Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Photo-promoted carbonylative carboxylation of alkenes to synthesize β-alkyl ketocarboxylic acid derivatives with CO and CO2 as two mixed C1 gaseous molecules
Hefei
Yang
ab,
Le-Cheng
Wang
ab,
Yuanrui
Wang
a and
Xiao-Feng
Wu
 *ab
*ab
aDalian National Laboratory for Clean Energy, Dalian Institute of Chemical Physics, Chinese Academy of Sciences, 116023 Dalian, Liaoning, China. E-mail: xwu2020@dicp.ac.cn
bLeibniz-Institut für Katalyse e.V., 18059 Rostock, Germany
First published on 7th November 2025
Abstract
Herein, we report a photocatalytic carbonylative carboxylation strategy for alkenes, achieving the cooperative utilization of both CO and CO2 as C1 building blocks within a single reaction system. This method enables the efficient synthesis of β-alkyl ketocarboxylic acid derivatives from abundant and low-cost starting materials under mild conditions.
Green foundation1. Utilization of both CO2 (greenhouse gas) and CO as C1 building blocks.2. A new photo-induced four-component carbonylation reaction toward efficient synthesis of β-alkyl ketocarboxylic acid derivatives. 3. Improvement of the reaction efficiency and lower CO pressure for further research. |
Due to environmental concerns and the need for sustainable development, the efficient utilization of chemical resources has become a critical priority. Carbon capture, storage, and utilization (CCSU) has also emerged as a burgeoning and urgent field of research.1 The carbonyl functional group, owing to its diverse forms of existence and unique catalytic properties, is widely present in various natural products and small-molecule pharmaceuticals.2–5 Carbon monoxide (CO) is an inexpensive and versatile C1 synthon that has become a key tool in the synthesis of a wide range of carbonylated compounds.6–11 In comparison, carbon dioxide (CO2), as another ideal C1 building block in chemical synthesis, represents a promising carbon utilization strategy through its conversion into value-added chemicals, leveraging its sustainability, low toxicity, and abundance.12–17 Despite carbon monoxide and carbon dioxide being used as C1 building blocks in equal measure, to our knowledge, to date few reaction studies have reported the simultaneous use of the two gases as different C1 modules for building compounds, due to their inherent differences in reactivity.
In recent years, with the development of novel transformation methods such as visible-light irradiation18,19 and transition-metal catalysis,20–22 extensive research has been conducted regarding the transformation and utilization of carbon monoxide (CO). Due to its unique electronic structure, reactions involving CO can be categorized into four main types: (1) through electrophilic acylmetal species;23,24 (2) through acyl cations initiated by strong acids;25 (3) through acyl anions formed by strong bases;26 and (4) through acyl radical intermediates by radical trapping.27,28 In contrast to carbon monoxide (CO), the carboxylation of carbon dioxide (CO2) is recognized as an important method for directly accessing carboxylic acids. However, its application presents challenges due to its thermodynamic stability and kinetic inertness.29,30 In classical CO2 carboxylation reactions, three strategies have been developed: generation of a transition metal-coordinated nucleophilic carboxyl metal species;31–34 direct SET-reduction of CO2 to CO2−;35–37 formation of a carbon negative ion, in which the SET process reduces the carbon radical (CO2 acts as an electrophilic reagent);38–42 intermediates are often produced as the first step and then quenched with hydrochloric acid to afford a series of carboxylate products (Fig. 1A). Despite these achievements in the two C1 modules, introducing two gaseous molecules simultaneously is still highly challenging. Leveraging the rapid development of alkene difunctionalization, in 2022, Wu and colleagues reported a copper-catalyzed 1,2-dicarbonylative cyclization reaction of alkenes, successfully incorporating two molecules of CO into the system.43 In the same year, Yu's team successfully achieved the dicarboxylation reaction of alkenes by introducing two CO2 gaseous molecules.44 From this perspective, utilizing CO and CO2 as inexpensive feedstocks for directly obtaining carbonyl and carboxyl groups, we designed a strategy to simultaneously incorporate both as carbonyl and carboxyl sources at the two termini of an alkene. This represents an unprecedented attempt (Fig. 1B). From the viewpoint of advancing green synthetic methodologies, exploring this new pathway for the simultaneous utilization of two C1 building blocks is valuable.
We have been attracted to developing a novel, milder, and greener reaction mode for the simultaneous introduction of both CO and CO2. The key to its success lies in achieving compatibility with the distinct properties of both C1 building blocks. The halogen atom transfer (XAT) strategy is an alternative to classical two-electron disconnection and provides a way to convert versatile alkyl halides into other valuable chemicals.45 Moreover, the reaction does not need to overcome the strong reducing nature of the C(sp3)–halogen bond (Ered ≥ −2.0 V vs. SCE) based on the XAT method, making it less dependent on the participation of transition metals. In recent years, α-aminoalkyl radicals have been used in XAT reactions involving various halides to produce the desired radicals. However, its own SET-reducing properties have been overlooked.46–48 Therefore, we endeavor to employ α-aminoalkyl radicals as mediators to bridge the reactions of CO and CO2. On the one hand, the desired radical species obtained via XAT can trap CO. On the other hand, the carbon radical precursor undergoes SET reduction to generate a carbanion, which then nucleophilically attacks CO2. In this report, we present a novel photocatalyzed carbonylative carboxylation of alkenes utilizing the combined incorporation of both CO and CO2 as C1 building blocks (Fig. 1C).
We initiated our studies on the dual C1 synthon strategy by evaluating the effects of various conditions on a model reaction by using 1,1-diphenylethylene (1a) and iodocyclohexane (2a) as substrates. After systematically compiling the reaction optimization data, the optimized conditions are summarized in Fig. 2 (see the SI for additional details). At the initial reaction stage, the occurrence of the reaction was successfully confirmed under the following conditions: using 3DPA2FBN as the catalyst, with the addition of Cy2NMe and Na2CO3, under a mixed CO/CO2 atmosphere, and irradiation with 15 W blue LEDs (Fig. 2, entry 1). When other solvents were substituted for DMSO, the yield of the desired product decreased (Fig. 2, entry 2). Subsequently, during our exploration of different catalysts, the use of Ir(dF(CF3)ppy)2(dtbbpy)PF6 increased the yield of 3a to 71% (Fig. 2, entry 3). Further screening of alternative bases failed to improve the yield of 3a (Fig. 2, entry 4). Upon exploration of diverse α-amino alkyl radicals, significantly fluctuating yields were observed, further confirming their crucial role in the reaction (Fig. 2, entry 5). Moreover, adjustment of the gas pressure ratio resulted in reduced yields (Fig. 2, entries 6 and 7). Finally, control experiments under light-exclusion conditions completely suppressed the reaction (Fig. 2, entry 8). It is worth noting that no desired reaction occurred in the absence of an organic base, and the yield decreased when the reaction time was shortened to 12 hours with incomplete conversion of substrates. Importantly, the obtained carboxylic acid product was methylated for easier purification.
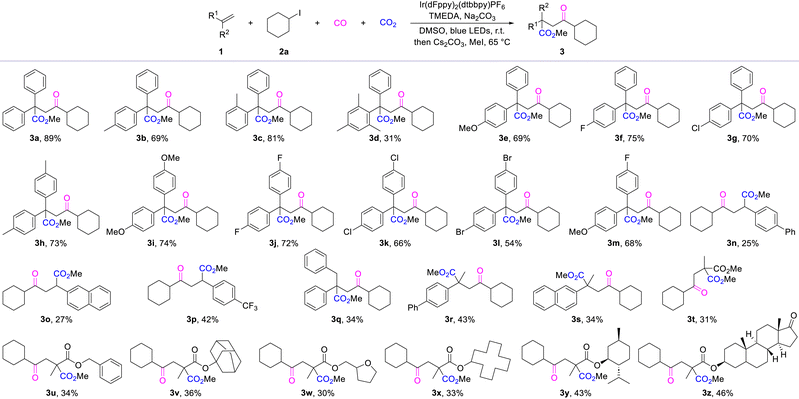
Having determined the optimal reaction conditions, we assessed the versatility of this novel olefinic carbonylative carboxylation by using a range of alkenes and representative iodocyclohexane under standard conditions (Table 1). From the experimental data, it can be observed that when adding substituents to a single phenyl group of an alkene, small steric hindrance does not cause a spatial effect on the reaction outcome, but as the steric hindrance effect increases significantly, the reaction yield decreases (3a–3d). Moreover, the corresponding products were obtained in good yields when the single phenyl ring bore substituents with varying electronic properties (3e–3g). Subsequently, when distinct functional groups were introduced onto each of the two phenyl rings, the target products were still obtained in moderate to good yields (3h–3m). These results demonstrate that the reaction exhibits good tolerance towards both electronic effects and steric hindrance in the substrates. To further expand the scope of alkene types, we also investigated alkenes bearing a single phenyl substituent. A decrease in reaction yield was observed for these substrates (3n–3o), which we postulate may arise from yield variations induced by differences in radical stability. Notably, when a CF3 group was incorporated onto the phenyl ring or methyl substitution was introduced at the α-position of the alkene, a clear increase in the yield of the desired product was observed (3p–3s). Notably, this protocol also proved applicable to α-methacrylate substrates, affording the target products in moderate yields (3t–3x). Finally, we successfully extended this methodology to the modification of bioactive molecular scaffolds, obtaining the desired products in good yields as well (3y–3z).
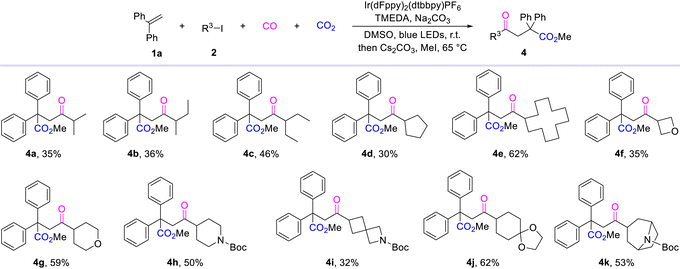
To further demonstrate the compatibility of this reaction, we explored the categories of alkyl iodides (Table 2). From the obtained results, we can observe that the reaction has good tolerance for small molecule iodinated alkanes, and the target products (4a–4c) are obtained in moderate yields. By contrast, a significant increase in yield was obtained when iodinated cyclic alkanes replaced chain alkanes (4d–4k). At the same time, the results show that, although the reaction is compatible with iodinated cycloalkanes containing heteroatoms, the presence of these heteroatoms still impacts the reaction yield (4f). This effect gradually decreases as the ring size increases and the distance from the heteroatom increases. Notably, the procedure can also be easily scaled up by performing the same reaction in several parallel vials in one autoclave. However, only a trace amount of the corresponding product was detected when tert-butyl iodide was tested as the substrate.
To gain a deeper understanding of the reaction pathway, we performed some control experiments (Fig. 3). First, adding TEMPO (2,2,6,6-tetramethylpiperidin-1-oxyl) to the system under standard conditions showed that the reaction was significantly inhibited. This suggests that the reaction probably proceeds via a radical pathway that competes with the breaking of the olefinic C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bond in the reaction (Fig. 3a). To verify the initiation step of the reaction and determine whether it undergoes a chain process, photocatalyst-free alkyl-generation conditions were used, and the presence of a reducing agent was excluded. We used catalytic (20 mol%) and equivalent (1.5 eq.) amounts of K2S2O8 as XAT initiators, respectively, but the target products were not obtained. However, the generation of 5a, 5b and 5c was successfully detected, suggesting that the XAT reagent was initiated in the first step and excluding the chain reaction process (Fig. 3b). When D2O was added to the reaction system, the presence of 90% deuterated H was observed at the benzyl position of the target product. This suggests that the reaction undergoes a benzyl carbon-negative process (Fig. 3c). This control experiment confirms the position of inserted CO and the source of the carbonyl group. Notably, a carboxylation reaction with CO2 was also reported in the literature, which then confirms the position of inserted CO2.49 Subsequently, to investigate the stability of the product, substrate 7 was subjected to the standard conditions. It was found that without the addition of TMEDA, compound 5a was obtained in 22% yield. Conversely, decarboxylation was markedly inhibited in the presence of TMEDA, with a 94% recovery of the starting material (Fig. 3d). To demonstrate the feasibility of this approach, we derivatized the product (Fig. 3e). The carboxylic acid product and the diol compound were obtained at high yields under suitable reaction conditions.
C bond in the reaction (Fig. 3a). To verify the initiation step of the reaction and determine whether it undergoes a chain process, photocatalyst-free alkyl-generation conditions were used, and the presence of a reducing agent was excluded. We used catalytic (20 mol%) and equivalent (1.5 eq.) amounts of K2S2O8 as XAT initiators, respectively, but the target products were not obtained. However, the generation of 5a, 5b and 5c was successfully detected, suggesting that the XAT reagent was initiated in the first step and excluding the chain reaction process (Fig. 3b). When D2O was added to the reaction system, the presence of 90% deuterated H was observed at the benzyl position of the target product. This suggests that the reaction undergoes a benzyl carbon-negative process (Fig. 3c). This control experiment confirms the position of inserted CO and the source of the carbonyl group. Notably, a carboxylation reaction with CO2 was also reported in the literature, which then confirms the position of inserted CO2.49 Subsequently, to investigate the stability of the product, substrate 7 was subjected to the standard conditions. It was found that without the addition of TMEDA, compound 5a was obtained in 22% yield. Conversely, decarboxylation was markedly inhibited in the presence of TMEDA, with a 94% recovery of the starting material (Fig. 3d). To demonstrate the feasibility of this approach, we derivatized the product (Fig. 3e). The carboxylic acid product and the diol compound were obtained at high yields under suitable reaction conditions.
Based on the experimental results, we proposed a possible reaction mechanism for this transformation (Fig. 4). First, the iridium photocatalyst is activated by light irradiation. The excited-state photocatalyst Ir* oxidizes α-aminoalkane I and then deprotonates to form an α-amino carbon radical II and an IrII complex. Then, the radical species II generates an alkyl radical B from iodinated alkane A through the XAT strategy. Subsequently, CO was captured by the radical B to obtain an acyl radical species C. A relatively stable benzyl radical D was formed after the addition of the radical C to an alkene, which was then further reduced to a benzyl carbon anion E under the action of the IrII complex. Finally, the carbon anion E reacted with CO2 through nucleophilic addition to afford the target product F. Meanwhile, the iridium photocatalyst is regenerated for the next catalytic cycle.
Conclusions
In summary, we have developed a new carbonylative carboxylation strategy for alkenes that enables the cooperative utilization of both CO and CO2 as C1 building blocks within a single reaction system through visible-light photocatalysis. This method demonstrates excellent substrate compatibility, enabling the synthesis of high-value β-alkyl ketocarboxylic acid derivatives from abundant and low-cost starting materials under mild conditions. It thus provides a new environmentally benign approach for the efficient utilization of C1 resources and expands the synthetic applications of dual C1 building blocks.Author contributions
X.-F. W. conceived and directed the project. H. Y., L. C. W. and Y. W. performed all the experiments and prepared the manuscript and the SI. X.-F. W. and H. Y. revised the manuscript.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: general comments, general procedure, analytical data and NMR spectra. See DOI: https://doi.org/10.1039/d5gc04231c.Acknowledgements
We acknowledge the financial support from the National Key R&D Program of China (2023YFA1507500) and the Dalian Institute of Chemical Physics (DICP).References
- D.-G. Yu and L.-N. He, Green Chem., 2021, 23, 3499–3501 RSC.
- (a) M. Yang, Y. Liu, X. Qi, Y. Zhao and X.-F. Wu, Green Synth. Catal., 2024, 5, 211–269 CAS; (b) L.-C. Wang and X.-F. Wu, Acc. Chem. Rev., 2025, 58, 1036–1050 CrossRef CAS PubMed; (c) Y. Wang, Z.-P. Bao, X.-D. Mao, M. Hou and X.-F. Wu, Chem. Soc. Rev., 2025, 54, 9530–9573 RSC.
- S. Sumino, A. Fusano, T. Fukuyama and I. Ryu, Acc. Chem. Res., 2014, 47, 1563–1574 CrossRef CAS.
- J. Yang, J. Liu, H. Neumann, R. Franke, R. Jackstell and M. Beller, Science, 2019, 366, 1514–1517 CrossRef CAS PubMed.
- I. Ziccarelli, R. Mancuso, F. Giacalone, C. Calabrese, V. La Parola, A. De Salvo, N. Della Ca, M. Gruttadauria and B. Gabriele, J. Catal., 2022, 413, 1098–1110 CrossRef CAS.
- M. Beller, Catalytic carbonylation reactions, Springer, Berlin, 2006 Search PubMed.
- M. Beller and X.-F. Wu, Transition metal catalyzed carbonylation reactions: carbonylative activation of C-X bonds, Springer, Berlin, 2013 Search PubMed.
- A. Börner and R. Franke, Hydroformylation: fundamentals, processes, and applications in organic synthesis, Wiley-VCH, Weinheim, 2016 Search PubMed.
- J.-B. Peng, F.-P. Wu and X.-F. Wu, Chem. Rev., 2018, 119, 2090–2127 CrossRef PubMed.
- H. Matsubara, T. Kawamoto, T. Fukuyama and I. Ryu, Acc. Chem. Res., 2018, 51, 2023–2035 CrossRef CAS PubMed.
- L.-J. Cheng and N. P. Mankad, Chem. Soc. Rev., 2020, 49, 8036–8064 RSC.
- Q. Liu, L. Wu, R. Jackstell and M. Beller, Nat. Commun., 2015, 6, 5933 CrossRef.
- A. Tortajada, F. Juliá-Hernández, M. Börjesson, T. Moragas and R. Martin, Angew. Chem., Int. Ed., 2018, 57, 15948–15982 CrossRef CAS PubMed.
- B. Grignard, S. Gennen, C. Jérôme, A. W. Kleij and C. Detrembleur, Chem. Soc. Rev., 2019, 48, 4466–4514 RSC.
- M. D. Burkart, N. Hazari, C. L. Tway and E. L. Zeitler, ACS Catal., 2019, 9, 7937–7956 CrossRef CAS.
- L. Zhang, Z. Li, M. Takimoto and Z. Hou, Chem. Rec., 2019, 20, 494–512 CrossRef.
- N. Iwasawa, Bull. Chem. Soc. Jpn., 2023, 96, 824–841 CrossRef.
- X.-W. Gu, Y. Zhang, F. Zhao, H.-J. Ai and X.-F. Wu, Chin. J. Catal., 2023, 48, 214–223 CrossRef.
- J. B. Peng, X. Qi and X. F. Wu, ChemSusChem, 2016, 9, 2279–2283 CrossRef PubMed.
- S. Zhang, H. Neumann and M. Beller, Chem. Soc. Rev., 2020, 49, 3187–3210 RSC.
- H.-Q. Geng and X.-F. Wu, Synthesis, 2024, 2595–2613 Search PubMed.
- X.-W. Gu and X.-F. Wu, Org. Chem. Front., 2023, 10, 1587–1591 RSC.
- M. Zhou, H.-Y. Zhao, S. Zhang, Y. Zhang and X. Zhang, J. Am. Chem. Soc., 2020, 142, 18191–18199 CrossRef PubMed.
- C. S. Day, S. J. Ton, C. Kaussler, D. Vrønning Hoffmann and T. Skrydstrup, Angew. Chem., Int. Ed., 2023, 62, e202308238 CrossRef PubMed.
- L. Omann, Z. W. Qu, E. Irran, H. F. T. Klare, S. Grimme and M. Oestreich, Angew. Chem., Int. Ed., 2018, 57, 8301–8305 CrossRef PubMed.
- H. Kai, K. Iwamoto, N. Chatani and S. Murai, J. Am. Chem. Soc., 1996, 118, 7634–7635 CrossRef.
- Y. Ding, J. Wu and H. Huang, J. Am. Chem. Soc., 2023, 145, 4982–4988 CrossRef PubMed.
- G. M. Torres, Y. Liu and B. A. Arndtsen, Science, 2020, 368, 318–323 CrossRef PubMed.
- C. S. Yeung, Angew. Chem., Int. Ed., 2019, 58, 5492–5502 CrossRef PubMed.
- (a) J.-H. Ye, T. Ju, H. Huang, L.-L. Liao and D.-G. Yu, Acc. Chem. Res., 2021, 54, 2518–2531 CrossRef PubMed; (b) A.-G. Wu, J. Ding, L. Zhao, H.-R. Li and L.-N. He, Chem. Rec., 2024, 24, e202400164 CrossRef CAS PubMed; (c) L.-Q. Qiu, H.-R. Li and L.-N. He, Acc. Chem. Res., 2023, 56, 2225–2240 CrossRef CAS PubMed; (d) Q.-W. Song, R. Ma, P. Liu, K. Zhang and L.-N. He, Green Chem., 2023, 25, 6538–6560 RSC.
- R. D. He and Y. Lu, Angew. Chem., Int. Ed., 2025, 64, e202424790 CrossRef CAS PubMed.
- K. Ukai, M. Aoki, J. Takaya and N. Iwasawa, J. Am. Chem. Soc., 2006, 128, 8706–8707 CrossRef CAS.
- A. Correa and R. Martín, J. Am. Chem. Soc., 2009, 131, 15974–15975 CrossRef CAS PubMed.
- J. Hong, M. Li, J. Zhang, B. Sun and F. Mo, ChemSusChem, 2019, 12, 6–39 CrossRef CAS.
- J. H. Ye, M. Miao, H. Huang, S. S. Yan, Z. B. Yin, W. J. Zhou and D. G. Yu, Angew. Chem., Int. Ed., 2017, 56, 15416–15420 CrossRef CAS PubMed.
- H. Huang, J.-H. Ye, L. Zhu, C.-K. Ran, M. Miao, W. Wang, H. Chen, W.-J. Zhou, Y. Lan, B. Yu and D.-G. Yu, CCS Chem., 2021, 3, 1746–1756 CrossRef CAS.
- W. Zhang, Z. Chen, Y.-X. Jiang, L.-L. Liao, W. Wang, J.-H. Ye and D.-G. Yu, Nat. Commun., 2023, 14, 3529 CrossRef CAS PubMed.
- V. R. Yatham, Y. Shen and R. Martin, Angew. Chem., Int. Ed., 2017, 56, 10915–10919 CrossRef CAS.
- L.-L. Liao, G.-M. Cao, Y.-X. Jiang, X.-H. Jin, X.-L. Hu, J. J. Chruma, G.-Q. Sun, Y.-Y. Gui and D.-G. Yu, J. Am. Chem. Soc., 2021, 143, 2812–2821 CrossRef CAS.
- L. Song, D. M. Fu, L. Chen, Y. X. Jiang, J. H. Ye, L. Zhu, Y. Lan, Q. Fu and D. G. Yu, Angew. Chem., Int. Ed., 2020, 59, 21121–21128 CrossRef CAS PubMed.
- J. Hou, A. Ee, H. Cao, H. W. Ong, J. H. Xu and J. Wu, Angew. Chem., Int. Ed., 2018, 57, 17220–17224 CrossRef CAS.
- H. Wang, Y. Gao, C. Zhou and G. Li, J. Am. Chem. Soc., 2020, 142, 8122–8129 CrossRef CAS PubMed.
- F. Zhao, X. W. Gu, R. Franke and X. F. Wu, Angew. Chem., Int. Ed., 2022, 61, e202214812 CrossRef CAS PubMed.
- T. Ju, Y.-Q. Zhou, K.-G. Cao, Q. Fu, J.-H. Ye, G.-Q. Sun, X.-F. Liu, L. Chen, L.-L. Liao and D.-G. Yu, Nat. Catal., 2021, 4, 304–311 CrossRef CAS.
- F. Juliá, T. Constantin and D. Leonori, Chem. Rev., 2021, 122, 2292–2352 CrossRef.
- X.-Y. Wang, Y.-Q. He, Y. Zhou, L. Lu, X.-R. Song, Z.-Z. Zhou, W.-F. Tian and Q. Xiao, Org. Lett., 2023, 25, 3847–3852 CrossRef CAS.
- B. Górski, A.-L. Barthelemy, J. J. Douglas, F. Juliá and D. Leonori, Nat. Catal., 2021, 4, 623–630 CrossRef.
- Z. Zhang, B. Górski and D. Leonori, J. Am. Chem. Soc., 2022, 144, 1986–1992 CrossRef CAS.
- (a) L.-L. Liao, G.-M. Cao, Y.-X. Jiang, X.-H. Jin, X.-L. Hu, J. J. Chruma, G.-Q. Sun, Y.-Y. Gui and D.-G. Yu, J. Am. Chem. Soc., 2021, 143, 2812–2821 CrossRef CAS; (b) W. Zhang, Z. Chen, Y.-X. Jiang, L.-L. Liao, W. Wang, J.-H. Ye and D.-G. Yu, Nat. Commun., 2023, 14, 3529 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2025 |



![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Reaction conditions: 1a (0.2 mmol), 2a (1.5 equiv.), 3DPA2FBN (2 mol%), Cy2NMe (1.5 equiv.), Na2CO3 (1.5 equiv.), CO2 (10 bar), CO (40 bar) in DMSO (1 mL), irradiation with 15 W blue LEDs at rt for 24 hours. Yields were determined by GC-FID analysis using
Reaction conditions: 1a (0.2 mmol), 2a (1.5 equiv.), 3DPA2FBN (2 mol%), Cy2NMe (1.5 equiv.), Na2CO3 (1.5 equiv.), CO2 (10 bar), CO (40 bar) in DMSO (1 mL), irradiation with 15 W blue LEDs at rt for 24 hours. Yields were determined by GC-FID analysis using