Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Improvement of vascular endothelial function by intake of lycopene-rich tomato juice in healthy adults: a randomized, placebo-controlled, double-blind, parallel-group comparative study
Kazutaka
Yoshida
 *,
Yuichiro
Nakazawa
*,
Yuichiro
Nakazawa
 ,
Shingo
Takahashi
,
Shingo
Takahashi
 and
Shigenori
Suzuki
and
Shigenori
Suzuki
Diet & Well-being Research Institute, Kagome Co., Ltd, 329-2762, 17 Nishitomiyama, Nasushiobara, Tochigi, Japan. E-mail: Kazutaka_Yoshida@kagome.co.jp
First published on 12th September 2025
Abstract
Maintaining normal vascular endothelial function is important for preventing arteriosclerosis. Flow-mediated dilatation (FMD) is widely used to evaluate vascular endothelial function. Lycopene, a carotenoid abundant in tomatoes, has been reported to improve FMD, but its effect after 4 weeks of intake remains unclear. We conducted a randomized, placebo-controlled, double-blind, parallel-group trial to assess the effects of lycopene. Seventy-five healthy adults with borderline FMD (4–7%) were randomly assigned to placebo juice (0.7 mg lycopene), tomato juice (TJ, 15.0 mg), or high-lycopene tomato juice (HLTJ, 26.7 mg) groups. FMD, serum lycopene, oxidative stress markers (malondialdehyde, 8-hydroxy-2′-deoxyguanosine, 8-iso-prostaglandin F2α), and nitrogen oxide concentrations were measured every 4 weeks over a 12-week intervention. The participants analyzed included 23 (placebo), 25 (TJ), and 24 (HLTJ). Data were analyzed using a linear mixed effects model adjusting for baseline covariates and a post hoc Dunnett's test with Bonferroni correction for intergroup comparisons. At week 12, FMD was significantly higher in the TJ (6.1 ± 0.5%, p < 0.001) and HLTJ groups (7.0 ± 0.7%, p < 0.001) than in the placebo group (5.4 ± 0.6%). In the HLTJ group, FMD at weeks 4 (6.2 ± 1.0%, p < 0.001) and 8 (6.7 ± 0.7%, p < 0.001) was significantly higher than in the placebo group (5.4 ± 0.9% and 5.3 ± 0.6%, respectively). Serum lycopene concentrations were significantly increased in TJ and HLTJ groups than in the placebo group. No significant differences were observed in oxidative stress or nitrogen oxide levels. These findings suggest that lycopene intake improves vascular endothelial function, though the mechanism remains unclear. Trial registration: UMIN000051176.
Introduction
The prevalence of cardiovascular disease (CVD) is increasing globally, and trends over the past decade have indicated a worsening of this burden. CVD is the largest contributor to the global disease burden, and atherosclerotic diseases are the primary mediators of CVD burden and trends.1 Atherosclerotic disease is the major cause of death in Japan, and the Japan Atherosclerosis Society published the Guidelines for Prevention of Atherosclerotic Cardiovascular Diseases in 2022 to prevent atherosclerotic CVD, which presents non-invasive diagnostic methods for arteriosclerosis prevention.2 Vascular endothelial function is one of them and is evaluated by measuring changes in forearm blood flow and the diameter of the brachial artery in response to endothelium-dependent increases in blood flow caused by drugs such as acetylcholine and reactive hyperemia after forearm ejection. One of the most commonly used techniques is flow-mediated dilatation (FMD), which is used to evaluate the degree of brachial artery dilation caused by reactive hyperemia after 5 min of restricted forearm blood flow. Given that FMD declines in the early stages of atherosclerosis,3,4 it is useful for the initial assessment of atherosclerotic CVDs. Normal FMD is ≥7%; 4–7% is borderline; and <4% is abnormal.5Lycopene is a carotenoid primarily found in tomatoes and tomato products, with strong singlet oxygen scavenging activity.6 Consumption of tomato-based products has been shown to be inversely associated with the risk of CVD in a cohort study.7 Another cohort study reported that lycopene intake was inversely associated with the incidence of CVD.8 Lycopene exhibits anti-inflammatory properties9 and inhibits the migration of endothelial cells by inhibiting the expression of vascular endothelial growth factor and increasing NO production.10 A human interventional study reported that consuming 70 g of tomato paste containing 33 mg of lycopene for 15 days significantly increased the FMD.11 Additionally, the consumption of tomato juice (intake amount was not specified) for 2 months significantly increased FMD.12 In addition, a meta-analysis that integrated three interventional studies evaluating the effect of continuous intake (for ≥1 week) of tomato-containing foods on FMD showed that tomato intake significantly increased FMD.13 In contrast, the ingestion of 70 g of tomato puree containing 46.2 mg of lycopene for 1 week resulted in no significant increase in FMD.14 Additionally, the ingestion of 80 g of tomato paste for 1 week (the amount of lycopene was not specified) resulted in no significant increase in postprandial FMD.15 To date, no placebo-controlled studies have investigated the effects of pure lycopene on FMD.
The effects of lycopene intake on FMD were significantly affected by the amount and duration of lycopene intake. The guidance issued by the European Food Safety Authority (EFSA), “Guidance for the scientific requirements for health claims related to antioxidants, oxidative damage, and cardiovascular health”, states that an increase in fasting FMD caused by the continuous consumption of food ingredients for more than 4 weeks has a beneficial physiological effect on vascular endothelial function.16 Considering these reports, it would be meaningful to clarify the effect of lycopene on vascular endothelial function by evaluating the effect of lycopene intake for ≥4 weeks on FMD. In addition, there have been no placebo-controlled studies evaluating the effect. Therefore, we conducted a randomized, placebo-controlled, double-blind, parallel-group comparison study in healthy adults with borderline FMD values to evaluate the change in FMD based on the long-term (≥4 weeks) intake of lower doses of lycopene compared to those in previous reports. In addition, the effects of lycopene on oxidative stress markers and nitrogen oxides (NOx), which may be related to the mechanism of lycopene function, were evaluated.
Methods
Study overview
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
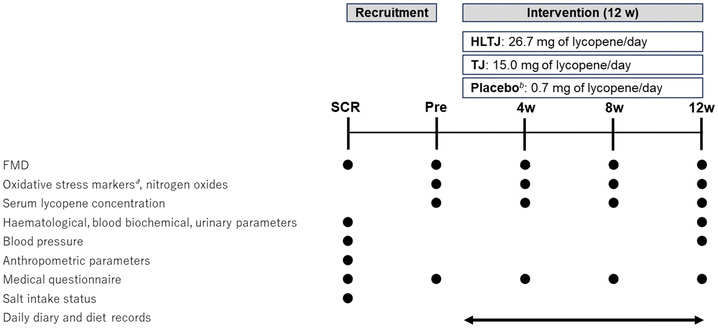
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1. Each participant consumed placebo juice containing 0.7 mg lycopene, TJ containing 15.0 mg lycopene, or HLTJ containing 26.7 mg lycopene for 12 weeks. Participants visited the hospital at 4, 8, and 12 weeks after starting the intervention, and data on FMD, oxidative stress markers, NOx, serum lycopene concentrations, virus tests, and medical questionnaires were collected. Hematological, blood biochemical and urinary parameters and blood pressure were measured at 12 weeks. The participants maintained their daily diaries and dietary records during the intervention period.
1. Each participant consumed placebo juice containing 0.7 mg lycopene, TJ containing 15.0 mg lycopene, or HLTJ containing 26.7 mg lycopene for 12 weeks. Participants visited the hospital at 4, 8, and 12 weeks after starting the intervention, and data on FMD, oxidative stress markers, NOx, serum lycopene concentrations, virus tests, and medical questionnaires were collected. Hematological, blood biochemical and urinary parameters and blood pressure were measured at 12 weeks. The participants maintained their daily diaries and dietary records during the intervention period.
Participants
The exclusion criteria were as follows: 1, subjects with a chronic disease and were undergoing treatment; 2, subjects with a history or current medical history of serious liver, kidney, gastrointestinal, heart, respiratory, endocrine, thyroid, adrenal, or other metabolic disease; 3, subjects diagnosed with arrhythmia during the SCR; 4, subjects who were judged as unsuitable for this study based on the SCR; 5, subjects with a history of gastrointestinal disease or gastrointestinal surgery that affected digestion and absorption; 6, subjects with allergies to drugs or foods (particularly test foods); 7, subjects who did not stop taking any supplement or a healthy food that may affect vascular endothelial function or blood pressure during the study period; 8, subjects who consumed tomato (≥40 g) or tomato products (e.g., ≥100 mL tomato juice or meals with a lot of tomato ketchup or tomato sauce) for ≥3 days a week; 9, subjects from whom >200 mL of blood was collected within 1 month or those from whom 400 mL of blood was collected within 3 months prior to the date of providing informed consent; 10, subjects with a history of drug dependence or alcohol dependence; 11, subjects who consumed ≥20 g of pure alcohol per day for ≥3 days a week; 12, subjects who smoked ≥21 cigarettes per day on an average (including e-cigarettes); 13, subjects who were shift workers or late night workers; 14, subjects who were pregnant and subjects who intended to conceive during the study period, and breastfeeding patients; 15, subjects who were participating in research involving other foods or medicines, subjects who used other study cosmetics or medicines, subjects who had participated in other studies within 1 month prior to the date of informed consent, and subjects who were willing to participate in other studies; and, 16, subjects who were considered unsuitable for this study by the investigators or researchers.
Dietary intervention
| Placebo | TJ | HLTJ | |
|---|---|---|---|
| Values represent the content per 190 g serving. TJ, tomato juice; HLTJ, high-lycopene tomato juice. | |||
| Energy, kcal | 45.6 | 36.1 | 30.4 |
| Protein, g | 2.3 | 1.5 | 1.5 |
| Fat, g | N. D. | N. D. | N. D. |
| Carbohydrate, g | 9.3 | 7.8 | 6.8 |
| Sugars, g | 9.1 | 6.8 | 5.7 |
| Dietary fiber, g | 0.2 | 1.0 | 1.1 |
| Lycopene, mg | 0.7 | 15.0 | 26.7 |
Data collection
The arm to be measured for each participant, i.e., left or right, was determined at the time of the SCR, and measurements were made on the same arm during the study period.
Endpoints
Safety analysis
Safety analysis was based on the incidence of side effects and adverse events during the intervention period and the results of hematological, blood biochemical, and urinary examinations.Statistical analysis
Data were expressed as mean value ± standard deviation, and EZR (Ver. 1.61)21 was used for statistical analysis. We used a linear mixed-effects model (LMM) to examine the effects of time and intervention on the outcome variable, while adjusting for baseline measurements. The model included the fixed effects of time (Pre, 4, 8, and 12 weeks), intervention group (Placebo, TJ, HLTJ), and baseline outcome values. A random intercept was included to account for individual variability across subjects. If a significant effect was detected by LMM, estimated marginal means were calculated and a post hoc Dunnett's test for intergroup comparisons between the placebo and TJ groups was performed. Bonferroni correction was applied to adjust the p-value for each comparison because of 3 comparisons by Dunnett's test. Specifically, the original p-value was tripled and adjusted p-values are reported for each test. All tests were performed at a 5% significance level.Results
Flow diagram and participant characteristics
Fig. 2 shows a flow diagram of the study participants. A total of 400 potential participants were screened for eligibility, and 65 were excluded because they did not meet the inclusion criteria. Furthermore, the top 150 patients were selected from among those with FMD ranging from 3% to 7%, and a pre-intervention examination was conducted. Five individuals who declined to participate in the study were excluded, and 75 participants with FMD ≥4% were selected in the ascending order of FMD. They were randomly allocated to the placebo (n = 25), TJ (n = 25), and HLTJ (n = 25) groups. Two participants (one in the placebo group and one in the HLTJ group) were excluded for personal reasons (injuries unrelated to the study), and 73 participants completed the study. One participant did not comply with the restrictions (intake of healthy foods that can affect vascular endothelial function), and 72 participants (placebo group, n = 23; TJ group, n = 25; HLTJ group, n = 24) were included in the analysis. The rate of adherence to the experimental food was 100% in all groups. The participant characteristics during the SCR examination were not significantly different between the groups (Table 2).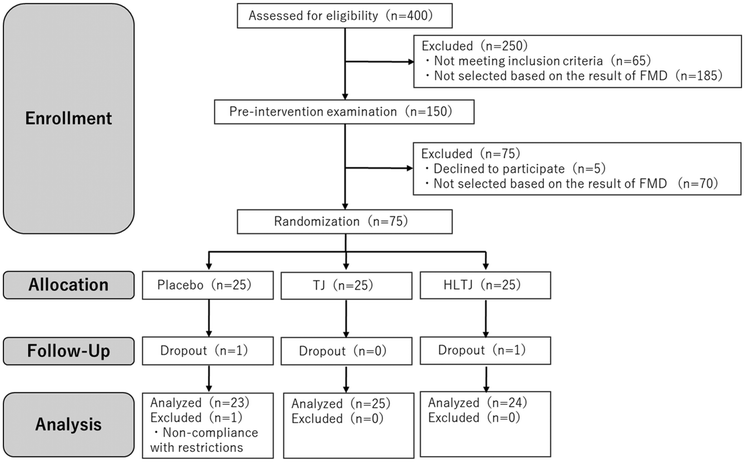 | ||
| Fig. 2 Flow diagram of the study participants. TJ, tomato juice; HLTJ, high-lycopene tomato juice; FMD, flow-mediated dilatation. | ||
| Placebo | TJ | HLTJ | |||
|---|---|---|---|---|---|
| Data are measured at the timing of SCR and expressed as numbers or mean ± standard deviation. The p-value is a comparison with placebo (Dunnett's test). TJ, tomato juice; HLTJ, high-lycopene tomato juice; M, male; F, female; BMI, body mass index; SBP, systolic blood pressure; DBP, diastolic blood pressure; FMD, flow-mediated dilatation. | |||||
| n | 23 | 25 | 24 | ||
| M/F | 9/14 | 9/16 | 8/16 | ||
| Age, years | 54.1 ± 6.6 | 54.6 ± 5.3 | p = 0.921 | 54.7 ± 5.2 | p = 0.915 |
| Height, cm | 161.8 ± 7.9 | 164.7 ± 8.9 | p = 0.363 | 160.6 ± 7.8 | p = 0.827 |
| Weight, kg | 59.0 ± 10.5 | 65.4 ± 13.8 | p = 0.135 | 60.9 ± 12.4 | p = 0.811 |
| BMI, kg m−2 | 22.4 ± 2.7 | 23.9 ± 3.6 | p = 0.176 | 23.4 ± 3.2 | p = 0.436 |
| SBP, mmHg | 123.7 ± 10.0 | 123.9 ± 10.7 | p = 0.996 | 124.1 ± 10.2 | p = 0.986 |
| DBP, mmHg | 77.2 ± 8.4 | 76.7 ± 9.0 | p = 0.970 | 75.1 ± 10.0 | p = 0.662 |
| FMD, % | 4.8 ± 0.7 | 5.0 ± 1.0 | p = 0.699 | 4.9 ± 0.9 | p = 0.880 |
Primary and secondary endpoints
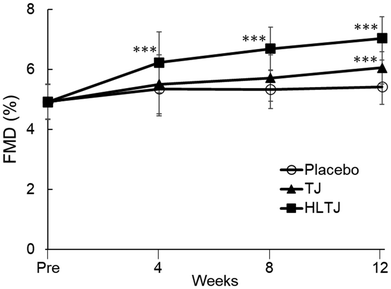
The mean value in FMD is shown in Fig. 3. The LMM revealed significant interaction between time and intervention (p for interaction <0.001). The post hoc Dunnett's test revealed that FMD at 12 weeks in the TJ (6.1 ± 0.5%, p < 0.001) and HLTJ groups (7.0 ± 0.7%, p < 0.001) was significantly higher than that in the placebo group (5.4 ± 0.6%). In the HLTJ group, FMD at 4 weeks (6.2 ± 1.0%, p < 0.001) and 8 weeks (6.7 ± 0.7%, p < 0.001) was significantly higher than that in the placebo group (5.4 ± 0.9%, 5.3 ± 0.6%, respectively). Detailed results of FMD are shown in Table S1. The results of secondary endpoints are shown in Table 3. No significant differences between the groups were observed in blood and urinary oxidative stress markers or urinary nitric oxide levels. A significant interaction between time and intervention was observed for serum lycopene concentrations in the LMM (p for interaction <0.001). The post hoc Dunnett's test revealed that serum lycopene concentrations were significantly higher in the TJ and HLTJ groups than those in the placebo group (p < 0.001 at 4, 8, and 12 weeks).| Outcomes | Group | Pre | 4 weeks | 8 weeks | 12 weeks | P for interaction |
|---|---|---|---|---|---|---|
| Data are expressed as mean ± standard deviation. ***p < 0.001 vs. placebo (adjusted p-value by Dunnett's test and Bonferroni correction). Pre, pre-intervention examination; TJ, tomato juice; HLTJ, high-lycopene tomato juice; FMD, flow-mediated dilation; NOx, nitrogen oxides; 8-OHdG, 8-hydroxy-2′-deoxyguanosine; MDA, malondialdehyde. | ||||||
| Urinary NOx (μmol mg−1 creatinine) | Placebo | 1.1 ± 0.6 | 1.3 ± 0.9 | 1.1 ± 0.5 | 1.2 ± 1.0 | 0.918 |
| TJ | 1.2 ± 0.8 | 1.3 ± 0.6 | 1.5 ± 1.3 | 1.6 ± 2.2 | ||
| HLTJ | 1.2 ± 0.7 | 1.2 ± 0.7 | 1.3 ± 0.9 | 1.5 ± 0.9 | ||
| Urinary 8-OHdG (ng mg−1 creatinine) | Placebo | 12.5 ± 17.6 | 9.7 ± 2.7 | 9.6 ± 3.8 | 10.2 ± 2.9 | 0.706 |
| TJ | 10.3 ± 4.5 | 9.8 ± 3.5 | 9.8 ± 3.4 | 10.5 ± 3.2 | ||
| HLTJ | 9.3 ± 3.1 | 10.1 ± 3.5 | 9.1 ± 2.5 | 10.0 ± 2.9 | ||
| Urinary 8-isoprostane (ng mg−1 creatinine) | Placebo | 4.5 ± 1.7 | 3.9 ± 1.3 | 3.8 ± 1.4 | 3.5 ± 1.0 | 0.998 |
| TJ | 4.3 ± 1.3 | 3.8 ± 1.5 | 3.8 ± 1.2 | 3.6 ± 1.0 | ||
| HLTJ | 4.1 ± 0.9 | 3.7 ± 1.4 | 3.6 ± 1.1 | 3.3 ± 1.3 | ||
| Serum MDA (nM) | Placebo | 608.7 ± 159.0 | 657.0 ± 190.9 | 655.7 ± 181.4 | 598.7 ± 182.8 | 0.483 |
| TJ | 616.0 ± 197.0 | 730.4 ± 232.6 | 702.8 ± 230.1 | 694.8 ± 227.0 | ||
| HLTJ | 617.5 ± 208.7 | 665.0 ± 155.6 | 630.8 ± 122.1 | 601.3 ± 152.5 | ||
| Serum lycopene (nM) | Placebo | 447.0 ± 237.1 | 422.2 ± 203.5 | 376.1 ± 153.5 | 419.2 ± 148.2 | <0.001 |
| TJ | 495.7 ± 273.5 | 759.6 ± 295.4*** | 781.2 ± 305.8*** | 842.0 ± 371.4*** | ||
| HLTJ | 508.9 ± 235.7 | 852.8 ± 348.5*** | 842.1 ± 288.7*** | 943.7 ± 293.8*** | ||
Safety analysis
The hematological, biochemical, and urinary parameters are shown in the SI (Table S2). Two markers (white blood cell count and lactate dehydrogenase) were significantly different in the placebo and TJ groups; however, no significant differences were observed between the placebo and HLTJ groups. There were five adverse events (elevation of blood TG, occult blood reaction, fracture of the right elbow, left knee meniscus posterior horn injury, and fatigue) during the intervention period; however, none of the symptoms were related to the intake of the experimental foods (Table S3). All serum samples returned negative results for virus testing.Discussion
This randomized, placebo-controlled, double-blind, parallel-group comparison study examined the effects of long-term lycopene intake on FMD in healthy adults with borderline FMD values. The results showed that FMD significantly increased after consuming tomato juice containing 15.0 mg of lycopene for 12 weeks or consuming tomato juice containing 26.7 mg of lycopene for ≥4 weeks. These results showed that long-term lycopene intake improved the FMD even at lower doses than those previously reported.11The effect of continuous lycopene intake (for more than 1 week) on FMD is influenced by the amount and duration of lycopene intake.11,12,14 Xaplanteris et al. reported that consuming 70 g of tomato paste (33 mg of lycopene) for 15 days significantly increased FMD compared to that in the control group.11 Furthermore, Samaras et al. reported a significant increase in FMD after drinking tomato juice (the amount of lycopene was not specified) for 2 months compared to that before the intervention.12 In contrast, Stangl et al. reported that consuming 70 g of tomato puree containing 46.2 mg of lycopene for 1 week did not significantly increase FMD.14 In our study, consuming 26.7 mg of lycopene for >4 weeks or 15.0 mg of lycopene for 12 weeks significantly increased the FMD. These results suggest that the effect of lycopene on improving vascular endothelial function is more likely to be exerted by consuming lycopene for >4 weeks. Although lycopene is gradually depleted from the blood over time,17,22,23 blood lycopene concentrations have been reported to increase significantly with continuous ingestion of tomato juice for 3 to 6 weeks.24–26 In this study, blood lycopene concentrations increased significantly after 4 weeks of tomato juice ingestion. Continuous intake of tomato juice for more than 4 weeks likely causes accumulation of lycopene in the body, which may explain the significant increase in FMD observed after 4 weeks of intake. The guidelines issued by the EFSA state that “an increase in fasting FMD caused by the continuous consumption of food ingredients for >4 weeks has a beneficial physiological effect on vascular endothelial function”.16 Increasing FMD by lycopene intake for >4 weeks in our study strongly supports the beneficial effect of lycopene on vascular endothelial function.
The FMD significantly increased in the placebo group after 8 and 12 weeks compared to the values before intake (p < 0.001, preliminary intragroup comparison by Dunnett's test). The Framingham Heart Study, a long-term ongoing cardiovascular cohort study, showed that FMD changes seasonally, increasing from winter to summer but not from summer to autumn.27 Given that the intervention period of this study was from summer to autumn, the increase in FMD in the placebo group was not considered to result from seasonal changes. Although the placebo food contained a very small amount (0.7 mg) of lycopene, the effect of lycopene was considered negligible because there was no significant increase in serum lycopene concentration in the placebo group. Water-soluble tomato components remained in the placebo food even though lycopene was almost completely removed by membrane permeation. Water-soluble tomato extract has anti-platelet aggregation effects28 and anti-inflammatory effects in human umbilical vein endothelial cells.29 The potential active ingredients in the water-soluble tomato components include adenosine, rutin, and chlorogenic acid.28,29 Based on these reports, the placebo food might have contained these water-soluble ingredients, which could have influenced the FMD. Previous reports11,12,14,15 evaluating the effects of tomato products on FMD compared the effects of tomato products on the test group with those not undergoing intervention; no food was set as an appropriate placebo. Our study evaluated the effect of lycopene on FMD more accurately than previous reports using a placebo food containing the water-soluble components of tomatoes.
The placebo juice in this study contained 0.8 g and 0.9 g less dietary fiber than TJ and HLTJ, respectively, because it was produced by removing lycopene from tomato juice through centrifugation and membrane permeation. A human interventional study reported that a meal containing 20.3 g of dietary fiber does not significantly increase postprandial FMD,30 suggesting that the amount of dietary fiber in TJ and HLTJ likely has little effect on FMD. In addition, centrifugation and membrane permeation may have removed other carotenoids (β-carotene, phytoene, and phytofluene). Although these components were not quantified in this study, previous reports indicated that 190 g of tomato juice contained approximately 0.3 mg of β-carotene, 3.8 mg of phytoene, and 1.7 mg of phytofluene.25,26,31 A human interventional study showed that ingestion of palm oil containing 21 mg of carotenes for 8 weeks did not increase FMD.32 Moreover, no human intervention studies have evaluated the effects of phytoene and phytofluene on vascular endothelial function. Although the contributions of β-carotene, phytoene, and phytofluene to the increase in FMD by tomato juice intake in this study cannot be completely ruled out, the effects are considered to be minimal.
NO is a substance secreted from the vascular endothelium that has beneficial effects on the vascular endothelium33 and plays an important role in improving FMD.3 An in vitro study showed that lycopene suppressed endothelial cell migration by suppressing vascular endothelial growth factor expression and increasing NO production.10 Furthermore, an animal study using a hyperhomocysteinemia rat model showed that administration of 20 mg kg−1 pure lycopene dissolved in corn oil for 12 weeks increased serum NO levels, ameliorated endothelial dysfunction, and prevented early arteriosclerosis induced by hyperhomocysteinemia.34 A human interventional study evaluating the effects of tomato paste on postprandial FMD showed a significant increase in urinary NO levels by tomato paste intake, but no significant change in FMD was observed.15 Xaplanteris et al. proposed that antioxidants such as lycopene in tomato paste may change the redox status of vascular endothelium and increase the production of vasodilators such as NO; however, NO was not measured in the study.11 Our study found no significant difference in the urinary NO level between the groups, even though the mean NO concentration at 12 weeks was higher in the TJ and HLTJ groups than in the placebo group. Measuring the serum NO concentration may be necessary to examine the relationship between lycopene-induced FMD improvement and NO production because a previous animal study showed an increase in serum NO concentration and amelioration of endothelial dysfunction.34
The increased production of reactive oxygen species is thought to reduce NO bioavailability and worsen vascular endothelial function.35 A human intervention study showed that the intake of tomato paste containing lycopene increased the FMD and improved the oxidative status measured by the total amount of plasma lipid peroxides.11 Another human intervention study showed that daily intake of 15 mg of lycopene supplement increased the activity of superoxide dismutase, an antioxidant enzyme, and improved reactive hyperemia peripheral arterial tonometry, an indicator of vascular endothelial function. Additionally, a correlation was observed between the changes in these two measures.36 Urinary 8-isoprostane and serum MDA levels, which are indicators of lipid oxidation, and the urinary 8-OHdG level, an indicator of DNA oxidation, were measured as indicators of oxidation status in our study, but no significant changes were observed by lycopene intake. Therefore, the antioxidant activity of lycopene might have contributed little to the increase in FMD observed in this study. In addition to its direct effects on oxidative stress, lycopene exerts antioxidant activity by modulating transcription factors such as NF-kB and Nrf2.37 The relationship between lycopene's effects on these transcription factors and its impact on vascular endothelial function remains to be investigated and is a topic for future study.
Oxidized LDL is thought to cause impairment of vascular endothelial function by decreasing the activity of NO synthase and increasing vascular endothelial adhesion factors.38 The plasma oxidized LDL concentration is positively correlated with the plasma LDL-C concentration.39 Additionally, blood LDL-C concentration is negatively correlated with FMD.40 A human interventional study showed that a 3-week intake of a high-tomato diet (400 mL TJ and 30 mg tomato ketchup daily) significantly reduced LDL-C levels.41 Another human intervention study found that intake of 50 g semi-dried tomato (providing 22.0–27.8 mg lycopene per day) for 12 weeks significantly reduced LDL-C levels.42 In addition, a meta-analysis of six human interventional studies showed that tomato supplementation was associated with significant reductions in LDL-C.13 Reduction in blood LDL-C concentration might be involved in the effect of lycopene on improving vascular endothelial function based on these reports. The serum LDL-C concentration was measured as a blood biochemical marker for safety analysis in our study. However, the relationship between changes in FMD and blood LDL-C concentration was unclear.
A safety analysis was conducted based on hematology, blood biochemistry, and urinary examination results and adverse event data collected from the daily diaries; however, no information concerning the safety of lycopene was obtained. Lycopene is thought to be an extremely safe component because synthetic lycopene and lycopene extract from tomatoes have received GRAS certification,43,44 and there have been few reports of serious adverse events caused by lycopene intake.
This study had some limitations. First, markers of oxidative stress and NO were measured to elucidate the mechanism of action of lycopene; however, the mechanism was not clear in our study alone. It would be necessary to measure other markers not assessed in our study, such as blood NOx, LDL-C, and oxidized LDL concentrations, to elucidate the underlying mechanism. Second, the effects of components other than lycopene could not be completely excluded, although our study evaluated the effect of lycopene more accurately than previous reports by using a placebo food containing water-soluble components of tomatoes. This is a common limitation of studies evaluating the physiological functions of food ingredients; however, an interventional study using lycopene supplements containing high concentrations of lycopene is important.
Conclusions
This randomized, placebo-controlled, double-blind, parallel-group comparison study showed that lycopene intake from tomato juice increased FMD, a representative indicator of vascular endothelial function. These findings support the beneficial effects of lycopene on vascular endothelial function. In the future, it will be important to evaluate markers such as blood NOx, LDL-C, and oxidized LDL concentrations to elucidate the underlying mechanisms.Author contributions
K. Y.: conceptualization, data curation, formal analysis, methodology, validation, and writing – original draft; Y. N.: conceptualization, formal analysis, methodology, investigation, validation, and writing – review and editing; S. T.: conceptualization, methodology, project administration, and writing – review and editing; S. S.: supervision and writing – review and editing.Conflicts of interest
KAGOME Co., Ltd supported this work and provided support in the form of salaries for authors K. Y., Y. N., S. T., and S.S.Data availability
The data supporting this article have been included as part of the SI. Supplementary information is available. Detailed results of FMD are shown in Table S1. The results of hematological, biochemical, and urinary parameters are shown in Table S2. Detailed information of the adverse events during the intervention period is shown in Table S3. See DOI: https://doi.org/10.1039/d5fo01397f.Other datasets are not readily available because the de-identified data described in the article, code book, and analytic codes will be made available upon request for the approval of an application for data use and execution of a Data Use Agreement and/or Material Transfer Agreement with KAGOME Co., Ltd. Requests to access the datasets were made by the corresponding author.
Acknowledgements
We thank all the participants in this study. We also thank Dr Nobuhiko Shioya, Dr Takatoshi Murata, and Dr Masahiko Tokushima for being the randomization manager, allocation manager, and principal investigator, respectively.References
- L. Nedkoff, T. Briffa, D. Zemedikun, S. Herrington and F. L. Wright, Global trends in atherosclerotic cardiovascular disease, Clin. Ther., 2023, 45, 1087–1091 CrossRef PubMed.
- T. Okamura, K. Tsukamoto, H. Arai, Y. Fujioka, Y. Ishigaki, S. Koba, H. Ohmura, T. Shoji, K. Yokote, H. Yoshida, M. Yoshida, J. Deguchi, K. Dobashi, A. Fujiyoshi, H. Hamaguchi, M. Hara, M. Harada-Shiba, T. Hirata, M. Iida, Y. Ikeda, S. Ishibashi, H. Kanda, S. Kihara, K. Kitagawa, S. Kodama, M. Koseki, Y. Maezawa, D. Masuda, T. Miida, Y. Miyamoto, R. Nishimura, K. Node, M. Noguchi, M. Ohishi, I. Saito, S. Sawada, H. Sone, M. Takemoto, A. Wakatsuki and H. Yanai, Japan Atherosclerosis Society (JAS) guidelines for prevention of atherosclerotic cardiovascular diseases 2022, J. Atheroscler. Thromb., 2024, 31, 641–853 CrossRef PubMed.
- M. C. Corretti, T. J. Anderson, E. J. Benjamin, D. Celermajer, F. Charbonneau, M. A. Creager, J. Deanfield, H. Drexler, M. Gerhard-Herman, D. Herrington, P. Vallance, J. Vita, R. Vogel and International Brachial Artery Reactivity Task Force, Guidelines for the ultrasound assessment of endothelial-dependent flow-mediated vasodilation of the brachial artery: a report of the International Brachial Artery Reactivity Task Force, J. Am. Coll. Cardiol., 2002, 39, 257–265 CrossRef.
- D. S. Celermajer, K. E. Sorensen, C. Bull, J. Robinson and J. E. Deanfield, Endothelium-dependent dilation in the systemic arteries of asymptomatic subjects relates to coronary risk factors and their interaction, J. Am. Coll. Cardiol., 1994, 24, 1468–1474 CrossRef PubMed.
- A. Tanaka, H. Tomiyama, T. Maruhashi, Y. Matsuzawa, T. Miyoshi, T. Kabutoya, K. Kario, S. Sugiyama, M. Munakata, H. Ito, S. Ueda, C. Vlachopoulos, Y. Higashi, T. Inoue and K. Node, Physiological Diagnosis Criteria for Vascular Failure Committee, Physiological diagnostic criteria for vascular failure, Hypertension, 2018, 72, 1060–1071 CrossRef.
- P. Di Mascio, S. Kaiser and H. Sies, Lycopene as the most efficient biological carotenoid singlet oxygen quencher, Arch. Biochem. Biophys., 1989, 274, 532–538 CrossRef PubMed.
- H. D. Sesso, S. Liu, J. M. Gaziano and J. E. Buring, Dietary lycopene, tomato-based food products and cardiovascular disease in women, J. Nutr., 2003, 133, 2336–2341 CrossRef.
- P. F. Jacques, A. Lyass, J. M. Massaro, R. S. Vasan and R. B. D'Agostino Sr, Relationship of lycopene intake and consumption of tomato products to incident CVD, Br. J. Nutr., 2013, 110, 545–551 CrossRef.
- C. F. Hung, T. F. Huang, B. H. Chen, J. M. Shieh, P. H. Wu and W. B. Wu, Lycopene inhibits TNF-alpha-induced endothelial ICAM-1 expression and monocyte-endothelial adhesion, Eur. J. Pharmacol., 2008, 586, 275–282 CrossRef.
- D. Treggiari, A. Dalbeni, A. Meneguzzi, P. Delva, C. Fava, B. Molesini, T. Pandolfini and P. Minuz, Lycopene inhibits endothelial cells migration induced by vascular endothelial growth factor A increasing nitric oxide bioavailability, J. Funct. Foods, 2018, 42, 312–318 CrossRef.
- P. Xaplanteris, C. Vlachopoulos, P. Pietri, D. Terentes-Printzios, D. Kardara, N. Alexopoulos, K. Aznaouridis, A. Miliou and C. Stefanadis, Tomato paste supplementation improves endothelial dynamics and reduces plasma total oxidative status in healthy subjects, Nutr. Res., 2012, 32, 390–394 CrossRef.
- A. Samaras, K. Tsarouhas, E. Paschalidis, G. Giamouzis, F. Triposkiadis, C. Tsitsimpikou, A. T. Becker, N. Goutzourelas and D. Kouretas, Effect of a special carbohydrate-protein bar and tomato juice supplementation on oxidative stress markers and vascular endothelial dynamics in ultra-marathon runners, Food Chem. Toxicol., 2014, 69, 231–236 CrossRef PubMed.
- H. M. Cheng, G. Koutsidis, J. K. Lodge, A. Ashor, M. Siervo and J. Lara, Tomato and lycopene supplementation and cardiovascular risk factors: A systematic review and meta-analysis, Atherosclerosis, 2017, 257, 100–108 CrossRef PubMed.
- V. Stangl, C. Kuhn, S. Hentschel, N. Jochmann, C. Jacob, V. Böhm, K. Fröhlich, L. Müller, C. Gericke and M. Lorenz, Lack of effects of tomato products on endothelial function in human subjects: results of a randomised, placebo-controlled cross-over study, Br. J. Nutr., 2011, 105, 263–267 CrossRef.
- A. Dalbeni, D. Treggiari, A. Tagetti, M. Bevilaqua, S. Bonafini, M. Montagnana, G. Scaturro, P. Minuz and C. Fava, Positive effects of tomato paste on vascular function after a fat meal in male healthy subjects, Nutrients, 2018, 10, 1310 CrossRef PubMed.
- EFSA Panel on Dietetic Products, Nutrition and Allergies (EFSA NDA Panel);, D. Turck, J. L. Bresson, B. Burlingame, T. Dean, S. Fairweather-Tait, M. Heinonen, K. I. Hirsch-Ernst, I. Mangelsdorf, H. J. McArdle, A. Naska, M. Neuhäuser-Berthold, G. Nowicka, K. Pentieva, Y. Sanz, A. Sjödin, M. Stern, D. Tomé, H. Van Loveren, M. Vinceti, P. Willatts, A. Martin, J. J. Strain, L. Heng, S. Valtueña Martínez and A. Siani, Guidance for the scientific requirements for health claims related to antioxidants, oxidative damage and cardiovascular health: (Revision 1), EFSA J., 2018, 16, e05136 Search PubMed.
- S. Oshima, H. Sakamoto, Y. Ishiguro and J. Terao, Accumulation and clearance of capsanthin in blood plasma after the ingestion of paprika juice in men, J. Nutr., 1997, 127, 1475–1479 CrossRef PubMed.
- K. Aizawa and T. Inakuma, Quantitation of carotenoids in commonly consumed vegetables in Japan, Food Sci. Technol. Res., 2007, 13, 247–252 CrossRef.
- T. Tsuchihashi, K. Masuda, H. Oniki, M. Sakaki, K. Arakawa, W. Kameda and H. Fukui, Assessment of salt intake by using a simple check sheet in hypertensive patients, J. Blood Pressure, 2013, 20, 1239–1243 Search PubMed.
- K. Yasutake, E. Miyoshi, T. Kajiyama, Y. Umeki, Y. Misumi, N. Horita, Y. Murata, K. Ohe, M. Enjoji and T. Tsuchihashi, Comparison of a salt check sheet with 24 h urinary salt excretion measurement in local residents, Hypertens. Res., 2016, 39, 879–885 CrossRef.
- Y. Kanda, Investigation of the freely available easy-to-use software “EZR” for medical statistics, Bone Marrow Transplant., 2013, 48, 452–458 CrossRef.
- C. L. Rock, M. E. Swendseid, R. A. Jacob and R. W. McKee, Plasma carotenoid levels in human subjects fed a low carotenoid diet, J. Nutr., 1992, 122, 96–100 Search PubMed.
- B. J. Burri, T. R. Neidlinger and A. J. Clifford, Serum carotenoid depletion follows first-order kinetics in healthy adult women fed naturally low carotenoid diets, J. Nutr., 2001, 131, 2096–2100 CrossRef PubMed.
- V. Böhm and R. Bitsch, Intestinal absorption of lycopene from different matrices and interactions to other carotenoids, the lipid status, and the antioxidant capacity of human plasma, Eur. J. Nutr., 1999, 38, 118–125 CrossRef PubMed.
- A. Nakamura, C. Itaki, A. Saito, T. Yonezawa, K. Aizawa, A. Hirai, H. Suganuma, T. Miura, Y. Mariya and S. Haghdoost, Possible benefits of tomato juice consumption: a pilot study on irradiated human lymphocytes from healthy donors, Nutr. J., 2017, 16, 27 Search PubMed.
- Y. Fukushi, Y. Mariya, K. Yamada, K. Yoshida, A. Sasa, H. Saito, A. Hirai, S. Suzuki, K. Aizawa, H. Suganuma and C. Itaki, Tomato juice consumption could improve breast skin adverse effects of radiotherapy in breast cancer patients, In Vivo, 2020, 34, 3013–3021 Search PubMed.
- M. E. Widlansky, J. A. Vita, M. J. Keyes, M. G. Larson, N. M. Hamburg, D. Levy, G. F. Mitchell, E. W. Osypiuk, R. S. Vasan and E. J. Benjamin, Relation of season and temperature to endothelium-dependent flow-mediated vasodilation in subjects without clinical evidence of cardiovascular disease (from the Framingham Heart Study), Am. J. Cardiol., 2007, 100, 518–523 CrossRef PubMed.
- N. O'Kennedy, D. Raederstorff and A. K. Duttaroy, Fruitflow®: the first European Food Safety Authority-approved natural cardio-protective functional ingredient, Eur. J. Nutr., 2017, 56, 461–482 CrossRef PubMed.
- J. Schwager, N. Richard, B. Mussler and D. Raederstorff, Tomato aqueous extract modulates the inflammatory profile of immune cells and endothelial cells, Molecules, 2016, 21, 168 CrossRef PubMed.
- G. A. Gaesser, J. Rodriguez, J. T. Patrie, C. M. Whisner and S. S. Angadi, Effects of glycemic index and cereal fiber on postprandial endothelial function, glycemia, and insulinemia in healthy adults, Nutrients, 2019, 11, 2387 Search PubMed.
- B. Olmedilla-Alonso, A. M. Benítez-González, R. Estévez-Santiago, P. Mapelli-Brahm, C. M. Stinco and A. J. Meléndez-Martínez, Assessment of food sources and the intake of the colourless carotenoids phytoene and phytofluene in Spain, Nutrients, 2021, 13, 4436 CrossRef CAS.
- W. Stonehouse, G. D. Brinkworth, C. H. Thompson and M. Y. Abeywardena, Short term effects of palm-tocotrienol and palm-carotenes on vascular function and cardiovascular disease risk: A randomised controlled trial, Atherosclerosis, 2016, 254, 205–214 CrossRef CAS.
- D. G. Harrison and H. Cai, Endothelial control of vasomotion and nitric oxide production, Cardiol. Clin., 2003, 21, 289–302 Search PubMed.
- X. Liu, D. Qu, F. He, Q. Lu, J. Wang and D. Cai, Effect of lycopene on the vascular endothelial function and expression of inflammatory agents in hyperhomocysteinemic rats, Asia Pac. J. Clin. Nutr., 2007, 16(Supplement 1), 244–248 Search PubMed.
- D. Tousoulis, A. M. Kampoli, C. Tentolouris, N. Papageorgiou and C. Stefanadis, The role of nitric oxide on endothelial function, Curr. Vasc. Pharmacol., 2012, 10, 4–18 CrossRef CAS PubMed.
- J. Y. Kim, J. K. Paik, O. Y. Kim, H. W. Park, J. H. Lee, Y. Jang and J. H. Lee, Effects of lycopene supplementation on oxidative stress and markers of endothelial function in healthy men, Atherosclerosis, 2011, 215, 189–195 CrossRef CAS PubMed.
- T. Bohn, M. L. Bonet, P. Borel, J. Keijer, J. F. Landrier, I. Milisav, J. Ribot, P. Riso, B. Winklhofer-Roob, Y. Sharoni, J. Corte-Real, Y. van Helden, M. R. Loizzo, B. Poljšak, M. Porrini, J. Roob, P. Trebše, R. Tundis, A. Wawrzyniak, R. Rühl and J. Dulińska-Litewka, Mechanistic aspects of carotenoid health benefits – where are we now?, Nutr. Res. Rev., 2021, 34, 276–302 CrossRef CAS.
- M. A. Incalza, R. D'Oria, A. Natalicchio, S. Perrini, L. Laviola and F. Giorgino, Oxidative stress and reactive oxygen species in endothelial dysfunction associated with cardiovascular and metabolic diseases, Vasc. Pharmacol., 2018, 100, 1–19 Search PubMed.
- L. P. van der Zwan, T. Teerlink, J. M. Dekker, R. M. A. Henry, C. D. A. Stehouwer, C. Jakobs, R. J. Heine and P. G. Scheffer, Circulating oxidized LDL: determinants and association with brachial flow-mediated dilation, J. Lipid Res., 2009, 50, 342–349 CrossRef CAS.
- Y. Takaeko, M. Kajikawa, S. Kishimoto, T. Yamaji, T. Harada, Y. Han, Y. Kihara, E. Hida, K. Chayama, C. Goto, Y. Aibara, F. M. Yusoff, T. Maruhashi, A. Nakashima and Y. Higashi, Low levels of low-density lipoprotein cholesterol and endothelial function in subjects without lipid-lowering therapy, J. Clin. Med., 2020, 9, 3796 CrossRef CAS.
- M. L. Silaste, G. Alfthan, A. Aro, Y. A. Kesäniemi and S. Hörkkö, Tomato juice decreases LDL cholesterol levels and increases LDL resistance to oxidation, Br. J. Nutr., 2007, 98, 1251–1258 Search PubMed.
- M. Nishimura, N. Tominaga, Y. Ishikawa-Takano, M. Maeda-Yamamoto and J. Nishihira, Effect of 12-week daily intake of the high-lycopene tomato (Solanum Lycopersicum), a variety named “PR-7”, on lipid metabolism: a randomized, double-blind, placebo-controlled, parallel-group study, Nutrients, 2019, 11, 1177 Search PubMed.
- G. R. N. Gras Notices, No. 119, Synthetic lycopene, https://www.hfpappexternal.fda.gov/scripts/fdcc/index.cfm?set=GRASNotices&id=119, (accessed 29 January, 2025).
- G. R. N. Gras Notices, No. 156, Tomato lycopene extract 6 percent, tomato lycopene extract 1.5 percent, and crystallized tomato lycopene extract, https://www.hfpappexternal.fda.gov/scripts/fdcc/index.cfm?set=GrASNotices&id=156, (accessed 29 January, 2025).
| This journal is © The Royal Society of Chemistry 2025 |