Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Structural diversities and magnetic properties of azido- and dicyanamido-bridged coordination polymers with 4,5-diphenylimidazole as a terminal ligand
Evangelia-Spyridoula
Fotopoulou
a,
Nikos
Panagiotou
b,
Anastasios J.
Tasiopoulos
 b,
Mark M.
Turnbull
b,
Mark M.
Turnbull
 *c,
Spyros P.
Perlepes
*c,
Spyros P.
Perlepes
 *a and
Vassilios
Nastopoulos
*a and
Vassilios
Nastopoulos
 *a
*a
aUniversity of Patras, Department of Chemistry, 26504 Patras, Greece. E-mail: nastopoulos@upatras.gr; perlepes@upatras.gr
bUniversity of Cyprus, Department of Chemistry, 1678 Nicosia, Cyprus
cClark University, Carlson School of Chemistry and Biochemistry, Worcester, MA 01610, USA. E-mail: MTurnbull@clarku.edu
First published on 19th August 2025
Abstract
With the aim of assessing the role and impact of pseudohalides on the molecular and supramolecular structures of copper(II)/heavily substituted imidazole complexes, the synthesis and characterization of the complexes {[Cu(N3)2(HL)]}n (1), {[Cu{N(CN)2}2(HL)]·Me2CO}n (2·Me2CO) and [Cu(NCO)2(HL)2]·MeCN (3·MeCN) have been carried out; HL is the 4,5-diphenylimidazole ligand. The organic molecule behaves as a monodentate ligand through the pyridine-type nitrogen atom of the imidazole ring. The 1D polymer 1 is made up of undulating {–Cu–(N3)2–Cu–(N3)2–} chains, with the HL ligands extending from both sides of the chains. The square pyramidal metal ions are alternately doubly bridged by two different types (basal–basal and basal–apical) of end-on azido groups. In 2·Me2CO, the N(CN)2− ions behave as end-to-end (μ-1,5 or 2.101) ligands forming a 2D polymeric lattice, consisting of four-membered CuII rings interconnected by four dicyanamido bridges; the five-coordinate CuII ion adopts a geometry intermediate between square pyramidal and trigonal bipyramidal. The metal centre in the mononuclear complex 3·MeCN is bonded to two HL ligands and two terminal isocyanato groups; the coordination geometry of CuII is closest to a seesaw conformation. The crystal structures of the complexes are built with a variety of supramolecular interactions, including the synthons N−H⋯X (X = N and O), which are critically discussed. The IR spectra of the compounds are discussed in terms of the coordination modes of the pseudohalido ligands. The magnetic properties of 1 and 2 have also been studied in the temperature and field ranges of 1.8–300 K and 0–50 kOe, respectively. Complex 1 exhibits both ferromagnetic and antiferromagnetic CuII⋯CuII exchange interactions propagated through the two different end-on bi-azido bridges, whereas the exchange interactions are negligible (if any) in the 2D polymer 2 as a consequence of the μ-1,5 ligation mode of the dicyanamido ligand and the resultant long CuII⋯CuII distances.
Introduction
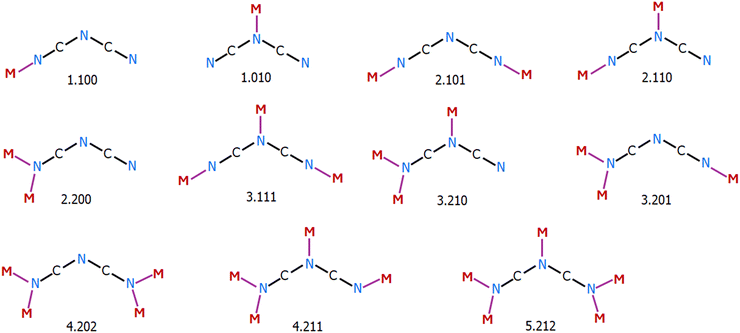
Linear pseudohalide ions such as N3−, NCO−, and NCS−, along with the larger bent dicyanamide ion, N(CN)2−, have been used as ligands in synthetic inorganic chemistry, due to their ability to coordinate with transition metal ions in various ways. For this reason, they are often named ‘schizophrenics of coordination chemistry’.1 When combined with ancillary terminal organic ligands, they facilitate the synthesis of compounds with interesting architectures and topologies.2 This approach enables the construction of polynuclear complexes and coordination polymers with diverse properties, structures and topologies. This class of materials holds significant potential in crystal engineering, with applications in molecular sensors,3–5 microporous materials for gas adsorption and storage,6 electrical conductivity,7,8 nonlinear optical activity,9,10 catalysis,11,12etc. Bridging pseudohalides can efficiently transmit magnetic interactions between paramagnetic metal centres. As a result, pseudohalides play a crucial role in molecular magnetic systems, spanning single-molecule magnets (SMMs),13 single-chain magnets (SCMs),14 higher-dimensional magnetic materials,15 and multifunctional systems such as photoactive and porous magnets.16,17The N3− ion is one of the most common bridging ligands among pseudohalides, widely used in the synthesis of metal clusters and coordination polymers, where it imparts intriguing magnetic and topological properties. It can act as either a terminal or a bridging ligand.1,2,18–22 The crystallographically verified coordination modes of the azido ligand are illustrated in Scheme 1. When functioning as a monoatomic bridging ligand, the azide ion can link up to four metal ions via end-on (EO) coordination.1 If both terminal nitrogen atoms participate in bonding, it acts as a bidentate bridging ligand, linking at least two metal centers through an end-to-end (EE) coordination mode. A combination of these two modes enables azide to bridge up to six metal ions. In some cases, a single compound may contain azido ligands adopting two or even three different coordination modes. It has been observed that the most common coordination mode of N3− in metal clusters is EO.2,18 In 1D, 2D, and 3D coordination polymers, the EE mode predominates, sometimes alternating with EO or forming mixed EE/EO networks.2,18,19 Coordination polymers exclusively featuring EO-bridged azides are rare. Due to the comparable stability of EO and EE coordination modes, steric hindrance is usually the key factor determining whether a complex adopts EO, EE, or a mixed EE/EO mode. The azide ion is widely used as a bridging ligand in the field of molecular magnetism, as its various coordination modes and the diverse exchange interactions it promotes lead to a range of magnetic behaviors.2,18,19 These include ferromagnetism, antiferromagnetism, ferrimagnetism, canted weak ferromagnetism, spin-flop transitions, single-molecule magnetism (SMM), and single-chain magnetism (SCM). Literature studies indicate that the end-on (EO) coordination mode typically favors ferromagnetic interactions, potentially leading to a high-spin ground state.2,18–21 In contrast, the end-to-end (EE) mode generally promotes antiferromagnetic interactions.2,18,21 However, the coordination mode alone is not the sole determining factor of magnetic coupling.
The relatively larger dicyanamide ion, N(CN)2−, is a flexible ligand that, in combination with transition metals, can form a wide range of compounds with diverse architectures and topologies. The dicyanamide ion has three potential donor sites and can act as either a terminal or a bridging ligand.23–25 In polynuclear complexes, N(CN)2− exhibits various coordination modes, binding to metal centers through the nitrogen atoms of the terminal nitrile groups, the central amido nitrogen, or both, functioning as a tridentate ligand. Consequently, dicyanamide can form coordination bonds with one to five metal ions. The coordination modes of dicyanamide, as revealed by X-ray crystallography, are illustrated in Scheme 2. These versatile binding properties enable dicyanamide to form 1D, 2D, and 3D structures, which may also exhibit interesting magnetic properties.23–28
Transition metal complexes with the cyanate ion (NCO−) are of particular interest due to its ambidentate nature, allowing it to coordinate to metal ions either as a terminal or a bridging ligand through its nitrogen or oxygen atom, or both.1,13,29–34 However, the number of polynuclear and polymeric complexes incorporating the cyanate ion is significantly smaller compared to those involving other pseudohalides. In most complexes, bonding occurs preferentially through the nitrogen atom. The N-site of NCO− lies at the boundary between hard and soft Lewis bases (HSAB), allowing it to bind to a wide range of metal ions. In contrast, the hard O-site has a stronger affinity for hard metal ions, although this preference can be influenced by the nature of other ligands and the stereochemical environment they impose. When NCO− acts as a monodentate ligand, it forms mononuclear complexes. However, as a bridging ligand, it can link metal centers via either end-on (EO) coordination modes or an end-to-end (EE) mode, as illustrated in Scheme 3.1,13,29–34
In recent years, we have studied a series of mononuclear complexes, primarily of the type MX2(HL)2 or [M(HL)4]X2 [M = Co2+, Ni2+, Cu2+ or Zn2+; X = Cl−, Br−, I−, NO3− or ClO4−; HL = 4,5-diphenylimidazole], to explore their assembly patterns, with a particular focus on both strong and weak intermolecular interactions.35–38 This work has been recently extended to 5-phenylimidazole.39,40 Imidazole and its derivatives41,42 are, among others, particularly interesting ligands in bioinorganic43,44 and metallosupramolecular45–47 chemistry. The monodentate HL ligand employed in this work (Scheme S1) coordinates to the metal ion via its pyridine-type N3 atom and contains a H-bond donor (the pyrrolic-type N1 atom), enabling the formation of motifs that may drive molecular self-assembly. Additionally, it supports π⋯π stacking interactions through its phenyl rings and the five-membered heterocyclic ring. There has been relatively little work on the coordination chemistry of heavily substituted imidazoles and, in particular, about HL.35–38,48–50 The X ions in these complexes coordinate terminally as either monodentate or bidentate ligands, thereby excluding the possibility of forming coordination polymers. Given the variation in their size and shapes, their influence on the metal's coordination geometry (when coordinated) or their spatial role in packing organization (when serving as counterions) has also been examined by our group.
In this context, it appeared interesting to explore the replacement of the halide ions with larger pseudohalides N3−, N(CN)2−, NCO− and SCN− in combination with 4,5-diphenylimidazole as an ancillary ligand in order to investigate their impact on the molecular and supramolecular organization, as well as the properties of the resulting complexes and coordination polymers. As reported, pseudohalides, when combined with other organic ligands and 3d metals, can promote the synthesis of compounds often exhibiting diverse architectures, topologies, and properties, making them a compelling area of study.
Copper(II) was selected as the metal for this study due to its versatile coordination numbers and geometries. Due to its 3d9 configuration, square planar or distorted tetrahedral (4-coordinate), square pyramidal or trigonal bipyramidal (5-coordinate), as well as distorted octahedral (6-coordinate) geometries dominate the coordination chemistry of copper(II). Additionally, the presence of one unpaired electron on this metal ion makes the interpretation of the magnetic properties of copper(II) complexes more straightforward compared to those of other 1st-row transition metal complexes.51,52
We report herein the results of the amalgamation of the above mentioned topics. Two coordination polymers, {[Cu(N3)2(HL)]}n (1) and {[Cu{N(CN)2}2(HL)]·Me2CO}n (2·Me2CO), together with the mononuclear complex [Cu(NCO)2(HL)2]·MeCN (3·MeCN), have been obtained and structurally/spectroscopically characterized, exploring the reaction system CuII/HL/pseudohalide; the magnetic properties of the polymeric complexes have also been studied. One synthetic or crystallization parameter at a time was varied during each set of experiments so as to assess its relative effect on the product identity.
Experimental
Materials and instruments
All reagents, solvents and starting materials were reagent grade, purchased from standard suppliers and used as received. All manipulations were performed under aerobic conditions. Microanalyses (C, H, and N) were performed by the University of Patras microanalytical service. IR spectra (4000–400 cm−1) were recorded using a Perkin-Elmer PC 16 FT-IR spectrometer with the samples prepared as KBr pellets. For magnetic studies, samples of compounds 1 and 2 were powdered and mounted in gelatine capsules placed in clear plastic straws. Magnetization data, as a function of field (0–50 kOe), were collected at 1.8 K for compounds 1 and 2 employing a Quantum Design MPMS-5 SQUID magnetometer. Several data points were recollected as the field was returned to zero; no hysteresis was observed. Data were then collected in a 1 kOe field as a function of temperature from 1.8 to 310 K. Measurements were corrected for the diamagnetism of the constituent atoms as estimated from Pascal's constants, the temperature independent paramagnetism of the Cu(II) ion and the gelatine capsule and straw (measured independently).Safety note: Azido and dicyanamido compounds and perchlorate salts are potentially explosive; such compounds should be synthesized and used in small quantities and treated with utmost care at all times.
Preparation of complexes
X-ray crystallography
A suitable single crystal of each of the three compounds was selected, coated with Paratone-N oil, and mounted in cryo-loops at the end of a copper pin. Diffraction data were collected using the ω-scan technique on a Rigaku Oxford Diffraction SuperNova diffractometer under a stream of nitrogen gas at 100(2) K; Cu Kα radiation (λ = 1.5418 Å) was used for compounds 1 and 2·Me2CO, while Mo Kα radiation (λ = 0.7107 Å) was used for compound 3·MeCN. Table 1 shows the summary of crystallographic data. Data were collected and processed using CRYSALIS CCD and RED software,53 respectively; the reflection intensities were corrected for absorption using the multiscan method. The structures were solved using classical direct methods (SIR92,54 Semi-Invariants Representation), the dual-space algorithm with SHELXT,55 or the charge flipping algorithm (Superflip56,57). The structural models were then refined by full-matrix least-squares on F2 with SHELXL-2019/3.58 All non-H atoms were refined anisotropically; carbon-bound H atoms were introduced at calculated positions and allowed to ride on their carrier atoms (riding model). All imidazole H atoms on the pyrrolic-type N1 atom of the ligands were located in difference Fourier maps and refined isotropically applying soft distance restraints. Considerable disorder has been observed in the crystal structure of compound 3·MeCN. One of the phenyl rings in the HL ligand exhibits positional disorder, which was treated by applying a two-part disorder model with a 57![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 43 domain ratio. Additionally, the MeCN solvent molecule is orientationally disordered (50
43 domain ratio. Additionally, the MeCN solvent molecule is orientationally disordered (50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50) about a two-fold axis running parallel to the b axis of the unit cell. Appropriate restraints were applied during refinement to ensure a chemically reasonable geometry for each disordered component.
50) about a two-fold axis running parallel to the b axis of the unit cell. Appropriate restraints were applied during refinement to ensure a chemically reasonable geometry for each disordered component.
| Compound reference | 1 | 2·Me2CO | 3·MeCN |
|---|---|---|---|
| Chemical formula | C15H12CuN8 | C19H12CuN8·C3H6O | C32H24CuN6O2·C2H3N |
| Formula mass g−1 mol−1 | 367.87 | 473.98 | 629.16 |
| Crystal system | Monoclinic | Monoclinic | Orthorhombic |
| Space group | P21/n | P21/n | Pbcn |
| a/Å | 9.2102(3) | 7.7400(2) | 17.0660(7) |
| b/Å | 5.5759(2) | 11.7233(3) | 12.7219(4) |
| c/Å | 30.9154(9) | 23.8510(5) | 13.6879(6) |
| α/° | 90 | 90 | 90 |
| β/° | 92.515(3) | 94.612(2) | 90 |
| γ/° | 90 | 90 | 90 |
| Unit cell volume Å−3 | 1586.14(9) | 2157.19(9) | 2971.8(2) |
| Z, Z′ | 4, 1 | 4, 1 | 4, 0.5 |
| Temperature/K | 100(2) | 100(2) | 100(2) |
| Radiation type, μ/mm−1 | CuKα, 2.071 | CuKα, 1.696 | MoKα, 0.779 |
| No. of reflections measured | 5882 | 13![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 654 654 |
24![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 688 688 |
| No. of independent reflections | 3022 [Rint = 0.0216] | 4159 [Rint = 0.0318] | 2759 [Rint = 0.0439] |
| Data/restraints/parameters | 3022/0/220 | 4159/1/294 | 2759/147/247 |
| Final R1 values (I > 2σ(I)) | 0.0306 | 0.0308 | 0.0666 |
| Final wR(F2) values (I > 2σ(I)) | 0.0790 | 0.0792 | 0.1548 |
| Final R1 values (all data) | 0.0373 | 0.0366 | 0.0719 |
| Final wR(F2) values (all data) | 0.0832 | 0.0834 | 0.1574 |
| Goodness of fit on F2 | 1.033 | 1.026 | 1.231 |
| Δρmax, Δρmin/e Å−3 | 0.376, −0.500 | 0.307, −0.369 | 1.039, −1.773 |
| CCDC number | 2456934 | 2456935 | 2456936 |
Geometric/crystallographic calculations were carried out using PLATON,59 OLEX260 and WINGX61 packages; molecular/packing graphics were prepared using MERCURY.62
Results and discussion
Synthetic comments
A variety of reactions were studied with the aim of preparing the largest possible number of compounds (discrete complexes or polymers) by exploring the CuII/HL/pseudohalide system (HL = 4,5-diphenylimidazole; pseudohalide = N3−, N(CN)2−, NCO−, and SCN−). Our trials included differing reactant ratios and concentrations, solvents, crystallization conditions and temperatures. Cu(II) is air stable, and the synthetic work was thus performed under aerobic conditions under the normal laboratory atmosphere. The reagents utilized in the experiments as sources for the respective ions in the reaction scheme involved Cu(ClO4)2·6H2O for copper(II) ions; NaN3 and (CH3)3SiN3 for azide ions; Na{N(CN)2} for dicyanamide ions; NaNCO for cyanate ions; and NH4SCN for thiocyanate ions. Two coordination polymers have been obtained, {[Cu(N3)2(HL)]}n (1) and {[Cu{N(CN)2}2(HL)]·Me2CO}n (2·Me2CO). Efforts to isolate an analogous coordination polymer using cyanates resulted in the formation of the mononuclear complex [Cu(NCO)2(HL)2]·MeCN (3·MeCN). Compounds were obtained by reactions with a CuII![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) HL
HL![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) pseudohalide ratio of 1
pseudohalide ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.5
2.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 for 1 and 3·MeCN and 1
2 for 1 and 3·MeCN and 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2
2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 for 2·Me2CO. Despite our efforts, no crystalline thiocyanato solids have been obtained yet. The formation of the complexes can be summarized by eqn (1)–(3):
2 for 2·Me2CO. Despite our efforts, no crystalline thiocyanato solids have been obtained yet. The formation of the complexes can be summarized by eqn (1)–(3): | (1) |
 | (2) |
 | (3) |
It should be mentioned that complexes 1–3 are the only products from their corresponding reaction mixtures irrespective of the CuII![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) HL reaction ratio. Thus, a large excess of HL (e.g., CuII
HL reaction ratio. Thus, a large excess of HL (e.g., CuII![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) HL = 1
HL = 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4) resulted again in the polymeric 1
4) resulted again in the polymeric 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 complexes 1 and 2, and thus mononuclear complexes [CuX2(HL)2], where X = N3 and N(CN)2, could not be isolated. An excess of metal (e.g., CuII
1 complexes 1 and 2, and thus mononuclear complexes [CuX2(HL)2], where X = N3 and N(CN)2, could not be isolated. An excess of metal (e.g., CuII![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) HL = 1
HL = 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) led again to the mononuclear complex 3, and the anticipated {[Cu(NCO)2(HL)]}n complex could not be prepared.
1) led again to the mononuclear complex 3, and the anticipated {[Cu(NCO)2(HL)]}n complex could not be prepared.
All crystalline products were characterized by IR spectroscopy, microanalyses and single-crystal X-ray diffraction; the magnetic properties of the polymeric compounds were also studied.
Description of the structures
For consistency in discussion and molecular comparison, the same numbering scheme has been assigned to the HL ligand atoms across all three compounds presented herein. Selected interatomic distances and angles for all compounds are provided in Table S1. Details of H-bonding, C–H⋯π and π⋯π interactions are given in Tables S2–S4, respectively. Various structural plots are shown in Fig. 1–5 and S1–S4.Compound 1 crystallizes as a 1D polymer in the monoclinic space group P21/n. The asymmetric unit comprises one CuII ion, two azido groups, and one 4,5-diphenylimidazole molecule (Fig. S1). The two azido groups (N4–N5–N6 and N7–N8–N9) act as monoatomic bridging ligands in an end-on (μ-1,1 or 2.20) mode connecting two metal centres through the N4 and N7 atoms, respectively. Due to crystallographic symmetry, each CuII centre is ultimately coordinated by four azido groups, with its coordination sphere completed by the pyridine-type N3 atom of a 4,5-diphenylimidazole ligand. As a result, the metal ions adopt a five-coordinate (CuN5) environment. According to the angular structural parameter τ5,63 the CuII coordination geometry is a distorted square pyramid, where the N7 symmetry-related atom (1 − x, 1 − y, −z), located at a distance of 2.330(2) Å from copper(II), occupies the apical position (Fig. S2). The τ5 value is 0.25, where τ5 = 0 corresponds to an ideal square pyramidal geometry and τ5 = 1 corresponds to an ideal trigonal bipyramidal geometry. The Cu–N bond lengths range from 1.968(2) to 2.330(2) Å. The bond length of the apical nitrogen atom is the largest, as expected. The N–N bond lengths in the two azido ligands, corresponding to the coordinated and central nitrogen atoms (i.e., N4–N5 = 1.215 Å and N7–N8 = 1.198 Å), are longer than those between the central and non-coordinated terminal nitrogen atoms (i.e., N5–N6 = 1.133 Å and N8–N9 = 1.156 Å, respectively). This elongation is attributed to the ‘loss’ of electron density at the two bridging nitrogen atoms. The N–N–N bond angles of the two nearly linear azido ligands are 179.2(2)° and 176.7(2)° for N4–N5–N6 and N7–N8–N9, respectively.
The 1D polymer {[Cu(N3)2(HL)]}n is made up of undulating Cu-azido chains, with the coordinated HL ligands extending from both sides of the chains (Fig. 1). These chains extend parallel to the b axis of the unit cell, with Cu⋯Cu distances of 3.093(1) and 3.317(1) Å between consecutive CuII atoms along the chain. At a supramolecular level, these parallel chains are linked by strong hydrogen bonds (synthons) of the type N1imidazole–H1⋯N9azide [N1⋯N9 = 2.923(2) Å], together with weak C–H⋯Nazide interactions, forming layers parallel to the ab plane (Fig. 2 and Table S2). These interactions contribute to the self-assembly and stabilization of the crystal structure. Finally, the three-dimensional structure is further stabilized by weak C–H⋯π interactions between the chains within each layer, as well as between adjacent layers (Fig. 2 and Table S3).
Compound 2·Me2CO, containing the dicyanamido ligand, crystallizes in the monoclinic space group P21/n. The asymmetric unit consists of a CuII atom coordinated with two dicyanamide ions and one molecule of 4,5-diphenylimidazole (HL). The structure also incorporates an acetone molecule as a consequence of the crystallization process (Fig. S3). The dicyanamide ions act as bridging ligands through their nitrogen atoms N4 and N6, as well as atoms N7 and N9 of the nitrile groups (coordination mode: end-to-end, 2.101 or μ-1,5). This arrangement forms a 2D polymeric structure parallel to the ab plane of the unit cell, consisting of four-membered metal rings interconnected by four dicyanamide bridges (Fig. 3, left). The coordination sphere of the metal ions is completed by the pyridine-like nitrogen atom (N3) of the 4,5-diphenylimidazole ligand (HL). The five-coordinate CuII ion (with a τ5 index value of 0.51) adopts a geometry intermediate between square pyramidal (τ5 = 0) and trigonal bipyramidal (τ5 = 1). When viewed along the a axis (Fig. 3, right), the 2D structure is best described as a corrugated surface parallel to the ab plane, with the four CuII ions of each ring positioned at the intersection of the planes of the corrugated surface. The dihedral angle between adjacent planes of this surface is 86.1(1)°. The Cu⋯Cu distances within the rings are 7.740(1) Å and 8.227(1) Å. Weak interactions, such as π⋯π stacking between aromatic rings of parallel layers and C–H⋯Ndicyanamide interactions, contribute to the self-assembly of the polymeric structure. Finally, the N1–H1 donor group does not engage in any H-bonding within the complex, presumably due to steric hindrance. Instead, it forms a hydrogen bond with the oxygen atom of an acetone molecule [N1imidazole⋯Oacetone = 2.848(2) Å], which accounts for the incorporation of this crystallization molecule into the crystal lattice (Tables S2 and S4).
The crystal structure of compound [Cu(NCO)2(HL)2]·MeCN (3·MeCN) is orthorhombic with the space group Pbcn. The mononuclear complex exhibits molecular symmetry, with a two-fold rotation axis passing through its CuII centre (Fig. 4). Additionally, the MeCN solvent molecules exhibit orientational disorder, occupying two symmetrically equivalent positions around the two-fold axis in a 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50 ratio. The metal centre of the complex is bonded to two HL ligands via the pyridine-type nitrogen atoms of their imidazole rings, along with two terminal cyanate ions coordinated through their nitrogen atoms (isocyanato ligands). According to the τ4 metrical parameter proposed by Yang, Powell, and Houser64 for assessing the geometry of 4-coordinate metal ions, the coordination geometry of CuII is closest to a seesaw configuration (τ4 = 0.38). This parameter ranges from 1 for ideal tetrahedral (Td) geometry to 0.85 for trigonal pyramidal (C3v) and 0 for square planar (D4h) geometry. The seesaw geometry, with an ideal C2v (mm2) symmetry, lies between trigonal pyramidal and square planar structures. However, in this case, the geometry is distorted due to steric restraints imposed by the bulky 4,5-diphenylimidazole ligands. As a result, only the two-fold axis is retained, allowing the two ligands to adopt a favourable ‘antiparallel’ arrangement with their aromatic rings oriented in opposite directions (Fig. 4). The NCO− ion is nearly linear with an N–C–O angle of 177.8(6)°. At the supramolecular level, strong N1imidazole–H1⋯ONCO hydrogen bonds [N1⋯O = 2.826(6) Å] assemble the complexes into 2D layers parallel to the ab plane (Fig. S4). These layers are further interconnected by a few C–H⋯π interactions that enhance the stability of the overall 3D architecture (Tables S2 and S3). The visualization of these interactions in the crystal structure of compound 3·MeCN using Hirshfeld surface analysis is shown in Fig. 5. The 3D molecular Hirshfeld surface is defined by the molecule and the proximity of its nearest neighbours, effectively capturing information about all intermolecular interactions. The MeCN solvent molecules are located in channels extending along the c axis of the unit cell. PLATON analysis indicates that, after solvent removal, the calculated void space of the channels accounts for 10.7% of the unit cell volume (Fig. S4).
50 ratio. The metal centre of the complex is bonded to two HL ligands via the pyridine-type nitrogen atoms of their imidazole rings, along with two terminal cyanate ions coordinated through their nitrogen atoms (isocyanato ligands). According to the τ4 metrical parameter proposed by Yang, Powell, and Houser64 for assessing the geometry of 4-coordinate metal ions, the coordination geometry of CuII is closest to a seesaw configuration (τ4 = 0.38). This parameter ranges from 1 for ideal tetrahedral (Td) geometry to 0.85 for trigonal pyramidal (C3v) and 0 for square planar (D4h) geometry. The seesaw geometry, with an ideal C2v (mm2) symmetry, lies between trigonal pyramidal and square planar structures. However, in this case, the geometry is distorted due to steric restraints imposed by the bulky 4,5-diphenylimidazole ligands. As a result, only the two-fold axis is retained, allowing the two ligands to adopt a favourable ‘antiparallel’ arrangement with their aromatic rings oriented in opposite directions (Fig. 4). The NCO− ion is nearly linear with an N–C–O angle of 177.8(6)°. At the supramolecular level, strong N1imidazole–H1⋯ONCO hydrogen bonds [N1⋯O = 2.826(6) Å] assemble the complexes into 2D layers parallel to the ab plane (Fig. S4). These layers are further interconnected by a few C–H⋯π interactions that enhance the stability of the overall 3D architecture (Tables S2 and S3). The visualization of these interactions in the crystal structure of compound 3·MeCN using Hirshfeld surface analysis is shown in Fig. 5. The 3D molecular Hirshfeld surface is defined by the molecule and the proximity of its nearest neighbours, effectively capturing information about all intermolecular interactions. The MeCN solvent molecules are located in channels extending along the c axis of the unit cell. PLATON analysis indicates that, after solvent removal, the calculated void space of the channels accounts for 10.7% of the unit cell volume (Fig. S4).
IR spectroscopy in brief
The three complexes were characterized by IR spectroscopy. The spectra exhibit the characteristic bands of the phenyl and imidazole rings of coordinated HL. The weak to medium band at ca. 3200 cm−1 is attributed to the ν(NH) vibration. The broadness and rather low wavelength of this band are both indicative of hydrogen bonding. We comment now on the bands due to the pseudohalide groups.The spectrum of 1 exhibits two strong bands at 2074 and 2050 cm−1, assigned to the asymmetric stretching mode of the monoatomic bridging azido ligands, νas(NNN).65,66 The appearance of two bands reflects the presence of two, crystallographically independent, 2.20 azido groups in the structure. The shoulder at 1278 cm−1 and the weak band at 574 cm−1 can be tentatively assigned to the νs(NNN) and δ(NNN) vibrations, respectively.65 The free dicyanamide ion in Na{N(CN)2} shows three sharp and medium to strong characteristic stretching bands in the 2290–2175 cm−1 region attributable to a νas(C–N) + νs(C–N) combination mode (2286 cm−1) and the νas(C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N) (2232 cm−1) and νs(C
N) (2232 cm−1) and νs(C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N) (2179 cm−1) vibrations.24,25,65 The shift of these bands to higher wavenumbers in the spectrum of 2 (2300–2194 cm−1) is characteristic of the bidentate bridging 2.101 ligation mode of N(CN)2−; monodentate coordination has been reported to have a minor effect on these features. The linear triatomic free (i.e., ionic) NCO− species belongs to the C∞v point group. The three normal modes of vibration (ν1, ν2, and ν3) are IR- and Raman-active. These vibrations are commonly described as unmixed group frequencies, even though this is an approximation. Thus, the pseudo-antisymmetric stretching frequency (ν1) is referred to as the ν(C
N) (2179 cm−1) vibrations.24,25,65 The shift of these bands to higher wavenumbers in the spectrum of 2 (2300–2194 cm−1) is characteristic of the bidentate bridging 2.101 ligation mode of N(CN)2−; monodentate coordination has been reported to have a minor effect on these features. The linear triatomic free (i.e., ionic) NCO− species belongs to the C∞v point group. The three normal modes of vibration (ν1, ν2, and ν3) are IR- and Raman-active. These vibrations are commonly described as unmixed group frequencies, even though this is an approximation. Thus, the pseudo-antisymmetric stretching frequency (ν1) is referred to as the ν(C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N) mode, the pseudo-symmetric stretching frequency (ν3) to the ν(C–O) mode and ν2 to the doubly degenerate deformation or bending δ(NCO) vibration.29 The ν1, ν2 and ν3 vibrations of the free cyanate ion (i.e., in KNCO) appear at 2160, 630 and 1250 cm−1, respectively. The corresponding modes in 3 are associated with the bands at 2216, 616 and 1276 cm−1. The shift of the stretching bands to higher wavenumbers in the spectrum of the complex, compared to those in KNCO, is indicative of N-coordination.29,65
N) mode, the pseudo-symmetric stretching frequency (ν3) to the ν(C–O) mode and ν2 to the doubly degenerate deformation or bending δ(NCO) vibration.29 The ν1, ν2 and ν3 vibrations of the free cyanate ion (i.e., in KNCO) appear at 2160, 630 and 1250 cm−1, respectively. The corresponding modes in 3 are associated with the bands at 2216, 616 and 1276 cm−1. The shift of the stretching bands to higher wavenumbers in the spectrum of the complex, compared to those in KNCO, is indicative of N-coordination.29,65
Magnetochemistry
Direct current (dc) magnetic susceptibility data (χ) on dried, analytically pure samples of 1 and 2 were collected in the 1.8–310 K range in an applied field of 1 kOe. Data are presented in Fig. 6, 7, S5 and S6. We describe the results using the emu convention.The magnetization (M) as a function of field (H) plot for complex 1 at 1.8 K (Fig. S5) is featureless. The curvature shows that it is approaching saturation near 5400 emu mol−1 at 50 kOe, but has not yet reached it. This is very reasonable for an S = 1/2 system with g slightly greater than 2.00, as is typical for Cu(II) complexes in the presence of weak antiferromagnetic interactions.
The χ as a function of T plot is interesting. The χ versus T data appear featureless, but the 1/χ versus T and χT versus T data (Fig. 6) clearly show the presence of magnetic exchange within the system. Although linear at high temperatures, the 1/χ data show a positive intercept, indicative of dominant ferromagnetic interactions, and a distinct curvature with decreasing temperature. The data were fit to the Curie–Weiss law from 310 K downward with a decreasing lower T limit until the quality of fit and the value of θ (Weiss constant) began to change significantly. This yielded a lower limit of 112 K with a Curie constant C = 0.402(2) emu K mol−1 Oe and θ = +15.1(9) K. Decreasing the lower limit to 101 K resulted in θ = +14.3(10) K and decreasing to 95 K gave θ = +13.7(8) K. The Curie constant was stable within the error over all these temperature ranges. The steady decrease in θ as the lower temperature decreases indicates the presence of weaker antiferromagnetic exchange in addition to ferromagnetic interactions. The plot of the χT versus T data confirms the presence of both ferromagnetic and antiferromagnetic interactions. As T decreases, the χT product slowly increases from 0.42 to a maximum of 0.49 emu K mol−1 Oe at 41 K. Below this temperature, the χT product begins to decrease rapidly, reaching a value of 0.27 emu K mol−1 Oe at 1.8 K. An examination of the molecular structure suggests an explanation for the presence of both interactions. Although there is a single, crystallographically unique CuII atom in the molecule, there are two different bi-azide bridges that link these metal ions into a chain (Fig. 1). One bridge (possessing the N4 atoms) not only exhibits nearly symmetrical azido coordination [Cu–N bond lengths of 1.988(2) and 2.009(2) Å], but also connects the CuII atoms through basal–basal Cu–N bonds. In contrast, the other bridging interaction (containing the N7 atoms) is quite unsymmetrical [Cu–N bond lengths of 1.989(2) and 2.330(1) Å], since one of these bonds lies in the apical site of the square pyramidal geometry. In other words, one azido nitrogen N7 occupies a position in the basal plane of one CuII atom, while the same nitrogen occupies the apical position of the neighboring metal ion (see Table S1). The Cu–N7–Cu and Cu–N4–Cu bridging angles are quite similar [100.1(1) and 101.4(1)°, respectively] and unlikely to be the cause of such a dramatic magnetic exchange shift. Given these observations, the χT versus T data were fit to a ferromagnetic dimer model, with a Curie–Weiss correction for weak interdimer interactions (the solid line in Fig. 6), resulting in C = 0.394(1) emu K mol−1 Oe, J/KB = 104(3) K and θ = −1.59(2) K. We propose67–76 that the stronger ferromagnetic interaction occurs via the basal–apical bridge as is often observed and these dimeric units are then linked via the weaker basal–basal interaction. It has been reported that the basal–apical bis(EO)-bridged dicopper(II) units, like the {Cu2(N7)2} unit in 1, are characterized by ferromagnetic CuII⋯CuII exchange interactions.67,71 Examples are the 1D compounds [Cu2(EO-N3)2(ta)(phen)2]72 (ta is the bridging terephthalato dianion) with J/KB = 17.8 K and the dimeric complex [Cu2(EO-N3)2(L)2]73 (L is the bidentate chelating 7-amino-4-methyl-5-aza-3-heptan-2-onato monoanion) with J/KB = 34.6 K. Exceptions to this general trend are known, for example, in the dimeric complexes [Cu2(EO-N3)2Cl2(phen)2]74 (phen is 1,10-phenanthroline) and [Cu2(EO-N3)2(dpt)2](ClO4)275 (dpt is the tridentate chelating ligand dipropylenetriamine) with almost negligible CuII⋯CuII exchange interaction. From the structural and magnetic viewpoints, the closest complex to 1 is the 1D compound {[Cu(N3)2(quin)]}n76 (quin is quinoline), which also possesses alternating basal–apical and basal–basal bi(EO)azido bridges. Similarly to 1, this complex exhibits ferromagnetic coupling (J/KB = 6.6 K) with the onset of a weak antiferromagnetic coupling at low temperatures; the smaller J constant (compared to 1) may be due to the geometry of the CuII ion, which is intermediate between square pyramidal and trigonal bipyramidal (τ5 = 0.47 in the quinoline compound vs. 0.25 in 1). The structurally similar 1D compounds {[Cu(EO-N3)2L′]}n, where L′ is 2,4-dimethylpyridine,77 2,5-dimethylpyridine,78 and 2-chloropyridine,79 have not been studied by dc magnetometry.
The M as a function of H plot (Fig. S6) for compound 2 increases steadily to 50 kOe where it nears saturation and reaches a value of ∼5500 emu mol−1, in good agreement with the expected value for a S = 1/2 ion with g slightly greater than 2 (∼6000 emu mol−1). M as a function of T was then measured in a 1000 Oe field from 1.8 to 310 K. The χ value rises monotonically with decreasing T, reaching a value of 1.32 × 10−3 emu mol−1 Oe at 310 K. The data, plotted as χT versus T and 1/χ versus T, are shown in Fig. 7. The 1/χ versus T plot is linear over the entire temperature range and was fit to the Curie–Weiss law to yield a Curie constant C of 0.405(1) emu K mol−1 Oe and a Weiss constant θ of virtually zero [−0.04(4) K], indicating the absence of magnetic exchange interactions over the whole temperature range. The χT versus T data were also fit to the Curie–Weiss law with the same result within experimental error. The small upturn in this plot at the lowest temperatures could signal extremely weak ferromagnetic interactions but is much more likely due to a small error in the background measurement. The negligible (if any) CuII⋯CuII exchange interactions are reasonable, given the 2.101 (μ-1,5) coordination mode of N(CN)2− and the resultant very long (>7.5 Å) copper(II)–copper(II) distances (vide supra).23
Conclusions and perspectives
The chemical message of this work is the ability of N3− and N(CN)2− to form coordination polymers of copper(II) in the presence of 4,5-diphenylimidazole (HL). The complexes have interesting molecular and supramolecular structures, and the dimensionality (1D versus 2D) of the product depends on the pseudohalide ligand used. In addition, the azido complex exhibits both ferromagnetic and antiferromagnetic CuII⋯CuII exchange interactions. Efforts to isolate analogous coordination polymers using NCO− were in vain, illustrating the greater preference of this ion for terminal coordination modes; the only product from the CuII/NCO−/HL ligand ‘blend’ is mononuclear.The structural impact of the imidazole NH donor group on the supramolecular organization of the present crystal structures, in relation to the mononuclear complexes with the HL ligand reported in earlier studies,35–38 deserves to be noted. In nearly all compounds, whether discrete or polymeric, the NH groups are involved in synthon formation, either through direct intermolecular interactions or mediated by counterions. In a few cases where such interactions are sterically hindered, suitably positioned and oriented crystallization solvents (e.g., H2O, Me2CO, etc.) bridge the NH groups or form terminal H bonds with them. It is evident that the molecular self-assembly in all structures is effectively directed by recurring N–H⋯X synthons (X = halide, pseudohalide or solvent) irrespective of the structural type of the compound formed.
With the results and experience obtained in this work, our attempts are directed, among others, towards (1) efforts to prepare CuII/SCN−/HL and CuII/C(CN)3−/HL complexes to investigate the influence of different pseudohalides on the product identity; (2) the use of other divalent 1st-row transition metals, e.g., MnII, FeII, CoII and NiII, in this chemistry with the aim to obtain coordination polymers with different structural types and magnetic properties; and (3) the replacement of HL with other heavily substituted imidazoles, e.g., 1-methyl-4,5-diphenylimidazole (L), in order to explore their influence on the molecular structures of the CuII/X−/imidazole complexes [X− = N3−, N(CN)2−, NCO−, etc.], but mainly to investigate the absence of hydrogen at N1 on the supramolecular motifs of the products that are expected to be different from those observed in the present study. Studies on perspectives 2 and 3, mentioned above, are advanced and will be submitted in due course. The ID polymeric complexes {[Co(N3)2(HL)]}n and {[Cu(N3)2(L)]}n have been structurally characterized (Fig. S7 and S8), and the study of their properties is in progress.
Author contributions
Evangelia-Spyridoula Fotopoulou: data curation, formal analysis, investigation, and methodology; Nikos Panagiotou: data curation, formal analysis, and investigation; Anastasios J. Tasiopoulos: formal analysis and methodology; Mark M. Turnbull: data curation, formal analysis, and software editing – original draft; Spyros P. Perlepes: conceptualization, investigation, methodology, writing – original draft, and supervision; Vassilios Nastopoulos: conceptualization, investigation, writing – original draft, supervision, review and editing.Conflicts of interest
There are no conflicts to declare.Data availability
The authors declare that the data supporting the findings of this study are available within the paper and its SI: structural schemes (Scheme S1), figures (Fig. S1–S4, S7 and S8) and tables (Tables S1–S4), and magnetic plots (Fig. S5 and S6). See DOI: https://doi.org/10.1039/d5dt01389e.CCDC 2456934–2456936 contain the supplementary crystallographic data for this paper.80a–c
References
- J. L. Burmeister, Coord. Chem. Rev., 1990, 105, 77–133 CrossRef CAS.
- A. Escuer, J. Esteban, S. P. Perlepes and T. C. Stamatatos, Coord. Chem. Rev., 2014, 275, 87–129 CrossRef CAS.
- S. R. Gottam, C.-T. Tsai, L.-W. Wang, C.-Y. Li, C.-C. Lin and S.-Y. Chu, J. Electrochem. Soc., 2020, 167, 027513 CrossRef CAS.
- M. K. Chattopadhyay, P. Ghorai, S. Datta, N. C. Jana and P. Banerjee, Cryst. Growth Des., 2024, 24, 5781–5793 CrossRef CAS.
- P. Ghorai, A. Dey, A. Hazra, B. Dutta, P. Brandão, P. P. Ray, P. Banerjee and A. Saha, Cryst. Growth Des., 2019, 19, 6431–6447 CrossRef CAS.
- A. R. Jeong, J. W. Shin, J. H. Jeong, S. Jeoung, H. R. Moon, S. Kang and K. S. Min, Inorg. Chem., 2020, 59, 15987–15999 CrossRef CAS PubMed.
- E. Coronado and P. Day, Chem. Rev., 2004, 104, 5419–5448 CrossRef PubMed.
- D. R. Talham, Chem. Rev., 2004, 104, 5479–5501 CrossRef.
- M. Boukabene, H. Brahim, D. Hadji and A. Guendouzi, Theor. Chem. Acc., 2020, 139, 47 Search PubMed.
- K. Kumar, O. Stefanczyk, K. Nakabayashi, K. Imoto, Y. Oki and S.-i. Ohkoshi, Adv. Opt. Mater., 2022, 10, 2101721 Search PubMed.
- J. Hossain, J. S. Gopinath, S. Tothadi, P. Parameswaran and S. Khan, Organometallics, 2022, 41, 3706–3717 CrossRef.
- E. B. Anderson and M. R. Buchmeiser, Synlett, 2012, 23, 185–207 CrossRef.
- A. K. Boudalis, Y. Sanakis, J. M. Clemente-Juan, B. Donnadieu, V. Nastopoulos, A. Mari, Y. Coppel, J.-P. Tuchagues and S. P. Perlepes, Chem. – Eur. J., 2008, 14, 2514–2526 CrossRef.
- T. C. Stamatatos, K. A. Abboud, A. Wernsdorfer and G. Christou, Inorg. Chem., 2009, 48, 807–809 CrossRef.
- D. I. Alexandropoulos, L. Cunha-Silva, A. Escuer and T. C. Stamatatos, Chem. – Eur. J., 2014, 20, 13860–13864 CrossRef.
- O. Sato, J. Tao and Y.-Z. Zhang, Angew. Chem., Int. Ed., 2007, 46, 2152–2187 CrossRef PubMed.
- P. Gütlich, Y. Garcia and T. Woike, Coord. Chem. Rev., 2001, 219–221, 839–879 CrossRef.
- A. Escuer and G. Aromí, Eur. J. Inorg. Chem., 2006, 4721–4736 CrossRef.
- J. Ribas, A. Escuer, M. Monfort, R. Vicente, R. Cortes, L. Lezama and T. Rojo, Coord. Chem. Rev., 1999, 193–195, 1027–1068 CrossRef.
- X.-Y. Wang, Z.-M. Wang and S. Gao, Chem. Commun., 2008, 281–294 RSC.
- T. C. Stamatatos and G. Christou, Inorg. Chem., 2009, 48, 3308–3322 CrossRef PubMed.
- G.-C. Guo and T. C. W. Mak, Angew. Chem., Int. Ed., 1998, 37, 3268–3270 CrossRef.
- S. R. Batten and K. S. Murray, Coord. Chem. Rev., 2003, 246, 103–130 CrossRef.
- G. S. Papaefstathiou, A. Escuer, F. A. Mautner, C. Raptopoulou, A. Terzis, S. P. Perlepes and R. Vicente, Eur. J. Inorg. Chem., 2005, 879–893 CrossRef.
- J. Kohout, L. Jäger, M. Hvastijová and J. Kožišek, J. Coord. Chem. Rev., 2000, 51, 169–218 CrossRef.
- F. A. Mautner, P. Jantscher, R. C. Fischer, A. Torvisco, R. Vicente, T. N. V. Karsili and S. S. Massoud, Magnetochemistry, 2019, 5, 41 CrossRef.
- N. de la Pinta, S. Martin, M. K. Urtiaga, M. G. Barandika, M. I. Arriortuo, L. Lezama, G. Madariaga and R. Cortés, Inorg. Chem., 2010, 49, 10445–10454 CrossRef PubMed.
- A. Escuer, F. A. Mautner, N. Sanz and R. Vicente, Inorg. Chem., 2000, 39, 1668–1673 CrossRef PubMed.
- A. H. Norbury, Adv. Inorg. Chem. Radiochem., 1975, 17, 231–386 CrossRef.
- F. Meyer, E. Kaifer, P. Kircher, K. Heinze and H. Pritzkow, Chem. – Eur. J., 1999, 5, 1617–1630 CrossRef.
- J. Martínez-Lillo, D. Armentano, G. De Munno, F. Lloret, M. Julve and J. Faus, Inorg. Chim. Acta, 2006, 359, 4343–4349 CrossRef.
- M. H. Sadhu and S. B. Kumar, Polyhedron, 2017, 123, 419–429 CrossRef.
- F. A. Mautner, M. Scherzer, C. Berger, R. C. Fischer, R. Vicente and S. S. Massoud, Polyhedron, 2015, 85, 20–26 CrossRef.
- Effendy, C. Di Nicola, M. Fianchini, C. Pettinari, B. W. Skelton, N. Somers and A. H. White, Inorg. Chim. Acta, 2005, 358, 763–795 CrossRef.
- K. A. Kounavi, C. Papatriantafyllopoulou, A. J. Tasiopoulos, S. P. Perlepes and V. Nastopoulos, Polyhedron, 2009, 28, 3349–3355 CrossRef.
- K. A. Kounavi, M. J. Manos, E. Moushi, C. Papatriantafyllopoulou, A. J. Tasiopoulos and V. Nastopoulos, Cryst. Growth Des., 2012, 12, 429–444 CrossRef.
- K. A. Kounavi, E. Moushi, M. J. Manos, C. Papatriantafyllopoulou, A. J. Tasiopoulos and V. Nastopoulos, CrystEngComm, 2012, 14, 6492–6502 RSC.
- K. A. Kounavi, A. A. Kitos, E. Moushi, M. J. Manos, C. Papatriantafyllopoulou, A. J. Tasiopoulos, S. P. Perlepes and V. Nastopoulos, CrystEngComm, 2015, 17, 7510–7521 RSC.
- M.-I. Delegkou, N. Panagiotou, C. Papatriantafyllopoulou, A. J. Tasiopoulos, D. Papaioannou, S. P. Perlepes and V. Nastopoulos, CrystEngComm, 2024, 26, 3574–3592 RSC.
- E. Loukopoulos, E. Moushi, C. Papatriantafyllopoulou, A. A. Kitos, A. J. Tasiopoulos, S. P. Perlepes and V. Nastopoulos, Acta Crystallogr., Sect. B: Struct. Sci., Cryst. Eng. Mater., 2024, 80, 347–359 CrossRef.
- E. C. Constable, Metals and Ligand Reactivity: An Introduction to the Organic Chemistry of Metal Complexes, VCH, Weinheim, Germany, 1996, pp. 240–245 Search PubMed.
- S.-S. Chen, CrystEngComm, 2016, 18, 6543–6565 RSC.
- S. J. Lippard and J. M. Berg, Principles of Bioinorganic Chemistry, University Science Books, Mill Valley, California, USA, 1994 Search PubMed.
- R. M. Roat-Malone, Bioinorganic Chemistry, Wiley, New Jersey, USA, 2002 Search PubMed.
- G. R. Desiraju, Angew. Chem., Int. Ed. Engl., 1995, 34, 2311–2327 CrossRef.
- C. B. Aakeröy, N. R. Champness and C. Janiak, CrystEngComm, 2010, 12, 22–43 RSC.
- J. D. Dunitz and A. Gavezzotti, Cryst. Growth Des., 2012, 12, 5873–5877 CrossRef.
- X.-J. Yang, F. Drepper, B. Wu, W.-H. Sun, W. Haehnel and C. Janiak, Dalton Trans., 2005, 256–267 RSC.
- J. G. Malecki, J. Coord. Chem., 2011, 64, 390–399 CrossRef CAS.
- X. Zhang, S. Li, Y.-J. He, T. Han, X.-L. Wang, B. Chen, K.-Y. Zou and Z.-X. Li, Z. Anorg. Allg. Chem., 2015, 641, 1874–1880 CrossRef.
- D. Reinen, Comments Inorg. Chem., 1983, 2, 227–246 CrossRef.
- F. A. Cotton, G. Wilkinson, C. A. Murillo and M. Bochmann, Advanced Inorganic Chemistry, Wiley, New York, USA, 6th edn, 1999, pp. 854–876 Search PubMed.
- CrysAlis PRO, ver. 1.171.39.46, Rigaku Oxford Diffraction, Yarnton, UK, 2018 Search PubMed.
- A. Altomare, G. Cascarano, C. Giacovazzo, A. Guagliardi, M. C. Burla, G. Polidori and M. Camalli, J. Appl. Crystallogr., 1994, 27, 435 Search PubMed.
- G. M. Sheldrick, Acta Crystallogr., Sect. A: Found. Adv., 2015, 71, 3–8 CrossRef PubMed.
- L. Palatinus and G. Chapuis, J. Appl. Crystallogr., 2007, 40, 786–790 CrossRef.
- L. Palatinus, S. J. Prathapa and S. van Smaalen, J. Appl. Crystallogr., 2012, 45, 575–580 CrossRef.
- G. M. Sheldrick, Acta Crystallogr., Sect. C: Struct. Chem., 2015, 71, 3–8 Search PubMed.
- A. L. Spek, Acta Crystallogr., Sect. D: Biol. Crystallogr., 2009, 65, 148–155 CrossRef PubMed.
- O. V. Dolomanov, L. J. Bourhis, R. J. Gildea, J. A. K. Howard and H. Puschmann, J. Appl. Crystallogr., 2009, 42, 339–341 CrossRef.
- L. J. Farrugia, J. Appl. Crystallogr., 2012, 45, 849–854 CrossRef.
- C. F. Macrae, I. Sovago, S. J. Cottrell, P. T. A. Galek, P. McCabe, E. Pidcock, M. Platings, G. P. Shields, J. S. Stevens, M. Towler and P. A. Wood, J. Appl. Crystallogr., 2020, 53, 226–235 CrossRef CAS.
- A. W. Addison, T. N. Rao, J. Reedijk, J. van Rijn and G. C. Verschoor, J. Chem. Soc., Dalton Trans., 1984, 1349–1356 RSC.
- L. Yang, D. R. Powell and R. P. Houser, Dalton Trans., 2007, 955–964 RSC.
- K. Nakamoto, Infrared and Raman Spectra of Inorganic and Coordination Compounds, 4th edn, Wiley, New York, USA, 1986, pp. 388–390 Search PubMed.
- A. Escuer, M. A. S. Goher, F. A. Mautner and R. Vicente, Inorg. Chem., 2000, 39, 2107–2112 CrossRef CAS PubMed.
- T. C. Stamatatos, G. S. Papaefstathiou, L. R. MacGillivray, A. Escuer, R. Vicente, E. Ruiz and S. P. Perlepes, Inorg. Chem., 2007, 46, 8843–8850 CrossRef CAS PubMed.
- L. K. Thomson and S. S. Tandon, Comments Inorg. Chem., 1996, 18, 125–144 CrossRef.
- C. Adamo, V. Barone, A. Bencini, F. Totti and I. Ciofini, Inorg. Chem., 1999, 38, 1996–2004 CrossRef CAS.
- E. Ruiz, J. Cano, S. Alvarez and P. Alemany, J. Am. Chem. Soc., 1998, 120, 11122–11129 CrossRef CAS.
- K. Matsumoto, S.-I. Ooi, K. Nakatsuka, W. Mori, S. Suzuki, A. Nakahara and Y. Nakao, J. Chem. Soc., Dalton Trans., 1985, 2095–2100 RSC.
- L. Li, D. Liao, Z. Jiang and S. Yan, Polyhedron, 2001, 20, 681–684 CrossRef CAS.
- J. P. Costes, F. Dahan, J. Ruiz and J. P. Laurent, Inorg. Chim. Acta, 1995, 239, 53–59 CrossRef CAS.
- H. H. Hammud, U. Kortz, S. Bhattacharya, S. Demirdjian, E. Hariri, S. Isber, E. S. Choi, B. Mirtamizdoust, M. Mroueh and C. F. Daher, Inorg. Chim. Acta, 2020, 506, 119533 CrossRef CAS.
- P. Manikandan, R. Muthukumaran, K. R. J. Thomas, B. Varghese, G. V. R. Chandramouli and P. T. Manoharan, Inorg. Chem., 2001, 40, 2378–2389 CrossRef CAS PubMed.
- J.-P. Zhao, Y. Xie, J.-R. Li and X.-H. Bu, Dalton Trans., 2016, 45, 1514–1524 RSC.
- M. A. S. Goher, A. E. H. Abdou, M. A. M. Abu-Youssef and F. A. Mautner, Transition Met. Chem., 2001, 26, 39–43 CrossRef CAS.
- M. A. S. Goher, F. A. Mautner and N. A. Al-Salem, Polyhedron, 1997, 16, 2239–2248 CrossRef CAS.
- M. A. S. Goher and F. A. Mautner, Polyhedron, 1998, 17, 1561–1570 CrossRef CAS.
- (a) E.-S. Fotopoulou, N. Panagiotou, A. J. Tasiopoulos, M. M. Turnbull, S. P. Perlepes and V. Nastopoulos, CCDC 2456934: Experimental Crystal Structure Determination, 2025, DOI:10.5517/ccdc.csd.cc2ngmyq; (b) E.-S. Fotopoulou, N. Panagiotou, A. J. Tasiopoulos, M. M. Turnbull, S. P. Perlepes and V. Nastopoulos, CCDC 2456935: Experimental Crystal Structure Determination, 2025, DOI:10.5517/ccdc.csd.cc2ngmzr; (c) E.-S. Fotopoulou, N. Panagiotou, A. J. Tasiopoulos, M. M. Turnbull, S. P. Perlepes and V. Nastopoulos, CCDC 2456936: Experimental Crystal Structure Determination, 2025, DOI:10.5517/ccdc.csd.cc2ngn0t.
| This journal is © The Royal Society of Chemistry 2025 |