Open Access Article
Open Access ArticleLanthanide complexes bearing a bioinspired CuII binding site with picomolar affinity: synthesis, structural, relaxometric and luminescence studies
Katharina
Zimmeter
a,
Bertrand
Vileno
 a,
Agnès
Pallier
b,
Carlos
Platas-Iglesias
a,
Agnès
Pallier
b,
Carlos
Platas-Iglesias
 c,
Peter
Faller
c,
Peter
Faller
 ad,
Célia S.
Bonnet
*b and
Angélique
Sour
ad,
Célia S.
Bonnet
*b and
Angélique
Sour
 *a
*a
aInstitut de Chimie (UMR 7177), Université de Strasbourg – CNRS, 4 Rue Blaise Pascal, 67070 Strasbourg, France. E-mail: a.sour@unistra.fr
bCentre de Biophysique Moléculaire, CNRS, Université d'Orléans, Rue Charles Sadron, 45071 Orléans, France. E-mail: celia.bonnet@cnrs-orleans.fr
cDepartamento de Química Fundamental, Universidade da Coruña, Campus da Zapateira, Rúa da Fraga 10, 15008 A Coruña, Spain
dInstitut Universitaire de France (IUF), 1 rue Descartes, 75231 Paris, France
First published on 29th September 2025
Abstract
In the pursuit of CuII-responsive MRI contrast agents with enhanced CuII affinity, we report the synthesis and characterization of two LnIII-DO3A-C3AmpicGH complexes (LnIII = GdIII, EuIII), featuring a tetradentate CuII-binding moiety, along with the corresponding mononuclear CuII-C3AmpicGH complex. The chelator coordinates CuII through two amide groups, a pyridine and an imidazole ring. The use of a propyl-amide linker between the LnIII- and CuII-binding moieties allowed an increased separation between the two metal centers. As a result, both LnIII–CuII-DO3A-C3AmpicGH and CuII-C3AmpicGH complexes exhibit the same CuII-affinity (log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) K = 11–12 at pH 7.4) and good selectivity over competing physiological metal ions, particularly ZnII (up to at least 500 equivalents). While the GdIII complex displayed no relaxivity changes upon CuII binding, the EuIII analogue showed a luminescence turn-off response. Spectroscopic analyses as well as density functional theory (DFT) calculations provide insights into the coordination environments of both metal ions, notably confirming the absence of amide coordination to the LnIII center, even in absence of CuII.
K = 11–12 at pH 7.4) and good selectivity over competing physiological metal ions, particularly ZnII (up to at least 500 equivalents). While the GdIII complex displayed no relaxivity changes upon CuII binding, the EuIII analogue showed a luminescence turn-off response. Spectroscopic analyses as well as density functional theory (DFT) calculations provide insights into the coordination environments of both metal ions, notably confirming the absence of amide coordination to the LnIII center, even in absence of CuII.
1. Introduction
Copper is an essential trace metal ion, required for various physiological processes in the human body, such as electron transfer and redox catalysis.1,2 An imbalance in copper ions—whether an excess or a deficiency—has been linked to various diseases, highlighting the critical importance of precisely regulating copper homeostasis in the body. In the extracellular medium, Cu ions are mainly in the oxidized CuII state and are never found as “free ions”, but are always bound to a carrier, which can be a protein, peptide or small molecule. Depending on the nature of the complex, this binding is either kinetically inert, such as Cu bound to the ferroxidase ceruloplasmin in the blood,3 or more labile, forming the “exchangeable copper (Cuexc) pool”. Cuexc is mainly bound to human serum albumin (HSA) in the blood, with an conditional association constant of log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Ka = 13 at pH 7.4.4,5 The exchangeable Cu pool is also present in extracellular fluids of the brain, particularly in the case of certain diseases. CuII can be bound to amyloid beta (Aβ) peptides with picomolar affinities (log
Ka = 13 at pH 7.4.4,5 The exchangeable Cu pool is also present in extracellular fluids of the brain, particularly in the case of certain diseases. CuII can be bound to amyloid beta (Aβ) peptides with picomolar affinities (log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Ka = 10 at pH 7.4 for Aβ1–16) in the case of Alzheimer's disease (AD).6–8 In the case of Parkinson's disease, Cu is considered to bind to the protein alpha synuclein (α-Syn), although this interaction is still discussed. In test tubes, experiments revealed two distinct CuII sites in α-Syn with nM and μM affinities respectively (site 1: α-Syn1–17: Ka = 0.94 × 10−9, site 2: α-Syn46–50: Ka = 1.5 × 10−6).9–11
Ka = 10 at pH 7.4 for Aβ1–16) in the case of Alzheimer's disease (AD).6–8 In the case of Parkinson's disease, Cu is considered to bind to the protein alpha synuclein (α-Syn), although this interaction is still discussed. In test tubes, experiments revealed two distinct CuII sites in α-Syn with nM and μM affinities respectively (site 1: α-Syn1–17: Ka = 0.94 × 10−9, site 2: α-Syn46–50: Ka = 1.5 × 10−6).9–11
It has been shown that the quantification of exchangeable copper in the blood has clinical significance for the diagnosis of Wilson's disease and a type of AD.12–14 Monitoring this CuII pool could also bring important information for a better understanding of the Cu metabolism. Consequently, the development of Cu-responsive sensors associated to imaging modalities has attracted significant attention. In the literature, CuII-responsive sensors reported so far rely on luminescence-based techniques, mostly involving quenching of the emission intensity upon interaction with paramagnetic CuII ions. In contrast, only a few examples are reported with the use of magnetic resonance imaging (MRI) as a detection method.15,16
MRI is a powerful technique that provides images with no depth limitation, high spatial resolution and allows real-time monitoring of dynamic processes. Contrast agents (CAs) based on gadolinium(III) complexes are commonly used to enhance image contrast. Upon interaction of the GdIII ion with surrounding water molecules, the relaxation rate of water protons is increased, resulting in an improved contrast. Relaxivity values define the efficiency of a CA to increase the relaxation rate (r1 for the longitudinal relaxation rate) of surrounding water protons.17–19 Cation-responsive CAs can be rationally engineered through the incorporation of an ion-binding site, and the change in relaxivity upon cation binding should be significantly pronounced to enable the detection of cations at low concentrations. This is generally achieved through a change in the number of GdIII-bound water molecules q and/or through a modification of the rotational correlation time τR of the complex upon cation interaction.16,20–24
Besides the relaxivity change upon metal binding, the CA should present an affinity tuned to that of the physiological Cuexc pool and its surrounding biochemical environment. This affinity should be sufficient to enable competitive extraction of CuII from its carrier, but not too strong to avoid CuII depletion. Typically, the CA should be able to retrieve CuII bound to HSA in the blood, or bound to Aβ- or α-Syn in the brain. Moreover, the CA should also show good selectivity for CuII over other metal ions, notably over zinc, more abundant in the body and more readily available.25–27 Finally, the kinetics of CuII capture and release should ideally match the temporal window of MRI acquisition, although achieving precise control over this parameter remains a challenge.
Fulfilling all these requirements is a real challenge, and since the first example of CuII-responsive CA was reported nearly twenty years ago,28 none of the complexes developed fulfill all the criteria. It should be however noted that one GdIII complex based on a τR change through an interaction with human serum albumin (HSA) was successfully used to detect CuII in the liver,29 although the selectivity vs. ZnII is not ideal.
In order to attain high relaxivity changes, a change in the hydration number of GdIII from 0 to 2 is highly desirable. We have previously developed a series of Gd-DO3A-based complexes linked to a pyridine-based switching arm, bearing a CuII binding site bioinspired by the N-terminal CuII binding motif of HSA (ATCUN).30,31 An unprecedented relaxivity increase of ca. 400% could be achieved with the compound Gd-DO3A-pyrGH (Fig. 1).30 Importantly, due to its bioinspired nature, this system is highly selective for CuII over ZnII. Unfortunately, the CuII affinity of the bioinspired binding site is drastically reduced when it is tethered to the GdIII complex, with an apparent binding constant (at pH 7.4) dropping from log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Kapp = 16 for the free binding site to log
Kapp = 16 for the free binding site to log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Kapp = 5.6 in the conjugated system.30,32 This is explained by the distortion of the CuII coordination sphere due to the proximity of the bulky GdDO3A moiety, along with possible electrostatic repulsion between CuII and GdIII ions and by the energy cost to switch the pyridine moiety from GdIII to CuII.
Kapp = 5.6 in the conjugated system.30,32 This is explained by the distortion of the CuII coordination sphere due to the proximity of the bulky GdDO3A moiety, along with possible electrostatic repulsion between CuII and GdIII ions and by the energy cost to switch the pyridine moiety from GdIII to CuII.
Therefore, one route to increase the affinity of the GdIII-based sensor for CuII is to increase the distance between the GdIII and CuII complexing moieties. Alkyl-amide groups have already been successfully used for ZnII and CaII detection, triggering an increase in the hydration number of the LnIII upon cation binding.33–35 It has been shown that the length of the linker, ethyl or propyl, plays a crucial role in the hydration number of the LnIII ion, being 1 and 0, respectively.35,36 For the propyl linker, the overall charge of the complex,37,38 as well as the steric hindrance of the group linked to the amide function are also important.39
This has prompted us to design GdIII-DO3A-C3AmpicH (Scheme 1) containing a Gd-DO3A subunit (DO3A: 1,4,7,10-tetraazacyclododecane-1,4,7-triacetate), a bioinspired CuII binding site, and a propyl amide function between the two. The bioinspired CuII binding site C3AmpicH is derived from previously studied ligands which are inspired by the ATCUN site of HSA, and which show ideal CuII affinity and selectivity versus ZnII.4,26,40 Here, we describe the synthesis of the compounds C3AmpicH and LnIII-DO3A-C3AmpicH, and we evaluate their properties by spectroscopy, relaxivity, and DFT calculations, both in the absence and in the presence of CuII.
 | ||
| Scheme 1 Chemical structure of the previously studied sensor Ln-DO3A-pyrGH, the new complexes Ln-DO3A-C3AmpicH (with Ln = GdIII, EuIII) and the CuII-binding ligand C3AmpicH. | ||
2. Results and discussion
2.1 Design and synthesis of DO3A-C3AmpicH and C3AmpicH
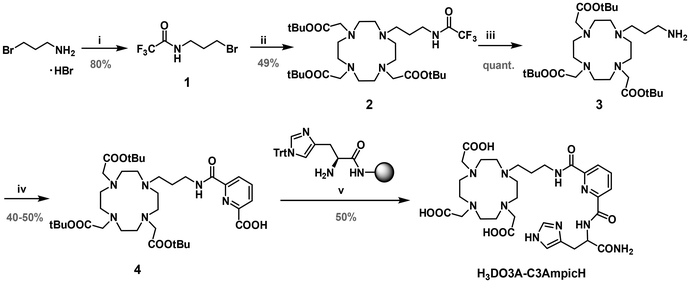
The ditopic ligand was obtained using a combination of solution and solid phase synthesis (SPS), as shown in Scheme 2. The DO3A subunit was linked to a protected 3-bromopropyl-amine chain 1 to give the protected compound 2. Deprotection of the amine under basic conditions afforded compound 3, which was then coupled with the monoactivated pyridine-2,6-dicarboxylic acid. The resulting compound 4 was then linked to a trityl (Trt)-protected histidine (His) residue on a Rink amide resin and the ligand H3DO3A-C3AmpicH was obtained after deprotection and cleavage from the resin.The CuII-binding ligand C3AmpicH was obtained by SPS on a Rink amide resin (section 1 of the SI). The His(Trt) residue was first attached to the resin, followed by the coupling of the monoactivated pyridine-2,6-dicarboxylic acid. The second carboxylic group was activated directly on the resin and coupled to propylamine and the ligand C3AmpicH was obtained after cleavage from the resin and removal of the trityl protecting group.
The final ligands were purified by preparative HPLC and characterized by 1D and 2D NMR and HR-ESI-MS (section 1 of the SI).
2.2 Relaxivity and luminescence responses of GdIII/EuIII-DO3A-C3AmpicH to CuII
The detection of CuII by GdIII-DO3A-C3AmpicH has been explored by relaxivity (r1) measurements in HEPES buffer (pH 7.4) at 60 MHz. As evidenced in Fig. 1A, no relaxivity change upon CuII coordination is observed. This can be explained by the absence of amide coordination to GdIII even in absence of CuII, which is in line with the high relaxivity values, and the DFT calculations (vide infra).The EuIII analogue EuIII-DO3A-C3AmpicH was also used to follow CuII binding by luminescence (Fig. 1B and C). It is well-known that a pyridine can sensitize EuIII luminescence41,42 and indeed, despite the distance between the EuIII ion and the pyridine moiety, it is also the case here. In the absence of CuII, the characteristic pattern of EuIII luminescence with emission maxima at 588 nm, 615 nm, 687 nm and 695 nm was observed. Upon addition of CuII, a linear luminescence decrease was observed and the complete quenching was obtained at a molar ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 (Cu
1 (Cu![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ligand). This turn-off luminescence response evidences CuII coordination to the LnIII complex. The sharp inflection point observed at 1 equiv. of added CuII indicates a strong binding.
ligand). This turn-off luminescence response evidences CuII coordination to the LnIII complex. The sharp inflection point observed at 1 equiv. of added CuII indicates a strong binding.
In order to understand this behavior, we investigated the CuII coordination mode, as well as the LnIII coordination sphere, both in the absence and presence of CuII, using electron paramagnetic resonance (EPR), luminescence and relaxivity measurements complemented by DFT calculations.
2.3 LnIII-coordination mode in the complexes LnIII-DO3A-C3AmpicH
First, the structure of LnIII-DO3A-C3AmpicH was investigated. Luminescence lifetime measurements were performed on EuIII-DO3A-C3AmpicH in H2O and D2O to assess the hydration state of the LnIII ion (Table S1 and Fig. S15). In H2O, the luminescence decays were best fitted by bi-exponential decay pointing to the presence of at least two species in solution with q = 0.1 and 1.7. The apparent predominance of the low-hydration species is however amplified, as the luminescence signal is weighted by the quantum yield of each species. The value of 1.7 is close to that of Eu-DO3A (q = 1.9) where the existence of an equilibrium strongly shifted towards the bis-hydrated species has been evidenced using temperature-dependent UV-Vis spectrophotometry.43 We certainly have the same phenomenon for EuIII-DO3A-C3AmpicH, which would be consistent with the relaxivity values observed (vide infra), and DFT calculations (vide infra).The NMR spectra of the corresponding diamagnetic YIII complex (Fig. S16) supports: (1) the presence of two species in solution in slow equilibrium at the NMR time scale in a ratio 85![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 15; and (2) the lack of coordination of the amide group to YIII as only broad signals for the macrocycle are observed, a fluxional behavior that is characteristic of complexes with flexible DO3A-type ligands. Altogether, this shows that there are several species in solution and the main species is bis-hydrated with no amide coordination.
15; and (2) the lack of coordination of the amide group to YIII as only broad signals for the macrocycle are observed, a fluxional behavior that is characteristic of complexes with flexible DO3A-type ligands. Altogether, this shows that there are several species in solution and the main species is bis-hydrated with no amide coordination.
This is also supported by DFT calculations (Fig. 2), which were used to model the mono-hydrated structure of the complex with the amide group coordinated to the metal ion. Subsequently, we explored the potential energy surface by increasing the Gd–Oamide distance, which generated a second energy minimum. Compared to the monohydrated LnIII structure with amide coordination, a difference of free energy of +7.9 kJ mol−1 is found. Inspection of the calculated structures shows that the angle formed between the planes of the pyridyl and amide groups increases from 33° to 43° upon amide coordination. Typically, aromatic secondary amides are bent out of the plane of aromatic unit by ∼30°, and thus amide coordination appears to be hindered by the presence of the pyridyl-amide group in the vicinity of the DO3A unit.44 Moreover, the inspection of the enthalpy and entropy contributions obtained with DFT shows that at 298 K the coordination of the amide group is disfavoured by an important entropy contribution of −TΔS = 12.8 kJ mol−1, which compensates the negative enthalpy contribution associated to amide coordination (ΔH = −4.9 kJ mol−1). Thus, amide coordination appears to be prevented by the loss of conformational entropy if it binds to the Ln ion.
This is surprising, as it contrasts with previous observations for a LnIII-DO3A complex bearing a propylamide arm linked to a pyridine, which was found to be non-hydrated.36 This is also in contrast with many propylamide systems developed so far that are either mono or non-hydrated. In our case, the amide is part of an extended aromatic system with a second amide substituted to the pyridine, which probably leads to increased steric hindrance, as reflected in the increase upon amide coordination of the dihedral angle involving the secondary amide and the plane of the pyridyl ring; loss of electronic effects, and rigidity. It shows that the steric hindrance, which remains difficult to evaluate, is important.
The nuclear magnetic resonance dispersion (NMRD) profiles of the corresponding GdIII complex were recorded at pH 7.3 (in HEPES buffer) between 40 kHz and 400 MHz at 25 °C and 37 °C (Fig. 2). As expected for small molecular complexes, the relaxivity decreases with increasing temperature. At 25 °C, 60 MHz, the relaxivity is quite high (r1 = 9.26 mM−1 s−1), which is consistent with a DO3A-type GdIII complex bearing two water molecules in the first coordination sphere.
The NMRD profiles were fitted to the Solomon Bloembergen and Morgan theory to gain access to the microscopic parameters of the complex that affect the observed relaxivity (see SI for equations and details). The values of the water exchange rate, its activation energy and the hydration number (q = 1.9) were set to those of GdDO3A43 due to the similar coordination sphere. Fig. 3 shows the experimental data and the best-fit curves, the parameters of which are reported in Table S2. The rotational correlation time was determined to be 122 ps, which is consistent with the size of the complex. We also checked that the decrease of the hydration number from 1.9 to 1.7 did not affect significantly the rotational correlation time.
 | ||
Fig. 3
1H NMRD profiles of Gd-DO3A-C3AmpicH (1.68 mM) at pH 7.3 (HEPES 0.1 M) at 37 °C ( ), and 25 °C ( ), and 25 °C ( ). The curves represent the best fit to the SBM theory with the parameters presented in Table S2. ). The curves represent the best fit to the SBM theory with the parameters presented in Table S2. | ||
2.4 CuII-coordination mode in the complexes EuIII–CuII-DO3A-C3AmpicH and CuII-C3AmpicH
The CuII coordination sphere was determined in both Cu-C3AmpicH and Eu–Cu-DO3A-C3AmpicH complexes. The CuII titrations were performed in HEPES buffer at pH 7.4 and followed by UV-visible spectroscopy (Fig. S17 and S19). Upon CuII addition, a broad d–d absorption band appeared, centered at 595 and 600 nm for Eu–Cu-DO3A-C3AmpicH and Cu-C3AmpicH, respectively. This indicates the formation of a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 Cu
1 Cu![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ligand complex, which is in accordance with luminescence titrations (Fig. 1B).
ligand complex, which is in accordance with luminescence titrations (Fig. 1B).
It is noteworthy that the d–d bands are more red-shifted than expected for the coordination to two amidates and two aromatic sp2 nitrogen atoms.45 Thus, we investigated the pH dependence of the CuII-coordination sphere of EuIII–CuII-DO3A-C3AmpicH and CuII-C3AmpicH by UV-Vis absorption spectroscopy and EPR. A hypsochromic shift of the absorption maximum of the d–d band was observed for both CuII-complexes from 630 nm (at pH below 6) to 580 nm (at pH above pH 8) (Fig. 4A and S20, S21). This shift can be attributed to a change in the coordination of CuII due to the deprotonation of the terminal amide when increasing the pH. Such pH-dependent behavior has previously been reported in the literature.46 At pH below 6, the CuII ion is linked to the oxygen of the terminal amide, then upon increasing the pH, the amidate is gradually formed and the CuII ion binds to the nitrogen of the amidate. At pH 7.4, both coordination modes are simultaneously present in solution, in line with a broad d–d band around 600 nm.
 | ||
| Fig. 4 pH-dependence of the CuII-binding to EuIII-DO3A-C3AmpicH. (A) UV-Vis: hypsochromic shift of the absorption maximum of the d–d band upon basification. Conditions: 100 μM EuIII-DO3A-C3AmpicH, 90 μM CuCl2, in H2O, addition of aqueous NaOH, room temperature. (B) EPR: conditions 600 μM EuIII-DO3A-C3AmpicH, 500 μM CuCl2, 75 mM HEPES pH 7.4 or CHES pH 10, 10% glycerol, 100 K. The indicated pH values have been measured on the pH-meter after the addition of glycerol and thus differ slightly from the pH values of the used buffer solutions. Inset: Peisach–Blumberg plot indicating the CuII-complex coordination sphere as a function of the values of A∥and g∥ as well as the overall charge of the molecule.48 (C) Structures showing the proposed CuII-coordination modes. | ||
To strengthen this hypothesis, low-temperature (100 K) EPR characterization of these complexes was performed at pH 7.0 and 9.4 (Fig. 4B and Table 1). EPR spectra evidenced the presence of two different coordination modes at pH 7.0 and 9.4, in line with a change from a neutral O to a negatively charged N. At this low temperature one coordination mode only is seen at each pH. At pH 7.0, the EPR spectrum suggests a CuII-coordination sphere composed of three nitrogens and one oxygen atom, and at pH 9.4, a 4N binding mode is highly probable. This is consistent with the UV-Vis absorption data (Fig. 4A), although the pH range in which the coordination change occurs was shifted, which may be due to the temperature difference between the two techniques. The g-tensors observed for the two species (g∥ > g⊥; g⊥ > 2.035) are characteristic of square-planar coordination with a dx2−y2 ground state.47
To further support this pH-dependent coordination mode, DFT calculations were performed for the CuII-C3AmpicH complex and the resulting structures are shown in Fig. S22. The EPR parameters obtained are in reasonable agreement with the experimental data, in particular when considering the difficulties associated to the calculation of EPR parameters in CuII complexes (Table 1 and Table S3).49,50 In particular, the deprotonation in the complex results in a 4N coordination environment that is characterized by a higher A∥ and a lower g∥ value than those obtained for the complex with 3N1O coordination (Table 1). This is in line with the changes expected for these coordination environments according to Peisach, as 3N1O coordination is generally characterized by higher g∥ and lower A∥ values than 4N coordination, an effect that is magnified by the increased positive charge of the 3N1O complex. We note that the experimental values of g∥/A∥, which are diagnostic of the coordination environment, show a very nice agreement with the DFT values (Table 1).
2.5 Estimation of the CuII-affinity of EuIII-DO3A-C3AmpicH and C3AmpicH
The affinity of EuIII-DO3A-C3AmpicH and C3Ampyr for CuII was evaluated by competition experiments with protein or peptides of biological relevance (HSA, Aβ1–16 and α-Syn1–6). These competition experiments were followed by UV-Vis absorption, circular dichroism and luminescence spectroscopies.The competition experiments between EuIII-DO3A-C3AmpicH and HSA for CuII, were first followed by UV-vis absorption experiments, monitoring the d–d bands of the CuII complexes. Measurements were performed both by addition of EuIII-DO3A-C3AmpicH to the equimolar CuII-HSA complex and by addition of the complex EuIII–CuII-DO3A-C3AmpicH to equimolar HSA (Fig. 5A and S23). Both CuII exchange reactions lead to the same end-point with a maximum absorbance at 535 nm, suggesting the coexistence of the complexes CuII-HSA (d–d band maximum at 530 nm) and EuIII–CuII-DO3A-C3AmpicH (d–d band maximum at 595 nm) at the equilibrium state. This also indicates that only a small fraction of CuII could be retrieved from HSA, which is in coherence with circular dichroism experiments (Fig. S24). Similar results were obtained with Cu-C3AmPicH (Fig. S25).
The CuII-affinity of EuIII-DO3A-C3AmpicH and C3AmpicH was further investigated using other physiological ligands with weaker CuII-affinity, like Aβ1–16 (log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Ka = 10)51 and α-Syn1–6 (α-Syn1–17: log
Ka = 10)51 and α-Syn1–6 (α-Syn1–17: log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Ka = 9)9,10 in equimolar conditions. UV-vis experiments showed that both Eu-DO3A-C3AmpicH and C3AmpicH immediately and completely retrieved Aβ1–16-bound CuII (Fig. 5B and S26, S27). On the other hand, Aβ1–16 failed to remove CuII from EuIII-DO3A-C3AmpicH and C3AmpicH, even when present in a 10-fold excess, indicating that these compounds have stronger affinity for CuII compared to Aβ1–16. As the d–d band of CuII-α-Syn1–6 at 625 nm is too close to the one of the studied Cu II-complexes, the CuII transfer between EuIII-DO3A-C3AmpicH and α-Syn1–6 was investigated by EuIII luminescence (Fig. 5C and S28) and the stronger affinity of EuIII-DO3A-C3AmpicH compared to α-Syn1–6 for CuII was confirmed.
Ka = 9)9,10 in equimolar conditions. UV-vis experiments showed that both Eu-DO3A-C3AmpicH and C3AmpicH immediately and completely retrieved Aβ1–16-bound CuII (Fig. 5B and S26, S27). On the other hand, Aβ1–16 failed to remove CuII from EuIII-DO3A-C3AmpicH and C3AmpicH, even when present in a 10-fold excess, indicating that these compounds have stronger affinity for CuII compared to Aβ1–16. As the d–d band of CuII-α-Syn1–6 at 625 nm is too close to the one of the studied Cu II-complexes, the CuII transfer between EuIII-DO3A-C3AmpicH and α-Syn1–6 was investigated by EuIII luminescence (Fig. 5C and S28) and the stronger affinity of EuIII-DO3A-C3AmpicH compared to α-Syn1–6 for CuII was confirmed.
To conclude, the affinities of Eu-DO3A-C3AmpicH and C3AmpicH for CuII are very similar, in contrast to the distinct affinities previously observed for Eu-DO3A-pyrGH vs. pyrGH. Hence, the propyl linker ensures an effective spatial separation between the LnIII and CuII complexes, and thereby reduces efficiently the electrostatic repulsion and steric hindrance between the two metal centers. The affinity of Eu-DO3A-C3AmpicH and C3AmpicH for CuII is between those of HSA and Aβ1–16, i.e. around log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Ka ∼ 11–12, which corresponds to a dissociation constant in the picomolar range (1–10 pM).
Ka ∼ 11–12, which corresponds to a dissociation constant in the picomolar range (1–10 pM).
2.6 Study of the CuII-selectivity
The selectivity of EuIII-DO3A-C3AmpicH and C3AmpicH for CuII over various metal ions was investigated by UV-Vis absorption and luminescence spectroscopy (Fig. S29–S32). In these experiments, 1 eq. CuII and 10 eq. ZnII were added to EuIII-DO3A-C3AmpicH and C3AmpicH in different orders. The addition of 10 eq. ZnII caused no change in the d–d transition region but led to a change in the absorption pattern below 300 nm (Fig. 6 and S29B), suggesting that ZnII interacts with the ligand. However, whatever the order of addition of the two metal ions, the CuII-complex was entirely formed. | ||
| Fig. 6 UV-Vis spectrum of the addition of 1 eq. CuII and 10 eq. ZnII to EuIII-DO3A-C3AmpicH, conditions: 100 μM EuIII-DO3A-C3AmpicH, 100 μM CuCl2, 1 mM ZnSO4, 10 mM TRIS pH 7.4. | ||
The selectivity of EuIII-DO3A-C3AmpicH for CuII over up to 500 eq. of ZnII was followed by luminescence spectroscopy. The addition of up to 5 eq. ZnII did not significantly impact the luminescence intensity of EuIII-DO3A-C3AmpicH. However, adding 10 or more equivalents resulted in an increase of up to 130% of the initial luminescence (Fig. S29A). This confirms some interactions between EuIII-DO3A-C3AmpicH and ZnII ions. Similar luminescence enhancements have been reported in the literature and are often attributed to an increase in rigidity of the complex upon ZnII binding.52–54 Another possibility is that ZnII induces conformational changes, bringing the pyridine sensitizer closer to EuIII, enhancing luminescence. The luminescence intensity remained high with the addition of up to 500 eq. of ZnII. With subsequent addition of CuII, the luminescence immediately decreased, confirming the selectivity for CuII over ZnII. Similar results were obtained in the presence of 65 eq. of CaII, 60 eq. of MgII, 1 eq. of MnII or FeIII (Fig. S30 and S31), confirming the selectivity of Eu-DO3A-C3AmpicH and C3AmPicH for CuII in these conditions.
In order to take into consideration that CuII is bound to a carrier in the body, competition experiments were also carried out in the presence of Aβ1–16. Thus, CuII and ZnII were added simultaneously to a solution containing EuIII-DO3A-C3AmpicH and Aβ1–16, in equimolar concentrations (Fig. S32). Given that Aβ1–16 has a 104-fold stronger affinity for CuII over ZnII, this experimental set-up represents more challenging conditions compared to the experiments in the absence of Aβ1–16. Immediately after the addition of CuII and ZnII, a d–d band centered at 600 nm emerged and showed no further temporal evolution in terms of intensity or position. The d–d band maximum was red-shifted by about 5 nm compared to EuIII–CuII-DO3A-C3AmpicH, suggesting that a minor part of CuII remains bound to Aβ1–16, whose absorption maximum lies around 635 nm when complexed to CuII.
3. Conclusion
The LnIII-DO3A-C3AmpicH complex was developed to bind CuII, and increase the distance between the LnIII- and CuII-binding sites in order to achieve a desirable affinity for CuII. Such design was successful as the CuII affinity of CuII-C3AmpicH and EuIII–CuII-DO3A-C3AmpicH are very similar, and in the appropriate range to compete with the Aβ1–16 peptide, or α-Synuclein. The selectivity for CuIIvs. other physiological cations, particularly ZnII, is very good. CuII could be detected by following the quenching of the luminescence of Eu-DO3A-C3AmpicH. This complex is able to detect CuII bound to Aβ or α-synuclein, key CuII reservoirs in Alzheimer's and Parkinson's diseases respectively, by luminescence. However, no change of relaxivities is observed upon CuII addition due to the absence of binding of the amide group to the LnIII in the absence of CuII, in contrast to what has been observed in the literature. This is due to the steric hindrance brought by the relatively rigid CuII binding site, as demonstrated by DFT calculations. Current efforts are pursued to further modify the CuII-binding site in order to obtain a relaxivity response.Author contributions
K. Z. performed synthesis of DO3A-C3AmpicH and C3AmpicH, Cu-binding studies, selectivity studies and contributed to analysis and writing. B. V. performed measurements, analysis and writing of EPR part; A. P. performed the relaxometric measurements, C. P.-I. performed DFT calculations and contributed to writing, P. F. supervised physico-chemical work and contributed to writing, C. B. supervised relaxometric measurements, did analysis and contributed to writing, A. S. supervised synthetic work and contributed to writing.Conflicts of interest
The authors declare no conflict of interest.Data availability
Data supporting this study are included within the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d5dt01735a.Acknowledgements
The French Ministry of Education and Research is acknowledged for a Ph.D. fellowship to Z. K. We also thank for the financial support from the Agence Nationale de la Recherche (SteRIC ANR-22-CE07-0030).References
- R. A. Festa and D. J. Thiele, Curr. Biol., 2011, 21, R877–R883 CrossRef PubMed.
- L. Chen, J. Min and F. Wang, Signal Transduction Targeted Ther., 2022, 7, 1–16 CrossRef PubMed.
- N. E. Hellman and J. D. Gitlin, Annu. Rev. Nutr., 2003, 22, 439–458 CrossRef.
- K. Bossak-Ahmad, T. Frączyk, W. Bal and S. C. Drew, ChemBioChem, 2020, 21, 331–334 CrossRef.
- S. Catalani, M. Paganelli, M. E. Gilberti, L. Rozzini, F. Lanfranchi, A. Padovani and P. Apostoli, J. Trace Elem. Med. Biol., 2018, 45, 176–180 CrossRef.
- V. Borghesani, B. Alies and C. Hureau, Eur. J. Inorg. Chem., 2018, 2018, 1–1 CrossRef.
- P. Faller and C. Hureau, Dalton Trans., 2009, 1080–1094 RSC.
- S. Bucossi, M. Ventriglia, V. Panetta, C. Salustri, P. Pasqualetti, S. Mariani, M. Siotto, P. M. Rossini and R. Squitti, J. Alzheimer's Dis., 2011, 24, 175–185 Search PubMed.
- R. De Ricco, D. Valensin, S. Dell'acqua, L. Casella, C. Hureau and P. Faller, ChemBioChem, 2015, 16, 2319–2328 CrossRef PubMed.
- T. Kowalik-Jankowska, A. Rajewska, K. Wiśniewska, Z. Grzonka and J. Jezierska, J. Inorg. Biochem., 2005, 99, 2282–2291 CrossRef PubMed.
- D. Valensin, F. Camponeschi, M. Luczkowski, M. C. Baratto, M. Remelli, G. Valensin and H. Kozlowski, Metallomics, 2011, 3, 292–302 CrossRef PubMed.
- A. Poujois, J. Poupon and F. Woimant, in Clinical and Translational Perspectives on WILSON DISEASE, ed. N. Kerkar and E. A. Roberts, Academic Press, 2019, pp. 249–255 Search PubMed.
- R. Squitti, R. Ghidoni, I. Simonelli, I. D. Ivanova, N. A. Colabufo, M. Zuin, L. Benussi, G. Binetti, E. Cassetta, M. Rongioletti and M. Siotto, J. Trace Elem. Med. Biol., 2018, 45, 181–188 CrossRef.
- S. El Balkhi, J. M. Trocello, J. Poupon, P. Chappuis, F. Massicot, N. Girardot-Tinant and F. Woimant, Clin. Chim. Acta, 2011, 412, 2254–2260 CrossRef PubMed.
- E. Falcone, M. Okafor, N. Vitale, L. Raibaut, A. Sour and P. Faller, Coord. Chem. Rev., 2021, 433, 213727 CrossRef CAS.
- K. P. Malikidogo, H. Martin and C. S. Bonnet, Pharmaceuticals, 2020, 13, 436 CrossRef CAS.
- J. Wahsner, E. M. Gale, A. Rodríguez-Rodríguez and P. Caravan, Chem. Rev., 2019, 119, 957–1057 CrossRef CAS PubMed.
- É. Tóth, L. Helm and A. E. Merbach, Contrast Agents I. Topics in Current Chemistry, Springer, Berlin, Heidelberg, 221st edn, 2002 Search PubMed.
- A. Merbach, L. Helm and É. Tóth, The Chemistry of Contrast Agents in Medical Magnetic Resonance Imaging, John Wiley and Sons, 2nd edn, 2013 Search PubMed.
- P. Yue, T. Nagendraraj, G. Wang, Z. Jin and G. Angelovski, Chem. Sci., 2024, 15, 20122–20154 RSC.
- E. L. Que and C. J. Chang, Chem. Soc. Rev., 2009, 39, 51–60 RSC.
- V. C. Pierre, S. M. Harris and S. L. Pailloux, Acc. Chem. Res., 2018, 51, 342–351 CrossRef CAS.
- P. Caravan, Chem. Soc. Rev., 2006, 35, 512–523 RSC.
- M. C. Heffern, L. M. Matosziuk and T. J. Meade, Chem. Rev., 2014, 114, 4496–4539 CrossRef CAS.
- J. P. C. Coverdale, J. P. Barnett, A. H. Adamu, E. J. Griffiths, A. J. Stewart and C. A. Blindauer, Metallomics, 2019, 11, 1805–1819 CrossRef CAS.
- W. Bal, M. Sokołowska, E. Kurowska and P. Faller, Biochim. Biophys. Acta, 2013, 1830, 5444–5455 CrossRef CAS PubMed.
- A. I. Bush, W. H. Pettingell, G. Multhaup, M. D. Paradis, J. P. Vonsattel, J. F. Gusella, K. Beyreuther, C. L. Masters and R. E. Tanzi, Science, 1994, 265, 1464–1467 CrossRef CAS PubMed.
- E. L. Que and C. J. Chang, J. Am. Chem. Soc., 2006, 128, 15942–15943 CrossRef CAS PubMed.
- N. N. Paranawithana, A. F. Martins, V. Clavijo Jordan, P. Zhao, S. Chirayil, G. Meloni and A. D. Sherry, J. Am. Chem. Soc., 2019, 141, 11009–11018 CrossRef CAS PubMed.
- K. Zimmeter, A. Pallier, B. Vileno, M. Sanadar, F. Szeremeta, C. Platas-Iglesias, P. Faller, C. S. Bonnet and A. Sour, Inorg. Chem., 2024, 63, 23067–23076 CrossRef CAS PubMed.
- M. Sanadar, K. Zimmeter, H. Martin, A. Pallier, B. Vileno, P. Faller, A. Sour and C. S. Bonnet, Eur. J. Inorg. Chem., 2025, 28, e202500049 CrossRef CAS.
- K. Zimmeter, B. Vileno, C. Platas-Iglesias, B. Vinjamuri, A. Sour and P. Faller, Inorg. Chem., 2023, 62, 9429–9439 CrossRef CAS PubMed.
- A. Mishra, N. K. Logothetis and D. Parker, Chem. – Eur. J., 2011, 17, 1529–1537 CrossRef CAS PubMed.
- M. Regueiro-Figueroa, S. Gündüz, V. Patinec, N. K. Logothetis, D. Esteban-Gómez, R. Tripier, G. Angelovski and C. Platas-Iglesias, Inorg. Chem., 2015, 54, 10342–10350 CrossRef CAS.
- P. Kadjane, C. Platas-Iglesias, P. Boehm-Sturm, V. Truffault, G. E. Hagberg, M. Hoehn, N. K. Logothetis and G. Angelovski, Chem. – Eur. J., 2014, 20, 7351–7362 CrossRef CAS PubMed.
- A. Congreve, D. Parker, E. Gianolio and M. Botta, Dalton Trans., 2004, 1441–1445 RSC.
- F. Oukhatar, H. Meudal, C. Landon, N. K. Logothetis, C. Platas-Iglesias, G. Angelovski and É. Tóth, Chem. – Eur. J., 2015, 21, 11226–11237 CrossRef CAS.
- M. Regueiro-Figueroa, S. Gündüz, V. Patinec, N. K. Logothetis, D. Esteban-Gómez, R. Tripier, G. Angelovski and C. Platas-Iglesias, Inorg. Chem., 2015, 54, 10342–10350 CrossRef CAS.
- S. Laine, J. F. Morfin, M. Galibert, V. Aucagne, C. S. Bonnet and É. Tóth, Molecules, 2021, 26, 2176 CrossRef CAS.
- C. Harford and B. Sarkar, Acc. Chem. Res., 1997, 30, 123–130 CrossRef CAS.
- D. M. Rudkevich, W. Verboom, E. van der Tol, C. J. Van Staveren, F. M. Kaspersen, J. W. Verhoeven and D. N. Reinhoudt, J. Chem. Soc., Perkin Trans. 2, 1995, 131–134 RSC.
- L. Pellegatti, J. Zhang, B. Drahos, S. Villette, F. Suzenet, G. Guillaumet, S. Petoud and É. Tóth, Chem. Commun., 2008, 6591–6593 RSC.
- É. Tóth, O. M. N. Dhubhghaill, G. Besson, L. Helm and A. E. Merbach, Magn. Reson. Chem., 1999, 37, 701–708 CrossRef.
- K. A. Brameld, B. Kuhn, D. C. Reuter and M. Stahl, J. Chem. Inf. Model., 2008, 48, 1–24 CrossRef CAS.
- H. Sigel and R. B. Martin, Chem. Rev., 1982, 82, 385–426 CrossRef CAS.
- A. Belczyk-Ciesielska, I. A. Zawisza, M. Mital, A. Bonna and W. Bal, Inorg. Chem., 2014, 53, 4639–4646 CrossRef CAS PubMed.
- A. W. Addison and E. Sinn, Inorg. Chem., 1983, 22, 1225–1228 CrossRef CAS.
- J. Peisach and W. E. Blumberg, Arch. Biochem. Biophys., 1974, 165, 691–708 CrossRef CAS PubMed.
- M. Drosou, C. A. Mitsopoulou, M. Orio and D. A. Pantazis, Magnetochemistry, 2022, 8, 36 CrossRef CAS.
- G. Sciortino, G. Lubinu, J.-D. Maréchal and E. Garribba, Magnetochemistry, 2018, 4, 55 CrossRef CAS.
- V. Borghesani, B. Alies and C. Hureau, Eur. J. Inorg. Chem., 2018, 2018, 1–1 CrossRef.
- K. C. Weerasinghe and L. Wang, ChemRxiv, 2023, preprint, DOI:10.26434/CHEMRXIV-2023-PJG54-V2.
- S. A. McFarland and N. S. Finney, J. Am. Chem. Soc., 2002, 124, 1178–1179 CrossRef CAS PubMed.
- J. Luo, W. S. Li, P. Xu, L. Y. Zhang and Z. N. Chen, Inorg. Chem., 2012, 51, 9508–9516 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2025 |