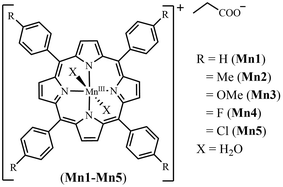
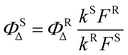Mn(III) porphyrins as photosensitizers: structural, photophysical and anticancer studies†
Bidisha
Bora
a,
Namisha
Das
a,
Jakia Parbin
Sultana
a,
Md Kausar
Raza
 *b and
Tridib K.
Goswami
*b and
Tridib K.
Goswami
 *a
*a
aDepartment of Chemistry, Gauhati University, Guwahati 781014, Assam, India. E-mail: tridibgoswami05@gmail.com
bDepartment of Inorganic and Physical Chemistry, Indian Institute of Science, Bangalore 560012, India. E-mail: kausarraza91@gmail.com
First published on 10th July 2025
Abstract
Herein, we synthesized, characterized and explored the photo-triggered anticancer activity of five Mn(III) porphyrins Mn1–Mn5, viz. (diaqua)meso-(tetraphenylporphyrinato)manganese(III) propionate, [Mn(III)TPP(H2O)2]+(C3H5O2−) or Mn1; (diaqua)meso-tetrakis(4-methylphenylporphyrinato)manganese(III) propionate, [Mn(III)TMeP(H2O)2]+(C3H5O2−) or Mn2; (diaqua)meso-tetrakis(4-methoxyphenylporphyrinato)manganese(III) propionate, [Mn(III)TMP(H2O)2]+(C3H5O2−) or Mn3; (diaqua)meso-tetrakis(4-fluorophenylporphyrinato)manganese(III) propionate, [Mn(III)FTPP(H2O)2]+(C3H5O2−) or Mn4 and (diaqua)meso-tetrakis(4-chlorophenylporphyrinato)manganese(III) propionate, [Mn(III)ClTPP(H2O)2]+(C3H5O2−) or Mn5, which remain virtually unexplored as photodynamic therapy (PDT) agents like other paramagnetic metalloporphyrins. These Mn(III) porphyrins, bearing different para-substituents on their meso-phenyl rings and two water molecules as axial ligands, were characterized using spectroscopic techniques and structurally through single-crystal X-ray diffraction, revealing an octahedral MnN4O2 geometry. Binding studies demonstrated a strong affinity of the metalloporphyrins for human serum albumin (HSA), indicating their potential for biological applications. The visible light-assisted generation of reactive oxygen species (ROS) by these Mn(III) porphyrins was confirmed via 1,3-diphenylisobenzofuran (DPBF) titration, identifying singlet oxygen (1O2) as one of the primary ROS. Photoredox activity under visible light, displayed by the Mn(III) porphyrins in the presence of ascorbic acid involving +3 and +2 oxidation states of manganese, further underscores the photochemotherapeutic potential of Mn1–Mn5. The ROS generation ability was further validated intracellularly in HeLa cells using Mn4 with the help of 2′,7′-dichlorofluorescein diacetate (DCFDA) assay under visible light irradiation (λ = 400–700 nm). Furthermore, among the five Mn(III) porphyrin complexes (Mn1–Mn5) evaluated for photo-triggered anticancer activity using MTT assays, Mn4 exhibited superior photocytotoxicity, with a half-maximal inhibitory concentration (IC50) of 4.93 ± 0.7 μM against HeLa cancer cells under visible light irradiation and negligible dark toxicity (IC50 > 50 μM). These results suggest that both type-I and type-II ROS generation pathways contribute to the observed photocytotoxicity. This study highlights the potential of paramagnetic metalloporphyrins, particularly Mn(III) porphyrins, in anticancer application by demonstrating their effectiveness as photosensitizers for photodynamic cancer therapy.
Introduction
Porphyrins belong to the family of tetrapyrrolic macrocycles that have gained massive research interest due to their many biological relevance and potential applications in diverse fields such as catalysis, sensing, and medicinal chemistry. Their unique optical, electronic, redox and catalytic properties make them ligands par excellence in both biology and chemistry.1–4 The use of porphyrins as photosensitizers (PSs) in photodynamic therapy (PDT) has grown considerably over the past few decades as a promising approach to combat cancer. PDT is a recently developed technique that relies on the ability of the PSs to generate cell-damaging reactive oxygen species (ROS) in the presence of light of an appropriate wavelength and molecular oxygen, thereby eliminating malignant cells.5–7 This therapy offers several advantages over traditional cancer treatments due to its non-invasiveness, spatiotemporal controllability and minimal systemic toxicity.8–10 Porphyrinic PSs are reported to preferentially accumulate in tumour cells over normal cells and exhibit photocytotoxicity via the type-II PDT pathway, i.e. through the production of singlet oxygen (1O2).8,9,11–13 However, some literature reports suggest that paramagnetic metalloporphyrins are not considered good photosensitizers as they negatively affect the 1O2 generation process.14,15 Nonetheless, in 2014, Antoni et al. reported the photocytotoxicity of a paramagnetic Cu(II) porphyrin in A2780 cancer cell lines, which was attributed to its ability to generate cytotoxic hydroxyl radicals (˙OH) upon exposure to red light.16 This study prompted us to explore the photocytotoxicity of other paramagnetic metalloporphyrins and their mechanism of action.Manganese plays an enormous roles in various biological activities, such as crucial roles in different metabolic functions, including cofactor for different type of enzymes, which calls for it use in pharmaceuticals.17 Mn-porphyrins bearing high positive charges were initially developed as SOD mimics and redox-active antioxidants.18 Their anticancer activity is thought to result from redox-induced cytotoxicity, which can affect the redox balance of cancer cells.18–20 However, there are only a few reports on the photoinduced anticancer activity of Mn-porphyrins.21–23 In 2003, Banfi et al. reported that positively charged Mn(III) porphyrins could oxidatively damage Plasmid Bluescript DNA in the presence of either an artificial oxygen donor or light,21 demonstrating the potential of Mn(III) porphyrins in PDT. In 2017, Lei Shi and co-workers demonstrated considerable photocytotoxicity of an anionic Mn(III) porphyrin in HepG2 cells.22 More recently, Nemeth and colleagues reported the PDT activity of a cationic Mn(III) pyridyl porphyrin in HeLa cells, and their EPR studies indicated that 1O2 was the primary cytotoxic species.23 In another study, Kim et al. showed that Mn(III) porphyrins undergo photoreduction in the presence of polynucleotides and can oxidize DNA via a photo-redox pathway.24
With these facts in mind, in this work, we synthesized five Mn(III) porphyrins bearing simple substituents at the para position of the meso-phenyl rings and two axial aqua ligands, viz. (diaqua)meso-(tetraphenylporphyrinato)manganese(III) propionate, [Mn(III)TPP(H2O)2]+(C3H5O2−) or Mn1; (diaqua)meso-tetrakis(4-methylphenyl porphyrinato)manganese(III) propionate, [Mn(III)TMeP(H2O)2]+(C3H5O2−) or Mn2; (diaqua)meso-tetrakis(4-methoxyphenylporphyrinato)manganese(III) propionate, [Mn(III)TMP(H2O)2]+(C3H5O2−) or Mn3; (diaqua)meso-tetrakis(4-fluorophenyl porphyrinato)manganese(III) propionate, [Mn(III)FTPP(H2O)2]+(C3H5O2−) or Mn4; and (diaqua)meso-tetrakis(4-chlorophenylporphyrinato)manganese(III) propionate, [Mn(III)ClTPP(H2O)2]+(C3H5O2−) or Mn5, in order to examine their photoinduced anticancer activity (Chart 1). The compounds were fully characterized by various spectroscopic and analytical techniques and structurally characterized by single crystal X-ray diffraction. They showed significant binding affinity towards human serum albumin (HSA) protein. UV-visible titration experiments with DPBF confirmed the generation of 1O2 by the compounds. The Mn(III) porphyrins also displayed redox shuttling between Mn(III) and Mn(II) in air in the presence of ascorbic acid and visible light, which is believed to induce additional oxidative stress in cells. Furthermore, 2′,7′-dichlorofluorescein diacetate (DCFDA) assay demonstrated the generation of ROS by the metalloporphyrins in the cellular environment. MTT assays showed significant photocytotoxicity of the Mn(III) porphyrins in HeLa cells under visible light.
Experimental section
Materials and measurements
Chemicals and solvents used in the work were obtained from commercial sources such as SD Fine Chemicals, India; Spectrochem, India; Loba Chemie, India; and Sigma-Aldrich, USA, and were used as received unless otherwise specified. Freshly purified pyrrole, prepared by passing it through a short column of basic alumina, was used for the synthesis of the free bases. Human serum albumin (HSA), 1,3-diphenylisobenzofuran (DPBF), L-ascorbic acid, Dulbecco's modified Eagle's medium (DMEM), MTT (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide), 2′,7′-dichlorofluorescein diacetate (DCFDA), Dulbecco's phosphate-buffered saline (DPBS) and fetal bovine serum (FBS) were all purchased from Sigma-Aldrich (USA). Tris-(hydroxylmethyl)aminomethane-HCl (Tris-HCl) buffer solution was prepared by using deionised and sonicated double-distilled water.Elemental CHN analyses were performed using a Thermo Finnigan Flash EA 1112 CHN analyzer. FT-IR spectra were recorded on a Bruker Alpha-II spectrophotometer, with samples prepared as KBr pellets. Electronic absorption and emission spectra were recorded on Shimadzu UV-1800 and Hitachi F-7000 spectrophotometers, respectively. 1H NMR spectra were recorded at room temperature on Bruker 300 MHz and 400 MHz NMR spectrometers. Room-temperature magnetic susceptibility measurements of the Mn(III) porphyrins were obtained using a Sherwood Scientific instrument (Cambridge, England), with Hg[Co(NCS)4] as the standard. The experimental susceptibility data were corrected for diamagnetic contributions.25 Electrochemical measurements were carried out at 25 °C using a CHI660D electrochemical workstation (CH Instruments, Inc., USA) with a conventional three-electrode configuration consisting of a glassy carbon working electrode, platinum wire auxiliary electrode and an Ag/AgCl reference electrode. Tetrabutylammonium perchlorate (TBAP, 0.1 M) in DMF was used as the supporting electrolyte. High-resolution electrospray ionization mass spectral measurements were recorded using a Waters Xevo G2-XS QTof mass spectrometer. Photoirradiation for cellular experiments was performed using a Luzchem Photoreactor (model LZC-1, Ontario, Canada) equipped with eight Sylvania white fluorescent tubes (λ = 400–700 nm). A TECAN microplate reader provided the formazan absorbance values in the MTT assay, and the resulting data were analyzed using GraphPad Prism 8 software. A fluorescence-activated cell sorting (FACS) Verse device (BD Biosciences) was used to conduct the flow cytometric analysis. The instrument is equipped with a MoFLo XDP cell sorter and analyzer featuring 10-color detection capability, and three lasers (λ = 488, 365, and 640 nm).
Synthesis
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) petroleum ether (1
petroleum ether (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1)] to obtain the pure ligands as purple crystalline solids [yield: 1.125 g, 19.23% for L1; 1.310 g, 19.87% for L2; 1.287 g, 19.8% for L3; 0.980 g, 14.30% for L4; 1.240 g, 16.45% for L5] (Scheme S1, ESI†).
1)] to obtain the pure ligands as purple crystalline solids [yield: 1.125 g, 19.23% for L1; 1.310 g, 19.87% for L2; 1.287 g, 19.8% for L3; 0.980 g, 14.30% for L4; 1.240 g, 16.45% for L5] (Scheme S1, ESI†).
Anal. calcd for C44H30N4 (L1): C, 85.97; H, 4.92; N, 9.11. Found: C, 85.71; H, 4.87; N, 9.23. Selected IR data (KBr, cm−1): 3314w (N–H), 3052w and 3021w (C–H), 2920w, 1678s, 1592m (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C), 1553m, 1470s (C
C), 1553m, 1470s (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C), 1437s, 1402m, 1347s (C–N), 1219w, 1174s, 1071m, 963vs, 799vs (C–Hbending), 700vs (vs, very strong; s, strong; m, medium; w, weak; br, broad). UV-Visible in DMSO, [λmax/nm (ε/M−1 cm−1)]: 416 (S, 1.87 × 105), 513 (Q, 8.42 × 103), 548 (Q, 3.8 × 103), 590 (Q, 2.60 × 103), 647 (Q, 2.56 × 103). 1H NMR (300 MHz, CDCl3): δ ppm, 8.88 (s, 8H, β PyH), 8.25–8.23 (d, J = 6.0 Hz, 8H, Ar o-H), 7.79–7.77 (d, J = 6.0 Hz, 12H, Ar m & p H), −2.76 (s, 2H, NH). ESI-MS in MeOH (m/z): 615.2551 for [M + H]+ (calcd: 615.2549).
C), 1437s, 1402m, 1347s (C–N), 1219w, 1174s, 1071m, 963vs, 799vs (C–Hbending), 700vs (vs, very strong; s, strong; m, medium; w, weak; br, broad). UV-Visible in DMSO, [λmax/nm (ε/M−1 cm−1)]: 416 (S, 1.87 × 105), 513 (Q, 8.42 × 103), 548 (Q, 3.8 × 103), 590 (Q, 2.60 × 103), 647 (Q, 2.56 × 103). 1H NMR (300 MHz, CDCl3): δ ppm, 8.88 (s, 8H, β PyH), 8.25–8.23 (d, J = 6.0 Hz, 8H, Ar o-H), 7.79–7.77 (d, J = 6.0 Hz, 12H, Ar m & p H), −2.76 (s, 2H, NH). ESI-MS in MeOH (m/z): 615.2551 for [M + H]+ (calcd: 615.2549).
Anal. calcd for C48H38N4 (L2): C, 85.94; H, 5.71; N, 8.35. Found: C, 85.73; H, 5.77; N, 8.27. Selected IR data (KBr, cm−1): 3326w (N–H), 3021w (C–H), 2902w, 1699w, 1557m (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C), 1509m, 1468s (C
C), 1509m, 1468s (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C), 1347s (C–N), 1217w, 1180s, 1108w, 1021w, 966vs, 846m, 799vs (C–Hbending), 733s, 615m. UV-Visible in DMSO, [λmax/nm (ε/M−1 cm−1)]: 418 (S, 2.8 × 105), 515 (Q, 1.2 × 104), 550 (Q, 6.2 × 103), 593 (Q, 3.5 × 103), 649 (Q, 3.9 × 103). 1H NMR (400 MHz, CDCl3): δ ppm, 8.86 (s, 8H, β PyH), 8.11–8.09 (d, J = 8.0 Hz, 8H, Ar o-H), 7.57–7.55 (d, J = 8.0 Hz, 8H, Ar m-H), 2.71 (s, 12H, CH3), −2.78 (s, 2H, NH). ESI-MS in MeOH (m/z): 671.3177 for [M + H]+ (calcd: 671.3175).
C), 1347s (C–N), 1217w, 1180s, 1108w, 1021w, 966vs, 846m, 799vs (C–Hbending), 733s, 615m. UV-Visible in DMSO, [λmax/nm (ε/M−1 cm−1)]: 418 (S, 2.8 × 105), 515 (Q, 1.2 × 104), 550 (Q, 6.2 × 103), 593 (Q, 3.5 × 103), 649 (Q, 3.9 × 103). 1H NMR (400 MHz, CDCl3): δ ppm, 8.86 (s, 8H, β PyH), 8.11–8.09 (d, J = 8.0 Hz, 8H, Ar o-H), 7.57–7.55 (d, J = 8.0 Hz, 8H, Ar m-H), 2.71 (s, 12H, CH3), −2.78 (s, 2H, NH). ESI-MS in MeOH (m/z): 671.3177 for [M + H]+ (calcd: 671.3175).
Anal. calcd for C48H38N4O4 (L3): C, 78.45; H, 5.21; N, 7.62. Found: C, 78.21; H, 5.29; N, 7.55. Selected IR data (KBr, cm−1): 3314w (N–H), 2928w (C–H), 2832w (C–H of –OCH3), 1744m, 1604s (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C), 1505s, 1464s (C
C), 1505s, 1464s (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C), 1347m (C–N), 1289s, 1248vs (C–O–C), 1174vs, 1108w, 1033s, 966s, 803s (C–Hbending), 737s, 640m. UV-Visible in DMSO, [λmax/nm (ε/M−1 cm−1)]: 421 (S, 1.77 × 105), 518 (Q, 7.06 × 103), 555 (Q, 5.48 × 103), 595 (Q, 2.40 × 103), 651 (Q, 3.9 × 103). 1H NMR (300 MHz, CDCl3): δ ppm, 8.86 (s, 8H, β PyH), 8.14–8.11 (d, J = 9.0 Hz, 8H, Ar o-H), 7.30–7.27 (d, J = 9.0 Hz, 8H, Ar m-H), 4.10 (s, 12H, OCH3), −2.78 (s, 2H, NH). ESI-MS in MeOH (m/z): 735.2908 for [M + H]+ (calcd: 735.2971).
C), 1347m (C–N), 1289s, 1248vs (C–O–C), 1174vs, 1108w, 1033s, 966s, 803s (C–Hbending), 737s, 640m. UV-Visible in DMSO, [λmax/nm (ε/M−1 cm−1)]: 421 (S, 1.77 × 105), 518 (Q, 7.06 × 103), 555 (Q, 5.48 × 103), 595 (Q, 2.40 × 103), 651 (Q, 3.9 × 103). 1H NMR (300 MHz, CDCl3): δ ppm, 8.86 (s, 8H, β PyH), 8.14–8.11 (d, J = 9.0 Hz, 8H, Ar o-H), 7.30–7.27 (d, J = 9.0 Hz, 8H, Ar m-H), 4.10 (s, 12H, OCH3), −2.78 (s, 2H, NH). ESI-MS in MeOH (m/z): 735.2908 for [M + H]+ (calcd: 735.2971).
Anal. calcd for C44H26N4F4 (L4): C, 76.96; H, 3.82; N, 8.16. Found: C, 76.84; H, 3.79; N, 8.22. Selected IR data (KBr, cm−1): 3309m (N–H), 3042w (C–H), 1699w, 1600m (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C), 1555w, 1503vs, 1470s (C
C), 1555w, 1503vs, 1470s (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C), 1400w, 1351m (C–N), 1225vs, 1157vs (Ar–F), 1091w, 968s, 846m, 807vs (C–Hbending), 729s. UV-Visible in DMSO, [λmax/nm (ε/M−1 cm−1)]: 414 (S, 4.2 × 104), 511 (Q, 2.7 × 103), 545 (Q, 1.2 × 103), 586 (Q, 7.8 × 102), 645 (Q, 5.7 × 102). 1H NMR (300 MHz, DMSO-d6 with NaOH): δ ppm, 8.53 (s, 8H, β PyH), 8.14–8.12 (d, J = 6.0 Hz, 8H, Ar o-H), 7.57–7.51 (t, J = 9 Hz, 8H, Ar m-H). ESI-MS in MeOH (m/z): 687.4095 for [M + H]+ (calcd: 687.2172).
C), 1400w, 1351m (C–N), 1225vs, 1157vs (Ar–F), 1091w, 968s, 846m, 807vs (C–Hbending), 729s. UV-Visible in DMSO, [λmax/nm (ε/M−1 cm−1)]: 414 (S, 4.2 × 104), 511 (Q, 2.7 × 103), 545 (Q, 1.2 × 103), 586 (Q, 7.8 × 102), 645 (Q, 5.7 × 102). 1H NMR (300 MHz, DMSO-d6 with NaOH): δ ppm, 8.53 (s, 8H, β PyH), 8.14–8.12 (d, J = 6.0 Hz, 8H, Ar o-H), 7.57–7.51 (t, J = 9 Hz, 8H, Ar m-H). ESI-MS in MeOH (m/z): 687.4095 for [M + H]+ (calcd: 687.2172).
Anal. calcd for C44H26Cl4N4 (L5): C, 70.23; H, 3.48; N, 7.45. Found: C, 70.11; H, 3.42; N, 7.49. Selected IR data (KBr, cm−1): 3312w (N–H), 2920m (C–H), 2850w, 2310w, 1656w, 1553m (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C), 1470s (C
C), 1470s (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C), 1394m, 1344m (C–N), 1174m, 1089vs (Ar–Cl), 1015s, 966s, 844m, 795vs (C–Hbending), 729s. UV-Visible in DMSO, [λmax/nm (ε/M−1 cm−1)]: 417 (S, 1.87 × 105), 514 (Q, 8.72 × 103), 549 (Q, 3.67 × 103), 590 (Q, 2.58 × 103), 647 (Q, 2.14 × 103). 1H NMR (300 MHz, CDCl3): δ 8.84 (s, 8H, β PyH), 8.15–8.12 (d, J = 9.0 Hz, 8H, Ar o-H), 7.76–7.74 (d J = 6.0 Hz, 8H, Ar m-H), −2.88 (s, 2H, NH). ESI-MS in MeOH (m/z): 753.0969 for [M + H]+ (calcd: 753.0960).
C), 1394m, 1344m (C–N), 1174m, 1089vs (Ar–Cl), 1015s, 966s, 844m, 795vs (C–Hbending), 729s. UV-Visible in DMSO, [λmax/nm (ε/M−1 cm−1)]: 417 (S, 1.87 × 105), 514 (Q, 8.72 × 103), 549 (Q, 3.67 × 103), 590 (Q, 2.58 × 103), 647 (Q, 2.14 × 103). 1H NMR (300 MHz, CDCl3): δ 8.84 (s, 8H, β PyH), 8.15–8.12 (d, J = 9.0 Hz, 8H, Ar o-H), 7.76–7.74 (d J = 6.0 Hz, 8H, Ar m-H), −2.88 (s, 2H, NH). ESI-MS in MeOH (m/z): 753.0969 for [M + H]+ (calcd: 753.0960).
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1). This was followed by the addition of 1.25 mmol (306 mg) Mn(OAc)2·4H2O and the reaction mixture was refluxed for 1 h.27 The organic layer was separated using a separating funnel, collected in chloroform, and evaporated to dryness in a rotary evaporator. The resulting solid was loaded onto a silica gel column and eluted with 1% ethanol in chloroform to remove any unreacted free base. The pure product, isolated as a greenish-black solid, was dried in vacuo over anhydrous calcium chloride, and the yield was calculated [yield: 0.162 g, 83.93% for Mn1; 0.170 g, 82.12% for Mn2; 0.178 g, 79.82% for Mn3, 0.142 g, 67.07% for Mn4; 0.197 g, 86.40% for Mn5] (Scheme S1, ESI†).
1). This was followed by the addition of 1.25 mmol (306 mg) Mn(OAc)2·4H2O and the reaction mixture was refluxed for 1 h.27 The organic layer was separated using a separating funnel, collected in chloroform, and evaporated to dryness in a rotary evaporator. The resulting solid was loaded onto a silica gel column and eluted with 1% ethanol in chloroform to remove any unreacted free base. The pure product, isolated as a greenish-black solid, was dried in vacuo over anhydrous calcium chloride, and the yield was calculated [yield: 0.162 g, 83.93% for Mn1; 0.170 g, 82.12% for Mn2; 0.178 g, 79.82% for Mn3, 0.142 g, 67.07% for Mn4; 0.197 g, 86.40% for Mn5] (Scheme S1, ESI†).
Anal. calcd for C47H37N4O4Mn (Mn1): C, 72.67; H, 4.80; N, 7.21. Found: C, 72.49; H, 4.88; N, 7.13. Selected IR data (KBr, cm−1): 3442br (O–H), 3052w & 2924w (C–H), 1631w, 1598m (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C), 1439w (C
C), 1439w (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C), 1384vs, 1350m (C–N), 1176w, 1072m, 1005vs, 834w, 793s (C–Hbending), 742s, 697s, 526w. UV-Visible in DMSO, [λmax/nm (ε/M−1 cm−1)]: 466 (S, 1.47 × 105), 568 (Q, 1.44 × 104), 603 (Q, 1.10 × 104). ESI-MS in MeCN (m/z): 667.1685 for [M-(2H2O + C3H5O2−)]+ (calcd: 667.1694). μeff = 4.5μB at 298 K.
C), 1384vs, 1350m (C–N), 1176w, 1072m, 1005vs, 834w, 793s (C–Hbending), 742s, 697s, 526w. UV-Visible in DMSO, [λmax/nm (ε/M−1 cm−1)]: 466 (S, 1.47 × 105), 568 (Q, 1.44 × 104), 603 (Q, 1.10 × 104). ESI-MS in MeCN (m/z): 667.1685 for [M-(2H2O + C3H5O2−)]+ (calcd: 667.1694). μeff = 4.5μB at 298 K.
Anal. calcd for C51H45N4O4Mn (Mn2): C, 73.55; H, 5.45; N, 6.73. Found: C, 73.32; H, 5.38; N, 6.79. Selected IR data (KBr, cm−1): 3438br (O–H), 2930w (C–H), 1631w, 1564m (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C), 1413w (C
C), 1413w (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C), 1384m, 1352m (C–N), 1209w, 1180w, 1107w, 1074w, 1001s, 799s (C–Hbending), 717m, 603w, 524w. UV-Visible in DMSO [λmax/nm (ε/M−1 cm−1)]: 465 (S, 1.29 × 105), 569 (Q, 1.42 × 104), 607 (Q, 1.36 × 104). ESI-MS in MeCN (m/z): 723.2330 for [M-(2H2O + C3H5O2−)]+ (calcd: 723.2320). μeff = 4.7μB at 298 K.
C), 1384m, 1352m (C–N), 1209w, 1180w, 1107w, 1074w, 1001s, 799s (C–Hbending), 717m, 603w, 524w. UV-Visible in DMSO [λmax/nm (ε/M−1 cm−1)]: 465 (S, 1.29 × 105), 569 (Q, 1.42 × 104), 607 (Q, 1.36 × 104). ESI-MS in MeCN (m/z): 723.2330 for [M-(2H2O + C3H5O2−)]+ (calcd: 723.2320). μeff = 4.7μB at 298 K.
Anal. calcd for C51H45N4O8Mn (Mn3): C, 68.30; H, 5.06; N, 6.25. Found: C, 68.11; H, 5.12; N, 6.17. Selected IR data (KBr, cm−1): 3444br (O–H), 2924w (C–H), 2832w (C–H of –OCH3), 1606s (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C), 1507s, 1460m (C
C), 1507s, 1460m (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C), 1384w, 1352s (C
C), 1384w, 1352s (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C), 1286m, 1248vs (C–O–C), 1174s, 1105w, 1074w, 1027w, 1001s, 803s (C–Hbending), 717m, 607m, 540w. UV-Visible in DMSO [λmax/nm (ε/M−1 cm−1)]: 467 (S, 1.38 × 105), 572 (Q, 1.46 × 104), 609 (Q, 1.66 × 104). ESI-MS in MeCN (m/z): 787.2111 for [M-(2H2O + C3H5O2−)]+ (calcd: 787.2117). μeff = 4.9μB at 298 K.
C), 1286m, 1248vs (C–O–C), 1174s, 1105w, 1074w, 1027w, 1001s, 803s (C–Hbending), 717m, 607m, 540w. UV-Visible in DMSO [λmax/nm (ε/M−1 cm−1)]: 467 (S, 1.38 × 105), 572 (Q, 1.46 × 104), 609 (Q, 1.66 × 104). ESI-MS in MeCN (m/z): 787.2111 for [M-(2H2O + C3H5O2−)]+ (calcd: 787.2117). μeff = 4.9μB at 298 K.
Anal. calcd for C47H33N4O4F4Mn (Mn4): C, 66.51; H, 3.92; N, 6.60. Found: C, 66.39; H, 3.84; N, 6.69. Selected IR data (KBr, cm−1): 3442br (O–H), 2975w & 2922w (C–H), 1602m (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C), 1564s, 1498s (C
C), 1564s, 1498s (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C), 1415m, 1341m (C–N), 1296w, 1225s, 1158s (Ar–F), 1072m, 1011s, 854w, 803s (C–Hbending), 719w, 597m, 532m. UV-Visible in DMSO [λmax/nm (ε/M−1 cm−1)]: 465 (S, 1.22 × 105), 567 (Q, 1.24 × 104), 603 (Q, 8.64 × 103). ESI-MS in MeCN (m/z): 739.1324 for [M-(2H2O + C3H5O2−)]+ (calcd: 739.1318). μeff = 4.4μB at 298 K.
C), 1415m, 1341m (C–N), 1296w, 1225s, 1158s (Ar–F), 1072m, 1011s, 854w, 803s (C–Hbending), 719w, 597m, 532m. UV-Visible in DMSO [λmax/nm (ε/M−1 cm−1)]: 465 (S, 1.22 × 105), 567 (Q, 1.24 × 104), 603 (Q, 8.64 × 103). ESI-MS in MeCN (m/z): 739.1324 for [M-(2H2O + C3H5O2−)]+ (calcd: 739.1318). μeff = 4.4μB at 298 K.
Anal. calcd for C47H33N4O4Cl4Mn (Mn5): C, 61.73; H, 3.64; N, 6.13. Found: C, 61.73; H, 3.61; N, 6.21. Selected IR data (KBr phase, cm−1): 3421br (O–H), 2977w & 2938w (C–H), 1715m, 1641w, 1566m (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C), 1484s (C
C), 1484s (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C), 1394m, 1339w (C–N), 1282w, 1205m, 1091vs (Ar–Cl), 1009vs, 883w, 850w, 803vs (C–Hbending), 717m, 632w, 562w, 501m. UV-Visible in DMSO [λmax/nm (ε/M−1 cm−1)]: 463 (S, 1.51 × 105), 567 (Q, 1.68 × 104), 605 (Q, 1.34 × 104). ESI-MS in MeCN (m/z): 805.0097 for [M-(2H2O + C3H5O2−)]+ (calcd: 805.0106). μeff = 4.6μB at 298 K.
C), 1394m, 1339w (C–N), 1282w, 1205m, 1091vs (Ar–Cl), 1009vs, 883w, 850w, 803vs (C–Hbending), 717m, 632w, 562w, 501m. UV-Visible in DMSO [λmax/nm (ε/M−1 cm−1)]: 463 (S, 1.51 × 105), 567 (Q, 1.68 × 104), 605 (Q, 1.34 × 104). ESI-MS in MeCN (m/z): 805.0097 for [M-(2H2O + C3H5O2−)]+ (calcd: 805.0106). μeff = 4.6μB at 298 K.
X-ray crystallographic procedure
The structures of the Mn(III) porphyrins Mn1, Mn2, Mn4 and Mn5 were determined by single-crystal X-ray diffraction. Diffraction-quality crystals were isolated by slow evaporation of aqueous propionic acid solutions of the metalloporphyrins over 2–3 days. Crystals were mounted on glass fibres using epoxy cement. All geometric and intensity data were collected at room temperature on a Bruker SMART APEX II CCD diffractometer equipped with a Mo-Kα fine-focus sealed-tube X-ray source (λ = 0.71073 Å), using increasing ω (width of 0.3° per frame) at a scan speed of 5 s per frame. Intensity data, collected in ω–2θ scan mode, were corrected for Lorentz-polarisation effects and for absorption using SADABS.28 Structure solution and refinement were performed using a combination of Patterson and Fourier methods, followed by full-matrix least-squares refinement using Bruker SHELXTL suite.29 For Mn5, data were collected using a dual-source (Cu/Mo) Rigaku Saturn 724+ CCD and a Synergy-S kappa diffractometer at room temperature. All hydrogen atoms belonging to the complex were refined isotropically, whereas all the non-hydrogen atoms were refined anisotropically. Perspective molecular views were generated using ORTEP.30HSA binding experiment
The binding affinity of the compounds Mn1–Mn5 for human serum albumin (HSA) were determined by tryptophan fluorescence quenching experiments using a solution of HSA (2 μM) in Tris-HCl buffer (pH 7.2). Quenching of the emission intensity of the single tryptophan residue (Trp-214) of HSA at 340 nm (excitation wavelength: 280 nm) by increasing concentrations of Mn1–Mn5 as quenchers was monitored using a fluorescence spectrophotometer. Plots of I0/I vs. [compound] were constructed, and the data were linearly fitted using the Stern–Volmer-like equation I0/I = 1 + K[Q], where I0 and I represent the emission intensities of HSA in the absence and presence of the quencher, respectively, and [Q] denotes the quencher concentration. The resulting slopes provided the binding constant KHSA values.31Partition coefficient of the metalloporphyrins between n-octanol and water
The partition coefficients of the Mn(III) porphyrins Mn1–Mn5 between n-octanol and water were determined using a modified shake-flask method reported in the literature.32 In summary, 5 ml solution of the compound in n-octanol-saturated water (OSW) was vortexed with 5 ml of water-saturated n-octanol (WSO) in a Falcon tube for ∼15–20 min. The biphasic mixture was then centrifuged at ∼3000 rpm for ∼5 min to separate the layers. The concentrations of Mn1–Mn5 in each layer were quantified by UV-visible spectroscopy. The partition coefficients of the compounds were determined by repeating the experiment three times at varying concentration of Mn1–Mn5 and averaging the results using the formula log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) P = log{[Mn1–Mn5]oct/[Mn1–Mn5]aq}.
P = log{[Mn1–Mn5]oct/[Mn1–Mn5]aq}.
UV-visible titration using DPBF to ascertain generation of 1O2
1,3-Diphenylisobenzofuran (DPBF) was used as a chemical probe to detect the generation of any singlet oxygen (1O2) by the Mn(III) porphyrins (Mn1–Mn5) under visible light irradiation. DPBF reacts specifically with 1O2 to form an endoperoxide, and a decrease in the absorbance intensity of DPBF at ∼415 nm indicates its conversion.33 The experiment was conducted in two different solvents, i.e. DMF and DMSO. A 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 molar ratio of DPBF to compound was prepared in the respective solvent, and the absorption maxima of DPBF were monitored before and after irradiation with visible light (irradiation provided by a 12 W LED bulb fitted inside a rectangular box of area 9 cm × 4.5 cm, λ = 400–700 nm) at various time intervals. The absorbance data recorded at different time interval of light exposure were fitted in a pseudo first-order rate equation: ln([A]t/[A]0) = −kt, where [A]0 and [A]t represent the absorbance of DPBF before and after light irradiation for a duration of “t”, and k is the DPBF decay constant, indicating the rate of 1O2 generation. The value of k was obtained from the slope of the plot.34 Various control experiments were also performed to confirm the formation of 1O2, including (a) photoirradiation of untreated DPBF for similar time intervals, (b) photoirradiation of compound-treated DPBF in the presence of a 1O2 quencher (NaN3) and (c) compound-treated DPBF kept in dark. Relative quantum yields of the compounds for 1O2 generation were calculated from the k values using TPP as the reference standard (ΦRΔ = 0.64 in DMF and 0.52 in DMSO), based on the following equation:35,36
1 molar ratio of DPBF to compound was prepared in the respective solvent, and the absorption maxima of DPBF were monitored before and after irradiation with visible light (irradiation provided by a 12 W LED bulb fitted inside a rectangular box of area 9 cm × 4.5 cm, λ = 400–700 nm) at various time intervals. The absorbance data recorded at different time interval of light exposure were fitted in a pseudo first-order rate equation: ln([A]t/[A]0) = −kt, where [A]0 and [A]t represent the absorbance of DPBF before and after light irradiation for a duration of “t”, and k is the DPBF decay constant, indicating the rate of 1O2 generation. The value of k was obtained from the slope of the plot.34 Various control experiments were also performed to confirm the formation of 1O2, including (a) photoirradiation of untreated DPBF for similar time intervals, (b) photoirradiation of compound-treated DPBF in the presence of a 1O2 quencher (NaN3) and (c) compound-treated DPBF kept in dark. Relative quantum yields of the compounds for 1O2 generation were calculated from the k values using TPP as the reference standard (ΦRΔ = 0.64 in DMF and 0.52 in DMSO), based on the following equation:35,36Here, the superscripts S and R denote sample and reference, respectively, and F is the absorption correction factor given by 1–10−OD, where OD is the absorbance of the compounds.
Photoreduction study in the presence of L-ascorbic acid
Ability of the Mn(III) porphyrins (Mn1–Mn5) to undergo photoreduction in the presence of a reducing agent such as L-ascorbic acid was monitored by UV-visible spectroscopy.37 A 10 μM solution of each Mn(III) porphyrin was treated with ascorbic acid (1.4 mM in case of Mn1, Mn4 and Mn5; 2 mM for Mn2 and Mn3) in 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 DMSO
1 DMSO![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Tris-HCl buffer medium. The varying amounts of ascorbic acid required for reduction likely reflect differences in the reduction potentials of the compounds. The resulting solutions were then exposed to visible light (λ = 400–700 nm) for 60 min in air, and UV-visible spectra were recorded at 15 min intervals during photoirradiation. A dark control experiment was also performed to validate the photoreduction behavior.
Tris-HCl buffer medium. The varying amounts of ascorbic acid required for reduction likely reflect differences in the reduction potentials of the compounds. The resulting solutions were then exposed to visible light (λ = 400–700 nm) for 60 min in air, and UV-visible spectra were recorded at 15 min intervals during photoirradiation. A dark control experiment was also performed to validate the photoreduction behavior.
DCFDA assay for detection of ROS
The DCFDA assay was used to detect the generation of cellular reactive oxygen species (ROS), using the most active Mn(III) porphyrin, i.e.Mn4, as a representative compound for the series. The experiment was conducted via flow cytometry following a reported protocol.38 Approximately 1 × 106 HeLa cells were incubated with Mn4 (10 μM) for 24 h. The cells were then treated with 10 μM DCFDA solution (in DMSO) in dark for 5 min at room temperature, followed by photoirradiation (400–700 nm) in PBS for varying durations (15, 30, 45 and 60 min). After irradiation, the cells were incubated for an additional 23 h in fresh medium. Corresponding dark control experiments were also conducted. Cells were harvested by trypsinization and resuspended as a single-cell suspension in PBS. Flow cytometry was used to assess the intracellular accumulation of H2DCF in HeLa cells via the FL-1 channel.Cell viability assay
The cytotoxicity of the compounds Mn1–Mn5 under visible light was evaluated in HeLa (human cervical carcinoma) cells using the MTT assay. This assay is based on the intracellular reduction of MTT dye by mitochondrial dehydrogenases in viable cells, resulting in the formation of dark blue, membrane-impermeable formazan crystals. The extent of absorbance at 540 nm provides a quantitative estimate of cell viability.39 HeLa cells were cultured in DMEM with 10% FBS, 100 IU ml−1 penicillin, 100 mg ml−1 streptomycin and 2 mM glutamax at 37 °C in a humidified incubator with 5% CO2. Monolayer adherent cultures were passaged every 4–5 days by trypsinization with 0.25% trypsin–EDTA. For the assay, ∼1 × 104 HeLa cells were seeded per well in a 96-well culture plate containing DMEM with 10% fetal bovine serum and incubated overnight. Different concentrations of Mn1–Mn5 were prepared by serial dilution in 1% DMSO–DMEM medium and added to the cells, followed by a 24 h incubation in the dark. Following incubation, the medium was replaced with 50 mM phosphate-buffered saline (PBS; pH 7.4, containing 150 mM NaCl), and one set of cells was photoirradiated for 1 h with visible light (λ = 400–700 nm) at a dose of 10 J cm−2. PBS was then replaced with 10% DMEM, and incubation was continued for an additional 23 h in the dark for light-exposed cells, and for 24 h in the dark for the unexposed cells. Subsequently, 25 μL of MTT solution (4 mg mL−1) was added to each well and incubated for an additional 3 h. After discarding the culture medium, formazan crystals were dissolved in DMSO, and absorbance was measured at 540 nm using a TECAN microplate reader (BioRad, Hercules, CA, USA). The cytotoxicity of the compounds was expressed in terms of their half-maximal inhibitory concentration (IC50) values, calculated using GraphPad Prism 8 software.Results and discussion
Synthesis and general aspects
Five Mn(III) meso-tetraaryalporphyrins (Mn1–Mn5) bearing different substituents at the para positions of the meso-aryl groups along with two water molecules as axial ligands were synthesized in good yields by reacting manganese(II) acetate tetrahydrate with the corresponding free bases (L1–L5) in a chloroform/propionic acid mixture. The formation of the free bases (L1–L5) was confirmed by 1H NMR and mass spectral data (Fig. S1–S10, ESI†). The corresponding metalloporphyrins (Mn1–Mn5) were characterised using various spectroscopic and analytical techniques, which supported the proposed structures. Selected physicochemical data are presented in Table 1. Characteristic N–H stretching vibrations at ∼3310–3325 cm−1 were observed in the IR spectra of the free bases (Fig. S11–S15, ESI†), which disappeared in case of their Mn(III) derivatives (Fig. S16–S20, ESI†). The absorption spectra of free-base porphyrins (L1–L5) in DMSO displayed a typical Soret (or B) band at ∼414–421 nm and four Q-bands spanning ∼511–651 nm. On metallation with manganese, the electronic spectra exhibited dramatic changes resembling those of hyperporphyrins.40 The Soret band was significantly red shifted (∼50 nm) to ∼460 nm, and the two Q-bands extended well into the red region, up to ∼605–610 nm (Fig. 1 and Fig. S21, ESI†). More specifically, the absorption intensities of the Q-bands in this region were markedly enhanced compared to their free-base counterparts, a feature highly desirable for PDT.9,14 These metalloporphyrins also exhibited additional absorption bands in the ∼370–400 nm range, which were attributed to LMCT transitions.40 Unlike the free-base porphyrins, the Mn(III) derivatives were found to be non-emissive in nature, as revealed by their emission spectra (Fig. 1 and Fig. S22, ESI†). The presence of a high-spin 3d4-Mn(III) center with four unpaired electrons was confirmed by the observed magnetic moment values of ∼4.5μB at 25 °C. The solution-phase stability of compounds Mn1–Mn5 was evidenced by their high-resolution ESI-MS spectra, which showed the base peak as [M-(2H2O + C3H5O2−)]+, corresponding to the loss of two weakly bound axial water molecules and a propionate counterion in acetonitrile (Fig. S23–S27, ESI†). Cyclic voltammetric measurements displayed a Mn(III)/Mn(II) redox couple at ∼−0.3 to −0.4 V vs. Ag/AgCl electrode, using TBAP (0.1 M) as the supporting electrolyte. All compounds exhibited quasi-reversible behavior with ΔEp > 60 mV for the Mn(III)/Mn(II) redox couple (Fig. 2 and Fig. S28, ESI†). The observed large ΔEp value is most likely associated with a change in the coordination number of the manganese center from six to five upon reduction of the Mn(III) porphyrins from d4 to d5.41 The negative potential required for one-electron reduction of Mn(III) porphyrins is consistent with the electrochemical stabilization of the reduced species. Mn4 and Mn5 were comparatively more readily reduced, likely due to the electron-withdrawing nature of the F and Cl atoms, respectively. | ||
| Fig. 1 (a) UV-visible absorption spectra of L4 and Mn4 (5 μM) (inset: enlarged view of the Q-bands) and (b) emission spectra of L4 and Mn4 (5 μM) in DMSO with λex set at their Soret band maxima. | ||
 | ||
| Fig. 2 Cyclic voltammetric responses of Mn4 (a) and Mn5 (b) in DMF using 0.1 M TBAP as the supporting electrolyte at a scan rate of 100 mV s−1vs. Ag/AgCl. | ||
| Compound |
λ
abs![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) a/nm (ε/103 M−1 cm−1) a/nm (ε/103 M−1 cm−1) |
E f/V (ΔEp/mV)b Mn(III)–Mn(II) |
μ
eff![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) c/μB c/μB |
log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Po/w Po/w |
Φ
Δ![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) d,e d,e |
|---|---|---|---|---|---|
| a 5 μM solution in DMSO. b Mn(III)–Mn(II) couple in DMF with 0.1 M TBAP, Ef = 0.5(Epa + Epc), ΔEp = (Epa − Epc), where Epa and Epc are the anodic and cathodic peak potentials, respectively. The potentials are referenced against the Ag/AgCl electrode. Scan rate = 100 mV s−1. c Magnetic moment measured at 298 K. d In DMF. e In DMSO. | |||||
| Mn1 | 466 (B, 147), 568 (Qy 14), 603 (Qx 11) | −0.373 (110) | 4.5 | 0.38 | 0.09, 0.04 |
| Mn2 | 465 (B, 129), 569 (Qy 14), 607 (Qx 13) | −0.400 (198) | 4.7 | 1.45 | 0.11, 0.06 |
| Mn3 | 467 (B, 138), 572 (Qy 14), 609 (Qx 16) | −0.402 (157) | 4.9 | 0.36 | 0.13, 0.08 |
| Mn4 | 465 (B, 122), 567 (Qy 12), 603 (Qx 8.6) | −0.351 (161) | 4.6 | 0.88 | 0.17, 0.09 |
| Mn5 | 463 (B, 151), 567 (Qy 16), 605 (Qx 13) | −0.336 (173) | 4.4 | 1.71 | 0.14, 0.08 |
Solubility, stability and aggregation properties
The Mn(III) porphyrins Mn1–Mn5 exhibited good solubility in MeOH, EtOH, MeCN, CH2Cl2, CHCl3, THF, DMF and DMSO, but were insoluble in hydrocarbon solvents. The compounds were found to be quite stable in DMSO–DMEM (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, v/v) solution phase for up to 24 h, as evidenced by their electronic absorption spectra in the ∼300–700 nm range (Fig. S29, ESI†). Photostability of the compounds was also evaluated in the same medium by irradiating the solutions with visible light for 60 min. The compounds remained stable under photoirradiation, suggesting their suitability for PDT without any significant degradation into undesirable oxidative products (Fig. S30, ESI†).42 Moreover, the Mn(III) porphyrins are expected to exhibit reduced aggregation in biological media due to the presence of two axial aqua ligands, which prevent individual porphyrin units from approaching each other closely.43 In the support of this, Mn1–Mn5 displayed sharp and intense absorption bands in DMSO–DMEM (1
1, v/v) solution phase for up to 24 h, as evidenced by their electronic absorption spectra in the ∼300–700 nm range (Fig. S29, ESI†). Photostability of the compounds was also evaluated in the same medium by irradiating the solutions with visible light for 60 min. The compounds remained stable under photoirradiation, suggesting their suitability for PDT without any significant degradation into undesirable oxidative products (Fig. S30, ESI†).42 Moreover, the Mn(III) porphyrins are expected to exhibit reduced aggregation in biological media due to the presence of two axial aqua ligands, which prevent individual porphyrin units from approaching each other closely.43 In the support of this, Mn1–Mn5 displayed sharp and intense absorption bands in DMSO–DMEM (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, v/v) media compared to the broad and less intense bands observed for the free-base porphyrins at similar concentrations, indicating significantly lower aggregation properties for the Mn(III) porphyrins (Fig. S31, ESI†). This finding suggests that Mn(III) porphyrins possess superior aqueous solubility compared to their free-base counterparts, thereby facilitating their evaluation for in vitro anticancer activities.
1, v/v) media compared to the broad and less intense bands observed for the free-base porphyrins at similar concentrations, indicating significantly lower aggregation properties for the Mn(III) porphyrins (Fig. S31, ESI†). This finding suggests that Mn(III) porphyrins possess superior aqueous solubility compared to their free-base counterparts, thereby facilitating their evaluation for in vitro anticancer activities.
Crystal structure
Structural characterization of the Mn(III) porphyrins Mn1, Mn2, Mn4 and Mn5 was successfully achieved by single-crystal X-ray diffraction analyses in the form of six coordinated cationic species [Mn(III)Por(H2O)2]+C3H5O2−. The crystal structure of Mn4 is entirely new, whereas those of other Mn(III) porphyrins, i.e.Mn1, Mn2 and Mn5, have been previously reported, albeit with different counterions crystallized from different solvents.44–49 To the best of our knowledge, the crystal structure of an Mn(III) porphyrin with propionate as the counterion has not been reported before. The ORTEP views of Mn4, Mn5 and the remaining compounds are shown in Fig. 3 (Fig. S32 and S33, ESI†). Mn4 crystallizes in the P![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) space group of the triclinic crystal system with 2 molecules per unit cell (Fig. S34, ESI†). Mn1 also crystallizes in the P
space group of the triclinic crystal system with 2 molecules per unit cell (Fig. S34, ESI†). Mn1 also crystallizes in the P![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) space group (triclinic) but with 1 molecule per unit cell (Fig. S35, ESI†). Mn2 and Mn5 crystallize in the P21/c space group of the monoclinic crystal system, with 2 molecules per unit cell (Fig. S36 and S37, ESI†). Selected crystallographic data are summarized in Table 2 (Table S1, ESI†). Selected bond distances and angle data are provided in Tables S2–S5 (ESI†). All the Mn(III) porphyrins exhibit an octahedral geometry around Mn(III) center, with four coordination sites occupied by the core nitrogen atoms of the flat planar porphyrin ring and two water molecules occupying the fifth and sixth coordination sites, i.e. the axial positions, with respect to the plan of the porphyrin ring. The Mn–N bond distances lie in the range of 2.0054–2.0207 Å. The Mn–O bond distances for the coordinated water molecules range from 2.2200–2.314 Å, indicating significant distortion in the octahedral structures imparted by tetragonal elongation anticipated in high-spin d4 Mn(III) complexes. The Mn–O and Mn–N bond distances are comparable to those reported structures that contain water, methanol, DMF, acrylamide etc. as axial ligands along with [Ag(CN)2]−, Br−, SbF6−, ClO4−etc. as counterions.44–49 The plane of the phenyl rings at the meso positions is not same with the plane of the porphyrin ring, suggesting a lack of conjugation between the two aromatic systems.
space group (triclinic) but with 1 molecule per unit cell (Fig. S35, ESI†). Mn2 and Mn5 crystallize in the P21/c space group of the monoclinic crystal system, with 2 molecules per unit cell (Fig. S36 and S37, ESI†). Selected crystallographic data are summarized in Table 2 (Table S1, ESI†). Selected bond distances and angle data are provided in Tables S2–S5 (ESI†). All the Mn(III) porphyrins exhibit an octahedral geometry around Mn(III) center, with four coordination sites occupied by the core nitrogen atoms of the flat planar porphyrin ring and two water molecules occupying the fifth and sixth coordination sites, i.e. the axial positions, with respect to the plan of the porphyrin ring. The Mn–N bond distances lie in the range of 2.0054–2.0207 Å. The Mn–O bond distances for the coordinated water molecules range from 2.2200–2.314 Å, indicating significant distortion in the octahedral structures imparted by tetragonal elongation anticipated in high-spin d4 Mn(III) complexes. The Mn–O and Mn–N bond distances are comparable to those reported structures that contain water, methanol, DMF, acrylamide etc. as axial ligands along with [Ag(CN)2]−, Br−, SbF6−, ClO4−etc. as counterions.44–49 The plane of the phenyl rings at the meso positions is not same with the plane of the porphyrin ring, suggesting a lack of conjugation between the two aromatic systems.
| Empirical formula | C44H28F4MnN4O2(C6H11O4)·3(C3H6O2) (Mn4) | C44H28Cl4MnN4O2(C6H11O4)·4(C3H6O2) (Mn5) |
|---|---|---|
| a R = ∑||Fo| − |Fc||/∑|Fo|. b R w = {∑[w(Fo2 − Fc2)2]/∑[w(Fo)2]}½; w = [σ2(Fo)2 + (AP)2 + BP]−1, where P = (Fo2 + 2Fc2)/3, A = 0.0921; B = 0.8341 (for Mn4) and A = 0.0871; B = 1.3753 (for Mn5). | ||
| F w, g M−1 | 1145.02 | 1284.90 |
| Crystal system | Triclinic | Monoclinic |
| Space group |
P![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) |
P21/c |
| a, Å | 9.2702(12) | 12.7394(2) |
| b, Å | 14.4396(18) | 9.48030(10) |
| c, Å | 22.878(3) | 26.7201(4) |
| α, ° | 72.099(4) | 90 |
| β, ° | 89.438(5) | 98.5660(10) |
| γ, ° | 72.965(5) | 90 |
| V, Å3 | 2775.4(6) | 3191.08(8) |
| Z | 2 | 2 |
| T, K | 296(2) | 294(2) |
| ρ calcd, g cm−3 | 1.370 | 1.337 |
| λ, Å | 0.71073 | 1.54184 |
| μ, cm−1 | 0.318 | 3.768 |
| Data/restraints/parameters | 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 091/6/755 091/6/755 |
6028/1/378 |
| F(000) | 1192 | 1336 |
| Goodness-of-fit | 1.039 | 1.076 |
| R(Fo)a, I > 2σ(I) [Rw(Fo)b] | 0.0707 [0.1664] | 0.0536 [0.1531] |
| R (all data) [Rw (all data)] | 0.1261 [0.1932] | 0.0594 [0.1582] |
| Largest diff. peak and hole (e Å−3) | 0.512, −0.475 | 0.644, −0.468 |
Binding affinity for HSA
Neutral and anionic porphyrins are known for their ability to form complexes with HSA.50 The affinity of the synthesized metalloporphyrins Mn1–Mn5 for human serum albumin (HSA) was evaluated by determining their binding constant values (KHSA) with HSA using a fluorescence quenching experiment. The emission intensity of HSA at 334 nm was gradually quenched, accompanied by a significant blue shift, in Tris-HCl buffer (pH 7.2) on increasing the concentration of Mn1–Mn5 (Fig. S38, ESI†). The observed decrease in emission intensity and the blue shift are attributed to structural changes in HSA on binding to the Mn(III) porphyrins, leading to perturbations in the microenvironment around the Trp-214 residue responsible for fluorescence. The blue shift in the emission maximum indicates a more hydrophobic environment surrounding the Trp-214 residue upon binding of the Mn(III) porphyrins to HSA.51,52 A linear plot of I0/I vs. [compound] yielded KHSA values in the following order: Mn5 > Mn4 > Mn2 > Mn3 ≥ Mn1 (Table S6, ESI†). Although this order is not completely parallel to their lipophilicity, it can be concluded that an increase in lipophilic character generally favors interaction with HSA. The KHSA values ranged from 9.08 (±0.01) × 104 to 4.03 (±0.02) × 105 M−1. Compared to these, the manganese derivative of protoporphyrin IX, Mn(III)PPIX, exhibited a relatively higher binding affinity towards HSA, with a KHSA value of 2.13 (±0.07) × 106 M−1.53 The moderate binding affinities of Mn1–Mn5 for HSA suggest that they can probably bind reversibly to HSA, potentially allowing them to be transported through blood plasma and released at the target tissue.Measurement of lipophilicity
An essential property for a PDT drug is its ability to incorporate and localize within vital cellular compartments. This is particularly crucial for hydrophobic compounds, for which passive diffusion across the cytoplasmic membrane is often a pre-requisite for cellular uptake.54 A partition coefficient between n-octanol and water serves as an indicator of a drug candidate's potential to diffuse through the lipid bilayer of cell membranes. The n-octanol/water partition coefficients (expressed as log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) PO/W) were determined using the shake-flask method and were found to follow the order: Mn5 > Mn2 > Mn4 > Mn1 ≥ Mn3 (Table 1). The Mn(III) porphyrins bearing –Cl and –CH3 substituents at their periphery were found to be highly lipophilic, with log
PO/W) were determined using the shake-flask method and were found to follow the order: Mn5 > Mn2 > Mn4 > Mn1 ≥ Mn3 (Table 1). The Mn(III) porphyrins bearing –Cl and –CH3 substituents at their periphery were found to be highly lipophilic, with log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) PO/W > 1.
PO/W > 1.
Detection of 1O2 generation using DPBF as a chemical probe
Paramagnetic metalloporphyrins such as those of Co(II) and Cu(II) are reported to be unsuitable candidates for 1O2 generation process.16,55 To evaluate whether the synthesized Mn(III) porphyrins are capable of generating 1O2 on electronic excitation, both extracellular and intracellular experiments were conducted using UV-visible titration with DPBF and a DCFDA assay as probes (Fig. 4). It is observed that DPBF, in the presence of Mn(III) porphyrins Mn1–Mn5, exhibited significant decrease in its absorption maximum under visible light irradiation (Fig. 4a and Fig. S39, ESI†). In contrast, the same experiment performed in the dark showed no significant changes in the DPBF absorption spectrum (Fig. 4a and Fig. S40, ESI†). Control experiments confirmed that DPBF did not undergo self-photodegradation under similar experimental conditions (Fig. S40, ESI†). To further verify whether the observed photodegradation of DPBF in the presence of Mn(III) porphyrins was due to 1O2 generation, a control experiment was performed using NaN3 as a 1O2 quencher (Fig. 4a and Fig. S41, ESI†). This results of this experiment closely resembled the photodegradation profile of untreated DPBF. This indicates that the significant decrease in absorption maximum of DPBF in the presence of Mn(III) porphyrins upon photoirradiation under visible light is due to the generation of 1O2. The rate constant values obtained from the linear plot of −ln([A]t/[A]0) vs. kt were 5.82 × 10−3 s−1, 8.20 × 10−3 s−1, 8.90 × 10−3 s−1, 1.14 × 10−2 s−1 and 9.10 × 10−3 s−1 for Mn1, Mn2, Mn3, Mn4 and Mn5, respectively, following the order: Mn4 > Mn5 ≥ Mn3 > Mn2 > Mn1 (Fig. S42, ESI†). The 1O2 quantum yields calculated from these rate constants were found to be 0.09, 0.11, 0.13, 0.17 and 0.14 for Mn1, Mn2, Mn3, Mn4 and Mn5, respectively (Table 1). The whole sets of these experiments were further repeated in DMSO, where a similar trend of 1O2 quantum yields was observed (Fig. S43–S46, ESI†). However, 1O2 quantum yield values obtained in DMSO were in the range of 0.04 to 0.09, which were lower compared to those in DMF (Table 1). This difference is likely due to the variable lifetime of 1O2 in different solvents.56 Although paramagnetic Mn(III) metal centre in porphyrin-type macrocycles promotes non-radiative decay pathways that limit 1O2 generation process, it does not completely eliminate the process. Thus, we observed some residual generation of 1O2 in the Mn(III) porphyrins. The unpaired electrons on Mn(III) in these compounds interact with the ring electrons and hence the (π,π*) states are not the typical singlets and triplets. The ground state becomes a quintet (5S0), and the quintet excited states (5S2 and 5S1) are derived from the excited ring (π,π*) singlet state, while a “tripmultiplet” manifold (3T1, 5T1, 7T1) arises from the lowest ring (π,π*) triplet state. Paramagnetism in Mn(III) porphyrin increases spin–orbit coupling, which in turn significantly enhances the rate of intersystem crossing compared to other metalloporphyrins (e.g. for MnIIITPP(Cl) τISC = 2.8 ps).57 Consequently, singlet excited states are rapidly converted to triplet manifolds. Although strong spin–orbit coupling also influences non-radiative decay of excited triplet states, some of these triplet excited states are still relatively long-lived compared to their excited singlet states. A portion of 5T1 state decays back to the ground state, while another portion decays to comparatively long-lived 7T1 state (Fig. S47, ESI†). This state can likely participate in energy transfer with molecular oxygen to generate 1O2. According to Harriman, for MnIIITPP(Cl), the energies of the 5T1 and 7T1 states are 1.8 eV and 1.6 eV, respectively, while the energy required for 1O2 generation is 0.98 eV.57–60 Although, the ΦΔ values of the Mn(III) porphyrins are lower than those of typical type-II PDT agents, they may still contribute to their photocytotoxicity due to their relatively better solubility and stability profiles.Mn(III)/Mn(II) redox shuttle in visible light
Mn(III) porphyrins can reversibly switch between +2 and +3 oxidation states. In general, Mn(III) porphyrins that bearing high positive charges on their periphery, such as water-soluble cationic Mn(III) porphyrins, are considered good SOD mimics. These compounds exhibit an optimum positive E1/2 value for the Mn(III)/Mn(II) redox cycle, making them readily reducible by common reducing agents.61 However, the E1/2 values of the Mn(III)/Mn(II) redox cycle for these Mn(III) porphyrins (Mn1–Mn5) fall within the negative range, rendering them difficult to reduce, especially under oxygenated conditions. As a result, reduction of such porphyrins is typically carried out under deaerated conditions.62 However, when we carried out the reduction experiment of these Mn(III) porphyrins with ascorbic acid, which is a cellular reductant, in 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 DMSO
1 DMSO![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Tris-HCl buffer in air with light irradiation, it was observed that with gradual increase in light irradiation, the UV-visible spectra of the porphyrins show some distinct changes that correlate to their reduction to Mn(II) porphyrin. The absorption intensity of the Soret band at ∼460 nm gradually decreases and a new band at approximately 434–440 nm appears, which is typical for Mn(II) porphyrins. The other two less intense bands observed at UV region (∼370–400 nm) also exhibited decrease in their absorption intensity, and the two Q-bands shifted somewhat towards the red region (Fig. 5 and Fig. S48, ESI†).37,62 When the light irradiation was stopped and the system was left exposed to air, it exhibited reversibility as shown by their UV-visible spectra (Fig. 5 and Fig. S49, ESI†). These findings suggest that light irradiation can catalyse the reduction of these Mn(III) porphyrins even in aerated conditions. Mn(III) porphyrins are generally stable against photodecomposition, and our photostability data confirmed this behavior (Fig. S30, ESI†). When the same experiment was conducted in the absence of light, the UV-visible spectra of these Mn(III) porphyrins did not show significant changes, suggesting that they are stable against the reduction process even at a higher concentration of ascorbic acid (5 mM), except Mn5 (Fig. 5 and Fig. S50, ESI†). This photo-redox activity of Mn(III) porphyrins in turn can imbalance the overall redox status of cancer cell. Some literature reports suggest that Mn(II) porphyrins are oxidized to Mn(III) porphyrin by molecular oxygen and, generating H2O2 during the process. The hydrogen peroxide produced may then lead to the formation of ROS via the Fenton reaction, which can induce oxidative stress that in turn can kill cancer cells.63,64 However, under our experimental conditions, we were unable to detect H2O2, likely due to its very low concentration. No reduction of Mn(III) to Mn(II) porphyrin was observed in the presence of glutathione, either in the dark or under visible light (Fig. S51, ESI†). This is likely due to coordination of the glutathione carboxylate group to the axial position of Mn(III) porphyrin, as previously in the literature.62 Furthermore, we were also unable to detect any hydroxyl radical formation by the Mn(III) porphyrins at physiological pH, either in the dark or under visible light irradiation, using terephthalic acid as a probe (Fig. S52, ESI†). Nonetheless, literature reports indicate that at alkaline pH (10–11), Mn(III) porphyrins can generate hydroxyl radicals through electron transfer reactions with hydroxide ions.37
Tris-HCl buffer in air with light irradiation, it was observed that with gradual increase in light irradiation, the UV-visible spectra of the porphyrins show some distinct changes that correlate to their reduction to Mn(II) porphyrin. The absorption intensity of the Soret band at ∼460 nm gradually decreases and a new band at approximately 434–440 nm appears, which is typical for Mn(II) porphyrins. The other two less intense bands observed at UV region (∼370–400 nm) also exhibited decrease in their absorption intensity, and the two Q-bands shifted somewhat towards the red region (Fig. 5 and Fig. S48, ESI†).37,62 When the light irradiation was stopped and the system was left exposed to air, it exhibited reversibility as shown by their UV-visible spectra (Fig. 5 and Fig. S49, ESI†). These findings suggest that light irradiation can catalyse the reduction of these Mn(III) porphyrins even in aerated conditions. Mn(III) porphyrins are generally stable against photodecomposition, and our photostability data confirmed this behavior (Fig. S30, ESI†). When the same experiment was conducted in the absence of light, the UV-visible spectra of these Mn(III) porphyrins did not show significant changes, suggesting that they are stable against the reduction process even at a higher concentration of ascorbic acid (5 mM), except Mn5 (Fig. 5 and Fig. S50, ESI†). This photo-redox activity of Mn(III) porphyrins in turn can imbalance the overall redox status of cancer cell. Some literature reports suggest that Mn(II) porphyrins are oxidized to Mn(III) porphyrin by molecular oxygen and, generating H2O2 during the process. The hydrogen peroxide produced may then lead to the formation of ROS via the Fenton reaction, which can induce oxidative stress that in turn can kill cancer cells.63,64 However, under our experimental conditions, we were unable to detect H2O2, likely due to its very low concentration. No reduction of Mn(III) to Mn(II) porphyrin was observed in the presence of glutathione, either in the dark or under visible light (Fig. S51, ESI†). This is likely due to coordination of the glutathione carboxylate group to the axial position of Mn(III) porphyrin, as previously in the literature.62 Furthermore, we were also unable to detect any hydroxyl radical formation by the Mn(III) porphyrins at physiological pH, either in the dark or under visible light irradiation, using terephthalic acid as a probe (Fig. S52, ESI†). Nonetheless, literature reports indicate that at alkaline pH (10–11), Mn(III) porphyrins can generate hydroxyl radicals through electron transfer reactions with hydroxide ions.37
Intracellular ROS generation
DCFDA assay was employed to evaluate the ability of Mn(III) porphyrins to generate reactive oxygen species in cancer cells, using the most active Mn(III) porphyrin, i.e.Mn4 (10 μM), as a representative compound. H2DCF, formed intracellularly from DCFDA via enzymatic conversion, is oxidized by ROS to yield DCF.65 The emissive property of DCF in green light (λem = 528 nm) was used to monitor the ability of Mn4 to generate ROS in HeLa cells through flow cytometry. A significant increase in DCF emission, resulting from intracellular oxidation of H2DCF by ROS generated upon photoexcitation of Mn4, was observed at various time points, viz. 15, 30, 45 and 60 min, in a time-dependent manner, as represented in the bar diagram (Fig. 4b). This was evident from the significant increase in emission count of DCF at different time in the light-exposed HeLa cells treated with Mn4. Control experiments using only cells in the dark, cells exposed to light alone and Mn4-treated cells kept in the dark did not show any significant ROS generation. The ROS generated by Mn4 on light exposure is believed to be primarily responsible of the observed cell death.Photocytotoxicity study
The toxicities of Mn(III) porphyrins Mn1–Mn5 against HeLa (human cervical carcinoma) cancer cell line under dark and visible light conditions (400–700 nm) were evaluated using the MTT assay, 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (Fig. 6). The compounds Mn1–Mn5 exhibited remarkable dose-dependent photocytotoxicity in visible light (400–700 nm) in HeLa cell, while showing minimal toxicity in the dark at concentrations up to 50 μM. As the solubility of the compounds were not very high in the permissible composition of DMSO–DMEM mixture, the maximum concentration of the compounds employed was restricted to 50 μM. A >5–10-fold increase in cytotoxicity was observed for Mn1–Mn5 in HeLa cells following 1 h of light exposure, compared to compound-treated cells kept in the dark under similar experimental conditions. Among the tested compounds, metalloporphyrin Mn4 was found to be the most phototoxic to HeLa cells, with an IC50 value of ∼4.9 μM under visible light, while remaining non-toxic in the dark up to a concentration of 50 μM. The phototoxicity index (PI), defined as the ratio of the IC50 value in the dark to that in the light, was >10 for Mn4. Mn5, which contains a chloro substituent in the para position of the meso-phenyl rings, exhibited significant photocytotoxicity, with an IC50 value of ∼5.5 μM in visible light and no apparent toxicity in the dark. This implies that the toxicity of the compound is enhanced by more than 9-fold (PI) under visible light. Mn(III) porphyrins bearing –CH3 and –OCH3 substituents in the para positions of the meso-phenyl rings of the porphyrins, i.e.Mn2 and Mn3, exhibited slightly higher IC50 values of ∼6.25 μM and ∼6.70 μM, respectively, under light exposure. Both compounds showed no significant toxicity toward HeLa cells in the dark at concentrations up to 50 μM. The PI values were found to be greater than 8.0 and 7.5 for Mn2 and Mn3, respectively. (Diaqua)meso(tetraphenylporphyrinato)manganese(III) propionate, i.e.Mn1, showed the lowest phototoxicity among the series, with an IC50 value of ∼9.54 μM in visible light. It is reported that porphyrins containing paramagnetic metal ions in the core such as Co(II), Cu(II), Mn(III) generally exhibit very poor singlet oxygen quantum yields and are therefore considered unsuitable for PDT.14,15 However, some studies have demonstrated that Mn(III) porphyrins can exert PDT effects via the generation of reactive oxygen species.22,23,66 Mn(III) porphyrin containing carboxyl groups at the meso positions showed a significant enhancement in cytotoxicity in HepG2 cells, with an IC50 value of ∼19.0 μM when irradiated with 625 nm LED light for 60 min. Photoinduced production of superoxide and hydroxide radicals was reported to be responsible for the photocytotoxicity, as revealed by EPR studies.22 A Mn(III) porphyrin, viz. Pt-MnPor-PPh3, bearing triphenylphosphonium cation and Pt(II) moieties at its meso-peripheral positions, exhibited significant PDT effect against HeLa cells upon light irradiation for 10 min using yellow light (4 J cm−2) generated from a 400 W tungsten lamp. The IC50 value was reported to be 30.74 μM under light irradiation, whereas no significant cytotoxicity was observed in the dark up to a concentration of 50 μM.66 Recently, a water-soluble Mn(III) porphyrin bearing cationic pyridyl side groups has been reported to show significant photocytotoxicity in HeLa cells when irradiated with a green LED of 527 nm.23 The IC50 values were found to be 178 and 4 μM in the dark and under light irradiation, respectively, giving a PI value of 44.5. The IC50 values of the Mn(III) porphyrins tested, including the Mn(III) porphyrin bearing cationic pyridyl groups, Photofrin®, protoporphyrin IX (PpIX) and cisplatin, are listed in Table 3.23,67–69 The observed photocytotoxicity is comparable to that of the clinically used PDT drug Photofrin®.67 PpIX, another established anticancer PDT drug, is found to be slightly more toxic than these metalloporphyrins, with IC50 values of 2.8 ± 0.5 μM and 2.1 ± 0.3 μM when irradiated at 510 nm (20 min, 5.0 J cm−2) at 540 nm (40 min, 9.5 J cm−2), respectively.68 The widely used chemotherapeutic agent cisplatin does not show any significant photocytotoxicity in HeLa cells under similar conditions.69 Overall, it can be concluded that the Mn(III) porphyrins exhibit significant photodynamic activity in HeLa cells under visible light irradiation through the generation of cytotoxic ROS. The MTT assay for the free-base porphyrins was not performed due to their poor solubility in the acceptable composition of DMSO-DMEM for photochemotherapeutic studies in cancer cells. In general, singlet oxygen quantum yields (ΦΔ) of free bases (e.g. ΦΔ for TPP 0.64 in DMF) are higher than those of the corresponding Mn(III) porphyrins. However, given their better solubility and lower aggregation properties in aqueous solutions, which normally limit the potential of hydrophobic free-base porphyrins as PS, the use of Mn(III) porphyrins for photodynamic therapeutic application cannot be precluded.42,70 | ||
| Fig. 6 Cell viability plots showing the cytotoxic effects of Mn1–Mn5 (a–e) on HeLa cells in the dark (black) and under visible light (400–700 nm, 10 J cm−2, 1 h) (red). | ||
| Compound | HeLa | ||
|---|---|---|---|
| IC50 (μM) darka | IC50 (μM) lightb | PIc | |
| a The IC50 values correspond to 24 h treatment followed by 24 h incubation in the dark. b The IC50 values correspond to 24 h treatment in the dark followed by photo-exposure to visible light (400–700 nm, 10 J cm−2) for 1 h. Subsequently, cells were incubated for 23 h in the dark. c PI (phototoxic index) is the ratio of the IC50 in the dark to that under visible light exposure. d The IC50 values are taken from ref. 23 and correspond to incubation of cells with the compound for 24 h in the dark, followed by illumination with a green LED (527 nm) for 15 min at a power of 45 mW. e The Photofrin® IC50 values are taken from ref. 67. Light source reported: red light at 633 nm. f The IC50 values are taken from ref. 68. g It corresponds to irradiation at 510 nm (20 min, 5.0 J cm−2). h It corresponds to irradiation at 540 nm (40 min, 9.5 J cm−2). i The IC50 value for 4 h treatment is taken from ref. 69. | |||
| Mn1 | >50 | 9.54 ± 1.2 | >5.2 |
| Mn2 | >50 | 6.25 ± 1.1 | >8.0 |
| Mn3 | >50 | 6.70 ± 0.9 | >7.5 |
| Mn4 | >50 | 4.93 ± 0.7 | >10.1 |
| Mn5 | >50 | 5.50 ± 0.7 | >9.1 |
| MnT(HeOH)PyP5+ | 178d | 4d | 44.5d |
| Photofrin® | >41e | 4.3 ± 0.2e | — |
| PpIX | >100f,g | 2.8 ± 0.5f,g | >35.7f,g |
| >100f,h | 2.1 ± 0.3f,h | >47.6f,h | |
| Cisplatin | 71.3 ± 2.9i | 68.7 ± 3.4i | — |
Conclusion
In summary, five meso-tetraaryl Mn(III) porphyrins bearing various para substituents on the meso-phenyl ring were synthesized, comprehensively characterized, and evaluated for their visible light-induced anticancer activity. The Mn(III) porphyrins exhibited strong binding affinity toward human serum albumin. Electrochemical studies via cyclic voltammetry provided insights into their redox behaviour, while lipophilicity studies highlighted the influence of substituents on their aqueous solubility and membrane permeability. Upon visible light irradiation, these complexes induced significant cytotoxicity in HeLa cells through the generation of ROS, as confirmed by DCFDA assay, while remaining essentially non-toxic in the dark. Photocytotoxicity data demonstrated that fluorine- and chlorine-substituted Mn(III) porphyrins, i.e., Mn4 and Mn5, respectively, displayed enhanced photocytotoxicity toward HeLa cells compared to the other compounds. In addition, these findings highlight the importance of substituent effects on the photophysical and biological properties of Mn(III) porphyrins, offering valuable insights for the rational design of next-generation photosensitizers for targeted cancer therapy. This study opens up the unexplored area of 3d metalloporphyrins for photodynamic therapy.Conflicts of interest
There are no conflicts to declare.Data availability
All relevant data supporting the findings of this study are included within the article and its ESI.†Acknowledgements
The authors are grateful to Department of Science and Technology (DST)-SERB, Government of India, Core Research Grant (CRG) (CRG/2023/002246) for financial assistance. We also acknowledge financial support from the DST-PURSE 2022 program (TPN-84912). The authors are thankful to DST and Department of SAIF, Gauhati University, for single crystal X-ray diffractometer facility. We also thank Dr Ranjit Thakuria, Dr Tejender S. Thakur, Dr S. Brahma and Dr Debajit Sarma for their kind assistance in X-ray data collection and structure solution. We sincerely thank Prof. A. R. Chakravarty, Indian Institute of Science, Bangalore, for allowing us to use the cell culture facility in his laboratory. JPS thanks DST-SERB for research fellowship.References
- M. O. Senge, N. N. Sergeeva and K. J. Hale, Chem. Soc. Rev., 2021, 50, 4730–4789 RSC.
- C. J. Kingsbury and M. O. Senge, Coord. Chem. Rev., 2021, 431, 213760 CrossRef CAS.
- M. A. Rajora, J. W. H. Lou and G. Zheng, Chem. Soc. Rev., 2017, 46, 6433–6469 RSC.
- S. Ishihara, J. Labuta, W. V. Rossom, D. Ishikawa, K. Minami, J. P. Hill and K. Ariga, Phys. Chem. Chem. Phys., 2014, 16, 9713–9746 RSC.
- A. F. dos Santos, D. R. Q. de Almeida, L. F. Terra, M. S. Baptista and L. Labriola, J. Cancer Metastasis Treat., 2019, 5, 25 Search PubMed.
- X. Wang, J. Peng, C. Meng and F. Feng, Chem. Sci., 2024, 15, 12234–12257 RSC.
- H. Abrahamse and M. R. Hamblin, Biochem. J., 2016, 473, 347–364 CrossRef CAS PubMed.
- L. M. Moreira, F. V. D. Santos, J. P. Lyon, M. Maftoum-Costa, C. Pacheco-Soares and N. S. D. Silva, Aust. J. Chem., 2008, 61, 741–754 CrossRef CAS.
- M. Ethirajan, Y. Chen, P. Joshi and R. K. Pandey, Chem. Soc. Rev., 2011, 40, 340–362 RSC.
- H. Shi, C. Imberti and P. J. Sadler, Inorg. Chem. Front., 2019, 6, 1623–1638 RSC.
- M. R. Detty, S. L. Gibson and S. J. Wagner, J. Med. Chem., 2004, 47, 3897–3915 CrossRef CAS PubMed.
- J. Kou, D. Dou and L. Yang, Oncotarget, 2017, 8, 81591–81603 CrossRef PubMed.
- Q. Zhang, J. He, W. Yu, Y. Li, Z. Liu, B. Zhou and Y. Liu, RSC Med. Chem., 2020, 11, 427–437 RSC.
- A. P. Castano, T. N. Demidova and M. R. Hamblin, Photodiagn. Photodyn. Ther., 2004, 1, 279–293 CrossRef CAS PubMed.
- T. L. C. Figueiredo, R. A. W. Johnstone, A. M. P. S. Sørensen, D. Burget and P. Jacques, Photochem. Photobiol., 1999, 69, 517–528 CAS.
- P. M. Antoni, A. Naik, I. Albert, R. Rubbiani, S. Gupta, P. Ruiz-Sanchez, P. Munikorn, J. M. Mateos, V. Luginbuehl, P. Thamyongkit, U. Ziegler, G. Gasser, G. Jeschke and B. Spingler, Chem. – Eur. J., 2014, 20, 1–6 CrossRef.
- L. Li and X. Yang, Oxid. Med. Cell. Longevity, 2018, 2018 DOI:10.1155/2018/7580707.
- B. Hasan, A. Tovmasyan, I. Batinic-Haberle and L. Benov, Redox Rep., 2021, 26, 85–93 CrossRef CAS PubMed.
- A. Tovmasyan, H. Sheng, T. Weitner, A. Arulpragasam, M. Lu, D. S. Warner, Z. Vujaskovic, I. Spasojevic and I. Batinic-Haberle, Med. Princ. Pract., 2013, 22, 103–130 CrossRef PubMed.
- A. Tovmasyan, R. S. Sampaio, M.-K. Boss, J. C. Bueno-Janice, B. H. Bader, M. Thomas, J. S. Reboucas, M. Orr, J. D. Chandler, Y.-M. Go, D. P. Jones, T. N. Venkatraman, S. Haberle, N. Kyui, C. D. Lascola, M. W. Dewhirst, I. Spasojevic, L. Benov and I. Batinic-Haberle, Free Radical Biol. Med., 2015, 89, 1231–1247 CrossRef CAS PubMed.
- S. Banfi, E. Cassani, E. Caruso and M. Cazzaro, Bioorg. Med. Chem., 2003, 11, 3595–3605 CrossRef CAS PubMed.
- L. Shi, Y.-Y. Jiang, T. Jiang, W. Yin, J.-P. Yang, M.-L. Cao, Y.-Q. Fang and H.-Y. Liu, Molecules, 2017, 22, 1084 CrossRef PubMed.
- T. Nemeth, A. Pallier, C. Çelik, Z. Garda, N. Yoshizawa-Sugata, H. Masai, É. Tóth and Y. Yamakoshi, Chem. Biomed. Imaging, 2025, 3, 5–14 CrossRef CAS PubMed.
- Y. H. Kim, S. D. Jung, M. H. Lee, C. Im, Y.-H. Kim, Y. J. Jang, S. K. Kim and D. W. Cho, J. Phys. Chem. B, 2013, 117, 9585–9590 CrossRef CAS PubMed.
- O. Kahn, Molecular Magnetism, VCH, Weinheim, 1993 Search PubMed.
- A. D. Adler, F. R. Longo, J. D. Finarelli, J. Goldmacher, J. Assour and L. Korsakoff, J. Org. Chem., 1967, 32, 476 CrossRef CAS.
- A. D. Adler, F. R. Longo, F. Kampas and J. Kim, J. Inorg. Nucl. Chem., 1970, 32, 2445–2448 CrossRef.
- N. Walker and D. Stuart, Acta Crystallogr., Sect. A: Found. Crystallogr., 1983, 39, 158–166 CrossRef.
- G. M. Sheldrick, Acta Crystallogr., Sect. A: Found. Crystallogr., 2008, 64, 112–122 CrossRef CAS PubMed.
- L. J. Farrugia, J. Appl. Crystallogr., 1997, 30, 565–565 CrossRef CAS.
- J. Seetharamappa and B. P. Kamat, Chem. Pharm. Bull., 2004, 52, 1053–1057 CrossRef CAS PubMed.
- J. Wu, D. G. J. Goodwin, K. Peter, D. Benoit, W. Li, D. H. Fairbrother and J. D. Fortner, Environ. Sci. Technol. Lett., 2014, 1, 490–494 CrossRef CAS.
- M. K. Raza, S. Gautam, A. Garai, K. Mitra, P. Kondaiah and A. R. Chakravarty, Inorg. Chem., 2017, 56, 11019–11029 CrossRef CAS PubMed.
- L.-X. Wang, J.-W. Li, J.-Y. Huang, J.-H. Li, L.-J. Zhang, D. Æshea and Z.-L. Chen, Tumor Biol., 2015, 36, 6839–6847 CrossRef CAS PubMed.
- S. Meng, Z. Xu, G. Hong, L. Zhao, Z. Zhao, J. Guo, H. Ji and T. Liu, Eur. J. Med. Chem., 2015, 92, 35–48 CrossRef CAS PubMed.
- P. Sen, R. Soy, S. Mgidlana, J. Mack and T. Nyokong, Dyes Pigm., 2022, 203, 110313 CrossRef CAS.
- A. Harriman and G. Porter, J. Chem. Soc., Faraday Trans. 2, 1979, 75, 1543–1552 RSC.
- H. Wang and J. A. Joseph, Free Radical Biol. Med., 1999, 27, 612–616 CrossRef CAS PubMed.
- T. Mosmann, J. Immunol. Methods, 1983, 65, 55–63 CrossRef CAS PubMed.
- C. C. Wamser and A. Ghosh, JACS Au, 2022, 2, 1543–1560 CrossRef CAS PubMed.
- L. E. Lieske, S. L. Hooe, A. W. Nichols and C. W. Machan, Dalton Trans., 2019, 48, 8633–8641 RSC.
- N. Mariño-Ocampo, L. Dibona-Villanueva, E. Escobar-Álvarez, D. Guerra-Díaz, D. Zúñiga-Núñez, D. Fuentealba and J. Robinson-Duggon, Photochem. Photobiol., 2023, 99, 469–497 CrossRef PubMed.
- B. Babu, E. Amuhaya, D. Oluwole, E. Prinsloo, J. Mack and T. Nyokong, Med. Chem. Commun., 2019, 10, 41–48 RSC.
- C. L. Hill and M. M. Williamson, Inorg. Chem., 1985, 24, 2836–2841 CrossRef CAS.
- M. M. Williamson and C. L. Hill, Inorg. Chem., 1987, 26, 4155–4160 CrossRef CAS.
- N. Xu, A. W. Bevak, B. R. Armstrong and D. R. Powell, Polyhedron, 2017, 127, 432–437 CrossRef CAS.
- R. Soman, S. Sujatha and C. Arunkumar, J. Porphyrins Phthalocyanines, 2016, 20, 833–842 CrossRef CAS.
- X. Chen, Z. D. Zhao, P. Wang and D. P. Zhang, Russ. J. Coord. Chem., 2014, 40, 320–324 CrossRef CAS.
- Z. N. Zahran, J. Lee, S. S. Alguindigue, M. A. Khan and G. B. Richter-Addo, Dalton Trans., 2004, 1, 44–50 RSC.
- O. A. Chaves, T. V. Acunha, B. A. Iglesias, C. S. H. Jesus and C. Serpa, J. Mol. Liq., 2020, 301, 112466 CrossRef CAS.
- I. Ling, M. Taha, N. A. Al-Sharji and O. K. Abou-Zied, Spectrochim. Acta, Part A, 2018, 194, 36–44 CrossRef CAS PubMed.
- A. Costa-Tuna, O. A. Chaves, R. J. S. Loureiro, S. Pinto, J. Pina and C. Serpa, Int. J. Biol. Macromol., 2024, 255, 128210 CrossRef CAS PubMed.
- J. Hu, E. H. Soraiz, C. N. Johnson, B. Demeler and L. Brancaleon, Int. J. Biol. Macromol., 2019, 134, 445–457 CrossRef CAS PubMed.
- J. A. Arnott and S. L. Planey, Expert Opin. Drug Discovery, 2012, 7, 863–875 CrossRef CAS PubMed.
- S. Mathai, T. A. Smith and K. P. Ghiggino, Photochem. Photobiol. Sci., 2007, 6, 995–1002 CrossRef CAS PubMed.
- T. Entradasa, S. Waldrona and M. Volk, J. Photochem. Photobiol., B, 2020, 204, 111787 CrossRef PubMed.
- L.-L. Wang, S.-H. Peng, H. Wang, L.-N. Ji and H.-Y. Liu, Phys. Chem. Chem. Phys., 2018, 20, 20141–20148 RSC.
- X. Yan, C. Kirmaier and D. Holten, Inorg. Chem., 1986, 25, 4774–4777 CrossRef CAS.
- G. Porter and M. R. Wright, Discuss. Faraday Soc., 1959, 27, 18–27 RSC.
- S. Joseph and S. K. A. Kumar, Coord. Chem. Rev., 2023, 496, 215408 CrossRef CAS.
- A. Tovmasyan, S. Carballal, R. Ghazaryan, L. Melikyan, T. Weitner, C. G. C. Maia, J. S. Reboucas, R. Radi, I. Spasojevic, L. Benov and I. Batinic-Haberle, Inorg. Chem., 2014, 53, 11467–11483 CrossRef CAS PubMed.
- S. M. A. Pinto, A. R. R. Ferreira, D. S. S. Teixeira, S. C. C. Nunes, A. L. M. B. de Carvalho, J. M. S. Almeida, Z. Garda, A. Pallier, A. A. C. C. Pais, C. M. A. Brett, É. Tóth, M. P. M. Marques, M. M. Pereira and C. F. G. C. Geraldes, Chem. – Eur. J., 2023, 29, e202301442 CrossRef CAS PubMed.
- I. A. Duncan, A. Harriman and G. Porter, J. Chem. Soc., Faraday Trans. 2, 1980, 76, 1415–1428 RSC.
- I. Batinic-Haberle, A. Tovmasyan, Z. Huang, W. Duan, L. Du, S. Siamakpour-Reihani, Z. Cao, H. Sheng, I. Spasojevic and A. A. Secord, Oxid. Med. Cell. Longev., 2021, 2021 DOI:10.1155/2021/6653790.
- D. Yu, Y. Zha, Z. Zhong, Y. Ruan, Z. Li, L. Sun and S. Hou, Sens. Actuators, B, 2021, 339, 129878 CrossRef CAS.
- M. Yang, J. Deng, D. Guo, Q. Sun, Z. Wang, K. Wang and F. Wu, Dyes Pigm., 2019, 166, 189–195 CrossRef CAS.
- A. Bhattacharyya, A. Jameei, A. Garai, R. Saha, A. A. Karande and A. R. Chakravarty, Dalton Trans., 2018, 47, 5019–5030 RSC.
- J. Karges, U. Basu, O. Blacque, H. Chao and G. Gasser, Angew. Chem., Int. Ed., 2019, 58, 14334–14340 CrossRef CAS PubMed.
- S. Saha, D. Mallick, R. Majumdar, M. Roy, R. R. Dighe, E. D. Jemmis and A. R. Chakravarty, Inorg. Chem., 2011, 50, 2975–2987 CrossRef CAS PubMed.
- V. A. Oliveira, H. Terenzi, L. B. Menezes, O. A. Chaves and B. A. Iglesias, J. Photochem. Photobiol., B, 2020, 211, 111991 CrossRef CAS PubMed.
Footnote |
| † Electronic supplementary information (ESI) available: Characterization, selected X-ray crystallographic data, HSA binding data (Tables S1–S6, Scheme S1, Fig. S1–S52). CCDC 2408932, 2408982, 2408984 and 2408985 for compounds Mn1, Mn2, Mn4 and Mn5, respectively. For ESI and crystallographic data in CIF or other electronic format see DOI: https://doi.org/10.1039/d4dt03432e |
| This journal is © The Royal Society of Chemistry 2025 |