Versatile functionalization of de-fluorinated FMOF-1 towards enhanced carbon capture and separation: a predictive molecular simulation study†
Rashida
Yasmeen
 a,
Sheikh M. S.
Islam
a,
Sheikh M. S.
Islam
 b,
Jincheng
Du
b,
Jincheng
Du
 *a and
Mohammad A.
Omary
*a and
Mohammad A.
Omary
 *b
*b
aDepartment of Materials Science & Engineering, University of North Texas, 1155 Union Circle, Denton, Texas-76203, USA. E-mail: Jincheng.Du@unt.edu
bDepartment of Chemistry, University of North Texas, 1155 Union Circle, Denton, Texas-76203, USA. E-mail: Omary@unt.edu
First published on 7th February 2025
Abstract
Fluorous metal–organic frameworks, FMOFs, represent a superhydrophobic class of MOFs containing –CF3 or –F groups in their pores. The primary objective of this research is to computationally design functionalized FMOF-1-X with X = –OCH3, –CN, –OH, –COOH, and –NH2 instead of –CF3 and analyze their CO2 adsorption and separation characteristics. Grand Canonical Monte Carlo (GCMC) simulations have been used to study the adsorption properties of CO2, CH4, and N2 in all structures. Henry's constant (KH) and isosteric heat of adsorption at infinite dilution (Qst0) estimated from molecular Monte Carlo simulations plus the binding energy (BE) from Möller–Plesset second-order perturbation theory (MP2) quantum-mechanical simulations characterize adsorbate–adsorbent interaction strengths. Such simulations predict a systematic enhancement of all KH, Qst0, and BE values in X-functionalized MOFs vs. the parent FMOF-1. Among such functional MOFs, the X = –COOH structure is predicted to exhibit the largest CO2 uptake in the low-pressure region due to the strongest CO2/–COOH interaction strength, as supported by the largest KH value (1.02 × 10−4 mol kg−1 Pa−1). In contrast, at high pressures (30 bar), the X = –OH structure is predicted to exhibit the highest CO2 uptake. Indeed, replacing the –CF3 groups in FMOF-1 by any aforementioned X group is expected to afford higher CO2 uptake in the GCMC-simulated adsorption isotherms compared to the parent material. The selective adsorption of CO2 over CH4 and N2 was determined using the ideal adsorbed solution theory (IAST) method at 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50 and 15
50 and 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 85 CO2/CH4 and CO2/N2 binary mixtures, respectively. The X = –COOH structure amounts to the largest selectivity (59.6 for CO2/CH4 and 128.7 for CO2/N2), i.e., nearly 40× and 43× higher vs. FMOF-1 (1.5 and 3 for CO2/CH4 and CO2/N2, respectively) at 298 K and 0.1 bar. The study herein of functionalized MOFs for CO2 separation, natural gas purification, landfill gas separation, and/or CO2 flue gas capture suggest that X = –OH, –COOH, and –NH2 are promising to enhance the adsorption capacity and selectivity.
85 CO2/CH4 and CO2/N2 binary mixtures, respectively. The X = –COOH structure amounts to the largest selectivity (59.6 for CO2/CH4 and 128.7 for CO2/N2), i.e., nearly 40× and 43× higher vs. FMOF-1 (1.5 and 3 for CO2/CH4 and CO2/N2, respectively) at 298 K and 0.1 bar. The study herein of functionalized MOFs for CO2 separation, natural gas purification, landfill gas separation, and/or CO2 flue gas capture suggest that X = –OH, –COOH, and –NH2 are promising to enhance the adsorption capacity and selectivity.
1. Introduction
Traditional porous materials such as silica, activated carbons, and zeolites have been studied as potential adsorbents for carbon capture and storage (CCS) for a while.1,2 However, these materials have not shown superior quality and bear the drawbacks of either low adsorption selectivity or smaller uptake capacity of CO2.3,4 In order to overcome the limitations of traditional porous solids, researchers have been exploring metal–organic frameworks (MOFs) extensively in recent times, for the enhanced adsorption and separation of CO2.5–8 With higher porosities and tunable chemical properties, MOFs have shown remarkable potential for adsorptive separation to date.9–14 To achieve improved adsorption and selectivity of CO2, the properties of MOFs could be tuned/have been tuned in different ways such as by controlling pore sizes, incorporating alkali–metal cations, introducing open metal sites, etc.15–17Ligand functionalization18–22 is a potential approach for the improvement of adsorbate–adsorbent interaction that could result in enhanced CO2 adsorption in MOFs. Addition of different functional groups to a linker can significantly affect the CO2 adsorption and separation capacities of the structures.23,24 Arstad et al.25 and An et al.26 found that amine groups can improve CO2 uptake in MOFs through the formation of larger binding sites compared to the parent MOFs. Couck et al.27 incorporated an amino group into MIL-53 (Al) and showed enhanced CO2/CH4 selectivity compared to the original MOF, MIL-53 (Al). Zheng et al.28 reported higher isosteric heats of adsorption (Qst) and improved CO2 uptake in acylamide-decorated MOFs than that of the unfunctionalized structures. Improved Qst was observed for amino functionalized MIL-53 (Al) as well with a value of ∼38.4 kJ mol−1, whereas the Qst value of the parent MIL-53 (Al) was <20 kJ mol−1.27 Torrisi et al.29,30 investigated the impact of functionalization on CO2 adsorption by incorporating –OH, –COOH, –NH2, and –CH3 groups into MIL-53 (lp). As per the work, significantly higher CO2/CH4 selectivity was predicted for –COOH and –OH functionalized MIL-53 (lp) compared to the parent MIL-53 (lp). Gu et al.31 used density functional theory (DFT) to study the effects of functional groups for the improvement of CO2 uptake by introducing –SO3H, –COOH, –NH2, –OH, –CN, –CH3, and –F groups into MOF-177. Additionally, some researchers have inspected the interaction strength of CO2 with different functionalized linkers using ab initio methods.32–34 Molecular simulations have been used widely by researchers to obtain useful information about the adsorption properties of MOFs, even prior to their synthesis.35–38 Inspired by the above work, we attempted to design various functionalized FMOFs by replacing –CF3 groups in the original structure by X = –OCH3, –CN, –OH–, –COOH, and –NH2 functional groups. Then we performed a systematic investigation of the CO2, CH4, and N2 adsorption behavior of all the MOFs. Henry's constant (KH) and isosteric heat of adsorption at infinite dilution (Qst0) were obtained by Monte Carlo (MC) simulation, whereas binding energy (BE) was calculated using Möller–Plesset second-order perturbation theory (MP2). The obtained KH, Qst0, and BE values were used to understand the adsorbate–adsorbent interaction in MOFs. The CO2 adsorption sites of MOFs were studied by analyzing the radial distribution functions (RDFs) of CO2 at room temperature. The CO2/CH4 and CO2/N2 adsorption selectivities were determined by Grand Canonical Monte Carlo (GCMC) simulations in all the X-functionalized MOF materials. Comprehensively, we inspected the effect of the X-functional groups on the uptake capacities and selective adsorption of CO2 over CH4 and N2.
2. Methodologies
2.1 Adsorbent model
FMOF-1 was considered as the reference adsorbent. This is a fluorous metal–organic framework, synthesized by Yang et al.39 The cell parameters and the coordinates of the framework atoms were taken from the experimental crystallographic data.40 FMOF-1 has a tetragonal crystal structure with the space group I![[4 with combining macron]](https://www.rsc.org/images/entities/char_0034_0304.gif) 2d and the lattice parameters are a = b = 14.0733 Å, c = 37.675 Å, and α = β = γ = 90°. No experimental crystallographic data are available for FMOF-1-X (X = –OCH3, –CN, –OH, –COOH, and –NH2). Materials Studio was used to construct the hypothetical structures.41 This program can efficiently generate feasible crystal structures and researchers are using this software to characterize MOF structures prior to their synthesis.42–44 Considering FMOF-1 as the parent structure, FMOF-1-X were constructed by replacing the –CF3 groups with the X-functional groups followed by structure optimization using the Forcite module, implemented in the Materials Studio program. The Universal Force Field45 was considered to describe the interactions between the framework atoms during the structure optimization. Final structures (FMOF-1-X) were obtained after completing the two step optimization procedure. Energy and density optimization data (Fig. S1–S5; see the ESI†) ensure reliable hypothetical crystal structures. Rather than having different lattice parameters, all the FMOF-1-X exhibit the same tetragonal crystal structure as the parent FMOF-1, with the space group I
2d and the lattice parameters are a = b = 14.0733 Å, c = 37.675 Å, and α = β = γ = 90°. No experimental crystallographic data are available for FMOF-1-X (X = –OCH3, –CN, –OH, –COOH, and –NH2). Materials Studio was used to construct the hypothetical structures.41 This program can efficiently generate feasible crystal structures and researchers are using this software to characterize MOF structures prior to their synthesis.42–44 Considering FMOF-1 as the parent structure, FMOF-1-X were constructed by replacing the –CF3 groups with the X-functional groups followed by structure optimization using the Forcite module, implemented in the Materials Studio program. The Universal Force Field45 was considered to describe the interactions between the framework atoms during the structure optimization. Final structures (FMOF-1-X) were obtained after completing the two step optimization procedure. Energy and density optimization data (Fig. S1–S5; see the ESI†) ensure reliable hypothetical crystal structures. Rather than having different lattice parameters, all the FMOF-1-X exhibit the same tetragonal crystal structure as the parent FMOF-1, with the space group I![[4 with combining macron]](https://www.rsc.org/images/entities/char_0034_0304.gif) 2d. The optimized structures of –OCH3, –CN, –OH, –COOH, and –NH2 functionalized MOFs along with the parent FMOF-1 are displayed in Fig. S6–S8.† The structural properties of the MOFs such as density, void fraction and pore volume were computed using Zeo++ software,46 whereas the RASPA2 program47 was used to determine the accessible surface area considering N2 as the probe molecule. The geometrical properties of all the MOFs are listed in Table 2.
2d. The optimized structures of –OCH3, –CN, –OH, –COOH, and –NH2 functionalized MOFs along with the parent FMOF-1 are displayed in Fig. S6–S8.† The structural properties of the MOFs such as density, void fraction and pore volume were computed using Zeo++ software,46 whereas the RASPA2 program47 was used to determine the accessible surface area considering N2 as the probe molecule. The geometrical properties of all the MOFs are listed in Table 2.
2.2 Interaction potential
In order to describe the non-bonded interactions during CO2, CH4, and N2 adsorption in MOFs, we used the interaction potential as a combination of truncated Lennard-Jones (LJ) and Coulomb potentials according to eqn (1): | (1) |
In our work, the atomic partial charges for the –CN, –OH, –COOH, and –NH2 functional groups were adopted from the works of Torrisi et al. and Gu et al.30,31 while the partial charges of –OCH3 were estimated using density functional theory (DFT) implemented in the DMol3 module of Materials Studio.41 During the DFT calculation, we used the PW91 functional along with the double-ξ numerical polarization (DNP) basis set. The partial charges of the X-functional groups were derived from the fitting of the energy surface potentials (ESPs). ESP charges are being widely used by researchers to study the gas adsorption isotherms for MOF structures,29–31 as this method generally gives a better description of the electrostatic potential around the different atomic species in MOFs.30 The partial charges for the rest of the framework atoms were taken from the work of Moghadam et al.40 We scaled the charges a little in order to make the framework charge neutral. All the framework charges are listed in Tables S2–S7.† The long-range electrostatic interactions were calculated via the Ewald summation method50 with the same cutoff distance of 12.8 Å considered for all the Lennard-Jones interactions.
In this study, CO2 and N2 molecules were modeled as the linear three-site rigid model,51 whereas CH4 was mimicked as a united spherical single site model.52 The C–O and N–N bond lengths were 1.16 Å and 1.10 Å, respectively, in CO2 and N2. The charges on C and O atoms were +0.70e and −0.35e in CO2. On the other hand, a charge of −0.482e and +0.964e was placed on N and on the center of mass, respectively, during the modeling of N2. These partial charges on the LJ sites were used to describe the internal quadrupole moment of CO2 and N2. We used the transferable potentials for the phase equilibria (TraPPE) force field to model the adsorbates (CO2, CH4, and N2). The potential parameters and the atomic charges of the adsorbates are listed in Table S8.† A 2 × 2 × 1 supercell was used for all MOF structures. The framework atoms were kept rigid during GCMC simulations.
2.3 Simulation details
Simulation studies have disclosed that Grand Canonical Monte Carlo (GCMC) simulations can accurately compute the gas loading in porous materials such as MOFs.53 We used GCMC to calculate the adsorption isotherms of pure CO2, CH4, N2 components, as well as binary mixtures of CO2/CH4 and CO2/N2 as a function of pressure. The calculations were carried out at 273 and 298 K up to a pressure of 30 bar in all the structures. The number of gas molecules in the adsorbed phase was allowed to fluctuate whereas the chemical potential (μ), volume (v), and temperature (T) were kept constant during the GCMC simulations. Random insertion, deletion, rotation and translation with equal probabilities were used in each MC run. The GCMC calculation at each pressure point consisted of 2.0 × 105 cycles. The first half was used for equilibration and the subsequent half was used to estimate the desired adsorption properties. Excess adsorption isotherms were simulated by considering the void fraction of both structures (details can be found in the ESI†).Henry's constants (KH) of the adsorbates were evaluated using the Widom particle insertion method.54 The enthalpy of adsorption, ΔH, was determined by the statistical average of the adsorbate binding energies at different available adsorbent sites according to eqn (2):
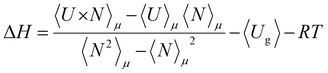 | (2) |
The isosteric heat of adsorption at infinite dilution, Qst0, was estimated using a single molecule of the adsorbates in the Canonical ensemble (NVT), as per the following equation:
| Qst0 = −ΔH = 〈Uhg〉 − 〈Uh〉 − 〈Ug〉 − RT | (3) |
To break down the relative contributions of van der Waals and coulombic interactions to the interaction energy between MOFs and adsorbate molecules, we performed energy minimizations of a single adsorbate molecule inside the MOF structures employing the NVT ensemble. From this force field-based molecular simulation, the minimum host-adsorbate energy was obtained via Baker's method56 and used for further analysis of the van der Waals and coulombic contributions to the interaction energy. Baker's minimization56 for 100 independent minimization attempts with the stopping criteria of the RMS gradient of 1.0 × 10−6 was also considered to find the favorable adsorption sites of the adsorbates (CO2, CH4, and N2). During Baker's minimization, framework atoms were held fixed, whereas a single molecule of the adsorbate gases was relaxed within the rigid MOF structures. Two types of trial moves, namely translation and rotation with equal probability, were attempted for the adsorbate molecules. In Baker's minimization method, the equilibrium geometries of the adsorbate molecules were estimated from the force field-based molecular simulations. All simulations, such as GCMC, Widom particle insertion calculations, and energy minimizations (via Baker's method), were carried out using the RASPA2 software package.47 The binding energy (BE) of the CO2 molecule with the various functional groups of the frameworks was computed by Möller–Plesset second-order perturbation theory (MP2) with a Def2TZVP basis set. These calculations were performed using the Gaussian 16 code.57 Further computational details can be found in section 3.4.
We also simulated the adsorption isotherms considering the bulk composition of 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50 in CO2/CH4 and 15
50 in CO2/CH4 and 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 85 in CO2/N2 binary mixtures. These compositions of the binary systems represent the landfill gas separation and flue gas separation, respectively.58 Ideal adsorbed solution theory (IAST)59 was invoked to predict the adsorption selectivity of CO2/CH4 and CO2/N2 binary mixtures from their pure component adsorption isotherms. The selectivities were predicted at the above-mentioned composition at 298 K with a pressure range up to 0–1 bar.
85 in CO2/N2 binary mixtures. These compositions of the binary systems represent the landfill gas separation and flue gas separation, respectively.58 Ideal adsorbed solution theory (IAST)59 was invoked to predict the adsorption selectivity of CO2/CH4 and CO2/N2 binary mixtures from their pure component adsorption isotherms. The selectivities were predicted at the above-mentioned composition at 298 K with a pressure range up to 0–1 bar.
All the structures were assessed in view of the three adsorbent evaluation criteria for their CO2 separation capability over CH4 and N2. The adsorbent evaluation criteria are: (1) CO2 uptake under adsorption conditions (mol kg−1), Nads; (2) working capacity of CO2 (mol kg−1), ΔN = Nads − Ndes, and (3) selectivity, 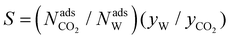 . Here, N is the adsorbed amount, y is the gas phase mol fraction, W indicates the weakly adsorbed gas component (CH4 or N2), and superscripts ads and des are the adsorption and desorption conditions, respectively. We measured these parameters for three cases as listed in Table 1 and ranked all the functional FMOFs based on their selectivity.
. Here, N is the adsorbed amount, y is the gas phase mol fraction, W indicates the weakly adsorbed gas component (CH4 or N2), and superscripts ads and des are the adsorption and desorption conditions, respectively. We measured these parameters for three cases as listed in Table 1 and ranked all the functional FMOFs based on their selectivity.
| Case | Mixture proportion | Adsorption pressure, Pads (bar) | Desorption pressure, Pdes (bar) |
|---|---|---|---|
| Natural gas purification | CO2/CH4 = 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 90 90 |
5 | 1 |
| Landfill gas separation | CO2/CH4 = 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50 50 |
1 | 0.1 |
| Flue gas separation | CO2/N2 = 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 85 85 |
1 | 0.1 |
The CO2, CH4, and N2 uptake was estimated at the partial pressure of the specific gas component under the adsorption and desorption conditions.
To confirm the reliability of the force field used in this study, we first simulated N2 adsorption in FMOF-1 at 77 K and compared it with the previously simulated data.40 We observe a similar trend of N2 adsorption but lower uptake compared to the previous simulated data. The slight variation in N2 loading could be due to the different void fractions used during simulations. However, the force field is validated by the identical shape of the isotherms depicted in Fig. S9.†
3. Results and discussion
From the geometrical properties of the structures listed in Table 2, we observe increased surface areas and pore volumes of –OCH3, –CN, –OH, and –NH2 functionalized MOFs compared to the parent FMOF-1 structure, while we notice a decreasing trend for the density. In contrast to the other functionalized MOFs, we observe a decreased surface area and pore volume for the –COOH functionalized MOF, compared with the parent FMOF-1. Likewise, a higher density of the –COOH functionalized MOF was found. We speculate that this opposite trend for the –COOH functionalized MOF is due to the size of the bulky –COOH group compared to the other functional groups. The pore size distributions (PSDs) of the MOFs are shown in Fig. 1. The channel diameter has increased to 6.93 Å in FMOF-1-CN, 6.99 Å in FMOF-1-OH, and 6.63 Å in FMOF-1-NH2 from 6.40 Å in the parent structure. On the other hand, the channel diameter has decreased to 5.55 Å and 5.31 Å, respectively, in FMOF-1-OCH3 and FMOF-1-COOH.| Structure | Density (g cm−3) | Pore volume (cm3 g−1) | He void fraction | Surface area (m2 g−1) |
|---|---|---|---|---|
| FMOF-1 | 1.666 | 0.248 | 0.413 | 826.537 |
| FMOF-1-OCH3 | 1.323 | 0.348 | 0.461 | 1215.416 |
| FMOF-1-CN | 1.330 | 0.332 | 0.442 | 1168.435 |
| FMOF-1-OH | 1.264 | 0.456 | 0.576 | 1492.484 |
| FMOF-1-COOH | 1.676 | 0.217 | 0.364 | 704.115 |
| FMOF-1-NH2 | 1.215 | 0.472 | 0.573 | 1523.289 |
N2 adsorption isotherms at 77 K for all the functionalized MOFs are depicted in Fig. S10.† All the studied MOFs display type I adsorption profiles, exhibiting characteristic microporous behavior.
3.1 Energy parameters at infinite dilution
Henry's constant, KH, and isosteric heat of adsorption at infinite dilution, Qst0, can reflect the adsorbate–adsorbent interaction strength in the Henry regime. The larger KH and Qst0 values indicate the greater adsorbate affinity to the adsorbent.60KH and Qst0 values for CO2, CH4, and N2 at 273 and 298 K were predicted from Monte Carlo (MC) simulations and the results at 298 K are listed in Tables 3 and 4, respectively. The values at 273 K are tabulated in Tables S9 and S10.†| Structure | Henry constant, KH (mol kg−1 Pa−1) | ||
|---|---|---|---|
| CO2 | CH4 | N2 | |
| FMOF-1 | 1.81 × 10−6 | 1.23 × 10−6 | 4.95 × 10−7 |
| FMOF-1-OCH3 | 4.10 × 10−6 | 2.12 × 10−6 | 7.55 × 10−7 |
| FMOF-1-CN | 5.11 × 10−6 | 1.34 × 10−6 | 6.14 × 10−7 |
| FMOF-1-OH | 3.64 × 10−5 | 1.32 × 10−6 | 7.90 × 10−7 |
| FMOF-1-COOH | 1.02 × 10−4 | 2.04 × 10−6 | 9.62 × 10−7 |
| FMOF-1-NH2 | 7.01 × 10−5 | 2.08 × 10−6 | 1.09 × 10−6 |
| Structure | Isosteric heat of adsorption at infinite dilution, Qst0 (kJ mol−1) | ||
|---|---|---|---|
| CO2 | CH4 | N2 | |
| FMOF-1 | −13.67 | −11.40 | −9.01 |
| FMOF-1-OCH3 | −16.94 | −13.18 | −10.35 |
| FMOF-1-CN | −17.75 | −11.63 | −9.56 |
| FMOF-1-OH | −29.33 | −10.44 | −9.63 |
| FMOF-1-COOH | −31.04 | −15.47 | −14.36 |
| FMOF-1-NH2 | −31.37 | −11.62 | −10.75 |
The order of KH and Qst0 was found as CO2 > CH4 > N2 for all the MOFs, implying stronger CO2–framework interactions compared to CH4–framework and N2–framework interactions. This phenomenon might be due to the larger permanent quadrupole moment of the CO2 molecule.61 According to the simulation, an increase in KH and Qst0 (except for the Qst0 value of CH4 in the –OH functionalized structure) values was noticed for all the X-functionalized MOFs compared to the parent MOF for all the adsorbates. Notably, –OH, –COOH, and –NH2 groups displayed significant improvement in the KH and Qst0 values for CO2. These enhanced values of the parameters could be described by the stronger polarity of the –OH, –COOH, and –NH2 groups compared to other functional groups. This assumption could be further supported by the results of the relative contribution of van der Waals (vdW) and coulombic interactions to the total interaction energy of CO2 in the studied MOFs, which has been discussed in section 3.3. The larger KH and Qst0 values of CO2, CH4, and N2 in all the X-functionalized MOFs compared to the parent MOF indicate greater affinity and stronger interaction strength to the frameworks.
3.2 Pure gas adsorption isotherms
We simulated the adsorption isotherms of pure component CO2, CH4, and N2 in all the X-functionalized MOFs along with the parent FMOF at 273 and 298 K with a pressure range up to 30 bar. Fig. 2 shows the CO2 uptake for all the MOF structures at 298 K in low-pressure (0.005–1 bar) and high-pressure (1–30 bar) regions. An enhanced CO2 uptake was observed for the X-functionalized MOFs compared to the parent MOF. The order of CO2 uptake up to 0.2 bar (Fig. 2a) is FMOF-1-COOH > FMOF-1-NH2 > FMOF-1-OH > FMOF-1-CN > FMOF-1-OCH3 > FMOF-1. This is consistent with the order of the predicted Henry's constant (KH values in Table 3). With the increase in pressure, a different CO2 adsorption profile was observed for the studied MOFs. In the low pressure region, gas adsorption is generally dominated by the host–guest interaction,62 as reflected in Henry's constant values. In contrast, in the high pressure region, the adsorption capacity primarily depends on the available free volume of the framework structure.62 As shown in Fig. 2, the –COOH functionalized MOF exhibits the highest CO2 uptake below 0.2 bar, while at 30 bar a comparable CO2 uptake was predicted for FMOF-1-COOH and FMOF-1. This comparable CO2 uptake is due to similar free volumes and surface areas of the structures (Table 2). At 1 bar pressure, the –OH and –NH2 functionalized structures show the maximum CO2 uptake with a value of ∼3.0 mol kg−1. This value is about 15-fold higher than that of the parent FMOF-1 (∼0.2 mol kg−1) and 1.5-fold higher than that of IRMOF-1 (2.1 mmol g−1 at 295 K),63 but lower than that of zeolite NaX and NaY structures (∼4.5 mol kg−1 at 298 K)64,65 at 1 bar. We also observe higher CO2 uptake for all the X-functionalized MOFs compared to the parent MOF in the high-pressure region. The highest CO2 uptake was observed for FMOF-1-OH with a value of 10.07 mol kg−1 at 30 bar at 298 K, which is 3.35-fold higher than that of the parent FMOF-1 with a value of 2.85 mol kg−1. A similar trend was observed for CO2 adsorption isotherms at 273 K, shown in Fig. S11.†Pure component CH4 and N2 adsorption isotherms at 298 K are depicted in Fig. 3 and 4 respectively, whereas Fig. S12 and S13† show the isotherms at 273 K. In the low pressure region (Fig. 3a), the order of CH4 uptake is FMOF-1-OCH3 > FMOF-1-NH2 > FMOF-1-COOH > FMOF-1-CN > FMOF-1-OH > FMOF-1. For N2 adsorption, the order was found as FMOF-1-NH2 > FMOF-1-COOH > FMOF-1-OCH3 > FMOF-1-OH > FMOF-1-CN > FMOF-1 (Fig. 4a). These orders of CH4 and N2 uptake are consistent with the orders of predicted Henry's constant (KH values in Table 3). In the high pressure region, a different order of CH4 and N2 adsorption was observed for the studied MOFs.
We observe an increase in CO2 uptake on a different scale than that of the increase in CH4 and N2 uptake with an increase in pressure up to 30 bar. The CO2 adsorption isotherms exhibit a sharp rise in the low pressure region compared to CH4 and N2 adsorption and then reach a plateau at around 20 bar. On the other hand, CH4 and N2 isotherms do not show a steep uptake in the low-pressure region. This expected finding is consistent with the relatively smaller Qst0 values of CH4 and N2 compared to the Qst0 value of CO2 (Table 4). CH4 and N2 uptake increases gradually with the pressure and does not saturate, even at the highest pressure of 30 bar. The maximum CH4 uptake at 1 bar was observed for FMOF-1-OCH3 (0.19 mol kg−1) which is 1.8 times higher than that of the parent FMOF-1 (0.11 mol kg−1). FMOF-1-NH2 shows the maximum N2 uptake with a value of 0.09 mol kg−1 which is 2.3 times higher compared to FMOF-1 (0.04 mol kg−1) under the same conditions (1 bar and 298 K).
3.3 Isosteric heats of adsorption, Qst
The isosteric heats of adsorption (Qst) for CO2, CH4, and N2 were obtained by GCMC simulations during the adsorption isotherm calculations. Fig. 5 and Fig. S14† display the Qst values as a function of pressure and uptake, respectively, at 298 K. It is observed that, for all the MOF structures, the Qst values of CO2 are higher compared to the values obtained for CH4 and N2. Consequently, the order of isosteric heat of adsorption is CO2 > CH4 > N2 for all the structures, consistent with the order of uptake capacity. At 0.005 bar, the isosteric heats of adsorption of CO2 were estimated as 16.92, 17.70, 29.42, 30.98, and 31.07 kJ mol−1, respectively, for –OCH3, –CN, –OH, –COOH, and –NH2 functionalized MOFs. All these Qst values are higher compared to the Qst value of CO2 for the parent FMOF-1 (13.65 kJ mol−1). The larger CO2Qst values for the X-functionalized MOFs than that of the parent MOF over the entire pressure range demonstrates the more energetic CO2 interaction with the X-functionalized MOFs. The CO2Qst values of FMOF-1-OH and FMOF-1-NH2 first decline to 29.24 and 28.15 kJ mol−1, respectively, at around 5 bar (Fig. 5a) due to the adsorbate–adsorbent interaction, i.e., the interactions of the CO2 quadrupole with the most active adsorption centers of the adsorbents play the dominant role in this region. Subsequently, we notice an increasing trend of CO2Qst with an increase in uptake or pressure, because of the increased CO2–CO2 interactions. For FMOF-1-COOH, the CO2Qst first increases up to 5 bar, reaches a value of 32.23 kJ mol−1 and then decreases with the increase in pressure or CO2 loadings. For FMOF-1-OH, FMOF-1-COOH, and FMOF-1-NH2, the presence of different energetic CO2 adsorption sites, i.e., surface heterogeneity,30 results in the maxima and minima of the Qst values of CO2 curves for these structures. For the other functionalized MOFs including the parent MOF, the relationship of Qstvs. pressure (Fig. 5a) or Qstvs. CO2 uptake (Fig. S14a†) is similar. The CO2Qst increases evenly to values of 27.69, 25.11, and 21.93 kJ mol−1 for FMOF-1-OCH3, FMOF-1-CN, and FMOF-1, respectively, up to 30 bar. The increasing behavior is due to the lateral interactions of the guest molecules with the increase in pressure (or the adsorbate concentration).66,67 | ||
| Fig. 5 GCMC simulated isosteric heats of adsorption for (a) CO2, (b) CH4, and (c) N2 at 298 K up to 30 bar. | ||
Fig. 5b and c depict the Qst values of CH4 and N2, respectively, at 298 K. For CH4, we observe a moderate variation in Qst with an increase in pressure or CH4 loading. The increase in CH4Qst values was found as 11.36 to 13.47 kJ mol−1 for FMOF-1, 13.18 to 15.97 kJ mol−1 for FMOF-1-OCH3, 11.56 to 12.96 kJ mol−1 for FMOF-1-CN, 10.43 to 11.90 kJ mol−1 for FMOF-1-OH, 15.46 to 17.42 kJ mol−1 for FMOF-1-COOH, and 11.58 to 13.74 kJ mol−1 for FMOF-1-NH2. For FMOF-1-OH, we notice lower Qst values throughout the entire pressure range compared to the parent MOF. This might be due to the larger void fraction of the –OH functionalized structure compared to the parent structure (Table 2) and the relatively smaller CH4KH value (Table 3). On the other hand, FMOF-1-CN shows a decreasing trend of Qst values compared to the parent MOF beyond 2.5 bar. This could be attributed to the similar KH values of CH4 in FMOF-1-OH and FMOF-1-CN as well as the comparable void fraction of FMOF-1-OH with FMOF-1 (0.442 vs. 0.413).
Unlike the Qst values of CO2 and CH4, the Qst value of N2 remains almost constant throughout the entire pressure range. This outcome indicates the relatively weaker interaction of the CH4 and N2 molecules with the MOF structures compared to the CO2–MOF interactions.
It is relevant to note that enhanced CO2, CH4, and N2 adsorption was observed for all the X-functionalized MOFs compared to the parent MOF, but the Qst orders of CO2, CH4, and N2 in those structures did not follow the same trend. This is because Qst is not the only factor that reflects the adsorption isotherm, rather it is one of many factors that affect the adsorption capacity and could reflect the adsorption amount to a certain extent.60 The Qst values at 273 K are depicted in Fig. S15–S17.†
From the relative contribution of van der Waals (vdW) and coulombic interactions to the total interaction energy of CO2, CH4, and N2 in the studied MOFs (Fig. 6), it is apparent that the significantly improved CO2 adsorption capacity in –OH, –COOH, and –NH2 functionalized structures is due to the enhanced coulombic interaction compared to the parent FMOF-1. On the other hand, for the N2-MOF interaction energy, the vdW interactions play the dominant role with a little coulombic contribution for those structures. In contrast, the interaction energy for CH4 was purely from the van der Waals interactions without any electrostatic contribution. This could be due to the larger quadrupole moment of CO2 (4.30 × 10−26 esu cm2) compared to the smaller quadrupole moment of N2 (1.52 × 10−26 esu cm2) and zero-quadrupole moment of CH4.61
 | ||
| Fig. 6 Interaction energy plot for (a) CO2, (b) CH4, and (c) N2 in the investigated MOFs, predicted from MC simulations. | ||
We performed additional simulations by omitting the coulombic interaction of the guest molecules with the framework atoms to investigate the effect of electrostatic interactions on CO2, CH4, and N2 adsorption at 298 K up to 1 bar (Fig. 7 and S18, 19†). Disregarding the electrostatic interaction, lower CO2 and N2 uptake was observed for all the MOF structures. In contrast, CH4 uptake was not influenced by turning off the coulombic interaction. We observe a larger variation of CO2 uptake while we consider the coulombic interaction than that of the uptake without considering the coulombic interaction. For N2 uptake, the variation was not as prominent as that of CO2. This finding is quite consistent with the results discussed above and demonstrates the importance of electrostatic interactions on enhanced CO2 adsorption in –OH, –COOH, and –NH2 functionalized structures.
3.4 Interaction of CO2 with the functional groups of the framework
The binding energy (BE) of CO2 with various functional groups of the framework was computed using Möller–Plesset second-order perturbation theory (MP2) with a triple-ξ valence basis set with polarization (Def2TZVP).68 The rationale for the selection of the Def2TZVP basis set is that it can efficiently capture the dispersion interaction.6,34,69 For BE calculations between CO2 and X-functional groups using MP2, a small model of the triazole ring containing two –CF3 or two X-substituents (at the 3,5-positions) was constructed (Fig. 8). Only a small model was constructed to reduce the computational cost. All the models (the triazole ring containing the –CF3 or X-substituents at the 3,5-positions) were optimized first using the MP2/Def2TZVP level of theory. Then geometry optimization was done for the CO2–triazole complex considering different geometries (positions and orientations) of CO2 around the functional groups. Eventually, the BE of CO2 around different functional groups was calculated as per the following equation:| BE = ECO2-triazole model − (ECO2+ Etriazole model) | (4) |
As per the optimized structures, the electropositive C atom of CO2 interacts with the electronegative N and O atoms of –CN and –COOH functional groups, while hydrogen bonding interactions were observed for –OCH3, –NH2, and –OH functional groups with distances of ∼3.50, 2.60, and 2.13 Å, respectively. The highest binding energy value was observed for the –NH2–CO2 structure (−21.09 kJ mol−1) followed by the –OH–CO2 structure (−18.10 kJ mol−1). Although the O(CO2)⋯H(–OH) distance exhibits a relatively smaller value (2.13 Å) than that of the average O(CO2)⋯H(–NH2) distance (2.60 Å), the multiple interaction between the terminal O atoms of CO2 with the H atoms of NH2 results in increased binding energy for the NH2–CO2 structure. For the –COOH–CO2 structure, we notice that the CO2 molecule is tilted towards the Ocarbonyl of the –COOH group with a distance of 3.00 Å and a binding energy of −17.97 kJ mol−1. We do not observe any hydrogen bonding interaction for the –COOH–CO2 structure, as the H(–COOH) atom is pointing away from the CO2 molecule in the optimized configuration. The –OCH3–CO2 structure shows the least BE value (−10.22 kJ mol−1) among all the structures.
The binding energies (BEs) of CO2 with different functional groups calculated using the MP2 level of theory follow the order of FMOF-1-OCH3 < FMOF-1 < FMOF-1-CN < FMOF-1-COOH < FMOF-1-OH < FMOF-1-NH2 (Fig. 8). This order shows a slight disagreement from the isosteric heat of adsorption at infinite dilution, Qst0, for CO2 predicted from force field-based molecular simulation, which follows the order of FMOF-1 < FMOF-1-OCH3 < FMOF-1-CN < FMOF-1-OH < FMOF-1-COOH < FMOF-1-NH2 (Table 4). More precisely, we observe the disagreement between FMOF-1 and FMOF-1-OCH3 and between FMOF-1-COOH and FMOF-1-OH.
For the calculations obtained by MP2, we observe a large number of interactions between the CO2 molecule and the F atoms (of –CF3 groups) in FMOF-1, compared to the number of interactions between the CO2 molecule and the H atoms (of –CH3 groups) in FMOF-1-OCH3. This results in an increased binding energy for the former. On the other hand, the smaller distance between O(CO2)⋯H(–OH) (2.132 Å) compared to the distance between C(CO2)⋯Ocarbonyl (–COOH) (3.001 Å) results in a slight increase in the binding energy value for FMOF-1-OH. This disagreement for the order of Qst0 and BE values obtained by force field-based molecular simulation and MP2/DFT calculations has also been reported by other researchers.6,31 However, we notice a significant enhancement of the Qst0 and BE values for –COOH, –OH, and –NH2 functionalized structures compared to that of the parent FMOF-1 structure, using both the force field-based molecular simulation and MP2 calculations.
The radial distribution functions (RDFs), g(r) between CO2 and various functional groups of the studied MOFs at 298 K and 0.1 bar are displayed in Fig. 9. For the parent FMOF-1, the g(r) value of O(CO2) around F(–CF3) is essentially zero with a distance r < 2.50 Å and exhibits a lower peak at ∼6.38 Å. On the other hand, a pronounced peak between O(CO2) and H(–OCH3) was observed at a distance between 3.42 and 3.78 Å for FMOF-1-OCH3, whereas a sharp peak between C(CO2) and N(–CN) was found at r = 3.54 Å for FMOF-1-CN. Unlike the other MOFs, FMOF-1-OH, FMOF-1-COOH, and FMOF-1-NH2 exhibit multiple peaks in g(r) curves. The g(r) between the O(CO2) and H(–OH) for FMOF-1-OH shows two distinct peaks at 2.94 and 7.54 Å. For FMOF-1-COOH, we observe three dominant peaks between C(CO2) and Ocarbonyl (–COOH) at nearly 3.00, 7.89, and 9.42 Å. Likewise, three major peaks between C(CO2) and H(–NH2) were found at nearly 2.66, 6.58, and 8.43 Å for FMOF-1-NH2. The presence of multiple peaks in the g(r) curves suggests multiple CO2 interactions with neighboring ligands’ X substituents in FMOF alternatives.6 This structural analysis reveals that CO2 molecules are preferentially adsorbed to X groups but at different distances, depending on the interaction strengths of CO2 with the new MOF structure.
 | ||
| Fig. 9 Radial distribution functions for CO2 near different functional groups of (a) FMOF-1, (b) FMOF-1-OCH3, (c) FMOF-1-CN, (d) FMOF-1-OH, (e) FMOF-1-COOH, and (f) FMOF-1-NH2 at 0.1 bar. | ||
We also performed Baker's minimization by inserting a single adsorbate molecule in the X-functionalized MOFs at 298 K in order to locate the favorable adsorption sites of the adsorbates. Baker's algorithm56 uses the eigenvalues of the Hessian matrix in order to locate true minima on the energy surface that corresponds to the equilibrium geometries. The positions of CO2 (Fig. 10) and CH4 & N2 (Fig. S20†) represent the preferred binding sites of the adsorbates in MOFs, predicted by Monte Carlo (MC) simulations.
 | ||
| Fig. 10 Adsorption sites of CO2 in (a) –OCH3, (b) –CN, (c) –OH, (d) –COOH, and (e) –NH2 functionalized MOFs after Baker's minimization. | ||
We also compared the equilibrium geometries of the adsorbate molecules after Baker's minimization, estimated from the force field-based molecular simulations with MP2 calculations. In MP2 calculations, we investigated the probable adsorption sites of CO2 by studying their dispersion interaction energies using a small model. Only the triazole ring containing two –CF3 or two X-substituents at the 3,5-positions was used in the molecular model for the study of the interaction energy of CO2 (Fig. 8). As per the MP2 calculation, CO2 molecules seem to interact with the –CF3 group for parent FMOF-1 and X-functional groups for X-functionalized MOF structures.
On the other hand, the equilibrium geometries of CO2 molecules after Baker's minimization estimated from the force field-based molecular simulations are shown in Fig. 10. According to Fig. 10, CO2 molecules are preferentially adsorbed to X-functional groups rather than the metal cluster sites. This finding is fairly consistent with the adsorption sites calculated from MP2. Still, there is a slight discrepancy between the binding distances of CO2 with X-functional groups, computed from force field-based molecular simulations and the MP2 model. For example, the distances between O(CO2)⋯H(–OCH3) in FMOF-1-OCH3 were 3.503 and 3.601 Å, respectively, obtained from the MP2 model and force field-based molecular simulations. This little variation could be qualitatively explained by comparing the two models. In the MP2 model, we considered a small segment of the framework, where the influence of neighboring linkers on CO2 adsorption had been ignored. In contrast, we considered the entire framework during the force field-based molecular simulations (Baker's minimization) to predict the CO2 adsorption sites, accounting for the influence of neighboring linkers on CO2 adsorption.
3.5 Adsorption of binary mixtures and their selectivity
We simulated the adsorption isotherms of CO2/CH4 and CO2/N2 gas mixtures with the composition of 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50 and 15
50 and 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 85 molar ratios, respectively, to study the carbon capture capability from landfill and flue gases. Fig. 11 shows the adsorption isotherms of the binary mixtures for all the MOFs at 298 K up to a pressure of 1 bar. CO2, CH4, and N2 adsorption exhibits a nearly linear isotherm with an increase in pressure except for CO2 adsorption in the –COOH functionalized MOF. We assume this non-linearity is due to the relatively higher pore volume of the other structures than that of the FMOF-1-COOH structure. We also notice preferential adsorption of CO2 over CH4 in all the MOFs for the CO2/CH4 mixture. Higher CO2 uptake was observed for the X-functionalized MOFs compared to the parent FMOF-1 structure. Especially for FMOF-1-NH2, dominant CO2 uptake was observed over CH4 due to the enhanced electrostatic interaction in addition to the dispersion interactions. In the case of the CO2/N2 gas mixture, the parent FMOF-1 displays higher N2 uptake over CO2 throughout the entire pressure range due to the higher mole fraction of N2 in the CO2/N2 binary mixture. On the other hand, for the FMOF-1-OCH3 and FMOF-1-CN structures, we notice lower N2 uptake over CO2, while for the remaining structures, vanishingly smaller N2 uptake was observed compared to CO2.
85 molar ratios, respectively, to study the carbon capture capability from landfill and flue gases. Fig. 11 shows the adsorption isotherms of the binary mixtures for all the MOFs at 298 K up to a pressure of 1 bar. CO2, CH4, and N2 adsorption exhibits a nearly linear isotherm with an increase in pressure except for CO2 adsorption in the –COOH functionalized MOF. We assume this non-linearity is due to the relatively higher pore volume of the other structures than that of the FMOF-1-COOH structure. We also notice preferential adsorption of CO2 over CH4 in all the MOFs for the CO2/CH4 mixture. Higher CO2 uptake was observed for the X-functionalized MOFs compared to the parent FMOF-1 structure. Especially for FMOF-1-NH2, dominant CO2 uptake was observed over CH4 due to the enhanced electrostatic interaction in addition to the dispersion interactions. In the case of the CO2/N2 gas mixture, the parent FMOF-1 displays higher N2 uptake over CO2 throughout the entire pressure range due to the higher mole fraction of N2 in the CO2/N2 binary mixture. On the other hand, for the FMOF-1-OCH3 and FMOF-1-CN structures, we notice lower N2 uptake over CO2, while for the remaining structures, vanishingly smaller N2 uptake was observed compared to CO2.
IAST was applied to calculate the adsorption selectivity of the MOF structures for the CO2/CH4 (50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50) and CO2/N2 (15
50) and CO2/N2 (15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 85) binary gas mixtures at 298 K (Fig. 12). The selectivity values exhibit a similar trend for both the mixtures. All the X-functionalized structures exhibit higher selectivity values than that of the parent MOF structure. This could be attributed to the relatively stronger CO2 interaction with the X-functionalized MOFs.6 It is apparent that –OH, –COOH, and –NH2 functionalized MOFs significantly enhance the CO2/CH4 and CO2/N2 selectivities compared to the original FMOF-1. For the CO2/CH4 gas mixture, the selectivity values were obtained as 59.6, 40.0, and 31.5, respectively, for the –COOH, –NH2, and –OH functionalized MOFs at a pressure of 0.1 bar. These values are much higher compared to the values obtained from –COOH, –NH2, and –OH substituted MIL-53 (lp), where the selectivity values are in the range of 12–17 at 0.1 bar.29 For the CO2/N2 gas mixture, FMOF-1-COOH exhibits the highest selectivity of 128.7 at 0.1 bar, which is higher than that of the dihydrofuran functional porous aromatic framework, DHF_PAF-1, having a value of 92.0 at infinite dilution.6 For both the CO2/CH4 and CO2/N2 gas mixtures, we notice that the selectivity values remain almost constant throughout the entire pressure range for FMOF-1, FMOF-1-OCH3, and FMOF-1-CN structures. On the other hand, the selectivity gradually increases with an increase in pressure for FMOF-1-OH, FMOF-1-NH2, and FMOF-1-COOH structures due to the amplified CO2–CO2 interactions at higher pressures.70 We also compared the selectivity values of the CO2/CH4 and CO2/N2 gas mixtures, derived from the corresponding Henry's constant ratios with the values obtained by the IAST method, at 0.1 bar and 298 K. The details of this comparison can be found in the ESI.†
85) binary gas mixtures at 298 K (Fig. 12). The selectivity values exhibit a similar trend for both the mixtures. All the X-functionalized structures exhibit higher selectivity values than that of the parent MOF structure. This could be attributed to the relatively stronger CO2 interaction with the X-functionalized MOFs.6 It is apparent that –OH, –COOH, and –NH2 functionalized MOFs significantly enhance the CO2/CH4 and CO2/N2 selectivities compared to the original FMOF-1. For the CO2/CH4 gas mixture, the selectivity values were obtained as 59.6, 40.0, and 31.5, respectively, for the –COOH, –NH2, and –OH functionalized MOFs at a pressure of 0.1 bar. These values are much higher compared to the values obtained from –COOH, –NH2, and –OH substituted MIL-53 (lp), where the selectivity values are in the range of 12–17 at 0.1 bar.29 For the CO2/N2 gas mixture, FMOF-1-COOH exhibits the highest selectivity of 128.7 at 0.1 bar, which is higher than that of the dihydrofuran functional porous aromatic framework, DHF_PAF-1, having a value of 92.0 at infinite dilution.6 For both the CO2/CH4 and CO2/N2 gas mixtures, we notice that the selectivity values remain almost constant throughout the entire pressure range for FMOF-1, FMOF-1-OCH3, and FMOF-1-CN structures. On the other hand, the selectivity gradually increases with an increase in pressure for FMOF-1-OH, FMOF-1-NH2, and FMOF-1-COOH structures due to the amplified CO2–CO2 interactions at higher pressures.70 We also compared the selectivity values of the CO2/CH4 and CO2/N2 gas mixtures, derived from the corresponding Henry's constant ratios with the values obtained by the IAST method, at 0.1 bar and 298 K. The details of this comparison can be found in the ESI.†
 | ||
| Fig. 12 Adsorption selectivity for (a) CO2/CH4 and (b) CO2/N2 binary gas mixtures at 298 K, up to 1 bar, as obtained by the IAST method. | ||
3.6 Evaluation of the FMOFs for CO2 separation
According to the adsorption evaluation criteria discussed earlier (section 2.3), we assessed the potential of all the MOFs for CO2 separation and capture. Tables 5 and 6 rank the MOFs for natural gas purification and landfill gas separation, respectively, in terms of their selectivity values.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 90)
90)
| Structure | N ads (mol kg−1) | ΔN (mol kg−1) | S |
|---|---|---|---|
| FMOF-1-OH | 1.63 | 1.26 | 30.06 |
| FMOF-1-NH2 | 2.01 | 1.41 | 23.83 |
| FMOF-1-COOH | 1.74 | 1.13 | 22.66 |
| FMOF-1-CN | 0.25 | 0.20 | 4.67 |
| FMOF-OCH3 | 0.21 | 0.17 | 2.40 |
| FMOF-1 | 0.10 | 0.08 | 1.94 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50)
50)
| Structure | N ads (mol kg−1) | ΔN (mol kg−1) | S |
|---|---|---|---|
| FMOF-1-OH | 1.63 | 1.44 | 28.28 |
| FMOF-1-NH2 | 2.01 | 1.68 | 21.51 |
| FMOF-1-COOH | 1.74 | 1.33 | 18.42 |
| FMOF-1-CN | 0.25 | 0.22 | 4.24 |
| FMOF-OCH3 | 0.21 | 0.19 | 2.10 |
| FMOF-1 | 0.10 | 0.08 | 1.75 |
For natural gas purification and landfill gas separation, the MOF structures follow the order of FMOF-1-OH > FMOF-1-NH2 > FMOF-1-COOH > FMOF-1-CN > FMOF-1-OCH3 > FMOF-1, obtained from the selectivity calculation as observed in Tables 5 and 6. Apparently, the selectivity values for the –OH functionalized MOF are 15.50 and 16.16-fold larger than that of the parent FMOF-1 structure for natural gas purification and landfill gas separation, respectively. As shown in Table 5, FMOF-1-OH, FMOF-1-NH2, and FMOF-1-COOH show relatively high selectivity values (30.06, 23.83, and 22.66) compared to the remaining MOF structures. These values are higher than those of many materials for natural gas purification such as zeolite-5A (20.0), zeolite-13X (18.9), Mg-MOF-74 (17.3), and amine-MIL-53(Al) (16.7).71 For landfill gas separation, the largest selectivity value was observed for the –OH functionalized MOF (28.28). This value is comparable to NaY (30.0), but smaller than that of NaX (40.0).72 Again, the selectivity values of –OH, –NH2, and –COOH functionalized MOFs (28.28, 21.51, and 18.42) are higher than those of many MOFs, for example, Mg-MOF-74 (12.5), zeolite-13X (13.2), and zeolite-5A (23.5).71,73 The presence of highly polar –OH, –NH2, and –COOH functional groups in MOFs leads to the higher adsorption of quadrupolar CO2 than that of nonpolar CH4 at the specified adsorption pressure, eventually resulting in larger selectivity values for the structures. Though FMOF-1-OH shows the largest selectivity value among all the structures, FMOF-1-NH2 seems to be the best adsorbent with respect to Nads and working capacity (ΔN) performance. We speculate these higher Nads and ΔN values are due to the relatively larger pore volume of FMOF-1-NH2 than those of the other structures.
For flue gas separation, the MOF structures follow a different order of performance (Table 7) compared to the above two cases, in terms of selectivity. FMOF-1-COOH shows the highest selectivity value (71.70) among all the MOFs, which is larger than those of Ni-MOF-74 (41.1) and zeolite-5A (61.8),71 but smaller than that of NaY (500).72 For flue gas separation, a strong CO2–MOF interaction is desired in the low-pressure region, usually at 0.15 bar, which will lead to very high CO2 uptake. CO2 shows the strongest interaction with FMOF-1-COOH as indicated by the largest CO2KH value (Table 3). This results in the highest CO2 uptake for the –COOH functionalized MOF at the stated adsorption pressure, 0.15 bar (Table 7). Nevertheless, FMOF-1-COOH exhibits relatively smaller N2 uptake (0.072 mol kg−1) compared to FMOF-1-NH2 (0.075 mol kg−1) at 0.15 bar. Consequently, we notice the largest selectivity for FMOF-1-COOH among all the evaluated MOFs for flue gas separation.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) N2 = 15
N2 = 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 85 binary mixture
85 binary mixture
| Structure | N ads (mol kg−1) | ΔN (mol kg−1) | S |
|---|---|---|---|
| FMOF-1-COOH | 0.92 | 0.78 | 71.70 |
| FMOF-1-NH2 | 0.84 | 0.73 | 63.42 |
| FMOF-1-OH | 0.37 | 0.33 | 38.32 |
| FMOF-1-CN | 0.08 | 0.07 | 10.51 |
| FMOF-OCH3 | 0.06 | 0.05 | 6.68 |
| FMOF-1 | 0.03 | 0.02 | 4.95 |
4. Conclusions
In this work, we have systematically investigated the effect of several de-fluorinating functional groups (X = –OCH3, –CN, –OH, –COOH, and –NH2) on gas adsorption (CO2, CH4, and N2) vs. the parent FMOF-1 (X = –CF3) up to a pressure of 30 bar. Henry's constant (KH) and isosteric heat of adsorption at infinite dilution (Qst0) for CO2 predicted via force field-based MC simulations reveal enhanced interactions between CO2 and the X-functionalized de-fluorinated MOFs. Greater CO2 uptake in the low-pressure region (<1 bar) is predicted for –OH, –COOH, and –NH2 functionalized structures compared to the –OCH3 and –CN functionalized structures. This finding suggests that the incorporation of polar functional groups into the linkers, containing lone pairs of electrons or acidic hydrogens, can significantly enhance the CO2 adsorption capacity of MOFs. However, in the high-pressure region, the adsorption properties of the MOFs are predominantly controlled by the structural properties of the materials, such as the free volume and the surface area, rather than the nature (polar or nonpolar) of the functional groups. This was verified by comparing all the gas uptake values of the MOF structures at 30 bar. The interaction energy values between CO2 and –COOH/–OH/–NH2 functionalized structures obtained by MP2 are qualitatively similar to that of the force field-based molecular simulation values. These interaction energies along with the radial distribution function of CO2 around different functional groups provide information about the binding sites of CO2 in the MOF structures investigated herein. The adsorption behavior of the multicomponent mixtures was predicted from the CO2/CH4 and CO2/N2 binary systems, using GCMC simulations. FMOF-1-OH is predicted to show the highest selectivity for CO2/CH4, while the highest CO2/N2 adsorption selectivity was predicted for FMOF-1-COOH at room temperature. The adsorbent evaluation criteria data obtained for CO2 separation indicate that –OH, –COOH, and –NH2 functionalized MOFs could be a promising class of porous materials for selective CO2 capture. These findings represent a predictive step for the experimental development of new materials towards enhancement of CO2 separation capability, which is particularly needed for various technologies. A complementary investigation aimed at sustaining the super-hydrophobicity of FMOFs via –CH3 instead of –CF3 groups, hence reducing the water interference (i.e., CO2/H2O and CH4/H2O selectivity instead of CO2/CH4 and CO2/N2 selectivity herein), is described elsewhere in a sister article planned to be published back-to-back with this one.Data availability
The data supporting this article have been included as part of the ESI.†Conflicts of interest
The authors report no conflict of interest of any kind.Acknowledgements
M. A. O. acknowledges support from the U.S. Nuclear Regulatory Commission (award 31310023M0019), the U.S. Department of Defense (award W911NF2210201), the Welch Foundation (B-1542), and the U.S. National Science Foundation (CHE-1413641 and an international supplement thereof, CHE-1545934). J. D. acknowledges the support of his group's contribution from the U.S. National Science Foundation (CMMI-1662288) and in part by the Advanced Research Projects Agency-Energy (ARPA-E) of the U.S. Department of Energy (DE AR0001613). The computational facilities have been supported by the National Science Foundation (CHE-1531468 and OAC-2117247) and the University of North Texas (Research Computing Services and Texas Advanced Computing Center). We also thank Prof. Randall Q. Snurr of Northwestern University, USA and Prof. Peyman Z. Moghadam of University College London, UK for providing access to the RASPA2 program (the latter also included initial training and troubleshooting).References
- R. Krishna, J. Phys. Chem. C, 2009, 113, 19756–19781 CrossRef CAS.
- R. Krishna and J. M. van Baten, J. Membr. Sci., 2010, 360, 323–333 CrossRef CAS.
- L. Kong, R. Zou, W. Bi, R. Zhong, W. Mu, J. Liu, R. P. S. Han and R. Zou, J. Mater. Chem. A, 2014, 2, 17771–17778 RSC.
- J. McEwen, J.-D. Hayman and A. Özgür Yazaydin, Chem. Phys., 2013, 412, 72–76 CrossRef CAS.
- C. Janiak and J. K. Vieth, New J. Chem., 2010, 34, 2366 RSC.
- R. Babarao, S. Dai and D. Jiang, Langmuir, 2011, 27, 3451–3460 CrossRef CAS PubMed.
- J.-R. Li, J. Sculley and H.-C. Zhou, Chem. Rev., 2012, 112, 869–932 CrossRef CAS PubMed.
- K. J. Hartlieb, J. M. Holcroft, P. Z. Moghadam, N. A. Vermeulen, M. M. Algaradah, M. S. Nassar, Y. Y. Botros, R. Q. Snurr and J. F. Stoddart, J. Am. Chem. Soc., 2016, 138, 2292–2301 CrossRef CAS PubMed.
- J. Hu, Y. Liu, J. Liu and C. Gu, AIChE J., 2020, 66, e16835 CrossRef CAS.
- Z. Zhang, Z.-Z. Yao, S. Xiang and B. Chen, Energy Environ. Sci., 2014, 7, 2868 RSC.
- T. Düren, L. Sarkisov, O. M. Yaghi and R. Q. Snurr, Langmuir, 2004, 20, 2683–2689 CrossRef PubMed.
- J.-R. Li, R. J. Kuppler and H.-C. Zhou, Chem. Soc. Rev., 2009, 38, 1477 RSC.
- R. J. Kuppler, D. J. Timmons, Q.-R. Fang, J.-R. Li, T. A. Makal, M. D. Young, D. Yuan, D. Zhao, W. Zhuang and H.-C. Zhou, Coord. Chem. Rev., 2009, 253, 3042–3066 CrossRef CAS.
- V. Finsy, S. Calero, E. García-Pérez, P. J. Merkling, G. Vedts, D. E. De Vos, G. V. Baron and J. F. M. Denayer, Phys. Chem. Chem. Phys., 2009, 11, 3515 RSC.
- D. M. D'Alessandro, B. Smit and J. R. Long, Angew. Chem., Int. Ed., 2010, 49, 6058–6082 CrossRef PubMed.
- X.-J. Wang, P.-Z. Li, Y. Chen, Q. Zhang, H. Zhang, X. X. Chan, R. Ganguly, Y. Li, J. Jiang and Y. Zhao, Sci. Rep., 2013, 3, 1149 CrossRef PubMed.
- X. Liu, M. Park, S. Hong, M. Oh, J. W. Yoon, J.-S. Chang and M. S. Lah, Inorg. Chem., 2009, 48, 11507–11509 CrossRef CAS PubMed.
- S. Li, Y. G. Chung, C. M. Simon and R. Q. Snurr, J. Phys. Chem. Lett., 2017, 8, 6135–6141 CrossRef CAS PubMed.
- M. Eddaoudi, J. Kim, N. Rosi, D. Vodak, J. Wachter, M. O'Keeffe and O. M. Yaghi, Science, 2002, 295, 469–472 CrossRef CAS PubMed.
- Y. H. Jhon, M. Cho, H. R. Jeon, I. Park, R. Chang, J. L. C. Rowsell and J. Kim, J. Phys. Chem. C, 2007, 111, 16618–16625 CrossRef CAS.
- T. Sagara, J. Klassen, J. Ortony and E. Ganz, J. Chem. Phys., 2005, 123, 014701 CrossRef PubMed.
- J. R. Karra and K. S. Walton, J. Phys. Chem. C, 2010, 114, 15735–15740 CrossRef CAS.
- K. K. Tanabe and S. M. Cohen, Chem. Soc. Rev., 2011, 40, 498–519 RSC.
- J. Hu, Y. Liu, J. Liu and C. Gu, Fuel, 2017, 200, 244–251 CrossRef CAS.
- B. Arstad, H. Fjellvåg, K. O. Kongshaug, O. Swang and R. Blom, Adsorption, 2008, 14, 755–762 CrossRef CAS.
- J. An, S. J. Geib and N. L. Rosi, J. Am. Chem. Soc., 2010, 132, 38–39 CrossRef CAS PubMed.
- S. Couck, J. F. M. Denayer, G. V. Baron, T. Rémy, J. Gascon and F. Kapteijn, J. Am. Chem. Soc., 2009, 131, 6326–6327 CrossRef CAS PubMed.
- B. Zheng, J. Bai, J. Duan, L. Wojtas and M. J. Zaworotko, J. Am. Chem. Soc., 2011, 133, 748–751 CrossRef CAS PubMed.
- A. Torrisi, R. G. Bell and C. Mellot-Draznieks, Cryst. Growth Des., 2010, 10, 2839–2841 CrossRef CAS.
- A. Torrisi, R. G. Bell and C. Mellot-Draznieks, Microporous Mesoporous Mater., 2013, 168, 225–238 CrossRef CAS.
- C. Gu, Y. Liu, W. Wang, J. Liu and J. Hu, Front. Chem. Sci. Eng., 2021, 15, 437–449 CrossRef CAS.
- A. Torrisi, C. Mellot-Draznieks and R. G. Bell, J. Chem. Phys., 2009, 130, 194703 CrossRef PubMed.
- K. D. Vogiatzis, A. Mavrandonakis, W. Klopper and G. E. Froudakis, ChemPhysChem, 2009, 10, 374–383 CrossRef CAS PubMed.
- A. Torrisi, C. Mellot-Draznieks and R. G. Bell, J. Chem. Phys., 2010, 132, 044705 CrossRef PubMed.
- T. Düren, Y.-S. Bae and R. Q. Snurr, Chem. Soc. Rev., 2009, 38, 1237 RSC.
- D. H. Jung, D. Kim, T. B. Lee, S. B. Choi, J. H. Yoon, J. Kim, K. Choi and S.-H. Choi, J. Phys. Chem. B, 2006, 110, 22987–22990 CrossRef CAS PubMed.
- A. Martín-Calvo, E. García-Pérez, J. M. Castillo and S. Calero, Phys. Chem. Chem. Phys., 2008, 10, 7085 RSC.
- T. Sagara, J. Klassen and E. Ganz, J. Chem. Phys., 2004, 121, 12543–12547 CrossRef CAS PubMed.
- C. Yang, X. Wang and M. A. Omary, J. Am. Chem. Soc., 2007, 129, 15454–15455 CrossRef CAS PubMed.
- P. Z. Moghadam, J. F. Ivy, R. K. Arvapally, A. M. dos Santos, J. C. Pearson, L. Zhang, E. Tylianakis, P. Ghosh, I. W. H. Oswald, U. Kaipa, X. Wang, A. K. Wilson, R. Q. Snurr and M. A. Omary, Chem. Sci., 2017, 8, 3989–4000 RSC.
- Material Studio Modeling Environment, Release 6, Accelrys Software Inc., San Diego, California, USA, 2011 Search PubMed.
- D. A. Gomez-Gualdron, O. V. Gutov, V. Krungleviciute, B. Borah, J. E. Mondloch, J. T. Hupp, T. Yildirim, O. K. Farha and R. Q. Snurr, Chem. Mater., 2014, 26, 5632–5639 CrossRef CAS.
- O. K. Farha, A. Özgür Yazaydın, I. Eryazici, C. D. Malliakas, B. G. Hauser, M. G. Kanatzidis, S. T. Nguyen, R. Q. Snurr and J. T. Hupp, Nat. Chem., 2010, 2, 944–948 CrossRef CAS PubMed.
- O. V. Gutov, W. Bury, D. A. Gomez-Gualdron, V. Krungleviciute, D. Fairen-Jimenez, J. E. Mondloch, A. A. Sarjeant, S. S. Al-Juaid, R. Q. Snurr, J. T. Hupp, T. Yildirim and O. K. Farha, Chem. – Eur. J., 2014, 20, 12389–12393 CrossRef CAS PubMed.
- A. K. Rappe, C. J. Casewit, K. S. Colwell, W. A. Goddard and W. M. Skiff, J. Am. Chem. Soc., 1992, 114, 10024–10035 CrossRef CAS.
- T. F. Willems, C. H. Rycroft, M. Kazi, J. C. Meza and M. Haranczyk, Microporous Mesoporous Mater., 2012, 149, 134–141 CrossRef CAS.
- D. Dubbeldam, S. Calero, D. E. Ellis and R. Q. Snurr, Mol. Simul., 2016, 42, 81–101 CrossRef CAS.
- M. Sedighi, M. R. Talaie, H. Sabzyan, S. Aghamiri and P. Chen, Fuel, 2022, 308, 121965 CrossRef CAS.
- K. Sladekova, C. Campbell, C. Grant, A. J. Fletcher, J. R. B. Gomes and M. Jorge, Adsorption, 2020, 26, 663–685 CrossRef CAS.
- P. P. Ewald, Ann. Phys., 1921, 369, 253–287 CrossRef.
- J. J. Potoff and J. I. Siepmann, AIChE J., 2001, 47, 1676–1682 CrossRef CAS.
- M. G. Martin and J. I. Siepmann, J. Phys. Chem. B, 1998, 102, 2569–2577 CrossRef CAS.
- D. Frenkel and B. Smit, Understanding Molecular Simulation: From algorithm to Applications, Elsevier Inc., 2023 Search PubMed.
- B. Widom, J. Chem. Phys., 1963, 39, 2808–2812 CrossRef CAS.
- T. J. H. Vlugt, E. García-Pérez, D. Dubbeldam, S. Ban and S. Calero, J. Chem. Theory Comput., 2008, 4, 1107–1118 CrossRef CAS PubMed.
- J. Baker, J. Comput. Chem., 1986, 7, 385–395 CrossRef CAS.
- M. J. Frisch, G. W. Trucks, H. B. Schlegel, G. E. Scuseria, M. A. Robb, J. R. Cheeseman, G. Scalmani, V. Barone, G. A. Petersson, H. Nakatsuji, X. Li, M. Caricato, A. V. Marenich, J. Bloino, B. G. Janesko, R. Gomperts, B. Mennucci, H. P. Hratchian, J. V. Ortiz, A. F. Izmaylov, J. L. Sonnenberg, D. Williams-Young, F. Ding, F. Lipparini, F. Egidi, J. Goings, B. Peng, A. Petrone, T. Henderson, D. Ranasinghe, V. G. Zakrzewski, J. Gao, N. Rega, G. Zheng, W. Liang, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, T. Vreven, K. Throssell, J. A. Montgomery Jr., J. E. Peralta, F. Ogliaro, M. J. Bearpark, J. J. Heyd, E. N. Brothers, K. N. Kudin, V. N. Staroverov, T. A. Keith, R. Kobayashi, J. Normand, K. Raghavachari, A. P. Rendell, J. C. Burant, S. S. Iyengar, J. Tomasi, M. Cossi, J. M. Millam, M. Klene, C. Adamo, R. Cammi, J. W. Ochterski, R. L. Martin, K. Morokuma, O. Farkas, J. B. Foresman and D. J. Fox, Gaussian 16, Revision C.01, Gaussian, Inc., Wallingford CT, 2016 Search PubMed.
- C. Altintas, G. Avci, H. Daglar, A. Nemati Vesali Azar, S. Velioglu, I. Erucar and S. Keskin, ACS Appl. Mater. Interfaces, 2018, 10, 17257–17268 CrossRef CAS PubMed.
- A. L. Myers and J. M. Prausnitz, AIChE J., 1965, 11, 121–127 Search PubMed.
- L. Qu, Z. Wang and L. Liu, Fire, 2023, 6, 355 CrossRef.
- P. Chowdhury, S. Mekala, F. Dreisbach and S. Gumma, Microporous Mesoporous Mater., 2012, 152, 246–252 CrossRef CAS.
- Z. Cai, C. E. Bien, Q. Liu and C. R. Wade, Chem. Mater., 2020, 32, 4257–4264 CrossRef CAS.
- S. R. Caskey, A. G. Wong-Foy and A. J. Matzger, J. Am. Chem. Soc., 2008, 130, 10870–10871 CrossRef CAS PubMed.
- S. Cavenati, C. A. Grande and A. E. Rodrigues, J. Chem. Eng. Data, 2004, 49, 1095–1101 Search PubMed.
- K. S. Walton, M. B. Abney and M. D. LeVan, Microporous Mesoporous Mater., 2006, 91, 78–84 CrossRef CAS.
- S. M. S. Islam, R. Yasmeen, G. Verma, S. M. Tekarli, V. N. Nesterov, S. Ma and M. A. Omary, Inorg. Chem., 2024, 63, 8664–8673 CrossRef CAS PubMed.
- H. W. B. Teo, A. Chakraborty and S. Kayal, Appl. Therm. Eng., 2017, 110, 891–900 CrossRef CAS.
- R. Ahlrichs, M. Bär, M. Häser, H. Horn and C. Kölmel, Chem. Phys. Lett., 1989, 162, 165–169 CrossRef CAS.
- Z. Xiang, S. Leng and D. Cao, J. Phys. Chem. C, 2012, 116, 10573–10579 CrossRef CAS.
- Y. Chen and J. Jiang, ChemSusChem, 2010, 3, 982–988 CrossRef CAS PubMed.
- Y. Bae and R. Q. Snurr, Angew. Chem., Int. Ed., 2011, 50, 11586–11596 CrossRef CAS PubMed.
- R. Krishna and J. M. van Baten, Phys. Chem. Chem. Phys., 2011, 13, 10593 RSC.
- D. Saha, Z. Bao, F. Jia and S. Deng, Environ. Sci. Technol., 2010, 44, 1820–1826 CrossRef CAS PubMed.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: https://doi.org/10.1039/d4dt03093a |
| This journal is © The Royal Society of Chemistry 2025 |







