Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Cocoa flavanols alleviate early diastolic dysfunction by decreasing left atrial volume in a randomized double blinded trial in healthy older individuals
Dragos Andrei
Duse†
 a,
Michael
Gröne†
a,
Niklas
Ophoff
a,
Nicolas
Kramser
a,
Hendrik
Schweers
a,
Javier
Ottaviani
a,
Michael
Gröne†
a,
Niklas
Ophoff
a,
Nicolas
Kramser
a,
Hendrik
Schweers
a,
Javier
Ottaviani
 b,
Hagen
Schroeter
b,
Christine
Quast
b,
Hagen
Schroeter
b,
Christine
Quast
 a,
Florian
Bönner
a,
Christian
Heiss
a,
Florian
Bönner
a,
Christian
Heiss
 c,
Roberto
Sansone
a,
Christian
Jung
ad,
Malte
Kelm
ad and
Ralf
Erkens
c,
Roberto
Sansone
a,
Christian
Jung
ad,
Malte
Kelm
ad and
Ralf
Erkens
 *ae
*ae
aDepartment of Cardiology, Pulmonology and Angiology Medical Faculty, Heinrich Heine University of Duesseldorf, Duesseldorf, Germany. E-mail: ralf.erkens@med.uni-duesseldorf.de
bMars Incorporated, 6885 Elm St, McLean, VA 22101, USA
cDepartment of Clinical and Experimental Medicine, Faculty of Health and Medical Science, University of Surrey, UK
dCardiovascular Research Institute Duesseldorf (CARID), Duesseldorf, Germany
eDepartment of Cardiology and Electrophysiology, St. Agnes-Hospital Bocholt, Klinikum Westmünsterland, Bocholt, Germany
First published on 27th October 2025
Abstract
Aims: Left atrial (LA) enlargement marks impaired cardiac filling and predicts future cardiovascular events. Dietary flavanols have been shown to reduce cardiovascular mortality, despite uncertain underlying mechanisms. In this study, we hypothesized that flavanol intake reduces LA volume in older individuals without cardiovascular diseases. Methods: In a substudy of the randomized, double-blinded Healthy Aging Through Dietary Intervention trial (NCT 05782309), we investigated the effects of cocoa flavanols on cardiac volumetry and diastolic function in healthy older individuals. LA and left-ventricular (LV) volumes as well as strain rates were measured by high-resolution cardiac magnetic resonance imaging before and after flavanol intake. Results: Sixty-three participants (59% male) aged ≥ 55 years received either 500 mg cocoa flavanols (containing 80 mg (–)-epicatechin) originating from cocoa extract (n = 30) or a control (n = 33) twice daily for 30 days. Flavanol intake counteracted subclinical cardiac dysfunction, evidenced by a 12.6 ± 3.5% reduction in maximal LA volume (p = 0.0063) and LA volume index (p = 0.0067) and ∼4.4 ± 1.9% reduction in LV end-diastolic volume (LVEDV) (p = 0.049) and LVEDV index (p = 0.041). Flavanol intake did not influence strain, strain rate, and systolic function parameters, while the systolic blood pressure decreased by 7 mmHg [∼4.7 ± 1.9% (p = 0.04)]. Blood pressure dynamics, gender or age of participants in the intervention group were not associated with LA volumetric changes. Conclusion: We provide evidence that cocoa flavanol intake mitigates early changes of diastolic dysfunction by reversing left atrial and left ventricular remodeling, thus providing novel insights into the mechanisms behind the beneficial cardiovascular effect of flavanol intake. The clinical trial registry number is NCT 05782309 (https://clinicaltrials.gov).
Introduction
The impaired relaxation pattern of the left ventricle is a common feature of the aging heart, causing increased filling pressures and consecutive LA remodeling and enlargement.1 As these changes occur slowly and continuously, they are often not clinically visible in the early stages, and individuals remain asymptomatic. Clinical detection, especially of these incipient states, remains challenging.2 To date, there is no known clinical, pharmacological or dietary intervention capable of reversing impaired cardiac relaxation patterns or diastolic dysfunction,3 despite the possibility that up to 20% of individuals older than 45 years may experience some degree of diastolic dysfunction.4 However, as filling pressures increase even further and the affected individuals transition from diastolic dysfunction to heart failure with preserved ejection fraction (HFpEF), they start experiencing the full spectrum of symptoms, including reduced exercise capacity, dyspnea, and edema accumulation. In addition to reflecting relaxation pressures in the left heart, LA enlargement has been verified and validated as a prognostic marker for future cardiovascular events in individuals with and without cardiovascular diseases.5–7Dietary flavanols occur in various food groups, including fruits, tea, and cocoa-derived products.8 The “COcoa Supplement and Multivitamin Outcomes Study (COSMOS)”, a large-scale randomized clinical trial investigating the intake of cocoa flavanols from cocoa extract and multivitamins in over 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 individuals during a median follow-up of 3.6 years, showed a 27% reduction in cardiovascular death in the flavanol intake group.9 Due to the growing evidence for cardiometabolic benefits of flavanols, the Academy of Nutrition and Dietetics released an intake recommendation of 400–600 mg of flavanols daily.10 In fact, several vasoprotective properties, such as the improvement of endothelial dysfunction and blood pressure reduction by flavanol intake, are known.11,12 Previously, we found that cocoa flavanols intake protected radial arteries against intima hyperplasia after catheterization-induced injury,13 and improved cardiovascular fitness in healthy older individuals after a 30-day flavanol supplementation period.14 In the present work, we sought to assess other potential mechanisms behind the beneficial cardiovascular effects of flavanols and hypothesized that 30-day intake of the same cocoa flavanol regimen would mediate beneficial changes in left atrial and left ventricular volumes, as surrogate parameters associated with diastolic dysfunction, and assessed using high-resolution CMR imaging, the gold standard method to assess cardiac volumetry,15 in older, otherwise healthy individuals.
000 individuals during a median follow-up of 3.6 years, showed a 27% reduction in cardiovascular death in the flavanol intake group.9 Due to the growing evidence for cardiometabolic benefits of flavanols, the Academy of Nutrition and Dietetics released an intake recommendation of 400–600 mg of flavanols daily.10 In fact, several vasoprotective properties, such as the improvement of endothelial dysfunction and blood pressure reduction by flavanol intake, are known.11,12 Previously, we found that cocoa flavanols intake protected radial arteries against intima hyperplasia after catheterization-induced injury,13 and improved cardiovascular fitness in healthy older individuals after a 30-day flavanol supplementation period.14 In the present work, we sought to assess other potential mechanisms behind the beneficial cardiovascular effects of flavanols and hypothesized that 30-day intake of the same cocoa flavanol regimen would mediate beneficial changes in left atrial and left ventricular volumes, as surrogate parameters associated with diastolic dysfunction, and assessed using high-resolution CMR imaging, the gold standard method to assess cardiac volumetry,15 in older, otherwise healthy individuals.
Methods
Study design and population
The present study represents a prespecified substudy of the randomized, double-blinded (participants and researchers), placebo-controlled, parallel-group Healthy Aging Through Dietary Intervention trial (clinicaltrials.gov registration number NCT 05782309). The original trial was performed in the Department of Cardiology of the University Hospital of Duesseldorf, Germany. Participants aged between 55 and 79 years were recruited from the local community via advertising banners in the hallways of the University Hospital of Duesseldorf and Heinrich-Heine University Duesseldorf. Participants received a 30-day intake of either a cocoa flavanols extract (2 × 500 mg daily per os, containing 80 mg of (–)-epicatechin per intake) or a control treatment (2 capsules, 2× day per os). Cocoa flavanols were defined as the sum of flavanol monomers and procyanidins with a degree of polymerization up to 7 (DP1–7), according to the AOAC2020.05 method16 (SI Table S1). Flavanol monomers, including (–)-epicatechin, were assessed using AOAC2013.04![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 17 (SI Table S1). While cocoa may contain flavanols with a degree of polymerization greater than 7, these procyanidins represent a smaller fraction of all procyanidins.18 Theobromine (100 mg) and caffeine (30 mg) were included in equal amounts in both the intervention and control capsules to regulate for their influence on the study's endpoints. Their amount was lower than the proven one to induce cardiovascular effects.19,20 Screened participants were excluded from the trial if they presented any cardiovascular, neurological, orthopedic, or metabolic disease or had any long-term drug intake. Treated hypertension was not considered an exclusion criterion. The primary endpoint of the Healthy Aging Through Dietary Intervention trial were changes in cardiopulmonary exercise capacity induced by 30-day cocoa flavanol intake. The results have been previously published by us.14
17 (SI Table S1). While cocoa may contain flavanols with a degree of polymerization greater than 7, these procyanidins represent a smaller fraction of all procyanidins.18 Theobromine (100 mg) and caffeine (30 mg) were included in equal amounts in both the intervention and control capsules to regulate for their influence on the study's endpoints. Their amount was lower than the proven one to induce cardiovascular effects.19,20 Screened participants were excluded from the trial if they presented any cardiovascular, neurological, orthopedic, or metabolic disease or had any long-term drug intake. Treated hypertension was not considered an exclusion criterion. The primary endpoint of the Healthy Aging Through Dietary Intervention trial were changes in cardiopulmonary exercise capacity induced by 30-day cocoa flavanol intake. The results have been previously published by us.14
The current substudy analyzed changes in cardiac volumetry, as specified among the secondary endpoints of the Healthy Aging Through Dietary Intervention trial. The heart chamber volumes and diastolic cardiac function were assessed by high-resolution cardiac magnetic resonance (CMR) at baseline and after 30-day intake. The maximal left-atrial volume (LAmax) and the end-diastolic left-ventricular volume (LVEDV) and their indexes (LAVI and LVIEDV, respectively) normalized to body surface area (BSA) were defined as the primary endpoints of the current substudy. Systolic and diastolic blood pressures were defined as secondary endpoints of the present substudy. Upon enrollment, several baseline measurements were performed to warrant study eligibility. These included blood pressure, echocardiography, electrocardiography, physical examination, laboratory parameters electrolytes, blood count, and the HF parameter N-terminal pro b-type natriuretic peptide (NT-proBNP) via standardized venous punction from the cubital vein. All participants gave informed verbal and written consent before enrolment. The ethics committee of Heinrich-Heine University approved our study (Approval Number R5761R), which was carried out in accordance with the ethical guidelines of the 1975 Declaration of Helsinki. The study was previously registered on clinicaltrials.gov (NCT 05782309). Study enrolment started in January 2019, and the measurements were completed by February 2021.
The data underlying this article will be shared on reasonable request to the corresponding author.
Randomization and intervention
Participants were randomized to the flavanol or control group with a double-blinded design. Randomization and allocation have been previously described in detail.14 Briefly, the allocation was performed by an independent study nurse, and it was disclosed by telephone. In the flavanol group, capsule intake provided 1000 mg of cocoa flavanols containing 160 mg of (–)-epicatechin per day. The intake amount of flavanol was chosen based on a previous study, in which we showed that the maximum change in flow-mediated dilation (FMD) occurred at an intake amount of 820 mg flavanol, containing 112 mg (–)-epicatechin.21 Participants from the control group received a flavanol-free, nutrient-matched control regimen. The detailed capsules composition is shown in SI Table S1. The capsules were sealed in containers, on which alphabetical codes were written double-blinded. Intake was per os and twice daily (a total of 4 capsules per day) in both groups. Participants were instructed to ingest the capsules with meals (breakfast and dinner), as recommended in ref. 22. Mars, incorporated provided all capsules. Capsules containing cocoa flavanols were indistinguishable in appearance from the control capsules and were handed in non-transparent, sealed containers. This regimen was maintained for 30 days, and compliance was assessed by counting the left-over capsules and the documented intake directly by the subjects. Subjects were asked to continue their current lifestyle with the same level of activity and habitual diet until the follow-up date. The amount of flavanol intake within the habitual diet was not quantified. Measurements occurred before the first intake of test capsules (baseline) and one day after the last test capsules (follow-up). Unblinding was undertaken by an independent study nurse after the database release was approved and after the study was completed.Cardiac magnetic resonance
CMR was performed on a 1.5 T MRI System (Achieva, Philips, Best Netherlands) using a 5-channel phased array coil as described previously.23 After scout and reference scans, the functional and geometric assessment was performed using cine steady-state free precession images in standard long-axis geometries (two-, three- and four-chamber view) as well as in short-axis orientation with full ventricular coverage from base to apex (repetition time/echo time = 3.3/1.6 ms, flip angle = 60°, spatial resolution = 1.5 × 1.5 × 8 mm3, 50 phases, two slices per breath-hold).Dedicated software (Circle CVI 42, Circle Cardiovascular Imaging Inc., Calgary, AB, Canada) was used to automatically delineate ventricular and atrial contours and calculate volumes, strain, and strain rate. Volumes were further indexed to BSA (calculated automatically by the MRI system) to assess end-diastolic volume index (LVIEDV), end-systolic volume index (LVIESV) and to assess the LV systolic functional parameters stroke volume index (SVi), cardiac index, and ejection fraction (EF). Global longitudinal strain (GLS), early and late longitudinal diastolic strain rate (GLSRe, GLSRa), as well as early and late circumferential diastolic strain rate (GCSRe, GCSRa) were derived from the integrated feature tracking algorithm. No significant manual corrections to the automated tracking were necessary.
Pulse wave velocity
Arterial pulse wave velocity (PWV) analysis was performed using applanation tonometry (SphygmoCor®, AtCor Medical, Australia). PWV was assessed via ECG-synchronized applanation tonometry of the left common carotid and femoral arteries. Distances were measured with a measuring band (difference in distance, Δd), and transit times were calculated from the latency between the R-wave and pulse wave onset (difference in time, Δt). PWV was then defined as PWV = Δd/ΔT.Blood pressure measurements
Peripheral blood pressure was measured automatically before CMR with an appropriate medical device (boso medicus, BOSCH + SOHN GmbH u. Co. KG). The measurement was performed on both brachial arteries in a supine position after 5 minutes of rest. The arithmetical mean was used for analyses.Statistics
Data are presented as mean ± standard deviation (SD), unless otherwise stated. Baseline characteristics were compared either by unpaired t-test (normally distributed numerical data), Mann–Whitney U test (non-normally distributed numerical data), or Fisher's exact test (categorical data), as appropriate. Variables measured at baseline and follow-up were compared between the groups by repeated measurements two-way ANOVA with the Bonferroni post hoc test. To exclude an association between LA remodeling of the intervention group and gender, baseline blood pressure, and body-mass-index, we performed logistic regression analysis. We defined positive LA remodeling as a >15% reduction from initial LA volume, which has been previously used in clinical studies.24 The sample size was calculated to correspond to the observed effects in FMD by a similar flavanol regimen, in which 30 days of flavanols induced a 30% improvement in flow-mediated dilation.25 We used mean reference values for LAmax diameter according to Maceira et al.26 of 73 ± 15 (SD) ml and expected a decrease in the intervention group of 17.5%. Considering an alpha level of 5% and a power of 80%, we calculated a sample size of 42. Including a drop-out rate of 10%, the final sample size was set for 47 participants. Since this was within our sample size initially calculated for analyses of flavanol on VO2max, as the primary endpoint of the Healthy Aging Through Dietary Intervention trial,14 no supplementary participants needed to be included for the current analysis.P-Values <0.05 were considered statistically significant. Statistical analysis was performed using Prism 9 (GraphPad Software, San Diego, CA, USA).
Results
Baseline characteristics
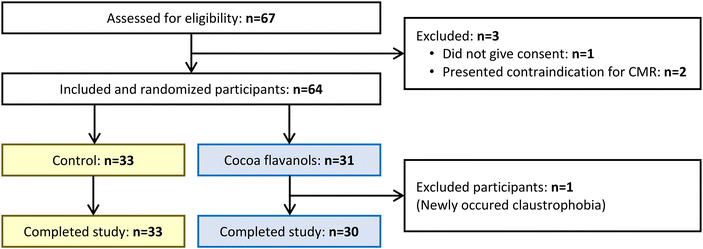
Sixty-seven participants adhered to the criteria for inclusion. One participant did not consent to the study; two had contraindications for CMR. The remaining 64 participants were included in the study. Both the flavanol and the control capsules were well tolerated. Following the intervention, one participant from the flavanol group developed claustrophobia from the first CMR examination and was excluded from the final analysis. All other participants who were included followed the predetermined protocol up to the follow-up and were included in the study's final analysis. The flow chart of the study is shown in Fig. 1. Baseline characteristics showed no significant differences for age (p = 0.95), BMI (p = 0.15), and BSA (p = 0.79) between groups. Detailed baseline characteristics are shown in Table 1.| Demographics | Control (n = 33) | Cocoa flavanols (n = 30) | p |
|---|---|---|---|
| Age, years | 62.9 ± 6.5 | 63.0 ± 4.4 | 0.95 |
| Male, n (%) | 18 (55) | 20 (63) | 0.61 |
| Height, cm | 173.9 ± 8.3 | 177.6 ± 9.4 | 0.11 |
| Weight, kg | 81.1 ± 12.6 | 80.6 ± 13.4 | 0.87 |
| Body mass index, kg m−2 | 26.7 ± 3.1 | 25.5 ± 3.6 | 0.15 |
| Body surface area, m2 | 1.98 ± 0.2 | 2.0 ± 0.2 | 0.79 |
| Systolic blood pressure, mmHg | 138.6 ± 16.0 | 137.9 ± 15.3 | 0.87 |
| Diastolic blood pressure, mmHg | 80.7 ± 9.7 | 80.3 ± 8.1 | 0.85 |
| Heart rate, 1 min−1 | 65.5 ± 10.1 | 62.7 ± 8.0 | 0.23 |
| Comorbidities | |||
| Type 2 diabetes mellitus, n (%) | 2 (6) | 1 (3) | >0.9999 |
| Hyperlipidemia, n (%) | 7 (21) | 5 (17) | 0.75 |
| Hypertension, n (%) | 14 (42) | 12 (40) | >0.9999 |
| Antihypertensive medication | |||
| ACEI or ARB, n (%) | 13 (39) | 11 (37) | >0.9999 |
| Beta-Blocker, n (%) | 2 (6) | 0 (0) | 0.49 |
| Calcium antagonists, n (%) | 3 (9) | 1 (3) | 0.61 |
| Thiazide-type diuretics, n (%) | 2 (6) | 0 (0) | 0.49 |
| Laboratory values | |||
| Hemoglobin, g dl−1 | 14.0 ± 0.8 | 13.6 ± 2.5 | 0.77 |
| eGFR, ml min−1/173 m2 | 86.3 ± 12.1 | 87.6 ± 14.4 | 0.72 |
| NT-proBNP, pg ml−1 | 84.3 ± 64.8 | 69.4 ± 54.5 | 0.25 |
Baseline cardiovascular function
CMR revealed similar LV systolic function at baseline between the two groups, mirrored by EF (65 ± 6.6% in the control versus 65.2 ± 7.1% in the flavanol group, p = 0.91), SV (94.7 ± 23.3 ml in the control versus 95.5 ± 17.6 ml in the flavanol group, p = 0.88) and CI (3.24 ± 0.78 l min−1 in the control versus 3.07 ± 0.56 l min−1 in the flavanol group, p = 0.34). In terms of afterload, blood pressure was comparable between groups (systolic BP: 137.2 ± 17.4 in the control versus 137.9 ± 15.3 mmHg in the flavanol group, p = 0.85; and diastolic BP: 80.7 ± 9.7 in the control versus 80.2 ± 8.1% in the flavanol group, p = 0.85). By applanation tonometry, we saw equivalent PWV of the two groups (9.0 ± 2.2 in the control versus 9.0 ± 2.2 m s−1 in the flavanol group, p = 0.64).Analysis of cardiac volumes showed similar LAmax (p = 0.46), LAVI (p = 0.32), LVEDV (p = 0.73), and LVIEDV (p = 0.76). Longitudinal SRe and circumferential SRe were comparable (p = 0.48 and p = 0.90). Altogether, these baseline cardiac characteristics mirrored well-balanced groups.
Cocoa flavanols improve markers of diastolic function
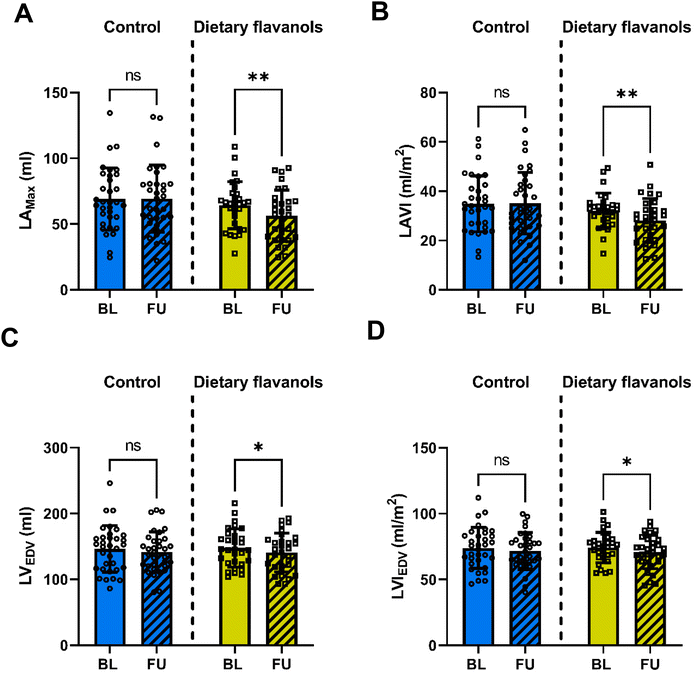
LAmax decreased by ∼12.6 ± 3.5% in the flavanol group (64.3 ± 17.9 ml at baseline versus 56.2 ± 19.5 ml at follow-up, Fig. 2A, p = 0.0063). Similarly, flavanol intake reduced LAVI by ∼12.6 ± 3.5% (SEM) (32.1 ± 7.2 ml m−2 at baseline versus 28.1 ± 8.9 ml m−2 at follow-up, Fig. 2B, p = 0.0067). The control regiment did not affect LAmax (69 ± 23.7 ml at baseline versus 69.3 ± 25.5 ml at follow-up, Fig. 2A, p = 0.99) or LAVI (34.8 ± 11.3 ml m−2 at baseline versus 35.1 ± 12.5 ml m−2 at follow-up, Fig. 2B, p = 0.97). In line with atrial changes, participants from the flavanol group had a lower LVEDV by ∼4.4 ± 1.9% (SEM) after intervention (147.8 ± 29 ml at baseline versus 141 ± 29.5 ml at follow-up, Fig. 2C, p = 0.0499) and LVIEDV by ∼4.4 ± 1.9% (SEM) (74.2 ± 11.5 ml at baseline versus 70.9 ± 15.6 ml at follow-up, Fig. 2D, p = 0.0413). The control regiment did not affect these parameters (LVEDV: 146.4 ± 35.6 at baseline versus 141.5 ± 30.9 ml at follow-up, Fig. 2C, p = 0.17; LVIEDV:74 ± 15.6 ml m−2 at baseline versus 71.7 ± 14 ml m−2 at follow-up, Fig. 2D, p = 0.17). Neither of the groups showed changes in longitudinal and circumferential SRe or SRa (Table 2).| Control (n = 33) | Cocoa flavanols (n = 30) | p(Δ) | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Baseline | +30 d | p | Δ [95% CI] | Baseline | +30 d | p | Δ [95% CI] | ||
| SBP, mmHg | 138.6 ± 16.0 | 133.0 ± 16.8 | 0.06 | −5.6 [−10.1; −1.1] | 137.9 ± 15.3 | 131.4 ± 13.5 | 0.04 | −6.5 [−12.6; −0.42] | 0.74 |
| DBP, mmHg | 80.7 ± 9.7 | 78.5 ± 7.2 | 0.19 | −2.1 [−5.0; 0.8] | 80.6 ± 8.0 | 77.2 ± 8.1 | 0.03 | −3.4 [−5.8; −1.1] | 0.49 |
| NT-proBNP, pg ml−1 | 84.4 ± 65.7 | 97.1 ± 96.3 | 0.14 | 13.7 [−5.0; 32.4] | 69.4 ± 54.5 | 68.8 ± 58.0 | 0.89 | 0.9 [−12.0; 13.8] | 0.26 |
| CMR | |||||||||
| EF, % | 65.0 ± 6.6 | 62.4 ± 5.3 | 0.02 | −2.5 [−4.6; 0.5] | 65.2 ± 7.1 | 65.0 ± 5.2 | 0.98 | −0.2 [−2.2; 1.8] | 0.09 |
| SV, ml | 94.7 ± 23.7 | 88.2 ± 19.9 | 0.02 | −6.5 [−11.0; 2.0] | 95.5 ± 17.6 | 91.1 ± 18.3 | 0.18 | −4.4 [−10.0; 1.3] | 0.55 |
| SVI, ml m−2 BSA | 47.8 ± 10.1 | 44.6 ± 8.9 | 0.02 | −3.2 [−5.4; 0.9] | 48.0 ± 6.98 | 45.9 ± 8.0 | 0.16 | −2.1 [−4.7; 0.5] | 0.54 |
| LA Max Volume, ml | 69.0 ± 23.7 | 69.3 ± 25.5 | 0.99 | 0.34 [−4.95; 5.64] | 64.3 ± 17.9 | 56.2 ± 19.5 | 0.0063 | −8.01 [−13.16; −2.86] | 0.03 |
| LAVI, ml m−2 BSA | 34.8 ± 11.3 | 35.1 ± 12.5 | 0.97 | 0.25 [−2.42; 2.91] | 32.1 ± 7.2 | 28.1 ± 8.9 | 0.0067 | −3.93 [−6.35; −1.51] | 0.02 |
| GLS, % | −15.7 ± 2.1 | −15.0 ± 2.3 | 0.07 | 0.77 [−0.04; 1.58] | −15.0 ± 1.9 | −14.7 ± 2.2 | 0.75 | 0.26 [−0.44; 0.96] | 0.34 |
| GLSRe, 1 s−1 | 0.71 ± 0.20 | 0.65 ± 0.23 | 0.22 | −0.06 [−0.14; 0.02] | 0.67 ± 0.19 | 0.67 ± 0.22 | 0.97 | −0.006 [−0.08; 0.06] | 0.33 |
| GLSRa, 1 s−1 | 0.72 ± 0.21 | 0.66 ± 0.23 | 0.29 | −0.05 [−0.13; 0.02] | 0.70 ± 0.19 | 0.66 ± 0.16 | 0.40 | −0.05 [−0.12; 0.03] | 0.92 |
Cocoa flavanol intake did not affect LV systolic function. This was evidenced by EF (65.2 ± 7.1% at baseline versus 65.0 ± 5.2% at follow-up, Table 2, p = 0.98) and SV (95.5 ± 17.6 ml at baseline versus 91.1 ± 18.3 ml at follow-up, Table 2, p = 0.18).
Cocoa flavanols improve blood pressure
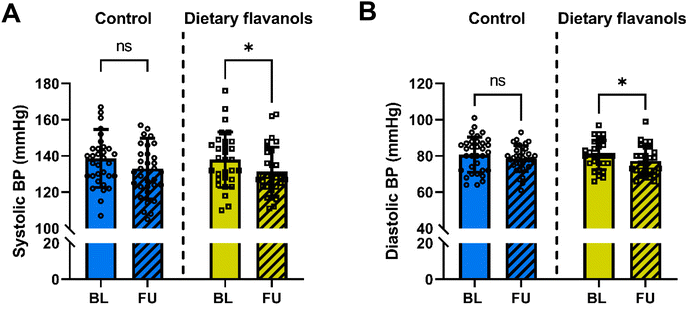
One month of flavanol administration led to a significant decrease in both systolic and diastolic blood pressure (Fig. 3): systolic blood pressure was reduced by 4.7 ± 1.9% (137.9 ± 15.3 mmHg at baseline versus 131.4 ± 13.5 mmHg at follow-up, p = 0.034) and diastolic blood pressure by 3.4 ± 1.6% (80.2 ± 8.1 mmHg at baseline versus 77.5 ± 8.1 mmHg at follow-up, p = 0.02).Gender, age and baseline blood pressure are not associated with positive LA remodeling
Neither elevated systolic blood pressure at baseline >130 mmHg (OR 2.6, 95% CI 0.49–20.6, p = 0.29), nor the absolute differences in blood pressure evoked by cocoa flavanols (OR per % reduction: 1.02, 95% CI 0.94–1.1, p = 0.62) were associated with positive LA remodeling. Similarly, gender, age, or body mass index were not associated with a higher degree of LA remodeling in a logistic regression model (OR for male participants: 0.52, 95% CI 0.31–2.45, p = 0.42; OR for age [per year]: 1.04, 95% CI 0.87–1.25, p = 0.66; OR for BMI [per unit]: 1.14, 95% CI 0.91–1.46, p = 0.26, SI Table S2).Discussion
Over a 30-day period, daily intake of 1000 mg of cocoa flavanol extract, containing 160 mg (–)-epicatechin, promoted cardiovascular health by reversing left atrial and left ventricular enlargement in healthy older individuals. The LA volume, whose enlargement was previously recognized as an age-related, subclinical manifestation of diastolic dysfunction, was reduced. Our present study is the first to provide a non-pharmacological, non-operative approach mediating beneficial changes in ventricular volumetry in healthy individuals. These outcomes were corroborated through high-resolution cine steady-state free precession CMR imaging, regarded as the gold standard for cardiac volumetry.15In a previous study, we demonstrated that the same cocoa flavanol intake regimen led to improvements in cardiorespiratory fitness in healthy older adults, particularly through increases in peak oxygen consumption and maximum exercise capacity.14 Given that diastolic dysfunction has been previously associated with reduced exercise capacity – including lower peak VO2 and tidal volume,27,28 the present findings may offer a mechanistic explanation for the previously observed functional benefits. However, further research is required to confirm and better understand this potential link. Consistent with our prior studies29,30 cocoa flavanol intake induced favorable changes in the systemic circulation, leading to reductions in systolic and diastolic blood pressure, which may have interacted with the observed cardiac alterations. However, there were no independent associations between baseline systolic blood pressure or its dynamics and the positive left atrial remodeling. Improvement in endothelial function, as measured by FMD, has been documented after both acute and chronic flavanol intake, summarized in detail,31 including in our previous trial using the same regimen.14 Notably, lower FMD has been associated with increased left-heart volumes.32 Endothelial dysfunction may theoretically contribute to increased cardiac afterload and pressure or volume overload, thereby promoting left atrial and ventricular remodeling. Taken together, these findings suggest that cocoa flavanol intake can potentially reverse early signs of diastolic dysfunction. Although speculative, it is possible that progression towards symptomatic diastolic dysfunction or the manifestation of heart failure with preserved ejection fraction could be delayed. The flavanol effects were independent of gender or systemic blood pressure at baseline.
The COSMOS trial, as the first large-scale, randomized-controlled trial to investigate flavanol intake in relation to cardiovascular disease and cardiovascular mortality,33 showed a reduction in cardiovascular death by ∼27%.9 The test material used in COSMOS consisted of a cocoa extract providing 500 mg of cocoa flavanols, including 80 mg of (–)-epicatechin, which is the same to the intervention used in the present study. The underlying mechanisms in terms of mortality reduction observed in COSMOS remain unknown. However, COSMOS recorded cardiovascular diseases with high mortality and morbidity, such as acute myocardial infarction, stroke, and the need for revascularizations of various arteries.9 Our current study might augment knowledge by providing insight into the cardiovascular benefits cocoa flavanol intake in age-related incipient dysfunctions.
Beyond the diastolic component, LA enlargement has been shown to be an independent long-term predictor of cardiovascular events in the general population.34 In our study cohort, the average LAVI of 34.8 ± 11.3 ml m−2 in the control and 32.1 ± 7.2 ml m−2 in the flavanol group was well situated within the standard physiological ranges of 17–59 in men and 17–61 ml m−2 in women.35 LA enlargement is often observed in individuals with cardiovascular diseases5,36,37 and its reduction to date has been proven so far only by drug intake,38,39 medical interventions40,41 or surgery42 but not by dietary interventions. We found a LA volume reduction achieved over one month of flavanol intake of ∼13%, uncovering the first dietary intervention to reduce LA volumes in healthy older individuals.
The daily intake of 1000 mg flavanol in our study exceeded the recommended intake range outlined by the Academy of Nutrition and Dietetics (400–600 mg daily)10 and the amount employed in the COSMOS trial (500 mg daily, containing 80 mg (–)-epicatechin).33 At the same time, whilst the COSMOS trial investigated long-term effects of flavanols intake from cocoa extract, mirrored by the median follow-up time of 3.6 years,9 we tested specific effects in a shorter timeframe, thus opting for a higher intake amount. This amount was selected based on a preliminary study from our laboratory, which indicated a dose-dependent effect on FMD, with the highest FMD response observed at approximately 820 mg of cocoa flavanols.21 Thus, it is possible that lower intake amounts of cocoa flavanols but consumed for much longer periods could result in similar beneficial health effects as reported here and thus, help to provide mechanistic insights behind the long-term cardiovascular effects of flavanols as shown in COSMOS and supported by the Academy of Nutrition and Dietetics recommendations.10 In our study, all test materials were well tolerated. This is in line with previously reported safety margins for flavanol consumption.22
No changes in LV systolic parameters, GLS or diastolic strain rate parameters were visible by flavanol intake. In an earlier clinical study, consuming 1 g of flavanols daily ameliorated chronic heart failure in patients with reduced EF, reflected by a reduction in NT-proBNP levels.43 Since our participants all showed normal LV systolic function at baseline, the effect of LV function improvement might be only in reduced systolic function before starting flavanol intake, however, further research is needed to confirm or deny this hypothesis.
Vascular effects evoked by flavanol were recorded as secondary endpoints of the present study. In the intervention group, systolic blood pressure was decreased by ∼6.5 mmHg. A recent meta-analysis summarized the effects of flavanols on blood pressure and found a slight but significant reduction in preponderantly healthy subjects, especially if the analyzed collective were prehypertensive or hypertensive.44 Our data align with this finding, whereas the higher amount of administered flavanol might explain the larger effect size. The potential for blood pressure reduction observed in our healthy cohort is similar to that achieved with commonly used antihypertensive medications, although flavanols cannot be considered a pharmacological agent.
Several limitations must be addressed. Most importantly, our study's first objective was to assess flavanol-mediated changes to diastolic function in healthy older individuals with normal systolic function. Data on CMR-derived strain analysis, especially diastolic strain rate, is rare. Effect sizes can only be estimated, and we could not define any cut-off criteria for pre-clinical diastolic dysfunction. In addition, due to our purely descriptive analysis, we could only show a reduction in LA volumes without establishing the physiological and biochemical mechanisms behind it. Furthermore, since this study represents one of the first successful interventions to reduce LA volume. Its effects on future CV events need further investigation for validation. Lastly, several participants included in the current analysis were part of our previously published RCT addressing the effects of flavanols on exercise capacity.14 As all participants were older than 55 and of Western-European descent, generalizability of our findings to younger people or other ethnic groups needs to be further investigated.
In an aging population worldwide, preventing cardiovascular disease and delaying disease onset improves health span and quality of life. Therefore, any intervention to preserve cardiovascular health is warranted. Healthy older individuals who consumed 1000 mg of cocoa flavanols over one month showed benefits in surrogate parameters for cardiac relaxation and peripheral blood pressure intake. Our data add to better understand the contributing mechanisms behind the growing evidence of flavanol's beneficial cardiovascular effects. Further investigations will be needed to determine if these changes take place when smaller amounts of flavanols or those containing lower amounts of (–)-epicatechin are consumed in longer time periods.
Conclusion
We provide evidence that 30-day flavanol intake of 1000 mg day−1, containing 160 mg of (–)-epicatechin, mitigates early changes of diastolic dysfunction by reversing left atrial and left ventricular remodeling in older, healthy individuals. These findings suggest a potential protective effect of flavanols against age-related cardiac changes and open the field for future research in flavanol and cardiac interactions.Author contributions
D. A. D.: data curation, writing – original draft; M. G.: data curation, funding acquisition, validation, writing – review & editing; N. O., N. K. & He. S.: investigation; J. O. & Ha. S.: methodology, resources; methodology, resources; C. Q., F. B., C. H. & R. S.: data curation; C. J. & M. K.: conceptualization, funding acquisition, project administration, supervision, writing – review & editing; R. E.: conceptualization, funding acquisition, methodology, project administration, writing – review & editing.Abbreviations
| BMI | Body mass index |
| BSA | Body surface area |
| CI | Cardiac index |
| CMR | Cardiac magnetic resonance |
| COSMOS | COcoa Supplement and Multivitamin Outcomes Study |
| EF | Ejection fraction |
| eNOS | Endothelial NO synthase |
| FMD | Flow-mediated vasodilation |
| GCSRe | Early circumferential diastolic strain rate |
| GCSRa | Late circumferential diastolic strain rate |
| GLS | Global longitudinal strain |
| GLSRe | Early longitudinal diastolic strain rate |
| GLSRa | Late longitudinal diastolic strain rate |
| HFpEF | Heart failure with persistent ejection fraction |
| LA | Left-atrial |
| LAmax volume | Maximum left-atrial volume |
| LAVI | Left-atrial volume index |
| LV | Left ventricle |
| LVEDV | End-distolic left-ventricular volume |
| LVIEDV | End-diastolic left-ventricular volume index |
| PWV | Pulse wave velocity |
| SD | Standard deviation |
| SEM | Standard error of the mean |
| SR | Strain rate |
| SV(I) | Stroke volume (index) |
Conflicts of interest
H. S. and J. O. are employees of Mars Edge (a segment of Mars, Incorporated). Mars Edge is dedicated to human health through nutrition, including flavanol research and flavanol-related activities. All other authors declare no conflicts of interest.Data availability
All data supporting the findings of this study are presented in the figures and tables. The raw data are not publicly available due to confidentiality restrictions but may be provided by the corresponding author upon reasonable request.Supplementary information (SI) is available. See DOI: https://doi.org/10.1039/d5fo02589c.
Acknowledgements
This study initiated by M. K., M. G. and R. E. was partially funded by an investigator-initiated grant from Mars Edge. Mars Edge is a segment of Mars Incorporated, dedicated to nutrition research and products. Mars Edge had no influence on study design, data collection, and analysis. The graphical abstract was created in BioRender (biorender.com).References
- A. Rossi, M. Gheorghiade, F. Triposkiadis, S. D. Solomon, B. Pieske and J. Butler, Left atrium in heart failure with preserved ejection fraction: structure, function, and significance, Circ.:Heart Failure, 2014, 7, 1042–1049 Search PubMed.
- L. Thomas, T. H. Marwick, B. A. Popescu, E. Donal and L. P. Badano, Left Atrial Structure and Function, and Left Ventricular Diastolic Dysfunction: JACC State-of-the-Art Review, J. Am. Coll. Cardiol., 2019, 73, 1961–1977 CrossRef.
- E. M. Jeong and S. C. Dudley, Jr., Diastolic dysfunction, Circ. J., 2015, 79, 470–477 CrossRef.
- M. M. Redfield, S. J. Jacobsen, J. C. Burnett, Jr., D. W. Mahoney, K. R. Bailey and R. J. Rodeheffer, Burden of systolic and diastolic ventricular dysfunction in the community: appreciating the scope of the heart failure epidemic, JAMA, 2003, 289, 194–202 CrossRef PubMed.
- L. Thomas and W. P. Abhayaratna, Left Atrial Reverse Remodeling: Mechanisms, Evaluation, and Clinical Significance, JACC: Cardiovasc. Imaging, 2017, 10, 65–77 Search PubMed.
- Z. Hao, G. Xu, M. Yuan, Y. Sun, R. Tan, Y. Liu and Y.-L. Xia, The predictive value of changes in left atrial volume index for rehospitalization in heart failure with preserved ejection fraction, Clin. Cardiol., 2023, 46, 151–158 CrossRef PubMed.
- B. D. Hoit, Left atrial size and function: role in prognosis, J. Am. Coll. Cardiol., 2014, 63, 493–505 CrossRef PubMed.
- J. Boyer and R. H. Liu, Apple phytochemicals and their health benefits, Nutr. J., 2004, 3, 5 CrossRef PubMed.
- H. D. Sesso, J. E. Manson, A. K. Aragaki, P. M. Rist, L. G. Johnson, G. Friedenberg, T. Copeland, A. Clar, S. Mora, M. V. Moorthy, A. Sarkissian, W. R. Carrick, G. L. Anderson and C. R. Group, Effect of cocoa flavanol supplementation for the prevention of cardiovascular disease events: the COcoa Supplement and Multivitamin Outcomes Study (COSMOS) randomized clinical trial, Am. J. Clin. Nutr., 2022, 115, 1490–1500 CrossRef.
- K. M. Crowe-White, L. W. Evans, G. G. C. Kuhnle, D. Milenkovic, K. Stote, T. Wallace, D. Handu and K. E. Senkus, Flavan-3-ols and Cardiometabolic Health: First Ever Dietary Bioactive Guideline, Adv. Nutr., 2022, 13, 2070–2083 CrossRef.
- H. Schroeter, C. Heiss, J. Balzer, P. Kleinbongard, C. L. Keen, N. K. Hollenberg, H. Sies, C. Kwik-Uribe, H. H. Schmitz and M. Kelm, (-)-Epicatechin mediates beneficial effects of flavanol-rich cocoa on vascular function in humans, Proc. Natl. Acad. Sci. U. S. A., 2006, 103, 1024–1029 CrossRef PubMed.
- C. Heiss, P. Kleinbongard, A. Dejam, S. Perré, H. Schroeter, H. Sies and M. Kelm, Acute consumption of flavanol-rich cocoa and the reversal of endothelial dysfunction in smokers, J. Am. Coll. Cardiol., 2005, 46, 1276–1283 CrossRef.
- M. Gröne, M. Schillings, D. Duse, N. Kramser, C. Quast, C. Heiss, R. Sansone, C. Jung, M. Kelm and R. Erkens, Cocoa flavanol supplementation preserves early and late radial artery function after transradial catheterization, Food Funct., 2023, 14, 4824–4835 RSC.
- M. Groene, D. A. Duse, N. Kramser, N. Ophoff, H. Schweers, C. Quast, F. Voss, R. Sansone, C. Heiss, C. Jung, M. Kelm and R. Erkens, Cocoa flavanols improve peakVO2 and exercise capacity in a randomized double blinded clinical trial in healthy elderly, Food Funct., 2023, 7562–7573 RSC.
- R. Krishnamurthy, B. Cheong and R. Muthupillai, Tools for cardiovascular magnetic resonance imaging, Cardiovasc. Diagn. Ther., 2014, 4, 104–125 Search PubMed.
- U. Bussy, H. You and C. Kwik-Uribe, Determination of Cocoa Flavanols and Procyanidins (by Degree of Polymerization DP1–7) in Cocoa-Based Products by Hydrophilic Interaction Chromatography Coupled With Fluorescence Detection: Collaborative Study, J. AOAC Int., 2022, 105, 1060–1068 CrossRef.
- P. Machonis, M. Jones, B. Schaneberg, C. Kwik-Uribe and D. Dowell, Method for the determination of catechin and epicatechin enantiomers in cocoa-based ingredients and products by high-performance liquid chromatography: First Action 2013.04, J. AOAC Int., 2014, 97, 506–509 CrossRef PubMed.
- U. Bussy, B. R. May, Y. Olanrewaju, G. Hewitt, N. Anderson, A. Crozier, J. I. Ottaviani and C. Kwik-Uribe, Reliable, accessible and transferable method for the quantification of flavanols and procyanidins in foodstuffs and dietary supplements, Food Funct., 2020, 11, 131–138 RSC.
- B. van den Bogaard, R. Draijer, B. E. Westerhof, A. H. van den Meiracker, G. A. van Montfrans and B. J. van den Born, Effects on peripheral and central blood pressure of cocoa with natural or high-dose theobromine: a randomized, double-blind crossover trial, Hypertension, 2010, 56, 839–846 CrossRef CAS.
- S. Khiali, A. Agabalazadeh, H. Sahrai, H. Bannazadeh Baghi, G. Rahbari Banaeian and T. Entezari-Maleki, Effect of Caffeine Consumption on Cardiovascular Disease: An Updated Review, Pharm. Med., 2023, 37, 139–151 CrossRef PubMed.
- R. Sansone, J. I. Ottaviani, A. Rodriguez-Mateos, Y. Heinen, D. Noske, J. P. Spencer, A. Crozier, M. W. Merx, M. Kelm, H. Schroeter and C. Heiss, Methylxanthines enhance the effects of cocoa flavanols on cardiovascular function: randomized, double-masked controlled studies, Am. J. Clin. Nutr., 2017, 105, 352–360 CrossRef CAS PubMed.
- J. I. Ottaviani, M. Balz, J. Kimball, J. L. Ensunsa, R. Fong, T. Y. Momma, C. Kwik-Uribe, H. Schroeter and C. L. Keen, Safety and efficacy of cocoa flavanol intake in healthy adults: a randomized, controlled, double-masked trial, Am. J. Clin. Nutr., 2015, 102, 1425–1435 CrossRef CAS.
- M. Gastl, A. Gotschy, M. Polacin, V. Vishnevskiy, D. Meyer, J. Sokolska, F. C. Tanner, H. Alkadhi, S. Kozerke and R. Manka, Determinants of myocardial function characterized by CMR-derived strain parameters in left ventricular non-compaction cardiomyopathy, Sci. Rep., 2019, 9, 15882 CrossRef.
- R. M. Inciardi, A. Bonelli, T. Biering-Sorensen, M. Cameli, M. Pagnesi, C. M. Lombardi, S. D. Solomon and M. Metra, Left atrial disease and left atrial reverse remodelling across different stages of heart failure development and progression: a new target for prevention and treatment, Eur. J. Heart Failure, 2022, 24, 959–975 CrossRef.
- J. Balzer, T. Rassaf, C. Heiss, P. Kleinbongard, T. Lauer, M. Merx, N. Heussen, H. B. Gross, C. L. Keen, H. Schroeter and M. Kelm, Sustained Benefits in Vascular Function Through Flavanol-Containing Cocoa in Medicated Diabetic Patients: A Double-Masked, Randomized, Controlled Trial, J. Am. Coll. Cardiol., 2008, 51, 2141–2149 CrossRef CAS PubMed.
- A. M. Maceira, J. Cosín-Sales, M. Roughton, S. K. Prasad and D. J. Pennell, Reference left atrial dimensions and volumes by steady state free precession cardiovascular magnetic resonance, J. Cardiovasc. Magn. Reson., 2010, 12, 65 CrossRef.
- A. M. Alonso-Gómez, L. Tojal Sierra, E. Fortuny Frau, L. Goicolea Güemez, A. Aboitiz Uribarri, M. P. Portillo, E. Toledo, H. Schröder, J. Salas-Salvadó and F. Arós Borau, Diastolic dysfunction and exercise capacity in patients with metabolic syndrome and overweight/obesity, Int. J. Cardiol. Heart Vasc., 2019, 22, 67–72 Search PubMed.
- A. L. Arruda, P. A. Pellikka, T. P. Olson and B. D. Johnson, Exercise capacity, breathing pattern, and gas exchange during exercise for patients with isolated diastolic dysfunction, J. Am. Soc. Echocardiogr., 2007, 20, 838–846 CrossRef PubMed.
- C. Heiss, R. Sansone, H. Karimi, M. Krabbe, D. Schuler, A. Rodriguez-Mateos, T. Kraemer, M. M. Cortese-Krott, G. G. Kuhnle, J. P. Spencer, H. Schroeter, M. W. Merx and M. Kelm, Impact of cocoa flavanol intake on age-dependent vascular stiffness in healthy men: a randomized, controlled, double-masked trial, Age, 2015, 37, 9794 CrossRef PubMed.
- R. Sansone, A. Rodriguez-Mateos, J. Heuel, D. Falk, D. Schuler, R. Wagstaff, G. G. Kuhnle, J. P. Spencer, H. Schroeter, M. W. Merx, M. Kelm and C. Heiss, Cocoa flavanol intake improves endothelial function and Framingham Risk Score in healthy men and women: a randomised, controlled, double-masked trial: the Flaviola Health Study, Br. J. Nutr., 2015, 114, 1246–1255 CrossRef CAS PubMed.
- Y. Sun, D. Zimmermann, C. A. De Castro and L. Actis-Goretta, Dose-response relationship between cocoa flavanols and human endothelial function: a systematic review and meta-analysis of randomized trials, Food Funct., 2019, 10, 6322–6330 RSC.
- J. Z. Xu, S. Y. Wu, Y. Q. Yan, Y. S. Xie, Y. R. Ren, Z. F. Yin, C. Z. Shi and C. Q. Wang, Left atrial diameter, flow-mediated dilation of brachial artery and target organ damage in Chinese patients with hypertension, J. Hum. Hypertens., 2012, 26, 41–47 CAS.
- P. M. Rist, H. D. Sesso, L. G. Johnson, A. K. Aragaki, L. Wang, S. Rautiainen, A. Hazra, D. K. Tobias, M. S. LeBoff, H. Schroeter, G. Friedenberg, T. Copeland, A. Clar, L. F. Tinker, R. P. Hunt, S. S. Bassuk, A. Sarkissian, D. C. Smith, E. Pereira, W. R. Carrick, E. S. Wion, J. Schoenberg, G. L. Anderson and J. E. Manson, Design and baseline characteristics of participants in the COcoa Supplement and Multivitamin Outcomes Study (COSMOS), Contemp. Clin. Trials, 2022, 116, 106728 CrossRef PubMed.
- M. Bombelli, R. Facchetti, C. Cuspidi, P. Villa, D. Dozio, G. Brambilla, G. Grassi and G. Mancia, Prognostic significance of left atrial enlargement in a general population: results of the PAMELA study, Hypertension, 2014, 64, 1205–1211 CrossRef CAS PubMed.
- N. Kawel-Boehm, S. J. Hetzel, B. Ambale-Venkatesh, G. Captur, C. J. Francois, M. Jerosch-Herold, M. Salerno, S. D. Teague, E. Valsangiacomo-Buechel, R. J. van der Geest and D. A. Bluemke, Reference ranges (“normal values”) for cardiovascular magnetic resonance (CMR) in adults and children: 2020 update, J. Cardiovasc. Magn. Reson., 2020, 22, 87 CrossRef.
- R. M. Inciardi, R. P. Giugliano, B. Claggett, D. K. Gupta, A. Chandra, C. T. Ruff, E. M. Antman, M. F. Mercuri, M. A. Grosso, E. Braunwald and S. D. Solomon, Left atrial structure and function and the risk of death or heart failure in atrial fibrillation, Eur. J. Heart Failure, 2019, 21, 1571–1579 CrossRef CAS PubMed.
- M. R. Zile, J. S. Gottdiener, S. J. Hetzel, J. J. McMurray, M. Komajda, R. McKelvie, C. F. Baicu, B. M. Massie and P. E. Carson, Prevalence and significance of alterations in cardiac structure and function in patients with heart failure and a preserved ejection fraction, Circulation, 2011, 124, 2491–2501 CrossRef PubMed.
- T. S. Tsang, M. E. Barnes, W. P. Abhayaratna, S. S. Cha, B. J. Gersh, A. P. Langins, T. D. Green, K. R. Bailey, Y. Miyasaka and J. B. Seward, Effects of quinapril on left atrial structural remodeling and arterial stiffness, Am. J. Cardiol., 2006, 97, 916–920 CrossRef CAS PubMed.
- A. V. Mattioli, M. Zennaro, S. Bonatti, L. Bonetti and G. Mattioli, Regression of left ventricular hypertrophy and improvement of diastolic function in hypertensive patients treated with telmisartan, Int. J. Cardiol., 2004, 97, 383–388 CrossRef CAS PubMed.
- L. F. Tops, V. Delgado, M. Bertini, N. A. Marsan, D. W. Den Uijl, S. A. Trines, K. Zeppenfeld, E. Holman, M. J. Schalij and J. J. Bax, Left atrial strain predicts reverse remodeling after catheter ablation for atrial fibrillation, J. Am. Coll. Cardiol., 2011, 57, 324–331 CrossRef.
- P. Reant, S. Lafitte, P. Jaïs, K. Serri, R. Weerasooriya, M. Hocini, X. Pillois, J. Clementy, M. Haïssaguerre and R. Roudaut, Reverse remodeling of the left cardiac chambers after catheter ablation after 1 year in a series of patients with isolated atrial fibrillation, Circulation, 2005, 112, 2896–2903 CrossRef.
- F. Antonini-Canterin, C. C. Beladan, B. A. Popescu, C. Ginghina, A. C. Popescu, R. Piazza, E. Leiballi, B. Zingone and G. L. Nicolosi, Left atrial remodelling early after mitral valve repair for degenerative mitral regurgitation, Heart, 2008, 94, 759–764 CrossRef CAS PubMed.
- R. De Palma, I. Sotto, E. G. Wood, N. Q. Khan, J. Butler, A. Johnston, M. T. Rothman and R. Corder, Cocoa flavanols reduce N-terminal pro-B-type natriuretic peptide in patients with chronic heart failure, ESC Heart Failure, 2016, 3, 97–106 CrossRef PubMed.
- K. Ried, P. Fakler and N. P. Stocks, Effect of cocoa on blood pressure, Cochrane Database Syst. Rev., 2017, 4, Cd008893 Search PubMed.
Footnote |
| † These authors contributed equally. |
| This journal is © The Royal Society of Chemistry 2025 |