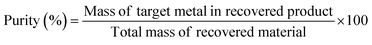Recycling and regeneration of failed layered oxide cathode materials for lithium-ion batteries
Changhao
Li
a,
Weihao
Zeng
*a,
Juan
Wang
a,
Zhongpeng
Li
a,
Jin
Zhang
a,
Xuanpeng
Wang
 b and
Shichun
Mu
b and
Shichun
Mu
 *a
*a
aState Key Laboratory of Advanced Technology for Materials Synthesis and Processing, Wuhan University of Technology, Wuhan 430070, China. E-mail: msc@whut.edu.cn
bZhongyu Feima New Material Technology Innovation Center (Zhengzhou) Co., Ltd, Zhengzhou, 45000, China
First published on 24th March 2025
Abstract
With broad usage of lithium-ion batteries (LIBs) in electronic devices and electric vehicles (EVs), a large number of decommissioned LIBs will be generated, which cause serious environmental pollution and waste of resources. Therefore, to reduce environmental pressure and realize secondary resource utilization of valuable metals, recycling decommissioned LIBs is urgent. At present, although pyrometallurgy, hydrometallurgy, direct regeneration and other methods have been used significantly in the recovery of failed LIB cathode materials, it is still necessary to formulate the best recovery strategy to achieve higher recovery efficiencies and value-added materials from spent cathodes. Thus, in this article, the latest progress in the recycling of retired LIB layered oxide cathode materials is reviewed in detail, and the recycling process, advantages and limits of each recycling method are analyzed. In view of the recycling challenges, the future development is prospected to promote the sustainable, environmentally friendly and efficient reutilization of failed LIB cathodes and contribute to the low-carbon circular economy.
Wider impactThis review systematically explores the critical developments in the recycling technologies for spent lithium-ion battery (LIB) layered oxide cathode materials. It focuses on the technical characteristics, advantages, limitations, and challenges of three primary methods: pyrometallurgy, hydrometallurgy, and direct regeneration. By integrating the latest research progress, the review highlights key unresolved issues in this field, such as the complexity of material separation, recovery efficiency, environmental impact, and industrialization potential, and proposes potential solutions. This research area has garnered significant attention primarily due to the increasing volume of spent LIBs, driven by the rapid global adoption of electrification and renewable energy applications. Resource scarcity, environmental pollution, and policy initiatives collectively position battery recycling as a critical solution for resource circularity and environmental protection, as well as a pivotal driver of the circular economy. In the future, the recycling technologies for layered oxide cathode materials will emphasize a balance among efficiency, cost-effectiveness, and environmental sustainability. Leveraging the strengths of multiple recycling methods and achieving process integration for higher efficiency will be the primary research trend. Furthermore, the development of high-value functional materials, low-energy green treatment methods, and automated industrial recycling processes is expected to further promote large-scale applications in this field. This review provides a comprehensive analysis and insights that not only serve as a valuable reference for researchers in technology optimization and application strategies but also help guide industry and policymakers in achieving synergistic development in resource management and technological innovation. Through this review, we aim to assist the field of materials science in addressing the technological bottlenecks of LIB recycling, inspire broader research discussions, and drive scientific progress in resource circularity and sustainable materials. Breakthroughs in this area will profoundly impact the interdisciplinary development of material design, energy management, and environmental protection, offering robust support for the global transition to green energy. |
1. Introduction
Over the past two decades, lithium-ion batteries (LIBs) have been widely used in 3C electronic products, electric vehicles, energy storage systems, and other fields due to their advantages of high energy density, long lifespan, light weight, high charging efficiency, and wide operating temperature range.1–3 However, this also indicates that a large number of LIBs will be retired in the future. While echelon utilization can delay the pressure of retiring power LIBs to some extent, they will inevitably become waste batteries with gradually decreasing capacity. Therefore, the key to solving the problem of spent LIBs lies in the efficient and reasonable recycling of retired LIBs.4–6 Since the cathode material accounts for more than 30% of the battery cost, it has naturally become the focus of the recycling process.According to the different commercial LIBs, the cathode materials are mainly divided into lithium iron phosphate (LFP), lithium manganate (LMO), and layered oxides including lithium cobalt (LCO) and lithium nickel cobalt manganese oxide (NCM).7,8 Among them, LCO batteries are widely used in electronic products due to their excellent cycle performance. Currently, LFP and NCM batteries dominate the automotive power battery market. LFP batteries are widely applied in electric vehicles due to their high stability and low cost. However, their low capacity fails to meet the requirements for long driving ranges in electric vehicles. This would lead to the gradual dominance of NCM batteries, particularly high-nickel NCM batteries, due to their high specific capacity.9–11 Compared with spent LCO and NCM, which contain rich valuable metals (such as nickel (Ni), cobalt (Co), and manganese (Mn)), the metal recovery value of spent LFP is relatively low. Therefore, recycling spent LCO and NCM batteries has high economic value and can reduce the dependence on valuable metal mineral resources.12–14
Currently, the recycling of failed lithium-ion cathode materials mainly relies on pyrometallurgy, hydrometallurgy, and direct regeneration methods (Fig. 1(a–c)).15–19 Among them, the pyrometallurgical recycling process is well-established, with roasting as the core process. The entire process is simple and meets the need for large-scale recovery treatment while effectively reducing costs. However, despite continuous optimization of the roasting process, the pyrometallurgical recycling still faces issues such as high energy consumption and significant emission of polluting gases. The hydrometallurgy process usually treats materials at lower temperatures, with the core focus being the leaching of ions from cathode materials and the separation of elements. It has the advantage of high recovery rates (generally above 90%) and moderate costs, but the process is prone to generating large amounts of toxic gases, landfill waste, and acidified wastewater, which require waste management to maintain economic feasibility. In contrast, the direct regeneration method can restore the electrochemical properties of the cathode material without damaging its structure, and theoretically, the recovery rate can reach 100%. It has significant advantages in terms of energy efficiency, resource utilization, and environmental impact. However, the black mass that is directly available is mostly a mixture of various cathode materials, which requires screening and classification to meet the requirements for direct regeneration. Additionally, the effectiveness of direct regeneration is linked to the failure behavior and extent of degradation of materials. In particular, high-nickel NCM cathode materials pose significant challenges for regeneration due to the complex phase transformations that occur after cycling. Moreover, due to cost and scale limitations, the direct regeneration is still in the experimental phase.
 | ||
| Fig. 1 Recovery mode summary diagram of the three recycling methods: (a) pyrometallurgical process18 (Copyright 2021, American Chemical Society). (b) Hydrometallurgical process17 (Copyright 2023, Royal Society of Chemistry). (c) Direct recycling19 (Copyright 2020, Elsevier). (d)–(f) Radar diagram displaying the advantages and disadvantages of the three methods15 (Copyright 2024, Elsevier). | ||
In the past decade, a large number of studies have emerged on non-closed-loop high-value recycling and closed-loop regeneration of spent lithium-ion cathode materials.20,21 However, due to the fragmentation and isolation of these studies, there is a lack of systematic research and evaluation on traditional closed-loop recycling and non-closed-loop high-value recovery of valuable metals from spent lithium-ion cathode materials. Therefore, this review systematically focuses on the recycling technologies of high-value layered oxide cathode materials (especially LCO and NCM materials). First, it summarizes the key technical indicators for recycling, then compares the core processes of pyrometallurgy, hydrometallurgy, and direct regeneration, and finally, in response to the current challenges faced by layered oxide cathodes, it proposes corresponding solutions and prospects future development trends, aiming to provide a reference for the efficient recycling and reuse of spent LIBs (Fig. 2).
2. Key considerations
Considering the various recycling methods of LIBs with their unique advantages and limitations, it is necessary to choose the most appropriate recycling method to ensure the efficiency, economy and environmental friendliness of the recycling process.22–26 Here are some of the main indicators to measure the effectiveness of recycling:(1) Recovery yield: during the recovery process, it is necessary to ensure efficient extraction of valuable metals such as lithium (Li), cobalt (Co), nickel (Ni) and manganese (Mn). It can be expressed as
(2) Purity: the purity evaluates the quality of the recovered material, reflecting its suitability for reuse, particularly in battery manufacturing. It can be quantified as
(3) Environmental impact: the recycling process should adopt low energy consumption and low emission processes to reduce the generation and release of waste water (gas) and other harmful substances into air, water and soil.
(4) Economic viability: the recycling process should be economically competitive, with lower cost than that of new mining and raw material synthesis. The optimization of equipment investment, energy consumption and labor costs need to be considered simultaneously.
(5) Process safety: the recycling process should have good operational safety to avoid dangerous chemical reactions or accidents such as fire and explosion during the recycling process.
(6) Scalability: the recycling process should be scalable and capable of processing large quantities of spent LIBs to meet the growing demand for battery recycling.
(7) Life cycle assessment (LCA): the recycling process should have a comprehensive LCA to measure environmental impact from raw material acquisition to waste disposal, which helps to identify the most sustainable recycling methods.
(8) Regulatory and standards compliance: the recycling process must comply with relevant laws and regulations and industry standards, including provisions on waste treatments, material reuse and environmental protection.
(9) Energy efficiency: the energy use efficiency of the recycling process should be as high as possible to reduce the overall energy consumption. Ideally, the energy needed for recycling should come from renewable sources as much as possible.
After comprehensively considering the above parameters, a parameter table from research papers has been derived for the three main recycling methods of failed layered oxide cathode materials (Table 1).
| Recycling method | Recovery elements | Recovery yield (%) | Environmental impact | Economic viability | Scalability |
|---|---|---|---|---|---|
| Pyro-27 | Li | 93.2 | High energy use | Economical | High |
| Pyro-28 | Li | 94.06 | High energy use | Economical | High |
| Pyro-29 | Li/Co | 92/100 | High energy use | Economical | High |
| Pyro-30 | Li/Co | 93/99 | High energy use | Economical | High |
| Pyro-31 | Li | 97 | High energy use | Economical | High |
| Hydr-32 | Li/Co | 95/95 | Moderate waste | Economically viable | Moderate |
| Hydr-33 | Li/Co/Mn/Ni | 98.2/99.96/99.95/99.96 | Moderate waste | Economically viable | Moderate |
| Hydr-34 | Li/Co/Mn/Ni | 99.07/98.64/99.31/99.31 | Moderate waste | Economically viable | Moderate |
| Hydr-35 | Li/Co/Mn | 100/100/92 | Moderate waste | Economically viable | Moderate |
| Hydr-36 | Li/Co/Ni | 100 | Moderate waste | Economically viable | Moderate |
| Direct37 | Li/Co | >99 | Low emission | High cost | Low |
| Direct38 | Li/Co | >99 | Low emission | High cost | Low |
| Direct39 | Li/Co/Mn/Ni | >99 | Low emission | High cost | Low |
| Direct40 | Li/Co/Mn/Ni | >99 | Low emission | High cost | Low |
| Direct41 | Li/Co/Mn/Ni | >99 | Low emission | High cost | Low |
3. Conventional recycling methods
3.1. Pretreatment
The structure of LIBs is complex, including multiple parts such as the shell, separator, electrolyte, cathode and anode.42 Therefore, before recycling processes, the pretreatment must be performed to achieve the separation of different parts.43–45 Spent LIBs usually have a certain residual voltage; if not properly treated, they are easy to cause spontaneous combustion and explosion, thus threatening the safety. Therefore, the primary task of pretreatment is safe discharge. For economic and safety reasons, the chemical discharge is usually performed by soaking the battery in a conductive salt solution (such as NaCl46,47 or NaSO448). After the discharge, the battery will be disassembled to separate the individual components. Later, the electrode active material needs to be separated from the collector. In industry, it is usually separated from other substances such as aluminum foil by mechanical separation, organic solvent dissolution, alkaline leaching and pyrolysis. In the mechanical separation method, the battery is first crushed by a vertical cutter, and the aluminum (Al), copper (Cu) and plastic shell materials are separated by vibration using a screen with a certain aperture, and the steel shell is separated by magnetic separation. After grinding, screening and magnetic separation, the active material powder is obtained.49 The solvent method adopts the principle of similar phase dissolution, using organic solvents such as N-methylpyrrolidone (NMP) to dissolve the binder, which realizes the separation of the active substance and the collector fluid.50 The alkali solution method mainly uses a strong alkali to selectively dissolve Al foil while valuable metals in the electrode material such as Li, Ni, Co and Mn are basically insoluble, achieving the separation of Al foil and active substances.51 The pyrolysis method capitalizes on the distinct decomposition temperatures of components within the electrode material. By precisely regulating the thermal conditions, the binders are decomposed, thereby enabling the separation of the cathode materials from the current collector.52 The pretreatment process is illustrated in Fig. 3.After pretreatment, the composition of the black powder of different layered oxide cathode materials is shown in Table 2:53
| Material type | Major components | Typical composition (wt%) | Impurities (wt%) |
|---|---|---|---|
| LCO | Li, Co | Li: ∼5–7 | Al, Cu: ∼2–5 |
| Co: ∼40–55 | |||
| NCM | Ni, Co, Mn, Li | Ni: 20–40 | ∼2–5 |
| Co: 10–20 | |||
| Mn: 10–20 | |||
| Li: ∼5–7 | |||
| NCA | Ni, Co, Al, Li | Ni: 40–50 | Al: 2–5 |
| Co: 5–10 | ∼3–6 | ||
| Li: ∼6 |
3.2. Pyrometallurgical recycling
The pyrometallurgical technology utilizes the melting temperature difference of different materials to realize the separation of materials.54–56 Taking LCO batteries as an example, in the pyrometallurgical process, the battery components (such as cathode, anode, binder, separators and electrolyte) are generally converted to cobalt-containing alloys (elemental cobalt), slag and gas during the high temperature process (Fig. 4(a)).43 Roasting as the core of the pyrometallurgical process of failed LIBs can be divided into three categories: direct roasting, atmosphere assisted and additive assisted roasting, and new roasting (Fig. 4(b)).18 At present, the emerging pyrometallurgical process achieves more efficient resource recovery and conversion by optimizing chemical conversion and thermodynamic and kinetic parameters when combined with the hydrometallurgical process. | ||
| Fig. 4 (a) LCO material conversion during pyrometallurgical processes43 (Copyright 2020, Elsevier). (b) Schematic of the development process of pyrometallurgical recycling18 (Copyright 2023, Royal Society of Chemistry). (c) Flowchart of recycling processes proposed by Georgi-Maschler et al.57 (Copyright 2017, Elsevier). | ||
 | ||
| Fig. 5 (a) and (b) Schematic diagram of the oxygen free roasting system (1 – Carbolite STF tube furnace, 2 – heating rod, 3 – alundum boat, 4 – alundum tube, 5 – power line, 6 – controller, 7 – inlet valve, 8 – outlet valve, 9 – barometer, 10 – sylphon bellows, 11 – vacuum pump) and schematic diagram of atmospheric assisted roasting recovery of a LCO battery61 (Copyright 2016, Elsevier). (c) Schematic of efficient and selective recovery of valuable metals from spent NCM63 (Copyright 2021, Elsevier). | ||
The main chemical equations of the carbothermal reaction process are as follows (eqn (1)–(6)):
| 4LiCoO2 → 2Li2O + O2 + 4CoO | (1) |
| C + O2 → CO2 | (2) |
| 2C + O2 → 2CO | (3) |
| Li2O + CO2 → 2Li2CO3 | (4) |
| 2CoO + C → 2Co + CO2 | (5) |
| CoO + CO → Co + CO2 | (6) |
In industrial applications, when carbon thermal reduction roasting is used to treat the cathode material, the cathode material usually needs to be separated from the collector during the pretreatment process. To simplify the process, Wang et al. employed the failed LiCoO2 and Al collector to recycle the metal by heat treatment under argon gas.64 According to the Gibbs free energy calculation results,65 LiCoO2 preferentially reacted with Al to form CoO, and part of the CoO was further reduced to Co metal by Al and the other part formed LiAlO2 with the Li2O generated by the reaction. Finally, Li and Al were leached by a strong alkali solution, and Co was recovered by acid leaching. Ma et al. used a similar method to recover Li, Co, Mn, Ni and other elements from the failed NCM cathode material under an argon atmosphere (Fig. 5c). These examples demonstrate the feasibility of adding reducing agents and using atmosphere assisted roasting in pyrometallurgical recycling.
The chemical reaction equations of thermite reduction are as follows (eqn (7)–(9)):
| 6LiCoO2 + 2Al → 3Li2O + 6CoO + Al2O3 | (7) |
| Li2O + Al2O3 → 2LiAlO2 | (8) |
| 3CoO + 2Al → 3Co + Al2O3 | (9) |
The atmosphere assisted roasting technology improves the recovery efficiency by reducing the reaction temperature and energy consumption, and can selectively recycle lithium at the same time. However, there are still some problems in practical application, such as excessive reagent and waste gas treatment. These challenges require further optimization to achieve a more efficient and environmentally friendly spent LIB cathode recycling process.
The additive assisted roasting technology mainly refers to the addition of inorganic salts or acids (such as chloride, sulfate, etc.) in the roasting process of spent LIBs and complete conversion of cathode materials through adding stoichiometric acids or salts (Fig. 6).66,67 On the one hand, the additive assisted roasting greatly enhances the recovery rate of lithium. Using the difference in the solubility of different metal salts, Li can be selectively recovered from raw materials. On the other hand, the additive assisted roasting technology significantly reduces the discharge of waste liquid. At present, the additive assisted roasting mainly includes sulfation roasting, chlorination roasting and nitrification roasting.68
 | ||
| Fig. 6 Schematic diagram of additive assisted roasting67 Copyright 2019, Royal Society of Chemistry. | ||
For sulfation roasting, the cathode material is converted to sulfate. Sulfation agents mainly include sulfuric acid, sulfate and sulfur-containing gases. Lin et al. added sulfuric acid during the roasting of LiCoO2, which partially destroyed the structure of the cathode material by producing some sulfates such as CoSO4 and released the Li element to form Li2SO4, and then lithium sulfate was separated and recovered by a simple water leaching process.67 Tang et al. mixed ammonium sulfate ((NH4)2SO4) with failed LiNi0.6Co0.2Mn0.2O2 (NCM622) and sintered the mixture at 350 °C, converting the cathode material into water-soluble sulfate. Its sulfation rate exceeded 98.5%.69
The chemical reaction equations of the sulphation roasting process are as follows (eqn (10)–(13)):
| 4LiCoO2 + 6H2SO4 + 22H2O → 2Li2SO4 + 4(CoSO4·7H2O) + O2 | (10) |
| 2CoSO4 + 4LiCoO2 → 2Li2SO4 + 6CoO + O2 | (11) |
| 2CoO + O2 → 2Co3O4 | (12) |
| 12LiCoO2 + 6H2SO4 → 6Li2SO4+ 4Co3O4 + 6H2O + O2 | (13) |
Chlorination roasting is a common technique used for element extraction in the metallurgical industry. Fan et al. adopted ammonium chloride (NH4Cl) as the chlorine source. LiCoO2 was chlorinated and calcinated at 350 °C for 20 min. Then LiCl and CoCl2 solutions were obtained by water immersion. The final leaching efficiency of Li and Co was close to 100%.68 Specifically, since NH4Cl breaks down NH3 and HCl during heating, HCl can destroy the surface structure of LiCoO2, and NH3 is used as a reducing agent to reduce cobalt, speeding up the chlorination roasting process.70
The chemical reaction equations of the chlorination roasting process are as follows (eqn (14) and (15)):
| 6LiCoO2 + 18NH4Cl → 6CoCl2 + 6LiCl + 12H2O + N2 + 16NH3 | (14) |
| 6LiCoO2 + 2NH3 → 6CoO + 3Li2O + 3H2O + N2 | (15) |
Compared with sulfation and chlorination roasting, nitrification roasting can further reduce the roasting temperature. Peng et al. nitrated the failed cathode to convert the metal component into the corresponding nitrate. After roasting at 250 °C for 1 h, the recovery rate of Li can reach more than 90%, and the remaining valuable metals were converted into insoluble oxides.71 In general, the nitrification roasting process is accompanied by the production of nitrogen oxides, which can be converted into nitric acid by pressurized acid adsorption, oxidation or catalyst recovery.72
Compared with the direct roasting method, incorporating auxiliary additives can effectively recover lithium from the cathode materials, which helps alleviate the shortage of lithium resources to a certain extent. In addition, compared with the atmosphere assisted roasting process, the heat treatment temperature can be reduced and the energy can be saved. From the perspective of reaction kinetics, the reaction rate of additive assisted roasting at high temperature is higher than that of hydrometallurgy, conducive to efficient treatment of failed cathode materials. However, polluting gases (such as sulfur oxides, nitrogen oxides, chlorine) may be produced during vulcanization, chlorination, and nitrification, which will cause environmental pollution and increasing cost issues.
 | ||
| Fig. 7 (a)–(c) Schematic of the FJH device, real-time temperature measurement with FJH activation and acid leaching results with and without FJH activation (Copyright 2023, American Association for the Advancement of Science). (d)–(f) Schematic representation of the FJH process combining magnetic separation and relithiation techniques, schematic diagram of FJH material change and the radar plot related to comparison among different recycling strategies74 (Copyright 2024, Springer Nature). | ||
Although the emergence of new roasting has obvious advantages (Fig. 7(f)), its essence is still to use the decomposition of substances and extraction of elements at high temperature, which is inevitably associated with high energy consumption and high emission. In addition, the ultra-high temperature of the process poses a safety challenge at scale. At the same time, like other roasting processes, the new roasting process still faces the problem of extraction and separation of elements, and the combination of the hydrometallurgical process makes the whole recovery process still complicated.
3.3. Hydrometallurgical recycling
Hydrometallurgical recycling is considered as one of the methods with high recovery quality and is widely used in the recovery of all kinds of failed LIBs.75,76 Compared with the pure pyrometallurgical process, the hydrometallurgical process is more mature in the recovery of spent LIBs. At the same time, after the pyrometallurgical roasting process, it is usually combined with the hydrometallurgical process for efficient recovery. The advantages of hydrometallurgical recovery are low treatment temperature, high recovery efficiency, high selectivity, low energy consumption and low gas emission.77,78 However, the significant problem is that the use of various chemical reagents will bring about the complexity of waste liquid treatments, which poses a challenge to the sustainable development strategy. Researchers are trying to reconcile the contradictions by optimizing the parameters and process. At present, the process of hydrometallurgical recovery mainly includes pretreatment, leaching of active substances, separation of valuable metals, and material regeneration (Fig. 8).49 Depending on the final recovered product, the hydrometallurgical recovery is mainly divided into two categories: valuable metal leaching and closed-loop regenerative battery cathode materials. The hydrometallurgical recovery has wide application prospects and significant advantages in the field of failure LIB recovery.79 | ||
| Fig. 9 (a) The classification diagram of leaching processes55 (Copyright 2024, Royal Society of Chemistry). (b) Schematic illustration of alkali leaching81 (Copyright 2022, Elsevier). (c) Flow chart of lithium battery leaching by CEC leaching82 (Copyright 2024, Springer Nature). (d) Schematic illustration of the leaching of LCO by bioleaching technology83 (Copyright 2023, Elsevier). (e) Proof-of-concept for the DES-based recycling loop from LNCO cathode materials84 (Copyright 2022, Wiley). | ||
In early studies, inorganic acids (such as sulfuric acid (H2SO4), hydrochloric acid (HCl), nitric acid (HNO3), and phosphoric acid (H3PO4)85) were used to leech metal ions. Because high-priced cobalt and manganese cannot exist as ions in aqueous solutions, it is necessary to add reducing agents (such as H2O2, Na2SO3, NaHSO3, and Na2S2O5) to nonreducing acids such as sulfuric acid, nitric acid, and phosphoric acid. The reducing agents promote the reduction of high-valence metal oxides in the cathode materials, lower their lattice energy, and accelerate the dissolution of metal oxides, thereby increasing the leaching rate. After adding a suitable amount of hydrogen peroxide, the leaching rate of Co and Mn can increase from about 30% to more than 97%. As for the reductive hydrochloric acid, the chlorine gas (Cl2) generated after the oxidation of Cl− is corrosive, which imposes certain requirements on the leaching equipment. Additionally, the treatment of exhaust gases increases the recovery cost. The chemical reaction equations of inorganic acid leaching process are as follows:
| 4LiCoO2 + 6H2SO4 → 4CoSO4 + O2 + 2Li2SO4 + 6H2O | (16) |
| 2LiCoO2 + 3H2SO4 + H2O2 → 2CoSO4 + O2 + Li2SO4 + 4H2O | (17) |
| 4LiCoO2 + 12HNO3 → 4Co(NO3)2 + O2 + 4LiNO3 + 6H2O | (18) |
| 2LiCoO2 + 8HCl → 2CoCl2 + Cl2 + 2LiCl + 4H2O | (19) |
In recent years, organic acids have attracted the attention of researchers because of their wide sources and easy degradation. The common organic acids in leaching include oxalic acid (H2C2O4), malic acid (C4H5O6) and citric acid (C6H8O7). Li et al. separately used DL-malic acid and citric acid to leach the failed LiCoO2; with the addition of an appropriate amount of a reducing agent, the leaching rates of cobalt and lithium both exceeded 90%.86 Oxalic acid and ascorbic acid (C6H8O6) are inherently reductive, which enhances the metal leaching efficiency without the need to add reducing agents. Zeng et al. took oxalic acid as a leaching agent for LiCoO2 leaching, and the leaching rate of Co and Li was as high as 97%.87 Citrus fruit juices are rich in ascorbic acid, which can be used as a metal element in LIB cathode materials, beneficial for further reduction of costs. Pant et al. used citrus juice as a leaching agent for leaching at 90 °C for 30 minutes; the leaching rates of Co, Mn, Ni and Li reached 94%, 99%, 98% and 100%, respectively.88 Li et al. proposed the concept of contact electrocatalysis (CEC) leaching using free radicals generated by contact electroionization, which can replace traditional reducing agents in the leaching process of LCO and NCM materials with citric acid. At 90 °C, the leaching rate of Li and Co was 100% and 92.19%, respectively, for LCO. At 70 °C, the leaching rate of Li, Ni, Mn and Co was 94.56%, 96.62%, 96.54% and 98.39%, respectively (Fig. 9(c)).82
The chemical reaction equations of the organic acid leaching process are as follows (eqn (20) and (21)):
| 2LiCoO2 + 4H2C2O4 → 2CoC2O4 + 2CO2 + Li2C2O4 + 4H2O | (20) |
| 6H3Cit + 2LiCoO2 + H2O2 → 2CoH2Cit + 2LiH2Cit + 4H2O + O2 | (21) |
In strong alkali environments, the complex reaction can occur between ammonia and metal ions. In practical applications, by adjusting the composition of the leaching agent, the ammonia leaching method can efficiently extract Ni and Co elements apart from Mn and Al. At the same time, it avoids the cost increase caused by alkali neutralization after acid leaching. Wang et al. reported the (NH4)2SO4–NH3·H2O–Na2SO3 leaching system to perform ammonia leaching treatments on the failed NCM622 cathode material. In the system, (NH4)2SO4 acted as a buffer and Na2SO3 reduced high-valence ions, accelerating the leaching process. Under appropriate conditions, the final leaching efficiency of Li, Ni and Co reached about 95%, and more than 90% of Mn was eventually left in the slag in the form of precipitation of (NH4)2Mn(SO3)·H2O (Fig. 9(b)).81 The ammonia leaching process can also be used in the combined pyro-hydro recovery process. For example, Yu et al. adopted a NH3·H2O–(NH4)2CO3 system to leach NCM materials after lignite roasting, and the results showed that the leaching rate of Co and Ni was nearly 100%, and Mn was hardly leached without reacting with ammonia.89
In recent years, the bioleaching process has been concerned in the recycling of decommissioned LIBs due to low cost, safety, environmental protection and mild process. By utilizing the cellular metabolism of certain autotrophic bacteria (such as Acidithiobacillus ferrooxidans and acidophilic bacteria), the acidic byproducts produced can effectively leach valuable metal ions from spent cathodes. Yun et al. isolated N10 and P7 strains from abandoned mines. After cultivation, these strains were used to leach spent LiNi0.33Co0.33Mn0.33O2 (NCM111), and the leaching rate of each metal was up to 95%.90 Liao et al. used gallic acid (GA) for mixed microbial cultivation to leach spent LiCoO2 (Fig. 9(d)). The corrosion of the biological acid led to the rapid release of Co and Li, and their leaching rate reached more than 98% in the best process.83 However, the survival of the strain and the metal leaching rate pose a challenge to the large-scale application of this method in industry.91,92
Because of their excellent selectivity and leaching efficiency, deep eutectic solvents (DESs) have attracted attention in the leaching of layered cathode materials.93–95 DESs are a binary or multicomponent system consisting of a hydrogen bond donor (HBD) and a hydrogen bond acceptor (HBA). Compared to the pure components, the system exhibits a lower melting point due to its asymmetric ions and low lattice energy. The common form of DESs is Cat+X−zY, where Cat+ refers to organic cations containing ammonium, phosphorus, and sulfur, and X and Y are Lewis bases and Lewis acids or Brønsted acids, respectively. Most DESs are reductive, meaning they can not only leach metals but also reduce high-valence metal ions to accelerate the leaching process.84 Salma et al. used a choline chloride glycol (ChCl-EG) system to leach a variety of cathode materials such as NMC111/NCA/NMC811. After the ChCl-EG homogeneous solution was prepared at 50 °C, the cathode material was added into a sealed microwave reactor vial and heated at 220 °C for a specified time under agitation. The microwave-assisted lithium selectivity can be induced and a significant extraction efficiency of 50% was achieved within 30 s, greatly reducing time and energy consumption.96 Gupta et al. leached the failed NCM material using the ethylene glycol tartaric acid (EG-TA) system. Under the optimum conditions of temperature (118 °C), reaction time (17 min), S/L (42 g L−1) and solid–liquid ratio (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), the metal extraction rate of Li, Ni, Co and Mn was 99.2%, 96.1%, 95.2% and 97.8%, respectively.97 Although most deep eutectic solvents can be recovered and reused, their high cost limits their large-scale applications.
1), the metal extraction rate of Li, Ni, Co and Mn was 99.2%, 96.1%, 95.2% and 97.8%, respectively.97 Although most deep eutectic solvents can be recovered and reused, their high cost limits their large-scale applications.
3.3.2.1. Solvent extraction method. The solvent extraction method is broadly applied in the separation and recovery of Ni, Co, Co and other metal ions in failed LIB leaching solution based on the solubility difference of compounds in solvents. The solvent extraction method has the advantages of low energy consumption, good separation effect and high product purity. Common extractants are di(6-ethylheptyl) phosphate (P204), 2-ethylhexyl dihydrogen phosphate (P507) and diisooctylphosphinic acid (Cyanex 272).98,99 In order to overcome the problem of the poor separation effect caused by similar properties of chemical metal ions, the chemical precipitation method is often combined with the solvent extraction method. During the recovery of ternary cathode materials, Chen et al. adjusted the pH value of the leaching solution using dimethylglyoxime (C4H8N2O2) and ammonium oxalate ((NH4)2C2O4) to precipitate Ni and Co successively, then Mn was extracted with D2EHPA, and finally lithium phosphate was precipitated to recover Li (Fig. 10(a)). Through this combined method, more than 98% of Ni, Co and Mn were recovered – Ni in the form of C4H8N2O2-Ni, Co in the form of CoC2O4·2H2O, and Mn, which is difficult to recover by conventional methods, in the form of MnSO4 – and the recovery rate of lithium reached 89%.100 To further improve the extraction efficiency and ion selectivity of the leach solution, some scholars have proposed collaborative extraction systems and multistage countercurrent extraction methods.99,101 Efficient and economical metal separation and recovery can be achieved through reasonable process design and extraction agent selection. However, due to complex operation and high cost, it still needs to be further optimized in large-scale industrial applications.
 | ||
| Fig. 10 (a) General flow sheet of the solvent extraction method100 (Copyright 2015, Elsevier). (b) Flow sheet of the chemical precipitation method. (c) The flow sheet of the ion-exchange method separating elements102 (Copyright 2025, Elsevier). (d) The effect of ion exchange absorption rate103 (Copyright 2019, Elsevier). (e) and (f) Diagram of the electrodeposition device and XRD pattern of the product104 (Copyright American Chemical Society). | ||
3.3.2.2. Chemical precipitation method. The chemical precipitation method selectively precipitates one or several metal elements by adding an appropriate amount of the anionic solution to the mixed solution and controlling the reaction conditions, so as to achieve the separation of metal elements (Fig. 10(b)). The chemical precipitation method is a mature, facile and common method for separating and recovering Li in ternary cathode materials. In the element separation and recovery of the failed battery leaching solution, the key is to select the appropriate precipitating agent and improve the selectivity of metal elements and the precipitation rate and product purity. Wang et al. employed the chemical precipitation method to separate NCM111 cathode material leaching solution. They separated Mn, Ni, Co and Li by the potassium permanganate oxidation precipitation, diketoxime precipitation, hydroxide precipitation and carbonate precipitation methods, respectively. The solubility of lithium carbonate decreases with the increase of temperature, and the recovery rate of lithium can reach 95%. After step-by-step precipitation, the purity of Mn, Ni, Co and Li products exceeded 95% (Fig. 10(c)).103 Binna et al. used oxalic acid as a precipitant to convert Co from the LiCoO2 leaching solution to cobalt oxalate. The resulting solution was pre-concentrated by evaporation until a concentration of 10 g L−1 of lithium was obtained. Next, carbon dioxide was blown in to precipitate Li in the form of carbonate. About 90% of Co recovered from the leaching solution was cobalt oxalate and 92% of Li was lithium carbonate.105 However, although chemical precipitation has the advantage of high recovery rates, the purity of the recovered products is not high and further processing and purification is required.
3.3.2.3. Ion exchange method. The ion exchange method can separate the metal ions from the leaching solution by using ion exchange resin with different adsorption coefficients. As early as the 1950s, it has been used for ion separation of valuable metals. This method has the advantages of simple process, low green energy and high efficiency separation when recovering valuable gold from leaching solution. In general, exchange resins are insoluble solids and can be divided into cation exchange resins and anion-exchange resins. Common resins used for ion exchange separation are weakly acidic and strongly acidic cation exchange resins. In the early days, Eom et al. resorted to a strongly acidic cationic resin to adsorb and recover Ni from wastewater, and the recycling rate of Ni could reach 99%.106 In recent years, Gong et al. adopted the hydrogen type strongly acidic exchange resin (HR) to adsorb valuable metal ions in the leaching solution of NCM materials. The exchange between metal ions and hydrogen ions on the HR skeleton eliminated metal separation with similar chemical properties and reduced manufacturing costs.107 Qing et al. proposed a green and efficient method based on co-precipitation and resin adsorption, and the precipitation efficiency of Al, Fe and Cu impurities through the co-precipitation process reached 95.5%, 92.4% and 88.2%, respectively. The loss of Li, Mn, Co and Ni was only 3.5%, 2.5%, 2.1% and 2.2%, respectively (Fig. 10(d)). Subsequently, the selective separation of Ni, Co, Mn and Li was achieved by adsorption of the D113 resin, and the ternary precursor and Li2CO3 were synthesized from the obtained product.102 However, the adsorption rate of ion-exchange resins limits their application in the process.
3.3.2.4. Electrodeposition method. By controlling the potential and current density, the electrodeposition method can recover valuable metals by inducing electrochemical reactions of metal ions in aqueous solutions, resulting in the deposition of metals or metal compounds. Electrodeposition offers advantages such as high selectivity, high product purity, lower costs, and the avoidance of secondary pollution. Lupi and his colleagues introduced Cyanex 272 to extract and separate Co, then to recover Ni by electrodeposition under the conditions of 250 A m−2 current density, 50 °C, and pH 3.0–3.2, achieving 87% electrolysis efficiency and reducing the Ni content in the treated solution to less than 100 mg L−1.108 Meng et al. used Li2CO3 and MnCO3 as raw materials to prepare Li- and Mn-rich leachates to investigate the electrochemical separation of Li and Mn (Fig. 10(e)). During electrodeposition, the anolyte and catholyte acted as a Li-rich solution and an amine solution loaded with CO2, with the process consuming only CO2. The separated products included Li2CO3, MnCO3, and MnO2 (Fig. 10(f)). They recovered Li2CO3 with a purity greater than 99.5% and a yield as high as 48.4%.104 However, electrochemical deposition still faces significant challenges in practical applications. On the one hand, electrodeposition has high energy consumption due to the continuous application of an electric field, increasing the energy cost of the process. On the other hand, for metal ions with similar reduction potentials, such as Co and Ni, co-deposition often occurs, reducing the separation efficiency. Therefore, in practical applications, other methods are often used to separate these metal ions before electrodeposition, and parameters such as current density, voltage, solution pH, and electrolysis time should be optimized to improve the recovery efficiency.
The coprecipitation method is one of the common methods for preparing cathode materials (Fig. 11(a)). In the co-precipitation stage, the co-precipitator not only affects the composition and form of the intermediate, but also determines the microstructure and electrochemical properties of the final regenerated material. Commonly used precipitating agents mainly include hydroxide, carbonate, and oxalate (Fig. 11(b)).112 In the study of closed-loop regeneration of cathode materials, Zhou et al. used oxalic acid as the precipitator to co-precipitate failed NCM523 from citric acid leaching to prepare the precursor. After precipitation, an appropriate amount of lithium hydroxide was added and kept at 880 °C for 10 h. After oxalate precipitation, the recovery of Ni, Co and Mn was all above 99% (Fig. 11(c)). Moreover, the regenerated cathode showed good electrochemical performance, and the capacity retention rate was 71.12% (the initial capacity was 156.9 mA h g−1) after 300 cycles at 0.5C (Fig. 11(d)). Compared with the pyrometallurgical and hydrometallurgical methods, this co-precipitation method possesses advantages in terms of cost, energy consumption and ease of operation (Fig. 11(e)).113 Tang et al. successfully regenerated the NCM622 cathode material with excellent electrochemical properties using a mixed solution of NaOH and NH3·H2O.69 He et al. used sodium carbonate as a co-precipitator and successfully regenerated NCM111 cathode materials at a pH value of 7.5. All these materials showed excellent electrochemical properties.114
 | ||
| Fig. 11 (a) and (b) Regeneration procedures of NCM via co-precipitation methods and comparison of the precursor of three co-precipitation strategies using hydroxide co-precipitation, carbonate co-precipitation, and oxalate co-precipitation112 (Copyright 2023, Elsevier). (c)–(e) The detailed regeneration procedure for spent LIBs, long cycling performance at 0.5C and comprehensive comparison of different battery recycling technologies113 (Copyright 2024, Elsevier). (f) and (g) Schematic illustration of the leaching of NCM and the coordination of the metal ions with D,L-malic acid and XRD patterns of the resynthesized LNCM115 (Copyright 2016, Royal Society of Chemistry). | ||
After leaching metal ions with organic acid, the sol–gel method utilizes the acidity and chelating properties of the organic acid. By adding a complexing agent to the leachate and adjusting the pH value of solutions, a gel with a unique three-dimensional structure is formed, which is then sintered to obtain regenerated materials. Yao et al. demonstrated the regeneration of the NCM111 material by adjusting the metal ion ratio and pH value of the leaching solution using D,L-malic acid as the leaching agent and chelating agent. After high-temperature treatment and through XRD analysis, it is confirmed that the layered structure of the regenerated material is restored, and the material exhibits superior electrochemical performance (Fig. 11(f) and (g)).115 Similarly, Li et al. used citric acid and lactic acid as the chelating agent to regenerate NCM111 materials by the sol–gel method, and the electrochemical performance of the recycled material reached the commercial level.116 The co-precipitation method and sol–gel method can not only improve the purity and performance of the recycled materials, but also significantly boost the efficiency and economy of the regeneration process. These methods provide effective solutions for the resource utilization of decommissioned LIBs and promote the sustainable development of the LIB industry.
4. Direct regeneration
The direct recycling of cathode materials is a promising method for the conversion of spent LIB resources. This technology has received increasing attention in recent years due to potential economic and environmental benefits.79,117–119 Through the stimulation of the external field, the direct regeneration method can supplement the missing elements of the failed material and carry out structural repair without breaking the main structure of the failed material. The repaired cathode material is expected to restore the initial properties of the material and return to service. The current direct regeneration technology for lithium cobalt oxide and ternary cathode materials mainly includes high-temperature solid-state methods, hydrothermal methods, and eutectic methods. In addition, the ion-thermal method, REDOX method and electrochemical method have also attracted extensive attention due to respective advantages. The continuous advancement of technology provides diversified and efficient solutions for the recycling of failed LIB cathode materials, and promotes the sustainable development of LIB recycling.4.1. Failure mechanism of layered oxide cathode materials
The direct regeneration of layered oxide cathode materials requires a clear understanding of their failure mechanism, and the degree of failure of the material will directly affect the performance and life of the battery120–122 (Fig. 12(a)). The capacity of LIBs directly depends on the number of effective Li ions and the number of active substances. During the charge and discharge cycles, Li ions repeatedly intercalate and de-intercalate between the cathode and anode. However, this process is not entirely reversible, and repeated cycling will lead to structural changes in the cathode material, reducing the number of effective lithium ions.123–125 Specifically, in LCO, as the number of cycles increases, the loss of Li+ triggers migration of the surrounding Co3+, resulting in instability of the crystal structure by generating spinel and rock salt phases such as Co3O4 and LiCo2O4 (Fig. 12(b)). The formation of these phases reduces the amount of active Li and significantly increases the diffusion resistance of Li ions, causing a decline in capacity.126,127 In NCM materials, the loss of Li+ creates additional lithium vacancies. Because the radii of Ni2+ and Li+ are similar (0.69 Å and 0.76 Å, respectively), they can easily diffuse into each other's sites, leading to Li–Ni antisite defects. This impedes the migration of Li+ and reduces the number of active Ni and Li, thereby accelerating the degradation of the material's performance.128,129 Additionally, the layered structure of NCM materials is also prone to transformation into spinel and rock-salt phases during cycling, accompanied by irreversible Li loss, further worsening the battery performance. | ||
| Fig. 12 (a) Aging mechanisms of LIBs120 (Copyright 2019, Elsevier). (b) Schematic diagram of the LCO failure mechanism127 (Copyright 2022, Elsevier). (c) The structure evolution of the NMC cathode under different voltages130 (Copyright 2014, Wiley). (d) Schematic diagram showing microcrack generation of NMC131 (Copyright 2018, American Chemical Society). | ||
In addition, the degradation of LCO and NCM is influenced by parameters such as voltage (Fig. 12(c)).123,130,132 Under high temperature or high voltage conditions, the decomposition of Li salts in the electrolyte generates HF, which promotes the acidic dissolution of metal ions, particularly Ni and Mn.133 On the other hand, the volume expansion and contraction of electrode materials during cycling may lead to the formation of cracks between grains, further exposing more grain surfaces to the electrolyte, promoting the dissolution of transition metals, and exacerbating the degradation of the cathode material. In particular, in high-nickel NCM materials, the Ni dissolution is most significant under high voltage conditions (Fig. 12(d)), which greatly impacts the cycle life of batteries.131,134
4.2. Regeneration methods
In the process of high temperature solid phase regeneration, with the increase of heat treatments, the degraded cathode particles begin to fuse, and then a gas channel will be usually generated inside the material; due to the high concentration of the Li environment outside the material, Li atoms effectively diffuse into the lithium vacancy of the degraded cathode material under high temperature drive, so as to repair the degraded crystal structure and composition, and restore its electrochemical activity.137 Nie et al. mixed the degraded LiCoO2 cathode material with Li2CO3 powder and calcined it in the air at 800–950 °C for 12 h. The structure of the regenerated LiCoO2 cathode material recovered well, and its electrochemical performance returned to the commercial level (Fig. 13(a)).138 Cao et al. mixed the degraded NCM523 cathode material with a certain amount of Li2CO3. After calcination at 850 °C for 12 h, the regenerated NCM523 material showed a discharge capacity of 162 mA h g−1. After 100 cycles, the capacity retention rate was as high as 91.9% (Fig. 13(b)).139 Gao and colleagues proposed a trace calcium ion-assisted molten salt strategy to achieve direct regeneration of LiCoO2 degradation. During the high-temperature solid-state reaction process, trace amounts of Ca2+ are added to adjust the regeneration kinetics of LiCoO2, while some Ca2+ can insert into lithium vacancies, achieving simultaneous calcium doping. The regenerated LiCoO2 exhibits excellent rate capacity and cycling performance, far surpassing those of LiCoO2 regenerated by traditional methods and of commercial origin.140
 | ||
| Fig. 13 (a) Flow sheet of regenerating LiCoO2 materials138 (Copyright 2015, Royal Society of Chemistry). (b) Electrochemical performance of regenerated materials139 (Copyright 2020, Springer Berlin Heidelberg). (c) Schematic of the regeneration mechanism of DR-NCM523 and LCMB-NCM523141 (Copyright 2024, Wiley). (d) Schematic of the repair process of spent NCM622 by pre-oxidation and Li supplement calcination142 (Copyright 2021, American Chemical Society). (e) Illustration of the morphology change during the calcination process with and without the surface rock salt phase143 (Copyright 2024, Wiley). (f) XRD patterns for D-LCO, U-NCM-8, U-NCM-10, U-NCM-12, and C-NCM materials144 (Copyright 2024, Wiley). (g) Schematic illustration of the direct upcycling method145 (Copyright 2021, American Chemical Society). | ||
To further optimize the effect of high temperature solid reaction, Zhuang et al. pretreated the failed NCM523 material with boric acid and successfully constructed the LiCo0.5Mn0.5BO3 (LCMB) channel on the particles (Fig. 13(c)). The channel was uniformly covered and combined with rock salt/spinel on the surface of spent NCM523, which reduced the diffusion potential barrier of Li+ at sintering temperature, greatly optimizing the addition amount of the Li supplementing agent and calcination time. Under the addition of only 25% Li and a sintering time of 2 h, the regenerated material can recover to 160 mA h g−1 specific capacity with 78% capacity retention after 300 cycles.141 Yang et al. introduced a pre-oxidation strategy to efficiently regenerate degraded NCM622 cathode materials. After soaking the cathode material with Na2S2O8 and NaOH solutions, they calcined the material with LiOH·H2O as the Li source at 850 °C in an O2 atmosphere for 5 h. The results showed that β-NiOOH produced by pre-oxidation helped to restore the crystal structure and reduce the Li/Ni mixing. The regenerated NCM622 material exhibited a high capacity of 153.82 mA h g−1 at a rate of 1C, and maintained a capacity retention rate of 94.74% after 100 cycles. The pre-oxidation successfully repaired the material structure and shortened the annealing process (Fig. 13(d)).142
The capacity of the material can be restored to some extent by adding the Li source and solid phase sintering, but the good secondary spherical morphology of the polycrystalline particles cannot be restored. Monocrystalline and high nickelization regeneration is an upgraded recycling route proposed in recent years, aiming to achieve the morphology remodeling and performance transcendence of failed cathode materials. Fan et al. calcined the failed polycrystalline NCM523 and Li2CO3 under an O2 atmosphere for 12 h. The rock salt phase on the surface of the layered material was successfully restored, and the monocrystalline upgrading of the material was completed. Studies have unveiled that the restoration of the rock salt phase was conducive to the monocrystalline transformation process of the material (Fig. 13(e)). The regenerated anode material maintained 93.5% capacity retention rate after 150 cycles of 1C. Even at a high cut-off voltage of 4.5 V or a high operating temperature of 45 °C, its performance exceeded that of the commercial cathode.143 Using the high temperature solid phase method, Yang et al. directly transformed the failed LCO into high-performance single crystal NCM111. By mixing the LiCoO2 powder with NiC4H6O4·4H2O, MnC4H6O4·4H2O and LiOH·H2O as Ni, Mn and Li sources, respectively, in a certain proportion, the powder was annealed at 850 °C in the air for 16 hours, followed by washing and a second annealing at 900 °C for 10 hours to complete the regeneration process. XRD results showed that the layered material well maintained after upgrading (Fig. 13(f)). The regenerated anode material had a high specific capacity (159.0 mA h g−1 at 0.1C) and excellent cycle stability (82.1% after 200 cycles at 1C).144 Gao et al. converted invalescent polycrystalline NCM111 to single crystalline LiNi0.8Co0.1Mn0.1O2 (NCM811) by the high temperature solid phase method. After NCM111 was evenly mixed with the Ni precursor, an appropriate amount of LiOH was added, and the particles were first maintained at 480 °C in an O2 atmosphere for 3 h, and then sintered at 850 °C for 12 h. The upgraded NCM811 had better performance than commercial polycrystals while maintaining good cycle stability (Fig. 13(g)).145 This idea provides an efficient and feasible way of upcycling, paving the way for the next generation of LIB cycles and upcycling. Although the high temperature solid phase method has the advantages of simplicity and green, it needs to be maintained at high temperature for a long time, resulting in the increase of energy loss and cost.
As early as 2004, Kim et al. repaired the degraded LiCoO2 cathode material using the hydrothermal method. The failed material was immersed in a stainless-steel autoclave in a tetrafluoroethylene (PTFE) container filled with concentrated LiOH solutions and held at 200 °C for 20 h. After repair, the material presented a capacity of 144.0 mA h g−1 at the first discharge and maintained a capacity retention rate of 92.2% after 40 cycles, proving the feasibility of the hydrothermal method in cathode material recovery.148 It is worth noting that the early hydrothermal repair is not annealed, which causes the faster degradation of materials in the later cycle. Shi et al. reported the direct regeneration of degraded NCM111 and NCM523 in LiOH solutions by the hydrothermal method (Fig. 14(a)). The regenerated cathode material after annealing had ideal stoichiometric ratios and high phase purity, and showed high specific capacity, good cycle stability and high rate performance.149 Subsequent studies have found that the surface LiOH shell formed in the hydrothermal process contributed to the transformation of the rock salt phase in the subsequent calcination step.150
 | ||
| Fig. 14 (a) Schematic illustration of the hydrothermal approach for direct regeneration of spent cathode materials149 (Copyright 2018, American Chemical Society). (b) Saturated vapor pressure of H2O under different temperatures associated with equipment changes during the hydrothermal repair process of NCM111151 (Copyright 2024, Elsevier). (c) Schematic of the failure and topotactic transformation regeneration process of NCM523152 (Copyright 2023, American Chemical Society). (d) and (e) Schematic illustration of the hydrothermal approach for high nickel upgrade of ternary materials and cycling performance of delithiated and regenerated NMC111 and upcycled NMC622 materials at 0.1C including the theoretical NMC622 capacity line153 (Copyright 2024, Elsevier). | ||
There are many factors affecting the regeneration of cathode materials by the hydrothermal method. Xu et al. systematically explored the effects of hydrothermal temperature, time, Li concentration and other parameters on the microstructure, composition and electrochemical properties of recycled cathode materials. The results show that, compared with the temperature factor, the relithiation time is more important to obtain high performance regenerated cathode materials. By optimizing the hydrothermal reaction time, unnecessary extension of the time can be avoided, thereby reducing the energy loss of the hydrothermal relithiation process. In addition, it was also found that the electrochemical properties of hydrothermal regenerated cathode materials were almost the same when partial KOH was replaced by LiOH to reduce Li concentration, indicating the applicability of the hydrothermal method in a wide range of Li concentration.154
To further optimize the effect of the hydrothermal process, Yu et al. conducted reduction pre-treatment of spent NCM111 and NCM622 before the hydrothermal process. After treatment with ethanol, hydrogen peroxide and ethylene glycol, the hydrothermal temperature could be reduced from 220 °C to only 90 °C (Fig. 14(b).). The results show that the reducing agent can effectively improve the relithiation kinetics of failed NCM materials. After hydrothermal annealing, the structure of the material was successfully repaired and the cycle performance was restored to the commercial level.151 Jia et al. added the failed NCM523 cathode powder into a certain concentration of ammonia, and subjected the mixture to hydrothermal treatment for 6 h at 180 °C. After drying, LiOH was mixed evenly and sintered at 500 °C for 2 h, and finally calcined at 850 °C for 10 h under an air atmosphere at a heating rate of 5 °C min−1 to obtain the regenerated NCM523 cathode material. The hydrothermal treatment transformed the topological orientation of the rock salt/spinel phase on the surface of the degraded cathode material to Ni0.5Co0.2Mn0.3(OH)2, which greatly improved the replenishment of Li during the regeneration process (Fig. 14(c)).152
The hydrothermal method can also achieve high-nickel upgrading of spent ternary materials. Krystal and her colleagues treated raw NCM111 with potassium persulfate (K2S2O8) to remove 35% of lithium. They then reacted the material with a high concentration of LiOH aqueous solution at 220 °C for 4 h. Subsequently, they added pre-calculated amounts of NiSO4·6H2O and Li2CO3 and performed heat treatment at 850 °C for 4 h to regenerate the NCM 622 cathode material (Fig. 14(d)). This regenerated material exhibits good cycling stability, with 91.5% capacity retention after 100 cycles (Fig. 14(e)).153 However, the hydrothermal method tends to focus on surface repair of layered materials, making it less effective in regenerating materials with severe structural damage compared to the high-temperature solid-state method, and the huge pressure generated during the hydrothermal reaction has potential safety problems.
Shi et al. chose the eutectic molten salt system of LiNO3–LiOH to repair the failed NCM523 material for the first time. The eutectic temperature of the LiNO3–LiOH molten salt system with optimal mixing ratios was lower than 200 °C (Fig. 15(a)). They heated the cathode material with the mixed molten salt at 300 °C for 4 h, washed the residual Li salt, and then briefly annealed it (Fig. 15(b)). The regenerated material exhibited 149.3 mA h g−1 at 1C and a capacity retention rate of 90% after 100 cycles.157 Jiang and colleagues used the LiOH–Li2CO3 eutectic molten salt system to directly repair the decommissioned NCM523 material, and the recycling performance of the recycled cathode material was also restored to the commercial level.158 Ma et al. selected the LiI–LiOH eutectic salt system with the lowest melting point of binary lithium salt (176 °C) to directly regenerate the waste NCM523 cathode by adding additives of Co2O3 and MnO2. The results showed that the simple one-step heating strategy not only simplified the regeneration process, but also promoted the structural repair further with the addition of additives. The recycled material was even slightly better than the commercial material after 100 cycles at 0.5C, and the capacity retention rate was greater than 80% after 200 cycles.159 From a cost point of view, replacing Li salts with other metal salts with a lower price seems to be a better choice without affecting the effect. Qin et al. introduced the LiOH–Na2SO4 eutectic molten salt system to repair the recycled polycrystalline LiNi0.88Co0.095Al0.025O2. LiOH played a major role in supplementing the loss of Li, and Na2SO4 had a high melting point and acted as a flux for ion migration and particle recrystallization. The regenerated material with the addition of Na2SO4 had better morphology and electrochemical properties, with an initial capacity of 187.2 mA h g−1 and a capacity retention rate of 85.2% after 250 cycles at 1C. The cycling performance was superior to that of commercial materials (Fig. 15(c) and (d)).160
 | ||
| Fig. 15 (a) and (b) Phase diagram of LiNO3 and LiOH and illustration of the relithiation process for Li composition recovery via the eutectic molten salt approach157 (Copyright 2019, Wiley). (c) and (d) SEM for different molten salts and cycling performance of R-NCA-LiOH and R-NCA-LiOH + Na2SO4 regenerated samples160 (Copyright 2023, Wiley). (e) and (f) HRTEM analysis of SNCM83 and UNCM83 (scale bars, 5 nm)161 (Copyright 2024, Wiley). | ||
Compared with the binary molten salt system, the ternary molten salt system shows lower eutectic point, lower density and larger volume, which further promotes the direct regeneration process. Yang et al. used the LiOH–KOH–Li2CO3 ternary eutectic system to regenerate the degraded LiCoO2 cathode material in an air atmosphere. Such a LiOH–KOH–Li2CO3 molten salt system provided a uniform Li-rich environment for the material during repair, which can effectively compensate the deficiency of Li ions and repair the damaged structure. The discharge capacity of the regenerated cathode material recovered to 144.5 mA h g−1, with a capacity retention rate of 92.5% after 200 cycles.162 Qin et al. used a ternary LiOH–LiNO3–CH3COOLi eutectic molten salt system to regenerate the used NCM523 cathode material. After the mixture of molten salts and spent materials was mixed evenly, it was maintained at 400 °C for 5 h. After washing of the residual Li salt, it was annealed at 850 °C for 6 h. The recycled material showed a reversible capacity of 160 mA h g−1 at 0.5C, and the retention rate was 93.7% after 100 cycles. The results demonstrate that the ternary molten salt system is effective in direct regeneration and degradation of cathode materials.163 To further improve the performance of recycled materials, some scholars have modified the materials by reducing the structural defects of the cathode materials through the regeneration of the eutectic molten salt system. Ji et al. converted waste current collector fragments into Al doping agents and used a LiOH·H2O–NaCl–KCl eutectic salt system for the regeneration and doping modification of polycrystalline LiNi0.83Co0.12Mn0.05O2 cathode materials. High-resolution transmission electron microscopy images before and after the regeneration treatment are shown in Fig. 15(e) and (f). The structure of the recycled material recovered well, and the material showed far better electrochemical performance than commercial materials under high pressure and high rate, with a capacity retention rate of 91.1% after 200 cycles in a 1.2 A h soft pack battery.161 The eutectic molten salt system shows great potential in the direct regeneration of degraded cathode materials, but how to meet the requirements of large-scale industrial processing and how to effectively reuse molten salt pose a challenge to the widespread application of this method.
The key parameters of these three methods used most in the direct regeneration of failed layered oxide anode materials are compared and presented in Table 3.
| Indicator | High-temperature solid state method | Hydrothermal method | Eutectic method |
|---|---|---|---|
| Remediation effects | Excellent for severe degradation | Effective for surface-level defects | Competitive; suitable for upgrades |
| Scope of application | Versatile; applicable to various materials | Limited to less degraded materials | Broad; adaptable to multiple materials |
| Cost-effectiveness | High energy and equipment costs | Moderate; energy-efficient | Cost-effective but salt recycling needed |
| Environmental impact | High emission; energy-intensive | Low emission; manageable waste | Minimal emission; salt waste treatment |
 | ||
| Fig. 16 (a) Synthesis scheme of three kinds of ionic liquids for the repair of NCM111164 (Copyright 2020, Wiley). (b) and (c) Schematic drawing of electrochemical configuration and the voltage profiles of the relithiation reaction on the EOL LixCoO2 electrode (WE) and a new LiCoO2 electrode (CE)165 (Copyright 2020, Wiley). (d) Schematic illustration of REDOX repair of LCO37 (Copyright 2024, Elsevier). (e) Proposed reaction scheme for relithiation of the EOL cathode materials via redox mediation166 (Copyright 2021, American Chemical Society). (f) Schematic of the repair process of S-LCO by ultrafast regeneration167 (Copyright 2023, American Chemical Society). (g) Battery cycling flow and comparison of the proposed and reported process168 (Copyright 2024, Elsevier). (h) Schematic of the repair process of magneto-electrochemical synergistic activation regeneration169 (Copyright 2024, Springer Nature). (i) Schematic of in-cell restoration of the NCM cathode using a sacrificial aluminum anode170 (Copyright 2024, Wiley). | ||
Electrochemical regeneration typically uses a two-electrode or three-electrode system, where the applied potential on the spent cathode serves as the main driving force for the relithiation process. Under the influence of the electric field, free Li+ ions in the electrolyte migrate to the lithium vacancies in the structure of the degraded cathode material and insert into the defective lattice of the spent material, replenishing lost Li+. Finally, a short annealing process was used to further remove the crystal water and repair the material structure.171 Yang et al. used this method to regenerate degraded LCO using a three-electrode system (Fig. 16(b)), in which the failed material, Ag/AgCl and non-degraded LCO served as the working electrode, reference electrode, and counter electrode, respectively, and LiSO4 solution served as the electrolyte. The electrochemical relithiation process was carried out at 0.4 mA cm−2 (Fig. 16(c)), and the regenerated cathode material was obtained by sintering in the air for 6 hours at 700 °C. The regenerated material had a capacity of 140 mA h g−1 at 0.1C rate, and the 100-week cycle capacity retention rate was 93%.165
The redox method can transport Li ions and electrons from the Li source to the cathode by the redox medium, and has also been applied in the regeneration of cathode materials. Because the relithiation process can be carried out at room temperature, and most of the media can be recycled, it has an advantage in saving resources and protecting the environment. Zhao et al. used a redox system composed of C2H5LiO and O2 to directly regenerate failed LCO cathode materials (Fig. 16(d)). After mixing the failed material with C2H5LiO solution in an oxygen atmosphere and calcination at 850 °C in the air atmosphere for 8 h, the crystal structure and surface defects of the regenerated LCO cathode material (RLCO) were reconstructed perfectly, and the specific discharge capacity, cycle stability and rate performance were restored to the commercial level. The regeneration process was carried out at room temperature and pressure, and the C2H5LiO solution can be regenerated by adding Li sheets.37 Park et al. used the quinone redox medium 3,5-di-tert-butylcatechol (DTBQ) to recharge delithiated NCM111 (Fig. 16(e)). They mixed DTBQ with dimethyl ether, then added a lithium source and NCM111 powder and stirred for 1 hour for the relithiation process. After filtration and washing, the material was annealed at 850 °C in the air for 4 hours to complete the regeneration. The results indicate that the lithium content of the regenerated material restored to the level before delithiation, and the electrochemical performance was excellent.166
As a non-destructive and ultra-fast method, the Joule heat method can achieve high energy and time efficient cathode material regeneration due to high heating rates, adjustable temperature and high cooling rates, which can greatly reduce the inevitable heat dissipation. Yin et al. regenerated the failed LCO within 8 s (Fig. 16(f)). The extremely short reaction time not only reduced the loss of the Li source at high temperature, but also avoided melting and possible corrosion reactions between the Li source and the container. The results show that the crystal structure of the cathode of LCO can be completely restored after rapid Joule heat repair, and the spinel Co3O4 formed in the long-term cycle process was transformed into the original LCO layer structure. In addition, the electrochemical properties of the regenerated LCO were comparable to those of fresh commercial cathode materials.167
The in situ regeneration method can directly regenerate decommissioned LIBs without disassembly, avoiding the complicated disassembly, separation and remanufacturing process of traditional battery recycling methods. Ogihara et al. proposed to realize in situ regeneration of spent batteries without disassembly by injecting specific recovery reagents into batteries with reduced battery capacity (Fig. 16(g)). They used potential-controlled naphthalene radical anions as a recovery reagent, and precisely controlled their potential through the dielectric effect of the solvent, so that they could selectively provide electrons and carrier Li+ for the cathode, and realized the capacity recovery of NCM111. Furthermore, they successfully demonstrated the effect in a large practical 4 A h class battery, proving the practicability and scalability of the method.168 Gong et al. used magneto-electrochemical co-activation targeting the positive electrode in assembled failed lithium-ion batteries (Fig. 16(h)). By inducing the transition of Ni3+ from high spin to low spin, the super-exchange interaction of Ni–O–transition metal (TM) is reduced. At the same time, the electrochemical reaction drives Li+ from the main material, restoring the inherent Li site, so that the low-mass positive electrode can be converted into a high-quality positive electrode. The repaired lithium-ion soft pack battery (SiC||NCM811, 8 A h nominal capacity) was rejuvenated and increased its capacity from 6.49 A h to 7.14 A h at 1C.169 Song et al. used spontaneous electrochemical corrosion of the Al collector to recover failed NCM cathode materials. The corrosion of Al was promoted by soaking in acetone (MeCN) solution containing lithium bromide (LiBr) (Fig. 16(i)). Meanwhile, lithium ions in the waste LIB electrolyte were utilized to achieve lithium replenishment. This in situ recovery strategy provides new insights into direct positive material recovery techniques that are economically viable and environmentally friendly.170
5. Challenges and countermeasures
Among the three mainstream recycling methods, the recycling technology for failed layered cathode materials has made significant progress at the current stage, but many challenges remain. Based on the current advancements in recycling LCO and NCM cathode materials, the existing challenges and potential countermeasures can be presented as follows:(1) First, improving the recovery rate, recovery efficiency, and product purity of valuable metals in traditional recycling processes is essential. In pyrometallurgical roasting, the incomplete reactions of certain metals, such as Li and Ni, limit the recovery efficiency. This issue arises due to the material characteristics, where lithium, owing to its low boiling point, tends to volatilize at high temperatures, while nickel forms stable oxide phases that are difficult to separate. These factors are compounded by process limitations, where high-temperature conditions may not be optimized for selective metal recovery. As a result, certain metals may remain in the slag or be lost as vapor, reducing overall recovery rates. Similarly, in hydrometallurgical processes, the ion leaching and precipitation rates rarely reach 100%, leaving room for significant improvement. The inefficiencies here are largely due to the kinetics of the leaching reactions and chemical similarities between metals such as Ni, Co, and Mn. Their similar ionic radii and oxidation states complicate their selective leaching, leading to incomplete separation and lower recovery efficiency. Additionally, the high consumption of reagents and the need for complex multi-stage purification processes add to the challenges, increasing both operational costs and time. Moreover, the separation and purification of transition metals like Ni, Co, and Mn, which share similar chemical properties, present substantial challenges. The difficulty of separating these metals is rooted in their similar electronegativity and overlapping chemical behaviors, making selective extraction complex without specialized reagents or multistep processes. The incomplete separation of impurities, such as Al or Fe, may further complicate purification and degrade the quality of the recovered materials. This, in turn, can negatively impact the performance of regenerated materials, particularly when the metals are not sufficiently purified, resulting in lower-quality recovered metals that may not meet the standards required for reuse in new batteries.
The key to addressing these challenges lies in the optimization of process parameters, which can be achieved through a more in-depth understanding of the reaction mechanisms involved. Specifically, studying the thermodynamic and kinetic factors that govern pyrometallurgical and hydrometallurgical processes will enable the optimization of various operational parameters. These include the selection of appropriate reagents, the adjustment of temperature and pressure conditions in roasting, and the optimization of leaching and precipitation steps. For instance, in pyrometallurgical roasting, controlling the gas composition (such as CO2/N2 ratios) can minimize lithium volatilization, while adjusting the temperature and pressure in the hydrometallurgical leaching step can enhance metal dissolution and separation. Moreover, the application of artificial intelligence (AI) and machine learning (ML) technologies presents a significant opportunity to further optimize the separation and purification processes. By incorporating AI and ML, we can model complex interactions between reagents, temperature, and metal phases more accurately. These technologies can help in predicting the best parameters for achieving higher recovery efficiency and product purity. For example, using machine learning algorithms to analyze data from experimental trials can allow the real-time optimization of operational parameters, such as reagent concentrations and reaction times, ensuring that the separation of valuable metals from impurities is more efficient and precise.
(2) Second, the direct regeneration process of the layered oxide anode material is not general enough, and the capacity recovery effect after regeneration is not ideal. On the one hand, the direct regeneration method has specific requirements for the quality of the spent cathode materials. Taking NCM as an example, the types and failure degrees of NCM obtained from different batches are different, which makes it difficult to regenerate the anode material at the same time due to differences in composition and degradation state. Therefore, obtaining pure failed cathode materials is essential for the direct regeneration strategies currently being developed. On the other hand, current direct regeneration technology for layered materials is still in the experimental stage, with regenerated materials often produced in gram or kilogram quantities, and most regeneration processes require stringent conditions, posing challenges for large-scale application. Whether regenerated materials can truly restore their capacity also warrants further investigation. The key to overcoming this challenge lies in developing new methods based on a deeper understanding of mechanisms. Clarifying the relithiation mechanism and developing a universal direct recycling method that accommodates various electrode materials and considers different levels of degradation could be the way forward. To improve regeneration performance, strategies such as upgrading to single-crystal, high-nickel materials, or modifying them through doping and coating could enhance the capacity and stability of regenerated materials, marking a potential future trend.
(3) Finally, the pollution control during the recycling process and the battery life cycle assessment should be strengthened. Waste and pollutants generated during recycling must be strictly controlled, especially when chemical solvents and high-temperature treatments are used. Additionally, a comprehensive assessment of the carbon and environmental footprints throughout the battery's life cycle is needed to ensure that the recycling process is genuinely environmentally friendly. The key to solving this problem is addressing pollution generation and control while reducing carbon emission. Developing and using low-toxicity, recyclable green chemical solvents can reduce environmental pollution during recycling. For example, researchers have developed various deep eutectic solvents that not only efficiently leach cathode materials but also allow for reuse. Comprehensive life cycle assessments can quantify the carbon footprint and environmental impact of different recycling process, and optimization based on the assessment results can maximize overall environmental benefits. Additionally, establishing strict pollution control standards for waste generated during recycling and exploring secondary uses for waste can help reduce the overall environmental burden.
6. Summary and prospect
As a large number of batteries gradually reach the limit of their service life, the recycling of waste lithium-ion batteries has become an urgent task. The layered oxide cathode materials have become the recycling focus as the most promising materials in the current battery recycling field. Currently, researchers have made significant progress through pyrometallurgy, hydrometallurgy, and direct regeneration technologies, but each method still faces numerous technical bottlenecks in terms of material separation, recovery efficiency, and product purity. Thus, we systematically analyze the development status and existing problems of these technologies, and propose corresponding countermeasures, such as optimizing solution treatment and improving the regeneration process, to enhance the recovery rates of valuable metals such as lithium, nickel, cobalt, and manganese, and improve the reutilization performance of cathode materials.In the future, the recycling technologies for LIB layered oxide cathode materials will place greater emphasis on precision and efficiency, with a focus on how to more effectively extract and reuse valuable metals. As direct regeneration technologies advance, new processes will concentrate more on maintaining the microstructure and electrochemical performance of the cathode materials, allowing the recycled materials to restore or approximate their original performance. Meanwhile, the synergistic development of recycling technologies and battery design will further enhance recovery efficiency by designing batteries that are easier to disassemble, separate, and recycle, thus reducing material loss and resource consumption. This integrated approach to design and recycling will drive the entire lithium battery industry chain towards more sustainable and efficient resource utilization, enabling a more environmentally friendly recycling process.
Data availability
Data availability is not applicable to this article as no new data were created or analyzed in this study.Conflicts of interest
The authors declare no conflict of interest.Acknowledgements
The authors acknowledge the support from the National Key Research and Development Program of China (No. 2023YFB3809300).References
- F. Degen, M. Winter, D. Bendig and J. Tuebke, Nat. Energy, 2023, 8, 1284 CAS.
- C.-Y. Wang, T. Liu, X.-G. Yang, S. Ge, N. V. Stanley, E. S. Rountree, Y. Leng and B. D. McCarthy, Nature, 2022, 611, 485 CrossRef CAS PubMed.
- Z. Cui, L. Kang, L. Li, L. Wang and K. Wang, Energy, 2022, 259, 124933 CAS.
- W. Yu, Y. Guo, S. Xu, Y. Yang, Y. Zhao and J. Zhang, Energy Storage Mater., 2023, 54, 172 CrossRef.
- K. Jia, G. Yang, Y. He, Z. Cao, J. Gao, H. Zhao, Z. Piao, J. Wang, A. M. Abdelkader, Z. Liang, R. V. Kumar, G. Zhou, S. Ding and K. Xi, Adv. Mater., 2024, 36, 2313273 CrossRef CAS PubMed.
- T. Yang, D. Luo, A. Yu and Z. Chen, Adv. Mater., 2023, 35, 2203218 CrossRef CAS PubMed.
- G. Harper, R. Sommerville, E. Kendrick, L. Driscoll, P. Slater, R. Stolkin, A. Walton, P. Christensen, O. Heidrich, S. Lambert, A. Abbott, K. S. Ryder, L. Gaines and P. Anderson, Nature, 2019, 575, 75 CAS.
- L. Chang, W. Yang, K. Cai, X. Bi, A. Wei, R. Yang and J. Liu, Mater. Horiz., 2023, 10, 4776 RSC.
- J.-C. Zhang, Z.-D. Liu, C.-H. Zeng, J.-W. Luo, Y.-D. Deng, X.-Y. Cui and Y.-N. Chen, Rare Met., 2022, 41, 3946 CrossRef CAS.
- M. Fan, Q. Meng, X. Chang, C.-F. Gu, X.-H. Meng, Y.-X. Yin, H. Li, L.-J. Wan and Y.-G. Guo, Adv. Energy Mater., 2022, 12, 2103630 CrossRef CAS.
- H.-H. Ryu, B. Namkoong, J.-H. Kim, I. Belharouak, C. S. Yoon and Y.-K. Sun, ACS Energy Lett., 2021, 6, 2726 CrossRef CAS.
- Z. Qin, Y. Zhang, W. Luo, T. Zhang, T. Wang, L. Ni, H. Wang, N. Zhang, X. Liu, J. Zhou and G. Chen, Angew. Chem., Int. Ed., 2023, 62, e202218672 CrossRef CAS PubMed.
- S. Zhou, Y. Zhang, Q. Meng, P. Dong, Z. Fei and Q. Li, J. Environ. Manage., 2021, 277, 111426 CAS.
- J. Quan, S. Zhao, D. Song, T. Wang, W. He and G. Li, Sci. Total Environ., 2022, 819, 153105 CrossRef CAS PubMed.
- P. Li, S. Luo, L. Zhang, Q. Liu, Y. Wang, Y. Lin, C. Xu, J. Guo, P. Cheali and X. Xia, J. Energy Chem., 2024, 89, 144 CrossRef CAS.
- K. Du, E. H. Ang, X. Wu and Y. Liu, Energy Environ. Mater., 2022, 5, 1012 CrossRef CAS.
- K. Davis and G. P. Demopoulos, RSC Sustainability, 2023, 1, 1932 RSC.
- M. Zhou, B. Li, J. Li and Z. Xu, ACS EST Engg., 2021, 1, 1369 CrossRef CAS.
- Y. Bai, N. Muralidharan, Y.-K. Sun, S. Passerini, M. S. Whittingham and I. Belharouak, Mater. Today, 2020, 41, 304 CrossRef CAS.
- J. Zhang, D. Chen, J. Jiao, W. Zeng and S. Mu, Green Energy Environ., 2024, 807 Search PubMed.
- R. Ma, S. Tao, X. Sun, Y. Ren, C. Sun, G. Ji, J. Xu, X. Wang, X. Zhang, Q. Wu and G. Zhou, Nat. Commun., 2024, 15, 7641 CrossRef CAS PubMed.
- F. Gu, J. Guo, X. Yao, P. A. Summers, S. D. Widijatmoko and P. Hall, J. Cleaner Prod., 2017, 161, 765 CrossRef.
- B. Huang, Z. Pan, X. Su and L. An, J. Power Sources, 2018, 399, 274 CrossRef CAS.
- Y. Yao, M. Zhu, Z. Zhao, B. Tong, Y. Fan and Z. Hua, ACS Sustainable Chem. Eng., 2018, 6, 13611 CAS.
- Y. Zhao, X. Yuan, L. Jiang, J. Wen, H. Wang, R. Guan, J. Zhang and G. Zeng, Chem. Eng. J., 2020, 383, 123089 CAS.
- T. Kim, S. Park, J. Bae, D. Jung, H. Cheon, W.-G. Lee and Y. Choi, Mater. Horiz., 2024, 11, 1008 RSC.
- W. Hou, X. Huang, R. Tang, Y. Min, Q. Xu, Z. Hu and P. Shi, Waste Manage., 2023, 155, 129 CrossRef CAS PubMed.
- Z. Yang, Y. Zhang, H. Yu, L. Liu, Y. Li, T. Hao, Q. Meng and P. Dong, J. Environ. Chem. Eng., 2023, 11, 110060 CrossRef CAS.
- P. Xu, C. Liu, X. Zhang, X. Zheng, W. Lv, F. Rao, P. Yao, J. Wang and Z. Sun, ACS Sustainable Chem. Eng., 2021, 9, 2271 CrossRef CAS.
- Y. Tang, H. Xie, B. Zhang, X. Chen, Z. Zhao, J. Qu, P. Xing and H. Yin, Waste Manage., 2019, 97, 140 CrossRef CAS PubMed.
- J. Zhang, J. Hu, W. Zhang, Y. Chen and C. Wang, J. Cleaner Prod., 2018, 204, 437 CrossRef CAS.
- E. G. Pinna, M. C. Ruiz, M. W. Ojeda and M. H. Rodriguez, Hydrometallurgy, 2017, 167, 66 CrossRef CAS.
- L. Li, J. Lu, Y. Ren, X. X. Zhang, R. J. Chen, F. Wu and K. Amine, J. Power Sources, 2012, 218, 21 CrossRef CAS.
- L.-P. He, S.-Y. Sun, Y.-Y. Mu, X.-F. Song and J.-G. Yu, ACS Sustainable Chem. Eng., 2017, 5, 714 CrossRef CAS.
- L. Zhuang, C. Sun, T. Zhou, H. Li and A. Dai, Waste Manage., 2019, 85, 175 CrossRef CAS PubMed.
- S. Virolainen, T. Wesselborg, A. Kaukinen and T. Sainio, Hydrometallurgy, 2021, 202, 105602 CrossRef CAS.
- X. Zhao, Z. Fei, J. Yan, Q. Meng and P. Dong, J. Energy Storage, 2024, 83, 110344 CrossRef.
- L. Kong, Z. Li, W. Zhu, C. R. Ratwani, N. Fernando, S. Karunarathne, A. M. Abdelkader, A. R. Kamali and Z. Shi, J. Colloid Interface Sci., 2023, 640, 1080 CrossRef CAS PubMed.
- S. Liu, J. Yang, S. Hao, S. Jiang, X. Li, O. Dolotko, F. Wu, Y. Li and Z. He, Chem. Eng. J., 2024, 479, 147607 CrossRef CAS.
- C. Xing, Y. Ying, B. Zhang, L. Liu, Y. Liu, Y. Zhang, L. Fei, M. Gan, J. Ye and H. Huang, Energy Storage Mater., 2024, 65, 103182 CrossRef.
- W. Chen, J. Li, H. Ji, R. Shi, J. Wang, Y. Zhu, J. Liu, R. Zhang, Z. Wu, X. Xiao, Z. Wei and G. Zhou, Adv. Mater., 2025, 37, 2416818 CrossRef CAS PubMed.
- Y. Zhang, Y. Lu, J. Jin, M. Wu, H. Yuan, S. Zhang, K. Davey, Z. Guo and Z. Wen, Adv. Mater., 2024, 36, e2308484 CrossRef PubMed.
- X. Zhang, L. Li, E. Fan, Q. Xue, Y. Bian, F. Wu and R. Chen, Chem. Soc. Rev., 2018, 47, 7239 CAS.
- R. Sommerville, P. Zhu, M. A. Rajaeifar, O. Heidrich, V. Goodship and E. Kendrick, Resour., Conserv. Recycl., 2021, 165, 105219 CrossRef CAS.
- X. Zheng, W. Gao, X. Zhang, M. He, X. Lin, H. Cao, Y. Zhang and Z. Sun, Waste Manage., 2017, 60, 680 CrossRef CAS PubMed.
- Y. He, T. Zhang, F. Wang, G. Zhang, W. Zhang and J. Wang, J. Cleaner Prod., 2017, 143, 319 CrossRef CAS.
- M. Lu, H. Zhang, B. Wang, X. Zheng and C. Dai, Int. J. Electrochem. Sci., 2013, 8, 8201 CrossRef CAS.
- H. Nie, L. Xu, D. Song, J. Song, X. Shi, X. Wang, L. Zhang and Z. Yuan, Green Chem., 2015, 17, 1276 RSC.
- Z. Liang, C. Cai, G. Peng, J. Hu, H. Hou, B. Liu, S. Liang, K. Xiao, S. Yuan and J. Yang, ACS Sustainable Chem. Eng., 2021, 9, 5750 CrossRef CAS.
- L. Yaug, G. Xi and Y. Xi, Ceram. Int., 2015, 41, 11498 CrossRef.
- J. Hu, J. Zhang, H. Li, Y. Chen and C. Wang, J. Power Sources, 2017, 351, 192 CrossRef CAS.
- D. Song, X. Wang, E. Zhou, P. Hou, F. Guo and L. Zhang, J. Power Sources, 2013, 232, 348 CrossRef CAS.
- J. Neumann, M. Petranikova, M. Meeus, J. D. Gamarra, R. Younesi, M. Winter and S. Nowak, Adv. Energy Mater., 2022, 12, 2102917 CrossRef CAS.
- B. K. Biswal, B. Zhang, P. Thi Minh Tran, J. Zhang and R. Balasubramanian, Chem. Soc. Rev., 2024, 53, 5552 RSC.
- H. Ji, J. Wang, J. Ma, H. M. Cheng and G. Zhou, Chem. Soc. Rev., 2023, 52, 8194 RSC.
- Y. Jin, T. Zhang and M. Zhang, Adv. Energy Mater., 2022, 12, 2201526 CrossRef CAS.
- T. Georgi-Maschler, B. Friedrich, R. Weyhe, H. Heegn and M. Rutz, J. Power Sources, 2012, 207, 173 CrossRef CAS.
- J. Li, Y. Lai, X. Zhu, Q. Liao, A. Xia, Y. Huang and X. Zhu, J. Hazard. Mater., 2020, 398, 122955 CrossRef CAS PubMed.
- V. Etacheri, R. Marom, R. Elazari, G. Salitra and D. Aurbach, Energy Environ. Sci., 2011, 4, 3243 RSC.
- J. Xiao, J. Li and Z. Xu, Environ. Sci. Technol., 2017, 51, 11960 CrossRef CAS PubMed.
- J. Li, G. Wang and Z. Xu, J. Hazard. Mater., 2016, 302, 97 CrossRef CAS PubMed.
- J. W. Kim and H. G. Lee, Metall. Mater. Trans. B, 2001, 32, 17 CrossRef.
- Y. Ma, J. Tang, R. Wanaldi, X. Zhou, H. Wang, C. Zhou and J. Yang, J. Hazard. Mater., 2021, 402, 123491 CrossRef CAS PubMed.
- W. Q. Wang, Y. C. Zhang, X. G. Liu and S. M. Xu, ACS Sustainable Chem. Eng., 2019, 7, 12222 CAS.
- M. J. Wang and A. Navrotsky, Solid State Ion, 2004, 166, 167 CrossRef CAS.
- D. Wang, X. Zhang, H. Chen and J. Sun, Miner. Eng., 2018, 126, 28 CrossRef CAS.
- J. Lin, C. Liu, H. Cao, R. Chen, Y. Yang, L. Li and Z. Sun, Green Chem., 2019, 21, 5904 RSC.
- E. Fan, L. Li, J. Lin, J. Wu, J. Yang, F. Wu and R. Chen, ACS Sustainable Chem. Eng., 2019, 7, 16144 CrossRef CAS.
- Y. Tang, X. Qu, B. Zhang, Y. Zhao, H. Xie, J. Zhao, Z. Ning, P. Xing and H. Yin, J. Cleaner Prod., 2021, 279, 123633 CrossRef CAS.
- J. Xiao, B. Niu and Z. Xu, J. Cleaner Prod., 2020, 277, 122718 CrossRef CAS.
- C. Peng, F. Liu, Z. Wang, B. P. Wilson and M. Lundstrom, J. Power Sources, 2019, 415, 179 CrossRef CAS.
- Y. Li, Y. Liu, L. Zhang, Q. Su and G. Jin, Chin. J. Chem. Eng., 2010, 18, 244 CAS.
- W. Chen, J. Chen, K. V. Bets, R. V. Salvatierra, K. M. Wyss, G. Gao, C. H. Choi, B. Deng, X. Wang, J. T. Li, C. Kittrell, N. La, L. Eddy, P. Scotland, Y. Cheng, S. Xu, B. Li, M. B. Tomson, Y. Han, B. I. Yakobson and J. M. Tour, Sci. Adv., 2023, 9, eadh5131 CAS.
- W. Chen, Y. Cheng, J. Chen, K. V. Bets, R. V. Salvatierra, C. Ge, J. T. Li, D. X. Luong, C. Kittrell, Z. Wang, E. A. McHugh, G. Gao, B. Deng, Y. Han, B. I. Yakobson and J. M. Tour, Nat. Commun., 2024, 15, 6250 CrossRef CAS PubMed.
- Y. Li, W. Lv, H. Huang, W. Yan, X. Li, P. Ning, H. Cao and Z. Sun, Green Chem., 2021, 23, 6139 RSC.
- J. C.-Y. Jung, N. Chow, A. Nacu, M. Melashvili, A. Cao, L. Khorbaladze, Z. Meseldzija, J. P.-C. Sui and J. Zhang, Int. J. Green Energy, 2021, 18, 1597 CrossRef CAS.
- M. Chen, X. Ma, B. Chen, R. Arsenault, P. Karlson, N. Simon and Y. Wang, Joule, 2019, 3, 2622 CrossRef CAS.
- X. Zheng, Z. Zhu, X. Lin, Y. Zhang, Y. He, H. Cao and Z. Sun, Engineering, 2018, 4, 361 CrossRef CAS.
- T. Yang, D. Luo, A. Yu and Z. Chen, Adv. Mater., 2023, 35, e2203218 CrossRef PubMed.
- R. Qadir and F. Gulshan, Mater. Sci. Appl., 2018, 09, 142 CAS.
- H. Wang, Z. Li, Q. Meng, J. Duan, M. Xu, Y. Lin and Y. Zhang, Hydrometallurgy, 2022, 208, 105809 CrossRef CAS.
- H. Li, A. Berbille, X. Zhao, Z. Wang, W. Tang and Z. L. Wang, Nat. Energy, 2023, 8, 1137 CrossRef CAS.
- X. J. Liao, M. Y. Ye, J. L. Liang, J. X. Jian, S. P. Li, Q. W. Gan, Z. H. Liu, Z. H. Mo, Y. Huang and S. Y. Sun, J. Hazard. Mater., 2023, 447, 130773 CrossRef CAS PubMed.
- R. Morina, D. Callegari, D. Merli, G. Alberti, P. Mustarelli and E. Quartarone, ChemSusChem, 2022, 15, e202102080 CrossRef CAS PubMed.
- X. Chen, H. Ma, C. Luo and T. Zhou, J. Hazard. Mater., 2017, 326, 77 CrossRef CAS PubMed.
- L. Li, J. Ge, F. Wu, R. Chen, S. Chen and B. Wu, J. Hazard. Mater., 2010, 176, 288 CrossRef CAS PubMed.
- X. Zeng, J. Li and B. Shen, J. Hazard. Mater., 2015, 295, 112 CrossRef CAS PubMed.
- D. Pant and T. Dolker, Waste Manage., 2017, 60, 689 CrossRef CAS PubMed.
- J. Yu, B. Ma, Z. Qiu, C. Wang and Y. Chen, ACS Sustainable Chem. Eng., 2023, 11, 9738 CAS.
- S. Yun, H. Jung, H. J. Lee, Y. Yang, J. S. Lee, M. Hur, B.-H. Lee, J. Ahn and G. Hwang, J. Ind. Eng. Chem., 2024, 135, 552 CAS.
- N. B. Horeh, S. M. Mousavi and S. A. Shojaosadati, J. Power Sources, 2016, 320, 257 CrossRef CAS.
- M. Baniasadi, F. Vakilchap, N. Bahaloo-Horeh, S. M. Mousavi and S. Farnaud, J. Ind. Eng. Chem., 2019, 76, 75 CrossRef CAS.
- M. K. Tran, M.-T. F. Rodrigues, K. Kato, G. Babu and P. M. Ajayan, Nat. Energy, 2019, 4, 339 CrossRef CAS.
- P. G. Schiavi, P. Altimari, M. Branchi, R. Zanoni, G. Simonetti, M. A. Navarra and F. Pagnanelli, Chem. Eng. J., 2021, 417, 129249 CrossRef CAS.
- K. Wang, T. Hu, P. Shi, Y. Min, J. Wu and Q. Xu, ACS Sustainable Chem. Eng., 2022, 10, 1149 CrossRef CAS.
- S. H. Alhashim, S. Bhattacharyya and P. M. Ajayan, Adv. Funct. Mater., 2024, 34, 2404570 CrossRef CAS.
- S. Gupta, K. K. Pant and G. Corder, J. Cleaner Prod., 2024, 461, 142624 CrossRef CAS.
- G. Granata, F. Pagnanelli, E. Moscardini, Z. Takacova, T. Havlik and L. Toro, J. Power Sources, 2012, 212, 205 CrossRef CAS.
- J. M. Zhao, X. Y. Shen, F. L. Deng, F. C. Wang, Y. Wu and H. Z. Liu, Sep. Purif. Technol., 2011, 78, 345 CrossRef CAS.
- X. Chen, T. Zhou, J. Kong, H. Fang and Y. Chen, Sep. Purif. Technol., 2015, 141, 76 CrossRef CAS.
- F. Vasilyev, S. Virolainen and T. Sainio, Sep. Purif. Technol., 2019, 210, 530 CrossRef CAS.
- Q. Lei, K. Zhou, X. Zhang, Q. Yang, Z. Qiu, C. Peng, D. He and W. Chen, Sep. Purif. Technol., 2025, 353, 128501 CrossRef CAS.
- R.-C. Wang, Y.-C. Lin and S.-H. Wu, Hydrometallurgy, 2009, 99, 194 CrossRef CAS.
- M. Shi, L. A. Diaz, J. R. Klaehn, A. D. Wilson and T. E. Lister, ACS Sustainable Chem. Eng., 2022, 10, 11773 CrossRef CAS.
- E. G. Pinna, N. Toro, S. Gallegos and M. H. Rodriguez, Materials, 2022, 15, 44 CrossRef CAS PubMed.
- T. H. Eom, C. H. Lee, J. H. Kim and C. H. Lee, Desalination, 2005, 180, 163 CrossRef CAS.
- S. Gong, E. Dong, B. Liu, Y. Chao, Y. Niu, G. Ji, W. Chen, K. Hou, S. Guo and L. Zhang, Sep. Purif. Technol., 2024, 348, 127771 CrossRef CAS.
- C. Lupi and M. Pasquali, Miner. Eng., 2003, 16, 537 CrossRef CAS.
- S. Feng, D. Li, J. Deng, Z. Yang, J. Zhang and Y. Zhou, Sep. Purif. Technol., 2025, 354, 128953 CrossRef CAS.
- Y. Luo, L. Ou and C. Yin, Sci. Total Environ., 2023, 875, 162567 CrossRef CAS PubMed.
- H. Yu, H. Yang, K. Chen, L. Yang, M. Huang, Z. Wang, H. Lv, C. Xu, L. Chen and X. Luo, Energy Storage Mater., 2024, 67, 103288 CrossRef.
- L. Yu, X. Liu, S. Feng, S. Jia, Y. Zhang, J. Zhu, W. Tang, J. Wang and J. Gong, Chem. Eng. J., 2023, 476, 146733 CrossRef CAS.
- M. M. Zhou, J. Shen, Y. Duan, Y. Z. Zuo, Z. W. Xing and R. P. Liu, Energy Storage Mater., 2024, 67, 103250 CrossRef.
- L.-P. He, S.-Y. Sun and J.-G. Yu, Ceram. Int., 2018, 44, 351 CrossRef CAS.
- L. Yao, H. Yao, G. Xi and Y. Feng, RSC Adv., 2016, 6, 17947 RSC.
- L. Li, E. Fan, Y. Guan, X. Zhang, Q. Xue, L. Wei, F. Wu and R. Chen, ACS Sustainable Chem. Eng., 2017, 5, 5224 CrossRef CAS.
- J. Wang, J. Ma, Z. Zhuang, Z. Liang, K. Jia, G. Ji, G. Zhou and H.-M. Cheng, Chem. Rev., 2024, 124(2839), 2839 CrossRef CAS PubMed.
- J. Wu, M. Zheng, T. Liu, Y. Wang, Y. Liu, J. Nai, L. Zhang, S. Zhang and X. Tao, Energy Storage Mater., 2023, 54, 120 CrossRef CAS.
- P. Xu, Q. Dai, H. Gao, H. Liu, M. Zhang, M. Li, Y. Chen, K. An, Y. S. Meng, P. Liu, Y. Li, J. S. Spangenberger, L. Gaines, J. Lu and Z. Chen, Joule, 2020, 4, 2609 CAS.
- C. Pastor-Fernandez, T. F. Yu, W. D. Widanage and J. Marco, Renewable Sustainable Energy Rev., 2019, 109, 138 CrossRef CAS.
- J. Wang, D. Li, W. Zeng, X. Chen, Y. Zhang, S. Zhang, Z. Li, C. Li and S. Mu, J. Energy Chem., 2025, 102, 534 CrossRef CAS.
- J. Feng, Z. Chen, W. Zhou and Z. Hao, Mater. Horiz., 2023, 10, 4686 RSC.
- J. Li, C. Lin, M. Weng, Y. Qiu, P. Chen, K. Yang, W. Huang, Y. Hong, J. Li, M. Zhang, C. Dong, W. Zhao, Z. Xu, X. Wang, K. Xu, J. Sun and F. Pan, Nat. Nanotechnol., 2021, 16, 599 CrossRef CAS PubMed.
- C. Xu, K. Marker, J. Lee, A. Mahadevegowda, P. J. Reeves, S. J. Day, M. F. Groh, S. P. Emge, C. Ducati, B. Layla Mehdi, C. C. Tang and C. P. Grey, Nat. Mater., 2021, 20, 84 CrossRef CAS PubMed.
- X. Zhang, Z. Wang, X. Li, Y. Su, Z. Ye, L. Zhang, Q. Huang, Y. Tang and J. Huang, Mater. Horiz., 2023, 10, 1856 CAS.
- R. Konar, S. Maiti, N. Shpigel and D. Aurbach, Energy Storage Mater., 2023, 63, 103001 CrossRef.
- J. Chen, Z. Lin, W. Xiang, B. Wu, G. Zhang, X. Wen, Y. Che, D. Ruan, W. Li and M. Chen, Electrochim. Acta, 2022, 436, 141374 CrossRef CAS.
- R. Jung, F. Linsenmann, R. Thomas, J. Wandt, S. Solchenbach, F. Maglia, C. Stinner, M. Tromp and H. A. Gasteiger, J. Electrochem. Soc., 2019, 166, A378 Search PubMed.
- D.-S. Ko, J.-H. Park, S. Park, Y. N. Ham, S. J. Ahn, J.-H. Park, H. N. Han, E. Lee, W. S. Jeon and C. Jung, Nano Energy, 2019, 56, 434 CrossRef CAS.
- S.-K. Jung, H. Gwon, J. Hong, K.-Y. Park, D.-H. Seo, H. Kim, J. Hyun, W. Yang and K. Kang, Adv. Energy Mater., 2014, 4, 1300787 CrossRef.
- H.-H. Ryu, K.-J. Park, C. S. Yoon and Y.-K. Sun, Chem. Mater., 2018, 30, 1155 Search PubMed.
- L. Du, X. Hou, D. Berghus, L. Frankenstein, R. Schmuch, J. Wang, E. Paillard, M. Winter, T. Placke and J. Li, J. Mater. Chem. A, 2023, 11, 3663 Search PubMed.
- Y. Ding, Y. Li, R. Xu, X. Han, K. Huang, X. Ke, B. Wang, M. Sui and P. Yan, Nano Energy, 2024, 126, 109685 CrossRef CAS.
- J. Hu, H. Wang, B. Xiao, P. Liu, T. Huang, Y. Li, X. Ren, Q. Zhang, J. Liu, X. Ouyang and X. Sun, Natl. Sci. Rev., 2023, 10, nwad252 CrossRef CAS PubMed.
- Y. Shin, S. Kim, S. Park, J. Lee, J. Bae, D. Kim, H. Joo, S. Ban, H. Lee, Y. Kim and K. Kwon, Renewable Sustainable Energy Rev., 2023, 187, 113693 CrossRef CAS.
- T. Wang, H. M. Luo, Y. C. Bai, J. L. Li, I. Belharouak and S. Dai, Adv. Energy Mater., 2020, 10, 2001204 CrossRef CAS.
- Z. Chi, J. Li, L. Wang, T. Li, Y. Wang, Y. Zhang, S. Tao, M. Zhang, Y. Xiao and Y. Chen, Green Chem., 2021, 23, 9099 RSC.
- H. Nie, L. Xu, D. Song, J. Song, X. Shi, X. Wang, L. Zhang and Z. Yuan, Green Chem., 2015, 17, 1276 RSC.
- L.-P. Yue, P. Lou, G.-H. Xu, H. Xu, G. Jin, L. Li, H. Deng, Q. Cheng, S. Tang and Y.-C. Cao, Ionics, 2020, 26, 2757 CrossRef CAS.
- G. Gao, Y. Zhu, S. Di, J. Zhao, C. Liu, S. Wang and L. Li, Acta Mater., 2024, 273, 119969 CrossRef CAS.
- Z. Zhuang, J. Li, H. Ji, Z. Piao, X. Wu, G. Ji, S. Liu, J. Ma, D. Tang, N. Zheng, J. Wang and G. Zhou, Adv. Mater., 2024, 36, e2313144 CrossRef PubMed.
- X. Yang, Y. Zhang, J. Xiao, Y. Zhang, P. Dong, Q. Meng and M. Zhang, ACS Sustainable Chem. Eng., 2021, 9, 16997 CrossRef CAS.
- M. Fan, X.-H. Meng, H. Guo, S. Xin, X. Chang, K.-C. Jiang, J.-C. Chen, Q. Meng and Y.-G. Guo, Adv. Mater., 2024, 36, 2405238 CrossRef CAS PubMed.
- Z. L. Yang, C. X. Xing, L. Chen, S. Q. Tao, Y. C. Zhang, G. R. Wang, P. Yang, J. Zhou, C. C. Zhang, J. Q. Chen and L. F. Fei, Adv. Funct. Mater., 2025, 35, 2409737 CrossRef CAS.
- H. Gao, Q. Yan, D. Tran, X. Yu, H. Liu, M. Li, W. Li, J. Wu, W. Tang, V. Gupta, J. Luo and Z. Chen, ACS Energy Lett., 2023, 8, 4136 CrossRef CAS.
- S. Sloop, L. Crandon, M. Allen, K. Koetje, L. Reed, L. Gaines, W. Sirisaksoontorn and M. Lerner, Sustainable Mater. Technol., 2020, 25, e00152 CrossRef CAS.
- H. Gao, Q. Yan, P. Xu, H. Liu, M. Li, P. Liu, J. Luo and Z. Chen, ACS Appl. Mater. Interfaces, 2020, 12, 51546 CrossRef CAS PubMed.
- D. S. Kim, J. S. Sohn, C. K. Lee, J. H. Lee, K. S. Han and Y. I. Lee, J. Power Sources, 2004, 132, 145 CrossRef CAS.
- Y. Shi, G. Chen, F. Liu, X. Yue and Z. Chen, ACS Energy Lett., 2018, 3, 1683 CrossRef CAS.
- Y. Guo, X. Liao, P. Huang, P. Lou, Y. Su, X. Hong, Q. Han, R. Yu, Y.-C. Cao and S. Chen, Energy Storage Mater., 2021, 43, 348 CrossRef.
- X. Yu, S. Yu, Z. Yang, H. Gao, P. Xu, G. Cai, S. Rose, C. Brooks, P. Liu and Z. Chen, Energy Storage Mater., 2022, 51, 54 CrossRef.
- K. Jia, J. Wang, Z. Zhuang, Z. Piao, M. Zhang, Z. Liang, G. Ji, J. Ma, H. Ji, W. Yao, G. Zhou and H.-M. Cheng, J. Am. Chem. Soc., 2023, 145, 7288 CrossRef CAS PubMed.
- K. Davis and G. P. Demopoulos, Next Energy, 2024, 4, 263 CrossRef.
- P. Xu, Z. Yang, X. Yu, J. Holoubek, H. Gao, M. Li, G. Cai, I. Bloom, H. Liu, Y. Chen, K. An, K. Z. Pupek, P. Liu and Z. Chen, ACS Sustainable Chem. Eng., 2021, 9, 4543 CrossRef CAS.
- D. Chen and S. Mu, Adv. Mater., 2024, 36, e2408285 CrossRef PubMed.
- M. V. Reddy, G. V. S. Rao and B. V. R. Chowdari, J. Power Sources, 2006, 159, 263 CrossRef CAS.
- Y. Shi, M. Zhang, Y. S. Meng and Z. Chen, Adv. Energy Mater., 2019, 9, 1900454 CrossRef.
- G. Jiang, Y. Zhang, Q. Meng, Y. Zhang, P. Dong, M. Zhang and X. Yang, ACS Sustainable Chem. Eng., 2020, 8, 18138 CrossRef CAS.
- J. Ma, J. Wang, K. Jia, Z. Liang, G. Ji, Z. Zhuang, G. Zhou and H.-M. Cheng, J. Am. Chem. Soc., 2022, 144, 20306 CrossRef CAS PubMed.
- Z. Qin, Y. Zhang, W. Luo, T. Zhang, T. Wang, L. Ni, H. Wang, N. Zhang, X. Liu, J. Zhou and G. Chen, Angew. Chem., Int. Ed., 2023, 62, e202218672 CAS.
- H. Ji, J. Wang, H. Qu, J. Li, W. Ji, X. Qiu, Y. Zhu, H. Ren, R. Shi, G. Ji, W. Zhao and G. Zhou, Adv. Mater., 2024, e2407029 Search PubMed.
- J. Yang, W. Wang, H. Yang and D. Wang, Green Chem., 2020, 22, 6489 RSC.
- Z. Qin, Z. Wen, Y. Xu, Z. Zheng, M. Bai, N. Zhang, C. Jia, H. B. Wu and G. Chen, Small, 2022, 18, 2106719 CrossRef CAS PubMed.
- T. Wang, H. Luo, Y. Bai, J. Li, I. Belharouak and S. Dai, Adv. Energy Mater., 2020, 10, 2001204 CrossRef CAS.
- T. Yang, Y. Lu, L. Li, D. Ge, H. Yang, W. Leng, H. Zhou, X. Han, N. Schmidt, M. Ellis and Z. Li, Adv. Sustainable Syst., 2020, 4, 1900088 CrossRef CAS.
- K. Park, J. Yu, J. Coyle, Q. Dai, S. Frisco, M. Zhou and A. Burrell, ACS Sustainable Chem. Eng., 2021, 9, 8214 CrossRef CAS.
- Y.-C. Yin, C. Li, X. Hu, D. Zuo, L. Yang, L. Zhou, J. Yang and J. Wan, ACS Energy Lett., 2023, 8, 3005 CrossRef CAS.
- N. Ogihara, K. Nagaya, H. Yamaguchi, Y. Kondo, Y. Yamada, T. Horiba, T. Baba, N. Ohba, S. Komagata, Y. Aoki, H. Kondo, T. Sasaki and S. Okayama, Joule, 2024, 8, 1364 CrossRef CAS.
- J. Song, H. Song, J. Song, G. Noh, H. Kim, J. Ma and J. J. Woo, Adv. Energy Mater., 2024, 2402106 Search PubMed.
- H. Gong, Y. Cao, B. Zhang, J. Zhang, Y. Zhang, H. Wang, S. Zhang, X. Wang, Y. Mao, S. Liu, C. Han, Q. Xiang, C. Zhou and J. Sun, Nat. Commun., 2024, 15, 10243 CAS.
- L. Zhang, Z. Xu and Z. He, ACS Sustainable Chem. Eng., 2020, 8, 11596 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2025 |