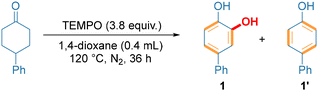Open Access Article
Open Access ArticleSynthesis of catechols from cyclohexanones via acid-regulated dual oxidative transformations with TEMPO†
Biping
Xu
 ab,
Xiaojie
Liu
ab,
Xiaojie
Liu
 ab,
Lei
Deng
a,
Yaping
Shang
ab,
Lei
Deng
a,
Yaping
Shang
 ab,
Xiaoming
Jie
ab,
Xiaoming
Jie
 *ab and
Weiping
Su
*ab and
Weiping
Su
 *ab
*ab
aState Key Laboratory of Structural Chemistry, Fujian Institute of Research on the Structure of Matter, Chinese Academy of Sciences, 155 Yangqiao Road West, Fuzhou 350002, China. E-mail: jiexiaoming@fjirsm.ac.cn; wpsu@fjirsm.ac.cn
bFujian Science & Technology Innovation Laboratory for Optoelectronic Information of China, Fuzhou, Fujian 350108, P. R. China
First published on 26th March 2025
Abstract
Herein, we disclosed a metal-free, operationally simple, general method for the straightforward synthesis of catechols via an α-oxygenation of cyclohexanone-initiated cascade oxidative reaction sequence. This metal-free method was particularly advantageous for the synthesis of pharmaceutically relevant catechols, as it eliminated the need for costly and potentially toxic metal catalysts. An exhibition of more than 60 cyclohexanone substrates proved the reliability of this protocol in directly synthesizing structurally diverse and synthetically useful bioactive molecules, which are conventionally unattainable by existing methods.
Green foundation1. This work advances the field of green chemistry by developing a novel, metal-free approach for the synthesis of catechols from cyclohexanones using inexpensive and bench-stable TEMPO and TsOH. This method eliminates the need for toxic and expensive metal catalysts and offers a broad substrate scope, providing a sustainable alternative for synthesizing pharmaceutically relevant catechols. Moreover, TEMPO serves not only as an oxidant in the reaction but also as an oxygen donor, incorporating oxygen atoms into the target catechol, thereby enhancing atom economy.2. The operationally simple protocol achieves high selectivity and efficiency, transforming readily available cyclohexanones into diverse catechols with over 60 substrates. It demonstrates excellent functional group tolerance and avoids hazardous reagents, aligning with the principles of green chemistry by minimizing waste, enhancing atom economy, and eliminating the use of toxic metals. 3. Future research could explore catalytic loading of nitroxides and acids by using environment-friendly terminal oxidants, such as air and O2. |
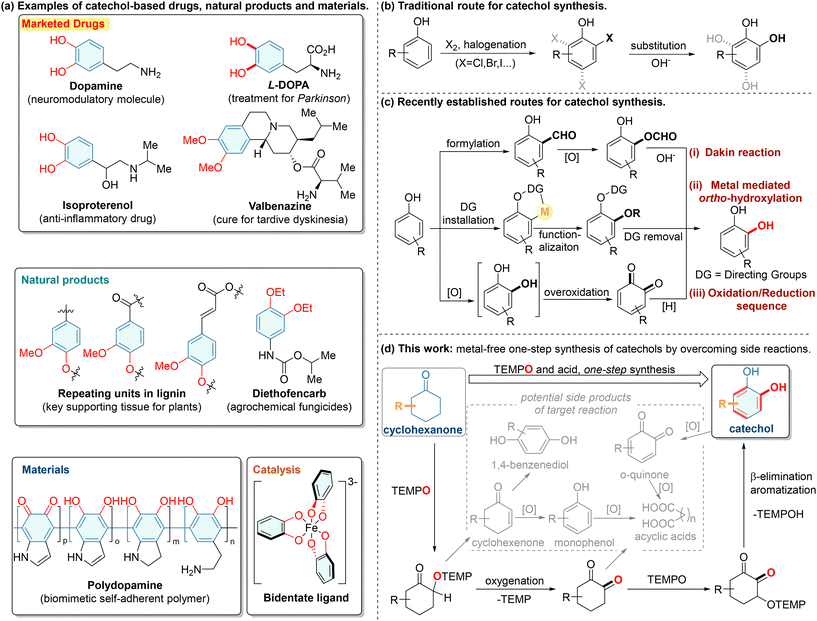
2,2,6,6-Tetramethylpiperidine-N-oxyl (TEMPO) is a persistent radical due to the delocalization of its unpaired electron across the nitrogen and oxygen atoms, as well as the steric protection provided by the bulky alkyl substituents on its nitrogen. Despite its inherent stability, TEMPO serves as a versatile oxidant, radical scavenger and catalyst for oxidation reactions, efficiently facilitating a wide range of chemical transformations in organic synthesis.1 Various TEMPO-enabled oxidation reactions involve the interaction of alkyl radicals with TEMPO to form alkoxyamine intermediates. Divergent transformations of these alkoxyamine intermediates can be precisely modulated through tailored reaction conditions, enabling the selective synthesis of diverse products with high efficiency based on established parameters.2 TEMPO-derived alkoxyamines generally undergo two primary reaction pathways. The first pathway involves the cleavage of N–O bonds in TEMPO-derived alkoxyamines, generating oxygenated products while releasing 2,2,6,6-tetramethylpiperidine (TEMPH) (Scheme 1a).2e–j Alternatively, alkoxyamines bearing β-C(sp3)–H bonds undergo β-elimination of TEMPOH, yielding alkenes as either final products or reactive intermediates (Scheme 1b).2k–o Herein, we reported TEMPO-mediated oxidation reactions that integrated these oxidation modes, providing a facile strategy to construct multi-functionalized catechols from readily available cyclohexanones (Scheme 1c).
Catechols, characterized by two hydroxyl groups lying on the ortho-positions of benzene rings, are extensively applied in contemporary organic synthesis, the pharmaceutical industry, and materials science, because catechol and its derivatives possess valuable pharmacological activities (Scheme 2a).3 About 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 metric tons of catechols are manufactured annually, and 78 drugs have been so far identified containing one or more catechol moieties (including 17 prescribed by the FDA),4 which collectively underscores the critical significance of catechol-based compounds. Despite the dramatic increase in the demand for catechols, the general synthetic methods for catechol synthesis remain in the infant stage, and thus, more effective methods are highly desirable.
000 metric tons of catechols are manufactured annually, and 78 drugs have been so far identified containing one or more catechol moieties (including 17 prescribed by the FDA),4 which collectively underscores the critical significance of catechol-based compounds. Despite the dramatic increase in the demand for catechols, the general synthetic methods for catechol synthesis remain in the infant stage, and thus, more effective methods are highly desirable.
Traditionally, catechols are industrially manufactured from phenols through a multi-step process consisting of an initial ortho-halogenation and a subsequent hydrolysis under harsh conditions (Scheme 2b).3a,b However, the low selectivity of this process often leads to the formation of unwanted side products, such as 1,4-benzenediol, 1,2,3-benzenetriol and 1,2,4-benzenetriol, which not only complicates the separation process but also wastes raw materials. In 1909, the Dakin oxidation was discovered and soon applied in catechol synthesis through a peroxide-initiated multi-step transformation of the salicylaldehyde framework [Scheme 2c(i)]. However, this was similarly restricted to relatively low total yields due to the lack of regioselectivity.5 Later the prevalence of transition-metal catalysis enabled rapid access to catechols through a directing-group-assisted ortho-C–H functionalization strategy that offered exclusive regioselectivity [Scheme 2c(ii)].6 Nevertheless, the indispensable need for expensive transition-metal catalysts, along with the need for the installation and removal of the directing groups, have limited the extensive application of this method in catechol synthesis. Although the ideal transformation, from phenols to catechols, has been achieved through enzyme catalysis with limited examples,7 similar chemical approaches invariably end up with over-oxidized quinones because of the lower oxidative potential of catechols compared to phenols, making additional reductive steps inevitable in such a detoured approach [Scheme 2c(iii)].8
Since two-step or longer synthetic procedures are generally required, the development of efficient and easy-to-handle methods for the syntheses of structurally diverse catechols from readily available substrates remains highly desired. Cyclohexanones are easily available and can be readily synthesized with a broad substrate scope.9 Prior elegant studies have demonstrated that cyclohexanones undergo oxidative dehydrogenation/aromatization under Pd-catalysis10 or photocatalysis11 to generate phenols. In a related study, Jiao's group reported an innovative and practical oxidative transformation of cyclohexanones to catechols,12 employing iodine as a catalyst and dimethyl sulfoxide as both the solvent and oxidant. However, a straightforward method that avoids the need to use corrosive reagents and offers improved functional group compatibility for the conversion of readily available cyclohexanones to catechols has yet to be achieved.
We envisioned that the reaction of cyclohexanones with TEMPO could provide one-step access to catechols, facilitating the development of a non-toxic, transition-metal-free approach favorable for pharmaceutical manufacturing.13 Considering that the conversion of cyclohexanones to catechols is expected to proceed via an α-C(sp3)–H oxygenation/β-elimination cascade with high regioselectivity, the development of such an approach must address the following challenges: controlling the competitive oxygenation and β-elimination to ensure reaction selectivity, as well as managing the inconsistent conditions and substrates required for these reactions. In this regard, the TEMPO-enabled C(sp3)–H oxygenation generally occurs at carbons(sp3) that are not tethered to another C(sp3)–H2h–j or through a reduction of TEMPO-derived alkoxyamine intermediates by metals.2f Besides, the TEMPO-enabled oxidation of C(sp3)–H on ethyl or longer alkyl groups mainly occurs via a β-elimination of TEMPOH to give alkenyl products.2a–d,k–o As described in Scheme 2d, the competition between oxygenation and β-elimination in the reaction of cyclohexanones with TEMPO may lead to varying products, such as 1,4-benzenediol, phenol, catechol, 1,2-cyclohexanedione14 or ring-opening side compounds,15 which underlines the challenges in developing a concise synthesis method for catechols from cyclohexanones using TEMPO.
Results and discussion
Discovery and optimization of the desired reaction
Our studies commenced with cyclohexanone and TEMPO to optimize the conditions for catechol synthesis (Table 1). Consistent with our speculation, the reaction between 4-phenylcyclohexanone with TEMPO occurred to produce the targeted catechol (1), but low yields were observed due to the formation of phenol as a side product (1′) (Table 1, entry 1). However, the elegant studies performed by Stahl and co-workers disclosed that Brønsted acids could regulate the redox property of TEMPO depending on their acid strength and improve the reactions of TEMPO.16 Additionally, Brønsted acids can also facilitate TEMPO-enabled oxygenation for the synthesis of carbonyl compounds by promoting N–O bond cleavage in the alkoxyamine intermediate.2j,17 Importantly, Brønsted acids generally promote the tautomerism of ketones to enols that are electron-rich and thus active toward oxidation transformation.18 Inspired by these elegant discoveries, we employed Brønsted acids as additives to achieve the efficient one-step syntheses of catechols. To our delight, we found that catalytic p-toluenesulfonic acid (TsOH) could improve the targeted reaction yield significantly (entry 2). Also, increasing the TsOH loading further raised the yield of catechol and decreased the yield of phenol (entries 2–5), as shown by the observation that one equivalent of TsOH enabled a 76% yield to be obtained (entry 5). Importantly, when NBu4OTs (0.5 equivalent) was used, the salt derived from the conjugate base of TsOH, combined with TsOH (1.0 equivalent), could serve as additives (entry 6) to obtain catechol in an optimal isolated yield of 82%, with the phenol completely suppressed.| Entry | Additives | Yieldb | |
|---|---|---|---|
| 1 (%) | 1′ (%) | ||
| a Standard conditions: 4-phenylcyclohexanone (0.2 mmol), TEMPO (3.8 equiv.), TsOH (1.0 equiv.), NBu4OTs (0.5 equiv.), 1,4-dioxane (0.4 mL), 120 °C, N2, 36 h. b Yields were determined by UPLC analysis using benzyl benzoate as an internal standard. c Isolated yields. | |||
| 1 | None | 26 | 33 |
| 2 | TsOH (0.1 equiv.) | 49 | 16 |
| 3 | TsOH (0.2 equiv.) | 56 | 12 |
| 4 | TsOH (0.5 equiv.) | 67 | 7 |
| 5 | TsOH (1.0 equiv.) | 76 | 5 |
| 6 | TsOH (1.0 equiv.) + NBu4OTs (0.5 equiv.) | 86 (82c) | Trace |
Substrate scope of reaction
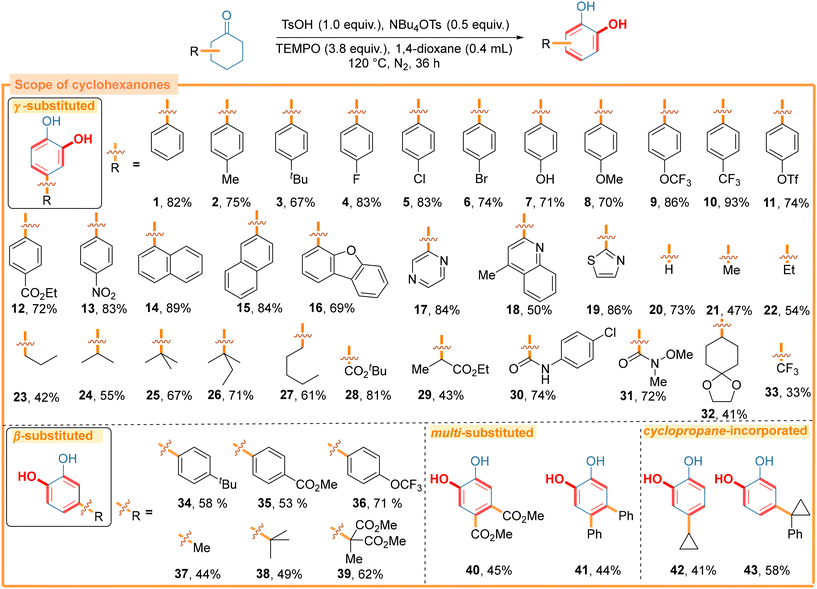
With the optimized conditions in hand, the versatility of the one-step synthesis of catechols was explored using various cyclohexanones (Scheme 3). 4-(Hetero)aryl-catechols generally exhibit high in vivo inhibition efficacy against inflammatory and HIV-1 reverse transcriptase-associated ribonuclease H.19 To our delight, cyclohexanones bearing various substituent groups at the γ-positions (1–15), as well as (hetero)aryl groups (16–19), produced the corresponding catechols in satisfactory yields, including the synthesis of the potential multitarget therapeutic drug 16.20 Due to the metal-free conditions, both halogens (F, Cl, Br; 4–6) and pseudo-halogen (OTf, 11) groups were well tolerated, which offered opportunities for further modifications. Cyclohexanones with a series of alkyl groups at the γ-positions also gave the corresponding products in moderate to good yields, regardless of their steric hindrance (21–27). Next, a range of polar functional groups, such as esters (28, 29), amide (30), Weinreb amide (31), ketal (32) and the pharmaceutically relevant trifluoromethyl (33) group, were all proven to be compatible with the proposed catechol synthesis.In comparison to γ-substituted ones, β-substituted cyclohexanones were more readily available by employing the well-established nucleophilic 1,4-conjugate addition between common cyclohexenones and nucleophilic reagents (Grignard reagents, organoboron reagents, etc.).9b,c Despite this advantage, converting the non-symmetrical β-substituted cyclohexanones into catechols may result in the production of two regioisomers due to the two different enolization orientations.
Pleasingly, our method enabled the regioselective transformation of β-substituted cyclohexanones to para-substituted catechols in good yields (34–39). Since γ-substituted and β-substituted cyclohexanones produced the same structural products, the use of more readily accessible β-substituted cyclohexanones further simplified the synthesis of para-substituted catechols. Meanwhile, 3,4-dually substituted cyclohexanones also underwent this transformation under standard conditions to form the tetra-substituted benzenoid products (40, 41), which opened up a new avenue for synthesizing structurally complex and functionally concentrated catechols. Interestingly, cyclohexanones substituted with cyclopropyl groups at either the γ- or β-position offered catechols bearing intact cyclopropyl groups without requiring the ring opening of cyclopropanes (42, 43).
Further exploration of the substrate scope demonstrated that both the positions and nature of several substituents impacted the chemo-selectivity of this transformation (Scheme 4). Cyclohexanone containing an α-methyl substituent produced a mixture of monophenol (44a) and catechol (44b) compounds with a nearly 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio. Moreover, cyclohexanone bearing an α-benzyl group gave a phenol (45a) in a poor yield without the formation of catechol (45b). The reaction outcome of these two cyclohexanones indicated that the α-substituents might reduce the reaction efficiency and inhibit the α-oxygenation of cyclohexanone. In agreement with this speculation, 2-phenylcyclohexanone produced a phenol (46a) in excellent yield and a catechol (46b) in poor yield. Both the N-tert-butyloxycarbonyl amino group (NH-Boc) (47) and N-methyl-N-tert-butyloxycarbonyl amino group (Me-N-Boc) (48) were found to be compatible with this reaction, but a mixture of phenol and catechol was observed in both cases. This was probably because the amino groups also acted as an active component21 to facilitate the dehydrogenative aromatization of cyclohexanones and impeded the α-oxygenation of cyclohexanones. Similarly, 3,5-diphenylcyclohexanone also showed good reactivity, producing a mixture of phenol (49a) and catechol (49b) in high yields, which could be attributed to the high reactivity of the phenyl moiety towards oxidation.
1 ratio. Moreover, cyclohexanone bearing an α-benzyl group gave a phenol (45a) in a poor yield without the formation of catechol (45b). The reaction outcome of these two cyclohexanones indicated that the α-substituents might reduce the reaction efficiency and inhibit the α-oxygenation of cyclohexanone. In agreement with this speculation, 2-phenylcyclohexanone produced a phenol (46a) in excellent yield and a catechol (46b) in poor yield. Both the N-tert-butyloxycarbonyl amino group (NH-Boc) (47) and N-methyl-N-tert-butyloxycarbonyl amino group (Me-N-Boc) (48) were found to be compatible with this reaction, but a mixture of phenol and catechol was observed in both cases. This was probably because the amino groups also acted as an active component21 to facilitate the dehydrogenative aromatization of cyclohexanones and impeded the α-oxygenation of cyclohexanones. Similarly, 3,5-diphenylcyclohexanone also showed good reactivity, producing a mixture of phenol (49a) and catechol (49b) in high yields, which could be attributed to the high reactivity of the phenyl moiety towards oxidation.
Synthetic applications
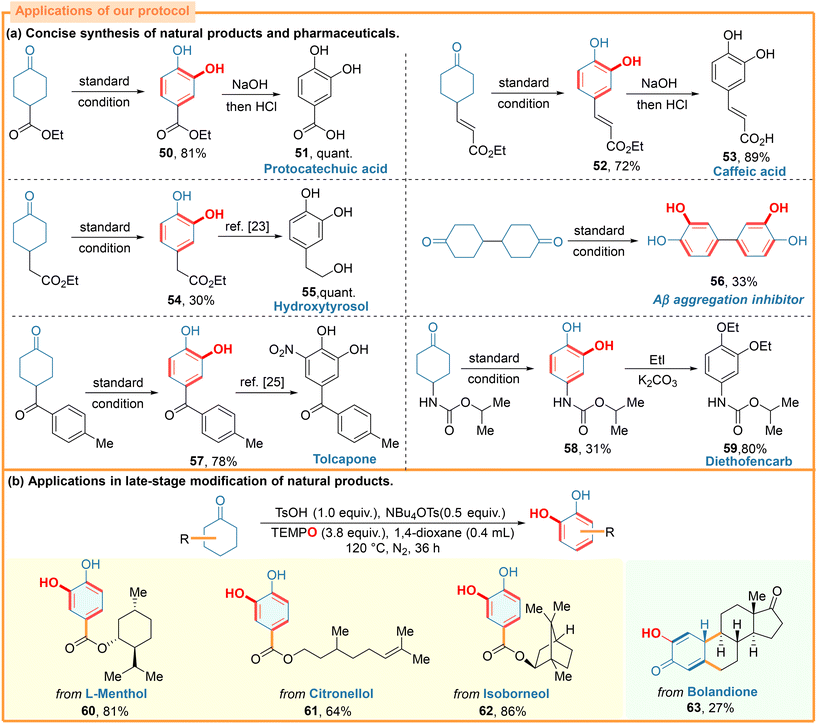
Notably, our one-step synthesis of catechols can enable the rapid syntheses of many natural products and pharmaceutical compounds starting from readily available compounds (Scheme 5). In this context, protocatechuic acid, a compound currently accessed by the alkaline fusion of vanillin, possesses a multitude of pharmacological activities including in vivo antiviral and nephroprotective activity.22 By using industrialized ethyl 4-oxocyclohexanecarboxylate as a starting material, our method offered a two-step approach to protocatechuic acid in a good total yield (51). Similarly, both γ-acrylate-substituted and γ-acetate-substituted cyclohexanones underwent acid-regulated reactions with TEMPO to produce the catechol derivatives (52, 54), which were then smoothly converted into the naturally occurring caffeic acid (53) and hydroxytyrosol (55)23via a one-step operation, respectively. Compound 56, featuring four phenolic hydroxyl groups, inhibits the aggregation of amyloid-β peptide in Alzheimer's, and acts as a superb bridging ligand for charge transfer.24 Our protocol also realized the use of commercially available 4,4′-bicyclohexanone for the one-step synthesis of the biological active compound 56. Compared with the known methods, which typically require multi-step synthetic routes involving palladium, our method not only offers a simplified synthesis of 56, but also circumvents the need for the employment of cytotoxic transition metals. Intriguingly, for substrates that contain two different carbonyls, TEMPO can distinguish them and selectively transform the cyclohexanone part into the corresponding compound 57, the direct precursor to the Alzheimer's inhibitor Tolcapone.25 Furthermore, the agrochemical diethofencarb (59), an effective marketed fungicide against benzimidazole-resistant fungi, could be quickly constructed through our method and a subsequent ethylation transformation.26Considering the pharmacological activity exhibited by some compounds bearing a catechol component,19,20 we also endeavored to apply our approach in the late-stage functionalization of structurally complex bioactive molecules, which could offer a streamlined synthesis of catechol-based pharmaceutically relevant molecules. In this domain, cyclohexanones embedded in natural products, such as L-menthol (60), citronellol (61) and isoborneol (62), could be transformed into the corresponding catechols with high efficiency. Notably, the more challenging steroid substrate, namely bolandione, which bears two carbonyl groups on its cyclic skeleton, underwent α-oxygenation and single β-H elimination on its six-membered ring to deliver product 63, in a marginal yield.
Mechanism studies
To elucidate the mechanism of this reaction, an array of experimental studies was carried out (Scheme 6). 4-Phenyl-cyclohexenone (64) was transformed under standard conditions to produce 4-phenylphenol (1′) as the main product, along with a small amount of 4-phenyl-catechol (1) (Scheme 6a). In contrast, the reaction of 4-phenyl-cyclohexane-1,2-dione (65) only formed 4-phenyl-catechol (1) without the formation of phenol 1′ (Scheme 6b). No conversion of phenol to catechol was observed when 1′ was used as the starting material (Scheme 6c). These reactions suggested that the oxidative transformation might proceed through an initial α-oxygenation of cyclohexanones to form 1,2-cyclohexanedione as an intermediate, followed by a dehydrogenative aromatization of 1,2-cyclohexanedione to yield the catechol. The possibility of an initial β-H elimination of cyclohexanone to form cyclohexenone (64) as an intermediate was excluded. Notably, although Brønsted acids can promote the disproportionation of TEMPO to its corresponding oxoammonium salt (TEMPO+) and hydroxylamine (TEMPOH),27 the reaction of 2,2,6,6-tetramethyl-1-oxo-piperidinium tetrafluoroborate (TEMPO+BF4−) with 4-phenylcyclohexanone did not generate the catechol product (Scheme 6d), indicating that the reaction was likely initiated by the interaction of cyclohexanone with TEMPO rather than the oxoammonium salt. The 18O-labeled experiment revealed that one of the oxygen atoms in catechol originated from TEMPO, as high-resolution mass spectrometry analysis indicated that the 18O ratio in the product catechol (18O-1) was nearly identical to that of the starting 18O-TEMPO. These mechanistic studies demonstrate that this acid-regulated oxidation of cyclohexanone represents a novel oxidation mode of TEMPO, as reactions between TEMPO and alkyl ketones via the isomerization of alkyl ketones into enols generally require external oxidants to oxidize the enols to carbon-centered radicals.28Grounded on the experimental results and previous studies,2a,29 a plausible mechanism for this catechol-synthesis reaction was proposed and shown in Scheme 6f. Initially, TsOH, a Brønsted acid, promoted the isomerization of cyclohexanone into the enol tautomer (I). TEMPO then abstracted a hydrogen atom from the hydroxyl group of enol (I) to form radical (II) at the α-carbon of cyclohexanone. This α-carbon-centered radical was trapped by another TEMPO molecule to give intermediate (III), which was further isomerized to the enol bearing a TEMPO substituent at the α-position (IV) due to the relatively strong acidity of the C(sp3)–H bond at the α-carbon bounded to TEMPO. The α-TEMPO enol (IV) underwent cleavage of the O–N bond of TEMPO through transition state V to form 1,2-cyclohexanedione (VI) along with the elimination of TEMPH. 1,2-Cyclohexanedione (VI) also underwent acid-promoted isomerization to give the α-carbonyl-enol intermediate (VII), which was then oxidized by TEMPO to form the 1,2-cyclohexanedione radical (VIII), followed by combination with another TEMPO molecule to form intermediate IX. Due to the intramolecular hydrogen bonding, the hydrogen abstraction from the hydroxyl group to eliminate TEMPH was geometrically unfavored. Consequently, intermediate IX underwent a β-elimination of TEMPOH to give the α-carbonyl enone intermediate (X). Finally, α-carbonyl enone (X) underwent an aromatization-driven isomerization via TsOH-mediated hydrogen migration to give catechol (XI) as the final product.
Conclusions
In conclusion, we have developed an acid-regulated method for the syntheses of catechols through the oxidation of cyclohexanones by TEMPO. This transformation consisted of two different TEMPO-enabled oxidation modes (oxygenation and β-elimination), thus representing a novel transformation, as these two modes generally required distinct reaction conditions and were only feasible with varied substrates. This acid-regulated protocol exhibited excellent functional group tolerance and was applicable for the concise synthesis and modification of bioactive compounds. Considering that catechols are key structural components in many varieties of bioactive compounds, we hope this general method for the efficient synthesis of catechols will find wide applications in organic synthesis.Author contributions
W. S. conceived and directed the project. B. X., X. J. and W. S. wrote the manuscript. B. X. developed and performed the methodology experiments and analyzed the data. X. L., L. D. and Y. S. prepared the starting materials.Data availability
The authors declare that the data supporting the findings of this study are available within the manuscript and the ESI,† as well as from the authors upon reasonable request.Conflicts of interest
There are no conflicts to declare.Acknowledgements
This work was supported by the National Key Research and Development Program of China (2018YFA0704502), the National Natural Science Foundation of China (Grant No. 21931011 and 22071243) and the Youth Innovation Promotion of the Chinese Academy of Sciences (2022304).References
- For reviews of TEMPO, see:
(a) L. Tebben and A. Studer, Angew. Chem., Int. Ed., 2011, 50, 5034–5068 CAS
; (b) D. Leifert and A. Studer, Chem. Rev., 2023, 123, 10302–10380 CAS
; (c) O. G. Mancheño and T. Stopka, Synthesis, 2013, 45, 1602–1611 Search PubMed
; (d) B. L. Ryland and S. S. Stahl, Angew. Chem., Int. Ed., 2014, 53, 8824–8838 CrossRef CAS PubMed
.
-
(a) B. Xu and W. Su, Angew. Chem., Int. Ed., 2022, 61, e202203365 CrossRef CAS
; (b) R. Hu, Y. Tao, X. Zhang and W. Su, Angew. Chem., Int. Ed., 2021, 60, 8425–8430 Search PubMed
; (c) P. Qian, Y. Deng, H. Mei, J. Han and Y. Pan, Chem. Commun., 2017, 53, 2958–2961 RSC
; (d) N. A. Eddy, C. B. Kelly, M. A. Mercadante, N. E. Leadbeater and G. Fenteany, Org. Lett., 2012, 14, 498–501 Search PubMed
. For TEMPO mediated oxygenative reactions, see: (e) A. Sanz-Marco, S. Martinez-Erro and B. Martin-Matute, Chem. – Eur. J., 2018, 24, 11564–11567 CrossRef CAS PubMed
; (f) A. de la Torre, D. Kaiser and N. Maulide, J. Am. Chem. Soc., 2017, 139, 6578–6581 CrossRef CAS PubMed
; (g) S. Nagasawa, Y. Sasano and Y. Iwabuchi, Chem. – Eur. J., 2017, 23, 10276–10279 CrossRef CAS
; (h) M. Hu, R. J. Song and J. H. Li, Angew. Chem., Int. Ed., 2015, 54, 608–612 CrossRef CAS
; (i) J. Yan, T. Ni, F. Yan, J. Zhang and F. Zhuang, RSC Adv., 2015, 5, 89906–89910 RSC
; (j) P. Feng, S. Song, L. H. Zhang and N. Jiao, Synlett, 2014, 2717–2720 CAS
. For TEMPO mediated dehydrogenative reactions, see: (k) F. Politano, W. P. Brydon, J. Nandi and N. E. Leadbeater, Molbank, 2021, M1180 CrossRef CAS
; (l) Y. Shang, X. Jie, K. Jonnada, S. N. Zafar and W. Su, Nat. Commun., 2017, 8, 2273 CrossRef
; (m) S. Nagasawa, Y. Sasano and Y. Iwabuchi, Angew. Chem., Int. Ed., 2016, 55, 13189–13194 CrossRef CAS PubMed
; (n) X. Jie, Y. Shang, X. Zhang and W. Su, J. Am. Chem. Soc., 2016, 138, 5623–5633 Search PubMed
; (o) T. A. Hamlin, C. B. Kelly and N. E. Leadbeater, Eur. J. Org. Chem., 2013, 3658–3661 Search PubMed
.
-
(a)
H. Fiege, H.-W. Voges, T. Hamamoto, S. Umemura, T. Iwata, H. Miki, Y. Fujita, H.-J. Buysch, D. Garbe and W. Paulus, Phenol Derivatives in Ullmann's Encyclopedia of Industrial Chemistry, Wiley-VCH, Weinheim, 2002 Search PubMed
; (b) B. A. Barner, Catechol in Encyclopedia of Reagents for Organic Synthesis, John Wiley and Sons, New York, 2004 Search PubMed
; (c) A. K. L. Yuen, G. A. Hutton, A. F. Masters and T. Maschmeyer, Dalton Trans., 2012, 41, 2545–2559 Search PubMed
; (d) J. H. Ryu, P. B. Messersmith and H. Lee, ACS Appl. Mater. Interfaces, 2018, 10, 7523–7540 Search PubMed
; (e) J. Liebscher, Eur. J. Org. Chem., 2019, 4976–4994 Search PubMed
.
- D.-P. Yang, H.-F. Ji, G.-Y. Tang, W. Ren and H.-Y. Zhang, Molecules, 2007, 12, 878–884 Search PubMed
.
-
(a) P. Bora, B. Bora and U. Bora, New J. Chem., 2021, 45, 17077–17084 Search PubMed
; (b) H. D. Dakin, Org. Synth., 1923, 3, 28 CrossRef
; (c) H. D. Dakin, Am. Chem. J., 1909, 42, 477–498 Search PubMed
.
-
(a) P. Andrade-Sampedro, J. M. Matxain and A. Correa, Adv. Synth. Catal., 2022, 364, 2072–2079 CrossRef CAS
; (b) Q. Wu, D. Yan, Y. Chen, T. Wang, F. Xiong, W. Wei, Y. Lu, W.-Y. Sun, J. J. Li and J. Zhao, Nat. Commun., 2017, 8, 14227 CrossRef
; (c) X. Yang, Y. Sun, Z. Chen and Y. Rao, Adv. Synth. Catal., 2014, 356, 1625 CAS
; (d) W. Liu and L. Ackermann, Org. Lett., 2013, 15, 3484–3486 CAS
; (e) C. Huang, N. Ghavtadze, B. Chattopadhyay and V. Gevorgyan, J. Am. Chem. Soc., 2011, 133, 17630–17633 CAS
.
-
(a) A.-W. Struck, M. R. Bennett, S. A. Shepherd, B. J. C. Law, Y. Zhuo, L. S. Wong and J. Micklefield, J. Am. Chem. Soc., 2016, 138, 3038–3045 Search PubMed
; (b) C. Morioka, Y. Tachi, S. Suzuki and S. Itoh, J. Am. Chem. Soc., 2006, 128, 6788–6789 CAS
.
-
(a) X. Xiao, N. S. Greenwood and S. E. Wengryniuk, Angew. Chem., Int. Ed., 2019, 58, 16181–16187 Search PubMed
; (b) A. Pezzella, L. Lista, A. Napolitano and M. d'Ischia, Tetrahedron Lett., 2005, 46, 3541–3544 CAS
; (c) D. Magdziak, A. A. Rodriguez, R. W. Van De Water and T. R. R. Pettus, Org. Lett., 2002, 4, 285–288 CAS
.
-
(a) A. V. Bay, E. J. Farnam and K. A. Scheidt, J. Am. Chem. Soc., 2022, 144, 7030–7037 CAS
; (b) S. Cren, P. Schär, P. Renaud and K. Schenk, J. Org. Chem., 2009, 74, 2942–2946 CAS
; (c) X. Lu and S. Lin, J. Org. Chem., 2005, 70, 9651–9653 Search PubMed
.
-
(a) Y. Izawa, D. Pun and S. S. Stahl, Science, 2011, 333, 209–213 CAS
; (b) D. Pun, T. Diao and S. S. Stahl, J. Am. Chem. Soc., 2013, 135, 8213–8221 Search PubMed
.
-
(a) H. P. Caldora, Z. Zhang, M. J. Tilby, O. Turner and D. Leonori, Angew. Chem., Int. Ed., 2023, 62, e202301656 CAS
; (b) L. Min, J. Lin and W. Shu, Chin. J. Chem., 2023, 41, 2773–2778 CrossRef CAS
.
- Y.-F. Liang, X. Li, X. Wang, M. Zou, C. Tang, Y. Liang, S. Song and N. Jiao, J. Am. Chem. Soc., 2016, 138, 12271–12277 CrossRef CAS
.
- C. J. Welch, J. Albaneze-Walker, W. R. Leonard, M. Biba, J. DaSilva, D. Henderson, B. Laing, D. J. Mathre, S. Spencer, X. Bu and T. Wang, Org. Process Res. Dev., 2005, 9, 198–205 CrossRef CAS
.
- M. Hayashi, M. Shibuya and Y. Iwabuchi, Synlett, 2012, 23, 1025–1030 CrossRef CAS
.
- R. Jastrzebski, B. M. Weckhuysen and P. C. Bruijnincx, Chem. Commun., 2013, 49, 6912–6914 Search PubMed
.
- J. B. Gerken, Y. Q. Pang, M. B. Lauber and S. S. Stahl, J. Org. Chem., 2017, 83, 7323–7330 Search PubMed
.
-
(a) R. Hu, F.-J. Chen, X. Zhang, M. Zhang and W. Su, Nat. Commun., 2019, 10, 3681 CAS
; (b) D. H. Hunter, D. H. R. Barton and W. J. Motherwell, Tetrahedron Lett., 1984, 25, 603–606 CrossRef CAS
.
-
F. A. Carey and R. J. Sundberg, Advanced Organic Chemistry Part A: Structure and Mechanisms, 5th edn, Springer, New York, 2007, p. 602 Search PubMed
.
-
(a) G. Tocco, F. Esposito, P. Caboni, A. Laus, J. A. Beutler, J. A. Wilson, A. Corona, S. F. J. Le Grice and E. Tramontano, J. Enzyme Inhib. Med. Chem., 2020, 35, 1953–1963 CAS
; (b) A. Corona, T. Masaoka, G. Tocco, E. Tramontano and S. F. J. Le Grice, Future Med. Chem., 2013, 5, 2127–2139 CAS
.
- F. Bruno, V. Krauth, S. M. Nabavi, V. Temml, F. Fratianni, G. Spaziano, F. Nazzaro, F. Roviezzo, J. Xiao, H. Khan, M. P. Romano, B. D'Agostino, O. Werz and R. Filosa, Eur. J. Med. Chem., 2022, 243, 114788 CAS
.
- H. Richter, R. Frohlich, C. G. Daniliuc and O. G. Mancheño, Angew. Chem., Int. Ed., 2012, 51, 8656–8660 CAS
.
- J. Song, Y. He, C. Luo, B. Feng, F. Ran, H. Xu, Z. Ci, R. Xu, L. Han and D. Zhang, Pharmacol. Res., 2020, 161, 105109 CrossRef CAS PubMed
.
- A. Procopio, S. Alcaro, M. Nardi, M. Oliverio, F. Ortuso, P. Sacchetta, D. Pieragostino and G. Sindona, J. Agric. Food Chem., 2009, 57, 11161–11167 CrossRef CAS PubMed
.
-
(a) C. B. Stevens, J. M. Hanna and R. K. Lammi, Bioorg. Med. Chem. Lett., 2013, 23, 1703–1706 CAS
; (b) J. Best, I. V. Sazanovich, H. Adams, R. D. Bennett, E. S. Davies, A. J. H. M. Meijer, M. Towrie, S. A. Tikhomirov, O. V. Bouganov, M. D. Ward and J. A. Weinstein, Inorg. Chem., 2010, 49, 10041–10056 CrossRef CAS PubMed
.
-
(a) P. Natarajan, R. Chaudhary, N. Rani, Sakshi and P. Venugopalan, Tetrahedron Lett., 2020, 61, 151529 CrossRef CAS
; (b) A. H. Rajput, W. Martin, M. H. Saint-Hilaire, E. Dorflinger and S. Pedder, Neurology, 1997, 49, 1066–1071 CrossRef CAS PubMed
.
- M. Fujimura, T. Kamakura and I. Yamaguchi, J. Pestic. Sci., 1992, 17, 237–242 CrossRef
.
- V. A. Golubev, V. D. Sen and G. Rozantsev, Bull. Acad. Sci. USSR, Div. Chem. Sci., 1979, 28, 1927–1931 CrossRef
.
-
(a) J. F. Van Humbeck, S. P. Simonovich, R. R. Knowles and D. W. C. MacMillan, J. Am. Chem. Soc., 2010, 132, 10012–10014 CrossRef CAS PubMed
; (b) M. P. Sibi and M. Hasegawa, J. Am. Chem. Soc., 2007, 129, 4124–4125 CAS
; (c) U. Jahn, P. Hartmann, I. Dix and P. G. Jones, Eur. J. Org. Chem., 2001, 3333–3355 CAS
.
-
(a) B. Xu, X. Liu, L. Deng, Y. Shang, X. Jie and W. Su, Sci. Adv., 2024, 10, eadn7656 CAS
; (b) X. Jie, Y. Shang, Z.-N. Chen, X. Zhang, W. Zhuang and W. Su, Nat. Commun., 2018, 9, 5002 Search PubMed
.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: https://doi.org/10.1039/d5gc00115c |
| This journal is © The Royal Society of Chemistry 2025 |