Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Synthesis of a pyrroloquinoline-containing polycyclic scaffold via decarboxylative [3+2] cycloaddition and aza-Wittig reactions
Lee
Price
,
Zonyuan
Li
and
Wei
Zhang
 *
*
Department of Chemistry and Centre for Green Chemistry, University of Massachusetts Boston, 100 Morrissey Boulevard, Boston, MA 02125, USA. E-mail: lee.price0214@gmail.com; wei2.zhang@umb.edu
First published on 1st October 2025
Abstract
A two-step reaction process involving decarboxylative [3+2] cycloaddition followed by the intramolecular aza-Wittig reaction is developed for making novel 5,5,6,6-fused polycyclic scaffold-containing pyrroloquinolines. The use of polymer-supported Ph3P in the aza-Wittig reaction simplified the removal of Ph3P(O) and reduced the loss of product during purification.
Introduction
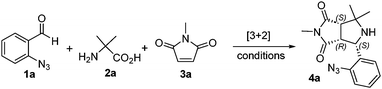
Pyrroloquinoline is a tricyclic system that can be found in alkaloids1,2 and synthetic drug molecules such as Blebbistatin.3a Pyrroloquinoline-derived molecules containing a maleimide moiety have been used in the development of antibody–drug conjugates (ADCs)3b (Fig. 1).Literature methods for making pyrroloquinolines and associated polycyclic systems include the Partyka group's six-step synthesis4 of pyrrololquinoline (Scheme 1a) and the Smalley group's method of using pre-assembled substrates for the aza-Witting reaction for the synthesis of 7H-benzo[4,5]isothiazolo[3,2-b]quinazolin-7-one-5,5-dioxide5 and other substituted dihydroisoquinolines (Scheme 1b). We have employed decarboxylative [3+2] cycloaddition adducts for the N-acylation and aza-Wittig reactions to make tetrahydropyrrolo[1,2-c]quinazolines (Scheme 1c).6 Reported in this paper is our effort to expand the scope of the intramolecular aza-Wittig reaction for making the dipyrrolo-dihydroisoquinoline ring system (Scheme 1Td).
Results and discussion
The synthesis of [3+2] adducts 4 was conducted by modifying reported procedures.6–14 Thus, a three-component reaction of 2-azidobenzaldehyde 1a, 2-amino-2-methylpropanoic acid 2a, and N-methyl maleimide 3a was optimized by exploring different solvents, reaction temperatures, heating methods and times (Table 1). It was found that the reaction of 1a, 2a, and 3a in the ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.2
1.2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.1 in MeCN and under microwave heating at 150 °C for 60 min gave product 4a in 68% isolated yield.
1.1 in MeCN and under microwave heating at 150 °C for 60 min gave product 4a in 68% isolated yield.
| Entryd | Solvent | T (°C) | Heating | t (min) | Yieldb (%) |
|---|---|---|---|---|---|
a 0.3 mmol scale; 1a![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2a 2a![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3a (1 3a (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.2 1.2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.1).
b Isolated yield.
c 110 °C oil bath.
d Reaction conditions for entries 1–3, 5 and 6: 0.30 mmol azidobenzaldehyde, 0.36 mmol 2-amino-2-methylpropanoic acid, and 0.33 mmol N-methyl maleimide. Entry 4: 0.5 mmol azidobenzaldehyde, 0.60 mmol 2-amino-2-methylpropanoic acid, and 0.55 mmol N-methyl maleimide. 1.1).
b Isolated yield.
c 110 °C oil bath.
d Reaction conditions for entries 1–3, 5 and 6: 0.30 mmol azidobenzaldehyde, 0.36 mmol 2-amino-2-methylpropanoic acid, and 0.33 mmol N-methyl maleimide. Entry 4: 0.5 mmol azidobenzaldehyde, 0.60 mmol 2-amino-2-methylpropanoic acid, and 0.55 mmol N-methyl maleimide.
|
|||||
| 1 | DMF | 150 | μw | 15 | 19 |
| 2 | THF | 150 | μw | 60 | 21 |
| 3 | DMAc | 150 | μw | 60 | 36 |
| 4 | MeCN + AcOH | 110 | Conventionalc | 360 | 63 |
| 5 | MeCN + AcOH | 150 | μw | 30 | 40 |
| 6 | MeCN | 150 | μw | 60 | 68 |
This kind of [3+2] cycloaddition is highly diastereoselective, giving products 4 as single diastereomers if R2 has two of the same groups.15–18 The stereochemistry of 4a was confirmed by 1H–1H NOESY, 1H–1H COSY, 1H–13C HMBC, and X-ray single crystal structure analysis. The X-ray single crystal structure for 4f was also obtained. In total, 14 analogs of [3+2] adducts were prepared by using five azidobenzaldehydes 1, six amino acids 2, and seven maleimides (including maleic anhydride) 3. The yields of [3+2] cycloaddition products 4a–4r were in the range of 50–71% (Scheme 2).
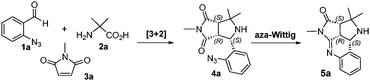
The aza-Wittig reaction was first attempted by performing a one-pot synthesis of [3+2] cycloaddition to make 4a followed by direct addition of Ph3P or n-Bu3P to the reaction mixture for the aza-Wittig reaction to make 5a. However, the yields of the products were 29% and 13%, respectively. Water generated from the [3+2] cycloaddition reaction could hydrolyze Ph3P and n-Bu3P and interrupted the reaction (Table 2). To address this issue, 4a was isolated and used for the aza-Wittig reaction. The reaction proceeded well, but the removal of phosphine oxide by ZnCl2 working up before flash column chromatography purification caused product decomposition and reduced the yield to <45%.18 Using polymer-supported Ph3P (PS-Ph3P) for the aza-Wittig reaction allowed the removal of PS-Ph3P(O) by filtration and avoided the step of ZnCl2 workup. The aza-Wittig reaction was then optimized by evaluating the amount of PS-Ph3P used, solvent, and other reaction conditions. It was found that the reaction of 5a with 2.4 equiv. of PS-Ph3P in MeCN under microwave heating at 110 °C for 1 h afforded product 5a in 90% yield (Table 2, entry 9).
| Entry | R3P (equiv.) | Condition | Solventb | Yieldc (%) |
|---|---|---|---|---|
| a 0.102 mmol scale of 4a. PS-Ph3P: 0.24 mmol: ∼3 mmol g−1 loading, ∼76% (w/w), 84.5 mg, Conditions: μw 110 °C, 1 h. b For the aza-Wittig reaction. c Isolated yield. | ||||
| 1 | Ph3P, 2.4 | One-pot | DMF | 29 |
| 2 | n-Bu3P, 2.4 | One-pot | DMF | 13 |
| 3 | PS-Ph3P, 2.4 | One-pot | MeCN | 50 |
| 3 | Ph3P, 2.4 | Stepwise | MeCN | 45 |
| 4 | n-Bu3P, 2.4 | Stepwise | MeCN | 41 |
| 5 | PS-Ph3P, 1.2 | Stepwise | DMF | 21 |
| 5 | PS-Ph3P, 2.4 | Stepwise | DMF | 38 |
| 6 | PS-Ph3P, 2.4 | Stepwise | DMAc | Trace |
| 7 | PS-Ph3P, 2.4 | Stepwise | THF | 32 |
| 8 | PS-Ph3P, 2.4 | Stepwise | Toluene | 42 |
| 9 | PS-Ph 3 P, 2.4 | Stepwise | MeCN | 90 |
Under the optimized aza-Wittig conditions, reactions of 4a–4r were conducted to explore the substrate scope and make analogs of product 5. The results presented in Table 3 indicate that the products were generated in up to 90% yield. The R1 group on azidobenzaldehydes has no significant impact on the product yields. For the R2 group on the amino acids, most products with R2 as dimethyl were generated in good yields. R2 is a single group leading to the formation of products 5m, 5n, 5p, and 5q as diastereomers. The yield of product 5o, which has no substituent group at R2, was only 25%. The R3 on maleimides seems to have more influence on the product yields. If R3 is a Me, Et, Pr, Cy, or Bn group, the products were formed in >50% yields. When R3 was Ph, the yield of product 5h was slightly lower (46%). With more hindered t-Bu as R3, product 5f was obtained in only 10% yield. Another interesting discovery was that the reaction of 4r, which was derived from maleic anhydride, gave product 5r, which was not stable enough to be isolated. A major by-product observed is the amine formation of the Staudinger reaction from hydrolysis of the imine, which would be the amine derivative of 4, which arose from a trace amount of water from the solvents used.
| a PS-Ph3P (2.4 equiv.) ∼3.0 mmol g−1 loading, 0.1–0.38 mmol scale of 4. |
|---|

|
To determine the stereochemistry of the aza-Wittig products, representative product 5a was subjected to 2D-NMR (HSQC, COSY, NOSEY, and HMBC) analysis (Fig. 2). The single crystal X-ray diffraction of 5a was obtained for structure confirmation. Likewise, X-ray crystal structure determination for products 5c and 5m was also conducted.
 | ||
| Fig. 2 1H–1H NOESY (purple), 1H–1H COSY (red), and 1H–13C HMBC (blue) NMRs and X-ray structure of 5a. | ||
The proposed reaction mechanism for the sequential decarboxylative [3+2] cycloaddition and aza-Wittig reactions is shown in Scheme 3. The condensation of azidobenzaldehyde 1 and α-amino acid 2 gives imine, which undergoes cyclization to form oxazolidinone. Decarboxylation of oxazolidinone gives the azomethine ylide, which undergoes 1,3-dipolar cycloaddition with maleimide 3 to form [3+2] adduct 4 stereoselectively.15 Upon addition of Ph3P, phosphazide is generated, which then converts to iminophosphorane after the elimination of N2. The [2+2] cycloaddition reaction of iminophosphorane with the carbonyl group affords oxazaphosphetane, which is followed by the elimination of Ph3(O)P to give aza-Wittig product 5.
Conclusions
In summary, we have developed a reaction sequence involving atom-economic and highly efficient three-component decarboxylative [3+2] cycloaddition for diastereoselective synthesis of precursors 4. The following aza-Wittig reaction of 4 leads to the formation of a pyrroloquinoline-containing 5,5,6,6-fused polycyclic scaffold. The polymer PS-Ph3P is used to simplify the removal of PS-Ph3P(O) and to avoid product decomposition during ZnCl2 workup prior to flash column purification. This is a straightforward and sustainable method for making pyrroloquinoline-containing polycyclic compounds, which may have potential biological interest.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: detailed experimental procedures, compound characterization, and NMR spectra. see DOI: https://doi.org/10.1039/d5nj03128a.CCDC 2450970 (4a), 2450974 (4f), 2450975 (5a), 2450976 (5c) and 2450977 (5n) contain the supplementary crystallographic data for this paper.19a–e
Acknowledgements
This research was supported by the NSF-MRI award CHE-2117246. We thank Jason Evans and Mynaja Ferguson for performing the HRMS analysis of the compounds. We would also like to thank Takeru Saito for his insight in this project.Notes and references
- S. M. Verbitski, C. L. Mayne, R. A. Davis, G. P. Concepcion and C. M. Ireland, J. Org. Chem., 2002, 67, 7124–7126 CrossRef CAS PubMed.
- L. Verotta, T. Pilati, M. Tatò, E. Elisabetsky, T. A. Amador and D. S. Nunes, J. Nat. Prod., 1998, 61, 392–396 CrossRef CAS.
- (a) B. I. Roman, S. Verhasselt and C. V. Stevens, J. Med. Chem., 2018, 61, 9410–9428 CrossRef CAS PubMed; (b) M. S. Kang, T. W. S. Kong, J. Y. X. Khoo and T.-P. Loh, Chem. Sci., 2021, 12, 13613–13647 RSC.
- R. A. Partyka, H. M. Holava and W. N. Beverung, Bristol Myers Co, US3974165A, August 1976.
- A.-B. N. Luheshi, S. M. Salem, R. K. Smalley, P. D. Kennewell and R. Westwood, Tetrahedron Lett., 1990, 31, 6561–6564 CrossRef CAS.
- X. Ma, X. Zhang, J. M. Awad, G. Xie, W. Qiu, R. E. Muriph and W. Zhang, Tetrahedron Lett., 2020, 61, 151392 CrossRef CAS.
- X. Zhang, K. Pham, S. Liu, M. Legris, A. Muthengi, J. P. Jasinski and W. Zhang, Beilstein J. Org. Chem., 2016, 12, 2204–2210 CrossRef CAS PubMed.
- X. Ma, S. Meng, X. Zhang, Q. Zhang, S. Yan, Y. Zhang and W. Zhang, Beilstein J. Org. Chem., 2020, 16, 1225–1233 CrossRef CAS.
- J. Muthengi, R. E. Erickson, R. E. Muriph and W. Zhang, J. Org. Chem., 2019, 84, 5927–5935 CrossRef PubMed.
- X. Ma, X. Zhang, W. Qiu, W. Zhang, B. Wan, J. Evans and W. Zhang, Molecules, 2019, 24, 601 CrossRef PubMed.
- X. Zhang, M. Legris, A. Muthengi and W. Zhang, Chem. Heterocycl. Comp., 2017, 53, 468–473 CrossRef CAS.
- X. Zhang, M. Liu, W. Zhang, M. Legris and W. Zhang, J. Fluorine Chem., 2017, 204, 18–22 CrossRef CAS.
- X. Zhang, X. Ma, W. Qiu, J. Awad and W. Zhang, Green Process. Synth., 2022, 11, 1128–1135 CrossRef CAS.
- X. Ma, W. Qiu, L. Liu, X. Zhang, J. Awad, J. Evans and W. Zhang, Green Synth. Catal., 2021, 2, 74–77 Search PubMed.
- R. Grigg, J. Montgomery and A. Somasunderam, Tetrahedron, 1992, 48, 10431–10442 CrossRef CAS.
- G. Pandey, P. Banerjee and S. R. Gadre, Chem. Rev., 2006, 106, 4484–4517 CrossRef CAS PubMed.
- I. Coldham and R. Hufton, Chem. Rev., 2005, 105, 2765–2810 CrossRef CAS PubMed.
- D. C. Batesky, M. J. Goldfogel and D. J. Weix, J. Org. Chem., 2017, 82, 9931–9936 CrossRef CAS PubMed.
- (a) CCDC 2450970: Experimental Crystal Structure Determination, 2025 DOI:10.5517/ccdc.csd.cc2n8fky; (b) CCDC 2450974: Experimental Crystal Structure Determination, 2025 DOI:10.5517/ccdc.csd.cc2n8fp2; (c) CCDC 2450975: Experimental Crystal Structure Determination, 2025 DOI:10.5517/ccdc.csd.cc2n8fq3; (d) CCDC 2450976: Experimental Crystal Structure Determination, 2025 DOI:10.5517/ccdc.csd.cc2n8fr4; (e) CCDC 2450977: Experimental Crystal Structure Determination, 2025 DOI:10.5517/ccdc.csd.cc2n8fs5.
| This journal is © The Royal Society of Chemistry and the Centre National de la Recherche Scientifique 2025 |