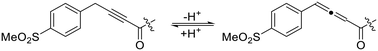Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Discovery of an internal alkyne warhead scaffold for irreversible hTG2 inhibition
Lavleen K.
Mader
 ,
Namita
Maunick
,
Jessica E.
Borean
,
Namita
Maunick
,
Jessica E.
Borean
 and
Jeffrey W.
Keillor
and
Jeffrey W.
Keillor
 *
*
Department of Chemistry and Biomolecular Sciences, University of Ottawa, Ottawa, Ontario K1N 6N5, Canada. E-mail: jkeillor@uottawa.ca
First published on 9th October 2025
Abstract
Human tissue transglutaminase (hTG2) is a multifunctional enzyme with both protein cross-linking and G-protein activity. Dysregulation of these functions has been implicated in diseases such as celiac disease and cancer, prompting the development of hTG2 inhibitors, many of which act covalently via a pendant electrophilic warhead. Most small molecule hTG2 inhibitors to date feature terminal, sterically minimal warheads, based on the assumption that bulkier electrophiles impair binding and reactivity. Here, we report structure–activity relationships (SAR) of a novel internal alkynyl warhead scaffold for irreversible inhibition of hTG2. This series includes one of the most potent non-peptidic hTG2 inhibitors reported to date. We demonstrate that this scaffold not only inhibits transamidase activity but also abolishes GTP binding, while exhibiting excellent isozyme selectivity. In addition, we investigate the tunability and stability of this warhead, providing insights into its broader applicability. Through detailed kinetic analysis, this study establishes a new scaffold for irreversible hTG2 inhibition and expands the design principles for covalent warheads beyond traditional terminal systems.
Introduction
Human tissue transglutaminase (hTG2) is one of nine enzymes in the transglutaminase (TGase) family, characterized by their ability to catalyze protein cross-linking in the extracellular matrix via a transamidation reaction involving a catalytic cysteine (Cys277).1,2 Uniquely, hTG2 is multifunctional: in addition to transamidation, it can bind guanosine triphosphate (GTP) and act as a G-protein in intracellular signalling. These two functions are mutually exclusive; each is associated with a distinct conformational state.1,3,4 In the extracellular space, high calcium concentrations favour an extended “open” conformation that exposes the transamidase active site but abolishes the GTP binding pocket. Conversely, under intracellular conditions, low calcium concentrations favour a compact “closed” conformation, allowing the GTP binding site to form and occluding the transamidase site.5–8 Dysregulation of either function has been linked to pathologies including celiac diseases,9 fibrosis,10 and cancer,11,12 making hTG2 an attractive therapeutic target.The conformationally-based exclusivity of the two functions enables a unique targeted covalent inhibition (TCI) strategy, whereby irreversible covalent modification of Cys277 in the transamidase site locks hTG2 in its open conformation, simultaneously blocking both of its functions.13 Several small molecule and peptidomimetic TCI scaffolds have been developed (Fig. 1),14–19 but peptidic inhibitors often suffer from poor pharmacokinetics, prompting increased interest in non-peptidic scaffolds.14
 | ||
| Fig. 1 Representative small molecule hTG2 inhibitors.15,17–19 | ||
A key determinant of covalent inhibition is the nature of the electrophilic warhead. In a recent study, we evaluated multiple warheads on a scaffold (EB-2-16) originally reported by Badarau et al.17,20 We found that effective Cys277 engagement generally required “terminal” warheads (i.e., minimally hindered electrophiles capable of accessing the active site tunnel). This is consistent with most reported irreversible hTG2 inhibitors, which typically feature a simple acrylamide warhead (Fig. 1). However, our group also previously identified CP4d,19 a structurally distinct inhibitor bearing a bulky, internal electrophilic warhead. Despite the presence of this warhead, CP4d acted reversibly and failed to react irreversibly with Cys277 or to lock hTG2 into the open state,11,21 and was not pursued further.
Here, we revisit CP4d with a library of alkynyl analogues, which interestingly, function as potent irreversible inhibitors that abolish GTP binding and exhibit high isozyme selectivity. These compounds feature an internal keto-alkyne-phenyl warhead, with tunable reactivity properties (kinact parameter). Our findings challenge the prevailing view that hTG2 covalent inhibitors require terminal, sterically minimal warheads, and provide a novel scaffold for future structure-based inhibitor design.
Results and discussion
Discovery of alkynyl scaffold
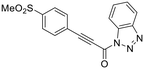
CP4d represents a peculiar class of hTG2 inhibitors. It bears an obvious electrophilic group and is a competitive inhibitor with the acyl-donor substrate of our standard activity assay (as a surrogate for transamidase activity), but does not irreversibly modify Cys277 or lock hTG2 in an open conformation.11,19,21 Given the structure of its warhead, CP4d would be expected to react covalently, even if only through reversible covalent modification, due to the strong electron withdrawing nitrophenyl group.22 Reactivity studies with CP4d and glutathione in solution showed rapid and irreversible GSH adduct formation in a matter of minutes, highlighting the high intrinsic reactivity of the warhead.23 These findings suggest that the compound may bind in the active site but not in a conformation that allows reaction with Cys277, leading to reversible competitive binding.Prior efforts in our group probed the reactivity of this scaffold with alkyne (KA22b) and bis-triazole derivatives.24 At the time it was assumed that these analogues were also reversible inhibitors, as kinetic analysis was performed using initial rate data over a very short period of time. However, an independent dialysis experiment later showed that no hTG2 activity was recovered after incubating with KA22b and dialyzing the inhibitor into buffer.23 Given the fact that the apparent IC50 and Ki values of this inhibitor were in the micromolar range, we ruled out “tight” binding as a cause of this result. This led us to our current hypothesis that the alkyne scaffold was in fact capable of irreversible inhibition and its evaluation and subsequent SAR analysis required rigorous irreversible inhibition kinetic analysis.
We evaluated KA22b (also referred to as 8A in this work) following our standard AL5 assay protocol, carried out under Kitz and Wilson conditions, and indeed observed classic time-dependent inhibition as expected for an irreversible inhibitor.25–27 The observed rate constants of inhibition (kobs) acquired from fitting of curves of absorbance vs. time (Fig. 2A) were plotted against inhibitor concentration corrected for the presence of a competitive substrate. The resulting hyperbolic curve (Fig. 2B) was fitted to a saturation kinetics model to obtain kinact = 0.90 ± 0.09 min−1 and KI = 1.80 ± 0.24 μM with an overall inhibitor efficiency of kinact/KI = 499 ± 0.85 × 103 M−1 min−1. This represents a remarkably efficient inhibitor to identify early in a campaign, on par with the lead scaffolds shown in Fig. 1.
We also demonstrated the irreversibility of inhibition by 8A by performing a substrate spike test as previously described,25 in which the assay is allowed to run until a plateau is reached in the uninhibited and inhibited reaction (Fig. 3). This plateau results from substrate depletion in the former, but complete enzyme inactivation in the latter. More substrate is then added (resulting in a concentration that is equivalent to its initial concentration in the assay), effectively diluting the reactions with substrate solution (Fig. 3, arrow). The activity of both reactions is then monitored again. The inhibition is confirmed to be irreversible as no activity is regained, whereas the activity of the uninhibited control returns to its initial activity, as observed in the first phase of the assay. The uninhibited control is crucial here to ensure that lack of activity is not due to enzyme degradation. Further validation of the irreversibility of this scaffold is provided by its ability to completely abolish GTP binding, unlike CP4d (see Inhibition of GTP binding below).
Design and synthesis
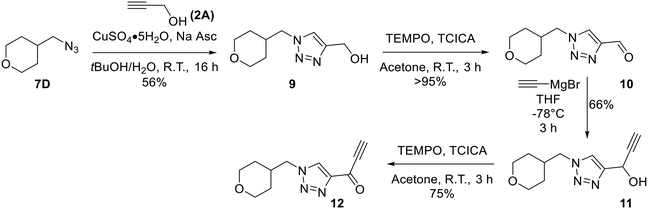
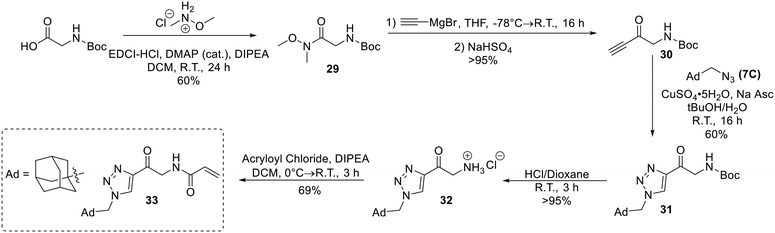
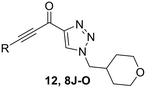
Confirmation of 8A as a potent irreversible hTG2 inhibitor prompted us to design and synthesize various derivatives to give better understanding of the pharmacophore of this scaffold and a potential binding mode, as well as reactivity of the warhead. 8A, while potent, poses several challenges. It is a very symmetrical, flat molecule which imparts poor solubility, and it also contains nitro groups which have mutagenic/toxicity potential.28 Moreover, the activated keto-alkyne warhead is very intrinsically reactive and has poor stability in GSH reactivity assays (see Intrinsic reactivity below).23 Therefore, prominent goals were to increase solubility and replace the aryl nitro groups, while tuning the warhead for stability.The synthesis of most analogues follows our previously reported procedure (Scheme 1).23,24 Aryl iodides 1A–H were either commercially available or synthesized according to known procedures (see Materials and methods). THP-protected propargyl alcohol (2B) was synthesized as previously described and azides 7A–J were synthesized by standard literature procedures. In the case of the pentafluorobenzene analogue (8K), additional protection and deprotection steps were required as side products from an SNAr reaction were observed when the free alcohol was used in the first Sonogashira reaction. Compound 12, bearing only a hydrogen at the terminal end of the alkyne, was also synthesized by a slightly modified route (Scheme 2), since working with a terminally unsubstituted alkyne (i.e., without a phenyl ring) would likely lead to highly reactive and volatile intermediates, making the original route challenging. Here, TEMPO/TCICA was used as the oxidant, since DMP resulted in poor yields.
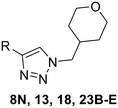
In the first series of compounds (8A–I), we altered the 1-position of the 1,4-substituted triazole with a variety of alkyl, cyclic/heterocyclic, and substituted phenyl moieties. When a group that replaced the nitro group while retaining potency and improving solubility was identified (see Kinetic evaluation below), that substituent was retained in the second series of compounds (8J–O, 12) to allow kinetic analysis without solubility as a barrier. 8J–O, and 12 probe the stereo electronic effects on the terminal phenyl ring attached to the alkyne warhead. Based on the results from the first two series (see Kinetic evaluation below), the elements that increased potency, solubility, and replaced nitro groups were combined to form compounds 8P–R.
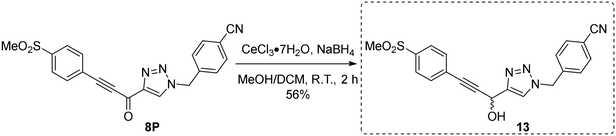
Next, we made a series of derivatives (13, 18, 23A–E) to probe and attempt to improve the stability (i.e., lower intrinsic reactivity) of the alkynyl warhead while retaining sufficient reactivity and binding affinity to inhibit hTG2. Compound 13, synthesized via a Luche reduction (Scheme 3), lowers the electrophilicity of the warhead by removing electron withdrawing affects from the adjacent carbonyl.
We also synthesized propiolamide derivatives 16, 18, 23A–E (Schemes 4 and 5), as the electron donation of the amine to the carbonyl is known to attenuate the intrinsic reactivity of α,β-unsaturated carbonyls.29 Some additional modifications were also made to the terminal substituent of the alkyne within the propiolamide series, to probe the effects of sterics and electronics, at the end of the alkyne, on intrinsic reactivity (see Intrinsic reactivity below).
Compound 16 was synthesized in similar fashion to a previously described protocol,24 and compound 18 was obtained by an amide coupling of intermediate 15 with propargyl amine, followed by a copper-catalysed click reaction.
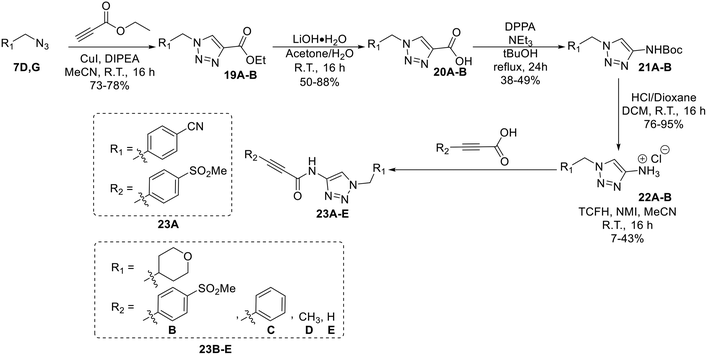
Synthesis of compounds 23A–E features a click reaction with ethyl propiolate and subsequent hydrolysis to produce intermediate 20. This intermediate was then subjected to a Curtius rearrangement and deprotection to install a free amine at the 4-position of the triazole (22). This intermediate was then coupled with various synthesized or commercially available propiolic acids using TCFH/NMI, which is known to facilitate challenging amide couplings with electron deficient amines.30
Given the stability that a terminal methyl group is known to provide to alkynone warheads (see Intrinsic reactivity), attempts were made to synthesize an analogue with a methylene unit between the alkynone and the phenyl group, to disrupt any conjugation that may contribute to increased electrophilicity of the warhead. However, attempts to synthesize this type of compound yielded rearrangement to an allene in mildly basic aqueous conditions (Scheme 6, data not shown), so this was not pursued further.
We also found it peculiar that hTG2 could not tolerate steric bulk around the warhead of a scaffold like EB-2-16, but apparently could do so for 8A, calling into question whether different small molecule scaffolds bind at all similarly to hTG2. No crystal structures are known of hTG2 in complex with a non-peptidic small molecule inhibitor; however, a structure obtained with Ac-P(DON)LPF-NH2 (PDB: 2Q3Z) is widely used for structure-based design,6 which features three distinct pockets: an A-site, the active site tunnel, and a hydrophobic D-site. This crystallographic structure features a distinct active site ‘tunnel’, in which it appears the warhead must fit to access Cys277. This was used to rationalize the need for ‘terminal’ warheads in EB-2-16.17 However, in another structure of hTG2 (PDB: 3S3S), obtained with an analogue of ZED013,31 which does feature a small but internal warhead, the upper wall of this tunnel is absent in the crystal structure, suggesting that it is dynamic in nature. Perhaps surpassing a certain threshold of steric bulk forces the tunnel open, to accommodate a larger warhead. Interestingly, molecular docking of these inhibitors in the 2Q3Zvs.3S3S structures produced significantly different poses. While EB-2-16 could be modelled in 2Q3Z (Fig. 4A), 8A could not, as none of the resulting poses placed the warhead in the tunnel near Cys277 (data not shown). Docking of 8A in 3S3S, however, resulted in a plausible binding mode shown in Fig. 4B. Given its more rigid and linear structure, it is unlikely that 8A could both occupy the D-site and wrap around the adjacent groove to access Cys277. However, binding in the A-site would allow the warhead direct access to Cys277 without compromising its extended conformation.
 | ||
| Fig. 4 Molecular modelling of (A) EB-2-16 in crystal structure PDB: 2Q3Z and (B) 8A in crystal structure PDB: 3S3S. Tunnel, D-site, and A-site are shown by arrows (see Materials and methods). | ||
To investigate these differences experimentally, we designed a series of ‘hybrid’ compounds (28, 33, 36, 38, 40) that combined elements of EB-2-16 with elements of 8A. If these two inhibitors bind similarly to TG2, some degree of cooperativity between their structural features would be expected, whereas the absence of cooperativity would suggest they adopt different binding poses.
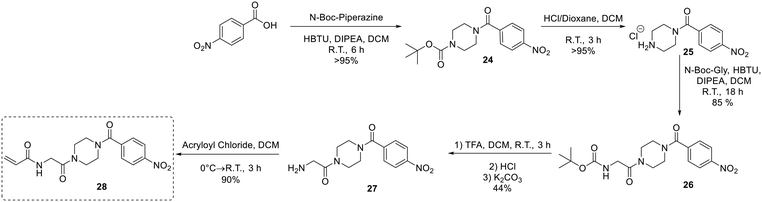
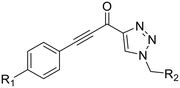
An analogue of EB-2-16 bearing a para-nitrophenyl group in the place of the adamantane group (28) was synthesized using our previously established protocol (Scheme 7),20 beginning with para-nitrobenzoic acid.
Next, an analogue replacing the piperazine group of EB-2-16 with a triazole was synthesized (33). This was achieved by accessing an alkynone derivative of Boc-glycine (30) via a Weinreb amide and subsequent Grignard reaction with ethynylmagnesium bromide. 30 was then clicked to 1-(azidomethyl)adamantane (7C), followed by deprotection of the amine and coupling to acryloyl chloride to obtain inhibitor 33 (Scheme 8).
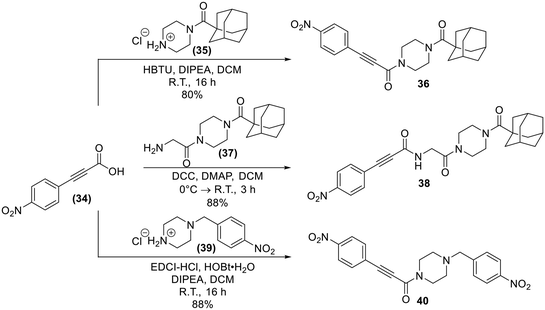
Lastly, analogues that combined the warhead of 8A with the scaffold of EB-2-16, with and without the glycine spacer were synthesized (36 and 38), as absence of the linker more closely resembles the size of 8A, but including the glycine linker is more consistent with the original EB-2-16 scaffold. We also included a compound with a para-nitrobenzyl replacing the adamantane carbonyl moiety (40), to resemble the rotatable bond and overall compound symmetry found in 8A. These compounds were obtained through amide couplings of intermediates we have previously synthesized (Scheme 9).20
Structure–activity relationships
All compounds were tested as described above for 8A. Whenever possible, individual kinact and KI parameters were determined using nonlinear regression. However, in cases where inhibitors showed poor solubility and/or poor inhibition, such that they could not be tested at concentrations high enough to produce a saturation plot, double reciprocal fitting was used to estimate these parameters. However, it should be noted that this double reciprocal approach often produces high errors in the fitting of individual rate and inhibition constants.25 In these cases, the ratio of kinact/KI is taken to be the more reliable measure of potency. All results were interpreted relative to benchmark values for 8A (kinact = 0.90 ± 0.09 min−1, KI = 1.80 ± 0.24 μM, kinact/KI = 499 ± 0.85 × 103 M−1 min−1) and EB-2-16 (kinact = 2.64 ± 0.39 min−1, KI = 3.98 ± 0.79 μM, kinact/KI = 662 ± 164 × 103 M−1 min−1).201-substituted triazole variants
We first started by altering the substituent at the 1-position of the triazole ring, with the goal of replacing the nitro group on the phenyl rings and increasing solubility, while retaining potency. We kept any substituents on the phenyl ring in the para position, as it was previously observed in CP4d and 8A derivatives that ortho and meta substituents led to a decrease in potency.19,23In general, we found that a variety of groups ranging from simple alkyl chains to more complex aromatic and aliphatic heterocycles could be tolerated at this position, while maintaining overall inhibitor efficiencies of kinact/KI ∼ 300–500 × 103 M−1 min−1 (Table 1). KI values in this series of compounds were around 1–2 μM and kinact ranged from about 0.5–1 min−1, diverging very little from parent compound 8A. However, it should be noted that moieties with a hydrogen bond acceptor showed slightly increased inhibition efficiency.
| Inh. | R | k inact (min−1) | K I (μM) | k inact/KI (×103 M−1 min−1) |
|---|---|---|---|---|
| a Determined by double reciprocal fitting. b No inhibition detected up to solubility limit of compound (1 μM). | ||||
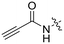
| 8A |
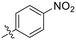
|
0.90 ± 0.09 | 1.80 ± 0.24 | 499 ± 85 |
| 8B |
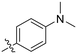
|
0.54 ± 0.38 | 1.03 ± 0.74 | 536 ± 27 |
| 8C |
|
n.d. | n.d. | n.d. |
| 8D |
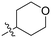
|
1.24 ± 0.19 | 2.30 ± 0.56 | 538 ± 156 |
| 8E |
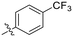
|
0.62 ± 0.29 | 1.75 ± 0.39 | 353 ± 136 |
| 8F |
|
0.89 ± 0.12 | 2.43 ± 0.33 | 368 ± 3 |
| 8G |
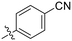
|
0.77 ± 0.07 | 0.96 ± 0.13 | 805 ± 128 |
| 8H |
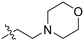
|
1.21 ± 0.14 | 2.84 ± 0.46 | 425 ± 84 |
| 8I |
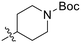
|
0.20 ± 0.02 | 0.40 ± 0.03 | 511 ± 15 |
The most efficient compounds from this series were the tetrahydropyranyl derivative (8D) and the para-cyanobenzyl derivative (8G). The latter showed roughly 2-fold increase in potency compared to parent compound 8A, owing largely to an increase in binding affinity (decrease in KI), suggesting that the presence of a cyano group as a hydrogen bond acceptor in this position was superior to a nitro (8A), amine (8B), ether (8D/8H), or ester (8I). The least efficient compound in this series was the para-trifluoromethyl derivative (8E). Fluorine is known to have variable effects on binding affinity with hTG2 depending on the scaffold. In the case of NM72 (Fig. 1) it provided a notable increase in binding affinity for this peptidomimetic scaffold.15 However, in the EB-2-16 scaffold (Fig. 1), the addition of a fluorine, particularly near the warhead, resulted in a significant loss of binding affinity.20 Here, it seems to not drastically reduce affinity but is not particularly desirable to include either. Note that 8C is not necessarily a poor inhibitor, but it displayed very poor solubility and could not be sufficiently tested. However, it was synthesized to serve as an interesting comparison to the ‘hybrid’ compound series discussed below.
Although 8G displayed the highest overall efficiency, for the majority of the subsequent series of compounds we chose to move forward with a tetrahydropyranyl group (8D) at the 4-position as it provided higher solubility and allowed us to test compounds at higher concentrations.
Warhead variants
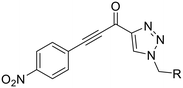
Next, we probed the effects of modifications to the terminal alkynyl substituent of the warhead on reactivity and binding affinity with hTG2 (Table 2). Surprisingly, completely removing the phenyl ring (12) or removing just its para substituent (8J) resulted in equally poor binding affinity. While both modifications also reduced reactivity kinact, retaining the phenyl ring resulted in 3-fold higher reactivity than having no phenyl ring at all. This suggests that the phenyl ring is not providing potency by way of binding affinity, but its conjugation to the alkyne is increasing the reactivity of the warhead. This is corroborated by the intrinsic reactivity of the warheads with glutathione (see Intrinsic reactivity).| Inh. | R/Structure | k inact (min−1) | K I or Ki (μM) | k inact/KI (×103 M−1 min−1) |
|---|---|---|---|---|
| a Determined by double reciprocal fitting. b Reversible inhibitor. c No inhibition detected up to solubility limit of compound (100 μM). | ||||
| 12 | H | 0.12 ± 0.05 | 19.4 ± 9.1 | 6.30 ± 1.00 |
| 8J |
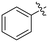
|
0.36 ± 0.02 | 23.9 ± 4.1 | 15.0 ± 3.0 |
| 8K |
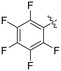
|
0.25 ± 0.02 | 7.30 ± 0.80 | 33.8 ± 4.3 |
| 8L |
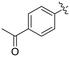
|
0.36 ± 0.05 | 1.36 ± 0.36 | 267 ± 81 |
| 8M |
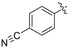
|
1.04 ± 0.18 | 2.99 ± 0.77 | 349 ± 109 |
| 8N |
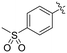
|
0.98 ± 0.14 | 2.05 ± 0.48 | 479 ± 131 |
| 8O |
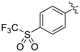
|
0.90 ± 0.13 | 3.19 ± 0.68 | 282 ± 74 |
| 13 |
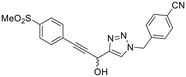
|
0.06 ± 0.01 | 18.9 ± 2.8 | 3.33 ± 0.54 |
| 16 |
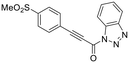
|
— | 0.19 ± 0.02 | — |
| 18 |
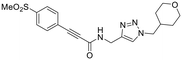
|
n.d. | n.d. | n.d. |
| 23A |
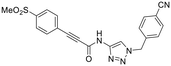
|
0.59 ± 0.13 | 11.4 ± 2.9 | 52.0 ± 2.0 |
| 23B |
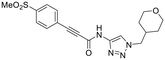
|
0.48 ± 0.06 | 21.5 ± 4.3 | 22.0 ± 4.0 |
Preliminary results previously showed that electron donating groups on this portion of the scaffold resulted in poor potency.23 Thus, we opted to keep electron withdrawing groups on the phenyl ring and investigate how the strength of the electron withdrawing group affects reactivity. Starting with a perfluorobenzene (8K), it seems that inductively withdrawing groups do not increase kinact at all, and fluorine at this position significantly decreases binding affinity, similar to what was observed with an α-fluoroacrylamide warhead on the EB-2-16 scaffold.20 Interestingly, while introducing and increasing the strength of a conjugated electron withdrawing group does increase kinact, this effect is not as strong as expected (e.g., 8Lvs.8M), and kinact seems to plateau around a value of 1 min−1, regardless of electron withdrawing group strength beyond a cyano group. However, again it seems that the presence of an electron withdrawing group containing a hydrogen bond acceptor, specifically one that contains a double-bonded oxygen, is required for optimal binding affinity (e.g., 8L and 8N).
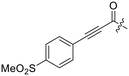
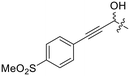
Previously we showed that a propiolamide warhead on the EB-2-16 scaffold had a kinact value of 6.83 ± 1.75 min−1.20 Compounds 8A–O are not propiolamides, as their keto-alkyne moiety is directly attached to the triazole ring, so there is very little attenuation of electrophilicity due to resonance from this side of the molecule. What is peculiar here, is that despite the high intrinsic reactivity of these compounds (see Intrinsic reactivity), their kinact values are relatively low. Given the generally higher binding affinity that this scaffold shows compared to EB-2-16, it seems that it is able to bind tightly to hTG2, but this bound conformation does not optimally position the warhead relative to Cys277 for reaction, resulting in lower kinact values. This could be due to improper positioning of these sterically hindered warheads in the active site tunnel, but it could also suggest a binding mode entirely different from the one proposed in (Fig. 4), perhaps accessing a pocket that has not yet been discovered crytallographically, since all crystal structures have been obtained with peptidomimetic inhibitors. The best inhibitor of this series was the methylsulfone derivative 8N, which allowed us to retain excellent inhibitor efficiency while replacing the second nitro group.
We also evaluated a variety of compounds with attenuated warhead reactivity. Inhibitor 13 features an internal propargyl alcohol rather than a keto-alkyne, in which the alkyne is much less activated (less electron deficient). Surprisingly, this inhibitor did still display time-dependent irreversible inhibition with hTG2, albeit with a very low kinact value of 0.06 ± 0.01 min−1 and lower binding affinity. Next, we investigated a variety of propiolamide derivatives, starting with a compound (16) inspired from a benzotriazole-cinnamoyl adduct (CP15n), from which CP4d was first discovered.19 Interestingly, this compound was a very potent but reversible inhibitor with a Ki of 190 nM (see SI). This suggests that having the triazole group and phenyl groups closer to the warhead in a fused ring structure impacted binding affinity very positively, but disrupted the positioning of the warhead such that it could no longer react with Cys277 at all. Strangely, it seems that minor structural changes can have significant impact on the exact binding site/pose of these compounds, without perturbing the overall binding affinity.
Other, more classic propiolamide derivatives 18 and 23A–B showed substantially reduced potency, largely due to a decrease in binding affinity, suggesting that more distance between the triazole and warhead leads to poorer binding. With the 2-atom spacer between the triazole and the warhead in compound 18, no significant inhibition was detected. However, with the 1-atom spacer in 23A–B, appreciable binding affinity had returned. The only difference between 23A and 23B is the substituent at the 1-position of the triazole. The para-cyanobenzyl derivative showed roughly 2-fold higher potency than the tetrahydropyranyl derivative. This is consistent the previous findings with compounds 8D and 8G (Table 1), confirming that there were no unusual cooperativity effects taking place when altering substituents at the 1 and 4-positions of the triazole together.
EB-2-16 and 8A hybrids
Compounds bearing elements of both the EB-2-16 and 8A scaffold were evaluated to determine if any cooperativity existed between their binding modes (Table 3). Inhibitor 28 features a para-nitrophenyl group in the place of the adamantane of EB-2-16. This derivative showed ∼50-fold reduction in binding affinity compared to EB-2-16. This suggests that the substituent at the 1-position of the triazole in 8A is not similar in its binding pose/interactions as the hydrophobic unit of EB-2-16.| Inh. | Structure | k inact (min−1) | K I (μM) | k inact/KI (×103 M−1 min−1) |
|---|---|---|---|---|
| a Determined by double reciprocal fitting. b No inhibition detected up to solubility limit of compound (50 μM). | ||||
| 28 |
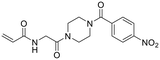
|
2.17 ± 0.45 | 181 ± 54 | 12.0 ± 4.3 |
| 33 |
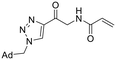
|
2.24 ± 0.56 | 16.9 ± 5.5 | 133 ± 54 |
| 36 |
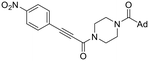
|
0.40 ± 0.22 | 17.7 ± 12.5 | 22.7 ± 3.8 |
| 38 |
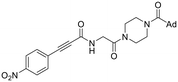
|
n.d. | n.d. | n.d. |
| 40 |
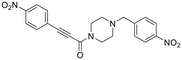
|
n.d. | n.d. | n.d. |
Inhibitor 33 contains a triazole ‘spacer’ unit in between the hydrophobic moiety and glycine linker/warhead component, instead of a piperazine. It should be noted, however, that the hydrophobic moiety does not contain a carbonyl linkage to the triazole like it does to piperazine in the parent compound. This is due to the fact that a click reaction with an acyl azide precursor would be needed to retain the carbonyl, and because this is synthetically impractical, we carried forward with a methylene linkage instead. Inhibitor 33 showed ∼4-fold decrease in binding affinity, but it was still a relatively efficient inhibitor. It is unclear whether the reduction in binding affinity came solely from replacing the triazole with the piperazine and/or removing the carbonyl linkage. However, given the fact that compound 40, in which the triazole of 8A was replaced with a piperazine, showed no significant inhibition at all, there seems to be little cooperativity between these ‘spacer’ units either.
Compounds 36 and 38 contain the warhead of 8A, but with the rest of the EB-2-16 scaffold, without and with the glycine linker respectively. Again, these compounds display significantly reduced potency compared to the parent scaffolds. However, it is interesting to note that the smaller size/length of 36, which is more similar to the size of 8A, shows greater potency than 38.
Taken together, these hybrid compounds reveal that the binding pose of these two small molecule scaffolds is not likely to be similar and that their optimization will require significantly different considerations when considering the types of interactions different structural elements may be engaged in. This is notably very different from traditional peptidic inhibitors that contain very similar structural features and binding modes.
Optimization of inhibitor potency
The results of the aforementioned SAR analyses suggested that the nitro group on the terminal phenyl group of the warhead could be replaced with a methylsulfone and that a para-cyanobenzyl group at the 1-position of the triazole increased binding affinity. Therefore, we combined these elements together to give inhibitor 8P. The synergy of these two changes pushed the inactivation constant (KI) down into the nanomolar range, while retaining a modest kinact (Table 4). We also synthesized and evaluated a derivative with a dimethylsulfonamide instead of a methylsulfone (8Q); however, this compound showed relatively poor solubility in our kinetic assay, and its overall inhibition efficiency was also lower. To achieve solubilizing effects similar to those of the tetrahydropyranyl moiety of 8D, we replaced the simple benzene ring with a pyridine in inhibitor 8R. This compound displayed the highest inhibitor efficiency among all compounds (kinact = 0.43 ± 0.02 min−1, KI = 0.48 ± 0.05 μM, kinact/KI = 910 ± 113 × 103 M−1 min−1), representing one of the most potent non-peptidic small molecule hTG2 inhibitors to date.Repeating molecular modelling with the most potent inhibitor 8R revealed some interesting interactions that may contribute to increased potency (and general affinity of this scaffold). In the binding pose shown in Fig. 5, the sulfonylphenyl group is engaged in pi-pi stacking with Trp332 and hydrogen bonding with Thr360, the triazole forms a hydrogen bond with Asn333, and the cyanopyridine group is positioned to engage in a pi-pi stacking interaction with Phe334 and a hydrogen bond with Gln169 via the nitrile group. The increased affinity observed with 8P and 8R compared to 8A may be due to the better positioning of the nitrile group, relative to the nitro group, to act as a hydrogen bond acceptor.
 | ||
| Fig. 5 Molecular modelling of 8R in crystal structure PDB: 3S3S. (A) Molecular model with key ligand-protein interactions shown by cyan dotted lines. (B) Ligand interaction map (see Materials and methods). | ||
Isozyme selectivity
Since hTG2 is part of a family of TGases it is always important to establish the selectivity of new scaffolds against a panel of other, therapeutically relevant TGases, namely TG1, TG6, TG3a, and FXIIIa. The AL5 assay described above for hTG2 inhibition was also used to monitor hTG1 and hTG6 inhibition,16 whereas for hTG3a and hFXIIIa, a commercially available continuous fluorescence assay that uses a peptidic FRET-quenched substrate was used.32,33 In each case the same effective inhibitor concentration ([I]/α, where α = (1 + ([S]/KM)), to account for competition with substrate) was used for each enzyme/substrate pair in the assays, in order to permit comparison between isozymes. A single kobs value was determined, in triplicate, for each isozyme with inhibitor 8R (see SI).16 As shown in Fig. 6 and the SI (page S12), 8R does not show significant inhibition of any of the other isozymes. In fact, this scaffold shows better selectivity than EB-2-16,20 and our other previously reported peptidomimetic scaffolds,14 making 8R one of the most selective non-peptidic hTG2 inhibitor scaffolds reported to date.Inhibition of GTP binding
One of the key advantages of covalent modification of Cys277 of hTG2 is that it locks hTG2 in its open conformation and thereby abolishes its ability to bind GTP. We have recently shown that molecules as small as acrylamide itself completely lock hTG2 in its open conformation, even if they have poor binding affinity.34 Therefore, it appears that any compound covalently modifying Cys277 will abolish GTP binding. Compound 8R was evaluated for its impact on GTP binding using our previously reported assay.34 The enzyme was incubated with and without inhibitor and then subjected to dialysis to wash out any unbound inhibitor. Both samples were then mixed with a nonhydrolyzable GTP analogue (GTPγS FL BODIPY) whose fluorescence increases when bound to hTG2. As shown in Fig. 7, enzyme incubated with inhibitor showed negligible GTP binding compared to the uninhibited control. This provides further validation that the alkynyl scaffold is in fact irreversible and likely reacting with Cys277.Intrinsic reactivity of alkynyl warheads
Although none of the warheads with attenuated reactivity were able to react/bind as efficiently with hTG2 as 8A, this type of internal warhead is underexplored in the field of TCIs. Therefore, we further investigated the intrinsic reactivity of different alkynyl warhead analogues by measuring the corrected second order rate constants (kcorr2) of their reaction with glutathione (GSH), in solution, under pseudo-first order conditions, at room temperature (Table 5). Reactions were carried out at pH 7.4 (phosphate buffer) or pH 10.4 (CAPS buffer) depending on how reactive the compound was and if rate acceleration was needed by increasing the pH to observe the reaction over a reasonable time frame. Corrected second order rate constants (i.e., corrected for pH and accounting for [GS−] in solution) provide a much more robust picture of reactivity than half-lives or pseudo-first order rate constants alone, as they are not dependent on assay conditions and allow direct comparison between literature values.35We started with compound 8N as a representative of the potent alkynyl hTG2 inhibition scaffold reported herein. Unfortunately, this compound showed rapid reaction with GSH and was highly unstable (t1/2 ≤ 10 min at pH 7.4). This is likely due to the fact there are two strong electron withdrawing groups attached directly to the alkyne (para-methylsulfone phenyl and ketone) making it a highly activated electrophile. Compound 16, a potent reversible inhibitor, also showed rapid consumption and instability in this assay. Due to its poor solubility, its reactivity with GSH was measured by 1H NMR in DMSO-d6 (see SI, page S13). Even in non-aqueous conditions, its half-life was under 10 minutes. With this compound, it is plausible that along with the alkyne, the amide bond could be cleaved by glutathione (or in solution during kinetic assays) due to the fact that benzotriazole is a good leaving group. For this reason, we also tested benzotriazole itself and intermediate 15 (Scheme 4) in our inhibition assay to see if inhibition actually resulted from one of these individual precursors rather than the compound itself due to hydrolysis. However, none of these precursors showed any inhibition of hTG2 on their own (see SI, page S9), suggesting that the inhibition observed in our assay was not due to degradation products. The evaluation of 8N and 16 provided half-lives that were less than 10 min, allowing us to set a limit of their kcorr2 values as being >100 M−1 min−1 (see Materials and methods for related equations).
The alkyne in compound 13 is not conjugated to a carbonyl, which explains why it is the least intrinsically reactive compound in this series with a kcorr2 of 0.12 ± 0.01 M−1 min−1. However, the conjugated methylsulfonylbenzene ring still provides significant activation, such that this compound is only about 4–8 fold less reactive than a standard N-alkyl acrylamide warhead, which typically have kcorr2 values of ∼0.4–0.8 M−1 min−1 at room temperature,20,29,36 suggesting that it is still a viable warhead, especially if it offers greater binding affinity with specific targets.
We also tested a variety of propiolamide derivatives with different terminal alkyne substituents. Compound 18, featuring an N-alkyl amide linkage showed a kcorr2 = 4.85 ± 0.19 M−1 min−1, whereas an analogue with an N-triazole amide linkage (23B) has a kcorr2 = 36.7 ± 1.0 M−1 min−1. This is likely because the electron donating capability of the nitrogen is stronger in the N-alkyl linkage, whereas the N-triazole linkage splits the electron donation through resonance between the adjacent carbonyl and the triazole, making the warhead overall about 10-fold more reactive. This is very similar to the difference in reactivity between N-alkyl and N-phenyl acrylamides.35,36
Next, we explored the effect of the methylsulfone group on the terminal phenyl ring. Removing this electron withdrawing group in compound 23C decreased the kcorr2 ∼ 4-fold to 11.2 ± 0.08 M−1 min−1 compared to 23B, revealing the significant contribution of this electron withdrawing group to warhead reactivity. Removing the phenyl group and replacing it with a methyl group (23D) resulted in a significant increase in stability, with a kcorr2 = 0.52 ± 0.08 M−1 min−1. This is an interesting result, as it seems that the steric hindrance conferred by the methyl group far outweighs any steric hindrance effects imparted by the phenyl group, and that the electronic effects of the phenyl group dominate. It is likely that, when directly attached to the alkyne, the planar phenyl ring is orthogonal to the anti-bonding pi-orbitals of the α,β-unsaturated carbonyl system. However, a tetrahedral methyl group is free to rotate and partially block attack from the incoming nucleophile. The stabilizing effects of the methyl group are also seen in compound 23E, where replacing the methyl group with a hydrogen (i.e., effectively removing the terminal substituent) resulted in significant loss of stability (∼140-fold) with a kcorr2 = 69.5 ± 5.81 M−1 min−1.
The kinact/KI ratio represents the second order rate constant for reaction between an inhibitor and enzyme, taking into account both binding affinity and reactivity. On the other hand, kcorr2 represents the second order rate constant for the reaction of inhibitor with glutathione thiolate (GS−). We divided (kinact/KI) by kcorr2 for every inhibitor for which these values were measured, to provide an ‘enzymatic efficiency ratio’ (EER), as we have previously described.20 This ratio gives information about how well the enzyme is able to facilitate thiol addition to the inhibitor, compared to GSH in solution. A higher ratio indicates higher expected on-target reactivity and selectivity. Interestingly, compound 13, which is a less efficient inhibitor than 23B, shows an EER of ∼28![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 (Table 5), roughly 50-fold higher than the EER obtained with 23B (∼600), meaning the enzyme is much more selective in facilitating the reaction with the intrinsically low reactivity warhead, likely due to binding affinity and/or conformational effects. This is generally true and consistent with what we found in our previous warhead screening study on EB-2-16.20 However, for good lead candidates this ratio should be on the order of millions,20 which is far above the EER values of our current series.
000 (Table 5), roughly 50-fold higher than the EER obtained with 23B (∼600), meaning the enzyme is much more selective in facilitating the reaction with the intrinsically low reactivity warhead, likely due to binding affinity and/or conformational effects. This is generally true and consistent with what we found in our previous warhead screening study on EB-2-16.20 However, for good lead candidates this ratio should be on the order of millions,20 which is far above the EER values of our current series.
These results point to some routes that may be explored to improve the stability and efficiency of these inhibitors. While it seems that propiolamide derivatives show significant decrease in binding affinity, and that the pharmacophore is very sensitive to changes at the 4-position of the triazole, attempts can be made to further improve the binding affinity of analogues like 13 and 23A/B, as they show modest inhibition efficiency and stability. Additionally, although attempts to synthesize a propargyl derivative of our scaffold (Scheme 6) by introducing a methylene unit between the alkyne and the phenyl ring were unsuccessful, it is likely that this type of modification, in principle, will increase stability similarly to a terminal methyl group. However, this type of modification will likely require more than one methylene unit to prevent allene formation, as well as more complex synthetic routes.
Conclusions
In this study, we revisited our previously reported reversible hTG2 inhibitor scaffold CP4d and discovered a unique and highly potent internal alkynyl scaffold (8A) that irreversibly inhibits hTG2. Comprehensive kinetic SAR analysis revealed how modifications to the triazole 1-substituent, warhead reactivity, and incorporation of elements from the EB-2-16 scaffold affect both binding affinity (KI) and reactivity (kinact). These insights guided the design of inhibitor 8R, in which potentially toxic nitrophenyl groups were replaced with methylsulfonyl-phenyl and cyano-pyridinyl substituents, while improving solubility and inhibitor efficiency substantially. With a kinact of 0.43 ± 0.02 min−1, a KI of 0.48 ± 0.05 μM, and a kinact/KI of 910 ± 113 × 103 M−1 min−1, 8R ranks among the most effective non-peptidic hTG2 inhibitors reported to date. It also demonstrates excellent isozyme selectivity and fully abolishes GTP binding.Hybrid derivatives combining the scaffolds of 8A and EB-2-16 showed no cooperative inhibition, suggesting distinct binding poses and underscoring the need for structural elucidation of non-peptidic hTG2 inhibitors via crystallography or cryo-EM to enhance rational structure-based design. Investigation into the intrinsic reactivity of various internal alkynyl warheads highlighted the stabilizing effect of terminal methyl groups and the significant influence of extended conjugation and electron-withdrawing groups on reactivity. Although the most potent inhibitors have limited stability, promising strategies such as propiolamide and propargyl derivatives have been identified for future optimization.
Overall, this work establishes a novel internal alkynyl warhead scaffold for irreversible hTG2 inhibition that not only provides a valuable chemical biology tool but also expands the scope of covalent warheads beyond traditional terminal electrophiles, with potential applications extending beyond hTG2.
Materials and methods
Synthesis
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 536 points. An automatic phase correction was applied and manually refined to provide a flatter baseline. A baseline correction, employing a Whittaker smoothing function with a 2.60 Hz filter and a smoothing factor of 16
536 points. An automatic phase correction was applied and manually refined to provide a flatter baseline. A baseline correction, employing a Whittaker smoothing function with a 2.60 Hz filter and a smoothing factor of 16![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 384 was employed on the spectrum between −2.00 and 15.00 ppm. Integration was measured using the manual integration tool and referenced to a relevant proton. Multiplet analysis was conducted using the algorithm as implemented in the software, confirmed by manual analysis. High resolution mass spectrometry (HRMS) was performed using a quadrupole time-of-flight (QTOF) or magnetic sector analyzer and electrospray ionization (ESI) or electron impact (EI), respectively. All measured m/z values were within 3 mmu of the calculated value (generally < 10 ppm discrepancy) unless otherwise noted. High performance liquid chromatography (HPLC) purity traces were collected on a Gilson-Mandel GXP271 with UV detection at 214 and 254 nm (Phenomenex Luna, 150 mm × 4.6 mm, 30 min, 1.5 mL min−1 flow rate, 5–95% or 20–80%, CH3CN with 0.1% TFA in H2O with 0.1% TFA, 30 min method).
384 was employed on the spectrum between −2.00 and 15.00 ppm. Integration was measured using the manual integration tool and referenced to a relevant proton. Multiplet analysis was conducted using the algorithm as implemented in the software, confirmed by manual analysis. High resolution mass spectrometry (HRMS) was performed using a quadrupole time-of-flight (QTOF) or magnetic sector analyzer and electrospray ionization (ESI) or electron impact (EI), respectively. All measured m/z values were within 3 mmu of the calculated value (generally < 10 ppm discrepancy) unless otherwise noted. High performance liquid chromatography (HPLC) purity traces were collected on a Gilson-Mandel GXP271 with UV detection at 214 and 254 nm (Phenomenex Luna, 150 mm × 4.6 mm, 30 min, 1.5 mL min−1 flow rate, 5–95% or 20–80%, CH3CN with 0.1% TFA in H2O with 0.1% TFA, 30 min method).
1-Iodo-4-(methylsulfonyl)benzene (1F)
To a solution of 4-(methylsulfonyl)aniline (2.00 g, 11.68 mmol, 1.0 eq.) in H2O (10 mL) and conc. HCl (2.5 mL) at 0 °C was added a solution of NaNO2 (0.88 g, 12.84 mmol, 1.1 eq.) in H2O (2 mL) dropwise. The reaction was stirred for 1 h and then a solution of KI (2.12 g, 12.84 mmol, 1.2 eq.) in H2O (2 mL) was added dropwise at 0 °C. The reaction was allowed to warm to R.T. and then refluxed for 1 h. The reaction mixture was then cooled to 0 °C prior to addition of sat. NaHSO3 (100 mL). The aqueous solution was extracted with Et2O (3 × 50 mL) and then washed with brine (100 mL). The organic layer was then dried over MgSO4 and concentrated in vacuo to give a yellow solid which was purified by flash column chromatography (30% EtOAc/Hex Rf = 0.47) to give a white powder (1.05 g, 35%). 1H NMR (500 MHz, CDCl3) δ 7.99 (d, J = 8.6 Hz, 2H), 7.70 (d, = 8.6 Hz, 2H), 3.09 (s, 3H). 13C NMR (125 MHz, CDCl3) δ 140.4, 138.8, 128.9, 101.7, 44.6. Characterization data are consistent with previously reported values.371-Bromo-4-((trifluoromethyl)sulfonyl)benzene (1G)
To a solution of 4-bromophenyl trifluoromethyl sulfide (0.30 mL, 1.94 mmol, 1.0 eq.) in DCM (20 mL) at 0 °C was added mCPBA (1.67 g, 9.70 mmol, 5.0 eq.) portion wise. The reaction mixture was allowed to warm to R.T while stirring overnight. The reaction mixture was then diluted with DCM (100 mL) and washed with sat. NaHCO3 (2 × 100 mL) and then brine (100 mL) followed by drying over MgSO4 and concentrating in vacuo. The residue was purified by flash column chromatography (5% EtOAc/Hex, Rf = 0.45, dry loading using celite) to give a white solid (0.50 g, 89%). 1H NMR (300 MHz, CDCl3) δ 7.90 (d, J = 9.0 Hz, 2H), 7.83 (d, J = 9.0 Hz, 2H). 13C NMR (75 MHz, CDCl3) δ 119.8 (q, JC,F = 324.3 Hz), 130.4, 132.3, 133.0, 133.4. 19F NMR (282 MHz, CDCl3) δ –78.25. Characterization data are consistent with previously reported values.382-(Prop-2-yn-1-yloxy)tetrahydro-2H-pyran (2B)
To a solution of propargyl alcohol (1.10 mL, 17.84 mmol, 1.0 eq.) in DCM (15 mL) were added dihydropyran (2.50 mL, 26.76 mmol, 1.5 eq.) and 4 drops of 12.5 M HCl. The reaction mixture was stirred vigorously and R.T. for 24 h, after which it was quenched with sat. NaHCO3 (50 mL). The organic layer was separated, and the aqueous layer was further extracted using DCM (3 × 50 mL). The combined organic layer was washed with brine (150 mL) and dried over MgSO4 before being concentrated in vacuo to give a colourless oil (2.14 g, 86%) 1H NMR (300 MHz, CDCl3) δ 4.83 (t, J = 3.0 Hz, 1H), 4.27 (dd, J = 15.6, 2.4 Hz, 2H), 3.90–3.74 (m, 1H), 3.60–3.47 (m, 1H), 2.42 (t, J = 2.4 Hz, 1H), 1.92–1.44 (m, 6H, overlapping with H2O). 13C NMR (75 MHz, CDCl3) δ 96.8, 79.7, 73.9, 61.9, 53.9, 30.2, 25.3, 19.0. Characterization data are consistent with previously reported values.39General procedure 1 (GP1): Sonogashira reaction24
In a flame-dried round-bottom flask filled with N2 were added para substituted aryl iodide or aryl bromide (1A–H) (1.0 eq., commercially available unless indicated otherwise), PdCl2(PPh3)2 (2 mol%) and CuI (2 mol%). The solids were dissolved in anhydrous MeCN (∼1 M) followed by the addition of NEt3 (4.0 eq.). The reaction mixture was stirred for 10 min at R.T. followed by dropwise addition of propargyl alcohol (2A) or THP-protected propargyl alcohol (2B) (1.2 eq.). The reaction was heated to reflux and stirred for 2–24 h. The reaction was concentrated in vacuo and then diluted with H2O (100 mL) and EtOAc (100 mL) and filtered through celite. The layers of the filtrate were separated, and the aqueous layer was extracted with EtOAc (2 × 100 mL). The combined organic layer was washed with sat. NH4OH (100 mL) and then water (100 mL) followed by drying over MgSO4 and concentrating in vacuo. The residue was purified by flash column chromatography as described below.3-(4-Nitrophenyl)prop-2-yn-1-ol (3A)
Synthesized according to GP1 using 4-iodonitrobenzene (1A) (1.24 g, 5.00 mmol) and purified by flash column chromatography (50% EtOAc/Hexanes, Rf = 0.61, dry loading using celite) to give an orange solid (0.74 g, 84%). 1H NMR (300 MHz, CDCl3) δ 8.19 (d, J = 8.7 Hz, 2H), 7.58 (d, J = 8.7 Hz, 2H), 4.54 (s, 2H), 1.70 (br s, overlapping with H2O, 1H). 13C NMR (75 MHz, CDCl3) δ 147.3, 132.4, 129.4, 123.6, 92.4, 83.8, 51.5. Characterization data are consistent with previously reported values.243-Phenylprop-2-yn-1-ol (3B)
Synthesized according to GP1 using 4-iodobenzene (1B) (1.00 g, 4.90 mmol) and purified by flash column chromatography (30% EtOAc/Hex, Rf = 0.39, dry loading using celite) to give a yellow solid (0.68 g, >95%). 1H NMR (400 MHz, CDCl3) δ 7.46–7.43 (m, 2H), 7.35–7.31 (m, 3H), 4.52 (s, 2H), 1.72 (br d, J = 2.0 Hz, 1H). 13C NMR (100 MHz, CDCl3) δ 131.7, 128.5, 128.3, 122.5, 87.7, 85.7, 51.6. Characterization data are consistent with previously reported values.232-((3-(Perfluorophenyl)prop-2-yn-1-yl)oxy)tetrahydro-2H-pyran (3C1)
Synthesized according to GP1 using iodopentafluorobenzene (1C) (1.00 mL, 7.50 mmol), 2B (0.87 g, 6.45 mmol), and purified by flash column chromatography (10% EtOAc/Hex, Rf = 0.53, dry loading using celite) to give a colourless oil (1.50 g, 65%). 1H NMR (300 MHz, CDCl3) δ 4.88 (t, J = 3.1 Hz, 1H), 4.54 (s, 2H), 4.01–3.77 (m, 1H), 3.61–3.54 (m, 1H), 1.97–1.41 (m, 6H). 13C NMR (75 MHz, CDCl3) δ 148.4–148.3 (m), 146.8–146.6 (m), 142.6–142.4 (m), 140.9–140.7 (m), 138.5–138.3 (m), 136.9–136.6 (m), 99.7–99.5 (m), 98.2, 97.1, 69.8, 62.1, 54.5, 30.2, 25.3, 18.9. 19F NMR (282 MHz, CDCl3) δ −135.79–−135.92 (m, 2F), −152.17–−152.34 (m, 1F), −161.66–−161.86 (m, 2F). HRMS (EI) calc'd for C14H11F5O2 [M˙]+: 306.0679, found: 306.0702.3-(Perfluorophenyl)prop-2-yn-1-ol (3C2)
To a solution of 3C1 (1.40 g, 4.90 mmol, 1.0 eq.) in MeOH (15 mL) was added pTsOH·H2O (0.10 g, 0.49 mmol, 0.1 eq.). The reaction was stirred for 1 h at R.T. at which point it was confirmed complete by TLC analysis. The reaction was concentrated in vacuo, and the residue was purified by flash column chromatography (10% EtOAc/Hex, Rf = 0.16, dry loading using celite) to give a white crystalline solid (0.92 g, 85%). 1H NMR (300 MHz, CDCl3) δ 4.60 (s, 2H), 1.93 (br s, 1H). 13C NMR (75 MHz, CDCl3) δ 148.4–148.2 (m), 146.7–146.5 (m), 142.7–142.5 (m), 140.9–140.7 (m), 138.5–138.3 (m), 136.9–136.6 (m), 99.8, 99.5–99.2 (m), 69.7, 51.5. 19F NMR (282 MHz, CDCl3) δ −135.90–−136.03 (m, 2F), −151.82–−151.98 (m, 1F), −161.46–−161.66 (m, 2F). HRMS (EI) calc'd for C9H3F5O [M˙]+: 222.0104, found: 222.0091.1-(4-(3-Hydroxyprop-1-yn-1-yl)phenyl)ethan-1-one (3D)
Synthesized according to GP1 using 4-iodoacetophenone (1D) (1.00 g, 4.06 mmol) and purified by flash column chromatography (10% EtOAc/DCM, Rf = 0.43, dry loading using celite) to give a yellow solid (0.65 g, 92%). 1H NMR (300 MHz, CDCl3) δ 7.92 (d, J = 8.5 Hz, 2H), 7.53 (d, J = 8.5 Hz, 2H), 4.54 (s, 2H), 2.61 (s, 3H). 13C NMR (75 MHz, CDCl3) δ 197.3, 136.5, 131.8, 128.2, 127.4, 90.4, 84.9, 51.6, 26.6. Characterization data are consistent with previously reported values.234-(3-Hydroxyprop-1-yn-1-yl)benzonitrile (3E)
Synthesized according to GP1 using 4-iodobenzonitrile (1E) (1.00 g, 4.37 mmol) and purified by flash column chromatography (50% EtOAc/Hex, Rf = 0.63, dry loading using celite) to give a yellow solid (0.60 g, 87%). 1H NMR (400 MHz, CDCl3) δ 7.59 (d, J = 8.4 Hz, 2H), 7.50 (d, J = 8.4 Hz, 2H), 4.51 (s, 2H). 13C NMR (100 MHz, CDCl3) δ 132.1, 132.0, 127.5, 118.3, 111.8, 91.7, 83.9, 51.4. Characterization data are consistent with previously reported values.403-(4-(Methylsulfonyl)phenyl)prop-2-yn-1-ol (3F)
Synthesized according to GP1 using 1F (0.60 g, 2.12 mmol) and purified by flash column chromatography (20% EtOAc/Hex, Rf = 0.37, dry loading using celite) to give an off-white solid (0.35 g, 80%). 1H NMR (300 MHz, CDCl3) δ 7.90 (d, J = 8.6 Hz, 2H), 7.61 (d, J = 8.6 Hz, 2H), 4.53 (s, 2H), 3.06 (s, 3H), 1.76 (br s, 1H). 13C NMR (75 MHz, CDCl3) δ 44.6, 51.5, 83.9, 91.9, 127.5, 128.8, 132.6, 140.0. Characterization data are consistent with previously reported values.413-(4-((Trifluoromethyl)sulfonyl)phenyl)prop-2-yn-1-ol (3G)
Synthesized according to GP1 using 1G (0.50 g, 1.73 mmol) and purified by flash column chromatography (5% EtOAc/DCM, Rf = 0.47, dry loading using celite) to give a brown oil (0.34 g, 73%). 1H NMR (600 MHz, CDCl3) δ 8.00 (d, J = 8.6 Hz, 2H), 7.70 (d, J = 8.6 Hz, 2H), 4.57 (s, 2H), 1.98 (br s, 1H). 13C NMR (150 MHz, CDCl3) δ 132.7, 131.5, 130.7, 130.4, 119.7 (q, JC,F = 324.3 Hz), 93.7, 83.5, 51.5. 19F NMR (282 MHz, CDCl3) δ −78.25. HRMS (EI) calc'd for C10H6F3O3S [M − H]˙+: 262.9990, found: 262.9950 (note: 5 mmu (15 ppm) discrepancy).4-(3-Hydroxyprop-1-yn-1-yl)-N,N-dimethylbenzenesulfonamide (3H)
Synthesized according to GP1 using 4-bromo-N,N-dimethylbenzenesulfonamide (1H) (1.00 g, 3.79 mmol) and purified by flash column chromatography (70% EtOAc/Hex, Rf = 0.41, dry loading using celite) to give a pale-yellow solid (0.77 g, 84%). 1H NMR (600 MHz, CDCl3) δ 7.72 (d, J = 8.6 Hz, 2H), 7.57 (d, J = 8.6 Hz, 2H), 2.71 (s, 6H). 13C NMR (150 MHz, CDCl3) δ 135.3, 132.2, 127.8, 127.5, 91.0, 84.2, 51.7, 38.0. HRMS (EI) calc'd for C11H13NO3S [M˙]+: 239.0616, found: 239.0636.General procedure 2 (GP2): DMP oxidation23
To a solution of alcohol (3A–H and 5A–H) (1.0 eq.) in DCM (∼0.15 M) at 0 °C was added DMP (1.2 eq.) portion wise. The ice bath was then removed, and the reaction was stirred at R.T. for 3 h. The reaction was concentrated in vacuo and diluted with EtOAc (100 mL). Sat. Na2S2O3 and NaHCO3 (150 mL![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 150 mL) were added to the reaction mixture and the solution was stirred vigorously until the precipitate dissolved. The layers were separated, and the aqueous layer was extracted with EtOAc (2 × 100 mL). The organic layer was washed with sat. NaHCO3 (100 mL), brine (200 mL), and then dried over MgSO4. After concentrating in vacuo, the residue was purified by flash column chromatography or carried forward without further purification as described below.
150 mL) were added to the reaction mixture and the solution was stirred vigorously until the precipitate dissolved. The layers were separated, and the aqueous layer was extracted with EtOAc (2 × 100 mL). The organic layer was washed with sat. NaHCO3 (100 mL), brine (200 mL), and then dried over MgSO4. After concentrating in vacuo, the residue was purified by flash column chromatography or carried forward without further purification as described below.
3-(4-Nitrophenyl)propiolaldehyde (4A)
Synthesized according to GP2 using 3A (0.74 g, 4.15 mmol) and purified by flash column chromatography (30% EtOAc/Hexanes, Rf = 0.57, dry loading using celite) to give a yellow solid (0.67 g, 92%). 1H NMR (300 MHz, CDCl3) δ 9.46 (s, 1H), 8.28 (d, J = 9.0 Hz, 2H), 7.78 (d, J = 9.0 Hz, 2H). 13C NMR (75 MHz, CDCl3) δ 176.1, 184.8, 133.9, 126.0, 123.9, 90.8, 90.6. Characterization data are consistent with previously reported values.233-Phenylpropiolaldehyde (4B)
Synthesized according to GP2 using 3B (0.63 g, 4.70 mmol) and the resulting solid was carried forward without further purification (5% EtOAc/DCM, Rf = 0.71) (0.60 g, >95%). 1H NMR (400 MHz, CDCl3) δ 9.44 (s, 1H), 7.64–7.61 (m, 2H), 7.53–7.48 (m, 1H), 7.44–7.40 (m, 2H). 13C NMR (100 MHz, CDCl3) δ 176.8, 133.3, 131.3, 128.7, 119.4, 95.1, 88.4. Characterization data are consistent with previously reported values.233-(Perfluorophenyl)propiolaldehyde (4C)
Synthesized according to modified procedure GP2 using 3C2 (0.30 g, 1.40 mmol). NaHCO3 (0.47 g, 4.60 mmol, 4.0 eq.) was added to the reaction to neutralize the AcOH in the reaction mixture. The resulting white powder (0.28 g, >95%) was carried forward without further purification (10% EtOAc/Hex, Rf = 0.41). 1H NMR (300 MHz, CDCl3) δ 9.41 (s, 1H). 13C NMR (75 MHz, CDCl3) δ 175.2, 149.9–149.7 (m), 146.5–146.3 (m), 145.6–145.2 (m), 142.1–141.7 (m), 139.4–139.2 (m), 136.3–136.0 (m), 97.3–97.7 (m, 2C overlapping), 60.7. 19F NMR (282 MHz, CDCl3) δ −132.68–−132.82 (m, 2F), −146.49–−146.65 (m, 1F), −160.17–−160.41 (m, 2F). HRMS (EI) calc'd for C9HF5O [M˙]+: 219.9948, found: 219.9937.3-(4-Acetylphenyl)propiolaldehyde (4D)
Synthesized according to GP2 using 3D (0.65 g, 3.73 mmol) and purified by flash column chromatography (10% EtOAc/DCM, Rf = 0.67, dry loading using celite) to give a yellow solid (0.60 g, 93%). 1H NMR (300 MHz, CDCl3) δ 9.46 (s, 1H), 7.99 (d, J = 8.7 Hz, 2H), 7.71 (d, J = 8.7 Hz, 2H), 2.64 (s, 3H). 13C NMR (75 MHz, CDCl3) δ 196.9, 176.4, 138.5, 133.3, 128.4, 123.9, 92.9, 89.8, 26.7. Characterization data are consistent with previously reported values.234-(3-Oxoprop-1-yn-1-yl)benzonitrile (4E)
Synthesized according to GP2 using 3E (0.60 g, 3.75 mmol) and the resulting yellow solid (0.59 g, >95%) was carried forward without further purification (50% EtOAc/Hex, Rf = 0.56). 1H NMR (300 MHz, CDCl3) δ 9.43 (s, 1H), 7.71–7.66 (m, 4H). 13C NMR (75 MHz, CDCl3) δ 176.2, 133.5, 132.4, 124.2, 117.8, 114.6, 91.3, 90.3. Characterization data are consistent with previously reported values.423-(4-(Methylsulfonyl)phenyl)propiolaldehyde (4F)
Synthesized according to GP2 using 3F (0.35 g, 1.68 mmol) and purified by flash column chromatography (5% EtOAc/DCM, Rf = 0.55, dry loading using celite) to give an off-white solid (0.31 g, 91%). 1H NMR (300 MHz, CDCl3) δ 9.46 (s, 1H), 8.00 (d, J = 8.7 Hz, 2H), 7.79 (d, J = 8.7 Hz, 2H), 3.08 (3H, s) 13C NMR (75 MHz, CDCl3) δ 176.2, 142.5, 133.8, 127.7, 125.1, 91.2, 89.2, 44.4. HRMS (EI) calc'd for C10H8O3S [M˙]+: 208.0194, found: 208.0181.3-(4-((Trifluoromethyl)sulfonyl)phenyl)propiolaldehyde (4G)
Synthesized according to GP2 using 3G (0.30 g, 0.76 mmol) and the resulting orange oil (0.30 g, >95%) was carried forward without further purification (5% EtOAc/DCM = 0.91). 1H NMR (300 MHz, CDCl3) δ 9.47 (s, 1H), 8.09 (d, J = 8.3 Hz, 2H), 7.87 (d, J = 8.3 Hz, 2H). 13C NMR (600 MHz, CDCl3) δ 175.9, 133.9, 133.2, 130.9, 128.1, 119.7 (q, JC,F = 324.3 Hz), 91.0, 89.9. 19F NMR (282 MHz, CDCl3) δ –77.89. HRMS (EI) calc'd for C10H5F3O3S [M˙]+: 261.9911, found: 261.9912.N,N-Dimethyl-4-(3-oxoprop-1-yn-1-yl)benzenesulfonamide (4H)
Synthesized according to GP2 using 3H (0.75 g, 3.12 mmol) and the resulting yellow solid (0.73 g, >95%) was carried forward without further purification (70% EtOAc/Hex, Rf = 0.67). 1H NMR (600 MHz, CDCl3) δ 9.47 (s, 1H), 7.84 (d, J = 8.0 Hz, 2H), 7.78 (d, J = 8.0 Hz, 2H), 2.77 (s, 6H). 13C NMR (150 MHz, CDCl3) δ 176.3, 138.0, 133.5, 127.9, 123.9, 91.8, 89.8, 37.9. HRMS (EI) calc'd for C11H11NO3S [M˙]+: 237.0460, found: 237.0473.General procedure 3 (GP3): Grignard reaction with ethynyl magnesium bromide24
In a flamed dried flask under N2, 4A–H (1.0 eq.) was dissolved in anhydrous THF (∼0.1 M) and the solution was cooled to −78 °C. Ethynylmagnesium bromide (2.0 eq.) was then added dropwise. The reaction was stirred at this temperature for 3 h and then quenched with H2O (10 mL) and sat. NH4Cl (5 mL), and then extracted with EtOAc (3 × 50 mL). The organic layer was dried over MgSO4 and concentrated in vacuo. The resulting solid was purified by flash column chromatography as described below.1-(4-Nitrophenyl)penta-1,4-diyn-3-ol (5A)
Synthesized according to GP3 using 4A (0.20 g, 1.14 mmol) and purified by flash column chromatography (30% EtOAc/hexanes, Rf = 0.27, dry loading using celite) to give a yellow solid (0.12 g, 54%). 1H NMR (300 MHz, CDCl3) δ 8.21 (d, J = 8.9 Hz, 2H), 7.62 (d, J = 8.9 Hz, 2H), 5.38 (dd, J = 7.5, 2.4 Hz, 1H), 2.67 (d, J = 2.4 Hz, 1H), 2.42 (br d, J = 7.5 Hz, 1H). 13C NMR (75 MHz, CDCl3) δ 147.6, 132.7, 128.5, 123.6, 90.3, 82.5, 80.0, 73.6, 52.4. Characterization data are consistent with previously reported values.241-Phenylpenta-1,4-diyn-3-ol (5B)
Synthesized according to GP3 using 4B (0.24 g, 1.84 mmol) and purified by flash column chromatography (30% EtOAc/Hexanes, Rf = 0.51, dry loading using celite) to give a yellow oil (0.25 g, 87%). 1H NMR (400 MHz, CDCl3) δ 7.50–7.47 (m, 2H), 7.38–7.31 (m, 3H), 5.37 (d, J = 2.4 Hz, 1H), 2.63 (d, J = 2.4 Hz, 1H), 2.44 (br s, 1H). 13C NMR (100 MHz, CDCl3) δ 131.8, 128.9, 128.3, 121.7, 85.4, 84.7, 80.8, 72.9, 52.5. Characterization data are consistent with previously reported values.231-(Perfluorophenyl)penta-1,4-diyn-3-ol (5C)
Synthesized according to GP3 using 4C (0.20 g, 0.90 mmol) and purified by flash column chromatography (10% EtOAc/hexanes, Rf = 0.21, dry loading using celite) to give a pale-yellow powder (0.12 g, 56%). 1H NMR (600 MHz, CDCl3) δ 5.40 (dd, J = 8.0, 2.3 Hz, 1H), 2.66 (d, J = 2.3 Hz, 1H), 2.53 (d, J = 8.0 Hz, 1H).13C NMR (125 MHz, CDCl3) δ 148.6–148.5 (m), 146.9–146.8 (m), 143.2–142.9 (m), 141.5–141.3 (m), 138.7–138.5 (m), 137.0–136.8 (m), 99.01–98.8 (m), 97.8, 79.6, 69.0, 52.6. 19F NMR (282 MHz, CDCl3) δ −135.15–−135.29 (m, 2F), −150.69–−150.86 (m, 1F), −161.12–−161.32 (m, 2F). HRMS (EI) calc'd for C11H3F5O [M˙]+: 246.0104, found: 246.0111.1-(4-(3-Hydroxypenta-1,4-diyn-1-yl)phenyl)ethan-1-one (5D)
Synthesized according to GP3 using 4D (0.20 g, 1.16 mmol) and purified by flash column chromatography (30% EtOAc/hexanes, Rf = 0.27, dry loading using celite) to give a yellow oil (0.20 g, 86%). 1H NMR (300 MHz, CDCl3) δ 7.92 (d, J = 8.5 Hz, 2H), 7.53 (d, J = 8.5 Hz, 2H), 4.54 (s, 2H), 2.61 (s, 3H). 13C NMR (75 MHz, CDCl3) δ 197.3, 136.5, 131.8, 128.2, 127.4, 90.4, 84.9, 51.6, 26.6. Characterization data are consistent with previously reported values.234-(3-Hydroxypenta-1,4-diyn-1-yl)benzonitrile (5E)
Synthesized according to GP3 using 4E (0.20 g, 1.30 mmol) and purified by flash column chromatography (30% EtOAc/hexanes, Rf = 0.31, dry loading using celite) to give a yellow solid (0.14 g, 61%). 1H NMR (300 MHz, CDCl3) δ 1H NMR (300 MHz, CDCl3) δ 7.62 (d, J = 8.8 Hz, 2H), 7.55 (d, J = 8.8 Hz, 2H), 5.37 (dd, J = 7.3, 2.3 Hz, 1H), 2.65 (d, J = 2.3 Hz, 1H), 2.44 (br d, J = 7.3 Hz, 1H). 13C NMR (75 MHz, CDCl3) δ 132.3, 132.0, 126.6, 118.2, 112.4, 89.5, 82.8, 80.1, 73.5, 52.4. HRMS (EI) calc'd for C12H7NO [M˙]+: 181.0528, found: 181.0516.1-(4-(Methylsulfonyl)phenyl)penta-1,4-diyn-3-ol (5F)
Synthesized according to GP3 using 4F (0.20 g, 0.96 mmol) and purified by flash column chromatography (15% EtOAc/DCM, Rf = 0.41, dry loading using celite) to give a yellow solid (0.15 g, 66%). 1H NMR (300 MHz, CDCl3) δ 7.89 (d, J = 8.7 Hz, 2H), 7.62 (d, J = 8.7 Hz, 2H), 5.37 (d, J = 2.3 Hz, 1H), 3.06 (s, 3H), 2.65 (d, J = 2.3 Hz, 1H). 13C NMR (75 MHz, CDCl3) δ 140.3, 132.6, 127.6, 127.4, 89.4, 82.6, 80.2, 73.5, 52.4, 44.4. HRMS (EI) calc'd for C12H10O3S [M˙]+: 234.0351, found: 234.0325.1-(4-((Trifluoromethyl)sulfonyl)phenyl)penta-1,4-diyn-3-ol (5G)
Synthesized according to GP3 using 4G (0.30 g, 1.14 mmol) and purified by flash column chromatography (8% EtOAc/DCM, Rf = 0.65, dry loading using celite) to give a dark orange oil (0.18 g, 54%). 1H NMR (300 MHz, CDCl3) δ 8.01 (d, J = 8.7 Hz, 2H), 7.73 (d, J = 8.7 Hz, 2H), 5.39 (d, J = 2.3 Hz, 1H), 2.66 (d, J = 2.3 Hz, 1H). 13C NMR (150 MHz, CDCl3) δ 133.0, 131.0, 130.7, 130.6, 119.7 (q, JC,F = 324.3 Hz), 91.4, 82.2, 79.9, 73.8, 52.4. 19F NMR (282 MHz, CDCl3) δ –78.16. HRMS (EI) calc'd for C12H7F3O3S [M˙]+: 288.0068, found: 288.0055.4-(3-Hydroxypenta-1,4-diyn-1-yl)-N,N-dimethylbenzenesulfonamide (5H)
Synthesized according to GP3 using 4H (0.30 g, 1.25 mmol) and the resulting yellow solid (0.27 g, 75%) was carried forward without further purification (70% EtOAc/Hex, Rf = 0.49). 1H NMR (600 MHz, CDCl3) δ 7.76 (d, J = 8.7 Hz, 2H), 7.64 (d, J = 8.7 Hz, 2H), 5.39 (dd, J = 7.6, 2.3 Hz, 1H), 2.73 (s, 6H), 2.67 (d, J = 2.3 Hz, 1H), 2.50 (d, J = 7.6 Hz, 1H). 13C NMR (150 MHz, CDCl3) δ 135.5, 132.4, 127.9, 127.7, 88.9, 82.9, 80.3, 73.5, 52.5, 37.9. HRMS (EI) calc'd for C13H13NO3S [M˙]+: 263.0616, found: 263.0639.1-(4-Nitrophenyl)penta-1,4-diyn-3-one (6A)
Synthesized according to GP2 using 5A (0.12 g, 0.60 mmol) and purified by flash column chromatography (30% EtOAc/hexanes, Rf = 0.50, dry loading using celite) to give a yellow solid (0.11 g, 88%). 1H NMR (300 MHz, CDCl3) δ 8.28 (d, J = 8.9 Hz, 2H), 7.79 (d, J = 8.9 Hz, 2H), 3.46 (s, 1H). 13C NMR (75 MHz, CDCl3) δ 159.4, 148.9, 134.0, 125.7, 123.8, 91.1, 88.0, 81.8, 80.3. Characterization data are consistent with previously reported values.241-Phenylpenta-1,4-diyn-3-one (6B)
Synthesized according to GP2 using 5B (0.25 g, 1.60 mmol) and the resulting solid (0.24 g, >95%) was carried forward without further purification (30% EtOAc/hexanes, Rf = 0.77). 1H NMR (400 MHz, CDCl3) δ 7.65–7.63 (m, 2H), 7.54–7.49 (m, 1H), 3.80 (s, 1H). 13C NMR (75 MHz, CDCl3) δ 160.1, 133.5, 131.5, 128.7, 119.1, 92.5, 89.0, 82.2, 79.0. Characterization data are consistent with previously reported values.231-(Perfluorophenyl)penta-1,4-diyn-3-one (6C)
Synthesized according to modified GP2 using 5C (0.12 g, 0.49 mmol) and the resulting white powder (0.11 g, >95%) was carried forward without further purification (10% EtOAc/Hex, Rf = 0.53). 1H NMR (600 MHz, CDCl3) δ 3.49 (s, 1H). 13C NMR (150 MHz, CDCl3) δ 158.7, 149.2–149.1 (m), 147.5–147.4 (m), 144.9–144.6 (m), 143.1–142.9 (m), 138.7–138.5 (m), 137.0–136.8 (m), 97.6, 97.0–96.7 (m), 81.5, 81.1, 74.5. 19F NMR (282 MHz, CDCl3) δ −131.87–−132.00 (m, 2F), −145.45–−145.62 (m, 1F), −159.49–−159.76 (m, 2F). HRMS (EI) calc'd for C11HF5O [M˙]+: 243.9948, found: 243.9922.1-(4-Acetylphenyl)penta-1,4-diyn-3-one (6D)
Synthesized according to GP2 using 5D (0.20 g, 1.0 mmol) and purified by flash column chromatography (30% EtOAc/hexanes, Rf = 0.43, dry loading using celite) to give a grey solid (0.18 g, 92%). 1H NMR (300 MHz, CDCl3) δ 7.99 (d, J = 8.7 Hz, 2H), 7.73 (d, J = 8.7 Hz, 2H), 3.43 (s, 1H), 2.64 (s, 3H). 13C NMR (75 MHz, CDCl3) δ 196.9, 159.8, 138.6, 133.5, 128.4, 123.6, 90.4, 90.2, 82.0, 79.7, 26.7. Characterization data are consistent with previously reported values.234-(3-Oxopenta-1,4-diyn-1-yl)benzonitrile (6E)
Synthesized according to GP2 using 5E (0.14 g, 0.77 mmol) and purified by flash column chromatography (30% EtOAc/Hexanes, Rf = 0.48, dry loading using celite) to give a yellow solid (0.12 g, 88%). 1H NMR (300 MHz, CDCl3) δ 7.71 (s, 4H), 3.44 (s, 1H). 13C NMR (75 MHz, CDCl3) δ 159.7, 133.7, 132.5, 124.0, 117.8, 114.9, 90.9, 88.7, 81.9, 80.4. HRMS (EI) calc'd for C12H5NO [M˙]+: 179.0371, found: 179.0362.1-(4-(Methylsulfonyl)phenyl)penta-1,4-diyn-3-one (6F)
Synthesized according to GP2 using 5F (0.14 g, 0.60 mmol) and the resulting yellow solid (0.14 g, >95%) was used without further purification (15% EtOAc/DCM, Rf = 0.69). 1H NMR (600 MHz, CDCl3) δ 8.00 (d, J = 8.6 Hz, 2H), 7.81 (d, J = 8.7 Hz, 2H), 3.45 (s, 1H), 3.06 (s, 3H). 13C NMR (150 MHz, CDCl3) δ 159.7, 142.7, 134.1, 127.8, 124.9, 90.5, 88.6, 82.0, 80.3, 77.4, 77.2, 76.9, 44.5. HRMS (EI) calc'd for C12H8O3S [M˙]+: 232.0194, found: 232.0187.1-(4-((Trifluoromethyl)sulfonyl)phenyl)penta-1,4-diyn-3-one (6G)
Synthesized according to GP2 using 5G (0.18 g, 0.62 mmol) the resulting oil was triturated with hexanes to give a beige solid (0.14 g, 80%) which was used without further purification (30% EtOAc/DCM, Rf = 0.62). 1H NMR (300 MHz, CDCl3) δ 8.09 (d, J = 8.3 Hz, 2H), 7.89 (d, J = 8.3 Hz, 2H), 3.49 (s, 1H). 13C NMR (150 MHz, CDCl3) δ 159.3, 134.0, 133.3, 130.9, 127.8, 119.7 (q, JC,F = 324.3 Hz), 91.4, 87.2, 81.7, 80.7. 19F NMR (282 MHz, CDCl3) δ –77.88. HRMS (EI) calc'd for C12H5F3O3S [M˙]+: 285.9911, found: 284.9909.N,N-Dimethyl-4-(3-oxopenta-1,4-diyn-1-yl)benzenesulfonamide (6H)
Synthesized according to GP2 using 5H (0.27 g, 1.03 mmol, 1.0 eq.) and the resulting yellow solid (0.26 g, >95%) was used without further purification (70% EtOAc/Hex, Rf = 0.75). 1H NMR (600 MHz, CDCl3) δ 7.81–7.77 (m, 4H), 3.45 (s, 1H), 2.74 (s, 6H). 13C NMR (150 MHz, CDCl3) δ 159.7, 138.2, 133.7, 127.9, 123.6, 90.3, 89.1, 81.9, 80.1, 37.9. HRMS (EI) calc'd for C13H11NO3S [M˙]+: 261.0460, found: 261.0442.General procedure 4: synthesis of azides
To a stirring solution of alkyl bromides or chlorides (1.0 eq., commercially available unless indicated otherwise) in DMF (∼0.5 M) was added NaN3 (2.0 eq.). The reaction was stirred at 60 °C for 16 h, after which it was concentrated in vacuo, diluted with water (20 mL), and then extracted with EtOAc (3 × 20 mL). The combined organic layer was washed with brine (25 mL), dried over MgSO4, and concentrated in vacuo to give azides (7A, E–J) which were used without further purification.1-(Azidomethyl)-4-nitrobenzene (7A)
Synthesized according to GP4 using 4-nitrobenzyl bromide (2.16 g, 10.00 mmol) to give a yellow oil (1.43 g, 80%). 1H NMR (300 MHz, CDCl3) δ 8.24 (d, J = 6.6 Hz, 2H), 7.50 (d, J = 6.6 Hz, 2H), 4.50 (s, 2H). 13C NMR (75 MHz, CDCl3) δ 142.4, 137.8, 124.8, 120.6, 55.8. Characterization data are consistent with previously reported values.24(4-(Dimethylamino)phenyl)methanol (7B1)
To a solution of 4-(dimethylamino)benzaldehyde (0.50 g, 3.35 mmol, 1.0 eq.) in EtOH (20 mL) at R.T. was added NaBH4 (0.14 g, 3.69 mmol, 1.1 eq.) portion wise. The reaction was stirred at R.T. for 5 h, concentrated in vacuo and then quenched with H2O (25 mL). The aqueous phase was extracted with EtOAc (3 × 25 mL) and the combined organic layer was washed with brine (50 mL), dried over MgSO4 and concentrated in vacuo to give a clear oil (0.48 g, 95%). 1H NMR (300 MHz, CDCl3) δ 7.24 (d, J = 8.7 Hz, 2H), 6.73 (d, J = 8.7 Hz, 2H), 4.56 (s, 2H), 2.95 (s, 6H). Characterization data are consistent with previously reported values.434-(Azidomethyl)-N,N-dimethylaniline (7B2)
To a solution of 7B1 (0.43 g, 2.81 mmol, 1.0 eq.) in anhydrous THF (15 mL) at 0 °C were added diphenylphosphoryl azide (0.91 mL, 4.22 mmol, 1.5 eq.) and DBU (0.63 mL, 4.22 mmol, 1.5 eq.) dropwise. The reaction was warmed to R.T. and stirred for 16 h. The reaction mixture was quenched with 1 M HCl (30 mL) and extracted with EtOAc (25 mL). The aqueous layer was then basified with 1 M NaOH to pH ≈ 8 and extracted with EtOAc (3 × 15 mL). The combined organic layer was washed with brine (50 mL), dried over MgSO4 and concentrated in vacuo to give a yellow oil which was purified by flash column chromatography (10% EtOAc/hexanes, Rf = 0.52, dry loading using celite) to give a clear colourless oil (0.23 g, 46%). 1H NMR (300 MHz, CDCl3) δ 7.19 (d, J = 8.7 Hz, 2H), 6.72 (d, J = 8.7 Hz, 2H), 4.23 (s, 2H), 2.97 (s, 6H). 13C NMR (75 MHz, CDCl3) δ 150.5, 129.6, 122.7, 112.4, 54.7, 40.5. Characterization data are consistent with previously reported values.44(Adamantan-1-yl)methyl methanesulfonate (7C1)
To a solution of 1-adamantanemethanol (1.00 g, 6.00 mmol, 1.0 eq.) in DCM (25 mL) was added NEt3 (2.10 mL, 15.00 mmol, 2.5 eq.) and the mixture was cooled to 0 °C. Then methanesulfonyl chloride (0.60 mL, 7.20 mmol, 1.2 eq.) was added dropwise and the reaction mixture was allowed to warm to R.T. and stirred for 4 h. The reaction was quenched with sat. NaHCO3 (25 mL) and extracted with EtOAc (3 × 30 mL). The combined organic layer was washed with sat. NH4Cl (50 mL), brine (50 mL), and then dried over MgSO4 and concentrated in vacuo to give a pale-yellow solid which was used without further purification (1.43 g, >95%). 1H NMR (300 MHz, CDCl3) δ 3.78 (s, 2H), 2.99 (2, 3H), 2.02 (apparent s, 3H), 1.76–1.63 (m, 6H), 1.57 (m, 6H). 13C NMR (75 MHz, CDCl3) δ 79.5, 38.9, 37.2, 36.9, 33.6, 28.0. Characterization data are consistent with previously reported values.451-(Azidomethyl)adamantane (7C2)
To a solution of 7C1 in DMF (10 mL) was added NaN3 (1.95 g, 30.00 mmol, 10.0 eq.) and the reaction was stirred at 120 °C for 48 h. The reaction mixture was quenched with H2O (20 mL) and extracted with 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4 EtOAc/hexanes (3 × 25 mL). The combined organic layer was washed with brine (25 mL), dried over MgSO4 and concentrated in vacuo to give a pale-yellow oil which was used without further purification (0.47 g, 82%). 1H NMR (300 MHz, CDCl3) δ 2.95 (s, 2H), 2.01–1.98 (m, 3H), 1.76–1.59 (m, 6H), 1.53–1.52 (m, 6H). 13C NMR (75 MHz, CDCl3) δ 64.3, 40.0, 36.8, 34.8, 28.2. Characterization data are consistent with previously reported values.45
4 EtOAc/hexanes (3 × 25 mL). The combined organic layer was washed with brine (25 mL), dried over MgSO4 and concentrated in vacuo to give a pale-yellow oil which was used without further purification (0.47 g, 82%). 1H NMR (300 MHz, CDCl3) δ 2.95 (s, 2H), 2.01–1.98 (m, 3H), 1.76–1.59 (m, 6H), 1.53–1.52 (m, 6H). 13C NMR (75 MHz, CDCl3) δ 64.3, 40.0, 36.8, 34.8, 28.2. Characterization data are consistent with previously reported values.45
4-(Azidomethyl)tetrahydro-2H-pyran (7D)
To a solution of tetrahydropyran-4-methanol (0.35 mL, 3.00 mmol, 1.0 eq.) in DCM (15 mL) was added NEt3 (1.00 mL, 7.50 mmol, 2.5 eq.) and the mixture was cooled to 0 °C. Then methanesulfonyl chloride (0.30 mL, 3.60 mmol, 1.2 eq.) was added dropwise and the reaction mixture was allowed to warm to R.T. and stirred for 4 h. The reaction was quenched with sat. NaHCO3 (15 mL) and extracted with DCM (2 × 10 mL). The combined organic layer was washed brine (25 mL) and then dried over MgSO4 and concentrated in vacuo to give a pale-yellow oil. The oil was dissolved in DMF (10 mL) and to this solution was added NaN3 (0.98 g, 15.00 mmol, 5.0 eq.) and the reaction was stirred at 80 °C for 16 h. The reaction mixture was quenched with H2O (20 mL) and extracted with Et2O (3 × 10 mL). The combined organic layer was washed with brine (25 mL), dried over MgSO4 and concentrated in vacuo to give a pale-yellow oil which was used without further purification (0.30 g, 72%). 1H NMR (300 MHz, CDCl3) δ 4.00–3.94 (m, 2H), 3.37 (td, J = 11.7, 2.1 Hz, 2H), 3.17 (d, J = 6.9 Hz, 2H), 1.83–1.72 (m, 1H), 1.68–1.60 (m, 2H), 1.41–1.24 (m, 2H). 13C NMR (75 MHz, CDCl3) δ 67.6, 57.3, 35.5, 30.5. Characterization data are consistent with previously reported values.461-(Azidomethyl)-4-(trifluoromethyl)benzene (7E)
Synthesized according to GP4 using 4-trifluoromethylbenzyl bromide (0.50 g, 2.10 mmol) give a clear colourless liquid (0.27 g, 65%). 1H NMR (300 MHz, CDCl3) δ 7.66 (d, J = 7.8 Hz, 2H), 7.45 (d, J = 7.8 Hz, 2H), 4.43 (s, 2H). 13C NMR (75 MHz, CDCl3) δ 139.4, 130.5 (q, JCF = 32.3 Hz), 128.3, 125.8 (q, JCF = 1.8 Hz), 124.0 (q, JCF = 270.3 Hz), 54.1. 19F NMR (283 MHz, CDCl3) δ −62.6. Characterization data are consistent with previously reported values.471-Azidohexane (7F)
Synthesized according to GP4 using 1-bromohexane (0.42 mL, 3.00 mmol) to give a clear colourless liquid (0.32 g, 84%). 1H NMR (300 MHz, CDCl3) δ 3.25 (t, J = 6.9 Hz, 2H), 1.66–1.51 (m, 2H), 1.45–1.20 (m, 6H), 0.89 (t, J = 6.9 Hz, 3H). 13C NMR (75 MHz, CDCl3) δ 51.6, 31.5, 28.9, 26.5, 22.6, 14.1. Characterization data are consistent with previously reported values.484-(Azidomethyl)benzonitrile (7G)
Synthesized according to GP4 using 4-cyanobenzyl bromide (0.50 g, 2.50 mmol) to give a clear colourless liquid (0.33 g, 83%). 1H NMR (300 MHz, CDCl3) δ 7.66 (d, J = 8.0 Hz, 2H), 7.43 (d, J = 8.0 Hz, 2H), 4.47 (s, 2H). 13C NMR (75 MHz, CDCl3) δ 140.8, 132.6, 128.5, 118.4, 112.1, 53.7. Characterization data are consistent with previously reported values.494-(2-Azidoethyl)morpholine (7H)
Synthesized according to GP4 using 4-(2-Chloroethyl)morpholine (0.18 mL, 1.30 mmol) to give a colourless oil (0.20 g, 96%). 1H NMR (400 MHz, CDCl3) 3.72 (t, J = 4.4 Hz, 4H), 3.35 (t, J = 6.0 Hz, 2H), 2.59 (t, J = 6.0 Hz, 2H), 2.59 (t, J = 4.4 Hz, 4H). δ13C NMR (100 MHz, CDCl3) δ 67.0, 57.7, 53.7, 48.0. Characterization data are consistent with previously reported values.50tert-Butyl 4-(azidomethyl)piperidine-1-carboxylate (7I)
Synthesized according to GP4 using tert-butyl 4-(bromomethyl)piperidine-1-carboxylate (0.50 g, 1.80 mmol) to give a colourless oil (0.41 g, 94%). 1H NMR (400 MHz, CDCl3) 4.11 (d, J = 12.8 Hz, 2H), 3.17 (d, J = 6.0 Hz, 2H), 2.67 (td, J = 13.2, 2.8 Hz, 2H), 1.73–1.63 (m, 3H), 1.44 (s, 9H), 1.20–1.09 (m, 2H). δ13C NMR (100 MHz, CDCl3) δ 154.9, 79.6, 57.1, 43.6, 36.4, 29.7, 28.6. Characterization data are consistent with previously reported values.515-(Azidomethyl)picolinonitrile (7J)
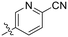
Synthesized according to GP4 using 2-cyano-5-bromomethylpyridine (0.40 g, 2.00 mmol) to give a brown solid (0.26 g, 82%). 1H NMR (600 MHz, CDCl3) δ 8.70 (s, 1H), 7.84 (dd, J = 8.3, 1.8 Hz, 1H), 7.75 (d, J = 8.0 Hz, 1H), 4.55 (s, 2H). 13C NMR (150 MHz, CDCl3) 150.3, 136.2, 135.3, 133.6, 128.4, 116.9, 51.5. Characterization data are consistent with previously reported values.52General procedure 4 (GP5): CuAAC reaction with CuI and DIPEA23
To a solution of 6A–H (1.0 eq.), 7A–J (1.0 eq.), and CuI (1.0 eq.) in anhydrous MeCN (∼0.1 M relative to 6) under N2 at R.T. was added DIPEA (1.0 eq.) dropwise and the reaction was stirred for 16 h. The reaction mixture was then diluted with DCM (20 mL) and washed with 1 M HCl (2 × 15 mL), brine (20 mL), sat. NH4OH (4 × 15 mL, until aqueous phase was no longer blue), and brine (20 mL) again. The organic layer was then dried over MgSO4 and concentrated in vacuo to give a residue which was purified by flash column chromatography as described below.1-(1-(4-Nitrobenzyl)-1H-1,2,3-triazol-4-yl)-3-(4-nitrophenyl)prop-2-yn-1-one (8A)
Synthesized according to GP5 using 6A (0.10 g, 0.50 mmol) and 7A (0.09 g, 0.50 mmol) and purified by flash column chromatography (1–5% EtOAc/DCM, in 2% EtOAc/DCM Rf = 0.23, dry loading using celite) to give a yellow solid (0.10 g, 54%). 1H NMR (300 MHz, DMSO-d6) δ 9.29 (s, 1H), 8.36 (d, J = 9.0 Hz, 2H), 8.26 (d, J = 8.7 Hz, 2H), 8.04 (d, J = 9.0 Hz, 2H), 7.59 (d, J = 8.7 Hz, 2H), 5.91 (s, 2H). 13C NMR (75 MHz, DMSO-d6) δ 168.2, 148.6, 147.4, 146.9, 142.6, 134.3, 130.9, 129.2, 125.4, 124.1, 124.0, 89.5, 88.7, 52.4. HRMS (EI) calc'd for C18H11N5O5 [M˙]+: 377.0760, found: 377.0750. Characterization data are consistent with previously reported values.231-(1-(4-(Dimethylamino)benzyl)-1H-1,2,3-triazol-4-yl)-3-(4-nitrophenyl)prop-2-yn-1-one (8B)
Synthesized according to GP5 using 6A (0.09 g, 0.45 mmol) and 7B (0.08 g, 0.45 mmol) and purified by flash column chromatography (1–5% EtOAc/DCM, in 2% EtOAc/DCM Rf = 0.27, dry loading using celite) to give an orange solid (0.12 g, 71%). 1H NMR (300 MHz, CDCl3) δ 8.27 (d, J = 9.0 Hz, 2H), 8.00 (s, 1H), 7.86 (d, J = 9.0 Hz, 2H), 7.21 (d, J = 8.7 Hz, 2H), 6.71 (d, J = 8.7 Hz, 2H), 5.91 (s, 2H), 2.98 (s, 6H). 13C NMR (75 MHz, CDCl3) δ 170.0, 161.2, 148.7, 148.0, 134.1, 130.0, 126.8, 126.7, 123.9, 120.1, 112.7, 90.1, 89.9, 54.7, 40.5. HRMS (EI) calc'd for C20H17N5O3 [M˙]+: 377.1331, found: 375.1324.1-(1-((Adamantan-1-yl)methyl)-1H-1,2,3-triazol-4-yl)-3-(4-nitrophenyl)prop-2-yn-1-one (8C)
Synthesized according to GP5 using 6A (0.08 g, 0.40 mmol) and 7C (0.08 g, 0.40 mmol) and purified by flash column chromatography (2% EtOAc/DCM, Rf = 0.43, dry loading using celite) to give a pale-yellow solid (0.10 g, 67%). 1H NMR (300 MHz, CDCl3) δ 8.28 (d, J = 9.0 Hz, 2H), 8.10 (s, 1H), 7.89 (d, J = 9.0 Hz, 2H), 4.12 (s, 2H), 2.03 (apparent s, 3H, 1.75–1.58 (m, 6H), 1.53–1.52 (m, 6H). 13C NMR (75 MHz, CDCl3) δ 170.1, 148.8, 147.6, 134.2, 128.0, 126.8, 123.9, 90.1 (2 alkyne C), 62.8, 40.3, 36.5, 34.4, 28.1. HRMS (ESI) calc'd for C22H22N4O3Na [M + Na]+: 413.1589, found: 413.1590.3-(4-Nitrophenyl)-1-(1-((tetrahydro-2H-pyran-4-yl)methyl)-1H-1,2,3-triazol-4-yl)prop-2-yn-1-one (8D)
Synthesized according to GP5 using 6A (0.08 g, 0.40 mmol) and 7D (0.05 g, 0.40 mmol) and purified using preparative thin layer chromatography by eluting 3 times (1% MeOH/DCM, Rf = 0.19) to give a pale-yellow solid (0.03 g, 19%).531H NMR (300 MHz, CDCl3) δ 8.27 (d, J = 9.0 Hz, 2H), 8.16 (s, 1H), 7.85 (d, J = 9.0 Hz, 2H), 4.33 (d, J = 7.2 Hz, 2H), 4.00–3.96 (m, 2H), 3.35 (td, J = 11.4, 2.1 Hz, 2H), 2.29–2.11 (m, 1H), 1.54–1.34 (m, 4H). 13C NMR (75 MHz, CDCl3) δ 169.8, 148.7, 147.8, 134.1, 127.3, 126.6, 123.8, 90.1, 89.8, 67.1, 56.3, 36.2, 30.2. HRMS (EI) calc'd for C17H16N4O4 [M˙]+: 340.1172, found: 340.1149.3-(4-Nitrophenyl)-1-(1-(4-(trifluoromethyl)benzyl)-1H-1,2,3-triazol-4-yl)prop-2-yn-1-one (8E)
Synthesized according to GP5 using 6A (0.08 g, 0.40 mmol) and 7E (0.08 g, 0.40 mmol) and purified using flash chromatography (1% EtOAc/DCM, Rf = 0.21, dry loading using celite) to give a beige solid (0.11 g, 71%). 1H NMR (300 MHz, CDCl3) δ 8.27 (d, J = 9.0 Hz, 2H), 8.17 (s, 1H), 7.85 (d, J = 9.0 Hz, 2H), 7.67 (d, J = 8.1 Hz, 2H), 7.43 (d, J = 8.1 Hz, 2H), 5.69 (s, 2H). 13C NMR (75 MHz, CDCl3) δ 169.7, 148.8, 148.5, 137.2, 134.2, 131.7 (q, JCF = 32.8 Hz), 128.6, 127.0, 126.6, 126.5 (q, JCF = 3.7 Hz), 123.9, 123.7 (q, JCF = 293.0 Hz), 90.4, 89.8, 54.1. 19F NMR (282 MHz, CDCl3) δ −62.8. HRMS (EI) calc'd for C19H11F3N4O3 [M˙]+: 400.0783, found: 400.0784.1-(1-Hexyl-1H-1,2,3-triazol-4-yl)-3-(4-nitrophenyl)prop-2-yn-1-one (8F)
Synthesized according to GP5 using 6A (0.08 g, 0.40 mmol) and 7F (0.05 g, 0.40 mmol) and purified using flash chromatography (0 → 3% EtOAc/DCM, 3% EtOAc/DCM Rf = 0.53, dry loading using celite) to give a pale-yellow solid (0.08 g, 61%). 1H NMR (300 MHz, CDCl3) δ 8.31 (d, J = 9.0 Hz, 2H), 8.22 (s, 1H), 7.91 (d, J = 9.0 Hz, 2H), 4.49 (t, J = 7.2 Hz, 2H), 2.04–1.95 (m, 2H), 1.43–1.29 (m, 6H), 0.92 (t, J = 7.2 Hz, 3H). 13C NMR (75 MHz, CDCl3) δ 169.9, 148.6, 147.9, 134.1, 126.7, 123.8, 90.0, 89.8, 51.0, 31.1, 30.1, 26.0, 22.4, 13.9. HRMS (EI) calc'd for C17H18N4O3 [M˙]+: 326.1379, found: 326.1357.4-((4-(3-(4-Nitrophenyl)propioloyl)-1H-1,2,3-triazol-1-yl)methyl)benzonitrile (8G)
Synthesized according to GP5 using 6A (0.06 g, 0.32 mmol) and 7G (0.05 g, 0.32 mmol) and purified by flash column chromatography (5% EtOAc/DCM, Rf = 0.29, dry loading using celite) to give a pale-yellow solid (0.09 g, 77%). 1H NMR (300 MHz, DMSO-d6) δ 9.24 (s, 1H), 8.31 (d, J = 9.0 Hz, 2H), 8.01 (d, J = 9.0 Hz, 2H), 7.84 (d, J = 8.4 Hz, 2H), 7.49 (d, J = 8.4 Hz, 2H), 5.82 (s, 2H). 13C NMR (75 MHz, DMSO-d6) δ 168.2, 148.6, 146.9, 140.7, 134.3, 132.8, 130.8, 128.8, 125.4, 118.5, 111.2, 89.5, 88.7, 52.7. HRMS (ESI) calc'd for C38H22N10O6 [2 M + Na]+: 737.1645, found: 737.1648.1-(1-(2-Morpholinoethyl)-1H-1,2,3-triazol-4-yl)-3-(4-nitrophenyl)prop-2-yn-1-one (8H)
Synthesized according to GP5 using 6A (0.08 g, 0.40 mmol) and 7H (0.06 g, 0.40 mmol), and purified by flash column chromatography (2% MeOH/EtOAc, Rf = 0.44, dry loading using celite) to give a pale-yellow solid (0.08 g, 56%). 1H NMR (400 MHz, CDCl3) δ 8.38 (s, 1H), 8.27 (d, J = 9.0 Hz, 2H), 7.87 (d, J = 9.0 Hz, 2H), 4.56 (t, J = 6.0 Hz, 2H), 3.70 (t, J = 4.8 Hz, 2H), 2.87 (t, J = 6.0 Hz, 2H), 2.52 (t, J = 4.8 Hz, 4H). 13C NMR (100 MHz, CDCl3) δ 170.0, 148.8, 148.0, 134.1, 127.7, 126.8, 123.9, 90.1, 90.0, 66.9, 57.6, 53.5, 47.7. HRMS (ESI) calc'd for C17H18N5O4 [M + H]+: 356.1359, found: 356.1320.tert-Butyl 4-((4-(3-(4-nitrophenyl)propioloyl)-1H-1,2,3-triazol-1-yl)methyl)piperidine-1-carboxylate (8I)
Synthesized according to GP5 using 6A (0.10 g, 0.50 mmol) and 7I (0.12 g, 0.50 mmol), and purified by flash column chromatography (10–30% EtOAc/DCM, Rf (30% EtOAc/DCM) = 0.72, dry loading using celite) to give a yellow solid (0.15 g, 68%). 1H NMR (400 MHz, CDCl3) δ 8.26 (d, J = 8.8 Hz, 2H), 8.15 (s, 1H), 7.85 (d, J = 8.8 Hz, 2H), 4.32 (d, J = 7.2 Hz, 2H), 4.15–4.07 (m, 2H), 2.67 (t, J = 12.8 Hz, 2H), 2.18–2.06 (m, 1H), 1.63–1.56 (m, 2H), 1.43 (s, 9H), 1.27–1.17 (m, 2H). 13C NMR (100 MHz, CDCl3) δ 169.8, 154.6, 148.7, 147.9, 134.1, 127.3, 126.6, 123.8, 90.2, 89.8, 79.8, 56.1, 43.2, 37.3, 29.4, 28.4. (ESI) calc'd for C22H25N5O5Na [M + Na]+: 462.1753, found: 462.1755.3-Phenyl-1-(1-((tetrahydro-2H-pyran-4-yl)methyl)-1H-1,2,3-triazol-4-yl)prop-2-yn-1-one (8J)
Synthesized according to GP5 using 6B (0.06 g, 0.39 mmol) and 7D (0.06 g, 0.39 mmol), and purified by flash column chromatography (10% EtOAc/DCM, Rf = 0.26, dry loading using celite) to give a pale-yellow solid (0.07 g, 62%). 1H NMR (300 MHz, CDCl3) δ 8.15 (s, 1H), 7.75–7.71 (m, 2H), 7.51–7.39 (m, 2H), 4.33 (d, J = 7.2 Hz, 2H), 4.02–3.97 (m, 2H), 3.37 (td, J = 11.3, 3.7 Hz, 2H), 2.29–2.15 (m, 1H), 1.58–1.33 (m, 4H). 13C NMR (75 MHz, CDCl3) δ 170.2, 148.2, 133.5, 131.1, 128.6, 127.2, 119.9, 94.2, 87.1, 77.2, 77.0, 76.8, 67.1, 56.2, 36.2, 30.2. HRMS (EI) calc'd for C17H17N3O2 [M˙]+: 295.1321, found: 295.1341.3-(Perfluorophenyl)-1-(1-((tetrahydro-2H-pyran-4-yl)methyl)-1H-1,2,3-triazol-4-yl)prop-2-yn-1-one (8K)
Synthesized according to GP5 using 6C (0.08 g, 0.33 mmol), and 7D (0.05 g, 0.33 mmol), and purified by preparative thin layer chromatography (5% MeOH/DCM, Rf = 0.20, developed 3×) to give a pale-yellow solid (0.08 g, 63%).531H NMR (600 MHz, CDCl3) δ 8.18 (s, 1H), 4.34 (d, J = 7.3 Hz, 2H), 4.03–3.97 (m, 2H), 3.37 (td, J = 11.7, 2. Hz, 2H), 2.31–2.15 (m, 1H), 1.57–1.35 (m, 4H). 13C NMR (150 MHz, CDCl3) δ 168.6, 149.1–149.0 (m), 147.5, 147.4–147.3 (m), 144.5–144.3 (m), 142.7–142.6 (m), 138.7–138.5 (m), 137.0–136.8 (m), 127.7, 97.6–97.4 (m), 96.4, 76.0, 67.1, 56.3, 36.2, 30.2. 19F NMR (282 MHz, CDCl3) δ −132.09–−132.23 (m, 2F), −146.59–−146.77 (m, 1F), −159.88–−160.04 (m, 2F). HRMS (EI) calc'd for C17H12F5N3O2 [M˙]+: 385.0850, found: 385.0839.3-(4-Acetylphenyl)-1-(1-((tetrahydro-2H-pyran-4-yl)methyl)-1H-1,2,3-triazol-4-yl)prop-2-yn-1-one (8L)
Synthesized according to GP5 using 6D (0.08 g, 0.41 mmol) and 7D (0.06 g, 0.41 mmol), and purified by flash column chromatography (20–40% EtOAc/DCM, in 20% EtOAc/DCM Rf = 0.26, dry loading using celite) to give a pale-yellow solid (0.05 g, 36%). 1H NMR (400 MHz, CDCl3) δ 8.16 (s, 1H), 8.00 (d, J = 8.4 Hz, 2H), 7.81 (d, J = 8.4 Hz, 2H), 4.34 (d, J = 6.0 Hz, 2H), 3.41–3.33 (m, 2H), 3.37 (td, J = 11.7, 2.1 Hz, 2H), 2.64 (s, 3H), 2.29–2.17 (m, 1H), 1.56–1.36 (m, 4H). 13C NMR (100 MHz, CDCl3) δ 197.1, 170.0, 148.0, 138.2, 133.4, 128.3, 127.1, 124.4, 92.1, 88.8, 67.1, 56.2, 36.2, 30.1, 26.7. HRMS (EI) calc'd for C19H19N3O3 [M˙]+: 337.1426, found: 337.1429.4-(3-Oxo-3-(1-((tetrahydro-2H-pyran-4-yl)methyl)-1H-1,2,3-triazol-4-yl)prop-1-yn-1-yl)benzonitrile (8M)
Synthesized according to GP5 using 6E (0.07 g, 0.39 mmol) and 7D (0.06 g, 0.39 mmol), and purified by flash column chromatography (20% EtOAc/DCM Rf = 0.29, dry loading using celite) to give a pale-yellow solid (0.05 g, 38%). 1H NMR (600 MHz, CDCl3) δ 8.16 (s, 1H), 7.81 (d, J = 8.7 Hz, 2H), 7.71 (d, J = 8.7 Hz, 2H), 4.34 (d, J = 7.2 Hz, 2H), 4.00 (m, 2H), 3.37 (td, J = 11.8, 2.3 Hz, 2H), 2.23 (m,1H), 1.57–1.49 (m, 2H), 1.50–1.36 (m, 2H). 13C NMR (150 MHz, CDCl3) δ 169.9, 148.0, 133.7, 132.3, 127.3, 124.8, 118.0, 114.3, 90.7, 89.4, 77.3, 77.1, 76.9, 67.2, 56.4, 36.3, 30.3. HRMS (EI) calc'd for C18H16N4O2 [M˙]+: 320.1273, found: 320.1240.3-(4-(Methylsulfonyl)phenyl)-1-(1-((tetrahydro-2H-pyran-4-yl)methyl)-1H-1,2,3-triazol-4-yl)prop-2-yn-1-one (8N)
Synthesized according to GP5 using 6F (0.08 g, 0.34 mmol) and 7D (0.05 g, 0.34 mmol), and purified by flash column chromatography (40% EtOAc/DCM Rf = 0.25, dry loading using celite) to give a pale-yellow solid (0.07 g, 55%). 1H NMR (300 MHz, CDCl3) δ 8.21 (s, 1H), 8.03 (d, J = 8.7 Hz, 2H), 7.93 (d, J = 8.7 Hz, 2H), 4.38 (d, J = 7.2 Hz, 2H), 4.03 (m, 2H), 3.40 (td, J = 11.7, 2.6 Hz, 2H), 3.12 (s, 3H), 2.26 (m, 1H), 1.59–1.39 (m, 4H). 13C NMR (75 MHz, CDCl3) δ 169.9, 147.9, 142.2, 133.9, 127.6, 127.3, 125.7, 90.6, 89.0, 67.1, 56.3, 44.4, 36.2, 30.1. HRMS (EI) calc'd for C18H19N3O4S [M˙]+: 373.1096, found: 373.1095.1-(1-((Tetrahydro-2H-pyran-4-yl)methyl)-1H-1,2,3-triazol-4-yl)-3-(4-((trifluoromethyl)sulfonyl)phenyl)prop-2-yn-1-one (8O)
Synthesized according to GP5 using 6G (0.08 g, 0.28 mmol) and 7D (0.04 g, 0.28 mmol), and purified by flash column chromatography (15% EtOAc/DCM Rf = 0.51, dry loading using celite) to give a beige solid (0.04 g, 33%). 1H NMR (600 MHz, CDCl3) δ 8.18 (s, 1H), 8.09 (d, J = 8.8 Hz, 2H), 7.97 (d, J = 8.8 Hz, 2H), 4.35 (d, J = 7.2 Hz, 2H), 4.00 (m, 2H), 3.37 (td, J = 11.6, 2.5 Hz, 2H), 2.32–2.11 (m, 1H), 1.59–1.27 (m, 4H). 13C NMR (150 MHz, CDCl3) δ 169.6, 147.7, 134.1, 132.8, 130.8, 128.7, 127.3, 119.7 (q, JC,F = 324.3 Hz), 90.3, 89.3, 67.1, 56.3, 36.2, 30.1. 19F NMR (282 MHz, CDCl3) δ –77.95. HRMS (EI) calc'd for C18H16F3N3O4S [M˙]+: 427.0814, found: 427.0787.4-((4-(3-(4-(Methylsulfonyl)phenyl)propioloyl)-1H-1,2,3-triazol-1-yl)methyl)benzonitrile (8P)
Synthesized according to GP5 using 6F (0.06 g, 0.25 mmol) and 7G (0.04 g, 0.25 mmol), and purified by flash column chromatography (20% EtOAc/DCM Rf = 0.4, dry loading using celite) to give a white powder (0.07 g, 95%). 1H NMR (300 MHz, CDCl3) δ 8.19 (s, 1H), 8.02 (d, J = 8.2 Hz, 2H), 7.92 (d, J = 8.2 Hz, 2H), 7.74 (d, J = 8.0 Hz, 2H), 7.43 (d, J = 8.0 Hz, 2H), 5.71 (s, 2H), 3.11 (s, 3H). 13C NMR (75 MHz, CDCl3) δ 169.5, 148.5, 142.2, 138.4, 133.9, 133.1, 128.6, 127.6, 126.8, 125.5, 117.8, 113.4, 90.8, 88.8, 77.2, 77.0, 76.8, 53.9, 44.3. HRMS (EI) calc'd for C20H14N4O3S [M˙]+: 390.0787, found: 390.0778.4-(3-(1-(4-Cyanobenzyl)-1H-1,2,3-triazol-4-yl)-3-oxoprop-1-yn-1-yl)-N,N-dimethylbenzenesulfonamide (8Q)
Synthesized according to GP5 using 6H (0.12 g, 0.44 mmol) and 7G (0.07 g, 0.44 mmol), and purified by flash column chromatography (15% EtOAc/DCM Rf = 0.53, dry loading using celite) to give a white powder (0.13 g, 72%). 1H NMR (300 MHz, CDCl3) δ 8.20 (s, 1H), 7.88 (d, J = 8.7 Hz, 2H), 7.83 (d, J = 8.7 Hz, 2H), 7.73 (d, J = 8.6 Hz, 2H), 7.43 (d, J = 8.6 Hz, 2H), 5.71 (s, 2H), 2.76 (s, 6H). 13C NMR (75 MHz, CDCl3) δ 169.6, 148.5, 138.5, 137.7, 133.7, 133.1, 128.6, 127.7, 126.9, 124.3, 117.8, 113.4, 91.3, 88.6, 53.8, 37.8. HRMS (EI) calc'd for C21H17N5O3S [M˙]+: 419.1052, found: 419.1056.5-((4-(3-(4-(Methylsulfonyl)phenyl)propioloyl)-1H-1,2,3-triazol-1-yl)methyl)picolinonitrile (8R)
Synthesized according to GP5 using 6G (0.06 g, 0.25 mmol) and 7J (0.04 g, 0.25 mmol), and purified by flash column chromatography (50% EtOAc/DCM Rf = 0.40, dry loading using celite) to give an off-white powder (0.05 g, 47%). 1H NMR (600 MHz, DMSO-d6) δ 9.25 (s, 1H), 8.83 (s, 1H), 8.09–87.99 (m, 6H), 5.91 (s, 2H), 3.30 (s, 3H). 13C NMR (150 MHz, DMSO-d6) 168.3, 150.9, 146.9, 142.8, 137.6, 135.3, 133.8, 132.4, 130.8, 129.1, 127.6, 123.9, 117.3, 89.3, 88.5, 50.4, 43.1. HRMS (ESI) calc'd for C19H13N5O3SNa [M + Na]+: 414.0637, found: 414.0649.(1-((Tetrahydro-2H-pyran-4-yl)methyl)-1H-1,2,3-triazol-4-yl)methanol (9)
To a solution of propargyl alcohol (0.11 mL, 1.82 mmol, 1.0 eq.) and 7D (0.26 g, 1.82 mmol, 1.0 eq.) in 6 mL of tBuOH/H2O (2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) were added CuSO4·5H2O (0.02 g, 0.09 mmol, 5 mol%) and sodium ascorbate (0.04 g, 0.18 mmol, 10 mol%). The mixture was stirred vigorously for 16 h at R.T. and then diluted with H2O (50 mL) and extracted with EtOAc (3 × 25 mL). The organic layer was washed with brine (50 mL), dried over MgSO4, and concentrated in vacuo to give a pale yellow oil (0.17 g, 50%) which was used without further purification (100% EtOAc, Rf = 0.13). 1H NMR (600 MHz, CDCl3) δ 7.54 (s, 1H), 4.24 (d, J = 7.2 Hz, 2H), 4.08–3.90 (m, 2H), 3.36 (td, J = 11.8, 2.1 Hz, 2H), 2.17 (m, 1H), 1.65–1.46 (m, 2H), 1.40 (m, 2H). 13C NMR (150 MHz, CDCl3) δ 147.8, 122.3, 67.2, 56.3, 55.8, 36.2, 30.2. HRMS (EI) calc'd for C9H15N3O2 [M˙]+: 197.1164, found: 197.1171.
1) were added CuSO4·5H2O (0.02 g, 0.09 mmol, 5 mol%) and sodium ascorbate (0.04 g, 0.18 mmol, 10 mol%). The mixture was stirred vigorously for 16 h at R.T. and then diluted with H2O (50 mL) and extracted with EtOAc (3 × 25 mL). The organic layer was washed with brine (50 mL), dried over MgSO4, and concentrated in vacuo to give a pale yellow oil (0.17 g, 50%) which was used without further purification (100% EtOAc, Rf = 0.13). 1H NMR (600 MHz, CDCl3) δ 7.54 (s, 1H), 4.24 (d, J = 7.2 Hz, 2H), 4.08–3.90 (m, 2H), 3.36 (td, J = 11.8, 2.1 Hz, 2H), 2.17 (m, 1H), 1.65–1.46 (m, 2H), 1.40 (m, 2H). 13C NMR (150 MHz, CDCl3) δ 147.8, 122.3, 67.2, 56.3, 55.8, 36.2, 30.2. HRMS (EI) calc'd for C9H15N3O2 [M˙]+: 197.1164, found: 197.1171.
1-((Tetrahydro-2H-pyran-4-yl)methyl)-1H-1,2,3-triazole-4-carbaldehyde (10)
To a solution of 9 (0.17 g, 0.91 mmol, 1.0 eq.) in acetone (10 mL) at 0 °C were added trichloroisocyanuric acid (0.19 g, 0.83 mmol, 1.05 eq.) and TEMPO (0.001 g, 0.008 mmol, 0.01 eq.). The reaction was allowed to gradually warm to R.T. over 3 h at which point TLC analysis confirmed completion (100% EtOAc, Rf = 0.68). The reaction was concentrated in vacuo, redissolved in DCM (50 mL) and washed with sat. NaHCO3 (25 mL), 1 M HCl (25 mL), and then brine (50 mL). The organic layer was dried over MgSO4 and concentrated in vacuo to give a yellow solid (0.16 g, >95%) which was used without further purification. 1H NMR (600 MHz, CDCl3) δ 10.16 (s, 1H), 8.10 (s, 2H), 4.34 (d, J = 7.2 Hz, 2H), 4.09–3.93 (m, 2H), 3.56–3.20 (td, J = 11.8, 2.1 Hz, 2H), 2.23 (m, 1H), 1.65–1.45 (m, 2H), 1.43 (m, 2H). 13C NMR (150 MHz, CDCl3) δ 185.1, 147.7, 125.5, 67.1, 56.2, 36.1, 30.1. HRMS (EI) calc'd for C9H13N3O2 [M˙]+: 195.1008, found: 195.1011.1-(1-((Tetrahydro-2H-pyran-4-yl)methyl)-1H-1,2,3-triazol-4-yl)prop-2-yn-1-ol (11)
In a flamed dried flask under N2, 10 (0.10 g, 0.51 mmol, 1.0 eq.) was dissolved in anhydrous THF (8 mL) and the solution was cooled to −78 °C. Ethynylmagnesium bromide (0.5 M in THF) (2.00 mL, 1.02 mmol, 2.0 eq.) was then added dropwise. The reaction was stirred for 4 h, then quenched with H2O (10 mL) and sat. NH4Cl (5 mL), and extracted with EtOAc (3 × 50 mL). The organic layer was dried over MgSO4 and concentrated in vacuo. The resulting solid was purified by flash column chromatography (50% EtOAc/DCM, Rf = 0.29, dry loading using celite) to give a yellow oil (0.08 g, 75%). 1H NMR (600 MHz, CDCl3) δ 7.56 (s, 1H), 5.64 (d, J = 3.0 Hz, 1H), 4.18 (d, J = 7.2 Hz, 2H), 3.99–3.83 (m, 2H), 3.29 (td, J = 11.8, 2.2 Hz, 2H), 2.56 (d, J = 2.3 Hz, 1H), 2.11 (m, 1H), 1.53–1.40 (m, 2H), 1.34 (m, 2H). 13C NMR (150 MHz, CDCl3) δ 146.8, 121.1, 81.0, 72.9, 66.1, 55.9, 54.9, 35.2, 29.2, 29.2. HRMS (EI) calc'd for C11H15N3O2 [M˙]+: 221.1164, found: 221.1159.1-(1-((Tetrahydro-2H-pyran-4-yl)methyl)-1H-1,2,3-triazol-4-yl)prop-2-yn-1-one (12)
To a solution of 11 (0.08 g, 0.36 mmol, 1.0 eq.) in acetone (5 mL) at 0 °C were added trichloroisocyanuric acid (0.09 g, 0.38 mmol, 1.05 eq.) and TEMPO (0.002 g, 0.004 mmol, 0.01 eq.). The reaction was allowed to gradually warm to R.T. over 3 h. The reaction was concentrated in vacuo, redissolved in DCM (10 mL) and washed with sat. NaHCO3 (10 mL), 1 M HCl (10 mL), and then brine (20 mL). The organic layer was dried over MgSO4 and concentrated in vacuo and purified by flash column chromatography (30% EtOAc/DCM, Rf = 0.62) to give a white solid (0.06 g, 75%). 1H NMR (600 MHz, CDCl3) δ 8.15 (s, 1H), 4.34 (d, J = 7.2 Hz, 2H), 4.03–3.93 (m, 2H), 3.53 (s, 1H), 3.38 (td, J = 11.9, 2.1 Hz, 2H), 2.23 (m, 1H), 1.55 (m, 2H), 1.44 (m, 2H). 13C NMR (150 MHz, CDCl3) δ 169.3, 147.5, 127.4, 81.4, 80.2, 67.1, 56.2, 36.1, 30.1. HRMS (EI) calc'd for C11H13N3O2 [M˙]+: 219.1008, found: 219.1008.4-((4-(1-Hydroxy-3-(4-(methylsulfonyl)phenyl)prop-2-yn-1-yl)-1H-1,2,3-triazol-1-yl)methyl)benzonitrile (13)
To a solution of 8P (0.03 g, 0.06 mmol, 1.0 eq.) in MeOH (2 mL) at R.T. was added CeCl3·7H2O (0.05 g, 0.13, 2.0 eq.) and the mixture was stirred for 10 min before the addition of NaBH4 (0.005 g, 0.128 mmol, 2.0 eq.) and stirring for another 1 h. The reaction mixture was then concentrated in vacuo, diluted with EtOAc (20 mL) and washed with 1 M HCl (2 × 10 mL) and brine (20 mL). The organic layer was then dried over MgSO4 and concentrated in vacuo to give a yellow solid which was purified by preparative thin layer chromatography (2.5% MeOH/DCM Rf = 0.13, developed 3×) to give a white powder (0.02 g, 60%, mixture of enantiomers).531H NMR (500 MHz, CDCl3) δ 7.93 (d, J = 8.7 Hz, 2H), 7.77–7.68 (m, 3H), 7.66 (d, J = 8.7 Hz, 2H), 7.42 (d, J = 8.4 Hz, 2H), 5.99 (s, 1H), 5.67 (s, 2H), 3.20 (s, 1H), 3.10 (s, 3H). 13C NMR (125 MHz, CDCl3) δ 148.4, 140.4, 139.3, 133.0, 132.6, 128.5, 127.8, 127.4, 121.7, 118.0, 113.0, 90.8, 84.0, 57.7, 53.6, 44.4. HRMS (ESI) calc'd for C20H16N4O3SNa [M + Na]+: 415.0837, found: 415.0841.Ethyl 3-(4-(methylsulfonyl)phenyl)propiolate (14)
In a flame dried round bottom flask filled with N2 were added 1G (0.50 g, 1.77 mmol, 1.0 eq.), PdCl2(PPh3)2 (0.03 g, 0.04 mmol, 2 mol%), CuI (0.01, 0.07 mmol, 4 mol%), and K2CO3 (0.50 g, 3.54 mmol, 2.0 eq.). The solids were dissolved in 5 mL of anhydrous THF followed by the addition of ethyl propiolate (0.72 mL, 7.08 mmol, 4.0 eq.). The reaction was heated to reflux and stirred for 24 h. The reaction was concentrated in vacuo and then diluted with H2O (100 mL) and EtOAc (50 mL) and filtered through celite. The layers of the filtrate were separated, and the aqueous layer was extract with EtOAc (2 × 50 mL). The combined organic layer was washed brine (100 mL) followed by drying over MgSO4 and concentrating in vacuo to give a brown solid. The residue was purified by flash column chromatography (40% EtOAc/Hex, Rf = 0.41, dry loading using celite) to give a brown solid (0.35 g, 77%). 1H NMR (600 MHz, CDCl3) δ 7.96 (d, J = 8.7 Hz, 2H), 7.76 (d, J = 8.7 Hz, 2H), 4.32 (q, J = 7.2 Hz, 2H), 3.07 (s, 3H), 1.37 (t, J = 7.2 Hz, 3H). 13C NMR (150 MHz, CDCl3) δ 153.3, 141.9, 133.5, 127.6, 125.4, 83.3, 83.0, 62.5, 44.3, 14.0. HRMS (EI) calc'd for C12H12O4S [M˙]+: 252.0456, found: 252.0435.3-(4-(Methylsulfonyl)phenyl)propiolic acid (15)
To a solution of 14 (0.33 g, 1.31 mmol, 1.0 eq.) in acetone/H2O (5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 mL) was added LiOH·H2O (0.22 g, 5.24 mmol, 4.0 eq.). The reaction was stirred for 4 h, concentrated in vacuo, and then diluted with sat. Na2CO3 (20 mL). The aqueous layer was washed with DCM (3 × 10 mL), acidified to pH ≈ 1, and extracted with EtOAc ((3 × 20 mL), The combined organic layer was washed brine (50 mL) followed by drying over MgSO4 and concentrating in vacuo to give a brown solid that was carried forward without further purification (40% EtOAc/Hex, Rf = 0) (0.22 g, >95%). 1H NMR (500 MHz, DMSO-d6) δ 8.01 (d, J = 8.7 Hz, 2H), 7.90 (d, J = 8.7 Hz, 2H), 3.29 (s, 3H). 13C NMR (125 MHz, DMSO-d6) δ 153.9, 142.2, 133.3, 127.4, 124.1, 83.9, 82.1, 43.1. HRMS (EI) calc'd for C10H8O4S [M˙]+: 224.0143, found: 224.0118.
1 mL) was added LiOH·H2O (0.22 g, 5.24 mmol, 4.0 eq.). The reaction was stirred for 4 h, concentrated in vacuo, and then diluted with sat. Na2CO3 (20 mL). The aqueous layer was washed with DCM (3 × 10 mL), acidified to pH ≈ 1, and extracted with EtOAc ((3 × 20 mL), The combined organic layer was washed brine (50 mL) followed by drying over MgSO4 and concentrating in vacuo to give a brown solid that was carried forward without further purification (40% EtOAc/Hex, Rf = 0) (0.22 g, >95%). 1H NMR (500 MHz, DMSO-d6) δ 8.01 (d, J = 8.7 Hz, 2H), 7.90 (d, J = 8.7 Hz, 2H), 3.29 (s, 3H). 13C NMR (125 MHz, DMSO-d6) δ 153.9, 142.2, 133.3, 127.4, 124.1, 83.9, 82.1, 43.1. HRMS (EI) calc'd for C10H8O4S [M˙]+: 224.0143, found: 224.0118.
1-(1H-Benzo[d][1,2,3]triazol-1-yl)-3-(4-(methylsulfonyl)phenyl)prop-2-yn-1-one (16)
To a solution of benzotriazole (0.21 g, 1.80 mmol, 4.0 eq.) in anhydrous DCM (3 mL) was added SOCl2 (0.03 mL, 0.45 mmol, 1.0 eq.) and the mixture was stirred for 30 min at R.T. followed by the addition of 15 (0.10 g, 0.45 mmol, 1.0 eq.). The reaction mixture was stirred for 3 h and then diluted with EtOAc (20 mL), filtered over celite, and washed with 2 M NaOH (3 × 10 mL). The organic layer was concentrated in vacuo to give a yellow solid which was washed with Et2O to give a white powder (0.04 g, 51%) (50% EtOAc/Hex Rf = 0.51). 1H NMR (400 MHz, CDCl3) δ 8.31 (d, J = 8.2 Hz, 1H), 8.18 (d, J = 8.2 Hz, 1H), 8.06 (d, J = 8.7 Hz, 2H), 7.98 (d, J = 8.7 Hz, 2H), 7.73 (t, J = 8.2 Hz, 1H), 7.58 (t, J = 8.2 Hz, 1H), 3.11 (s, 3H). 13C NMR (100 MHz, CDCl3) δ 149.9, 146.5, 142.9, 134.3, 131.0, 130.9, 127.9, 127.1, 124.8, 120.7, 114.3, 92.5, 83.4, 44.5. HRMS (ESI) calc'd for C16H11N3O3S [M + Na]+: 348.0419, found: 348.0389.3-(4-(Methylsulfonyl)phenyl)-N-(prop-2-yn-1-yl)propiolamide (17)
To a solution of 15 (0.10 g, 0.45 mmol, 1.1 eq.) and propargyl amine (0.03 mL, 0.41 mmol, 1.0 eq.) in anhydrous DCM (3 mL) under N2 was added a solution of DCC (0.09 g, 0.25 mol, 1.1 eq.) and DMAP (0.001 g, 0.008 mmol, 2 mol%) in anhydrous DCM (2 mL). The reaction was stirred at R.T. for 3 h and then diluted EtOAc (20 mL) and filtered over celite. The organic layer was washed with 5% AcOH (3 × 10 mL), brine (20 mL), sat. NaHCO3 (3 × 10 mL), and then brine (20 mL) again. The combined organic layer was dried over MgSO4 and concentrated in vacuo to give a brown solid that was purified by flash column chromatography (50% EtOAc/Hex, Rf = 0.18) to give a pale-yellow solid (0.05 g, 42%). 1H NMR (500 MHz, CDCl3 + CD3OD) δ 7.93 (d, J = 6.5 Hz, 2H), 7.72 (d, J = 6.5 Hz, 2H), 4.14–4.00 (m, 1H), 3.47–3.37 (m, 2H), 3.13 (s, 4H), 1.69–1.66 (m, 1H). 13C NMR (125 MHz, CDCl3 + CD3OD) δ 152.5, 141.3, 133.2, 127.5, 125.9, 85.2, 82.7, 78.3, 71.9, 44.2, 33.7. HRMS (EI) calc'd for C13H11O3S [M˙]+: 261.0460, found: 261.0466.3-(4-(Methylsulfonyl)phenyl)-N-((1-((tetrahydro-2H-pyran-4-yl)methyl)-1H-1,2,3-triazol-4-yl)methyl)propiolamide (18)
To a solution of 17 (0.05 g, 0.19 mmol, 1.0 eq.) and 7D (0.03 g, 0.19 mmol, 1.0 eq.) in tBuOH/H2O (2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 mL) were added CuSO4·5H2O (0.003 g, 0.009 mmol, 5 mol%) and sodium ascorbate (0.004 g, 0.019 mmol, 10 mol%). The reaction mixture was stirred vigorously for 16 h and was then concentrated in vacuo to give a yellow solid, which was purified by flash column chromatography (2% MeOH/EtOAc Rf = 0.23, dry loading using celite) to give a white powder (0.04 g, 51%). 1H NMR (600 MHz, CDCl3) δ 7.98 (d, J = 8.6 Hz, 2H), 7.74 (d, J = 8.6 Hz, 2H), 7.64 (s, 1H), 6.97 (s, 1H), 4.67 (d, J = 5.7 Hz, 2H), 4.28 (d, J = 7.1 Hz, 2H), 4.02 (dd, J = 11.4, 2.9 Hz, 2H), 3.40 (td, J = 11.8, 2.3 Hz, 2H), 3.11 (s, 3H), 2.26–2.16 (m, 1H), 1.58–1.54 (m, 2H), 1.49–1.39 (m, 2H). 13C NMR (150 MHz, CDCl3) δ 152.6, 143.4, 141.6, 133.3, 127.6, 125.8, 122.9, 85.4, 82.7, 67.1, 55.9, 44.3, 36.2, 35.3, 30.3. HRMS (ESI) calc'd for C19H22N4O4SNa [M + Na]+: 425.1259, found: 425.1241.
2 mL) were added CuSO4·5H2O (0.003 g, 0.009 mmol, 5 mol%) and sodium ascorbate (0.004 g, 0.019 mmol, 10 mol%). The reaction mixture was stirred vigorously for 16 h and was then concentrated in vacuo to give a yellow solid, which was purified by flash column chromatography (2% MeOH/EtOAc Rf = 0.23, dry loading using celite) to give a white powder (0.04 g, 51%). 1H NMR (600 MHz, CDCl3) δ 7.98 (d, J = 8.6 Hz, 2H), 7.74 (d, J = 8.6 Hz, 2H), 7.64 (s, 1H), 6.97 (s, 1H), 4.67 (d, J = 5.7 Hz, 2H), 4.28 (d, J = 7.1 Hz, 2H), 4.02 (dd, J = 11.4, 2.9 Hz, 2H), 3.40 (td, J = 11.8, 2.3 Hz, 2H), 3.11 (s, 3H), 2.26–2.16 (m, 1H), 1.58–1.54 (m, 2H), 1.49–1.39 (m, 2H). 13C NMR (150 MHz, CDCl3) δ 152.6, 143.4, 141.6, 133.3, 127.6, 125.8, 122.9, 85.4, 82.7, 67.1, 55.9, 44.3, 36.2, 35.3, 30.3. HRMS (ESI) calc'd for C19H22N4O4SNa [M + Na]+: 425.1259, found: 425.1241.
Ethyl 1-(4-cyanobenzyl)-1H-1,2,3-triazole-4-carboxylate (19A)
To a solution of 7G (0.20 g, 1.26 mmol, 1.0 eq.), ethyl propiolate (0.13 mL, 1.26 mmol, 1.0 eq.), and CuI (0.24 g, 1.26 mmol, 1.0 eq.) in anhydrous MeCN (10 mL) under N2 at R.T. was added DIPEA (0.22 mL, 1.26 mmol, 1.0 eq) dropwise and the reaction was stirred for 16 h. The reaction mixture was then diluted with DCM (20 mL) and washed with 1 M HCl (2 × 15 mL), brine (20 mL), sat. NH4OH (4 × 15 mL, until the aqueous phase was no longer blue), and brine (20 mL) again. The organic layer was then dried over MgSO4 and concentrated in vacuo to give a brown solid that was purified by flash column chromatography (10% EtOAc/DCM Rf = 0.47, dry loading using celite) to give the title compound as white fluffy needles (0.35 g, 78%). 1H NMR (500 MHz, CDCl3) δ 8.05 (s, 1H), 7.69 (d, J = 8.4 Hz, 2H), 7.37 (d, J = 8.4 Hz, 2H), 5.65 (s, 2H), 4.41 (q, J = 7.1 Hz, 2H), 1.39 (t, J = 7.1 Hz, 3H). 13C NMR (125 MHz, CDCl3) δ 160.6, 141.2, 139.0, 133.2, 128.7, 127.6, 118.1, 113.4, 77.4, 77.2, 76.9, 61.6, 53.8, 14.4. HRMS (ESI) calc'd for C13H12N4O2Na [M + Na]+: 279.0858, found: 279.0828.1-(4-Cyanobenzyl)-1H-1,2,3-triazole-4-carboxylic acid (20A)
To a solution of 19A (0.10 g, 0.40 mmol, 1.0 eq.) in acetone/H2O (5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 mL) was added LiOH·H2O (0.07 g, 1.60 mmol, 4.0 eq.). The reaction was stirred for 4 h, concentrated in vacuo, and then diluted with sat. Na2CO3 (10 mL). The aqueous layer was washed with DCM (3 × 10 mL), acidified to pH ≈ 1, and extracted with EtOAc (3 × 20 mL). The combined organic layer was washed brine (20 mL) followed by drying over MgSO4 and concentrating in vacuo to give a white powder that was carried forward without further purification (50% EtOAc/Hex, Rf = 0) (0.08 g, 88%). 1H NMR (500 MHz, DMSO-d6) δ 8.82 (s, 1H), 7.86 (d, J = 8.7 Hz, 2H), 7.49 (d, J = 8.7 Hz, 2H), 5.77 (s, 2H). 13C NMR (125 MHz, DMSO-d6) δ 162.0, 141.4, 140.5, 129.9, 129.3, 119.0, 111.5, 52.9. HRMS (ESI) calc'd for C11H8N4O2Na [M + Na]+: 251.0545, found: 251.0557.
1 mL) was added LiOH·H2O (0.07 g, 1.60 mmol, 4.0 eq.). The reaction was stirred for 4 h, concentrated in vacuo, and then diluted with sat. Na2CO3 (10 mL). The aqueous layer was washed with DCM (3 × 10 mL), acidified to pH ≈ 1, and extracted with EtOAc (3 × 20 mL). The combined organic layer was washed brine (20 mL) followed by drying over MgSO4 and concentrating in vacuo to give a white powder that was carried forward without further purification (50% EtOAc/Hex, Rf = 0) (0.08 g, 88%). 1H NMR (500 MHz, DMSO-d6) δ 8.82 (s, 1H), 7.86 (d, J = 8.7 Hz, 2H), 7.49 (d, J = 8.7 Hz, 2H), 5.77 (s, 2H). 13C NMR (125 MHz, DMSO-d6) δ 162.0, 141.4, 140.5, 129.9, 129.3, 119.0, 111.5, 52.9. HRMS (ESI) calc'd for C11H8N4O2Na [M + Na]+: 251.0545, found: 251.0557.
tert-Butyl (1-(4-cyanobenzyl)-1H-1,2,3-triazol-4-yl)carbamate (21A)
To a suspension of 20A (0.83 g, 3.65 mmol, 1.0 eq.) in tBuOH (20 mL) were added NEt3 (0.63 mL, 4.40 mmol, 1.2 eq.) and DPPA (0.95 mL, 4.40 mmol, 1.2 eq.). The reaction was stirred for 24 h at 90 °C and then concentrated in vacuo. The crude residue was purified by flash column chromatography (70% EtOAc/Hex Rf = 0.71, dry loading using celite) to give a white powder (0.41 g, 38%). 1H NMR (500 MHz, CDCl3) δ 7.60 (s, 1H), 7.56 (d, J = 8.3 Hz, 2H), 7.25 (d, J = 8.3 Hz, 2H), 7.19 (s, 1H), 5.41 (s, 2H), 1.39 (s, 9H). 13C NMR (125 MHz, CDCl3) δ 152.1, 145.0, 139.7, 132.9, 128.5, 118.2, 112.9, 111.6, 81.5, 53.9, 28.2. HRMS (ESI) calc'd for C15H17N5O2Na [M + Na]+: 322.1280, found: 322.1304.4-((4-Amino-1H-1,2,3-triazol-1-yl)methyl)benzonitrile hydrochloride (22A)
A solution of 21A (0.20 g, 0.67 mmol) in DCM![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4 M HCl in Dioxanes (2 mL
4 M HCl in Dioxanes (2 mL![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 mL) was stirred at R.T. for 16 h. The reaction was concentrated, and the residue was washed with Et2O to give a yellow solid (0.13 g, >95%) which was used without further purification. 1H NMR (500 MHz, DMSO-d6) δ 8.27 (s, 1H), 7.88 (d, J = 8.3 Hz, 2H), 7.50 (d, J = 8.3 Hz, 2H), 5.75 (s, 2H). 13C NMR (125 MHz, DMSO-d6) δ 141.0, 139.7, 132.8, 128.8, 118.5, 117.3, 111.1, 52.8. HRMS (ESI) calc'd for C10H10N5 [M]+: 200.0936, found: 200.0925.
2 mL) was stirred at R.T. for 16 h. The reaction was concentrated, and the residue was washed with Et2O to give a yellow solid (0.13 g, >95%) which was used without further purification. 1H NMR (500 MHz, DMSO-d6) δ 8.27 (s, 1H), 7.88 (d, J = 8.3 Hz, 2H), 7.50 (d, J = 8.3 Hz, 2H), 5.75 (s, 2H). 13C NMR (125 MHz, DMSO-d6) δ 141.0, 139.7, 132.8, 128.8, 118.5, 117.3, 111.1, 52.8. HRMS (ESI) calc'd for C10H10N5 [M]+: 200.0936, found: 200.0925.
Ethyl 1-((tetrahydro-2H-pyran-4-yl)methyl)-1H-1,2,3-triazole-4-carboxylate (19B)
To a solution of 7D (0.40 g, 2.83 mmol, 1.0 eq.), ethyl propiolate (0.30 mL, 2.83 mmol, 1.0 eq.), and CuI (0.54 g, 2.83 mmol, 1.0 eq.) in anhydrous MeCN (10 mL) under N2 at R.T. was added DIPEA (0.50 mL, 2.83 mmol, 1.0 eq.) dropwise and the reaction was stirred for 16 h. The reaction mixture was then diluted with DCM (50 mL) and washed with 1 M HCl (2 × 25 mL), brine (25 mL), sat. NH4OH (4 × 25 mL, until aqueous phase was no longer blue), and brine (25 mL) again. The organic layer was then dried over MgSO4 and concentrated in vacuo to give an orange solid which was washed with Et2O to give the title compound as pale-orange powder (0.50 g, 73%) that was carried forward without further purification. 1H NMR (500 MHz, CDCl3) δ 8.05 (s, 1H), 4.42 (q, J = 7.1 Hz, 2H), 4.29 (d, J = 7.1 Hz, 2H), 4.04–3.90 (m, 2H), 3.35 (td, J = 11.7, 2.4 Hz, 2H), 2.19 (m, 1H), 1.61–1.14 (m, 7H). 13C NMR (125 MHz, CDCl3) δ 160.9, 140.4, 127.9, 67.3, 61.5, 56.2, 36.3, 30.3, 14.5. HRMS (ESI) calc'd for C11H17N3O3Na [M + Na]+: 262.1184, found: 262.1168.1-((Tetrahydro-2H-pyran-4-yl)methyl)-1H-1,2,3-triazole-4-carboxylic acid (20B)
To a solution of 19B (0.50 g, 2.09 mmol, 1.0 eq.) in acetone/H2O (20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 mL) was added LiOH·H2O (0.35 g, 8.36 mmol, 4.0 eq.). The reaction was stirred for 4 h, concentrated in vacuo, and then diluted with sat. Na2CO3 (25 mL). The aqueous layer was washed with DCM (3 × 25 mL), acidified to pH ≈ 1, and extracted with EtOAc (3 × 25 mL). The combined organic layer was washed with brine (25 mL), dried over MgSO4, and concentrated in vacuo to give a white powder that was carried forward without further purification (50% EtOAc/Hex, Rf = 0) (0.21 g, 50%). 1H NMR (500 MHz, DMSO-d6) δ 8.68 (s, 1H), 4.32 (d, J = 7.2 Hz, 2H), 3.82 (dd, J = 11.5, 2.6 Hz, 1H), 3.24 (td, J = 11.5, 2.6 Hz, 2H), 2.10 (m,1H), 1.62–0.96 (m, 4H). 13C NMR (125 MHz, DMSO-d6) δ 162.2, 140.0, 129.8, 66.8, 55.2, 35.8, 30.1. HRMS (ESI) calc'd for C9H13N3O2Na [M + Na]+: 234.0837, found: 234.0855.
5 mL) was added LiOH·H2O (0.35 g, 8.36 mmol, 4.0 eq.). The reaction was stirred for 4 h, concentrated in vacuo, and then diluted with sat. Na2CO3 (25 mL). The aqueous layer was washed with DCM (3 × 25 mL), acidified to pH ≈ 1, and extracted with EtOAc (3 × 25 mL). The combined organic layer was washed with brine (25 mL), dried over MgSO4, and concentrated in vacuo to give a white powder that was carried forward without further purification (50% EtOAc/Hex, Rf = 0) (0.21 g, 50%). 1H NMR (500 MHz, DMSO-d6) δ 8.68 (s, 1H), 4.32 (d, J = 7.2 Hz, 2H), 3.82 (dd, J = 11.5, 2.6 Hz, 1H), 3.24 (td, J = 11.5, 2.6 Hz, 2H), 2.10 (m,1H), 1.62–0.96 (m, 4H). 13C NMR (125 MHz, DMSO-d6) δ 162.2, 140.0, 129.8, 66.8, 55.2, 35.8, 30.1. HRMS (ESI) calc'd for C9H13N3O2Na [M + Na]+: 234.0837, found: 234.0855.
tert-Butyl (1-((tetrahydro-2H-pyran-4-yl)methyl)-1H-1,2,3-triazol-4-yl)carbamate (21B)
To a suspension of 20B (0.90 g, 4.26 mmol, 1.0 eq.) in tBuOH (20 mL) were added NEt3 (0.90 mL, 6.39 mmol, 1.2 eq.) and DPPA (1.40 mL, 6.39 mmol, 1.2 eq.). The reaction was stirred for 24 h at 90 °C and then concentrated in vacuo. The crude residue was purified by flash column chromatography (80% EtOAc/Hex Rf = 0.48, dry loading using celite) to give a white powder (0.59 g, 49%). 1H NMR (600 MHz, CDCl3) δ 7.71 (s, 1H), 7.15 (s, 1H), 4.17 (d, J = 7.2 Hz, 2H), 3.97 (apparent dd, J = 9.4, 4.6 Hz, 2H), 3.36 (td, J = 11.8, 2.2 Hz, 2H), 2.16 (m, 2H), 1.57–1.44 (m, 11H), 1.40 (m, 2H). 13C NMR (150 MHz, CDCl3) δ 152.2, 144.2, 112.0, 81.3, 67.3, 56.3, 36.2, 30.3, 28.3. HRMS (ESI) calc'd for C13H22N4O3Na [M + Na]+: 305.1590, found: 305.1595.1-((Tetrahydro-2H-pyran-4-yl)methyl)-1H-1,2,3-triazol-4-amine hydrochloride (22B)
A solution of 21B (0.25 g, 0.89 mmol) in DCM![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4 M HCl in dioxanes (3 mL
4 M HCl in dioxanes (3 mL![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 mL) was stirred at R.T. for 16 h. The reaction was concentrated, and the residue was washed with Et2O to give a white powder (0.20 g, >95%) which was used without further purification. 1H NMR (500 MHz, DMSO-d6) δ 8.11 (s, 1H), 4.30 (d, J = 7.1 Hz, 2H), 3.91–3.74 (m, 2H), 3.26 (td, J = 11.5, 2.5, 2H), 2.09 (m, 1H), 1.44–1.37 (m, 2H), 1.34–1.11 (m, 2H). 13C NMR (125 MHz, DMSO-d6) δ 139.8, 117.5, 66.8, 55.7, 35.9, 30.1. HRMS (ESI) calc'd for C8H15N4 [M]+: 183.1246, found: 183.1248.
3 mL) was stirred at R.T. for 16 h. The reaction was concentrated, and the residue was washed with Et2O to give a white powder (0.20 g, >95%) which was used without further purification. 1H NMR (500 MHz, DMSO-d6) δ 8.11 (s, 1H), 4.30 (d, J = 7.1 Hz, 2H), 3.91–3.74 (m, 2H), 3.26 (td, J = 11.5, 2.5, 2H), 2.09 (m, 1H), 1.44–1.37 (m, 2H), 1.34–1.11 (m, 2H). 13C NMR (125 MHz, DMSO-d6) δ 139.8, 117.5, 66.8, 55.7, 35.9, 30.1. HRMS (ESI) calc'd for C8H15N4 [M]+: 183.1246, found: 183.1248.
General procedure 6 (GP6): amide coupling with TCFH and NMI30
To a solution of propiolic acids (1.0 eq., commercially available unless indicated otherwise), 22A or 22B (1.3 eq.), and NMI (3.5 eq.) in anhydrous MeCN (∼0.1 M relative to acid) at R.T. was added TCFH (1.2 eq.). The reaction was stirred at R.T. for 3 h and then diluted with EtOAc (20 mL). The organic layer was washed with 5% AcOH (3 × 10 mL), brine (20 mL), sat. NaHCO3 (3 × 10 mL), and brine again (20 mL). The organic layer was then dried over MgSO4 and concentrated in vacuo. The crude residue was purified by flash column chromatography as described below.N-(1-(4-Cyanobenzyl)-1H-1,2,3-triazol-4-yl)-3-(4-(methylsulfonyl)phenyl)propiolamide (23A)
Synthesized according to GP6 using 15 (0.08 g, 0.39 mmol) and 22A (0.12 g, 0.50 mmol), and purified by flash column chromatography (40% EtOAc/Hex Rf = 0.40, dry loading using celite) to give a white powder (0.06 g, 36%). 1H NMR (500 MHz, DMSO-d6) δ 12.08 (s, 1H), 8.39 (s, 1H), 8.03 (d, J = 8.5 Hz, 2H), 7.91 (d, J = 8.5 Hz, 2H), 7.86 (d, J = 8.4 Hz, 2H), 7.49 (d, J = 8.4 Hz, 2H), 5.72 (s, 2H), 3.28 (s, 3H). 13C NMR (125 MHz, DMSO-d6) δ 148.8, 142.6, 142.1, 141.9, 141.4, 133.3, 142.8, 128.8, 127.5, 124.6, 118.6, 115.3, 110.9, 85.4, 83.4, 52.5, 43.2. HRMS (ESI) calc'd for C20H15N5O3Na [M + Na]+: 428.0793, found: 428.0811.3-(4-(Methylsulfonyl)phenyl)-N-(1-((tetrahydro-2H-pyran-4-yl)methyl)-1H-1,2,3-triazol-4-yl)propiolamide (23B)
Synthesized according to GP6 using 15 (0.16 g, 0.70 mmol) and 22B (0.20 g, 0.91 mmol), and the resulting residue was suspended in MeOH (10 mL) and DCM (1 mL) and then filtered to give the title compound as a pale-yellow solid which was used without further purification (0.02 g, 7%) (50% EtOAc/Hex Rf = 0.59). 1H NMR (600 MHz, CDCl3) δ 11.98 (s, 1H), 8.21 (s, 1H), 8.00 (d, J = 8.7 Hz, 2H), 7.87 (d, J = 8.7 Hz, 1H), 4.24 (d, J = 7.1 Hz, 2H), 3.79 (apparent d, J = 11.5 Hz, 3H), 3.25–3.17 (m, 5H), 2.10–2.02 (m, 1H), 1.53–1.12 (m, 4H). 13C NMR (150 MHz, CDCl3) δ 149.2, 142.7, 142.6, 133.7, 128.0, 125.1, 115.8, 86.0, 83.8, 66.9, 55.4, 55.4, 43.7, 36.0, 30.2. HRMS (ESI) calc'd for C18H20N4O4Na [M + Na]+: 411.1103, found: 411.1108.3-Phenyl-N-(1-((tetrahydro-2H-pyran-4-yl)methyl)-1H-1,2,3-triazol-4-yl)propiolamide (23C)
Synthesized according to GP6 using phenylpropiolic acid (0.07 g, 0.46 mmol) and 22B (0.12 g, 0.55 mmol), and the resulting residue was suspended in MeOH (5 mL) and DCM (0.5 mL) and then filtered to give the title compound as a pale-yellow solid which was used without further purification (0.03 g, 21%) (50% EtOAc/Hex Rf = 0.65). 1H NMR (600 MHz, DMSO-d6) δ 8.21 (s, 1H), 7.80–7.52 (m, 2H), 7.60–7.23 (m, 3H), 4.27 (d, J = 7.1 Hz, 2H), 3.83 (apparent dd, J = 12.0, 4.1 Hz, 2H), 3.25 (td, J = 11.7, 2.3 Hz, 2H), 2.09 (ddd, J = 11.3, 7.4, 4.0 Hz, 1H), 1.49–1.05 (m, 4H). 13C NMR (150 MHz, CDCl3) δ 149.7, 142.9, 132.9, 131.2, 129.5, 120.0, 115.6, 85.8, 83.8, 66.9, 55.4, 36.0, 30.2. HRMS (ESI) calc'd for C17H18N4O2Na [M + Na]+: 333.1327, found: 333.1337.N-(1-((Tetrahydro-2H-pyran-4-yl)methyl)-1H-1,2,3-triazol-4-yl)but-2-ynamide (23D)
Synthesized according to GP6 using 2-butynoic acid (0.02 g, 0.27 mmol) and 22B (0.07 g, 0.35 mmol), and purified by flash column chromatography (50% EtOAc/Hex Rf = 0.54, dry loading using celite) to give a white powder (0.02 g, 31%). 1H NMR (300 MHz, CDCl3) δ 8.70 (s, 1H), 7.97 (s, 1H), 4.20 (d, J = 7.2 Hz, 2H), 3.98 (apparent dd, J = 12.2, 4.3 Hz, 2H), 3.36 (td, J = 11.7, 2.4 Hz, 2H), 2.16 (ddd, J = 11.4, 7.9, 4.0 Hz, 1H), 1.53–1.30 (m, 4H). 13C NMR (150 MHz, CDCl3) δ 149.8, 142.8, 114.3, 86.4, 74.4, 67.4, 56.5, 36.3, 30.4, 4.0. HRMS (ESI) calc'd for C12H16N4O2Na [M + Na]+: 271.1171, found: 271.1192.N-(1-((Tetrahydro-2H-pyran-4-yl)methyl)-1H-1,2,3-triazol-4-yl)propiolamide (23E)
Synthesized according to GP6 using propiolic acid (0.02 mL, 0.30 mmol) and 22B (0.08 g, 0.37 mmol), and purified by flash column chromatography (50% EtOAc/Hex Rf = 0.51, dry loading using celite) to give a pale-yellow solid (0.03 g, 43%). 1H NMR (600 MHz, CDCl3 + CD3OD) δ 7.95 (s, 1H), 4.15 (d, J = 7.2 Hz, 3H), 3.97–3.82 (m, 2H), 3.33–3.29 (m, 2H, overlapping with residual methanol), 3.07 (s, 1H), 2.09 (ddd, J = 11.1, 7.4, 3.8 Hz, 1H), 1.49–1.41 (m, 2H), 1.41–1.27 (m, 2H). 13C NMR (150 MHz, CDCl3 + CD3OD) δ 149.1, 142.5, 114.8, 76.4, 75.5, 75.5, 67.1, 56.1, 35.9, 30.0. HRMS (ESI) calc'd for C11H14N4O2Na [M + Na]+: 257.1014, found: 257.1034.tert-Butyl 4-(4-nitrobenzoyl)piperazine-1-carboxylate (24)
4-Nitrobenzoic acid (0.49 g, 2.95 mmol, 1.1 eq.), HBTU (1.32 g, 3.48 mmol, 1.3 eq.), and DIPEA (1.40 mL, 8.04 mmol, 3.0 eq.) were stirred in DCM (25 mL) at R.T. for 15 min, after which N-Boc-piperazine (0.50 g, 2.68 mmol, 1.0 eq.) was added. The reaction was stirred for 3 h and then diluted with EtOAc (100 mL). The organic layer was washed with 5% AcOH (3 × 25 mL), brine (50 mL), sat. NaHCO3 (50 mL), and brine (50 mL) again, and then dried over MgSO4 and concentrated in vacuo to give a yellow solid (0.90 g, >95%), which was used without further purification. 1H NMR (300 MHz, CDCl3) δ 8.29 (d, J = 8.9 Hz, 2H), 7.57 (d, J = 8.9 Hz, 2H), 3.76–3.35 (m, 8H), 1.46 (s, 9H). 13C NMR (75 MHz, CDCl3) δ 167.3, 153.8, 147.8, 142.0, 128.3, 123.7, 79.2, 46.7 (2C), 41.4 (2C), 28.0. Characterization data are consistent with previously reported values.54(4-Nitrophenyl)(piperazin-1-yl)methanone hydrochloride (25)
To a solution of 24 (0.50 g, 1.50 mmol, 1.0 eq.) in DCM (5 mL) was added 4 M HCl/dioxane (5 mL) and the reaction was stirred for 3 h at R.T. Upon completion, the reaction was concentrated in vacuo and the resulting solid was washed with DCM (3 × 10 mL) to give a white solid (0.37 g, 93%). 1H NMR (300 MHz, DMSO-d6) δ 9.41 (br s, 2H), 8.31 (d, J = 8.9 Hz, 2H), 7.75 (d, J = 8.9 Hz, 2H), 3.85 (apparent br s, 2H), 3.50 (apparent br s, 2H), 3.18–3.12 (m, 4H). 13C NMR (75 MHz, DMSO-d6) δ 168.2, 148.5, 142.2, 128.2, 124.1, 49.0, 46.6, 46.0, 43.5. Characterization data are consistent with previously reported values.54tert-Butyl (2-(4-(4-nitrobenzoyl)piperazin-1-yl)-2-oxoethyl)carbamate (26)
N-Boc-glycine (0.25 g, 1.43 mmol, 1.1 eq.), HBTU (0.64 g, 1.69 mmol, 1.3 eq.), and DIPEA (0.90 mL, 5.20 mmol, 3.0 eq.) were stirred in DCM (15 mL) at R.T. for 15 min after which 25 (0.37 g, 1.30 mmol, 1.0 eq.) was added. The reaction was stirred for 3 h and then diluted with EtOAc (100 mL). The organic layer was washed with 5% AcOH (3 × 25 mL), brine (50 mL), sat. NaHCO3 (50 mL), and brine (50 mL) again, and then dried over MgSO4 and concentrated in vacuo. The residue was washed with hexanes (3 × 25 mL) to give a yellow solid (0.44 g, 85%), which was used without further purification. 1H NMR (300 MHz, CDCl3) δ 8.31 (d, J = 8.9 Hz, 2H), 7.59 (d, J = 8.9 Hz, 2H), 5.43 (br s, 1H), 3.99 (br s, 2H), 3.81–3.41 (m, 8H), 1.44 (s, 9H). 13C NMR (75 MHz, CDCl3) δ 168.4, 167.5, 155.9, 148.8, 141.1, 128.3, 124.2, 80.1, 47.3, 44.3, 42.3 (3C), 28.5. HRMS (ESI) calc'd for C18H24N4O6Na [M + Na]+: 415.1576, found: 415.1594.2-Amino-1-(4-(4-nitrobenzoyl)piperazin-1-yl)ethan-1-one (27)
26 (0.38 g, 0.97 mmol, 1.0 eq.) was dissolved in DCM (5.0 mL) and treated with TFA (1.00 mL, 5.90 mmol, 6.0 eq.) until the starting material was completely consumed by TLC (10% MeOH/DCM) in 3 h. The reaction mixture was diluted with DCM (15 mL) and washed with 0.5 M HCl (3 × 25 mL). The pH of the combined aqueous layer was adjusted to 9–10 using sat. K2CO3 and then extracted using DCM (3 × 35 mL). The combined organic layer was dried over MgSO4 and concentrated in vacuo to give a white solid (0.12 g, 44%) (10% MeOH/DCM, Rf = 0.27). 1H NMR (300 MHz, DMSO-d6) δ 8.29 (d, J = 8.9 Hz, 2H), 7.70 (d, J = 8.9 Hz, 2H), 3.61–3.27 (m, 19H). 13C NMR (75 MHz, DMSO-d6) δ 171.5, 167.3, 147.9, 142.0, 128.4, 123.8, 46.7, 43.3, 42.6 41.4. HRMS (EI) calc'd for C13H16N4O4 [M˙]+: 292.1172, found: 292.1175.N-(2-(4-(4-Nitrobenzoyl)piperazin-1-yl)-2-oxoethyl)acrylamide (28)
27 (0.12 g, 0.35 mmol, 1.0 eq.) and DIPEA (0.20 mL, 1.10 mmol, 3.0 eq.) were dissolved in anhydrous DCM (5.0 mL) and cooled to 0 °C. To this solution was added acryloyl chloride (0.0630 mL, 0.39 mmol, 1.1 eq.) and the reaction was stirred under nitrogen for 3 h. The reaction was concentrated and purified by flash column chromatography (5% MeOH/EtOAc, Rf = 0.33) to give a white solid (0.12 g, 90%). 1H NMR (300 MHz, CDCl3) δ 8.31 (d, J = 8.9 Hz, 2H), 7.59 (d, J = 8.9 Hz, 2H), 6.67 (br s, 1H), 6.35–6.29 (m, 2H), 5.69 (dd, J = 9.9, 1.8 Hz, 1H), 4.18 (s, 2H), 3.81–3.45 (m, 8H). 13C NMR (75 MHz, CDCl3) δ 168.4, 167.0, 165.5, 148.8, 141.0, 130.3, 128.3, 127.3, 124.2, 47.2, 44.4, 42.2 (2C), 41.4. HRMS (ESI) calc'd for C16H18N4O5Na [M + Na]+: 369.1175, found: 369.1171.tert-Butyl (2-(methoxy(methyl)amino)-2-oxoethyl)carbamate (29)
To a solution of N-Boc-Gly (2.00 g, 11.42 mmol, 1.0 eq.), N,O-dimethylhydroxylamine hydrochloride (1.23 g, 12.56, 1.1 eq.), and DIPEA (8.00 mL, 45.68 mmol, 4.0 eq.) in DCM (50 mL) at 0 °C were added EDCI-HCl (2.41 g, 12.56 mmol, 1.1 eq.) and DMAP (0.14 g, 1.14 mmol, 0.1 eq.). The reaction was allowed to warm to R.T. and stirred for 24 h. The reaction was diluted with EtOAc (150 mL) and washed with 1 M HCl (50 mL), brine (50 mL), sat. NaHCO3 (2 × 50 mL), and brine again (50 mL). The organic layer was then dried over MgSO4 and concentrated in vacuo to give the product as a white solid (1.50 g, 60%). 1H NMR (300 MHz, CDCl3) δ 5.26 (br s, 1H), 4.08–4.09 (d, J = 5.1 Hz, 2H), 3.71 (s, 3H), 3.20 (s, 3H), 1.45 (s, 9H). 13C NMR (75 MHz, CDCl3) δ 170.3, 156.0, 79.6, 61.5, 41.7, 32.4, 28.3. Characterization data are consistent with previously reported values.55tert-Butyl (2-oxobut-3-yn-1-yl)carbamate (30)
In a flame-dried round-bottom flask flushed with nitrogen, 29 (0.97 g, 4.60 mmol, 1.0 eq.) was dissolved in in anhydrous THF (80 mL) and the solution was cooled to −78 °C. Ethynyl magnesium bromide (0.5 M in THF) (37.00 mL, 18.40 mmol, 4.0 eq.) was then added dropwise and the reaction was allowed to warm to R.T. over 16 h. The reaction was quenched with cold 1 M NaHSO4 (50 mL) and concentrated to removed majority of the THF. The aqueous layer was then extracted with EtOAc (3 × 100 mL). The combined organic layer was washed with sat. NaHCO3 (100 mL) and brine (100 mL), dried over MgSO4, and concentrated in vacuo to give an orange oil (0.83 g, >95%) which was used without further purification. 1H NMR (300 MHz, CDCl3) δ 5.16 (br s, 1H), 4.21–4.19 (d, J = 5.1 Hz, 2H), 3.38 (s, 1H), 1.48 (s, 9H). 13C NMR (75 MHz, CDCl3) δ 183.1, 155.6, 81.7, 80.3, 79.7, 52.2, 28.3. Characterization data are consistent with previously reported values.55tert-Butyl (2-(1-((adamantan-1-yl)methyl)-1H-1,2,3-triazol-4-yl)-2-oxoethyl)carbamate (31)
To a solution of 30 (0.50 g, 2.73 mmol, 1.0 eq.) and 7C (0.52 g, 2.73 mmol, 1.0 eq.) in 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 tBuOH/H2O (14 mL) were added freshly prepared aqueous solutions of CuSO4·5H2O (50 mg mL−1) (0.70 mL, 0.14 mmol, 0.05 eq.) and 1 M sodium ascorbate (0.55 mL, 0.55 mmol, 0.2 eq.). The reaction mixture was stirred vigorously overnight, at which point the reaction was still in complete by TLC. Another 0.2 mL of CuSO4·5H2O solution and 0.1 mL of sodium ascorbate solution were added, and the reaction was stirred for another 3 h, after which the reaction mixture was diluted with water (25 mL) and extracted with EtOAc (3 × 20 mL). The combined organic layer was dried over MgSO4 and concentrated in vacuo to give a yellow oil which was purified by flash column chromatography (dry loading with celite, 40% EtOAc/hexanes, Rf = 0.38) to give a white foam (0.46 g, 40%). 1H NMR (300 MHz, CDCl3) δ 8.01 (s, 1H), 5.32 (br s, 1H), 4.76–4.74 (d, J = 5.1 Hz, 2H), 4.07 (s, 2H), 2.00 (apparent s, 3H), 1.72–1.55 (m, 6H), 1.48 (apparent s, 6H), 1.46 (s, 9H). 13C NMR (75 MHz, CDCl3) δ 190.0, 155.9, 145.4, 127.0, 79.9, 62.6, 48.3, 40.2, 36.5, 34.3, 28.5, 28.1. HRMS (ESI) calc'd for C16H18N4O5Na [M + Na]+: 397.2216, found: 397.2203.
1 tBuOH/H2O (14 mL) were added freshly prepared aqueous solutions of CuSO4·5H2O (50 mg mL−1) (0.70 mL, 0.14 mmol, 0.05 eq.) and 1 M sodium ascorbate (0.55 mL, 0.55 mmol, 0.2 eq.). The reaction mixture was stirred vigorously overnight, at which point the reaction was still in complete by TLC. Another 0.2 mL of CuSO4·5H2O solution and 0.1 mL of sodium ascorbate solution were added, and the reaction was stirred for another 3 h, after which the reaction mixture was diluted with water (25 mL) and extracted with EtOAc (3 × 20 mL). The combined organic layer was dried over MgSO4 and concentrated in vacuo to give a yellow oil which was purified by flash column chromatography (dry loading with celite, 40% EtOAc/hexanes, Rf = 0.38) to give a white foam (0.46 g, 40%). 1H NMR (300 MHz, CDCl3) δ 8.01 (s, 1H), 5.32 (br s, 1H), 4.76–4.74 (d, J = 5.1 Hz, 2H), 4.07 (s, 2H), 2.00 (apparent s, 3H), 1.72–1.55 (m, 6H), 1.48 (apparent s, 6H), 1.46 (s, 9H). 13C NMR (75 MHz, CDCl3) δ 190.0, 155.9, 145.4, 127.0, 79.9, 62.6, 48.3, 40.2, 36.5, 34.3, 28.5, 28.1. HRMS (ESI) calc'd for C16H18N4O5Na [M + Na]+: 397.2216, found: 397.2203.
1-(1-((Adamantan-1-yl)methyl)-1H-1,2,3-triazol-4-yl)-2-aminoethan-1-one hydrochloride (32)
To a solution of 31 (0.24 g, 0.64 mmol, 1.0 eq.) in DCM (5 mL) was added 5 mL of HCl (4 M in dioxane). The reaction was stirred for 4 h at room temperature and then concentrated in vacuo to give an off-white solid which was washed with DCM (3 × 20 mL) and used without further purification (0.18 g, 89%). 1H NMR (300 MHz, CD3OD) δ 8.65 (s, 1H), 4.58 (s, 2H), 4.19 (s, 2H), 1.97 (apparent s, 3H), 1.73–1.61 (m, 6H), 1.55–1.54 (m, 6H). 13C NMR (75 MHz, CD3OD) δ 185.6, 128.5, 126.9, 61.7, 42.0, 39.8, 36.2, 33.8, 28.2. HRMS (ESI) calc'd for C15H23N4O [M]+: 275.1872, found: 275.1890.N-(2-(1-((Adamantan-1-yl)methyl)-1H-1,2,3-triazol-4-yl)-2-oxoethyl)acrylamide (33)
A solution of 32 (0.15 g, 0.48 mmol, 1.0 eq.) and DIPEA (0.25 mL, 1.44 mmol, 3.0 eq.) in anhydrous DCM (7 mL) was stirred at 0 °C until complete dissolution of the amine. Acryloyl chloride (0.04 mL, 0.53 mmol, 1.1 eq.) was then added dropwise and the reaction was allowed to stir at R.T. for 3 h and then concentrated in vacuo. The crude product was purified by flash column chromatography (dry loading using celite, 100% EtOAc, Rf = 0.58) to give a white solid (0.11 g, 69%). 1H NMR (300 MHz, CDCl3) δ 8.05 (s, 1H), 6.61 (apparent s, 1H), 6.36–6.18 (m, 2H), 5.68 (dd, J = 9.6, 1.5 Hz, 1H), 4.94 (d, J = 4.8 Hz, 1H), 4.07 (s, 2H), 1.99 (apparent s, 3H), 1.71–1.54 (m, 6H), 1.49 (apparent s, 6H). 13C NMR (75 MHz, CDCl3) δ 189.3, 165.6, 145.2, 130.5, 127.2, 127.1, 62.6, 47.3, 40.2, 36.5, 34.3, 28.1. HRMS (EI) calc'd for C18H24N4O2 [M˙]+: 328.1899, found: 328.1874.1-(4-(Adamantane-1-carbonyl)piperazin-1-yl)-3-(4-nitrophenyl)prop-2-yn-1-one (36)
To a stirring solution of 35, which was synthesized as previously described,20 (0.19 g, 0.58 mmol, 1.1 eq.) and 34, which was synthesized as previously described,24 (0.10 g, 0.52 mmol, 1.0 eq.) in anhydrous DCM (10 mL) was added HBTU (0.32 g, 0.84 mmol, 1.6 eq.) and Hünig's base (0.36 mL, 2.09 mmol, 4.0 eq.). The reaction mixture was stirred overnight at R.T, under N2. The reaction was confirmed complete by TLC (10% MeOH/DCM). The reaction mixture was concentrated in vacuo and the solid was resuspended in EtOAc (30 mL). The organic phase was washed with 5% AcOH (3 × 25 mL), brine (25 mL), NaHCO3 (25 mL) and then brine again (25 mL). The organic phase was dried over MgSO4 and concentrated in vacuo to give a yellow solid, which was purified by silica gel column chromatography (10% MeOH/DCM, Rf = 0.74, dry loading using celite), giving a pale-yellow solid (0.18 g, 80%). 1H NMR (300 MHz, CDCl3) δ 8.30–8.20 (d, J = 9.0 Hz, 2H), 7.77–7.67 (d, J = 9.0 Hz, 2H), 3.84–3.64 (m, 8H), 2.14–2.04 (m, 3H), 2.04–1.90 (m, 6H), 1.86–1.65 (m, 6H). 13C NMR (75 MHz, CDCl3) δ 176.2, 152.3, 148.3, 133.2, 126.9, 123.8, 88.5, 84.5, 47.3, 45.3, 42.1, 39.2, 36.5, 28.4. HRMS (ESI) calc'd for C24H27N3O4Na [M + Na]+: 444.1894, found: 444.1899.N-(2-(4-(Adamantane-1-carbonyl)piperazin-1-yl)-2-oxoethyl)-3-(4-nitrophenyl)propiolamide (38)
Compound 37, which was synthesized as previously described,20 (0.10 g, 0.33 mmol, 1.0 eq.) and 34 (0.07 g, 0.36 mmol, 1.1 eq.) were stirred in anhydrous DCM (2 mL) under N2 at 0 °C. A solution of DCC (0.07 g, 0.36 mmol, 1.1 eq.) and DMAP (0.001 g, 0.008 mmol, 2 mol%) in anhydrous DCM (2 mL) was added, the ice bath was removed and the reaction was stirred for 3 h. The mixture was filtered through celite and the filtrate was diluted with EtOAc (50 mL). The organic phase was washed with 5% AcOH (3 × 15 mL), brine (25 mL), NaHCO3 (25 mL), and brine again (25 mL). The organic phase was dried over MgSO4 and concentrated in vacuo to give a yellow solid, which was purified by silica gel column chromatography (8% MeOH/DCM, Rf = 0.42, dry loading using celite) (0.14 g, 88%). 1H NMR (300 MHz, CDCl3) δ 8.33–8.25 (d, J = 9.0 Hz, 2H), 7.78–7.72 (d, J = 9.0 Hz, 2H), 4.27–4.19 (d, J = 3.0 Hz, 2H), 3.87–3.66 (m, 6H), 3.57–3.39 (m, 2H), 2.15–2.08 (m, 3H), 2.06–2.00 (m, 6H), 1.86–1.67 (m, 6H) 13C NMR (75 MHz, CDCl3) δ 176.12, 165.7, 152.2, 148.3, 133.4, 126.7, 123.7, 86.2, 82.5, 45.2, 44.6, 41.8, 41.6, 39.1, 36.5, 28.4. HRMS (ESI) calc'd for C26H30N4O5Na [M + Na]+: 501.2126, found: 501.2114.1-(4-(4-Nitrobenzyl)piperazin-1-yl)-3-(4-nitrophenyl)prop-2-yn-1-one (40)
39, which was synthesized as previously described,56 (0.05 g, 0.25 mmol, 1.1 eq.), 34 (0.06 g, 0.23 mmol, 1.0 eq.), EDCI-HCl (0.05 g, 0.27 mmol, 1.2 eq.) and HOBt·H2O (0.04 g, 0.27 mmol, 1.2 eq.) were stirred in dry DCM (2 mL) under N2. After 24 h, the reaction mixture was diluted with DCM (15 mL) and washed with H2O (2 × 15 mL), followed by brine (15 mL). The organic layer was then dried over MgSO4 and concentrated in vacuo, to give a white solid, which was purified by silica gel column chromatography, giving a white solid (5% DCM/EtOAc, Rf = 0.52, dry loading using celite) (0.02 g, 20%). 1H NMR (300 MHz, CDCl3) δ 8.28–8.15 (m, 4H), 7.74–7.64 (d, J = 9 Hz, 2H), 7.62–7.43 (d, J = 9 Hz, 2H), 3.95–3.79 (m, 2H), 3.79–3.69 (m, 2H), 3.69–3.54 (s, 2H), 2.70–2.37 (m, 4H). 13C NMR (75 MHz, CDCl3) δ 152.1, 148.2, 147.4, 145.5, 133.1, 129.5, 127.0, 123.8, 88.1, 84.8, 61.8, 52.4, 47.0, 41.6. HRMS (ESI) calc'd for C20H18N4O5 [M + H]+: 395.1357, found: 395.1387.TG2 inhibition assay
Recombinant TG2 was expressed from E. coli and purified as described previously.57 TG2 activity was determined according to a previously published colorimetric activity assay using the chromogenic substrate Cbz-Glu(γ-pnitrophenylester)Gly (AL5).58 In order to determine irreversible inhibition parameters for each inhibitor, enzymatic assays were run under Kitz and Wilson conditions,25,26 in the presence of 100 μM AL5 substrate, in triplicate. A large excess of substrate was used to ensure that any curvature in the slope is not attributed to depletion of the substrate, but rather to time-dependent inhibition. Buffered solutions of 50 mM of 3-(4-morpholino)propanesulfonic acid (MOPS) (pH 6.9), 7.5 mM CaCl2, 100 μM AL5, and various concentrations of inhibitor (from 0.25 to 1200 μM, depending on the inhibitor) were prepared in a 96-well polystyrene microplate with a final volume of 200 μL at 25 °C. AL5 and inhibitor stocks were prepared in DMSO ensuring that the final concentration of this co-solvent did not exceed 10% v/v. To initiate the enzymatic reaction, 5 mU mL−1 (0.25 μM) TG2, or water for the blank, was added to the well and the formation of the hydrolysis product, p-nitrophenolate, was followed at 405 nm for 20 min (a period of time over which the positive control is linear and not being impacted by substrate depletion) using a BioTek Synergy 4 plate reader. Background AL5 hydrolysis was corrected for by subtracting the blank from each reaction. Observed first order rate constants of inactivation (kobs) were obtained by fitting the inhibition data sets by non-linear regression to mono-exponential association eqn (1) using GraphPad Prism software.| Abst = Absmax(1 − e−kobst) | (1) |
The rate constants measured at different inhibitor concentrations were then fitted by non-linear regression to a saturation kinetics model, using eqn (2).
 | (2) |
In order to correct for the competition with the assay substrate, AL5, the inhibitor concentrations were divided by α, which equals (1 + ([S]/KM)), where KM = 10 μM. Inhibition parameters, kinact and KI, were then extrapolated from the fitting.
In cases where a saturation plot could not be generated due to solubility constraints and only a few inhibitor concentrations tested, a double-reciprocal plot of (eqn (2)) was made to estimate kinact and KI according to eqn (3).
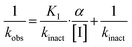 | (3) |
Simple linear regression was performed on the kobs values measured at the lowest concentrations of each inhibitor for validation of kinact/KI ratios.
For reversible inhibitor 16, initial rates of inhibition (vi) were measured at various inhibitor concentrations instead of kobs values. These rates were then normalized with respect to the uninhibited control (% vi/v0). These percent relative rate values were plotted against the inhibitor concentration on a logarithmic scale to generate a IC50 curve. An IC50 value was determined using four-parameter fitting eqn (4) in GraphPad Prism 9.
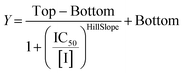 | (4) |
An estimated Ki value was then calculated using the Cheng–Prusoff eqn (5)59
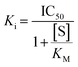 | (5) |
Isozyme selectivity
First, for each inhibitor, a single corrected inhibitor concentration ([I]/α) was chosen, which would result in the complete inactivation of TG2 in under 5 min. Then for each isozyme, the concentration of inhibitor was adjusted according to the value of α determined by the concentration and KM value of the substrate of each isozyme assay. In this way the effective inhibitor concentration ([I]/α) remained constant for each isozyme.20The activities of TG1 and TG6 were measured by a colorimetric assay using the substrate AL5. Assays were performed as previously described for TG2 under Kitz & Wilson conditions established for each transglutaminase isoform by varying the concentration of substrate to 112 μM and 436 μM of AL5 for TG1 and TG6, respectively. The reaction was initiated with the addition of enzyme, 0.10 μM TG1 or 0.32 μM TG6. Formation of the hydrolysis product, p-nitrophenolate, was followed at 405 nm for 100 min for TG1 or 60 min for TG6.16
The isopeptidase activities of activated TG3a and hFXIIIa (purchased from Zedira) were measured by a fluorescence-based assay based on the use of the peptidic FRET-quenched probe A101 (Zedira).16,32 The final assay mixture comprised 50 mM TRIS (pH 7.0), 10 mM CaCl2, 100 mM NaCl, 2.8 mM TCEP, 50 μM A101 and 14 mM H-Gly-OMe. The reaction was monitored at 25 °C using a BioTek Synergy 4 plate reader (Ex/Em: 318/413 nm). Enzymatic inhibition assays were run under Kitz and Wilson conditions, which were established for TG3a and FXIIIa at a substrate (A101) concentration of 50 μM using enzyme concentrations of 0.17 μM and 0.11 μM for TG3a and FXIIIa, respectively. Experiments were completed at least in triplicate. A kobs value was obtained for each isozyme as described above.
GTP binding assay
GTP binding experiments were performed following a previously described protocol.34 hTG2 (20 μg) was incubated at 25 °C for 30 min with or without an irreversible inhibitor (each at a concentration of 2 × KI) with 15 mM CaCl2 in 100 mM MOPS (pH = 6.91). The buffer was then exchanged via dialysis to 100 mM MOPS (pH = 7.0), 1 mM EGTA, and 5 mM MgCl2 to remove calcium using a 14-kDa molecular weight cut-off membrane cuvette (purchased from Millipore-Sigma, Oakville, ON, Canada). The fluorescent, nonhydrolyzable GTP analogue BODIPY GTP-γ-S (purchased from Invitrogen, Waltham, MA, USA), whose fluorescence increases when bound to protein, was then added at a final concentration of 0.5 μM. Fluorescence was then measured on a microplate reader after 10 min of incubation (Ex/Em: 490/520 nm).Intrinsic reactivity
HPLC traces were collected by Gilson-Mandel GXP271 high performance liquid chromatography (HPLC) with UV detection at 214 and 254 nm (Phenomenex Luna, 150 mm × 4.6 mm, 30 min, 1.5 mL min−1 flow rate, 5–95% CH3CN with 0.1% TFA in H2O with 0.1% TFA, 30 min method).Intrinsic reactions were monitored with a 10-fold excess of GSH for compounds 8N, 18, 23B, 23C, 23E and a 100-fold excess for compounds 13 and 23D, depending on the stability of the warhead. In 2 mL HPLC vials fitted with a pre-slit screw cap, 1250 μL of aqueous buffer was added (pH = 7.4, phosphate buffer for 8N, 16, 18, 23B, 23C, 23E and pH = 10.4 CAPS buffer for 13 and 23D, buffer stability over 24 h was confirmed in all cases). Then, 150 μL of a 25- or 250-mM stock solution of GSH in the same buffer was added. The reactions were initiated by the addition of 150 μL of 2.5-mM warhead stock solution in DMSO to a final concentration of 0.25 mM with 10% v/v DMSO as co-solvent. The final concentration of GSH was either 2.5 or 25 mM. Once initiated, the HPLC vials were inverted, placed on the autosampler tray and an initial aliquot was taken immediately. The vials were incubated at 22 °C with aliquots taken at pre-determined time points over the course of at least 5–6 half lives. The disappearance or decrease in the area under the curve (AUC) of the chromatogram of each compound was measured in triplicate.
The decreasing AUC of the inhibitor peaks of the HPLC chromatograms were fitted to a mono-exponential decay model, with the constraint that the lower plateau = 0 (eqn (6)).
| AUC = AUC0(e−kobst) | (6) |
Second order rates constants were then calculated by dividing these pseudo-first order rate constants by the concentration of excess thiol (eqn (7)).
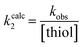 | (7) |
The final, corrected second order rate constants were then calculated by dividing by the fraction of thiolate at the reaction pH, using the corresponding pKα value (8.7) of the thiol (eqn (8)).35
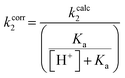 | (8) |
For compound 16 (12 mM), due to limited aqueous solubility, a stability half-life (t½) with 80 mM GSH was determined by 1H NMR in DMSO-d6.23
Molecular docking in molecular operating software (MOE)
Two crystals were selected as a target receptor enzyme, and were imported from their PDB files, namely 2Q3Z and 3S3S. The structure was prepared using the preparation tool from MOE. First, water molecules, salts and ions were removed from the structure. All hydrogen atoms were then added (electrostatics: 1/r2, dielectric: 2, solvent: 80, van der Waals: 12–6) and the protein structures were finally verified, and corrected manually, for any problems or warnings such as chain breaks, termini missing or unreasonable charges. Ligands were drawn in ChemDraw and imported to MOE; partial charges were calculated using a MMFF94x forcefield and the system was eventually minimized following a 0.0001 kcal mol−1 Å−2 gradient. After solvation, minimization was repeated.The “compute” tool from MOE was used to perform docking analysis of each ligand by a non-covalent approach. Ligand placement was achieved using the Triangle Matcher protocol (London dG) to produce 30 poses. In addition, a Rigid Receptor refinement protocol was performed (GBVI/WSA dG) and a total of 5 to 15 final poses were obtained. Finally, the top pose (lowest S-score) was chosen and using the builder tool from MOE, the covalent bond between residue CYS277 and the warhead of the bound inhibitors were manually created, prior to minimization of the system (0.001 kcal mol−1 Å−2).
Author contributions
Conceptualization: J. W. K. and L. K. M.; funding acquisition: J. W. K.; supervision: J. W. K.; investigation and methodology: L. K. M., N. M., J. E. B.; writing – original draft: L. K. M., writing – reviewing and editing: all authors.Conflicts of interest
There are no conflicts of interest to declare.List of abbreviations
| AcOH | Acetic acid |
| Ad | Adamantane |
| Boc | tert-Butyloxycarbonyl |
| Cys | Cysteine |
| DCC | N,N′-Dicyclohexylcarbodiimide |
| DCM | Dichloromethane |
| DIPEA | Diisopropylethylamine |
| DMAP | 4-Dimethylaminopyridine |
| DMF | Dimethylformamide |
| DMP | Dess–Martin Periodinane |
| DPPA | Diphenylphosphoryl azide |
| EDCI | 1-Ethyl-3-(3-dimethylaminopropyl)carbodiimide |
| Et2O | Diethyl ether |
| EtOAc | Ethyl acetate |
| EtOH | Ethanol |
| HBTU | N,N,N′,N′-Tetramethyl-O-(1H-benzotriazol-1-yl)uronium hexafluorophosphate |
| HOBt | Hydroxybenzotriazole |
| MeCN | Acetonitrile |
| MeOH | Methanol |
| Na Asc | Sodium Ascorbate |
| NEt3 | Triethylamine |
| NMI | N-Methyl imidazole |
| R.T. | Room temperature |
| tBuOH | tert-butanol |
| TCFH | N′-tetramethylformamidinium hexafluorophosphate |
| TCICA | Trichloroisocyanuric acid |
| TEMPO | 2,2,6,6-Tetramethylpiperidinyloxy |
| TFA | Trifluoroacetic acid |
| THF | Tetrahydrofuran |
| THP | Tetrahydropyranyl |
Data availability
The data supporting this article (kinetic graphs, HPLC traces, and NMR spectra) have been included as part of the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d5md00777a.Acknowledgements
The authors are grateful to Dr. Kim Y. P. Apperley, for her discovery of the irreversible inhibitor KA22b, carefully documented in her doctoral thesis.23 J. W. K is grateful to the Natural Sciences and Engineering Research Council of Canada (NSERC) and the Canadian Institutes of Health Research (CIHR) for funding. L. K. M. thanks NSERC for a CGS-D Award.References
- S. Gundemir, G. Colak, J. Tucholski and G. V. Johnson, Transglutaminase 2: a molecular Swiss army knife, Biochim. Biophys. Acta, 2012, 1823(2), 406–419 CrossRef CAS PubMed.
- J. W. Keillor, C. M. Clouthier, K. Y. P. Apperley, A. Akbar and A. Mulani, Acyl transfer mechanisms of tissue transglutaminase, Bioorg. Chem., 2014, 57, 186–197 CrossRef CAS PubMed.
- R. L. Eckert, M. T. Kaartinen, M. Nurminskaya, A. M. Belkin, G. Colak, G. V. Johnson and K. Mehta, Transglutaminase regulation of cell function, Physiol. Rev., 2014, 94(2), 383–417 CrossRef CAS.
- J. S. Chen and K. Mehta, Tissue transglutaminase: an enzyme with a split personality, Int. J. Biochem. Cell Biol., 1999, 31(8), 817–836 CrossRef CAS.
- S. Liu, R. A. Cerione and J. Clardy, Structural basis for the guanine nucleotide-binding activity of tissue transglutaminase and its regulation of transamidation activity, Proc. Natl. Acad. Sci. U. S. A., 2002, 99(5), 2743–2747 CrossRef CAS PubMed.
- D. M. Pinkas, P. Strop, A. T. Brunger and C. Khosla, Transglutaminase 2 Undergoes a Large Conformational Change upon Activation, PLoS Biol., 2007, 5(12), e327 CrossRef.
- G. E. Begg, S. R. Holman, P. H. Stokes, J. M. Matthews, R. M. Graham and S. E. Iismaa, Mutation of a Critical Arginine in the GTP-binding Site of Transglutaminase 2 Disinhibits Intracellular Cross-linking Activity*, J. Biol. Chem., 2006, 281(18), 12603–12609 CrossRef CAS.
- A. S. Sewa, H. A. Besser, I. I. Mathews and C. Khosla, Structural and mechanistic analysis of Ca2+-dependent regulation of transglutaminase 2 activity using a Ca2+-bound intermediate state, Proc. Natl. Acad. Sci. U. S. A., 2024, 121(28), e2407066121 CrossRef CAS PubMed.
- D. Schuppan, M. Mäki, K. E. A. Lundin, J. Isola, T. Friesing-Sosnik, J. Taavela, A. Popp, J. Koskenpato, J. Langhorst, Ø. Hovde, M.-L. Lähdeaho, S. Fusco, M. Schumann, H. P. Török, J. Kupcinskas, Y. Zopf, A. W. Lohse, M. Scheinin, K. Kull, L. Biedermann, V. Byrnes, A. Stallmach, J. Jahnsen, J. Zeitz, R. Mohrbacher and R. Greinwald, A Randomized Trial of a Transglutaminase 2 Inhibitor for Celiac Disease, N. Engl. J. Med., 2021, 385(1), 35–45 CrossRef CAS.
- T. S. Johnson, M. Fisher, J. L. Haylor, Z. Hau, N. J. Skill, R. Jones, R. Saint, I. Coutts, M. E. Vickers, A. M. El Nahas and M. Griffin, Transglutaminase inhibition reduces fibrosis and preserves function in experimental chronic kidney disease, J. Am. Soc. Nephrol., 2007, 18(12), 3078–3088 CrossRef CAS.
- C. Kerr, H. Szmacinski, M. L. Fisher, B. Nance, J. R. Lakowicz, A. Akbar, J. W. Keillor, T. Lok Wong, R. Godoy-Ruiz, E. A. Toth, D. J. Weber and R. L. Eckert, Transamidase site-targeted agents alter the conformation of the transglutaminase cancer stem cell survival protein to reduce GTP binding activity and cancer stem cell survival, Oncogene, 2017, 36(21), 2981–2990 CrossRef CAS PubMed.
- Z. Szondy, I. Korponay-Szabó, R. Király, Z. Sarang and G. J. Tsay, Transglutaminase 2 in human diseases, Biomedicine, 2017, 7(3), 15 CrossRef PubMed.
- J. W. Keillor, N. Chabot, I. Roy, A. Mulani, O. Leogane and C. Pardin, Irreversible inhibitors of tissue transglutaminase, Adv. Enzymol. Relat. Areas Mol. Biol., 2011, 78, 415–447 CAS.
- N. J. Cundy, J. Arciszewski, E. W. J. Gates, S. L. Acton, K. D. Passley, E. Awoonor-Williams, E. K. Boyd, N. Xu, É. Pierson, C. Fernandez-Ansieta, M. R. Albert, N. M. R. McNeil, G. Adhikary, R. L. Eckert and J. W. Keillor, Novel irreversible peptidic inhibitors of transglutaminase 2, RSC Med. Chem., 2023, 14(2), 378–385 RSC.
- N. M. R. McNeil, E. W. J. Gates, N. Firoozi, N. J. Cundy, J. Leccese, S. Eisinga, J. D. A. Tyndall, G. Adhikary, R. L. Eckert and J. W. Keillor, Structure-activity relationships of N-terminal variants of peptidomimetic tissue transglutaminase inhibitors, Eur. J. Med. Chem., 2022, 232, 114172 CrossRef CAS.
- A. Akbar, N. M. R. McNeil, M. R. Albert, V. Ta, G. Adhikary, K. Bourgeois, R. L. Eckert and J. W. Keillor, Structure-Activity Relationships of Potent, Targeted Covalent Inhibitors That Abolish Both the Transamidation and GTP Binding Activities of Human Tissue Transglutaminase, J. Med. Chem., 2017, 60(18), 7910–7927 CrossRef CAS.
- E. Badarau, Z. Wang, D. L. Rathbone, A. Costanzi, T. Thibault, C. E. Murdoch, S. El Alaoui, M. Bartkeviciute and M. Griffin, Development of Potent and Selective Tissue Transglutaminase Inhibitors: Their Effect on TG2 Function and Application in Pathological Conditions, Chem. Biol., 2015, 22(10), 1347–1361 CrossRef CAS PubMed.
- M. E. Prime, F. A. Brookfield, S. M. Courtney, S. Gaines, R. W. Marston, O. Ichihara, M. Li, D. Vaidya, H. Williams, A. Pedret-Dunn, L. Reed, S. Schaertl, L. Toledo-Sherman, M. Beconi, D. Macdonald, I. Muñoz-Sanjuan, C. Dominguez and J. Wityak, Irreversible 4-Aminopiperidine Transglutaminase 2 Inhibitors for Huntington's Disease, ACS Med. Chem. Lett., 2012, 3(9), 731–735 CrossRef CAS.
- C. Pardin, I. Roy, W. D. Lubell and J. W. Keillor, Reversible and Competitive Cinnamoyl Triazole Inhibitors of Tissue Transglutaminase, Chem. Biol. Drug Des., 2008, 72(3), 189–196 CrossRef CAS.
- L. Mader, S. K. I. Watt, H. R. Iyer, L. Nguyen, H. Kaur and J. W. Keillor, The war on hTG2: warhead optimization in small molecule human tissue transglutaminase inhibitors, RSC Med. Chem., 2023, 14(2), 277–298 RSC.
- C. Pardin, I. Roy, R. A. Chica, E. Bonneil, P. Thibault, W. D. Lubell, J. N. Pelletier and J. W. Keillor, Photolabeling of tissue transglutaminase reveals the binding mode of potent cinnamoyl inhibitors, Biochemistry, 2009, 48(15), 3346–3353 CrossRef CAS PubMed.
- L. K. Mader and J. W. Keillor, Methods for kinetic evaluation of reversible covalent inhibitors from time-dependent IC50 data, RSC Med. Chem., 2025, 16, 2517–2531 RSC.
- K. Y. P. Apperley, Reversible and photolabile inhibitors for human tissue transglutaminase, Ph.D. Thesis, University of Ottawa, 2017, DOI:10.20381/ruor-20873.
- K. Y. P. Apperley, I. Roy, V. Saucier, N. Brunet-Filion, S.-P. Piscopo, C. Pardin, É. De Francesco, C. Hao and J. W. Keillor, Development of new scaffolds as reversible tissue transglutaminase inhibitors, with improved potency or resistance to glutathione addition, MedChemComm, 2017, 8(2), 338–345 RSC.
- L. K. Mader, J. E. Borean and J. W. Keillor, A practical guide for the assay-dependent characterisation of irreversible inhibitors, RSC Med. Chem., 2025, 16, 63–76 RSC.
- R. Kitz and I. B. Wilson, Esters of Methanesulfonic Acid as Irreversible Inhibitors of Acetylcholinesterase, J. Biol. Chem., 1962, 237(10), 3245–3249 CrossRef CAS.
- L. K. Mader and J. W. Keillor, Fitting of kinact and KI Values from Endpoint Pre-incubation IC50 Data, ACS Med. Chem. Lett., 2024, 15(5), 731–738 CrossRef CAS.
- K. Nepali, H.-Y. Lee and J.-P. Liou, Nitro-Group-Containing Drugs, J. Med. Chem., 2019, 62(6), 2851–2893 CrossRef CAS.
- M. E. Flanagan, J. A. Abramite, D. P. Anderson, A. Aulabaugh, U. P. Dahal, A. M. Gilbert, C. Li, J. Montgomery, S. R. Oppenheimer, T. Ryder, B. P. Schuff, D. P. Uccello, G. S. Walker, Y. Wu, M. F. Brown, J. M. Chen, M. M. Hayward, M. C. Noe, R. S. Obach, L. Philippe, V. Shanmugasundaram, M. J. Shapiro, J. Starr, J. Stroh and Y. Che, Chemical and Computational Methods for the Characterization of Covalent Reactive Groups for the Prospective Design of Irreversible Inhibitors, J. Med. Chem., 2014, 57(23), 10072–10079 CrossRef CAS.
- G. L. Beutner, I. S. Young, M. L. Davies, M. R. Hickey, H. Park, J. M. Stevens and Q. Ye, TCFH–NMI: Direct Access to N-Acyl Imidazoliums for Challenging Amide Bond Formations, Org. Lett., 2018, 20(14), 4218–4222 CrossRef CAS PubMed.
- I. Lindemann, J. Böttcher, K. Oertel, J. Weber, M. Hils, R. Pasternack, U. Linne, A. Heine and G. Klebe, Inhibitors of transglutaminase 2: A therapeutic option in celiac disease, RCSB Protein Data Bank, 2011, 3S3S Search PubMed.
- K. Oertel, A. Hunfeld, E. Specker, C. Reiff, R. Seitz, R. Pasternack and J. Dodt, A highly sensitive fluorometric assay for determination of human coagulation factor XIII in plasma, Anal. Biochem., 2007, 367(2), 152–158 CrossRef CAS PubMed.
- R. Király, K. Thangaraju, Z. Nagy, R. Collighan, Z. Nemes, M. Griffin and L. Fésüs, Isopeptidase activity of human transglutaminase 2: disconnection from transamidation and characterization by kinetic parameters, Amino Acids, 2016, 48(1), 31–40 CrossRef.
- P. Navals, A. M. M. Rangaswamy, P. Kasyanchyk, M. V. Berezovski and J. W. Keillor, Conformational Modulation of Tissue Transglutaminase via Active Site Thiol Alkylating Agents: Size Does Not Matter, Biomolecules, 2024, 14(4), 496 CrossRef CAS PubMed.
- S. K. I. Watt, J. G. Charlebois, C. N. Rowley and J. W. Keillor, A mechanistic study of thiol addition to N-phenylacrylamide, Org. Biomol. Chem., 2022, 20(45), 8898–8906 RSC.
- S. K. I. Watt, J. G. Charlebois, C. N. Rowley and J. W. Keillor, A mechanistic study of thiol addition to N-acryloylpiperidine, Org. Biomol. Chem., 2023, 21(10), 2204–2212 RSC.
- S. Xu, H.-H. Chen, J.-J. Dai and H.-J. Xu, Copper-Promoted Reductive Coupling of Aryl Iodides with 1,1,1-Trifluoro-2-iodoethane, Org. Lett., 2014, 16(9), 2306–2309 CrossRef CAS PubMed.
- G. Schwertz, M. S. Frei, M. C. Witschel, M. Rottmann, U. Leartsakulpanich, P. Chitnumsub, A. Jaruwat, W. Ittarat, A. Schäfer, R. A. Aponte, N. Trapp, K. Mark, P. Chaiyen and F. Diederich, Conformational Aspects in the Design of Inhibitors for Serine Hydroxymethyltransferase (SHMT): Biphenyl, Aryl Sulfonamide, and Aryl Sulfone Motifs, Chem. – Eur. J., 2017, 23(57), 14345–14357 CrossRef CAS PubMed.
- M. Liutkus, C. Dacquet, V. Audinot-Bouchez, J. Boutin, D.-H. Caignard, A. Ktorza and R. Gree, Synthesis of a Novel Series of 8-HETE Analogs and their Biological Evaluation Towards the PPAR Nuclear Receptors, J. Enzyme Inhib. Med. Chem., 2008, 5(8), 503–511 CAS.
- Z. Chen, P. Liang, F. Xu, Z. Deng, L. Long, G. Luo and M. Ye, Metal-Free Aminothiation of Alkynes: Three-Component Tandem Annulation toward Indolizine Thiones from 2-Alkylpyridines, Ynals, and Elemental Sulfur, J. Org. Chem., 2019, 84(19), 12639–12647 CrossRef CAS.
- M. Dell'Acqua, V. Pirovano, G. Confalonieri, A. Arcadi, E. Rossi and G. Abbiati, Synthesis of 3-benzylisoquinolines by domino imination/cycloisomerisation of 2-propargylbenzaldehydes, Org. Biomol. Chem., 2014, 12(40), 8019–8030 RSC.
- A. Nowak-Król, B. Koszarna, S. Y. Yoo, J. Chromiński, M. K. Wȩcławski, C.-H. Lee and D. T. Gryko, Synthesis of trans-A2B2-Porphyrins Bearing Phenylethynyl Substituents, J. Org. Chem., 2011, 76(8), 2627–2634 CrossRef.
- C. Wiles, P. Watts and S. J. Haswell, Clean and selective oxidation of aromatic alcohols using silica-supported Jones' reagent in a pressure-driven flow reactor, Tetrahedron Lett., 2006, 47(30), 5261–5264 CrossRef CAS.
- G. Vaidyanathan, D. McDougald, J. Choi, M. Pruszynski, E. Koumarianou, Z. Zhou and M. R. Zalutsky, N-Succinimidyl 3-((4-(4-[18F]fluorobutyl)-1H-1,2,3-triazol-1-yl)methyl)-5-(guanidinomethyl)benzoate ([18F]SFBTMGMB): a residualizing label for 18F-labeling of internalizing biomolecules, Org. Biomol. Chem., 2016, 14(4), 1261–1271 RSC.
- D. T. Pournara, A. Durner, E. Kritsi, A. Papakostas, P. Zoumpoulakis, A. Nicke and M. Koufaki, Design, Synthesis, and in vitro Evaluation of P2X7 Antagonists, ChemMedChem, 2020, 15(24), 2530–2543 CrossRef PubMed.
- Y. Sun, H. Zheng, L. Qian, Y. Liu, D. Zhu, Z. Xu, W. Chang, J. Xu, L. Wang, B. Sun, L. Gu, H. Yuan and H. Lou, Targeting GDP-Dissociation Inhibitor Beta (GDI2) with a Benzo[a]quinolizidine Library to Induce Paraptosis for Cancer Therapy, JACS Au, 2023, 3(10), 2749–2762 CrossRef CAS PubMed.
- R. Valderrama-Callejón, E. L. Vargas, I. Alonso, M. Tortosa and M. Belén Cid, Diboron Reagents in N−N Bond Cleavage of Hydrazines, N-Nitrosamines, and Azides: Reactivity and Mechanistic Insights, Chem. – Eur. J., 2025, 31(26), e202404081 CrossRef.
- E. P. Ávila, L. A. de Oliveira, B. A. D. Neto, M. V. de Almeida and J. R. Pliego Jr., Flavanone-enabled CuAAC Reaction: Noninnocent Reagents Driving a Mononuclear Mechanism Over the Dinuclear Paradigm, Chem. – Eur. J., 2025, 31(20), e202500121 CrossRef.
- A. Srivastava, L. Aggarwal and N. Jain, One-Pot Sequential Alkynylation and Cycloaddition: Regioselective Construction and Biological Evaluation of Novel Benzoxazole–Triazole Derivatives, ACS Comb. Sci., 2015, 17(1), 39–48 CrossRef CAS PubMed.
- X. Li, Y. Wu, Y. Wang, Q. You and X. Zhang, Click Chemistry Synthesis of Novel Natural Product-Like Caged Xanthones Bearing a 1,2,3-Triazole Moiety with Improved Druglike Properties as Orally Active Antitumor Agents, Molecules, 2017, 22(11), 1834 CrossRef PubMed.
- R. Jannapu Reddy, M. Waheed, A. Haritha Kumari and G. Rama Krishna, Interrupted CuAAC-Thiolation for the Construction of 1,2,3-Triazole-Fused Eight-Membered Heterocycles from O-/N-Propargyl derived Benzyl Thiosulfonates with Organic Azides, Adv. Synth. Catal., 2022, 364(2), 319–325 CrossRef CAS.
- A. Roth-Rosendahl and E. Svernhage, Preparation of azetidinylbenzamidines and related compounds for combination therapy of arrhythmia or coagulation controlled complications thereof, WO03/101956A1, 2003.
- J. J. Hayward, L. Mader and J. F. Trant, Giving Preparative Thin-Layer Chromatography Some Tender Loving Care, Synthesis, 2022, 54(10), 2391–2394 CrossRef CAS.
- L.-x. Wang, X.-b. Zhou, M.-l. Xiao, N. Jiang, F. Liu, W.-x. Zhou, X.-k. Wang, Z.-b. Zheng and S. Li, Synthesis and biological evaluation of substituted 4-(thiophen-2-ylmethyl)-2H-phthalazin-1-ones as potent PARP-1 inhibitors, Bioorg. Med. Chem. Lett., 2014, 24(16), 3739–3743 CrossRef CAS.
- L. Šenica, U. Grošelj, M. Kasunič, D. Kočar, B. Stanovnik and J. Svete, Synthesis of Enaminone-Based Vinylogous Peptides, Eur. J. Org. Chem., 2014, 2014(15), 3067–3071 CrossRef.
- S. S. Palimkar, V. S. More, P. H. Kumar and K. V. Srinivasan, Synthesis of an indole containing KDR kinase inhibitor by tandem Sonogashira coupling-5-endo-dig-cyclization as a key step, Tetrahedron, 2007, 63(51), 12786–12790 CrossRef CAS.
- I. Roy, O. Smith, C. M. Clouthier and J. W. Keillor, Expression, purification and kinetic characterisation of human tissue transglutaminase, Protein Expression Purif., 2013, 87(1), 41–46 CrossRef CAS PubMed.
- A. Leblanc, C. Gravel, J. Labelle and J. W. Keillor, Kinetic Studies of Guinea Pig Liver Transglutaminase Reveal a General-Base-Catalyzed Deacylation Mechanism, Biochemistry, 2001, 40(28), 8335–8342 CrossRef CAS.
- Y. Cheng and W. H. Prusoff, Relationship between the inhibition constant (KI) and the concentration of inhibitor which causes 50 per cent inhibition (I50) of an enzymatic reaction, Biochem. Pharmacol., 1973, 22(23), 3099–3108 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2025 |