Recent advances in the effect of simulated gastrointestinal digestion and encapsulation on peptide bioactivity and stability
Chenlong
Chen†
 a,
Wancong
Yu†
b,
Xiaohong
Kou†
a,
Yujia
Niu
a,
Jiaxin
Ji
a,
Ying
Shao
a,
Shuqi
Wu
a,
Mengyi
Liu
a and
Zhaohui
Xue
a,
Wancong
Yu†
b,
Xiaohong
Kou†
a,
Yujia
Niu
a,
Jiaxin
Ji
a,
Ying
Shao
a,
Shuqi
Wu
a,
Mengyi
Liu
a and
Zhaohui
Xue
 *a
*a
aSchool of Chemical Engineering and Technology, Tianjin University, Tianjin 300350, China. E-mail: zhhxue@tju.edu.cn
bBiotechnology Research Institute, Tianjin Academy of Agricultural Sciences, Tianjin 300384, China
First published on 13th February 2025
Abstract
Food-derived bioactive peptides have garnered significant attention from researchers due to their specific biological functions, including antihypertensive, antioxidant, antidiabetic, anticancer, anti-inflammatory, and anti-osteoporosis properties. Despite extensive in vitro research, the bioactivity of these peptides may be compromised in the gastrointestinal tract due to enzymatic hydrolysis before reaching the bloodstream or target cells. Therefore, understanding the fate of bioactive peptides during digestion is crucial before advancing to clinical trials and commercial applications. To exert their health-promoting effects, these peptides must maintain their bioactivity throughout digestion. Encapsulation has emerged as a promising strategy for protecting peptides in the gastrointestinal tract. This review examines the effects of in vitro simulated gastrointestinal digestion on peptide bioactivity and stability, highlighting recent research on encapsulation strategies designed to enhance their gastrointestinal stability. Furthermore, the review addresses existing research gaps and suggests future research directions to advance our understanding and the application of bioactive peptides.
1. Introduction
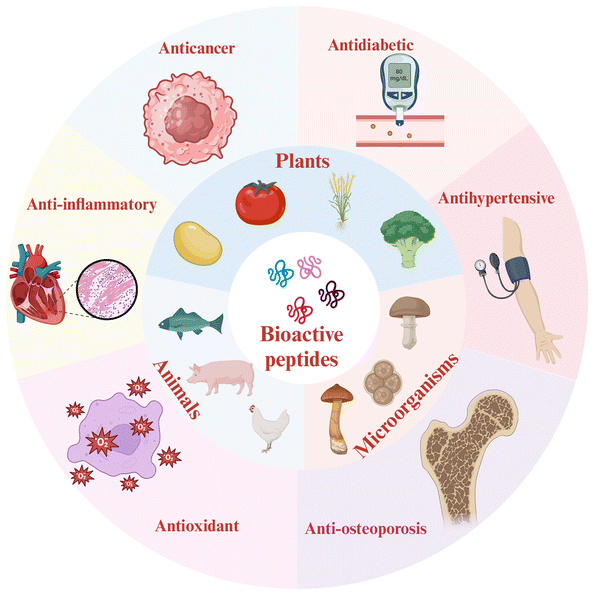
Food-derived bioactive peptides are fragments of 2–20 amino acid residues.1 Beyond basic nutrition, these peptides exhibit various bioactivities, including antihypertensive,2 antioxidant,3 anticancer,4 antidiabetic,5 anti-inflammatory,6 and anti-osteoporosis7 properties (Fig. 1). They are released from parent proteins through protease hydrolysis, fermentation, or germination.8 Parent proteins can be obtained from animals, plants, microorganisms, and their derivatives9 (Fig. 1), providing abundant sources of peptides. Unlike synthetic drugs, which may lead to severe side effects,10 bioactive peptides offer promising health benefits as natural alternatives, due to their low molecular weight, low toxicity, selectivity, and high bioactivity.11,12Current research on bioactive peptides predominantly focuses on in vitro studies, where the efficacy observed does not always correlate with their in vivo functionality. This discrepancy is primarily due to the degradation of bioactive peptides by digestive enzymes in the gastrointestinal tract, resulting in a loss of bioactivity. Once ingested, bioactive peptides will encounter numerous digestive proteases and peptidases. These include pepsin, trypsin, chymotrypsin, elastase, carboxypeptidases A/B, and brush border peptidases such as several aminopeptidases and carboxypeptidase M.13 Pepsin preferentially hydrolyzes peptide bonds at the carboxyl-terminal Phe, Leu, Tyr, and Trp residues.14 Trypsin, an endopeptidase, tends to cleave peptides at the carboxyl-terminal Arg and Lys residues.13 Chymotrypsin, a serine protease, cleaves aromatic amino acid residues like Trp, Tyr, and Phe, except when adjacent to Pro.15 Elastase has an affinity for uncharged residues such as Ala, Gly, and Ser.16 Carboxypeptidases are metalloproteinases with a zinc atom in their active site. Pancreatic carboxypeptidases include types A and B. Carboxypeptidase A hydrolyzes peptide bonds involving aromatic amino acids at the carboxyl-terminal end, whereas carboxypeptidase B cleaves Lys or Arg residues from the same terminus.13 Fortunately, the stability of bioactive peptides can be improved through encapsulation. This method converts sensitive materials into stable components, effectively shielding them from gastrointestinal digestion.17
Human and animal in vivo digestion models generally involve in vivo imaging18 and wireless telemetry techniques.19 Although these models provide accurate physiological data on the digestion of bioactive peptides, they involve long research periods, high costs, and ethical concerns related to surgical approaches and animal mortality.20 Consequently, in vivo digestion models present practical research challenges. To overcome these limitations, several in vitro static and dynamic digestion models have been developed and optimized to assess the effect of gastrointestinal digestion on peptide bioactivity and stability in recent years. Among these, static digestion models are the most widely used due to their ease of operation, lack of specific equipment requirements, and low cost.20 INFOGEST is a standardized and practical static digestion method.21 In this method, during the oral phase, solid foods are minced to simulate mastication. The oral digestion phase for liquid foods is optional. Subsequently, the mixture is prepared by combining simulated salivary fluid, (SSF) with solid or liquid food at a 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50 (v/w and v/v) ratio. The mixture contains salivary alpha-amylase and CaCl2 at concentrations of 75 U mL−1 and 0.75 mM, respectively, with water added as necessary to dilute the stock solution of SSF. The mixture is incubated at 37 °C for 2 min. During the gastric phase, under standard conditions, the final ratio of simulated gastric fluid (SGF) (containing 0.075 mM CaCl2 and pepsin with an activity of 2000 U mL−1 in the final digestion mixture) to liquid food or oral bolus is 50
50 (v/w and v/v) ratio. The mixture contains salivary alpha-amylase and CaCl2 at concentrations of 75 U mL−1 and 0.75 mM, respectively, with water added as necessary to dilute the stock solution of SSF. The mixture is incubated at 37 °C for 2 min. During the gastric phase, under standard conditions, the final ratio of simulated gastric fluid (SGF) (containing 0.075 mM CaCl2 and pepsin with an activity of 2000 U mL−1 in the final digestion mixture) to liquid food or oral bolus is 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50 (v/v), at a digestion time of 2 h, a temperature of 37 °C, and a pH of 3. During the intestinal phase, the pH is adjusted to 7, the temperature remains at 37 °C, and the digestion time is also 2 h. The final ratio of simulated intestinal fluid (SIF) to gastric chyme is also 50
50 (v/v), at a digestion time of 2 h, a temperature of 37 °C, and a pH of 3. During the intestinal phase, the pH is adjusted to 7, the temperature remains at 37 °C, and the digestion time is also 2 h. The final ratio of simulated intestinal fluid (SIF) to gastric chyme is also 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50 (v/v). The enzyme in SIF can be a porcine pancreatic enzyme or individual enzymes, with the final digestion mixture containing bile salts (10 mM), CaCl2 (0.3 mM), chymotrypsin (25 U mL−1), and trypsin (100 U mL−1). On this basis, an enhanced version, INFOGEST 2.0, was developed and detailed in a 2019 article published in Nature Protocols. Unlike INFOGEST, INFOGEST 2.0 requires the inclusion of the oral digestion phase to ensure consistent dilution of food, regardless of whether the food is solid or liquid. Additionally, INFOGEST 2.0 introduces gastric lipase, significantly impacting fat digestion.22 Although static digestion has many advantages, it still presents limitations such as maintaining constant pH and enzyme activity, a lack of gastric emptying, and non-gradual addition of digestive fluids. Therefore, static digestion is only suitable for assessing digestion endpoints rather than the digestion process itself. Hence, the recently developed semi-dynamic digestion protocol has enhanced the secretion of digestive fluids, pH control, and gastric emptying compared to the static method. In this protocol, SGF is gradually added. The pH is adjusted by adding HCl, reaching pH 2 by the end of gastric digestion. Gastric emptying occurs at a linear rate, and this protocol recommends delivering calories to the intestinal phase at a rate of 2 kcal min−1.23 However, neither static nor semi-dynamic digestion methods can accurately simulate gastrointestinal peristalsis and the internal structure of the gastrointestinal tract. Therefore, in recent years, researchers have developed numerous dynamic digestion models that not only simulate the continuous secretion of digestive fluids, dynamically adjust pH, and facilitate gastric emptying, but also replicate the real environment and peristalsis of the gastrointestinal tract.24 Nevertheless, due to cost and operational limitations, dynamic digestion models are not as widely applied as static digestion models.
50 (v/v). The enzyme in SIF can be a porcine pancreatic enzyme or individual enzymes, with the final digestion mixture containing bile salts (10 mM), CaCl2 (0.3 mM), chymotrypsin (25 U mL−1), and trypsin (100 U mL−1). On this basis, an enhanced version, INFOGEST 2.0, was developed and detailed in a 2019 article published in Nature Protocols. Unlike INFOGEST, INFOGEST 2.0 requires the inclusion of the oral digestion phase to ensure consistent dilution of food, regardless of whether the food is solid or liquid. Additionally, INFOGEST 2.0 introduces gastric lipase, significantly impacting fat digestion.22 Although static digestion has many advantages, it still presents limitations such as maintaining constant pH and enzyme activity, a lack of gastric emptying, and non-gradual addition of digestive fluids. Therefore, static digestion is only suitable for assessing digestion endpoints rather than the digestion process itself. Hence, the recently developed semi-dynamic digestion protocol has enhanced the secretion of digestive fluids, pH control, and gastric emptying compared to the static method. In this protocol, SGF is gradually added. The pH is adjusted by adding HCl, reaching pH 2 by the end of gastric digestion. Gastric emptying occurs at a linear rate, and this protocol recommends delivering calories to the intestinal phase at a rate of 2 kcal min−1.23 However, neither static nor semi-dynamic digestion methods can accurately simulate gastrointestinal peristalsis and the internal structure of the gastrointestinal tract. Therefore, in recent years, researchers have developed numerous dynamic digestion models that not only simulate the continuous secretion of digestive fluids, dynamically adjust pH, and facilitate gastric emptying, but also replicate the real environment and peristalsis of the gastrointestinal tract.24 Nevertheless, due to cost and operational limitations, dynamic digestion models are not as widely applied as static digestion models.
Understanding the fate of bioactive peptides during digestion is crucial before proceeding to clinical trials and commercial applications. In brief, these peptides must maintain their bioactivity, determining whether they can be broadly used in functional foods or nutraceuticals. This review summarizes the effect of in vitro simulated gastrointestinal digestion on the bioactivity and stability of various bioactive peptides and explores recent research on encapsulation methods to enhance their gastrointestinal stability.
2. The effect of gastrointestinal digestion on peptide bioactivity and stability
Bioactive peptides, originating from various sources and having different functions and molecular sizes, may experience different bioactivity changes during gastrointestinal digestion. Table 1 shows the effect of in vitro simulated gastrointestinal digestion on peptide bioactivity and stability, emphasizing its role as the initial barrier to the in vivo effectiveness of biopeptides. The following review is organized according to the functional classification of bioactive peptides.| Bioactivity | Protein source | Enzyme used for hydrolysis | Hydrolysate name/peptide sequences | Results | Ref. |
|---|---|---|---|---|---|
| Angiotensin-converting enzyme (ACE) inhibitory | Tilapia (Oreochromis niloticus) | Properase E | LSGYGP | Maintain good digestive stability for 4 h | 25 |
| Bitter almond albumin | Papain and thermolysin | RPPSEDEDQE | ACE-inhibitory capacity was not significantly reduced | 26 | |
| Walnut glutelin-1 hydrolysates | Pepsin | VERGRRITSV and FVIEPNITPA | ACE inhibitory rate was reduced to 76.9% and 70.3%, respectively | 27 | |
| Ginkgo seed globulin | Alcalase and thermolysin | EASPKPV | The IC50 (half maximal inhibitory concentration) of EASPKPV increased from 87.66 ± 2.96 mmol L−1 to 93.56 ± 7.38 mmol L−1 | 28 | |
| Oil palm kernel globulin | Papain | EVPQAYIP | 71.23% of its activity was retained | 29 | |
| Oil palm kernel glutelin-2 | Alcalase, flavourzyme, pepsin, and trypsin | VIEPR, LPILR, ADVFNPR and VVLYK | The activity of these peptides did not significantly decrease | 30 | |
| Naked oat bran albumin hydrolysates | Papain and flavourzyme | QYVPF and GYHGH | The IC50 values of digested QYVPF and GYHGH were 260.39 and 368.87 μmol L−1, respectively, showing no significant difference from the untreated peptide values of 243.36 and 321.94 μmol L−1 | 31 | |
| Styela clava | Commercial proteases (flavourzyme, protamex, and kojizyme) and digestive proteases (pepsin and papain) | LWHTH | After digestion by pepsin, trypsin, and α-chymotrypsin, LWHTH maintained its ACE inhibitory effects with IC50 values of 22.31, 19.62, and 21.25 μM, respectively, compared to 16.42 ± 0.45 μM for undigested LWHTH, demonstrating high stability | 32 | |
| Spent hen protein | Protex 26L, pepsin, and thermoase | Spent hen muscle protein hydrolysate (SPH) prepared by protex 26L, pepsin, and thermoase (SPH-26L, SPH-P, and SPH-T) | The activity of SPH-T remained unchanged, whereas the activities of SPH-P and SPH-26L were reduced | 33 | |
| Foxtail millet protein (FMPH) | Alkaline | FPGVSPF, SPAQLLPF, LVPYRP, and WYWPQ | WYWPQ and LVPYRP exhibited superior gastrointestinal stability, maintaining 85.22 ± 2.06% and 83.22 ± 0.39% of their activity, respectively | 34 | |
| Larimichthys crocea protein (LCP) | Trypsin and papain | IPYADFK, FYEPFM, NWPWMK, LYDHLGK, INEMLDTK, IHFGTTGK | The activities of NWPWMK, FYEPFM, and IPYADFK remained nearly unchanged, whereas LYDHLGK, INEMLDTK, and IHFGTTGK showed a decrease in activity | 35 | |
| Antioxidant | Millet bran glutelin-2 | Papain | CFMTY, CTGTPYC, and RGLLLPSMSNAP | CFMTY (IC50 value of 2,2′-azinobis (3-ethyl-benzothiazoline-6-sulfonic acid) (ABTS+) scavenging activity increased from 80.85 to 81.07 μmol L−1) and CTGTPYC (IC50 rose from 87.78 to 92.73 μmol L−1) remained stable, whereas RGLLLPSMSNAP (IC50 increased from 214.17 to 1236.01 μmol L−1) was unstable | 36 |
| Heads and bones of hybrid groupers | Alcalase | A fraction with a molecular weight of less than 1 kDa obtained from bone hydrolysate after ultrafiltration (HB-IV) | The hydroxyl radical (HO˙) scavenging and reducing power were enhanced | 37 | |
| Carp (Cyprinus carpio) skin gelatin | Protamex® (novozymes) enzyme preparation | Unfractionated hydrolysate and AY | Retain the overall antioxidant effect | 38 | |
| Round scad | Protamex and pancreatin | WCPFSRSF | The content of WCPFSRSF dropped to less than 1% following gastrointestinal digestion, significantly impacting its antioxidant activity | 39 | |
| Coconut cake albumin | Trypsin, favourzyme, pepsin, and Alcalase | KAQYPYV, KIIIYN and KILIYG | The HO˙ scavenging activities of KIIIYN and KILIYG decreased remarkably, while KAQYPYV could retain its antioxidant activity | 40 | |
| Spirulina sp. LEB 18 | Protemax 580L | Protein hydrolysates obtained at the 90 and 120 min reaction time points (H90 and H120) | The activity of H90 remained unaffected, while the activity of H120 decreased | 41 | |
| Soybeans | Trypsin, papain, protamex, Alcalase 2.4L, and neutrase 0.8L | SFQSeM | SFQSeM remained mostly intact, with only 11.92% ± 2.76% hydrolysis observed | 42 | |
| Meat and visceral mass of marine snail Neptunea arthritica cumingii | — | YSQLENEFDR and YIAEDAER | The 2,2′-diphenyl-1-picrylhydrazyl (DPPH˙) radical scavenging and ACE inhibitory activities of both peptides slightly increased | 43 | |
| Tomato seed proteins | Alcalase | The fraction of tomato seed protein hydrolysate (TSPH) (F2) | Peptides in fraction F2 had high gastrointestinal stability | 44 | |
| Wheat protein | Alkaline protease | Selenium-chelating polypeptides derived from wheat protein (WPH-Se) | WPH-Se showed resistance to gastrointestinal digestion and preserved its antioxidant effects | 45 | |
| Antidiabetic | Chlorella pyrenoidosa protein | Trypsin and pepsin | FLKPLGSGK, QIYTMGK, FLFVAEAIYK and QHAGTKAK | QIYTMGK and QHAGTKAK resisted digestive enzymes, whereas FLKPLGSGK and FLFVAEAIYK were unstable | 46 |
| Atlantic salmon (Salmo salar) co-product (trimmings and skin gelatin) | Flavourzyme 500L and Alcalase 2.4L | Salmon trimmings hydrolysate and gelatin hydrolysate | The salmon trimmings hydrolysate retained its dipeptidyl peptidase IV (DPP-IV) inhibitory and glucagon-like peptide-1 (GLP-1) secretory activities and improved its insulin secretory activity. In contrast, the gelatin hydrolysate lost its GLP-1 secretory activity | 47 | |
| Atlantic salmon (Salmo salar) trimmings | Corolase PP | Salmon trimmings protein hydrolysate (STP-C1) | The DPP-IV inhibitory activity was retained, and the peptide profiles remained similar | 48 | |
| Orange by-products | Pepsin | Fractions 45–49 and 50–54 | The α-glucosidase inhibition and α-amylase inhibition activity increased or remained unchanged | 49 | |
| Rubing cheese by-product | Papain | YPVEPF | The α-glucosidase inhibitory activity retention rate was 19.64% | 50 | |
| Poultry by-product | FoodPro PNL | YA, IY, and VL | All three dipeptides were stable | 51 | |
| Quinoa | — | Malts | The DPP-IV inhibitory activity of most malts could be significantly increased | 52 | |
| Anti-inflammatory | Dry-cured Xuanwei hams | — | Xuanwei ham peptides (XHPs) | Its anti-inflammatory effect was enhanced because of the release of novel peptides | 53 |
| Alcohol dehydrogenase (ADH) activating ability | Chickpea protein | Alcalase | Chickpea protein hydrolysates (CPHs-Pro-30) | The ADH activation rate could be maintained above 80% | 54 |
| Elastase inhibitory | Walnut (Juglans regia L.) meal | Alkaline protease | FFVPF | It could resist digestion in the gastric environment but was almost entirely hydrolyzed after gastrointestinal digestion | 16 |
| Antibacterial | Atlantic mackerel | Protamex | AMGAP | Only 2% of the intact peptide remained after digestion | 55 |
| Hepatocyte-protective | Jinhua ham | — | KRQKYD | 36.60% of KRQKYD was split into smaller fragments after intestinal digestion, yet it still demonstrated strong hepatocyte-protective activity | 56 |
2.1 ACE inhibitory peptides
ACE is associated with two blood pressure systems: the renin–angiotensin system (RAS) and the nitric oxide system (NOS). In the RAS, angiotensinogen (AGT, DRVYIHPFHLVI) is cleaved by renin to form the inactive decapeptide angiotensin I (ANG I, DRVYIHPFHL). ANG I is then hydrolyzed by ACE to form the bioactive octapeptide angiotensin II (ANG II), a potent vasoconstrictor that causes blood vessel constriction, thereby increasing blood pressure.57–59 This process is crucial for blood pressure regulation. In the NOS, bradykinin (BK) binds to β-receptors, increasing intracellular Ca2+ levels, which stimulates endothelial nitric oxide synthase (eNOS) to convert L-arginine into nitric oxide (NO).60 NO is a vasodilator that aids in maintaining normal blood pressure.61 However, ACE can deactivate BK, resulting in insufficient vasodilation and increased blood pressure.62Although synthetic ACE inhibitors like lisinopril, enalapril, benzapril, fosinopril, and captopril57 are effective at lowering blood pressure, their use as therapeutic agents is limited by side effects like taste disturbances, skin rashes, angioedema, hypotension, and dry cough.15 Food-derived ACE inhibitory peptides are considered natural and safer alternatives to synthetic drugs. However, they must either maintain their intact structure or be hydrolyzed into active products during gastrointestinal digestion, then be absorbed by the intestines to reach the cardiovascular system and exhibit their bioactivity.63
Some ACE inhibitory peptides’ bioactivity remains unchanged or increases after gastrointestinal digestion. Ma et al.2 hydrolyzed Moringa oleifera (MO) leaf using Alcalase enzymes, obtaining two ACE inhibition peptides, LGF and GLFF, with IC50 values of 0.29 ± 0.13 mM and 0.31 ± 0.04 mM, respectively. Both LGF and GLFF resisted intact gastrointestinal digestion, indicating that their bioactivity remained unchanged after digestion. Their ACE inhibitory activity and gastrointestinal stability might be due to their relatively high content of hydrophobic amino acids, such as Phe and Leu. A novel ACE inhibitory peptide, LLLLPKP (IC50 = 10.22 μM), isolated from yeast extract, was hydrolyzed into smaller peptides, particularly LLLLPK, LLLL, and LPKP, which also showed potential ACE inhibitory activity, during simulated gastrointestinal digestion. Its overall activity was not significantly affected. The ACE inhibitory activity of LLLLPKP and its digestion products might be attributed to their short chain lengths and the hydrophobic amino acid Leu.64 KYPHVF, an ACE inhibitory peptide, remained intact when exposed to gastrointestinal proteases. Its ACE inhibitory activity was unaffected during digestion, demonstrating its stability. Additionally, 44 potential antihypertensive targets were identified through network pharmacology, suggesting that KYPHVF was a multi-target, multi-pathway antihypertensive peptide.65 A recently identified ACE inhibitory peptide (NDRP) from soybean protein exhibits high stability during digestion. The activity of the digested sample only decreased to 82.47 ± 1.07%, compared to 94% for the pure peptide.66 Garcia-Mora et al.67 discovered that pepsin and pancreatin digestion enhanced the ACE inhibitory activity of lentil-derived peptides by releasing smaller peptide fragments. For example, the IC50 of LLSGTQNQPSFLSGF decreased from 119.75 ± 3.04 to 21.25 ± 1.17 μM.
However, gastrointestinal digestion may decrease or even eliminate the ACE inhibitory activities of some peptides. A pea-derived tripeptide, LRW, could not lower the blood pressure of spontaneous hypertensive rats (SHRs) due to its low gastrointestinal stability.68 The ACE inhibitory activities of two peptides from ovalbumin, YAEERYPIL and RADHPFL, decreased by approximately 100-fold after simulated digestion.69 The ACE inhibitory peptides DKVGINY and KGYGGVSL were highly susceptible to gastrointestinal digestion, with their IC50 values significantly increasing after the action of pepsin and pancreatic extract. The IC50 of DKVGINY increased from 25.2 ± 1.0 to 254.5 ± 47.0 μg mL−1, and the IC50 of KGYGGVSL increased from 0.8 ± 0.1 to 2.0 ± 0.1 μg mL−1.70
2.2 Antioxidant peptides
Reactive oxygen species (ROS), such as hydrogen peroxide, hydroxy radicals, superoxide radicals, and peroxyl radicals,71 are free radicals primarily generated by the mitochondrial respiratory chain and play a vital role in the body. However, excessive accumulation of ROS disrupts redox homeostasis, leading to oxidative stress, which is closely linked to various degenerative aging diseases, including cardiovascular diseases, Parkinson's, Alzheimer's, and cancer.72–74 Under normal circumstances, the antioxidant system maintains the balance between oxidants and antioxidants in biological tissues, protecting the body from oxidative damage. However, as age advances, the body's antioxidant function gradually diminishes, necessitating supplementation with exogenous antioxidants. Several synthetic chemical antioxidants, such as propyl gallate, butylated hydroxyanisole, tert-butylhydroquinone, and butylated hydroxytoluene,75,76 are widely used due to their excellent antioxidant properties. However, these antioxidants can have toxic side effects on human health during usage. Consequently, natural food-derived antioxidant peptides have attracted significant attention in recent years. The antioxidant potential of these peptides arises from their synergistic effects, including inhibiting lipid peroxidation, chelating transition metals, scavenging free radicals, and inducing gene expression to deactivate intracellular ROS. Peptide antioxidant activity is closely related to the peptide's composition, hydrophobicity, and conformation.77 Histidine, tyrosine, cysteine, proline, lysine, tryptophan, methionine, and aromatic amino acids can effectively scavenge free radicals, enhancing the antioxidant activity of peptides.78,79As natural alternatives to synthetic antioxidants, antioxidant peptides or hydrolysates must resist degradation by gastrointestinal enzymes, enter the portal circulation, and reach target organs and tissues in an active form to exert antioxidant effects. Some antioxidant peptides demonstrate robust functional stability during in vitro simulated gastrointestinal digestion. Antioxidant peptides GAC and SSC, purified from purple wheat bran protein hydrolysates, exhibited significant Trolox equivalent antioxidant capacity (TEAC) and demonstrated strong digestive stability.3 Furthermore, the peptides PKK, YEGGD, and GPGLM demonstrated antioxidant activity by scavenging DPPH˙ (EC50 = 0.978 ± 0.006, 1.062 ± 0.032, 1.149 ± 0.039 mg mL−1), HO˙ (EC50 = 1.158 ± 0.032, 1.234 ± 0.027, 1.285 ± 0.016 mg mL−1), ABTS+ (EC50 = 0.188 ± 0.002, 0.200 ± 0.002, 0.216 ± 0.007 mg mL−1), and O2− (EC50 = 0.924 ± 0.003, 0.933 ± 0.011, 0.969 ± 0.014 mg mL−1). These peptides remained highly stable under simulated gastrointestinal digestion for 240 min, with minimal decreases in DPPH˙ scavenging activities of 3.25%, 3.87%, and 4.30%, respectively.80 Chen et al.81 studied the effect of gastrointestinal digestion on the antioxidant activity of L. plantarum 60 fermented goat milk. Their findings revealed a slight decrease in DPPH˙ radical scavenging activity (from 68.16% to 52.68%), demonstrating good stability during gastrointestinal digestion. Zhan et al.82 isolated the peptide YDQLPEPRKPIE from porcine plasma hydrolysates; it exhibited significant antioxidant activity, especially after in vitro digestion. The antioxidant activity of YDQLPEPRKPIE was promoted by acidic amino acids (e.g., D, E) and hydrophobic amino acids (e.g., I, P, L). Additionally, amino acids and peptides (e.g., R, K, P, KP, RK, PR, PIE, PE, YDQL) generated during gastrointestinal digestion could more effectively scavenge free radicals by the resonance of the imidazole ring and interacting with other peptides. Xiao et al.83 found that chicken hydrolysates (CHs) exhibited significant antioxidant activity and resistance to simulated gastrointestinal digestion.
However, some antioxidant peptides are unstable during gastrointestinal digestion. The antioxidant peptide SNAAC, derived from Spanish dry-cured ham, is highly susceptible to gastrointestinal enzymes, resulting in over 90% decrease in activity compared to pre-digestion levels. MALDI-ToF MS analysis revealed the intact peptide's degradation and the SNAA fragment's formation during the gastric and gastrointestinal phases.84 The peptide AEEEYPDL from Spanish dry-cured ham can be digested into AEEEY and PDL fragments. Its oxygen radical absorbance capacity (ORAC) decreased from nearly 900 nM TE mg−1 to 250 nM TE mg−1, and its ABTS+ radical scavenging capacity exhibited 90% reduction after gastrointestinal digestion.85 MDLFTE and WPPD, peptides isolated from blood cockle (Tegillarca granosa), demonstrated instability during simulated gastrointestinal digestion. Their initial EC50 values (MDLFTE: 0.47 ± 0.03 mg mL−1; WPPD: 0.38 ± 0.04 mg mL−1) were significantly lower than the values after digestion (MDLFTE: 5.52 ± 0.36 mg mL−1; WPPD: 2.74 ± 0.42 mg mL−1).86
2.3 DPP-IV inhibitory peptides
Approximately 90% of diagnosed diabetes cases are attributed to type-2 diabetes, which primarily arises from inadequate insulin secretion and utilization.87 It is recognized as a multifaceted disease due to its association with several complications, including cardiovascular disease, lower limb amputation, kidney failure, and blindness.88 Fortunately, several synthetic drugs (such as pyrimidinone, xanthine, and arylmethylamine analogs) have been developed to manage type-2 diabetes.89 However, they often cause adverse side effects, such as hypertension and liver damage. Consequently, there is significant interest in natural alternatives like bioactive peptides. DPP-IV inhibitors, in particular, are emerging as promising therapeutic options due to their minimal risk of hypoglycemia.90,91 The key hormones regulating blood sugar are the incretins, specifically glucose-dependent insulinotropic polypeptide (GIP) and glucagon-like peptide-1 (GLP-1), which promptly stimulate insulin synthesis and secretion. DPP-IV, an enzyme, is primarily recognized for its antagonism ability against incretin hormones.92 Inhibiting DPP-IV prolongs the half-life of incretin hormones, thereby benefiting insulin secretion, glycemic control, and the treatment of type-2 diabetes.93Gastrointestinal digestive enzymes generally interact with DPP-IV inhibitory peptides, making them susceptible to degradation. As a result, their DPP-IV inhibitory activity often decreases or is lost after gastrointestinal digestion. For instance, the DPP-IV inhibitory activities of collagen peptides from sheep skin, GIOGVGPF, GPAGPIGPV, and GPAGPOGFPG, significantly decreased after digestion (IC50 of GIOGVGPF increased from 125.42 ± 0.81 μM to 214.12 ± 0.82 μM, IC50 of GPAGPIGPV increased from 84.19 ± 0.72 μM to 144.52 ± 0.23 μM, IC50 of GPAGPOGFPG increased from 67.12 ± 0.64 μM to 122.45 ± 0.54 μM).94 Harnedy-Rothwell et al.95 produced six blue whiting soluble protein hydrolysates (BW-SPHs). Among these, BW-SPH-B, BW-SPH-D, BW-SPH-E, and BW-SPH-F exhibited a significant reduction in DPP-IV inhibitory activity after simulated gastrointestinal digestion. Similarly, the activity of Bambara bean protein hydrolysates produced with Alcalase decreased from 44.253 ± 1.327% to 8.996 ± 0.270% after digestion.96 Pérez-Gálvez et al.97 employed the INFOGEST method to simulate the gastrointestinal digestion of lupin hydrolysates. The results demonstrated a significant reduction in the DPP-IV inhibitory activity of lupin hydrolysates. This reduction might be attributed to the digestion of bioactive peptides ranging from 1 to 3 kDa in lupin hydrolysates, releasing inactive low-molecular-weight peptide fragments. Conversely, the hydrolysis of digestive enzymes may enhance the activity of certain peptides by producing fragments with stronger DPP-IV inhibitory capacities. Rendon-Rosales et al.98 identified twelve peptides in fermented milk. Among them, P1 (QEPVLGPVRGPFIIV), P2 (YIPIQYVLS), P8 (SLPQNIPPL), and P9 (NAVPITPTLN) exhibited increased DPP-IV inhibitory effects after digestion. Digested NAVPITPTLN and SLPQNIPPL exhibited the highest activity with IC50 values of 298.47 μM and 368.24 μM, respectively. Hydrophobic amino acids and Pro residues in the digestion products of peptides P8 (SL, PQNIPPL) and P9 (NA, PITPTL) could facilitate their binding to the DPP-IV catalytic site. Pei et al.5 identified the DPP-IV inhibitory peptide VPLVM from broccoli. The hydrophobic Pro and Leu residues near the N-terminus conferred DPP-IV inhibitory activity to VPLVM. Most VPLVM remained undigested by gastrointestinal enzymes, demonstrating an overall stability of 84.58%. A portion of VPLVM was hydrolyzed into VPLV and PLV. Moreover, mice experienced a 20.7% reduction in blood glucose levels 30 min after VPLVM administration compared to the control group. The DPP-IV inhibitory peptides NLEIILR and TQMVDEEIMEKFR demonstrated good stability after simulated gastrointestinal digestion. The inhibitory ability of TQMVDEEIMEKFR increased by 21.00%, while the activity of NLEIILR remained unchanged.99 Simulated gastrointestinal digestion in vitro of VPA, VPW, IPL, and IPR present in Chlorella vulgaris suggested that these peptides were likely resistant to gastrointestinal digestion and could retain their DPP-IV inhibitory activity. The hydrophobic Val residue at the N-terminus and the Pro residue at the second position from the N-terminus of these peptides might play key roles in their activity and gastrointestinal stability.100
2.4 Anticancer peptides
Despite advancements in research and treatment methods, cancer remains increasingly prevalent worldwide and is a significant cause of morbidity and mortality. Most cancers, apart from chronic leukemia, are still regarded as chronic diseases that cannot be completely eradicated. Conventional cancer treatments, like chemotherapy and radiotherapy, often cause severe side effects due to damage to healthy cells and tissues.101 The demand for innovative treatments has spotlighted peptides as potential drug candidates due to their several key advantages over conventional chemotherapy molecules. Peptides exhibit low toxicity, strong specificity, high affinity, and good tissue penetration.102 Cancer cells can evade signals that inhibit growth, allowing them to resist cell death and multiply. Therefore, inducing apoptosis has emerged as a viable approach for managing cancer. Research has demonstrated that peptides are involved in multiple stages of carcinogenesis, such as angiogenesis, apoptosis, and metastasis.103Similarly, gastrointestinal digestion may affect the activity of anticancer peptides. For instance, Sah et al.104 discovered that simulating the gastrointestinal tract significantly enhanced the antiproliferative activities of the anticancer peptides from synbiotic yoghurt against HT-29 colon cancer cells; this was attributed to the generation of potent bioactive peptides. Specifically, the digestate of YQEPVLGPVRGPFPIIV (at 3 mg mL−1) exhibited an activity of 86.63 ± 0.57%, while the digestate of SLPQNIPPLTQTPVVVPPF (at 3 mg mL−1) showed an activity of 87.60 ± 0.89%. Both undigested peptides (H. Met-Gly-Pro-Pro-Gly-Leu-Ala-Gly-Ala-Pro-Gly-Glu-Ala-Gly-Arg. OH) isolated from flathead by-products and those resulting from gastric and gastrointestinal digestion possessed anticancer activity, inhibiting the proliferation of HT-29 cells by up to 28.89%, 29.68%, and 38.3%, respectively. The highest rate of cell death (3.54 ± 2.30%) was observed in HT-29 cells treated with peptides digested by pepsin and pancreatin.105 Xue et al.106 investigated the inhibitory activity of chickpea peptide CPe-III (RQSHFANAQP) on tumor proliferation in vitro. They conducted both in vitro simulated and in vivo digestion in mice, identifying Ser-His dipeptide as a common product in both processes. Further research revealed that this metabolite was a potential anticancer agent, inhibiting tumor cell proliferation.
2.5 Bioactive peptides with other functions
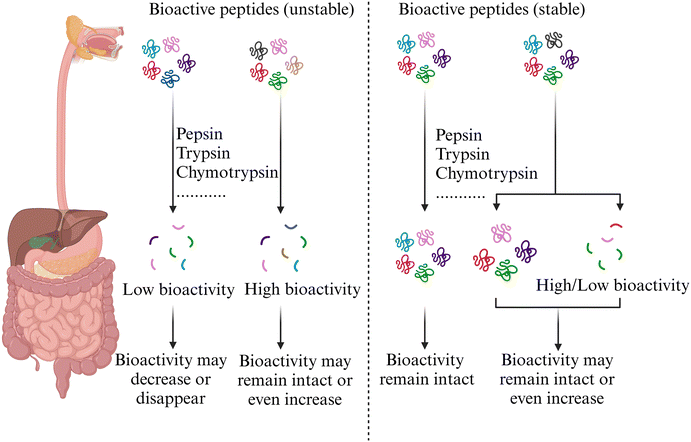
Besides investigating the ACE inhibitory peptides, antioxidant peptides, DPP-IV inhibitory peptides, and anticancer peptides, extensive research has also focused on the gastrointestinal stability of other bioactive peptides. For instance, Li et al.107 suggested that peptides derived from Staphylococcus simulans QB7-fermented sausage, inhibiting cyclooxygenase 2 (COX2) or vascular cell adhesion molecule 1 (VCAM-1) expression, likely consisted of different sequences. Peptides inhibiting COX2 expression probably showed greater resistance to gastrointestinal digestion, whereas those targeting VCAM-1 expression might have been less resistant, degrading into smaller peptides while retaining remarkable VCAM-1 inhibitory capability. The peptide identification results indicated that gastrointestinal digestion led to the release of numerous peptide sequences containing positively charged and hydrophobic amino acids. This finding explained why the digested peptides exhibited high anti-inflammatory activity. The anti-inflammatory activity of IVYPWTQR from goose blood hydrolysate slightly decreased after pepsin digestion, as 45.19% of IVYPWTQR was cleaved into smaller fragments, including IV (20.00%) and YPWTQR (25.19%), which were resistant to trypsin.108 Bovine-elastin-derived elastase inhibition peptides GAGQPFPI, FPGIG, and FFPGAG exhibit digestion resistances of 66.3%, 64.0%, and 61.3%, respectively.109 Yang et al.110 investigated the α-glucosidase inhibitory activity of hot-pressed peanut meal protein hydrolysates (PMHs) after in vitro gastrointestinal digestion. They discovered that this activity remained stable and even increased in the final stages. The ADH activation activity of chicken breast muscle hydrolysates (CBMHs) was reduced by 33.42% after simulated intestinal digestion compared to the untreated sample, due to the degradation of hydrophobic peptides into less active forms.111 Liu et al.112 identified cholesterol-lowering peptides ALPM, AVFK, and HTSGY from whey protein and found that their activity decreased by 5.43 ± 0.87%, 13.32 ± 1.30%, and 9.52 ± 0.89% after digestion, while their bioavailability was 68.19%, 72.16%, and 83.66%, respectively. The retention of cholesterol-lowering activity might stem from an elevated level of hydrophobic amino acids, enhancing their capacity to bind conjugated bile acids. The peptide β-casein (f193–209) (m/z 1881), derived from Prato cheese and exhibiting immunomodulatory, ACE inhibitory, antimicrobial, and antithrombotic activities, displayed a prominent peak after static digestion and was completely hydrolyzed during dynamic digestion. However, other studies demonstrated that this peptide could resist gastrointestinal digestion.113 This suggests that different experimental protocols may lead to contradictory results, emphasizing the need for developing standardized experimental methods. Vivanco-Maroto et al.114 employed a semi-dynamic protocol to investigate the effects of simulated gastrointestinal digestion on peptides in precursor micellar casein and casein hydrolysates that could induce the secretion of GLP-1 and cholecystokinin (CCK) from STC-1 cells. The results indicated that the peptides VYPFPGPIPN, YPFPGPI, and VYPFPGPI derived from β-casein consistently exhibited high intensity during the intestinal digestion process.The activity of bioactive peptides after digestion hinges on their stability and the bioactivity of their digestion products (Fig. 2). If the peptide remains relatively stable during gastrointestinal digestion or its hydrolysis products have high bioactivity, its overall activity will largely remain intact or even increase.64–67,80,98 Conversely, if the peptide is highly susceptible to digestive enzymes and the resulting small peptides exhibit low bioactivity, the peptide's bioactivity may significantly decrease or nearly disappear after digestion.68,84,86,95 Since peptide bioactivity is easily affected by gastrointestinal digestion, several studies employ simulated gastrointestinal digestion to derive bioactive peptides. For instance, the novel ACE inhibitory peptide GHIITVAR (IC50 = 3.60 ± 0.10 μM) was identified from sesame (Sesamum indicum L.) protein using in vitro two-stage simulated gastrointestinal digestion.115 Zhang et al.116 identified five novel antioxidant peptides through simulated digestion. Among these, IREADIDGDGQVN (1.78 mM), PEILPDGDHD (1.18 mM), and ASDEQDSVRL (1.45 mM) exhibited the lowest DPPH˙ radical scavenging IC50 values. Additionally, APLEEPSSPH demonstrated the most effective Fe2+ chelating capability with an IC50 of 0.09 mM. Suárez et al.117 utilized an in vitro dynamic simulated digestion system to identify the peptide sequence IERGEGIMGV, which was likely to inhibit ACE, from the final digesta of an amaranth beverage (AB). Wu et al.118 found that the digestive products of fermented soymilk contained bioactive peptides with α-glucosidase, DPP IV and ACE inhibitory and antioxidant activities. The highest relative quantity of bioactive peptides was released through the combined action of Propionibacterium shermanii and Lactobacillus plantarum. ACE inhibitory peptides LLR and AWR, derived from the gastrointestinal digestion products of sufu hydrolysates, exhibited IC50 values of 94.01 ± 5.07 μM and 98.04 ± 2.56 μM, respectively. In vivo experiments demonstrated that both peptides significantly reduced diastolic and systolic blood pressure in spontaneously hypertensive rats.119 A recent review screened 17![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 675 articles and selected 31 that assessed the structural stability of bioactive peptides during in vitro simulated gastrointestinal digestion.120 This research identified 93 unique peptide sequences and systematically elucidated the characteristics of stable peptides. The findings indicated that stable peptides had higher proportions of Pro and hydrophobic residues, shorter chain length and lower molecular weight, no C-terminal Leu, more basic amino acid residues, and branched-chain aliphatic N-terminal residues.
675 articles and selected 31 that assessed the structural stability of bioactive peptides during in vitro simulated gastrointestinal digestion.120 This research identified 93 unique peptide sequences and systematically elucidated the characteristics of stable peptides. The findings indicated that stable peptides had higher proportions of Pro and hydrophobic residues, shorter chain length and lower molecular weight, no C-terminal Leu, more basic amino acid residues, and branched-chain aliphatic N-terminal residues.
3. Encapsulation strategies to enhance the gastrointestinal stability of bioactive peptides
Due to the uncertain bioactivity of digestion products, directly preparing bioactive peptides with gastrointestinal enzymes or designing peptides to resist digestive enzymes offers benefits but reduces diversity. Therefore, developing strategies to enhance the gastrointestinal stability of bioactive peptides is crucial. Encapsulation, including microcapsules, gels, emulsions, liposomes, and nanofibers (Fig. 3), is a prevalent approach today, as illustrated in Table 2.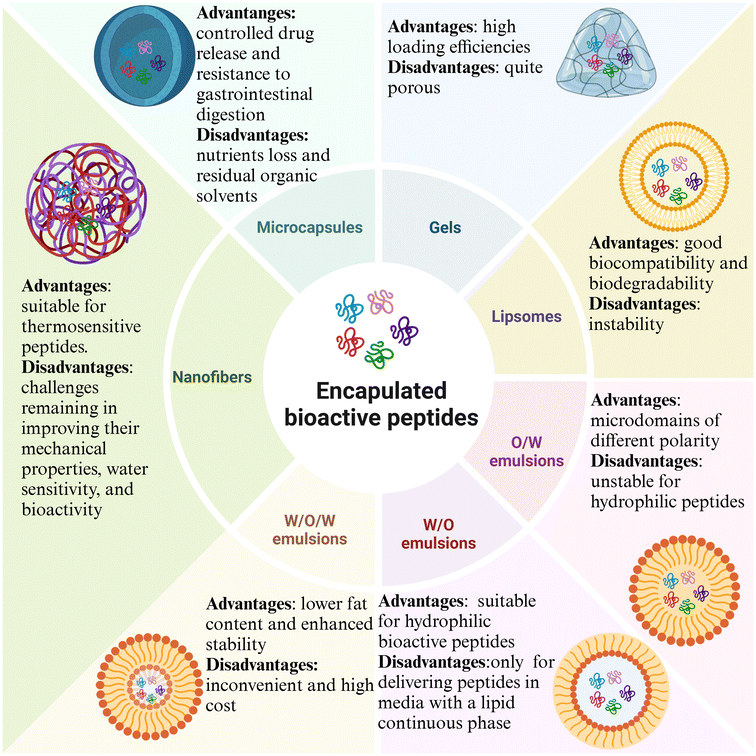 | ||
| Fig. 3 Schematic diagrams of microcapsules, gels, emulsions, liposomes, and nanofibers for the encapsulation of bioactive peptides. | ||
| Encapsulation method | Material | Hydrolysate name/peptide sequences | Advantage | Ref. |
|---|---|---|---|---|
| Microcapsule | 10% cluster dextrin | Fractionated riceberry bran protein hydrolysate obtained by ultrafiltration <3 kDa (URBPH < 3 kDa) | Encapsulated peptides retained their antioxidant activity better during digestion compared to the free form | 121 |
| Agar (Agarforce®) and maltodextrin (15 DE) | Peptides obtained from brewer's spent grain (BSG-P) | Microencapsulation improved the hypolipidemic activity of BSG-P observed under simulated digestion | 122 | |
| Pyropia columbina phycocolloids | Brewers’ spent grain ACE-I inhibitory peptides | Encapsulated peptides exhibited a lower IC50 value (2.4 ± 0.2 mg mL−1 protein) than unencapsulated peptides (7.2 ± 0.3 mg mL−1 protein) | 123 | |
| Alginate | Plantaricin EF (PlnEF) | Mixing PlnEF protected by crosslinked alginate microcapsules (CLAMs) into peanut butter (PB) safeguarded it from proteolysis and enhanced its intestinal release | 124 | |
| Gel | Chitosan-tripolyphosphate (TPP) | Atlantic codfish skin-derived collagen hydrolysates | Encapsulated hydrolysates released approximately 58% of their content during digestion | 125 |
| Casein and alginate | Protein nanoparticles consisting of a zein core and a whey protein shell | These microgels can potentially retain and protect protein nanoparticles in the gastric phase while facilitating their release in the intestinal phase | 126 | |
| Emulsion | Amaranth proteins and sunflower oil | Amaranth protein hydrolysates (AH) | The ACE inhibitory activity was maintained after simulated gastrointestinal digestion | 127 |
| Canola oil and tween 80 | Collagen peptide (CP) | W1/O/W2 emulsions achieved the highest collagen peptide retention, with an encapsulation efficiency of over 90% | 128 | |
| Polyethylene glycol 40 stearate (PEG 40 St) and soybean oil | Bovine lactoferrin (LF) | After gastric digestion, 84% of the LF in these emulsions was protected. Under intestinal conditions, the PEG-LF emulsions demonstrated good stability and slower lipolysis | 129 | |
| Liposome | CS-TPP/liposomes | Fish-purified antioxidant peptide fractionated by RP-HPLC (F5) | The antioxidant activity of F5 loaded into liposomes was retained after digestion | 130 |
| Soy phosphatidylcholine liposomes with the addition of glycerol | Collagen hydrolysate | The liposome membrane effectively protected the active compounds from interactions within the gel matrix or degradation | 131 | |
| Nanofiber | Bioactive electrospun fish sarcoplasmic proteins (FSP) | Insulin | Insulin was efficiently encapsulated, allowing for its controlled release in simulated intestinal fluid while protecting against chymotrypsin degradation | 132 |
| Electrospun chitosan/poly(vinyl alcohol) (CS/PVA) | Fish-purified antioxidant peptide (AOP) | The encapsulated antioxidant peptide retained its activity and showed sustained release | 133 |
The digestive behavior of these wall materials is crucial for protecting bioactive peptides during digestion and ensuring their controlled release, thereby enhancing their bioavailability and therapeutic efficacy. Polysaccharides and proteins are the primary wall materials used to encapsulate bioactive peptides, including forms such as microcapsules, gels, and nanofibers. Digestion of polysaccharide wall materials, primarily starch, begins with salivary amylase in the oral cavity. Although the acidic environment of the stomach inhibits this process, pancreatic amylase, lactase, sucrase, and maltase in the intestines further break down the polysaccharides. Conversely, if the wall material is a dietary fiber-type polysaccharide, such as pectin or alginate, its resistance to digestion may make it suitable for encapsulating bioactive peptides intended for absorption in the colon.134 It is worth noting that when dietary fiber-type polysaccharides (e.g., alginate) and starch are used together as wall materials, the release of the core materials from the system may be compromised.135 Furthermore, when polysaccharides are used as encapsulating materials, swelling may occur during the digestion process. For example, during the intestinal phase, higher pH levels increase electrostatic repulsion between gel-based pectin polymer chains, resulting in swelling and the subsequent release of peptides.134
If the wall material is a protein, such as casein or gelatin, digestive enzymes initiate its degradation with pepsin in the stomach and subsequently break it down further with intestinal proteases like trypsin and chymotrypsin, leading to the controlled release of bioactive peptides. Additionally, the acidic pH of the stomach may induce the dissociation of protein-based delivery systems. As the gastric pH approaches the isoelectric point of proteins, they tend to aggregate and precipitate, thereby compromising the stability of the delivery system.136 When proteins are combined with polysaccharides as wall materials, various electrostatic interactions between proteins and polysaccharides, as well as between core materials and polysaccharides, may affect the digestion of wall materials, potentially altering the release profile of bioactive peptides.137
Lipids are utilized as encapsulating materials in liposome and emulsion delivery systems. Lipid digestion is initiated by gastric lipase and continues in the intestinal phase, where pancreatic lipase plays a key role. Furthermore, bile salt aids in the removal of free fatty acids and proteins from the surface of oil droplets, thereby increasing the available surface area for enzymatic action.138 In emulsions, emulsifiers, such as polysaccharides and proteins, significantly affect lipid digestion rates by determining the system stability and oil droplet size.139 Liposomes, composed of one or more phospholipid bilayers, are unstable in the gastrointestinal tract. Their stability can be enhanced through modifications, as discussed in detail in subsequent sections.
3.1 Microcapsules
In recent years, the food industry has extensively employed microcapsules for the encapsulation of bioactive peptides.140,141 It involves enclosing sensitive materials within a physical barrier, forming small particles. Various materials have been used as wall materials for preparing microcapsules, including maltodextrin, gum arabic, chitosan, casein, and sodium alginate. Spray-drying and freeze-drying are commonly used methods for microencapsulating various bioactive compounds.142 Spray drying is a dehydration technique that converts a feed solution into powder. The process involves atomization of the feed solution, drying, and particle separation.143 Freeze-drying creates microcapsules by sublimating frozen emulsion. This process includes freezing, ice sublimation, and secondary drying.144 Encapsulating bioactive peptides in microcapsules provides several advantages, including controlled drug release rates, reduced toxicity, resistance to gastrointestinal digestion, protection against chemical and enzymatic degradation, and enhanced bioavailability.145 Corrêa et al.146 encapsulated bioactive sheep whey hydrolysates using prebiotic gum arabic as the wall material. The microcapsules demonstrated higher antioxidant and antihypertensive activities after simulated gastrointestinal digestion than non-encapsulated bioactive sheep whey hydrolysates. Cian et al.147 used agar and maltodextrin to prepare spray-dried microcapsules of brewers’ spent grain protein hydrolysate (BSGH). During in vitro simulated gastrointestinal digestion, encapsulated BSGH had a cleavage rate of 39.2%, compared to 66.5% for non-encapsulated BSGH. Microencapsulation also preserved the in vitro antioxidant activity. Cian et al.148 examined the bioactivity of Phaseolus lunatus peptides microencapsulated with maltodextrin and gum arabic as wall materials following digestion. The results indicated that microencapsulation preserved these peptides’ α-glucosidase, α-amylase, and DPP-IV inhibition activity. Xun et al.149 prepared caffeic acid grafted chitosan–sodium alginate microcapsules using microfluidics to efficiently encapsulate silkworm pupae bioactive peptide. Peptide release from the microcapsules was 9.76 ± 0.23% during simulated gastric digestion, and the microcapsules demonstrated good intestinal release. Additionally, the DPPH˙ radical scavenging activity of the microcapsules was 31.15 ± 0.99%, compared to 13.31 ± 0.63% in the control group.Current research on microcapsules primarily focuses on microparticle characteristics, encapsulation efficiency, processing parameters, and drug release duration. Utilizing non-toxic and safer food ingredients as wall materials opens new avenues for the development of microcapsules. Recently, plant proteins were employed as encapsulating agents, driven by the growing trend in specialized markets to restrict or limit animal products.150 For instance, potato protein, with its functional properties and valuable nutritional content, is a potential alternative wall material. Furthermore, studies show that modifying plant proteins can significantly improve microparticle encapsulation efficiency through heat treatment, enzymatic hydrolysis, and acylation.151 Future efforts should concentrate on devising strategies to enhance the preservation of peptide activity within microcapsules.152
3.2 Micro- and nanogels
Micro- and nanogels primarily consist of water and cross-linked polymer molecules, often polysaccharides and proteins.153 Common polysaccharides include alginate, chitosan, methyl cellulose, and pectin, while common proteins are albumin, caseins, gelatin, and ovalbumin.154 Their functional properties can be customized for specific applications by precisely controlling their dimensions, porosities, compositions, interfacial characteristics, and morphologies.155 Various methods can be used to prepare these colloidal particles, such as emulsion templating, coacervation, antisolvent precipitation, and injection gelation.153,156 Gels can achieve high loading efficiencies (LEs) due to favorable partitioning and have been extensively studied for encapsulating, protecting, and delivering bioactive agents.155 Free-Manjarrez et al.157 found that Greek-style yogurt with common bean protein hydrolysates encapsulated in a gel showed 44% reduction in bitterness and 52% decrease in astringency compared to control yogurt with unencapsulated hydrolysates. Additionally, yogurt containing encapsulated hydrolysates inhibited α-amylase (92.47%) and DPP-IV (75.24%) after digestion. Obuobi et al.158 encapsulated antimicrobial peptides within DNA nanogels, preserving bioactivity and structural integrity. They demonstrated that the sustained release of these encapsulated peptides resulted in significant antimicrobial effectiveness against S. aureus.However, gels enable bioactive peptides to diffuse out easily due to their porous nature. This issue can be mitigated by reducing the pore size of the gel or enhancing the interactions between the peptides and the biopolymer.156
3.3 Liposomes
Liposomes are colloidal structures composed of one or more phospholipid bilayers, with the non-polar tails of the molecules facing inward due to hydrophobic interactions.159,160 Liposomes can encapsulate hydrophilic bioactive peptides between the polar head groups or within their aqueous core. Additionally, hydrophobic bioactive peptides can be embedded within the bilayers. Due to their phospholipid composition, liposomes may exhibit good biocompatibility, biodegradability, cell membrane permeability, and low toxicity and immunogenicity, making them widely used for encapsulating, protecting, and delivering bioactive peptides.160 Pavlovic et al.161 encapsulated soy protein hydrolysate in liposomes and found that, under simulated gastrointestinal conditions, liposomes provided the sustained release of the encapsulated hydrolysates. In the first 75 min, only 20% of the encapsulated soy peptides diffused out. The osteogenic activity of osteogenic peptides (OPs) encapsulated in nanoliposomes remains unaffected, with their cumulative release in phosphate buffer solution (PBS) within 8 h (58.2%) being significantly slower than that of non-encapsulated OPs (68.5%).162However, a major drawback of liposomes as delivery systems is their instability during digestion.163 Therefore, it is essential to consider their digestibility and optimize various factors to achieve ideal liposome digestion performance. For instance, improving liposome stability during digestion can be achieved by altering the type of phospholipid used. Huang et al.164 developed liposomes modified with stearic acid (Lipo-SA) and decanoic acid (Lipo-DA) to encapsulate the peptide LKPNM. After 180 min of incubation in simulated gastric fluid, the peptide release percentages for Lipo-Pep, Lipo-SA-Pep, and Lipo-DA-Pep were 77.73 ± 2.03%, 67.01 ± 0.61%, and 68.51 ± 0.77%, respectively. In simulated intestinal fluid, the corresponding release percentages were 85.58 ± 2.01%, 72.4 ± 3.79%, and 78.24 ± 3.99%, indicating that modified liposomes exhibited better peptide preservation. Niu et al.165 discovered that liposomes containing bile salts (BS-liposomes) demonstrated enhanced gastrointestinal stability and prolonged residence time compared to conventional liposomes.
3.4 Emulsions, nanoemulsions, and multiple emulsions
Emulsions are colloidal suspensions formed by blending water and oil, with one liquid forming small droplets within the other.166 These emulsions can be categorized as water-in-oil (W/O) or oil-in-water (O/W) based on the arrangement of the water and oil phases. Furthermore, they can be classified as either emulsions (d > 100 nm) or nanoemulsions (d < 100 nm) based on the mean droplet diameter.167 According to McClements,17 since both emulsions and nanoemulsions are thermodynamically unstable systems with similar structural and physicochemical properties, the term “emulsion” is used in this article to describe both types.From a drug delivery perspective, emulsions are appealing because they contain microdomains of different polarity within a single-phase solution, enabling the solubilization of hydrophilic or hydrophobic materials. However, significant challenges exist in encapsulating hydrophilic peptides into the oily core of O/W emulsions. Due to their polarity, peptides may remain at the oil–water interface.168 Additionally, bioactive peptides can adsorb onto the surfaces of emulsifier-coated oil droplets because of electrostatic interactions, compromising protection.169 Current strategies to better protect them include converting peptides into a hydrophobic form for incorporation into O/W emulsions and encapsulating protein-coated droplets within other protective systems, such as microgels.17,155 In contrast, W/O emulsions are more suitable for encapsulating hydrophilic bioactive peptides because their hydrophilic interior better accommodates these peptides.170 For example, Gao et al.171 effectively encapsulated bitter peptides in W/O emulsions, significantly mitigating their bitterness. Additionally, encapsulation improved the gastric stability of the peptides, shielded them from pepsin in the stomach, and facilitated the delivery of more intact peptides to the intestines. Nevertheless, W/O emulsions are only effective at delivering peptides in drugs, supplements, or functional foods with a lipid continuous phase, such as oil-filled capsules.166 Fortunately, W/O/W emulsions can overcome this drawback. They are created by dispersing a W/O primary emulsion into an external aqueous phase containing an emulsifier.172 There are two main reasons for the steady increase in researchers’ interest in W/O/W systems. Firstly, W/O/W emulsions have a lower fat content than traditional O/W emulsions, while maintaining a similar mouthfeel. Secondly, they offer enhanced stability and more effectively protect sensitive water-soluble components. However, W/O/W systems also present challenges, including difficult and time-consuming preparation, high costs, and susceptibility to decomposition during storage.172 Giroux et al.173 investigated the impact of the oil phase composition (butter, linseed oil, or mineral oil) and the degree of hydrolysis (0%, 3.5%, or 7%) on the release kinetics of casein-derived peptides encapsulated in W/O/W double emulsions during digestion. Peptides with 3.5% and 7% hydrolysis showed faster release rates compared to the unhydrolyzed group in both digestion phases. After the gastric phase, peptide release ranged from 4% to 42%. During the intestinal phase, peptide release in double emulsions with linseed oil and butter exceeded 80%, whereas release in emulsions with mineral oil was below 18% due to its indigestibility.
3.5 Nanofibers
Electrospinning is a facile, innovative, and promising technique for preparing nanofibers with porosity and a high surface-to-volume ratio.174 Bioactive peptides can be encapsulated into nanofibers by mixing them with a biopolymer solution and electrospinning. Commonly used biopolymers include proteins such as whey protein concentrate (WPC), collagen, and zein, as well as polysaccharides like chitosan, pullulan, dextrans, and starch.175 Since nanofibers are prepared under ambient conditions, they are more suitable for encapsulating thermosensitive peptides compared to techniques such as spray drying.174 Additionally, the room temperature processing route enables efficient encapsulation by enhancing the stability of bioactive peptides. Recently, nanofibers produced via electrospinning attracted significant attention. Rajanna et al.176 synthesized casein-derived peptide-loaded pullulan nanofibers via electrospinning at pullulan concentrations of 120 and 140 g kg−1, achieving mean diameters ranging from 60.45 to 133.05 nm and encapsulation efficiencies between 72.95% and 86.04%. Peptides from electrospun nanofibers exhibited sustained release under gastrointestinal pH conditions. Using electrospinning, Kirbas and Altay177 prepared pullulan–sodium alginate nanofiber carriers loaded with L-carnosine (Car). The electrospinning process preserved Car antioxidant activity and enabled its sustained release during digestion.Although nanofibers hold significant promise for various applications, challenges remain in improving their mechanical properties, water sensitivity, and bioactivity. Methods such as modifying biopolymers, and adding nanoparticles and bioactive components can improve the properties of nanofibers. For instance, antimicrobial aromatic compounds (AACs) are extensively used as property modifiers and bioactive components in various biopolymer nanofibers to improve their thermal properties, hydrophobicity, functionality, and mechanical properties.178 Future research should delve deeper into the interactions between small molecules and biopolymers, elucidating the effects of these molecules on biopolymer properties.
4. Conclusion and future research perspectives
Bioactive peptides isolated from food proteins exhibit ACE inhibitory, antioxidant, DPP-IV inhibitory, anticancer, and anti-inflammatory activities. However, their in vitro activity does not always correlate with in vivo functionality due to hydrolysis by digestive enzymes. The activity of bioactive peptides after exposure to digestive enzymes can remain unchanged, decrease, or increase, depending on the stability of the peptides and the bioactivity of their hydrolysis products. If a bioactive peptide exhibits good gastrointestinal stability or its hydrolysis products retain high activity during transit, its overall activity may be maintained or even enhanced. Conversely, if a peptide is highly susceptible to gastrointestinal enzymes and its hydrolysis products have low activity, digestion will significantly reduce the activity. Therefore, the effect of gastrointestinal digestion on peptide bioactivity is uncertain. To address this uncertainty, some studies directly prepare bioactive peptides through simulated gastrointestinal digestion, but this method is time-consuming and less effective. Therefore, enhancing the gastrointestinal stability of bioactive peptides is a more reliable approach to maintaining their activity. Encapsulation is widely used to improve bioactive peptides’ gastrointestinal stability and bioavailability.The ability of bioactive peptides to reach target organs and cells in their active form depends on their gastrointestinal stability and stability during transepithelial transport, blood metabolism, and kidney and liver metabolism (Fig. 4). To save resources, reduce costs, and protect experimental subjects, preliminary in vitro pharmacokinetic studies are necessary before clinical trials. Bioactive peptides must retain their activity after being processed by brush border peptidases and passing through intestinal epithelial cells. The primary mechanisms for bioactive peptide transport across intestinal epithelial cells include peptide transportable 1 (PepT1), the paracellular route, and transcytosis. Once bioactive peptides enter the bloodstream, they encounter various peptidases and proteases and must resist their action. Common peptidases in the blood include aminopeptidase N, carboxypeptidase N, plasmin, DPP4, furin, and neprilysin.179 Additionally, bioactive peptides are easily filtered and excreted from the body by the kidneys and metabolized by the liver.
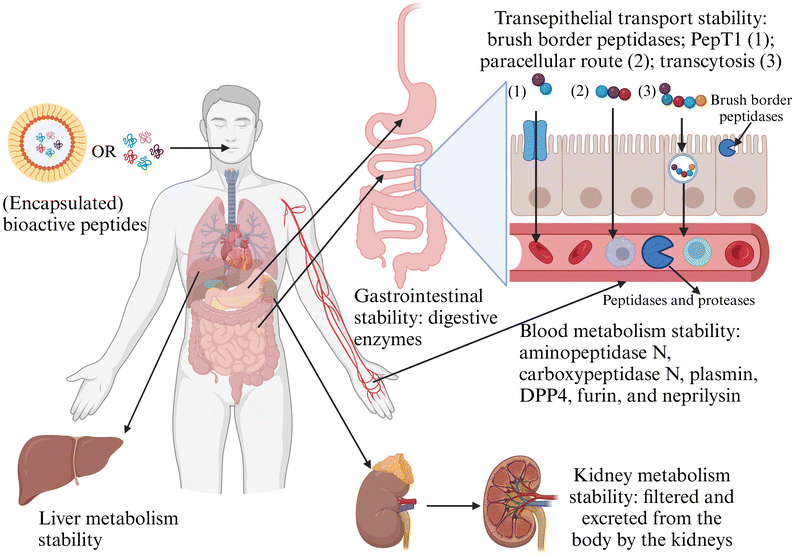 | ||
| Fig. 4 Gastrointestinal, transepithelial transport, blood metabolism, liver metabolism, and kidney metabolism stability challenges encountered by bioactive peptides. | ||
Furthermore, bioactive peptides’ allergenicity, toxicity, and bitterness must be considered before clinical trials and commercial application. Proteins are the primary allergens in food, and the development of some novel food processing technologies may help avoid or control these allergens.180In vitro tests, such as the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay, are commonly used to assess the potential toxicity of proteins.181 The taste of bioactive peptides is a crucial factor affecting their commercial application. Current techniques to reduce peptide bitterness include encapsulation, enzymatic treatment, and removal of hydrophobic residues.182
If in vitro experiments confirm that bioactive peptides can maintain their activity under various conditions and are non-allergenic and non-toxic, further in vivo experiments are necessary to support their beneficial impact. Martin et al.183 investigated the in vivo antihypertensive effects of a whey protein hydrolysate containing peptides WL and IW. Although the hydrolysate significantly reduced plasma ACE activity (to 86.4 ± 5.9% and 75.1 ± 6.9% of baseline activity after ingestion of 5 g and 50 g hydrolysate, respectively), it did not lead to a reduction in blood pressure over the study period. Recently, the in vivo antihypertensive effects of processed rice bran, containing the antihypertensive peptide LRA, were evaluated over a 12-week trial. The results indicated that a daily dosage of 43 μg LRA significantly reduced the blood pressure of volunteers with mild hypertension compared to the placebo group (p < 0.05).184 A milk protein hydrolysate with α-glucosidase inhibitory activity was shown to exert a postprandial glycemic effect. Low doses of the hydrolysate significantly reduced postprandial plasma glucose concentrations in subjects compared to the placebo (p < 0.05) and mildly regulated insulin secretion.185 Similarly, the in vivo activities of certain encapsulated bioactive peptides have been validated. A 35-day study demonstrated that encapsulating carp skin gelatin hydrolysate in furcellaran-based microcapsules reduced its in vivo antioxidant activity. This outcome may be attributed to the incomplete digestion of the wall materials in the small intestine, preventing the hydrolysate from being released. Therefore, selecting an appropriate wall material is crucial for ensuring that bioactive peptides can exert their effects in vivo.186 During a 100-day administration period, encapsulated brewers’ spent grain peptides effectively lowered blood glucose and blood pressure in an insulin-resistant and hypertensive rat model induced by a sucrose-rich diet.187 Clinical trials are essential for bridging in vitro activity with in vivo function and represent a crucial stage for the commercial application of bioactive peptides. However, few animal models and even fewer clinical trials have been conducted, with the existing ones primarily focusing on antihypertensive peptides. Future research must provide robust evidence for the health claims surrounding bioactive peptides through well-designed clinical trials.
Regulatory approvals are essential for protecting consumers from fraudulent claims, deceptive practices, and risks related to bioactive peptides.188 First, it is necessary to determine whether bioactive peptides should be classified as food or drugs. Generally, they are classified based on their intended use. If a product is consumed for sustenance and nourishment, it should be considered a food; if it is designed to treat a disease, it should be regarded as a drug.189 Bioactive peptides are usually classified as food for human consumption, complying with the same safety regulations as traditional food items.190 Although researchers have isolated numerous food-derived bioactive peptides, only a few have reached commercial applications. Additional animal studies, clinical trials, and regulatory approvals are needed to advance the commercial application of bioactive peptides.
Due to factors such as low cost, minimal equipment requirements, and ease of operation, most current studies use static methods to investigate the effects of simulated gastrointestinal digestion on bioactive peptides, with few employing semi-dynamic or dynamic approaches. Although static methods are convenient, they are primarily suited to evaluating digestion endpoints rather than the digestion process itself. Moreover, simple mixed solutions cannot fully replicate actual body fluids, potentially leading to inaccurate results. Another limitation is that different studies may employ various static methods to simulate gastrointestinal digestion. For example, Ma et al.2 added 1% pepsin to the solution and incubated it for 2 h at a pH of 2.0. Then, they adjusted the pH to 7.5, added 1% trypsin, and incubated the mixture for another 4 h. However, Gallego et al.85 added pepsin (2000 U mL−1) to the solution, digesting the mixture at a pH of 3.0 for 3 h. Then, they adjusted the pH to 7.0 and added pancreatic lipase (2000 U mL−1), pancreatic α-amylase (200 U mL−1), chymotrypsin (25 U mL−1), and trypsin (100 U mL−1) for another 3 h of incubation. Variations in enzyme amounts and reaction conditions, like solution composition, reaction time, and pH, may result in different outcomes. The enzyme source is also crucial but different studies may use trypsin from bovine or porcine origins, affecting the results. Therefore, future studies should concentrate on developing more standardized and realistic digestion simulation methods that closely mimic human conditions.
In recent years, bioinformatics has been utilized to predict peptides’ allergenicity, toxicity, taste-evoking properties, potential bioactivity, and the hydrolytic actions of gastrointestinal proteases, enhancing efficiency and reducing costs.191 For example, Nongonierma and FitzGerald192 performed in silico gastrointestinal digestion on eight casein-derived DPP-IV inhibitory peptides and found that NP and IP were resistant to cleavage by pepsin, trypsin, and chymotrypsin. However, in silico simulated digestion differs from actual digestion. In silico gastrointestinal digestion is a database-based simulation assuming that all peptide bonds theoretically susceptible to a given digestive enzyme are hydrolyzed. However, peptide hydrolysis is often incomplete under actual conditions due to factors such as temperature, pH, duration, the availability of bonds, and the complexity of the gastrointestinal environment. As bioinformatics advances, exploring more realistic in silico gastrointestinal digestion simulations becomes increasingly valuable.
Bioactive peptides demonstrate significant potential for disease prevention and treatment. However, challenges such as poor gastrointestinal stability must be addressed before commercial application, necessitating further attention and research for their development.
Author contributions
Chenlong Chen: conceptualization, formal analysis, investigation, methodology, project administration, software, visualization, writing – original draft, writing – review & editing. Wancong Yu: formal analysis, investigation, methodology, writing-review & editing. Xiaohong Kou: conceptualization, visualization, writing – original draft. Yujia Niu: software, visualization. Jiaxin Ji: project administration. Ying Shao: writing – original draft. Shuqi Wu: visualization. Mengyi Liu: project administration. Zhaohui Xue: conceptualization, formal analysis, methodology, funding acquisition, writing – review & editing.Data availability
No primary research results, software or code have been included and no new data were generated or analysed as part of this review.Conflicts of interest
There are no conflicts to declare.Acknowledgements
This work was supported by grants from the National Natural Science Foundation of China (No. 31571825), the 2024 Graduate International Teaching System Construction Project (ENT24004), the Tianjin University Independent Innovation Foundation (2024XC-0001), and the 2024 Education and Teaching Reform Project (XK2415). BioRender.com was used to create the figures.References
- M. Chalamaiah, S. K. Ulug, H. Hong and J. Wu, Regulatory requirements of bioactive peptides (protein hydrolysates) from food proteins, J. Funct. Foods, 2019, 58, 123–129 CrossRef CAS.
- K. Ma, Y. Wang, M. Wang, Z. Wang, X. Wang, X. Ju and R. He, Antihypertensive activity of the ACE-renin inhibitory peptide derived from Moringa oleifera protein, Food Funct., 2021, 12, 8994–9006 RSC.
- Y. Zhao, Q. Zhao and Q. Lu, Purification, structural analysis, and stability of antioxidant peptides from purple wheat bran, BMC Chem., 2020, 14, 58 CrossRef CAS PubMed.
- M. Xie, D. Liu and Y. Yang, Anti-cancer peptides: classification, mechanism of action, reconstruction and modification, Open Biol., 2020, 10, 200004 CrossRef CAS PubMed.
- J. Pei, Z. Liu, D. Pan, Y. Zhao, Y. Dang and X. Gao, Transport, Stability, and In vivo Hypoglycemic Effect of a Broccoli-Derived DPP-IV Inhibitory Peptide VPLVM, J. Agric. Food Chem., 2022, 70, 4934–4941 CrossRef CAS.
- E. B. Okeke, R. O. Abioye, E. Ventura-Santana, X. Sun and C. C. Udenigwe, TNKPVI, a Putative Bioaccessible Pharmacophore of Anti-Inflammatory Potato Patatin-Derived Decapeptide DIKTNKPVIF, Molecules, 2022, 27, 3869 CrossRef CAS PubMed.
- Y. Zhang, Z. Zheng and Y. Liu, Anti-osteoporosis effects and underpinning mechanisms of food-derived bioactive peptides: A review, Trends Food Sci. Technol., 2024, 147, 104431 CrossRef CAS.
- C. Sosalagere, B. A. Kehinde and P. Sharma, Isolation and functionalities of bioactive peptides from fruits and vegetables: A reviews, Food Chem., 2022, 366, 130494 Search PubMed.
- E. B. Daliri, D. H. Oh and B. H. Lee, Bioactive Peptides, Foods, 2017, 6, 32 Search PubMed.
- M. Tu, S. Cheng, W. Lu and M. Du, Advancement and prospects of bioinformatics analysis for studying bioactive peptides from food-derived protein: Sequence, structure, and functions, TrAC, Trends Anal. Chem., 2018, 105, 7–17 CrossRef CAS.
- E. B. Daliri, B. H. Lee and D. H. Oh, Current trends and perspectives of bioactive peptides, Crit. Rev. Food Sci. Nutr., 2018, 58, 2273–2284 CrossRef CAS.
- L. Kang, T. Han, H. Cong, B. Yu and Y. Shen, Recent research progress of biologically active peptides, BioFactors, 2022, 48, 575–596 CrossRef CAS.
- D. C. Whitcomb and M. E. Lowe, Human Pancreatic Digestive Enzymes, Dig. Dis. Sci., 2007, 52, 1–17 CrossRef CAS PubMed.
- Y. Hamuro, S. J. Coales, K. S. Molnar, S. J. Tuske and J. A. Morrow, Specificity of immobilized porcine pepsin in H/D exchange compatible conditions, Rapid Commun. Mass Spectrom., 2008, 22, 1041–1046 CrossRef CAS PubMed.
- S. Y. Lee and S. J. Hur, Antihypertensive peptides from animal products, marine organisms, and plants, Food Chem., 2017, 228, 506–517 CrossRef CAS.
- Y. Xiong, P. Peng, S. Chen, M. Chang, Q. Wang, S. Yin and D. Ren, Preparation, identification, and molecular docking of novel elastase inhibitory peptide from walnut (Juglans regia L.) meal, Food Chem.:Mol. Sci., 2022, 5, 100139 CAS.
- D. J. McClements, Encapsulation, protection, and delivery of bioactive proteins and peptides using nanoparticle and microparticle systems: A review, Adv. Colloid Interface Sci., 2018, 253, 1–22 CrossRef CAS.
- A. R. Mackie, H. Rafiee, P. Malcolm, L. Salt and G. van Aken, Specific food structures supress appetite through reduced gastric emptying rate, Am. J. Physiol.: Gastrointest. Liver Physiol., 2013, 304, G1038–G1043 CrossRef CAS.
- M. Koziolek, F. Schneider, M. Grimm, C. Modeβ, A. Seekamp, T. Roustom, W. Siegmund and W. Weitschies, Intragastric pH and pressure profiles after intake of the high-caloric, high-fat meal as used for food effect studies, J. Controlled Release, 2015, 220, 71–78 CrossRef CAS PubMed.
- L. Amigo and B. Hernandez-Ledesma, Current Evidence on the Bioavailability of Food Bioactive Peptides, Molecules, 2020, 25, 4479 CAS.
- M. Minekus, M. Alminger, P. Alvito, S. Ballance, T. Bohn, C. Bourlieu, F. Carrière, R. Boutrou, M. Corredig, D. Dupont, C. Dufour, L. Egger, M. Golding, S. Karakaya, B. Kirkhus, S. Le Feunteun, U. Lesmes, A. Macierzanka, A. Mackie, S. Marze, D. J. McClements, O. Ménard, I. Recio, C. N. Santos, R. P. Singh, G. E. Vegarud, M. S. J. Wickham, W. Weitschies and A. Brodkorb, A standardised static in vitro digestion method suitable for food - an international consensus, Food Funct., 2014, 5, 1113–1124 CAS.
- A. Brodkorb, L. Egger, M. Alminger, P. Alvito, R. Assunção, S. Ballance, T. Bohn, C. Bourlieu-Lacanal, R. Boutrou, F. Carrière, A. Clemente, M. Corredig, D. Dupont, C. Dufour, C. Edwards, M. Golding, S. Karakaya, B. Kirkhus, S. Le Feunteun, U. Lesmes, A. Macierzanka, A. R. Mackie, C. Martins, S. Marze, D. J. McClements, O. Ménard, M. Minekus, R. Portmann, C. N. Santos, I. Souchon, R. P. Singh, G. E. Vegarud, M. S. J. Wickham, W. Weitschies and I. Recio, INFOGEST static in vitro simulation of gastrointestinal food digestion, Nat. Protoc., 2019, 14, 991–1014 CAS.
- A. Mulet-Cabero, L. Egger, R. Portmann, O. Ménard, S. Marze, M. Minekus, S. Le Feunteun, A. Sarkar, M. M. Grundy, F. Carrière, M. Golding, D. Dupont, I. Recio, A. Brodkorb and A. Mackie, A standardised semi-dynamic in vitro digestion method suitable for food - an international consensus, Food Funct., 2020, 11, 1702–1720 Search PubMed.
- D. Dupont, M. Alric, S. Blanquet-Diot, G. Bornhorst, C. Cueva, A. Deglaire, S. Denis, M. Ferrua, R. Havenaar, J. Lelieveld, A. R. Mackie, M. Marzorati, O. Menard, M. Minekus, B. Miralles, I. Recio and P. Van den Abbeele, Can dynamic in vitro digestion systems mimic the physiological reality?, Crit. Rev. Food Sci. Nutr., 2019, 59, 1546–1562 CAS.
- J. Chen, B. Ryu, Y. Zhang, P. Liang, C. Li, C. Zhou, P. Yang, P. Hong and Z. Qian, Comparison of an angiotensin-I-converting enzyme inhibitory peptide from tilapia (Oreochromis niloticus) with captopril: inhibition kinetics, in vivo effect, simulated gastrointestinal digestion and a molecular docking study, J. Sci. Food Agric., 2020, 100, 315–324 CAS.
- N. Qin, C. Chen, N. Zhang, L. Song, Y. Li, L. Guo, R. Liu and W. Zhang, Bitter Almond Albumin ACE-Inhibitory Peptides: Purification, Screening, and Characterization In Silico, Action Mechanisms, Antihypertensive Effect In vivo, and Stability, Molecules, 2023, 28, 6002 CAS.
- J. Wang, G. Wang, Y. Zhang, R. Zhang and Y. Zhang, Novel Angiotensin-Converting Enzyme Inhibitory Peptides Identified from Walnut Glutelin-1 Hydrolysates: Molecular Interaction, Stability, and Antihypertensive Effects, Nutrients, 2022, 14, 151 CAS.
- W. Gao, M. Liu and Y. Wang, Isolation, virtual screening, action mechanisms, chelation with zinc ions, and stability of ACE-inhibitory peptides from ginkgo seed globulin, RSC Adv., 2023, 13, 30528–30538 CAS.
- L. Zhang, D. Pan, L. Shao, Y. Zheng, W. Hao, Y. Kan, J. Cao, H. Yu and J. Liu, Oil palm kernel globulin antihypertensive peptides: isolation and characterization, ACE inhibition mechanisms, zinc-chelating activity, security and stability, Front. Pharmacol., 2023, 14, 1225256 CAS.
- Y. Zheng, Y. Li, Y. Zhang, X. Ruan and R. Zhang, Purification, characterization, synthesis, in vitro ACE inhibition and in vivo antihypertensive activity of bioactive peptides derived from oil palm kernel glutelin-2 hydrolysates, J. Funct. Foods, 2017, 28, 48–58 CrossRef CAS.
- Y. Li, J. Li, C. Cheng, Y. Zheng, H. Li, Z. Zhu, Y. Yan, W. Hao and N. Qin, Study on the In Silico Screening and Characterization, Inhibition Mechanisms, Zinc-Chelate Activity, and Stability of ACE-Inhibitory Peptides Identified in Naked Oat Bran Albumin Hydrolysates, Foods, 2023, 12, 2268 CrossRef CAS PubMed.
- N. Kang, S. Ko, H. Kim, H. Yang, G. Ahn, S. Lee, T. Lee, J. Lee and Y. Jeon, Structural Evidence for Antihypertensive Effect of an Antioxidant Peptide Purified from the Edible Marine Animal Styela clava, J. Med. Food, 2020, 23, 132–138 CrossRef CAS.
- H. Fan, W. Yu, W. Liao and J. Wu, Spent Hen Protein Hydrolysate with Good Gastrointestinal Stability and Permeability in Caco-2 Cells Shows Antihypertensive Activity in SHR, Foods, 2020, 9, 1384 CrossRef CAS.
- Y. Zhu, C. Chen, Z. Dai, H. Wang, Y. Zhang, Q. Zhao, Y. Xue and Q. Shen, Identification, screening and molecular mechanisms of natural stable angiotensin-converting enzyme (ACE) inhibitory peptides from foxtail millet protein hydrolysates: a combined In Silico and in vitro study, Food Funct., 2024, 15, 7782–7793 RSC.
- Z. Yang, C. Wang, B. Huang, Y. Chen, Z. Liu, H. Chen and J. Chen, Biodirected Screening and Preparation of Larimichthys crocea Angiotensin-I-Converting Enzyme-Inhibitory Peptides by a Combined In vitro and In Silico Approach, Molecules, 2024, 29, 1134 CrossRef CAS PubMed.
- B. Xu, X. Wang, Y. Zheng, Y. Li, M. Guo and Z. Yan, Novel antioxidant peptides identified in millet bran glutelin-2 hydrolysates: Purification, In Silico characterization and security prediction, and stability profiles under different food processing conditions, LWT–Food Sci. Technol., 2022, 164, 113634 CrossRef CAS.
- P. Chan, P. Matanjun, C. Budiman, R. Shapawi and J. Lee, Novel Peptide Sequences with ACE-Inhibitory and Antioxidant Activities Derived from the Heads and Bones of Hybrid Groupers (Epinephelus lanceolatus x Epinephelus fuscoguttatus), Foods, 2022, 11, 3991 CrossRef CAS PubMed.
- J. Tkaczewska, M. Bukowski and P. Mak, Identification of Antioxidant Peptides in Enzymatic Hydrolysates of Carp (Cyprinus Carpio) Skin Gelatin, Molecules, 2019, 24, 97 CrossRef.
- Q. Zhang, L. Zheng, D. Luo and M. Zhao, In vitro Simulated Gastrointestinal Digestion Stability of a Neuroprotective Octapeptide WCPFSRSF and Prediction of Potential Bioactive Peptides in Its Digestive Fragments by Multiple Bioinformatics Tools, J. Agric. Food Chem., 2023, 71, 6987–6998 CrossRef CAS.
- Y. Zheng, Y. Li and G. Li, ACE-inhibitory and antioxidant peptides from coconut cake albumin hydrolysates: purification, identification and synthesis, RSC Adv., 2019, 9, 5925–5936 RSC.
- A. M. Pereira, C. R. Lisboa, T. D. Santos and J. A. V. Costa, Bioactive stability of microalgal protein hydrolysates under food processing and storage conditions, J. Food Sci. Technol., 2019, 56, 4543–4551 CrossRef CAS PubMed.
- X. Zhang, H. He, J. Xiang and T. Hou, Screening and bioavailability evaluation of anti-oxidative selenium-containing peptides from soybeans based on specific structures, Food Funct., 2022, 13, 5252–5261 RSC.
- S. Zhang, L. Han, Y. Shi, X. Li, X. Zhang, H. Hou, H. Lin and K. Liu, Two Novel Multi-Functional Peptides from Meat and Visceral Mass of Marine Snail Neptunea arthritica cumingii and Their Activities In vitro and In vivo, Mar. Drugs, 2018, 16, 473 CrossRef CAS.
- N. Meshginfar, A. S. Mahoonak, F. Hosseinian, M. Ghorbani and A. Tsopmo, Production of antioxidant peptide fractions from a by-product of tomato processing: mass spectrometry identification of peptides and stability to gastrointestinal digestion, J. Food Sci. Technol., 2018, 55, 3498–3507 CrossRef CAS PubMed.
- Y. Hou, X. Chen, M. Zhang, S. Yang, A. Liao, L. Pan, Z. Wang, X. Shen, X. Yuan and J. Huang, Selenium-Chelating Peptide Derived from Wheat Gluten: In vitro Functional Properties, Foods, 2024, 13, 1819 CrossRef CAS PubMed.
- Y. Li, G. Aiello, E. M. A. Fassi, G. Boschin, M. Bartolomei, C. Bollati, G. Roda, A. Arnoldi, G. Grazioso and C. Lammi, Investigation of Chlorella pyrenoidosa Protein as a Source of Novel Angiotensin I-Converting Enzyme (ACE) and Dipeptidyl Peptidase-IV (DPP-IV) Inhibitory Peptides, Nutrients, 2021, 13, 1624 CrossRef CAS PubMed.
- P. A. Harnedy, V. Parthsarathy, C. M. McLaughlin, M. B. O'Keeffe, P. J. Allsopp, E. M. McSorley, F. P. M. O'Harte and R. J. FitzGerald, Atlantic salmon (Salmo salar) co-product-derived protein hydrolysates: A source of antidiabetic peptides, Food Res. Int., 2018, 106, 598–606 CrossRef CAS PubMed.
- A. C. Neves, P. A. Harnedy, M. B. O'Keeffe and R. J. FitzGerald, Bioactive peptides from Atlantic salmon (Salmo salar) with angiotensin converting enzyme and dipeptidyl peptidase IV inhibitory, and antioxidant activities, Food Chem., 2017, 218, 396–405 CrossRef CAS PubMed.
- S. N. Mazloomi, A. S. Mahoonak, L. Mora, M. Ghorbani, G. Houshmand and F. Toldra, Pepsin Hydrolysis of Orange By-Products for the Production of Bioactive Peptides with Gastrointestinal Resistant Properties, Foods, 2021, 10, 679 CrossRef CAS PubMed.
- Y. Li, Y. Fan, J. Liu, Z. Meng, A. Huang, F. Xu and X. Wang, Identification, characterization and in vitro activity of hypoglycemic peptides in whey hydrolysates from rubing cheese by-product, Food Res. Int., 2023, 164, 112382 CrossRef CAS PubMed.
- L. Sorokina, N. T. Solberg, S. Koga, S. B. Ronning, N. K. Afseth, S. R. Wilson, A. Rieder and S. G. Wubshet, In vitro gastrointestinal stability and intestinal absorption of ACE-1 and DPP4 inhibitory peptides from poultry by-product hydrolysates, Food Funct., 2024, 15, 7364–7374 RSC.
- T. D. U. Kröber, M. Holzer, R. Kerpes, V. K. Mittermeier-Kleßinger, C. Dawid and T. Becker, Enrichment and Quantitation of Dipeptidyl Peptidase IV Inhibitory Peptides in Quinoa upon Systematic Malting, J. Agric. Food Chem., 2024, 72, 11480–11492 CrossRef PubMed.
- L. Xing, L. Fu, F. Toldra, S. Teng, Y. Yin and W. Zhang, The stability of dry-cured ham-derived peptides and its anti-inflammatory effect in RAW264.7 macrophage cells, Int. J. Food Sci. Technol., 2023, 58, 1575–1585 CrossRef CAS.
- R. Zan, L. Zhu, G. Wu and H. Zhang, Identification of Novel Peptides with Alcohol Dehydrogenase (ADH) Activating Ability in Chickpea Protein Hydrolysates, Foods, 2023, 12, 1574 CrossRef CAS PubMed.
- C. Offret, I. Fliss, L. Bazinet, A. Marette and L. Beaulieu, Identification of A Novel Antibacterial Peptide from Atlantic Mackerel belonging to the GAPDH-Related Antimicrobial Family and Its In vitro Digestibility, Mar. Drugs, 2019, 17, 413 CrossRef CAS PubMed.
- W. Nie, F. Xu, K. Zhou, J. Deng, Y. Wang and B. Xu, Stability and transepithelial transport of oligopeptide (KRQKYD) with hepatocyte-protective activity from Jinhua ham in human intestinal Caco-2 monolayer cells, Food Sci. Hum. Wellness, 2024, 13, 1503–1512 CrossRef CAS.
- B. Hernández-Ledesma, M. d. M. Contreras and I. Recio, Antihypertensive peptides: Production, bioavailability and incorporation into foods, Adv. Colloid Interface Sci., 2011, 165, 23–35 CrossRef PubMed.
- S. M. Martínez-Sánchez, J. A. Gabaldón-Hernández and S. Montoro-García, Unravelling the molecular mechanisms associated with the role of food-derived bioactive peptides in promoting cardiovascular health, J. Funct. Foods, 2020, 64, 103645 CrossRef.
- F. J. Warner, A. I. Smith, N. M. Hooper and A. J. Turner, Angiotensin-converting enzyme-2: a molecular and cellular perspective, Cell. Mol. Life Sci., 2004, 61, 2704–2713 CrossRef CAS PubMed.
- A. Dhaval, N. Yadav and S. Purwar, Potential Applications of Food Derived Bioactive Peptides in Management of Health, Int. J. Pept. Res. Ther., 2016, 22, 377–398 CrossRef CAS.
- R. E. Aluko, Antihypertensive Peptides from Food Proteins, Annu. Rev. Food Sci. Technol., 2015, 6, 235–262 CrossRef CAS PubMed.
- B. A. Murray and R. J. FitzGerald, Angiotensin converting enzyme inhibitory peptides derived from food proteins: biochemistry, bioactivity and production, Curr. Pharm. Des., 2007, 13, 773–791 CrossRef CAS PubMed.
- C. Jao, S. Huang and K. Hsu, Angiotensin I-converting enzyme inhibitory peptides: Inhibition mode, bioavailability, and antihypertensive effects, Biomedicine, 2012, 2, 130–136 CrossRef.
- J. Zhang, L. Liang, Y. Shan, X. Zhou, B. Sun, Y. Liu and Y. Zhang, Antihypertensive Effect, ACE Inhibitory Activity, and Stability of Umami Peptides from Yeast Extract, J. Agric. Food Chem., 2023, 71, 17263–17272 CAS.
- R. Li, Y. Zhuang, L. Lin, L. Li, X. Fan and L. Sun, In vitro simulated gastrointestinal digestion stability and in vivo antihypertensive effect of the peptide KYPHVF and its network pharmacology, J. Funct. Foods, 2023, 107, 105672 CAS.
- S. Sangiorgio, N. Vidovic, G. Boschin, G. Aiello, P. Arcidiaco, A. Arnoldi, C. F. Morelli, M. Rabuffetti, T. Recca, L. Scarabattoli, D. Ubiali and G. Speranza, Preparation, Characterization and In vitro Stability of a Novel ACE-Inhibitory Peptide from Soybean Protein, Foods, 2022, 11, 2667 CAS.
- P. Garcia-Mora, M. Martin-Martinez, M. A. Bonache, R. Gonzalez-Muniz, E. Penas, J. Frias and C. Martinez-Villaluenga, Identification, functional gastrointestinal stability and molecular docking studies of lentil peptides with dual antioxidant and angiotensin I converting enzyme inhibitory activities, Food Chem., 2017, 221, 464–472 CAS.
- H. Fan, K. Wu and J. Wu, Pea-derived tripeptide LRW fails to reduce blood pressure in spontaneously hypertensive rats due to its low gastrointestinal stability and transepithelial permeability, Food Biosci., 2022, 49, 101964 CrossRef CAS.
- M. Miguel, M. A. Aleixandre, M. Ramos and R. López-Fandino, Effect of simulated gastrointestinal digestion on the antihypertensive properties of ACE-inhibitory peptides derived from ovalbumin, J. Agric. Food Chem., 2006, 54, 726–731 CrossRef CAS PubMed.
- T. Tavares, M. D. M. Contreras, M. Amorim, M. Pintado, I. Recio and F. X. Malcata, Novel whey-derived peptides with inhibitory effect against angiotensin-converting enzyme: In vitro effect and stability to gastrointestinal enzymes, Peptides, 2011, 32, 1013–1019 CrossRef CAS PubMed.
- Y. Jiang, J. Sun, J. Chandrapala, M. Majzoobi, C. Brennan, X. A. Zeng and B. Sun, Recent progress of food–derived bioactive peptides: Extraction, purification, function, and encapsulation, Food Front., 2024, 5, 1240–1264 CrossRef CAS.
- M. Chen, P. Ning, Y. Jiao, Z. Xu and Y. Cheng, Extraction of antioxidant peptides from rice dreg protein hydrolysate via an angling method, Food Chem., 2021, 337, 128069 CrossRef CAS PubMed.
- J. T. Ryan, R. P. Ross, D. Bolton, G. F. Fitzgerald and C. Stanton, Bioactive Peptides from Muscle Sources: Meat and Fish, Nutrients, 2011, 3, 765–791 CrossRef CAS PubMed.
- M. Wang, X. Sun, W. Luo, S. Božović, C. Gong and J. Ren, Characterization and analysis of antioxidant activity of walnut-derived pentapeptide PW5 via nuclear magnetic resonance spectroscopy, Food Chem., 2021, 339, 128047 CrossRef CAS PubMed.
- Y. Jiang, R. Wang, Z. Yin, J. Sun, B. Wang, D. Zhao, X. Zeng, H. Li, M. Huang and B. Sun, Optimization of Jiuzao protein hydrolysis conditions and antioxidant activity in vivo of Jiuzao tetrapeptide Asp-Arg-Glu-Leu by elevating the Nrf2/Keap1-p38/PI3K-MafK signaling pathway, Food Funct., 2021, 12, 4808–4824 RSC.
- L. Ren, Y. Yang, C. Ma, J. Fan, X. Bian, B. Liu, D. Wang, P. Zhu, Y. Fu and N. Zhang, Identification and In Silico analysis of novel antioxidant peptides in broken rice protein hydrolysate and its cytoprotective effect against H2O2-induced 2BS cell model, Food Res. Int., 2022, 162, 112108 CrossRef CAS PubMed.
- C. G. Rizzello, D. Tagliazucchi, E. Babini, G. S. Rutella, D. L. T. Saa and A. Gianotti, Bioactive peptides from vegetable food matrices: Research trends and novel biotechnologies for synthesis and recovery, J. Funct. Foods, 2016, 27, 549–569 CrossRef CAS.
- E. Maestri, M. Marmiroli and N. Marmiroli, Bioactive peptides in plant-derived foodstuffs, J. Proteomics, 2016, 147, 140–155 CrossRef CAS.
- C. C. Udenigwe and R. E. Aluko, Food Protein–Derived Bioactive Peptides: Production, Processing, and Potential Health Benefits, J. Food Sci., 2012, 77, R11–R24 CrossRef CAS PubMed.
- W. Cai, X. Hu, Y. Wang, C. Chi and B. Wang, Bioactive Peptides from Skipjack Tuna Cardiac Arterial Bulbs: Preparation, Identification, Antioxidant Activity, and Stability against Thermal, pH, and Simulated Gastrointestinal Digestion Treatments, Mar. Drugs, 2022, 20, 626 CrossRef CAS PubMed.
- L. Chen, Y. Hui, T. Gao, G. Shu and H. Chen, Function and characterization of novel antioxidant peptides by fermentation with a wild Lactobacillus plantarum 60, LWT–Food Sci. Technol., 2021, 135, 110162 CrossRef CAS.
- J. Zhan, G. Li, Y. Dang and D. Pan, Antioxidant stability of a novel peptide from porcine plasma hydrolysates by in vitro digestion/HepG-2 model, Eur. Food Res. Technol., 2022, 248, 797–806 CrossRef CAS.
- C. Xiao, F. Toldra, M. Zhao, F. Zhou, D. Luo, R. Jia and L. Mora, In vitro and In Silico analysis of potential antioxidant peptides obtained from chicken hydrolysate produced using Alcalase, Food Res. Int., 2022, 157, 111253 CrossRef CAS PubMed.
- M. Gallego, L. Mora, M. Reig and F. Toldra, Stability of the potent antioxidant peptide SNAAC identified from Spanish dry-cured ham, Food Res. Int., 2018, 105, 873–879 CrossRef CAS PubMed.
- M. Gallego, L. Mora and F. Toldra, Characterisation of the antioxidant peptide AEEEYPDL and its quantification in Spanish dry-cured ham, Food Chem., 2018, 258, 8–15 CrossRef CAS.
- X. Yang, Y. Qiu, Y. Zhao, C. Chi and B. Wang, Purification and Characterization of Antioxidant Peptides Derived from Protein Hydrolysate of the Marine Bivalve Mollusk Tergillarca granosa, Mar. Drugs, 2019, 17, 251 CrossRef CAS.
- M. A. Guney, D. S. Lorberbaum and L. Sussel, Pancreatic β cell regeneration: to β or not to β, Curr. Opin. Physiol., 2020, 14, 13–20 CrossRef PubMed.
- C. Jao, C. Hung, Y. Tung, P. Lin, M. Chen and K. Hsu, The development of bioactive peptides from dietary proteins as a dipeptidyl peptidase IV inhibitor for the management of type 2 diabetes, Biomedicine, 2015, 5, 14 Search PubMed.
- N. Li, L. Wang, B. Jiang, X. Li, C. Guo, S. Guo and D. Shi, Recent progress of the development of dipeptidyl peptidase-4 inhibitors for the treatment of type 2 diabetes mellitus, Eur. J. Med. Chem., 2018, 151, 145–157 Search PubMed.
- B. Ahrén, DPP-4 inhibitors, Best Pract. Res., Clin. Endocrinol. Metab., 2007, 21, 517–533 CrossRef PubMed.
- M. Zhang, L. Zhu, G. Wu, T. Liu, X. Qi and H. Zhang, Food-derived dipeptidyl peptidase IV inhibitory peptides: Production, identification, structure-activity relationship, and their potential role in glycemic regulation, Crit. Rev. Food Sci. Nutr., 2022, 64, 2053–2075 CrossRef PubMed.
- I. M. E. Lacroix and E. C. Y. Li-Chan, Food-derived dipeptidyl-peptidase IV inhibitors as a potential approach for glycemic regulation – Current knowledge and future research considerations, Trends Food Sci. Technol., 2016, 54, 1–16 Search PubMed.
- A. Brandelli, D. J. Daroit and A. P. F. Corrêa, Whey as a source of peptides with remarkable biological activities, Food Res. Int., 2015, 73, 149–161 CrossRef CAS.
- B. Wang, Z. Yu, W. Yokoyama, B. Chiou, M. Chen, F. Liu and F. Zhong, Collagen peptides with DPP-IV inhibitory activity from sheep skin and their stability to in vitro gastrointestinal digestion, Food Biosci., 2021, 42, 101161 CrossRef CAS.
- P. A. Harnedy-Rothwell, N. Khatib, S. Sharkey, R. A. Lafferty, S. Gite, J. Whooley, F. P. M. O'Harte and R. J. FitzGerald, Physicochemical, Nutritional and In vitro Antidiabetic Characterisation of Blue Whiting (Micromesistius poutassou) Protein Hydrolysates, Mar. Drugs, 2021, 19, 383 Search PubMed.
- M. A. M. Mune, S. R. Minka and T. Henle, Investigation on antioxidant, angiotensin converting enzyme and dipeptidyl peptidase IV inhibitory activity of Bambara bean protein hydrolysates, Food Chem., 2018, 250, 162–169 Search PubMed.
- R. Pérez-Gálvez, C. Berraquero-García, J. L. Ospina-Quiroga, F. J. Espejo-Carpio, M. C. Almécija, A. Guadix, P. J. García-Moreno and E. Guadix, Influence of In Vitro Digestion on Dipeptidyl Peptidase-IV (DPP-IV) Inhibitory Activity of Plant-Protein Hydrolysates Obtained from Agro-Industrial By-Products, Foods, 2024, 13, 2691 Search PubMed.
- M. A. Rendon-Rosales, M. J. Torres-Llanez, M. A. Mazorra-Manzano, A. F. Gonzalez-Cordova, A. Hernandez-Mendoza and B. Vallejo-Cordoba, In vitro and In Silico evaluation of multifunctional properties of bioactive synthetic peptides identified in milk fermented with Lactococcus lactis NRRL B-50571 and NRRL B-50572, LWT–Food Sci. Technol., 2022, 154, 112581 CAS.
- J. J. Song, Q. Wang, M. Du, X. M. Ji and X. Y. Mao, Identification of dipeptidyl peptidase-IV inhibitory peptides from mare whey protein hydrolysates, J. Dairy Sci., 2017, 100, 6885–6894 CAS.
- Q. Zhu, X. Chen, J. Wu, Y. Zhou, Y. Qian, M. Fang, J. Xie and D. Wei, Dipeptidyl peptidase IV inhibitory peptides from Chlorella vulgaris: In Silico gastrointestinal hydrolysis and molecular mechanism, Eur. Food Res. Technol., 2017, 243, 1739–1748 CAS.
- A. C. Buzaid and M. Atkins, Practical guidelines for the management of biochemotherapy-related toxicity in melanoma, Clin. Cancer Res., 2001, 7, 2611–2619 CAS.
- M. Ortiz-Martinez, R. Winkler and S. García-Lara, Preventive and therapeutic potential of peptides from cereals against cancer, J. Proteomics, 2014, 111, 165–183 CAS.
- E. G. de Mejia and V. P. Dia, The role of nutraceutical proteins and peptides in apoptosis, angiogenesis, and metastasis of cancer cells, Cancer Metastasis Rev., 2010, 29, 511–528 Search PubMed.
- B. N. P. Sah, T. Vasiljevic, S. McKechnie and O. N. Donkor, Antioxidant peptides isolated from synbiotic yoghurt exhibit antiproliferative activities against HT-29 colon cancer cells, Int. Dairy J., 2016, 63, 99–106 CrossRef CAS.
- R. Nurdiani, T. Vasiljevic, T. K. Singh, O. N. Donkor, A. A. Prihanto and T. S. Kusuma, Stability of an anticancer peptide isolated from Flathead by-products during in vitro gastrointestinal digestion, Funct. Foods Health Dis., 2022, 12, 198–207 CrossRef CAS.
- Z. Xue, H. Wen, C. Wang, L. Zhai, A. Cheng and X. Kou, CPe-III-S Metabolism in vitro and in vivo and Molecular Simulation of Its Metabolites Using a p53-R273H Mutant, J. Agric. Food Chem., 2016, 64, 7095–7103 CrossRef CAS PubMed.
- H. Li, H. Fan, Z. Wang, Q. Zhu and J. Wu, Effect of simulated gastrointestinal digestion on antioxidant, and anti-inflammatory activities of bioactive peptides generated in sausages fermented with Staphylococcus simulans QB7, Food Sci. Hum. Wellness, 2024, 13, 1662–1671 CrossRef CAS.
- Y. Du, S. Zhu, R. Wang, X. Chen and K. Cai, Isolation and Identification of Anti-Inflammatory Peptide from Goose Blood Hydrolysate to Ameliorate LPS-Mediated Inflammation and Oxidative Stress in RAW264.7 Macrophages, Molecules, 2022, 27, 8816 CrossRef CAS PubMed.
- J. Wu, Q. Li, C. Cui and J. Xu, Screening of novel bovine-elastin-derived peptides with elastase inhibition and photoprotective potential: a combined In Silico and in vitro study, J. Sci. Food Agric., 2023, 104, 716–726 CrossRef PubMed.
- X. Yang, D. Wang, Y. Dai, L. Zhao, W. Wang and X. Ding, Identification and Molecular Binding Mechanism of Novel α-Glucosidase Inhibitory Peptides from Hot-Pressed Peanut Meal Protein Hydrolysates, Foods, 2023, 12, 663 CrossRef CAS PubMed.
- C. Xiao, F. Zhou, L. Zheng, Y. Cai, G. Su, D. Luo and M. Zhao, Chicken breast-derived alcohol dehydrogenase-activating peptides in response to physicochemical changes and digestion simulation: The vital role of hydrophobicity, Food Res. Int., 2020, 136, 109592 CrossRef CAS PubMed.
- M. Liu, F. Liu, T. Zhang, X. Zhao, X. Wang, W. Kong, L. Cui, H. Luo, L. Guo and Y. Guo, Transportation of whey protein-derived peptides using Caco-2 cell model and identification of novel cholesterol-lowering peptides, Food Nutr. Res., 2023, 67, 9079 Search PubMed.
- D. P. Baptista, M. K. Salgaço, K. Sivieri and M. L. Gigante, Use of static and dynamic in vitro models to simulate Prato cheese gastrointestinal digestion: Effect of Lactobacillus helveticus LH-B02 addition on peptides bioaccessibility, LWT–Food Sci. Technol., 2020, 134, 110229 CrossRef CAS.
- S. M. Vivanco-Maroto, V. Gallo, B. Miralles and I. Recio, CCK and GLP-1 response on enteroendocrine cells of semi-dynamic digests of hydrolyzed and intact casein, Food Res. Int., 2023, 171, 113047 CrossRef CAS PubMed.
- R. Wang, X. Lu, Q. Sun, J. Gao, L. Ma and J. Huang, Novel ACE Inhibitory Peptides Derived from Simulated Gastrointestinal Digestion in vitro of Sesame (Sesamum indicum L.) Protein and Molecular Docking Study, Int. J. Mol. Sci., 2020, 21, 1059 CrossRef CAS PubMed.
- J. Zhang, H. Du, G. Zhang, F. Kong, Y. Hu, S. Xiong and S. Zhao, Identification and characterization of novel antioxidant peptides from crucian carp (Carassius auratus) cooking juice released in simulated gastrointestinal digestion by UPLC-MS/MS and in silico analysis, J. Chromatogr. B:Anal. Technol. Biomed. Life Sci., 2020, 1136, 121893 CrossRef CAS PubMed.
- S. E. Suárez, H. Rabesona, O. Ménard, J. Jardin, M. Anton and M. C. Añón, Dynamic digestion of a high protein beverage based on amaranth: Structural changes and antihypertensive activity, Food Res. Int., 2024, 187, 114416 CrossRef PubMed.
- X. Wu, H. Liu, J. Han, Z. Zhou, J. Chen and X. Liu, Introducing Bacillus natto and Propionibacterium shermanii into soymilk fermentation: A promising strategy for quality improvement and bioactive peptide production during in vitro digestion, Food Chem., 2024, 455, 139585 CrossRef CAS PubMed.
- J. Li, H. Hu, F. Chen, C. Yang, W. Yang, Y. Pan, X. Yu and Q. He, Characterization, mechanisms, structure-activity relationships, and antihypertensive effects of ACE inhibitory peptides: rapid screening from sufu hydrolysate, Food Funct., 2024, 15, 9224–9234 RSC.
- T. Ahmed, X. Sun and C. C. Udenigwe, Role of structural properties of bioactive peptides in their stability during simulated gastrointestinal digestion: A systematic review, Trends Food Sci. Technol., 2022, 120, 265–273 CrossRef CAS.
- O. Selamassakul, N. Kaisangsri, C. Sonklin, R. Kaprasob, A. Uthairatanakij and N. Laohakunjit, Effects of cluster dextrin encapsulation on the properties and antioxidant stability of fractionated Riceberry protein hydrolysate powder prepared by bromelain, Food Chem., 2024, 439, 138161 CrossRef CAS PubMed.
- M. R. Ferreira, A. G. Garzon, M. E. Oliva, R. E. Cian, S. R. Drago and M. E. D'Alessandro, Lipid-lowering effect of microencapsulated peptides from brewer's spent grain in high-sucrose diet-fed rats, Food Biosci., 2022, 49, 101981 CrossRef CAS.
- R. E. Cian, P. R. Salgado, A. N. Mauri and S. R. Drago, Pyropia columbina phycocolloids as microencapsulating material improve bioaccessibility of brewers’ spent grain peptides with ACE-I inhibitory activity, Int. J. Food Sci. Technol., 2020, 55, 1311–1317 CrossRef CAS.
- L. Wei, D. Wong, T. Jeoh and M. L. Marco, Intestinal delivery of encapsulated bacteriocin peptides in cross-linked alginate microcapsules, Food Res. Int., 2024, 188, 114473 CAS.
- I. Silva, B. M. C. Vaz, S. Sousa, M. M. Pintado, E. R. Coscueta and S. P. M. Ventura, Gastrointestinal delivery of codfish Skin-Derived collagen Hydrolysates: Deep eutectic solvent extraction and bioactivity analysis, Food Res. Int., 2024, 175, 113729 CAS.
- L. Zou, Z. Zhang, R. Zhang, W. Liu, C. Liu, H. Xiao and D. J. McClements, Encapsulation of protein nanoparticles within alginate microparticles: Impact of pH and ionic strength on functional performance, J. Food Eng., 2016, 178, 81–89 CrossRef CAS.
- S. Suárez and M. C. Añón, Amaranth proteins emulsions as delivery system of Angiotensin-I converting enzyme inhibitory peptides, Food Hydrocolloids, 2019, 90, 154–161 Search PubMed.
- Y. Jo, H. P. Karbstein and U. S. van der Schaaf, Collagen peptide-loaded W1/O single emulsions and W1/O/W2 double emulsions: influence of collagen peptide and salt concentration, dispersed phase fraction and type of hydrophilic emulsifier on droplet stability and encapsulation efficiency, Food Funct., 2019, 10, 3312–3323 RSC.
- Z. Niu, I. Thielen, S. M. Loveday and H. Singh, Emulsions Stabilised by Polyethylene Glycol (PEG) 40 Stearate and Lactoferrin for Protection of Lactoferrin during In vitro Digestion, Food Biophys., 2021, 16, 40–47 CrossRef.
- L. Ramezanzade, S. F. Hosseini, B. Akbari-Adergani and A. Yaghmur, Cross-linked chitosan-coated liposomes for encapsulation of fish-derived peptide, LWT–Food Sci. Technol., 2021, 150, 112057 CrossRef CAS.
- D. Marin, A. Aleman, A. Sanchez-Faure, P. Montero and M. C. Gomez-Guillen, Freeze-dried phosphatidylcholine liposomes encapsulating various antioxidant extracts from natural waste as functional ingredients in surimi gels, Food Chem., 2018, 245, 525–535 CrossRef CAS PubMed.
- K. Stephansen, M. Garcia-Diaz, F. Jessen, I. S. Chronakis and H. M. Nielsen, Bioactive protein-based nanofibers interact with intestinal biological components resulting in transepithelial permeation of a therapeutic protein, Int. J. Pharm., 2015, 495, 58–66 CrossRef CAS PubMed.
- S. F. Hosseini, Z. Nahvi and M. Zandi, Antioxidant peptide-loaded electrospun chitosan/poly (vinyl alcohol) nanofibrous mat intended for food biopackaging purposes, Food Hydrocolloids, 2019, 89, 637–648 CrossRef CAS.
- T. A. Comunian, S. Drusch and A. Brodkorb, Advances of plant-based structured food delivery systems on the in vitro digestibility of bioactive compounds, Crit. Rev. Food Sci. Nutr., 2022, 62, 6485–6504 CrossRef CAS PubMed.
- G. Feltre, F. S. Almeida, A. C. K. Sato, G. C. Dacanal and M. D. Hubinger, Alginate and corn starch mixed gels: Effect of gelatinization and amylose content on the properties and in vitro digestibility, Food Res. Int., 2020, 132, 109069 CrossRef CAS PubMed.
- A. Mulet-Cabero, A. Torcello-Gómez, S. Saha, A. R. Mackie, P. J. Wilde and A. Brodkorb, Impact of caseins and whey proteins ratio and lipid content on in vitro digestion and ex vivo absorption, Food Chem., 2020, 319, 126514 CrossRef CAS PubMed.
- O. Norkaew, P. Thitisut, S. Mahatheeranont, B. Pawin, P. Sookwong, S. Yodpitak and A. Lungkaphin, Effect of wall materials on some physicochemical properties and release characteristics of encapsulated black rice anthocyanin microcapsules, Food Chem., 2019, 294, 493–502 CrossRef CAS PubMed.
- J. Calvo-Lerma, A. Asensio-Grau, A. Heredia and A. Andrés, Lessons learnt from MyCyFAPP Project: Effect of cystic fibrosis factors and inherent-to-food properties on lipid digestion in foods, Food Res. Int., 2020, 133, 109198 CrossRef CAS PubMed.
- Y. Tan, Z. Zhang, J. M. Mundo and D. J. McClements, Factors impacting lipid digestion and nutraceutical bioaccessibility assessed by standardized gastrointestinal model (INFOGEST): Emulsifier type, Food Res. Int., 2020, 137, 109739 CrossRef CAS PubMed.
- F. Paulo and L. Santos, Design of experiments for microencapsulation applications: A review, Mater. Sci. Eng., C, 2017, 77, 1327–1340 CrossRef CAS PubMed.
- H. R. Sharif, P. A. Williams, M. K. Sharif, S. Abbas, H. Majeed, K. G. Masamba, W. Safdar and F. Zhong, Current progress in the utilization of native and modified legume proteins as emulsifiers and encapsulants – A review, Food Hydrocolloids, 2018, 76, 2–16 CrossRef CAS.
- C. Anandharamakrishnan, C. D. Rielly and A. G. F. Stapley, Spray-freeze-drying of whey proteins at sub-atmospheric pressures., Dairy Sci. Technol., 2010, 90, 321–334 CrossRef CAS.
- D. Ma, B. Yang, J. Zhao, D. Yuan and Q. Li, Advances in protein-based microcapsules and their applications: A review, Int. J. Biol. Macromol., 2024, 263, 129742 CrossRef CAS PubMed.
- C. Wen, X. Lin, J. Tang, M. Fan, G. Liu, J. Zhang and X. Xu, New perspective on protein-based microcapsules as delivery vehicles for sensitive substances: A review, Int. J. Biol. Macromol., 2024, 270, 132449 CrossRef CAS PubMed.
- M. Malmsten, Soft drug delivery systems, Soft Matter, 2006, 2, 760–769 RSC.
- A. P. F. Corrêa, F. F. Veras, C. C. Lago, C. P. Z. Noreña and A. Brandelli, Microencapsulation upholds biological activities of sheep whey hydrolysates and protects against in vitro gastrointestinal digestion, Int. Dairy J., 2023, 138, 105554 CrossRef.
- R. E. Cian, M. E. Oliva, A. G. Garzon, M. R. Ferreira, M. E. D’Alessandro and S. R. Drago, In vitro and in vivo antithrombotic and antioxidant properties of microencapsulated brewers’ spent grain peptides, Int. J. Food Sci. Technol., 2022, 57, 3872–3879 CrossRef CAS.
- R. E. Cian, A. Campos-Soldini, L. Chel-Guerrero, S. R. Drago and D. Betancur-Ancona, Bioactive Phaseolus lunatus peptides release from maltodextrin/gum arabic microcapsules obtained by spray drying after simulated gastrointestinal digestion, Int. J. Food Sci. Technol., 2019, 54, 2002–2009 CrossRef CAS.
- X. Xun, Z. Zhang, Z. Yuan, K. Tuhong, C. Yan, Y. Zhan, G. Kang, Q. Wu and J. Wang, Novel caffeic acid grafted chitosan-sodium alginate microcapsules generated by microfluidic technique for the encapsulation of bioactive peptides from silkworm pupae, Sustainable Chem. Pharm., 2023, 32, 100974 CrossRef CAS.
- C. Galves, A. K. Stone, J. Szarko, S. Liu, K. Shafer, J. Hargreaves, M. Siarkowski and M. T. Nickerson, Effect of pH and defatting on the functional attributes of safflower, sunflower, canola, and hemp protein concentrates, Cereal Chem., 2019, 96, 1036–1047 CrossRef CAS.
- C. Galves, G. Galli and L. Kurozawa, Potato protein: current review of structure, technological properties, and potential application on spray drying microencapsulation, Crit. Rev. Food Sci. Nutr., 2023, 63, 6564–6579 CrossRef CAS PubMed.
- J. Pei, X. Gao, D. Pan, Y. Hua, J. He, Z. Liu and Y. Dang, Advances in the stability challenges of bioactive peptides and improvement strategies, Curr. Res. Food Sci., 2022, 5, 2162–2170 CrossRef CAS PubMed.
- A. Matalanis, O. G. Jones and D. J. McClements, Structured biopolymer-based delivery systems for encapsulation, protection, and release of lipophilic compounds, Food Hydrocolloids, 2011, 25, 1865–1880 CrossRef CAS.
- O. G. Jones and D. J. McClements, Functional Biopolymer Particles: Design, Fabrication, and Applications, Compr. Rev. Food Sci. Food Saf., 2010, 9, 374–397 CrossRef CAS PubMed.
- S. L. Perry and D. J. McClements, Recent Advances in Encapsulation, Protection, and Oral Delivery of Bioactive Proteins and Peptides using Colloidal Systems, Molecules, 2020, 25, 1161 CrossRef CAS PubMed.
- D. J. McClements, Recent progress in hydrogel delivery systems for improving nutraceutical bioavailability, Food Hydrocolloids, 2017, 68, 238–245 CrossRef CAS.
- S. Free-Manjarrez, L. Mojica, H. Espinosa-Andrews and N. Morales-Hernandez, Sensory and Biological Potential of Encapsulated Common Bean Protein Hydrolysates Incorporated in a Greek-Style Yogurt Matrix, Polymers, 2022, 14, 854 CrossRef CAS PubMed.
- S. Obuobi, V. Mayandi, N. A. M. Nor, B. J. Lee, R. Lakshminarayanan and P. L. R. Ee, Nucleic acid peptide nanogels for the treatment of bacterial keratitis, Nanoscale, 2020, 12, 17411–17425 RSC.
- B. Maherani, E. Arab-Tehrany, M. R. Mozafari, C. Gaiani and M. Linder, Liposomes: A Review of Manufacturing Techniques and Targeting Strategies, Curr. Nanosci., 2011, 7, 436–452 CrossRef CAS.
- R. R. Sawant and V. P. Torchilin, Liposomes as ‘smart’ pharmaceutical nanocarriers, Soft Matter, 2010, 6, 426–444 RSC.
- N. V. Pavlovic, J. R. Jovanovic, V. B. Dordevic, B. D. Balanc, B. M. Bugarski and Z. D. Knezevic-Jugovic, Production and characterization of liposomes with encapsulated bioactive soy protein hydrolysate, Hem. Ind., 2020, 74, 327–339 CrossRef.
- D. Zhu, W. Ma, M. Yang, S. Cheng, L. Zhang and M. Du, Protection of osteogenic peptides in nanoliposomes: Stability, sustained release, bioaccessibility and influence on bioactive properties, Food Chem., 2024, 436, 137683 CrossRef CAS PubMed.
- B. S. Esposto, P. Jauregi, D. R. Tapia-Blácido and M. Martelli-Tosi, Liposomes vs. chitosomes: Encapsulating food bioactives, Trends Food Sci. Technol., 2021, 108, 40–48 CrossRef CAS.
- R. Huang, H. Song, X. Wang, H. Shen, S. Li and X. Guan, Fatty acids-modified liposomes for encapsulation of bioactive peptides: Fabrication, characterization, storage stability and in vitro release., Food Chem., 2023, 440, 138139 CrossRef PubMed.
- M. Niu, Y. N. Tan, P. Guan, L. Hovgaard, Y. Lu, J. Qi, R. Lian, X. Li and W. Wu, Enhanced oral absorption of insulin-loaded liposomes containing bile salts: A mechanistic study, Int. J. Pharm., 2014, 460, 119–130 CrossRef CAS PubMed.
- D. J. McClements, Food emulsions principles, practices, and techniques, CRC Press, Boca Raton, FL, USA, 3rd edn, 2015 Search PubMed.
- D. J. McClements, Nanoemulsions versus microemulsions: terminology, differences, and similarities, Soft Matter, 2012, 8, 1719–1729 RSC.
- D. J. McClements and C. E. Gumus, Natural emulsifiers—Biosurfactants, phospholipids, biopolymers, and colloidal particles: Molecular and physicochemical basis of functional performance, Adv. Colloid Interface Sci., 2016, 234, 3–26 CrossRef CAS PubMed.
- D. J. McClements, Design of Nano–Laminated Coatings to Control Bioavailability of Lipophilic Food Components, J. Food Sci., 2010, 75, R30–R42 CrossRef CAS PubMed.
- N. Anton, J. Benoit and P. Saulnier, Design and production of nanoparticles formulated from nano-emulsion templates—A review, J. Controlled Release, 2008, 128, 185–199 CrossRef CAS PubMed.
- Y. Gao, X. Wu, D. J. McClements, C. Cheng, Y. Xie, R. Liang, J. Liu, L. Zou and W. Liu, Encapsulation of bitter peptides in water-in-oil high internal phase emulsions reduces their bitterness and improves gastrointestinal stability, Food Chem., 2022, 386, 132787 CrossRef CAS PubMed.
- G. Muschiolik and E. Dickinson, Double Emulsions Relevant to Food Systems: Preparation, Stability, and Applications, Compr. Rev. Food Sci. Food Saf., 2017, 16, 532–555 CrossRef CAS PubMed.
- H. J. Giroux, G. Robitaille and M. Britten, Controlled release of casein-derived peptides in the gastrointestinal environment by encapsulation in water-in-oil-in-water double emulsions, LWT–Food Sci. Technol., 2016, 69, 225–232 CrossRef CAS.
- P. Wen, M. Zong, R. J. Linhardt, K. Feng and H. Wu, Electrospinning: A novel nano-encapsulation approach for bioactive compounds, Trends Food Sci. Technol., 2017, 70, 56–68 CrossRef CAS.
- B. Ghorani and N. Tucker, Fundamentals of electrospinning as a novel delivery vehicle for bioactive compounds in food nanotechnology, Food Hydrocolloids, 2015, 51, 227–240 CrossRef CAS.
- D. Rajanna, H. A. Pushpadass, F. M. E. Emerald, N. V. Padaki and B. S. Nath, Nanoencapsulation of casein–derived peptides within electrospun nanofibres, J. Sci. Food Agric., 2022, 102, 1684–1698 CrossRef PubMed.
- Z. Kirbas and F. Altay, Incorporating antioxidative peptides within nanofibrous delivery vehicles: Characterization and in vitro release kinetics, Food Biosci., 2023, 53, 102660 CrossRef CAS.
- Q. Ke, K. Ma, Y. Zhang, Q. Meng, X. Huang and X. Kou, Antibacterial aroma compounds as property modifiers for electrospun biopolymer nanofibers of proteins and polysaccharides: A review, Int. J. Biol. Macromol., 2023, 253, 126563 CrossRef CAS PubMed.
- X. Lai, J. Tang and M. ElSayed, Recent advances in proteolytic stability for peptide, protein, and antibody drug discovery, Expert Opin. Drug Discovery, 2021, 16, 1467–1482 CrossRef CAS PubMed.
- H. Li, K. Zhu, H. Zhou, W. Peng and X. Guo, Comparative study of four physical approaches about allergenicity of soybean protein isolate for infant formula, Food Agric. Immunol., 2016, 27, 604–623 CrossRef CAS.
- B. P. Hurley, W. Pirzai, A. D. Eaton, M. Harper, J. Roper, C. Zimmermann, G. S. Ladics, R. J. Layton and B. Delaney, An experimental platform using human intestinal epithelial cell lines to differentiate between hazardous and non-hazardous proteins, Food Chem. Toxicol., 2016, 92, 75–87 CrossRef CAS PubMed.
- A. Görgüç, E. Gençdağ and F. M. Yılmaz, Bioactive peptides derived from plant origin by-products: Biological activities and techno-functional utilizations in food developments – A review, Food Res. Int., 2020, 136, 109504 CrossRef PubMed.
- M. Martin, D. Hagemann, T. T. Nguyen, L. Schwarz, S. Khedr, M. L. Moskopp, T. Henle and A. Deussen, Plasma concentrations and ACE-inhibitory effects of tryptophan-containing peptides from whey protein hydrolysate in healthy volunteers, Eur. J. Nutr., 2020, 59, 1135–1147 CrossRef CAS PubMed.
- Y. Ogawa, N. Shobako, I. Fukuhara, H. Satoh, E. Kobayashi, T. Kusakari, M. Suwa, M. Matsumoto and A. Ishikado, Rice Bran Supplement Containing a Functional Substance, the Novel Peptide Leu-Arg-Ala, Has Anti-Hypertensive Effects: A Double-Blind, Randomized, Placebo-Controlled Study, Nutrients, 2019, 11, 726 CrossRef CAS PubMed.
- T. Sartorius, A. Weidner, T. Dharsono, A. Boulier, M. Wilhelm and C. Schön, Postprandial Effects of a Proprietary Milk Protein Hydrolysate Containing Bioactive Peptides in Prediabetic Subjects, Nutrients, 2019, 11, 1700 CrossRef CAS PubMed.
- J. Tkaczewska, E. Jamróz, E. Piątkowska, B. Borczak, J. Kapusta-Duch and M. Morawska, Furcellaran-Coated Microcapsules as Carriers of Cyprinus carpio Skin-Derived Antioxidant Hydrolysate: An In Vitro and In Vivo Study, Nutrients, 2019, 11, 2502 CrossRef CAS PubMed.
- A. G. Garzón, M. D. R. Ferreira, R. E. Cian, M. E. Oliva, M. E. D'Alessandro and S. R. Drago, Microencapsulated bioactive peptides from brewer's spent grain promotes antihypertensive and antidiabetogenic effects on a hypertensive and insulin–resistant rat model, J. Food Biochem., 2022, 46, e14283 CrossRef PubMed.
- B. P. Singh, R. E. Aluko, S. Hati and D. Solanki, Bioactive peptides in the management of lifestyle-related diseases: Current trends and future perspectives, Crit. Rev. Food Sci. Nutr., 2022, 62, 4593–4606 CrossRef CAS PubMed.
- S. Chakrabarti, S. Guha and K. Majumder, Food-Derived Bioactive Peptides in Human Health: Challenges and Opportunities, Nutrients, 2018, 10, 1738 CrossRef PubMed.
- P. J. Patil, M. Usman, C. Zhang, A. Mehmood, M. Zhou, C. Teng and X. Li, An updated review on food-derived bioactive peptides: Focus on the regulatory requirements, safety, and bioavailability, Compr. Rev. Food Sci. Food Saf., 2022, 21, 1732–1776 CrossRef PubMed.
- Z. Du, J. Comer and Y. Li, Bioinformatics approaches to discovering food-derived bioactive peptides: Reviews and perspectives, TrAC, Trends Anal. Chem., 2023, 162, 117051 CrossRef CAS.
- A. B. Nongonierma and R. J. FitzGerald, Inhibition of dipeptidyl peptidase IV (DPP-IV) by proline containing casein-derived peptides, J. Funct. Foods, 2013, 5, 1909–1917 CrossRef CAS.
Footnote |
| † These authors contributed equally. |
| This journal is © The Royal Society of Chemistry 2025 |