Open Access Article
Open Access ArticleBlackthorn juice from central Italy exhibits selective anticancer activity by inducing apoptosis and autophagy in gastric carcinoma cells
Matteo
Micucci†
 a,
Giovanni
Caprioli†
a,
Giovanni
Caprioli†
 b,
Ilaria
Versari
c,
Laura
Acquaticci
b,
Gianni
Sagratini
b,
Federico
Gianfanti
b,
Ilaria
Versari
c,
Laura
Acquaticci
b,
Gianni
Sagratini
b,
Federico
Gianfanti
 *a,
Francesco
Onesimo
a,
Irene
Faenza
c,
Sabrina
Burattini
a,
Michele
Mari
*a,
Francesco
Onesimo
a,
Irene
Faenza
c,
Sabrina
Burattini
a,
Michele
Mari
 a,
Michele
Retini
a,
Michele
Retini
 a,
Riham
Osman
a,
Sara
Salucci†
*c and
Michela
Battistelli†
a,
Riham
Osman
a,
Sara
Salucci†
*c and
Michela
Battistelli†
 a
a
aDepartment of Biomolecular Sciences, University of Urbino Carlo Bo, 61029 Urbino, Italy. E-mail: federico.gianfanti@uniurb.it
bChemistry Interdisciplinary Project (ChIP), School of Pharmacy, University of Camerino, 62032 Camerino, Italy
cDepartment of Biomedical and NeuroMotor Sciences, University of Bologna, 40126 Bologna, Italy. E-mail: sara.salucci@unibo.it
First published on 7th October 2025
Abstract
Blackthorn (Prunus spinosa L.) juice, a polyphenol-rich blend derived from the Marche region of Italy, was chemically characterized and evaluated for its selective effects against gastric cancer cells. The juice contained a mix of polyphenols, including neochlorogenic acid, cyanidin-3-glucoside, rutin, and smaller molecules – such as gallic, caffeic, and vanillic acids – characterized by high absorption rates. In vitro, the juice significantly reduced the viability of AGS and KATO III gastric cancer cells, while sparing non-tumorigenic GES-1 cells. In malignant cells, the juice activated Caspase-3, upregulated Bax, and downregulated Bcl-xL, while also modulating autophagy by increasing LC3B-II levels and decreasing Beclin-1. Mitochondrial impairment was confirmed by cardiolipin loss and cytochrome c release into the cytoplasm. These findings support the potential of Prunus spinosa L. juice as a selective, innovative functional beverage for gastric cancer prevention, acting beyond nutrition but before the need for medical intervention.
1. Introduction
Gastric carcinoma remains a persistent global health challenge, characterized by marked geographical variations in both incidence and burden. In 2020, it accounted for over one million new cases and nearly 770 000 deaths worldwide, ranking as the fifth most diagnosed cancer and the fourth leading cause of cancer-related mortality globally.1 Within the EU-27 alone, gastric cancer ranked as the tenth most diagnosed malignancy and the seventh leading cause of cancer-related death, accounting for approximately 100 000 new cases and 75 000 deaths in 2021.2 In Italy, gastric cancer remains among the ten most prevalent malignancies, particularly affecting older populations.3 The pathogenesis of gastric cancer is complex and multifactorial, involving genetic susceptibility, chronic inflammation, often driven by Helicobacter pylori infection, and modifiable environmental exposures. Among these, dietary habits play a crucial role. A high intake of red and processed meats, alcohol, and salt-preserved foods has been strongly associated with an increased risk of gastric cancer. Conversely, regular consumption of fruits and vegetables appears to exert a protective effect. In this context, polyphenols have gained increasing attention for their anti-inflammatory, antioxidant, and anti-proliferative properties.4,5 Recent studies confirmed that vegetable-based diets are significantly associated with a reduced risk of gastrointestinal malignancies, including gastric cancer.5–7 Sub-classes of compounds, including hydroxybenzaldehydes, tyrosols, and hydroxycoumarins, have demonstrated particularly strong protective associations, with risk reductions exceeding 50% in the highest intake quartiles.7 Within this context, the concept of functional juice becomes particularly relevant. Functional foods and beverages are conventionally designed to positively affect physiological functions beyond basic nutrition. Some of these products are inherently functional due to their natural richness in polyphenols, flavonoids, and other bioactive molecules. Prunus spinosa L., commonly known as blackthorn, is one such plant. Its dark-purple drupes have been recognized in traditional cuisine across Europe. Phytochemical studies have revealed a high content of anthocyanins, flavonols, and hydroxycinnamic acids.8 This study aims to explore the biological effects of a highly concentrated juice obtained from Prunus spinosa L., harvested in the Marche region of Italy, within the nutraceutical space “beyond food, before pharmaceuticals”.9 As preventive oncology shifts towards plant-based tools, this intervention offers a valuable approach, complementing these broader.4,10 While numerous plant extracts have been extensively studied in relation to gastric carcinogenesis,11–13 the investigation of whole, unprocessed fruits, particularly those naturally rich in bioactive compounds, remains limited. This evidence highlights the need to further investigate lesser-explored fruits, such as Prunus spinosa L., as promising candidates for nutraceutical applications in gastric cancer prevention. In this context, the present study evaluates the chemical composition of Prunus juice and its impact on cell viability, mitochondrial integrity, and the expression of apoptotic and autophagic markers in two human gastric adenocarcinoma cell lines (AGS and KATO III), as well as in a healthy gastric epithelial model (GES-1). Therefore, we established that the juice obtained from Prunus spinosa L. exhibits a rich polyphenolic profile and exerts biological effects against gastric cancer.2. Materials and methods
2.1. Production and chemical composition of Prunus spinosa L. juice
| No. | Compounds | Precursor ion, m/z | Product ion, m/z | Fragm-entor, V | Collision energy, V | Polarity | Retention time (Rt, min) |
|---|---|---|---|---|---|---|---|
| a These product ions were used for quantification. | |||||||
| 1 | Gallic acid | 169 | 125.2a | 97 | 12 | Negative | 6.96 |
| 2 | Neochlorogenic acid | 353 | 191.2a, 179 | 82 | 12, 12 | Negative | 9.52 |
| 3 | Delphinidin-3-galactoside | 465.01 | 303a | 121 | 20 | Positive | 11.36 |
| 4 | (+)-Catechin | 289 | 245.2a,109.2 | 131 | 8, 20 | Negative | 11.44 |
| 5 | Procyanidin B2 | 576.99 | 576.99a, 321.2 | 160 | 0, 32 | Negative | 12.41 |
| 6 | Chlorogenic acid | 353 | 191.2a, 127.5 | 82 | 12, 20 | Negative | 12.42 |
| 7 | p-Hydroxybenzoic acid | 137 | 93.2a | 92 | 16 | Negative | 12.86 |
| 8 | (−)-Epicatechin | 289 | 245.1a, 109.1 | 126 | 8, 20 | Negative | 13.03 |
| 9 | Cyanidin-3-glucoside | 449 | 287.3a, 255.6 | 121 | 20, 20 | Positive | 13.14 |
| 10 | Petunidin-3-glucoside | 479.01 | 317a, 302 | 121 | 20, 44 | Positive | 13.26 |
| 11 | 3-Hydroxybenzoic acid | 137 | 93.2a | 88 | 8 | Negative | 13.59 |
| 12 | Caffeic acid | 179 | 135.2a, 134.1 | 92 | 12, 24 | Negative | 13.65 |
| 13 | Vanillic acid | 167 | 152.4a, 108.1 | 88 | 12, 20 | Negative | 14.32 |
| 14 | Pelargonidin-3-glucoside | 433.01 | 271a, 121 | 116 | 24, 50 | Positive | 14.52 |
| 15 | Pelagonidin-3-rutinoside | 579.01 | 271a | 145 | 32 | Positive | 14.56 |
| 16 | Malvidin-3-galactoside | 493.01 | 331a, 315.1 | 121 | 20, 50 | Positive | 14.64 |
| 17 | Syringic acid | 196.9 | 182.2a, 121.2 | 93 | 8, 12 | Negative | 15.28 |
| 18 | Procyanidin A2 | 575 | 575a, 285 | 170 | 0, 20 | Negative | 16.18 |
| 19 | p-Coumaric acid | 163 | 119.2a, 93.2 | 83 | 12, 36 | Negative | 16.70 |
| 20 | Ferulic acid | 193 | 134.2a, 131.6 | 83 | 12, 8 | Negative | 17.10 |
| 21 | Rutin | 609 | 300.2a, 271.2 | 170 | 32, 50 | Negative | 17.73 |
| 22 | Hyperoside | 465.01 | 303a, 61.1 | 97 | 8, 50 | Positive | 18.33 |
| 23 | Isoquercitrin | 463 | 271.2a,300.2 | 155 | 44, 24 | Negative | 18.36 |
| 24 | Delphinidin-3,5-diglucoside | 462.9 | 300.1a | 165 | 24 | Negative | 18.38 |
| 25 | Phloridzin | 435.39 | 273a, 167 | 155 | 8, 28 | Negative | 18.83 |
| 26 | Quercitrin | 446.99 | 300.2a, 301.2 | 160 | 24, 16 | Negative | 19.61 |
| 27 | Myricetin | 316.99 | 179.1a, 182 | 150 | 16, 24 | Negative | 19.61 |
| 28 | Naringin | 578.99 | 271.3a, 151.3 | 170 | 32, 44 | Negative | 19.62 |
| 29 | Kaempferol-3-glucoside | 447 | 284.2a, 255.2 | 170 | 24, 40 | Negative | 19.77 |
| 30 | Hesperidin | 611.01 | 303a, 334.8 | 112 | 20, 12 | Positive | 20.19 |
| 31 | Ellagic acid | 301 | 301a, 229 | 170 | 0, 24 | Negative | 21.41 |
| 32 | Quercetin | 300.99 | 151.2a, 179.2 | 145 | 16, 12 | Negative | 21.87 |
| 33 | Phloretin | 272.99 | 167a, 123 | 116 | 8, 20 | Negative | 22.30 |
| 34 | Kaempferol | 287.01 | 153a, 69.1 | 60 | 36, 50 | Positive | 23.84 |
| 35 | Isorhamnetin | 314.99 | 300.2a, 196.1 | 145 | 16, 4 | Negative | 24.57 |
2.2. Cell lines and reagents
Commercial human gastric cancer cell lines (AGS and KATO III) were purchased from ATCC (American Type Culture Collection, Manassas, VA, USA). Healthy human epithelial gastric cells (GES-1) were kindly provided by Prof. Mario Dell'Agli (Department of Pharmacological and Biomolecular Sciences “Rodolfo Paoletti”, University of Milan, 20133 Milan, Italy). AGS cells, isolated from a female patient with gastric adenocarcinoma, were grown in DMEM/F12 medium (Gibco, Thermo Fisher Scientific, Waltham, MA, USA) supplemented with 100 U mL−1 penicillin (Gibco, Thermo Fisher Scientific, Waltham, MA, USA), 100 mg mL−1 streptomycin (Gibco, Thermo Fisher Scientific, Waltham, MA, USA), 2 mM L-glutamine (Gibco, Thermo Fisher Scientific, Waltham, MA, USA) and 10% heat-inactivated fetal bovine serum (FBS, Euroclone Milano, Italy). KATO III cells were obtained in vitro from a pleural effusion of a 55-year-old, Asian male stomach cancer patient and cultured with Iscove's Modified Dulbecco's Medium (30-2005™, ATCC) supplemented with 20% FBS. GES-1 cells, a non-cancerous gastric epithelial immortalized cell line, were used as a control. These cells, derived from normal gastric epithelial tissue, were cultured in RPMI-1640 medium (Gibco, Thermo Fisher Scientific, Waltham, MA, USA) supplemented with 10% FBS, 100 U mL−1 penicillin, 100 mg mL−1 streptomycin, and 1% L-glutamine (Gibco, Thermo Fisher Scientific, Waltham, MA, USA). All cell lines were maintained at 37 °C with 5% CO2.11 Lyophilized Prunus spinosa L. juice was dissolved in DMSO to generate a stock solution of 100 mg mL−1, which was subsequently diluted in growth medium before being added to the cells.2.3. MTT assay
The MTT assay was employed to evaluate the cytotoxicity of Prunus spinosa L. juice and its main compounds on gastric epithelial cells. The selected compounds were purchased from Med Chem Express (MCE): neochlorogenic acid (HY-N0722), cyanidin 3-O-glucoside chloride (HY-N0640), rutin (HY-N0148), vanillic acid (HY-N0708). Cells were seeded into 96-well plates at a density of 10 000 cells per well and incubated at 37 °C with 5% CO2 for 24 hours. After incubation, the medium was replaced with 100 μL of Prunus spinosa L. dissolved in fresh medium at concentrations ranging from 0 to 500 μg mL−1. As controls, untreated cells and cells treated with an equivalent concentration of DMSO were considered. For the selected compounds, we calculated the starting dose considering their percentage in 500 μg mL−1 of Prunus spinosa L. juice. The plates were incubated at 37 °C for 72 hours. Subsequently, 10 μL of MTT solution (R&D Systems, Minneapolis, MN, USA) were added to each well and incubated for 3 hours at 37 °C. Then, 100 μL of Detergent Reagent (R&D Systems, Minneapolis, MN, USA) were used to dissolve the purple formazan crystals formed by the reduction of MTT by metabolically active cells. Absorbance was measured at 570 nm using a microplate reader. Cytotoxicity was calculated based on the absorbance values, allowing for the assessment of the cytotoxic effects of Prunus spinosa L. juice on gastric cell lines. The graphs were generated using GraphPad Prism v.9.0 (GraphPad Software, La Jolla, CA, USA); R2 > 0.6 was considered significant.2.4. Trypan Blue assay
Cell viability of GES-1 cells treated with equivalent concentrations of DMSO was assessed and compared to untreated controls by means of the Trypan Blue (TB) exclusion assay. The same experimental approach has been used to evaluate the effect of Prunus spinosa L. juice on gastric cell viability. Briefly, cells were washed and suspended (1.0 × 105 cells per mL) in a solution of 1× phosphate-buffered saline (PBS) with 0.5 mM EDTA (Life Technologies, Carlsbad, CA, USA) containing 0.2% bovine serum albumin (BSA, Sigma-Aldrich). 50 μL of the cell suspension were added to an equal volume of 0.4% TB. The solution was mixed thoroughly and allowed to stand for 5 minutes at room temperature (RT). 10 μL of the solution were transferred to a hemocytometer, and viable (clear) and dead (blue) cells were counted. The number of live cells divided by the total number of counted cells (clear and blue) gave the percentage of viability. Viability graphs were generated using GraphPad Prism v.9.0 (GraphPad Software, La Jolla, CA, USA). For statistical analysis, one-way ANOVA with Turkey's multiple comparisons test was performed at both 24 and 48 hours of treatment. Data were considered statistically significant when p < 0.05 (*), highly significant when p < 0.005 (**), and even more highly significant when p < 0.0005 (***).2.5. Transmission electron microscopy (TEM)
Cells were fixed (1 hour at RT) in 2.5% glutaraldehyde in 0.1 M phosphate buffer. Samples were then washed, post-fixed in 1% osmium tetroxide (1 hour at RT), and dehydrated in graded ethanol solutions. Finally, samples were embedded in epoxy resin at 60 °C for three days. Semi-thin sections were cut and stained with toluidine blue and examined by a light microscope. Ultrathin sections were contrasted with UranyLess and lead citrate and observed with a Philips CM10 Electron Microscope.2.6. Protein extraction and western blotting (WB)
Cells were lysed in radioimmunoprecipitation assay lysis buffer (RIPA) containing EDTA-free protease and phosphatase inhibitor cocktails (Thermo Fisher Scientific). Cells were lysed by vortexing for 30 minutes at 4 °C and then cleared of cellular debris by centrifugation at 14 000g for 15 minutes at 4 °C. The protein concentration of the cleared lysates was determined using the Pierce BCA protein assay kit (Thermo Fisher Scientific). Proteins were separated by sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE) on 4–12% gradient gels and immunoblotted. Primary antibodies used were as follows: Cell Signaling Technologies (Beverly, MA, USA): anti-Caspase-3 (Cat. No. 9665S, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1000), anti-Caspase-3 cleaved (Cat. No. 9661, 1
1000), anti-Caspase-3 cleaved (Cat. No. 9661, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1000), anti-Beclin-1 (Cat. No, 3738S, 1
1000), anti-Beclin-1 (Cat. No, 3738S, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1000), anti-LC3B (Cat. No. 2775S, 1
1000), anti-LC3B (Cat. No. 2775S, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1000). Santa Cruz Biotechnology (Dallas, TX, USA): anti-Actin (Cat. No. sc-1616, 1
1000). Santa Cruz Biotechnology (Dallas, TX, USA): anti-Actin (Cat. No. sc-1616, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500), anti-Bax (Cat. No. sc-23959, 1
500), anti-Bax (Cat. No. sc-23959, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1000), anti-Bcl-x S/L (Cat. No. sc-1041, 1
1000), anti-Bcl-x S/L (Cat. No. sc-1041, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1000). After four washes in PBS containing 0.1% Tween-20, samples were incubated for 1 hour at RT with peroxidase-conjugated secondary antibody diluted 1
1000). After four washes in PBS containing 0.1% Tween-20, samples were incubated for 1 hour at RT with peroxidase-conjugated secondary antibody diluted 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 000 in PBS-Tween-20 and washed as described above. Detection of protein bands using the iBright Imaging System (Invitrogen) was done by incubating the membranes with Westar Antares ECL substrate (Cyanagen, Bologna, Italy).
10 000 in PBS-Tween-20 and washed as described above. Detection of protein bands using the iBright Imaging System (Invitrogen) was done by incubating the membranes with Westar Antares ECL substrate (Cyanagen, Bologna, Italy).
2.7. Confocal laser scanning microscopy (CLSM) – immunofluorescence
Cells were grown on coverslips, then fixed in 4% paraformaldehyde in 0.1 M PBS, pH 7.4, for 30 minutes, and permeabilized with 0.20% Triton X-100 in PBS, for 10 minutes at RT. Samples were rinsed again with PBS, treated with 2% BSA and 5% normal goat serum (NGS) in PBS (PBS–NGS–BSA mixture) for 30 minutes at RT. Afterwards, they were incubated with Cyt C, a monoclonal mouse primary antibody (anti-cytochrome C antibody, 7H8.2C12, A. Becton Dickinson, PharMingen), against cytochrome c (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 100 in the PBS–NGS–BSA mixture) overnight at 4 °C. The next day, samples were rinsed with PBS and incubated with AlexaFluor 488 conjugated goat anti-mouse secondary antibody (Millipore, Burlington, MA, USA; 1
100 in the PBS–NGS–BSA mixture) overnight at 4 °C. The next day, samples were rinsed with PBS and incubated with AlexaFluor 488 conjugated goat anti-mouse secondary antibody (Millipore, Burlington, MA, USA; 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50 in the PBS–BSA–NGS mixture) for 1 hour at RT in the dark. Control sections for non-specific staining were treated with the same incubation procedure by omitting the primary antibody. Slices were finally mounted with the Vectashield mounting media (Vectorlabs, Newark, CA, USA). Images were acquired by means of a Leica TCS-SP5 confocal microscope, connected to a DMI 6000 CS inverted microscope (Leica Microsystems CMS GmbH, 35578 Wetzlar, Germany) and analyzed using the software Leica Application Suite Advanced Fluorescence (LAS AF Version 2012.1.1). Samples were examined using an oil immersion objective lens (100× N.A. 1.25). Excitation was 488 nm (FITC), and the emission signal was detected at 519 nm. CLSM images are presented as single-plane images, and image analysis was carried out using ImageJ 1.54j software (National Institutes of Health).
50 in the PBS–BSA–NGS mixture) for 1 hour at RT in the dark. Control sections for non-specific staining were treated with the same incubation procedure by omitting the primary antibody. Slices were finally mounted with the Vectashield mounting media (Vectorlabs, Newark, CA, USA). Images were acquired by means of a Leica TCS-SP5 confocal microscope, connected to a DMI 6000 CS inverted microscope (Leica Microsystems CMS GmbH, 35578 Wetzlar, Germany) and analyzed using the software Leica Application Suite Advanced Fluorescence (LAS AF Version 2012.1.1). Samples were examined using an oil immersion objective lens (100× N.A. 1.25). Excitation was 488 nm (FITC), and the emission signal was detected at 519 nm. CLSM images are presented as single-plane images, and image analysis was carried out using ImageJ 1.54j software (National Institutes of Health).
2.8. 10-Nonyl acridine orange (NAO) detection
To assess the presence of peroxidation events, fresh cell pellets were treated with the fluorescent probe NAO. Briefly, 50 nM NAO was added to each sample for 10 minutes at RT; the specimens were then rinsed in PBS and finally observed through a CLSM. Excitation and emission signals were 488 nm and 519 nm for NAO.15 Densitometric analysis of NAO immunofluorescence was performed with ImageJ 1.54j Software (National Institutes of Health).The graphs were generated using GraphPad Prism v.9.0 (GraphPad Software, La Jolla, CA, USA). Data are presented as mean ± standard deviation (SD) and derive from at least three independent experiments. Data were analyzed using unpaired t-test, to identify specific differences between treatment and control groups. Results were considered statistically significant when p < 0.05 (*) and highly significant when p < 0.005 (**).
3. Results
3.1. Chemical composition of Prunus spinosa L. juice
The HPLC-MS/MS analysis of Prunus spinosa L. juice revealed a total polyphenol content of 996.78 mg kg−1, comprising various classes of bioactive compounds, including phenolic acids, flavanols, flavonols, and anthocyanins (Table 2). Among the phenolic acids, neochlorogenic acid (628.92 mg kg−1) was the most abundant, followed by vanillic acid (106.78 mg kg−1), caffeic acid (26.60 mg kg−1), chlorogenic acid (5.54 mg kg−1), and gallic acid (3.58 mg kg−1). The flavanol fraction included procyanidin A2 (62.04 mg kg−1), while catechin, epicatechin, and procyanidin B2 were not found. The major flavonol was rutin (39.04 mg kg−1), with lower concentrations of hyperoside (0.74 mg kg−1) and quercitrin (2.18 mg kg−1). Anthocyanins, responsible for the characteristic pigmentation and bioactivity of Prunus spinosa L., were dominated by cyanidin-3-glucoside (114.35 mg kg−1), followed by delphinidin-3,5-diglucoside (7.00 mg kg−1). This chemical profile highlights the richness of Prunus spinosa L. in polyphenols, particularly hydroxycinnamic acids and anthocyanins. Results are reported in Table 2 and are expressed in mg kg−1.| Compound | Mg kg−1 |
|---|---|
| Gallic acid | 3.58 |
| Neochlorogenic acid | 628.92 |
| Delphindin-3-galactoside | n.d. |
| Catechin | n.d. |
| Procyanidin B2 | n.d. |
| Chlorogenic acid | 5.54 |
| 4-Hydroxy benzoic acid | n.d. |
| Epicatechin | n.d. |
| Cyanidin-3-glucoside | 114.35 |
| Petunidin-3-glucoside | n.d. |
| 3-Hydroxy benzoic acid | n.d. |
| Caffeic acid | 26.60 |
| Vanillic acid | 106.78 |
| Pelargonidin-3-glucoside | n.d. |
| Pelargonidin-3-rutinoside | n.d. |
| Malvidin-3-galactoside | n.d. |
| Syringic acid | n.d. |
| Procyanidin A2 | 62.04 |
| p-Coumaric acid | n.d. |
| Ferulic acid | n.d. |
| Rutin | 39.04 |
| Hyperoside | 0.74 |
| Isoquercitrin | n.d. |
| Delphindin-3,5-diglucoside | 7.00 |
| Phloridzin | n.d. |
| Quercitrin | 2.18 |
| Myricetin | n.d. |
| Naringin | n.d. |
| Kaempferol-3-glucoside | n.d. |
| Hesperidin | n.d. |
| Ellagic acid | n.d. |
| Quercetin | n.d. |
| Phloretin | n.d. |
| Kaempferol | n.d. |
| Isorhamnetin | n.d. |
| TOTAL | 996.78 |
The concentration of polyphenols in Prunus spinosa L. juice was lower compared to that reported for Prunus spinosa L. fruit methanolic extracts.16 Nevertheless, the predominance of neochlorogenic acid and cyanidin 3-glucoside among the other polyphenols was confirmed.16,17
3.2. Prunus spinosa L. juice exerts anti-proliferative effects on gastric cancer cells while sparing healthy gastric cells
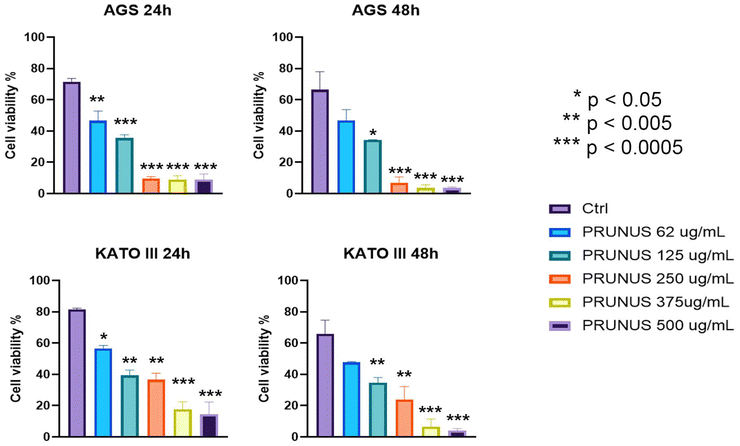
The cytotoxic potential of Prunus spinosa L. juice on gastric cancer cells was evaluated using the MTT assay. As illustrated in Fig. 1A, a 72-hour treatment with Prunus spinosa L. juice resulted in a dose-dependent reduction in cell viability in both AGS and KATO III gastric adenocarcinoma cell lines. Non-linear regression analysis revealed a comparable antiproliferative effect in both cell lines, with calculated IC50 values of 65.63 μg mL−1 for AGS and 69.76 μg mL−1 for KATO III cells, and an R2 value of 0.83, indicating a strong goodness-of-fit. Notably, the same treatment did not induce cytotoxic effects in non-tumorigenic gastric epithelial GES-1 cells. Instead, a slight increase in cell viability was observed in GES-1 cells treated with Prunus spinosa L. juice at concentrations up to 500 μg mL−1 (Fig. 1B), suggesting a potential pro-proliferative and cytoprotective effect on healthy gastric epithelial cells. Importantly, the viability of GES-1 cells treated with DMSO at concentrations equivalent to those used for juice treatment was comparable to that of untreated cells (Fig. 1C). Therefore, DMSO-treated cells were employed as controls in all subsequent experiments. These findings were further supported by ultrastructural analyses of juice-treated GES-1 cells, which revealed well-preserved cell viability (Fig. 1D and G), nuclear morphology – including an intact double membrane and diffuse chromatin (Fig. 1E and H) – as well as preserved viability of mitochondria and other subcellular organelles (Fig. 1F and I). Furthermore, no variation in Caspase-3 and LC3B expression was observed between untreated and Prunus juice-treated GES-1 cells (Fig. 1J). In conclusion, Prunus spinosa L. juice does not induce cell death or morphological signs of cellular stress in healthy gastric epithelial cells, supporting its safety profile for non-malignant tissue. To further investigate the impact of Prunus spinosa L. juice on gastric cancer, AGS and KATO III cells were assessed for viability using the TB exclusion assay (Fig. 2). Specifically, the cells were treated with increasing concentrations (62.5, 125, 250, 375 and 500 μg mL−1) of Prunus spinosa L. juice for 24 and 48 hours. At 375 and 500 μg mL−1, both AGS and KATO III cells exhibited near-complete loss of viability at both time points (Fig. 2). At these highest doses, plasma membrane integrity was lost, leading to necrosis. Therefore, concentrations of 125 and 250 μg mL−1, which still induced significant cell death without considerable necrosis, were selected for further experiments. Altogether, these results demonstrate that Prunus spinosa L. juice induces cytotoxicity in gastric cancer cell lines in a dose- and time-dependent manner.3.3. Prunus spinosa L. juice modulates autophagy and triggers apoptosis in gastric cancer cells
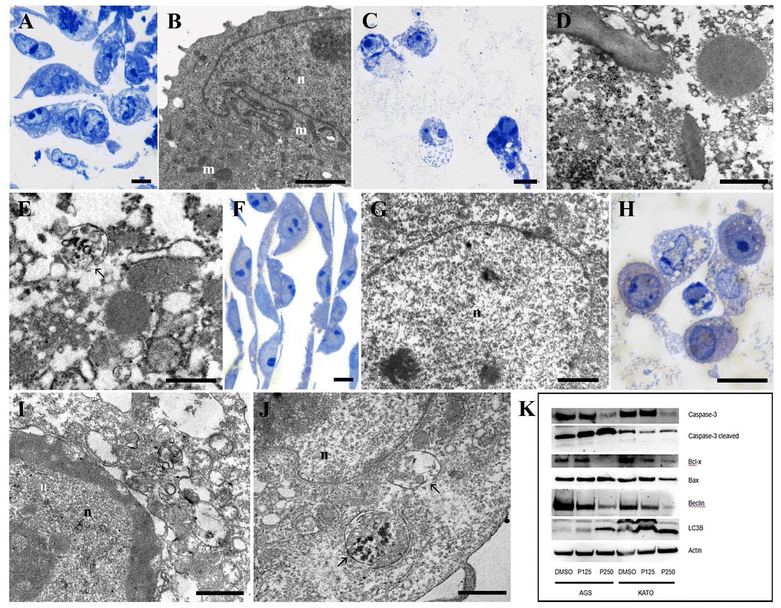
To investigate the morphological effects of Prunus spinosa L., TEM ultrastructural analysis was performed on gastric cancer cells. AGS and KATO III cells were treated with 125 μg mL−1 of the juice for 48 hours, and their morphology was compared with that of DMSO-treated control cells. Untreated AGS cells exhibited a preserved nuclear architecture, including a well-preserved nuclear membrane and diffuse chromatin, along with numerous structurally intact mitochondria (Fig. 3A and B). In contrast, AGS cells treated with Prunus spinosa L. juice displayed morphological hallmarks of apoptosis, such as cytoplasmic vacuolization and large autophagic vacuoles (Fig. 3C–E). A similar ultrastructural pattern was observed in KATO III cells exposed to the juice, with prominent autophagic vacuole formation suggesting activation of programmed cell death pathways (Fig. 3H–J). Conversely, DMSO-treated KATO III cells maintained normal morphology, with well-preserved mitochondrial and nuclear compartments (Fig. 3F and G). To elucidate the molecular mechanisms underlying the cytotoxic effects of Prunus spinosa L. juice, the expression of key autophagy- and apoptosis-related proteins was examined by WB in cells treated for 24 hours with 125 μg mL−1 and 250 μg mL−1 of the juice, compared to untreated control cells (Fig. 3K). In AGS cells, treatment with Prunus spinosa L. juice markedly increased the expression levels of cleaved/activated Caspase-3 in a dose-dependent manner, along with a progressive loss of the inactive full-length form, suggesting proteolytic activation. This was accompanied by dose-dependent downregulation of the anti-apoptotic protein Bcl-x, whereas the pro-apoptotic Bax showed a modest upregulation following juice treatment. Notably, increasing concentrations of juice also led to a significant reduction in the expression of the autophagy initiator Beclin-1. In parallel, a clear activation of the autophagosome marker LC3B was observed, evidenced by the accumulation of its lipidated form, LC3B-II, after Prunus spinosa L. juice treatment. In KATO III cells, the expression of autophagy-related proteins was similarly modulated. Beclin-1 levels displayed a dose-dependent downregulation, whereas LC3B activation was strongly enhanced in response to increasing juice concentrations. Bcl-x expression appears reduced by the treatment, while Bax exhibited a biphasic modulation: its expression increased upon treatment with 125 μg mL−1 of the juice but decreased at a higher concentration (250 μg mL−1). Regarding Caspase-3, although a progressive reduction in the full-length protein was observed, this only led to a partial accumulation of the cleaved, active form. Altogether, these findings suggest a complex, cell-type-dependent response to Prunus spinosa L. juice, promoting cell death by directly triggering the apoptotic cascade or activating alternative autophagy-related cytotoxicity mechanisms. To better characterize the behavior of autophagic vacuoles, a time-course experiment was conducted on AGS and KATO cells treated with 125 μg ml−1Prunus spinosa L. juice for 0, 12, 24, 36, and 48 hours, employing both ultrastructural analysis and WB. The results (Fig. 4) showed no presence of autophagic vacuoles at time 0 in both cell models (Fig. 4A and G). The activation of the autophagic process begins at 12 hours of treatment in AGS cells (Fig. 4A and F). The image shows a phagophore in an attempt to surround the damaged organelles (Fig. 4B). After 12 h of treatment, the presence of damaged mitochondria could be observed in Kato III cells (Fig. 4H). The endoplasmic reticulum was observed to align alongside the damaged mitochondria (Fig. 4H). The autophagosomes together with autophagolysosomes and LC3B-II expression appeared after 24 hours of exposure (Fig. 3K and 4C, I) and became particularly evident at 36 hours of treatment (Fig. 4D, E, J and M), especially in KATO cells (Fig. 4J and M). After 48 hours, the amount of autophagosomes and LC3B-II expression decreases in both cell lines (Fig. 4E, F, K, L and M), likely reflecting the progression of advanced stages of cell death. In AGS cells, death appeared predominantly apoptotic, as evidenced by the presence of micronuclei and condensed chromatin as early as 36 hours after exposure to Prunus spinosa L. juice (Fig. 4D and E). Conversely, in KATO cells, autophagy remained the main death mechanism. Indeed, after 48 hours of exposure, fully vacuolated cells (Fig. 4L) were observed, whereas other cells underwent secondary necrosis (Fig. 4K and L).3.4. Prunus spinosa L. juice impairs mitochondrial integrity and triggers peroxidative damage and cytochrome c release
To further confirm the presence of apoptotic processes, the integrity of the mitochondrial membrane was investigated. Its evaluation was done by NAO staining, which was performed on AGS and KATO III cells treated with 125 μg mL−1 of the Prunus spinosa L. juice for 48 hours. NAO dye binds to cardiolipin and accumulates within intact mitochondria. When this interaction is disrupted, for instance upon ROS production, the release of Cyt C and apoptosis initiation occur.18 As expected, NAO-associated green fluorescence was substantially reduced in AGS and KATO III cells after 48 hours of treatment compared to control cells. In untreated cells, a strong cytoplasmic signal was observed, indicative of cardiolipin-rich, functionally intact mitochondrial membranes. In contrast, juice-treated cells exhibited a marked reduction in signal intensity, suggesting the occurrence of cardiolipin-targeted peroxidative events leading to mitochondrial membrane disruption (Fig. 5 and 6, top left). Quantitative analysis of NAO fluorescence further confirmed a statistically significant reduction in signal intensity (p < 0.01) in gastric cancer cells exposed to Prunus spinosa L. juice (Fig. 5 and 6, top right). Under normal conditions, the mitochondrial enzyme Cyt C is retained in the intermembrane space. Upon cardiolipin oxidation, Cyt C is released into the cytoplasm, where it promotes apoptosome assembly and the progression of apoptosis.19,20 Consistently, immunofluorescence analysis of untreated AGS and KATO III cells revealed a distinct, punctate Cyt C signal localized to mitochondria. In contrast, a 48-hour treatment with Prunus spinosa L. juice caused a redistribution of Cyt C fluorescence to a diffuse cytoplasmic pattern, indicative of mitochondrial membrane disruption and consistent with outer membrane permeabilization and activation of the intrinsic apoptotic pathway (Fig. 5 and 6). Brightfield images of juice-treated AGS and KATO III cells further revealed morphological features typical of apoptosis, including cell rounding, membrane blebbing, and reduced adhesion, in contrast to the well-preserved cellular and nuclear architecture, intact membranes, and organized cytoplasm observed in untreated cells (Fig. 5 and 6). Taken together, these findings highlight the cytotoxic potential of Prunus spinosa L. juice, which induces mitochondrial dysfunction, Cyt C release, and morphological alterations consistent with apoptosis in gastric cancer cells.3.5. Individual phenolic compounds from Prunus spinosa L. juice do not affect gastric cancer cell viability
Given the observed antitumoral activity of Prunus spinosa L. juice, the cytotoxic potential of its individual phenolic constituents was evaluated. MTT cell viability assay was performed on AGS and KATO III cells exposed for 72 hours to neochlorogenic acid (NCA), cyanidin-3-glucoside (C3G), vanillic acid (VA), and rutin, at concentrations corresponding to their amounts in 500 μg mL−1 of Prunus spinosa L. juice and at lower dilutions. As shown in Fig. 7, none of the four compounds at these concentrations exhibited cytotoxic effects against AGS or KATO III cells.4. Discussion
Gastric cancer remains a global health challenge, representing the fifth most diagnosed malignancy and the fourth leading cause of cancer-related mortality worldwide.21,22 Although surgery and (neo)adjuvant chemo-radiotherapy can be effective, their benefits are often offset by significant side effects and the emergence of drug resistance.23,24 Growing interest has emerged in preventing gastric cancer through safer strategies. Epidemiological and preclinical evidence links plant-rich diets to a reduced risk of gastric cancer.25 Numerous plant-derived bioactive compounds show anti-cancer activity while exerting minimal toxicity on healthy cells.26 These substances may contribute to the elimination of early malignant cells or to the prevention of carcinogenesis. In this context, our study bridges food sciences and oncology by investigating the antitumoral potential of a fruit juice derived from Prunus spinosa L. against gastric cancer. Specifically, we evaluated the cytotoxic potential of Prunus spinosa L. juice against transformed gastric epithelial cells and assessed its safety in healthy gastric epithelial cells (Fig. 8). In our in vitro experiments, Prunus spinosa L. juice exhibited cytotoxic and pro-apoptotic activities against human gastric adenocarcinoma cells (AGS) and signet-ring carcinoma cells (KATO III), while remarkably sparing healthy gastric epithelial cells. Treated cancer cells displayed dose-dependent growth inhibition, accompanied by robust induction of apoptosis and autophagy. In contrast, the viability of normal GES-1 cells was slightly enhanced rather than impaired upon exposure to the juice, suggesting a potential trophic effect at specific doses. This differential response underscores a desirable selective toxicity, targeting malignant cells while preserving normal cells. The ability of blackthorn juice to orchestrate the induction of both apoptotic and autophagic pathways in gastric cancer cells suggests a multitarget mechanism of action. This concerted activation has gained increasing recognition in cancer biology and may underlie the growth inhibition observed in AGS and KATO III cells, suggesting that Prunus spinosa L. juice could target key regulators involved in the crosstalk between autophagy and apoptosis, such as the Bcl-2 protein family.27,28 Experimentally, juice-treated AGS and KATO III cells exhibited reduced Beclin-1 levels alongside increased LC3B-II expression. These molecular events are characteristic of Beclin-1-independent autophagy, a process known to contribute significantly to caspase-dependent apoptosis. Notably, Prunus spinosa L. juice exhibited no cytotoxicity toward non-transformed gastric GES-1 cells, even at high concentrations. Similar selectivity has been observed in various food-derived compounds with anticancer potential. For instance, ellagic acid was recently shown to inhibit proliferation and induce apoptosis in AGS gastric cancer cells in vitro and in vivo, sparing normal cells.12 Our findings dovetail nicely with a growing body of literature on fruit extracts, plant polyphenols, and other dietary compounds exhibiting antitumor effects in gastric cancer models. Several phytochemicals present in Prunus juice, also found in other foods, demonstrate strikingly similar activities, including inhibition of gastric cancer cell proliferation, induction of apoptosis, and frequent involvement of autophagic pathways: rutin selectively inhibits proliferation and induces apoptosis in AGS and MGC803 gastric cancer cells without affecting GES-1 cell viability;29 C3G exhibits selective antiproliferative and pro-apoptotic effects on human gastric adenocarcinoma cells while sparing healthy epithelial GES-1 cells;30 Chlorogenic acid (CGA) exerts dose-dependent cytotoxic effects on AGS cells through cell cycle blockage and apoptosis activation;31 gallic acid (GA) selectively blocks gastric epithelial transformation, while suppressing tumor growth.32,33 Notably, the biological effects of Prunus spinosa L. juice observed in our study align well with this antitumoral activity. Its ability to modulate apoptosis, disrupt mitochondrial integrity, and induce autophagy, while sparing non-malignant gastric cells, underscores a mechanistic convergence with well-characterized dietary polyphenols. The bioaccessibility profile of these phytochemicals have been extensively investigated. NCA, the most abundant compound in the extract, is almost completely metabolized by the intestinal microflora into simpler and rapidly-absorbed phenolic acids, thereby serving as a valuable precursor of bioavailable antitumoral phenolic metabolites.34,35 Similarly, anthocyanin-derived direct metabolites, including glycated chalcones and simple phenolic acids, are detected in the systemic circulation and may contribute to the health benefits associated with anthocyanin intake. Chemically less complex molecules found in the extract – including gallic, vanillic, and caffeic acids have favorable absorption profiles and can be readily distributed to target tissues where they exert their biological activities.36,37 Rutin, the main dietary source of quercetin, is hydrolyzed by the cecal bacteria and absorbed as quercetin, which exerts cytotoxic effects against gastric cancer cells.38,39 Taken together, these bioaccessibility profiles highlight the translational potential of our blend, as the identified phytochemicals – either in their intact form or as direct metabolites – are able to reach the systemic circulation and exert their biological effects. | ||
| Fig. 8 Graphic representation of the biological role of Prunus spinosa L. juice (Prunus blend) in healthy epithelial GES-1 cells and gastric cancer AGS and KATO III cells. | ||
Consistent with existing literature, our study highlights the crosstalk between autophagy and apoptosis that emerges in gastric cancer cells upon exposure to dietary anti-cancer agents. The interplay between these two processes plays a pivotal role in early carcinogenesis and its prevention. Controlled activation of autophagy may be beneficial for gastric cancer prevention by triggering self-destructive mechanisms in premalignant or highly stressed cells. Numerous antitumoral phytochemicals appear capable of shifting autophagy from a survival mechanism to a cytotoxic one in cancer cells. For example, myricetin-induced autophagy was shown to promote cytotoxicity, indicating that autophagy contributed to apoptosis rather than counteracting it.40 In line with this evidence, our time-course ultrastructural and WB analyses indicate that autophagy contributes to the cytotoxic activity of Prunus spinosa L. juice against gastric cancer cells, either as the predominant mechanism or as an upstream driver that triggers apoptosis. In both AGS and KATO III models, LC3B-II upregulation and autophagosome generation confirmed the engagement of autophagy. In AGS cells, this response culminated in autophagy-mediated apoptosis, as indicated by increased cleaved Caspase-3 levels, Bax upregulation, and Bcl-x downregulation. Conversely, although KATO III cells also exhibited LC3B-II accumulation, their death was associated with a necroptotic shift in the cell death programme, as evidenced by reduced Caspase-3 activation and morphological signs of secondary necroptosis (plasma membrane perforation and cellular collapse). Moreover, the biphasic modulation of Bax expression suggests that, while apoptosis is engaged at lower doses of Prunus juice, it does not represent the predominant mechanism driving cytotoxicity at higher concentrations. Instead, cell death in KATO III cells appears to rely mainly on LC3B-dependent autophagy, a form of autophagy-mediated cell death that can occur independently of Bax.41 Overall, Prunus juice exerts strong cytotoxicity against gastric cancer cells through a dual mechanism involving both pro-apoptotic and pro-autophagic activities, ultimately inducing either apoptotic cell death or autophagy-associated secondary necroptosis. Notably, individual phenolic constituents of the blend failed to replicate the cytotoxic effects of Prunus juice in AGS and KATO III cells. This evidence aligns with the nutraceutical paradigm, which seeks to transcend the “one-disease, one target drug” concept by employing crude extracts that deliver functional blends of active compounds capable of engaging multiple targets simultaneously, thus yielding superior biological outputs. Indeed, cancer pathogenesis and progression are multifactorial and thus more susceptible to combination regimens than to single-agent strategies.42 Within this framework, our results further substantiate the value of this functional product, demonstrating that its cytotoxic activity arises from the complex interplay of its active constituents.
Altogether, the observed anti-proliferative and pro-apoptotic effects of Prunus juice on gastric cancer cells are encouraging, especially considering its favorable safety profile in normal cells. This suggests a potential role for Prunus-derived nutraceuticals in intercepting early malignant transformation or suppressing the outgrowth of dormant neoplastic lesions in the stomach. The presented data paves the way for further investigation in more complex experimental models. For instance, assessing the effects of Prunus spinosa L. juice in animal models of gastric carcinogenesis may elucidate its ability to reduce tumor incidence or burden, while mechanistic studies may help pinpoint the most bioactive constituents and clarify how they might be optimized. Moreover, combining such food-derived blends with existing preventive strategies – such as Helicobacter pylori eradication and targeted dietary modifications – may offer an integrated approach to gastric cancer prevention. The integration of such functional substances into preventive strategies may be further facilitated by the identification of predictive biomarkers of gastric cancer susceptibility, allowing for tailored interventions in at-risk populations.43,44 Altogether, our findings contribute to this translational trajectory, suggesting that food-derived blends such as Prunus spinosa L. juice hold significant potential for the development of evidence-based, food-derived strategies for cancer prevention and management.
Author contributions
Matteo Micucci, Federico Gianfanti, Michela Battistelli, Giovanni Caprioli: conceptualization, investigation, supervision, writing – review & editing – original draft. Matteo Micucci: project administration, funding acquisition, methodology, and visualization. Sara Salucci and Irene Faenza: writing – review & editing – original draft, supervision, investigation, software, validation. Riham Osman: software, investigation. Michele Mari, Michele Retini, and Sabrina Burattini: data curation, writing – review & editing – original draft. Francesco Onesimo, Ilaria Versari, Laura Acquaticci, and Gianni Sagratini: data curation.Conflicts of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.Data availability
Data will be made available on request.Supplementary information (SI) is available. See DOI: https://doi.org/10.1039/d5fo02227d.
Acknowledgements
The authors thank Samuela Capellacci, University of Urbino, Carlo Bo and IOR Istituto Oncologico Romagnolo Coop. Soc. https://www.ior-romagna.it/. This work was financially supported by the University of Urbino Carlo Bo.References
- A. P. Thrift, T. N. Wenker and H. B. El-Serag, Global burden of gastric cancer: epidemiological trends, risk factors, screening, and prevention, Nat. Rev. Clin. Oncol., 2023, 20, 338–349 Search PubMed.
- European Cancer Information System, 2022. https://ecis.jrc.ec.europa.eu, accessed 01/09/2022.
- OECD/European Commission (2025). EU Country Cancer Profile, Italy, 2025, EU Country Cancer Profiles, OECD Publishing, Paris Search PubMed.
- M. Micucci, B. X. Kwan, H. Yee, C. Minting, M. Mari, M. Retini, S. Burattini, R. Osman, U. J. Okeke, F. O. Abdullah, F. Gianfanti and M. Battistelli, Matching traditional Chinese medicine and western medicine-based research: Advanced nutraceutical development for proactive gastric cancer prevention, World J. Gastrointes. Oncol., 2024, 16, 3798–3819 Search PubMed.
- T. Bai, J. Peng, X. Zhu and C. Wu, Vegetarian diets and the risk of gastrointestinal cancers: a meta-analysis of observational studies, Eur. J. Gastroenterol. Hepatol., 2023, 35, 1244–1252 Search PubMed.
- M. De Araújo Fagundes, R. Alves Carnauba, G. A. Fernandes, P. Pimentel de Assumpção and M. P. Curado, Polyphenol intake and gastric cancer: A case-control study in the Brazilian Amazon region, Cancer Epidemiol., 2024, 88, 102518 Search PubMed.
- M. Rubín-García, F. Vitelli-Storelli, A. J. Molina, R. Zamora-Ros, N. Aragonés, E. Adarnaz, G. Castaño-Vinyals, M. Obón-Santacana, I. Gómez-Acebo, A. Molina-Barceló, G. Fernández-Tardón, J. J. Jiménez-Moleón, J. Alguacil, M. D. Chirlaque, E. Toledo, B. Pérez-Gómez, M. Pollán, M. Kogevinas and V. Martín, Association between Polyphenol Intake and Gastric Cancer Risk by Anatomic and Histologic Subtypes: MCC-Spain, Nutrients, 2020, 12, 3281 Search PubMed.
- H. Džudžević-Čančar, A. Dedić, A. Alispahić and I. Špánik, Validation of an Isocratic HPLC Method for Simultaneous Estimation of Major Phytosterols in Prunus spinosa L. extracts, Acta Chim. Slov., 2024, 71, 305–313 Search PubMed.
- A. Santini, S. M. Cammarata, G. Capone, A. Ianaro, G. C. Tenore, L. Pani and E. Novellino, (2018). Nutraceuticals: opening the debate for a regulatory framework, Br. J. Clin. Pharmacol., 2018, 84, 659–6720 Search PubMed.
- S. Sritharan, S. Guha, S. Hazarika and N. Sivalingam, (2022). Meta analysis of bioactive compounds, miRNA, siRNA and cell death regulators as sensitizers to doxorubicin induced chemoresistance, Apoptosis, 2022, 27, 622–646 Search PubMed.
- M. Micucci, A. Bartoletti Stella, F. O. Abdullah, S. Burattini, I. Versari, M. Canale, F. D'Agostino, D. Roncarati, D. Piatti, G. Sagratini, G. Caprioli, M. Mari, M. Retini, I. Faenza, M. Battistelli and S. Salucci, Paradigm Shift in Gastric Cancer Prevention: Harnessing the Potential of Aristolochia olivieri Extract, Int. J. Mol. Sci., 2023, 24, 16003 Search PubMed.
- H. Cheshomi, A. R. Bahrami, H. Rafatpanah and M. M. Matin, The effects of ellagic acid and other pomegranate (Punica granatum L.) derivatives on human gastric cancer AGS cells, Hum. Exp. Toxicol., 2022, 41, 9603271211064534 Search PubMed.
- M. Micucci, M. Battistelli, S. Burattini, M. Mari, M. Retini, R. Osman, I. Versari, A. Bartoletti Stella, F. Gianfanti, J. O. Udodinma, F. Onesimo, S. Riela, M. Canale, A. Palumbo Piccionello, I. Faenza and S. Salucci, Food waste to wellness: a grape pomace blend in gastric cancer prevention, Food Sci. Nutr., 2025, 13, e70350 Search PubMed.
- A. M. Mustafa, S. Angeloni, D. Abouelenein, L. Acquaticci, J. Xiao, G. Sagratini, F. Maggi, S. Vittori and G. Caprioli, A new HPLC-MS/MS method for the simultaneous determination of 36 polyphenols in blueberry, strawberry and their commercial products and determination of antioxidant activity, Food Chem., 2022, 367, 130743 Search PubMed.
- S. Salucci, V. Baldassarri, B. Canonico, S. Burattini, M. Battistelli, M. Guescini, S. Papa, V. Stocchi and E. Falcieri, Melatonin behavior in restoring chemical damaged C2C12 myoblasts, Microsc, Res, Tech., 2016, 79, 532–540 Search PubMed.
- A. Magiera, M. E. Czerwińska, A. Owczarek, A. Marchelak, S. Granica and M. A. Olszewska, Polyphenol-enriched extracts of Prunus spinosa fruits: Anti-inflammatory and antioxidant effects in human immune cells ex vivo in relation to phytochemical profile, Molecules, 2022, 27, 1691 Search PubMed.
- J. M. Veličković, D. A. Kostić, G. S. Stojanović, S. S. Mitić, M. N. Mitić, S. S. Ranđelović and A. S. Đorđević, Phenolic composition, antioxidant and antimicrobial activity of the extracts from Prunus spinosa L. fruit, Hem. Ind., 2014, 68, 297–303 Search PubMed.
- C. Ferlini and G. Scambia, Assay for apoptosis using the mitochondrial probes, Rhodamine123 and 10-N-nonyl acridine orange, Nat. Protoc., 2017, 2, 3111–3114 Search PubMed.
- M. Hüttemann, P. Pecina, M. Rainbolt, T. H. Sanderson, V. E. Kagan, L. Samavati, J. W. Doan and I. Lee, The multiple functions of cytochrome c and their regulation in life and death decisions of the mammalian cell: From respiration to apoptosis, Mitochondrion, 2011, 11, 369–381 Search PubMed.
- C. Garrido, L. Galluzzi, M. Brunet, P. E. Puig, C. Didelot and G. Kroemer, Mechanisms of cytochrome c release from mitochondria, Cell Death Differ., 2006, 13, 1423–1433 Search PubMed.
- K. M. Sjoquist and J. R. Zalcberg, Gastric cancer: past progress and present challenges, Gastric Cancer, 2015, 18, 205–209 Search PubMed.
- J. L. Lin, J. X. Lin, G. T. Lin, C. M. Huang, C. H. Zheng, J. W. Xie, J. B. Wang, J. Lu, Q. Y. Chen and P. Li, Global incidence and mortality trends of gastric cancer and predicted mortality of gastric cancer by 2035, BMC Public Health, 2024, 24, 1763 Search PubMed.
- H. Yuan, M. Bao, M. Chen, J. Fu and S. Yu, Advances in Immunotherapy and Targeted Therapy for Gastric Cancer: A Comprehensive Review, Br. J. Hosp. Med., 2025, 86, 1–24 Search PubMed.
- H. L. Wiraswati, I. F. Ma'ruf, J. Sharifi-Rad and D. Calina, Piperine: an emerging biofactor with anticancer efficacy and therapeutic potential, Biofactors, 2025, 51, e2134 Search PubMed.
- A. Rudzińska, P. Juchaniuk, J. Oberda, J. Wiśniewska, W. Wojdan, K. Szklener and S. Mańdziuk, Phytochemicals in Cancer Treatment and Cancer Prevention—Review on Epidemiological Data and Clinical Trials, Nutrients, 2023, 15, 1896 Search PubMed.
- C. X. Ng, M. M. Affendi, P. P. Chong and S. H. Lee, The Potential of Plant-Derived Extracts and Compounds to Augment Anticancer Effects of Chemotherapeutic Drugs, Nutr. Cancer, 2022, 74, 3058–3076 Search PubMed.
- H. Xi, S. Wang, B. Wang, X. Hong, X. L. M. Liu, R. Shen and Q. Dong, The role of interaction between autophagy and apoptosis in tumorigenesis (Review), Oncol. Rep., 2022, 48, 208 Search PubMed.
- Y. El Mahi, Z. N. Nizami, A. F. Wali, A. Al Neyadi, M. Magramane, M. Al Azzani, K. Arafat, S. Attoub, A. H. Eid and R. Iratni, Rhus coriaria induces autophagic and apoptotic cell death in pancreatic cancer cells, Front. Pharmacol., 2024, 15, 1412565 Search PubMed.
- H. Huang, J. Shi, W. Chen and L. Liu, Rutin suppresses the malignant biological behavior of gastric cancer cells through the Wnt/β-catenin pathway, Discover Oncol., 2024, 15, 407 Search PubMed.
- F. J. Olivas-Aguirre, J. Rodrigo-García, N. D. Martínez-Ruiz, A. I. Cárdenas-Robles, S. O. Mendoza-Díaz, E. Álvarez-Parrilla, G. A. González-Aguilar, L. A. de la Rosa, A. Ramos-Jiménez and A. Wall-Medrano, Cyanidin-3-O-glucoside: Physical-Chemistry, Foodomics and Health Effects, Molecules, 2016, 21, 1264 Search PubMed.
- N. Jafari, S. J. Zargar, M. R. Delnavazi and N. Yassa, Cell Cycle Arrest and Apoptosis Induction of Phloroacetophenone Glycosides and Caffeoylquinic Acid Derivatives in Gastric Adenocarcinoma (AGS) Cells, Anticancer Agents Med. Chem., 2018, 18, 610–616 Search PubMed.
- W. Liao, Y. Wen, J. Wang, M. Zhao, S. Lv, N. Chen, Y. Li, L. Wan, Q. Zheng, Y. Mou, Z. Zhao, J. Tang and J. Zeng, Gallic acid alleviates gastric precancerous lesions through inhibition of epithelial mesenchymal transition via Wnt/β-catenin signaling pathway, J. Ethnopharmacol., 2023, 302, 115885 Search PubMed.
- C. L. Tsai, Y. M. Chiu, T. Y. Ho, C. T. Hsieh, D. C. Shieh, Y. J. Lee, G. J. Tsay and Y. Y. Wu, Gallic Acid Induces Apoptosis in Human Gastric Adenocarcinoma Cells, Anticancer Res., 2018, 38, 2057–2067 Search PubMed.
- D. Del Rio, A. Stalmach, L. Calani and A. Crozier, (2010). Bioavailability of coffee chlorogenic acids and green tea flavan-3-ols, Nutrients, 2010, 2, 820–833 Search PubMed.
- H. T. Chang, I. L. Chen, C. T. Chou, W. Z. Liang, D. H. Kuo, P. Shieh and C. R. Jan, Effect of caffeic acid on Ca(2+) homeostasis and apoptosis in SCM1 human gastric cancer cells, Arch. Toxicol., 2013, 87, 2141–2150 Search PubMed.
- A. Faria, I. Fernandes, N. Mateus and C. Calhau, Bioavailability of Anthocyanins, in Natural Products, ed. K. Ramawat and J. M. Mérillon, Springer, Berlin, Heidelberg, 2013 Search PubMed.
- C. I. Victoria-Campos, J. J. Ornelas-Paz, N. E. Rocha-Guzmán, J. A. Gallegos-Infante, M. L. Failla, J. D. Pérez-Martínez, C. Rios-Velasco and V. Ibarra-Junquera, Gastrointestinal metabolism and bioaccessibility of selected anthocyanins isolated from commonly consumed fruits, Food Chem., 2022, 383, 132451 Search PubMed.
- C. Manach, C. Morand, C. Demigné, O. Texier, F. Régérat and C. Rémésy, Bioavailability of rutin and quercetin in rats, FEBS Lett., 1977, 409, 12–16 Search PubMed.
- L. Ding, S. Dang, M. Sun, D. Zhou, Y. Sun, E. Li, S. Peng, J. Li and G. Li, Quercetin induces ferroptosis in gastric cancer cells by targeting SLC1A5 and regulating the p-Camk2/p-DRP1 and NRF2/GPX4 Axes, Free Radicals Biol. Med., 2024, 213, 150–163 Search PubMed.
- S. H. Han, J. H. Lee, J. S. Woo, G. H. Jung, S. H. Jung, E. J. Han, B. Kim, S. D. Cho, J. S. Nam, J. H. Che and J. Y. Jung, Myricetin induces apoptosis and autophagy in human gastric cancer cells through inhibition of the PI3K/Akt/mTOR pathway, Heliyon, 2022, 8, e09309 Search PubMed.
- F. McCoy, J. Hurwitz, N. McTavish, I. Paul, C. Barnes, B. O'Hagan, K. Odrzywol, J. Murray, D. Longley, G. McKerr and D. A. Fennell, Obatoclax induces Atg7-dependent autophagy independent of beclin-1 and BAX/BAK, Cell Death Dis., 2010, 1, e108 Search PubMed.
- L. K. Caesar and N. B. Cech, Synergy and antagonism in natural product extracts: when 1 + 1 does not equal 2, Nat. Prod. Rep., 2019, 36, 845–936 Search PubMed.
- S. Vickram, S. S. Infant, S. Manikandan, D. Jenila Rani, C. M. Mathan Muthu and H. Chopra, Immune biomarkers and predictive signatures in gastric cancer: Optimizing immunotherapy responses, Pathol., Res. Pract., 2025, 265, 155743 Search PubMed.
- L. Bian, B. Hu, F. Li, Y. Gu, C. Hu, Y. Chen, B. Deng, H. Fang, X. Zhu, Y. Chen, X. Fu, T. Wang, Q. She, M. Zhu, Y. Jiang, J. Dai, H. Xu, H. Ma, Z. Xu, Z. Hu, H. Shen, Y. Ding, C. Yan and G. Jin, Single-cell eQTL mapping reveals cell-type-specific genes associated with the risk of gastric cancer, Cell Genomics, 2025, 5, 100812 Search PubMed.
Footnote |
| † These authors equally contributed to the work. |
| This journal is © The Royal Society of Chemistry 2025 |