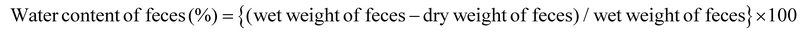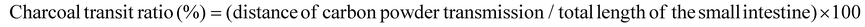Effects of lyophilized Korean rice wine (Makgeolli) on intestinal function and bowel activity in loperamide-induced constipation in rats
Soyoung Jung a,
Yunjung Lee
a,
Yunjung Lee b,
Yoo Kyoung Park
b,
Yoo Kyoung Park *a and
Eunju Park
*a and
Eunju Park *b
*b
aDepartment of Medical Nutrition, AgeTech-Service Convergence Major, Kyung Hee University, Yongin 17104, Republic of Korea. E-mail: ypark@khu.ac.kr
bDepartment of Food and Nutrition, Kyungnam University, Masan 51767, Republic of Korea. E-mail: pej@kyungman.ac.kr
First published on 2nd September 2025
Abstract
Constipation is an increasingly common gastrointestinal disorder worldwide. Makgeolli, a traditional Korean rice wine, is celebrated for its rich nutrient content, including probiotics and yeasts, which are known to promote intestinal motility. However, there is limited empirical research on its effects on bowel movements and overall gastrointestinal health. This study aims to explore the impact of Makgeolli on intestinal function and bowel activity using a loperamide-induced constipation model in rats. We divided six-week-old Sprague-Dawley rats into four groups: a normal control (NC), a constipation-induced group (LO, loperamide 4 mg kg−1), a positive control (L-SP, loperamide + sodium picosulfate 5 mg kg−1), and a Makgeolli treatment group (L-MG, loperamide + Makgeolli 200 mg kg−1). After two weeks of treatment, the L-MG group showed significant improvements in fecal parameters and gastrointestinal transit compared to the LO group (p < 0.05). Additionally, DNA damage in colonic cells and fecal water was significantly reduced in the L-MG group (p < 0.05). The activity of harmful intestinal enzymes, such as β-glucosidase, Tryptophanase, Nitroreductase, and β-glucuronidase, was also significantly decreased (p < 0.05). Furthermore, analysis of plasma ORAC levels and lipid peroxides (conjugated dienes) indicated a notable reduction in oxidative stress and lipid peroxidation in the L-MG group (p < 0.05). The mRNA expression levels of pro-inflammatory cytokines (COX-2, INOS, TNF-α) in colon tissue were significantly reduced (p < 0.05). Therefore, Makgeolli shows potential as a functional food that could improve bowel function and facilitate defecation, while also providing antioxidant and anti-inflammatory benefits that may help alleviate constipation.
Introduction
Makgeolli is a traditional Korean fermented alcoholic beverage made from grains such as non-glutinous rice, glutinous rice, and barley, using a fermenting agent called nuruk.1,2 Rich in nutrients such as vitamin B, lysine, leucine, and arginine, Makgeolli is also a notable source of lactic acid bacteria (LAB), which contribute to gut health by enhancing intestinal motility and suppressing harmful bacteria.3,4 Jin et al.5 isolated seven species of LAB from commercially available Makgeolli and identified Lactobacillus paracasei and Lactobacillus arizonensis as the dominant species. Other species, including L. plantarum, L. harbinensis, L. parabuchneri, L. brevis and L. hilgardii, were less prevalent, confirming the presence of Lactobacillus strains. Similarly, Seo et al.6 reported that L. plantarum strains exhibited superior characteristics, and 16S rRNA gene sequence analysis showed high similarity with reference strains: L. plantarum RW (99% sequence similarity), L. plantarum WCFS1 (98%), and L. brevis ATCC 367 (92%). According to Kim et al.,5 LAB isolated from Makgeolli stored at 4 °C were identified as Pediococcus species, whereas at 20 °C, L. plantarum and L. brevis were predominant. Additionally, Kim et al.7 found L. crustorum and L. plantarum to be dominant, confirming that the predominant LAB in commercially available raw Makgeolli belong to the genera Lactobacillus and Pediococcus.LAB are known to support lactose digestion, modulate immune responses, reduce blood cholesterol, and synthesize vitamins, thereby improving gut health.8,9 Recent studies suggest that Makgeolli consumption can modulate the composition of gut microbiota more distinctly than other alcoholic beverages.10 Research has shown that using mixed strains of LAB is more effective than single strains in managing gastrointestinal disorders, including irritable bowel syndrome.11
Constipation is not a diagnosis but rather a symptom of gastrointestinal disorders, characterized by bowel obstruction, insufficient defecation, and incomplete evacuation.12 Typical symptoms include infrequent bowel movement, small stool volume, abdominal discomfort, bloating, and a persistent sense of incomplete evacuation.13 Prolonged constipation has also been linked to increased oxidative stress in the body.14
In addition to its probiotic content, Makgeolli has demonstrated antioxidant activity due to its fermentation-derived bioactive compounds, with enhanced effects observed when supplemented with functional ingredients such as seaweed.15
Previous studies have reported that LAB from Makgeolli possess antimicrobial, anticancer, and immune-enhancing effects,16 and that Makgeolli itself may exert anti-inflammatory and anticancer actions by inhibiting angiogenesis and adipocyte differentiation.17 However, most existing research has focused on its fermentation properties such as raw materials and organic acid composition rather than its physiological effects.1,18 Therefore, this study aims to evaluate the effects of Makgeolli on intestinal function and bowel movement using a loperamide-induced constipation model in rats, while also assessing its potential in alleviating oxidative damage associated with constipation.
Materials and methods
Materials
The experimental sample, Makgeolli, was obtained from Morning of Woopo Co., Ltd (Korea). Initial analysis showed that the raw Makgeolli contained a LAB concentration of 2.6 × 108 CFU mL−1 and a fiber content of 9.93 g per 100 g. A total of 341.57 g of fresh Makgeolli, corresponding to the full volume obtained from a single bottle, underwent lyophilization for 72 hours at −98 °C, resulting in 58.59 g of dried product, with a yield rate of 17.16%. The fiber content was maintained during the drying process, with the final powder containing 9.94 g of fiber per 100 g, as confirmed by mass balance calculations. Based on nutrient analysis conducted using a food composition database program (Can-Pro 6.0, The Korean Nutrition Society, Korea), Makgeolli typically contains carbohydrates (1.56 g per 100 mL), proteins (0.98 g per 100 mL), organic acids, polyphenols, and live lactic acid bacteria, along with dietary fiber. However, in this study, only fiber content and bacterial concentration were directly measured, as the primary focus was on its functional effects. The lyophilized powder was stored at −20 °C until further analysis. For the experimental procedures, sodium picosulfate, used as the control drug, and loperamide hydrochloride, used to induce constipation, were acquired from Sigma Chemical Company (St. Louis, MO, USA).Animals and experimental design
All animal procedures were performed in accordance with the Guidelines for the Care and Use of Laboratory Animals of Kyungnam University and approved by the Animal Ethics Committee of Kyungnam University (approval no. KUIAC_19_06). Thirty-two male Sprague-Dawley rats (aged 6 weeks; Coatech, Inc., Pyeongtaek, Korea) were acclimatized for one week on a Lab-chow diet (2018S Teklad global 18% protein rodent diet, USA). After this period, they were divided into four groups of eight, based on weight: Normal Control Group (NC), Constipation-Induced Group (LO), Control Drug Group (L-SP), and Makgeolli Group (L-MG). The NC group received sterile distilled water (DW), while the LO group was orally administered 4 mg kg−1 of loperamide for 14 days. To enhance constipation severity, loperamide (3 mg kg−1) dissolved in 0.5% Tween-20 was additionally administered subcutaneously twice daily on days 12 to 14. The loperamide dosage protocol was selected based on previous studies demonstrating effective induction of constipation in rats without excessive toxicity or weight loss.19 The L-SP group was treated with sodium picosulfate at 5 mg kg−1 dissolved in sterile DW, following previously reported effective doses for improving bowel motility in constipated animal models.20 The L-MG group was given 200 mg kg−1 of Makgeolli orally once a day. The animals were individually housed in cages within the animal laboratory and provided with their respective diets and distilled water. Throughout the study, they were kept under a 12 hour light/dark cycle at a temperature of 23 ± 2 °C and a relative humidity of 50 ± 5% (Fig. 1).Fecal number and number of fecal pellets in the distal colon
The number of fecal pellets was measured in two phases: before and during the induction of constipation. The number of fecal pellets remaining in the distal colon was assessed after the experimental animals were sacrificed.Fecal water content
To measure fecal water content, all animals were individually housed in separate cages, and fecal samples were collected on days 0 and 14 of the experiment. The feces excreted from each cage were collected at 10:00 a.m. and subsequently dried in an oven at 70 °C for 24 hours. The fecal weight was measured using an electronic balance, and the water content was calculated using the following formula:Gastrointestinal transit ratio
The gastrointestinal transit rate was evaluated on the 14th day of dietary intake, following an 18 hour fasting period. On the 15th day, the animals were orally administered the experimental diet, followed by a dose of activated charcoal diet (0.5 mL), which consisted of a 3% suspension of activated charcoal in 0.5% aqueous methylcellulose. Thirty minutes after administration, the animals were euthanized, and the gastrointestinal tract was removed. The gastrointestinal transit rate was calculated by measuring the distance traveled by the activated charcoal relative to the total length of the small intestine.Analysis of DNA damage in colonic cells
Gut harmful enzyme activity
Plasma oxygen radical absorbance capacity (ORAC) analysis
Plasma was treated with 2,2′-azobis(2-amidinopropane) dihydrochloride (AAPH, final reaction concentration of 20 nM) and the fluorescent standard, fluorescein (final reaction concentration of 40 nM), to induce the generation of peroxyl radicals. Additionally, a Trolox standard was used at a final concentration of 1 μM. The fluorescence decay rate, which indicates the reduction of peroxyl radicals, was measured every 2 minutes for 2 hours using the GENios fluorescence plate reader (FLUOstar OPTIMA, BMG Labtech, Ortenberg, Germany) at an excitation wavelength of 485 nm and an emission wavelength of 535 nm. The ORAC value was calculated by determining the total area under the curve of fluorescence decay for each sample and expressed as 1 μM Trolox equivalents (TE).Blood lipid peroxidation
Inflammatory cytokine messenger RNA (mRNA) expression in colonic tissues
cDNA was mixed with 10X Taq polymerase buffer (containing 15 mM MgCl2), 0.2 mM deoxynucleotide triphosphates (dNTPs), 0.125 U Taq polymerase, and 0.25 μM primers. Amplification was performed using a DNA thermal cycler (MyCycler TM thermal cycler, BIO-RAD, USA). The reaction conditions included an initial denaturation at 95 °C for 5 minutes, followed by denaturation at 95 °C for 15 seconds, annealing at the temperature specified in Table 1 for 30 seconds, extension at 72 °C for 40 seconds, and a final extension at 72 °C for 10 minutes. Results were expressed as relative concentrations, using β-actin as the housekeeping gene.| Primer | Product Size(bp) | Sequence | |
|---|---|---|---|
| COX-2 | R | 203 | gtgaagtgctgggcaaagaa |
| L | 203 | tgggaagctttctccaacct | |
| INOS | R | 221 | ggggttttctccacgttgtt |
| L | 221 | cttggtgaggggactggact | |
| TNF-α | R | 139 | tcttctgtctactgaacttcgg |
| L | 139 | gctacgggcttgtcactc | |
| IL-1β | R | 234 | gtgaagtcaactatgtcccg |
| L | 234 | cctgtcctgtgtgatgaaag | |
| β-action | R | 132 | cgtgcgtgacattaaagaga |
| L | 132 | ttgccgatagtgatgacct |
Statistical analysis
All statistical analyses were conducted using Statistical Package for the Social Sciences (SPSS) version 23.0 (IBM, Chicago, IL, USA). Each variable was presented as mean ± standard deviation (SD), and significance testing for mean values was conducted at a 95% confidence level (p < 0.05). Duncan's multiple-range test was employed to assess the significance of differences among intervals.Results
Weight gain, dietary intake, and dietary efficiency
The results for weight gain, dietary intake, and dietary efficiency for each group during the study period are presented in Table 2. The L-SP group, which received sodium picosulfate, experienced a significant weight reduction compared to the NC group. Although the LO and the L-MG group, which received Makgeolli, showed a trend towards weight reduction when compared to the NC group, these differences were not statistically significant. There were no significant differences in dietary intake among the groups. However, the L-MG group demonstrated significantly higher dietary efficiency compared to the LO group.| Group | NC | LO | L-SP | L-MG |
|---|---|---|---|---|
| NC: normal control, LO: loperamide, L-SP: loperamide + sodium picosulfate, L-MG: loperamide + Makgeolli. Bars with different letters are significantly different at p < 0.05 by Duncan's multiple range test. Values are the mean ± S.D. Values with different letters are significantly different at p < 0.05 after Duncan's multiple range test within group. ns: not significant.a FER : food efficiency ratio (weight gain g/food intake g). | ||||
| Weight gain (g day−1) | 6.5 ± 0.3b | 5.8 ± 0.3ab | 5.5 ± 0.2a | 6.1 ± 0.2ab |
| Food intake (g day−1) | 21.7 ± 0.7NS | 21.6 ± 0.5 | 20.5 ± 0.7 | 20.4 ± 0.4 |
| FERa (%) | 4.2 ± 0.1b | 3.8 ± 0.1a | 3.8 ± 0.1a | 4.2 ± 0.1b |
Fecal number and number of fecal pellets in the distal colon
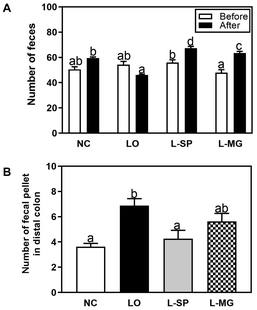
The results of the fecal number and the number of fecal pellets in the distal colon of the experimental animals induced with constipation are presented in Fig. 2. Initially, there were no significant differences in the number of fecal pellets among all groups before the induction of constipation with loperamide. However, after constipation induction, the LO group exhibited a statistically significant decrease in fecal number compared to the NC group, confirming successful induction by loperamide. Following the induction of constipation, both the L-SP and L-MG groups showed a significant increase in fecal number compared to the LO group. Regarding the number of fecal pellets in the distal colon, the LO group had significantly more fecal pellets, showing an increase of 99%, compared to the NC group. Conversely, the L-SP and L-MG groups had significant reductions in fecal pellets in the distal colon by 72.2% and 44.3%, respectively, compared to the LO group.Analysis of fecal water content
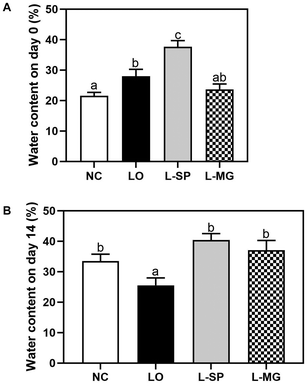
As shown in Fig. 3, fecal water content was measured using activated charcoal. Before constipation induction, there were no significant differences in fecal water content among the groups. However, after loperamide administration, the fecal water content in the LO group decreased significantly by 23.9% compared to the NC group, confirming the successful induction of constipation. In contrast, the L-SP and L-MG groups showed significantly higher fecal water content than the LO group by 20.8% and 10.8%, respectively and their levels were comparable to that of the NC group. These findings suggest that Makgeolli supplementation effectively prevented the reduction in fecal water content caused by loperamide-induced constipation.Small intestinal transit rate
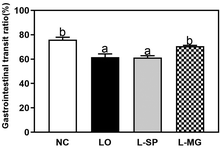
As shown in Fig. 4, the small intestinal transit rate was measured using activated charcoal. The results indicated a transit rate of 75.9% in the NC group. In contrast, the LO group, which received loperamide alone, showed a significantly reduced transit rate of 61.7%, confirming the induction of constipation. Compared to the constipated LO group, there was no significant difference in the transit rate for the L-SP group. However, the L-MG group, which received Makgeolli, exhibited a significantly increased transit rate of 70.7%. This suggests that the administration of Makgeolli can improve the small intestinal transit rate to a level similar to that of the NC group.Analysis of DNA damage in colonic cells
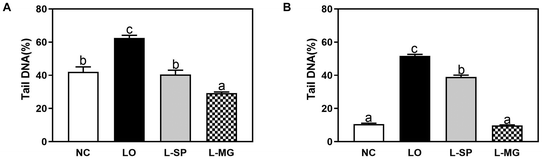
To examine changes in DNA damage within colon cells during loperamide-induced constipation, colon cells were isolated from rectal tissue and subjected to a comet assay, as shown in Fig. 5. The LO group exhibited a significant 46.3% increase in DNA damage compared to the NC group. However, the L-SP and L-MG groups showed a significant reduction in DNA damage compared to the LO group. This suggests that the antioxidants present in Makgeolli may help inhibit DNA damage in colon cells.Analysis of DNA damage in fecal water
To analyze the impact of toxic substances in residual feces on cellular damage in the colon, fecal water extracted from fecal pellets was tested on HT-29 colon cancer cells for DNA damage, as depicted in Fig. 5. HT-29 cells were treated with 75 μM H2O2 to observe significant changes in DNA damage. The LO group exhibited the highest level of DNA damage, indicating that loperamide-induced constipation causes cellular damage in the colon. In contrast, the L-SP and L-MG groups showed significant reductions in DNA damage by 119.8% and 397.3%, respectively, compared to the LO group. Impressively, the L-MG group demonstrated DNA damage inhibition levels similar to those of the NC group.Analysis of harmful intestinal enzyme activity
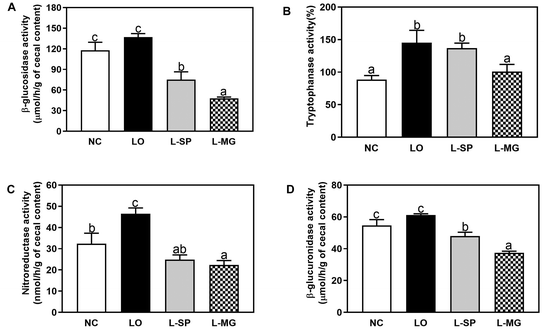
The effects of Makgeolli on the activity of harmful enzymes in the cecum of SD rats with loperamide-induced constipation were investigated. The activities of β-glucosidase, tryptophanase, nitroreductase, and β-glucuronidase in cecal extracts were analyzed, as illustrated in Fig. 6. β-Glucosidase activity was 16.3% higher in the NC group compared to the LO group treated with loperamide, although this difference was not statistically significant. The L-SP and L-MG groups showed significant reductions in β-glucosidase activity by 52.6% and 75.5%, respectively, compared to the LO group, effectively suppressing the loperamide-induced upregulation. Similarly, the activities of tryptophanase, nitroreductase, and β-glucuronidase were increased by loperamide treatment. Tryptophanase activity in the L-SP group was reduced by 9.3% compared to the LO group, but this reduction was not significant. However, nitroreductase and β-glucuronidase activities were significantly reduced by 43.8% and 12.1%, respectively. In the L-MG group, enzyme activities were significantly reduced by 75.7%, 49.1%, 74.7%, and 43.4%, respectively, compared to the LO group.Plasma ORAC analysis
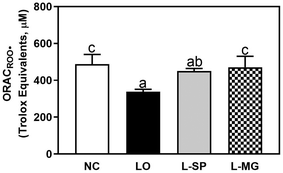
The impact on plasma antioxidant capacity was assessed by measuring plasma ORAC activity, as illustrated in Fig. 7. The LO group exhibited a 30.8% decrease in ORAC activity compared to the NC group, indicating an increase in oxidative stress in the blood due to constipation. The L-SP group showed a 23.2% decrease in ORAC activity compared to the LO group, but this was not statistically significant. In contrast, the L-MG group demonstrated a significant 27.5% increase in ORAC activity compared to the LO group.Blood lipid peroxidation (CD, TBARS)
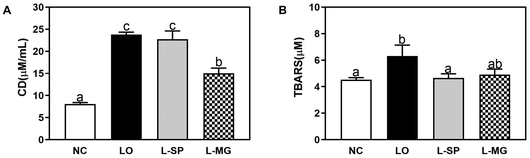
The effects on lipid peroxidation in the blood were analyzed by measuring TBARS (thiobarbituric acid reactive substances) and CD (conjugated dienes), as depicted in Fig. 8. The results for CD indicated that the LO group exhibited a significant 193.3% increase compared to the NC group. There was no significant difference between the L-SP and LO groups. However, compared to the L-MG group, the LO group showed a significant 108.9% increase. In the TBARS analysis, the LO group showed a significant 40% increase compared to the NC group, suggesting that constipation induced by loperamide promotes lipid peroxidation in the plasma. The L-SP group showed a significant decrease in TBARS compared to the LO group. In contrast, the L-MG group exhibited a 31.7% decrease compared to the LO group, although this was not statistically significant.Inflammatory cytokine mRNA expression in colonic tissues
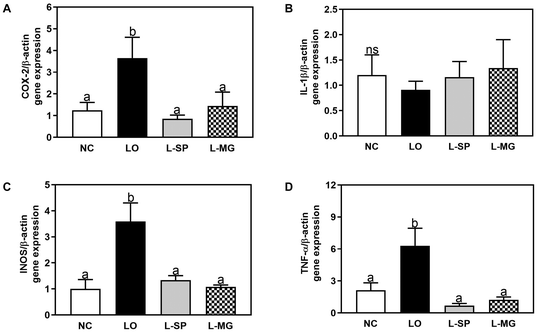
The mRNA expression levels of pro-inflammatory cytokines (COX-2, INOS, TNF-α, and IL-β) in colon tissue are shown in Fig. 9. There were no significant differences in the expression levels of IL-β among the groups. However, the LO group exhibited a significant increase in the mRNA expression levels of COX-2, INOS, and TNF-α compared to the NC group. Both the L-SP and L-MG groups showed a significant reduction in the mRNA expression levels of COX-2, INOS, and TNF-α compared to the LO group. These results suggest that the administration of Makgeolli may enhance anti-inflammatory effects in the body.Discussion
Constipation, one of the most common gastrointestinal disorders worldwide, requires clinical attention as its pharmacological treatment may lead to serious adverse effects and complications, including myocardial infarction and diarrhea.24 Therefore, food and dietary interventions are recommended for the prevention and treatment of constipation.25 Makgeolli, a traditional Korean alcoholic beverage, is rich in nutrients, lactic acid bacteria, and yeast and has been reported to promote smooth bowel movements and inhibit harmful bacteria.4,7This study examined the effects of various interventions on weight gain, food intake, and feed efficiency across different experimental groups. While no statistically significant differences were observed among the groups in these parameters, the LO group exhibited a trend toward reduced weight gain and feed efficiency. Previous research has reported that loperamide can reduce both body weight and food intake, which aligns with our findings that loperamide may contribute to decreased weight gain and feed efficiency.26 These results are consistent with existing studies, reinforcing the proposed mechanisms underlying loperamide's effects. The study analyzed stool count and colon stool content in animals with induced constipation. Before induction, there were no significant differences between groups. However, post-induction, the LO group showed a significant reduction in stool count. Loperamide, commonly used to treat diarrhea, thins the mucus layer and inhibits bowel movements, extending transit time while reducing dietary intake, leading to bloating and decreased appetite.27 The L-SP and L-MG groups showed a significant increase in stool count compared to the LO group. Sodium picosulfate, a stimulant laxative, induces colonic contractions and increases water and electrolyte secretion. It converts to its active metabolite bis-(p-hydroxyphenyl)-pyridyl-2-methane (BHPM), which stimulates peristalsis by acting directly on the colonic mucosa.28 This study also found that Makgeolli had similar effects. Makgeolli is known to contain beneficial probiotics.5–7 Previous research has identified L. paracasei and L. arizonensis as the predominant strains among the seven LAB isolated from Makgeolli.5 Other studies have also highlighted the superiority of L. plantarum strains, with L. plantarum RW (99%), L. plantarum WCFS1 (98%), and L. brevis ATCC 367 (92%) being reported.6 These findings suggest that Makgeolli by-products may have potential benefits for alleviating constipation. In this study, the Makgeolli was lyophilized before being administered to the animals. This step aimed to preserve beneficial microorganisms while minimizing potential confounding factors. Makgeolli typically contains ethanol at levels comparable to other traditional fermented rice beverages. However, lyophilization largely removes volatile compounds, including ethanol, through sublimation under low temperature and vacuum conditions. Previous studies using alcohol dilution–lyophilization methods have confirmed the effective removal of residual ethanol from fermented beverages.29 As a result, the ethanol concentration in the reconstituted Makgeolli solution used in this study was negligible, and it is unlikely to have influenced gut microbial counts or other physiological parameters.
Futhermore, Research has shown that using mixed strains of LAB such as Bifidobacterium bifidum, B. infantis, B. longum, Lactobacillus casei, L. plantarum, and L. rhamnosus can be more effective than single strains in managing gastrointestinal disorders, including irritable bowel syndrome and functional constipation.11
In the study, the LO group exhibited a significant decrease in transit rate compared to the NC group. Loperamide, known to activate μ-opioid receptors in the myenteric plexus, inhibits excitatory neurotransmission and reduces gastrointestinal motility by affecting calcium channel activation and membrane depolarization.30 Makgeolli intake significantly improved intestinal transit rate, attributable to its probiotic content. Probiotics enhance lactose absorption, boost immunity, lower blood cholesterol, and synthesize vitamins, thus improving the gut environment and inhibiting harmful bacterial growth, thereby preventing and treating diseases related to poor gut health.8,9
DNA damage is an early step in the chemical carcinogenesis process, and inhibition of DNA damage is known to be a primary defense mechanism against carcinogen-induced cancer.31 In this study, the L-MG group showed inhibition of both colon tissue and fecal water DNA damage, which is thought to be due to the bioactive compounds in rice wine, including B vitamins, organic acids, inositol, acetylcholine, and riboflavin.32 Makgeolli contains phenolic compounds such as tyrosol and ferulic acid, which are known to exhibit strong antioxidant activity by scavenging free radicals and inhibiting lipid peroxidation.33 Tyrosol also reduces cholesterol absorption and demonstrates anti-inflammatory and anticancer effects, while ferulic acid, commonly found in grains like rice and barley, contributes to antioxidant defense.34,35 These fermentation-derived compounds may explain the improved plasma antioxidant capacity observed in the L-MG group. In addition, previous studies have isolated antioxidant compounds such as tyrosol and ferulic acid from Makgeolli, which demonstrated significant radical scavenging activity in DPPH and ABTS assays.36 Moreover, Makgeolli lees were shown to activate the NRF2 pathway in vitro, increasing expression of endogenous antioxidant enzymes like SOD, GPx, and CAT,37 suggesting a potential mechanism by which Makgeolli exerts its plasma antioxidant capacity. In serum lipid peroxides, loperamide is known to induce stress and alter serum lipid levels.38 During stress, the bloodstream is characterized by a surge of catabolic hormones such as epinephrine, norepinephrine, and cortisol.39 Epinephrine is known to promote lipid breakdown.40 Constipation can induce systemic oxidative stress by prolonging fecal retention in the colon, leading to increased production and absorption of harmful metabolites. This effect may be further exacerbated by the depletion of antioxidant nutrients such as vitamins C and E, and by reduced activity of antioxidant enzymes like superoxide dismutase (SOD) and catalase (CAT), ultimately may promote lipid peroxidation.41 These mechanisms are consistent with the elevated levels of serum lipid peroxidation observed in the LO group in this study. The results of this study are consistent with the observed decrease in serum lipid levels in the LO group.
We analyzed the impact on harmful enzyme activity in the intestine, specifically β-glucosidase, tryptophanase, nitroreductase, and β-glucuronidase. The activity of these enzymes, produced by intestinal microbes, is closely associated with the circulation of toxic and carcinogenic substances.42 Specifically, colonic β-glucosidase and tryptophanase have been linked to the onset of colorectal cancer.43 Previous studies have reported that certain bacteria can reduce levels of intestinal toxins and improve gut function.44 Furthermore, the suppression of harmful enzyme activity has been associated with enhanced gastrointestinal function and relief from constipation.45 For example, nitroreductase reduces nitrate to nitrite, which can react with amines or amides in food to form N-nitroso compounds—potential carcinogens capable of inducing DNA mutations.46 Likewise, β-glucuronidase, mainly produced by harmful bacteria such as Clostridium perfringens, E. coli, Bacteroides, Eubacterium and Peptostreptococcus, contributes to gut inflammation and carcinogenesis by generating toxic metabolites.47 The consumption of Makgeolli demonstrated an effect of inhibiting these harmful enzyme activities, thereby improving gut function. In our study, Makgeolli intake effectively suppressed the activity of these harmful enzymes, thereby improving gut function.48 Although β-glucosidase is also produced by probiotic bacteria and may play a beneficial role under eubiotic conditions, its excessive production under dysbiotic conditions such as those seen in colorectal cancer can lead to the accumulation of toxic metabolites and carcinogens.49 Previous studies support this dual role: Jeon et al. (2002)49 demonstrated that certain probiotic bacteria can produce β-glucosidase and hydrolyze isoflavone glycosides during fermentation, highlighting its beneficial effects under normal gut conditions. In contrast, Cheng et al. (2022) reported that excessive activity of β-glucuronidase a functionally similar microbial enzyme under dysbiotic conditions promotes the accumulation of toxic metabolites and the development of colorectal cancer.50 Based on these findings, it is reasonable to suggest that β-glucosidase, when excessively activated under dysbiotic conditions, could similarly contribute to harmful metabolite and carcinogen production. Therefore, the reduced β-glucosidase activity observed in the L-MG group is likely due to a decrease in harmful enzyme activity associated with microbial imbalance, rather than an inhibition of beneficial bacteria. This suggests that Makgeolli has potential benefits in reducing harmful enzymatic activities in the gut, which may contribute to overall gut health and the alleviation of constipation.
The results of mRNA expression for inflammatory cytokines (COX-2, INOS, TNF-α, and IL-β) in colon tissues indicated an increase in expression levels in the LO group compared to the NC group, suggesting that intestinal inflammation induced by loperamide is confirmed by the increased expression of these cytokines. The low expression level of IL-1β observed in the LO group may be associated with its time-dependent expression pattern, in which IL-1β is rapidly upregulated during the early phase of inflammation and subsequently decreases as the inflammatory response progresses toward resolution. Conversely, the L-SP and L-MG groups showed a significant reduction in the expression levels of COX-2, INOS, and TNF-α compared to the LO group. These findings imply that the consumption of Makgeolli may enhance anti-inflammatory effects in the body and positively influence constipation control. According to previous studies, similar mild inflammation and activation of immune responses have been observed in patients with irritable bowel syndrome (IBS).51 Although interpreting the correlation between intestinal permeability and colonic inflammation is complex due to potential contributions from small intestinal permeability, it is noted that upper gastrointestinal motility is impaired in patients with constipation. This inflammatory response may also be relevant to those suffering from constipation.52 It has been reported that pharmacological agents like loperamide, which induce delayed intestinal transit and constipation, can lead to changes in the gut microbiota.53 Gastrointestinal diseases are closely associated with gut microbiota, and the regulation of this microbiota plays a significant role in the progression of gastrointestinal disorders.54 The human gut harbors a diverse array of bacteria that maintain symbiotic or antagonistic relationships, forming an ecosystem predominantly composed of anaerobes.55 Previous research indicates that Lactobacilli and Bifidobacteria, known for their beneficial effects such as immune enhancement and digestive promotion, have the ability to stabilize gut microbiota.56 The increased expression of inflammatory cytokines observed in this study suggests potential interactions with inflammatory signaling pathways such as the NF-κB pathway. This may provide important clues for future research to elucidate the pathological mechanisms of inflammation and constipation, warranting further investigation into the interactions between gut microbiota and inflammatory cytokines.
Conclusion
In this study, we investigated the effects of Makgeolli on improving bowel function and defecation activity in loperamide-induced constipated SD rats over a period of 14 days. The L-SP group received sodium picosulfate, and the L-MG group received Makgeolli. Measured parameters included improvements in constipation, DNA damage, harmful enzyme activity in the intestine, lipid peroxides in the blood, and the expression of pro-inflammatory cytokine mRNA in colon tissue. The results showed that after constipation was induced, the L-MG group had a significant increase in fecal output and a significant decrease in the number of fecal pellets in the intestine compared to the NC group. Analysis of DNA damage in colon tissue and fecal water on HT-29 colon cancer cells revealed that Makgeolli consumption reduced DNA damage that was increased due to constipation. Tryptophanase activity in the L-MG group was similar to that of the NC group, indicating an inhibitory effect on harmful enzymes. Furthermore, the impact of constipation-induced intestinal damage on blood lipid peroxides was analyzed. The LO group showed a significant increase in TBARS and CD levels compared to the NC group, confirming that constipation promotes lipid peroxidation in plasma. In contrast, the L-MG group exhibited an improvement in lipid peroxidation. The mRNA expression analysis of pro-inflammatory cytokines in colon tissue showed no significant differences in IL-β expression levels among the groups. However, COX-2, INOS, and TNF-α expression levels were significantly increased in the LO group compared to the NC group. In contrast, the L-SP and L-MG groups showed significant reductions in the expression levels of COX-2, INOS, and TNF-α compared to the LO group. These results suggest that Makgeolli significantly improves constipation and enhances bowel function in loperamide-induced constipated animals. Consequently, Makgeolli has the potential to be considered a functional food for preventing constipation.Author contributions
Soyoung Jung: conceptualization, methodology, investigation, formal analysis, writing – original Draft. Yunjung Lee: investigation, data curation, validation. Yoo Kyoung Park: conceptualization, methodology, writing – review & editing, supervision, project administration. Eunju Park: conceptualization, methodology, writing – review & editing, supervision, project administration.Conflicts of interest
The authors declare no conflict of interest.Data availability statement
The data are available from the corresponding author on reasonable request.Acknowledgements
This work was supported by the Technology Development Program (S2718916) funded by the Ministry of SMEs and Startups (MSS, Korea). This work was supported by the BK21 Plus Program “AgeTech-Service Convergence Major” through the National Research Foundation (NRF) funded by the Ministry of Education of Korea [5120200313836].References
- J.-S. Lee, T.-S. Lee, S.-O. Park and B.-S. Noh, Flavor components in mash of takju prepared by different raw materials, Korean J. Food Sci. Technol., 1996, 28(2), 316–323 Search PubMed.
- S. J. Rhee, C.-Y. J. Lee, K. K. Kim and C.-H. Lee, Comparison of the traditional (Samhaeju) and industrial (Chongju) rice wine brewing in Korea, Food Sci. Biotechnol., 2003, 12(3), 242–247 Search PubMed.
- G. Lee, Characteristic and new technology of Korean Takju, Korean J. Appl. Microbial. Bioeng., 1994, 7, 36–46 Search PubMed.
- S.-E. Jung and S.-H. Kim, Probiotic properties of lactic acid bacteria isolated from commercial raw makgeolli, Korean J. Food Sci. Technol., 2015, 47(1), 44–50 Search PubMed.
- J. Jin, S.-Y. Kim, Q. Jin, H.-J. Eom and N.-S. Han, Diversity analysis of lactic acid bacteria in Takju, Korean rice wine, J. Microbiol. Biotechnol., 2008, 18(10), 1678–1682 Search PubMed.
- D.-H. Seo, J.-H. Jung, H.-Y. Kim, Y.-R. Kim, S.-J. Ha and Y.-C. Kim, et al., Identification of lactic acid bacteria involved in traditional Korean rice wine fermentation, Food Sci. Biotechnol., 2007, 16(6), 994–998 Search PubMed.
- Y.-H. Kim, J.-H. Min, M.-G. Kang, J.-H. Kim, B.-H. Ahn and H.-K. Kim, et al., Physicochemical properties, lactic acid bacteria content and physiological functionalities of Korean commercial Makgeolli, Microbiol. Biotechnol. Lett., 2012, 40(4), 325–332 CrossRef.
- S. Y. Park, G. E. Ji, Y. T. Ko, H. K. Jung, Z. Ustunol and J. J. Pestka, Potentiation of hydrogen peroxide, nitric oxide, and cytokine production in RAW 264.7 macrophage cells exposed to human and commercial isolates of Bifidobacterium, Int. J. Food Microbiol., 1999, 46(3), 231–241 CrossRef PubMed.
- S. Salminen, A. von Wright, L. Morelli, P. Marteau, D. Brassart and W. M. de Vos, et al., Demonstration of safety of probiotics—a review, Int. J. Food Microbiol., 1998, 44(1–2), 93–106 CrossRef PubMed.
- J. Cha, S.-E. Park, E.-J. Kim, S.-H. Seo, S. W. Roh and T. W. Whon, et al., Consumption of Korean rice wine alters certain human gut microbial genera in different ways from consumption of other alcoholic beverages: A randomized, cross-over trial, LWT, 2024, 200, 116152 CrossRef.
- N.-L.-H. Bekkali, M. E. Bongers, M. M. Van den Berg, O. Liem and M. A. Benninga, The role of a probiotics mixture in the treatment of childhood constipation: a pilot study, Nutrition journal, 2007, 6, 1–6 CrossRef PubMed.
- C. Koebnick, I. Wagner, P. Leitzmann, U. Stern and H. F. Zunft, Probiotic beverage containing Lactobacillus casei Shirota improves gastrointestinal symptoms in patients with chronic constipation, Can. J. Gastroenterol. Hepatol., 2003, 17(11), 655–659 Search PubMed.
- Chronic constipation, ed. A. E. Bharucha and A. Wald, Mayo Clinic Proceedings, Elsevier, 2019 Search PubMed.
- M. Camilleri, W. G. Thompson, J. W. Fleshman and J. H. Pemberton, Clinical management of intractable constipation, Ann. Intern. Med., 1994, 121(7), 520–528 CrossRef CAS PubMed.
- A. Vermorken, E. Andrès and Y. Cui, chronic constipation–a warning sign for oxidative stress?, Aliment. Pharmacol. Ther., 2015, 42(3), 385–386 CrossRef CAS PubMed.
- J.-H. Yun, D.-S. Yim, J.-Y. Kang, B.-Y. Kang, E.-A. Shin and M.-J. Chung, et al., Identification of Lactobacillus ruminus SPM0211 isolated from healthy Koreans and its antimicrobial activity against some pathogens, Arch. Pharmacal Res., 2005, 28(6), 660–666 CrossRef CAS.
- L. S.-J. Lee, J. H. Kim, Y. W. Jung, S. Park, W. C. Shin and C. S. Park, et al., Composition of organic acids and physiological functionality of commercial Makgeolli, Korean J. Food Sci. Technol., 2011, 43(2), 206–212 CrossRef.
- J. C. Song and H. J. Park, Takju brewing using the uncooked germed brown rice at second stage mash, J. Korean Soc. Food Sci. Nutr., 2003, 32(6), 847–854 CrossRef CAS.
- O. Adeniyi, M. Edache, I. Abi and R. Ediale, Ameliorative Effects of Virgin Coconut Oil in Loperamide Induced Constipation in Wistar Rats, Gut, 2020, 1, 2 Search PubMed.
- Y. N. Dey, M. M. Wanjari, B. Srivastava, D. Kumar, D. Sharma and J. Sharma, et al., Beneficial effect of standardized extracts of Amorphophallus paeoniifolius tuber and its active constituents on experimental constipation in rats, Heliyon, 2020, 6(5), e04023 CrossRef PubMed.
- P. S. Kanna, C. Mahendrakumar, B. Indira, S. Srivastawa, K. Kalaiselvi and T. Elayaraja, et al., Chemopreventive effects of vanadium toward 1, 2−dimethylhydrazine–induced genotoxicity and preneoplastic lesions in rat colon, Environ. Mol. Mutagen., 2004, 44(2), 113–118 CrossRef CAS PubMed.
- D.-H. Kim, H.-J. Kang, S.-W. Kim and K. Kobashi, pH-inducible β-glucosidase and β-glucuronidase of intestinal bacteria, Chem. Pharm. Bull., 1992, 40(6), 1667–1669 CrossRef CAS PubMed.
- D.-H. Kim, J.-H. Lee, E.-A. Bae and M. J. Han, Induction and inhibition of indole production of intestinal bacteria, Arch. Pharmacal Res., 1995, 18(5), 351–355 CrossRef CAS.
- J.-F. Zhou, J.-G. Lou, S.-L. Zhou and J.-Y. Wang, Potential oxidative stress in children with chronic constipation, World J. Gastroenterol., 2005, 11(3), 368 CrossRef PubMed.
- C. Cirillo and R. Capasso, Constipation and botanical medicines: an overview, Phytother. Res., 2015, 29(10), 1488–1493 CrossRef.
- Y. A. Chumakova, V. Bashkatova and S. Sudakov, Changes in feeding behavior after peripheral loperamide administration in rats, Bull. Exp. Biol. Med., 2011, 150(4), 398–400 CrossRef CAS.
- A. Shimotoyodome, S. Meguro, T. Hase, I. Tokimitsu and T. Sakata, Decreased colonic mucus in rats with loperamide-induced constipation, Comp. Biochem. Physiol., Part A:Mol. Integr. Physiol., 2000, 126(2), 203–212 CrossRef.
- D. Krueger, I. Demir, G. Ceyhan, F. Zeller and M. Schemann, bis-(p-hydroxyphenyl)-pyridyl-2-methane (BHPM)—the active metabolite of the laxatives bisacodyl and sodium picosulfate—enhances contractility and secretion in human intestine in vitro, Neurogastroenterol. Motil., 2018, 30(7), e13311 CrossRef PubMed.
- D. Shirane, H. Tanaka, Y. Sakurai, S. Taneichi, Y. Nakai and K. Tange, et al., Development of an alcohol dilution–lyophilization method for the preparation of mRNA-LNPs with improved storage stability, Pharmaceutics, 2023, 15(7), 1819 CrossRef.
- P. Holzer, Opioid receptors in the gastrointestinal tract, Regul. Pept., 2009, 155(1–3), 11–17 CrossRef PubMed.
- S. Barth, C. Fähndrich, A. Bub, H. Dietrich, B. Watzl and F. Will, et al., Cloudy apple juice decreases DNA damage, hyperproliferation and aberrant crypt foci development in the distal colon of DMH-initiated rats, Carcinogenesis, 2005, 26(8), 1414–1421 CrossRef PubMed.
- J.-Y. Kim, K.-W. Sung, H.-W. Bae and Y.-H. Yi, pH, acidity, color, reducing sugar, total sugar, alcohol and organoleptic characteristics of puffed rice powder added Takju during fermentation, Korean J. Food Sci. Technol., 2007, 39(3), 266–271 Search PubMed.
- J.-H. Ha, D.-U. Lee, J.-T. Lee, J.-S. Kim, C.-S. Yong and J.-A. Kim, et al., 4-Hydroxybenzaldehyde from Gastrodia elata B1. is active in the antioxidation and GABAergic neuromodulation of the rat brain, J. Ethnopharmacol., 2000, 73(1–2), 329–333 CrossRef PubMed.
- R. M. Lamuela-Raventós, E. Gimeno, M. Fitó, A.-I. Castellote, M. Covas and M. C. De La Torre-Boronat, et al., Interaction of olive oil phenol antioxidant components with low-density lipoprotein, Biol. Res., 2004, 37(2), 247–252 Search PubMed.
- A. Ramos-Comerzana and M. Monteoliva-Sánchez, Biopharmaceutical potencial of olive industrial wastes, Ars Pharm., 2000, 41(1), 129–136 Search PubMed.
- S. Wang, H. Lee, J. Cho, K. Park and J. Moon, Isolation and identificati on of antioxidants from makge olli, Han'guk Sikp'um Kwahakhoechi, 2012, 44, 14–20 Search PubMed.
- M. Jeon and N. Rahman, Cytoprotective effect of Makgeolli lees on paraquat induced oxidative stress in A549 cells via activation of NRF2 and antioxidant genes, J. Microbiol. Biotechnol., 2016, 26(2), 277–286 CrossRef CAS.
- J. E. Kim, Y. J. Choi, S. J. Lee, J. E. Gong, Y. J. Lee and J. E. Sung, et al., Antioxidant activity and laxative effects of tannin-enriched extract of Ecklonia cava in loperamide-induced constipation of SD rats, PLoS One, 2021, 16(2), e0246363 CrossRef CAS.
- I. Brenner, J. Zamecnik, P. Shek and R. Shephard, The impact of heat exposure and repeated exercise on circulating stress hormones, Eur. J. Appl. Physiol. Occup. Physiol., 1997, 76, 445–454 CrossRef CAS.
- U. Freyschuss, P. Hjemdahl, A. Juhlin-Dannfelt and B. Linde, Cardiovascular and metabolic responses to low dose adrenaline infusion: an invasive study in humans, Clin. Sci., 1986, 70(2), 199–206 CrossRef CAS.
- J.-F. Zhou, J.-G. Lou, S.-L. Zhou and J.-Y. Wang, Potential oxidative stress in children with chronic constipation, World J. Gastroenterol., 2005, 11(3), 368 CrossRef PubMed.
- C. Guerin-Danan, C. Chabanet, C. Pedone, F. Popot, P. Vaissade and C. Bouley, et al., Milk fermented with yogurt cultures and Lactobacillus casei compared with yogurt and gelled milk: influence on intestinal microflora in healthy infants, Am. J. Clin. Nutr., 1998, 67(1), 111–117 CrossRef CAS PubMed.
- K.-T. Chung, G. E. Fulk and M. W. Slein, Tryptophanase of fecal flora as a possible factor in the etiology of colon cancer, J. Natl. Cancer Inst., 1975, 54(5), 1073–1078 CrossRef CAS.
- E. A. Griffiths, L. C. Duffy, F. L. Schanbacher, H. Qiao, D. Dryja and A. Leavens, et al., In vivo effects of bifidobacteria and lactoferrin on gut endotoxin concentration and mucosal immunity in Balb/c mice, Dig. Dis. Sci., 2004, 49, 579–589 CrossRef CAS.
- H. M. An, E. H. Baek, S. Jang, D. K. Lee, M. J. Kim and J. R. Kim, et al., Efficacy of Lactic Acid Bacteria (LAB) supplement in management of constipation among nursing home residents, Nutr. J., 2010, 9, 1–7 CrossRef PubMed.
- R. Hughes, A. Cross, J. Pollock and S. Bingham, Dose-dependent effect of dietary meat on endogenous colonic N-nitrosation, Carcinogenesis, 2001, 22(1), 199–202 CrossRef CAS.
- M. Mroczynska and Z. Libudzisz, Beta-glucuronidase and beta-glucosidase activity of Lactobacillus and Enterococcus isolated from human feces, Pol. J. Microbiol., 2010, 59(4), 265–269 CAS.
- M. Sharma and G. Shukla, Administration of metabiotics extracted from probiotic Lactobacillus rhamnosus MD 14 inhibit experimental colorectal carcinogenesis by targeting Wnt/β-catenin pathway, Front. Oncol., 2020, 10, 746 CrossRef PubMed.
- K.-S. Jeon, G.-E. Ji and I.-K. Hwang, Assay of β-glucosidase activity of bifidobacteria and the hydrolysis of isoflavone glycosides by Bifidobacterium sp. Int-57 in soymilk fermentation, J. Microbiol. Biotechnol., 2002, 12(1), 8–13 CAS.
- K.-W. Cheng, C.-H. Tseng, I.-J. Chen, B.-C. Huang, H.-J. Liu and K.-W. Ho, et al., Inhibition of gut microbial β-glucuronidase effectively prevents carcinogen-induced microbial dysbiosis and intestinal tumorigenesis, Pharmacol. Res., 2022, 177
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) , 106115 CrossRef CAS.
, 106115 CrossRef CAS. - V. S. Chadwick, W. Chen, D. Shu, B. Paulus, P. Bethwaite and A. Tie, et al., Activation of the mucosal immune system in irritable bowel syndrome, Gastroenterology, 2002, 122(7), 1778–1783 CrossRef.
- M. V. Penning, J. B. V. M. Delemarre and A. A. M. Masclee, Proximal gastric motor and, sensory function in slow transit constipation, Scand. J. Gastroenterol., 2001, 36(12), 1267–1273 CrossRef.
- P. C. Kashyap, A. Marcobal, L. K. Ursell, M. Larauche, H. Duboc and K. A. Earle, et al., Complex interactions among diet, gastrointestinal transit, and gut microbiota in humanized mice, Gastroenterology, 2013, 144(5), 967–977 CrossRef.
- Y. Deng, M. Li, L. Mei, L. Cong, Y. Liu and B. Zhang, et al., Manipulation of intestinal dysbiosis by a bacterial mixture ameliorates loperamide-induced constipation in rats, Benefic. Microbes, 2018, 9(3), 453–464 CAS.
- T. Mitsuoka, Bifidobacteria and their role in human health, J. Ind. Microbiol., 1990, 6, 263–267 CrossRef.
- K. Kimura, A. L. McCartney, M. A. McConnell and G. W. Tannock, Analysis of fecal populations of bifidobacteria and lactobacilli and investigation of the immunological responses of their human hosts to the predominant strains, Appl. Environ. Microbiol., 1997, 63(9), 3394–3398 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2025 |