Open Access Article
Open Access ArticleEffects of kefirs made from whole milk or whey on the pancreas-intestine axis in a mouse model of acute pancreatitis
Diego
Aguirre-Ramírez
 ade,
Elisa
Abás
ade,
Elisa
Abás
 f,
Álex
Gómez
f,
Álex
Gómez
 cd,
Lluís
Luján
cd,
Lluís
Luján
 cd,
Estela
Pérez
cd,
Estela
Pérez
 c,
Lourdes
Sánchez
c,
Lourdes
Sánchez
 bd and
Laura
Grasa
bd and
Laura
Grasa
 *ade
*ade
aDepartamento de Farmacología, Fisiología y Medicina Legal y Forense, Facultad de Veterinaria, Universidad de Zaragoza, Spain. E-mail: lgralo@unizar.es
bDepartamento de Producción Animal y Ciencia de los Alimentos, Facultad de Veterinaria, Universidad de Zaragoza, Spain
cDepartamento de Patología Animal, Facultad de Veterinaria, Universidad de Zaragoza, Spain
dInstituto Agroalimentario de Aragón IA2 (UNIZAR-CITA), Zaragoza, Spain
eInstituto de Investigación Sanitaria de Aragón (IIS Aragón), Zaragoza, Spain
fInstituto de Síntesis Química y Catálisis Homogénea, Universidad de Zaragoza-CSIC, Zaragoza, Spain
First published on 24th November 2025
Abstract
Background: acute pancreatitis (AP) is a severe inflammatory condition, often involving intestinal barrier dysfunction. Fermented foods like kefir, are potential therapeutic adjuvants due to their gut-modulatory properties. This study aimed to compare the protective effects of kefir made from pasteurized whole milk (PMK) or whey (WK) on pancreatic injury, intestinal barrier integrity, inflammation, and oxidative stress in a cerulein-induced mouse model of AP. Methods: male BALB/c mice were assigned to six groups: control, AP, PMK, WK, and AP combined with either PMK or WK. AP was induced by cerulein injections on days 10 and 11. Mice received kefir or saline via oral gavage for 14 days. Pancreatic and ileal tissues were analyzed for histopathological damage, serum amylase and lipase levels, gene expression of inflammatory and tight junction markers (TNF-α, IL-6, IL-1β, occludin, ZO-1), oxidative stress (MDA, carbonyls), and intestinal function. Results: AP induction was confirmed by elevated serum amylase/lipase and severe pancreatic damage. While both PMK and WK partially reduced pancreatic histopathological damage, they failed to reduce serum amylase and lipase levels. Furthermore, PMK, but not WK, partially attenuated pancreatic IL-6 and IL-1β expression, and both kefirs reduced ileal TNF-α. However, neither kefir prevented the AP-induced downregulation of tight junction genes, the increase in oxidative stress markers in either tissue, or the impairment of intestinal transit and contractility. Conclusion: kefir supplementation offers localized anti-inflammatory benefits in a severe AP model. It is insufficient to mitigate key systemic markers of pancreatic injury, restore gut barrier integrity, or reverse functional dysmotility. These findings suggest that the therapeutic potential of kefir may be limited in the context of severe acute inflammatory conditions.
Introduction
Acute pancreatitis (AP) is a significant inflammatory disease of the pancreas, characterized by the premature activation and release of digestive enzymes, leading to pancreatic autodigestion, tissue damage, and glandular dysfunction that can extend to distant organs.1 Its incidence has notably increased, with reported rates varying globally2 and despite advances in supportive care, AP-associated mortality remains a great concern.3 While gallstones and alcohol abuse are common etiological factors,4 the intricate pathophysiology involves initial acinar cell injury, aberrant zymogen activation, and a subsequent cascade of inflammatory mediators5,6 contributing to both local and systemic complications.Among the organs frequently affected by AP, the intestine plays a critical role, highlighting the concept of a “pancreas-intestine axis”. Early in AP, intestinal motility is often disrupted, potentially due to the systemic inflammatory response, which can lead to luminal stasis and enteric bacterial overgrowth.7,8 Crucially, a significant proportion of AP patients exhibit increased intestinal permeability, facilitating bacterial translocation from the gut lumen into systemic circulation, thereby exacerbating inflammation and potentially leading to sepsis and multi-organ failure.9 This compromised gut barrier function underscores the interplay where pancreatic exocrine insufficiency can impact gut immunity,10 and conversely, intestinal health can influence pancreatic function.11,12
Current AP management primarily involves supportive care, including nutritional strategies, with endoscopic, radiological, and surgical interventions reserved for specific complications.6,13 Given the profound intestinal involvement in AP, interventions aimed at restoring gut homeostasis are of increasing interest. Probiotics, for instance, have been investigated for their potential to mitigate AP-associated intestinal complications by reducing inflammation, maintaining gut barrier integrity, and preventing dysbiosis-related infections.5,14,15 In this context, fermented foods rich in probiotics and bioactive compounds, such as kefir, represent a promising avenue for strategies to complement therapeutics.
Kefir, a traditional fermented beverage, is produced by the symbiotic action of a complex mixture of bacteria (lactic acid and acetic acid bacteria) and yeasts embedded in a polysaccharide-protein matrix known as kefiran.16 It is recognized for a wide array of health benefits, including improved digestion, antimicrobial activity, and modulation of immune responses, primarily attributed to its probiotic content and bioactive metabolites.17,18 Notably, kefir has demonstrated anti-inflammatory and antioxidant properties17,19 and has been suggested to modulate gut microbiota and potentially influence pancreatic enzyme levels,20 making it a compelling candidate for investigation in AP.
Furthermore, the substrate used for kefir fermentation can significantly influence its final composition and bioactive potential. Whey, a nutrient-rich byproduct of the dairy industry, is abundant in bioactive proteins (e.g., β-lactoglobulin, α-lactalbumin, lactoferrin) and peptides known for their antimicrobial, antioxidant, and immunomodulatory properties.21,22 Fermentation of whey with kefir cultures could potentially enhance the bioavailability of its components or generate novel bioactive compounds, offering unique beneficial properties compared to traditional whole milk kefir. However, the specific effects of kefirs, derived from different substrates, such as whey, on the interconnected pancreatic and intestinal disturbances characteristic of AP remain largely unexplored.
Therefore, considering the detrimental impact of AP on the pancreas-intestine axis and the potential gut-protective and immunomodulatory effects of fermented dairy products, this study aimed to investigate and compare the effects of kefirs made from pasteurized whole milk (PMK) or whey (WK) on pancreatic injury, intestinal barrier integrity, inflammatory responses, and oxidative stress in a cerulein-induced mouse model of acute pancreatitis. Our hypothesis was that both kefir beverages, could ameliorate AP-induced damage along the pancreas-intestine axis.
Experimental
Preparation of kefir drinks
Two different types of kefir drinks were obtained using as base substrate pasteurized fresh whole milk (PMK), obtained from the dairy industry Villacorona S.A. (El Burgo de Ebro, Zaragoza, Spain) or whey (WK). The procedure to obtain whey from pasteurized milk was performed by adding CaCl2 30% to the milk in the proportion 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1000 (v/v). The mix was stirred and kept at 34 °C for 45 min. After the coagulum was formed, it was mechanically cut and filtered to obtain the final whey. The composition of the kefir culture used for the two types of dairy drinks was obtained from Abiasa S.L. (Pontevedra, Spain) and consisted of Lactococcus lactis subsp. lactis, Lactococcus lactis subsp. cremoris, Lactobacillus delbrueckii subsp. bulgaricus, Lactobacillus helveticus, Lacticaseibacillus rhamnosus, Lacticaseibacillus paracasei, Lactobacillus acidophilus, Streptococcus thermophilus, Bifidobacterium bifidum and Leuconostoc mesenteroides. After the mixture of kefir culture with the substrates, drinks were kept at 25 °C for 22 hours, undergoing pH and acidity measurements at the beginning and at the end of the process. Kefir drinks were kept at 4 °C for two days before their use in the animal model of acute pancreatitis.
1000 (v/v). The mix was stirred and kept at 34 °C for 45 min. After the coagulum was formed, it was mechanically cut and filtered to obtain the final whey. The composition of the kefir culture used for the two types of dairy drinks was obtained from Abiasa S.L. (Pontevedra, Spain) and consisted of Lactococcus lactis subsp. lactis, Lactococcus lactis subsp. cremoris, Lactobacillus delbrueckii subsp. bulgaricus, Lactobacillus helveticus, Lacticaseibacillus rhamnosus, Lacticaseibacillus paracasei, Lactobacillus acidophilus, Streptococcus thermophilus, Bifidobacterium bifidum and Leuconostoc mesenteroides. After the mixture of kefir culture with the substrates, drinks were kept at 25 °C for 22 hours, undergoing pH and acidity measurements at the beginning and at the end of the process. Kefir drinks were kept at 4 °C for two days before their use in the animal model of acute pancreatitis.
Murine model of acute pancreatitis
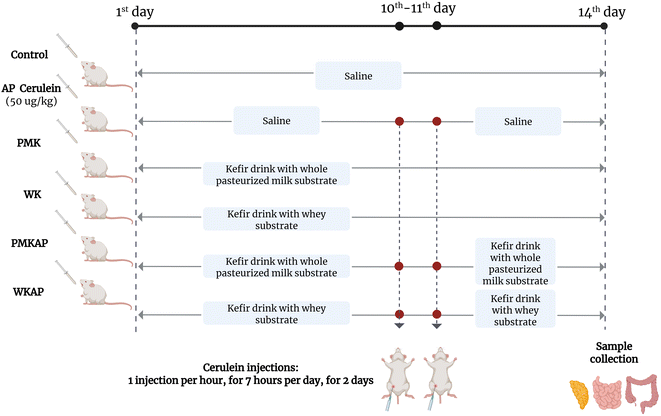
All procedures were approved by the Ethic Committee for Animal Experiments of the University of Zaragoza (Project License PI40/17). The care and use of animals were performed as stated in the Spanish Policy for Animal Protection RD53/2013, which meets the European Union Directive 2010/63 on the protection of animals used for experimental and other scientific purposes. All animals were kept in a conventional laboratory animal facility at the University of Zaragoza under a 12 h light/dark cycle, at a range of temperatures between 20–22 °C, with free access to animal chow and water.The study was carried out with 30 male BALB/cJRj mice (6–8 weeks old, Janvier Labs, Le Genest–Saints–Isle, France). For the experiment, mice were randomly divided into 6 groups (n = 5 per group): control, acute pancreatitis (AP), kefir made from pasteurized whole milk (PMK), kefir made from whey (WK), PMK + AP (PMKAP) and KW + AP (WKAP). Acute pancreatitis was induced via six daily intraperitoneal injections of cerulein 50 µg kg−1 in a volume of 100 μl administered hourly for 2 consecutive days (days 10 and 11). Similar doses of cerulein have shown to induce acute pancreatitis in mice.23–25 Control group received 6 IP injections of 100 μl saline at days 10 and 11. Control and AP groups were orally gavaged with 100 μl of saline for 14 days. PMK and WK groups received 100 μL of the respective kefir dairy drink by gastric gavage for 14 days. Based on body surface area (BSA) conversion,26 this dose corresponds to a human equivalent dose (HED) of approximately 20–28 mL day−1 for a 60–70 kg adult, which is well within the range of typical kefir consumption in humans. PMKAP and WKAP received the respective kefir dairy drink for 14 days and acute pancreatitis was induced on days 10 and 11 (Fig. 1).
Cerulein was obtained from MedChemExpress (FI-6934, Monmouth Junction, NJ, USA). A stock solution 1.25 mg ml−1 was prepared by dissolving the compound in a mixture of 10% dimethyl sulfoxide, 40% PEG 300, 5% Tween 80 and 45% saline according to the instructions of manufacturer.
Histopathological assessment
On day 14 of the study, mice were humanely sacrificed by cervical dislocation. Samples of pancreas and intestine were collected and fixed in 10% neutral buffered formaldehyde for 24 h, dehydrated, paraffin-embedded and four µm sections were obtained and stained with hematoxylin and eosin. Microscopic pancreatic evaluation determined the percentage of affected tissue, estimating globally the parameters presented in Table 1. For the ileum, a neutrophil counting was carried out in 10 consecutive fields of the intestine at 400× in the control and AP groups, only. In both cases, coded slides were evaluated by two persons unaware of the treatments.| Parameter | Description |
|---|---|
| Pancreatic degeneration | Loss of pancreatic acinar structure: |
| - Loss of staining | |
| - Indistinct cell borders | |
| - Cytoplasmic vacuolization | |
| - Nuclear pyknosis | |
| Acute inflammation | Viable neutrophils scattered in the interacinar fibrovascular tissue |
| Chronic inflammation | Infiltration of lymphocytes and plasma cells |
Lipase and amylase levels
Blood samples were extracted by intracardiac puncture and centrifuged at 5000 rpm for 10 min at 4 °C. The serum was collected for further assays. The activity of the lipase was detected using 10 μl of serum and the Lipase Activity Assay Kit MAK046 (Sigma-Aldrich, Madrid, Spain). This kit uses a coupled enzyme reaction that results in a colorimetric product that can be measured at 570 nm after 45 min, according to the manufacturer's instructions. The colorimetric product is proportional to the amount of lipase activity express as milliunits per mL, where one unit generates one micromole of glycerol. The activity of the amylase was detected using 10 μl of serum and the Amylase Activity Assay Kit MAK009 (Sigma Aldrich). This kit determined the amylase activity using a coupled enzymatic assay that results in a product which can be measured colorimetrically (405 nm). The samples were measured after 60 min according to the manufacturer's instructions. One unit obtained is the amount of amylase that cleaves ethylidene-pNP-G7 to generate 1.0 µmol of p-nitrophenol per min at 25 °C.Gene expression by real time-PCR
Tissue samples from pancreas and ileum were excised and preserved in RNAlater solution (Ambion, Thermo Fisher Scientific, Madrid, Spain) for 24 hours at 4 °C and then stored at −80 °C. Total RNA was isolated using the NZY Total RNA Isolation Kit (NZYTech, Lisbon, Portugal), according to manufacturer's specifications. cDNA was obtained from RNA using a NZY First-Strand cDNA Synthesis Kit (NZYTech) following the supplier's protocol. cDNAs obtained by reverse transcription were used to determine the mRNA expression levels of several genes related with inflammation and intestinal permeability. Real time PCR was carried out using the StepOne Plus Real-Time PCR System (Applied Biosystems, Carlsbad, CA, USA), using the NZY Speedy qPCR Green Master Mix (NZYTech) and specific primers listed in Table 2. Each sample was run in triplicate, and the mean Ct was determined from the three runs. Relative mRNA expression under each experimental condition was expressed as ΔCt = Ctgene − Ctcalibrators, with HPRT1 and YWHAZ as housekeeping genes for pancreas, and β-Actin for ileum. Then, the relative gene expression levels were calculated as ΔΔCt = ΔCtcontrol − ΔCttreatment, and finally converted and expressed as fold difference (2−ΔΔCt).| Gene | Forward primer (5′–3′) | Reverse primer (5′–3′) | Ref. |
|---|---|---|---|
| HPRT1 | CTGACCTGCTGGATTACA | GCGACCTTGACCATCTTT | 48 |
| β-Actin | TCTACAATGAGCTGCGTG | CCTTAATGTCACGCACG | 49 |
| YWHAZ | CAGTAGATGGAGAAAGATTTGC | GGGACAATTAGGGAAGTAAGT | 50 |
| IL-1β | AGAGCATCCAGCTTCAAATCTC | CAGTTGTCTAATGGGAACGTCA | 51 |
| IL-6 | GTTGCCTTCTTGGGACTGATG | ATTGCCATTGCACAACTCTTT | 51 |
| iNOS | GTTCTCAGCCCAACAATACAAGA | GTGGACGGGTCGATGTCA | 52 |
| TNF-α | TTAGAAAGGGGATTATGGCTA | ACTCTCCCTTTGCAGAACTCAG | 51 |
| ZO-1 | ACTCCCACTTCCCCAAAAAC | CCACAGCTGAAGGACTCACA | 53 |
| Occludin | AGTACATGGCTGCTGCTGATG | CCCACCATCCTCTTGATGTGT | 54 |
Measurement of oxidative stress
Pancreas and ileum samples were collected from each animal and immediately snap-frozen in liquid nitrogen. Samples were stored at −80 °C until analysis. For biochemical assays, frozen tissues were homogenized in ice-cold Tris buffer (50 mM, pH 7.4) using an Ultra Turrax homogenizer (Yellowline DI 25, IKA-Werke, Staufen, Germany). The homogenates were centrifuged (11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000g, 10 min, 4 °C) and the resulting supernatants were used for subsequent analyses. Total protein concentration in each homogenate was determined using the Bradford assay (Bio-Rad, Hercules, CA, USA), with bovine serum albumin (BSA) as the standard.
000g, 10 min, 4 °C) and the resulting supernatants were used for subsequent analyses. Total protein concentration in each homogenate was determined using the Bradford assay (Bio-Rad, Hercules, CA, USA), with bovine serum albumin (BSA) as the standard.
Lipid peroxidation was assessed by measuring the levels of malondialdehyde (MDA) and 4-hydroxyalkenals (4-HDA). The method is based on the reaction of these aldehydes with N-methyl-2-phenylindole, which generates a stable chromophore with a maximum absorbance at 586 nm. 1,1,3,3-Tetramethoxypropane was used as an MDA precursor for the calibration curve. Results were normalized to protein concentration and expressed as nmol of (MDA + 4-HDA) per mg of protein.
Oxidative protein damage was determined by measuring the carbonyl group content, as described previously by our group.27 Briefly, 100 µL of sample homogenate were incubated with 200 µL of 10 mM dinitrophenylhydrazine (DNPH, dissolved in 2.5 M HCl) for 1 hour at 37 °C in the dark. Subsequently, proteins were precipitated with 325 µL of 20% (w/v) trichloroacetic acid (TCA) on ice. Following centrifugation (11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000g, 3 min, 4 °C), the protein pellet was washed three times with 1 mL of an ethanol
000g, 3 min, 4 °C), the protein pellet was washed three times with 1 mL of an ethanol![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ethyl acetate mixture (1
ethyl acetate mixture (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, v/v) to remove excess reagent. The final pellet was resuspended in 700 µL of 6 M guanidine (in phosphate buffer) and incubated at 37 °C for 15 min to ensure complete dissolution. Absorbance was measured at 375 nm, using 6 M guanidine as a blank. Results were expressed as nmol of carbonyl groups per mg of protein.
1, v/v) to remove excess reagent. The final pellet was resuspended in 700 µL of 6 M guanidine (in phosphate buffer) and incubated at 37 °C for 15 min to ensure complete dissolution. Absorbance was measured at 375 nm, using 6 M guanidine as a blank. Results were expressed as nmol of carbonyl groups per mg of protein.
Intestinal transit
After the administration of the treatments, mice were kept for 14 hours in fasting. One hour prior to the start of the experiment, the animals were transferred to an individual cage for individual monitoring. A tracer consisting of 200 μl of an Evans blue marker (5% solution of gum arabic and 5% non-absorbable Evans blue dye) was administered by gastric gavage. Total gastrointestinal transit was measured by recording the time (min) it took the animals to expel the first dark blue pellet.Muscle contractility studies
Ileum from mice was rapidly harvested and located in an ice-cold Krebs buffer. Ileum segments (10 mm-length) with intact mucosa were suspended in the longitudinal direction of the smooth muscle fibers in an organ bath, thermostatically controlled at 37 °C with Krebs solution and gassed continuously (95% O2 and 5% CO2). Each segment was connected to an isometric force transducer (Pioden UF1, Graham Bell House, Canterbury, UK) and stretched passively to an initial tension of 0.5 g. The mechanical activity signal was amplified (The Mac Lab Bridge Amp, AD Instruments Inc., Milford, MA, USA) to a range of 2 mV and recorded to be analyzed and digitized (two samples/second/channel) in the Mac Lab System/8e computer program (AD Instruments Inc., Milford, MA, USA). Before testing, the segments were equilibrated for 1 h in Krebs buffer, changed every 20 min. After this period of equilibration, the spontaneous motility patterns of ileum from all animals were acquired. To study the effect of the different conditions of acute pancreatitis and treatments with the kefir drinks on the intestinal contractility, we added acetylcholine (ACh, 100 µM) or serotonin (5-hydroxytryptamine, 5-HT, 100 µM) for 3 min to the organ bath.ACh or 5-HT-induced contractions were measured as integrated mechanical activity (IMA) as follows: IMA = A1 − A0, where A1 and A0 are the integrated area per second after and before adding the substances, respectively. The IMA was expressed as millinewtons per second. The ACh or 5-HT contractions of the different groups of animals were expressed as a percentage of control group (100%).
Statistical analysis
Results were expressed as the mean ± standard error of the mean (SEM). All statistical analysis were performed using the statistical software GraphPad Prism v8.0.2 (GraphPad Software, San Diego, CA, USA). For the analysis of all results, the normality of data was verified with Shapiro–Wilk Test. For data that followed a normal distribution, an analysis of variance (ANOVA) was executed followed by a Dunnett's test; for data that did not follow a normal distribution, a non-parametric Kruskal–Wallis test was done, followed by a Dunn's test. P values <0.05 were considered to be statistically significant.Results
Histopathological assessment
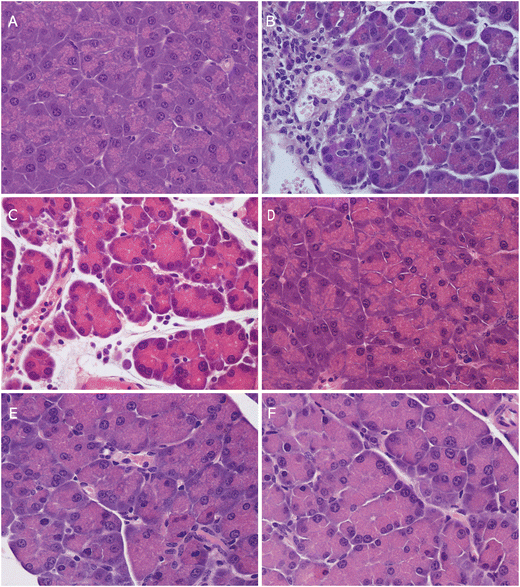
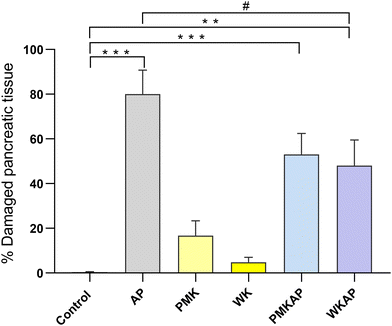
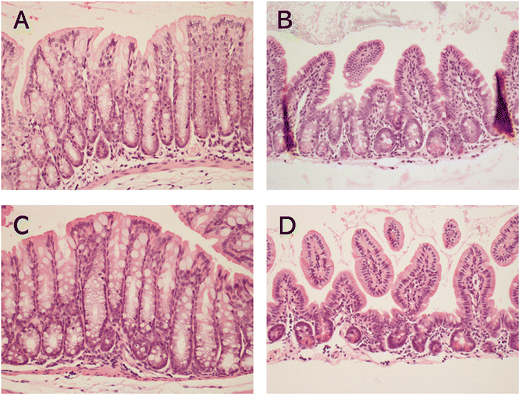
The AP group displayed pronounced histopathological alterations characterized by dense neutrophilic inflammatory cell infiltration, accompanied by interstitial edema and loss of normal acinar organization, typical of acute inflammatory response to cerulein-induced pancreatic injury, whereas the control group maintained a well-preserved pancreatic structure (Fig. 2A, B and 3). In the PMK group (Fig. 2C), slight but not significant inflammatory changes (Fig. 3) were observed compared to the control group, while the WK group (Fig. 2D) maintained a structure similar to the control. The PMKAP group (Fig. 2E) presented mild signs of AP but tissue damage was less severe, and the integrity of the pancreatic acini was better preserved, though no statistically significant differences were observed compared with the AP group (Fig. 3). Similarly, in the WKAP group (Fig. 2F), inflammatory infiltration and cellular changes were present but they were milder than in the AP group, differences not reaching a significant level (Fig. 3).Acute pancreatitis did not induce discernible pathological changes in the intestine. The histomorphology of the ileum, including the integrity of the villi, was unaltered in the AP group compared to control (Fig. 4A and B). Number of neutrophils infiltrating the ileum mucosa was similar in the control (4.6 ± 1.94) and AP (3.6 ± 1.47) groups, differences not showing a significant level (p = 0.688). Similarly, the colonic mucosa retained its normal structure, with no signs of edema, inflammatory infiltrate, or epithelial damage in either group (Fig. 4C and D).
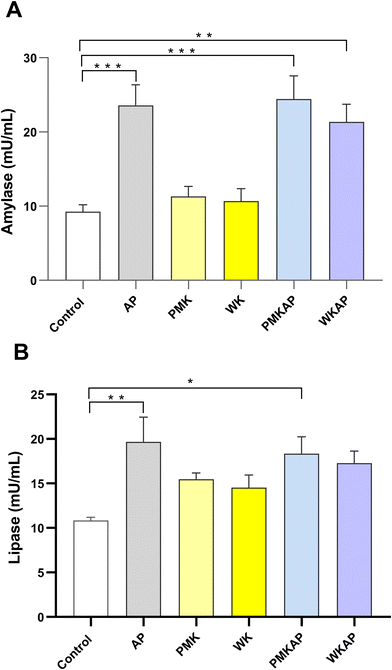
Serum marker levels
To evaluate the effect of PMK and WK on pancreatic damage, serum levels of amylase and lipase were measured (Fig. 5). The induction of acute pancreatitis resulted in a strong and statistically significant increase in serum amylase (Fig. 5A) and lipase (Fig. 5B) activities compared to the control group, thus confirming the successful establishment of the acute pancreatitis model.Treatment with PMK and WK individually did not modify significantly the amylase and lipase levels respect to control group (Fig. 5). Notably, the combined treatment groups (PMKAP and WKAP) failed to mitigate the increase in pancreatic damage markers. Amylase levels in the PMKAP and WKAP groups remained elevated, showing no significant difference from the AP group, and were still significantly higher than the control group (Fig. 5A). Analogously, lipase levels in these groups showed no significant reduction compared to the AP group (Fig. 5B).
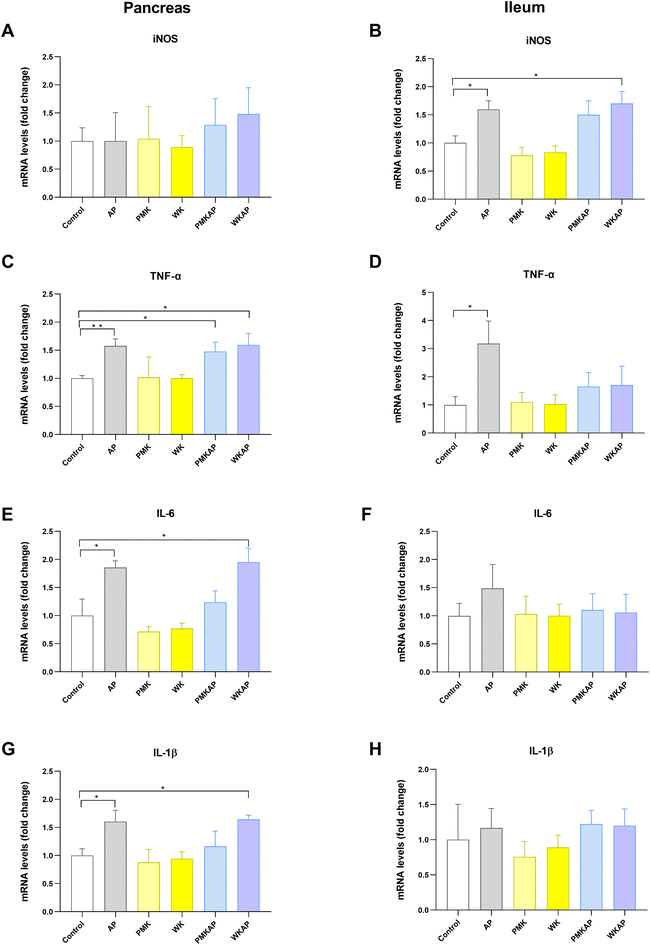
Gene expression levels of inflammatory mediators
In pancreatic tissue, the induction of acute pancreatitis (AP group) led to a significant upregulation of TNF-α (Fig. 6C), IL-6 (Fig. 6E), and IL-1β (Fig. 6G) expression compared to the control group. In contrast, the mRNA levels of iNOS remained unchanged across all groups (Fig. 6A). Individual treatment with PMK or WK did not modify the expression of TNF-α, IL-6, and IL-1β compared to the control group (Fig. 6C, E and G). The expression of TNF-α, IL-6, and IL-1β in WKAP groups remained elevated, showing no significant difference from the AP group, indicating that the supplementation with WK failed to exert a protective effect (Fig. 6C, E and G). However, the levels of IL-6, and IL-1β in PMKAP groups were partially reduced and similar to the control group, indicating that the supplementation with PMK exert a protective effect on the pancreas (Fig. 6E and G).To assess distant inflammation, gene expression was analyzed in the ileum. Acute pancreatitis also induced an inflammatory response in this tissue, as evidenced by a significant increase in the expression of iNOS (Fig. 6B) and TNF-α (Fig. 6D) compared to controls. Unlike in the pancreas, no significant changes were observed in the mRNA levels of IL-6 (Fig. 6F) or IL-1β (Fig. 6H) in the ileum. Consistent with the findings in the pancreas, individual treatments with PMK and WK did not modify the expression of iNOS and TNF-α compared to the control group (Fig. 6B and D). The groups PMKAP and WKAP failed to reduce the expression of iNOS, which remained at high levels similar to the AP group (Fig. 6B), but reduced the expression of TNF-α to control levels, indicating that the supplementation with PMK and WK exert a protective effect in the ileum (Fig. 6D).
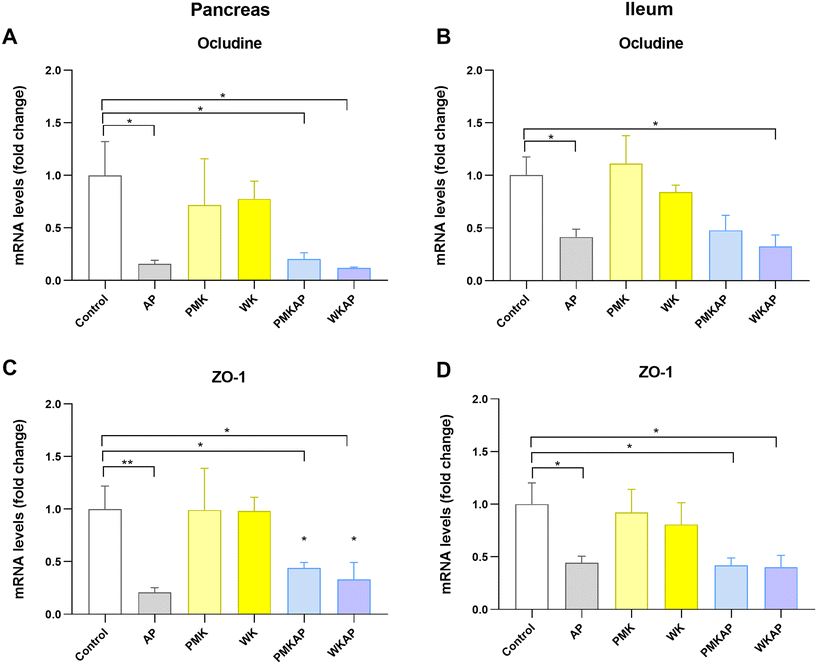
Gene expression levels of tight junction proteins
In the pancreas, acute pancreatitis induced a drastic and significant downregulation of both occludin (Fig. 7A) and ZO-1 (Fig. 7C) expression compared to the control group, indicating a loss of local barrier integrity. In the pancreas, these tight junction proteins are mainly expressed in epithelial ductal cells, and their dysregulation compromises ductal epithelial barrier function and integrity. Individual treatment with either PMK or WK did not modify the expression of these proteins in pancreas. The treatments with PMK or WK failed to prevent the loss of tight junction gene expression in pancreas from mice with AP. Occludin and ZO-1 levels in PMKAP and WKAP groups remained as low as in the untreated AP group and were still significantly below control levels (Fig. 7A and C).A similar disruption of the distant barrier was observed in the ileum, where the AP group showed a significant reduction in the mRNA levels of occludin (Fig. 7B) and ZO-1 (Fig. 7D). Individual treatment with either PMK or WK did not modify the expression of these proteins in ileum. Consistent with all other findings, the PMK and WK treatments were ineffective at preserving tight junction protein expression in the ileum from mice with AP, with levels remaining significantly depressed (Fig. 7B and D).
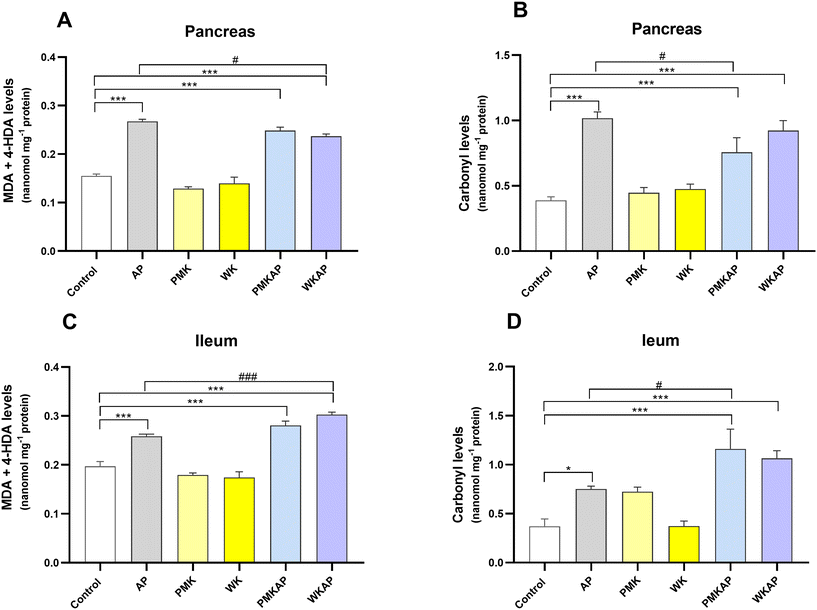
Evaluation of oxidative stress
To investigate the role of oxidative stress, we measured markers of lipid peroxidation (MDA + 4-HDA) and protein oxidation (carbonyls) in both pancreatic and ileal tissue (Fig. 8). In both the pancreas and intestine, the induction of AP caused a significant increase in both MDA + 4-HDA levels (Fig. 8A and C) and carbonyl levels (Fig. 8B and D) compared to the control group. Treatment with PMK or WK did not modify the oxidative stress and both MDA + 4-HDA and carbonyl levels were similar to control in pancreas and intestine (Fig. 8). The supplementation with PMK or WK failed to provide any protection against oxidative stress induced by AP. MDA + 4-HDA and carbonyl levels in PMKAP and WKAP groups were not significantly different from the AP group in pancreas or ileum (Fig. 8).Intestinal transit and muscle contractility
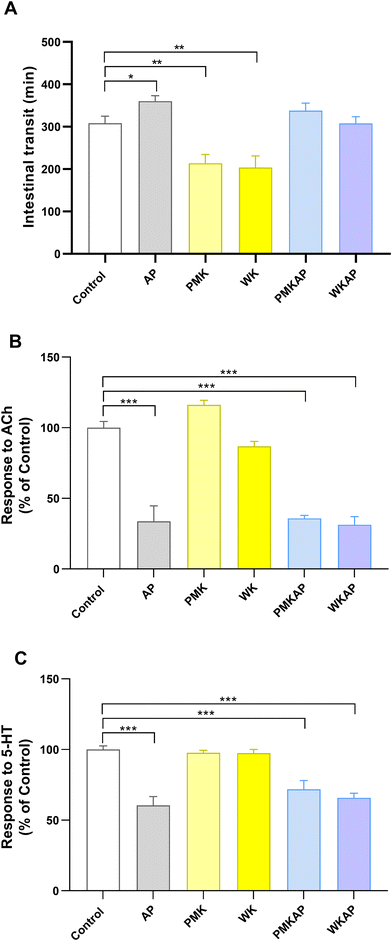
To determine the functional consequences of AP, we evaluated gastrointestinal transit time and ex vivo ileal contractility (Fig. 9). The AP group exhibited a slight but significant delay in intestinal transit compared to the control group (Fig. 9A). In stark contrast, individual treatments with either PMK or WK significantly accelerated transit time (Fig. 9A). In the groups PMKAP and WKAP transit times remained prolonged and were not significantly different from the AP group.To investigate the mechanisms underlying this dysmotility, we assessed the contractile response of isolated ileum strips to both acetylcholine (ACh) and serotonin (5-HT). As shown in Fig. 9B and C, AP induced a severe impairment in the contractile response to both ACh and 5-HT. Individual treatments with either PMK or WK did not modify the ACh or 5-HT contractile response respect to control (Fig. 9B and C). In the groups PMKAP and WKAP, ACh or 5-HT contractile responses remained decreased and were not significantly different from the AP group.
Discussion
This study provides a comprehensive evaluation of the potential protective effects of kefir made from pasteurized milk (PMK) and whey kefir (WK) supplementation against cerulein-induced acute pancreatitis (AP) and its associated distant organ injury in the intestine. Our findings reveal a complex and nuanced response, where kefir offered only partial and compartment-specific benefits. While both kefir beverages types modestly attenuated pancreatic histopathological damage and ileal TNF-α expression, they failed to mitigate key systemic markers of pancreatic injury, functional gut impairments, oxidative stress, and the disruption of tight junction protein expression. These results challenge the notion of a broad protective effect of kefir in severe inflammatory conditions like AP, underscoring both the complexity of functional food interactions and the preventive – not curative-intention of this treatment in modulating disease progression.A cornerstone of our study was the successful induction of a robust AP model, confirmed by severe pancreatic tissue damage, including edema, inflammatory infiltration, and acinar cell disarray, and a dramatic elevation in serum amylase and lipase levels. These are hallmark features of clinical and experimental AP,28,29 validating our model for investigating therapeutic interventions. Interestingly, while the individual administration of PMK or WK did not induce any adverse effects, their co-administration with cerulein yielded mixed results. We observed a partial amelioration of the histological damage score in the pancreas for both PMKAP and WKAP groups. This suggests that kefir components may interfere with some aspects of the inflammatory cascade, such as leukocyte infiltration or edema formation, consistent with previous studies demonstrating the anti-inflammatory properties of kefir-derived peptides and exopolysaccharides.30,31
However, this subtle histopathological improvement was discordant with the systemic biochemical markers. Neither PMK nor WK supplementation could prevent the massive surge in serum amylase and lipase, which are diagnostic and prognostic indicators of AP severity.32 This discrepancy suggests that while kefir may modulate the local inflammatory microenvironment to some extent, it is insufficient to prevent the initial acinar cell injury and massive enzyme release that drive the pathology of AP. This finding is crucial, as it implies that the protective mechanism is not aimed at the primary insult but rather at secondary inflammatory amplification, indicating a preventive effect. A particularly noteworthy finding was the differential effect observed in the pancreas, where whole milk (PMK), but not whey (WK), partially reduced IL-6 and IL-1β expression. This disparity likely stems from the significant compositional differences between the two fractions. Whole milk has higher concentrations of fat, protein, and minerals compared to whey, and it is plausible that these compounds would have exerted a more potent immunomodulatory effect on pancreatic resident immune cells than those of WK. Indeed, both bovine milk and whey are known to contain numerous bioactive proteins—such as α-lactalbumin, β-lactoglobulin, immunoglobulins, lactoferrin, and lactoperoxidase—with recognized anti-inflammatory properties.33 Although in our study the microbiota of kefir beverages made from milk and whey has not been characterized, other studies have found that the structure and microbiota of kefir grains and beverages remained consistent when using cheese whey instead of milk as a substrate, including the dominant microbiota bacteria from the Lactobacillus genus.34
The gut–pancreas axis is a critical determinant of AP severity, as intestinal barrier dysfunction can lead to bacterial translocation and the systemic amplification of inflammation.35,36 A surprising finding of our study was the absence of overt histopathological damage in the ileum and colon of AP mice, despite molecular and functional disturbances. This indicates that significant gut pathology in AP can occur even before it is histopathologically apparent. Indeed, we found that AP induced a strong inflammatory response in the ileum, characterized by increased expression of iNOS and TNF-α. Both PMK and WK effectively normalized TNF-α expression, consistent with their previously described preventive anti-inflammatory activity in the gut.37 This localized anti-inflammatory effect, however, did not translate into broader protection.
Critically, either kefir beverage could prevent the AP-induced downregulation of tight junction genes (occludin and ZO-1) in either the pancreas or the ileum. The loss of these key barrier proteins is a pivotal event in AP, contributing to increased pancreatic permeability38 and increased intestinal permeability that fuels systemic inflammation.39 The inability of kefir to preserve tight junction expression, despite reducing TNF-α, suggests that other, more potent pathways triggered by AP, perhaps involving myosin light chain kinase (MLCK) activation or other pro-inflammatory cytokines, are the dominant drivers of barrier loss in this severe model.40,41
Consistent with previous reports, acute pancreatitis (AP) induced a marked increase in lipid and protein oxidation in both the pancreas and the ileum.42,43 However, contrary to our initial hypothesis, kefir administration failed to mitigate this effect in the pancreas. A possible explanation for this discrepancy is the treatment duration, which may have been insufficient to elicit a protective antioxidant response. Furthermore, the antioxidant properties of kefir are highly species and even strain-dependent.44 While certain strains have shown antioxidant activity in other models, such as inflammatory bowel disease,45 it is plausible that the specific microbial consortium in our kefir beverages did not possess the necessary antioxidant capacity within the context of AP.
These molecular disruptions culminated in severe functional consequences. AP induced a significant delay in gastrointestinal transit and profoundly impaired ileal muscle contractility in response to both cholinergic and serotonergic stimulation, classic features of intestinal dysmotility associated with pancreatitis.46,47 Although PMK and WK alone exhibited a prokinetic effect, they were completely unable to reverse the AP-induced dysmotility, and their potential preventive action could not be demonstrated under the conditions of this study. This suggests that the damage to the enteric nervous system (ENS) and/or smooth muscle cells caused by the systemic inflammation and oxidative stress in AP is too severe to be overcome by the beneficial effects of kefir.
Our study has several limitations. We assessed a single time point after AP induction, which provides a snapshot but not the full dynamic evolution of the disease and recovery. Although anti-inflammatory benefit of kefir is supported in our study by gene expression analysis, other mechanisms involved in the pancreas–intestine axis require further investigation, including microbiota sequencing and metabolomic analysis. Moreover, the use of kefir-based beverages in this context should be considered as a complementary or preventive nutritional approach rather than a therapeutic or palliative treatment.
In conclusion, our study demonstrates that PMK and WK may offer localized anti-inflammatory benefits in a severe model of acute pancreatitis, but they are insufficient to counteract the systemic disease hallmarks, including pancreatic enzyme release, gut barrier disruption, oxidative stress, and functional intestinal dysmotility. These findings urge for caution when extrapolating the benefits of functional foods to severe pathological conditions, as kefir-based beverages should be regarded as having preventive potential as complementary aids in maintaining health.
Author contributions
D. A.-R.: investigation, methodology, formal analysis, data curation, writing – original draft. E. A.: investigation, methodology. A. G: formal analysis. L. L.: formal analysis, writing – review & editing. E. P.: formal analysis. L. S.: conceptualization, project administration, funding acquisition, writing – review & editing. L. G.: conceptualization, project administration, funding acquisition, supervision, writing – review & editing.Conflicts of interest
There are no conflicts to declare.Data availability
All raw and processed data generated during this study have been deposited in Zenodo and are publicly available at: https://doi.org/10.5281/zenodo.17608463.Acknowledgements
This work was funded by the European Union and the Spanish State Research Agency under project PID2022-139104OB-I00 and the Gobierno de Aragón (A20_23R). D. Aguirre-Ramírez was supported by a grant by the European Union Next-Generation EU.References
- M. S. Petrov and D. Yadav, Global epidemiology and holistic prevention of pancreatitis, Nat. Rev. Gastroenterol. Hepatol., 2019, 16, 175–184 CrossRef PubMed.
- G. Sarri, Y. Guo, I. Iheanacho and J. Puelles, Moderately severe and severe acute pancreatitis : a systematic review of the outcomes in the USA and European Union-5, BMJ Open Gastroenterol., 2019, 6, e000248 CrossRef PubMed.
- C. C. Popa, D. C. Badiu, O. C. Rusu, V. T. Grigorean, S. I. Neagu and C. R. Strugaru, Mortality prognostic factors in acute pancreatitis, J. Med. Life, 2016, 9, 413–418 CAS.
- L. Boxhoorn, R. P. Voermans, S. A. Bouwense, M. J. Bruno, R. C. Verdonk, M. A. Boermeester, H. C. van Santvoort and M. G. Besselink, Acute pancreatitis, Lancet, 2020, 396, 726–734 CrossRef PubMed.
- S. Gou, Z. Yang, T. Liu, H. Wu and C. Wang, Use of probiotics in the treatment of severe acute pancreatitis: a systematic review and meta-analysis of randomized controlled trials, Crit. Care, 2014, 18, R57 CrossRef PubMed.
- M. A. Mederos, H. A. Reber and M. D. Girgis, Acute Pancreatitis: A Review, J. Am. Med. Assoc., 2021, 325, 382–390 CrossRef CAS PubMed.
- P. Pulipati and R. Talukdar, Gut microbiome linked to pancreatitis, Curr. Opin. Physiol., 2021, 23, 100470 CrossRef CAS.
- J. Zhao, C. Zhong, Z. He, G. Chen and W. Tang, Effect of da-cheng-qi decoction on pancreatitis-associated intestinal dysmotility in patients and in rat models, J. Evidence-Based Complementary Altern. Med., 2015, 2015, 895717 Search PubMed.
- J. Beiriger, A. Khan, B. Yan, H. Ross, M. Wang, M. Carducci, N. S. Parra, S. Chowdhury, R. Erwin, P. Forrest, S. Chen and A. Gerber, Comprehensive Review of Acute Pancreatitis Pain Syndrome, Gastrointest. Disord., 2023, 5, 144–166 Search PubMed.
- Y. Kurashima, T. Kigoshi, S. Murasaki, F. Arai, K. Shimada, N. Seki, Y. G. Kim, K. Hase, H. Ohno, K. Kawano, H. Ashida, T. Suzuki, M. Morimoto, Y. Saito, A. Sasou, Y. Goda, Y. Yuki, Y. Inagaki, H. Iijima, W. Suda, M. Hattori and H. Kiyono, Pancreatic glycoprotein 2 is a first line of defense for mucosal protection in intestinal inflammation, Nat. Commun., 2021, 12, 1067 CrossRef CAS PubMed.
- S. J. Konturek, R. Zabielski, J. W. Konturek and J. Czarnecki, Neuroendocrinology of the pancreas; role of brain-gut axis in pancreatic secretion, Eur. J. Pharmacol., 2003, 481, 1–14 CrossRef CAS PubMed.
- Z. Zhang, I. Tanaka, Z. Pan, P. B. Ernst, H. Kiyono and Y. Kurashima, Intestinal homeostasis and inflammation: Gut microbiota at the crossroads of pancreas-intestinal barrier axis, Eur. J. Immunol., 2022, 52, 1035–1046 CrossRef CAS PubMed.
- J. Walkowska, N. Zielinska, R. S. Tubbs, M. Podgorski, J. Dlubek-Ruxer and L. Olewnik, Diagnosis and Treatment of Acute Pancreatitis, Diagnostics, 2022, 12(8), 1974 CrossRef CAS PubMed.
- D. Werawatganon, S. Vivatvakin, K. Somanawat, S. Tumwasorn, N. Klaikeaw, P. Siriviriyakul and M. Chayanupatkul, Effects of probiotics on pancreatic inflammation and intestinal integrity in mice with acute pancreatitis, BMC Complementary Med. Ther., 2023, 23, 166 CrossRef CAS PubMed.
- M. M. Zhang, J. Q. Cheng, Y. R. Lu, Z. H. Yi, P. Yang and X. T. Wu, Use of pre-, pro- and synbiotics in patients with acute pancreatitis: a meta-analysis, World J. Gastroenterol., 2010, 16, 3970–3978 CrossRef PubMed.
- D. D. Rosa, M. M. S. Dias, L. M. Grzeskowiak, S. A. Reis, L. L. Conceicao and M. Peluzio, Milk kefir: nutritional, microbiological and health benefits, Nutr. Res. Rev., 2017, 30, 82–96 CrossRef CAS PubMed.
- V. Ganatsios, P. Nigam, S. Plessas and A. Terpou, Kefir as a Functional Beverage Gaining Momentum towards Its Health Promoting Attributes, Beverages, 2021, 7, 48 CrossRef CAS.
- D. Aguirre-Ramírez, I. Abad, E. Pinilla, M. D. Pérez, L. Grasa and L. Sánchez, Antibacterial activity and antioxidant capacity of dairy kefir beverages, Int. J. Dairy Technol., 2025, 78(1), e13153 CrossRef.
- S. M′hir, K. Rtibi, A. Mejri, M. Ziadi, H. Aloui, M. Hamdi and L. Ayed, Development of a Novel Whey Date Beverage Fermented with Kefir Grains Using Response Surface Methodology, J. Chem., 2019, 1218058 Search PubMed.
- M. Tiss, Z. Souiy, N. b. Abdeljelil, M. Njima, L. Achour and K. Hamden, Fermented soy milk prepared using kefir grains prevents and ameliorates obesity, type 2 diabetes, hyperlipidemia and Liver-Kidney toxicities in HFFD-rats, J. Funct. Foods, 2020, 67, 103869 CrossRef CAS.
- A. Belles, I. Abad, L. Sanchez and L. Grasa, Whey and Buttermilk-Based Formulas Modulate Gut Microbiota in Mice with Antibiotic-Induced Dysbiosis, Mol. Nutr. Food Res., 2023, e2300248, DOI:10.1002/mnfr.202300248.
- S. Patel, Emerging trends in nutraceutical applications of whey protein and its derivatives, J. Food Sci. Technol., 2015, 52, 6847–6858 CrossRef CAS PubMed.
- M. Bombardo, E. Saponara, E. Malagola, R. Chen, G. M. Seleznik, C. Haumaitre, E. Quilichini, A. Zabel, T. Reding, R. Graf and S. Sonda, Class I histone deacetylase inhibition improves pancreatitis outcome by limiting leukocyte recruitment and acinar-to-ductal metaplasia, Br. J. Pharmacol., 2017, 174, 3865–3880 CrossRef CAS PubMed.
- R. Chen, T. Hornemann, S. Stefanic, E. M. Schraner, R. Zuellig, T. Reding, E. Malagola, D. C. Henstridge, A. P. Hills, R. Graf and S. Sonda, Serine administration as a novel prophylactic approach to reduce the severity of acute pancreatitis during diabetes in mice, Diabetologia, 2020, 63, 1885–1899 CrossRef CAS PubMed.
- M. Durst, T. R. Graf, R. Graf, M. Kron, M. Arras, D. Zechner, R. Palme, S. R. Talbot and P. Jirkof, Analysis of Pain and Analgesia Protocols in Acute Cerulein-Induced Pancreatitis in Male C57BL/6 Mice, Front. Physiol., 2021, 12, 744638 CrossRef PubMed.
- S. Reagan-Shaw, M. Nihal and N. Ahmad, Dose translation from animal to human studies revisited, FASEB J., 2008, 22, 659–661 CrossRef CAS PubMed.
- S. Gonzalo, L. Grasa, M. P. Arruebo, M. A. Plaza and M. D. Murillo, Extracellular signal-regulated kinase (ERK) is involved in LPS-induced disturbances in intestinal motility, Neurogastroenterol. Motil., 2011, 23, e80–e90 CrossRef CAS PubMed.
- L. Bettac, S. Denk, T. Seufferlein and M. Huber-Lang, Complement in Pancreatic Disease-Perpetrator or Savior?, Front. Immunol., 2017, 8, 15 Search PubMed.
- G. Klöppel, in The Pancreas, 2018, ch. 17, pp. 193–198, DOI:10.1002/9781119188421.ch19.
- K. L. Rodrigues, J. C. Carvalho and J. M. Schneedorf, Anti-inflammatory properties of kefir and its polysaccharide extract, Inflammopharmacology, 2005, 13, 485–492 CrossRef CAS PubMed.
- M. K. Seo, E. J. Park, S. Y. Ko, E. W. Choi and S. Kim, Therapeutic effects of kefir grain Lactobacillus-derived extracellular vesicles in mice with 2,4,6-trinitrobenzene sulfonic acid-induced inflammatory bowel disease, J. Dairy Sci., 2018, 101, 8662–8671 CrossRef CAS PubMed.
- D. Yadav, N. Agarwal and C. S. Pitchumoni, A critical evaluation of laboratory tests in acute pancreatitis, Am. J. Gastroenterol., 2002, 97, 1309–1318 CrossRef CAS PubMed.
- D. E. Chatterton, D. N. Nguyen, S. B. Bering and P. T. Sangild, Anti-inflammatory mechanisms of bioactive milk proteins in the intestine of newborns, Int. J. Biochem. Cell Biol., 2013, 45, 1730–1747 CrossRef CAS PubMed.
- K. T. Magalhaes, M. A. Pereira, A. Nicolau, G. Dragone, L. Domingues, J. A. Teixeira, J. B. de Almeida Silva and R. F. Schwan, Production of fermented cheese whey-based beverage using kefir grains as starter culture: evaluation of morphological and microbial variations, Bioresour. Technol., 2010, 101, 8843–8850 CrossRef CAS PubMed.
- C. Zhang, G. Li, T. Lu, L. Liu, Y. Sui, R. Bai, L. Li and B. Sun, The Interaction of Microbiome and Pancreas in Acute Pancreatitis, Biomolecules, 2023, 14(1), 59 CrossRef PubMed.
- X. Y. Li, C. He, Y. Zhu and N. H. Lu, Role of gut microbiota on intestinal barrier function in acute pancreatitis, World J. Gastroenterol., 2020, 26, 2187–2193 CrossRef CAS PubMed.
- C. G. Vinderola, J. Duarte, D. Thangavel, G. Perdigon, E. Farnworth and C. Matar, Immunomodulating capacity of kefir, J. Dairy Res., 2005, 72, 195–202 CrossRef CAS PubMed.
- M. Schmitt, H. Klonowski-Stumpe, M. Eckert, R. Luthen and D. Haussinger, Disruption of paracellular sealing is an early event in acute caerulein-pancreatitis, Pancreas, 2004, 28, 181–190 CrossRef PubMed.
- H. Fukui, Increased Intestinal Permeability and Decreased Barrier Function: Does It Really Influence the Risk of Inflammation?, Inflamm. Intest. Dis., 2016, 1, 135–145 CrossRef PubMed.
- W. Q. He, J. Wang, J. Y. Sheng, J. M. Zha, W. V. Graham and J. R. Turner, Contributions of Myosin Light Chain Kinase to Regulation of Epithelial Paracellular Permeability and Mucosal Homeostasis, Int. J. Mol. Sci., 2020, 21(3), 993 CrossRef CAS PubMed.
- Y. J. Xiong, C. O. Wang, L. Q. Shi, L. Wang, Z. J. Zhou, D. P. Chen, J. Y. Wang and H. S. Guo, Myosin Light Chain Kinase: A Potential Target for Treatment of Inflammatory Diseases, Front. Pharmacol., 2017, 8, 292 CrossRef PubMed.
- F. Bishehsari, A. Sharma, K. Stello, C. Toth, M. R. O'Connell, A. C. Evans, J. LaRusch, V. Muddana, G. I. Papachristou and D. C. Whitcomb, TNF-alpha gene (TNFA) variants increase risk for multi-organ dysfunction syndrome (MODS) in acute pancreatitis, Pancreatology, 2012, 12, 113–118 CrossRef CAS PubMed.
- E. Bozkir, B. Yilmaz, H. Sharma, T. Esatbeyoglu and F. Ozogul, Challenges in water kefir production and limitations in human consumption: A comprehensive review of current knowledge, Heliyon, 2024, 10(13), e33501 CrossRef CAS PubMed.
- S. Abbring, G. Hols, J. Garssen and B. van Esch, Raw cow's milk consumption and allergic diseases - The potential role of bioactive whey proteins, Eur. J. Pharmacol., 2019, 843, 55–65 CrossRef CAS PubMed.
- Y. Kudoh, S. Matsuda, K. Igoshi and T. Oki, Antioxidative Peptide from Milk Fermented with Lactobacillus delbrueckii subsp. bulgaricus IFO13953, Nippon Shokuhin Kagaku Kogaku Kaishi, 2001, 48, 44–50 CrossRef CAS.
- T. C. Seerden, B. Y. De Winter, R. M. Van Den Bossche, A. G. Herman, P. A. Pelckmans and J. G. De Man, Regional differences in gastrointestinal motility disturbances during acute necrotising pancreatitis, Neurogastroenterol. Motil., 2005, 17, 671–679 CrossRef CAS PubMed.
- T. C. Seerden, J. G. De Man, P. Holzer, R. M. Van den Bossche, A. G. Herman, P. A. Pelckmans and B. Y. De Winter, Experimental pancreatitis disturbs gastrointestinal and colonic motility in mice: effect of the prokinetic agent tegaserod, Neurogastroenterol. Motil., 2007, 19, 856–864 CrossRef CAS PubMed.
- B. Buey, A. Belles, E. Latorre, I. Abad, M. D. Perez, L. Grasa, J. E. Mesonero and L. Sanchez, Comparative effect of bovine buttermilk, whey, and lactoferrin on the innate immunity receptors and oxidative status of intestinal epithelial cells, Biochem. Cell Biol., 2021, 99, 54–60 CrossRef CAS PubMed.
- R. R. Negi, S. V. Rana, V. Gupta, R. Gupta, V. D. Chadha, K. K. Prasad and D. K. Dhawan, Over-Expression of Cyclooxygenase-2 in Colorectal Cancer Patients, Asian Pac. J. Cancer Prev., 2019, 20, 1675–1681 CrossRef CAS PubMed.
- Z. Yan, J. Gao, X. Lv, W. Yang, S. Wen, H. Tong and C. Tang, Corrigendum to “Quantitative Evaluation and Selection of Reference Genes for Quantitative RT-PCR in Mouse Acute Pancreatitis”, BioMed Res. Int., 2017, 3498537 Search PubMed.
- H. Huang, Y. Liu, J. Daniluk, S. Gaiser, J. Chu, H. Wang, Z. S. Li, C. D. Logsdon and B. Ji, Activation of nuclear factor-kappaB in acinar cells increases the severity of pancreatitis in mice, Gastroenterology, 2013, 144, 202–210 CrossRef CAS PubMed.
- M. M. LaRue, S. Parker, J. Puccini, M. Cammer, A. C. Kimmelman and D. Bar-Sagi, Metabolic reprogramming of tumor-associated macrophages by collagen turnover promotes fibrosis in pancreatic cancer, Proc. Natl. Acad. Sci. U. S. A., 2022, 119, e2119168119 CrossRef CAS PubMed.
- I. Hwang, E. J. Hong, H. Yang, H. S. Kang, C. Ahn, B. S. An and E. B. Jeung, Regulation of tight junction gene expression in the kidney of calbindin-D9k and/or -D28k knockout mice after consumption of a calcium- or a calcium/vitamin D-deficient diet, BMC Biochem., 2014, 15, 6 CrossRef PubMed.
- M. Meynier, E. Baudu, N. Rolhion, M. Defaye, M. Straube, V. Daugey, M. Modoux, I. Wawrzyniak, F. Delbac, R. Villeger, M. Meleine, E. Borras Nogues, C. Godfraind, N. Barnich, D. Ardid, P. Poirier, H. Sokol, J. M. Chatel, P. Langella, V. Livrelli, M. Bonnet and F. A. Carvalho, AhR/IL-22 pathway as new target for the treatment of post-infectious irritable bowel syndrome symptoms, Gut Microbes, 2022, 14, 2022997 CrossRef PubMed.
| This journal is © The Royal Society of Chemistry 2025 |