Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
The effects of three different daily plant stanol ester intakes on influenza vaccination responses: a double-blind, randomized, placebo-controlled intervention study in older adults
Willem
Zwaan
 ,
Lieve
van Brakel
,
Ronald P.
Mensink
and
Jogchum
Plat
*
,
Lieve
van Brakel
,
Ronald P.
Mensink
and
Jogchum
Plat
*
Department of Nutrition and Movement Sciences, Institute of Nutrition and Translational Research in Metabolism (NUTRIM), Maastricht University Medical Centre, Maastricht, the Netherlands. E-mail: j.plat@maastrichtuniversity.nl
First published on 24th November 2025
Abstract
Aging is accompanied by a gradual decline in immune function. Previously, we demonstrated that daily intake of 4 g plant stanols, provided as fatty-acid esters, increased antibody titre responses following different vaccinations. Effects of lower intakes, however, are unknown. We therefore evaluated whether daily consumption of 2, 3 or 4 g of plant stanols, provided as fatty-acid esters, improved immune responses following influenza vaccination in older adults. A double-blind, randomized, placebo-controlled parallel trial was performed. Older participants (59–87 years) consumed products with added plant stanols (2, 3 or 4 g d−1 provided as fatty-acid esters) or control products for approximately 14 days prior to vaccination until 4 weeks post-vaccination. Antibody titres, serum metabolic risk markers, and haematological and immunological parameters were measured and statistically evaluated using linear mixed models. Data of 128 participants were analysed. Changes in IgM and IgG concentrations did not differ significantly between the control and plant stanol groups. Also, changes in Hemagglutination Inhibition (HI) antibody titres were not significantly different between the groups for either the Phuket or Victoria strains. Serum LDL-cholesterol concentrations decreased significantly in the 4 g d−1 and 3 g d−1 groups compared with control. Serum LDL-cholesterol concentrations in the 2 g d−1 group were also lowered, but did not reach statistical significance. No between-group differences in haematological and immunological parameters were observed. Daily consumption of 2, 3 or 4 g of plant stanols provided as fatty-acid esters, did not improve vaccination responses following annual influenza vaccination in men and women aged 60 years or older.
Introduction
Ageing is characterized by various physiological and health-related changes, including a gradual decline in immune function. This phenomenon, known as immunosenescence,1,2 may result in age-related compromised immune responses, increasing the susceptibility to infections, and consequently, the risk of hospitalisation and mortality following infections.3 Influenza virus infections, for example, result annually in 3 to 5 million cases of severe illness and 290![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 to 650
000 to 650![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 deaths worldwide, mainly in older adults.4,5 Vaccinations remain the most effective strategy for preventing or decreasing the incidence of severe illness and associated mortality following influenza infections.1,3 However, due to immunosenescence, older adults may show weaker immune responses to vaccinations as compared to younger adults.6 Therefore, it seems essential to boost vaccination responses in susceptible populations. A promising approach to modulate immune responses is through dietary interventions.7 However, due to the complex nature of the immune system, assessing the impact of nutritional interventions on immune functioning is challenging. The European Food Safety Authority (EFSA) states that the stimulation of vaccine responses, either through an increased number of individuals acquiring protective antibody concentrations or through increases in vaccine-specific antibody titres, is an appropriate outcome for determining the immunomodulating effects of dietary approaches.1,8,9 Therefore, a vaccination model such as monitoring immune responses after the seasonal influenza vaccination, can be considered as the gold standard to determine effects of foods and food components on immune function.7
000 deaths worldwide, mainly in older adults.4,5 Vaccinations remain the most effective strategy for preventing or decreasing the incidence of severe illness and associated mortality following influenza infections.1,3 However, due to immunosenescence, older adults may show weaker immune responses to vaccinations as compared to younger adults.6 Therefore, it seems essential to boost vaccination responses in susceptible populations. A promising approach to modulate immune responses is through dietary interventions.7 However, due to the complex nature of the immune system, assessing the impact of nutritional interventions on immune functioning is challenging. The European Food Safety Authority (EFSA) states that the stimulation of vaccine responses, either through an increased number of individuals acquiring protective antibody concentrations or through increases in vaccine-specific antibody titres, is an appropriate outcome for determining the immunomodulating effects of dietary approaches.1,8,9 Therefore, a vaccination model such as monitoring immune responses after the seasonal influenza vaccination, can be considered as the gold standard to determine effects of foods and food components on immune function.7
Several well-controlled randomized intervention trials have already shown that dietary components such as micronutrients and omega-3 long-chain polyunsaturated fatty acids can positively affect the immune system in susceptible populations.1,10–13 Previously, we have shown that daily consumption of 4 grams plant stanols, provided as their fatty-acid esters, increased antibody titres in patients with asthma after a hepatitis A vaccination14 or in individuals with overweight/obesity after a COVID-19 vaccination.15 These findings suggest that plant stanol esters may also be beneficial for immunoscenescent populations such as older adults. Moreover, whether comparable effects on immune responses can be achieved at lower intakes of plant stanols, as opposed to the previously used 4 grams per day (g d−1), is unknown. We therefore examined whether daily consumption of 2, 3 or 4 grams of plant stanols provided as fatty-acid esters improved immune responses following the annual influenza vaccination, compared to control, in men and women aged 60 years or older during the 2022/2023 influenza season. Secondary aims were to assess the effects of the different daily plant stanol intakes on (1) haematological and immunological parameters (high-sensitivity C-reactive protein [hs-CRP], cytokines and leukocyte counts) and (2) metabolic parameters (serum lipid and lipoprotein profiles, and glucose).
Methods
Study participants
Male and female participants were recruited via advertisements in local newspapers and university buildings in Maastricht and surrounding areas, the Netherlands. Participants were eligible if they were aged 60 years or older before May 1, 2023, had a BMI between 20 and 35 kg m−2, were not using food products enriched with plant sterols/stanols, were willing to abstain from these products during the study, and were willing to keep the intake of fish oil and vitamin supplements constant during the study period. Participants were excluded if they had already received an influenza vaccination in 2022, experienced an influenza infection in 2022, had known allergic reactions to any of the ingredients in the study products or vaccine, donated blood in the month prior to the study or planned to do so during the study, regularly consumed more than 20 units of alcohol per week or used soft and/or hard drugs. Initial screening was conducted via a telephone questionnaire and individuals who met all inclusion criteria and were willing to participate were invited to the university. During the initial visit to the university, weight and height were measured to calculate BMI to ensure that the self-reported weight and height were correct. All participants provided signed informed consent prior to the measurements.Study design
This double-blind, randomized, placebo-controlled clinical trial with a parallel design lasted six weeks, in which six visits to the metabolic research unit of our university were scheduled. During these visits, venous blood samples were collected, and anthropometrics were measured. Participants started consuming the study products immediately after the first visit, generally two weeks – but at least nine days – before receiving the influenza vaccination. Visit 2 was scheduled approximately 1 day before, but never after receiving the vaccination. The next four visits 3–6 were scheduled for four consecutive weeks immediately following the vaccination (Fig. 1). Scheduling of the influenza vaccination was imposed for each participant individually by the general practitioner according to the vaccination program authorized by the Dutch government. As a result, slight differences in the number of days between the first two visits occurred. The influenza vaccinations in this study were either the Vaxigrip Tetra (Sanofi Pasteur Europe, Lyon, France), Invluvac Tetra (Abbott Biologicals BV, Weesp, the Netherlands) or the Fluarix Tetra (GlaxoSmithKline BV, Amersfoort, the Netherlands). All visits were scheduled between September 2022 and December 2022. Before each visit, participants had to refrain from alcohol for at least 24 hours. In addition, participants were fasted for at least 12 hours before visits 1, 2 and 6. Participants kept a diary during the study to record possible medical complaints, medication use, side effects of the vaccination, visits to a hospital or general practitioner, and the number of study products consumed. Dietary intakes of the participants were estimated via a food frequency questionnaire (FFQ) at the beginning (visit 1) and the end (visit 6) of the study. Participants were instructed to maintain their habitual diet throughout the study. This study was approved by the medical ethical committee of Maastricht University and Medical Centre (NL82182.068.22) and registered at clinicaltrials.gov (NCT05706350).Study products
Participants were randomly allocated to the control group, or to one of the three intervention groups (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4) using a computer-generated block randomisation with a block size of 8. The test products were soft chews provided by Raisio Nutrition Ltd (Raisio, Finland) containing per chew 0.5 g plant stanols, provided as their fatty-acid esters and handed out to the participant by a independent researcher. When referred to our own study products, this can be described from now on as plant stanols. The control chews had a comparable composition, but contained no plant stanols. Participants in the intervention groups consumed either 4, (2.0 gr plant stanols), 6 (3.0 gr plant stanols) or 8 (4.0 gr plant stanols) chews per day, equally divided over breakfast and dinner to ensure optimal bioavailability of the plant stanols. Participants in the control group received 6 control chews without added plant stanols per day (3 with breakfast plus 3 with dinner). The chews had to be stored at room temperature and study products that were left over at the end of the study had to be returned to the research unit.
4) using a computer-generated block randomisation with a block size of 8. The test products were soft chews provided by Raisio Nutrition Ltd (Raisio, Finland) containing per chew 0.5 g plant stanols, provided as their fatty-acid esters and handed out to the participant by a independent researcher. When referred to our own study products, this can be described from now on as plant stanols. The control chews had a comparable composition, but contained no plant stanols. Participants in the intervention groups consumed either 4, (2.0 gr plant stanols), 6 (3.0 gr plant stanols) or 8 (4.0 gr plant stanols) chews per day, equally divided over breakfast and dinner to ensure optimal bioavailability of the plant stanols. Participants in the control group received 6 control chews without added plant stanols per day (3 with breakfast plus 3 with dinner). The chews had to be stored at room temperature and study products that were left over at the end of the study had to be returned to the research unit.
Biochemical analysis
During each visit, blood was collected in serum separator tubes (Becton, Dickson and Company, New Jersey, USA). These 8.5 ml tubes were centrifuged at 1300g for 10 minutes at 21 °C after coagulation at room temperature for at least 30 minutes. During visits 1, 2, and 6, blood was collected in two additional EDTA tubes (Becton, Dickson and Company). After sampling, these EDTA tubes (4 ml) were directly placed on ice. One tube was centrifuged at 1300g for 10 minutes at 4 °C, while the second tube was used fresh for leukocyte total and differential cell counts without centrifugation. After centrifugation, serum and EDTA plasma aliquots were snap-frozen in liquid nitrogen and stored at −80 °C until further analysis.Serum non-cholesterol sterols
Non-cholesterol sterols were measured in the serum samples of visits 1, 2, 4 and 6. Serum cholesterol and cholestanol concentrations were quantified using gas chromatography with flame ionization detector (GC-FID), as described previously.16 In addition, serum concentrations of plant stanols (sitostanol and campestanol), plant sterols (sitosterol and campesterol), and intermediates in the endogenous cholesterol synthesis pathways (desmosterol and lathosterol) were analysed using a gas chromatography triple quadrupole mass spectrometer (GC-MS/MS) as described.16 Serum non-cholesterol sterol concentrations were standardized for total cholesterol (TC) concentrations, as analysed with the GC-FID. Changes in serum TC-standardized sitostanol and campestanol concentrations were used as compliance markers.Antibody responses
The Haemagglutination Inhibition (HI) assay was performed to determine the amount of antibodies against the specific influenza virus in serum samples from visits 2, 3, 4, 5 and 6. The assay was conducted according to a standardized protocol by Viroclinics-DDL, a Cerba Research Company (Rotterdam, the Netherlands), for two influenza strains that were present in the vaccines given to the participants: A/Victoria/2570/2019 (H1N1)pdm09-like virus (A/Victoria/2570/2019, IVR-215) and B/Phuket/3073/2013-like virus (B/Phuket/3073/2013, wild type). Briefly, serum samples were pre-treated overnight at 37 °C with Receptor Destroying Enzyme (RDE), provided as a Vibrio cholerae culture filtrate, to remove any non-specific inhibitors and to allow quantitation of virus-specific antibodies only. Next, samples were incubated at 56 °C for 1 hour to inactivate RDE. Turkey erythrocytes (TRBC) were collected by standard phlebotomy and centrifugation, washed in a phosphate-buffered saline solution (PBS), and resuspended to a 1% (v/v) solution. Two-fold serial dilutions of pretreated serum samples were made in 96-wells microtiter plates, all in duplicate. Then, a fixed amount of virus antigen was added to each well and the plate was carefully mixed before incubation for 1 hour at room temperature. After incubation, 1% TRBC suspension was added to all wells and incubation was continued for 1 hour at 4 °C. The plate read out was performed by visual scoring of TRBC sedimentation by two separate technicians, each one analysing one of the duplicate plates. Duplicate results were subsequently cross-checked for consistency. Results were only accepted if the duplicate plates titres were within one two-fold dilution. After acceptance, results of the duplicates were averaged. For the HI assay outcomes, the geometric mean titres (GMTs) were calculated. Besides the HI assay, serum anti-Flu B/Phuket HA IgM and IgG concentrations were determined in the samples from visits 2, 3, 4, 5 and 6 (Meso Scale Diagnostics, Rockville, MD, USA). The IgM serum antibody titres and IgG antibody titres were measured using respectively the V-PLEX Respiratory Panel 1 (IgM) and V-PLEX Respiratory Panel 1 (IgG) (Meso Scale Diagnostics, Rockville, MD, USA). For the A/Victoria/2570/2019 strain, no panel was available.Cytokines, hs-CRP and leukocyte count
Circulating cytokine concentrations (interleukin-6 [IL-6], IL-8 and tumour necrosis factor α [TNFα]) were analysed in serum samples from visits 1, 2 and 6 according to the protocol of the manufacturer (V-PLEX Human Inflammatory Panel II (4-plex) and Meso Scale Diagnostics, Rockville, MD, USA). Serum hs-CRP concentrations (CRP CP, Horiba ABX) were analysed in samples from visits 1,2 and 6. Leukocyte total and differential count (visits 1,2 and 6) were analysed in fresh EDTA full blood using the haematology analyser Sysmex XN900 (Sysmex Europe GmbH) at the Central Diagnostics Laboratory of the MUMC+.Serum lipids, lipoproteins and glucose
Lipids, lipoproteins and glucose were analysed in samples of visits 1, 2 and 6 at the Laboratory of the Central Diagnostics at the MUMC+. Serum triacylglycerol (TAG), TC and HDL cholesterol (HDL-C) concentrations were determined enzymatically by using cholesterol esterase and cholesterol oxidase (all using Cobas Pro c503 module). Additionally, plasma glucose concentrations were analysed using an enzymatic reference method with hexokinase on the Cobas Pure c303 module. Serum LDL cholesterol (LDL-C) concentrations were calculated using the Friedewald formula.17Statistical analyses
The power calculation was based on estimated effect sizes and SDs using results from earlier vaccination studies performed at our department, in which the effects of plant stanol esters were examined on hepatitis A and COVID-19 vaccination responses.14,15 Based on these earlier findings, we aimed for a true effect size of 60% (SD: 86%) in the vaccine specific immune response between any of the three intervention groups and the control group. The study was not powered to detect significant differences between the three plant stanol doses. For a power of 80% and a 2-sided significance level of 0.017, a sample size of 172 participants was needed. All data are presented as means ± SD or median (IQR) when not normally distributed. Normality was checked by visual inspection of histograms and Q–Q plots. Differences between the groups at baseline were analysed using independent sample t tests, chi-square, or Mann–Whitney U tests as appropriate. To determine possible significant differences in changes over time between the plant stanol groups and the control group, linear mixed model analyses were performed. A per protocol analysis was used to investigate the effect of the intervention. For each model the optimal covariance structure was determined using a top-down approach, starting with most complex covariance structure (e.g., unstructured) and sequentially testing more simple structures. The optimal structure was chosen using maximum likelihood estimates, assessed by Akaike's Information Criterion (AIC), Bayesian Information Criterion (BIC), and likelihood ratio tests where applicable. In all models, sex, time, and study group were included as fixed factors. Subject number was added as random factor. The interaction term group × time was included in the initial model, but excluded in the final model when it did not reach statistical significance. If this interaction term was significant, a Bonferroni correction was performed to determine at which timepoints the outcomes were different between the plant stanol and control groups. To correct for inter-individual differences in study duration, the time between the start of the study (visit 1) and the day before vaccination (visit 2) was added to all models. This variable, called time V1-vaccination, was removed when not statistically significant. All models were corrected for corresponding baseline concentrations. Differences in dietary intake (FFQ) between groups were compared using ANOVA or a Kruskal Wallis test as appropriate. In additional analysis of the antibody outcomes, all three intervention groups were combined and compared to the control group using the same approaches. P-Values of <0.05 were considered to be statistically significant, except for group-to-control group comparison where p < 0.017 was considered as statistically significant. All analysis were performed using IBM SPSS Statistics version 28.0 for Windows (IBM Corp, NY, USA).Results
Study participants
The flow chart shows that 206 individuals received detailed study information, of which ultimately 139 were enrolled in the study (Fig. 2). These participants were randomized to receive study products from one of the intervention groups providing 2, 3, or 4 g plant stanols, or the control products. Nine participants dropped out due to various reasons. Moreover, two participants from the plant stanol groups were excluded, because they were considered as non-compliant based on a lack of change in TC-standardized plant stanol levels. Ultimately, the data of 128 participants who completed the study was included in the statistical analysis. The mean age of the total population was 70 ± 7 years, and the mean BMI was 26.1 ± 3.2 kg m−2. When comparing the four study groups at baseline, BMI was significantly higher in the 4 g d−1 group as compared to the other groups, while serum HDL-C was significantly lower in the 4 g d−1 group compared to all other groups. All other baseline characteristics were comparable between the four study groups (Table 1).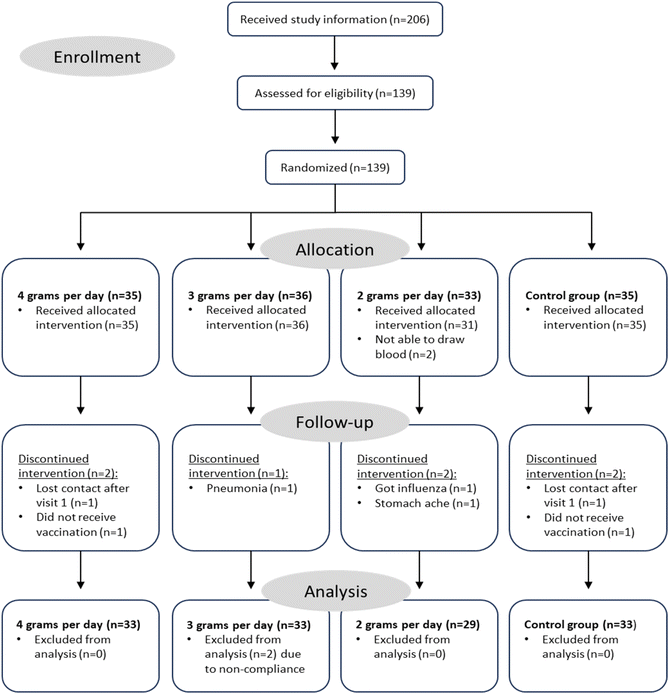 | ||
| Fig. 2 Flow chart of the study. In total, 130 participants completed the study and 128 participants were included in the analysis. | ||
| Total group (N = 128) | 4 g d−1 group (N = 33) | 3 g d−1 group(N = 33) | 2 g d−1 group (N = 29) | Control group (N = 33) | Between groups p-value | |
|---|---|---|---|---|---|---|
| A = ANOVA, K = Kruskal Wallis test, C = Chi-square, Tukey post-hoc test used when p-value of ANOVA test was statistically significant.a Indicates that 4 g d−1 group has a significantly higher BMI at baseline compared to the other three groups.b Indicates that 4 g d−1 group has a significantly lower HDL-C baseline value compared to the 2 g d−1 group. Abbreviations: time V1-vaccination = time between the start of the study and the influenza vaccination date, SD = standard deviation, IQR = interquartile range presented as Q1–Q3, BMI = body mass index, TAG = triacylglycerol, TC = total cholesterol, HDL-C = high density lipoprotein cholesterol, LDL-C = low density lipoprotein cholesterol, hs-CRP = high sensitive C-reactive protein. Statistically significant if p ≤ 0.05. | ||||||
| Age, years (SD)A | 70 (7) | 71 (7) | 71 (7) | 70 (7) | 69 (7) | 0.42 |
| Sex (male), N (%)C | 60 (46.2) | 18 (54.5) | 14 (42.4) | 16 (55.2) | 11 (33.3) | 0.17 |
| Time V1-vaccination, days (IQR)K | 14 (12–14) | 14 (13–15) | 13 (12–15) | 14 (12–14) | 14 (12–14) | 0.89 |
| BMI, kg m−2 (SD)A | 26.1 (3.2) | 27.9a (3.4) | 25.5 (3.0) | 25.4 (2.8) | 25.6 (3.2) | <0.01 |
| TAG, mmol L−1 (SD)A | 1.31 (0.50) | 1.37 (0.50) | 1.24 (0.48) | 1.23 (0.45) | 1.40 (0.56) | 0.39 |
| TC, mmol L−1 (SD)A | 5.62 (1.2) | 5.55 (1.17) | 5.45 (1.16) | 5.49 (1.01) | 5.96 (1.34) | 0.28 |
| HDL-C, mmol L−1 (SD)A | 1.64 (0.45) | 1.45b (0.42) | 1.69 (0.50) | 1.79 (0.47) | 1.66 (0.37) | 0.02 |
| LDL-C, mmol L−1 (SD)A | 3.37 (1.02) | 3.47 (0.95) | 3.20 (0.96) | 3.15 (0.94) | 3.66 (1.19) | 0.16 |
| Glucose, mmol L−1 (SD)A | 5.58 (0.50) | 5.75 (0.96) | 5.58 (0.79) | 5.50 (0.51) | 5.49 (1.07) | 0.62 |
| Hs-CRP, mg dL−1 (IQR)K | 1.47 (0.73–3.62) | 1.65 (0.85–3.15) | 1.59 (0.72–3.89) | 1.30 (0.65–1.92) | 1.74 (0.86–4.70) | 0.23 |
Non-cholesterol sterol concentrations and compliance
In all three intervention groups, changes in levels of serum TC-standardized plant stanol and other non-cholesterol sterols were observed (SI Table S1). Serum TC-standardized sitostanol levels increased in all three intervention groups from visit 2 onwards with significant differences in changes as compared to the control group at V2, V4 and V6 (group × time p < 0.01). Table 2 shows that TC-standardized campestanol levels were also significantly increased in all intervention groups (group × time p < 0.01). These changes clearly indicated excellent compliance. The validated marker for intestinal cholesterol absorption, i.e. the TC-standardized campesterol level,18 was significantly reduced in the three intervention groups from V2 onwards. At visits 4 and 6, serum TC-standardized sitosterol levels were also significantly lower in all three intervention groups (group × time p < 0.01). Serum TC-standardized lathosterol levels, the validated marker for endogenous cholesterol synthesis,18 were significantly increased (group effect p = 0.01). In more detail, the changes vs. control for the 4 g d−1 group (20.5 [8.11, 32.9] µmol mmol−1 × 100, p < 0.01) and the 3 g d−1 group (15.0 [2.81, 27.3] µmol mmol−1 × 100, p = 0.02) significantly differed. The change for the 2 g d−1 group vs. control (13.0 [0.22, 25.8] µmol mmol−1 × 100, p = 0.05) did not reach statistical significance (Table 2). Finally, changes in serum TC-standardized desmosterol and cholestanol levels did not significantly differ between the intervention groups and the control group (p = 0.05 and p = 0.12 respectively).| p-Value | |||||
|---|---|---|---|---|---|
| Group vs. control group | Estimated marginal difference (95% CI) | Group vs. control group | Group | Group × time | |
| All models were corrected for gender and baseline concentrations. Abbreviations: 95%CI = 95% confidence interval, time V1-vaccination = time between the start of the study and the influenza vaccination date (this variable differed per participant and therefore was tested in all models), N/A = not applicable. If group × time or time V1-vaccination was N/A, this variable was not included in the final model because it did not reach statistical significance. If group was not significant, N/A is shown for the group vs. group comparison.a Significant differences between a group and the control group at visits 2,4 and 6.b Significant differences between a group and the control group at visits 2, 4 and 6 except for group 2 at visit 2. Statistically significant if p ≤ 0.05, for group vs. control group statistically significant if p ≤ 0.017. | |||||
| Compliance markers (TC-standardized plant stanol levels (µmol mmol −1 × 100)) | |||||
| Sitostanola | N/A | N/A | N/A | N/A | <0.01 |
| Campestanola | N/A | N/A | N/A | N/A | <0.01 |
| TC-standardized plant sterol and cholestanol levels (µmol mmol −1 × 100) | |||||
| Sitosterolb | N/A | N/A | N/A | N/A | <0.01 |
| Campesterola | N/A | N/A | N/A | N/A | <0.01 |
| Cholestanol | 4 g d−1 | −6.44 (−15.2, 2.30) | N/A | 0.12 | N/A |
| 3 g d−1 | −9.94 (−18.5, −1.42) | N/A | |||
| 2 g d−1 | −8.01 (−16.9, 0.85) | N/A | |||
| TC-standardized intermediates in endogenous cholesterol synthesis pathways (µmol mmol −1 × 100) | |||||
| Lathosterol | 4 g d−1 | 20.5 (8.11, 32.9) | <0.01 | 0.01 | N/A |
| 3 g d−1 | 15.0 (2.81, 27.3) | 0.02 | |||
| 2 g d−1 | 13.0 (0.22, 25.8) | 0.05 | |||
| Desmosterol | 4 g d−1 | 7.02 (1.78, 12.3) | N/A | 0.05 | N/A |
| 3 g d−1 | 5.36 (0.18, 10.5) | N/A | |||
| 2 g d−1 | 5.10 (−0.30, 10.5) | N/A | |||
Antibodies
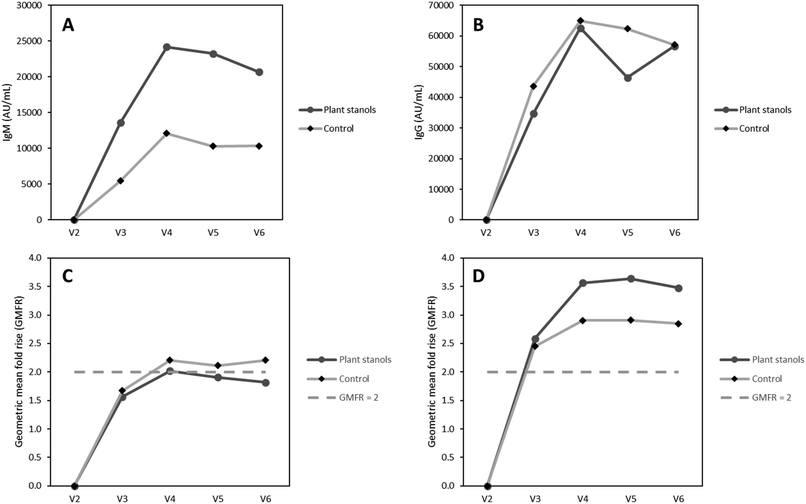
Differences in IgM antibody titres against the Flu B/Phuket/2013 HA between any of the three intervention groups and the control group did not reach statistical significance at any of the time points (Table 3). At visit 4, the average increase in IgM concentrations compared to pre-vaccination was 22![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 081 AU ml−1 (SD 65062) in the 4 g d−1 group. For the control group, the average increase was 12
081 AU ml−1 (SD 65062) in the 4 g d−1 group. For the control group, the average increase was 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 102 AU ml−1 (SD 28144), resulting in an 82% higher increase in the 4 g d−1 group compared to control group. However, this difference did not reach statistical significance. Also, the specific influenza B/Phuket/2013 HA IgG antibody titres did not significantly differ between any of the intervention groups and the control group. Averages for each group at each visit of IgM and IgG anti-influenza concentrations against the Phuket strain are presented in SI Table S2a and b. When the results of the three intervention groups (2-, 3-, and 4-gram plant stanols) were combined, the average increase at visit 4 in IgM concentrations compared to pre-vaccination was 24
102 AU ml−1 (SD 28144), resulting in an 82% higher increase in the 4 g d−1 group compared to control group. However, this difference did not reach statistical significance. Also, the specific influenza B/Phuket/2013 HA IgG antibody titres did not significantly differ between any of the intervention groups and the control group. Averages for each group at each visit of IgM and IgG anti-influenza concentrations against the Phuket strain are presented in SI Table S2a and b. When the results of the three intervention groups (2-, 3-, and 4-gram plant stanols) were combined, the average increase at visit 4 in IgM concentrations compared to pre-vaccination was 24![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 166 AU ml−1 (SD 74317) (Fig. 3A). This observed difference between the combined intervention groups and the control group was also not statistically different. In addition, increases in IgG concentrations of the combined intervention groups were not statistically different compared to the control group (Table 3). The highest average increase in IgG antibodies was observed at visit 4, corresponding with an increase of 64
166 AU ml−1 (SD 74317) (Fig. 3A). This observed difference between the combined intervention groups and the control group was also not statistically different. In addition, increases in IgG concentrations of the combined intervention groups were not statistically different compared to the control group (Table 3). The highest average increase in IgG antibodies was observed at visit 4, corresponding with an increase of 64![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 978 AU ml−1 (SD 51469) for the control group and 62
978 AU ml−1 (SD 51469) for the control group and 62![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 614 AU ml−1 (SD 107260) for the combined intervention groups (Fig. 3B). In the HI assay, the geometric means of the Victoria strain were increased by at least a 2.0 geometric mean fold rise (GMFR) in all groups, which is in line with the requested increase by the Medicinal Products for Human use (CHMP) requirements for influenza vaccines. However, no significant differences were observed between any of the intervention groups and the control group (SI Table S2a and b). In the HI assay of the Phuket strain, multiple samples had undetectable values, and the threshold of 2.0 of the GMFR was reached in only 102 participants (Table 4). Similar to the Victoria strain, no differences were observed between any of the intervention groups and the control group for the Phuket strain. When the three intervention groups were combined, conclusions did not change (Fig. 3C and D). In addition, no significant differences between the combined intervention groups and the control group were observed in seroprotection or seroconversion (Table 4).
614 AU ml−1 (SD 107260) for the combined intervention groups (Fig. 3B). In the HI assay, the geometric means of the Victoria strain were increased by at least a 2.0 geometric mean fold rise (GMFR) in all groups, which is in line with the requested increase by the Medicinal Products for Human use (CHMP) requirements for influenza vaccines. However, no significant differences were observed between any of the intervention groups and the control group (SI Table S2a and b). In the HI assay of the Phuket strain, multiple samples had undetectable values, and the threshold of 2.0 of the GMFR was reached in only 102 participants (Table 4). Similar to the Victoria strain, no differences were observed between any of the intervention groups and the control group for the Phuket strain. When the three intervention groups were combined, conclusions did not change (Fig. 3C and D). In addition, no significant differences between the combined intervention groups and the control group were observed in seroprotection or seroconversion (Table 4).
| p-Value | |||||||
|---|---|---|---|---|---|---|---|
| Group vs. control group | Estimated marginal difference (95% CI) | Group vs. control group | Group | Time | Group × time | Time V1-vaccination | |
| All models were corrected for gender and baseline concentrations. Abbreviations: 95%CI = 95% confidence interval, time V1-vaccination = time between the start of the study and the influenza vaccination date (this variable differed per participant and therefore was tested in all models), N/A = not applicable, Ig = immunoglobulin, HI = hemagglutination inhibition. If group × time or time V1-vaccination was N/A this variable was not included in the final model because it did not reach significance. If group was not significant, N/A is shown for the group vs. group comparison. | |||||||
| IgM (AU ml−1) | 4 g d−1 | −2539 (−8395, 3316) | N/A | 0.81 | <0.01 | N/A | N/A |
| 3 g d−1 | −53.6 (−6004, 5897) | N/A | |||||
| 2 g d−1 | −493 (−6514, 5529) | N/A | |||||
| Combined | −1098 (−5869, 3673) | N/A | 0.65 | <0.01 | N/A | N/A | |
| IgG (AU ml−1) | 4 g d−1 | −10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 049 (−40 049 (−40![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 916, 17 916, 17![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 390) 390) |
N/A | 0.85 | <0.01 | N/A | N/A |
| 3 g d−1 | 2113 (−29![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 259, 33 259, 33![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 485) 485) |
N/A | |||||
| 2 g d−1 | −4204 (−37![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 262, 28 262, 28![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 854) 854) |
N/A | |||||
| Combined | −2876 (−28![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 096, 22 096, 22![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 343) 343) |
N/A | 0.82 | <0.01 | N/A | N/A | |
| HI assay Victoria strain | 4 g d−1 | −17.4 (−144, 109) | N/A | 0.96 | 0.03 | N/A | N/A |
| 3 g d−1 | 13.0 (−112, 138) | N/A | |||||
| 2 g d−1 | 9.60 (−140, 121) | N/A | |||||
| Combined | 2.38 (−101, 106) | N/A | 0.96 | 0.03 | N/A | N/A | |
| HI assay Phuket strain | 4 g d−1 | −8.48 (−24.7, 7.71) | N/A | 0.50 | <0.01 | N/A | N/A |
| 3 g d−1 | 2.83 (−12.8, 18.4) | N/A | |||||
| 2 g d−1 | 1.35 (−15.0, 17.7) | N/A | |||||
| Combined | −1.25 (−14.4, 11.9) | N/A | 0.85 | <0.01 | N/A | N/A | |
| Requirementsa | Victoria strain | Phuket strain | |||
|---|---|---|---|---|---|
| Stanol groups (N = 94) | Control group (N = 32) | Stanol groups (N = 76) | Control group (N = 25) | ||
| a Requirements for people ≥60 years according to the CHMP immunological criteria for HI titres of influenza vaccines. Abbreviations: HI = hemagglutination inhibition, CHMP = committee for medicinal products for human use. | |||||
| Geometric mean fold rise (GMFR) | >2.0 | Yes | Yes | Yes | Yes |
| Seroconversion rate (>4-fold increase) | >30% | 41% | 39% | 26% | 28% |
| Seroprotection rate (HI titre > 40) before vaccination | — | 67% | 77% | 33% | 45% |
| Seroprotection rate (HI titre > 40) after vaccination | >60% | 98% | 97% | 96% | 84% |
| Seroprotection after but not before vaccination | - | 31% | 20% | 63% | 39% |
Cytokines, hs-CRP and leucocytes
Changes in serum cytokine (IL-6, IL-8 and TNF-α) and hs-CRP concentrations did not significantly differ between the intervention groups and the control group throughout the study (Table 5). All intervention groups as well as the control group showed a slight increase in TNFα over time, resulting in a significant time effect. Total leukocyte count slightly increased over time (p = 0.05) in all groups, while changes in total and differential leukocyte counts did not differ significantly between the groups (Table 6).| p-Value | |||||||
|---|---|---|---|---|---|---|---|
| Group vs. control group | Estimated marginal difference (95% CI) | Group vs. control group | Group | Time | Group × time | Time V1-vaccination | |
| All models were corrected for gender and baseline concentrations. Abbreviations: 95%CI = 95% confidence interval, time V1-vaccination = time between the start of the study and the influenza vaccination date (this variable differed per participant and therefore was tested in all models), N/A = not applicable, IL = interleukin, TNFα = tumour necrose factor α. If group × time or time V1-vaccination was N/A this variable was not included in the final model because it did not reach significance. If group was not significant, N/A is shown for the group vs. group comparison. Statistically significant if p-value ≤ 0.05. | |||||||
| hs-CRP, mg dL−1 | 4 g d−1 | 0.40 (−1.47, 2.27) | N/A | 0.79 | 0.44 | N/A | N/A |
| 3 g d−1 | 0.70 (−1.14, 2.55) | N/A | |||||
| 2 g d−1 | 0.94 (−1.00, 2.87) | N/A | |||||
| IL-6 | 4 g d−1 | −0.25 (−0.55, 0.27) | N/A | 0.35 | 0.18 | N/A | N/A |
| 3 g d−1 | −0.23 (−0.52, 0.07) | N/A | |||||
| 2 g d−1 | −0.15 (−0.46, 0.16) | N/A | |||||
| IL-8 | 4 g d−1 | 0.66 (−0.40, 1.73) | N/A | 0.60 | 0.14 | N/A | N/A |
| 3 g d−1 | −0.32 (−1.34, 0.71) | N/A | |||||
| 2 g d−1 | −0.48 (−1.60, 0.59) | N/A | |||||
| TNF-α | 4 g d−1 | −0.01 (−0.15, 0.14) | N/A | 0.98 | 0.02 | N/A | N/A |
| 3 g d−1 | −0.02 (−0.15, 0.15) | N/A | |||||
| 2 g d−1 | −0.02 (−0.18, 0.13) | N/A | |||||
| p-Value | |||||||
|---|---|---|---|---|---|---|---|
| Group vs. control group | Estimated marginal difference (95% CI) | Group vs. control group | Group | Time | Group × time | Time V1-vaccination | |
| All models were corrected for gender and baseline concentrations. Abbreviations: 95%CI = 95% confidence interval, time V1-vaccination = time between the start of the study and the influenza vaccination date (this variable differed per participant and therefore was tested in all models), N/A = not applicable. If group × time or time V1-vaccination was N/A this variable was not included in the final model because it did not reach significance. If group was not significant, N/A is shown for the group vs. group comparison. Statistically significant if p-value ≤ 0.05. | |||||||
| Leukocytes, 109/L | 4 g d−1 | 0.22 (−0.15, 0.59) | N/A | 0.67 | 0.05 | N/A | N/A |
| 3 g d−1 | 0.17 (−0.19, 0.54) | N/A | |||||
| 2 g d−1 | 0.14 (−0.24, 0.52) | N/A | |||||
| Neutrophils, % | 4 g d−1 | −0.56 (−2.93, 1.81) | N/A | 0.43 | 0.20 | N/A | N/A |
| 3 g d−1 | −1.53 (−3.91, 0.86) | N/A | |||||
| 2 g d−1 | −1.79 (−4.23, 0.66) | N/A | |||||
| Lymphocytes, % | 4 g d−1 | 0.70 (−1.37, 2.77) | N/A | 0.13 | 0.90 | N/A | N/A |
| 3 g d−1 | 2.20 (0.13, 4.26) | N/A | |||||
| 2 g d−1 | 1.89 (−0.24, 4.03) | N/A | |||||
| Monocytes, % | 4 g d−1 | −0.50 (−1.22, 0.21) | N/A | 0.28 | 0.64 | N/A | N/A |
| 3 g d−1 | −0.29 (−1.00, 0.42) | N/A | |||||
| 2 g d−1 | 0.15 (−0.58, 0.89) | N/A | |||||
| Eosinophils, % | 4 g d−1 | −0.31 (−0.69, 0.06) | N/A | 0.10 | 0.05 | N/A | N/A |
| 3 g d−1 | −0.41 (−0.78, −0.05) | N/A | |||||
| 2 g d−1 | −0.43 (−0.81, −0.04) | N/A | |||||
| Basophils, % | 4 g d−1 | 0.12 (−0.03, 0.26) | N/A | 0.39 | 0.55 | N/A | N/A |
| 3 g d−1 | 0.02 (−0.12, 0.16) | N/A | |||||
| 2 g d−1 | 0.05 (−0.10, 0.20) | N/A | |||||
Serum lipids, lipoproteins and glucose
Serum LDL-C concentrations were significantly decreased in the 4 g d−1 and the 3 g d−1 with respectively 7.8% (−0.27 [−0.42, −0.12] mmol L−1, p < 0.01) and 6.8% (−0.22 [−0.37, −0.07] mmol L−1, p < 0.01) as compared to control. In the 2 g d−1 group serum LDL-C concentrations were lowered with 6.0% (−0.19 [−0.35, −0.03] mmol L−1), but this difference did not reach statistical significance. Changes in serum TC concentrations statistically differed between all intervention groups and the control group (all p ≤ 0.01) with −0.36 [−0.55, −0.17] mmol L−1, −0.24 [−0.43, −0.05] mmol L−1 and −0.26 [−0.46, −0.07] mmol L−1 for 4, 3 and 2 g d−1 respectively. For serum HDL-C concentrations no significant differences in changes between the groups were observed. Furthermore, a significant reduction in serum TAG concentrations for the 4 g d−1 and 2 g d−1 groups (estimated marginal difference compared with the control group 95% CI −0.19 [−0.33, −0.03] mmol L−1; p = 0.02 and −0.21 [−0.37, −0.05] mmol L−1; p < 0.01 respectively) was found (Table 7). Finally, fasting glucose concentrations did not differ between the groups (group p = 0.74).| p-Value | |||||||
|---|---|---|---|---|---|---|---|
| (In mmol L−1) | Group vs. control group | Estimated marginal difference (95% CI) | Group vs. control group | Group | Time | Group × time | Time V1-vaccination |
| All models were corrected for gender and baseline concentrations. Abbreviations: 95%CI = 95% confidence interval, time V1-vaccination = time between the start of the study and the influenza vaccination date (this variable differed per participant and therefore was tested in all models), N/A = not applicable, TC = total cholesterol, LDL = low-density lipoprotein, HDL = high-density lipoprotein, TAG = triacylglycerol. If group × time or time V1-vaccination was N/A this variable was not included in the final model because it did not reach significance. If group was not significant, N/A is shown for the group vs. group comparison. Statistically significant if p-value ≤ 0.05, except for the group vs. group comparison (p ≤ 0.017 is significant). | |||||||
| TC | 4 g d−1 | −0.36 (−0.55, −0.17) | <0.01 | <0.01 | 0.07 | N/A | N/A |
| 3 g d−1 | −0.24 (−0.43, −0.05) | 0.01 | |||||
| 2 g d−1 | −0.26 (−0.46, −0.07) | 0.01 | |||||
| LDL-C | 4 g d−1 | −0.27 (−0.42, −0.12) | <0.01 | <0.01 | 0.81 | N/A | N/A |
| 3 g d−1 | −0.22 (−0.37, −0.07) | <0.01 | |||||
| 2 g d−1 | −0.19 (−0.35, −0.03) | 0.02 | |||||
| HDL-C | 4 g d−1 | 0.00 (−0.07, 0.07) | N/A | 0.99 | 0.01 | N/A | N/A |
| 3 g d−1 | 0.01 (−0.06, 0.07) | N/A | |||||
| 2 g d−1 | −0.00 (−0.07, 0.07) | N/A | |||||
| TAG | 4 g d−1 | −0.19 (−0.33, −0.03) | 0.02 | 0.04 | <0.01 | N/A | N/A |
| 3 g d−1 | −0.11 (−0.26, 0.04) | 0.16 | |||||
| 2 g d−1 | −0.21 (−0.37, −0.05) | <0.01 | |||||
| Glucose | 4 g d−1 | 0.08 (−0.08, 0.24) | N/A | 0.74 | 0.63 | N/A | N/A |
| 3 g d−1 | 0.06 (−0.10, 0.21) | N/A | |||||
| 2 g d−1 | 0.08 (−0.08, 0.25) | N/A | |||||
Anthropometrics and dietary intakes
Weight, BMI, waist circumference, hip circumference, and WHR remained stable over time and did not differ between the groups (SI Table S3). Changes in dietary intakes were small and comparable between intervention groups and the control group, no significant differences were observed (SI Table S4).Discussion
In this randomized placebo-controlled double-blind intervention study, daily consumption of 2, 3 or 4 grams of plant stanols, provided as their fatty-acid esters, did not improve vaccination responses after the annual influenza vaccination in men and women aged 60 years or older. In addition, no differences in changes were observed in total or differential leukocytes counts as well as other immunological parameters. Compliance was excellent as shown by the changes in serum TC-standardized sitostanol and campestanol levels as well as the changes in serum LDL-C in the plant stanol ester groups as compared to control.In previous studies, we have shown that plant stanol ester consumption provided as their fatty-acid esters (4 gram stanols per day) significantly increased antibody production after vaccination in immunocompromised individuals.14,15 In one study, significant increases in serum anti-COVID-19 IgM Spike antibody titres (up to 139%) were shown post-vaccination in adults with overweight or obesity.15 In the second study, significant increases in serum anti-hepatitis-A antibody titres (up to 77%) were shown post-vaccination in obese and overweight adults with allergic asthma.14 Contrary to our hypothesis, in the present study we found no significant effects of plant stanols on IgM or IgG anti-influenza antibody titres post-vaccination. Although the observed increase in the IgM antibody titre in the 4 gram per day group as compared to the change in the control group was not significant, the effect size of the increased IgM concentrations was 81% at visit 4. This effect size is slightly higher then a priori anticipated during the design of the study and in line with observed effect sizes in previous vaccination studies using 4 grams per day of plant stanols.14,15 This raises the question of how this study differed from the two previous studies, which had even lower numbers of participants, but effect sizes higher than anticipated that did reach statistical significance. Compared to the previous studies,14,15 baseline antibody titres at the start of this study were higher in the present study and varied more between the participants. Also, responses in antibody titres after the vaccination varied more widely between subjects, as evident from the larger between-subject variability. A major difference with our earlier studies is that in those studies a primary immune response against a foreign pathogen was evaluated, which is most likely not the case for the majority of the participants in the present study receiving the influenza vaccination. In the Netherlands, participants of 60+ years of age can annually receive the influenza vaccination and therefore our participants probably already encountered a different number influenza infections and vaccinations during the past years. Previous studies have demonstrated that multiple vaccinations and earlier influenza infections can both lead to higher pre-vaccination antibody titres and reduced vaccination responses, resulting in larger variation between individuals. This results in a wider range of responses and consequently a higher SD.1,19–22 This different increase in variation may partially explain why the observed change of 81% in the IgM values did not reach statistical significance in this study.
In addition to IgM and IgG antibodies titres, vaccine-specific HI antibody titres were also measured. However, in line with the IgM and IgG data, no significant effects of plant stanol ester intake on HI antibody titres were observed against the A/Victoria/2570/2019 (H1N1)pdm09-like virus or the B/Phuket/3073/2013-like virus strains. The Victoria strain is an influenza A H1N1 virus, while the Phuket strain is an influenza B virus. Although both strains were present in the same amounts in the vaccines (15 µg HA per strain), HI antibody titres against the Victoria strain were almost double as compared to those against the Phuket strain. These strain specific effects agree with the results of a meta-analysis showing statistically significant increases in HI antibody titres against the H1N1 strain, but not against H3N2 and B strains.23 Although it is difficult to explain these strain-specific differences, it is known that previous vaccinations and infections with closely related influenza strains can lead to cross-reactivity and therefore can affect pre- and post-vaccination antibody titres.1,21 In line with our findings for IgM and IgG, other studies have shown that repeated vaccinations with the same strains not only resulted in higher pre-vaccination HI titres but also less pronounced increases in HI titres post-vaccination.4,19,24–27 One explanation could be that high pre-vaccination titres result in reduced B cell boosting responses, which translates into less pronounced increased antibody titres post-vaccination.19,28,29 We therefore speculate that, in line with the explanation for IgM, the difference in HI titres did not translate into statistical significance, due to the high variation in responses between participants.
In addition to evaluating antibodies using the HI assay, we used the seroprotection and seroconversion rates to compare our results to other studies. In all groups and for both strains, seroprotection was reached since HI titres were ≥40 in more than 60% of the individuals. In addition, our study population showed seroconversion rates ranging between 28–41% while populations older adults normally achieves rates between 10–30%. This suggests that the older adults in our study were immunologically relatively healthy as compared to previous studies. Moreover, reviews showed that older adults failed to reach protective HI antibody titres after vaccination, which also contrasts our observations.6,30,31 In other words, the relatively younger and healthier population in our study could have resulted in overall higher antibody responses, leaving less room for additional improvement due to the intervention.
Aging, and the consequent immunosenescent state, also translates in altered cytokine production, characterised by elevated concentrations of the pro-inflammatory cytokines IL-6, IL-8 and TNF-α.32,33 However, no effects of plant stanol ester intake on plasma concentrations of these cytokines were found. This is in line with our earlier study evaluating effects of plant stanols on the COVID-19 vaccination response in people with overweight/obesity, in which also no effects on circulating cytokine concentrations were observed.15
In the Netherlands, all individuals aged 60 years and older are invited to receive an influenza vaccination. This study therefore included a diverse population with minimal exclusion criteria reflecting a real-world context. While this approach strengthens the generalizability of our findings, it can also be viewed as a limitation due to inherent variability in the population, as increased age, frailty and BMI can decrease the immune response even more. This variability may have made it more challenging to detect significant changes. Other strengths of this study were the excellent compliance, as well as the unchanged anthropometrics and food intake during the study. However, there are also limitations that need to be addressed. Most important, we were not able to recruit all desired participants which may have resulted in an underpowered study and therefore verifying the results is needed.
Vaccination models are considered as the gold standard for determining the effects of foods and food components on immune function.7 However, we propose that this gold standard can be made more specific, hence multiple studies have been performed on influenza vaccination responses and elicit different results, not only between studies but even between different strains within the same vaccine.1,19–21,23 The vaccination response already seem to depend on age, serum TC and LDL-C concentrations and other interpersonal differences.15 However, the vaccination response after the influenza vaccination is also dependent on strain differences, the number of previous infections, previous number of vaccinations and the timing of these vaccinations. To standardize investigating immune responses as well as possible, we would like to propose that the gold standard should be analysing a primary vaccination response.
Conclusions
To conclude, daily consumption of 2, 3 or 4 grams of plant stanols provided as their fatty-acid esters did not significantly improve vaccination responses following the annual influenza vaccination in men and women aged 60 years or older. Future studies should aim to examine the effects of lower intakes of plant stanols (<4 g d−1) using a similar study design, but with a specific focus on the primary vaccination response or in a more specific, less healthy, study population.Author contributions
Willem Zwaan: conceptualization, investigation, formal analysis, writing – original draft, Lieve van Brakel: conceptualization, investigation, writing – review & editing, Ronald P. Mensink: conceptualization, formal analysis, writing – original draft, supervision, Jogchum Plat: conceptualization, formal analysis, writing – original draft, supervision.Conflicts of interest
JP has financial support from Raisio Nutrition Ltd. All other authors report no conflict of interest. All other authors have no conflicts to declare.Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.Supplementary information (SI) is available. See DOI: https://doi.org/10.1039/d5fo03652f.
Acknowledgements
This work was supported by Raisio Nutrition Ltd, who also provided the study products but was not involved in the design, implementation, analysis, and interpretation of the study. The authors thank all volunteers for participating in this study, B. Winkens for statistical advice, A. Lasfar and E. Stoop for practical support throughout this study, M. C. J. M. Konings for the biochemical analyses.References
- C. Laue, Y. Stevens, M. van Erp, E. Papazova, E. Soeth, A. Pannenbeckers, E. Stolte, R. Böhm, S. L. Gall, X. Falourd, S. Ballance, S. H. Knutsen, I. Pinheiro, S. Possemiers, P. M. Ryan, R. P. Ross, C. Stanton, J. M. Wells, S. V. Werf, J. J. Mes and J. Schrezenmeir, Adjuvant Effect of Orally Applied Preparations Containing Non-Digestible Polysaccharides on Influenza Vaccination in Healthy Seniors: A Double-Blind, Randomised, Controlled Pilot Trial, Nutrients, 2021, 13(8), 2683 CrossRef PubMed.
- A. L. Gruver, L. L. Hudson and G. D. Sempowski, Immunosenescence of ageing, J. Pathol., 2007, 211(2), 144–156 CrossRef PubMed.
- D. Loukov, A. Naidoo and D. M. Bowdish, Immunosenescence: implications for vaccination programs in the elderly, Vaccine:Dev. Ther., 2015, 17–29 Search PubMed.
- L. Hansen, F. Zhou, H. Amdam, M. C. Trieu and R. J. Cox, Repeated Influenza Vaccination Boosts and Maintains H1N1pdm09 Neuraminidase Antibody Titers, Front. Immunol., 2021, 12, 748264 CrossRef.
- A. D. Iuliano, K. M. Roguski, H. H. Chang, D. J. Muscatello, R. Palekar, S. Tempia, C. Cohen, J. M. Gran, D. Schanzer, B. J. Cowling, P. Wu, J. Kyncl, L. W. Ang, M. Park, M. Redlberger-Fritz, H. Yu, L. Espenhain, A. Krishnan, G. Emukule, L. van Asten, S. Pereira da Silva, S. Aungkulanon, U. Buchholz, M. A. Widdowson and J. S. Bresee, Global Seasonal Influenza-associated Mortality Collaborator Network. Estimates of global seasonal influenza-associated respiratory mortality: a modelling study, Lancet, 2018, 391(10127), 1285–1300 CrossRef PubMed.
- K. Goodwin, C. Viboud and L. Simonsen, Antibody response to influenza vaccination in the elderly: a quantitative review, Vaccine, 2006, 24(8), 1159–1169 CrossRef PubMed.
- R. Albers, J. M. Antoine, R. Bourdet-Sicard, P. C. Calder, M. Gleeson, B. Lesourd, S. Samartín, I. R. Sanderson, J. Van Loo, F. W. Vas Dias and B. Watzl, Markers to measure immunomodulation in human nutrition intervention studies, Br. J. Nutr., 2005, 94(3), 452–481 CrossRef PubMed.
- EFSA Panel on Dietetic Products, Allergies, Guidance on the scientific requirements for health claims related to gut and immune function, EFSA J., 2011, 9(4), 1984 CrossRef.
- EFSA Panel on Dietetic Products, Allergies, Guidance on the scientific requirements for health claims related to the immune system, the gastrointestinal tract and defence against pathogenic microorganisms, EFSA J., 2016, 14(1), 4369 Search PubMed.
- P. C. Calder, E. F. Ortega, S. N. Meydani, Y. Adkins, C. B. Stephensen, B. Thompson and H. Zwickey, Nutrition, Immunosenescence, and Infectious Disease: An Overview of the Scientific Evidence on Micronutrients and on Modulation of the Gut Microbiota, Adv. Nutr., 2022, 13(5), S1–S26 CrossRef PubMed.
- C. J. Huang, M. C. Zourdos, E. Jo and M. J. Ormsbee, Influence of physical activity and nutrition on obesity-related immune function, Sci. World J., 2013, 2013, 752071 CrossRef PubMed.
- M. Eggersdorfer, M. M. Berger, P. C. Calder, A. F. Gombart, E. Ho, A. Laviano and S. N. Meydani, Perspective: Role of Micronutrients and Omega-3 Long-Chain Polyunsaturated Fatty Acids for Immune Outcomes of Relevance to Infections in Older Adults-A Narrative Review and Call for Action, Adv. Nutr., 2022, 13(5), 1415–1430 CrossRef.
- P. C. Calder, M. M. Berger, A. F. Gombart, G. A. McComsey, A. R. Martineau and M. Eggersdorfer, Micronutrients to Support Vaccine Immunogenicity and Efficacy, Vaccines, 2022, 10(4), 568 CrossRef.
- F. Brüll, E. De Smet, R. P. Mensink, A. Vreugdenhil, A. Kerksiek, D. Lütjohann, G. Wesseling and J. Plat, Dietary plant stanol ester consumption improves immune function in asthma patients: results of a randomized, double-blind clinical trial, Am. J. Clin. Nutr., 2016, 103(2), 444–453 CrossRef PubMed.
- L. van Brakel, R. P. Mensink, D. Lütjohann and J. Plat, Plant stanol consumption increases anti-COVID-19 antibody responses, independent of changes in serum cholesterol concentrations: a randomized controlled trial, Am. J. Clin. Nutr., 2024, 119(4), 969–980 CrossRef CAS PubMed.
- D. S. Mackay, P. J. Jones, S. B. Myrie, J. Plat and D. Lütjohann, Methodological considerations for the harmonization of non-cholesterol sterol bio-analysis, J. Chromatogr. B:Anal. Technol. Biomed. Life Sci., 2014, 957, 116–122 CrossRef CAS.
- W. T. Friedewald, R. I. Levy and D. S. Fredrickson, Estimation of the concentration of low-density lipoprotein cholesterol in plasma, without use of the preparative ultracentrifuge, Clin. Chem., 1972, 18(6), 499–502 CrossRef CAS . PMID: 4337382.
- T. A. Miettinen, R. S. Tilvis and Y. A. Kesäniemi, Serum plant sterols and cholesterol precursors reflect cholesterol absorption and synthesis in volunteers of a randomly selected male population, Am. J. Epidemiol., 1990, 131(1), 20–31 CrossRef CAS PubMed.
- M. Sanyal, T. H. Holmes, H. T. Maecker, R. A. Albrecht, C. L. Dekker, X. S. He and H. B. Greenberg, Diminished B-Cell Response After Repeat Influenza Vaccination, J. Infect. Dis., 2019, 219(10), 1586–1595, DOI:10.1093/infdis/jiy685 , Erratum in: J Infect Dis. 2019 Apr 19;219(10):1683.
- Y. Hirota, M. Kaji, S. Ide, S. Goto and T. Oka, The hemagglutination inhibition antibody responses to an inactivated influenza vaccine among healthy adults: with special reference to the prevaccination antibody and its interaction with age, Vaccine, 1996, 14(17–18), 1597–1602 CrossRef CAS.
- M. C. Trieu, F. Zhou, S. L. Lartey, S. Sridhar, S. Mjaaland and R. J. Cox, Augmented CD4+ T-cell and humoral responses after repeated annual influenza vaccination with the same vaccine component A/H1N1pdm09 over 5 years, npj Vaccines, 2018, 3, 37 CrossRef PubMed.
- G. Mai, C. Zhang, C. Lan, J. Zhang, Y. Wang, K. Tang, J. Tang, J. Zeng, Y. Chen, P. Cheng, S. Liu, H. Long, Q. Wen, A. Li, X. Liu, R. Zhang, S. Xu, L. Liu, Y. Niu, L. Yang, Y. Wang, D. Yin, C. Sun, Y. Q. Chen, W. Shen, Z. Zhang and X. Du, Characterizing the dynamics of BCR repertoire from repeated influenza vaccination, Emerging Microbes Infect., 2023, 12(2), 2245931 CrossRef.
- T. L. Yeh, P. C. Shih, S. J. Liu, C. H. Lin, J. M. Liu, W. T. Lei and C. Y. Lin, The influence of prebiotic or probiotic supplementation on antibody titers after influenza vaccination: a systematic review and meta-analysis of randomized controlled trials, Drug Des., Dev. Ther., 2018, 12, 217–230 CrossRef CAS PubMed.
- R. Pyhälä, V. Kumpulainen, S. Alanko and T. Forsten, HI antibody kinetics in adult volunteers immunized repeatedly with inactivated trivalent influenza vaccine in 1990–1992, Vaccine, 1994, 12(10), 947–952 CrossRef.
- D. Hobson, F. A. Baker and R. L. Curry, Effect of influenza vaccines in stimulating antibody in volunteers with prior immunity, Lancet, 1973, 2(7821), 155–156 CrossRef CAS.
- S. Sasaki, X. S. He, T. H. Holmes, C. L. Dekker, G. W. Kemble, A. M. Arvin and H. B. Greenberg, Influence of prior influenza vaccination on antibody and B-cell responses, PLoS One, 2008, 3(8), e2975 CrossRef.
- F. Wen, J. Guo and S. Huang, A meta-analysis identified genes responsible for distinct immune responses to trivalent inactivated and live attenuated influenza vaccines, J. Cell Physiol., 2019, 234(4), 5196–5202 CrossRef CAS.
- A. H. Ellebedy, R. Nachbagauer, K. J. L. Jackson, Y. N. Dai, J. Han, W. B. Alsoussi, C. W. Davis, D. Stadlbauer, N. Rouphael, V. Chromikova, M. McCausland, C. Y. Chang, M. Cortese, M. Bower, C. Chennareddy, A. J. Schmitz, V. I. Zarnitsyna, L. Lai, A. Rajabhathor, C. Kazemian, R. Antia, M. J. Mulligan, A. B. Ward, D. H. Fremont, S. D. Boyd, B. Pulendran, F. Krammer and R. Ahmed, Adjuvanted H5N1 influenza vaccine enhances both cross-reactive memory B cell and strain-specific naive B cell responses in humans, Proc. Natl. Acad. Sci. U. S. A., 2020, 117(30), 17957–17964 CrossRef CAS.
- S. F. Andrews, K. Kaur, N. T. Pauli, M. Huang, Y. Huang and P. C. Wilson, High preexisting serological antibody levels correlate with diversification of the influenza vaccine response, J. Virol., 2015, 89(6), 3308–3317 CrossRef CAS PubMed.
- R. B. Fulton and S. M. Varga, Effects of aging on the adaptive immune response to respiratory virus infections, Aging Health, 2009, 5(6), 775 CrossRef PubMed.
- S. N. Crooke, I. G. Ovsyannikova, G. A. Poland and R. B. Kennedy, Immunosenescence and human vaccine immune responses, Immun. Ageing, 2019, 16, 25 CrossRef PubMed.
- C. Hieber, S. Grabbe and M. Bros, Counteracting Immunosenescence-Which Therapeutic Strategies Are Promising?, Biomolecules, 2023, 13(7), 1085 CrossRef CAS PubMed.
- B. J. Andonian, J. A. Hippensteel, K. Abuabara, E. M. Boyle, J. F. Colbert, M. J. Devinney, A. S. Faye, B. Kochar, J. Lee, R. Litke, D. Nair, S. E. Sattui, A. Sheshadri, A. N. Sherman, N. Singh, Y. Zhang and S. C. LaHue, Inflammation and aging-related disease: A transdisciplinary inflammaging framework, Geroscience, 2025, 47(1), 515–542 CrossRef PubMed.
| This journal is © The Royal Society of Chemistry 2025 |