Template-driven synthesis of tin selenide nanosheets and their composites for supercapacitor applications
Nisha
Kushwah
a,
Divya
Nechiyil
b,
Jyoti
Prakash
 cd,
Suresh M.
Chopade
a,
Sushil Swaroop
Pathak
e,
Amey
Wadawale
a,
Sanjay
Kumar
f and
Gotluru
Kedarnath
cd,
Suresh M.
Chopade
a,
Sushil Swaroop
Pathak
e,
Amey
Wadawale
a,
Sanjay
Kumar
f and
Gotluru
Kedarnath
 *ad
*ad
aChemistry Division, Bhabha Atomic Research Centre, Trombay, Mumbai-400085, India. E-mail: kedar@barc.gov.in
bManipal Institute of Applied Physics, Manipal Academy of Higher Education, Manipal, Karnataka-576104, India
cMaterials Group, Bhabha Atomic Research Centre, Mumbai-400085, India
dHomi Bhabha National Institute, Anushaktinagar, Mumbai-400094, India
eLaboratoire de chimie de la matière condensée de Paris, Sorbonne Universite, Paris 75005, France
fMaterials Science Division, Bhabha Atomic Research Centre, Mumbai-400085, India
First published on 25th July 2025
Abstract
Bulk SnSe and SnSe2, with direct-band gaps of 1.3 eV and 1.84 eV, are promising materials for optoelectronics, lithium-ion batteries, thermoelectrics, and supercapacitors, due to their excellent electrochemical performance for energy storage. In this context, a new molecular precursor, [Me2Sn(SeC4H3N2)2], has been derived from bis(2-pyrazinyl)diselenide {(2-pyzSe)2} and structurally characterised by single crystal X-ray diffraction (sc-XRD) that serves as a building block for the fabrication of tin selenide (SnSe) nanosheets and tin selenide/g-C3N4 composites. The electrochemical performance of the synthesised nanosheets and composites was evaluated for their potential use in supercapacitor applications. The band gaps of rectangular SnSe (1.85 eV) and hexagonal SnSe2 (2.21 eV) nanosheets, and tin selenide/g-C3N4 composites (1.89 and 1.91 eV) exhibited a blue shift compared to those of bulk SnSe (Eg = 1.3 eV) and SnSe2 (Eg = 1.84 eV). The tin selenide/g-C3N4 composite utilised in supercapacitor applications exhibited a specific capacitance of 140 F g−1 at a current density of 1 A g−1. Remarkably, it retained 85% of this specific capacitance after 5000 cycles, demonstrating outstanding cycling durability.
Introduction
The increasing global demand for energy and environmental problems, accompanied by dwindling fossil fuel reserves, are compelling us to look at clean and sustainable renewable energy sources. To address these issues, advances in effective energy conversion and storage devices, viz. solar cells,1–4 electrochemical water splitting cells,5–8 batteries9 and supercapacitors,10 will be essential. Among these, supercapacitors (SCs) are remarkable electrochemical energy storage devices, which present higher power densities, better safety compliance and more prompt storage and release of charge compared to batteries. Furthermore, the SCs can operate in pollution-free and rapidly changing temperature conditions, leading to extended and enduring charge/discharge cycles. In this direction, 2D layered metal chalcogenide nanostructure-based energy conversion and storage devices can further lead to path-breaking technologies. Within the class of 2D layered metal chalcogenides, tin selenide has been verified as a suitable material for its applicability in the fields of thermoelectrics,1 photodetectors,2 solar cells,3 gas sensing,4 topological insulators,5 anode materials for batteries,6,9 supercapacitors10 and other related applications. Nevertheless, the category of application is strongly dependent on size, shape, composition, phase-driven properties, etc. Typically, tin selenide exists in two forms: SnSe and SnSe2. SnSe inherently grows in an orthorhombic crystal system with bulk direct- and indirect-energy gaps of 1.3 and 0.9 eV, respectively. In contrast, SnSe2, an innate n-type semiconductor, features a CdI2-type crystal lattice and has bulk direct- and indirect-energy gaps of 1.84 and 1.07 eV, respectively.9 The first form of tin selenide exhibits optical and structural anisotropy, making it suitable for photovoltaic3 and thermoelectric applications,10,11 while SnSe2 shows promise in superconductivity,12 alkali metal ion battery anodes,13 photocatalysis,14 and as an absorber for pulsed lasers.15Additionally, the focus has also been on SnSe for supercapacitor (SC) applications. However, glitches associated with the composition or phase of tin selenides are often found during their synthesis. Accordingly, quite a few methods, including solution-phase synthesis or solvent-less approaches have emerged over a period for the synthesis of nano-tin selenides by choosing either dual-source16–19 or single-source molecular precursor (SSMP)20–32 methods. In the case of dual-source methods, tin sources like Sn, SnCl2,19 SnCl4, tin-oleate complex, Sn6O4(OH)4, Sn[N(SiMe3)2]2, etc. and selenium sources such as Se,16 SeO2,18 and Se ligand/compound17 have been employed for the synthesis of tin selenide nanostructures with suitable selection of reaction conditions. When adopting the SSMP method, the complexes (Bz3Sn)2Se (Bz = benzyl),20 [Sn(SeC5H4N)2]2,21 [{Me2NC(NCy)2}2Sn![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) Se],22 [{(Me3Si)2N}2Sn(SePh)2],23 and [{Sn[N(Me3Si)2]2(μ-Se)}2]23 have been utilised for the synthesis of SnSe nanomaterials, whereas (Ph2SnSe)3
Se],22 [{(Me3Si)2N}2Sn(SePh)2],23 and [{Sn[N(Me3Si)2]2(μ-Se)}2]23 have been utilised for the synthesis of SnSe nanomaterials, whereas (Ph2SnSe)3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 24 yielded Sn-contaminated SnSe. While our group also has effectively employed quite a few dialkyl-tin complexes consisting of heterocyclic-based internally functionalised hemilabile ligands as molecular templates for the synthesis of either SnSe or SnSe2 nanostructures or thin films, by varying the alkyl groups either on tin or the ligand itself.25–28 Recently, Mathur et al. demonstrated the controlled decomposition of [SnIV(SeC2H4N(Me)C2H4Se)2] and [SnIVCl2(SeC2H4N(Me)C2H4Se)] into SnSe and SnSe2 phases.17 Moreover, [Sn(Ph2PSe2)2]29 and [Bu2Sn(PhCOSe)2]30 were employed for the deposition of SnSe thin films, while [SnCl4(nBu2Se)2]31 and [SnCl4{nBuSe(CH2)2SenBu}]32 were employed for the growth of SnSe2 thin films.
24 yielded Sn-contaminated SnSe. While our group also has effectively employed quite a few dialkyl-tin complexes consisting of heterocyclic-based internally functionalised hemilabile ligands as molecular templates for the synthesis of either SnSe or SnSe2 nanostructures or thin films, by varying the alkyl groups either on tin or the ligand itself.25–28 Recently, Mathur et al. demonstrated the controlled decomposition of [SnIV(SeC2H4N(Me)C2H4Se)2] and [SnIVCl2(SeC2H4N(Me)C2H4Se)] into SnSe and SnSe2 phases.17 Moreover, [Sn(Ph2PSe2)2]29 and [Bu2Sn(PhCOSe)2]30 were employed for the deposition of SnSe thin films, while [SnCl4(nBu2Se)2]31 and [SnCl4{nBuSe(CH2)2SenBu}]32 were employed for the growth of SnSe2 thin films.
Despite these achievements for the synthesis of tin selenide nanostructures, very few synthetic protocols for the synthesis of SnSe have been presented in the literature for supercapacitor applications.19,33 For instance, Zhang et al.34 have reported SnSe disc-shaped nanosheets which exhibit a specific capacitance of 210 F g−1 at 1 A g−1, while SnSe2 nano-discs showcase 144 F g−1 at 1 A g−1 with 99% retention of capacitance after 1000 cycles. Ni et al. reported a specific capacitance of 214.3 F g−1 for microwave-synthesised SnSe.16 Nevertheless, molecular template-based controlled synthesis of 2D SnSe nanosheets and their g-C3N4 composites for supercapacitor applications is rare to nonexistent, even though a number of merits have been listed for SSMPs.35,36 For instance, SSMPs in general comprise pre-existing M–E (M = metal and E = chalcogen) linkages, allowing molecular-level mixing, resulting in greater regulation of composition and defects. The judicious selection of ligands for SSMP synthesis is useful for appreciating the cleaner decomposition of SSMP at relatively low temperatures, ensuring high-quality target materials. Further, the versatility of SSMP as a source for both nanomaterial synthesis and film deposition makes it handy for device applications.35,36 Therefore, the synthesis of SnSe nanosheets for supercapacitor applications has been envisaged and implemented through the SSMP route.
With this perspective, a new and monomeric pyrizinylselenolate of Sn(IV), [Me2Sn(SeC4H3N2)2] (1), has been synthesised and unequivocally characterised using sc-XRD. Additionally, a modest and scalable synthesis of high-quality SnSe nanosheets and their g-C3N4 composites has been investigated for supercapacitor applications, utilising the aforementioned complex for the first time. Furthermore, a g-C3N4-induced phase transition from SnSe to SnSe2 has also been observed during the preparation of composites. A supercapacitor made up of a tin selenide-g-C3N4 composite exhibits a specific capacitance of 140 F g−1 at 1 A g−1 with 85% retention of this specific capacitance up to 5000 cycles. This indicates long cycle durability with reasonable specific capacitance for the tin selenide system.
Experimental section
Materials
Prior to use, all reagents were dried and purified using standard methods. Me2SnCl2, NaBH4, oleylamine (OAm), KOH and analytical grade solvents were purchased from commercial sources. The diselenide, {2-SeC4H3N2}2 (1H NMR (CDCl3) δ: 2.37 (s, Me); 6.71 (s, CH-5). 13C{1H} NMR(CDCl3) δ: 23.7 (Me); 117.2 (CH-5); 165.7 (C-2); 167.5 (C-4,6). 77Se{1H} NMR (CDCl3) δ: 486 ppm) was prepared according to a previously reported method in the literature.37The CNT films were synthesized using the floating-catalyst chemical vapor deposition (FC-CVD) method.38,39 Briefly, a catalyst solution comprising thiophene, ferrocene, and ethanol in a weight ratio of 0.3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2
2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 32 was preheated to 150 °C and introduced into the reactor at a flow rate of 0.2 mL min−1. Argon and hydrogen gases were used as carriers at a total flow rate of 1 L min−1. CNT soot was formed inside the reactor at 1250 °C, and the resulting CNT network was continuously collected in a glove box on a rotating steel rod, yielding a free-standing CNT film. The CNT aerogel films were immersed in a concentrated acid mixture of H2SO4 and HNO3 (3
32 was preheated to 150 °C and introduced into the reactor at a flow rate of 0.2 mL min−1. Argon and hydrogen gases were used as carriers at a total flow rate of 1 L min−1. CNT soot was formed inside the reactor at 1250 °C, and the resulting CNT network was continuously collected in a glove box on a rotating steel rod, yielding a free-standing CNT film. The CNT aerogel films were immersed in a concentrated acid mixture of H2SO4 and HNO3 (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 v/v) to improve surface wettability, thereby facilitating better electrolyte penetration and enhancing electrochemical performance. The resulting CNT substrate is highly conductive (∼5000 S cm−1), with a thickness of ∼100 μm, and serves as a current collector for the electrode material.
1 v/v) to improve surface wettability, thereby facilitating better electrolyte penetration and enhancing electrochemical performance. The resulting CNT substrate is highly conductive (∼5000 S cm−1), with a thickness of ∼100 μm, and serves as a current collector for the electrode material.
Physical measurements
All reactions were conducted under an anhydrous and inert atmosphere. A Thermo Fischer Flash EA1112 CHNS elemental analyser was employed for elemental analyses. Electronic spectra were acquired on a UV–vis Jasco V-630 spectrophotometer. The multinuclear NMR spectra (1H, 13C{1H} and 125Se{1H}) were obtained on a Bruker Avance-II spectrometer operating at 300, 75.47 and 57.24 MHz, respectively. Internal calibration of chemical shifts was performed relative to the chloroform peak for 1H and 13C{1H} NMR spectra while external calibration was undertaken in the case of 77Se{1H} NMR spectra with respect to Ph2Se2 (δ 463 ppm relative to Me2Se) in CDCl3. A NitzschSTA 409 PC-Luxx TG-DTA instrument calibrated with CaC2O4·H2O was used for thermogravimetric analyses (TGA). The TG curves were recorded at a heating rate of 10 °C min−1 under a flow of argon. A Philips PW-1820 diffractometer employing CuKα radiation was utilised to record powder X-ray diffraction patterns. A 532 nm wavelength from a DPSS laser (OXXIUS-LC-532) was utilised to record the Raman spectra. The Raman-scattered light was detected using a CCD (ANDOR)-based monochromator (ANDOR-SR-750C) together with an edge filter. The FE-SEM and EDS measurements were carried out on Zeiss Auriga and Oxford instruments, respectively. A Zeiss Libra 200 FE transmission electron microscope (TEM) operating at an accelerating voltage of 200 kV was used for TEM studies. The samples for TEM were prepared by placing a drop of sample dispersed in chloroform on a carbon-coated copper grid. Optical diffuse reflectance measurements in the range 200–1800 nm (0.68 eV to 6.2 eV) were performed on a JASCO V-670 two-beam spectrometer with a diffuse reflectance (DR) attachment consisting of an integration sphere coated with barium sulfate, which was used as a reference material. Measured reflectance data were converted to absorption (A) using the Kubelka–Munk remission function.40 The band gaps of the samples were estimated by extrapolating the linear portion of the plot to the X (energy) axis.X-ray crystallography
The crystallographic data for the complex [Me2Sn(SeC4H3N2)2] (1) were collected using CuKα radiation (λ = 1.54184 Å) from a single crystal at 298(2) K on an XtaLAB Synergy, Dualflex, HyPix four-circle diffractometer with a micro-focus sealed X-ray tube using a mirror as a monochromator and a HyPix detector. All data were integrated with CrysAlis PRO, and a multi-scan absorption correction using SCALE3 ABSPACK was applied.41 The structures were solved by iterative methods using OLEX and refined by full-matrix least-squares methods against F2 by SHELXL-2017/1.42,43 Hydrogen atoms were placed in idealised positions and were set riding on the respective parent atoms. All non-hydrogen atoms were refined with anisotropic thermal parameters. The structure was refined (weighted least squares refinement on F2) to convergence. The crystal and structure refinement data are detailed in Table S1, SI. All figures were drawn using ORTEP and Mercury.44,45 Crystallographic data (including structural factors) for the structures reported in this paper have been deposited with the Cambridge Crystallographic Data Centre (CCDC no. 2388509 for [Me2Sn(SeC4H3N2)2] (1)).Electrochemical measurements
To analyse the performance of the supercapacitor, various electrochemical measurements, including cyclic voltammetry (CV), galvanostatic charge–discharge (GCD), and electrochemical impedance spectroscopy (EIS) were conducted using a three-electrode configuration. The CNT and CNT-TS films were utilised as the working electrode (WE). The electrochemical measurements were carried out using a CH1760E Bipot electrochemical workstation. In the case of three-electrode measurements, the reference electrode (RE) employed was Ag/AgCl (1 M saturated KCl), the counter electrode (CE) consisted of Pt wire, and 1 M KOH was used as the electrolyte. CV and GCD curves of the functionalized CNT substrate films are shown in Fig. S1.Preparation of complex
Preparation of nanostructures and their composites
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 were performed to separate the SnSe nanosheets. The sample was designated TS-1 and characterised with different tools.
1 were performed to separate the SnSe nanosheets. The sample was designated TS-1 and characterised with different tools.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 were performed to separate the tin selenide nanocomposite. The sample was named TS-3 and characterized using different tools. In another experiment, the above procedure was repeated with only a change in the amount of g-C3N4 (7 mg). The resulting sample was named TS-4 and characterised.
1 were performed to separate the tin selenide nanocomposite. The sample was named TS-3 and characterized using different tools. In another experiment, the above procedure was repeated with only a change in the amount of g-C3N4 (7 mg). The resulting sample was named TS-4 and characterised.
Results and discussion
Chemical synthesis and characterisation
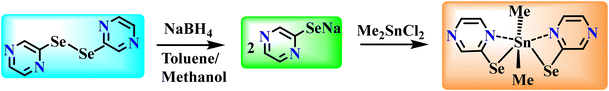
The reaction of a sodium salt of pyrazinylselenolate with dimethyltin dichloride in 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 stoichiometry afforded pyrazinylselenolate complex, [Me2Sn(SeC4H3N2)2] (1) (Scheme 1) as a light-orange solid, soluble in most organic solvents. This solid when recrystallised in chloroform offered rod-shaped crystals. The complex was probed by multinuclear NMR, supported by elemental analyses. The 1H and 13C{1H} NMR spectra presented the anticipated peaks and their multiplicities. The pyrazine ring proton resonances of [Me2Sn(SeC4H3N2)2] (1) were moved upfield relative to the equivalent signals for the pristine ligand. The 77Se{1H} NMR spectrum of [Me2Sn(SeC4H3N2)2] (1) presented only one peak at 180 ppm, pointing to the presence of a single species in the solution (ligand 77Se{1H} = 443 ppm). The 77Se{1H} NMR spectrum of complex 1 exhibited a single resonance at 180 ppm flanked by tin satellites with 1J(119Sn–77Se) = 718 Hz, indicating direct interaction of Sn and Se. The selenium resonance peak of the complex shows that it is shielded with respect to the selenium signal of the diselenide ligand.
1 stoichiometry afforded pyrazinylselenolate complex, [Me2Sn(SeC4H3N2)2] (1) (Scheme 1) as a light-orange solid, soluble in most organic solvents. This solid when recrystallised in chloroform offered rod-shaped crystals. The complex was probed by multinuclear NMR, supported by elemental analyses. The 1H and 13C{1H} NMR spectra presented the anticipated peaks and their multiplicities. The pyrazine ring proton resonances of [Me2Sn(SeC4H3N2)2] (1) were moved upfield relative to the equivalent signals for the pristine ligand. The 77Se{1H} NMR spectrum of [Me2Sn(SeC4H3N2)2] (1) presented only one peak at 180 ppm, pointing to the presence of a single species in the solution (ligand 77Se{1H} = 443 ppm). The 77Se{1H} NMR spectrum of complex 1 exhibited a single resonance at 180 ppm flanked by tin satellites with 1J(119Sn–77Se) = 718 Hz, indicating direct interaction of Sn and Se. The selenium resonance peak of the complex shows that it is shielded with respect to the selenium signal of the diselenide ligand.
The estimated value of 2J(119Sn–1H) of complex 1 is 74.9 Hz, less than the value anticipated for dimethyltin complexes with coordination number 6, while the magnitude of 1J(119Sn–13C) (∼483.7 Hz) is somewhat larger than those documented for tetra-coordinated diorganotin(IV) complexes.47 The ∠C–Sn–C estimated for [Me2Sn(SeC4H3N2)2] (1) using the equation below is 117.85°,48 while the same value determined by sc-XRD analysis is 127.32(16)°, indicating that the structure of complex 1 existing in the solid state also remains intact in the solution.
| |1J(119Sn–13C)| = 10.7(∠C–Sn–C) − 778 |
X-ray crystallography for structural determination
Recrystallisation of [Me2Sn(SeC4H3N2)2] (1) was achieved by slow evaporation of a solution of 1 in chloroform to yield rod-shaped pale-yellow crystals. The molecular structure of [Me2Sn(SeC4H3N2)2] (1) with atomic numbering scheme and its crystal packing are revealed in Fig. 1. Certain inter-atomic parameters (bond length and angles) are given in Tables S1 and S2. The complex grows in a monoclinic crystal system (space group P21/C), while the unit cell constitutes four molecules of complex 1 (Fig. 1). The central Sn atom of [Me2Sn(SeC4H3N2)2] (1) is located on a C2 axis with a distorted trapezoidal bipyramidal geometry defined by two methyl groups, and two selenium and two nitrogen atoms from two 2-pyrazine selenolate ligands, considering Sn⋯N weak interactions. The configuration around tin is similar to organo-tin pyridyl selenolates, [Me2Sn(2-SeC5H4N)2],22 [Me2Sn{2-SeC5H3(Me-5)N}2]24 and the analogous thiolato complex, [Me2Sn(SC5H2N)2].49The Sn–Se bond distance (2.6175(4) and 2.6090(4)) are in agreement with those reported for [tBu2Sn{SeC4H(Me-4,6)2N2}2] (2.615(3) and 2.618(3) Å) and [tBu2Sn{SeC4H(Me-4,6)2N2}Cl] (2.583(3) and 2.595(3) Å)23 and shorter than those reported for [Sn(SeC5H4N)2]2 (2.681, 2.759 Å)17 [Sn(SePh)2] (2.668, 2.675, 2.683, 2.673 Å)50 and [tBu2Sn(2-SeC5H4N)2] (2.622(2) Å).25,51 The molecule is slightly puckered with Sn⋯N distances (2.860 and 2.679 Å) longer than their covalent radii (2.15 Å), but are significantly smaller than the sum of their van der Waals (vdW) radii (3.72 Å),52 suggesting a weak intramolecular interaction between tin and nitrogen atoms and in agreement with those reported for [Me2Sn{2-SeC4H(Me-4,6)2N}2] (2.671 Å).28 These non-covalent interactions between Sn and tin present a case of chelation leading to a stable complex. The hexacoordination around tin is further corroborated by the 1J(119Sn–13C) value of ∼718 Hz, which falls in the range that is observed for six-coordination compounds of tin,53 indicating that the weak interactions detected in the solid state are also present in solution. This is in contrast to what has been observed in [Me2Sn{2-SeC5H2(Me-4,6)2N}2].28 The Sn–C distances (2.118(4) and 2.126(3) Å) are in agreement with those of similar diorganotin pyridyl and pyrimidyl complexes25–27,48 along with complexes of type [Me2Sn(SC5H4N)2] (∼2.13 Å) and [Me2Sn(C9H6NO)2] (∼2.15 Å).48,54 The Se–Sn–Se bond angle in dimethyl tin complexes of pyridyl and pyrimidyl selenide are in the range of 91.23(4) to 94.25(8)°, while in the present case, the same angle falls at the lower end of the range, at 91.524(13)°.22–25 The angles, ∠N–Sn–N and ∠Se–Sn–Se in 1 (145.10 and 91.57(2)°, respectively) are squeezed vis-à-vis the same angles in [Me2Sn(SeC5H4N)2] (152.1(2) and 94.25(8)°),25 whereas ∠Se–Sn–Se is opened by ∼4.5° relative to ∠S–Sn–S of bis[3-(2-pyridyl)-2-thienyl]di-p-tolylstannane (86.91(7)°).55 Opening of ∠Se–Sn–Se with respect to ∠S–Sn–S might be expected because of the presence of a relatively more bulky Se atom in the former compared to the S atom in the latter. The N–Sn–Se bite angles (62.59° and 60.91°) of complex 1 are similar to those of the analogous dimethyltin complex with a 2-selenopyridyl ligand, [Me2Sn(SeC5H4N)2] (62.61° and 60.69°),22 demonstrating that the ring effect has no impact on ∠N–Sn–Se bite angles.
The crystal packing of 1 in 3D space shows that the neighbouring [Me2Sn(SeC4H3N2)2] (1) molecules are held together by short contacts between the Se2 atoms of two adjacent asymmetric units and the N2 atom of the asymmetric unit with the H atom of the C10B–H10B fragment of the nearby unit. The Se⋯Se distance (3.685 Å) is shorter than the vdW radius of the Se atoms (3.8 Å), suggesting weak secondary interactions. In contrast, the C10–H10B⋯N2 distances (C10–H10B⋯N2 = 2.693 Å) are shorter than the vdW radii of the two atoms (3.2 Å), signifying that strong non-bonding secondary interactions hold neighboring asymmetric units together. Additionally, adjoining asymmetric units in the same layer are sliding over each other through C–H⋯N interactions, while adjacent molecules in two bordering layers are linked together by weak Se⋯Se interactions.
Thermogravimetric analysis
Thermal traits of complex 1 were examined by thermogravimetric analysis (TGA) and differential thermogravimetry (DTG) (Fig. S2, SI) in the range of 30–1000 °C under a flowing argon atmosphere. The complex underwent a two-step decomposition. The first step had an onset temperature of 210 °C and a completion temperature of 300 °C with a weight loss of 83%. However, the experimental weight loss of 83% is significantly greater than the expected weight losses for SnSe (57.48%) and Sn (74.46%), pointing out the substantial volatility of the precursor, in turn making the precursor suitable for CVD applications.56 The volatility of the precursor is corroborated by the presence of volatile pyrazine moieties in complex 1. Additionally, a steady weight loss was noted even after 520 °C, which might be because of the loss of volatile tin.Fabrication and characterisation of tin selenide nanostructures and their g-C3N4 composites
Gathering thermal behavioural inputs of complex 1 from the above measurements, a synthesis protocol for tin selenide (SnSe) nanosheets and their g-C3N4 composites has been planned by the thermolysis of [Me2Sn(SeC4H3N2)2] (1) in the high-boiling coordinating solvent, oleylamine (OAm). A thermolysis temperature of 250 °C was preferred for the decomposition, even though the required decomposition temperature obtained from TG is 292 °C. This may be vindicated by the lowering of the thermal degradation temperature as a consequence of solvation of the complex by the coordinating solvent.57 The selection of OAm as a solvent for thermolysis was driven by many factors: its high boiling point of 360 °C, the strong affinity of the nitrogen of the –NH2 group for tin, leading to effective surface passivation of nanostructures and its role in catalysing of the thermal disintegration of the complex at relatively low temperatures compared to its solid-state decomposition temperature, as predicted by TG curves.58 Thus, tin selenide nanosheets were synthesised by the heat-up method in OAm at 250 °C for 10 min. The p-XRD patterns (Fig. S3, SI) of the same exhibit Bragg's reflections at 2θ = 21.48, 25.35, 29.38, 30.62, 37.86, 41.38, 43.50, 44.30, 47.30, 49.70, 52.64, 54.21 and 57.50°, corresponding to orthorhombic SnSe (ICSD no. 12863; JCPDS no. 48-1224). These peaks can be marked as the reflections emanating from (101), (201), (011), (111), (311), (102), (202), (501), (112), (511), (312), (420) and (502), whereas peak broadening specifies that the particle dimensions are in the nano-region. In contrast, a tin selenide composite prepared through pyrolysis revealed reflections matching trigonal SnSe2 (ICSD no. 43594; JCPDS no. 23-0602). While p-XRD patterns of g-C3N4 composites of tin selenide with different weight percentages of g-C3N4 prepared by thermolysis revealed peaks consistent with orthorhombic SnSe (ICSD no. 12863; JCPDS no. 48-1224) along with trigonal SnSe2 (ICSD no. 43594; JCPDS no. 23-0602) (Fig. S4, SI). The extent of formation of SnSe2 increases with the increasing amount of g-C3N4 used during the synthesis of the composites, which is determined by the relative intensities of SnSe and SnSe2 peaks. The details of the reaction conditions and the phase of the material are listed in Table S3, SI.Notably, the introduction of g-C3N4 to prepare an SnSe/g-C3N4 composite through a thermolysis experiment yields an SnSe–SnSe2/g-C3N4 composite instead of the required product, which has been designated as a tin selenide composite. This indicates that the addition of g-C3N4 drives the formation of SnSe2 phase during the preparation of the composite. Furthermore, an attempt to prepare an SnSe/g-C3N4 composite by the pyrolysis of [Me2Sn(SeC4H3N2)2] (1) and g-C3N4 together in a furnace yields the SnSe2/g-C3N4 composite (TS-2). The phase purity of these nanostructures and composites is further validated by their elemental compositions according to EDS (Fig. S5–S11, SI). These compositional studies revealed that the atom% ratio of Sn![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Se is 1.01
Se is 1.01![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.00 and 1
1.00 and 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.92 for TS-1 and TS-2, respectively, confirming the SnSe and SnSe2 compositions for these samples. Moreover, the uniform distribution of the integral elements within these samples was verified by 2D elemental mapping analysis.
1.92 for TS-1 and TS-2, respectively, confirming the SnSe and SnSe2 compositions for these samples. Moreover, the uniform distribution of the integral elements within these samples was verified by 2D elemental mapping analysis.
The phase purity of rectangular SnSe nanosheets (TS-1) and hexagonal SnSe2 (TS-2) was further evaluated by Raman spectroscopy in order to examine the binary-phase impurities present in the samples through the phonon modes of the nanomaterial. Further, the presence of g-C3N4 in composite samples can also be confirmed by Raman spectroscopy. The Raman spectrum of the TS-1 sample (Fig. S12, SI) exhibits a peak at 106 cm−1 and a shoulder at 156 cm−1 corresponding to the Bg and Ag modes of SnSe, while TS-2 shows peaks at 111 and 185 cm−1 related to in-plane Eg and out-of-plane A1g modes of SnSe2, consistent with literature reports for SnSe59 and SnSe2.60
The morphology, dimension and phase of TS-1 to TS-4 were determined by FE-SEM, TEM and selective area electron diffraction (SAED) patterns. SEM images of TS-1 and TS-2 revealed rectangular sheets and hexagonal sheets, as shown in Fig. 2(a) and (b), while SEM images of TS-3 and TS-4 revealed a mixture of rectangular and hexagonal sheets (Fig. 2(c) and (d)). TEM images of TS-1 to TS-4 (Fig. 2(e–h)) confirm the same. The average thickness of rectangular sheets (TS-1) and hexagonal sheets (TS-2) is 46 and 65 nm, respectively (Table S3, SI), while the lateral dimensions of the hexagonal sheets are in the range of 350–450 nm. SAED patterns of rectangular and hexagonal sheets exhibit lattice planes, (111), (511), (502) (Fig. 2(i)) and (002), (101), (103) (Fig. 2(j)), matching orthorhombic SnSe (ICSD no. 12863/JCPDS no. 48-1224) and trigonal SnSe2 (ICSD no. 43594; JCPDS no. 23-0602) phases, respectively.
In contrast, the SAED patterns of TS-3 (Fig. 2(k)) and TS-4 (Fig. 2(l)) reveal the lattice planes (111) (SnSe), (011), (102), (003) (SnSe2) and (201), (502) (SnSe), (002), (400), (110), (013) (SnSe2), respectively, corresponding to orthorhombic SnSe and trigonal SnSe2 indicating that both TS-3 and TS-4 contain a mixture of SnSe and SnSe2, in conformity with the respective SEM and TEM images. The discrepancy between p-XRD and TEM sizes is mainly due to the dependency of the full width at half maximum of the p-XRD peak (which can be used to estimate particle size using the Scherrer equation) on crystal distortions, defects and instrumental broadening in addition to scattering domain size.61
Exploring the optical behaviour of tin selenide nanosheets and g-C3N4 nanocomposites
SnSe and SnSe2 have direct-band gaps of 1.3 (ref. 41) and 1.84 eV,62 respectively, with an absorption coefficient of ∼105 cm−2, a high carrier concentration and p-type conductivity, and they show significant applicability in solar photovoltaics,63 thermoelectrics64 photodetectors, anode materials for batteries65 and supercapacitors.66 Besides, the effectiveness of these applications can be further boosted by replacing the bulk materials with their nanostructures, due to dimension-driven and morphology-driven changes in properties. Of these properties, the optical band gap is worth investigating as it has an impact on the efficiency of these applications. Hence, probing of the optical bandgap of the synthesised nanosheets and composites by diffused reflectance spectroscopy (DRS) is worthwhile. These band gap measurements were carried out by plotting [F(R)]nvs. energy (hν), where F(R) is the Kubelka–Munk (KM) function, which can be stated as follows:| [F(R)]n = A(hν − Eg) |
 | ||
| Fig. 3 Plots of [F(R)]2vs. energy (eV) created by KM transformation of DRS data of TS-1 to TS-4, for calculating direct-band gap energies. | ||
As Bohr's radius of bulk SnSe is around 23 nm (ref. 67) and the thickness of rectangular sheets (TS-1) and hexagonal sheets (TS-2) in the present study is 46 and 65 nm, respectively, quantum confinement may be ruled out. Hence, the blue shift in band gap values can be ascribed either to lattice distortion or to surface lattice defects. Nevertheless, band gap values of all the samples (TS-1 to TS-4) are in the range 1.85–2.21 eV, indicating their reasonable conductivity and suitability as materials for supercapacitors.
Electrochemical performance of tin selenide nanosheets and composite nanostructures
The pristine SnSe rectangular nanosheets (TS-1) and tin selenide/g-C3N4 composites (TS-2, TS-3 and TS-4) with band gaps in the range of 1.85–2.21 eV are appropriate for a supercapacitor. Given this, these samples were evaluated for supercapacitor applications, and cyclic voltammetry (CV), electrochemical impedance spectrometry (EIS), galvanostatic charge–discharge (GCD), and cyclic stability studies were performed in a three-electrode setup employing a 1 M KOH solution as the electrolyte to investigate the supercapacitor performance.To develop flexible supercapacitors, all solid-state supercapacitors (SSCs) were employed using SnSe nanosheets (TS-1) and their g-C3N4 composites (TS-2, TS-3 and TS-4) as active materials. The energy storage performance of these samples was studied by coating the samples on CNT film, as reported elsewhere46 and using these coated CNT films as electrodes. Fig. 4(a) and (b) depict CV analytical results performed on TS-1 to TS-4 electrodes. The CV plots of CNT-TS (Fig. 4(a)) were acquired at a scan rate of 100 mV s−1 and a potential range of −0.6 to 0.4 V. The CV plots are symmetric and nearly rectangular due to the surface electro-sorption and the subsequent reversible surface redox reactions of SnSe nanosheets (TS-1) using cations/anions.
The larger surface area under the CV curve of TS-3, relative to the other electrodes, specifies a higher capacitance for the TS-3 electrode. GCD measurements were performed to evaluate the specific capacitance of the flexible electrodes. Fig. 4(b) reveals the GCD shapes of the TS-1 to TS-4 electrodes. All TS electrodes display a non-linear GCD profile, representing the pseudocapacitive nature of faradaic reactions in the TS. The TS-3 electrode shows an extended charge–discharge time, signifying its higher capacitance relative to the other electrodes. This improvement can be attributed to the synergistic effect between SnSe nanosheets and g-C3N4. The incorporation of g-C3N4 enhances the electrochemical pseudocapacitance by providing additional redox-active sites, increasing surface area, and facilitating better ion diffusion throughout the electrode matrix. The specific capacitance (Cs) is determined with the following formula:
| Cs = (I × Δt)/(m × ΔV) |
Conclusions
It is notable that the synthesis of phase-pure tin selenides employing dual sources consistently involves subtle nuances. Therefore, the molecular template method for the synthesis of quality tin selenide nanostructures is simple and accessible. Consequently, a new, air-stable and moisture-stable pyrizinylselenolate of tin(IV), [Me2Sn(SeC4H3N2)2], has been produced, structurally characterized and used as a molecular template for the synthesis of SnSe nanosheets and tin selenide/g-C3N4 composites. These materials were evaluated for their crystal structure, shape, and energy gap. Experimentally determined optical energy gaps of these materials are in a suitable range for a supercapacitor material. A supercapacitor made up of a tin selenide/g-C3N4 composite (TS-3) remained stable for more than 5000 cycles and established a specific capacitance of 140 F g−1 at a current density of 1 A g−1, showing good cyclability with reasonable specific capacitance. Even though the molecular template method is quite versatile for metal chalcogenides, the applicability of these materials for supercapacitors is still to be determined. Hence, the current study of the molecular templated synthesis of tin selenide and its composites for supercapacitor applications can overcome the application blockade.Author contributions
N. K., D. N., and J. P. were responsible for data curation, formal analysis, investigation, validation, visualisation, and writing – original draft. S. S. P. was responsible for conceptualisation, data curation, formal analysis, investigation, validation, resources, and writing – review & editing. S. C. contributed to data curation, formal analysis and investigation. A. W. and S. K. contributed to data curation, investigation, and validation. G. K. was responsible for conceptualisation, formal analysis, investigation, methodology, project administration, resources, supervision, validation, writing – original draft and writing – review & editing.Conflicts of interest
The authors declare no conflicts of interest.Data availability
EDS spectra and 2D mapping of the constituent elements of TS-1 to TS-4. See DOI: https://doi.org/10.1039/d5dt01255d.CCDC 2388509 contains the supplementary crystallographic data for this paper.68
Acknowledgements
We would like to express our gratitude to Dr A. C. Bhasikuttan, Associate Director of the Chemistry Group and Dr N. Choudhury, Chemistry Division at Bhabha Atomic Research Centre, for their support of this work. We also extend our thanks to the Head of the Radiation Technology Development Division (RTDD) for their assistance with the TG measurements. S. S. P. gratefully acknowledges Prof. David Portehault, Laboratoire de Chimie de la Matière Condensée de Paris (LCMCP), Sorbonne Université, for his kind support.References
- W. Shi, M. Gao, J. Wei, J. Gao, C. Fan, E. Ashalley, H. Li and Z. Wang, Adv. Sci., 2018, 5, 1700602–1700624 CrossRef PubMed.
- M. Kang, S. Rathi, I. Lee, L. Li, M. A. Khan, D. Lim, Y. Lee, J. Park, A. T. Pham, A. T. Duong, S. Cho, S. J. Yun and G. H. Kim, J. Nanosci. Nanotechnol., 2017, 18, 4243–4247 CrossRef PubMed.
- Z. Li, Y. Guo, F. Zhao, C. Nie, H. Li, J. Shi, X. Liu, J. Jiang and S. Zuo, RSC Adv., 2020, 10, 16749–16755 RSC.
- X. Wang, Y. Liu, J. Dai, Q. Chen, X. Huang and W. Huang, Chem. – Eur. J., 2020, 26, 3870–3876 CrossRef CAS PubMed.
- Y. Sun, Z. Zhong, T. Shirakawa, C. Franchini, D. Li, Y. Li, S. Yunoki and X. Q. Chen, Phys. Rev. B: Condens. Matter Mater. Phys., 2013, 88, 235122 CrossRef.
- Z. Li, J. Ding and D. Mitlin, Acc. Chem. Res., 2015, 48, 1657–1665 CrossRef CAS PubMed.
- S. S. Pathak, S. Dey, A. Kumar, S. Gamre, T. Gadly and G. Kedarnath, J. Phys. Chem. C, 2024, 128(19), 8058–8067 CrossRef CAS.
- Y. Tomar, S. S. Pathak, S. Jain and L. S. Panchakarla, Isr. J. Chem., 2022, 62, e202100080 CrossRef CAS.
- J. M. Gonzalez and I. I. Oleynik, Phys. Rev. B, 2016, 94, 125443 CrossRef.
- C. Chang, M. Wu, D. He, Y. Pei, C. F. Wu, X. Wu, H. Yu, F. Zhu, K. Wang, Y. Chen, L. Huang, J. F. Li, J. He and L. D. Zhao, Science, 2018, 360, 778–783 CrossRef CAS PubMed.
- L. D. Zhao, S. H. Lo, Y. S. Zhang, H. Sun, G. J. Tan, C. Uher, C. Wolverton, V. P. Dravid and M. G. Kanatzidis, Nature, 2014, 508, 373–377 CrossRef CAS PubMed.
- Z. Shao, Z. G. Fu, S. Li, Y. Cao, Q. Bian, H. Sun, Z. Zhang, H. Gedeon, X. Zhang, L. Liu, Z. Cheng, F. Zheng, P. Zhang and M. Pan, Nano Lett., 2019, 19, 5304–5312 CrossRef CAS PubMed.
- Z. Wei, L. Wang, M. Zhuo, W. Ni, H. Wang and J. Ma, J. Mater. Chem. A, 2018, 6, 12185–12214 RSC.
- Y. Fan, J. Wang and M. Zhao, Nanoscale, 2019, 11, 14836–14843 RSC.
- M. Wang, Z. Wang, X. Xu, S. Duan and C. Du, Nanotechnology, 2019, 30, 265703 CrossRef CAS PubMed.
- D. Ni, Y. Chen, X. Yang, C. Liu and K. Cai, J. Alloys Compd., 2018, 737, 623–629 CrossRef CAS.
- F. Hartl, V. Brune, S. Lüggert, C. Hegemann, D. van Gerven, M. Wilhelm, S. Ji, H. Choi and S. Mathur, Inorg. Chem., 2023, 62, 6274–6287 CrossRef CAS PubMed.
- S. Wu, S. Zhou, Q. Feng, H. Zhao, X. Xu, T. Cui, H. Zhang, X. Wang and Q. Yang, J. Energy Storage, 2021, 42, 103032 CrossRef.
- B. Pandit, C. D. Jadhav, P. G. Chavan, H. S. Tarkas, J. V. Sali, R. B. Gupta and B. R. Sankapal, IEEE Trans. Power Electron., 2020, 35, 11344–11351 Search PubMed.
- P. Boudjouk, D. J. Seidler, D. Grier and G. J. McCarthy, Chem. Mater., 1996, 8, 1189–1196 CrossRef CAS.
- Y. Cheng, T. J. Emge and J. G. Brennan, Inorg. Chem., 1996, 35, 342–346 CrossRef CAS PubMed.
- I. Y. Ahmet, J. R. Thompson and A. L. Johnson, Eur. J. Inorg. Chem., 2018, 1670–1678 CrossRef CAS.
- J. R. Thompson, I. Y. Ahmet, A. L. Johnson and G. Kociok-Köhn, Eur. J. Inorg. Chem., 2016, 4711–4720 CrossRef CAS.
- S. R. Bahr, P. Boudjouk and G. J. McCarthy, Chem. Mater., 1992, 4, 383–388 CrossRef CAS.
- R. K. Sharma, G. Kedarnath, A. Wadawale, C. A. Betty, B. Vishwanadh and V. K. Jain, Dalton Trans., 2012, 41, 12129–12138 RSC.
- A. Tyagi, G. Kedarnath, A. Wadawale, A. Y. Shah, V. K. Jain and B. Vishwandh, RSC Adv., 2016, 6, 8367–8376 RSC.
- A. Tyagi, G. Karmakar, A. Wadawale, A. Y. Shah, G. Kedarnath, A. P. Srivastava, V. Singh and V. K. Jain, J. Organomet. Chem., 2018, 873, 15–21 CrossRef CAS.
- G. Karmakar, K. K. Halankar, A. Tyagi, B. P. Mandal, A. P. Wadawale, G. Kedarnath, A. P. Srivastava and V. Singh, Dalton Trans., 2021, 50, 15730–15742 RSC.
- P. Kevin, S. N. Malik, M. A. Malik and P. O'Brien, Chem. Commun., 2014, 50, 14328–14330 RSC.
- M. D. Khan, M. Aamir, M. Sohail, M. Sher, N. Baig, J. Akhtar, M. A. Malik and N. Revaprasadu, Dalton Trans., 2018, 47, 5465–5473 RSC.
- C. Gurnani, S. L. Hawken, A. L. Hector, R. Huang, M. Jura, W. Levason, J. Perkins, G. Reid and G. B. G. Stenning, Dalton Trans., 2018, 47, 2628–2637 RSC.
- C. H. de Groot, C. Gurnani, A. L. Hector, R. Huang, M. Jura, W. Levason and G. Reid, Chem. Mater., 2012, 24, 4442–4449 CrossRef CAS.
- M. A. Dar, S. R. Ahmed, M. A. Rather, S. Kalpana, A. A. Rather, S. R. Ahamed and Z. Ahmad, Inorg. Chem. Commun., 2023, 153, 110838 CrossRef CAS.
- C. Zhang, H. Yin, M. Han, Z. Dai, H. Pang, Y. Zheng, Y. Q. Lan, J. Bao and J. Zhu, ACS Nano, 2014, 8, 3761–3770 CrossRef CAS PubMed.
- G. Kedarnath, in Handbook on synthesis strategies for advanced materials, ed. A. K. Tyagi and R. S. Ningthoujam, Springer Singapore, Singapore, 2021, vol. 15, pp. 467–501 Search PubMed.
- G. Kedarnath and V. K. Jain, New J. Chem., 2023, 47, 20688 RSC.
- N. Kushwah, S. Chopade, A. Wadawale, P. B. Sharma and G. Kedarnath, RSC Adv., 2023, 13, 36392 RSC.
- A. Kaushal, R. Alexander, D. Mandal, J. B. Joshi and K. Dasgupta, Chem. Eng. J., 2023, 452, 139142 CrossRef CAS.
- R. Alexander, A. Kaushal, J. Prakash, P. T. Rao, D. Sen and K. Dasgupta, J. Porous Mater., 2023, 30, 507–520 CrossRef CAS.
- B. Philips-Invernizzi, D. Dupont and C. Cazé, Opt. Eng., 2001, 40, 1082–1092 CrossRef.
- P. D. Antunez, J. J. Buckley and R. L. Brutchey, Nanoscale, 2011, 3, 2399–2411 RSC.
- Rigaku Oxford Diffraction, CrysAlis PRO, Rigaku Oxford Diffraction, Yarnton, England, 2015 Search PubMed.
- O. V. Dolomanov, L. J. Bourhis, R. J. Gildea, J. A. K. Howard and H. Puschmann, J. Appl. Crystallogr., 2009, 42(2), 229–341 CrossRef.
- G. M. Sheldrick, Acta Crystallogr., Sect. C, 2015, 71, 3–8 CrossRef PubMed.
- (a) L. J. Farrugia, J. Appl. Crystallogr., 1997, 30, 565 CrossRef CAS; (b) C. F. Macrae, P. R. Edgington, P. McCabe, E. Pidcock, G. P. Shields, R. Taylor, M. Towler and J. van der Streek, J. Appl. Crystallogr., 2006, 39, 453–457 CrossRef CAS.
- D. Nechiyil, J. Mor, R. Alexander, S. K. Sharma, K. Dasgupta and J. Prakash, J. Energy Storage, 2024, 85, 111207 CrossRef.
- C. Vatsa, Ph. D. Thesis, University of Mumbai, 1990.
- T. P. Lockhart and F. Davidson, Organometallics, 1987, 6, 2471–2478 CrossRef CAS.
- M. V. Castaño, A. Macías, A. Castiñeiras, A. S. González, E. G. Martínez, J. S. Casas, J. Sordo, W. Hiller and E. E. Castellano, J. Chem. Soc., Dalton Trans., 1990, 1001–1005 RSC.
- A. Eichhöfer, J. J. Jiang, H. Sommer, F. Weigend, O. Fuhr, D. Fenske, C. Y. Su and G. Buth, Eur. J. Inorg. Chem., 2010, 410–418 CrossRef.
- J. Otera, T. Yano and K. Kusakabe, Bull. Chem. Soc. Jpn., 1983, 56, 1057–1059 CrossRef CAS.
- A. Bondi, J. Phys. Chem., 1964, 68, 441–452 CrossRef CAS.
- T. P. Lockhart, W. F. Manders and J. J. Zuckerman, J. Am. Chem. Soc., 1985, 107, 4546–4547 CrossRef CAS.
- E. O. Schlemper, Inorg. Chem., 1967, 6, 2012–2017 CrossRef CAS.
- P. J. Smith and A. P. Tupciauskas, Annu. Rep. NMR Spectrosc., 1978, 8, 291–370 CrossRef CAS.
- J. R. Thompson, I. Y. Ahmet, A. L. Johnson and G. Kociok-Köhn, Eur. J. Inorg. Chem., 2016, 4711–4720 CrossRef CAS.
- N. Kushwah, G. Kedarnath, A. Wadawale, K. K. Halankar, B. P. Mandal, M. Jafar and B. Vishwanadh, Inorg. Chem., 2024, 62, 8823–8834 CrossRef PubMed.
- M. D. Khan, M. Akhtar, M. A. Malik, N. Revaprasadu and P. O'Brien, ChemistrySelect, 2018, 3, 2943–2950 CrossRef CAS.
- X. Z. Li, J. Xia, L. Wang, Y. Y. Gu, H. Q. Cheng and X. M. Meng, Nanoscale, 2017, 9, 14558 RSC.
- H. Guan, H. Li, J. Ming, J. Hong, J. Dong, W. Qiu, W. Zhu, J. Yu, Z. Chen, G. Peng and H. Lu, J. Lightwave Technol., 2020, 38, 6089–6096 CAS.
- (a) L. J. Farrugia, J. Appl. Crystallogr., 1997, 30, 565 CrossRef CAS; (b) C. F. Macrae, P. Edgington, P. McCabe, E. Pidcock, G. P. Shields, R. Taylor, M. Towler and J. van der Streek, J. Appl. Crystallogr., 2006, 39, 453–457 CrossRef CAS.
- M. Kumar, S. Rani, Y. Singh, K. S. Gour and V. N. Singh, RSC Adv., 2021, 11, 6477–6503 RSC.
- M. Kumar, S. Rani, Y. Singh, Mamta, A. Kumar and V. N. Singh, Sol. Energy, 2022, 232, 146–153 CrossRef CAS.
- M. W. van de Putte and M. Huijben, Appl. Surf. Sci., 2023, 623, 157034 CrossRef CAS.
- Z. Wei, L. Wanga, M. Zhuo, W. Ni, H. Wang and J. Ma, Mater. Chem. A, 2018, 6, 12185–12214 RSC.
- S. Harish and P. U. Sathyakam, J. Energy Storage, 2022, 52, 104966 CrossRef.
- J. J. Buckley, F. A. Rabuffetti, H. L. Hinton and R. L. Brutchey, Chem. Mater., 2012, 24, 3514–3516 CrossRef CAS.
- N. Kushwah, D. Nechiyil, J. Prakash, S. M. Chopade, S. S. Pathak, A. Wadawale, S. Kumar and G. Kedarnath, CCDC 2388509: Experimental Crystal Structure Determination, 2025, DOI:10.5517/ccdc.csd.cc2l5fpx.
| This journal is © The Royal Society of Chemistry 2025 |