Mass production of robust hydrogel electrolytes for high-performance zinc-ion batteries†
Linlin
Ma
ab,
Xiaojing
Liu
ab,
Jihao
Fan
b,
Xiaodong
Yu
b,
Longsheng
Cao
*b and
Chuangqi
Zhao
 *ab
*ab
aSchool of Chemistry and Materials Science, University of Science and Technology of China, Hefei, 230026, P. R. China. E-mail: zhaochuangqi@ustc.edu.cn
bSuzhou Institute for Advanced Research, University of Science and Technology of China, Suzhou, 215123, P. R. China. E-mail: caolsh@ustc.edu.cn
First published on 15th January 2025
Abstract
Hydrogel electrolytes are crucial for solving the problems of random zinc dendrite growth, hydrogen evolution reactions, and uncontrollable passivation. However, their complex fabrication processes pose challenges to achieving large-scale production with excellent mechanical properties required to withstand multiple cycles of mechanical loads while maintaining high electrochemical performance needed for the new-generation flexible zinc-ion batteries. Herein, we present a superspreading-based strategy to produce robust hydrogel electrolytes consisting of polyvinyl alcohol, sodium alginate and sodium acetate. The hydrogel electrolytes have a tensile strength of 54.1 ± 2.5 MPa, a fracture strain of up to 1113 ± 37%, and a fracture toughness of 374.1 ± 6.1 MJ m−3, showcasing endurance of 2500 cycles at 80% strain without damage. Besides, the hydrogel electrolytes feature a high ionic conductivity of 14 mS cm−1 and a Zn2+ transference number of 0.62, as interfacial regulation enables the symmetric cell to achieve 1300 hours of highly stable and reversible zinc plating/stripping. As a preliminary attempt toward mass production, soft-pack batteries assembled using modified hydrogel electrolytes demonstrate robust machinability, with minimal voltage change after being bent and deformed 100 times. This work is expected to pave the way for developing a convenient hydrogel electrolyte for effective and stable zinc-ion batteries.
New conceptsWe demonstrate a novel technique that can produce large-area and robust hydrogel electrolytes in a continuous, roll-to-roll fashion via a superspreading-based strategy. Compared with current methods, our strategy has several unprecedented advances in the field of hydrogel electrolytes: (1) our strategy can realize the large-area fabrication of hydrogel electrolytes in a roll-to-roll production system. The fabricated hydrogel electrolytes are defect free and uniform. (2) The mechanical properties of our hydrogel electrolytes are superior to those of all existing hydrogel electrolytes. The tensile strength and toughness are as high as 54.1 ± 2.5 Mpa and 374.1 ± 6.1 MJ m−3, showcasing an endurance of 2500 cycles at 80% strain without damage. (3) The electrochemical performance of our hydrogel electrolyte configured zinc-ion batteries is superior to those of other reported hydrogel electrolytes. The ionic conductivity and Zn2+ transference number are as high as 14 mS cm−1 and 0.62, as interfacial regulation enables the symmetric cell to achieve 1300 hours of highly stable and reversible zinc plating/stripping. We believe that our design concept together with the listed achievements (1)–(3) will bring a significant advancement in the continuous preparation of hydrogel electrolytes. |
1. Introduction
The demand for energy storage devices that exhibit convenience and stability is increasing with the continuous development of smart electronic devices. Zinc-ion batteries have attracted widespread attention due to their low oxygen reduction potential (−0.76 V) compared to the standard hydrogen electrode, high theoretical specific capacity (820 mA h g−1), low toxicity, and abundant zinc resources.1,2 However, uncontrolled dendrite growth at the electrolyte–electrode interface, accompanied by active water-induced side reactions and interfacial polarization in aqueous systems,3,4 leads to unavoidable anode volume expansion and short circuits, ultimately compromising the stability of high-performance zinc ion batteries. Thus, it is highly desirable to develop viable strategies in interface optimization to protect the zinc anode from these processes.As alternatives to traditional liquid electrolytes, hydrogel electrolytes have been extensively studied and used in zinc-ion batteries.5,6 The polymer network of hydrogel electrolytes contains adjustable functional groups, which can regulate ion transport behavior and improve desolvation kinetics.7 Moreover, the excellent mechanical properties of the hydrogel electrolyte can maintain the integrity of the structure under multiple cycles of load,8,9 effectively facilitate convenient processing and prevent rampant interface evolution,10,11 resulting in efficient, safe and convenient zinc-ion batteries. Recently, numerous reports have focused on improving the ionic conductivity and Zn2+ transference number of hydrogel electrolytes by altering the polymer matrix12,13 and introducing functional groups,14,15 establishing a stable interface between the electrode and the electrolyte.16,17 Extensive efforts have also been made to strengthen or toughen hydrogel electrolytes through methods such as double-network formation,18 multiple crosslinking incorporation,19 nanofiller integration,20 and creation of nano-crystalline domains21 or anisotropic structures.22 Nevertheless, these strategies typically involve complex techniques and limited mechanical properties, creating obstacles in scaling up production. Therefore, it is still a great challenge to realize a large-area hydrogel electrolyte with high mechanical properties and electrochemical performance simultaneously.
Herein, we present a superspreading-based strategy for the continuous fabrication of composite hydrogel electrolytes with high toughness, optimized ionic conductivities and an elevated Zn2+ transference number. Consequently, the composite hydrogel electrolytes enable Zn‖Zn symmetric cells to achieve reversible plating/stripping for over 1300 h. Additionally, soft-pack batteries assembled using our hydrogel electrolytes show minimal voltage change under repeated bending deformation. We envision that our strategy will offer a universal and efficient pathway toward the next generation of zinc-ion batteries, combining both effectiveness and safety to meet the large-area energy storage demand.
2. Results and discussion
2.1. Mechanism and fabrication of composite hydrogel electrolytes
Fig. 1a presents a schematic illustration that describes the mechanism and fabrication of polyvinyl alcohol–sodium alginate–sodium acetate (denoted as PVA–SA–acetate) composite hydrogel electrolyte.The metal ion (Ca2+) aqueous solution was extruded from an array of syringes onto the non-woven fabric surface. The mixed dilute solution of PVA, SA and sodium acetate spread rapidly and entirely (defined as superspreading) (Movie S1, ESI†) on the surface of this nonwoven fabric impregnated with Ca2+ solution within one second in air (Movie S2, ESI†).23 The Ca2+ created crosslinks of SA, and consequently the superspreading layer containing PVA, SA and sodium acetate was converted into a calcium alginate (CA) hydrogel film. This CA hydrogel film could then be readily separated from the non-woven fabric surface after immersion in the water bath. After the hydrogel film was dried, it was soaked in a 2M ZnSO4 aqueous electrolyte to sufficiently complete ion exchange, resulting in the mass production of a PVA–SA–acetate hydrogel electrolyte (Fig. 1b).
The hydrogel electrolyte prepared via this process exhibits good mechanical stability and can lift a weight of 1.5 kg (Fig. 1c). Scanning electron microscopy (SEM) images were further taken to analyze the interior structures of hydrogels. As depicted in the cross-section morphology, PVA–SA–acetate features hierarchical pores with diameters of 2–3 μm (Fig. 1d). These well-organized interconnecting pores are postulated to provide additional pathways for ion transport.24 Fourier-transform infrared (FTIR) spectroscopy (Fig. 1e) reveals the structural information and internal molecular interaction of the individual PVA, SA, and PVA–SA–acetate hydrogel electrolyte. The peaks plotted at 1616 and 1438 cm−1 are derived from the typical –COO– asymmetric and symmetric stretching vibrations, respectively.25 The significant blue shift of –COO– asymmetric stretching after hydrogel formation is attributed to the coordination of Zn2+ ions under electrostatic forces, which is quite conducive to the confinement and rapid guidance of Zn2+ ions along polymer chains. In this context, the –COO– groups, with a p–π conjugated structure between carbon and oxygen atoms, could attract Zn2+ ions for pre-embedding in the binding sites and thereby accelerating transport kinetics.26 Moreover, FTIR results manifest the presence of massive hydrophilic functional groups in PVA–SA–acetate, which contributes to the immobilization of water molecules through hydrogen bonds. And the O–H stretching vibration peaks exhibited a significant blueshift, indicating that PVA, SA and sodium acetate formed more abundant hydrogen bonds, which destroyed the original hydrogen bond network of H2O (Fig. S1, ESI†).
2.2. Mechanical properties of composite hydrogel electrolytes
The mechanical performance of hydrogel electrolytes is crucial as it directly impacts the adaptability of zinc ion batteries to external forces and deformations.27 The prepared composite hydrogel electrolytes can be engineered into arbitrary shapes without any damage under different mechanical manipulations, including knotting and twisting (Fig. S2, ESI†).We quantitatively studied the mechanical properties of the PVA–acetate, SA–acetate, PVA–SA and PVA–SA–acetate composite hydrogel electrolyte by tensile testing, in which the strength and toughness of PVA–SA–acetate is superior to the others (Fig. 2a and b). Subsequently, by precisely adjusting the ratio among the PVA, SA and sodium acetate, the PVA–SA-0.03 hydrogel electrolytes exhibit a tensile strength of 54.1 ± 2.5 MPa, a fracture strain of up to 1113 ± 37%, and a fracture toughness of 374.1 ± 6.1 MJ m−3 (Fig. 2c and d). The substantial enhancement in the mechanical strength of the PVA–SA–acetate hydrogel is mainly attributed to the intermolecular hydrogen bonds of PVA and SA to provide additional physical cross-linking sites which enhances the stability and mechanical strength of the hydrogel.28 Furthermore, the addition of sodium acetate effectively enhances the toughness of the hydrogel electrolytes through ion-induced chain aggregation.29
 | ||
| Fig. 2 Mechanical properties of hydrogel electrolytes. (a) Stress–strain curves of PVA–SA–acetate, PVA–SA, PVA–acetate and SA–acetate. (b) The histogram of strength and toughness of PVA–SA–acetate, PVA–SA, PVA–acetate and SA–acetate. (c) Stress–strain curves of PVA–SA-x (x = 0.00, 0.01, 0.03, 0.05, 0.07, and 0.09) hydrogel electrolytes. (d) The histogram of strength and toughness of PVA–SA-x hydrogel electrolytes. (e) Strength versus strain of this work and other reported hydrogel electrolytes. (f) Toughness versus strength of this work and other reported hydrogel electrolytes. The data used are summarized in Table S1 and Table S2 (ESI†). | ||
Our PVA–SA–acetate hydrogel electrolytes have high strength and strain, surpassing the values seen in many reported tough hydrogel electrolytes (Fig. 2e and Table S1, ESI†). The PVA–SA–acetate demonstrated excellent toughness of 374.1 ± 6.1 MJ m−3 in the absence of flaws, as the direct result of their combination of high strength and high ductility (Fig. 2f and Table S2, ESI†). Since Zn plating/stripping poses repeated stress to the hydrogel electrolyte, excellent mechanical properties under multiple cycles are critical for long-term stability. Under continuous loading–unloading multiple cycles, the mechanical properties of PVA–SA–acetate do not decay dramatically at a strain of 50% after 2500 cycles (Fig. S3a, ESI†). Encouragingly, PVA–SA–acetate can still be realized without any damage even under 80% strain (Fig. S3b, ESI†).
2.3. Electrochemical performance of composite hydrogel electrolytes configured in symmetric cells
In addition to mechanical performance, regulating the interface and optimizing the Zn2+deposition process is also crucial for inhibiting dendrite formation and mitigating parasitic reaction through hydrogel electrolytes to enhance the stability of zinc anodes.29,30 The ionic conductivity value of PVA–SA–acetate was calculated as 14 mS cm−1 through electrochemical impedance spectroscopy (EIS), which is higher than that of liquid electrolyte (4.8 mS cm−1, representing 2M ZnSO4 with a glass cellulose separator) (Fig. 3a).Then, the Zn2+ transference number (tZn2+) is calculated from the steady current method to validate the fast ion transport in PVA–SA–acetate. The PVA–SA–acetate exhibits a higher Zn2+ transference number of 0.62 (Fig. 3b) compared to the liquid electrolyte (0.49) (Fig. S4, ESI†). It can be inferred that the PVA–SA–acetate hydrogel electrolyte, with polar functional groups and additional ion transport pathways, can modulate Zn2+ deposition behavior, ensure uniform ion distribution, and reduce concentration polarization at the interface, further alleviating dendrite growth.
To reveal the stability and reversibility of the zinc anode, long-term galvanostatic cycling performances were conducted in a Zn‖Zn symmetrical cell. With the assistance of the PVA–SA–acetate hydrogel electrolyte, the Zn anode sustains a significantly prolonged lifespan of up to 1100 h at 0.5 mA cm−2 and 0.5 mA h cm−2 which is almost thirteen times that in a liquid electrolyte (Fig. 3c). Additionally, the symmetric cell with the aid of the PVA–SA–acetate hydrogel electrolyte holds steady as the current density increases from 0.1 to 2 mA cm−2 without any abnormal voltage response (Fig. S5, ESI†). The outstanding cycle life of the PVA–SA–acetate symmetrical cell is attributed to the high strength, toughness and good mechanical stability under multiple cycles of the hydrogel electrolytes, which prevents hydrogel damage during cycling and enables reversible zinc ion plating/stripping. Moreover, it demonstrates stable cycling at a current density of 2 mA cm−2 (at the first several cycles, apply a 0.5 mA cm−2 for activation), emphasizing the decisive role of PVA–SA–acetate hydrogel electrolyte in high current tolerance through enhanced transport kinetics (Fig. 3d and Fig. S6, ESI†).
To investigate this mechanism, morphological changes in the deposited Zn electrode after cycling were analyzed. Through SEM characterization (Fig. S7, ESI†), a significant amount of layered dendrites was observed on the zinc surface in the liquid electrolyte. In contrast, a more uniform coating was observed on the zinc surface in the PVA–SA–acetate hydrogel electrolytes indicating the gradual formation of a preferred homogeneous deposition structure during cycling.31 To further reveal the Zn deposition morphology, atomic force microscopy (AFM) was also performed on the Zn surface after 100 cycles (Fig. 4a–c). Clearly, compared to the pristine state and the PVA–SA–acetate hydrogel electrolyte, the Zn surface under the liquid electrolyte appears quite rough and uneven, indicating the presence of a thick layer of dendrites. To accurately investigate the short-circuit failure mechanism of the Zn anode, the morphology of Zn//PVA–SA–acetate and Zn//liquid electrolyte at different current densities were characterized using SEM. The top view of the zinc anode exhibits a smooth surface with no dendrite formation at areal capacities of 1 mA h cm−2 and 2 mA h cm−2 (Fig. 4d and e). Even at a high capacity of 4 mA h cm−2, a planar morphology can be maintained (Fig. 4f). In contrast, the dendrites can be observed on the zinc surface in the presence of the liquid electrolyte. When the capacity is 1 mA h cm−2, vertically aligned nanosheet-like dendrites can be seen on the zinc surface (Fig. 4g). At areal capacities of 2 mA h cm−2 (Fig. 4h) and 4 mA h cm−2 (Fig. 4i), rapid accumulation can be observed. In comparison, the PVA–SA–acetate hydrogel electrolytes significantly suppress the growth of Zn dendrites.
Simultaneously, in consideration of the plating/stripping reversibility, Zn‖Ti asymmetric cells were also assembled to measure coulombic efficiency (CE).
The cell with PVA–SA–acetate demonstrates a stabilized CE of 99.6% (Fig. S8, ESI†), further indicating excellent reversibility. To elucidate the failure mechanism of zinc plating/stripping processes, the electrochemical properties of different electrolytes were deeply investigated. Compared to liquid electrolytes, the HER in the PVA–SA–acetate hydrogel electrolyte are retarded, suggesting the improved corrosion-resistance ability (Fig. 4j).
The corrosion current of the Zn anode calculated from the Tafel curve in the PVA–SA–acetate hydrogel electrolyte is 2.97 μA, which is significantly lower than that in the liquid electrolyte (16.6 μA) (Fig. 4k). It can be attributed to the abundant presence of hydrophilic carboxyl groups, more free water is transformed into bound water, reducing the electrochemical activity of water and reducing the occurrence of side reactions.32
Further verified by ex situ XRD (Fig. S9, ESI†), a large amount of (Zn(OH)2)3(ZnSO4)(H2O)5 was formed on the Zn surface with liquid electrolyte after cycling, which was consistent with the EDS mapping images of elemental composition (Fig. S10, ESI†). For the Zn surface with PVA–SA–acetate, no distinctive by-product was detected in the XRD pattern. Additionally, the desolvation activation energies (Ea) of hydrated Zn ions were calculated by using the Arrhenius equation (Fig. 4l and Fig. S11, 12, ESI†) to be 23.8 and 22.2 kJ mol−1 for the liquid electrolyte and PVA–SA–acetate hydrogel electrolyte, respectively. It manifested that PVA–SA–acetate hydrogel electrolyte could remove the water sheath from [Zn(H2O)6]2+ and accelerate the desolvation process.33 In addition, the low charge transfer resistance (Rct) indicates that the charge transfer process of the PVA–SA–acetate anode is more efficient and the electroplating kinetics is faster, which is attributed to the high ion conductivity network and abundant active sites in the PVA–SA–acetate hydrogel electrolytes (Fig. S13, ESI†).34
2.4. Electrochemical performance of composite hydrogel electrolytes configured full cells
Based on the ameliorated electrochemical stability of electrolyte–anode interface, the full cell with PVA–SA–acetate was assembled to get more insight into the multiple merits of such a hydrogel electrolyte (Fig. 5a).The Zn‖PVA–SA–acetate‖MnO2 full cell shows a capacity retention rate of 83.3% after 200 cycles at the current density of 0.1 A g−1 (Fig. 5b). The cyclic voltammetry (CV) curves of the full cell show the redox peaks at 1.35 V and 1.61 V, representing the reversible insertion and deinsertion of Zn ions within the cathode (Fig. S14, ESI†). Charge–discharge curves at different current densities (Fig. S15, ESI†) demonstrate good reversibility and clear discharge plateaus. A large specific capacity of 341.48 mA h g−1 is achieved at the current density of 0.02 A g−1, and even after testing under high current densities, 96.7% of the capacity (331.13 mA h g−1) is retained when the current density returns to 0.02 A g−1 (Fig. S16, ESI†), while only 50% is retained in the liquid electrolyte.
Various characterization studies were conducted to illustrate the excellent electrochemical performance of the Zn‖PVA–SA–acetate‖MnO2 full cell. The peak currents from CV curves at different scan rates (Fig. S17a, ESI†) were used to calculate the b values of 0.48 for the liquid electrolyte and 0.55 for PVA–SA–acetate, indicating that the kinetics of the Zn‖PVA–SA–acetate‖MnO2 full cell are controlled by ion diffusion. The CV curve of the Zn‖PVA–SA–acetate‖MnO2 full cell at a scan rate of 5 mV s−1, where diffusion contributes 54.78% to the total capacity, demonstrating partial diffusion contribution of the MnO2 electrode, enabling high rate capability (Fig. S17b, ESI†). Additionally, the capacitance-controlled kinetics become more prominent, showcasing the rapid kinetics and ion diffusion of the Zn‖PVA–SA–acetate‖MnO2 full cell (Fig. S17c, ESI†).
Furthermore, Zn–MnO2 pouch cells (length is 5 cm, width is 4 cm) with PVA–SA–acetate are assembled to validate the practical application of the PVA–SA–acetate hydrogel electrolyte (Fig. 5c). The pouch cell underwent 100 bending experiments, with no significant change in voltage reading (Fig. 5d and Fig. S18, ESI†). Through destructive tests such as bending, piercing, and even cutting, the pouch cells exhibit minimal voltage fluctuations, indicating excellent flexibility and strong adhesion of PVA–SA–acetate at electrolyte–electrode interface (Fig. 5e, f and Fig. S19, ESI†). In addition, our PVA–SA–acetate has also been assembled into larger pouch batteries (length is 12 cm, width is 10 cm) with steady voltage reading (Fig. S20, ESI†). These excellent characteristics enable the hydrogel electrolytes to be widely used in quasi-solid-state energy storage devices.
3. Conclusions
In summary, we have presented a generalized and scalable strategy to continuously fabricate composite PVA–SA–acetate hydrogel electrolytes. PVA–SA–acetate has excellent mechanical properties under multiple cycles of loads, with high strength and high toughness. It also showed improved ionic conductivity and Zn2+ transference number attributed to abundant polar functional groups and specific pore structures, which enhanced Zn2+ transport kinetics. In addition, the optimized hydrogen bond network significantly reduces the free water, accelerates the Zn2+ desolvation process, and promotes homogeneous deposition at the interface, thereby minimizing water-related side reactions and inhibiting dendrite growth. Our strategy holds significant potential to stabilize the electrolyte–electrode interface and chart a new direction for the development of effective, convenient and stable zinc-ion batteries in mass production.4. Experiments
4.1. Preparation of PVA–SA–acetate composite hydrogel electrolyte
First, we made stock solutions for the polymers dispersing 8 g of polyvinyl alcohol (PVA) solid in 92 mL deionized water to obtain an 8 wt% PVA solution and 4 g of sodium alginate (SA) solid in 96 mL deionized water to obtain a 4 wt% SA solution. Sodium acetate was dispersed in deionized water to obtain the sodium acetate solution (the solution concentrations are 0.01 mol L−1, 0.03 mol L−1, 0.05 mol L−1, 0.07 mol L−1, 0.09 mol L−1). PVA, SA and sodium acetate mixture solution denoted as PVA–SA–acetate. For different concentrations of sodium acetate, the mixture solutions were denoted as PVA–SA-0.01, PVA–SA-0.03, PVA–SA-0.05, PVA–SA-0.07 and PVA–SA-0.09. During the fabrication process, the metal ion aqueous solution (1 M CaCl2) and PVA–SA–acetate solution were continuously extruded from the array of syringes onto the non-woven fabric surface respectively. The distance between two adjacent syringes was set as 1 cm, the flow rate of the metal ion aqueous solution was determined as 60 mL h−1, the PVA–SA–acetate solution was 90 mL h−1, and the moving speed of the non-woven fabrics was 5 mm s−1. Because of the superspreading of the dropped solution on the nonwoven fabric and in situ cross-linking of SA and calcium ions, the reaction solution layer could be rapidly solidified into a hydrogel film on the nonwoven fabric interface, and this hydrogel film could be easily separated from the nonwoven fabric surface when the hydrogel film was transferred into a water bath. Finally, the hydrogel film was immersed into 2M ZnSO4 to obtain the PVA–SA–acetate hydrogel electrolyte.4.2. Preparation of the MnO2 cathode
Dispersing potassium permanganate (KMnO4) (0.237 g) and manganese sulfate monohydrate (MnSO4·H2O) (0.3803 g) separately in deionized water (15 mL), with stirring to dissolve, the KMnO4 solution was added slowly to the MnSO4·H2O solution with stirring for 30 minutes. The above solution was transferred into a hydrothermal vessel, and heated at 160 °C for 12 hours. After cooling down to the temperature, the precursor was washed three times with deionized water, and vacuum dried at 80 °C to obtain manganese dioxide (MnO2) cathode material.4.3. Materials characterization
The SEM images were obtained using a Quanta S operated at 20 kV. Optical imaging of the hydrogel electrolyte was performed using a Canon R10. ATR FTIR spectra were obtained using iN10. The crystal structures were characterized using a Germany Bruker X-Ray diffractometer (Cu Ka, 30 KV and 20 mA) with λ = 0.154 nm.4.4. Tensile testing
All mechanical tests were conducted using a tensile machine ESM303 from Mark-10 Corporation. The stress–strain curves were obtained by dividing the measured force by the initial cross-sectional area and dividing the measured displacement via the initial clamp distance. Tests were performed at a rate of 5 mm min−1.4.5. Electrochemical measurements
A CR2032 coin cell was assembled to evaluate the electrochemical performance. We prepared the working electrode by mixing the as-prepared MnO2 (70%), super P (20%) and polyvinylidene difluoride (PVDF) binder (10%) in N-methyl-2-pyrrolidone (NMP) solvent and then coating on Ti foil. Cyclic voltammogram (CV) curves were collected at different scan rates (with a voltage range from 0.9 to 1.8 V (vs. Zn/Zn2+) in a full-cell system). The electrochemical impedance spectroscopy (EIS) was collected using a CHI760 in the frequency ranging from 0.01 to 10−5 Hz. Meanwhile, the galvanostatic charging and discharging process were used to conduct cycling performance and specific capacity.4.6. The calculation of ionic conductivity
The EIS was collected by the CHI760 in the frequency ranging from 0.01 to 10−5 Hz. The relation between σ and R is calculated according to the following eqn (1):35 | (1) |
4.7. The Calculation of ion migration activation energy
The electrochemical impedance spectroscopy (EIS) was collected by the CHI 760 in the frequency ranging from 0.01 to 10−5 Hz at different temperatures. The relation between Ea and R is calculated according to the following eqn (2):36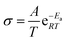 | (2) |
4.8. The calculation of diffusion coefficients
| i = aVb | (3) |
| i(v) = k1v + k2v1/2 | (4) |
Author contributions
All authors have given approval to the final version of the manuscript.Data availability
The datasets generated during and/or analyzed during the current study are available from the authors on reasonable request.Conflicts of interest
There are no conflicts to declare. The authors declare no conflict of interest.Acknowledgements
This research was supported by the Youth Fund of the National Natural Science Foundation of China (22305244); the Start-up Funding for Suzhou Institute for Advanced Research, University of Science and Technology of China (KY2260080033); the Start-up Funding for University of Science and Technology of China (KY2060000233); and the CAS Hundred Talent Program (KJ2060007007). The authors thank the Physical and Chemical Analysis Center at Suzhou Institute for Advanced Research, University of Science and Technology of China, the Key Laboratory of Precision and Intelligent Chemistry, University of Science and Technology of China and Suzhou Key Laboratory of Bioinspired Interfacial Science.References
- L. Zhou, T. Du, J. Li, Y. Wang, W. Luo and J. Wang, Mater. Horiz., 2022, 9, 2722–2751 RSC.
- Y. Liu, A. Gao, J. Hao, X. Li, J. Ling, F. Yi, Q. Li and D. Shu, Chem. Eng. J., 2023, 452, 139605 CrossRef CAS.
- Y. Jen, S. Wang, J. Zhang, W. Lu, X. Zhou and T. Chen, Mater. Horiz., 2021, 8, 351–369 RSC.
- X. Wang, S. Zheng, J. Xiong, Z. Liu, Q. Li, W. Li and F. Yan, Adv. Mater., 2024, 2313845 CrossRef CAS.
- Y. Wu, Y. Zhang, H. Wu, J. Wen, S. Zhang, H. Zhang, H. Xue, J. Gao and Y. Mai, Adv. Mater., 2023, 35, 2210624 CrossRef CAS.
- H. Wan, B. Wu, L. Hou and P. Wu, Adv. Mater., 2024, 36, 2307290 CrossRef CAS.
- Z. Chen, T. Shen, M. Zhang, X. Xiao, H. Wang, Y. Luo, Z. Jin and C. Li, Adv. Funct. Mater., 2024, 2314864 CrossRef CAS.
- S. Lin, X. Liu, J. Liu, H. Yuk, H. C. Loh, G. A. Parada, C. Settens, J. Song, A. Masic, G. H. Mckinley and X. Zhao, Sci. Adv., 2019, 5, eaau8528 CrossRef.
- Z. Chen, T. Shen, M. Zhang, X. Xiao, H. Wang, Q. Lu, Y. Luo, Z. Jin and C. Li, Adv. Funct. Mater., 2024, 2314864 CrossRef CAS.
- M. Sun, G. Ji, M. Li and J. Zheng, Adv. Funct. Mater., 2024, 2402004 CrossRef CAS.
- H. Zhang, X. Gan, Y. Yan and J. Zhou, Nano-Micro Lett., 2024, 16, 106 CrossRef CAS.
- Q. Liu, Z. Yu, Q. Zhuang, J. Kim, F. Kang and B. Zhang, Adv. Mater., 2023, 35, 2300498 CrossRef CAS PubMed.
- Q. Fu, S. Hao, X. Zhang, H. Zhao, F. Xu and J. Yang, Energy Environ. Sci., 2023, 16, 1291–1311 RSC.
- Y. Yan, S. Duan, B. Liu, S. Wu, B. Yao, Y. Du, T. Wang, Y. Li and X. He, Adv. Mater., 2023, 35, 2211673 CrossRef CAS.
- Q. He, Y. Zhong, J. Li, Y. Yang, S. Liang, Z. Chang, G. Fang and A. Pan, Adv. Energy Mater., 2024, 14, 2400170 CrossRef CAS.
- Y. Song, W. Lu, H. Yang, C. Wu, W. Wei, G. Kuang, Y. Chen and X. Ouyang, Nano Energy, 2024, 120, 109094 CrossRef CAS.
- J. Sarpong, C. Chang, J. Hao, T. Li, X. Deng, C. Han and B. Li, Adv. Mater., 2024, 2403214 CrossRef.
- Z. Wang, X. Zheng, T. Ouchi, T. Kouznetsova, H. Beech, S. Ron, T. Matsuda and B. Bowser, Science, 2021, 374, 193–196 CrossRef CAS PubMed.
- P. Lin, S. Ma, X. Wang and F. Zhou, Adv. Mater., 2015, 27, 2054–2059 CrossRef CAS PubMed.
- Z. Hu and G. Chen, Adv. Mater., 2014, 26, 5950–5956 CrossRef CAS.
- S. Lin, X. Liu, J. Liu, H. Yuk, H. Loh, G. Parada, C. Settens, J. Song, A. Masic and X. Zhao, Sci. Adv., 2019, 5, eaau8528 CrossRef.
- M. Mredha, Y. Guo, T. Nonoyama, T. Nakajima, T. Kurokawa and J. Gong, Adv. Mater., 2018, 30, 1704937 CrossRef PubMed.
- C. Zhao, P. Zhang, J. Zhou, M. Liu, S. Wang and L. Jiang, Nature, 2020, 580, 210–215 CrossRef CAS PubMed.
- S. Huang, Y. Jiao and P. Wu, Adv. Mater., 2022, 34, 2110140 CrossRef CAS PubMed.
- C. Liu, F. Guo, Q. Yang, H. Mi, C. Ji, N. Yang and J. Qiu, Small Methods, 2023, 7, 2201398 CrossRef CAS.
- L. Sun, Y. Yao, L. Dai, M. Jiao, B. Ding, Q. Yu, J. Tang and B. Liu, Energy Storage Mater., 2022, 47, 187–194 CrossRef.
- H. Xia, G. Xu, X. Cao, C. Miao, H. Zhang, P. Chen, Y. Zhou, W. Zhang and Z. Sun, Adv. Mater., 2023, 35, 2301996 CrossRef CAS.
- Y. Meng, J. Zhou and T. Qian, Adv. Funct. Mater., 2022, 32, 2206653 CrossRef CAS.
- M. Hua, S. Wu, Y. Ma, Y. Zhao, Z. Chen, H. Zhou, X. Zhu and X. He, Nature, 2021, 590, 594–599 CrossRef CAS.
- C. Tian, J. Wang and Y. Hu, Angew. Chem., Int. Ed., 2023, e202310970 CAS.
- H. Lu, J. Hu, L. Wang, J. Li, Y. Li, J. Zhao and B. Xu, Adv. Funct. Mater., 2022, 32, 2112540 CrossRef CAS.
- M. Sun, G. Ji, M. Li and J. Zheng, Adv. Funct. Mater., 2024, 2402004 CrossRef CAS.
- H. Zhang, X. Gan, Z. Song and J. Zhou, Angew. Chem., Int. Ed., 2023, 62, e202217833 CrossRef CAS.
- H. Lu, J. Hu, L. Wang, J. Li, X. Ma, Z. Zhu, H. Li, Y. Zhao, Y. Li, J. Zhao and B. Xu, Adv. Funct. Mater., 2022, 32, 2112540 CrossRef CAS.
- D. Feng, Y. Jiao and P. Wu, Angew. Chem., Int. Ed., 2023, 62, e202215060 CrossRef CAS.
- J. Wang, H. Tang, H. Wang, R. Yu and D. Wang, Mater. Chem. Front., 2017, 1, 414–430 RSC.
- H. Lindström, S. Södergren, A. Solbrand, H. Rensmo, J. Hjelm, A. Hagfeldt and S. Lindquist, J. Phys. Chem. B, 1997, 101, 7717–7722 CrossRef.
- M. Zhang, Y. Yang, M. Li, Q. Shang, J. Yu, K. Shen, Y. Zhang and Y. Cheng, Adv. Mater., 2023, 35, 2301551 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: https://doi.org/10.1039/d4mh01716a |
| This journal is © The Royal Society of Chemistry 2025 |




