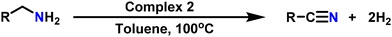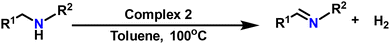Acceptorless oxidant-free dehydrogenation of amines catalyzed by Ru–hydride complexes of amide-acid/ester ligands†
Samanta
Yadav
and
Rajeev
Gupta
 *
*
Department of Chemistry, University of Delhi, Delhi-110 007, India. E-mail: rgupta@chemistry.du.ac.in
First published on 11th February 2025
Abstract
Traditional dehydrogenation of amines involves the transfer of hydrogen molecule(s) from a substrate to an acceptor. In acceptorless dehydrogenation, hydrogen gas is liberated without an oxidant, providing an efficient synthetic method. Acceptorless dehydrogenation of primary amines to nitriles without using an oxidant or hydrogen acceptor is significant yet challenging. Herein, we present efficient Ru-based catalysts capable of carrying out such a transformation with hydrogen gas as the only by-product. A new class of air and moisture-stable ruthenium–hydride complexes (1–4) of amide-acid/ester-based ligands have been synthesized and characterized. Crystal structures of two representative complexes, 2 and 3, illustrate the bidentate N–O coordination mode of the ligands. At the same time, additional binding sites are occupied by one hydride, one CO, and two PPh3 co-ligands. The catalytic behavior of these complexes is explored towards the oxidant-free, acceptorless, and selective dehydrogenation of primary and secondary amines affording nitriles and imines, respectively. Among four Ru(II) complexes, complex 2 showed the best catalytic activity for the dehydrogenation of amines. A wide variety of both primary and secondary amines were utilized to explore the substrate scope. The catalytic system tolerated both electron-withdrawing and electron-releasing substituents on amine substrates. Various control experiments and mechanistic studies were carried out to support the dehydrogenation of amines by using complex 2 as a representative catalyst.
Introduction
Nitriles are an eminent category of functional groups that are a part of multifaceted compounds involved in a wide variety of biologically active and industrial-relevant compounds.1–3 Various methods have been employed to synthesize nitriles, but they suffer from poor atom economy, limited selectivity, use of noxious reagents, and harsh reaction conditions.4 Typical synthetic methods include the cyanation of alkyl/aryl halides, dehydration of amides, metal-catalyzed cyanation, and Sandmeyer-type reactions.5–9 On the other hand, industrial methods usually depend on the ammoxidation process that operates at high temperatures.10 Most conventional methods for this transformation require an oxidant, which necessitates oxidant-compatible functional groups, thus significantly limiting the scope of amine oxidation reactions.11–13An alternative synthetic method involves transition-metal-catalyzed double dehydrogenation of primary amines, considered one of the most suitable synthetic methods.3,4,10,14,15 Although this method produces nitriles in excellent yields, many of such transition-metal catalyzed oxidation processes require large quantities of both oxidant and base for efficient catalytic conversion, which increases the amount of undesirable waste products while limiting the substrate scope for sensitive ones.9,10,15 Nevertheless, double dehydrogenation of primary amines to nitriles is a noteworthy but demanding task as it involves the removal of two molecules of hydrogen while several competing pathways, such as coupling and transamination reactions, co-exist (Scheme 1).3,9,16,17
 | ||
| Scheme 1 Acceptorless double dehydrogenation of primary amines, including a competing coupling pathway. | ||
Till now, only a few examples are available in the literature for the selective, acceptorless, and oxidant-free dehydrogenation of primary amines to nitriles. The first example was reported by Szymczak and co-workers in 2013 using (NNN)Ru–H complex as a catalyst (complex a, Scheme 2).3 The authors proposed an inner-sphere mechanism that involved proton transfer from a coordinated amine to Ru–H followed by H2 release.18 Another example is an iridium–pincer complex, [C6H3-2,6-(OPtBu2)2IrH2], which required a hydrogen acceptor and a base to afford nitrile products via an inner-sphere mechanism involving N–H oxidative addition followed by β-H elimination (complex b, Scheme 2).19 An elegant Ru complex was reported by Bera and co-workers for an acceptorless dehydrogenation of primary amines to nitriles although it required the addition of a strong base to achieve the enhanced reactivity (complex c, Scheme 2).14 Albrecht and co-workers have reported a pyridyl-triazole-based Ru–cymene complex which catalyzes the oxidation of both aromatic and aliphatic amines to nitriles using dioxygen as the terminal oxidant (complex d, Scheme 2).15 Very recently, Tang and co-workers have proposed a tandem outer-sphere–inner-sphere mechanism using a Co catalyst (complex e, Scheme 2) for the dehydrogenation of amines via an imine intermediate.4 However, due to limited examples of dehydrogenation of amine, it is still indistinct whether an inner-sphere, an outer-sphere, or a cooperative mechanism is dominant for such a double dehydrogenation process.18
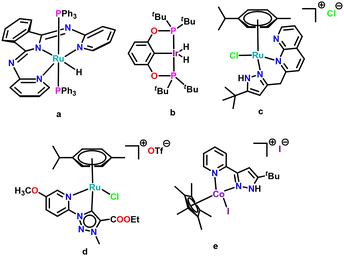 | ||
| Scheme 2 Selected examples of metal complexes known for catalyzing the dehydrogenation of primary amines. | ||
Although these examples highlight the significance of acceptorless dehydrogenation of amines, harsh reaction conditions, including the strong bases, demand the development of newer catalysts.4,14,20 Herein, we present Ru–hydride-based complexes for an acceptorless and oxidant-free dehydrogenation (DH) of amines. This work delineates the synthesis, characterization, and utilization of ruthenium complexes of pyridine–amide-based ligands for the catalytic conversion of amines to nitriles. The DH catalytic activity is found to be influenced by both the selection of a Ru(II) complex and the associated ligand. We show that an elegant ligand design can overcome challenges associated with acceptorless and oxidant-free DH of amines.
Results and discussion
Synthesis and characterization of ligands and their Ru(II) complexes
The amide-based ligands appended with ester (HL1 and HL3) and carboxylic acid groups (HL2 and HL4) were designed as viable hydrogen bond acceptors or donors to facilitate substrate binding and activation.21,22 The ester–amide ligands HL1 and HL3 were synthesized by coupling 2-picolinic acid with 4-aminoethyl benzoate or 3-aminoethyl benzoate. The acid–amide-based ligands HL2 and HL4 were synthesized by the base-catalyzed hydrolysis of HL1 and HL3, respectively. These ligands react with [Ru(H)(Cl)(CO)(PPh3)3] in a MeOH–CHCl3 mixture to form the corresponding [Ru–H] complexes 1–4 (Scheme 3). Both ligands and their Ru(II) complexes were thoroughly characterized using various spectroscopic techniques.The CO stretching bands for complexes 1–4 were observed at 1921–1930 cm−1.23 The Ru–H signals were found to merge with the CO bands and appeared at 1957–1962 cm−1.24,25 The C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O group of ester in 1 and 3 were displayed at a higher wave number, i.e., 1706 and 1709 cm−1, compared to the acid group in 2 and 4, which appeared at 1699 and 1697 cm−1, respectively.23 The amidic C
O group of ester in 1 and 3 were displayed at a higher wave number, i.e., 1706 and 1709 cm−1, compared to the acid group in 2 and 4, which appeared at 1699 and 1697 cm−1, respectively.23 The amidic C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O bands for complexes 1–4 (1602–1610 cm−1) were blue-shifted compared to the respective ligands (1575–1592 cm−1) (Fig. S1–S8, ESI†). This advocates their coordination with the metal center (vide infra).24,25 The amidic N–H stretches in ligands resonated at 3319–3342 cm−1. These bands appeared even after metal complexation but were found red-shifted at 3225–3265 cm−1. Such a fact suggests that a ligand binds with a Ru(II) ion through an amidic–O atom, subsequently confirmed by the X-ray crystallography (vide infra).
O bands for complexes 1–4 (1602–1610 cm−1) were blue-shifted compared to the respective ligands (1575–1592 cm−1) (Fig. S1–S8, ESI†). This advocates their coordination with the metal center (vide infra).24,25 The amidic N–H stretches in ligands resonated at 3319–3342 cm−1. These bands appeared even after metal complexation but were found red-shifted at 3225–3265 cm−1. Such a fact suggests that a ligand binds with a Ru(II) ion through an amidic–O atom, subsequently confirmed by the X-ray crystallography (vide infra).
In the 1H NMR spectra of Ru(II) complexes, the aromatic proton signals were shifted downfield compared to the ligands (Fig. S9–S16, ESI†). The signals for the N–H groups remain intact in 1–4 but were noted at higher δ values than their corresponding ligands. For complexes 1–4, the hydride signal appeared as a triplet at −10.27 (JH,P = 18.8 Hz), −10.23 (JH,P = 18.7 Hz), −10.20 (JH,P = 18.7 Hz), and −10.38 ppm (JH,P = 18.8 Hz), respectively.26,27 The 13C NMR spectra of complexes 1–4 showed CO group resonating at 203–211 ppm (Fig. S17–S24, ESI†). The 31P NMR spectra for complexes 1–4 exhibited a singlet at 45.58–46.21 ppm (Fig. S25–S28, ESI†).26,28 Such a fact asserted the presence of two PPh3 groups trans to each other in a magnetically equivalent environment around a Ru center. The absorption spectra of complexes 1–4 exhibit bands at 415–435 nm and 300–305 nm, tentatively assigned to charge transfer and ligand-based transitions, respectively (Fig. S29, ESI†).24,26 All ligands and their metal complexes were also characterized by the ESI+ mass spectra (Fig. S30–S37, ESI†). The observed isotopic distribution pattern for complexes 1–4 matched excellently with the simulated patterns, while the most abundant peak was assigned to [M − Cl]+ species.
To understand the redox behavior of Ru(II) complexes, cyclic voltametric studies were performed in DMF (Fig. S38, ESI†). The complexes 1–4 exhibited reductive responses in the negative potential region corresponding to RuII/RuI redox couple. For example, complexes 1 and 3 showed quasi-reversible reductive responses with E1/2 at −1.84 V (ΔE = 130 mV) and −1.86 V (ΔE = 120 mV).29 Both these complexes also displayed irreversible responses in the negative potential region with Epc at −1.72 V and −1.70 V, assigned to ligand-based reductive events.30 Similarly, complexes 2 and 4 exhibited quasi-reversible reductive responses at −1.90 V (ΔE = 90 mV) and −1.88 V (ΔE = 90 mV). Additionally, all four complexes showed irreversible oxidative responses with Epa values of 1.39, 1.24, 1.42, and 1.29 V, respectively, corresponding to RuIII/RuII redox event.30
Crystal structures
The structure and geometry of complexes 2 and 3 were confirmed with the help of the single crystal diffraction studies (Fig. 1 and Tables S1 and S2, ESI†). The molecular structures of complexes 2 and 3 displayed a distorted octahedral geometry around the Ru(II) center. The ligands acted as the bidentate entities, occupying two coordination sites, while the remaining sites were ligated by one hydride ion, one CO, and two PPh3 groups.25,27 Each Ru(II) ion was coordinated to two PPh3 molecules, present trans to each other.24,27 The P1–Ru–P2 bond angles for complexes 2 and 3 were 172.37(4) and 169.016(19)°, respectively.26,27 The N1–Ru–H1 bond angles for both complexes were 165.9(9) and 174.2(6)°, signifying that the hydride ligand is located approximately at the trans position to that of the pyridyl ring. The CO ligand in both complexes was present at trans position to the amidic C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O group with bond angles of 172.64(17) and 173.49(7)°.25–27 The Ru–PPh3 bond lengths in both complexes were slightly longer than that of the Ru–H, Ru–CO, Ru–Oamide, and Ru–Npyridine bonds.27 The Ru–Oamide and Ru–Npyridine bond lengths for 2 and 3 were 2.153(3) and 2.164(11) Å; and 2.176(3) and 2.174(15) Å, respectively. The Ru–Oamide bonds were relatively shorter when compared to the Ru–Npyridine bonds in both complexes. Such a fact indicates that the binding of the amidic–O was stronger than that of pyridyl–N.25,27,31 In both complexes, one chloride ion was present outside the primary coordination sphere, which balanced the mono-cationic charge on the complexes.
O group with bond angles of 172.64(17) and 173.49(7)°.25–27 The Ru–PPh3 bond lengths in both complexes were slightly longer than that of the Ru–H, Ru–CO, Ru–Oamide, and Ru–Npyridine bonds.27 The Ru–Oamide and Ru–Npyridine bond lengths for 2 and 3 were 2.153(3) and 2.164(11) Å; and 2.176(3) and 2.174(15) Å, respectively. The Ru–Oamide bonds were relatively shorter when compared to the Ru–Npyridine bonds in both complexes. Such a fact indicates that the binding of the amidic–O was stronger than that of pyridyl–N.25,27,31 In both complexes, one chloride ion was present outside the primary coordination sphere, which balanced the mono-cationic charge on the complexes.
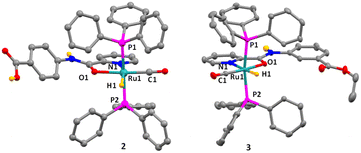 | ||
| Fig. 1 Crystal structures of Ru(II) complexes 2 and 3. Thermal ellipsoids are drawn at a 30% probability level, whereas selected hydrogen atoms have been shown for clarity. | ||
Dehydrogenation of amines
Nitrile- and imine-containing compounds are considered significant as they are key building blocks for the synthesis of numerous organic compounds, including carboxylic acids, amides, amines, and heterocyclic compounds in addition to various industrially important products such as pharmaceuticals, dyes, pesticides, and polymers.32 A noteworthy method for the preparation of nitriles is the oxidant-free and base-free dehydrogenation of amines catalyzed by transition metal complexes.14,19 The dehydrogenation of amines has been very little investigated and documented, whereas the conventional methods involve the excessive use of bases, high temperatures, and hydrogen acceptors.10,32 We have utilized the Ru–H complexes 1–4 for attempting DH of both primary and secondary amines to form nitriles and imines, respectively, generating hydrogen as the only by-product.Initially, the reaction conditions for the acceptorless DH were optimized by taking benzylamine as a model substrate in toluene while using 2.0 mol% of complex 2 as a representative catalyst. This reaction resulted in 95% conversion to benzonitrile (Table 1). To examine the role of a catalyst, the DH reaction was performed in the absence of complex 2, which did not result in any product formation (entry 1, Table 1). Two different Ru-precursors were employed to explore their catalytic efficiency; however, they only formed a negligible amount of the product (entries 2 and 3). The solvent screening was done using different solvents, such as dioxane, MeOH, DMF, CH3CN, DCM, THF, and toluene (entry 4). The best DH result was obtained in toluene (entry 5). As the DH of amines requires higher temperatures, toluene expectedly provided the best result. The remaining Ru–H complexes were also utilized for the DH of benzylamine, resulting in comparatively lower product yield (entries 6–8). Notably, the best DH results were obtained with the amide–acid-based complexes 2 and 4. We believe that the ability of the free –COOH group to form hydrogen bonds in these two complexes is a critical factor (vide infra).
| Entry | Catalyst | Solvent | Time (h) | Yieldb (%) |
|---|---|---|---|---|
| a Reaction conditions: benzylamine (1.0 mmol), catalyst (2.0 mol%), solvent (5 mL), temperature (100 °C), time (24 h). b Determined by gas chromatography. c Catalyst (1.0 mol%). d 85 °C. e 90 °C. | ||||
| 1 | — | Toluene | 24 | 0 |
| 2 | [Ru(CO)2Cl2]n | Toluene | 24 | 7 |
| 3 | RuH(CO)Cl(PPh3)3 | Toluene | 24 | 5 |
| 4 | 2 | Dioxane, MeOH, DMF, CH3CN, DCM, THF | 24 | 8, 0, 10, 4, 0, 0 |
| 5 | 2 | Toluene | 24 | 95 |
| 6 | 1 | Toluene | 24 | 90 |
| 7 | 3 | Toluene | 24 | 92 |
| 8 | 4 | Toluene | 24 | 93 |
| 9 | 5 | Toluene | 24 | 38 |
| 10c | 2 | Toluene | 24 | 72 |
| 11 | 2 | Toluene | 12 | 52 |
| 12d | 2 | Toluene | 24 | 65 |
| 13e | 2 | Toluene | 24 | 78 |
Therefore, to substantiate the effect of the appended group on the catalytic activity, we synthesized a ruthenium complex, complex 5, of the unsubstituted ligand. The complex 5 was characterized by 1H, 13C, and 31P NMR spectra (Fig. S39–S41†). Subsequently, the catalytic activity of complex 5 was evaluated by carrying out DH of benzylamine. However, this complex was quite inferior in its catalytic activity and only resulted in a 38% yield of benzonitrile (entry 9; Table 1). It is, therefore, evident that the presence of an appended group in a ligand scaffold plays a vital role in DH catalysis, presumably due to its ability to form hydrogen bonds (vide infra).33,34
When the catalyst load is decreased (entry 10), the yield also decreases, and therefore, 2.0 mol% of a catalyst is ideal for the acceptorless DH of amines. The other reaction parameters, such as temperature and time, were also optimized (entries 11–13). The best reaction conditions for the acceptorless DH of benzylamine were as follows: 2.0 mol% complex 2 using toluene as a solvent at 100 °C for 24 h (entry 5). The homogeneity of the catalysis was established by performing the mercury drop and the 1,10-phenanthroline poisoning tests.3 These studies did not result in any appreciable change in the product yield, thus confirming the homogeneity of DH.
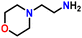
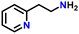
After optimizing the reaction conditions, the substrate scope was extended for the acceptorless DH of assorted primary amines (Table 2). Double DH of butylamine and octylamine provided excellent yield of butyronitrile and octanenitrile, respectively (entries 1 and 2). Substrate 4-(2-aminoethyl)morpholine afforded a high yield of 2-morpholino-acetonitrile (entry 3). Similarly, 2-phenoxyethylamine, 4-phenyl-butylamine, 2-(2-pyridyl)ethylamine, and 2-(4-nitrophenyl) ethan-1-amine were converted to their corresponding nitriles in high yields (entries 4–7).
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) a
a
| Entry | Substrate | Product | Yieldb (%) |
|---|---|---|---|
| a Reaction conditions: amine (1.0 mmol), catalyst (2.0 mol%), toluene (5 mL), temperature (100 °C), time (24 h). b Yields were determined by gas chromatography. The selected products were characterized by 1H NMR spectroscopy (Fig. S42–S45, ESI†). | |||
| 1 |

|
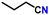
|
85 |
| 2 |
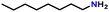
|

|
88 |
| 3 |
|

|
78 |
| 4 |

|
|
75 |
| 5 |

|

|
82 |
| 6 |
|

|
85 |
| 7 |

|

|
80 |
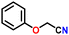
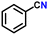
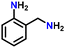
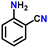
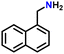
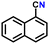
Subsequently, the substrate scope was extended to various aromatic primary amines (Table 3). As already discussed, double DH of benzylamine produced an excellent yield of benzonitrile (entry 1). The next aim was to study the effect of sterically and electronically distinct substituents on benzylamine.3,14,15 The electron-donating groups, such as –CH3, –NH2, and –OH on benzylamine, afforded the corresponding substituted benzonitriles in excellent yields (entries 2–5).14 The chemoselectivity was evaluated by carrying out the double DH of 2-aminobenzylamine and 4-aminobenzylamine, possessing both –NH2 and –CH2NH2 functional groups (entries 3 and 4).3 The exclusive formation of 2-aminobenzonitrile and 4-aminobenzonitrile confirmed the excellent chemoselectivity of the DH catalysis. Both these reactions adequately exemplify the advantages of opting for DH instead of conventional aerobic oxidation methods.10,12,35 The benzylamines substituted with electron-withdrawing groups, such as –F, –Cl, –Br, and –NO2, produced the respective benzonitriles in a slightly lower yield (entries 6–11).3,14 Notably, heterocyclic aromatic amines also produced high yields of the corresponding nitriles, while the effect of a nitrogen atom's position in the ring was not pronounced (entries 12–14). Lastly, bulkier 1-naphthylmethylamine was efficiently converted to 1-naphthonitrile (entry 15).
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) a
a
| Entry | Substrate | Product | Yieldb (%) |
|---|---|---|---|
| a Reaction conditions: amine (1.0 mmol), catalyst (2.0 mol%), toluene (5 mL), temperature (100 °C), time (24 h). b Yields were determined by gas chromatography. The selected products were characterized by 1H NMR spectroscopy (Fig. S46–S60, ESI†). | |||
| 1 |

|
|
94 |
| 2 |

|

|
95 |
| 3 |
|
|
96 |
| 4 |
|

|
97 |
| 5 |
|
|
95 |
| 6 |
|
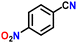
|
85 |
| 7 |

|

|
90 |
| 8 |
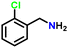
|
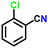
|
88 |
| 9 |

|
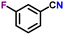
|
85 |
| 10 |

|

|
87 |
| 11 |
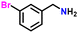
|

|
85 |
| 12 |
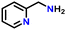
|
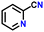
|
87 |
| 13 |

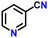
|
|
85 |
| 14 |

|
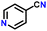
|
90 |
| 15 |
|
|
87 |
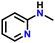
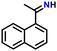
After obtaining noteworthy results for the double DH of assorted primary amines, we explored the DH of secondary amines and other nitrogen-containing heterocycles (Table 4).17,36 As per the literature, introducing a nitrogen atom in a cyclic system eases the DH process by lowering the catalytic reaction's endothermicity.10 Gratifyingly, DH of 2-(methylamino)pyridine and 4-(methylamino)pyridine afforded good yields of N-(pyridin-2-yl)methanimine and N-(pyridin-4-yl)methanimine, respectively (entries 1 and 2). Further, 1-(naphthalen-1-yl)ethylamine and N-(2-(pyridin-2-yl)ethyl)ethanamine also produced respective imines in good yields (entries 3 and 4). Furthermore, DH of indoline yielded indole as the sole product (entry 5). Importantly, both N-benzylaniline and dibenzylamine afforded the respective dehydrogenated products in excellent yields (entries 6 and 7). These results assert both the high efficiency and selectivity of the present ruthenium hydride catalysts.10,35 Although good selectivity and efficiency were achieved for the DH of secondary amines, the yield was comparatively lower than primary amines, most probably due to the presence of steric hindrance on the secondary amines.10,32
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) a
a
| Entry | Substrate | Product | Yieldb (%) |
|---|---|---|---|
| a Reaction conditions: amine (1.0 mmol), catalyst (2.0 mol%), toluene (5 mL), temperature (100 °C), time (24 h). b Yields were determined by gas chromatography. The selected products were characterized by 1H NMR spectroscopy (Fig. S61–S63, ESI†). | |||
| 1 |
|
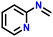
|
85 |
| 2 |
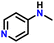
|
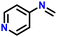
|
82 |
| 3 |
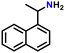
|
|
78 |
| 4 |
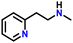
|
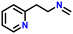
|
77 |
| 5 |
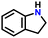
|
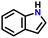
|
80 |
| 6 |
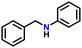
|
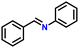
|
75 |
| 7 |

|

|
78 |
Binding and mechanistic studies
Binding and molecular docking studies were performed to get an insight into the mechanism of DH of amines. The binding ability of complex 2 towards benzylamine was investigated with the help of UV-Vis spectral titration in CH3OH (Fig. 2a).30,37,38 During the spectral titration, three isosbestic points were observed at 305, 320, and 420 nm, suggesting an interaction between complex 2 and benzylamine (Table S3 and Fig. S64†). The Job's plot suggested a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 stoichiometry between complex 2 and benzylamine (Fig. 2b).39 This fact was further supported by a linear regression fitting showing a 1
1 stoichiometry between complex 2 and benzylamine (Fig. 2b).39 This fact was further supported by a linear regression fitting showing a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 stoichiometry between complex 2 and benzylamine with a binding constant (Kb) of 1.23 × 103 M−1 (Fig. 2c).30,38
1 stoichiometry between complex 2 and benzylamine with a binding constant (Kb) of 1.23 × 103 M−1 (Fig. 2c).30,38
The molecular docking studies provided further evidence for the interaction of benzylamine with complex 2 (Fig. S65, Table S4, ESI†).40 The docked structure exhibited that benzylamine interacts with complex 2 through a hydrogen bond between its –NH2 group, and the –COOH group of the Ru–H complex. In addition, complex 2 showed dihydrogen bonding interaction with the –NH2 group of benzylamine through its metal-bound hydride group. Furthermore, π⋯π stacking was also observed between one of the phenyl rings of the PPh3 group of 2 to that of the arene ring of benzylamine.37,38 All these interactions aided in bringing a substrate (i.e., benzylamine) closer to the catalyst (i.e., complex 2) and assisted in DH catalysis.37
The proton NMR spectrum of a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 mixture of complex 2 and benzylamine was measured to further substantiate binding and molecular docking studies. The 1H NMR spectrum of this mixture exhibited notable shifts in the signals compared to the proton NMR spectrum of only complex 2 (Fig. S66, ESI†). The Ru–H signal was also found to shift from −10.35 to −10.31 ppm. Collectively, all studies attributed that a molecule of benzylamine was held in place by the involvement of various hydrogen bonding and π⋯π interactions, as illustrated by the molecular docking studies.
1 mixture of complex 2 and benzylamine was measured to further substantiate binding and molecular docking studies. The 1H NMR spectrum of this mixture exhibited notable shifts in the signals compared to the proton NMR spectrum of only complex 2 (Fig. S66, ESI†). The Ru–H signal was also found to shift from −10.35 to −10.31 ppm. Collectively, all studies attributed that a molecule of benzylamine was held in place by the involvement of various hydrogen bonding and π⋯π interactions, as illustrated by the molecular docking studies.
Based on all experiments and literature reports, a tentative mechanism is proposed for the acceptorless double DH of benzylamine as a representative substrate (Scheme S1, ESI†). In the first step, a molecule of benzylamine interacts with the catalyst through hydrogen bonding and π⋯π stacking to generate species A. It is worth mentioning that binding, molecular docking, and proton NMR spectral studies have provided evidence for this step (Scheme 4). This step was followed by the irreversible release of the first molecule of H2, leading to the generation of intermediate B. Intermediate B undergoes β-hydride elimination to form the Ru–imine intermediate C. The next step is the irreversible release of a second molecule of H2 to form the species D. Finally, a molecule of PPh3 replaces benzonitrile as the final product, thus completing the catalytic cycle. The formation of the imine intermediate was not detected in the reaction mixture, indicating that the release of the second molecule of H2 is fast. Importantly, the release of two equiv. of H2 was quantified using a gas burette apparatus, while its detection was ascertained with the help of gas chromatography. Such a fact supports the proposed mechanism.14
Conclusions
A new class of ruthenium hydride complexes supported with pyridine–amide-based ligands containing appended ester and/or acid groups was presented. All Ru(II) complexes were thoroughly characterized using various spectroscopic techniques, while crystal structures were obtained for a few representative cases. The catalytic behavior of these Ru–H complexes was explored for the oxidant-free and acceptorless dehydrogenation of both primary and secondary amines to nitriles and imines, respectively. These Ru–H complexes supported a wide substrate scope and operated under mild reaction conditions. These ruthenium catalysts tolerated both electron-withdrawing and electron-releasing groups present on a substrate. Various control experiments and mechanistic studies supported the mechanism for the dehydrogenation of amines catalyzed by the ruthenium hydride complexes. These results elegantly illustrate that strategic ligand design can overcome challenges associated with oxidant-free and acceptorless dehydrogenation of amines.Experimental
Materials
All reagents and chemicals were procured from commercial sources and used without further purification. The solvents were dried and/or purified using the standard literature procedures.41 Precursor [Ru(H)(Cl)(CO)(PPh3)3] was prepared according to a reported literature method.29Synthesis of ligands
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O (3
H2O (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, v/v) and treated with 2 equivalents of NaOH. The reaction mixture was stirred for 12 h at room temperature. The resulting solution was neutralized by 4 N HCl and the mixture was stirred for 1 h. After removal of THF under vacuum white precipitates of HL2 appeared that were filtered under suction. Yield: 0.82 g (87%). Anal. calcd for C13H10N2O3: C, 64.46%; H, 4.16%; N, 11.56%. Found: C, 64.92%; H, 4.77%; N, 12.03%. FTIR spectrum (diamond ATR, cm−1): 3325, 1685, 1606, 1525, 1431, 1284, 1109, 769, 678. 1H NMR spectrum (400 MHz, DMSO-d6, 25 °C) δ (ppm) 12.74 (s, 1H), 10.87 (s, 1H), 8.71 (d, J = 4.6 Hz, 1H), 8.14 (d, J = 7.8 Hz, 1H), 8.08–7.97 (m, 3H), 7.90 (d, J = 8.7 Hz, 2H), 7.65 (dd, J = 6.3, 4.8 Hz, 1H). 13C NMR spectrum (100 MHz, DMSO-d6, 25 °C) δ (ppm) 167.46, 163.48, 150.10, 149.03, 142.95, 138.76, 130.74, 127.72, 126.30, 123.15, 120.15. MS spectrum (ESI+, MeOH): m/z calcd for C15H14N2O3 [M + H+] 243.0691, found: 243.0767.
1, v/v) and treated with 2 equivalents of NaOH. The reaction mixture was stirred for 12 h at room temperature. The resulting solution was neutralized by 4 N HCl and the mixture was stirred for 1 h. After removal of THF under vacuum white precipitates of HL2 appeared that were filtered under suction. Yield: 0.82 g (87%). Anal. calcd for C13H10N2O3: C, 64.46%; H, 4.16%; N, 11.56%. Found: C, 64.92%; H, 4.77%; N, 12.03%. FTIR spectrum (diamond ATR, cm−1): 3325, 1685, 1606, 1525, 1431, 1284, 1109, 769, 678. 1H NMR spectrum (400 MHz, DMSO-d6, 25 °C) δ (ppm) 12.74 (s, 1H), 10.87 (s, 1H), 8.71 (d, J = 4.6 Hz, 1H), 8.14 (d, J = 7.8 Hz, 1H), 8.08–7.97 (m, 3H), 7.90 (d, J = 8.7 Hz, 2H), 7.65 (dd, J = 6.3, 4.8 Hz, 1H). 13C NMR spectrum (100 MHz, DMSO-d6, 25 °C) δ (ppm) 167.46, 163.48, 150.10, 149.03, 142.95, 138.76, 130.74, 127.72, 126.30, 123.15, 120.15. MS spectrum (ESI+, MeOH): m/z calcd for C15H14N2O3 [M + H+] 243.0691, found: 243.0767.
Synthesis of Ru(II) complexes
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) mixture was treated with ligand HL1 (0.0284 g, 0.10 mmol) and refluxed at 65 °C for 6 h which resulted in a yellow-colored solution. After completion of the reaction, volume was reduced to one-third using a rotary evaporator, and diethyl ether was added until the precipitation was complete. The product was then collected by filtration and dried under vacuum. Single crystals suitable for X-ray analysis were obtained by the vapor diffusion of diethyl ether into a saturated methanol–chloroform solution of the complex. Yield: 0.088 g (90%). Anal. calcd for C52H45N2O4P2ClRu: C 65.03, H 4.72, N 2.92; found: C 65.55, H 5.12, N 3.33. FTIR spectrum (diamond, cm−1): 1957, 1923, 1706, 1597, 1431, 1271, 1055, 696, 509. UV/Vis spectrum (MeOH, λmax, nm): 300, 435. 1H NMR spectrum (400 MHz, CDCl3, 25 °C) δ (ppm) 8.20 (d, J = 5.3 Hz, 1H), 7.91 (d, J = 7.8 Hz, 1H), 7.44 (t, J = 7.6 Hz, 2H), 7.36–7.30 (m, 11H), 7.19 (dd, J = 11.9, 4.6 Hz, 8H), 7.07 (t, J = 7.6 Hz, 12H), 6.46–6.36 (m, 1H), 6.20 (d, J = 8.7 Hz, 2H), 4.27 (q, J = 7.2 Hz, 2H), 1.33 (t, J = 7.1 Hz, 3H). 13C NMR spectrum (100 MHz, CDCl3, 25 °C) δ (ppm) 211.37, 180.70, 167.48, 151.43, 146.69, 137.12, 135.81, 133.27, 132.10, 131.87, 130.2, 128.47, 127.99, 126.93, 126.19, 123.81. 31P NMR (160 MHz, CDCl3, 25 °C) δ (ppm) 46.21. MS spectrum (ESI+, MeOH): m/z calcd for C52H45N2O4P2Ru [M − Cl]+ 925.1898, found: 925.1894.
1) mixture was treated with ligand HL1 (0.0284 g, 0.10 mmol) and refluxed at 65 °C for 6 h which resulted in a yellow-colored solution. After completion of the reaction, volume was reduced to one-third using a rotary evaporator, and diethyl ether was added until the precipitation was complete. The product was then collected by filtration and dried under vacuum. Single crystals suitable for X-ray analysis were obtained by the vapor diffusion of diethyl ether into a saturated methanol–chloroform solution of the complex. Yield: 0.088 g (90%). Anal. calcd for C52H45N2O4P2ClRu: C 65.03, H 4.72, N 2.92; found: C 65.55, H 5.12, N 3.33. FTIR spectrum (diamond, cm−1): 1957, 1923, 1706, 1597, 1431, 1271, 1055, 696, 509. UV/Vis spectrum (MeOH, λmax, nm): 300, 435. 1H NMR spectrum (400 MHz, CDCl3, 25 °C) δ (ppm) 8.20 (d, J = 5.3 Hz, 1H), 7.91 (d, J = 7.8 Hz, 1H), 7.44 (t, J = 7.6 Hz, 2H), 7.36–7.30 (m, 11H), 7.19 (dd, J = 11.9, 4.6 Hz, 8H), 7.07 (t, J = 7.6 Hz, 12H), 6.46–6.36 (m, 1H), 6.20 (d, J = 8.7 Hz, 2H), 4.27 (q, J = 7.2 Hz, 2H), 1.33 (t, J = 7.1 Hz, 3H). 13C NMR spectrum (100 MHz, CDCl3, 25 °C) δ (ppm) 211.37, 180.70, 167.48, 151.43, 146.69, 137.12, 135.81, 133.27, 132.10, 131.87, 130.2, 128.47, 127.99, 126.93, 126.19, 123.81. 31P NMR (160 MHz, CDCl3, 25 °C) δ (ppm) 46.21. MS spectrum (ESI+, MeOH): m/z calcd for C52H45N2O4P2Ru [M − Cl]+ 925.1898, found: 925.1894.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) mixture with ligand HL3 (0.0284 g, 0.10 mmol). Yield: 0.086 g (88%). Anal. calcd for C52H45N2O4P2ClRu: C 65.03, H 4.72, N 2.92; found: C 65.43, H 4.92, N 3.12. FTIR spectrum (diamond, cm−1): 3365, 1971, 1930, 1709, 1592, 1438, 1263, 1093, 752, 690. UV/Vis spectrum (MeOH, λmax, nm): 305, 416. 1H NMR spectrum (400 MHz, CDCl3, 25 °C) δ (ppm) 12.49 (s, 1H), 9.63 (d, J = 8.0 Hz, 1H), 8.43 (s, 1H), 7.85 (d, J = 7.8 Hz, 2H), 7.76 (d, J = 5.2 Hz, 1H), 7.71 (t, J = 7.9 Hz, 1H), 7.47 (dd, J = 12.3, 5.4 Hz, 12H), 7.29 (dd, J = 11.0, 7.5 Hz, 5H), 7.20 (t, J = 7.3 Hz, 12H), 6.83–6.77 (m, 1H), 4.44 (q, J = 7.1 Hz, 2H), 1.43 (t, J = 7.1 Hz, 3H), −10.20 (t, J = 18.7 Hz, 1H). 13C NMR spectrum (100 MHz, CDCl3, 25 °C) δ (ppm): 167.90, 151.48, 137.15, 136.36, 133.31, 132.07, 131.74, 131.54, 128.64, 124.11, 14.59. 31P NMR (160 MHz, CDCl3, 25 °C) δ (ppm) 46.17. MS spectrum (ESI+, MeOH): m/z calcd for C52H45N2O4P2Ru [M − Cl]+ 925.1898, found: 925.1942.
1) mixture with ligand HL3 (0.0284 g, 0.10 mmol). Yield: 0.086 g (88%). Anal. calcd for C52H45N2O4P2ClRu: C 65.03, H 4.72, N 2.92; found: C 65.43, H 4.92, N 3.12. FTIR spectrum (diamond, cm−1): 3365, 1971, 1930, 1709, 1592, 1438, 1263, 1093, 752, 690. UV/Vis spectrum (MeOH, λmax, nm): 305, 416. 1H NMR spectrum (400 MHz, CDCl3, 25 °C) δ (ppm) 12.49 (s, 1H), 9.63 (d, J = 8.0 Hz, 1H), 8.43 (s, 1H), 7.85 (d, J = 7.8 Hz, 2H), 7.76 (d, J = 5.2 Hz, 1H), 7.71 (t, J = 7.9 Hz, 1H), 7.47 (dd, J = 12.3, 5.4 Hz, 12H), 7.29 (dd, J = 11.0, 7.5 Hz, 5H), 7.20 (t, J = 7.3 Hz, 12H), 6.83–6.77 (m, 1H), 4.44 (q, J = 7.1 Hz, 2H), 1.43 (t, J = 7.1 Hz, 3H), −10.20 (t, J = 18.7 Hz, 1H). 13C NMR spectrum (100 MHz, CDCl3, 25 °C) δ (ppm): 167.90, 151.48, 137.15, 136.36, 133.31, 132.07, 131.74, 131.54, 128.64, 124.11, 14.59. 31P NMR (160 MHz, CDCl3, 25 °C) δ (ppm) 46.17. MS spectrum (ESI+, MeOH): m/z calcd for C52H45N2O4P2Ru [M − Cl]+ 925.1898, found: 925.1942.
Typical procedure for the dehydrogenation of amines
An amine substrate (1.00 mmol) was added to a 10 mL round bottom flask containing a solution of complex 2 (2.0 mol%) in toluene (5 mL). The round bottom flask was capped with a septum and then heated to 100 °C under N2 atmosphere. The progress of the reaction was monitored by thin-layer chromatography (TLC). After 24 h, the reaction mixture was cooled, and the solvent was evaporated under reduced pressure. The residue was dissolved in ethyl acetate, and yield was determined by gas chromatography. The products were purified by the silica gel chromatography using hexanes![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ethyl acetate (3
ethyl acetate (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) mixture which afforded the corresponding nitrile as the isolated product. The identity and the purity of the nitrile products were confirmed by a combination of GC and NMR spectral techniques. As control and optimization experiments used benzylamine as a reactant that produces benzonitrile as a product on DH, so, the calibration plot was studied for this reactant/product combination (Fig. S67, ESI†).
1) mixture which afforded the corresponding nitrile as the isolated product. The identity and the purity of the nitrile products were confirmed by a combination of GC and NMR spectral techniques. As control and optimization experiments used benzylamine as a reactant that produces benzonitrile as a product on DH, so, the calibration plot was studied for this reactant/product combination (Fig. S67, ESI†).
Estimation of evolved H2 during the dehydrogenation of benzylamine
Benzylamine (1.00 mmol) was added to a 10 mL round bottom flask containing a solution of complex 2 (2.0 mol%) in toluene (5 mL) and heated to 100 °C. The round bottom flask was connected to a gas burette apparatus through a cold trap to eliminate the toluene vapors. The reaction was allowed to continue till the evolution of gas had stopped. The volume of the water displaced by the H2 gas was recorded. This experiment was repeated three times to get reasonable concordant readings, and the amount of H2 gas evolved was calculated using the formula given below:where nH2 is the number of moles of H2 gas produced; P(atm) is atmospheric pressure = 760 Torr; P(water) is vapour pressure of water at 298 K = 23.8 Torr; V is the volume of water displaced = 45.7 mL; R = 62.3635 L Torr K−1 mol−1; and T = 298 K.
1H NMR spectral data for some of the representative products
Job's plot
Stock solutions of an identical concentration (1 mM) of complex 2 and benzylamine (substrate) were prepared in MeOH. The absorbance in each case, with different complex-substrate ratios but equal volume was recorded. The Job's plot was drawn by plotting the intensity vs. mole fraction of benzylamine.Physical measurements
Elemental analysis data were obtained by the Elementar Analysen Systeme GmbH Vario EL-III instrument. The 1H and 13C NMR spectra were recorded with a JEOL 400 MHz instrument. The FTIR spectra were recorded with an Agilent Cary-630 spectrometer having a diamond ATR. The conductivity measurements were done with an Eutech Instruments conductivity bridge (model: Con-510). The absorption spectra were recorded with a PerkinElmer Lambda-950 spectrophotometer. ESI+ mass spectra were measured with an Agilent Q-TOF LC-MS mass spectrometer. The cyclic voltametric experiments were conducted on the CH Instruments electrochemical analyzer (model 1120 A). The cell contained a glassy–carbon working electrode, a platinum wire auxiliary electrode, and an Ag/Ag+ reference electrode.42,43 The stock solutions were ca. 1 mM in Ru(II) complex and 0.1 M in supporting electrolyte, tetrabutylammonium perchlorate.X-ray crystallography
The X-ray intensity data for complexes 2 and 3 were collected at 273 K with an Oxford XCalibur CCD diffractometer equipped with a graphite monochromatic Mo-Kα radiation (λ = 0.71073 Å).44 Data reduction was performed with the CrysAllisPro program (Oxford Diffraction ver. 171.34.40).23 The structures were solved by the direct methods using the SHELXL program and refined on F2 using all data by full-matrix least-squares procedures with SHELXL-2018/3.45 The hydrogen atoms were placed at the calculated positions and included in the last cycles of the refinement.46 For complex 3, restrain command DELU was applied to obtain reasonable thermal parameters. For both complexes 2 and 3, some disordered electron density could not be resolved, and therefore, the solvent masking procedure (PLATON SQUEEZE) was used.47 The electron counts of 36 (two methanol) and 67 (three methanol and one water) were recovered for complexes 2 and 3, respectively. Such solvent molecules have been included in both the empirical formula and formula weight of complexes. All calculations were done using the WinGX crystallographic software package.45,47 The crystallographic data collection and structure solution parameters are summarized in Table S1 (ESI).†Molecular docking studies
The molecular docking studies were performed by using the CIF file of the structurally characterized complex 2 and an energy-minimized chemical drawing of benzylamine using Hex 8.0.0 software.48 The binding affinity of the metal complex was calculated against the receptor molecule of benzylamine. The binding interactions (docking images) were visualized in Mercury 4.2.0 (Build 257471).Data availability
The data related to this manuscript are available in the manuscript, as well as the ESI.†Conflicts of interest
There are no conflicts to declare.Acknowledgements
RG gratefully acknowledges financial support from the Institution of Eminence, University of Delhi. SY thanks CSIR, New Delhi for her SRF. The authors thank the USIC of this university for the instrumental facilities. Reviewers’ comments were very helpful in the revision stage.References
- F. F. Fleming, L. Yao, P. C. Ravikumar, L. Funk and B. C. Shook, J. Med. Chem., 2010, 53, 7902–7917 CrossRef CAS PubMed.
- D. Srimani, M. Feller, Y. Ben-David and D. Milstein, Chem. Commun., 2012, 48, 11853–11855 RSC.
- K.-N. T. Tseng, A. M. Rizzi and N. K. Szymczak, J. Am. Chem. Soc., 2013, 135, 16352–16355 CrossRef CAS PubMed.
- H. Tian, C.-Y. Ding, R.-Z. Liao, M. Li and C. Tang, J. Am. Chem. Soc., 2024, 146, 11801–11810 CrossRef CAS PubMed.
- K. Yamaguchi, H. Fujiwara, Y. Ogasawara, M. Kotani and N. Mizuno, Angew. Chem., Int. Ed., 2007, 46, 3922–3925 CrossRef CAS PubMed.
- K. Ishihara, Y. Furuya and H. Yamamoto, Angew. Chem., 2002, 114, 3109–3112 CrossRef.
- S. S. Moirangthem, D. Ahanthem, S. D. Khongbantabam and W. S. Laitonjam, ACS Omega, 2022, 7, 31348–31351 CrossRef CAS PubMed.
- A. Falk, A.-L. Göderz and H.-G. Schmalz, Angew. Chem., Int. Ed., 2013, 52, 1576–1580 CrossRef CAS PubMed.
- T. Achard, J. Egly, M. Sigrist, A. Maisse-François and S. Bellemin-Laponnaz, Chem. – Eur. J., 2019, 25, 13271–13274 CrossRef CAS PubMed.
- M. Kannan, P. Barteja, P. Devi and S. Muthaiah, J. Catal., 2020, 386, 1–11 CrossRef CAS.
- W. P. Griffith, B. Reddy, A. G. F. Shoair, M. Suriaatmaja, A. J. P. White and D. J. Williams, J. Chem. Soc., Dalton Trans., 1998, 2819–2826 RSC.
- S. Aiki, A. Taketoshi, J. Kuwabara, T. Koizumi and T. Kanbara, J. Organomet. Chem., 2011, 696, 1301–1304 CrossRef CAS.
- J. Kim and S. S. Stahl, ACS Catal., 2013, 3, 1652–1656 CrossRef CAS PubMed.
- I. Dutta, S. Yadav, A. Sarbajna, S. De, M. Hölscher, W. Leitner and J. K. Bera, J. Am. Chem. Soc., 2018, 140, 8662–8666 CrossRef CAS PubMed.
- M. Olivares, P. Knörr and M. Albrecht, Dalton Trans., 2020, 49, 1981–1991 RSC.
- S. Hazra, P. Pilania, M. Deb, A. K. Kushawaha and A. J. Elias, Chem. – Eur. J., 2018, 24, 15766–15771 CrossRef CAS PubMed.
- S. Budweg, K. Junge and M. Beller, Catal. Sci. Technol., 2020, 10, 3825–3842 RSC.
- L. V. A. Hale, T. Malakar, K.-N. T. Tseng, P. M. Zimmerman, A. Paul and N. K. Szymczak, ACS Catal., 2016, 6, 4799–4813 CrossRef CAS.
- W. H. Bernskoetter and M. Brookhart, Organometallics, 2008, 27, 2036–2045 CrossRef CAS.
- X. Nie, Y. Zheng, L. Ji, H. Fu, H. Chen and R. Li, J. Catal., 2020, 391, 378–385 CrossRef CAS.
- J. P. Shanahan, D. M. Mullis, M. Zeller and N. K. Szymczak, J. Am. Chem. Soc., 2020, 142, 8809–8817 CrossRef PubMed.
- R. Mitsuhashi, Y. Imai, T. Suzuki and Y. Hayashi, Molecules, 2022, 27, 2098 CrossRef CAS PubMed.
- K. Nakamoto, in Handbook of Vibrational Spectroscopy, John Wiley & Sons, Ltd, 2006 Search PubMed.
- Z. Almodares, S. J. Lucas, B. D. Crossley, A. M. Basri, C. M. Pask, A. J. Hebden, R. M. Phillips and P. C. McGowan, Inorg. Chem., 2014, 53, 727–736 CrossRef CAS PubMed.
- S. Panda, A. Singha Hazari, M. Gogia and G. K. Lahiri, Inorg. Chem., 2020, 59, 1355–1363 CrossRef CAS PubMed.
- S. Nag, R. J. Butcher and S. Bhattacharya, Eur. J. Inorg. Chem., 2007, 2007, 1251–1260 CrossRef.
- M. Maji and S. Kundu, Dalton Trans., 2019, 48, 17479–17487 RSC.
- B. Procacci, S. B. Duckett, M. W. George, M. W. D. Hanson-Heine, R. Horvath, R. N. Perutz, X.-Z. Sun, K. Q. Vuong and J. A. Welch, Organometallics, 2018, 37, 855–868 CrossRef CAS.
- P. Vijayan, S. Yadav, S. Yadav and R. Gupta, Inorg. Chim. Acta, 2020, 502, 119285 CrossRef CAS.
- S. Yadav, P. Vijayan, S. Yadav and R. Gupta, Dalton Trans., 2021, 50, 3269–3279 RSC.
- S. M. Ashraf, W. Kandioller, M.-G. Mendoza-Ferri, A. A. Nazarov, C. G. Hartinger and B. K. Keppler, Chem. Biodiversity, 2008, 5, 2060–2066 CrossRef CAS PubMed.
- C. Gunanathan and D. Milstein, Chem. Rev., 2014, 114, 12024–12087 CrossRef CAS PubMed.
- B. Das, A. Rahaman, A. Shatskiy, O. Verho, M. D. Kärkäs and B. Åkermark, Acc. Chem. Res., 2021, 54, 3326–3337 CrossRef CAS PubMed.
- X.-H. Yang, K. Wang, S.-F. Zhu, J.-H. Xie and Q.-L. Zhou, J. Am. Chem. Soc., 2014, 136, 17426–17429 CrossRef CAS PubMed.
- B. Karimi, O. Yari, M. Khorasani, H. Vali and F. Mansouri, ChemCatChem, 2018, 10, 1783–1787 CrossRef CAS.
- D. Ventura-Espinosa, A. Marzá-Beltrán and J. A. Mata, Chem. – Eur. J., 2016, 22, 17758–17766 CrossRef CAS PubMed.
- S. Pachisia, R. Kishan, S. Yadav and R. Gupta, Inorg. Chem., 2021, 60, 2009–2022 CrossRef CAS PubMed.
- D. Prabha, S. Pachisia and R. Gupta, Inorg. Chem. Front., 2021, 8, 1599–1609 RSC.
- S. Yadav and R. Gupta, Inorg. Chem., 2022, 61, 15463–15474 CrossRef CAS PubMed.
- A. W. Ghoorah, M.-D. Devignes, M. Smaïl-Tabbone and D. W. Ritchie, Proteins: Struct., Funct., Bioinf., 2013, 81, 2150–2158 CrossRef CAS PubMed.
- D. D. Perrin, W. L. F. Armarego and D. R. Perrin, Purification of Laboratory Chemicals, Pergamon Press, Oxford, UK, 1980 Search PubMed.
- A. Brajter-Toth, J. Am. Chem. Soc., 1996, 118, 10339–10340 CrossRef CAS.
- N. G. Connelly and W. E. Geiger, Chem. Rev., 1996, 96, 877–910 CrossRef CAS PubMed.
- CrysAlisPro, v. 1.171.33.49b, Oxford Diffraction Ltd., Abingdon, 2009 Search PubMed.
- G. M. Sheldrick, Acta Crystallogr., Sect. A: Found. Crystallogr., 2008, 64, 112–122 CrossRef CAS PubMed.
- L. J. Farrugia, J. Appl. Crystallogr., 2012, 45, 849–854 CrossRef CAS.
- A. L. Spek, PLATON, A Multipurpose Crystallographic Tool, Utrecht University, The Netherlands, 2002 Search PubMed.
- D. W. Ritchie, Hex, Inria, 2012. https://hex.loria.fr/ Search PubMed.
Footnote |
| † Electronic supplementary information (ESI) available. CCDC 2402741 and 2402742. For ESI and crystallographic data in CIF or other electronic format see DOI: https://doi.org/10.1039/d4dt03201b |
| This journal is © The Royal Society of Chemistry 2025 |