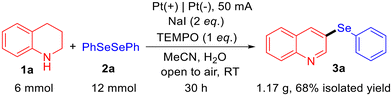Tunable selective electrochemical selenization of tetrahydroquinolines with diselenides†
Lan-Xi
Zong‡
,
Yu-Fang
Tan‡
 ,
Yu-Hao
Yang
,
Yan-Hong
He
,
Yu-Hao
Yang
,
Yan-Hong
He
 * and
Zhi
Guan
* and
Zhi
Guan
 *
*
Key Laboratory of Applied Chemistry of Chongqing Municipality, School of Chemistry and Chemical Engineering, Southwest University, Chongqing 400715, China. heyh@swu.edu.cn, guanzhi@swu.edu.cn
First published on 7th February 2025
Abstract
A simple and environmentally friendly electrochemical synthesis method is presented for the selenylation of tetrahydroquinolines with diselenides. By adjusting the reaction conditions, different products can be synthesized. Using NaI as the electrolyte and 2,2,6,6-tetramethylpiperidinooxy (TEMPO) as a redox mediator, C-3 selenylated quinolines were successfully synthesized, achieving the challenging C-3 substitution of quinolines. In the absence of TEMPO, C-6 selenylated tetrahydroquinolines were obtained. This green method offers excellent selectivity and a broad substrate scope, with reaction conditions that can be easily tuned to achieve different outcomes.
Green foundation1. For the first time, 3-(phenylselanyl)quinoline was directly produced from tetrahydroquinolines via electrochemical selenization/oxidation. By simply adjusting the reaction conditions, the selectivity can be tuned to produce 6-(phenylselanyl)-tetrahydroquinoline.2. Using the electrochemical green method, hydrogen is the only by-product. This green method offers excellent selectivity and a broad substrate scope, tunable selectivity for different products, oxidant-free and metal-free processes and insensitivity to air and moisture. 3. Continuous-flow electrochemistry can be used to improve the reaction efficiency. With this technique, the electrolysis can be performed at low concentrations (or absence) of the electrolyte or mediator, and the yield of the gram-scale experiment can also be improved. |
Introduction
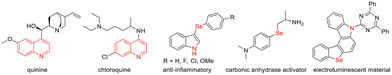
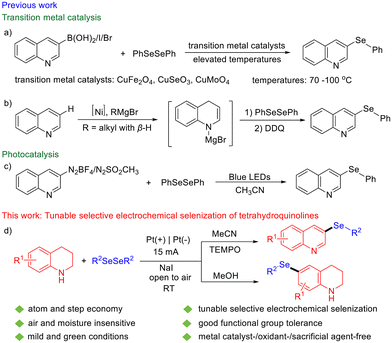
The quinoline skeleton is prevalent in numerous natural products and pharmaceuticals, recognized for its broad spectrum of biological activities, including antimalarial,1 antibacterial,2 antitumor,3 and antituberculosis4 effects. For example, quinine and chloroquine5 are the most famous antimalarial drugs containing a quinoline scaffold (Fig. 1). Organoselenium compounds also exhibit unique chemical, physical and drug-related biological properties.6,7 Among the various organoselenium compounds, it is worth noting that molecules containing both nitrogen and selenium are widely prevalent (Fig. 1). Their applications in medicine and materials science make them particularly appealing for chemical synthesis and biology research.8,9 Given the significant properties of selenium compounds, incorporating selenium into the privileged quinoline framework holds substantial promise for advancing drug discovery and materials chemistry.Due to the influence of the sp2 hybridized nitrogen atom, electrophilic substitution reactions of quinoline primarily occur at the benzene ring. In contrast, nucleophilic substitution reactions take place on the pyridine ring, predominantly at the C-2 and C-4 positions. Moreover, various C-2 functionalization of quinolines has been accomplished through the addition of nucleophilic radicals to N-heteroarenes, such as in the Minisci reaction. Consequently, substitution at the C-3 position of quinoline remains a challenging task. While some progress has been made in the research of quinoline C-3 substitution,10,11 reports on seleno substitution at this position are still limited, with only a few sporadic examples. For instance, the synthesis of 3-(phenylselanyl)quinoline was accomplished through the cross-coupling reaction of quinolin-3-ylboronic acid, 3-iodoquinoline, or 3-bromoquinoline with 1,2-diphenyldiselane. These reactions employed catalysts such as CuFe2O4,12 CuSeO3,13 or CuMoO4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 14 and were carried out at elevated temperatures ranging from 70 to 100 °C (Fig. 2a). A nickel-catalyzed method for C-3 selective selenization of unactivated quinoline with 1,2-diphenyldiselane was developed, utilizing Ni(dppp)Cl2, a Grignard reagent, and DDQ under an argon atmosphere. In this process, quinoline first underwent a 1,4-addition with a nickel hydride species generated through β-H elimination of alkyl nickel intermediate, forming 1,4-dihydroquinoline. This intermediate subsequently reacted with 1,2-diphenyldiselane via nucleophilic attack, followed by oxidative aromatization, to produce 3-(phenylselanyl)quinoline (Fig. 2b).15 Alternatively, 3-(phenylselanyl)quinoline could be synthesized through visible light-mediated reactions. These methods involved either the reaction of a quinoline-derived diazonium salt16 or a quinoline-derived azo sulfone17 with 1,2-diphenyldiselane (Fig. 2c). Furthermore, a separate approach using phenylseleninic anhydride and tetrahydroquinoline was reported, yielding 3-(phenylselanyl)quinoline (14%) and 6-(phenylselanyl)-tetrahydroquinoline (36%).18 These elegant works have made significant contributions to quinoline C-3 seleno substitution. However, there are still some limitations, such as the need for pre-functionalization, the use of transition metals, high temperatures, moisture-sensitive reagents or oxidants.
14 and were carried out at elevated temperatures ranging from 70 to 100 °C (Fig. 2a). A nickel-catalyzed method for C-3 selective selenization of unactivated quinoline with 1,2-diphenyldiselane was developed, utilizing Ni(dppp)Cl2, a Grignard reagent, and DDQ under an argon atmosphere. In this process, quinoline first underwent a 1,4-addition with a nickel hydride species generated through β-H elimination of alkyl nickel intermediate, forming 1,4-dihydroquinoline. This intermediate subsequently reacted with 1,2-diphenyldiselane via nucleophilic attack, followed by oxidative aromatization, to produce 3-(phenylselanyl)quinoline (Fig. 2b).15 Alternatively, 3-(phenylselanyl)quinoline could be synthesized through visible light-mediated reactions. These methods involved either the reaction of a quinoline-derived diazonium salt16 or a quinoline-derived azo sulfone17 with 1,2-diphenyldiselane (Fig. 2c). Furthermore, a separate approach using phenylseleninic anhydride and tetrahydroquinoline was reported, yielding 3-(phenylselanyl)quinoline (14%) and 6-(phenylselanyl)-tetrahydroquinoline (36%).18 These elegant works have made significant contributions to quinoline C-3 seleno substitution. However, there are still some limitations, such as the need for pre-functionalization, the use of transition metals, high temperatures, moisture-sensitive reagents or oxidants.
Electrochemical synthesis, which uses electricity as a cost-effective, renewable, and eco-friendly alternative to stoichiometric oxidants or reducing agents, has emerged as a vital method in organic synthesis.19 Recently, significant advancements have been achieved in the electrochemical synthesis of chalcogen-containing compounds.20,21 In this context, we developed a novel method for synthesizing C-3 selenylated quinolines. For the first time, these compounds were directly produced from tetrahydroquinolines via electrochemical selenization/oxidation. Tetrahydroquinolines exhibit structural versatility and high reactivity, allowing interactions with various functional groups, making them widely applicable in synthesis. Additionally, by simply adjusting the reaction conditions, the selectivity can be tuned to produce C-6 selenylated tetrahydroquinolines. Herein, we report this straightforward and tunable electrochemical selenization of tetrahydroquinolines with diselenides, achieving the selective synthesis of C-3 selenylated quinolines and C-6 selenylated tetrahydroquinolines. The reaction features mild reaction conditions, high atom efficiency, excellent selectivity, good functional group tolerance, and a broad substrate scope.
Results and discussion
Initially, tetrahydroquinoline (1a) and 1,2-diphenyldiselane (2a) were selected as model substrates for the reaction, which was performed in an undivided electrolytic cell. A systematic screening of various parameters led to the identification of optimal reaction conditions: 1a (0.3 mmol, 1 equiv.), 2a (3 equiv.), MeCN (6 mL) and H2O (0.2 mL) as the solvent, NaI (2 equiv.) as the electrolyte, 2,2,6,6-tetramethylpiperidinooxy (TEMPO, 1 equiv.) as the mediator, and a constant current of 15 mA applied at room temperature (RT), using platinum electrodes for both the anode and cathode (see ESI, 3.1 Tables S1–S9†). To further validate the reaction, several control experiments were conducted (Table 1). Under standard conditions, the desired product 3a was obtained in 85% isolated yield (Table 1, entry 1). In contrast, no reaction occurred without the application of electricity (Table 1, entry 2). When no water was added to the reaction system, the voltage rose rapidly after applying electricity and exceeded 30 V in just 4 minutes (Table 1, entry 3). Excessively high voltage is unfavourable for the reaction, as it may lead to over-electrolysis of the substrates and products. After adding 0.10–0.25 mL of water, the system voltage remained between 2.4 and 3.7 V throughout the electrolysis, allowing the reaction to proceed smoothly. When the amount of water added increased from 0.1 mL to 2.0 mL, the yield of product 3a improved from 61% to 85%. However, further increasing the water amount to 2.5 mL reduced the yield to 70% (as shown in the ESI, Table S9†). Therefore, we hypothesize that the addition of water enhances the conductivity of the reaction system, and an appropriate amount of water improves the product yield. In the absence of TEMPO, 3a formation was barely detectable. However, 6-(phenylselanyl)-tetrahydroquinoline (4a) (structure shown in Fig. 4) was obtained in 40% yield, accompanied by the formation of various complex byproducts (Table 1, entry 4). When NaI was omitted as the electrolyte, the reaction voltage overloaded (Table 1, entry 5). Replacing NaI with NH4I resulted in a 49% yield (Table 1, entry 6). Switching NaI to NaClO4 led to almost no formation of 3a, with many side reactions observed (Table 1, entry 7). Using 9-azabicyclo[3.3.1]nonane N-oxyl instead of TEMPO yielded a 45% yield (Table 1, entry 8). When DMF was used as the solvent instead of MeCN, the yield dropped to 33% (Table 1, entry 9). Adjusting the current either higher or lower also reduced the yield (Table 1, entries 10 & 11). Furthermore, using electrodes other than platinum for both the anode and cathode resulted in a lower yield of 3a (Table 1, entries 12–14).| Entry | Variation from standard conditions | Yieldb (%) |
|---|---|---|
| a Standard conditions: a mixture of 1a (0.3 mmol, 1 equiv.), 2a (0.9 mmol, 3 equiv.), NaI (0.6 mmol, 2 equiv.), TEMPO (0.3 mmol, 1 equiv.) and H2O (0.2 mL) in MeCN (6 mL) under a constant current of 15 mA (Pt anode: 1 cm × 1 cm × 0.2 mm; Pt cathode: 1 cm × 1 cm × 0.2 mm) in an undivided cell at RT for 6 h. b Isolated yield. c No reaction. d 4a was obtained in 40% yield. | ||
| 1 | — | 85 |
| 2 | Without current | N.R.c |
| 3 | Without H2O | High voltage |
| 4 | Without TEMPO | Traced |
| 5 | Without NaI | Voltage overload |
| 6 | NH4I instead of NaI | 49 |
| 7 | NaClO4 instead of NaI | Trace |
| 8 | 9-Azabicyclo[3.3.1]nonane N-oxyl instead of TEMPO | 45 |
| 9 | DMF instead of MeCN | 33 |
| 10 | 12 mA instead of 15 mA | 46 |
| 11 | 18 mA instead of 15 mA | 51 |
| 12 | C(+) | Pt(−) instead of Pt(+) | Pt(−) | 64 |
| 13 | C(+) | C(−) instead of Pt(+) | Pt(−) | 67 |
| 14 | Pt(+) | C(−) instead of Pt(+) | Pt(−) | 46 |
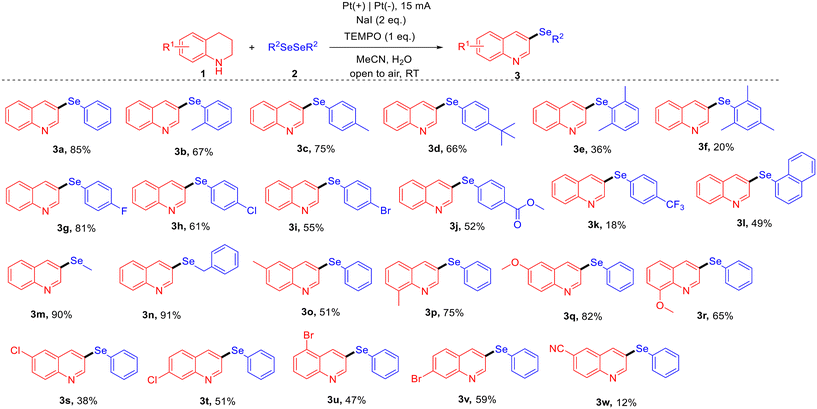
Under the optimized reaction conditions, the substrate scope for the synthesis of C-3 selenylated quinolines was explored (Fig. 3). The investigation began with tetrahydroquinoline (1a) as the coupling partner to evaluate various diselenide substrates. When the phenyl ring of 1,2-diphenyldiselane was substituted with methyl or tert-butyl groups, the products were obtained in good yields of 66–75% (3b–3d). Dimethyl or trimethyl substitutions on the phenyl ring led to low yields (3e, 3f), likely due to steric hindrance. Halogen-substituted (F, Cl, Br) 1,2-diphenyldiselane provided satisfactory yields of 55–81% (3g–3i). Similarly, ester-substituted diselenide delivered the desired product in a moderate yield of 52% (3j). However, when 1,2-bis (4-(trifluoromethyl)phenyl)diselane was used, the corresponding product was obtained in only 18% yield (3k), accompanied by the formation of a substantial amount of quinoline as a byproduct. Furthermore, 1,2-di(naphthalen-1-yl)diselane proved compatible with the reaction, providing the corresponding product in 49% yield (3l). In addition to aromatic diselenides, aliphatic diselenides such as dimethyl diselenide and benzyl diselenide also performed exceptionally well, giving the desired products in excellent yields of 90–91% (3m, 3n). The substrate scope of tetrahydroquinolines was then explored using 1,2-diphenyldiselane (2a) as the coupling partner. Tetrahydroquinolines substituted with methyl or methoxy groups at the C-6 or C-8 positions provided the corresponding products in moderate to good yields, ranging from 51% to 82% (3o–3r). Cl or Br-substituted tetrahydroquinolines achieved yields of 38–59% (3s–3v). Interestingly, 6-Cl-tetrahydroquinoline gave a lower yield compared to 7-Cl-tetrahydroquinoline, while 5-Br-tetrahydroquinoline produced a lower yield than 7-Br-tetrahydroquinoline, indicating that the position of the substituent affects the reactivity of tetrahydroquinolines. However, the introduction of a cyano group at the C-6 position of tetrahydroquinoline significantly diminished reactivity, resulting in a notably low yield of only 12% (3w), likely due to the strong electron-withdrawing nature of the cyano group.
To verify the practicality of the reaction, a gram-scale experiment was conducted (Scheme 1). Using 1a (6 mmol) and 2a (12 mmol) as substrates, the reaction yielded product 3a in 68% isolated yield (1.17 g). This result confirms the feasibility of performing the reaction on a larger scale (see ESI, 2.3†).
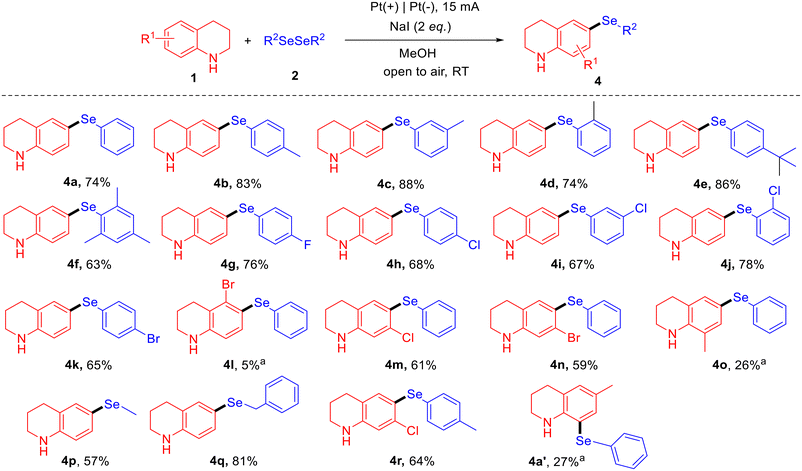
During the optimization of reaction conditions for synthesizing 3-(phenylselanyl)quinoline (3a) from the model substrates tetrahydroquinoline (1a) and 1,2-diphenyldiselane (2a), we observed that omitting TEMPO led to the formation of an alternative product, 6-(phenylselanyl)-tetrahydroquinoline (4a). Therefore, we further optimized the reaction conditions specifically for the synthesis of 4a. After systematically screening various parameters, the optimal reaction conditions were identified: 1a (0.3 mmol, 1 equiv.), 2a (3 equiv.), MeOH (6 mL) as the solvent, NaI (2 equiv.) as the electrolyte, with a constant current of 15 mA at room temperature, using platinum electrodes for both the anode and cathode (see ESI, 3.2 Tables S10–S15†). Under these optimized conditions, 4a was obtained in 74% isolated yield.
The substrate scope for the synthesis of C-6 selenylated tetrahydroquinolines was explored under the optimized reaction conditions (Fig. 4). First, the reactions between tetrahydroquinoline and various substituted 1,2-diphenyldiselenides were investigated. When the phenyl rings of 1,2-diphenyldiselenide were substituted with methyl groups at the ortho, meta, or para positions, the corresponding products were obtained in good yields of 74–88% (4b–4d). The reaction with para-tert-butyl-substituted 1,2-diphenyldiselenide afforded the product in a high yield of 86% (4e). However, the reaction with trimethyl-substituted 1,2-diphenyldiselenide gave a lower yield of 63% (4f), likely due to steric hindrance. Reactions with 1,2-diphenyldiselenides bearing various halogens (F, Cl, Br) at different positions afforded the corresponding products in satisfactory yields of 65–78% (4g–4k). When 5-bromotetrahydroquinoline was used as the substrate, its reaction with 1,2-diphenyldiselane yielded the corresponding product 4l in only 5% yield. Even after extending the reaction time to 12 hours, a significant amount of unreacted starting material remained. When the C-7 position of tetrahydroquinoline was substituted with Cl or Br, the target products could be obtained in moderate yields (4m, 4n, 4r). 8-Methyltetrahydroquinoline also participated in the reaction, but the target product 4o was obtained in a low yield of only 26%, with a substantial quantity of unreacted starting material remaining. In addition to diaryl diselenides, dialkyl diselenides were also compatible with the reaction. 1,2-Dimethyldiselane and 1,2-dibenzyldiselane provided products 4p and 4q with satisfactory yields of 57% and 81%, respectively. Interestingly, under these reaction conditions, C-6-substituted tetrahydroquinoline, 6-methyl-tetrahydroquinoline, also participated in the reaction, leading to the formation of C-8 selenylated tetrahydroquinoline (4a′), although only a 27% yield was obtained, with a large amount of starting material remaining. A possible reaction mechanism for the formation of 4a′ is illustrated in Section 4.3 of the ESI.†
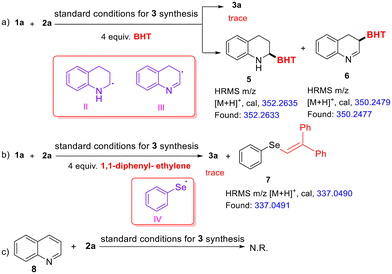
Next, to gain deeper insights into the reaction mechanism, a series of experiments were conducted. To confirm whether the reaction involves a radical process, radical trapping experiments were performed (Scheme 2a & b). Under the standard conditions for synthesizing products 3, adding 4 equivalents of the radical scavengers butylated hydroxytoluene (BHT) and 1,1-diphenyl-ethylene (DPE) separately to the model reaction completely inhibited the formation of product 3a. Meanwhile, high-resolution mass spectrometry (HRMS) detected the adducts 5, 6, and 7 formed between the radicals and the scavengers. These findings indicate that the reaction proceeds through a radical pathway, with radicals II, III and IV likely serving as intermediates. However, under the standard conditions for synthesizing products 4, only adduct 7 was detected. (see ESI, 4.2 for details†).
Furthermore, to determine whether the formation of 3a involves the initial oxidation of tetrahydroquinoline to quinoline, followed by its reaction with diselenide, a control experiment was conducted (Scheme 2c). Under the standard conditions for synthesizing 3a, replacing tetrahydroquinoline with quinoline (8) did not result in the formation of 3a. This suggests that the formation of 3a is initiated by the reaction of tetrahydroquinoline with 1,2-diphenyldiselane, followed by oxidation to produce the target compound.
Circular voltammetry (CV) experiments were performed using nBu4NClO4 as the supporting electrolyte (see ESI, 4.1†). The results showed that the oxidation potentials of NaI (+0.75 V vs. SCE in MeCN) and TEMPO (+0.85 V vs. SCE in MeCN) were lower than those of 1a (+1.10 V vs. SCE in MeCN) and 2a (+1.55 V vs. SCE in MeCN) (Fig. 5). Thus, it can be inferred that in the reaction, NaI and TEMPO are preferentially oxidized at the anode. Meanwhile, the reduction potentials of 1a and 2a were measured, revealing values of −2.2 V (vs. SCE in MeCN) for 1a and −1.75 V (vs. SCE in MeCN) for 2a (Fig. 6). These results indicate that although both 1a and 2a are reducible, they exhibit relatively high reduction potentials.
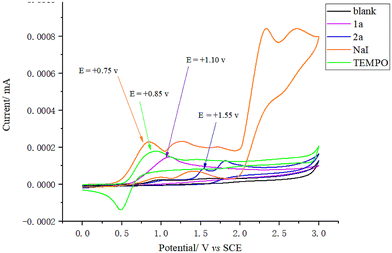 | ||
| Fig. 5 Cyclic voltammogram of NaI, TEMPO, 1a, and 2a in an electrolyte of nBu4NClO4 (0.05 M) in MeCN from 0 to +3.0 V. | ||
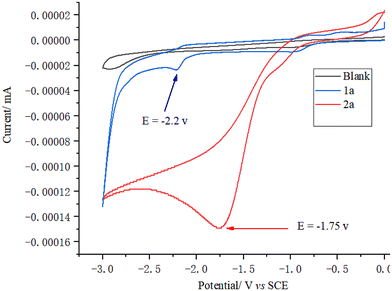 | ||
| Fig. 6 Cyclic voltammogram of 1a and 2a in an electrolyte of nBu4NClO4 (0.05 M) in MeCN from 0 to −3.0 V. | ||
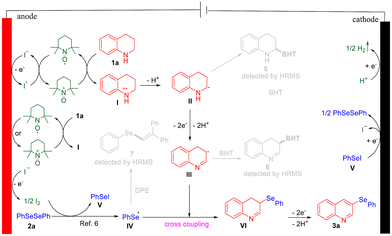
Finally, on the basis of the mechanistic exploration experiments and related literature,6,22,23 we proposed a possible reaction mechanism for the synthesis of 3a. During the optimization of reaction conditions, it was found that in the absence of TEMPO, 3a was barely formed, and significant side reactions occurred. Thus, we hypothesized that both TEMPO and NaI might function as redox mediators, ensuring high selectivity in the reaction.6,24 The proposed mechanism is outlined as follows (Fig. 7). First, I− loses an electron at the anode to form an iodine radical,23 which subsequently undergoes a single electron transfer (SET) with TEMPO to generate TEMPO+. Alternatively, TEMPO may also be directly oxidized to TEMPO+ at the anode. The TEMPO+ then participates in another SET with tetrahydroquinoline (1a), yielding radical intermediate I.10,25 Radical intermediate I loses a proton to form radical intermediate II, which is further oxidized to produce radical intermediate III. Simultaneously, I− undergoes self-coupling at the anode after losing an electron, producing I2. I2 reacts with 1,2-diphenyldiselane (2a), forming phenylselenyl radical IV and phenylselanyl iodide V.6 Subsequently, radical intermediates III and IV undergo radical cross-coupling to form intermediate VI, which undergoes electrooxidative aromatization to yield the desired product 3a. At the cathode, V is reduced to release I− and regenerate diselenide, while protons are reduced to form hydrogen gas. Intermediates II, III, and IV were captured individually by radical scavengers and identified using HRMS.
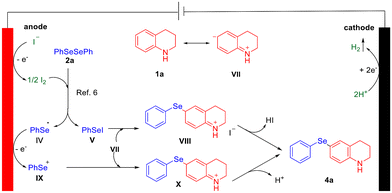
Based on the relevant literature and the experimental results, a possible mechanism for the generation of 4a is proposed (Fig. 8). Initially, I− is oxidized at the anode to produce I2, which reacts with 2a to generate the phenylselenyl radical IV and phenylselanyl iodide V.6 In tetrahydroquinoline (1a), the sp3-N atom enhances the nucleophilicity at the C-6 position, enabling 1a to resonate into VII. The intermediate V, acting as an electrophile, attacks the C-6 position of VII, leading to the formation of intermediate VIII, which subsequently eliminates one molecule of HI and undergoes aromatization to yield the target product 4a. Additionally, the phenylselenyl radical IV undergoes oxidation at the anode to form the selenium cation IX, which then attacks VII to produce intermediate X. This intermediate undergoes deprotonation and aromatization to form the target product 4a. Simultaneously, H+ is reduced at the cathode to form hydrogen gas.
By comparing the two reactions mentioned above, we observe an intriguing phenomenon. In the presence of TEMPO, the reaction yields product 3a; however, in its absence, product 4a is formed. We speculate that this difference arises as follows: TEMPO, with its relatively low oxidation potential, is readily oxidized to TEMPO+, which can undergo SET with tetrahydroquinoline (1a). This process allows 1a to be oxidized at a lower potential, generating the radical intermediate I, which ultimately leads to the formation of product 3a. In contrast, without TEMPO, 1a is less susceptible to oxidation, favoring an electrophilic substitution pathway that produces product 4a. In both cases, the indirect electrolysis facilitated by TEMPO or I− operates at a lower potential than direct electrolysis, thereby enhancing the reaction efficiency and selectivity.6,24
Conclusion
In summary, we have developed a simple and environmentally friendly electrochemical synthesis method for the selenylation of tetrahydroquinolines with diselenides. By adjusting the reaction conditions, various products can be selectively synthesized. Utilizing TEMPO as a redox mediator successfully achieves the challenging C-3 selenylation of quinolines, while in the absence of TEMPO, C-6 selenylated tetrahydroquinolines are obtained. This green approach not only exhibits excellent selectivity and broad functional group tolerance but also provides flexibility in adjusting reaction conditions to produce diverse outcomes. Given the valuable properties of selenium compounds and the privileged structures of quinolines and tetrahydroquinolines, we believe this method holds significant promise for advancing drug discovery and synthetic chemistry.Author contributions
Lan-Xi Zong: investigation, methodology, writing – original draft; Yu-Fang Tan: methodology, investigation; Yu-Hao Yang: methodology, investigation; Yan-Hong He: conceptualization, funding acquisition, supervision, writing – review & editing; Zhi Guan: conceptualization, funding acquisition, supervision, writing – review & editing.Data availability
The data supporting this article have been included as part of the ESI.†Conflicts of interest
The authors declare no conflict of interest.Acknowledgements
We gratefully acknowledge financial support from the National Natural Science Foundation of China (No. 22378334 and 22477103), and Innovation Research 2035 Pilot Plan of Southwest University (SWU-XDZD22011).References
- G. Huang, C. M. Solano, J. Melendez, J. Shaw, J. Collins, R. Banks, A. K. Arshadi, R. Boonhok, H. Min, J. Miao, D. Chakrabarti and Y. Yuan, J. Med. Chem., 2020, 63, 11756–11785 CrossRef CAS PubMed.
- A. Dorababu, Arch. Pharm., 2022, 355, e2100345, DOI:10.1002/ardp.202100345.
- Y. Luo, X. Yue, P. Wei, A. Zhou, X. Kong and S. Alimzhanova, Sci. Total Environ., 2020, 747, 141136–141146 CrossRef CAS PubMed.
- R. S. Upadhayaya, P. D. Shinde, S. A. Kadam, A. N. Bawane, A. Y. Sayyed, R. A. Kardile, P. N. Gitay, S. V. Lahore, S. S. Dixit, A. Foldesi and J. Chattopadhyaya, Eur. J. Med. Chem., 2011, 46, 1306–1324 CrossRef CAS PubMed.
- K. Kaur, M. Jain, R. P. Reddy and R. Jain, Eur. J. Med. Chem., 2010, 45, 3245–3264 CrossRef CAS PubMed.
- P. Qu, Y. Q. Jiang, Y. H. Wang and G. Q. Liu, Green Chem., 2023, 25, 7485–7507 RSC.
- (a) C. W. Nogueira, G. Zeni and J. B. T. Rocha, Chem. Rev., 2004, 104, 6255–6286 CrossRef CAS PubMed; (b) A. J. Mukherjee, S. S. Zade, H. B. Singh and R. B. Sunoj, Chem. Rev., 2010, 110, 4357–4416 CrossRef CAS PubMed; (c) W. Hou, H. Dong, X. Zhang, Y. Wang, L. Su and H. Xu, Drug Discovery Today, 2022, 27, 2268–2277 CrossRef CAS PubMed.
- (a) Q. Guan, C. Han, D. Zuo, M. A. Zhai, Z. Li, Q. Zhang, Y. Zhai, X. Jiang, K. Bao, Y. Wu and W. Zhang, Eur. J. Med. Chem., 2014, 87, 306–315 CrossRef CAS PubMed; (b) Z. Wen, J. Xu, Z. Wang, H. Qi, Q. Xu, Z. Bai, Q. Zhang, K. Bao, Y. Wu and W. Zhang, Eur. J. Med. Chem., 2015, 90, 184–194 CrossRef CAS PubMed.
- (a) S. Shabaan, L. A. Ba, M. Abbas, T. Burkholz, A. Denkert, A. Gohr, L. A. Wessjohann, F. Sasse, W. Weber and C. Jacob, Chem. Commun., 2009, 4702–4704 RSC; (b) A. Capperucci, M. Coronnello, F. Salvini, D. Tanini, S. Dei, E. Teodori and L. Giovannelli, Bioorg. Chem., 2021, 110, 104812–104820 CrossRef CAS PubMed; (c) S. Shaaban, D. Vervandier-Fasseur, P. Andreoletti, A. Zarrouk, P. Richard, A. Negm, G. Manolikakes, C. Jacob and M. Cherkaoui-Malki, Bioorg. Chem., 2018, 80, 43–56 CrossRef CAS PubMed; (d) A. Arora, S. Singh, P. Oswal, D. Nautiyal, G. K. Rao, S. Kumar and A. Kumar, Coord. Chem. Rev., 2021, 438, 213885–213920 CrossRef CAS.
- X. Y. Zhou, M. Zhang, Z. Liu, J. H. He and X. C. Wang, J. Am. Chem. Soc., 2022, 144, 14463–14470 CrossRef CAS PubMed.
- (a) R. Francke and R. D. Little, Chem. Soc. Rev., 2014, 43, 2492–2521 RSC; (b) J. E. Nutting, M. Rafiee and S. S. Stahl, Chem. Rev., 2018, 118, 4834–4885 CrossRef CAS PubMed; (c) F. Wang and S. S. Stahl, Acc. Chem. Res., 2020, 53, 561–574 CrossRef CAS PubMed.
- D. Kundu, N. Mukherjee and B. C. Ranu, RSC Adv., 2013, 3, 117–125 RSC.
- A. K. Pradhan, P. K. Behera, P. Choudhury, P. Behera, A. Swain, P. Bag, D. Pany and L. Rout, ChemistrySelect, 2023, 8, e202300378 CrossRef.
- P. Choudhury, A. K. Pradhan, S. Jena, B. K. Sahoo, S. K. Sahu, P. K. Behera, P. Behera, A. Swain and L. Rout, Eur. J. Org. Chem., 2022, e202201194 CrossRef CAS.
- X. Sheng, M. Yan, B. Zhang, W. Y. Wong, N. Kambe and R. Qiu, ACS Catal., 2023, 13, 9753–9765 CrossRef CAS.
- X. Ren, Q. Liu, Z. Yang, Z. Wang and X. Chen, Chin. Chem. Lett., 2023, 34, 107821–107825 CrossRef CAS.
- J. Liu, M. Tian, Y. Li, X. Shan, A. Li, K. Lu, M. Fagnoni, S. Protti and X. Zhao, Eur. J. Org. Chem., 2020, 7358–7367 CrossRef.
- D. H. R. Barton, X. Lusinchi and P. Hillibet, Tetrahedron, 1985, 41, 4727–4738 CrossRef CAS.
- (a) H.-T. Tang, Y.-Z. Pan and Y.-M. Pan, Green Chem., 2023, 25, 8313–8327 RSC; (b) M. Yan, Y. Kawamata and P. S. Baran, Chem. Rev., 2017, 117, 13230–13319 CrossRef CAS PubMed; (c) M. Regnier, C. Vega, D. I. Ioannou and T. Noël, Chem. Soc. Rev., 2024, 53, 10741–10760 RSC; (d) Y. Yuan, J. Yang and A. W. Lei, Chem. Soc. Rev., 2021, 50, 10058–10086 RSC.
- (a) X.-Q. Zhou, H.-T. Tang, F.-H. Cui, Y. Liang, S.-H. Li and Y.-M. Pan, Green Chem., 2023, 25, 5024–5029 RSC; (b) Z. Y. Chen, Y. Wang, C. J. Hu, D. X. Wang, P. L. Lei, H. Yi, Y. Yuan and A. W. Lei, Org. Lett., 2022, 24, 3307–3312 CrossRef CAS PubMed.
- W.-J. Wei, X.-Y. Wang, H.-T. Tang, F.-H. Cui, Y.-Q. Wu and Y.-M. Pan, Sci. China: Chem., 2024, 67, 3382–3388 CrossRef CAS.
- (a) R. Francke and R. D. Little, Chem. Soc. Rev., 2014, 43, 2492–2521 RSC; (b) J. E. Nutting, M. Rafiee and S. S. Stahl, Chem. Rev., 2018, 118, 4834–4885 CrossRef CAS PubMed; (c) F. Wang and S. S. Stahl, Acc. Chem. Res., 2020, 53, 561–574 CrossRef CAS PubMed.
- C. Li, C. C. Zeng, L. M. Hu, F. L. Yang, S. J. Yoo and R. D. Little, Electrochim. Acta, 2013, 114, 560–566 CrossRef CAS.
- C. C. Zeng, N. T. Zhang, C. M. Lam and R. D. Little, Org. Lett., 2012, 14, 1314–1317 CrossRef CAS PubMed.
- (a) A. Palma, J. Cárdenas and B. A. Frontana-Uribe, Green Chem., 2009, 11, 283–293 RSC; (b) M. Rafiee, K. C. Miles and S. S. Stahl, J. Am. Chem. Soc., 2015, 137, 14751–14757 CrossRef CAS PubMed.
Footnotes |
| † Electronic supplementary information (ESI) available. See DOI: https://doi.org/10.1039/d4gc06230b |
| ‡ These authors contributed equally to this work. |
| This journal is © The Royal Society of Chemistry 2025 |