Development of nanosized materials using organosulphur compounds as building blocks for applications in catalysis and electrocatalysis
Anupma
Tyagi
 a,
Suraj
Purohit
a,
Suraj
Purohit
 a,
Nivedita
Kunwar
a,
Anurag
Bahuguna
a,
Kamal K.
Pant
b and
Arun
Kumar
a,
Nivedita
Kunwar
a,
Anurag
Bahuguna
a,
Kamal K.
Pant
b and
Arun
Kumar
 *a
*a
aDepartment of Chemistry, School of Physical Sciences, Doon University, Dehradun-248012, India. E-mail: arunkaushik@gmail.com; akumar.ch@doonuniversity.ac.in
bDepartment of Chemical Engineering, IIT Delhi, New Delhi-110016, India
First published on 8th October 2025
Abstract
Over the past few decades, there has been extensive utilization of organosulphur compounds for the development of nanocatalysts for various chemical reactions. A wide array of organosulphur compounds and their preformed molecular complexes with numerous metals (including palladium, cobalt, copper, lead, nickel and silver) have been used for this purpose. Such compounds, complexes and nanocatalysts can be developed using diverse techniques. This article encompasses the strategies employed in designing relevant organosulphur compounds and exploring their utility in developing catalytically active nanomaterials. Various methodologies are discussed for the synthesis of nanomaterials, such as the single source precursor (SSP) method, reduction method for colloidal synthesis and indirect generation of nanoparticles. The intricacies of different synthetic methodologies are revealed in detail in this article. Additionally, the applications of such nanocatalysts in a diverse range of chemical transformations (such as Suzuki coupling, Sonogashira coupling, Heck coupling, C–O coupling reaction, and cross-dehydrogenative coupling (CDC)) are the main focus of this article. Their use in electrochemical studies is also rationalized in the context of the reactions such as the hydrogen evolution reaction (HER), oxygen evolution reaction (OER) and overall water splitting. A critical assessment of the variations in catalytic performances and the mechanistic aspects of nanocatalysis is also included. Moreover, it sheds light on the future perspectives of this field.
1. Introduction
Nanoscience is considered one of the key technology areas of the 21st century.1,2 It is well known that materials have unique properties at the nanoscale. Some of these properties are remarkable. The Lycurgus cup, consisting of gold and silver nanoparticles, was an early example of nanostructured materials.3 When lit from the outside, it appeared opaque green. However, when it is lit inside, it glows red.3 The word “nanomaterial” refers to any solid with nanometer-scale dimensions. The European Commission (EC) defined “nanomaterials” for the first time in 2011 as “a natural, incidental, or manufactured material containing particles in an unbound state or as an aggregate or as an agglomerate”.4 Nanocatalysis has been a very important field in the domain of nanoscience since a long period of time.5Nanocatalysts have gained much popularity because they exhibit exceptional properties, such as selectivity,6,7 high activity, durability, recoverability, recyclability and other unique attributes. Such properties are rarely observed in the bulk counterparts of these catalysts. Gold nanoparticles exhibit unexpected catalytic activity, which differs from that of the bulk gold. This can be taken as a good example of nanocatalysis.8–11 Nanocatalysis is sometimes referred to as a “semiheterogeneous”12 or “soluble heterogeneous”13 mode of catalysis. Nanoparticles can behave as active and stable heterogeneous catalysts or support materials for various catalytic groups.14 Hence, they also act as a bridge between homogeneous and heterogeneous catalysis.14 Such particles have a large surface area, which enables them to be in better contact with the reactants. As far as the ease of separation from reaction mixtures is concerned, nanostructured catalysts can function as heterogeneous catalysts. Like homogeneous catalytic systems, they exhibit customisable catalytic activity and selectivity at the nanoscale.15,16 It is worth noting that nanocatalysts with superior activity, stability, and selectivity6,7 can be developed and synthesised simply by controlling their sizes, shapes, and morphologies.14,17
Nanocatalysis is also referred to as a tool to promote green chemistry, i.e., a philosophy that promotes sustainable principles for reducing or eliminating chemicals and chemical processes that may create harm to the environment. It is primarily guided by twelve key concepts given by Paul Anastas and John Warner. Catalysis is one of the most significant principles in green chemistry. High-performance catalysts may open the door to green chemistry. For example, the use of metals (catalysts) in low quantities for performing the reactions successfully may result in considerable cost savings in chemical processes. This objective may be achieved using green and sustainable catalysts with high activity, improved selectivity, hassle-free recovery from the reaction media, durability or stability, recyclability, and low-cost. The majority of these critical challenges might be addressed using nano-sized metal particles as catalysts. As a result, catalysis by metals at the nanoscale is also a tool to enhance the sustainability of chemical processes. If such catalysts are highly active, the requirement and use of extreme reaction conditions may be avoided, resulting in energy-efficient processes. The increase in the selectivity of such catalysts reduces the formation of the by-products and allows chemical reactions to occur in a specific manner with low consumption of the substances, i.e., reactants. Through the enhancement in atomic efficiency and waste avoidance, the increase in the recyclability of catalytically active nanomaterials may also promote green chemistry.18
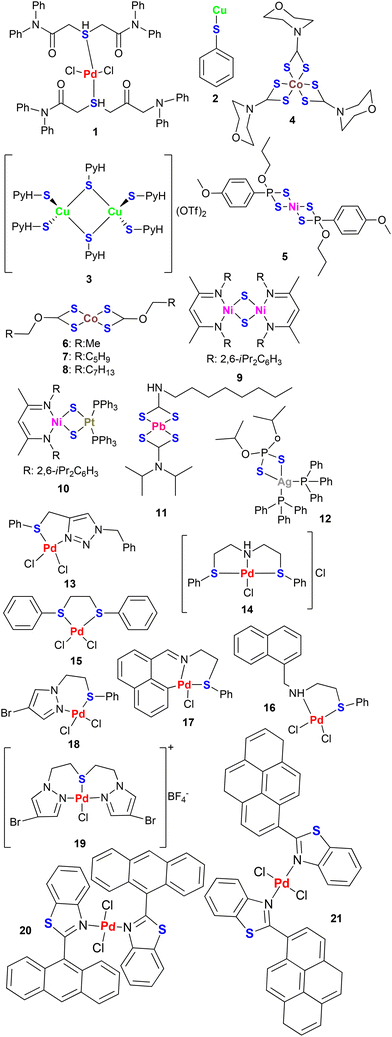
Organosulphur compounds are very significant in the context of the development of all kinds of catalysts, including homogeneous, heterogeneous and nanocatalysts.19,20 Such compounds have sulphur covalently bonded with carbon in the framework of the organic molecules. Sulphur is an element identified in 1777 by the French chemist Antonie Lavoisier (1743–1794).21 It exhibits an electron donation ability and shares similarities with non-metals (like selenium) of the same group of the periodic table. However, many of its properties (such as high electronegativity, smaller size, high electron affinity, and non-metallic character) are different from those of selenium. A large variety of organosulphur compounds have been designed and explored for the development of a wide range of catalysts and electrocatalysts. Such compounds are extensively used in various forms, such as thioethers, thiols, thiosemicarbazones, thiourea, Schiff bases, sulphur-containing N-heterocyclic carbenes (NHCs), and sulphur-containing pincer ligands. These compounds behave as versatile ligands and play a crucial role in the development of diverse nanocatalytic systems. A few reviews have been published in the area of nanocatalysis. However, an analysis of recent developments in the field of the role of organosulphur compounds in the field of catalysis and electrocatalysis has rarely been focused on.
This article comprehensively delves into the advancements related to the synthesis and utilization of organosulphur compounds in the development of nano-catalytic systems. It extensively covers various types of chemical transformations carried out using such nanocatalysts, including Suzuki–Miyaura coupling, Heck coupling, Sonogashira coupling, C–O coupling, and cross-dehydrogenative coupling (CDC). An in-depth analysis of the electrochemical applications of these nanocatalysts for the hydrogen evolution reaction (HER), oxygen evolution reaction (OER), and overall water splitting is also included. Additionally, this article critically evaluates the diversity in the catalytic potential, recyclability, and mechanistic aspects of catalysis. It also includes a comparative analysis focusing on the efficiency and other advantages of this unique class of catalysts.
2. Roles of ligands and preformed metal complexes in the development of nanocatalytic systems
In the case of homogeneous catalysts, the role and function of the ligands is quite clear in determining and rationalizing the activity, selectivity and efficiency of catalytically active molecular complexes. However, the same is not true in the case of nanocatalysts and heterogeneous catalysts. It is assumed that the ligands play multiple roles in metal-based nanocatalytic systems.22 Stabilization of the particles in the nano-size range is one of them. Other roles are yet to be studied thoroughly and established fully. As far as the role in stabilization is concerned, it is well-established that a stabilizing ligand primarily functions to prevent agglomeration, which is a tendency of the nanoscale materials. Hence, the stabilizing ligand renders them catalytically active and makes them suitable for applications in catalysis. It is unambiguous that dispersion of stabilized NPs, their catalytic performance (i.e., the outcome or consequence of their activity and efficiency), composition, solubility, morphology, recyclability and size are affected by the stabilizing ligand or capping agents up to a small or significant extent.23 The interaction between metal nanoparticles and stabilizing agent may be a strong covalent linkage, or chemisorption, or electrostatic in nature. The possibility of Ostwald ripening becomes low when the ligand adheres tightly to the metal surface. When the binding is very much strong, the circumstances are not favourable for the molecules that are approaching the surface of the nanoparticles. Such a condition can undermine catalytic activity by competing with substrate binding.24 Hence, for optimized catalytic activity and stability of the catalytic system, proper binding strength of the stabilizing ligand is crucial.23,24 The adoption of the reaction pathway and the coordination sphere of the metal is also manipulated by metal–ligand interaction and steric repulsions. The nature of the stabilizing ligand also affects the stability of nanocatalysts. It is also rationalized that two different interfaces are in existence due to a capping agent-generated organic shell on the nanoparticle surface (i.e. the metal–ligand interface and the ligand–solution interface).23 If the capping agent is not suitable, it can act as a “poison” for the nanocatalyst and limit the availability of the active sites.25 Hence, several studies have been performed on “ligand-free” synthesis of noble metal-based nanocatalytic systems.26 Nonetheless, due to the numerous benefits, such as controlling the size and the facet of nanoparticles during nanoparticle synthesis, ligands are utilized as building blocks for the development of nanocatalysts. Most of the efforts to date have been limited to exploring the role of molecular ligands in controlling the size and facet of nanoparticles during nanoparticle synthesis.22 Stabilizers such as dendrimers, surfactants, polymers, ionic liquids, and organofluorous ligands are used for the development of nanocatalysts.27,28 Many times, they completely encapsulate the nanoparticles, rendering them catalytically inactive. Therefore, organic ligands, such as thiols, have been found to be stabilizers. In some cases, such compounds lead to the formation of larger aggregates, and the depression in catalytic efficiency is negligible.29Organosulphur compounds have a strong coordinating site due to which they can bond with transition metals very well. Thus, they act as highly efficient stabilizers for metal nanoparticles.24 They have been utilized for designing nanocatalysts (Chart 1) using three approaches. One is the formation of NPs through the preformed molecular complexes, which provide high and uniform dispersion of the catalytically active nanocatalytic species.30 The other approach is to use them as stabilizers for transition metal nanoparticles due to the excellent affinity of S donor groups with transition metals. The third approach is through the isolation and separation of in situ-generated nanoparticles during chemical transformations. These particles not only exhibit long term stability in solution and good catalytic activity but also provide anti fouling and responsive properties for many biomedical and catalytic applications.24,31,32 Such sulphur containing compounds have also been reported to lead to the development of the catalytic systems, which play a central role in forming selective products. In such cases, their asymmetric nature leads to the formation of optically active products.33
Unlike the nanocatalysts, the preformed metal complexes have a definite and more specified role for the ligands in the context of catalytic activity.34 The nature and structure of the coordination sphere around the metal are completely determined by the ligand molecule(s). Thus, they play a significant controlling role on the activity and reactivity of the metal complexes in catalysis. The local ligand-induced microenvironment allows the molecular complex to grant permission or impose a restriction on the substrate molecules during their movement to reach the catalytically active metal site. This is how the ligand molecules play an important role in regulating the kinetic reactivity of the metal complex, leading to narrowing down or broadening the substrate scope for a specific reaction.34 The efficiency of the catalyst also relies on the lability of the bond between the metal and donor atom. In addition to activity and efficiency, ligands also affect the solubility of metal complexes in different solvents.36 As a result, ligand design has garnered significant attention from research groups worldwide.35
During the past two decades, the preformed metal complexes (Chart 2) have also been utilized to design tailor-made nanocatalytic systems for various catalytic and electrochemical applications. Such complexes have been used as single source precursors (SSPs) for synthesizing nanocatalytic systems or they have been used to carry out the in situ generation of the nanoparticles during catalysis.
3. Methodologies utilized for developing nanocatalytic systems using organosulphur-based ligands and metal complexes
Nanocatalytic systems (Charts 3–5) have been studied extensively in terms of their development and characterization before being used in catalysis or electrocatalysis. In the development of such systems, organosulphur compounds have been utilized in three ways. The use of different techniques and strategies can be exercised only after a thorough investigation and understanding of the capabilities of organosulphur compounds in designing nanocatalytic systems for improved catalytic performance.3.1. Single source precursor (SSP) method: use of organosulphur compounds as building blocks for single source molecular precursors
In this method, preformed metal complexes of organosulphur compounds are used as single source precursors for developing catalytically active and stable (Chart 3) metal sulphides. The main advantage of this method is the ease with which it leads to the synthesis of the product (i.e., nanomaterial). Such an ease includes the occurrence of the reaction at a moderate temperature and a straightforward protocol.Additional advantages are the purity of nanostructures, reduction in toxicity risks and negligible issues related to air and moisture sensitivity.
Flower-shaped nanocatalyst 22 (i.e., Pd16S7) has been synthesized by the thermolysis route (Scheme 1). In the first step, ligand L1 reacts with the palladium precursor Na2PdCl4 to obtain metal complex 1. Thereafter, nanosized Pd16S7 is synthesized by the thermolysis of complex 1 in trioctylphosphine (TOP) at 280 °C. The size of this nanocatalyst (Fig. 1) is nearly 31–40 nm.37
 | ||
| Scheme 1 Development of the flower-shaped Pd16S7 nanocatalyst 22 using metal complex 1 as a single source precursor.37 | ||
 | ||
| Fig. 1 Size distribution of the Pd16S7 nanocatalyst 22. Reproduced from ref. 37 with permission from the Royal Society of Chemistry. Copyright {2017}. | ||
Nanocatalyst 23 (i.e., Cu2S) has been developed using the SSP method. Complex 2 is subjected to thermolysis (Scheme 2) at 320 °C to obtain this catalyst. The size of the nanoparticles (Fig. 2) is in the range of 28–36 nm.38
 | ||
| Scheme 2 Development of nanocatalyst 23 using metal complex 2 as a single source precursor.38 | ||
 | ||
| Fig. 2 Size distribution of the Cu2S nanocatalyst 23. Reprinted (adapted) with permission from ref. 38. Copyright {2018}, the American Chemical Society. | ||
Nanocatalytic system 24 (i.e., Cu9S5) has been developed using a similar method in which copper complex 3 is used as a single source precursor.39 This complex is synthesized (Scheme 3) by reacting Cu(OTf)2 and 2-mercaptopyridine thiol in DCM for 8 hours under an inert atmosphere. When this complex is subjected to thermolysis in oleylamine at 250 °C, it leads to the generation of Cu9S5 hexagonal nanocrystals (24). The HR-TEM data clearly indicate that the d(101) lattice fringes have an interplanar distance of 0.33 nm. This confirms the purity of the nanocatalyst.39
 | ||
| Scheme 3 Synthesis of the dinuclear copper(I) complex 3 and its use as a single source precursor to generate the nanoparticle 24.39 | ||
Nanocatalyst 25 is CoS (sulphur rich). It is made up of nanoplates that have a slightly sulphur-rich composition. Similarly, nanocatalyst 26 is CoS (cobalt rich). In this case, the nanoplates have a cobalt-rich composition. Both of them are obtained when cobalt complex 4 is thermolyzed (Scheme 4) in oleylamine (OLA). Temperature influences the final product in this case. When the process of thermolysis is carried out at 200 °C, the formation of the 25 (sulphur rich CoS) occurs. When the thermolysis is performed at 260 °C, 26 (cobalt rich CoS) is obtained (Scheme 4). P-XRD and XPS investigations provide sufficient data to confirm the successful formation of both the nanocatalysts.40
 | ||
| Scheme 4 Synthesis of the cobalt sulphide nanoplates 25 and 26.40 | ||
Nanocatalytic systems 27, 28, and 29 have NiS (nickel sulphide).41 They have been synthesized using a sulphur ligated complex of nickel (i.e., the 5). Thermal degradation of this complex is carried out to obtain them. When thermolysis is carried out (Scheme 5) in the mixture of n-hexadecylamine and 1-octadecene system at 300 °C, hexagonal nanocrystals 27 are obtained. Interestingly, when a single molecular precursor (i.e., complex 5) is thermolyzed at 400 °C under nitrogen atmosphere and solvent free conditions, it yields hexagonal nanocrystals of 28 (Scheme 5). However, when the complex is thermolyzed at 300 °C in tri-octylphosphine oxide and ODE, rhombohedral nanocrystals of NiS (29) are obtained. P-XRD data are sufficient to indicate that these crystals contain NiS.41
 | ||
| Scheme 5 Synthesis of the nanocatalytic systems 27–29.41 | ||
Nanocatalytic systems 30, 31 and 32 are self-capped nanoparticles of CoS (cobalt sulphide).42 They are synthesized (Scheme 6) by the thermal decomposition of cobalt xanthate complexes 6, 7, and 8, respectively. The thermolytic process is carried out at 250 °C under solvent free conditions (Scheme 6). The composition and morphology of the synthesized systems are verified using P-XRD, XPS, SEM and TEM analyses.42 The TEM data reveal that nanoparticles 30, 31 and 32 are spherical in shape. The size of the nanoparticles decreases with the increase in alkyl chain length; this could be due to the possibility of better dispersion of nanoparticles with longer alkyl chains. The order of the size of the nanoparticles is 30 > 31 > 32.42
 | ||
| Scheme 6 Synthesis of nanoparticles 30–32 by thermolyzing the complexes 6–8 in solvent-free conditions.42 | ||
Moreover, 33 and 34 are interesting nanocatalytic systems.43 Both of them contain nickel sulphide (NiS). Nanocatalyst 34 is a core–shell bimetallic alloy system. It has a nickel–platinum alloy (NiPt3) protected by amorphous NiS structures (Scheme 7). Additionally, 33 is synthesized (Scheme 7) by the thermolysis of complex 9 under N2 atmosphere at 200 °C. Interestingly, the development of 34 involves the thermolysis of a hetero-bimetallic complex of nickel and platinum (i.e., complex 10) under similar conditions.43 The Ni–Pt nanoparticles, i.e., 34, are approximately 2 nm in size. The EDX data revealed that the compositions of Ni, Pt and S in the nanoparticles are 40%, 30% and 30%, respectively.43
 | ||
| Scheme 7 Synthesis of nanoparticles 33 and 34.43 | ||
Nanocatalytic systems 35 and 36 are the nanoparticles of PbS.44 Nanocatalytic system 35 is developed (Scheme 8) by dissolving complex 11 (the Pb complex of an organosulphur compound) in oleic acid and thereafter infusing the same into hexadecylamine (HDA) at 360 °C for 1 hour. Similarly, nanocatalytic system 36 is synthesized (Scheme 8) using a thermogravimetric analyzer (TGA), in which complex 11 is placed in an alumina pan and the temperature is gradually increased from 30–900 °C with a continuous flow of nitrogen, yielding nanoparticles 36.44 Nanoparticles 35 and 36 are spherical in shape. The nanoparticles synthesized using a solvent (oleic acid and HDA), i.e., 35, are smaller in size (1.82–2.44 nm) in comparison to the nanoparticles synthesized without any solvent, i.e., 36, with a size in the range of 3.16–5.95 nm.44
 | ||
| Scheme 8 Synthesis of nanoparticles 35 and 36 using complex 11.44 | ||
Nanocatalytic system 37 contains extremely thin Ag2S nanowires. The wires crystallize in a monoclinic system. They are synthesized by the thermal degradation of molecular complex 12 at a moderate temperature (Scheme 9), i.e., 140 °C in a mixture of octadecylamine, oleylamine, and n-decylboronic acid. These nanowires have an average diameter of 2.5 nm. However, their length is approximately 500–700 nm.45
 | ||
| Scheme 9 Synthesis of the silver nanowire 37 using complex 12.45 | ||
3.2. Nanocatalysts as by-products: use of molecular complexes of organosulphur compounds as dispensers
There are some remarkable reports on the generation of nanocatalysts during the course of homogeneous catalysis by palladium complexes (Chart 4) of organosulphur compounds. These nanocatalysts are either nanosized binary phases of palladium sulphide or palladium(0) species. The properties of such nanocatalysts are greatly dependent on the molecular structure of the organosulphur ligands. These nanocatalysts, which are probably the true catalytic species in the reactions, can be isolated and separated from the reaction mixtures. They have a good shelf-life and can be stored.Nanocatalysts 38 and 39 have palladium in a zero oxidation state.46 The size (5–6 nm) of the particles in 39 is larger than that (3–5 nm) in 38. Both of them are generated (Scheme 10) by complex 13 in which the Pd(II) is bound to the nitrogen of the 1,2,3-triazole unit of the ligand. When complex 13 is used to catalyze Suzuki–Miyaura coupling (Scheme 10), 38 is formed. When the same complex is used for the catalysis of Heck coupling reactions (Scheme 10), 39 is obtained.46
 | ||
| Scheme 10 Generation of nanoparticles 38 and 39 as by-products of the reactions (SMC and Heck coupling) catalyzed by the preformed molecular complex 13.46 | ||
Nanocatalyst 40 has the quantum dots of palladium.47 Their size is approximately 1–3 nm (Fig. 3). When complex 14 is used to catalyze the Suzuki coupling reaction, quantum dots are formed and released (Scheme 11) in the reaction mixture. They are stabilized by the fragments of the organosulphur ligand of complex 14. They can be isolated and separated from the reaction mixture. They can be stored. They have the ability to show independent catalytic activity.47
 | ||
| Fig. 3 Size distribution of the Pd(0) quantum dot nanocatalyst 40. Reproduced from ref. 47 with permission from the Royal Society of Chemistry, Copyright {2013}. | ||
 | ||
| Scheme 11 Generation of nanoparticle 40 as a by-product of the SMC reaction catalyzed by the preformed molecular complex 14.47 | ||
Nanocatalytic system 41 has nanosized species of Pd(0).48 Their size is approximately 7 nm. When Pd(II) complex 15 is used (Scheme 12) as a catalyst for the SMC reaction, the generation of 41 occurs (Scheme 12) in the reaction mixture. During the Suzuki coupling reaction, the nucleation of discrete palladium is likely to occur, leading to the formation of peanut-shaped nanoparticles (Scheme 12). The size and shape of the nanoparticles depend on the nature of the coordinated atoms of the organosulphur ligand with palladium in complex 15.48
 | ||
| Scheme 12 Generation of nanoparticle 41 as a by-product of the Suzuki–Miyaura coupling reaction catalyzed by the preformed molecular complex 15.48 | ||
Nanocatalysts 42 and 43 contain nanoparticles of palladium.49 In the case of 42, the size of the NPs is 5 nm. However, 43 contains NPs of a slightly larger size (6 nm). Moreover, 42 is generated (Scheme 13) by complex 16 during the catalysis of the Suzuki coupling reaction. In this complex, an organosulphur ligand is coordinated with palladium in bidentate mode. Palladium(II) complex 17 (in which an organosulphur ligand is coordinated to the metal in tridentate mode) leads to the formation of 43. Both complexes have nitrogen as a hard donor and sulphur as a soft donor (Scheme 13) in their molecular structures.49 Interestingly, in complex 17, the ligand is a pincer-type (S, N, C donor) compound.49
 | ||
| Scheme 13 Generation of nanoparticles 42 and 43 as by-products of the SMC reaction catalyzed by the preformed molecular complexes 16 and 17.49 | ||
When Pd(II) complexes 16 and 17 are used for catalyzing the Sonogashira coupling reaction, they lead to the formation of nanoparticles (Scheme 14) 44 and 45, respectively. The difference in size between 44 and 45 is greater than that in the cases of 42 and 43. The sizes of the nanoparticles are 3 nm and 6 nm for 44 and 45, respectively.49
 | ||
| Scheme 14 Generation of nanoparticles 44 and 45 as by-products of the Sonogashira coupling reaction catalyzed by the preformed molecular complexes 16 and 17.49 | ||
A variety of binary phases of PdxSy (Pd4S, Pd3S, Pd2.8S, Pd16S7, PdS, and PdS2) are known. The majority of such phases are prepared by gas sulphuration with H2S–H2.50–56 Moreover, 46 and 47 are the nanoparticles of the different binary phases of palladium sulphide. Additionally, 46 contains the nanoparticles of Pd4S (Scheme 15).57 In addition, 47 has a different composition. Its nanoparticles are made up of Pd5S2 (Scheme 15). They have significant differences in their sizes. The sizes of these nanoparticles are 19 nm and 5 nm for 46 and 47, respectively.57 The structure of Pd4S was reported in 1962.58 It is isostructural with Pd4Se. The electrical conductivity of Pd4S is similar to that of the metal,59,60 but its tendency for the permeability of H2 is different from that of the metallic Pd.61,62 In the recent past, palladium sulphides have attracted the attention of chemists. Due to the interactions between Pd and S, their physicochemical properties are unique. Such properties include tunable catalytic behaviour, enhanced electron density modulation and improved thermal stability.63 Their catalytic applications have not been given sufficient attention. Only a few reports on Pd4S exist to understand its catalytic potential,64,65 electrocatalytic potential,66,67 photocatalytic potential59 and hydrogen permeability.62 It has been found to show catalytic potential for selective gas phase alkyne hydrogenation,64 liquid phase alkyne hydrogenation,54,63 Suzuki coupling,65 oxidation of methane68 and oxygen reduction reaction.66 As far as the methodologies of its synthesis are concerned, gas sulphuration with H2S–H2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50–56 has also been reported as a method for its synthesis. It has also been synthesized using direct methods. However, such methods are challenging due to the harsh reaction conditions typically required.52,53,62,68 The SSP route has rarely been reported for this.66 To overcome these limitations, single-source precursor (SSP) methodologies have emerged as elegant and controllable strategies for the synthesis of metal chalcogenide nanomaterials.69–72 Another interesting approach for its synthesis is in situ generation. Moreover, 46 and 47 are generated (Scheme 15) by complexes 18 and 19 during the catalysis of the Suzuki–Miyaura coupling reaction, respectively. Complex 19 has a tridentate ligand with two nitrogen and one sulphur donor atom (Scheme 15), and complex 18 has a bidentate (N, S donor) ligand.57 Interestingly, the nanoparticles generated via these complexes are not Pd(0) nanoparticles; they are Pd4S (46) and Pd5S2 (47).57
50–56 has also been reported as a method for its synthesis. It has also been synthesized using direct methods. However, such methods are challenging due to the harsh reaction conditions typically required.52,53,62,68 The SSP route has rarely been reported for this.66 To overcome these limitations, single-source precursor (SSP) methodologies have emerged as elegant and controllable strategies for the synthesis of metal chalcogenide nanomaterials.69–72 Another interesting approach for its synthesis is in situ generation. Moreover, 46 and 47 are generated (Scheme 15) by complexes 18 and 19 during the catalysis of the Suzuki–Miyaura coupling reaction, respectively. Complex 19 has a tridentate ligand with two nitrogen and one sulphur donor atom (Scheme 15), and complex 18 has a bidentate (N, S donor) ligand.57 Interestingly, the nanoparticles generated via these complexes are not Pd(0) nanoparticles; they are Pd4S (46) and Pd5S2 (47).57
 | ||
| Scheme 15 Generation of nanoparticles 46 and 47 as by-products of the SMC reaction catalysed by the preformed molecular complexes 18 and 19.57 | ||
Nanocatalysts 48 and 49 also contain the binary phases of palladium sulphide.73 Thus, 48 contains the nanoparticles of PdS, which are 10–12 nm in size. However, 49 has the nanoparticles of Pd16S7, which are agglomerated (revealed by HR-TEM analysis). When the Suzuki–Miyaura coupling reaction is carried out using Pd(II) complexes 20 and 21 (Scheme 16) as catalysts, 48 and 49 are generated. These nanoparticles are isolated and separated for their independent use as the catalysts.73
 | ||
| Scheme 16 In situ generation of nanoparticles 48 and 49 during the Suzuki–Miyaura coupling reaction using the preformed molecular complexes 20 and 21.73 | ||
3.3. Use of organosulphur compounds as stabilizers for the development of nanocatalysts
The synthesis of nanoparticles can be achieved through various techniques, such as sol–gel,74–79 hydrothermal,80–82 microemulsion,83,84 chemical co-precipitation,85–87 and spray pyrolysis.88,89 The science of nanoparticles hinges on two key factors: achieving the development of the nanoparticles with controlled size and shape, and rendering them the potential for specific applications. Such applications also include catalysis and electrocatalysis. Controlled size and shape are primarily achieved through the use of stabilizing agents, such as ligands (thiols, amines, and phosphines), surfactants (ammonium salts), and polymers (polyvinyl alcohols). The selectivity6,7,15,16 and reactivity of nanoparticles play crucial roles in influencing the outcome of a reaction. The activity is dependent on many factors, including the surface area of nanoparticles. The involvement of inter-atomic interactions and the adjustment of under-coordinated/less coordinated surface atoms constitute the basis on which the properties of nanomaterials may be tuned. Such a possibility is rarely present in their bulk counterparts. Reduction in atomic coordination significantly impacts the performance of surfaces, nano-solids, and amorphous solids in terms of bond relaxation and energy.90,91The stability of nanoparticles against aggregation can be achieved using thermodynamic and kinetic approaches. The thermodynamic approach is adopted to suppress Ostwald ripening by minimizing the difference in chemical potential and surface energy between the nanoparticles (NPs).92 On the basis of the results of the study on the thermodynamic behaviour of supported metal particles in various reaction environments, a general strategy has been suggested for the inhibition of the sintering of such NPs through theoretical calculations.92 In this study, it has been observed that the stability of the particles and the rate of Ostwald ripening are determined by their size and distribution. When the sizes of the NPs are in a narrow range and their distribution is homogeneous, they have similar chemical potential and surface energy. In such cases, the Ostwald ripening rate is very low. Moreover, this objective can be achieved using a support with high surface activation energy. Therefore, in the preparation of NPs with uniform size and distribution, the selection of a suitable support is crucial for enhancing the stability of supported nano-catalysts.92 The kinetic approach provides the basis for the use of an external medium (such as high temperature or high pressure) to limit the size of the nanoparticles. The rate of aggregation can be determined using various techniques, such as spectral turbidimetry, time-resolved dynamic light scattering (TR-DLS), and fluorescence correlation spectroscopy (FCS).93
Organosulphur compounds have sulphur as the donor site. They behave as very good stabilizers in the development of metal nanoparticles.24 Nanoparticles of palladium with high catalytic potential have been developed using such compounds as stabilizers. In this method, a source of palladium (e.g. Na2PdCl4) is reduced in the presence of an organosulphur compound, and catalytically active and stable nanoparticles of metal (Chart 5), which are stabilized with such a compound, are produced. Such nanoparticles can be isolated and stored. The stabilizing ligands play an important role in dictating the size, shape, dispersion, and inter-particle spacing of nanoparticles. These compounds also affect the catalytic efficiency and recyclability of the synthesized NPs through ligation. The stabilizer compounds, which have functional groups such as amine, alcohol, carbonyl, thiol, hydroxy and phosphine, inhibit strong chemisorptions to the metal surface.94 Therefore, the stabilizing ligand must be strong enough to stabilize the nanoparticle but should not have extremely strong interactions that hinder the catalytic activity of the NPs.
Nanoparticles 50 and 51 are palladium nanoparticles stabilized by an organosulphur compound. They have been synthesized using L17 (a reduced Schiff base) as a stabilizer (Scheme 17). The size of nanoparticles has been found to be in a range of 2–3 nm in 50 and 4–5 nm in 51. The only difference between them is the ratio of metal and stabilizing ligand (Scheme 17), with 50 having a ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 (metal precursor
1 (metal precursor![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ligand), while 51 has a ratio of 1
ligand), while 51 has a ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4 (metal precursor
4 (metal precursor![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ligand). Ligand L17 and palladium precursor Na2PdCl4 have been reduced using a reducing agent with different concentrations of metal and ligand to obtain NPs 50 and 51.95
ligand). Ligand L17 and palladium precursor Na2PdCl4 have been reduced using a reducing agent with different concentrations of metal and ligand to obtain NPs 50 and 51.95
 | ||
| Scheme 17 Development of the Pd(0) nanoparticles 50 and 51 using the organosulphur ligand L17 as the stabilizer.95 | ||
Ultrasmall nanoparticles 52, 53 and 54 have been synthesized (Scheme 18) using ligand L18 (containing the combination of sulphur and nitrogen donors) as a stabilizer.96 Similar to the above-described NPs, the difference between these NPs is the ratio between the ligand and metal precursor. The reaction between ligand L18 and Na2PdCl4 with different ratios (metal precursor![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ligand) is utilized to obtain 52 (1
ligand) is utilized to obtain 52 (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), 53 (1
1), 53 (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4) and 54 (4
4) and 54 (4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) NPs. The size of NPs has been found to be in the range of 1–2 nm for 52, 2–3 nm for 53 and 4–6 nm for 54.96
1) NPs. The size of NPs has been found to be in the range of 1–2 nm for 52, 2–3 nm for 53 and 4–6 nm for 54.96
 | ||
| Scheme 18 Development of the Pd(0) nanoparticles 52–54 using the organosulphur ligand L18 as the stabilizer.96 | ||
Nanoparticles 55–60 have been formed by n-didocosyl sulphide ligand L16 (Scheme 19). The nanoparticles have been synthesized by the reaction between different palladium precursors [i.e., Na2PdCl4, Pd(OAc)2 and Pd(MeCN)2Cl2] and ligand L16 with different ratios, such as 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, 1
1, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 and 1
2 and 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4, for each precursor to give nanoparticles 55–60. The NPs synthesized using Na2PdCl4 are found to be uniformly dispersed and hence have better catalytic activity among all.97
4, for each precursor to give nanoparticles 55–60. The NPs synthesized using Na2PdCl4 are found to be uniformly dispersed and hence have better catalytic activity among all.97
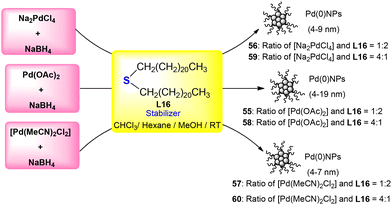 | ||
| Scheme 19 Development of the Pd(0) nanoparticles 55–60 using the organosulphur ligand L16 as the stabilizer.97 | ||
Pd–Ni-based nanocatalytic system 61 has been synthesized (Scheme 20) by reducing Ni(NO3)2 and Pd(OAc)2 in the presence of 2-mercaptoethanol (L24) as a stabilizer98 in an aqueous basic (NaOH) solution. The pH of the mixture is maintained at around 11.5. The nanoparticles have been characterized using PXRD, SEM, IR and UV spectroscopic techniques. The majority of the nanoparticles have been obtained with less than 20 nm. The average particle size of nanoparticle 61 is 8.6 nm.98
 | ||
| Scheme 20 Development of the Pd–Ni nanoparticle 61 using the organosulphur ligand L24 as the stabilizer.98 | ||
4. Application in catalysis
4.1. Suzuki–Miyaura C–C cross coupling reaction
The Suzuki–Miyaura coupling reaction involves forming a C–C bond (Scheme 21) between an aryl halide and organoborane compounds.99 This reaction is highly valued due to the simplicity of its process, such as mild conditions, the use of non-hazardous reagents, and its adaptability and utility across various industries, including the production of natural products and agrochemicals.99,100 Extensive research has been conducted on the catalysts (developed using organosulphur compounds) for this reaction. Numerous nanocatalytic systems are also among them. Their applications provide quite interesting results.Pd16S7 nanoflowers (22) are highly active nanocatalysts for the SMC reaction. Low catalyst loading (i.e., 0.5 mol%) is sufficient for catalyzing this reaction within 3 hours. When 4-bromoacetophenone and phenyl boronic acid are reacted (Scheme 22) in the presence of its low (0.5 mol%) concentration, the coupled product is obtained with 91% yield.37 It is recyclable up to four runs, and the yield obtained after the 4th cycle is 38%.37
 | ||
| Scheme 22 Suzuki–Miyaura coupling using Pd16S7 (22) nanoflowers.37 | ||
Nanocatalyst 38 (developed from complex 13) is very much useful.46 It has the ability to catalyse the SMC reactions independently. The substrates including 4-bromobenzaldehyde, 4-bromoanisole and 4-bromoacetophenone react with phenylboronic acid in the presence of a very low quantity (nearly 5 mg) of this catalyst. The reaction between p-bromobenzaldehyde and phenyl boronic acid results in a 94% yield of the coupled product (Scheme 23) in a short duration of just 3 hours.46
 | ||
| Scheme 23 Variation in the performance of in situ generated nanocatalysts 38 and 47 in the Suzuki–Miyaura coupling reaction.46,57 | ||
However, nanocatalyst 47 (i.e., NPs of Pd5S2) works at a relatively high (10 mg) catalyst loading for the same reaction (Scheme 23) and allows for the formation of the product in 02 hours.
Moreover, 41 (peanut-shaped nanoparticles) is used as a nanocatalyst for the SMC reaction (Scheme 24) of phenylboronic acid with 4-bromobenzaldehyde. Its low quantity (10 mg) gives the coupled products, i.e., biphenyl-4-carboxaldehyde, in a very high (94%) yield in a very short duration of 02 hours at 100 °C. The reaction of 4-bromoanisole suffers from a low reaction rate due to the presence of the OMe group (an electron donating substituent) at the para position to the C–Br bond. It is converted to 4-methoxybiphenyl (the cross-coupled product) with a relatively low (78%) yield after a long duration of 12 hours under the same reaction conditions.48
 | ||
| Scheme 24 Effects of electron-withdrawing and electron-donating substrates on the performance of nanocatalyst 41.48 | ||
Nanocatalysts 42 and 43 (generated by complex 16 and palladacycle 17) also catalyze the Suzuki coupling reactions.49 Their low quantity (2.0 mol%) allows the reactions to proceed and gives the coupled product with good yields, i.e., 71–95%. Nanocatalyst 42 performs better than nanocatalyst 43 in all the coupling reactions. The size of 42 (5 nm) is slightly larger than that of 43 (4 nm). The particles of 42 are more dispersed than those of 43. This could be the reason for the better activity of 42 in the coupling reactions. For example, when 4-bromobenzonitrile is reacted with phenyl boronic acid, the presence of 42 gives the product in a 91% yield. The same reaction gives the product (Scheme 25) in 81% yield in the presence of 43 under the same reaction conditions.49
 | ||
| Scheme 25 Effect of the dispersion of NPs on the efficiency of the SMC reaction.49 | ||
Nanocatalysts 46 and 47 have also been independently utilized as catalysts for the SMC reactions. There is no significant variation between their catalytic performances. The coupling of 4-bromobenzaldehyde with phenyl boronic acid gave 92 and 95% yields within 2 hours (Scheme 23) in the presence of 10 mg of the nanocatalyst (i.e., 46 and 47, respectively).57 The high surface to volume ratio and uniformity in distribution of NPs are likely to be among the reasons and the factors that are responsible for their enhanced catalytic activity.57
 | ||
| Scheme 26 Suzuki–Miyaura coupling reaction catalyzed by NP 61via ultrasonication.98 | ||
 | ||
| Scheme 27 Suzuki–Miyaura coupling reaction catalyzed by NP 40 in water.47 | ||
 | ||
| Scheme 28 Effect of agglomeration on the performance of in situ generated nanocatalysts 48 and 49 in Suzuki–Miyaura coupling.73 | ||
 | ||
| Scheme 29 Suzuki–Miyaura coupling reaction catalyzed by nanocatalyst 51.95 | ||
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1. Palladium is present in low concentrations, and the stabilizing ligand is present in high concentrations. Ligands could encapsulate the palladium (or the active sites) in such a way that the reactants could not reach the active sites efficiently. Nanocatalyst 52 has shown the highest efficiency (Scheme 30) and reactivity among the three NPs, i.e., 52–54, possibly due to their relatively small size and high uniformity in the dispersion.96
1. Palladium is present in low concentrations, and the stabilizing ligand is present in high concentrations. Ligands could encapsulate the palladium (or the active sites) in such a way that the reactants could not reach the active sites efficiently. Nanocatalyst 52 has shown the highest efficiency (Scheme 30) and reactivity among the three NPs, i.e., 52–54, possibly due to their relatively small size and high uniformity in the dispersion.96
 | ||
| Scheme 30 Suzuki–Miyaura coupling reaction catalyzed by nanocatalyst 52.96 | ||
Nanocatalytic system 55–60, developed by the reduction method using L16 as the stabilizer, has the potential for catalysing the SMC reactions of bromoarenes.97 Among them, the highest activity is exhibited by nanoparticle 56, the development of which is carried out using sodium tetrachloropalladate (Na2PdCl4). They have very high uniformity in size and dispersion. When 4-bromoanisole and phenylboronic acid are reacted (Scheme 31) at 0.1 mol% catalyst loading, nanocatalyst 56 gives an excellent yield (i.e., ≥99%) in a short reaction time of 1 hour. However, 55, 57, 58, and 59 give moderate yields. Moreover, 60 has shown negligible efficiency for the reaction, and the product is obtained in trace amounts. The order of efficiency of these catalysts has been found to be 56 > 58 > 57 > 59 > 55 > 60.97
 | ||
| Scheme 31 Suzuki coupling reaction catalyzed by nanocatalyst 56.97 | ||
4.2. Heck coupling reaction
The Heck coupling reaction forms substituted olefins (Scheme 32) by the treatment of an aryl halide with terminal alkenes.101,102 This reaction is feasible under mild conditions and can accommodate a variety of functional groups, such as aldehyde, amines, esters and nitro groups, without compromising the results (i.e., high yields of the products).103 It is widely used in the production of fine chemicals.101,102 Organosulphur compounds are often used to develop various types of catalytic systems for this reaction.104 Like the Suzuki coupling reaction, it has also been observed in this reaction that some nanoparticles or nano species are generated in situ during the catalysis of this reaction with preformed metal complexes. Such nanoparticles (e.g., 39) can be isolated from the reaction mixtures and stored for their applications as independent nanocatalysts.46Moreover, 39 is utilized for the Heck coupling reaction of aryl bromides (such as 4-bromobenzaldehyde and 4-bromoacetophenone), which have highly active electron withdrawing groups with n-butyl acrylate. When 4-bromobenzaldehyde and n-butyl acrylate are reacted (Scheme 33) in the presence of 30 mg of the catalyst, i.e., 39, the yield of the product obtained is 75%. This nanocatalyst is unable to convert 4-bromoanisole into the product.46
 | ||
| Scheme 33 Heck coupling reaction using the nanocatalytic system 39.46 | ||
4.3. Sonogashira coupling
Sonogashira coupling is a reaction in which a terminal alkyne and an aryl halide combine (Scheme 34) with each other through the formation of a new C–C bond. This method is commonly used to produce pharmaceuticals, natural products, organic compounds and nanomaterials.105Like the Suzuki and Heck coupling reactions, during the catalysis of Sonogashira coupling reactions by Pd(II) complexes 16 and 17, Pd(0) nanoparticles, i.e., 44 and 45, are generated in situ in the reaction mixture.49 These nanoparticles (i.e., 44 and 45) are isolated and stored. They can be used independently for catalysis. They produce moderate to high yields (ranging from 42 to 91%) in Sonogashira coupling reactions. When 1-bromo-4-nitrobenzene is reacted with phenyl acetylene utilizing a 2.0 mol% concentration of the nanocatalyst without the addition of any co-catalyst, 44 and 45 (Scheme 35) give the desired product in 76% and 91% yields, respectively.49 This high performance of 45 could be attributed to the smaller size of Pd(0) NPs in this case.
 | ||
| Scheme 35 Variation in the performance (due to the size of nanoparticles) of in situ-generated nanocatalysts 44 and 45 in Sonogashira coupling.49 | ||
4.4. O-Arylation of phenol
In O-arylation, phenol combines with aryl halides (Scheme 36) to form diaryl ethers. This type of C–O coupling is widely used in the production of polymers106 and agrochemicals.106 Many palladium-based nanocatalytic systems have been developed for this reaction.106Nanocatalyst 22 (i.e., flower-shaped nanoparticles of Pd16S7), obtained from palladium complex 1 using the single source precursor method, has the ability to carry out the catalysis of this reaction.37 This nanocatalyst is used for the coupling of phenol with a variety of aryl bromides at a low catalyst loading of 0.5 mol% under mild reaction conditions. When 1-bromo-4-nitrobenzene and phenol are reacted at 100 °C (Scheme 37) in the presence of a 0.5 mol% concentration of the catalyst, the coupled product is obtained with a 92% yield. The bromides with electron donating groups, such as –CH3, –H or –OCH3, show less reactivity compared to the ArBr, which has electron withdrawing groups, such as –CHO, –NO2, –CN or –COCH3.37
 | ||
| Scheme 37 O-Arylation of phenol using nanocatalyst 22.37 | ||
4.5. Cross-dehydrogenative coupling (CDC)
Cross-dehydrogenative coupling is a chemical reaction (Scheme 38) that forms a C–C bond or C–heteroatom bond by removing two hydrogen atoms from two compounds.107,108 The formation of such bonds is significant in the synthesis and functionalization of complex organic molecules, including natural products and pharmaceuticals. CDC reactions are often catalyzed by metals and can proceed under mild conditions, offering a versatile and efficient approach to C–C or C–heteroatom bond formation in organic synthesis.107Nanocatalytic system 23 has been explored for cross dehydrogenative coupling (Scheme 39) of N,N-dimethylbenzylamines with azoles under solvent free conditions.38 When N,N-dimethyl-1-phenylmethanamine and 1H-1,2,3-triazole are reacted together in the presence of 1.0 mol% concentration of this catalyst (Scheme 39), the desired product is obtained with a 74% yield. This catalyst is also reusable for up to 5 reaction cycles with only a slight loss in activity. Depending on the substrate used, the yield of the product can reach up to 89% in the case of catalysis with the 23.38
 | ||
| Scheme 39 Cross-dehydrogenative coupling reaction catalyzed by nanocatalyst 23.38 | ||
The proposed mechanism of the reaction using nanocatalyst 23 is depicted in Scheme 40.38 In the first step, copper-catalyzed decomposition of tert-butyl hydroperoxide (TBHP) takes place (Scheme 40) to generate a tert-butoxyl radical. A radical cation is generated via a single electron transfer (SET) from an amine. After this step, the abstraction of the sp3 hydrogen of the radical cation takes place to generate iminium-type intermediate I or II (Scheme 40). In the next step, triazole/carbazole reacts with compound I, which results in the formation of compound III (Scheme 40). In the last step, the C–N coupled product is formed (Scheme 40) via a nucleophilic substitution reaction between the azole and iminium ion, regenerating nanocatalyst 23 for the next catalytic cycle. The organosulphur compound (i.e., 2) utilized for the synthesis of nanocatalyst 23 does not directly influence the reaction mechanism; however, it has a role in the development of the particular binary phase Cu2S nanoparticles (23), which are used as catalysts for this reaction.38
 | ||
| Scheme 40 Proposed mechanism of cross-dehydrogenative coupling reaction catalyzed by nanocatalyst 23.38 | ||
5. Applications in electrocatalysis
Metal sulphides have emerged as highly promising catalysts in electrochemical processes over the last decade owing to their diverse range of properties. Such properties encompass high surface area, excellent electrical conductivity, metallic structure and unique morphology. It is worth noting that these materials have demonstrated high potential in electrocatalytic reactions, like the hydrogen evolution reaction (HER), oxygen evolution reaction (OER) and overall water splitting. These properties make them useful and crucial for advancement in electrocatalysis.5.1. Hydrogen evolution reaction (HER)
Nanocatalysts 25, 26, 33, 34 and 37 are interesting species for the hydrogen evolution reaction (HER) (Table 1). This reaction involves the electrochemical process in which molecular hydrogen is generated (Scheme 41).| S. no. | Catalyst | Current density (mA cm−2) | Overpotential towards the HER (mV) | Tafel slope for the HER (mV dec−1) | Overpotential towards the OER (mV) | Tafel slope for the OER (mV dec−1) | Ref. |
|---|---|---|---|---|---|---|---|
| 1. | 24 (Cu9S5) | 10 | — | — | 293 | 67 ± 3 | 39 |
| 2. | 25 (CoS S rich) | 10 | 132 | 159 | 307 | 96 | 40 |
| 3. | 26 (CoS Co rich) | 10 | 153 | 154 | 276 | 82 | 40 |
| 4. | 27 (NiS hexagonal nanocrystals) | 50 | 273 | — | 389 | — | 41 |
| 5. | 28 (NiS hexagonal nanocrystals) | 50 | 259 | — | 389 | — | 41 |
| 6. | 29 (NiS rhombohedral nanocrystals) | 50 | 231 | — | 371 | — | 41 |
| 7. | 30 (CoS obtained from 6) | 10 | 200 | 126 | 325 | 77 | 42 |
| 8. | 31 (CoS obtained from 7) | 10 | 232 | 138 | 361 | 67 | 42 |
| 9. | 32 (CoS obtained from 8) | 10 | 235 | 101 | 339 | 77 | 42 |
| 10. | 33 (NiS) | −10 | 107 | 71 | — | — | 43 |
| 11. | 34 (NiPt3 coated with NiS) | −10 | 12 | 31 | — | — | 43 |
| 12. | 37 (Ag2S) | 10 | −88 | 52 | — | — | 45 |
Overpotentials of 132 mV and 153 mV have been observed when 25 and 26 are employed as catalysts (Scheme 39) for the hydrogen evolution process.40 These are cobalt sulphide (CoS) nanoparticles. The value of the Tafel slope is 159 mV dec−1 for 25. However, this value is slightly low (154 mV dec−1) for 26. When they are applied for hydrogen evolution studies, a standard three-electrode system [with samples on nickel foam as the working electrode, platinum wire as the counter electrode, and saturated calomel electrode (SCE) as a reference in 1.0 M KOH electrolyte] is used for the experiments. Nyquist plots at various overpotentials were also obtained and understood. It can be noticed that with the increase in potential, the curves from the Nyquist plots begin to transform from a straight line into a slightly semicircle for both 25 and 26. This is because a high level of potential results in a faster reaction, showing enhanced activity of CoS electrodes (i.e., electrocatalysts 25 and 26).40
The potentials of 33 and 34 have also been studied in electrocatalysis for the HER.43 When a reductive potential is applied at a rate of 1 mV per second, nanocatalyst 34 shows overpotential values of just 12 mV and 73 mV at the HER current densities of −10 and −100 mA cm−2, respectively. However, 33 shows the efficiency towards the HER with overpotential values of 107 and 229 mV at current densities of −10 and −100 mA cm−2, respectively. The values of Tafel slopes provide important information to enhance understanding and compare kinetics. This value is 71 mV dec−1 for 33 and 31 mV dec−1 for 34. Chronoamperometry has also been performed on 33 and 34 coated nickel foam electrodes. Moreover, 34 exhibits consistent stability over 24 hours at the HER overpotentials of 12 and 75 mV. However, 33 requires a greater overpotential (107 mV) to maintain a steady current density of −10 mA cm−2 for two days.43
In addition, 37 (i.e., Ag2S nanowires) exhibits strong HER activity in comparison to the bulk form of Ag2S.45 It is ultrathin in size. It has high electrocatalytic activity (Scheme 42), a highly reduced level of resistance, and significantly high durability for the HER process.45 At 10 mA cm−2, it has a substantially reduced overpotential of −88 mV, while bulk Ag2S has an overpotential of 242 mV (Scheme 42) at the same current density. Furthermore, 37 has a Tafel slope value of 52 mV dec−1, indicating a more effective HER mechanism than the bulk material (bulk counterpart) for which 141 mV dec−1 is the value of the Tafel slope. An electrochemical impedance spectroscopic (EIS) study has also been carried out. The results of this study reveal that 37 has a lower charge transfer resistance (51.4 Ω) than the bulk material (138.2 Ω), indicating greater electrocatalytic activity. When 37 is subjected to chronoamperometric studies, it is found to demonstrate outstanding stability, with a reduction of only 11% in starting current density after 12 hours. The electrochemically active surface area (ECSA) of 37 is relatively larger than the bulk Ag2S. This is likely to be the main reason for its enhanced electrocatalytic activity.45
 | ||
| Scheme 42 Electrocatalytic HER using nanocatalysts 25 and 37.40,45 | ||
5.2. Oxygen evolution reaction (OER)
24, 25 and 26 are interesting nanosized species for the oxygen evolution reaction (OER) (Table 1). This reaction involves the electrochemical process in which molecular oxygen is generated (Scheme 43) and evolved.The potential of nanocatalytic system 24 has been studied for the OER.39 It is deposited on a highly conducting nickel foam electrode. The value of the overpotential achieved using this material is only 293 mV (Scheme 44) to obtain a current density of 10 mA cm−1. In the past, CuO and Cu nanoparticles were synthesised and deposited on NF39 in the same manner as adopted in the case of 24. The values of Tafel slopes are used to evaluate the OER kinetics for 24/NF, CuO/NF, and Cu/NF, which are 67 ± 3 mV dec−1, 92 ± 4 mV dec−1, and 146 ± 3 mV dec−1, respectively. Tafel slope value for 24/NF is lower than that for CuO/NF and Cu/NF. This indicates that 24/NF has high ease for electron transport, favourable electrocatalytic reaction kinetics, and considerable catalytic activity towards the OER. In addition, 24/NF has a high ECSA (0.89 cm2), indicating the presence of more active OER sites, as well as a low charge transfer resistance. This is also an indication of the increased charge transport across the electrode/electrolyte and vice versa. Its long-term durability has been assessed using chronoamperometry at 1.55 V (vs. RHE) and maintained at a current density value ≥10 mA cm−2 for 12 hours, demonstrating the catalyst's intrinsic stability over a longer run.39
 | ||
| Scheme 44 Electrocatalytic OER using nanocatalysts 24, 25 and 26.39,40 | ||
Moreover, 25 and 26 exhibit the ability to carry out the oxygen evolution reaction (OER), with overpotentials of 307 mV and 276 mV, respectively.40 The values of Tafel slopes for 25 and 26 are 96 and 82 mV dec−1, respectively. It can be observed that the value of the Tafel slope for 26 is lesser than that of 25. This indicates greater electrocatalytic performance (Scheme 44) of 26 due to faster electron transfer and OER kinetics in this case. Electrochemical impedance spectroscopic (EIS) measurements at various potentials were also performed and studied using Nyquist plots. Additionally, 26 has a smaller semicircle diameter at 0.45 V. This also indicates a reduced level of charge transport resistance and a quick rate of electron transfer. As a result, enhanced electrocatalytic performance and quick electron transfer are observed in the case of 26, and it shows an improved electrocatalytic performance.40
5.3. Overall water splitting
27, 28, and 29 have nickel sulphide (NiS) nanoparticles, and they show electrocatalytic performance (for overall water splitting) for the oxygen evolution reaction (OER).41 When the polarisation curves are compared using linear sweep voltammetry (LSV) at a current density of 50 mA cm−2, it is noticed that 29 has the lowest overpotential of 371 mV, while the value of overpotentials for 27 and 28 is 389 mV. Thus, 29 also has superior electrochemical stability. Low potential variations of roughly 5 mV are present between the 1st and 1000th cycles of LSV at 50 mA cm−2. The combination of low overpotential and high electrochemical stability demonstrates that 29 is an excellent electrocatalyst for the oxygen evolution process. Similarly, the electrocatalytic activities of nickel sulphide samples for the hydrogen evolution processes (HERs) have been investigated and understood. To obtain a current density of 50 mA cm−2, 29 requires the lowest overpotential of 231 mV, while 27 and 28 have overpotentials of 273 mV and 259 mV, respectively. Interestingly, 29 outperforms the other samples in terms of efficiency for both processes, i.e., the OER and HER. It is worth noting that 27 (with the millerite phase) is the most efficient in terms of charge storage efficiency. As a result, it can be concluded that different phases may perform differently in energy generation and storage applications.41Nanocatalyst 30–32 has cobalt sulphide (CoS) nanoparticles. They have also been utilized for the study of overall water splitting processes.42 Their electrocatalytic activity for the OER is investigated, studied and understood using LSV. The polarisation curves demonstrate that 30 has the lowest overpotential value (325 mV) at a current density of 10 mA cm−2. They have quick and efficient reaction kinetics in electrocatalytic processes. The values of the Tafel slope for 30, 31, and 32 are 77 mV dec−1, 67 mV dec−1, and 77 mV dec−1, respectively. When the nanosized electrocatalyst has small particles, it has a large active surface area, leading to an improvement in electrochemical behaviour and characteristics. All the three species display stable current density during the 18-hour long-term stability test. Nanocatalyst 30–32 has also been investigated for the hydrogen evolution process (HER). Moreover, 30 is the most effective electrocatalyst for the HER reaction, with a Tafel slope of 126 mV dec−1 and the lowest overpotential of 200 mV at 10 mA cm−2. The size and surface area of its nanoparticles also have a direct and visible effect on HER efficiency. For both the OER and HER reactions, 30 exhibits better efficiency in comparison to 31 and 32. The studies reveal that 30 has a reduced level of over potential of 325 mV for the OER and 200 mV for the HER at a current density of 10 mA cm−2.42
6. Challenges and alternative approaches
Nanocatalytic systems developed utilizing organosulphur compounds offer the advantage of being recyclable and reusable for multiple cycles without any significant loss in catalytic activity. Advancements in this field can lead to the development of a comparatively stable, highly active, and efficient catalytic system, as the bonding of sulphur with metal is likely to create favourable electronic properties on the metal centre with a stable environment for catalytic reactions. Moreover, the scope of catalytic systems (developed using organosulphur compounds) encompasses a wide range of chemical transformations, such as Heck coupling, cross-dehydrogenative coupling (CDC), Suzuki coupling and O-arylation of phenols. There are several other chemical transformations to which the use of such nanocatalysts can be extended. In the field of electrocatalysis, although various noble and non-noble metal sulfides have been utilized, there is still a need for the development of novel electrocatalytic systems with improved activity for electrochemical reactions using organosulphur compounds. Therefore, diverse approaches can be followed for synthesising novel organosulphur compounds and for applying them in the development of nanocatalytic systems. The potential of such systems in various fields, such as catalysis and electrocatalysis, can be harnessed at a wider level.7. Future scope and conclusion
This article focuses on advancements related to the synthesis and employment of organosulphur compounds in the field of the development of nanocatalysts and electrocatalysts. Nowadays, nanocatalysis is a crucial aspect of both academic and industrial research and development. Nanocatalysts are obtained using organosulphur compounds using three distinct methods, specifically the single source precursor route, in situ generation during homogeneous catalysis of coupling reactions by preformed sulphur ligated palladium complexes, and the reduction method involving the utilization of organosulphur compounds as stabilizers. This article delves into the applications of nanocatalytic systems in catalyzing various chemical transformations, including Suzuki–Miyaura coupling, Heck coupling, Sonogashira coupling, O-arylation of phenol, and cross-dehydrogenative coupling (CDC). It also highlights their utilization in electrochemical applications, such as the hydrogen evolution reaction (HER), oxygen evolution reaction (OER), and overall water splitting. This article provides a critical analysis of these catalytic systems in terms of the advantages of their use. It also emphasizes the potential and scope for further extensive and in-depth studies on the design and application of such catalysts, highlighting the immense possibilities in the field of catalysis for various other transformations and electrocatalysis. It has been demonstrated that small-sized and highly dispersed nanoparticles are more efficient in catalysis. In the reduction method, when Na2PdCl4 is used as a precursor, it gives more uniformly dispersed nanoparticles than those obtained using Pd(OAc)2 and Pd(MeCN)2Cl2 as the source of palladium. Therefore, Na2PdCl4 is the better choice in terms of precursor for the development of palladium containing nanocatalysts using organosulphur compounds as the stabilizers and reduction process as the method. In the field of catalysis, the use of mainly palladium-based nanocatalysts has been discussed. There is a scope of development and use of other metal nanoparticles, such as Ni and Cu, using similar strategies. The development of such nanoparticles using the reduction method or the single source precursor (SSP) route would be interesting. In terms of electrocatalysis, although various non-noble metal-based nanoparticles have been utilized, Pt and Ag containing nanoparticles outperform other systems. Therefore, there is a gap that can be filled by developing such non-noble metal-based nanoparticles, which can perform better than or at par with noble metal-based catalysts.Conflicts of interest
There are no conflicts to declare.Data availability
No primary research results, software or code have been generated as a part of this perspective.Acknowledgements
A. T. acknowledges DST for the INSPIRE Fellowship [DST/INSPIRE Fellowship/2021/IF210531]. S. P. acknowledges DST for the INSPIRE Fellowship [DST/INSPIRE Fellowship/2021/IF210150]. A. K. acknowledges the support of DST under the PURSE scheme [file number: SR/PURSE/2023/199] and Anusandhan National Research Foundation (ANRF) for financial support under ANRF TARE (previously SERB TARE) with file number [TAR/2023/000169].References
- G. Pacchioni, Nat. Mater., 2009, 8, 167–168 CrossRef CAS PubMed.
- S. Malik, K. Muhammad and Y. Waheed, Molecules, 2023, 28, 661 CrossRef CAS.
- T. Kumar, R. K. Pandey, R. Kumar, C. V. Sudheep, S. Sreelakshmi, S. Awasthi, Vandana and R. Singhal, Historical Perspective of Nanotechnology and Functionalized Nanomaterials, in Functionalized Nanomaterials Based Supercapacitor: Design, Performance and Industrial Applications, ed. C. M. Hussain and M. B. Ahamed, Springer Nature, Cham, 2023, pp. 1–24 Search PubMed.
- G. Liden, Ann. Occup. Hyg., 2011, 55, 1–5 CAS.
- B. Zhou, R. Balee and R. Groenendaal, Nanotechol. Law Bus., 2005, 2, 222 Search PubMed.
- L. Jin, B. Liu, S. S. Duay and J. He, Catalysts, 2017, 7, 44 CrossRef.
- Z. Li, M. Li, Z. Bian, Y. Kathiraser and S. Kawi, Appl. Catal., B, 2016, 188, 324–341 CrossRef CAS.
- C. T. Campbell, Science, 2004, 306, 234–235 CrossRef CAS.
- M. Haruta, Chem. Rec., 2003, 3, 75–87 CrossRef CAS.
- Z. Ma and S. Dai, Nano Res., 2011, 4, 3–32 CrossRef CAS.
- S. A. C. Carabineiro, N. Bogdanchikova, M. Avalos-Borja, A. Pestryakov, P. B. Tavares and J. L. Figueiredo, Nano Res., 2011, 4, 180–193 CrossRef CAS.
- D. Astruc, F. Lu and J. R. Aranzaes, Angew. Chem., Int. Ed., 2005, 44, 7852–7872 CrossRef CAS.
- J. A. Widegren and R. G. Finke, J. Mol. Catal. A: Chem., 2003, 198, 317–341 CrossRef CAS.
- V. Polshettiwar and R. S. Varma, Green Chem., 2010, 12, 743–754 RSC.
- K. N. L. Hoang, S. M. McClain, S. M. Meyer, C. A. Jalomo, N. B. Forney and C. J. Murphy, Chem. Commun., 2022, 58, 9728–9741 RSC.
- K. An and G. A. Somorjai, ChemCatChem, 2012, 4, 1512–1524 CrossRef CAS.
- G. A. Somorjai, H. Frei and J. Y. Park, J. Am. Chem. Soc., 2009, 131, 16589–16605 CrossRef CAS PubMed.
- J. A. Gladysz, Pure Appl. Chem., 2001, 73, 1319–1324 CrossRef.
- A. Tyagi, N. Kunwar, S. Purohit, K. K. Pant and A. Kumar, Polyhedron, 2025, 277, 117569 CrossRef CAS.
- A. Tyagi, S. Purohit, P. Oswal, V. Negi, S. Rawat, N. Bhatt, P. Sharma, A. K. Singh and A. Kumar, Sulphur donor containing Schiff base ligands in Homogeneous catalysis: Synthesis, Characterization and Applications, in Homogeneous catalysis Concepts and Basics, ed. M. R. Rahimpour, M. A. Makarem, T. Roostaei and M. Meshksar, Elsevier, Amsterdam, 2024, pp. 93–125 Search PubMed.
- E. Tobler, Sulfur, Enslow Publishing, New York, 2018 Search PubMed.
- L. Lu, S. Zou and B. Fang, ACS Catal., 2021, 11, 6020–6058 CrossRef CAS.
- A. Heuer-Jungemann, N. Feliu, I. Bakaimi, M. Hamaly, A. Alkilany, I. Chakraborty, A. Masood, M. F. Casula, A. Kostopoulou, E. Oh, K. Susumu, M. H. Stewart, I. L. Menditz, E. Statakis, W. J. Parak and A. G. Kanaras, Chem. Rev., 2019, 119, 4819–4880 CrossRef CAS.
- L. M. Rossi, L. L. Vono, M. A. Garcia, T. L. Faria and J. A. Lopez-Sanchez, Top. Catal., 2013, 56, 1228–1238 CrossRef CAS.
- R. D. Neal, R. A. Hughes, P. Sapkota, S. Ptasinska and S. Neretina, ACS Catal., 2020, 10, 10040–10050 CrossRef CAS.
- L. Lu, H. Zheng, Y. Li, Y. Zhou and B. Fang, Chem. Eng. J., 2023, 451, 138668 CrossRef CAS.
- A. Fihri, M. Bouhrara, B. Nekoueishahraki, J. M. Basset and V. Polshettiwar, Chem. Soc. Rev., 2011, 40, 5181–5203 RSC.
- A. Roucoux, J. Schulz and H. Patin, Chem. Rev., 2002, 102, 3757–3778 CrossRef CAS.
- Y. A. Attia and S. H. Abdel-Hafez, New J. Chem., 2018, 42, 9606–9611 RSC.
- M. Zdražil, Catal. Today, 1988, 3, 269–365 CrossRef.
- X. Li, A. Sotto, J. Li and B. V. D. Bruggen, J. Membr. Sci., 2017, 524, 502–528 CrossRef CAS.
- C. J. Huang, L. C. Wang, C. Y. Liu, A. S. Chiang and Y. C. Chang, Biointerphases, 2014, 9, 029010 CrossRef CAS.
- K. Hiroi, Y. Suzuki, I. Abe, Y. Hasegawa and K. Suzuki, Tetrahedron: Asymmetry, 1998, 9, 3797–3817 CrossRef CAS.
- D. Zaramella, P. Scrimin and L. J. Prins, J. Am. Chem. Soc., 2012, 134, 8396 CrossRef CAS.
- V. K. Jain, Coord. Chem. Rev., 2021, 427, 213546 CrossRef CAS.
- A. Arora, P. Oswal, G. K. Rao, J. Kaushal, S. Kumar, A. K. Singh and A. Kumar, ChemistrySelect, 2019, 4, 10765 CrossRef CAS.
- P. Singh and A. K. Singh, Dalton Trans., 2017, 46, 10037–10049 RSC.
- S. Gupta, N. Chandna, A. K. Singh and N. Jain, J. Org. Chem., 2018, 83, 3226–3235 CrossRef CAS.
- B. Chakraborty, S. Kalra, R. Beltrán-Suito, C. Das, T. Hellmann, P. W. Menezes and M. Driess, Chem. – Asian J., 2020, 15, 852–859 CrossRef CAS.
- C. Gervas, M. D. Khan, S. Mlowe, C. Zhang, C. Zhao, R. K. Gupta, M. P. Akerman, P. Mashazi, T. Nyokong and N. Revaprasadu, ChemElectroChem, 2019, 6, 2560–2569 CrossRef CAS.
- G. E. Ayom, M. D. Khan, T. Ingsel, W. Lin, R. K. Gupta, S. J. Zamisa, W. E. Zyl and N. Revaprasadu, Chem. – Eur. J., 2020, 26, 2693–2704 CrossRef CAS.
- R. Akram, M. D. Khan, C. Zequine, C. Zhao, R. K. Gupta, M. Akhtar, J. Akhtar, M. A. Malik, N. Revaprasadu and M. H. Bhatti, Mater. Sci. Semicond. Process., 2020, 109, 104925 CrossRef CAS.
- C. Panda, P. W. Menezes, S. Yao, J. Schmidt, C. Walter, J. N. Hausmann and M. Driess, J. Am. Chem. Soc., 2019, 141, 13306–13310 CrossRef CAS.
- M. A. Agoro, J. Z. Mbese and E. L. Meyer, Molecules, 2020, 25, 1919 CrossRef CAS PubMed.
- S. Khirid, R. Biswas, S. Meena, R. A. Patil, Y.-R. Ma, R. S. Devan, R. S. Dhayal and K. K. Haldar, ChemistrySelect, 2020, 5, 10593–10598 CrossRef CAS.
- F. Saleem, G. K. Rao, A. Kumar, S. Kumar, M. P. Singh and A. K. Singh, RSC Adv., 2014, 4, 56102–56111 RSC.
- S. Kumar, G. K. Rao, A. Kumar, M. P. Singh and A. K. Singh, Dalton Trans., 2013, 42, 16939–16948 RSC.
- G. K. Rao, A. Kumar, F. Saleem, M. P. Singh, S. Kumar, B. Kumar, G. Mukherjee and A. K. Singh, Dalton Trans., 2015, 44, 6600–6612 RSC.
- R. Bhaskar, A. K. Sharma, M. K. Yadav and A. K. Singh, Dalton Trans., 2017, 46, 15235–15248 RSC.
- F. Ortloff, J. Bohnau, U. Kramar, F. Graf and T. Kolb, Appl. Catal., B, 2016, 182, 550–561 CrossRef CAS.
- I. J. Ferrer, P. Diaz-Chao, A. Pascual and C. Sánchez, Thin Solid Films, 2007, 515, 5783–5786 CrossRef CAS.
- W. Xu, J. Ni, Q. F. Zhang, F. Feng, Y. Z. Xiang and X. N. Li, J. Mater. Chem. A, 2013, 1, 12811–12817 RSC.
- Q. F. Zhang, W. Xu, X. N. Li, D. H. Jiang, Y. Z. Xiang, J. G. Wang, J. Cen, S. Romano and J. Ni, Appl. Catal., A, 2015, 497, 17–21 CrossRef CAS.
- A. J. Mccue, A. Guerrero-Ruiz, I. Rodríguez-Ramos and J. A. Anderson, J. Catal., 2016, 340, 10–16 CrossRef CAS.
- F. Menegazzo, P. Canton, F. Pinna and N. Pernicone, Catal. Commun., 2008, 9, 2353–2356 CrossRef CAS.
- A. J. Mccue and J. A. Anderson, Cheminform, 2014, 4, 272–294 CAS.
- K. N. Sharma, H. Joshi, V. V. Singh, P. Singh and A. K. Singh, Dalton Trans., 2013, 42, 3908–3918 RSC.
- F. Grønvold and E. Røst, Acta Crystallogr., 1962, 15, 11–13 CrossRef.
- L. L. Long, A. Y. Zhang, Y. X. Huang, X. Zhang and H. Q. Yu, J. Mater. Chem. A, 2015, 3, 4301–4306 RSC.
- H. Fischmeister, Acta Chem. Scand., 1959, 13, 852–853 CrossRef CAS.
- B. D. Morreale, B. H. Howard, O. Iyoha, R. M. Enick, C. Ling and D. S. Sholl, Ind. Eng. Chem. Res., 2007, 46, 6313–6319 CrossRef CAS.
- C. P. O'Brien, A. J. Gellman, B. D. Morreale and J. B. Miller, J. Membr. Sci., 2011, 371, 263–267 CrossRef.
- D. Albani, M. Shahrokhi, Z. Chen, S. Mitchell, R. Hauert, N. López and J. Pérez-Ramírez, Nat. Commun., 2018, 9, 2634 CrossRef PubMed.
- W. Jing, S. Mo, W. Zhang, W. Zhou, K. Liu, J. Wei, R. Qin and N. Zheng, Precis. Chem., 2024, 2, 200–207 CrossRef CAS.
- V. V. Singh, U. Kumar, S. N. Tripathi and A. K. Singh, Dalton Trans., 2014, 43, 12555–12563 RSC.
- W. Wang, D. Zhao, J. Yu, L. Shi, Y. Wang and H. Chen, Nano Res., 2023, 16, 2597–2603 CrossRef.
- Y. Xu, K. Ren, T. Ren, M. Wang, S. Yu, Z. Wang, X. Li, L. Wang and H. Wang, J. Mater. Chem. A, 2020, 8, 19873–19878 RSC.
- L. Ma, S. Yuan, H. Zhu, T. Jiang, X. Zhu, C. Lu and X. Li, Catalysts, 2019, 9, 410 CrossRef CAS.
- M. Bochmann, Chem. Vap. Deposition, 1996, 2, 85–96 CrossRef CAS.
- V. K. Jain, J. Chem. Sci., 2006, 118, 547–552 CrossRef CAS.
- A. A. Tedstone, A. B. Jumah, E. Asuquo and A. A. Garforth, R. Soc. Open Sci., 2022, 9, 211353 CrossRef CAS PubMed.
- S. Shen, Y. Zhang, L. Peng, B. Xu, Y. Du, M. Deng, H. Xu and Q. Wang, CrystEngComm, 2011, 13, 4572–4579 RSC.
- P. Oswal, A. Arora, S. Singh, G. K. Rao, S. Kumar, A. K. Singh and A. Kumar, Catal. Commun., 2021, 149, 106242 CrossRef CAS.
- S. B. Somvanshi, S. R. Patade, D. D. Andhare, S. A. Jadhav, M. V. Khedkar, P. B. Kharat, P. P. Khirade and K. Jadhav, J. Alloys Compd., 2020, 835, 155422 CrossRef CAS.
- S. B. Somvanshi, S. A. Jadhav, M. V. Khedkar, P. B. Kharat, S. More and K. Jadhav, Ceram. Int., 2020, 46, 13170–13179 CrossRef CAS.
- S. B. Somvanshi, P. B. Kharat, T. S. Saraf, S. B. Somwanshi, S. B. Shejul and K. M. Jadhav, Mater. Res. Innovations, 2020, 25, 169–174 CrossRef.
- A. Bhosale, S. B. Somvanshi, V. Murumkar and K. Jadhav, Ceram. Int., 2020, 46, 15372–15378 CrossRef CAS.
- S. B. Somvanshi, P. B. Kharat, M. V. Khedkar and K. Jadhav, Ceram. Int., 2020, 46, 7642–7653 CrossRef CAS.
- S. B. Somvanshi, M. V. Khedkar, P. B. Kharat and K. Jadhav, Ceram. Int., 2020, 46, 8640–8650 CrossRef CAS.
- J. A. Darr, J. Zhang, N. M. Makwana and X. Weng, Chem. Rev., 2017, 117, 11125–11238 CrossRef CAS PubMed.
- Y. Guo, Z. Wang, H. Shao and X. Jiang, Carbon, 2013, 52, 583–589 CrossRef CAS.
- M. Zhu, Y. Wang, D. Meng, X. Qin and G. Diao, J. Phys. Chem. C, 2012, 116, 16276–16285 CrossRef CAS.
- A. K. Ganguli, T. Ahmad, S. Vaidya and J. Ahmed, Pure Appl. Chem., 2008, 80, 2451–2477 CrossRef CAS.
- M. López-Quintela, J. Rivas, M. Blanco and C. Tojo, Synthesis of nanoparticles in microemulsions, in Nanoscale materials, Springer, Berlin, 2004, pp. 135–155 Search PubMed.
- S. R. Patade, D. D. Andhare, S. B. Somvanshi, S. A. Jadhav, M. V. Khedkar and K. Jadhav, Ceram. Int., 2020, 46, 25576–25583 CrossRef CAS.
- P. B. Kharat, S. More, S. B. Somvanshi and K. Jadhav, J. Mater. Sci.: Mater. Electron., 2019, 30, 6564–6574 CrossRef CAS.
- S. B. Somvanshi, R. V. Kumar, J. S. Kounsalye, T. S. Saraf and K. Jadhav, J. Phys.: Conf. Ser., 2020, 1644, 012046 CrossRef.
- A. R. Chavan, S. B. Somvanshi, P. P. Khirade and K. Jadhav, RSC Adv., 2020, 10, 25143–25154 RSC.
- H. Kardile, S. B. Somvanshi, A. R. Chavan, A. Pandit and K. Jadhav, Optik, 2020, 207, 164462 CrossRef CAS.
- C. Q. Sun, Prog. Solid State Chem., 2007, 35, 1–159 CrossRef CAS.
- L. Pan, M. Gu, G. Ouyang and C. Q. Sun, Key Eng. Mater., 2010, 444, 17–45 CAS.
- R. Ouyang, J.-X. Liu and W.-X. Li, J. Am. Chem. Soc., 2013, 135, 1760 CrossRef CAS PubMed.
- F. Gambinossi, S. E. Mylon and J. K. Ferri, Adv. Colloid Interface Sci., 2015, 222, 332–349 CrossRef CAS PubMed.
- L. M. Rossi, J. L. Fiorio, M. A. S. Garcia and C. P. Ferraz, Dalton Trans., 2018, 47, 5889–5915 RSC.
- P. Sharma, A. Arora, P. Oswal, G. K. Rao, J. Kaushal, S. Kumar, S. Kumar, M. P. Singh, A. K. Singh and A. Kumar, Polyhedron, 2019, 171, 120–127 CrossRef CAS.
- P. Oswal, A. Arora, J. Kaushal, G. K. Rao, S. Kumar, A. K. Singh and A. Kumar, RSC Adv., 2019, 9, 22313–22319 RSC.
- S. Kumar, G. K. Rao, A. Kumar, M. P. Singh, F. Saleem and A. K. Singh, RSC Adv., 2015, 5, 20081–20089 RSC.
- K. Said, A. Mesni and R. B. Salem, Lett. Org. Chem., 2020, 17, 36–45 CrossRef CAS.
- P. Devendar, R.-Y. Qu, W.-M. Kang, B. He and G.-F. Yang, J. Agric. Food Chem., 2018, 66, 8914 CrossRef CAS PubMed.
- A. T. K. Koshvandi, M. M. Heravi and T. Momeni, Appl. Organomet. Chem., 2018, 32, e4210 CrossRef.
- S. Jagtap, Catalysts, 2017, 7, 267 CrossRef.
- Y.-H. Ding, H.-X. Fan, J. Long, Q. Zhan and Y. Chen, Bioorg. Med. Chem. Lett., 2013, 23, 6087 CrossRef CAS PubMed.
- J. G. D. Vries, Can. J. Chem., 2001, 79, 1086 CrossRef.
- P. Oswal, A. Arora, S. Gairola, A. Datta and A. Kumar, New J. Chem., 2021, 45, 21449–21487 RSC.
- M. Karak, L. C. A. Barbosa and G. C. Hargaden, RSC Adv., 2014, 4, 53442 RSC.
- K. Nejati, S. Ahmadi, M. Nikpassand, P. D. K. Nezhada and E. Vessally, RSC Adv., 2018, 8, 19125 RSC.
- L. Lv and Z. Li, Fe-Catalyzed Cross-Dehydrogenative Coupling Reactions. Ni- and Fe-Based Cross-Coupling Reactions, in Ni- and Fe- Based Cross-Coupling Reactions, ed. A. Correa, Springer, Cham, 2016, pp. 225–263 Search PubMed.
- C. J. Li, Acc. Chem. Res., 2009, 42, 335–344 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2025 |