Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Standardized fenugreek seed extract alleviates menopausal sleep disturbances via GABAA, melatonin, and adenosine receptors in ovariectomized (OVX) rodents
Hyowon
Lee
 a,
Hoon
Kim
a,
Hoon
Kim
 b,
Hyung Joo
Suh
b,
Hyung Joo
Suh
 *a and
Hyeon-Son
Choi
*a and
Hyeon-Son
Choi
 *c
*c
aDepartment of Integrated Biomedical and Life Science, Graduate School, Korea, University, Seoul 02841, Republic of Korea. E-mail: suh1960@korea.ac.kr; hyowon24@korea.ac.kr; Tel: +82-2-3290-5639
bDepartment of Food and Nutrition, Anyang University, Samdeok-ro 37beon-gil, Manan-gu, Anyang-si, Gyeonggi-do 14028, Republic of Korea. E-mail: hkim@anyang.ac.kr
cDepartment of Food Nutrition, Sangmyung University, Seoul 03016, Republic of Korea. E-mail: hsc1970@smu.ac.kr; Fax: +82-2-2287-0104; Tel: +82-2-2287-6155
First published on 5th November 2025
Abstract
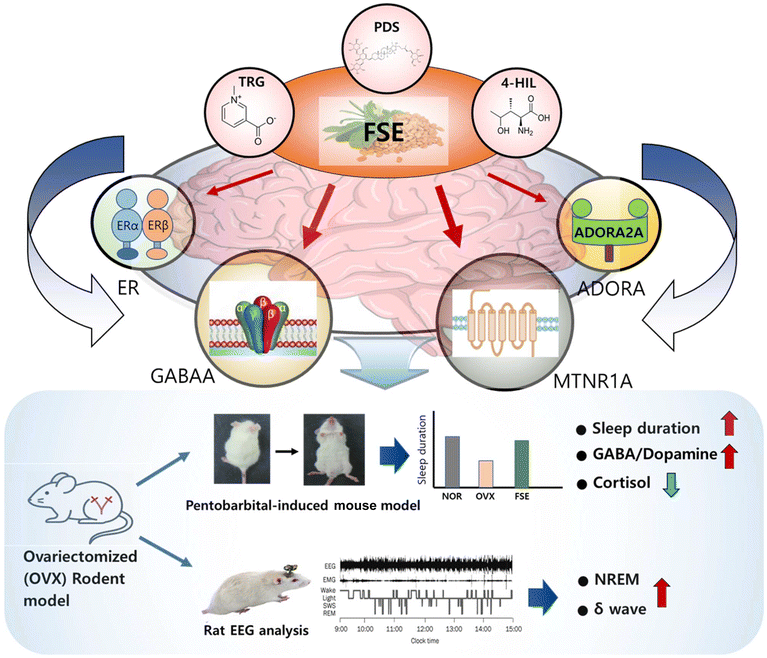
Fenugreek seed (Trigonella foenum-graecum L.), a dietary component traditionally used in food and herbal medicine, is known for its wide range of health-promoting properties. However, its effects on menopause-associated sleep disturbances remain largely unexplored. This study investigated the potential of a standardized fenugreek seed extract (FSE), enriched in trigonelline (1.72%), protodioscin (6.17%), and 4-hydroxy-L-isoleucine (2.55%), to alleviate sleep impairments in ovariectomized (OVX) mice, a model of menopause-related symptoms. FSE administration significantly improved OVX-induced sleep disturbances by increasing the sleep duration in a pentobarbital-induced hypnotic test. Mechanistic investigations showed that FSE activated GABAA and melatonin receptor 1A (MTNR1A), confirmed by antagonist treatment with picrotoxin, flumazenil, and luzindole. Electroencephalogram (EEG) analysis revealed enhanced delta activity during NREM sleep, indicating improved sleep quality. FSE restored neurochemical and hormonal balance by elevating GABA and dopamine levels while reducing cortisol. It also increased the expression of estrogen receptors ERα, ERβ, and GPR30, without altering systemic estradiol levels. Furthermore, the adenosine A2A receptor (ADORA2A) was significantly elevated. Administration of trigonelline, protodioscin, and 4-hydroxy-L-isoleucine, either individually or in combination, replicated the sleep-promoting effects of FSE, supporting their role in the overall bioactivity. This study highlights FSE and its bioactive compounds as promising functional food ingredients for improving menopausal sleep disorders by modulating GABAergic, melatonergic, and adenosinergic signaling pathways, as well as estrogen receptor expression and neurohormonal homeostasis.
Introduction
Sleep is a vital biological process essential for physical health, cognitive function, emotional well-being, and immune defense.1 However, modern lifestyles, characterized by stress, irregular schedules, and screen exposure, contribute to a growing prevalence of sleep disorders, linked to cardiovascular disease, obesity, diabetes, and mental health disorders such as anxiety and depression.2,3 Poor sleep quality also accelerates biological aging and increases mortality risk.4 Sleep disturbances arise from multiple factors, including poor sleep hygiene, work-related stress, and medical conditions like insomnia and sleep apnea.5,6Current management of sleep disorders typically involves lifestyle changes, cognitive-behavioral therapy, and pharmacological interventions.7,8 Sedative-hypnotics, including benzodiazepines, non-benzodiazepine receptor agonists, and melatonin supplements, are commonly prescribed to promote sleep onset and maintenance.7,9 However, these drugs are associated with side effects such as dependency, daytime drowsiness, dizziness, memory impairment, and an increased fall risk, particularly in older adults. Long-term use can also lead to tolerance and withdrawal symptoms, potentially worsening sleep problems over time.10 These limitations have spurred interest in food-derived bioactive components as safer, evidence-based natural alternatives to promote sleep health, particularly through natural ingredients with biochemical potential.
While sleep disorders affect diverse populations, menopausal women are particularly vulnerable due to hormonal changes. Menopause, occurring between ages 45 and 55 years, is characterized by a decline in estrogen and progesterone, leading to hot flashes, mood swings, fatigue, cognitive impairments, and sleep disturbances.11,12 These sleep disruptions, affecting up to 50% of menopausal women, are driven by hormonal fluctuations, night sweats and circadian dysregulation, significantly contributing to daytime fatigue and reduced quality of life.13,14 Psychological stressors, including anxiety, further exacerbate these sleep problems, highlighting the need for effective, low-risk interventions.
Fenugreek (Trigonella foenum-graecum), a medicinal herb native to the Mediterranean and widely cultivated in Asia and Europe, is commonly used as a food ingredient, with its seeds serving as a spice to enhance flavor and nutrition in various cuisines.15 It has been traditionally used for a wide range of health benefits, including its antioxidant, anti-inflammatory, digestive support, glycemic control, and hormonal balancing properties.16–18 Fenugreek seed extract (FSE), rich in bioactive constituents, such as saponins, flavonoids, and alkaloids, trigonelline, protodioscin, and 4-hydroxy-L-isoleucine,18,19 is hypothesized to alleviate menopause-related sleep through its endocrine-modulating properties and potential to influence neurotransmitter systems. However, despite these promising traditional uses and proposed mechanisms, FSE's efficacy and specific underlying mechanisms for sleep improvement remain largely unexplored.
To address this knowledge gap, our study investigates the potential of FSE and its bioactive components in alleviating sleep disorders induced by estrogen deficiency, using an ovariectomized (OVX) rodent model. We further aim to elucidate the underlying mechanisms, focusing on GABAergic, melatonergic, and adenosinergic pathways critical to sleep regulation. By exploring FSE's role as a functional food ingredient, this research highlights its significance in food chemistry and health applications, addressing the growing demand for safe, natural interventions for menopause-related sleep disorders.
Materials and methods
Preparation of FSE
The seeds of Trigonella foenum-graecum L. were obtained from Juyeong NS Co., Ltd (Seoul, Korea). The fenugreek seed extract (FSE) used in this study was commercially prepared by Juyeong NS Co., Ltd. According to the manufacturer's procedure, the seeds were flaked or powdered using a pulverizer to achieve a particle size of 0.8–1.2 mm. The powdered seeds were then subjected to commercial-scale extraction using 70% ethanol at 30–35 °C for 12 to 15 h. The resulting mixture was then subjected to evaporation and concentration. The extract was purified through fabric filtration with a 2 µm mesh, followed by spray drying. The dried extract was then blended with 0.5% (w/w) silica. The final product obtained was fenugreek seed extract (FSE). FSE was standardized to contain not less than 6.0% protodioscin, 2.5% 4-hydroxy-L-isoleucine, and 1.7% trigonelline (w/w) based on HPLC analysis across three independent batches (see the section “HPLC analysis of FSE-derived compounds”). For experimental use, FSE powder was dissolved in an appropriate vehicle prior to administration to animals.Chemicals and materials
Pentobarbital (Entobar) was purchased from Hanlim Pharm. Co. Ltd (Yongin, Korea). The GABAA receptor antagonists—bicuculline (BIC), flumazenil (FMZ), and picrotoxin (PIX)—were obtained from Sigma-Aldrich (St Louis, MO, USA). The melatonin antagonists luzindole (LUZ) and 4-phenyl-1-(4-phenylbutyl) piperidine (4-P-PDOT, 4PP) were also sourced from Sigma-Aldrich. Additionally, trigonelline hydrochloride (TRG), 4-hydroxy-L-isoleucine (4-HIL), protodioscin (PDS), and dimethyl sulfoxide (DMSO) were acquired from Sigma-Aldrich.Animal maintenance and experimental design
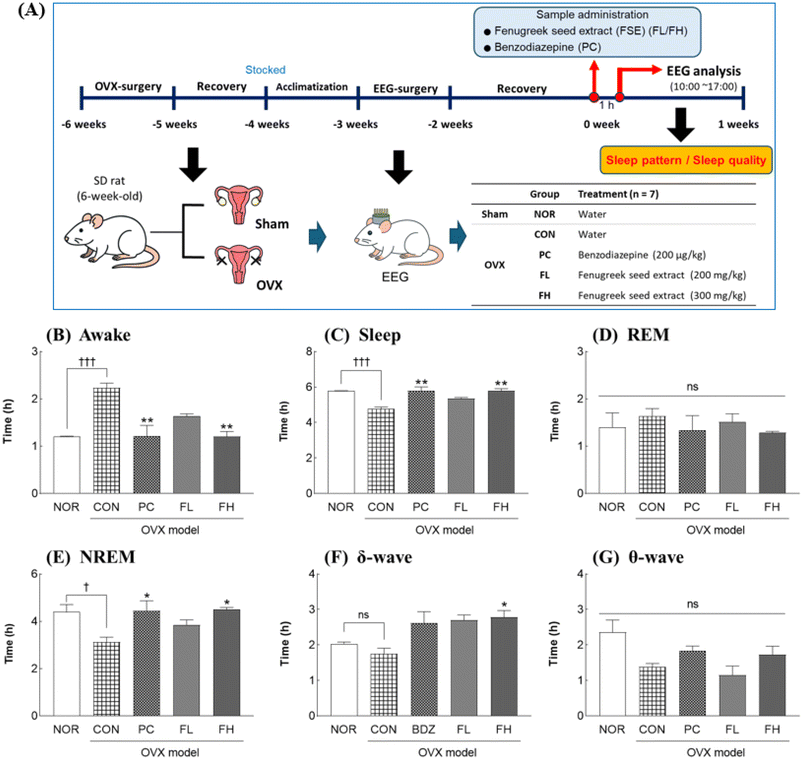
Female ICR mice (4 weeks old, 18–20 g; Orient Bio, Seongnam, Korea) were acclimatized for one week before undergoing ovariectomy (OVX) or sham surgery. After surgery, the mice were allowed a 1-week recovery period, followed by a 4-week post-OVX period during which serum estrogen levels were measured weekly to confirm the estrogen-deficient status. Prior to behavioral testing, mice were fasted for 24 h. Oral administration of FSE or the vehicle was then performed, and 40 minutes later, pentobarbital (42 mg kg−1, i.p.) was administered to assess sleep latency and duration. The mice were randomly assigned to treatment groups (n = 7 per group). The sham-operated groups included: normal control (NOR, vehicle only), positive control (PC-sham, benzodiazepine 200 μg kg−1), and FSE high-dose (FH-sham, 300 mg kg−1). The OVX groups included: OVX control (CON-OVX, vehicle), OVX positive control (PC-OVX, benzodiazepine 200 μg kg−1), FSE low-dose (FL-OVX, 200 mg kg−1), and FSE high-dose (FH-OVX, 300 mg kg−1) (Fig. 1). Female SD rats (6 weeks old, 160–180 g; Biolink, Eumseong, Korea) underwent ovariectomy (OVX) or sham surgery and were allowed 1 week of post-OVX recovery. Following this, the rats experienced an additional 1-week acclimation period prior to EEG transmitter implantation (surgical electrode implantation). After implantation, the rats were allowed 1 week for post-surgical recovery, followed by 2 weeks of additional recovery before initiation of oral FSE or vehicle administration. EEG recordings commenced 1 hour after sample administration, conducted 7 h per day (10:00–17:00) for 6 consecutive days. SD rats were randomly assigned to five groups (n = 7 per group). These included a normal (NOR) sham-operated control (vehicle, water); an OVX control (CON) group of ovariectomized (OVX) model rats (vehicle, water); a positive control (PC) group of OVX model rats (benzodiazepine, 200 µg kg−1); an FSE low-dose (FL) group of OVX model rats (FSE, 200 mg kg−1); and an FSE high-dose (FH) group of OVX model rats (FSE, 300 mg kg−1).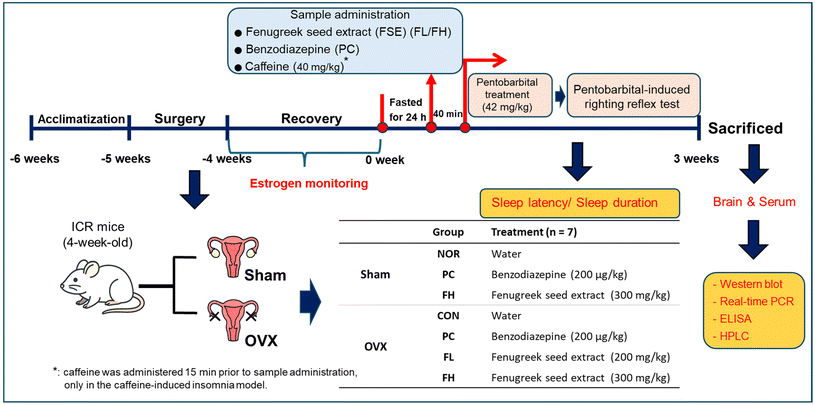 | ||
| Fig. 1 Experimental procedure for the pentobarbital-induced sleep behavior test in the OVX mouse model. | ||
All animals were housed in a controlled environment (21 ± 1 °C, 50%–55% humidity, 12 h light/dark cycle) with ad libitum access to food (Feed Lab Korea, Guri, Korea) and water. All animal experiments were conducted in accordance with the guidelines approved by the Institutional Animal Care and Use Committee of Korea University (KUIACUC-2024-0025).
Ovariectomy and confirmation
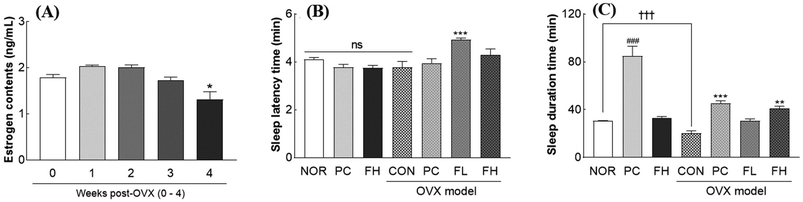
Ovariectomy was performed using a bilateral dorsolateral incision under aseptic conditions, as previously described20 with minor modifications. Briefly, the mice were anesthetized with isoflurane (3% for induction, 1.5–2% for maintenance) and their lateral flank shaved. A small incision was made on each side to expose the ovaries, which were carefully excised after ligating the underlying vasculature. The incision sites were then sutured, and tramadol (20 mg kg−1, subcutaneously) was administered for pain relief. After surgery, the mice received acetaminophen-supplemented water (0.47 mg mL−1) for three days and monitored for distress. To determine the appropriate time point for molecular analyses, serum estrogen levels were measured weekly post-OVX (Fig. 2). As shown, significant reductions in estrogen were observed from week 4. The mice that did not exhibit a sufficient reduction in estrogen or failed to enter sleep within 15 minutes after pentobarbital injection were excluded from further analyses. After applying these criteria, the final sample size for each group was n = 7, with 1–2 mice excluded per group. Therefore, all subsequent molecular and biochemical analyses, including brain tissue collection for RNA and protein assays, were conducted at this 4-week time point to ensure that measurements reflected established estrogen-deficient conditions.Evaluation of sleep behaviors
Antagonist treatment in pentobarbital-induced sleep test
The study utilized GABAA receptor antagonists—picrotoxin (PIX, 4 mg kg−1), bicuculline (BIC, 6 mg kg−1), and flumazenil (FMZ, 10 mg kg−1)—and melatonin receptor antagonists—luzindole (LUZ, 30 mg kg−1) and 4-PPDOT (4PP, 10 mg kg−1). All antagonists were prepared in corn oil containing 5% DMSO. Each antagonist or its corresponding vehicle (corn oil with 5% DMSO) was administered intraperitoneally to ICR mice. Twenty minutes later, FSE (300 mg kg−1) was given orally. The remainder of the experiment followed the same conditions as those of the standard pentobarbital-induced sleep test.EEG analysis in SD rats
Electrodes and EEG transmitters (Emka Technologies, Paris, France) were surgically implanted onto the skull surface of SD rats.21 Following a one-week recovery, EEG measurements were conducted for 7 h per session (10:00–17:00) for 6 d, with signals collected at a 500 Hz sampling rate. Spectrum data were analyzed with 1 Hz spacing using the following standard frequency bands: δ (0.5–4 Hz), θ (4–8 Hz), α (8–12 Hz), β (12–30 Hz), and γ (30–60 Hz). Sleep structure and parameters (wakefulness, total sleep time, NREM, REM, theta, and delta waves) were evaluated using a fast Fourier transform algorithm implemented in ECG Auto 3 software (version 3.3, Emka Technologies, Paris, France).Gene expression analysis–real-time PCR
After 4 weeks of oral FSE administration (5 times per week), whole brain tissues were collected from sacrificed ICR mice. RNA was extracted from 50 mg of homogenized tissue in 1 mL of TRIzol (Invitrogen, Carlsbad, CA, USA) using the TissueLyser II (Qiagen, Hilden, Germany), following the manufacturer's instructions (Thermo Fisher Scientific, Waltham, MA, USA) which included genomic DNA removal. RNA samples were quantified, and only those with OD260/OD280 ratios of 1.8–2.0 (INNO-M, LTek, Seongnam, Republic of Korea) were used for subsequent quantitative real-time PCR (qRT-PCR) analysis. Complementary DNA (cDNA) was synthesized using a reverse transcription-polymerase kit (iNtRON Biotechnology, Seongnam, Korea). qRT-PCR was performed using the SYBR Green system (Applied Biosystems, CA, USA) on an AriaMx system (Agilent Technologies, Santa Clara, CA, USA). Primer sequences are detailed in Table S1.Western blot analysis
Protein was extracted from brain tissue (100 mg) using RIPA buffer supplemented with protease and phosphatase inhibitors (PPC1010, Sigma-Aldrich). Following centrifugation (10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000g for 10 min at 4 °C), protein content was quantified via BCA assay (BCA1, Sigma-Aldrich). For SDS-PAGE, 50 μg of denatured protein per sample was separated on a 10% polyacrylamide gel (100 V, for 90 min) and then transferred to an Immobilon-P PVDF membrane (Millipore, Burlington, MA, USA). The membrane was blocked with 5% BSA for 1 h and incubated overnight at 4 °C with the primary antibody (1
000g for 10 min at 4 °C), protein content was quantified via BCA assay (BCA1, Sigma-Aldrich). For SDS-PAGE, 50 μg of denatured protein per sample was separated on a 10% polyacrylamide gel (100 V, for 90 min) and then transferred to an Immobilon-P PVDF membrane (Millipore, Burlington, MA, USA). The membrane was blocked with 5% BSA for 1 h and incubated overnight at 4 °C with the primary antibody (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1000). After three 10 min washes with TBST, the membrane was incubated with an HRP-conjugated secondary antibody (goat anti-rabbit IgG-HRP, 1
1000). After three 10 min washes with TBST, the membrane was incubated with an HRP-conjugated secondary antibody (goat anti-rabbit IgG-HRP, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5000, #35401, Cell Signaling Technology) at room temperature for 2 h. After three additional TBST washes, protein bands were visualized using an ECL detection system (32106, Thermo Fisher Scientific) and an imaging system (the Alliance Q9 system, UVITECH, Warwickshire, UK). The antibodies used are presented in Table S2.
5000, #35401, Cell Signaling Technology) at room temperature for 2 h. After three additional TBST washes, protein bands were visualized using an ECL detection system (32106, Thermo Fisher Scientific) and an imaging system (the Alliance Q9 system, UVITECH, Warwickshire, UK). The antibodies used are presented in Table S2.
Measurement of GABA, dopamine, cortisol, and estrogen levels
For GABA and dopamine analyses, brain tissue (50 mg) was homogenized in 700 μL of 40 mM Tris-HCl (pH 7.2) using a TissueLyser II (Qiagen). The supernatant was collected by centrifugation (10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000g for 15 min, 4 °C). GABA and dopamine levels were analyzed using HPLC with fluorescence detection on a Waters AccQ-Tag column (Waters, Milford, MA, USA) and an HPLC system with UV detection on a YMC-Pack Pro C18 column, respectively, as previously described21,22 (Fig. S1). After 4 weeks of oral administration, blood was collected from sacrificed mice via cardiac puncture into serum separator tubes, clotted at room temperature for 30 min. Serum was obtained by centrifugation (3000g, 20 min, 4 °C). Cortisol and estrogen levels were measured using a cortisol ELISA kit (Abcam, Cambridge, UK, ab108665) and an estrogen ELISA kit (Assay Genie, Dublin, Ireland, UNFI0011), respectively, following the manufacturer's instructions. Absorbance was measured at 450 nm using a microplate reader (INNO M, LTek).
000g for 15 min, 4 °C). GABA and dopamine levels were analyzed using HPLC with fluorescence detection on a Waters AccQ-Tag column (Waters, Milford, MA, USA) and an HPLC system with UV detection on a YMC-Pack Pro C18 column, respectively, as previously described21,22 (Fig. S1). After 4 weeks of oral administration, blood was collected from sacrificed mice via cardiac puncture into serum separator tubes, clotted at room temperature for 30 min. Serum was obtained by centrifugation (3000g, 20 min, 4 °C). Cortisol and estrogen levels were measured using a cortisol ELISA kit (Abcam, Cambridge, UK, ab108665) and an estrogen ELISA kit (Assay Genie, Dublin, Ireland, UNFI0011), respectively, following the manufacturer's instructions. Absorbance was measured at 450 nm using a microplate reader (INNO M, LTek).
HPLC analysis of FSE-derived compounds
For the HPLC analysis, a 150 mg fenugreek seed extract (FSE) sample was dissolved in distilled water, sonicated for 30 min, cooled and then brought to a final volume of 50 mL. A test solution was subsequently prepared by mixing 80 μL of this solution with 400 μL of borate buffer (pH 8.5) for 30 s, followed by the addition of 80 μL of OPA solution. The analysis was conducted using an Alliance 1260 Infinity II HPLC system (Waters, Milford, MA, USA) equipped with an autosampler, a quaternary pump, and a UV detector. For trigonelline, the filtered supernatant was injected into an ODS C18 column (250 × 4.6 mm, 5 μm) at 35 °C, detected at 265 nm with a water/acetonitrile gradient (98![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 to 30
2 to 30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 70). For 4-HIL, the sample was injected into a Capcell Pak C18 UG 120 column (250 × 4.6 mm, 5 μm; YMC), detected at 338 nm with a flow rate of 0.7 mL min−1 and a 10 μl injection volume, using a gradient of 20 mM Na2HPO4 (pH 7.8) and 90% acetonitrile (85
70). For 4-HIL, the sample was injected into a Capcell Pak C18 UG 120 column (250 × 4.6 mm, 5 μm; YMC), detected at 338 nm with a flow rate of 0.7 mL min−1 and a 10 μl injection volume, using a gradient of 20 mM Na2HPO4 (pH 7.8) and 90% acetonitrile (85![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 15 to 10
15 to 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 90). Protodioscin detection involved injecting the sample into a Phenomenex Luna C18(2) column (100 × 4.6 mm, 3 μm), detected at 205 nm, at 1.0 mL min−1 with a 10 μL injection volume, employing a gradient of 0.1% formic acid in water and 0.1% formic acid in acetonitrile (78
90). Protodioscin detection involved injecting the sample into a Phenomenex Luna C18(2) column (100 × 4.6 mm, 3 μm), detected at 205 nm, at 1.0 mL min−1 with a 10 μL injection volume, employing a gradient of 0.1% formic acid in water and 0.1% formic acid in acetonitrile (78![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 22 to 10
22 to 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 90). System calibration was achieved using prepared standards ranging from 20 to 100 μg mL−1.
90). System calibration was achieved using prepared standards ranging from 20 to 100 μg mL−1.
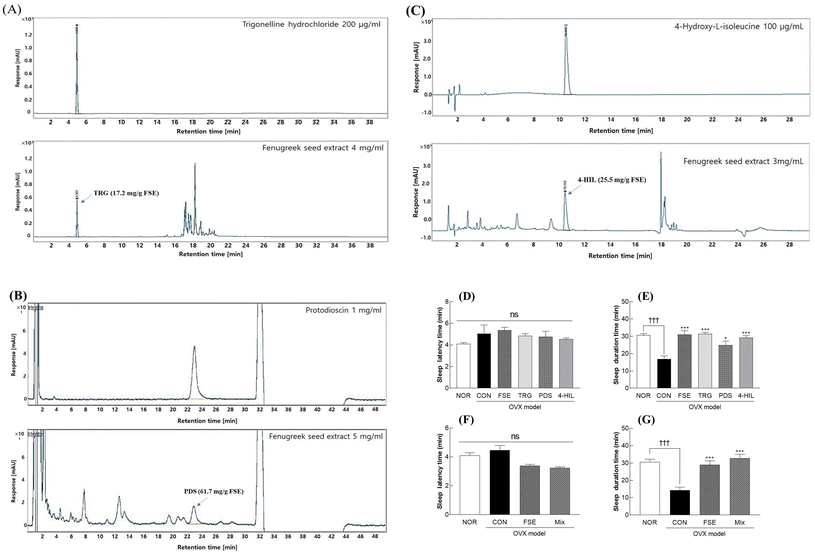
HPLC analyses were performed on three independent batches of FSE to confirm consistent content of key bioactive compounds. Quantification of trigonelline, 4-hydroxy-L-isoleucine, and protodioscin in FSE was carried out by comparing peak areas to those of external standards. Calibration curves were constructed using serial dilutions of each standard compound at appropriate concentrations: trigonelline (50–200 μg mL−1), 4-hydroxy-L-isoleucine (20–100 μg mL−1), and protodioscin (200–1000 μg mL−1). These ranges were selected based on expected compound concentrations in FSE. Each analyte showed excellent linearity within its respective range with coefficients of determination (R2) exceeding 0.99. As a result, the FSE was found to contain 6.17% protodioscin, 2.55% 4-hydroxy-L-isoleucine, and 1.72% trigonelline (w/w).
Statistical analysis
Statistical analysis was performed using the Statistical Package for Social Science (SPSS) version 26.0 (SPSS Inc., Chicago, IL, USA). Data were expressed as mean ± standard error of the mean (SEM). Group differences were analyzed using one-way ANOVA, followed by Tukey's multiple comparison test where appropriate. Statistical significance was set at a P-value < 0.05. Additionally, a t-test was conducted to compare specific group pairs.Results
FSE restores sleep impairment induced by ovariectomy-associated estrogen deficiency in ICR mice
Ovariectomy (OVX) successfully induced a menopause-like condition in ICR mice, as evidenced by a significant reduction in estrogen levels. Four weeks post-surgery, estrogen levels were notably decreased by 26.4% (p < 0.05) compared to those of normal (NOR) mice (Fig. 2A). Additionally, no significant changes in the weight of animal organs were observed in OVX mice compared to those of NOR mice (Table S3). This estrogen deficiency profoundly impacted the sleep duration: the OVX group (n = 7) exhibited a significant decrease in the sleep duration by 33.5% (p < 0.001) in the pentobarbital-induced sleep model (Fig. 2C), confirming that the menopause-like state negatively affects sleep. Regarding sleep latency, the fenugreek seed extract (FSE) generally did not cause significant changes across most doses. However, a low dose (FL, 200 mg kg−1) of FSE led to a 30.5% increase in sleep latency (Fig. 2B, p < 0.001). Crucially, FSE demonstrated a significant ability to counteract OVX-induced sleep disturbances. While a high dose of FSE (FH) did not alter sleep in sham (non-OVX) mice, it effectively restored sleep in OVX mice. In the pentobarbital-induced sleep model, both the positive control benzodiazepine (PC) and the high dose of FSE (FH) significantly counteracted the OVX-induced sleep reduction. PC increased the sleep duration by approximately 2.2-fold, and FH significantly increased it by 2.0-fold (Fig. 2C, p < 0.001). The low dose of FSE (FL) did not significantly affect the sleep duration in OVX mice (Fig. 2C). These findings indicate that FSE, particularly at the high dose, effectively mitigates OVX-induced sleep disturbance and restores sleep.FSE restores the sleep duration in an ovariectomized (OVX) mouse model of caffeine-induced insomnia
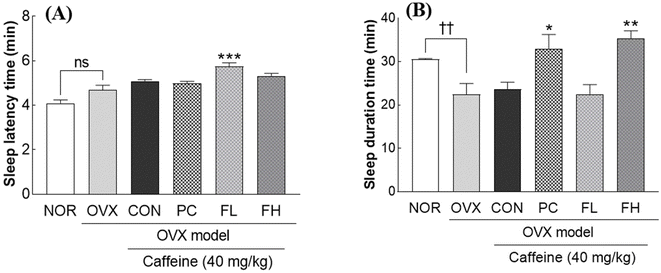
In an OVX model combined with caffeine administration, caffeine did not further exacerbate OVX-induced sleep disturbances, showing no significant additive effect on either sleep latency or sleep duration compared to OVX alone (Fig. 3A and B). Both OVX and caffeine independently reduced the sleep duration by 22.4% to 26.4% (p < 0.01 vs. NOR, Fig. 3B). Regarding sleep latency, a low dose of FSE (FL, 200 mg kg−1) significantly increased sleep latency by 13.1% compared to that of the OVX + caffeine control (CON) group (Fig. 3A, p < 0.001). Conversely, the high-dose FSE (FH) and the positive control (PC) groups did not significantly alter sleep latency (Fig. 3A). For the sleep duration, both the positive control (benzodiazepine, PC) and the high dose of FSE (FH) effectively counteracted the sleep reduction caused by OVX and caffeine. PC significantly increased the sleep duration by 39.2% (p < 0.05), while FH demonstrated a pronounced effect, significantly increasing the sleep duration by 49.0% (p < 0.01), compared to the CON group (Fig. 3B). The low dose of FSE (FL) did not significantly alter the sleep duration in this model (Fig. 3B). These results indicate that FSE, particularly at a high dose, effectively restores sleep even under conditions where OVX and caffeine contribute to sleep disturbance.FSE upregulates GABA and melatonin receptors in the brains of ovariectomized (OVX) mice
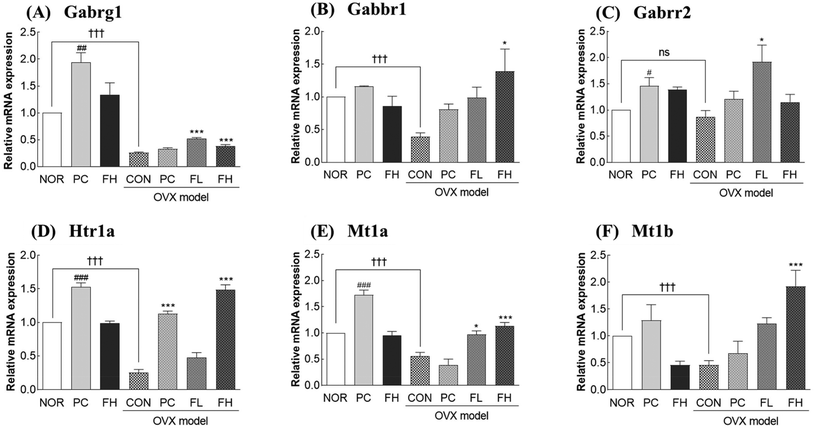
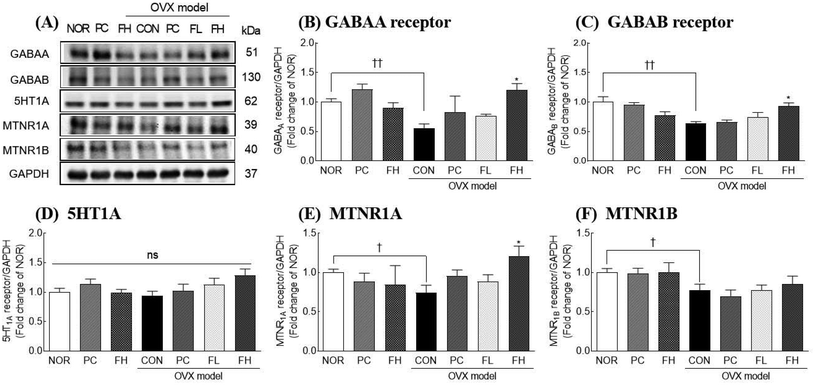
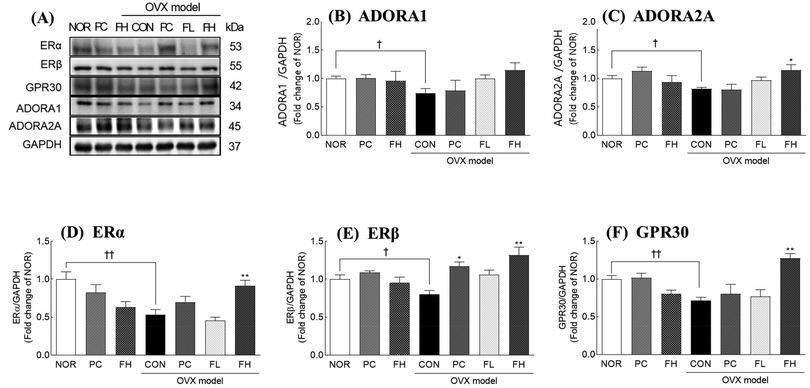
OVX significantly downregulated GABAergic, serotonergic, and melatonergic signaling pathways, as reflected by reduced receptor expression at both mRNA and protein levels in the brain. FSE administration effectively reversed the OVX-derived decreases. Specifically, at the mRNA level, GABAA receptor (Gabrg1) gene expression in OVX mice decreased by 74.0% compared to that in the NOR group (Fig. 4A). Treatment with FSE significantly increased this mRNA level, with the low dose (FL) achieving a 100% increase (p < 0.001) and the high dose (FH) showing a 46.1% increase (p < 0.001). Furthermore, FSE upregulated GABAB receptor gene expression (Gabbr1 and Gabbr2) in OVX mice, with FL increasing Gabbr1 and Gabbr2 by 2.5-fold and 2.2-fold, respectively, while FH increased them by 3.5-fold and 32.1% (Fig. 4B and C). Similarly, serotonin (Htr1a) and melatonin receptor (Mt1a, Mt1b) mRNA levels were significantly increased by FSE. 5HTR expression rose by 92.0% with FL and 5.9-fold with FH (Fig. 4D). For melatonin receptors, FL and FH increased Mt1a by 73.2% and 2.0-fold, respectively, and Mt1b by 2.6-fold and 1.1-fold, compared to those of the OVX control group (Fig. 4E and F). At the protein levels, similar regulatory patterns were observed, though with some notable exceptions. Specifically, FH administration significantly increased GABAA and GABAB receptor protein levels by 38.1% and 2.1-fold, respectively, compared to the OVX control group (Fig. 5B and C, p < 0.05). Additionally, FH upregulated MTNR1A protein levels by 63.5% relative to the OVX control group (Fig. 5E). However, 5HT1A (serotonin receptor) and MTNR1B (a melatonin receptor) protein levels showed only slight, non-significant increases (Fig. 5D and F). Taken together, these findings indicate that FSE effectively counteracts OVX-induced disruptions in neurotransmitter receptor expression, particularly within the GABAergic and melatonergic systems.FSE upregulates adenosine and estrogen receptor proteins in ovariectomized (OVX) mice
In OVX mice, protein levels of adenosine receptors, including ADORA1 and ADORA2A, were significantly reduced (Fig. 6A–C, p < 0.05). FSE administration induced an increasing trend in the protein levels of these receptors. While the increase in ADORA1 protein was not statistically significant, ADORA2A protein levels increased by 42.1% compared to the OVX control (CON) group, at a dose of 300 mg kg−1 (Fig. 6C, p < 0.05). This suggests that FSE partially regulates adenosine receptors, potentially contributing to sleep. Additionally, estrogen receptors, specifically ERα, ERβ, and GPR30, which were also significantly reduced in OVX mice (p < 0.01, p < 0.05, and p < 0.01, respectively), showed an increase in protein levels with FSE treatment. At a dose of 300 mg kg−1, FSE administration significantly increased ERα by 71.6%, ERβ by 63.7%, and GPR30 by 78.8% compared to those of the OVX control group (CON) (Fig. 6D–F, p < 0.01). These results indicate that FSE can increase estrogenic response by increasing the expression of estrogen receptors.FSE improves NREM sleep and delta wave activity in ovariectomized (OVX) SD rats
In ovariectomized (OVX) SD rats, FSE effectively counteracted OVX-induced sleep disturbances by reducing awake time and increasing the total sleep duration. Among the FSE fractions (FL and FH) tested, only the FH fraction demonstrated significant changes compared to the OVX control group. Specifically, FH administration led to a 21.6% increase in total sleep time (Fig. 7B and C). This FH-induced increase in sleep time was primarily attributable to an enhancement in non-rapid eye movement (NREM) sleep, which increased by 44.2% (Fig. 7E), while the rapid eye movement (REM) sleep duration remained unchanged (Fig. 7D). Furthermore, within the NREM sleep phase, FH significantly increased delta (δ) wave activity by 59.7%, suggesting an improvement in sleep quality (Fig. 7F). Although FH also showed a trend toward increasing theta (θ) wave activity, this change was not statistically significant (Fig. 7G). These results collectively demonstrate that FH effectively mitigates OVX-induced sleep disturbances by enhancing delta wave activity during NREM sleep, thereby improving sleep architecture.FSE enhances sleep via GABA subunits and melatonin receptor 1 in ovariectomized (OVX) mice
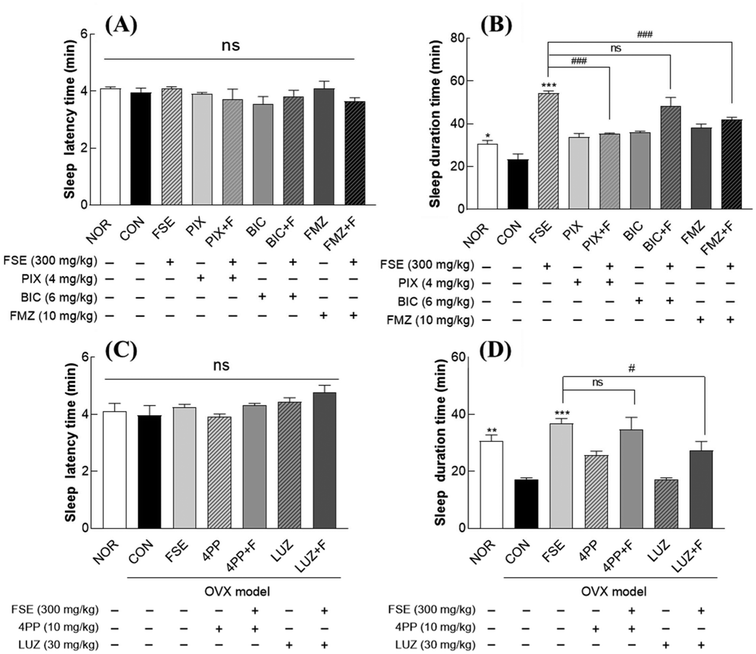
In OVX models treated with receptor antagonists, sleep latency was not significantly different among any of the groups (Fig. 8A and C). Regarding the GABAergic pathway, co-administration of FSE with picrotoxin (PIX) or flumazenil (FMZ) abolished the FSE-induced increase in the sleep duration, maintaining it at a level comparable to that of the OVX control group (Fig. 8B). This indicates that FSE-induced sleep enhancement is associated with the GABAA receptor Cl− ion channel (targeted by PIX) and benzodiazepine binding site located at the α1–γ2 subunit interface (targeted by FMZ). Conversely, co-administration of bicuculin (BIC) and FSE significantly increased the sleep duration by 34.1% compared to the BIC-alone group (Fig. 8B), indicating that FSE does not act through the BIC-binding site (α–β interface) of the GABA receptor. For the melatonergic receptor pathway, co-administration of 4-phenyl-2-propionaminotertrain (4PP), an antagonist of melatonin receptor 2 (MTNR2), with FSE resulted in a sleep duration comparable to that with FSE alone (Fig. 8D), suggesting that MTNR2 is not the primary target of FSE-mediated sleep enhancement. Conversely, co-administration of FSE and luzindole (LUZ), an MTNR1 and MTNR2 antagonist, significantly reduced the sleep duration compared to that with FSE alone (Fig. 8D), indicating that FSE-mediated sleep enhancement is closely associated with melatonin receptor 1 (MTNR1) activation. Taken together, these findings indicate that FSE promotes sleep by modulating the Cl− channel and α1/γ2 subunit of the GABAA receptor, and the MTNR1 receptor.FSE restores GABA and dopamine levels and reduces cortisol in ovariectomized (OVX) mice
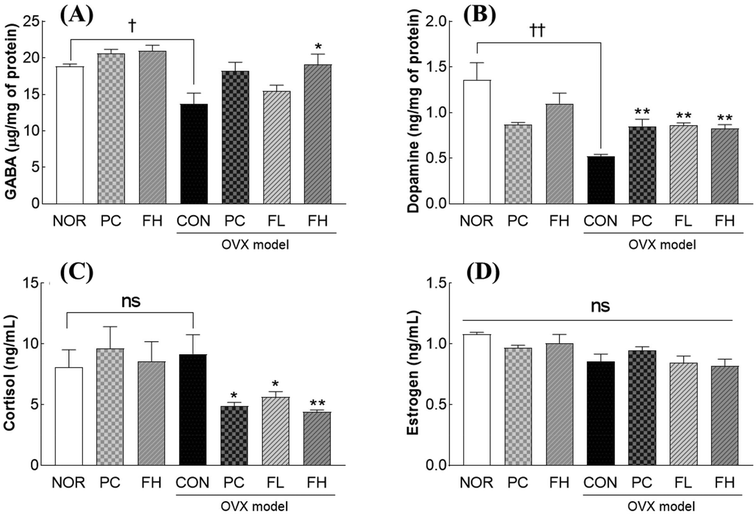
OVX significantly decreased brain GABA levels by 27.3%, whereas FH administration increased the GABA level by 39.5% compared to that with the OVX control (Fig. 9A and Fig. S1). Similarly, OVX caused a 61.7% decrease in dopamine levels (Fig. 9B and Fig. S1). However, both benzodiazepine (PC) and FSE (FL and FH) significantly restored dopamine levels; FL increased them by 65.3% and FH by 57.6%, respectively (Fig. 9B). While cortisol levels showed a non-significant slight increase under OVX conditions (Fig. 9C), both benzodiazepine (PC) and FH separately significantly reduced cortisol levels. PC decreased cortisol by 46.7% (p < 0.05) and FH by 51.7% (p < 0.01), respectively, compared to the OVX control group (Fig. 9C). In contrast, estrogen levels were significantly decreased by 20.8% in OVX mice, while FSE administration did not significantly alter estrogen levels (Fig. 9D). These findings indicate that FSE partially ameliorates OVX-induced disturbances in neurotransmitter and hormone levels, particularly by restoring GABA and dopamine levels and reducing elevated cortisol.FSE-derived compounds, especially trigonelline, increase the sleep duration in ovariectomized (OVX) mice
HPLC analysis identified key compounds in FSE: trigonelline (TRG) at 17.2 mg g−1, protodioscin (PDS) at 61.7 mg g−1, and 4-hydroxy-L-isoleucine (4-HIL) at 25.5 mg g−1 (Fig. 10A–C). In in vivo experiments, these compounds were administered individually or in combination at doses equivalent to their concentrations in the 300 mg kg−1 FSE (i.e., 5.16 mg kg−1 for TRG, 18.51 mg kg−1 for PDS, and 4.44 mg kg−1 for 4-HIL). Sleep latency did not significantly differ between the FSE-treated group and the individual compound groups (Fig. 10D and F). However, all three individual compounds significantly increased the sleep duration, counteracting the decrease induced by OVX (Fig. 10E). Specifically, TRG, PDS, and 4-HIL increased the sleep duration by 86.5%, 48.2%, and 73.6%, respectively, with TRG exhibiting effects similar to FSE (Fig. 10E, p < 0.001). When administered in combination, the mixture of these compounds produced a 2.2-fold increase in the sleep duration compared to that in the OVX control group, an effect higher than that observed with FSE alone (Fig. 10G). These results demonstrate that the three major compounds in FSE—TRG, PDS, and 4-HIL—effectively mitigate OVX-induced sleep disturbances, with TRG identified as the most potent compound in improving the sleep duration (Fig. 10).Discussion
This study investigated the effects of fenugreek seed extract (FSE) on sleep behavior in a menopausal-like condition and explored its underlying molecular mechanisms. Our findings demonstrated that FSE significantly alleviates sleep disturbances by modulating GABAergic and melatonergic pathways. In particular, antagonist-based animal studies revealed that FSE targets specific sites on the GABAA and MTNR1A receptors to mitigate OVX-induced sleep disruption. Additionally, FSE partially restored neurotransmitter and hormone imbalances induced by OVX, normalizing GABA and dopamine levels while reducing elevated cortisol. EEG analysis further showed that FSE predominantly enhances NREM sleep by increasing delta wave activity. Notably, although previous studies reported no sedative effects of fenugreek seeds at doses up to 200 mg kg−1 in normal pentobarbital-induced sleep models,16 our data revealed that a higher dose of 600 mg kg−1 significantly prolonged the sleep duration even under normal conditions (Fig. S2). Interestingly, FSE was effective at a lower dose (300 mg kg−1) in the OVX model (Fig. 2), suggesting greater efficacy under menopausal-like conditions. These findings demonstrate that FSE holds promise as a functional food ingredient targeting sleep disturbances associated with menopause, potentially offering a natural alternative to pharmacological or hormone-based interventions.During menopause, the decline in estrogen and progesterone is a major contributor to poor sleep quality, manifesting as insomnia, sleep fragmentation, and night sweats.12 These hormonal changes are known to disrupt neuroendocrine balance, leading to elevated cortisol and increased stress, which further exacerbate sleep disturbances.11 In our study, the OVX model exhibited reduced levels of GABA and dopamine, along with an increase in cortisol (Fig. 9). These neurochemical alterations were consistent with the observed impairment in the sleep duration (Fig. 2). Importantly, FSE administration effectively ameliorated this dysregulation, restoring GABA and dopamine levels while reducing cortisol, thereby improving sleep parameters (Fig. 2 and 9). Despite the significant decline in circulating estrogen in OVX mice, FSE did not restore estrogen levels (Fig. 9D). Instead, it upregulated ERα, ERβ, and GPR30 (Fig. 6), suggesting a receptor-mediated mechanism that may offer advantages over conventional hormone replacement therapy (HRT), which carries systemic risks due to elevated hormone exposure. Interestingly, a clinical study has reported that fenugreek extract supplementation increased circulating estradiol in postmenopausal women.23 This divergence from our findings may reflect species-specific differences in hormonal feedback regulation, or gut microbiota metabolism. Nevertheless, caution is warranted when translating these findings to humans, as placebo responses and inter-individual variability—including metabolic and hormonal status—may influence sleep outcomes. Future clinical studies should include placebo controls and account for such heterogeneity to accurately assess the efficacy of FSE for sleep improvement. Collectively, these findings suggest that the phytoestrogenic actions of FSE—particularly its ability to modulate receptor signaling and neuroendocrine balance—may contribute to the improvement of sleep disturbances associated with menopause.
Sleep regulation is a complex neurobiological process governed by multiple interacting systems, including the GABAergic, melatonergic, serotonergic, and adenosinergic pathways.24 Among these, the GABAergic system is the principal inhibitory pathway that promotes sleep initiation and maintenance, particularly by activating a brain region called the ventrolateral preoptic nucleus (VLPO), which suppresses wake-promoting centers.24 GABAA receptor agonists such as benzodiazepines—used as a positive control in this study—enhance GABAergic inhibition and are widely prescribed as hypnotics.25 Our antagonist-based animal study provided mechanistic insights into FSE's action on the GABAergic system. Specifically, picrotoxin (PIX) and flumazenil (FMZ), which block distinct sites on the GABAA receptor, were used to detect the receptor's involvement. PIX inhibits the chloride ion channel, whereas FMZ acts as a competitive antagonist at the benzodiazepine-binding site located at the α1–γ2 interface.26,27 Co-administration of either PIX or FMZ with FSE abolished the sleep-enhancing effects of FSE (Fig. 8B), suggesting that both sites may be essential targets of FSE's action. In contrast, co-treatment with bicuculline (BIC)—which binds the α–β subunit interface and also functions as a GABA-binding site antagonist28—did not significantly reduce the sleep-promoting effect of FSE compared to that with FSE alone. This suggests that the α–β site may not be a primary interaction site. However, since BIC alone reduced the sleep duration, the observed FSE-induced enhancement in the BIC + FSE group may be due to FSE-mediated upregulation of endogenous GABA levels (Fig. 8B and 9A), thus partially overcoming receptor blockade. Together, these results suggest that FSE mitigates OVX-induced sleep disturbances by enhancing GABAA receptor function through both ligand-binding site modulation and increased GABA availability. This is further supported by elevated GABAA receptor expression at both mRNA and protein levels following FSE treatment (Fig. 4 and 5). Nonetheless, our functional analysis focused exclusively on GABAA receptors. The potential involvement of GABAB receptors in FSE-induced sleep regulation remains to be elucidated. Future studies using GABAB-specific antagonists such as CGP55845 will be required to clarify their role in FSE's sleep-enhancing effects.
The melatonergic system, another critical sleep-regulating pathway, modulates the sleep–wake cycle via melatonin secretion from the pineal gland in response to light–dark cycles.29 Melatonin promotes sleep onset by binding to MTNR1A (Mt1a) and MTNR1B (Mt1b) receptors, thereby reinforcing circadian rhythms and suppressing wake-promoting neuronal activity.29 To elucidate the involvement of these receptors in FSE-induced sleep enhancement, we employed melatonin receptor-specific antagonists (LUZ, 4-P-PDOT). Our data indicated that the FSE-mediated increase in the sleep duration is primarily associated with melatonin receptor 1A (MTNR1A), as demonstrated by the significant reduction in the sleep duration upon co-administration of luzindole (LUZ), a non-selective MTNR1A/MTNR1B antagonist (Fig. 8D). In contrast, 4-phenyl-2-propionamidotetralin (4-P-PDOT), a selective MTNR1B antagonist, had no significant effect. This functional specificity is further supported by protein expression: although both Mt1a and Mt1b were transcriptionally upregulated (Fig. 4E and F), only MTNR1A protein levels significantly increased after FSE treatment (Fig. 5E), indicating that MTNR1A is the key receptor mediating FSE-induced sleep promotion.
Additionally, the serotonergic system is closely linked to sleep regulation, playing a complex dual role in the sleep–wake cycle.30 Serotonin (5-HT), released from the brainstem, promotes wakefulness and also contributes to melatonin production, linking these two systems in regulating sleep cycles.30 FSE significantly upregulated 5-HT1A (Htr1a) mRNA levels but did not affect its protein expression (Fig. 4D and 5D). This discrepancy between mRNA and protein levels may reflect post-transcriptional regulation or differences in protein stability, and should be considered when interpreting the functional outcomes. Therefore, further studies, including antagonist experiments, are necessary to clarify FSE's precise role in 5-HT1A receptor regulation.
Our investigation into the adenosinergic system provided important insights into FSE's multifaceted sleep-regulating effects, particularly its capacity to counteract wake-promoting stimuli. Adenosine receptors ADORA1 and ADORA2A were significantly downregulated in OVX mice (Fig. 6A–C). While FSE treatment showed a non-significant upward trend in ADORA1 protein levels, it significantly increased ADORA2A protein expression by 42.1% (Fig. 6C, p < 0.05). This modulation of adenosine receptors is especially relevant to FSE's ability to mitigate caffeine-induced insomnia. Behavioral data demonstrated that caffeine treatment in OVX mice reduced the sleep duration to a level comparable to those of OVX mice without caffeine, suggesting overlapping sleep-disrupting pathways. Nevertheless, FSE significantly prolonged the sleep duration despite caffeine administration (Fig. 3). Given that caffeine acts primarily as an antagonist of ADORA1 and ADORA2A,31 FSE appears to facilitate endogenous adenosine's sleep-promoting effects even under partial receptor blockade. These findings suggest that FSE exerts its sleep-enhancing effects through multiple pathways, including the GABAA receptor, MTNR1A receptor, and adenosine receptors as key targets.
Beyond neurotransmitter systems, FSE also altered the sleep architecture, as evidenced by enhanced delta wave activity in EEG recordings—indicative of deeper, more restorative non-rapid eye movement (NREM) sleep.32 This effect may underlie FSE's ability to alleviate OVX-induced sleep disturbances. Delta waves play a key role in synaptic plasticity, growth hormone secretion, and immune regulation.33 Notably, a decline in delta wave activity is commonly observed with aging and in sleep disorders such as insomnia and obstructive sleep apnea,34 underscoring the clinical significance of preserving delta oscillations. These findings suggest that FSE may serve as a natural, food-derived therapeutic agent for sleep disorders related to hormonal imbalance. While FSE primarily influenced NREM sleep, no significant changes were observed in REM sleep, which is associated with processes such as emotional regulation and memory consolidation.35 Importantly, abnormal EEG patterns—such as reduced delta activity in neurodegenerative diseases like Alzheimer's and excessive theta activity in ADHD—have been proposed as potential biomarkers.36,37 Since FSE modulates EEG-defined sleep architecture, its effects may extend beyond symptomatic relief, potentially contributing to the development of sleep-based biomarkers for neurological and endocrine disorders.
The compositional analysis of FSE revealed that its major constituents—trigonelline (TRG), protodioscin (PDS), and 4-hydroxy-L-isoleucine (4-HIL)—each significantly increased the sleep duration when administered individually to OVX mice with sleep disturbances (Fig. 10E). To the best of our knowledge, there are few reports directly investigating the sleep-promoting effects of these compounds in OVX models. Our study thus provides novel evidence that TRG, PDS, and 4-HIL significantly enhance the sleep duration under estrogen-deficient conditions, highlighting their potential functional roles as active constituents of standardized FSE.
Although previous studies have documented various bioactivities of these compounds—TRG exhibiting conditional estrogenic and neuroprotective properties,38 PDS showing androgenic and stress-attenuating effects,39 and 4-HIL demonstrating insulinotropic and metabolic regulatory actions40,41—direct evidence for their sleep-promoting roles has been limited. Nonetheless, these known physiological actions provide plausible mechanistic insights. TRG may facilitate sleep regulation by restoring neuroendocrine balance and alleviating estrogen deficiency–related perturbations;38 PDS could help stabilize hypothalamic–pituitary–adrenal (HPA) axis activity, thereby mitigating stress-induced sleep fragmentation;39 and 4-HIL might indirectly enhance sleep quality through improved energy metabolism and insulin sensitivity.40,41 Collectively, these effects are likely to act in concert to produce the observed enhancement of NREM sleep and delta wave activity. Importantly, the combined administration of TRG, PDS, and 4-HIL resulted in an even greater increase in the sleep duration than that of FSE itself (Fig. 10G). This suggests that these compounds not only serve as bioactive markers but also act as functional contributors to FSE's sleep-regulating effects. While the observed enhancement does not necessarily indicate a synergistic interaction, it supports the notion of additive or complementary actions among the compounds. Formal interaction analyses, such as isobolographic or combination index methods, were not performed in this study; however, future studies could apply these approaches to clarify potential synergistic mechanisms. Furthermore, considering that fenugreek seeds are rich in various phytochemicals—including polyphenols, flavonoids, alkaloids, and saponins18—other constituents beyond TRG, PDS, and 4-HIL may also contribute to the sleep-promoting effects observed in OVX-induced models.
The low (200 mg kg−1) and high (300 mg kg−1) doses of FSE were selected based on previous studies16 and our pilot experiments (Fig. S2) demonstrated physiological activity without toxicity. As noted above, 200 mg kg−1 was previously reported as ineffective in normal sleep models,16 whereas 300 mg kg−1 produced significant improvement in the OVX model (Fig. S2D), supporting its appropriateness as an effective dose under estrogen-deficient conditions. Both doses were effective, and no adverse effects were observed during the 4-week treatment period, indicating safety within this range. Although the difference between the low and high doses appears relatively small, significant differences in sleep-promoting activity were observed, highlighting a dose-dependent effect without apparent toxicity. To provide a translational perspective, human-equivalent doses (HEDs) were calculated using the standard body surface area normalization method (HED mg kg−1 = animal dose × [animal Km/human Km], Km: mouse 3, human 37). Accordingly, the 200 mg kg−1 and 300 mg kg−1 doses correspond to ∼16.2 mg kg−1 and ∼24.3 mg kg−1 in humans, translating to ∼973 mg and ∼1459 mg per day for a 60 kg adult—levels achievable for dietary supplementation or functional food use. Future studies should explore a broader dose–response relationship to fully characterize the optimal therapeutic window and confirm long-term safety of FSE.
Regarding standardization, the extract used in this study was characterized for TRG, PDS, and 4-HIL, quantified via HPLC with marker compounds, ensuring reproducible bioactivity. Although this standardization allowed consistent experimental results, the optimal proportions of these compounds for maximal sleep-promoting activity remain to be determined. Future investigations could systematically adjust the relative concentrations of TRG, PDS, and 4-HIL to identify formulations that maximize efficacy while maintaining safety. Additionally, quality control parameters such as extract purity, batch-to-batch consistency, and stability during storage should be considered in the standardization process to support potential functional food or nutraceutical applications.
The actions of FSE-derived compounds—trigonelline (TRG), protodioscin (PDS), and 4-hydroxy-L-isoleucine (4-HIL)—on sleep-related receptors such as GABAA and melatonin largely depend on their ability to cross the blood–brain barrier (BBB). Among them, TRG exhibits effective BBB permeability. Following oral administration, plasma TRG levels peaked at approximately 200 μg mL−1 within 5 min and gradually declined, while TRG concentrations in the cerebral cortex increased progressively from 5 min up to 24 h, indicating sustained accumulation in the brain.42 In contrast, the BBB permeability of PDS remains poorly understood, highlighting the need for pharmacokinetic and neuropharmacological studies to clarify its central effects. Conversely, 4-HIL, due to its high polarity, is unlikely to penetrate the BBB.43 However, its high gastrointestinal absorption43 suggests that it may exert effects via the enteric nervous system (ENS), implying a potential role for the brain–gut axis in mediating FSE's sleep-regulatory actions. Future studies should explore both the BBB permeability of PDS and the mechanistic contribution of the brain–gut axis, particularly in the context of hormone-related sleep disturbances.
This study has several limitations. It was performed using a short-term ovariectomized mouse model, which may not fully reflect long-term adaptations to FSE administration. Circadian rhythm-related parameters were not assessed, limiting our understanding of the temporal aspects of FSE-mediated sleep regulation. Additionally, long-term safety and toxicity were not evaluated. Future studies addressing these aspects will be essential to confirm the sustained efficacy and safety of FSE.
In conclusion, fenugreek seed extract (FSE) effectively alleviated OVX-induced sleep disturbances through a multifaceted mechanism (Fig. 11). The primary actions involved modulation of GABAergic and melatonergic pathways, with additional contributions from adenosinergic signaling and estrogen receptor upregulation. HPLC analysis identified trigonelline (TRG), protodioscin (PDS), and 4-hydroxy-L-isoleucine (4-HIL) as key bioactive compounds. TRG, which has been reported to cross the blood–brain barrier,42 may contribute to FSE's central effects on sleep regulation. Antagonist-based studies using FSE confirmed functional interactions with GABAA and MTNR1A receptors, while ADORA2A upregulation counteracted caffeine-induced insomnia. FSE also enhanced delta wave activity during NREM sleep, indicating improved restorative sleep, and increased estrogen receptor (ERα, ERβ, and GPR30) expression without altering circulating estrogen levels, supporting its potential as a safer alternative to hormone replacement therapy. To the best of our knowledge, this is the first study to comprehensively demonstrate the sleep-enhancing properties of FSE in a menopausal context, advancing functional food research by linking receptor-level mechanisms with pathway modulation. Collectively, these findings position FSE as a promising functional food ingredient for dietary supplements or fortified foods targeting menopause-related sleep disorders.
Author contributions
Hyowon Lee: investigation, validation, resources, methodology, writing – review & editing, and formal analysis. Hoon Kim: investigation, validation, resources, visualization, and methdology. Hyung Joo Suh and Hyeon-Son Choi: conceptualization, methodology, supervision, writing – original draft preparation, project administration, and writing – review & editing.Conflicts of interest
The authors declare that they have no competing interests.Abbreviations
| ADORA1 | Adenosine A1 receptor |
| ADORA2A | Adenosine A2A receptor |
| BBB | Blood brain barrier |
| BIC | Bicuculline |
| EEG | Electroencephalogram |
| ERα | Estrogen receptor alpha |
| ERβ | Estrogen receptor beta |
| FMZ | Flumazenil |
| FSE | Fenugreek seed extract |
| 5HT1A (Htr1a) | 5-Hydroxytryptamine receptor 1A receptor |
| 5HTR | 5-Hydroxytryptamine receptor |
| GABA | γ-Aminobutyric acid |
| GABAA | GABA type A receptor |
| GABAB | GABA type B receptor |
| Gabrg1 | Gamma-aminobutyric acid type A receptor subunit gamma1 |
| Gabbr1 | Gamma-aminobutyric acid type B receptor subunit 1 |
| Gabrr2 | Gamma-aminobutyric acid type C receptor subunit rho2 |
| GPR30 | G-protein coupled estrogen receptor 30 |
| 4-HIL | 4-Hydroxy-L-isoleucine |
| LUZ | Luzindole |
| MTNR1A (Mt1a) | Melatonin receptor 1A |
| MTNR1B (Mt1b) | Melatonin receptor 1B |
| NREM | Non-rapid eye movement |
| OVX | Ovariectomized |
| PDS | Protodioscin |
| PIX | Picrotoxin |
| 4PP (4-P-PDOT) | 4-Phenyl-1-(4-phenylbutyl) piperidine |
| REM | Rapid eye movement |
| TRG | Trigonelline |
Data availability
The data supporting this article have been included as part of the supplementary information (SI). The Supplementary Information includes chromatograms of GABA and dopamine levels (Fig. S1), the effects of FSE on sleep latency and total sleep time in normal and OVX mice (Fig. S2), PCR primer sequences (Table S1), antibodies used in the study (Table S2), and data on organ weights in ICR mice (Table S3). See DOI: https://doi.org/10.1039/d5fo03393d.References
- X. Y. Tai, C. Chen, S. Manohar and M. Husain, Impact of sleep duration on executive function and brain structure, Commun. Biol., 2022, 5, 201 CrossRef PubMed.
- T. Shochat, Impact of lifestyle and technology developments on sleep, Nat. Sci. Sleep, 2012, 4, 1931 Search PubMed.
- S. Garbarino, P. Lanteri, N. L. Bragazzi, N. Magnavita and E. Scoditti, Role of sleep deprivation in immune-related disease risk and outcomes, Commun. Biol., 2021, 4, 1304 CrossRef PubMed.
- M. L. Sambou, X. Zhao, T. Hong, J. Fan, T. B. Basnet, M. Zhu, C. Wang, D. Hang, Y. Jiang and J. Dai, Associations between sleep quality and health span: A prospective cohort study based on 328,850 UK Biobank Participants, Front. Genet., 2021, 12, 663449 CrossRef PubMed.
- N. S. Joo, For the sleep-related study, Korean J. Fam. Med., 2010, 31, 745–746 CrossRef.
- Y. Sun, M. Wei, Q. Zhao, J. Yang, J. Gao and J. Dai, Mediating effect of sleep quality on the association between job stress and health-related productivity loss among workers in R&D enterprises in Shanghai, Front. Public Health, 2024, 12, 1331458 CrossRef PubMed.
- L. Chen, J. S. Bell, R. Visvanathan, S. N. Hilmer, T. Emery, L. Robson, J. M. Hughes and E. C. K. Tan, The association between benzodiazepine use and sleep quality in residential aged care facilities: a cross-sectional study, BMC Geriatr., 2016, 16, 196 CrossRef PubMed.
- J. D. Edinger, J. T. Arnedt, S. M. Bertisch, C. E. Carney, J. J. Harrington, K. L. Lichstein, M. J. Saeia, W. M. Trpxel, E. S. Zhou, U. Kazmi, J. L. Heald and J. L. Martin, Behavioral and psychological treatments for chronic insomnia disorder in adults: an American Academy of Sleep Medicine systematic review, meta-analysis, and GRADE assessment, J. Clin. Sleep Med., 2021, 17, 263–298 CrossRef PubMed.
- J.-L. Yue, X.-W. Chang, J.-W. Zheng, L. Shi, Y.-J. Xiang, J.-Y. Que, K. Yuan, J.-H. Deng, T. Teng, Y.-Y. Li, W. Sun, H.-Q. Sun, M. V. Vitiello, X.-D. Tang, X.-Y. Zhou, Y.-P. Bao, J. Shi and L. Lu, Efficacy and tolerability of pharmacological treatments for insomnia in adults: A systematic review and network meta-analysis, Sleep Med. Rev., 2023, 68, 101746 CrossRef CAS PubMed.
- J. Ong and S. Suh, Utilizing cognitive-behavioral therapy for insomnia to facilitate discontinuation of sleep medication in chronic insomnia patients, Sleep Med. Res., 2012, 3, 1–6 CrossRef.
- A. Y. Cohn, L. K. Grant, M. D. Nathan, A. Wiley, M. Abramson, J. A. Harder, S. Crawford, E. B. Klerman, F. A. J. L. Scheer, U. B. Kaiser, S. A. Rahman and H. Joffe, Effects of sleep fragmentation and estradiol decline on cortisol in a human experimental model of menopause, J. Clin. Endocrinol. Metab., 2023, 108, e1347–e1357 CrossRef CAS PubMed.
- Z. Pan, S. Wen, X. Qiao, M. Yang, X. Shen and L. Xu, Different regimens of menopausal hormone therapy for improving sleep quality: a systematic review and meta-analysis, Menopause, 2022, 29, 627–635 CrossRef PubMed.
- A. Dorsey, L. de Lecea and K. J. Jennings, Neurobiological and hormonal mechanisms regulating women's sleep, Front. Neurosci., 2020, 14, 625397 CrossRef PubMed.
- N. Asiamah, C. Cronin, J. E. Abbott and S. Smith, Interactions of depression, anxiety, and sleep quality with menopausal symptoms on job satisfaction among middle-aged health workers in England: a STROBE-based analysis, Hum. Resour. Health, 2024, 22, 64 CrossRef PubMed.
- K. Srinivasan, Fenugreek (Trigonella foenum-graecum): A review of health beneficial physiological effects, Food Rev. Int., 2006, 22, 203–224 CrossRef CAS.
- T. Assad and R. A. Khan, Effect of methanol extract of Trigonella foenum-graecum L. seeds on anxiety, sedation and motor coordination, Metab. Brain Dis., 2017, 32, 343–349 CrossRef PubMed.
- K. C. Nagulapalli Venkata, A. Swaroop, D. Bagchi and A. Bishayee, A small plant with big benefits: Fenugreek (Trigonella foenum-graecum Linn.) for disease prevention and health promotion, Mol. Nutr. Food Res., 2017, 61, 1600950 CrossRef PubMed.
- M. H. Alu'datt, T. Rababah, S. Al-Ali, C. C. Tranchant, S. Gammoh, M. Alrosan, S. Kubow, T.-C. Tan and S. Ghatasheh, Current perspectives on fenugreek bioactive compounds and their potential impact on human health: A review of recent insights into functional foods and other high value applications, J. Food Sci., 2024, 89, 1835–1864 CrossRef PubMed.
- A. Jikah and G. Christopher, The bioactive constituents and biological properties of Trigonella Foenum-Graecum (Fenugreek): A comprehensive review, Int. J. Sci. Res. Arch., 2024, 13, 2140–2149 CrossRef.
- Y. Zhang, Z. Zhang, N. Kang and X. Sheng, Generation of a mouse artificial decidualization model with ovariectomy for endometrial decidualization research, J. Visualized Exp., 2022, 185, e64278 Search PubMed.
- K. Jo, S. Kim, K. B. Hong and H. J. Suh, Nelumbo nucifera promotes non-rapid eye movement sleep by regulating GABAergic receptors in rat model, J. Ethnopharmacol., 2021, 267, 13511 CrossRef PubMed.
- H. Kim, H. Kim, H. J. Suh and H. S. Choi, Lactobacillus brevis-fermented gamma-aminobutyric acid ameliorates depression- and anxiety-like behaviors by activating the brain-derived neurotrophic factor-tropomyosin receptor kinase B signaling pathway in BALB/C Mice, J. Agric. Food Chem., 2024, 72, 2977–2988 CrossRef CAS PubMed.
- S. Shamshad Begum, H. K. Jayalakshmi, H. G. Vidyavathi, G. Gopakumar, I. Abin, M. Balu, K. Keetha, S. V. Suesha, M. Vasundhara and M. Krishnakumar, A novel extract of fenugreek husk (FenuSMART) alleviates postmenopausal symptoms and helps to establish the hormonal balance: A randomized, double-blind, placebo-controlled study, Phytother. Res., 2016, 30, 1775–1784 CrossRef CAS PubMed.
- C. Falup-Pecurariu, S. Diaconu, D. Tint and O. Falup-Pecurariu, Neurobiology of sleep (Review), Exp. Ther. Med., 2021, 21, 272 CrossRef PubMed.
- U. Rudolph and F. Knoflach, Beyond classical benzodiazepines: novel therapeutic potential of GABAA receptor subtypes, Nat. Rev. Drug Discovery, 2011, 10, 685–697 CrossRef CAS PubMed.
- R. W. Olsen, Picrotoxin-like channel blockers of GABAA receptors, Proc. Natl. Acad. Sci. U. S. A., 2006, 103, 6081–6082 CrossRef CAS PubMed.
- S. A. Safavynia, G. Keating, I. Spiegel, J. A. Fidler, M. Kreuzer, D. B. Rye, A. Jenkins and P. S. Garcia, The effects of GABAA receptor modulation by flumazenil on emergence from general Anesthesia, Anesthesiology, 2016, 125, 147 CrossRef CAS PubMed.
- G. A. Johnston, Advantages of an antagonist: bicuculline and other GABA antagonists, Br. J. Pharmacol., 2013, 169, 328–336 CrossRef CAS PubMed.
- H. K. Kim and K. I. Yang, Melatonin and melatonergic drugs in sleep disorders, Transl. Clin. Pharmacol., 2022, 30, 163–171 CrossRef PubMed.
- M. Jouvet, Sleep and serotonin: an unfinished story, Neuropsychopharmacology, 1999, 21, 24S–27S CAS.
- L. López-Cruz, J. D. Salamone and M. Correa, Caffeine and selective adenosine receptor antagonists as new therapeutic tools for the motivational symptoms of depression, Front. Pharmacol., 2018, 9, 526 CrossRef PubMed.
- M. Schönauer and D. Pöhlchen, Sleep spindles, Curr. Biol., 2018, 28, R1129–R1130 CrossRef PubMed.
- S. Diekelmann and J. Born, The memory function of sleep, Nat. Rev. Neurosci., 2010, 11, 114–126 CrossRef CAS PubMed.
- B. A. Mander, J. R. Winer and M. P. Walker, Sleep and human aging, Neuron, 2017, 94, 19–36 CrossRef CAS PubMed.
- B. Rasch and J. Born, About sleep's role in memory, Physiol. Rev., 2013, 93, 681–766 CrossRef CAS PubMed.
- M. M. Lansbergen, M. Arns, M. van Dongen-Boomsma, D. Spronk and J. K. Buitelaar, The increase in theta/beta ratio on resting-state EEG in boys with attention-deficit/hyperactivity disorder is mediated by slow alpha peak frequency, Prog. Neuro-Psychopharmacol. Biol. Psychiatry, 2011, 35, 47–52 CrossRef PubMed.
- Y. F. Lee, D. Gerashchenko, I. Timofeev, B. J. Bacskai and K. V. Kastanenka, Slow wave sleep is a promising intervention target for alzheimer's disease, Front. Neurosci., 2020, 14, 705 CrossRef PubMed.
- K. M. Bhat, T. Nagamma and A. Konuri, Medicinal value of phytoestrogens in trigonella foenum-graecum L (Fenugreek)-review, Biomed. Pharmacol. J., 2023, 16, 1885 CAS.
- A. Maheshwari, N. Verma, A. Swaroop, M. Bagchi, H. G. Preuss, K. Tiwari and D. Bagchi, Efficacy of Furosap(TM), a novel trigonella foenum-graecum seed extract, in enhancing testosterone level and improving sperm profile in male volunteers, Int. J. Med. Sci., 2017, 14, 58–66 CrossRef CAS PubMed.
- A. Avalos-Soriano, R. De la Cruz-Cordero, J. L. Rosado and T. Garcia-Gasca, 4-Hydroxyisoleucine from fenugreek (Trigonella foenum-graecum): Effects on insulin resistance associated with obesity, Molecules, 2016, 21, 1596 CrossRef PubMed.
- M. I. Zafar and F. Gao, 4-Hydroxyisoleucine: A potential new treatment for type 2 diabetes Mellitus, Biodrugs, 2016, 30, 255–262 CrossRef CAS PubMed.
- M. M. Farid, X. Yang, T. Kuboyama and C. Tohda, Trigonelline recovers memory function in alzheimer's disease model mice: evidence of brain penetration and target molecule, Sci. Rep., 2020, 10, 16424 CrossRef CAS PubMed.
- I. Ahmad, A. E. Kuznetsov, A. S. Pirzada, K. F. Alsharif, M. Daglia and H. Khan, Computational pharmacology and computational chemistry of 4-hydroxyisoleucine: physicochemical, pharmacokinetic, and DFT-based approaches, Front. Chem., 2023, 11, 1145974 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2025 |