Strain-tunable optoelectronic and photocatalytic properties of 2D GaN/MoSi2P4 heterobilayers: potential optoelectronic/photocatalytic materials†
Huabing
Shu
 *a,
Feifan
Wang
b,
Kai
Ren
*a,
Feifan
Wang
b,
Kai
Ren
 *c and
Jiyuan
Guo
*c and
Jiyuan
Guo
 a
a
aSchool of Science, Jiangsu University of Science and Technology, Zhenjiang 212001, China. E-mail: shuhuabing@just.edu.cn
bJiangsu Co-Innovation Centre of Efficient Processing and Utilization of Forest Resources, College of Chemical Engineering, Nanjing Forestry University, Nanjing 210037, China
cSchool of Mechanical and Electronic Engineering, Nanjing Forestry University, Nanjing 210037, China. E-mail: kairen@njfu.edu.cn
First published on 17th December 2024
Abstract
Herein, we propose a new GaN/MoSi2P4 van der Waals (vdWs) heterostructure constructed by vertically stacking GaN and MoSi2P4 monolayers. Its electronic, optical, and photocatalytic properties are explored via DFT+G0W0+BSE calculations. The calculated binding energy and phonon spectrum demonstrated the material's high stabilities. The projected band structure of GaN/MoSi2P4 suggests that it has a desirable direct bandgap and displays type-I band alignment. It also exhibits a particularly large absorption coefficient for visible and near-infrared light while considering electron–hole interactions. Intriguingly, a small biaxial tensile strain of +1% can transform the band alignment to type-II using a direct Z-scheme mechanism for water splitting. The Z-scheme optimizes redox ability, thus perfectly engulfing the redox potentials of water and showing excellent photocatalytic activity in different layers. Our findings indicate that the GaN/MoSi2P4 vdWs heterostructure is a promising optoelectronic and photocatalytic material.
1 Introduction
Two-dimensional (2D) materials have attracted tremendous interest owing to their novel properties and promising applications in electronics and optoelectronics.1–3 For a single 2D material, there exists a limitation in photocatalytic, photovoltaic, and optoelectronic devices owing to the swift recombination of photoexcited charge carriers. The vertical stacking of diverse 2D materials facilitates the fabrication of van der Waals (vdWs) heterostructures, which have been achieved experimentally, such as graphene/MoS2,4 MoSe2/MoS2,5 and h-BN/WS2.6 At present, vdWs engineering has been regarded as the most feasible way to integrate the intrinsic properties of two pristine 2D materials.7–9 More importantly, semiconducting vdWs heterostructures can offer a large active junction area to absorb more photons and induce more exciton states, thus leading to enhanced light absorption and photocurrent. In such vdWs heterostructures, type-II band alignment at interfaces is especially preferred owing to effective space segregation of photoexcited electrons and holes, consequently suppressing the recombination of electron–hole (e–h) pairs. In addition, type-II vdWs heterostructures with the Z-scheme can optimize redox ability through interface electron–hole recombination and facilitate the hydrogen evolution reaction (HER) and oxygen evolution reaction (OER),10 thereby ensuring the photocatalytic activity of the vdWs heterostructure. As a result, the design and exploration of new 2D vdWs heterostructures are highly necessary for photocatalytic, photovoltaic, and optoelectronic devices.Very recently, a novel 2D MoSi2N4 monolayer has been successfully synthesized by employing chemical vapor deposition, which displayed excellent environmental stability, a high strength of ∼66 GP, a moderate indirect bandgap of ∼1.94 eV, and a high hole (electron) mobility of ∼1200 (270) cm2 V−1 s−1.11 P and N atoms belong to the same main group and possess the same number of valence electrons. A MoSi2P4 monolayer has also been demonstrated to be dynamically and thermally stable12 and exhibits a direct bandgap of ∼1.32 eV at the HSE06 level. The predicted hole (electron) mobility can achieve a large value of ∼1429 (258) cm2 V−1 s−1,13 which is significantly larger than those (hole: 200 cm2 V−1 s−1; electron: 72 cm2 V−1 s−1)14 of monolayer MoS2. A MoSi2P4 monolayer with a direct gap also shows a tunable electronic property under a wide strain range and an outstanding absorption of visible light.13,15 However, monolayer gallium nitride (GaN) has also been synthesized by applying graphene-encapsulated growth technique and sonochemical exfoliation.16,17 The GaN monolayer is a wide indirect semiconductor with a bandgap of ∼4.44 eV (ref. 18) and high carrier mobility of ∼103 cm2 V−1 s−1,19 leading to the prominent absorption of near-ultraviolet light. The electronic and optical properties of 2D GaN can be broadly modulated under external strain.18 Some theoretical reports have also pointed out that monolayer GaN can form a type-II heterostructure with other monolayer MX2 (such as WS2, WSe2, and MoS2).20,21 These suggest that GaN and MoSi2P4 monolayers have potential applications in electronic and optoelectronic devices.
Because MoSi2P4 and GaN monolayers are both semiconductors with the same hexagonal structure and similar lattice parameters (MoSi2P4: 3.46 Å;22 GaN: 3.20 Å (ref. 18)), a GaN/MoSi2P4 vdWs heterobilayer stacked vertically by GaN and MoSi2P4 monolayers is proposed, and the following questions arise immediately: (1) Can a GaN monolayer be stacked on a MoSi2P4 monolayer to form structurally stable vdWs heterostructures? (2) What are the electronic and optical characteristics of such vdWs heterostructures? (3) Can such vdWs heterostructures be used in device design? To answer these questions, this study employs first-principles calculations to investigate GaN/MoSi2P4 vdWs heterobilayer with six stacking configurations. The structural, electronic, photocatalytic, and optical properties of the GaN/MoSi2P4 with AA5 stacking mode are investigated systemically owing to their relatively high stabilities. AA5 GaN/MoSi2P4 displays type-I band alignment, which is unfavorable for separating photoexcited e–h pairs. Its optical spectrum shows desirable absorption from the near-infrared to the visible range. Notably, band alignment can be modulated by the application of in-plane biaxial strains. Specifically, the transition from type-I to type-II band alignment can be achieved by introducing a small tensile strain. Our results point out the potential of the AA5 GaN/MoSi2P4 heterobilayer as a novel strain-tunable optoelectronic and photocatalytic material.
2 Computational details and methods
All the ground-state calculations were performed using Quantum ESPRESSO code.23 A vacuum thickness of 23 Å in the direction perpendicular to the surface was adopted to reduce the coupling between the two adjacent layers. Perdew–Burke–Ernzerhof (PBE) functional24 was used to optimize the structures of the observed systems. We used the vdW-DF method to describe the weak interlayer coupling,25 and an energy cutoff of 952 eV was utilized for the plane wave expansion. The convergence thresholds for force and energy were established at 10−2 eV and 10−5 eV Å−1, respectively. The Brillouin zone was sampled using a k-point mesh with dimensions of 18 × 18 × 1. The phonon band structure was computed using the density functional perturbation theory,26 and the acoustic sum rule was used for the vibrational frequencies of the Γ point. To examine the thermal stability, ab initio molecular dynamics (AIMD) simulations were executed, in which a 4 × 4 × 1 supercell was adopted at 400 K and dynamics simulations lasted for 10 ps (a time step of 1 fs). The Nosé algorithm was employed to control the temperature in the NVT ensemble.The free energy change (ΔG) during the hydrogen reduction and water oxidation reactions was calculated utilizing the method developed by Nørskov et al.27,28 The ΔG of an electrochemical reaction can be obtained by applying the following equation:
| ΔG = ΔE + ΔEzpe − TΔS + ΔGpH + ΔGU, | (1) |
The quasi-particle (QP) energies were obtained by applying the G0W0 method along with plasmon-pole approximation.30,31 We used 982 unoccupied bands, 70 Ry cutoff energy for the dielectric matrix, response cutoffs of 12 Ry, and box-shaped truncation of 30 a.u. to obtain the converged dielectric function, where the convergence of the QP bandgap was within 50 meV. The excitonic effects have been included with a fine k-point grid of 36 × 36 × 1 by solving the Bethe-Salpeter equation (BSE):32,33
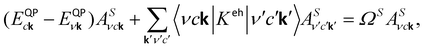 | (2) |
3 Results and discussion
3.1 Structure and electronic properties
The isolated GaN and MoSi2P4 monolayers are first investigated by their crystals and band structures, as shown in Fig. S1 of ESI.† The optimized lattice constants (a/b) for the hexagonal GaN and MoSi2P4 monolayers are 3.201 Å and 3.468 Å, respectively. The MoSi2P4 monolayer is predicted to have a direct bandgap of about 0.685 eV by applying the PBE method, which is further increased to 1.335 eV using the G0W0 method (as shown in Fig. S2†). These calculation results align well with previously reported theoretical predictions.36–39 The calculated phonon dispersions of GaN and MoSi2P4 monolayers can be found in Fig. S3 of the ESI,† in which they are suggested to be dynamically stable owing to no negative phonon frequency. Previously, their thermal stabilities were verified at a high temperature of 1000 K for GaN monolayer40 (700 K for MoSi2P4 monolayer41).Next, six possible high-symmetry stacking patterns are considered AA1–AA6 configurations, and their optimized structures are depicted in Fig. 1. These stacking configurations have been estimated by their binding energies (Eb), which can judge their energetic stabilities, thus pointing out the feasibility of experimental preparation. The binding energy can be calculated using the following equation: Eb = (EGaN/MoSi2P4 − EGaN − EMoSi2P4)/S, where EGaN/MoSi2P4, EGaN, and EMoSi2P4 are total energies of GaN/MoSi2P4 heterobilayer, GaN monolayer, and MoSi2P4 monolayer, respectively. S is the interface area of the GaN/MoSi2P4 heterobilayer. The obtained Eb values are presented in Fig. 2a, in which the Eb values are all negative and limited in the energy range of −12 to −18 meV Å−2. The estimated binding energies of GaN/MoSi2P4 heterobilayers are comparable to those (−15 to −20 meV Å−2) of typical vdWs layered materials,42 indicating that the interlayer interaction between GaN and MoSi2P4 layers belongs to the vdWs force. In particular, the AA5 configuration with a lattice constant of 3.407 Å and an interlayer distance of 3.429 Å provides the lowest binding energy of approximately −17 meV Å−2, suggesting that it is the most favorable energetically and can be fabricated experimentally. Fig. 2b shows the phonon spectra of the AA5 configuration, in which no imaginary frequency is found, suggesting its dynamical stability. In addition, the thermal stability of AA5 GaN/MoSi2P4 is confirmed by the AIMD simulations, as depicted in Fig. 3. It can be found that the structure is intact at a temperature of 400 K. Therefore, the subsequent discussions are mainly focused on GaN/MoSi2P4 heterobilayer with AA5 configuration.
 | ||
| Fig. 1 Top and side views of optimized configurations for GaN/MoSi2P4 heterobilayers with six stacking patterns. | ||
 | ||
Fig. 3 Variable total energy and temperature of the AA5 GaN/MoSi2P4 heterosbilayer during ab initio molecular dynamics heating 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 fs at 400 K. 000 fs at 400 K. | ||
For GaN/MoSi2P4 heterobilayer with AA5 configuration, the projected band structure obtained by the PBE method is shown in Fig. 4a. It can be found that the band structure of every monolayer is retained perfectly, further implying that the interface of the heterostructure is supported by the weak vdWs interaction. Additionally, AA5 exhibits a direct bandgap of about 0.825 eV at the PBE level, which shows that the bandgap (0.685 eV) of the isolated MoSi2P4 monolayer can be improved by forming the vdWs heterobilayer. The valence band maximum (VBM) and conduction band minimum (CBM) of the AA5 GaN/MoSi2P4 heterobilayer are at the K point, identical to the isolated MoSi2P4 monolayer. Fig. 4a shows that both the VBM and CBM originate from the MoSi2P4 layer, which is further verified by the calculated partial charge distributions for the VBM and CBM (see the insets in Fig. 4b). This suggests that the AA5 configuration is a typical type-I vdWs heterostructure, which makes it suitable for light-emitting applications. Additionally, the differential charge density along the c direction is calculated, as shown in Fig. S4,† where the accumulated and depleted electrons are suggested via yellow and cyan, respectively. It can be found that the electron is gathered near the MoSi2P4 layer, while the GaN layer loses the electron.
In addition, the spin–orbit coupling effects on the band structure of the AA5 configuration are checked owing to the heavier Mo atom, as shown in Fig. S5.† The bandgap decreases to 0.741 eV at the PBE level, which is attributed to the spin splitting of the VBM at the K point. The spin splitting Δsoc of the VBM is approximately 87 meV, while that of the CBM is only 4 meV. Previously, the MoS2 monolayer has illustrated that the Δsoc of the VBM is 146 meV.43 The spin splitting of the VBM from the Mo atom can induce the splitting of the lowest-energy peak in the optical absorption spectrum; however, they do not influence the whole optical absorption of the MoS2 monolayer.43,44 Therefore, the spin–orbit coupling effects are not included in the following calculations. We also calculated the quasi-particle (QP) band structure of the AA5 configuration by applying the G0W0 method (Fig. 4b), in which the band structure is acceptable considering only electron–electron interactions. Under the G0W0 approximation, the PBE bandgap of 0.825 eV is corrected to 1.479 eV. The more accurate quasi-particle bandgap of 1.479 eV together with its direct nature indicates that the AA5 GaN/MoSi2P4 heterobilayer has potential in optoelectronic applications.
In addition, it is noteworthy that the two highest valence bands at the K point are very close and the second valence band originates from the GaN layer (Fig. 4a). There is only an energy difference of 63 meV between the two valence bands, suggesting that the two valence bands compete under an external factor, such as in-plane strain. In the experiment, the in-plane tensile strain on the 2D materials can be achieved easily compared to the in-plane compressive strain.45,46 Thus, we explore only the effects of biaxial tensile strain on the valence band edge of the AA5 configuration. The calculated quasi-particle band structures of the AA5 configuration under different biaxial tensile strains (δ) are provided in Fig. 5a–c. As the applied tensile strain increases, the valence band edge changes. When the δ is increased to +1%, the GaN and MoSi2P4 layers contribute to the VBM and CBM, respectively, as shown in the inset of Fig. 5a. This illustrates that the band alignment in the AA5 GaN/MoSi2P4 heterobilayer is transformed from type-I to type-II at δ = +1%. Additionally, the type-II band alignment and direct nature of the bandgap can be retained within the examined strain range from +1% to +5% for AA5 GaN/MoSi2P4. The predicted quasi-particle bandgap decreases linearly from 1.325 eV (δ = +1%) to 0.751 eV (δ = +5%) with the increased tensile strains. Although the PBE method underestimates the bandgap of the AA5 GaN/MoSi2P4, the changing trend in the bandgap seems to be the same as the G0W0 method in the observed strain range of 0–+5% (Fig. 5d). In addition, the phonon dispersions of AA5 GaN/MoSi2P4 at +5% tensile strain are calculated, and the calculated results are shown in Fig. S6.† No negative frequency is found, suggesting that the strained structure is still stable dynamically.
The formed type-II heterostructure usually conforms to the double-transfer mode or the Z-scheme mode under illumination, as illustrated in Fig. S7 of the ESI.† To confirm the mode in the strained GaN/MoSi2P4, we perform a Bader's charge calculation in AA5 GaN/MoSi2P4. The calculation result shows that the electron of about 9.07 × 10−3e near the interface is transferred from the GaN layer to the MoSi2P4 layer at a biaxial tensile strain of +1%, which is less than 1.12 × 10−2e of the pristine structure. The direction of charge transfer is unchanged in the observed strain range of 0–+5%, and the transferred charge is reduced to 8.53 × 10−3e (+5%). In fact, the electron transfer at the interface leaves some holes at the GaN layer, causing an electric polarized field (E1). The direction of the E1 is pointed from the GaN layer to the MoSi2P4 layer. Once the light illuminates the AA5 heterobilayer, the electric polarized field induced by the charge transfer can hold back the transfers of the photogenerated electrons of the GaN layer and the hole of the MoSi2P4 layer by the reverse electric field force. However, this can increase the interface e–h recombination between the photogenerated electron from the MoSi2P4 layer and the photogenerated hole from the GaN layer, which should be attributed to the short electron-migration distance between the GaN and MoSi2P4 layers, thus forming a direct Z-scheme in the strained AA5 GaN/MoSi2P4 heterobilayer. Here, we show only the Z-scheme GaN/MoSi2P4 at the biaxial tensile strain of +1% due to the similar case at other observed strains, as shown in Fig. 6, in which the transfer processes of photogenerated electrons and holes can be understood clearly under the illumination.
 | ||
| Fig. 6 Transfer processes of photogenerated electrons and holes in a direct Z-scheme AA5 GaN/MoSi2P4 heterobilayer at a biaxial tensile strain of +1%. | ||
It is also found that the interlayer bandgaps of the Z-scheme AA5 GaN/MoSi2P4 under different tensile strains are all smaller than the intralayer bandgaps in the GaN and MoSi2P4 layers, as illustrated in Fig. S8 of the ESI.† With increased tensile strains, the interlayer bandgap is greatly reduced from 1.325 eV (δ = 0) to 0.751 (δ = +5%). This indicates that the interlayer e–h recombination is much faster than that of the intralayer with the increased strains. When interlayer e–h recombination occurs, the excess electrons and holes can gather on the CBM of the GaN layer and VBM of the MoSi2P4 layer, respectively. This direct Z-scheme in AA5 GaN/MoSi2P4 is formed by a more positive potential of the VBM and a more negative potential of the CBM, achieving a strong oxidation and reduction ability. Taking the vacuum level as a reference, the predicted CBM of the GaN layer and the VBM of the MoSi2P4 layer in the strained systems are located at about −4.024/−3.856/−3.824 eV and −5.977/−5.949/−5.920 eV for the +1%/+3%/+5% tensile strain, respectively. In particular, the case of +1% is depicted in Fig. 6. Two band edges under the observed tensile strains can well straddle the redox potentials of water at pH = 0 (reduction/oxidation potential: −4.440/−5.670 eV), satisfying the thermodynamic conditions for overall water splitting. The pH-dependent water redox potentials (−4.440 + 0.059 × pH; −5.670 + 0.059 × pH)47,48 yield a reduction/oxidation potential of −4.027/−5.257 eV at pH = 7, indicating that the AA5 GaN/MoSi2P4 heterobilayer in the strain range of +1%–+5% can also act as a water-splitting photocatalyst in a neutral environment.
In photocatalytic water splitting, the magnitude of the external potentials generated by photoexcited carriers directly dictates whether the half-reactions for water splitting can proceed spontaneously. The external potential (Ue) for the hydrogen reduction reaction via photoexcited electrons is identified as 0.416/0.584/0.616 V for the +1%/+3%/+5% biaxial tensile strain at pH = 0. The energy disparity between the CBM of GaN and the hydrogen reduction potential serves to define Ue. Conversely, Uh, which represents the external potential of photoexcited holes in water oxidation, is characterized by the energy differential between the VBM of the MoSi2P4 layer and the hydrogen reduction potential.49 It is found to be 1.537/1.509/1.480 V for the +1%/+3%/+5% biaxial tensile strain at pH = 0. The calculated values of Uh and Ue may generate adequate driving forces to facilitate both the oxygen evolution reaction (OER) and hydrogen evolution reaction (HER). Furthermore, this suggests that the photoexcited electrons on the GaN and photoexcited holes on the MoSi2P4 would preferentially migrate to react with water rather than with themselves, thus generating significant resistance to incident light-induced corrosion.49,50 These results indicate that the hydrogen and oxygen evolution reaction can happen readily and be separated naturally from different layers. Thus, the strained GaN/MoSi2P4 heterobilayer can act as a direct Z-scheme high-efficiency photocatalyst for water splitting to H2 and O2.
3.2 Overall water splitting
For observed AA5 GaN/MoSi2P4 heterobilayer under biaxial tensile strains of +1%–+5%, the first requisite for overall water splitting is acquired by their suitable band edge positions. Nevertheless, it remains unclear whether the photoexcited electrons and holes in the strained AA5 GaN/MoSi2P4 possess sufficient driving force to facilitate the overall water splitting process, specifically the OER and HER. To make it clear, we take AA5 GaN/MoSi2P4 under a biaxial tensile strain of +1%, for example, and explore the OER and HER mechanisms to confirm the photocatalytic activity.The OER on the surface of the MoSi2P4 layer is first investigated by following the four-electron (4e−) reaction pathway:
| * + H2O → OH* + H+ + e−, | (3) |
| OH* → O* + H+ + e−, | (4) |
| O* + H2O → O*OH* + H+ + e−, | (5) |
| O*OH* → O*O* + H+ + e−, | (6) |
| O*O* → O2, | (7) |
Unlike water oxidation, the active site for the HER is the N site of the GaN layer. The distinct active sites for the two half reactions can markedly enhance the efficiency of photocatalytic processes. In the absence of light stimulation (U = 0), the GaN layer shows a thermodynamically favorable ΔG of −0.679 eV for the production of H* species, following the reaction pathway: * + H+ + e− → H*, suggesting an exothermic process that can proceed spontaneously under dark conditions. The process of creating H2 from H* species is endothermic at pH = 0 (H* + H+ + e− → H2 + *), as shown in Fig. 6d. Under an external potential Ue of 0.416 V provided by the photogenerated electrons of the heterobilayer, the free-energy profile for the hydrogen reduction reaction exhibits a downhill trajectory for both elementary steps. Consequently, the +1% strained GaN/MoSi2P4 heterobilayer can efficiently split pure water when exposed to light, as both OER and HER can proceed.
3.3 Optical properties
An efficient photocatalyst should possess a strong light-harvesting performance for the incident sunlight, especially the infrared and visible lights (accounting for ∼93% of the sunlight). The optical properties of 2D materials are significantly influenced by electron–hole couplings primarily due to the substantial effects of excitons. To elucidate the light-harvesting efficacy, an analysis of the optical characteristics of the AA5 GaN/MoSi2P4 heterobilayer is conducted under different biaxial tensile strains (0, +1%, +3%, and +5%) by applying the state-of-the-art G0W0+BSE method. Fig. 8a depicts the imaginary component ε2(ω) of frequency-dependent dielectric function ε(ω) of the pristine AA5 GaN/MoSi2P4, where the G0W0+BSE (G0W0+RPA) curve corresponds to an optical absorption spectrum, including (excluding) the photogenerated e–h couplings. Compared with the G0W0+RPA profile, the G0W0+BSE profile is largely red-shifted due to the outstanding e–h couplings, in which a large number of exciton states with large oscillator strengths are observed in the energy region of 0.5–3.16 eV. In particular, two bright bound exciton states below the QP bandgap (1.479 eV) are located at 0.951 and 1.282 eV (as shown in the orange arrows in Fig. 8a). For a direct semiconductor, the first bright exciton state is very important and can be captured by experimental observation. By analysing the oscillator strength, the first bright bound exciton state is from the VBM → CBM vertical transition at the K-point (Fig. 4b). Thus, the excitation energy of 0.951 eV is regarded as the optical gap of pristine AA5 GaN/MoSi2P4, which can be compared with the experimentally measured gap. Further, the binding energy (Eb-exciton) of the first bright bound exciton is estimated to be 0.528 eV by the difference between the QP bandgap (Eg-QP) and excitation energy (Ee), i.e. Eb-exciton = Eg-QP − Ee, which can resist thermal fluctuation at room temperature. The calculated exciton binding energy of pristine AA5 GaN/MoSi2P4 is compared to those of other 2D materials.49,51Fig. 8b shows the strain-dependent optical absorptions of AA5 GaN/MoSi2P4, in which the ε2(ω) is observed at the strains of δ = +1%, +3%, +5%. The focused first absorption peak is significantly red-shifted by about 0.463 eV from 0.951 eV (δ = 0) to 0.492 eV (δ = +5%). The first absorption peaks of the strained systems are also examined and verified to be from the VBM → CBM vertical transitions at the K-point. The estimated Eb-exciton corresponding to the first absorption peaks is changed from 528 meV from δ = 0 to 259 meV for δ = +5%, as listed in Table 1, indicating that the binding energy of exciton in the AA5 GaN/MoSi2P4 can be tuned significantly by the applied biaxial strains. Smaller Eb-exciton can result in the photogenerated electron around the hole delocalized in a larger space, thus making the photogenerated e–h pair recombination difficult. In addition, compared with that at δ = 0, the optical absorption of AA5 GaN/MoSi2P4 under the biaxial tensile strain of +5% reveals a significant enhancement in the near-infrared and visible range. Consequently, the biaxial tensile strain in the AA5 GaN/MoSi2P4 heterobialyer can induce smaller exciton binding energy and enhance light absorption.
| δ | E g-PBE | E g-QP | E e | E b-exciton |
|---|---|---|---|---|
| 0 | 0.825 | 1.479 | 0.951 | 0.528 |
| +1% | 0.688 | 1.325 | 0.810 | 0.515 |
| +3% | 0.350 | 1.024 | 0.644 | 0.380 |
| +5% | 0.101 | 0.751 | 0.492 | 0.259 |
Finally, to further measure the light harvesting ability of the AA5 GaN/MoSi2P4 heterobilayer for the sunlight, we calculated its light absorption coefficient α(ω) by applying the following equation:52,53
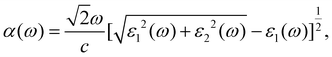 | (8) |
4 Conclusions
In summary, by employing DFT+G0W0+BSE calculations, this study systematically investigates the structural, electronic, photocatalytic, and optical properties of a new GaN/MoSi2P4 van der Waals heterobilayer. The dynamical, energetic, and thermodynamic stabilities are verified by the phonon spectrum, binding energy, and AIMD. The GaN/MoSi2P4 with type-I band alignment is predicted to have a direct quasi-particle bandgap of 1.479 eV at the G0W0 level. It also exhibits a strong absorption coefficient of more than 106 cm−1 for visible and near-infrared lights. The calculated binding energy of exciton in the GaN/MoSi2P4 is 528 meV, which can withstand thermal fluctuation at room temperature. More interestingly, a +1% biaxial tensile strain can transform the GaN/MoSi2P4 from type-I to type-II. The type-II nature in the strained GaN/MoSi2P4 can facilitate the effective separation of photogenerated electron–hole pairs. The observed built-in electric field oriented from the GaN layer to the MoSi2P4 layer further indicates that it constitutes a direct Z-scheme heterostructure. The formed Z-scheme can greatly optimize the redox ability. Additionally, the distinct layers of GaN/MoSi2P4 facilitate both water oxidation and hydrogen reduction half-reactions, which are driven by the potentials generated from photoexcited electrons and holes. With the increase in applied biaxial tensile strain, the quasi-particle bandgap is significantly reduced to 0.751 eV (+5%) and significantly enhanced absorptions for the visible and near-infrared lights are observed. These tunable properties in the GaN/MoSi2P4 vdWs heterobilayer can endow it as a potential material for optoelectronic and photocatalytic water splitting devices.Data availability
The data supporting this work are available from the corresponding author upon reasonable request.Conflicts of interest
There are no conflicts to declare.Acknowledgements
This work was supported by the foundations from Jiangsu University of Science and Technology (No. 1052931610), Natural Science Foundation of Jiangsu Province of China (BK20220407), Natural Science Foundation of China (No. 12404273), and China Postdoctoral Fund Project (2024M750835, 2024T170251).References
- X. Li, L. Tao, Z. Chen, H. Fang, X. Li, X. Wang, J.-B. Xu and H. Zhu, Appl. Phys. Rev., 2017, 4, 021306 Search PubMed.
- C. Chang, W. Chen, Y. Chen, Y. Chen, Y. Chen, F. Ding, C. Fan, H. Jin Fan, Z. Fan, C. Gong, Y. Gong, Q. He, X. Hong, S. Hu, W. Hu, W. Huang, Y. Huang, W. Ji, D. Li, L.-J. Li, Q. Li, L. Lin, C. Ling, M. Liu, N. Liu, Z. Liu, K. Ping Loh, J. Ma, F. Miao, H. Peng, M. Shao, L. Song, S. Su, S. Sun, C. Tan, Z. Tang, D. Wang, H. Wang, J. Wang, X. Wang, X. Wang, A. T. S. Wee, Z. Wei, Y. Wu, Z.-S. Wu, J. Xiong, Q. Xiong, W. Xu, P. Yin, H. Zeng, Z. Zeng, T. Zhai, H. Zhang, H. Zhang, Q. Zhang, T. Zhang, X. Zhang, L.-D. Zhao, M. Zhao, W. Zhao, Y. Zhao, K.-G. Zhou, X. Zhou, Y. Zhou, H. Zhu, H. Zhang and Z. Liu, Acta Phys.-Chim. Sin., 2021, 37, 2108017 Search PubMed.
- Y. Yin, Q. Gong, M. Yi and W. Guo, Adv. Funct. Mater., 2023, 33, 2214050 CrossRef CAS.
- B. Cho, J. Yoon, S. K. Lim, A. R. Kim, D.-H. Kim, S.-G. Park, J.-D. Kwon, Y.-J. Lee, K.-H. Lee, B. H. Lee, H. C. Ko and M. G. Hahm, ACS Appl. Mater. Interfaces, 2015, 7, 16775–16780 CrossRef CAS.
- N. Zhang, A. Surrente, M. Baranowski, D. K. Maude, P. Gant, A. Castellanos-Gomez and P. Plochocka, Nano Lett., 2018, 18, 7651–7657 CrossRef CAS PubMed.
- B. Du, Y. Li, M. Jiang, H. Zhang, L. Wu, W. Wen, Z. Liu, Z. Fang and T. Yu, Nano Lett., 2022, 22, 1649–1655 CrossRef CAS PubMed.
- M. Yankowitz, Q. Ma, P. Jarillo-Herrero and B. J. LeRoy, Nat. Rev. Phys., 2019, 1, 112–125 CrossRef CAS.
- H. Wang, Z. Li, D. Li, P. Chen, L. Pi, X. Zhou and T. Zhai, Adv. Funct. Mater., 2021, 31, 2103106 CrossRef CAS.
- E. Barré, M. Dandu, S. Kundu, A. Sood, F. H. da Jornada and A. Raja, Nat. Rev. Mater., 2024, 9, 499–508 CrossRef.
- R. Zhang, L. Zhang, Q. Zheng, P. Gao, J. Zhao and J. Yang, J. Phys. Chem. Lett., 2018, 9, 5419–5424 CrossRef CAS PubMed.
- Y.-L. Hong, Z. Liu, L. Wang, T. Zhou, W. Ma, C. Xu, S. Feng, L. Chen, M.-L. Chen, D.-M. Sun, X.-Q. Chen, H.-M. Cheng and W. Ren, Science, 2020, 369, 670–674 CrossRef CAS.
- H. Yao, C. Zhang, Q. Wang, J. Li, Y. Yu, F. Xu, B. Wang and Y. Wei, Nanomaterials, 2021, 11, 559 CrossRef CAS.
- X. Liu, H. Zhang, Z. Yang, Z. Zhang, X. Fan and H. Liu, Phys. Lett. A, 2021, 420, 127751 CrossRef CAS.
- Q. H. Wang, K. Kalantar-Zadeh, A. Kis, J. N. Coleman and M. S. Strano, Nat. Nanotechnol., 2012, 7, 699–712 CrossRef CAS PubMed.
- Y. Wang and Y. Ding, Appl. Phys. Lett., 2022, 121, 073101 CrossRef CAS.
- T. K. Sahu, S. P. Sahu, K. P. S. S. Hembram, J.-K. Lee, V. Biju and P. Kumar, NPG Asia Mater., 2023, 15, 49 CrossRef CAS.
- Z. Y. Al Balushi, K. Wang, R. K. Ghosh, R. A. Vilá, S. M. Eichfeld, J. D. Caldwell, X. Qin, Y.-C. Lin, P. A. DeSario, G. Stone, S. Subramanian, D. F. Paul, R. M. Wallace, S. Datta, J. M. Redwing and J. A. Robinson, Nat. Mater., 2016, 15, 1166 CrossRef CAS PubMed.
- H. Shu, X. Niu, X. Ding and Y. Wang, Appl. Surf. Sci., 2019, 479, 475–481 CrossRef CAS.
- W.-J. Yin, X.-L. Zeng, B. Wen, Q.-X. Ge, Y. Xu, G. Teobaldi and L.-M. Liu, Front. Phys., 2020, 16, 33501 CrossRef.
- J. Wang, H. Shu, P. Liang, N. Wang, D. Cao and X. Chen, J. Phys. Chem. C, 2019, 123, 3861–3867 CrossRef CAS.
- Z. Cui, K. Ren, Y. Zhao, X. Wang, H. Shu, J. Yu, W. Tang and M. Sun, Appl. Surf. Sci., 2019, 492, 513–519 CrossRef CAS.
- N. B. Singh, R. Mondal, J. Deb, D. Paul and U. Sarkar, ACS Appl. Nano Mater., 2024, 7, 6704–6711 CrossRef CAS.
- P. Giannozzi, S. Baroni, N. Bonini, M. Calandra, R. Car, C. Cavazzoni, D. Ceresoli, G. L. Chiarotti, M. Cococcioni, I. Dabo, A. Dal Corso, S. de Gironcoli, S. Fabris, G. Fratesi, R. Gebauer, U. Gerstmann, C. Gougoussis, A. Kokalj, M. Lazzeri, L. Martin-Samos, N. Marzari, F. Mauri, R. Mazzarello, S. Paolini, A. Pasquarello, L. Paulatto, C. Sbraccia, S. Scandolo, G. Sclauzero, A. P. Seitsonen, A. Smogunov, P. Umari and R. M. Wentzcovitch, J. Phys.: Condens. Matter, 2009, 21, 395502 CrossRef PubMed.
- J. P. Perdew, K. Burke and M. Ernzerhof, Phys. Rev. Lett., 1996, 77, 3865–3868 CrossRef CAS.
- G. Román-Pérez and J. M. Soler, Phys. Rev. Lett., 2009, 103, 096102 CrossRef.
- S. Baroni, S. de Gironcoli, A. Dal Corso and P. Giannozzi, Rev. Mod. Phys., 2001, 73, 515–562 CrossRef CAS.
- J. K. Nørskov, J. Rossmeisl, A. Logadottir, L. Lindqvist, J. R. Kitchin, T. Bligaard and H. Jónsson, J. Phys. Chem. B, 2004, 108, 17886–17892 CrossRef PubMed.
- Á. Valdés, Z.-W. Qu, G.-J. Kroes, J. Rossmeisl and J. K. Nørskov, J. Phys. Chem. C, 2008, 112, 9872–9879 CrossRef.
- NIST Computational Chemistry Comparison and Benchmark Database, 2018, vol. NIST Standard Reference Database Number 101, Release 19, edited by Russell D. Johnson III, available at https://cccbdb.nist.gov/.
- R. W. Godby and R. J. Needs, Phys. Rev. Lett., 1989, 62, 1169 CrossRef.
- A. Oschlies, R. W. Godby and R. J. Needs, Phys. Rev. B: Condens. Matter Mater. Phys., 1995, 51, 1527 CrossRef CAS PubMed.
- E. E. Salpeter and H. A. Bethe, Phys. Rev., 1951, 84, 1232 CrossRef.
- G. Onida, L. Reining and A. Rubio, Rev. Mod. Phys., 2002, 74, 601–659 CrossRef CAS.
- L. Yang, C. D. Spataru, S. G. Louie and M. Y. Chou, Phys. Rev. B: Condens. Matter Mater. Phys., 2007, 75, 201304 CrossRef.
- A. Marini, C. Hogan, M. Grüning and D. Varsano, Comput. Phys. Commun., 2009, 180, 1392–1403 CrossRef CAS.
- H. Şahin, S. Cahangirov, M. Topsakal, E. Bekaroglu, E. Akturk, R. T. Senger and S. Ciraci, Phys. Rev. B: Condens. Matter Mater. Phys., 2009, 80, 155453 CrossRef.
- B. Mortazavi, B. Javvaji, F. Shojaei, T. Rabczuk, A. V. Shapeev and X. Zhuang, Nano Energy, 2021, 82, 105716 CrossRef CAS.
- Y. Guo, J. Min, X. Cai, L. Zhang, C. Liu and Y. Jia, J. Phys. Chem. C, 2022, 126, 4677–4683 CrossRef CAS.
- M. Sun, M. Re Fiorentin, U. Schwingenschlögl and M. Palummo, npj 2D Mater. Appl., 2022, 6, 81 CrossRef CAS.
- A. Onen, D. Kecik, E. Durgun and S. Ciraci, Phys. Rev. B, 2016, 93, 085431 CrossRef.
- X. Wang, W. Ju, D. Wang, X. Li and J. Wan, Phys. Chem. Chem. Phys., 2023, 25, 18247–18258 RSC.
- T. Björkman, A. Gulans, A. V. Krasheninnikov and R. M. Nieminen, Phys. Rev. Lett., 2012, 108, 235502 CrossRef PubMed.
- A. Ramasubramaniam, Phys. Rev. B: Condens. Matter Mater. Phys., 2012, 86, 115409 CrossRef.
- K. F. Mak, C. Lee, J. Hone, J. Shan and T. F. Heinz, Phys. Rev. Lett., 2010, 105, 136805 CrossRef.
- M. Pandey, C. Pandey, R. Ahuja and R. Kumar, Nano Energy, 2023, 109, 108278 CrossRef CAS.
- S. Yang, Y. Chen and C. Jiang, InfoMat, 2021, 3, 397–420 CrossRef.
- V. Chakrapani, J. C. Angus, A. B. Anderson, S. D. Wolter, B. R. Stoner and G. U. Sumanasekera, Science, 2007, 318, 1424–1430 CrossRef CAS.
- Y. Xu and M. A. A. Schoonen, Am. Mineral., 2000, 85, 543–556 CrossRef CAS.
- M. Qiao, J. Liu, Y. Wang, Y. Li and Z. Chen, J. Am. Chem. Soc., 2018, 140, 12256–12262 CrossRef CAS PubMed.
- S. Chen and L.-W. Wang, Chem. Mater., 2012, 24, 3659–3666 CrossRef CAS.
- H. Seksaria, A. Kishore and A. De Sarkar, Phys. Chem. Chem. Phys., 2024, 26, 22882–22893 RSC.
- F. Karlický and J. Turoň, Carbon, 2018, 135, 134–144 CrossRef.
- X. Liu, P. Gao, W. Hu and J. Yang, J. Phys. Chem. Lett., 2020, 11, 4070–4079 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: https://doi.org/10.1039/d4nr04545a |
| This journal is © The Royal Society of Chemistry 2025 |






