for the realization of high-performance electronic devices. Significant research efforts have focused on the synthesis of InAsxP1−x quantum dots in InP NWs, which are promising for single photon emission.9,10Notably, improved optical properties have been observed when these QDs are inserted into an InAsxP1−x segment with a reduced InAs fraction,11 a strategy that requires precise control over the formation of ternary NWs. To date, ternary alloy engineering in NWs has mainly concentrated on compositional control.8,12,13 However, a defect-free structure of InAsxP1−x is absolutely required for all envisioned applications, because structural defects are known to degrade both the electronic and optical performance of the material. Due to the different band structures of the WZ and ZB phases, their intermixing in the same NW introduces electron and phonon scattering at the interfaces and the formation of charge traps.14–16 Avoiding polytypism remains challenging even in binary InP and InAs NWs, since the NW growth along the 〈111〉 direction promotes the formation of stacking faults due to the small energy difference between the WZ and ZB stacking in this direction.17 The crystal phase purity in both InP and InAs NWs has been shown to depend on several growth parameters, including growth temperature, V/III ratio, total precursor fluxes and diameter. The latter dependence has been ascribed to the Gibbs–Thomson effect.18 Defect-free WZ InP NWs have been demonstrated, to the best of our knowledge, only in NWs with very small diameters (up to 20 nm).16 In contrast, InAs NWs tend to form in the WZ crystal phase even at larger diameters, although the diameter dependence has also been reported for this material.18 Here, we present a study on the crystal phase purity of ternary InAsxP1−x NWs as a function of the NW diameter and chemical composition. We find the existence of a critical composition, above which the InAsxP1−x NWs are pure WZ. This critical composition increases with the NW diameter.
In general, the crystal phase trends in VLS III–V ternary NWs have not been systematically quantified and remain largely unknown. Consequently, we have developed a semi-quantitative model for the composition- and diameter-dependent polytypism in such NWs and show that the percentage of the WZ phase is generally a function of two variables, the NW diameter 2R and composition, represented by the InAs fraction in the ternary alloy, x. The diameter dependence originates from the contributions of edges separating the NW facets to the overall surface energy balance, which become important for thin NWs. Hence, the critical composition xc in VLS III–V ternary NWs, corresponding to the transition from a polytypic WZ/ZB intermix at x < xc to a pure WZ structure at x > xc, depends on the NW diameter. For the InAsxP1−x material, the critical composition increases with the NW diameter, which explains the data. This work extends the approaches originally developed for binary systems to a more complex case of VLS III–V ternary NWs, providing insights into the interplay between the diameter, composition and crystal phase. To the best of our knowledge, we demonstrate for the first time a diameter-dependent critical composition required to achieve phase-pure ternary wurtzite NWs. The results should be useful from a general perspective and for crystal phase engineering in thin InAsxP1−x NWs, and can be translated to other material systems.
2. Results and discussion
Defect-free WZ InP NWs grown from 10 nm diameter Au colloids have already been reported in the literature.19 At this diameter, the WZ crystal phase is energetically preferred also for InAs NWs.18 To explore the dependence of the crystal phase on the NW diameter and composition, we decided to focus on larger diameters, where a mixed ZB/WZ crystal phase typically occurs in both binary NWs.18,20,21 Different samples were grown using Au colloidal nanoparticles (NPs) of 20 nm and 30 nm diameters, which resulted in the NW diameters of ∼25 nm and up to ∼45 nm, respectively. All the NW samples investigated in this work were grown using the same procedure. After heating the InP (111)B substrate to 400 ± 10 °C under a tert-butyl phosphine (TBP) line pressure of 1 Torr, the NW growth was initiated by introducing trimethylindium (TMIn), TBP and, when required, tert-butyl arsine (TBAs). The growth was carried out for 120 minutes to obtain NWs with lengths of 1090 ± 200 nm for 20 nm diameter colloids and 330 ± 30 nm for 30 nm diameter colloids.Fig. 1 shows the results for the NWs grown from 20 nm diameter NPs. Additional TEM images are reported in section S1 of the SI. In panel (a), a 45°-tilted view SEM image shows an ensemble of binary InP NWs grown using TMIn and TBP precursor line pressures of 0.30 Torr and 1.05 Torr, respectively. Two different NW morphologies are highlighted with orange and green boxes. The former, referred to as “type 1” in the following, is characterized by the enhanced radial growth, indicated by the red arrow in Fig. 1(a). This enhancement has previously been reported in the literature22 and is attributed to the NW sections with a high density of stacking faults. This is also confirmed in our sample. Panel (b) of Fig. 1 presents a high-resolution transmission electron microscopy (HRTEM) image of such a NW, showing the presence of crystal defects. In contrast, the latter type of NW, labelled “type 2” in the following, displays a nearly untapered morphology with no significant radial growth or bulges along its length. This morphology is characterized by a low stacking fault density or even a fully defect-free WZ crystal phase, as demonstrated by the HRTEM image in Fig. 1(c).
To identify the optimal growth conditions for obtaining pure WZ binary InP NWs with an untapered geometry, we studied the growth under different V/III ratios by varying the TBP line pressure from 0.50 Torr to 1.50 Torr and keeping the TMIn pressure constant at 0.30 Torr. In all cases, we obtained NW ensembles with both morphologies. Hence, we selected the growth conditions that minimized the occurrence of type 1 NWs with high stacking fault density. Based on this criterion, the optimal TBP line pressure was identified as 1.05 Torr (i.e., the sample in Fig. 1(a)). Under these precursor fluxes, approximately 42% of the NWs exhibit the unwanted type 1 morphology, while the remaining portion corresponds to type 2 morphology. A total of 50 NWs were analyzed for the percentage estimation. After selecting the best conditions for binary InP NWs of this diameter, we investigated the growth of InAsxP1−x ternary NWs. To evaluate the effect of As incorporation on the crystal purity of the NWs, we grew a series of samples at the same temperature of 400 °C, keeping the TMIn line pressure at 0.30 Torr and the constant total group-V line pressure (TBAs + TBP) at 1.05 Torr. Fig. 1(d) and (g) show the SEM images of two different InAsxP1−x samples, grown using TBAs and TBP line pressures of 0.10 Torr and 0.95 Torr, respectively (resulting in x = 0.09), and 0.20 Torr and 0.85 Torr, respectively (resulting in x = 0.36). TEM analysis of the first sample in Fig. 1(e) and (f) reveals that stacking faults are still present in the InAs0.09P0.91 NWs, but their occurrence is reduced compared to InP NWs. Only ∼36% of InAs0.09P0.91 NWs exhibit crystal defects. In contrast, Fig. 1(h) shows a TEM image of a defect-free InAs0.36P0.64 WZ NW. The corresponding HRTEM image in panel (i) demonstrates the absence of any stacking faults. All NWs of this sample exhibit the defect-free WZ crystal phase. Therefore, a higher fraction of InAs “x” in ternary InAsxP1−x NWs grown from 20 nm Au NPs improves the crystal quality in the WZ phase. By comparing the SEM images of these samples, it is evident that the incorporation of As into the NWs leads to increased radial growth, resulting in more-tapered NWs grown at the same temperature. This effect is likely due to the higher sticking coefficient of As than that of P,8 which enhances the nucleation probability on the NW sidewalls. Additionally, the average axial growth rate decreases monotonically with the increase of the As fraction from 13 nm min−1 in pure InP NWs to 9 nm min−1 in InAs0.36P0.64 NWs.
Fig. 2 presents the results of a similar study for NWs grown from Au NPs of 30 nm diameter. Additional TEM images are shown in section S2 of the SI. Panel (a) shows a 45°-tilted SEM image of an ensemble of binary InP NWs grown at the TMIn and TBP line pressures of 0.30 Torr and 0.40 Torr, respectively. As above, several V/III ratios were explored to identify the optimal growth conditions for binary InP NWs. However, in this case, the two distinct morphologies observed for 20 nm diameter colloids are not evident. The NWs appear highly defective across the entire range of TBP line pressures. Employing a TBP line pressure of 1.05 Torr, as for thinner NWs in Fig. 1(a), resulted in NWs with a high radial growth rate and limited elongation. Consequently, we decided to lower the TBP flux to 0.40 Torr, which gave nearly untapered NWs with negligible radial growth, as demonstrated in Fig. 2(a). Fig. 2(b) and (c) show the TEM and HRTEM images of a representative NW from this sample, where a high stacking fault density is clearly visible. The transition from a mixed ZB/WZ structure to a pure WZ phase is observed for higher InAs fractions in NWs, as observed earlier for 20 nm Au NPs. However, achieving a pure WZ phase requires a higher InAs fraction than that for 20 nm Au NPs. Fig. 2(d) shows an ensemble of InAs0.46P0.54 NWs grown under TBAs and TBP line pressures of 0.15 Torr and 0.25 Torr, respectively. These NWs remain highly defective, as demonstrated by the representative TEM image in Fig. 2(e) and the corresponding HRTEM image in Fig. 2(f). Defect-free WZ NWs shown in Fig. 2(g), (h) and (i) are grown under TBAs and TBP line pressures of 0.20 Torr and 0.20 Torr, respectively. This results in a higher InAs fraction of x = 0.54, and does not lead to any significant change in either the axial or radial growth rate. In contrast to the earlier suggestion,22 lower axial growth rates are not the only requirement for achieving a pure WZ phase in III–V NWs, at least in our case of ternary InAsxP1−x NWs. It should be noted, however, that the increased InAs fraction results in a lower vertical yield of NWs, with a significant fraction of the NWs failing to nucleate. This can be seen in Fig. 2(g), where some catalyst NPs without any NWs are clearly visible on the substrate. This should be related to difficult nucleation of NWs on the substrate surface under the growth conditions that were chosen to obtain untapered NWs in the pure WZ phase rather than to maximize their yield.
It is clear at this point that the crystal phase of ternary InAsxP1−x NWs strongly depends on the InAs fraction, with a critical composition xc at which the structure transforms from highly defective to pure WZ. This critical composition increases with the NW diameter (or, equivalently, with the Au NP diameter from which the NWs grow). Previous studies have speculated that defect formation in both InP and InAs binary NWs is related to the concentration of liquid In in the catalyst droplets.18,19 Therefore, we performed TEM-EDS measurements of the droplet compositions and contact angles for all the NWs discussed above. At least 20 NWs were investigated for each sample. These post-growth measurements were taken after cooling the samples in the absence of group V fluxes. This method has previously been demonstrated not to significantly alter the catalyst composition in binary NWs, providing an estimation of the catalyst composition during growth.23 Here, we used the same cooling procedure, so that we can compare the measured composition of the catalyst nanoparticles of the different samples after growth. The data in Table 1 show that the In/Au ratio inside the catalysts varies between 20 and 30 nm diameter samples. However, there is no significant change in the NP composition between binary InP and ternary InAsxP1−x NWs. These results indicate that, unlike in the case of binary InP NWs,19 the crystal phase purity of ternary InAsxP1−x NWs is not directly linked to the In content in the catalyst NPs (of course, both As and P atoms were not detected in the NPs due to their high volatility). Additionally, Table 1 shows no significant change in the average contact angle of the NPs on top of different NWs.
| NP diameter (nm) | InAs fraction x | In (at%) | Au (at%) | Average contact angle (°) |
|---|---|---|---|---|
| 20 | 0 | 54 ± 2 | 46 ± 2 | 94 ± 5 |
| 20 | 0.09 | 52 ± 8 | 48 ± 8 | 87 ± 5 |
| 20 | 0.36 | 49 ± 3 | 51 ± 3 | 93 ± 3 |
| 30 | 0 | 65 ± 2 | 35 ± 2 | 84 ± 8 |
| 30 | 0.46 | 68.6 ± 0.9 | 31.4 ± 0.9 | 100 ± 5 |
| 30 | 0.54 | 65.3 ± 0.8 | 34.7 ± 0.8 | 98 ± 3 |
3. Modeling
To understand the composition- and diameter-dependent polytypism and crystal phase purity of ternary InAsxP1−x NWs under optimal conditions, we have developed the following energetic model. In the first approximation, we ignored the vapor–solid growth of tapered shells around the NW cores, thus assuming that the crystal phase is determined during the VLS growth step of the NW core. This was earlier justified for binary III–V NWs, where the tapered shells inherit the crystal phase of the VLS cores.24–27 According to ref. 26–31, the conditions for the formation of VLS NWs in the WZ phase are equivalent to the preferred formation of vertical WZ facets relative to the truncated ZB facets: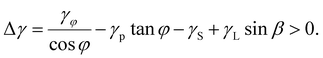 | (1) |
Here, β is the droplet contact angle, φ is the angle of the truncated facet with respect to the vertical facet, γφ is the solid–liquid surface energy of the truncated facet, γP is the solid–liquid surface energy of the horizontal facet under the droplet, γS is the solid–vapor surface energy of the vertical WZ facet, and γL is the liquid–vapor surface energy of the droplet. The two solid–liquid surface energies γφ and γP are phase-independent.24,32 The geometries of the NW tip and the interfaces of interest are illustrated in Fig. 3(a). The surface energies γ in eqn (1) are usually considered radius-independent.26–32 This is valid for thick enough NWs with negligible contribution of the edges separating the side facets. For thin NWs, these edges start to play an important role in the total formation energy of hexahedral NWs.24,33–35 The liquid surface and planar horizontal facet under the droplet have no edges; hence, γL and γp are independent of the NW top radius (which is directly related to the nominal radius of Au colloids R).
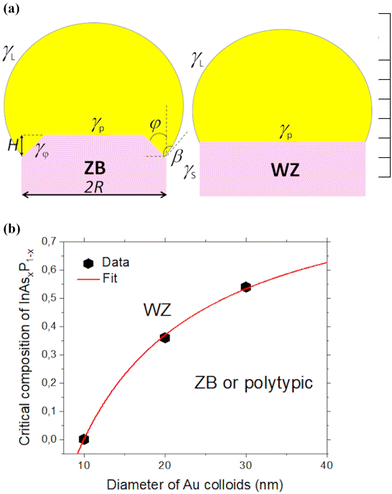 | ||
| Fig. 3 (a) Illustration of the truncated ZB and planar WZ growth interfaces under the catalyst droplets, showing the surface energies of interest. (b) Critical composition of InAsxP1−x NWs xcversus the Au nanoparticle diameter (symbols), fitted with eqn (6) at x∞c = 0.96, A = 5 nm and B = 3 nm (line). | ||
Six equivalent facets of truncated ZB NWs and vertical WZ NWs are separated by edges. The surface energies γ per the surface area are ∼RH, where H is the truncation height (see Fig. 3(a)). Hence, the edge energies are inversely proportional to R. The total surface energies with the edge contributions are given by ref. 24 and 34:
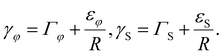 | (2) |
Here, Γ denotes the surface energy of flat facets and ε is the specific edge energy per unit length, with γφ → Γφ at large R. For InAsxP1−x ternary NWs, the solid–vapor and liquid–vapor surfaces and edge energies depend on the composition x. The surface energies and edge energies of III–V ternary NWs in both ZB and WZ phases are unknown. Therefore, we used the simplest linear dependence on the composition in the first approximation:1,13
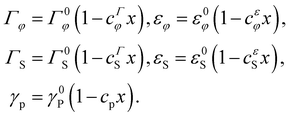 | (3) |
Here, the surface and edge energies labelled “0” correspond to pure InP NWs, and x-independent coefficients c describe linear interpolation of the surface and edge energies between pure InP and InAs. According to the calculations of ref. 36, the vapor–solid surface energy Γ0S of unpassivated vertical WZ facets (1![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 00) and (11
00) and (11![[2 with combining macron]](https://www.rsc.org/images/entities/char_0032_0304.gif) 0) at zero temperature is 23.5% lower for InAs NWs, corresponding to positive cΓS = 0.235. This corresponds to the well-known trend of a stable WZ phase in binary InAs NWs, where the ZB stacking is the exception rather than the rule.12,13 However, nothing is known about the edge energies of ternary NWs and the solid–liquid surface energies Γφ and γp, which is why the other coefficients c in eqn (3) can be of either sign.
0) at zero temperature is 23.5% lower for InAs NWs, corresponding to positive cΓS = 0.235. This corresponds to the well-known trend of a stable WZ phase in binary InAs NWs, where the ZB stacking is the exception rather than the rule.12,13 However, nothing is known about the edge energies of ternary NWs and the solid–liquid surface energies Γφ and γp, which is why the other coefficients c in eqn (3) can be of either sign.
The surface energy of a liquid droplet should be independent of the negligibly small concentrations of P and As atoms, and even of the Au concentration, because the lower surface energy liquid In accumulates at the droplet surface.28–30 The droplet contact angle β is the only kinetic parameter in eqn (1). It generally depends on the total V/III ratio and As/P flux ratio (and hence the composition x) due to kinetic growth effects.23 We used linear interpolation of sin![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) β in eqn (1) of the form: sin
β in eqn (1) of the form: sin![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) β(x) ≅ sin
β(x) ≅ sin![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) β0 + (cos
β0 + (cos![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) β0)cβx, with the coefficient cβ, and β0 is the average contact angle of the droplets on top of pure InP NWs. This assumes a small variation of the contact angle compared to β0. Post-growth measurements (see Table 1) show that the average contact angle of all the NWs is only slightly above 90°. In this case, the composition dependence of the contact angle is very weak, but this is not critical for our semi-quantitative model.
β0)cβx, with the coefficient cβ, and β0 is the average contact angle of the droplets on top of pure InP NWs. This assumes a small variation of the contact angle compared to β0. Post-growth measurements (see Table 1) show that the average contact angle of all the NWs is only slightly above 90°. In this case, the composition dependence of the contact angle is very weak, but this is not critical for our semi-quantitative model.
Using eqn (2) and (3) in eqn (1), we obtain the condition of the preferred WZ phase formation in the form Δγ(R, x) > 0. This allows us to determine the critical composition xc corresponding to the ZB-to-WZ phase transition as a function of the radius R. After some manipulations, eqn (1) can be put in the form
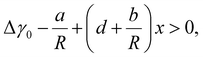 | (4) |
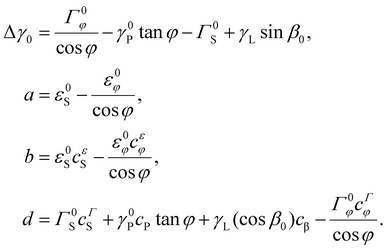 | (5) |
Clearly, Δγ0 is the surface energy difference that determines the crystal phase of thick InP NWs at a contact angle β0, a/R is the composition-independent edge contribution, and the x term contains two contributions from the surface (d) and edge (b/r) energies. The WZ phase is energetically preferred when
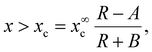 | (6) |
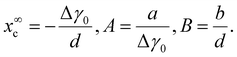 | (7) |
The parameters x∞c, A and B combine together the largely unknown energetic factors influencing the crystal phase trend in thin III–V ternary NWs, and yield a simple analytical shape of the critical curve xc(R). For x below the critical curve xc(R), the crystal phase can be polytypic rather than pure ZB due to a periodically changing geometry of the growth interface.26,30
Our model describes different crystal phase trends depending on the signs and magnitudes of the coefficients in eqn (6). The data shown in Fig. 3(b) demonstrate that the pure WZ phase of InAsxP1−x grown in our experiments requires higher fractions of InAs for larger radii. The first data point in Fig. 3(b) corresponds to WZ InP NWs at R = 5 nm,19 which requires A = 5 nm in eqn (6). A monotonically increasing dependence xc(R) in the investigated range of NW compositions and Au radii requires positive x∞c and positive A. From eqn (7), this corresponds to differently signed coefficients Δγ0 and d, and equally signed coefficients Δγ0 and a (Δγ0 > 0, a > 0 and d < 0, or Δγ0 < 0, a < 0 and d > 0). The sign of B in eqn (6) is less important. For example, the data points can reasonably be fitted by xc = x∞c(1 − A/R) at B ≅ 0. The fit shown in Fig. 3(b) is obtained with x∞c = 0.96, A = 5 nm and B = 3 nm. This corresponds to the pure WZ phase of very thin InP NWs (R ≤ 5 nm), ZB or the polytypic phase of thicker InP NWs, and the almost pure WZ phase of thick InAs NWs under our growth conditions. The trend of a more stable WZ phase of InAs NWs relative to InP and all other binary III–V NWs is well known and has been reproduced by different epitaxy techniques.24,37
Experimental details
The NW samples in this work are epitaxially grown by means of Au-assisted VLS growth in a Riber C-21 chemical beam epitaxy (CBE) system. Trimethylindium (TMIn), pre-cracked tert-butyl arsine (TBAs) and tert-butyl phosphine (TBP) are used as gaseous metal–organic precursors, for which it is possible to measure the line pressures as a reference for the fluxes introduced into the chamber. The substrate temperature is measured with a pyrometer, providing an accuracy of ±10 °C. Prior to the mounting of the substrate in the reactor chamber, Au catalyst nanoparticles are deposited through colloidal dispersion. Commercial aqueous solutions of Au colloids with nominal diameters of 20 nm and 30 nm (BBInternational EM.GCnn) are employed. Fe-doped InP(111)B substrates are first functionalized by wetting the surface through dropcasting of 0.1% poly-L-lysine solution for 30s, then rinsed in deionized water and blow-dried under N2 flux. The colloid solution is dropcast onto the substrate surface, which is then rinsed in DI water and blow-dried. Before inserting the sample into the growth chamber, the substrate is outgassed at 300 °C on a heating stage for around five minutes to get rid of the moisture.After growth, the samples are imaged employing scanning electron microscopy (Zeiss Merlin SEM) operating at 5 kV, from both top and side views, to check the NW morphology, and the ternary alloy composition of the NWs is measured through energy-dispersive X-ray spectroscopy (EDS) analysis employing a Bruker Quantax EDS system mounted on a Zeiss Ultraplus scanning electron microscope. Transmission electron microscopy (TEM), scanning transmission electron microscopy (STEM) and STEM–EDS of the catalyst nanoparticles were performed using a JEOL JEM-F200 multipurpose microscope, working at 200 kV and equipped with a Schottky field-emission gun and a silicon drift detector. TEM images were recorded with a GATAN RIO16 CMOS camera. The crystal structure of 20 NWs per sample is analyzed by TEM for statistical purposes. The percentage of defective NWs is calculated by counting the number of NWs in which stacking faults are observed, regardless of their density and distribution along the growth axis, over the total number of NWs measured.
4. Conclusions
In summary, we have demonstrated Au-catalyzed formation of InAsxP1−x NWs in a pure WZ crystal phase for high enough fractions of InAs in the ternary alloy. The critical composition xc required for the formation of the pure WZ phase in ternary NWs increases from 0.36 for 20 nm diameter to 0.54 for 30 nm diameter Au nanoparticles, and is expected to increase further for larger diameters. While a more predominant WZ phase in binary InAs NWs relative to InP NWs has been known, the composition- and diameter-dependent crystal phase purity of Au-catalyzed VLS ternary InAsxP1−x NWs with small diameters has been studied here for the first time to our knowledge. The developed model explains the data on surface energetic grounds, where the effective surface energies vary with the NW composition, and the diameter dependence is due to the edge contributions to the overall energy balance. A more advanced study should include kinetic contributions to the phase purity of III–V ternary NWs. These obtained results call for further systematic investigations of the crystal phase transitions in InAsxP1−x NWs under a wider range of growth conditions and Au nanoparticle diameters, and similar investigations for other III–V ternary NWs grown by different methods.Author contributions
G. B., V. Z. and L. S. performed the growth of the NWs; G. B. performed the TEM measurements; V. G. D. carried out the modeling part; F. B. contributed to writing, review and editing. All authors have read and agreed to the published version of the manuscript.Conflicts of interest
There are no conflicts to declare.Data availability
The authors confirm that all data supporting the findings of this study are available within the manuscript.Supplementary information is available. In the SI, additional TEM images of the samples presented in the main text are provided. S1 contains the images of NWs obtained with 20 nm diameter colloids, S2 the the images of NWs obtained with 30 nm diameter colloids. See DOI: https://doi.org/10.1039/d5nr02409a
Acknowledgements
G.B., V.Z. and L.S. acknowledge the Center for Instrument Sharing of the University of Pisa (CISUP) for the TEM facilities and Prof. Enrico Mugnaioli and Dr. Michele Alderighi for the help during the TEM measurement sessions. This research was funded by the Horizon EIC-PATHFINDEROPEN-01-01 QCEED (no. 101185617) and the PNRR MUR PE0000023-NQSTI projects. VGD acknowledges St-Petersburg State University for the research project no. 129360164.References
- J. Johansson and K. A. Dick, CrystEngComm, 2011, 13, 7175 RSC.
- P. Caroff, J. Bolinsson and J. Johansson, IEEE J. Sel. Top. Quantum Electron., 2011, 17, 829–846 Search PubMed.
- R. Singh and G. Bester, Phys. Rev. Lett., 2009, 103, 063601 CrossRef PubMed.
- B. C. da Silva, O. D. D. Couto, H. T. Obata, M. M. de Lima, F. D. Bonani, C. E. de Oliveira, G. M. Sipahi, F. Iikawa and M. A. Cotta, Sci. Rep., 2020, 10, 7904 CrossRef PubMed.
- S. Assali, J. Greil, I. Zardo, A. Belabbes, M. W. A. De Moor, S. Koelling, P. M. Koenraad, F. Bechstedt, E. P. A. M. Bakkers and J. E. M. Haverkort, J. Appl. Phys., 2016, 120, 044304 CrossRef.
- I. Kim, H. S. Kim and H. Ryu, Molecules, 2019, 24, 3249 CrossRef CAS.
- J. Liu, H. Yang, V. Khayrudinov, H. Lipsanen, H. Nie, K. Yang, B. Zhang and J. He, Photonics Res., 2021, 9, 1811–1819 CrossRef.
- A. I. Persson, M. T. Björk, S. Jeppesen, J. B. Wagner, L. R. Wallenberg and L. Samuelson, Nano Lett., 2006, 6, 403–407 CrossRef CAS.
- D. Dalacu, P. J. Poole and R. L. Williams, Nanotechnology, 2019, 30, 232001 CrossRef CAS.
- G. Bucci, V. Zannier, F. Rossi, A. Musiał, J. Boniecki, G. Sęk and L. Sorba, ACS Appl. Mater. Interfaces, 2024, 16, 26491–26499 CrossRef CAS PubMed.
- S. Haffouz, P. J. Poole, J. Jin, X. Wu, L. Ginet, K. Mnaymneh, D. Dalacu and R. L. Williams, Appl. Phys. Lett., 2020, 117, 113102 CrossRef CAS.
- E. D. Leshchenko and V. G. Dubrovskii, Nanomaterials, 2023, 13, 1659 CrossRef CAS PubMed.
- M. Ghasemi, E. D. Leshchenko and J. Johansson, Nanotechnology, 2021, 32, 072001 CrossRef CAS PubMed.
- K. A. Dick, C. Thelander, L. Samuelson and P. Caroff, Nano Lett., 2010, 10(9), 3494–3499 CrossRef CAS.
- M. E. Reimer, G. Bulgarini, A. Fognini, R. W. Heeres, B. J. Witek, M. A. M. Versteegh, A. Rubino, T. Braun, M. Kamp, S. Höfling, D. Dalacu, J. Lapointe, P. J. Poole and V. Zwiller, Phys. Rev. B, 2016, 93, 195316 CrossRef.
- D. Dalacu, K. Mnaymneh, J. Lapointe, X. Wu, P. J. Poole, G. Bulgarini, V. Zwiller and M. E. Reimer, Nano Lett., 2012, 12(11), 5919–5923 CrossRef CAS PubMed.
- T. Chiaramonte, L. H. G. Tizei, D. Ugarte and M. A. Cotta, Nano Lett., 2011, 11(5), 1934–1940 CrossRef CAS PubMed.
- J. Johansson, K. A. Dick, P. Caroff, M. E. Messing, J. Bolinsson, K. Deppert and L. Samuelson, J. Phys. Chem. C, 2010, 114(9), 3837–3842 CrossRef CAS.
- F. Wang, C. Wang, Y. Wang, M. Zhang, Z. Han, S. Yip, L. Shen, N. Han, E. Y. B. Pun and J. C. Ho, Sci. Rep., 2016, 6, 32910 CrossRef CAS.
- S. Lehmann, J. Wallentin, D. Jacobsson, K. Deppert and K. A. Dick, Nano Lett., 2013, 13(9), 4099–4105 CrossRef CAS.
- S. Paiman, Q. Gao, H. H. Tan, C. Jagadish, X. Zhang and J. Zou, J. Cryst. Growth, 2013, 383, 100–105 CrossRef CAS.
- P. J. Poole, D. Dalacu, X. Wu, J. Lapointe and K. Mnaymneh, Nanotechnology, 2012, 23, 385205 CrossRef CAS PubMed.
- V. Zannier, F. Rossi, V. G. Dubrovskii, D. Ercolani, S. Battiato and L. Sorba, Nano Lett., 2018, 18(1), 167–174 CrossRef CAS PubMed.
- V. G. Dubrovskii, Nucleation theory and growth of nanostructures, Springer, Heidelberg – New York – Dordrecht – London, 2014 Search PubMed.
- V. G. Dubrovskii, N. V. Sibirev, X. Zhang and R. A. Suris, Cryst. Growth Des., 2010, 10, 3949 CrossRef CAS.
- C. Y. Wen, J. Tersoff, K. Hillerich, M. C. Reuter, J. H. Park, S. Kodambaka, E. A. Stach and F. M. Ross, Phys. Rev. Lett., 2011, 107, 025503 CrossRef.
- D. Jacobsson, F. Panciera, J. Tersoff, M. C. Reuter, S. Lehmann, S. Hofmann, K. A. Dick and F. M. Ross, Nature, 2016, 531, 317 CrossRef CAS PubMed.
- V. G. Dubrovskii, Cryst. Growth Des., 2017, 17, 2544 CrossRef CAS.
- V. G. Dubrovskii, N. V. Sibirev, N. N. Halder and D. Ritter, J. Phys. Chem. C, 2019, 123, 18693 CrossRef CAS.
- F. Panciera, Z. Baraissov, G. Patriarche, V. G. Dubrovskii, F. Glas, L. Travers, U. Mirsaidov and J. C. Harmand, Nano Lett., 2020, 20, 1669 CrossRef CAS.
- V. G. Dubrovskii and F. Glas, Cryst. Growth Des., 2024, 24, 9660 CrossRef CAS.
- F. Glas, J. C. Harmand and G. Patriarche, Phys. Rev. Lett., 2007, 99, 146101 CrossRef.
- T. Akiyama, K. Sano, K. Nakamura and T. Ito, Jpn. J. Appl. Phys., 2006, 45, L275 CrossRef CAS.
- M. Rosini and R. Magri, ACS Nano, 2010, 4, 6021 CrossRef PubMed.
- V. Pankoke, P. Kratzer and S. Sakong, Phys. Rev. B: Condens. Matter Mater. Phys., 2011, 84, 075455 CrossRef.
- N. V. Sibirev, M. A. Timofeeva, A. D. Bolshakov, M. V. Nazarenko and V. G. Dubrovskii, Phys. Solid State, 2010, 52, 1531 CrossRef.
- K. A. Dick, P. Caroff, J. Bolinsson, M. E. Messing, J. Johansson, K. Deppert, L. R. Wallenberg and L. Samuelson, Semicond. Sci. Technol., 2010, 25, 024009 CrossRef.
| This journal is © The Royal Society of Chemistry 2025 |


