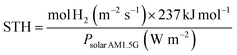Advances and challenges of metal organic frameworks (MOFs) and derivatives in photoelectrocatalytic water splitting
Xin-Jie
Tian
a,
Si-Jia
Guo
a,
Zheng-Yi
Wu
a,
Na
Xu
a,
Guanqun
Han
*b,
Yong-Ming
Chai
a and
Bin
Dong
 *a
*a
aState Key Laboratory of Heavy Oil Processing, College of Chemistry and Chemical Engineering, China University of Petroleum (East China), Qingdao 266580, P. R. China. E-mail: dongbin@upc.edu.cn; Fax: +86-532-86981156; Tel: +86-532-86981156
bSchool of Advanced Energy, Sun Yat-sen University (Shenzhen), Shenzhen 518107, P. R. China. E-mail: hangq5@mail.sysu.edu.cn
First published on 22nd August 2025
Abstract
Photoelectrocatalytic (PEC) hydrogen production technology combines the advantages of photocatalysis and electrocatalysis and utilizes solar energy to drive water splitting, which is a technology for sustainable energy systems. However, its low photocatalytic water splitting efficiency results in relatively small hydrogen production. And the cost-effectiveness of PEC water splitting technology and the overall solar energy conversion efficiency to hydrogen remains a great challenge. Metal–organic frameworks (MOFs) are porous materials created through the coordination of metal ions or clusters with organic bridging ligands via ligand bonds. They offer high specific surface areas, abundant metal active sites, large pore volumes, and customizable structures and compositions, making them highly favorable for applications in photoelectrocatalysis. This review discusses the advancements in photoelectrocatalytic hydrogen production technology using metal–organic frameworks (MOFs) and derivatives. It covers the principles of photoelectrocatalysis, preparation methods for MOF catalysts and strategies for performance enhancement. These strategies include improving light absorption, enhancing carrier separation efficiency, and ensuring stability. The paper also discusses the current challenges and future directions of photoelectrocatalytic water splitting technology. Overall, this review offers a thorough theoretical framework and practical insights for researchers in this field.
Wider impactPhotoelectrocatalytic hydrogen production technology combines the advantages of photocatalysis and electrocatalysis and utilizes solar energy to drive water splitting, which is a technology for sustainable energy systems. However, its low photocatalytic water splitting efficiency results in relatively small hydrogen production. And the cost-effectiveness of PEC water splitting technology and the overall solar energy conversion efficiency to hydrogen remains a great challenge. Metal–organic frameworks (MOFs) are porous materials created through the coordination of metal ions or clusters with organic bridging ligands via ligand bonds, which can promote the development of photoelectrocatalytic hydrogen production technology. |
1. Introduction
Against the backdrop of surging global energy demand and intensifying environmental problems, the development of efficient and sustainable clean energy technologies has become an important issue in the scientific community. Among them, photoelectrocatalytic water splitting technology is considered as one of the key ways to realize a green hydrogen energy economy by solar-driven splitting of water into hydrogen and oxygen. Electrocatalysis has high efficiency and controllability with high energy conversion rates,1–4 but it requires continuous supply of electrical energy, low hydrogen production efficiency, high cost, and complex devices.5–8 As a green technology for energy production and environmental remediation that directly harnesses solar energy, photocatalysis exhibits significant advantages.9–12 However, the rapid recombination of charge carriers during the photocatalytic process leads to low catalytic activity.13–15 In contrast, photoelectrocatalysis integrates the merits of both photocatalysis and electrocatalysis, emerging as a key technology for achieving solar-to-hydrogen energy conversion. It has been successfully applied in hydrogen production, oxygen generation, carbon dioxide reduction, organic compound oxidation, and wastewater treatment.16–19 This approach involves irradiating a semiconductor to generate electron–hole pairs, while simultaneously applying a gradient potential bias to minimize charge carrier recombination. The specific steps and basic principles are systematically described in Part 2. However, photoelectrocatalytic hydrogen production still faces low light absorption efficiency, low charge separation efficiency, and poor catalyst stability in practical applications.20–23 In order to solve these problems, researchers have extensively studied the synthesis, modification and reaction mechanism of photoelectrocatalysts, and significant progress has been achieved.The most commonly used photocatalysts include metal oxides, sulfides, and nitrides.24–28 The limited light-absorbing capacity of metal oxides, the tendency of reaction kinetics to be slow, the ease of photocorrosion and chemical corrosion, and the high cost of preparation have hindered their widespread adoption.29,30 The sensitivity of sulfide catalysts to photodegradation and oxidation leads to instability and rapid deactivation during operation, lower visible light absorption efficiency, and performance can be significantly affected by pH conditions.31,32 Although nitride catalysts have narrow bandgaps, a narrower bandgap is often accompanied by an upward shift in the conduction band (CB) potential (reduced reducing ability) or a downward shift in the valence band (VB) potential (insufficient oxidizing ability);33 narrow bandgaps are more prone to carrier recombination;34 furthermore, the valence band of many narrow bandgap nitrides is primarily composed of N 2p orbitals. Under illumination, the strongly oxidizing holes generated can attack the material's own N3− ions or metal-nitrogen bonds, leading to structural damage (e.g., g-C3N4 + 6h+ → 3C2N2 + N2↑), i.e., the material itself becomes a sacrificial agent for photocatalytic oxidation, resulting in photodegradation and poor stability.35 Synthesis and processing are technically challenging and costly, and nitrides tend to show limited selectivity for hydrogen production, often favoring other by-products, further hampering their efficiency.36–39 Carbon-based materials catalysts tend to have low electrical conductivity and electron transfer efficiency, limiting the overall photocatalytic efficiency. They have short lifetimes under harsh reaction conditions such as high pH or high temperature. The drawbacks of these catalysts limit their wide application.40–42
Metal–organic framework (MOF) materials and their derivatives show great potential in photoelectrocatalysis due to their tunable pore structures, high specific surface areas and abundant active sites, and they are classified as novel materials with excellent photocatalytic functions.43–49 MOFs are formed by the self-assembly of metal nodes with organic ligands via coordination bonds, and their structural flexibility and chemical designability provide unique advantages for optimizing light absorption, charge separation, and surface catalytic activity.50–55
Currently, research on MOF materials is showing two significant trends. On the one hand, efforts are being made to synthesize MOF materials with more complex structures. By precisely designing the combination of ligands and metal nodes, introducing multifunctional groups, or constructing hierarchical pore structures, it is possible to finely tune the crystal structure, electronic structure, and transport properties of MOF materials, thereby endowing them with richer physicochemical properties. For example, certain MOF materials with complex topological structures can provide unique microenvironments that promote the adsorption and activation of reactant molecules, thereby enhancing the selectivity and activity of catalytic reactions.56 On the other hand, research efforts are focused on developing MOF materials with enhanced functionality and improved stability. In terms of functional enhancement, methods such as doping with heteroatoms, loading co-catalysts, or constructing heterojunctions are employed to optimize the electronic structure of MOF materials, thereby enhancing their light absorption capacity, charge separation, and transport efficiency, and ultimately improving their photocatalytic performance. In terms of stability enhancement, appropriate synthesis methods and post-processing techniques are employed to enhance the skeletal rigidity of MOF materials, improving their resistance to factors such as light, heat, and chemical environments. This ensures that they maintain stable structure and performance during long-term photocatalytic reactions, laying the foundation for practical applications.57
This paper reviews the research progress of MOFs and their derivatives in photoelectrocatalytic water splitting in recent years, focusing on their preparation methods, performance optimization mechanisms such as electronic structure modulation and interfacial effects, as well as challenges such as insufficient stability and mechanism studies. The research and development of photoelectrocatalytic hydrogen production is a form of solar energy utilization, which can promote the progress of renewable energy technologies. Understanding the principles and mechanisms of photoelectrocatalysis provides references and insights for other renewable energy technologies, and by integrating experimental and theoretical research results, we aim to provide theoretical basis and technological references for the development of the next generation of high-efficiency and low-cost catalysts.
2. PEC water splitting fundamentals
During the photocatalytic process, upon irradiation of a photocatalyst (semiconductor) with photons whose energy exceeds its bandgap, electrons in the VB are excited to the CB. This excitation results in the formation of negative charge carriers (eCB−) in the CB, while leaving behind positive holes (hVB+) in the VB. The loss of photocatalytic efficiency is mainly due to the direct binding of eCB− to hVB+ or ˙OH as shown in the following equations:54| eCB− + hVB+ → photocatalyst + heat | (1) |
| eCB− + ˙OH → OH− | (2) |
Photoelectrocatalytic technology enhances photocatalytic efficiency by integrating electrochemical techniques into the photocatalytic process.58–60 The photoelectrocatalytic mechanism is similar to the traditional photocatalytic mechanism, based on the energy band structure theory, the light-trapping materials such as semiconductors and their co-catalysts act as photoelectrodes, and the process of HER and OER occurs on the two photoelectrodes through the reaction of photogenerated electrons and holes with water under the effect of light and applied bias voltage, as shown in Fig. 1, respectively. The difference of the principle is that the introduction of an applied electric field promotes the separation of carriers to a certain extent, which in turn improves the efficiency of the reaction. Therefore, photoelectrocatalysis is concerned with the electrochemical properties of the photoelectrodes under light conditions, where the photogenerated carriers catalyze the electrochemical reaction process at the photoelectrode–solution interface. The technology has great potential for application in various fields. The photoelectrocatalytic hydrogen production process consists of three main stages: light absorption, charge separation and reaction.61 In the photocatalytic process, when the energy of photons (from solar energy) is larger than the semiconductor band gap, intrinsic ionization is induced at the photoelectroanode, and the absorption of photons excites electrons residing in the VB to the CB, yielding mobile electrons (eCB−) in the CB and leaving behind positively charged holes (hVB+) in the VB, which triggers the charge separation and these electron or hole pairs act as charge carriers. In photoelectrocatalytic reactions, the charge separation process is a key step in the photoelectrocatalytic reaction, which drives the subsequent chemical reactions.62
The steps of the photoelectrocatalytic reaction are roughly divided into three steps, as shown in Fig. 2. When light is irradiated on the photocatalyst semiconductor, the reaction of eqn (3) occurs.63
| 2hv → 2e− + 2h+ | (3) |
When the electrolyte is in contact with the photoelectrode, the Fermi energy levels of the two need to be aligned, leading to a redistribution of charge at the interface, creating a space charge region and generating a built-in electric field. This electric field drives electrons and holes to migrate in opposite directions: holes are pushed towards the surface and OER occurs, while electrons move towards the bulk phase.62 Water is decomposed by cavities at the photoelectrode anode, releasing gaseous oxygen (eqn (4)).64
| Anode: 2h+ + H2O(l) → 1/2O2(g) + 2H+(aq) | (4) |
Electrons are transferred under bias through an external circuit to the photocathode, where a HER occurs, where H+ ions produced due to the splitting of water then move across the electrolyte towards the cathode to be reduced and produce H2 (eqn (5)).65
| Cathode: 2e− + 2H+(aq) → H2(g) | (5) |
The total net reaction in a PEC cell is shown in eqn (6)66
| 2hv + H2O(l) → 1/2O2(g) + H2(g) | (6) |
Therefore, it is known that the factors affecting photoelectrocatalytic water splitting are (1) the light absorption range, (2) the separation of electron–hole pairs and (3) the surface electrochemical reaction. To optimize the performance of photoelectrocatalytic hydrogen production, it is necessary to improve the photo-absorption capacity, carrier separation efficiency and reactivity of the photocatalyst.67
3. PEC water splitting performance evaluation parameters
In the assessment of the hydrogen evolution reaction (HER) efficiency during photoelectrochemical (PEC) water splitting, it is essential to put forward efficiency criteria and develop suitable comparative metrics for evaluating the performance of PEC systems. It is worth noting that among the many indicators used to evaluate the performance of advanced photoelectrode materials, solar-to-hydrogen (STH) conversion efficiency is widely regarded as one of the most important, comprehensive, and practically meaningful core calibration efficiencies:Among these, jsc is the current density of the photoelectrode at 0 V in a two-electrode setup, Psolar is the incident power on the active photoelectrode, and AM 1.5G is the standard representation of 1 sun, equivalent to an irradiance of 100 mW cm−2. A is the illuminated electrode area, and ηF is the Faraday efficiency for hydrogen evolution.
In photocatalytic experiments, parameters such as photocurrent density are frequently used as performance evaluation criteria. The photocurrent density jp refers to the ratio of the photocurrent produced by a photoelectrode to its illuminated area under one-sun irradiation. As a key indicator, it directly mirrors the performance of the photoelectrode and is affected by multiple factors. Specifically, it is generally determined by the light absorption rate, the separation efficiency of electron–hole pairs in the bulk phase, and the surface charge injection efficiency, while also varying with the applied bias voltage. The photocurrent density can be expressed via the formula below:68,69
| jp = jmax × (ηabs × ηsep × ηinj) |
Additionally, the photoconversion efficiency formula:69–73
Another evaluation parameter is the applied bias photon current efficiency (ABPE). ABPE primarily reflects the overall energy conversion efficiency of a photocatalytic system under applied bias. In most cases, ABPE is reported as an efficiency value for the performance of a photocathode. ABPE measures the overall conversion efficiency of light energy to hydrogen energy, integrating light absorption, charge separation, interfacial reaction kinetics, and bias-assisted energy utilization efficiency. However, the applied bias photon-to-current efficiency (ABPE) is computed based on the voltage applied across the photoelectrode. As a modified form of STH efficiency under a specific applied voltage, ABPE requires measurements to be performed in a two-electrode setup for accurate determination.74 The calculation of ABPE can be expressed by the following equation:
Subsequently, the performance metrics of PEC water splitting are briefly elaborated with respect to light absorption efficiency, charge carrier separation efficiency, and catalytic activity.
3.1. Light absorption efficiency
In the field of photocatalysis, the formula Jabs = Jmax × ηabs is used to quantify the upper limit of a photocathode's light-capturing capacity under ideal charge separation and injection conditions, serving as the foundation for evaluating a material's intrinsic photoresponse performance. Jabs: the maximum achievable photogenerated current density (unit: mA cm−2), representing the current density when all carriers generated by the material absorbing photons theoretically participate in the reaction; Jmax: the theoretical maximum photocurrent density (unit: mA cm−2), referring to the limiting current value under specific wavelength sunlight irradiation at 100% quantum efficiency (i.e., each photon generates one electron–hole pair and all are utilized); ηabs: the light absorption efficiency of a semiconductor photovoltaic electrode reflects the material's ability to absorb incident light, with values ranging from 0–100%. The calculation formula is ηabs = 1–10(−A) (where A is the absorbance). The absorbance of semiconductors is measured using UV-vis diffuse reflectance spectroscopy. Jabs can be obtained by multiplying the Jmax and ηabs values for each wavelength using the trapezoidal integration method.In the photoexcitation process of semiconductor materials, the transition of valence band electrons to the conduction band follows the basic quantum model of the photoelectric effect. Within this model, the photoelectric effect can be delineated by the equation Ek = hν − W, where Ek represents the maximum kinetic energy of the excited electron after it breaks free from its confinement; h is Planck's constant; ν is the frequency of the incident photon; W is the work function of the material, which represents the minimum energy required to excite an electron from near the Fermi level to the material's outer layer (vacuum level). To excite an electron from the valence band top to the conduction band bottom, it must obtain sufficient energy to overcome the bandgap Eg. At this point, W is conceptually equivalent to Eg. This is because the excitation of electrons across the bandgap is the physical prerequisite for the generation of free carriers. Therefore, the minimum “escape work” required for the generation of photo-generated carriers (electron–hole pairs) is the bandgap Eg of the semiconductor. When Ek = 0, this is the minimum frequency required for light to excite carriers. Based on the formula and existing bandgap values, the minimum required frequency of light can be determined. Using the relationship between the speed of light c, frequency ν, and wavelength λ, c = νλ, the threshold frequency can be converted to the corresponding maximum wavelength.75
By characterizing light absorption efficiency, it is possible to more specifically identify key factors contributing to the excellent catalytic performance and enhanced light absorption properties achieved by photoconductive electrode materials.
3.2. Carrier separation efficiency
In photocatalytic water splitting systems, even if the photoanode material exhibits excellent light absorption properties, the efficiency with which the photoexcited electron–hole pairs are spatially separated and transported to the reaction interface (charge separation efficiency, denoted as ηsep) often becomes a bottleneck limiting the overall performance of the catalyst. Hence, the measurement of ηsep is equally crucial. In the presence of electron scavengers, photocurrent peaks associated with recombination or reverse reactions in the material can be minimized. Under such circumstances, the correlation between the photocurrent density in the electron scavenger system (JES) and ηsep can be expressed as ηsep ≈ JES/Jabs. Here, Jabs denotes the absorbed photon flux density, while JES refers to the extracted/collected charge carrier flux density. ηsep allows for an intuitive assessment of the photoelectrode material's capability to generate and separate photoexcited carriers. However, the exact value of Jabs is sometimes inaccessible,76 making it impossible to accurately determine ηsep.In such cases, we introduce several other efficiency measurement methods to assess carrier separation efficiency. The incident photon current efficiency (IPCE) is commonly used to calibrate the performance of such photoelectrodes77,78 (compared to RHE). IPCE can be calculated based on different wavelengths in the spectrum, describing the wavelength-dependent photocurrent response, and can be calculated (in simplified form) as:
| IPCE = ηabs × ηsep × ηinj |
Therefore, from the IPCE spectrum, ηsep can be derived.
IPCE quantifies the overall efficiency as a function of incident wavelength, incorporating losses from reflected or transmitted photons. To evaluate the intrinsic performance of a PEC device, these optical losses must be accounted for to determine the actual efficiency. This corrected metric, known as the absorbed photon-to-current efficiency (APCE), is calculated via:
Through APCE analysis, ηsep can be accurately determined.
For investigating the electronic properties of the photoelectrode, the flat-band potential (VF) and charge carrier density (ND) can be extracted using the following expressions:79
Transient photocurrent is a key technology for evaluating the photoelectron-hole separation efficiency in photoelectrodes. By analyzing the response kinetics of photocurrent to pulsed light irradiation, the charge separation, recombination, and transport efficiencies can be quantitatively analyzed. The larger the photocurrent, the higher the separation efficiency.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) steady: steady-state photocurrent (effective separation and participation of carriers in the reaction); Jph,
steady: steady-state photocurrent (effective separation and participation of carriers in the reaction); Jph,![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) max: initial peak current under illumination (total photo-generated carriers).
max: initial peak current under illumination (total photo-generated carriers).
In cases where the carrier separation efficiency of the fabricated photoelectrode is observed to be excessively low, targeted strategies can be employed to enhance the internal charge separation efficiency.
3.3. Catalytic efficiency
In the process of photocatalytic water splitting, after photogenerated charge carriers are successfully spatially separated within the material, they ultimately need to migrate to the electrode/electrolyte interface and participate in the target redox reaction on the catalyst surface. However, the accumulation of charge carriers on the surface and reaction efficiency are not spontaneously efficient but are strictly limited by the kinetics of the interface reaction. The efficiency of this critical step is typically quantified using the carrier injection efficiency (ηinj), which is also referred to as surface catalytic efficiency or interfacial charge transfer efficiency. ηinj can be calculated by comparing the photocurrent density with or without an electron scavenger (ηinj = JPEC/JES). By studying ηinj, one can gain further insight into the performance of the prepared photoelectrode and accurately improve its defects.While catalytic efficiency serves as a critical parameter for assessing the overall performance of a photocathode, it fails to capture the material's stability and selectivity. Faradaic efficiency (FE) is a key indicator for evaluating the reaction selectivity, stability, and charge utilization efficiency of a photocathode, directly reflecting how much of the transferred charge is effectively used for the target reaction (rather than side reactions or energy loss). FE < 100% indicates the presence of competitive side reactions, photocorrosion, and interface recombination. Given that not all photogenerated electrons participate in water splitting reactions, photocorrosion of the photoelectrode results in a Faraday efficiency (FE) significantly lower than 100%, as calculated by the equation below:

For gaseous products, the formula is converted to:
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 485 C mol−1); Nproduct: experimental measurement of the amount of product (mol).
485 C mol−1); Nproduct: experimental measurement of the amount of product (mol).
Since photocatalysis is a combination of photocatalysis and electrocatalysis, the commonly used evaluation parameters for electrocatalysis are also applicable to photocatalysis. The Tafel slope is an important parameter for determining catalyst activity. It is the graphical representation of the current generated in an electrochemical cell versus the electrode potential of a specific metal. In simple terms, the Tafel plot describes the electrode's ability to generate current in response to changes in applied potential. It correlates reaction rate with overpotential. The smaller the Tafel slope, the faster the electron transfer.83 Overpotential is a parameter used to assess catalyst activity. The potential applied above the equilibrium potential to initiate the reaction and release the product at the electrode is referred to as overpotential. At least 1.23 V is required to decompose water into its components, however, a substantial overpotential is generally required due to the complexity of reaction kinetics.84 For water splitting reactions, an optimal catalyst should possess a small Tafel slope and minimal overpotential.85
The turnover frequency (TOF) is defined as the number of substrate molecules converted per unit time by each active site on the catalyst surface (unit: s−1). Its core significance lies in eliminating the interference of non-intrinsic factors such as catalyst dosage, surface area, and dispersion, directly reflecting the inherent catalytic ability of the active sites. In electrocatalytic reactions, the TOF value directly indicates the catalytic efficiency of active sites: a higher TOF value means that each active site completes more catalytic cycles per unit time, indicating stronger intrinsic catalytic activity of the catalyst. The formula for TOF is:
For example, in the hydrogen evolution reaction (HER):
Additionally, the stability of photoelectrode materials constitutes another crucial parameter that demands consideration. As photoelectrode materials exposed to electrolytes for extended periods can be severely hindered by photochemical and electrochemical corrosion, which impede surface reactions. An excellent photoelectrode should maintain its performance over extended periods.86 An important factor in the practical application of materials is their stability during electrochemical studies, which primarily depends on the properties of the electrolyte and electrode. Typically, directly preparing catalytic materials is a better strategy than coating them onto electrodes to extend the stability of test materials even in alkaline media. Material stability can be assessed using chronoamperometry, cyclic stability testing, in situ FTIR, XRD, and SEM analysis.87–91
4. Preparation of MOF and its derivative photoanodes
Fujita et al.92 discovered the application of MOFs in catalysis in 1994. Subsequently, Alvaro et al.93 demonstrated that MOFs have semiconducting properties. Yu and Mahata et al.94,95 conducted a preliminary study on the preparation of MOFs with photocatalytic properties. In 2009, Mori's group reported the use of MOFs as photocatalysts for water splitting,96,97 and found that MOFs could significantly improve the performance of the photoelectrocatalytic oxygen evolution reaction, and this discovery contributed to the rapid development of MOF-based materials for use as photocatalysts for water splitting. The structure of MOFs usually consists of organic ligands and metal centers, where the organic ligands act as supports and the metal centers have in a connections role. In the synthesis of MOFs, different organic ligands and metal centers are used to form a desirable flexible molecular structure that can be easily modified in terms of physical and chemical properties. So far, various physical and chemical synthesis methods have been investigated, highlighting the following commonly used ones.984.1. Sedimentation
Deposition is a technique to transfer substances from the gas or liquid phase to a solid surface. In the preparation of MOF photocatalytic materials, MOF components, precursors or functional additives (such as semiconductor quantum dots99 and nanoparticles100) are deposited onto conductive substrates using techniques such as electrochemical deposition,101 atomic layer deposition,102 chemical vapor deposition (CVD),103 and sol–gel deposition.104 This process results in the formation of a composite material exhibiting enhanced photoelectrocatalytic performance. This method is highly controllable. By adjusting parameters such as temperature,105 sedimentation time106 and precursor concentration,107 the thickness, morphology and composition of MOF films can be precisely controlled. The deposition method can directly grow MOF films on complex shapes or flexible substrates (such as conductive glass,108 carbon cloth109 and metal oxides110), facilitating their integration into photocatalytic devices (such as photoelectrodes111). Through layer-by-layer deposition or co-deposition methods, heterojunction structures comprising MOFs and other functional materials like semiconductors112 and conductive polymers113 can be engineered to improve light absorption and enhance carrier separation efficiency. The n–n heterojunction composite CeBTC@FeBTC/NIF, developed by Dong et al.114 was created using a two-step electrodeposition technique. This composite demonstrated a notable improvement in photocatalytic efficiency by optimizing light absorption through a distinctive structural configuration and modulation of energy bands, as shown in Fig. 3. Cardenas-Morcoso et al.115 synthesized cobalt-based ZIF-67 MOF modified BiVO4-based photoanodes. Heat treatment of the deposited MOF in air converted ZIF-67 into highly porous CoOx co-catalysts, which resulted in substantial improvement in catalytic performance compared to bare BiVO4 electrodes. Tang et al.116 prepared MOF-derived Co3C/TiO2 nanocages by electrostatic adsorption deposition of nanosheets of ZIF on TiO2/Cu2O annealed in pure argon for 6 h. The MOF was used as an electrostatic deposition method for the deposition of ZIF-67 on TiO2/Cu2O. The MOF was then used as a catalyst in a cobalt-based photoanode. The synergistic effect of the heterostructure-optimized microstructure and components significantly improved the photocurrent density and carrier separation efficiency of PEC and enhanced the water oxidation performance of PEC. Zhou et al.117 utilized imidazolium-based cobalt (Co) complexes (Co-MIm) to cover rod-shaped BiVO4 electrodes for photoelectrochemical (PEC) water splitting via a simple in situ deposition method. With the help of Co-MIm, the charge separation and injection efficiency of BiVO4 photoanodes were improved. The method somewhat requires specialized equipment and is costly. Compared with traditional methods such as the solvothermal method, the deposition method (especially electrochemical deposition) may generate amorphous or low-crystallinity MOF, which reduces the porosity and exposure of catalytically active sites and affects the photovoltaic performance and low efficiency of scale-up production. Certain deposition methods have specific requirements for substrate conductivity or surface functional groups such as electrochemical deposition requires a conductive substrate,118 limiting the binding of MOF to certain functional substrates. Post-deposition annealing or activation treatments may be required to enhance crystallinity or remove impurities, increasing process complexity.119 | ||
| Fig. 3 (a) Flow chart of the synthesis of CeBTC@FeBTC/NIF. (b) Diagram of the microscopic reaction during electrodeposition.114 Copyright 2024, Elsevier. | ||
4.2. In situ growth method
The in situ growth method is a method to grow MOF materials with photoelectrocatalytic activity directly on the surface of a conductive substrate by chemical reaction. This method offers structural controllability. By adjusting the type and proportion of metal ions and organic ligands, as well as reaction conditions such as temperature, pH, and solvent,120–122 the crystal structure, porosity and surface chemistry of MOFs can be precisely controlled, thereby optimizing their photocatalytic performance. The in situ-grown MOF materials are tightly bonded to the substrate surface through chemical bonding, which is not easy to come off and improves the stability and lifetime of the materials. The preparation process is simple and usually carried out at room temperature and pressure without complex equipment, which makes it easy to operate and realize large-scale preparation. Liu123et al. reported the use of interfacial engineering to aid energy level modulation to improve the PEC water splitting performance of BiVO4 photoanodes. An integrated photoanode NiCo-MOF-CuCrO2-BiVO4 was prepared by coupling a CuCrO2 hole transport layer (HTL) and in situ growth of NiCo-MOF, as shown in Fig. 4. This integrated photoanode exhibits excellent PEC water oxidation performance and very high stability. It was shown that coupling a CuCrO2 HTL and in situ growth of NiCo-MOF had a synergistic effect in improving the PEC water oxidation performance of BiVO4 photoanodes. He et al.124 reported the synthesis of TiO2/Ti-MOF heterojunctions by in situ self-growth, with selective anchoring of dense Pt monoatoms, for high-efficiency photodegradation of water to produce hydrogen. The TiO2/Ti-MOF heterojunction follows a Z-type electron transfer mechanism, and the MOF phase, which serves as a selective anchoring site for the co-catalyst Pt, is demonstrated to be an electron-rich region of the heterojunction, which facilitates the further participation of the photogenerated electrons separated through the heterojunction in the hydrogen precipitation reaction. The catalyst exhibited significantly enhanced visible light hydrogenolysis activity (12.4 mmol g−1 h−1). However, the in situ growth method is very sensitive to reaction conditions such as temperature, concentration, pH and so on,125–128 which need to be precisely controlled to ensure the uniform growth and high quality of MOF materials. Limited material selectivity and the limited variety of substrate materials and functional additives currently available for the in situ growth method may limit its application in some specific areas. The yield of the in situ growth method may be lower compared to hydrothermal and solvothermal methods, etc., and further optimization is needed to improve the preparation efficiency. | ||
| Fig. 4 Schematic synthesis route of the NiCo-MOF-CuCrO2-BiVO4 photoanode.123 Copyright 2025, Wiley. | ||
4.3. Solvothermal
Solvothermal synthesis is the predominant approach for preparing MOFs. In this technique, metal salts and organic linkers are dissolved in a mixed solvent of organic and aqueous phases, followed by a reaction conducted within a sealed vessel at elevated temperatures (100–500 °C) for 12–48 hours. During this process, metal cations and organic linkers undergo self-assembly to form a crystalline metal–organic scaffold. The advantage of the solvent thermal method for preparing MOFs is that the high-temperature, high-pressure environment greatly promotes the solubility and diffusion rate of the reactants, which is conducive to the orderly arrangement of atoms/molecules and crystal growth. Typically, MOF crystals with very high crystallinity and highly ordered structures can be obtained. The products have high specific surface area and porosity. The structure and morphology can be adjusted by controlling the reaction parameters.129–132 Wang et al.129 used a solvothermal method to synthesize a three-dimensional (3D) highly crystalline cluster-type Ti-MOF by synergistically assembling hexagonal prismatic {[Ti6O6[iPrO6]]}6+ clusters and rhombic Cu2I2 dimers using isonicotinic acid, as shown in Fig. 5. This demonstrates the high crystallinity of MOFs synthesized via the solvothermal method. Liu et al.130 synthesized a highly crystalline MOF-808 with uniform size and regular structure via the solvent method at 130 °C for 5 hours. The resulting MOF material exhibits a specific surface area exceeding 1000 m2 g−1 and a pore volume exceeding 0.50 cm3 g−1, along with high thermal stability, as shown in Table 1. Kouser et al.131 synthesized Zn(INA) microcrystalline MOFs. The structure of the MOFs was confirmed via XRD, SEM, and TEM imaging, and their purity was verified through EDS analysis. BET studies indicated that it has a large surface area and pore volume, suggesting potential for various industrial applications. This demonstrates that MOFs synthesized by the solvothermal method have high surface area and high porosity. Hu et al.132 used hexahydrate nickel chloride (NiCl2·6H2O) as the metal salt and terephthalic acid (PTA) as the organic ligand. By altering the type of solvent, they employed a one-step solvothermal method to self-assemble high-loading nickel-based MOF materials (Ni-MOF/NF) on the surface of foam nickel. The better the solvent's solubility for PTA or the higher the pH value, the faster the deprotonation rate of PTA in the solution, and the faster the nucleation rate of the material. The self-grown nickel-based MOF materials exhibit three distinct morphologies—spherical clusters, plate-like, and bulk-under different solvent systems, with corresponding changes in loading capacity. This demonstrates the strong controllability of the solvothermal method. | ||
| Fig. 5 (a) Structure of the hexagon-shaped unit {[Ti6O6[iPrO6[INA6]]]}. (b) Coordination environment of the Cu2I2 dimer. (c) View of the cage. (d) Crystal photograph of MTM-1.129 Copyright 2017, RSC. | ||
| Sample | BET surface area/(m2 g−1) | Pore volume/(cm3 g−1) |
|---|---|---|
| MOF-808-130 °C-1 h | 504 | 0.29 |
| MOF-808-130 °C-5 h | 1004 | 0.53 |
| MOF-808-130 °C-10 h | 1171 | 0.61 |
| MOF-808-130 °C-15 h | 1083 | 0.59 |
| MOF-808-130 °C-20 h | 1232 | 0.68 |
However, the reaction conditions are harsh, requiring high temperatures (120–200 °C) and high-pressure environments, with equipment dependent on pressure-resistant reactors, posing high operational risks. It is energy-intensive and time-consuming. Reaction times typically range from 6 to 24 hours, and energy consumption does not meet green chemistry requirements. The extensive use of toxic solvents such as DMF not only increases costs and environmental impact but also risks residual solvents blocking pores, necessitating complex post-processing (e.g., ethanol exchange, vacuum drying). Issues such as high temperatures and pressures, prolonged reaction times, extensive solvent use, and potential batch-to-batch variability pose significant challenges when scaling up from laboratory to industrial production.133–135 As an important method for synthesizing MOF materials, researchers have identified some approaches to address some of these drawbacks, but further exploration of solutions or alternative preparation methods is still needed. For example:
1. The mother liquor-induced synthesis method not only reduces the thermodynamic barriers to crystal nucleation but also induces crystal nucleation and growth, significantly reducing crystallization time while increasing material crystallinity and purity. Professor Jiang Haolong's team successfully induced the synthesis of 100% pure MOFs by pre-synthesizing a small amount of pure-phase zirconium-based MOFs as seeds and adding them to the reaction system, while reducing the reaction time to 1–10 hours.136
2. Poor sample reproducibility is primarily due to the failure to accurately control reaction parameters, including reaction time, temperature, pressure, reactant concentration, flow rate, and surface area to volume ratio. Effective control of these parameters ensures catalyst reproducibility.133
3. Establishing databases, sharing of open data and the adoption of transparent research practices are capable of substantially improving the reproducibility of studies focused on porous materials.133
4. Seeking non-toxic and inexpensive organic solvents or utilizing hydrothermal synthesis instead of solvent thermal synthesis. HKUST-1 was synthesized via hydrothermal and solvent thermal methods,137 with hydrothermal synthesis capable of producing particles with high specific surface area.134,138,139
5. Replacing high temperatures with room temperature (RT). The RT synthesis of HKUST-1 (MOF-199) has also been demonstrated by researchers to be scalable. RT synthesis involves mixing the copper acetate metal precursor with a pre-homogenized aqueous suspension of H3BTC for one hour to achieve a space-time yield (STY) of 2035 kg m3 day−1. Typically, RT synthesis requires pre-activating ligands using salt-like ligands or by adding a base to deprotonate the ligands, as this promotes faster nucleation during synthesis. In terms of surface area and crystallinity, the properties of MOFs synthesized by this method are comparable to those of traditional and industrial syntheses. Although RT synthesis can be easily scaled up for production and offers higher yields and lower energy consumption compared to current industrial production of HKUST-1, the limitations of using deprotonated ligands for MOF synthesis significantly restrict the flexibility of RT synthesis.140–143
4.4. Calcination
Calcination for the preparation of MOF photoelectrocatalytic materials is a method of treating MOF materials at high temperatures in order to change their physical and chemical properties and thus improve their photoelectrocatalytic properties. The calcination process removes impurities and volatiles from the MOF materials and improves their thermal and chemical stability. The calcined MOF materials have higher stability and catalytic activity, and can be applied in a wider range of fields, such as environmental pollution control, energy conversion, and organic synthesis, etc. Pan et al.144 employed a two-step calcination process to convert ZIF-8 for the preparation of porous carbon (C). As illustrated in Fig. 6, the resulting modified structure successfully incorporates C doping into the ZnO lattice while preserving its cubic morphology. Li et al.145 were the first to demonstrate that core–shell heterojunctions derived from MOFs exhibit enhanced photoelectrochemical (PEC) water splitting efficiency. The photovoltaic electrodes were prepared by coating MOF NH2-MIL-125 (Ti) onto Fe2O3 nanorods using a solvothermal procedure and subsequent two-step calcination to form TixFe1−xOy shell/Fe2O3 core nanorod arrays. However, the calcination method may lead to structural damage, which may collapse the structure of the MOF material or decompose the organic ligands, affecting its catalytic performance. The calcination process consumes a lot of energy and time, which increases the preparation cost. In addition, the use and maintenance of the calcination equipment also require a certain cost investment. Limited material selectivity and the limited variety of MOF materials currently available for the calcination method may limit their application in some specific areas. | ||
| Fig. 6 Schematic illustration of the fabrication of the ZnO@M@-ZIF-67 photoanode for PEC water splitting.144 Copyright 2016, Elsevier. | ||
4.5. Mechanochemical method
Mechanochemical synthesis involves grinding metal salts and organic ligands to prepare MOFs. Molecular bonds are disrupted by mechanical forces during milling, leading to the formation of new products through re-coordination. The first study of mechanochemical synthesis of MOFs was reported by Pichon et al.146 in 2006. The process involves no solvent and the target product can be synthesized within 10 minutes under mild conditions, offering time efficiency but yielding products with low purity and poor crystallinity. To address these limitations, researchers have modified the protocol by introducing a liquid-assisted grinding (LAG) approach. Specifically, Friščić's group147 incorporated a minimal amount of solvent during the milling stage, leveraging the liquid's structural-directing effects to enhance reaction kinetics and crystal quality (Fig. 7). Despite the advantages of high synthesis efficiency and reduced solvent usage in mechanochemical methods, the resulting MOFs often exhibit suboptimal crystallinity and impurity inclusion.148 | ||
| Fig. 7 (a) Expected MOF 1 assembly; (b) MOF isomers 1 a and 1 b; red O, gray C, blue N, purple Zn. (c) PXRD patterns (top to bottom): 60 min neat grinding (red; no reaction), 60 min LAG (blue; partial reaction), simulated pattern for the dabco-Hta salt and for 1 b (in square-root scale). Dotted lines indicate reflections of ZnO; reflections of residual Hta are labeled “☆”.147 Copyright 2010, Wiley. | ||
4.6. Ultrasonic method
Sonochemical synthesis, in simple terms, is a synthetic process that harnesses ultrasound-generated energy to regulate chemical reactions. Among the various methods for the synthesis of MOFs, the ultrasonic method is an economical and eco-friendly method, which can provide high yields under mild operating conditions. Yang et al.149 used an ultrasound-assisted method to uniformly coat an ultrathin CoFe MOF layer on the surface of a BiVO4 photoanode to construct a type II heterojunction and to promote charge separation and transfer. The optimized photoanode achieved a photocurrent density of 3.92 mA cm−2 at 1.23 V vs. RHE and a photonic current efficiency (IPCE) of 89% at 400 nm, as shown in Fig. 8. However, the reactivity of this method exhibits limited controllability, often leading to the emergence of parasitic reactions and the incorporation of impurities within the synthesized products. | ||
| Fig. 8 Schematic illustration of the synthesis process of BVO/CoFe MOF(NA) and BVO@CoFe MOF(NA) photoanodes.149 Copyright 2023, Wiley. | ||
4.7. Microwave method
The microwave method was first reported in 2005.150–152 As an emerging material synthesis technique, it has been widely used for the preparation of MOFs in recent years. Its main working principle is based on the unique mechanism of microwave–matter interaction, which promotes intermolecular coordination reactions of reactants through rapid and uniform heating, thus realizing efficient synthesis. MIL-125(Cr) represents the initial MOF synthesized through microwave-assisted methods, exhibiting a 44% yield after 4 hours at 220 °C. In contrast, conventional heating methods required 4 days to attain a comparable yield.153 Wu et al.154 used a microwave-assisted synthesis method to prepare soluble porphyrin MOFs (Al-PMOF), as shown in Fig. 9. Since Al-PMOF itself does not possess active sites for water splitting to produce hydrogen, (Pd(CH3CN)2Cl2) was introduced as a precursor for Pd. Pd nanoparticles were deposited onto Al-PMOF via photodeposition, enabling the use of Pd nanoparticles as co-catalysts in photocatalytic water splitting experiments. The microwave method significantly reduced reaction time (30 minutes at 140 °C, compared to 16 hours at 180 °C using traditional methods). Compared to bulk MOF (Bulk-Al-PMOF), the photocatalytic water splitting activity for hydrogen production was enhanced by 14 times. The synthesis of MOFs using microwave irradiation can effectively decrease reaction times and enhance the crystal quality of MOFs. Moreover, this method embodies environmentally friendly practices, energy efficiency, and operational simplicity. Nonetheless, it is essential to acknowledge that this approach necessitates precise instrumentation for regulating microwave energy and mandates that metal ions possess strong electromagnetic wave absorption capabilities, thereby somewhat constraining its applicability. Table 2 summarizes the advantages and disadvantages of various methods. | ||
| Fig. 9 Schematic illustration of the synthesis process of S-Al-PMOF.154 | ||
| Synthesis method | Advantages | Disadvantages |
|---|---|---|
| Sedimentation | 1. High controllability | 1. May have substrate requirements |
| 2. May increase process complexity | ||
| In situ growth method | 1. High controllability | 1. Reaction conditions must be precisely controlled |
| 2. High stability | 2. Material selection is limited | |
| 3. Simple preparation process | 3. Yields may be low | |
| 4. No complex equipment required | ||
| 5. Easy to operate | ||
| Solvothermal | 1. No special equipment required | 1. Long synthesis time |
| 2. Crystals can be grown | 2. Low yield; difficult to produce on a large scale | |
| Calcination | 1. Improve thermal stability and chemical stability | 1. Requires a significant amount of energy and time |
| 2. Enhance catalytic activity | 2. The use and maintenance of calcination equipment incurs costs | |
| 3. Applicable to a wider range of fields | 3. Material selection is limited | |
| Mechanochemical method | 1. No solvents involved | 1. The product has low purity and poor crystal form |
| 2. Mild reaction | ||
| 3. Time-saving | ||
| Ultrasonic method | 1. Low cost | 1. The reaction is difficult to control |
| 2. Environmentally friendly | 2. May be accompanied by side effects | |
| 3. Mild operating conditions | ||
| Microwave method | 1. Highly efficient synthesis | 1. Microwave energy is difficult to control |
| 2. Metal ions must have good electromagnetic wave absorption capacity | ||
| 3. Weak universality | ||
5. Performance optimization strategies
Since the photoelectrocatalytic water splitting mechanism was mentioned earlier, the steps of photoelectrocatalysis are divided into light absorption to produce carriers, carrier separation, and surface chemical reaction. Therefore, the performance of photoelectrode materials is limited by several energy loss mechanisms as follows: (1) when the incident photon energy exceeds the semiconductor band gap, the excess energy is dissipated as heat or photoluminescence, resulting in a low utilization of light energy. (2) Energy band structure mismatch and poor conductivity can hinder carrier separation, leading to lower catalytic efficiency. (3) Photogenerated carriers may be complexed in the bulk phase or on the surface, resulting in fewer carriers effectively participating in the reaction. (4) HER/OER at high energy barriers may result in carrier depletion while simultaneously inducing electrode polarization or material corrosion. To optimize their performance, ideal photoelectrode materials should be developed through three key strategies: enhancing light absorption, modulating carrier dynamics, and engineering surface reactions. These approaches are essential for achieving efficient conversion of solar energy into hydrogen.The bandgap (Eg) of MOFs directly determines their light absorption range, and narrow bandgap MOF materials can utilize visible light or even near-infrared light. Therefore, it is feasible to reduce the bandgap of MOFs by modifying them and developing new MOFs. Moreover, the efficiency of photogenerated charge carrier (electrons and holes) migration is a critical determinant of photoelectrode performance. The construction of rational heterostructures of MOFs together with other semiconductor materials is the key to promote charge separation. Surface electrochemical reactions constitute the final and rate-determining step in photoelectrochemical water splitting. The surface properties of photoelectrode materials critically govern the efficiency of charge carrier utilization. Specifically, surface defects, dislocations, and active sites act as reaction centers where photogenerated electrons and holes participate in redox reactions. However, these defects can either enhance or impede catalytic performance depending on their type, density, and distribution.62,155 The direct contact of electrolytes with the photoelectrode and the attachment of H2/O2 bubbles are the two key factors leading to the deterioration of the surface electrochemical reaction. The former destroys the material structure through chemical/photocorrosion mechanisms, and the latter exacerbates the reaction inhomogeneity through physical hindrance and local potential fluctuations. Moreover, for long-term photoelectrocatalytic reactions, the stability of the photoelectrode material must be high, as shown in Fig. 10. Therefore, further improving the stability of MOFs is also an important research direction. Future research will focus on designing and optimizing the photoelectrocatalytic materials, investigating the energy band structure, surface state and carrier transport mechanism of the photoelectrocatalytic materials, and improving the photoelectrocatalytic activity, light absorption performance, carrier mobility and stability of the photoelectrocatalytic materials through structural design of the materials, component modulation and doping modification.
5.1. Light absorption extension and energy level structure
Light absorption in photoelectrocatalysts refers to the process of photon interaction with the material, which determines the onset of light excitation, affects charge separation and migration, and is a key step in applications such as hydrogen production by photoelectrocatalysis. The light absorption efficiency is a measure of the photocatalyst's ability to absorb light. In photoelectrocatalysis, carrier production is directly related to the material's bandgap width and absorption coefficient, and increasing the light absorption efficiency is essential to increase the production of electron–hole pairs.156 The bandgap width determines the light absorption capacity of the photoelectrode material throughout the solar spectrum, thereby establishing the material's maximum potential efficiency for solar-to-hydrogen (STH) conversion. Heterojunction materials featuring a narrow bandgap structure exhibit enhanced light absorption within the solar spectrum. However, the bandgap cannot be excessively narrow as it must still supply an ample driving force for both the oxygen and hydrogen precipitation reactions.157 The energy band structure of a photoelectrocatalytic material is an important property that determines the energy state of the photoexcited carriers. Photoelectrodes for PEC water splitting devices must meet specific criteria regarding their energy band positions, including appropriate band gap, valence band, and conduction band levels. It is essential for the conduction band edge of these materials to be positioned lower than the water reduction potential E (H+/H2), while the valence band edge should be situated above the oxygen reduction potential E (O2/H2O). Semiconductor materials are ideal for PEC photoelectrodes because of their tunable energy band edge positions and bandgap widths. To achieve optimal performance, these semiconductors must efficiently utilize most of the visible light spectrum and maintain stability under dim light conditions in order to use sunlight to decompose water into commercially viable hydrogen. Optimizing the energy band structure is key to improving photoelectrocatalytic performance. Researchers have explored various material modification methods such as elemental doping, composites and ligand functionalization to further improve the performance. Table 3 briefly describes the extended photoabsorption catalytic materials reported in recent years for PEC water cracking.| Photoelectrode | Modification method | H2 evolution rate | Light absorption range | Bandgap energies | Photocurrent density vs. RHE | Ref. |
|---|---|---|---|---|---|---|
| CuSL–CuBr | Heteroatom doping | 50.28 mmol g−1 h−1 | 250–600 nm | 2.30 eV | 0.64 μA cm−2 | 158 |
| Mn-FeBTC/NIF | Heteroatom doping | — | 380–800 nm | 1.53 eV | — | 159 |
| Cu3HHAE2 | Composite material | — | up to 1600 nm | 0.87 | 260 μA cm−2 (0.1 M Na2SO4 0 V) | 160 |
| Ag/Au HPNS-Cd0.8Zn0.2S NSs | Composite material | — | 365–850 nm | 1.7 eV | 0.16 mA cm−2 (0.65 V) | 161 |
| Co3C−x/TiO2 | Ligand functionalization | — | 390–850 nm | 1.42 eV | 2.6 mA cm−2 (1 M NaOH 1.23 V) | 163 |
| Ti6((μ3-O)2(Oi-Pr)8) (LA)2·i-PrOH (S1) | Ligand functionalization | — | 200–510 nm | 2.85 eV | 2. 12 μA cm−2 (0.2 M Na2SO4) | 165 |
 | ||
| Fig. 11 (a) Cu 2p XPS spectrum of CuSL–CuBr. (b) PXRD patterns of CuSL–CuBr after immersion in solutions of varying pH for one week. (c) UV-vis diffuse reflectance spectrum of CuSL–CuBr and CuSL–CuCl. (d) Mott–Schottky plot of CuSL–CuBr. (e) UPS spectra of CuSL–CuBr. (f) Band structure diagram of CuSL–CuBr and CuSL–CuCl.158 Copyright 2025, Wiley. | ||
Dong et al.159 deposited the semiconductor Mn-FeBTC on a well-conducting nickel iron foam and used a trace Mn doping strategy to efficiently modulate the BIEF of the Mott Schottky heterojunction formed by FeBTC and a metal substrate. The Mn-FeBTC/NIF composite exhibited enhanced light absorption across the 380–800 nm range, peaking at 68.5% at 738 nm. Additionally, there was a 0.28% increase in maximum absorption intensity at 597 nm, which is due to the increase in photosensitivity caused by the decrease in band gap due to Mn doping.
 | ||
| Fig. 12 (a and b) Variable-temperature electrical conductivities of Cu3HHAE2. (c) Vis-NIR absorption spectroscopy of Cu3HHAE2. (d) UPS results of Cu3HHAE2; the left image is the low kinetic energy region of the UPS spectra, and the right image is the low binding energy region (HOMO).160 Copyright 2022, Wiley. | ||
 | ||
| Fig. 13 (a) UV-vis DRS spectra and (b) normalized photoluminescence spectroscopy of TiO2, Co3C−x/TiO2 (x = 1, 3, 6, 9) and Co3C.163 Copyright 2018, Wiley. | ||
Hu et al.164 first modified the –NH2 group onto MIL-125 using a ligand functionalization strategy to obtain NH2-MIL-125, and then successfully prepared Cu–NH2-MIL-125 by Cu ion coordination. This dual modification strategy resulted in a reduced band gap and a wider range of light absorption, exhibiting excellent piezoelectric–photocatalytic activity (2884.2 μmol g−1 h−1). In addition, by introducing chromophores or photosensitizers with strong light-absorbing ability, the light-absorbing ability of the MOF materials can be enhanced and their photoresponsive range can be extended to the visible region. Yang et al.165 utilized naphthalene diimide (NDI) ligands, H2LA, as photo-active sensitizing molecules, and designed and synthesized the cyclic titanium–oxygen clusters Ti6((μ3-O)2(Oi-Pr)8)(LA)2·i-PrOH (S1). As shown by ultraviolet-visible (UV-vis) spectroscopy and photocurrent tests, the NDI ligand not only extended the absorption edge of S1 to 500 nm, but also caused S1 to display a photocurrent density as high as 2.12 μA cm−2, thus demonstrating the importance of the photoreactive ligand in the modulation of the photoresponse of TOCs, as shown in Fig. 14.
 | ||
| Fig. 14 Solid-state UV-vis spectra and band gap: (a) absorption spectra of S1, S2 and ligands; (b) and (c) Kubelka–Munk transformation of diffuse reflectance data of S1 and S2.165 | ||
5.2. Photogenerated charge transport and utilization
The efficiency of photocatalytic reactions is directly dependent on the effective separation of photogenerated electrons and holes. During photocatalysis, electron–hole pairs are generated by the absorption of light energy by the semiconductor material, but if these carriers are compounded before migrating to the surface to participate in the reaction, it will lead to energy waste and reduce the overall efficiency. For example, studies of Al-doped SrTiO3 have shown166 that by suppressing carrier complexation and optimizing the charge separation mechanism, the quantum efficiency can be close to 100%, and high efficiency water splitting can be achieved even with extremely short carrier lifetimes (sub-nanosecond). Optimizing charge separation also enhances the photocurrent density. The carrier separation efficiency directly affects the rate of redox reaction on the surface of the photoelectrode. For example, atomic layer mesoporous In2O3−x/In2S3 heterojunction167 shortens the carrier migration distance through interfacial strong coupling and mesoporous structure, and achieves a photocurrent density of 1.28 mA cm−2 (1.23 V vs. RHE), which is significantly higher than that of a single material. Some materials are limited in their light absorption or charge transport ability due to bandgap or defect issues, which need to be optimized by carrier separation strategies. For example, Sn-doped SrTiO3 suppresses Ti3+ defects (carrier complex centers) by replacing Sr sites168 with Sn2+ to improve the overall water splitting efficiency by a factor of 10.4. Enhancing the carrier separation efficiency is the key to the practicalization of photoelectrocatalytic water splitting technology. Therefore, future studies of photoelectrocatalytic MOF materials need to further explore the carrier separation mechanism with low energy consumption and high stability to promote the large-scale application of green hydrogen energy. Table 4 briefly describes the catalytic materials that have been reported in recent years to enhance carrier separation efficiency for PEC water cracking.| Photoelectrode | Modification method | Charge recombination lifetime | Faradaic efficiency | Photocurrent density 1.23 V vs. RHE | Ref. |
|---|---|---|---|---|---|
| BiVO4/CoFe MOF | Heterojunction | 89 ns | 92% | 3.92 mA cm−2 (1 M KBi) | 149 |
| Co-agZIF-62/NiO/BiVO4 | Interface engineering | 22.12 ns | — | 5.34 MA cm−2 | 169 |
| NiCo-MOFCuCrO2-BiVO4 | Interface engineering | — | 95.3% | 5.75 mA cm−2 | 170 |
| Ov-BiVO4/MIL-101 | Defective engineering | 6.62 ns | — | 5.91 mA cm−2 (1 M KBi pH = 9) | 171 |
| Vw-WO3 | Defective engineering | 13.61 | Nearly 1 | 4.0 mA cm−2 (Na2SO4 pH = 7) | 172 |
| Co3O4/CoV-MOF/BiVO4 | Defective engineering | 10 ns | 6.0 mA cm−2 | 173 | |
| (Co, Ni)-MOF/WO3/W | Electronic control | — | 90.07% | 2.96 mA cm−2 (0.5 M Na2SO4) | 174 |
| BiVO4 @aNiFc-MOFs | Electronic control | 35.31 ns | 92.1% | 4.34 mA cm−2 (0.5 M KBi) | 175 |
 | ||
| Fig. 15 Femtosecond transient absorption (fs-TA) spectra of (A) BVO and (B) BVO/CoFe MOF(NA) excited at 375 nm. (C) Normalized fs-TA spectra probed at 620 nm for the BVO and BVO/CoFe MOF(NA) under 375 nm excitation. (D) Electron transfer pathways involved in BVO and BVO/CoFe MOF(NA) heterostructure systems. τre represents charge recombination lifetime.149 Copyright 2023, Wiley. | ||
 | ||
| Fig. 16 A schematic illustration of the charge transfer process on MO and Co-agZIF-62/NiO/MO photoanodes.169 Copyright 2023, Wiley. | ||
Tian et al.123 used an interface engineering-assisted energy level modulation strategy to effectively optimize the photocatalytic water oxidation performance of bismuth vanadate photoanodes. The coupling of the CuCrO2 HTL (hole transport layer) with the BiVO4 photoanode generated an in-built electric field, which efficiently facilitated the interfacial migration of carriers. In addition, the in situ growth of NiCo-MOF exhibits a dual effect. On the one hand, the specific functional groups present in NiCo-MOF can effectively passivate the surface defect states of the BiVO4 photoanode, thus significantly reducing the surface recombination of carriers. On the other hand, in NiCo-MOF, the bimetallic elements Ni and Co as OER active sites can significantly accelerate the kinetic process of the water oxidation reaction. Under the synergistic effect of the CuCrO2 HTL and further in situ growth of NiCo-MOF, the photogenerated holes can migrate to the semiconductor/electrolyte interface rapidly and participate in the water oxidation reaction in time, which greatly enhances the water oxidation performance of the PEC of the BiVO4 photoanode, as shown in Fig. 17.
 | ||
| Fig. 17 Working mechanism diagram of the BiVO4 photoanode, CuCrO2-BiVO4 photoanode and NiCo-MOF-CuCrO2-BiVO4 photoanode.123 Copyright 2025, Wiley. | ||
5.2.3. Defective engineering
Defect engineering is also an effective method to enhance carrier separation efficiency. Defect engineering significantly enhances the carrier separation efficiency of MOF catalysts in PEC water splitting through electronic structure modulation, surface property optimization, and carrier dynamics enhancement (the pores or interfaces formed by the defects promote the reactant diffusion and electron transfer).170 Xin et al.171 proposed a molecular epitaxial growth technique based on oxygen vacancy (Ov)-driven molecular epitaxial growth on the BiVO4 surface to prepare a uniform MIL-101 layer (Ov-BiVO4/MIL-101 composite structure). This technique generates a high concentration of oxygen vacancies on the BiVO4 surface by electrochemical etching, which promotes the molecular-level binding of MIL-101 to BiVO4 and significantly improves the interfacial charge separation and transport efficiency. The photocurrent density of the Ov-BiVO4/MIL-101 composite photoelectrode reaches 5.91 mA cm−2 at 1.23 VRHE potential, which is 2.93 times higher than that of the conventional BiVO4, and achieves a 4.33% in the unbiased solar-hydrogen conversion system with a solar-hydrogen efficiency. Wang et al.172 proposed an innovative strategy: reducing the effective mass of holes and significantly enhancing their mobility by modulating the metal vacancies (VMs) in semiconductor metal oxides. It is shown that this strategy exhibits universality in all three transition metal oxides (WO3, TiO2 and Bi2O3), and ultimately achieves a photocurrent enhancement by a factor of 4.4 and a stability of more than 120 h in PEC water splitting applications. Calculations show that the formation of VW (tungsten vacancy) in WO3 results in a positive shift of the valence band maximum (VBM) and the creation of a “band-tailed state”, as shown in Fig. 18a and c. Energy band calculations show that the effective mass m* of the hole decreases from 2.69 m0 to 0.95 m0 (65% reduction) after the introduction of the metal vacancy, which implies a significant increase in the hole mobility, as shown in Fig. 18b and d. Therefore, it can be concluded that the tungsten vacancy (VW) reduces the effective mass of the hole, which enhances the hole mobility and makes the carrier separation more efficient. Xu et al.173 constructed CoV-MOFs with ligand defects on the surface of BiVO4 by photoelectrochemical treatment, which enhances the interfacial bonding and inhibits the V5+ dissolution. The optimized photoanode achieved a photocurrent density of 6.0 mA cm−2 at 1.23 V and operated stably at 0.6 V for 500 hours. The ligand defects modulated the energy band structure to enhance the light absorption and charge transfer efficiency. | ||
| Fig. 18 Theoretical calculation of the energy band structures. (a) and (c), calculated density of states (DOS) of WO3 (a) and Vw-WO3 (c). (b) and (d), Energy band structures of WO3 (b) and Vw-WO3 (d).172 Copyright 2025, Nature. | ||
5.2.4. Bandgap engineering
Strategies for bandgap engineering using semiconductor photocatalysts can influence the transport paths of electrons and holes, thereby improving carrier separation efficiency. In addition, altering the bandgap position can enhance their redox capabilities. Dong et al.174 decorated the surface of coral-like WO3/W films with hierarchical, flower-like arrays of stabilized heterogeneous (Co, Ni)-metal–organic framework (MOF) composites. The resulting (Co, Ni)-MOF/WO3/W photoanode exhibited a high photocurrent density of 2.96 mA cm−2 at 1.23 V vs. RHE in a 0.5 M neutral Na2SO4 electrolyte and achieved a solar-to-hydrogen (STH) efficiency of 2.17% under AM 1.5 G illumination. Light-assisted Kelvin probe force microscopy (KPFM) revealed the spatial distribution and segregation dynamics of photogenerated charges. The energy band alignment facilitated efficient charge separation: the conduction band (CB) of (Co, Ni)-MOF lies above that of WO3, enabling spontaneous electron transfer from (Co, Ni)-MOF to WO3. These electrons then traverse the WO3/W Schottky junction to reach the cathode under bias, while the photogenerated holes accumulate in the valence band (VB) of (Co, Ni)-MOF and participate in the oxygen evolution reaction (OER), as illustrated in Fig. 19. This mechanism effectively suppresses charge recombination, enhancing solar-driven photoelectrochemical water splitting. | ||
| Fig. 19 The schematic illustration of separation and transfer paths of photogenerated charges for the (Co, Ni)-MOF/WO3/W photoanode during the PEC process.174 Copyright 2024, Elsevier. | ||
Li et al.175 proposed a strategy to enhance the quasi-Fermi level of MIL-53 (Fe) by injecting high-energy electrons via EY, thereby boosting photocatalytic activity. The integration of characterization techniques and theoretical simulations revealed that dye sensitization is capable of efficiently injecting high-energy electrons into Fe-based metal–organic frameworks (Fe-MOFs) and raising their quasi-Fermi level upon light irradiation, as shown in Fig. 20. After EY has sufficiently elevated the quasi-Fermi level of Fe-MOFs under light irradiation, depositing Pt nanoparticles on the Fe-MOF can reduce charge transfer resistance and improve charge separation, thereby lowering the overpotential for H2 evolution.
 | ||
| Fig. 20 (a) Energy diagrams of MIL-53(Fe) before and after EY sensitization. (b and c) Side views of the charge densities at the EY-MIL-53(Fe) interface showing the electron unoccupied and occupied states. Color: white (H); grey (C); red (O); orange (Fe); brown (Br).175 Copyright 2022, Elsevier. | ||
5.3. Electrode reactions and generation of products
 | ||
| Fig. 21 Structure of the LP–MOF for overall photocatalytic water splitting and the proposed ‘Z-scheme’ electron-transfer chain in the LP–MOF system. The bilayer in the illustration represents a section of the liposome. The HER-MOF for hydrogen evolution is incorporated between the hydrophobic chains, and the WOR-MOF for water oxidation is in the aqueous phase. The redox shuttles TCBQ/TCBQH and Fe3+/Fe2+ connect the HER and WOR sides, functioning in the lipid phase and the aqueous phase, respectively. Electron-transfer chains (shown by blue arrows) and photons (represented by yellow lightning shapes) form the ‘Z-scheme’. Inset, schematic representation of the liposome with the two MOFs embedded within the hydrophobic bilayer (HER-MOF) and the hydrophilic interior (WOR-MOF).187 Copyright 2021, Nature. | ||
To gain a deeper understanding of the photoelectrocatalytic process, it is helpful to study the electrode reaction kinetics. For example, we can study the migration of photogenerated charges on the electrodes and the reaction process measured current–voltage curves and electrochemical impedance spectra, which can help to optimize the electrode structure and improve the photoelectrocatalytic efficiency. For example, Yang et al.149 coated the surface of BiVO4 nanoarrays with a CoFe MOF (NA) nanolayer, resulting in the BiVO4/CoFe MOF (NA) catalyst. EIS confirmed a significant reduction in interfacial resistance, as shown in Fig. 22a; the current–voltage curve showed a photocurrent density of 3.92 mA cm−2 at 1.23 V, a 6.03-fold increase compared to pure BiVO4, due to significantly improved charge transfer and separation efficiency, as shown in Fig. 22b. At 400 nm, the BVO/CoFe MOF(NA) photoanode exhibits the highest IPCE, with an APCE as high as 96%, indicating that almost all absorbed photons at this wavelength effectively drive the water oxidation reaction, as shown in Fig. 22c.
 | ||
| Fig. 22 (a) EIS Nyquist plots under light irradiation. (b) Chopped light i–t curves of BVO, BVO@CoFe MOF(NA), BVO/CoFe MOF(5-ana), and BVO/CoFe MOF(NA). (c) IPCE spectra (inset shows the APCE spectra).149 Copyright 2023, Wiley. | ||
Co-catalysts can improve the reactivity of photoelectrocatalysts and promote the splitting of water molecules and hydrogen generation on the surface of photoelectrocatalysts; certain co-catalysts with specific functions, such as noble metals and transition metal oxides, can improve the reactivity and establish an effective electron transport pathway by providing active centers or promoting the adsorption of intermediate species. For example, Wang et al.188 investigated the effect of MOF-derived co-catalysts on the photocatalytic performance of BiVO4 photoanodes. Porous BiVO4-based thin films were prepared using electro-deposition and thermal annealing methods. The BiVO4 electrodes were then immersed in a Co-MOF (ZIF-67) precursor solution, and the porous BiVO4 surface was subjected to in situ modification at room temperature. The resulting BiVO4-MOF photoanode was annealed at 450 °C for 2 hours in a N2 atmosphere, converting the MOF into a carbon matrix containing cobalt-based active reaction centers, ultimately yielding the BiVO4-MOF-N2 catalyst. The MOF-derived co-catalyst significantly enhanced the photocurrent density of the BiVO4-MOF-N2 photocathode to 2.32 mA cm−2 (1.23 V vs. RHE), approximately 2.64 times that of the pristine BiVO4. Compared to the pure MOF co-catalyst, the MOF-derived co-catalyst exhibits higher charge transfer efficiency and lower charge transfer resistivity during the charge transfer process from BiVO4 to the cobalt co-active center, due to the excellent conductivity of the carbon matrix.
The formation of photocatalytic products is influenced by various factors, including light conditions, temperature, pH, and different coupling reactions.189–197 The formation of products in photoelectrocatalysis is significantly affected by light intensity and spectral distribution. Intense light increases the rate of photogenerated charge production, thus improving the efficiency of photoelectrocatalysis. However, excessive light can also cause problems such as photocorrosion and thermal effects, which can negatively affect the photoelectrocatalytic process. Therefore, it is crucial to select appropriate light conditions to optimize product generation. Therefore, selecting appropriate light conditions and designing suitable catalysts are critical for optimizing product formation. For example, metal sulfide photocatalysts are typically unsuitable as single-particle photocatalysts for water splitting due to photodegradation. Therefore, Iwashina et al.198 utilized various metal sulfides as hydrogen-evolving photocatalysts and RGO-TiO2 composites as oxygen-evolving photocatalysts for Z-diagram water splitting. When unloaded CuGaS2 was combined with RGO-TiO2 composites, H2 and O2 were not released in stoichiometric amounts, indicating that H2 release was primarily due to photocorrosion. Pt particles loaded via adsorption served as effective hydrogen release sites, enabling H2 and O2 to be released in stoichiometric amounts while mitigating photocorrosion. Temperature has an important effect on the efficiency of PEC hydrolysis. The PEC efficiency of MOF materials can be effectively optimized by adjusting the temperature. The effect of temperature on the formation of photoelectrocatalytic products is mainly characterized by their influence on the adsorption properties of the reactants and the potential alteration of the chemical reaction pathways. In some cases, lower temperatures may lead to higher product selectivity, while higher temperatures may promote chemical reactions. The pH of the solution also has a significant effect on the photoelectrochemical water splitting process. The normal operation of a PEC cell requires the electrolyte to be in a specific pH environment, so the equilibrium state of the PEC water splitting reaction will be largely regulated by the pH of the electrolyte solution. When the solution pH changes, the species adsorbed on the electrode surface and their charge states also change, which in turn affects the rate and efficiency of the PEC water splitting reaction. Variations in pH also can cause corrosion of the photo-electrode materials. So it is necessary to regulate the pH of the electrolyte and to design catalysts that are stable over a wide range of pH values.199 The coupling reactions of hydrogen evolution reactions also affect the efficiency of water splitting. Hydrogen production can be promoted by replacing the slow OER with a thermodynamically more favorable reaction. For example, Zhou et al.200 used the oxidation reaction of benzyl alcohol to benzaldehyde to replace the OER reaction. On a Bi2MoO6@TiO2 NTA photocathode, the selectivity of benzyl alcohol conversion to benzaldehyde reaches 100%. The electrons generated in this controlled and selective one-step oxidation reaction are used as a source for synergistic hydrogen production. The hydrogen production rate is 5.5 times higher than when using the OER. The efficiency of this hydrogen production process reaches up to 85%. Photoelectrocatalytic electrode reactions and product generation processes are complex and involve multiple factors and processes. An in-depth understanding of the mechanism of photoelectrocatalytic electrode reactions and an in-depth study of the mechanism of photoelectrocatalytic reactions can help to improve the efficiency of photoelectrocatalysis.
5.4. Stability
In photocatalytic processes, catalyst stability is compromised by photodegradation, dissolution of active sites, or structural collapse. Therefore, catalyst stability is a key parameter for evaluating the performance of photocatalysts and is also critical to their reliability in practical applications. Research into stability enhancement strategies (such as ligand engineering, controlling the coordination environment, and adding co-catalysts) can lead to the development of highly stable photocatalysts. | ||
| Fig. 23 Photoelectrocatalytic OER activity, PL intensity and band gaps. (a) and (b) In situ electrochemical photoluminescence spectroscopy (a) and band potential diagram (b) of A-ZIF with 100 CV cycles at 0.85–1.55 V. (c) Amperometric plots and O2 evolution rates of A-ZIF and AE-ZIF at certain potentials of 1.65 V and visible light. (d) and (e) Band potential diagram (d) and in situ electrochemical PL spectroscopy (e) of AE-ZIF with 100 CV cycles at 0.85–1.55 V. Copyright 2024, Elsevier.201 Copyright 2024, Nature. | ||
BiVO4 is considered a promising photoanode material due to its suitable energy band structure. After years of development, although the oxygen precipitation activity of BiVO4 photoanodes has been dramatically improved, the poor photostability of BiVO4 photoanodes due to the dissolution of V5+ ions during photoelectrocatalytic processes has become a bottleneck challenge that restricts its transition from the laboratory to practical applications. The main challenge of BiVO4 photoanodes lies in their photocorrosion problems, especially the V5+ dissolution leading to decreased stability. Xu et al.173 designed Co3O4/CoV-MOF/BiVO photoanode catalysts, which, due to the presence of ligand defects and the formation of a large number of unpaired sites in CoV-MOF, are able to enhance BiVO4 surface V5+ bonding between V5+ ions and CoV-MOF, thus effectively inhibiting the dissolution of V5+ in BiVO4 during the hydrolysis process, and significantly improving photostability, stable operation for 500 h at 0.6 VRHE. The interfacial V–O coordination bonds of PEC-0 h without ligand defects and PEC-20 h with ligand defects are shown in Fig. 24a and b, respectively, where the carboxyl group in CoV-MOF interacts with the oxygen vacancies in the vanadium–oxygen tetrahedra of BVO to form a V–O coordination bond. As shown in Fig. 24c and d, the interfacial V–O bond of PEC-20 h has a shorter bond length of about 1.92 nm compared with that of PEC-0 h. Bader charge analysis of the O atoms also indicates that the electron transfer in PEC-20 h (0.48 e) is higher than that in PEC-0 h (0.05 e). As shown in Fig. 21e and f, PEC-20 h with ligand defects showed a larger V–O binding energy of 2.44 eV. In contrast, the V–O binding energy of PEC-0 h was only 0.87 eV, which is consistent with the V–O bond length. Fig. 24g–j show the localized density of states (DOS) and the corresponding pCOHP of the V–O bonds of PEC-0 and PEC-20 h. It is clear that the V–O bond of PEC-20 h exhibits a larger overlap of the density of states, which implies that the interaction between V and O atoms is stronger. The pCOHP effectively reflects the charge distribution and chemical bonding properties. The more positive the pCOHP value is below the Fermi energy level, the more bonding states there are and the stronger the bonds are. In Fig. 24i and j, PEC-20 h shows stronger bonding states. Thus, the PEC treatment leads to ligand defects in CoV-MOF, effectively inhibits the dissolution of V atoms from the BVO lattice and leads to ultra-stable hydrolysis properties.
 | ||
| Fig. 24 Understanding the mechanisms of improved stability through PEC treatment. (a) and (b) Schematic diagrams of the interaction between different types of MOF molecules and BVO surface, where (a) is the CoV-MOF without ligand defects (PEC-0 h) and (b) is the CoV-MOF with ligand defects (PEC-20 h). Schematic diagrams of the electron transfer and bond length on interfacial V–O coordinated bonds for (c) PEC-0 h and (d) PEC-20 h. The interfacial V–O bond energy of (e) PEC-0 h and (f) PEC-20 h. Local DOS plots of (g) PEC-0 h and (h) PEC-20 h, and corresponding -pCOHP of V–O bonds for (i) PEC-0 h and (j) PEC-20 h.108 Copyright 2025, ACS. | ||
 | ||
| Fig. 25 Schematic presentation of the reaction process of overall water splitting with the PUM sample under visible light irradiation.202 | ||
6. Conclusion and prospects
Recent advancements in the utilization of MOFs and derivatives for the photoelectrocatalytic splitting of water are reviewed. Studies have demonstrated the robust catalytic activity of MOFs and their derivatives in this process. Various critical factors influencing photoelectrocatalytic hydrogen production are extensively analyzed, including light absorption and utilization, charge separation and transport, surface activity and catalyst stability. Despite the promising prospects of this technology, it is confronted with certain challenges and constraints.1. Although MOFs are a class of crystalline porous materials with tunable pore sizes, they have been shown to be highly suitable for integrating different catalytic active species into the pore space to enhance catalytic activity. For example, MOFs can be used to accommodate metal nanoclusters. However, controlling the size of MOF pores is challenging.203 Furthermore, photocatalytic water splitting is a liquid-phase dynamic process, and the accessibility of active sites is a critical parameter, with the diffusion of reactants being limited by the inherent small pore size. Therefore, it is essential to exploit the potential of pore engineering to improve the performance of MOFs in diverse liquid-phase applications.204
2. The bandgap of MOF is usually wide and only absorbs UV light with low utilization of visible light, limiting the solar energy conversion efficiency.
3. Certain MOFs are prone to decomposition in aqueous solutions or when exposed to light, particularly becoming significantly deactivated over prolonged reaction times, and specific organic ligands being easily degraded by additional oxidants. Consequently, there exists an urgent need for the development of high-efficiency and stable MOF photocatalysts.
4. The high interfacial resistance between the organic ligands and inorganic nodes of MOF makes the photogenerated electron–hole pairs easy to be compounded, resulting in short carrier lifetime.
5. Scaling up the synthesis of MOFs from laboratory scale to industrial production poses significant challenges, primarily due to their high production costs and poor reproducibility. Therefore, corresponding measures need to be taken to promote the commercialization and industrialization of MOFs.
Future direction of development:
The key to photocatalytic hydrogen production technology is to improve the efficiency of photocatalysis and hydrogen yield. To meet this challenge, significant efforts are still needed. MOF materials used for water splitting can be explored in the following aspects, as shown in Fig. 26.
1. In situ characterization: characterizing photocatalytic and photoelectrocatalytic mechanisms under in situ conditions is intricate. Employing time-resolved spectroscopy and operando techniques is essential to dynamically monitor the reaction interface process. This approach allows for a comprehensive and precise understanding of the role of MOF-based materials in the photoelectrocatalytic mechanism. Investigating photogenerated charge generation, transfer, and reaction processes, is conducive to elucidating the kinetic mechanism of photoelectrocatalytic hydrogen production reactions, offering a more scientifically grounded foundation for optimizing reaction conditions. These studies will serve as a theoretical roadmap for designing highly efficient catalysts.
2. Develop next-generation MOFs/MOF-derived photocatalysts with higher activity. Design strategies: (1) precise control: precisely control the coordination patterns of metal nodes (coordination number, geometric configuration) and organic ligands (conjugated nature, connectivity) to precisely coordinate and regulate the charge transfer pathways of MOFs. (2) Multilevel pore structure design. Pores of different sizes serve distinct functions, e.g., micropores (<2 nm) provide active sites, mesopores (2–50 nm) facilitate mass transfer, and macropores (>50 nm) enhance light penetration.205–207 This can enhance activity by adjusting the pore structure of MOFs. (3) Heterostructure construction and interface engineering: Improve carrier separation efficiency. (4) Interdisciplinary integration: drawing inspiration from artificial photosynthesis systems (e.g., liposome-MOF) and energy materials (e.g., perovskite solar cells) to advance the development of novel photoanodes.208,209 (5) Development of co-catalysts: some high-performance co-catalysts can significantly enhance the efficiency of photocatalytic hydrogen production. For example, when metal oxides such as MnOx form heterojunction structures with MOF catalysts and their derivatives, electrons and holes accumulate on the co-catalyst and participate in redox reactions, prolonging the lifetime of photo-generated electrons and holes, thereby facilitating electron–hole separation and improving hydrogen production efficiency. Additionally, precious metals such as Pt and Pd are commonly used as co-catalysts to provide active sites that promote water splitting. In the future, the development of efficient and stable composite co-catalysts could further enhance the performance and stability of photocatalytic hydrogen production.
3. Design of novel MOFs; currently, commonly used MOF templates include zeolitic imidazolate frameworks (ZIFs), University of Oslo (UiO), and Material Institute of Lavoisier (MIL). It is necessary to explore novel MOFs with designed structures and active sites. By precisely arranging the active sites and synergistically combining them with multi-level structures, their light absorption capacity, charge separation, and migration performance can be improved.
4. The transition of MOF materials from laboratory-scale synthesis to commercial-scale production and application faces several challenges. (1) Presently, standardized protocols for the synthesis and characterization of porous materials are insufficient. The absence of standardized methods makes it challenging to compare different research results and ensure experimental reproducibility. For example, establishing databases, promoting data sharing, and enhancing research transparency can significantly improve the reproducibility of porous material research. At the same time, strict control of synthesis parameters (such as time, temperature, pressure, concentration, flow rate, specific surface area, etc.) is key to ensuring technical reproducibility and reducing costs.133 (2) Process greening: promote atmospheric pressure aqueous phase synthesis and continuous flow technology, and phase out high-energy-consuming high-pressure reactions.210,211 Raw material recycling: Establish a waste ligand/waste MOF recycling network to promote the high-value utilization of industrial waste.212
5. Development of novel photocatalytic systems: Future research must prioritize the design of innovative system architectures, optimize the integration of photocatalytic devices and systems, reduce costs, and enhance practicality. Additionally, reaction conditions must be optimized, and stringent energy efficiency metrics must be established.
Overall, metal–organic frameworks (MOFs) and derivatives offer unique advantages not present in conventional materials, making them a promising platform for advancing photocatalysis and photoelectrocatalysis in energy and environmental contexts. The continuous technological progress enabling a more comprehensive exploration of these materials underscores their potential for future applications in energy and beyond.
Conflicts of interest
There are no conflicts to declare.Data availability
No primary research results, software or code have been included and no new data were generated or analysed as part of this review.Acknowledgements
This work is financially supported by the National Natural Science Foundation of China (22479161) and the Shenzhen Science and Technology Program (JCYJ20240813151113018) and the Fundamental Research Funds for the Central Universities (24CX03012A).References
- X. W. Lv, W. W. Tian and Z. Y. Yuan, Electrochem. Energy Rev., 2023, 6, 23 CrossRef CAS.
- J. G. Hou, Y. Z. Wu, B. Zhang, S. Y. Cao, Z. W. Li and L. C. Sun, Adv. Funct. Mater., 2019, 29, 1808367 CrossRef.
- Y. J. Li, L. Zhou and S. J. Guo, EnergyChem., 2021, 3, 100053 CrossRef CAS.
- G. Chen, C. Wei, Y. P. Zhu and H. T. Huang, EcoMat., 2023, 5, e12294 CrossRef CAS.
- X. Xiao, L. J. Yang, W. P. Sun, Y. Chen, H. Yu, K. K. Li, B. H. Jia, L. Zhang and T. Y. Ma, Small, 2022, 18, 2105830 CrossRef CAS PubMed.
- Z. X. Li, M. L. Hu, P. Wang, J. H. Liu, J. S. Yao and C. Y. Li, Coord. Chem. Rev., 2021, 439, 213953 CrossRef CAS.
- C. L. Luo, X. J. Liu, K. Yang, J. Xu, Z. J. Zhu, Z. H. Tang, S. L. Shen, J. C. Fan, D. Luo, N. A. H. Alshammari, Z. M. El-Bahy, X. T. Xu, Y. H. Xue and L. K. Pan, Coord. Chem. Rev., 2024, 516, 215936 CrossRef CAS.
- H. S. Su, X. X. Chang and B. J. Xu, Chin. J. Catal., 2022, 43, 2757–2771 CrossRef CAS.
- S. T. Nishioka1, F. E. Osterloh, X. C. Wang, T. E. Mallouk and K. Maeda, Nat. Rev. Methods Primers, 2023, 3, 42 CrossRef.
- S. Lin, H. W. Huang, T. Y. Ma and Y. H. Zhang, Adv. Sci., 2021, 8, 20024258 Search PubMed.
- C. F. Fu, X. J. Wu and J. L. Yang, Adv. Mater., 2018, 30, 1802106 CrossRef PubMed.
- K. Sun, Y. Y. Qian and H. L. Jiang, Angew. Chem., Int. Ed., 2023, 62, e202217565 CrossRef CAS PubMed.
- C. B. Bie, L. X. Wang and J. G. Yu, Chem, 2022, 8, 1567–1574 CAS.
- V. H. Dang, T. A. Nguyen, M. V. Le, D. Q. Nguyen, Y. H. Wang and J. C. S. Wu, Chem. Eng. J., 2024, 484, 149213 CrossRef CAS.
- L. H. Lin, T. Hisatomi, S. S. Chen, T. Takata and K. Domen, Trends Chem., 2020, 2, 813–824 CrossRef CAS.
- T. T. Yao, X. R. An, H. X. Han, J. Q. J. Chen and C. Li, Adv. Energy Mater., 2018, 8, 1800210 CrossRef.
- J. Z. Jia, Y. T. Wang, Y. Y. Cha, Z. W. Wang, J. F. Huang, D. N. Wang, H. Li, K. L. Guo, J. Li, J. E. Huang, Y. Tang and C. L. Xu, Adv. Funct. Mater., 2025, 2500568 CrossRef CAS.
- K. Poonia, P. Singh, A. Singh, A. Singh, S. Thakur, Q. V. Le, T. Ahamad, P. Raizada, C. Y. Wang, L. H. Nguyen and V. H. Nguyen, Environ. Chem. Lett., 2023, 21, 265–283 CrossRef CAS.
- Y. Huang, L. Zou, Y. B. Huang and R. Cao, Chin. J. Catal., 2025, 70, 207–229 CrossRef.
- L. Y. Yang, F. Li and Q. J. Xiang, Mater. Horiz., 2024, 11, 1638–1657 RSC.
- D. Liu, X. Y. Wan, T. T. Kong, W. W. Hanc and Y. J. Xiong, J. Mater. Chem. A, 2022, 10, 5878–5888 RSC.
- Z. R. Liu, X. Yu and L. L. Li, Chin. J. Catal., 2020, 41, 534–549 CrossRef CAS.
- L. V. Bora and N. V. Bora, Fuel, 2025, 381, 133642 CrossRef CAS.
- T. T. Wu, G. Q. Deng and C. Zhen, J. Mater. Sci. Technol., 2021, 73, 9–22 CrossRef CAS.
- C. Y. Toe, S. J. Zhou, M. Gunawan, X. X. Lu, Y. H. Ng and R. Amal, J. Mater. Chem. A, 2021, 9, 20277–20319 RSC.
- K. R. G. Lim, A. D. Handoko, S. K. Nemani, B. Wyatt, H. Y. Jiang, J. W. Tang, B. Anasori and Z. W. She, ACS Nano, 2020, 14, 10834–10864 CrossRef CAS PubMed.
- T. Tong, M. M. Zhang, W. F. Chen, X. Q. Huo, F. H. Xu, H. C. Yan, C. Lai, W. J. Wang, S. Y. Hu, L. Qin and D. L. Huang, Coord. Chem. Rev., 2024, 500, 215498 CrossRef CAS.
- F. Qiao, Nano Res. Energy, 2025, 4, e9120132 CrossRef.
- W. W. Li, J. L. Shi, K. H. L. Zhang and J. L. MacManus-Driscoll, Mater. Horiz., 2020, 7, 2832–2859 RSC.
- Y. Zhang, Y. Y. Zhou, D. L. Sun, Y. X. Nie, D. Y. Wu, L. Ban, B. Tang, S. Yang, H. Li, T. Y. Ma and H. Zhang, Coord. Chem. Rev., 2025, 527, 216395 CrossRef CAS.
- S. Ali, S. H. Bakhtiar, A. Ismail, P. M. Ismail, S. Hayat, A. Zada, X. Q. Wu, A. N. Alodhayb, M. Zahid, F. Raziq, J. B. Yi and L. Qiao, Coord. Chem. Rev., 2025, 523, 216237 CrossRef CAS.
- X. D. Wang, J. L. Xie and C. M. Li, J. Mater. Chem. A, 2015, 3, 1235–1242 RSC.
- C. H. Jiang, Y. H. Zheng, W. G. Yang and F. R. Wan, J. Univ. Sci. Technol., 2007, 29(2), 139–141 CAS.
- H. An, B. Lin, C. Xue, X. Q. Yan, Y. Z. Dai, J. J. Wei and G. D. Yang, Chin. J. Catal., 2018, 39, 654–663 CrossRef CAS.
- G. J. Liu, S. Ye, P. L. Yan, F. Q. Xiong, P. Fu, Z. L. Wang, Z. Chen, J. Y. Shi and C. Li, Energy Environ. Sci., 2016, 9, 1327–1334 RSC.
- B. P. Mishraa and K. Parida, J. Mater. Chem. A, 2021, 9, 10039–10080 RSC.
- L. Mao, X. Y. Cai, H. Gao, X. G. Diao and J. Y. Zhang, Nano Energy, 2017, 39, 172–182 CrossRef CAS.
- S. Mondal, G. Mark, L. Abisdris, J. Y. Li, T. Shmila, J. Tzadikov, M. Volokh, L. D. Xing and M. Shalom, Mater. Horiz., 2023, 10, 1363–1372 RSC.
- W. W. Yu, T. Zhang and Z. K. Zhao, Appl. Catal., B, 2020, 278, 119342 CrossRef CAS.
- M. J. Wang, M. Langer, R. Altieri, M. Crisci, S. Osella and T. Gatti, ACS Nano, 2024, 18(13), 9245–9284 CrossRef CAS PubMed.
- M. Ali, A. S. Anjum, A. Bibi, S. Wageh, K. C. Sun and S. H. Jeong, Carbon, 2022, 196, 649–662 CrossRef CAS.
- H. Xiao, W. X. Wang, M. Zhao, Z. M. Fu, M. Bai, L. Zhang, J. M. Zhang, E. G. Luo, J. Zhang, H. S. Wu and J. F. Jia, Chem. Eng. J., 2024, 490, 151415 CrossRef CAS.
- Y. d Pan, R. Abazari, B. Tahir, S. Sanati, Y. C. Zheng, M. Tahir and J. K. Gao, Coord. Chem. Rev., 2024, 499, 215538 CrossRef CAS.
- Y. J. He, W. Liu and J. Q. Liu, J. Colloid Interface Sci., 2024, 661, 409–435 CrossRef CAS PubMed.
- Z. Wang, X. D. Jin, L. P. Yan, Y. Z. Yang and X. G. Liu, Microchim. Acta, 2023, 190, 28 CrossRef CAS PubMed.
- D. d Hu, R. T. Guo, J. S. Yan, S. H. Guo and W. G. Pan, Nanoscale, 2024, 16, 2185–2219 RSC.
- X. L. Li, S. S. Rajasree, J. R. Yu and P. Deria, Dalton Trans., 2020, 49, 12892–12917 RSC.
- A. Thiam, J. A. Lopez-Ruiz, D. Barpaga and S. Garcia-Segura, Molecules, 2021, 26, 5713 CrossRef CAS PubMed.
- H. L. Zhu, D. X. Liu, D. T. Zou and J. Y. Zhang, J. Mater. Chem. A, 2018, 6, 6130–6154 RSC.
- A. García-Sánchez, M. Gomez-Mendoza, M. Barawi, I. J. Villar-Garcia, M. Liras, F. Gándara and V. A. de la Peña O’Shea, J. Am. Chem. Soc., 2020, 142(1), 318–326 CrossRef PubMed.
- Q. K. Shi, M. Cheng, Y. Liu, J. Wang, G. X. Zhang, L. Li, L. Du, G. F. Wang and H. D. Liu, Coord. Chem. Rev., 2024, 499, 215500 CrossRef CAS.
- J. Patel, G. Bury and Y. L. Pushkar, Small, 2024, 20, 2310106 CrossRef CAS PubMed.
- J. Wang, C. Xue, W. Q. Yao, J. Liu, X. X. Gao, R. L. Zong, Z. Yang, W. J. Jin and D. P. Tao, Appl. Catal., B, 2019, 250, 369–381 CrossRef CAS.
- R. Ifraemov, R. Shimoni, W. H. He, G. M. Penga and I. Hod, J. Mater. Chem. A, 2019, 7, 3046–3053 RSC.
- S. Gulati, S. Vijaya, Mansi, S. Kumar, B. Harikumar, M. Trivedi and R. S. Varma, Coord. Chem. Rev., 2023, 474, 214853 CrossRef CAS.
- H. Furukawa, K. E. Cordova, M. O'Keeffe and O. M. Yaghi, Science, 2013, 341, 1230444 CrossRef PubMed.
- H. Wang, X. Yuan, Y. Wu, X. Chen, L. Leng and G. Zeng, J. Hazard. Mater., 2015, 300, 393–403 Search PubMed.
- M. Ali, E. Pervaiz, T. Noor, O. Rabi, R. Zahra and M. H. Yang, Int. J. Energy Res., 2020, 1–37 Search PubMed.
- L. V. Bora and N. V. Bora, Fuel, 2025, 381, 133642 CrossRef CAS.
- F. H. Mu, B. L. Dai, W. Zhao, L. L. Zhang, J. M. Xu and X. J. Guo, Chin. Chem. Lett., 2020, 31, 1773–1781 CrossRef CAS.
- L. Y. Yang, F. Li and Q. J. Xiang, Mater. Horiz., 2024, 11, 1638 RSC.
- T. T. Yao, X. R. An, H. X. Han, J. Q. J. Chen and C. Li, Adv. Energy Mater., 2018, 8, 1800210 CrossRef.
- J. Liu, W. Fu, Y. Liao, J. Fan and Q. Xiang, J. Mater. Sci. Technol., 2021, 91, 224–240 CrossRef CAS.
- Y. Li, D. Zhang, J. Fan and Q. Xiang, Chin. J. Catal., 2021, 42, 627–636 CrossRef CAS.
- M. A. Marwat, M. Humayun, M. W. Afridi, H. Zhang, M. R. Abdul Karim and M. Ashtar, ACS Appl. Energy Mater., 2021, 4, 12007–12031 CrossRef CAS.
- F. Li, X. Yue, L. Cheng, J. Fan and Q. Xiang, Chem. Eng. J., 2023, 452, 139463 CrossRef CAS.
- J. Chen, R. Abazari, K. A. Adegoke, N. W. Maxakato, O. S. Bello, M. Tahir, S. Tasleem, S. Sanati, A. M. Kirillov and Y. T. Zhou, Coord. Chem. Rev., 2022, 469, 214664 CrossRef CAS.
- S. Cho, J. W. Jang, K. H. Lee and J. S. Lee, APL Mater., 2014, 2, 010703 CrossRef.
- J. J. Cen, Q. Y. Wu, M. Z. Liu and A. Orlov, Green Energy Environ., 2017, 2(2), 100–111 CrossRef.
- R. T. Gao, X. T. Guo, S. J. Liu, X. Y. Zhang, X. H. Liu, Y. G. Su and L. Wang, Appl. Catal., B, 2022, 304, 120883 CrossRef CAS.
- Z. Lin, Y. Q. Wang, Z. M. Peng, Y. C. Huang, F. Q. Meng, J. L. Chen, C. L. Dong, Q. H. Zhang, R. Z. Wang, D. M. Zhao, J. Chen, L. Gu and S. H. Shen, Adv. Energy Mater., 2022, 2200716 CrossRef CAS.
- L. T. Shi, C. C. Wu, Y. Wang, Y. H. Dou, D. Yuan, H. Li, H. W. Huang, Y. Zhang, I. D. Gates, X. D. Sun and T. Y. Ma, Adv. Funct. Mater., 2022, 2202571 CrossRef CAS.
- W. W. Zhang, L. J. Chen, S. Dai, C. X. Zhao, C. Ma, L. Wei, M. H. Zhu, S. Y. Chong, H. F. Yang, L. J. Liu, Y. Bai, M. J. Yu, Y. J. Xu, X. W. Zhu, Q. Zhu, S. H. An, R. S. Sprick, M. A. Little, X. F. Wu, S. Jiang, Y. Z. Wu, Y. B. Zhang, H. Tian, W. H. Zhu and A. I. Cooper, Nature, 2022, 604, 72 CrossRef CAS PubMed.
- T. Bak, J. Nowotny, M. Rekas and C. C. Sorrell, Int. J. Hydrogen Energy, 2002, 27, 991–1022 CrossRef CAS.
- Y. W. Dong, H. J. Liu, X. Wang, F. L. Wang, X. Y. Zhang, Q. X. Lv, C. Y. Liu, Y. M. Chai and B. Dong, Appl. Catal., B, 2023, 328, 122464 CrossRef CAS.
- M. Li, Q. Wang, Y. He, Y. Wang, K. Wang, P. Tsiakaras and S. Song, Coord. Chem. Rev., 2023, 495, 215373 CrossRef CAS.
- N. E. Gorji, U. Reggiani and L. Sandrolini, Sol. Energy, 2012, 86(3), 920–925 CrossRef CAS.
- H. Dotan, K. Sivula, M. Grätzel, A. Rothschild and S. C. Warren, Energy Environ. Sci., 2011, 4(3), 958–964 RSC.
- B. K. Kim, M. J. Kim and J. J. Kim, Appl. Catal., B, 2022, 308, 121226 CrossRef CAS.
- G. Heidari, M. Rabani and B. Ramezanzadeh, Int. J. Hydrogen Energy, 2017, 42, 9545–9552 CrossRef CAS.
- F. A. Chaves, P. C. Feijoo and D. Jimenez, Nanoscale Adv., 2020, 2, 3252–3262 RSC.
- S. Hamrouni, M. S. AlKhalifah, M. S. El-Bana, S. K. Zobaidi and S. Belgacem, Appl. Phys. A: Mater. Sci. Process., 2018, 124, 555 CrossRef.
- G. Kear and F. C. Walsh, The characteristics of a true Tafel slope, Corrosion Mater., 2005, 30(6), 51–55 Search PubMed.
- A. J. Bard, L. R. Faulkner, J. Leddy and C. G. Zoski, Electrochemical Methods: Fundamentals and Applications, Wiley, New York, NY, 1980, vol. 2 Search PubMed.
- Y. N. Guo, T. Park, J. W. Yi, J. Henzie, J. Kim, Z. L. Wang, B. Jiang, Y. Bando, Y. Sugahara, J. Tang and Y. Yamauchi, Adv. Mater., 2019, 31(17), 1807134 CrossRef PubMed.
- Y. Zhang, H. Lv, Z. Zhang, L. Wang, X. Wu and H. Xu, Adv. Mater., 2021, 33, 2008264 CrossRef CAS PubMed.
- M. Tahir, L. Pan, F. Idrees, X. W. Zhang, L. Wang, J. J. Zou and Z. L. Wang, Nano Energy, 2017, 37, 136–157 CrossRef CAS.
- J. H. Li, Y. S. Wang, Y. C. Chen and C. W. Kung, Appl. Sci., 2019, 9(12), 2427 CrossRef CAS.
- W. W. Zhan, Q. Kuang, J. Z. Zhou, X. J. Kong, Z. X. Xie and L. S. Zheng, J. Am. Chem. Soc., 2013, 135(5), 1926–1933 CrossRef CAS PubMed.
- T. Y. Ma and S. Dai, J. Am. Chem. Soc., 2014, 136(39), 13925–13931 CrossRef CAS PubMed.
- M. Ding, X. Cai and H. L. Jiang, Chem. Sci., 2019, 10(44), 10209–10230 RSC.
- M. Fujita, Y. J. Kwon, S. Washizu and K. Ogura, J. Am. Chem. Soc., 1994, 116(3), 1151–1152 CrossRef CAS.
- M. Alvaro, E. Carbonell, B. Ferrer, F. X. Llabrés i Xamena and H. Garcia, Chem. – Eur. J., 2007, 13, 5106–5112 CrossRef CAS PubMed.
- Z. T. Yu, Z. L. Liao, Y. S. Jiang, G. H. Li, G. D. Li and J. S. Chen, Chem. Commun., 2004, 1814–1815 RSC.
- P. Mahata, G. Madras and S. Natarajan, J. Phys. Chem. B, 2006, 110, 13759–13768 CrossRef CAS PubMed.
- S. J. Liu, C. Zhang, Y. D. Sun, Q. Chen, L. F. He, K. Zhang, J. Zhang, B. Liu and L. F. Chen, Coord. Chem. Rev., 2020, 413, 213266 CrossRef CAS.
- B. Y. Chen, T. D. Hu, S. Y. Shan, Y. F. Zhi, C. R. Zhang and Q. Wu, Acta Mater. Compositae Sin., 2022, 39, 2073–2088 CAS.
- Q. Q. Ge, M. Xu, X. Liang and F. W. Wang, Chem. Ind. Eng. Prog., 2023, 42, 4692–4705 CAS.
- C. Pan, J. Y. Chao, F. S. Niu, S. H. Xie, H. Y. Gu, T. H. Su, K. Hu, D. W. Zhang, K. Liu, G. F. Liu, T. F. Xie, Z. T. Li and L. M. Zhang, Adv. Mater. Interfaces, 2022, 9, 2101678 CrossRef CAS.
- J. Kosco, M. Bidwell, H. Cha, T. Martin, C. T. Howells, M. Sachs, D. H. Anjum, S. G. Lopez, Y. Zou, A. Wadsworth, W. M. Zhang, L. S. Zhang, J. Tellam, R. Sougrat, F. Laquai, D. M. DeLongchamp, J. R. Durrant and I. McCulloch, Nat. Mater., 2020, 19, 559–565 CrossRef CAS PubMed.
- X. F. Wu, D. S. Chen, W. Ma and K. K. Huang, Chin. J. Appl. Chem., 2022, 39(4), 694–696 CAS.
- W. Liu, D. Y. Liu, Z. Y. Zhang and Z. K. Sun, Particuology, 2024, 91, 19–28 CrossRef CAS.
- M. ChoeJin, Y. Koo, I. Park, H. Ohtsu, J. H. Shim, H. C. Choi and S. S. Park, J. Am. Chem. Soc., 2022, 144(37), 16726–16731 CrossRef PubMed.
- L. T. Yu, J. Liu, X. J. Xu, L. G. Zhang, R. Z. Hu, J. W. Liu, L. Z. Ouyang, L. C. Yang and M. Zhu, ACS Nano, 2017, 11(5), 5120–5129 CrossRef CAS PubMed.
- F. J. Claire, M. A. Solomos, J. Kim, G. Q. Wang, M. A. Siegler, M. F. Crommie and T. J. Kempa, Nat. Commun., 2020, 11, 5524 CrossRef CAS PubMed.
- S. J. Xie, Z. Y. Zhou, X. Zhang and J. Fransaer, Chem. Soc. Rev., 2023, 52, 4292–4312 RSC.
- Q. Liu, Y. R. Miao, L. F. Villalobos, S. X. Li, H. Y. Chi, C. L. Chen, M. Tohidi Vahdat, S. Q. Song, D. J. Babu, J. Hao, Y. Han, M. Tsapatsis and K. V. Agrawal, Nat. Mater., 2023, 22, 1387–1393 CrossRef CAS PubMed.
- D. Balestri, Y. Roux, M. Mattarozzi, C. Mucchino, L. Heux, D. Brazzolotto, V. Artero, C. Duboc, P. Pelagatti, L. Marchi and M. Gennari, Inorg. Chem., 2017, 56(24), 14801–14808 CrossRef CAS PubMed.
- X. F. Li, M. Y. Lu, H. Y. Yu, T. H. Zhang, J. Liu, J. H. Tian and R. Z. Yang, ChemElectroChem, 2019, 6(17), 4507–4510 CrossRef CAS.
- B. Gikonyo, F. B. Liu, S. Hawila, A. Demessence, H. G. Baldovi, S. Navalón, C. Marichy and A. Fateeva, Molecules, 2023, 28(15), 5876 CrossRef CAS PubMed.
- W. M. A. El Rouby, M. Antuch, S. M. You and P. Millet, Electrochim. Acta, 2020, 339, 135882 CrossRef CAS.
- P. Sindhu and N. Ballav, Inorg. Chem., 2023, 62(28), 10887–10891 CrossRef CAS PubMed.
- C. Young, J. Wang, J. Kim, Y. Sugahara, J. Henzie and Y. Yamauchi, Chem. Mater., 2018, 30(10), 3379–3386 CrossRef CAS.
- Y. W. Dong, X. J. Zhai, Y. Wu, Y. N. Zhou, Y. C. Li, J. Nan, S. T. Wang, Y. M. Chai and B. Dong, J. Colloid Interface Sci., 2024, 658, 258–266 CrossRef CAS PubMed.
- D. Cardenas-Morcoso, R. Ifraemov, M. García-Tecedor, I. Liberman, S. Gimenez and I. Hod, J. Mater. Chem. A, 2019, 7, 11143–11149 RSC.
- R. Tang, S. J. Zhou, L. Y. Zhang and L. W. Yin, Adv. Funct. Mater., 2018, 28, 1706154 CrossRef.
- S. Q. Zhou, P. F. Yue, J. W. Huang, L. Wang, H. D. She and Q. Z. Wang, Chem. Eng. J., 2019, 371, 885–892 CrossRef CAS.
- D. B. Bailmare, B. V. Malozyomov and A. D. Deshmukh, Commun. Chem., 2024, 7, 178 CrossRef CAS PubMed.
- Y. J. Tang, H. Zheng, Y. Wang, W. Zhang and K. Zhou, Adv. Funct. Mater., 2021, 31, 2102648 CrossRef CAS.
- J. J. Feng, W. W. Li, T. X. Chen, Z. P. Zeng, M. Tian, W. X. Ji, Y. Guo, S. X. Min and X. Y. Liu, Adv. Sci., 2025, 12, 2411673 CrossRef CAS PubMed.
- M. Q. Xu, R. Zhang, J. He, Z. Y. Chen, J. C. Jiang, Z. X. Li, Y. J. Liu, D. Chen and Y. H. Ma, Sep. Purif. Technol., 2025, 354(5), 129093 CrossRef CAS.
- M. S. Xu, P. F. Zhu, Q. H. Cai, M. C. Bu, C. H. Zhang, H. Wu, Y. Z. He, M. Fu, S. Q. Li and X. Y. Liu, Chin. Chem. Lett., 2024, 35(10), 109524 CrossRef CAS.
- K. G. Tian, Z. Xu, H. Yang, G. L. Chen, P. F. An, J. Zhang, S. Z. Liu and J. Q. Yan, Adv. Energy Mater., 2025, 15, 2404477 CrossRef CAS.
- X. Y. He, Y. J. Ding, Z. N. Huang, M. Liu, M. F. Chi, Z. L. Wu, C. U. Segre, C. S. Song, X. Wang and X. W. Guo, Angew. Chem., 2023, 62, e202217439 CrossRef CAS PubMed.
- E. W. Liu, Z. L. Yang, L. J. Han, Y. P. Huang and K. K. Liao, Acta Opt. Sin., 2021, 41(07), 144–151 Search PubMed.
- Y. Z. Zhang, T. Cheng, Y. Wang, W. Y. Lai, H. Pang and W. Huang, Adv. Mater., 2016, 28, 5242–5248 CrossRef CAS PubMed.
- W. B. Wu, Z. X. Yang and T. L. Guo, Chin. J. Vac. Sci. Technol., 2023, 43(7), 563–568 Search PubMed.
- Y. C. Li, J. Su, Y. Zhao, L. Feng, L. Gao, X. Y. Xu, Y. Yin, Y. Yin, Y. F. Liu, P. W. Xiao, L. Yuan, J. S. Qin, Y. Y. Wang, S. Yuan, H. Zheng and J. L. Zuo, J. Am. Chem. Soc., 2023, 145(18), 10227–10235 CrossRef CAS PubMed.
- C. Wang, C. Liu, X. He and Z. M. Sun, Chem. Commun., 2017, 53, 11670–11673 RSC.
- P. Liu, C. H. Zhou, Z. F. Ren, Q. Lin, Y. Zhou, J. Gu and Y. H. Shan, Chem. Mater., 2021, 49(12), 103–107 Search PubMed.
- S. Kouser, A. Hezam and S. A. Khanum, J. Mol. Struct., 2025, 1321, 140061 CrossRef CAS.
- W. M. Hu, Q. Ma, Y. Q. He, H. B. Liu, J. Q. Liu and X. H. Xia, Chin. J. Inorg. Chem., 2020, 36(3), 485–493 CAS.
- M. Nazari, F. Zadehahmadi, M. M. Sadiq, A. L. Sutton, H. Mahdavi and M. R. Hill, Commun. Mater., 2024, 5, 170 CrossRef.
- T. Paul, A. Juma, R. Alqerem, G. Karanikolos, H. A. Arafat and L. F. Dumee, J. Environ. Chem. Eng., 2023, 11, 111112 CrossRef CAS.
- D. Chakraborty, A. Yurdusen, G. Mouchaham, F. Nouar and C. Serre, Adv. Funct. Mater., 2024, 34, 2309089 CrossRef CAS.
- L. Zhang, J. Y. Zou and G. X. Peng, Biol. Chem. Eng., 2023, 9(03), 59–61 CAS.
- M. Taddei, D. A. Steitz, J. A. Bokhoven and M. Ranocchiari, Chem. - Eur. J., 2016, 22(10), 3245–3249 CrossRef CAS PubMed.
- S. Hübner, S. Kressirer, D. Kralisch, C. Bludszuweit-Philipp, K. Lukow, I. Jänich, A. Schilling, H. Hieronymus, C. Liebner and K. Jähnisch, ChemSusChem, 2012, 5(2), 279–288 CrossRef PubMed.
- M. Shukuya and A. Hammache, 2002: VTT Technical Research Centre of Finland Search PubMed.
- M. Sánchez-Sánchez, N. Getachew, K. Díaz, M. Díaz-García, Y. Chebude and I. Díaz, Green Chem., 2015, 17(3), 1500–1509 RSC.
- O. Shekhah, H. Wang, D. Zacher, R. A. Fische and C. Wöll, Angew. Chem., Int. Ed., 2009, 48(27), 5038–5041 CrossRef CAS PubMed.
- J. Huo, M. Brightwell, S. E. Hankari, A. Garai and D. Bradshaw, J. Mater. Chem. A, 2013, 1(48), 15220 RSC.
- W. K. Yu, M. Xu, X. Liang, J. Y. Wang, W. Y. Fang and F. W. Wang, Sep. Purif. Technol., 2022, 303, 122155 CrossRef CAS.
- L. Pan, T. Muhammad, L. Ma, Z. F. Huang, S. B. Wang, L. Wang, J. J. Zou and X. W. Zhang, Appl. Catal. Environ., 2016, 189, 181–191 CrossRef CAS.
- C. H. Li, C. L. Huang, X. F. Chuah, D. S. Raja, C. T. Hsieh and S. Y. Lu, Chem. Eng J., 2019, 361, 660–670 CrossRef CAS.
- A. Pichon, A. Lazuen-Garay and S. L. James, CrystEngComm, 2006, 8, 211–214 RSC.
- T. Friscic, D. G. Reid, I. Halasz, R. S. Stein, R. E. Dinnebier and M. J. Duer, Angew. Chem., 2010, 122, 724–727 CrossRef.
- G. Lin, B. Zeng, J. Li, Z. Y. Wang, S. X. Wang, T. Hu and L. B. Zhang, Chem. Eng. J., 2023, 460, 141710 CrossRef CAS.
- X. Y. Yang, Z. W. Chen, X. Z. Yue, X. Du, X. H. Hou, L. Y. Zhang, D. L. Chen and S. S. Yi, Small, 2023, 19, 2205246 CrossRef CAS PubMed.
- G. A. Tompsett, W. C. Conner and K. S. Yngvesson, Chem. Phys. Chem., 2006, 7, 296–319 CrossRef CAS PubMed.
- Y. R. Lee, J. Kim and W. S. Ahn, Korean J. Chem. Eng., 2013, 30, 1667–1680 CrossRef CAS.
- U. Mueller, M. Schubert, F. Teich, H. Puetter, K. Schierle-Arndt and J. Pastré, ChemInform, 2006, 16, 626–636 CAS.
- U. Müller, M. Schubert, F. Teich, H. Puetter, K. Schierle-Arndt and J. Pastré, ChemInform, 2006, 16, 626–636 Search PubMed.
- Q. Y. Wu, C. X. Zhang, K. Sun and H. L. Jiang, Acta Chim. Sin., 2020, 78, 688–694 CrossRef CAS.
- L. Cheng, D. Zhang, Y. Liao, J. Fan and Q. Xiang, Chin. J. Catal., 2021, 42, 131–140 CrossRef CAS.
- X. Yue, J. Fan and Q. Xiang, Adv. Funct. Mater., 2022, 32, 2110258 CrossRef CAS.
- X. Zhang, D. Xue, S. Jiang, H. Xia, Y. Yang, W. Yan, J. Hu and J. Zhang, InfoMat, 2022, 4, e12257 CrossRef CAS.
- Y. J. Cheng, J. Q. Zhao, X. F. Ma, H. L. Zheng, L. He, J. Zhang and Q. P. Lin, Adv. Mater., 2025, 2503756 CrossRef CAS PubMed.
- Y. W. Dong, H. J. Liu, X. Wang, F. L. Wang, X. Y. Zhang, Q. X. Lv, C. Y. Liu, Y. M. Chai and B. Dong, Appl. Catal., B, 2023, 328, 122464 CrossRef CAS.
- Y. Lu, H. X. Zhong, J. Li, A. M. Dominic, Y. M. Hu, Z. Gao, Y. L. Jiao, M. J. Wu, H. Y. Qi, C. H. Huang, L. J. Wayment, U. Kaiser, E. Spiecker, I. M. Weidinger, W. Zhang, X. L. Feng and R. H. Dong, Angew. Chem., Int. Ed., 2022, 61, e202208163 CrossRef CAS PubMed.
- C. P. Li, S. R. Li, T. X. Hang, F. F. Guo, X. D. Zhu and T. F. Liu, Small Struct., 2022, 3, 2100071 CrossRef CAS.
- Y. Q. Liu, X. Xin, Y. H. Shi, Z. F. Zhao, J. D. Tan, D. Yang and Z. Y. Jiang, Chem. Eng. J., 2024, 482, 149193 CrossRef CAS.
- R. Tang, S. J. Zhou, L. Y. Zhang and L. W. Yin, Adv. Funct. Mater., 2018, 28, 1706154 CrossRef.
- H. J. Hu, X. N. Li, K. L. Zhang, G. Yan, W. X. Kong, A. Q. Qin, Y. L. Ma, A. S. Li, K. Wang, H. W. Huang, X. D. Sun and T. Y. Ma, Adv. Mater., 2025, 2419023 CrossRef CAS PubMed.
- Y. Yang, Q. X. Zhao, Q. Zheng and W. M. Xuan, J. Donghua Univ., 2023, 40, 590–599 Search PubMed.
- R. H. Li, T. Takata, B. B. Zhang, C. Feng, Q. B. Wu, C. H. Cui, Z. M. Zhang, K. Domen and Y. B. Li, Angew. Chem., Int. Ed., 2023, 62, e202313537 CrossRef CAS PubMed.
- J. G. Hou, S. Y. Cao, Y. Q. Sun, Y. Z. Wu, F. Liang, Z. H. Lin and L. C. Sun, Adv. Energy Mater., 2018, 8, 1701114 CrossRef.
- Y. S. Chen, M. D. Cai, Y. M. Cao, S. S. A. Shuaib, J. Q. Bai, F. Chen, J. W. Xue, Y. X. Wei and S. Sun, Catal. Sci. Technol., 2025, 15, 3316–3324 RSC.
- Y. R. Song, Y. H. Ren, H. J. Cheng, Y. Y. Jiao, S. B. Shi, L. H. Gao, H. M. Xie, J. F. Gao, L. C. Sun and J. G. Hou, Angew. Chem., Int. Ed., 2023, 62, e202306420 CrossRef CAS PubMed.
- B. Y. Liu, X. Wang, Y. J. Zhang, L. C. Xu, T. S. Wang, X. Xiao, S. C. Wang, L. Z. Wang and W. Huang, Angew. Chem., Int. Ed., 2023, 62, e202217346 CrossRef CAS PubMed.
- Y. L. Xin, J. Tian, X. Q. Xiong, C. L. Wu, S. A. C. Carabineiro, X. G. Yang, Z. X. Chen, Y. Xia and Y. X. Jin, Adv. Mater., 2025, 37, 2417589 CrossRef CAS.
- J. Wang, K. Liu, W. R. Liao, Y. C. Kang, H. R. Xiao, Y. K. Chen, Q. Y. Wang, T. Luo, J. W. Chen, H. M. Li, T. S. Chan, S. Y. Chen, E. Pensa, L. Y. Chai, F. Y. Liu, L. X. Jiang, C. X. Liu, J. W. Fu, E. Cortés and M. Liu, Nat. Catal., 2025, 8, 229–238 CrossRef CAS.
- L. C. Xu, Y. J. Zhang, B. Y. Liu, K. Wan, X. Wang, T. S. Wang, L. Z. Wang, S. C. Wang and W. Huang, ACS Nano, 2025, 19, 15863–15875 CrossRef CAS PubMed.
- P. Y. Dong, J. K. Pan, L. H. Zhang, X. L. Yang, M. H. Xie and J. L. Zhang, Appl. Catal., B, 2024, 350, 123925 CrossRef CAS.
- S. M. Li, F. Wu, R. B. Lin, J. Wang, C. X. Li, Z. Q. Li, J. Jiang and Y. J. Xiong, Chem. Eng. J., 2022, 429, 132217 CrossRef CAS.
- M. Ali, E. Pervaiz, U. Sikandar and Y. Khan, Int. J. Hydrogen Energy, 2021, 46, 18257–18283 CrossRef CAS.
- D. Kumar, J. E. Abraham, M. Varghese, J. George, M. Balachandran and J. Cherusseri, Int. J. Hydrogen Energy, 2024, 50, 118–141 CrossRef CAS.
- M. Khosravi and M. R. Mohammadi, Photosynth. Res., 2022, 154, 329–352 CrossRef CAS PubMed.
- N. Joseph, P. M. Shafi and A. C. Bose, Energy Fuels, 2020, 34, 6558–6597 CrossRef CAS.
- B. Fabre, C. Falaise and E. Cadot, ACS Catal., 2022, 12, 12055–12091 CrossRef CAS.
- M. Fawaz, R. Bahadur, N. P. Dharmarajan, J. H. Yang, C. I. Sathish, A. M. Sadanandan, V. Perumalsamy, G. Singh, X. W. Guan, P. Kumar and A. Vinu, Carbon, 2023, 214, 118345 CrossRef CAS.
- S. J. Li, L. M. Zhang, W. Q. Zhao, S. H. Yuan, L. Yang, X. Q. Chen, P. Ge, W. Sun and X. B. Ji, Energy Storage Mater., 2020, 32, 477–496 CrossRef.
- E. Pervaiz, M. Ali, M. A. Abbasi, T. Noor, Z. Said and H. Alawadhi, Int. J. Hydrogen Energy, 2022, 47, 26915–26955 CrossRef CAS.
- N. Li, J. Liu, B. X. Dong and Y. Q. Lan, Angew. Chem., Int. Ed., 2020, 59, 20779–20793 CrossRef CAS PubMed.
- R. Kulkarni, L. P. Lingamdinne, R. R. Karri, Z. H. Momin, J. R. Koduru and Y. Y. Chang, Coordin. Chem. Rev., 2023, 497, 215460 CrossRef CAS.
- W. J. Liu, F. Qiao, J. Yang, J. R. Yuan, Y. Xie, T. Wang, J. Z. Hu, J. H. Zheng, R. Ren, X. M. Kang, Y. Zhao and J. W. Zhang, Nano Res., 2023, 16, 10401–10411 CrossRef CAS.
- H. H. Hu, Z. Y. Wang, L. Y. Cao, L. Z. Zeng, C. K. Zhang, W. B. Lin and C. Wang, Nat. Chem., 2021, 13, 358–366 CrossRef CAS PubMed.
- D. Wang, J. L. Gu, H. L. Wang, M. Z. Liu, Y. H. Liu and X. T. Zhang, Appl. Surf. Sci., 2023, 619, 156710 CrossRef CAS.
- Q. C. Lin, W. M. Liao, J. Y. Li, B. W. Ye, D. T. Chen, X. X. Zhou, P. H. Li, M. Li, M. D. Li and J. He, Angew. Chem., Int. Ed., 2025, 64, e202423070 CrossRef CAS PubMed.
- Z. Y. Zhou, Y. N. Xie, W. Z. Zhu, H. Y. Zhao, N. J. Yang and G. H. Zhao, Appl. Catal., B, 2021, 286, 119868 CrossRef CAS.
- S. Kim, D. S. Han and H. Park, Appl. Catal., B, 2021, 284, 119745 CrossRef CAS.
- C. Liu, D. Fu and H. Li, Environ. Technol., 2012, 33(7), 791–799 CrossRef CAS PubMed.
- J. Y. Sun, H. Maimaiti, B. Xu, L. R. Feng, J. Z. Bao and X. W. Zhao, Appl. Surf. Sci., 2022, 585, 152701 CrossRef CAS.
- Y. C. Miao and M. F. Shao, Chin. J. Catal., 2022, 43, 595–610 CrossRef CAS.
- Y. Z. Zhang, Y. R. Dai, H. H. Li, L. F. Yin and M. R. Hoffmann, Commun. Mater., 2020, 1, 66 CrossRef PubMed.
- S. Ye, W. H. Feng, J. J. Li, H. Y. Zhong, J. Y. Weng and H. Li, J. Electroanal. Chem., 2022, 927, 116975 CrossRef CAS.
- C. H. Zhou, J. H. Li, J. C. Wang, C. Y. Xie, Y. Zhang, L. Li, T. S. Zhou, J. Bai, H. Zhu and B. X. Zhou, Appl. Catal., B, 2023, 324, 122229 CrossRef CAS.
- K. Iwashina, A. Iwase, Y. H. Ng, R. Amal and A. Kudo, J. Am. Chem. Soc., 2015, 137, 604–607 CrossRef CAS PubMed.
- M. Fekete, W. Riedel, A. F. Patti and L. Spiccia, Nanoscale, 2014, 6, 7585–7593 RSC.
- Z. Y. Zhou, Y. N. Xie, W. Z. Zhu, H. Y. Zhao, N. J. Yang and G. H. Zhao, Appl. Catal., B, 2021, 286, 119868 CrossRef CAS.
- Z. A. Huang, Z. Z. Wang, H. Rabl, S. Naghdi, Q. C. Zhou, S. Schwarz, D. H. Apaydin, Y. Yu and D. Eder, Nat. Commun., 2024, 15, 9393 CrossRef CAS PubMed.
- J. J. Zhang, T. Y. Bai, H. Huang, M. H. Yu, X. B. Fan, Z. Chang and X. H. Bu, Adv. Mater., 2020, 32, 2004747 CrossRef CAS PubMed.
- H. Wang, X. K. Liu, Y. L. Zhao, Z. H. Sun, Y. Lin, T. Yao and H. L. Jiang, Nat. Sci. Rev., 2024, 11, 252 CrossRef PubMed.
- S. Naghdi, A. Cherevan, A. Giesriegl, R. Guillet-Nicolas, S. Biswas, T. Gupta, J. Wang, T. Haunold, B. C. Bayer, G. Rupprechter, M. C. Toroker, F. Kleitz and D. Eder, Nat. Commun., 2022, 13, 282 CrossRef CAS PubMed.
- K. Li, J. Yang and J. L. Gu, Acc. Chem. Res., 2022, 55(16), 2235–2247 CrossRef CAS PubMed.
- D. X. Yang, Q. Z. Xiong, Y. Wang, Y. Chen, L. B. Li and J. P. Li, Chem. Ind. Eng. Prog., 2024, 43(04), 1882–1896 CAS.
- Y. Yao, X. Y. Zhao, G. G. Chang, X. Y. Yang and B. L. Chen, Small, 2023, 4, 2200187 CrossRef CAS.
- W. G. Huang, B. Chan, Y. W. Yang, P. Chen, J. J. Wang, L. Casey, C. Atzori, T. Schulli, O. Mathon, H. G. Hackbarth, N. M. Bedford, D. Appadoo, X. M. Li, T. G. Lin, R. J. Lin, J. Lee, Z. L. Wang, V. Chen, A. K. Cheetham, L. Z. Wang and J. W. Hou, J. Am. Chem. Soc., 2025, 147, 3195–3205 CrossRef CAS PubMed.
- H. H. Hu, Z. Y. Wang, L. Y. Cao, L. Z. Zeng, C. K. Zhang, W. B. Lin and C. Wang, Nat. Chem., 2021, 13, 358–366 CrossRef CAS PubMed.
- B. B. Chen, D. Fan, R. V. Pinto, I. Dovgaliuk, S. Nandi, D. Chakraborty, N. G. Moncada, A. Vimont, C. J. McMonagle, M. Bordonhos, A. A. Mohtar, I. Cornu, P. Florian, N. Heymans, M. Daturi, G. D. Weireld, M. Pinto, F. Nouar, G. Maurin, G. Mouchaham and C. Serre, Adv. Sci., 2024, 11, 2401070 CrossRef CAS PubMed.
- D. Senthil Raja and D. H. Tsai, Chem. Commun., 2024, 60, 8497–8515 RSC.
- H. Y. Chu, Z. X. Liu, C. C. Wan and P. Wang, Chem. Commun., 2024, 60, 8350–8359 RSC.
| This journal is © The Royal Society of Chemistry 2025 |