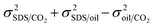Open Access Article
Open Access ArticleMicrofluidic investigation of CO2 foam flow in a heterogeneous porous medium
Nikoo
Moradpour
and
Peichun Amy
Tsai
 *
*
Department of Mechanical Engineering, University of Alberta, Edmonton, Alberta T6G 2R3, Canada. E-mail: peichun.amy.tsai@ualberta.ca
First published on 25th July 2025
Abstract
CO2 foam has emerged as a promising alternative to CO2 gas for mobility control in enhanced oil recovery (EOR) applications, yet the pore-scale dynamics and mechanisms governing foam flow and oil displacement in heterogeneous porous media remain relatively underexplored. This study investigates the pore-scale behavior of CO2 foam flow and its efficacy for EOR in a microfluidic heterogeneous porous medium under ambient conditions. The microfluidic device, composed of parallel high- and low-permeable regions with a permeability ratio of 5.8, enabled direct visualization of foam generation, propagation, and oil displacement dynamics. A flow-focusing geometry was used to produce stable foam bubbles ranging from 30–270 μm, with size and texture governed by gas pressure and liquid flow rates. The foam morphology and transport characteristics were further analyzed as functions of the gas injection ratio (Rg), revealing that increasing Rg led to a higher gas areal fraction (Fg), increased gas trapping, and reduced foam velocity (![[V with combining macron]](https://www.rsc.org/images/entities/i_char_0056_0304.gif) f) and texture (nf). Oil displacement experiments using SDS solution, CO2 gas, and CO2 foam showed distinct differences in performance. SDS solution and CO2 foam formed relatively stable displacement fronts in the high-permeable zone, while CO2 gas exhibited severe viscous fingering and early breakthrough. Foam flooding achieved significantly higher oil recovery (up to 100%) at faster rates compared to gas injection, owing to foam-induced pore blocking and vertical flow diversion into the low-permeable zone. These findings provide insights for optimizing foam-based EOR processes and highlight the value of microfluidics for resolving multiphase transport phenomena at the pore scale.
f) and texture (nf). Oil displacement experiments using SDS solution, CO2 gas, and CO2 foam showed distinct differences in performance. SDS solution and CO2 foam formed relatively stable displacement fronts in the high-permeable zone, while CO2 gas exhibited severe viscous fingering and early breakthrough. Foam flooding achieved significantly higher oil recovery (up to 100%) at faster rates compared to gas injection, owing to foam-induced pore blocking and vertical flow diversion into the low-permeable zone. These findings provide insights for optimizing foam-based EOR processes and highlight the value of microfluidics for resolving multiphase transport phenomena at the pore scale.
1 Introduction
Foams comprise gas bubbles dispersed in a surfactant solution,1,2 forming a dynamic, multiphase system often described as a colloidal dispersion.3 However, foams differ from classical colloidal systems due to their athermal nature, with structural dynamics governed primarily by interfacial and hydrodynamic forces,4 enabling them to modify flow behavior in porous media. Foams have been widely applied in subsurface processes, including enhanced oil recovery (EOR),5–7 soil and aquifer remediation,8 carbon dioxide sequestration,9,10 hydraulic fracturing,11 and hydrogen storage.12,13 Foam improves displacement efficiency by mitigating gas channeling and enhancing sweep efficiency, compared to gas, water, or surfactant flooding alone, particularly in heterogeneous media.14CO2 gas injection is a well-established EOR technique,15 expected to be used for 1.6 million barrel of oil production per day by 2040.16 However, its poor mobility control due to the gas's inherently low density and viscosity drives viscous fingering, gravity override, and preferential flow along high-permeable zones.17 This unfavorable behavior is quantified by the mobility ratio (MR) between the displacing and residing fluids, defined as:
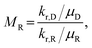 | (1) |
Foam mitigates these mobility challenges by increasing the displacing fluid's viscosity (μD), thereby shifting MR toward more favorable values and suppressing unstable flow.20 The enhanced resistance in foam flow stems from interfacial viscosity at gas–liquid boundaries21 and capillary resistance from foam lamellae confined in pores.22 Foam can also accumulate in high-permeable regions, diverting flow into less-swept, low-permeable zones.6
Conventional core flooding has long been used to assess foam performance in porous media, wherein fluids are injected through rock cores to quantify oil displacement under field-relevant conditions.23,24 While valuable for evaluating bulk displacement efficiency, these methods are inherently limited by the opacity of geological samples, which obscures the pore-scale mechanisms governing foam evolution and oil displacement.25,26 To address this, 2D Hele–Shaw cells of different configurations, such as empty chambers,20,27 bead packs,28 and patterned plates,29 have been used to offer visual access but are limited in mimicking realistic pore geometries. Advanced imaging tools, such as X-ray computed tomography (CT)30 and nuclear magnetic resonance (NMR),31 have also been applied for detailed internal observations, but are constrained by high cost, resolution limits, and experimental complexity.32
Microfluidic platforms have emerged as powerful tools for resolving pore-scale processes with exceptional spatial and temporal resolution.25,26,33–36 These lab-on-a-chip systems replicate complex porous architectures on transparent chips, enabling direct observation of foam generation, transport, and collapse under controlled flow conditions. Advantages such as optical clarity, low fluid consumption, high reproducibility, and rapid prototyping make microfluidics an increasingly indispensable asset for studying multiphase interactions, wettability alterations, and interfacial transport in porous media.25,33,34,37
Recently, microfluidic experiments have played an important role in advancing foam characterization and optimization for EOR applications. Oil is known to destabilize foams by disrupting films, limiting their effectiveness in EOR. To address this, recent microfluidic studies have examined foam stability under varied conditions—including oil composition,38,39 gas component,40,41 brine salinity,39,42 pressure and temperature,41,43 and surfactant type and concentration.44,45 Foam coarsening was observed to increase at higher gas ratios, higher temperatures, lower surfactant concentrations, or when using gas types with higher solubility.41 Bubble population dynamics were shown to depend on the interplay between foam generation and decay.46 Moreover, Lopes et al.47 recommended adding N2 to CO2 gas to decrease bubble coarsening through gas diffusion.
Foam texture—the size, shape, and distribution of gas bubbles—has emerged as a key parameter, besides velocity, governing flow resistance in porous media.48–51 Foam texture is influenced by pore size distribution, gas fraction, surfactant type, and bubble generation and decay dynamics.1,17,52 The transition between “wet” (low gas fraction) and “dry” (high gas fraction) foams is regulated by capillary pressure thresholds and the corresponding water saturation levels.53 At high liquid saturation, foam bubbles are small and easily transported, while at reduced liquid contents and low flow velocities, bubbles tend to coarsen, decreasing foam strength. Microfluidic studies have visualized this transition and its influence on pressure drop and flow redistribution.51,54 Foams with higher gas ratios tend to enhance foam viscosity and improve propagation and diversion efficiency.42,45,49 Wang et al.54 showed that increasing the gas ratio up to 0.8–0.9 increases bubble size, lamellae density, and apparent viscosity. However, some studies indicate that at high gas ratios, coarser foam texture may reduce viscosity.51
Microfluidic visualizations have consistently revealed that fluid displacement by foam is an efficient method to suppress viscous/capillary fingering, thereby improving sweep efficiency.5,6,22,55 In homogeneous devices, only CO2 foam generated a stable displacement front, outperforming liquid, gas, and supercritical CO2 injections.55 Similarly, air foam injections produced better sweep efficiency than air gas, primarily due to enhanced mobility control and delayed breakthrough.6 Conn et al. showed that foam could displace oil from both high- and low-permeability zones and intermediate fractures, achieving up to 80% oil recovery, in contrast to 50% and 20% for water and WAG (water-alternating-gas), respectively.5 Surfactant and surfactant-nanoparticle foams can enhance recovery compared to water injection, reaching 77% and 94%.56 These findings are also supported by Guo et al., who observed foam-enhanced recovery gains exceeding 30% over water and gas flooding alone.57,58
Foam injection also serves as an effective means of mitigating preferential flow pathways commonly encountered during gas injection in heterogeneous porous media.5,59 Microfluidic experiments reveal that foam bubbles preferentially occupy and block high-permeability channels, promoting fluid redistribution into adjacent, previously unswept regions.46,60 This selective occupation is governed by capillary pressure (Pc), which increases as the pore size decreases. When Pc exceeds the entry pressure for gas, the non-wetting phase is excluded from low-permeability pores, thereby encouraging aqueous phase invasion and improved sweep.22 This interplay between capillary thresholds and the foam structure supports foam's unique ability to propagate in heterogeneous porous zones.58
Despite these advances, pore-scale displacement of oil by CO2 foam remains insufficiently understood due to the complex interfacial interactions between oil, gas–liquid foam structures, and solid surfaces within porous matrices. A comprehensive investigation into how injection conditions and foam formulation affect displacement dynamics is critical for optimizing foam performance across EOR, aquifer remediation, and CO2 sequestration scenarios. In this study, we employ a heterogeneous microfluidic platform to visualize and quantify CO2 foam transport and oil displacement behaviors, focusing on the role of the gas fraction and displacing fluid type in controlling sweep efficiency and mobility stabilization.
2 Experimental
In this study, we conducted three distinct sets of experiments designed to quantify and understand the structure, flow transport, and oil displacement performance of CO2 foam in a heterogeneous microfluidic porous medium. First, we characterized foam bubble generation at the microfluidic flow-focusing junction at varying gas injection pressures (Pg) and liquid flow rates (Ql), prior to entry into the microfluidic porous medium. Second, we investigated foam transport dynamics within the porous medium (without a residing fluid), examining the influence of the gas ratio (Rg) on fluid distribution, bubble velocity, and overall foam velocity. Third, we evaluated oil displacement performance using SDS solution, CO2 gas, and CO2 foam, with emphasis on how variations in Ql, Pg, and Rg impact breakthrough behavior and recovery efficiency. Their respective results are presented in sections 3.1–3.3.2.1 Microfluidic chip design and fabrication
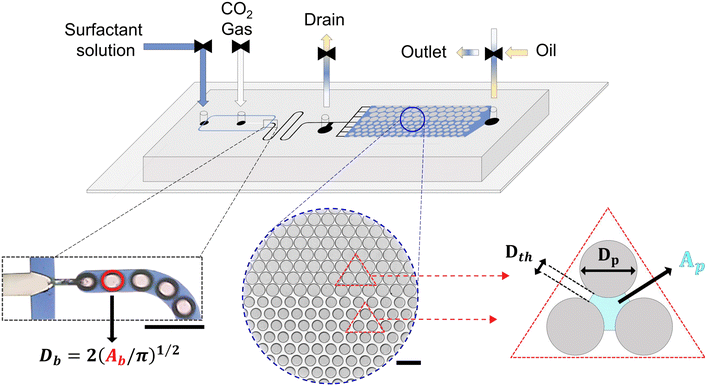
A heterogeneous porous medium (27![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 800 μm × 12
800 μm × 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 400 μm) was designed, comprising two homogeneous regions with distinct porosities and permeabilities (see Fig. 1). All the pillars are circular and arranged in an orthorhombic lattice to represent rock grains. The high-permeable (HP) region contains pillars of diameter Dp = 210 μm, while the low-permeable (LP) region features larger pillars of Dp = 250 μm. The throat distances, Dth, are 60 μm and 21 μm, corresponding to porosities of 45% and 23% in the HP and LP regions, respectively. The geometric parameters—pillar size, pore body, and throat dimensions—were selected based on petrophysical data available in the literature for sandstone reservoirs61–69 to closely mimic natural rock structures.
400 μm) was designed, comprising two homogeneous regions with distinct porosities and permeabilities (see Fig. 1). All the pillars are circular and arranged in an orthorhombic lattice to represent rock grains. The high-permeable (HP) region contains pillars of diameter Dp = 210 μm, while the low-permeable (LP) region features larger pillars of Dp = 250 μm. The throat distances, Dth, are 60 μm and 21 μm, corresponding to porosities of 45% and 23% in the HP and LP regions, respectively. The geometric parameters—pillar size, pore body, and throat dimensions—were selected based on petrophysical data available in the literature for sandstone reservoirs61–69 to closely mimic natural rock structures.
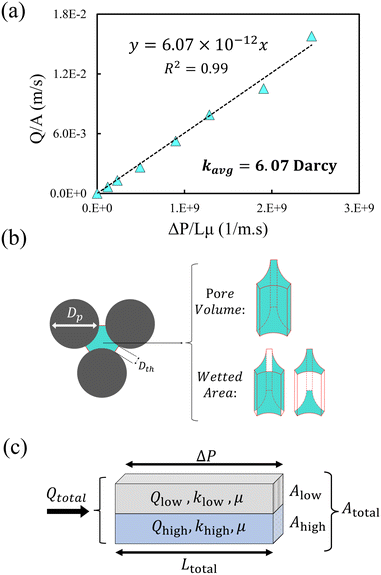
The total average permeability, Kavg, of the porous medium was estimated by injecting pure water at various flow rates (Appendix 5.1), measuring the pressure drop using a differential pressure transducer (Omega 0–50 psi), and applying Darcy's law. The average permeability of the medium (including both low and high permeable regions) was estimated to be 6.07 Darcy. As the permeability of each homogeneous layer is proportional to the porosity and hydraulic radius,70 the permeability ratio between the layers was determined to be 5.8. The hydraulic radii were estimated based on wetted surface areas and pore volumes. Considering the permeability averaging rule for parallel layers, the permeabilities of the low- and high-permeable layers were estimated to be 1.8 and 10.1 Darcy, respectively (see Appendix 5.1 for detailed calculations).
Foam was generated in the same chip using a flow-focusing geometry placed upstream of the porous medium (Fig. 1). Here, a surfactant solution and CO2 gas were co-injected and converged at a 60 μm-wide orifice to produce bubbles. A serpentine (winding) channel segment was incorporated between the foam generation region and the porous medium. The bubbles traveled through the winding channels connected to a drain channel, which remained open during stabilization to prevent unsteady foam entry into the porous structure. The winding channels served both as a temporary reservoir for the generated bubbles and as a hydraulic resistance element that isolates the foam generator from downstream pressure variations, thereby facilitating stable bubble formation and delivery. All flow and distribution channels, including those leading to the porous medium, were uniformly 200 μm wide.
The microfluidic pattern was fabricated on a 6′′ silicon wafer coated with a positive photoresist (AZ P4330) using maskless photolithography (MLA 150, Heidelberg Instruments), followed by uniform etching to a depth of 100 μm (ICPRIE – Oxford Estrelas) to create the microfluidic mold. A soft lithography approach was then used to cast and cure PDMS on a hydrophobized mold (treatedwith Trichloro(octadecyl)silane, Sigma Aldrich) to produce the chips.
All inlet and outlet ports (1.5 mm diameter) were punched using a coring tool (EMS Rapid-Core 1.5) to facilitate tubing connections. The PDMS slab was bonded to a glass slide to enclose the channels. Both surfaces were cleaned with isopropanol and exposed to oxygen plasma to functionalize them with silanol groups, enabling strong, permanent PDMS–glass bonding.
2.2 Materials and methods
The surfactant used to generate foam was 0.5 wt% sodium dodecyl sulphate (SDS), dissolved in Milli-Q water (resistivity > 20 MΩ cm). To enhance visual contrast with the oil phase and improve image processing, 1.0 wt% Nile Blue dye was added to the aqueous solution. The gas phase used was CO2 gas with 99.99% purity (Linde). For oil displacement experiments, a paraffinic oil (ISOPAR M, ExxonMobil) with a viscosity of 3.2 cP was employed. We initially conducted exploratory experiments with oils of higher viscosities (10 cP and 35 cP); however, the higher injection pressures required to saturate and subsequently displace these oils exceeded the mechanical integrity of the PDMS–glass bonding, resulting in de-bonding and fluid leakage. To address this limitation in future studies, we plan to employ alternative microfluidic materials—such as fully glass-based or silicon–glass hybrid chips—with tailored porosity and permeability designs. These platforms offer greater pressure tolerance while maintaining optical accessibility, enabling systematic investigation of foam–oil displacement dynamics over a broader range of viscosity ratios.The contact angles of the aqueous phase and the oil droplets on PDMS were measured in the presence of air to be 86° and 18°, respectively (Appendix 5.3). On bare glass, the contact angles were 32° for the aqueous phase and 12° for the oil, indicating that the porous medium—composed of a glass base and PDMS features—was overall more hydrophobic (or oil-wet). The wettability of the microfluidic chip remained unchanged after exposure to both the SDS solution and ISOPAR M oil, indicating no appreciable alteration from surfactant adsorption or oil contact. In addition, the interfacial tension (IFT), σ, between various fluids was measured using pendant drop tensiometry.71,72 The results showed the following values for the SDS solution and oil used: σSDS,air = 36 mN m−1, σoil,air = 16 mN m−1, σSDS,CO2 = 38 mN m−1, σoil,CO2 = 18 mN m−1, and σSDS,oil = 17 mN m−1 (see Appendix 5.4).
The stability of foam bubbles could be evaluated using the IFT values to calculate the entering (E), bridging (B), and spreading (S) coefficients following established criteria.73,74 According to the results in Table 1, ISOPAR M oil can enter the gas–liquid interface (E > 0), rupture it (B > 0), and spread along it (S > 0), leading to foam destabilization.
The blue aqueous solution and ISOPAR M oil were injected using a syringe pump (Chemyx) operated in constant flow rate (Ql) mode, and the gas flow was controlled via a pressure-regulated mass flow controller (ALICAT) operated in constant pressure mode (Pg), producing a corresponding gas flow rate (Qg). Microfluidic valves (Upchurch Scientific) were used to open or close the outlet and drain ports as needed.
2.3 Image processing
The microfluidic chip was mounted and secured on an inverted microscope (Zeiss Axio Observer 7 Materials) connected to a high-speed camera (HSC, Photron Nova S9) for experimental visualization. The captured videos were processed and analyzed using Fiji (ImageJ) and MATLAB. In all image analyses in this work, a 2D flow condition was assumed.Different magnifications (2.5×, 5×, and 10×) and frame rates (125–15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 fps) were used depending on the resolution required for each specific analysis. Images were converted to grayscale, and fast Fourier transform (FFT) band-pass filtering75,76 was applied for shading correction. Thresholding was then used to isolate region of interest, such as bubble areas or fluid saturation. Each pixel was assigned to a single fluid phase.
000 fps) were used depending on the resolution required for each specific analysis. Images were converted to grayscale, and fast Fourier transform (FFT) band-pass filtering75,76 was applied for shading correction. Thresholding was then used to isolate region of interest, such as bubble areas or fluid saturation. Each pixel was assigned to a single fluid phase.
Bubble sizes were calculated assuming a circular geometry, using the relation 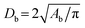 , where Db is the bubble diameter and Ab is the bubble area measured from the 2D images (see Fig. 1). The main oil displacement experiments were carried out at a low magnification of 2.5× to maximize the field of view, which limited resolution and contrast, particularly in the low-permeable area.
, where Db is the bubble diameter and Ab is the bubble area measured from the 2D images (see Fig. 1). The main oil displacement experiments were carried out at a low magnification of 2.5× to maximize the field of view, which limited resolution and contrast, particularly in the low-permeable area.
3 Results and discussion
In this study, we evaluated the dynamics of CO2 foam flow and assess its potential for EOR application in porous media. Therefore, the results are presented in three parts: a) characterization of bubbles generated at the flow-focusing foam generator, b) analysis of foam size and velocity in the porous medium, and c) oil displacement by SDS solution, CO2 gas, and CO2 foam in the porous medium.3.1 Foam bubble size characterization
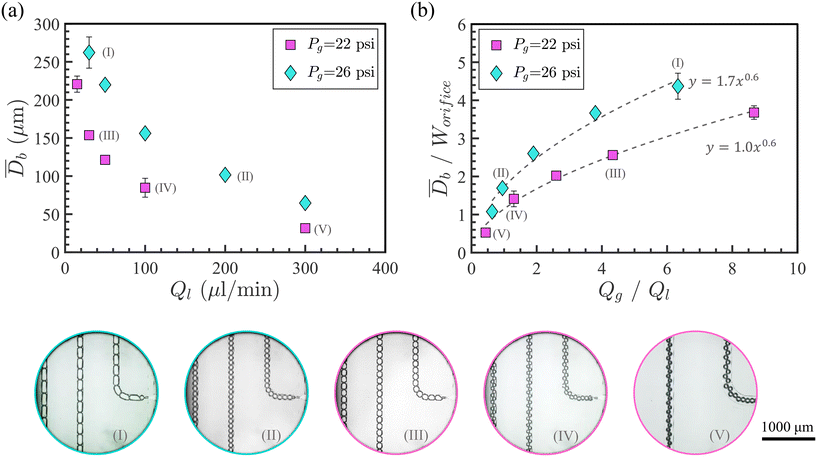
To generate different foam textures using the flow-focusing geometry, gas was injected at constant pressure while varying the liquid flow rate. Two different gas pressures—Pg = 22 and 26 psi—were applied, and the liquid flow rates (Ql) ranged from 25–300 μL min−1 and 30–300 μL min−1, respectively. All experiments in this part were captured at a frame rate of 5000 fps.During these experiments, the outlet port remained open, which allowed the bubbles to travel through the winding channels and into the porous medium, while the drain port was closed. The resulting bubble trains remained stable within the region of interest (ROI shown in Fig. 2 snapshots)–flow-focusing foam generation area–and throughout the observation period. Each experiment was repeated 3 to 4 times to ensure reproducibility.
To calculate the average diameter of the bubbles for each condition, all bubble diameters (Db) in a single representative frame were averaged and reported as ![[D with combining macron]](https://www.rsc.org/images/entities/i_char_0044_0304.gif) b. Fig. 2(a) shows the variation of
b. Fig. 2(a) shows the variation of ![[D with combining macron]](https://www.rsc.org/images/entities/i_char_0044_0304.gif) b as a function of Ql for both gas pressures, indicating that
b as a function of Ql for both gas pressures, indicating that ![[D with combining macron]](https://www.rsc.org/images/entities/i_char_0044_0304.gif) b decreases with increasing Ql at constant gas pressure.
b decreases with increasing Ql at constant gas pressure.
Fig. 2(b) presents the relationship between the normalized bubble size, ![[D with combining macron]](https://www.rsc.org/images/entities/i_char_0044_0304.gif) b/Worifice, and the flow rate ratio, Qg/Ql, at both gas pressures, along with representative experimental snapshots. Here,
b/Worifice, and the flow rate ratio, Qg/Ql, at both gas pressures, along with representative experimental snapshots. Here, ![[D with combining macron]](https://www.rsc.org/images/entities/i_char_0044_0304.gif) b is the average bubble diameter obtained in Fig. 2(a) and Worifice = 60 μm is the constant width of the orifice in the flow-focusing generator.
b is the average bubble diameter obtained in Fig. 2(a) and Worifice = 60 μm is the constant width of the orifice in the flow-focusing generator.
At a constant gas flow rate—Qg = 0.13 mL min−1 and 0.19 mL min−1 at gas pressures Pg = 22 psi and Pg = 26 psi, respectively—increasing Ql resulted in smaller bubbles. Higher liquid flow rates impose greater shear stress on the (constant-pressure) gas thread, increasing the bubble pinch-off frequency at the expense of bubble size.77–79 Moreover, higher gas pressure generated larger bubbles. The bubble diameters ranged from 30–270 μm.
Previous studies using microfluidic flow-focusing bubble generators with different dimensions have proposed a power-law relation, ![[D with combining macron]](https://www.rsc.org/images/entities/i_char_0044_0304.gif) b/Worifice = a(Qg/Ql)b, to predict the bubble size.78,80 Similarly, power-law fits to our data (with R2 values of 0.98 and 0.97) in Fig. 2(b) confirm that the bubble size can be reliably predicted under these injection conditions. The constants a and b appear to depend on the flow-focusing geometry, surface wettability, and fluid properties.78,80 Both fitted curves exhibit similar power-law exponents of 0.6, which aligns with the reported range (0.3 < b < 1.0) in the literature.80 This power-law exponent (b) is primarily determined by the flow focusing geometry,80 while the coefficient a appears to be more sensitive to variations in the injection gas pressure.
b/Worifice = a(Qg/Ql)b, to predict the bubble size.78,80 Similarly, power-law fits to our data (with R2 values of 0.98 and 0.97) in Fig. 2(b) confirm that the bubble size can be reliably predicted under these injection conditions. The constants a and b appear to depend on the flow-focusing geometry, surface wettability, and fluid properties.78,80 Both fitted curves exhibit similar power-law exponents of 0.6, which aligns with the reported range (0.3 < b < 1.0) in the literature.80 This power-law exponent (b) is primarily determined by the flow focusing geometry,80 while the coefficient a appears to be more sensitive to variations in the injection gas pressure.
3.2 Foam flow in the microfluidic porous medium
The flow of generated bubbles in the porous medium was captured to investigate the distribution of fluid phases in terms of 2D areal fractions—CO2 gas (Fg), both trapped and mobile—and the bubble velocities. From this point forward, all experiments were conducted at a constant gas pressure of Pg = 22 psi. Liquid flow rates were varied from 10 to 400 μL min−1, resulting in gas ratios (Rg) ranging from 0.25 to 0.93. The gas ratio Rg is defined as the ratio of the gas flow rate (Qg) to the total flow rate (including both gas and liquid injection rates):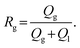 | (2) |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 fps. Due to camera memory limitation (2 second videos at this frame rate), videos were recorded when the fluid distribution was established in the porous medium. The middle snapshot of each video, e.g., Fig. 2(a-I-left), was analyzed to calculate the distribution fractions. During the period of the captured videos, fluid saturation barely changed, as confirmed by the representative videos in the supplementary information. Each experiment was repeated 3–4 times for reproducibility.
000 fps. Due to camera memory limitation (2 second videos at this frame rate), videos were recorded when the fluid distribution was established in the porous medium. The middle snapshot of each video, e.g., Fig. 2(a-I-left), was analyzed to calculate the distribution fractions. During the period of the captured videos, fluid saturation barely changed, as confirmed by the representative videos in the supplementary information. Each experiment was repeated 3–4 times for reproducibility.
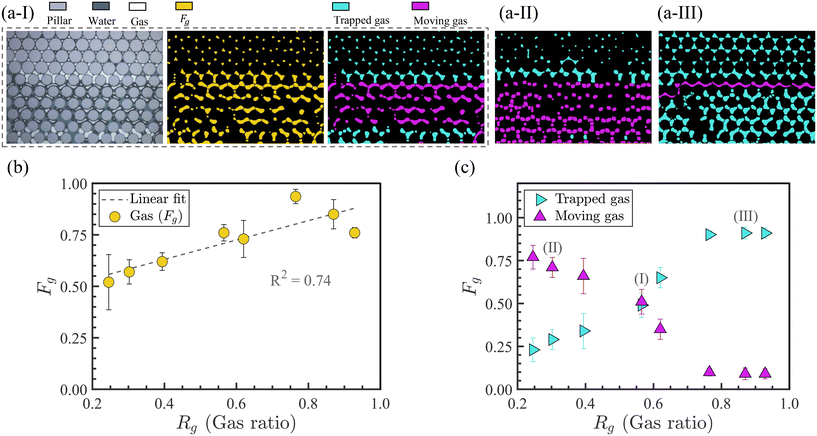
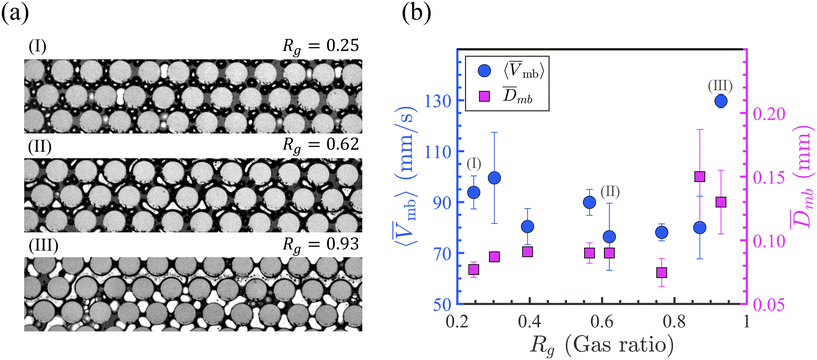
As revealed in Fig. 3(b), increasing Rg from 0.25 to 0.93 resulted in higher gas fractions in the porous medium. This is expected because with increased gas ratios, less water volume is introduced into the device, leading to greater Fg. The data indicate that between 50% and 93% of the pore area is occupied by the gas phase using different tested ranges of wet to dry foams. A linear fit with a coefficient of determination R2 = 0.74 indicates a similar increasing trend in this range of gas ratios.
Each experiment revealed the coexistence of mobile and trapped gas bubbles: some bubbles flowed through the porous structure due to hydrodynamic pressure drop, while others became immobilized, trapped in the pore structures. Thus, we categorized the gas phase into trapped and moving components, and defined their fractions as follows: the trapped (or moving) gas fraction is the ratio of the area occupied by trapped (or moving) gas to the total area occupied by the gas phase.
In Fig. 3(a-I-right), the yellow gas region is divided into purple (moving) and cyan (trapped) regions (to distinguish the moving and trapped bubbles). As shown in Fig. 3(c), increasing Rg resulted in a larger proportion of trapped gas. At Rg = 0.25, the trapped gas fraction is 0.23, which increases to 0.91 at Rg = 0.93.
Beyond Rg = 0.76, the trapped Fg does not change significantly and reaches a relative plateau value close to 0.90. Fig. 3(a-I-right), (a-II), and (a-III) correspond to the labeled data points in Fig. 3(c). As the foam propagates and accumulates in porous media, certain pathways become increasingly blocked, leading to local increases in flow resistance. To sustain fluid transport, flow is redirected through less obstructed routes. In all cases, a central pathway—the horizontal channel within the high-permeability zone closest to the main inlet flow channel—remained unblocked and allowed bubble movement. This persistent open channel likely represents the least resistive path due to its direct alignment with the inlet, enabling preferential flow. This path contributes to ≈10% of the pore volume and accounts for the observed plateau.
Even at the highest moving gas fraction of 0.77 at Rg = 0.25, no moving bubbles were detected in the low-permeable zone. This implies that although the foam bubbles overcome the critical capillary pressure to enter the pore areas in both zones, mobile bubbles exclusively traveled through the high-permeable zone, which offered the path of least resistance.
Across all gas ratios, ![[D with combining macron]](https://www.rsc.org/images/entities/i_char_0044_0304.gif) b/Dth > 1 in both zones, indicating that bubbles generated by the flow focusing geometry needed to shrink or break to enter the pore throats of both zones. This ratio is smaller (0.5–4.7) for the high-permeable zone than in the low-permeable zone (1.5–13.3). As discussed, foam bubbles in the microfluidic porous media are either trapped or mobile. The average sizes of moving bubbles,
b/Dth > 1 in both zones, indicating that bubbles generated by the flow focusing geometry needed to shrink or break to enter the pore throats of both zones. This ratio is smaller (0.5–4.7) for the high-permeable zone than in the low-permeable zone (1.5–13.3). As discussed, foam bubbles in the microfluidic porous media are either trapped or mobile. The average sizes of moving bubbles, ![[D with combining macron]](https://www.rsc.org/images/entities/i_char_0044_0304.gif) mb, and trapped bubble areas Ātb were obtained by averaging the corresponding detected bubbles from the same representative frames used in Fig. 3(a–c).
mb, and trapped bubble areas Ātb were obtained by averaging the corresponding detected bubbles from the same representative frames used in Fig. 3(a–c).
The comparison between ![[D with combining macron]](https://www.rsc.org/images/entities/i_char_0044_0304.gif) mb and Dth, in addition to the visualized results, reveals that regardless of Rg, the bubbles flow only through the high-permeable zone as the less resistive path and they do not need to shrink much (maximum
mb and Dth, in addition to the visualized results, reveals that regardless of Rg, the bubbles flow only through the high-permeable zone as the less resistive path and they do not need to shrink much (maximum ![[D with combining macron]](https://www.rsc.org/images/entities/i_char_0044_0304.gif) mb/Dth is around 2). In contrast, entering the low-permeable zone would require bubbles to shrink by a factor of 4–8. This preferential foam flow aligns with previous observations that foams tend to migrate through a larger pore pathway.81
mb/Dth is around 2). In contrast, entering the low-permeable zone would require bubbles to shrink by a factor of 4–8. This preferential foam flow aligns with previous observations that foams tend to migrate through a larger pore pathway.81
Finally, a comparison of trapped bubble areas (Ātb) with local pore areas (Ap) showed that trapped bubbles conformed closely to the pore size, with Atb/Ap ∼ 1 in both high-permeable and low-permeable zones. Being restricted by the pillar walls, this bubble alignment phenomenon has been observed81 and even used for spatial patterning.82
The solubility and diffusivity of CO2 in water at atmospheric pressure and ambient temperature (14.7 psi and 25 °C) are relatively low.83–86 Under these conditions, the estimated Peclet numbers in our system are considerably high (>1600), indicating that convective transport strongly dominates over diffusion. In addition, the observed foam bubble sizes remain relatively constant during flow, further supporting that CO2 dissolution in water via diffusion is negligible under our experimental conditions.
The displacements of all the bubbles' centroids were tracked and measured throughout the image sequences. The velocities of moving bubbles were then averaged to obtain a single mean value, 〈![[V with combining macron]](https://www.rsc.org/images/entities/i_char_0056_0304.gif) mb〉. Bubbles were classified as mobile or trapped based on their measured velocity. A threshold of 1 pixel per frame was used to distinguish the two populations: bubbles with velocities below this value were considered stationary (trapped), while those with higher velocities were classified as mobile.
mb〉. Bubbles were classified as mobile or trapped based on their measured velocity. A threshold of 1 pixel per frame was used to distinguish the two populations: bubbles with velocities below this value were considered stationary (trapped), while those with higher velocities were classified as mobile.
Fig. 4(a and b-I to III) show snapshots of three representative moving foam bubbles at different Rg values along with the corresponding average velocity results. As shown, 〈![[V with combining macron]](https://www.rsc.org/images/entities/i_char_0056_0304.gif) mb〉 generally falls in the range of 75–95 mm s−1, except at Rg = 0.93, where 〈
mb〉 generally falls in the range of 75–95 mm s−1, except at Rg = 0.93, where 〈![[V with combining macron]](https://www.rsc.org/images/entities/i_char_0056_0304.gif) mb〉 = 130 mm s−1. In contrast to uniform bubbles generated at the flow-focusing junction (Fig. 2(a)), moving bubbles within the porous medium displayed less uniformity and were more influenced by the local porous structure. As shown in Fig. 4(b), the mean size of the moving bubbles,
mb〉 = 130 mm s−1. In contrast to uniform bubbles generated at the flow-focusing junction (Fig. 2(a)), moving bubbles within the porous medium displayed less uniformity and were more influenced by the local porous structure. As shown in Fig. 4(b), the mean size of the moving bubbles, ![[D with combining macron]](https://www.rsc.org/images/entities/i_char_0044_0304.gif) mb, remains relatively unchanged (0.074–0.091 mm) for Rg ≤ 0.76, but increases at higher gas ratios. Bubbles at Rg ≤ 0.76 are well separated and more circular (Fig. 4(a-I and II)), whereas Rg > 0.76 caused connected large bubbles (Fig. 4(a-III)). Moreover, bubble velocity appears to have a weak dependence on the gas ratio, as no clear correlation is observed in 〈
mb, remains relatively unchanged (0.074–0.091 mm) for Rg ≤ 0.76, but increases at higher gas ratios. Bubbles at Rg ≤ 0.76 are well separated and more circular (Fig. 4(a-I and II)), whereas Rg > 0.76 caused connected large bubbles (Fig. 4(a-III)). Moreover, bubble velocity appears to have a weak dependence on the gas ratio, as no clear correlation is observed in 〈![[V with combining macron]](https://www.rsc.org/images/entities/i_char_0056_0304.gif) mb〉 with increasing Rg from 0.25 to 0.93 (see Fig. 4(b)).
mb〉 with increasing Rg from 0.25 to 0.93 (see Fig. 4(b)).
In addition to hydrodynamic pressure, the Jamin effect plays a significant role in governing foam flow dynamics within porous media.87–89 When a bubble approaches a pore constriction, its leading edge compresses while the trailing edge remains relatively undeformed, producing a curvature difference that results in additional capillary resistance.88 Two factors help reduce this resistance: (1) smaller bubbles require less energy to deform, weakening the Jamin effect, and (2) higher gas entrapment increases the pressure gradient across the porous medium, enhancing foam bubble velocity. These competing factors may explain the lack of a consistent trend in 〈![[V with combining macron]](https://www.rsc.org/images/entities/i_char_0056_0304.gif) mb〉 versus Rg.
mb〉 versus Rg.
The average foam velocity (![[V with combining macron]](https://www.rsc.org/images/entities/i_char_0056_0304.gif) f), incorporating both moving and trapped bubbles, was estimated as:
f), incorporating both moving and trapped bubbles, was estimated as:
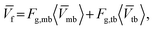 | (3) |
![[V with combining macron]](https://www.rsc.org/images/entities/i_char_0056_0304.gif) f denotes the average foam velocity; 〈
f denotes the average foam velocity; 〈![[V with combining macron]](https://www.rsc.org/images/entities/i_char_0056_0304.gif) tb〉 represents the trapped foam velocity, which is assumed to be zero (as its traveling distance is less than 1 pixel between consecutive frames); 〈
tb〉 represents the trapped foam velocity, which is assumed to be zero (as its traveling distance is less than 1 pixel between consecutive frames); 〈![[V with combining macron]](https://www.rsc.org/images/entities/i_char_0056_0304.gif) mb〉 is the velocity of moving bubbles as obtained in Fig. 4(b).
mb〉 is the velocity of moving bubbles as obtained in Fig. 4(b).
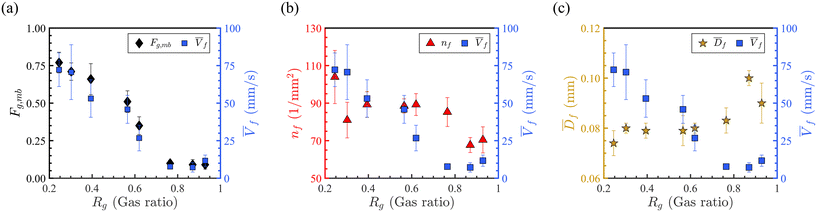
As revealed in Fig. 5(a-right axis), the overall foam velocity ![[V with combining macron]](https://www.rsc.org/images/entities/i_char_0056_0304.gif) f decreases from approximately 73 mm s−1 to 7 mm s−1, with increasing gas ratio. Comparing trends in
f decreases from approximately 73 mm s−1 to 7 mm s−1, with increasing gas ratio. Comparing trends in ![[V with combining macron]](https://www.rsc.org/images/entities/i_char_0056_0304.gif) f and Fg,mb reveals that bubble entrapment has a stronger influence on overall foam velocity than on the velocity of individual moving bubbles (Fig. 4(b)). In other words, a less mobile gas fraction results in slower foam propagation. It is important to emphasize that
f and Fg,mb reveals that bubble entrapment has a stronger influence on overall foam velocity than on the velocity of individual moving bubbles (Fig. 4(b)). In other words, a less mobile gas fraction results in slower foam propagation. It is important to emphasize that ![[V with combining macron]](https://www.rsc.org/images/entities/i_char_0056_0304.gif) f accounts for all bubbles, while 〈
f accounts for all bubbles, while 〈![[V with combining macron]](https://www.rsc.org/images/entities/i_char_0056_0304.gif) mb〉 only represents the moving ones.
mb〉 only represents the moving ones.
Furthermore, foam velocity variation with Rg is compared to foam texture—defined as the number of bubbles per unit area (nf)—and to the mean bubble diameter of the same foam (![[D with combining macron]](https://www.rsc.org/images/entities/i_char_0044_0304.gif) f) in Fig. 5(b) and (c), respectively. These parameters account for both moving and trapped bubbles. The results show that overall, larger bubbles (or lower foam texture) are associated with slower foam movement. The mean bubble diameter
f) in Fig. 5(b) and (c), respectively. These parameters account for both moving and trapped bubbles. The results show that overall, larger bubbles (or lower foam texture) are associated with slower foam movement. The mean bubble diameter ![[D with combining macron]](https://www.rsc.org/images/entities/i_char_0044_0304.gif) f ranges from 740 μm to 1000 μm, while nf ranges from 70 to 104 mm−2. This inverse relationship reflects that the increased gas ratio leads to larger bubbles and lower foam texture, which reduces foam mobility. These trends are clearly seen in the comparison between Fig. 5(b) and (c).
f ranges from 740 μm to 1000 μm, while nf ranges from 70 to 104 mm−2. This inverse relationship reflects that the increased gas ratio leads to larger bubbles and lower foam texture, which reduces foam mobility. These trends are clearly seen in the comparison between Fig. 5(b) and (c).
In summary, a higher foam gas ratio results in a greater fraction of the porous medium being occupied by CO2 gas, much of which is trapped rather than flowing. Due to capillary barriers, mobile bubbles consistently flow through the high-permeable zone, while trapped bubbles accumulate in both high- and low-permeable zones depending on Rg. This increased entrapment results in larger average bubble size and lower foam texture, thereby reducing the overall foam velocity.
3.3 Oil displacement experiments by gas, aqueous solution, and foam
In this section, we investigated the enhanced oil recovery (EOR) performance of three displacement strategies by (1) CO2 gas, (2) SDS aqueous solution, and (3) CO2 foam. All experiments were conducted using a 2.5× magnification and 125–250 fps, which provided a field of view of 4096 μm × 4096 μm. The displacement was monitored near the center of the porous medium, away from the outlet, to minimize capillary end effects.55,90 Sudden reductions in capillary pressure near the outlet may lead to local accumulation of wetting fluid and distortion of fluid distribution analysis.ISOPAR M oil was injected from the outlet at a constant flow rate of 100 μl min−1. The drain port remained open to allow the oil phase to exit, while the gas and liquid injection ports were closed (using Idex shut-off valves) to prevent oil from entering the foam generation area. Once the microfluidic porous medium was fully saturated with oil, the respective injection port(s) were opened depending on the displacement fluid. Continued oil injection maintained a back pressure to prevent entry of unstable displacing fluid into the porous space.
The displacing fluid flowed out through the drain port until stable injection conditions were reached, i.e., constant gas pressure (for CO2 gas injections), constant liquid flow rate (for SDS solution injections), or steady bubble regime (for foam injections). Once stabilized, the outlet was opened to atmospheric pressure, the drain port was closed to redirect the displacing fluid into the porous medium, and data acquisition began.
The tested conditions included five CO2 gas pressures (15–28 psi), five SDS solution flow rates (5–100 μm min−1), and six foam gas ratios (Rg = 0.57–0.98 at Pg = 22 psi). Each experiment was repeated 3 to 4 times to ensure reproducibility.
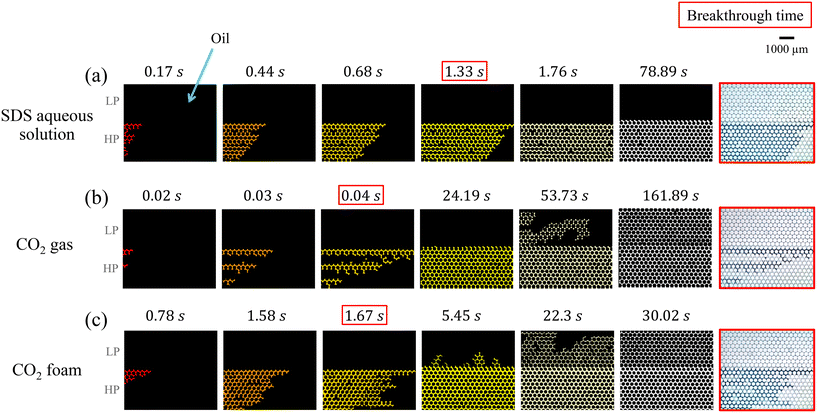
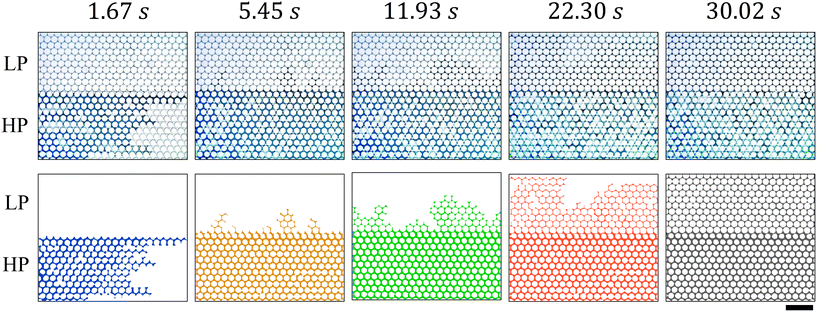
The image sequences in Fig. 6 illustrate oil displacement by the three fluids: (a) SDS aqueous solution, (b) CO2 gas, and (c) CO2 foam. In all cases, displacement initiated in the high-permeable (HP) zone (labeled “HP” in Fig. 6). The SDS solution (Ql = 25 μl min−1) forms a relatively stable displacement front and exhibits a delayed breakthrough at Tbt = 1.33 s, as shown in Fig. 6(a). Although the high-permeable (HP) zone was quickly swept (within 2 s), the solution fails to enter the low-permeable (LP) zone, even after extended injection (up to 78.89 s).
In contrast, CO2 gas injection (at Pg = 22 psi) led to viscous fingering in the direction of flow (left to right), producing an early breakthrough at Tbt = 0.02 s (Fig. 6(b)). Although this unstable gas–oil interface delayed complete oil displacement in the HP zone (24.19 s), gas was able to penetrate the LP zone, achieving nearly complete oil recovery after a prolonged injection period of 161.89 s. The improved sweep may be attributed to a larger pressure, estimated using Darcy's law (along with the chip geometry, fluid properties,91,92 and operating conditions) to be 0.23 psi for SDS solution vs. 7.30 psi for CO2 gas (see Appendix 5.1). Viscous fingering was less severe in the LP zone due to its significantly lower permeability, ≈6× lower than that of the HP zone.
Fig. 6(c) shows the oil displacement by CO2 foam (Rg = 0.87), which achieved a relatively stable front (in the HP zone) similar to SDS solution, but with deeper penetration (in the LP zone) like gas. CO2 foam broke through at Tbt = 1.67 s and recovered oil from the LP zone in just 30.02 s, about 5× faster than the pure gas injection. This better performance stemmed from foam lamellae trapping gas bubbles, thereby creating additional flow resistance and suppressing viscous fingering. Consequently, CO2 foam exhibits both delayed breakthrough and enhanced oil recovery efficiency, combining the benefits of stable propagation (in the HP zone) and deeper penetration (in the LP area).
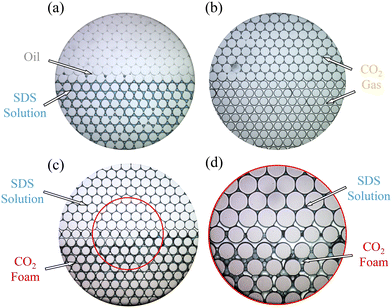
To elucidate the fluid distributions during the microfluidic EOR process, oil displacement experiments under the same conditions as in Fig. 6 were repeated at a higher magnification (5×). The resulting final fluid distributions (after the displacement) are shown in Fig. 7. Consistent with previous observations, the SDS solution (blue) failed to displace oil (gray) in the LP zone (Fig. 7(a)). In contrast, both CO2 gas and foam effectively swept oil from the field of view (Fig. 7(b) and (c)). Fig. 7(c) and (d), acquired at 5× and 10× magnifications, respectively, reveal that the high- and low-permeable zones are mainly displaced by foam and liquid, respectively. This behavior may be attributed to the fact that the non-wetting gas phase overcomes opposing capillary forces in the high-permeable zone more readily. Similar trends have been reported in the literature for heterogeneous porous media, where lower-permeability regions tend to become liquid-rich, while injected foam bubbles preferentially fill the higher-permeability zones.5,22
Breakthrough time (Tbt) is a key performance metric in EOR field applications, as it correlates with sweep efficiency and operational feasibility for a given strategy.93–95 To ensure cost-effectiveness, the volume of produced gas (gas–oil ratio) and water (water cut), relative to oil, must remain sufficiently low.
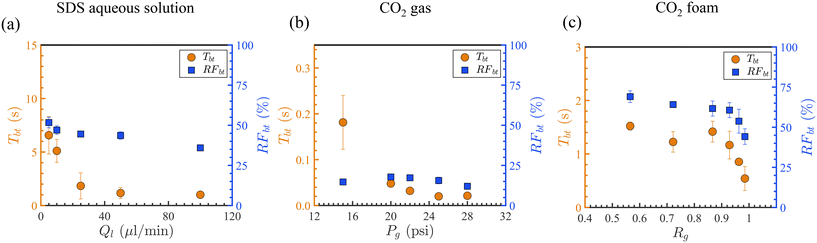
Fig. 8 presents the Tbt and oil recovery factor at breakthrough (RFbt) for all the displacements.
For the SDS aqueous solution, Tbt decreases from 6.6 s to 1.0 s as the injection flow rate (Ql) increases from 5 μl min−1 to 100 μl min−1 (Fig. 8(a)). This is expected, as higher flow rates result in greater hydrodynamic pressure drops, leading to faster fluid propagation and consequently earlier breakthrough. However, a higher Ql, with a shorter Tbt, reduces sweep efficiency, with an RFbt as low as 40% at Ql = 100 μL min−1. The maximum oil recovery at breakthrough for SDS solution injection is 52%. Contact angle measurements confirm that the microfluidic chip is hydrophobic. Even at the lowest flow rate (Ql), the displacement front remains stable, and no lateral capillary fingering is observed. This suggests that oil displacement by the SDS solution is primarily facilitated by desirable viscosity contrast (M < 1) rather than capillary forces. A comparative analysis of the displacement behavior using pure water versus SDS solution would provide further insight into the role of surfactants and capillary effects. However, such a comparison lies beyond the scope of the present study.
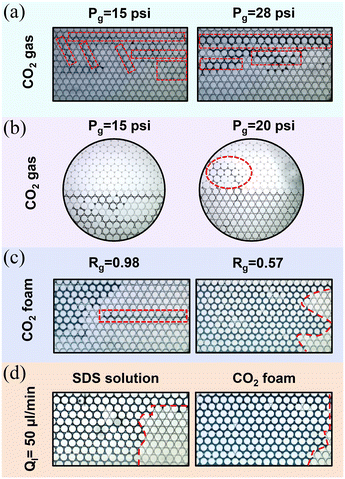
Injection of CO2 gas accelerates the breakthrough times from 0.18 s down to 0.02 s when Pg = 15–28 psi (Fig. 8(b)). In all gas injection experiments, even at the lowest injection pressure Pg, gas fingers through the oil toward the outlet, as shown in Fig. 9(a). Viscous fingering reduced the sweep efficiency, with RFbt being reduced to 12–18%, significantly lower than that of SDS solution and CO2 foam with stable displacement fronts. At Pg = 15 psi, the oil recovery factor by CO2 gas is low despite a delayed breakthrough, as shown in Fig. 9(b). This is because the resulting pressure drop (≈0.3 psig) is too small to effectively push the gas through the porous medium, slowing down the displacement process. Observations show that at this pressure, CO2 cannot enter the LP zone, likely due to its higher entry capillary pressure. Capillary pressure is defined as Pc = Pnon-wet − Pwet = 2γκc![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) cos(θ), where γ is the interfacial tension, θ is the contact angle, and
cos(θ), where γ is the interfacial tension, θ is the contact angle, and 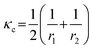 is the average curvature of the porous medium; r1 and r2 are the two principal pore radii or radii of curvature of the meniscus. Here,
is the average curvature of the porous medium; r1 and r2 are the two principal pore radii or radii of curvature of the meniscus. Here, 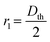 and
and 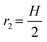 ; Dth is the diameter of the pore throat (60 and 21 μm for the high- and low-permeable zones, respectively), and H is the height of the microfluidic chip (100 μm). Assuming a constant contact angle (cosθ = 1) for both regions, and using the measured pore throat radii, the estimated capillary entry pressures (for gas displacing oil) are approximately 960 Pa for the high-permeability region and 2074 Pa for the low-permeability region. According to Fig. 9(b), it seems that Pg = 15 psi is insufficient to overcome this 1114 Pa pressure difference between the two zones and unable to invade the LP zone. However, when the gas pressure increases to 20 psi, CO2 gas begins to penetrate the LP zone, suggesting that the gas critical entry pressure (Poil + Pc) lies between 15 and 20 psi.
; Dth is the diameter of the pore throat (60 and 21 μm for the high- and low-permeable zones, respectively), and H is the height of the microfluidic chip (100 μm). Assuming a constant contact angle (cosθ = 1) for both regions, and using the measured pore throat radii, the estimated capillary entry pressures (for gas displacing oil) are approximately 960 Pa for the high-permeability region and 2074 Pa for the low-permeability region. According to Fig. 9(b), it seems that Pg = 15 psi is insufficient to overcome this 1114 Pa pressure difference between the two zones and unable to invade the LP zone. However, when the gas pressure increases to 20 psi, CO2 gas begins to penetrate the LP zone, suggesting that the gas critical entry pressure (Poil + Pc) lies between 15 and 20 psi.
Foam flooding significantly improves recovery. As Rg increases from 0.57 to 0.98, Tbt decreases from 1.2 s to 0.54 s, while RFbt ranges from 44% to 70% (Fig. 8(c)). Foam injection at Pg = 22 psi improves the breakthrough time (Tbt = 0.03 s) by over an order of magnitude compared to gas and enhances recovery by 27% or more. Our results reveal that replacing CO2 gas injection with CO2 foam at 0.57 ≤ Rg ≤ 0.98 is beneficial by stabilizing the displacing front, delaying the breakthrough time, and increasing the oil sweep efficiency.
In contrast to the SDS solution, increasing Ql in foam flooding (i.e., lowering Rg) improved oil displacement with longer Tbt and higher oil RFbt. Two factors may contribute to this trend. First, contact between the foam and oil can destabilize the foam, leading to segregated gas slugs, mild viscous fingering, and earlier breakthrough (Fig. 6(c), 1.67 s). As shown in Table 1, ISOPAR M oil exhibits positive entering (E > 0), bridging (B > 0), and spreading (S > 0) coefficients, indicating that the oil can penetrate the gas–liquid interface, rupture the foam lamellae, and spread along the interfacial film. These characteristics suggest a strong tendency for foam destabilization. At the foam–oil front, this leads to lamella rupture, gas slug release, and the onset of gas fingering, as illustrated in Fig. 9(c). The destabilization reduces foam's ability to maintain a stable displacement front, resulting in shorter breakthrough times (Tbt) and lower oil recovery at breakthrough (oil RFbt), as shown in Fig. 8(c). This effect can become more pronounced at higher Rg due to the increased gas content and reduced availability of the foam stabilizer (e.g., surfactant). Moreover, as discussed in section 1, foam flow in porous media can occur in either the high Rg (high-quality) or low Rg (low-quality) regimes. At a constant gas flow rate (22 psi here), the low Rg regime is relatively insensitive to variations in the liquid flow rate, resulting in minimal changes in pressure drop. In contrast, upon transitioning into the high Rg regime, even small reductions in the liquid flow rate lead to significant decreases in pressure drop.53 Numerous studies have observed shear-thinning behavior in the high Rg regime.51,96,97 A lower foam viscosity in this regime causes shorter Tbt, potentially contributing to the decreasing trend observed in Fig. 8(c) beyond Rg = 0.87. Second, gas injection pressure enhances the hydrodynamic driving force, accelerating the displacement front while maintaining stability and improving recovery (Fig. 9(d)). Under the conditions tested, foam destabilization likely dominates the observed trend in Fig. 8(c), while the added driving force explains the superior performance of foam over SDS solution, with higher recovery achieved despite shorter Tbt.
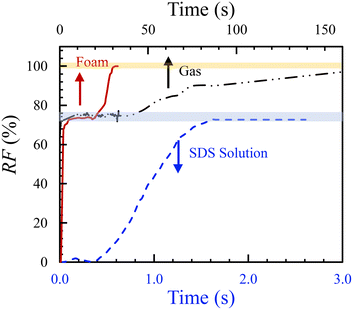
Fig. 10 displays the dynamic performance of oil production (recovery factor, RF, versus time) by the three representative displacing fluids: SDS solution (100 μl min−1, blue), CO2 gas (22 psi, black), and CO2 foam (Rg = 0.57, red). The oil RF at each time was calculated by the ratio of the displaced to the initial oil-saturated area. Note that the SDS curve follows the bottom time axis, while the gas and foam curves correspond to the top axis. The horizontal gray lines indicate the recovery limits for the high-permeable zone (∼70%) and full displacement (100%).
Consistent with the observations in Fig. 6, the SDS solution sweeps the HP zone smoothly, forming a stable displacement front, and reaches a plateau at ≈70%, the high-permeable RF. However, it fails to displace oil from the LP zone, likely due to insufficient injection pressure to overcome the zone's capillary entry pressure. In contrast, both CO2 gas and foam reach the HP zone plateau and proceed to displace oil from the LP zone—after ≈40 s and 15 s, respectively. The gas pressure of 22 psi in both cases is sufficient to overcome the capillary threshold in the LP region.
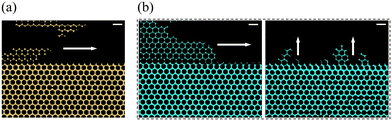
Furthermore, CO2 foam achieves complete recovery in just 31 s, whereas gas alone requires over 150 s—roughly five times longer. This enhanced performance is attributed to two main factors: (1) foam suppresses viscous fingering, in contrast to pure gas (Fig. 11(a and b)), and (2) foam lamellae may block pores in the HP zone, raising the pressure drop in the porous medium and redirecting flow into the LP zone (Fig. 11(b, right)). In addition, while foam initially sweeps the HP zone, it is the liquid phase that ultimately penetrates and displaces oil from the LP region.
4 Conclusions
We experimentally investigate pore-scale CO2 foam dynamics and oil displacement behavior in a microfluidic heterogeneous porous medium, designed to mimic dual-permeability reservoir structures. The microfluidic device was fabricated from PDMS and featured two parallel homogeneous zones—high- and low-permeable regions—characterized by a permeability ratio of 5.8 and porosities of 45% and 23%, respectively. Oil-displacement experiments were conducted under ambient conditions using ISOPAR M oil (as the displaced fluid) and three displacing agents: SDS aqueous solution, CO2 gas, and CO2 foam.Stable foam bubbles were generated via a flow-focusing geometry with an orifice width Worifice = 60 μm. Bubble sizes ranged from 30 μm to 270 μm and were sensitive to both gas pressure (Pg = 22 and 26 psi) and the liquid flow rate (Ql = 25–300 μL min−1). Larger foam bubbles formed at lower Ql and higher Pg. The bubble diameter normalized by the orifice width (![[D with combining macron]](https://www.rsc.org/images/entities/i_char_0044_0304.gif) b/Worifice) followed a power-law trend against the gas-to-liquid flow rate ratio (Qg/Ql), with an exponent of 0.6 under constant Pg, aligning with literature models of foam formation in microfluidic systems.
b/Worifice) followed a power-law trend against the gas-to-liquid flow rate ratio (Qg/Ql), with an exponent of 0.6 under constant Pg, aligning with literature models of foam formation in microfluidic systems.
Foam's structure and velocity were characterized by analyzing the gas areal fraction (Fg), foam texture (nf), and velocity when foam flowed in the microfluidic porous medium at a constant gas pressure of 22 psi and varied liquid flow rates of 10 to 400 μL min−1, producing gas ratios (Rg) of 0.25–0.93. As Rg increased, Fg rose linearly from 0.50 to 0.94, while nf decreased, indicating coarser foam. Subdivision of Fg into trapped and mobile components revealed that higher Rg resulted in significantly greater gas trapping, with the trapped gas fraction (Fg,tb) increasing from 0.10 to 0.90. Spatial analysis showed that trapped bubbles exclusively occupied the low-permeable zone, while both trapped and mobile bubbles were present in the high-permeable region, which served as the primary flow pathway. Despite the changes in the foam structure, the average diameter of moving bubbles (![[D with combining macron]](https://www.rsc.org/images/entities/i_char_0044_0304.gif) mb) remained relatively stable (0.074–0.091 mm) for Rg ≤ 0.76. Their velocities (〈
mb) remained relatively stable (0.074–0.091 mm) for Rg ≤ 0.76. Their velocities (〈![[V with combining macron]](https://www.rsc.org/images/entities/i_char_0056_0304.gif) mb〉) ranged from 75 to 95 mm s−1 without a clear trend. However, the overall foam velocity (
mb〉) ranged from 75 to 95 mm s−1 without a clear trend. However, the overall foam velocity (![[V with combining macron]](https://www.rsc.org/images/entities/i_char_0056_0304.gif) f), estimated using the relative proportions of mobile and trapped gas, decreased with Rg, reflecting the growing impact of gas entrapment on flow resistance.
f), estimated using the relative proportions of mobile and trapped gas, decreased with Rg, reflecting the growing impact of gas entrapment on flow resistance.
Oil displacement experiments provided a comparative evaluation of recovery performance across different displacing fluids. Both SDS solution and CO2 foam formed stable displacement fronts in the high-permeable zone, whereas CO2 gas led to pronounced viscous fingering and very early breakthrough. For SDS solution, increasing Ql from 5 to 100 μL min−1 reduced Tbt from 6.6 to 1.0 s, but also decreased RFbt from 51.7% to 35.9%. CO2 gas yielded Tbt values between 0.02 and 0.18 s as Pg increased from 15 to 28 psi; however, RFbt remained consistently low (12–18%) due to rapid fingering and inefficient sweep. Foam flooding, by contrast, demonstrated delayed breakthrough and improved recovery: Tbt ranged from 1.52 to 0.54 s and RFbt from 69.1% to 44.2% as Rg increased from 0.57 to 0.98.
Although higher Rg improved bubble generation, it also promoted foam destabilization in contact with oil, likely due to the reduced surfactant availability and shear-thinning rheology. Nevertheless, CO2 foam injection significantly outperformed CO2 gas in recovery dynamics. Complete oil recovery (100%) was achieved within 31 s using CO2 foam, compared to over 150 s with CO2 gas—demonstrating the foam's ability to overcome capillary entry pressures in the low-permeable zone more efficiently. This improvement was attributed to foam lamellae that blocked high-permeability pathways, increased resistance, and redirected flow vertically into unswept low-permeable regions. Such mechanisms were absent in gas-only injection, which relied solely on pressure-driven flow and suffered from bypassing.
These findings highlight that CO2 foam's powerful potential for enhancing mobility control, sweep efficiency, and displacement depth in heterogeneous porous media. A promising recovery strategy could involve a sequential injection: first deploying SDS solution to establish a stable front in high-permeable channels, followed by CO2 foam to divert flow into low-permeable areas. Operational parameters—including surfactant concentration, gas/liquid flow rates, foam quality (Rg), and injection pressure—must be optimized based on desired outcomes, technical constraints, and reservoir conditions.
Future work should explore the effect of reservoir-relevant factors such as elevated temperature and pressure, brine salinity, crude oil properties, and surface mineralogy on foam stability and flow behavior. These factors are essential for scaling up microfluidic insights to practical EOR, aquifer remediation, and carbon sequestration applications.
Author contributions
Nikoo Moradpour: writing original draft, visualization, software, methodology, investigation, formal analysis. Peichun Amy Tsai: conceptualization, methodology, writing, review & editing, validation, visualization, supervision, funding acquisition.Conflicts of interest
There are no conflicts to declare.Data availability
Supplementary information: two videos are provided in the supplementary information (SI): Video: Fig2_Foam bubble characterization: bubble regimes generated by the flow-focusing geometry at varied gas pressures (Pg) and liquid flow rates (Ql). This is a representative video for the results in Fig. 2. Video: Fig4_Effect of gas ratio on bubble velocity: (left) trapped and moving bubbles in a representative porous medium at Rg = 0.25 and (right) moving bubbles (white) in the microfluidic porous medium at the gas ratios Rg = 0.25 (top), Rg = 0.63 (middle), and Rg = 0.93 (bottom). This is a representative video for the results in Fig. 4. See DOI: https://doi.org/10.1039/D5LC00544B.Data analysis was conducted using ImageJ Fiji and MATLAB.
The data supporting the findings of this study are available upon reasonable request.
5 Appendix
5.1 Permeability of the porous medium
The average permeability, kavg, is estimated by injecting pure water into the microfluidic chip and recording the pressure drop, ΔP, across the porous medium. The water injection flow rate is varied in the range of Q = 25–600 μl min−1. Fig. 12(a) shows the result of Q/A versus ΔP/μL. L and A are the length and cross-sectional area of the porous medium, and μ is the water viscosity. According to Darcy's law, the slope of the linear fit in this plot represents the permeability of the porous medium, kavg = 6.07 × 10−12 m2 or 6.07 Darcy.We used the Kozeny–Carman equation to find the ratio of the permeability of the low- and high- permeable layers using their hydraulic diameter, Dh, and porosity, ϕ.70,98Dh is defined as the pore volume/wetted area, indicated in Fig. 12(b).
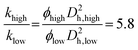 | (4) |
Given that the average permeability of two parallel layers (see Fig. 12(c)) can be determined using Darcy's law, the relationship is expressed as follows:
| Qtotal = Qhigh + Qlow, | (5) |
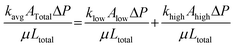 | (6) |
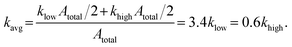 | (7) |
5.2 Darcy's law
Fluid flow through porous media can be described using Darcy's law:99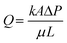 | (8) |
To estimate ΔP for oil displacement by SDS solution, Ql = 25 μl min−1, A = 6.3 × 10−7 m2, L = 1.4 m, K = 6.07 × 10−12 m2, and μ is very close to 1 cP at an SDS concentration of 0.5 wt%.91,92 Therefore, ΔP = 0.23 psi.
5.3 Contact-angle measurement results
Droplets of SDS solution (0.5 wt%) and ISOPAR M oil were placed on clean surfaces of bare glass slides and PDMS slabs. A high-resolution camera captured snapshots of the liquid droplets on the solid substrates. Fiji ImageJ software was used to find the left and right contact angles (inner side as shown in Fig. 13). The values were averaged to determine the reported ones.5.4 IFT measurement results
Interfacial tension (IFT) between the fluids was obtained using the pendant drop technique. For gas–liquid IFT measurements, a transparent cuvette was filled with the liquid phase and the gas bubbles were released using a syringe and a U-shaped needle. Oil droplets were released in the SDS solution similar to that above. The captured images were processed for drop shape analysis71,72 and the attained IFT values are listed in Fig. 14. | ||
| Fig. 14 The interfacial tension, σ, measured between different fluids (air, CO2, SDS solution, and ISOPAR M oil) using the pendant drop method. | ||
5.5 Oil displacement by CO2 foam
The raw and binarized temporal sequence of oil displacement by CO2 foam is displayed in Fig. 15. The foam is injected at Pg = 22 psi with Rg = 0.87. This figure represents the same experiment shown in Fig. 6(c).Acknowledgements
We gratefully acknowledge the support from the Canada First Research Excellence Fund (CFREF), the Future Energy System (FES T02-P05 CCUS projects) at the University of Alberta, and the Canada Foundation for Innovation (CFI 34546). P. A. T. holds a Canada Research Chair (CRC) in Fluids and Interfaces and gratefully acknowledges funding from the Natural Sciences and Engineering Research Council of Canada (NSERC), in particular the NSERC Canada Research Chairs Program (CRC 233147) and Discovery Grant (RGPIN-2020-05511).Notes and references
- D. L. Weaire and S. Hutzler, The physics of foams, Oxford University Press, 1999 Search PubMed.
- W. Drenckhan and S. Hutzler, Adv. Colloid Interface Sci., 2015, 224, 1–16 CrossRef CAS PubMed.
- L. L. Schramm, Emulsions, foams, suspensions, and aerosols: microscience and applications, John Wiley & Sons, 2014 Search PubMed.
- B. Dollet and C. Raufaste, C. R. Phys., 2014, 15, 731–747 CrossRef CAS.
- C. A. Conn, K. Ma, G. J. Hirasaki and S. L. Biswal, Lab Chip, 2014, 14, 3968–3977 RSC.
- K. Ma, R. Liontas, C. A. Conn, G. J. Hirasaki and S. L. Biswal, Soft Matter, 2012, 8, 10669–10675 RSC.
- P. Nguyen, H. Fadaei and D. Sinton, Energy Fuels, 2014, 28, 6221–6227 CrossRef CAS.
- A. Karthick, B. Roy and P. Chattopadhyay, J. Environ. Manage., 2019, 243, 187–205 CrossRef CAS PubMed.
- S. S. Datta, I. Battiato, M. A. Fernø, R. Juanes, S. Parsa, V. Prigiobbe, E. Santanach-Carreras, W. Song, S. L. Biswal and D. Sinton, Lab Chip, 2023, 23, 1358–1375 RSC.
- W. R. Rossen, R. Farajzadeh, G. J. Hirasaki and M. Amirmoshiri, SPE Improved Oil Recovery Conference?, 2022, p. D021S014R001.
- W. Wanniarachchi, P. Ranjith, M. Perera, T. Rathnaweera, D. Zhang and C. Zhang, Eng. Fract. Mech., 2018, 194, 117–135 CrossRef.
- T. Lu, Z. Li and L. Du, J. Chem. Eng., 2024, 481, 148575 CrossRef CAS.
- K. R. Chaturvedi, S. Bajpai, J. Trivedi and T. Sharma, Energy Fuels, 2022, 36, 5036–5046 CrossRef CAS.
- P. Liu, X. Zhang, Y. Wu and X. Li, J. Pet. Sci. Eng., 2017, 150, 208–216 CrossRef CAS.
- Z.-x. Liu, Y. Liang, Q. Wang, Y.-j. Guo, M. Gao, Z.-b. Wang and W.-l. Liu, J. Pet. Sci. Eng., 2020, 193, 107449 CrossRef CAS.
- C. McGlade, G. Sondak and M. Han, Whatever happened to enhanced oil recovery?, International energy agency technical report, 2018 Search PubMed.
- N. Moradpour, J. Yang and P. A. Tsai, Curr. Opin. Colloid Interface Sci., 2024, 101845 CrossRef CAS.
- T. Muther, H. A. Qureshi, F. I. Syed, H. Aziz, A. Siyal, A. K. Dahaghi and S. Negahban, J. Pet. Explor. Prod. Technol., 2022, 12, 1463–1488 CrossRef.
- R. Santos, W. Loh, A. Bannwart and O. Trevisan, Braz. J. Chem. Eng., 2014, 31, 571–590 CrossRef.
- N. Moradpour, R. Azadi and P. A. Tsai, Colloids Surf., A, 2025, 705, 135533 CrossRef CAS.
- H. Manikantan and T. M. Squires, J. Fluid Mech., 2020, 892, P1 CrossRef CAS PubMed.
- S. Xiao, Y. Zeng, E. D. Vavra, P. He, M. Puerto, G. J. Hirasaki and S. L. Biswal, Langmuir, 2018, 34, 739–749 CrossRef CAS PubMed.
- R. Phukan, S. B. Gogoi and P. Tiwari, Colloids Surf., A, 2020, 597, 124799 CrossRef CAS.
- K. R. Chaturvedi and T. Sharma, Chem. Eng. Sci., 2021, 235, 116484 CrossRef CAS.
- V. A. Lifton, Lab Chip, 2016, 16, 1777–1796 RSC.
- A. Anbari, H. T. Chien, S. S. Datta, W. Deng, D. A. Weitz and J. Fan, Small, 2018, 14, 1–15 CrossRef PubMed.
- K. Osei-Bonsu, N. Shokri and P. Grassia, J. Colloid Interface Sci., 2016, 462, 288–296 CrossRef CAS PubMed.
- M. J. Shojaei, K. Osei-Bonsu, S. Richman, P. Grassia and N. Shokri, Ind. Eng. Chem. Res., 2018, 58, 1068–1074 CrossRef.
- B. Géraud, S. A. Jones, I. Cantat, B. Dollet and Y. Méheust, Water Resour. Res., 2016, 52, 773–790 CrossRef.
- M. Simjoo, Y. Dong, A. Andrianov, M. Talanana and P. Zitha, Ind. Eng. Chem. Res., 2013, 52, 6221–6233 CrossRef CAS.
- G. Shi, K. Tang, F. Wang, Q. Luo, L. Bai, P. Sun and D. Zhu, Energy Fuels, 2020, 35, 465–472 CrossRef.
- S. Li, Q. Wang, K. Zhang and Z. Li, Fuel, 2020, 263, 116648 CrossRef CAS.
- D. Sinton, Lab Chip, 2014, 14, 3127–3134 RSC.
- J. Yang and P. A. Tsai, Chem. Eng. Sci., 2024, 300, 120543 CrossRef CAS.
- T. H. M. Ho and P. A. Tsai, Lab Chip, 2020, 20(20), 3806–3814 RSC.
- T. H. M. Ho, D. Sameoto and P. A. Tsai, Chem. Eng. Res. Des., 2021, 174, 116–126 CrossRef CAS.
- J. Yang, M. Saadat, I. Azizov, M. Dudek, G. Øye and P. A. Tsai, Lab Chip, 2022, 22(24), 4974–4983 RSC.
- J. Zhao, F. Torabi and J. Yang, Fuel, 2021, 287, 119443 CrossRef CAS.
- N. Yekeen, M. A. Manan, A. K. Idris, A. M. Samin and A. R. Risal, J. Pet. Sci. Eng., 2017, 159, 115–134 CrossRef CAS.
- M. Lv, Z. Liu, C. Ji, L. Jia and Y. Jiang, Ind. Eng. Chem. Res., 2018, 57, 15172–15180 CrossRef CAS.
- W. Yu, X. Zhou and M. Y. Kanj, Langmuir, 2022, 38, 2895–2905 CrossRef CAS PubMed.
- A. Gizzatov, S. Pierobon, Z. AlYousef, G. Jian, X. Fan, A. Abedini and A. I. Abdel-Fattah, Sci. Rep., 2021, 11, 3360 CrossRef CAS PubMed.
- Z. Xu, Z. Li, S. Cui, B. Li, D. Chen, Q. Zhang, L. Zheng and M. M. Husein, J. Pet. Sci. Eng., 2022, 211, 110170 CrossRef CAS.
- Q. Lv, R. Zheng, T. Zhou, X. Guo, W. Wang, J. Li and Z. Liu, Fuel, 2022, 330, 125533 CrossRef CAS.
- G. Jian, A. Gizzatov, M. Kawelah, Z. AlYousef and A. I. Abdel-Fattah, Appl. Energy, 2021, 292, 116815 CrossRef CAS.
- B. Benali, T. L. Føyen, Z. P. Alcorn, M. Haugen, J. Gauteplass, A. R. Kovscek and M. A. Fernø, Int. J. Greenhouse Gas Control, 2022, 114, 103607 CrossRef CAS.
- L. F. Lopes, J. M. Façanha, L. Maqueira, F. R. Ribeiro and A. Pérez-Gramatges, J. Pet. Sci. Eng., 2021, 207, 109141 CrossRef CAS.
- W. Yan, C. A. Miller and G. J. Hirasaki, Colloids Surf., A, 2006, 282, 348–359 CrossRef.
- S. Jones, N. Getrouw and S. Vincent-Bonnieu, Soft Matter, 2018, 14, 3497–3503 RSC.
- A. Kovscek, T. Patzek and C. Radke, Chem. Eng. Sci., 1995, 50, 3783–3799 CrossRef CAS.
- N. Nazari and A. R. Kovscek, Lab Chip, 2022, 22, 3489–3498 RSC.
- W. R. Rossen, Colloids Surf., A, 2003, 225, 1–24 CrossRef CAS.
- J. Alvarez, H. Rivas and W. Rossen, SPE J., 2001, 6, 325–333 CrossRef.
- Y. Wang, M. Puerto, C. Bai, K. Ma, K. Mateen, G. J. Hirasaki and S. L. Biswal, Ind. Eng. Chem. Res., 2025, 64, 2995–3003 CrossRef CAS.
- X. Zheng, N. Mahabadi, T. S. Yun and J. Jang, J. Geophys. Res.: Solid Earth, 2017, 122, 1634–1647 CrossRef CAS.
- Q. Sun, Z. Li, S. Li, L. Jiang, J. Wang and P. Wang, Energy Fuels, 2014, 28, 2384–2394 CrossRef CAS.
- F. Guo, S. A. Aryana, Y. Wang, J. F. McLaughlin and K. Coddington, Int. J. Greenhouse Gas Control, 2019, 87, 134–141 CrossRef CAS.
- F. Guo and S. A. Aryana, J. Pet. Sci. Eng., 2018, 164, 155–163 CrossRef CAS.
- D. Li, G. Xin and S. Ren, ACS Omega, 2022, 7, 36503–36509 CrossRef CAS PubMed.
- S. Jones, N. Getrouw and S. Vincent-Bonnieu, Soft Matter, 2018, 14, 3490–3496 RSC.
- A. E. Peksa, K.-H. A. Wolf and P. L. Zitha, Mar. Pet. Geol., 2015, 67, 701–719 CrossRef.
- B. A. Abuamarah and B. S. Nabawy, J. Nat. Gas Sci. Eng., 2021, 88, 103807 CrossRef.
- P.-R. Thomson, M. Jefferd, B. L. Clark, D. Chiarella, T. M. Mitchell and S. Hier-Majumder, Mar. Pet. Geol., 2020, 122, 104614 CrossRef.
- P.-R. Thomson, R. Ellis, D. Chiarella and S. Hier-Majumder, Front. Earth Sci., 2020, 8, 246 CrossRef.
- M. Kashif, Y. Cao, G. Yuan, M. Asif, K. Javed, J. N. Mendez, D. Khan and L. Miruo, Pet. Sci., 2019, 16, 981–1000 CrossRef CAS.
- S. Ma, G. Jin, R. Sy and H. Kesserwan, SPE Kingdom of Saudi Arabia Annual Technical Symposium and Exhibition, 2018, p. SPE-192220.
- R. Kareem, P. Cubillas, J. Gluyas, L. Bowen, S. Hillier and H. C. Greenwell, J. Pet. Sci. Eng., 2017, 149, 436–455 CrossRef CAS.
- M. Wetzel, T. Kempka and M. Kühn, Minerals, 2021, 11, 151 CrossRef CAS.
- H. Safari, B. J. Balcom and A. Afrough, Comput. Geosci., 2021, 156, 104895 CrossRef.
- E. J. Peters, Advanced Petrophysics: Geology, porosity, absolute permeability, heterogeneity, and geostatistics, Greenleaf Book Group, 2012, vol. 1 Search PubMed.
- J. D. Berry, M. J. Neeson, R. R. Dagastine, D. Y. Chan and R. F. Tabor, J. Colloid Interface Sci., 2015, 454, 226–237 CrossRef CAS PubMed.
- A. Esfandiarian, A. Maghsoudian, A. Davarpanah, Y. Tamsilian and S. Kord, J. Pet. Sci. Eng., 2022, 215, 110607 CrossRef CAS.
- N. D. Denkov, K. G. Marinova and S. S. Tcholakova, Adv. Colloid Interface Sci., 2014, 206, 57–67 CrossRef CAS PubMed.
- R. Aveyard, B. Binks, P. Fletcher, T. Peck and C. Rutherford, Adv. Colloid Interface Sci., 1994, 48, 93–120 CrossRef CAS.
- J. Broeke, J. M. M. Pérez and J. Pascau, Image processing with ImageJ, Packt Publishing Ltd, 2015 Search PubMed.
- T. Ferreira and W. Rasband, ImageJ user guide, ImageJ/Fiji, 2012 Search PubMed.
- P. Garstecki, I. Gitlin, W. DiLuzio, G. M. Whitesides, E. Kumacheva and H. A. Stone, Appl. Phys. Lett., 2004, 85, 2649–2651 CrossRef CAS.
- N. Dietrich, S. Poncin, N. Midoux and H. Z. Li, Langmuir, 2008, 24, 13904–13911 CrossRef CAS PubMed.
- T. Fu, D. Funfschilling, Y. Ma and H. Z. Li, Microfluid. Nanofluid., 2010, 8, 467–475 CrossRef CAS.
- S. Cleve, A. Lassus, C. Diddens, B. Van Elburg, E. Gaud, S. Cherkaoui, M. Versluis, T. Segers and G. Lajoinie, J. Fluid Mech., 2023, 972, A27 CrossRef.
- B. Géraud, Y. Méheust, I. Cantat and B. Dollet, Phys. Rev. Lett., 2017, 118, 098003 CrossRef PubMed.
- Z. Huang, M. Su, Q. Yang, Z. Li, S. Chen, Y. Li, X. Zhou, F. Li and Y. Song, Nat. Commun., 2017, 8, 1–9 CrossRef PubMed.
- Y. Zhang, W. Geng, M. Chen, X. Xu, L. Jiang and Y. Song, Energy Fuels, 2023, 37, 19695–19703 CrossRef CAS.
- H. M. Polat, F. M. Coelho, T. J. Vlugt, L. F. Mercier Franco, I. N. Tsimpanogiannis and O. A. Moultos, J. Chem. Eng. Data, 2024, 69, 3296–3329 CrossRef CAS PubMed.
- J. J. Carroll, J. D. Slupsky and A. E. Mather, J. Phys. Chem. Ref. Data, 1991, 20, 1201–1209 CrossRef CAS.
- L. W. Diamond and N. N. Akinfiev, Fluid Phase Equilib., 2003, 208, 265–290 CrossRef CAS.
- S. Liu, X. Dou, Q. Zeng and J. Liu, J. Pet. Sci. Eng., 2021, 196, 107635 CrossRef CAS.
- S. Ansari and D. S. Nobes, Fluids, 2022, 7, 333 CrossRef.
- F. Mo, Z. Du, X. Peng, Y. Tang and H. Sun, J. Nat. Gas Sci. Eng., 2017, 44, 314–327 CrossRef.
- D. D. Huang and M. M. Honarpour, J. Pet. Sci. Eng., 1998, 19, 103–117 CrossRef CAS.
- T. P. Niraula, S. K. Shah, S. K. Chatterjee and A. Bhattarai, Karbala Int. J. Mod. Sci., 2018, 4, 26–34 CrossRef.
- Y. Chai, X. Li, J. Geng, J. Pan, Y. Huang and D. Jing, Colloid Polym. Sci., 2019, 297, 1025–1035 CrossRef CAS.
- H. B. Todd and J. G. Evans, SPE Rocky Mountain Petroleum Technology Conference/Low-Permeability Reservoirs Symposium, 2016, p. SPE-180270 Search PubMed.
- L. Xue, P. Liu and Y. Zhang, Water, 2023, 15, 1342 CrossRef CAS.
- L. Zhao, T. Liu, F. Lu, J. Sun and Y. Yao, ACS Omega, 2024, 9, 28505–28519 CrossRef CAS PubMed.
- W. Osterloh and M. Jante Jr, SPE Improved Oil Recovery Conference?, 1992, p. SPE-24179.
- R. Gajbhiye and S. Kam, Chem. Eng. Sci., 2011, 66, 1536–1549 CrossRef CAS.
- P. C. Carman, Chem. Eng. Res. Des., 1997, 75, S32–S48 CrossRef.
- T. Ahmed, Reservoir engineering handbook, Gulf professional publishing, 2018 Search PubMed.
| This journal is © The Royal Society of Chemistry 2025 |